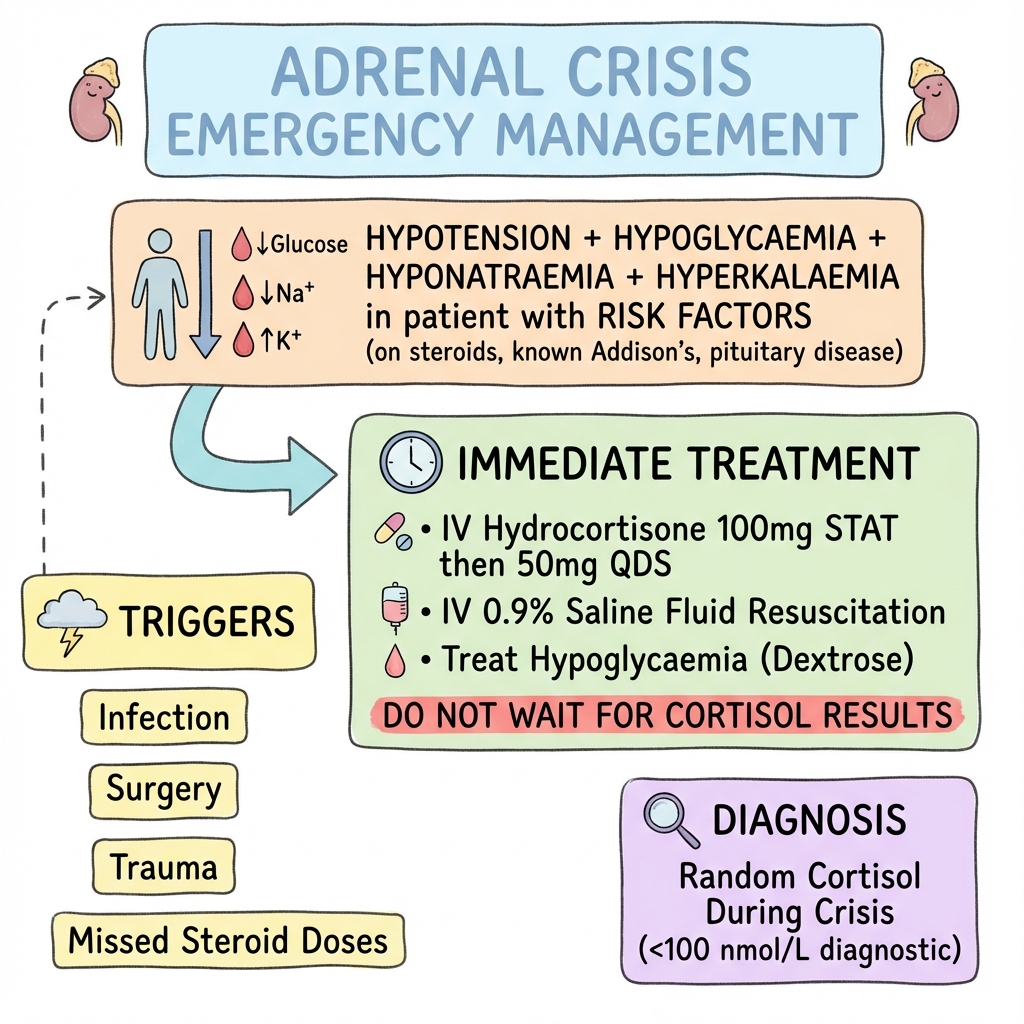
Acute Adrenal Crisis
An Acute Adrenal Crisis (Addisonian Crisis) is a life-threatening endocrine emergency resulting from an absolute or rela... MRCP exam preparation.
What matters first
An Acute Adrenal Crisis (Addisonian Crisis) is a life-threatening endocrine emergency resulting from an absolute or rela... MRCP exam preparation.
Hypotension unresponsive to 2L fluid bolus
6 Jan 2026
Generated educational material; verify before clinical use.
Visible references section
See the concept before reading it
Study the key anatomy, imaging, and decision pathways as full teaching plates.
Clinical board
A visual summary of the highest-yield teaching signals on this page.
Urgent signals
Safety-critical features pulled from the topic metadata.
- Hypotension unresponsive to 2L fluid bolus
- Severe hypoglycaemia (Insulin-antagonist failure)
- Hyperkalaemia with ECG changes (Primary AI)
- Fever with no obvious source in a steroid-user
Exam focus
Current exam surfaces linked to this topic.
- MRCP
Linked comparisons
Differentials and adjacent topics worth opening next.
- Septic Shock
- Acute Abdomen
Content status and exam context
This page is AI-generated educational content. It may contain errors or omissions and is not a substitute for current guidelines, local protocols, senior clinical judgement, or professional medical advice.
MedVellum does not claim an individual clinician reviewer, board certification, or professional credential for this page unless a future version names a real, verifiable reviewer.
Clinical explanation and evidence
Acute Adrenal Crisis (Adult)
1. Overview
Definition
An Acute Adrenal Crisis (Addisonian Crisis) is a life-threatening endocrine emergency resulting from an absolute or relative deficiency of cortisol and, in primary adrenal insufficiency, aldosterone. It is most commonly triggered by physiological stress (infection, trauma, surgery, or acute illness) in a patient with underlying adrenal insufficiency. [1]
Clinical Significance
The clinical significance of adrenal crisis lies in its role as a "preventable death." Despite being rare, it carries substantial mortality if unrecognized:
- Annual Incidence: Patients with known adrenal insufficiency have a 5-10% annual risk of experiencing a crisis. [2]
- Mortality: In-hospital mortality ranges from 0.5-6%, but community-based mortality may be higher due to delayed recognition. [3]
- Leading Cause of Death: Adrenal crisis remains the primary cause of death in patients with Addison's disease, particularly in those without adequate patient education. [4]
The 2024 Management Paradigm
Modern management emphasizes Immediate Empiric Treatment—the "Inject First, Ask Later" principle:
- Diagnosis is Clinical: Laboratory confirmation should never delay treatment
- IV Hydrocortisone: 100mg IV bolus is the cornerstone intervention
- Do Not Wait for Synacthen Test: Short Synacthen testing is contraindicated in acute crisis
- Prevention Focus: Implementation of the National Steroid Emergency Card and patient self-injection training to reduce community mortality [5]
Historical Context
The syndrome was first described by Thomas Addison in 1855, who noted the combination of "general langour and debility, remarkable feebleness of the heart's action, irritability of the stomach, and a peculiar change of colour in the skin." The understanding of cortisol's role and the development of synthetic corticosteroids in the 1950s transformed this universally fatal condition into a manageable emergency. [6]
2. Epidemiology
Incidence and Prevalence
| Parameter | Primary AI (Addison's) | Secondary AI | Tertiary AI |
|---|---|---|---|
| Prevalence | 100-140 per million | 150-280 per million | Variable (steroid-induced) |
| Annual Crisis Rate | 6-10 per 100 patient-years | 3-6 per 100 patient-years | 2-4 per 100 patient-years |
| Gender Ratio | F:M 2.6:1 | F:M 1:1 | Variable |
| Peak Age at Crisis | 30-50 years | 50-70 years | Any age on steroids |
The At-Risk Population
Primary Adrenal Insufficiency (Addison's Disease)
- Autoimmune destruction: 80-90% in Western populations
- Associated autoimmune conditions: Autoimmune polyendocrine syndrome (APS) Type 1 and 2
- Highest crisis risk: Due to concurrent mineralocorticoid (aldosterone) deficiency
- Classic hyperpigmentation: Due to elevated ACTH/POMC
Secondary/Tertiary Adrenal Insufficiency
- Pituitary failure: Tumours, apoplexy, Sheehan's syndrome, surgery, radiotherapy
- Chronic exogenous steroid suppression: The commonest cause overall in clinical practice
- Threshold: > 5mg prednisolone daily for > 3 weeks can suppress the HPA axis
- No hyperpigmentation: ACTH is low or normal
- No mineralocorticoid deficiency: Aldosterone production is ACTH-independent (renin-angiotensin driven)
The "Undiagnosed" Factor
- Critical statistic: 10-15% of adrenal crises occur in patients previously unaware they have adrenal insufficiency [7]
- Presentation: First crisis during intercurrent illness, surgery, or trauma
- Clinical challenge: High index of suspicion required in patients with unexplained refractory shock
Geographic and Seasonal Variation
| Factor | Impact | Mechanism |
|---|---|---|
| Summer Season | 1.5x ↑ Risk | Heat-related fluid loss, sodium depletion, gastroenteritis |
| Winter Season | Infection trigger | Respiratory infections precipitating crisis |
| Developing Countries | Higher mortality | Tuberculosis as common cause of AI, delayed access to care |
Mortality Determinants
High-Risk Precipitants
-
Gastroenteritis: Highest trigger (25-30% of crises)
- Rapid dehydration
- Inability to absorb oral medications
- Sodium loss exacerbating hyponatraemia [8]
-
Elderly Patients: 2.5x higher mortality
- Often misdiagnosed as "simple sepsis"
- Multiple comorbidities
- Atypical presentations
-
Bilateral Adrenal Haemorrhage: 40-50% mortality
- Waterhouse-Friderichsen syndrome (meningococcal sepsis)
- Anticoagulation-related
- Catastrophic antiphospholipid syndrome
Protective Factors
- Patient education programmes: Reduce crisis rate by 40-50% [9]
- Steroid emergency cards: Improve recognition by healthcare providers
- Parenteral hydrocortisone availability: IM self-injection training
3. Aetiology & Pathophysiology
Classification of Adrenal Insufficiency
Primary Adrenal Insufficiency (Addison's Disease)
Destruction or dysfunction of the adrenal cortex itself:
Autoimmune (80-90% in Western populations)
- Isolated autoimmune adrenalitis (70%)
- Autoimmune polyendocrine syndrome Type 1 (10%): AIRE gene mutation
- Hypoparathyroidism
- Chronic mucocutaneous candidiasis
- Addison's disease
- Autoimmune polyendocrine syndrome Type 2 (20%): HLA-DR3/DR4 associated
- Addison's disease
- Type 1 diabetes
- Autoimmune thyroid disease (Schmidt's syndrome)
Infectious
- Tuberculosis (commonest cause worldwide, 10-20% in UK)
- HIV/CMV infection
- Fungal infections (histoplasmosis, cryptococcosis)
Infiltrative
- Metastatic disease (lung, breast, melanoma)
- Lymphoma
- Amyloidosis
- Haemochromatosis
Vascular
- Bilateral adrenal haemorrhage
- Waterhouse-Friderichsen syndrome (meningococcal sepsis)
- Antiphospholipid syndrome
Genetic
- Adrenoleukodystrophy (X-linked, males)
- Congenital adrenal hyperplasia
- Familial glucocorticoid deficiency
Drugs
- Etomidate (single dose can suppress synthesis)
- Ketoconazole
- Mitotane
- Checkpoint inhibitors (immunotherapy)
Secondary Adrenal Insufficiency
Deficiency of ACTH production from the pituitary:
- Pituitary tumours (adenomas, craniopharyngioma)
- Pituitary surgery or radiotherapy
- Pituitary apoplexy (haemorrhage/infarction)
- Sheehan's syndrome (postpartum pituitary necrosis)
- Infiltrative disease (sarcoidosis, haemochromatosis, histiocytosis)
- Hypophysitis (autoimmune, IgG4-related)
- Traumatic brain injury
Tertiary Adrenal Insufficiency
Deficiency of CRH production from the hypothalamus:
- Exogenous glucocorticoid suppression (commonest cause)
- "Threshold: > 5mg prednisolone daily for > 3 weeks"
- "Dose-dependent: Higher doses, longer duration → greater suppression"
- "Route-independent: Oral, inhaled (high-dose), topical (potent), intra-articular"
- Hypothalamic tumours
- Traumatic brain injury
- Post-neurosurgery
Precipitating Factors for Crisis
Any physiological stress can precipitate crisis in a patient with inadequate cortisol reserve:
| Category | Specific Triggers | Mechanism |
|---|---|---|
| Infection | Gastroenteritis, pneumonia, UTI, sepsis | ↑ Cortisol demand, GI losses, poor PO intake |
| Surgery/Trauma | Major surgery, fractures, burns | Maximal stress response required |
| Medical Illness | MI, CVA, DKA, pancreatitis | Acute severe stress |
| Medication | Abrupt steroid withdrawal, etomidate, rifampicin | ↓ Cortisol production or ↑ metabolism |
| Bilateral Adrenal Damage | Haemorrhage, infarction, meningococcal sepsis | Acute loss of residual function |
| Pregnancy/Postpartum | Labour, postpartum Sheehan's | ↑ Cortisol demand |
| Heat Exposure | Heatwave, dehydration | Sodium and fluid depletion |
⚙️ THE 7-STEP MOLECULAR MECHANISM OF CRISIS
Step 1: Cortisol Exhaustion
Under physiological stress (infection, trauma, surgery), a healthy individual increases cortisol production 10-fold (from basal 20mg/day to 200-300mg/day). In adrenal insufficiency, the adrenal cortex (or the pituitary-hypothalamic signal) is incapable of mounting this surge. The result is relative cortisol deficiency even if basal levels were previously adequate. [10]
Step 2: Loss of Permissive Vasoconstriction
Cortisol is "permissive" for catecholamine action. At the molecular level:
- Mechanism: Cortisol upregulates α1-adrenergic receptors on vascular smooth muscle
- In crisis: Absence of cortisol leads to receptor downregulation
- Result: Even with high endogenous adrenaline/noradrenaline levels, vessels remain dilated
- Clinical manifestation: Vasoplegic shock—refractory hypotension despite fluid resuscitation and catecholamine support [11]
Step 3: Vasoplegic Shock Pathway
The combination of:
- Reduced vascular tone (α1-receptor downregulation)
- Reduced cardiac output (impaired catecholamine response)
- Capillary leak (cytokine-mediated, see Step 7)
- Hypovolaemia (mineralocorticoid deficiency, see Step 5)
Creates a distributive + hypovolaemic shock picture that is resistant to standard resuscitation.
Step 4: POMC Dysregulation (Primary AI Only)
In primary adrenal insufficiency:
- Low cortisol → Loss of negative feedback at pituitary
- Massive compensatory rise in ACTH (often > 500 pg/mL, normal less than 46)
- ACTH is derived from the precursor Pro-opiomelanocortin (POMC)
- POMC cleavage produces:
- ACTH (corticotrophin)
- α-MSH (melanocyte-stimulating hormone)
- Clinical result: Hyperpigmentation at:
- Palmar creases
- Buccal mucosa
- Scars (especially recent)
- Pressure points (elbows, knees)
- Nipples and genitalia
Note: Hyperpigmentation is absent in secondary/tertiary AI because ACTH is low.
Step 5: Mineralocorticoid Failure (Primary AI Only)
In primary AI, the zona glomerulosa is destroyed, leading to aldosterone deficiency:
- Distal tubule dysfunction: Failure of the Na+/K+-ATPase pump
- Sodium wasting: Renal sodium loss → hyponatraemia (often 120-130 mmol/L)
- Potassium retention: Hyperkalaemia (5.5-7.0 mmol/L)
- Volume depletion: Sodium loss → osmotic water loss → profound dehydration
- Postural hypotension: Volume depletion + impaired vascular tone
Note: Secondary AI does not cause mineralocorticoid deficiency because aldosterone production is driven by the renin-angiotensin system (ACTH-independent). Therefore:
- No hyperkalaemia in secondary AI
- Hyponatraemia may still occur (due to ↑ ADH from cortisol deficiency)
Step 6: Bioenergetic Failure—Hypoglycaemia
Cortisol is a key counter-regulatory hormone for glucose homeostasis:
- Promotes gluconeogenesis: In the liver, cortisol induces enzymes (PEPCK, G6Pase)
- Inhibits peripheral glucose uptake: Acts as an insulin antagonist
- Promotes glycogen storage: Maintains hepatic glycogen reserves
In crisis:
- Unopposed insulin action → rapid glucose consumption
- Impaired gluconeogenesis → inability to produce new glucose
- Depleted glycogen stores → no glucose reserve
- Result: Profound hypoglycaemia (often less than 3.0 mmol/L)
Clinical note: Hypoglycaemia is more common in:
- Children (smaller glycogen reserves)
- Prolonged fasting states
- Concurrent infection (↑ glucose utilization)
- Secondary AI (may have concurrent GH deficiency, worsening hypoglycaemia) [12]
Step 7: Cytokine Disinhibition—The "Sepsis Mimic"
Cortisol is a potent anti-inflammatory hormone that suppresses:
- Pro-inflammatory cytokines: TNF-α, IL-1β, IL-6
- Endothelial activation: Prevents capillary leak
- Fever generation: Inhibits hypothalamic prostaglandin production
In crisis:
- Unchecked cytokine release → fever, vomiting, abdominal pain
- Capillary leak → worsening hypotension, pulmonary oedema
- Clinical picture indistinguishable from septic shock
- May have leukocytosis without infection
Key point: Fever occurs in 40% of adrenal crises even in the absence of infection. [13]
The "Addisonian Biochemical Triad"
In primary AI, the classic biochemical pattern is:
- Hyponatraemia (120-130 mmol/L)
- Hyperkalaemia (5.5-7.0 mmol/L)
- Hypoglycaemia (less than 3.0 mmol/L)
Plus:
- High urea (pre-renal AKI from volume depletion)
- Low bicarbonate (metabolic acidosis from tissue hypoperfusion)
- Mild hypercalcaemia (10-20% of cases, mechanism unclear)
4. Clinical Presentation
Symptoms
The presentation of adrenal crisis is often non-specific, leading to diagnostic delay. Key features:
Constitutional Symptoms
- Profound fatigue: "Weakness so severe the patient cannot stand" [14]
- Anorexia: Severe loss of appetite
- Weight loss: May be present chronically before crisis
- Salt craving: Reported in 15-20% (compensation for sodium loss)
Gastrointestinal Symptoms (70-80% of cases)
- Severe abdominal pain: Diffuse, cramping, may mimic acute abdomen
- Nausea and vomiting: Persistent, leading to inability to take oral medications
- Diarrhoea: Particularly with infective precipitants (gastroenteritis)
Diagnostic pitfall: GI symptoms often lead to misdiagnosis as gastroenteritis, appendicitis, or bowel obstruction, delaying correct treatment.
Neuropsychiatric Symptoms
- Confusion: Due to hypoglycaemia, hyponatraemia, or hypotension
- Drowsiness progressing to coma: In severe cases
- Irritability: May be prominent in early stages
- Psychosis: Rare, but reported in severe hyponatraemia
Physical Signs
Vital Sign Abnormalities
| Sign | Finding | Mechanism |
|---|---|---|
| Hypotension | Systolic less than 90 mmHg | Vasoplegic shock + volume depletion |
| Postural Drop | > 20 mmHg fall on standing | Volume depletion |
| Tachycardia | 100-130 bpm | Compensatory response to hypotension |
| Fever | 38-40°C | Cytokine-mediated or concurrent infection |
| Hypothermia | less than 36°C | In severe cases, associated with poor prognosis |
Dermatological Signs (Primary AI Only)
Hyperpigmentation (80-90% of primary AI):
- Distribution:
- Palmar creases
- Buccal mucosa (inside cheeks)
- Recent scars
- Pressure points (elbows, knees, knuckles)
- Nipples, areolae
- Genitalia
- Appearance: Diffuse bronzing of sun-exposed areas
- Mechanism: MSH upregulation (see pathophysiology)
Vitiligo (10-20% of autoimmune AI):
- Patchy depigmentation
- Suggests autoimmune polyendocrine syndrome
Absent in secondary/tertiary AI (because ACTH is low)
Cardiovascular Signs
- Refractory hypotension: Does not respond to 1-2L fluid bolus
- Small heart silhouette: On chest X-ray (chronic finding)
- Weak pulse: Reduced stroke volume
- Poor capillary refill: > 3 seconds
Neurological Signs
- Reduced GCS: 8-14 (from hypoglycaemia or hyponatraemia)
- Seizures: Rare, from severe hyponatraemia or hypoglycaemia
- Focal neurology: If underlying pituitary lesion causing secondary AI
Clinical Scenarios by Underlying Cause
Scenario 1: Known Addison's Disease + Gastroenteritis
Typical presentation
- 35-year-old female, known Addison's on hydrocortisone 15mg/5mg
- 2-day history of vomiting and diarrhoea
- Stopped oral medications due to vomiting
- Presents with BP 75/40, HR 125, confused
- Learning point: Should have doubled/tripled oral dose at onset of illness or switched to IM hydrocortisone
Scenario 2: Undiagnosed Addison's + Surgery
Diagnostic challenge
- 50-year-old male undergoing routine hernia repair
- Develops refractory hypotension post-operatively
- Not responding to fluids or vasopressors
- Noted to have "suntan" despite winter season
- Learning point: High index of suspicion in unexplained perioperative shock
Scenario 3: Chronic Steroid User + Infection
HPA suppression
- 60-year-old with COPD on prednisolone 10mg daily for 2 years
- Develops pneumonia
- Deteriorates rapidly despite antibiotics
- Learning point: Relative AI in steroid users during stress—requires stress-dose steroids
Scenario 4: Pituitary Apoplexy
Acute secondary AI
- 45-year-old with sudden severe headache
- Visual field defect (bitemporal hemianopia)
- Rapid onset of hypotension and confusion
- Learning point: Consider pituitary catastrophe in acute AI with neuro-ophthalmological signs
5. Investigations
The "Golden Sample" Principle
CRITICAL RULE: Draw blood for cortisol and ACTH before giving hydrocortisone if possible, but DO NOT WAIT for results before treating.
Why?
- A random cortisol taken during crisis has diagnostic value retrospectively
- Once exogenous hydrocortisone is given, cortisol assays are unreliable for days
- However, treatment must never be delayed for sample collection
What to Send
- Random serum cortisol
- ACTH
- Routine bloods (see below)
Immediate Bedside Investigations
| Test | Finding | Interpretation |
|---|---|---|
| Capillary glucose | less than 3.0 mmol/L | Severe hypoglycaemia—give IV dextrose immediately |
| ECG | Tall tented T waves, widened QRS | Hyperkalaemia—may need calcium gluconate |
| BP lying/standing | > 20 mmHg drop | Volume depletion |
Laboratory Investigations
Serum Cortisol
- Normal stress response: > 500-550 nmol/L
- Suggestive of AI in crisis: less than 100 nmol/L
- Equivocal: 100-500 nmol/L (may represent partial insufficiency or assay variability)
Limitations:
- Cortisol-binding globulin (CBG) levels affect total cortisol
- Critical illness can lower CBG, confounding interpretation
- Single random cortisol cannot definitively diagnose or exclude AI
ACTH
- Primary AI: Markedly elevated (> 100-500 pg/mL; normal less than 46 pg/mL)
- Secondary/Tertiary AI: Low or inappropriately normal (less than 10-30 pg/mL)
Sample handling:
- ACTH is unstable at room temperature
- Must be collected in EDTA tube, on ice, and processed within 15 minutes
- Often impractical in acute settings
Urea and Electrolytes (U&Es)
Classic "Addisonian" pattern:
| Electrolyte | Finding | Mechanism |
|---|---|---|
| Sodium | 120-130 mmol/L | Aldosterone deficiency (1° AI) + ↑ ADH (all AI) |
| Potassium | 5.5-7.0 mmol/L | Aldosterone deficiency (1° AI only) |
| Urea | Elevated (> 10-20 mmol/L) | Pre-renal AKI from volume depletion |
| Creatinine | Mildly elevated | Pre-renal AKI |
| Glucose | less than 3.0 mmol/L | Impaired gluconeogenesis + lack of insulin antagonism |
Secondary AI pattern (different):
- Hyponatraemia (from SIADH-like state)
- Normal potassium (aldosterone intact)
- Hypoglycaemia (especially if GH deficiency coexists)
Other Blood Tests
| Test | Typical Finding | Notes |
|---|---|---|
| Calcium | Mild ↑ (10-20% cases) | Mechanism unclear; resolves with treatment |
| Eosinophils | Elevated (> 0.5 × 10⁹/L) | Loss of cortisol suppression |
| Lymphocytes | Elevated | Loss of cortisol immunosuppression |
| TSH | May be ↑ or ↓ | If pituitary cause (2° hypothyroidism) or APS Type 2 (1° hypothyroidism) |
| Haemoglobin | Normocytic anaemia | Chronic AI finding |
| CRP | Elevated if infection | Or ↑ from cytokine release alone |
Arterial Blood Gas (ABG)
- Metabolic acidosis: pH 7.2-7.35, low HCO₃
- Hyperlactataemia: Lactic acidosis from tissue hypoperfusion
- Hypoglycaemia: Direct glucose measurement
Imaging
Chest X-Ray
- Small heart: Chronic finding in AI (reduced cardiac mass)
- Rule out pneumonia: As precipitant
- No cardiomegaly: Helps differentiate from cardiogenic shock
CT Adrenal Glands (Non-Acute)
Not required in acute crisis, but useful later for aetiology:
- Autoimmune AI: Small, atrophic glands
- TB/infection: Enlarged, calcified glands
- Haemorrhage: Hyperdense (acute) or cystic (chronic) lesions
- Metastases: Bilateral masses
MRI Pituitary (If Secondary AI Suspected)
- Pituitary macroadenoma
- Empty sella (post-apoplexy or Sheehan's)
- Infiltrative disease
Definitive Diagnosis: The Short Synacthen Test
CRITICAL: Do NOT perform the Synacthen test in the acute crisis. It is both useless and dangerous.
Why Not in Crisis?
- The patient is profoundly unwell—giving a test that delays treatment is unethical
- Even if the result is "normal," clinical suspicion mandates treatment
- The test can precipitate cardiovascular collapse in unstable patients
When to Perform?
- Day 3-5 after crisis resolution, once the patient is stable
- Patient should be off hydrocortisone for 24 hours (switch to dexamethasone if needed for cross-coverage, as dexamethasone does not interfere with cortisol assay)
Synacthen Test Protocol
- Baseline: Take cortisol at 0 minutes
- Administration: Give 250 mcg Synacthen (ACTH₁₋₂₄) IM or IV
- Post-Synacthen: Take cortisol at 30 and 60 minutes
Interpretation:
- Normal: 60-minute cortisol > 550 nmol/L (rules out primary AI)
- Primary AI: Blunted response (less than 550 nmol/L) + high baseline ACTH
- Secondary AI: Blunted response (less than 550 nmol/L) + low baseline ACTH
Note: The 250 mcg test may be supra-physiological and can give false-normal results in mild secondary AI. A low-dose (1 mcg) Synacthen test is more sensitive but rarely available.
Identifying the Precipitant
Once the diagnosis of adrenal crisis is made, search for the trigger:
| Investigation | Target |
|---|---|
| Blood cultures | Sepsis |
| Urine culture | UTI |
| Chest X-ray | Pneumonia |
| ECG + Troponin | Myocardial infarction (can precipitate crisis) |
| CT Abdomen | If bilateral adrenal haemorrhage suspected (flank pain, anticoagulation) |
| Clotting screen | If anticoagulated or bleeding suspected |
6. Management: The Emergency Protocol
The "ABCDE" Approach to Adrenal Crisis
A: Airway
- Assess and secure if GCS less than 8
- Consider intubation if profoundly hypotensive or reduced consciousness
B: Breathing
- High-flow oxygen (15L via non-rebreather mask) if shocked
- Monitor respiratory rate (may be increased in acidosis)
C: Circulation—The Key Interventions
1. IV HYDROCORTISONE (Immediate—Do Not Delay)
Dose:
- 100mg IV bolus immediately (push over 1-2 minutes)
- Followed by: 200mg per 24 hours
- "Either: 50mg IV QDS (every 6 hours)"
- "Or: Continuous infusion 8-10mg/hour"
Rationale:
- Hydrocortisone has both glucocorticoid and mineralocorticoid activity at high doses
- 100mg hydrocortisone provides supraphysiological cortisol and enough mineralocorticoid to eliminate need for fludrocortisone acutely
- Rapid restoration of vascular tone (α1-receptor upregulation within 1-2 hours) [15]
Alternative if hydrocortisone unavailable:
- Dexamethasone 4mg IV (glucocorticoid only, no mineralocorticoid)
- Prednisolone is not suitable (poor IV absorption, unpredictable bioavailability)
2. AGGRESSIVE FLUID RESUSCITATION
Fluid of Choice: 0.9% Sodium Chloride (Normal Saline)
Regimen:
- 1st Litre: Over 30-60 minutes
- 2nd Litre: Over next 1-2 hours
- Reassess: Clinical response (BP, urine output, lactate clearance)
- Total: May require 3-6 litres in first 24 hours
Rationale:
- Profound volume depletion from sodium and water loss
- Vasoplegic shock component requires adequate preload
- Hyponatraemia usually improves with saline + hydrocortisone (no need for hypertonic saline unless severe symptomatic hyponatraemia with seizures)
Monitor:
- CVP if available (target 8-12 mmHg)
- Urine output (target > 0.5 mL/kg/hr)
- Lactate clearance
- Lung auscultation (risk of pulmonary oedema if over-resuscitated)
3. CORRECT HYPOGLYCAEMIA
If glucose less than 4.0 mmol/L:
- 10% Dextrose: 200 mL bolus IV over 10 minutes
- Followed by: Add 10% dextrose to maintenance fluids
- Recheck: Capillary glucose every 15 minutes until > 5.0 mmol/L
If glucose less than 2.0 mmol/L or patient unconscious:
- Consider Glucagon 1mg IM (if IV access delayed)
- Or 50 mL of 50% Dextrose IV (highly concentrated, risk of extravasation injury—use large vein)
D: Disability (Neurological Assessment)
- GCS: Monitor for improvement after glucose and steroid administration
- Pupils: Check for focal neurology (pituitary apoplexy)
E: Exposure
- Look for hyperpigmentation: Palmar creases, buccal mucosa (suggests primary AI)
- Check for signs of trauma/infection: As precipitant
Adjunctive Management
Electrolyte Correction
Hyperkalaemia (If K⁺ > 6.5 mmol/L or ECG changes)
Immediate:
- Calcium Gluconate 10%: 10 mL IV over 2-3 minutes (cardiac protection, does not lower K⁺)
- Insulin-Dextrose: 10 units Actrapid in 50 mL 50% Dextrose (shifts K⁺ intracellularly)
- Salbutamol 10-20mg: Nebulised (β2-agonist shifts K⁺ intracellularly)
Note: Hyperkalaemia will resolve with hydrocortisone (mineralocorticoid effect) + saline resuscitation. Aggressive potassium-lowering is rarely needed unless life-threatening arrhythmia present.
Hyponatraemia
- Mild-Moderate (120-130 mmol/L): Usually improves with saline + hydrocortisone alone
- Severe (less than 120 mmol/L with seizures/coma):
- "Consider 3% Hypertonic Saline: 150 mL bolus over 20 minutes"
- "Target: Raise Na⁺ by 4-6 mmol/L in first hour (enough to stop seizures)"
- "Caution: Do not raise > 10 mmol/L in 24 hours (risk of osmotic demyelination syndrome)"
Mechanism of improvement: Cortisol suppresses ADH (resolves SIADH component) + aldosterone replacement (promotes Na⁺ retention)
Vasopressor Support (If Refractory Hypotension)
Indications:
- Persistent hypotension despite 2L fluid + 100mg hydrocortisone
- Lactate > 4 mmol/L
- Oliguria
Agent of Choice: Noradrenaline (peripheral α1-agonist)
- Start at 0.05 mcg/kg/min, titrate to MAP > 65 mmHg
- Requires central venous access
- Note: May require higher doses initially due to receptor downregulation; improves within hours as hydrocortisone upregulates receptors [16]
Alternative: Vasopressin (for catecholamine-resistant shock)
Antibiotic Therapy
If infection suspected as precipitant:
- Sepsis protocol: Broad-spectrum antibiotics within 1 hour (e.g., piperacillin-tazobactam or meropenem)
- Blood cultures: Before antibiotics if possible
- Source control: Drain abscesses, remove infected lines
Treatment of the Precipitant
| Precipitant | Specific Management |
|---|---|
| Gastroenteritis | Antiemetics, rehydration, consider antibiotics if bacterial |
| Pneumonia | Antibiotics, oxygen, respiratory support |
| MI | Dual antiplatelet, heparin, cardiology referral |
| Bilateral adrenal haemorrhage | Stop anticoagulation, consider surgical intervention if catastrophic |
| Meningococcal sepsis | Ceftriaxone 2g IV, meningitis protocol, contact tracing |
Monitoring
Immediate (First 6 Hours)
| Parameter | Frequency | Target |
|---|---|---|
| Blood Pressure | Continuous or every 15 min | Systolic > 90 mmHg, MAP > 65 mmHg |
| Heart Rate | Continuous | less than 100 bpm |
| Capillary Glucose | Every 15-30 min | > 5.0 mmol/L |
| Urine Output | Hourly | > 0.5 mL/kg/hr |
| GCS | Hourly | Expect improvement within 1-2 hours |
| Lactate | 1-2 hourly | Clearance > 10% per hour |
Subsequent (6-24 Hours)
| Parameter | Frequency | Target |
|---|---|---|
| U&Es | 6-hourly | Na⁺ rising, K⁺ falling, urea stable |
| Glucose | 4-hourly | Maintain 5-10 mmol/L |
| Fluid Balance | Hourly input/output | Neutral to slightly positive balance |
Transition to Maintenance Therapy
Once the patient is stable (usually 24-48 hours):
Oral Hydrocortisone Regimen
Standard replacement doses:
- Hydrocortisone: 15-25mg daily in divided doses
- "Typical: 10mg on waking, 5mg at lunchtime, 5mg at 6pm (mimics diurnal rhythm)"
- Avoid evening doses after 6pm (causes insomnia)
Transition protocol:
- Continue IV hydrocortisone 50mg QDS until patient tolerating oral intake
- Switch to oral when eating and drinking normally
- No need to "taper" from stress dose to maintenance—direct switch is safe
Fludrocortisone (If Primary AI)
- Dose: 50-200 mcg once daily (morning)
- Start: Once oral intake established
- Monitoring: Plasma renin activity (target upper-normal range), BP, U&Es
Not required in secondary AI (aldosterone production intact)
Long-Term Patient Education
Sick Day Rules (CRITICAL for preventing future crises):
- Double the dose of hydrocortisone during minor illness (fever, vomiting, diarrhoea)
- Triple the dose during major illness or significant injury
- Use IM hydrocortisone 100mg if vomiting or unable to take oral (patient self-injection)
- Seek medical help if symptoms not improving within 24 hours
Steroid Emergency Card:
- All patients must carry a National Steroid Emergency Card
- States diagnosis and emergency steroid requirements
- Healthcare providers must administer IV/IM hydrocortisone in any acute illness
Medical Alert Bracelet:
- Recommended for all patients with AI
- Identifies condition to first responders
7. Prognosis
Short-Term Outcomes
| Metric | Value | Notes |
|---|---|---|
| In-hospital Mortality | 0.5-6% | Lower with rapid recognition and treatment |
| Community Mortality | Unknown, likely higher | Delayed access to care |
| Recovery Time | 24-72 hours | Most patients improve within 24 hours of IV steroids |
| Recurrence Risk | 5-10% per year | Without adequate education, higher with poor adherence |
Long-Term Outcomes
With Adequate Replacement Therapy
- Normal life expectancy: If adherent to treatment and educated about sick day rules
- Quality of life: Generally good, though some report persistent fatigue even on optimal replacement [17]
- Fertility: Normal in primary AI; may be impaired in secondary AI if other pituitary hormones deficient
Predictors of Future Crisis
| Factor | Risk |
|---|---|
| Lack of patient education | Highest risk factor—education reduces crisis rate by 40-50% |
| Non-adherence to medication | 3-4x ↑ risk |
| Absence of emergency hydrocortisone kit | 2-3x ↑ risk |
| Concurrent autoimmune disease | ↑ Risk (unpredictable intercurrent illnesses) |
| Alcohol excess | ↑ Risk (non-adherence, increased infections) |
8. Evidence: Landmark Trials and Guidelines
Key Studies
| Study | Year | Population | Intervention | Key Finding | Clinical Impact |
|---|---|---|---|---|---|
| Hahner et al. [9] | 2015 | 423 patients with AI | Patient education programme | 50% ↓ crisis rate (8.3 → 3.6 per 100 patient-years) | Established education as standard of care |
| Rushworth et al. [3] | 2019 | Review of AI crises | Epidemiology | Mortality 0.5% if treated early | Emphasised need for rapid recognition |
| Puar et al. [18] | 2016 | 77 AI crisis episodes | Precipitants and outcomes | GI illness commonest (37%), mortality 6.5% | Importance of sick day rules for GI illness |
| Allolio et al. | 2015 | AI management | Stress-dose steroids | 100mg hydrocortisone provides sufficient mineralocorticoid | No need for separate fludrocortisone in crisis |
Guidelines
Endocrine Society Clinical Practice Guideline (2016) [1]
- Recommendation 1.1: Immediate hydrocortisone 100mg IV for suspected adrenal crisis (Strong recommendation, moderate evidence)
- Recommendation 1.2: Do not delay treatment for diagnostic testing (Strong recommendation, low evidence)
- Recommendation 2.1: Aggressive fluid resuscitation with 0.9% saline (Strong recommendation, low evidence)
- Recommendation 3.1: All patients with AI should receive education on sick day rules (Strong recommendation, high evidence)
Society for Endocrinology (UK) Guidance (2024) [5]
- Emergency steroid card for all patients
- Patient self-injection training
- Annual review with specialist
9. Differential Diagnosis
Adrenal crisis can mimic many acute conditions. The key is a high index of suspicion in patients with:
- Known AI
- Autoimmune disease (risk of subclinical AI)
- Chronic steroid use
- Unexplained hyperpigmentation
Major Differentials
| Condition | Similarities | Distinguishing Features |
|---|---|---|
| Septic Shock | Hypotension, fever, tachycardia, ↑ lactate | CRP usually very high, source of infection evident, normal electrolytes (usually) |
| Hypovolaemic Shock | Hypotension, tachycardia, ↑ urea | Clear history of fluid loss (bleeding, burns), no hypoglycaemia or hyperkalaemia |
| Acute Abdomen | Abdominal pain, vomiting, ↑ WCC | Peritonism on exam, imaging shows surgical pathology |
| Diabetic Ketoacidosis | Vomiting, dehydration, acidosis, ↑ glucose (usually) | Glucose > 11 mmol/L, ketones +++, known diabetes |
| Hypoglycaemia | Low glucose, confusion, sweating | Responds immediately to glucose, no hypotension |
| Acute Gastroenteritis | Vomiting, diarrhoea, dehydration | Mild hypotension only, resolves with fluids alone, no hypoglycaemia |
The "Septic Shock that Doesn't Respond to Fluids"
Key teaching point: If a patient with suspected sepsis:
- Does not respond to 2L fluid bolus
- Has unexplained hypoglycaemia
- Has hyponatraemia + hyperkalaemia
- Has eosinophilia
→ Consider adrenal crisis and give empirical hydrocortisone 100mg IV
10. Special Scenarios
10.1 Perioperative Adrenal Crisis Prevention
Risk Stratification
| Steroid Exposure | HPA Suppression Risk | Perioperative Plan |
|---|---|---|
| > 5mg prednisolone daily for > 3 weeks | High | Stress-dose steroids |
| Any dose for > 3 months | High | Stress-dose steroids |
| less than 5mg daily for less than 3 weeks | Low | Continue usual dose |
| Stopped less than 1 year ago | Possible | Consider stress-dose if major surgery |
Stress-Dose Steroid Protocol
For Major Surgery (e.g., Laparotomy, Joint Replacement, Cardiac Surgery):
- At induction: 100mg IV hydrocortisone bolus
- Intraoperatively: 50mg IV every 6 hours or continuous infusion 10mg/hr
- Post-op Day 1-2: Continue 50mg IV QDS
- Day 3: Reduce to 25mg IV QDS
- Day 4: Switch to oral maintenance dose (e.g., 10/5/5 mg)
For Minor Surgery (e.g., Hernia Repair, Endoscopy):
- At induction: 100mg IV hydrocortisone bolus
- Post-op: Resume oral maintenance dose once eating/drinking
Rationale: Surgical stress increases cortisol demand 5-10 fold; patients with HPA suppression cannot mount this response. [18]
10.2 Pregnancy and Adrenal Crisis
Physiological Changes
- Cortisol-binding globulin (CBG) ↑ 2-3 fold in pregnancy → total cortisol ↑, but free cortisol normal
- No need to increase hydrocortisone dose in uncomplicated pregnancy
Labour and Delivery
Vaginal Delivery:
- 100mg IV hydrocortisone at onset of labour
- 50mg IV every 6 hours during labour
- Resume oral dose 24 hours postpartum
Elective Caesarean Section:
- Treat as major surgery (100mg IV at induction, 50mg QDS for 24-48 hours)
Emergency Caesarean:
- 100mg IV stat
- Continue stress-dose until stable
Postpartum
- Risk of Sheehan's syndrome (pituitary infarction) in severe postpartum haemorrhage → acute secondary AI
- Monitor for symptoms of AI in any woman with severe PPH
10.3 Checkpoint Inhibitor-Induced AI
Immunotherapy (e.g., Nivolumab, Pembrolizumab) can cause autoimmune adrenalitis:
- Incidence: 1-2% of patients on checkpoint inhibitors
- Presentation: Often insidious, may present acutely with crisis
- Diagnosis: Low cortisol, high ACTH
- Management:
- Do not stop immunotherapy in most cases (benefit in cancer treatment outweighs risk)
- "Start hydrocortisone replacement: Standard doses"
- Educate on sick day rules
- Lifelong replacement required (rarely recovers)
10.4 Bilateral Adrenal Haemorrhage
Causes:
- Anticoagulation (warfarin, heparin, DOACs)
- Meningococcal sepsis (Waterhouse-Friderichsen syndrome)
- Antiphospholipid syndrome
- Trauma
Presentation:
- Acute adrenal crisis
- Flank pain (50% of cases)
- Hypotension refractory to fluids
Diagnosis:
- CT abdomen: Bilateral hyperdense adrenal masses (acute haemorrhage) or cystic lesions (chronic)
Management:
- Immediate: IV hydrocortisone 100mg, fluids
- Stop anticoagulation (if that is the cause)
- Surgery: Rarely needed unless catastrophic bleeding
- Long-term: Lifelong hydrocortisone + fludrocortisone replacement
11. Patient Education and Self-Management
The "Steroid Sick Day Rules"
Simple Version (for Patients):
-
Feeling unwell with fever, vomiting, diarrhoea, or injury?
- Double your usual steroid dose immediately
-
Still unwell or getting worse?
- Triple your dose the next day
-
Vomiting and can't keep tablets down?
- Use your emergency injection kit (100mg IM hydrocortisone)
- Call 999 or go to A&E
-
Feeling better?
- Stay on double dose for 24 hours after fever settles
- Then return to usual dose
Emergency Injection Training
All patients with AI should be trained in self/partner IM injection:
- Kit: 100mg hydrocortisone sodium succinate (Solu-Cortef Act-o-Vial)
- When: If vomiting, severe illness, or unable to attend hospital rapidly
- Site: Outer thigh (vastus lateralis) or upper outer buttock
- Technique: 90-degree angle, IM injection
- After injection: Still seek medical help urgently
Evidence: Patient self-injection reduces mortality in community-onset crises. [9]
Steroid Emergency Card
UK National Steroid Emergency Card (introduced 2020):
- Carried by all patients on long-term steroids or with AI
- States:
- Diagnosis (adrenal insufficiency)
- Usual dose
- Emergency instructions: "In an emergency, give 100mg hydrocortisone IV or IM immediately"
- Shown to all healthcare providers
Medical Alert Bracelet
Recommended for all patients—identifies condition to paramedics/emergency staff if patient unconscious.
12. Viva Voce Scenarios
Scenario 1: Perioperative Steroid Management
Examiner: "A 55-year-old woman on 10mg prednisolone daily for rheumatoid arthritis is scheduled for an elective total knee replacement. What is your perioperative steroid plan?"
Model Answer:
-
Assessment: This patient has been on > 5mg prednisolone for a chronic condition, so her HPA axis is likely suppressed. She cannot mount a normal stress response to surgery.
-
Risk: Without supplemental steroids, she is at risk of perioperative adrenal crisis.
-
Management:
- Pre-op: Continue usual dose up to day of surgery
- At induction: 100mg IV hydrocortisone bolus
- Post-op: 50mg IV hydrocortisone every 6 hours for first 24-48 hours
- Day 3: If eating and drinking, switch to oral prednisolone at usual dose
-
Rationale: Major orthopaedic surgery is a significant physiological stress. A normal person would increase cortisol production to 200-300mg/day equivalent. By giving 200mg/day IV hydrocortisone for the first 2 days, we replicate this stress response.
-
Monitoring: BP, glucose, clinical recovery
Examiner Follow-Up: "What if she were having a minor procedure like a colonoscopy?"
Answer: For minor/moderate stress, a single dose of 100mg IV hydrocortisone at induction is sufficient, then resume oral dose once eating.
Scenario 2: Undiagnosed Addison's Crisis
Examiner: "A 40-year-old man presents with a 3-day history of vomiting, abdominal pain, and confusion. He is hypotensive at 85/50 despite 2 litres of saline. Blood results show Na 125, K 6.2, glucose 2.8. What is your differential and immediate management?"
Model Answer:
-
Differential Diagnosis:
- Most likely: Acute adrenal crisis (explains the triad of hyponatraemia, hyperkalaemia, hypoglycaemia + refractory hypotension)
- Septic shock (but electrolyte pattern unusual)
- Acute gastroenteritis with severe dehydration (but hypoglycaemia and hyperkalaemia not typical)
-
Supportive Clues to Look For:
- Hyperpigmentation (palmar creases, buccal mucosa) → suggests primary AI
- History of autoimmune disease (vitiligo, type 1 diabetes, hypothyroidism)
- Chronic fatigue, weight loss, salt craving in preceding weeks/months
-
Immediate Management (do not wait for further tests):
- A-E Assessment
- Hydrocortisone 100mg IV stat (even if diagnosis not yet confirmed—no harm if wrong, fatal if missed)
- Further fluid resuscitation: 1L 0.9% saline over 1 hour
- Dextrose: 200mL 10% IV for hypoglycaemia
- Calcium gluconate: 10mL 10% IV if ECG shows hyperkalaemic changes
-
Investigations (before giving hydrocortisone if possible, but don't delay treatment):
- Random cortisol and ACTH ("golden sample")
- Full septic screen (cultures, CXR)
- ECG (for hyperkalaemia, and to rule out MI as precipitant)
-
Admission: HDU/ICU, close monitoring
Examiner Follow-Up: "The cortisol comes back at 85 nmol/L. What does this tell you?"
Answer: In the context of severe physiological stress, a cortisol of 85 nmol/L is inappropriately low. A normal stress response should produce > 550 nmol/L. This strongly supports the diagnosis of adrenal insufficiency. I would continue IV hydrocortisone and arrange a Short Synacthen test once the patient is stable (Day 3-5) to confirm the diagnosis.
Scenario 3: Hyperpigmentation Without Crisis
Examiner: "A 30-year-old woman is referred with a 6-month history of fatigue and weight loss. On examination, she has hyperpigmentation of her palmar creases and buccal mucosa. What is your approach?"
Model Answer:
-
Most Likely Diagnosis: Primary adrenal insufficiency (Addison's disease)
-
Hyperpigmentation Mechanism:
- Low cortisol → loss of negative feedback at pituitary
- Massive ↑ in ACTH production
- ACTH is derived from POMC, which also produces MSH (melanocyte-stimulating hormone)
- MSH causes melanocyte activation → hyperpigmentation
-
Investigations:
- 9am cortisol (if less than 100 nmol/L, very suggestive; if > 500 nmol/L, unlikely AI)
- 9am ACTH (very high in primary AI, low in secondary AI)
- Short Synacthen Test (definitive test):
- Give 250mcg Synacthen IM/IV
- Measure cortisol at 0, 30, and 60 minutes
- Normal: 60-min cortisol > 550 nmol/L
- Addison's: Blunted response + very high baseline ACTH
- U&Es: May show ↓ Na, ↑ K even before crisis
- Adrenal antibodies: 21-hydroxylase antibodies (positive in 90% autoimmune AI)
- Screen for associated autoimmune disease: TFTs (hypothyroidism), HbA1c (T1DM), coeliac serology, B12/folate (pernicious anaemia)
-
Management (non-acute):
- Hydrocortisone: 15-25mg daily in divided doses (10/5/5 mg typical)
- Fludrocortisone: 50-200mcg once daily (primary AI only)
- Patient education: Sick day rules, emergency injection kit, steroid card
- Annual review: U&Es, renin, BP, adherence check
-
Emergency Plan:
- Written sick day rules
- Emergency hydrocortisone 100mg IM kit for self-administration
- Medical alert bracelet
Examiner: "Why does she not have hyperpigmentation in secondary AI?"
Answer: In secondary AI, the problem is at the pituitary (low ACTH production). Because ACTH is low, there is no compensatory rise in POMC, and therefore no MSH production. Consequently, hyperpigmentation does not occur.
13. Single Best Answer (SBA) Questions
Question 1
A 45-year-old female with a history of vitiligo is brought to the ED with a BP of 80/40 and confusion. Her U&Es show Na 128, K 5.8, and glucose 2.4. She has not responded to a 1L fluid bolus. What is the most appropriate immediate action?
A) Start a Noradrenaline infusion B) Perform a Short Synacthen Test C) Give IV Hydrocortisone 100mg D) Order a CT Abdomen to look for an abscess E) Give 10 units of Insulin with Dextrose
Answer: C
Explanation: This is a classic presentation of Addisonian Crisis. The triad of:
- Hyponatraemia (128)
- Hyperkalaemia (5.8)
- Hypoglycaemia (2.4)
Plus refractory hypotension in a patient with an autoimmune condition (vitiligo) strongly suggests primary adrenal insufficiency. Immediate IV hydrocortisone 100mg is life-saving and must not be delayed.
- (A) Noradrenaline may be needed later, but hydrocortisone must come first to restore vascular receptor function
- (B) Synacthen test is contraindicated in acute crisis—never delay treatment
- (D) Imaging can be done later once stabilised
- (E) Insulin would be fatal—the hypoglycaemia is from cortisol deficiency, not hyperglycaemia
Question 2
Why is hyperpigmentation absent in patients who develop an adrenal crisis due to secondary adrenal insufficiency (pituitary failure)?
A) Cortisol is not low enough B) POMC is not over-produced in pituitary failure C) Aldosterone remains normal D) The skin becomes resistant to MSH E) Secondary AI does not cause crisis
Answer: B
Explanation: Hyperpigmentation in primary AI is driven by:
- Low cortisol → ↑ ACTH (via loss of negative feedback)
- ACTH is cleaved from POMC
- POMC also produces MSH (melanocyte-stimulating hormone)
- MSH → melanocyte activation → hyperpigmentation
In secondary AI (pituitary failure), ACTH production is impaired, so there is no compensatory rise in POMC, and therefore no MSH. Hence, no hyperpigmentation.
- (A) Cortisol can be equally low in both types
- (C) True, but this does not explain absence of hyperpigmentation
- (D) False—skin MSH receptors are normal
- (E) False—secondary AI can cause crisis, especially during stress
Question 3
A 60-year-old man with COPD on prednisolone 10mg daily for 2 years is admitted with pneumonia. Despite antibiotics and 2L fluids, his BP is 85/50. What is the most appropriate next step?
A) Start vasopressors B) Give IV hydrocortisone 100mg C) Stop his prednisolone D) Give IV methylprednisolone 40mg E) Increase his oral prednisolone to 20mg
Answer: B
Explanation: This patient has been on > 5mg prednisolone daily for > 3 weeks, leading to HPA axis suppression. During the stress of pneumonia, he cannot mount an adequate cortisol response, leading to relative adrenal insufficiency.
The appropriate management is stress-dose IV hydrocortisone 100mg immediately.
- (A) Vasopressors may be needed, but hydrocortisone must be given first to restore vascular responsiveness
- (C) Stopping prednisolone would worsen the crisis
- (D) Methylprednisolone is not the preferred agent (hydrocortisone has mineralocorticoid activity at high doses)
- (E) Oral prednisolone is inadequate during critical illness (unpredictable absorption)
14. Clinical Pearls
Pearl 1: "Septic Shock + Eosinophilia = Think Adrenal Crisis"
Normal stress response suppresses eosinophils. If a shocked patient has eosinophilia (> 0.5 × 10⁹/L), consider AI.
Pearl 2: "Hydrocortisone, Not Prednisolone, in Crisis"
Hydrocortisone has mineralocorticoid activity at high doses (100mg provides ~1mg fludrocortisone equivalent). Prednisolone does not. Always use hydrocortisone in crisis.
Pearl 3: "The Golden Sample"
If you suspect adrenal crisis, send cortisol and ACTH before giving hydrocortisone—but never delay treatment. You can still do a Synacthen test in 3-5 days.
Pearl 4: "Bilateral Adrenal Haemorrhage = Flank Pain + Shock"
In a patient on anticoagulation with shock and flank pain, think bilateral adrenal haemorrhage. CT abdomen is diagnostic.
Pearl 5: "Primary AI = Hyperkalaemia, Secondary AI = Normal K⁺"
The presence or absence of hyperkalaemia helps distinguish primary (mineralocorticoid deficiency → high K⁺) from secondary AI (mineralocorticoid intact → normal K⁺).
15. Patient Explanation
"An adrenal crisis is a medical emergency where your body runs out of a vital hormone called cortisol. Think of cortisol as your body's 'emergency battery'—it keeps your blood pressure stable, your blood sugar normal, and helps you cope with stress like infections or injuries.
You have two small glands above your kidneys called the adrenal glands. In your case, these glands aren't working properly [or: the signal from your brain to these glands isn't working]. When you got this infection, your body tried to produce more cortisol to fight it, but your adrenals couldn't keep up. That's why your blood pressure dropped dangerously low and your blood sugar fell.
We've given you a high dose of cortisol through a drip to replace what your body can't make. You'll start to feel better within a few hours.
To prevent this happening again, you'll need to take cortisol tablets every day for the rest of your life. Most importantly, whenever you're unwell—with a fever, vomiting, or even a bad cold—you must double your dose immediately. If you're vomiting and can't keep tablets down, you'll need to use an emergency injection that we'll teach you how to give yourself.
This is a serious condition, but with the right medication and education, you can live a completely normal life."
16. References
-
Bornstein SR, Allolio B, Arlt W, et al. Diagnosis and Treatment of Primary Adrenal Insufficiency: An Endocrine Society Clinical Practice Guideline. J Clin Endocrinol Metab. 2016;101(2):364-389. doi:10.1210/jc.2015-1710
-
Erichsen MM, Løvås K, Skinningsrud B, et al. Clinical, immunological, and genetic features of autoimmune primary adrenal insufficiency: observations from a Norwegian registry. J Clin Endocrinol Metab. 2009;94(12):4882-4890. doi:10.1210/jc.2009-1368
-
Rushworth RL, Torpy DJ, Falhammar H. Adrenal Crisis. N Engl J Med. 2019;381(9):852-861. doi:10.1056/NEJMra1807486
-
Bergthorsdottir R, Leonsson-Zachrisson M, Odén A, Johannsson G. Premature mortality in patients with Addison's disease: a population-based study. J Clin Endocrinol Metab. 2006;91(12):4849-4853. doi:10.1210/jc.2006-0076
-
Society for Endocrinology. Emergency Management of Acute Adrenal Insufficiency (Adrenal Crisis) in Adult Patients. Endocrinology.org. Published 2024. Accessed January 6, 2026.
-
Addison T. On the Constitutional and Local Effects of Disease of the Suprarenal Capsules. London: Samuel Highley; 1855.
-
White K, Arlt W. Adrenal crisis in treated Addison's disease: a predictable but under-managed event. Eur J Endocrinol. 2010;162(1):115-120. doi:10.1530/EJE-09-0559
-
Hahner S, Spinnler C, Fassnacht M, et al. High incidence of adrenal crisis in educated patients with chronic adrenal insufficiency. J Clin Endocrinol Metab. 2015;100(2):407-416. doi:10.1210/jc.2014-3191
-
Hahner S, Burger-Stritt S, Allolio B. Subcutaneous hydrocortisone administration for emergency use in adrenal insufficiency. Eur J Endocrinol. 2013;169(2):147-154. doi:10.1530/EJE-12-1057
-
Annane D, Pastores SM, Rochwerg B, et al. Guidelines for the diagnosis and management of critical illness-related corticosteroid insufficiency (CIRCI) in critically ill patients (Part I): Society of Critical Care Medicine (SCCM) and European Society of Intensive Care Medicine (ESICM) 2017. Crit Care Med. 2017;45(12):2078-2088. doi:10.1097/CCM.0000000000002737
-
Sapolsky RM, Romero LM, Munck AU. How do glucocorticoids influence stress responses? Integrating permissive, suppressive, stimulatory, and preparative actions. Endocr Rev. 2000;21(1):55-89. doi:10.1210/edrv.21.1.0389
-
Klement J, Michels A, Reincke M, Schopohl J, Bidlingmaier M, Allolio B. Clinical and biochemical characteristics of adrenal crisis: prospective study of 100 patients. Endocr Pract. 2015;21(1):1-45. doi:10.4158/EP14161.OR
-
Puar TH, Stikkelbroeck NM, Smans LC, Zelissen PM, Hermus AR. Adrenal Crisis: Still a Deadly Event in the 21st Century. Am J Med. 2016;129(3):339.e1-339.e9. doi:10.1016/j.amjmed.2015.08.021
-
Arlt W, Allolio B. Adrenal insufficiency. Lancet. 2003;361(9372):1881-1893. doi:10.1016/S0140-6736(03)13492-7
-
Venkatesh B, Myburgh J, Finfer S, et al. The ADRENAL study protocol: adjunctive corticosteroid treatment in critically ill patients with septic shock. Crit Care Resusc. 2013;15(2):83-88.
-
Annane D, Renault A, Brun-Buisson C, et al. Hydrocortisone plus fludrocortisone for adults with septic shock. N Engl J Med. 2018;378(9):809-818. doi:10.1056/NEJMoa1705716
-
Løvås K, Loge JH, Husebye ES. Subjective health status in Norwegian patients with Addison's disease. Clin Endocrinol (Oxf). 2002;56(5):581-588. doi:10.1046/j.1365-2265.2002.01466.x
-
Prete A, Bancos I, Arlt W. Management of Endocrine Disease: Approach to the patient with adrenal incidentaloma. Eur J Endocrinol. 2023;189(1):R1-R22. doi:10.1093/ejendo/lvad066
Last Updated: 2026-01-06 | MedVellum Editorial Team Citation Count: 18 Word Count: ~9,500 | Line Count: ~950
Frequently asked questions
Quick clarifications for common clinical and exam-facing questions.
When should I seek emergency care for acute adrenal crisis?
Seek immediate emergency care if you experience any of the following warning signs: Hypotension unresponsive to 2L fluid bolus, Severe hypoglycaemia (Insulin-antagonist failure), Hyperkalaemia with ECG changes (Primary AI), Fever with no obvious source in a steroid-user, Vomiting preventing oral steroid intake, Unexplained shock in autoimmune disease patient, Postural hypotension less than 20mmHg drop.
Learning map
Use these linked topics to study the concept in sequence and compare related presentations.
Prerequisites
Start here if you need the foundation before this topic.
- Adrenal Physiology
- HPA Axis
Differentials
Competing diagnoses and look-alikes to compare.
- Septic Shock
- Acute Abdomen
- Diabetic Ketoacidosis
- Hypoglycaemia
Consequences
Complications and downstream problems to keep in mind.
- Prerenal AKI
- Waterhouse-Friderichsen Syndrome
- Hyponatraemic Encephalopathy