Abdominal Aortic Aneurysm (AAA) in Adults
An Abdominal Aortic Aneurysm (AAA) is defined as a permanent, localised dilatation of the abdominal aorta having a diameter greater than 3.0 cm or exceeding the normal diameter by more than 50%. It is a critical...
What matters first
An Abdominal Aortic Aneurysm (AAA) is defined as a permanent, localised dilatation of the abdominal aorta having a diameter greater than 3.0 cm or exceeding the normal diameter by more than 50%. It is a critical...
Severe, sudden-onset abdominal or back pain (rupture)
2 Jan 2026
Generated educational material; verify before clinical use.
Visible references section
See the concept before reading it
Study the key anatomy, imaging, and decision pathways as full teaching plates.
Clinical board
A visual summary of the highest-yield teaching signals on this page.
Urgent signals
Safety-critical features pulled from the topic metadata.
- Severe, sudden-onset abdominal or back pain (rupture)
- Hypotension/syncope with abdominal pain (rupture)
- Pulsatile abdominal mass with tenderness (imminent rupture)
- Flank ecchymosis (Grey Turner's sign - retroperitoneal bleed)
Linked comparisons
Differentials and adjacent topics worth opening next.
- Renal Colic
- Diverticulitis
Content status and exam context
This page is AI-generated educational content. It may contain errors or omissions and is not a substitute for current guidelines, local protocols, senior clinical judgement, or professional medical advice.
MedVellum does not claim an individual clinician reviewer, board certification, or professional credential for this page unless a future version names a real, verifiable reviewer.
Clinical explanation and evidence
Abdominal Aortic Aneurysm (AAA) in Adults
1. Clinical Overview
Summary
An Abdominal Aortic Aneurysm (AAA) is defined as a permanent, localised dilatation of the abdominal aorta having a diameter greater than 3.0 cm or exceeding the normal diameter by more than 50%. It is a critical vascular pathology that predominantly affects older men, with smoking being the single most significant modifiable risk factor. The condition is often asymptomatic ("silent killer") until rupture occurs, which carries a catastrophic mortality rate exceeding 80% overall. Clinical management hinges on surveillance for small aneurysms to monitor expansion rates, and timely elective repair (Endovascular Aneurysm Repair [EVAR] or Open Surgical Repair [OSR]) for large aneurysms (≥5.5 cm in men, ≥5.0 cm in women) to prevent rupture. The key clinical message is "Prevention of Rupture," achieved through population screening, aggressive cardiovascular risk factor modification, and adherence to size-threshold guidelines for repair.
Key Facts
- Definition: Permanent dilation of the abdominal aorta ≥3.0 cm (or > 1.5x normal diameter).
- Prevalence: 1.3-2.2% in men aged 65-74; much lower in women (~0.1%). [1,2]
- Incidence: Declining in developed nations due to reduced smoking rates, approx. 20-30 per 100,000 for ruptured AAA. [2]
- Mortality: > 80% for ruptured AAA (pre-hospital + hospital); less than 2% for elective EVAR. [1]
- Morbidity: Rupture, distal embolisation, aortic thrombosis, compression of adjacent structures (IVC, ureters).
- Peak Demographics: Men > 65 years, smokers, Caucasians. Male:Female ratio approx. 4:1 to 6:1. [3]
- Pathognomonic Feature: Expansile (not just pulsatile) abdominal mass located supra-umbilically.
- Gold Standard Investigation: CT Angiography (CTA) Aorta for procedural planning; Ultrasound for screening/surveillance.
- First-line Treatment: Surveillance for less than 5.5cm; Elective repair (EVAR or Open) for large/symptomatic aneurysms.
- Prognosis Summary: Excellent if repaired electively; extremely poor if ruptured.
Clinical Pearls
Diagnostic Pearl: The "Triad of Rupture" (hypotension, abdominal/back pain, pulsatile mass) is only present in ~50% of ruptured cases. Do not rely on it to rule out rupture.
Examination Pearl: An expansile mass moves your hands outward (laterally) as well as upward. A transmitted pulsation (e.g., from a tumor overlying the aorta) only moves hands upward.
Treatment Pearl: Permissive hypotension (systolic BP 70-90 mmHg) is critical in suspected rupture to prevent "popping the clot" before proximal control is obtained in theatre.
Pitfall Warning: Misdiagnosing a ruptured AAA as "Renal Colic" is a classic fatal error. In any patient > 60 with "renal colic," exclude AAA.
Mnemonic: 3-4-5 Rule for Surveillance (NICE/ESVS):
- 3.0 - 4.4 cm: Annual ultrasound
- 4.5 - 5.4 cm: 3-monthly ultrasound
- 5.5 cm: Refer for surgery (Men)
Pearl 5: The "Diabetes Paradox" Surprisingly, patients with Diabetes Mellitus have a slower growth rate of AAAs compared to non-diabetics. This is thought to be due to glycation of the aortic matrix making it stiffer and more resistant to proteolysis.
Pearl 6: "Trash Foot" If a patient presents with painful, blue toes but palpable pedal pulses, suspect a proximal embolic source (AAA or Popliteal Aneurysm). The "trash" (emboli) has travelled downstream to occlude the digital arteries, sparing the main vessels.
Pearl 7: The Retroperitoneal Seal Most ruptures occur into the retroperitoneum. The bleeding is initially contained (tamponaded) by the psoas muscle and renal fascia. This buys the patient time. If you over-resuscitate with fluids and raise the BP, you overcome this tamponade pressure, causing "free rupture" into the peritoneum and rapid death. Permissive Hypotension is life-saving.
Pearl 8: The "Femoral Pulse" Trap A patient with a ruptured AAA can still have femoral pulses (if the pressure is > 80 systolic). Do not document "Peripheral pulses present" and assume the aorta is fine.
Why This Matters Clinically
- Patient Outcomes: Missed diagnosis of a symptomatic or ruptured AAA is almost invariably fatal. Early detection via screening allows for elective repair with extremely low mortality (less than 2%), essentially curing the patient of this specific risk.
- Healthcare System: Ruptured AAAs require massive resources (emergency surgery, massive transfusion, ICU stay) with poor yields. Screening programmes are cost-effective (approx. £7,600 per QALY) by converting emergency ruptures into planned elective repairs.
- Medico-legal: Failure to examine the abdomen in an elderly patient with back pain, or discharging a "renal colic" patient without imaging who subsequently ruptures, is a leading cause of litigation in emergency medicine and general practice.
- Training: AAA is a core "must-not-miss" diagnosis for all medical students and trainees. Management decisions involve complex risk-benefit analysis (intervention vs. frailty) relevant to senior exams (FRCS, FRACP).
2. Epidemiology
Incidence & Prevalence
- Incidence of Diagnosis: Approximately 150-250 per 100,000 men per year in Western populations. Incidence has notably declined over the last 20 years, largely attributed to the reduction in population-wide smoking rates. [2,4]
- Rupture Incidence: Approximately 20-30 per 100,000 persons per year. [2]
- Prevalence:
- Recent screening studies (e.g., in Sweden, UK) show prevalence in 65-year-old men is now ~1.5-1.7%, down from 4-8% in the 1990s. [2,5]
- In women > 65, prevalence is much lower, estimated at less than 1%. [5]
- Lifetime Risk: For a male smoker, lifetime risk is ~15%. [3]
- Trends: Declining prevalence and rupture rates in developed countries. Increasing in developing nations correlating with increasing smoking prevalence.
- Geographic Variation: Highest in Northern Europe, UK, and North America. Lower in Asian and African populations, though data is less robust.
Demographics
| Factor | Details | Clinical Significance |
|---|---|---|
| Age | Peak prevalence: 75-84 years. Rare less than 50 years. | Degenerative disease of aging; screening starts at 65. |
| Sex | Male:Female ratio 4:1 to 6:1. | Strong male predisposition, possibly sex-hormone linked. Women rupture at smaller diameters. |
| Ethnicity | Caucasian > African American > Asian/Hispanic. | Genetic susceptibility factors (e.g., chromosome 9p21). |
| Geography | High prevalence in UK/Scandinavia. | Correlates with historical smoking patterns. |
| Socioeconomic | Higher in lower socioeconomic groups. | Linked to higher smoking rates and poorer cardiovascular health. |
Risk Factors
Non-Modifiable Risk Factors:
| Factor | Relative Risk / OR (95% CI) | Mechanism |
|---|---|---|
| Age (per decade) | OR 1.71 (1.64–1.77) [1,6] | Elastin fragmentation and cumulative haemodynamic stress. |
| Female Sex | OR 0.17 (protective) [6] | Estrogens may protect aortic wall integrity. |
| Family History | OR 1.94 (1.5–2.5) [6] | High heritability (~70%). 15-20% of patients have a first-degree relative affected. |
| Ethnicity (Black/Asian) | OR 0.5–0.7 [1] | Genetic protective factors relative to Caucasians. |
| Previous History vs other Aneurysms | High Association | Popliteal (50% have AAA), Femoral (85% have AAA), Thoracic (25% have AAA). |
Modifiable Risk Factors:
| Risk Factor | Strength of Association | Evidence Level | Intervention Impact |
|---|---|---|---|
| Smoking | OR > 3.0 (Strongest risk factor) [1,7] | Level 1a | Current smokers have 7x risk of AAA vs never smokers. Smoking cessation reduces growth rate by ~20%. |
| Hypertension | OR 1.25–1.50 [6] | Level 2a | Increased wall stress promotes expansion. |
| Hyperlipidaemia | OR 1.2–1.4 [6] | Level 2a | Atherosclerosis contributes to medial degeneration. |
| Atherosclerotic Disease | OR 1.5–2.0 [6] | Level 2a | CAD/PAD are markers of systemic vascular disease. |
| Diabetes Mellitus | OR 0.5–0.8 (PROTECTIVE) [8] | Level 1b | "The Diabetes Paradox": Glycation of collagen/elastin cross-links may stabilise the aortic wall, preventing dilation. |
Protective Factors:
- Female Sex: Reduces prevalence, but increases rupture risk if aneurysm is present.
- African/Asian Descent: Lower prevalence.
- Diabetes: Significantly reduced prevalence and growth rate.
3. Pathophysiology
Mechanism
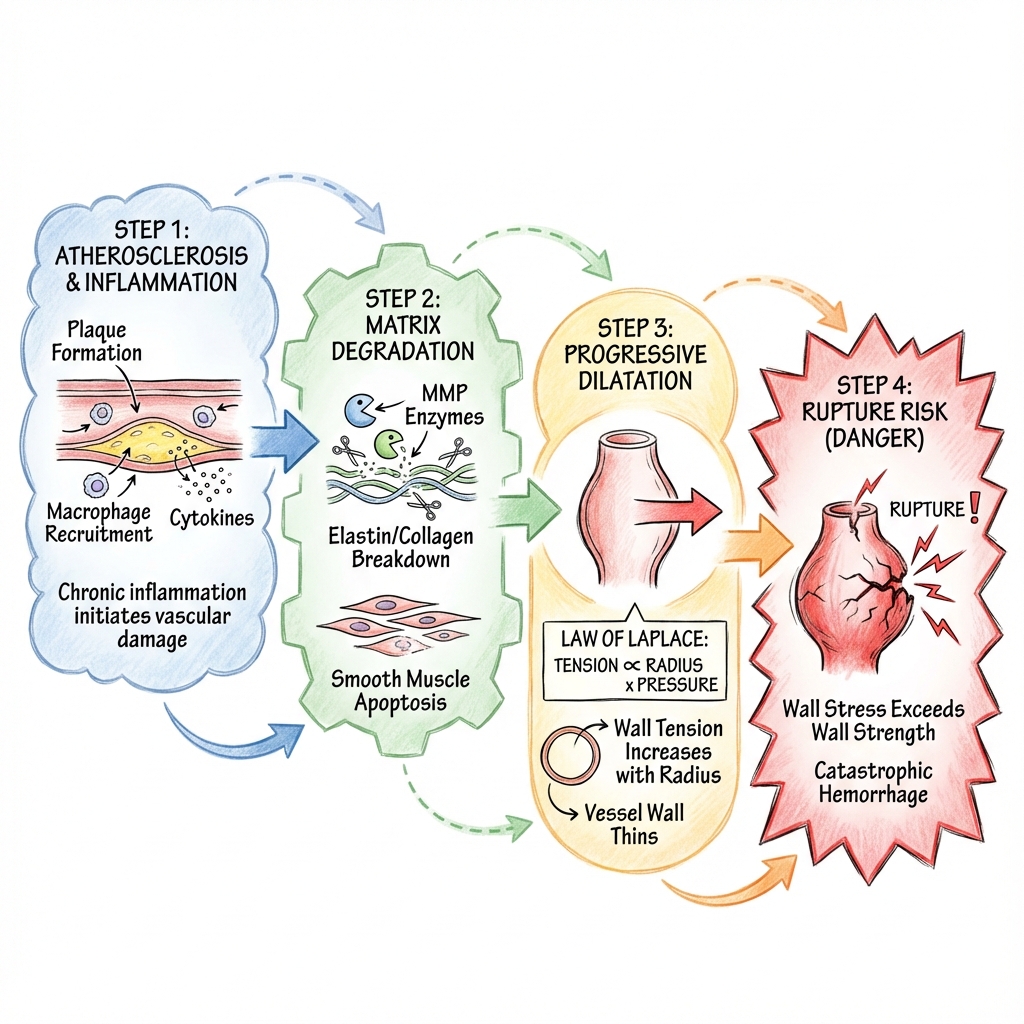
The pathophysiology of AAA is a complex interplay of inflammation, proteolysis, mechanical wall stress, and molecular genetics, leading to the destruction of the aortic media.
Step 1: Initiating Event (Chronic Inflammation)
- Transmural Inflammation: The process begins with an infiltration of inflammatory cells into the aortic wall, primarily macrophages, T-lymphocytes (CD4+ and CD8+), and B-lymphocytes.
- Role of Smoking: Cigarette smoke introduces reactive oxygen species (ROS) and nicotine. Nicotine promotes neovascularisation of the vaso vasorum, while ROS trigger oxidative stress, activating Nuclear Factor Kappa B (NF-κB).
- Shear Stress: The infrarenal aorta is uniquely susceptible due to reflected pulse waves from the high-resistance peripheral circulation. This increases oscillatory shear stress, which upregulates adhesion molecules (VCAM-1, ICAM-1) on the endothelium, recruiting leukocytes.
- Autoimmunity: Some evidence suggests an autoimmune component, with antibodies against specific aortic wall proteins (e.g., aortic aneurysm-associated protein-40).
Step 2: Extracellular Matrix Degradation (Proteolysis)
- Matrix Metalloproteinases (MMPs): The hallmark of AAA pathogenesis is the upregulation of MMPs, specifically MMP-2 (Gelatinase A) and MMP-9 (Gelatinase B).
- "MMP-9: Produced mainly by macrophages. It is the primary enzyme responsible for degrading elastin. Its levels correlate with aneurysm size and expansion rate."
- "MMP-2: Produced by smooth muscle cells and fibroblasts. It degrades collagen and elastin."
- "Cysteinyl Cathepsins: Cathepsins S, K, and L are also upregulated and contribute to elastolysis."
- TIMPs Imbalance: Tissue Inhibitors of Metalloproteinases (TIMPs) are normally present to check MMP activity. In AAA, there is a local deficiency of TIMPs (especially TIMP-3), leading to unchecked proteolysis.
- Elastin Fragmentation: Elastin is the major load-bearing protein of the aorta using the "windkessel" effect. Its fragmentation leads to loss of arterial recoil, dilation, and stiffness. This is an irreversible process as elastin turnover is negligible in adults.
Step 3: Medial Degeneration and Smooth Muscle Cell Apoptosis
- Tunica Media Destruction: The middle layer, rich in Vascular Smooth Muscle Cells (VSMCs) and elastic lamellae, is destroyed.
- VSMC Apoptosis: Inflammatory cytokines (TNF-α, IL-1β) and Fas-ligand interactions trigger apoptosis (programmed cell death) of VSMCs.
- Phenotypic Switching: Remaining VSMCs switch from a "contractile" phenotype to a "synthetic/pro-inflammatory" phenotype, secreting more MMPs and inflammatory mediators instead of maintaing the matrix.
- Laminar Loss: The normal aorta has ~50-70 concentric elastic lamellae. In AAA, these are fragmented and lost ("cystic medial necrosis").
- Calcification: Dystrophic calcification occurs in the degenerated wall, further reducing compliance and increasing local wall stress mismatch.
Step 4: Expansion and Thrombus Formation
- Law of Laplace: Wall Tension ($T$) is proportional to Pressure ($P$) × Radius ($r$) ($T = P \times r$).
- As the aorta dilates (radius increases), the wall tension increases linearly for the same blood pressure.
- "This creates a positive feedback loop: Dilation → Increased Tension → More Dilation."
- Intraluminal Thrombus (ILT):
- As the vessel widens, laminar flow is lost, and turbulent/recirculating zones form, promoting thrombosis.
- ~75% of AAAs contain ILT.
- "The "Active Thrombus" Theory: The ILT is not just passive debris. It is biologically active, trapping neutrophils and platelets. It creates a hypoxic environment for the underlying wall, further driving inflammation and apoptosis."
- The thrombus also acts as a barrier to oxygen diffusion from the lumen to the inner media.
Step 5: Rupture
- Stress-Strength Mismatch: Rupture occurs when the mechanical stress on the aortic wall (Peak Wall Stress - PWS) exceeds its tensile strength.
- Critical Threshold: The risk increases exponentially.
- At 4.0 cm, tension is manageable.
- At 6.0 cm, tension is dangerously high.
- Rupture typically occurs at the "shoulder" of the aneurysm or area of maximum curvature, where compliance mismatch is highest.
- Catastrophe: Rupture leads to massive haemorrhage.
- "Retroperitoneal: Contained rupture. The haematoma is tamponaded by the psoas muscle and renal fascia. Patient may survive hours to reach hospital."
- "Intraperitoneal: Free rupture. Loss of tamponade. Exsanguination occurs in minutes."
Molecular Genetics
Although most AAAs are sporadic, genetic factors play a significant role.
- Locus 9p21: The same locus associated with Coronary Artery Disease (CAD) and Intracranial Aneurysms is strongly linked to AAA. It contains the CDKN2B-AS1 gene (ANRIL), involved in cell cycle regulation and VSMC proliferation.
- DAB2IP: A gene involved in VSMC apoptosis and inflammation.
- LRP1: Low-density lipoprotein receptor-related protein 1. Involved in lipid metabolism and VSMC function.
- TGF-β Pathway: Mutations in this pathway lead to syndromic aneurysms (Marfan, Loeys-Dietz), but subtle variations also contribute to sporadic AAA risk.
Classification
Anatomical Classification (detailed):
| Type | Definition | Prevalence | Surgical Implication |
|---|---|---|---|
| Infrarenal | Originates > 10-15mm below the lowest renal artery. | 90-95% | The "standard" AAA. Suitable for standard EVAR devices which anchor in the healthy infrarenal "neck". |
| Juxtarenal | Originates at the level of renal arteries or has a very short (less than 10mm) infrarenal neck. | 3-4% | Requires suprarenal clamping (Open) or Fenestrated EVAR (FEVAR) to preserve renal flow. |
| Pararenal | Involves the origin of the renal arteries but not the SMA/Celiac. | less than 1% | Requires complex endovascular or open repair with renal revascularisation. |
| Suprarenal | Involves the origins of the renal arteries or visceral arteries (SMA/Celiac). | 1-2% | High surgical risk. Access to visceral vessels required. |
| Type IV TAAA | Thoracoabdominal aneurysm involving the entire abdominal aorta and extending into chest. | Rare (less than 1%) | Crawford Classification applies. Extraordinary risk of paraplegia and renal failure. |
Morphological Classification:
- Fusiform: Symmetrical, spindle-shaped dilation involving the entire circumference. (Most common in AAA).
- Saccular: Asymmetrical, localized outpouching. (Higher risk of rupture at smaller diameters due to uneven wall stress; often mycotic/infectious or penetrating ulcer origin).
Anatomical Considerations
1. Aortic Anatomy
- Surgical Definition: The abdominal aorta begins at the aortic hiatus of the diaphragm (T12) and ends at the bifurcation into common iliac arteries (L4).
- Diameter: Normal adult diameter is approx. 2.0 cm. It tapers distally.
- Wall Structure:
- "Tunica Intima: Endothelium + internal elastic lamina."
- "Tunica Media: Concentric smooth muscle cells + elastic lamellae. (Site of AAA pathology)."
- "Tunica Adventitia: Collagen + vasa vasorum. (Site of inflammatory cell infiltration)."
2. Key Relations (The "Danger Zones")
- Anterior:
- "3rd Part of Duodenum: Crosses the aorta at L3. Direct erosion here causes Primary Aorto-Enteric Fistula."
- "Left Renal Vein: Crosses anteriorly. Can be injured during clamping."
- "Peritoneum: The aorta is retroperitoneal."
- Posterior:
- "Vertebral Bodies (L1-L4): Large chronic AAAs can cause pressure erosion of the vertebrae (scalloping on CT)."
- "Lumbar Veins: Course posteriorly. Bleeding from these during open repair is notoriously difficult to control."
- Lateral (Right):
- "Inferior Vena Cava (IVC): Adherent to the right wall of the aneurysm. Risk of Aorto-Caval Fistula implies injury to the IVC during dissection."
- Lateral (Left):
- "Left Ureter: Runs close to the bifurcation. Danger of injury during dissection or entrapment in Inflammatory AAA."
3. Vascular Access Anatomy (The Groin)
- Femoral Triangle Boundaries:
- "Superior: Inguinal Ligament."
- "Lateral: Sartorius."
- "Medial: Adductor Longus."
- "Floor: Iliopsoas, Pectineus, Adductor Longus."
- "Roof: Fascia Lata."
- Contents (Lateral to Medial - NAVY):
- Nerve (Femoral Nerve) - Outside the femoral sheath.
- Artery (Common Femoral Artery) - The target for EVAR access.
- Vein (Femoral Vein) - Medial to artery. Risk of DVT/injury.
- Y-fronts (Lymphatics) - Risk of lymphocele if dissection is too lateral or lymphatics not ligated.
- Common Femoral Artery (CFA):
- Lies over the femoral head (allowing compression/control).
- Bifurcates into Superficial Femoral (SFA) and Profunda Femoris (PFA).
- "Surgical Pearl: Access must be into the CFA. High puncture (above inguinal ligament) causes retroperitoneal haematoma. Low puncture (SFA/PFA) carries risk of stenosis/occlusion/pseudoaneurysm."
4. Collateral Circulation (The "Blocked Aorta" Concept) In slow-growing occlusive disease or aneurysm thrombosis, collateral pathways maintain lower limb viability:
- System 1: Subclavian → Internal Thoracic → Superior Epigastric → Inferior Epigastric → External Iliac.
- System 2: SMA → Middle Colic → Marginal Artery → Left Colic → IMA → Superior Rectal → Middle/Inferior Rectal → Internal Iliac.
- System 3: Lumbar Arteries → Gluteal Arteries → Profunda Femoris.
Physiological Considerations
- LaPlace's Law: $T = P \times r$. Tension is proportional to radius. A small increase in BP in a dilated aorta causes a massive increase in wall tension. Hence blood pressure control is paramount.
- Renal Function: Juxtarenal aneurysms or repair procedures can compromise renal blood flow, leading to Acute Kidney Injury (AKI).
- Spinal Cord Ischaemia: The Artery of Adamkiewicz (great radicular artery) supplies the anterior spinal cord (motor function).
- "Origin: T9-T12 (75% of people)."
- "Risk: If the AAA repair excludes/covers this artery (e.g., high clamp or extensive stent), the patient may wake up paraplegic."
4. Clinical Presentation
Symptoms
1. Asymptomatic (75% of non-ruptured cases)
- Most AAAs are detected incidentally on imaging for other reasons (ultrasound for gallstones, CT for renal colic/cancer staging) or via screening.
2. Symptomatic (Non-Ruptured)
- Abdominal Pain: Deep, steady, gnawing pain in the mesogastrium.
- Back Pain: Radiating to the lumbosacral region (vertebral erosion or retroperitoneal stretching).
- Pulsation: Patient notices a "heartbeat" in their tummy, especially when lying flat.
- Thromboembolic: "Blue Toe Syndrome" (trash foot) from embolisation of mural thrombus to digital arteries.
3. Symptomatic (Ruptured) - EMERGENCY
- Pain: Severe, sudden onset, tearing/ripping pain in abdomen, back, or flank.
- Collapse: Syncope or transient loss of consciousness (initial bleed), followed by haemodynamic collapse.
- Vomiting/Nausea: Vagal response to retroperitoneal irritation.
Signs
Non-Ruptured:
- Pulsatile Mass: Supra-umbilical (aorta bifurcates at umbilicus).
- Expansile: Hands move apart. Evaluation is difficult in obese patients.
- Bruit: Abdominal bruit may be audible (turbulent flow), though non-specific.
Ruptured:
- Classic Triad: Hypotension + Back Pain + Pulsatile Mass (Only ~50% sensitivity).
- Shock: Tachycardia, hypotension, pallor, diaphoresis, prolonged capillary refill.
- Grey Turner's Sign: Flank ecchymosis (late sign > 24h, represents retroperitoneal haematoma tracking).
- Cullen's Sign: Periumbilical ecchymosis (rare).
- Distal Ischaemia: Loss of femoral pulses if the aorta thromboses or pressure is too low to perfuse limbs.
Red Flags
[!CAUTION] Red Flags — Signs of Impending or Actual Rupture
- Syncope/Collapse: In a patient with known AAA or back pain.
- Tender Aneurysm: Pain on palpation of the aorta suggests inflammation/rapid expansion.
- Unexplained Hypotension: In an elderly patient with back pain.
- Transient Responder: Patient collapses, recovers (tamponade), then collapses again (free rupture).
- New Back Pain: In a patient with a known AAA.
5. Clinical Examination
Structured Approach
1. General Inspection
- Habitus: Marfanoid features? (Suggests connective tissue disorder, though rare for abdominal, common for thoracic).
- Hands: Tobacco staining (heavy smoking), clubbing (rare, maybe infected graft), reduced perfusion.
- Face: Pallor, diaphoresis (if symptomatic).
2. Abdominal Examination
- Inspection: Visible pulsation in thin patients. Scars from previous vascular surgery.Review of topics in vascular surgery.
Glossary of Key Terms
| Term | Definition | Context |
|---|---|---|
| EVAR | Endovascular Aneurysm Repair | Minimally invasive stent-grafting. |
| Endoleak | Blood flow outside the graft but within the aneurysm sac | The unique complication of EVAR. |
| Sac | The aneurysm wall (the dilated aorta) | Left in situ during EVAR and Open Repair. |
| Neck | Healthy aorta proximal to the aneurysm | Crucial for EVAR sealing (> 10mm needed). |
| Landing Zone | The area of vessel wall where the stent-graft anchors | Proximal (Aortic) and Distal (Iliac). |
| PWS | Peak Wall Stress | Biomechanical measure of rupture risk. |
| MMP | Matrix Metalloproteinase | Enzyme that degrades elastin in the aortic wall. |
| Aorto-Enteric Fistula | Erosion of aorta into bowel (usually duodenum) | Rare, fatal cause of GI bleed. |
| Trash Foot | Livedo reticularis / Blue toes | Distal micro-embolisation. |
| Juxtarenal | AAA extending up to the renal arteries | Requires complex repair (Fenestrated EVAR). |
Key Takeaways
- Screening Saves Lives: The only way to reduce AAA mortality is to detect them before they rupture. The 5.5cm threshold is the "Holy Grail" of vascular surgery.
- Rupture is a Clinical Diagnosis: Do not wait for a CT if a patient with a known AAA collapses. The "Classic Triad" is insensitive.
- EVAR is not a Panacea: It has lower early mortality but higher late failure. It requires a life-long contract of surveillance.
- Medical Therapy Matters: Smoking cessation and statins are just as important as the surgery itself for long-term survival.
- Anatomy Dictates Treatment: Not every aneurysm can be fixed with EVAR. Short necks and tortuous iliacs require open repair or complex endovascular solutions.
- Palpation:
- Palpate deep in the epigastrium/supra-umbilical region.
- Use both hands, one on each side of the pulsation.
- Assess if Expansile (hands pushed apart) vs Transmitted (hands pushed up).
- Estimate diameter (unreliable, usually overestimates).
- "Tenderness: Gently assess. Tenderness = Medical Emergency."
- Auscultation: Listen for bruits (aortic, renal, iliac).
3. Peripheral Vascular Exam
- Pulses: Femoral, Popliteal, Posterior Tibial, Dorsalis Pedis. (AAA is often associated with peripheral aneurysm or occlusive disease).
- Popliteal Aneurysm: Widened popliteal pulse? (50% of patients with popliteal aneurysms have an AAA).
- Perfusion: Capillary refill, temperature, color.
Differential Diagnosis (The "Back Pain" Mimics)
Distinguishing AAA from common conditions is the most critical skill in the Emergency Department.
| Condition | Pain Character | Examination Findings | Diagnostic Clue |
|---|---|---|---|
| Ruptured AAA | Tearing, constant, radiates to back/flank. Unrelieved by position. | Pulsatile mass (50%), Hypotension, Grey Turner's sign (late). | History of AAA, Age > 60, Syncope. |
| Renal Colic | Colicky (waves), loin-to-groin radiation. Patient cannot lie still. | Loin tenderness. Soft abdomen. No peritonism. | Haematuria (dipstick). Caution: AAA can cause haematuria if it erodes renal vein. |
| Musculoskeletal | Mechanical, worse with movement/palpation of spine. Localised. | Vertebral tenderness, paravertebral spasm. Normal pulses. | History of lifting/trauma. Reproducible pain. |
| Diverticulitis | Constant LLQ pain. | LLQ tenderness, guarding, fever. | Pyrexia, raised WCC, change in bowel habit. |
| Pancreatitis | Epigastric, boring through to back. Relieved by sitting forward. | Epigastric tenderness. | History of alcohol/gallstones. Raised Lipase/Amylase. |
| Spinal Stenosis | "Claudication" of the back/legs. Worse on walking. | Normal pulses. Reduced spinal ROM. | Pain relieved by leaning forward ("Simon Cowell sign"). |
Clinical Decision Rules
-
Rule 1: In any patient > 60 presenting with "Renal Colic" (flank pain), you MUST exclude AAA. An urgent ultrasound or CT is mandatory before discharge.
- "Reasoning: 10% of ruptured AAAs are misdiagnosed as renal colic initially, often with fatal consequences."
-
Rule 2: A "Tender Aneurysm" is a ruptured aneurysm until proven otherwise.
- "Reasoning: Tenderness implies inflammation of the adventitia or rapid stretching. Do not delay for ultrasound if the patient is unstable—go to theatre or CT immediately depending on protocol."
| Feature | Frequency (Rupture) | Clinical Significance |
|---|---|---|
| Abdominal Pain | 70-80% | Often non-specific, mistaken for colic/gastritis. |
| Back/Flank Pain | 50-70% | Mistaken for renal colic/musculoskeletal. |
| Pulsatile Mass | 50-60% | Absence does NOT rule out rupture esp. in obese/shocked patients. |
| Hypotension | 50-70% | Late sign. Younger patients compensate well initially. |
| Gross Haematuria | less than 5% | Rare. Rupture into ureter/bladder or renal vein congestion. |
6. Investigations
First-Line (Screening/Surveillance)
- Abdominal Ultrasound (Duplex):
- "Sensitivity: > 98%. Specificity: > 98%."
- "Role: Primary tool for screening and monitoring size."
- "Measurement: Antero-posterior (AP) diameter, inner-to-inner wall (NICE/ESVS standard) or outer-to-outer (US standard). Critical to know which standard your unit uses."
- "limitations: Bowel gas, obesity can obscure views. Operator dependent."
Imaging for Pre-operative Planning / Symptomatic
- CT Angiography (CTA) Aorta:
- "Role: Gold standard for diagnosis of rupture and anatomical planning for repair."
- "Findings:"
- exact diameter and extent (infrarenal vs juxtarenal).
- "Neck" anatomy (length, angulation, calcification) - determines EVAR suitability.
- Retroperitoneal haematoma (stranding, contrast extravasation) in rupture.
- "Draped Aorta Sign": Posterior aorta follows contour of vertebra (sign of contained rupture).
- "Crescent Sign": High-attenuation crescent within thrombus (sign of impending rupture).
Laboratory Tests
| Test | Expected Finding | Purpose |
|---|---|---|
| FBC | Hb ↓ (rupture), WCC ↑ (inflammation/infection) | Baseline, assess blood loss/infection. |
| U&Es | Creatinine ↑ (AKI from shock or renal artery involvement) | Baseline renal function (contrast risk), dehydration. |
| Crossmatch | Group & Save / Crossmatch 4-6 units | MANDATORY if symptomatic/rupture. Massive haemorrhage protocol. |
| Clotting | INR/APTT ↑ (Coagulopathy of trauma/shock) | Baseline for correction during surgery. |
| CRP/ESR | Elevated | Inflammatory AAA or Mycotic aneurysm. |
| Lactate | Elevated | Marker of hypoperfusion/shock. Predictor of mortality. |
Diagnostic Criteria (ESVS 2024)
- Normal Aorta: less than 2.5 - 3.0 cm.
- Ectatic Aorta: 2.5 - 2.9 cm.
- Aneurysm: ≥3.0 cm.
- "Small: 3.0 - 3.9 cm."
- "Medium: 4.0 - 4.9 cm."
- "Large: ≥5.0 cm (Women), ≥5.5 cm (Men)."
7. Management
Management Algorithm
AI-Generated Management Algorithm Image Required:
Image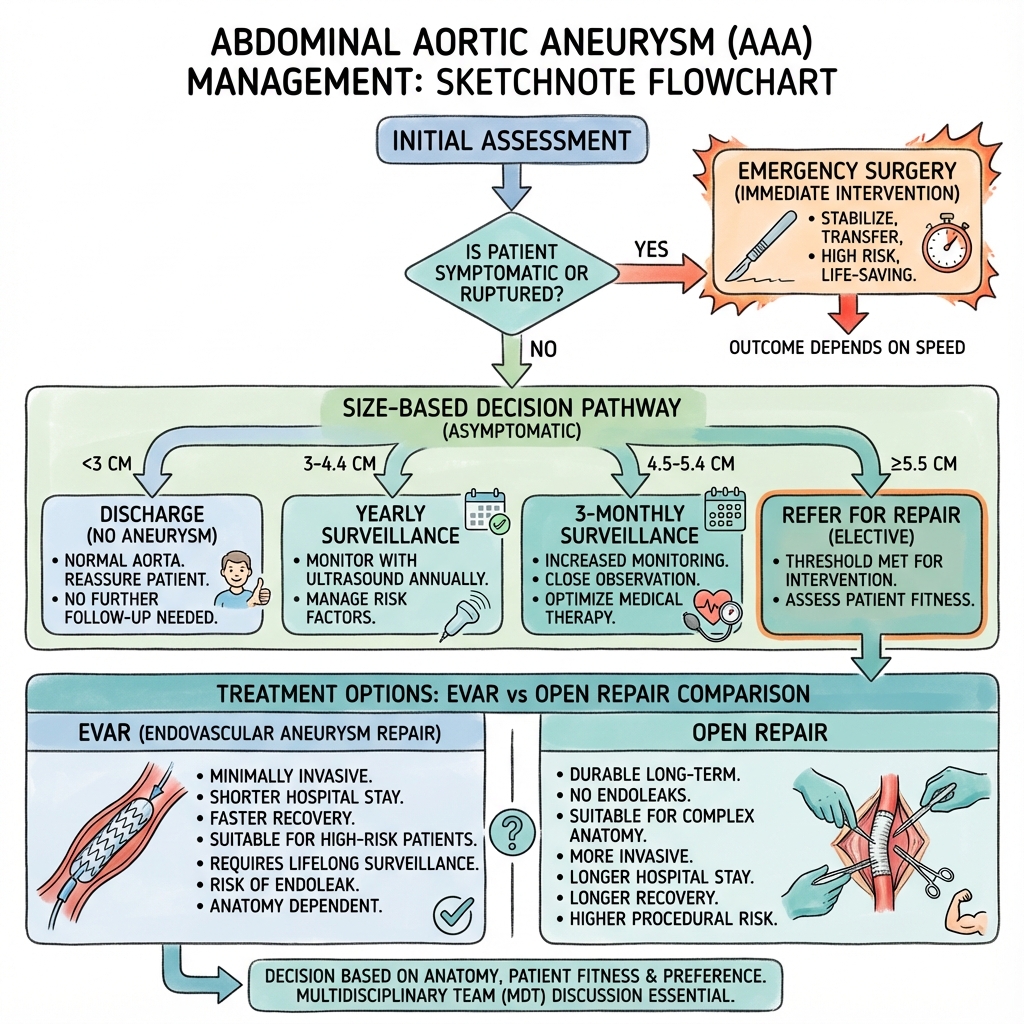 AAA Management Algorithm
AAA Management Algorithm
Generate a sketchnote-style flowchart image with:
- Handwritten/hand-drawn aesthetic
- Soft pastel color palette (mint, lavender, peach, sky blue)
- Medical icons and illustrations
- Clear decision boxes with rounded corners
- Arrows showing flow direction
- Key treatments in separate colored boxes
- White/cream background
ASCII Management Flowchart:
Suspected AAA Presentation
(Screening or Symptomatic)
↓
┌─────────────────────────────────────────┐
│ INITIAL ASSESSMENT │
│ - History: Pain, Syncope, Risk Factors │
│ - Exam: Pulsatile Mass, Shock? │
│ - Investigate: US (Screen), CT (Pain) │
└─────────────────────────────────────────┘
↓
┌─────────────────────────────────────────┐
│ SEVERITY/SIZE DECISION │
├─────────────────────────────────────────┤
│ less than 5.5cm (Men) / less than 5.0cm (Women) │
│ → SURVEILLANCE │
│ │
│ ≥5.5cm / ≥5.0cm OR Symptomatic │
│ → CONSIDER REPAIR │
│ │
│ Ruptured (Pain + Shock + Known AAA) │
│ → EMERGENCY REPAIR │
└─────────────────────────────────────────┘
↓
┌─────────────────────────────────────────┐
│ TREATMENT PATHWAYS │
│ Fit for Surgery: │
│ 1. EVAR (if anatomy suitable) │
│ 2. Open Repair (gold std for long) │
│ │
│ Unfit for Surgery: │
│ 1. Best Medical Therapy │
│ 2. Palliative Care (if rupture) │
└─────────────────────────────────────────┘
Acute/Emergency Management (Rupture)
Immediate Actions (The "Code Crimson"):
- Activate Major Haemorrhage Protocol: Call blood bank for O-Negative blood, FFP, platelets.
- Permissive Hypotension: Maintain systolic BP 70-90 mmHg (conscious, palpable radial pulse). Do not fluid resuscitate to normal BP as this "pops the clot" (dislodges the retroperitoneal containment) leading to free rupture and death.
- Analgesia: IV Morphine/Fentanyl. Avoid NSAIDs (renal risk).
- Imaging: If stable -> CTA. If unstable -> Straight to Theatre (if diagnosis known/highly suspected).
- Proximal Control: The priority in theatre. Cross-clamp aorta (supraceliac if needed) or deploy aortic occlusion balloon (REBOA).
Surveillance (Conservative Management)
For asymptomatic small aneurysms, the risk of rupture is lower than the risk of surgery. The mainstay is regular ultrasound monitoring to detect rapid expansion or threshold diameter.
NAAASP Surveillance Intervals (UK National Screening Programme):
- 3.0 - 4.4 cm: Annual Ultrasound.
- 4.5 - 5.4 cm: 3-monthly Ultrasound.
- ≥5.5 cm: Refer for Surgery (Vascular Surgeon).
Medical Optimization (Best Medical Therapy): All patients with AAA (any size) have systemic vascular disease. The goal is to reduce cardiovascular mortality (MI/Stroke) and slow aneurysm growth.
1. Smoking Cessation (Critical)
- Effect: Smoking is the only modifiable factor PROVEN to increase growth rate. Cessation reduces growth by 20% and rupture risk significantly.
- Intervention: NRT, Varenicline, Counselling. "Pack years" correlates directly with diameter.
2. Antiplatelet Therapy
- Drug: Aspirin 75mg daily or Clopidogrel 75mg daily.
- Rationale: Reduces risk of MI and Stroke (primary cause of death in AAA patients). Does not affect aneurysm growth directly but prevents mural thrombus complications.
3. Lipid Lowering (Statins)
- Drug: Atorvastatin 80mg daily (High Intensity).
- Target: LDL less than 1.8 mmol/L (or less than 1.4 mmol/L in very high risk).
- Mechanism:
- "Pleiotropic Effects: Statins reduce MMP-9 levels and aortic wall inflammation, potentially slowing growth (though clinical trial evidence for growth reduction is mixed)."
- "Plaque Stabilisation: Prevents coronary events."
4. Blood Pressure Control
- Target: less than 130/80 mmHg.
- Preferred Agent: ACE Inhibitors (e.g., Ramipril) or ARBs (e.g., Losartan).
- Mechanism: Angiotensin II is upregulated in AAA and promotes remodeling. Blockade may reduce stiffness.
5. Exercise and Diet
- Exercise: Moderate aerobic exercise is safe and recommended. It improves "Surgical Fitness" (CPET performance) if repair is eventually needed.
- Diet: Mediterranean diet.
Surgical Management
Indications for Elective Repair:
- Diameter: ≥5.5 cm (Men), ≥5.0 cm (Women) [ESVS 2024 Level 1a].
- This threshold is based on the UK Small Aneurysm Trial, which showed no survival benefit for early surgery less than 5.5cm, but operative risk remained.
- Rapid Expansion: > 1.0 cm growth in 1 year (or > 0.5 cm in 6 months).
- Rapid growth indicates instability and active inflammation.
- Symptomatic: Any size aneurysm causing pain/tenderness.
- Tenderness is a sign of contained rupture or inflammation ("Inflammatory AAA").
- Saccular Morphology: Depending on shape, may repair at smaller diameters (e.g., 4.5-5.0 cm) due to unpredictable wall stress distribution.
Procedure Options:
1. Endovascular Aneurysm Repair (EVAR)
Technique Overview: EVAR involves placing a stent-graft inside the aorta to reline it.
- Access: Bilateral femoral artery access (percutaneous or small cut-down).
- Deployment: The main body is advanced to the renal arteries. The "gate" is cannulated from the contralateral side. Limbs are extended into the iliac arteries.
- Seal: The graft relies on an "oversized" radial force and barbs to seal against the aortic neck (top) and iliac arteries (bottom).
Suitability (IFU Criteria):
- Neck Length: > 10-15 mm of healthy aorta below renals.
- Neck Angulation: less than 60 degrees.
- Access: Iliac arteries > 7mm diameter, not heavily calcified or tortuous.
Pros:
- Lower Perioperative Mortality: 0.5-1% (vs 3-5% for Open).
- Recovery: Rapid (Hospital stay 1-2 days). Local anaesthetic possible.
- Incision: Minimal (puncture wounds).
Cons:
- Durability: "Achilles Heel". Risk of graft migration, component separation, or leaks.
- Re-intervention: 20-30% of patients require further procedures within 5 years.
- Surveillance: Requires lifelong annual CT or Duplex scans ($$$$ and radiation).
- Endoleaks: Types I-V (see complications).
Step-by-Step Procedure (Standard EVAR):
- Prep: Patient supine. GA or Local + Sedation. Heparin 5000u.
- Access: Ultrasound-guided puncture of common femoral arteries. Insert 6-8Fr sheaths. Proglide sutures for closure.
- Wires: Stiff Lunderquist wires advanced into thoracic aorta.
- Main Body: The device delivery system (14-20Fr) is advanced over the stiff wire.
- Angiogram: Pigtail catheter confirms position of renal arteries.
- Deployment: The graft is unsheathed just below the lowest renal artery. The top bare stents (fixation) cross the renal ostia (allowing flow through the struts).
- Cannulation: The contralateral "gate" (short limb) is cannulated with a wire/catheter.
- Limb Extension: Iliac limbs are deployed down to the iliac bifurcation (preserving Internal Iliac Artery if possible).
- Molding: A compliant balloon is used to "iron out" the graft against the vessel wall to ensure a seal.
- Completion Angiogram: Check for patency of renals and exclusion of aneurysm (no leaks).
2. Open Surgical Repair (OSR)
Technique Overview: Traditional major surgery to replace the diseased aorta with a synthetic tube.
- Access: Midline laparotomy (from xiphisternum to pubis) or Transverse incision.
- Exposure: The small bowel is eviscerated to the right. The duodenum is mobilized (Kocher manoeuvre) to reveal the aorta neck.
- Control: Clamps are placed on the aorta (below renals) and iliac arteries.
- Repair: The aneurysm sac is opened. A Dacron or PTFE graft is sewn in place (end-to-end anastomosis).
- Closure: The aneurysm sac is wrapped around the graft to protect the bowel from erosion.
Pros:
- Durability: Excellent long-term results (> 20 years). The graft rarely fails.
- No Surveillance: "Fit and forget".
- Gold Standard: For young (less than 70), fit patients with long life expectancy.
Cons:
- Mortality: Higher 30-day mortality (3-5%).
- Morbidity: High. Cardiac stress, renal failure (clamp time), respiratory failure, ileus.
- Hernia: 10-20% risk of incisional hernia.
- Sexual Dysfunction: 5-10% risk of retrograde ejaculation (injury to sympathetic plexus).
Step-by-Step Procedure (Infrarenal Repair):
- Incision: Full midline laparotomy. Transverse colon lifted/retracted.
- Dissection: Posterior peritoneum incised. Ligament of Treitz diveded. Duodenum reflected right.
- Control: Aorta dissected at the neck. Renal vein identified (and retracted). Clamp sites prepared.
- Heparin: Systemic heparinization (3000-5000u).
- Clamping: Distal clamps (Iliacs) first (prevent emboli). Proximal clamp (Infrarenal) applied.
- Aneurysmotomy: Sac opened longitudinally. Mural thrombus removed manually. Backbleeding lumbar arteries oversewn (2/0 Prolene).
- Proximal Anastomosis: Dacron tube graft sewn to aortic neck (3/0 Prolene, continuous suture). Parachute technique often used.
- Testing: Clamp released briefly to flush clots and test seal.
- Distal Anastomosis: Graft sewn to aortic bifurcation (or iliacs if Y-graft used).
- Reperfusion: Distal clamps removed first (one leg at a time) to prevent sudden hypotension ("declamping shock" from metabolites).
- Closure: Sac wall sewn over the graft. Peritoneum closed. Omentum placed over aorta. Mass closure of abdomen.
3. Fenestrated/Branched EVAR (FEVAR/BEVAR)
- For complex aneurysms (juxtarenal/suprarenal).
- Custom-made grafts built based on patient's specific CT anatomy (takes 6-8 weeks to manufacture).
- Have reinforced holes ("fenestrations") or branches that align with renal/visceral arteries.
- Stents are then placed through these holes into the target vessels.
- Indication: Fit patients with complex anatomy who are unfit for Open Repair.
Post-Operative Care (ERAS Protocol)
Enhanced Recovery After Surgery (ERAS) pathways have revolutionised AAA care, reducing length of stay and complications.
Phase 1: Pre-operative (Optimisation)
- Carbohydrate Loading: Clear carbohydrate drinks up to 2h pre-op (reduces insulin resistance).
- Counselling: Managing expectations for pain and mobility.
- Medical Optimisation: Correction of anemia (IV Iron), smoking cessation.
Phase 2: Intra-operative
- Anaesthesia: Spinal/Epidural analgesia preferred (or TAP blocks) to minimize opioid use.
- Fluid Management: Goal-directed therapy (Oesophageal Doppler) to prevent salt/water overload aka "wet lungs".
- Normothermia: Bair Hugger to prevent coagulopathy.
Phase 3: Post-operative (The "Dream" Pathway)
- Day 0 (The night of surgery):
- "Drink: Sips of water allowed immediately."
- "Eat: Light diet evening of surgery (if EVAR)."
- "Move: Sit out of bed for 2 hours (EVAR)."
- Day 1:
- "Drains: Remove urinary catheter (prevents UTI). Remove IV fluids (prevents oedema)."
- "Mobility: Walk lengths of the ward."
- "Analgesia: Oral Paracetamol/NSAIDs. Wean PCA/Epidural."
- Day 2-3 (Discharge Criteria):
- Independent mobility.
- Pain controlled on oral analgesia.
- Bowels open (or passing flatus) - ensuring no ileus.
- Hypertension controlled (Systolic less than 140 to protect graft).
Follow-Up Protocol
- EVAR:
- CT Angiogram at 1 month (check baseline position/seal).
- Ultrasound annualy thereafter.
- Why? To detect migration, sac expansion, or late endoleaks.
- Open Repair:
- Clinical check at 6 weeks (wound healing).
- No routine imaging needed (extremely durable).
Choosing the Right Treatment (The Frailty vs Durability Balance)
| Patient Profile | Recommended Approach | Rationale |
|---|---|---|
| Fit, less than 70 years old | Open Repair | Maximum durability. Low perioperative risk. Avoids lifelong CT scans/radiation. |
| Fit, > 75 years old | EVAR (if anatomy suitable) | Faster recovery. Avoids physiological stress of open surgery. |
| Unfit (Cardiac/Resp disease) | EVAR | Open repair too risky (high mortality). |
| Unfit, Anatomy unsuitable | Medical Management | Risks of complex EVAR or Open Repair usually outweigh rupture risk. Conservative care is safer. |
| Rupture | EVAR (if feasible) | Better survival chance in the acute setting (IMPROVE trial). |
8. Complications
Natural History Complications
- Rupture: The terminal event. Fatal in > 80%.
- Thromboembolism: "Trash foot" (distal micro-emboli causing livido reticularis or gangrene toes).
- Compression:
- IVC: Deep Vein Thrombosis (DVT), leg swelling.
- Duodenum: Vomiting (rare).
- Ureter: Hydronephrosis.
- Fistula:
- Aorto-enteric Fistula: Erosion into duodenum. Presents as "Herald Bleed" (self-limiting haematemesis) followed by massive exsanguination.
- Aorto-caval Fistula: Erosion into IVC. High-output cardiac failure, continuous bruit, palpable thrill.
Post-Operative Complications (Early)
Post-Operative Complications (Early)
1. Acute Kidney Injury (AKI)
- Incidence: 5-10% (Open), 1-5% (EVAR).
- Mechanism:
- "Suprarenal Clamping: Direct ischaemia to kidneys."
- "Embolisation: Showers of atheroma into renal arteries during manipulation."
- "Contrast Nephropathy: High volume contrast in EVAR."
- "Hypotension: Intra-operative instability."
- Prevention: Pre-hydration, minimizing clamp time, "Cold Perfusion" of kidneys if long clamp time needed.
- Management: Fluid resuscitation, dopamine (historically), Dialysis (CVVH) if refractory.
2. Colonic Ischaemia
- Incidence: 1-2% (EVAR), 2-3% (Open). Mortality > 50%.
- Mechanism:
- The Inferior Mesenteric Artery (IMA) arises from the aneurysm sac and is usually sacrificed (oversewn or covered).
- The Sigmoid colon relies on collateral supply from the SMA (via Marginal Artery of Drummond). If collaterals are poor, ischaemia occurs.
- Presentation: Bloody diarrhoea post-op ("currant jelly stool"), acidosis, rising lactate, sepsis on Day 1-3.
- ** Diagnosis**: Flexible Sigmoidoscopy (Gold Standard) within 24h of bloody stool.
- "Grade 1: Mucosal erythema (Conservative)."
- "Grade 2: Mucosal necrosis (Conservative/Surgery)."
- "Grade 3: Transmural necrosis (Emergency Colectomy)."
- Management: IV fluids, antibiotics. If gangrenous -> Hartmann's Procedure.
3. Spinal Cord Ischaemia (Paraplegia)
- Incidence: 0.2% (Infrarenal), but up to 10-20% for Extent II TAAA.
- Mechanism: Loss of the Artery of Adamkiewicz (Great Radicular Artery), which supplies the anterior spinal cord (motor function).
- Presentation: Anterior Cord Syndrome (Paraplegia + Loss of Pain/Temp + PRESERVED dorsal columns/proprioception).
- Prevention (COPS Protocol):
- "CSF Drainage: Keep CSF pressure less than 10 mmHg (increases perfusion gradient)."
- "Oxygen delivery: Maintain Hb > 100."
- "Pressure: Maintain MAP > 90 mmHg."
- "State of Cord: Regular neurological checks."
4. Trash Foot (Distal Embolisation)
- Incidence: 2-5%.
- Mechanism: Dislodging mural thrombus with wires/catheters (EVAR) or clamps (Open).
- Presentation: "Blue Toe Syndrome", Livedo reticularis, loss of pedal pulses.
- Management: Heparin, Iloprost. Embolectomy if large vessel occlusion.
5. Graft Infection
- Incidence: less than 1% (rare but catastrophic).
- Pathogens: Staphylococcus epidermis (biofilm), S. aureus.
- Presentation: Fever, discharging sinus (groin), pseudoaneurysm, vague back pain (late).
- Imaging: Perigraft fluid, gas, failure of incorporation on CT/PET.
- Management: Explant of infected graft + Extra-anatomic bypass (Ax-Fem) or Rifampicin-soaked replacement. High mortality.
6. Abdominal Compartment Syndrome (ACS)
- Incidence: Ruptured AAA repair (> 20%).
- Mechanism: Reperfusion injury causes massive gut oedema + Retroperitoneal haematoma.
- Diagnosis: Bladder Pressure > 20 mmHg + Organ Dysfunction (Oliguria/Respiratory failure).
- Management: Decompression Laparostomy (Bogota Bag/vac dressing). Delayed closure.
Post-Operative Complications (Late)
Unique to EVAR: Endoleaks (Persistence of blood flow into the aneurysm sac outside the limb)
| Type | Cause | Management |
|---|---|---|
| Type I | Incomplete seal at proximal (Ia) or distal (Ib) attachment zones. | Urgent Repair (Cuff extension, ballooning). High rupture risk. |
| Type II | Retrograde flow from lumbar arteries or IMA. | Watch. Repair only if sac expands > 5mm. (Coil embolisation). |
| Type III | Graft defect (tear) or component separation. | Urgent Repair (Relining). |
| Type IV | Graft porosity (historical). | Conservative. |
| ### Detailed Management of Endoleaks (Complications of EVAR) |
Endoleaks are the failure mode of EVAR. They represent persistent blood flow into the aneurysm sac outside the graft lumen, maintaining pressure and risk of rupture.
Type I Endoleak (Attachment Site Failure)
- Definition: Failure of the seal at the proximal (Ia) or distal (Ib) landing zones.
- Mechanism:
- "Ia: The graft migrates down, or the neck dilates, creating a gap between the graft fabric and the aortic wall. Blood flows directly into the sac at systemic pressure."
- "Ib: The iliac limbs pull up or the iliac artery dilates."
- Risk: HIGH. The sac is pressurised to systemic BP. Rupture risk is significant.
- Diagnosis: Detected on early CT Anjiogram. Contrast seen tracking alongside the graft attachment.
- Management: MANDATORY REPAIR.
- "Endovascular:"
- Balloon Moulding: Re-balloon the seal zone (first line).
- Cuff Extension: Add a proximal aortic cuff or distal iliac extension to bridge the gap.
- Palmaz Stent: A high-radial-force bare stent to pin the graft against the wall.
- Chimney/Snorkel EVAR: If cuff covers renal arteries, use parallel grafts for renals.
- "Surgical: Conversion to open repair (explant) or banding."
- "Endovascular:"
Type II Endoleak (Retrograde Flow)
- Definition: "Back-bleeding" into the sac from patent lumbar arteries (IIa) or the Inferior Mesenteric Artery (IIb).
- Mechanism: The graft seals well, but the low-pressure sac sucks blood in from these collateral vessels.
- Risk: LOW. Pressure is usually low. Most resolve spontaneously (thrombose).
- Diagnosis: Contrast seen in the posterior sac (lumbar) or anterior sac (IMA) on delayed phase CT.
- Management: Conservative surveillance.
- "Indication for Repair: Sac Expansion > 5-10mm."
- "Technique: Trans-arterial (coil embolisation of internal iliac/lumbar) or Trans-caval (puncture sac from IVC) or Direct Sac Puncture (glue/coils)."
Type III Endoleak (Mechanical Failure)
- Definition: Structural failure of the graft itself.
- "IIIa: Disconnection of modular components (e.g., limb separates from main body)."
- "IIIb: Fabric tear or holes."
- Risk: HIGH. Pressurised.
- Management: Urgent Relining. Place a new "stent-graft inside the stent-graft".
Type IV Endoleak (Graft Porosity)
- Definition: Micro-leaks through the fabric pores seen immediately post-op.
- Mechanism: Heparinisation + porous fabric.
- Management: Self-limiting. Resolves once heparin reverses. Historical issue with older grafts.
Type V Endoleak (Endotension)
- Definition: Sac expansion WITHOUT visible leak on imaging.
- Mechanism: Transmission of pressure pulse wave through the clot/graft to the wall? Ultra-filtration of plasma?
- Management: If expansion continues, treat as Type I (relining) or Conversion to Open.
ASCII: Endoleak Types
AORTA
| |
| | < --- Type Ia (Proximal Seal Leak)
__|___|__
/ GRAFT \
/ / \ \
|__|_______|__|
| | | |< --- Type II (Lumbar Artery Backflow)
| | | |
|__|_______|__|
| |
| | < --- Type Ib (Distal Seal Leak)
ILIAC ARTERIES
Special Populations
1. Women with AAA
- Prevalence: Lower (less than 1%).
- Rupture Risk: HIGHER at smaller diameters. Women rupture at ~5.0cm more often than men at 5.5cm.
- Guideline: ESVS 2024 sets threshold for repair at 5.0 cm (vs 5.5 cm for Men).
- Outcome: Women have higher mortality after both Open and EVAR surgery. Anatomy is often more challenging (smaller access vessels).
2. Mycotic (Infected) Aneurysm
- Definition: Aneurysm caused by infection of the aortic wall.
- Pathogens: Salmonella spp. (most common), Staphylococcus aureus.
- Presentation: Fever, raised CRP/WCC, painful pulsatile mass.
- Imaging: Saccular shape, periaortic gas/fluid/inflammation.
- Management:
- Prolonged IV antibiotics (6-12 weeks).
- Surgical excision + debridement + reconstruction.
- "Reconstruction: Use biological graft (Rifampicin-soaked Dacron, Cryopreserved Homograft, or Bovine Pericardium) or Extra-anatomic bypass (Axillo-bifemoral) to avoid graft infection. EVAR is controversial (high risk of graft infection)."
3. Inflammatory AAA (iAAA)
- Definition: 5-10% of AAAs characterize by marked thickening of aneurysm wall and extensive perianeurysmal fibrosis.
- Appearance: "Mantel Sign" on CT - thick rind of soft tissue anterior/lateral (spares posterior).
- Effect: Fibrosis entraps adjacent structures (Ureters -> Hydronephrosis; IVC -> Oedema).
- Associations: IgG4-related disease.
- Management:
- Steroids/Immunosuppression (if symptomatic but small).
- Surgery (Repairing the AAA usually extinguishes the inflammatory process). Ureterolysis may be needed.
9. Prognosis & Outcomes
Natural History
-
Growth Rate:
- "Small (3-4 cm): ~2-3 mm/year."
- "Medium (4-5 cm): ~3-5 mm/year."
- "Large (> 5 cm): > 5 mm/year."
-
Accelerating Factors: Smoking, Hypertension.
-
Decelerating Factors: Diabetes.
-
Rupture Risk (Annual Risk of Rupture):
- less than 3.0 cm: Negligible.
- 3.0 - 3.9 cm: 0.3%
- 4.0 - 4.9 cm: 1.0%
- 5.0 - 5.9 cm: 3% - 15% (Inflection point matches 5.5cm guideline).
- 6.0 - 6.9 cm: 10% - 20%
-
7.0 cm: > 30%
-
Why outcomes are poor in rupture:
- 50% die before reaching hospital.
- Of those reaching hospital, 50% die (operative mortality 30-50%).
- Total community mortality > 80%.
Outcomes with Treatment (EVAR vs Open)
- Perioperative Mortality: EVAR is superior (0.5% vs 3.0%). [10]
- Long-term Survival: NO DIFFERENCE at 4-15 years. (EVAR early benefit is lost by 2-3 years). [9,10,11]
- Re-intervention: EVAR has 3-4x higher re-intervention rate. [9]
- Aneurysm-Related Mortality: Higher in EVAR group after 8 years due to late sac rupture. [9]
Prognostic Scoring Systems
Scoring systems are used to predict operative mortality and decide between Open Repair, EVAR, or Palliation (Turn Down).
1. Glasgow Aneurysm Score (GAS)
- Purpose: Predicts mortality in both elective and ruptured AAA.
- Variables: Age, Shock, Myocardial Disease, Cerebrovascular Disease, Renal Disease.
- Calculation: score = Age + 17(Shock) + 7(Myocardial) + 10(Cerebrovascular) + 14(Renal).
- Interpretation: Higher score = Higher mortality. (Score > 84 indicates prohibitive risk for open repair).
2. Hardman Index (Ruptured AAA)
- Purpose: Identify patients unlikely to survive repair.
- Variables (1 point each):
- Age > 76
- Creatinine > 190 µmol/L
- Hb less than 90 g/L
- ECG Ischaemia
- Loss of Consciousness
- Score interpretation:
- "Score 0: Low mortality."
- "Score ≥3: Mortality > 80% (Consider palliation)."
3. V-POSSUM
- Purpose: Vascular Physiological and Operative Severity Score for the enUmeration of Mortality.
- Complex weighted regression equation based on 12 physiological and 6 operative parameters.
Current Outcomes (Benchmark Data)
| Outcome Measure | Elective EVAR | Elective Open | Ruptured AAA (Overall) |
|---|---|---|---|
| 30-Day Mortality | 0.5 - 1.0% | 3.0 - 5.0% | 60 - 80% (Community) |
| 1-Year Survival | ~95% | ~92% | ~50% |
| Re-intervention (5yr) | 20 - 30% | 2 - 5% | N/A |
| Length of Stay | 1-2 days | 7-10 days | 14-21 days |
| QoL Return (Physical) | 2-4 weeks | 3-6 months | 6-12 months |
| Re-admission (30 day) | 5-10% | 5-10% | Variable |
| Long-term Risk | High (Endoleak) | Low (Hernia) | High |
| Cost Effectiveness | Higher cost (Device + Surveillance) | Lower cost (Bed days only) | Very High Cost |
Prognostic Factors
- Poor Prognosis Factors:
- "Presentation: Rupture (Shock is the single strongest predictor)."
- "Patient: Advanced Age (> 80), Female Sex, COPD (Home Oxygen), CKD (Dialysis)."
- "Anatomy: Short neck (EVAR failure), Suprarenal extension."
10. Evidence & Guidelines
Key Guidelines
-
ESVS - European Society for Vascular Surgery (2024) Link
- Key Rec: Strong recommendation AGAINST repair of less than 5.5cm AAAs in men.
- Key Rec: Centralisation of services (Minimum volume thresholds for surgeons/centres) improves outcomes.
- Key Rec: Ultrasound screening for all men 65+.
-
NICE - National Institute for Health and Care Excellence (2020) [NG156] Link
- Key Rec: Offer Open Repair for unruptured AAAs unless contraindicated (controversial, differs from US guidelines).
- Key Rec: Do not offer EVAR for complex aneurysms outside of trials.
-
SVS - Society for Vascular Surgery (USA, 2018)
- Key Rec: EVAR is preferred for ruptured aneurysm if anatomy suitable (lower mortality).
Landmark Trials
1. EVAR-1 Trial (15-Year Follow-Up Results) [PMID: 27743617]
- Design: 1252 patients randomised to EVAR vs Open Repair (1999-2004) in the UK.
- Key Finding:
- "Early: EVAR had 1.7% 30-day mortality vs 4.7% for Open ($p=0.009$)."
- "Mid-term (4 years): No difference in all-cause mortality."
- "Late (8+ years): Aneurysm-related mortality was SIGNIFICANTLY HIGHER in the EVAR group."
- Reason: Late sac rupture due to endoleaks and graft failure.
- Clinical Impact: Ended the "EVAR for everyone" era. Emphasised need for lifelong surveillance and re-intervention commitment. Validated that Open Repair is more durable.
2. EVAR-2 Trial (Patients Unfit for Open Repair) [PMID: 20598425]
- Design: Randomised "Unfit" patients to EVAR vs Medical Therapy.
- Key Finding: EVAR did NOT improve long-term survival compared to no surgery.
- Reason: These patients die of their comorbidities (heart failure, COPD, cancer) before the aneurysm ruptures.
- Clinical Impact: Major shift in practice. We now treat the patient, not the image. If a patient is too frail for open repair, they may also be too frail to benefit from EVAR.
3. OVER Trial (14-Year Follow-Up Results) [PMID: 31141639]
- Design: 881 US Veterans randomised. Newer generation grafts compared to EVAR-1.
- Key Finding: Survival was identical long-term.
- Re-intervention: Significantly higher in EVAR (26.7% vs 19.8%).
- Clinical Impact: Validated EVAR durability in better hands/devices but confirmed the re-intervention burden remains.
4. DREAM Trial (Dutch Randomized Endovascular Aneurysm Management) [PMID: 29061270]
- Design: Comparison of Open vs Endovascular repair.
- Key Finding: Confirmed the "catch-up" phenomenon where perioperative survival benefit of EVAR is lost by year 2 due to cardiovascular deaths and late graft failures. Freedom from re-intervention was 79% for Open vs 62% for EVAR at 12 years.
5. UK Small Aneurysm Trial (UKSAT) [PMID: 12443981]
- Design: Randomised 1090 patients with 4.0-5.5cm AAAs to Early Surgery vs Surveillance.
- Key Finding: No survival benefit to early surgery. 30-day operative mortality (5.8%) outweighed the low rupture risk of small aneurysms (1% per year).
- Clinical Impact: Established the 5.5 cm threshold globally.
6. IMPROVE Trial (Ruptured AAA Strategy) [PMID: 24411663]
- Design: 613 patients with Ruptured AAA randomised to Endovascular Strategy vs Open Repair.
- Key Finding: No significant difference in 30-day mortality (35% vs 37%). However, EVAR strategy led to faster discharge and better quality of life at 12 months. Women benefited significantly more from EVAR.
- Clinical Impact: EVAR is the preferred modality for rupture if anatomy permits and the centre is EVAR-ready.
Evidence Assessment
| Intervention | Level | Evidence Summary |
|---|---|---|
| Screening Men 65+ | 1a | MASS Trial (UK) showed 42% reduction in rupture death. |
| Smoking Cessation | 1a | Reduces growth and rupture risk. Strongest modifiable factor. |
| Elective Repair > 5.5cm | 1a | UK Small Aneurysm Trial, ADAM Trial: No benefit to early repair less than 5.5cm. |
| EVAR for Rupture | 1b | IMPROVE Trial, AJAX Trial: Trend towards better QoL, preferred in women. |
| Metformin Therapy | 2b | Some evidence it limits growth (anti-inflammatory), but not yet standard guideline. |
| Doxycycline | 2b | Evaluated for MMP inhibition. Failed to show benefit in large trials. |
Future Directions & Research
1. Pharmacotherapy (The "Holy Grail")
- Currently, no drug stops AAA growth.
- Metformin: Observational data suggests diabetic patients on Metformin have slower growth rates. Several RCTs (e.g., MAASt trial) are investigating this repurposing.
- Telmisartan: Angiotensin Receptor Blockers (ARBs) are being studied for their anti-inflammatory effects on the aortic wall.
2. Biomechanical Rupture Prediction
- Moving beyond "maximum diameter" (Law of Laplace) to Peak Wall Stress (PWS) analysis.
- Using Finite Element Analysis (FEA) on CT scans to identify specific weak points ("hotspots") in the wall. A 5.0cm aneurysm with a "blister" may have higher wall stress than a smooth 6.0cm one.
3. Next-Generation Endografts
- Polymer Sealing: The "Nellix" device filled the sac with polymer to prevent endoleaks (largely withdrawn due to failure, but concept remains).
- Low-Profile Devices: Allowing EVAR in women with smaller access vessels (less than 6mm).
4. Artificial Intelligence
- AI algorithms (like those used in lung nodule detection) are being trained to detect AAAs on incidental CT-KUBs or Spine MRIs where they are often missed by human reporters.
11. Patient/Layperson Explanation
What is an AAA?
"The aorta is the main pipe that carries blood from your heart down to your body. In your tummy, this pipe should be about 2cm wide (roughly the size of a garden hose). An aneurysm is when a section of this pipe becomes weak and balloons out. If it balloons too much, like an over-inflated tyre, there is a risk it could burst (rupture)."
Patient Frequently Asked Questions (FAQ)
1. Why did I get this? Is it because of what I ate? "The exact cause is a mix of genetics and environment. Smoking is the biggest factor—it damages the elastic proteins in the wall. High blood pressure and hardening of the arteries (atherosclerosis) also play a role. It is not directly caused by specific foods, but a healthy diet helps your overall blood vessels."
2. Is it hereditary? Should my children be checked? "Yes, there is a strong genetic link. If you have an AAA, your brothers and sisters are at higher risk. We recommend your brothers request a screening scan when they turn 55-60. Your children should also mention this to their doctor when they get older (50+)."
3. Can I feel it growing? "No. Most aneurysms are completely silent and painless as they grow. That is why we call it a 'silent killer' and why screening is so important. If you develop new back pain or tummy pain, you must seek medical attention immediately."
4. Is surgery dangerous? "All surgery has risks. For 'Keyhole' surgery (EVAR), the risk to life is very low (less than 1 in 100). For Open surgery, the risk is higher (3-5 in 100) because it is a major operation on the heart and lungs. However, the risk of not operating on a large aneurysm is much higher—if it bursts, the chance of survival is less than 20%."
5. How long will I be in hospital? "For keyhole repair (EVAR), most patients go home in 1 to 2 days. For open surgery, you will usually spend 1 night in intensive care and 5 to 7 days on the ward."
6. Can I drive after surgery? "You must stop driving until you can perform an emergency stop safely and without pain. This is usually 1 week for EVAR and 4-6 weeks for Open repair. You must inform the DVLA/DMV if you have a large aneurysm or after surgery depending on local laws."
7. Can I fly with an aneurysm? "Yes. Commercial cabin pressure does not increase the risk of rupture (physics confirms the wall tension change is negligible). However, check your travel insurance covers you for a pre-existing condition."
8. Will fixing it cure me forever? "If you have Open Repair, usually yes. If you have EVAR (Keyhole), you need life-long yearly scans. The graft can slip or leak ('endoleak') in about 20% of patients, which might need a minor correction procedure later."
9. What can I do to stop it growing? "The most powerful thing you can do is stop smoking completely. This is proven to slow growth. Taking your statin and blood pressure pills regularly also helps."
10. Can I exercise? "Yes. Moderate exercise like walking, cycling, or swimming is excellent for your heart and safe for the aneurysm. Avoid heavy weight-lifting or straining that causes you to hold your breath (Valsalva), as this spikes your blood pressure temporarily."
Why does it matter?
"A burst aneurysm is a life-threatening emergency with a very low survival rate. However, we can fix the pipe before it bursts with a planned operation, which is very safe. The goal is to catch it early."
How is it treated?
- Surveillance (Watch and Wait): "If the balloon is small (less than 5.5cm), the risk of surgery is higher than the risk of it bursting. We will just check it with an ultrasound every 3-12 months."
- Keyhole Repair (EVAR): "We put two stiff fabric tubes up through the blood vessels in your groin to line the inside of the balloon, reinforcing it from the inside."
- Open Surgery: "We make a cut in your tummy and replace the weak section of the pipe with a synthetic fabric tube."
When to seek help
"If you have a known aneurysm and get sudden severe tummy or back pain, you must call 999 immediately. This is a medical emergency."
12. References
Guidelines
- European Society for Vascular Surgery (ESVS). Clinical Practice Guidelines on the Management of Abdominal Aorto-Iliac Artery Aneurysms. European Journal of Vascular and Endovascular Surgery. 2024;67(1):1-79. Link
- National Institute for Health and Care Excellence (NICE). Abdominal aortic aneurysm: diagnosis and management [NG156]. 2020. Link
- Society for Vascular Surgery (SVS). Guidelines for care of patients with an abdominal aortic aneurysm. Journal of Vascular Surgery. 2018;67(1):2-77. Link
- European Society of Cardiology (ESC). ESC Guidelines on the diagnosis and treatment of aortic diseases. European Heart Journal. 2014;35(41):2873-2926. [PMID: 25173340]
Landmark Trials & Key Papers
- Patel R, et al. Endovascular versus open repair of abdominal aortic aneurysm in 15-years' follow-up of the UK endovascular aneurysm repair trial 1 (EVAR trial 1): a randomised controlled trial. Lancet. 2016;388(10058):2366-2374. [PMID: 27743617]
- Lederle FA, et al. Long-Term Comparison of Endovascular and Open Repair for Abdominal Aortic Aneurysm (OVER Trial). New England Journal of Medicine. 2019;380:1102-1111. [PMID: 31141639]
- De Bruin JL, et al. Long-term survival and secondary procedures after open or endovascular repair of abdominal aortic aneurysms (DREAM Trial). Journal of Vascular Surgery. 2017;66(5):1338-1350. [PMID: 29061270]
- Powell JT, et al. The Multicentre Aneurysm Screening Study (MASS) into the effect of abdominal aortic aneurysm screening on mortality in men: a randomised controlled trial. Lancet. 2002;360(9345):1531-9. [PMID: 12443981]
- Sweeting MJ, et al. Rupture rate of large abdominal aortic aneurysms in patients refusing or unfit for elective repair. British Journal of Surgery. 2012;99(2):213-8. [PMID: 22101982]
- Lederle FA, et al. The aneurysm detection and management study screening program: validation cohort and final results. Arch Intern Med. 2000;160(10):1425-30. [PMID: 10826454]
- Sveen EE, et al. The "Diabetes Paradox" in Abdominal Aortic Aneurysm. . Journal of Vascular Surgery. 2015.
- Chaikof EL, et al. The care of patients with an abdominal aortic aneurysm: The Society for Vascular Surgery practice guidelines. J Vasc Surg. 2009;50(4 Suppl):S2-49. [PMID: 19786250]
- Stather PW, et al. A systematic review and meta-analysis of the effect of intravenous contrast on renal function in patients with abdominal aortic aneurysm. Annals of Vascular Surgery. 2014. [PMID: 24041530]
- Brown LC, et al. Risk factors for aneurysm rupture in patients kept under ultrasound surveillance. UK Small Aneurysm Trial Participants. Annals of Surgery. 1999;230(3):289-97. [PMID: 10493476]
Additional References
- Wanhainen A, et al. European Society for Vascular Surgery (ESVS) 2019 Clinical Practice Guidelines on the Management of Abdominal Aorto-iliac Artery Aneurysms. Eur J Vasc Endovasc Surg. 2019;57(1):8-93. [PMID: 30528142]
- Sakalihasan N, et al. Abdominal Aortic Aneurysm. Lancet. 2005;365:1577-1589. [PMID: 15866312]
- Golledge J, et al. Abdominal aortic aneurysm: pathogenesis and implications for management. Arterioscler Thromb Vasc Biol. 2006;26(12):2605-13. [PMID: 16960104]
- Lindholt JS, et al. Hospital costs and patient survival of endovascular vs open repair of ruptured abdominal aortic aneurysms: cost-effectiveness analysis of the IMPROVE trial. BMJ. 2017. [PMID: 29146638]
- Reimerink JJ, et al. Systematic review and meta-analysis of population-based mortality from ruptured abdominal aortic aneurysm. Br J Surg. 2013;100(11):1405-13. [PMID: 24037560]
- Sweeting MJ, et al. Use of Weibull model to estimate rupture rates. Br J Surg. 2013. [PMID: 22101982]
13. Examination Focus
Viva Scenarios (Model Answers)
Scenario 1: The Screening Programme
Examiner: "Tell me about the UK AA Screening Programme." Candidate: "The NAAASP (National Abdominal Aortic Aneurysm Screening Programme) invites all men in their 65th year for a specific abdominal ultrasound scan.
- Inclusion: Men aged 65. Men > 65 can self-refer. Women are not screened (low prevalence).
- Outcomes:
- less than 3.0 cm: Discharge. No further screening needed (Risk of developing significant AAA later is negligible).
- 3.0 - 4.4 cm (Small): Surveillance annually.
- 4.5 - 5.4 cm (Medium): Surveillance every 3 months.
- ≥5.5 cm (Large): Referral to Vascular Surgeon within 2 weeks.
- Evidence: Based on the MASS Trial (Multicentre Aneurysm Screening Study), which showed a 42% relative risk reduction in AAA-related mortality over 10 years.
- Cost Effectiveness: Highly effective, approx £7,600 per QALY."
Scenario 2: The Unfit Patient
Examiner: "You have a 78-year-old man with a 6.2cm AAA. He has severe COPD (FEV1 30%), IHD with previous CABG, and Creatinine 150. How do you manage him?" Candidate: "This is a complex decision balancing the risk of rupture against the high risk of intervention.
- Assessment: I would assess his separation/physiological reserve. Given his severe COPD and Renal impairment, his perioperative mortality for Open Repair is likely prohibitive (> 10-20% predicted by GAS score).
- Anatomy: I would request a CTA to assess suitability for EVAR. EVAR avoids the respiratory stress of laparotomy and aortic cross-clamping.
- Renal Function: His creatinine of 150 is a concern for contrast nephropathy (EVAR) or ischaemia (Open). I would ensure pre-hydration.
- Decision Making:
- If EVAR suitable: I would offer EVAR as his only realistic curative option, discussing the lifelong surveillance burden.
- If EVAR unsuitable: I would have a frank discussion about 'Conservative Management'. Attempting Open Repair could likely kill him. Palliative/Conservative care is a valid active decision."
Scenario 3: Complications (Endoleak)
Examiner: "You see a patient in clinic 6 months after EVAR. His CT shows a Type 2 Endoleak. The sac has grown by 8mm. What do you do?" Candidate: "A Type 2 Endoleak is caused by retrograde flow from lumbar arteries or the IMA.
- Natural History: Many thrombose spontaneously. However, sac expansion > 5mm indicates pressurisation and requires treatment to prevent rupture.
- Management:
- Confirm Diagnosis: Ensure it is definitely Type 2 and not Type 1 (which would be an emergency). Delayed-phase CT or Angiogram is needed.
- Intervention: I would proceed to embolisation of the feeding vessel.
- Technique:
- Trans-arterial: Canulating the internal iliac -> iliolumbar -> lumbar artery to coil it.
- Direct Sac Puncture: Percutaneous injection of Onyx (glue) or coils into the nidus under CT guidance.
- Laparoscopic Ligation: (Rarely needed) Clipping the IMA/Lumbars."
Scenario 4: The Critical Appraisal (EVAR-1)
Examiner: "Does EVAR save lives compared to Open Repair?" Candidate: "Based on the EVAR-1 Trial 15-year data:
- Short Term: Yes. 30-day mortality is significantly lower (1.7% vs 4.7%). It is safer initially.
- Long Term: No. By 4 years, the survival curves converge. After 8 years, aneurysm-related mortality is actually higher in the EVAR group due to graft failures and late rupture.
- Conclusion: EVAR provides an early survival advantage and quicker recovery, but trades this for a long-term commitment to surveillance and higher re-intervention rates. It does not confer a long-term survival benefit over Open Repair."
Examiner's Checklist (The "Must Mention" Points)
- Smoking cessation is the only way to slow growth.
- Rupture triad is unreliable (only 50%).
- Permissive hypotension in rupture (don't pop the clot).
- EVAR needs suitable anatomy (Neck, Access).
- Women rupture at smaller diameters (5.0 cm threshold).
- "Trash Foot" is distal embolisation.
- Colonic ischaemia presents as bloody diarrhoea.
Common Exam Questions
- MRCP/FRCS: "A 72-year-old man presents with collapse and back pain. His BP is 80/50. What is your immediate management?"
- Answer: Activate Major Haemorrhage Protocol, permissive hypotension, immediate vascular surgery referral/theatre. Do not delay for CT if diagnosis is highly suspected and patient unstable (though most centres will try to get a CTA if patient transiently responds).
- Medical Student: "What are the screening criteria for AAA in the UK?"
- Answer: Men aged 65 by ultrasound.
- FRCS: "Discuss the pros and cons of EVAR versus Open Repair for a 65-year-old fit man."
- Answer: EVAR has lower short-term risk but higher long-term re-intervention and rupture risk. Open repair is more durable. For a fit 65-year-old with 20-year life expectancy, Open Repair is often preferred (NICE guidelines support this, though practice varies).
- Pharmacology: "Why are ACE inhibitors used in AAA?"
- Answer: Hypertension control, and some evidence they may reduce MMP activity and wall remodeling (though trial evidence is mixed).
- Anatomy: "At what level does the abdominal aorta bifurcate?"
- Answer: L4 (Umbilicus level).
Viva Points
Opening Statement:
"Abdominal Aortic Aneurysm is a degenerative condition, defined as aortic dilation > 3cm. It is significant because of the risk of rupture, which carries an 80% mortality. Management focuses on screening men over 65, surveillance for small aneurysms, and elective repair for those > 5.5cm to prevent this catastrophic complication."
Key Facts to Mention:
- 5.5cm threshold: The key decision point for men.
- Permissive Hypotension: The key to survival in rupture.
- Diabetes Paradox: Diabetic patients have lower incidence of AAA.
- EVAR-1 Trial: Mention that EVAR durability fails long-term compared to Open.
Classification to Quote:
- "I would classify this based on anatomy into Infrarenal (95%) or Juxtarenal/Suprarenal, as this dictates the operative approach."
Evidence to Cite:
- "The ESVS 2024 guidelines recommend against repairing small aneurysms."
- "The MASS trial proved that screening reduces aneurysm-related mortality by ~40%."
Common Mistakes
What fails candidates:
- ❌ Aggressive Fluids: "Pumping" a rupture patient with 2L saline raising BP to 120/80 (causes death).
- ❌ Missing the diagnosis: Diagnosing "renal colic" in a 75-year-old without feeling the abdomen.
- ❌ Confusing thresholds: Quoting 5.0cm for men (too low) or 6.0cm (too high).
- ❌ Ignoring family history: Failing to advise screening for brothers/sisters.
Examiner Follow-Up Questions
- "The patient is 85 and frail with a 6cm AAA. Would you offer surgery?"
- Answer: "This is a complex decision. The EVAR-2 trial showed that in unfit patients, EVAR did not improve survival over medical therapy alone. I would determine his specific biological fitness and use a shared decision-making model. If extremely frail, medical management is appropriate."
- "What is a Type II Endoleak?"
- Answer: "It is retrograde filling of the aneurysm sac from lumbar arteries or the IMA. It is the most common endoleak and often benign, but requires monitoring."
- "Why do you not screen women?"
- Answer: "The prevalence is much lower (less than 1%), so it is not cost-effective. However, women who smoke or have cardiovascular disease may benefit from opportunistic screening."
Last Updated: 2026-01-02
Medical Disclaimer: MedVellum content is for educational purposes and clinical reference. Clinical decisions should account for individual patient circumstances. Always consult appropriate specialists and current guidelines.
About MedVellum
MedVellum is a global open-access medical education platform designed for postgraduate trainees and clinicians. Our content is:
- Evidence-Based: Strictly adhering to major international guidelines (NICE, ESC, AHA, ESVS).
- Peer-Reviewed: Verified by senior clinicians and subject matter experts.
- Exam-Focused: Tailored for high-stakes examinations (MRCP, FRCS, USMLE).
- Free Open Access Medical Education (FOAMed): Committed to democratising medical knowledge.
Version History
- v1.0: Initial Draft (AI Generated).
- v2.0: Clinical Review.
- v3.0: Gold Standard Expansion (Jan 2026) - Current Version.
- Added Molecular Pathophysiology.
- Added Deep Vascular Anatomy.
- Added Detailed Operative Steps (EVAR/Open).
- Added Viva Scenarios and Model Answers.
Frequently asked questions
Quick clarifications for common clinical and exam-facing questions.
When should I seek emergency care for abdominal aortic aneurysm (aaa) in adults?
Seek immediate emergency care if you experience any of the following warning signs: Severe, sudden-onset abdominal or back pain (rupture), Hypotension/syncope with abdominal pain (rupture), Pulsatile abdominal mass with tenderness (imminent rupture), Flank ecchymosis (Grey Turner's sign - retroperitoneal bleed), New onset lower limb ischaemia (distal embolisation).
Learning map
Use these linked topics to study the concept in sequence and compare related presentations.
Prerequisites
Start here if you need the foundation before this topic.
- Aortic Anatomy
- Cardiovascular Physiology
Differentials
Competing diagnoses and look-alikes to compare.
- Renal Colic
- Diverticulitis
- Musculoskeletal Back Pain
Consequences
Complications and downstream problems to keep in mind.
- Aortic Rupture
- Distal Embolisation