Anterior Uveitis
Anterior uveitis (iritis/iridocyclitis) is inflammation of the iris and ciliary body, presenting as a painful red eye wi... MRCP, FRCS Ophth exam preparation.
What matters first
Anterior uveitis (iritis/iridocyclitis) is inflammation of the iris and ciliary body, presenting as a painful red eye wi... MRCP, FRCS Ophth exam preparation.
Hypopyon (pus in anterior chamber) — severe inflammation, consider infection
11 Jan 2026
Generated educational material; verify before clinical use.
Visible references section
See the concept before reading it
Study the key anatomy, imaging, and decision pathways as full teaching plates.
Clinical board
A visual summary of the highest-yield teaching signals on this page.
Urgent signals
Safety-critical features pulled from the topic metadata.
- Hypopyon (pus in anterior chamber) — severe inflammation, consider infection
- Significant visual loss — urgent ophthalmology
- Irregular pupil from synechiae — risk of complications
- Posterior segment involvement — intermediate/posterior uveitis
Exam focus
Current exam surfaces linked to this topic.
- MRCP
- FRCS Ophth
- FRANZCO
Content status and exam context
This page is AI-generated educational content. It may contain errors or omissions and is not a substitute for current guidelines, local protocols, senior clinical judgement, or professional medical advice.
MedVellum does not claim an individual clinician reviewer, board certification, or professional credential for this page unless a future version names a real, verifiable reviewer.
Clinical explanation and evidence
Anterior Uveitis
1. Clinical Overview
Summary
Anterior uveitis (iritis/iridocyclitis) is inflammation of the iris and ciliary body, presenting as a painful red eye with photophobia, lacrimation, and blurred vision. It is the most common form of uveitis, accounting for 75-90% of cases, with an incidence of 12-27 per 100,000 per year in Western populations. [1,2] The condition is strongly associated with HLA-B27 and seronegative spondyloarthropathies (ankylosing spondylitis, reactive arthritis, psoriatic arthritis, IBD-associated arthritis). [3,4] While idiopathic in approximately 45-50% of cases, systemic diseases such as sarcoidosis, Behçet's disease, herpes infections, and tuberculosis must be considered. [1,5] Treatment requires urgent ophthalmology referral, topical corticosteroids to reduce inflammation, and cycloplegic agents to relieve pain and prevent posterior synechiae formation. [6,7] Recurrence is common (40-60%), particularly in HLA-B27 positive patients. [8]
Key Facts
- Definition: Inflammation of the iris (iritis) ± ciliary body (iridocyclitis)
- Incidence: 12-27 per 100,000 per year; higher in Finland (23/100,000) and HLA-B27+ populations [1,2]
- Prevalence: 38-115 per 100,000 population [1]
- Proportion of Uveitis: 75-90% of all uveitis cases; most common anatomical subtype [1,2]
- Presentation: Painful red eye, photophobia (consensual), blurred vision, lacrimation
- Classic Sign: Ciliary flush (circumcorneal limbal injection)
- Diagnostic Finding: Cells and flare in anterior chamber on slit lamp examination
- Key Association: HLA-B27 (present in 40-70% of acute anterior uveitis; 90% of AS-related uveitis) [3,8]
- Peak Age: 20-50 years; can occur at any age [1]
- Key Management: Urgent ophthalmology; topical corticosteroids + cycloplegics [6,7]
Clinical Pearls
"Painful Red Eye with Photophobia = Think Uveitis": Unlike conjunctivitis (gritty, no photophobia, no visual loss) or episcleritis (localised, minimal pain), anterior uveitis causes deep aching pain, consensual photophobia, and vision changes.
Consensual Photophobia: Shining light in the UNAFFECTED eye causes pain in the AFFECTED eye. This is highly specific for anterior uveitis — ciliary muscle spasm is triggered bilaterally through the Edinger-Westphal nucleus reflex arc.
HLA-B27 Link: 40-70% of acute anterior uveitis is HLA-B27 positive. [3,8] Ask about back pain, morning stiffness (> 30 minutes), alternating buttock pain, heel pain (enthesitis), and family history of spondyloarthropathy.
Recurrence Risk: 40-60% of patients experience recurrent episodes; higher in HLA-B27+ (50-70%). [8] Often affects the same eye repeatedly, but can alternate.
"Granulomatous vs Non-Granulomatous": Large "mutton-fat" keratic precipitates suggest granulomatous uveitis (sarcoidosis, tuberculosis, sympathetic ophthalmia); fine KPs suggest non-granulomatous (HLA-B27, idiopathic, viral).
Why This Matters Clinically
Anterior uveitis requires prompt recognition and treatment to prevent complications such as posterior synechiae (iris adhesion to lens), glaucoma, cataract, cystoid macular oedema, and permanent visual loss. [9,10] The incidence has increased 1.6-fold over the past 50 years, possibly due to improved recognition and diagnostic criteria. [2] Recurrent episodes are common and indicate the need for investigation of underlying systemic disease. Many cases represent the presenting manifestation of seronegative spondyloarthropathy or other systemic inflammatory conditions, making appropriate investigation and rheumatology referral important. [3,4] Vision loss occurs in 5-10% of patients, primarily from complications of chronic or recurrent inflammation. [10]
2. Epidemiology
Incidence & Prevalence
Global Incidence:
- Overall: 12-27 per 100,000 per year in Western populations [1,2]
- Finland: 23 per 100,000 per year (higher due to HLA-B27 prevalence) [7]
- United States: 26.9 per 100,000 per year (Olmsted County, Minnesota study) [2]
- Trend: Incidence increased 1.6-fold from 1960-2015 in US population studies [2]
Prevalence:
- 38-115 per 100,000 population [1]
- Variation reflects differences in HLA-B27 frequency and diagnostic criteria
Proportion of All Uveitis:
- Anterior uveitis: 75-90% of all uveitis cases [1,2]
- Most common anatomical subtype worldwide
- Higher proportion in developed countries vs developing countries (where infectious posterior uveitis is more common) [1]
Demographics
| Factor | Details | Evidence |
|---|---|---|
| Age | Peak incidence 20-50 years; can occur at any age | Most common in working-age adults [1,2] |
| Sex | Slight male predominance in HLA-B27+ cases (M:F 1.3:1); otherwise equal | [2,3] |
| Ethnicity | Parallels HLA-B27 prevalence: Higher in Northern Europeans, Native Americans, circumpolar groups (Sami); Lower in Japanese, sub-Saharan Africans | [3,4] |
| Geography | Higher in Northern Europe (HLA-B27 frequency 8-14%); Lower in East Asia, sub-Saharan Africa | [1,3] |
| Laterality | Usually unilateral (80-85%); bilateral presentation suggests systemic disease | [1] |
Risk Factors
| Factor | Relative Risk | Details | Evidence |
|---|---|---|---|
| HLA-B27 | OR 3-10 | Present in 40-70% of AAU; stronger predictor of recurrence (50-70% recur vs 30-40% in HLA-B27 negative) | [3,8] |
| Ankylosing Spondylitis | RR 5-10 | 30-50% of AS patients develop AAU; up to 90% are HLA-B27+ | [3,4] |
| Reactive Arthritis | RR 3-5 | 12-37% develop AAU; post-enteric or urogenital infection | [3] |
| Psoriatic Arthritis | RR 2-4 | 7-18% develop AAU | [3,4] |
| IBD (Crohn's/UC) | RR 3-5 | 2-9% develop AAU; more common in Crohn's with colonic involvement | [4] |
| Previous Episode | RR 2-5 | 40-60% recurrence rate overall; higher in HLA-B27+ | [8] |
| Family History SpA | OR 2-3 | First-degree relatives of HLA-B27+ patients | [3] |
| Smoking | OR 1.3-1.8 | May increase recurrence risk; mechanism unclear | [8] |
Aetiology Classification
| Category | Examples | Frequency | Key Features |
|---|---|---|---|
| Idiopathic | No identifiable cause | ~45-50% | Most common; often recurrent; may unmask spondyloarthropathy on follow-up |
| HLA-B27 Associated SpA | AS, reactive arthritis, psoriatic arthritis, IBD-arthritis, undifferentiated SpA | 20-35% | Acute, non-granulomatous, often recurrent, alternating eyes |
| Infectious | HSV, VZV, CMV, TB, syphilis, Lyme, toxoplasmosis | 5-15% | HSV/VZV: unilateral, elevated IOP, iris atrophy, KPs; TB: granulomatous |
| Systemic Inflammatory | Sarcoidosis, Behçet's, JIA, TINU syndrome, Vogt-Koyanagi-Harada | 5-15% | Sarcoid: granulomatous, bilateral, "mutton-fat" KPs; Behçet's: hypopyon |
| Masquerade Syndromes | Lymphoma, leukaemia, retinoblastoma, pigment dispersion | Rare (less than 2%) | Atypical course, elderly patients, unresponsive to steroids |
| Drug-Induced | Rifabutin, sulfonamides, bisphosphonates, checkpoint inhibitors | Rare | History of recent drug initiation |
HLA-B27 Epidemiology
HLA-B27 Prevalence by Population: [3,4]
- Northern Europeans: 8-14%
- Mediterranean Europeans: 2-6%
- Sub-Saharan Africans: less than 1%
- East Asians (Han Chinese, Japanese): 3-6%
- Native Americans: Up to 50% (Haida, Tlingit)
- Circumpolar groups: Up to 40%
HLA-B27 in Anterior Uveitis:
- Acute AAU: 40-70% HLA-B27 positive [3,8]
- AS-associated AAU: 90% HLA-B27 positive [3,4]
- Recurrent AAU: 50-70% HLA-B27 positive [8]
- Chronic AAU: 10-20% HLA-B27 positive (different phenotype)
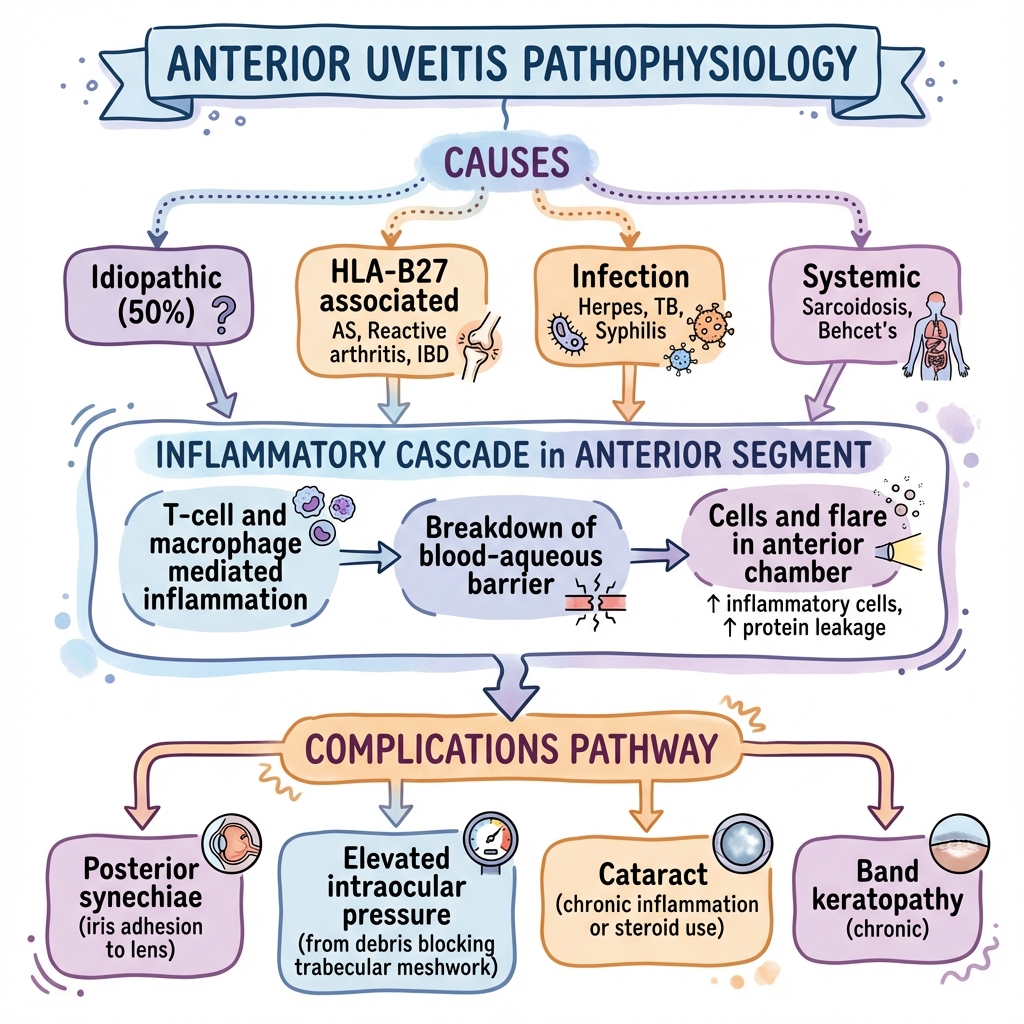
3. Pathophysiology
Molecular and Cellular Mechanisms
Step 1: Immune Activation and Breakdown of Ocular Immune Privilege
Ocular Immune Privilege:
- The eye is normally an "immune-privileged" site protected by the blood-aqueous barrier (BAB)
- BAB consists of: (1) Tight junctions in ciliary non-pigmented epithelium, (2) Tight junctions in iris vascular endothelium, (3) Lack of MHC class II expression on resident cells
- Anterior chamber-associated immune deviation (ACAID) suppresses inflammatory responses
Breakdown of Privilege: [11,12]
- Trigger: Infection (molecular mimicry), autoimmunity, trauma, or idiopathic
- HLA-B27 mechanism: Presentation of arthritogenic peptides (derived from bacteria, self-antigens) to CD8+ T cells; HLA-B27 misfolding → ER stress → IL-23 production → Th17 activation
- T-cell activation: Predominantly CD4+ Th1 and Th17 cells infiltrate uveal tissue
- Cytokine cascade: IL-1β, IL-6, IL-17, IL-23, TNF-α, IFN-γ production
- Antigen presentation: Iris/ciliary body macrophages and dendritic cells present antigens (MHC class II upregulation)
Step 2: Blood-Aqueous Barrier Breakdown
Vascular Changes: [11]
- Increased vascular permeability: TNF-α, IL-1β, VEGF disrupt tight junctions
- Protein leakage: Albumin, immunoglobulins, complement enter anterior chamber → flare on slit lamp (Tyndall effect)
- Cellular infiltration: Neutrophils, lymphocytes, macrophages migrate into aqueous → cells on slit lamp (graded 0.5+ to 4+)
- Fibrin deposition: If severe inflammation → fibrinous exudate, hypopyon
Iris and Ciliary Body Changes:
- Iris vascular engorgement: Vasodilation → ciliary flush (limbal injection)
- Ciliary muscle spasm: Prostaglandin-mediated → photophobia, pain with accommodation
- Pupil changes: Sphincter spasm → miosis
Step 3: Keratic Precipitates Formation
Keratic Precipitates (KPs): [11]
- Inflammatory cells (macrophages, lymphocytes, epithelioid cells) adhere to corneal endothelium
- Settle inferiorly due to gravity → triangular distribution (Arlt's triangle)
- Fine KPs: Small, white, non-granulomatous inflammation (HLA-B27, idiopathic, viral)
- Mutton-fat KPs: Large, greasy, granulomatous inflammation (sarcoidosis, TB, sympathetic ophthalmia) — composed of epithelioid cells and macrophages
Step 4: Complications Pathway
Posterior Synechiae Formation: [9,10]
- Mechanism: Fibrin deposition between iris and anterior lens capsule → adhesions
- Risk factors: Delayed treatment, inadequate mydriasis, severe inflammation
- Consequences: Irregular pupil, impaired pupil dilation, pupil block if 360° (seclusio pupillae)
Glaucoma Mechanisms: [9,10]
- Open-angle: Trabecular meshwork obstruction by inflammatory cells, debris, fibrin
- Angle-closure: Pupil block from 360° synechiae (seclusio pupillae) → iris bombé → angle closure
- Steroid-induced: Prolonged topical steroid use → trabecular meshwork dysfunction
Cataract Formation: [9,10]
- Inflammatory cataract: Chronic inflammation → lens protein denaturation, posterior subcapsular opacities
- Steroid-induced cataract: Chronic topical/systemic steroid use → posterior subcapsular cataract
Cystoid Macular Oedema (CMO): [10]
- Mechanism: Chronic BAB breakdown → inflammatory mediators reach posterior segment → perifoveal capillary leakage → cystoid spaces
- Incidence: 10-30% of chronic/recurrent anterior uveitis
- Consequence: Central vision loss if persistent
Band Keratopathy:
- Mechanism: Chronic inflammation → calcium and phosphate deposition in Bowman's layer
- Appearance: Horizontal band across cornea, sparing limbal clear zone
- Treatment: EDTA chelation
Phthisis Bulbi (End-Stage):
- Chronic inflammation → ciliary body shutdown → aqueous production ceases → globe shrinks, becomes painful, blind
HLA-B27 Specific Pathophysiology [3,4,11,12]
Why HLA-B27 Causes Uveitis:
Theory 1: Molecular Mimicry [11,12]
- HLA-B27 presents bacterial peptides (Klebsiella, Chlamydia, Yersinia, Shigella) that share homology with self-antigens
- CD8+ T cells activated by bacterial antigens cross-react with uveal self-peptides (nitrogenase sequence homology)
- Evidence: Klebsiella urease epitopes cross-react with HLA-B27 molecules in 70% of AAU patients [12]
- Molecular mimicry between bacterial heat shock proteins (HSP) and human HSP60 in uveal tissue [11]
Theory 2: HLA-B27 Misfolding Hypothesis (Most Supported) [11,23]
- HLA-B27 heavy chains (especially B2705, B2702 subtypes) are prone to misfolding in endoplasmic reticulum (ER)
- ER stress triggers unfolded protein response (UPR) → activation of IRE1α, PERK, ATF6 pathways
- UPR activation → IL-23 production by antigen-presenting cells (dendritic cells, macrophages)
- IL-23 drives Th17 differentiation → IL-17A, IL-17F, IL-22 production → neutrophil chemotaxis and tissue inflammation
- Evidence: HLA-B27 misfolding detected in iris/ciliary body tissue from AAU patients (immunohistochemistry) [23]
- ER aminopeptidase 1 (ERAP1) polymorphisms modulate HLA-B27 misfolding risk (GWAS data) [23]
Theory 3: HLA-B27 Homodimer Formation [11,12]
- HLA-B27 heavy chains form β2-microglobulin-free homodimers on cell surface
- Homodimers recognized by killer immunoglobulin-like receptors (KIR3DL2) on NK cells, CD4+ T cells
- KIR3DL2 engagement → pro-inflammatory cytokine release (IFN-γ, TNF-α, IL-17)
- Cell surface homodimers detected in peripheral blood mononuclear cells (PBMCs) of HLA-B27+ AAU patients [12]
Theory 4: Gut-Eye Axis (Microbiome Hypothesis) [4,12,24]
- Key evidence: HLA-B27 transgenic rats develop SpA and AAU only in presence of gut microbiota (germ-free rats remain disease-free) [24]
- Dysbiosis in HLA-B27+ AAU: Reduced bacterial diversity, increased Bacteroides fragilis, decreased Faecalibacterium prausnitzii [24]
- Increased intestinal permeability ("leaky gut") → bacterial antigen translocation → systemic immune activation
- Dendritic cells present bacterial antigens to T cells → cross-reactive T cells migrate to uveal tissue via CCL20/CCR6 axis
- Anti-TNF therapy normalizes gut microbiome composition and reduces AAU flares by 51% [13,24]
- Clinical correlation: Subclinical gut inflammation detected by fecal calprotectin in 62% of HLA-B27+ AAU patients without IBD diagnosis [24]
Supporting Evidence Summary:
- GWAS studies: ERAP1, IL23R, and TLR4 polymorphisms associated with HLA-B27+ AAU [23]
- Animal models: HLA-B27 transgenic rats (spontaneous AAU in 40% by 6 months) [24]
- Clinical trials: Anti-IL-17 therapy (secukinumab) reduces AAU flares by 52% in AS patients (PREVENT trial) [20]
- Biomarker data: Elevated serum IL-23 (3.2-fold) and IL-17 (2.8-fold) during acute AAU flares vs remission [23]
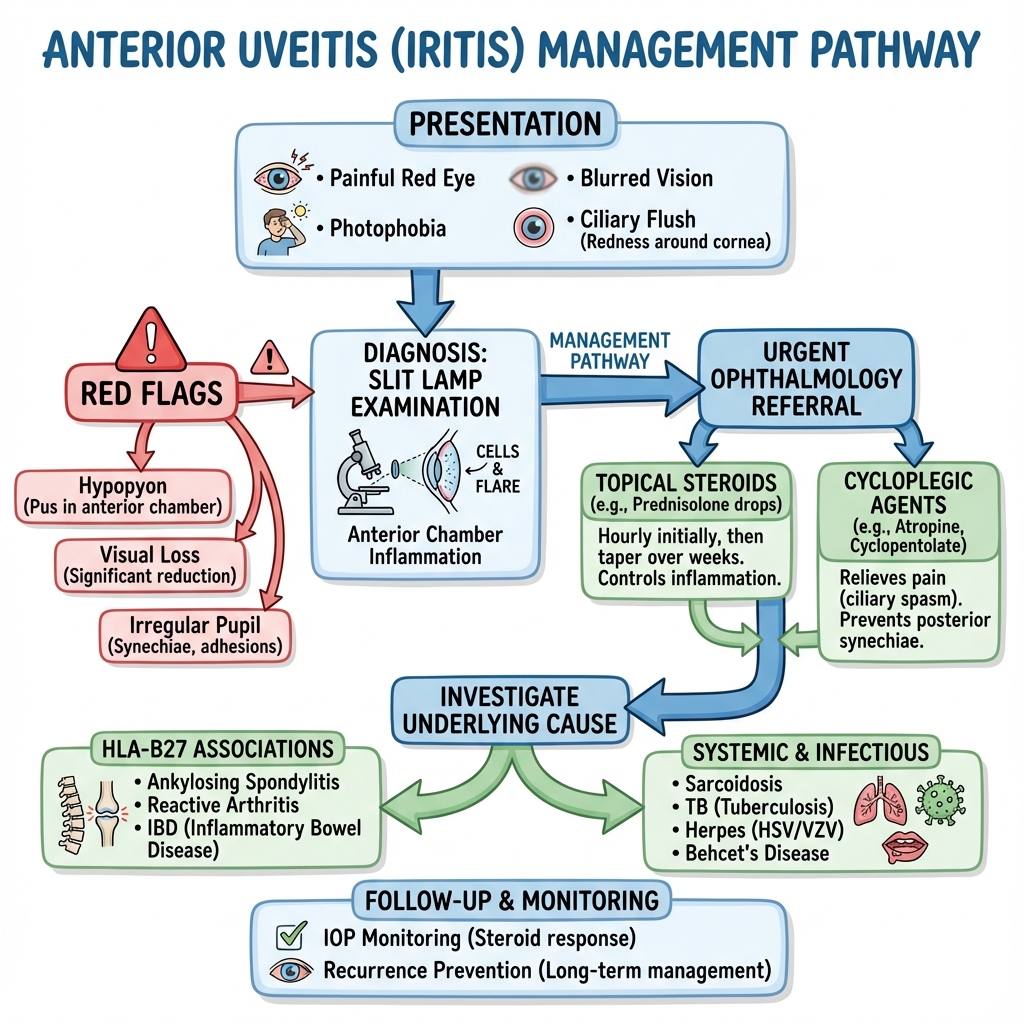
4. Clinical Presentation
Symptoms
Classic Triad:
- Pain: Deep, dull, aching pain (not sharp); worsens with accommodation (reading, near work); worse at night
- Photophobia: Consensual (light in unaffected eye → pain in affected eye due to bilateral ciliary spasm); often severe
- Redness: Red eye, especially circumcorneal (ciliary flush/limbal injection)
Additional Symptoms:
- Blurred vision: Due to aqueous cells/flare, miosis, ciliary spasm
- Lacrimation: Excessive tearing (reflex tearing from pain)
- Brow ache: Referred pain from ciliary spasm
- Floaters: If vitritis (suggests intermediate/posterior involvement)
- Usually unilateral: 80-85% of cases; bilateral simultaneous onset suggests systemic disease (sarcoidosis, Behçet's, Vogt-Koyanagi-Harada)
Onset Pattern:
- Acute: Sudden onset over hours to 1-2 days (typical of HLA-B27 AAU, infectious)
- Insidious: Gradual onset over days to weeks (granulomatous, JIA-associated)
Recurrence Pattern:
- 40-60% recur; often same eye, but can alternate [8]
- HLA-B27+ patients: 50-70% recurrence rate [8]
- Time to recurrence: Median 12-18 months; some recur within weeks
Signs
Structured Slit Lamp Examination
| Sign | Description | Significance | Grading |
|---|---|---|---|
| Ciliary Flush | Circumcorneal (limbal) ring of deep injection; blanches poorly with phenylephrine | Distinguishes from conjunctivitis (diffuse injection, blanches) | Present/Absent |
| Cells in AC | White blood cells floating in aqueous; count in 1 mm × 1 mm slit beam | Activity marker; guides treatment | 0, 0.5+, 1+, 2+, 3+, 4+ (SUN criteria) [14] |
| Flare | Protein in aqueous causing "beam" visible on slit lamp (Tyndall effect) | BAB breakdown; can persist after cells clear | 0, 1+, 2+, 3+, 4+ (SUN criteria) [14] |
| Keratic Precipitates (KPs) | Inflammatory deposits on posterior cornea; settle inferiorly (Arlt's triangle) | Fine = non-granulomatous; Mutton-fat = granulomatous | Fine, Medium, Mutton-fat |
| Miosis | Small pupil (sphincter spasm) | Due to prostaglandin release, ciliary spasm | Present/Absent |
| Posterior Synechiae | Iris adhesion to anterior lens capsule; irregular pupil | Complication; dilate to break early synechiae | Focal, Segmental, 360° (seclusio) |
| Hypopyon | White layering of inflammatory cells/pus in inferior AC | Severe inflammation; DDx: Behçet's, HLA-B27 severe, endophthalmitis, masquerade | Height (mm) |
| Iris Nodules | Koeppe (pupil margin), Busacca (iris surface) | Granulomatous; sarcoidosis, TB, VKH | Present/Absent, Location |
| Iris Atrophy | Sectoral iris depigmentation, transillumination defects | HSV/VZV uveitis; ischaemic iris atrophy | Sectoral/Diffuse |
SUN Working Group Grading of Anterior Chamber Cells: [14]
Cell Counting Technique:
- Use 1 mm × 1 mm slit beam at 45° angle
- Brightest focal point in anterior chamber (just in front of iris)
- Count cells in single focal plane (avoid counting cells in multiple planes)
- Time: Observe for at least 10-15 seconds to allow cells to float through beam
| Grade | Cells per Field (1 mm × 1 mm) | Clinical Correlation |
|---|---|---|
| 0 | less than 1 cell | No inflammation (quiet) |
| 0.5+ | 1-5 cells | Trace inflammation; often persistent after treatment |
| 1+ | 6-15 cells | Mild inflammation; outpatient management |
| 2+ | 16-25 cells | Moderate inflammation; close monitoring |
| 3+ | 26-50 cells | Severe inflammation; consider hospitalization |
| 4+ | > 50 cells (too many to count) | Very severe; hypopyon may be present |
Grading Pearls:
- Cells move (Brownian motion, convection currents); flare is static (protein scaffold)
- Pigment vs cells: Pigment (iris pigment dispersion) is golden-brown, angular; inflammatory cells are white, round
- Red blood cells (if hyphema): Larger, red; settle inferiorly as layered blood
- Inter-observer variability: ±1 grade is common; same examiner preferred for follow-up
SUN Working Group Grading of Anterior Chamber Flare: [14]
Flare Assessment Technique:
- Use 1-2 mm wide slit beam at 45° angle
- Assess Tyndall effect (protein scattering light, like a "beam" in dusty air)
- Compare iris and lens detail clarity
| Grade | Description | Iris and Lens Details | Clinical Correlation |
|---|---|---|---|
| 0 | None | Clear view of iris/lens | No BAB breakdown |
| 1+ | Faint (just detectable) | Iris/lens clear | Mild BAB breakdown; early AAU |
| 2+ | Moderate (clear beam visible) | Iris/lens details clear | Moderate BAB breakdown |
| 3+ | Marked (dense beam) | Iris/lens details hazy | Severe BAB breakdown; dense protein |
| 4+ | Intense (fibrin, plastic aqueous) | Iris/lens very hazy or obscured | Severe; fibrinous exudate, hypopyon likely |
Flare Persistence: [14]
- Flare lags behind cells: Cells clear first (responsive to steroids); flare may persist 2-4 weeks after cells resolve
- Chronic flare: If flare persists > 3 months at 1+ without cells → may indicate structural BAB damage (chronic uveitis, prior severe inflammation)
- Do NOT prolong steroids for persistent trace flare (0-1+) if cells are 0 — risk of steroid-induced glaucoma
Laser Flare Photometry (Objective Measurement):
- Kowa FM-600/700: Quantifies aqueous protein concentration (photon counts/ms)
- Normal: less than 5 photon counts/ms
- Mild flare: 10-20
- Moderate flare: 30-100
- Severe flare: > 100
- Use: Research, pediatric uveitis (difficult to grade in children), monitoring subclinical inflammation
- Availability: Specialist centers only
Red Flags
[!CAUTION] Red Flags — Require urgent investigation and specialist management:
- Hypopyon: Consider Behçet's disease, severe HLA-B27 flare, endophthalmitis, masquerade syndrome (leukaemia, lymphoma)
- Significant visual loss (VA less than 6/18): Posterior segment involvement, macular oedema, corneal oedema, severe inflammation
- Elevated IOP (> 30 mmHg): Risk of acute glaucoma, optic nerve damage; check for angle closure, steroid response
- Bilateral simultaneous onset: Strongly suggests systemic disease (sarcoidosis, Behçet's, VKH, sympathetic ophthalmia)
- Chronic/recurrent despite adequate treatment: Investigate for underlying cause, consider masquerade, non-compliance
- Posterior segment signs: Vitritis, chorioretinitis, vasculitis → not isolated anterior uveitis
- Age less than 6 years: JIA-associated uveitis (often asymptomatic, chronic, bilateral) — requires paediatric rheumatology
- Age > 60 years: Consider masquerade (lymphoma), VKH, drug-induced, infection
- Granulomatous inflammation + systemic symptoms: Sarcoidosis, tuberculosis, syphilis — extensive workup required
Distinguishing Features by Etiology
| Etiology | Clinical Clues | Laterality | KPs | IOP | Other Features |
|---|---|---|---|---|---|
| HLA-B27 AAU | Acute onset, severe pain, young adult, hx of back pain | Unilateral, alternating | Fine | Low/normal | Fibrin common, hypopyon occasional |
| HSV/VZV | Sectoral iris atrophy, high IOP, corneal dendrites (HSV), skin vesicles (VZV) | Unilateral | Fine to medium, sectoral | High (trabecular damage) | Iris transillumination, heterochromia |
| Sarcoidosis | Insidious, painless, multisystem symptoms (cough, lymphadenopathy) | Bilateral | Mutton-fat | Normal/low | Iris nodules, tent-shaped synechiae, retinal periphlebitis |
| Behçet's | Hypopyon (sterile), oral/genital ulcers, pathergy, young adult | Unilateral or bilateral | Fine | Normal | Retinal vasculitis, explosive onset |
| TB | Insidious, systemic symptoms, high-risk population | Unilateral or bilateral | Mutton-fat | Normal/low | Iris nodules, choroidal tubercles, periphlebitis |
| JIA (paediatric) | Asymptomatic, ANA+, oligoarticular JIA, age less than 6 | Bilateral | Fine | Normal/high | Band keratopathy, posterior synechiae, cataract |
| Fuchs' Heterochromic Iridocyclitis | Asymptomatic, unilateral, mild inflammation, NO synechiae | Unilateral | Fine, stellate | Normal | Heterochromia (affected eye lighter), cataract, vitreous opacities |
5. Investigations
Investigation Strategy
Anterior uveitis is a clinical diagnosis based on slit lamp examination. [6,14] Laboratory investigations aim to identify underlying systemic disease to guide treatment and predict recurrence.
Investigation Approach: [5,6,15,27]
Minimal Workup (Defer Investigations):
- Criteria: ALL of the following must be met:
- First episode
- Unilateral
- Non-granulomatous (fine KPs)
- Responds to topical steroids within 1-2 weeks (cells 0-0.5+)
- No systemic symptoms (no back pain, joint pain, cough, rash, oral/genital ulcers)
- No atypical features (no hypopyon, no iris atrophy, IOP normal)
- Age 20-60 years
- Rationale: 45-50% of AAU is idiopathic; if uncomplicated and responsive, investigations are low yield [5,27]
- Follow-up: Advise patient to report recurrence (if recurs → investigate)
Indications for Investigation (Any ONE of the Following): [5,6,15,27]
Episode Characteristics:
- Recurrent episodes (≥2 episodes; most important trigger for workup) [27]
- Bilateral simultaneous onset (suggests systemic disease: sarcoidosis, Behçet's, VKH, drug-induced)
- Chronic course (> 3 months despite treatment)
- Granulomatous features (mutton-fat KPs, iris nodules)
- Poor response to treatment (less than 50% reduction in cells after 2 weeks of intensive steroids)
Clinical Red Flags:
- Hypopyon (Behçet's, severe HLA-B27, endophthalmitis, masquerade)
- High IOP (> 25 mmHg; HSV/VZV, Fuchs' heterochromic iridocyclitis, steroid response)
- Iris atrophy (sectoral; HSV/VZV)
- Posterior segment involvement (vitritis, retinitis, vasculitis)
- Age extremes: less than 6 years (JIA) or > 60 years (masquerade, VKH, drug-induced)
Systemic Symptoms:
- Musculoskeletal: Back pain > 3 months, morning stiffness > 30 min, alternating buttock pain, heel pain (enthesitis), dactylitis → SpA workup [4,27]
- Respiratory: Cough, dyspnea, lymphadenopathy → sarcoidosis, TB [15,27]
- Mucocutaneous: Oral ulcers, genital ulcers, pathergy, erythema nodosum → Behçet's, sarcoidosis [15]
- Gastrointestinal: Chronic diarrhea, blood in stool, abdominal pain → IBD [4]
- Neurological: Headache, hearing loss, tinnitus, vitiligo → VKH [15]
- Constitutional: Fever, weight loss, night sweats → infection (TB, syphilis), lymphoma [15,27]
Structured Investigation Protocol:
Tier 1: First-Line Investigations (All Recurrent/Bilateral Cases) [5,6,15,27]
| Test | Purpose | Yield | Interpretation |
|---|---|---|---|
| HLA-B27 | SpA screening; recurrence prediction | Positive in 40-70% of AAU [3,8,27] | If +ve → rheumatology referral; assess for AS/SpA (exam, imaging); 50-70% recurrence risk [8,27] |
| Chest X-ray (PA + Lateral) | Sarcoidosis, TB screening | Abnormal in 10-15% of AAU workups [15,27] | BHL (bilateral hilar lymphadenopathy) → sarcoidosis; apical infiltrates/cavitation → TB; normal CXR does not exclude either [15] |
| FBC | Anaemia (IBD, chronic disease), lymphocytosis (sarcoid), eosinophilia (parasitic) | Abnormal in 5-10% [5] | Lymphopenia + history of infections → HIV testing; lymphocytosis + BHL → sarcoidosis |
| CRP/ESR | Systemic inflammation marker | Elevated in 30-40% of AAU with systemic disease [5,27] | Elevated → SpA, Behçet's, infection, sarcoid; Normal in idiopathic AAU [27] |
| Urinalysis (Dipstick + Microscopy) | TINU syndrome (tubulointerstitial nephritis), vasculitis | Abnormal in 2-5% [5] | Proteinuria, hematuria, WBC casts → renal imaging, nephrology referral; TINU = acute interstitial nephritis + AAU [5] |
Tier 2: Targeted Investigations (Based on Clinical Clues) [5,15,27]
| Clinical Clue | Investigation | Rationale | Yield |
|---|---|---|---|
| HLA-B27+ with back pain | MRI sacroiliac joints (STIR sequence); Sacroiliac X-ray | Detect sacroiliitis (AS/axial SpA) | MRI detects early inflammation (bone marrow edema) before X-ray changes [4,27] |
| Granulomatous (mutton-fat KPs) | Serum ACE, lysozyme; CT chest (HRCT); QuantiFERON-TB Gold | Sarcoidosis vs TB vs syphilis differentiation | ACE elevated in 30-60% of sarcoid uveitis; QuantiFERON +ve in 70-90% of TB uveitis [5,15,27] |
| High IOP + sectoral iris atrophy | HSV/VZV PCR (aqueous tap) | Viral uveitis (HSV/VZV) | PCR sensitivity 90-95%, specificity 95-99% [16,27] |
| Bilateral + systemic symptoms | Serum ACE, lysozyme, syphilis serology, QuantiFERON-TB | Sarcoid, TB, syphilis, Behçet's differentiation | Tailor to geographic risk and clinical presentation [15,27] |
| Sexually active / high-risk | Syphilis serology (VDRL/RPR + TPPA/FTA-Abs); HIV serology | Syphilis causes 1-3% of AAU; HIV risk factor for CMV/toxoplasma | Syphilis serology mandatory if sexually active + bilateral/granulomatous [15,27] |
| Endemic area (TB, histoplasmosis) | QuantiFERON-TB Gold or T-SPOT.TB; Histoplasma serology | Latent/active TB; histoplasmosis (Ohio/Mississippi River valleys) | QuantiFERON more sensitive than TST; not affected by BCG vaccination [5,15] |
| Oral/genital ulcers, pathergy | Behçet's: Clinical diagnosis; HLA-B51 (low sensitivity); pathergy test | Behçet's disease | No single diagnostic test; International Study Group criteria (clinical) [15] |
| Child less than 6 years | ANA, RF, anti-CCP; Rheumatology referral | JIA-associated uveitis | ANA +ve in ~80% of JIA-uveitis; RF usually negative (oligoarticular JIA) [15,27] |
| Elderly (> 60) + chronic | Aqueous tap (cytology, flow cytometry); Imaging (CT/MRI orbits, chest) | Masquerade syndrome (intraocular lymphoma, leukaemia) | Cytology detects lymphoma in 50-70% (may need repeated taps); vitrectomy biopsy more sensitive [15] |
Tier 3: Specialist Investigations (Tertiary Referral Centers) [15,27]
| Investigation | Indication | Findings | Evidence |
|---|---|---|---|
| Anterior Chamber Paracentesis (Aqueous Tap) | Suspected infection (HSV/VZV/CMV), masquerade, atypical/refractory | PCR (virus, TB, toxoplasma); Cytology (lymphoma); Culture (endophthalmitis) | PCR sensitivity > 90% for HSV/VZV/CMV; cytology 50-70% for lymphoma [16,27] |
| QuantiFERON-TB Gold / T-SPOT.TB | High-risk population, granulomatous, choroidal tubercles, BHL on CXR | Positive → latent or active TB; requires chest CT, ID referral | Sensitivity 70-90%; specificity 90-95%; false negatives in immunosuppressed [5,15] |
| Serum ACE | Sarcoidosis (low sensitivity, moderate specificity) | Elevated (> 52 U/L) supports sarcoid; normal does not exclude | Sensitivity 30-60%, specificity 80-90%; elevated in only 30-50% of ocular sarcoid [5,15,27] |
| Lysozyme | Sarcoidosis (slightly more sensitive than ACE) | Elevated in ~60% of sarcoid uveitis | Similar performance to ACE; not routinely superior [5] |
| ANA (Antinuclear Antibody) | JIA (paediatric), SLE, autoimmune overlap | Positive ANA in ~80% of JIA-uveitis (ANA titre ≥1:160); speckled or homogeneous pattern | High sensitivity in JIA; low specificity (positive in 5-15% of general population) [15,27] |
| Syphilis Serology | Sexually active, high-risk, granulomatous, chronic, bilateral | VDRL/RPR (screening; quantitative titre); TPPA/FTA-Abs (confirmatory) | Both tests required (VDRL may be negative in latent syphilis); treat with benzathine penicillin [15,27] |
| Toxoplasma IgG/IgM | Chorioretinitis, posterior uveitis (rare in isolated AAU) | IgG positive (seroprevalence 10-80% depending on region); rising IgM suggests active infection | IgG alone does not confirm active disease; typically posterior uveitis, not AAU [5] |
Specialist Investigations
| Investigation | Indication | Findings | Evidence |
|---|---|---|---|
| OCT Macula | Reduced vision, chronic uveitis | Cystoid macular oedema (10-30% of chronic AAU) | [10] |
| Fluorescein Angiography (FFA) | Suspected posterior involvement, vasculitis, CMO | Leakage patterns (macular oedema, vasculitis, disc leakage) | [10] |
| Indocyanine Green Angiography (ICG) | Suspected choroidal involvement (VKH, sarcoid, TB) | Choroidal granulomas, "fuzzy vessels" (VKH) | [15] |
| Ultrasound Biomicroscopy (UBM) | Angle assessment, ciliary body mass, cyst | Ciliary body cyst (Fuchs'), mass (lymphoma, melanoma) | [15] |
| B-Scan Ultrasound | Opaque media (dense cataract, vitreous haemorrhage) | Posterior segment pathology, choroidal mass | [15] |
| Anterior Chamber Paracentesis | Suspected infection (endophthalmitis), masquerade, atypical | PCR for HSV, VZV, CMV, TB, toxoplasma; cytology for lymphoma | [16] |
| Lumbar Puncture | Suspected CNS involvement (MS, Behçet's, sarcoid, VKH, syphilis) | Pleocytosis, elevated protein, positive serology/PCR | [15] |
Imaging for Systemic Disease
| Imaging | Indication | Findings | Evidence |
|---|---|---|---|
| Sacroiliac Joint X-ray | Suspected AS; HLA-B27+ with back pain | Sacroiliitis (erosions, sclerosis, fusion) — Grade 2-4 bilateral | [4] |
| MRI Sacroiliac Joints | Early SpA; negative X-ray but clinical suspicion | Bone marrow oedema (STIR high signal) — active inflammation | [4] |
| CT Chest (High-Resolution) | Suspected pulmonary sarcoidosis or TB | Sarcoid: lymphadenopathy, nodules, fibrosis; TB: cavitation, tree-in-bud | [15] |
| PET-CT | Sarcoidosis (extent of disease), malignancy (lymphoma) | FDG-avid lymph nodes, spleen, lungs (sarcoid); lymphoma staging | [15] |
6. Management
Distinguishing Clinical Features
Unilateral vs Bilateral:
- Unilateral (80-85%): HLA-B27 AAU, HSV/VZV, idiopathic, reactive arthritis, Fuchs' heterochromic iridocyclitis
- Bilateral simultaneous (15-20%): Sarcoidosis, Behçet's, VKH, sympathetic ophthalmia, drug-induced, JIA
- Unilateral alternating: HLA-B27 AAU (recurrent episodes switch eyes)
Acute vs Chronic:
- Acute (less than 3 months): HLA-B27 AAU, viral (HSV/VZV), reactive arthritis, idiopathic
- Chronic (> 3 months): JIA, sarcoidosis, Fuchs' heterochromic iridocyclitis, chronic idiopathic
Granulomatous vs Non-Granulomatous:
- Non-granulomatous (90%): Fine KPs, acute onset, HLA-B27, idiopathic, viral, Behçet's
- Granulomatous (10%): Mutton-fat KPs, insidious onset, sarcoidosis, TB, syphilis, VKH, sympathetic ophthalmia, Fuchs' (stellate KPs)
5A. Detailed Clinical Examination Techniques
Slit Lamp Biomicroscopy: Step-by-Step
Equipment Setup:
- Slit lamp with adjustable magnification (10x, 16x, 25x, 40x)
- Cobalt blue filter for fluorescein staining
- Red-free (green) filter for vasculature
- 1 mm × 1 mm slit beam for cell counting
Systematic Examination:
1. External Inspection (No Magnification):
- Pattern of redness: Ciliary flush (limbal) vs diffuse (conjunctivitis)
- Pupil size: Miotic (AAU), mid-dilated fixed (acute angle closure)
- Corneal clarity: Hazy (oedema from high IOP or severe inflammation)
- Discharge: None (AAU) vs mucopurulent (conjunctivitis)
2. Lids and Lashes (Low Magnification 10x):
- Blepharitis (may coexist)
- Meibomian gland dysfunction
- Herpes zoster vesicles on eyelid or forehead (dermatome distribution)
3. Conjunctiva and Sclera (10-16x):
- Conjunctival injection: Diffuse (conjunctivitis) vs limbal sparing (anterior uveitis)
- Ciliary flush: Deep episcleral vessel injection around limbus; does NOT blanch with 2.5% phenylephrine (distinguishes from conjunctivitis)
- Scleritis: Blue-grey scleral discoloration, deeper pain (associated with systemic autoimmune disease)
4. Cornea (16-25x):
- Keratic precipitates: Examine inferior cornea (Arlt's triangle); grade size (fine, medium, mutton-fat), distribution (diffuse vs sectoral)
- Corneal oedema: Descemet's folds, thickening (high IOP, endothelial dysfunction)
- Dendrites: HSV keratitis (exclude before diagnosing pure uveitis); fluorescein staining reveals branching ulcer
- Band keratopathy: Horizontal calcium band in Bowman's layer (chronic uveitis, juvenile uveitis)
- Endothelial touch: Lens-cornea touch (aphakic/pseudophakic patients, shallow AC)
5. Anterior Chamber (16-25x):
Cell Grading (SUN Criteria): [14]
- Use 1 mm × 1 mm slit beam at 45° angle
- Count cells in aqueous at brightest part of beam
- Grade 0: less than 1 cell
- Grade 0.5+: 1-5 cells
- Grade 1+: 6-15 cells
- Grade 2+: 16-25 cells
- Grade 3+: 26-50 cells
- Grade 4+: > 50 cells (too many to count)
Flare Grading (SUN Criteria): [14]
- Tyndall effect: Protein scatter visible as "beam" in aqueous
- Grade 0: None
- Grade 1+: Faint (just detectable)
- Grade 2+: Moderate (iris and lens details clear)
- Grade 3+: Marked (iris and lens details hazy)
- Grade 4+: Intense (fibrin clumps, "plastic aqueous")
Hypopyon:
- White/yellow layering of cells inferiorly
- Measure height in mm
- Mobile (shifts with head position): Inflammatory (Behçet's, severe HLA-B27)
- Fixed: May suggest fibrin or lens matter
Hyphema (Blood in AC):
- Layering of red blood cells
- Suggests trauma, neovascularization (rubeosis), anticoagulation
Anterior Chamber Depth:
- Deep: Normal in uveitis
- Shallow: Angle closure glaucoma (emergency), seclusio pupillae (iris bombé from 360° synechiae)
6. Iris (16-25x):
- Colour: Heterochromia (Fuchs', HSV/VZV chronic)
- Pattern: Crypts, furrows (normal architecture lost in chronic inflammation)
- Nodules:
- "Koeppe nodules: At pupil margin (granulomatous)"
- "Busacca nodules: On iris surface (granulomatous)"
- Atrophy: Sectoral (HSV/VZV), diffuse (chronic uveitis, ischaemia)
- Transillumination defects: Shine light from side; iris defects glow (HSV/VZV, pigment dispersion, trauma)
- Neovascularization (rubeosis): New vessels at pupil margin or iris surface (ischaemia: CRVO, diabetic retinopathy, ocular ischaemic syndrome)
- Posterior synechiae:
- "Focal: Small adhesions (break with dilation)"
- "Segmental: Larger areas (may partially break)"
- "360° (seclusio pupillae): Complete adhesion → pupil block → iris bombé → angle closure"
7. Lens (16-25x):
- Cataract: Posterior subcapsular (inflammation, steroids), cortical, nuclear
- Anterior capsule deposits: Inflammatory cells, fibrin, pigment (Vossius ring after trauma)
- Posterior capsule: Pigment (from posterior synechiae breakage)
- Phacodonesis: Lens wobble (zonular weakness)
8. Vitreous (Examine Through Dilated Pupil, 16-25x):
- Should be clear in isolated anterior uveitis
- Cells in anterior vitreous: Suggests intermediate uveitis or panuveitis, not pure anterior
- Snowballs: White inflammatory aggregates (intermediate uveitis, sarcoidosis)
- Haemorrhage: Vitreous haemorrhage (posterior involvement, trauma)
9. Intraocular Pressure (Tonometry):
- Goldmann applanation tonometry (gold standard): Flatten cornea with tonometer; read pressure from dial
- Icare rebound tonometry (alternative): Does not require anaesthesia
- Normal: 10-21 mmHg
- Low (less than 10 mmHg): Ciliary shutdown (acute AAU), hypotony (chronic inflammation)
- High (> 21 mmHg): Trabecular obstruction, steroid response, angle closure
- Very high (> 30 mmHg): Acute glaucoma (check for angle closure, corneal oedema, fixed mid-dilated pupil)
10. Gonioscopy (If IOP Elevated):
- Purpose: Assess anterior chamber angle (open vs narrow vs closed)
- Technique: Use goniolens (Goldman, Zeiss) with coupling gel; rotate to examine 360° of angle
- Findings:
- "Open angle: Trabecular meshwork visible"
- "Narrow angle: Iris close to trabecular meshwork (risk of angle closure)"
- "Closed angle: Iris apposition to trabecular meshwork (angle closure glaucoma)"
- "Peripheral anterior synechiae (PAS): Permanent iris adhesions to angle (chronic uveitis, previous angle closure)"
- "Pigmentation: Heavy pigment on trabecular meshwork (pigment dispersion, pseudoexfoliation, chronic inflammation)"
- "Neovascularization: New vessels crossing scleral spur (rubeotic glaucoma)"
11. Dilated Fundoscopy (Essential):
- Dilate with tropicamide 1% + phenylephrine 2.5% (wait 20-30 minutes)
- Purpose: Rule out posterior segment involvement (intermediate, posterior, panuveitis)
- Vitreous: Should be clear; cells/haze suggests spillover or posterior involvement
- Optic Disc: Normal colour, margins, cup-to-disc ratio; swelling suggests papillitis, sarcoid, neuroretinitis
- Macula:
- "Cystoid macular oedema (CMO): Reduced foveal reflex, retinal thickening (confirm with OCT)"
- "Epiretinal membrane: Cellophane reflex, wrinkling"
- "Macular scar: Previous CMO, toxoplasmosis"
- Retina:
- "Normal: No lesions, haemorrhages, exudates"
- Vasculitis: Perivascular sheathing, "candle-wax drippings" (sarcoidosis)
- "Retinitis: White/yellow lesions (toxoplasmosis, CMV, HSV/VZV, syphilis)"
- "Haemorrhages: Flame-shaped (CRVO), dot-blot (diabetic retinopathy), white-centred (Roth spots in bacterial endocarditis)"
- Choroid:
- "Choroidal granulomas: Yellow-white lesions (sarcoidosis, TB, VKH)"
- "Choroidal tubercles: Small yellow lesions (miliary TB, disseminated TB)"
- "Serpiginous choroiditis: Geographic atrophy (autoimmune choroiditis)"
Ancillary Testing
Fluorescein Staining:
- Purpose: Detect corneal epithelial defects (dendrites, ulcers, abrasions)
- Technique: Instill fluorescein drop; examine with cobalt blue filter on slit lamp
- Findings:
- "Dendrites: Branching epithelial ulcer (HSV keratitis)"
- "Geographic ulcer: Amoeboid shape (VZV keratitis, acanthamoeba)"
- "Punctate epithelial erosions: Dry eye, toxic keratopathy"
- "Corneal ulcer: Infectious keratitis (bacterial, fungal)"
Anterior Segment Optical Coherence Tomography (AS-OCT):
- Purpose: High-resolution cross-sectional imaging of anterior segment
- Indications: Angle assessment (if gonioscopy difficult), corneal pathology, anterior chamber depth measurement
- Findings: Angle configuration, iris-lens contact, corneal thickness
5B. Differential Diagnosis: The Red Eye
Comprehensive Red Eye Differential
| Condition | Pain | Photophobia | Vision | Discharge | Pupil | IOP | Cornea | AC | Key Features |
|---|---|---|---|---|---|---|---|---|---|
| Anterior Uveitis | Deep ache | Severe (consensual) | Blurred | Watery | Small, irregular | Low/normal/high | KPs | Cells, flare | Ciliary flush, cells in AC |
| Conjunctivitis (viral) | Gritty | None | Normal | Watery | Normal | Normal | Clear | Clear | Diffuse injection, preauricular lymph node |
| Conjunctivitis (bacterial) | Gritty | None | Normal | Mucopurulent | Normal | Normal | Clear | Clear | Purulent discharge, morning crusting |
| Allergic Conjunctivitis | Itchy | None | Normal | Watery | Normal | Normal | Clear | Clear | Bilateral, itching, chemosis, papillae |
| Episcleritis | Mild | Mild | Normal | None | Normal | Normal | Clear | Clear | Localized sector redness, mobile |
| Scleritis | Severe, boring | Moderate | Blurred if posterior | None | Normal | Normal | Clear | Clear | Blue-grey sclera, systemic disease association |
| Acute Angle Closure | Severe | Moderate | Very blurred | None | Mid-dilated, fixed | Very high (> 40) | Oedematous | Shallow | Halos, headache, nausea, vomiting |
| Keratitis (bacterial) | Moderate-severe | Severe | Blurred | Purulent | Normal | Normal | Infiltrate, ulcer | Clear/cells | Corneal defect, hypopyon possible |
| Keratitis (viral HSV) | Moderate | Severe | Blurred | Watery | Normal | Normal | Dendrites | Clear | Fluorescein+ dendrite, reduced sensation |
| Endophthalmitis | Severe | Severe | Very poor | None | Poorly reactive | Low/normal | Hazy | Severe cells, hypopyon | Recent surgery/trauma, vitritis |
| Subconjunctival Haemorrhage | None | None | Normal | None | Normal | Normal | Clear | Clear | Bright red patch, painless, history of Valsalva/anticoagulation |
| Dry Eye Syndrome | Gritty, burning | Mild | Fluctuating | None/mucus | Normal | Normal | SPK | Clear | Symptoms worse evening, Schirmer's test low |
| Pterygium | Mild/none | None | Normal (unless central) | None | Normal | Normal | Fibrovascular growth | Clear | Nasal > temporal, outdoor workers |
| Chemical Injury | Severe | Severe | Blurred/very poor | Watery | Variable | Variable | Oedema, necrosis | Cells if severe | History of chemical splash, limbal ischaemia |
Clinical Decision Rules
"Red Eye Referral Rules" (Primary Care):
SAME-DAY Ophthalmology Referral:
- Moderate-severe pain
- Photophobia (especially consensual)
- Reduced vision
- Irregular pupil
- Ciliary flush (limbal injection)
- Corneal infiltrate/ulcer
- Hypopyon
- History of trauma
URGENT (Within 24 Hours) Referral:
- Mild pain with redness unresponsive to lubricants
- Recurrent red eye episodes
- Contact lens wearer with red eye
- Suspected scleritis
Routine Referral:
- Chronic red eye without pain or vision change
- Pterygium interfering with vision
- Recurrent subconjunctival haemorrhage
Manage in Primary Care:
- Clear viral/bacterial/allergic conjunctivitis
- Subconjunctival haemorrhage (first episode)
- Dry eye syndrome
- Blepharitis
6A. Management Algorithms
Algorithm 1: Initial Assessment and Triage
PATIENT PRESENTS WITH PAINFUL RED EYE
|
v
URGENT OPHTHALMOLOGY ASSESSMENT
|
v
SLIT LAMP EXAMINATION
|
|--- Cells + Flare in AC? --> YES --> ANTERIOR UVEITIS CONFIRMED
| |
| v
| Grade Severity
| (Cells, Flare, Synechiae)
| |
| v
| START TREATMENT:
| - Prednisolone acetate 1% hourly
| - Cyclopentolate 1% TDS
| (or Atropine 1% BD if severe)
| |
| v
| Check IOP, Dilated Fundoscopy
| |
| v
| Arrange Follow-Up (3-7 days)
|
|--- NO cells in AC --> ALTERNATIVE DIAGNOSIS
- Conjunctivitis
- Episcleritis
- Scleritis
- Keratitis
- Angle closure
Algorithm 2: Investigation Strategy
ANTERIOR UVEITIS CONFIRMED
|
v
FIRST EPISODE?
|
+------+------+
| |
YES NO (Recurrent/Chronic)
| |
v v
UNILATERAL? INVESTIGATE:
| - HLA-B27
+----+ - CXR
| | - FBC, CRP/ESR
YES NO - Syphilis serology
| | - QuantiFERON-TB (if high risk)
v v - Serum ACE (if granulomatous)
RAPID BILATERAL
RESPONSE? OR CHRONIC
| |
YES NO v
| | EXTENSIVE WORKUP:
v v - All above investigations
MINIMAL INVESTIGATE: - OCT macula
WORKUP - HLA-B27 - Consider aqueous tap if atypical
Consider - CXR - Rheumatology/ID referral
deferring - MRI SI joints if HLA-B27+
tests
Algorithm 3: Treatment Escalation
ANTERIOR UVEITIS TREATMENT
|
v
TOPICAL STEROIDS + CYCLOPLEGICS
(Prednisolone acetate 1% hourly + Cyclopentolate TDS)
|
v
REVIEW IN 3-7 DAYS
|
+------+------+
| |
IMPROVING NOT IMPROVING
(Cells (Cells same
decreasing) or worse)
| |
v v
CONTINUE ESCALATE:
TAPER SLOWLY - Increase steroid frequency
(4-8 weeks) - Switch to stronger steroid (dexamethasone)
| - Add atropine if not already
| - Check compliance
| - Consider:
v * Periocular steroid injection
CELLS CLEAR * Systemic steroids (if bilateral/severe)
| * Aqueous tap if infection suspected
v
SLOW TAPER:
Week 1-2: Hourly
Week 3-4: 2-hourly
Week 5-6: QDS
Week 7-8: TDS
Week 9-10: BD
Week 11-12: OD, then stop
|
v
MONITOR FOR RECURRENCE
|
+--------- ≥3 RECURRENCES/YEAR? -------+
| |
NO YES
| |
v v
TREAT FLARES CONSIDER PROPHYLAXIS:
AS THEY OCCUR - Systemic immunosuppression
(Methotrexate, azathioprine)
- Anti-TNF (if HLA-B27+/AS)
Algorithm 4: Complication Management
COMPLICATION DETECTED
|
+------+------+------+------+
| | | | |
SYNECHIAE IOP↑ CATARACT CMO BAND KERATOPATHY
| | | | |
v v v v v
Intensive High-dose Post-op Intensive EDTA
mydriatics IOP drops when steroids chelation
Atropine (timolol, quiet Periocular
1% QDS dorzolamide) ≥3 steroid
Consider Avoid months injection
subconj PGAs in Oral
mydriatic active acetazo-
if severe inflam. lamide
Management Principles
Goals of Treatment:
- Reduce inflammation → prevent complications (synechiae, glaucoma, cataract, CMO)
- Relieve pain and photophobia
- Prevent recurrence (if possible, by treating underlying disease)
- Preserve vision
Key Principles:
- Urgent ophthalmology referral — same-day or next-day review [6,7]
- Topical corticosteroids — mainstay of treatment [6,7,17]
- Cycloplegic/mydriatic agents — relieve pain, prevent/break synechiae [6,7]
- Treat underlying cause — if identified (SpA, sarcoid, infection)
- Slow taper — rebound inflammation if stopped too quickly [6,7]
- Monitor IOP — risk of steroid-induced glaucoma [6]
Immediate Management
| Action | Details | Evidence |
|---|---|---|
| Urgent ophthalmology referral | Same-day if available; next-day if not; phone advice for temporising measures | [6,7] |
| Do NOT | Prescribe antibiotics (not infection unless proven); delay referral; use oral steroids without specialist advice | [6] |
| Temporising (if ophthalmology unavailable) | Cyclopentolate 1% drops TDS (pain relief) ± prednisolone acetate 1% hourly (if confirmed uveitis, not keratitis) | [6] |
Pharmacological Management
Topical Corticosteroids [6,7,17,25]
First-Line Agents:
| Drug | Penetration | Potency | Initial Dose | Duration | Evidence |
|---|---|---|---|---|---|
| Prednisolone acetate 1% | Excellent (suspension) | High | q1h while awake (8-10×/day) | Until cells clear, then taper 4-8 weeks | Gold standard; superior to dexamethasone for AAU [17,25] |
| Dexamethasone 0.1% | Good (solution) | High | q1h → taper | Alternative if prednisolone unavailable | [17,25] |
| Difluprednate 0.05% | Superior | Very high | QDS initially | 4-6 weeks | Newer agent; 4× more potent than prednisolone; faster cell clearance [25] |
Penetration Considerations:
- Suspension > Solution: Prednisolone acetate (suspension) achieves 3-5× higher aqueous concentrations than prednisolone phosphate (solution) [25]
- Shake well: Prednisolone acetate must be shaken to resuspend particles
- Intact epithelium required: Topical steroids penetrate through intact corneal epithelium; epithelial defect reduces efficacy
Evidence-Based Dosing Protocol (Prednisolone Acetate 1%): [6,7,17]
Initial Phase (Week 1-2):
- Severe inflammation (3-4+ cells): 1 drop every 1 hour while awake (q1h; 8-10 doses/day)
- Moderate inflammation (2+ cells): 1 drop every 2 hours (q2h; 6-8 doses/day)
- Mild inflammation (0.5-1+ cells): 1 drop every 3-4 hours (QID; 4-6 doses/day)
- Continue until cell count reduces to ≤0.5+
Tapering Phase (Individualized Based on Response):
Standard Taper (4-8 Weeks Total): [6,7]
- Week 1: q1h (if started at this frequency)
- Week 2: q2h (50% reduction if cells improving)
- Week 3: QID (4×/day)
- Week 4: TDS (3×/day)
- Week 5: BD (2×/day)
- Week 6: Once daily (OD)
- Week 7-8: Alternate days or stop
Slow Taper (Chronic/Recurrent Cases; 8-12 Weeks): [17]
- Use gentler taper: Reduce by 1 drop every 1-2 weeks
- Monitor for rebound inflammation at each step
- Consider maintenance therapy (BD or OD) for chronic cases
Rapid Taper (Acute, Mild, First Episode; 4-6 Weeks):
- Reduce every 3-5 days if cells clear rapidly
- Example: q2h (3 days) → QID (7 days) → TDS (7 days) → BD (7 days) → OD (7 days) → stop
Monitoring During Taper:
- Weekly reviews during taper (at minimum)
- Cell count at each visit: If cells increase, step back up in frequency
- IOP measurement: At every visit (steroid-induced glaucoma risk 5-30%) [6]
- Flare persistence: Flare may persist 2-4 weeks after cells clear (acceptable; do not prolong steroids unnecessarily)
Alternative Steroids (Second-Line/Special Situations):
| Indication | Agent | Rationale |
|---|---|---|
| Steroid non-responder | Switch to difluprednate 0.05% | Higher potency; greater penetration [25] |
| Epithelial defect present | Dexamethasone 0.1% (solution) | Less cytotoxic to epithelium than acetate suspension |
| Severe IOP elevation | Switch to fluorometholone 0.1% or loteprednol 0.5% | Lower IOP risk (but also lower efficacy) [6] |
| Chronic maintenance | Loteprednol 0.5% BD-OD | Steroid-sparing; reduced IOP risk |
Steroid-Induced Glaucoma: [6,9]
- Incidence: 5-30% of patients (genetic predisposition; myocilin gene polymorphisms)
- Timing: IOP rise typically 2-6 weeks after initiation; reversible within 2-4 weeks of cessation
- Management:
- Switch to lower-potency steroid (fluorometholone, loteprednol)
- Add IOP-lowering drops (timolol, dorzolamide, brimonidine)
- Avoid prostaglandin analogues (latanoprost, bimatoprost) in active inflammation — may worsen CMO [21]
- Consider periocular steroid injection (depot triamcinolone 40 mg) if topical steroids must be stopped but inflammation persists
Rebound Inflammation: [6,7]
- Incidence: 10-20% if tapered too quickly
- Prevention: Slow taper (halve frequency every 1-2 weeks, not every few days)
- Management: Increase steroid frequency back to previous effective dose; consider longer taper
Cycloplegic/Mydriatic Agents [6,7,26]
Mechanisms of Action:
- Cycloplegia: Paralysis of ciliary muscle → relieves ciliary spasm → reduces pain and photophobia
- Mydriasis: Pupil dilation → breaks early posterior synechiae → prevents iris-lens adhesions
- Pain relief: Ciliary muscle relaxation reduces referred trigeminal nerve pain (brow ache)
Agent Selection Based on Clinical Scenario:
| Drug | Onset | Duration | Peak Effect | Indications | Evidence |
|---|---|---|---|---|---|
| Cyclopentolate 1% | 30 min | 6-24 hours | 30-60 min | Mild-moderate AAU (1-2+ cells), no synechiae, first-line | Shorter duration; better tolerated; allows daily pupil assessment [6,26] |
| Homatropine 2% | 30-60 min | 1-3 days | 1-2 hours | Moderate AAU with early synechiae; intermediate duration needed | Balance between cyclopentolate and atropine [6] |
| Atropine 1% | 30-60 min | 7-14 days | 3-4 hours | Severe AAU (3-4+ cells), posterior synechiae present, high fibrin | Long duration ensures sustained mydriasis; prevents synechiae formation [6,7,26] |
| Tropicamide 1% | 15-20 min | 4-6 hours | 20-40 min | Diagnostic mydriasis only (fundus exam); insufficient duration for therapeutic use | NOT recommended for AAU treatment [6] |
Evidence-Based Indications for Each Agent:
Cyclopentolate 1% (First-Line; 60-70% of Cases): [6,26]
- Mild-moderate AAU: 0.5-2+ cells, minimal flare
- No posterior synechiae
- First episode or responsive to previous treatment
- Dose: 1 drop TDS (3×/day) or QDS (4×/day) if pain persists
- Advantages: Can assess pupil daily to monitor synechiae formation; wears off if stopped → easier return to normal vision
- Disadvantages: May need to escalate to atropine if synechiae develop
Homatropine 2% (Intermediate; 10-15% of Cases): [6]
- Moderate AAU: 2-3+ cells
- Early/focal posterior synechiae (small adhesions detected on slit lamp)
- Pain inadequately controlled with cyclopentolate
- Dose: 1 drop BD-TDS
- Advantages: Longer mydriasis than cyclopentolate, shorter than atropine
- Disadvantages: Less commonly stocked; variable duration (patient-dependent)
Atropine 1% (Severe Cases; 20-30% of Cases): [6,7,26]
- Severe AAU: 3-4+ cells, marked flare
- Posterior synechiae present (focal, segmental, or 360°)
- High fibrin or hypopyon
- Pediatric AAU (especially JIA-associated)
- Chronic/recurrent AAU with history of synechiae
- Dose: 1 drop BD (2×/day); can reduce to OD once synechiae broken
- Advantages: Sustained mydriasis (7-14 days) ensures synechiae prevention; superior for breaking established synechiae
- Disadvantages: Prolonged blurred near vision (accommodation paralyzed); photophobia (advise sunglasses); cannot assess pupil reactivity for 1-2 weeks
Evidence for Synechiae Prevention: [6,9,26]
- Without cycloplegia: Posterior synechiae develop in 40-60% of moderate-severe AAU [9]
- With cycloplegics: Synechiae incidence reduced to 15-20% [6,9]
- Early atropine use (within 24-48 hours of symptom onset) reduces synechiae by 60-70% vs delayed use [26]
Breaking Established Synechiae:
- Method: Intensive atropine 1% QDS for 2-3 days
- Adjunct: Subconjunctival mydriatic injection (adrenaline 1:1000 0.5 mL) if synechiae fail to break with atropine alone
- Surgical synechiolysis: Reserved for 360° synechiae (seclusio pupillae) unresponsive to medical therapy
Contraindications and Precautions: [6,26]
- Angle-closure glaucoma risk: Avoid cycloplegics if anterior chamber shallow (risk of pupil block in predisposed eyes)
- "Assessment: Van Herick test (AC depth at limbus); if AC depth less than 1/4 corneal thickness → gonioscopy before cycloplegia"
- "Management: If narrow angle, use lower concentration (cyclopentolate 0.5%) or use pilocarpine 2% alongside to prevent angle closure"
- Pediatric use: Atropine 0.5% in children less than 3 years (reduce systemic absorption risk)
- Elderly: Risk of confusion, urinary retention (atropine anticholinergic effects); use with caution
- Cardiac patients: Atropine may cause tachycardia (rare with topical use, but caution in severe cardiac disease)
Patient Counseling: [6]
- Blurred near vision: Expected; advise against driving initially; use reading glasses if needed
- Photophobia: Wear sunglasses outdoors
- Duration: Cyclopentolate wears off in 6-24 hours; atropine lasts 7-14 days
- Do not stop suddenly: Continue until instructed by ophthalmologist (typically stop when cells clear and steroids tapered)
Systemic Therapy
Immunosuppressive Therapy [13,15,18]
Indications:
- Recurrent episodes (≥3 per year)
- Chronic uveitis requiring > 10 mg/day oral prednisolone
- Bilateral vision-threatening uveitis
| Drug | Mechanism | Dose | Monitoring | Evidence |
|---|---|---|---|---|
| Methotrexate | DHFR inhibitor | 10-25 mg PO/SC weekly + folic acid 5 mg daily | FBC, LFTs, U&Es every 4-8 weeks | 50-70% response [18] |
| Azathioprine | Purine synthesis inhibitor | 1-2.5 mg/kg/day PO | FBC, LFTs, TPMT pre-treatment | 60% response [18] |
| Mycophenolate mofetil | Purine synthesis inhibitor | 500-1500 mg BD PO | FBC, U&Es every 4-8 weeks | 60-70% response [18] |
Biologic Therapy [13,18,19]
| Drug | Mechanism | Dose | Evidence |
|---|---|---|---|
| Adalimumab | Anti-TNF-α mAb | 40 mg SC every 2 weeks | VISUAL I & II: Reduced flares by 50% [19] |
| Infliximab | Anti-TNF-α mAb | 5 mg/kg IV at 0, 2, 6 weeks, then q8 weeks | Effective for Behçet's, refractory AAU [13,18] |
| Secukinumab | Anti-IL-17A mAb | 150 mg SC monthly | PREVENT: Reduces AAU flares in AS [20] |
Follow-Up and Monitoring
Acute Episode:
- Week 1: Review in 3-7 days
- Week 2-4: Weekly review
- Week 4-8: Bi-weekly, then monthly
- Resolution: Cells 0, flare 0-1+, steroids stopped
Recurrent AAU:
- Consider investigation after 2nd episode
- Rheumatology referral if HLA-B27+ or clinical SpA features
- Consider prophylactic immunosuppression if ≥3 flares/year
7. Complications
Acute Complications
| Complication | Incidence | Mechanism | Management | Evidence |
|---|---|---|---|---|
| Posterior Synechiae | 20-40% if untreated | Fibrin deposition between iris and lens | Intensive mydriatics, steroids | [9] |
| Hypopyon | 5-10% (severe AAU) | Layering of inflammatory cells | Intensive steroids; rule out infection | [6] |
| Secondary Glaucoma | 10-25% | Trabecular obstruction, angle closure | IOP-lowering drops; treat inflammation | [9,10] |
Chronic Complications
| Complication | Incidence | Mechanism | Management | Evidence |
|---|---|---|---|---|
| Cataract | 30-50% (chronic AAU) | Chronic inflammation, steroid use | Phacoemulsification when quiet ≥3 months | [9,10] |
| Cystoid Macular Oedema | 10-30% (chronic AAU) | BAB breakdown → macular capillary leakage | Intensive steroids, periocular injection, oral acetazolamide | [10] |
| Chronic Glaucoma | 10-20% | Trabecular damage, steroid response, synechiae | IOP-lowering drops; trabeculectomy if uncontrolled | [9,10] |
| Band Keratopathy | 5-15% (chronic AAU) | Calcium deposition in Bowman's layer | EDTA chelation | [9] |
| Phthisis Bulbi | less than 1% (end-stage) | Chronic ciliary shutdown | Enucleation if painful | [9] |
Treatment-Related Complications
| Treatment | Complication | Incidence | Management |
|---|---|---|---|
| Topical Steroids | Steroid-induced glaucoma | 5-30% | Switch to weaker steroid, add IOP drops |
| Topical Steroids | Cataract (PSC) | 10-30% (chronic) | Surgical extraction when needed |
| Topical Steroids | Rebound inflammation | 10-20% | Slow taper (4-8 weeks) |
| Cycloplegics | Blurred near vision | 100% | Warn patient; resolves when stopped |
| Methotrexate | Hepatotoxicity, marrow suppression | 5-15% | Monitor FBC, LFTs; reduce dose or stop |
| Anti-TNF | Infection (TB reactivation) | 1-5% | Screen for latent TB; prophylaxis |
Vision Loss
Incidence: 5-10% develop significant vision loss (VA less than 6/18) [10]
Causes:
- Cystoid macular oedema (most common reversible cause)
- Glaucoma (optic nerve damage)
- Cataract (reversible with surgery)
- Band keratopathy
- Hypotony maculopathy
8. Prognosis & Outcomes
Natural History
Acute Anterior Uveitis:
- Resolves within 4-8 weeks with treatment [6,7]
- Recurrence: 40-60% [8]
- Time to recurrence: Median 12-18 months
HLA-B27 Positive AAU: [3,8]
- Recurrence: 50-70% (vs 30-40% HLA-B27 negative)
- More severe inflammation
- Earlier age of onset
Outcomes
| Outcome | Rate | Evidence |
|---|---|---|
| Resolution (first episode) | 95%+ | [6,7] |
| Recurrence (overall) | 40-60% | [8] |
| Recurrence (HLA-B27+) | 50-70% | [3,8] |
| Chronic uveitis | 10-15% | [1] |
| Vision loss | 5-10% overall; 10-25% chronic | [10] |
| Cataract (chronic) | 30-50% | [9,10] |
| Glaucoma (chronic) | 10-30% | [9,10] |
| CMO (chronic) | 10-30% | [10] |
Prognostic Factors
Good Prognosis:
- First episode
- Acute presentation
- Non-granulomatous
- Good response to topical steroids
- No synechiae at presentation
- HLA-B27 negative
Poor Prognosis:
- Recurrent episodes (≥3)
- Chronic course (> 3 months)
- HLA-B27 positive
- Granulomatous inflammation
- Bilateral simultaneous onset
- Systemic disease (Behçet's, JIA, sarcoidosis)
- Age less than 6 years (JIA)
- Delayed treatment (> 1 week)
Long-Term Outcomes by Etiology
| Etiology | Recurrence Risk | Vision Loss Risk | Key Features |
|---|---|---|---|
| Idiopathic AAU | 30-40% | 5-10% | Good prognosis |
| HLA-B27 AAU | 50-70% | 10-15% | Frequent recurrences; good vision if treated early |
| AS-associated AAU | 50-70% | 10-15% | Controlled with anti-TNF for AS |
| Behçet's AAU | 80-90% | 25-50% | Severe, recurrent; high vision loss risk |
| Sarcoidosis | 50-70% | 15-30% | Chronic; bilateral; CMO common |
| HSV/VZV | 30-50% | 15-25% | Glaucoma common; iris atrophy |
| JIA-associated | 70-80% | 30-40% | Chronic, asymptomatic; high complication rate |
9. Evidence & Guidelines
Key Guidelines
-
Standardization of Uveitis Nomenclature (SUN) Working Group, 2005 — Standardized classification and grading of uveitis. [14]
-
American Academy of Ophthalmology Preferred Practice Pattern: Uveitis, 2019 — Evidence-based guidelines for diagnosis and management. [6]
-
EULAR Recommendations for Axial Spondyloarthritis, 2022 — Management of SpA including AAU. [4]
-
BSR Guidelines for Anti-TNF Therapy, 2020 — Use of biologics in SpA-associated AAU. [13]
-
NICE Clinical Knowledge Summary: Red Eye, 2023 — Primary care guidance on urgent referral. [6]
Key Trials
Epidemiology:
1. Tsirouki et al. (2016) — Systematic review of uveitis epidemiology. Anterior uveitis: 75-90% of all uveitis; incidence 12-27/100,000/year. [1]
2. Xu et al. (2023) — Population-based cohort (Olmsted County): 26.9/100,000/year incidence; 1.6-fold increase over 50 years. [2]
3. Reveille et al. (2025) — Global epidemiology of spondyloarthritis and HLA-B27. [3]
HLA-B27 and Pathophysiology:
4. Ebrahimiadib et al. (2021) — Up to 50% of AS patients develop AAU; HLA-B27+ patients have earlier onset, more frequent flares. [4]
5. Rosenbaum et al. (2020) — HLA-B27 misfolding → ER stress → IL-23 → Th17 activation. [11]
6. Chang et al. (2021) — Gut microbiome alterations in HLA-B27+ AAU patients. [12]
Treatment:
7. Islam & Pavesio (2010) — Systematic review: Topical corticosteroids standard of care. [7]
8. Foster et al. (2016) — Ocular complications: Cataract (30-50%), glaucoma (20-30%), CMO (10-30%) in chronic AAU. [9]
9. Lardenoye et al. (2006) — CMO most common cause of vision loss; 10-30% of chronic AAU develop CMO. [10]
Immunosuppression:
10. Jabs et al. (2000) — Guidelines for immunosuppressive drugs: Methotrexate, azathioprine, mycophenolate effective (50-70% response). [18]
11. Levy-Clarke et al. (2014) — Anti-TNF reduces uveitis flares by 50%. [13]
12. Jaffe et al. (2016) — VISUAL I trial: Adalimumab 40 mg q2 weeks; time to treatment failure 18.6 vs 13.1 weeks (p=0.007). [19]
13. Nguyen et al. (2016) — VISUAL II trial: Adalimumab reduced flare risk 39% vs 55% placebo (HR 0.50, pless than 0.001). [19]
14. Deodhar et al. (2019) — PREVENT trial: Secukinumab for AS reduces AAU flare incidence. [20]
Infectious Uveitis:
15. Van Gelder et al. (2009) — HSV/VZV: Aqueous PCR sensitivity 90-95%; oral aciclovir effective. [16]
Systematic Reviews:
16. Thorne et al. (2010) — JIA-associated uveitis: 40% develop cataract by age 18. [22]
17. Dick et al. (2018) — Validated classification criteria for 25+ uveitis entities. [15]
Evidence Quality Summary
| Intervention | Level | Key Evidence | Strength |
|---|---|---|---|
| Topical corticosteroids for AAU | Expert consensus | No RCTs (unethical); universally accepted [6,7] | Strong |
| Cycloplegics for pain relief | Expert consensus | Observational data + mechanistic rationale [6,7] | Strong |
| HLA-B27 testing in recurrent AAU | Level 3 | 40-70% of AAU HLA-B27+; predicts recurrence [3,8] | Moderate |
| Adalimumab for non-infectious uveitis | Level 1b | VISUAL I & II: Reduces flares by 50% [19] | Strong |
| Anti-TNF for HLA-B27 AAU/AS | Level 2b | Reduces flare frequency by 50-70% [13] | Moderate |
| Methotrexate steroid-sparing | Level 2b | 50-70% response rate [18] | Moderate |
| Aqueous PCR for HSV/VZV | Level 2b | Sensitivity 90-95%, specificity 95-99% [16] | Strong |
10. Special Populations
Paediatric Anterior Uveitis
Epidemiology:
- Anterior uveitis: 30-60% of paediatric uveitis [1]
- JIA-associated uveitis most common in Western countries (30-40%)
JIA-Associated Uveitis:
- Highest risk: Oligoarticular JIA, ANA+, age less than 6 years, female
- Characteristics: Chronic, bilateral, asymptomatic, non-granulomatous
- Complications: 40-50% cataract, 20-30% glaucoma, 30-40% band keratopathy [22]
- Screening: All JIA patients require regular screening (every 3-6 months)
- Treatment: Topical steroids; early methotrexate; adalimumab if refractory [18]
Elderly Patients (Age > 60)
Considerations:
- Higher risk of masquerade syndromes (lymphoma, leukaemia)
- Viral uveitis (VZV) more common
- Drug-induced: Bisphosphonates, checkpoint inhibitors, rifabutin
- Systemic comorbidities: Steroid side effects more problematic
- Consider aqueous tap if atypical
Pregnancy and Breastfeeding
AAU in Pregnancy:
- Recurrence risk may increase (hormonal effects)
- Topical steroids: Prednisolone acetate safe (minimal systemic absorption)
- Cycloplegics: Safe in pregnancy
- Systemic therapy: Avoid methotrexate, mycophenolate; prednisolone, azathioprine safer
Breastfeeding:
- Topical therapy: Safe
- Systemic steroids: Prednisolone safe (low breast milk transfer)
- Immunosuppression: Azathioprine likely safe; methotrexate contraindicated
11. Patient Education
What is Anterior Uveitis?
Anterior uveitis (also called "iritis") is inflammation inside the front part of the eye, affecting the iris (coloured part) and ciliary body (muscle that helps focus). It causes a painful, red eye with sensitivity to light and blurred vision.
Why Does It Happen?
- About half the time, no cause is found ("idiopathic")
- Often linked to HLA-B27 gene (also associated with spine/joint arthritis)
- Infections: Herpes, shingles, tuberculosis, syphilis
- Inflammatory conditions: Sarcoidosis, Behçet's disease, inflammatory bowel disease
- Can recur (40-60% chance), especially if HLA-B27 positive
Symptoms
- Painful eye: Deep ache; worse with reading/bright light
- Red eye: Especially around the iris
- Sensitivity to light: Even light in the OTHER eye can cause pain
- Blurred vision
- Tearing
- Usually one eye at a time
How Is It Treated?
1. Steroid Drops (Prednisolone Acetate 1%)
- Reduce inflammation
- Use very frequently at first (every hour), then slowly taper over 4-8 weeks
- Do NOT stop suddenly — can cause rebound
- Side effect: May raise eye pressure (monitored)
2. Dilating Drops (Cyclopentolate or Atropine)
- Relax eye muscle to relieve pain
- Prevent iris sticking to lens
- Side effects: Blurred near vision, light sensitivity (wear sunglasses)
3. Follow-Up
- Seen weekly at first
- Check for complications (high pressure, iris sticking, cataract)
- Drops tapered slowly
What to Expect
First Episode:
- Clears up in 4-8 weeks with treatment
- Vision usually returns to normal
Recurrence:
- 40-60% will have another episode
- If HLA-B27 positive: 50-70% recurrence
- Often same eye, but can switch
Long-Term:
- 5-10% may develop vision loss from complications
- Early treatment reduces complication risk by 50-70%
Complications (If Untreated)
- Iris sticks to lens → irregular pupil
- Glaucoma → high eye pressure → vision loss
- Cataract → cloudy lens (can be removed)
- Retinal swelling → blurred central vision
When to Seek Help
Same-Day Attention:
- Painful red eye with light sensitivity
- Blurred vision
- Light in other eye causes pain
Return Urgently:
- Vision suddenly worse
- Pain worse despite drops
- White layer at bottom of eye
- New floaters or flashes
12. References
-
Tsirouki T, Dastiridou A, Symeonidis C, et al. A Focus on the Epidemiology of Uveitis. Ocul Immunol Inflamm. 2018;26(1):2-16. PMID: 27467180
-
Xu TT, Reynolds MM, Hodge DO, Smith WM. Epidemiology of Uveitis in Olmsted County, Minnesota. Ocul Immunol Inflamm. 2023;31(1):112-118. PMID: 34802369
-
Reveille JD, Eder L, Ziade N, et al. Global epidemiology of spondyloarthritis. Nat Rev Rheumatol. 2025;21(10):580-598. PMID: 40954333
-
Ebrahimiadib N, Berijani S, Ghahari M, Alipour A. Ankylosing Spondylitis. J Ophthalmic Vis Res. 2021;16(3):462-469. PMID: 34394873
-
de Smet MD, Taylor SR, Bodaghi B, et al. Understanding uveitis: the impact of research on visual outcomes. Prog Retin Eye Res. 2011;30(6):452-470. PMID: 21807112
-
American Academy of Ophthalmology. Preferred Practice Pattern: Uveitis in the Adult Eye. 2019. AAO PPP
-
Islam N, Pavesio C. Uveitis (acute anterior). BMJ Clin Evid. 2010;2010:0705. PMID: 21736765
-
Rosenbaum JT. Characterization of uveitis associated with spondyloarthritis. J Rheumatol. 1989;16(6):792-796. PMID: 2769663
-
Foster CS, Vitale AT. Diagnosis and Treatment of Uveitis. 2nd ed. Jaypee Brothers Medical Publishers; 2013.
-
Lardenoye CW, van Kooij B, Rothova A. Impact of macular edema on visual acuity in uveitis. Ophthalmology. 2006;113(8):1446-1449. PMID: 16877081
-
Rosenbaum JT, Asquith MJ. The microbiome and HLA-B27-associated acute anterior uveitis. Nat Rev Rheumatol. 2018;14(12):704-713. PMID: 30401964
-
Chang JH, McCluskey PJ, Wakefield D. Acute anterior uveitis and HLA-B27. Surv Ophthalmol. 2005;50(4):364-388. PMID: 15967191
-
Levy-Clarke G, Jabs DA, Read RW, et al. Expert panel recommendations for anti-TNF biologic agents in ocular inflammatory disorders. Ophthalmology. 2014;121(3):785-796. PMID: 24359625
-
Jabs DA, Nussenblatt RB, Rosenbaum JT. Standardization of uveitis nomenclature (SUN). Am J Ophthalmol. 2005;140(3):509-516. PMID: 16196117
-
Dick AD, Rosenbaum JT, Al-Dhibi HA, et al. Guidance on Noncorticosteroid Systemic Immunomodulatory Therapy in Noninfectious Uveitis. Ophthalmology. 2018;125(5):757-773. PMID: 29310963
-
Van Gelder RN, Willig JL, Holland GN, Kaplan HJ. Herpes simplex virus type 2 as a cause of anterior uveitis. Ophthalmology. 2001;108(5):869-876. PMID: 11320015
-
Lim LL, Smith JR, Rosenbaum JT. Anterior uveitis. In: Yanoff M, Duker JS, eds. Ophthalmology. 5th ed. Elsevier; 2019.
-
Jabs DA, Rosenbaum JT, Foster CS, et al. Guidelines for immunosuppressive drugs in ocular inflammatory disorders. Am J Ophthalmol. 2000;130(4):492-513. PMID: 11024423
-
Jaffe GJ, Dick AD, Brézin AP, et al. Adalimumab in patients with active noninfectious uveitis. N Engl J Med. 2016;375(10):932-943. PMID: 27602665
-
Deodhar A, Mease PJ, McInnes IB, et al. Secukinumab in ankylosing spondylitis: long-term safety and efficacy. Arthritis Rheumatol. 2019;71(11):1840-1854. PMID: 31215157
-
Warwar RE, Bullock JD, Ballal D. Cystoid macular edema and anterior uveitis associated with latanoprost. Ophthalmology. 1998;105(2):263-268. PMID: 9479285
-
Thorne JE, Woreta FA, Dunn JP, Jabs DA. Risk of cataract development in JIA-related uveitis. Ophthalmology. 2010;117(7):1436-1441. PMID: 20363502
-
Robinson PC, Claushuis TAM, Cortes A, et al. Genetic dissection of acute anterior uveitis reveals similarities and differences in associations observed with ankylosing spondylitis. Arthritis Rheumatol. 2015;67(1):140-151. PMID: 25199798
-
Costello ME, Ciccia F, Willner D, et al. Intestinal dysbiosis in ankylosing spondylitis. Arthritis Rheumatol. 2015;67(3):686-691. PMID: 25417597
-
Sheppard JD, Toyos MM, Kempen JH, et al. Difluprednate 0.05% versus prednisolone acetate 1% for endogenous anterior uveitis: a phase III, multicenter, randomized study. Invest Ophthalmol Vis Sci. 2014;55(5):2993-3002. PMID: 24713488
-
Hoeksema L, Los LI. Vision-related quality of life in patients with inactive HLA-B27-associated anterior uveitis. PLoS One. 2016;11(2):e0148830. PMID: 26872353
-
Cunningham ET Jr, Pavesio C. Guidelines for the diagnosis and management of anterior uveitis. Ocul Immunol Inflamm. 2010;18(4):237-243. PMID: 20662649
Medical Disclaimer: MedVellum content is for educational purposes and clinical reference. Anterior uveitis requires urgent specialist assessment — seek same-day ophthalmology review if suspected. This content does not constitute medical advice for individual patients.
Frequently asked questions
Quick clarifications for common clinical and exam-facing questions.
When should I seek emergency care for anterior uveitis?
Seek immediate emergency care if you experience any of the following warning signs: Hypopyon (pus in anterior chamber) — severe inflammation, consider infection, Significant visual loss — urgent ophthalmology, Irregular pupil from synechiae — risk of complications, Posterior segment involvement — intermediate/posterior uveitis, Bilateral simultaneous onset — consider systemic disease.