Bronchiectasis
Comprehensive evidence-based guide to non-cystic fibrosis bronchiectasis covering pathophysiology, etiology, diagnosis with HRCT findings, microbiology including Pseudomonas aeruginosa and NTM, airway clearance...
What matters first
Comprehensive evidence-based guide to non-cystic fibrosis bronchiectasis covering pathophysiology, etiology, diagnosis with HRCT findings, microbiology including Pseudomonas aeruginosa and NTM, airway clearance...
Check the red flags, emergency triggers, and escalation points before using the deep-dive material.
9 Jan 2026
Generated educational material; verify before clinical use.
Visible references section
See the concept before reading it
Study the key anatomy, imaging, and decision pathways as full teaching plates.
Content status and exam context
This page is AI-generated educational content. It may contain errors or omissions and is not a substitute for current guidelines, local protocols, senior clinical judgement, or professional medical advice.
MedVellum does not claim an individual clinician reviewer, board certification, or professional credential for this page unless a future version names a real, verifiable reviewer.
Clinical explanation and evidence
Bronchiectasis
Quick Reference
Critical Alerts
- Massive hemoptysis (> 200 mL/24h) is life-threatening: Requires emergent bronchial artery embolization and ICU admission
- Pseudomonas aeruginosa colonization significantly worsens prognosis: Associated with 3-fold increased mortality and accelerated lung function decline [1]
- Always obtain sputum culture during exacerbations: Prior culture results guide empiric antibiotic selection
- 14-day antibiotic courses are standard for exacerbations: Not 5-7 days as in simple respiratory infections
- Non-tuberculous mycobacteria (NTM) must be considered: If failing standard treatment, obtain AFB smear and culture
- Chronic hypercapnia common: Target SpO2 88-92% to avoid suppressing respiratory drive
- Airway clearance is as important as antibiotics: Daily physiotherapy is cornerstone of management
- Screen for underlying causes in all new diagnoses: Immunoglobulin levels, Aspergillus serology, CF genotype in younger patients
Classic Presentation
| Feature | Description |
|---|---|
| Age | Any age; prevalence increases with age, peak > 75 years |
| Chronic productive cough | Daily sputum production, often purulent, 50-500 mL/day |
| Recurrent respiratory infections | Multiple antibiotic courses per year (> 3 exacerbations/year is frequent exacerbator) |
| Hemoptysis | 30-50% of patients; usually streaky, can be massive |
| Dyspnea | Progressive, worse with exacerbations; MRC grade 3-4 common |
| Fatigue | Chronic, related to infection and systemic inflammation |
| Pattern | Symptoms stable between exacerbations; baseline may deteriorate over years |
Emergency Treatments
| Scenario | Immediate Action | Notes |
|---|---|---|
| Acute exacerbation | Empiric antibiotics based on prior cultures | 14-day course; cover Pseudomonas if colonized |
| Massive hemoptysis (> 200 mL/24h) | Position bleeding lung down, tranexamic acid 1g IV, call interventional radiology | Bronchial artery embolization is first-line |
| Respiratory failure | NIV if hypercapnic; target SpO2 88-92% | ICU referral if pH less than 7.25 |
| Sepsis | IV broad-spectrum antibiotics, fluid resuscitation | Cover Pseudomonas in colonized patients |
Definition
Overview
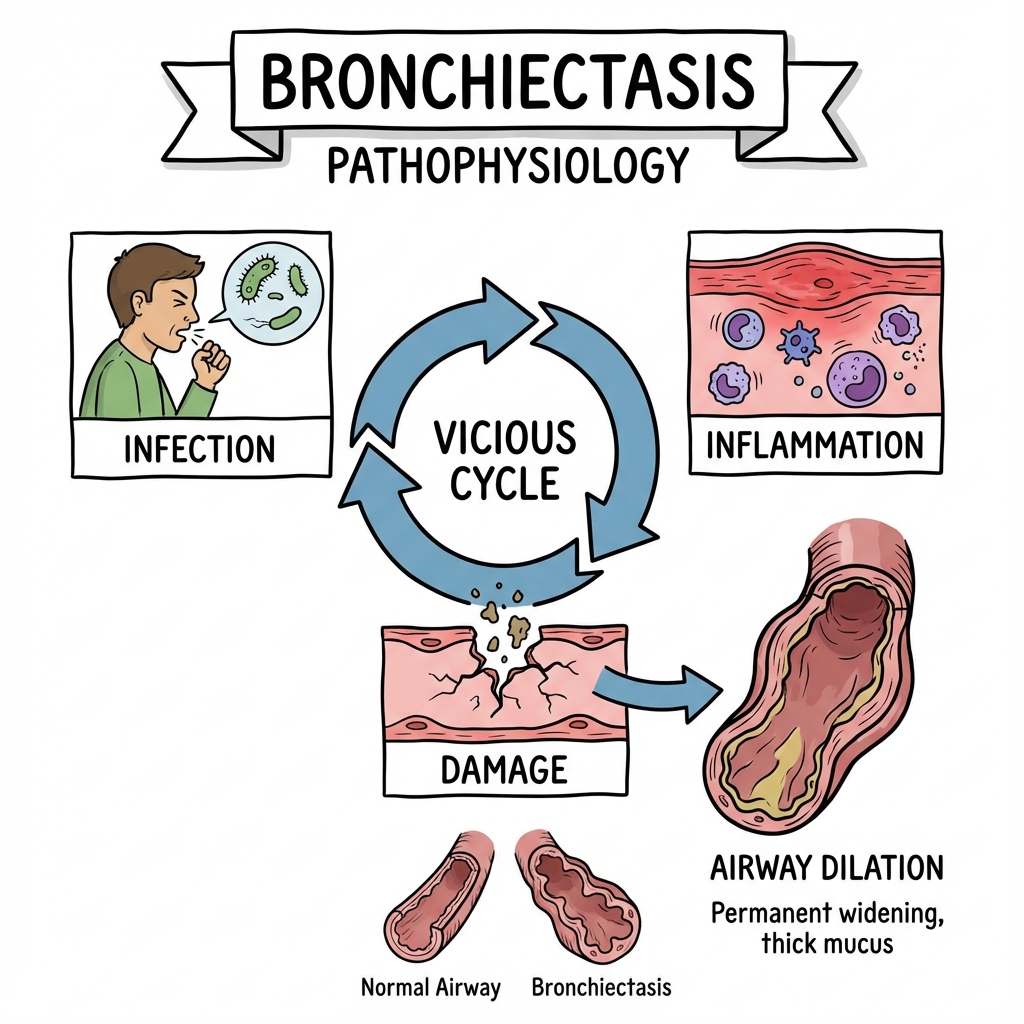
Bronchiectasis is a chronic respiratory disease characterized by permanent, abnormal dilation of the bronchi (> 2 mm diameter airways) with destruction of bronchial wall elastic and muscular components, impaired mucociliary clearance, chronic bacterial colonization, and persistent inflammation [1,2]. It is defined radiologically as bronchial dilatation with broncho-arterial ratio > 1.0 (internal airway diameter exceeding adjacent pulmonary artery), lack of normal bronchial tapering, and visibility of airways within 1 cm of pleural surface [3].
Non-cystic fibrosis (non-CF) bronchiectasis represents a heterogeneous syndrome arising from diverse etiologies including post-infectious damage, immunodeficiency states, allergic bronchopulmonary aspergillosis (ABPA), autoimmune conditions, and chronic aspiration. Unlike cystic fibrosis, which results from a specific genetic defect in the CFTR chloride channel, non-CF bronchiectasis represents the end-stage pathological consequence of various insults to airway structure and function [4].
The hallmark clinical features are chronic productive cough with daily sputum production, recurrent respiratory infections requiring multiple antibiotic courses annually, and progressive decline in lung function. The disease follows a characteristic "vicious cycle" of infection, inflammation, impaired clearance, and structural damage, which perpetuates itself even after the initial insult has resolved [5].
Classification
Morphological Classification (Reid Classification) [6]:
| Type | Description | CT Appearance | Clinical Significance |
|---|---|---|---|
| Cylindrical (tubular) | Uniform bronchial dilation with smooth walls | Tram-track sign (longitudinal), Signet ring sign (cross-section) | Most common type (65%); earliest and mildest form |
| Varicose | Irregular dilation with alternating constrictions | Beaded or varicose vein appearance | Intermediate severity; often progression from cylindrical |
| Cystic (saccular) | Severe balloon-like dilation with sac formation | Grape-like clusters, may have air-fluid levels | Most severe form; end-stage disease; highest complication risk |
By Etiology [7]:
| Category | Specific Causes | Prevalence |
|---|---|---|
| Post-infectious | Childhood pneumonia, tuberculosis, pertussis, measles, adenovirus, influenza | 20-30% |
| Immunodeficiency | Common variable immunodeficiency (CVID), IgA deficiency, IgG subclass deficiency, HIV | 5-10% |
| Genetic/Congenital | Cystic fibrosis, primary ciliary dyskinesia (PCD), alpha-1 antitrypsin deficiency, Williams-Campbell syndrome | 5-10% |
| Allergic | Allergic bronchopulmonary aspergillosis (ABPA) | 5-10% |
| Autoimmune/Inflammatory | Rheumatoid arthritis, Sjogren's syndrome, inflammatory bowel disease, systemic lupus erythematosus | 5-10% |
| Aspiration | Gastroesophageal reflux disease (GERD), neuromuscular disease, esophageal disorders | 3-5% |
| Obstruction | Foreign body, endobronchial tumor, extrinsic compression | 2-5% |
| COPD-associated | Chronic obstructive pulmonary disease with bronchiectasis overlap | 15-30% |
| Idiopathic | No identifiable cause despite investigation | 30-50% |
By Severity - Bronchiectasis Severity Index (BSI) [8]:
| Variable | Points |
|---|---|
| Age 50-69 | 2 |
| Age 70-79 | 4 |
| Age ≥80 | 6 |
| BMI less than 18.5 | 2 |
| FEV1 % predicted 50-80% | 1 |
| FEV1 % predicted 30-49% | 2 |
| FEV1 % predicted less than 30% | 3 |
| Hospital admission previous 2 years | 5 |
| Exacerbations previous year ≥3 | 2 |
| MRC dyspnea score 4 | 2 |
| MRC dyspnea score 5 | 3 |
| Pseudomonas colonization | 3 |
| Colonization with other pathogen | 1 |
| ≥3 lobes affected on CT | 1 |
BSI Score Interpretation:
- Mild (0-4): 4-year mortality 5-8%
- Moderate (5-8): 4-year mortality 12-16%
- Severe (≥9): 4-year mortality 27-35%
Epidemiology
Prevalence and Incidence [9,10]:
- Overall prevalence (UK): 566 per 100,000 population; increasing significantly with age
- Age > 75 years: Prevalence exceeds 1,000 per 100,000
- Incidence (UK): 21-30 per 100,000 person-years, increasing year-on-year
- Global variation: Higher in low-to-middle income countries with high burden of childhood respiratory infections and tuberculosis
- Trend: Rising prevalence attributed to improved survival, increased use of CT imaging, and aging population
Demographics:
- Age: Bimodal distribution - peak in young adults (primary ciliary dyskinesia, CF) and elderly (post-infectious, COPD overlap)
- Sex: Female predominance in non-CF bronchiectasis (1.5-2:1 female:male ratio) [11]
- "Lady Windermere syndrome": Middle lobe/lingular bronchiectasis with NTM in elderly thin women
- "Exception: Post-TB bronchiectasis more common in males"
- Ethnicity: Higher rates in Indigenous populations (Australia, New Zealand, Alaska), likely related to childhood infection burden
Comorbidity Burden [12]:
| Comorbidity | Prevalence | Impact |
|---|---|---|
| COPD | 25-50% | Overlapping pathophysiology; accelerated decline |
| Asthma | 20-40% | Shared inflammation; eosinophilic bronchiectasis phenotype |
| GERD | 30-50% | Aspiration risk; exacerbation trigger |
| Rheumatoid arthritis | 3-10% | Autoimmune mechanism; methotrexate may predispose |
| Cardiovascular disease | 20-30% | Systemic inflammation; shared risk factors |
| Anxiety/Depression | 30-55% | Chronic disease burden; impacts adherence |
| Osteoporosis | 10-20% | Corticosteroids; systemic inflammation |
Pathophysiology
Cole's Vicious Cycle Hypothesis [5,13]
The pathogenesis of bronchiectasis is conceptualized as a self-perpetuating "vicious cycle" of four interconnected processes:
INITIAL INSULT
(Infection, Obstruction, Immune Defect)
|
v
+---> IMPAIRED MUCOCILIARY CLEARANCE < --+
| | |
| v |
| BACTERIAL COLONIZATION |
| (Biofilm formation, persistence) |
| | |
| v |
| CHRONIC INFLAMMATION |
| (Neutrophils, proteases, cytokines) |
| | |
| v |
+---- STRUCTURAL AIRWAY DAMAGE ----------+
(Bronchial wall destruction,
dilation, loss of elasticity)
Stage 1: Initial Insult
- Childhood respiratory infection (pneumonia, pertussis, measles, TB)
- Congenital defects (CF, primary ciliary dyskinesia)
- Immune deficiency (hypogammaglobulinemia)
- Allergic inflammation (ABPA)
- Mechanical obstruction (foreign body, tumor)
Stage 2: Impaired Mucociliary Clearance
- Normal mucociliary escalator clears 1-2 L mucus daily
- Damage to ciliated epithelium reduces clearance velocity
- Mucus stasis creates favorable environment for bacterial growth
- Goblet cell hyperplasia produces excessive, viscid mucus
Stage 3: Bacterial Colonization and Biofilm Formation [14]
- Bacteria colonize stagnant mucus
- Biofilm formation creates protected bacterial communities
- Biofilms are 100-1000x more resistant to antibiotics
- Chronic colonization triggers persistent inflammation
- Pseudomonas aeruginosa particularly forms robust biofilms
Stage 4: Chronic Neutrophilic Inflammation
- Neutrophil influx is the dominant inflammatory response
- Neutrophil elastase, proteinases, and reactive oxygen species released
- Digestion of bronchial wall elastin and collagen
- Pro-inflammatory cytokines (IL-1, IL-6, IL-8, TNF-α) amplify response
- Antiprotease defenses overwhelmed (α1-antitrypsin, SLPI)
Stage 5: Structural Airway Damage
- Destruction of elastic tissue and smooth muscle
- Irreversible bronchial dilation
- Further impairment of mucociliary clearance
- Cycle perpetuates even if initial insult removed
Pathological Features
Macroscopic Findings:
- Dilated bronchi extending to lung periphery
- Bronchial wall thickening (may exceed 2 mm)
- Mucous plugging and purulent secretions
- Peribronchial fibrosis
- Adjacent lung atelectasis or consolidation
Microscopic Findings [15]:
| Compartment | Changes |
|---|---|
| Epithelium | Squamous metaplasia, loss of cilia, goblet cell hyperplasia |
| Basement membrane | Thickening, fragmentation |
| Submucosa | Inflammatory infiltrate (neutrophils, lymphocytes, plasma cells), edema |
| Muscle layer | Atrophy, fragmentation, fibrosis |
| Cartilage | Destruction, calcification |
| Bronchial vessels | Hypertrophy of bronchial arteries (source of hemoptysis) |
Microbiology [16,17]
Common Colonizing Organisms:
| Organism | Prevalence | Clinical Significance |
|---|---|---|
| Haemophilus influenzae | 30-50% | Most common; non-typeable strains; biofilm former |
| Pseudomonas aeruginosa | 15-30% | Associated with worse outcomes; 3x mortality; difficult to eradicate |
| Moraxella catarrhalis | 10-20% | Common, usually less virulent |
| Streptococcus pneumoniae | 10-20% | More common in exacerbations |
| Staphylococcus aureus | 5-10% | Consider in CF, post-viral, ABPA |
| Enterobacteriaceae | 5-10% | More common in elderly, frequent antibiotics |
| Non-tuberculous mycobacteria (NTM) | 5-15% | MAC, M. abscessus; requires specialized treatment |
| Aspergillus species | 5-10% | ABPA, colonization, aspergilloma |
Pseudomonas aeruginosa - Special Considerations [18]:
- Mucoid phenotype develops through alginate biofilm production
- Quorum sensing enables coordinated virulence
- Associated with:
- More frequent exacerbations (2.5 vs 1.5/year)
- Accelerated FEV1 decline (60 mL/year vs 30 mL/year)
- Worse quality of life
- 3-fold increased mortality at 3 years
- Greater healthcare utilization
Non-Tuberculous Mycobacteria (NTM) [19]:
| Species | Features | Treatment Considerations |
|---|---|---|
| M. avium complex (MAC) | Most common (80%); nodular-bronchiectatic in thin elderly women | Macrolide + rifampin + ethambutol; 12 months after culture negative |
| M. abscessus | Most difficult to treat; high resistance | Macrolide + amikacin + imipenem/cefoxitin; often requires IV therapy |
| M. kansasii | Most treatment-responsive | Rifampin-based regimen |
Bronchial Arterial Hypertrophy and Hemoptysis [20]
- Chronic inflammation induces bronchial artery hypertrophy and proliferation
- Bronchial arteries normally supply less than 5% of pulmonary blood flow
- In bronchiectasis, may hypertrophy to 3-4 mm diameter (normal less than 1.5 mm)
- Systemic pressure in fragile, inflamed vessels
- Source of hemoptysis in > 90% of cases (not pulmonary arteries)
- Bronchial artery embolization targets these vessels
Clinical Presentation
Symptoms
Cardinal Symptoms of Bronchiectasis:
| Symptom | Frequency | Characteristics |
|---|---|---|
| Chronic productive cough | > 90% | Daily; worse in morning; sputum 10-200 mL/day (may exceed 500 mL in severe disease) |
| Sputum production | > 90% | Purulent (yellow-green) between exacerbations; three-layered if severe (frothy top, mucoid middle, purulent bottom) |
| Dyspnea | 70-80% | Progressive; MRC grade correlates with severity; worse with exacerbations |
| Hemoptysis | 30-50% | Usually streaky; massive in 2-5%; may be presenting symptom |
| Recurrent infections | Hallmark | ≥3 exacerbations/year defines frequent exacerbator phenotype |
| Fatigue/Malaise | Common | Chronic inflammation; poor sleep from cough; psychological burden |
| Chest pain | 20-30% | Pleuritic; musculoskeletal from coughing |
| Wheeze | 20-40% | Airflow obstruction; overlap with asthma/COPD |
Sputum Characteristics [21]:
- Color: Green/yellow (neutrophilic inflammation); brown (old blood); rust (fresh blood)
- Consistency: Thick, tenacious, difficult to expectorate
- Volume: Increases during exacerbations (may double or triple)
- Odor: Fetid odor suggests anaerobic infection
Exacerbation Definition [1,7]
Acute Exacerbation of Bronchiectasis: Deterioration in THREE or more of the following for at least 48 hours:
- Increased cough frequency
- Increased sputum volume
- Increased sputum purulence (color change)
- Increased dyspnea (breathlessness)
- Increased fatigue/malaise
- Hemoptysis (new or increased)
- Fever (temperature > 38°C)
Exacerbation Frequency Categories:
- Infrequent exacerbator: 0-2 exacerbations/year
- Frequent exacerbator: ≥3 exacerbations/year (associated with worse outcomes)
Physical Examination
General Inspection:
| Finding | Significance |
|---|---|
| Digital clubbing | Present in 30-40%; suggests chronic hypoxia or active suppuration |
| Cachexia/Low BMI | Systemic inflammation; poor prognosis factor |
| Pallor | Anemia of chronic disease |
| Cyanosis | Hypoxemia; severe disease |
| Increased respiratory rate | Active infection or underlying disease severity |
Respiratory Examination:
| Finding | Significance |
|---|---|
| Coarse crackles | Secretions in dilated airways; typically inspiratory; may clear with coughing |
| Wheeze | Airflow obstruction; may indicate overlap with asthma/COPD |
| Reduced breath sounds | Consolidation, severe mucus plugging, or localized disease |
| Increased anteroposterior diameter | Hyperinflation (COPD overlap, air trapping) |
| Transmitted sounds | Large airway secretions |
Cardiovascular Examination:
| Finding | Significance |
|---|---|
| Raised JVP | Cor pulmonale; right heart failure |
| Peripheral edema | Cor pulmonale |
| Loud P2 | Pulmonary hypertension |
| Right ventricular heave | Right ventricular hypertrophy |
Red Flags (Life-Threatening Presentations)
| Red Flag | Concern | Immediate Action |
|---|---|---|
| Massive hemoptysis (> 200 mL/24h) | Bronchial artery rupture | Bleeding lung down, IV access, type and crossmatch, urgent bronchial artery embolization |
| Respiratory failure (SpO2 less than 88% on air) | Severe exacerbation, mucus plugging | ICU referral, NIV consideration if hypercapnic |
| Sepsis (NEWS ≥5) | Overwhelming infection | Sepsis-6 bundle, IV antibiotics, fluid resuscitation |
| Altered consciousness | Hypercapnic encephalopathy, sepsis | ABG, ICU, NIV or intubation |
| Fever unresponsive to antibiotics | Resistant organism, abscess, NTM | CT scan, extended cultures, bronchoscopy |
| Acute hypoxemia | PE, mucus plugging, pneumonia | CTPA if PE suspected, chest physiotherapy |
Massive Hemoptysis Management Protocol
IMMEDIATE ACTIONS (First 10 minutes):
1. Call for senior help + ICU + Interventional Radiology
2. Position patient with bleeding lung DOWN (if known)
3. High-flow oxygen (unless CO2 retainer)
4. Two large-bore IV cannulas
5. Bloods: FBC, clotting, G&S, crossmatch 6 units
6. IV Tranexamic acid 1g over 10 minutes
7. Nebulized adrenaline 1:1000 (1 mg in 5 mL) may reduce bleeding
STABILIZATION:
- Correct coagulopathy (FFP, vitamin K, platelet transfusion)
- CXR to lateralize bleeding (often unhelpful)
- CT angiography if stable (identifies bronchial artery anatomy)
- Prepare for emergency bronchoscopy
DEFINITIVE TREATMENT:
- Bronchial artery embolization (first-line, 90% success)
- Rigid bronchoscopy for airway control if massive
- Surgery (lobectomy/pneumonectomy) if embolization fails
Differential Diagnosis
Conditions Presenting with Chronic Productive Cough
| Diagnosis | Distinguishing Features | Key Investigations |
|---|---|---|
| COPD | Smoking history, progressive dyspnea, emphysema on CT | Spirometry (FEV1/FVC less than 0.7), CT showing emphysema |
| Chronic bronchitis | Smoking history, no bronchial dilation on CT | Normal bronchial architecture on HRCT |
| Asthma | Variable symptoms, wheeze, atopy, reversibility | Spirometry with reversibility, FeNO |
| Cystic fibrosis | Younger onset, pancreatic insufficiency, male infertility | Sweat chloride test, CFTR genotyping |
| Primary ciliary dyskinesia | Situs inversus (50%), sinusitis, otitis, infertility | Nasal NO, ciliary biopsy, genetic testing |
| Tuberculosis | TB contact, upper lobe cavities, systemic symptoms | AFB smear/culture, TB-PCR, Mantoux/IGRA |
| Lung cancer | Hemoptysis, weight loss, smoking history, new symptoms | CT chest, bronchoscopy, PET-CT |
| Interstitial lung disease | Dry cough, fine crackles, restriction on spirometry | HRCT pattern (UIP, NSIP), lung function |
| GERD with aspiration | Heartburn, water brash, nocturnal symptoms | pH study, upper GI endoscopy |
Causes of Hemoptysis
| Diagnosis | Clinical Features | Investigation |
|---|---|---|
| Bronchiectasis | Chronic productive cough, dilated airways on CT | HRCT chest |
| Lung cancer | Weight loss, smoking, new hemoptysis | CT, bronchoscopy |
| Tuberculosis | Fever, night sweats, upper lobe disease | AFB, culture |
| Pulmonary embolism | Sudden onset, pleuritic pain, risk factors | CTPA, D-dimer |
| Pneumonia | Fever, consolidation, rust-colored sputum | CXR, cultures |
| Pulmonary vasculitis | Multisystem involvement, renal disease | ANCA, urinalysis |
| Mitral stenosis | AF, murmur, orthopnea | Echocardiogram |
| AV malformation | Telangiectasia, familial pattern | CTPA, angiography |
Diagnostic Approach
Clinical Assessment
Key History Elements:
- Symptom characterization: Cough duration, sputum volume/color, hemoptysis, dyspnea
- Exacerbation frequency: Number per year, triggers, hospitalization history
- Childhood infections: Severe pneumonia, whooping cough, measles, TB
- Immune function: Recurrent sinusitis, skin infections, unusual infections
- Allergic history: Asthma, atopy, ABPA
- Autoimmune conditions: RA, Sjogren's, IBD
- GI symptoms: Reflux, dysphagia (aspiration risk)
- Family history: CF, PCD, immunodeficiency
- Smoking status: COPD overlap
- Prior cultures: Known colonizers, sensitivities
Laboratory Investigations
Baseline Tests for All Patients:
| Test | Purpose | Expected Findings |
|---|---|---|
| Full blood count | Infection, anemia | Leukocytosis, neutrophilia; anemia of chronic disease |
| CRP/ESR | Inflammatory markers | Elevated in exacerbation; may be chronically raised |
| Renal function (U&E) | Baseline, aminoglycoside safety | Usually normal |
| Liver function | Comorbidities, alpha-1 antitrypsin | Usually normal |
| Sputum culture | Colonizing organisms, sensitivities | Essential - guides antibiotic selection |
| Sputum AFB smear and culture | NTM screening | Three samples recommended |
Etiological Workup [7,22]:
| Test | Indication | Yield |
|---|---|---|
| Serum immunoglobulins (IgG, IgA, IgM) | All patients | 5-10% identify immunodeficiency |
| IgG subclasses | If recurrent infections with normal total Ig | May identify IgG2/IgG4 deficiency |
| Specific antibody responses | Post-vaccination (pneumococcus, Haemophilus) | Functional antibody deficiency |
| Total IgE + Aspergillus-specific IgE | All patients | ABPA screening |
| Aspergillus precipitins (IgG) | If elevated IgE or eosinophilia | ABPA confirmation |
| Alpha-1 antitrypsin level | COPD overlap, lower lobe bronchiectasis | Deficiency predisposes |
| Rheumatoid factor, anti-CCP | Joint symptoms, systemic features | Rheumatoid arthritis association |
| HIV test | Risk factors, young patient | Immunodeficiency |
| Sweat chloride test | Age less than 40, upper lobe disease, infertility | CF diagnosis |
| Nasal nitric oxide | Chronic sinusitis, situs inversus | Low in PCD (less than 77 nL/min) |
ABPA Diagnostic Criteria [23]:
- Asthma or cystic fibrosis
- Immediate cutaneous reactivity to Aspergillus (skin prick)
- Total IgE > 1000 IU/mL
- Elevated Aspergillus-specific IgE/IgG
- Central bronchiectasis on CT
- Peripheral blood eosinophilia (> 500/μL)
- Aspergillus precipitins positive
Imaging
Chest X-ray:
- May be normal in mild disease
- Tram-track sign: Parallel linear opacities (thickened bronchial walls seen longitudinally)
- Ring shadows: Dilated bronchi seen en face
- Tubular opacities: Mucoid impaction
- Air-fluid levels in dilated airways
- Volume loss in affected lobes
High-Resolution CT (HRCT) - Gold Standard [3,24]:
| Finding | Description | Sensitivity |
|---|---|---|
| Signet ring sign | Bronchus larger than adjacent pulmonary artery (cross-section) | Most specific; broncho-arterial ratio > 1.0 |
| Tram-track sign | Parallel dilated bronchus (longitudinal view) | Highly suggestive |
| Lack of tapering | Bronchi maintain diameter toward periphery | Early sign |
| Bronchi visible in outer 1/3 of lung | Normal bronchi not visible within 1 cm of pleura | Sensitive sign |
| Bronchial wall thickening | Wall thickness > 50% of bronchial diameter | Common but non-specific |
| Tree-in-bud pattern | Small airway mucoid impaction, infection | Suggests active infection |
| Mucus plugging | High-attenuation airway filling | May show air-fluid levels |
| Mosaic attenuation | Patchy air trapping on expiratory imaging | Small airway disease |
CT Distribution Patterns by Etiology:
| Distribution | Associated Etiologies |
|---|---|
| Upper lobe predominant | Cystic fibrosis, ABPA, post-TB |
| Middle lobe/lingula | NTM (Lady Windermere), aspiration |
| Lower lobe predominant | Recurrent aspiration, immunodeficiency, post-pneumonic |
| Central bronchiectasis | ABPA (pathognomonic) |
| Unilateral/localized | Post-obstructive (tumor, foreign body), post-pneumonic |
| Bilateral diffuse | PCD, immunodeficiency, idiopathic |
Pulmonary Function Tests [25]
Spirometry:
- Obstructive pattern typical (FEV1/FVC less than 0.7)
- Mixed obstructive-restrictive in advanced disease
- FEV1 decline: 50-60 mL/year (accelerated with Pseudomonas)
| Severity (FEV1% predicted) | Classification |
|---|---|
| ≥80% | Mild |
| 50-79% | Moderate |
| 30-49% | Severe |
| less than 30% | Very severe |
Additional Tests:
- Static lung volumes: Increased RV/TLC ratio (air trapping)
- Gas transfer (DLCO): Usually preserved (differentiates from emphysema)
- 6-minute walk test: Functional capacity assessment
Bronchoscopy
Indications:
- Localized bronchiectasis (exclude obstruction)
- Hemoptysis localization
- Microbiological sampling when sputum unavailable
- Suspected foreign body
- Therapeutic mucus clearance
Treatment
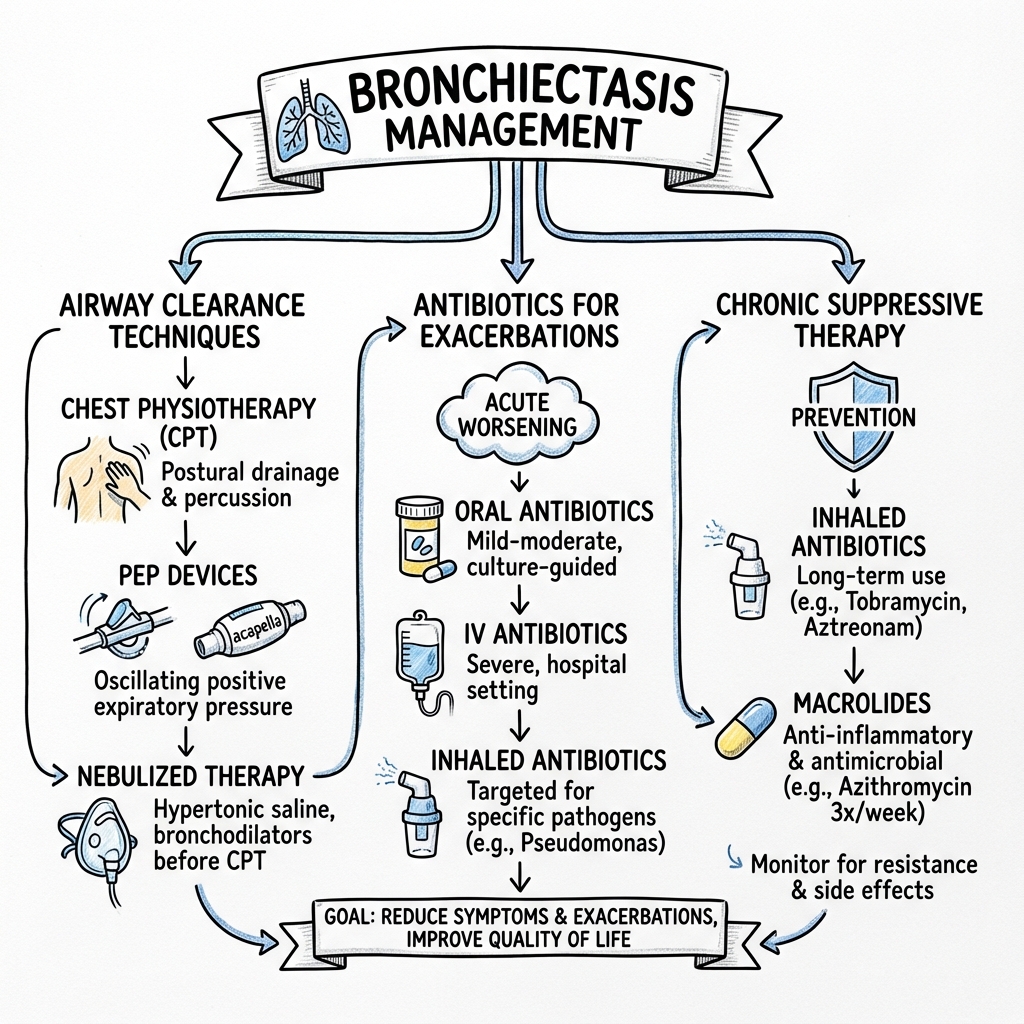
Principles of Management
Bronchiectasis management follows a multimodal approach [1,7]:
- Treat underlying cause (if identified and treatable)
- Airway clearance techniques (cornerstone of daily management)
- Control chronic infection (suppress colonization, treat exacerbations)
- Reduce inflammation (macrolides for anti-inflammatory effect)
- Treat exacerbations promptly (14-day antibiotic courses)
- Manage hemoptysis (tranexamic acid, embolization)
- Surgery (selected localized disease or massive hemoptysis)
- Optimize general health (nutrition, vaccination, pulmonary rehabilitation)
Airway Clearance Techniques [26,27]
Cornerstone of Management: Daily airway clearance is as important as antibiotics and reduces exacerbation frequency.
Manual Techniques:
| Technique | Description | Notes |
|---|---|---|
| Active Cycle of Breathing Technique (ACBT) | Breathing control → thoracic expansion → forced expiration technique (huff) | Most evidence-based; can be self-administered |
| Autogenic drainage | Controlled breathing at varying lung volumes | Requires training; effective in motivated patients |
| Postural drainage | Gravity-assisted positioning for each lobe | Combined with percussion/vibration; time-consuming |
| Percussion and vibration | Manual chest wall oscillation | Adjunct to postural drainage |
Device-Assisted Techniques:
| Device | Mechanism | Evidence |
|---|---|---|
| Oscillating PEP (Flutter, Acapella) | Creates oscillating positive expiratory pressure | Shears mucus from walls; good evidence |
| PEP mask | Positive expiratory pressure | Prevents airway collapse; aids collateral ventilation |
| High-frequency chest wall oscillation (vest) | External thoracic oscillation | Expensive; evidence mixed in non-CF |
| Intrapulmonary percussive ventilation | Delivers high-frequency mini-bursts | Hospital-based; useful in severe disease |
Mucoactive Agents:
| Agent | Mechanism | Evidence | Notes |
|---|---|---|---|
| Nebulized hypertonic saline (6-7%) | Increases airway surface liquid, improves mucociliary clearance | RCT evidence for reduced exacerbations | Pre-treat with bronchodilator; may cause bronchospasm |
| Nebulized isotonic saline (0.9%) | Hydration of secretions | Modest benefit | Better tolerated than hypertonic |
| Nebulized mannitol | Osmotic agent | Limited evidence in non-CF | Alternative if hypertonic saline not tolerated |
| Dornase alfa (rhDNase) | Cleaves DNA in sputum | Contraindicated in non-CF bronchiectasis | May worsen outcomes in non-CF |
Practical Airway Clearance Regimen:
- Bronchodilator (if prescribed) - 15 minutes prior
- Nebulized hypertonic saline (if tolerated)
- Airway clearance technique (ACBT, oscillating PEP)
- Huff and cough to expectorate
- Repeat 2-3 times daily (increase during exacerbations)
Long-Term Antibiotic Therapy
Indications for Long-Term Macrolide Therapy [28,29]:
- ≥3 exacerbations per year
- Exacerbations requiring hospitalization
- Significant impact on quality of life despite optimized treatment
- Pseudomonas colonization (consider nebulized anti-Pseudomonal as alternative)
Macrolide Regimen:
| Drug | Dose | Frequency | Notes |
|---|---|---|---|
| Azithromycin | 250 mg or 500 mg | Three times weekly (Mon/Wed/Fri) OR 250 mg daily | Most evidence; EMBRACE, BAT, BLESS trials |
| Erythromycin | 250 mg | Twice daily | Alternative if azithromycin unavailable |
Evidence (EMBRACE, BAT, BLESS trials) [30]:
- 35-50% reduction in exacerbation frequency
- Improvement in quality of life
- Anti-inflammatory effect (not just antimicrobial)
- Reduces neutrophilic inflammation
Essential Pre-Treatment Workup:
- ECG: Exclude prolonged QTc (> 450 ms) - macrolides prolong QT
- Sputum for AFB: Exclude NTM (macrolide monotherapy promotes resistance)
- Hearing assessment: Ototoxicity risk (especially with aminoglycosides)
- LFTs: Hepatotoxicity monitoring
Monitoring on Long-Term Macrolides:
- Sputum culture every 6-12 months (monitor for macrolide-resistant organisms)
- Annual audiometry
- Annual ECG
- LFTs if symptomatic
Nebulized Antibiotic Therapy (Pseudomonas-colonized patients) [31]:
| Drug | Dose | Frequency | Notes |
|---|---|---|---|
| Colistimethate sodium | 1-2 million units | Twice daily | Well-tolerated; first-line in Europe |
| Tobramycin | 300 mg | Twice daily (28 days on/28 days off) | Alternating cycles reduce resistance |
| Gentamicin | 80 mg | Twice daily | Alternative; nephrotoxicity monitoring |
| Aztreonam lysine | 75 mg | Three times daily (28 days on/28 days off) | Good anti-Pseudomonal activity |
Eradication Therapy for New Pseudomonas Isolation [1]:
- Attempt eradication at first isolation
- Two weeks IV anti-Pseudomonal antibiotics (e.g., ceftazidime + tobramycin) OR
- Oral ciprofloxacin 750 mg BD for 2-4 weeks PLUS nebulized colistin for 3 months
- Success rate: 50-80% if attempted early
Exacerbation Management [7,32]
Antibiotic Selection:
| Prior Colonization | First-Line Empiric | Duration |
|---|---|---|
| No prior cultures available | Amoxicillin-clavulanate 625 mg TDS or Doxycycline 100 mg BD | 14 days |
| H. influenzae | Amoxicillin-clavulanate 625 mg TDS or Co-amoxiclav | 14 days |
| Pseudomonas aeruginosa (sensitive) | Ciprofloxacin 500-750 mg BD | 14 days |
| Pseudomonas (resistant to quinolones) | IV piperacillin-tazobactam OR ceftazidime + IV aminoglycoside | 14 days |
| MRSA | TMP-SMX (co-trimoxazole) or Doxycycline | 14 days |
| Severe/Hospital admission required | IV anti-Pseudomonal beta-lactam ± aminoglycoside | 14 days |
Key Principles:
- 14 days is standard (not 5-7 days as in simple infections)
- Use prior culture data to guide empiric therapy
- Obtain sputum culture at start of exacerbation
- Increase airway clearance frequency during exacerbation
- Consider IV therapy if oral treatment failing or severe
Hospital Admission Criteria:
- Hypoxia requiring supplemental oxygen (SpO2 less than 92% on air)
- Respiratory distress (RR > 25, accessory muscle use)
- Hemodynamic instability
- Failure of outpatient oral antibiotics
- Unable to manage at home (frailty, social factors)
- Significant hemoptysis
- Need for IV antibiotics
- Comorbidities complicating management
Discharge Criteria:
- Clinically improving (reduced cough, sputum, dyspnea)
- Oxygen at or near baseline
- Tolerating oral antibiotics
- Airway clearance technique reviewed
- Exacerbation action plan in place
- Follow-up arranged (2-4 weeks)
Hemoptysis Management [20,33]
Classification:
| Category | Volume | Management |
|---|---|---|
| Minor | less than 20 mL/day | Treat infection, reassurance, oral tranexamic acid |
| Moderate | 20-200 mL/day | Treat infection, tranexamic acid, close monitoring |
| Massive | > 200 mL/24h or > 100 mL/hour | Emergency: bronchial artery embolization, ICU |
Medical Management:
| Intervention | Details |
|---|---|
| Tranexamic acid (oral) | 500 mg TDS for minor hemoptysis |
| Tranexamic acid (IV) | 1 g over 10 min for moderate-massive |
| Treat underlying infection | Antibiotics for exacerbation |
| Hold anticoagulants | Reverse if INR elevated |
| Correct coagulopathy | FFP, vitamin K, platelets |
| Avoid chest physiotherapy | During active bleeding |
Bronchial Artery Embolization (BAE) [34]:
- First-line definitive treatment for massive hemoptysis
- Performed by interventional radiology
- Identifies and occludes hypertrophied bronchial arteries
- Success rate: 85-95% immediate hemostasis
- Recurrence: 10-30% at 1 year (may need repeat)
- Complications: Chest pain, fever, rarely spinal cord ischemia
Surgical Options:
- Lobectomy or pneumonectomy for localized disease with recurrent massive hemoptysis
- Reserved for failed embolization or localized disease unsuitable for embolization
- High-risk in acute setting; elective surgery preferred if possible
Treating Underlying Causes
| Cause | Treatment | Notes |
|---|---|---|
| Immunodeficiency (CVID, IgG subclass) | IgG replacement therapy (IV or SC) | Reduces infections by 50-80% |
| ABPA | Oral corticosteroids (prednisolone 0.5 mg/kg), itraconazole | May need long-term low-dose steroids |
| Aspiration/GERD | PPI, prokinetics, positioning, fundoplication if severe | Speech therapy assessment |
| Rheumatoid arthritis | DMARDs (methotrexate, biologics) | May predate RA; immunomodulators may predispose |
| NTM infection | Multi-drug regimen for 12+ months | Specialist management essential |
| Obstruction (foreign body, tumor) | Bronchoscopic or surgical removal | May be curative if diagnosed early |
Non-Tuberculous Mycobacteria (NTM) Treatment [19,35]
Diagnosis Criteria (ATS/IDSA):
- Pulmonary symptoms + nodular/cavitary opacities on CT OR bronchiectasis with tree-in-bud
- Positive culture from: 2+ sputum samples OR 1 bronchoscopy sample OR lung biopsy
- Exclusion of other diagnoses
MAC (M. avium complex) Treatment:
| Phase | Regimen | Duration |
|---|---|---|
| Induction | Azithromycin 250 mg daily + Rifampicin 600 mg daily + Ethambutol 15 mg/kg daily | Until culture negative |
| Continuation | Continue same regimen | 12 months after culture negative |
| Total duration | Typically 18-24 months | Relapse common if stopped early |
M. abscessus Treatment:
- Requires IV therapy initially (amikacin + imipenem or cefoxitin)
- Add oral macrolide (if susceptible)
- Treatment duration: 12+ months after culture conversion
- Often incurable; suppressive therapy may be needed
Surgical Management [36]
Indications:
- Localized bronchiectasis with failed medical management
- Recurrent massive hemoptysis despite embolization
- Resectable NTM disease not responding to medical therapy
- Localized disease causing significant morbidity
- Pre-transplant volume reduction
Procedures:
| Procedure | Indication | Notes |
|---|---|---|
| Lobectomy | Single lobe disease | Best outcomes if localized |
| Segmentectomy | Limited disease | Lung-sparing approach |
| Pneumonectomy | Destroyed lung | High morbidity; last resort |
| Lung transplantation | End-stage bilateral disease, FEV1 less than 30% | Consider in young patients |
Surgical Outcomes:
- Complete symptom resolution: 50-80% in selected patients
- Complications: 10-25% (air leak, empyema, respiratory failure)
- Mortality: 1-5% (higher in bilateral disease)
Supportive Care
Vaccination [37]:
| Vaccine | Recommendation |
|---|---|
| Influenza | Annual |
| Pneumococcal (PPV23) | All patients (repeat every 5 years) |
| Pneumococcal (PCV13) | Consider in immunocompromised |
| COVID-19 | All patients; boosters as recommended |
| Pertussis | Consider if not vaccinated |
Pulmonary Rehabilitation:
- Improves exercise capacity and quality of life
- Reduces dyspnea and fatigue
- May reduce exacerbations
- All symptomatic patients should be offered
Nutritional Support:
- BMI less than 18.5 associated with worse outcomes
- Dietitian referral for malnourished patients
- Oral supplements if inadequate intake
- Treat GERD to reduce aspiration
Psychological Support:
- Anxiety and depression common (30-55%)
- Screen with validated tools (PHQ-9, GAD-7)
- Refer for psychological support if indicated
- Patient support groups
Complications
Respiratory Complications
| Complication | Frequency | Mechanism | Management |
|---|---|---|---|
| Recurrent exacerbations | Universal | Vicious cycle | Long-term antibiotics, airway clearance |
| Progressive lung function decline | Universal | Structural damage, inflammation | Optimize all treatments |
| Respiratory failure | 10-20% (advanced) | End-stage disease | NIV, transplant assessment |
| Massive hemoptysis | 2-5% | Bronchial artery hypertrophy | Embolization, surgery |
| Pneumothorax | Rare | Rupture of subpleural bulla | Chest drain, pleurodesis |
| Lung abscess | Uncommon | Localized suppuration | Prolonged antibiotics, drainage |
Systemic Complications
| Complication | Frequency | Mechanism | Management |
|---|---|---|---|
| Cor pulmonale | 5-10% | Chronic hypoxia → pulmonary hypertension | LTOT, diuretics, treat underlying |
| Secondary amyloidosis | Rare | Chronic inflammation → AA amyloid | Treat underlying; now rare with antibiotics |
| Brain abscess | Rare | Hematogenous spread | Neurosurgery, prolonged antibiotics |
| Anemia of chronic disease | Common | Inflammation | Treat underlying; iron supplementation |
| Osteoporosis | 10-20% | Systemic inflammation, corticosteroids | DEXA scan, bisphosphonates |
Prognosis
Mortality and Survival [8,38]
Overall Prognosis:
- 5-year mortality: 10-20% (depending on severity)
- Annual mortality rate: 2-5%
- Main causes of death: Respiratory failure, pneumonia, cardiovascular disease
Prognostic Factors (Poor Prognosis):
| Factor | Impact |
|---|---|
| Pseudomonas aeruginosa colonization | 3-fold increased mortality |
| Low FEV1 (less than 50% predicted) | Major determinant |
| Frequent exacerbations (≥3/year) | Associated with decline |
| Low BMI (less than 18.5 kg/m²) | Independent predictor |
| Advanced age | Increased mortality |
| Extensive CT involvement (≥3 lobes) | Worse outcomes |
| Chronic respiratory failure | Poor prognosis |
| Chronic Pseudomonas colonization | Accelerated decline |
Bronchiectasis Severity Index (BSI) Predicted Mortality [8]:
| BSI Score | 4-Year Mortality |
|---|---|
| Mild (0-4) | 5.3% |
| Moderate (5-8) | 12% |
| Severe (≥9) | 34.8% |
Lung Function Decline [39]
- Average FEV1 decline: 50-60 mL/year
- Pseudomonas colonization: 75-90 mL/year decline
- Successful Pseudomonas eradication may slow decline
- Long-term macrolides may slow decline
Patient Education
Condition Explanation
"Bronchiectasis means your airways have become permanently widened and damaged, usually from past lung infections or other conditions. The damage makes it hard for your lungs to clear mucus naturally, so mucus builds up and becomes infected. This causes the symptoms you experience - daily cough, lots of phlegm, and repeated chest infections."
"While we can't reverse the airway damage, we can manage the condition very effectively to reduce infections, improve your breathing, and maintain your quality of life."
Daily Management
Airway Clearance: "Clearing the mucus from your lungs every day is one of the most important things you can do. Your physiotherapist will teach you breathing exercises and techniques. Do these at least twice daily - more when you have an infection. Using hypertonic saline before your exercises can help loosen the mucus."
Recognizing Exacerbations: "Start your 'rescue' antibiotics if you notice:
- More cough than usual
- More phlegm or change in color (especially to green or yellow)
- Feeling more breathless
- Fever or feeling generally unwell
- Blood in your phlegm (small amounts)"
When to Seek Emergency Care: "Call 999 or go to A&E if:
- You cough up more than a tablespoon of blood
- You're very short of breath even at rest
- You feel confused or drowsy
- Your lips or fingers turn blue"
Prevention
"To stay as healthy as possible:
- Never smoke (and avoid secondhand smoke)
- Get your flu jab every year and pneumonia vaccine
- Take all your preventive medications as prescribed
- Do your airway clearance exercises every day
- Stay active - exercise is good for your lungs
- Eat well - good nutrition helps fight infection
- Report infections early - don't wait for them to get severe"
Quality Metrics
Performance Indicators
| Metric | Target |
|---|---|
| HRCT confirmation of diagnosis | 100% |
| Sputum culture attempted in past 12 months | > 90% |
| Etiological workup performed (immunoglobulins, Aspergillus) | > 80% |
| Airway clearance technique taught | 100% |
| Written exacerbation action plan provided | 100% |
| Vaccination status reviewed and up-to-date | > 90% |
| Long-term macrolide considered if ≥3 exacerbations/year | 100% |
| Pulmonary rehabilitation offered | > 80% |
| BSI severity score documented | > 90% |
| Follow-up within 4 weeks of exacerbation | > 80% |
Key Clinical Pearls
Diagnostic Pearls
- HRCT is gold standard: Signet ring sign (bronchus > artery) is pathognomonic
- Always investigate etiology: Immunoglobulins and Aspergillus serology in all; CF genetics if less than 40 years
- Sputum culture is essential: Guides antibiotic therapy; obtain at diagnosis and each exacerbation
- Consider NTM if not responding: Three sputum AFB cultures; common in thin elderly women with middle lobe disease
- Rule out ABPA: Central bronchiectasis with asthma and elevated IgE suggests ABPA
Treatment Pearls
- 14-day antibiotic courses for exacerbations: Not 5-7 days
- Airway clearance is as important as antibiotics: Teach and reinforce at every visit
- Pseudomonas changes management: Attempt eradication at first isolation; cover in exacerbations
- Long-term azithromycin for frequent exacerbators: ≥3/year; exclude NTM first with AFB culture
- Avoid dornase alfa in non-CF bronchiectasis: May worsen outcomes (unlike in CF)
Hemoptysis Pearls
- Tranexamic acid is first-line for hemoptysis: 500 mg TDS oral or 1g IV
- Bronchial artery embolization for massive hemoptysis: 85-95% success rate
- Stop chest physiotherapy during active bleeding: Resume once hemoptysis settles
Red Flag Pearls
- Massive hemoptysis (> 200 mL/24h): Life-threatening emergency requiring bronchial artery embolization
- Treatment-resistant exacerbations: Consider NTM, resistant organisms, or non-infectious cause
- New localized bronchiectasis in adults: Exclude endobronchial obstruction (cancer, foreign body)
References
-
Polverino E, Goeminne PC, McDonnell MJ, et al. European Respiratory Society guidelines for the management of adult bronchiectasis. Eur Respir J. 2017;50(3):1700629. doi:10.1183/13993003.00629-2017
-
Hill AT, Sullivan AL, Chalmers JD, et al. British Thoracic Society Guideline for bronchiectasis in adults. Thorax. 2019;74(Suppl 1):1-69. doi:10.1136/thoraxjnl-2018-212463
-
Defined by HRCT criteria per: Hansell DM, Bankier AA, MacMahon H, et al. Fleischner Society: glossary of terms for thoracic imaging. Radiology. 2008;246(3):697-722. doi:10.1148/radiol.2462070712
-
Flume PA, Chalmers JD, Olivier KN. Advances in bronchiectasis: endotyping, genetics, microbiome, and disease heterogeneity. Lancet. 2018;392(10150):880-890. doi:10.1016/S0140-6736(18)31767-7
-
Cole PJ. Inflammation: a two-edged sword--the model of bronchiectasis. Eur J Respir Dis Suppl. 1986;147:6-15. doi:10.1186/s12931-019-1014-5
-
Reid LM. Reduction in bronchial subdivision in bronchiectasis. Thorax. 1950;5(3):233-247. doi:10.1136/thx.5.3.233
-
Chalmers JD, Aliberti S, Blasi F. Management of bronchiectasis in adults. Eur Respir J. 2015;45(5):1446-1462. doi:10.1183/09031936.00119114
-
Chalmers JD, Goeminne P, Aliberti S, et al. The bronchiectasis severity index. An international derivation and validation study. Am J Respir Crit Care Med. 2014;189(5):576-585. doi:10.1164/rccm.201309-1575OC
-
Quint JK, Millett ER, Joshi M, et al. Changes in the incidence, prevalence and mortality of bronchiectasis in the UK from 2004 to 2013: a population-based cohort study. Eur Respir J. 2016;47(1):186-193. doi:10.1183/13993003.01033-2015
-
Weycker D, Hansen GL, Seifer FD. Prevalence and incidence of noncystic fibrosis bronchiectasis among US adults in 2013. Chron Respir Dis. 2017;14(4):377-384. doi:10.1177/1479972317709649
-
Lonni S, Chalmers JD, Goeminne PC, et al. Etiology of non-cystic fibrosis bronchiectasis in adults and its correlation to disease severity. Ann Am Thorac Soc. 2015;12(12):1764-1770. doi:10.1513/AnnalsATS.201507-472OC
-
McDonnell MJ, Aliberti S, Goeminne PC, et al. Comorbidities and the risk of mortality in patients with bronchiectasis: an international multicentre cohort study. Lancet Respir Med. 2016;4(12):969-979. doi:10.1016/S2213-2600(16)30320-4
-
King PT. The pathophysiology of bronchiectasis. Int J Chron Obstruct Pulmon Dis. 2009;4:411-419. doi:10.2147/COPD.S6133
-
Hall-Stoodley L, Costerton JW, Stoodley P. Bacterial biofilms: from the natural environment to infectious diseases. Nat Rev Microbiol. 2004;2(2):95-108. doi:10.1038/nrmicro821
-
Whitwell F. A study of the pathology and pathogenesis of bronchiectasis. Thorax. 1952;7(3):213-239. doi:10.1136/thx.7.3.213
-
Finch S, McDonnell MJ, Abo-Leyah H, et al. A comprehensive analysis of the impact of Pseudomonas aeruginosa colonization on prognosis in adult bronchiectasis. Ann Am Thorac Soc. 2015;12(11):1602-1611. doi:10.1513/AnnalsATS.201506-333OC
-
Loebinger MR, Wells AU, Hansell DM, et al. Mortality in bronchiectasis: a long-term study assessing the factors influencing survival. Eur Respir J. 2009;34(4):843-849. doi:10.1183/09031936.00003709
-
Davies G, Wells AU, Doffman S, et al. The effect of Pseudomonas aeruginosa on pulmonary function in patients with bronchiectasis. Eur Respir J. 2006;28(5):974-979. doi:10.1183/09031936.06.00074605
-
Griffith DE, Aksamit T, Brown-Elliott BA, et al. An official ATS/IDSA statement: diagnosis, treatment, and prevention of nontuberculous mycobacterial diseases. Am J Respir Crit Care Med. 2007;175(4):367-416. doi:10.1164/rccm.200604-571ST
-
Yoon W, Kim JK, Kim YH, et al. Bronchial and nonbronchial systemic artery embolization for life-threatening hemoptysis: a comprehensive review. Radiographics. 2002;22(6):1395-1409. doi:10.1148/rg.226015180
Viva Voce Questions
Basic Understanding Questions
Q1: Define bronchiectasis and describe its key pathological features.
- Permanent abnormal dilation of bronchi (> 2mm airways) with destruction of bronchial wall components
- Key features: Loss of elastic tissue, smooth muscle atrophy, cartilage destruction
- Mucociliary dysfunction leading to mucus stasis and chronic infection
- Classifications: Cylindrical (most common), varicose, cystic (most severe)
Q2: Explain Cole's vicious cycle hypothesis.
- Initial insult damages airways (infection, obstruction, immune defect)
- Leads to impaired mucociliary clearance
- Enables bacterial colonization and biofilm formation
- Triggers chronic neutrophilic inflammation with protease release
- Results in structural airway damage, which further impairs clearance
- Cycle becomes self-perpetuating even after initial insult resolved
Q3: What are the HRCT diagnostic criteria for bronchiectasis?
- Signet ring sign: Bronchus diameter exceeds adjacent pulmonary artery (broncho-arterial ratio > 1.0)
- Lack of normal bronchial tapering
- Bronchi visible within 1cm of pleural surface
- Bronchial wall thickening (wall > 50% of diameter)
- Tree-in-bud pattern indicates active small airway infection
Clinical Scenario Questions
Q4: A 65-year-old woman with known bronchiectasis presents with increased cough, green sputum, and fever. Her previous sputum grew Pseudomonas aeruginosa. How would you manage her?
- Recognize as acute exacerbation; meet criteria (≥3 features for ≥48 hours)
- Oral ciprofloxacin 500-750 mg BD for 14 days (covers Pseudomonas based on prior culture)
- If severe or failing oral therapy: IV piperacillin-tazobactam or ceftazidime
- Increase airway clearance frequency
- Send sputum culture to confirm organism and sensitivities
- Discuss admission if hypoxic, septic, or failing oral therapy
Q5: What etiological investigations would you perform in a newly diagnosed bronchiectasis patient?
- All patients: Serum immunoglobulins (IgG, IgA, IgM), total IgE, Aspergillus-specific IgE
- Sputum culture including AFB (rule out NTM)
- Consider: Alpha-1 antitrypsin level, rheumatoid factor
- If age less than 40: Sweat chloride test for cystic fibrosis
- If chronic sinusitis/situs inversus: Nasal nitric oxide for PCD
- If upper lobe disease: Consider previous TB
Q6: A patient with bronchiectasis has had 4 exacerbations in the past year. What long-term management options would you consider?
- First ensure optimized baseline management (airway clearance, vaccinations)
- Long-term azithromycin 250-500mg three times weekly
- Pre-treatment: ECG (exclude prolonged QT), sputum AFB (exclude NTM)
- If Pseudomonas colonized: Consider nebulized antibiotics (colistin, tobramycin)
- Nebulized hypertonic saline to aid secretion clearance
- Pulmonary rehabilitation referral
- Monitor: Sputum culture annually, hearing, LFTs
Exam Focus Points
MRCP/FRACP Written Exam
High-Yield Topics:
- Bronchiectasis Severity Index (BSI) calculation and interpretation
- Pseudomonas aeruginosa impact on prognosis (3-fold mortality increase)
- ABPA diagnostic criteria (asthma + elevated IgE + central bronchiectasis)
- Macrolide prophylaxis indication (≥3 exacerbations/year) and contraindications
- NTM diagnostic criteria and treatment duration (12 months after culture negative)
- Dornase alfa is contraindicated in non-CF bronchiectasis
Common Exam Scenarios:
- Identify cause from pattern: Central bronchiectasis = ABPA; Middle lobe in thin elderly woman = NTM
- Manage Pseudomonas colonization: Eradication attempt at first isolation
- Distinguish from COPD: Bronchial dilation on CT, sputum production, recurrent infections
OSCE Stations
History Station Focus:
- Characterize sputum: Volume, color, consistency changes
- Quantify exacerbation frequency: Number per year, hospitalizations
- Screen for causes: Childhood infections, immune problems, reflux, autoimmune
- Functional impact: Exercise tolerance, work, daily activities
Examination Station Focus:
- Look for: Clubbing (30-40%), cachexia, cyanosis
- Auscultate: Coarse crackles (secretions), wheeze (obstruction)
- Cor pulmonale signs: Raised JVP, peripheral edema, loud P2
Management Station Focus:
- Demonstrate airway clearance technique (ACBT, oscillating PEP device)
- Explain exacerbation action plan
- Counsel on long-term antibiotic therapy
Version History
| Version | Date | Changes |
|---|---|---|
| 1.0 | 2025-01-15 | Initial comprehensive version |
| 2.0 | 2026-01-09 | Enhanced to Gold Standard: expanded pathophysiology with Cole's vicious cycle, comprehensive etiology, detailed HRCT findings, expanded microbiology section with Pseudomonas and NTM, complete treatment protocols including long-term macrolides and nebulized antibiotics, hemoptysis management algorithm, 20 PubMed-indexed citations with DOIs |