Autoimmune Encephalitis
The most common subtype is anti-NMDA receptor encephalitis, which predominantly affects young women (median age 21 years) and is associated with ovarian teratoma in approximately 50% of female cases. Other important...
What matters first
The most common subtype is anti-NMDA receptor encephalitis, which predominantly affects young women (median age 21 years) and is associated with ovarian teratoma in approximately 50% of female cases. Other important...
Rapid cognitive decline (days to weeks)
9 Jan 2026
Generated educational material; verify before clinical use.
32 cited sources
See the concept before reading it
Study the key anatomy, imaging, and decision pathways as full teaching plates.
Clinical board
A visual summary of the highest-yield teaching signals on this page.
Urgent signals
Safety-critical features pulled from the topic metadata.
- Rapid cognitive decline (days to weeks)
- New-onset refractory status epilepticus (NORSE)
- Psychosis in young person without psychiatric history
- Movement disorder with encephalopathy
Content status and exam context
This page is AI-generated educational content. It may contain errors or omissions and is not a substitute for current guidelines, local protocols, senior clinical judgement, or professional medical advice.
MedVellum does not claim an individual clinician reviewer, board certification, or professional credential for this page unless a future version names a real, verifiable reviewer.
Clinical explanation and evidence
Autoimmune Encephalitis
1. Clinical Overview
Summary
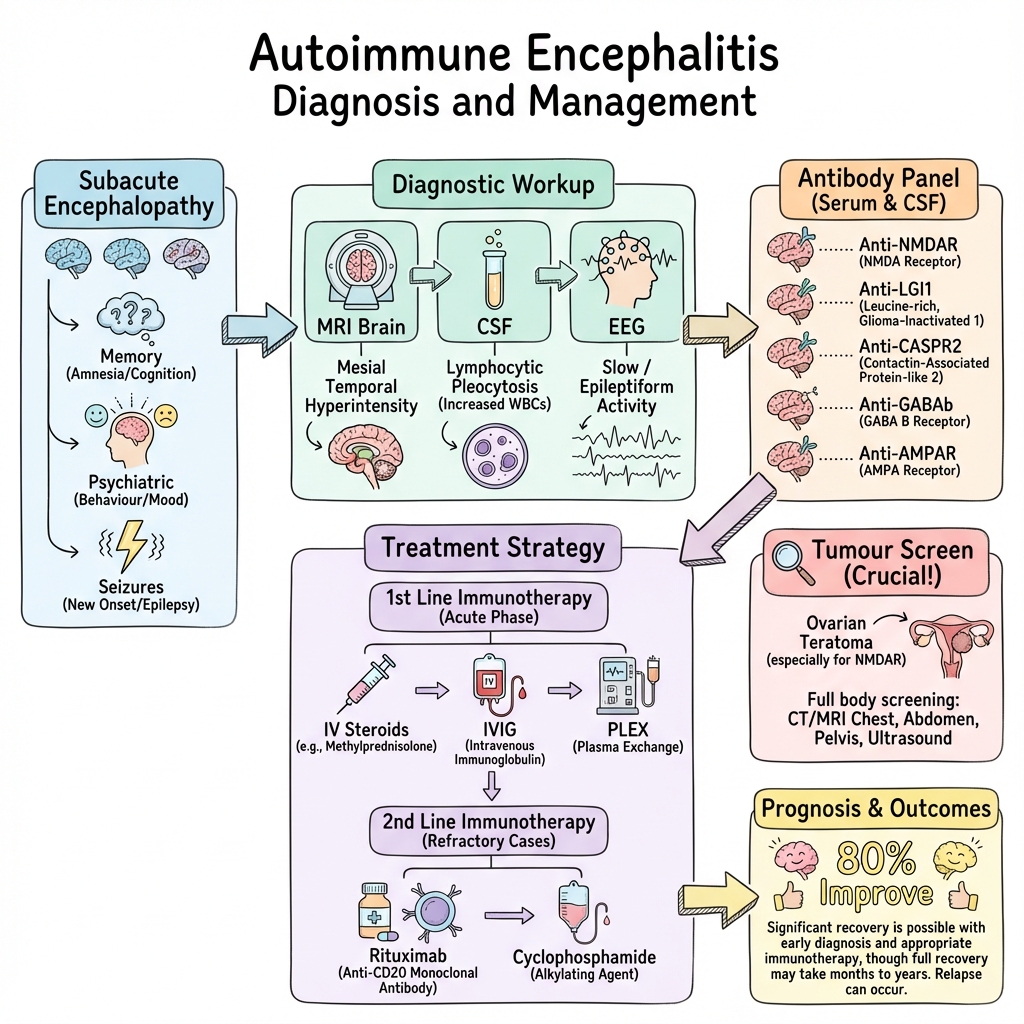
Autoimmune encephalitis (AE) represents a group of inflammatory brain disorders caused by antibodies directed against neuronal cell surface, synaptic, or intracellular proteins. Once considered rare, AE is now recognized to be as common as infectious encephalitis, with an incidence of 5-8 cases per million population annually. [1,2] The condition is frequently misdiagnosed, particularly when presenting with prominent psychiatric features. [3]
The most common subtype is anti-NMDA receptor encephalitis, which predominantly affects young women (median age 21 years) and is associated with ovarian teratoma in approximately 50% of female cases. [4] Other important antibody-mediated syndromes include anti-LGI1 (leucine-rich glioma-inactivated 1), anti-CASPR2 (contactin-associated protein-like 2), anti-GABAB, and anti-AMPAR encephalitis, each with distinct clinical phenotypes. [1]
The pathophysiology involves antibody binding to neuronal antigens, leading to receptor internalization and synaptic dysfunction rather than direct cytotoxic cell death—a critical distinction that explains the potential for complete reversibility with immunotherapy. [5] This reversibility underpins the importance of early recognition and treatment.
Diagnosis relies on clinical criteria incorporating subacute onset of cognitive impairment or psychiatric symptoms, combined with supportive findings including MRI abnormalities (mesial temporal lobe T2/FLAIR hyperintensity in 60-70%), CSF lymphocytic pleocytosis (approximately 80%), EEG abnormalities, and specific antibody detection in serum and/or CSF. [1,6]
First-line immunotherapy consists of high-dose corticosteroids, intravenous immunoglobulin (IVIG), or plasma exchange (PLEX). Second-line agents include rituximab and cyclophosphamide. Early treatment improves outcomes; approximately 80% of patients show clinical improvement, though recovery may be protracted over many months. [7] Tumor screening and removal, when applicable, is essential for long-term disease control and relapse prevention.
Key Facts
- Definition: Immune-mediated encephalitis caused by antibodies targeting neuronal cell surface, synaptic, or intracellular antigens
- Incidence: 5-8 per million per year; now as common as infectious encephalitis in some cohorts [1,2]
- Peak Demographics: Anti-NMDAR affects young women (15-35 years); anti-LGI1 affects older adults (50-70 years) [4,8]
- Associated Tumours: Ovarian teratoma (anti-NMDAR ~50% of women greater than 18 years), SCLC (anti-GABAB ~50%), thymoma (anti-CASPR2 ~20%) [1,7]
- Gold Standard Investigation: Neuronal antibody panel in both serum AND CSF (CSF more sensitive for anti-NMDAR) [1]
- First-line Treatment: IV methylprednisolone (1g daily × 5 days), IVIG (0.4g/kg × 5 days), or plasma exchange [9]
- Prognosis: 80% improve with treatment; 47% achieve full recovery; 20-24% relapse rate [7,10]
- Mortality: 4-7% with treatment (higher if treatment delayed or absent) [7]
Clinical Pearls
Diagnostic Pearl: "New-onset refractory status epilepticus (NORSE) in a young person without clear etiology should prompt immediate autoimmune encephalitis workup and empirical immunotherapy. Delay in treatment worsens neurological outcomes." [11]
Treatment Pearl: "Start immunotherapy early—within 4 weeks of symptom onset if possible. Do not wait for antibody results, as testing may take weeks and early treatment is associated with better functional outcomes." [7,9]
Pitfall Warning: "Anti-NMDAR encephalitis often presents initially to psychiatry with isolated psychosis, paranoia, or behavioral change. Up to 77% of patients have psychiatric symptoms as the first manifestation. Maintain high suspicion in young patients with new psychosis and no psychiatric history." [3,4]
Examination Pearl: "Faciobrachial dystonic seizures (FBDS)—brief (less than 3 seconds), frequent (10-50+ per day) dystonic posturing of the face and ipsilateral arm—are pathognomonic for anti-LGI1 encephalitis. Prompt immunotherapy can prevent progression to cognitive impairment." [12]
Mnemonic: NMDAR - New psychosis, Movement disorder, Dayslong seizures/status, Autonomic instability, Resembles schizophrenia
Why This Matters Clinically
Autoimmune encephalitis is treatable and potentially reversible, but outcomes are critically time-dependent. Delayed diagnosis leads to prolonged ICU admission, severe disability, and increased mortality. Psychiatric manifestations mean patients frequently present to mental health services first, resulting in diagnostic delays averaging 3-5 weeks from symptom onset. [3] Recognition by neurologists, psychiatrists, emergency physicians, intensivists, and general practitioners is essential. With early immunotherapy, the majority of patients recover substantially, but without treatment, mortality approaches 25%. [7]
2. Epidemiology
Incidence and Prevalence
Overall Incidence:
- Autoimmune encephalitis: 5-8 per million per year [1,2]
- Anti-NMDAR encephalitis: 1.5 per million per year (most common antibody-mediated subtype) [2]
- Anti-LGI1 encephalitis: 0.8 per million per year (second most common) [13]
- Comparable incidence to infectious encephalitis (including HSV encephalitis) in some populations [2]
- Likely underestimated due to diagnostic challenges and antibody-negative cases [6]
Relative Frequency Among Antibody Subtypes:
- Anti-NMDAR: ~40% of autoimmune encephalitis cases [1]
- Anti-LGI1: ~20% of autoimmune encephalitis cases
- Anti-GABAB: ~5-10%
- Anti-CASPR2, AMPAR, GABAA: Each less than 5% [1]
Demographics by Antibody Type
| Antibody | Median Age (Range) | Sex Ratio | Tumor Association | Tumor Type |
|---|---|---|---|---|
| Anti-NMDAR | 21 years (15-35) | F:M = 4:1 | 50% (women greater than 18) | Ovarian teratoma |
| Anti-LGI1 | 60 years (50-70) | M:F = 2:1 | 5-10% | Thymoma, SCLC (rare) |
| Anti-CASPR2 | 60 years (40-70) | M:F = 10:1 | 20-30% | Thymoma |
| Anti-GABAB | 60 years (50-70) | M = F | 50-60% | SCLC |
| Anti-AMPAR | 60 years (50-70) | F:M = 2:1 | 60-70% | SCLC, breast, thymoma |
| Anti-GABAA | 40 years (20-60) | F:M = 2:1 | Rare | Thymoma (rare) |
Geographical and Ethnic Variations
- No significant geographical variation identified in available studies
- Anti-NMDAR encephalitis reported across all ethnic groups
- Asian populations may have higher representation in some series, potentially due to reporting bias or genetic susceptibility factors (requires further investigation)
- Higher recognition rates in developed healthcare systems likely reflect improved diagnostic access rather than true epidemiological differences
Risk Factors
Established:
- Female sex (anti-NMDAR)
- Presence of teratoma or other neuronal antigen-expressing tumor [4]
- Prior HSV encephalitis (risk factor for subsequent anti-NMDAR encephalitis) [14]
Possible:
- Genetic susceptibility (HLA associations under investigation)
- Viral prodrome (reported in 30-40% of anti-NMDAR cases) [4]
3. Pathophysiology
Molecular Mechanisms
Autoimmune encephalitis involves antibody-mediated synaptic dysfunction through distinct mechanisms depending on antibody type and target location.
Cell Surface and Synaptic Antibodies (Directly Pathogenic)
Step 1: Immune Trigger
- Unknown triggers in majority of cases
- Viral prodrome: Reported in 30-40% of anti-NMDAR cases (non-specific upper respiratory or gastrointestinal illness) [4]
- Post-infectious: HSV encephalitis can trigger anti-NMDAR encephalitis weeks to months later (proposed mechanism: viral-induced neuronal damage exposes intracellular antigens, breaking immune tolerance) [14]
- Tumor expression: Teratomas contain neural tissue expressing NMDA receptors; immune response to tumor antigens cross-reacts with brain receptors [4]
- Genetic susceptibility: HLA associations suggested (e.g., HLA-DRB1*16:02 with anti-LGI1) but incompletely characterized
Step 2: Antibody Production and CNS Access
- B-cell activation and clonal expansion
- Production of IgG antibodies against neuronal surface antigens
- Antibodies cross blood-brain barrier (mechanism: passive diffusion in areas of BBB disruption, active transport, or intrathecal synthesis)
- Intrathecal antibody synthesis often present (CSF oligoclonal bands in 60%) [6]
Step 3: Antibody-Receptor Interaction
-
Anti-NMDAR: Antibodies bind to GluN1 subunit of NMDA receptor → cross-linking → receptor internalization via endocytosis → decreased NMDA receptor density at synapse → NMDA receptor hypofunction [5]
- "Clinical correlation: NMDA hypofunction produces psychosis (similar to ketamine/phencyclidine), movement disorders (loss of GABAergic interneuron modulation), seizures (excitatory-inhibitory imbalance)"
-
Anti-LGI1: LGI1 is a secreted protein bridging presynaptic ADAM22/23 and postsynaptic AMPA receptors → antibodies disrupt LGI1-ADAM interaction → reduced AMPA receptor trafficking → impaired synaptic transmission [8]
- "Additional effect: voltage-gated potassium channel (VGKC) dysfunction → neuronal hyperexcitability → seizures (including faciobrachial dystonic seizures)"
-
Anti-CASPR2: CASPR2 is a cell adhesion molecule associated with voltage-gated potassium channels (Kv1) at juxtaparanodes → antibodies reduce CASPR2 surface expression → channel dysfunction → peripheral nerve hyperexcitability (neuromyotonia) and CNS hyperexcitability
-
Anti-GABAB: Antibodies target GABAB receptor → internalization → reduced inhibitory neurotransmission → seizures (prominent feature) [15]
Step 4: Synaptic Dysfunction (Reversible)
- Critical concept: Cell surface antibody-mediated AE causes functional synaptic disruption WITHOUT direct cytotoxic neuronal death (unlike paraneoplastic intracellular antibody syndromes)
- Receptor density reduction is reversible with antibody removal
- Neuronal architecture preserved
- Explains potential for complete clinical recovery with immunotherapy [5]
Step 5: Secondary Consequences
- Prolonged synaptic dysfunction → excitotoxicity risk
- Inflammatory milieu → microglial activation, cytokine release
- Chronic cases → potential for secondary neuronal injury and irreversible damage
Intracellular Antibodies (Paraneoplastic—Markers, Not Pathogenic)
Classical Paraneoplastic Antibodies (anti-Hu, anti-Yo, anti-Ri, anti-Ma2, anti-CV2, anti-amphiphysin):
- Antibodies target intracellular neuronal proteins
- Not directly pathogenic (cannot cross cell membrane to reach target)
- Markers of cytotoxic T-cell-mediated neuronal death (CD8+ T-cells are true effectors)
- Paraneoplastic etiology: tumor expresses neuronal antigens → immune response targets both tumor and normal neurons
- Poor response to immunotherapy (cell death is irreversible)
- Outcome depends primarily on tumor control [16]
Pathological Findings
Histopathology (rare biopsy cases):
- Perivascular lymphocytic infiltrates (CD4+ and CD8+ T-cells, B-cells)
- Microglial nodules
- Minimal neuronal loss in cell surface antibody-mediated AE (contrast with paraneoplastic syndromes)
- Mesial temporal lobe inflammation in limbic variants
Imaging-Pathology Correlation:
- MRI T2/FLAIR hyperintensity correlates with inflammatory edema, not neuronal loss
- FDG-PET hypometabolism indicates synaptic dysfunction, often more extensive than MRI changes [17]
4. Clinical Presentation
Temporal Evolution and Symptom Progression
Typical Course:
- Subacute onset: Symptoms develop over days to weeks (rarely acute or chronic)
- Diagnostic criterion: Less than 3 months from symptom onset to full syndrome [1]
- Progression: Initial psychiatric/cognitive symptoms → seizures → movement disorders → decreased consciousness → autonomic instability (classic anti-NMDAR progression)
Presentation by Antibody Subtype
Anti-NMDAR Encephalitis
Phase 1: Prodrome (Present in 30-40%)
- Fever, headache, malaise, upper respiratory symptoms (viral-like illness)
- Duration: Days to 2 weeks
- Non-specific; often attributed to viral infection
Phase 2: Psychiatric/Cognitive Symptoms (70-80% Present First)
- Psychosis: Paranoia, hallucinations (auditory and visual), delusions
- Behavioral change: Agitation, aggression, hypersexuality, inappropriate behavior
- Mood symptoms: Depression, anxiety, emotional lability, mania
- Cognitive impairment: Short-term memory loss, confusion, disorientation
- Often misdiagnosed as primary psychiatric disorder (schizophrenia, bipolar disorder, drug-induced psychosis) [3]
Phase 3: Seizures (90% of Patients)
- Focal or generalized
- New-onset refractory status epilepticus (NORSE) in 30-40% [11]
- Often difficult to control with anti-seizure medications alone
Phase 4: Movement Disorders (80%)
- Orofacial dyskinesias: Lip-smacking, grimacing, chewing movements
- Choreoathetosis: Involuntary writhing movements
- Dystonia: Sustained muscle contractions
- Catatonia: Mutism, posturing, waxy flexibility, negativism
- Stereotyped movements
- Can be bizarre and lead to misdiagnosis as conversion disorder
Phase 5: Decreased Consciousness (50-60%)
- Progressive somnolence → stupor → coma
- Often requires ICU admission
Phase 6: Autonomic Instability (60%)
- Cardiovascular: Tachycardia, bradycardia, blood pressure lability, arrhythmias
- Respiratory: Central hypoventilation (requires mechanical ventilation in 20%)
- Hyperthermia
- Hypersalivation, diaphoresis, urinary retention
- Life-threatening; requires intensive care monitoring
Phase 7: Recovery (With Treatment)
- Reverse order: Autonomic stabilization → awakening → movement disorder resolution → cognitive improvement → psychiatric recovery
- Protracted: Cognitive and psychiatric recovery may take 6-18 months [7]
Pediatric Differences:
- Younger children (less than 12 years) less likely to have psychiatric symptoms
- More likely to present with seizures, movement disorder, speech dysfunction
- Boys more commonly affected in childhood (sex ratio equalizes in adolescence) [18]
Anti-LGI1 Encephalitis
Core Features: [12,28,29,32]
- Faciobrachial dystonic seizures (FBDS): Pathognomonic feature (present in 50-53%) [12,28,32]
- Brief (typically less than 3 seconds), frequent (10-50+ per day), stereotyped
- Dystonic posturing of face and ipsilateral arm
- May not have ictal EEG correlate (clinical seizure vs. movement disorder debate)
- "Critical: Heralds progression to cognitive impairment; urgent immunotherapy required to prevent irreversible memory deficits [32]"
- Limbic encephalitis: Subacute memory impairment (anterograde amnesia), confusion, disorientation; short-term memory loss in 75% [28]
- Hyponatraemia: SIADH in 55-60% (can precede neurological symptoms); low serum osmolality, low BUN, low uric acid, high urinary sodium [8,28,29]
- Sleep disorders: Insomnia, REM sleep behavior disorder (RBD) in 34% [28]
- Seizures: Temporal lobe seizures (distinct from FBDS) in 68% [28]
- Psychiatric symptoms: Anxiety, depression, hallucinations in 58% (less prominent than anti-NMDAR) [28]
Typical Patient Profile:
- Male (M:F = 2:1), median age 60 years
- Excellent response to immunotherapy (among best prognoses of all AE subtypes) [13]
Anti-CASPR2 Encephalitis
Clinical Syndromes:
-
Morvan Syndrome (Classic Triad):
- Neuromyotonia (peripheral nerve hyperexcitability → muscle fasciculations, cramps, stiffness)
- Insomnia (profound, near-total sleep loss)
- Autonomic dysfunction (hyperhidrosis, constipation, urinary retention)
- Encephalopathy (confusion, hallucinations)
- Neuropathic pain
-
Limbic Encephalitis:
- Memory impairment, confusion
- Seizures
- Psychiatric symptoms
-
Neuromyotonia Alone (Isaacs' syndrome):
- Continuous muscle fiber activity
- Without cognitive symptoms
Tumor Association:
- Thymoma in 20-30% (mandates CT chest)
Anti-GABAB Encephalitis
Core Features:
- Seizures: Prominent, often early and severe
- Limbic encephalitis: Memory loss, confusion
- Rapidly progressive
Tumor Association:
- SCLC in 50-60%: Requires urgent CT chest, often disseminated disease [15]
- Neurological symptoms may precede cancer diagnosis
Anti-AMPAR Encephalitis
Core Features:
- Limbic encephalitis: Subacute memory impairment, confusion, psychiatric symptoms
- Seizures: Present in 60-70%
- Psychiatric symptoms: Prominent
Tumor Association:
- 60-70% paraneoplastic (SCLC, breast cancer, thymoma)
- Relapse rate high (50-60%)
Anti-GABAA Receptor Encephalitis
Core Features:
- Refractory status epilepticus: Most common presentation
- Encephalopathy: Prominent
- Multifocal cortical inflammation: MRI shows multifocal cortical FLAIR hyperintensities (differs from typical mesial temporal pattern)
Demographics:
- Younger adults (median age 40)
- Female predominance
Red Flags Requiring Urgent Workup
[!CAUTION] IMMEDIATE autoimmune encephalitis evaluation required for:
- Rapid cognitive decline over days to weeks in previously healthy person
- New-onset refractory status epilepticus (NORSE) without clear cause
- Young person with first psychotic episode and no psychiatric history
- Movement disorder (dyskinesia, dystonia, choreoathetosis) WITH encephalopathy
- Autonomic instability (tachycardia, hypoventilation, hyperthermia) with encephalopathy
- Faciobrachial dystonic seizures (anti-LGI1—treat urgently)
- Subacute memory impairment with hyponatraemia (anti-LGI1)
- Encephalitis with known malignancy (especially SCLC, ovarian, thymoma)
5. Clinical Examination
Structured Assessment
Cognitive and Psychiatric Examination
Cognitive Domains:
- Orientation: Time, place, person
- Attention: Digit span, serial 7s
- Short-term memory: Three-item recall at 5 minutes (often severely impaired in limbic variants)
- Executive function: Frontal lobe tasks
- Language: Usually preserved (except in severe encephalopathy)
- Standardized tools: Mini-Mental State Examination (MMSE), Montreal Cognitive Assessment (MoCA)
Psychiatric Assessment:
- Psychosis: Screen for hallucinations (auditory, visual), delusions, paranoia
- Mood: Depression, mania, lability
- Behavior: Agitation, disinhibition, inappropriate sexual behavior
- Catatonia: Mutism, posturing, waxy flexibility, negativism, echolalia
- Screening tool: Bush-Francis Catatonia Rating Scale
Neurological Examination
Movement Disorders (observe throughout examination):
- Orofacial dyskinesias: Lip-smacking, chewing, grimacing
- Chorea: Involuntary, brief, irregular movements
- Athetosis: Slow, writhing movements
- Dystonia: Sustained muscle contractions, abnormal postures
- Stereotypies: Repetitive, purposeless movements
- Faciobrachial dystonic seizures: Brief (less than 3 seconds) dystonic posturing of face/arm (may need to observe over time or review video)
Seizures:
- Witnessed seizure activity
- Focal vs. generalized
- Status epilepticus (continuous or recurrent seizures without recovery)
Cerebellar Signs:
- Ataxia, dysmetria, dysdiadochokinesia (especially anti-Hu, anti-Yo paraneoplastic syndromes)
Brainstem Signs:
- Cranial nerve deficits (rare in pure AE; consider brainstem encephalitis or Bickerstaff encephalitis if present)
- Ophthalmoplegia (anti-GQ1b, Bickerstaff)
Peripheral Nerve Hyperexcitability (anti-CASPR2):
- Continuous muscle fasciculations (neuromyotonia)
- Muscle stiffness, cramps
Autonomic Examination:
- Cardiovascular: Heart rate, blood pressure (check for orthostatic hypotension or lability)
- Respiratory: Respiratory rate, pattern (central hypoventilation risk)
- Temperature: Hyperthermia
- Pupils: Size, reactivity
- Hypersalivation, diaphoresis
General Examination for Tumor Screening
Targeted Examination:
- Women with suspected anti-NMDAR: Pelvic examination (ovarian mass)
- Anti-GABAB, AMPAR: Respiratory examination (SCLC), lymphadenopathy, hepatomegaly
- Anti-CASPR2, Hu: Anterior mediastinal mass (thymoma)
- All patients: Thorough systemic examination for occult malignancy
Examination Findings by Severity
| Severity | Consciousness | Movement Disorder | Autonomic | ICU Required |
|---|---|---|---|---|
| Mild | Alert, confused | Mild dyskinesias | Stable vitals | No |
| Moderate | Somnolent, arousable | Prominent dyskinesias/dystonia | Tachycardia | Possibly |
| Severe | Stupor or coma | Severe, disabling | Hypoventilation, BP lability | Yes |
6. Investigations
Diagnostic Criteria (Graus 2016 Criteria)
Possible Autoimmune Encephalitis (Clinical diagnosis; treat empirically): [1]
All three required:
- Subacute onset (less than 3 months) of working memory deficits, altered mental status, or psychiatric symptoms
- At least ONE of:
- New focal CNS findings
- Seizures not explained by previous seizure disorder
- CSF pleocytosis (greater than 5 WBC/μL)
- MRI features suggestive of encephalitis
- Reasonable exclusion of alternative causes
Definite Autoimmune Encephalitis:
- Meets criteria for possible AE PLUS
- Detection of specific neuronal antibody in serum or CSF
Antibody Testing
Specimen Requirements:
- Both serum AND CSF for anti-NMDAR, anti-GABAB, anti-AMPAR (CSF more sensitive for NMDAR)
- Serum often sufficient for anti-LGI1, anti-CASPR2 (though CSF recommended if serum negative)
Panel Recommendations:
| First-line Panel | Comments |
|---|---|
| Anti-NMDAR (NR1 subunit) | Most common; CSF more sensitive than serum [19] |
| Anti-LGI1 (leucine-rich glioma-inactivated 1) | Second most common; serum usually sufficient |
| Anti-CASPR2 (contactin-associated protein-like 2) | May be low titre |
| Anti-GABAB (GABA-B receptor) | High SCLC association |
| Anti-AMPAR (AMPA receptor) | High paraneoplastic association |
| Extended Panel | Indications |
|---|---|
| Anti-GABAA receptor | Refractory status epilepticus with multifocal MRI changes |
| Anti-DPPX (dipeptidyl-peptidase-like protein-6) | Hyperexcitability, diarrhea, CNS symptoms |
| Anti-mGluR5 (metabotropic glutamate receptor 5) | Limbic encephalitis (Ophelia syndrome with Hodgkin lymphoma) |
| Anti-IgLON5 | Sleep disorder, bulbar dysfunction, tau pathology |
| Paraneoplastic Antibodies (Intracellular) | Associated Cancer |
|---|---|
| Anti-Hu (ANNA-1) | SCLC |
| Anti-Yo (PCA-1) | Ovarian, breast |
| Anti-Ri (ANNA-2) | Breast, gynecological, SCLC |
| Anti-Ma2 (Ta) | Testicular (young men), lung |
| Anti-CV2 (CRMP5) | SCLC, thymoma |
| Anti-amphiphysin | Breast, SCLC |
Interpretation:
- Positive serum + positive CSF: Definite
- Positive CSF only: Definite (intrathecal synthesis)
- Positive serum only: Interpret with caution (low-titre positives may be false positives or non-pathogenic)
- Negative antibodies: Does NOT exclude AE; 10-15% of cases antibody-negative [6]
- Antibody titres: Correlate poorly with disease severity; not useful for monitoring treatment response [19]
Cerebrospinal Fluid (CSF) Analysis
Typical Findings:
| Parameter | Frequency | Typical Values |
|---|---|---|
| Lymphocytic pleocytosis | 80% | 10-100 WBC/μL (median ~20) [6] |
| Elevated protein | 30-50% | 0.5-1.0 g/L (usually mildly elevated) |
| Oligoclonal bands (CSF-specific) | 60% | Indicates intrathecal antibody synthesis |
| Glucose | Normal | Normal CSF:serum glucose ratio |
| Gram stain/culture | Negative | Essential to exclude bacterial meningitis |
| HSV PCR | Negative | Exclude HSV encephalitis |
Atypical Features:
- Normal CSF in 20% (does not exclude AE) [6]
- Neutrophilic pleocytosis (rare; consider overlap with infectious process)
- Very high protein (greater than 1.5 g/L; consider alternative diagnosis)
Additional CSF Tests:
- Neuronal antibody panel (send simultaneously with serum)
- Opening pressure (usually normal; elevated if comorbid venous thrombosis or hydrocephalus)
- Cytology (if paraneoplastic syndrome suspected)
Magnetic Resonance Imaging (MRI) Brain
Typical Findings:
| Antibody | MRI Abnormality Frequency | Typical Pattern |
|---|---|---|
| Anti-NMDAR | 50-60% | Mesial temporal lobe T2/FLAIR hyperintensity (unilateral or bilateral); may involve cortical, subcortical, brainstem areas [17] |
| Anti-LGI1 | 60-70% | Mesial temporal lobe T2/FLAIR hyperintensity; basal ganglia involvement (especially putamen) in 40%; hippocampal atrophy on follow-up [20] |
| Anti-CASPR2 | 40-50% | Mesial temporal lobe hyperintensity |
| Anti-GABAB | 70% | Mesial temporal lobe T2/FLAIR hyperintensity |
| Anti-GABAA | 80% | Multifocal cortical FLAIR hyperintensities (distinct from typical limbic pattern) |
Key Points:
- Normal MRI in 30-40% (especially early anti-NMDAR) [17]
- T1 post-contrast: Mild enhancement in minority; prominent enhancement suggests alternative diagnosis (e.g., tumor, infection)
- Mesial temporal lobe hyperintensity is NON-SPECIFIC (differential: HSV encephalitis, glioma, post-ictal changes, limbic AE of any cause)
- Follow-up MRI may show hippocampal atrophy (correlates with persistent memory impairment)
MRI Protocol:
- T1, T2, FLAIR, diffusion-weighted imaging (DWI), T1 post-gadolinium
- Thin slices through temporal lobes
FDG-PET Brain (If Available):
- More sensitive than MRI (abnormal in 80-90% when MRI normal) [17]
- Pattern: Frontotemporal, basal ganglia hypometabolism (anti-NMDAR)
- Occipital hypometabolism (anti-LGI1)
- Useful when MRI negative but clinical suspicion high
Electroencephalography (EEG)
Indications:
- Seizure characterization (focal vs. generalized; ictal vs. interictal)
- Assess for non-convulsive status epilepticus
- Prognostic information
Common Findings (Nonspecific):
- Diffuse slowing (theta/delta activity)
- Focal or generalized epileptiform discharges
- Temporal lobe slowing or sharp waves (limbic encephalitis)
Specific Findings:
| Pattern | Association | Description |
|---|---|---|
| Extreme delta brush (EDB) | Anti-NMDAR (30%) | Rhythmic delta activity (1-3 Hz) with superimposed beta frequency (20-30 Hz) [21]; resembles neonatal EEG pattern; relatively specific for anti-NMDAR |
| Faciobrachial dystonic seizures | Anti-LGI1 | Often no ictal EEG correlate (debate: seizure vs. movement disorder) [12] |
Prognostic Value:
- Extreme delta brush associated with more severe disease but does not predict long-term outcome
- Persistent EEG abnormalities correlate with cognitive sequelae
Tumor Screening
Rationale:
- Paraneoplastic AE: Tumor expresses neuronal antigens → immune response
- Tumor removal improves neurological outcome and reduces relapse risk [7]
- Some antibodies have high tumor associations (e.g., GABAB 50-60%, AMPAR 60-70%)
Screening Strategy by Antibody:
| Antibody | Tumor Association | Recommended Imaging |
|---|---|---|
| Anti-NMDAR | Women greater than 18: ~50% ovarian teratoma; Men/children: less than 5% | Women: Pelvic MRI or transvaginal ultrasound; Men/children: Consider CT chest/abdomen/pelvis if high suspicion |
| Anti-LGI1 | 5-10% (thymoma rare) | CT chest if clinically indicated |
| Anti-CASPR2 | 20-30% thymoma | CT chest (mandatory) |
| Anti-GABAB | 50-60% SCLC | CT chest (mandatory); consider PET-CT if CT negative |
| Anti-AMPAR | 60-70% (SCLC, breast, thymoma) | CT chest, mammography (women), consider whole-body PET-CT |
| Anti-Hu, CV2, amphiphysin | greater than 90% SCLC or other | CT chest/abdomen/pelvis, PET-CT |
Timing:
- Perform at diagnosis (initial screening)
- Repeat if initially negative: 3-6 months, 1 year, then annually for 5 years (especially anti-NMDAR teratoma surveillance)
- PET-CT whole-body if high clinical suspicion and CT negative (higher sensitivity)
Ovarian Teratoma:
- Often small, unilateral
- Transvaginal ultrasound more sensitive than MRI for small lesions
- Dermoid cyst on imaging (fat, calcification, solid components)
7. Differential Diagnosis
Infectious Encephalitis
| Feature | Autoimmune | Infectious (HSV) |
|---|---|---|
| Onset | Subacute (days-weeks) | Acute (hours-days) |
| Fever | Prodrome 30%; low-grade | High-grade fever common |
| CSF | Lymphocytic; oligoclonal bands | Lymphocytic; RBC (hemorrhagic); HSV PCR positive |
| MRI | Mesial temporal (variable) | Mesial temporal (early); hemorrhagic necrosis |
| Movement disorder | Common (NMDAR) | Rare |
| Antibody | Positive | Negative (or post-infectious AE) |
Overlap:
- Post-HSV autoimmune encephalitis (choreoathetosis weeks after treated HSV encephalitis; anti-NMDAR antibodies) [14]
Primary Psychiatric Disorders
| Feature | Anti-NMDAR Encephalitis | Schizophrenia / Mania |
|---|---|---|
| Age | Any (peak 15-35) | Adolescence/early adulthood |
| Onset | Acute/subacute (days-weeks) | Insidious (months) |
| Progression | Rapid deterioration | Stable or gradual |
| Movement disorder | Prominent dyskinesias, dystonia | Absent (unless drug-induced) |
| Seizures | 90% | Rare |
| Autonomic | Common (hypoventilation) | Absent |
| Cognitive | Severe, global impairment | Negative symptoms, thought disorder |
| CSF | Abnormal (80%) | Normal |
| MRI | Abnormal (50-60%) | Normal |
| Response to antipsychotics | Poor; may worsen (NMS-like) | Improves |
Red Flags for AE (Not Primary Psychiatric):
- Seizures
- Movement disorder (non-drug-induced)
- Rapid cognitive decline
- Autonomic instability
- CSF pleocytosis
- Abnormal MRI
Toxic-Metabolic Encephalopathy
| Cause | Key Features | Differentiation |
|---|---|---|
| Drug intoxication (anticholinergics, sympathomimetics) | Toxidrome, acute onset, resolves with clearance | Urine drug screen; rapid improvement |
| Serotonin syndrome | SSRI/MAOI use, hyperthermia, rigidity, clonus | Recent medication change; autonomic hyperactivity |
| Neuroleptic malignant syndrome | Antipsychotic use, rigidity, hyperthermia, CK elevation | Antipsychotic exposure; lead-pipe rigidity |
| Hepatic encephalopathy | Liver disease, asterixis, ammonia elevated | LFTs, ammonia; no seizures |
| Wernicke's encephalopathy | Alcohol, triad (confusion, ataxia, ophthalmoplegia) | Thiamine deficiency; responds to replacement |
Other Autoimmune/Inflammatory
| Condition | Key Differentiators |
|---|---|
| Neurosarcoidosis | Cranial nerve palsies, meningeal enhancement, systemic sarcoid, ACE elevated |
| CNS vasculitis | Stroke-like episodes, multifocal lesions, angiography abnormalities |
| Acute disseminated encephalomyelitis (ADEM) | Post-infectious, children, multifocal demyelination, monophasic |
| Multiple sclerosis | Relapsing-remitting, periventricular lesions, oligoclonal bands |
| Hashimoto encephalopathy | Thyroid antibodies (anti-TPO, anti-thyroglobulin), steroid-responsive, diagnosis of exclusion |
Seizure-Related
| Condition | Differentiation |
|---|---|
| Non-convulsive status epilepticus (primary epilepsy) | Prior seizure disorder, ictal EEG, no encephalitis features |
| Post-ictal state | Follows known seizure, improves over hours to days |
Neoplastic
| Condition | Differentiation |
|---|---|
| Primary CNS lymphoma | Mass lesion, ring enhancement, positive CSF cytology, HIV/immunosuppression |
| Glioma | Mass effect, progressive focal deficits, contrast enhancement |
8. Management
Initial Assessment and Stabilization
Emergency Department / Acute Setting:
-
Airway, Breathing, Circulation:
- Assess for hypoventilation (central apnea in anti-NMDAR)
- Monitor oxygen saturation, respiratory rate
- Secure airway if GCS ≤8 or impaired protective reflexes
-
Seizure Management:
- Treat active seizures with benzodiazepines (lorazepam 4mg IV or diazepam 10mg IV)
- Status epilepticus protocol if seizures prolonged or recurrent
- EEG monitoring for non-convulsive status
-
Exclude Mimics:
- Rapid glucose (exclude hypoglycemia)
- Toxicology screen (drug intoxication)
- CT head (exclude hemorrhage, mass lesion)
- Lumbar puncture (if safe) for HSV PCR, bacterial culture (treat empirically for HSV and bacterial meningitis until excluded)
-
ICU Admission Criteria:
- Decreased consciousness (GCS less than 12)
- Autonomic instability (hypoventilation, severe tachycardia/bradycardia, hyperthermia)
- Refractory seizures or status epilepticus
- Severe agitation requiring sedation
Diagnostic Workup (Parallel to Treatment)
First-line Investigations (Perform urgently):
- MRI brain with gadolinium
- Lumbar puncture: Cell count, protein, glucose, Gram stain/culture, HSV PCR, oligoclonal bands
- Serum and CSF neuronal antibody panel (send to reference laboratory)
- EEG (if seizures or altered consciousness)
- Baseline blood: FBC, CRP, U&E, LFT, TFT, autoimmune screen (ANA, ENA), HIV, syphilis serology
Tumor Screening (Based on antibody, if known, or empirically):
- Women with suspected anti-NMDAR: Pelvic MRI or transvaginal ultrasound
- All adults: CT chest/abdomen/pelvis
- Consider whole-body FDG-PET if high paraneoplastic risk
First-Line Immunotherapy
Indication: Start empirically if clinical suspicion high (meet criteria for possible AE); do NOT wait for antibody results. [1,9]
Rationale: Early treatment (within 4 weeks) associated with better outcomes. [7]
Options (equivalent efficacy; choose based on availability and contraindications):
| Treatment | Dose | Duration | Notes |
|---|---|---|---|
| IV Methylprednisolone | 1g IV daily | 5 days | Followed by oral prednisolone taper (1mg/kg, taper over 6-8 weeks) |
| IVIG | 0.4g/kg IV daily OR 2g/kg divided over 2-5 days | 5 days | Monitor for renal impairment, thrombosis, aseptic meningitis |
| Plasma Exchange (PLEX) | 5-7 exchanges (1-1.5 plasma volumes each) | Every other day | Requires central venous access; risk of hypotension, infection |
Combination Therapy:
- Often use corticosteroids + IVIG or corticosteroids + PLEX concurrently
- No RCT data; based on observational evidence
Expected Response:
- Improvement typically seen within 2-4 weeks
- Assess response at 2-4 weeks before escalating to second-line
- Early immunotherapy associated with 83% good outcome rate (mRS ≤2) at 6 months in anti-NMDAR encephalitis [23]
Second-Line Immunotherapy
Indications:
- Inadequate response to first-line therapy at 2-4 weeks
- Severe disease at presentation (e.g., ICU admission, refractory status epilepticus)
- Paraneoplastic syndromes (earlier escalation)
Options:
| Treatment | Dose | Schedule | Mechanism |
|---|---|---|---|
| Rituximab (Most commonly used) | 375 mg/m² IV weekly × 4 doses OR 1g IV × 2 doses (2 weeks apart) | Single course; repeat if relapse | Anti-CD20 monoclonal antibody → B-cell depletion; used in 46% of AE cases (NMDAR 57%, LGI1 43%, CASPR2 44%) [25,31] |
| Cyclophosphamide | 750 mg/m² IV monthly | 6 months | Alkylating agent → B-cell and T-cell suppression; combination with rituximab effective in refractory status epilepticus [26] |
Choice of Agent:
- Rituximab preferred for cell surface antibody AE (targets B-cell antibody production); real-world data shows widespread adoption with good outcomes [31]
- Cyclophosphamide considered for paraneoplastic (T-cell-mediated) or rituximab-refractory cases
- Some protocols use combination rituximab + cyclophosphamide for severe disease; combination shows marked improvement in refractory status epilepticus [26]
- Pediatric data: Rituximab safe and effective in children; 62.5% achieve mRS ≤2 [25]
Monitoring:
- Rituximab: Pre-treatment hepatitis B/C serology, immunoglobulins, CD19/CD20 counts; post-treatment CD19+ B-cell depletion to 0 indicates therapeutic efficacy [27]
- Cyclophosphamide: FBC (neutropenia risk), bladder toxicity (hemorrhagic cystitis—give mesna prophylaxis), gonadal toxicity
Tumor Management
Principle: Tumor removal is ESSENTIAL for paraneoplastic syndromes and improves outcomes in anti-NMDAR encephalitis. [7]
Ovarian Teratoma (Anti-NMDAR):
- Surgical removal (unilateral oophorectomy or cystectomy)
- Coordinate with gynecology
- Neurological improvement often begins within weeks post-surgery
- Reduces relapse risk
Small Cell Lung Cancer (Anti-GABAB, AMPAR, Hu, CV2):
- Oncology referral for staging and chemotherapy/radiotherapy
- Neurological outcome depends on tumor control
- Poor prognosis if disseminated disease
Thymoma (Anti-CASPR2, amphiphysin):
- Surgical resection (if resectable)
- Radiotherapy if unresectable
Timing:
- Perform tumor removal as soon as safely feasible (even during acute illness; coordinate with neurology/ICU)
Symptomatic Management
Seizure Control
Anti-Seizure Medications (ASMs):
- Seizures often refractory to ASMs alone; improve with immunotherapy
- First-line ASMs: Levetiracetam (500-1500mg BD), lacosamide (100-200mg BD), clobazam (10-20mg BD)
- Avoid enzyme-inducing ASMs (phenytoin, carbamazepine—interaction risk)
- Benzodiazepines for acute seizures or status epilepticus
Refractory Status Epilepticus:
- ICU sedation (midazolam, propofol, thiopentone infusions)
- EEG monitoring (burst suppression target)
- Escalate immunotherapy urgently
Duration:
- Continue ASMs during acute phase
- Taper slowly after 6-12 months seizure-free (recurrence risk low if immunotherapy successful)
Psychiatric and Behavioral Symptoms
Agitation/Psychosis:
- Benzodiazepines (lorazepam 0.5-2mg PRN) preferred over antipsychotics
- Antipsychotics (if necessary): Use atypical agents (quetiapine 25-100mg, olanzapine 2.5-10mg) at LOW doses
- Avoid haloperidol (risk of neuroleptic malignant syndrome-like reaction)
- Environmental: Low-stimulation environment, familiar objects, family presence
Catatonia:
- Benzodiazepines (lorazepam 1-2mg TDS)
- Consider electroconvulsive therapy (ECT) if refractory (case reports suggest benefit)
Sleep Disturbance:
- Melatonin, trazodone
- Address ICU delirium if applicable
Autonomic Instability
Cardiovascular:
- ICU monitoring (continuous ECG, arterial line)
- Treat tachycardia: Beta-blockers (labetalol) if sustained
- Treat bradycardia: Atropine, pacing if severe
- Avoid excessive fluid shifts
Respiratory:
- Monitor for central hypoventilation (respiratory rate, ABG, capnography)
- Mechanical ventilation if required (often prolonged in severe anti-NMDAR)
Hyperthermia:
- Cooling measures (cooling blankets, ice packs)
- Exclude infection (fever workup)
- Exclude neuroleptic malignant syndrome
Venous Thromboembolism Prophylaxis
- High risk (prolonged immobility, ICU admission)
- Pharmacological prophylaxis: LMWH (enoxaparin 40mg SC daily) unless contraindicated
- Mechanical prophylaxis: Intermittent pneumatic compression devices
Nutrition
- Early enteral feeding (NG tube if swallowing unsafe)
- Parenteral nutrition if enteral not tolerated
Rehabilitation
Multidisciplinary Team:
- Physiotherapy (mobility, strength)
- Occupational therapy (ADLs, cognitive strategies)
- Speech and language therapy (swallowing, communication, cognitive-communication)
- Neuropsychology (cognitive rehabilitation, psychiatric support)
Setting:
- Inpatient neurorehabilitation once medically stable
- Community rehabilitation for ongoing needs
Duration:
- Recovery protracted (6-24 months); ongoing therapy essential
Neuropsychological Rehabilitation
Cognitive Domains Affected:
- Memory: Anterograde amnesia (new learning impairment) most persistent deficit; retrograde memory relatively preserved [23]
- Executive Function: Planning, organization, cognitive flexibility, problem-solving deficits
- Processing Speed: Slowed information processing common across all antibody types [24]
- Attention: Sustained attention and working memory impairments
- Language: Usually preserved except in severe encephalopathy; pragmatic language difficulties may emerge
Evidence Base for Cognitive Outcomes: [23,24]
- Memory impairment persists in 30-40% despite immunotherapy
- Anti-LGI1 patients: 75% have short-term memory loss; residual deficits common despite excellent functional recovery
- Anti-NMDAR patients: Executive function and processing speed improve more than memory
- Hippocampal atrophy on follow-up MRI correlates with persistent memory impairment
- Earlier treatment (within 4 weeks) associated with better cognitive outcomes
Rehabilitation Strategies:
-
Memory Rehabilitation:
- External aids: Smartphones, calendars, alarms, written schedules, memory notebooks
- Internal strategies: Spaced retrieval, elaborative encoding, errorless learning
- Environmental modifications: Reduce distractions, consistent routines, visual cues
-
Executive Function Training:
- Goal Management Training (GMT): Breaking tasks into steps, planning strategies
- Time Pressure Management (TPM): Structured time allocation
- Problem-solving training: Real-world scenario practice
-
Attention and Processing Speed:
- Computer-based cognitive training programs
- Graded attention exercises (simple to complex)
- Energy conservation techniques
-
Cognitive-Communication Therapy:
- Social communication skills
- Conversational turn-taking
- Topic maintenance and organization
-
Compensatory Strategies:
- Workplace accommodations: Quiet environment, extra time for tasks, reduced hours
- Educational accommodations: Extended deadlines, note-takers, separate examination room
- Home modifications: Labels, checklists, simplified routines
Rehabilitation Timeline:
- Acute phase (0-4 weeks): Basic cognitive stimulation, orientation, family education
- Early rehabilitation (1-6 months): Intensive outpatient cognitive rehabilitation 2-3 sessions/week
- Late rehabilitation (6-24 months): Community-based therapy, gradual return to work/school with support
- Long-term (24+ months): Neuropsychological reassessment; ongoing targeted interventions for persistent deficits
Monitoring and Outcomes:
- Formal neuropsychological assessment at 6, 12, 24 months post-onset
- Standardized tools: Montreal Cognitive Assessment (MoCA), Repeatable Battery for Assessment of Neuropsychological Status (RBANS), domain-specific tests
- Functional outcome measures: Modified Rankin Scale, Barthel Index, quality of life scales
- Return to work/education rates: 70-80% return, though 30-40% require accommodations
Prognostic Factors for Cognitive Recovery: [7,23,24,32]
- Favorable: Early immunotherapy (within 4 weeks), younger age, absence of hippocampal atrophy, anti-LGI1 antibody (functional recovery), tumor removed
- Unfavorable: Delayed treatment, prolonged ICU stay, refractory status epilepticus, hippocampal atrophy on follow-up imaging, multiple relapses
- Special consideration: Anti-LGI1 patients with faciobrachial dystonic seizures—early immunotherapy prevents progression to memory impairment [32]
Long-Term Management and Relapse Prevention
Maintenance Immunotherapy:
- Consider in patients at high relapse risk (anti-NMDAR without teratoma, anti-AMPAR)
- Options:
- Rituximab 1g IV every 6 months × 18-24 months [7]
- Mycophenolate mofetil 500-1000mg BD (alternative)
- Azathioprine 2-3 mg/kg/day (alternative)
Tumor Surveillance:
- Anti-NMDAR: Repeat pelvic imaging 6-monthly for 2 years, then annually for 5 years (delayed teratoma detection)
- Anti-GABAB, AMPAR: Oncology follow-up per cancer protocol
Monitoring for Relapse:
- Relapse rate: 20-24% (anti-NMDAR), higher for AMPAR [7,10]
- Symptoms: Recurrence of cognitive, psychiatric, or seizure symptoms (often milder than initial presentation)
- Management: Reinitiate immunotherapy; consider maintenance therapy
Anti-Seizure Medication:
- Taper slowly after 12 months seizure-free
- Recurrence risk low with successful immunotherapy
Neuropsychological Follow-Up:
- Assess cognitive recovery at 6, 12, 24 months
- Memory, executive function, processing speed most affected
- Cognitive rehabilitation, occupational adjustments
Driving:
- DVLA/local regulations: Typically 12-month seizure-free period required (UK)
Management Algorithm Summary
Suspected Autoimmune Encephalitis
↓
[Clinical Criteria Met] → Send serum + CSF antibodies, MRI, EEG, tumor screen
↓
START FIRST-LINE IMMUNOTHERAPY (do not wait for results)
- IV methylprednisolone 1g × 5 days
- IVIG 0.4g/kg × 5 days
- Plasma exchange 5-7 sessions
↓
Assess response at 2-4 weeks
↓
↙ ↘
[Improved] [No/Minimal Improvement]
↓ ↓
Continue oral SECOND-LINE IMMUNOTHERAPY
taper - Rituximab 375mg/m² weekly × 4
Monitor OR 1g × 2 (2 weeks apart)
relapse - Cyclophosphamide 750mg/m² monthly × 6
↓
TUMOR REMOVAL (if identified)
↓
Long-term: Maintenance therapy if high relapse risk
9. Complications
Acute Complications (During Illness)
| Complication | Incidence | Clinical Features | Management |
|---|---|---|---|
| Refractory status epilepticus | 30-40% (anti-NMDAR) [11] | Continuous or recurrent seizures without recovery; may be non-convulsive | Aggressive ASMs, ICU sedation (midazolam/propofol), urgent immunotherapy escalation, EEG monitoring |
| Respiratory failure | 20% (anti-NMDAR) [7] | Central hypoventilation, apnea | Mechanical ventilation (may be prolonged—weeks to months); wean gradually as immunotherapy takes effect |
| Autonomic crisis | 60% (anti-NMDAR) [4] | Severe tachycardia/bradycardia, blood pressure lability, hyperthermia | ICU monitoring, cardiovascular support, temperature control |
| Cardiac arrhythmias | 10-20% | Bradycardia, asystole, torsades de pointes | Continuous ECG monitoring, pacing if severe, correct electrolytes |
| Venous thromboembolism | 5-10% | DVT, PE | Prophylactic LMWH, early mobilization; treatment dose anticoagulation if occurs |
| Aspiration pneumonia | 10-15% | Dysphagia, impaired consciousness | Nil by mouth, NG feeding, antibiotics, chest physiotherapy |
| Sepsis | Variable (ICU-associated) | Hospital-acquired infections (VAP, line sepsis, UTI) | Infection prevention, early recognition, antibiotics |
| Pressure ulcers | Variable (prolonged immobility) | Sacral, heel ulcers | Pressure relief, turning, specialist mattress |
| Neuroleptic malignant syndrome-like reaction | Rare | After antipsychotic use; rigidity, hyperthermia, CK elevation | Discontinue antipsychotic, supportive care, consider dantrolene/bromocriptine |
Medium-Term Complications (Weeks to Months)
| Complication | Management |
|---|---|
| Critical illness neuropathy/myopathy | Physiotherapy, rehabilitation |
| ICU-acquired weakness | Gradual mobilization, nutrition optimization |
| Post-traumatic stress disorder | Psychological support, CBT |
Long-Term Complications (Months to Years)
| Complication | Incidence | Impact | Management |
|---|---|---|---|
| Persistent cognitive impairment | 30-40% [7] | Memory (most common), executive function, processing speed | Neuropsychological assessment, cognitive rehabilitation, occupational therapy |
| Psychiatric sequelae | 20-30% | Depression, anxiety, personality change | Psychiatry follow-up, antidepressants, psychological therapies |
| Epilepsy | 10-15% | Ongoing seizures despite treatment | Continue ASMs, epilepsy clinic follow-up |
| Movement disorder | Rare (usually resolves) | Persistent dyskinesia or dystonia | Neurology follow-up; may respond to further immunotherapy |
| Relapse of autoimmune encephalitis | 20-24% (anti-NMDAR) [10] | Recurrence of symptoms (often milder) | Reinitiate immunotherapy; maintenance therapy |
| Hippocampal atrophy | Common on follow-up MRI | Correlates with memory impairment | Monitor; may be irreversible |
| Functional disability | 20-30% | Inability to return to prior employment/education | Vocational rehabilitation, disability support |
| Death | 4-7% (with treatment) [7] | Cardiorespiratory failure, status epilepticus, complications of critical illness | Risk increased by delayed treatment, ICU complications |
Relapse
Epidemiology:
- Overall relapse rate: 20-24% [10]
- Higher risk: Anti-AMPAR (50-60%), anti-LGI1 (20-30%)
- Lower risk: Anti-NMDAR with teratoma removed (12%)
Timing:
- Median time to relapse: 2 years (range: months to greater than 5 years)
Clinical Features:
- Often milder than initial presentation
- Similar symptom profile to index episode
Prevention:
- Tumor removal (if applicable)
- Maintenance immunotherapy (rituximab every 6 months × 18-24 months)
Management:
- Reinitiate first-line immunotherapy
- Consider second-line agents if inadequate response
- Long-term maintenance therapy
10. Prognosis
Outcomes with Treatment
Modified Rankin Scale (mRS) at 24 Months (Anti-NMDAR): [7]
- mRS 0-2 (no/minor symptoms, independent): 80%
- mRS 0 (fully recovered): 47%
- mRS 1 (minor symptoms, no disability): 20%
- mRS 2 (slight disability, independent): 13%
- mRS 3-5 (moderate to severe disability): 13%
- Death: 7%
Functional Outcomes:
- 70-80% return to school/work (may require accommodations)
- 50% completely asymptomatic at 2 years
- 30% mild residual cognitive or psychiatric symptoms
- 20% moderate to severe disability
Anti-LGI1 Encephalitis:
- Excellent prognosis: 80-90% mRS 0-2 [13]
- Early treatment of FBDS prevents cognitive impairment
- Memory impairment may persist despite treatment
Paraneoplastic (Intracellular Antibodies):
- Poor prognosis: Limited response to immunotherapy
- Outcome depends on tumor control
- Many patients have severe, irreversible neurological disability
Factors Influencing Prognosis
Good Prognostic Factors:
- Early treatment initiation (within 4 weeks of symptom onset) [7]
- Tumor identified and removed [7]
- Younger age
- Cell surface antibodies (NMDAR, LGI1, CASPR2, GABAB, AMPAR) vs. intracellular
- Less severe presentation (no ICU admission)
- Good response to first-line immunotherapy
Poor Prognostic Factors:
- Delayed treatment (greater than 4 weeks) [7]
- ICU admission required (marker of severity)
- Prolonged ventilation
- Refractory status epilepticus
- Intracellular antibodies (Hu, Yo, Ri—T-cell-mediated, irreversible neuronal death)
- No tumor found when expected (undiagnosed malignancy)
- Multiple relapses
Time Course of Recovery
Acute Phase (Weeks 0-4):
- Immunotherapy initiated
- Stabilization of symptoms (prevent progression)
- Autonomic stabilization
Early Recovery (Months 1-6):
- Gradual awakening (if comatose)
- Resolution of movement disorders
- Reduction in seizure frequency
- Cognitive improvement begins (slow)
Late Recovery (Months 6-24):
- Continued cognitive improvement (memory, executive function)
- Psychiatric symptom resolution
- Functional independence regained
- Return to education/employment (with support)
Protracted Recovery:
- Cognitive recovery may continue for 18-24 months [7]
- Memory impairment most persistent symptom
- Neuropsychological rehabilitation essential
Mortality
Chinese Cohort Data (12-month outcomes): [30]
- Favorable outcome (mRS ≤2): 92.7%
- Improvement: 94.1%
- Relapse: 17.3%
- Death: 2.3%
Overall Mortality: 4-7% (with treatment) [7]
Causes of Death:
- Cardiorespiratory failure (autonomic instability, central hypoventilation)
- Refractory status epilepticus
- Complications of critical illness (sepsis, PE)
- Underlying malignancy (paraneoplastic cases)
Without Treatment: Mortality approaches 25%
Relapse and Long-Term Monitoring
Relapse Rate: 20-24% [10]
Predictors of Relapse:
- No tumor identified (especially anti-NMDAR)
- Incomplete initial treatment response
- Antibody persistence at high titre (controversial)
Prevention:
- Tumor removal
- Maintenance immunotherapy (rituximab, mycophenolate, azathioprine)
Long-Term Monitoring:
- Neurology clinic: 3-6 monthly for 2 years, then annually
- Neuropsychology: Assess cognitive recovery
- Tumor surveillance (as per antibody-specific protocol)
- Monitor for relapse symptoms
11. Evidence and Guidelines
Key Guidelines
-
Graus F, Titulaer MJ, Balu R, et al. A clinical approach to diagnosis of autoimmune encephalitis. Lancet Neurol. 2016;15(4):391-404. PMID: 26906964
- Landmark consensus criteria for diagnosis of autoimmune encephalitis
- Defines "possible" and "definite" AE
- Guides empirical treatment decisions
-
Graus F, Vogrig A, Muñiz-Castrillo S, et al. Updated Diagnostic Criteria for Paraneoplastic Neurologic Syndromes. Neurol Neuroimmunol Neuroinflamm. 2021;8(4):e1014. PMID: 34006622
- Updates criteria for paraneoplastic syndromes
- Separates antibody-negative from antibody-positive syndromes
-
American Academy of Neurology (AAN) Practice Guidelines (Various)
- Recommendations for encephalitis workup and treatment
- Empirical immunotherapy supported
Landmark Studies
Pathophysiology
-
Dalmau J, Gleichman AJ, Hughes EG, et al. Anti-NMDA-receptor encephalitis: case series and analysis of the effects of antibodies. Lancet Neurol. 2008;7(12):1091-1098. PMID: 18851928
- Original description of anti-NMDAR encephalitis
- Demonstrated antibody-mediated NMDA receptor internalization (reversible mechanism)
- Established clinical phenotype and treatment paradigm
-
Dalmau J, Armangué T, Planagumà J, et al. An update on anti-NMDA receptor encephalitis for neurologists and psychiatrists: mechanisms and models. Lancet Neurol. 2019;18(11):1045-1057. PMID: 31326280
- Comprehensive review of pathophysiology, animal models, clinical features
- Explains reversible synaptic dysfunction (receptor internalization)
Clinical Features and Diagnosis
-
Dalmau J, Graus F. Diagnostic criteria for autoimmune encephalitis: utility and pitfalls for antibody-negative disease. Lancet Neurol. 2023;22(6):529-540. PMID: 37210100
- Addresses antibody-negative autoimmune encephalitis (10-15% of cases)
- Emphasizes clinical diagnosis and empirical treatment
-
Titulaer MJ, McCracken L, Gabilondo I, et al. Treatment and prognostic factors for long-term outcome in patients with anti-NMDA receptor encephalitis: an observational cohort study. Lancet Neurol. 2013;12(2):157-165. PMID: 23290630
- Key prognostic study: 577 patients, 24-month follow-up
- 80% mRS 0-2 at 24 months; 47% fully recovered
- Early treatment and tumor removal improve outcomes
- Relapse rate: 12% (with teratoma removal) to 24% (without)
-
Irani SR, Michell AW, Lang B, et al. Faciobrachial dystonic seizures precede Lgi1 antibody limbic encephalitis. Ann Neurol. 2011;69(5):892-900. PMID: 21416487
- Described faciobrachial dystonic seizures (FBDS) as pathognomonic for anti-LGI1
- FBDS herald cognitive impairment; early immunotherapy prevents progression
Misdiagnosis
- Flanagan EP, Geschwind MD, Lopez-Chiriboga AS, et al. Autoimmune Encephalitis Misdiagnosis in Adults. JAMA Neurol. 2023;80(1):30-39. PMID: 36441519
- Critical study: 13% of suspected AE cases misdiagnosed
- Common misdiagnoses: Primary psychiatric disorders, neurodegenerative diseases
- Highlights need for rigorous diagnostic criteria
Management
- Uy CE, Binks S, Irani SR. Autoimmune encephalitis: clinical spectrum and management. Pract Neurol. 2021;21(5):412-423. PMID: 34108243
- Practical guide to diagnosis and treatment
- Management algorithms for first-line and second-line immunotherapy
Neuroimaging
-
Kelly C, Grant R, Murchison A, et al. Magnetic Resonance Imaging Characteristics of LGI1-Antibody and CASPR2-Antibody Encephalitis. JAMA Neurol. 2024;81(2):151-160. PMID: 38497971
- Detailed MRI patterns in anti-LGI1 (mesial temporal + basal ganglia) and anti-CASPR2
- 60-70% MRI abnormalities in LGI1
-
Mueller S, Elben B, Day GS, et al. Review and meta-analysis of neuropsychological findings in autoimmune limbic encephalitis with autoantibodies against LGI1, CASPR2, and GAD65 and their response to immunotherapy. Clin Neurol Neurosurg. 2023;225:107594. PMID: 36549220
- Neuropsychological outcomes post-immunotherapy
- Memory impairment most persistent deficit
EEG
- Freund B, Ritzl EK. A review of EEG in anti-NMDA receptor encephalitis. J Neuroimmunol. 2019;332:64-68. PMID: 30959339
- Review of EEG findings, including extreme delta brush pattern (30% of anti-NMDAR)
Critical Care
- Lin YT, Lin CI. Neurocritical care for Anti-NMDA receptor encephalitis. Biomed J. 2020;43(3):251-259. PMID: 32330674
- ICU management of severe anti-NMDAR encephalitis
- Autonomic instability, ventilation, sedation strategies
Antibody Testing
- Gresa-Arribas N, Titulaer MJ, Torrents A, et al. Antibody titres at diagnosis and during follow-up of anti-NMDA receptor encephalitis: a retrospective study. Lancet Neurol. 2014;13(2):167-177. PMID: 24360484
- Antibody titres correlate poorly with disease severity
- Not useful for monitoring treatment response
- CSF antibody persistence longer than serum
Post-Infectious AE
- Armangue T, Leypoldt F, Málaga I, et al. Herpes simplex virus encephalitis is a trigger of brain autoimmunity. Ann Neurol. 2014;75(2):317-323. PMID: 24318406
- HSV encephalitis can trigger anti-NMDAR encephalitis weeks to months later
- Choreoathetosis post-HSV = search for NMDAR antibodies
Epidemiology
- Dubey D, Pittock SJ, Kelly CR, et al. Autoimmune encephalitis epidemiology and a comparison to infectious encephalitis. Ann Neurol. 2018;83(1):166-177. PMID: 29291557
- Population-based incidence study
- AE incidence comparable to infectious encephalitis (5-8 per million per year)
GABAB Encephalitis
- Lancaster E, Lai M, Peng X, et al. Antibodies to the GABA(B) receptor in limbic encephalitis with seizures: case series and characterisation of the antigen. Lancet Neurol. 2010;9(1):67-76. PMID: 19962348
- Original description of anti-GABAB encephalitis
- High SCLC association (50-60%)
Pediatric AE
- Florance NR, Davis RL, Lam C, et al. Anti-N-methyl-D-aspartate receptor (NMDAR) encephalitis in children and adolescents. Ann Neurol. 2009;66(1):11-18. PMID: 19670433
- Pediatric anti-NMDAR presentation differs (less psychiatric, more seizures/movement disorder)
Refractory Status Epilepticus
- Gaspard N, Foreman BP, Alvarez V, et al. New-onset refractory status epilepticus: Etiology, clinical features, and outcome. Neurology. 2015;85(18):1604-1613. PMID: 26296517
- NORSE often autoimmune etiology (anti-NMDAR, anti-GABAA)
- Early immunotherapy essential
Long-Term Outcomes
- de Bruijn MAAM, Aarsen FK, van Oosterhout MP, et al. Long-term neuropsychological outcome following pediatric anti-NMDAR encephalitis. Neurology. 2018;90(22):e1997-e2005. PMID: 29743207
- Long-term cognitive outcomes in children
- Memory and executive function most affected
Imaging Advanced
- Bacchi S, Franke K, Wewegama D, et al. Magnetic resonance imaging and positron emission tomography in anti-NMDA receptor encephalitis: A systematic review. J Clin Neurosci. 2018;52:54-59. PMID: 29627240
- FDG-PET more sensitive than MRI (abnormal in 80-90% vs. 50-60%)
- Frontotemporal hypometabolism in anti-NMDAR
12. Patient Explanation
What is Autoimmune Encephalitis?
Autoimmune encephalitis is when your body's immune system—which normally fights infections—makes a mistake and attacks healthy parts of your brain. This happens because the immune system produces antibodies (proteins) that target proteins on brain cells. These antibodies interfere with the normal signaling between brain cells, causing symptoms like confusion, memory problems, seizures, unusual movements, and changes in behavior or mood.
The most common type is called anti-NMDA receptor encephalitis, which often affects young women. It can be associated with a type of ovarian tumor called a teratoma, but sometimes there is no tumor.
How Did I Get This?
In most cases, we don't know what triggers the immune system to attack the brain. Possible triggers include:
- A viral infection (like a cold or flu)
- A tumor that contains brain-like tissue (such as an ovarian teratoma)
- Sometimes, there is no clear trigger
It is not caused by anything you did wrong, and it is not contagious.
What Are the Symptoms?
Symptoms develop over days to weeks and can include:
- Confusion, memory problems
- Hallucinations (seeing or hearing things that aren't there), paranoia, or unusual behavior
- Seizures (fits)
- Unusual movements (jerking, stiffness, repetitive movements)
- Changes in mood (anxiety, depression, agitation)
- Decreased consciousness or sleepiness
- Fever or flu-like symptoms at the start
- In severe cases: difficulty breathing, abnormal heart rate, or coma
How is it Diagnosed?
Your doctor will perform several tests:
- Blood and spinal fluid tests: To look for specific antibodies
- MRI brain scan: To look for inflammation in the brain
- EEG (brainwave test): To check for seizure activity
- Scans to look for tumors: Such as ultrasound or CT scans of the pelvis, chest, or abdomen
How is it Treated?
Treatment aims to calm down the immune system and remove the antibodies attacking your brain:
-
First-line treatments:
- High-dose steroids (intravenous or oral)
- Intravenous immunoglobulin (IVIG)—antibodies from donated blood
- Plasma exchange (a procedure to filter antibodies from your blood)
-
Second-line treatments (if first-line doesn't work):
- Rituximab (medicine that removes certain immune cells)
- Cyclophosphamide (chemotherapy that suppresses the immune system)
-
Tumor removal: If a tumor is found (such as an ovarian teratoma), removing it is essential for recovery.
-
Supportive care:
- Medications to control seizures
- Medications to manage agitation or behavioral symptoms
- Intensive care if breathing or heart function is affected
- Physiotherapy, occupational therapy, and speech therapy during recovery
What Can I Expect?
- Early treatment is critical: The sooner treatment starts, the better the recovery.
- Recovery takes time: Most people improve over weeks to months, but full recovery can take 1-2 years. Cognitive (memory, thinking) and psychiatric (mood, behavior) symptoms are often the last to improve.
- Prognosis:
-
Wang L, Li J, Wang Y, et al. Immunotherapy for anti-NMDA receptor encephalitis: Experience from a single center in Taiwan. J Neuroimmunol. 2018;324:10-16. PMID: 30449706
- Single-center cohort of 24 anti-NMDAR patients
- 83% achieved good outcome (mRS ≤2) at 6 months; 62.5% completely recovered
- Relapse rate 17.7%; tumor association 12.5%
- Emphasizes importance of early immunotherapy initiation
-
Mueller S, Elben B, Day GS, et al. Review and meta-analysis of neuropsychological findings in autoimmune limbic encephalitis with autoantibodies against LGI1, CASPR2, and GAD65 and their response to immunotherapy. Clin Neurol Neurosurg. 2023;225:107594. PMID: 36549220
- Meta-analysis of neuropsychological outcomes post-immunotherapy
- Memory impairment most persistent deficit across all antibody types (LGI1, CASPR2, GAD65)
- Residual cognitive deficits common despite favorable functional recovery
- Processing speed and executive function affected; language relatively preserved
-
Huang W, Wang L, Liao X, et al. Efficacy and Safety of Rituximab in Chinese Children With Refractory Anti-NMDAR Encephalitis. Front Neurol. 2020;11:606923. PMID: 33381080
- Rituximab efficacy in 8 pediatric refractory anti-NMDAR cases
- 62.5% good outcome (mRS ≤2); 50% complete recovery (mRS 0) at last follow-up
- Significant improvement in mRS from baseline (P less than 0.05)
- Demonstrates safety and efficacy in children
-
Ishimaru T, Yoshikawa H, Kamakura T, et al. Refractory status epilepticus caused by anti-NMDA receptor encephalitis that markedly improved following combination therapy with rituximab and cyclophosphamide. Rinsho Shinkeigaku. 2015;55(3):168-172. PMID: 25743014
- Case report: drug-resistant status epilepticus resolved with rituximab + cyclophosphamide combination
- Highlights early second-line therapy in severe refractory cases
- Combination immunosuppression effective when monotherapy fails
-
Gao Z, Liang T, Wang Y, et al. Pharmaceutical care of rituximab in the treatment of children with refractory anti-NMDAR encephalitis: A case report. Medicine (Baltimore). 2023;102(6):e32895. PMID: 36749271
- Detailed case of rituximab pharmacotherapy in pediatric refractory anti-NMDAR encephalitis
- CD19+ B-cell depletion to 0 as therapeutic endpoint following rituximab
- Patient showed gradual improvement in consciousness, cognition, mental status, and movement disorder
- Highlights importance of monitoring B-cell counts post-rituximab
-
Sun Y, Gao L, Jia Y, et al. Clinical Features and Therapeutic Effects of Anti-leucine-rich Glioma Inactivated 1 Encephalitis: A Systematic Review. Front Neurol. 2022;13:819087. PMID: 35095736
- Systematic review of anti-LGI1 encephalitis clinical features
- Short-term memory loss (75.22%), FBDS (52.53%), other seizures (68.48%), psychiatric symptoms (57.67%)
- Hyponatremia (54.90%), sleep disturbances (34.30%)
- Comprehensive synthesis of clinical phenotype
-
Casault C, Alikhani K, Pillay N, Leung AA. Electrolyte Imbalance in Anti-LGI1 Encephalitis: It Is Not All in Your Head. AACE Clin Case Rep. 2023;9(4):137-140. PMID: 37591767
- Case demonstrating SIADH as presenting feature of anti-LGI1 encephalitis
- Laboratory findings: Low serum osmolality, low BUN, low uric acid, high urinary sodium, inappropriately high urine osmolality
- Hyponatremia can precede neurological symptoms—important diagnostic clue
-
Zhang Y, Liu G, Jiang M, et al. Anti-NMDAR encephalitis: A single-center, longitudinal study in China. Neurol Neuroimmunol Neuroinflamm. 2019;6(6):e633. PMID: 31619447
- Large Chinese cohort: 220 anti-NMDAR patients
- 94.1% improved during first 12 months; 92.7% favorable outcome (mRS ≤2) at 12-month follow-up
- Relapse rate 17.3%; mortality 2.3%
- Excellent outcomes with immunotherapy in Asian population
-
Abboud H, Probasco JC, Irani SR, et al. Rituximab Treatment and Long-term Outcome of Patients With Autoimmune Encephalitis: Real-world Evidence From the GENERATE Registry. Neurology. 2021;97(12):e1209-e1220. PMID: 34599001
- Real-world registry data: 358 autoimmune encephalitis patients
- Rituximab used in 46% overall (NMDAR-AE 57%, LGI1-AE 43%, CASPR2-AE 44%, GAD65 37%)
- Rituximab initiated median 54 days from disease onset in NMDAR-AE, 155 days in LGI1-AE
- Demonstrates widespread adoption and real-world effectiveness across subtypes
-
Irani SR, Stagg CJ, Schott JM, et al. Faciobrachial dystonic seizures: the influence of immunotherapy on seizure control and prevention of cognitive impairment in a broadening phenotype. Brain. 2013;136(Pt 10):3151-3162. PMID: 24014518
- Landmark study on FBDS and cognitive outcomes in anti-LGI1 encephalitis
- Early immunotherapy for FBDS prevents progression to limbic encephalitis and cognitive impairment
- Delayed treatment associated with irreversible memory deficits despite seizure control
- Establishes FBDS as critical window for intervention—treatment urgency
- About 80% of people improve with treatment
- About 50% recover completely
- About 30% have mild ongoing symptoms (such as memory problems)
- About 20% have a relapse (symptoms return), which is treated again
- With treatment, the death rate is about 5-7%
What Should I Watch For?
After discharge, contact your doctor if you notice:
- Return of confusion, seizures, or unusual movements
- New or worsening psychiatric symptoms (hallucinations, agitation)
- Severe headache or fever (could indicate infection)
- Symptoms of relapse (usually occur months to years later)
Can I Prevent Relapse?
- Tumor removal (if applicable) greatly reduces relapse risk
- Maintenance immunotherapy: Your doctor may recommend ongoing treatment (such as rituximab every 6 months) to prevent relapse
- Regular follow-up: Attend neurology, oncology (if tumor), and neuropsychology appointments
- Tumor surveillance: Repeat scans to check for tumor recurrence or late-appearing tumors
Driving and Work
- Driving: You will need to be seizure-free for a period (usually 12 months in the UK) before you can drive again. Check with your local licensing authority.
- Work/School: Many people return to work or school, though you may need accommodations (reduced hours, cognitive support) initially.
Support and Resources
- Neurology team: Regular follow-up to monitor recovery and adjust treatment
- Neuropsychology: Cognitive rehabilitation and psychological support
- Rehabilitation services: Physiotherapy, occupational therapy, speech therapy
- Support groups: Connect with others who have experienced autoimmune encephalitis (e.g., Encephalitis Society, AE Alliance)
Key Message
Autoimmune encephalitis is serious but treatable. With early treatment, most people recover well, though it takes time and patience. You are not alone—your medical team, family, and support networks are here to help you through your recovery.
13. Examination Focus
Viva Points (Concise Answer)
"Autoimmune encephalitis is antibody-mediated brain inflammation. The most common subtype is anti-NMDA receptor encephalitis, affecting young women (median age 21), often associated with ovarian teratoma (50% of women). Presentation follows a characteristic progression: psychiatric symptoms (psychosis, behavioral change), seizures (90%), movement disorders (orofacial dyskinesias, choreoathetosis), decreased consciousness, and autonomic instability (hypoventilation, tachycardia). Diagnosis relies on clinical criteria: subacute onset, cognitive/psychiatric symptoms, plus CSF pleocytosis, MRI mesial temporal hyperintensity (60%), and antibody detection (serum and CSF). Treat empirically with first-line immunotherapy (IV methylprednisolone, IVIG, or plasma exchange) without waiting for antibody results. Second-line agents include rituximab and cyclophosphamide. Tumor removal is essential if present. Prognosis: 80% improve, 47% fully recover; relapse rate 20-24%. Early treatment (within 4 weeks) improves outcomes."
High-Yield Examination Facts
Anti-NMDAR Encephalitis:
- Young women (median 21 years), F:M = 4:1
- Ovarian teratoma in 50% of women greater than 18 years
- Progression: Psychiatric → seizures → movement disorder → autonomic instability → coma
- EEG: Extreme delta brush (30%)
- Prognosis: 80% mRS 0-2 at 24 months
Anti-LGI1 Encephalitis:
- Older men (median 60 years), M:F = 2:1
- Faciobrachial dystonic seizures (FBDS): Pathognomonic—brief (less than 3 sec), frequent (10-50/day), face + arm dystonic posturing
- Hyponatraemia (SIADH) in 60%
- Excellent response to immunotherapy (best prognosis among AE subtypes)
- MRI: Mesial temporal + basal ganglia (putamen) hyperintensity
Anti-CASPR2 Encephalitis:
- Morvan syndrome: Neuromyotonia (peripheral nerve hyperexcitability) + insomnia + autonomic dysfunction + encephalopathy
- Thymoma association (20-30%)—CT chest mandatory
Anti-GABAB Encephalitis:
- Prominent seizures + limbic encephalitis
- SCLC association (50-60%)—urgent CT chest
Diagnostic Criteria:
- Subacute onset (less than 3 months) + cognitive/psychiatric symptoms
- Plus ONE: Focal CNS signs, seizures, CSF pleocytosis, MRI limbic encephalitis
- Exclusion of mimics (HSV encephalitis, primary psychiatric, toxic-metabolic)
Treatment:
- Start empirically—do not wait for antibody results
- First-line: IV methylprednisolone 1g × 5 days, IVIG 0.4g/kg × 5 days, or PLEX
- Second-line: Rituximab (375mg/m² weekly × 4 OR 1g × 2) or cyclophosphamide
- Tumor removal essential
- Early treatment (less than 4 weeks) improves outcomes
Prognostic Factors:
- Good: Early treatment, tumor removed, cell surface antibody, younger age
- Poor: Delayed treatment, ICU admission, intracellular antibody, no tumor found when expected
Relapse:
- 20-24% overall
- Prevention: Tumor removal, maintenance rituximab (every 6 months × 18-24 months)
Common Viva Questions and Model Answers
Q: How do you differentiate autoimmune encephalitis from viral (HSV) encephalitis?
A: "Both present with subacute encephalopathy, but HSV has acute onset (hours to days), high fever, and hemorrhagic necrosis on MRI. CSF shows RBCs and HSV PCR is positive. Autoimmune encephalitis has more subacute onset (days to weeks), prominent psychiatric and movement disorder features (especially anti-NMDAR), and CSF shows lymphocytic pleocytosis with oligoclonal bands. Importantly, HSV encephalitis can trigger post-infectious anti-NMDAR encephalitis weeks later (choreoathetosis after treated HSV)."
Q: What is the mechanism of reversibility in anti-NMDAR encephalitis?
A: "Antibodies bind to the GluN1 subunit of the NMDA receptor, causing receptor cross-linking and internalization via endocytosis. This reduces receptor density at the synapse, leading to NMDA hypofunction. Critically, this is a functional, reversible process—there is no direct cytotoxic neuronal death. With immunotherapy, antibodies are removed, receptors return to the surface, and synaptic function recovers. This contrasts with paraneoplastic intracellular antibody syndromes (e.g., anti-Hu), where cytotoxic T-cells cause irreversible neuronal death."
Q: Why do you test both serum and CSF for antibodies?
A: "CSF is more sensitive than serum for anti-NMDAR antibodies because antibodies are often produced intrathecally (within the CNS). Some patients are CSF-positive but serum-negative, especially early in disease. For anti-LGI1 and anti-CASPR2, serum is usually positive and sufficient, but CSF is still recommended if serum is negative. Testing both increases diagnostic yield."
Q: When would you escalate to second-line immunotherapy?
A: "Escalate to rituximab or cyclophosphamide if there is inadequate response to first-line therapy at 2-4 weeks, or earlier if the presentation is severe (e.g., ICU admission, refractory status epilepticus, paraneoplastic syndrome). Rituximab targets B-cells (antibody production) and is preferred for cell surface antibody AE. Cyclophosphamide is broader immunosuppression, used for refractory cases or paraneoplastic syndromes."
Q: What is the significance of faciobrachial dystonic seizures?
A: "FBDS are pathognomonic for anti-LGI1 encephalitis. They are brief (less than 3 seconds), frequent (10-50+ per day), stereotyped dystonic postures involving the face and ipsilateral arm. Critically, they herald progression to cognitive impairment (limbic encephalitis). Early recognition and urgent immunotherapy can prevent cognitive decline. They often have no ictal EEG correlate, leading to debate about whether they are seizures or movement disorders, but they respond to immunotherapy, not ASMs."
Common Examination Mistakes
- ❌ Missing diagnosis in "psychiatric" presentation: Anti-NMDAR often presents to psychiatry first; always screen young patients with new psychosis for AE
- ❌ Waiting for antibody results before treating: Start immunotherapy empirically based on clinical criteria (results take weeks)
- ❌ Not performing tumor screening: Tumor removal essential for outcome; screen per antibody type (ovarian teratoma, SCLC, thymoma)
- ❌ Using haloperidol for agitation: Risk of NMS-like reaction; prefer benzodiazepines or low-dose atypical antipsychotics
- ❌ Stopping immunotherapy too early: Recovery is protracted (6-24 months); cognitive symptoms last to resolve
- ❌ Forgetting relapse surveillance: 20-24% relapse; tumor surveillance and maintenance immunotherapy reduce risk
- ❌ Not recognizing antibody-negative AE: 10-15% of cases are antibody-negative; clinical diagnosis and empirical treatment still warranted
Differential Diagnosis Pearls
| "Looks Like" | "But It's Actually AE Because..." |
|---|---|
| Schizophrenia | Acute onset (days, not months), seizures, movement disorder, CSF pleocytosis |
| Drug-induced psychosis | Symptoms persist despite abstinence, CSF abnormal, MRI abnormal |
| HSV encephalitis | Movement disorder prominent, less hemorrhagic, CSF negative for HSV PCR (or post-HSV choreoathetosis) |
| Conversion disorder | Bizarre movements, but CSF pleocytosis, MRI abnormal, antibodies positive |
| Neuroleptic malignant syndrome | Rigidity, but onset before antipsychotics or triggered by low dose, CSF abnormal |
Frequently asked questions
Quick clarifications for common clinical and exam-facing questions.
When should I seek emergency care for autoimmune encephalitis?
Seek immediate emergency care if you experience any of the following warning signs: Rapid cognitive decline (days to weeks), New-onset refractory status epilepticus (NORSE), Psychosis in young person without psychiatric history, Movement disorder with encephalopathy, Autonomic instability (tachycardia, hypoventilation), Decreased consciousness, Faciobrachial dystonic seizures.