Antiphospholipid Syndrome
Diagnostic Pearl : Antiphospholipid antibodies must be positive on TWO separate occasions, at least 12 weeks apart, to confirm diagnosis. Transient aPL positivity is common following infections, medications, or...
What matters first
Diagnostic Pearl : Antiphospholipid antibodies must be positive on TWO separate occasions, at least 12 weeks apart, to confirm diagnosis. Transient aPL positivity is common following infections, medications, or...
Catastrophic APS (multi-organ thrombosis)
9 Jan 2026
Generated educational material; verify before clinical use.
Visible references section
See the concept before reading it
Study the key anatomy, imaging, and decision pathways as full teaching plates.
Clinical board
A visual summary of the highest-yield teaching signals on this page.
Urgent signals
Safety-critical features pulled from the topic metadata.
- Catastrophic APS (multi-organ thrombosis)
- Stroke in young patient
- Recurrent pregnancy loss
- DVT/PE with positive aPL
Content status and exam context
This page is AI-generated educational content. It may contain errors or omissions and is not a substitute for current guidelines, local protocols, senior clinical judgement, or professional medical advice.
MedVellum does not claim an individual clinician reviewer, board certification, or professional credential for this page unless a future version names a real, verifiable reviewer.
Clinical explanation and evidence
Antiphospholipid Syndrome
1. Clinical Overview
Summary
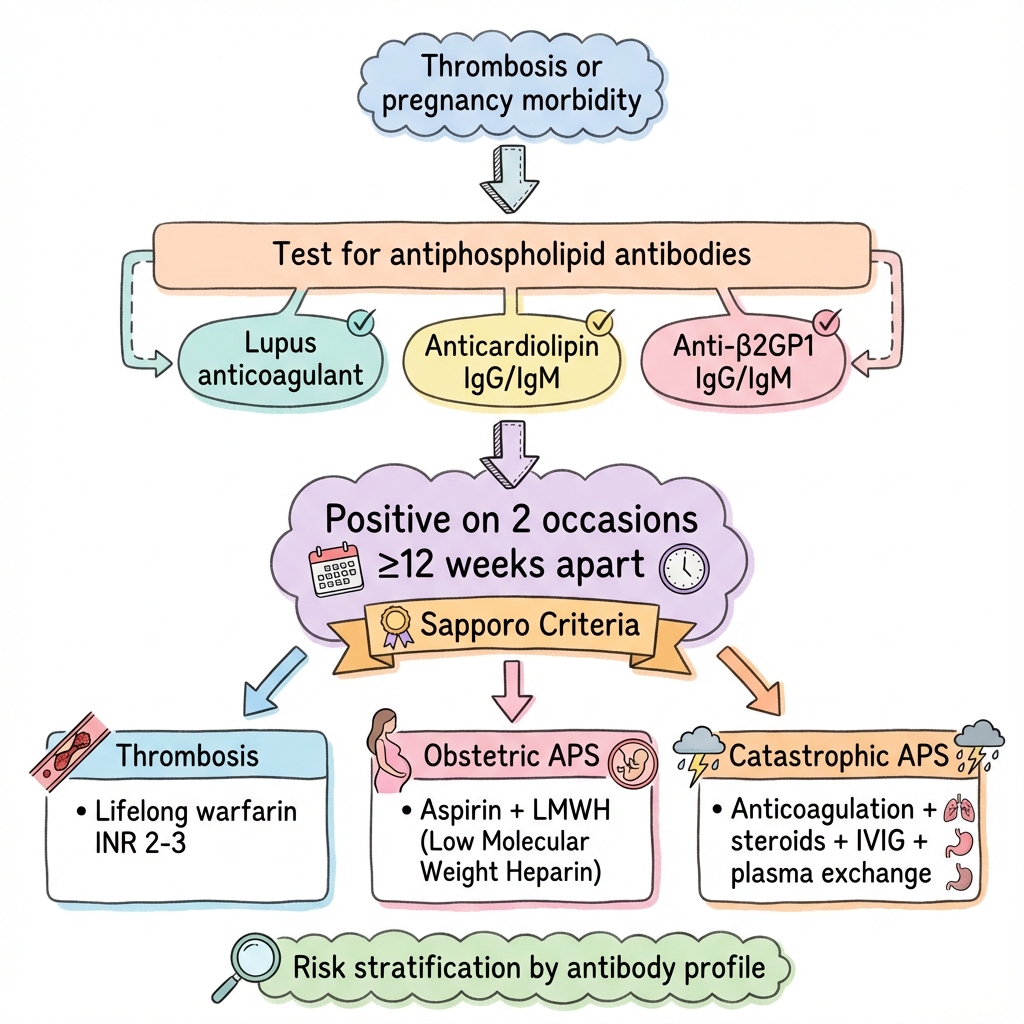
Antiphospholipid syndrome (APS) is an acquired autoimmune thrombophilia characterised by arterial and/or venous thrombosis and/or pregnancy morbidity in the presence of persistent antiphospholipid antibodies (aPL). It represents the most common acquired cause of thrombophilia and a major cause of recurrent pregnancy loss. APS can be primary (isolated) or secondary (most commonly associated with systemic lupus erythematosus). The three clinically relevant aPL are lupus anticoagulant (LA), anticardiolipin antibodies (aCL), and anti-β2-glycoprotein I antibodies (anti-β2GPI). Diagnosis requires both clinical criteria (thrombosis or pregnancy morbidity) and laboratory criteria (persistent aPL positivity on two occasions ≥12 weeks apart). Management involves lifelong anticoagulation for thrombotic APS (warfarin target INR 2-3) and aspirin plus low-molecular-weight heparin (LMWH) for obstetric APS. Direct oral anticoagulants (DOACs) are generally contraindicated, particularly in triple-positive patients, due to increased thrombotic risk. Catastrophic APS (CAPS) is a rare, life-threatening variant characterised by rapid multi-organ failure requiring urgent multimodal treatment. [1,2,3]
Key Facts
- Definition: Autoimmune thrombophilia with persistent aPL causing thrombosis and/or pregnancy morbidity
- Incidence: 2-5 per 100,000 per year in general population; aPL positivity in 30-40% of SLE patients
- Peak Demographics: Women 20-40 years (especially obstetric APS); overall female:male ratio 5:1
- Common Manifestations: Deep vein thrombosis (most common venous), stroke (most common arterial), recurrent miscarriage
- Pathognomonic Feature: Persistent aPL (≥12 weeks apart) plus thrombosis or pregnancy morbidity
- Gold Standard Diagnosis: Revised Sapporo (Sydney) classification criteria; updated 2023 ACR/EULAR criteria
- First-line Treatment: Warfarin INR 2-3 (thrombotic APS); Aspirin 75-100mg + prophylactic LMWH (obstetric APS)
- Prognosis: Good with appropriate anticoagulation; 50-70% recurrence risk without treatment; CAPS mortality 30-50%
Clinical Pearls
Diagnostic Pearl: Antiphospholipid antibodies must be positive on TWO separate occasions, at least 12 weeks apart, to confirm diagnosis. Transient aPL positivity is common following infections, medications, or malignancy and does not constitute APS.
Treatment Pearl: DOACs (rivaroxaban, apixaban) are generally NOT recommended in APS, particularly in triple-positive patients. The TRAPS and ASTRO-APS trials demonstrated significantly higher thrombosis rates with rivaroxaban compared to warfarin in high-risk APS. Warfarin remains the standard of care.
Pitfall Warning: Lupus anticoagulant causes a prolonged APTT (activated partial thromboplastin time) in laboratory testing, but paradoxically is associated with THROMBOSIS, not bleeding. The name is misleading—it reflects the in vitro effect on clotting assays, not clinical bleeding.
Risk Stratification: Triple-positive patients (LA + aCL + anti-β2GPI all positive) have the highest thrombotic risk and worst outcomes, with thrombosis rates up to 5-fold higher than single-positive patients. This subgroup requires particularly aggressive management.
Mnemonic: APS CLOT - Antibodies (aPL), Pregnancy loss, SLE association; Catastrophic variant, Livedo reticularis, Obstetric morbidity, Thrombosis
Why This Matters Clinically
APS is the most common acquired thrombophilia, affecting approximately 1 in 2,000 individuals. It accounts for 10-15% of all recurrent pregnancy losses and up to 20% of strokes in young adults (less than 50 years). Recognition of APS guides critical management decisions including lifelong anticoagulation strategies, pregnancy planning, and avoidance of high-risk medications (e.g., oestrogen-containing contraceptives). Misdiagnosis or delayed diagnosis can lead to recurrent life-threatening thrombotic events, pregnancy loss, and progression to catastrophic APS. The condition requires specialist multidisciplinary input from rheumatology, haematology, and obstetrics. [4,5]
2. Epidemiology
Incidence and Prevalence
General Population:
- Incidence of APS: 2-5 per 100,000 per year
- Prevalence of APS: Estimated 40-50 per 100,000
- Transient aPL positivity in general population: 1-5%
- Persistent aPL positivity: 0.5-1%
High-Risk Populations:
- SLE patients with aPL: 30-40%
- SLE patients who develop thrombosis: 10-15%
- Recurrent pregnancy loss patients: 10-15% have aPL
- Young stroke patients (less than 50 years): 10-20% have aPL
- Deep vein thrombosis patients: 5-10% have aPL [6,7]
Demographics
| Factor | Details |
|---|---|
| Age of Onset | Most commonly 20-40 years; can occur at any age including children |
| Sex Ratio | Female:Male 5:1 overall; obstetric APS exclusively female; thrombotic APS F:M ~2:1 |
| Ethnicity | All ethnicities affected; some studies suggest higher prevalence in African-American and Hispanic populations |
| Geography | Worldwide distribution; no clear geographic clustering |
Primary vs Secondary APS
| Type | Definition | Prevalence |
|---|---|---|
| Primary APS | APS without underlying autoimmune disease | 50-60% of APS cases |
| Secondary APS | APS associated with SLE or other autoimmune disease | 40-50% of APS cases |
| Associated Conditions | SLE (most common), Sjögren's syndrome, rheumatoid arthritis, systemic sclerosis | Variable |
Obstetric vs Thrombotic Manifestations
Obstetric-Only APS: 35-40% of APS patients (pregnancy morbidity without thrombosis) Thrombotic-Only APS: 40-45% of APS patients (thrombosis without pregnancy morbidity) Combined Obstetric + Thrombotic: 15-20% of APS patients [8,9]
3. Pathophysiology
Molecular Pathogenesis
APS is characterised by a complex interplay between autoantibodies, endothelial dysfunction, platelet activation, and complement activation. The pathogenesis is multifactorial and not fully elucidated.
Step 1: Antiphospholipid Antibody Production
The trigger for aPL production remains largely unknown. Proposed mechanisms include:
- Molecular Mimicry: Infection-related antigens (particularly viral and bacterial) may share epitopes with phospholipid-binding proteins, triggering cross-reactive antibody production
- Genetic Predisposition: HLA-DR, HLA-DQ associations; specific polymorphisms in complement and coagulation genes
- Environmental Triggers: Certain infections (CMV, EBV, parvovirus B19), medications (procainamide, hydralazine, phenytoin), and malignancies can induce transient or persistent aPL
The clinically relevant autoantibodies primarily target β2-glycoprotein I (β2GPI), a plasma protein that binds to phospholipid surfaces. Specifically, antibodies directed against domain I of β2GPI appear most pathogenic. [10,11]
Step 2: Endothelial Cell Activation
- aPL-β2GPI complexes bind to endothelial cell surface receptors (annexin A2, apolipoprotein E receptor 2)
- Activation of endothelial cells via Toll-like receptor 4 (TLR4) and other pattern recognition receptors
- Upregulation of adhesion molecules (E-selectin, VCAM-1, ICAM-1)
- Increased tissue factor expression on endothelial surface → shift to procoagulant state
- Disruption of endothelial protective mechanisms:
- Reduced production of prostacyclin (PGI2) and nitric oxide (NO)
- Decreased thrombomodulin expression
- Impaired protein C activation
- Enhanced von Willebrand factor release [12]
Step 3: Platelet Activation and Aggregation
- Direct binding of aPL-β2GPI complexes to platelet surface GPIIb/IIIa receptors
- Increased platelet activation markers (P-selectin, CD63)
- Enhanced platelet aggregation and thromboxane A2 production
- Formation of platelet-leukocyte aggregates
- Increased microparticle release from activated platelets
- Enhanced thrombin generation on platelet phospholipid surfaces [13]
Step 4: Complement Activation
- Alternative complement pathway activation is critical for thrombosis
- C5a generation recruits and activates neutrophils
- Neutrophil extracellular trap (NET) formation contributes to thrombosis
- Complement activation particularly important in pregnancy morbidity:
- C5a-mediated trophoblast injury
- Complement deposition in placenta
- Impaired placental angiogenesis
- Eculizumab (C5 inhibitor) shows promise in refractory cases [14,15]
Step 5: Coagulation Cascade Dysregulation
- Enhanced tissue factor-mediated thrombin generation
- Resistance to activated protein C (acquired)
- Reduced fibrinolysis (impaired tissue plasminogen activator release, increased PAI-1)
- Direct interference with natural anticoagulants (protein C, protein S, antithrombin)
- Prothrombin activation via anti-prothrombin antibodies
Step 6: Thrombosis Formation
The culmination of the above mechanisms leads to:
- Arterial thrombosis (stroke, MI, peripheral arterial occlusion)
- Venous thrombosis (DVT, PE, cerebral venous sinus thrombosis, Budd-Chiari)
- Microvascular thrombosis (particularly in CAPS)
- Thrombosis can occur in any vascular bed
- Recurrence is common without anticoagulation (50-70%) [16]
Pregnancy Morbidity Mechanisms
Pregnancy complications in APS are distinct from thrombotic manifestations:
Placental Mechanisms:
- Spiral artery thrombosis and placental infarction
- Impaired trophoblast invasion and differentiation
- Anti-β2GPI antibodies directly inhibit trophoblast function
- Reduced syncytiotrophoblast formation
- Defective placentation leading to insufficiency
Complement-Mediated Injury:
- Complement activation at maternal-fetal interface
- C5a-induced trophoblast injury and apoptosis
- Neutrophil recruitment and inflammation
- Impaired angiogenesis and spiral artery remodelling
Clinical Consequences:
- Early pregnancy loss (less than 10 weeks): thrombosis and inflammation at implantation site
- Late pregnancy loss (≥10 weeks): placental insufficiency
- Intrauterine growth restriction (IUGR): chronic placental insufficiency
- Pre-eclampsia: endothelial dysfunction and placental ischaemia
- Preterm delivery: placental abruption, severe pre-eclampsia [17,18]
Triple Positivity: Highest Risk Phenotype
Patients positive for all three criteria aPL (LA + aCL + anti-β2GPI) represent the highest-risk subgroup:
- 5-fold increased thrombosis risk compared to single-positive
- Higher risk of arterial events (especially stroke)
- Increased CAPS risk
- Worse pregnancy outcomes
- Higher recurrence rates despite anticoagulation
- Contraindication to DOACs (strong evidence of harm)
- May require higher intensity anticoagulation or combination therapy [19,20]
Catastrophic APS (CAPS) Pathophysiology
CAPS represents an extreme manifestation characterised by:
- Widespread microvascular thrombosis affecting ≥3 organ systems
- Triggered by infection (47%), surgery (17%), malignancy, anticoagulation withdrawal, or idiopathic
- "Cytokine storm" with massive inflammatory response
- Systemic endothelial activation and injury
- Consumptive coagulopathy may develop
- Microangiopathic haemolytic anaemia
- Multiorgan failure over days to weeks
- Mortality 30-50% despite aggressive treatment [21]
4. Clinical Presentation
Thrombotic Manifestations
APS can cause thrombosis in any vascular territory. The clinical presentation varies by location and severity.
Venous Thrombosis (Most Common)
| Site | Manifestation | Frequency in APS |
|---|---|---|
| Deep Vein Thrombosis (DVT) | Leg swelling, pain, erythema; most commonly lower limb | 30-40% of APS patients |
| Pulmonary Embolism (PE) | Dyspnoea, pleuritic chest pain, haemoptysis | 15-20%; often with DVT |
| Cerebral Venous Sinus Thrombosis | Headache, papilloedema, seizures, focal deficits | 2-5%; important in young adults |
| Hepatic Vein (Budd-Chiari) | Hepatomegaly, ascites, abdominal pain | 1-2%; may be presenting feature |
| Renal Vein Thrombosis | Flank pain, haematuria, acute kidney injury | 1-2% |
| Retinal Vein Occlusion | Sudden painless vision loss | 1-5% |
| Adrenal Vein Thrombosis | Adrenal insufficiency, haemorrhage | Rare |
Arterial Thrombosis
| Site | Manifestation | Frequency in APS |
|---|---|---|
| Ischaemic Stroke | Focal neurological deficit; most common arterial event | 15-20% of APS patients |
| Transient Ischaemic Attack (TIA) | Temporary neurological symptoms less than 24 hours | 5-10% |
| Myocardial Infarction | Chest pain, ST elevation/depression, troponin rise | 3-5%; especially young patients |
| Peripheral Arterial Thrombosis | Limb ischaemia, claudication, gangrene | 2-5% |
| Mesenteric Artery Thrombosis | Acute abdominal pain, bowel ischaemia | 1-2%; high mortality |
| Retinal Artery Occlusion | Sudden painless vision loss | 1-5% |
Microvascular Thrombosis
- Livedo Reticularis: Lacy, reticular, purplish skin discolouration on legs, trunk, arms; present in 20-25% of APS patients
- Livedoid Vasculopathy: Painful purpuric lesions, ulceration, white atrophic scars (atrophie blanche)
- Digital Ischaemia: Blue discolouration, digital infarcts, gangrene
- Skin Necrosis: Especially following warfarin initiation (warfarin-induced skin necrosis)
- Renal Thrombotic Microangiopathy: Proteinuria, hypertension, renal impairment
Obstetric Manifestations (Revised Sapporo Criteria)
Pregnancy morbidity in APS includes:
Early Pregnancy Loss:
- ≥3 consecutive unexplained spontaneous miscarriages less than 10 weeks gestation
- Morphologically normal embryo/fetus
- Excludes anatomical, genetic, or hormonal abnormalities
Late Pregnancy Loss:
- ≥1 unexplained fetal death ≥10 weeks gestation
- Morphologically normal fetus by ultrasound or direct examination
Premature Birth:
- ≥1 premature birth less than 34 weeks gestation
- Due to eclampsia, severe pre-eclampsia, or placental insufficiency
- Recognisable features of placental insufficiency
Additional Obstetric Features (not classification criteria but common):
- Recurrent second-trimester loss
- Severe early-onset pre-eclampsia (less than 34 weeks)
- Severe intrauterine growth restriction (IUGR)
- Placental abruption
- HELLP syndrome (haemolysis, elevated liver enzymes, low platelets) [22]
Non-Criteria Manifestations
APS has a wide range of associated features beyond classification criteria:
Haematological
- Thrombocytopenia: Present in 20-40%; usually mild (50,000-150,000/μL); rarely severe
- Haemolytic Anaemia: Autoimmune haemolytic anaemia (AIHA); may be part of Evans syndrome
- Bone Marrow Necrosis: Rare, presents with fever, bone pain, pancytopenia
Cardiac
- Valvular Heart Disease: Mitral and aortic valve thickening and regurgitation; "Libman-Sacks" endocarditis
- Valvular Vegetations: Sterile vegetations on valve leaflets
- Intracardiac Thrombus: Particularly in atria; may mimic myxoma
- Coronary Artery Disease: Premature atherosclerosis and MI
- Cardiomyopathy: Rare; may be ischaemic or inflammatory
Renal
- APS Nephropathy: Chronic thrombotic microangiopathy
- Acute Thrombotic Microangiopathy: Mimics haemolytic uraemic syndrome (HUS)
- Renal Artery Stenosis: Hypertension, renovascular disease
- Renal Infarction: Flank pain, haematuria
- End-Stage Renal Disease: 10-20% of APS nephropathy progress to ESRD
Neurological
- Cognitive Dysfunction: Memory impairment, difficulty concentrating; "brain fog"
- Seizures: Generalised or focal; may be presenting feature
- Chorea: Involuntary movements; more common in young women with SLE-APS
- Migraine: Often with aura; higher frequency than general population
- Transverse Myelitis: Spinal cord inflammation and infarction
- Multiple Sclerosis-Like Syndrome: White matter lesions on MRI
- Psychiatric Manifestations: Depression, psychosis, anxiety
Pulmonary (Non-Thrombotic)
- Pulmonary Hypertension: Chronic thromboembolic or primary; affects 3-4%
- Diffuse Alveolar Haemorrhage: Rare, life-threatening
- Acute Respiratory Distress Syndrome (ARDS): Particularly in CAPS
- Fibrosing Alveolitis: Rare chronic manifestation
Gastrointestinal
- Hepatic: Budd-Chiari syndrome, hepatic infarction, nodular regenerative hyperplasia
- Intestinal: Mesenteric ischaemia, bowel infarction, ischaemic colitis
- Splenic: Splenic infarction, functional asplenia
- Pancreatic: Pancreatitis, pancreatic infarction
Musculoskeletal
- Avascular Necrosis (AVN): Femoral head most common; also humeral head, knee
- Bone Infarction: Bone pain, may be multifocal
Dermatological
- Livedo Reticularis: Lacy reticular pattern; 20-25% of patients
- Cutaneous Ulceration: Particularly legs
- Subungual Splinter Haemorrhages: Small linear haemorrhages under nails
- Pseudo-Vasculitic Lesions: Purpura, ecchymoses
- Sneddon Syndrome: Livedo reticularis + stroke + hypertension [23]
Catastrophic APS (CAPS)
CAPS is a rare (less than 1% of APS), life-threatening variant:
Clinical Features:
- Acute multiorgan failure evolving over days to weeks
- Small vessel thrombosis affecting ≥3 organ systems
- Histopathological evidence of multiple small vessel occlusions
- Persistent aPL positivity
Common Organ Involvement:
- Renal (71%): Acute kidney injury, thrombotic microangiopathy, malignant hypertension
- Pulmonary (64%): ARDS, pulmonary haemorrhage, PE
- Cerebral (62%): Encephalopathy, stroke, seizures, coma
- Cardiac (51%): Myocardial infarction, heart failure, valve dysfunction
- Cutaneous (50%): Livedo reticularis, purpura, digital ischaemia, gangrene
- Hepatic: Hepatic infarction, Budd-Chiari
- Gastrointestinal: Mesenteric ischaemia, bowel infarction
Precipitating Factors:
- Infection (47% of cases): most common trigger
- Surgery (17%): particularly abdominal, orthopaedic
- Anticoagulation withdrawal or subtherapeutic levels (7%)
- Pregnancy or puerperium (6%)
- Medications (5%): particularly oral contraceptives
- Malignancy (4%)
- Idiopathic (18%): no identifiable trigger
Mortality:
- 30-50% overall
- Higher with renal involvement, CNS involvement, or underlying SLE
- Improved survival with early aggressive treatment
- Recurrence risk 15-20% [24,25]
Red Flags Requiring Urgent Assessment
[!CAUTION] Emergency Red Flags
- Stroke or TIA in patient less than 50 years (consider APS in differential)
- Recurrent venous thrombosis despite adequate anticoagulation
- Thrombosis in unusual site (mesenteric, cerebral venous sinus, Budd-Chiari)
- ≥3 consecutive early pregnancy losses or ≥1 unexplained fetal death ≥10 weeks
- Multiorgan failure with thrombocytopenia and positive aPL (CAPS)
- Livedo reticularis with systemic features (thrombosis, neurological symptoms)
- Acute renal failure with microangiopathic haemolytic anaemia and thrombocytopenia
- Young patient with MI and no traditional cardiovascular risk factors
5. Clinical Examination
General Inspection
Vital Signs:
- Blood pressure: may be elevated (renal involvement, hypertension common)
- Pulse: tachycardia (if PE or acute thrombosis)
- Respiratory rate: tachypnoea (PE, pulmonary hypertension)
- Oxygen saturation: hypoxia (if pulmonary involvement)
General Appearance:
- Well vs unwell
- Pallor (anaemia from haemolysis)
- Cyanosis (digital ischaemia, hypoxia)
- Signs of systemic illness or multiorgan failure (CAPS)
Skin and Soft Tissue
Livedo Reticularis:
- Lacy, reticulated, purple-red discolouration
- Most prominent on legs, thighs, trunk, buttocks, arms
- Non-blanching, irregular net-like pattern
- Worsens with cold exposure
- Distinguish from livedo racemosa (broken, irregular pattern; more associated with vasculitis)
Other Dermatological Signs:
- Cutaneous ulceration (especially lower legs)
- Digital infarcts or gangrene
- Subungual splinter haemorrhages
- Purpura or petechiae (if thrombocytopenic)
- Ecchymoses (bruising)
- Nail fold capillary changes (in SLE-associated APS)
- Photosensitive rash (suggests SLE)
Cardiovascular Examination
Inspection:
- Signs of DVT: leg swelling, erythema, visible venous collaterals
- Peripheral oedema (heart failure, renal failure)
Palpation:
- Calf tenderness (DVT)
- Homan's sign (unreliable, do not rely on)
- Pedal pulses (assess for peripheral arterial disease)
Auscultation:
- Cardiac murmurs: mitral regurgitation, aortic regurgitation (valvular disease)
- Loud P2, parasternal heave (pulmonary hypertension)
- Irregular pulse (atrial fibrillation)
Other:
- Jugular venous pressure (JVP): elevated if right heart failure or PE
- Blood pressure: both arms (assess for renal artery stenosis, dissection)
Respiratory Examination
Inspection:
- Respiratory distress (PE, ARDS, pulmonary haemorrhage)
- Use of accessory muscles
- Cyanosis
Percussion/Auscultation:
- Reduced breath sounds (pleural effusion from PE)
- Crackles (pulmonary oedema, haemorrhage)
- Normal in many cases of PE
Abdominal Examination
Inspection:
- Distension (ascites from Budd-Chiari, portal hypertension)
- Surgical scars (previous thrombotic complications)
Palpation:
- Hepatomegaly (Budd-Chiari, hepatic vein thrombosis, right heart failure)
- Splenomegaly (splenic infarction, SLE, portal hypertension)
- Tenderness (mesenteric ischaemia, splenic/hepatic infarction)
- Renal angle tenderness (renal vein thrombosis, infarction)
Auscultation:
- Abdominal bruits (renal artery stenosis)
Neurological Examination
Cognitive Assessment:
- Mini-Mental State Examination (MMSE)
- Cognitive impairment, memory problems ("brain fog")
- Psychiatric features (depression, psychosis)
Cranial Nerves:
- Visual field defects (stroke, retinal artery/vein occlusion)
- Fundoscopy: retinal haemorrhages, cotton wool spots, vessel occlusion
- Other cranial nerve palsies (brainstem stroke)
Motor/Sensory:
- Focal neurological deficits (stroke, TIA)
- Hemiparesis, hemisensory loss
- Reflexes: hyperreflexia, Babinski sign (upper motor neuron lesion)
Movement Disorders:
- Chorea (involuntary, irregular movements)
- Tremor
Coordination:
- Cerebellar signs (ataxia, dysdiadochokinesia, intention tremor)
Musculoskeletal and Obstetric History
Joints and Gait:
- Arthralgia common in SLE-associated APS; limited range of motion (avascular necrosis)
- Antalgic gait (AVN of hip), ataxic gait (cerebellar stroke), hemiparetic gait (stroke)
Pregnancy History (Women of Reproductive Age):
- Pattern and timing of pregnancy losses (first vs second/third trimester)
- Complications: pre-eclampsia, placental insufficiency, IUGR, preterm delivery
- Live births and infertility history
Associated Autoimmune Disease:
- SLE: Malar rash, discoid rash, photosensitive rash, oral ulcers, alopecia, serositis, arthritis
- Sjögren's, systemic sclerosis, rheumatoid arthritis features
6. Investigations
Antiphospholipid Antibody Testing
Diagnosis of APS requires laboratory demonstration of persistent aPL. Testing must be performed correctly to avoid false positives/negatives.
Three Criteria Antiphospholipid Antibodies
| Test | Method | Positive Threshold | Notes |
|---|---|---|---|
| Lupus Anticoagulant (LA) | Functional clotting assay | Positive by dilute Russell's viper venom time (dRVVT) and/or activated partial thromboplastin time (APTT)-based assays | Most specific for thrombosis; requires 2 different assays (APTT + dRVVT recommended) |
| Anticardiolipin IgG (aCL IgG) | ELISA | > 40 GPL units OR > 99th percentile (medium-high titre) | Titre-dependent risk; low titres (less than 40) not diagnostic |
| Anticardiolipin IgM (aCL IgM) | ELISA | > 40 MPL units OR > 99th percentile (medium-high titre) | Less specific than IgG; some experts question clinical significance |
| Anti-β2GPI IgG | ELISA | > 99th percentile OR > 40 units | Highly specific; domain I-specific assays under development |
| Anti-β2GPI IgM | ELISA | > 99th percentile OR > 40 units | Less specific than IgG |
Critical Testing Principles
Timing Requirements:
- MUST test on TWO separate occasions ≥12 weeks apart
- Single positive test is insufficient for diagnosis
- Allows exclusion of transient aPL (infections, medications)
- Ideally, confirmatory testing 12-16 weeks after initial positive
Pre-Analytical Considerations:
- Avoid testing during acute thrombosis or infection (may cause transient positivity)
- Ideally test ≥2 weeks after acute event
- Anticoagulation effects:
- "Warfarin: interferes with LA testing (APTT, dRVVT prolonged); may test on DOACs or before starting warfarin if possible"
- "Heparin: interferes with LA testing; test before starting or use anti-Xa-resistant assays"
- "DOACs: interfere with LA testing; avoid or use DOAC-specific calibration"
- For aCL and anti-β2GPI (ELISA), anticoagulation does not interfere
Triple Positivity:
- Positivity for all three: LA + aCL (IgG or IgM) + anti-β2GPI (IgG or IgM)
- Highest thrombotic risk (5-fold increase vs single positive)
- Contraindication to DOACs
- May require intensified anticoagulation [26,27]
Classification Criteria
Revised Sapporo (Sydney) Criteria (2006)
Definite APS requires ≥1 clinical criterion AND ≥1 laboratory criterion:
Clinical Criteria:
-
Vascular Thrombosis
- One or more clinical episodes of arterial, venous, or small vessel thrombosis
- In any tissue or organ
- Confirmed by objective validated criteria (imaging, Doppler, histopathology)
- Superficial vein thrombosis NOT included
- Histopathology: thrombosis without significant vessel wall inflammation
-
Pregnancy Morbidity
- One or more unexplained deaths of morphologically normal fetus ≥10 weeks gestation, OR
- One or more premature births of morphologically normal neonate less than 34 weeks due to eclampsia, severe pre-eclampsia, or placental insufficiency, OR
- Three or more unexplained consecutive spontaneous abortions less than 10 weeks gestation (excluding anatomical, genetic, hormonal abnormalities)
Laboratory Criteria (present on ≥2 occasions, ≥12 weeks apart):
- Lupus anticoagulant (LA) present
- Anticardiolipin IgG or IgM antibody (medium-high titre: > 40 GPL/MPL or > 99th percentile)
- Anti-β2-glycoprotein I IgG or IgM antibody (> 99th percentile)
2023 ACR/EULAR Classification Criteria
Recently updated criteria with weighted point system (published August 2023):
Entry Criterion:
- At least one positive aPL test (any of the three criteria aPL)
Clinical Domains (points):
- Macrovascular venous thromboembolism (5 points)
- Macrovascular arterial thrombosis (5 points)
- Microvascular thrombosis (3 points)
- Pregnancy loss ≥10 weeks (4 points)
- Pregnancy loss less than 10 weeks with morphologically normal embryo (2 points)
- Delivery less than 34 weeks due to placental insufficiency or pre-eclampsia (4 points)
Laboratory Domains (points):
- LA positive on 2 occasions ≥12 weeks apart (5 points)
- aCL IgG/IgM > 99th percentile on 2 occasions (4 points if high, 2 points if medium titre)
- Anti-β2GPI IgG/IgM > 99th percentile on 2 occasions (4 points if high, 2 points if medium titre)
Classification: APS if total score ≥3 points (with ≥1 clinical + ≥1 laboratory criterion)
The new criteria aim to improve specificity and better risk-stratify patients. [28,29]
Additional Laboratory Tests
Full Blood Count (FBC)
- Thrombocytopenia: Present in 20-40%; usually mild (50,000-150,000/μL)
- Anaemia: Normocytic (chronic disease) or haemolytic (microangiopathic haemolytic anaemia in CAPS; direct Coombs-positive AIHA)
- Leukopenia: If associated SLE
- Blood film: Schistocytes (if microangiopathic haemolysis), spherocytes (if AIHA)
Coagulation Studies
- APTT: Prolonged if LA present (paradoxical finding given thrombotic tendency)
- Prothrombin Time (PT): Usually normal; prolonged if on warfarin
- Mixing Study: If prolonged APTT, mixing with normal plasma will NOT correct (distinguishes inhibitor from factor deficiency)
- D-dimer: Elevated if acute thrombosis
- Fibrinogen: Normal or elevated; reduced in CAPS with DIC
Autoimmune Screen (Exclude Secondary APS)
- Antinuclear Antibody (ANA): Positive in 30-40% of primary APS; > 95% in SLE-associated APS
- Anti-dsDNA: Specific for SLE (not typically positive in primary APS)
- Extractable Nuclear Antigens (ENA): Anti-Ro, anti-La (Sjögren's), anti-Sm (SLE-specific), anti-RNP
- Complement (C3, C4): Low in SLE-associated APS (active lupus); normal in primary APS
- Rheumatoid Factor (RF): If suspect rheumatoid arthritis
- Anti-CCP: If suspect rheumatoid arthritis
Renal Function
- Serum Creatinine, eGFR: Assess for APS nephropathy or acute kidney injury (CAPS)
- Urinalysis: Proteinuria, haematuria (renal involvement, thrombotic microangiopathy)
- Urine Protein:Creatinine Ratio (PCR): Quantify proteinuria
Liver Function Tests
- AST, ALT: Elevated if hepatic infarction, Budd-Chiari
- ALP, GGT: Cholestatic pattern in Budd-Chiari
- Bilirubin: Elevated if haemolysis or hepatic dysfunction
Cardiac Biomarkers
- Troponin: If suspect MI
- BNP/NT-proBNP: If heart failure, pulmonary hypertension
Thrombophilia Screen (Selected Cases)
May identify coexistent inherited thrombophilia (additive risk): Protein C/S, antithrombin, Factor V Leiden, prothrombin G20210A, homocysteine. Test when not on anticoagulation; warfarin reduces protein C/S levels.
Imaging Investigations
Venous Doppler: DVT assessment; non-compressible vein, intraluminal thrombus.
CTPA: PE diagnosis; intraluminal filling defect in pulmonary artery.
Echocardiography (TTE/TOE): Valvular disease (thickening, regurgitation, vegetations), intracardiac thrombus, pulmonary hypertension. TOE more sensitive for valve abnormalities.
MRI Brain: Stroke/TIA assessment; acute/chronic infarcts, white matter hyperintensities. MRI Spine: Transverse myelitis (T2 hyperintensity). MRI Pelvis/Hip: Avascular necrosis (bone marrow oedema, femoral head collapse).
CT Brain: Acute stroke (hypodense ischaemia, hyperdense haemorrhage). CT Abdomen: Mesenteric ischaemia, Budd-Chiari, renal infarction.
V/Q Scan: Chronic thromboembolic pulmonary hypertension; mismatched perfusion defects.
Renal Biopsy: APS nephropathy; thrombotic microangiopathy, vascular changes. Risk of bleeding.
Histopathology: Thrombosis with minimal vessel inflammation (vs vasculitis); placental infarction, spiral artery thrombosis in obstetric APS.
7. Management
General Principles
Goals of Treatment:
- Prevent recurrent thrombosis (secondary prevention)
- Optimize pregnancy outcomes (obstetric APS)
- Manage acute thrombotic events
- Prevent progression to catastrophic APS
- Treat underlying autoimmune disease if secondary APS
Multidisciplinary Approach:
- Rheumatology (diagnosis, overall management, SLE management)
- Haematology (anticoagulation management, complex cases)
- Obstetrics (high-risk pregnancy care)
- Cardiology (valvular disease, pulmonary hypertension)
- Neurology (stroke, cognitive dysfunction)
- Nephrology (APS nephropathy)
Management Overview
Thrombotic APS: Warfarin INR 2-3 (lifelong) for venous events; warfarin INR 2-3 +/- aspirin 75mg for arterial events. Avoid DOACs.
Obstetric-Only APS: Aspirin 75-100mg + prophylactic LMWH during pregnancy; 70-80% live birth rate.
Prior Thrombosis + Pregnancy: Therapeutic LMWH throughout pregnancy; switch from warfarin before conception.
Thrombotic APS Management
First Venous Thrombotic Event
Standard Treatment:
- Warfarin: Target INR 2-3
- Duration: Lifelong (indefinite)
- Bridging: Start LMWH or unfractionated heparin immediately; overlap with warfarin for ≥5 days and until INR therapeutic (≥2.0) for 24 hours
Evidence:
- Multiple studies show high recurrence risk (50-70%) if anticoagulation stopped
- No clear benefit of higher-intensity anticoagulation (INR 3-4) for venous events in most patients
- DOACs are generally NOT recommended (see below) [30]
Alternative in Selected Cases:
- Long-term LMWH: If warfarin contraindicated, intolerant, or unreliable INR control
- "Dose: Therapeutic dose (e.g., enoxaparin 1.5 mg/kg once daily or 1 mg/kg twice daily)"
- "Monitoring: Anti-Xa levels (target 0.5-1.0 for twice daily, 1.0-2.0 for once daily)"
First Arterial Thrombotic Event
Standard Treatment:
- Warfarin: Target INR 2-3
- Aspirin: Consider adding low-dose aspirin 75-100 mg daily (some evidence for additional benefit in arterial APS)
- Duration: Lifelong
High-Intensity Warfarin (Controversial):
- Some experts recommend higher INR target (3-4) for arterial events or recurrent thrombosis
- Limited evidence; consider in:
- Recurrent arterial events despite INR 2-3
- Triple-positive patients with arterial thrombosis
- Increased bleeding risk must be weighed
Stroke-Specific Considerations:
- Acute ischaemic stroke: thrombolysis (tPA) if within 4.5 hours and eligible
- Antiplatelet vs anticoagulation debate: warfarin superior to aspirin for secondary stroke prevention in APS
- Address cardiovascular risk factors: hypertension, diabetes, dyslipidaemia, smoking
Recurrent Thrombosis Despite Anticoagulation
Assessment:
- Confirm compliance with anticoagulation
- Check INR control (time in therapeutic range)
- Exclude alternative causes (malignancy, other thrombophilia)
- Consider drug interactions affecting warfarin levels
Management Options:
- Increase INR target to 3-4 (high-intensity warfarin)
- Add aspirin 75-100 mg daily to warfarin
- Switch to therapeutic LMWH (particularly if poor INR control)
- Consider hydroxychloroquine 200-400 mg daily (immunomodulatory, antithrombotic effects)
- Rarely: add second antiplatelet (clopidogrel) or immunosuppression (rituximab) in refractory cases [31]
Direct Oral Anticoagulants (DOACs): Generally Contraindicated
Evidence Against DOACs in APS:
TRAPS Trial (2018):
- Randomized trial: rivaroxaban vs warfarin in triple-positive APS
- STOPPED EARLY due to excess thrombotic events in rivaroxaban arm
- 4-fold increase in arterial thrombotic events with rivaroxaban
- Conclusion: Rivaroxaban is INFERIOR and potentially harmful in triple-positive APS [32]
ASTRO-APS Trial (2019):
- Randomized trial: apixaban vs warfarin in APS (mixed antibody profiles)
- Trend toward more thrombotic events with apixaban (not statistically significant)
- Study underpowered
Current Recommendations:
- Avoid DOACs in APS, particularly:
- Triple-positive patients (absolute contraindication)
- Arterial thrombosis history
- High-risk aPL profile
- Possible exceptions (expert opinion, limited data):
- Single-positive aPL (not meeting criteria for APS)
- Isolated aCL or anti-β2GPI positivity (not LA-positive)
- Venous thrombosis only, low-risk profile
- Patient unable to take warfarin and LMWH not feasible
- If DOAC used, must be shared decision with patient understanding risks; close monitoring required
Why DOACs Fail in APS:
- Mechanisms unclear
- Hypotheses: inadequate inhibition of Factor Xa in hypercoagulable APS state; different pharmacokinetics; failure to inhibit contact pathway activation [33]
Obstetric APS Management
Pregnancy in APS requires specialized high-risk obstetric care with multidisciplinary input.
Obstetric-Only APS (No Prior Thrombosis)
Recurrent Early Pregnancy Loss (less than 10 weeks):
- Low-dose aspirin 75-100 mg daily: start preconception, continue until delivery
- Prophylactic LMWH: start once pregnancy confirmed (positive β-hCG or gestational sac on ultrasound)
- Enoxaparin 40 mg subcutaneous once daily, OR
- Dalteparin 5,000 units subcutaneous once daily
- Continue until: 6 weeks postpartum (highest thrombotic risk period)
Late Pregnancy Loss (≥10 weeks) or Severe Pre-eclampsia/Placental Insufficiency:
- Low-dose aspirin 75-100 mg daily: start preconception
- Prophylactic or intermediate-dose LMWH: start once pregnancy confirmed
- Some experts use intermediate dose for late loss/severe complications
- Enhanced fetal monitoring: Serial growth scans, umbilical artery Doppler from 24 weeks
- Timing of delivery: Individualized; consider delivery at 37-38 weeks if uncomplicated
Success Rates:
- Live birth rate with aspirin + LMWH: 70-80%
- Without treatment: 30-50% live birth rate
Adjunctive Therapies (Refractory Obstetric APS):
- Hydroxychloroquine 200-400 mg daily (safe in pregnancy; may improve outcomes)
- Low-dose prednisolone 10-20 mg daily (if recurrent loss despite aspirin+LMWH; risk of gestational diabetes, hypertension)
- Intravenous immunoglobulin (IVIG) 0.4 g/kg monthly (limited evidence; expensive; consider if refractory)
- Therapeutic plasma exchange: Rarely used; case reports only [34,35]
Prior Thrombosis + Pregnancy
- Therapeutic LMWH throughout pregnancy
- Enoxaparin 1 mg/kg subcutaneous twice daily (or 1.5 mg/kg once daily)
- Monitor anti-Xa levels (target 0.6-1.0 for twice daily dosing, 4 hours post-dose)
- Low-dose aspirin 75-100 mg daily
- Switch from warfarin: BEFORE conception (warfarin is teratogenic in weeks 6-12)
- Ideally plan pregnancy; stop warfarin and start LMWH before attempting conception
- If unplanned pregnancy on warfarin, switch to LMWH as soon as pregnancy confirmed
- Postpartum anticoagulation: Resume warfarin 5-7 days postpartum; continue LMWH until therapeutic INR
- Duration postpartum: ≥6 weeks; then lifelong warfarin as per thrombotic APS protocol
Labour, Delivery, and Preconception
Labour/Delivery: Stop LMWH 12-24 hours before neuraxial anaesthesia (prophylactic vs therapeutic dose). Restart 4-12 hours postpartum. Warfarin and LMWH safe in breastfeeding.
Preconception: Stop warfarin, start LMWH before conception. Optimize SLE activity. Folic acid 5mg daily. Address cardiovascular risk factors. Multidisciplinary team (obstetrics, rheumatology, haematology).
Primary Thromboprophylaxis (Asymptomatic aPL Carriers)
Low-Risk (Single-Positive, Low Titre): Cardiovascular risk modification, avoid oestrogen contraceptives/HRT. Thromboprophylaxis during high-risk periods (surgery, hospitalization, long flights). Consider low-dose aspirin in select cases.
High-Risk (Triple-Positive, High Titre, SLE-Associated): Low-dose aspirin 75-100mg daily (stronger evidence in SLE-aPL). Hydroxychloroquine 200-400mg daily if SLE. Aggressive thromboprophylaxis during high-risk periods. [36]
Catastrophic APS (CAPS) Management
CAPS is a medical emergency requiring ICU-level care.
First-Line Treatment (Triple Therapy):
-
Anticoagulation:
- Unfractionated heparin IV (target APTT 1.5-2.5× control) OR
- Therapeutic LMWH subcutaneous
- Continue lifelong anticoagulation (warfarin) after acute phase
-
High-Dose Corticosteroids:
- Methylprednisolone 500-1,000 mg IV daily for 3 days, THEN
- Prednisolone 1 mg/kg/day orally (taper slowly)
-
Plasma Exchange (PLEX) OR Intravenous Immunoglobulin (IVIG):
- Plasma exchange: 5-7 sessions over 10-14 days; removes aPL and cytokines
- IVIG: 0.4 g/kg/day for 5 days OR 1 g/kg for 2 days
- PLEX and IVIG appear equally effective; choice depends on local availability
Supportive Care:
- Treat precipitating factors (antibiotics for infection, surgery if indicated)
- Organ support:
- "Renal: Dialysis if acute kidney injury/renal failure"
- "Respiratory: Mechanical ventilation if ARDS"
- "Cardiovascular: Inotropic support if shock"
- Avoid triggers: Maintain therapeutic anticoagulation, avoid nephrotoxic drugs
Second-Line/Refractory CAPS:
- Rituximab 375 mg/m² IV weekly for 4 weeks (B-cell depletion)
- Eculizumab (C5 complement inhibitor): Loading dose 900 mg IV, then 1,200 mg at 1 week, then 1,200 mg every 2 weeks
- Requires meningococcal vaccination (give 2 weeks before if possible, or give antibiotics prophylaxis)
- Promising results in case series; very expensive
- Cyclophosphamide IV (in SLE-associated CAPS)
Outcomes:
- Mortality 30-50% despite treatment
- Survivors require lifelong anticoagulation (high recurrence risk)
- Multiorgan damage may persist (chronic kidney disease, neurological sequelae) [37,38]
Non-Criteria Manifestations
Thrombocytopenia: Mild (50,000-150,000) requires no treatment. Severe (less than 50,000): corticosteroids, IVIG, rituximab if refractory. Balance with thrombotic risk.
Valvular Disease: Serial echo annually. Surgery if severe regurgitation. Endocarditis prophylaxis not routine.
Cognitive Dysfunction: Optimize anticoagulation, hydroxychloroquine, cognitive rehabilitation.
APS Nephropathy: Anticoagulation, BP control (less than 130/80 mmHg), ACE inhibitor/ARB for proteinuria. Monitor renal function. ESRD may require dialysis/transplant.
Monitoring and Emerging Therapies
Follow-Up (All APS Patients): INR monthly (warfarin; time in therapeutic range ≥70%), renal function 6-12 monthly, blood pressure 3-6 monthly, cardiovascular risk annual, FBC 6-12 monthly. SLE-APS: Monitor ANA, dsDNA, complement 3-6 monthly. Pregnancy: Antenatal visits 2-4 weekly, growth scans from 24 weeks, umbilical Doppler, BP monitoring, anti-Xa if therapeutic LMWH. Post-Thrombotic: Annual screening for pulmonary hypertension, CTEPH, post-thrombotic syndrome.
Emerging Therapies: Hydroxychloroquine (200-400mg daily; reduces thrombotic risk in SLE, safe in pregnancy). Statins (cardiovascular risk reduction). Rituximab (refractory APS, CAPS; variable efficacy). Eculizumab (C5 inhibitor; CAPS, refractory pregnancy APS; expensive, requires meningococcal vaccination). Novel β2GPI-targeted anticoagulants (experimental). [39]
8. Complications
Thrombotic Complications
| Complication | Incidence | Management | Prognosis |
|---|---|---|---|
| Recurrent Venous Thrombosis | 10-30% over 10 years (on treatment) | Reassess anticoagulation adequacy; consider INR 3-4, add aspirin, switch to LMWH | High recurrence if suboptimal anticoagulation |
| Recurrent Arterial Thrombosis | 15-20% over 10 years | Higher-intensity anticoagulation (INR 3-4), add aspirin, aggressive CV risk modification | Higher morbidity/mortality than venous events |
| Stroke | 15-20% of APS patients; most common arterial event | Secondary prevention with warfarin +/- aspirin; rehabilitation | Residual disability common; recurrence risk high |
| Myocardial Infarction | 3-5% of APS; more common in young patients | Standard ACS treatment + lifelong anticoagulation | Premature coronary disease; recurrent MI risk |
| Pulmonary Embolism | 15-20%; often recurrent | Lifelong anticoagulation | Post-PE syndrome; CTEPH in 3-4% |
| Chronic Thromboembolic Pulmonary Hypertension (CTEPH) | 3-4% of APS with PE | Pulmonary endarterectomy if operable; riociguat if inoperable; lifelong anticoagulation | Progressive dyspnoea; right heart failure; mortality 10-15% |
Pregnancy Complications
| Complication | Incidence (Untreated) | Incidence (Treated) | Management |
|---|---|---|---|
| Recurrent Pregnancy Loss | 50-80% | 20-30% | Aspirin + LMWH; 70-80% live birth rate with treatment |
| Pre-eclampsia | 30-50% | 10-20% | Low-dose aspirin (reduces risk 50%); early delivery if severe |
| Intrauterine Growth Restriction (IUGR) | 30-40% | 15-20% | Fetal monitoring, umbilical artery Doppler; delivery at 37-38 weeks |
| Preterm Delivery (less than 37 weeks) | 40-50% | 20-30% | Corticosteroids for fetal lung maturity if less than 34 weeks |
| Placental Abruption | 10-20% | 5-10% | Emergency Caesarean section if fetal compromise |
| Maternal Thrombosis | 5-12% during pregnancy/postpartum | 1-2% | Therapeutic LMWH if thrombosis occurs |
Organ-Specific Complications
Renal:
- APS Nephropathy: 10-20% of APS patients; chronic thrombotic microangiopathy
- Progressive proteinuria, hypertension, declining eGFR
- 10-20% progress to end-stage renal disease (ESRD) requiring dialysis/transplantation
- Acute Kidney Injury: In CAPS, acute thrombotic microangiopathy
- Renal Transplant: Higher thrombotic risk; requires lifelong anticoagulation
Cardiac:
- Valvular Disease: Mitral regurgitation, aortic regurgitation; 10-30% of APS patients
- Progressive; may require surgical repair/replacement
- Ischaemic Cardiomyopathy: Following MI or microvascular thrombosis
- Intracardiac Thrombus: High embolic risk; requires therapeutic anticoagulation
Neurological:
- Cognitive Impairment: "Brain fog," memory problems; affects 20-40%
- Progressive in some; impacts quality of life
- Vascular Dementia: Following multiple strokes
- Seizures: 5-15% of APS; may require long-term anticonvulsants
- Permanent Neurological Deficit: Following stroke; residual hemiparesis, dysphasia
Pulmonary:
- Pulmonary Hypertension: 3-4% of APS; may be CTEPH or primary
- Progressive; right heart failure; mortality 10-15% at 5 years
- Diffuse Alveolar Haemorrhage: Rare; high mortality (50%)
Haematological:
- Severe Thrombocytopenia (less than 20,000): Rare; bleeding risk vs thrombotic risk
- Evans Syndrome: Autoimmune haemolytic anaemia + thrombocytopenia
Musculoskeletal:
- Avascular Necrosis (AVN): Femoral head most common; may require hip replacement
- Risk increased by corticosteroid use
Catastrophic APS Outcomes
- Mortality: 30-50% despite aggressive treatment
- Recurrence: 15-20% of CAPS survivors have recurrent CAPS
- Chronic Organ Damage:
- Chronic kidney disease / ESRD (common)
- Neurological sequelae (cognitive impairment, stroke deficits)
- Cardiac dysfunction (cardiomyopathy, valve disease)
- Limb amputation (if severe digital ischaemia/gangrene)
Medication-Related Complications
Warfarin:
- Major bleeding: 1-3% per year
- Intracranial haemorrhage: 0.2-0.5% per year (higher if INR supratherapeutic)
- Warfarin-induced skin necrosis: Rare; paradoxical thrombosis in skin microvasculature (protein C deficiency relative to vitamin K-dependent procoagulants)
LMWH:
- Bleeding: Less than warfarin
- Heparin-induced thrombocytopenia (HIT): Rare with LMWH (less than 1%)
- Osteoporosis: With long-term use (pregnancy); calcium/vitamin D supplementation
Corticosteroids:
- Infection, osteoporosis, avascular necrosis, diabetes, hypertension, weight gain, cataracts
9. Prognosis
Overall Outcomes
With Appropriate Anticoagulation:
- Recurrent thrombosis rate: 2-5% per year (venous events)
- Recurrent thrombosis rate: 5-10% per year (arterial events; higher risk)
- Overall survival: Generally good; 10-year survival > 90% in non-CAPS patients
Without Anticoagulation (or Subtherapeutic):
- Recurrent thrombosis rate: 50-70% over 5-10 years
- High morbidity and mortality
Obstetric APS:
- Live birth rate with aspirin + LMWH: 70-80%
- Live birth rate without treatment: 30-50%
- Recurrent pregnancy loss remains significant problem in ~20-30% despite treatment
Catastrophic APS:
- Mortality: 30-50% despite aggressive treatment
- Recurrence in survivors: 15-20%
- Long-term organ damage common [40]
Prognostic Factors
Poor Prognostic Factors (Higher Thrombotic Risk):
| Factor | Risk Increase |
|---|---|
| Triple-Positive aPL (LA + aCL + anti-β2GPI) | 5-fold increase in thrombosis vs single-positive |
| High-Titre aPL | Titre > 99th percentile vs 95th percentile |
| Lupus Anticoagulant Positivity | Higher risk than isolated aCL or anti-β2GPI |
| Prior Arterial Thrombosis | Arterial recurrence rate higher than venous |
| SLE-Associated APS | Higher CAPS risk; more severe manifestations |
| Cardiovascular Risk Factors | Hypertension, diabetes, smoking, dyslipidaemia additively increase risk |
| Male Sex | Men have higher thrombotic recurrence (some studies) |
| Multiple Non-Criteria Manifestations | Livedo, thrombocytopenia, valve disease indicate more severe disease |
Good Prognostic Factors:
- Single-positive aPL (low-medium titre)
- Obstetric-only APS (no thrombosis)
- Good anticoagulation control (time in therapeutic range > 70%)
- No underlying SLE
- Female sex (for thrombotic risk; obstetric manifestations female-only)
Risk Stratification
High-Risk APS:
- Triple-positive
- Prior arterial thrombosis (especially stroke, MI)
- Recurrent thrombosis despite anticoagulation
- Catastrophic APS
- SLE-associated APS
Moderate-Risk APS:
- Double-positive aPL
- Single venous thrombotic event
- High-titre single-positive aPL
Low-Risk APS:
- Single-positive aPL (low-medium titre)
- Obstetric-only APS without thrombosis
Asymptomatic aPL Carriers:
- Persistent aPL without clinical events
- Annual thrombosis risk 0.5-2%
- Higher if triple-positive, high-titre, or SLE-associated
Long-Term Morbidity
Cumulative Damage:
- Post-thrombotic syndrome (chronic leg swelling, ulceration) after DVT: 20-50%
- Chronic thromboembolic pulmonary hypertension (CTEPH): 3-4% of PE patients
- Stroke-related disability: 30-50% have residual deficits
- Cognitive impairment: 20-40% have progressive "brain fog"
- Chronic kidney disease: 10-20% develop APS nephropathy; 2-5% reach ESRD
- Quality of life: Reduced in many patients due to chronic anticoagulation, recurrent events, organ damage
Mortality
Causes of Death in APS:
- Catastrophic APS (30-50% mortality)
- Stroke (cerebral infarction or haemorrhage)
- Myocardial infarction
- Pulmonary embolism
- Infection (immunosuppression-related in SLE-APS)
- Bleeding complications (anticoagulation-related)
Overall Mortality:
- Primary APS: 10-year survival > 90%
- SLE-associated APS: 10-year survival 80-90% (SLE contributes to mortality)
- CAPS: 30-50% acute mortality; survivors have 10-year survival ~60-70%
Life Expectancy:
- With appropriate management, most APS patients have near-normal life expectancy
- Arterial events and CAPS significantly reduce life expectancy
- Adherence to anticoagulation critical [41]
10. Evidence and Guidelines
Key Guidelines
-
EULAR Recommendations for Management of Antiphospholipid Syndrome (2019)
- Tektonidou MG et al. Ann Rheum Dis. 2019;78(10):1296-1304. PMID: 31092409
- Comprehensive evidence-based recommendations for thrombotic and obstetric APS management
- Emphasizes warfarin over DOACs; aspirin + LMWH for obstetric APS
- Addresses primary thromboprophylaxis, cardiovascular risk, and non-criteria manifestations [1]
-
ACR/EULAR Antiphospholipid Syndrome Classification Criteria (2023)
- Barbhaiya M et al. Arthritis Rheumatol. 2023;75(10):1687-1702. PMID: 37635643
- Updated classification criteria with weighted scoring system
- Improved specificity; includes microvascular manifestations
- Distinguishes clinical and laboratory domain scoring [28]
-
International Consensus Statement on Classification Criteria for APS (Sydney Criteria, 2006)
- Miyakis S et al. J Thromb Haemost. 2006;4(2):295-306. PMID: 16420554
- Revised Sapporo criteria; most widely used for clinical diagnosis
- Defines clinical and laboratory criteria; requires persistence of aPL (≥12 weeks) [3]
-
16th International Congress on Antiphospholipid Antibodies Task Force Report (2020)
- Cohen H et al. Lupus. 2021;30(10):1685-1697. PMID: 33100166
- Comprehensive update on all aspects of APS
- Addresses controversies: DOACs, seronegative APS, emerging therapies [42]
-
British Society for Haematology Guidelines on Investigation and Management of APS (2012, Update in Progress)
- Covers laboratory testing, anticoagulation management, pregnancy
- Practical UK-focused guidance
Landmark Trials and Studies
Anticoagulation Trials
TRAPS Trial (2018) — Rivaroxaban vs Warfarin in Triple-Positive APS
- Pengo V et al. Blood. 2018;132(13):1365-1371. PMID: 30002145
- Randomized, open-label, non-inferiority trial
- Stopped early due to significantly higher thrombotic events in rivaroxaban arm
- 4 arterial thromboses in rivaroxaban group vs 0 in warfarin group
- Conclusion: Rivaroxaban is INFERIOR to warfarin in triple-positive APS; avoid DOACs in this population [32]
ASTRO-APS Trial (2019) — Apixaban vs Warfarin in APS
- Woller SC et al. Arterioscler Thromb Vasc Biol. 2022;42(1):e1-e7. PMID: 34662890
- Randomized trial; mixed aPL profiles (not restricted to triple-positive)
- Trend toward more thrombotic events with apixaban (not statistically significant)
- Underpowered study; reinforces caution with DOACs in APS
RAPS Trial (2019) — Rivaroxaban vs Warfarin in APS (Mixed Profile)
- Ordi-Ros J et al. Ann Intern Med. 2019;171(10):685-694. PMID: 31610549
- Non-inferiority trial in non-triple-positive APS
- Did NOT demonstrate non-inferiority of rivaroxaban
- Some arterial events in rivaroxaban arm
- Conclusion: Insufficient evidence to support DOACs even in non-triple-positive APS
Warfarin Intensity Trials
- High-intensity warfarin (INR 3-4) vs standard (INR 2-3)
- No clear benefit of high-intensity for venous events; increased bleeding
- May consider for recurrent arterial events (limited evidence)
Obstetric APS Trials
FRUIT-RCT — Aspirin + Heparin for Recurrent Miscarriage with Inherited Thrombophilia or aPL
- de Jong PG et al. Lancet. 2014;384(9955):1673-1683.
- Aspirin + LMWH did NOT improve live birth in inherited thrombophilia
- Benefit confirmed in aPL-positive women (subgroup analysis)
PREGNANTS Study — Hydroxychloroquine in Obstetric APS
- Ongoing trial; hydroxychloroquine adjunct to aspirin + LMWH
- Preliminary data suggest possible benefit
Catastrophic APS
CAPS Registry — International Registry (> 500 Patients)
- Rodriguez-Pinto I et al. Autoimmun Rev. 2024;23(9):103601. PMID: 39174151
- Largest dataset on CAPS epidemiology, triggers, treatment, outcomes
- Mortality 30-50%; infection most common trigger (47%)
- Triple therapy (anticoagulation + steroids + PLEX/IVIG) associated with better outcomes [24]
Key Evidence
Pathophysiology:
- β2-Glycoprotein I is major antigenic target; domain I-specific antibodies most pathogenic
- Complement activation critical in pregnancy morbidity (animal models)
- Endothelial activation and NET formation contribute to thrombosis
Triple Positivity:
- Pengo V et al. Blood. 2011;117(20):5298-5303. PMID: 21378274
- Triple-positive patients have 5-fold higher thrombotic risk than single-positive
- Strongest predictor of thrombotic events
Primary Thromboprophylaxis:
- Aspirin may reduce first thrombosis in SLE patients with aPL
- No clear benefit in asymptomatic aPL carriers without SLE (ongoing debate)
Hydroxychloroquine:
- Reduces thrombotic risk in SLE-APS patients
- Possible benefit in primary APS (observational data)
- Safe in pregnancy; increasingly recommended [43]
11. Patient Explanation
What is Antiphospholipid Syndrome?
Antiphospholipid syndrome, or APS, is a condition where your immune system makes antibodies that increase the risk of blood clots forming in your arteries and veins. These antibodies are called "antiphospholipid antibodies." The condition can also cause problems during pregnancy, including miscarriages.
APS is sometimes called "sticky blood syndrome" because the blood has a greater tendency to clot than normal.
What Causes APS?
We don't fully understand what triggers APS. It's an autoimmune condition, meaning your immune system mistakenly attacks your own body. In APS, the antibodies target proteins that help regulate blood clotting. This can happen on its own (primary APS) or alongside other autoimmune diseases like lupus (secondary APS).
What Are the Symptoms?
APS itself doesn't cause symptoms day-to-day. Symptoms appear when blood clots form or during pregnancy complications:
Blood Clots:
- Deep vein thrombosis (DVT): Swelling, pain, and redness in your leg
- Pulmonary embolism (PE): Shortness of breath, chest pain
- Stroke: Sudden weakness, difficulty speaking, vision problems
- Other clots: Can occur in any part of the body
Pregnancy Problems:
- Recurrent miscarriages (especially 3 or more in the first 3 months)
- Late pregnancy loss (after 3 months)
- Premature birth due to complications like pre-eclampsia
Other Features:
- Lacy purple-red skin pattern on your legs (livedo reticularis)
- Low platelet count (usually mild)
- Migraine headaches
How is APS Diagnosed?
Diagnosis requires both:
- Clinical event: A blood clot or pregnancy problem
- Positive antibody tests: Two positive blood tests for antiphospholipid antibodies, done at least 12 weeks apart
The two-test requirement is important because antibodies can be temporarily positive after infections or with certain medications.
How is APS Treated?
If You've Had a Blood Clot:
- Blood thinners (anticoagulants): Usually warfarin tablets, taken lifelong
- Target INR 2-3 (INR is a blood test that measures how thin your blood is)
- Regular blood tests (INR checks) to make sure the dose is right
- Important: Newer blood thinners (DOACs like rivaroxaban, apixaban) are generally NOT recommended in APS because they don't work as well
If You're Pregnant or Planning Pregnancy:
- Low-dose aspirin (75-100 mg daily): Start before conception
- Heparin injections (LMWH): Daily injections throughout pregnancy
- These medications dramatically improve the chance of a successful pregnancy (70-80% live birth rate)
- Close monitoring throughout pregnancy with extra scans
- Stop warfarin before conception (it can harm the baby); switch to heparin injections
If You Have Antibodies But No Symptoms:
- Your doctor may recommend:
- Low-dose aspirin (in some cases)
- Heparin injections during high-risk times (surgery, long flights, pregnancy)
- Avoiding estrogen-containing birth control pills
- "Healthy lifestyle: Don't smoke, control blood pressure and cholesterol"
Living with APS
Medication Adherence:
- Take your warfarin every day as prescribed
- Attend regular INR blood tests (usually monthly once stable)
- Carry a card or wear a bracelet saying you're on anticoagulants
Lifestyle:
- Avoid activities with high bleeding risk (contact sports, etc.)
- Tell all doctors and dentists you're on blood thinners before procedures
- Eat a consistent amount of vitamin K foods (leafy greens); don't cut them out, just be consistent
- Limit alcohol
Pregnancy Planning:
- Always discuss pregnancy plans with your doctor beforehand
- Switch from warfarin to heparin injections before trying to conceive
- Specialist antenatal care throughout pregnancy
Avoid High-Risk Factors:
- Estrogen contraceptives (the pill); use progesterone-only or non-hormonal methods
- Hormone replacement therapy (HRT)
- Smoking (increases clot risk)
- Prolonged immobility (long flights, bed rest)
Watch for Warning Signs:
- Leg swelling, pain, redness (DVT)
- Sudden shortness of breath, chest pain (PE)
- Sudden weakness, vision changes, speech difficulty (stroke)
- Unusual bleeding (if on anticoagulants)
- Seek urgent medical attention if these occur
Prognosis
With appropriate treatment:
- Most people with APS lead normal, active lives
- Lifelong anticoagulation dramatically reduces the risk of further blood clots (from 50-70% recurrence to 2-5%)
- Women with obstetric APS have a 70-80% chance of successful pregnancy with treatment
- Regular follow-up with your specialist is important
Without treatment, there is a very high risk of recurrent blood clots and pregnancy complications.
Questions to Ask Your Doctor
- What is my specific antibody profile? Am I triple-positive?
- What is my target INR, and how often do I need blood tests?
- If I'm planning pregnancy, when should I switch medications?
- What are the warning signs of a blood clot that I should watch for?
- Do I need to avoid any medications or foods?
- Are there any activities I should avoid?
12. References
-
Tektonidou MG, Andreoli L, Limper M, et al. EULAR recommendations for the management of antiphospholipid syndrome in adults. Ann Rheum Dis. 2019;78(10):1296-1304. PMID: 31092409
-
Sammaritano LR, Bermas BL, Chakravarty EE, et al. 2020 American College of Rheumatology Guideline for the Management of Reproductive Health in Rheumatic and Musculoskeletal Diseases. Arthritis Rheumatol. 2020;72(4):529-556. PMID: 31866276
-
Miyakis S, Lockshin MD, Atsumi T, et al. International consensus statement on an update of the classification criteria for definite antiphospholipid syndrome (APS). J Thromb Haemost. 2006;4(2):295-306. PMID: 16420554
-
Petri M. Epidemiology of antiphospholipid syndrome. Best Pract Res Clin Rheumatol. 2020;34(1):101462. PMID: 32413497
-
Ruiz-Irastorza G, Crowther M, Branch W, Khamashta MA. Antiphospholipid syndrome. Lancet. 2010;376(9751):1498-1509. PMID: 20822807
-
Cervera R, Piette JC, Font J, et al. Antiphospholipid syndrome: clinical and immunologic manifestations and patterns of disease expression in a cohort of 1,000 patients. Arthritis Rheum. 2002;46(4):1019-1027. PMID: 11953980
-
Giannakopoulos B, Krilis SA. The pathogenesis of the antiphospholipid syndrome. N Engl J Med. 2013;368(11):1033-1044. PMID: 23484830
-
Schreiber K, Sciascia S, de Groot PG, et al. Antiphospholipid syndrome. Nat Rev Dis Primers. 2018;4:17103. PMID: 29321641
-
Zuo Y, Barbhaiya M, Gockman K, et al. Antiphospholipid syndrome: a clinical perspective. Chin Med J (Engl). 2020;133(8):929-940. PMID: 32265421
-
de Groot PG, Urbanus RT. The significance of autoantibodies against β2-glycoprotein I. Blood. 2012;120(2):266-274. PMID: 22553314
-
Pierangeli SS, Espinola RG, Liu X, Harris EN. Thrombogenic effects of antiphospholipid antibodies are mediated by intercellular cell adhesion molecule-1, vascular cell adhesion molecule-1, and P-selectin. Circ Res. 2001;88(2):245-250. PMID: 11157681
-
Aldabie G, AlMarzooqi A, Aljohani R, et al. Navigating Obstetric antiphospholipid syndrome: pathophysiology, diagnosis, and therapeutic advances. Expert Rev Clin Immunol. 2026;22(1):51-68. PMID: 41498538
-
Tan Y, Qiao J, Yang S, et al. H3K4me3-Mediated FOXJ2/SLAMF8 Axis Aggravates Thrombosis and Inflammation in β2GPI/Anti-β2GPI-Treated Monocytes. Adv Sci (Weinh). 2024;11(20):e2308445. PMID: 38639399
-
Girardi G, Berman J, Redecha P, et al. Complement C5a receptors and neutrophils mediate fetal injury in the antiphospholipid syndrome. J Clin Invest. 2003;112(11):1644-1654. PMID: 14660741
-
Coss S, Braunstein EM. Complement system and human autoimmune diseases. J Autoimmun. 2023;137:102979. PMID: 36535812
-
Newman PJ, Zuraw BL. Antiphospholipid Syndrome: An Antibody-Mediated Disease With Emerging Therapeutic Opportunities. Arterioscler Thromb Vasc Biol. 2025;45(1):25-33. PMID: 40386969
-
D'Ippolito S, Marana R, Di Nicuolo F, et al. Antiphospholipid Syndrome in Pregnancy: New and Old Pathogenetic Mechanisms. Int J Mol Sci. 2023;24(5):4306. PMID: 36834614
-
Alijotas-Reig J, Esteve-Valverde E, Anunciação M, et al. Pathogenesis, Diagnosis and Management of Obstetric Antiphospholipid Syndrome: A Comprehensive Review. J Clin Med. 2022;11(3):675. PMID: 35160128
-
Pengo V, Ruffatti A, Legnani C, et al. Clinical course of high-risk patients diagnosed with antiphospholipid syndrome. J Thromb Haemost. 2010;8(2):237-242. PMID: 19874470
-
Moore GW, Foxton E, Platton S, et al. Triple-positive antiphospholipid syndrome does not guarantee positivity in each lupus anticoagulant assay. J Thromb Haemost. 2023;21(11):3177-3187. PMID: 37597725
-
Salter BS, Weiner MM, Trinh MA, et al. Catastrophic antiphospholipid syndrome: a CAPS-tivating hematologic disease. Blood. 2024;144(23):2331-2343. PMID: 39644034
-
Soto-Peleteiro A, Gómez-Puerta JA, Cervera R. Obstetric antiphospholipid syndrome. Lupus. 2024;33(8):825-834. PMID: 39174149
-
Depietri L, Veropalumbo MR, Leone MC, Ghirarduzzi A. Antiphospholipid Syndrome: State of the Art of Clinical Management. Cardiovasc Drugs Ther. 2025;39(3):533-550. PMID: 37572208
-
Rodriguez-Pinto I, Moitinho M, Santacreu I, et al. Catastrophic antiphospholipid syndrome: Lessons from the "CAPS Registry". Autoimmun Rev. 2024;23(9):103601. PMID: 39174151
-
Bitsadze V, Khizroeva J, Makatsariya A, et al. Catastrophic Antiphospholipid Syndrome. Hematol Oncol Clin North Am. 2024;38(2):433-451. PMID: 38203837
-
Cohen H, Cuadrado MJ, Erkan D, et al. 16th International Congress on Antiphospholipid Antibodies Task Force Report on Antiphospholipid Syndrome Treatment Trends. Lupus. 2020;29(12):1571-1593. PMID: 33100166
-
Devreese KMJ, Ortel TL, Pengo V, de Laat B. Laboratory criteria for antiphospholipid syndrome: communication from the SSC of the ISTH. J Thromb Haemost. 2018;16(4):809-813. PMID: 29380531
-
Barbhaiya M, Zuily S, Naden R, et al. The 2023 ACR/EULAR Antiphospholipid Syndrome Classification Criteria. Arthritis Rheumatol. 2023;75(10):1687-1702. PMID: 37635643
-
Barbhaiya M, Zuily S, Naden R, et al. 2023 ACR/EULAR antiphospholipid syndrome classification criteria. Ann Rheum Dis. 2023;82(10):1258-1270. PMID: 37640450
-
Cohen H, Hunt BJ, Efthymiou M, et al. Rivaroxaban versus warfarin to treat patients with thrombotic antiphospholipid syndrome, with or without systemic lupus erythematosus (RAPS): a randomised, controlled, open-label, phase 2/3, non-inferiority trial. Lancet Haematol. 2016;3(9):e426-e436. PMID: 27570089
-
Tektonidou MG. Antiphospholipid syndrome nephropathy: from pathogenesis to treatment. Front Immunol. 2018;9:1181. PMID: 29915586
-
Pengo V, Denas G, Zoppellaro G, et al. Rivaroxaban vs warfarin in high-risk patients with antiphospholipid syndrome. Blood. 2018;132(13):1365-1371. PMID: 30002145
-
Giron-Ortega A, Ruiz-Irastorza G. Direct-acting oral anticoagulants in antiphospholipid syndrome: A systematic review. Autoimmun Rev. 2023;22(7):103342. PMID: 37105842
-
Mak A, Cheung MW, Cheak AA, Ho RC. Combination of heparin and aspirin is superior to aspirin alone in enhancing live births in patients with recurrent pregnancy loss and positive anti-phospholipid antibodies: a meta-analysis of randomized controlled trials and meta-regression. Rheumatology (Oxford). 2010;49(2):281-288. PMID: 19933595
-
Barros RP, Ramakrishnan R, Chaturvedi S. Obstetric antiphospholipid syndrome. Best Pract Res Clin Haematol. 2021;34(3):101295. PMID: 34318477
-
Arnaud L, Mathian A, Ruffatti A, et al. Efficacy of aspirin for the primary prevention of thrombosis in patients with antiphospholipid antibodies: an international and collaborative meta-analysis. Autoimmun Rev. 2014;13(3):281-291. PMID: 24189283
-
Parepalli SA, Malemud CJ. Antiphospholipid Syndrome and Catastrophic Antiphospholipid Syndrome: A Comprehensive Review of Pathogenesis, Clinical Features, Diagnosis, and Novel Treatment Approaches. Antibodies (Basel). 2024;13(3):73. PMID: 39252716
-
Okunlola OO, Mbadugha CC, Davis F, et al. Catastrophic Antiphospholipid Syndrome: A Review of Current Evidence and Future Management Practices. Cureus. 2024;16(10):e71163. PMID: 39429267
-
Sciascia S, Baldovino S, Schreiber K, et al. Thrombotic risk assessment in APS: the Global APS Score (GAPSS). Lupus. 2018;27(9):1535-1539. PMID: 29973135
-
Cervera R, Serrano R, Pons-Estel GJ, et al. Morbidity and mortality in the antiphospholipid syndrome during a 10-year period: a multicentre prospective study of 1000 patients. Ann Rheum Dis. 2015;74(6):1011-1018. PMID: 24464962
-
Windisch R, Lau C, Mariampillai K, et al. Antiphospholipid Syndrome: Thrombotic and Vascular Complications. Rheum Dis Clin North Am. 2025;51(1):115-135. PMID: 37607079
-
Cohen H, Cuadrado MJ, Erkan D, et al. 16th International Congress on Antiphospholipid Antibodies Task Force Report on Antiphospholipid Syndrome Treatment Trends. Lupus. 2020;29(12):1571-1593. PMID: 33100166
-
Rand JH, Wu XX, Quinn AS, et al. Hydroxychloroquine directly reduces the binding of antiphospholipid antibody-beta2-glycoprotein I complexes to phospholipid bilayers. Blood. 2008;112(5):1687-1695. PMID: 18577708
13. Examination Focus
MRCP/FRACP Viva Points
Opening Statement (30 seconds):
"Antiphospholipid syndrome is an acquired autoimmune thrombophilia characterised by arterial or venous thrombosis and/or pregnancy morbidity in the presence of persistent antiphospholipid antibodies. The three criteria antibodies are lupus anticoagulant, anticardiolipin, and anti-β2-glycoprotein I, which must be positive on two occasions at least 12 weeks apart. It can be primary or secondary, most commonly associated with SLE. Management involves lifelong anticoagulation with warfarin targeting INR 2-3 for thrombotic APS, and aspirin plus LMWH for obstetric APS. DOACs are contraindicated, particularly in triple-positive patients. Catastrophic APS is a rare, life-threatening variant requiring aggressive multimodal treatment."
Key Facts for Rapid Recall
Three Criteria Antiphospholipid Antibodies:
- Lupus anticoagulant (LA) — most specific for thrombosis
- Anticardiolipin (aCL) IgG/IgM
- Anti-β2-glycoprotein I (anti-β2GPI) IgG/IgM
Classification Criteria (Revised Sapporo/Sydney):
- Clinical: ≥1 thrombosis OR pregnancy morbidity
- Laboratory: ≥1 aPL positive on ≥2 occasions ≥12 weeks apart
- Definite APS = ≥1 clinical + ≥1 laboratory
Triple Positivity:
- LA + aCL + anti-β2GPI all positive
- 5-fold increased thrombotic risk vs single-positive
- Highest-risk phenotype
- Absolute contraindication to DOACs
Management Essentials:
- Venous thrombosis: Warfarin INR 2-3 (lifelong)
- Arterial thrombosis: Warfarin INR 2-3 +/- aspirin (lifelong)
- Obstetric APS: Aspirin 75-100mg + prophylactic LMWH (pregnancy only)
- Prior thrombosis + pregnancy: Therapeutic LMWH throughout pregnancy
- AVOID DOACs (especially triple-positive): TRAPS trial showed harm
Catastrophic APS (CAPS):
- Multi-organ thrombosis (≥3 organs) over days
- Mortality 30-50%
- Treatment: Anticoagulation + steroids + PLEX/IVIG
- Common triggers: infection (47%), surgery (17%)
Common MRCP/PACES Scenarios
Scenario 1: Young Woman with Recurrent DVT
- Suspect APS (acquired thrombophilia)
- Test for aPL (LA, aCL, anti-β2GPI) × 2 (≥12 weeks apart)
- Exclude SLE (ANA, dsDNA, complement)
- If confirmed APS: Warfarin INR 2-3 lifelong
- Counsel: Avoid estrogen contraceptives; plan pregnancy (switch to LMWH)
Scenario 2: Stroke in 35-Year-Old with No Risk Factors
- Consider APS in differential (common cause in young stroke)
- Check aPL, thrombophilia screen, cardiovascular risk factors
- If APS: Warfarin INR 2-3 + aspirin 75mg (lifelong)
- Address modifiable risk factors
- Rehabilitation, secondary stroke prevention
Scenario 3: Recurrent Miscarriage (3 First-Trimester Losses)
- Investigate for obstetric APS
- aPL testing × 2 (≥12 weeks apart)
- Exclude anatomical (pelvic ultrasound), genetic (karyotyping), hormonal causes
- If APS: Aspirin + LMWH in future pregnancies (70-80% live birth rate)
- Preconception counselling, high-risk obstetric care
Scenario 4: Patient on Warfarin for APS, Planning Pregnancy
- Stop warfarin BEFORE conception (teratogenic weeks 6-12)
- Switch to therapeutic LMWH preconception
- Start aspirin 75-100mg daily
- Arrange high-risk antenatal care
- Folic acid 5mg daily
- Monitor: growth scans, Doppler, anti-Xa levels
- Postpartum: Resume warfarin after 5-7 days, continue LMWH until therapeutic INR
Scenario 5: Triple-Positive APS, Recurrent Thrombosis on Warfarin INR 2.5
- Assess adherence, time in therapeutic range
- Check for intercurrent illness, malignancy
- Consider:
- Increase INR target to 3-4 (high-intensity)
- Add aspirin 75mg daily
- Switch to therapeutic LMWH
- Add hydroxychloroquine
- Exclude other prothrombotic factors
- Multidisciplinary input (haematology, rheumatology)
Common Mistakes to Avoid
❌ Diagnosing APS on single positive aPL test
- MUST have 2 tests ≥12 weeks apart (transient positivity common)
❌ Using DOACs in APS, especially triple-positive
- TRAPS trial: rivaroxaban INFERIOR to warfarin; increased arterial events
- Warfarin remains standard of care
❌ Continuing warfarin in pregnancy
- Warfarin is teratogenic (weeks 6-12); switch to LMWH BEFORE conception
❌ Missing SLE in secondary APS workup
- 30-40% of SLE patients have aPL
- Always check ANA, dsDNA, complement in aPL-positive patients
❌ Forgetting postpartum anticoagulation
- Highest thrombotic risk period: 6 weeks postpartum
- Continue LMWH for ≥6 weeks; resume warfarin if prior thrombosis
❌ Assuming prolonged APTT means bleeding risk
- Lupus anticoagulant prolongs APTT but causes THROMBOSIS, not bleeding
- Paradoxical in vitro effect vs in vivo thrombosis
❌ Undertreating CAPS
- Requires triple therapy: anticoagulation + steroids + PLEX/IVIG
- Delayed treatment increases mortality
❌ Forgetting cardiovascular risk modification
- APS patients have high arterial event risk
- Address hypertension, smoking, dyslipidaemia, diabetes
Differential Diagnoses (APS Mimics)
Thrombotic Presentation:
- Inherited thrombophilias (Factor V Leiden, prothrombin G20210A, protein C/S/antithrombin deficiency)
- Malignancy-associated thrombosis
- Heparin-induced thrombocytopenia (HIT)
- Myeloproliferative neoplasms (polycythaemia vera, essential thrombocythaemia)
- Paroxysmal nocturnal haemoglobinuria (PNH)
- Hyperhomocysteinaemia
Pregnancy Loss:
- Anatomical (uterine septum, cervical incompetence)
- Genetic (parental karyotype abnormalities)
- Endocrine (thyroid disease, diabetes, PCOS)
- Inherited thrombophilias
- Infection
Catastrophic APS Mimics:
- Thrombotic thrombocytopenic purpura (TTP)
- Haemolytic uraemic syndrome (HUS)
- Disseminated intravascular coagulation (DIC)
- HELLP syndrome (pregnancy)
- Heparin-induced thrombocytopenia (HIT)
- Sepsis with multiorgan failure
Key Papers to Know
- TRAPS (2018): Rivaroxaban inferior to warfarin in triple-positive APS; DOACs contraindicated
- Revised Sapporo Criteria (2006): Classification criteria for APS diagnosis
- ACR/EULAR 2023 Criteria: Updated weighted scoring system for APS classification
- EULAR 2019 Recommendations: Comprehensive management guidelines for APS
- CAPS Registry: Largest dataset on catastrophic APS; mortality 30-50%, infection trigger 47%
Last Updated: 2026-01-09
Frequently asked questions
Quick clarifications for common clinical and exam-facing questions.
When should I seek emergency care for antiphospholipid syndrome?
Seek immediate emergency care if you experience any of the following warning signs: Catastrophic APS (multi-organ thrombosis), Stroke in young patient, Recurrent pregnancy loss, DVT/PE with positive aPL, Livedo reticularis with thrombosis.