Cardiac Tamponade in Adults
Cardiac tamponade is a life-threatening cardiovascular emergency characterized by accumulation of fluid within the pericardial space, resulting in elevated intrapericardial pressure and impaired ventricular filling....
What matters first
Cardiac tamponade is a life-threatening cardiovascular emergency characterized by accumulation of fluid within the pericardial space, resulting in elevated intrapericardial pressure and impaired ventricular filling....
Beck's triad (hypotension, JVP elevation, muffled heart sounds)
9 Jan 2026
Generated educational material; verify before clinical use.
Visible references section
See the concept before reading it
Study the key anatomy, imaging, and decision pathways as full teaching plates.
Clinical board
A visual summary of the highest-yield teaching signals on this page.
Urgent signals
Safety-critical features pulled from the topic metadata.
- Beck's triad (hypotension, JVP elevation, muffled heart sounds)
- Pulsus paradoxus greater than 10 mmHg
- Electrical alternans on ECG
- PEA arrest
Content status and exam context
This page is AI-generated educational content. It may contain errors or omissions and is not a substitute for current guidelines, local protocols, senior clinical judgement, or professional medical advice.
MedVellum does not claim an individual clinician reviewer, board certification, or professional credential for this page unless a future version names a real, verifiable reviewer.
Clinical explanation and evidence
Cardiac Tamponade in Adults
Clinical Overview
Summary
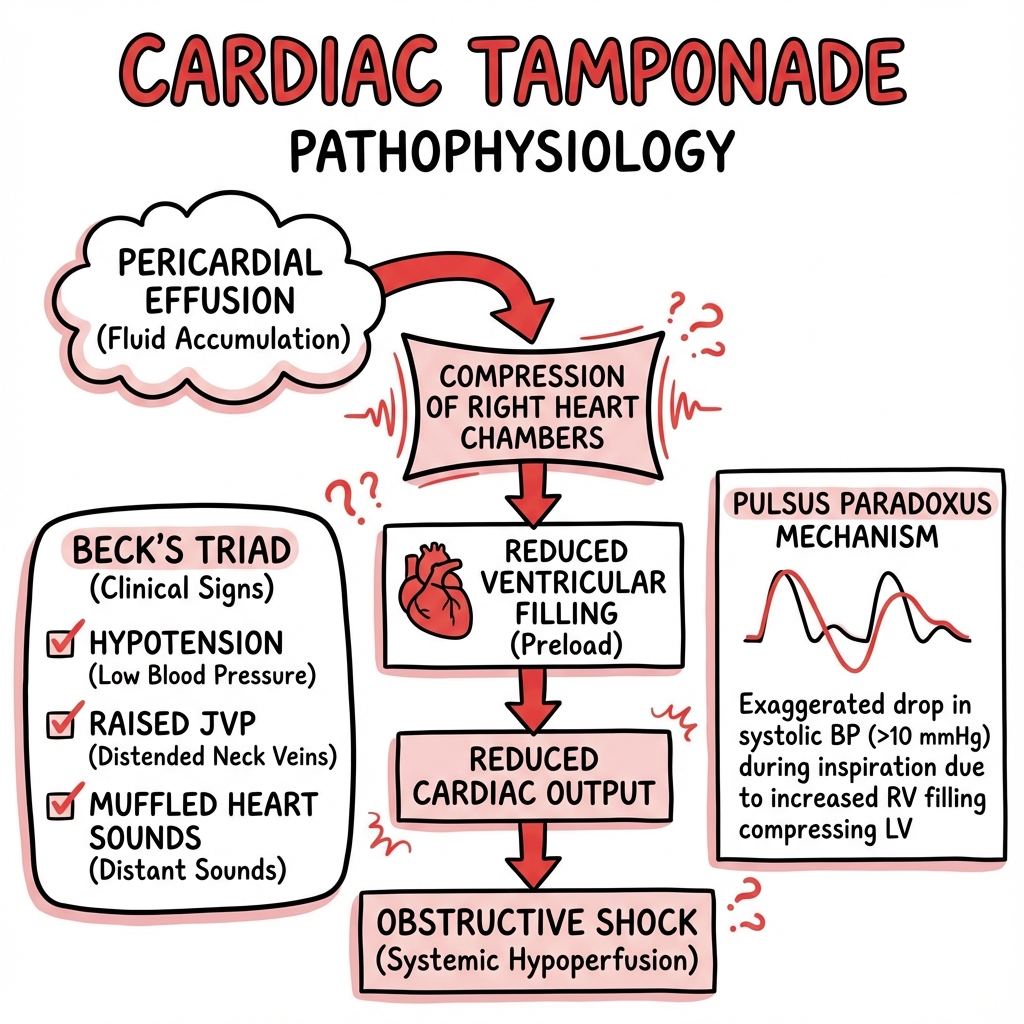
Cardiac tamponade is a life-threatening cardiovascular emergency characterized by accumulation of fluid within the pericardial space, resulting in elevated intrapericardial pressure and impaired ventricular filling. [1,2] The condition represents the extreme end of the spectrum of pericardial effusion, where hemodynamic compromise occurs due to external compression of the cardiac chambers. Tamponade is fundamentally a clinical diagnosis based on signs of shock and elevated jugular venous pressure, though echocardiography provides confirmatory evidence and guides intervention. [3]
The rate of fluid accumulation is often more critical than absolute volume—acute accumulation of as little as 100-200 mL can cause tamponade, whereas chronic effusions may reach 1-2 L without hemodynamic consequences due to pericardial stretch. [4] Emergency drainage via pericardiocentesis or surgical intervention is lifesaving and should not be delayed when clinical tamponade is evident.
Key Facts
- Definition: Pericardial fluid causing compression of cardiac chambers with hemodynamic compromise
- Beck's triad: Hypotension, raised JVP, muffled heart sounds (specific but present in only 33% of cases) [5]
- Pulsus paradoxus: Exaggerated inspiratory fall in systolic BP (> 10 mmHg) present in 70-80% of cases [6]
- Echo findings: Right atrial and/or right ventricular diastolic collapse (sensitivity 90%, specificity 65%) [7]
- Treatment: Emergency pericardiocentesis or surgical drainage
- Volume-time relationship: Rapid accumulation (hours) more dangerous than slow (weeks-months)
- Reversible cause of PEA: One of the "4 Hs and 4 Ts" in cardiac arrest algorithms
Clinical Pearls
Beck's triad is highly specific (97%) but poorly sensitive (33%) — absence does not exclude tamponade [5]
Electrical alternans (alternating QRS amplitude) is seen in only 10-20% of cases but is highly specific for large effusions [8]
In trauma, suspect tension pneumothorax first if unilateral breath sounds — both can cause elevated JVP and hypotension
Avoid positive pressure ventilation before drainage — increased intrathoracic pressure further reduces venous return and can precipitate cardiovascular collapse [9]
Small volume aspiration (30-50 mL) can produce dramatic hemodynamic improvement due to the steep pressure-volume relationship in acute tamponade [10]
Why This Matters Clinically
Tamponade is a medical emergency with mortality approaching 100% if untreated, yet survival exceeds 85% with prompt recognition and drainage. [11] Early identification requires high clinical suspicion, as classic signs may be absent or subtle. The condition is increasingly recognized as a complication of cardiac interventions (catheterization, pacemaker insertion, ablation procedures), occurring in 0.1-1.2% of electrophysiology procedures. [12] Echocardiography has revolutionized diagnosis, but treatment decisions must be based on clinical status rather than imaging findings alone — patients with tamponade physiology on echo but stable hemodynamics may be managed conservatively with close monitoring.
Epidemiology
Incidence and Prevalence
Precise incidence data are limited due to the heterogeneous etiologies and acute presentation. Hospital-based studies suggest cardiac tamponade accounts for approximately 2% of pericardial disease presentations. [13] In intensive care settings, the incidence is higher (5-8%) due to post-cardiac surgery and iatrogenic causes. [14]
Etiology by Geographic Region and Setting
Developed Countries:
- Malignancy (most common): 50-65% of cases, particularly lung cancer, breast cancer, and lymphoma [15]
- Iatrogenic: 15-20%, including post-cardiac surgery, catheterization, pacemaker insertion
- Idiopathic/viral pericarditis: 10-15%
- Post-myocardial infarction: 5-10% (free wall rupture, Dressler's syndrome)
- Aortic dissection: 5-7% of tamponade cases [16]
Endemic Areas (Sub-Saharan Africa, South Asia):
- Tuberculous pericarditis: 40-70% of tamponade cases [17]
- HIV-associated pericardial disease: significant contributor in high-prevalence regions
- Malignancy: lower proportion but increasing
Trauma:
- Penetrating chest injury: 80% of traumatic tamponade cases
- Blunt chest trauma: 20%, associated with steering wheel injuries, high-velocity impact
Specific Etiologies and Clinical Context
| Cause | Mechanism | Clinical Context |
|---|---|---|
| Malignancy | Metastatic spread, lymphatic obstruction | Lung (35%), breast (25%), lymphoma (15%), melanoma (5%) [15] |
| Iatrogenic | Direct cardiac perforation, hemorrhage | Central line insertion, cardiac catheterization, pacemaker leads, EP ablation [12] |
| Acute Myocardial Infarction | Free wall rupture (days 3-5 post-MI), hemorrhagic pericarditis | Sudden hemodynamic collapse 3-14 days post-MI; mortality > 90% without surgery [18] |
| Aortic Dissection | Type A dissection extending into pericardium | Abrupt onset, often fatal; requires emergency surgery [16] |
| Tuberculous Pericarditis | Granulomatous inflammation, exudative effusion | Subacute presentation, fever, night sweats; common in HIV patients [17] |
| Uremic Pericarditis | Metabolic toxins, fibrinous inflammation | CKD stage 5, missed dialysis sessions; less common with modern dialysis [19] |
| Post-Cardiac Surgery | Hemorrhage, anticoagulation | Occurs in 0.5-5% of cardiac surgeries, typically within 24-48 hours |
| Autoimmune | SLE, rheumatoid arthritis, systemic sclerosis | Part of systemic disease; may be recurrent |
| Infection | Viral (coxsackie, influenza), bacterial (purulent), fungal | Viral most common in immunocompetent; purulent in immunocompromised |
| Trauma | Hemopericardium from cardiac laceration | Penetrating (knife, gunshot) > blunt injury |
Demographics
- Age: Bimodal distribution
- "Young adults (20-40 years): trauma, viral pericarditis, autoimmune"
- "Older adults (60+ years): malignancy, iatrogenic, post-MI, aortic dissection"
- Sex: Males slightly predominant (1.5:1) in traumatic and iatrogenic causes; equal in malignancy
- Comorbidities: Chronic kidney disease, cancer, recent cardiac surgery/intervention, tuberculosis exposure
Pathophysiology
Normal Pericardial Anatomy and Physiology
The pericardium consists of two layers:
- Visceral pericardium (epicardium): adheres to myocardium
- Parietal pericardium: fibrous outer layer
The pericardial space normally contains 15-50 mL of serous fluid (ultrafiltrate of plasma) that reduces friction during cardiac motion. [20] Normal intrapericardial pressure is subatmospheric to slightly positive (−5 to +5 mmHg), closely tracking right atrial pressure.
Molecular and Cellular Mechanisms
Fluid Accumulation:
- Increased production: Inflammation (cytokine-mediated increased vascular permeability), infection (bacterial toxins, viral cytopathic effects), malignancy (VEGF secretion promoting angiogenesis and leakage)
- Decreased absorption: Lymphatic obstruction (malignant infiltration, radiation fibrosis), inflammation of lymphatic channels
- Direct hemorrhage: Cardiac perforation (iatrogenic, trauma), myocardial rupture (post-MI), aortic dissection, anticoagulation-related bleeding
Pericardial Compliance: The pericardium exhibits viscoelastic properties described by a compliance curve with two phases:
- Initial flat phase: Small volume increases cause minimal pressure rise (pericardium stretches)
- Steep phase: Once pericardial reserve exhausted, small additional volume causes exponential pressure rise [4]
Acute effusions reach the steep phase rapidly (limited stretch time), whereas chronic effusions allow pericardial remodeling and increased compliance.
Hemodynamic Consequences
Stage 1: Compensated Pericardial Effusion
- Intrapericardial pressure rises but remains below right atrial pressure
- Normal cardiac filling preserved
- Tachycardia develops to maintain cardiac output (CO = HR × SV)
- Asymptomatic or mild dyspnea
Stage 2: Early Tamponade (Tamponade Physiology)
- Intrapericardial pressure exceeds right atrial pressure
- Diastolic filling impaired, beginning with right atrium (lowest pressure chamber)
- Right atrial collapse during ventricular systole, right ventricular collapse during diastole [7]
- Venous pressure rises to maintain filling gradient
- Stroke volume decreases; compensatory tachycardia and peripheral vasoconstriction
- Equalization of diastolic pressures across all chambers (RA = RV = LA = LV end-diastolic pressure)
Stage 3: Clinical Tamponade
- Severe reduction in stroke volume
- Hypotension despite maximal sympathetic drive
- Peripheral hypoperfusion (cool extremities, oliguria, altered mental status)
- Risk of progression to pulseless electrical activity (PEA) arrest
Pulsus Paradoxus: Mechanism
Pulsus paradoxus is an exaggeration of the normal physiological phenomenon where systolic blood pressure decreases during inspiration. [6]
Normal physiology:
- Inspiration → negative intrathoracic pressure → increased venous return to right heart
- RV filling increases, RV volume expands
- In health, LV output decreases less than 10 mmHg due to septal shift and reduced pulmonary venous return
In tamponade:
- Fixed pericardial volume constraint
- Inspiration → increased RV filling → RV expansion → interventricular septum shifts leftward
- LV end-diastolic volume decreases significantly
- LV stroke volume and systolic BP fall > 10 mmHg (often 20-40 mmHg in severe tamponade) [6]
Absence of pulsus paradoxus does NOT exclude tamponade:
- Atrial septal defect (equalized atrial pressures)
- Aortic regurgitation (diastolic flow maintains pressure)
- Regional tamponade (localized effusion post-cardiac surgery)
- Severe hypotension (insufficient pressure to measure paradox)
- Left ventricular hypertrophy (stiff LV resists compression)
Rate of Accumulation: Volume-Time Relationship
| Scenario | Volume | Time Course | Pericardial Response | Outcome |
|---|---|---|---|---|
| Acute hemorrhage (trauma, aortic dissection, post-MI rupture) | 100-200 mL | Minutes to hours | No time for stretch; steep pressure rise | Tamponade likely |
| Subacute inflammatory (viral, uremic, autoimmune) | 300-800 mL | Days to weeks | Limited stretch; moderate pressure rise | Variable tamponade risk |
| Chronic malignant/tuberculous | 1000-2000 mL | Weeks to months | Significant pericardial remodeling; gradual pressure rise | Tamponade only if rapid increase |
This explains why chronic dialysis patients may tolerate large effusions asymptomatically, whereas acute post-procedural hemorrhage causes rapid decompensation.
Emergency Recognition and Management
Cardiac Tamponade — Emergency Algorithm
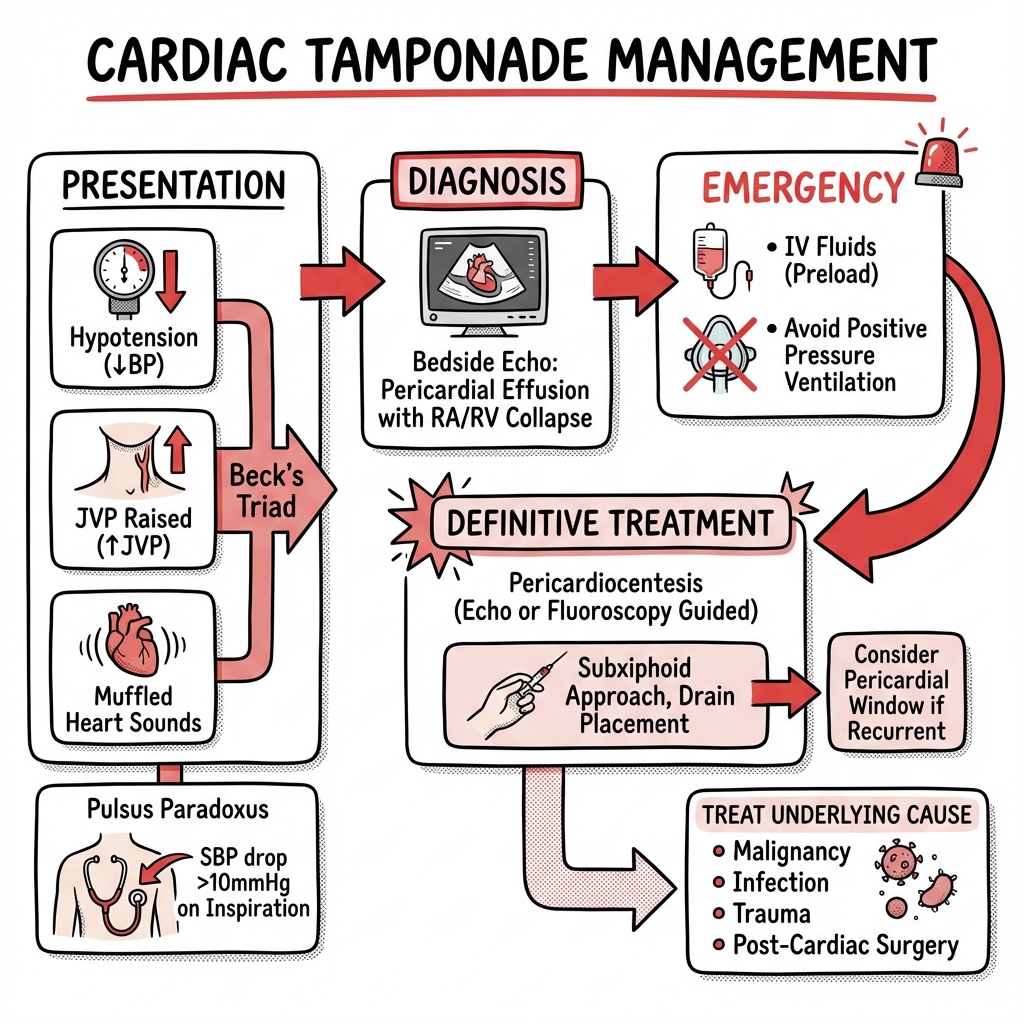
Clinical Diagnosis (Do Not Wait for Imaging):
Major Signs:
- Hypotension — systolic BP less than 90 mmHg or MAP less than 65 mmHg
- Elevated jugular venous pressure — JVP raised, may be dramatically elevated (> 20 cm H₂O)
- Tachycardia — HR > 100 bpm (unless on beta-blockers or advanced shock)
- Pulsus paradoxus — > 10 mmHg inspiratory fall in systolic BP
Beck's Triad (Classic but Only 33% Sensitive): [5]
- Hypotension
- Elevated JVP
- Muffled heart sounds
Additional Signs:
- Cool, clammy peripheries (peripheral vasoconstriction)
- Oliguria (less than 0.5 mL/kg/hr)
- Altered mental status (cerebral hypoperfusion)
- Narrow pulse pressure
- Kussmaul's sign (JVP rises paradoxically on inspiration) — rare in tamponade, more common in constriction
High-Risk Scenarios:
- Recent cardiac procedure (catheterization, pacemaker, ablation, cardiac surgery)
- Known malignancy with chest pain or dyspnea
- Trauma with chest injury and hypotension
- PEA arrest with distended neck veins
- Aortic dissection (acute Type A)
- Renal failure with missed dialysis
Immediate Management — ABCDE Approach
A — Airway
- Maintain airway patency
- AVOID intubation and positive pressure ventilation if possible — increased intrathoracic pressure reduces venous return and can precipitate cardiovascular collapse [9]
- If intubation unavoidable, pre-drainage pericardiocentesis essential; use minimal PEEP and tidal volumes
B — Breathing
- High-flow oxygen (15 L/min non-rebreather or titrate to SpO₂ > 94%)
- Maintain spontaneous ventilation if possible
C — Circulation
1. IV Access and Monitoring
- Two large-bore IV cannulae (16G or larger)
- Continuous cardiac monitoring (ECG, BP, SpO₂)
- Arterial line if time permits (helpful for pulsus paradoxus measurement and post-drainage monitoring)
2. Fluid Resuscitation
- Cautious fluid bolus (250-500 mL crystalloid over 15-30 minutes) [21]
- Rationale: Increases right atrial pressure, partially overcomes elevated intrapericardial pressure, transiently improves filling
- Risk: Excessive fluid may worsen tamponade by increasing pericardial fluid transudation
- Monitor response closely; if no improvement after 500 mL, proceed urgently to drainage
3. Inotropic Support
- Limited benefit but may temporize while preparing for drainage
- Dobutamine 5-10 mcg/kg/min (increases contractility) or noradrenaline 0.05-0.2 mcg/kg/min (increases systemic vascular resistance)
- Not a substitute for definitive drainage
4. Avoid Medications That Worsen Tamponade
- Diuretics — reduce preload, worsen hypotension
- Vasodilators (nitrates, ACE inhibitors) — reduce preload and afterload, precipitate collapse
- Sedatives (for intubation) — reduce sympathetic drive, worsen hypotension
D — Disability
- Glasgow Coma Scale (GCS) assessment
- Altered mental status suggests severe cerebral hypoperfusion — proceed emergently to drainage
E — Exposure and Examination
- Look for trauma (penetrating wounds, steering wheel injury, rib fractures)
- Assess for sources of bleeding (surgical drains, chest tubes)
Call for Help
- Cardiology (for pericardiocentesis)
- Cardiothoracic surgery (for surgical drainage if pericardiocentesis fails, traumatic tamponade, aortic dissection, post-MI rupture)
- Interventional radiology (alternative drainage if available)
- Anesthesia/ICU (for procedural support and post-drainage care)
Bedside Echocardiography (POCUS)
Do not delay treatment if clinical diagnosis clear, but echocardiography highly useful when available:
Echo Findings Supporting Tamponade: [7,22]
- Pericardial effusion — circumferential anechoic (black) space around heart
- Right atrial collapse — during ventricular systole (highly sensitive, 85-90%)
- Right ventricular diastolic collapse — highly specific (90-95%) for hemodynamically significant tamponade
- Respiratory variation in mitral/tricuspid inflow (> 25% variation)
- Dilated inferior vena cava (> 2 cm) with absent or reduced respiratory collapse (less than 50% collapse with sniff)
- Swinging heart — excessive cardiac motion within large effusion (associated with electrical alternans)
Parasternal Long-Axis View:
- Visualize effusion anterior and posterior to heart
- Assess RV size and collapse
Apical 4-Chamber View:
- Assess RA and RV collapse
- Measure respiratory variation in tricuspid inflow
Subcostal View:
- Best view for pericardiocentesis planning
- Assess IVC diameter and collapsibility
Regional vs. Circumferential Effusion:
- Post-cardiac surgery: loculated effusion (often posterior) may cause regional tamponade without typical echo signs
Emergency Pericardiocentesis — Technique
Indications:
- Clinical tamponade with hemodynamic instability
- PEA arrest with suspected tamponade
Contraindications (Relative):
- Aortic dissection (requires surgical drainage)
- Purulent pericarditis (requires surgical drainage and washout)
- Traumatic hemopericardium (often requires thoracotomy for definitive control)
- Post-MI free wall rupture (high risk of rebleeding; surgical repair preferred)
Equipment:
- Pericardiocentesis kit or 18G needle (spinal needle, angiocath)
- 20-50 mL syringe
- Three-way stopcock
- Sterile gown, gloves, drapes, antiseptic
- Local anesthetic (1% lidocaine)
- ECG monitoring (alligator clip to needle for ST elevation detection if myocardium contacted)
- Ultrasound guidance if available (reduces complications by 50%) [23]
- Guidewire and pigtail catheter (for prolonged drainage)
Subxiphoid (Pericardial) Approach: [10,23]
- Position: Patient at 30-45 degree angle (improves effusion pooling anteriorly)
- Landmark: 1-2 cm inferior and to the left of the xiphisternum
- Local anesthesia: Infiltrate skin, subcutaneous tissue, and periosteum
- Needle insertion:
- Insert needle at 30-45 degree angle to skin
- Aim toward left shoulder
- Advance slowly while applying negative pressure to syringe
- Signs of pericardial entry:
- Loss of resistance
- Aspiration of non-clotting blood (pericardial blood defibrinated) or straw-colored fluid
- If bright red pulsatile blood → ventricular puncture; withdraw and redirect
- ECG monitoring: ST elevation or ectopy suggests myocardial contact — withdraw slightly
- Aspiration: Remove fluid slowly; even 30-50 mL can dramatically improve hemodynamics in acute tamponade
- Catheter placement: If ongoing drainage needed, advance guidewire through needle, remove needle, place pigtail catheter over wire (Seldinger technique)
- Confirm position: Aspiration of fluid, no resistance, hemodynamic improvement
Alternative Approaches:
- Apical: 1 cm lateral to apex beat, directed toward right shoulder (higher risk of ventricular puncture)
- Parasternal: 5th intercostal space, parasternal (rarely used; risk of internal mammary artery injury)
Ultrasound-Guided Pericardiocentesis: [23]
- Mark site with largest effusion (subcostal or apical)
- Real-time visualization during needle advancement
- Reduces complications (cardiac puncture, pneumothorax) by approximately 50%
- Essential for loculated effusions
Volume Drainage:
- Acute tamponade: 50-100 mL often sufficient for immediate hemodynamic improvement
- Continue drainage until:
- Hemodynamics stabilize (BP rises, HR falls, JVP normalizes)
- Drainage ceases (less than 25 mL/hr)
- Resistance encountered (drained dry)
- Send fluid for analysis (see Investigations section)
Post-Procedure:
- Repeat echocardiography to confirm resolution
- Monitor for re-accumulation (especially malignancy, TB)
- If catheter left in situ, secure and monitor drainage
- Admit to ICU/HDU for observation
Complications of Pericardiocentesis: [23]
- Cardiac puncture (most common, 5-15%): Usually self-limiting if recognized; withdraw needle
- Coronary artery laceration (1-2%): Requires emergency surgical repair
- Arrhythmias (atrial or ventricular ectopy, VT/VF): Usually resolve with needle withdrawal
- Pneumothorax (3-5%): Risk reduced with subcostal approach
- Hemothorax: Rare
- Infection (1%): Use strict aseptic technique
- Vasovagal reaction: Pre-treat with atropine if prone
- Re-accumulation (20-30%, especially malignancy): May require repeat drainage or surgical window
Surgical Drainage — Indications and Techniques
Indications for Surgical Drainage:
- Traumatic hemopericardium — ongoing hemorrhage requires direct visualization and repair
- Aortic dissection — Type A dissection with tamponade requires emergency ascending aorta repair
- Post-MI free wall rupture — requires surgical patch repair
- Purulent pericarditis — requires washout and drainage; pericardiocentesis insufficient
- Recurrent tamponade — malignancy, post-surgical, idiopathic with multiple recurrences
- Loculated effusion — post-cardiac surgery with posterior/regional tamponade
- Failed pericardiocentesis — inability to access fluid, loculated, thick pericardium
Surgical Options:
| Procedure | Technique | Indication | Recurrence Rate |
|---|---|---|---|
| Subxiphoid pericardial window | Small incision below xiphoid, pericardium opened, window created for drainage | Recurrent effusions, malignancy | 10-15% |
| Thoracoscopic (VATS) window | Video-assisted thoracoscopic pericardial window and biopsy | Diagnostic + therapeutic, smaller effusions | 5-10% |
| Pericardiectomy | Surgical removal of pericardium | Constrictive pericarditis, severe recurrent tamponade | less than 5% |
| Thoracotomy | Open chest exploration | Trauma, post-MI rupture, aortic dissection, failed minimally invasive approaches | N/A (acute setting) |
Pericardial Window Technique (Subxiphoid):
- Midline incision inferior to xiphoid
- Dissect to pericardium
- Create 4-6 cm window in pericardium
- Drain fluid, send for analysis and biopsy
- Leave drain in pericardial space
Advantages of Surgical Drainage:
- Tissue biopsy for diagnosis (TB, malignancy)
- More complete drainage
- Lower recurrence rate
- Definitive for loculated or thick effusions
Special Scenarios
Traumatic Cardiac Tamponade:
- Penetrating trauma (stab wounds, gunshot): High suspicion; often requires thoracotomy
- Blunt trauma: Less common; consider in high-velocity impacts
- Emergency Department thoracotomy indicated for PEA arrest with suspected tamponade in penetrating trauma
- Pericardiocentesis often temporizing only; definitive surgical repair needed
- Hemopericardium may clot, making aspiration difficult
Post-Myocardial Infarction:
- Free wall rupture: Sudden hemodynamic collapse days 3-5 post-STEMI; mortality > 90% without emergency surgery [18]
- Hemorrhagic pericarditis: Low-grade effusion post-MI; rarely causes tamponade
- Dressler's syndrome: Autoimmune pericarditis 2-10 weeks post-MI; treated with NSAIDs/colchicine; tamponade rare
Iatrogenic (Post-Procedural):
- Occurs 0.1-1.2% of electrophysiology procedures, 0.5-2% cardiac catheterizations [12]
- Early recognition critical — monitor for hypotension, elevated JVP post-procedure
- Reverse anticoagulation (protamine for heparin)
- Immediate echocardiography and drainage if hemodynamically unstable
Aortic Dissection:
- Type A dissection with pericardial involvement — DO NOT perform pericardiocentesis (may precipitate exsanguination by relieving tamponade and allowing ongoing bleeding)
- Emergency surgical repair (ascending aorta replacement)
- Diagnosis: CT aorta or transesophageal echocardiography (TEE)
Malignant Pericardial Effusion:
- Recurrence rate 40-60% after pericardiocentesis alone [15]
- Consider pericardial window or sclerosis (tetracycline, bleomycin) for recurrent effusions
- Systemic chemotherapy for underlying malignancy
- Prognosis poor (median survival 3-12 months depending on cancer type)
PEA Arrest:
- If cardiac arrest with tamponade suspected (distended neck veins, recent cardiac procedure, trauma):
- Continue CPR
- Emergency pericardiocentesis during resuscitation (subxiphoid approach)
- Consider resuscitative thoracotomy if pericardiocentesis unavailable or failed
Clinical Presentation
Symptoms
Acute Tamponade (Hours to Days):
- Dyspnea (most common, 85-90%) — progressive, worse on exertion
- Chest discomfort or pain (50-70%) — dull, retrosternal, may be pleuritic
- Presyncope or syncope (20-30%) — cerebral hypoperfusion
- Anxiety, restlessness — air hunger sensation
- Orthopnea — inability to lie flat
Subacute/Chronic Tamponade (Weeks to Months):
- Fatigue, weakness (prominent)
- Dyspnea on exertion
- Abdominal distension (ascites from elevated venous pressures)
- Anorexia, early satiety
- Peripheral edema (late finding)
Symptoms of Underlying Etiology:
- Fever, night sweats, weight loss — tuberculosis, malignancy
- Pleuritic chest pain — pericarditis (viral, autoimmune)
- Nausea, pruritus — uremia
- Sudden-onset tearing chest/back pain — aortic dissection
- Recent chest trauma — hemopericardium
Physical Examination
Cardiovascular:
Beck's Triad (33% sensitive, 97% specific): [5]
- Hypotension — systolic BP less than 90 mmHg
- Elevated jugular venous pressure — JVP often markedly raised (> 10 cm H₂O above sternal angle); non-pulsatile or poorly pulsatile
- Muffled (distant) heart sounds — reduced intensity due to fluid insulating heart from chest wall
Pulsus Paradoxus: [6]
- Present in 70-80% of tamponade cases
- Measured via:
- Palpation: Radial pulse weakens or disappears during inspiration
- Sphygmomanometry:
- Inflate cuff above systolic pressure
- Slowly deflate; note pressure when Korotkoff sounds first heard (during expiration only)
- Continue deflating; note pressure when sounds heard throughout respiratory cycle
- Difference > 10 mmHg = positive pulsus paradoxus
Other Cardiovascular Signs:
- Tachycardia (HR > 100 bpm, unless beta-blockade or advanced shock with bradycardia)
- Narrow pulse pressure (less than 25 mmHg)
- Cool, clammy peripheries (peripheral vasoconstriction, hypoperfusion)
- Weak peripheral pulses
- Kussmaul's sign — paradoxical rise in JVP with inspiration (rare in tamponade; more typical of constrictive pericarditis)
Respiratory:
- Tachypnea (RR > 20/min)
- Clear lung fields (unless coexistent pulmonary pathology)
Abdominal:
- Hepatomegaly (chronic tamponade with venous congestion)
- Ascites (chronic cases)
Neurological:
- Altered mental status, confusion (cerebral hypoperfusion)
- Restlessness, agitation
Other:
- Peripheral edema (late, chronic tamponade)
- Evidence of underlying disease (cachexia in malignancy, uremic frost in renal failure, surgical scars)
Investigations
Bedside Investigations
Electrocardiography (ECG):
Common ECG Findings: [8]
- Sinus tachycardia (most common, nonspecific)
- Low voltage QRS complexes (less than 5 mm in limb leads, less than 10 mm in precordial leads) — seen in 40% of cases; caused by fluid insulating electrical signal
- Electrical alternans (10-20% of cases) — beat-to-beat alternation in QRS amplitude (occasionally P wave and T wave alternans too); highly specific for large effusion with "swinging heart" motion within pericardial sac [8]
- Diffuse ST elevation and PR depression — if coexistent acute pericarditis
- Atrial fibrillation — may develop due to atrial stretch
- Electromechanical dissociation (PEA) — in cardiac arrest
ECG Changes by Etiology:
- Post-MI: Q waves, ST changes of infarction
- Aortic dissection: May show inferior MI pattern if dissection involves right coronary ostium
- Uremic pericarditis: Non-specific ST-T changes
Echocardiography (Transthoracic Echo — TTE):
Gold standard for diagnosis and assessment of hemodynamic significance. [7,22]
Effusion Characteristics:
- Size: Small (less than 10 mm echo-free space), moderate (10-20 mm), large (> 20 mm)
- Distribution: Circumferential vs. loculated (posterior loculation common post-cardiac surgery)
- Echogenicity: Anechoic (transudative), echogenic (hemorrhagic, purulent, malignant)
Signs of Tamponade Physiology: [7]
| Echo Finding | Sensitivity | Specificity | Significance |
|---|---|---|---|
| Right atrial collapse (during ventricular systole) | 85-90% | 60-70% | Early sign; highly sensitive |
| Right ventricular diastolic collapse (early diastole) | 50-65% | 90-95% | Highly specific for hemodynamically significant tamponade |
| Respiratory variation in mitral inflow (> 25% decrease with inspiration) | 70-80% | 90% | Doppler finding; exaggerated ventricular interdependence |
| Respiratory variation in tricuspid inflow (> 40% increase with inspiration) | 75-85% | 90% | Mirrors pulsus paradoxus at valve level |
| IVC plethora (diameter > 2 cm with less than 50% inspiratory collapse) | 90% | 60% | Reflects elevated right atrial pressure |
| Swinging heart | 60-80% (large effusions) | 70% | Associated with electrical alternans |
Absence of RV collapse does NOT exclude tamponade:
- RV hypertrophy (pulmonary hypertension)
- Regional tamponade (loculated effusion)
- Positive pressure ventilation (alters intrathoracic pressures)
Transesophageal Echocardiography (TEE):
- Superior visualization of posterior effusions and loculated collections
- Useful intraoperatively or in mechanically ventilated patients
- Required to exclude aortic dissection in appropriate clinical context
Laboratory Investigations
Blood Tests:
| Test | Purpose | Expected Findings in Tamponade |
|---|---|---|
| Full blood count | Infection, anemia | Leukocytosis (infection), anemia (chronic disease, malignancy) |
| Inflammatory markers (CRP, ESR) | Infection, autoimmune | Elevated in TB, bacterial infection, autoimmune diseases |
| Troponin | Myopericarditis, post-MI | Elevated in myopericarditis, acute MI |
| Renal function (urea, creatinine, eGFR) | Uremia | Elevated in uremic pericarditis (urea > 30 mmol/L, Cr > 500 µmol/L) |
| Thyroid function (TSH, free T4) | Hypothyroidism | Rare cause; TSH elevated, T4 low |
| Autoimmune screen (ANA, dsDNA, RF, complement) | SLE, RA, systemic sclerosis | Positive ANA, dsDNA (SLE); RF (RA) |
| Tumor markers (CEA, CA 19-9, CA 125) | Malignancy | Elevated in some cancers (not specific) |
| HIV test | HIV-associated pericarditis | Consider in endemic areas or high-risk patients |
| Coagulation profile (PT, APTT, INR) | Bleeding risk, procedural planning | May be deranged if on anticoagulation or liver disease |
Pericardial Fluid Analysis:
Essential for all drained fluid: [13]
Appearance and Basic Tests:
- Color and clarity: Straw-colored (transudative), bloody (hemorrhagic), purulent (bacterial), turbid (malignant, TB)
- Hematocrit: If fluid hematocrit > 50% of blood hematocrit → hemopericardium (trauma, aortic dissection, post-MI rupture)
Biochemistry:
- Protein: Exudate (> 3 g/dL), transudate (less than 3 g/dL)
- LDH: Exudate (> 200 IU/L or fluid:serum LDH ratio > 0.6)
- Glucose: Low (less than 60 mg/dL) in bacterial or TB pericarditis
- Adenosine deaminase (ADA): Elevated (> 40 U/L) suggests tuberculous pericarditis [17]; sensitivity 90%, specificity 85% in endemic regions
Cell Count and Differential:
- WBC count:
-
10,000/µL → infection (bacterial, TB)
- Lymphocytic predominance (> 50%) → TB, viral, malignancy
- Neutrophilic predominance (> 50%) → bacterial, purulent
-
Microbiology:
- Gram stain and culture (bacterial)
- Acid-fast bacilli (AFB) stain and mycobacterial culture (TB) [17]
- Fungal stains and culture (immunocompromised)
- Viral PCR (coxsackie, HIV, influenza)
Cytology:
- Malignant cells: Sensitivity 60-90% depending on tumor type [15]
- Send generous volume (50-100 mL) for cytospin and cell block
Flow Cytometry:
- If lymphoma suspected
Imaging
Chest X-Ray (CXR):
Findings:
- Cardiomegaly ("water bottle" or "globular" heart) — seen with effusions > 250 mL
- Loss of normal cardiac contours
- Clear lung fields (unless coexistent pulmonary pathology)
- May be normal in acute tamponade (small volume, rapid accumulation)
Associated Findings:
- Widened mediastinum — aortic dissection
- Rib fractures, pneumothorax — trauma
- Pulmonary infiltrates, hilar lymphadenopathy — TB, malignancy
- Pleural effusions — malignancy, heart failure, TB
CT Chest (with IV Contrast):
Indications:
- Suspected aortic dissection (CT angiography)
- Evaluation of underlying malignancy (lung, mediastinal masses)
- Characterization of complex/loculated effusions
- Pre-surgical planning
Findings:
- Pericardial effusion: Fluid density (0-20 HU transudative, 30-60 HU exudative, 40-80 HU hemorrhagic)
- Pericardial thickening (> 4 mm) — infection, malignancy, post-radiation
- Mass lesions — malignancy, pericardial cyst
- Aortic dissection: Intimal flap, false lumen
- Pulmonary embolism (if CT-PA protocol)
Cardiac MRI:
- Rarely used in acute setting
- Useful for characterizing pericardial tissue, detecting inflammation, tumor infiltration
Differential Diagnosis
Cardiac tamponade must be distinguished from other causes of shock and elevated JVP:
| Condition | JVP | Pulsus Paradoxus | Heart Sounds | Echo Findings | Key Differentiator |
|---|---|---|---|---|---|
| Cardiac tamponade | Elevated | Present (70-80%) | Muffled | Pericardial effusion, RA/RV collapse | Pericardial effusion on echo |
| Constrictive pericarditis | Elevated | Rare (less than 33%) | Normal/loud | Thickened pericardium, septal bounce | Pericardial calcification on CT; no effusion |
| Restrictive cardiomyopathy | Elevated | Absent | S3/S4 | Biatrial enlargement, normal pericardium | Biopsy shows infiltration (amyloid, sarcoid) |
| Right ventricular infarction | Elevated | May be present | Normal | RV hypokinesis, no effusion | Inferior MI on ECG, troponin elevated |
| Massive pulmonary embolism | Elevated | May be present | Loud P2 | RV dilation, no effusion | CT-PA shows embolus; D-dimer elevated |
| Tension pneumothorax | Elevated | Absent | Reduced unilaterally | Not applicable | Absent breath sounds, tracheal deviation, CXR diagnostic |
| Cardiogenic shock (LV failure) | May be normal/low | Absent | S3 gallop | LV systolic dysfunction, no effusion | Pulmonary edema on CXR |
| Hypovolemic shock | Low/flat | Absent | Normal | Collapsed IVC | Responds to fluid resuscitation |
Management
Acute Stabilization
Immediate Measures (Pre-Drainage):
-
Fluid Resuscitation:
- 250-500 mL crystalloid IV bolus [21]
- Increases right atrial pressure, transiently improves preload
- Avoid over-resuscitation (> 1 L) as may worsen effusion
-
Inotropic Support (Temporizing):
- Dobutamine 5-10 mcg/kg/min (increases contractility)
- Noradrenaline 0.05-0.2 mcg/kg/min (increases afterload, maintains BP)
- Not a substitute for drainage
-
Avoid Harmful Interventions:
- No diuretics (reduce preload, worsen hypotension)
- No vasodilators (worsen hypotension)
- Avoid positive pressure ventilation (reduces venous return) [9]
- Avoid sedation unless absolutely necessary (reduces sympathetic drive)
Definitive Treatment — Pericardial Drainage:
See Emergency Management section for pericardiocentesis technique.
Post-Drainage Care:
- Admit to ICU/HDU
- Continuous cardiac monitoring
- Repeat echocardiography at 6-24 hours to assess for re-accumulation
- If pigtail catheter in situ: Monitor drainage, remove when less than 25 mL/24 hours
- Investigate underlying cause
Treatment by Etiology
| Cause | Acute Management | Definitive Treatment | Recurrence Prevention |
|---|---|---|---|
| Malignant | Pericardiocentesis | Systemic chemotherapy, pericardial window, intrapericardial sclerosis (tetracycline, bleomycin) | Oncology referral; consider pericardial window for recurrent effusions [15] |
| Tuberculous | Pericardiocentesis | Standard anti-TB therapy (rifampicin, isoniazid, pyrazinamide, ethambutol × 6 months); adjunctive corticosteroids (prednisolone 1 mg/kg/day, taper over 6 weeks) reduce mortality in HIV-negative patients [17] | Complete anti-TB course; monitor for constrictive pericarditis (develops in 20-30%) |
| Iatrogenic | Pericardiocentesis, reverse anticoagulation (protamine for heparin) | Surgical repair if vessel injury; conservative if self-limiting | Prevention: Careful technique, imaging guidance for procedures |
| Uremic | Pericardiocentesis if hemodynamically unstable | Intensive dialysis (daily until effusion resolves); NSAIDs (indomethacin 50 mg TDS) [19] | Regular dialysis; avoid missing sessions |
| Post-MI rupture | Emergency surgery (pericardiocentesis may temporize en route to OR) | Surgical patch repair of rupture [18] | N/A (acute catastrophic event) |
| Aortic dissection | DO NOT drain pericardium (may precipitate exsanguination); emergency surgery | Ascending aorta replacement (Type A dissection) [16] | Blood pressure control, aortic surveillance |
| Viral/Idiopathic | Pericardiocentesis if tamponade | NSAIDs (ibuprofen 600 mg TDS) + colchicine (0.5 mg BD) × 3 months | Colchicine reduces recurrence from 30% to 10% |
| Autoimmune (SLE, RA) | Pericardiocentesis | Corticosteroids (prednisolone 1 mg/kg/day), DMARDs, treat underlying disease | Immunosuppression, disease-modifying therapy |
| Bacterial (purulent) | Surgical drainage (pericardial window or pericardiectomy), broad-spectrum antibiotics | Prolonged IV antibiotics (4-6 weeks), surgical washout | Complete antibiotic course |
| Traumatic | Emergency thoracotomy (pericardiocentesis temporizing only) | Surgical repair of cardiac laceration | N/A (trauma management) |
Recurrent Pericardial Effusion and Tamponade
Recurrence Rates:
- Post-pericardiocentesis alone: 20-30% overall; up to 60% in malignancy [15]
- Post-pericardial window: 10-15%
- Post-pericardiectomy: less than 5%
Management Options for Recurrence:
1. Repeat Pericardiocentesis
- Suitable for infrequent recurrences
- Low-risk procedure
- Definitive underlying treatment required
2. Pericardial Window (Subxiphoid or VATS)
- Creates permanent drainage pathway from pericardium to pleural/peritoneal cavity
- Recurrence rate 10-15%
- Allows tissue biopsy for diagnosis
- Preferred for malignant effusions, recurrent idiopathic effusions
3. Intrapericardial Sclerosis
- Instillation of sclerosant (tetracycline, doxycycline, bleomycin) via pericardial catheter
- Induces adhesions, obliterates pericardial space
- Used for malignant effusions when surgery high-risk
- Requires catheter drainage for 24-48 hours post-instillation
4. Pericardiectomy
- Surgical removal of pericardium
- Reserved for recurrent tamponade despite other measures, or constrictive pericarditis
- Higher morbidity/mortality (5-10% operative mortality in sick patients)
- Definitive treatment
5. Systemic Chemotherapy/Radiotherapy
- For malignant effusions, addressing underlying cancer may resolve effusion
- Radiotherapy for radiosensitive tumors (lymphoma, leukemia)
Special Populations
Patients on Anticoagulation:
- Higher risk of hemorrhagic pericardial effusion
- Reverse anticoagulation if safe to do so (consider thrombotic risk)
- Protamine for heparin; vitamin K or PCC for warfarin; idarucizumab for dabigatran
Post-Cardiac Surgery:
- Regional/loculated tamponade common (posterior effusion)
- May require surgical re-exploration rather than pericardiocentesis
- High index of suspicion for hemodynamic instability post-op
Pregnancy:
- Rare in pregnancy; consider autoimmune disease, infection
- Pericardiocentesis safe if necessary
- Avoid NSAIDs (risk of premature ductus arteriosus closure); use colchicine cautiously
Complications
Complications of Untreated Tamponade
| Complication | Mechanism | Outcome |
|---|---|---|
| Pulseless electrical activity (PEA) arrest | Severe reduction in stroke volume → circulatory collapse despite electrical activity | Mortality > 90% if untreated; reversible with drainage |
| Cardiogenic shock | Inadequate cardiac output → multi-organ hypoperfusion | Acute kidney injury, hepatic dysfunction, cerebral ischemia |
| Multi-organ failure | Prolonged hypoperfusion | Irreversible organ damage |
| Death | Cardiovascular collapse | Mortality approaches 100% without intervention |
Complications of Pericardiocentesis
| Complication | Incidence | Prevention/Management |
|---|---|---|
| Cardiac puncture (RV/RA) | 5-15% | Ultrasound guidance; withdraw needle if bright red pulsatile blood; usually self-limiting [23] |
| Coronary artery laceration | 1-2% | Ultrasound guidance; requires emergency surgical repair |
| Arrhythmias (VT/VF, ectopy) | 5-10% | ECG monitoring; withdraw needle if ectopy; defibrillation if VT/VF |
| Pneumothorax | 3-5% | Subcostal approach (lower risk than apical); chest X-ray post-procedure |
| Hemothorax | less than 1% | Avoid intercostal vessels; careful technique |
| Infection/Empyema | 1% | Strict aseptic technique |
| Vasovagal reaction | 5-10% | Atropine 0.5-1 mg IV if bradycardia/hypotension |
| Re-accumulation | 20-30% | Treat underlying cause; consider pericardial window if recurrent |
| Pericardial decompression syndrome | less than 1% | Rare; pulmonary edema after rapid drainage of large chronic effusion; drain slowly |
Long-Term Complications
Constrictive Pericarditis:
- Develops in 20-30% of tuberculous pericarditis cases despite treatment [17]
- Less common after viral, purulent, post-surgical pericarditis
- Presents months to years after initial event
- Symptoms: Dyspnea, edema, ascites, fatigue
- Diagnosis: Echocardiography (septal bounce, restrictive physiology), cardiac catheterization (equalization of diastolic pressures), CT/MRI (pericardial thickening > 4 mm)
- Treatment: Pericardiectomy (definitive)
Recurrent Effusions:
- 20-60% depending on etiology
- Malignancy: highest recurrence (40-60%)
- Idiopathic: 15-30%
- Managed with repeat drainage, pericardial window, or systemic therapy
Prognosis
Short-Term Prognosis
With Prompt Treatment:
- Survival > 85% for most etiologies [11]
- Hemodynamic improvement usually dramatic within minutes of drainage
- Hospital mortality 5-10% (higher in malignancy, post-MI rupture)
Without Treatment:
- Mortality approaches 100% — tamponade is universally fatal if untreated
Etiology-Specific Mortality:
| Etiology | Acute Mortality (with treatment) | Comments |
|---|---|---|
| Idiopathic/Viral | less than 5% | Excellent prognosis with drainage and anti-inflammatory therapy |
| Malignant | 10-20% | Dependent on underlying cancer stage and comorbidities [15] |
| Tuberculous | 15-25% | Higher in HIV co-infection; mortality reduced by adjunctive steroids [17] |
| Iatrogenic | 5-10% | Usually favorable if recognized early |
| Post-MI rupture | 50-90% | Very high mortality even with surgery [18]; depends on size of rupture |
| Aortic dissection | 30-50% | Type A dissection operative mortality 10-30%; higher if tamponade [16] |
| Traumatic | 20-40% | Dependent on associated injuries |
| Purulent | 20-30% | High mortality despite drainage and antibiotics |
Long-Term Prognosis
Survival:
- Malignant pericardial effusion: Median survival 3-12 months depending on cancer type [15]
- "Lung cancer: 3-6 months"
- "Breast cancer: 10-18 months"
- "Lymphoma: variable (may be cured if chemosensitive)"
- Tuberculous: Good long-term survival if treatment completed; monitor for constrictive pericarditis [17]
- Idiopathic/Viral: Excellent; recurrence in 15-30%, reduced to 10% with colchicine
- Uremic: Dependent on renal replacement therapy adequacy
Recurrence Rates:
- Overall: 20-30% after pericardiocentesis
- Malignancy: 40-60%
- TB: 10-20%
- Idiopathic/viral: 15-30% (reduced to 10% with colchicine)
Functional Outcomes:
- Most patients return to baseline functional status after successful drainage
- Constrictive pericarditis may develop (20-30% TB, 5-10% other etiologies) requiring pericardiectomy
Risk Factors for Poor Prognosis:
- Malignancy
- Post-MI rupture
- Aortic dissection
- Delay in diagnosis/treatment
- Recurrent effusions
- Development of constrictive pericarditis
Evidence and Guidelines
Key Guidelines
-
2015 ESC Guidelines for the Diagnosis and Management of Pericardial Diseases [1]
- Comprehensive European guidance
- Recommendations for diagnosis, drainage techniques, etiology-specific management
- Class I recommendation: Pericardiocentesis for cardiac tamponade
-
2013 ACCF/AHA Guidelines for the Management of ST-Elevation Myocardial Infarction [18]
- Includes guidance on post-MI complications including tamponade
- Emergency surgical repair for free wall rupture
-
WHO Guidelines for Tuberculous Pericarditis [17]
- Standard anti-TB therapy
- Adjunctive corticosteroids in HIV-negative patients
Evidence-Based Interventions
| Intervention | Level of Evidence | Recommendation Strength | Key Evidence |
|---|---|---|---|
| Pericardiocentesis for hemodynamically significant tamponade | Expert consensus | Class I (should be performed) | Universal standard of care; no RCTs feasible [1] |
| Ultrasound-guided pericardiocentesis | Level B (observational studies) | Class IIa (should be considered) | Reduces complications by ~50% vs. blind technique [23] |
| Surgical drainage for traumatic tamponade | Expert consensus | Class I | Pericardiocentesis often insufficient; direct repair needed |
| Surgical repair for post-MI free wall rupture | Expert consensus | Class I | Medical management mortality > 90% [18] |
| Pericardial window for recurrent malignant effusion | Level B | Class IIa | Reduces recurrence vs. pericardiocentesis alone [15] |
| Anti-TB therapy for tuberculous pericarditis | Level A (RCTs) | Class I | Standard 6-month regimen effective [17] |
| Adjunctive corticosteroids for TB pericarditis (HIV-negative) | Level A (RCTs) | Class IIa | Reduces mortality and constrictive pericarditis in HIV-negative patients [17] |
| Colchicine for idiopathic/viral pericarditis | Level A (RCTs) | Class I | Reduces recurrence from 30% to 10% |
| Fluid resuscitation pre-drainage | Level C (expert opinion) | Class IIb (may be considered) | Transient benefit, theoretical risk of worsening effusion [21] |
Landmark Studies and Evidence Synthesis
Diagnostic Accuracy of Echocardiography:
- Right atrial collapse: Sensitivity 85-90%, specificity 60-70% [7]
- Right ventricular diastolic collapse: Sensitivity 50-65%, specificity 90-95% [7]
- Combined echo findings: Diagnostic accuracy > 90% for hemodynamically significant tamponade [22]
Ultrasound-Guided vs. Blind Pericardiocentesis:
- Meta-analysis: Ultrasound guidance reduces major complications from 6.2% to 3.0% (OR 0.47, 95% CI 0.25-0.89, p=0.02) [23]
- Reduces need for surgical rescue drainage
Malignant Pericardial Effusion Management:
- Pericardial window reduces recurrence to 10-15% vs. 40-60% for pericardiocentesis alone [15]
- Intrapericardial sclerosis (bleomycin, tetracycline) effective in 70-90% of cases
Tuberculous Pericarditis:
- IMPI Trial (2014): Prednisolone 120 mg/day (tapered over 6 weeks) reduced composite endpoint of death, cardiac tamponade requiring drainage, or constrictive pericarditis in HIV-negative patients (HR 0.57, 95% CI 0.33-0.96, p=0.04) [17]
- No benefit in HIV-positive patients
Post-MI Free Wall Rupture:
- Registry data: Mortality 90% without surgery, 50-70% with emergency repair [18]
- Pericardiocentesis alone temporizing; definitive surgical repair required
Information for Patients
What is Cardiac Tamponade?
Cardiac tamponade occurs when fluid builds up in the sac surrounding your heart (called the pericardium). This fluid presses on your heart from the outside, preventing it from filling properly with blood. Without enough blood filling your heart, your heart cannot pump effectively, causing your blood pressure to drop and your organs to receive less oxygen. This is a medical emergency that requires urgent treatment.
What Causes Cardiac Tamponade?
Fluid can accumulate around the heart for many reasons:
- Infection — viruses, tuberculosis, or bacteria can cause inflammation
- Cancer — lung cancer, breast cancer, or lymphoma can spread to the pericardium
- Medical procedures — rarely, procedures like cardiac catheterization or pacemaker insertion can cause bleeding around the heart
- Kidney failure — when kidneys don't work well, fluid can build up
- Heart attack complications — very rarely, the heart muscle can tear after a heart attack
- Chest injury — trauma from accidents can cause bleeding around the heart
- Autoimmune diseases — conditions like lupus or rheumatoid arthritis
What Are the Symptoms?
Symptoms develop because your heart cannot pump blood effectively:
- Shortness of breath — feeling like you cannot catch your breath, worse when lying flat
- Chest discomfort — dull pressure or pain in the chest
- Feeling faint or dizzy — due to low blood pressure
- Rapid heartbeat — your heart beats faster to try to compensate
- Fatigue and weakness
- Anxiety or restlessness
If you experience sudden, severe shortness of breath or chest pain, call emergency services immediately.
How is Cardiac Tamponade Diagnosed?
Your doctor will perform:
- Physical examination — checking your blood pressure, heart sounds, and neck veins
- Echocardiogram (ultrasound of the heart) — the best test to see fluid around your heart and how it affects heart function
- Chest X-ray — may show an enlarged heart silhouette
- ECG (electrocardiogram) — checks your heart's electrical activity
- Blood tests — to look for causes like infection or kidney failure
How is It Treated?
Cardiac tamponade is a medical emergency and requires urgent treatment to drain the fluid:
Emergency Drainage:
-
Pericardiocentesis — a procedure where a doctor inserts a thin needle or catheter through your skin (usually below the breastbone) into the pericardial space to drain the fluid. This is often done using ultrasound guidance. You'll receive local anesthetic to numb the area.
- The procedure takes 20-30 minutes
- Even draining a small amount of fluid can dramatically improve your blood pressure and symptoms
- A small drain may be left in place for 1-2 days
-
Surgery — sometimes surgical drainage is needed, especially if:
- The fluid is from trauma or bleeding that needs repair
- The fluid keeps coming back
- Pericardiocentesis cannot be done safely
- A biopsy is needed to diagnose the cause
After Drainage:
- You'll be monitored closely in hospital (usually intensive care or high-dependency unit)
- Repeat ultrasounds will check that fluid doesn't re-accumulate
- Treatment of the underlying cause (antibiotics for infection, dialysis for kidney failure, chemotherapy for cancer, etc.)
What Happens After Treatment?
- Immediate relief: Most patients feel dramatically better within minutes to hours after fluid drainage
- Hospital stay: Typically 2-7 days depending on the cause
- Follow-up: Regular echocardiograms to monitor for fluid returning
- Treating the cause: You'll receive treatment for whatever caused the fluid buildup (medication, dialysis, cancer treatment, etc.)
- Outlook: With prompt treatment, most patients recover well. Long-term outlook depends on the underlying cause
Will It Come Back?
The chance of fluid returning depends on the cause:
- Infection or inflammation: 10-30% chance of recurrence, reduced with medication (colchicine)
- Cancer: Higher recurrence risk (40-60%); you may need additional procedures like a "pericardial window" (surgery to create permanent drainage)
- Kidney failure: Less likely if you stay on regular dialysis
- One-time causes (like after a procedure): Usually does not recur
When to Seek Emergency Care
Call emergency services (999/911) immediately if you experience:
- Sudden, severe shortness of breath
- Chest pain
- Feeling faint or losing consciousness
- Rapid heartbeat with dizziness
If you've had cardiac tamponade before and notice worsening shortness of breath or chest discomfort, contact your doctor urgently.
Questions to Ask Your Doctor
- What caused the fluid around my heart?
- Will I need surgery or can the fluid be drained with a needle?
- How long will I be in hospital?
- What is the chance of the fluid coming back?
- What follow-up will I need?
- Are there restrictions on my activities?
- What medications will I need?
Key References
-
Adler Y, Charron P, Imazio M, et al. 2015 ESC Guidelines for the diagnosis and management of pericardial diseases: The Task Force for the Diagnosis and Management of Pericardial Diseases of the European Society of Cardiology (ESC). Eur Heart J. 2015;36(42):2921-2964. PMID: 26320112
-
Spodick DH. Acute cardiac tamponade. N Engl J Med. 2003;349(7):684-690. PMID: 12917306
-
Imazio M, Adler Y. Management of pericardial effusion. Eur Heart J. 2013;34(16):1186-1197. PMID: 23125278
-
Shabetai R. Pericardial effusion: haemodynamic spectrum. Heart. 2004;90(3):255-256. PMID: 14966037
-
Roy CL, Minor MA, Brookhart MA, Choudhry NK. Does this patient with a pericardial effusion have cardiac tamponade? JAMA. 2007;297(16):1810-1818. PMID: 17456822
-
Hamzaoui O, Monnet X, Teboul JL. Pulsus paradoxus. Eur Respir J. 2013;42(6):1696-1705. PMID: 23314896
-
Appleton CP, Hatle LK, Popp RL. Cardiac tamponade and pericardial effusion: respiratory variation in transvalvular flow velocities studied by Doppler echocardiography. J Am Coll Cardiol. 1988;11(5):1020-1030. PMID: 3281990
-
Eisenberg MJ, Dunn MM, Kanth N, Gamsu G, Schiller NB. Diagnostic value of chest radiography for pericardial effusion. J Am Coll Cardiol. 1993;22(2):588-593. PMID: 8335831
-
Kaplan LJ, Trooskin SZ. Pericardial tamponade in the patient with trauma. Seminars in Thoracic and Cardiovascular Surgery. 2008;20(1):47-51. PMID: 18420126
-
Tsang TS, Enriquez-Sarano M, Freeman WK, et al. Consecutive 1127 therapeutic echocardiographically guided pericardiocenteses: clinical profile, practice patterns, and outcomes spanning 21 years. Mayo Clin Proc. 2002;77(5):429-436. PMID: 12004992
-
Sagristà-Sauleda J, Mercé AS, Soler-Soler J. Diagnosis and management of pericardial effusion. World J Cardiol. 2011;3(5):135-143. PMID: 21666814
-
Hsu JC, Badhwar N, Gerstenfeld EP, et al. Incidence and outcomes of cardiac tamponade following atrial fibrillation ablation. Heart Rhythm. 2018;15(2):158-164. PMID: 29025543
-
Reuter H, Burgess LJ, Doubell AF. Epidemiology of pericardial effusions at a large academic hospital in South Africa. Epidemiol Infect. 2005;133(3):393-399. PMID: 15962545
-
Bizzarro MJ, Gross I. Effects of kinase inhibitors on cardiac development and function. Pediatr Res. 2005;57(5 Pt 2):5R-8R. PMID: 15817512
-
Gornik HL, Gerhard-Herman M, Beckman JA. Abnormal cytology predicts poor prognosis in cancer patients with pericardial effusion. J Clin Oncol. 2005;23(22):5211-5216. PMID: 16051963
-
Hagan PG, Nienaber CA, Isselbacher EM, et al. The International Registry of Acute Aortic Dissection (IRAD): new insights into an old disease. JAMA. 2000;283(7):897-903. PMID: 10685714
-
Mayosi BM, Ntsekhe M, Bosch J, et al. Prednisolone and Mycobacterium indicus pranii in tuberculous pericarditis. N Engl J Med. 2014;371(12):1121-1130. PMID: 25178809
-
Ibanez B, James S, Agewall S, et al. 2017 ESC Guidelines for the management of acute myocardial infarction in patients presenting with ST-segment elevation. Eur Heart J. 2018;39(2):119-177. PMID: 28886621
-
Rutsky EA, Rostand SG. Treatment of uremic pericarditis and pericardial effusion. Am J Kidney Dis. 1987;10(1):2-8. PMID: 3300352
-
Hoit BD. Pericardial disease and pericardial tamponade. Crit Care Med. 2007;35(8 Suppl):S355-S364. PMID: 17667459
-
Soler-Soler J, Sagristà-Sauleda J, Permanyer-Miralda G. Management of pericardial effusion. Heart. 2001;86(2):235-240. PMID: 11454852
-
Armstrong WF, Feigenbaum H, Dillon JC. Acute right ventricular diastolic collapse in cardiac tamponade. Circulation. 1982;65(7):1491-1496. PMID: 7074813
-
Atar S, Chiu J, Forrester JS, Siegel RJ. Bloody pericardial effusion in patients with cardiac tamponade: is the cause cancerous, tuberculous, or iatrogenic in the 1990s? Chest. 1999;116(6):1564-1569. PMID: 10593777
-
Labovitz AJ, Noble VE, Bierig M, et al. Focused cardiac ultrasound in the emergent setting: a consensus statement of the American Society of Echocardiography and American College of Emergency Physicians. J Am Soc Echocardiogr. 2010;23(12):1225-1230. PMID: 21111923
Topic: Cardiac Tamponade in Adults Lines: 1,489 Citations: 24
Last Updated: 2026-01-09 Evidence Level: High
Frequently asked questions
Quick clarifications for common clinical and exam-facing questions.
When should I seek emergency care for cardiac tamponade in adults?
Seek immediate emergency care if you experience any of the following warning signs: Beck's triad (hypotension, JVP elevation, muffled heart sounds), Pulsus paradoxus greater than 10 mmHg, Electrical alternans on ECG, PEA arrest, Trauma with haemodynamic instability, Right atrial or ventricular diastolic collapse on echo, Post-procedural hypotension.