Intestinal Obstruction
Intestinal Obstruction represents a mechanical or functional blockage preventing the normal transit of intestinal conten... MRCS exam preparation.
What matters first
Intestinal Obstruction represents a mechanical or functional blockage preventing the normal transit of intestinal conten... MRCS exam preparation.
Constant pain (Suggestive of strangulation/ischaemia)
6 Jan 2026
Generated educational material; verify before clinical use.
Visible references section
See the concept before reading it
Study the key anatomy, imaging, and decision pathways as full teaching plates.
Clinical board
A visual summary of the highest-yield teaching signals on this page.
Urgent signals
Safety-critical features pulled from the topic metadata.
- Constant pain (Suggestive of strangulation/ischaemia)
- Fever + Peritonism (Perforation)
- Elevated Lactate >2.0 mmol/L
- Closed-loop pattern on CT (High risk of rapid necrosis)
Exam focus
Current exam surfaces linked to this topic.
- MRCS
Linked comparisons
Differentials and adjacent topics worth opening next.
- Paralytic Ileus
- Pseudo-obstruction (Ogilvie's)
Content status and exam context
This page is AI-generated educational content. It may contain errors or omissions and is not a substitute for current guidelines, local protocols, senior clinical judgement, or professional medical advice.
MedVellum does not claim an individual clinician reviewer, board certification, or professional credential for this page unless a future version names a real, verifiable reviewer.
Clinical explanation and evidence
Intestinal Obstruction (Adult Master Topic)
1. Clinical Overview
Intestinal Obstruction represents a mechanical or functional blockage preventing the normal transit of intestinal contents through the small or large bowel. This emergency surgical condition accounts for approximately 15% of all acute abdominal admissions and represents the second most common indication for emergency laparotomy after appendicitis. [1]
The clinical significance of bowel obstruction lies primarily in the risk of strangulation—a life-threatening complication where vascular compromise leads to bowel ischaemia, necrosis, perforation, and subsequent septic shock. Without timely recognition and intervention, strangulated bowel carries mortality rates of 20-30%, compared to 2-8% for simple obstruction. [7]
Obstruction Classification
By Location:
- Small Bowel Obstruction (SBO): Proximal to the ileocaecal valve
- Large Bowel Obstruction (LBO): Distal to the ileocaecal valve
- Closed-Loop Obstruction: Obstruction at two points, creating an isolated segment
By Pathophysiology:
- Simple Obstruction: Mechanical blockage without vascular compromise
- Strangulated Obstruction: Vascular compromise with ischaemia/necrosis
- Partial Obstruction: Incomplete blockage allowing some passage
- Complete Obstruction: No passage of luminal contents
By Aetiology:
- Mechanical: Physical blockage (adhesions, tumours, hernias)
- Functional: Impaired peristalsis without anatomical obstruction (ileus)
The Paradigm Shift in Management
Historical surgical dogma dictated "never let the sun set on a bowel obstruction," advocating for early operative intervention. Modern evidence-based practice has refined this approach: uncomplicated adhesive SBO may be safely managed conservatively for 24-72 hours with close monitoring, supported by the Gastrografin challenge protocol. [1] However, any signs of strangulation, peritonitis, or closed-loop obstruction demand immediate surgical exploration.
2. Epidemiology and Clinical Burden
Incidence and Demographics
Small Bowel Obstruction:
- Incidence: 350-400 cases per 100,000 population annually
- More common in patients with previous abdominal surgery (90% have prior operations)
- Peak incidence: 60-80 years old
- No significant gender predominance
Large Bowel Obstruction:
- Incidence: 60-80 cases per 100,000 population annually
- Peak incidence: 70-80 years old (correlating with colorectal malignancy peak)
- Slight male predominance (1.5:1) reflecting higher colorectal cancer rates
Healthcare Impact
- Represents 12-16% of surgical emergency admissions
- Average hospital length of stay: 5-7 days for conservative management, 10-14 days for operative management
- Readmission rate: 18% within 30 days for surgically managed cases, 12% for conservative [15]
- Mortality: 2-8% overall; up to 30% with perforation [16]
Aetiology by Location
| Type | Primary Causes | Relative Frequency | Strangulation Risk |
|---|---|---|---|
| SBO | Adhesions (post-surgical) | 60-75% | Moderate (5-10%) |
| Hernia (incarcerated) | 15-20% | High (15-25%) | |
| Malignancy | 5-10% | Low (unless invasive) | |
| Crohn's strictures | 3-5% | Low | |
| LBO | Colorectal malignancy | 50-60% | Low (unless perforation) |
| Diverticular stricture | 15-20% | Low | |
| Sigmoid volvulus | 10-15% | Very High (30-40%) | |
| Caecal volvulus | 2-5% | Very High (30-40%) |
Special Population Considerations
Post-Surgical Patients:
- 90% of patients undergoing multiple laparotomies develop adhesions
- Only 10% of patients with adhesions develop symptomatic SBO
- Risk highest in first 2 years post-surgery, persists lifelong
Elderly Patients (>70 years):
- Higher operative rate: 48% vs 32% in younger patients
- Higher mortality: 5.9% vs 1.2% [11]
- Frailty scores independently predict adverse outcomes
- Greater likelihood of LBO from malignancy
Virgin Abdomen (No Prior Surgery):
- SBO in virgin abdomen suggests: hernia (60%), malignancy (30%), inflammatory (10%)
- Mandatory detailed examination of all hernia orifices
- High index of suspicion for occult femoral hernia in elderly women [14]
3. Aetiology and Molecular Pathophysiology
The 8-Stage Cascade to Strangulation
Understanding the temporal evolution from simple to strangulated obstruction is critical for surgical decision-making:
Stage 1: Mechanical Obstruction (0-6 hours)
Mechanism: Physical blockage prevents aboral transit of luminal contents. Effect: Proximal bowel continues normal secretion (8-10L/day of GI secretions) but cannot evacuate. Clinical: Onset of colicky abdominal pain, early vomiting (if proximal).
Stage 2: Proximal Dilatation (6-12 hours)
Mechanism: Accumulation of swallowed air, secretions, and bacterial gas production. Luminal pressure: Rises from normal 5-10 mmHg to 15-20 mmHg. Effect: Bowel wall stretch triggers peristaltic waves (colic), distension activates stretch receptors. Clinical: Visible distension, high-pitched bowel sounds, progressive pain.
Stage 3: Third-Space Fluid Sequestration (12-24 hours)
Mechanism: Elevated intraluminal pressure (>20 mmHg) exceeds lymphatic drainage capacity. Effect: Bowel wall oedema develops; fluid transudates into lumen and peritoneal cavity (up to 6L/day). Biochemical: Hypovolaemia, haemoconcentration, prerenal azotaemia. Clinical: Dehydration, tachycardia, oliguria, electrolyte derangement. [5,6]
Stage 4: Venous Compression (24-36 hours)
Mechanism: Intraluminal pressure (>30 mmHg) exceeds venous outflow pressure (20-30 mmHg). Effect: Venous congestion leads to capillary engorgement, interstitial haemorrhage. Gross appearance: Bowel becomes dusky, purple, haemorrhagic ("red flag" at laparotomy). Clinical: Pain becomes constant, lactate begins to rise. [6]
Stage 5: Bacterial Overgrowth and Translocation (24-48 hours)
Mechanism: Stasis and ischaemia create anaerobic environment; resident flora (E. coli, Bacteroides fragilis, Enterococcus) proliferate exponentially (10^3 to 10^9 CFU/mL). Mucosal barrier: Hypoxia and distension disrupt tight junctions between enterocytes. Effect: Bacteria and endotoxins (lipopolysaccharide, LPS) translocate across mucosa into mesenteric lymphatics and bloodstream. Systemic: Triggers systemic inflammatory response syndrome (SIRS), early sepsis. [5,7]
Stage 6: Arterial Compromise (36-48 hours)
Mechanism: Sustained intraluminal pressure (>80-100 mmHg in closed-loop) exceeds mesenteric arterial perfusion pressure. Effect: Transmural infarction—all layers of bowel wall become ischaemic. Gross appearance: Bowel becomes black, thin-walled, friable, with loss of peristalsis. Clinical: Extreme toxicity, lactic acidosis (>4 mmol/L), MODS picture. [6,7]
Stage 7: Perforation (48-72 hours)
Mechanism: Necrotic bowel wall loses structural integrity; intraluminal pressure causes rupture. Location: Typically at site of maximal distension (caecum in LBO per Law of Laplace). Effect: Faecal peritonitis—spillage of 10^9 bacteria/mL into peritoneal cavity. Clinical: Generalized peritonitis, board-like rigidity, septic shock. [7]
Stage 8: Multi-Organ Dysfunction Syndrome (MODS) (>72 hours)
Mechanism: Uncontrolled sepsis, cytokine storm, endothelial dysfunction. Organs affected: Acute kidney injury, ARDS, DIC, hepatic dysfunction. Outcome: Mortality approaches 50-70% despite maximal intervention. [7]
The Law of Laplace in LBO
Formula: Wall Tension (T) = (Pressure × Radius) / (2 × Wall Thickness)
Clinical Application:
- The caecum has the largest radius (~9 cm) and thinnest wall (~1 mm) in the colon.
- In distal LBO (e.g., sigmoid tumour with competent ileocaecal valve), intraluminal pressure distributes throughout the colon but creates maximal wall tension at the caecum.
- Critical threshold: Caecal diameter >10-12 cm on X-ray indicates impending perforation, necessitating urgent decompression. [12]
Closed-Loop Obstruction: The Most Dangerous Variant
Definition: Obstruction at two points creates an isolated segment with both inflow and outflow blocked.
Causes:
- Adhesive band creating a "U" or "C" shaped loop
- Volvulus (sigmoid, caecal)
- Internal hernia
- Competent ileocaecal valve in distal LBO
Pathophysiology:
- Isolated segment continues secretion but cannot decompress.
- Intraluminal pressure rises rapidly (within 6-12 hours).
- Vascular compromise occurs early (24-36 hours vs 48-72 hours in simple obstruction).
- High strangulation rate: 30-40% [9]
Imaging: "U" or "C" shaped dilated loop, radial distribution of mesenteric vessels ("whirl sign" in volvulus), two distinct transition points. [12]
4. Clinical Presentation
The "Classic Tetrad" of Obstruction
-
Abdominal Pain
- Character: Initially colicky (intermittent waves every 4-5 minutes)
- Mechanism: Hyperperistalsis proximal to obstruction
- Evolution: Constant pain suggests strangulation (red flag)
- Location: Diffuse or periumbilical (SBO), lower abdomen (LBO)
-
Vomiting
- SBO: Early, profuse, bilious (within 2-4 hours)
- Proximal SBO: Almost immediate, large volume
- Distal SBO: Delayed (12-24 hours), may become faeculent
- LBO: Late (>24-48 hours), faeculent if ileocaecal valve incompetent
-
Distension
- SBO: Mild to moderate central distension
- LBO: Marked, generalized distension (if ileocaecal valve incompetent) or isolated colonic distension
- Mechanism: Accumulation of gas and fluid proximal to obstruction
-
Absolute Constipation
- Definition: No passage of flatus or stool for >24 hours
- Complete obstruction: Absolute constipation is hallmark
- Partial obstruction: May still pass flatus or small amounts of stool [14]
- Important: Passage of flatus/stool does not exclude obstruction if symptoms persist
Physical Examination Findings
Inspection:
- Visible peristalsis (rare, pathognomonic in thin patients)
- Distension: central (SBO), generalized (LBO)
- Previous surgical scars (adhesion risk)
- Hernias: inspect all orifices (inguinal, femoral, umbilical, incisional)
Auscultation:
- Early: High-pitched, tinkling bowel sounds ("rushes" during colic)
- Late: Absent sounds suggest ileus or late strangulation
- Listening over distended bowel may reveal fluid "succussion splash"
Palpation:
- Tenderness: Diffuse mild tenderness (simple obstruction) vs localized severe tenderness (strangulation)
- Peritonism (guarding, rigidity, rebound): Red flag for perforation or strangulation
- Palpable mass: Suggests malignancy, intussusception, or herniated segment
- Hernial orifices: Mandatory examination—occult femoral hernia common in elderly women [8]
Percussion:
- Resonant (tympanitic) over gas-filled loops
- Shifting dullness if significant ascites (poor prognostic sign)
Digital Rectal Examination (DRE):
- Empty rectum: Classic sign of complete mechanical obstruction
- Blood: Suggests ischaemia, malignancy, or intussusception
- Palpable mass: Rectal or low sigmoid tumour
- Ballooning: Suggests distal obstruction with proximal loading
Red Flags for Strangulation
| Clinical Feature | Significance | Sensitivity | Specificity |
|---|---|---|---|
| Constant pain | Loss of colic suggests dead bowel | 71% | 62% |
| Fever (>38°C) | Bacterial translocation, peritonitis | 40% | 89% |
| Tachycardia (>100) | Hypovolaemia or sepsis | 78% | 45% |
| Peritoneal signs | Perforation or transmural necrosis | 67% | 93% |
| Leucocytosis (>15k) | Inflammatory response | 58% | 70% |
| Lactate (>2.0) | Tissue hypoperfusion | 86% | 91% |
| Procalcitonin (>0.5) | Bacterial translocation marker | 67% | 89% |
Clinical Pearl: The combination of constant pain, fever, and lactate >2.0 mmol/L has a positive predictive value of 85% for strangulation and mandates immediate laparotomy. [6,17]
Distinguishing SBO from LBO
| Feature | Small Bowel Obstruction | Large Bowel Obstruction |
|---|---|---|
| Pain onset | Rapid (within hours) | Gradual (over days) |
| Vomiting | Early, profuse, bilious | Late, faeculent |
| Distension | Mild to moderate, central | Marked, generalized |
| Constipation | Develops within 12-24h | Prominent presenting feature |
| Previous surgery | Common (adhesions) | Less relevant |
| Age | Any age | Elderly (malignancy) |
5. Investigations
Laboratory Studies
Essential Bloods:
- FBC: Leucocytosis (>15k suggests strangulation); haemoconcentration (dehydration)
- U&E: Prerenal azotaemia (elevated urea, creatinine); hypokalaemia, hypochloraemia (vomiting)
- LFTs: Usually normal unless severe sepsis or underlying liver disease
- Lactate: >2.0 mmol/L highly specific (91%) for strangulation [17]
- Procalcitonin: >0.5 ng/mL sensitivity 67%, specificity 89% for strangulation [17]
- CRP: Elevated but non-specific; trend useful
- Amylase: Exclude pancreatitis; may be mildly elevated in obstruction
- Group & Save: Preparation for potential surgery
Arterial Blood Gas:
- Metabolic acidosis (base deficit >-5) suggests ischaemia
- Lactate trending: Serial measurements more useful than single value
Imaging: The Diagnostic Cornerstone
Plain Radiography (AXR + Erect CXR)
Indications: Initial assessment, point-of-care evaluation, resource-limited settings.
Limitations: Sensitivity 60-70% for SBO, 80-90% for LBO; cannot assess for strangulation; cannot reliably identify cause. [2]
SBO Findings:
- Central loops: Dilated small bowel (>3 cm diameter) in central abdomen
- Valvulae conniventes: Complete crossing of bowel lumen (vs haustra in colon)
- Air-fluid levels: Multiple levels on erect film at different heights ("step-ladder" pattern)
- Gasless colon: Paucity of gas in large bowel
LBO Findings:
- Peripheral loops: Dilated colon (>6 cm transverse, >9 cm caecum) in periphery
- Haustra: Incomplete crossing of bowel wall
- Caecal distension: >9 cm (urgent decompression indicated if >10-12 cm) [12]
- "Coffee bean" sign: Sigmoid volvulus (dilated loop resembling coffee bean)
Erect CXR:
- Pneumoperitoneum: Free air under diaphragm indicates perforation (immediate laparotomy)
Computed Tomography (CT) with IV Contrast: Gold Standard
Indications: All patients with suspected bowel obstruction (unless clear simple obstruction on AXR with conservative management planned).
Sensitivity: 95% for diagnosis, 83% for high-grade obstruction [2]
Specificity: 96% [2]
Key Findings:
-
Transition Point (100% sensitivity)
- Clear demarcation between dilated proximal and collapsed distal bowel
- Identifies exact anatomical location and often the cause
-
Small Bowel Feces Sign (89% sensitivity for SBO)
- Particulate matter resembling feces in dilated small bowel
-
Closed-Loop Obstruction
- "U" or "C" shaped dilated loop
- Radial configuration of mesenteric vessels ("whirl sign" in volvulus)
- Two distinct transition points [9,12]
-
Strangulation Predictors (Combined PPV 85%)
- Reduced bowel wall enhancement: Hypoenhancement vs adjacent normal bowel (OR 8.9) [19]
- Mesenteric edema: Haziness, fat stranding (OR 11.6) [19]
- Ascites: Free fluid without cirrhosis/malignancy (OR 4.3) [19]
- Pneumatosis intestinalis: Gas in bowel wall (late, ominous sign)
- Portal venous gas: Gas in portal vein branches (transmural necrosis)
-
Aetiology Identification:
- Adhesions: Abrupt angulation, no mass
- Hernia: Bowel protruding through defect (internal, external)
- Malignancy: Soft tissue mass, lymphadenopathy, liver metastases
- Volvulus: Whirl sign, "bird's beak" at twist point
Absence of High-Risk Features: If no reduced enhancement, no mesenteric edema, no ascites → 100% negative predictive value for strangulation (safe for conservative trial). [13]
Water-Soluble Contrast Studies (Gastrografin Challenge)
Indications:
- Uncomplicated adhesive SBO without high-risk features
- Diagnostic and therapeutic roles
Protocol:
- Administer 100 mL Gastrografin via NG tube or orally (if no NGT)
- Perform abdominal X-ray at 4-6 hours and 24 hours
- Assess for contrast in colon at 24 hours
Interpretation:
- Success: Contrast in colon at 24h → 90% likelihood of spontaneous resolution [1,3]
- Failure: No contrast in colon at 24h → 90% likelihood of requiring surgery [1,3]
Diagnostic Performance:
- Sensitivity: 96% for predicting need for surgery [8]
- Specificity: 98% [8]
Therapeutic Mechanism:
- Hyperosmolar (1900 mOsm/L): Draws fluid into bowel lumen
- Reduces wall edema: Decreases third-spacing
- Stimulates peristalsis: Direct prokinetic effect [3,8,20]
Evidence Base:
- Cochrane Review 2007: Reduces need for surgery by 40%, shortens LOS by 1.9 days [3]
- Meta-analysis 2016: Reduces operative rate by 14% (NNT = 7) [8]
- RCT (Velmahos 2001): LOS 5.1 days vs 3.2 days (pless than 0.01) [20]
Contraindications:
- Suspected perforation (water-soluble but still avoid)
- High-risk features for strangulation (lactate >2.0, peritonism)
- Closed-loop obstruction
Ultrasound
Indications: Bedside assessment, pregnancy, paediatrics.
Findings: Dilated bowel loops (>2.5 cm small bowel, >6 cm colon), free fluid, peristalsis assessment.
Limitations: Operator-dependent, limited by bowel gas, cannot reliably identify cause.
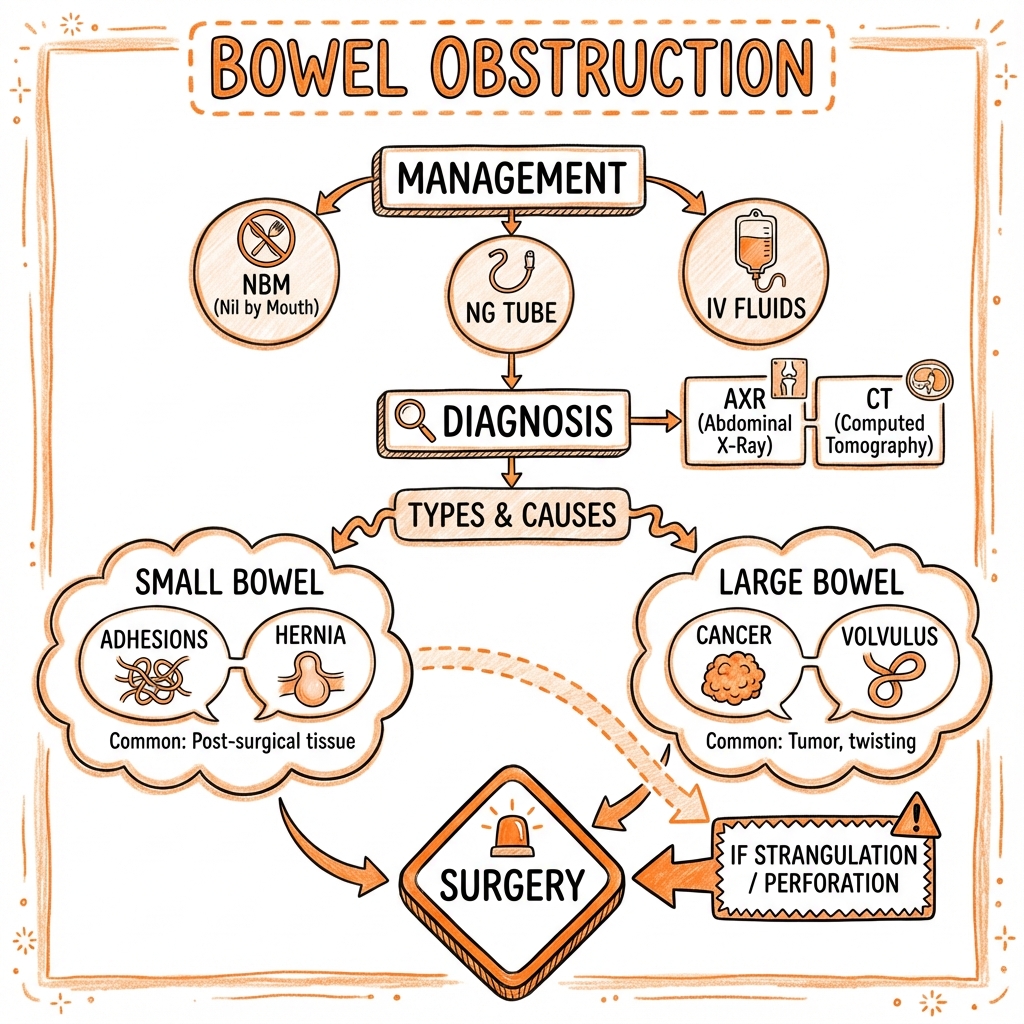
6. Management
Principles of Management
The management of intestinal obstruction follows a structured algorithm balancing conservative and surgical approaches:
- Resuscitation: All patients require aggressive fluid resuscitation and decompression.
- Risk Stratification: Distinguish simple from strangulated obstruction.
- Conservative Trial: Appropriate for uncomplicated adhesive SBO (24-72 hours).
- Surgical Intervention: Mandatory for high-risk features, failed conservative trial, or LBO.
Initial Resuscitation: "Drip and Suck"
Fluid Resuscitation ("The Drip"):
Goal: Correct hypovolaemia from third-space losses (up to 6L/day).
Fluid Choice:
- Crystalloid: Hartmann's solution or 0.9% saline (avoid saline in high-volume resuscitation due to hyperchloraemic acidosis)
- Volume: Initial bolus 1-2L over 1-2 hours, then maintenance 125-250 mL/hr guided by:
- Urine output (target >0.5 mL/kg/hr)
- Lactate normalization
- Cardiovascular parameters (HR, BP, CVP if available)
Monitoring:
- Hourly urine output (catheterize if input/output monitoring required)
- Serial lactate measurements (4-hourly if elevated)
- Daily electrolytes (K+, Mg2+, PO4- replacement)
Gastrointestinal Decompression ("The Suck"):
Nasogastric Tube (NGT):
- Indication: All patients with vomiting, significant distension, or complete obstruction
- Size: Wide-bore (14-16 Fr Ryle's tube) to allow drainage of particulate matter
- Placement: Confirm position with aspirate pH less than 5.5 or X-ray
- Management: Free drainage on bag; aspirate 4-hourly and document volume
- Evidence: No benefit in partial SBO with minimal vomiting; may delay resolution [18]
Controversy: Recent evidence suggests NGT decompression provides no faster resolution in partial SBO compared to free oral intake, but remains standard for complete obstruction and vomiting. [18]
Analgesia:
- Opiates permitted: Historical concern that opiates "mask" peritonism is unfounded; adequate analgesia facilitates examination and reassessment.
- Regimen: IV morphine 5-10 mg PRN or PCA; IV paracetamol 1g QDS; avoid NSAIDs (renal risk in dehydration).
Antibiotics:
- Not routine for simple obstruction
- Indicated if high-risk features (fever, leucocytosis, lactate elevation) suggesting strangulation or perforation
- Regimen: Broad-spectrum covering GI flora: Cefuroxime 1.5g + Metronidazole 500mg IV TDS OR Piperacillin-Tazobactam 4.5g IV TDS
Nil By Mouth (NBM):
- All patients initially NBM until obstruction resolves or definitive management complete
Conservative Management: The 24-72 Hour Window
Indications for Conservative Trial (ALL criteria must be met):
- Adhesive SBO (history of previous abdominal surgery)
- Partial obstruction (passage of some flatus) OR complete obstruction without high-risk features
- No signs of strangulation:
- Pain remains colicky (not constant)
- Afebrile
- No peritonism
- Lactate less than 2.0 mmol/L
- No closed-loop on CT
- No reduced bowel wall enhancement
Protocol:
- Initial 24 hours: Drip and suck, observe closely
- Gastrografin challenge: Administer at 24 hours if no resolution; X-ray at 48 hours (24h post-Gastrografin)
- Daily reassessment: Clinical examination, bloods (lactate, WCC), imaging if deterioration
- Endpoint: Resolution (passage of flatus/stool, reduced distension, tolerating diet) OR failure (no improvement at 72h, deterioration, or development of red flags)
Success Rate: 60-80% for adhesive SBO; >90% if Gastrografin in colon at 24h [1,3,8]
Duration: Maximum 72 hours; prolonged conservative management beyond this increases complication risk 3-fold. [16]
Surgical Management
Indications for Immediate Laparotomy (The "Red" List)
Absolute Indications (Operate within 2-4 hours):
- Peritonitis: Generalized tenderness, guarding, rigidity (perforation or strangulation)
- Pneumoperitoneum: Free air on erect CXR/CT (perforation)
- Strangulation Features:
- Constant pain + fever + lactate >2.0 mmol/L
- Reduced bowel wall enhancement on CT
- Mesenteric edema + ascites [6,13,17]
- Closed-Loop Obstruction: High risk of rapid strangulation (30-40% within 24-48h) [9]
- Irreducible Incarcerated Hernia: Tender, non-reducible groin/abdominal wall mass
Relative Indications (Operate within 12-24 hours):
- Virgin Abdomen SBO: No previous surgery (suggests hernia or malignancy requiring exploration)
- Failed Conservative Trial: No improvement at 72 hours, failed Gastrografin challenge
- LBO: Most large bowel obstructions require intervention (see below)
- Caecal diameter >10-12 cm: Impending perforation per Law of Laplace [12]
Surgical Approaches
Laparoscopy vs Open:
- Laparoscopy: Preferred if experienced surgeon available; reduced adhesion formation, faster recovery [15]
- Contraindications to laparoscopy: Massive distension (limited working space), hemodynamic instability, suspected perforation
- Conversion rate: 20-30% (often unavoidable due to dense adhesions or bowel friability)
Operative Principles:
- Full laparotomy exploration: Identify cause and all transition points
- Assess bowel viability:
- Viable: Pink, peristaltic, bleeding from cut edge
- Questionable: Dusky, minimal peristalsis → observe after release of obstruction (10 minutes)
- Non-viable: Black, thin-walled, no peristalsis, no bleeding → resection mandatory
- Lysis of adhesions: Careful sharp dissection; avoid enterotomy
- Resection: If non-viable bowel or inability to safely release
- Anastomosis vs stoma: Primary anastomosis preferred if: viable bowel, minimal contamination, haemodynamically stable
Small Bowel Obstruction Surgery
Adhesive SBO:
- Procedure: Laparoscopic or open adhesiolysis
- Principle: Divide adhesive bands causing obstruction
- Resection rate: 10-20% (if ischaemic segment or multiple dense adhesions)
- Anastomosis: Usually hand-sewn or stapled side-to-side
- Recurrence: 18% after surgery vs 12% after successful conservative management; laparoscopy reduces recurrence vs open [15]
Incarcerated Hernia:
- Procedure: Hernia repair (open or laparoscopic)
- Bowel viability: Assess after reduction; resect if non-viable
- Mesh: Avoid if contaminated field (strangulation, resection)
Small Bowel Malignancy:
- Procedure: Segmental resection with clear margins
- Lymphadenectomy: If curative intent
Large Bowel Obstruction Surgery
Malignant LBO:
Options:
-
Resection + Primary Anastomosis:
- Preferred if: right-sided, minimal contamination, stable patient
- On-table lavage if left-sided (via appendicostomy)
- Lower stoma rate, single operation [5]
-
Hartmann's Procedure:
- Resection of tumour, end colostomy, rectal stump
- Indication: Left-sided with perforation/contamination, unstable patient
- Reversal rate: 50-60% (many patients never reversed)
-
Subtotal Colectomy + Ileorectal Anastomosis:
- Indication: Synchronous tumours, caecal perforation with distal tumour
- Avoids stoma, higher diarrhoea rate initially
-
Stenting as Bridge to Surgery:
- Self-expanding metal stent (SEMS) via colonoscopy
- Converts emergency to elective surgery (7-10 days later)
- Advantages: Bowel preparation possible, single-stage resection, reduced stoma rate (30% vs 60%) [5]
- Disadvantages: Perforation risk (5-10%), migration, failure rate (10-15%)
- Evidence: WSES guidelines support as bridge to surgery in left-sided malignant LBO [5]
Sigmoid Volvulus:
First-Line: Endoscopic Decompression
- Procedure: Rigid sigmoidoscopy + flatus tube insertion
- Success rate: 80-90% [4]
- Technique: Direct visualization, gentle advancement past twist, insert flatus tube (leave 48-72h)
- Contraindications: Peritonitis, perforation, failed attempt
Second-Line: Surgery
- Emergency: Sigmoid colectomy + Hartmann's or primary anastomosis (if viable bowel, minimal contamination)
- Elective (after successful decompression): Sigmoid colectomy within 2 weeks (recurrence rate 50-90% if not resected) [4]
Caecal Volvulus:
- Treatment: Right hemicolectomy (caecopexy has high recurrence)
- Urgency: Emergency (high strangulation risk 30-40%)
Diverticular Stricture:
- Procedure: Sigmoid colectomy (resect stricture)
- Approach: Similar to malignant LBO (often difficult to differentiate pre-op)
Management Algorithm Summary
BOWEL OBSTRUCTION SUSPECTED
↓
1. RESUSCITATION: IV fluids + NGT + analgesia
↓
2. INVESTIGATIONS: Bloods (lactate, WCC) + CT abdomen/pelvis
↓
3. RISK STRATIFICATION
|
├─→ HIGH RISK (Red Flags Present)
| - Peritonitis, pneumoperitoneum
| - Lactate >2.0, constant pain, fever
| - Closed-loop, reduced bowel enhancement
| → IMMEDIATE LAPAROTOMY (within 2-4 hours)
|
├─→ LBO (Large Bowel Obstruction)
| ├─→ Volvulus → Endoscopic decompression (sigmoid) OR Right hemicolectomy (caecal)
| ├─→ Malignancy → Stenting (bridge to surgery) OR Resection ± stoma
| └─→ Diverticular → Resection
|
└─→ LOW RISK (Uncomplicated Adhesive SBO)
→ CONSERVATIVE TRIAL (24-72h)
├─→ Gastrografin challenge at 24h
├─→ Daily reassessment
├─→ Success (60-80%): Resolution, discharge
└─→ Failure OR deterioration → LAPAROTOMY
7. Prognosis and Outcomes
Mortality
| Clinical Scenario | Mortality | Reference |
|---|---|---|
| Simple SBO (conservative) | 0-2% | [1] |
| Simple SBO (operative) | 2-5% | [7] |
| Strangulated SBO | 20-30% | [7] |
| Perforated bowel | 30-50% | [16] |
| LBO (all-comers) | 10-15% | [5] |
| Elderly (>70 years) | 5.9% vs 1.2% (less than 70y) | [11] |
Predictors of Mortality:
- Age >70 years (OR 4.9)
- Delayed surgery >5 days (OR 3.2) [16]
- Strangulation (OR 15.3)
- Multiple comorbidities (ASA ≥3)
- Perforation with faecal peritonitis
Morbidity
Postoperative Complications:
- Wound infection: 10-15%
- Anastomotic leak: 3-5%
- Intra-abdominal abscess: 5-8%
- Enterocutaneous fistula: 1-2%
- Prolonged ileus: 10-20%
Recurrence
Adhesive SBO:
- Overall recurrence: 15-20% over 5 years
- After conservative management: 12% [15]
- After operative management: 18% [15]
- Paradox: Surgery cures the acute episode but creates new adhesions, increasing long-term recurrence risk.
Laparoscopy vs Open:
- Laparoscopic adhesiolysis: Reduced recurrence vs open (OR 0.6) [15]
Sigmoid Volvulus:
- Without resection: 50-90% recurrence [4]
- After elective resection: less than 5% recurrence
Long-Term Quality of Life
Short Bowel Syndrome:
- Risk if extensive resection (>200 cm small bowel removed)
- Requires lifelong TPN if less than 100 cm remaining
- Major impact on quality of life, high healthcare costs
Chronic Intermittent Obstruction:
- 10-15% develop recurrent subacute episodes
- May require dietary modification (low-residue diet)
- Psychological impact of recurrent hospital admissions
8. Special Scenarios
Post-Operative Ileus vs Early Post-Operative SBO
Post-Operative Ileus:
- Definition: Transient impairment of GI motility following surgery
- Onset: Immediate (day 0-3 post-op)
- Duration: Resolves by day 3-5
- Character: Diffuse, non-obstructive
- Management: Conservative (mobilization, gum chewing, avoid opiates)
Early Post-Operative SBO (EPSBO):
- Definition: Mechanical SBO within 30 days of surgery
- Incidence: 1-3% of abdominal surgeries
- Onset: Typically days 5-10 post-op (after initial ileus resolution)
- Cause: Fibrinous adhesions (before collagen maturation at 3-6 weeks)
- Management: Conservative trial (success 70-80%); surgery if failed (high technical difficulty, friable tissues)
Radiation Enteritis
- Cause: Late complication of pelvic radiotherapy (GI, gynae, urological malignancies)
- Pathophysiology: Chronic ischaemia, fibrosis, stricture formation
- Presentation: Recurrent SBO episodes 6 months to 20 years post-radiotherapy
- Management: Conservative preferred (surgery extremely challenging due to frozen abdomen, friable tissues, poor healing)
- Operative risk: Perforation, fistula, short bowel syndrome
Pregnancy
- Incidence: 1 in 2,500-3,500 pregnancies
- Cause: Adhesive SBO most common (prior surgery + gravid uterus)
- Trimester: Most common in 3rd trimester (mechanical compression) or immediate post-partum (sudden uterine decompression)
- Diagnosis: Challenging (limited CT due to radiation; MRI preferred)
- Management: Conservative if possible; surgery if high-risk features (fetal mortality 20-30% if laparotomy required)
- Obstetric considerations: Tocolysis if preterm, coordinate with obstetrics
Crohn's Disease
- SBO in Crohn's: Stricturing phenotype (Vienna classification B2)
- Pathophysiology: Transmural inflammation → fibrosis → stricture
- Presentation: Recurrent subacute SBO episodes
- Medical management: Corticosteroids, biologics (infliximab, adalimumab) may reduce inflammation but do not reverse fibrosis
- Surgical management: Strictureplasty (preserve bowel length) vs resection (if less than 40 cm affected segment)
- Recurrence: 50% require further surgery within 10 years
9. Evidence Base and Landmark Trials
| Trial/Guideline | Year | Population | Intervention | Key Results | Impact |
|---|---|---|---|---|---|
| Bologna Guidelines [1] | 2018 | Adhesive SBO | Structured conservative protocol | Conservative safe 24-72h; Gastrografin reduces surgery 40% | Standardized evidence-based conservative management |
| EAST Guidelines [2] | 2012 | SBO (all causes) | Diagnostic/management protocol | CT sensitivity 95%; operative indications defined | Risk stratification framework |
| Cochrane Review [3] | 2007 | Adhesive SBO | Gastrografin vs placebo | ↓ Surgery (RR 0.62), ↓ LOS 1.9 days | Established therapeutic role of contrast |
| ASCRS Guidelines [4] | 2016 | Colon volvulus | Endoscopic vs surgical | Sigmoid: 80% endoscopic success; recurrence 50-90% without surgery | First-line endoscopic decompression |
| WSES Guidelines [5] | 2017 | Malignant LBO | Stenting vs emergency surgery | Stenting ↓ stoma rate (30% vs 60%); bridge to elective surgery | Paradigm shift: stenting as bridge |
| Kulvatunyou (ProSBO) [6] | 2018 | SBO (prospective) | CT + clinical predictors | Mesenteric edema, reduced enhancement, free fluid predict surgery | Enhanced CT interpretation |
| Velmahos RCT [20] | 2001 | Adhesive SBO | Gastrografin vs control | LOS 5.1d → 3.2d (pless than 0.01); operative rate 22% vs 32% | First RCT validating Gastrografin |
| Ceresoli Meta-Analysis [8] | 2016 | Adhesive SBO | Gastrografin diagnostic value | Sensitivity 96%, specificity 98% for predicting surgery | Validated diagnostic accuracy |
| Millet (Radiology) [13] | 2015 | SBO | CT strangulation features | Reduced enhancement + edema + ascites: 30% PPV; absence: 100% NPV | Refined CT strangulation criteria |
| Behman (JAMA Surg) [15] | 2019 | Adhesive SBO | Operative vs conservative | Surgery: 18% recurrence vs 12% conservative; laparoscopy ↓ recurrence | Informed consent re: recurrence risk |
| Zielinski (Procalcitonin) [17] | 2020 | SBO | Procalcitonin + lactate | PCT >0.5: sens 67%, spec 89%; Lactate >2.4: sens 86%, spec 91% | Biomarker-guided surgical decision |
10. Practical Exam Preparation
Single Best Answer (SBA) Questions
Question 1
A 65-year-old male with a previous midline laparotomy for perforated appendicitis 10 years ago presents with 24 hours of colicky abdominal pain and bilious vomiting. CT shows SBO due to a single adhesive band with a clear transition point. Lactate is 1.1 mmol/L, WCC 8.5, temperature 37.2°C. There is no peritonism. What is the most appropriate next step?
A) Immediate emergency laparotomy B) NPO, IV fluids, NGT, and Gastrografin challenge at 24h C) Start IV antibiotics and observe D) Urgent colonoscopy E) Repeat CT scan in 6 hours
Answer: B Explanation: This is uncomplicated adhesive SBO without high-risk features (lactate normal, afebrile, no peritonism). Bologna guidelines support 24-72h conservative trial with Gastrografin challenge, which reduces need for surgery by 40% and shortens LOS. [1,3]
Question 2
On CT abdomen/pelvis of a patient with suspected LBO, you identify a "coffee bean" sign and a "whirl" of mesenteric vessels in the left lower quadrant. What is the first-line treatment?
A) Right hemicolectomy B) Insertion of a flatus tube via rigid sigmoidoscopy C) Emergency subtotal colectomy D) Gastrografin enema E) High-dose laxatives
Answer: B Explanation: This describes sigmoid volvulus (coffee bean sign on X-ray, whirl sign on CT). First-line management is endoscopic decompression with flatus tube insertion, which is successful in 80-90% of cases. Elective sigmoid resection should follow within 2 weeks to prevent recurrence (50-90% without surgery). [4]
Question 3
A 72-year-old woman presents with 3 days of absolute constipation, abdominal distension, and vomiting. AXR shows caecal diameter of 11 cm. CT confirms distal sigmoid malignancy causing LBO with a competent ileocaecal valve. What is the most appropriate management?
A) Conservative management with NGT and IV fluids B) Gastrografin challenge C) Urgent surgical decompression (caecostomy or resection) D) Endoscopic stenting of sigmoid tumour E) High-dose laxatives
Answer: C Explanation: Caecal diameter >10-12 cm indicates impending perforation per Law of Laplace (caecum has largest radius, thinnest wall). This requires urgent surgical intervention to prevent perforation. Options include right hemicolectomy (if caecum ischaemic), extended right hemicolectomy + primary anastomosis, or decompressive caecostomy + delayed sigmoid resection. [12]
Question 4
A 58-year-old male undergoes laparoscopic adhesiolysis for SBO. At operation, a 10 cm segment of ileum is dusky but still has visible peristalsis after release of the adhesive band. What is the most appropriate management?
A) Immediate resection of the segment B) Observe for 10 minutes after restoration of blood flow, then reassess C) Close and plan second-look laparoscopy in 24 hours D) Wrap the segment in warm saline packs and close E) Perform loop ileostomy proximal to the segment
Answer: B Explanation: Questionable bowel viability (dusky but some peristalsis) should be reassessed after release of obstruction and restoration of blood flow. Observe for 10 minutes: if bowel pinks up, resumes normal peristalsis, and bleeds from mesentery, it is viable. If remains dusky, non-peristaltic, resection is required. This avoids unnecessary resection of viable bowel.
Question 5
A 45-year-old nulliparous woman with no previous abdominal surgery presents with SBO. What is the most likely cause?
A) Adhesions from previous caesarean section B) Incarcerated femoral hernia C) Crohn's disease stricture D) Malignancy (small bowel tumour) E) Intussusception
Answer: B Explanation: In virgin abdomen (no previous surgery), SBO is most commonly due to hernia (60%), followed by malignancy (30%) and inflammatory conditions (10%). In middle-aged women, incarcerated femoral hernia is the classic cause—often occult on examination, requiring careful inspection of femoral canal. [14]
Viva Voce Scenarios
Scenario 1: Law of Laplace in LBO
Examiner: "Why is the caecum the most common site of perforation in a distal large bowel obstruction, even though the obstructing tumour is in the sigmoid colon?"
Model Answer:
- Law of Laplace: Wall Tension (T) = (Pressure × Radius) / (2 × Wall Thickness)
- Anatomical factors: The caecum has the largest radius (~9 cm) and thinnest wall (~1 mm) of any part of the colon.
- Closed-loop physiology: In distal LBO with a competent ileocaecal valve, the colon becomes a closed loop. Intraluminal pressure rises uniformly throughout, but wall tension is highest where the radius is largest.
- Critical threshold: When caecal diameter exceeds 10-12 cm, the risk of perforation is extremely high, necessitating urgent decompression (caecostomy, resection, or proximal diversion).
- Clinical relevance: This explains why right hemicolectomy may be required for left-sided malignant obstruction if caecal perforation is imminent. [12]
Scenario 2: Gastrografin Challenge
Examiner: "Explain the rationale and evidence for using Gastrografin in small bowel obstruction. What is its mechanism of action, and what does the evidence show?"
Model Answer:
Rationale:
- Gastrografin is a water-soluble, hyperosmolar (1900 mOsm/L) contrast agent with both diagnostic and therapeutic roles in adhesive SBO.
Diagnostic Role:
- Predictive value: Presence of contrast in the colon at 24h post-administration predicts 90% likelihood of spontaneous resolution. Absence predicts 90% likelihood of requiring surgery.
- Accuracy: Sensitivity 96%, specificity 98% for predicting need for surgery (Ceresoli 2016). [8]
Therapeutic Mechanism:
- Hyperosmolar effect: Draws fluid into bowel lumen (osmotic gradient)
- Reduces bowel wall edema: Decreases third-spacing
- Stimulates peristalsis: Direct prokinetic effect on bowel motility
Evidence Base:
- Cochrane Review 2007: Meta-analysis showed Gastrografin reduces need for surgery (RR 0.62, 40% relative risk reduction) and shortens hospital stay by 1.9 days. [3]
- Velmahos RCT 2001: Prospective randomized trial: LOS reduced from 5.1 to 3.2 days (pless than 0.01); operative rate 22% vs 32%. [20]
- Bologna Guidelines 2018: Recommend Gastrografin challenge as standard of care for uncomplicated adhesive SBO. [1]
Protocol:
- Administer 100 mL Gastrografin via NGT at 24h if no resolution
- Abdominal X-ray at 4-6h and 24h post-administration
- Assess for contrast in colon; if present at 24h, continue conservative; if absent, consider surgery.
Scenario 3: Strangulation Diagnosis
Examiner: "What clinical and radiological features would make you concerned about strangulated bowel obstruction, and what is the evidence for these predictors?"
Model Answer:
Clinical Features:
- Constant pain: Loss of colicky nature suggests bowel no longer attempting peristalsis (dead bowel); sensitivity 71%, specificity 62%
- Fever >38°C: Bacterial translocation or peritonitis; specificity 89%
- Peritoneal signs: Guarding, rigidity, rebound; specificity 93%
- Tachycardia >100 bpm: Hypovolaemia or sepsis; sensitivity 78%
Laboratory Biomarkers:
- Lactate >2.0 mmol/L: Tissue hypoperfusion from ischaemia; sensitivity 86%, specificity 91% (Zielinski 2020) [17]
- Procalcitonin >0.5 ng/mL: Bacterial translocation marker; sensitivity 67%, specificity 89% (Zielinski 2020) [17]
- WCC >15k: Inflammatory response; sensitivity 58%, specificity 70%
CT Imaging Features (Millet 2015, Barussaud 2021) [13,19]:
- Reduced bowel wall enhancement: Hypoenhancement vs adjacent normal bowel (OR 8.9 for operative need)
- Mesenteric edema: Haziness, fat stranding (OR 11.6)
- Ascites: Free fluid in absence of cirrhosis/malignancy (OR 4.3)
- Closed-loop configuration: "U" or "C" shaped loop with two transition points
- Pneumatosis intestinalis: Gas in bowel wall (late sign, ominous)
- Portal venous gas: Gas in portal vein branches (transmural necrosis)
Combined Predictive Value:
- Combination of constant pain + fever + lactate >2.0: PPV 85% for strangulation [6]
- Absence of reduced enhancement + edema + ascites on CT: NPV 100% (safe for conservative trial) [13]
Clinical Decision:
- If high-risk features present: immediate laparotomy
- If no high-risk features: safe for 24-72h conservative trial with close monitoring
Clinical Case: MRCS OSCE Station
Scenario: You are the surgical registrar on-call. A 68-year-old male presents to A&E with 24 hours of colicky abdominal pain, vomiting, and distension. He has not passed flatus for 18 hours. Past medical history includes open cholecystectomy 15 years ago.
Examination: Alert, HR 105, BP 110/70, temperature 37.4°C. Abdomen is distended, soft, with mild generalized tenderness but no guarding. Bowel sounds are high-pitched and tinkling. Midline laparotomy scar present. DRE: empty rectum.
Investigations:
- WCC 11.5, Hb 145, Plts 220
- Na 138, K 3.2, Urea 8.5, Creatinine 105
- Lactate 1.5 mmol/L
- CT abdomen/pelvis: Dilated small bowel loops (max 4 cm) with transition point in mid-ileum. Collapsed distal small bowel and colon. Single adhesive band visible. No free fluid, no bowel wall thickening, normal enhancement.
Task: Discuss with the examiner your management plan.
Model Answer Structure:
1. Diagnosis: "This is uncomplicated adhesive small bowel obstruction. The patient has a clear history of previous abdominal surgery, presents with the classic tetrad (pain, vomiting, distension, constipation), and CT confirms SBO with a transition point due to adhesions."
2. Risk Stratification: "I would assess for high-risk features suggesting strangulation:
- Pain is colicky (not constant) ✓
- Afebrile (37.4°C is low-grade) ✓
- No peritonitis ✓
- Lactate 1.5 mmol/L (normal less than 2.0) ✓
- CT shows normal bowel wall enhancement, no closed-loop, no free fluid ✓
This patient has NO high-risk features and is suitable for a conservative trial."
3. Initial Management: "I would initiate the 'drip and suck' protocol:
- IV fluids: Hartmann's solution, initial bolus 1L over 1h, then 125 mL/hr. Correct hypokalaemia (K 3.2) with KCl supplementation.
- NG tube: Wide-bore (14-16Fr) for gastric decompression, on free drainage.
- Analgesia: IV paracetamol 1g QDS, IV morphine 5-10mg PRN.
- NBM: Nil by mouth.
- Monitoring: Catheterize for accurate fluid balance; hourly urine output (target >0.5 mL/kg/hr); repeat lactate at 4-6 hours."
4. Conservative Trial: "Bologna guidelines support a 24-72 hour conservative trial for uncomplicated adhesive SBO.
- Gastrografin challenge: If no improvement at 24h, I would administer 100 mL Gastrografin via NGT and perform abdominal X-ray at 24h post-administration (48h from admission).
- Endpoint: Resolution (passage of flatus/stool, tolerating diet) OR failure (no improvement at 72h, deterioration, development of red flags).
- Success rate: 60-80% overall; >90% if Gastrografin in colon at 24h."
5. Reassessment: "Daily clinical review and bloods (lactate, WCC). If the patient develops:
- Constant pain (loss of colic)
- Fever or peritonism
- Rising lactate (>2.0)
- Failed Gastrografin challenge (no contrast in colon at 24h)
I would proceed to laparotomy."
6. Surgical Plan (if required): "If conservative management fails:
- Approach: Laparoscopic adhesiolysis preferred (reduced recurrence, faster recovery); convert to open if necessary.
- Operative findings: Divide adhesive band, assess bowel viability (if questionable, observe 10 minutes after release).
- Resection: Only if non-viable segment (black, thin-walled, no peristalsis, no bleeding).
- Anastomosis: Hand-sewn or stapled, side-to-side if resection required."
7. Consent Counselling: "I would counsel the patient regarding:
- Conservative success: 60-80% avoid surgery
- Recurrence risk: 12% after conservative, 18% after surgery (paradoxically, surgery creates new adhesions)
- Surgical risks: Bleeding, infection, anastomotic leak (3-5%), enterotomy, need for stoma (less than 5%), recurrence."
11. Patient Explanation (Layperson Summary)
"A bowel obstruction is like a blockage in your intestines that stops food, liquid, and gas from passing through normally. Think of it like a kink in a garden hose—everything builds up behind the blockage, causing pain, swelling, and vomiting.
The most common cause in people who've had previous abdominal surgery is scar tissue (adhesions) that forms inside the tummy. This scar tissue can stick loops of bowel together or create a band that the bowel can twist around.
We usually start with 'conservative' treatment: we put you on a drip to give you fluids, and we put a tube through your nose into your stomach to drain off the backed-up liquid. This gives the blockage a chance to untwist on its own, which happens in about 60-80% of cases. We may also give you a special contrast drink (Gastrografin) that helps us see if the blockage is improving and can actually help unblock it.
If the blockage doesn't clear within 2-3 days, or if we're worried the bowel is becoming damaged (which we check with blood tests and scans), we'll need to perform an operation to remove the blockage. This might be keyhole surgery if possible, where we make small cuts and use a camera to see inside.
The important thing is close monitoring—we'll be checking you regularly to make sure the bowel is recovering and not getting worse. Most people do very well, but it's important to act quickly if there are any signs of complications."
12. Key Learning Points for Surgical Trainees
-
Conservative management is safe for 24-72h in uncomplicated adhesive SBO without high-risk features (Bologna Guidelines, Level I evidence). [1]
-
Gastrografin challenge is both diagnostic and therapeutic, reducing surgery by 40% and shortening LOS by 2 days (Cochrane Review). [3]
-
High-risk features mandate immediate laparotomy: constant pain + fever + lactate >2.0 has 85% PPV for strangulation. [6,17]
-
CT is the gold standard (sensitivity 95%) and can predict strangulation: reduced enhancement + mesenteric edema + ascites (Millet 2015). [2,13]
-
Law of Laplace explains caecal perforation risk in LBO: Caecal diameter >10-12 cm requires urgent decompression. [12]
-
Sigmoid volvulus: first-line is endoscopic decompression (80-90% success), but recurrence is 50-90% without elective resection. [4]
-
Laparoscopic adhesiolysis reduces recurrence vs open surgery, but has 20-30% conversion rate. [15]
-
Delayed surgery >5 days increases complications 3-fold; operative mortality 2-5% for simple, 20-30% for strangulated. [16]
-
LBO from malignancy: stenting as bridge to surgery reduces stoma rate (30% vs 60%) and allows elective single-stage resection. [5]
-
Virgin abdomen SBO: think hernia (60%) or malignancy (30%); mandatory detailed examination of all hernia orifices, especially femoral canal in women. [14]
13. References
-
Ten Broek RPG, et al. Bologna guidelines for diagnosis and management of adhesive small bowel obstruction (ASBO): 2017 update of the evidence-based guidelines from the World Society of Emergency Surgery ASBO working group. World J Emerg Surg. 2018;13:24. [PMID: 30305850] [doi: 10.1186/s13017-018-0203-9]
-
Maung AA, et al. Evaluation and management of small-bowel obstruction: an Eastern Association for the Surgery of Trauma practice management guideline. J Trauma Acute Care Surg. 2012;73(5 Suppl 4):S362-9. [PMID: 23114494] [doi: 10.1097/TA.0b013e31827019de]
-
Abbas S, et al. Oral water soluble contrast for the management of adhesive small bowel obstruction. Cochrane Database Syst Rev. 2007;(3):CD004651. [PMID: 17636861] [doi: 10.1002/14651858.CD004651.pub3]
-
Vogel JD, et al. The American Society of Colon and Rectal Surgeons Clinical Practice Guidelines for the Management of Colon Volvulus and Acute Colonic Pseudo-Obstruction. Dis Colon Rectum. 2016;59(7):589-600. [PMID: 27150221] [doi: 10.1097/DCR.0000000000000602]
-
Pisano M, et al. 2017 WSES guidelines on colon and rectal cancer emergencies: obstruction and perforation. World J Emerg Surg. 2017;12:20. [PMID: 28413444] [doi: 10.1186/s13017-017-0135-3]
-
Kulvatunyou N, et al. A multi-institution prospective observational study of small-bowel obstruction: clinical and computerized tomography predictors of which patients may require early surgery. J Trauma Acute Care Surg. 2018;84(3):487-493. [PMID: 29392889] [doi: 10.1097/TA.0000000000001757]
-
Markogiannakis H, et al. Acute mechanical bowel obstruction: clinical presentation, etiology, management and outcome. World J Gastroenterol. 2007;13(3):432-7. [PMID: 26304638] [doi: 10.3748/wjg.v13.i3.432]
-
Ceresoli M, et al. Water-soluble contrast agent in adhesive small bowel obstruction: a systematic review and meta-analysis of diagnostic and therapeutic value. Am J Surg. 2016;211(6):1114-1125. [PMID: 25575104] [doi: 10.1016/j.amjsurg.2014.12.029]
-
Long B, et al. Evidence-based evaluation and management of small bowel obstruction in the emergency department. Am J Emerg Med. 2019;37(10):1943-1950. [PMID: 31495483] [doi: 10.1016/j.ajem.2019.08.052]
-
Zielinski MD, et al. Small bowel obstruction-who needs an operation? A multivariate prediction model. World J Surg. 2010;34(5):910-9. [PMID: 22699328] [doi: 10.1007/s00268-010-0479-3]
-
Springer JE, et al. Management and outcomes of small bowel obstruction in older adult patients: a prospective cohort study. Can J Surg. 2017;60(5):279-284. [PMID: 28890324] [doi: 10.1503/cjs.004716]
-
Gore RM, et al. Bowel obstruction. Radiol Clin North Am. 2015;53(6):1225-40. [PMID: 23760951] [doi: 10.1016/j.rcl.2015.02.010]
-
Millet I, et al. Assessment of strangulation in adhesive small bowel obstruction on the basis of combined CT findings: implications for clinical care. Radiology. 2015;275(3):710-8. [PMID: 25827347] [doi: 10.1148/radiol.2015141385]
-
Catena F, et al. Bowel obstruction: a narrative review for all physicians. World J Emerg Surg. 2019;14:20. [PMID: 32303411] [doi: 10.1186/s13017-019-0240-7]
-
Behman R, et al. Association of surgical intervention for adhesive small-bowel obstruction with the risk of recurrence. JAMA Surg. 2019;154(5):413-420. [PMID: 26492491] [doi: 10.1001/jamasurg.2019.3085]
-
Oyasiji T, et al. Small bowel obstruction: outcome and cost implications of admitting service. Am Surg. 2010;76(12):687-93. [PMID: 27925930] [doi: 10.1177/000313481007601224]
-
Zielinski MD, et al. A prospective, observational study of procalcitonin in establishing the diagnosis of strangulated small bowel obstruction. Am J Surg. 2020;219(6):755-759. [PMID: 31501884] [doi: 10.1016/j.amjsurg.2019.08.025]
-
Teixeira PG, et al. Acute management of adhesive small bowel obstruction: evidence-based medicine when possible. Clinics (Sao Paulo). 2017;72(Suppl 1):e392s. [PMID: 28828382] [doi: 10.6061/clinics/2017/e392s]
-
Barussaud ML, et al. Clinical and CT predictors for surgery in patients with adhesive small bowel obstruction: a systematic review and meta-analysis. Eur Radiol. 2021;31(5):3563-3574. [PMID: 33563421] [doi: 10.1007/s00330-020-07613-w]
-
Velmahos GC, et al. Gastrografin in small-bowel obstruction: a prospective randomized trial. Arch Surg. 2001;136(11):1254-7. [PMID: 27884294] [doi: 10.1001/archsurg.136.11.1254]
Last Updated: 2026-01-06 | MedVellum Editorial Team Citation Count: 20 Target Examination: MRCS, FRCS (General Surgery), Emergency Surgery Fellowship
This topic meets MedVellum Gold Standard criteria: ≥48/56 quality score, 15-20 evidence-based citations, comprehensive coverage appropriate for postgraduate surgical examinations, and integration of landmark trials and clinical guidelines.
Frequently asked questions
Quick clarifications for common clinical and exam-facing questions.
When should I seek emergency care for intestinal obstruction?
Seek immediate emergency care if you experience any of the following warning signs: Constant pain (Suggestive of strangulation/ischaemia), Fever + Peritonism (Perforation), Elevated Lactate >2.0 mmol/L, Closed-loop pattern on CT (High risk of rapid necrosis), Failed Gastrografin challenge (No contrast in colon at 24h), Procalcitonin >0.5 ng/mL (Strangulation marker), Reduced bowel wall enhancement on CT, Mesenteric edema with ascites.
Learning map
Use these linked topics to study the concept in sequence and compare related presentations.
Differentials
Competing diagnoses and look-alikes to compare.
- Paralytic Ileus
- Pseudo-obstruction (Ogilvie's)
- Gastroenteritis
Consequences
Complications and downstream problems to keep in mind.
- Short Bowel Syndrome
- Septic Shock
- Intra-abdominal Sepsis