Acute Porphyria
The acute porphyrias are a group of rare, inherited metabolic disorders characterized by life-threatening "neurovisceral" attacks. These conditions result from partial deficiencies in enzymes of the heme biosynthetic...
What matters first
The acute porphyrias are a group of rare, inherited metabolic disorders characterized by life-threatening "neurovisceral" attacks. These conditions result from partial deficiencies in enzymes of the heme biosynthetic...
Respiratory paralysis / Breathlessness
4 Jan 2026
Generated educational material; verify before clinical use.
Visible references section
See the concept before reading it
Study the key anatomy, imaging, and decision pathways as full teaching plates.
Clinical board
A visual summary of the highest-yield teaching signals on this page.
Urgent signals
Safety-critical features pulled from the topic metadata.
- Respiratory paralysis / Breathlessness
- Severe hyponatraemia (less than 125 mmol/L)
- Bulbar palsy / Dysphagia
- Status epilepticus
Linked comparisons
Differentials and adjacent topics worth opening next.
- Hyponatraemia
- Peripheral Neuropathy
Content status and exam context
This page is AI-generated educational content. It may contain errors or omissions and is not a substitute for current guidelines, local protocols, senior clinical judgement, or professional medical advice.
MedVellum does not claim an individual clinician reviewer, board certification, or professional credential for this page unless a future version names a real, verifiable reviewer.
Clinical explanation and evidence
Acute Porphyria
1. Clinical Overview
Summary
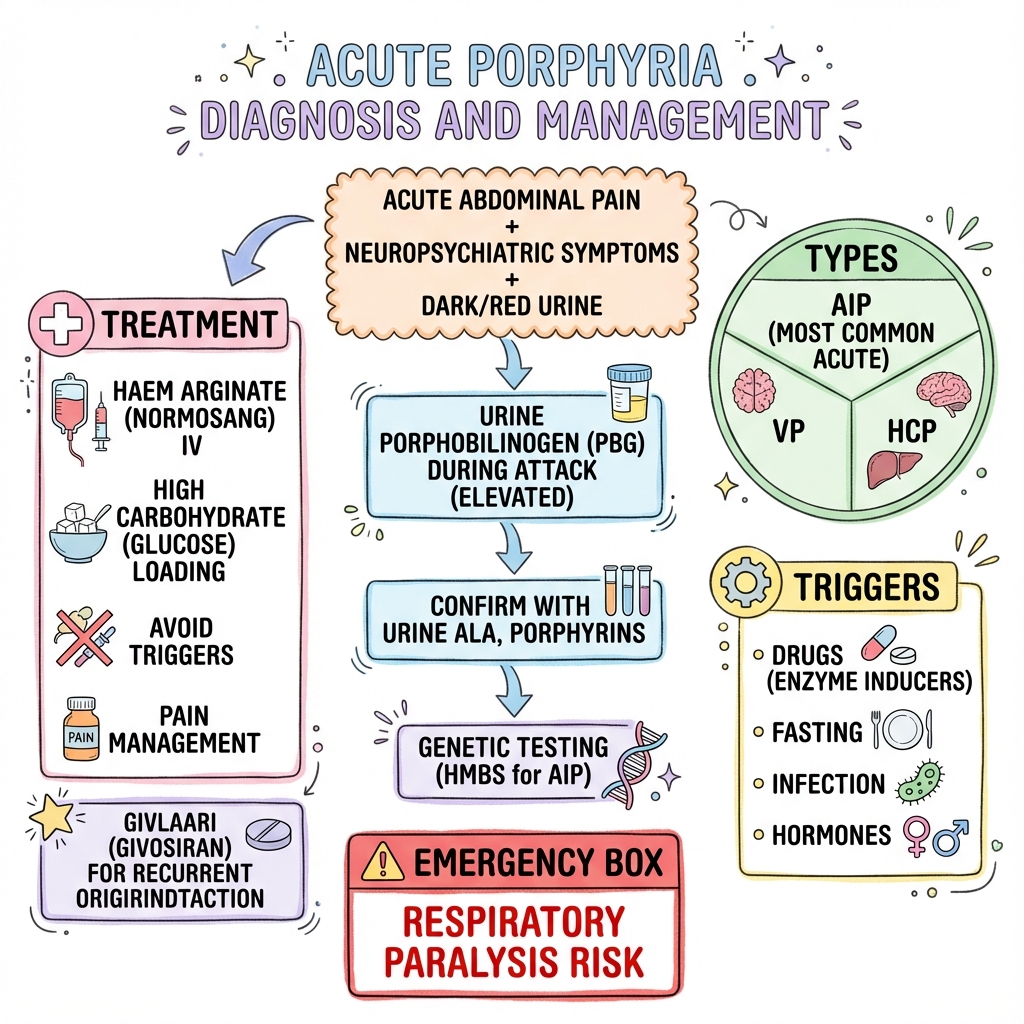
The acute porphyrias are a group of rare, inherited metabolic disorders characterized by life-threatening "neurovisceral" attacks. These conditions result from partial deficiencies in enzymes of the heme biosynthetic pathway, leading to the accumulation of toxic heme precursors, specifically delta-aminolevulinic acid (ALA) and porphobilinogen (PBG). The most common and clinically significant form is Acute Intermittent Porphyria (AIP).
Attacks are typically precipitated by external triggers—most notably porphyrinogenic drugs, fasting, hormonal changes, and stress—which upregulate hepatic ALA synthase-1 (ALAS1). Known as "The Little Imitator," acute porphyria presents with a bewildering array of symptoms: severe abdominal pain (often without peritonitis), neuropsychiatric disturbances, autonomic dysfunction, and peripheral neuropathy. Early recognition is critical; if untreated, attacks can progress to respiratory paralysis or irreversible neurological damage. Management hinges on the rapid administration of intravenous heme arginate and the absolute avoidance of precipitating triggers.
Key Facts
- Definition: Inherited defects in heme synthesis causing accumulation of neurotoxic precursors (ALA/PBG).
- The "Little Imitator": Frequently misdiagnosed as surgical abdomen, appendicitis, or psychiatric illness.
- Classic Tetrad: Abdominal pain + Neuropsychiatric symptoms + Hyponatraemia + Dark urine.
- Pathognomonic Finding: Marked elevation of Urinary PBG (essential for diagnosis during an attack).
- Heme Pathway Hook: Deficiency in PBG Deaminase (also known as Hydroxymethylbilane synthase) is the hallmark of AIP.
- Triggers: Cytochrome P450 inducers (barbiturates, sulfonamides), fasting (low carbohydrate), and progesterone.
- Emergency Management: IV Heme Arginate (Normosang) 3-4 mg/kg/day for 4 days.
- Prevention: Givosiran (RNA interference therapy) is the first-line preventive for recurrent attacks.
Clinical Pearls
Diagnostic Pearl: The "Fresh Urine Test". In an acute attack, urine may appear normal but turns dark or "port-wine" colored when exposed to light and air as PBG polymerizes to porphobilin.
Safety Pearl: NEVER give barbiturates or sulfonamides to a patient with suspected porphyria. Use a dedicated database (e.g., www.drugs-porphyria.org) for every prescription.
Pitfall Warning: Acute porphyria causes "Pain out of proportion to signs." A patient screaming in agony with a soft, non-tender abdomen and no fever should trigger immediate porphyria screening.
Physiological Pearl: The "Carbohydrate Effect". Glucose suppresses hepatic ALAS1 transcription. In mild attacks, high carbohydrate loading (300g/day) may abort the crisis.
2. Epidemiology
Incidence & Prevalence
- Prevalence: Clinically overt acute porphyria is rare (approx. 1-2 per 100,000 in Europe).
- Genetic Prevalence: The prevalence of HMBS mutations (AIP) is much higher (approx. 1 in 1,700), indicating low penetrance (~1-10%). Most carriers never experience an attack.
- Geographic Variation:
- AIP: Most common worldwide; particularly high in Northern Sweden (1 in 1,000 due to founder effect).
- Variegate Porphyria (VP): Highly prevalent in the South African Afrikaner population (1 in 300) due to a founder effect.
- Hereditary Coproporphyria (HCP): Rare globally.
Demographics
- Age: Attacks are rare before puberty and after menopause. Peak incidence is in the 3rd and 4th decades (ages 20-45).
- Sex: Significant female predominance (approx. 4:1 to 5:1), largely due to the role of cyclic hormonal triggers (progesterone) and oral contraceptive use.
- Race: Affects all ethnic groups; VP is specifically frequent in South Africans of Dutch descent.
Risk Factors & Triggers
| Trigger Category | Examples | Mechanism |
|---|---|---|
| Pharmacological | Barbiturates, Sulfonamides, Rifampicin, Carbamazepine, Phenytoin | Induction of Cytochrome P450 enzymes depletes hepatic heme, triggering ALAS1 feedback upregulation. |
| Endocrine | Progesterone, Luteal phase of menstrual cycle, Pregnancy | Direct induction of ALAS1 mRNA in the liver. |
| Nutritional | Fasting, Low-calorie diets, Gastric bypass, Liquid diets | Starvation induces PGC-1α, a potent co-activator of ALAS1. |
| Toxic/Lifestyle | Alcohol (binge drinking), Smoking, Organic solvents | Oxidative stress and P450 induction. |
| Physiological | Infection, Surgery, Psychological stress | Systemic metabolic demand and cortisol-mediated ALAS1 induction. |
3. Pathophysiology
The Heme Biosynthetic Pathway
Heme synthesis is an 8-step pathway occurring partially in the mitochondria and partially in the cytosol.
- Rate-Limiting Step (Mitochondria): Glycine + Succinyl-CoA $\rightarrow$ $\delta$-Aminolevulinic Acid (ALA).
- Enzyme: ALA Synthase (ALAS).
- ALAS1 (Liver): Regulated by heme levels (negative feedback).
- ALAS2 (Erythroid): Regulated by iron levels.
- Step 2 (Cytosol): 2 molecules of ALA $\rightarrow$ Porphobilinogen (PBG).
- Enzyme: ALA Dehydratase (ALAD). (Inhibited by Lead).
- Step 3 (The AIP Defect): 4 molecules of PBG $\rightarrow$ Hydroxymethylbilane (HMB).
- Enzyme: PBG Deaminase (HMBS) (also called Hydroxymethylbilane Synthase).
- In AIP, this enzyme is ~50% deficient.
- Step 4: HMB $\rightarrow$ Uroporphyrinogen III.
- Enzyme: Uroporphyrinogen III Synthase. (Defect = Congenital Erythropoietic Porphyria).
- Step 5: Uroporphyrinogen III $\rightarrow$ Coproporphyrinogen III.
- Enzyme: Uroporphyrinogen Decarboxylase. (Defect = Porphyria Cutanea Tarda).
- Step 6 (Mitochondria): Coproporphyrinogen III $\rightarrow$ Protoporphyrinogen IX.
- Enzyme: Coproporphyrinogen Oxidase (CPOX). (Defect = HCP).
- Step 7: Protoporphyrinogen IX $\rightarrow$ Protoporphyrin IX.
- Enzyme: Protoporphyrinogen Oxidase (PPOX). (Defect = VP).
- Step 8: Protoporphyrin IX + $Fe^{2+}$ $\rightarrow$ Heme.
- Enzyme: Ferrochelatase. (Defect = Erythropoietic Protoporphyria).
Mechanism of the Acute Attack
- Enzyme Deficiency: A partial block exists (e.g., HMBS in AIP). Under basal conditions, the 50% enzyme activity is sufficient to maintain heme levels.
- Upregulation of ALAS1: A trigger (e.g., a drug or fasting) demands more heme or increases ALAS1 production.
- Precursor Accumulation: The pathway is "flooded" from the top. The defective enzyme becomes a bottleneck.
- Toxic Overflow: Massive amounts of ALA and PBG accumulate in the liver and spill into the plasma and urine.
Neurotoxicity
The exact mechanism of neurological damage involves:
- Direct Neurotoxicity of ALA: ALA is structurally similar to $\gamma$-aminobutyric acid (GABA) and can act as a GABA receptor agonist or antagonist, disrupting inhibitory neurotransmission.
- Oxidative Stress: ALA auto-oxidation generates reactive oxygen species (ROS), causing axonal degeneration and Schwann cell damage.
- Heme Deficiency: Local heme deficiency in nerve cells may impair mitochondrial function, cytochrome P450 function, and tryptophan metabolism.
Pathological Classification
| Condition | Enzyme Defect | Primary Accumulation | Features |
|---|---|---|---|
| AIP | HMBS (PBG Deaminase) | ALA, PBG | Purely neurovisceral; No skin signs. |
| VP | PPOX | ALA, PBG, Protoporphyrin | Neurovisceral + Skin photosensitivity. |
| HCP | CPOX | ALA, PBG, Coproporphyrin | Neurovisceral + Skin photosensitivity. |
| ALAD Deficiency | ALAD | ALA (Normal PBG) | Extremely rare; mimics lead poisoning. |
4. Clinical Presentation
"The Little Imitator"
Acute porphyria is notorious for mimicking other conditions. Diagnosis is often delayed by years (median delay 15 years).
1. Visceral Symptoms (90% of attacks)
- Abdominal Pain: Severe, diffuse, poorly localized. Often described as "tearing" or "cramping."
- Character: Constant pain with sudden exacerbations.
- Examination: Remarkably soft abdomen without guarding or rebound (no peritonitis).
- Associated: Nausea, vomiting, severe constipation (ileus due to autonomic neuropathy), and urinary retention.
2. Autonomic Dysfunction (80% of attacks)
- Tachycardia: Often the earliest and most consistent sign (often > 100 bpm).
- Hypertension: Can be labile or sustained; reflects sympathetic overactivity.
- Sweating, Tremor, and Low-grade fever.
3. Neurological Manifestations (20-50% of attacks)
- Peripheral Neuropathy: Primarily motor, axonal, and symmetric. Often starts in the proximal upper limbs (shoulder girdle) rather than distal legs.
- Progression: Can rapidly evolve into quadriparesis and respiratory failure (mimicking Guillain-Barré).
- CNS: Seizures (often exacerbated by hyponatraemia), confusion, hallucinations, and coma.
4. Psychiatric Symptoms
- Anxiety, agitation, depression, or overt psychosis.
- Many patients are historically misdiagnosed with "conversion disorder," "hysteria," or "somatization."
5. Electrolyte Imbalance
- Hyponatraemia: Present in 25-40% of attacks. Caused by SIADH (from hypothalamic damage) or salt-wasting from vomiting and renal losses. Worsening hyponatraemia can trigger seizures.
5. Clinical Examination
Structured Approach
- Vital Signs: Look for tachycardia and hypertension (autonomic overactivity).
- Abdominal Exam:
- Inspection: May see distension (ileus).
- Palpation: Soft, non-tender or mildly tender. Crucial finding: Lack of peritonism despite severe pain.
- Neurological Exam:
- Motor: Proximal muscle weakness (shoulder girdle/thighs). Check for reduced or absent reflexes (though they may be preserved initially).
- Cranial Nerves: Check for bulbar palsy (dysphagia, dysphonia) – a sign of impending respiratory failure.
- Sensory: Less common, but "bathing suit" distribution of sensory loss can occur.
- Skin (VP and HCP only): Look for blisters, erosions, milia, and scarring on sun-exposed areas (back of hands, face). AIP has NO skin involvement.
- Urine Inspection: Observe color. Freshly voided urine is usually pale but may darken to a reddish-brown "burgundy" or "port-wine" color upon standing in light.
6. Investigations
The Diagnostic Gold Standard
Rule 1: In a suspected acute attack, the ONLY test that matters initially is Spot Urinary PBG.
1. First-Line Tests (During Attack)
- Urinary PBG (Spot): Must be quantitative. Levels are typically 20–100 times the upper limit of normal during an attack.
- Note: Levels may decrease between attacks, so testing must occur during symptoms.
- Light Protection: Samples must be wrapped in foil and sent to the lab immediately.
- Urinary ALA: Also markedly elevated.
- Urine Color: Darkening on exposure to UV light.
2. Specialist Differentiation (After PBG confirmation)
- Plasma Porphyrin Scan:
- AIP: Usually normal.
- VP: Pathognomonic fluorescence peak at 624-628 nm.
- HCP: Peak at 618 nm.
- Fecal Porphyrins: Useful to distinguish VP (high protoporphyrin) from HCP (high coproporphyrin III).
3. Genetic Testing
- Role: Once the biochemical diagnosis is made, genetic sequencing (e.g., HMBS gene) is performed to identify the specific mutation.
- Clinical Utility: Essential for family screening (identifying asymptomatic carriers).
4. Bedside/Routine Labs
- Sodium: Hyponatraemia is a key clue.
- WCC/CRP: Usually normal (helps rule out infection/inflammation).
- LFTs: May show mild transaminitis.
- Imaging: Abdominal X-ray may show dilated loops of bowel (ileus). CT abdomen is usually unremarkable (done to rule out other causes).
7. Management
Management Algorithm (The "Porphyria Protocol")
Acute Abdominal Pain + Neuro/Psych Symptoms
↓
Check Spot Urine PBG (Protect sample from light)
↓
┌────────────────┴────────────────┐
PBG Elevated (> 5x ULN) PBG Normal
(Acute Porphyria Attack) (Search for other causes)
↓
1. ADMIT to hospital.
2. ELIMINATE triggers (Check all drugs).
3. ANALGESIA (Opioids are safe).
4. ANTI-EMETICS (Ondansetron safe).
↓
┌────────────────┴────────────────┐
Mild Attack (No weakness/Low Na) Severe Attack (Weakness/Seizures/High PBG)
- High Carbohydrate (300g/day) - IV HEME ARGINATE (Normosang)
- Monitor Neuro status q4h - 3-4 mg/kg IV daily for 4 days
- If no improvement in 24h → Heme - Central line access (Vein irritant)
↓
Long-term: Genetic counseling, Trigger avoidance, Givosiran if recurrent.
Acute Attack Management
1. Immediate Stabilization
- Fluid Resuscitation: Use 0.9% Saline. Avoid hypotonic fluids (risk of worsening hyponatraemia).
- Nutrition: If tolerated, high carbohydrate diet. If NBM, IV 10% Dextrose (at least 300g glucose/24h). Note: Glucose is supportive but not a substitute for heme in severe attacks.
2. Specific Therapy: Heme Arginate (Normosang)
- Mechanism: Provides exogenous heme, which exerts negative feedback on ALAS1 via heme-repressor protein, rapidly shutting down the production of ALA and PBG.
- Dose: 3-4 mg/kg (max 250mg) IV daily for 4 days.
- Administration: Must be diluted in Human Albumin (minimizes phlebitis) and given via a large vein or central line.
- Contraindications: Hypersensitivity to heme.
3. Symptom Control (Safe Drugs)
- Pain: Morphine, Pethidine, Fentanyl. (Avoid NSAIDs if renal function is impaired).
- Nausea: Ondansetron, Prochlorperazine.
- Agitation: Chlorpromazine, Lorazepam.
- Tachycardia/HTN: Propranolol (Beta-blockers are safe).
- Seizures: Levetiracetam (Keppra) or Gabapentin. Warning: Phenytoin and Carbamazepine are highly unsafe.
Pharmacology: Safe vs. Unsafe Drugs
| SAFE | UNSAFE (Avoid) |
|---|---|
| Paracetamol, Aspirin, Ibuprofen | Barbiturates (Thiopental, Phenobarbital) |
| Morphine, Fentanyl, Oxycodone | Sulfonamides (Sulfamethoxazole/Trimethoprim) |
| Penicillins, Cephalosporins, Gentamicin | Anticonvulsants (Phenytoin, Carbamazepine, Valproate) |
| Ondansetron, Domperidone | Hormones (Progesterone, Oral Contraceptives) |
| Heparin, Warfarin | Antibiotics (Rifampicin, Nitrofurantoin, Clarithromycin) |
| Insulin, Metformin | Anaesthetics (Etomidate, Ketamine) |
| Beta-blockers, ACE Inhibitors | Calcium Channel Blockers (Nifedipine - use with caution) |
8. Differential Diagnosis
| Condition | Porphyria Mimicry | Key Differentiators |
|---|---|---|
| Acute Appendicitis | Abdominal pain, vomiting. | Appendicitis has localized tenderness (McBurney's), fever, and raised WCC. |
| Guillain-Barré Syndrome | Ascending motor paralysis. | GBS usually has distal onset, protein-cytological dissociation in CSF, and normal PBG. |
| Lead Poisoning | Abdominal colic, neuropathy, ALA elevation. | Lead poisoning has "lead lines" on gums, basophilic stippling, and normal PBG. |
| Conversion Disorder | Bizarre neuro/psych symptoms. | Porphyria has objective markers: hyponatraemia, tachycardia, and elevated PBG. |
| Acute Psychosis | Agitation, hallucinations. | Primary psychosis lacks the visceral pain and autonomic signs of porphyria. |
9. Complications
Acute Complications
- Respiratory Paralysis: Progression of motor neuropathy to the diaphragm. Requires urgent mechanical ventilation.
- Seizures: Often multifactorial (ALA toxicity + hyponatraemia).
- Wernicke's Encephalopathy: Can be precipitated by high glucose loading in malnourished patients (give Thiamine).
- Aspiration Pneumonia: Secondary to bulbar palsy and impaired swallowing.
Long-term Complications
- Chronic Hypertension: Develops in > 50% of AIP patients due to chronic autonomic damage.
- Chronic Kidney Disease: Multifactorial (hypertension + nephrotoxicity of ALA).
- Hepatocellular Carcinoma (HCC): Risk is significantly increased in AIP (60-fold increase). Annual ultrasound and AFP screening is recommended after age 50, even in asymptomatic carriers.
- Chronic Neuropathic Pain: Some patients develop chronic neuropathy or "smouldering" symptoms between attacks.
10. Prognosis & Outcomes
- Mortality: Historically high (25%), but now less than 5% in developed centers due to heme arginate and better ICU care.
- Recovery: Most patients recover fully from the visceral and psychiatric symptoms within days of starting heme.
- Neuropathy Recovery: Nerve regeneration is slow (1mm/day). Severe motor weakness may take 6–12 months to improve, and some residual deficit is possible if treatment was delayed.
- Recurrence:
- Sporadic: Most patients have only one or two attacks in their lifetime.
- Recurrent: 3-5% of patients (mostly women) suffer from "Recurrent Acute Porphyria" (> 4 attacks per year). This is often linked to the menstrual cycle.
11. Evidence & Guidelines
Landmark Trials & Therapies
- Givosiran (ENVISION Trial, 2020):
- Design: Phase 3 RCT of Givosiran (RNAi targeting ALAS1) vs. Placebo in patients with recurrent attacks.
- Outcome: 74% reduction in annualized attack rate.
- Impact: Established Givosiran as the gold standard for prevention of recurrent porphyria.
- The "Carbohydrate Effect": While widely practiced, evidence shows glucose is significantly less effective than heme. Guidelines (ASPC/BPA) now emphasize early heme rather than prolonged glucose trials in severe attacks.
- Liver Transplantation: For intractable, life-threatening cases failing all medical therapy. It "cures" the metabolic defect in the liver (the source of ALA/PBG).
Guidelines
- British Porphyria Association (BPA): Standard protocols for emergency admission and safe drug lists.
- Porphyrias Consortium (USA): Expert consensus on diagnosis, management, and long-term HCC surveillance.
- European Porphyria Network (EPNET): International registry and standardized diagnostic criteria.
12. Patient Explanation
"Your body uses a 'production line' to make heme, which is the part of your blood that carries oxygen. In your case, one of the machines on this production line is working at half-speed. Usually, this isn't a problem. However, certain triggers—like some medications, fasting, or hormones—try to speed up the production line. Because of the slow machine, the 'raw materials' (called ALA and PBG) start to pile up. These raw materials are toxic to your nerves. This causes the severe pain and other symptoms you are feeling. The treatment we give (Heme) tells your body to stop the production line immediately, allowing the toxic levels to drop."
13. References
- Puy H, Gouya L, Deybach JC. Porphyrias. Lancet. 2010;375(9718):924-937. [PMID: 20226990]
- Balwani M, et al. Phase 3 Trial of Givosiran for Acute Intermittent Porphyria. N Engl J Med. 2020;382(24):2289-2301. [PMID: 32521132]
- Stein PE, et al. Acute intermittent porphyria: fatal complications of a treatable disease. Lancet. 2013;382(9896):1000.
- Bonkovsky HL, et al. Porphyrin and heme metabolism and the porphyrias. Hepatology. 2013;58(4):1199-1201.
- The European Porphyria Network (EPNET). Drug Database for Acute Porphyria. www.drugs-porphyria.org
- Anderson KE, et al. Recommendations for the diagnosis and molecular genetic testing of the acute porphyrias. Ann Intern Med. 2005.
14. Examination Focus
Viva Questions & Model Answers
Q1: What is the most important first step in managing a patient with suspected acute porphyria? A: The most critical step is to obtain a spot urine sample for quantitative PBG measurement (protected from light) and simultaneously check the patient's current medications against a porphyria-safe drug database to eliminate any potential triggers.
Q2: A patient with known AIP presents with seizures. Which anticonvulsant would you use? A: I would use Levetiracetam (Keppra) or Gabapentin, as they are proven to be safe. I would strictly avoid Phenytoin, Carbamazepine, and Sodium Valproate, as they strongly induce the heme pathway and will catastrophically worsen the attack.
Q3: Explain the "Carbohydrate Effect" in porphyria. A: Glucose and other carbohydrates suppress the transcription of hepatic ALAS1 by inhibiting the co-activator PGC-1$\alpha$. This reduces the production of neurotoxic precursors (ALA/PBG). It is used as a supportive measure in mild attacks.
Q4: Why does hyponatraemia occur in acute porphyria? A: It is primarily due to SIADH caused by toxic damage to the hypothalamus or posterior pituitary, but can be exacerbated by gastrointestinal losses and poor intake.
MRCP/USMLE Style Pearls
- Proximal motor weakness + Abdominal pain = Porphyria until proven otherwise.
- Normal WCC in a patient with severe abdominal pain is a strong negative predictor for surgical causes and should raise suspicion of porphyria.
- Variegate Porphyria vs. AIP: If the question mentions skin blisters/photosensitivity, the answer is VP or HCP, not AIP.
- The 624nm Peak: If you see "fluorescence at 624nm" in a question, they are testing for Variegate Porphyria.
Last Updated: 2026-01-04
Frequently asked questions
Quick clarifications for common clinical and exam-facing questions.
When should I seek emergency care for acute porphyria?
Seek immediate emergency care if you experience any of the following warning signs: Respiratory paralysis / Breathlessness, Severe hyponatraemia (less than 125 mmol/L), Bulbar palsy / Dysphagia, Status epilepticus, Rapidly progressive ascending paralysis.
Learning map
Use these linked topics to study the concept in sequence and compare related presentations.