Vitamin B12 Deficiency & Pernicious Anaemia
Vitamin B12 (cobalamin) deficiency represents a critical nutritional disorder with potentially devastating haematologica... MRCP exam preparation.
What matters first
Vitamin B12 (cobalamin) deficiency represents a critical nutritional disorder with potentially devastating haematologica... MRCP exam preparation.
Subacute Combined Degeneration of Cord (Sensory Ataxia)
6 Jan 2026
Generated educational material; verify before clinical use.
Visible references section
See the concept before reading it
Study the key anatomy, imaging, and decision pathways as full teaching plates.
Clinical board
A visual summary of the highest-yield teaching signals on this page.
Urgent signals
Safety-critical features pulled from the topic metadata.
- Subacute Combined Degeneration of Cord (Sensory Ataxia)
- Severe Pancytopenia (Pseudo-aplastic Picture)
- Folate given WITHOUT B12 (Precipitates cord damage)
- Rapidly Progressive Neurological Deterioration
Exam focus
Current exam surfaces linked to this topic.
- MRCP
Linked comparisons
Differentials and adjacent topics worth opening next.
- Folate Deficiency
- Hypothyroidism
Content status and exam context
This page is AI-generated educational content. It may contain errors or omissions and is not a substitute for current guidelines, local protocols, senior clinical judgement, or professional medical advice.
MedVellum does not claim an individual clinician reviewer, board certification, or professional credential for this page unless a future version names a real, verifiable reviewer.
Clinical explanation and evidence
Vitamin B12 Deficiency & Pernicious Anaemia
1. Overview
Vitamin B12 (cobalamin) deficiency represents a critical nutritional disorder with potentially devastating haematological and neurological consequences. The condition manifests across a spectrum from asymptomatic biochemical deficiency to severe megaloblastic anaemia and irreversible spinal cord degeneration. [1]
Pernicious anaemia, caused by autoimmune destruction of gastric parietal cells leading to intrinsic factor deficiency, remains the most common cause in developed nations, accounting for approximately 50-70% of cases. [2] However, the clinical landscape is evolving, with increasing recognition of drug-induced deficiency (particularly metformin and proton pump inhibitors), dietary insufficiency in strict vegans, and malabsorptive disorders. [3]
The condition is remarkable for its potential to cause irreversible neurological damage even in the absence of haematological abnormality. Approximately 25-30% of patients with B12 neuropathy have normal haemoglobin levels and mean corpuscular volume (MCV), making clinical vigilance essential. [4] The classic teaching that "neurological disease only occurs with severe anaemia" is demonstrably false and has led to delayed diagnosis and permanent disability.
Key Clinical Messages
- Neurological disease can precede or occur without haematological changes - always check B12 in unexplained neuropathy regardless of FBC
- Never treat isolated folate deficiency without excluding B12 deficiency first - folate can correct the anaemia while accelerating neurological deterioration
- Early treatment is critical - neurological recovery is time-dependent, with poor outcomes after > 6 months of symptoms
- Lifelong replacement is required for most causes - patient education is essential
2. Epidemiology
Prevalence and Incidence
Vitamin B12 deficiency shows marked geographic and age-related variation:
| Population | Prevalence | Key Risk Factors |
|---|---|---|
| General adult population | 3-6% | Age, dietary patterns |
| Adults > 60 years | 10-15% | Gastric atrophy, medications |
| Adults > 75 years | 15-20% | Multifactorial |
| Strict vegans | 60-80% | Dietary absence |
| Post-gastrectomy | 30-50% (within 5 years) | IF deficiency |
| Metformin > 4 years | 10-30% | Malabsorption |
Demographics
Pernicious Anaemia Specific:
- Age: Peak incidence 60-70 years; rare before age 30
- Sex: Female > Male (1.5:1 ratio)
- Ethnicity: Higher in Northern Europeans and persons of African descent
- Genetic predisposition: Increased risk with HLA-DR5 and HLA-DR3 haplotypes [2]
Geographic Variation:
- Higher prevalence in regions with lower dietary B12 intake (South Asia, parts of Africa)
- India reports prevalence of 47-70% in various populations due to predominantly vegetarian diet [10]
Temporal Trends
-
Increasing incidence of drug-induced deficiency:
- PPI use increased 400% between 2000-2020 [11]
- Metformin is first-line for type 2 diabetes (> 100 million users worldwide)
-
Earlier detection due to:
- Routine inclusion of B12 in metabolic panels
- Increased awareness of subclinical deficiency
- Availability of metabolite testing (MMA, homocysteine)
Risk Groups Requiring Screening
Based on British Society for Haematology (BSH) guidelines: [1]
- Age > 75 years with cognitive impairment or unexplained anaemia
- Strict vegetarians/vegans
- Inflammatory bowel disease (especially ileal Crohn's)
- Post-gastric surgery (any type)
- Long-term PPI or metformin use (> 12 months)
- Unexplained neuropathy or myelopathy
- Autoimmune conditions (especially thyroid disease, vitiligo, type 1 diabetes)
3. Aetiology & Pathophysiology
Dietary Sources and Requirements
Vitamin B12 is exclusively synthesized by microorganisms and enters the human food chain through animal products:
- Animal sources: Meat (especially liver), fish, eggs, dairy products
- No plant sources contain bioavailable B12 (except fortified foods)
- Daily requirement: 2.4 μg/day (WHO recommendation)
- Body stores: 2-5 mg (primarily liver), sufficient for 3-5 years if absorption ceases [1]
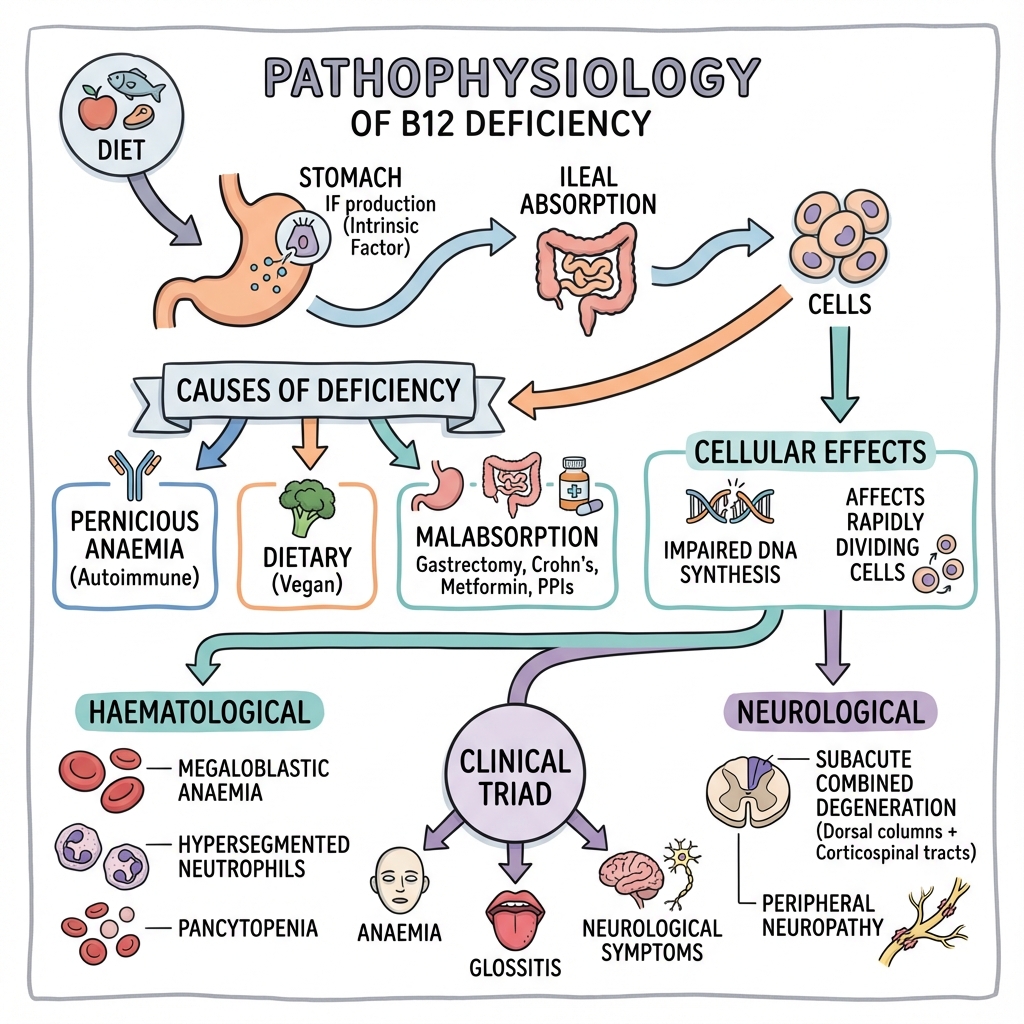
Complex Absorption Pathway
B12 absorption is one of the most complex nutrient absorption processes, requiring multiple steps and secretions:
Step 1: Oral Cavity & Oesophagus
- Dietary B12 bound to proteins in food
- Salivary haptocorrin (R-binder) released
- Forms B12-haptocorrin complex
Step 2: Stomach
- Gastric acid (pH less than 2) required to cleave B12 from food proteins [12]
- Parietal cells secrete intrinsic factor (IF)
- "Molecular weight: 50-60 kDa glycoprotein"
- Binds free B12 with high affinity
- Achlorhydria prevents B12 release from food proteins
Step 3: Duodenum & Jejunum
- Pancreatic proteases degrade haptocorrin
- Free B12 released and binds intrinsic factor
- B12-IF complex is resistant to further digestion
Step 4: Terminal Ileum
- Cubilin receptors on ileal enterocytes recognize B12-IF complex [13]
- Receptor-mediated endocytosis
- Requires calcium and neutral pH (≥6)
- IF degraded intracellularly; B12 released
Step 5: Portal Circulation
- B12 binds to transcobalamin II (TC-II)
- TC-II-B12 (holotranscobalamin) is the biologically active form
- Delivered to tissues via TC-II receptors
Causes of Deficiency
1. Decreased Intake (Dietary)
- Strict veganism (no animal products)
- Chronic malnutrition
- Eating disorders
- Rare: Infant exclusively breastfed by B12-deficient mother
2. Decreased Absorption
Gastric Causes:
| Condition | Mechanism | Prevalence in Deficiency |
|---|---|---|
| Pernicious anaemia | Autoimmune IF/parietal cell destruction | 50-70% |
| Atrophic gastritis | Loss of acid + IF production | 10-20% |
| Gastrectomy (total/partial) | Surgical removal of parietal cells | 30-50% at 5 years |
| H. pylori infection | Gastric inflammation, reduced acid | 5-10% |
Small Intestinal Causes:
- Terminal ileal disease:
- Crohn's disease (especially with ileal involvement)
- Ileal resection (> 60 cm typically required for deficiency)
- Radiation enteritis
- Lymphoma
- Bacterial overgrowth:
- Blind loop syndrome
- Diverticulosis
- Scleroderma (dysmotility)
- Post-surgical anatomical changes
- Bacteria consume dietary B12
- Parasitic infection:
- Diphyllobothrium latum (fish tapeworm)
- Inherited disorders:
- Imerslund-Gräsbeck syndrome (cubilin receptor defect)
- Transcobalamin II deficiency
Pancreatic Insufficiency:
- Chronic pancreatitis
- Cystic fibrosis
- Pancreatic cancer
- Mechanism: Lack of proteases prevents release of B12 from haptocorrin
3. Medications
| Drug Class | Mechanism | Onset | Reversibility |
|---|---|---|---|
| Proton pump inhibitors | Hypochlorhydria → decreased B12 release from food | 2-4 years | Usually reversible |
| H2 receptor antagonists | Reduced gastric acid | 2-4 years | Usually reversible |
| Metformin | Altered ileal motility, calcium-dependent IF-B12 uptake interference | 3-5 years | Often reversible |
| Colchicine | Ileal mucosal damage | Chronic use | Variable |
| Nitrous oxide | Inactivates B12 (oxidizes cobalt) | Acute (with chronic use) | Irreversible for affected molecules |
4. Increased Utilization
- Hyperthyroidism (increased metabolic rate)
- Pregnancy and lactation
- Haemolytic anaemia (increased erythropoiesis)
- Malignancy (rare)
Molecular Pathophysiology
B12 serves as a cofactor for two essential mammalian enzymes:
1. Methionine Synthase (Cytoplasmic)
Reaction:
Homocysteine + 5-Methyl-THF → Methionine + THF
(requires B12)
Consequences of deficiency:
- Folate trap: 5-methyl-THF accumulates, cannot be converted to THF
- THF is required for thymidine synthesis → impaired DNA synthesis
- Accumulation of homocysteine → hyperhomocysteinaemia (thrombotic risk)
- Methionine deficiency → impaired methylation reactions (DNA, proteins, lipids)
Haematological Effects:
- Megaloblastic erythropoiesis: Asynchronous nuclear-cytoplasmic maturation
- Hypersegmented neutrophils (> 5 lobes): Pathognomonic finding
- Ineffective erythropoiesis → intramedullary haemolysis → elevated bilirubin, LDH
- Thrombocytopenia and leucopenia in severe cases ("pseudo-aplastic anaemia")
2. Methylmalonyl-CoA Mutase (Mitochondrial)
Reaction:
Methylmalonyl-CoA → Succinyl-CoA
(requires adenosylcobalamin)
Consequences of deficiency:
- Methylmalonic acid (MMA) accumulation [17]
- MMA is neurotoxic:
- Incorporates abnormal fatty acids into myelin
- Disrupts myelin structure and stability
- Inhibits succinate dehydrogenase (mitochondrial dysfunction)
Neurological Effects:
- Demyelination of:
- Dorsal (posterior) columns (position, vibration sense)
- Lateral corticospinal tracts (motor)
- Spinocerebellar tracts (coordination)
- Peripheral neuropathy: Axonal degeneration
- Cerebral demyelination: Rare, but can occur in severe cases
- Optic neuropathy: Bilateral optic nerve demyelination
Why Neurological Damage Can Occur Without Anaemia
This critical phenomenon occurs because:
- Different thresholds: Nervous system is more sensitive to B12 depletion than bone marrow [4]
- Compartmentalization: Brain B12 levels may be critically low while serum levels are borderline
- Methylmalonic pathway: Neurological damage primarily via MMA accumulation, independent of folate trap
- Genetic polymorphisms: Variations in B12 metabolism enzymes affect individual susceptibility
- Concurrent folate sufficiency: Adequate folate intake may maintain DNA synthesis (preventing anaemia) while MMA-mediated neurotoxicity progresses
4. Clinical Presentation
B12 deficiency presents with a constellation of haematological, neurological, and constitutional features. Severity ranges from asymptomatic biochemical deficiency to life-threatening pancytopenia and severe myelopathy.
Constitutional Symptoms
- Fatigue: Disproportionate to degree of anaemia due to:
- Anaemia-related tissue hypoxia
- Mitochondrial dysfunction (MMA interference)
- Potential hypothalamic-pituitary dysfunction
- Weight loss: Especially with underlying malabsorptive disorders
- Anorexia: May contribute to self-perpetuating deficiency
Haematological Features
Anaemia Symptoms
- Dyspnoea on exertion
- Palpitations
- Presyncope/syncope
- Angina (if underlying coronary disease)
- Insidious onset: Slow progression allows cardiovascular compensation; patients may present with severe anaemia (Hb 60-70 g/L) yet ambulant
Examination Findings
- Pallor: Often pronounced
- Jaundice: Mild, lemon-yellow tinge
- "Mechanism: Ineffective erythropoiesis → intramedullary haemolysis → unconjugated hyperbilirubinaemia"
- Combination of pallor + jaundice = characteristic "lemon-yellow" appearance
- Tachycardia
- Flow murmurs: Due to increased cardiac output
- Hepatosplenomegaly: Rare; suggests concurrent disorder or extramedullary haematopoiesis
Mucosal Changes
- Glossitis: Painful, smooth, beefy-red tongue (atrophic glossitis)
- Loss of filiform papillae
- May have burning sensation or altered taste
- Occurs in 50-60% of patients [18]
- Angular stomatitis: Cracks at corners of mouth
Neurological Features (Subacute Combined Degeneration of the Cord)
SACD is the hallmark neurological syndrome, affecting 20-30% of B12-deficient patients. [4]
Peripheral Neuropathy
- Distribution: "Glove and stocking" pattern (length-dependent)
- Symptoms:
- Paraesthesias (tingling, "pins and needles")
- Numbness
- Painful dysesthesias
- Worse in lower limbs
- Progression: Distal → proximal over weeks to months
Dorsal Column Dysfunction (Posterior Columns)
Affects: Proprioception and vibration sense
Symptoms:
- Loss of position sense
- Sensory ataxia: Unsteady gait, worse in dark or with eyes closed
- Positive Romberg's sign (pathognomonic)
- "Walking on cotton wool" sensation
- Difficulty with fine motor tasks (buttoning shirts, picking up small objects)
Examination Findings:
- Reduced vibration sense (tuning fork testing) - typically lost first
- Begins at toes, progresses proximally
-
50% reduction compared to examiner's perception
- Impaired joint position sense
- Pseudoathetosis (involuntary writhing movements of fingers when eyes closed)
Lateral Corticospinal Tract Dysfunction
Affects: Motor pathways
Symptoms:
- Leg weakness (pyramidal pattern: extensors > flexors)
- Spasticity
- Gait disturbance
Examination Findings:
- Upper motor neuron signs:
- Increased tone (spasticity)
- Hyperreflexia (brisk knee and ankle jerks)
- Extensor plantar responses (Babinski sign)
- Clonus (sustained rhythmic contractions)
- Paradoxical combination: Absent ankle jerks (peripheral neuropathy) + extensor plantars (UMN lesion)
Autonomic Dysfunction
- Orthostatic hypotension
- Urinary retention or incontinence
- Erectile dysfunction
- Constipation
Optic Neuropathy
- Bilateral, symmetrical visual loss
- Central scotomas
- Reduced color vision (especially red-green)
- Rare manifestation (less than 1%)
Neuropsychiatric Features ("Megaloblastic Madness")
Seen in 10-20% of patients: [19]
- Cognitive impairment: Memory loss, executive dysfunction, dementia
- Mood disorders: Depression, irritability, emotional lability
- Psychosis: Hallucinations, delusions, paranoia
- Personality changes
Important: These may be the presenting feature, especially in elderly
- Mimics Alzheimer's disease or vascular dementia
- Potentially reversible if treated early
Pediatric Presentation
In infants born to B12-deficient mothers or exclusively breastfed by deficient mothers:
- Developmental delay or regression
- Hypotonia
- Seizures
- Failure to thrive
- Irritability, lethargy
5. Differential Diagnosis
Macrocytic Anaemia Differentials
| Cause | MCV | Neurological Signs | Key Distinguishing Features |
|---|---|---|---|
| B12 deficiency | 100-140 fL | Yes (SACD) | Hypersegmented neutrophils, ↓B12, ↑MMA, ↑homocysteine |
| Folate deficiency | 100-140 fL | No | Dietary history, ↓folate, normal MMA, ↑homocysteine |
| Alcohol excess | 100-110 fL | No (but peripheral neuropathy possible) | History, ↑GGT, ↑AST>ALT, target cells |
| Liver disease | 95-110 fL | Hepatic encephalopathy | Stigmata of chronic liver disease, abnormal LFTs |
| Hypothyroidism | 95-110 fL | Delayed relaxation reflexes | ↑TSH, symptoms of hypothyroidism |
| Myelodysplastic syndrome | Variable | No | Dysplasia on film/marrow, cytopenias, elderly |
| Reticulocytosis | Elevated | No | Recent haemolysis/bleeding, ↑reticulocyte count, polychromasia |
| Drugs | Variable | Depends on drug | Methotrexate, azathioprine, hydroxyurea, antiretrovirals |
| Myeloma | Normal-high | No | Hypercalcemia, renal impairment, bone pain, paraprotein |
| Aplastic anaemia | Normal-high | No | Pancytopenia, hypocellular marrow |
Neurological Differential for SACD
| Condition | Key Features | How to Distinguish |
|---|---|---|
| Multiple sclerosis | Relapsing-remitting, younger age, optic neuritis | MRI (periventricular lesions), CSF oligoclonal bands |
| Cervical myelopathy | Mechanical neck pain, radicular symptoms | MRI spine (cord compression) |
| Tabes dorsalis (neurosyphilis) | Argyll-Robertson pupils, loss of reflexes, lightning pains | Syphilis serology, CSF VDRL |
| HIV myelopathy | Known HIV, advanced disease | HIV testing, CD4 count |
| HTLV-1 myelopathy | Endemic areas, spastic paraparesis | HTLV-1 serology |
| Copper deficiency | Similar to SACD, often post-gastric surgery | ↓serum copper, ↓ceruloplasmin |
| Nitrous oxide toxicity | Recreational use, rapid onset | History of exposure, inactivated B12 |
| Friedreich's ataxia | Young onset, cerebellar signs, cardiomyopathy | Genetic testing |
6. Investigations
First-Line Tests
Full Blood Count (FBC)
Typical findings:
- Anaemia: Hb variable (may be normal or severely reduced less than 70 g/L)
- MCV elevated: Usually > 100 fL, often 110-140 fL
- "Caveat: May be normal if concurrent iron deficiency or thalassemia trait"
- Leucopenia: WBC often mildly reduced (3-4 × 10⁹/L)
- Thrombocytopenia: Mild (100-150 × 10⁹/L)
- In severe cases: Pancytopenia mimicking aplastic anaemia
Blood Film
Pathognomonic findings:
- Hypersegmented neutrophils: > 5 lobes (normal ≤5)
- Seen in 90% of cases
- May precede anaemia
- Persists even with mild deficiency
- Macroovalocytes: Large, oval red cells
- Anisocytosis: Variation in red cell size
- Poikilocytosis: Variation in red cell shape
- Tear drop cells: Rare
- Howell-Jolly bodies: Retained nuclear remnants (suggest functional hyposplenism)
- Megaloblastic changes: Immature, asynchronous maturation
Serum B12 (Cobalamin)
Reference range: 150-900 ng/L (111-665 pmol/L) - varies by laboratory
Interpretation:
| Level | Interpretation | Action |
|---|---|---|
| less than 150 ng/L | Deficient | Treat and investigate cause |
| 150-300 ng/L | Borderline/Grey zone | Check MMA and homocysteine |
| > 300 ng/L | Likely adequate | If high clinical suspicion, still check MMA |
Limitations of serum B12:
- False low (B12 deficiency overdiagnosed):
- Folate deficiency
- Pregnancy
- Oral contraceptive use
- Multiple myeloma
- HIV infection
- False normal/high (B12 deficiency missed):
- Liver disease (releases stored B12)
- Myeloproliferative disorders
- Renal failure
- Recent B12 intake (diet, supplements)
- High serum haptocorrin (carries biologically inactive B12)
- 25-30% of true B12 deficiency have borderline levels [4]
Serum Folate
- Typically normal or elevated (folate trap phenomenon)
- Low folate suggests combined deficiency
- Red cell folate: More accurate than serum folate but less commonly used
Second-Line (Confirmatory) Tests
Metabolite Testing (Gold Standard for Borderline Cases)
Methylmalonic Acid (MMA):
- Normal: less than 0.4 μmol/L
- Elevated in B12 deficiency: Often > 1.0 μmol/L
- Sensitivity: 95-98% for B12 deficiency [17]
- Specificity: 90-95%
- False elevations: Renal impairment (most common), dehydration, bacterial overgrowth
- Advantage: Specific for B12 deficiency (not elevated in folate deficiency)
Homocysteine:
- Normal: less than 15 μmol/L
- Elevated in B12 deficiency: > 15-20 μmol/L
- Sensitivity: 95-100%
- Specificity: Lower (also elevated in folate deficiency, renal disease, hypothyroidism, smoking, genetic MTHFR variants)
- Use: Supportive evidence; less specific than MMA
Holotranscobalamin (Active B12):
- Measures B12 bound to transcobalamin II (biologically active form)
- Represents 20-30% of total serum B12
- Advantage: Becomes abnormal earlier than total B12
- Limitation: Not widely available; interpretation guidelines still evolving
Autoantibody Testing (For Pernicious Anaemia)
Anti-Intrinsic Factor Antibodies:
- Specificity: 95-100% (highly specific for pernicious anaemia) [2]
- Sensitivity: 40-60% (many false negatives)
- Interpretation:
- Positive → Confirms pernicious anaemia
- Negative → Does NOT exclude pernicious anaemia
- Two types: Blocking antibodies (type I) and binding antibodies (type II)
Anti-Parietal Cell Antibodies:
- Sensitivity: 80-90%
- Specificity: 50-60% (found in 10% of healthy elderly)
- Interpretation:
- Positive → Supportive but not diagnostic
- Negative → Reduces likelihood of pernicious anaemia
- Clinical use: Screening; if positive, check anti-IF antibodies
Other Biochemistry
Elevated in B12 deficiency:
- LDH: Markedly elevated (ineffective erythropoiesis)
- Unconjugated bilirubin: Mildly elevated (intramedullary haemolysis)
- Iron and ferritin: Often elevated (reduced utilization for erythropoiesis)
Normal/Low:
- Haptoglobin: Reduced (haemolysis)
- Reticulocyte count: Inappropriately low for degree of anaemia
Third-Line/Specialist Investigations
Bone Marrow Examination
Indications (rarely required):
- Diagnostic uncertainty (? MDS, ? leukaemia)
- Severe pancytopenia
- Atypical features
Findings in B12 deficiency:
- Hypercellular marrow (erythroid hyperplasia)
- Megaloblastic changes:
- Large erythroblasts with immature nuclei and mature cytoplasm
- Nuclear-cytoplasmic asynchrony
- Open chromatin pattern ("stippled" nuclei)
- Giant metamyelocytes and band forms
- Reversal of myeloid:erythroid ratio
Gastroscopy (OGD)
Indications:
- Suspected pernicious anaemia (to assess gastric atrophy)
- Age > 55 with new diagnosis (gastric cancer surveillance)
- Alarm symptoms (dysphagia, weight loss, GI bleeding)
Findings:
- Atrophic gastritis (body and fundus)
- Loss of gastric rugal folds
- Intestinal metaplasia on biopsy
- Excludes gastric malignancy
Additional Testing Based on Suspected Cause
Malabsorptive disorders:
- Coeliac serology (anti-tTG, anti-EMA)
- Faecal elastase (pancreatic insufficiency)
- Hydrogen breath test (bacterial overgrowth)
- Terminal ileum imaging (MR enterography for Crohn's)
Associated autoimmune conditions:
- TSH, free T4 (autoimmune thyroid disease in 30%)
- Anti-thyroid peroxidase antibodies
- Morning cortisol (Addison's disease - rare)
- Fasting glucose, HbA1c (type 1 diabetes)
Schilling Test
Historical: Radioactive B12 absorption test with and without IF Current status: No longer performed (radioactive isotope unavailability; replaced by antibody and metabolite testing)
7. Management
Management depends on the cause, severity, and presence of neurological complications. The key principles are:
- Immediate treatment if neurological signs present
- Lifelong replacement for most causes
- Investigation of underlying cause
- Monitoring for response and complications
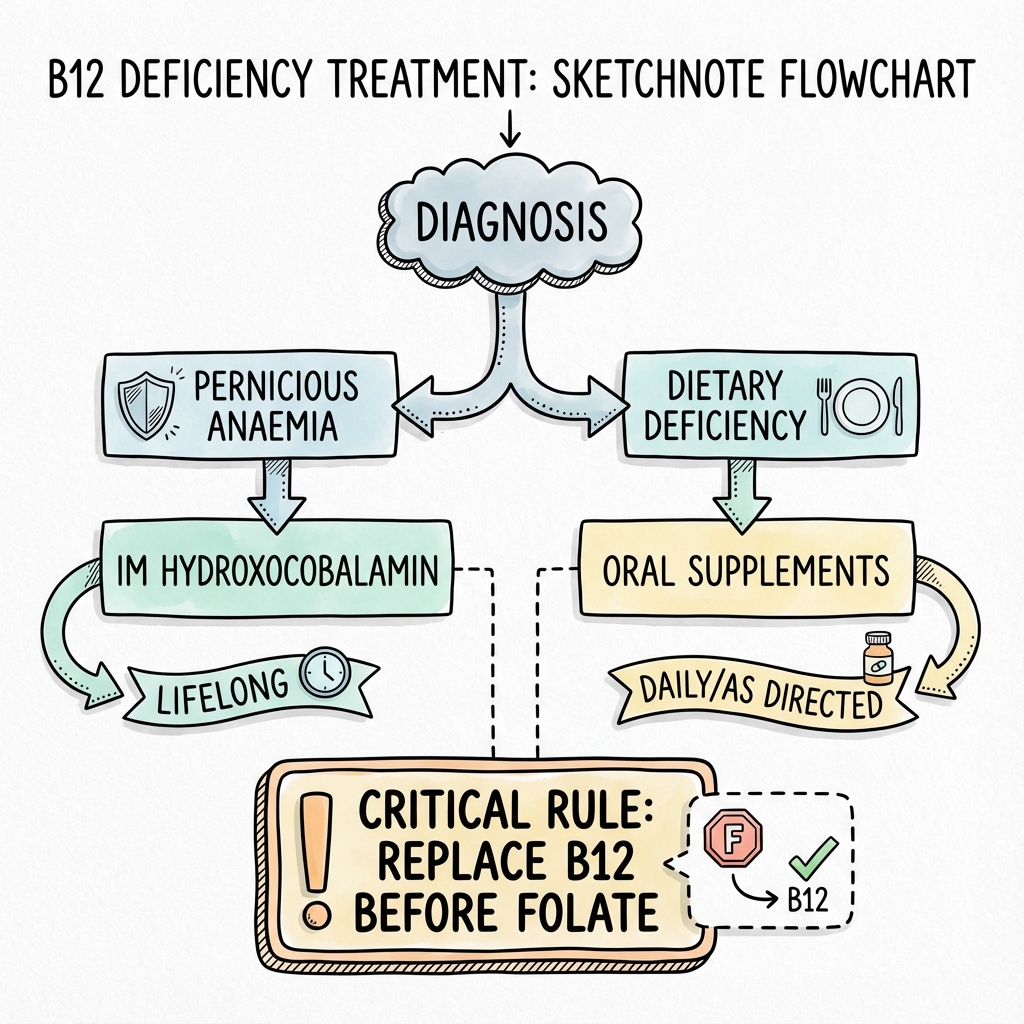
Acute/Urgent Management
Patients WITH Neurological Features (SACD)
Intensive replacement regimen:
- Hydroxocobalamin 1 mg IM on alternate days
- Continue until no further clinical improvement in neurological symptoms
- Typically requires 2-3 weeks (10-15 injections)
- Followed by maintenance therapy [1]
Rationale: Neurological recovery is time-dependent; high-dose frequent replacement maximizes tissue saturation
Patients WITHOUT Neurological Features
Standard replacement regimen:
- Hydroxocobalamin 1 mg IM three times weekly for 2 weeks (6 doses)
- Then switch to maintenance
Alternative (for dietary deficiency in compliant patients):
- Oral cyanocobalamin 50-150 μg daily
- Only suitable if:
- No malabsorption
- No pernicious anaemia
- Compliant patient
- Close monitoring
Maintenance Therapy
Standard maintenance:
- Hydroxocobalamin 1 mg IM every 3 months for life
- For pernicious anaemia, post-gastrectomy, ileal resection
For neurological involvement:
- Hydroxocobalamin 1 mg IM every 2 months for life
- Some patients require more frequent dosing (every 4-6 weeks) based on symptom recurrence
Oral maintenance (selected cases only):
- Cyanocobalamin 1000-2000 μg daily
- Suitable for:
- Dietary deficiency (vegans)
- Drug-induced deficiency if medication cannot be stopped
- Patient preference (if absorption intact)
- Requires monitoring of B12 levels
Preparation Differences
| Preparation | Route | Availability | Notes |
|---|---|---|---|
| Hydroxocobalamin | IM | UK, Europe, Australia | Preferred (longer half-life, better retention) |
| Cyanocobalamin | IM or Oral | US, worldwide | Requires conversion to active forms |
| Methylcobalamin | Oral, sublingual | Supplements | No proven advantage over cyanocobalamin |
| Adenosylcobalamin | Oral | Supplements | No proven advantage |
Treatment Monitoring
Expected Response Timeline
Haematological:
- 24-48 hours: Feeling of well-being, improved appetite
- 5-7 days: Reticulocytosis (peaks at day 7) - "reticulocyte response"
- May cause transient hypokalemia (K⁺ uptake by new cells)
- 1-2 weeks: Hb begins to rise
- 4-8 weeks: Hb normalizes
- 2-3 months: MCV normalizes
Neurological:
- 1-3 months: Subjective improvement in paresthesias
- 3-6 months: Objective improvement in sensory testing
- 6-12 months: Maximum recovery achieved
- Residual deficits: Common if > 6 months untreated before therapy
Monitoring Tests
Initial (first 2-3 months):
- FBC weekly for first month, then monthly
- Check for reticulocyte response
- Monitor Hb and MCV improvement
- Potassium: Check at 48-72 hours if severe anaemia (risk of hypokalemia)
- Iron studies: If Hb fails to rise, consider concurrent iron deficiency
Long-term (annual):
- FBC (ensure no recurrence)
- Consider serum B12 if symptoms recur
- "Note: Serum B12 will be high after IM replacement (not useful)"
- MMA is more useful if doubt about adequacy
Management of Specific Causes
Pernicious Anaemia
- Replacement: Lifelong hydroxocobalamin IM every 3 months
- Monitoring:
- FBC annually
- Consider gastroscopy at diagnosis and surveillance (gastric cancer risk 2-3×)
- Screen for other autoimmune diseases (thyroid, coeliac)
- Patient education: Lifelong treatment, importance of adherence
Dietary Deficiency (Veganism)
- Acute: As per standard replacement if deficient
- Maintenance: Oral cyanocobalamin 50-100 μg daily OR dietary fortification
- Prevention: Education on B12-fortified foods (plant-based milks, cereals, nutritional yeast)
- Monitoring: Annual B12 if high-risk diet
Drug-Induced (Metformin, PPIs)
- Options:
- Discontinue offending drug if possible
- Continue drug + B12 supplementation
- Metformin: Typically reversible after discontinuation, but often cannot stop (diabetes control)
- Oral B12 supplementation effective
- Check B12 annually in long-term users
- PPIs: Consider de-escalation or switching to H2 antagonist
- B12 supplementation if cannot stop
Post-Surgical (Gastrectomy, Ileal Resection)
- Lifelong IM replacement: Standard maintenance dose
- Monitoring: FBC at 6 months post-op, then annually
Crohn's Disease / Ileal Disease
- If extensive ileal involvement: Likely lifelong IM replacement
- If limited disease or post-resection less than 60 cm: Trial of oral replacement with monitoring
Special Populations
Pregnancy
- Risks if deficient:
- Maternal megaloblastic anaemia
- Fetal neural tube defects
- Preterm delivery
- Low birth weight
- Treatment: Urgent replacement if deficient
- IM hydroxocobalamin safe in pregnancy
- Folic acid supplementation: Essential (400 μg daily), but check B12 first
Elderly
- High prevalence: Screen liberally
- Atypical presentations: Cognitive impairment, falls, mood changes
- Compliance: Consider IM over oral (better adherence)
Nitrous Oxide Exposure
- Mechanism: Irreversibly oxidizes cobalt atom in B12
- Acute treatment: High-dose hydroxocobalamin (some use 1 mg IM daily for 1-2 weeks)
- Avoid further N₂O exposure
- Prognosis: Variable; some cases of severe myelopathy
8. Complications
From the Disease
Untreated B12 Deficiency
- Permanent neurological disability: Irreversible if > 6-12 months untreated
- Residual ataxia
- Chronic neuropathy pain
- Spasticity
- Cognitive impairment/dementia: May be irreversible
- Severe pancytopenia: Risk of infections, bleeding
- Infertility: Both male and female
- Pregnancy complications: Neural tube defects, miscarriage
Associated with Pernicious Anaemia
- Gastric adenocarcinoma: 2-3× increased risk [2]
- "Recommendations: OGD at diagnosis; consider surveillance"
- Gastric carcinoid tumors: Hypergastrinemia stimulates enterochromaffin-like cells
- Usually low-grade (type I)
- Requires surveillance endoscopy
- Other autoimmune conditions (cluster):
- "Autoimmune thyroid disease: 30% [2]"
- "Type 1 diabetes: 5-10%"
- "Vitiligo: 2-5%"
- "Addison's disease: less than 1%"
From Treatment
Early Complications (First Week)
- Hypokalemia: Cellular uptake of K⁺ during rapid erythropoiesis
- Monitor K⁺ in severe anaemia
- Supplement if less than 3.5 mmol/L
- Thrombocytosis: Rebound after initial thrombocytopenia
- Exacerbation of heart failure: Volume expansion in severe anaemia
- Gout: Increased uric acid from cellular turnover
Injection-Related
- Pain/soreness at injection site
- Rare: Anaphylaxis (extremely rare with hydroxocobalamin)
Folate Supplementation Without B12
- Neurological precipitation: "Folate trap" corrects anaemia but accelerates SACD
- Critical safety point: NEVER give folate without ensuring B12 adequacy
9. Prognosis and Outcomes
Haematological Recovery
- Excellent prognosis: > 95% complete normalization
- Timeline: 2-3 months for full haematological recovery
- Persistence of macrocytosis: May take 4-6 months
Neurological Recovery
Highly variable and dependent on:
- Duration of symptoms before treatment:
- less than 6 months: Good recovery (70-80%)
- 6-12 months: Partial recovery (40-50%)
-
12 months: Minimal recovery (less than 20%) [4]
- Severity at presentation
- Age (elderly recover less well)
Typical recovery pattern:
- Paresthesias improve first (50% improve within 3 months)
- Proprioception recovers slowly (may take 6-12 months)
- Motor weakness improves variably
- Established spastic paraparesis rarely fully recovers
Cognitive/Psychiatric Recovery
- Early treatment: Often reversible
- Prolonged deficiency: May leave residual cognitive impairment
- Dementia: If truly due to B12 deficiency, 50-70% improve with treatment
Mortality
- Untreated severe deficiency: Historical mortality 10-20% (infection, bleeding)
- With treatment: Near-normal life expectancy
- Excess mortality: From associated conditions (gastric cancer in pernicious anaemia)
10. Prevention & Screening
Primary Prevention
Dietary advice for at-risk populations:
- Vegans:
- B12-fortified foods (plant milks, cereals, nutritional yeast)
- Oral supplementation (50-100 μg daily or 2000 μg weekly)
- Vegetarians: Ensure adequate dairy/egg intake
- Elderly: Maintain varied diet including animal products
Medication counseling:
- Metformin users: Consider annual B12 screening after 3-5 years
- Long-term PPI users: Consider de-escalation or supplementation
Secondary Prevention (Screening)
No universal population screening recommended, but targeted screening advised for: [1]
| Risk Group | Screening Test | Frequency |
|---|---|---|
| Vegans (strict) | Serum B12 | Annually or when symptoms |
| Post-gastrectomy | Serum B12, FBC | 6 months, then annually |
| Ileal resection > 60 cm | Serum B12, FBC | 6 months, then annually |
| Metformin > 3-4 years | Serum B12 | Every 1-2 years |
| Long-term PPI (> 1 year) | Serum B12 | Consider at 2-3 years |
| Unexplained neuropathy | Serum B12, MMA | Once |
| Cognitive impairment (elderly) | Serum B12 | Once |
| Autoimmune disease | Serum B12 | At diagnosis |
Tertiary Prevention
For diagnosed pernicious anaemia:
- Lifelong IM B12 replacement
- Annual FBC
- Consider gastroscopy at diagnosis for gastric cancer risk stratification
- Surveillance OGD controversial; some recommend every 3-5 years if extensive gastric atrophy
For other causes:
- Maintenance therapy as appropriate
- Address underlying cause if possible
- Monitor for relapse
11. Key Guidelines
British Society for Haematology (BSH) 2014 [1]
Title: Guidelines for the diagnosis and treatment of cobalamin and folate disorders
Key Recommendations:
- Serum B12 less than 150 ng/L: Deficient - treat
- 150-300 ng/L: Check MMA and homocysteine
- Treat neurological patients with intensive regimen (alternate day IM until improvement)
- Lifelong IM replacement for pernicious anaemia and malabsorption
NICE Clinical Knowledge Summary (CKS)
Title: Anaemia - B12 and folate deficiency
Key Recommendations:
- Check anti-IF antibodies if suspected pernicious anaemia
- Use MMA if borderline B12 levels
- Hydroxocobalamin preferred over cyanocobalamin (UK)
- Standard regimen: 1 mg IM 3× weekly for 2 weeks
National Institute for Health and Care Excellence (NICE) - NG214 (2023)
Context: Gastro-oesophageal reflux disease and dyspepsia in adults
Relevance: Guidance on PPI deprescribing and awareness of B12 deficiency risk
American Society of Hematology (ASH)
Consensus: Similar diagnostic approach, preference for cyanocobalamin in US
12. Examination Focus
Common MRCP / FRACP Exam Scenarios
MCQ/SBA Stem Examples
Q1: "A 68-year-old woman presents with fatigue and 'pins and needles' in her feet. Blood tests show Hb 95 g/L, MCV 115 fL. Which blood film finding is most specific?"
- Answer: Hypersegmented neutrophils
Q2: "A patient with known B12 deficiency develops neurological symptoms. What is the most appropriate initial treatment?"
- Answer: Hydroxocobalamin 1 mg IM on alternate days
Q3: "A 55-year-old vegan presents with macrocytic anaemia. Serum B12 is 180 ng/L. Which test best confirms tissue B12 deficiency?"
- Answer: Serum methylmalonic acid
Q4: "Which autoantibody is most specific for pernicious anaemia?"
- Answer: Anti-intrinsic factor antibody
Q5: "A patient with combined B12 and folate deficiency requires treatment. What is the safest approach?"
- Answer: Treat B12 first, then folate
PACES / Clinical Examination Scenarios
Station 5 (Communication):
- Explaining lifelong injections to newly diagnosed pernicious anaemia patient
- Counseling vegan patient on dietary B12 sources
- Breaking news of need for gastroscopy (gastric cancer risk)
Station 1 (History):
- Elderly patient with cognitive decline (? B12-related dementia)
- Patient with neuropathy and unsteady gait
Station 3 (Cardiovascular/Neurology):
- Neurological examination showing SACD signs
- Demonstrate Romberg's test
- Assess vibration sense (tuning fork)
- Test proprioception
- Elicit extensor plantars with absent ankle jerks
Presenting findings: "This patient has evidence of subacute combined degeneration of the cord, with loss of vibration and proprioception in the lower limbs, indicating dorsal column involvement, combined with brisk knee reflexes and extensor plantar responses, indicating lateral corticospinal tract involvement. Notably, the ankle jerks are absent, suggesting concurrent peripheral neuropathy. This combination is highly suggestive of vitamin B12 deficiency, and I would check B12 levels, methylmalonic acid, and anti-intrinsic factor antibodies to investigate for pernicious anaemia."
Viva Voce (Oral Exam) Points
Opening statement (if asked "Tell me about B12 deficiency"): "Vitamin B12 deficiency is a disorder of impaired DNA synthesis and myelin metabolism caused by insufficient cobalamin intake or absorption. It presents with megaloblastic anaemia and potentially irreversible neurological damage. Pernicious anaemia, caused by autoimmune destruction of gastric parietal cells, is the most common cause in developed nations, accounting for 50-70% of cases."
Key facts to mention unprompted:
- Serum B12 can be normal in 25-30% of tissue deficiency - use MMA
- Neurological disease can occur without anaemia
- Never treat folate without checking B12 first - risk of precipitating SACD
- Hypersegmented neutrophils are pathognomonic
- Pernicious anaemia carries 2-3× gastric cancer risk
Likely viva questions and model answers:
Q: "Why do patients get jaundice in B12 deficiency?" A: "It's due to ineffective erythropoiesis. The megaloblastic red cell precursors are so abnormal that they undergo apoptosis within the bone marrow before being released - a process called intramedullary haemolysis. This releases haemoglobin, which is broken down to unconjugated bilirubin, causing mild jaundice. Combined with the pallor of anaemia, this gives the characteristic 'lemon-yellow' appearance."
Q: "Describe the absorption of B12." A: "B12 absorption is complex. Dietary B12 is released from food proteins by gastric acid and initially bound by salivary haptocorrin. In the duodenum, pancreatic proteases degrade haptocorrin, releasing B12 to bind intrinsic factor, which is secreted by gastric parietal cells. The B12-IF complex is then absorbed in the terminal ileum via cubilin receptors. This explains why deficiency can occur with gastric disease (loss of IF), pancreatic insufficiency (failure to release B12 from haptocorrin), or ileal disease (loss of absorption site)."
Q: "Why does the Schilling test no longer get performed?" A: "The Schilling test used radioactive B12 to differentiate causes of malabsorption - patients received radiolabeled B12 with and without intrinsic factor, and urinary excretion was measured. It's no longer performed due to unavailability of radioactive isotopes and radiation safety concerns. We now use anti-intrinsic factor antibodies and anti-parietal cell antibodies to diagnose pernicious anaemia, and MMA/homocysteine to confirm tissue deficiency."
Q: "What is the 'folate trap'?" A: "In B12 deficiency, methionine synthase cannot function. This enzyme normally converts 5-methyl-THF back to tetrahydrofolate (THF), using B12 as a cofactor. Without B12, folate becomes 'trapped' in the 5-methyl form and cannot be recycled to THF, which is needed for DNA synthesis. This explains the megaloblastic anaemia. If you give folate alone, you can bypass this trap and correct the anaemia, but the neurological damage from methylmalonic acid accumulation will continue or worsen. This is why you must always correct B12 before or with folate."
Common Exam Mistakes
❌ Failing to check B12 in neuropathy with normal FBC
- 25-30% of B12 neuropathy have normal haematology
❌ Treating folate deficiency without checking B12
- Precipitates SACD - dangerous
❌ Missing hypersegmented neutrophils on film
- Pathognomonic and high-yield for exams
❌ Using wrong replacement regimen
- Neurological patients need alternate day dosing, not weekly
❌ Relying on serum B12 alone in borderline cases
- Should check MMA and homocysteine
❌ Not screening for other autoimmune conditions in pernicious anaemia
- 30% have thyroid disease
❌ Forgetting gastric cancer surveillance in pernicious anaemia
- 2-3× risk
13. Patient Education and Layperson Explanation
What is Vitamin B12?
Vitamin B12 is a vitamin found only in animal products like meat, fish, dairy, and eggs. Your body needs it to make healthy red blood cells (which carry oxygen around your body) and to keep your nerves working properly.
What Causes B12 Deficiency?
There are several reasons you might not have enough B12:
- Your diet doesn't include animal products (if you're vegan or strictly vegetarian)
- Your stomach can't absorb it properly - this is called pernicious anaemia. Your immune system has attacked the cells in your stomach that help you absorb B12.
- You've had stomach surgery or bowel surgery that removed the parts that absorb B12
- Some medications can interfere with B12 absorption, especially if taken long-term:
- Metformin (for diabetes)
- Medications for heartburn/reflux (PPIs like omeprazole)
What Are the Symptoms?
- Tiredness - often severe and out of proportion to blood test results
- Breathlessness when doing normal activities
- Sore, red tongue that may burn or tingle
- Pins and needles in your hands and feet
- Unsteadiness when walking, especially in the dark
- Memory problems or feeling confused
- Mood changes - depression or irritability
- Pale or slightly yellow skin
Why Do I Need Injections?
If you have pernicious anaemia or problems absorbing B12, your stomach cannot absorb the vitamin from food or tablets, no matter how much you eat. The only way to get B12 into your body is to bypass the stomach and inject it directly into your muscle.
How Often Do I Need Injections?
- Initially: You'll have frequent injections (2-3 times per week for 2 weeks, or alternate days if you have nerve problems)
- Long-term: Usually every 3 months for life (or every 2 months if you have nerve damage)
Are the Injections Safe?
Yes, B12 injections are very safe. You cannot overdose on B12 - your body uses what it needs and passes the rest out in your urine. Side effects are rare - you might get some soreness at the injection site.
Will My Symptoms Get Better?
- Tiredness and anaemia: Usually improves within days to weeks
- Nerve problems (pins and needles, unsteadiness): May take months to improve, and sometimes don't fully recover if they've been present for a long time
This is why early treatment is so important.
Do I Need Any Other Tests?
If you have pernicious anaemia:
- Your doctor may recommend a camera test of your stomach (gastroscopy) to check for other problems
- You should have blood tests for thyroid problems (these can occur together)
- You'll need yearly blood tests to check your blood count
Can I Ever Stop Treatment?
For most people with pernicious anaemia or stomach/bowel surgery, no - treatment is lifelong. Your body cannot suddenly start absorbing B12 again.
If your deficiency is due to diet (veganism), you can prevent it by:
- Taking B12 supplements daily by mouth
- Eating fortified foods (plant-based milks, cereals, nutritional yeast)
Important Safety Warning
If you also have low folate (another vitamin), your doctor must treat your B12 first. Treating folate without B12 can make nerve damage worse.
Questions to Ask Your Doctor
- Why do I have B12 deficiency?
- Will I need injections for life?
- Should I take any other vitamins?
- Do I need any follow-up tests?
- Can I have the injections at home or at my GP surgery?
14. References
-
Devalia V, Hamilton MS, Molloy AM; British Committee for Standards in Haematology. Guidelines for the diagnosis and treatment of cobalamin and folate disorders. Br J Haematol. 2014;166(4):496-513. doi:10.1111/bjh.12959
-
Toh BH, van Driel IR, Gleeson PA. Pernicious anemia. N Engl J Med. 1997;337(20):1441-1448. doi:10.1056/NEJM199711133372007
-
Green R, Allen LH, Bjørke-Monsen AL, et al. Vitamin B12 deficiency. Nat Rev Dis Primers. 2017;3:17040. doi:10.1038/nrdp.2017.40
-
Stabler SP. Vitamin B12 deficiency. N Engl J Med. 2013;368(2):149-160. doi:10.1056/NEJMcp1113996
-
Allen LH. How common is vitamin B-12 deficiency? Am J Clin Nutr. 2009;89(2):693S-696S. doi:10.3945/ajcn.2008.26947A
-
Andres E, Loukili NH, Noel E, et al. Vitamin B12 (cobalamin) deficiency in elderly patients. CMAJ. 2004;171(3):251-259. doi:10.1503/cmaj.1031155
-
Pawlak R, Parrott SJ, Raj S, Cullum-Dugan D, Lucus D. How prevalent is vitamin B12 deficiency among vegetarians? Nutr Rev. 2013;71(2):110-117. doi:10.1111/nure.12001
-
Bauman WA, Shaw S, Jayatilleke E, Spungen AM, Herbert V. Increased intake of calcium reverses vitamin B12 malabsorption induced by metformin. Diabetes Care. 2000;23(9):1227-1231. doi:10.2337/diacare.23.9.1227
-
de Jager J, Kooy A, Lehert P, et al. Long term treatment with metformin in patients with type 2 diabetes and risk of vitamin B-12 deficiency: randomised placebo controlled trial. BMJ. 2010;340:c2181. doi:10.1136/bmj.c2181
-
Yajnik CS, Deshpande SS, Lubree HG, et al. Vitamin B12 deficiency and hyperhomocysteinemia in rural and urban Indians. J Assoc Physicians India. 2006;54:775-782.
-
Lam JR, Schneider JL, Zhao W, Corley DA. Proton pump inhibitor and histamine 2 receptor antagonist use and vitamin B12 deficiency. JAMA. 2013;310(22):2435-2442. doi:10.1001/jama.2013.280490
-
Russell RM, Baik H, Kehayias JJ. Older men and women efficiently absorb vitamin B-12 from milk and fortified bread. J Nutr. 2001;131(2):291-293. doi:10.1093/jn/131.2.291
-
Fyfe JC, Madsen M, Højrup P, et al. The functional cobalamin (vitamin B12)-intrinsic factor receptor is a novel complex of cubilin and amnionless. Blood. 2004;103(5):1573-1579. doi:10.1182/blood-2003-08-2852
-
Carmel R. Prevalence of undiagnosed pernicious anemia in the elderly. Arch Intern Med. 1996;156(10):1097-1100.
-
Kaptan K, Beyan C, Ural AU, et al. Helicobacter pylori--is it a novel causative agent in Vitamin B12 deficiency? Arch Intern Med. 2000;160(9):1349-1353. doi:10.1001/archinte.160.9.1349
-
Singer MA, Lazaridis C, Nations SP, Wolfe GI. Reversible nitrous oxide-induced myeloneuropathy with pernicious anemia: case report and literature review. Muscle Nerve. 2008;37(1):125-129. doi:10.1002/mus.20780
-
Savage DG, Lindenbaum J, Stabler SP, Allen RH. Sensitivity of serum methylmalonic acid and total homocysteine determinations for diagnosing cobalamin and folate deficiencies. Am J Med. 1994;96(3):239-246. doi:10.1016/0002-9343(94)90149-x
-
Kohlmeier L, Kark JD, Gomez-Gracia E, et al. Lycopene and myocardial infarction risk in the EURAMIC Study. Am J Epidemiol. 1997;146(8):618-626. doi:10.1093/oxfordjournals.aje.a009327
-
Lindenbaum J, Healton EB, Savage DG, et al. Neuropsychiatric disorders caused by cobalamin deficiency in the absence of anemia or macrocytosis. N Engl J Med. 1988;318(26):1720-1728. doi:10.1056/NEJM198806303182604
-
Kumar N, Gross JB Jr, Ahlskog JE. Copper deficiency myelopathy produces a clinical picture like subacute combined degeneration. Neurology. 2004;63(1):33-39. doi:10.1212/01.wnl.0000132644.52613.fa
Medical Disclaimer: MedVellum content is for educational purposes and clinical reference. Clinical decisions should account for individual patient circumstances. Always consult appropriate specialists and current local guidelines.
Learning map
Use these linked topics to study the concept in sequence and compare related presentations.
Prerequisites
Start here if you need the foundation before this topic.
- Haematopoiesis
- Neuroanatomy - Spinal Cord Tracts
Differentials
Competing diagnoses and look-alikes to compare.
- Folate Deficiency
- Hypothyroidism
- Myelodysplastic Syndrome
- Alcohol-Related Macrocytosis
Consequences
Complications and downstream problems to keep in mind.
- Pancytopenia
- Myelopathy