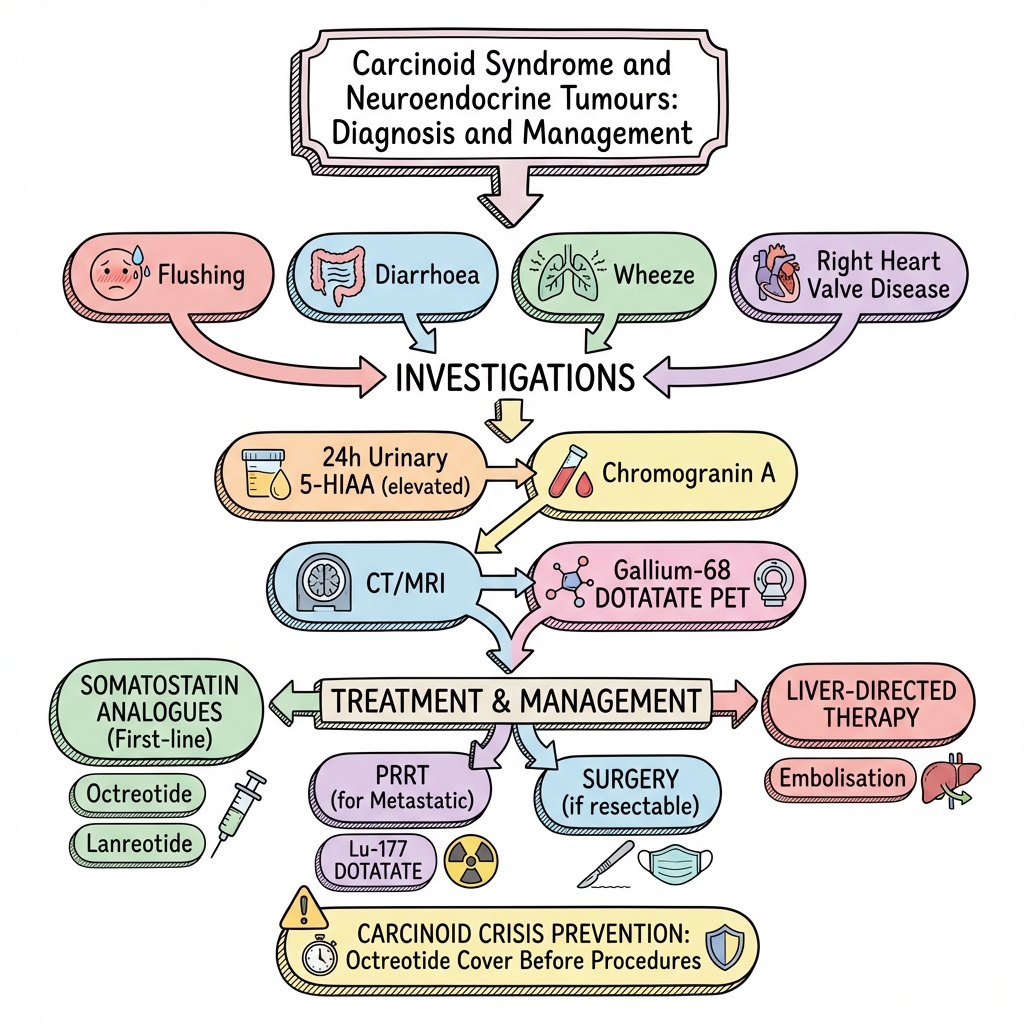
Carcinoid Syndrome and Neuroendocrine Tumours
Neuroendocrine tumours (NETs) represent a heterogeneous group of neoplasms arising from neuroendocrine cells distributed... MRCP exam preparation.
What matters first
Neuroendocrine tumours (NETs) represent a heterogeneous group of neoplasms arising from neuroendocrine cells distributed... MRCP exam preparation.
Carcinoid crisis
5 Jan 2026
Generated educational material; verify before clinical use.
Visible references section
See the concept before reading it
Study the key anatomy, imaging, and decision pathways as full teaching plates.
Clinical board
A visual summary of the highest-yield teaching signals on this page.
Urgent signals
Safety-critical features pulled from the topic metadata.
- Carcinoid crisis
- Carcinoid heart disease
- Bowel obstruction
- Rapid tumour progression
Exam focus
Current exam surfaces linked to this topic.
- MRCP
Linked comparisons
Differentials and adjacent topics worth opening next.
- VIPoma
- Phaeochromocytoma
Content status and exam context
This page is AI-generated educational content. It may contain errors or omissions and is not a substitute for current guidelines, local protocols, senior clinical judgement, or professional medical advice.
MedVellum does not claim an individual clinician reviewer, board certification, or professional credential for this page unless a future version names a real, verifiable reviewer.
Clinical explanation and evidence
Carcinoid Syndrome and Neuroendocrine Tumours
1. Overview
Neuroendocrine tumours (NETs) represent a heterogeneous group of neoplasms arising from neuroendocrine cells distributed throughout the body, with the highest concentration in the gastrointestinal tract and lungs. These cells are capable of producing and secreting various hormones and bioactive substances. The term "carcinoid" historically referred to these tumours due to their relatively indolent behaviour compared to conventional adenocarcinomas, though this terminology is being replaced by the more precise classification of neuroendocrine tumours. [1]
Carcinoid syndrome is a distinctive paraneoplastic syndrome that occurs in 10-20% of patients with NETs, typically when tumours metastasise to the liver or originate in sites that drain directly into the systemic circulation, bypassing hepatic first-pass metabolism. [2] The syndrome is characterised by episodic flushing, secretory diarrhoea, bronchospasm, and right-sided cardiac valve disease, resulting from the release of bioactive substances including serotonin, tachykinins, histamine, and prostaglandins into the systemic circulation. [3]
The clinical significance of NETs extends beyond their hormonal effects. While often slow-growing, they represent a spectrum of malignancies ranging from indolent, well-differentiated tumours with excellent long-term survival to aggressive, poorly differentiated neuroendocrine carcinomas with dismal prognosis. Understanding the biological behaviour, classification systems, and therapeutic approaches is essential for optimal patient management. [4]
2. Epidemiology
Incidence and Prevalence
The incidence of neuroendocrine tumours has increased substantially over the past four decades, with current estimates suggesting 6.98 per 100,000 population annually in the United States, representing a more than 6-fold increase since the 1970s. [5] This dramatic rise is largely attributed to increased detection of small, incidental tumours through widespread use of cross-sectional imaging and endoscopic procedures, though some true increase in incidence may also contribute. [6]
The prevalence is estimated at approximately 35 per 100,000 population, reflecting the relatively indolent nature of many NETs and prolonged survival times. [5] However, only 10-20% of patients with NETs develop carcinoid syndrome, and this typically occurs in the setting of hepatic metastases or when primary tumours drain outside the portal venous system. [2,7]
| Statistic | Value | Source |
|---|---|---|
| Annual incidence (all NETs) | 6.98 per 100,000 | [5] |
| Prevalence | 35 per 100,000 | [5] |
| Proportion developing syndrome | 10-20% | [2,7] |
| 5-year survival (localised) | 89-97% | [8] |
| 5-year survival (distant metastases) | 25-42% | [8] |
Demographics and Risk Factors
NETs demonstrate a slight female predominance (female:male ratio approximately 1.2:1) and typically present in the sixth decade of life, with median age at diagnosis of 60-65 years. [5] However, age at presentation varies considerably by tumour location and functional status.
Risk Factors:
- Genetic syndromes: Multiple Endocrine Neoplasia type 1 (MEN-1) accounts for approximately 10% of gastroduodenal NETs and predisposes to pancreatic NETs, with lifetime risk exceeding 50%. [9]
- Chronic atrophic gastritis: Increases risk of type 1 gastric carcinoids through hypergastrinaemia-induced enterochromaffin-like cell hyperplasia. [10]
- Inflammatory bowel disease: Modest increased risk, particularly for small bowel NETs. [11]
- Ethnicity: Highest incidence reported in Black populations (8.48 per 100,000) compared to White (6.84 per 100,000) and Asian populations. [5]
Anatomical Distribution
Small intestine NETs represent the most common primary site for tumours causing carcinoid syndrome (44% of all NETs), followed by lung (25%), rectum (16%), and pancreas (7%). [5,12] However, carcinoid syndrome is predominantly associated with small bowel primaries (particularly jejunoileal) due to their propensity for liver metastases and high serotonin production. [13]
3. Aetiology and Pathophysiology
Neuroendocrine Cell Origin and Differentiation
Neuroendocrine cells are specialised epithelial cells distributed throughout the body, with the highest density in the diffuse neuroendocrine system of the gastrointestinal tract and respiratory system. These cells share characteristics with both endocrine cells (hormone secretion) and neurons (dense core secretory granules, neural markers). [1]
NETs arise through transformation of these neuroendocrine cells, though the precise molecular events triggering neoplastic change remain incompletely understood. Unlike many epithelial malignancies, NETs infrequently harbour mutations in common oncogenes (e.g., KRAS, BRAF) or tumour suppressors (e.g., TP53). Instead, they demonstrate distinctive genetic alterations:
- Chromosome 18 loss: Present in up to 88% of small intestinal NETs, involving loss of SMAD4 and other tumour suppressor genes. [14]
- MEN1 mutations: Found in 44% of pancreatic NETs (sporadic cases) and essentially all MEN-1 syndrome-associated tumours. [15]
- Chromatin remodelling genes: Mutations in DAXX and ATRX occur in approximately 43% of pancreatic NETs and predict longer survival. [16]
Molecular Pathogenesis - Advanced Detail:
The pathogenesis of NETs involves complex interactions between genetic alterations, epigenetic modifications, and signalling pathway dysregulation:
Genetic Alterations by Site:
- Pancreatic NETs: MEN1 (44%), DAXX/ATRX (43%), mTOR pathway genes (15%)
- Small bowel NETs: Chromosome 18q loss (88%), CDKN1B mutations (8%)
- Lung NETs: MEN1 mutations (rare in typical carcinoids, common in atypical carcinoids)
Key Signalling Pathways:
- mTOR pathway: Dysregulated in pancreatic NETs, providing rationale for everolimus therapy
- PI3K/AKT pathway: Activated in subset of NETs
- VEGF pathway: Contributes to hypervascular phenotype, targeted by sunitinib
- Somatostatin receptor expression: SSTR2 most commonly expressed, basis for somatostatin analogue therapy and molecular imaging
Epigenetic Factors:
- DNA methylation patterns distinguish NET subtypes
- Histone modifications affect tumour behaviour
- MicroRNA dysregulation influences proliferation and metastasis
Hormonal Production and Carcinoid Syndrome Pathophysiology
Neuroendocrine tumours are capable of synthesising and secreting numerous bioactive substances. Carcinoid syndrome results from systemic exposure to these mediators, which occurs when hepatic metabolism is bypassed. [3]
Key Mediators and Effects:
-
Serotonin (5-hydroxytryptamine): Principal mediator, produced from tryptophan via 5-hydroxytryptophan
- Effects: Diarrhoea (increased intestinal motility and secretion), fibrosis (valvular and mesenteric), possibly flushing
- Metabolism: Converted to 5-hydroxyindoleacetic acid (5-HIAA) by monoamine oxidase and aldehyde dehydrogenase
- Clinical correlation: Urinary 5-HIAA serves as diagnostic biomarker [17]
-
Tachykinins (substance P, neurokinin A, neuropeptide K):
- Effects: Flushing (vasodilation), bronchospasm, intestinal motility
- Particularly elevated: In foregut NETs and during carcinoid crisis [18]
-
Histamine: Particularly from gastric carcinoids
- Effects: Flushing, pruritus, gastric acid hypersecretion
- Clinical correlation: Responds to histamine receptor antagonists
-
Prostaglandins: Contribute to diarrhoea and flushing
- May explain partial response to prostaglandin inhibitors (NSAIDs) in some patients
-
Kallikrein: Produces bradykinin
- Effects: Hypotension, flushing, bronchospasm
- Clinical significance: May contribute to carcinoid crisis
Hepatic Metabolism Bypass Mechanism
Under normal circumstances, bioactive substances produced by intestinal NETs are released into the portal venous system and undergo first-pass hepatic metabolism, where they are efficiently inactivated. Carcinoid syndrome develops when this protective mechanism is circumvented by: [19]
- Hepatic metastases: Allow direct secretion into hepatic veins and systemic circulation
- Retroperitoneal drainage: Tumours draining via lumbar or ovarian veins bypass portal system
- High tumour burden: Overwhelms hepatic metabolic capacity
- Primary sites outside portal drainage: Lung, ovarian, retroperitoneal primaries release directly into systemic circulation
Carcinoid Heart Disease Pathophysiology
Carcinoid heart disease develops in 20-50% of patients with carcinoid syndrome and represents a major determinant of morbidity and mortality. [20] The pathophysiology involves serotonin and other mediators inducing fibroblast proliferation and deposition of fibrous plaques on valvular and endocardial surfaces.
Characteristic Features:
- Right-sided predominance: Left heart usually spared due to pulmonary inactivation of mediators
- Valvular lesions: Tricuspid regurgitation (> 90% of cases), pulmonary stenosis (50-80%), less commonly pulmonary regurgitation
- Plaque composition: Smooth muscle cells, myofibroblasts, and extracellular matrix on valve surfaces
- Left heart involvement: Rare (less than 10%), associated with high mediator levels, patent foramen ovale, or bronchial NETs [21]
Detailed Mechanisms of Carcinoid Heart Disease:
Molecular Mechanisms:
- 5-HT2B receptor activation on cardiac fibroblasts triggers TGF-β signalling
- Promotes fibroblast proliferation and collagen deposition
- Valvular endothelial cells undergo endothelial-to-mesenchymal transition
- Results in thickened, retracted, immobile valve leaflets
Risk Factors for Development:
- High urinary 5-HIAA levels (> 300 μmol/24h carries significantly increased risk)
- Prolonged duration of carcinoid syndrome
- Tachykinin elevation
- High plasma NT-proBNP levels
Haemodynamic Consequences:
- Tricuspid regurgitation leads to right atrial dilatation, elevated CVP
- Pulmonary stenosis causes RV hypertrophy, reduced cardiac output
- Progressive RV dysfunction and right heart failure
- Eventual hepatic congestion and peripheral oedema
Timing and Prevention:
- Develops typically after years of untreated syndrome
- Adequate symptom control with SSAs may reduce risk
- Telotristat (tryptophan hydroxylase inhibitor) potentially cardioprotective
4. Clinical Presentation
Classic Carcinoid Syndrome Features
The cardinal manifestations of carcinoid syndrome result from systemic effects of secreted bioactive substances. Symptom severity varies from mild and intermittent to debilitating and constant, with progression over time if untreated. [22]
Major Clinical Features:
| Feature | Frequency | Characteristics | Triggers |
|---|---|---|---|
| Flushing | 85-95% | Sudden, red-purple discolouration of face and upper trunk; 2-5 minutes duration | Alcohol, stress, tyramine-rich foods, physical exertion |
| Diarrhoea | 70-80% | Secretory, watery, up to 10-20 bowel movements daily; nocturnal symptoms common | May be constant or episodic |
| Abdominal pain | 40-50% | Cramping, associated with diarrhoea or mesenteric ischaemia/fibrosis | Variable |
| Bronchospasm | 10-20% | Wheezing, dyspnoea; may be severe | Same as flushing triggers |
| Carcinoid heart disease | 20-50% | Progressive dyspnoea, peripheral oedema, ascites | Develops over years |
Flushing Characteristics by Tumour Location
Flushing patterns vary according to primary tumour site and mediator profile: [23]
- Midgut NETs (small intestine, appendix): Brief (2-5 min), cyanotic-red, affecting face and upper chest; rarely accompanied by lacrimation or periorbital oedema
- Foregut NETs (bronchial, gastric): Prolonged (hours), bright red, patchy distribution; often accompanied by periorbital oedema, lacrimation, and facial swelling; may cause prominent dermatographism
- Hindgut NETs (rectum): Carcinoid syndrome extremely rare
Diarrhoea Pattern and Mechanism
Carcinoid diarrhoea is characteristically secretory, persisting despite fasting, and associated with: [24]
- High volume: May exceed 2-3 litres daily in severe cases
- Watery consistency: Without blood or mucus (unless concomitant bowel obstruction)
- Nocturnal symptoms: Distinguishes from functional bowel disorders
- Electrolyte abnormalities: Hypokalemia, metabolic acidosis in severe cases
- Steatorrhoea: May occur due to rapid transit or mesenteric ischaemia
Mechanisms:
- Serotonin-induced increased intestinal secretion and motility
- Vasoactive intestinal peptide (VIP) co-secretion in some cases
- Bile salt malabsorption from rapid transit
- Mesenteric vascular insufficiency from tumour-associated fibrosis
Carcinoid Heart Disease Clinical Features
Cardiac manifestations develop insidiously, often attributed initially to other causes. [20,21]
Clinical Presentation:
- Early stage: Fatigue, reduced exercise tolerance (subtle findings)
- Established disease: Progressive dyspnoea on exertion, peripheral oedema
- Advanced disease: Right heart failure with ascites, hepatomegaly, elevated jugular venous pressure
Examination Findings:
- Tricuspid regurgitation: Pansystolic murmur at left sternal edge, increased with inspiration (Carvallo's sign), prominent CV wave in JVP, hepatic pulsation
- Pulmonary stenosis: Ejection systolic murmur at left upper sternal edge, may have ejection click
- Right ventricular heave: Parasternal lift indicating RV hypertrophy
- Signs of right heart failure: Peripheral oedema, ascites, hepatomegaly
Non-Classical Presentations
Asymptomatic/Incidental NETs: Many NETs are discovered incidentally during imaging or surgery for unrelated indications. Small (less than 2 cm) rectal NETs are frequently found during screening colonoscopy. Appendiceal NETs are discovered in approximately 0.3% of appendectomy specimens. [25]
Mesenteric Fibrosis and Ischaemia: Fibrotic reaction in mesentery can cause:
- Intestinal kinking and obstruction
- Mesenteric vascular compromise with chronic ischaemia
- Abdominal angina-type pain after meals
- CT findings of mesenteric mass with radiating strands and calcification [26]
Pellagra-like Syndrome: Excessive conversion of dietary tryptophan to serotonin depletes substrate for niacin synthesis, causing pellagra (dermatitis, diarrhoea, dementia) in approximately 5% of patients with extensive disease and high serotonin production. [27]
Carcinoid Crisis - Critical Emergency:
Carcinoid crisis represents a life-threatening manifestation characterised by severe flushing, profound hypotension, bronchospasm, tachycardia, and altered mental status. [28]
Triggers:
- Anaesthesia induction and surgical manipulation of tumour
- Chemotherapy or embolisation of tumour
- Stress (sepsis, trauma)
- Medications (catecholamines, which paradoxically may worsen hypotension)
Pathophysiology: Massive, uncontrolled release of mediators overwhelms compensatory mechanisms, causing:
- Profound vasodilation and distributive shock
- Severe bronchospasm impairing ventilation
- Myocardial depression
- Increased capillary permeability
Clinical Features:
- Intense generalised flushing (may become cyanotic)
- Hypotension (SBP less than 90 mmHg), refractory to fluid resuscitation
- Tachycardia (> 120 bpm)
- Severe bronchospasm, respiratory distress
- Altered consciousness, confusion
- May progress to cardiovascular collapse
Management:
- Immediate octreotide: 100-500 μg IV bolus, followed by continuous infusion (50-100 μg/h)
- Fluid resuscitation: Cautious, monitor for fluid overload in heart disease
- Bronchodilators: Salbutamol for bronchospasm
- Avoid catecholamines: May worsen mediator release; if vasopressor needed, vasopressin preferred
- ICU monitoring: Invasive haemodynamic monitoring, mechanical ventilation if required
Prevention:
- Pre-operative octreotide: Start 100-500 μg SC 2-4 hours before surgery, or continuous infusion
- Maintain throughout perioperative period
- Gradually wean postoperatively over 48-72 hours
5. Differential Diagnosis
The diverse manifestations of carcinoid syndrome necessitate broad differential consideration, particularly for flushing and diarrhoea, which are common symptoms with numerous causes.
Flushing Differential Diagnosis
| Condition | Distinguishing Features | Key Investigations |
|---|---|---|
| Menopause | Hot flashes typically last less than 5 min, upper body distribution, associated with night sweats; age 45-55 years | FSH, oestradiol |
| Phaeochromocytoma | Flushing accompanied by severe headache, palpitations, hypertension; paroxysmal symptoms | 24h urinary metanephrines, plasma metanephrines |
| Medullary thyroid carcinoma | Flushing less prominent; may have neck mass, diarrhoea, elevated calcitonin | Serum calcitonin, CEA; neck ultrasound |
| Mastocytosis | Flushing with pruritus, urticaria, anaphylaxis risk; Darier's sign | Serum tryptase, bone marrow biopsy |
| VIPoma | Profuse watery diarrhoea dominant (> 3L/day), hypokalaemia, achlorhydria (WDHA syndrome) | Serum VIP, fasting gut hormones |
| Drug-induced | Niacin, calcium channel blockers, nitrates; temporal relationship with medication | Medication history |
| Alcohol/foods | Alcohol flushing (especially with disulfiram), scromboid poisoning, spicy foods | History, exclusion diet |
| Renal cell carcinoma | May produce PTHrP or other mediators; haematuria, flank mass | Renal imaging, urinalysis |
| Rosacea | Persistent facial erythema, telangiectasia, papulopustular lesions | Clinical diagnosis |
Diarrhoea Differential Diagnosis
Secretory Diarrhoea (continues during fasting):
- VIPoma: Extreme volume (> 3L), severe hypokalaemia, achlorhydria
- Gastrinoma: Associated peptic ulcer disease, oesophagitis; elevated fasting gastrin
- Medullary thyroid carcinoma: Elevated calcitonin
- Bile acid malabsorption: Responds to bile acid sequestrants; SeHCAT scan
- Microscopic colitis: Chronic watery diarrhoea; normal colonoscopy, abnormal histology
- Addison's disease: Hyperpigmentation, hypotension, electrolyte abnormalities
Osmotic/Malabsorptive Diarrhoea (ceases with fasting):
- Coeliac disease: Anti-TTG antibodies, duodenal biopsy
- Lactose intolerance: Hydrogen breath test
- Pancreatic insufficiency: Faecal elastase, imaging for chronic pancreatitis
Inflammatory Diarrhoea:
- Inflammatory bowel disease: Blood/mucus in stool, elevated CRP/faecal calprotectin
Bronchospasm Differential
- Asthma: Usually without flushing/diarrhoea; reversibility testing
- COPD: Smoking history, persistent obstruction
- Anaphylaxis: Acute onset, urticaria, hypotension, identifiable trigger
- Angiotensin-converting enzyme inhibitor-induced cough/bronchospasm: Medication history
Cardiac Differential
Right-sided valvular disease requires consideration of:
- Rheumatic heart disease: Usually left-sided predominant; history of rheumatic fever
- Infective endocarditis: Fever, positive blood cultures, vegetations on echo
- Congenital heart disease: Ebstein's anomaly, ASD
- Ergot-derivative drugs: Parkinson's disease medications (pergolide, cabergoline)
- Fenfluramine-phentermine (historical)
- Endomyocardial fibrosis: Tropical eosinophilia, hypereosinophilic syndrome
6. Investigations
Biochemical Confirmation
Urinary 5-Hydroxyindoleacetic Acid (5-HIAA):
The 24-hour urinary 5-HIAA remains the gold standard biochemical test for carcinoid syndrome diagnosis, with sensitivity 73% and specificity 100% when significantly elevated. [17,29]
Protocol:
- Collect 24-hour urine with acid preservative
- Avoid interfering substances for 48-72 hours prior:
- "Foods: Bananas, pineapple, kiwi, plums, avocados, walnuts, pecans, aubergine (all high in serotonin)"
- "Medications: Paracetamol (acetaminophen), caffeine, nicotine, guaifenesin, phenobarbital"
- Normal range: less than 40 μmol/24h (varies by laboratory)
- Diagnostic threshold: > 2-3 times upper limit of normal highly suggestive
- Limitations:
- Foregut NETs may not produce serotonin (false negative)
- Hindgut NETs rarely produce serotonin
- Renal impairment may cause false elevation
Chromogranin A (CgA):
A non-specific marker secreted by neuroendocrine cells; elevated in 60-80% of NET patients but lacks specificity for carcinoid syndrome. [30]
Interpretation:
- Useful for diagnosis, monitoring treatment response, detecting recurrence
- Correlates with tumour burden
- False positives: Proton pump inhibitors (most common), atrophic gastritis, renal failure, hypertension, inflammatory bowel disease, other neuroendocrine conditions
- Recommendation: Discontinue PPIs for 2 weeks before testing (use H2 antagonists if needed)
- Rising levels suggest disease progression
Other Biomarkers:
| Test | Indication | Interpretation |
|---|---|---|
| Plasma/urinary serotonin | Alternative to 5-HIAA | Less commonly used; difficult assay |
| Pancreastatin | More specific than CgA | May predict disease progression |
| Neuron-specific enolase | Poorly differentiated NECs | Non-specific |
| Plasma NT-proBNP | Cardiac involvement screening | Elevated in carcinoid heart disease |
| Plasma tachykinins | Research setting | Not widely available |
Anatomical Localisation
Computed Tomography (CT) - Multiphasic Protocol:
Dedicated NET protocol CT with arterial and portal venous phases essential for detection, as NETs are typically hypervascular. [31]
Findings:
- Primary tumour: Hypervascular mass, often small (less than 2 cm)
- Liver metastases: Multiple hypervascular lesions, best seen in arterial phase
- Mesenteric involvement: Spiculated mesenteric mass with radiating strands, calcification
- Complications: Bowel obstruction, mesenteric vascular involvement
Sensitivity: 73-80% for primary tumour detection, > 90% for liver metastases
Magnetic Resonance Imaging (MRI):
Superior soft tissue characterisation, particularly for liver metastases. [32]
Protocol: T1, T2, diffusion-weighted imaging (DWI), dynamic contrast enhancement Liver metastases appearance:
- T2 hyperintense
- Arterial enhancement with washout
- Restricted diffusion on DWI Advantages: No ionising radiation, superior liver lesion characterisation, better for small lesions
68Gallium-DOTATATE PET/CT:
The most sensitive imaging modality for NETs, exploiting somatostatin receptor (SSTR) expression. [33,34]
Mechanism: 68Ga-labelled somatostatin analogue binds to SSTRs (particularly SSTR2), allowing detection of NET deposits Sensitivity/Specificity: 93-96% sensitivity, 91-98% specificity for NET detection Advantages:
- Whole-body imaging in single study
- Detects disease missed on conventional imaging in 30-60% of cases
- Essential for selecting patients for peptide receptor radionuclide therapy (PRRT)
- Identifies unsuspected sites of disease (bone, peritoneum)
Krenning Score (SSTR expression intensity):
- Grade 0: No uptake
- Grade 1: Very low uptake
- Grade 2: Uptake ≤ liver
- Grade 3: Uptake > liver
- Grade 4: Uptake >> liver
PRRT eligibility: Requires Krenning score ≥2 (uptake at least equal to liver)
18F-FDG PET/CT:
Limited role in well-differentiated NETs (low metabolic activity), but valuable for poorly differentiated neuroendocrine carcinomas (NECs) with high proliferative indices. [35]
Indication: Grade 3 NETs and NECs (Ki-67 > 20%) Prognostic value: FDG avidity in well-differentiated NETs suggests aggressive biology
Endoscopic Evaluation
Gastroduodenoscopy: For suspected foregut (gastric, duodenal) primaries Colonoscopy: For suspected hindgut (colonic, rectal) primaries Video capsule endoscopy: May identify small bowel primaries missed on cross-sectional imaging Deep enteroscopy (balloon-assisted): Allows biopsy of small bowel lesions identified on imaging
Cardiac Assessment
Echocardiography - Transthoracic (TTE):
Screening investigation for all patients with carcinoid syndrome; repeat annually or with symptom change. [36]
Findings:
- Tricuspid valve: Thickened, retracted leaflets with restricted mobility; severe regurgitation in advanced disease
- Pulmonary valve: Thickening, stenosis, or mixed stenosis/regurgitation
- Right atrium/ventricle: Dilatation, RV dysfunction
- Severity grading: Doppler assessment of regurgitant jet, RV function
Sensitivity: 58-66% for detecting carcinoid heart disease
Transoesophageal Echocardiography (TOE):
Superior visualisation of valve morphology; used for:
- Detailed pre-operative assessment
- When TTE images inadequate
- Assessing suitability for valve repair vs replacement
Cardiac MRI:
Emerging role for:
- Detailed RV function and volume assessment
- Identification of myocardial fibrosis (late gadolinium enhancement)
- Pre-operative planning
Biomarkers:
- NT-proBNP: Elevated in cardiac involvement; useful for screening and monitoring
- Troponin: Usually normal unless acute decompensation
Histopathological Evaluation and WHO Classification
Tissue diagnosis essential for confirming NET diagnosis and determining grade, which guides prognosis and treatment. [37,38]
WHO 2019 Classification for GEP-NETs:
| Grade | Mitotic Count (per 2 mm²) | Ki-67 Index | Differentiation |
|---|---|---|---|
| G1 | less than 2 | less than 3% | Well-differentiated |
| G2 | 2-20 | 3-20% | Well-differentiated |
| G3 | > 20 | > 20% | Well-differentiated NET or poorly differentiated NEC |
Note: Grade 3 subdivided into:
- Well-differentiated NET G3: Ki-67 > 20%, but maintains differentiated morphology
- Poorly differentiated NEC: Ki-67 > 20%, loss of differentiation; further subdivided into small cell and large cell NECs
Immunohistochemistry:
- Chromogranin A: Positive in most well-differentiated NETs
- Synaptophysin: Neuroendocrine marker, often positive
- Ki-67: Proliferation marker, essential for grading
- Serotonin: Often positive in small bowel NETs causing carcinoid syndrome
- TTF-1: Positive in lung NETs
- CDX-2: Positive in GI NETs
Detailed Grading and Prognostic Implications:
Grade 1 NETs:
- Excellent prognosis: 5-year survival > 90% for localised disease
- Slow growth, minimal proliferative activity
- May be suitable for observation alone if small and asymptomatic
- High SSTR expression, excellent targets for SSA therapy and PRRT
Grade 2 NETs:
- Intermediate prognosis: 5-year survival 70-80% for localised disease
- More aggressive than G1 but still well-differentiated
- May require more aggressive treatment
- Usually maintain SSTR expression
Grade 3 NETs/NECs:
- Heterogeneous group with divergent outcomes:
- "Well-differentiated G3 NETs: Behave more like aggressive G2 tumours; may respond to PRRT; 5-year survival 30-50%"
- "Poorly differentiated NECs: Highly aggressive; managed with platinum-based chemotherapy (similar to small cell lung cancer); 5-year survival less than 10-20%"
- Ki-67 threshold of 55% may distinguish NET G3 (Ki-67 20-55%) from NEC (Ki-67 > 55%)
- FDG-PET usually positive in NECs, helping distinguish from well-differentiated G3 NETs
Site-Specific Variations:
- Lung carcinoids: Classified as typical (G1) or atypical (G2/3) based on mitotic rate and presence of necrosis
- Pancreatic NETs: More heterogeneous behaviour; functional vs non-functional
- Rectal NETs: Generally indolent; size (less than 1 cm, 1-2 cm, > 2 cm) main prognostic factor
7. Management
Management of NETs and carcinoid syndrome requires multidisciplinary approach involving oncology, surgery, endocrinology, cardiology, and radiology. Treatment strategy depends on tumour grade, stage, functional status, and patient factors.
General Principles
Multidisciplinary Team (MDT) Discussion: All NET cases should be discussed in specialist NET MDT, as management is complex and evolving. [39]
Treatment Goals:
- Control hormonal symptoms (in functional tumours)
- Reduce tumour burden
- Prevent disease progression
- Manage complications
- Optimise quality of life
Symptom Control - Somatostatin Analogues (SSAs)
Somatostatin analogues represent first-line therapy for carcinoid syndrome symptom control and have demonstrated anti-tumour effects. [40,41,42]
Mechanism of Action:
- Bind to somatostatin receptors (SSTR2, SSTR5) on NET cells
- Inhibit hormone secretion
- Reduce tumour cell proliferation
- Induce apoptosis
- Anti-angiogenic effects
First-Generation SSAs:
| Agent | Formulation | Dose | Administration |
|---|---|---|---|
| Octreotide LAR | Long-acting release | 20-30 mg IM every 4 weeks | Deep IM injection; initial short-acting octreotide bridge required (100-200 μg SC TDS for 2 weeks) |
| Lanreotide Autogel | Slow-release depot | 120 mg SC every 4 weeks | Deep SC injection; no bridge required |
| Octreotide SC | Short-acting | 100-200 μg SC TDS | For breakthrough symptoms or pre-operative |
Second-Generation SSA:
| Agent | Advantage | Dose |
|---|---|---|
| Pasireotide LAR | Broader SSTR binding (SSTR1,2,3,5) | 40-60 mg IM monthly |
Efficacy for Symptom Control:
- Diarrhoea control: 70-80% of patients
- Flushing control: 60-70% of patients
- May require dose escalation: Increase to 30-40 mg monthly or shorten interval to every 3 weeks
Anti-Tumour Effects - Key Trials:
PROMID Trial [40]:
- 85 patients with well-differentiated midgut NETs
- Octreotide LAR 30 mg vs placebo
- Median time to progression: 14.3 vs 6 months (HR 0.34, p=0.000072)
- Established SSAs as anti-proliferative agents
CLARINET Trial [41]:
- 204 patients with non-functioning GEP-NETs (pancreatic and intestinal)
- Lanreotide Autogel 120 mg vs placebo
- 24-month PFS: 65.1% vs 33% (HR 0.47, pless than 0.001)
- Confirmed anti-tumour effect across GEP-NETs
Side Effects:
- Gastrointestinal: Abdominal cramps, nausea, steatorrhoea (10-20%)
- Biliary: Gallstones (20-50%), usually asymptomatic; routine cholecystectomy not indicated
- Metabolic: Hyperglycaemia (worsening diabetes in 10%), hypothyroidism (rare)
- Injection site: Pain, nodules with long-acting formulations
Additional Symptom Control Agents
Telotristat Ethyl:
Oral tryptophan hydroxylase inhibitor, reducing peripheral serotonin synthesis. [43]
Indication: Add-on therapy for carcinoid diarrhoea inadequately controlled by SSAs Dose: 250 mg TDS Efficacy (TELESTAR trial): Reduced bowel movements by ≥30% in 44% vs 20% placebo Side Effects: Nausea, headache, elevated liver enzymes (monitor LFTs) Note: Must be taken with SSAs (not monotherapy)
Anti-Diarrhoeal Agents:
- Loperamide: 4-16 mg/day in divided doses
- Codeine phosphate: 30-60 mg QDS
- Cholestyramine: For bile acid malabsorption component
Anti-Histamines:
- H1-blockers (chlorphenamine): For pruritus, flushing in histamine-secreting tumours
- H2-blockers (ranitidine, famotidine): For gastric acid hypersecretion
Surgical Management
Curative-Intent Surgery:
Complete surgical resection offers only curative option for NETs. [44]
Indications:
- Localised disease without distant metastases
- Limited liver metastases amenable to complete resection
- Symptomatic primary tumour (obstruction, bleeding)
Approach by Site:
- Small bowel: Segmental resection with wide mesenteric lymphadenectomy
- Appendix: Appendectomy if less than 1 cm; right hemicolectomy if > 2 cm, caecal involvement, or lymphovascular invasion
- Rectum: Local excision if less than 1-2 cm; TME if > 2 cm or adverse features
- Pancreas: Enucleation for small, superficial lesions; formal resection for larger tumours
- Gastric: Endoscopic resection (type 1, less than 1 cm); surgical resection for type 2/3 or larger lesions
Cytoreductive Surgery:
Even with metastatic disease, debulking may improve symptom control and outcomes. [45]
Indications:
-
90% tumour reduction achievable (R90 resection)
- Symptomatic primary despite medical therapy
- Complications (obstruction, bleeding)
Outcomes: Improved symptom control in 80-95%; median symptom-free survival 4.5-5 years
Liver-Directed Therapies:
For liver-dominant metastatic disease not amenable to complete resection. [46]
| Technique | Mechanism | Indications | Outcomes |
|---|---|---|---|
| Hepatic resection | Complete removal of involved segments | Unilobar disease, less than 50% involvement | 5-year survival 60-80% |
| Radiofrequency ablation (RFA) | Thermal destruction | Small lesions (less than 3 cm), limited number | Local control 80-90% |
| Trans-arterial embolisation (TAE) | Ischaemic necrosis | Multiple bilobar metastases | Symptom control 80%, median duration 12 months |
| Trans-arterial chemoembolisation (TACE) | Embolisation + chemotherapy | Multiple bilobar metastases | Similar to TAE; no clear superiority |
| Selective internal radiotherapy (SIRT) | Yttrium-90 microspheres | Diffuse liver involvement | Symptom control 70-90%, PFS 13-22 months |
Complications: Post-embolisation syndrome (fever, pain, nausea) in 50-70%; carcinoid crisis risk (prevent with octreotide cover)
Peptide Receptor Radionuclide Therapy (PRRT)
Major advance in NET management, delivering targeted radiotherapy to SSTR-expressing tumours. [47]
177Lutetium-DOTATATE (Lutathera):
NETTER-1 Trial [47]:
- 229 patients with progressive midgut NETs
- 177Lu-DOTATATE (7.4 GBq every 8 weeks × 4) + octreotide LAR vs octreotide LAR 60 mg
- Efficacy:
- "Median PFS: 20 months (PRRT) vs 8.4 months (control) - HR 0.21, pless than 0.001"
- "Overall response rate: 18% vs 3%"
- 79% reduction in progression or death risk
- Trend towards OS benefit (median not reached vs 36.3 months)
Eligibility Criteria:
- Well-differentiated NET (G1 or G2, selected G3)
- SSTR-positive on 68Ga-DOTATATE PET (Krenning score ≥2)
- Progressive disease or high tumour burden
- Adequate organ function (creatinine less than 150 μmol/L, platelets > 75×10⁹/L, Hb > 8 g/dL)
Administration:
- 4 cycles of 7.4 GBq 177Lu-DOTATATE every 8 weeks
- Continue SSAs for symptom control
- Anti-emetics for nausea
- Amino acid infusion (lysine/arginine) to protect kidneys
Side Effects:
- Haematological: Cytopenias (usually grade 1-2); myelodysplastic syndrome/acute leukaemia rare (2-3%)
- Renal: Reduced GFR in 10-20%; monitor renal function
- Hepatic: Transient transaminitis
- Hormonal crisis: Rare; prevented by adequate SSA coverage
- Nausea/vomiting: Common during infusion
Outcomes:
- Disease control rate (CR+PR+SD): 83%
- Symptom improvement: 60-70%
- Quality of life improvement demonstrated
Systemic Therapy
Targeted Molecular Therapies:
Everolimus (mTOR inhibitor):
RADIANT-2 Trial (carcinoid) [48]:
- 429 patients with advanced NETs and carcinoid syndrome
- Everolimus 10 mg daily + octreotide LAR vs placebo + octreotide LAR
- Median PFS: 16.4 vs 11.3 months (HR 0.77, p=0.026 in central review; p=0.036 in investigator assessment)
RADIANT-3 Trial (pancreatic NETs) [49]:
- Median PFS: 11.0 vs 4.6 months (HR 0.35, pless than 0.001)
Indications: Progressive well-differentiated NETs (pancreatic, GI, lung) Dose: 10 mg oral daily Side effects: Stomatitis (60%), infections, hyperglycaemia, pneumonitis (10%), rash
Sunitinib (multi-targeted tyrosine kinase inhibitor):
Indication: Progressive pancreatic NETs (licensed for pancreatic only) Dose: 37.5 mg oral daily continuous Efficacy: Median PFS 11.4 vs 5.5 months (HR 0.42, pless than 0.001) Side effects: Fatigue, hypertension, hand-foot syndrome, diarrhoea, cytopenias
Chemotherapy:
Indications: Poorly differentiated NECs (Ki-67 > 20%, particularly > 55%), rapidly progressive disease
Regimens:
- Platinum-based (for NEC): Cisplatin/carboplatin + etoposide (response rate 40-60% in NECs)
- Temozolomide-based (for pancreatic NETs): Temozolomide + capecitabine (response rate 30-40%)
- Streptozocin-based (historical, mainly pancreatic): Streptozocin + 5-FU
Poor efficacy in well-differentiated NETs; reserve for high-grade tumours
Management of Carcinoid Heart Disease
Medical Optimisation:
- Aggressive SSA therapy: High-dose/increased frequency to minimise mediator exposure
- Diuretics: Furosemide for fluid overload
- Telotristat: May reduce further valvular damage
- Avoid: Serotonin antagonists (e.g., cyproheptadine) - limited efficacy
Surgical Intervention:
Indications for Valve Surgery: [50]
- Severe symptomatic valvular disease (NYHA III-IV)
- Progressive RV dysfunction
- Refractory right heart failure
Timing Considerations:
- Ideally after tumour control achieved with medical/surgical therapy
- Pre-operative optimisation with SSAs essential
- High perioperative morbidity/mortality (15-20%) - requires specialist centre
Procedure:
- Tricuspid valve: Replacement usually required (repair rarely feasible due to leaflet retraction)
- Bioprosthetic preferred over mechanical (avoid anticoagulation)
- Pulmonary valve: Replacement if severe stenosis
- Concomitant left-sided valve surgery if involved (rare)
Perioperative Management:
- Continuous octreotide infusion (50-100 μg/h) from 12h pre-op
- Invasive haemodynamic monitoring
- Avoid catecholamines (may precipitate crisis); use vasopressin if vasopressor needed
- Post-operative intensive monitoring (48-72h)
Outcomes:
- Operative mortality: 10-20% (high-risk surgery)
- Symptomatic improvement: 80% of survivors
- 5-year survival: 50-60%
Perioperative Management and Carcinoid Crisis Prevention
Pre-operative Assessment:
- Biochemical control: 5-HIAA ideally normalised or significantly reduced
- Cardiac evaluation: Echo to assess RV function, valvular disease
- Volumetric assessment: CT/MRI to assess tumour burden
Prophylaxis Protocol: [28]
Low-Risk Procedures (diagnostic endoscopy, minor surgery):
- Octreotide 100-200 μg SC 2-4h before procedure
- Additional boluses available for breakthrough symptoms
High-Risk Procedures (tumour resection, liver embolisation, major surgery):
- Octreotide 100-500 μg IV bolus at induction
- Followed by continuous infusion 50-100 μg/h throughout surgery and first 24-48h post-op
- Gradually wean over 48-72h based on clinical response
Intraoperative Management:
- Avoid histamine-releasing agents (morphine, atracurium, tubocurarine)
- Use fentanyl or remifentanil for analgesia
- Gentle handling of tumour to minimise mediator release
- Invasive haemodynamic monitoring
- If hypotension: fluids, octreotide bolus, vasopressin (NOT catecholamines)
Post-operative Care:
- ICU/HDU monitoring
- Continue octreotide infusion
- Monitor for complications: carcinoid crisis, heart failure, bleeding
8. Complications
Tumour-Related Complications
| Complication | Frequency | Mechanism | Management |
|---|---|---|---|
| Intestinal obstruction | 20-30% | Mesenteric fibrosis, tumour mass effect, kinking | Bowel rest, NGT decompression; surgery if persistent |
| Mesenteric ischaemia | 10-15% | Tumour-associated fibrosis of mesenteric vessels | Angiography ± revascularisation; surgical resection of ischaemic bowel |
| GI bleeding | 10% | Ulceration at tumour site, varices from portal involvement | Endoscopic therapy; embolisation; surgery |
| Pellagra | 5% | Tryptophan depletion from excessive serotonin synthesis | Niacin supplementation (100-500 mg daily) |
Treatment-Related Complications
SSA Therapy:
- Cholelithiasis: 20-50%; usually asymptomatic, monitor with annual ultrasound
- Steatorrhoea: 10-20% from pancreatic enzyme suppression; may require pancreatic enzyme replacement
- Hyperglycaemia: Monitor glucose in diabetics; may require insulin adjustment
PRRT:
- Myelodysplastic syndrome/leukaemia: 2-3%; occurs years after treatment
- Renal insufficiency: 10-20%; avoid in pre-existing severe renal impairment
- Hormonal crisis: Rare with adequate SSA cover
Liver-Directed Therapies:
- Post-embolisation syndrome: 50-70% (fever, pain, nausea); self-limiting, 3-5 days
- Hepatic abscess: 1-2%; requires antibiotics ± drainage
- Hepatic failure: Rare if less than 50% liver embolised per session
Cardiac Complications
- Progressive right heart failure: Requires diuretics, consider surgery
- Atrial fibrillation: Common with RA dilatation; rate control, anticoagulation
- Sudden cardiac death: Rare; arrhythmic or haemodynamic
9. Prognosis
Prognosis in NETs varies dramatically based on grade, stage, primary site, and functional status. [8,51]
Survival by Stage
| Stage | 5-Year Survival |
|---|---|
| Localised | 89-97% |
| Regional | 75-83% |
| Distant metastases | 25-42% |
Prognostic Factors
Favourable:
- Low grade (G1 > G2 > G3)
- Localised disease
- Complete surgical resection
- Small bowel or rectal primary
- Low Ki-67 index (less than 5%)
- Young age
- Absence of carcinoid heart disease
Unfavourable:
- High grade (G3, particularly poorly differentiated NEC)
- Liver metastases with high tumour burden
- Pancreatic primary (more aggressive than small bowel)
- Ki-67 > 20%
- Carcinoid heart disease (reduces median survival by 50%)
- Elevated markers (5-HIAA > 300 μmol/24h, CgA markedly elevated)
Site-Specific Prognosis
- Small bowel: Best prognosis among metastatic GEP-NETs; median OS 10-12 years for G1/G2
- Pancreatic: More aggressive; median OS 5-7 years for metastatic disease
- Rectal: Excellent prognosis if less than 2 cm; > 95% 5-year survival
- Lung carcinoid: Typical carcinoid 90% 5-year survival; atypical 50-70%
Impact of Treatment
Modern multimodal therapy has significantly improved outcomes:
- PRRT: Added median PFS of 11.6 months in NETTER-1
- SSAs: Extended median PFS by 8.3 months (PROMID) and significant delay in CLARINET
- Targeted therapy: PFS benefit of 6-8 months with everolimus/sunitinib
- Surgical cytoreduction: Symptom-free survival 4.5-5 years
Many patients live > 10 years even with metastatic disease, particularly G1/G2 small bowel primaries managed in specialist centres.
10. Follow-Up and Surveillance
Monitoring Strategy
Functional Tumours:
- Clinical assessment: Every 3-4 months initially, then 6-monthly when stable
- Biochemistry: CgA, 5-HIAA every 3-6 months
- Imaging: CT/MRI abdomen/pelvis every 6-12 months
- 68Ga-DOTATATE PET: Every 12-24 months or when change in management considered
- Echocardiography: Annually for carcinoid syndrome patients; more frequent if cardiac involvement
Non-Functional Tumours:
- Clinical + biochemistry: Every 6 months
- Imaging: Every 6-12 months depending on grade and stability
- PET: As needed for management decisions
Post-Resection:
- More intensive surveillance first 2 years (recurrence risk highest)
- Lifelong follow-up required (late recurrences occur)
11. Prevention and Screening
No population-based screening exists for sporadic NETs.
Surveillance in Genetic Syndromes:
MEN-1:
- Annual biochemistry (fasting gut hormones, chromogranin A) from age 5-10 years
- MRI abdomen from age 10-15 years every 1-2 years
- High risk (50-80%) of pancreatic NETs by age 70
Von Hippel-Lindau (VHL):
- Increased risk of pancreatic NETs
- Annual MRI abdomen from age 15-16 years
Neurofibromatosis Type 1 (NF1):
- Increased risk of duodenal/peri-ampullary NETs (somatostatinomas)
- Consider screening endoscopy if symptomatic
12. Key Guidelines
European Neuroendocrine Tumour Society (ENETS) Guidelines (Multiple publications 2016-2020):
- Comprehensive guidelines covering diagnosis, staging, and treatment of GEP-NETs
- Site-specific recommendations for gastric, duodenal, pancreatic, small bowel, colonic NETs
- Biomarker and imaging recommendations
North American Neuroendocrine Tumor Society (NANETS) Guidelines (2010-2017):
- Consensus guidelines on diagnosis and management
- Specific protocols for functional syndrome management
National Comprehensive Cancer Network (NCCN) Guidelines (Updated annually):
- Evidence-based treatment algorithms
- Systemic therapy selection based on grade and site
UK/Ireland Neuroendocrine Tumour Society (UKI-NETS) Guidelines (2020):
- NHS-context specific recommendations
- Patient pathway guidance
Examination Focus
Common Exam Questions
-
"A 58-year-old presents with flushing and diarrhoea. How would you investigate?"
- Focused history: timing, triggers, associated symptoms
- Exclude common causes (menopause, medications)
- Biochemistry: 24h urinary 5-HIAA (avoid interfering foods), chromogranin A
- Imaging: CT chest/abdomen/pelvis (multiphasic), 68Ga-DOTATATE PET
- Cardiac: echocardiography
- Tissue: biopsy for histology and grading
-
"What is the WHO 2019 grading system for GEP-NETs?"
- Grade 1: Ki-67 less than 3%, mitoses less than 2 per 2mm²
- Grade 2: Ki-67 3-20%, mitoses 2-20 per 2mm²
- Grade 3: Ki-67 > 20%, mitoses > 20 per 2mm²
- Subdivided into well-differentiated NET G3 vs poorly differentiated NEC
-
"What is the evidence for somatostatin analogues in NET treatment?"
- Symptom control: 60-80% response rate for flushing/diarrhoea
- Anti-tumour effects:
- PROMID: Octreotide LAR extended TTP from 6 to 14.3 months in midgut NETs
- CLARINET: Lanreotide improved 24-month PFS from 33% to 65% in GEP-NETs
- Generally well-tolerated; gallstones most common side effect
-
"How would you manage a patient undergoing surgery for carcinoid?"
- Pre-operative assessment: Biochemical control, cardiac evaluation, imaging
- Prophylaxis:
- Octreotide 100-500 μg IV at induction
- Continuous infusion 50-100 μg/h throughout surgery and 24-48h post-op
- Intraoperative: Avoid histamine-releasing agents, gentle tumour handling, invasive monitoring
- Complications: Carcinoid crisis (flushing, hypotension, bronchospasm) - treat with octreotide bolus, fluids, avoid catecholamines
-
"What is peptide receptor radionuclide therapy (PRRT)?"
- Targeted radiotherapy using radiolabelled somatostatin analogues
- 177Lu-DOTATATE binds to SSTR-expressing tumours, delivering beta radiation
- NETTER-1 trial: Median PFS 20 vs 8.4 months compared to octreotide alone in progressive midgut NETs
- Requires SSTR-positive disease on 68Ga-DOTATATE PET (Krenning ≥2)
- 4 cycles of 7.4 GBq every 8 weeks
- Side effects: Cytopenias, renal impairment, rare MDS/leukaemia
Viva Points
Opening Statement: "Neuroendocrine tumours are neoplasms arising from neuroendocrine cells, most commonly in the gastrointestinal tract and lungs. Carcinoid syndrome occurs in 10-20% of patients, typically when liver metastases allow bioactive substances—particularly serotonin—to bypass hepatic first-pass metabolism. The classic tetrad comprises flushing, secretory diarrhoea, bronchospasm, and right-sided valvular heart disease."
Epidemiology to quote:
- Incidence 6.98 per 100,000 and rising
- Small bowel most common primary for carcinoid syndrome
- Only 10-20% of NET patients develop syndrome (requires hepatic metastases or extra-portal drainage)
Classification system: "The WHO 2019 classification grades GEP-NETs based on Ki-67 proliferation index: Grade 1 less than 3%, Grade 2 3-20%, Grade 3 > 20%. Grade 3 is further subdivided into well-differentiated NET G3 and poorly differentiated neuroendocrine carcinoma, with markedly different prognoses and treatment strategies."
Diagnosis:
- Biochemical: 24-hour urinary 5-HIAA (sensitivity 73%, specificity 100%), chromogranin A
- Imaging: 68Ga-DOTATATE PET most sensitive (93-96%), CT/MRI for anatomical detail
- Cardiac: Echocardiography to detect carcinoid heart disease (20-50% of syndrome patients)
- Tissue: Biopsy for histological confirmation and Ki-67 grading
First-line treatment with evidence:
- Somatostatin analogues (octreotide LAR 20-30 mg IM monthly, lanreotide 120 mg SC monthly)
- "PROMID trial: Extended TTP from 6 to 14.3 months in midgut NETs"
- "CLARINET trial: Improved PFS in GEP-NETs (HR 0.47)"
- Surgery for localised disease or cytoreduction if > 90% resection achievable
- PRRT for progressive disease: NETTER-1 showed median PFS 20 vs 8.4 months
Carcinoid crisis: "A life-threatening complication characterised by severe flushing, profound hypotension, bronchospasm, and altered consciousness, triggered by anaesthesia, surgery, or tumour manipulation. Prevention requires octreotide 100-500 μg IV bolus and continuous infusion perioperatively. Treatment includes further octreotide boluses, fluids, and vasopressin if needed—avoid catecholamines as they may worsen mediator release."
Carcinoid heart disease mechanism: "Results from chronic serotonin exposure causing fibroblast proliferation and fibrous plaque deposition on right-sided valves, predominantly affecting tricuspid (regurgitation > 90%) and pulmonary valves (stenosis 50-80%). Left heart typically spared due to pulmonary inactivation of serotonin. Requires valve replacement in severe symptomatic cases, though operative mortality is high (10-20%)."
Common Mistakes
❌ Failing to consider carcinoid syndrome in differential for flushing/diarrhoea:
- Many candidates focus only on common causes (IBS, menopause) and miss this important diagnosis
❌ Not recognising requirement for hepatic metastases for syndrome:
- Primary small bowel NET without liver involvement rarely causes syndrome due to hepatic first-pass metabolism
❌ Ordering 5-HIAA without dietary restriction:
- Serotonin-rich foods (bananas, pineapples, walnuts) cause false positives - must avoid 48-72h before collection
❌ Not recognising SSAs have anti-tumour effects:
- Previously thought purely symptomatic; PROMID/CLARINET established as antiproliferative therapy
❌ Inadequate perioperative octreotide in known carcinoid:
- Single pre-operative dose insufficient; requires continuous infusion for high-risk procedures
❌ Using catecholamines for hypotension in carcinoid crisis:
- May paradoxically worsen hypotension and trigger further mediator release; vasopressin preferred
❌ Not screening for cardiac involvement:
- Annual echocardiography essential in carcinoid syndrome; cardiac disease major determinant of mortality
❌ Confusing NET grading with staging:
- WHO grade (Ki-67 based) is distinct from TNM stage; both independently prognostic
Model Answers
Q: "How would you investigate a patient with suspected carcinoid syndrome?"
A: "I would approach this systematically with biochemical confirmation, anatomical localisation, and assessment of complications.
Biochemistry: The gold standard is 24-hour urinary 5-HIAA, with sensitivity 73% and specificity 100% when significantly elevated. Importantly, the patient must avoid serotonin-rich foods—including bananas, pineapple, kiwi, walnuts, and avocados—for 48-72 hours before collection, and discontinue interfering medications like paracetamol. I would also measure chromogranin A, though this is non-specific and may be falsely elevated by proton pump inhibitors, which should ideally be stopped 2 weeks prior.
Imaging: For anatomical localisation, I would arrange multiphasic CT of chest, abdomen, and pelvis, as NETs are hypervascular and best seen in arterial phase. However, the most sensitive imaging modality is 68Ga-DOTATATE PET/CT, which detects SSTR-expressing disease with 93-96% sensitivity and is essential for selecting patients for PRRT. It frequently detects disease missed on conventional imaging.
Cardiac assessment: Given that 20-50% of carcinoid syndrome patients develop cardiac involvement, I would arrange transthoracic echocardiography to look for characteristic right-sided valvular disease—tricuspid regurgitation and pulmonary stenosis—and may measure NT-proBNP as a screening biomarker.
Histology: Tissue diagnosis is essential to confirm NET, determine grade (Ki-67 index), and guide treatment. If the primary site is unknown after imaging, endoscopy or biopsy of accessible metastases would be appropriate.
This comprehensive assessment allows confirmation of diagnosis, identification of primary site, staging, grading, and detection of complications, which together inform the treatment strategy to be discussed in a multidisciplinary NET meeting."
Q: "Describe the mechanism of action and evidence for PRRT in neuroendocrine tumours."
A: "Peptide receptor radionuclide therapy, or PRRT, is a form of targeted molecular radiotherapy that exploits the high expression of somatostatin receptors on neuroendocrine tumour cells.
Mechanism: The therapy uses 177Lutetium-DOTATATE, which is a radiolabelled somatostatin analogue. This compound binds to somatostatin receptors—particularly SSTR2—on tumour cell surfaces. After binding, it is internalized into the cell, where the beta-radiation from lutetium-177 causes direct DNA damage and cell death. Because SSTR expression is high on NET cells but low on most normal tissues, this achieves selective tumour targeting while minimizing systemic toxicity.
Patient Selection: Eligibility requires demonstration of SSTR-positive disease on 68Ga-DOTATATE PET scan, typically with a Krenning score of at least 2, meaning tumour uptake should be at least equal to liver. The treatment is indicated for well-differentiated, progressive NETs with adequate organ function.
Evidence: The landmark NETTER-1 trial, published in the New England Journal of Medicine in 2017, randomized 229 patients with progressive midgut NETs to either four cycles of 177Lu-DOTATATE every 8 weeks plus octreotide LAR, versus high-dose octreotide LAR 60 mg alone. The results showed a dramatic improvement in median progression-free survival—20 months in the PRRT group versus 8.4 months in the control group, with a hazard ratio of 0.21 representing a 79% reduction in risk of progression or death. Objective response rate was 18% versus 3%, and there was a trend toward overall survival benefit.
Administration and Toxicity: Patients receive 7.4 GBq per cycle over four cycles given every 8 weeks, with amino acid infusion to protect the kidneys. The main side effects are haematological cytopenias—usually grade 1-2—and gradual decline in renal function in 10-20% of patients. There is a small long-term risk of myelodysplastic syndrome or acute leukaemia in approximately 2-3% of patients, occurring several years after treatment.
This represents a major advance in NET management and has become standard of care for progressive, SSTR-positive disease."
References
-
Modlin IM, Oberg K, Chung DC, et al. Gastroenteropancreatic neuroendocrine tumours. Lancet Oncol. 2008;9(1):61-72. doi:10.1016/S1470-2045(07)70410-2
-
Halperin DM, Shen C, Dasari A, et al. Frequency of carcinoid syndrome at neuroendocrine tumour diagnosis: a population-based study. Lancet Oncol. 2017;18(4):525-534. doi:10.1016/S1470-2045(17)30110-9
-
Vinik AI, Woltering EA, Warner RR, et al. NANETS consensus guidelines for the diagnosis of neuroendocrine tumor. Pancreas. 2010;39(6):713-734. doi:10.1097/MPA.0b013e3181ebaffd
-
Dasari A, Shen C, Halperin D, et al. Trends in the incidence, prevalence, and survival outcomes in patients with neuroendocrine tumors in the United States. JAMA Oncol. 2017;3(10):1335-1342. doi:10.1001/jamaoncol.2017.0589
-
Yao JC, Hassan M, Phan A, et al. One hundred years after "carcinoid": epidemiology of and prognostic factors for neuroendocrine tumors in 35,825 cases in the United States. J Clin Oncol. 2008;26(18):3063-3072. doi:10.1200/JCO.2007.15.4377
-
Lawrence B, Gustafsson BI, Chan A, et al. The epidemiology of gastroenteropancreatic neuroendocrine tumors. Endocrinol Metab Clin North Am. 2011;40(1):1-18. doi:10.1016/j.ecl.2010.12.005
-
Halperin DM, Kulke MH, Yao JC. A tale of two tumors: treating pancreatic and extrapancreatic neuroendocrine tumors. Annu Rev Med. 2015;66:1-16. doi:10.1146/annurev-med-081813-023925
-
Modlin IM, Lye KD, Kidd M. A 5-decade analysis of 13,715 carcinoid tumors. Cancer. 2003;97(4):934-959. doi:10.1002/cncr.11105
-
Thakker RV, Newey PJ, Walls GV, et al. Clinical practice guidelines for multiple endocrine neoplasia type 1 (MEN1). J Clin Endocrinol Metab. 2012;97(9):2990-3011. doi:10.1210/jc.2012-1230
-
Delle Fave G, O'Toole D, Sundin A, et al. ENETS consensus guidelines update for gastroduodenal neuroendocrine neoplasms. Neuroendocrinology. 2016;103(2):119-124. doi:10.1159/000443168
-
Hemminki K, Li X. Incidence trends and risk factors of carcinoid tumors: a nationwide epidemiologic study from Sweden. Cancer. 2001;92(8):2204-2210. doi:10.1002/1097-0142(20011015)92:8less than 2204::aid-cncr1564> 3.0.co;2-r
-
Modlin IM, Kidd M, Latich I, et al. Current status of gastrointestinal carcinoids. Gastroenterology. 2005;128(6):1717-1751. doi:10.1053/j.gastro.2005.03.038
-
Niederle MB, Hackl M, Kaserer K, Niederle B. Gastroenteropancreatic neuroendocrine tumours: the current incidence and staging based on the WHO and European Neuroendocrine Tumour Society classification. Endocr Relat Cancer. 2010;17(4):909-918. doi:10.1677/ERC-10-0152
-
Kytola S, Hoog A, Nord B, et al. Comparative genomic hybridization identifies loss of 18q22-qter as an early and specific event in tumorigenesis of midgut carcinoids. Am J Pathol. 2001;158(5):1803-1808. doi:10.1016/S0002-9440(10)64136-3
-
Jiao Y, Shi C, Edil BH, et al. DAXX/ATRX, MEN1, and mTOR pathway genes are frequently altered in pancreatic neuroendocrine tumors. Science. 2011;331(6021):1199-1203. doi:10.1126/science.1200609
-
Marinoni I, Kurrer AS, Vassella E, et al. Loss of DAXX and ATRX are associated with chromosome instability and reduced survival of patients with pancreatic neuroendocrine tumors. Gastroenterology. 2014;146(2):453-460. doi:10.1053/j.gastro.2013.10.020
-
Tellez MR, Mamikunian G, O'Dorisio TM, et al. A single fasting plasma 5-HIAA value correlates with 24-hour urinary 5-HIAA values and other biomarkers in midgut neuroendocrine tumors (NETs). Pancreas. 2013;42(3):405-410. doi:10.1097/MPA.0b013e318271c0d5
-
Zandee WT, Kamp K, van Adrichem RC, et al. Effect of hormone secretory syndromes on neuroendocrine tumor prognosis. Endocr Relat Cancer. 2017;24(7):R261-R274. doi:10.1530/ERC-16-0538
-
Bhattacharyya S, Davar J, Dreyfus G, Caplin ME. Carcinoid heart disease. Circulation. 2007;116(24):2860-2865. doi:10.1161/CIRCULATIONAHA.107.691147
-
Møller JE, Pellikka PA, Bernheim AM, et al. Prognosis of carcinoid heart disease: analysis of 200 cases over two decades. Circulation. 2005;112(21):3320-3327. doi:10.1161/CIRCULATIONAHA.105.553750
-
Dobson R, Burgess MI, Pritchard DM, Cuthbertson DJ. The clinical presentation and management of carcinoid heart disease. Int J Cardiol. 2014;173(1):29-32. doi:10.1016/j.ijcard.2014.02.037
-
Ruszniewski P, Delle Fave G, Cadiot G, et al. Well-differentiated gastric tumors/carcinomas. Neuroendocrinology. 2006;84(3):158-164. doi:10.1159/000098009
-
Warner RRP, Mani S, Profeta J, Grunstein E. Serotonin carcinoid and bronchial carcinoid tumor: A clinical review. Ann Clin Lab Sci. 1996;26(6):465-489. PMID: 8908495
-
Pape UF, Böhmig M, Berndt U, et al. Survival and clinical outcome of patients with neuroendocrine tumors of the gastroenteropancreatic tract in a German referral center. Ann N Y Acad Sci. 2004;1014:222-233. doi:10.1196/annals.1294.025
-
Concors SJ, Sarpel U. A contemporary review of small bowel neuroendocrine tumors. Int J Surg. 2016;28:146-153. doi:10.1016/j.ijsu.2016.02.092
-
Druce MR, Bharwani N, Akker SA, et al. Intra-abdominal fibrosis in a recent cohort of patients with neuroendocrine ('carcinoid') tumours of the small bowel. QJM. 2010;103(3):177-185. doi:10.1093/qjmed/hcp180
-
Shah GM, Shah RG, Veillette H, et al. Biochemical assessment of niacin deficiency among carcinoid cancer patients. Am J Gastroenterol. 2005;100(10):2307-2314. doi:10.1111/j.1572-0241.2005.00215.x
-
Condron ME, Pommier SJ, Pommier RF. Continuous infusion of octreotide combined with perioperative octreotide bolus does not prevent intraoperative carcinoid crisis. Surgery. 2016;159(1):358-365. doi:10.1016/j.surg.2015.05.036
-
Tormey WP, FitzGerald RJ. The clinical and laboratory correlates of an increased urinary 5-hydroxyindoleacetic acid. Postgrad Med J. 1995;71(839):542-545. doi:10.1136/pgmj.71.839.542
-
Marotta V, Zatelli MC, Sciammarella C, et al. Chromogranin A as circulating marker for diagnosis and management of neuroendocrine neoplasms: more flaws than fame. Endocr Relat Cancer. 2018;25(1):R11-R29. doi:10.1530/ERC-17-0269
-
Sundin A, Vullierme MP, Kaltsas G, Plockinger U. ENETS consensus guidelines for the standards of care in neuroendocrine tumors: radiological examinations. Neuroendocrinology. 2009;90(2):167-183. doi:10.1159/000184855
-
Dromain C, de Baere T, Lumbroso J, et al. Detection of liver metastases from endocrine tumors: a prospective comparison of somatostatin receptor scintigraphy, computed tomography, and magnetic resonance imaging. J Clin Oncol. 2005;23(1):70-78. doi:10.1200/JCO.2005.01.013
-
Deppen SA, Liu E, Blume JD, et al. Safety and efficacy of 68Ga-DOTATATE PET/CT for diagnosis, staging, and treatment management of neuroendocrine tumors. J Nucl Med. 2016;57(5):708-714. doi:10.2967/jnumed.115.163865
-
Hofman MS, Lau WF, Hicks RJ. Somatostatin receptor imaging with 68Ga DOTATATE PET/CT: clinical utility, normal patterns, pearls, and pitfalls in interpretation. Radiographics. 2015;35(2):500-516. doi:10.1148/rg.352140164
-
Binderup T, Knigge U, Loft A, et al. 18F-fluorodeoxyglucose positron emission tomography predicts survival of patients with neuroendocrine tumors. Clin Cancer Res. 2010;16(3):978-985. doi:10.1158/1078-0432.CCR-09-1759
-
Bhattacharyya S, Toumpanakis C, Caplin ME, Davar J. Analysis of 150 patients with carcinoid syndrome seen in a single year at one institution in the first decade of the twenty-first century. Am J Cardiol. 2008;101(3):378-381. doi:10.1016/j.amjcard.2007.08.045
-
Nagtegaal ID, Odze RD, Klimstra D, et al. The 2019 WHO classification of tumours of the digestive system. Histopathology. 2020;76(2):182-188. doi:10.1111/his.13975
-
Rindi G, Klimstra DS, Abedi-Ardekani B, et al. A common classification framework for neuroendocrine neoplasms: an International Agency for Research on Cancer (IARC) and World Health Organization (WHO) expert consensus proposal. Mod Pathol. 2018;31(12):1770-1786. doi:10.1038/s41379-018-0110-y
-
Singh S, Asa SL, Dey C, et al. Diagnosis and management of gastrointestinal neuroendocrine tumors: An evidence-based Canadian consensus. Cancer Treat Rev. 2016;47:32-45. doi:10.1016/j.ctrv.2016.05.003
-
Rinke A, Müller HH, Schade-Brittinger C, et al. Placebo-controlled, double-blind, prospective, randomized study on the effect of octreotide LAR in the control of tumor growth in patients with metastatic neuroendocrine midgut tumors: a report from the PROMID Study Group. J Clin Oncol. 2009;27(28):4656-4663. doi:10.1200/JCO.2009.22.8510
-
Caplin ME, Pavel M, Ćwikła JB, et al. Lanreotide in metastatic enteropancreatic neuroendocrine tumors. N Engl J Med. 2014;371(3):224-233. doi:10.1056/NEJMoa1316158
-
Hofland J, Kaltsas G, de Herder WW. Advances in the diagnosis and management of well-differentiated neuroendocrine neoplasms. Endocr Rev. 2020;41(2):371-403. doi:10.1210/endrev/bnz004
-
Kulke MH, Hörsch D, Caplin ME, et al. Telotristat ethyl, a tryptophan hydroxylase inhibitor for the treatment of carcinoid syndrome. J Clin Oncol. 2017;35(1):14-23. doi:10.1200/JCO.2016.69.2780
-
Mayo SC, de Jong MC, Pulitano C, et al. Surgical management of hepatic neuroendocrine tumor metastasis: results from an international multi-institutional analysis. Ann Surg Oncol. 2010;17(12):3129-3136. doi:10.1245/s10434-010-1154-5
-
Chambers AJ, Pasieka JL, Dixon E, Rorstad O. The palliative benefit of aggressive surgical intervention for both hepatic and mesenteric metastases from neuroendocrine tumors. Surgery. 2008;144(4):645-651. doi:10.1016/j.surg.2008.06.008
-
Kennedy AS, Dezarn WA, McNeillie P, et al. Radioembolization for unresectable neuroendocrine hepatic metastases using resin 90Y-microspheres: early results in 148 patients. Am J Clin Oncol. 2008;31(3):271-279. doi:10.1097/COC.0b013e31815e4557
-
Strosberg J, El-Haddad G, Wolin E, et al. Phase 3 trial of 177Lu-Dotatate for midgut neuroendocrine tumors. N Engl J Med. 2017;376(2):125-135. doi:10.1056/NEJMoa1607427
-
Pavel ME, Hainsworth JD, Baudin E, et al. Everolimus plus octreotide long-acting repeatable for the treatment of advanced neuroendocrine tumours associated with carcinoid syndrome (RADIANT-2): a randomised, placebo-controlled, phase 3 study. Lancet. 2011;378(9808):2005-2012. doi:10.1016/S0140-6736(11)61742-X
-
Yao JC, Shah MH, Ito T, et al. Everolimus for advanced pancreatic neuroendocrine tumors. N Engl J Med. 2011;364(6):514-523. doi:10.1056/NEJMoa1009290
-
Bhattacharyya S, Jagtap V, Baliga V, et al. Outcomes, risks and complications of cardiac surgery for carcinoid heart disease. Eur J Cardiothorac Surg. 2011;40(1):168-172. doi:10.1016/j.ejcts.2010.10.035
-
Garcia-Carbonero R, Capdevila J, Crespo-Herrero G, et al. Incidence, patterns of care and prognostic factors for outcome of gastroenteropancreatic neuroendocrine tumors (GEP-NETs): results from the National Cancer Registry of Spain (RGETNE). Ann Oncol. 2010;21(9):1794-1803. doi:10.1093/annonc/mdq022
Last Updated: 2026-01-05
Learning map
Use these linked topics to study the concept in sequence and compare related presentations.
Prerequisites
Start here if you need the foundation before this topic.
- Neuroendocrine System Physiology
Differentials
Competing diagnoses and look-alikes to compare.
- VIPoma
- Phaeochromocytoma
- Medullary Thyroid Carcinoma
Consequences
Complications and downstream problems to keep in mind.
- Carcinoid Heart Disease
- Carcinoid Crisis