Abdominal Aortic Aneurysm
Abdominal aortic aneurysm is a focal dilatation of the abdominal aorta, most often infrarenal, that matters because it can remain silent for years and then present catastrophically with rupture. This page gives a...
What matters first
Abdominal aortic aneurysm is a focal dilatation of the abdominal aorta, most often infrarenal, that matters because it can remain silent for years and then present catastrophically with rupture. This page gives a...
Sudden severe abdominal, back, flank, or groin pain
25 Apr 2026
Generated educational material; verify before clinical use.
15 cited sources
See the concept before reading it
Study the key anatomy, imaging, and decision pathways as full teaching plates.
Clinical board
A visual summary of the highest-yield teaching signals on this page.
Urgent signals
Safety-critical features pulled from the topic metadata.
- Sudden severe abdominal, back, flank, or groin pain
- Collapse, syncope, or new hypotension
- Known AAA with new pain
- Pulsatile abdominal mass
Exam focus
Current exam surfaces linked to this topic.
- ACEM Fellowship
- CICM Fellowship
- FRCS / FRACS Vascular Surgery
Linked comparisons
Differentials and adjacent topics worth opening next.
- Renal Colic (Adult)
- Acute Myocardial Infarction
- Ruptured Abdominal Aortic Aneurysm
- Aortic Dissection
Content status and exam context
This page is AI-generated educational content. It may contain errors or omissions and is not a substitute for current guidelines, local protocols, senior clinical judgement, or professional medical advice.
MedVellum does not claim an individual clinician reviewer, board certification, or professional credential for this page unless a future version names a real, verifiable reviewer.
Clinical explanation and evidence
Abdominal Aortic Aneurysm
Answer Card
An abdominal aortic aneurysm is an abdominal aortic diameter of 3.0 cm or more, or a focal dilatation at least 50% larger than expected. Most clinically important AAAs are infrarenal. [1-3]
Think rupture in any older patient, especially an ever-smoker or hypertensive patient, with new abdominal or back pain, collapse, loss of consciousness, or shock. Immediate bedside ultrasound is appropriate if rupture is suspected. [2]
Standard infrarenal AAAs are usually considered for repair at 5.5 cm, if symptomatic, or if growth exceeds 1 cm in 1 year. Women rupture more often at smaller diameters, so threshold discussions should be individualized. [1-3,6]
EVAR has lower early operative mortality in suitable anatomy, but needs lifelong imaging and has more late graft-related reinterventions. Open repair is more invasive but more durable in fit patients. [1-3,10-12]
Ruptured AAA is a vascular time-critical diagnosis. Do not wait for the classic triad. Severe new abdominal, back, flank, or groin pain plus collapse, hypotension, known AAA, or an older smoking history is enough to activate immediate imaging and vascular discussion. NICE specifically recommends considering rupture in people with new abdominal/back pain, cardiovascular collapse, or loss of consciousness, especially when risk factors are present. [2]
Why This Topic Matters
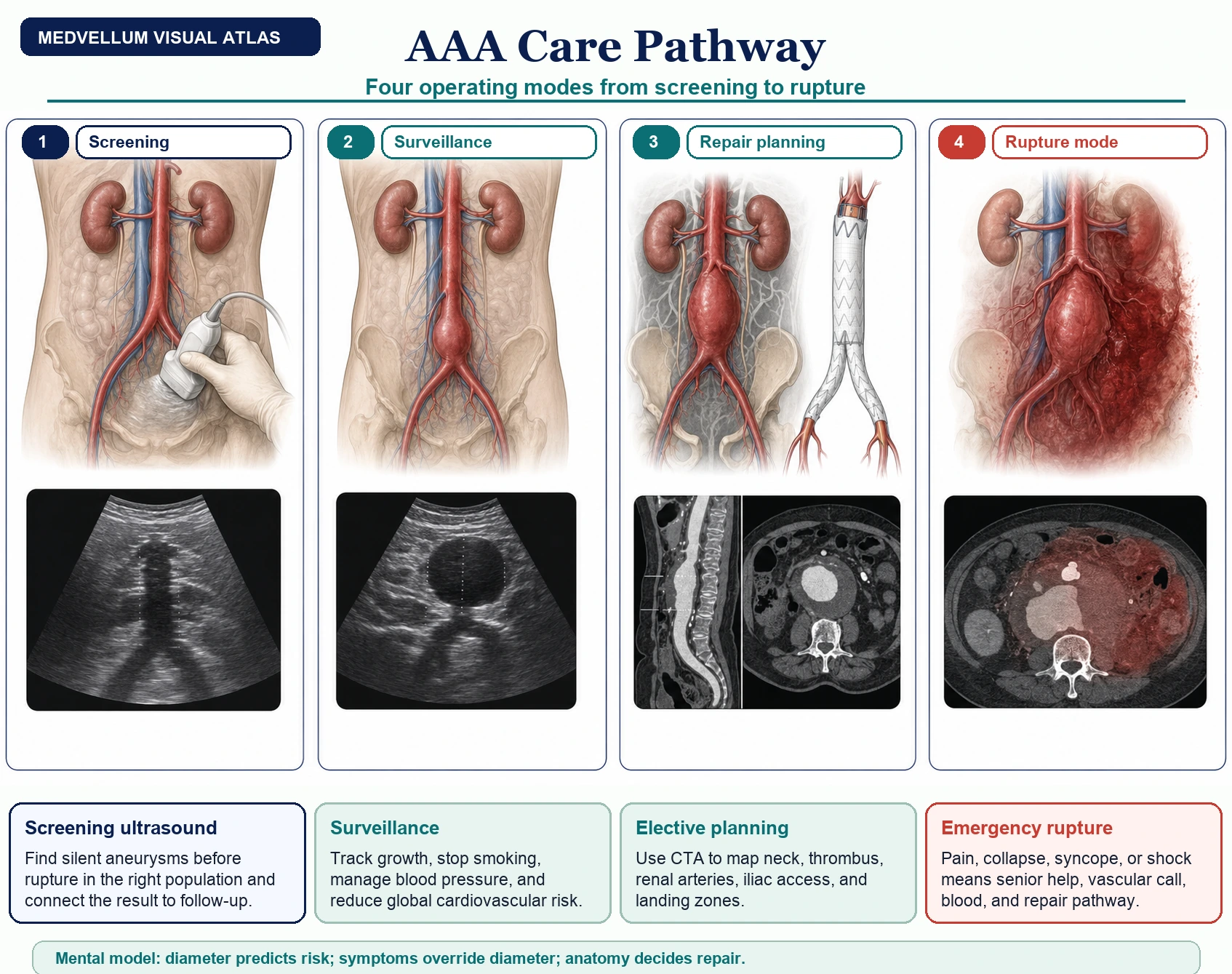
AAA is a perfect visual medicine problem: one disease moves between screening, surveillance, elective operative planning, and immediate rupture resuscitation. Good care starts by recognizing which mode you are in, then switching the whole pathway around that state. [1-5]
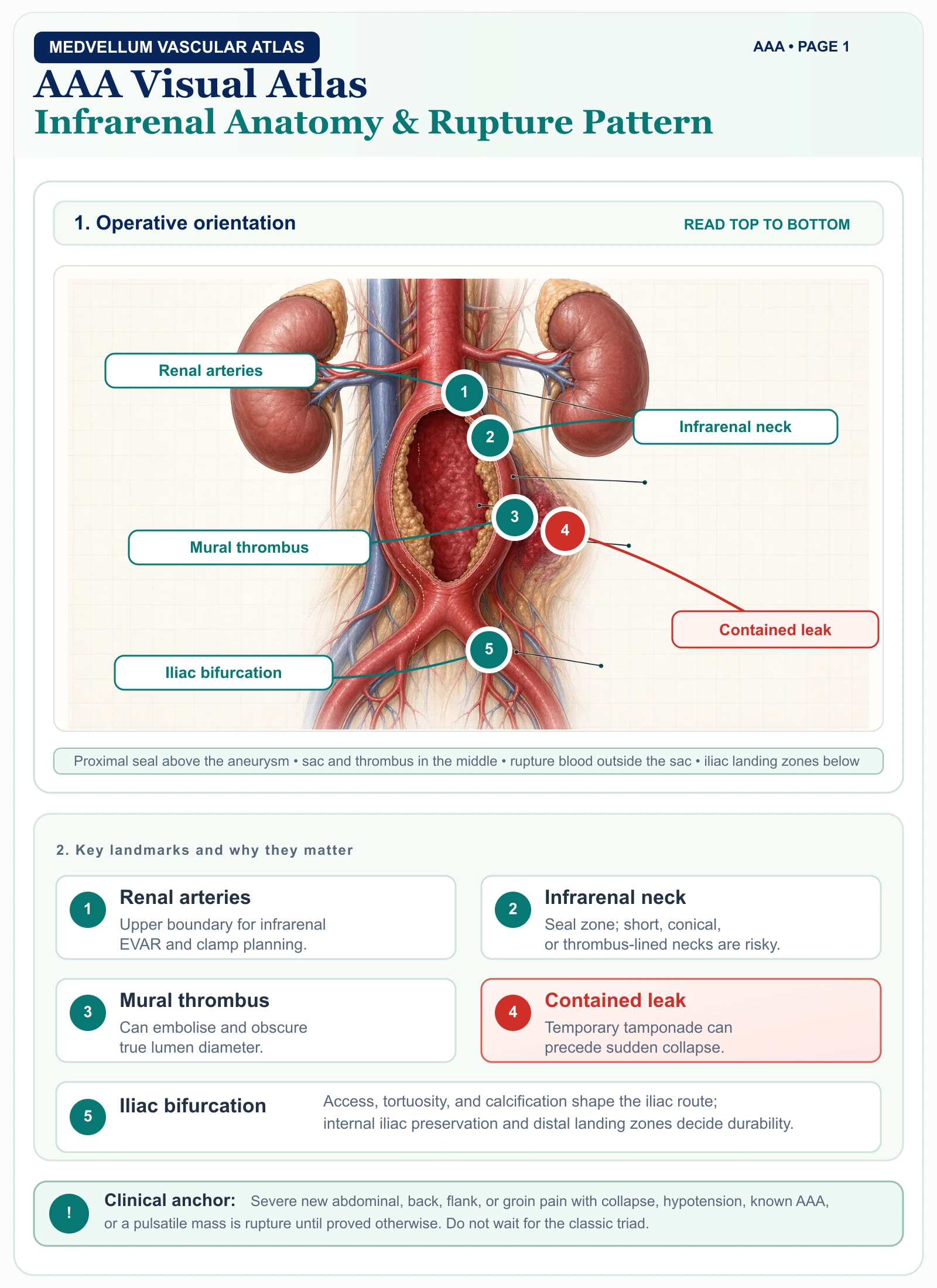
Anatomy You Must See
Most AAAs arise below the renal arteries and above the aortic bifurcation. That infrarenal location explains why standard EVAR is possible: the endograft needs a normal proximal neck below the renal arteries and adequate iliac landing zones.
| Structure | Why it matters |
|---|---|
| Renal arteries | Define the upper boundary for standard infrarenal EVAR. Short, conical, thrombus-lined, or angulated necks increase type Ia endoleak risk. |
| Inferior mesenteric artery | Often sacrificed during repair; colonic perfusion depends on collateral circulation. |
| Lumbar arteries | Common source of type II endoleak after EVAR. |
| Common/internal iliac arteries | Iliac aneurysm or poor access can change device planning and pelvic ischaemia risk. |
| Third part of duodenum | Site of aorto-enteric fistula, especially after prior aortic grafting. |
| Retroperitoneum | Rupture may be temporarily contained, creating deceptive transient stability. |
The infrarenal neck is the business end of EVAR planning. Ask five questions every time you read the CT angiogram: Is the neck long enough, narrow enough, straight enough, free enough of thrombus/calcification, and parallel enough to seal?
Neck length, diameter, angulation, thrombus, and calcification determine whether the device can seal below the renal arteries.
Fusiform, saccular, inflammatory, infected, or rapidly expanding aneurysms do not carry the same risk or planning assumptions.
Tortuous, calcified, narrow, or aneurysmal iliac arteries can block device delivery or require adjunctive iliac procedures.
Lumbar and inferior mesenteric branches explain type II endoleak; renal and internal iliac branches set the danger zones.
Pathophysiology In One Diagram
Initiation: smoking, age, hypertension, family history, atherosclerotic burden, and inherited matrix biology injure the aortic wall. Smoking is the strongest modifiable risk factor. [1-4]
Inflammation: macrophages, lymphocytes, cytokines, oxidative stress, and protease signalling accumulate in the aortic media and adventitia. [13]
Matrix failure: elastin fragmentation, collagen remodelling, smooth muscle cell loss, and matrix metalloproteinase activity reduce wall strength. [13]
Biomechanical escalation: as diameter increases, wall stress rises. A larger, thinner, stiffer aneurysm is less tolerant of pressure and pulsatile load.
Clinical consequence: slow asymptomatic enlargement, embolisation from mural thrombus, tenderness from expansion, or rupture into the retroperitoneum/peritoneum.
Risk Factors And Protective Signals
Risk rises strongly after 60 years. Screening systems usually target older men because prevalence is highest in this group. [2,4]
The dominant modifiable risk factor for development, growth, and rupture. Offer smoking cessation support to every smoker with AAA. [1-4]
AAA is less common in women, but rupture risk is higher once an aneurysm is present, so threshold discussions need individualization. [2,6]
First-degree relatives have increased risk. Document family history and consider earlier imaging according to local policy. [1-3]
Treat blood pressure and global cardiovascular risk in line with standard prevention guidance; rupture and cardiovascular mortality travel together. [2]
Diabetes is associated with lower AAA prevalence and slower expansion, but it is not protective enough to reassure anyone with pain or rupture symptoms. [14]
Screening works because AAA is common enough in selected older male populations, ultrasound is accurate and non-invasive, and elective repair is safer than emergency rupture repair. The MASS randomized trial showed reduced aneurysm-related mortality from population screening, and later surveillance meta-analysis refined how frequently small aneurysms need follow-up. [4,5]
Presentation
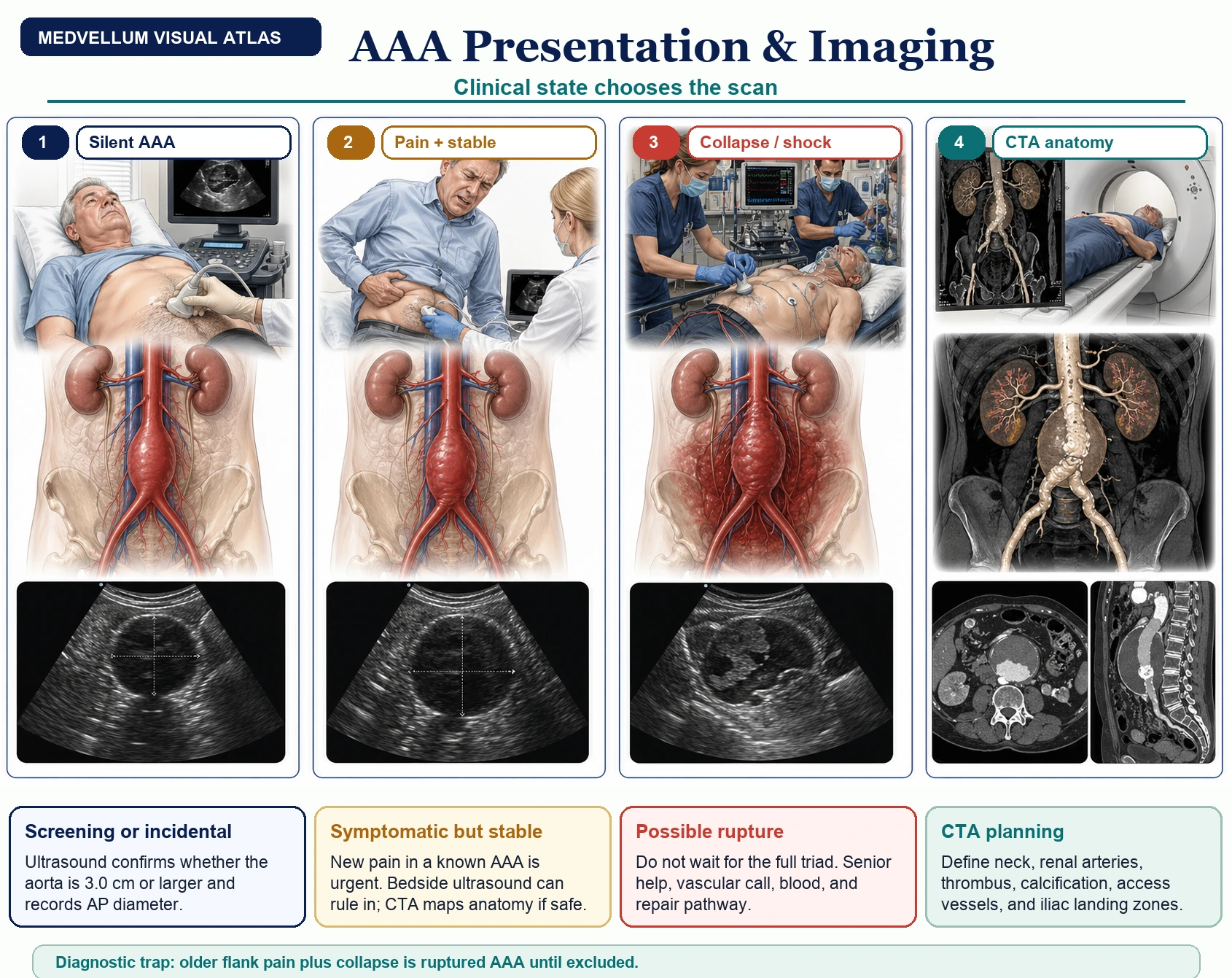
Most AAAs are found by screening ultrasound or unrelated imaging before symptoms appear.
New pain in a known AAA suggests expansion, tenderness, inflammatory aneurysm, embolisation, infection, or contained leak. Treat it as urgent.
Back, flank, groin, abdominal, or testicular pain with collapse, syncope, hypotension, or shock should activate the vascular pathway. [2]
Renal colic, lumbar pain, GI bleeding, sepsis, or unexplained shock can all hide AAA rupture, especially in older ever-smokers. [2]
Renal colic trap: older patient + flank pain + collapse, hypotension, or vascular risk factors = exclude AAA before treating the presentation as uncomplicated ureteric colic.
Diagnosis And Imaging
Ultrasound answers the first question: is the aorta 3.0 cm or larger, and what is the maximum AP diameter?
Bedside ultrasound is the fastest rule-in test. If suspicion remains after a non-diagnostic scan, keep the vascular pathway moving. [2]
Arterial-phase CTA is the anatomy map: neck, renal arteries, thrombus, calcification, access vessels, iliac landing zones. [1-3]
CTA is useful if the patient can tolerate it and it changes repair strategy; unstable physiology still needs senior vascular decision-making.
| Situation | First useful test | What you need to know |
|---|---|---|
| Screening or incidental asymptomatic AAA | Ultrasound | Maximum anterior-posterior diameter using the local measurement convention. NICE specifies inner-to-inner AP diameter in the NHS screening programme. [2] |
| Suspected symptomatic or ruptured AAA | Bedside aortic ultrasound immediately if available | If AAA is seen, or ultrasound is unavailable/non-diagnostic but suspicion remains, discuss immediately with the regional vascular service. [2] |
| Elective repair planning | Thin-slice arterial-phase CT angiography | Neck length/diameter/angulation, renal and iliac anatomy, access vessels, thrombus, calcification, and suitability for EVAR/open/complex repair. [1-3] |
| Suspected rupture and candidate for repair | CT angiography if it will not delay definitive care | Confirms rupture, morphology, access, and EVAR feasibility. Unstable patients still need senior vascular decision-making in real time. [1,2,11] |
Surveillance And Repair Decision Pathway
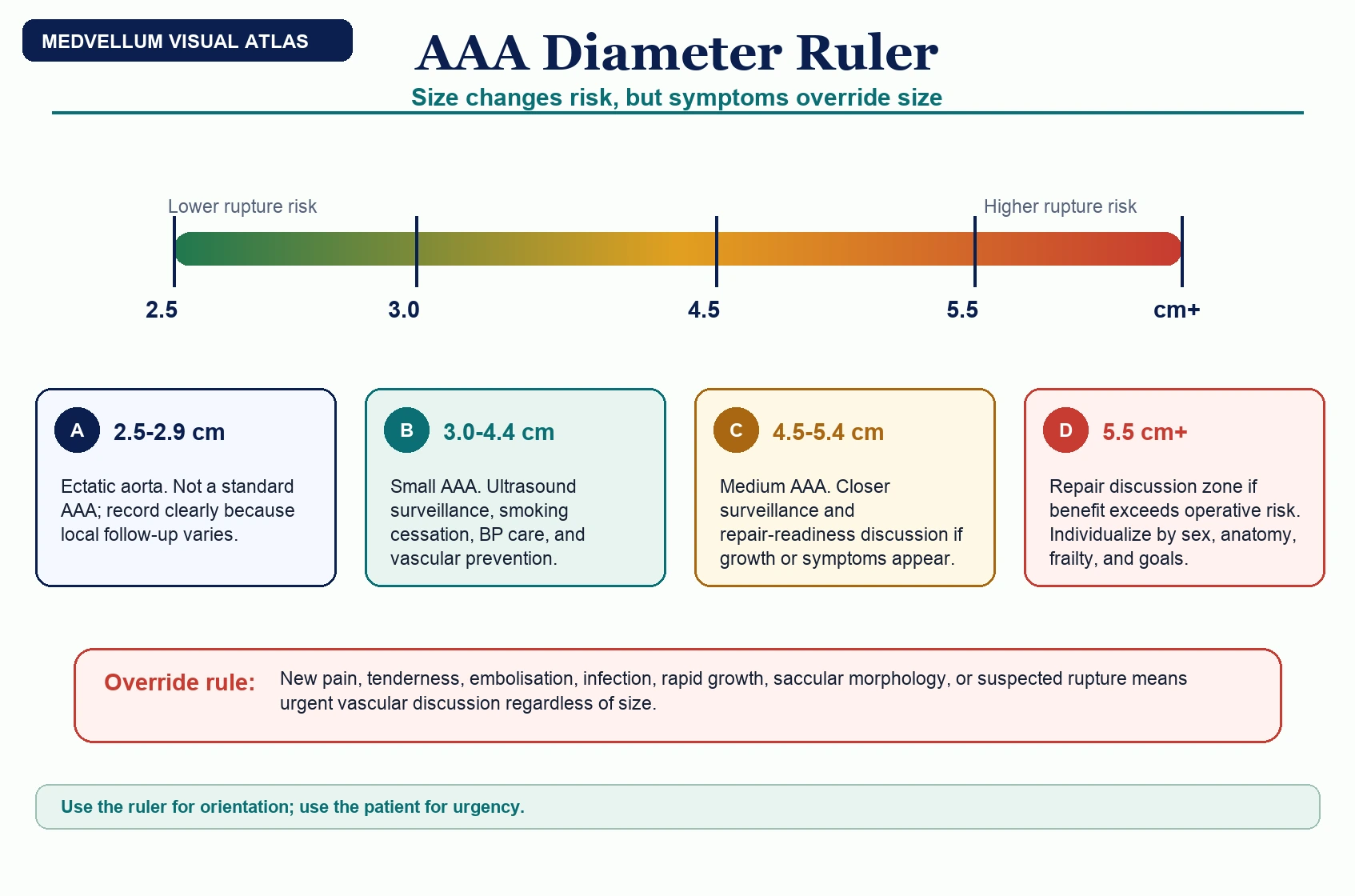
Aorta less than 3.0 cm: no AAA by standard definition. Re-enter screening only if local screening policy or a new clinical indication applies. [1-3]
AAA 3.0-4.4 cm: ultrasound surveillance. Optimize cardiovascular prevention, smoking cessation, and blood pressure care. [1-3,5]
AAA 4.5-5.4 cm: closer ultrasound surveillance and vascular clinic review. Check growth rate, symptoms, sex, morphology, and operative fitness. [1-3,5]
Any symptom, rapid growth, saccular/inflammatory/mycotic morphology, or diameter 5.5 cm or more: discuss repair rather than routine surveillance. [1-3]
Repair planning: CT angiography, cardiopulmonary/anaesthetic assessment, shared decision-making, anatomy review, and choice between EVAR, open repair, complex EVAR, or conservative management. [1-3]
After repair: EVAR requires lifelong surveillance for endoleak, migration, sac expansion, limb occlusion, and graft infection. Open repair follow-up focuses on graft complications and cardiovascular risk. [1-3,10-12]
Use size bands as a ruler, not a permission slip. A 5.5 cm asymptomatic infrarenal AAA often enters repair discussion, but any painful, tender, infected, rapidly growing, saccular, embolising, or possibly ruptured aneurysm needs urgent vascular review regardless of diameter. [1-3]
Management
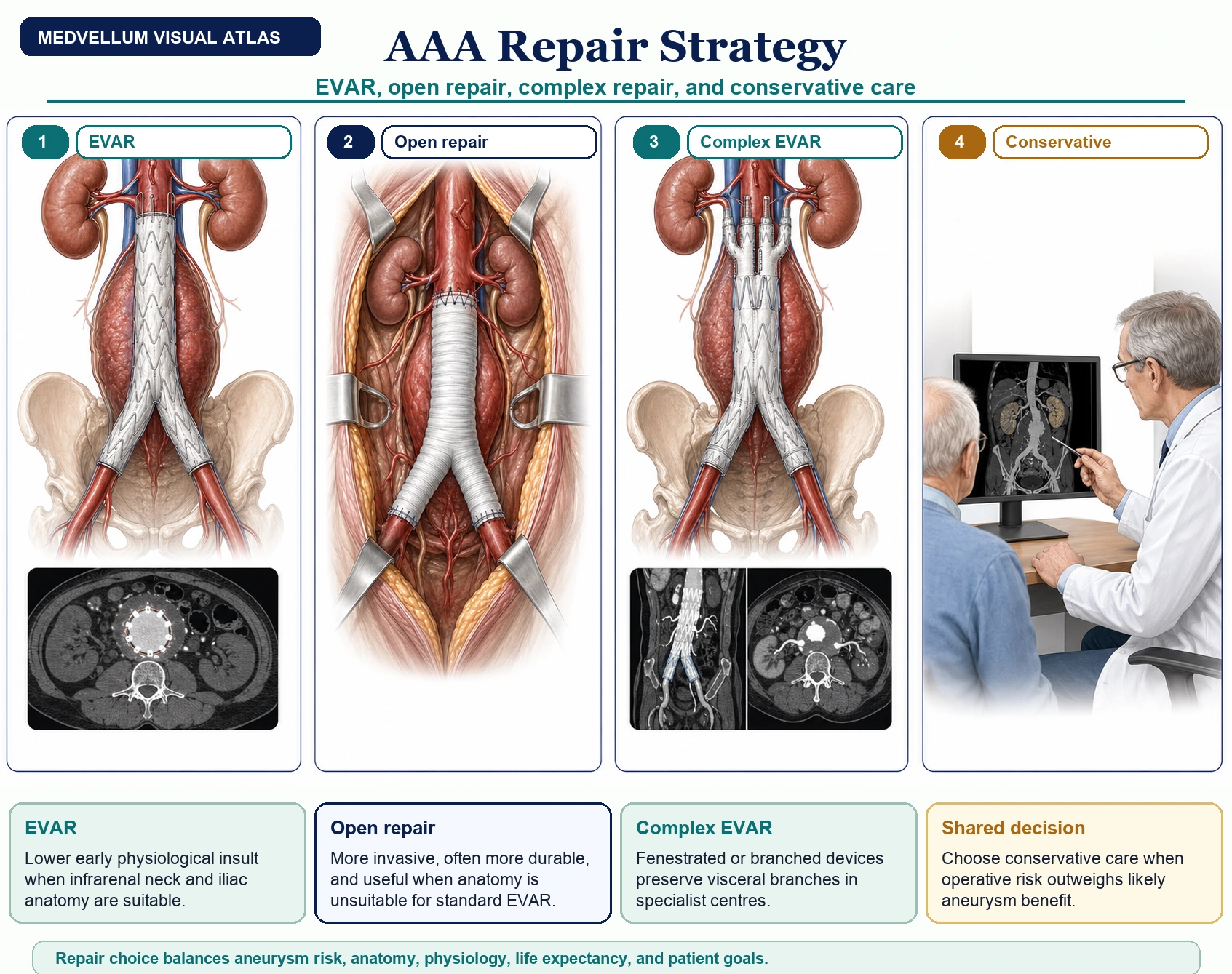
Everyone With AAA
Explain diagnosis, rupture risk, surveillance plan, and symptoms that require urgent care. [2]
Offer smoking cessation; treat blood pressure and global cardiovascular risk according to standard prevention guidance. [2]
Record the measurement convention because small differences matter near thresholds. NICE specifies inner-to-inner AP diameter in the NHS screening programme. [2]
Clarify family history and advise first-degree relatives to discuss risk assessment with their clinician. [1-3]
Stable Elective AAA
Decision-making should combine:
- Aneurysm diameter and growth rate.
- Symptoms.
- Sex-specific rupture considerations.
- Neck and iliac anatomy.
- Patient frailty, cardiopulmonary reserve, renal function, and life expectancy.
- Patient goals and tolerance of lifelong surveillance/reintervention risk.
| Repair option | Best fit | Trade-off |
|---|---|---|
| EVAR | Suitable infrarenal anatomy, higher open surgical risk, hostile abdomen, need for lower early physiological insult | Lower early operative mortality in trials, but more late imaging, endoleaks, reinterventions, and device durability concerns. [10-12] |
| Open repair | Fit patient, long expected survival, anatomy unsuitable for standard EVAR, or preference for durable repair | More invasive with higher early physiological burden, but less late graft-related reintervention than EVAR in randomized follow-up. [10-12] |
| Complex EVAR / fenestrated / branched | Juxtarenal, pararenal, or complex anatomy in specialist centres | Requires high-volume expertise, careful consent, and rigorous follow-up. [1,2] |
| Conservative management | Operative risk outweighs likely aneurysm benefit | Must include clear safety-netting, risk reduction, and shared decision documentation. [2] |
Suspected Ruptured AAA
Rupture mode is not normal shock mode. If rupture is suspected, call the regional vascular service early, obtain immediate bedside ultrasound if available, avoid delays for non-essential tests, and consider permissive hypotension during transfer when appropriate. NICE recommends leaving the referring unit within 30 minutes after acceptance for emergency vascular assessment. [2]
Older patient, ever-smoker, known AAA, sudden abdominal/back/flank/groin pain, collapse, syncope, or unexplained shock.
Senior help, vascular call, anaesthesia, blood bank, immediate bedside aortic ultrasound if it does not delay transfer.
Avoid reflex crystalloid normalization. Use blood products and restrictive resuscitation/permissive hypotension when appropriate. [2]
EVAR-capable or open vascular repair pathway, CTA only when physiology and local workflow make it useful rather than delaying repair.
Key steps:
- Senior ED/acute clinician, vascular surgeon, anaesthetist, and radiology involvement.
- Two large-bore IV lines or rapid infuser access, blood products available, group and crossmatch.
- Immediate bedside aortic ultrasound if it can be done without delaying transfer.
- CT angiography if stable enough and if it informs repair strategy.
- Restrictive volume resuscitation/permissive hypotension during transfer when clinically appropriate. [2]
- Choose endovascular strategy vs open repair based on anatomy, physiology, team skill, and local pathway. [1,2,11]
Complications To Look For
Before Repair
- Rupture or contained leak.
- Distal embolisation, including blue toe syndrome.
- Aorto-caval fistula: high-output cardiac failure, machinery abdominal bruit, venous congestion.
- Aorto-enteric fistula: herald GI bleed followed by collapse.
- Mycotic aneurysm: fever, inflammatory markers, rapid growth, saccular morphology.
After EVAR
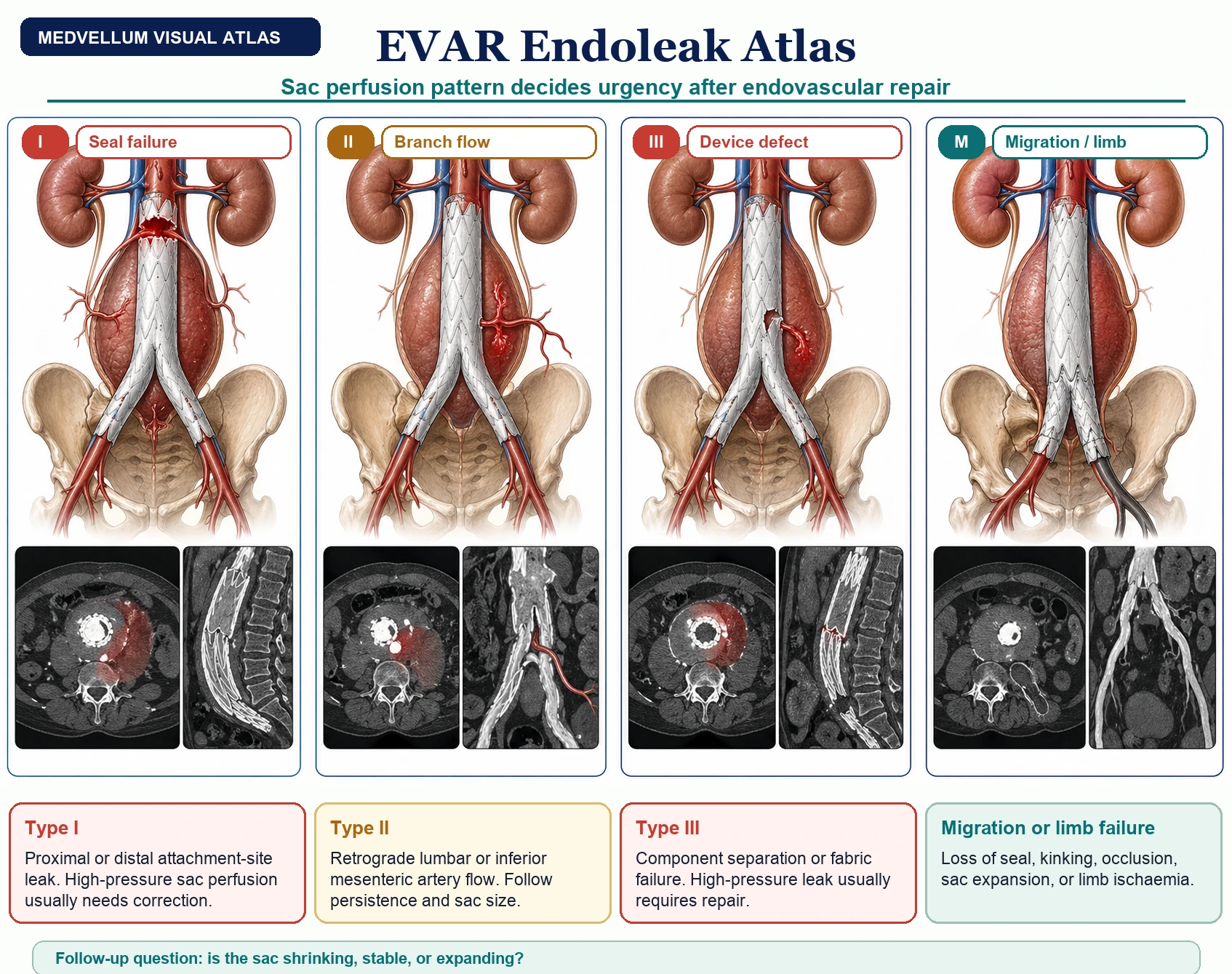
| Complication | Pattern |
|---|---|
| Type I endoleak | Inadequate seal at proximal or distal attachment site; high-pressure and usually needs urgent correction. |
| Type II endoleak | Retrograde flow from lumbar or inferior mesenteric branches; monitor sac size and persistence. |
| Type III endoleak | Device defect or component separation; high-pressure and usually requires repair. |
| Migration/kinking/limb occlusion | Presents as sac expansion, loss of seal, or limb ischaemia. |
| Graft infection | Rare but catastrophic; fever, pain, sepsis, perigraft gas/fluid. |
Exam And Viva Framework
Five sentences that pass most AAA viva stations:
- AAA is usually an infrarenal aortic diameter of 3.0 cm or more.
- Most AAAs are asymptomatic until expansion or rupture.
- Rupture must be considered in older patients with new abdominal/back pain, collapse, or shock.
- Elective repair is usually considered at 5.5 cm, rapid growth, or symptoms, but women and complex morphology require individualized discussion.
- EVAR lowers early operative stress in suitable anatomy but trades that for lifelong surveillance and more late graft-related reintervention.
OSCE case: A 72-year-old man with a 50 pack-year smoking history presents with sudden left flank pain, sweating, and near-syncope. BP is 88/54 mmHg. The urine dip has trace blood.
Expected candidate actions: recognize ruptured AAA as a cannot-miss diagnosis; call senior help; request immediate bedside aortic ultrasound if available; alert vascular/anaesthetic teams; obtain bloods and crossmatch; avoid anchoring on renal colic; avoid aggressive crystalloid resuscitation; arrange urgent vascular transfer or CT angiography according to stability and local pathway.
Patient Explanation
"An abdominal aortic aneurysm means the main artery in the abdomen has become widened. Small aneurysms are usually watched with ultrasound because the risk of an operation can be greater than the risk of rupture. Larger aneurysms, fast-growing aneurysms, or aneurysms causing pain need specialist review because repair may become safer than waiting. If you have known AAA and develop sudden severe tummy, back, flank, or groin pain, collapse, or fainting, call emergency services."
References
- Wanhainen A, Van Herzeele I, Bastos Goncalves F, et al. European Society for Vascular Surgery (ESVS) 2024 Clinical Practice Guidelines on the Management of Abdominal Aorto-Iliac Artery Aneurysms. Eur J Vasc Endovasc Surg. 2024;67(2):192-331.
- National Institute for Health and Care Excellence. Abdominal aortic aneurysm: diagnosis and management. NICE guideline NG156. Published 19 March 2020.
- Chaikof EL, Dalman RL, Eskandari MK, et al. The Society for Vascular Surgery practice guidelines on the care of patients with an abdominal aortic aneurysm. J Vasc Surg. 2018;67(1):2-77.e2.
- Ashton HA, Buxton MJ, Day NE, et al. The Multicentre Aneurysm Screening Study (MASS) into the effect of abdominal aortic aneurysm screening on mortality in men. Lancet. 2002;360(9345):1531-1539.
- RESCAN Collaborators. Surveillance intervals for small abdominal aortic aneurysms: a meta-analysis. JAMA. 2013;309(8):806-813.
- Sweeting MJ, Thompson SG, Brown LC, Powell JT. Meta-analysis of individual patient data to examine factors affecting growth and rupture of small abdominal aortic aneurysms. Br J Surg. 2012;99(5):655-665.
- Lederle FA, Wilson SE, Johnson GR, et al. Immediate repair compared with surveillance of small abdominal aortic aneurysms. N Engl J Med. 2002;346(19):1437-1444.
- United Kingdom Small Aneurysm Trial Participants. Long-term outcomes of immediate repair compared with surveillance of small abdominal aortic aneurysms. N Engl J Med. 2002;346(19):1445-1452.
- Thompson SG, Brown LC, Sweeting MJ, et al. Systematic review and meta-analysis of growth and rupture rates of small abdominal aortic aneurysms. Health Technol Assess. 2013;17(41):1-118.
- United Kingdom EVAR Trial Investigators. Endovascular versus open repair of abdominal aortic aneurysm. N Engl J Med. 2010;362(20):1863-1871.
- IMPROVE Trial Investigators. Endovascular strategy versus open repair for ruptured abdominal aortic aneurysm: three year results. BMJ. 2017;359:j4859.
- van Schaik TG, Yeung KK, Verhagen HJ, et al. Long-term survival and secondary procedures after open or endovascular repair of abdominal aortic aneurysms. J Vasc Surg. 2017;66(5):1379-1389.
- Golledge J, Muller J, Daugherty A, Norman P. Abdominal aortic aneurysm: pathogenesis and implications for management. Arterioscler Thromb Vasc Biol. 2006;26(12):2605-2613.
- De Rango P, Farchioni L, Fiorucci B, Lenti M. Diabetes and abdominal aortic aneurysms. Eur J Vasc Endovasc Surg. 2014;47(3):243-261.
- Lederle FA, Johnson GR, Wilson SE, et al. The aneurysm detection and management study screening program: validation cohort and final results. Arch Intern Med. 2000;160(10):1425-1430.
Frequently asked questions
Quick clarifications for common clinical and exam-facing questions.
What size defines an abdominal aortic aneurysm?
An abdominal aortic aneurysm is usually defined as an abdominal aortic diameter of 3.0 cm or greater, or a focal dilatation at least 50% larger than expected.
When is an AAA usually considered for elective repair?
Repair is usually considered when the aneurysm is symptomatic, reaches 5.5 cm, or grows by more than 1 cm in a year, with individualized decision-making for sex, anatomy, operative fitness, and local guidance.
What symptoms suggest rupture?
New severe abdominal, back, flank, or groin pain with collapse, hypotension, known AAA, or a pulsatile mass should be treated as possible rupture until excluded.
Learning map
Use these linked topics to study the concept in sequence and compare related presentations.
Prerequisites
Start here if you need the foundation before this topic.
Related Topics
Adjacent pages worth reading next.
Differentials
Competing diagnoses and look-alikes to compare.
Consequences
Complications and downstream problems to keep in mind.