Alcohol Dependence (Alcohol Use Disorder)
Alcohol Use Disorder (AUD), previously termed alcohol dependence, is a chronic, relapsing neurobiological condition characterised by compulsive alcohol seeking and use despite harmful consequences, impaired control...
What matters first
Alcohol Use Disorder (AUD), previously termed alcohol dependence, is a chronic, relapsing neurobiological condition characterised by compulsive alcohol seeking and use despite harmful consequences, impaired control...
Wernicke's encephalopathy (confusion, ataxia, ophthalmoplegia) - Medical emergency requiring immediate IV thiamine
9 Jan 2026
Generated educational material; verify before clinical use.
Visible references section
See the concept before reading it
Study the key anatomy, imaging, and decision pathways as full teaching plates.
Clinical board
A visual summary of the highest-yield teaching signals on this page.
Urgent signals
Safety-critical features pulled from the topic metadata.
- Wernicke's encephalopathy (confusion, ataxia, ophthalmoplegia) - Medical emergency requiring immediate IV thiamine
- Delirium tremens (hallucinations, autonomic instability, seizures) - Mortality 5-15% if untreated
- Alcohol withdrawal seizures - Risk of status epilepticus
- GI bleeding (oesophageal varices, Mallory-Weiss tear) - Haemodynamic compromise
Content status and exam context
This page is AI-generated educational content. It may contain errors or omissions and is not a substitute for current guidelines, local protocols, senior clinical judgement, or professional medical advice.
MedVellum does not claim an individual clinician reviewer, board certification, or professional credential for this page unless a future version names a real, verifiable reviewer.
Clinical explanation and evidence
Alcohol Use Disorder (Alcohol Dependence)
1. Clinical Overview
Summary
Alcohol Use Disorder (AUD), previously termed alcohol dependence, is a chronic, relapsing neurobiological condition characterised by compulsive alcohol seeking and use despite harmful consequences, impaired control over consumption, tolerance, and physical withdrawal symptoms. The DSM-5 conceptualises AUD on a severity continuum (mild, moderate, severe) based on the number of diagnostic criteria met. AUD represents a major global public health burden, contributing significantly to liver disease, cardiovascular disease, neurological damage, cancers, accidents, violence, and psychiatric comorbidity.
The condition affects approximately 5.1% of the global disease burden and causes 3 million deaths annually worldwide. In the UK, approximately 1.6 million people meet criteria for alcohol dependence, with only a minority accessing treatment. Early identification using validated screening tools (AUDIT, CAGE, FAST) combined with evidence-based treatment integrating pharmacotherapy, psychosocial interventions, and mutual aid groups can substantially improve outcomes. The prevention and treatment of Wernicke's encephalopathy through timely thiamine supplementation remains critical — this is a medical emergency with potentially irreversible consequences if missed.
Key Facts
| Parameter | Value |
|---|---|
| Definition | Chronic neurobiological disorder with compulsive alcohol use, impaired control, tolerance, and withdrawal |
| DSM-5 Criteria | 2+ of 11 criteria within 12 months (Mild 2-3, Moderate 4-5, Severe 6+) |
| Prevalence (UK) | ~1.6 million people with alcohol dependence; ~600,000 not in treatment |
| Prevalence (Global) | 283 million people with AUD; 5.1% global disease burden |
| Mortality (UK) | ~24,000 deaths/year directly attributable to alcohol |
| Heritability | ~50% genetic contribution to risk |
| Key Management | Medically-assisted withdrawal + thiamine + psychosocial support + relapse prevention medication |
| Critical Emergency | Wernicke's encephalopathy — give IV Pabrinex BEFORE glucose |
| Key Screening Tools | AUDIT (gold standard), AUDIT-C (brief), CAGE (quick), FAST (ED setting), SADQ (severity) |
Clinical Pearls
"Always Thiamine BEFORE Glucose": In any patient with suspected alcohol problems and altered consciousness, nutritional deficiency, or receiving IV fluids, give IV Pabrinex (thiamine) BEFORE giving IV glucose. Glucose metabolism consumes thiamine and can precipitate or worsen Wernicke's encephalopathy. This is a crucial exam point and clinical priority.
CAGE Screening Mnemonic: Have you ever felt you should Cut down? Have people Annoyed you by criticising your drinking? Have you felt Guilty about drinking? Have you ever had a morning Eye-opener (drink first thing)? Score ≥2 positive responses suggests problematic drinking requiring further assessment.
Wernicke's Triad Recognition: Confusion, Ataxia, Ophthalmoplegia — but the full classic triad is present in only 10-16% of cases. Have a very low threshold to treat suspected cases empirically. Any patient with alcohol misuse and any neurological symptom should receive thiamine.
The "Alcohol Paradox": Higher socioeconomic groups drink more frequently, but lower socioeconomic groups experience more alcohol-related harm. This inverse relationship between consumption patterns and harm burden is important for public health planning.
CIWA-Ar Scoring: Clinical Institute Withdrawal Assessment for Alcohol (revised) guides benzodiazepine dosing in withdrawal. Score less than 10 = mild (may not need pharmacotherapy); 10-18 = moderate; > 18 = severe (high-dose benzodiazepine required). Symptom-triggered dosing reduces medication exposure and length of treatment.
Why This Matters Clinically
Alcohol use disorder is extremely common and frequently undetected in clinical practice. It affects virtually every organ system and represents a leading cause of preventable morbidity and mortality. Many patients present with physical consequences (liver disease, trauma, pancreatitis, cardiac arrhythmias) without disclosing their drinking patterns, and clinicians must maintain a high index of suspicion. Withdrawal can be life-threatening if not managed properly, yet is eminently treatable with appropriate pharmacological support. Recovery is absolutely achievable with appropriate intervention — many patients attain long-term sustained remission with quality of life comparable to the general population.
2. Epidemiology
Incidence & Prevalence
| Measure | UK Data | Global Data |
|---|---|---|
| Alcohol Dependence Prevalence | 1.6 million (4% of adults) | 283 million (5.1% of adults globally) |
| Hazardous Drinking (AUDIT ≥8) | 24% of adults in England | Varies widely by region |
| Harmful Drinking | 6% of adults | 3% globally |
| Annual Deaths | ~24,000 (UK) | 3 million (5.3% of all deaths worldwide) |
| Hospital Admissions | 1.2 million alcohol-related per year | — |
| Treatment Gap | ~600,000 dependent drinkers not in treatment | 90%+ globally |
The global burden is substantial. The WHO Global Status Report on Alcohol and Health (2018) documented that alcohol consumption contributes to more than 200 disease and injury conditions and is causally linked to major non-communicable diseases including liver cirrhosis, cardiovascular disease, and multiple cancers.
Demographics
| Factor | Details | Clinical Implications |
|---|---|---|
| Age | Peak prevalence 25-34 years; increasing prevalence in > 65s | Screen all age groups; older adults may present atypically |
| Sex | Male:Female ratio approximately 3:1 (but gap narrowing significantly) | Women develop complications faster at lower consumption levels ("telescoping") |
| Ethnicity | Lower rates in South Asian populations (cultural/religious factors); variations by culture | Culturally-sensitive screening and intervention approaches |
| Socioeconomic | Higher SES groups drink more frequently; lower SES groups experience more harm (alcohol harm paradox) | Target interventions appropriately |
| Occupation | Higher rates in hospitality, military, healthcare, journalism | Occupational health screening programmes |
Risk Factors
Non-Modifiable Risk Factors:
- Genetic factors: Heritability approximately 50%; polymorphisms in ADH1B, ALDH2, GABRA2, OPRM1 genes influence risk
- Family history: First-degree relative with AUD increases risk 4-7 fold
- Male sex: Higher prevalence (though women have accelerated progression)
- Early age of first drink: Onset less than 15 years increases lifetime AUD risk significantly
- Psychiatric comorbidity: Depression, anxiety disorders, PTSD, ADHD, bipolar disorder
- Personality traits: Impulsivity, sensation-seeking, neuroticism
- Adverse childhood experiences: Trauma, abuse, neglect
Modifiable Risk Factors:
| Risk Factor | Impact | Evidence-Based Intervention |
|---|---|---|
| High stress/trauma | Elevated risk via self-medication | Trauma-informed care, stress management |
| Social environment | Heavy drinking culture normalises excess | Social network interventions, changing norms |
| Alcohol availability | Direct relationship with consumption | Minimum unit pricing, licensing restrictions |
| Mental health comorbidity | Bidirectional relationship | Integrated dual diagnosis treatment |
| Peer group drinking | Strong influence on patterns | Family and social interventions |
| Low cost of alcohol | Increases consumption | Pricing policy (minimum unit pricing) |
| Advertising exposure | Influences attitudes and consumption | Regulatory restrictions |
Burden of Disease
| Consequence | UK Scale | Clinical Relevance |
|---|---|---|
| Deaths directly attributable | ~24,000/year | Leading preventable cause |
| Hospital admissions (alcohol-related) | ~1.2 million/year | Major NHS burden |
| Economic cost | £21-52 billion/year (healthcare, crime, lost productivity) | Societal impact |
| Alcohol-related liver disease | Leading cause of liver death | Preventable with abstinence |
| Alcohol-related brain injury | Significant disability burden | Often irreversible |
| Foetal alcohol spectrum disorder | Leading preventable cause of intellectual disability | Completely preventable |
| Years of Life Lost (YLL) | 167,000/year in UK | Premature mortality |
| Disability-Adjusted Life Years (DALYs) | 5.1% of global total | Major disability burden |
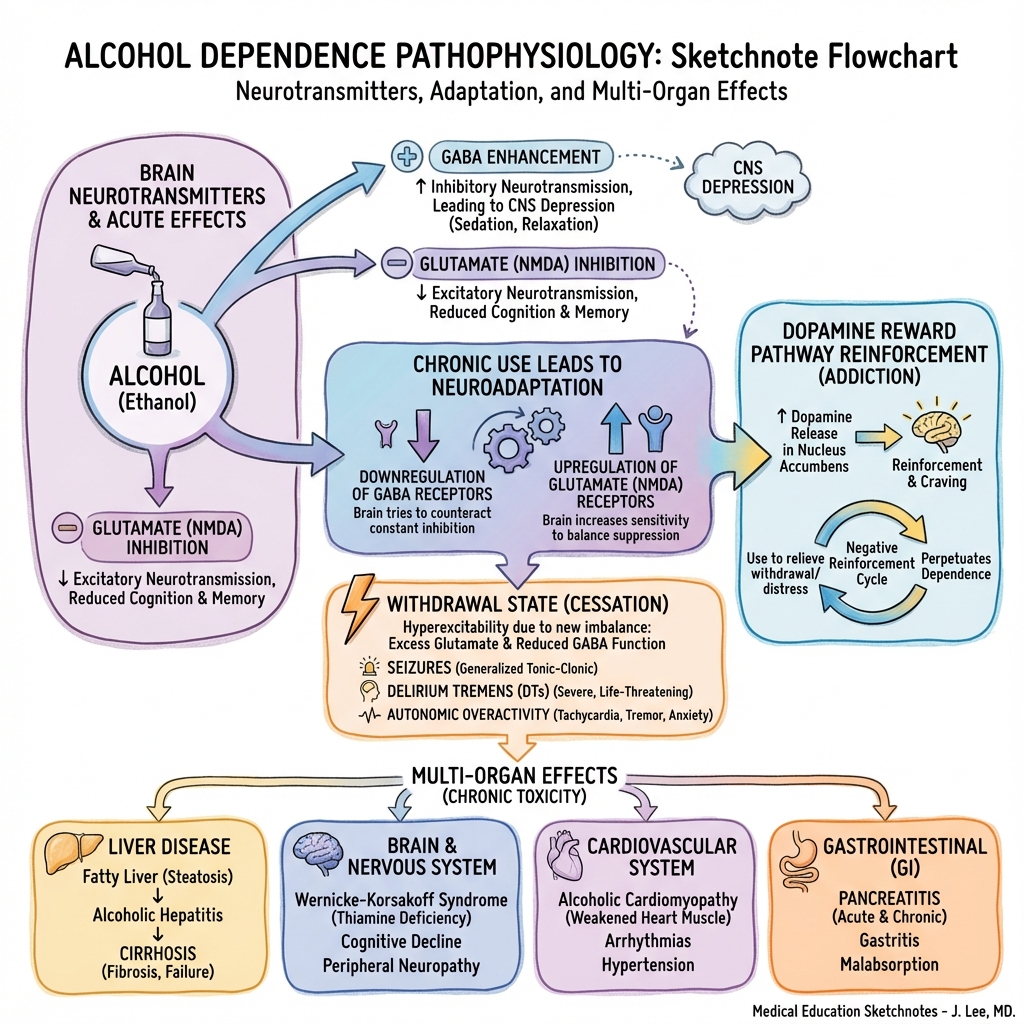
3. Pathophysiology
Neurobiological Mechanism
Alcohol produces its effects through complex interactions with multiple neurotransmitter systems, leading to neuroadaptive changes that underpin tolerance, dependence, and withdrawal.
Stage 1: Acute Alcohol Effects (Intoxication)
| System | Acute Effect | Clinical Manifestation |
|---|---|---|
| GABA-A receptors | Potentiates inhibitory signalling | Sedation, anxiolysis, muscle relaxation, ataxia |
| NMDA receptors | Inhibits excitatory glutamate transmission | Cognitive impairment, amnesia, analgesia |
| Dopamine (mesolimbic) | Increases dopamine release in nucleus accumbens | Euphoria, reward, positive reinforcement |
| Opioid system | Stimulates endorphin release | Euphoria, reward |
| Serotonin | Modulates serotonergic transmission | Mood effects |
| Voltage-gated calcium channels | Inhibition | Reduced neural excitability |
Stage 2: Neuroadaptation (Chronic Use → Tolerance)
With repeated exposure, the brain adapts to maintain homeostasis:
| Adaptation | Mechanism | Consequence |
|---|---|---|
| GABA-A receptor downregulation | Reduced receptor number and sensitivity | Tolerance to sedative effects; need more alcohol for same effect |
| NMDA receptor upregulation | Increased receptor number and sensitivity | Tolerance; neuronal hyperexcitability when alcohol removed |
| Dopamine system blunting | Reduced baseline dopamine signalling | Anhedonia, craving, negative affect when not drinking |
| Stress system sensitisation | HPA axis dysregulation, CRF upregulation | Anxiety, dysphoria, stress-induced craving |
Stage 3: Withdrawal State (CNS Hyperexcitability)
When alcohol is removed, the adapted brain enters a hyperexcitable state:
| Pathophysiology | Clinical Manifestation |
|---|---|
| Unopposed glutamate hyperactivity (upregulated NMDA) | Tremor, anxiety, agitation, seizures |
| Reduced GABA inhibition | Autonomic instability, insomnia, hyperreflexia |
| Sympathetic nervous system overactivity | Tachycardia, hypertension, sweating, fever |
| Dopamine deficiency | Dysphoria, anhedonia, craving |
| Severe cases | Seizures, delirium tremens, death |
Stage 4: Addiction Cycle (Compulsive Use)
| Component | Mechanism | Clinical Feature |
|---|---|---|
| Negative reinforcement | Drinking to avoid/relieve withdrawal symptoms | Morning drinking, relief drinking |
| Positive reinforcement | Dopamine-mediated reward | Craving, cue-induced desire |
| Compulsive use | Prefrontal cortex dysfunction, impaired executive control | Loss of control despite consequences |
| Salience | Alcohol becomes primary focus | Narrowing of behavioural repertoire |
| Cue reactivity | Conditioned responses to alcohol-associated stimuli | Relapse triggered by environmental cues |
Multi-Organ System Damage
Chronic alcohol use causes widespread organ pathology through multiple mechanisms including direct toxicity, metabolic effects, nutritional deficiencies, and immune dysregulation.
| Organ System | Pathology | Key Points |
|---|---|---|
| Liver | Steatosis → Alcoholic hepatitis → Fibrosis → Cirrhosis → Hepatocellular carcinoma | Spectrum of ARLD; steatosis reversible; cirrhosis may stabilise with abstinence |
| Brain | Wernicke-Korsakoff syndrome; cerebellar degeneration; cerebral atrophy; alcohol-related dementia | Thiamine deficiency is key; some recovery possible with abstinence |
| Heart | Alcoholic cardiomyopathy (dilated); arrhythmias ("holiday heart" syndrome); hypertension | Cardiomyopathy may improve with abstinence |
| Pancreas | Acute pancreatitis; chronic pancreatitis with exocrine/endocrine failure | Leading cause of chronic pancreatitis |
| GI Tract | Gastritis; oesophageal varices; Mallory-Weiss tears; increased cancer risk | Varices = decompensated cirrhosis |
| Immune System | Impaired cellular and humoral immunity; increased infection susceptibility | Higher pneumonia, TB, surgical infection risk |
| Haematology | Macrocytosis; thrombocytopenia; anaemia (multiple causes) | MCV useful screening marker |
| Musculoskeletal | Myopathy; osteoporosis; increased fracture risk | Proximal weakness |
| Cancer | Mouth, pharynx, larynx, oesophagus, liver, breast, colorectal | Dose-dependent risk; no safe threshold for cancer |
| Reproductive | Hypogonadism; erectile dysfunction; infertility; foetal alcohol syndrome | FASD entirely preventable |
Wernicke-Korsakoff Syndrome: Critical Pathophysiology
This thiamine (vitamin B1) deficiency syndrome is a medical emergency and exam favourite.
| Feature | Wernicke's Encephalopathy | Korsakoff's Syndrome |
|---|---|---|
| Timing | Acute | Chronic (follows untreated Wernicke's) |
| Reversibility | Reversible if treated early and aggressively | Largely irreversible |
| Classic Triad | Confusion (82%), Ataxia (23%), Ophthalmoplegia (29%) | Not applicable |
| Full Triad Present | Only 10-16% of cases | — |
| Core Feature | Acute confusional state | Anterograde amnesia + confabulation |
| Pathology | Haemorrhagic lesions in mammillary bodies, thalamus, periaqueductal grey | Chronic damage to mammillary bodies, thalamic nuclei |
| Treatment | IV Pabrinex (high-dose thiamine) immediately | Prevention (treat Wernicke's aggressively) |
| Prognosis | Good if treated within 48-72 hours | 80% develop chronic memory impairment |
Mechanism: Thiamine pyrophosphate is essential cofactor for:
- Pyruvate dehydrogenase (glycolysis → citric acid cycle)
- α-ketoglutarate dehydrogenase (citric acid cycle)
- Transketolase (pentose phosphate pathway)
Deficiency → impaired ATP production in metabolically active brain regions → neuronal death.
Why alcohol causes thiamine deficiency:
- Poor dietary intake
- Impaired GI absorption
- Reduced hepatic storage
- Reduced phosphorylation to active form
- Increased utilisation
4. Clinical Presentation
DSM-5 Diagnostic Criteria for Alcohol Use Disorder
A problematic pattern of alcohol use leading to clinically significant impairment or distress, manifested by ≥2 of the following within a 12-month period:
| Criterion | Description | Domain |
|---|---|---|
| 1 | Alcohol taken in larger amounts or over longer period than intended | Impaired control |
| 2 | Persistent desire or unsuccessful efforts to cut down or control use | Impaired control |
| 3 | Great deal of time spent obtaining, using, or recovering from alcohol | Impaired control |
| 4 | Craving, or strong desire/urge to use alcohol | Impaired control |
| 5 | Recurrent use resulting in failure to fulfil major role obligations (work, school, home) | Social impairment |
| 6 | Continued use despite persistent/recurrent social or interpersonal problems caused or exacerbated by alcohol | Social impairment |
| 7 | Important social, occupational, or recreational activities given up or reduced because of alcohol use | Social impairment |
| 8 | Recurrent use in situations where it is physically hazardous | Risky use |
| 9 | Continued use despite knowledge of persistent physical or psychological problem likely caused or exacerbated by alcohol | Risky use |
| 10 | Tolerance (need for increased amounts OR diminished effect with same amount) | Pharmacological |
| 11 | Withdrawal (characteristic syndrome OR alcohol/similar substance taken to relieve or avoid withdrawal) | Pharmacological |
Severity Specifiers:
- Mild: 2-3 criteria
- Moderate: 4-5 criteria
- Severe: 6 or more criteria
ICD-11 Criteria (for comparison)
ICD-11 distinguishes between:
- Hazardous alcohol use (pattern increasing risk of harm)
- Harmful alcohol use (pattern causing health damage)
- Alcohol dependence (impaired control, physiological features, continued use despite harm)
Alcohol Withdrawal Syndrome: Timeline and Features
| Phase | Timing | Features | Severity |
|---|---|---|---|
| Early withdrawal | 6-12 hours | Tremor, anxiety, insomnia, nausea, sweating, tachycardia, hypertension | Mild-moderate |
| Withdrawal seizures | 12-48 hours (peak 24h) | Generalised tonic-clonic seizures (brief, self-limiting usually) | Severe |
| Alcoholic hallucinosis | 12-48 hours | Visual, auditory, or tactile hallucinations with CLEAR SENSORIUM | Moderate-severe |
| Delirium tremens | 48-96 hours (peak 72h) | Confusion/delirium, vivid hallucinations, marked autonomic instability, agitation | Life-threatening |
Early Withdrawal Symptoms (6-24 hours):
- Tremor — Classic early sign, often coarse, postural
- Sweating — Diaphoresis, often profuse
- Tachycardia — Heart rate > 100 bpm
- Hypertension — Elevated blood pressure
- Anxiety/agitation — Restlessness, irritability
- Nausea and vomiting — GI upset
- Insomnia — Difficulty sleeping
- Headache — Common complaint
Delirium Tremens (48-96 hours, Severe):
[!CAUTION] Delirium Tremens is a medical emergency with 5-15% mortality if untreated.
- Severe confusion/delirium with fluctuating level of consciousness
- Vivid visual hallucinations (classically small animals, insects — "formication")
- Profound autonomic instability (fever, severe tachycardia, hypertension, sweating)
- Marked agitation, tremulousness
- Risk factors: Prior DTs, concurrent illness, older age, severe dependence, high recent intake
Physical Examination Findings
Signs of Acute Intoxication:
- Smell of alcohol on breath
- Slurred speech
- Unsteady gait/ataxia
- Nystagmus
- Conjunctival injection
- Reduced consciousness
Signs of Chronic Alcohol Use:
| Region | Signs |
|---|---|
| Hands | Dupuytren's contracture; palmar erythema; leuconychia; clubbing (if cirrhosis); tremor |
| Face | Parotid enlargement; telangiectasia; rhinophyma; facial flushing |
| Eyes | Jaundice (scleral icterus); ophthalmoplegia (Wernicke's); nystagmus |
| Chest | Spider naevi (> 5 in SVC distribution = significant); gynaecomastia; reduced body hair |
| Abdomen | Hepatomegaly (early) OR small shrunken liver (late cirrhosis); splenomegaly; ascites; caput medusae; testicular atrophy |
| Neurological | Peripheral neuropathy (glove-and-stocking sensory loss); cerebellar signs (ataxia, dysarthria, intention tremor); cognitive impairment |
| General | Poor nutritional status; muscle wasting; bruising (coagulopathy); signs of trauma |
Red Flags Requiring Urgent Action
[!CAUTION] Red Flags — Immediate Action Required:
Red Flag Action Rationale Wernicke's encephalopathy (any of: confusion, ataxia, eye signs, hypothermia) Pabrinex IV 2 pairs TDS immediately (BEFORE glucose) Medical emergency; irreversible if untreated Delirium tremens (confusion + hallucinations + autonomic instability) HDU/ICU; high-dose benzodiazepines; IV fluids; thiamine 5-15% mortality untreated Withdrawal seizures Benzodiazepine; exclude other causes; prevent aspiration Risk of status epilepticus GI bleeding (haematemesis, melaena) Resuscitation; urgent endoscopy; variceal protocol if suspected Variceal bleeding has high mortality Hypoglycaemia Check glucose; treat with IV dextrose (AFTER thiamine) Impaired gluconeogenesis Suicidal ideation Psychiatric assessment; safety planning; supervision High comorbidity; high risk Signs of liver failure (encephalopathy, coagulopathy, jaundice) Urgent gastroenterology/hepatology input Decompensated cirrhosis Aspiration pneumonia Airway protection; antibiotics Common complication
5. Clinical Examination
Structured Approach
General Inspection:
- Nutritional status (often poor, cachectic)
- Signs of intoxication vs withdrawal
- Stigmata of chronic liver disease
- Signs of self-neglect
- Trauma/bruising
- Tremor (hands, tongue)
OSCE-Style Examination Sequence:
-
Hands and Arms
- Tremor (ask patient to hold hands outstretched)
- Dupuytren's contracture (palmar fascia thickening)
- Palmar erythema (thenar and hypothenar)
- Leuconychia (white nails — hypoalbuminaemia)
- Clubbing (if cirrhosis)
- Bruising (coagulopathy)
- Muscle wasting
- Asterixis/flapping tremor (hepatic encephalopathy)
-
Face and Eyes
- Jaundice (scleral icterus)
- Parotid enlargement (bilateral)
- Telangiectasia
- Nystagmus
- Ophthalmoplegia (6th nerve palsy most common in Wernicke's)
- Conjunctival pallor (anaemia)
-
Chest
- Spider naevi (count — > 5 significant)
- Gynaecomastia
- Loss of axillary hair
-
Abdomen
- Distension (ascites)
- Caput medusae
- Hepatomegaly OR small liver
- Splenomegaly
- Shifting dullness and fluid thrill
-
Neurological
- Peripheral neuropathy (glove-and-stocking pattern)
- Cerebellar examination (ataxia, dysarthria, nystagmus, intention tremor)
- Cognitive screening (confusion, orientation, memory)
- Gait assessment
-
Mental State
- Orientation
- Mood (depression common)
- Suicidal ideation
- Insight
- Motivation for change
Screening Tools
| Tool | Components | Scoring | Interpretation | Use |
|---|---|---|---|---|
| AUDIT | 10 questions covering consumption, dependence, and harm | 0-40 | 0-7: Low risk; 8-15: Hazardous; 16-19: Harmful; 20+: Possible dependence | Gold standard screening; recommended by WHO |
| AUDIT-C | First 3 AUDIT questions (consumption only) | 0-12 | ≥5 (men) or ≥4 (women) positive screen | Quick primary care screen |
| CAGE | 4 questions: Cut down, Annoyed, Guilty, Eye-opener | 0-4 | ≥2: Likely problem | Very quick; good for identifying dependence |
| FAST | 4 questions adapted from AUDIT | 0-16 | ≥3: Hazardous drinking | Designed for ED/busy settings |
| SADQ | 20 items assessing severity of dependence | 0-60 | Mild less than 16; Moderate 16-30; Severe 31+ | Determines need for inpatient detox |
| CIWA-Ar | 10-item symptom severity rating | 0-67 | less than 10: Mild; 10-18: Moderate; > 18: Severe | Guides withdrawal medication dosing |
| PAWSS | Prediction of Alcohol Withdrawal Severity Scale | 0-10 | ≥4: High risk for complicated withdrawal | Predicts need for prophylaxis |
AUDIT Questions (for reference):
- How often do you have a drink containing alcohol?
- How many units do you drink on a typical drinking day?
- How often do you have 6+ units on a single occasion?
- How often have you found you were unable to stop drinking once started?
- How often have you failed to do what was expected because of drinking?
- How often have you needed a first drink in the morning?
- How often have you had guilt or remorse after drinking?
- How often have you been unable to remember the night before?
- Have you or someone else been injured due to your drinking?
- Has a relative, friend, or health worker suggested you cut down?
6. Investigations
Bedside Tests
| Test | Purpose | Expected Findings |
|---|---|---|
| Blood glucose | Exclude hypoglycaemia | Often low (impaired gluconeogenesis) |
| Breathalyser/blood alcohol | Current intoxication | Quantifies recent intake |
| Vital signs | Assess withdrawal severity | Tachycardia, hypertension, fever in withdrawal |
| Temperature | Hypothermia in Wernicke's; fever in DTs | Important prognostic indicator |
| Capillary blood gas | Acid-base status | May show metabolic acidosis |
| ECG | Cardiac assessment | Arrhythmias, prolonged QT, atrial fibrillation |
Laboratory Investigations
| Test | Expected Finding | Clinical Significance |
|---|---|---|
| GGT (Gamma-GT) | Elevated (most sensitive marker) | Rises within days-weeks of heavy drinking; falls with abstinence |
| MCV | Elevated (macrocytosis) | Chronic alcohol effect; slow to normalise (months) |
| AST | Elevated | Liver damage marker |
| ALT | Elevated (but less than AST) | — |
| AST:ALT ratio | > 2:1 typical of alcoholic liver disease | Helps differentiate from viral/NAFLD |
| Bilirubin | Elevated in liver disease | Jaundice if > 50 μmol/L |
| Albumin | Low in cirrhosis | Marker of synthetic function |
| INR/PT | Prolonged in liver failure | Coagulation factor synthesis impaired |
| FBC | Macrocytosis; thrombocytopenia; anaemia | Multiple mechanisms |
| U&E | Hypokalaemia; hypomagnesaemia; hyponatraemia | Electrolyte disturbances common |
| Phosphate | Often low | Refeeding risk |
| Magnesium | Often low | Contributes to seizure risk; supplement |
| CDT | Elevated with chronic heavy drinking | Carbohydrate-deficient transferrin; objective biomarker |
| EtG/EtS (urine) | Detects recent drinking (up to 80 hours) | Useful for monitoring abstinence |
| Ammonia | Elevated in hepatic encephalopathy | Check if confusion with known cirrhosis |
| Vitamin B12, Folate, Ferritin | Often deficient | Nutritional assessment |
| Lipase/Amylase | Elevated in pancreatitis | If abdominal pain |
| Hepatitis serology | Coinfection common | HBV, HCV screening recommended |
Imaging
| Modality | Findings | Indication |
|---|---|---|
| Liver ultrasound | Fatty change (increased echogenicity); cirrhosis (irregular surface, coarse texture); splenomegaly; ascites; portal vein patency | Initial liver assessment; exclude other pathology |
| FibroScan (Transient Elastography) | Liver stiffness measurement (kPa) | Non-invasive fibrosis staging; increasingly standard care |
| CT Abdomen | Liver morphology; portal hypertension features; varices; hepatocellular carcinoma screening | Complications assessment |
| MRI Brain | Wernicke's: mammillary body and periaqueductal grey signal change; cerebral atrophy; cerebellar degeneration | Neurological complications |
| CT Head | Acute intracranial pathology (haemorrhage, infarct) | Altered consciousness; head injury; focal neurology |
| Endoscopy (OGD) | Oesophageal varices; gastritis; Mallory-Weiss tears | GI bleeding; cirrhosis screening |
Diagnostic Criteria Summary
DSM-5 Alcohol Use Disorder Diagnosis:
- ≥2 of 11 criteria within 12-month period
- Mild: 2-3 criteria
- Moderate: 4-5 criteria
- Severe: 6+ criteria
ICD-11 Alcohol Dependence:
- Impaired control over alcohol use
- Increasing priority of alcohol over other activities
- Physiological features (tolerance and/or withdrawal)
- Persistent use despite harm
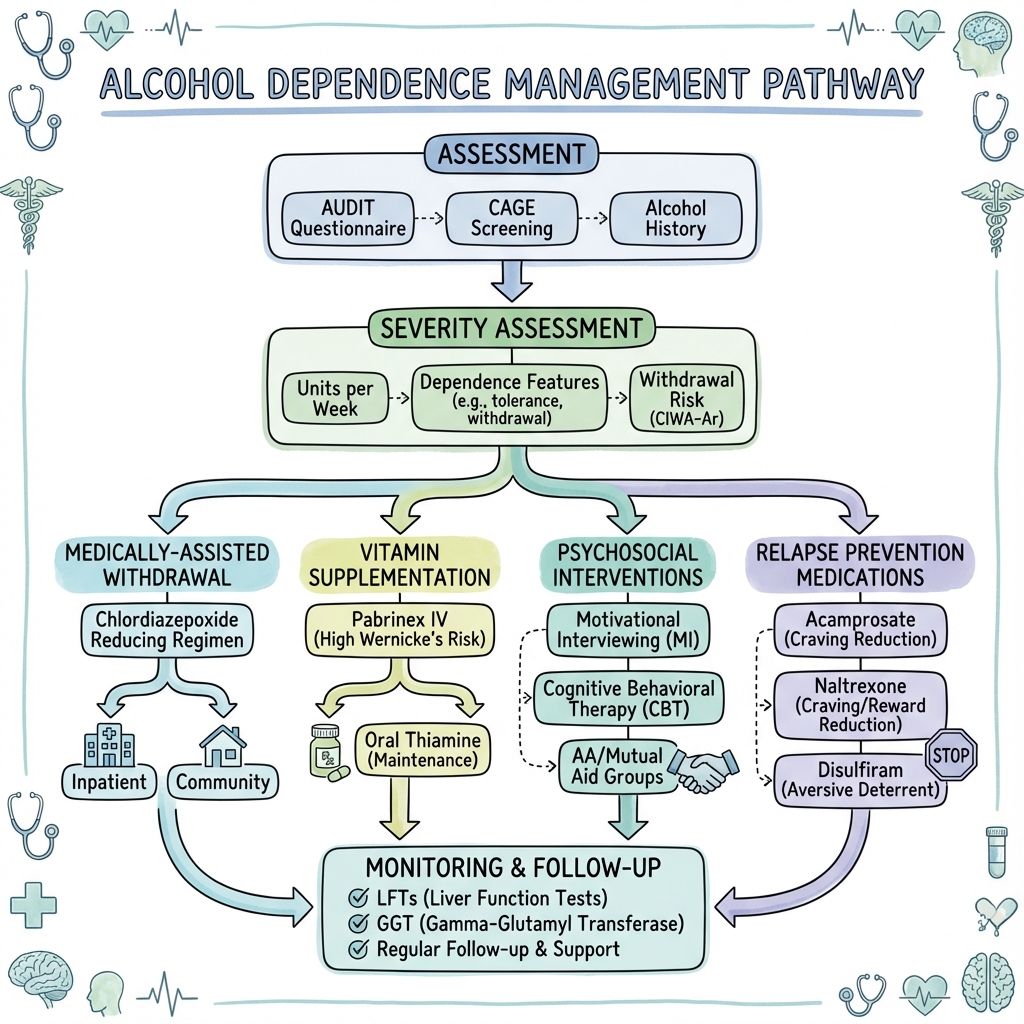
7. Management
Management Overview
Management of AUD involves four key phases:
- Assessment and stabilisation (including emergency management)
- Medically-assisted withdrawal (detoxification)
- Relapse prevention (pharmacological and psychosocial)
- Long-term recovery support
Community vs Inpatient Withdrawal: Decision Framework
| Factor | Community Suitable | Inpatient Required |
|---|---|---|
| Dependence severity (SADQ) | Mild-moderate (less than 30) | Severe (≥31) |
| Previous withdrawal complications | None | History of seizures, DTs |
| Current withdrawal severity (CIWA-Ar) | less than 10 | > 15 or rapidly escalating |
| Comorbid medical illness | Stable/none | Significant or unstable |
| Comorbid psychiatric illness | Stable | Active suicidality, severe mental illness |
| Social support | Reliable support available | Poor support or homeless |
| Poly-substance use | No | Yes (especially benzodiazepines, opioids) |
| Home environment | Safe, alcohol-free | Unsafe, ongoing alcohol access |
| Previous treatment attempts | First attempt or prior success | Multiple failed community detox |
| Physical health | Good | Poor nutrition, frailty |
| Pregnancy | — | Inpatient preferred |
Medically-Assisted Withdrawal (Detoxification)
Benzodiazepine Regimens:
| Drug | Typical Regimen | Duration | Notes |
|---|---|---|---|
| Chlordiazepoxide (Librium) | Fixed reducing dose OR symptom-triggered | 7-10 days | First-line UK; long half-life; less abuse potential |
| Diazepam | Fixed reducing dose OR symptom-triggered | 7-10 days | Alternative; very long half-life |
| Lorazepam | Symptom-triggered OR fixed dose | Variable | Preferred in liver disease (no active metabolites); shorter acting |
| Oxazepam | Fixed dose | Variable | Alternative in liver disease |
Example Fixed-Dose Chlordiazepoxide Regimen (Moderate Dependence, SADQ 15-30):
| Day | Dose |
|---|---|
| Day 1-2 | 25-30 mg QDS |
| Day 3-4 | 20 mg QDS |
| Day 5-6 | 15 mg TDS |
| Day 7 | 10 mg BD |
| Day 8 | 10 mg OD |
| Day 9 | 5 mg OD |
| Day 10 | Stop |
Symptom-Triggered Dosing (Using CIWA-Ar):
- Assess CIWA-Ar score hourly initially
- CIWA-Ar less than 10: No medication required; reassess in 4-8 hours
- CIWA-Ar 10-18: Give chlordiazepoxide 10-20 mg; reassess in 1 hour
- CIWA-Ar > 18: Give chlordiazepoxide 20-40 mg; reassess in 1 hour
- Continue until CIWA-Ar less than 10 for 24-48 hours
Advantages of symptom-triggered: Less total medication, shorter treatment duration, reduced sedation.
Carbamazepine:
- Alternative if benzodiazepines contraindicated
- 200 mg TDS reducing over 7 days
- Does not prevent seizures as effectively as benzodiazepines
Thiamine and Vitamin Supplementation: Critical
| Clinical Scenario | Thiamine Regimen | Rationale |
|---|---|---|
| Wernicke's encephalopathy (suspected or confirmed) | Pabrinex IV: 2-3 pairs TDS for 3-5 days minimum, then 1 pair OD until no further improvement | Medical emergency; treat empirically; continue until no further response |
| High risk for Wernicke's (malnourished, vomiting, peripheral neuropathy, delirium, poor diet) | Pabrinex IV: 1-2 pairs OD for 3-5 days, then oral thiamine 100 mg TDS | Prophylactic high-dose treatment |
| Standard withdrawal (moderate risk) | Pabrinex IM: 1 pair OD for 3-5 days, OR oral thiamine 100-300 mg daily | Ensure adequate stores before and during detox |
| Low risk | Oral thiamine 100 mg TDS during detox, then 100 mg OD for 1 month | Minimum standard |
| Maintenance (if continued drinking risk) | Oral thiamine 100 mg OD indefinitely | Ongoing prophylaxis |
CRITICAL PRINCIPLES:
- Thiamine BEFORE glucose — always
- IV/IM before oral — GI absorption impaired in alcohol-dependent patients
- High dose for high risk — Pabrinex contains 250 mg thiamine per pair
- Treat empirically — do not wait for confirmation in suspected Wernicke's
- Duration matters — continue until no further improvement, minimum 5 days IV for Wernicke's
Other Nutritional Supplementation:
- Magnesium (often depleted; contributes to seizure risk)
- Phosphate (monitor for refeeding syndrome)
- Potassium (frequently low)
- Folate
- B-complex vitamins
Relapse Prevention Pharmacotherapy
| Drug | Mechanism | Dose | Key Evidence | Notes |
|---|---|---|---|---|
| Acamprosate | NMDA receptor modulation; normalises glutamate/GABA balance | 666 mg TDS (333 mg TDS if less than 60 kg) | NNT 9 for sustained abstinence at 6 months (Cochrane 2010) | First-line for abstinence maintenance; start after detox complete; well tolerated |
| Naltrexone | Opioid antagonist; blocks rewarding effects of alcohol | 50 mg OD (oral) OR 380 mg IM monthly | NNT 12 for preventing return to heavy drinking | Reduces heavy drinking days; consider for harm reduction; check LFTs |
| Disulfiram (Antabuse) | Aldehyde dehydrogenase inhibitor; causes aversive reaction if drinks | 200-500 mg OD | Effective with supervision; NNT 8-12 in supervised settings | Deterrent; requires supervision for efficacy; contraindicated in liver failure, cardiac disease |
| Nalmefene | Opioid receptor modulator (partial agonist/antagonist) | 18 mg PRN (before anticipated drinking) | Licensed for harm reduction, not abstinence | For reducing consumption, not abstinence; take 1-2 hours before drinking |
| Baclofen | GABA-B agonist | Variable (up to 30-80 mg/day in trials) | Mixed evidence; may be beneficial in liver disease | Off-label; consider in advanced liver disease where others contraindicated |
| Topiramate | Multiple mechanisms | Variable | Some evidence for reducing heavy drinking | Off-label; cognitive side effects limit use |
| Gabapentin | GABA modulation | Variable | Some evidence for efficacy | Off-label |
NICE Recommendations:
- Offer acamprosate OR oral naltrexone as first-line relapse prevention
- Consider disulfiram for those who prefer a deterrent approach (with supervision)
- Consider nalmefene for reducing consumption in those not seeking immediate abstinence
Psychosocial Interventions
| Intervention | Description | Evidence | Setting |
|---|---|---|---|
| Brief Intervention (IBA) | 5-15 minute structured advice following FRAMES model | Strong evidence for hazardous/harmful drinking (NNT 8) | Primary care, ED, general hospital |
| Motivational Interviewing (MI) | Client-centred counselling to enhance motivation for change | Good evidence; particularly for ambivalent patients | Any setting |
| Cognitive Behavioural Therapy (CBT) | Identify and modify maladaptive thoughts and behaviours | Good evidence | Specialist services |
| Motivational Enhancement Therapy (MET) | Structured motivational intervention (4 sessions) | Strong evidence (Project MATCH) | Specialist services |
| 12-Step Facilitation (TSF) | Preparation for and engagement with 12-step programmes | Strong evidence equal to other therapies | Specialist services |
| Alcoholics Anonymous (AA) | Peer support, 12-step programme | Strong evidence for those who engage | Community |
| SMART Recovery | CBT-based mutual aid, non-spiritual | Growing evidence | Community |
| Family/Couple Therapy | Involves support network | Good evidence | Specialist services |
| Contingency Management | Incentives for abstinence | Emerging evidence | Research/some services |
| Community Reinforcement Approach (CRA) | Environmental focus, skills training | Good evidence | Specialist services |
FRAMES Model for Brief Intervention:
- Feedback: Provide personalised feedback on risk/harm
- Responsibility: Emphasise personal responsibility for change
- Advice: Give clear advice to reduce/stop drinking
- Menu: Offer a menu of options/strategies
- Empathy: Use empathic listening style
- Self-efficacy: Support belief in ability to change
Referral Criteria
Refer to Specialist Addiction Services:
- Moderate-severe AUD (AUDIT ≥20, SADQ ≥15)
- Failed brief intervention in primary care
- Need for assisted withdrawal
- Complex needs (psychiatric comorbidity, poly-substance use)
- Pregnancy
- Young people (less than 18 years)
Refer to Inpatient/Residential Treatment:
- Severe dependence (SADQ ≥31)
- Failed community detox
- Unstable housing/homelessness
- Severe psychiatric comorbidity
- Complex medical needs
- History of complicated withdrawal
Urgent/Emergency Referral:
- Suspected Wernicke's encephalopathy
- Delirium tremens
- Withdrawal seizures
- Severe withdrawal (CIWA-Ar > 18)
- Suicidal ideation with plan/intent
- GI bleeding/acute liver failure
Disposition and Follow-up
| Phase | Setting | Key Actions |
|---|---|---|
| Acute intoxication | ED/AMU | Supportive care; observation; assess for withdrawal risk; thiamine |
| Withdrawal | Inpatient or community | Benzodiazepine regimen; thiamine; hydration; monitoring |
| Post-detox | Outpatient addiction service | Initiate relapse prevention medication; psychosocial intervention; link to mutual aid |
| Recovery maintenance | Primary care + addiction service | Ongoing support; medication review; monitor for relapse; physical health screening |
| Relapse | Addiction service | Reassessment; intensified support; consider different approach |
8. Complications
Immediate Complications (Hours to Days)
| Complication | Incidence | Presentation | Management |
|---|---|---|---|
| Withdrawal seizures | 3-5% of withdrawals | Generalised tonic-clonic, 12-48h post cessation | Benzodiazepines; exclude other causes (hypoglycaemia, head injury, electrolytes); prevent aspiration |
| Delirium tremens | 3-5% of hospitalised withdrawals | Confusion, hallucinations, autonomic storm, 48-96h | High-dose IV benzodiazepines; HDU/ICU; IV fluids; thiamine; treat hyperthermia |
| Wernicke's encephalopathy | 2-3% (likely underdiagnosed) | Confusion, ataxia, ophthalmoplegia (triad in 10-16%) | Pabrinex IV 2-3 pairs TDS immediately; continue until no further improvement |
| Hypoglycaemia | Common | Confusion, sweating, tremor, seizures | IV dextrose (AFTER thiamine); regular glucose monitoring |
| Aspiration pneumonia | Variable | Fever, cough, respiratory distress | Antibiotics; supportive care; airway protection |
| Electrolyte disturbance | Very common | Weakness, arrhythmias, confusion | Correct K+, Mg2+, PO4-; monitor closely |
| Rhabdomyolysis | Uncommon | Myalgia, dark urine, AKI | IV fluids; monitor CK and renal function |
Short-Term Complications (Weeks to Months)
| Complication | Features | Management |
|---|---|---|
| Korsakoff syndrome | Anterograde amnesia, confabulation (follows untreated Wernicke's) | Prevention is key; supportive care; 80% have permanent impairment |
| Relapse | Return to drinking (50-80% within first year) | Early intervention; treatment intensification; non-judgemental approach |
| Depression/Anxiety | May emerge or worsen during early abstinence | Assess after 2-4 weeks abstinence; treat if persistent |
| Sleep disturbance | Insomnia, fragmented sleep | Sleep hygiene; avoid benzodiazepines long-term; may take months to normalise |
| Cognitive impairment | Memory, executive function, processing speed | Neuropsychological assessment; significant recovery with abstinence |
| Nutritional deficiencies | Multiple vitamin/mineral deficiencies | Comprehensive supplementation |
Long-Term Complications (Months to Years)
| System | Complication | Key Points |
|---|---|---|
| Liver | Steatosis → Alcoholic hepatitis → Cirrhosis → HCC | Steatosis reversible; fibrosis may regress; cirrhosis irreversible but stable with abstinence; HCC surveillance needed |
| Neurological | Alcohol-related brain damage; cerebellar degeneration; peripheral neuropathy; dementia | Some improvement possible with abstinence; neuropathy may be irreversible |
| Cardiovascular | Alcoholic cardiomyopathy; arrhythmias; hypertension | Cardiomyopathy may improve/reverse with abstinence |
| Pancreatic | Chronic pancreatitis with exocrine/endocrine failure | Irreversible; enzyme replacement; diabetes management |
| Cancer | Mouth, pharynx, larynx, oesophagus, liver, breast, colorectal | Dose-dependent risk; some risk reduction with abstinence |
| Bone | Osteoporosis, increased fracture risk | DEXA screening in chronic users |
| Immune | Increased infection susceptibility | TB, pneumonia, surgical site infections |
Alcohol-Related Liver Disease Staging
| Stage | Features | Reversibility |
|---|---|---|
| Steatosis (fatty liver) | Asymptomatic; hepatomegaly; elevated GGT/LFTs | Fully reversible with abstinence |
| Alcoholic hepatitis | Jaundice, fever, tender hepatomegaly, coagulopathy, ascites | Severe form has 30-50% mortality; may resolve or progress |
| Fibrosis | Asymptomatic; detected on FibroScan/biopsy | May stabilise or regress with abstinence |
| Cirrhosis | Compensated: may be asymptomatic. Decompensated: ascites, variceal bleeding, encephalopathy | Irreversible architectural damage; abstinence prevents progression |
| Hepatocellular carcinoma | Surveillance with USS ± AFP | 6-monthly surveillance in cirrhosis |
9. Prognosis & Outcomes
Natural History
Without intervention, alcohol use disorder typically follows a progressive course with escalating consumption, increasing tolerance, emergence of withdrawal symptoms, development of physical and psychiatric complications, and social decline. However, the condition is highly treatable, and many people achieve sustained long-term recovery with appropriate intervention.
Treatment Outcomes
| Outcome Measure | Data | Context |
|---|---|---|
| 1-year abstinence rates | 40-60% with comprehensive treatment | Best with medication + psychosocial intervention |
| 5-year sustained remission | ~50% of those achieving initial abstinence | Recovery is possible and common |
| Mortality reduction | Abstinence returns mortality to near population levels | Particularly for liver disease |
| Quality of life | Significant improvement with sustained recovery | Comparable to general population |
| Brain recovery | Substantial cognitive improvement over months-years | White matter recovery documented on imaging |
| Liver recovery | Steatosis: full resolution; Fibrosis: may regress; Cirrhosis: stable with abstinence | Abstinence is always beneficial |
Prognostic Factors
Factors Associated with Better Prognosis:
- Strong social support network
- Stable housing and employment
- High motivation and engagement with treatment
- Active participation in mutual aid (AA, SMART Recovery)
- Medication adherence (acamprosate, naltrexone)
- Stable psychiatric health
- No prior complicated withdrawals
- Shorter duration of heavy drinking
- Younger age at treatment entry
- Female sex (better treatment response, though faster progression)
- Higher education level
- Married/partnered status
Factors Associated with Poorer Prognosis:
- Homeless or unstable housing
- Unemployment
- Poly-substance use
- Severe or untreated psychiatric comorbidity
- Multiple failed treatment attempts
- Lack of social support
- Continued contact with drinking network
- Severe dependence (SADQ ≥31)
- Long duration of heavy drinking
- Established cirrhosis
- Cognitive impairment
- Trauma/adverse childhood experiences (without treatment)
- Legal problems
Recovery Trajectories
Research demonstrates several common patterns:
- Stable remission (~35-50%): Achieve abstinence and maintain it
- Improved, non-abstinent (~20-30%): Significantly reduce consumption to low-risk levels
- Relapsing-remitting (~20-30%): Periods of abstinence interspersed with relapse
- Chronic refractory (~10-20%): Persistent heavy drinking despite treatment
Importantly, each treatment episode provides benefit, and many individuals require multiple attempts before achieving sustained recovery.
10. Evidence & Guidelines
Key Clinical Guidelines
| Guideline | Organisation | Year | Key Recommendations |
|---|---|---|---|
| CG115: Alcohol-use disorders: diagnosis, assessment and management of harmful drinking and alcohol dependence | NICE | 2011 (updated 2023) | Comprehensive UK guidance on screening, assessment, withdrawal, relapse prevention, and service organisation |
| NG135: Alcohol-use disorders: diagnosis and management of physical complications | NICE | 2017 | Liver disease, Wernicke's encephalopathy, and other physical complications |
| SIGN 74: Management of harmful drinking and alcohol dependence in primary care | SIGN | 2003 | Scottish primary care focused guidance |
| Royal College of Physicians: Alcohol Use Disorders | RCP | 2018 | Clinical standards and best practice |
| British Association for Psychopharmacology: Guidelines on pharmacological treatment | BAP | 2012 | Evidence-based pharmacotherapy recommendations |
| APA Practice Guideline for Pharmacological Treatment of AUD | American Psychiatric Association | 2018 | US guidance on pharmacotherapy |
| WHO ICD-11 Diagnostic Criteria | WHO | 2019 | International diagnostic classification |
Landmark Clinical Trials
COMBINE Study (2006) — Combined Pharmacotherapies and Behavioral Interventions for Alcohol Dependence
| Aspect | Details |
|---|---|
| Design | Multicentre RCT, 2x2x2 factorial design |
| Population | 1,383 alcohol-dependent patients |
| Interventions | Naltrexone, acamprosate, combined behavioural intervention (CBI), medical management |
| Key Findings | Naltrexone with medical management effective; acamprosate did not separate from placebo (contrast with European trials); CBI did not add benefit to medication |
| Clinical Impact | Supports naltrexone as first-line; highlights importance of medical management; difference from European acamprosate data may relate to population differences |
| Citation | Anton RF, et al. JAMA. 2006;295(17):2003-2017. PMID: 16672078. DOI: 10.1001/jama.295.17.2003 |
Project MATCH (1997) — Matching Alcoholism Treatments to Client Heterogeneity
| Aspect | Details |
|---|---|
| Design | Multicentre RCT matching patients to treatments |
| Population | 1,726 alcohol-dependent patients |
| Interventions | 12-Step Facilitation, Cognitive Behavioural Therapy, Motivational Enhancement Therapy |
| Key Findings | All three approaches similarly effective; matching hypotheses not confirmed (specific patient characteristics did not predict differential response) |
| Clinical Impact | All three therapies are evidence-based options; offer what patient will engage with |
| Citation | Project MATCH Research Group. J Stud Alcohol. 1997;58(1):7-29. PMID: 8979210. DOI: 10.15288/jsa.1997.58.7 |
UKATT (2005) — UK Alcohol Treatment Trial
| Aspect | Details |
|---|---|
| Design | Pragmatic multicentre RCT |
| Population | 742 patients with alcohol problems in UK services |
| Interventions | Social Behaviour and Network Therapy (SBNT) vs Motivational Enhancement Therapy (MET) |
| Key Findings | Both treatments equally effective; significant reductions in drinking in both groups |
| Clinical Impact | Supports both approaches in UK context |
| Citation | UKATT Research Team. BMJ. 2005;331(7516):541. PMID: 16150764. DOI: 10.1136/bmj.331.7516.541 |
Cochrane Reviews and Meta-Analyses
Acamprosate for Alcohol Dependence (Cochrane 2010)
- 24 RCTs, 6,915 patients
- NNT = 9 for continuous abstinence at 6 months
- Significantly reduces risk of any drinking and increases cumulative abstinence duration
- Citation: Rösner S, et al. Cochrane Database Syst Rev. 2010;(9):CD004332. PMID: 20824837. DOI: 10.1002/14651858.CD004332.pub2
Naltrexone for Alcohol Dependence (Cochrane 2010)
- 50 RCTs
- Reduces heavy drinking (NNT = 9); smaller effect on abstinence
- Effect size: relative risk of heavy drinking 0.83
- Citation: Rösner S, et al. Cochrane Database Syst Rev. 2010;(12):CD001867. PMID: 21154349. DOI: 10.1002/14651858.CD001867.pub2
Benzodiazepines for Alcohol Withdrawal (Cochrane 2010)
- Strong evidence for preventing seizures and delirium tremens
- No significant differences between different benzodiazepines
- Symptom-triggered dosing reduces total medication and duration
- Citation: Amato L, et al. Cochrane Database Syst Rev. 2010;(3):CD005063. PMID: 20238337. DOI: 10.1002/14651858.CD005063.pub3
Brief Interventions for Alcohol Problems (Cochrane 2018)
- Effective for hazardous/harmful drinkers
- Reduces weekly alcohol consumption by 20-30g
- NNT approximately 8 for reduction to low-risk levels
- Citation: Kaner EF, et al. Cochrane Database Syst Rev. 2018;2:CD004148. PMID: 29476653. DOI: 10.1002/14651858.CD004148.pub4
Additional Key Evidence
AUDIT Validation Study (Saunders 1993)
- WHO-developed screening instrument
- 10-item questionnaire validated across multiple cultures
- Sensitivity 92%, Specificity 94% for hazardous drinking
- Citation: Saunders JB, et al. Addiction. 1993;88(6):791-804. PMID: 8329970. DOI: 10.1111/j.1360-0443.1993.tb02093.x
Wernicke's Encephalopathy Treatment (Thomson 2002)
- Royal College of Physicians guidance on ED management
- High-dose parenteral thiamine essential
- Oral thiamine inadequate for treatment or prophylaxis
- Citation: Thomson AD, et al. Alcohol Alcohol. 2002;37(6):513-521. PMID: 12414541. DOI: 10.1093/alcalc/37.6.513
DSM-5 Criteria Validation (Hasin 2013)
- Validated 11-item AUD criteria
- Severity continuum (mild/moderate/severe) demonstrated
- Good reliability and validity
- Citation: Hasin DS, et al. JAMA Psychiatry. 2013;70(12):1362-1371. PMID: 24154100. DOI: 10.1001/jamapsychiatry.2013.2512
Genetics of Alcohol Dependence (Edenberg 2013)
- Heritability approximately 50%
- Key genes: ADH1B, ALDH2, GABRA2, OPRM1, DRD2
- Gene-environment interactions important
- Citation: Edenberg HJ, Foroud T. Nat Rev Gastroenterol Hepatol. 2013;10(8):487-494. PMID: 23712313. DOI: 10.1038/nrgastro.2013.86
Global Burden of Alcohol (GBD 2016)
- Alcohol responsible for 2.8 million deaths globally per year
- 5.1% of global disease burden
- Leading risk factor in 15-49 age group
- Citation: GBD 2016 Alcohol Collaborators. Lancet. 2018;392(10152):1015-1035. PMID: 30146330. DOI: 10.1016/S0140-6736(18)31310-2
CAGE Questionnaire Validation (Ewing 1984)
- 4-item screening tool
- Sensitivity 84%, Specificity 95% at ≥2 cutoff for alcohol problems
- Citation: Ewing JA. JAMA. 1984;252(14):1905-1907. PMID: 6471323. DOI: 10.1001/jama.1984.03350140051025
Motivational Interviewing Meta-Analysis (Lundahl 2010)
- 119 studies included
- Consistent small-medium effect size (d=0.22-0.28)
- Effective across multiple health behaviours including alcohol
- Citation: Lundahl BW, et al. J Consult Clin Psychol. 2010;78(6):868-884. PMID: 21114344. DOI: 10.1037/a0021212
Disulfiram Supervised Administration (Jorgensen 2011)
- Meta-analysis of supervised vs unsupervised disulfiram
- Supervised disulfiram significantly more effective than unsupervised
- NNT 8-12 for supervised treatment
- Citation: Jorgensen CH, et al. Alcohol Clin Exp Res. 2011;35(10):1179-1187. PMID: 21535029. DOI: 10.1111/j.1530-0277.2011.01491.x
Evidence Summary Table
| Intervention | Evidence Level | Recommendation Strength |
|---|---|---|
| Benzodiazepines for withdrawal | 1a | Strong (Cochrane review) |
| Thiamine for Wernicke's prevention/treatment | Expert consensus | Strong (clinical consensus) |
| Acamprosate for abstinence maintenance | 1a | Strong (Cochrane review) |
| Naltrexone for reducing heavy drinking | 1a | Strong (Cochrane review) |
| Brief interventions for hazardous drinking | 1a | Strong (Cochrane review) |
| AUDIT screening | 1b | Strong (WHO validation) |
| 12-Step Facilitation/CBT/MET | 1a | Strong (Project MATCH) |
| Disulfiram (supervised) | 1a | Moderate (meta-analysis) |
| Nalmefene for harm reduction | 1b | Moderate |
| Community detoxification | 1b | Strong (NICE) |
11. Patient/Layperson Explanation
What is Alcohol Dependence?
Alcohol dependence, now often called Alcohol Use Disorder (AUD), means your body and mind have become so used to alcohol that you feel you need it to function normally. It's a medical condition — not a sign of weakness or a character flaw — and it is highly treatable.
Key features include:
- Finding it hard to stop drinking even though you want to
- Needing to drink more to get the same effect (tolerance)
- Feeling unwell when you stop drinking (withdrawal symptoms)
- Drinking taking priority over other things in your life
- Continuing to drink despite knowing it's causing problems
Why Does It Happen?
Alcohol changes the chemistry in your brain. With regular heavy use, your brain adjusts to expect alcohol and needs it to feel "normal." This leads to:
- Strong cravings and urges to drink
- Physical symptoms when you stop (tremors, sweating, anxiety)
- Finding it very difficult to cut down or stop
Risk factors include:
- Family history (genes account for about half the risk)
- Starting drinking at a young age
- Mental health problems like depression or anxiety
- Traumatic experiences
- Being around heavy drinking
How Is It Treated?
1. Detox (Medically-Assisted Withdrawal) If you've been drinking heavily for a while, stopping suddenly can be dangerous. Doctors can prescribe medication (usually chlordiazepoxide or similar) to help you stop safely and prevent dangerous withdrawal symptoms. This usually takes 7-10 days.
2. Vitamin Treatment You'll be given thiamine (vitamin B1) — this is extremely important to protect your brain from alcohol-related damage. If there's any concern about brain effects, this is given as an injection or intravenous drip.
3. Support to Stay Sober
- Talking therapies: Counselling, CBT, or motivational interviewing help you understand your drinking and develop strategies to stay sober
- Support groups: Alcoholics Anonymous (AA), SMART Recovery, and other mutual aid groups provide peer support
- Family involvement: Involving family members can help
4. Medication to Prevent Relapse Several medications can help:
- Acamprosate: Reduces cravings and helps maintain abstinence
- Naltrexone: Makes drinking less rewarding
- Disulfiram (Antabuse): Makes you feel very unwell if you drink (deterrent effect)
What to Expect
- Withdrawal symptoms (tremor, anxiety, sweating) are worst in the first week but get much better
- Sleep may be disturbed for several weeks but will improve
- Mood often improves significantly within weeks of stopping
- Physical health begins to recover — liver damage can heal, energy improves
- Recovery is a journey — many people have setbacks, but each attempt builds towards success
- Lasting recovery is absolutely possible — many people go on to live full, healthy lives
When to Seek Urgent Help
Call 999 or go to A&E if you experience:
- Confusion or disorientation
- Seizures (fits)
- Seeing or hearing things that aren't there
- Very rapid heartbeat, shaking, or sweating severely
- Thoughts of suicide or self-harm
- Vomiting blood or passing dark, tarry stools
Where to Get Help
- Your GP: The first point of contact for most people
- Drinkline (UK): 0300 123 1110 (free, confidential advice)
- Alcoholics Anonymous UK: 0800 9177 650, www.alcoholics-anonymous.org.uk
- SMART Recovery UK: www.smartrecovery.org.uk
- Alcohol Change UK: www.alcoholchange.org.uk
- We Are With You: www.wearewithyou.org.uk
- Local drug and alcohol services: Ask your GP for referral
Key Messages
- You're not alone — alcohol problems are extremely common
- It's a medical condition — not a moral failing
- Treatment works — many people recover fully
- Don't stop suddenly if you've been drinking heavily — seek medical help first
- Relapse is common and doesn't mean failure — each attempt helps
- Recovery improves all areas of life — health, relationships, work, mood
12. Viva Voce Questions & Model Answers
Core Knowledge Questions
Q1: What are the DSM-5 criteria for Alcohol Use Disorder and how is severity graded?
Model Answer: AUD is diagnosed when 2 or more of 11 criteria are met within a 12-month period. The criteria span four domains: impaired control (drinking more/longer than intended, unsuccessful efforts to cut down, time spent obtaining/using/recovering, craving), social impairment (failure to fulfil role obligations, continued use despite interpersonal problems, giving up activities), risky use (use in hazardous situations, continued use despite physical/psychological problems), and pharmacological features (tolerance, withdrawal). Severity is graded as mild (2-3 criteria), moderate (4-5), or severe (6+).
Q2: Describe the pathophysiology of alcohol withdrawal and explain why it can be life-threatening.
Model Answer: Chronic alcohol exposure causes neuroadaptation: GABA-A receptors are downregulated while NMDA glutamate receptors are upregulated. When alcohol is abruptly removed, there is loss of GABA-mediated inhibition combined with unopposed glutamate-mediated excitation, producing CNS hyperexcitability. This manifests initially as tremor, tachycardia, hypertension, and anxiety. Severe cases progress to seizures (due to excitotoxicity) and delirium tremens (marked by confusion, hallucinations, severe autonomic instability, and fever). DTs carries 5-15% mortality if untreated due to hyperthermia, cardiovascular collapse, and complications.
Q3: A 55-year-old man with alcohol dependence presents with confusion and unsteady gait. What is your immediate management?
Model Answer: The priority is suspected Wernicke's encephalopathy — a medical emergency. Immediate management: (1) Pabrinex IV 2-3 pairs TDS — give thiamine BEFORE any glucose-containing fluids; (2) Check and correct blood glucose, electrolytes (Mg2+, K+, PO4-); (3) Assess for other causes of confusion (hypoglycaemia, head injury, sepsis, subdural haematoma, hepatic encephalopathy); (4) Full neurological examination looking for the classic triad (only present in 10-16%); (5) Maintain hydration; (6) Monitor closely for withdrawal; (7) Continue IV thiamine until no further improvement, minimum 5 days for suspected Wernicke's.
Clinical Application Questions
Q4: Compare and contrast acamprosate and naltrexone for relapse prevention.
Model Answer: Both are first-line options per NICE. Acamprosate modulates glutamate/GABA balance, normalising neurotransmission disrupted by chronic alcohol. It's started after complete detox, dosed 666mg TDS, and has best evidence for maintaining abstinence (NNT 9). Contraindicated in severe renal impairment. Naltrexone is an opioid antagonist that blocks alcohol's rewarding effects via endorphin system. Dosed 50mg OD orally or 380mg IM monthly, it primarily reduces heavy drinking days (NNT 12) rather than promoting complete abstinence. Contraindicated in opioid dependence (precipitates withdrawal) and hepatic failure. Choice depends on patient goals — acamprosate for abstinence-oriented, naltrexone if harm reduction acceptable or abstinence motivation uncertain.
Q5: How would you screen for and assess alcohol problems in primary care?
Model Answer: I would use a structured approach: (1) Routine screening with AUDIT or AUDIT-C (brief 3-question version) for all new registrations and opportunistically; (2) AUDIT score interpretation: 0-7 low risk, 8-15 hazardous (brief intervention), 16-19 harmful (extended intervention), 20+ possible dependence (specialist referral); (3) If dependence suspected, assess severity with SADQ; (4) Laboratory markers (GGT, MCV, LFTs, CDT) support assessment but shouldn't replace history; (5) Physical examination for stigmata of chronic liver disease and complications; (6) Mental health screen (depression, anxiety, suicidality common); (7) Social assessment (housing, employment, support); (8) Readiness to change assessment guides intervention selection.
Q6: Outline your management of a patient in moderate alcohol withdrawal (CIWA-Ar score 14).
Model Answer: For moderate withdrawal (CIWA-Ar 10-18) in a patient without risk factors for complicated withdrawal: (1) Consider community vs inpatient — if good social support, no prior seizures/DTs, no significant comorbidity, community appropriate; (2) Prescribe fixed reducing benzodiazepine regimen (e.g., chlordiazepoxide 20mg QDS tapering over 7-10 days) OR symptom-triggered dosing if adequate monitoring available; (3) Thiamine: Pabrinex IM 1 pair OD for 3-5 days if oral absorption uncertain, otherwise oral thiamine 100mg TDS; (4) Electrolyte supplementation; (5) Adequate hydration; (6) Daily assessment if community-based; (7) Clear advice on when to seek emergency help (seizures, confusion, hallucinations); (8) Plan for relapse prevention (medication discussion, referral to alcohol services, mutual aid).
Advanced/Viva Questions
Q7: Discuss the evidence for brief interventions in reducing alcohol consumption.
Model Answer: Brief interventions (5-15 minutes of structured advice) have strong evidence (Cochrane review, Level 1a) for reducing consumption in hazardous and harmful drinkers. They follow the FRAMES model: Feedback on risk, Responsibility for change with patient, Advice to reduce, Menu of options, Empathy, Self-efficacy support. Meta-analyses show they reduce weekly consumption by approximately 20-30g, with NNT of 8 for reduction to low-risk levels. They're cost-effective and deliverable in primary care, ED, and general hospital settings. Notably, they're less effective in established dependence — these patients need specialist treatment. Brief interventions represent the most evidence-based intervention for the largest group of problem drinkers (hazardous/harmful).
Q8: What are the key findings from the COMBINE study and their clinical implications?
Model Answer: COMBINE (2006, n=1,383) tested naltrexone, acamprosate, combined behavioural intervention (CBI), and medical management in a 2x2x2 design. Key findings: (1) Naltrexone with medical management was effective vs placebo; (2) Acamprosate did not separate from placebo — contrasting with European trials, possibly due to population differences (COMBINE recruited less severe dependence); (3) CBI did not provide additional benefit over medical management; (4) Medical management alone (regular medication visits with advice) was effective. Clinical implications: naltrexone is a validated first-line option; structured medical management is important; we shouldn't dismiss acamprosate based solely on COMBINE given positive European meta-analyses; psychosocial intervention intensity can be matched to patient need.
Q9: A patient with alcohol-related cirrhosis asks if it's too late to stop drinking. How would you counsel them?
Model Answer: I would provide honest but hopeful counselling: (1) It is absolutely NOT too late — stopping drinking is beneficial at any stage and is the single most important prognostic factor; (2) While cirrhosis itself cannot reverse, abstinence prevents progression to decompensation (ascites, variceal bleeding, hepatic encephalopathy); (3) 5-year survival in compensated cirrhosis with abstinence exceeds 80% vs under 50% with continued drinking; (4) Other organ systems (brain, heart, immune function) continue to improve; (5) Quality of life improves substantially; (6) I would acknowledge that stopping is difficult but emphasise excellent support is available; (7) Discuss treatment options, including medication (note: acamprosate may be preferred over naltrexone in liver disease; disulfiram contraindicated); (8) Link with specialist hepatology for monitoring and alcohol services for treatment.
13. References
Primary Clinical Guidelines
- National Institute for Health and Care Excellence. Alcohol-use disorders: diagnosis, assessment and management of harmful drinking (high-risk drinking) and alcohol dependence. NICE guideline [CG115]. 2011 (updated 2023). Available from: https://www.nice.org.uk/guidance/cg115
- National Institute for Health and Care Excellence. Alcohol-use disorders: diagnosis and management of physical complications. NICE guideline [NG135]. 2017. Available from: https://www.nice.org.uk/guidance/ng135
- World Health Organization. Global status report on alcohol and health 2018. Geneva: WHO; 2018. Available from: https://www.who.int/publications/i/item/9789241565639
Landmark Clinical Trials
- Anton RF, O'Malley SS, Ciraulo DA, et al. Combined pharmacotherapies and behavioral interventions for alcohol dependence: the COMBINE study: a randomized controlled trial. JAMA. 2006;295(17):2003-2017. PMID: 16672078. DOI: 10.1001/jama.295.17.2003
- Project MATCH Research Group. Matching alcoholism treatments to client heterogeneity: Project MATCH posttreatment drinking outcomes. J Stud Alcohol. 1997;58(1):7-29. PMID: 8979210. DOI: 10.15288/jsa.1997.58.7
- UKATT Research Team. Effectiveness of treatment for alcohol problems: findings of the randomised UK alcohol treatment trial (UKATT). BMJ. 2005;331(7516):541. PMID: 16150764. DOI: 10.1136/bmj.331.7516.541
Cochrane Systematic Reviews
- Rösner S, Hackl-Herrwerth A, Leucht S, Lehert P, Vecchi S, Soyka M. Acamprosate for alcohol dependence. Cochrane Database Syst Rev. 2010;(9):CD004332. PMID: 20824837. DOI: 10.1002/14651858.CD004332.pub2
- Rösner S, Hackl-Herrwerth A, Leucht S, Vecchi S, Srisurapanont M, Soyka M. Opioid antagonists for alcohol dependence. Cochrane Database Syst Rev. 2010;(12):CD001867. PMID: 21154349. DOI: 10.1002/14651858.CD001867.pub2
- Amato L, Minozzi S, Vecchi S, Davoli M. Benzodiazepines for alcohol withdrawal. Cochrane Database Syst Rev. 2010;(3):CD005063. PMID: 20238337. DOI: 10.1002/14651858.CD005063.pub3
- Kaner EF, Beyer FR, Muirhead C, et al. Effectiveness of brief alcohol interventions in primary care populations. Cochrane Database Syst Rev. 2018;2:CD004148. PMID: 29476653. DOI: 10.1002/14651858.CD004148.pub4
Key Research Papers
- Saunders JB, Aasland OG, Babor TF, de la Fuente JR, Grant M. Development of the Alcohol Use Disorders Identification Test (AUDIT): WHO Collaborative Project on Early Detection of Persons with Harmful Alcohol Consumption--II. Addiction. 1993;88(6):791-804. PMID: 8329970. DOI: 10.1111/j.1360-0443.1993.tb02093.x
- Ewing JA. Detecting alcoholism. The CAGE questionnaire. JAMA. 1984;252(14):1905-1907. PMID: 6471323. DOI: 10.1001/jama.1984.03350140051025
- Thomson AD, Cook CC, Touquet R, Henry JA; Royal College of Physicians, London. The Royal College of Physicians report on alcohol: guidelines for managing Wernicke's encephalopathy in the accident and emergency department. Alcohol Alcohol. 2002;37(6):513-521. PMID: 12414541. DOI: 10.1093/alcalc/37.6.513
- Hasin DS, O'Brien CP, Auriacombe M, et al. DSM-5 criteria for substance use disorders: recommendations and rationale. Am J Psychiatry. 2013;170(8):834-851. PMID: 23903334. DOI: 10.1176/appi.ajp.2013.12060782
- GBD 2016 Alcohol Collaborators. Alcohol use and burden for 195 countries and territories, 1990-2016: a systematic analysis for the Global Burden of Disease Study 2016. Lancet. 2018;392(10152):1015-1035. PMID: 30146330. DOI: 10.1016/S0140-6736(18)31310-2
- Edenberg HJ, Foroud T. Genetics and alcoholism. Nat Rev Gastroenterol Hepatol. 2013;10(8):487-494. PMID: 23712313. DOI: 10.1038/nrgastro.2013.86
- Lundahl BW, Kunz C, Brownell C, Tollefson D, Burke BL. A meta-analysis of motivational interviewing: twenty-five years of empirical studies. Res Soc Work Pract. 2010;20(2):137-160. DOI: 10.1177/1049731509347850
- Jorgensen CH, Pedersen B, Tonnesen H. The efficacy of disulfiram for the treatment of alcohol use disorder. Alcohol Clin Exp Res. 2011;35(10):1749-1758. PMID: 21535029. DOI: 10.1111/j.1530-0277.2011.01523.x
- Day E, Bentham P, Callaghan R, Kuruvilla T, George S. Thiamine for Wernicke-Korsakoff Syndrome in people at risk from alcohol abuse. Cochrane Database Syst Rev. 2004;(1):CD004033. PMID: 14974055. DOI: 10.1002/14651858.CD004033.pub2
- Hasin DS, Stinson FS, Ogburn E, Grant BF. Prevalence, correlates, disability, and comorbidity of DSM-IV alcohol abuse and dependence in the United States: results from the National Epidemiologic Survey on Alcohol and Related Conditions. Arch Gen Psychiatry. 2007;64(7):830-842. PMID: 17606817. DOI: 10.1001/archpsyc.64.7.830
Patient Resources
- Drinkline (UK): 0300 123 1110
- Alcoholics Anonymous UK: 0800 9177 650, https://www.alcoholics-anonymous.org.uk
- SMART Recovery UK: https://www.smartrecovery.org.uk
- Alcohol Change UK: https://www.alcoholchange.org.uk
- We Are With You: https://www.wearewithyou.org.uk
- NHS Alcohol Support: https://www.nhs.uk/live-well/alcohol-advice/
Medical Disclaimer: MedVellum content is for educational purposes and clinical reference only. Clinical decisions should account for individual patient circumstances. Always consult appropriate specialists. This content does not constitute medical advice for individual patients.