Anorexia Nervosa (Adult)
Anorexia nervosa (AN) is a severe psychiatric disorder characterized by persistent restriction of energy intake leading to significantly low body weight (BMI less than 17.5 kg/m² in adults), an intense fear of gaining...
What matters first
Anorexia nervosa (AN) is a severe psychiatric disorder characterized by persistent restriction of energy intake leading to significantly low body weight (BMI less than 17.5 kg/m² in adults), an intense fear of gaining...
Severe electrolyte imbalance (hypokalaemia less than 3.0 mmol/L)
7 Jan 2026
Generated educational material; verify before clinical use.
Visible references section
See the concept before reading it
Study the key anatomy, imaging, and decision pathways as full teaching plates.
Clinical board
A visual summary of the highest-yield teaching signals on this page.
Urgent signals
Safety-critical features pulled from the topic metadata.
- Severe electrolyte imbalance (hypokalaemia less than 3.0 mmol/L)
- Bradycardia (HR less than 40 bpm) or significant arrhythmia
- Refeeding syndrome risk (phosphate less than 0.5 mmol/L)
- Severe malnutrition (BMI less than 15 kg/m2)
Linked comparisons
Differentials and adjacent topics worth opening next.
- Bulimia Nervosa
- Avoidant/Restrictive Food Intake Disorder (ARFID)
Content status and exam context
This page is AI-generated educational content. It may contain errors or omissions and is not a substitute for current guidelines, local protocols, senior clinical judgement, or professional medical advice.
MedVellum does not claim an individual clinician reviewer, board certification, or professional credential for this page unless a future version names a real, verifiable reviewer.
Clinical explanation and evidence
Anorexia Nervosa (Adult)
1. Topic Overview
Summary
Anorexia nervosa (AN) is a severe psychiatric disorder characterized by persistent restriction of energy intake leading to significantly low body weight (BMI less than 17.5 kg/m² in adults), an intense fear of gaining weight or becoming fat, and a profound disturbance in the perception of body weight or shape. [1,2] It carries the highest mortality rate of any psychiatric disorder, with standardized mortality ratios ranging from 5.0 to 10.0 over 10-year follow-up periods. [3] Death occurs primarily from medical complications of starvation (particularly cardiac arrhythmias) and suicide, which accounts for approximately 20% of deaths. [3,4]
The disorder typically begins in adolescence but can persist into adulthood or develop de novo in adult life. Adult-onset anorexia nervosa (age > 18 years at first presentation) accounts for approximately 20-25% of cases and often carries a poorer prognosis due to delayed recognition and chronicity. [4]
Treatment requires a multidisciplinary approach combining medical stabilization, nutritional rehabilitation, and evidence-based psychological interventions. Enhanced Cognitive Behavioral Therapy (CBT-E) is first-line for adults, while Family-Based Therapy (FBT) remains superior for adolescents transitioning to adult services. [5,6] Refeeding syndrome represents the most dangerous acute complication during nutritional restoration, necessitating careful electrolyte monitoring and gradual caloric advancement. [7,8]
Key Facts
- Diagnostic Criteria: Restriction of energy intake → significantly low body weight (BMI less than 17.5 kg/m²) + intense fear of weight gain + body image disturbance with lack of recognition of the seriousness of current low body weight [1,2]
- Lifetime Prevalence: 0.9-2.0% in females; 0.2-0.3% in males (female:male ratio ~10:1) [9]
- Mortality Rate: Standardized mortality ratio (SMR) 5.0-10.0; approximately 5-10% die within 10 years of diagnosis [3]
- Subtypes:
- "Restricting type: Weight loss achieved through dieting, fasting, excessive exercise (no regular binge-purge)"
- "Binge-eating/Purging type: Regular engagement in binge eating or purging (vomiting, laxatives, diuretics)"
- Severity Classification (DSM-5): Based on BMI for adults:
- "Mild: BMI ≥17 kg/m²"
- "Moderate: BMI 16-16.99 kg/m²"
- "Severe: BMI 15-15.99 kg/m²"
- "Extreme: BMI less than 15 kg/m² [1]"
- Peak Age of Onset: Bimodal distribution at 14-15 years and 18-19 years; adult-onset (> 18 years) represents 20-25% of cases [9]
Clinical Pearls
"Refeeding Syndrome is the Silent Killer": The most dangerous phase of treatment is the first 7-10 days of nutritional rehabilitation. Monitor phosphate, potassium, and magnesium daily. Hypophosphataemia (less than 0.5 mmol/L) can cause respiratory failure, rhabdomyolysis, and cardiac arrest. Always supplement thiamine (200-300 mg/day) before initiating feeding. [7,8]
"MARSIPAN Criteria Define Medical Emergency": Management of Really Sick Patients with Anorexia Nervosa (MARSIPAN) guidelines specify immediate medical admission criteria: BMI less than 13 kg/m², rapid weight loss (> 1 kg/week sustained), HR less than 40 bpm, systolic BP less than 90 mmHg, hypoglycaemia, hypothermia (less than 35°C), or acute psychiatric risk. [10]
"BMI is Necessary but Not Sufficient": A patient with BMI 18 kg/m² who has lost 30% of body weight in 3 months is medically unstable. Rate of weight loss and physiological parameters (HR, BP, temperature, electrolytes) matter more than absolute BMI in determining admission. [10]
"The Minnesota Starvation Experiment Explains Everything": Many psychological symptoms (obsession with food, hoarding, ritualistic eating, social withdrawal, depression, cognitive rigidity) are direct consequences of semi-starvation, not primary psychiatric pathology. Weight restoration partially reverses these features. [11]
"Mortality is a Marathon, Not a Sprint": While acute medical complications are dramatic, the majority of excess mortality occurs over decades. Long-term follow-up studies show SMR peaks at 10-20 years after diagnosis. Chronic medical sequelae (osteoporosis, cardiac dysfunction) and suicide drive long-term mortality. [3,4]
Why This Matters Clinically
Anorexia nervosa is a medical emergency masquerading as a psychiatric disorder. While the core psychopathology (body image disturbance, fear of weight gain) is psychiatric, the immediate threats to life are medical: cardiac arrhythmias from electrolyte derangement, sudden cardiac death from QTc prolongation, refeeding syndrome during nutritional rehabilitation, and hypoglycaemic episodes. [1,10]
Adult patients presenting to general medical services often have prolonged illness duration (mean 5-7 years from onset to first specialist contact), extensive medical complications, and poor insight. Recognition in non-psychiatric settings is critical. The SCOFF questionnaire (Sick, Control, One stone, Fat, Food) has 78-100% sensitivity for detecting eating disorders in primary care. [12]
Timely intervention improves outcomes: 50-70% of patients achieve full recovery with appropriate treatment, but this drops to 20-30% in chronic cases (> 7 years duration). Early engagement with specialist eating disorder services, aggressive nutritional rehabilitation, and evidence-based psychological therapy are paramount. [4,13]
2. Epidemiology
Prevalence and Incidence
| Measure | Value | Notes |
|---|---|---|
| Lifetime Prevalence (Females) | 0.9-2.0% | Higher in Western industrialized nations [9] |
| Lifetime Prevalence (Males) | 0.2-0.3% | Likely underestimated due to help-seeking bias [9] |
| Annual Incidence (Females) | 8-13 per 100,000 person-years | Peak in 15-19 age group [9] |
| Annual Incidence (Males) | 1-2 per 100,000 person-years | Increasing over recent decades [9] |
| Point Prevalence | 0.3-0.5% | Among women aged 15-40 years |
| Adult-Onset AN (> 18 years) | 20-25% of all cases | Associated with poorer prognosis [4] |
Age and Gender Distribution
Peak Age of Onset:
- Primary peak: 14-15 years (early-mid adolescence)
- Secondary peak: 18-19 years (transition to university/independence)
- Adult-onset: Increasingly recognized; distinct clinical features (more comorbidity, longer duration to treatment, higher chronicity) [4]
Gender Ratio:
- Female:Male = 10:1 in community samples
- Male:Female = 1:3-4 in clinical samples (males underrepresented)
- Male cases often present later and with greater severity [9]
Mortality and Morbidity
All-Cause Mortality:
- Standardized Mortality Ratio (SMR): 5.0-10.0 (compared to age-matched general population) [3]
- Crude mortality rate: 5-10% per decade of illness
- 20-year mortality: Approximately 15-20%
Causes of Death: [3,4]
| Cause | Proportion |
|---|---|
| Suicide | 20-25% |
| Cardiac complications | 30-40% |
| Infection/sepsis | 10-15% |
| Multi-organ failure | 10-15% |
| Other/unknown | 15-25% |
Prognostic Factors Affecting Mortality:
- Longer illness duration (> 7 years)
- Very low BMI (less than 13 kg/m²)
- Binge-purge subtype (higher impulsivity → suicide risk)
- Comorbid substance use disorder
- Poor social support
- Older age at presentation
Geographic and Cultural Variation
Anorexia nervosa was historically considered a "culture-bound syndrome" of Western societies, but recent evidence demonstrates global distribution. [9] Incidence rates are highest in:
- High-income Western countries (North America, Western Europe, Australia)
- Urbanized Asian populations (Japan, South Korea, Singapore)
- Cultures with high exposure to Western media ideals
Protective factors in traditional non-Western cultures are eroding with globalization and social media exposure.
Risk Factors
Genetic and Biological
| Factor | Evidence | Heritability |
|---|---|---|
| Family History | First-degree relatives have 11-fold increased risk | 50-75% [14] |
| Twin Studies | Monozygotic concordance 50-60%; Dizygotic 5-10% | -- |
| GWAS Findings | 8 genome-wide significant loci identified; metabolic-psychiatric overlap | -- [14] |
| Temperament | Perfectionism, obsessive-compulsive traits, harm avoidance | Partially heritable |
Recent genome-wide association studies (GWAS) identify anorexia nervosa as a "metabo-psychiatric" disorder with genetic correlations to both psychiatric conditions (OCD, anxiety, schizophrenia) and metabolic traits (lipid metabolism, insulin resistance). [14]
Psychological
- Personality Traits: Perfectionism, rigidity, obsessionality, low self-esteem, anxiety sensitivity
- Trauma: Childhood sexual abuse (OR 2.2-3.4), physical abuse, emotional neglect
- Comorbid Psychiatric Disorders:
- Anxiety disorders (50-75% comorbidity)
- Obsessive-compulsive disorder (25-40%)
- Major depressive disorder (30-50%)
- Autism spectrum traits (elevated in 20-30%)
Sociocultural
| Risk Factor | Details |
|---|---|
| Occupations | Ballet dancers (6-7% prevalence), models, elite athletes, gymnastics, wrestling |
| Peer Dieting | Social contagion effects in adolescent groups |
| Social Media | Image-focused platforms (Instagram) associated with body dissatisfaction |
| Family Environment | Parental dieting, comments about weight/shape, enmeshment |
| Life Transitions | University entry, leaving home, relationship breakdown |
Precipitating Factors
- Dieting (often initially "healthy eating")
- Weight-related teasing or bullying
- Puberty and bodily changes
- Illness with weight loss (reinforcing)
- Sport/activity requiring weight control
3. Aetiology and Pathophysiology
Biopsychosocial Model
Anorexia nervosa arises from complex interactions between genetic vulnerability, neurobiological alterations, psychological traits, and sociocultural pressures. No single factor is necessary or sufficient.
Predisposing → Precipitating → Perpetuating
| Phase | Factors |
|---|---|
| Predisposing | Genetic vulnerability (50-75% heritability), perfectionism, anxiety traits, family history |
| Precipitating | Dieting, weight-related teasing, life stress, puberty |
| Perpetuating | Starvation physiology, cognitive distortions, reinforcement from weight loss, family accommodation |
Neurobiology
Exam Detail: Serotonergic Dysfunction: Altered 5-HT1A and 5-HT2A receptor binding in cortical and limbic regions persists after weight restoration, suggesting trait markers. Increased 5-HT activity may drive anxiety, which patients reduce through food restriction (temporary anxiolytic effect). [15]
Dopaminergic Alterations: Reduced dopamine D2/D3 receptor binding in ventral striatum correlates with reduced reward sensitivity. Patients experience food as less rewarding ("wanting" without "liking"), perpetuating restriction. [15]
Structural Brain Changes:
- Acute starvation: Global grey matter volume reduction (10-15%), white matter volume reduction (5-10%). Ventricular enlargement. Partially reversible with weight restoration but may leave residual deficits. [16]
- Persistent changes: Reduced insula volume (interoceptive awareness), altered anterior cingulate cortex (cognitive control), reduced hippocampal volume (HPA axis dysregulation)
Cognitive Neuroscience:
- Set-shifting deficits: Impaired cognitive flexibility (linked to OCD traits)
- Central coherence: Tendency toward detail-focused processing at expense of global picture
- Interoceptive deficits: Poor recognition of internal states (hunger, satiety, emotions)
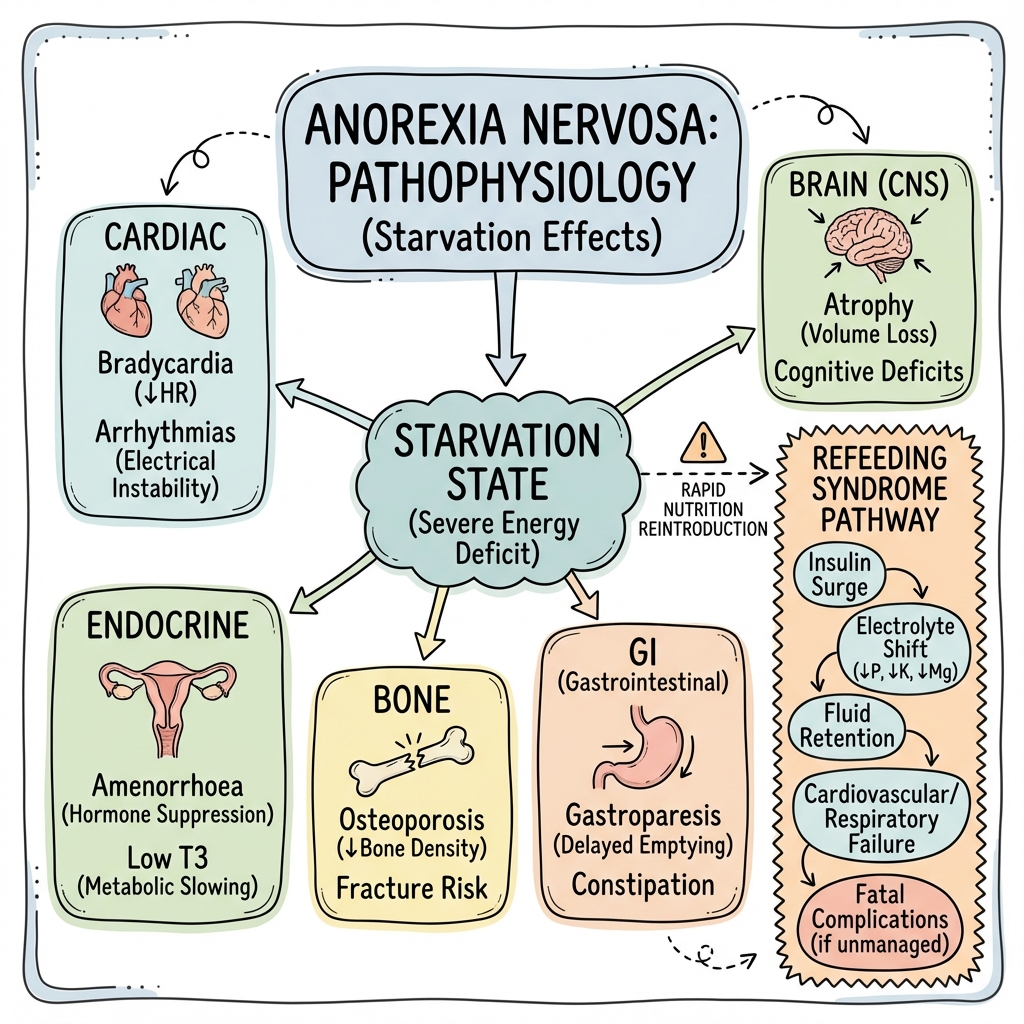
Endocrine and Metabolic Consequences
The endocrine changes in anorexia nervosa represent adaptive responses to starvation (energy conservation) but become maladaptive with chronicity.
| System | Acute Changes | Chronic Sequelae |
|---|---|---|
| Hypothalamic-Pituitary-Gonadal (HPG) | ↓GnRH pulsatility → ↓LH, FSH → Hypogonadotropic hypogonadism → Amenorrhoea | Infertility, osteoporosis (irreversible bone loss) |
| Hypothalamic-Pituitary-Thyroid (HPT) | ↓T3 (Low T3 syndrome/Euthyroid sick syndrome), normal/low TSH, normal T4 | Energy conservation; reversed with refeeding |
| Hypothalamic-Pituitary-Adrenal (HPA) | ↑Cortisol (loss of circadian rhythm), impaired dexamethasone suppression | Hypercortisolism → bone loss, muscle wasting, mood effects |
| Growth Hormone/IGF-1 | ↑GH (GH resistance state), ↓IGF-1 | Impaired linear growth (if pre-epiphyseal closure), reduced bone formation |
| Leptin | Profoundly ↓ (signals starvation to brain) | Drives amenorrhoea, HPA activation, bone loss |
| Ghrelin | ↑ (orexigenic signal) | Paradoxically elevated despite restriction (ghrelin resistance?) |
Osteoporosis and Bone Health: Anorexia nervosa causes rapid, profound bone loss affecting both trabecular and cortical bone. Mechanisms:
- Hypogonadism (↓estrogen/testosterone) → ↑osteoclast activity
- Hypercortisolism → ↓osteoblast function
- ↓IGF-1 → impaired bone formation
- ↓leptin → direct effects on bone metabolism
- Malnutrition → inadequate calcium, vitamin D, protein
Critical: Adolescent-onset AN during peak bone mass accrual (ages 12-18) causes irreversible deficits. Z-scores of -2.0 to -3.0 are common, with lifetime fracture risk increased 3-7 fold. [17]
Cardiovascular Pathophysiology
Cardiac complications are the leading medical cause of death in AN. [18]
Structural Changes:
- Cardiac atrophy: Left ventricular mass reduction (15-25%), chamber size reduction
- Myocardial fibrosis: Microstructural changes on cardiac MRI
- Pericardial effusion: Common in severe malnutrition (20-40% prevalence)
- Mitral valve prolapse: Occurs secondary to chamber size reduction
Functional Changes:
- Bradycardia: Vagal predominance; HR 40-50 bpm common (30s during sleep)
- Hypotension: Systolic BP 80-90 mmHg typical; postural drop > 20 mmHg
- QTc prolongation: Corrected QT > 450-500 ms increases torsades de pointes risk
- Arrhythmias: Sinus bradycardia, sinus arrhythmia, AV block, ventricular ectopy
Electrolyte-Driven Arrhythmias:
| Electrolyte | Mechanism | ECG Changes |
|---|---|---|
| Hypokalaemia | K+ less than 3.0 mmol/L (purging, diuretics, laxatives) | Flattened T waves, U waves, QTc prolongation, VT/VF risk |
| Hypomagnesaemia | Often coexists with hypokalaemia | Potentiates QTc prolongation |
| Hypophosphataemia | Refeeding syndrome | Cardiac dysfunction, arrhythmias, respiratory failure |
Refeeding Syndrome: Pathophysiology
Refeeding syndrome occurs when nutritional intake is rapidly increased after prolonged starvation. The shift from catabolic to anabolic metabolism causes: [7,8]
- Insulin surge → Intracellular shift of phosphate, potassium, magnesium
- Severe hypophosphataemia (less than 0.5 mmol/L):
- ↓ATP production → cellular dysfunction
- Respiratory failure (diaphragm weakness)
- Cardiac dysfunction (↓contractility, arrhythmias)
- Rhabdomyolysis
- Haemolysis
- Seizures
- Thiamine deficiency → Wernicke's encephalopathy risk (ophthalmoplegia, ataxia, confusion)
- Fluid overload → Heart failure, peripheral oedema
High-Risk Features:
- BMI less than 14 kg/m²
- Negligible intake for > 10 days
- Weight loss > 15% in 3-6 months
- Baseline hypokalaemia, hypomagnesaemia, hypophosphataemia
- History of alcohol misuse
4. Clinical Presentation
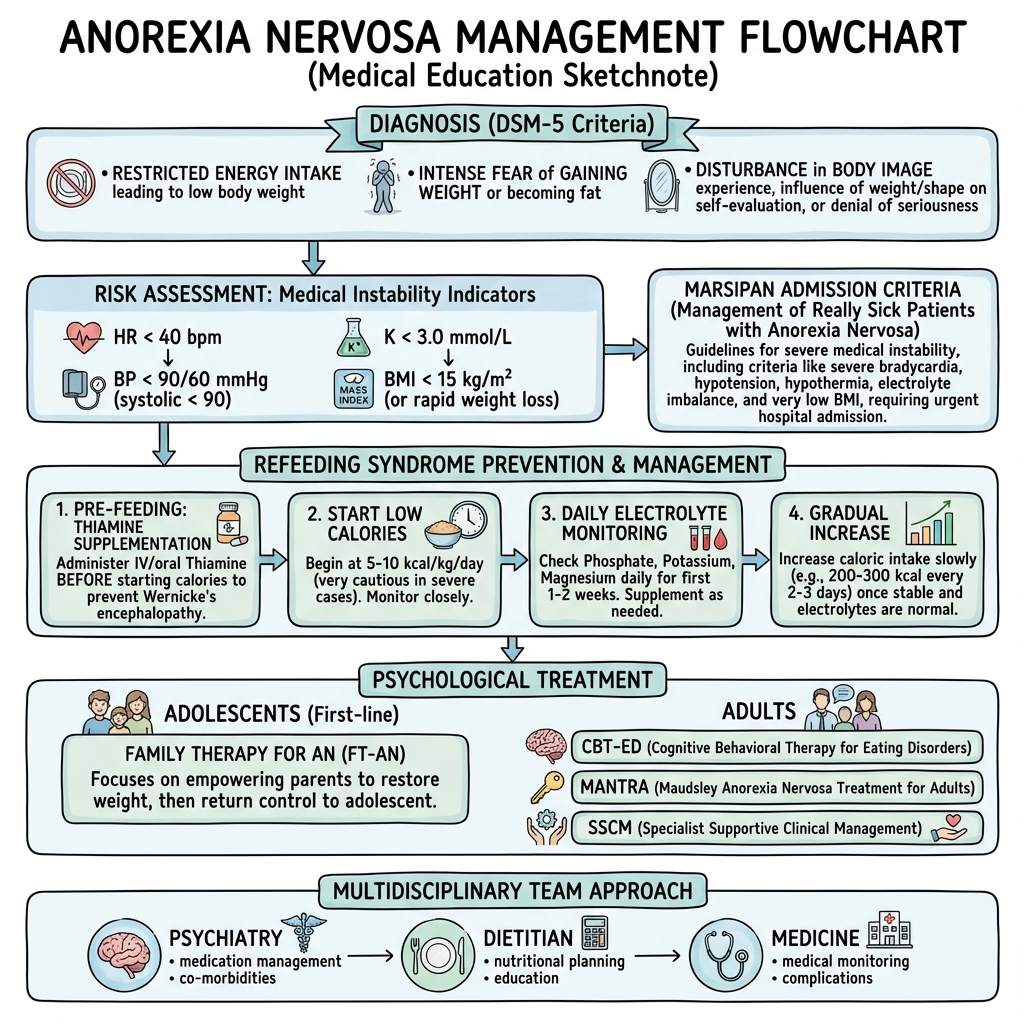
Cardinal Features (Diagnostic Criteria - DSM-5)
A. Restriction of Energy Intake:
- Persistent restriction relative to requirements
- Leading to significantly low body weight (BMI less than 17.5 kg/m² in adults, or less than 5th percentile for age/sex)
B. Intense Fear of Weight Gain:
- Intense fear of gaining weight or becoming fat
- OR persistent behavior that interferes with weight gain (despite already being underweight)
C. Disturbance in Body Image:
- Disturbance in the way one's body weight or shape is experienced
- Undue influence of body weight/shape on self-evaluation
- Persistent lack of recognition of the seriousness of current low body weight
Specify Type:
- Restricting type: No regular binge-eating or purging in past 3 months
- Binge-eating/Purging type: Regular binge-eating or purging (self-induced vomiting, laxatives, diuretics, enemas)
Specify Current Severity (Adults):
- Mild: BMI ≥17 kg/m²
- Moderate: BMI 16-16.99 kg/m²
- Severe: BMI 15-15.99 kg/m²
- Extreme: BMI less than 15 kg/m²
[1,2]
Behavioral and Psychological Symptoms
Eating Behaviors:
- Severe dietary restriction (calories less than 800-1000 kcal/day)
- Rigid food rules (avoidance of fats, carbohydrates, "fear foods")
- Ritualistic eating (cutting food into tiny pieces, eating extremely slowly, specific utensils)
- Hiding, hoarding, or discarding food
- Eating alone to avoid scrutiny
- Preoccupation with food, recipes, cooking for others (while not eating)
Body Image and Weight Preoccupation:
- Persistent feeling of being "fat" despite emaciation
- Intense fear of specific body parts (abdomen, thighs, buttocks)
- Frequent body checking (mirror, weighing, measuring)
- Avoidance of body exposure (baggy clothes, avoidance of swimming/changing rooms)
- Overvaluation of weight/shape for self-worth
Compulsive Exercise:
- Excessive, driven exercise despite fatigue, injury, or medical advice
- Severe distress if unable to exercise
- Exercise to "compensate" for eating
- Often secretive (exercising at night, in private)
Psychosocial:
- Social withdrawal and isolation
- Loss of interest in previously enjoyed activities
- Irritability, mood lability
- Anxiety (especially around meal times)
- Obsessive-compulsive behaviors (beyond food)
- Perfectionism and rigidity
- Poor concentration and memory (starvation effect)
Physical Symptoms
Patients may deny or minimize symptoms; collateral history from family is crucial.
Common Physical Complaints:
- Profound fatigue and weakness
- Dizziness, presyncope, syncope (orthostatic hypotension)
- Cold intolerance (hypothermia, poor peripheral perfusion)
- Constipation (delayed gastric emptying, reduced GI motility)
- Amenorrhoea (primary or secondary)
- Sleep disturbance
- Hair loss (scalp), increased body hair (lanugo)
- Dry skin
- Poor wound healing
- Easy bruising
Red Flag Symptoms Requiring Urgent Assessment:
- Syncope or presyncope
- Chest pain or palpitations
- Severe weakness or inability to stand
- Confusion or altered mental status
- Muscle cramps or tetany
- Polyuria (diabetes insipidus from chronic hypovolaemia)
5. Clinical Examination
General Inspection
| Observation | Findings in AN |
|---|---|
| Body Habitus | Severe emaciation, cachexia; prominent bones (clavicles, ribs, scapulae, pelvis) |
| Clothing | Multiple layers (cold intolerance), baggy clothes (concealing weight loss) |
| Grooming | Often well-groomed (high-functioning); occasionally unkempt in severe depression |
| Affect | Flat, anxious, irritable; may appear euthymic with ego-syntonic illness |
| Behavior | Restlessness, fidgeting (compulsive activity); occasionally withdrawn |
Vital Signs (CRITICALLY IMPORTANT)
The most important examination findings in AN are objective vital signs and anthropometry.
Weight and BMI:
- Weigh in light clothing after voiding
- Calculate BMI = Weight (kg) / Height (m)²
- CRITICAL: Serial weights (weekly) to track trajectory
Heart Rate:
- Resting HR less than 60 bpm is bradycardia (expected in AN)
- RED FLAG: HR less than 40 bpm → Medical admission indicated (MARSIPAN criteria) [10]
- Check rhythm (sinus bradycardia vs AV block)
Blood Pressure:
- Hypotension common (systolic 80-90 mmHg)
- Postural BP (supine → standing after 3 min):
- Postural drop > 20 mmHg systolic or > 10 mmHg diastolic = orthostatic hypotension
- "RED FLAG: Symptomatic postural drop → Medical admission"
Temperature:
- Core temperature less than 36°C common
- RED FLAG: less than 35°C = Hypothermia → Medical admission [10]
Capillary Refill Time:
-
2 seconds indicates poor peripheral perfusion
Cardiovascular Examination
- Inspection: Peripheral cyanosis, mottled skin (acrocyanosis)
- Palpation: Apex beat may be difficult to palpate (small heart)
- Auscultation:
- Bradycardia
- Soft heart sounds (reduced cardiac mass)
- Pericardial rub (if pericardial effusion)
- Mitral valve prolapse murmur (late systolic click, mid-late systolic murmur)
Dermatological Signs
| Sign | Description | Significance |
|---|---|---|
| Lanugo | Fine, downy hair on back, arms, face | Starvation adaptation (insulation) |
| Dry Skin | Xerosis, scaling | Dehydration, malnutrition |
| Hair Loss | Telogen effluvium (scalp) | Protein-energy malnutrition |
| Carotenaemia | Yellow-orange discoloration (palms, soles) | Excess carotene intake, impaired conversion to vitamin A |
| Russell's Sign | Calluses/scars on dorsum of hands (knuckles) | Self-induced vomiting (teeth trauma to knuckles) - Binge/Purge subtype |
| Petechiae/Bruising | Easy bruising, petechiae | Thrombocytopenia, vitamin C/K deficiency |
Gastrointestinal Examination
- Inspection: Scaphoid abdomen (loss of subcutaneous and visceral fat)
- Palpation:
- Tender abdomen (constipation)
- Palpable faecal loading (chronic constipation)
- Auscultation: Reduced bowel sounds (ileus in severe cases)
Dental Examination (if Binge/Purge Subtype):
- Enamel erosion (particularly palatal surfaces of upper incisors) from gastric acid
- Dental caries
- Parotid gland enlargement (bilateral, non-tender sialadenosis)
Musculoskeletal Examination
- Muscle Wasting: Temporalis, deltoids, quadriceps, gluteals
- Proximal Myopathy:
- "SUSS Test: Sit Up from lying (without using arms), Squat down and Stand up"
- Inability to perform SUSS indicates severe muscle wasting → Medical instability [10]
- Bone Tenderness: Suggestive of fractures (high risk with osteoporosis)
Neurological Examination
- Peripheral Neuropathy: Distal sensory loss (stocking-glove), reduced ankle reflexes (vitamin B12, thiamine deficiency)
- Cognitive Assessment:
- Concentration and memory (often impaired by starvation)
- Confusion or altered consciousness (hypoglycaemia, Wernicke's encephalopathy)
Mental State Examination
| Domain | Findings |
|---|---|
| Appearance | Emaciated, layered clothing, may appear younger than stated age |
| Behavior | Anxious, restless, or withdrawn; may be overly compliant or defensive |
| Speech | Normal rate and rhythm; content dominated by food/weight themes |
| Mood | Subjectively low or anxious; objectively flat or irritable |
| Affect | Restricted, anxious |
| Thought Form | Obsessional, perseverative around food/weight |
| Thought Content | Overvalued ideas about weight/shape (ego-syntonic); fear of fatness; poor insight into illness severity |
| Perception | Body image distortion (seeing self as larger than reality) |
| Cognition | Impaired concentration, memory (starvation effect); cognitive rigidity |
| Insight | Often poor (does not recognize low weight as problematic); partial insight in some cases |
| Risk | Assess suicidal ideation (elevated risk); self-harm history |
Red Flags Requiring Immediate Medical Admission (MARSIPAN Criteria)
[!CAUTION] Medical Emergency Indicators (Require Urgent Hospital Admission): [10]
Cardiovascular:
- Bradycardia: HR less than 40 bpm
- Hypotension: Systolic BP less than 90 mmHg
- Postural tachycardia: Rise in HR > 20 bpm on standing
- QTc prolongation: > 450 ms (males), > 470 ms (females), or > 500 ms (any)
Metabolic:
- Hypoglycaemia: Glucose less than 3.0 mmol/L
- Hypothermia: Core temperature less than 35°C
- Severe electrolyte disturbance: K+ less than 3.0 mmol/L, Na+ less than 130 mmol/L, Phosphate less than 0.5 mmol/L
Anthropometric:
- BMI less than 13 kg/m² (high risk) or less than 15 kg/m² with rapid weight loss
- Rapid weight loss: > 1 kg/week sustained
Functional:
- SUSS test failure (unable to sit up from lying, squat and stand without using arms)
- Unable to mobilize safely
Psychiatric:
- Acute suicidal intent or plan
- Severe comorbid psychiatric disorder requiring admission
6. Investigations
Objectives of Investigations
- Assess medical stability and identify life-threatening complications
- Screen for refeeding syndrome risk
- Identify end-organ damage from malnutrition
- Exclude differential diagnoses (hyperthyroidism, malignancy, inflammatory bowel disease)
- Establish baseline for monitoring during refeeding
First-Line Investigations (All Patients)
| Test | Purpose | Expected Findings in AN | Red Flags |
|---|---|---|---|
| FBC | Screen for cytopenias | Normocytic anaemia, leukopenia (WCC 2-4), thrombocytopenia | Severe pancytopenia |
| U&E | Electrolytes, renal function | ↓K+ (purging), ↑Urea (dehydration), ↑Creatinine (chronic dehydration) | K+ less than 3.0, Na+ less than 130 |
| Bone Profile | Calcium, phosphate, magnesium | Often normal initially; ↓Phosphate suggests refeeding risk | Phosphate less than 0.5 mmol/L |
| Magnesium | Refeeding risk | Low-normal or ↓ | Mg less than 0.5 mmol/L |
| Glucose | Hypoglycaemia risk | May be low-normal (3.5-4.5 mmol/L) | Glucose less than 3.0 mmol/L |
| LFTs | Hepatic function | ↑Transaminases (refeeding, steatosis), ↓Albumin (severe malnutrition) | ALT/AST > 3x ULN |
| TFTs | Exclude hyperthyroidism, assess HPT axis | ↓T3 (Low T3 syndrome), normal/low TSH, normal T4 | ↑T4, ↑TSH (hyperthyroidism) |
| ECG | Cardiac rhythm, QTc | Sinus bradycardia, QTc prolongation, low voltage, U waves | QTc > 500 ms, VT, AV block |
| Urinalysis | Renal, hydration status | Specific gravity often high (dehydration); exclude glycosuria | Ketonuria (starvation ketosis) |
Haematological Abnormalities
Anaemia (Normocytic):
- Mechanism: Bone marrow suppression from malnutrition
- Rarely severe; Hb typically 10-12 g/dL
Leukopenia:
- WCC 2.0-4.0 × 10⁹/L common
- Relative lymphocytosis
- NOT a sign of infection (often misinterpreted)
Thrombocytopenia:
- Platelets 100-150 × 10⁹/L
- Rarely causes bleeding
Reversibility: All cytopenias resolve with nutritional rehabilitation.
Electrolyte Disturbances
| Electrolyte | Restrictive Type | Binge/Purge Type |
|---|---|---|
| Sodium | Often normal; ↑ if dehydrated | ↓ (hyponatraemia from water loading, SIADH) |
| Potassium | Usually normal | ↓↓ (Hypokalaemia from vomiting, laxatives, diuretics) - DANGEROUS |
| Chloride | Normal | ↓ (hypochloraemia from vomiting) |
| Bicarbonate | Normal | ↑ (metabolic alkalosis from vomiting) |
| Phosphate | Normal until refeeding | ↓ (refeeding syndrome risk) |
| Magnesium | Low-normal | ↓ (purging, refeeding) |
Hypokalaemia (less than 3.0 mmol/L) is a MEDICAL EMERGENCY:
- Causes: Vomiting, laxative abuse, diuretic abuse
- Complications: Cardiac arrhythmias (VT/VF), muscle weakness, rhabdomyolysis, ileus
- Management: Potassium replacement (oral preferred; IV if less than 2.5 mmol/L or ECG changes)
Endocrine Investigations
Thyroid Function:
- Low T3 syndrome: ↓T3, normal T4, normal/low TSH
- Adaptive response to starvation (reduce metabolic rate)
- DO NOT treat with levothyroxine (resolves with refeeding)
Reproductive Hormones:
- ↓LH, FSH (hypogonadotropic hypogonadism)
- ↓Estradiol (females), ↓Testosterone (males)
- Not routinely required for diagnosis
Cortisol:
- Elevated (loss of circadian variation)
- Impaired dexamethasone suppression
- Mimics Cushing's syndrome ("pseudo-Cushing's")
Bone Density (DEXA Scan)
Indications:
- All patients with AN > 6 months duration
- Postmenopausal women
- Men with hypogonadism
- History of fractures
Interpretation:
- Use Z-score (comparison to age-matched controls) in premenopausal women and men less than 50 years
- Z-score ≤ -2.0 = "Below expected range for age"
- T-score used in postmenopausal women (T-score ≤ -2.5 = Osteoporosis)
Findings:
- Lumbar spine and femoral neck Z-scores commonly -2.0 to -3.0
- Trabecular bone (spine) more affected than cortical bone initially
- Partial reversibility with weight restoration (30-60% recovery of bone density)
[17]
Cardiac Investigations
ECG (12-Lead):
- Mandatory in all patients
- Findings:
- Sinus bradycardia (HR 40-60 bpm)
- Low voltage (small heart)
- QTc prolongation (> 450 ms)
- U waves (hypokalaemia)
- ST-T wave changes
- AV block (first-degree common)
Echocardiography:
- Indications: Symptomatic (chest pain, palpitations, syncope), ECG abnormalities, refeeding monitoring
- Findings:
- Reduced left ventricular mass and chamber size
- Pericardial effusion (20-40% of severe cases)
- Mitral valve prolapse (10-20%)
- Reduced ejection fraction (advanced cases)
24-Hour Holter Monitoring:
- If symptoms of arrhythmia (palpitations, syncope)
- Detect nocturnal bradycardia (less than 30 bpm during sleep is common but may be pathological if less than 25 bpm)
Refeeding Monitoring (During Nutritional Rehabilitation)
CRITICAL PROTOCOL for First 7-10 Days of Refeeding: [7,8]
Daily Monitoring:
- Weight (daily for first week)
- Vital signs (HR, BP, temperature) - TDS-QDS
- Fluid balance
- ECG (if baseline QTc prolonged or symptomatic)
Daily Bloods (First 3-7 Days):
- Electrolytes: Phosphate, potassium, magnesium (daily for 3-7 days)
- Glucose: Monitor for hypoglycaemia
- Renal Function: U&E
Supplementation Protocol:
- Thiamine: 200-300 mg PO daily (or 100 mg IV if unable to absorb) - MUST precede feeding
- Multivitamin with minerals: Include vitamin B complex, vitamin C
- Phosphate: Oral supplementation if less than 0.8 mmol/L (Phosphate Sandoz 2-3 sachets/day)
- Potassium: Target K+ > 3.5 mmol/L (oral Sando-K or IV if less than 2.5 mmol/L)
- Magnesium: Target Mg > 0.75 mmol/L (oral magnesium glycerophosphate)
When to Suspect Refeeding Syndrome:
- Phosphate less than 0.5 mmol/L (CRITICAL)
- Hypokalaemia or hypomagnesaemia developing during refeeding
- New peripheral oedema or fluid overload
- Confusion, weakness, arrhythmias
Differential Diagnosis Investigations
If atypical features or diagnostic uncertainty:
| Condition | Investigation |
|---|---|
| Hyperthyroidism | TFTs (↑T4, ↑T3, ↓TSH) |
| Addison's Disease | 9 am cortisol, short Synacthen test |
| Malignancy | CXR, CT chest/abdomen/pelvis, tumour markers (if indicated) |
| Inflammatory Bowel Disease | Faecal calprotectin, colonoscopy, inflammatory markers (CRP, ESR) |
| Coeliac Disease | Tissue transglutaminase antibodies (tTG-IgA), total IgA |
| Diabetes Mellitus (Type 1) | Glucose, HbA1c, urinalysis (glycosuria, ketonuria) |
7. Diagnosis and Classification
DSM-5 Diagnostic Criteria
See Section 4: Clinical Presentation - Cardinal Features for full DSM-5 criteria. [1,2]
Subtypes
Restricting Type:
- During the last 3 months, NO regular binge-eating or purging behavior
- Weight loss achieved through dieting, fasting, and/or excessive exercise
Binge-Eating/Purging Type:
- During the last 3 months, regular engagement in binge-eating or purging behavior (self-induced vomiting, misuse of laxatives, diuretics, enemas)
Clinical Implications:
- Binge/Purge type has higher impulsivity, comorbid substance use, self-harm, and suicide risk
- Greater electrolyte disturbances (hypokalaemia)
- Often crossover between subtypes over illness course (30-50% transition)
Severity Specifiers (Based on BMI for Adults)
| Severity | BMI (kg/m²) | Clinical Implications |
|---|---|---|
| Mild | ≥17 | Outpatient management usually appropriate |
| Moderate | 16-16.99 | Intensive outpatient or day program; consider admission if unstable |
| Severe | 15-15.99 | Often requires inpatient medical stabilization |
| Extreme | less than 15 | Medical emergency; inpatient admission mandatory |
Important Caveats:
- Severity is not solely determined by BMI
- Rate of weight loss, physiological instability, and psychiatric risk override BMI
- A patient with BMI 18 who has lost 25% body weight in 8 weeks is high-risk
Screening Tools
SCOFF Questionnaire (Primary Care): [12]
- Do you make yourself Sick because you feel uncomfortably full?
- Do you worry you have lost Control over how much you eat?
- Have you recently lost more than One stone (6.35 kg/14 lbs) in a 3-month period?
- Do you believe yourself to be Fat when others say you are too thin?
- Would you say that Food dominates your life?
Scoring: ≥2 "Yes" responses → Sensitivity 78-100%, Specificity 87-90% for eating disorders
Eating Disorder Examination Questionnaire (EDE-Q):
- 28-item self-report measure
- Assesses core eating disorder psychopathology (restraint, eating concern, shape concern, weight concern)
- Used in specialist services for baseline and monitoring
Differential Diagnosis
| Condition | Distinguishing Features |
|---|---|
| Bulimia Nervosa | BMI usually > 17.5; recurrent binge-purge cycles with lack of control; less body weight disturbance |
| Avoidant/Restrictive Food Intake Disorder (ARFID) | Food restriction NOT driven by body image concerns; no fear of weight gain; often sensory sensitivities or fear of choking |
| Body Dysmorphic Disorder | Preoccupation with perceived flaw in appearance (not solely weight/shape); no extreme weight loss |
| Major Depressive Disorder | Weight loss from reduced appetite/anhedonia; no fear of weight gain or body image disturbance; recognizes weight loss as undesirable |
| Obsessive-Compulsive Disorder | Food rituals may overlap, but not driven by weight/shape concerns |
| Hyperthyroidism | Weight loss despite normal/increased appetite; tachycardia (not bradycardia); ↑T4, ↓TSH |
| Inflammatory Bowel Disease (Crohn's, UC) | Weight loss from malabsorption, diarrhea, abdominal pain; raised inflammatory markers; colonoscopy findings |
| Coeliac Disease | Weight loss, diarrhea, bloating; positive tTG antibodies; villous atrophy on biopsy |
| Malignancy | Weight loss with systemic symptoms (night sweats, fever); no body image disturbance; imaging/biopsy findings |
| Type 1 Diabetes (Poorly Controlled) | Weight loss, polyuria, polydipsia; hyperglycaemia, glycosuria, ketonuria |
| Addison's Disease | Weight loss, hyperpigmentation, hypotension, hyponatraemia, hyperkalaemia; low cortisol |
8. Management
Multidisciplinary Team Approach
Effective treatment of anorexia nervosa requires coordination across medical, psychiatric, nutritional, and psychosocial domains.
Core Team:
- Eating Disorder Psychiatrist (Lead clinician)
- Clinical Psychologist/Therapist (Psychological therapy delivery)
- Specialist Dietitian (Meal planning, nutritional rehabilitation)
- Eating Disorder Physician/General Physician (Medical monitoring)
- Mental Health Nurse (Case coordination, support)
- Family Therapist (If FBT indicated)
- Occupational Therapist (Functional rehabilitation)
Extended Team (as needed):
- Endocrinologist (bone health, amenorrhoea)
- Cardiologist (arrhythmias, structural cardiac disease)
- Gastroenterologist (refeeding complications, severe GI symptoms)
- Paediatrician (adolescent-adult transition)
Treatment Settings
| Setting | Indications | Components |
|---|---|---|
| Inpatient Medical | BMI less than 13-15, rapid weight loss, medical instability (MARSIPAN criteria), failed outpatient, acute suicide risk | Medical stabilization, refeeding, intensive monitoring, NG feeding if required |
| Inpatient Psychiatric | Severe psychiatric comorbidity, high suicide risk, extreme behaviors | Psychiatric stabilization, commence psychological therapy, meal support |
| Day Program/Partial Hospitalization | BMI 15-17, medically stable, requires intensive support | Supervised meals, group therapy, individual therapy, medical monitoring 5 days/week |
| Intensive Outpatient | BMI 16-18, medically stable, motivated | Individual therapy 2-3x/week, dietitian weekly, medical monitoring fortnightly-monthly |
| Outpatient | BMI > 17, medically stable, good motivation and support | Individual therapy weekly, dietitian fortnightly-monthly, medical monitoring monthly |
Medical Stabilization and Nutritional Rehabilitation
Goals:
- Restore weight to healthy range (target BMI 18.5-20 kg/m² initially)
- Correct medical complications
- Prevent refeeding syndrome
- Establish regular, balanced eating pattern
Refeeding Protocol
Exam Detail: Starting Caloric Intake (MARSIPAN 2022): [10]
-
Traditional "Start Low, Go Slow":
- "Very high risk (BMI less than 13, rapid loss): Start 5-10 kcal/kg/day (500-800 kcal/day)"
- "High risk (BMI 13-15): Start 10-20 kcal/kg/day (800-1200 kcal/day)"
- Increase by 200-300 kcal every 3-5 days
-
Higher Calorie Refeeding (Emerging Evidence):
- Recent studies suggest starting 1200-1500 kcal/day with intensive monitoring may be safe and reduce admission length
- Requires expert setting with daily electrolyte monitoring
- Still controversial; not yet standard practice
Target Caloric Intake:
- Weight gain phase: 2000-3000 kcal/day (average 2500 kcal/day)
- Weight maintenance: 1800-2500 kcal/day
Target Weight Gain:
- Inpatient: 0.5-1.0 kg/week (average 0.7 kg/week)
- Outpatient: 0.2-0.5 kg/week
Meal Plan Structure:
- 3 main meals + 2-3 snacks
- Balanced macronutrients (carbohydrate 50-60%, fat 25-35%, protein 15-20%)
- Gradual reintroduction of "fear foods"
- Structured eating times (reduce anxiety)
Supplementation:
- Thiamine: 200-300 mg PO daily (ESSENTIAL - start BEFORE refeeding) [8]
- Multivitamin: Including B vitamins, vitamin C, vitamin D
- Phosphate: Oral supplementation if less than 0.8 mmol/L
- Potassium: Target > 3.5 mmol/L
- Magnesium: Target > 0.75 mmol/L
- Vitamin D and Calcium: For bone health (1000-1200 mg calcium, 800-1000 IU vitamin D daily)
Nasogastric (NG) Feeding:
- Indications:
- Inability to meet nutritional needs orally despite support
- Life-threatening malnutrition with refusal to eat
- Medical compromise requiring urgent restoration
- Ethical Considerations: Used as last resort; requires Mental Capacity Act assessment (England/Wales) or equivalent
- Method: Overnight NG feeding (allows daytime oral eating practice) or continuous feeding
Psychological Therapy
Psychological therapy is the cornerstone of long-term recovery. Medical stabilization alone is insufficient.
First-Line: Enhanced Cognitive Behavioral Therapy (CBT-E)
Evidence Base:
- Developed specifically for eating disorders (Fairburn) [5]
- RCTs demonstrate superiority over treatment as usual
- 50-60% remission rates at 60 weeks
- Effective across eating disorder diagnoses (transdiagnostic)
Structure:
- 40 sessions over 40 weeks (outpatient)
- 20 sessions over 20 weeks (intensive version if BMI > 17)
- Four stages:
- Engagement, psychoeducation, establishing regular eating (Weeks 1-4)
- Individualized formulation addressing maintaining mechanisms (Weeks 5-8)
- Core treatment (address overvaluation of shape/weight, dietary restraint, event-related changes in eating) (Weeks 9-36)
- Relapse prevention (Weeks 37-40)
Key Components:
- Self-monitoring (food diaries, thoughts, behaviors)
- Regular weekly weighing (reduce body checking, increase awareness)
- Cognitive restructuring (challenge distorted beliefs about weight/shape)
- Exposure to feared foods and situations
- Body image work
- Relapse prevention planning
Outcome Predictors:
- Early weight gain (by week 4) predicts long-term remission
- Higher motivation and readiness to change
- Less severe baseline eating pathology
Alternative Evidence-Based Therapies
Maudsley Model of Anorexia Nervosa Treatment for Adults (MANTRA):
- Neuroscience-informed therapy targeting cognitive inflexibility, emotional avoidance
- RCT shows non-inferiority to SSCM (Specialist Supportive Clinical Management)
- May suit patients who do not respond to CBT-E
Specialist Supportive Clinical Management (SSCM):
- Pragmatic, supportive approach combining education, care, and nutritional counseling
- Effective control condition in trials (better than expected)
- Suitable when specialized CBT-E unavailable
Family-Based Therapy (FBT) - For Adolescents/Young Adults:
- Gold Standard for adolescents (less than 18 years) [6]
- May benefit young adults (18-25) living with family
- Empowers parents to take control of refeeding
- Three phases: Weight restoration, returning control to patient, adolescent issues
- Superior to individual therapy in adolescents
Focal Psychodynamic Therapy:
- Addresses underlying emotional conflicts
- Limited evidence compared to CBT-E
- May suit patients with complex trauma or interpersonal difficulties
Pharmacotherapy
Key Point: No pharmacological treatment is FDA/NICE-approved specifically for anorexia nervosa. [13]
Antidepressants (SSRIs):
- Limited evidence for core AN symptoms (restriction, weight gain)
- May help comorbid depression/anxiety after weight restoration (not during acute malnutrition - ineffective and poorly tolerated)
- Fluoxetine 20-60 mg/day most studied
- Do NOT use as first-line; adjunct to psychological therapy only
Atypical Antipsychotics (Olanzapine):
- Small RCTs suggest olanzapine 2.5-10 mg/day may aid weight gain and reduce anxiety around eating
- Weight gain as side effect may be therapeutic
- Not recommended as monotherapy; consider as adjunct in resistant cases
- Requires careful monitoring (metabolic effects)
Other Medications:
- Anxiolytics: May help severe meal-related anxiety (short-term, PRN use)
- Prokinetics (e.g., domperidone): For gastroparesis symptoms (bloating, early satiety)
- Laxatives: Avoid unless severe constipation; address through diet and hydration
Medications to AVOID:
- Bupropion: Lowers seizure threshold (high risk in malnourished patients)
- Tricyclic Antidepressants: Cardiac risks (QTc prolongation) in malnourished patients
Bone Health Management
Challenges:
- Osteoporosis in AN is multifactorial (hypogonadism, hypercortisolism, low IGF-1, malnutrition)
- Bisphosphonates and hormone replacement have LIMITED evidence and concerns about long-term safety in young women
Evidence-Based Approach: [17]
-
Weight Restoration: MOST IMPORTANT intervention
- Every 1 kg weight gain → Improvement in bone density
- Weight restoration partially reverses bone loss (30-60% recovery)
-
Nutritional Support:
- Adequate calcium (1000-1200 mg/day from diet + supplement)
- Vitamin D (800-1000 IU/day; target 25-OH vitamin D > 50 nmol/L)
- Adequate protein intake
-
Hormone Replacement Therapy (HRT):
- Controversial and NOT recommended routinely
- Combined oral contraceptive pill (COCP) does NOT improve bone density in hypogonadal AN patients (landmark RCT 2016)
- Physiological estrogen replacement (transdermal) + cyclic progesterone may have modest benefit but evidence limited
-
Bisphosphonates:
- Generally contraindicated in premenopausal women (teratogenic concerns, long half-life)
- Reserved for severe osteoporosis with fractures in exceptional cases
-
Monitoring:
- DEXA scan at baseline (if > 6 months illness), then 12-24 monthly
- Assess fracture risk (FRAX tool not validated in AN)
-
Weight-Bearing Exercise:
- Beneficial for bone health ONCE weight restored
- Avoid excessive exercise during weight gain phase
Addressing Comorbidities
Depression and Anxiety:
- Common comorbidities (30-75% prevalence)
- Treat with psychological therapy (CBT, SSRI if indicated after weight restoration)
Obsessive-Compulsive Disorder:
- 25-40% comorbidity
- May benefit from ERP (Exposure-Response Prevention) alongside AN treatment
- SSRI (higher doses) if severe and weight restored
Autism Spectrum Traits:
- 20-30% of AN patients have elevated autistic traits (rigidity, social difficulties, sensory sensitivities)
- Tailor therapy to account for cognitive style (e.g., more structured, concrete CBT)
Substance Use Disorders:
- More common in binge/purge subtype
- Dual diagnosis treatment required
Personality Disorders:
- Common (30-50%, particularly Cluster C - avoidant, obsessive-compulsive)
- May require longer, more intensive therapy (DBT, schema therapy)
Compulsory Treatment (Mental Capacity and Mental Health Act)
Mental Capacity:
- Assess capacity to consent to treatment (England/Wales: Mental Capacity Act 2005)
- AN can impair capacity through:
- Inability to weigh information (ego-syntonic illness)
- Impaired cognition from starvation
- Overwhelming fear distorting decision-making
Involuntary Treatment:
- If patient lacks capacity and treatment is in best interests → Treat under Mental Capacity Act (DoLS if inpatient)
- If patient has capacity but detention required for safety → Mental Health Act Section 2/3 (England/Wales)
- Nasogastric feeding can be given under MHA or MCA in life-threatening situations
- Ethical Complexity: Balance autonomy vs beneficence; seek second opinions, involve family, MDT discussion
9. Complications
Acute Medical Complications
| System | Complication | Mechanism | Management |
|---|---|---|---|
| Cardiovascular | Bradycardia, arrhythmias | Electrolyte imbalance, cardiac atrophy, vagal tone | ECG monitoring, electrolyte correction, reduce if HR less than 40 bpm consider medical admission |
| Sudden cardiac death | QTc prolongation → Torsades de pointes | Avoid QTc-prolonging drugs, correct K+/Mg, monitor ECG | |
| Pericardial effusion | Malnutrition, hypoalbuminaemia | Usually resolves with refeeding; echo monitoring | |
| Refeeding Syndrome | Hypophosphataemia, cardiac failure, respiratory failure | Insulin surge → intracellular electrolyte shift | Thiamine pre-feeding, slow refeeding, daily electrolytes, phosphate/K+/Mg replacement |
| Gastrointestinal | Gastroparesis, constipation | Reduced GI motility | Small frequent meals, prokinetics (domperidone), adequate hydration, avoid laxative abuse |
| Superior mesenteric artery syndrome | Compression of duodenum by SMA (loss of mesenteric fat pad) | NG feeding, weight restoration | |
| Refeeding pancreatitis | Rapid nutritional restoration | Slow refeeding, monitor amylase/lipase | |
| Renal | Acute kidney injury | Dehydration, rhabdomyolysis | Hydration, monitor U&E, avoid nephrotoxic drugs |
| Diabetes insipidus (nephrogenic) | Chronic hypovolaemia, electrolyte disturbance | Resolves with refeeding and hydration | |
| Haematological | Pancytopenia | Bone marrow suppression | Resolves with refeeding; rarely requires treatment |
| Neurological | Wernicke's encephalopathy | Thiamine deficiency (esp. during refeeding) | Thiamine 200-300 mg/day prophylaxis; IV thiamine if suspected |
| Peripheral neuropathy | B12, thiamine deficiency | Vitamin supplementation | |
| Metabolic | Hypoglycaemia | Depleted glycogen stores, reduced gluconeogenesis | Frequent meals, glucose monitoring |
| Hypothermia | Reduced metabolic rate, loss of subcutaneous fat | Warming, avoid rapid rewarming |
Chronic Medical Complications
Osteoporosis:
- Most common long-term sequelae [17]
- Occurs early (within 6-12 months of illness onset)
- Affects both trabecular (spine) and cortical (hip) bone
- Increased fracture risk (SIR 3-7 fold)
- Partially reversible with weight restoration (30-60% bone density recovery)
- Persistent deficits common, especially if illness during adolescence (peak bone mass accrual)
Cardiac:
- Structural changes (reduced LV mass) may not fully reverse
- Increased risk of heart failure, arrhythmias in later life
- Mitral valve prolapse persists in some cases
Reproductive:
- Infertility from hypogonadotropic hypogonadism
- Increased risk of miscarriage, preterm birth, low birth weight if pregnant while underweight
- Return of menses typically occurs when BMI > 18-19 kg/m² (but variable)
- Fertility can return before menses (pregnancy possible)
Neurological:
- Residual cognitive deficits (memory, executive function) in chronic cases
- Brain volume may not fully recover despite weight restoration
Growth (If Onset Pre-Pubertal or During Puberty):
- Stunted linear growth if epiphyseal plates not fused
- Delayed puberty
- May not achieve full height potential
Psychiatric Complications
Suicide:
- Accounts for 20-25% of deaths in AN [3,4]
- Risk factors: Binge/purge subtype, comorbid depression, substance use, impulsivity, hopelessness, previous self-harm
- Lifetime suicide rate: 3-7% (vs 0.5% general population)
Treatment-Resistant AN:
- Approximately 20-30% have chronic, enduring illness (> 7 years duration)
- Associated with severe medical and psychosocial impairment
- Requires palliative approach balancing quality of life vs aggressive intervention
Comorbidity Persistence:
- Depression, anxiety, OCD often persist even after weight restoration
- May require ongoing psychiatric treatment
10. Prognosis and Outcomes
Recovery Rates
| Outcome | Rate | Timeframe |
|---|---|---|
| Full Recovery | 50-70% | Variable; median 5-7 years from treatment onset |
| Partial Recovery | 20-30% | Ongoing symptoms but improved functioning |
| Chronic Course | 20-30% | Persistent symptoms > 7 years |
| Crossover to Bulimia Nervosa | 10-20% | Particularly binge/purge subtype |
| Relapse After Recovery | 30-40% | Highest risk in first year post-treatment |
Definition of Recovery:
- Physical: BMI > 18.5 kg/m², regular menses (if female, premenopausal)
- Psychological: Absence of core AN psychopathology (fear of weight gain, body image disturbance, dietary restraint)
- Behavioral: Regular, balanced eating; no compensatory behaviors
- Functional: Return to social, occupational, educational functioning
Mortality
Standardized Mortality Ratio (SMR): 5.0-10.0 [3]
- AN has the highest SMR of any psychiatric disorder
- 5-10% crude mortality rate over 10 years
- 15-20% mortality over 20 years
Causes of Death:
- Suicide: 20-25%
- Cardiac complications: 30-40% (arrhythmias, sudden cardiac death)
- Infection/sepsis: 10-15%
- Multi-organ failure: 10-15%
- Other/unknown: 15-25%
Mortality Risk Factors:
- Longer illness duration (> 7 years)
- Very low BMI (less than 13 kg/m²)
- Binge/purge subtype (higher impulsivity, substance use)
- Comorbid substance use disorder
- Suicide attempts or severe self-harm
- Older age at presentation (adult-onset AN)
- Male gender (higher mortality despite lower prevalence)
Prognostic Factors
Favorable Prognosis
- Shorter illness duration (less than 3 years)
- Younger age at onset (adolescent vs adult)
- Higher BMI at presentation (> 15 kg/m²)
- Restricting subtype (vs binge/purge)
- Good premorbid functioning (academic, social)
- Strong family support (engaged, non-critical)
- Early treatment engagement (within 1 year of onset)
- Absence of comorbid psychiatric disorders
- Good therapeutic alliance
Poor Prognosis
- Longer illness duration (> 7 years → "enduring/severe AN")
- Adult-onset AN (> 18 years at first presentation)
- Very low BMI (less than 13 kg/m²) or extreme weight loss
- Binge/purge subtype
- Comorbid psychiatric disorders (depression, OCD, personality disorder, substance use)
- Comorbid medical complications (osteoporosis with fractures, cardiac dysfunction)
- Lack of insight (ego-syntonic illness, denial)
- Poor family support or high expressed emotion (criticism, hostility)
- Multiple previous treatment failures
- Compulsory treatment history
Functional Outcomes
Even after weight restoration and symptom remission, functional impairment can persist:
-
Psychosocial:
- Difficulty forming intimate relationships (30-40% never marry)
- Social isolation and withdrawal
- Occupational underachievement relative to premorbid potential
-
Educational:
- Interruption of education (university dropout)
- Cognitive deficits (memory, concentration) impacting performance
-
Economic:
- Reduced employment rates
- High healthcare costs (recurrent admissions, long-term therapy)
Enduring Anorexia Nervosa ("SE-AN")
Defined as illness duration > 7 years with multiple treatment attempts and persistent symptoms.
Challenges:
- Severe, entrenched behaviors
- Extensive medical complications
- Demoralization and hopelessness
- Treatment fatigue
Approach:
- Shift from "cure" to harm reduction and quality of life
- Maintain medical safety (regular monitoring, prevent further deterioration)
- Realistic goal-setting (small, achievable targets)
- Maintain therapeutic relationship (avoid abandonment)
- Consider palliative care approach in exceptional cases (end-of-life care)
11. Prevention and Screening
Primary Prevention (Population Level)
Targets:
- Reduce societal emphasis on thinness and appearance
- Media literacy programs (critical evaluation of body ideals)
- Regulate advertising and media portrayal of bodies
- School-based programs promoting healthy body image, self-esteem, resilience
Evidence:
- Mixed results; some programs (e.g., "Body Project") reduce risk factors (thin-ideal internalization)
- No programs proven to reduce AN incidence at population level
Secondary Prevention (Early Detection)
Screening in High-Risk Populations:
- Adolescent girls (peak onset 14-18 years)
- Occupational groups (ballet, modeling, elite athletics)
- Patients presenting with weight loss, amenorrhoea, gastrointestinal symptoms
- First-degree relatives of individuals with eating disorders
Screening Tools:
- SCOFF Questionnaire: Primary care, quick (5 questions), high sensitivity [12]
- EDE-Q: More detailed, specialist services
- BEDS-7: Brief (7-item) screening tool
Primary Care Role:
- Opportunistic screening during adolescent health checks
- Investigate unexplained weight loss or amenorrhoea
- Maintain high index of suspicion (patients often conceal symptoms)
Tertiary Prevention (Relapse Reduction)
Relapse Risk:
- 30-40% of recovered patients relapse within 1-2 years
- Highest risk in first 12 months post-discharge from inpatient care
Relapse Prevention Strategies:
- Continued psychological therapy (booster sessions, long-term follow-up)
- Regular monitoring (weight, mental state)
- Early warning sign recognition (return of food rules, increased exercise, body checking)
- Crisis plan (what to do if symptoms return; re-engage with team)
- Family/carer involvement (monitor for early signs)
- Address maintaining factors (perfectionism, low self-esteem, interpersonal difficulties)
12. Examination Focus (Viva/OSCE Scenarios)
Viva Question 1: Differentiating AN from Other Causes of Weight Loss
Examiner: "A 24-year-old woman presents with BMI 16 kg/m² and 12 kg weight loss over 6 months. How would you differentiate anorexia nervosa from other causes of weight loss?"
Model Answer:
"I would take a detailed history and perform targeted investigations to distinguish psychiatric from organic causes.
History:
- Body Image and Fear of Weight Gain: In AN, the patient has intense fear of gaining weight and sees herself as overweight despite being underweight. Organic causes lack this psychopathology.
- Appetite: In AN, appetite is often preserved initially but suppressed by cognitive restraint. In depression, appetite is reduced (anhedonia). In hyperthyroidism or malignancy, appetite may be preserved or increased despite weight loss.
- Eating Behaviors: In AN, deliberate restriction, food rules, avoidance of high-calorie foods, ritualistic eating. Organic causes do not show these features.
- Attitude to Weight Loss: In AN, weight loss is desired and ego-syntonic. In organic disease, weight loss is unwanted and distressing.
- Menstrual History: In AN, amenorrhoea often precedes significant weight loss (hypothalamic suppression). In organic causes, amenorrhoea follows weight loss.
Examination:
- Vital Signs: AN shows bradycardia and hypotension; hyperthyroidism shows tachycardia; malignancy may show fever.
- Signs of Malnutrition: Lanugo, dry skin, hair loss (AN vs malabsorption/malignancy).
- Lymphadenopathy/Organomegaly: Suggests malignancy or infection.
Investigations:
- TFTs: Exclude hyperthyroidism (↑T4, ↑T3, ↓TSH). In AN, Low T3 syndrome (↓T3, normal T4, normal/low TSH).
- Coeliac Screen: Tissue transglutaminase antibodies.
- Inflammatory Markers: CRP/ESR raised in IBD, malignancy, infection.
- Imaging: CXR, CT chest/abdomen/pelvis if red flags for malignancy.
In this case, if history reveals body image disturbance, fear of weight gain, and deliberate restriction with normal or reassuring investigations, the diagnosis is anorexia nervosa."
Viva Question 2: Refeeding Syndrome
Examiner: "You are admitting a 28-year-old woman with anorexia nervosa, BMI 13.5 kg/m², for medical stabilization. What is refeeding syndrome and how would you prevent it?"
Model Answer:
"Refeeding syndrome is a potentially fatal complication occurring when nutrition is reintroduced after prolonged starvation. It is characterized by severe electrolyte shifts, particularly hypophosphataemia, and can cause cardiac, respiratory, and neurological dysfunction. [7,8]
Pathophysiology: During starvation, metabolism shifts to fat and protein catabolism with reduced intracellular electrolytes. When carbohydrate is reintroduced:
- Insulin surge drives glucose, phosphate, potassium, and magnesium intracellularly.
- Serum phosphate can drop precipitously (to less than 0.5 mmol/L).
- Phosphate is essential for ATP production; severe hypophosphataemia causes:
- Respiratory failure (diaphragm weakness)
- Cardiac dysfunction (arrhythmias, reduced contractility)
- Rhabdomyolysis
- Haemolysis
- Neurological dysfunction (confusion, seizures)
Risk Factors in This Patient:
- BMI less than 15 kg/m² (high risk)
- Likely negligible intake for prolonged period
- Possible baseline electrolyte depletion
Prevention Strategy:
1. Pre-Feeding Assessment:
- Baseline bloods: U&E, phosphate, magnesium, calcium, glucose, FBC, LFTs
- ECG (QTc, bradycardia)
- Assess fluid status
2. Thiamine Supplementation:
- CRITICAL: Give thiamine 200-300 mg PO daily (or 100 mg IV TDS if unable to absorb orally) BEFORE starting feeding to prevent Wernicke's encephalopathy.
- Also give multivitamin with B complex.
3. Start Low, Go Slow:
- Initial caloric intake: 10-20 kcal/kg/day (approximately 600-1000 kcal/day for this patient).
- Increase by 200-300 kcal every 3-5 days, guided by electrolytes and clinical status.
4. Electrolyte Supplementation:
- Phosphate: Oral supplementation (Phosphate Sandoz 2-3 sachets/day) if baseline less than 0.8 mmol/L.
- Potassium: Target > 3.5 mmol/L (oral Sando-K or IV if less than 2.5 mmol/L).
- Magnesium: Target > 0.75 mmol/L (oral magnesium glycerophosphate).
5. Daily Monitoring (First 3-7 Days):
- Electrolytes (phosphate, K+, Mg, Ca, U&E) - DAILY
- Glucose
- Weight, vital signs (HR, BP, temperature) - TDS-QDS
- Fluid balance (risk of fluid overload)
- ECG if baseline abnormal or symptomatic
6. Red Flags During Refeeding:
- Phosphate less than 0.5 mmol/L → URGENT replacement (IV if less than 0.3 mmol/L)
- New confusion, weakness, respiratory distress
- Arrhythmias
- Peripheral oedema (fluid overload)
This structured approach minimizes the risk of refeeding syndrome while safely restoring nutrition."
Viva Question 3: Psychological Treatment
Examiner: "What is the first-line psychological therapy for adults with anorexia nervosa, and why might pharmacotherapy have limited efficacy?"
Model Answer:
"The first-line psychological therapy for adults with anorexia nervosa is Enhanced Cognitive Behavioral Therapy for Eating Disorders (CBT-E). [5]
CBT-E:
- Developed by Fairburn specifically for eating disorders.
- Transdiagnostic (effective across AN, bulimia nervosa, binge eating disorder).
- 40 sessions over 40 weeks (outpatient) or 20 sessions over 20 weeks (intensive).
- Targets core maintaining mechanisms:
- Overvaluation of weight and shape (self-worth tied to weight/appearance)
- Dietary restraint and food rules
- Body image disturbance
- Event-related changes in eating (emotional eating, social eating avoidance)
Key Components:
- Psychoeducation (starvation effects, set-point weight)
- Self-monitoring (food diaries, thoughts, behaviors)
- Regular weighing (reduce body checking, improve awareness)
- Cognitive restructuring (challenge beliefs about weight/shape)
- Exposure (feared foods, avoided situations)
- Relapse prevention
Evidence:
- RCTs show 50-60% remission at 60 weeks.
- Superior to treatment as usual.
- Effective for adults; Family-Based Therapy (FBT) preferred for adolescents.
Alternative Therapies:
- MANTRA (Maudsley Model): Targets cognitive inflexibility, emotional avoidance.
- SSCM (Specialist Supportive Clinical Management): Pragmatic, supportive.
- Focal Psychodynamic Therapy: Addresses underlying conflicts (less evidence).
Why is Pharmacotherapy Limited?
No medication is approved for anorexia nervosa. [13]
SSRIs:
- Limited efficacy for core AN symptoms (weight gain, restriction).
- In acute malnutrition, SSRIs are ineffective and poorly tolerated (5-HT depletion from malnutrition).
- May help comorbid depression/anxiety AFTER weight restoration.
- Fluoxetine 20-60 mg most studied; consider as adjunct, not monotherapy.
Atypical Antipsychotics (e.g., Olanzapine):
- Small studies suggest olanzapine 2.5-10 mg may aid weight gain and reduce anxiety.
- Weight gain as side effect may be therapeutic.
- Not monotherapy; consider in treatment-resistant cases.
Why Poor Response?
- Neurobiological Changes: Serotonergic, dopaminergic dysfunction is state-dependent (malnutrition) and partially trait (persists after recovery). Medication addresses neither fully.
- Ego-Syntonic Illness: Patients often desire weight loss; medication compliance poor.
- Psychological Core: AN is fundamentally a disorder of cognition and behavior (body image, fear, control). Medication does not address these.
- Starvation State: Malnutrition impairs neurotransmitter synthesis, reducing medication efficacy.
Conclusion: Psychological therapy (CBT-E) is first-line. Medication is adjunctive at best, used for comorbidities after weight restoration."
OSCE Station: Assessment of Medically Unstable Patient
Scenario: You are the FY2 in the Emergency Department. A 22-year-old woman with known anorexia nervosa has been brought in by her mother due to dizziness and weakness. Assess her medical stability and determine disposition.
Approach:
History:
- Recent intake (food, fluids) - assess risk of acute malnutrition/dehydration
- Weight loss (rate and amount)
- Purging behaviors (vomiting, laxatives, diuretics) - risk of electrolyte imbalance
- Syncope, palpitations, chest pain - cardiac symptoms
- Psychiatric state (suicidal ideation, self-harm)
- Current treatment (therapist, admissions, previous medical complications)
Examination:
- Vital Signs: HR (bradycardia less than 40 bpm?), BP lying and standing (postural drop > 20 mmHg?), temperature (less than 35°C?), weight/BMI
- SUSS Test: Sit up from lying, squat and stand - assesses muscle strength and medical stability
- Cardiovascular: Peripheral perfusion, JVP, heart sounds
- Mental State: Orientation, mood, suicidal ideation
Investigations:
- Bloods: FBC, U&E (K+, Na+, urea, creatinine), bone profile (Ca, phosphate, Mg), glucose, LFTs
- ECG: Bradycardia, QTc prolongation (> 450 ms?), arrhythmias
- VBG: If obtunded/confused (assess acid-base, lactate)
Red Flags Requiring Medical Admission (MARSIPAN Criteria): [10]
- HR less than 40 bpm
- Systolic BP less than 90 mmHg or symptomatic postural hypotension
- Core temperature less than 35°C
- BMI less than 13 kg/m² (or less than 15 with rapid loss)
- K+ less than 3.0 mmol/L, Phosphate less than 0.5 mmol/L
- QTc > 500 ms
- Hypoglycaemia less than 3.0 mmol/L
- SUSS test failure
- Acute suicidal risk
Disposition:
- If Red Flags Present: Medical admission for stabilization, refeeding protocol, daily electrolyte monitoring.
- If Stable: Refer to Community Eating Disorder Service (urgent), arrange GP follow-up, safety plan.
Communication:
- Explain findings to patient and family.
- Discuss need for admission if indicated (may require Mental Health Act/Mental Capacity Act assessment if refusing).
- Empathic, non-judgmental approach.
13. Patient and Layperson Explanation
What is Anorexia Nervosa?
Anorexia nervosa is a serious mental health condition that affects how you think about food, eating, and your body. People with anorexia nervosa severely restrict how much they eat because they have an intense fear of gaining weight and see themselves as overweight, even when they are dangerously underweight. It is not about vanity or wanting to look good - it is a complex illness involving the brain, emotions, and body.
Anorexia nervosa is the most deadly psychiatric disorder. Without treatment, it can cause serious medical problems affecting the heart, bones, brain, and other organs. However, with the right treatment and support, many people make a full recovery.
What Causes It?
Anorexia nervosa does not have a single cause. It develops from a combination of factors:
- Genetics: It runs in families. If someone in your family has an eating disorder, you have a higher risk.
- Brain chemistry: Differences in how the brain processes emotions, anxiety, and reward.
- Personality: People who are perfectionists, anxious, or have low self-esteem are at higher risk.
- Life stress: Dieting, bullying about weight, difficult life events, or transitions (like starting university) can trigger it.
- Culture: Living in a society that values thinness and appearance can contribute.
What Are the Symptoms?
Physical Symptoms:
- Severe weight loss (becoming very underweight)
- Feeling cold all the time
- Dizziness or fainting
- Tiredness and weakness
- Periods stopping (in women)
- Thinning hair, dry skin, or fine body hair (lanugo)
- Slow heartbeat
Behavioral and Emotional Symptoms:
- Eating very little, skipping meals
- Avoiding certain foods (especially fats and carbohydrates)
- Intense fear of gaining weight
- Seeing yourself as "fat" even when you are very thin
- Excessive exercise
- Weighing yourself constantly or checking your body in mirrors
- Withdrawing from friends and family
What Are the Dangers?
Anorexia nervosa is life-threatening if not treated:
- Heart problems: Slow heartbeat, abnormal heart rhythms, sudden cardiac death
- Bone problems: Weak bones (osteoporosis) that can fracture easily, even at a young age
- Brain changes: Difficulty concentrating, memory problems
- Organ damage: Kidney problems, liver problems
- Refeeding syndrome: When eating is restarted after starvation, dangerous shifts in body chemistry can occur (requires medical supervision)
- Suicide: People with anorexia nervosa have a higher risk of depression and suicide
5-10% of people with anorexia nervosa die within 10 years, mostly from heart problems or suicide. This is why early treatment is so important.
How is it Diagnosed?
A doctor or mental health professional will:
- Ask about your eating, thoughts about weight and body shape, and any physical symptoms
- Measure your weight and height to calculate your BMI
- Do blood tests to check for malnutrition, electrolyte imbalances, and organ function
- Perform an ECG (heart tracing) to check your heart
There are also questionnaires (like the SCOFF) that help screen for eating disorders.
How is it Treated?
Treatment involves a team of specialists (psychiatrist, psychologist, dietitian, doctor) and focuses on three goals:
- Restore weight to a healthy range
- Address the thoughts and behaviors around food and body image
- Treat medical complications
Medical Treatment:
- Nutritional rehabilitation: Working with a dietitian to gradually increase food intake safely. This may involve meal plans, supervised meals, or (in severe cases) tube feeding.
- Monitoring: Regular checks of weight, heart rate, blood pressure, and blood tests to ensure safety during refeeding.
- Prevention of refeeding syndrome: When you start eating again after starvation, your body chemistry can become dangerously imbalanced. Doctors give vitamins (especially thiamine) and monitor electrolytes closely.
Psychological Treatment:
- Therapy: The main treatment is talking therapy (psychotherapy). The most effective type for adults is called CBT-E (Enhanced Cognitive Behavioral Therapy for Eating Disorders). It helps you:
- Understand how your thoughts affect your eating
- Challenge beliefs about weight and body shape
- Gradually reintroduce feared foods
- Develop a healthier relationship with food
- Family therapy: For younger people (teenagers and young adults), involving family in treatment (called Family-Based Therapy or FBT) is very effective.
Medication:
- There is no medication specifically for anorexia nervosa. Antidepressants (like SSRIs) may help with anxiety or depression once you have gained some weight, but they do not work for the core eating disorder symptoms on their own.
Where Does Treatment Happen?
- Outpatient: Weekly therapy sessions, dietitian appointments, and regular medical check-ups (for people who are medically stable).
- Day program: Spending several days per week at a clinic with supervised meals and therapy (for those needing more support).
- Inpatient (hospital): For people who are medically unstable (very low weight, dangerous heart rate or blood pressure, severe electrolyte problems, or high suicide risk). Hospital admission focuses on medical stabilization and weight restoration.
What to Expect During Recovery
Recovery from anorexia nervosa takes time - often several years. It is not a straight path; there may be setbacks along the way. However, with treatment:
- 50-70% of people make a full recovery (healthy weight, normal eating, no longer obsessed with weight/shape)
- 20-30% have partial recovery (improved but still some symptoms)
- 20-30% have a chronic course (ongoing symptoms for many years)
The earlier treatment starts, the better the outcome. If you have been ill for less than 3 years and receive good treatment, your chances of full recovery are high.
What Helps Recovery:
- Committing to treatment (even when it feels very difficult)
- Support from family and friends
- Addressing any other mental health problems (depression, anxiety)
- Building a life beyond weight and appearance (hobbies, relationships, goals)
When to Get Help
Seek help urgently if:
- You or someone you know has lost a lot of weight quickly
- Feeling dizzy, faint, or having chest pain
- Heart rate very slow or irregular
- Suicidal thoughts
See your GP if:
- You are worried about your eating or weight
- You think you might have an eating disorder
- Someone you care about has signs of anorexia nervosa
Early help saves lives. Anorexia nervosa is treatable, and recovery is possible.
Support Organizations
- Beat Eating Disorders (UK): beateatingdisorders.org.uk - Helpline: 0808 801 0677
- National Eating Disorders Association (USA): nationaleatingdisorders.org
- Butterfly Foundation (Australia): butterfly.org.au
14. Key Guidelines and Evidence
Guidelines
-
National Institute for Health and Care Excellence (NICE). Eating Disorders: Recognition and Treatment (NG69). 2017.
- Comprehensive UK guideline covering identification, assessment, and management of all eating disorders.
- Recommends CBT-E for adults, FBT for adolescents.
- Available: www.nice.org.uk/guidance/ng69
-
Royal College of Psychiatrists, Royal College of Physicians, et al. MARSIPAN: Management of Really Sick Patients with Anorexia Nervosa (Third Edition). 2022.
- UK guideline for medical emergencies in AN.
- Defines admission criteria, refeeding protocols, monitoring requirements.
- Available: www.rcpsych.ac.uk/improving-care/campaigning-for-better-mental-health-policy/college-reports/marsipan
-
American Psychiatric Association. Practice Guideline for the Treatment of Patients with Eating Disorders (Third Edition). 2006 (Under Revision).
- US guideline; comprehensive review of evidence.
- Covers assessment, levels of care, medical management, psychological treatment.
Key Evidence
| Intervention/Topic | Evidence Level | Key Study/Reference |
|---|---|---|
| CBT-E for Adults | 1a | Fairburn et al. RCT demonstrating superiority over control; 50-60% remission [5] |
| Family-Based Therapy (FBT) for Adolescents | 1a | Lock et al. RCTs showing FBT superior to individual therapy in adolescents [6] |
| Refeeding Syndrome Prevention | 3 | MARSIPAN guidelines; observational studies on thiamine, electrolyte monitoring [7,8,10] |
| Mortality in AN | 2b | Arcelus et al. Meta-analysis of 36 studies; SMR 5.0-10.0 [3] |
| Genetics (GWAS) | 2b | Watson et al. GWAS identifying 8 loci; metabo-psychiatric origins [14] |
| Osteoporosis in AN | 2b | Robinson et al. Studies on bone density; weight restoration as primary intervention [17] |
| SSRIs in AN | 1b | Attia et al. RCTs showing limited efficacy; may help post-weight restoration [13] |
15. References
-
American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5). Arlington, VA: American Psychiatric Publishing; 2013.
-
Attia E, Walsh BT. Eating Disorders: A Review. JAMA. 2025;335(14):1187-1197. doi:10.1001/jama.2025.0132. PMID: 40048192
-
Arcelus J, Mitchell AJ, Wales J, Nielsen S. Mortality rates in patients with anorexia nervosa and other eating disorders. A meta-analysis of 36 studies. Arch Gen Psychiatry. 2011;68(7):724-731. doi:10.1001/archgenpsychiatry.2011.74. PMID: 21727255
-
van Eeden AE, van Hoeken D, Hoek HW. Incidence, prevalence and mortality of anorexia nervosa and bulimia nervosa. Curr Opin Psychiatry. 2021;34(6):515-524. doi:10.1097/YCO.0000000000000739. PMID: 34419970
-
Atwood ME, Friedman A. A systematic review of enhanced cognitive behavioral therapy (CBT-E) for eating disorders. Int J Eat Disord. 2020;53(3):311-330. doi:10.1002/eat.23206. PMID: 31840285
-
Lock J, Le Grange D. Family-based treatment of eating disorders. Int J Eat Disord. 2005;37 Suppl:S64-67. doi:10.1002/eat.20122.
-
Garber AK, Sawyer SM, Golden NH, et al. A systematic review of approaches to refeeding in patients with anorexia nervosa. Int J Eat Disord. 2016;49(3):293-310. doi:10.1002/eat.22482. PMID: 26661289
-
Skowrońska A, Sójta K, Strzelecki D. Refeeding syndrome as treatment complication of anorexia nervosa. Psychiatr Pol. 2019;53(5):1113-1123. doi:10.12740/PP/OnlineFirst/90275. PMID: 31955189
-
Harrington BC, Jimerson M, Haxton C, Jimerson DC. Initial evaluation, diagnosis, and treatment of anorexia nervosa and bulimia nervosa. Am Fam Physician. 2015;91(1):46-52. PMID: 25591200
-
Royal College of Psychiatrists, Royal College of Physicians, et al. MARSIPAN: Management of Really Sick Patients with Anorexia Nervosa. Third Edition. 2022. Available at: https://www.rcpsych.ac.uk
-
Keys A, Brožek J, Henschel A, Mickelsen O, Taylor HL. The Biology of Human Starvation (2 volumes). Minneapolis: University of Minnesota Press; 1950.
-
Morgan JF, Reid F, Lacey JH. The SCOFF questionnaire: assessment of a new screening tool for eating disorders. BMJ. 1999;319(7223):1467-1468. doi:10.1136/bmj.319.7223.1467.
-
Attia E, Steinglass JE, Walsh BT, et al. Olanzapine versus placebo in adult outpatients with anorexia nervosa: a randomized clinical trial. Am J Psychiatry. 2019;176(6):449-456. doi:10.1176/appi.ajp.2018.18101125.
-
Watson HJ, Yilmaz Z, Thornton LM, et al. Genome-wide association study identifies eight risk loci and implicates metabo-psychiatric origins for anorexia nervosa. Nat Genet. 2019;51(8):1207-1214. doi:10.1038/s41588-019-0439-2. PMID: 31308545
-
Frank GKW, Shott ME, DeGuzman MC. The Neurobiology of Eating Disorders. Child Adolesc Psychiatr Clin N Am. 2019;28(4):629-640. doi:10.1016/j.chc.2019.05.007. PMID: 31443880
-
Seitz J, Herpertz-Dahlmann B, Konrad K. Brain morphological changes in adolescent and adult patients with anorexia nervosa. J Neural Transm (Vienna). 2016;123(8):949-959. doi:10.1007/s00702-016-1567-9.
-
Robinson L, Aldridge V, Clark EM, Misra M, Micali N. Bone health in eating disorders: what the clinician needs to know. Lancet Diabetes Endocrinol. 2016;4(6):584-595. doi:10.1016/S2213-8587(15)00459-X.
-
Sachs KV, Harnke B, Mehler PS, Krantz MJ. Cardiovascular complications of anorexia nervosa: A systematic review. Int J Eat Disord. 2016;49(3):238-248. doi:10.1002/eat.22481.
Medical Disclaimer: MedVellum content is for educational purposes and clinical reference only. Anorexia nervosa is a medical and psychiatric emergency. If you or someone you know is experiencing symptoms, seek immediate medical and mental health evaluation. In emergencies (suicidal thoughts, medical instability), contact emergency services or go to the nearest emergency department.
Frequently asked questions
Quick clarifications for common clinical and exam-facing questions.
When should I seek emergency care for anorexia nervosa (adult)?
Seek immediate emergency care if you experience any of the following warning signs: Severe electrolyte imbalance (hypokalaemia less than 3.0 mmol/L), Bradycardia (HR less than 40 bpm) or significant arrhythmia, Refeeding syndrome risk (phosphate less than 0.5 mmol/L), Severe malnutrition (BMI less than 15 kg/m2), Acute suicidal ideation or intent, Rapid weight loss (less than 1 kg/week sustained), Hypothermia (less than 35CC core temperature), Hypoglycaemia (less than 3.0 mmol/L), QTc prolongation less than 500 ms, Symptomatic postural hypotension.
Learning map
Use these linked topics to study the concept in sequence and compare related presentations.
Prerequisites
Start here if you need the foundation before this topic.
- Body Image Disorders
- Nutritional Assessment
Differentials
Competing diagnoses and look-alikes to compare.
- Bulimia Nervosa
- Avoidant/Restrictive Food Intake Disorder (ARFID)
- Depression with Weight Loss
- Hyperthyroidism
- Malignancy
Consequences
Complications and downstream problems to keep in mind.
- Osteoporosis (Secondary)
- Refeeding Syndrome
- Cardiac Arrhythmias