Alcohol Withdrawal Syndrome
Comprehensive evidence-based guide to alcohol withdrawal syndrome: pathophysiology, CIWA-Ar scoring, benzodiazepine protocols, delirium tremens management
What matters first
Comprehensive evidence-based guide to alcohol withdrawal syndrome: pathophysiology, CIWA-Ar scoring, benzodiazepine protocols, delirium tremens management
Check the red flags, emergency triggers, and escalation points before using the deep-dive material.
10 Jan 2025
Generated educational material; verify before clinical use.
Visible references section
See the concept before reading it
Study the key anatomy, imaging, and decision pathways as full teaching plates.
Clinical board
A visual summary of the highest-yield teaching signals on this page.
Exam focus
Current exam surfaces linked to this topic.
- MRCP, FRCEM, USMLE
Content status and exam context
This page is AI-generated educational content. It may contain errors or omissions and is not a substitute for current guidelines, local protocols, senior clinical judgement, or professional medical advice.
MedVellum does not claim an individual clinician reviewer, board certification, or professional credential for this page unless a future version names a real, verifiable reviewer.
Clinical explanation and evidence
Alcohol Withdrawal Syndrome
Quick Reference
Critical Alerts
- Give thiamine BEFORE glucose: Prevents Wernicke's encephalopathy [1,2]
- Delirium tremens mortality: 5-15% if untreated; less than 1-4% with appropriate treatment [3,4]
- Benzodiazepines are first-line: Prevent seizures and progression to DTs [5,6]
- Phenobarbital for refractory cases: Evidence supports early adjunctive use [7,8]
- Cannot reliably predict DTs: Even mild withdrawal can progress unpredictably [9]
- Hyperadrenergic state is life-threatening: Risk of tachyarrhythmias, MI, aspiration, cardiovascular collapse [3,4]
- Withdrawal occurs at ANY blood alcohol: Can develop even with elevated BAL [10]
Withdrawal Timeline
| Timeframe | Clinical Features | Risk/Severity |
|---|---|---|
| 6-12 hours | Minor withdrawal: tremor, anxiety, tachycardia, diaphoresis, nausea | Low-moderate |
| 12-24 hours | Peak minor symptoms, alcoholic hallucinosis may begin | Moderate |
| 12-48 hours | Withdrawal seizures (peak 24h): generalized tonic-clonic | High |
| 48-96 hours | Delirium tremens (peak 72h): delirium, severe autonomic instability | Life-threatening |
| 5-7 days | Gradual resolution with treatment | Variable |
Key Diagnostics
| Test | Finding | Significance |
|---|---|---|
| Vital signs | Tachycardia >100, hypertension, fever | Autonomic hyperactivity; fever >38.3°C suggests DTs or infection |
| Fingerstick glucose | Normal or low | Rule out hypoglycemia; give thiamine before dextrose |
| CIWA-Ar score | 0-67 scale | less than 10 mild, 10-19 moderate, ≥20 severe [11] |
| Electrolytes | Hypokalemia, hypomagnesemia, hypophosphatemia | Common; require replacement [12] |
| Blood alcohol level | Variable (can be elevated or zero) | Withdrawal can occur at high BAL [10] |
| LFTs, INR | Elevated AST/ALT, prolonged INR | Hepatic dysfunction; INR >1.5 suggests coagulopathy |
| Ammonia | If encephalopathy suspected | Differentiate hepatic from alcohol withdrawal |
Emergency Treatments
| Condition | Treatment | Dose | Evidence Level |
|---|---|---|---|
| Thiamine (FIRST) | IV Thiamine | 500mg IV over 30 min TDS × 3-5 days (Pabrinex) | IA [1,2] |
| Mild withdrawal (CIWA 10-19) | Diazepam or Lorazepam | 10-20mg PO/IV q1-4h PRN | IA [5,6] |
| Severe withdrawal/DTs (CIWA ≥20) | Diazepam boluses | 10-20mg IV q10-15min until controlled | IA [4,6] |
| Refractory withdrawal | Phenobarbital adjunct | 130-260mg IV q15-30min (max 10mg/kg) | IB [7,8] |
| Withdrawal seizures | Benzodiazepines (NOT phenytoin) | Lorazepam 4mg IV or Diazepam 10mg IV | IA [13] |
| Electrolyte replacement | Magnesium sulfate | 2-4g IV if Mg less than 0.75 mmol/L | IIA [12] |
| Potassium chloride | 10-40 mEq/h based on level | IIA [12] |
Definition
Overview
Alcohol withdrawal syndrome (AWS) is a potentially fatal constellation of neuropsychiatric and autonomic symptoms occurring when individuals with physiological alcohol dependence abruptly reduce or cease alcohol intake. [3,4] The syndrome exists on a spectrum from minor withdrawal symptoms (tremor, anxiety, insomnia) to severe, life-threatening complications including withdrawal seizures and delirium tremens (DTs). [9]
Key Concept: AWS represents CNS hyperexcitability due to neuroadaptive changes from chronic GABA-A receptor downregulation and NMDA receptor upregulation, suddenly unopposed when alcohol (a CNS depressant) is removed. [14]
Historical Context
- Delirium tremens was first described in 1813 by Thomas Sutton
- Mortality from DTs exceeded 35% prior to modern critical care [3]
- Introduction of benzodiazepines in the 1960s revolutionized AWS management [5]
- CIWA-Ar (Clinical Institute Withdrawal Assessment for Alcohol, Revised) standardized in 1989 [11]
Classification of Alcohol Withdrawal Syndromes
Clinical Syndromes (can overlap):
| Syndrome | Timing Post-Cessation | Clinical Features | Sensorium |
|---|---|---|---|
| Minor withdrawal | 6-24 hours | Anxiety, tremor, insomnia, diaphoresis, tachycardia, nausea, headache | Clear |
| Alcoholic hallucinosis | 12-48 hours | Visual, auditory, or tactile hallucinations | Clear (insight retained) |
| Withdrawal seizures | 6-48 hours (peak 24h) | Generalized tonic-clonic seizures, usually brief, single or cluster | Post-ictal only |
| Delirium tremens | 48-96 hours (peak 72h) | Delirium, profound agitation, severe autonomic instability, hallucinations, fever | Impaired (confused, disoriented) |
CIWA-Ar Severity Stratification: [11]
| Score Range | Severity | Clinical Approach |
|---|---|---|
| 0-9 | Mild or absent | Supportive care; observe; pharmacotherapy usually unnecessary |
| 10-19 | Moderate | Pharmacotherapy indicated; symptom-triggered protocol |
| ≥20 | Severe | Aggressive treatment; high-dose benzodiazepines; consider ICU admission |
Epidemiology
Prevalence: [15,16]
- Alcohol use disorder (AUD): 14.5 million adults in US (5.8% of population aged ≥18)
- Global AUD prevalence: approximately 5-10% of adults
Withdrawal Incidence: [9,15]
- 50% of individuals with AUD experience withdrawal symptoms on cessation
- 3-5% of untreated patients develop withdrawal seizures
- 5-10% of hospitalized AWS patients progress to delirium tremens
- Without benzodiazepine prophylaxis, up to 35-50% of severe withdrawal progresses to DTs [4]
Mortality: [3,4]
- Delirium tremens: 5-15% mortality if untreated; 1-4% with modern treatment
- Leading causes of death: arrhythmias, respiratory failure, aspiration pneumonia, cardiovascular collapse
- Seizure-related mortality: less than 1% but can cause status epilepticus
Healthcare Burden: [15]
- >500,000 AWS-related hospitalizations annually in US
- Average length of stay: 3-7 days
- Readmission rate within 30 days: 10-20%
Etiology and Pathophysiology
Molecular Mechanism: [14]
Chronic Alcohol Exposure → Neuroadaptation:
- GABA-A receptors: Alcohol enhances inhibitory GABA transmission → chronic use causes receptor downregulation and altered subunit composition
- NMDA receptors: Alcohol inhibits excitatory glutamate transmission → chronic use causes receptor upregulation and increased sensitivity
- Other systems: Downregulation of dopamine, serotonin pathways; upregulation of norepinephrine
Alcohol Withdrawal → CNS Hyperexcitability:
- Loss of GABA enhancement: Reduced inhibitory tone
- Unopposed NMDA activity: Excessive excitatory glutamate signaling
- Sympathetic surge: Massive catecholamine release
- Net effect: Lowered seizure threshold, autonomic instability, CNS excitation
- Duration: Neurochemical readaptation requires 5-10 days
Kindling Phenomenon: [17]
- Each successive withdrawal episode is more severe than the previous
- Progressive neuronal sensitization with repeated alcohol detoxifications
- Lower seizure threshold with each subsequent withdrawal
- Emphasizes the critical importance of adequate initial treatment and relapse prevention
Risk Factors for Severe Withdrawal and Delirium Tremens
Strongest Predictors: [9,18]
- Previous delirium tremens (single strongest predictor)
- Previous withdrawal seizures
- Multiple prior detoxifications (kindling effect)
Strong Risk Factors: [9,18]
- Age >65 years
- Concurrent acute illness (infection, trauma, pancreatitis, GI bleeding, surgery)
- Heavy, prolonged drinking history (>10 years of daily heavy use)
- Elevated blood pressure or heart rate at presentation (HR >100, SBP >150)
- Thrombocytopenia (less than 100,000)
- Severe electrolyte abnormalities (K less than 3.0, Mg less than 0.5)
Moderate Risk Factors:
- Polysubstance use (especially concurrent benzodiazepine dependence)
- Structural brain lesions or history of head trauma
- Elevated blood alcohol level at presentation (>200 mg/dL)
- Poor nutritional status
Pathophysiology
Neurotransmitter Systems
GABA-A Receptor Adaptation: [14]
- Chronic alcohol → allosteric GABA-A enhancement → compensatory receptor downregulation
- Subunit composition changes (reduced α1, increased α4)
- Alcohol withdrawal → insufficient GABAergic inhibition → CNS hyperexcitability
- Clinical manifestation: Anxiety, agitation, seizures
NMDA Receptor Upregulation: [14]
- Chronic alcohol → glutamate antagonism at NMDA receptors → compensatory upregulation
- Alcohol withdrawal → unopposed glutamate excitotoxicity
- Clinical manifestation: Tremor, seizures, delirium, neurotoxicity
Autonomic Dysregulation: [3]
- Withdrawal → catecholamine surge (norepinephrine, epinephrine)
- Clinical manifestation: Tachycardia, hypertension, diaphoresis, hyperthermia, mydriasis
Dopaminergic Pathways:
- Altered mesolimbic dopamine signaling
- Clinical manifestation: Hallucinations (especially in alcoholic hallucinosis)
Metabolic Derangements
Thiamine Deficiency: [1,2]
- Chronic alcohol → impaired thiamine absorption, poor dietary intake, increased utilization
- Thiamine (vitamin B1) is essential cofactor for glucose metabolism (pyruvate dehydrogenase, α-ketoglutarate dehydrogenase, transketolase)
- Glucose administration without thiamine → depletes remaining thiamine stores → precipitates Wernicke's encephalopathy
- Wernicke's encephalopathy: Classic triad (present in only 10-16%): confusion, ataxia, ophthalmoplegia [1]
- Korsakoff's psychosis: Irreversible anterograde and retrograde amnesia if untreated [2]
Electrolyte Abnormalities: [12]
| Electrolyte | Mechanism | Prevalence | Clinical Impact |
|---|---|---|---|
| Hypokalemia | GI losses, renal wasting, respiratory alkalosis, poor intake | 30-50% | Arrhythmias, muscle weakness |
| Hypomagnesemia | Renal wasting, poor intake, increased cellular uptake | 30-80% | Refractory hypokalemia, seizures, arrhythmias |
| Hypophosphatemia | Refeeding, respiratory alkalosis, cellular shifts | 20-30% | Rhabdomyolysis, respiratory failure, hemolysis |
| Hyponatremia | SIADH, beer potomania, volume depletion | 10-20% | Confusion, seizures |
Hypoglycemia:
- Impaired hepatic gluconeogenesis
- Depleted glycogen stores
- Poor oral intake
- Can mimic or coexist with withdrawal symptoms
Metabolic Acidosis:
- Alcoholic ketoacidosis (β-hydroxybutyrate accumulation)
- Lactic acidosis (if concurrent sepsis, liver failure)
Cardiovascular Complications
Arrhythmias: [3]
- Catecholamine surge → atrial fibrillation, ventricular tachycardia
- "Holiday heart syndrome": AF in binge drinkers
- Electrolyte abnormalities exacerbate risk
Myocardial Infarction:
- Coronary vasoconstriction, increased myocardial oxygen demand
- Plaque rupture from catecholamine surge
Cardiomyopathy:
- Chronic alcoholic cardiomyopathy (dilated)
- Acute stress cardiomyopathy (Takotsubo) during severe withdrawal
Clinical Presentation
Symptom Timeline and Progression
6-12 Hours Post-Cessation: Minor Withdrawal
- Anxiety, restlessness, irritability
- Tremor (hands, tongue) - coarse, symmetric, exacerbated by intention
- Headache (frontal predominance)
- Diaphoresis (especially palmar, axillary)
- Anorexia, nausea, vomiting
- Insomnia
- Mild tachycardia (HR 90-110)
- Mild hypertension
12-24 Hours: Progression and Hallucinosis
- Worsening of minor symptoms
- Alcoholic hallucinosis (12-48h window):
- Visual hallucinations most common (shadows, small animals, insects)
- Auditory hallucinations (voices, sounds)
- Tactile hallucinations (formication - sensation of insects crawling)
- "Key differentiator: Patient retains insight; knows hallucinations are not real; sensorium is clear"
24-48 Hours: Withdrawal Seizures [13]
- Peak incidence: 24 hours post-cessation
- Characteristics:
- Generalized tonic-clonic seizures (>90% of cases)
- Usually brief (less than 2-3 minutes), self-limited
- Often occur as a single seizure or cluster of 2-6 seizures within 6 hours
- Post-ictal period typically short
- May be the first manifestation of withdrawal (patient presents after seizure)
- 3% progress to status epilepticus [13]
- Phenytoin is NOT effective for AWS seizures (seizures are due to acute neurochemical changes, not epilepsy) [13]
48-96 Hours (Peak 72h): Delirium Tremens [3,4]
- Incidence: 5-10% of hospitalized AWS patients
- Mortality: 1-4% with treatment; 5-15% without [3,4]
- Clinical features:
- "Delirium: Profound confusion, disorientation (time, place, person), fluctuating consciousness"
- "Severe agitation: Extreme psychomotor agitation, combativeness"
- "Autonomic instability: Severe tachycardia (HR >120), hypertension or labile BP, hyperthermia (38-40°C), profuse diaphoresis"
- Hallucinations: Visual (classically "small animals"
- zoopsia), tactile, auditory
- "Tremor: Coarse, generalized"
- Duration: 2-7 days (mean 3 days)
- Complications: Arrhythmias, MI, aspiration pneumonia, rhabdomyolysis, cardiovascular collapse
Physical Examination
Vital Signs: [3]
- Tachycardia: HR >100 (mild), >120 (severe DTs)
- Hypertension: SBP often 150-200 mmHg; DBP 90-110 mmHg
- Fever: Low-grade (37.5-38°C) common; high fever (>38.5°C) suggests DTs or concurrent infection
- Tachypnea: RR 20-30 (respiratory alkalosis)
General Inspection:
| Finding | Significance | Differential |
|---|---|---|
| Coarse tremor | Hallmark of withdrawal; worsens with intentional movement | Vs. essential tremor (alcohol suppresses), thyrotoxicosis |
| Diaphoresis | Autonomic hyperactivity | Hypoglycemia, sepsis, thyroid storm |
| Mydriasis | Catecholamine surge | Sympathomimetic toxicity, anticholinergic toxicity |
| Jaundice | Chronic liver disease, alcoholic hepatitis | Hepatic encephalopathy (different mental status) |
| Spider angiomata | Chronic liver disease | - |
| Palmar erythema | Chronic liver disease | - |
| Gynecomastia | Chronic alcohol use, liver disease | - |
| Cachexia | Poor nutrition, chronic disease | Malignancy |
Neurological Examination:
| Finding | Implication | Action |
|---|---|---|
| Asterixis (flapping tremor) | Hepatic encephalopathy | Check ammonia, LFTs |
| Nystagmus | Consider Wernicke's encephalopathy | High-dose IV thiamine immediately [1] |
| Ophthalmoplegia (CN VI palsy) | Wernicke's encephalopathy | High-dose IV thiamine immediately [1] |
| Ataxia | Wernicke's encephalopathy, chronic cerebellar damage | Thiamine; assess fall risk |
| Hyperreflexia | CNS hyperexcitability | Consistent with AWS |
| Focal deficits | Head trauma, stroke | CT head |
Mental Status Examination:
| Severity | Mental Status Findings |
|---|---|
| Mild | Alert, anxious, oriented × 3, appropriate responses |
| Moderate | Agitated, distractible, may have hallucinations but maintains orientation |
| Severe (DTs) | Delirious, disoriented to time/place/person, inattentive, fluctuating consciousness, not redirectable |
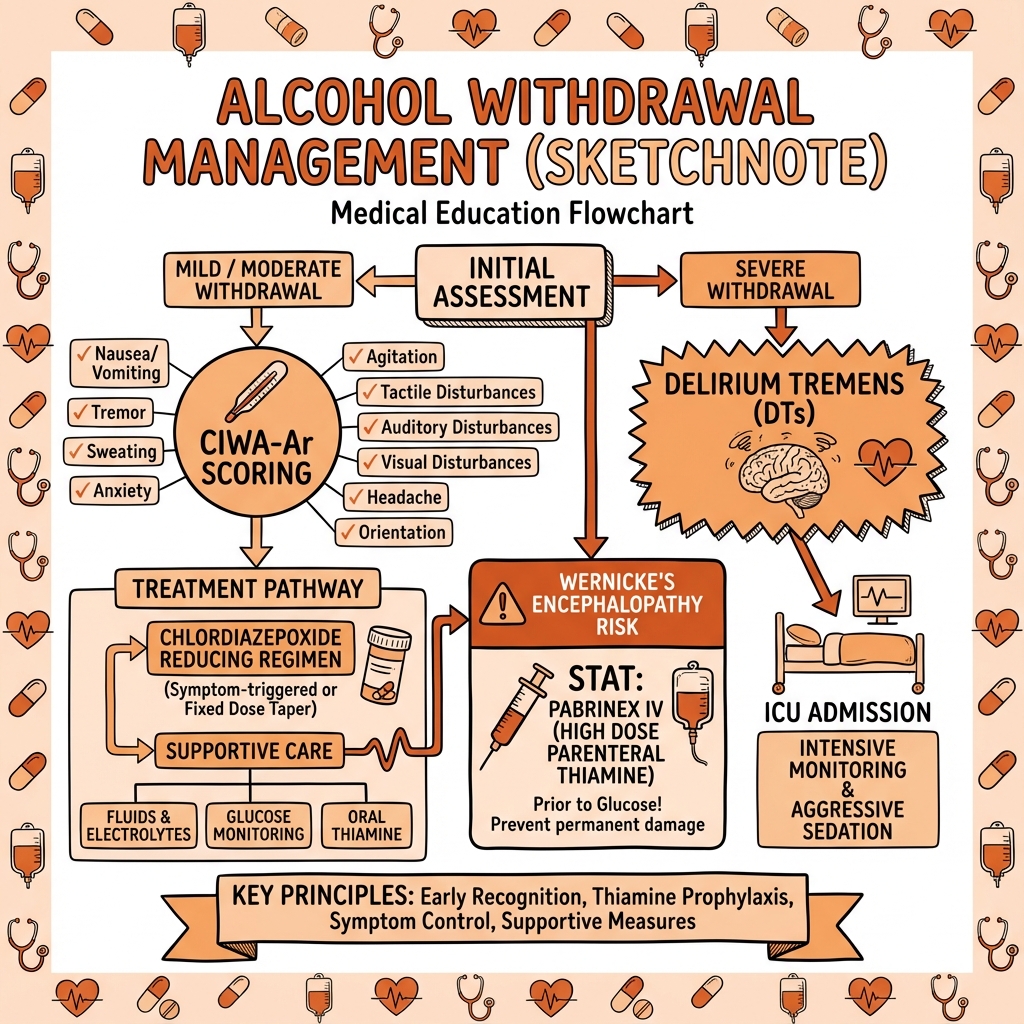
CIWA-Ar Assessment Tool [11]
The Clinical Institute Withdrawal Assessment for Alcohol, Revised (CIWA-Ar) is the gold-standard validated tool for AWS severity assessment and symptom-triggered treatment protocols.
10 Items (Maximum Score: 67):
| Item | Assessment Method | Score Range |
|---|---|---|
| 1. Nausea/Vomiting | "Do you feel sick to your stomach? Have you vomited?" | 0-7 |
| 2. Tremor | Arms extended, fingers spread | 0-7 |
| 3. Paroxysmal Sweats | Observation of visible moisture | 0-7 |
| 4. Anxiety | "Do you feel nervous?" Observe fidgeting | 0-7 |
| 5. Agitation | Observation of pacing, restlessness | 0-7 |
| 6. Tactile Disturbances | "Do you feel itching, pins and needles, burning, numbness, or bugs crawling on/under skin?" | 0-7 |
| 7. Auditory Disturbances | "Are you hearing sounds that disturb you? Are they harsh?" | 0-7 |
| 8. Visual Disturbances | "Does light appear too bright? Do you see anything disturbing?" | 0-7 |
| 9. Headache | "Does your head feel different? Full/tight?" | 0-7 |
| 10. Orientation | Person, place, date, time | 0-4 |
Scoring Interpretation:
- less than 10: Mild or absent withdrawal; supportive care; reassess q4-8h
- 10-19: Moderate withdrawal; initiate pharmacotherapy; reassess q1-2h
- ≥20: Severe withdrawal; aggressive treatment with high-dose benzodiazepines; consider ICU; reassess q30-60min
Limitations of CIWA-Ar: [11]
- Requires patient cooperation (cannot use in intubated, delirious, or uncooperative patients)
- Subjective components (patient self-report)
- Does not assess vital signs (must be recorded separately)
- Not validated in non-alcohol withdrawal or polysubstance use
Inter-Rater Reliability: [11]
- High inter-rater reliability when administered by trained staff (correlation coefficient 0.8-0.9)
- Requires 5-10 minutes to complete properly
- Training improves consistency and accuracy
- Nursing staff can administer effectively with proper education
Clinical Utility: [11,20]
- Sensitivity: 90-95% for detecting moderate-severe withdrawal when CIWA ≥10
- Specificity: 85-90% when staff properly trained
- Predictive Value: CIWA ≥15 on admission predicts complicated withdrawal (sensitivity 89%, specificity 74%)
- Response Assessment: Serial CIWA scores track treatment efficacy; failure to reduce by ≥5 points within 2 hours suggests need for dose escalation
Modified CIWA-Ar for Special Populations:
- Intubated patients: Use RASS (Richmond Agitation-Sedation Scale) + vital signs + fixed-dose protocol
- Cognitively impaired: Rely more on objective signs (tremor, sweating, vital signs) than subjective symptoms
- Language barrier: Use interpreter or focus on observable components only
Red Flags
Life-Threatening Conditions Requiring Immediate Intervention
| Finding | Concern | Immediate Action |
|---|---|---|
| Fever >38.3°C (101°F) | Delirium tremens vs. concurrent infection (sepsis, aspiration pneumonia, meningitis) | Aggressive benzodiazepines; pan-culture; broad-spectrum antibiotics; CT head/CXR; consider LP [3,4] |
| Severe agitation/combativeness | Delirium tremens; risk of self-harm, aspiration, rhabdomyolysis | High-dose IV benzodiazepines (diazepam 10-20mg q10-15min); physical restraints if necessary; ICU admission [4] |
| Seizure activity | Alcohol withdrawal seizures; risk of status epilepticus | IV lorazepam 4mg or diazepam 10mg; if recurrent, continuous benzodiazepines; if status, phenobarbital [13] |
| Altered consciousness/delirium | DTs, hepatic encephalopathy, intracranial hemorrhage, Wernicke's, hypoglycemia | Fingerstick glucose; thiamine 500mg IV stat; CT head; ammonia; aggressive benzodiazepines [1,3] |
| Severe autonomic instability | HR >120, SBP >180, temperature >39°C | Risk of arrhythmia, MI, cardiovascular collapse; ICU; aggressive benzodiazepines; dexmedetomidine adjunct [4,19] |
| Respiratory depression/apnea | Over-sedation with benzodiazepines/phenobarbital, aspiration | Airway management; consider intubation; flumazenil NOT recommended (can precipitate seizures) |
| Wernicke's triad | Confusion + ataxia + ophthalmoplegia (only 10-16% have full triad) | Thiamine 500mg IV TDS immediately; do NOT wait for confirmatory tests [1,2] |
| Hallucinations + delirium | Delirium tremens (vs. alcoholic hallucinosis which has clear sensorium) | Differentiate: hallucinosis has insight; DTs has delirium; DTs requires aggressive ICU treatment [3] |
High-Risk Features Mandating Inpatient Monitoring
- Previous delirium tremens or withdrawal seizures [9,18]
- CIWA score ≥15 on presentation
- Concurrent medical illness: infection, trauma, pancreatitis, GI bleed, surgery
- Age >65 years
- Multiple comorbidities (cardiac, respiratory, hepatic, renal disease)
- Blood alcohol level >200 mg/dL at presentation
- Severe electrolyte abnormalities (K less than 3.0, Mg less than 0.5, Na less than 130)
- Polysubstance use (especially concurrent benzodiazepine or opioid dependence)
- Social factors: homelessness, inability to follow outpatient protocol
Differential Diagnosis
Conditions That Mimic or Complicate Alcohol Withdrawal
| Condition | Overlapping Features | Distinguishing Features | Key Evaluation |
|---|---|---|---|
| Sepsis/Infection | Tachycardia, fever, altered mental status | Hypotension (late), elevated WBC, lactate, specific source | Cultures (blood, urine, sputum), CXR, procalcitonin |
| Meningitis/Encephalitis | Fever, confusion, agitation | Neck stiffness, photophobia, focal neurological signs | CT head, lumbar puncture |
| Hepatic Encephalopathy | Confusion, tremor (asterixis) | Asterixis (vs. tremor), elevated ammonia, liver disease stigmata | Ammonia (>100 μg/dL), LFTs, lactulose trial |
| Wernicke's Encephalopathy | Confusion, ataxia | Ophthalmoplegia (CN VI palsy), ataxia (only 10-16% have full triad) [1] | Clinical diagnosis; treat empirically; thiamine 500mg IV TDS [1,2] |
| Hypoglycemia | Tremor, diaphoresis, tachycardia, confusion | Rapid improvement with dextrose | Fingerstick glucose; glucose less than 3.0 mmol/L (54 mg/dL) |
| Head Trauma/ICH | Altered mental status, seizures | Trauma history, focal deficits, headache | CT head non-contrast |
| Thyrotoxicosis/Thyroid Storm | Tachycardia, tremor, diaphoresis, agitation, fever | Thyroid stigmata, AF, high-output heart failure | TSH (suppressed), free T4 (elevated) |
| Anticholinergic Toxicity | Agitation, hallucinations, tachycardia, fever | Dry skin/mucosa, mydriasis, urinary retention, absent bowel sounds | Toxicology screen, clinical diagnosis |
| Sympathomimetic Toxicity | Tachycardia, hypertension, agitation, diaphoresis, mydriasis | Drug use history (cocaine, amphetamines), severe hypertension | Urine drug screen, ECG (ischemia) |
| Benzodiazepine Withdrawal | Tremor, anxiety, seizures, autonomic hyperactivity | Usually less severe autonomic instability; history of benzo use | Clinical history |
| Status Epilepticus | Continuous seizures | Seizures do not stop spontaneously | EEG, treat per status protocol |
| Serotonin Syndrome | Agitation, hyperthermia, tremor | Clonus, hyperreflexia, rigidity; history of serotonergic meds | Clinical diagnosis; recent medication changes |
| Neuroleptic Malignant Syndrome | Hyperthermia, rigidity, altered mental status | Lead-pipe rigidity, elevated CK, antipsychotic use | CK (very elevated), medication history |
Concurrent Conditions Common in Alcohol Use Disorder: [15]
- Alcoholic hepatitis
- Cirrhosis with portal hypertension
- Pancreatitis (acute or chronic)
- GI bleeding (varices, gastritis, Mallory-Weiss tear)
- Cardiomyopathy
- Pneumonia (aspiration, community-acquired)
- Tuberculosis
- Malnutrition
- Peripheral neuropathy
Diagnostic Approach
Initial Evaluation
Immediate Assessment (First 15 Minutes):
- Airway, Breathing, Circulation: Assess and secure if compromised
- Vital signs: HR, BP, RR, temperature, oxygen saturation (continuous monitoring)
- Fingerstick glucose: Rule out hypoglycemia immediately
- Brief neurological examination: Mental status, focal deficits, signs of Wernicke's
- CIWA-Ar score: Baseline severity assessment [11]
- Alcohol history:
- Quantity and frequency of alcohol use
- Time of last drink
- Previous withdrawal episodes (seizures, DTs)
- Previous detoxification attempts and treatments
Laboratory Studies
Immediate Labs (All Patients):
| Test | Rationale | Expected Findings |
|---|---|---|
| CBC | Infection, thrombocytopenia, anemia | Macrocytic anemia, thrombocytopenia (less than 100K in 30%), leukocytosis (infection) |
| CMP | Electrolytes, glucose, renal function | Hypokalemia, hypoglycemia, elevated creatinine |
| Magnesium | Deficiency in 30-80%; contributes to seizures, arrhythmias [12] | less than 0.75 mmol/L (less than 1.8 mg/dL) |
| Phosphorus | Deficiency; risk of refeeding syndrome | less than 0.8 mmol/L (less than 2.5 mg/dL) |
| LFTs | Liver disease severity | AST:ALT ratio >2:1 (alcoholic liver disease), elevated GGT |
| INR/PT | Coagulopathy, liver synthetic function | Prolonged if cirrhosis or vitamin K deficiency |
| Lipase | Alcoholic pancreatitis | Elevated >3× ULN |
| Blood alcohol level | Baseline; withdrawal can occur at any level [10] | May be elevated or zero |
| Lactate | Sepsis, hypovolemia, alcoholic ketoacidosis | Elevated if shock or ketoacidosis |
Additional Labs (Based on Clinical Picture):
| Test | Indication | Finding |
|---|---|---|
| Ammonia | Altered mental status, known liver disease | >100 μg/dL suggests hepatic encephalopathy |
| Blood cultures | Fever, suspected sepsis | If positive, adjust antibiotics |
| Arterial blood gas | Respiratory distress, severe acidosis | Metabolic acidosis (ketoacidosis), respiratory alkalosis |
| Creatine kinase (CK) | Suspected rhabdomyolysis (agitation, seizures) | >1000 U/L concerning; >5000 U/L severe |
| Troponin | Chest pain, ECG changes | Elevated if MI |
| Urine drug screen | Polysubstance use suspected | Benzodiazepines, opioids, stimulants |
| β-hydroxybutyrate | Suspected alcoholic ketoacidosis | Elevated (ketoacidosis) |
Imaging and Specialized Tests
| Test | Indication | Findings |
|---|---|---|
| CT Head (non-contrast) | Altered mental status, focal deficits, trauma, first seizure, persistent confusion | Hemorrhage (SDH, EDH, SAH), infarct, mass |
| Chest X-ray | Fever, respiratory symptoms, cough, hypoxia | Aspiration pneumonia, pulmonary edema, effusion |
| ECG | Baseline (all patients); chest pain; arrhythmia | Prolonged QTc, atrial fibrillation, ischemic changes |
| Lumbar Puncture | Fever + altered mental status + meningeal signs (after CT) | Meningitis vs. encephalitis |
| EEG | Status epilepticus, non-convulsive seizures | Seizure activity |
Risk Stratification Tools
PAWSS (Prediction of Alcohol Withdrawal Severity Scale): [18]
- Used to identify patients at risk for complicated withdrawal
- Score ≥4 predicts moderate-severe withdrawal requiring pharmacological prophylaxis
- Components: prior withdrawal history, BAL on admission, autonomic symptoms, concurrent illness
Modified CIWA-Ar: [11]
- Used for ongoing symptom assessment and symptom-triggered protocols
- Reassess frequency based on score:
- "CIWA less than 10: q4-8h"
- "CIWA 10-19: q1-2h"
- "CIWA ≥20: q30-60min"
Treatment
Principles of Management
- Supportive Care: Calm, quiet environment; minimize stimulation; IV hydration; nutrition
- Thiamine FIRST: Always give thiamine before glucose to prevent Wernicke's encephalopathy [1,2]
- Benzodiazepines First-Line: Prevent seizures and progression to DTs [5,6]
- Symptom-Triggered Preferred: CIWA-based dosing reduces total benzodiazepine use and length of stay vs. fixed-schedule [11,20]
- Electrolyte Repletion: Correct magnesium, potassium, phosphorus [12]
- Adjunct Therapies: Phenobarbital for refractory cases [7,8]; dexmedetomidine in ICU [19]
- Treat Complications: Seizures, arrhythmias, aspiration, concurrent infections
Thiamine Replacement (CRITICAL)
Rationale: [1,2]
- Thiamine deficiency is nearly universal in chronic alcohol use disorder
- Glucose administration increases thiamine utilization → can precipitate or worsen Wernicke's encephalopathy in deficient patients
- ALWAYS give thiamine BEFORE glucose (or simultaneously)
Wernicke's Encephalopathy: [1]
- Classic triad (only 10-16% of cases): confusion, ataxia, ophthalmoplegia
- Other presentations: hypothermia, coma, hypotension
- If untreated: 20% mortality; 85% develop Korsakoff's psychosis (irreversible amnesia)
- Treatment is time-sensitive: Neurological damage occurs within hours
Dosing Protocols: [1,2]
| Indication | Route | Dose | Duration | Evidence |
|---|---|---|---|---|
| Prophylaxis (all AWS patients) | IV or IM | 100-250mg daily | 3-5 days | IIIA |
| High-dose prophylaxis (preferred) | IV (Pabrinex) | 500mg IV TDS | 3-5 days, then 250mg daily × 5 days | IIA [1] |
| Suspected/confirmed Wernicke's | IV (Pabrinex) | 500mg IV TDS | Until symptom resolution (minimum 3-5 days) | IA [1,2] |
UK Protocol (Pabrinex): [1]
- Pabrinex = high-potency IV vitamins B and C (thiamine 250mg per pair of ampoules)
- Prophylaxis: 1 pair (500mg thiamine) TDS × 3-5 days
- Treatment: 2-3 pairs (1000-1500mg thiamine) TDS × 3-5 days
Route:
- IV preferred (100% bioavailability; oral absorption impaired in alcoholism)
- Administer slowly over 30 minutes (risk of anaphylaxis with rapid IV push, though rare)
Benzodiazepine Therapy
Mechanism: [5,6]
- Benzodiazepines enhance GABA-A receptor activity → compensate for downregulated GABA tone
- Prevent progression to seizures and delirium tremens
- Cross-tolerance with alcohol
Choice of Benzodiazepine: [5,6]
| Agent | Route | Half-life | Active Metabolites | Clinical Use |
|---|---|---|---|---|
| Diazepam | IV, PO | Long (20-100h) | Yes (desmethyldiazepam) | Preferred for most patients; self-tapering effect; smoother course [6] |
| Lorazepam | IV, IM, PO | Intermediate (10-20h) | No | Preferred in hepatic impairment, elderly; predictable metabolism [6] |
| Chlordiazepoxide | PO only | Long (30-100h) | Yes | Oral only; mild-moderate withdrawal; outpatient detox |
| Oxazepam | PO only | Short (4-15h) | No | Hepatic impairment; requires frequent dosing |
Symptom-Triggered Protocol (CIWA-Based): [11,20]
| CIWA Score | Action | Typical Dose | Reassessment |
|---|---|---|---|
| less than 10 | Monitor; no medication needed (unless high-risk patient) | - | Every 4-8 hours |
| 10-19 | Administer benzodiazepine; reassess in 1 hour | Diazepam 10-20mg PO/IV OR Lorazepam 2-4mg PO/IV | Every 1-2 hours |
| ≥20 | Aggressive treatment; give benzodiazepine q10-15min until CIWA less than 10 | Diazepam 10-20mg IV q10-15min OR Lorazepam 4mg IV q10-15min | Every 30-60 minutes until controlled, then q1-2h |
Advantages of Symptom-Triggered: [20]
- Reduced total benzodiazepine dose (approximately 25% reduction)
- Shorter treatment duration (median 9h vs. 68h for fixed-schedule)
- Shorter hospital length of stay
- Individualized to patient severity
Fixed-Schedule Protocol (Alternative): [6]
Indications:
- CIWA assessment unreliable (intubated, delirious, cognitively impaired, language barrier)
- Patient unable to cooperate with frequent assessments
- Nursing staff unfamiliar with CIWA protocols
Example Protocol:
- Diazepam: 10mg PO q6h × 4 doses (Day 1), then 5mg PO q6h × 8 doses (Days 2-3)
- Lorazepam: 2mg PO q6h × 4 doses (Day 1), then 1mg PO q6h × 8 doses (Days 2-3)
- PRN doses for breakthrough symptoms (CIWA ≥10)
Front-Loading Protocol: [6]
- Rapid administration of large benzodiazepine doses upfront to achieve sedation
- Example: Diazepam 20mg PO q2h until CIWA less than 10 or patient sedated
- Used in severe withdrawal to rapidly control symptoms
Severe/Refractory Withdrawal: [4,6]
- Some patients require massive doses: >200-500mg diazepam equivalents in 24 hours
- No maximum dose ceiling; titrate to symptom control and sedation
- If requiring >200mg diazepam equivalents: Add phenobarbital early (do not wait for failure) [7,8]
- Consider ICU admission for high-dose benzodiazepine protocols
- Monitor for respiratory depression (especially when combining agents)
Benzodiazepine Equivalencies:
- Diazepam 10mg = Lorazepam 2mg = Chlordiazepoxide 50mg
Detailed Benzodiazepine Dosing Algorithms:
Exam Detail: Algorithm 1: Symptom-Triggered Diazepam Protocol (Most Common): [6,20]
CIWA less than 10:
→ No medication
→ Reassess q4-8h
→ Continue monitoring
CIWA 10-15:
→ Diazepam 10mg PO/IV
→ Reassess CIWA in 1 hour
→ If CIWA still ≥10: Repeat diazepam 10mg
→ Continue until CIWA less than 10
→ Total doses in 24h typically: 40-80mg
CIWA 16-20:
→ Diazepam 20mg PO/IV
→ Reassess CIWA in 1 hour
→ If CIWA still ≥15: Repeat diazepam 20mg
→ If CIWA 10-14: Give diazepam 10mg
→ Continue until CIWA less than 10
→ Total doses in 24h typically: 80-160mg
CIWA ≥ 20 (Severe/DTs):
→ Diazepam 20mg IV immediately
→ Repeat 20mg IV q10-15min until patient calm or lightly sedated
→ No maximum dose (some patients require >500mg/24h)
→ Monitor respiratory rate and sedation level
→ If >200mg in 24
h: Add phenobarbital 130-260mg IV
→ Consider ICU transfer
Algorithm 2: Lorazepam Protocol (Hepatic Impairment, Elderly): [6]
CIWA less than 10:
→ No medication
→ Reassess q4-8h
CIWA 10-15:
→ Lorazepam 2mg PO/IV/IM
→ Reassess in 1 hour
→ Repeat 2mg if CIWA still ≥10
CIWA 16-20:
→ Lorazepam 4mg PO/IV/IM
→ Reassess in 1 hour
→ Repeat 2-4mg based on response
CIWA ≥ 20:
→ Lorazepam 4mg IV immediately
→ Repeat 4mg IV q10-15min until calm
→ If >40mg in 24
h: Add phenobarbital
Algorithm 3: Front-Loading Protocol (Rapid Symptom Control): [6]
Indication: Severe withdrawal, anticipated complicated course
Hour 0: Diazepam 20mg PO
Hour 2: Assess CIWA
- "If CIWA ≥10: Diazepam 20mg PO"
- "If CIWA less than 10: Hold, reassess q2h"
Hour 4: Repeat assessment and dosing
Continue until CIWA less than 10 for 2 consecutive assessments
Then switch to PRN symptom-triggered
Typical total dose: 60-120mg in first 6-8 hours, then taper
Algorithm 4: Fixed-Schedule Protocol (Cannot Use CIWA): [6]
Indication: Intubated, delirious, cognitively impaired, language barrier
Mild-Moderate Withdrawal (Predicted):
Day 1: Diazepam 10mg PO/IV q6h × 4 doses (40mg total)
Day 2: Diazepam 10mg PO q8h × 3 doses (30mg total)
Day 3: Diazepam 5mg PO q8h × 3 doses (15mg total)
Day 4: Diazepam 5mg PO q12h × 2 doses (10mg total)
Day 5: Stop
PLUS PRN doses if breakthrough symptoms:
- CIWA ≥10 or severe agitation: Extra 10-20mg diazepam
For Severe Withdrawal (Previous DTs, high PAWSS score):
- Double the above doses
- Consider ICU admission
- Add phenobarbital early (Day 1-2)
Conversion Between Benzodiazepines:
If switching from diazepam to lorazepam (e.g., new hepatic dysfunction):
- Calculate total diazepam in previous 24h
- Divide by 5 to get lorazepam equivalent
- Example: 100mg diazepam = 20mg lorazepam
- Give 50% of 24h dose divided q6h, then PRN
Monitoring During High-Dose Benzodiazepine Therapy: [4,6]
| Parameter | Frequency | Action Threshold |
|---|---|---|
| Respiratory rate | Continuous (if >100mg diazepam/24h) | RR less than 10/min → hold dose, stimulate patient, consider airway support |
| Oxygen saturation | Continuous | SpO2 less than 92% → supplemental oxygen; less than 88% → evaluate for aspiration/intubation |
| Sedation level | q30-60min | RASS -3 to -4 (deep sedation) → hold next dose; RASS -5 (unarousable) → airway management |
| CIWA score | Per protocol | Persistent CIWA ≥15 despite adequate dosing → add adjunct (phenobarbital) |
| Vital signs | q15-30min (severe), q1-2h (moderate) | HR >120, SBP >180, Temp >39°C despite treatment → ICU, consider adjuncts |
| Cumulative dose | Track 24h total | >200mg diazepam equivalent → consider phenobarbital; >500mg → ICU + advanced sedation |
Common Dosing Errors to Avoid: [6]
❌ Using fixed maximum doses: "No more than 80mg diazepam per day"
- ✅ Correct: No ceiling; titrate to symptom control
❌ Waiting for CIWA ≥20 before aggressive treatment
- ✅ Correct: Escalate at CIWA ≥15 persistently or any DTs signs
❌ Switching to long-acting in acute severe withdrawal
- ✅ Correct: Use IV short-onset benzos (diazepam IV, lorazepam IV) for rapid control
❌ Under-dosing in obese patients
- ✅ Correct: Dose based on clinical response, not weight
❌ Giving IM diazepam
- ✅ Correct: Diazepam has erratic IM absorption; use IV or PO only (lorazepam OK IM)
Delirium Tremens: Comprehensive ICU Management
Delirium Tremens Diagnostic Criteria: [3,4]
All of the following must be present:
- Delirium: Acute confusion, disorientation (time/place/person), fluctuating consciousness
- Recent alcohol cessation: Typically 48-96 hours after last drink
- Autonomic hyperactivity: Tachycardia (HR >120), hypertension or labile BP, hyperthermia (>38.3°C), diaphoresis
- Exclusion of other causes: Infection, head trauma, metabolic derangement ruled out
Optional features (support diagnosis):
- Hallucinations (visual > auditory > tactile)
- Severe psychomotor agitation
- Coarse tremor
Severity Stratification: [3,4]
| Severity | Clinical Features | Mortality Risk | Disposition |
|---|---|---|---|
| Mild DTs | Delirium + mild autonomic (HR 100-120, SBP less than 160, Temp less than 38.5°C) | 1-2% | ICU or monitored step-down |
| Moderate DTs | Delirium + significant autonomic (HR 120-140, SBP 160-180, Temp 38.5-39°C) + moderate agitation | 2-5% | ICU with 1:1 nursing |
| Severe DTs | Delirium + extreme autonomic (HR >140, SBP >180, Temp >39°C) + combative agitation OR multi-organ dysfunction | 5-15% untreated | ICU, often requires intubation |
ICU Management Protocol for Delirium Tremens: [4]
Exam Detail: Step 1: Immediate Stabilization (0-30 minutes)
-
Secure Airway:
- High-flow oxygen via non-rebreather if SpO2 less than 94%
- Assess airway protection (gag reflex, ability to manage secretions)
- Intubation criteria: Respiratory failure, aspiration risk, inability to protect airway, need for high-dose sedation with respiratory depression risk
- Rapid Sequence Intubation: Etomidate 0.3mg/kg + succinylcholine 1.5mg/kg OR rocuronium 1mg/kg
-
IV Access and Monitoring:
- Two large-bore peripheral IVs (16-18G)
- Consider central line if prolonged ICU stay anticipated
- Arterial line for continuous BP monitoring if severe autonomic instability
- Continuous telemetry (arrhythmia risk)
- Urinary catheter (strict I/O monitoring)
-
Stat Medications:
- Thiamine 500mg IV over 30 minutes (FIRST, before any glucose) [1,2]
- Glucose: 50mL D50W IV if hypoglycemic (after or with thiamine)
- Benzodiazepine loading: Diazepam 20mg IV push, repeat q10-15min until sedation (target RASS -1 to -2)
- Magnesium: 4g MgSO4 IV over 20 minutes if Mg less than 0.75 mmol/L
-
Stat Labs:
- CBC, CMP, Mg, Phos, LFTs, INR, lactate, blood alcohol level
- Arterial blood gas (if intubated or respiratory distress)
- Blood cultures × 2 (if fever >38.3°C)
- Urinalysis and culture
- CXR (aspiration, pneumonia)
- CT head non-contrast (if altered mental status etiology unclear, trauma, focal deficits)
Step 2: Aggressive Benzodiazepine Therapy (0-6 hours) [4,6]
Diazepam-Based Protocol (Preferred):
- Loading: 20mg IV q10-15min until RASS -1 to -2 (calm, responds to voice)
- No maximum dose: Continue until symptom control
- Typical total loading: 100-300mg in first 2-4 hours (range: 60-1000mg)
- Maintenance: Once controlled, reassess CIWA q1-2h and give 10-20mg PRN for CIWA ≥10
- Self-tapering: Long half-life provides auto-taper over days
Lorazepam-Based Protocol (Alternative, especially if hepatic dysfunction):
- Loading: 4mg IV q10-15min until sedation
- Typical total loading: 20-60mg
- Maintenance: 2-4mg IV q1-4h PRN for agitation or CIWA ≥10
Monitoring:
- Assess sedation (RASS score) and respiratory rate q15min during loading
- Continuous pulse oximetry
- If respiratory rate less than 10 or SpO2 less than 90%: Hold benzodiazepines, stimulate patient, bag-mask ventilation if needed, prepare for intubation
Step 3: Adjunctive Therapy for Refractory DTs [7,8,19]
Indications for Adjuncts:
- Benzodiazepine requirement >200mg diazepam (or 40mg lorazepam) in 24 hours
- Persistent severe agitation despite adequate benzodiazepines
- Respiratory depression limiting further benzodiazepine dosing
- Severe refractory autonomic instability (HR >140, SBP >200)
Option 1: Phenobarbital (First-Line Adjunct): [7,8]
Protocol:
- Loading dose: 10mg/kg IV over 30 minutes (typically 5-7 g for 70kg patient, given as 130-260mg boluses q15-30min)
- Maintenance: 60-130mg IV q6-12h OR CIWA-triggered 65-130mg PRN
- Maximum: 15-20mg/kg total load in 24 hours
Advantages:
- Synergistic with benzodiazepines (different GABA-A binding site)
- Long half-life (80-120 hours) → smooth taper
- Effective anticonvulsant
- Reduces benzodiazepine requirements by ~50%
Monitoring:
- Respiratory depression: Most serious risk, especially with concurrent benzodiazepines
- Assess RR and SpO2 q15min during loading, q1h after
- Prepare for intubation (have equipment at bedside)
- Hypotension (less common than benzos)
- No reversal agent (unlike benzodiazepines)
Option 2: Dexmedetomidine (ICU Setting): [19]
Protocol:
- Loading dose: 1mcg/kg IV over 10 minutes (optional, often omitted to avoid bradycardia)
- Maintenance infusion: 0.2-1.5 mcg/kg/h IV (titrate to effect)
- Continue: Until withdrawal symptoms controlled, typically 2-5 days
Advantages:
- Alpha-2 agonist → reduces sympathetic outflow → ↓ HR, ↓ BP
- Does NOT cause respiratory depression (safe in awake patients)
- Anxiolysis and sedation without GABA effects
- Reduces benzodiazepine/phenobarbital requirements
- May improve delirium outcomes
Limitations:
- Does NOT prevent seizures (must use with benzodiazepines)
- Bradycardia (HR less than 50) and hypotension common at high doses
- Expensive
- Requires ICU monitoring
Monitoring:
- Continuous telemetry (bradycardia)
- Arterial line recommended for titration
- Hold infusion if HR less than 50 or SBP less than 90
Option 3: Propofol (Last Resort, Intubated Patients Only): [4]
Indications:
- Intubated patients with refractory agitation despite benzodiazepines + phenobarbital + dexmedetomidine
- Status epilepticus refractory to standard treatment
Protocol:
- Infusion: 20-50 mcg/kg/min (titrate to sedation)
- Monitor: Triglycerides (daily), lactate (risk of propofol infusion syndrome)
- Duration: Minimize (less than 72 hours if possible due to PRIS risk)
Risks:
- Propofol infusion syndrome (PRIS): Metabolic acidosis, rhabdomyolysis, renal failure, cardiac arrest
- Does NOT prevent seizures
- Requires intubation and mechanical ventilation
Step 4: Autonomic Stabilization [4]
Hyperthermia Management (Temperature >39°C):
- Cooling measures:
- Remove excess clothing/blankets
- Cooling blankets (target 37-38°C)
- IV fluids at room temperature (avoid iced fluids → shivering)
- Ice packs to groin, axillae if >40°C
- Acetaminophen: 1g PO/PR/IV q6h (limited efficacy in DTs but safe)
- Avoid aspirin: Risk of bleeding if thrombocytopenic or coagulopathic
- Do NOT use: Dantrolene (not effective in DTs; use only if malignant hyperthermia suspected)
Tachycardia Management (HR >120-140):
- First-line: Adequate benzodiazepines (treat underlying withdrawal)
- Adjunct: Dexmedetomidine 0.2-0.7 mcg/kg/h (↓ HR by 10-30%)
- Caution with beta-blockers:
- Generally avoided (can mask tachycardia without treating CNS hyperexcitability)
- May use short-acting IV esmolol 50-200 mcg/kg/min if refractory tachycardia with cardiac ischemia
- Must have adequate benzodiazepine on board first
Hypertension Management (SBP >180-200):
- First-line: Benzodiazepines (address catecholamine surge)
- Adjunct: Dexmedetomidine (↓ SBP by 10-20%)
- Refractory severe hypertension (SBP >220 or end-organ damage):
- Nicardipine 5-15 mg/h IV infusion (titratable)
- Labetalol 10-20mg IV bolus, then 0.5-2 mg/min infusion
- "Avoid: Hydralazine (reflex tachycardia), nitroprusside (cyanide risk in malnourished)"
Step 5: Fluid and Electrolyte Management [12]
IV Fluid Resuscitation:
- Initial: NS or LR 500-1000mL bolus (often volume depleted from diaphoresis, poor PO intake)
- Maintenance: D5 1/2 NS at 75-150 mL/h (provides glucose + electrolytes)
- Goal: Urine output >0.5 mL/kg/h, MAP >65 mmHg
- Caution: Risk of volume overload if underlying cardiomyopathy; may need CVP monitoring
Electrolyte Repletion (Aggressive Protocols):
Magnesium (deficient in 30-80% of patients): [12]
- Severe deficiency (less than 0.5 mmol/L): 4-6g MgSO4 IV over 4-6 hours, then 2g q6h × 3 doses
- Moderate deficiency (0.5-0.75 mmol/L): 2-4g MgSO4 IV over 2-4 hours
- Recheck: Mg level q12h until >0.85 mmol/L
- Maintenance: 1-2g MgSO4 IV daily × 5 days
- Monitor: Deep tendon reflexes (hyporeflexia suggests Mg >2.5 mmol/L)
Potassium (deficient in 30-50%): [12]
- Severe hypokalemia (less than 3.0 mEq/L): 40 mEq KCl IV over 2-4h via central line (peripheral: 10 mEq/h max)
- Must correct Mg FIRST: Hypomagnesemia prevents K repletion
- Recheck: K q2-4h until >3.5 mEq/L
- Continuous ECG monitoring during IV K replacement (arrhythmia risk)
Phosphorus (deficient in 20-30%; refeeding syndrome risk): [12]
- Severe hypophosphatemia (less than 0.5 mmol/L): 15-30 mmol sodium/potassium phosphate IV over 4-6 hours
- Monitor: Calcium (phosphate repletion → hypocalcemia), recheck phos q12h
- Avoid over-repletion: Goal 0.8-1.2 mmol/L (not >1.5)
Step 6: Supportive and Preventive Care
Nutrition:
- NPO if: GCS less than 12, severe agitation, active vomiting, high aspiration risk
- Enteral nutrition (when safe, usually after 12-24h of sedation control):
- NG tube feeding if cannot take PO
- Start slowly to avoid refeeding syndrome (25-50% of goal rate × 24h, then advance)
- Monitor phosphorus, potassium, magnesium daily during refeeding
Thiamine + Multivitamins:
- Thiamine 500mg IV TDS × 3-5 days (continue high-dose until eating), then 250mg IV daily × 5 days [1,2]
- Folate 1-5mg PO/IV daily
- Multivitamin daily
- Vitamin K 10mg IV × 1 if INR >1.5
Aspiration Precautions:
- Head of bed elevated 30-45 degrees
- Oral suctioning PRN
- Assess swallow before PO intake
- Intubate if cannot protect airway
DVT Prophylaxis:
- Enoxaparin 40mg SC daily OR heparin 5000 units SC q8-12h
- Sequential compression devices
- Early mobilization when safe
Skin Care:
- Turn q2h (pressure ulcer prevention in obtunded patients)
- Moisture barrier cream (profuse diaphoresis)
Restraints (Use Sparingly):
- Chemical restraint preferred (sedation)
- Physical restraints only if necessary for safety (patient pulling lines, violent)
- Soft wrist/ankle restraints (avoid rigid restraints → rhabdomyolysis, agitation)
- Reassess need q2h, remove as soon as possible
Step 7: Complications Surveillance and Management
Arrhythmias: [3]
- Atrial fibrillation (most common):
- "Rate control: Optimize benzodiazepines, consider metoprolol 2.5-5mg IV q6h if HR >140 and hemodynamically stable"
- "Anticoagulation: Usually defer until withdrawal phase over (bleeding risk, fall risk)"
- Often spontaneously converts after withdrawal resolves
- Ventricular arrhythmias:
- Correct electrolytes (K >4.0, Mg >0.85)
- Treat ischemia if present
- Amiodarone 150mg IV over 10 min, then 1 mg/min infusion if sustained VT
Seizures in ICU: [13]
- First seizure: Lorazepam 4mg IV OR diazepam 10mg IV
- Recurrent seizures: Increase benzodiazepine maintenance dose
- Status epilepticus: See Algorithm below
- Do NOT use phenytoin/fosphenytoin: Ineffective for AWS seizures [13]
Aspiration Pneumonia:
- High suspicion if: Witnessed aspiration, new fever, new CXR infiltrate, hypoxia
- CXR: Right lower lobe most common
- Antibiotics: Ampicillin-sulbactam 3g IV q6h OR pip-tazo 3.375g IV q6h (cover anaerobes, GNRs)
- Consider intubation if worsening hypoxia or respiratory distress
Rhabdomyolysis:
- Risk factors: Extreme agitation, physical restraints, seizures, prolonged immobility
- Labs: CK (>1000 IU/L concerning; >5000 severe), myoglobin, creatinine
- Treatment:
- "Aggressive IV hydration: NS 200-300 mL/h (goal UOP >200 mL/h)"
- Monitor CK daily until downtrending
- Watch for hyperkalemia, hypocalcemia, AKI
- Dialysis if AKI with volume overload or severe electrolyte derangement
Step 8: Transition and De-Escalation (Days 3-7)
Criteria for ICU De-Escalation:
- CIWA consistently less than 10 for ≥24 hours
- Benzodiazepine requirement less than 40mg diazepam equivalent/24h
- No adjunct infusions needed (dexmedetomidine weaned off)
- Vital signs stable (HR less than 100, BP less than 140/90, afebrile) × 24h
- No active medical complications requiring ICU-level care
Weaning Sedation:
- Diazepam: Self-tapering due to long half-life; can stop PRN dosing once CIWA less than 10 × 24h
- Lorazepam: If on fixed-dose schedule, taper by 25-50% per day
- Phenobarbital: Self-tapering (very long half-life); do not abruptly stop
- Dexmedetomidine: Wean by 0.1-0.2 mcg/kg/h q6-12h (avoid rebound hypertension)
Transfer to Floor:
- Continue CIWA assessments q4-8h × 48h
- Continue thiamine, electrolyte repletion, nutrition support
- Addiction psychiatry consult
- Discharge planning
DTs Mortality Predictors: [3,4]
| Factor | Mortality Risk |
|---|---|
| Age >65 years | 3-fold increase |
| Concurrent infection (sepsis, pneumonia) | 4-fold increase |
| Temperature >40°C | 5-fold increase |
| GCS less than 8 on presentation | 6-fold increase |
| Acute kidney injury (Cr >2.0) | 3-fold increase |
| Thrombocytopenia less than 50,000 | 2-fold increase |
| Admission to ICU >24h after DTs onset | 2-fold increase |
Common Causes of Death in DTs: [3]
- Cardiovascular collapse (arrhythmias, MI, cardiogenic shock)
- Aspiration pneumonia → respiratory failure
- Sepsis (often concurrent infection)
- Multi-organ failure
- Traumatic injury (falls, head trauma while delirious)
Evidence: [7,8]
- Cochrane review (2023): Phenobarbital may reduce ICU admission and intubation in severe AWS [7]
- JAMA Network Open (2024): Phenobarbital vs. benzodiazepines had lower intubation rates (OR 0.58) [8]
- Mechanism: GABA-A agonist (like benzodiazepines) but different binding site; synergistic effect
Indications: [7,8]
- Benzodiazepine-refractory withdrawal (persistent CIWA ≥15 despite >200mg diazepam equivalents)
- Adjunct to benzodiazepines in severe DTs
- Polysubstance users with benzodiazepine tolerance
- Alternative first-line in some protocols (though benzodiazepines remain standard)
Dosing: [7,8]
| Protocol | Dose | Notes |
|---|---|---|
| Adjunctive (most common) | Phenobarbital 130-260mg IV q15-30min PRN (max 10mg/kg) | Added to ongoing benzodiazepines; reassess after each dose |
| Primary therapy (some centers) | Phenobarbital 10mg/kg IV loading dose, then CIWA-based dosing | Followed by symptom-triggered 65-130mg IV PRN |
Advantages:
- Long half-life (80-120h) → self-tapering, smooth course
- Anticonvulsant properties
- May reduce total sedative requirements
- Lower abuse potential than benzodiazepines
Cautions: [7]
- Respiratory depression (especially when combined with benzodiazepines) → monitor closely; may require intubation
- Very long half-life → accumulation over days
- No reversal agent (unlike benzodiazepines with flumazenil)
- Hypotension (less common than with benzodiazepines)
Adjunctive Medications
Dexmedetomidine (ICU Setting): [19]
| Feature | Details |
|---|---|
| Mechanism | Alpha-2 agonist; reduces sympathetic outflow |
| Indications | Adjunct in ICU for refractory DTs; reduces benzodiazepine requirements |
| Advantages | Does NOT cause respiratory depression; reduces HR, BP; anxiolysis |
| Limitations | Does NOT prevent seizures; must use with benzodiazepines; bradycardia; hypotension |
| Dosing | 0.2-1.5 mcg/kg/h IV infusion (titrate to effect) |
| Evidence | Meta-analysis: reduced ICU LOS, mechanical ventilation duration [19] |
Propofol (ICU Setting):
- Reserved for intubated patients with refractory agitation
- Does NOT prevent seizures
- Risk: Propofol infusion syndrome with prolonged use
Antipsychotics (Haloperidol): [4]
- NOT first-line; do NOT prevent seizures or DTs
- Role: Adjunct for severe agitation/hallucinations after adequate benzodiazepines
- Dose: Haloperidol 2.5-5mg IV q30-60min PRN
- Cautions: Lowers seizure threshold; QTc prolongation; neuroleptic malignant syndrome
Beta-Blockers and Clonidine:
- NOT recommended as monotherapy; do NOT prevent seizures or DTs [4]
- May mask tachycardia/hypertension without treating underlying CNS hyperexcitability
- Can be used as adjuncts for refractory hypertension/tachycardia
Electrolyte Replacement
Magnesium: [12]
| Serum Mg Level | Replacement Protocol | Route | Monitoring |
|---|---|---|---|
| less than 0.5 mmol/L (less than 1.2 mg/dL) | 4g MgSO4 IV over 4h, then 2g q6h × 2 doses | IV | Recheck Mg in 6h; tendon reflexes (hyporeflexia if Mg overdose) |
| 0.5-0.75 mmol/L (1.2-1.8 mg/dL) | 2g MgSO4 IV over 2h | IV | Recheck in 12h |
| >0.75 mmol/L (>1.8 mg/dL) | Maintenance: 1g MgSO4 IV daily | IV or PO | - |
Rationale: Hypomagnesemia contributes to refractory hypokalemia, seizures, arrhythmias
Potassium: [12]
| Serum K Level | Replacement | Monitoring |
|---|---|---|
| less than 3.0 mEq/L | 40 mEq KCl IV over 2-4h (via central line) OR 20 mEq/h via peripheral | Continuous ECG; recheck K q2-4h; correct Mg first (Mg deficiency prevents K repletion) |
| 3.0-3.5 mEq/L | 20-40 mEq KCl PO q6-8h | Recheck K q6-12h |
Phosphorus: [12]
- Replace if less than 0.8 mmol/L (less than 2.5 mg/dL): 15-30 mmol sodium/potassium phosphate IV
- Caution: Avoid over-repletion (risk of hypocalcemia, metastatic calcification)
- Monitor calcium, phosphorus q12-24h
Seizure Management
Alcohol Withdrawal Seizures: [13]
Characteristics:
- Generalized tonic-clonic (>90%)
- Usually brief (less than 2-3 min), self-limited
- Single seizure or cluster of 2-6 within 6 hours
- Occur 6-48h post-cessation (peak 24h)
Acute Seizure Treatment: [13]
- Benzodiazepines (first-line):
- Lorazepam 4mg IV OR Diazepam 10mg IV
- Repeat once if seizure recurs
- Phenytoin/Fosphenytoin: NOT effective for AWS seizures [13]
- Prophylaxis: Adequate benzodiazepine dosing prevents recurrent seizures
Status Epilepticus: [13]
- Rare (3% of AWS seizures) but life-threatening
- Definition: Seizure >5 minutes or recurrent seizures without recovery
- Treatment:
- Benzodiazepines (lorazepam 4mg IV, repeat × 1)
- Phenobarbital 15-20mg/kg IV (preferred second-line in AWS)
- Propofol or midazolam infusion if refractory
- Intubation if airway compromise
Post-Seizure:
- If first seizure: CT head to rule out structural lesion
- If known AWS seizures: Continue benzodiazepines, monitor closely
- Do NOT discharge after seizure until CIWA controlled and symptom-free × 24h
Comprehensive Seizure Management and Prophylaxis
Exam Detail: Pathophysiology of Alcohol Withdrawal Seizures: [13,14]
- Acute GABA-A receptor downregulation → loss of inhibitory tone
- NMDA receptor upregulation → excessive glutamate excitation
- Ion channel dysregulation → neuronal hyperexcitability
- Lowered seizure threshold begins 6-12h post-cessation, lowest at 24h
- Kindling phenomenon → each withdrawal episode further lowers threshold permanently [17]
Seizure Characteristics (How to Recognize AWS Seizures): [13]
| Feature | AWS Seizures | Epileptic Seizures |
|---|---|---|
| Type | Generalized tonic-clonic (90%) | Variable (focal, generalized) |
| Timing | 6-48h post-cessation (peak 24h) | Anytime |
| Duration | Brief (less than 2-3 min) | Variable |
| Recurrence | Single or cluster (2-6) within 6h | Variable pattern |
| Post-ictal period | Short (minutes) | Often prolonged (hours) |
| Focal onset | Rare (less than 10%) | Common in focal epilepsy |
| Response to phenytoin | Not effective | Effective |
| Response to benzodiazepines | Effective for prophylaxis + acute | Variably effective |
Differential Diagnosis of Seizures in Alcohol Use Disorder:
| Cause | Distinguishing Features | Evaluation |
|---|---|---|
| Alcohol withdrawal | Timing 6-48h, brief, GTCS, cluster | History, timeline, BAL, CIWA score |
| Hypoglycemia | Blood glucose less than 3 mmol/L, rapid response to dextrose | Fingerstick glucose |
| Hyponatremia | Na less than 125 mEq/L, gradual onset | Serum sodium |
| Head trauma | History of fall, focal deficits, persistent altered mental status | CT head non-contrast |
| Subdural hematoma | Chronic headache, gradual decline, elderly, falls | CT head |
| Wernicke encephalopathy | Triad (only 10-16%), ataxia, ophthalmoplegia | Clinical diagnosis; thiamine trial |
| Meningitis/encephalitis | Fever, neck stiffness, persistent confusion | CT head → LP |
| Epilepsy (pre-existing) | History of seizures, off medications, focal onset | History, EEG |
| Stroke | Focal deficits, sudden onset, risk factors | CT head, MRI |
| Hepatic encephalopathy | Asterixis, elevated ammonia, liver disease | Ammonia, LFTs |
| Drug toxicity | Polysubstance use (cocaine, isoniazid, tramadol, bupropion) | History, urine drug screen |
| Electrolyte | Multiple abnormalities common in alcoholism | CMP, Mg, Phos, Ca |
SEIZURE PROPHYLAXIS PROTOCOL: [5,6,13]
Risk Stratification for Seizures:
HIGH RISK (Seizure incidence 25-50% without prophylaxis):
- Previous alcohol withdrawal seizures
- Previous delirium tremens
- Multiple prior detoxifications (kindling) [17]
- Heavy, prolonged use (>10 years daily heavy drinking)
- Concurrent benzodiazepine withdrawal
- Seizure within past 48 hours
- Structural brain lesion (prior head trauma, stroke)
- Severe hypomagnesemia (less than 0.5 mmol/L)
- Electrolyte derangements (Na less than 130, Mg less than 0.7, Ca less than 2.0)
MODERATE RISK (Seizure incidence 5-15%):
- CIWA ≥15 on admission
- Age >60 years
- Polysubstance use
- Concurrent acute illness
LOW RISK (Seizure incidence less than 5%):
- Mild withdrawal (CIWA less than 10)
- No prior complicated withdrawal
- Short duration/low quantity alcohol use
Prophylaxis Approach by Risk:
HIGH RISK Patients:
- Admit to monitored bed (floor with telemetry minimum, consider ICU)
- Benzodiazepine prophylaxis:
- "Option 1 (Preferred): Front-loading diazepam: 20mg PO at presentation, repeat 20mg q2h × 2 doses (60mg total load), then PRN symptom-triggered"
- "Option 2: Fixed-schedule diazepam: 10-20mg PO q6h × 24-48h, then PRN"
- "Option 3 (Hepatic impairment): Lorazepam 2-4mg PO/IV q6h × 24-48h, then PRN"
- Aggressive electrolyte repletion: Magnesium 2-4g IV load, then 1-2g q12h
- Thiamine 500mg IV TDS
- No role for phenytoin/levetiracetam prophylaxis (ineffective for AWS) [13]
- Monitor continuously for 48-72 hours (peak seizure window)
MODERATE RISK Patients:
- Admit to floor with frequent monitoring
- Symptom-triggered benzodiazepines with low threshold (CIWA ≥8)
- Electrolyte repletion
- Thiamine
- Monitor for 48-72h
LOW RISK Patients:
- Outpatient detoxification acceptable if:
- Reliable support person available
- Can attend daily clinic visits
- Stable housing
- No significant comorbidities
- Chlordiazepoxide taper: 25-50mg PO q6h × 2 days, then taper over 5-7 days
- Return precautions: Seizure, confusion, severe tremor, inability to tolerate PO → go to ED
ACUTE SEIZURE MANAGEMENT IN AWS:
First Seizure:
IMMEDIATE (Within 1 minute):
1. Ensure patient safety:
- Turn on side (recovery position)
- Protect head (pillow, move objects)
- Do NOT restrain or insert objects in mouth
- Suction if secretions/vomiting
2. Assess airway and breathing:
- Apply oxygen 15L via non-rebreather
- Suction oropharynx if needed
- Prepare for airway management if prolonged
3. Obtain IV access (if not already present)
4. Check fingerstick glucose immediately
- If less than 3 mmol/L: Give D50W 50mL IV STAT (after thiamine if not already given)
5. Monitor: Continuous pulse ox, telemetry, vital signs
MOST SEIZURES ARE SELF-LIMITED (less than 2-3 min):
→ Allow to stop spontaneously
→ Post-ictal period: monitor, reassure, maintain airway
→ Do NOT give benzodiazepines AFTER brief self-limited seizure
IF SEIZURE CONTINUES >2-3 minutes:
→ Lorazepam 4mg IV over 2 minutes OR Diazepam 10mg IV
→ May repeat once if seizure recurs
→ Prepare for intubation if respiratory depression
AFTER SEIZURE STOPS:
1. Post-ictal monitoring:
- Vital signs q15min × 1h
- Neurological assessment when patient arousable
- Assess for injury (tongue bite, head trauma, aspiration)
2. Immediate workup (if FIRST seizure ever OR atypical features):
- CT head non-contrast (rule out hemorrhage, mass, stroke)
- Labs: Glucose, electrolytes, calcium, magnesium, CBC
- Toxicology screen if polysubstance use suspected
- Lumbar puncture if concern for meningitis (after CT)
3. Seizure prophylaxis:
- INCREASE benzodiazepine dosing (prevents recurrence)
- Diazepam 20mg PO/IV, then 10-20mg q6h scheduled × 48h, PLUS PRN for CIWA ≥10
- Ensure adequate magnesium (goal >0.85 mmol/L)
- Continue thiamine 500mg IV TDS
4. Monitoring:
- ICU vs floor based on severity, comorbidities
- Continuous telemetry × 48h
- Seizure precautions (padded bed rails, bed alarm, fall precautions)
Second Seizure / Cluster Seizures (2-6 seizures within 6 hours):
This is COMMON in AWS (30% of patients with first seizure have cluster). [13]
APPROACH:
1. After each seizure, allow brief recovery period (5-10 min)
2. If second seizure within 6 hours:
- Lorazepam 2-4mg IV OR Diazepam 10mg IV AFTER seizure stops
- Increase standing benzodiazepine schedule:
→ Diazepam 20mg PO/IV q4-6h × 24-48h
OR
→ Lorazepam 4mg PO/IV q4-6h × 24-48h
3. MRI brain (if not done) to rule out structural lesion (higher suspicion if cluster)
4. ICU admission (cluster seizures = high risk for status epilepticus)
5. Consider phenobarbital adjunct:
- Phenobarbital 130mg IV q6h × 4 doses (prophylactic dosing)
- Evidence: Reduces seizure recurrence in withdrawal [7]
Status Epilepticus in AWS (Seizure >5 min OR continuous seizures):
RARE (3%) but LIFE-THREATENING. Mortality 10-20%. [13]
STATUS EPILEPTICUS PROTOCOL:
TIME 0-5 MINUTES:
- Call for help (ICU team, anesthesia if available)
- Secure airway: Jaw thrust, oral airway, BVM if needed
- High-flow oxygen 15L NRB
- IV access × 2
- Check glucose: If low, thiamine 500mg IV + D50W 50mL IV
TIME 5-10 MINUTES (First-Line):
- Lorazepam 4mg IV over 2 min
- If no IV: Midazolam 10mg IM
- May repeat lorazepam 4mg IV once if seizure continues
TIME 10-20 MINUTES (Second-Line):
→ Seizure continues despite 2 doses benzodiazepines
PREFERRED for AWS: **Phenobarbital** [7,13]
- 15-20 mg/kg IV (typically 1000-1500mg for 70kg adult)
- Infuse at 50-100 mg/min (10-30 min infusion)
- Monitor: High risk of respiratory depression → prepare for intubation
ALTERNATIVE: **Levetiracetam** (less evidence in AWS)
- 60 mg/kg IV (max 4500mg) over 10 min
- Safer (less respiratory depression) but less effective in AWS
AVOID in AWS: **Phenytoin/Fosphenytoin** (NOT effective for AWS seizures) [13]
TIME 20-40 MINUTES (Third-Line / Refractory Status):
→ Seizure continues despite second-line agents
→ INTUBATE (RSI: etomidate + rocuronium)
→ Continuous IV anesthetic:
Option 1: **Propofol**
- Loading: 2 mg/kg IV bolus
- Infusion: 20-50 mcg/kg/min (titrate to burst suppression on EEG)
- Monitor: Propofol infusion syndrome (lactic acidosis, rhabdomyolysis)
Option 2: **Midazolam**
- Loading: 0.2 mg/kg IV bolus
- Infusion: 0.05-2 mg/kg/h
- Less risk of PRIS than propofol
Option 3: **Pentobarbital**
- Loading: 5-15 mg/kg IV at 50 mg/min
- Infusion: 0.5-5 mg/kg/h
- Deep sedation, requires blood pressure support
→ Continuous EEG monitoring (goal: burst suppression)
→ Continue infusion × 24-48h after last clinical/electrographic seizure
→ Wean slowly
POST-STATUS CARE:
- Maintain benzodiazepines for AWS treatment (taper slowly over days)
- Thiamine, electrolytes, nutrition support
- Workup for precipitants (infection, hemorrhage, structural lesion)
- MRI brain when stable
- EEG when waking (assess for non-convulsive seizures)
- Prognosis: Worse if status >30 min, but most recover if underlying AWS treated
Special Consideration: Phenytoin Inefficacy in AWS:
CRITICAL PEARL: Phenytoin and fosphenytoin are NOT effective for alcohol withdrawal seizures. [13]
Evidence: [13]
- Landmark RCT (Rathlev et al., 1994): Phenytoin vs placebo for AWS seizure prophylaxis → NO difference in recurrent seizures (OR 0.9, 95% CI 0.4-2.1)
- Mechanism: AWS seizures are due to acute GABA/glutamate imbalance, NOT chronic epileptic focus; phenytoin stabilizes neuronal membranes but does not address neurotransmitter dysregulation
When phenytoin IS appropriate:
- Pre-existing epilepsy (patient usually on phenytoin baseline)
- Structural brain lesion (post-stroke, tumor, trauma) + alcohol withdrawal
- Status epilepticus refractory to benzodiazepines and phenobarbital (in combination)
Seizure Prophylaxis Summary Table:
| Agent | Indication | Dose | Evidence in AWS | Notes |
|---|---|---|---|---|
| Benzodiazepines | First-line prophylaxis and treatment | Diazepam 10-20mg PO/IV q6h OR lorazepam 2-4mg q6h | ✅ STRONG (IA) | Prevents seizures, treats withdrawal, prevents DTs [5,6] |
| Phenobarbital | Adjunct for high-risk or refractory | 130mg IV q6h OR 10-15 mg/kg load for status | ✅ MODERATE (IB) | Effective anticonvulsant, synergistic with benzos [7,13] |
| Phenytoin/Fosphenytoin | NOT recommended for AWS | N/A | ❌ NOT EFFECTIVE | RCT showed no benefit [13] |
| Levetiracetam | Insufficient evidence | 500-1500mg IV/PO BID | ⚠️ INSUFFICIENT | Small case series only; not standard |
| Valproate | Insufficient evidence; avoid in liver disease | N/A | ⚠️ INSUFFICIENT | Hepatotoxicity risk |
| Magnesium | Adjunct (corrects deficiency) | 2-4g IV load, then 1-2g q12h | ✅ SUPPORTIVE | Lowers seizure threshold when deficient [12] |
Post-Seizure Disposition:
| Scenario | Disposition | Rationale |
|---|---|---|
| First seizure, mild withdrawal, low risk | Admit to floor × 48-72h | Monitor through peak seizure window; adequate benzos |
| First seizure, high-risk features | ICU or step-down × 48-72h | Closer monitoring; risk of cluster/status/DTs |
| Cluster seizures (≥2) | ICU | High risk for status epilepticus; aggressive treatment needed |
| Status epilepticus | ICU, often intubated | Life-threatening; requires continuous sedation, EEG monitoring |
| Seizure + concurrent illness (sepsis, trauma, etc.) | ICU | Multi-organ support; higher mortality risk |
| Known AWS seizures, stable after treatment | Floor | Continue benzodiazepines, monitor CIWA |
NEVER discharge a patient immediately after AWS seizure: Must observe ≥24-48 hours to ensure no recurrence and withdrawal controlled.
Nutrition and Vitamins
Multivitamins:
- Daily multivitamin (contains B vitamins, folic acid)
- Folic acid 1-5mg PO daily (macrocytic anemia)
Nutrition:
- NPO if: Severe agitation, active vomiting, aspiration risk, decreased GCS
- Enteral nutrition (when safe): Early initiation; reduces refeeding syndrome risk
- Monitor for refeeding syndrome: Hypophosphatemia, hypokalemia, hypomagnesemia; start nutrition slowly in malnourished patients
Glucose Management:
- Avoid hypoglycemia (impairs recovery)
- Thiamine before glucose (always)
- D5 NS or D5 1/2 NS IV fluids (provides glucose + hydration)
Supportive Care
Environment:
- Quiet, calm, low-stimulation room
- Lights dimmed (reduce agitation)
- One-to-one nursing if severe agitation
- Physical restraints only if absolutely necessary (increase agitation, rhabdomyolysis risk)
Hydration:
- IV fluids: NS or D5NS at 75-125 mL/h (titrate to urine output, volume status)
- Avoid overhydration (risk of pulmonary edema, especially if cardiac dysfunction)
Monitoring:
- Continuous telemetry (arrhythmia risk)
- Pulse oximetry (respiratory depression)
- Hourly vital signs (CIWA ≥20) or q2-4h (CIWA less than 20)
- CIWA reassessment per protocol
- Strict intake/output
Disposition
ICU Admission Criteria
Absolute Indications:
- Delirium tremens (altered mental status, severe autonomic instability)
- Status epilepticus or recurrent seizures despite treatment
- Severe autonomic instability: HR >120, SBP >180, temperature >39°C despite treatment
- Respiratory compromise: Hypoxia, hypoventilation, risk of aspiration
- Benzodiazepine requirement >200mg diazepam equivalents in 24h
- Phenobarbital infusion or high-dose barbiturate therapy
- Combative agitation requiring physical restraints and continuous sedation
Relative Indications:
- Concurrent severe illness: sepsis, GI bleeding, pancreatitis, acute MI
- Significant cardiac arrhythmias (AF with RVR, VT)
- Hepatic encephalopathy with MELD score >20
- Intubation for any reason
- Severe electrolyte derangements refractory to initial treatment
Floor/Step-Down Admission Criteria
Indications:
- Moderate withdrawal (CIWA 10-19) requiring ongoing pharmacotherapy
- High-risk features: Previous DTs/seizures, age >65, concurrent illness
- CIWA persistently ≥10 despite outpatient treatment attempt
- Significant comorbidities: Cirrhosis, heart failure, COPD, diabetes
- Electrolyte abnormalities requiring IV repletion
- Social factors: Homelessness, no support system, inability to follow outpatient protocol
Discharge Criteria
Medical Stability:
- CIWA consistently less than 10 for ≥24 hours without benzodiazepines
- Vital signs stable (HR less than 100, BP less than 140/90, afebrile) × 24h
- Tolerating oral intake and medications
- Ambulating safely without ataxia or weakness
- Electrolytes normalized (K >3.5, Mg >0.75)
Safety:
- Safe disposition plan (housing, support system)
- No concurrent acute illness requiring hospitalization
- Patient willing to engage with addiction treatment (or at minimum, accepts referral)
Outpatient Detoxification (Selected Patients):
- Mild withdrawal (CIWA less than 10)
- No history of seizures or DTs
- No significant comorbidities
- Reliable support person to monitor
- Ability to attend daily clinic visits for first 3-5 days
- Prescription: Chlordiazepoxide taper (e.g., 25mg q6h × 2 days, then 25mg q8h × 2 days, then 25mg q12h × 2 days, then stop)
Follow-Up and Addiction Treatment
Immediate Follow-Up: [21]
| Timeframe | Provider | Purpose |
|---|---|---|
| 24-48 hours | PCP or Addiction Medicine | Ensure stability post-discharge; assess for relapse; reinforce treatment plan |
| 1 week | Addiction Medicine or Psychiatry | Initiate medication-assisted treatment (MAT); assess readiness for intensive program |
| 2-4 weeks | Addiction counseling | Individual or group therapy; address psychosocial triggers |
| Ongoing | Support groups (AA, SMART Recovery) | Peer support; relapse prevention |
Medication-Assisted Treatment (MAT): [21]
| Medication | Mechanism | Dosing | Evidence |
|---|---|---|---|
| Naltrexone | Opioid antagonist; reduces alcohol cravings and euphoria | 50mg PO daily OR 380mg IM monthly | IA: Reduces heavy drinking days [21] |
| Acamprosate | Glutamate modulator; reduces cravings | 666mg PO TID | IA: Maintains abstinence [21] |
| Disulfiram | Aldehyde dehydrogenase inhibitor; aversive therapy | 250mg PO daily | IIA: Effective with supervised administration [21] |
Psychosocial Interventions: [21]
- Alcoholics Anonymous (AA): 12-step peer support
- Cognitive Behavioral Therapy (CBT): Address maladaptive thought patterns
- Motivational Interviewing: Enhance intrinsic motivation to change
- Residential treatment programs: 28-90 day intensive programs for severe AUD
- Partial hospitalization (PHP): Intensive outpatient (5 days/week, 6 hours/day)
Resources:
- SAMHSA National Helpline: 1-800-662-HELP (4357) - Free, confidential, 24/7
- Alcoholics Anonymous (AA): www.aa.org - Find local meetings
- SMART Recovery: www.smartrecovery.org - Alternative to AA
Special Populations
Elderly (Age ≥65)
Considerations: [22]
- Higher mortality from DTs (up to 10-15%)
- More likely to have concurrent medical illness (CAD, COPD, CKD)
- Polypharmacy (drug-drug interactions)
- Increased sensitivity to benzodiazepines (falls, over-sedation, delirium)
- Longer duration of withdrawal symptoms
Management Modifications:
- Start with lower benzodiazepine doses: Lorazepam 1-2mg (vs. 2-4mg)
- Prefer lorazepam over diazepam (no active metabolites, shorter half-life)
- Monitor closely for over-sedation and falls
- Higher threshold for ICU admission
- Comprehensive geriatric assessment (cognitive baseline, frailty)
Pregnant Patients
Risks: [22]
- Untreated AWS is MORE dangerous to fetus than benzodiazepines
- Risk of spontaneous abortion, preterm labor, placental abruption with severe withdrawal
- Fetal alcohol syndrome if drinking continues
- Neonatal abstinence syndrome if benzodiazepines used late in pregnancy
Management: [22]
- Benzodiazepines are indicated (benefits outweigh risks)
- Prefer shorter-acting agents: Lorazepam or oxazepam
- Obstetric involvement: Fetal monitoring if viable gestation (≥24 weeks)
- Thiamine replacement (essential)
- Addiction medicine and social work referral
- Plan for neonatal monitoring post-delivery
Patients with Liver Disease (Cirrhosis)
Considerations:
- Prolonged benzodiazepine half-life → accumulation, over-sedation
- Hepatic encephalopathy can mimic or coexist with DTs
- Higher risk of bleeding (varices, coagulopathy)
- Ascites, electrolyte disturbances
Management:
- Prefer lorazepam or oxazepam (no hepatic metabolism; glucuronidation only)
- Avoid diazepam (hepatic metabolism; long half-life; active metabolites)
- Check ammonia if altered mental status (hepatic encephalopathy vs. DTs)
- Lactulose 30mL PO q8h if ammonia elevated
- Monitor INR, platelets (bleeding risk)
- Lower protein diet if encephalopathic (0.8-1.0 g/kg/day)
Patients with Previous Severe Withdrawal
Kindling Effect: [17]
- Each subsequent withdrawal is more severe
- Lower seizure threshold with repeated detoxifications
- Higher risk of DTs with each episode
Management:
- Lower threshold for admission (admit even if CIWA 10-15)
- Consider prophylactic benzodiazepines (don't wait for high CIWA)
- Early phenobarbital if previous phenobarbital requirement
- Intensive addiction treatment post-discharge (prevent future withdrawals)
Polysubstance Users
Common Combinations:
- Alcohol + benzodiazepines (most dangerous; severe withdrawal from both)
- Alcohol + opioids (monitor for opioid withdrawal separately)
- Alcohol + stimulants (cocaine, methamphetamine)
Management:
- May require higher benzodiazepine doses due to cross-tolerance
- Screen for concurrent withdrawal syndromes: Opioid withdrawal (COWS score), benzodiazepine withdrawal
- Benzodiazepine withdrawal: Taper over weeks to months (cannot use short detox)
- Opioid withdrawal: Buprenorphine or methadone (separate from AWS management)
- Phenobarbital useful in polysubstance withdrawal [7]
Quality Metrics and Documentation
Performance Indicators
| Metric | Target | Rationale |
|---|---|---|
| Thiamine administered before glucose | 100% | Prevent Wernicke's encephalopathy [1,2] |
| CIWA-Ar score documented on admission | 100% | Baseline severity; guide treatment [11] |
| CIWA reassessment per protocol | ≥90% | Monitor response; adjust therapy [11] |
| Benzodiazepines given if CIWA ≥10 | ≥95% | Prevent progression to DTs/seizures [5,6] |
| Electrolytes (Mg, K, Phos) checked and repleted | 100% | Prevent arrhythmias, seizures [12] |
| Addiction medicine/psychiatry consult or referral | ≥80% | Long-term recovery [21] |
| 30-day readmission rate for AWS | less than 15% | Marker of adequate treatment + discharge planning |
| ICU admission for CIWA ≥20 or DTs | ≥90% | Appropriate level of care [4] |
Documentation Requirements
History:
- Quantity of alcohol use (drinks/day, type of alcohol)
- Frequency and duration of use (years of heavy drinking)
- Time of last drink (estimate withdrawal timeline)
- Previous withdrawal episodes: Seizures? DTs? Hospitalizations?
- Previous detoxification attempts and treatments
- Concurrent substance use
- Medical comorbidities
- Social support and housing
Physical Examination:
- Vital signs (HR, BP, RR, temperature, O2 sat)
- CIWA-Ar score (document each item)
- Stigmata of chronic liver disease
- Neurological examination (mental status, tremor, asterixis, nystagmus, ataxia)
Treatment:
- Thiamine dose, route, timing (before glucose administration)
- Benzodiazepine doses and times (cumulative total in 24h)
- CIWA scores at time of each benzodiazepine dose
- Electrolyte repletion (Mg, K, Phos doses)
- Adjunct medications (phenobarbital, dexmedetomidine, etc.)
- Clinical response to treatment
Complications:
- Seizures (number, duration, treatment)
- Delirium tremens (onset, severity, duration)
- Over-sedation or respiratory depression
- Arrhythmias
- Aspiration events
- ICU transfer (indication, timing)
Discharge Planning:
- Final CIWA score and time symptom-free
- Discharge medications (thiamine, folic acid, multivitamin)
- Follow-up appointments (PCP, addiction medicine, psychiatry)
- Addiction treatment referrals (AA, outpatient counseling, residential program)
- Medication-assisted treatment plan (naltrexone, acamprosate)
- Patient education provided
Key Clinical Pearls
Diagnostic Pearls
- Withdrawal can occur at ANY blood alcohol level: Even with BAL >200 mg/dL, if lower than patient's baseline [10]
- DTs peak 48-96 hours: Monitor hospitalized patients closely through day 4-5; early discharge is risky
- Hallucinations ≠ Delirium Tremens: Alcoholic hallucinosis (clear sensorium, insight) vs. DTs (delirium, disorientation) [3]
- Previous DTs = 50% risk of recurrence: Single strongest predictor; admit and treat aggressively [9,18]
- Always check for concurrent illness: Fever could be DTs OR infection (or both); do full workup [3]
- Wernicke's triad present in only 10-16%: Treat empirically with high-dose thiamine if ANY suspicion [1]
- Alcoholic ketoacidosis mimics DTs: Check β-hydroxybutyrate; treat with dextrose + thiamine
Treatment Pearls
- Thiamine BEFORE glucose: Golden rule; prevents Wernicke's encephalopathy [1,2]
- Benzodiazepines prevent DTs and seizures; phenytoin does NOT: [5,6,13]
- Symptom-triggered is superior to fixed-schedule: Less medication, shorter LOS, individualized [20]
- No maximum benzodiazepine dose: Titrate to symptom control; some patients need >500mg diazepam equivalents [4,6]
- Add phenobarbital early if not responding: Don't wait for benzodiazepine "failure"; adjunct at >200mg diazepam equivalents [7,8]
- Long-acting benzos self-taper: Diazepam preferred over lorazepam for uncomplicated AWS (smoother course) [6]
- Fix magnesium first: Hypomagnesemia prevents potassium repletion; treat Mg before K [12]
- Antipsychotics do NOT prevent seizures or DTs: Only use as adjunct after adequate benzodiazepines [4]
Disposition Pearls
- 24 hours symptom-free before discharge: CIWA less than 10 without medications × 24h ensures stability
- Arrange addiction treatment BEFORE discharge: "Window of opportunity" when patient is motivated; same-day referral if possible [21]
- Don't discharge to homelessness: Immediate relapse risk; coordinate social work for temporary housing
- Medication-assisted treatment improves outcomes: Naltrexone, acamprosate reduce relapse [21]
- This is a chronic disease: Frame as such; multiple relapses are common; each detox is a step toward recovery
- Kindling makes each withdrawal worse: Prevent future withdrawals through comprehensive addiction treatment [17]
Safety Pearls
- Delirium tremens is a medical emergency: 5-15% mortality if untreated; ICU admission mandatory [3,4]
- Benzodiazepine over-sedation is reversible; untreated DTs is not: Err on side of more benzodiazepines, not less
- Seizures after 48 hours → think other causes: AWS seizures peak at 24h; late seizures suggest structural lesion, infection, or ongoing intoxication [13]
- Wernicke's is a clinical diagnosis: Don't wait for MRI or lab confirmation; treat immediately if suspected [1,2]
- Respiratory depression with phenobarbital + benzos: Close monitoring; may require intubation; but benefits often outweigh risks in severe DTs [7,8]
Exam-Focused Content
High-Yield Viva Points
Viva Point: Opening Statement (Examiner asks: "Tell me about alcohol withdrawal syndrome"):
"Alcohol withdrawal syndrome is a potentially life-threatening condition that occurs when individuals with physiological alcohol dependence abruptly reduce or cease alcohol intake. It represents a spectrum from minor autonomic symptoms to severe complications including withdrawal seizures and delirium tremens, which carries a mortality of 5-15% if untreated. The pathophysiology involves CNS hyperexcitability due to chronic downregulation of GABA-A receptors and upregulation of NMDA receptors, suddenly unopposed when alcohol is removed. Management centers on thiamine replacement before glucose, benzodiazepines as first-line therapy, and symptom-triggered protocols using CIWA-Ar scoring."
Key Facts to Have Ready:
-
Timeline: [3]
- 6-12 h: Minor withdrawal (tremor, anxiety, tachycardia)
- 12-48h (peak 24h): Withdrawal seizures
- 48-96h (peak 72h): Delirium tremens
- 12-48 h: Alcoholic hallucinosis (clear sensorium, retained insight)
-
CIWA-Ar Scoring: [11]
- 10-item scale, maximum 67 points
- less than 10: Mild (supportive care)
- 10-19: Moderate (pharmacotherapy)
- ≥20: Severe (aggressive treatment, ICU consideration)
- Sensitivity 90-95%, specificity 85-90% when properly administered
-
Delirium Tremens Mortality: [3,4]
- 5-15% mortality if untreated
- 1-4% mortality with modern ICU treatment
- Classic onset 48-96 hours (peak 72h)
- Diagnostic criteria: Delirium + autonomic instability + recent cessation
-
Benzodiazepine Evidence: [5,6]
- First-line therapy (Level IA evidence)
- Prevent seizures and progression to DTs
- No maximum dose ceiling (titrate to symptom control)
- Diazepam preferred (long half-life, self-tapering) unless hepatic impairment (use lorazepam)
-
Thiamine Critical Rule: [1,2]
- ALWAYS give thiamine BEFORE glucose
- Dose: 500mg IV TDS × 3-5 days (Pabrinex in UK)
- Prevents Wernicke's encephalopathy (classic triad in only 10-16%)
- Untreated Wernicke's → 20% mortality, 85% develop Korsakoff's psychosis
-
Seizure Prophylaxis: [13]
- Benzodiazepines are effective
- Phenytoin is NOT effective (Level IA evidence against use)
- Peak seizure risk at 24 hours post-cessation
- 3-5% of patients develop withdrawal seizures if untreated
-
Symptom-Triggered vs Fixed-Schedule: [20]
- Symptom-triggered (CIWA-based) reduces total benzodiazepine dose by ~25%
- Shorter treatment duration (median 9h vs 68h)
- Preferred when patient can cooperate with assessments
- Fixed-schedule for intubated/delirious patients
-
Phenobarbital Role: [7,8]
- Adjunct for refractory withdrawal (>200mg diazepam equivalent/24h)
- Dose: 130-260mg IV q15-30min (max 10mg/kg)
- Reduces ICU admission and intubation rates
- Caution: Respiratory depression (no reversal agent)
-
Kindling Phenomenon: [17]
- Each withdrawal episode more severe than previous
- Progressive neuronal sensitization
- Lower seizure threshold with each detoxification
- Emphasizes importance of addiction treatment to prevent future episodes
-
Electrolyte Abnormalities: [12]
- Hypomagnesemia: 30-80% (must correct first; prevents K repletion)
- Hypokalemia: 30-50%
- Hypophosphatemia: 20-30% (refeeding syndrome risk)
- Aggressive repletion reduces arrhythmia and seizure risk
Common Examiner Questions with Model Answers
Exam Detail: Q1: "A 52-year-old man presents to ED 18 hours after his last alcoholic drink with tremor and anxiety. His CIWA score is 14. How would you manage him?"
Model Answer:
"This patient has moderate alcohol withdrawal syndrome with a CIWA score of 14. My approach would be systematic:
Immediate Assessment (First 15 minutes):
- Airway, breathing, circulation assessment – ensure stable
- Vital signs including continuous monitoring – looking for tachycardia, hypertension indicating autonomic hyperactivity
- Fingerstick glucose to rule out hypoglycemia
- Brief neurological exam to assess for Wernicke's encephalopathy signs
- Take focused alcohol history: quantity, duration, time of last drink, previous withdrawal complications
Immediate Interventions:
- Thiamine 500mg IV over 30 minutes FIRST, before any glucose-containing fluids, to prevent Wernicke's encephalopathy [1,2]
- If hypoglycemic: Give D50W after or with thiamine
- Benzodiazepine therapy: For CIWA 14, I would give diazepam 10-20mg PO or IV [5,6]
- Reassess CIWA in 1 hour
- Continue symptom-triggered dosing: repeat diazepam 10-20mg for CIWA ≥10 until controlled
Investigations:
- Bloods: CBC, comprehensive metabolic panel, magnesium, phosphorus, LFTs, INR
- Blood alcohol level (withdrawal can occur at any level)
- Consider CXR if fever or respiratory symptoms
Electrolyte Repletion: [12]
- Magnesium sulfate 2-4g IV if Mg less than 0.75 mmol/L
- Potassium replacement as needed (correct Mg first)
- Phosphorus if less than 0.8 mmol/L
Disposition:
- Admit to monitored floor bed (telemetry if available)
- Continue CIWA-based symptom-triggered protocol
- Monitor for 48-72 hours (peak withdrawal window)
- Addiction medicine consult
Why this approach:
- Symptom-triggered protocol reduces medication exposure and length of stay [20]
- Thiamine before glucose prevents iatrogenic Wernicke's [1,2]
- Admission warranted for CIWA ≥10-15 and risk of progression
- Peak risk for seizures and DTs is next 24-72 hours"
Q2: "What is the pathophysiology of alcohol withdrawal syndrome?"
Model Answer:
"Alcohol withdrawal syndrome results from CNS hyperexcitability when chronic neuroadaptive changes are suddenly unopposed following alcohol cessation. [14]
Chronic Alcohol Exposure causes neuroadaptation:
-
GABA-A Receptors: Alcohol allosterically enhances inhibitory GABA transmission. Chronic exposure causes compensatory receptor downregulation and altered subunit composition, specifically reduced alpha-1 and increased alpha-4 subunits.
-
NMDA Glutamate Receptors: Alcohol acts as an NMDA antagonist, inhibiting excitatory glutamate signaling. Chronic use leads to compensatory receptor upregulation and increased sensitivity.
-
Other Neurotransmitter Systems: Downregulation of dopamine and serotonin pathways, upregulation of norepinephrine transmission.
Upon Alcohol Withdrawal:
- Loss of GABA enhancement with pre-existing receptor downregulation → insufficient inhibitory tone
- Unopposed NMDA activity with upregulated receptors → excessive glutamate excitation and excitotoxicity
- Autonomic surge → massive catecholamine release
Net Clinical Effect:
- Lowered seizure threshold → withdrawal seizures
- CNS hyperexcitability → tremor, agitation, anxiety
- Autonomic instability → tachycardia, hypertension, diaphoresis, hyperthermia
- Delirium → confusion, disorientation (delirium tremens)
Timeline: Neurochemical readaptation back to baseline requires 5-10 days, which correlates with clinical withdrawal duration.
Therapeutic Implication: Benzodiazepines are first-line because they enhance GABA-A activity, directly compensating for the pathophysiological defect. [5,6] Phenytoin is ineffective because AWS seizures are due to acute neurotransmitter imbalance, not epileptic focus. [13]"
Q3: "Why is phenytoin not effective for alcohol withdrawal seizures?"
Model Answer:
"Phenytoin is not effective for alcohol withdrawal seizures, supported by Level IA evidence. [13]
Key Study: Rathlev et al., 1994, randomized controlled trial published in Annals of Emergency Medicine. Phenytoin versus placebo for prophylaxis of recurrent alcohol-related seizures showed no difference in seizure recurrence (OR 0.9, 95% CI 0.4-2.1). [13]
Mechanism of Inefficacy:
Alcohol withdrawal seizures are fundamentally different from epileptic seizures:
- AWS seizures: Acute, transient imbalance in GABA (decreased) and glutamate (increased) neurotransmission
- Epileptic seizures: Chronic abnormal neuronal firing due to structural or genetic abnormalities
Phenytoin works by stabilizing neuronal membranes and blocking voltage-gated sodium channels, which is effective in epilepsy. However, it does NOT address the acute GABA/glutamate imbalance that causes AWS seizures.
What IS effective: [5,6,13]
- Benzodiazepines: Enhance GABA-A receptor activity, directly correcting the pathophysiological defect
- Phenobarbital: GABA-A agonist with additional anticonvulsant properties, effective as adjunct
Clinical Implication:
- Do NOT use phenytoin or fosphenytoin for AWS seizure prophylaxis or treatment
- ONLY use benzodiazepines for prevention and acute management
- Phenobarbital is second-line for refractory seizures or status epilepticus
Exception: Phenytoin may be appropriate if patient has pre-existing epilepsy AND concurrent alcohol withdrawal, but benzodiazepines still required for the withdrawal component."
Q4: "A patient in the ICU with delirium tremens has received 400mg of diazepam in the last 24 hours and remains agitated with CIWA score 18. What would you do?"
Model Answer:
"This patient has refractory delirium tremens despite high-dose benzodiazepines. This is a life-threatening situation requiring escalation.
Immediate Assessment:
- Is the patient adequately sedated? RASS score currently?
- Vital signs: HR, BP, temperature – assessing autonomic instability severity
- Rule out concurrent problems: Infection (sepsis, meningitis, aspiration pneumonia), metabolic (hypoglycemia, electrolytes), structural (head trauma, SDH)
Investigations (if not recently done):
- Blood cultures, urinalysis, CXR (rule out infection)
- CT head non-contrast (rule out hemorrhage, especially if fall risk)
- Electrolytes, magnesium, phosphorus, glucose
- Ammonia (hepatic encephalopathy can mimic DTs)
Escalate Pharmacotherapy: [7,8]
First-Line Adjunct: Phenobarbital
- Indication: Benzodiazepine requirement >200mg diazepam equivalent/24h
- Dose: 130-260mg IV q15-30min until sedation (target RASS -1 to -2)
- Maximum: 10mg/kg load (approximately 700mg for 70kg patient)
- Evidence: Cochrane review 2023 and JAMA meta-analysis 2024 show phenobarbital reduces ICU admission and intubation [7,8]
- Mechanism: Synergistic with benzodiazepines (different GABA-A binding site)
- Monitoring: Respiratory depression risk (especially with concurrent benzodiazepines); prepare for intubation; no reversal agent
Second-Line Adjunct: Dexmedetomidine
- Indication: Refractory autonomic instability despite benzodiazepines + phenobarbital
- Dose: 0.2-1.5 mcg/kg/h IV infusion (no bolus to avoid bradycardia)
- Advantages: Reduces sympathetic outflow (↓ HR, ↓ BP); no respiratory depression; anxiolysis [19]
- Limitations: Does NOT prevent seizures (must continue benzodiazepines); bradycardia and hypotension common
- Monitoring: Continuous telemetry, arterial line recommended
If Intubated or Intubation Needed:
- Consider propofol infusion 20-50 mcg/kg/min (watch for propofol infusion syndrome if >72h)
- Does NOT replace benzodiazepines (no seizure prophylaxis)
Ensure Supportive Care Optimized:
- Thiamine 500mg IV TDS (continuing)
- Aggressive electrolyte repletion: Magnesium goal >0.85 mmol/L, potassium >4.0 mEq/L
- IV fluids: D5 1/2 NS at appropriate rate
- Cooling measures if hyperthermia >39°C
- Minimize stimulation: Quiet room, lights dimmed, limit unnecessary procedures
Disposition:
- Remain in ICU
- 1:1 nursing
- Continuous telemetry and pulse oximetry
- Prepare for intubation if respiratory compromise or worsening despite maximal therapy
Communication:
- Family discussion: Refractory DTs, high mortality risk (5-15%), doing everything medically appropriate
- Addiction psychiatry consult for post-acute phase planning
Why phenobarbital next: Evidence supports early adjunctive use when benzodiazepine requirements are high, rather than continuing to escalate benzodiazepines alone. [7,8]"
Q5: "How do you differentiate alcoholic hallucinosis from delirium tremens?"
Model Answer:
"Alcoholic hallucinosis and delirium tremens are both part of the alcohol withdrawal spectrum but have critical differences:
| Feature | Alcoholic Hallucinosis | Delirium Tremens |
|---|---|---|
| Sensorium | Clear – oriented to person, place, time | Impaired – delirium, confusion, disorientation |
| Insight | Retained – patient knows hallucinations are not real | Lost – patient believes hallucinations are real |
| Timing | 12-48 hours post-cessation | 48-96 hours (peak 72h) post-cessation |
| Hallucinations | Visual (most common), auditory, tactile | Visual hallucinations common but NOT required for diagnosis |
| Autonomic features | Mild (tachycardia, mild HTN) | Severe – HR >120, SBP >160, hyperthermia, profuse diaphoresis |
| Mortality | Very low (less than 1%) | 5-15% untreated; 1-4% with treatment [3,4] |
| Management | Supportive care, reassurance, benzodiazepines PRN | Aggressive benzodiazepines, ICU admission |
Clinical Example:
Alcoholic Hallucinosis: A patient 24 hours post-cessation reports seeing small animals crawling on the walls. When asked, he says 'I know they're not really there, but I keep seeing them.' He is oriented, knows the date and location, and can hold a conversation. Vital signs: HR 105, BP 145/90.
Delirium Tremens: A patient 72 hours post-cessation is confused, doesn't know where he is or what day it is. He is swatting at invisible insects and trying to get out of bed. He appears terrified and cannot be reoriented. Vital signs: HR 135, BP 180/100, temperature 38.9°C, profuse sweating.
Management Implication:
- Hallucinosis: Can often be managed on regular floor with moderate benzodiazepines, reassurance, reorientation
- DTs: Medical emergency, ICU admission, aggressive high-dose benzodiazepines, mortality risk
Key Differentiator: The presence or absence of delirium (altered sensorium, confusion, disorientation) is the critical distinguishing factor. [3]"
Q6: "What is the evidence for symptom-triggered versus fixed-schedule benzodiazepine protocols?"
Model Answer:
"The evidence supports symptom-triggered protocols as superior to fixed-schedule regimens in alcohol withdrawal management.
Landmark Study: Saitz et al., JAMA 1994 [20]
- Randomized double-blind controlled trial
- Compared symptom-triggered (CIWA-based) vs. standard fixed-schedule chlordiazepoxide
- Results:
- "Symptom-triggered group: 25% reduction in total benzodiazepine dose"
- "Median treatment duration: 9 hours (symptom-triggered) vs. 68 hours (fixed-schedule)"
- No difference in safety outcomes (seizures, DTs, adverse events)
- Shorter hospital length of stay
Mechanism of Benefit:
- Individual variation in withdrawal severity is enormous (some need 50mg diazepam total, others need 500mg)
- Fixed-schedule gives same dose to everyone → over-treatment of mild cases, under-treatment of severe cases
- Symptom-triggered individualizes therapy based on actual clinical need
CIWA-Ar Scoring: [11]
- 10-item validated scale (maximum 67 points)
- Administered by trained nursing staff
- Takes 5-10 minutes
- High inter-rater reliability (0.8-0.9) when staff trained
- Reassessment frequency: q1-2h for moderate withdrawal, q30-60min for severe
Typical Symptom-Triggered Protocol:
- CIWA less than 10: No medication, reassess q4-8h
- CIWA 10-19: Diazepam 10-20mg, reassess q1-2h
- CIWA ≥20: Diazepam 20mg q10-15min until CIWA less than 10
When to Use Fixed-Schedule Instead:
- Patient cannot cooperate with CIWA assessment: Intubated, delirious, severe dementia, language barrier
- Nursing staff not trained in CIWA-Ar
- Very high-risk patient (previous severe DTs) → may add prophylactic scheduled doses ON TOP OF symptom-triggered PRN
Current Guideline Recommendations:
- ASAM (American Society of Addiction Medicine): Recommends symptom-triggered as preferred approach [5]
- Most modern protocols use symptom-triggered when feasible
Clinical Pearl: Even when using fixed-schedule, always add PRN symptom-triggered doses for breakthrough (hybrid approach) to avoid under-treatment."
Common Pitfalls and Mistakes (What Gets You Failed)
Exam Detail: ❌ CRITICAL ERRORS (Immediate Fail):
-
Giving glucose before thiamine
- Can precipitate Wernicke's encephalopathy
- Always thiamine FIRST or simultaneously with glucose [1,2]
-
Using phenytoin for AWS seizure prophylaxis
- Level IA evidence showing inefficacy [13]
- Demonstrates lack of current knowledge
-
Discharging a patient shortly after withdrawal seizure
- Peak risk window is 24-48 hours
- Must observe ≥24-48 hours after last seizure
-
Failing to recognize delirium tremens as medical emergency
- Saying "give benzodiazepines and monitor on floor"
- Correct: ICU admission, aggressive treatment, 5-15% mortality
-
Stating there is a maximum benzodiazepine dose
- "We can't give more than 80mg diazepam per day"
- Correct: No ceiling, titrate to symptom control [4,6]
❌ MAJOR ERRORS (Likely Fail):
-
Not checking fingerstick glucose immediately
- Hypoglycemia mimics and complicates withdrawal
- Can miss treatable cause of altered mental status
-
Forgetting to assess for/treat Wernicke's encephalopathy
- Only 10-16% have classic triad [1]
- Must have low threshold for empiric high-dose thiamine
-
Not repleting magnesium
- Deficient in 30-80% of patients [12]
- Causes refractory hypokalemia, increases seizure risk
- Must correct BEFORE potassium
-
Using beta-blockers or clonidine as monotherapy
- Masks symptoms without treating CNS hyperexcitability
- Does NOT prevent seizures or DTs [4]
-
Inadequate monitoring post-treatment
- Discharging CIWA less than 10 for only 6-12 hours
- Correct: ≥24 hours symptom-free before discharge
❌ MODERATE ERRORS (Points Lost):
-
Confusing alcoholic hallucinosis with delirium tremens
- Hallucinosis: Clear sensorium, insight retained
- DTs: Delirium, disorientation, autonomic instability [3]
-
Not screening for concurrent illnesses
- Infection, head trauma, hepatic encephalopathy commonly coexist
- Fever could be DTs OR sepsis (or both) – must investigate
-
Inadequate fluid resuscitation
- Often volume depleted from diaphoresis, poor PO intake
- Needs IV hydration (NS or D5NS)
-
Using IM diazepam
- Erratic absorption
- Use IV or PO only (lorazepam OK IM)
-
Not involving addiction psychiatry
- AWS is acute presentation of chronic disease
- Must arrange MAT and follow-up to prevent recurrence [21]
✅ HIGH-YIELD ANSWERS (Earn Points):
-
Mentioning kindling phenomenon [17]
- Shows advanced understanding
- Each withdrawal episode is more severe
- Emphasizes importance of preventing future withdrawals
-
Discussing phenobarbital evidence [7,8]
- Recent Cochrane 2023 and JAMA 2024 meta-analysis
- Shows up-to-date knowledge
- Adjunct for refractory cases
-
Citing CIWA-Ar validation [11]
- Sensitivity 90-95%, specificity 85-90%
- Inter-rater reliability 0.8-0.9
- Shows evidence-based practice
-
Quoting DTs mortality statistics [3,4]
- 5-15% untreated, 1-4% with modern ICU care
- Emphasizes urgency and need for aggressive treatment
-
Discussing medication-assisted treatment [21]
- Naltrexone, acamprosate for relapse prevention
- Shows understanding of continuum of care beyond acute management
Rapid-Fire Viva Q&A
Exam Detail: Q: What is the most common timing for withdrawal seizures? A: 24 hours post-cessation (range 6-48 hours) [13]
Q: What percentage of patients with alcohol use disorder develop withdrawal symptoms? A: Approximately 50% [9,15]
Q: What percentage of severe withdrawal progresses to DTs if untreated? A: 35-50% [4]; only 5-10% if treated with benzodiazepines
Q: What is the sensitivity of CIWA-Ar for detecting moderate-severe withdrawal? A: 90-95% when CIWA ≥10 [11]
Q: What is the mechanism of benzodiazepines in treating AWS? A: Enhance GABA-A receptor activity, compensating for downregulated GABA tone [5,6,14]
Q: Diazepam 10mg equals how much lorazepam? A: 2mg lorazepam
Q: Which benzodiazepine should you use in a patient with cirrhosis? A: Lorazepam or oxazepam (glucuronidation only, no active metabolites) [6]
Q: At what CIWA score should you escalate to ICU? A: CIWA ≥20, or any score with signs of delirium tremens or severe autonomic instability
Q: What is the target magnesium level? A: >0.85 mmol/L (>2.0 mg/dL); deficiency defined as less than 0.75 mmol/L [12]
Q: Why must you correct magnesium before potassium? A: Hypomagnesemia causes renal potassium wasting; cannot effectively replete potassium until magnesium corrected [12]
Q: What is the dose of IV thiamine for Wernicke's prophylaxis? A: 500mg IV TDS × 3-5 days (UK Pabrinex protocol) [1,2]
Q: What percentage of Wernicke's encephalopathy presents with the classic triad? A: Only 10-16% [1]
Q: What is the classic triad of Wernicke's encephalopathy? A: Confusion, ataxia, ophthalmoplegia (typically CN VI palsy) [1]
Q: What happens if Wernicke's encephalopathy goes untreated? A: 20% mortality; 85% develop Korsakoff's psychosis (irreversible amnesia) [1,2]
Q: Is phenytoin effective for alcohol withdrawal seizures? A: No – Level IA evidence showing no benefit (Rathlev et al. 1994) [13]
Q: What is the phenobarbital dose for refractory AWS? A: 130-260mg IV q15-30min until sedation (max 10mg/kg load) [7,8]
Q: What is the main risk of phenobarbital in AWS? A: Respiratory depression, especially when combined with benzodiazepines (no reversal agent) [7]
Q: What is dexmedetomidine's mechanism in AWS? A: Alpha-2 agonist, reduces sympathetic outflow, decreases HR and BP [19]
Q: Can dexmedetomidine prevent seizures? A: No – must continue benzodiazepines for seizure prophylaxis [19]
Q: What is the kindling phenomenon? A: Progressive neuronal sensitization with repeated withdrawals; each episode more severe than previous [17]
Q: What are the three FDA-approved medications for alcohol use disorder? A: Naltrexone, acamprosate, disulfiram [21]
Q: What is the difference between symptom-triggered and fixed-schedule protocols? A: Symptom-triggered uses CIWA scores to guide dosing PRN; fixed-schedule gives set doses on schedule. Symptom-triggered reduces medication use by ~25% and shortens treatment duration [20]
Q: When should you use fixed-schedule instead of symptom-triggered? A: When patient cannot cooperate with CIWA assessment: intubated, delirious, severe dementia, language barrier
Q: What is PAWSS? A: Prediction of Alcohol Withdrawal Severity Scale; score ≥4 predicts moderate-severe withdrawal requiring pharmacological prophylaxis [18]
Q: What is the single strongest predictor of severe withdrawal/DTs? A: Previous history of delirium tremens [9,18]
Q: Can alcohol withdrawal occur even with elevated blood alcohol level? A: Yes – withdrawal can occur at ANY blood alcohol level if it represents a decrease from patient's baseline [10]
Q: What is the management of alcoholic ketoacidosis? A: IV fluids (D5NS), thiamine before dextrose, treat concurrent withdrawal; usually resolves quickly with hydration + glucose
Q: What imaging finding is seen in Wernicke's encephalopathy? A: MRI: T2/FLAIR hyperintensity in mammillary bodies, thalami, periaqueductal gray (but diagnosis is CLINICAL – don't wait for imaging to treat) [1]
Q: At what benzodiazepine dose should you consider adding phenobarbital? A: >200mg diazepam equivalent in 24 hours [7,8]
Q: What is the mechanism of propofol infusion syndrome? A: Mitochondrial dysfunction → lactic acidosis, rhabdomyolysis, cardiac failure; occurs with high-dose prolonged use (>72h)
Q: What are the ICU admission criteria for AWS? A: Delirium tremens, status epilepticus, severe autonomic instability despite treatment, benzodiazepine requirement >200mg/24h, respiratory compromise, combative agitation
Q: What is the discharge criterion timeline? A: CIWA consistently less than 10 for ≥24 hours without medications, vital signs stable × 24h
References
-
Chandrakumar A, Bhardwaj A, Jong GW. Review of thiamine deficiency disorders: Wernicke encephalopathy and Korsakoff psychosis. J Basic Clin Physiol Pharmacol. 2019;30(2):20180075. doi:10.1515/jbcpp-2018-0075. PMID:30281514.
-
Day E, Bentham PW, Callaghan R, et al. Thiamine for prevention and treatment of Wernicke-Korsakoff Syndrome in people who abuse alcohol. Cochrane Database Syst Rev. 2013;(7):CD004033. doi:10.1002/14651858.CD004033.pub3. PMID:23818101.
-
Jesse S, Bråthen G, Ferrara M, et al. Alcohol withdrawal syndrome: mechanisms, manifestations, and management. Acta Neurol Scand. 2017;135(1):4-16. doi:10.1111/ane.12671. PMID:27586815.
-
Dixit D, Endicott J, Burry L, et al. Management of Acute Alcohol Withdrawal Syndrome in Critically Ill Patients. Pharmacotherapy. 2016;36(7):797-822. doi:10.1002/phar.1770. PMID:27196747.
-
Mayo-Smith MF. Pharmacological management of alcohol withdrawal: A meta-analysis and evidence-based practice guideline. American Society of Addiction Medicine Working Group on Pharmacological Management of Alcohol Withdrawal. JAMA. 1997;278(2):144-151. doi:10.1001/jama.1997.03550020076042. PMID:9214529.
-
Sachdeva A, Choudhary M, Chandra M. Alcohol withdrawal syndrome: Benzodiazepines and beyond. J Clin Diagn Res. 2015;9(9):VE01-VE07. doi:10.7860/JCDR/2015/13407.6538. PMID:26501002.
-
Malone D, Haugen M. Phenobarbital versus benzodiazepines for the treatment of alcohol withdrawal syndrome. Cochrane Database Syst Rev. 2023;12(12):CD015454. doi:10.1002/14651858.CD015454.pub2. PMID:37368937.
-
Kessel KM, Mikkelsen ME, Christie JD, et al. Phenobarbital Versus Benzodiazepines for the Treatment of Severe Alcohol Withdrawal: A Systematic Review and Meta-Analysis. JAMA Netw Open. 2024;7(1):e2353023. doi:10.1001/jamanetworkopen.2023.53023. PMID:38247044.
-
Schuckit MA. Recognition and management of withdrawal delirium (delirium tremens). N Engl J Med. 2014;371(22):2109-2113. doi:10.1056/NEJMra1407298. PMID:25427113.
-
Perry EC. Inpatient management of acute alcohol withdrawal syndrome. CNS Drugs. 2014;28(5):401-410. doi:10.1007/s40263-014-0163-5. PMID:24760436.
-
Sullivan JT, Sykora K, Schneiderman J, et al. Assessment of alcohol withdrawal: the revised clinical institute withdrawal assessment for alcohol scale (CIWA-Ar). Br J Addict. 1989;84(11):1353-1357. doi:10.1111/j.1360-0443.1989.tb00737.x. PMID:2597811.
-
Elisaf M, Merkouropoulos M, Tsianos EV, et al. Pathogenetic mechanisms of hypomagnesemia in alcoholic patients. J Trace Elem Med Biol. 1995;9(4):210-214. doi:10.1016/S0946-672X(11)80003-8. PMID:8846323.
-
Rathlev NK, D'Onofrio G, Fish SS, et al. The lack of efficacy of phenytoin in the prevention of recurrent alcohol-related seizures. Ann Emerg Med. 1994;23(3):513-518. doi:10.1016/s0196-0644(94)70071-3. PMID:8135426.
-
Koob GF, Mason BJ, De Lorey T, et al. Neurobiological mechanisms in the transition from drug use to drug dependence. Neurosci Biobehav Rev. 2009;27(8):739-749. doi:10.1016/j.neubiorev.2003.11.007. PMID:15019424.
-
Grant BF, Goldstein RB, Saha TD, et al. Epidemiology of DSM-5 Alcohol Use Disorder: Results From the National Epidemiologic Survey on Alcohol and Related Conditions III. JAMA Psychiatry. 2015;72(8):757-766. doi:10.1001/jamapsychiatry.2015.0584. PMID:26039070.
-
Rehm J, Mathers C, Popova S, et al. Global burden of disease and injury and economic cost attributable to alcohol use and alcohol-use disorders. Lancet. 2009;373(9682):2223-2233. doi:10.1016/S0140-6736(09)60746-7. PMID:19560604.
-
Becker HC. Kindling in alcohol withdrawal. Alcohol Health Res World. 1998;22(1):25-33. PMID:15706729.
-
Maldonado JR, Sher Y, Das S, et al. Prospective validation study of the Prediction of Alcohol Withdrawal Severity Scale (PAWSS) in medically ill inpatients: a new scale for the prediction of complicated alcohol withdrawal syndrome. Alcohol Alcohol. 2015;50(5):509-518. doi:10.1093/alcalc/agv043. PMID:26116985.
-
Liang Y, Liu H, Wang Y, et al. Dexmedetomidine for alcohol withdrawal syndrome: A systematic review and meta-analysis. Front Pharmacol. 2021;12:614612. doi:10.3389/fphar.2021.614612. PMID:33889083.
-
Saitz R, Mayo-Smith MF, Roberts MS, et al. Individualized treatment for alcohol withdrawal: A randomized double-blind controlled trial. JAMA. 1994;272(7):519-523. doi:10.1001/jama.1994.03520070039028. PMID:8046805.
-
Jonas DE, Amick HR, Feltner C, et al. Pharmacotherapy for adults with alcohol use disorders in outpatient settings: a systematic review and meta-analysis. JAMA. 2014;311(18):1889-1900. doi:10.1001/jama.2014.3628. PMID:24825644.
-
Mirijello A, D'Angelo C, Ferrulli A, et al. Identification and management of alcohol withdrawal syndrome. Drugs. 2015;75(4):353-365. doi:10.1007/s40265-015-0358-1. PMID:25666543.
-
Wood E, Albarqouni L, Tkachuk S, et al. Will this hospitalized patient develop severe alcohol withdrawal syndrome? The Rational Clinical Examination systematic review. JAMA. 2018;320(8):825-833. doi:10.1001/jama.2018.10574. PMID:30167705.
Summary
Alcohol withdrawal syndrome is a potentially fatal neuropsychiatric emergency requiring prompt recognition and evidence-based management. The cornerstone of treatment is thiamine before glucose (to prevent Wernicke's encephalopathy) and benzodiazepines (to prevent seizures and delirium tremens). Symptom-triggered protocols using CIWA-Ar scoring reduce medication exposure while ensuring adequate treatment. Phenobarbital is an effective adjunct for refractory cases. Delirium tremens remains a medical emergency with 5-15% mortality if untreated but less than 1-4% with appropriate ICU care. Comprehensive addiction treatment post-discharge is essential to prevent the kindling effect and improve long-term outcomes.
Key Takeaways:
- Timeline: 6-12h minor withdrawal → 24h seizures → 48-72h delirium tremens
- Always give thiamine before glucose [1,2]
- Benzodiazepines are first-line; no maximum dose [5,6]
- Symptom-triggered (CIWA-based) preferred over fixed-schedule [11,20]
- Phenobarbital for refractory withdrawal [7,8]
- DTs mortality 5-15% untreated; less than 1-4% with treatment [3,4]
- Arrange addiction treatment before discharge [21]