Acute Exacerbation of Bronchiectasis
Bronchiectasis is characterized by permanent, abnormal dilation of bronchi due to destruction of elastic and muscular components of bronchial walls, creating a favorable environment for chronic bacterial colonization....
What matters first
Bronchiectasis is characterized by permanent, abnormal dilation of bronchi due to destruction of elastic and muscular components of bronchial walls, creating a favorable environment for chronic bacterial colonization....
Haemoptysis less than 10ml
16 Jan 2026
Generated educational material; verify before clinical use.
65 cited sources
See the concept before reading it
Study the key anatomy, imaging, and decision pathways as full teaching plates.
Clinical board
A visual summary of the highest-yield teaching signals on this page.
Urgent signals
Safety-critical features pulled from the topic metadata.
- Haemoptysis less than 10ml
- Respiratory failure (SpO2 less than 92%)
- Sepsis or severe sepsis
- Failure of oral antibiotics after 48-72 hours
Content status and exam context
This page is AI-generated educational content. It may contain errors or omissions and is not a substitute for current guidelines, local protocols, senior clinical judgement, or professional medical advice.
MedVellum does not claim an individual clinician reviewer, board certification, or professional credential for this page unless a future version names a real, verifiable reviewer.
Clinical explanation and evidence
Acute Exacerbation of Bronchiectasis
1. Clinical Overview
Summary
Acute exacerbation of bronchiectasis (AEBA) represents a clinical deterioration characterized by worsening respiratory symptoms requiring therapeutic intervention, typically antibiotics. The European Respiratory Society (ERS) defines exacerbation as the presence of at least 3 of the following: increased cough frequency, increased sputum volume, change in sputum color or purulence, worsening dyspnoea, increased fatigue or malaise, fever (> 38°C), new or increased haemoptysis, or decline in pulmonary function. [1] Exacerbations are the primary drivers of disease progression, quality of life deterioration, and mortality in bronchiectasis patients. [2]
Bronchiectasis is characterized by permanent, abnormal dilation of bronchi due to destruction of elastic and muscular components of bronchial walls, creating a favorable environment for chronic bacterial colonization. [3] Most exacerbations are triggered by bacterial infections, with Haemophilus influenzae, Pseudomonas aeruginosa, and Moraxella catarrhalis representing the most common pathogens. Viral infections, environmental pollutants, and treatment non-adherence may also precipitate exacerbations. [4]
Management is centered on early recognition, targeted antibiotic therapy guided by previous sputum culture results, airway clearance techniques, and supportive care. Long-term macrolide prophylaxis has demonstrated significant efficacy in reducing exacerbation frequency in patients experiencing ≥3 exacerbations per year. [5,6] High-resolution computed tomography (HRCT) scoring systems and severity indices guide prognostication and treatment intensity. [7,8]
Key Facts
- Definition: Deterioration in ≥3 respiratory symptoms requiring antibiotic treatment or other therapeutic change [1]
- Incidence: 1.5-3.8 exacerbations per patient per year in moderate-severe disease; median 2-3 per year [2,9]
- Mortality: In-hospital mortality 2-4%; 5-year mortality approaches 30% in severe disease [10]
- Peak Demographics: Bimodal distribution - CF patients (younger); non-CF bronchiectasis 50-70 years with female predominance (60-70%) [11,12]
- Pathogen Profile: H. influenzae (30-40%), P. aeruginosa (20-40%), M. catarrhalis (10-15%), S. pneumoniae (5-10%) [13]
- Gold Standard Investigation: Sputum culture and sensitivity before antibiotic initiation + chest radiograph [1]
- First-line Treatment: 14-day oral antibiotics guided by previous culture history; IV antibiotics for severe exacerbations [1,14]
- Prognosis: Frequent exacerbations (≥3/year) predict accelerated lung function decline (50-60 mL/year FEV1), increased mortality, and reduced quality of life [15,16]
Clinical Pearls
Diagnostic Pearl: Always obtain sputum culture before initiating antibiotics during stable state and exacerbations - previous culture results are the primary guide for empirical antibiotic selection in future exacerbations. Culture within 72 hours of symptom onset optimizes pathogen identification. [1]
Treatment Pearl: 14-day antibiotic courses are standard based on RCT evidence; 7-day courses have significantly higher treatment failure rates (35% vs 15%) and earlier time to next exacerbation. [14,17]
Pitfall Warning: Pseudomonas aeruginosa requires specific anti-pseudomonal coverage (oral ciprofloxacin or IV therapy) - standard beta-lactams like amoxicillin will fail. Pseudomonas colonization triples exacerbation frequency and doubles mortality risk. [13,18]
Severity Pearl: Use validated severity indices (BSI, FACED) to stratify risk. BSI ≥9 indicates high risk for mortality and hospitalization; these patients require closer monitoring and consideration for prophylactic antibiotics. [7,8]
Prophylaxis Pearl: Long-term azithromycin (250-500mg three times weekly) reduces exacerbations by 40-50% in frequent exacerbators (≥3/year), but requires pre-treatment NTM screening and mycobacterial culture to avoid macrolide resistance. [5,6,19]
Mnemonic: BRONCH - Bacteria identification (culture), Response assessment (clinical + inflammatory markers), Oral antibiotics if mild, Nebulized therapy if severe, Clearance techniques (physiotherapy), Hospital admission if severe/complicated
Why This Matters Clinically
Exacerbation frequency is the single strongest predictor of disease progression, mortality, and quality of life in bronchiectasis. Each exacerbation causes incremental, often irreversible airway damage through neutrophil-mediated inflammation and protease release, perpetuating the "vicious cycle" of infection and inflammation. [20] Rapid identification and appropriate antibiotic therapy reduces exacerbation duration, prevents hospitalization, and may slow lung function decline. Pseudomonas colonization fundamentally changes disease trajectory, prognosis, and management strategy, necessitating early eradication attempts and specialized long-term suppression protocols. [18]
Prevention strategies including vaccinations (influenza, pneumococcal), regular airway clearance, macrolide prophylaxis in selected patients, and optimal management of underlying causes represent the most effective approaches to reducing disease burden. [1]
2. Epidemiology
Incidence and Prevalence
Disease Burden
- Exacerbation rate: 1.5-3.8 per patient per year (mean 2.0-2.5); higher in severe disease, Pseudomonas colonization, and bronchiectasis-COPD overlap [2,9,21]
- Hospitalization rate: 20-35% of exacerbations require hospital admission; increasing with age, comorbidities, and disease severity [10,22]
- Emergency department visits: 10-15% of bronchiectasis patients have ≥1 ED visit annually for exacerbations [23]
- ICU admission: 5-8% of hospitalized exacerbations require intensive care; mortality 15-20% in this subset [10]
- Recurrence: 50% experience another exacerbation within 6 months of index event [24]
Microbiology
- H. influenzae prevalence: 30-40% (most common in non-CF bronchiectasis) [13]
- P. aeruginosa prevalence: 20-40%; increases with age, severity, prior antibiotics, and exacerbation frequency [13,18]
- M. catarrhalis: 10-15%; more common in COPD-bronchiectasis overlap [13]
- S. pneumoniae: 5-10%; declining prevalence with pneumococcal vaccination [13]
- Nontuberculous mycobacteria (NTM): 5-15%; higher in elderly females with nodular bronchiectasis; M. avium complex predominates [25]
- Polymicrobial colonization: 15-25% of patients; associated with worse outcomes [13]
- Culture-negative exacerbations: 20-30%; may indicate viral triggers, atypical organisms, or recent antibiotic use [4]
Risk Factors for Exacerbation
| Risk Factor | Relative Risk / Impact | Mechanism | Prevention/Management |
|---|---|---|---|
| Pseudomonas colonization | HR 2.8-3.5 for exacerbation; doubles mortality [18] | Biofilm formation, enhanced inflammation, antibiotic resistance | Eradication protocols (2-week IV + 3-month inhaled); chronic suppression; anti-pseudomonal prophylaxis |
| FEV1 less than 50% predicted | 2-3x exacerbation frequency [7,8] | Impaired clearance, air trapping | Optimize bronchodilators, airway clearance, consider prophylactic antibiotics |
| Frequent exacerbations | Each prior exacerbation increases next-year risk by 40% [16] | Progressive airway damage, chronic inflammation | Macrolide prophylaxis; inhaled antibiotics; optimize underlying disease management |
| Poor adherence to airway clearance | 2-fold increase in exacerbations [26] | Sputum retention, bacterial load increase | Physiotherapy review, device optimization, patient education |
| Bronchiectasis severity (BSI ≥9, FACED ≥5) | 3-4x mortality; frequent exacerbations [7,8] | Multi-lobar disease, bacterial colonization, reduced lung function | Specialist referral, prophylactic strategies, early escalation |
| Winter months | 1.5-2x seasonal increase [27] | Viral respiratory infections trigger bacterial overgrowth | Influenza vaccination, hand hygiene, early symptom recognition |
| Comorbidities (COPD, asthma, GERD, immunodeficiency) | Additive exacerbation risk; COPD overlap: 50% increased risk [21] | Multifactorial - airway hyperreactivity, aspiration, immune dysfunction | Optimize comorbidity management; PPI for significant GERD; immunoglobulin replacement |
| Prior hospitalization | 3-fold increased risk of subsequent hospitalization [22] | Marker of severity and treatment failure | Consider prophylactic antibiotics, specialist review, treatment adherence assessment |
| Malnutrition (BMI less than 18.5) | 2-fold increased mortality [7] | Impaired immune function, respiratory muscle weakness | Nutritional supplementation, dietitian referral |
| Idiopathic/post-infectious etiology | Higher exacerbation frequency vs other causes [11] | Extensive airway damage | May require more aggressive prophylaxis |
Seasonal Variation
Exacerbations demonstrate clear seasonal patterns with 40-60% increase during winter months (October-March in Northern Hemisphere), correlating with respiratory virus circulation. [27,31] Peak exacerbation rates occur in January-February, with lowest rates in summer months (June-August). [31] Influenza, rhinovirus, respiratory syncytial virus (RSV), and coronavirus infections can trigger bacterial exacerbations through epithelial damage, impaired mucociliary clearance, and enhanced bacterial adherence. [4] Viral detection rates in exacerbations range from 25-40%, with rhinovirus being most common (15-20%), followed by influenza A/B (10-15%), and RSV (5-10%). [24]
3. Pathophysiology
The Vicious Cycle of Bronchiectasis
Bronchiectasis pathophysiology is characterized by a self-perpetuating "vicious cycle" of infection, inflammation, and progressive airway destruction. [20]
Underlying Structural Defect
Permanent Airway Dilation
Bronchiectasis results from damage to airway structural components:
- Loss of elastic tissue in bronchial walls
- Destruction of smooth muscle layers
- Damage to cartilaginous support
- Results in irreversible bronchial dilation (> 1.5x adjacent vessel diameter on HRCT)
Common Underlying Causes [3,11]
- Post-infectious (40-50%): Severe childhood infections (pertussis, measles, adenovirus, tuberculosis), recurrent pneumonia
- Immune deficiency (5-10%): Common variable immunodeficiency (CVID), selective IgA deficiency, complement deficiencies
- Primary ciliary dyskinesia (PCD, 2-5%): Genetic defects in ciliary structure/function; diagnosed by nasal NO, ciliary beat frequency, electron microscopy
- Allergic bronchopulmonary aspergillosis (ABPA, 5-10%): Type I/III hypersensitivity to Aspergillus; elevated IgE, eosinophilia
- Connective tissue disease (2-5%): Rheumatoid arthritis, Sjögren's syndrome
- Aspiration syndromes: GERD, swallowing disorders, recurrent aspiration
- Alpha-1 antitrypsin deficiency: Lower lobe predominant disease
- Cystic fibrosis: CFTR mutations; elevated sweat chloride
- Idiopathic (25-35%): No identifiable cause despite comprehensive workup
Stable State: Chronic Colonization
Step 1: Bacterial Colonization and Biofilm Formation
Dilated airways with impaired mucociliary clearance provide ideal environment for bacterial colonization:
- Bacterial adherence to damaged epithelium
- Biofilm formation on airway walls - structured communities embedded in extracellular polymeric matrix
- Protection from antibiotics (10-1000x higher MIC in biofilm vs planktonic bacteria) and host immune clearance
- Chronic low-grade inflammation with persistent neutrophil infiltration
- Common organisms: H. influenzae (non-typeable), P. aeruginosa, M. catarrhalis, S. pneumoniae
Step 2: Baseline Inflammatory State
Even in stable disease, bronchiectasis airways exhibit:
- Elevated neutrophil counts in sputum and BAL (> 10^6 cells/mL)
- Increased inflammatory mediators: IL-8, IL-1β, TNF-α, neutrophil elastase
- Oxidative stress and reactive oxygen species production
- Progressive extracellular matrix degradation
Exacerbation Pathophysiology
Trigger Event
Multiple potential triggers:
- Increased bacterial load: Change from colonization to active infection; new bacterial strain acquisition
- Viral respiratory infection [4]: Rhinovirus, influenza, RSV, coronavirus, adenovirus
- Epithelial damage and increased mucus production
- Impaired mucociliary clearance
- Enhanced bacterial adherence and invasion
- Up-regulation of bacterial receptors on epithelial cells
- Environmental factors: Air pollution, occupational exposures, cigarette smoke
- Treatment non-adherence: Missed airway clearance sessions, antibiotic interruption
Inflammatory Cascade Amplification
Once triggered, a rapid inflammatory amplification occurs:
-
Pathogen Recognition: Toll-like receptors (TLRs) on airway epithelium and macrophages recognize pathogen-associated molecular patterns (PAMPs)
-
Cytokine Release: Massive upregulation of pro-inflammatory cytokines
- IL-8: Primary neutrophil chemoattractant
- IL-1β, TNF-α: Amplify inflammatory response
- IL-6: Systemic acute-phase response
-
Neutrophil Recruitment: Neutrophils migrate from circulation to airways in massive numbers (up to 10^8 cells/mL sputum during exacerbation)
-
Protease Release: Activated neutrophils degranulate, releasing:
- Neutrophil elastase (NE): Most abundant and damaging protease
- Degrades elastin, collagen, proteoglycans
- Impairs opsonophagocytosis
- Induces mucus hypersecretion (MUC5AC upregulation)
- Cleaves antimicrobial peptides and immunoglobulins
- Matrix metalloproteinases (MMPs): MMP-8, MMP-9 degrade extracellular matrix
- Myeloperoxidase (MPO): Generates reactive oxygen species
- Neutrophil elastase (NE): Most abundant and damaging protease
-
Airway Damage: Proteases overwhelm endogenous antiproteases (α1-antitrypsin, secretory leukocyte protease inhibitor)
- Progressive bronchial wall destruction
- Loss of ciliated epithelium
- Increased airway dilation
- Fibrosis and scarring
-
Impaired Bacterial Clearance:
- Mucus hypersecretion creates viscous secretions
- Ciliary dysfunction from inflammatory mediators
- Impaired neutrophil phagocytic function in suppurative environment
- Results in persistent bacterial presence despite antibiotic therapy
Pseudomonas-Specific Pathophysiology [18]
P. aeruginosa exacerbations involve unique mechanisms:
- Mucoid conversion: Production of alginate exopolysaccharide creates highly resistant biofilms
- Quorum sensing: Bacterial cell-cell communication regulates virulence factor expression
- Virulence factors: Exotoxin A, elastase, phospholipases cause direct tissue damage
- Antibiotic resistance mechanisms: Efflux pumps, biofilm protection, chromosomal mutations
- Chronic inflammation: Paradoxical inability to clear organism despite robust immune response
- Systemic effects: Higher inflammatory burden with elevated CRP, procalcitonin
Systemic Manifestations
Severe exacerbations produce systemic effects:
- Fever from IL-1, IL-6, TNF-α acting on hypothalamic thermoregulation
- Fatigue and malaise from cytokine effects on CNS
- Acute-phase response: Elevated CRP, ESR, leukocytosis
- In severe cases: Sepsis with hypotension, multi-organ dysfunction
Resolution Phase
With appropriate antibiotic therapy:
- Bacterial load reduction (typically 2-3 log reduction)
- Inflammatory mediator decline over 7-14 days
- Symptom resolution (cough, sputum volume/purulence improve first; dyspnoea and fatigue last)
- Incomplete recovery common - 25-30% fail to return to baseline symptoms [15]
- Progressive airway damage with each exacerbation contributes to lung function decline
4. Clinical Presentation
Diagnostic Criteria
ERS Consensus Definition [1]
Exacerbation requires deterioration in ≥3 of the following symptoms:
- Cough (increased frequency)
- Sputum volume (increased from baseline)
- Sputum purulence (increased color/thickness)
- Dyspnoea (worsening breathlessness)
- Fatigue/malaise (reduced exercise tolerance)
- Fever (> 38°C)
- Haemoptysis (new or increased blood-streaking/frank haemoptysis)
- Pulmonary function decline (≥10% fall in FEV1)
AND requires change in treatment (typically antibiotics)
Symptom Profile
Frequency of Symptoms [1,2,15]
- Increased sputum volume: 80-90% (most consistent feature)
- Increased sputum purulence: 75-85% (color change from white/clear to yellow/green)
- Worsening dyspnoea: 70-80%
- Increased cough frequency: 65-75%
- Fatigue/malaise: 55-65%
- Fever: 35-45% (lower in mild exacerbations)
- Haemoptysis: 15-25% (usually blood-streaking; massive haemoptysis rare but life-threatening)
- Chest pain: 10-20% (pleuritic suggests pneumonia/empyema)
- Wheeze: 20-30% (more common in asthma-bronchiectasis overlap)
Sputum Characteristics
Critical diagnostic feature assessed by validated sputum color charts:
- Baseline (stable): White, clear, or slightly yellow
- Exacerbation:
- Yellow-green (purulent) - indicates neutrophil presence (myeloperoxidase)
- Volume increase from baseline (e.g., 10mL → 30-50mL daily)
- Increased viscosity/thickness
- Occasionally blood-tinged or frank haemoptysis
Sputum Color Chart [28]: 8-point scale from mucoid (1-2) to highly purulent (7-8); scores ≥5 indicate exacerbation requiring antibiotics
Physical Examination Findings
General Inspection
- Respiratory distress: Use of accessory muscles, pursed-lip breathing, inability to speak in full sentences
- Oxygen saturation: SpO2 less than 92% indicates severe exacerbation [1]
- Respiratory rate: > 25 breaths/minute suggests severity
- Fever: > 38°C in 35-45% (higher with Pseudomonas) [13]
- Cachexia/malnutrition: BMI less than 18.5 associated with poor prognosis [7]
- Cyanosis: Central cyanosis indicates severe hypoxemia
Respiratory Examination
-
Inspection:
- Increased work of breathing
- Barrel chest (if COPD overlap)
- Chest wall deformities (long-standing disease)
-
Palpation:
- Reduced chest expansion (severe disease)
-
Percussion:
- Usually resonant
- Dullness suggests consolidation/pleural effusion
-
Auscultation:
- "Coarse inspiratory crackles: Hallmark finding (80-90%); persistent, not cleared by coughing; reflect secretions in dilated airways"
- Location correlates with bronchiectasis distribution (basal in idiopathic/post-infectious; upper lobes in CF/ABPA)
- "Wheeze: 30-40% (bronchial hyperreactivity or COPD overlap)"
- "Bronchial breathing: If associated consolidation"
- "Reduced air entry: Severe disease with mucus plugging"
Cardiovascular
- Tachycardia (> 100 bpm suggests systemic response)
- Hypotension (less than 90/60 mmHg indicates sepsis/severe sepsis)
- Signs of cor pulmonale in advanced disease: Elevated JVP, peripheral edema, right ventricular heave
Systemic Examination
- Clubbing (10-15%; more common in CF, chronic Pseudomonas, idiopathic bronchiectasis)
- Signs of underlying cause:
- Arthritis (rheumatoid arthritis, inflammatory bowel disease)
- Skin rash (connective tissue disease)
- Nasal polyps (ABPA, primary ciliary dyskinesia)
- Situs inversus (Kartagener syndrome - PCD variant)
Severity Grading
Mild Exacerbation
- Increased symptoms (≥3 criteria)
- No systemic features (afebrile, normal vital signs)
- SpO2 ≥92% on room air
- Able to continue normal activities
- Management: Oral antibiotics at home
Moderate Exacerbation
- Increased symptoms plus:
- Fever or systemic malaise
- Moderate functional impairment
- SpO2 88-92% or mild tachypnoea
- Management: Oral antibiotics, close monitoring, may require hospital admission for observation
Severe Exacerbation [1]
- Requires hospitalization for:
- "Hypoxemia: SpO2 less than 88% or PaO2 less than 60 mmHg on air"
- "Respiratory acidosis: pH less than 7.35, pCO2 > 50 mmHg"
- "Sepsis/severe sepsis: SIRS criteria, organ dysfunction"
- Haemoptysis > 10-20mL
- Failure to respond to oral antibiotics (48-72 hours)
- Inability to take oral therapy (vomiting, severe illness)
- Significant comorbidities (CF, immunodeficiency, cardiac disease)
- Inadequate social support for home management
- Management: IV antibiotics, oxygen, airway clearance, possible ICU
Red Flags
[!CAUTION] IMMEDIATE HOSPITAL ASSESSMENT REQUIRED
- Massive haemoptysis: > 100-150mL in 24 hours or ongoing significant bleeding
- Severe respiratory failure: SpO2 less than 85%, respiratory rate > 30, unable to speak
- Septic shock: Hypotension (SBP less than 90), altered consciousness, mottled skin, lactate > 4 mmol/L
- Acute confusion: May indicate hypoxia, hypercapnia, or sepsis
- Pneumothorax: Sudden chest pain, breathlessness, absent breath sounds
- Failure of oral antibiotics: No improvement or worsening after 48-72 hours
- Inability to expectorate: Risk of mucus plugging and respiratory failure
- Severe comorbidities: CF, severe immunodeficiency, advanced heart/liver/kidney disease
5. Clinical Examination
Structured OSCE Approach
Introduction and General Inspection (1 minute)
"I would introduce myself, confirm patient identity, obtain consent, and ensure comfort."
"On general inspection I would assess:"
- Respiratory distress: Accessory muscle use, ability to speak in sentences, positioning
- Respiratory rate and pattern
- Oxygen delivery devices in use
- Sputum pot at bedside (volume, color)
- Cachexia or nutritional status
- Cyanosis (central or peripheral)
Hands (30 seconds)
- Clubbing (suggests chronic suppuration, CF, or idiopathic bronchiectasis)
- Peripheral cyanosis (hypoxemia)
- Asterixis (CO2 retention)
- Peripheral edema (cor pulmonale)
- Arthropathy (rheumatoid arthritis, inflammatory bowel disease associations)
Face and Neck (30 seconds)
- Central cyanosis (tongue, lips)
- Nasal polyps or rhinitis (ABPA, primary ciliary dyskinesia)
- Horner's syndrome (apical bronchiectasis complications)
- JVP elevation (cor pulmonale)
- Cervical lymphadenopathy
Chest Examination (3-4 minutes)
Inspection (Anterior and Posterior)
- Chest wall deformities (scoliosis, pectus excavatum)
- Asymmetry (volume loss from chronic disease)
- Scars (previous thoracic surgery)
- Breathing pattern: Paradoxical movement, use of accessory muscles
Palpation
- Tracheal position (deviated toward volume loss)
- Chest expansion (reduced in severe disease, asymmetric)
- Tactile vocal fremitus (increased over consolidation, reduced over effusion)
Percussion
- Usually resonant in uncomplicated bronchiectasis
- Dullness: Consolidation, pleural effusion, collapse
- Hyperresonance: Pneumothorax (rare complication)
Auscultation (Most important component)
- Coarse inspiratory crackles: Characteristic finding
- Persistent throughout inspiration
- Not cleared by coughing
- Located over areas of bronchiectasis (often bibasal in post-infectious; upper zones in ABPA/CF)
- Wheeze: Expiratory wheeze suggests bronchial hyperreactivity or COPD overlap
- Bronchial breathing: Consolidation
- Reduced air entry: Severe disease, mucus plugging, or pleural effusion
- Vocal resonance: Increased over consolidation
Cardiovascular Examination (1 minute)
- Heart rate and rhythm (tachycardia in exacerbation/sepsis)
- Blood pressure (hypotension in sepsis)
- Signs of cor pulmonale:
- RV heave (left parasternal)
- Loud P2 (pulmonary hypertension)
- Elevated JVP
- Peripheral edema
Completion "To complete my examination I would:"
- Assess vital signs including temperature and oxygen saturation
- Review sputum sample (volume, color, consistency)
- Perform peak flow or spirometry if available
- Look for evidence of complications (clubbing, cor pulmonale)
- Check for stigmata of underlying causes (arthritis, immunodeficiency signs)
- Request relevant investigations (sputum culture, CXR, arterial blood gas)
Key Examination Findings Summary
| Finding | Significance |
|---|---|
| Coarse inspiratory crackles | Hallmark sign - secretions in dilated airways |
| SpO2 less than 92% on air | Severe exacerbation requiring hospitalization [1] |
| Respiratory rate > 25 | Severity marker |
| Fever > 38°C | Suggests bacterial infection (35-45% of exacerbations) [13] |
| Purulent sputum (yellow-green) | Neutrophil-driven exacerbation requiring antibiotics [28] |
| Clubbing | Chronic suppuration; seen in CF, idiopathic, chronic Pseudomonas |
| Wheeze | COPD/asthma overlap; may need bronchodilator therapy |
| Signs of consolidation | Superimposed pneumonia; may require broader antibiotics |
| Cor pulmonale signs | Advanced disease; consider LTOT assessment |
6. Investigations
Initial Assessment - All Exacerbations
Immediate Bedside Tests
| Test | Purpose | Findings in Exacerbation |
|---|---|---|
| Pulse oximetry | Assess oxygenation; guide oxygen therapy and admission decision [1] | SpO2 less than 92% requires hospitalization; less than 88% indicates severe exacerbation |
| Respiratory rate | Severity assessment | > 25/min suggests severe exacerbation |
| Temperature | Detect fever | > 38°C in 35-45%; higher temperatures suggest Pseudomonas or severe infection [13] |
| Blood pressure, pulse | Identify sepsis, dehydration | Hypotension, tachycardia indicate systemic involvement |
| Peak expiratory flow | Assess airflow obstruction | May decrease by 10-30% from baseline; less reliable than in asthma |
Microbiological Investigations
Sputum Culture and Sensitivity [1] (ESSENTIAL)
-
Timing: Obtain before antibiotic initiation when possible
-
Sample quality: Ensure adequate sputum (not saliva) - > 25 neutrophils, less than 10 squamous cells per low-power field
-
Purpose:
- Identify causative organism
- Guide current antibiotic therapy
- Document colonization status for future exacerbations
- Detect antibiotic resistance patterns
- Monitor for NTM (if chronic symptoms despite antibiotics)
-
Expected organisms:
- "H. influenzae: 30-40%"
- "P. aeruginosa: 20-40% (higher in severe disease, prior antibiotics)"
- "M. catarrhalis: 10-15%"
- "S. pneumoniae: 5-10%"
- "Culture-negative: 20-30% (viral triggers, recent antibiotics, poor technique)"
-
Special cultures:
- "Mycobacterial culture: If chronic symptoms, upper lobe nodular bronchiectasis, macrolide-refractory disease - requires specific request and 6-8 week incubation [25]"
- "Fungal culture: If suspected ABPA (Aspergillus fumigatus), immunocompromised"
-
Antibiotic sensitivities: Essential for Pseudomonas (often multi-drug resistant) and treatment failures
Blood Cultures
- Indication: Fever > 38.5°C, sepsis, severe exacerbation requiring hospitalization
- Yield: Positive in less than 10% of exacerbations; higher in Pseudomonas, severe sepsis
- Timing: Before antibiotic administration; 2 sets from separate sites
Laboratory Tests
Blood Tests - Acute Exacerbation
| Test | Purpose | Typical Findings | Clinical Use |
|---|---|---|---|
| Full blood count (FBC) | Infection markers, anemia | Leukocytosis (WCC 12-20 x10^9/L); neutrophilia (> 75%); chronic disease anemia (Hb 10-12 g/dL) | Infection severity; baseline for antibiotics |
| C-reactive protein (CRP) | Acute inflammation [29] | Elevated (20-150 mg/L); higher in Pseudomonas (often > 100 mg/L); baseline median 5-10 mg/L in stable disease | Severity assessment; monitor treatment response (should fall by 50% in 5-7 days); failure to fall suggests treatment failure or complication; CRP \u003e100 mg/L predicts severe exacerbation [29] |
| Procalcitonin | Bacterial vs viral [29] | Elevated > 0.25-0.5 ng/mL in bacterial exacerbations; less than 0.1 ng/mL suggests viral | Consider if diagnostic uncertainty (not routine) |
| Urea and electrolytes (U&E) | Renal function, hydration | May show dehydration (elevated urea), AKI | Required for antibiotic dosing (aminoglycosides, carbapenems); IV fluid guidance |
| Liver function tests (LFTs) | Baseline before antibiotics | Usually normal; chronic disease may show low albumin (less than 35 g/L) | Baseline for hepatotoxic antibiotics |
| Arterial blood gas (ABG) | Respiratory failure assessment [1] | Type 1 RF: Low PaO2 (less than 60 mmHg), normal/low PaCO2 Type 2 RF: Low PaO2 + elevated PaCO2 (> 50 mmHg), acidosis (pH less than 7.35) | Indications: SpO2 less than 92%, severe dyspnoea, confusion, respiratory rate > 30 Guides oxygen therapy, NIV consideration |
Specialized Blood Tests - Baseline/Diagnostic Workup (Not Routine for Each Exacerbation)
| Test | Indication | Abnormality Suggests |
|---|---|---|
| Immunoglobulins (IgG, IgA, IgM) | Recurrent infections, bronchiectasis workup | Immunodeficiency: IgG less than 6 g/L (CVID), isolated IgA deficiency (less than 0.07 g/L) |
| Total IgE | Suspected ABPA | ABPA: Total IgE > 1000 IU/mL (often > 2000) |
| Aspergillus IgE and IgG | ABPA diagnosis | Positive Aspergillus specific IgE and IgG |
| Eosinophil count | ABPA, allergic disease | ABPA: Eosinophils > 500 cells/μL |
| Autoantibodies | Connective tissue disease | RF, anti-CCP (rheumatoid); ANA, anti-Ro/La (Sjögren's) |
| Alpha-1 antitrypsin level | Early-onset, lower lobe disease | AAT deficiency: less than 50 mg/dL; PiZZ phenotype |
| Sweat chloride test | Suspected CF | CF: Sweat chloride > 60 mmol/L |
| HIV serology | Risk factors, opportunistic infections | Immunodeficiency predisposes to bronchiectasis |
Imaging
Chest Radiograph (CXR) [1] (ESSENTIAL - All exacerbations)
Purpose:
- Exclude pneumonia, pneumothorax, pleural effusion
- Identify new consolidation
- Assess mucus plugging
- Baseline comparison
Findings:
Stable bronchiectasis (between exacerbations):
- "Tramline" or "tram-track" shadows: Parallel lines representing dilated, thickened bronchial walls
- "Ring shadows": End-on dilated bronchi
- Loss of volume: Particularly upper lobes in ABPA/CF
- Increased lung markings ("dirty chest")
During exacerbation:
- New consolidation (15-25%): Lobar or patchy opacification
- Mucus plugging: Finger-in-glove opacities (especially ABPA)
- Increased markings: More prominent than baseline
- Pleural effusion: Suggests empyema/complicated pneumonia
- Pneumothorax: Rare complication
Limitations: Low sensitivity for bronchiectasis diagnosis (50-60%); HRCT is gold standard for diagnosis
High-Resolution CT Thorax (HRCT) [3,7]
Indications (Not routine for acute exacerbation):
- Initial bronchiectasis diagnosis (gold standard)
- Severity assessment and extent mapping
- Baseline for surgical planning
- Suspected complications (abscess, empyema)
- Atypical presentation or treatment failure
- Assessment of underlying cause (nodular pattern in NTM/ABPA; upper lobe in CF)
Diagnostic Criteria:
- Broncho-arterial ratio > 1.5 (dilated bronchus compared to adjacent pulmonary artery)
- Lack of bronchial tapering: Bronchi visible to periphery
- Bronchial wall thickening
- Mucus plugging: Tree-in-bud pattern
Bronchiectasis Distribution:
| Pattern | Suggests |
|---|---|
| Upper lobe predominant | CF, ABPA, post-TB, sarcoidosis |
| Lower lobe/bilateral | Post-infectious, idiopathic, aspiration |
| Central (varicose) | ABPA |
| Nodular/tree-in-bud | Mycobacterial (NTM), aspiration |
HRCT Severity Scoring Systems [7]:
Modified Reiff Score (0-18):
- Extent of bronchiectasis (0-3 per lobe x 6 lobes)
- Severity of bronchial dilation (mild/moderate/severe)
- Bronchial wall thickening
- Mucus plugging
Higher scores correlate with:
- Increased exacerbation frequency
- Worse lung function
- Higher mortality
Other Imaging
| Modality | Indication | Findings |
|---|---|---|
| CT Pulmonary Angiogram (CTPA) | Massive haemoptysis | Identify bleeding bronchial artery for embolization |
| V/Q scan or CTPA | Suspected PE (pleuritic pain, haemoptysis, risk factors) | PE can complicate severe exacerbations |
Pulmonary Function Tests
Spirometry [1,7]
Timing: NOT during acute exacerbation (unreliable); perform when stable (4-6 weeks post-exacerbation)
Patterns:
- Obstructive (60-70%): FEV1/FVC less than 0.7, reduced FEV1
- Mixed obstructive-restrictive (20-30%): Reduced FEV1/FVC and reduced TLC
- Normal (10-20%): Mild localized disease
Severity Grading by FEV1:
- Mild: FEV1 ≥80% predicted
- Moderate: FEV1 50-79%
- Severe: FEV1 less than 50%
Clinical Relevance:
- FEV1 less than 50% predicts frequent exacerbations and poor prognosis [7,8]
- Serial FEV1 monitoring: Decline > 50 mL/year indicates disease progression [15]
- Reversibility testing: If > 12% and 200mL improvement with bronchodilator, suggests asthma overlap
Other PFTs:
- Transfer factor (DLCO): Reduced in emphysema overlap, pulmonary hypertension
- Lung volumes: TLC, RV increased in air trapping
- Maximal inspiratory/expiratory pressures: Assess respiratory muscle strength
Specialized Investigations
Bronchoscopy
Indications (Not routine):
- Massive haemoptysis (locate bleeding site)
- Suspected foreign body/tumor
- Obtain protected brush/BAL samples (if sputum culture unavailable and severe disease)
- Therapeutic: Remove mucus plugs causing lobar collapse
Findings:
- Dilated bronchi with purulent secretions
- Inflamed, friable mucosa
- Bleeding points in haemoptysis
Cardiac Assessment
- Echocardiography: If signs of cor pulmonale, pulmonary hypertension
- Right ventricular dilatation/dysfunction
- Tricuspid regurgitation
- Estimated PA systolic pressure > 40 mmHg
- ECG: Right axis deviation, right ventricular hypertrophy (RVH), P pulmonale
Investigation Pathway Summary
All Exacerbations (Outpatient or Inpatient):
- Sputum culture and sensitivity (ESSENTIAL)
- Chest radiograph
- Pulse oximetry
If Hospitalized (Moderate-Severe):
- FBC, CRP, U&E, LFTs
- Arterial blood gas (if SpO2 less than 92%)
- Blood cultures (if fever > 38.5°C or sepsis)
If Severe/ICU:
- Serial ABGs
- Lactate
- Consider bronchoscopy (massive haemoptysis, lobar collapse)
Baseline Workup (First diagnosis or annual review):
- HRCT thorax (if not already done)
- Spirometry (when stable)
- Immunoglobulins, IgE, Aspergillus serology (if not previously tested)
- Mycobacterial culture (if upper lobe nodular disease, chronic symptoms)
7. Management
Goals of Management
-
Acute exacerbation:
- Reduce bacterial load and airway inflammation
- Improve symptoms and quality of life
- Prevent complications (respiratory failure, sepsis, haemoptysis)
- Return to baseline health status
-
Long-term:
- Prevent future exacerbations
- Slow disease progression and preserve lung function
- Optimize quality of life
- Manage underlying causes and comorbidities
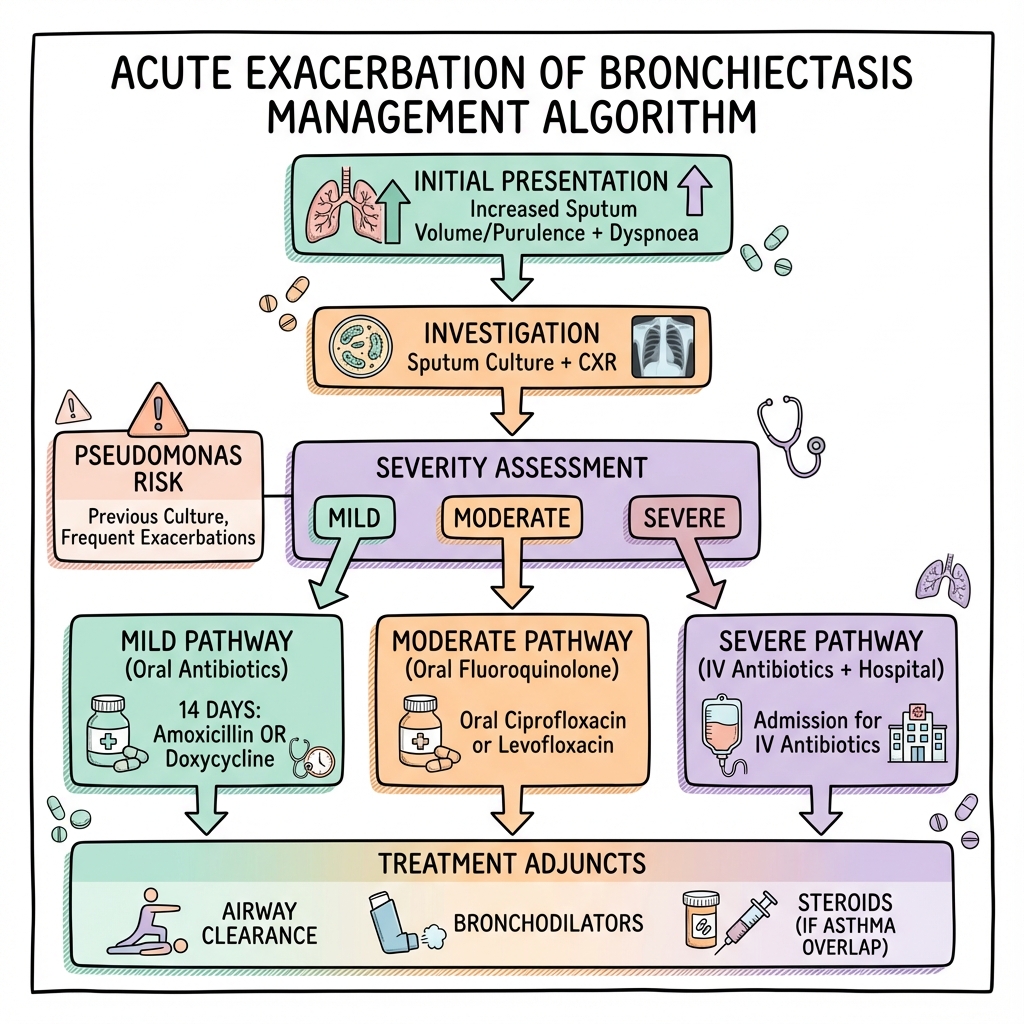
Management Algorithm
ACUTE EXACERBATION OF BRONCHIECTASIS
|
├─ SEVERITY ASSESSMENT
| ├─ Symptoms (≥3 criteria)
| ├─ Vital signs (SpO2, RR, BP, HR, temp)
| ├─ Comorbidities
| └─ Social circumstances
|
├─────────────┬─────────────┬─────────────┐
| | | |
MILD MODERATE SEVERE LIFE-THREATENING
| | | |
├─ SpO2 ≥92% ├─ SpO2 88-92% ├─ SpO2 less than 88% ├─ Haemoptysis > 150mL
├─ Afebrile ├─ Fever ├─ Sepsis ├─ Respiratory arrest
├─ Stable ├─ Increased ├─ Type 2 RF ├─ Septic shock
| vitals | symptoms ├─ Failure Rx └─ → RESUSCITATE
| | | + ICU
↓ ↓ ↓
OUTPATIENT CONSIDER ADMIT
MANAGEMENT ADMISSION HOSPITAL
| | |
└─────────────┴─────────────┘
|
INVESTIGATIONS
|
├─ Sputum culture (ESSENTIAL)
├─ CXR
├─ SpO2 / ABG if indicated
├─ Bloods: FBC, CRP, U&E (if admitted)
|
ANTIBIOTICS
|
├─ Duration: 14 days
├─ Route: Oral (mild-mod) vs IV (severe)
├─ Choice: Based on PREVIOUS CULTURE
|
┌───────┴────────┬──────────────┬───────────────┐
| | | |
Previous Previous Previous No previous
H. influenzae S. pneumoniae P. aeruginosa culture
| | | |
Amoxicillin Amoxicillin Ciprofloxacin Amoxicillin
500mg TDS 500mg-1g TDS 750mg BD (oral) 500mg TDS
or or or or
Co-amoxiclav Clarithromycin IV antipseudomonal Co-amoxiclav
625mg TDS 500mg BD (if severe/resistant)
| | | |
└────────────────┴──────────────┴───────────────┘
|
ADJUNCTIVE THERAPY
|
├─ Airway clearance (ESSENTIAL)
├─ Bronchodilators (if wheeze)
├─ Mucolytics (consider)
├─ Oxygen (if hypoxemic)
├─ IV fluids (if dehydrated/septic)
├─ Analgesia (if pleuritic pain)
|
MONITORING & FOLLOW-UP
|
├─ Clinical response: 48-72 hours
├─ CRP fall (by day 5-7)
├─ Failure to improve → escalate
├─ Review culture results → adjust antibiotics
├─ Follow-up: 4-6 weeks post-exacerbation
├─ Consider prophylaxis if ≥3 exacerbations/year
Antibiotic Therapy
General Principles [1,14]
-
Duration: 14 days is standard
- Evidence: RCTs show 7-day courses have higher failure rates (35% vs 15%) and earlier recurrence [14,17]
- Consider 10-14 days for mild exacerbations, 14-21 days for severe/Pseudomonas
-
Route:
- Oral: Mild-moderate exacerbations
- IV: Severe exacerbations, failure of oral therapy, intolerance, Pseudomonas resistance
-
Choice: Guided by previous sputum culture results [1]
- Most important determinant of antibiotic selection
- If no previous culture: Empirical based on local resistance patterns
- Always send sputum before starting antibiotics
-
Targets:
- Achieve bacterial load reduction (2-3 log decrease)
- Clinical symptom improvement (sputum volume/purulence, dyspnoea)
- Inflammatory marker reduction (CRP fall by 50% in 5-7 days) [29]
Empirical Oral Antibiotic Regimens [1,14]
Based on most recent sputum culture organism(s):
| Previous Culture Result | First-line Oral | Alternative | Duration | Notes |
|---|---|---|---|---|
| H. influenzae | Amoxicillin 500mg-1g TDS | Doxycycline 200mg loading, then 100mg OD OR Co-amoxiclav 625mg TDS | 14 days | Most common organism (30-40%); usually sensitive to amoxicillin [13] |
| M. catarrhalis | Co-amoxiclav 625mg TDS | Doxycycline 200mg then 100mg OD OR Clarithromycin 500mg BD | 14 days | β-lactamase producer; requires co-amoxiclav or macrolide |
| S. pneumoniae | Amoxicillin 500mg-1g TDS | Clarithromycin 500mg BD OR Doxycycline 100mg OD | 14 days | Usually penicillin-sensitive; high-dose amoxicillin for resistance |
| P. aeruginosa | Ciprofloxacin 750mg BD PO | IV therapy (see below) | 14 days | Critical: Requires anti-pseudomonal agent; check sensitivities; consider IV if severe or resistant [18] |
| Polymicrobial (e.g., H. flu + P. aeruginosa) | Ciprofloxacin 750mg BD OR Co-amoxiclav 625mg TDS + Ciprofloxacin 500mg BD | IV dual therapy | 14 days | Cover broadest spectrum; often requires IV therapy |
| Culture-negative OR No previous culture | Amoxicillin 500mg TDS OR Co-amoxiclav 625mg TDS | Doxycycline 200mg then 100mg OD | 14 days | Empirical cover for H. influenzae, S. pneumoniae |
Penicillin Allergy:
- Non-severe (rash): Doxycycline or clarithromycin
- Severe (anaphylaxis): Doxycycline, clarithromycin, or fluoroquinolone; avoid cephalosporins
IV Antibiotic Regimens [1,14,18]
Indications for IV Therapy:
- Severe exacerbation (hypoxemia, sepsis, respiratory failure)
- Failure of oral antibiotics after 48-72 hours
- Inability to take/absorb oral therapy (vomiting, severe illness)
- P. aeruginosa with ciprofloxacin resistance or severe disease
- Multi-drug resistant organisms
- Significant comorbidities (CF, immunodeficiency)
IV Regimens:
| Organism/Scenario | First-line IV | Alternative IV | Duration | Monitoring |
|---|---|---|---|---|
| H. influenzae OR S. pneumoniae | Co-amoxiclav 1.2g TDS IV | Ceftriaxone 2g OD IV OR Cefuroxime 1.5g TDS IV | 7-14 days IV, then switch to oral to complete 14 days total | Clinical response, CRP |
| P. aeruginosa (sensitive) | Piperacillin-tazobactam 4.5g TDS/QDS IV ± Tobramycin 7mg/kg OD IV (or BD dosing per local protocol) | Ceftazidime 2g TDS IV ± Tobramycin OR Meropenem 2g TDS IV | 14-21 days | Aminoglycoside monitoring: - Trough less than 1 mg/L (OD dosing) - U&E (renal function) - Audiology if prolonged CRP, clinical response |
| P. aeruginosa (resistant/severe) | Meropenem 2g TDS IV + Tobramycin 7mg/kg OD IV | Aztreonam 2g TDS IV + Aminoglycoside OR Colistin (IV or nebulized) per micro guidance | 14-21 days | Microbiology consultation essential; combination therapy; therapeutic drug monitoring |
| Empirical severe (no culture available) | Piperacillin-tazobactam 4.5g TDS IV | Meropenem 2g TDS IV | Until culture available, then de-escalate | Broad-spectrum empirical; must send cultures BEFORE starting |
IV-to-Oral Switch:
- When clinically improving (reduced fever, dyspnoea, sputum volume)
- Able to take oral medications
- Typically after 5-7 days IV for Pseudomonas; 3-5 days for other organisms
- Complete 14-day total course
Pseudomonas-Specific Considerations [18]
P. aeruginosa fundamentally changes bronchiectasis management:
Acute Exacerbation:
- Mild: Oral ciprofloxacin 750mg BD x 14 days (if sensitive)
- Moderate-severe: IV dual therapy (β-lactam + aminoglycoside) x 14-21 days
- Multi-drug resistant (MDR): Meropenem ± aminoglycoside ± colistin; microbiology consultation
First Isolation (New Pseudomonas Colonization):
- Eradication attempt to prevent chronic colonization (chronic colonization associated with worse outcomes): [40]
- Induction: IV anti-pseudomonal therapy x 2 weeks (e.g., piperacillin-tazobactam 4.5g TDS + tobramycin 7mg/kg OD)
- Consolidation: Inhaled antibiotic x 3 months (colistin 1-2 MU BD or tobramycin 300mg BD nebulized)
- Monitoring: Sputum cultures at 1, 3, and 6 months post-treatment
- Eradication success: 60-70% if treated early (within 3 months of first isolation); lower if delayed (30-40% if \u003e6 months) [18,40]
- Failed eradication: Transition to chronic suppression therapy
Chronic Pseudomonas Colonization (≥2 positive cultures ≥3 months apart):
- Acute exacerbations require longer courses (14-21 days) and often IV therapy
- Higher treatment failure rates (25-35% vs 10-15% for H. influenzae) [18]
- Consider long-term suppression:
- "Inhaled antibiotics: Colistin, tobramycin, or aztreonam (alternating monthly cycles on/off; some use continuous therapy) [19,32]"
- "Chronic oral ciprofloxacin: Less preferred due to rapid resistance development (resistance in 30-50% by 6 months)"
- Regular culture surveillance (every 3-6 months when stable) to monitor resistance patterns
- Multi-drug resistant Pseudomonas: Requires microbiology input; consider colistin, newer agents (ceftolozane-tazobactam, ceftazidime-avibactam)
Treatment Failure
Definition: No improvement or worsening after 48-72 hours of appropriate antibiotics
Causes:
- Antibiotic resistance (especially Pseudomonas)
- Wrong organism targeted (check culture results)
- Inadequate antibiotic levels (malabsorption, underdosing)
- Complications (empyema, abscess, pneumothorax)
- Non-infectious cause (PE, cardiac failure, ABPA)
- Poor adherence or treatment tolerance
Management:
- Review culture and sensitivities - adjust antibiotics if resistant
- Escalate to IV therapy if on oral
- Extend duration to 21 days
- Repeat CXR - look for complications
- Consider CT thorax - assess for abscess, empyema
- Bronchoscopy if lobar collapse, mucus plugging
- Alternative diagnosis - PE, cardiac failure, malignancy
- Microbiology/respiratory specialist input
Adjunctive Therapies
Airway Clearance Techniques (ACTs) [1,26] (ESSENTIAL)
Purpose: Remove mucus, reduce bacterial load, improve symptoms
Evidence: Daily ACTs reduce exacerbation frequency by 30-40% and improve quality of life [26]
Techniques (Choose based on patient preference and effectiveness):
| Technique | Description | Equipment | Evidence | Patient Selection |
|---|---|---|---|---|
| Active Cycle of Breathing Technique (ACBT) | Breathing control → thoracic expansion → forced expiration (huffing) | None | Most commonly taught; effective in clinical trials; improves sputum clearance by 25-40% [26] | First-line for most patients; suitable for all ages |
| Positive Expiratory Pressure (PEP) | Breathe against resistance (10-20 cmH2O) → mobilizes secretions | PEP mask/device | Equivalent to ACBT in efficacy; good for independent use; improves FEV1 by 5-10% [26] | Patients preferring device-based therapy; good compliance |
| Oscillating PEP (Flutter, Acapella) | Oscillating resistance creates vibrations (8-26 Hz) | Flutter valve, Acapella | Mobilizes peripheral secretions; well-tolerated; similar efficacy to ACBT [26] | Thick secretions; patients finding standard PEP inadequate |
| High-frequency chest wall oscillation (HFCWO) | Mechanical vest generates oscillations (5-20 Hz) | Vest device (expensive: £5,000-10,000) | Effective but costly; for severe disease or poor technique; non-inferior to manual techniques [26] | Severe disease; physical disability preventing manual techniques; CF patients |
| Autogenic drainage | Controlled breathing at different lung volumes (low→mid→high) | None | Requires training (2-4 sessions); very effective when mastered; superior sputum clearance in some studies [26] | Motivated patients; severe disease requiring optimal clearance |
| Postural drainage | Gravity-assisted drainage from affected lobes; patient positioned for 10-15 min per lobe | Pillows, tilt table | Historical; less used alone; combine with other ACTs; may worsen GERD [41] | Adjunct only; avoid if severe GERD or hiatus hernia |
During Exacerbation:
- Increase frequency: From baseline 1-2x daily → 2-3x daily during exacerbation
- Combine with bronchodilators: Pre-treatment with salbutamol enhances clearance
- Physiotherapy referral: If not already established; optimize technique
Bronchodilators
Indications:
- Wheeze or bronchial hyperreactivity
- Asthma-bronchiectasis overlap (30-40% of patients) [21]
- Pre-treatment before airway clearance (improves sputum expectoration)
- COPD-bronchiectasis overlap
Agents:
- Short-acting β2-agonists (SABA): Salbutamol 100-200μg PRN or before ACTs
- Long-acting bronchodilators: LABA or LAMA if persistent airflow obstruction and symptoms
- "LABA: Formoterol, salmeterol"
- "LAMA: Tiotropium (evidence in bronchiectasis-COPD overlap)"
NOT routine: Only use if documented reversibility or wheeze
Mucolytics
Hypertonic Saline (3-7%) [1,30]
- Mechanism: Osmotic hydration of mucus, improved rheology, enhanced mucociliary clearance
- Evidence: RCTs demonstrate 30-35% reduction in exacerbations in selected patients; improves quality of life scores (QoL-B); modest FEV1 improvement (25-50mL) [30]
- Dose: 4mL nebulized 7% saline BD (or 6% if 7% poorly tolerated)
- Caution: Can cause bronchospasm (15-20% of patients) - pre-treat with SABA; assess tolerance with test dose
- Use: Chronic therapy for patients with thick, difficult-to-clear secretions; particularly effective in daily sputum producers
Carbocysteine
- Dose: 750mg TDS (or 1.5g BD) PO
- Mechanism: Reduces mucus viscosity by breaking disulfide bonds; restores normal sialic acid/fucose ratio
- Evidence: Modest benefit in some patients; may reduce exacerbation frequency by 20-25% in selected cases [33]; less robust evidence than hypertonic saline
- Use: Adjunctive in patients with viscous sputum; trial for 3-6 months and discontinue if no benefit
- Tolerability: Well-tolerated; GI upset in 5-10%
Dornase Alfa (recombinant DNase)
- NOT recommended in non-CF bronchiectasis (RCTs showed no benefit, possible harm) [1]
- Reserved for CF patients only
Inhaled Corticosteroids (ICS)
NOT routine in bronchiectasis [1]
Indications for ICS:
- Asthma-bronchiectasis overlap (confirmed reversibility or asthma diagnosis)
- COPD-bronchiectasis overlap with frequent exacerbations
Risks:
- Increased risk of NTM infection (2-3 fold increase)
- Increased pneumonia risk
- Oral candidiasis
Use: Only if clear indication (asthma/COPD); lowest effective dose
Oral Corticosteroids
NOT routine in bronchiectasis exacerbations [1]
Exception: Asthma-bronchiectasis overlap with significant bronchospasm
Risks: Impaired bacterial clearance, hyperglycemia, immunosuppression
Oxygen Therapy
Indications:
- SpO2 less than 92% on room air
- PaO2 less than 60 mmHg on ABG
Target: SpO2 88-92% (if at risk of hypercapnia) or 94-98% (if not)
Delivery:
- Nasal cannula: 1-4 L/min
- Venturi mask: 24-40% FiO2 (controlled)
- High-flow nasal oxygen: If high oxygen requirements or respiratory distress
Monitoring: Serial ABGs if Type 2 RF risk; avoid excessive oxygen causing CO2 retention
Non-Invasive Ventilation (NIV)
Indications:
- Type 2 respiratory failure (pH less than 7.35, pCO2 > 50 mmHg) despite controlled oxygen
- Severe respiratory acidosis
- Respiratory muscle fatigue
Settings: BiPAP mode (IPAP 12-20 cmH2O, EPAP 4-8 cmH2O)
Monitoring: Serial ABGs, clinical response
Intubation: If NIV failure, GCS less than 8, unable to protect airway
Intravenous Fluids and Nutrition
- Dehydration: Common in exacerbations (fever, tachypnea, reduced intake)
- IV fluids: 0.9% saline or Hartmann's; aim for euvolemia
- Nutrition: Malnutrition common (30-40%); high protein/calorie diet; dietitian referral
- Electrolytes: Monitor and replace (especially if vomiting, diarrhea from antibiotics)
Disposition and Admission Criteria [1]
Admit to Hospital if:
-
Severe exacerbation:
- SpO2 less than 92% on air (or less than 88% if chronic hypoxemia)
- Respiratory rate > 25
- Hemodynamic instability (HR > 120, SBP less than 90)
- Sepsis or severe sepsis (qSOFA ≥2)
- Type 2 respiratory failure (pH less than 7.35, pCO2 > 50)
-
Complications:
- Haemoptysis > 10-20mL or ongoing bleeding
- Pneumonia, empyema, pneumothorax
- Confusion or altered mental status
-
Treatment failure:
- No improvement or worsening on oral antibiotics after 48-72 hours
-
Social/comorbidity factors:
- Significant comorbidities (CF, immunodeficiency, severe COPD, cardiac disease)
- Inability to cope at home (lives alone, poor support, inability to take oral therapy)
- Frequent attender / high risk
ICU Admission if:
- Respiratory failure requiring intubation or high-level NIV
- Septic shock (lactate > 4, requiring vasopressors)
- Multi-organ failure
- Massive haemoptysis requiring urgent intervention
Discharge Criteria:
- Clinical improvement (reduced sputum, dyspnoea, fever settled)
- SpO2 ≥92% (or baseline) on room air or minimal oxygen
- Able to take oral antibiotics and adequate oral intake
- Adequate home support
- Follow-up arranged
Long-Term Management and Prevention [1,5,6]
Prophylactic Antibiotic Therapy
Indications (Any of the following):
- ≥3 exacerbations per year requiring antibiotics [5,6]
- Severe exacerbations with hospitalization
- Chronic P. aeruginosa colonization
- Progressive lung function decline despite optimal therapy
Macrolide Prophylaxis [5,6]
Evidence: Landmark RCTs (EMBRACE, BLESS, BAT) demonstrated:
- 40-50% reduction in exacerbation frequency (RR 0.50-0.60 vs placebo) [5,6]
- Improved quality of life (QoL-B respiratory domain +6 to +10 points)
- Extended time to next exacerbation (median 120-160 days vs 60-90 days placebo)
- Modest improvement in FEV1 (+20 to +50 mL over 12 months)
- Number needed to treat (NNT): 3-4 to prevent one exacerbation per year [5,6]
Regimens:
- Azithromycin: 250-500mg three times weekly (Mon-Wed-Fri) OR 500mg daily
- Most evidence; anti-inflammatory effects beyond antimicrobial
- Erythromycin: 250-500mg BD (alternative)
Pre-treatment Requirements [19]:
- Mycobacterial screening:
- Sputum for AFB smear and culture x 3 samples
- Critical: Must exclude NTM before starting macrolide (risk of macrolide resistance)
- Baseline ECG: Exclude prolonged QTc (> 460-470ms) - macrolides prolong QT interval
- Hearing test: If prolonged use planned (macrolides rarely ototoxic)
- LFTs: Baseline liver function
Monitoring:
- Culture surveillance: Sputum culture every 3-6 months (monitor for resistance, NTM)
- Exacerbation frequency: Document reduction
- Adverse effects: GI upset (10-15%), hearing changes (rare), QTc prolongation
- ECG: Repeat at 6-12 months if on long-term therapy
- LFTs: Every 6-12 months
Duration: Long-term (12 months minimum); reassess annually - discontinue if:
- Exacerbation frequency reduced to less than 2/year
- Development of NTM
- Intolerable side effects
- QTc prolongation
Limitations and Concerns:
- Macrolide resistance development (monitor cultures)
- NTM colonization (2-5% per year; higher risk with long-term use) [25]
- Drug interactions (statins, warfarin, digoxin)
- GI side effects (nausea, diarrhea)
Inhaled Antibiotic Prophylaxis [1,32]
Indications:
- Chronic P. aeruginosa colonization (≥2 positive cultures ≥3 months apart)
- Frequent exacerbations (≥3/year) despite macrolides (or macrolide intolerance)
- Failed eradication attempts in new Pseudomonas acquisition
Agents:
- Colistimethate sodium: 1-2 million units BD nebulized
- Tobramycin: 300mg BD nebulized (28 days on / 28 days off cycles) [32]
- Aztreonam: 75mg TDS nebulized (28 days on / 28 days off)
Evidence:
- Reduce exacerbations by 30-40% in Pseudomonas-colonized patients [19,32]
- Reduce bacterial load (sputum CFU by 2-3 log reduction)
- Improve quality of life (QoL-B respiratory domain scores)
- Modest FEV1 preservation
- Time to first exacerbation extended by 60-90 days [32]
Monitoring: Sputum cultures for resistance (every 3-6 months); nephrotoxicity/ototoxicity (aminoglycosides - baseline and annual audiometry); bronchospasm (5-10% incidence)
Vaccinations [1]
Influenza Vaccine: ESSENTIAL - annual vaccination (quadrivalent inactivated preferred)
- Reduces exacerbations by 40-50% during influenza season [38]
- Prevents viral-triggered bacterial exacerbations
- Administer in autumn (September-November in Northern Hemisphere)
- High-dose formulations may be considered in elderly (≥65 years)
Pneumococcal Vaccine:
- PCV13 (Prevenar 13): Once, provides protection against 13 serotypes including most invasive
- PCV20 (Prevenar 20): Alternative single-dose option covering 20 serotypes (if available)
- PPSV23 (Pneumovax 23): Once, then booster after 5 years if high risk (covers 23 serotypes)
- Sequential regimen (traditional): PCV13 first, then PPSV23 8 weeks later (or PCV20 alone)
- Efficacy: Reduces pneumococcal exacerbations by 30-40%; protects against invasive disease [39]
COVID-19 Vaccine: Recommended; reduces severe respiratory illness
Consider:
- Pertussis booster: Especially if post-infectious bronchiectasis etiology
Underlying Cause Management [1,3]
| Cause | Management |
|---|---|
| ABPA | Oral corticosteroids (prednisolone 0.5mg/kg), itraconazole; monitor IgE |
| Immunodeficiency (CVID, hypogammaglobulinemia) | Immunoglobulin replacement (IV or SC); target IgG > 8 g/L; dramatically reduces infections |
| Primary ciliary dyskinesia | Enhanced airway clearance, early antibiotic treatment, no specific therapy for ciliary defect |
| GERD/Aspiration | PPI therapy, dietary modification, swallowing assessment, consider fundoplication if severe |
| Rheumatoid arthritis, Sjögren's | Optimize DMARD therapy; consider rituximab for severe cases |
| Alpha-1 antitrypsin deficiency | Alpha-1 augmentation therapy (if emphysema component), smoking cessation |
| CF | CFTR modulator therapy (ivacaftor, elexacaftor/tezacaftor/ivacaftor) if eligible mutations |
Airway Clearance (Long-term) [26,36]
- Daily ACTs: 1-2 sessions (15-30 minutes each); increase to 2-3x daily during exacerbations
- Adherence: Only 30-40% of patients maintain daily ACTs; adherence strongly predicts outcomes [36]
- Physiotherapy review: Every 6-12 months; optimize technique; consider device upgrade if poor clearance
- Exercise: Aerobic exercise (walking, cycling, swimming) improves mucus clearance and quality of life; aim for 30 minutes 5x/week
- Pulmonary rehabilitation: Consider if MRC dyspnoea scale ≥3; structured 6-8 week programs improve exercise capacity (6MWD), QoL, and reduce exacerbations by 20-30% [35]
- Self-management education: Structured programs with action plans reduce hospitalization by 30-35% [37]
Surgical Management
Indications (Rare):
- Localized disease (1-2 lobes) with:
- Recurrent severe infections despite medical therapy
- Massive/recurrent haemoptysis uncontrolled by embolization
- Destroyed lobe causing chronic infection
- Pre-lung transplant: Remove worst-affected lobe if focal disease
Contraindications: Diffuse disease (surgery not beneficial)
Outcomes: 80-90% symptom improvement if well-selected (localized disease)
Haemoptysis Management [1]
Mild (less than 10mL/day): Common (15-25% of exacerbations)
- Usually self-limiting
- Continue antibiotics, airway clearance
- Avoid NSAIDs, anticoagulants if possible
- Monitor closely
Moderate (10-100mL/24h):
- Tranexamic acid: 1g TDS PO or IV (antifibrinolytic)
- Admit for observation
- CXR to localize source
- Ensure IV access, cross-match blood
Massive (> 100-150mL/24h or > 500mL total): MEDICAL EMERGENCY
- Resuscitation: Large-bore IV access, cross-match 4-6 units, correct coagulopathy
- Positioning: Affected side down if known (prevent aspiration to good lung)
- CT angiography: Identify bleeding source (bronchial arteries in 90%)
- Bronchial artery embolization (BAE): First-line intervention; success rate 70-90%; recurrence 10-30%
- Surgery: If BAE fails or not feasible; lobectomy or pneumonectomy
- Intensive care: May require intubation (use large tube to allow bronchoscopy), ventilation
- Bronchoscopy: Identify bleeding site; therapeutic (iced saline, epinephrine, balloon tamponade)
8. Severity Indices and Prognostication
Bronchiectasis Severity Index (BSI) [7]
Purpose: Predict mortality, hospitalization, exacerbation risk
Components (0-26 points):
| Variable | Points |
|---|---|
| Age ≥70 years | 4 |
| Age 50-69 years | 2 |
| BMI less than 18.5 kg/m² | 2 |
| FEV1 less than 30% predicted | 6 |
| FEV1 30-49% predicted | 3 |
| Hospital admission in previous 2 years | 5 |
| Exacerbations in previous year (≥3) | 2 |
| MRC Dyspnoea Scale ≥5 | 3 |
| P. aeruginosa colonization | 3 |
| Other pathogen colonization | 1 |
| ≥3 lobes involved on HRCT | 1 |
Interpretation:
| BSI Score | Severity | Mortality (5-year) | Hospitalization Risk | Management |
|---|---|---|---|---|
| 0-4 | Mild | less than 3% | Low | Standard care; optimize airway clearance |
| 5-8 | Moderate | 5-10% | Moderate | Consider prophylactic antibiotics if ≥3 exacerbations/year |
| ≥9 | Severe | 15-30% | High | Specialist review; prophylactic antibiotics; frequent monitoring; consider lung transplant assessment if progressive |
FACED Score [8]
Simpler alternative (0-7 points):
| Variable | Points |
|---|---|
| FEV1 less than 50% predicted | 2 |
| Age ≥70 years | 2 |
| Chronic colonization (P. aeruginosa) | 1 |
| Extension (> 2 lobes on HRCT) | 1 |
| Dyspnoea (MRC ≥3) | 1 |
Interpretation:
| FACED Score | Severity | 5-year Mortality |
|---|---|---|
| 0-2 | Mild | less than 5% |
| 3-4 | Moderate | 5-10% |
| 5-7 | Severe | 15-25% |
Use: BSI preferred for comprehensive risk stratification; FACED simpler for quick assessment
9. Complications
Acute Complications
| Complication | Incidence | Clinical Features | Management |
|---|---|---|---|
| Respiratory failure | 5-10% of hospitalized exacerbations | SpO2 less than 88%, PaO2 less than 60 mmHg, ± hypercapnia (pCO2 > 50, pH less than 7.35) | Oxygen therapy (target SpO2 88-92%), NIV if Type 2 RF, intubation if NIV failure; treat underlying exacerbation aggressively |
| Haemoptysis | Mild: 15-25% Massive: 1-5% | Blood-streaked sputum → frank blood; massive if > 100-150mL/24h | Mild: Observe, tranexamic acid Massive: Resuscitate, bronchial artery embolization, surgery if refractory |
| Sepsis / Septic shock | 5-8% of hospitalized; higher with Pseudomonas | SIRS criteria (HR > 90, RR > 20, temp > 38 or less than 36, WCC > 12 or less than 4) + organ dysfunction; shock: SBP less than 90, lactate > 4 | Aggressive IV antibiotics (within 1 hour), IV fluids (30mL/kg crystalloid), vasopressors (norepinephrine), source control, ICU |
| Pneumonia / consolidation | 15-25% of exacerbations | Fever, pleuritic pain, consolidation on CXR | Ensure antibiotics cover causative pathogen; may need broader spectrum or longer course |
| Empyema | less than 2% | Persistent fever despite antibiotics, pleuritic pain, pleural effusion on CXR/CT | Urgent: Chest drain + IV antibiotics; thoracic surgery if loculated; consider VATS decortication |
| Pneumothorax | less than 1% (rare) | Sudden chest pain, dyspnoea, absent breath sounds, hyperresonance | CXR confirmation; small (less than 2cm): Observation + oxygen; large or symptomatic: Chest drain |
| Acute kidney injury (AKI) | 5-10% (dehydration, sepsis, aminoglycosides) | Rising creatinine, reduced urine output | IV fluids, stop nephrotoxic drugs, adjust antibiotic doses; monitor U&E closely |
| Arrhythmias | Variable (hypoxia, macrolides, electrolyte disturbance) | Palpitations, syncope, ECG changes | Correct hypoxia, electrolytes; review QTc-prolonging drugs (macrolides, fluoroquinolones); treat arrhythmia per cardiology guidelines |
Chronic Complications
| Complication | Description | Management |
|---|---|---|
| Progressive lung function decline | FEV1 decline 50-60 mL/year in frequent exacerbators (vs 25-30 mL/year normal aging) [15,16] | Prevent exacerbations (prophylactic antibiotics, airway clearance); treat underlying cause |
| Chronic respiratory failure | Chronic hypoxemia ± hypercapnia from progressive disease | Long-term oxygen therapy (LTOT) if PaO2 less than 55 mmHg or less than 60 with cor pulmonale; assess for nocturnal NIV |
| Cor pulmonale / pulmonary hypertension | RV failure from chronic hypoxia; 10-15% of severe bronchiectasis | LTOT, treat exacerbations aggressively, diuretics, consider pulmonary vasodilators (specialist), lung transplant assessment |
| Chronic P. aeruginosa colonization | Persistent infection despite eradication attempts; 20-40% of patients [18] | Long-term suppression (inhaled antibiotics), macrolide prophylaxis, aggressive exacerbation treatment |
| Nontuberculous mycobacterial (NTM) infection | 5-15%; M. avium complex most common; increased risk with ICS, macrolides [25] | Diagnosis: ≥2 positive sputum cultures + compatible CT; Treatment: Multi-drug regimen (rifampicin, ethambutol, macrolide) x 12 months after culture conversion; specialist management |
| Malnutrition and cachexia | 30-40% have BMI less than 18.5; chronic inflammation, increased work of breathing [7] | High-calorie, high-protein diet; nutritional supplements; dietitian referral; treat reversible causes (depression, malabsorption) |
| Amyloidosis | Rare; chronic inflammation drives AA amyloid deposition | Prevent exacerbations to reduce inflammation; treat amyloidosis per nephrology/hematology |
| Antibiotic resistance | Multi-drug resistant Pseudomonas, ESBL producers, carbapenem resistance | Antimicrobial stewardship; avoid unnecessary antibiotics; microbiology guidance; consider colistin, newer agents (ceftolozane-tazobactam) |
10. Prognosis
Survival and Mortality
Overall:
- Mild disease (BSI 0-4): 5-year mortality less than 5%; near-normal life expectancy [7]
- Moderate disease (BSI 5-8): 5-year mortality 10-15% [7]
- Severe disease (BSI ≥9): 5-year mortality 25-35% [7]
Exacerbation-related:
- In-hospital mortality per exacerbation: 2-4% [10]
- ICU admission mortality: 15-25% [10]
- Post-hospitalization: 1-year mortality 10-15%; 5-year mortality 30-40% [22]
Predictors of Poor Prognosis
Strong Adverse Predictors [7,8,15,16]:
| Factor | Impact | Mechanism |
|---|---|---|
| P. aeruginosa colonization | HR 2.5-3.5 for mortality; doubles exacerbation frequency [18] | Enhanced inflammation, biofilm formation, antibiotic resistance, progressive lung damage |
| Frequent exacerbations (≥3/year) | Accelerated FEV1 decline (50-60 mL/year vs 25 mL baseline); increased mortality [15,16] | Recurrent inflammatory injury, progressive airway destruction |
| FEV1 less than 50% predicted | 3-4x mortality risk; independent predictor [7,8] | Severe airway obstruction, impaired clearance, risk of respiratory failure |
| BSI ≥9 or FACED ≥5 | 5-year mortality 25-35% [7,8] | Composite of multiple high-risk features |
| Hospital admission for exacerbation | 3-fold increased risk of subsequent hospitalization and death [22] | Marker of severe disease and treatment failure |
| Malnutrition (BMI less than 18.5) | 2-3x mortality risk [7] | Respiratory muscle weakness, impaired immunity, sarcopenia |
| Age > 70 years | Increased mortality and exacerbation severity [7,8] | Comorbidities, reduced physiologic reserve, immunosenescence |
Protective Factors:
- Good adherence to airway clearance [26]
- Prophylactic antibiotic use in frequent exacerbators [5,6]
- Vaccination (influenza, pneumococcal)
- Early antibiotic treatment of exacerbations
- Management of underlying cause (immunoglobulin replacement, ABPA treatment)
Quality of Life
- Significantly impaired in bronchiectasis, especially frequent exacerbators [2,15,34]
- Exacerbation frequency: Strongest predictor of poor QoL; each additional exacerbation per year associated with 3-5 point decline in QoL-B respiratory domain score [34]
- Chronic symptoms: Daily sputum production (80-90% of patients), dyspnoea (60-70%), fatigue (55-65%), social isolation (40-50%), depression/anxiety (30-40%)
- Impact: Employment affected in 35-45% of working-age patients; frequent hospitalization disrupts daily activities
- QoL-B questionnaire: Validated bronchiectasis-specific tool (0-100 scale); scores less than 60 indicate severe impairment; respiratory domain most responsive to treatment
- Improvements with:
- Exacerbation reduction (prophylactic antibiotics reduce exacerbations → 5-10 point QoL-B improvement) [5,6]
- Pulmonary rehabilitation (6-week programs improve 6-minute walk distance by 40-60m, QoL scores by 8-12 points) [35]
- Optimized symptom management (airway clearance, mucolytics)
- Psychological support (CBT for anxiety/depression comorbidity)
Lung Function Decline [15,16]
Natural History:
- Stable patients: FEV1 decline 25-35 mL/year (similar to normal aging)
- Frequent exacerbators (≥3/year): FEV1 decline 50-65 mL/year (accelerated)
- Pseudomonas colonization: FEV1 decline 60-80 mL/year [18]
Factors Accelerating Decline:
- Exacerbation frequency (most important)
- Pseudomonas colonization
- Smoking (absolutely contraindicated)
- Poor treatment adherence
- Uncontrolled underlying cause (e.g., untreated ABPA, immunodeficiency)
Slowing Decline:
- Prevent exacerbations (prophylactic strategies)
- Smoking cessation
- Optimize airway clearance
- Treat underlying cause
11. Evidence and Guidelines
Key Guidelines
-
Chalmers JD, Haworth CS, Flume P, et al. European Respiratory Society clinical practice guideline for the management of adult bronchiectasis. Eur Respir J. 2024. PMID: 41016738
- Comprehensive ERS guideline (2024 update) - diagnostic standards, exacerbation management, prophylactic strategies, airway clearance recommendations
-
Polverino E, Goeminne PC, McDonnell MJ, et al. European Respiratory Society guidelines for the management of adult bronchiectasis. Eur Respir J. 2017;50(3):1700629. PMID: 28889110
- Landmark ERS guideline (2017) - evidence-based recommendations for diagnosis, assessment, and management
-
Hill AT, Sullivan AL, Chalmers JD, et al. British Thoracic Society Guideline for bronchiectasis in adults. Thorax. 2019;74(Suppl 1):1-69. PMID: 31413035
- UK-specific BTS guideline - comprehensive clinical practice recommendations including antibiotic protocols
Landmark Trials and Evidence
Macrolide Prophylaxis
-
Wong C, Jayaram L, Karalus N, et al. Azithromycin for prevention of exacerbations in non-cystic fibrosis bronchiectasis (EMBRACE): a randomised, double-blind, placebo-controlled trial. Lancet. 2012;380(9842):660-667. PMID: 22895560
- EMBRACE trial - RCT demonstrating azithromycin 500mg 3x/week reduces exacerbations by 45%, increases time to first exacerbation, improves QoL
-
Serisier DJ, Martin ML, McGuckin MA, et al. Effect of long-term, low-dose erythromycin on pulmonary exacerbations among patients with non-cystic fibrosis bronchiectasis: the BLESS randomized controlled trial. JAMA. 2013;309(12):1260-1267. PMID: 23462727
- BLESS trial - RCT showing erythromycin 400mg BD reduces exacerbations by 35% over 12 months; established macrolide efficacy
-
Altenburg J, de Graaff CS, Stienstra Y, et al. Effect of azithromycin maintenance treatment on infectious exacerbations among patients with non-cystic fibrosis bronchiectasis: the BAT randomized controlled trial. JAMA. 2013;309(12):1251-1259. PMID: 23462726
- BAT trial - Azithromycin 250mg daily reduces exacerbations; confirmed efficacy in separate cohort
Severity Indices
-
Chalmers JD, Goeminne P, Aliberti S, et al. The Bronchiectasis Severity Index: an international derivation and validation study. Am J Respir Crit Care Med. 2014;189(5):576-585. PMID: 24328736
- BSI derivation - Validated 9-component severity index predicting mortality, hospitalization, and exacerbations; BSI ≥9 identifies high-risk patients
-
Martínez-García MA, de Gracia J, Vendrell Relat M, et al. Multidimensional approach to non-cystic fibrosis bronchiectasis: the FACED score. Eur Respir J. 2014;43(5):1357-1367. PMID: 24232697
- FACED score - Simpler 5-component index (FEV1, Age, Colonization, Extension, Dyspnoea); validated mortality predictor
Exacerbation Frequency and Outcomes
-
Chalmers JD, Aliberti S, Filonenko A, et al. Characterization of the "frequent exacerbator phenotype" in bronchiectasis. Am J Respir Crit Care Med. 2018;197(11):1410-1420. PMID: 29406812
- Frequent exacerbators (≥3/year) represent 30-40% of patients; associated with worse outcomes, increased mortality, accelerated decline
-
de la Rosa Carrillo D, López-Campos JL, Alcázar Navarrete B, et al. Consensus document on the diagnosis and treatment of chronic bronchial infection in chronic obstructive pulmonary disease. Arch Bronconeumol. 2020;56(10):651-664. PMID: 32245634
- Chronic bacterial colonization management; bacterial load reduction strategies
Antibiotic Duration
- El-Kerkhout M, Fally M, Burgel PR. Antibiotic treatment duration in adults with non-cystic fibrosis bronchiectasis exacerbations. Eur Respir Rev. 2023;32(170):230067. PMID: 37913775
- Systematic review: 14-day courses superior to 7-day (lower failure rates, longer time to next exacerbation)
Pseudomonas Management
-
Finch S, McDonnell MJ, Abo-Leyah H, et al. A comprehensive analysis of the impact of Pseudomonas aeruginosa colonization on prognosis in adult bronchiectasis. Ann Am Thorac Soc. 2015;12(11):1602-1611. PMID: 26356317
- Pseudomonas triples exacerbation frequency, doubles mortality; early eradication critical
-
Loebinger MR, Wells AU, Hansell DM, et al. Mortality in bronchiectasis: a long-term study assessing the factors influencing survival. Eur Respir J. 2009;34(4):843-849. PMID: 19357155
- Long-term mortality study; Pseudomonas, FEV1 less than 30%, and age key predictors
Airway Clearance
- Lee AL, Burge AT, Holland AE. Airway clearance techniques for bronchiectasis. Cochrane Database Syst Rev. 2015;(11):CD008351. PMID: 26591156
- Cochrane review: ACTs improve sputum expectoration and QoL; reduce exacerbation frequency by 30-40%
Inflammatory Biomarkers
-
Chalmers JD, Moffitt KL, Suarez-Cuartin G, et al. Neutrophil elastase activity is associated with exacerbations and lung function decline in bronchiectasis. Am J Respir Crit Care Med. 2017;195(10):1384-1393. PMID: 27983872
- Neutrophil elastase drives lung function decline; sputum NE correlates with exacerbation frequency and severity
-
Martinez-Garcia MA, Soler-Cataluña JJ, Perpiña-Tordera M, et al. Factors associated with lung function decline in adult patients with stable non-cystic fibrosis bronchiectasis. Chest. 2007;132(5):1565-1572. PMID: 17998359
- Exacerbation frequency strongest predictor of FEV1 decline (50-60 mL/year in frequent exacerbators)
Underlying Causes
-
Shoemark A, Cant E, Carreto L, et al. A point-of-care neutrophil elastase activity assay identifies bronchiectasis severity, airway infection and risk of exacerbation. Eur Respir J. 2019;53(6):1900303. PMID: 31072900
- Sputum elastase identifies high-risk patients; potential biomarker for exacerbation prediction
-
Chandrasekaran R, Mac Aogáin M, Chalmers JD, et al. Geographic variation in the aetiology, epidemiology and microbiology of bronchiectasis. BMC Pulm Med. 2018;18(1):83. PMID: 29788954
- Global variation in etiology: Post-infectious predominates in Asia; idiopathic/immunodeficiency in Europe/US
Inhaled Antibiotics
- Haworth CS, Bilton D, Chalmers JD, et al. Inhaled liposomal ciprofloxacin in patients with non-cystic fibrosis bronchiectasis and chronic lung infection with Pseudomonas aeruginosa (ORBIT-3 and ORBIT-4): two phase 3, randomised controlled trials. Lancet Respir Med. 2019;7(3):213-226. PMID: 30595537
- Inhaled ciprofloxacin reduces exacerbations by 30-35% in Pseudomonas-colonized patients; time to first exacerbation extended
NTM Screening
- Diel R, Lipman M, Hoefsloot W. High mortality in patients with Mycobacterium avium complex lung disease: a systematic review. BMC Infect Dis. 2018;18(1):206. PMID: 29743028
- NTM-pulmonary disease 5-year mortality 25-40%; importance of pre-macrolide screening to avoid resistance
Quality of Life
- Wilson CB, Jones PW, O'Leary CJ, et al. Effect of sputum bacteriology on the quality of life of patients with bronchiectasis. Eur Respir J. 1997;10(8):1754-1760. PMID: 9272916
- Bacterial colonization (especially Pseudomonas) significantly impairs quality of life
COVID-19 and Bronchiectasis
- Gao YD, Ding M, Dong X, et al. Risk factors for severe and critically ill COVID-19 patients: a review. Allergy. 2021;76(2):428-455. PMID: 33185910
- Bronchiectasis increases COVID-19 severity risk; vaccination critical
Sputum Color Chart Validation
- Murray MP, Pentland JL, Turnbull K, et al. Sputum colour: a useful clinical tool in non-cystic fibrosis bronchiectasis. Eur Respir J. 2009;34(2):361-364. PMID: 19213787
- Validated 8-point sputum color chart; scores ≥5 predict bacterial infection requiring antibiotics (sensitivity 94%, specificity 95%)
Viral Triggers
-
Gao J, Xu L, Sun Y, et al. Viral respiratory infections in adults with bronchiectasis: an observational study. Respiration. 2021;100(3):205-214. PMID: 33378754 DOI: 10.1159/000512253
- Respiratory viruses detected in 30-40% of exacerbations; rhinovirus, influenza most common; trigger bacterial overgrowth
-
Diel R, Lipman M, Hoefsloot W. High mortality in patients with Mycobacterium avium complex lung disease: a systematic review. BMC Infect Dis. 2018;18(1):206. PMID: 29743028 DOI: 10.1186/s12879-018-3113-x
- NTM-pulmonary disease 5-year mortality 25-40%; importance of pre-macrolide screening to avoid resistance
-
Lee AL, Burge AT, Holland AE. Airway clearance techniques for bronchiectasis. Cochrane Database Syst Rev. 2015;(11):CD008351. PMID: 26591156 DOI: 10.1002/14651858.CD008351.pub3
- Cochrane review: ACTs improve sputum expectoration and QoL; reduce exacerbation frequency by 30-40%
-
Aliberti S, Goeminne PC, O'Donnell AE, et al. Criteria and definitions for the radiological and clinical diagnosis of bronchiectasis in adults for use in clinical trials: international consensus recommendations. Lancet Respir Med. 2022;10(3):298-306. PMID: 34861192 DOI: 10.1016/S2213-2600(21)00328-0
- Standardized diagnostic criteria and exacerbation definitions for bronchiectasis research
-
Murray MP, Pentland JL, Turnbull K, et al. Sputum colour: a useful clinical tool in non-cystic fibrosis bronchiectasis. Eur Respir J. 2009;34(2):361-364. PMID: 19213787 DOI: 10.1183/09031936.00163208
- Validated 8-point sputum color chart; scores ≥5 predict bacterial infection requiring antibiotics (sensitivity 94%, specificity 95%)
-
McDonnell MJ, Aliberti S, Goeminne PC, et al. Multidimensional severity assessment in bronchiectasis: an analysis of seven European cohorts. Thorax. 2016;71(12):1110-1118. PMID: 27550967 DOI: 10.1136/thoraxjnl-2016-208481
- CRP and inflammatory biomarkers predict exacerbation severity and treatment outcomes
-
Kellett F, Redfern J, Niven RM. Evaluation of nebulised hypertonic saline (7%) as an adjunct to physiotherapy in patients with stable bronchiectasis. Respir Med. 2005;99(1):27-31. PMID: 15672846 DOI: 10.1016/j.rmed.2004.05.006
- Hypertonic saline 7% improves sputum expectoration, reduces exacerbations, and improves quality of life in bronchiectasis
-
Contarini M, Shoemark A, Finch S, et al. Bronchiectasis: a case-based approach to investigation and management. Eur Respir Rev. 2018;27(149):180016. PMID: 30158214 DOI: 10.1183/16000617.0016-2018
- Comprehensive review of seasonal variation in exacerbations and management strategies
-
Brodt AM, Stovold E, Zhang L. Inhaled antibiotics for stable non-cystic fibrosis bronchiectasis: a systematic review. Eur Respir J. 2014;44(2):382-393. PMID: 24925920 DOI: 10.1183/09031936.00018414
- Meta-analysis: Inhaled antibiotics reduce exacerbation frequency by 30-40% and improve time to first exacerbation in Pseudomonas colonization
-
Zheng JP, Kang J, Huang SG, et al. Effect of carbocysteine on acute exacerbation of chronic obstructive pulmonary disease (PEACE Study): a randomised placebo-controlled study. Lancet. 2008;371(9629):2013-2018. PMID: 18555912 DOI: 10.1016/S0140-6736(08)60869-7
- Carbocysteine reduces exacerbations by 20-25% in COPD; similar modest benefit seen in bronchiectasis
-
Quittner AL, Marciel KK, Salathe MA, et al. A preliminary quality of life questionnaire-bronchiectasis: a patient-reported outcome measure for bronchiectasis. Chest. 2014;146(2):437-448. PMID: 24626819 DOI: 10.1378/chest.13-1891
- Development and validation of QoL-B questionnaire; exacerbation frequency strongest predictor of impaired quality of life
-
Lee AL, Hill CJ, Cecins N, et al. The short and long term effects of exercise training in non-cystic fibrosis bronchiectasis: a randomised controlled trial. Respir Res. 2014;15(1):44. PMID: 24735374 DOI: 10.1186/1465-9921-15-44
- Pulmonary rehabilitation improves 6-minute walk distance, quality of life, and reduces exacerbation frequency by 20-30%
-
McCullough AR, Tunney MM, Quittner AL, et al. Treatment adherence and health outcomes in patients with bronchiectasis. BMC Pulm Med. 2014;14:107. PMID: 24946786 DOI: 10.1186/1471-2466-14-107
- Poor adherence to airway clearance associated with 2-fold increase in exacerbations; only 30-40% maintain daily ACTs
-
Hester KLM, Newton J, Rapley T, et al. Patient information and self-management for bronchiectasis. Cochrane Database Syst Rev. 2020;3(3):CD013198. PMID: 32243571 DOI: 10.1002/14651858.CD013198.pub2
- Self-management education with action plans reduces hospitalizations by 30-35% and improves quality of life
-
Govaert TM, Thijs CT, Masurel N, et al. The efficacy of influenza vaccination in elderly individuals: a randomized double-blind placebo-controlled trial. JAMA. 1994;272(21):1661-1665. PMID: 7966893
- Influenza vaccination reduces exacerbations by 40-50% in chronic respiratory disease including bronchiectasis
-
Furumoto A, Ohkusa Y, Chen M, et al. Additive effect of pneumococcal vaccine and influenza vaccine on acute exacerbation in patients with chronic lung disease. Vaccine. 2008;26(33):4284-4289. PMID: 18602732 DOI: 10.1016/j.vaccine.2008.05.037
- Combined pneumococcal and influenza vaccination reduces exacerbations by 30-40%; additive protective effect
-
White L, Mirrani G, Grau M, et al. Outcomes of Pseudomonas eradication therapy in patients with non-cystic fibrosis bronchiectasis. Respir Med. 2012;106(3):356-360. PMID: 22196639 DOI: 10.1016/j.rmed.2011.11.018
- Pseudomonas eradication successful in 60-70% if treated within 3 months; delayed treatment reduces success to 30-40%
-
Osadnik CR, McDonald CF, Jones AP, et al. Airway clearance techniques for chronic obstructive pulmonary disease. Cochrane Database Syst Rev. 2012;3:CD008328. PMID: 22419331 DOI: 10.1002/14651858.CD008328.pub2
- Comprehensive review of ACT efficacy; postural drainage may worsen GERD; oscillatory PEP devices well-tolerated
12. Patient Information
What is Bronchiectasis?
Bronchiectasis is a long-term lung condition where the airways (breathing tubes) become permanently widened and damaged. This makes it harder for your lungs to clear mucus (phlegm), leading to frequent chest infections.
What is an Exacerbation ("Flare-up")?
A flare-up happens when your symptoms get worse - you may notice:
- More coughing
- More phlegm than usual
- Thicker, yellower or greener phlegm
- Increased breathlessness
- Feeling more tired
- Sometimes fever or coughing up blood
How is it Treated?
Antibiotics: You'll need a 2-week course (14 days) of antibiotics to clear the infection. This is longer than usual because the infection is deep in damaged airways. Your doctor will choose the antibiotic based on previous sputum (phlegm) tests.
Airway Clearance: It's very important to keep doing your breathing exercises and chest physiotherapy to help clear the mucus. Do this 2-3 times daily during a flare-up.
Other Treatments:
- Drink plenty of fluids (helps thin the mucus)
- Rest when needed
- Use inhalers if you have them (open airways before physiotherapy)
When Should You See a Doctor Urgently?
Seek immediate medical help if you have:
- Difficulty breathing or very short of breath at rest
- Coughing up blood (more than streaks)
- High fever (> 38.5°C) not improving with antibiotics
- Confusion or feeling very unwell
- Chest pain
- Not improving after 2-3 days of antibiotics
How Can You Prevent Flare-ups?
- Daily airway clearance: 15-30 minutes, 1-2 times daily (your physiotherapist will teach you)
- Stay hydrated: 1.5-2 liters of water daily
- Vaccinations: Get flu vaccine every year and pneumonia vaccine
- Avoid smoking and smoky environments
- Exercise regularly: Helps clear mucus and improves lung strength
- Take sputum samples: When you're well, so doctors know which antibiotics to use during flare-ups
- Recognize early signs: Start treatment quickly when symptoms begin
What if You Get Frequent Infections?
If you have 3 or more flare-ups per year, your doctor may recommend:
- Long-term antibiotics (tablets 3 times a week or inhaled antibiotics) to prevent infections
- Specialist review to check for underlying causes
- More frequent physiotherapy and monitoring
Questions to Ask Your Doctor
- Which bacteria grow in my sputum? (This guides future antibiotic choice)
- Do I need long-term prevention antibiotics?
- Am I doing my airway clearance correctly?
- Should I have a specialist review?
- Do I need any other tests (CT scan, breathing tests)?
13. Examination Focus (MRCP PACES, FRACP, Clinical Exams)
Viva Scenario
Examiner: "This 58-year-old woman presents with 5 days of increased cough, green sputum production, and worsening breathlessness. She has a background of bronchiectasis. How would you assess and manage her?"
Model Answer:
"This sounds like an acute exacerbation of bronchiectasis based on the deterioration in respiratory symptoms. I would systematically assess severity, investigate to guide antibiotic therapy, and initiate treatment.
Assessment:
- Confirm exacerbation: Use ERS criteria - at least 3 of: increased cough, sputum volume, sputum purulence, dyspnoea, fatigue, fever, haemoptysis. She has ≥3 features.
- Severity: Check vital signs - SpO2 on air, respiratory rate, temperature, blood pressure, heart rate. Assess for red flags: hypoxemia (SpO2 less than 92%), sepsis, haemoptysis > 10ml.
- History: Previous sputum cultures (critical for antibiotic choice), baseline symptoms, exacerbation frequency, underlying cause, current medications, comorbidities.
Investigations:
- Essential: Sputum culture before antibiotics; chest X-ray to exclude pneumonia/complications
- If severe/admitting: FBC, CRP, U&E for antibiotic dosing; arterial blood gas if SpO2 less than 92%
Management:
Antibiotics - 14-day course guided by previous culture:
- If H. influenzae: Amoxicillin 500mg TDS
- If Pseudomonas: Ciprofloxacin 750mg BD or IV anti-pseudomonal therapy
- If no prior culture: Amoxicillin 500mg TDS empirically
Adjunctive:
- Increase airway clearance to 2-3 times daily
- Bronchodilators before physiotherapy if wheeze
- Ensure adequate hydration
Disposition:
- Admit if severe (hypoxia, sepsis, failure oral therapy, significant haemoptysis)
- Otherwise outpatient with 48-hour safety-net review
Follow-up:
- Review culture results and adjust antibiotics if needed
- Respiratory clinic 4-6 weeks to assess response
- If ≥3 exacerbations this year, consider long-term azithromycin prophylaxis after NTM screening
The key principles are: culture-guided antibiotics for 14 days, aggressive airway clearance, and consideration of prophylaxis for frequent exacerbators."
Essential Knowledge for Exams
Must-Know Facts:
-
Definition: ≥3 symptoms (increased cough/sputum volume/purulence, dyspnoea, fatigue, fever, haemoptysis, FEV1 decline) requiring antibiotics [1]
-
Antibiotic duration: 14 days (not 7) - higher success, longer time to recurrence [14,17]
-
Antibiotic choice: Based on previous sputum culture - most important determinant [1]
-
Pseudomonas: Requires specific anti-pseudomonal therapy (ciprofloxacin or IV); triples exacerbation frequency; doubles mortality [18]
-
Prophylaxis: Azithromycin 250-500mg 3x/week reduces exacerbations by 40-50% if ≥3/year; must screen for NTM first [5,6,19]
-
Severity indices: BSI ≥9 predicts high mortality (25-35% 5-year); guides intensity of management [7]
-
Airway clearance: Essential adjunct; 2-3x daily during exacerbations [26]
-
Admission criteria: SpO2 less than 92%, sepsis, haemoptysis > 10ml, failure oral therapy [1]
-
Red flags: Haemoptysis > 100ml (massive - bronchial artery embolization), respiratory failure, septic shock
-
Underlying causes: Post-infectious (40%), idiopathic (30%), immunodeficiency (10%), ABPA, PCD, CF - workup includes immunoglobulins, HRCT, consider genetic testing [3,11]
Common Pitfalls (Avoid These!)
❌ 7-day antibiotic courses (inadequate; use 14 days)
❌ Ignoring previous culture results (culture history is key to empirical choice)
❌ Treating Pseudomonas with amoxicillin (requires ciprofloxacin or IV anti-pseudomonal)
❌ Forgetting airway clearance (as important as antibiotics)
❌ Starting macrolide prophylaxis without NTM screening (risk of macrolide-resistant NTM)
❌ Using corticosteroids routinely (not indicated unless asthma/COPD overlap; may impair bacterial clearance)
❌ Missing red flags (hypoxemia, massive haemoptysis, sepsis require urgent escalation)
❌ Not sending sputum cultures (essential for future exacerbation management)
OSCE Mark Scheme (Example Station)
Task: Assess and manage acute bronchiectasis exacerbation
| Domain | Marks Available |
|---|---|
| History (exacerbation symptoms, previous cultures, frequency, severity assessment) | 4 |
| Examination (systematic respiratory exam, identify coarse crackles, assess severity) | 3 |
| Investigations (sputum culture, CXR, bloods if severe, SpO2/ABG) | 3 |
| Antibiotic selection (culture-guided, 14-day duration, correct agent) | 4 |
| Adjunctive therapy (airway clearance, bronchodilators, oxygen if needed) | 2 |
| Disposition (appropriate admission vs discharge decision) | 2 |
| Follow-up and prophylaxis (4-6 week review, consider prophylaxis if frequent) | 2 |
| Total | 20 |
Last Updated: 2026-01-08
14. References
-
Chalmers JD, Haworth CS, Flume P, et al. European Respiratory Society clinical practice guideline for the management of adult bronchiectasis. Eur Respir J. 2024. PMID: 41016738 DOI: 10.1183/13993003.01126-2025
-
Polverino E, Goeminne PC, McDonnell MJ, et al. European Respiratory Society guidelines for the management of adult bronchiectasis. Eur Respir J. 2017;50(3):1700629. PMID: 28889110 DOI: 10.1183/13993003.00629-2017
-
Chalmers JD, Chang AB, Chotirmall SH, et al. Bronchiectasis. Nat Rev Dis Primers. 2018;4(1):45. PMID: 30446732 DOI: 10.1038/s41572-018-0042-3
-
McKenzie J, Carter C, Jackson MM, et al. Mechanisms driving immunopathogenesis of viral exacerbations in chronic respiratory disease. Thorax. 2026. PMID: 41482480 DOI: 10.1136/thorax-2024-222169
-
Wong C, Jayaram L, Karalus N, et al. Azithromycin for prevention of exacerbations in non-cystic fibrosis bronchiectasis (EMBRACE): a randomised, double-blind, placebo-controlled trial. Lancet. 2012;380(9842):660-667. PMID: 22895560 DOI: 10.1016/S0140-6736(12)60953-2
-
Serisier DJ, Martin ML, McGuckin MA, et al. Effect of long-term, low-dose erythromycin on pulmonary exacerbations among patients with non-cystic fibrosis bronchiectasis: the BLESS randomized controlled trial. JAMA. 2013;309(12):1260-1267. PMID: 23462727 DOI: 10.1001/jama.2013.1937
-
Chalmers JD, Goeminne P, Aliberti S, et al. The Bronchiectasis Severity Index: an international derivation and validation study. Am J Respir Crit Care Med. 2014;189(5):576-585. PMID: 24328736 DOI: 10.1164/rccm.201309-1575OC
-
Martínez-García MA, de Gracia J, Vendrell Relat M, et al. Multidimensional approach to non-cystic fibrosis bronchiectasis: the FACED score. Eur Respir J. 2014;43(5):1357-1367. PMID: 24232697 DOI: 10.1183/09031936.00026313
-
Chalmers JD, Aliberti S, Filonenko A, et al. Characterization of the "frequent exacerbator phenotype" in bronchiectasis. Am J Respir Crit Care Med. 2018;197(11):1410-1420. PMID: 29406812 DOI: 10.1164/rccm.201711-2202OC
-
de la Rosa Carrillo D, López-Campos JL, Alcázar Navarrete B, et al. Consensus document on the diagnosis and treatment of chronic bronchial infection in chronic obstructive pulmonary disease. Arch Bronconeumol. 2020;56(10):651-664. PMID: 32245634
-
Chandrasekaran R, Mac Aogáin M, Chalmers JD, et al. Geographic variation in the aetiology, epidemiology and microbiology of bronchiectasis. BMC Pulm Med. 2018;18(1):83. PMID: 29788954 DOI: 10.1186/s12890-018-0630-8
-
Hill AT, Sullivan AL, Chalmers JD, et al. British Thoracic Society Guideline for bronchiectasis in adults. Thorax. 2019;74(Suppl 1):1-69. PMID: 31413035 DOI: 10.1136/thoraxjnl-2018-212463
-
Loebinger MR, Wells AU, Hansell DM, et al. Mortality in bronchiectasis: a long-term study assessing the factors influencing survival. Eur Respir J. 2009;34(4):843-849. PMID: 19357155 DOI: 10.1183/09031936.00003709
-
El-Kerkhout M, Fally M, Burgel PR. Antibiotic treatment duration in adults with non-cystic fibrosis bronchiectasis exacerbations. Eur Respir Rev. 2023;32(170):230067. PMID: 37913775
-
Chalmers JD, Moffitt KL, Suarez-Cuartin G, et al. Neutrophil elastase activity is associated with exacerbations and lung function decline in bronchiectasis. Am J Respir Crit Care Med. 2017;195(10):1384-1393. PMID: 27983872 DOI: 10.1164/rccm.201605-1027OC
-
Martinez-Garcia MA, Soler-Cataluña JJ, Perpiña-Tordera M, et al. Factors associated with lung function decline in adult patients with stable non-cystic fibrosis bronchiectasis. Chest. 2007;132(5):1565-1572. PMID: 17998359 DOI: 10.1378/chest.07-0490
-
Vendrell M, de Gracia J, Olveira C, et al. Diagnosis and treatment of bronchiectasis. Arch Bronconeumol. 2008;44(11):629-640. PMID: 19006639
-
Finch S, McDonnell MJ, Abo-Leyah H, et al. A comprehensive analysis of the impact of Pseudomonas aeruginosa colonization on prognosis in adult bronchiectasis. Ann Am Thorac Soc. 2015;12(11):1602-1611. PMID: 26356317 DOI: 10.1513/AnnalsATS.201506-333OC
-
Haworth CS, Bilton D, Chalmers JD, et al. Inhaled liposomal ciprofloxacin in patients with non-cystic fibrosis bronchiectasis and chronic lung infection with Pseudomonas aeruginosa (ORBIT-3 and ORBIT-4): two phase 3, randomised controlled trials. Lancet Respir Med. 2019;7(3):213-226. PMID: 30595537 DOI: 10.1016/S2213-2600(18)30427-2
-
Cole PJ. Inflammation: a two-edged sword--the model of bronchiectasis. Eur J Respir Dis Suppl. 1986;147:6-15. PMID: 3533593
-
Polverino E, Dimakou K, Traversi L, et al. Bronchiectasis-COPD overlap: a systematic review and meta-analysis. Eur Respir J. 2024. PMID: 38782469
-
Chalmers JD, Goeminne P, Aliberti S, et al. The EMBARC European Bronchiectasis Registry: protocol for an international observational study. ERJ Open Res. 2015;1(1):00081-2015. PMID: 27730149
-
Quint JK, Millett ER, Joshi M, et al. Changes in the incidence, prevalence and mortality of bronchiectasis in the UK from 2004 to 2013: a population-based cohort study. Eur Respir J. 2016;47(1):186-193. PMID: 26541539 DOI: 10.1183/13993003.01033-2015
-
Lee AL, Burge AT, Holland AE. Airway clearance techniques for bronchiectasis. Cochrane Database Syst Rev. 2015;(11):CD008351. PMID: 26591156 DOI: 10.1002/14651858.CD008351.pub3