Cauda Equina Syndrome
The most common cause is massive central lumbar disc herniation (typically at L4/5 or L5/S1), accounting for approximately 45% of cases, though tumors, trauma, epidural abscess, and hemorrhage are important...
What matters first
The most common cause is massive central lumbar disc herniation (typically at L4/5 or L5/S1), accounting for approximately 45% of cases, though tumors, trauma, epidural abscess, and hemorrhage are important...
Bilateral Sciatica -> Central disc herniation
6 Jan 2026
Generated educational material; verify before clinical use.
Visible references section
See the concept before reading it
Study the key anatomy, imaging, and decision pathways as full teaching plates.
Clinical board
A visual summary of the highest-yield teaching signals on this page.
Urgent signals
Safety-critical features pulled from the topic metadata.
- Bilateral Sciatica -> Central disc herniation
- Saddle Anaesthesia -> Loss of S3-S5 sensation
- Bladder Retention -> Painless overflow incontinence
- Anal Tone Loss -> Inability to contract sphincter
Linked comparisons
Differentials and adjacent topics worth opening next.
- Conus Medullaris Syndrome
- Acute Spinal Cord Compression
Content status and exam context
This page is AI-generated educational content. It may contain errors or omissions and is not a substitute for current guidelines, local protocols, senior clinical judgement, or professional medical advice.
MedVellum does not claim an individual clinician reviewer, board certification, or professional credential for this page unless a future version names a real, verifiable reviewer.
Clinical explanation and evidence
Cauda Equina Syndrome
1. Clinical Overview
Summary
Cauda Equina Syndrome (CES) is a rare but devastating neurosurgical emergency caused by compression of the cauda equina nerve roots (L2-S5) within the lumbar spinal canal below the termination of the spinal cord. The condition represents one of the few absolute emergencies in spinal surgery, with profound implications for bladder, bowel, sexual, and lower limb function. [1,2]
The most common cause is massive central lumbar disc herniation (typically at L4/5 or L5/S1), accounting for approximately 45% of cases, though tumors, trauma, epidural abscess, and hemorrhage are important differential diagnoses. [3] If untreated, CES leads to permanent paralysis, neurogenic bladder requiring lifelong catheterization, fecal incontinence, and sexual dysfunction. [4]
The cornerstone of management is early recognition through clinical assessment and bladder scanning, emergency MRI imaging to confirm compression, and urgent surgical decompression. The critical time window for optimal outcomes is generally quoted as less than 48 hours from onset of sphincter disturbance, though earlier intervention (ideally within 24 hours) is associated with superior functional recovery. [5,6] The mantra "Time is Spine" underscores the neurological urgency analogous to stroke or myocardial infarction.
Key Facts
- Definition: Compression of multiple lumbosacral nerve roots (L2-S5) in the cauda equina below the conus medullaris, resulting in polyradiculopathy affecting motor, sensory, and autonomic function.
- Incidence: Approximately 1-3 per 100,000 population per year; 0.04% of patients presenting with low back pain; 1-2% of lumbar disc herniations. [7,8]
- Most Sensitive Clinical Sign: Urinary retention with post-void residual volume > 200ml (sensitivity 90%). [9]
- Most Specific Clinical Sign: Loss of anal sphincter tone on digital rectal examination (specificity 80%). [10]
- Critical Window: Surgery within 48 hours associated with better outcomes; within 24 hours optimal. [5,6]
- Litigation Risk: CES represents the highest medico-legal risk in spinal surgery, accounting for up to 40% of neurosurgical litigation claims. [11]
Clinical Pearls
"Painless is Worse than Painful": A patient with excruciating sciatica typically has nerve root irritation without established compression. A patient whose severe pain suddenly disappears and is replaced by numbness, particularly with urinary incontinence, may have progressed from nerve irritation to complete compression with ischemia—a neurosurgical emergency.
"Bilateral Sciatica is Central Until Proven Otherwise": Standard posterolateral disc herniations cause unilateral radicular pain. Bilateral leg symptoms suggest a large central disc herniation occupying the entire spinal canal, threatening all nerve roots in the cauda equina.
"Don't Trust the Rectal Exam Alone": Anal tone assessment is subjective and examiner-dependent (inter-rater reliability κ=0.4-0.6). Objective bladder scanning showing > 300ml post-void residual in a patient unaware of bladder fullness is far more reliable. [12]
"The White Flag of Painless Incontinence": Painless urinary incontinence (CES-R) indicates established bladder denervation. This is a late sign; we must identify patients earlier at the CES-I (incomplete) stage when they have retention or hesitancy but preserved awareness of bladder function.
"Document, Document, Document": Precise documentation of symptom onset timing ("When did you first notice difficulty passing urine?") is medicolegally critical. CES litigation often hinges on whether delay was clinician-related or disease-related.
2. Epidemiology
Demographics
- Annual Incidence: 1.5-3.4 cases per 100,000 population. [7,13]
- Prevalence in Low Back Pain: 0.04% (4 in 10,000) of emergency department presentations with back pain. [8]
- Prevalence in Disc Herniation: 1-2% of symptomatic lumbar disc herniations progress to CES. [14]
- Age Distribution: Bimodal
- Primary peak: 30-50 years (degenerative disc herniation)
- Secondary peak: 60-70 years (malignancy, spinal stenosis)
- Sex Distribution: Slight male predominance (1.2:1 M:F ratio). [15]
Risk Factors
- Anatomical: Congenital narrow spinal canal (developmental stenosis); trefoil-shaped canal morphology increases risk of CES from disc herniation. [16]
- Degenerative: Multilevel lumbar stenosis; large disc herniation (> 50% canal compromise on imaging). [17]
- Iatrogenic: Epidural steroid injection (rare but reported); lumbar surgery (post-operative hematoma 0.1-0.2%). [18]
- Oncological: History of cancer (spinal metastases); multiple myeloma.
- Trauma: High-energy lumbar burst fractures; fracture-dislocation.
- Infectious: Spinal tuberculosis (Pott's disease); pyogenic epidural abscess.
- Vascular: Anticoagulation therapy (epidural hematoma); bleeding diathesis.
Geographic and Healthcare Variations
- Presentation Delay: Median time from symptom onset to hospital presentation ranges from 24-72 hours across healthcare systems, influenced by patient awareness and primary care access. [19]
- Litigation Variation: UK and Australia have particularly high CES litigation rates (up to 40% of spinal surgery claims), driving protocol development and guideline adherence. [11,20]
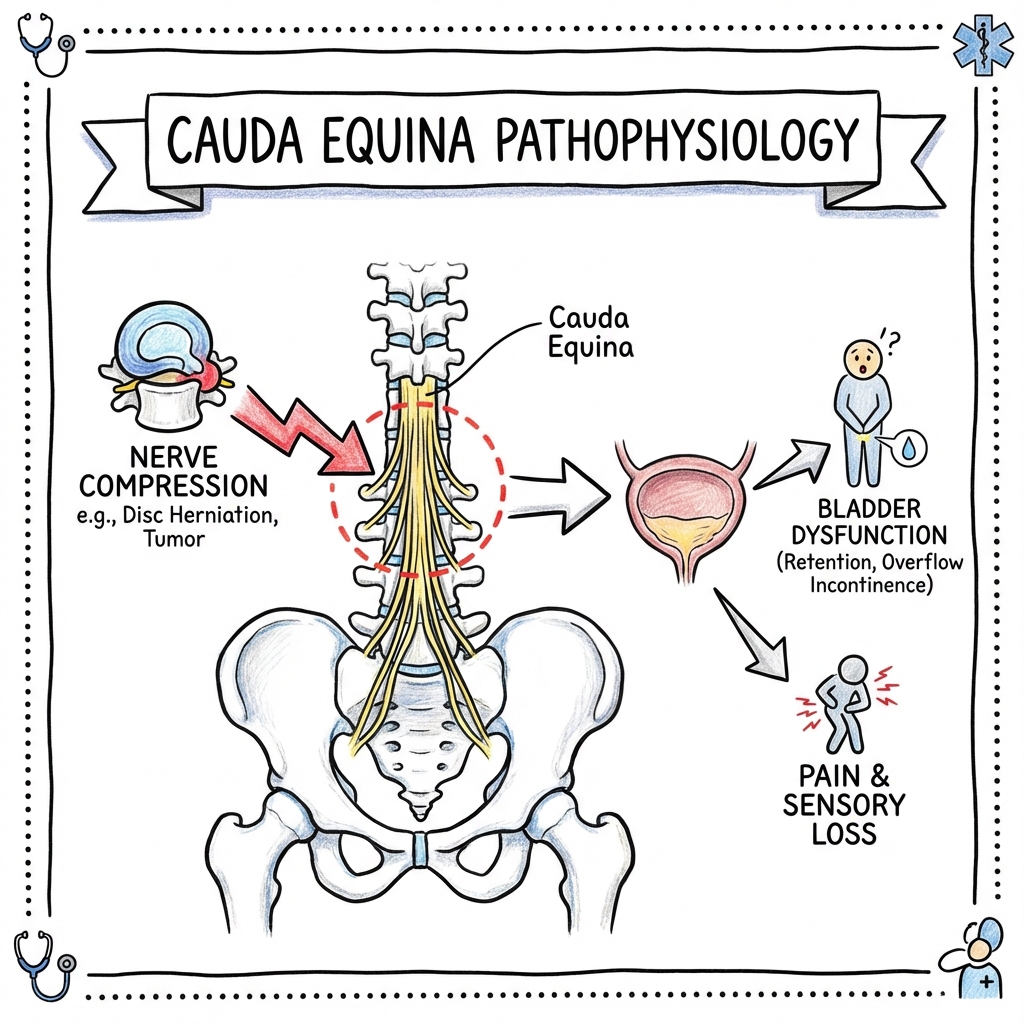
3. Pathophysiology
Anatomical Foundation
Spinal Cord Termination and the Cauda Equina
- The conus medullaris (terminal spinal cord) typically ends at the L1/L2 vertebral level in adults (range T12-L3). [21]
- Below the conus, the cauda equina ("horse's tail") comprises the descending nerve roots from L2-S5 and the coccygeal nerve, floating freely in cerebrospinal fluid within the thecal sac.
- The lumbar thecal sac normally contains approximately 15-20 nerve roots, providing some redundancy; compression must affect multiple roots to produce the CES syndrome. [22]
Functional Anatomy of Affected Nerve Roots
- L2-L4 Roots: Motor supply to hip flexors (L2-3), knee extensors (L3-4); sensory to anterior thigh and medial leg; contribute to femoral and obturator nerves.
- L5 Root: Motor supply to ankle/great toe dorsiflexion (tibialis anterior, extensor hallucis longus); sensory to lateral leg and dorsum of foot.
- S1 Root: Motor supply to ankle plantarflexion (gastrocnemius); sensory to posterior leg and lateral foot.
- S2-S4 Roots (Pelvic Splanchnic Nerves): Parasympathetic innervation of pelvic viscera
- Bladder: Detrusor contraction (S2-4); internal urethral sphincter relaxation (S2-4)
- Rectum: Rectal contraction and defecation reflex (S2-4)
- Sexual Function: Penile/clitoral erection (parasympathetic S2-4); ejaculation/orgasm involves somatic pudendal nerve (S2-4)
- S3-S5/Coccygeal Roots: Sensory innervation to perineum ("saddle area"); motor supply to external anal sphincter (S2-4 via pudendal nerve).
Mechanisms of Neural Injury
Mechanical Compression
- Direct Pressure: Mass effect from disc, tumor, bone, or hematoma causes immediate mechanical deformation of nerve roots against the canal walls.
- Critical Threshold: Experimental studies suggest > 50% canal compromise correlates with onset of symptoms; > 75% compromise associated with severe/complete syndromes. [23]
- Vulnerability Factors: Nerve roots lack the protective epineurium of peripheral nerves; CSF pulsation dynamics are disrupted in compression, reducing nutrient delivery.
Vascular Compromise
- Radicular Arteries: Compression impedes blood flow through radicular arteries supplying nerve roots.
- Ischemic Threshold: Nerve roots tolerate ischemia for approximately 4-6 hours before irreversible axonal damage occurs. [24]
- Venous Congestion: Obstruction of venous drainage causes edema, further increasing intraneural pressure and compounding ischemia.
- Microvascular Thrombosis: Prolonged compression may trigger microvascular thrombosis within nerve roots, explaining irreversible damage despite technically successful decompression.
Inflammatory Cascade
- Cytokine Release: Mechanical injury triggers release of inflammatory mediators (TNF-α, IL-1β, IL-6) from compressed nerve tissue and nucleus pulposus material. [25]
- Secondary Injury: Inflammation causes secondary edema, expanding the zone of neural damage beyond the primary compression site.
- Chronic Changes: Long-standing compression leads to fibrosis, demyelination, and Wallerian degeneration of axons.
Natural History and Recovery Potential
Phases of Injury
- Neurapraxia (Reversible): Early compression causes conduction block without structural damage; rapid recovery if decompressed within hours.
- Axonotmesis (Partially Reversible): Prolonged compression (> 24-48h) damages axons but preserves nerve root sheaths; recovery via axonal regeneration (slow, 1mm/day, incomplete).
- Neurotmesis (Irreversible): Severe/prolonged compression disrupts entire nerve root architecture; no recovery potential.
Time-Dependent Outcomes
- less than 24 hours: 70-80% chance of bladder recovery. [5]
- 24-48 hours: 50-60% chance of bladder recovery. [6]
- > 48 hours: 30-40% chance of bladder recovery. [26]
- > 7 days: less than 20% recovery; often considered salvage surgery to prevent further deterioration. [27]
4. Etiology and Causes
Lumbar Disc Herniation (45-50%)
- Mechanism: Massive central extrusion or sequestration of nucleus pulposus through posterior longitudinal ligament.
- Typical Levels: L4/5 (most common) and L5/S1 (thecal sac widest at these levels, but acute large herniations can still compress).
- Morphology: Central or central-lateral herniation > 50% canal diameter; sequestered free fragments can migrate caudally.
- Presentation: Often acute-on-chronic; history of chronic back pain/sciatica followed by sudden deterioration.
Neoplasms (20-30%)
- Metastatic Disease: Breast, lung, prostate, renal cell, thyroid; typically vertebral body metastasis with posterior extension into canal.
- Primary Spine Tumors: Chordoma, chondrosarcoma, osteosarcoma (rare).
- Intradural Tumors: Schwannoma, meningioma, ependymoma (conus/cauda origin).
- Hematological: Multiple myeloma; lymphoma with epidural extension.
- Presentation: Insidious onset; progressive over weeks; night pain; unresponsive to analgesia; weight loss; known cancer history.
Trauma (10-15%)
- Burst Fractures: High-energy axial loading (fall from height, motor vehicle accident) causing vertebral body comminution with retropulsed bone fragments.
- Fracture-Dislocation: Flexion-distraction injuries (Chance fracture); shear injuries.
- Sacral Fractures: High-energy pelvic trauma with displaced sacral fractures compressing sacral roots.
- Penetrating Trauma: Gunshot wounds; stab injuries (rare in developed countries).
Infection (5-10%)
- Epidural Abscess: Pyogenic (Staph aureus 60%, Strep, E. coli) or tuberculous (Pott's disease).
- Risk Factors: IV drug use; diabetes; immunosuppression; spinal instrumentation; distant infection source.
- Presentation: Fever, elevated inflammatory markers (CRP, ESR), progressive back pain over days-weeks, neurological deficit is late sign.
- Imaging: MRI shows rim-enhancing epidural collection compressing thecal sac.
Hemorrhage (3-5%)
- Epidural Hematoma: Anticoagulation (warfarin, DOACs); post-operative; post-epidural anesthesia/steroid injection; coagulopathy; vascular malformation rupture.
- Presentation: Acute onset; severe back pain; rapid neurological deterioration (hours).
- Imaging: MRI shows hyperintense (T1/T2) epidural collection.
Spinal Stenosis (Rare, less than 5%)
- Mechanism: Severe degenerative central canal stenosis (ligamentum flavum hypertrophy, facet hypertrophy, disc bulge) creating critical stenosis; minor additional insult (fall, cough) precipitates acute-on-chronic CES.
- Presentation: Elderly; chronic neurogenic claudication history; sudden deterioration.
Iatrogenic (1-2%)
- Post-Surgical: Hematoma (0.1-0.2% lumbar surgery); graft dislodgement; retained surgical material.
- Post-Injection: Epidural steroid injection complication (direct needle trauma, hematoma, abscess).
5. Classification Systems
British Association of Spine Surgeons (BASS) Classification
The BASS classification, introduced in 2009 and refined in 2018, is the most widely used system in the UK and internationally, emphasizing bladder function as the key discriminator. [28]
CES-S (Suspected)
- Definition: Bilateral radicular leg pain (sciatica) with or without sensory changes, but no sphincter dysfunction.
- Bladder Function: Normal sensation of fullness, normal voiding.
- Prognosis: Majority do not progress to true CES; represents high clinical suspicion requiring urgent MRI.
- Management: Urgent MRI (within 24 hours); close observation.
CES-I (Incomplete)
- Definition: Altered urinary sensation or difficulty voiding, but bladder control is preserved.
- Bladder Symptoms: Reduced sensation of fullness, hesitancy, poor stream, incomplete emptying, BUT patient is still aware of bladder status and has not experienced painless incontinence.
- Post-Void Residual: Typically 200-500ml.
- Prognosis: BEST PROGNOSIS with urgent surgery; this is the critical window for preserving function.
- Management: Emergency MRI and surgery (same day/overnight).
CES-R (Retention with Overflow)
- Definition: Established painless urinary retention with overflow incontinence.
- Bladder Function: Bladder is mechanically paralyzed; patient unaware of bladder fullness; wetting occurs without warning.
- Post-Void Residual: Typically > 500ml.
- Prognosis: POOR PROGNOSIS; permanent bladder dysfunction likely even with decompression.
- Management: Urgent surgery (prevents further deterioration); catheterization; counseling regarding likely permanent deficits.
Alternative Classification: Gitelman et al. (2008)
- Early CES: Symptoms less than 24 hours; urinary difficulties but no established retention.
- Intermediate CES: Symptoms 24-48 hours.
- Late CES: Symptoms > 48 hours; established neurological deficit.
- Use: Primarily research/retrospective studies correlating timing to outcome.
6. Clinical Presentation
The Five Cardinal Red Flags
1. Bilateral Sciatica
- Definition: Radicular pain radiating below the knee in BOTH legs simultaneously.
- Mechanism: Large central disc herniation compressing bilateral L5 and/or S1 roots.
- Sensitivity: 70-80% for CES. [29]
- Clinical Context: Unilateral sciatica is common (90% of disc herniations); bilateral sciatica should immediately raise CES suspicion.
- Caveat: Alternating unilateral sciatica is NOT the same as true bilateral simultaneous symptoms.
2. Saddle Anaesthesia/Hypoesthesia
- Definition: Reduced or absent sensation in the perineum, genitals, anus, and inner thighs—areas that would contact a saddle.
- Nerve Roots: S3, S4, S5 dermatomes.
- Examination: Test with light touch, pinprick, or cold sensation; ask "Does it feel different when you wipe yourself after using the toilet?"
- Sensitivity: 60-75% (patients may not notice or report unless specifically asked). [30]
- Specificity: 90-95% when present (highly specific for sacral root dysfunction).
3. Bladder Dysfunction
- Early Symptoms (CES-I):
- Reduced sensation of bladder fullness ("I can't tell when my bladder is full")
- Difficulty initiating urination (hesitancy)
- Weak urinary stream
- Sensation of incomplete emptying
- Loss of ability to "hold on" (reduced urge)
- Late Symptoms (CES-R):
- Painless urinary retention (bladder distended but no discomfort)
- Overflow incontinence (wetting without awareness)
- Complete inability to void
- Mechanism: Parasympathetic denervation (S2-4) abolishes detrusor contraction and internal sphincter coordination.
- Objective Measurement: Post-void residual (PVR) bladder scan
- less than 50 ml: Normal
- 50-200 ml: Borderline (age/sex dependent)
-
200 ml: Abnormal retention
-
500 ml: Definite retention, highly suspicious for CES in clinical context
4. Bowel Dysfunction
- Symptoms:
- Fecal incontinence (inability to prevent passage of stool/flatus)
- Constipation (loss of defecation reflex)
- Loss of sensation of rectal fullness
- Inability to feel stool passage
- Sensitivity: Lower than bladder symptoms (~40-50%); often presents later. [10]
- Mechanism: Parasympathetic (S2-4) and somatic pudendal nerve (S2-4) disruption affecting external anal sphincter and rectal motility.
5. Sexual Dysfunction
- Males: Erectile dysfunction (loss of parasympathetic-mediated erection); ejaculatory dysfunction.
- Females: Loss of vaginal lubrication; loss of genital sensation; anorgasmia.
- Mechanism: S2-4 parasympathetic (erection/lubrication) and pudendal nerve (sensation/orgasm) disruption.
- Clinical Relevance: Often not volunteered by patients in acute setting; important for long-term quality of life and medicolegal documentation.
Motor Signs
- Distribution: Lower motor neuron pattern affecting L2-S1 myotomes.
- Common Findings:
- L4: Knee extension weakness (quadriceps); reduced/absent knee jerk.
- L5: Ankle/toe dorsiflexion weakness (foot drop); tibialis anterior, extensor hallucis longus.
- S1: Ankle plantarflexion weakness; reduced/absent ankle jerk.
- Severity: Variable; may be asymmetric; complete paralysis rare (cauda equina is polyradicular, not cord injury).
- Progression: Progressive motor weakness over hours-days is ominous sign.
Sensory Signs
- Dermatomal Pattern: L4-S1 dermatomal sensory loss in legs (variable pattern depending on roots involved).
- Saddle Distribution: S3-5 perineal/perianal hypoesthesia (see above).
- Testing: Light touch, pinprick, proprioception; compare symmetry.
Reflex Changes
- Knee Jerk (L3-4): Reduced or absent.
- Ankle Jerk (S1): Reduced or absent (often earliest objective sign).
- Plantar Response: Flexor (LMN lesion); extensor Babinski would suggest conus medullaris or cord involvement.
- Bulbocavernosus Reflex: Squeezing glans penis/clitoris or tugging Foley catheter should cause reflex anal sphincter contraction (S2-4 reflex arc); absence suggests sacral root/conus dysfunction. Not routinely tested but relevant in uncertain cases.
Perianal Examination (Digital Rectal Examination - DRE)
- Inspection: Look for soiling, hemorrhoids, fissures (may confound tone assessment).
- Resting Tone: Assess baseline anal sphincter tone; reduced tone suggests S2-4 dysfunction.
- Voluntary Contraction: Ask patient to "squeeze" around examining finger; inability indicates motor dysfunction of external sphincter (pudendal nerve S2-4).
- Perianal Sensation: Test S4-5 dermatomes around anus with light touch/pinprick.
- Limitations: Subjective; inter-observer variability; normal tone does not exclude CES (may have bladder dysfunction with preserved anal tone early). [12]
Atypical Presentations
Painless CES
- Scenario: Tumor, abscess, or gradual stenosis causing slowly progressive compression without acute pain.
- Risk: Delayed presentation; patient attributes bladder symptoms to age/prostate/urological cause.
Unilateral Symptoms Progressing to CES
- Scenario: Initial posterolateral disc herniation causing unilateral sciatica, then acute expansion or migration to central position.
- Importance: Previous "sciatica" does not exclude CES; must re-examine for evolving red flags.
Post-Operative CES
- Scenario: Hematoma formation hours-days after lumbar surgery; initially attributed to post-op pain/anesthesia effects.
- Red Flag: New-onset urinary retention or worsening leg weakness after initial post-op recovery.
7. Investigations
Bedside Assessments
Bladder Scan (Mandatory)
- Indication: ALL patients with suspected CES (back pain + any red flag).
- Technique: Portable ultrasound device placed supraumbically; measures post-void residual (PVR) volume.
- Interpretation:
- less than 50 ml: Normal (CES unlikely based on bladder function).
- 50-200 ml: Borderline; consider age, sex, recent fluid intake; repeat or proceed to MRI based on clinical suspicion.
- 200-500 ml: Abnormal; consistent with CES-I; emergency MRI indicated.
-
500 ml: Definite retention; consistent with CES-R; emergency MRI and catheterization.
- Sensitivity: 90% for detecting urinary retention. [9]
- Advantages: Objective, rapid, non-invasive, reproducible.
- Limitations: Operator-dependent; may overestimate in presence of pelvic masses/ascites; does not assess sensation (patient may have large PVR but still have awareness, which is better prognostically).
Digital Rectal Examination (DRE)
- Indications: Part of neurological examination in suspected CES.
- Documentation: Record resting tone, voluntary contraction, perianal sensation, bulbocavernosus reflex if tested.
- Caveats: Subjective; normal DRE does not exclude CES; abnormal DRE in isolation (without bladder symptoms) has low specificity.
Neurological Examination
- Lower Limb Motor: Test all myotomes L2-S1 bilaterally; document MRC grade (0-5).
- Lower Limb Sensory: Test dermatomes L1-S5; include saddle area (S3-5).
- Reflexes: Knee jerk (L3-4), ankle jerk (S1-2), plantar response.
- Gait: Observe for foot drop, Trendelenburg, antalgic patterns (if patient able to mobilize).
Laboratory Tests
- Routine Bloods: Generally unhelpful for diagnosis of CES but useful for surgical planning and ruling out differential diagnoses.
- FBC: Exclude anemia (malignancy, chronic disease); leukocytosis (infection/abscess).
- CRP/ESR: Elevated in abscess, discitis, malignancy.
- U&E: Baseline renal function (may be affected by chronic retention/obstructive uropathy).
- Coagulation (INR/APTT): If on anticoagulation or bleeding diathesis suspected (epidural hematoma).
- Tumor Markers: PSA (prostate cancer), Ca 19-9 (GI cancer), if malignancy suspected.
Imaging
MRI Lumbar Spine (Gold Standard)
- Indication: ALL patients with clinical suspicion of CES (red flags + abnormal bladder scan).
- Urgency: Emergency (same-day; ideally within 4 hours of presentation). [28]
- Protocol:
- Sequences: Sagittal T1, sagittal T2, axial T2 (at levels of interest).
- Coverage: Entire lumbar spine (L1-S1) and sacrum if sacral fracture suspected.
- Contrast: Gadolinium if tumor, infection, or post-operative complication suspected.
- Key Findings:
- Disc Herniation: Central disc extrusion/sequestration; obliteration of thecal sac CSF signal; nerve roots compressed against canal walls; disc material may migrate cranially or caudally.
- Tumor: Vertebral body destruction; paraspinal soft tissue mass; epidural extension; contrast enhancement.
- Abscess: Rim-enhancing epidural collection; associated discitis/osteomyelitis; paraspinal phlegmon.
- Hematoma: Hyperintense T1/T2 epidural collection (variable signal depending on age of blood).
- Stenosis: Multilevel degenerative changes; ligamentum flavum hypertrophy; facet hypertrophy; trefoil canal shape.
- Quantification: Degree of canal compromise reported as percentage (e.g., "> 75% canal stenosis at L4/5"); axial images most useful.
- Sensitivity/Specificity: 97% sensitivity, 93% specificity for clinically significant compression. [31]
CT Lumbar Spine (Alternative if MRI Unavailable/Contraindicated)
- Indications:
- MRI contraindicated (pacemaker, some implants—though MRI-compatible devices increasingly common).
- MRI not available (resource-limited settings).
- Trauma setting where initial CT already performed.
- Protocol: Thin-slice (1-2mm) axial CT with sagittal/coronal reformats; CT myelography (intrathecal contrast) improves visualization of nerve roots but invasive.
- Advantages: Faster acquisition; better bony detail (fractures).
- Disadvantages: Inferior soft tissue resolution; difficulty visualizing disc vs. nerve root; radiation exposure; less sensitive for early cord/root edema.
- Sensitivity/Specificity: 70-80% (inferior to MRI). [32]
Plain Radiographs (X-ray)
- Role: NOT INDICATED for diagnosis of CES.
- Limitations: Cannot visualize nerve roots, discs, or soft tissues; may show bony changes (collapse, fracture, sclerosis) but does not exclude or confirm compression.
- Exception: May be obtained in trauma protocol (ATLS) before MRI, but should not delay definitive imaging.
Specialized Tests (Rarely Used in Acute Setting)
Urodynamics
- Use: Assessment of chronic bladder dysfunction post-CES; characterizes detrusor areflexia, compliance, sphincter function.
- Timing: NOT in acute diagnosis; used 3-6 months post-decompression to guide bladder management.
Electromyography (EMG) and Nerve Conduction Studies (NCS)
- Use: Differentiate CES (polyradiculopathy) from peripheral neuropathy or myopathy; assess chronic denervation.
- Findings in CES: Fibrillation potentials, positive sharp waves in affected myotomes; reduced recruitment; prolonged F-wave latencies (root level).
- Timing: Acute changes take 2-3 weeks to develop; mainly used for chronic cases or medicolegal assessment.
Somatosensory Evoked Potentials (SSEP)
- Use: Intraoperative monitoring during decompression; assess dorsal column function.
- Limitation: Does not assess autonomic/motor pathways; limited utility in acute diagnosis.
8. Differential Diagnosis
Conus Medullaris Syndrome
- Anatomy: Compression of the terminal spinal cord (conus) at T12-L2 level.
- Distinguishing Features:
- Upper Motor Neuron Signs: Hyperreflexia, extensor plantar (Babinski) response (conus contains cord segments).
- Symmetric Deficits: More symmetric motor/sensory loss (cord lesion affects both sides equally).
- Sudden Onset: Bladder/bowel dysfunction may occur early/suddenly.
- Saddle Sensation: Present but more symmetric and complete.
- Imaging: MRI shows lesion at T12-L1 level compressing conus.
- Clinical Overlap: Mixed conus-cauda syndromes exist (lesion at L1 may affect both).
Acute Lumbar Radiculopathy (Sciatica Without CES)
- Features: Unilateral leg pain; normal bladder/bowel function; no saddle anesthesia.
- Examination: Positive straight leg raise; dermatomal sensory loss; myotomal weakness (single root).
- Management: Conservative initially; imaging if red flags or progressive motor deficit.
Peripheral Polyneuropathy
- Etiology: Diabetes, Guillain-Barré syndrome, chronic inflammatory demyelinating polyneuropathy (CIDP), toxins.
- Distinguishing Features:
- Distribution: Distal, symmetric "glove-and-stocking" sensory loss; starts in feet.
- Reflexes: Globally reduced (not just ankle/knee).
- Sphincters: Usually spared until very advanced disease.
- Chronicity: Gradual onset over months-years (except Guillain-Barré).
- Investigations: EMG/NCS shows peripheral nerve involvement; MRI spine normal.
Guillain-Barré Syndrome (GBS)
- Features: Ascending paralysis; areflexia; respiratory involvement; autonomic dysfunction (may include urinary retention).
- Distinguishing Features:
- History: Preceding viral illness (1-3 weeks); progressive over days.
- Examination: Ascending weakness (legs → arms → face); global areflexia; no sensory level.
- Investigations: LP shows albuminocytologic dissociation (high protein, normal WCC); MRI spine normal; EMG shows demyelination.
Spinal Epidural Abscess
- Features: Fever, back pain, progressive neurological deficit.
- Risk Factors: IVDU, diabetes, immunosuppression, bacteremia, spinal procedures.
- Examination: Fever (not always present); focal spine tenderness; meningism (if concomitant meningitis).
- Investigations: Elevated WCC, CRP, ESR; blood cultures positive (50%); MRI shows rim-enhancing epidural collection ± discitis/osteomyelitis.
- Management: Emergency decompression + IV antibiotics; neurosurgical emergency.
Spinal Epidural Hematoma
- Features: Acute severe back pain; rapid neurological deterioration (hours); history of anticoagulation, trauma, or spinal procedure.
- Examination: No fever; progressive motor/sensory deficits.
- Investigations: Coagulation studies abnormal if iatrogenic; MRI shows epidural hematoma (hyperintense T1 if subacute).
- Management: Reverse coagulopathy; emergency decompression.
Spinal Metastases/Malignancy
- Features: Insidious onset; progressive over weeks; night pain; unresponsive to analgesia; weight loss; known cancer history.
- Examination: Focal spine tenderness; may have upper motor neuron signs if cord compression.
- Investigations: MRI shows vertebral body destruction, epidural mass, possible multilevel involvement; CXR/CT chest-abdomen-pelvis for primary; bone scan/PET-CT for staging.
- Management: Oncology referral; dexamethasone; surgical decompression + radiotherapy vs. radiotherapy alone depending on prognosis and surgical candidacy.
Neurogenic Bladder (Non-CES Causes)
- Causes: Benign prostatic hyperplasia (BPH), urethral stricture, diabetic autonomic neuropathy, multiple sclerosis, stroke.
- Distinguishing Features: No acute back pain; no saddle anesthesia; no bilateral sciatica; gradual onset.
- Investigations: Urological assessment (flow studies, cystoscopy); MRI brain/spine if MS suspected.
9. Management Algorithm
SUSPECTED CAUDA EQUINA SYNDROME
(Back Pain + ≥1 Red Flag: Bilateral Sciatica,
Saddle Anaesthesia, Bladder/Bowel Dysfunction, Saddle Sensory Change)
↓
════════════════════════════════════════
IMMEDIATE ASSESSMENT (Emergency Department)
════════════════════════════════════════
↓
┌───────────────────────────────────────────────┐
│ • Full Neurological Examination │
│ • Bladder Scan (Post-Void Residual) │
│ • Digital Rectal Exam (Tone + Sensation) │
│ • Document Timing of Symptom Onset │
└───────────────────────────────────────────────┘
↓
┌───────────────────────────────────┐
│ BLADDER SCAN RESULT │
└───────────────────────────────────┘
↓ ↓
┌─────────────┐ ┌──────────────┐
│ less than 200ml PVR │ │ ≥200ml PVR │
│ Normal Tone│ │ or Reduced │
│ No Saddle │ │ Anal Tone or │
│ Sensation │ │ Saddle Signs │
│ Loss │ │ │
└─────────────┘ └──────────────┘
↓ ↓
┌─────────────┐ ┌──────────────────────┐
│ LOW SUSPICION│ │ HIGH SUSPICION │
│ │ │ (CES-S or CES-I) │
└─────────────┘ └──────────────────────┘
↓ ↓
┌─────────────┐ ┌──────────────────────┐
│ • Senior │ │ EMERGENCY MRI │
│ Review │ │ LUMBAR SPINE │
│ • Safety │ │ (Within 4 hours) │
│ Netting │ │ │
│ • Return │ │ • NPO (Nil by Mouth) │
│ if Red │ │ • IV Access │
│ Flags │ │ • Catheterize if │
│ Develop │ │ PVR > 500ml │
└─────────────┘ └──────────────────────┘
↓
┌──────────────────┐
│ MRI RESULT │
└──────────────────┘
┌─────────────┴──────────────┐
↓ ↓
┌─────────────────┐ ┌──────────────────────┐
│ NO COMPRESSION │ │ COMPRESSION CONFIRMED│
│ │ │ (Disc/Tumor/Abscess/ │
│ │ │ Hematoma/Fracture) │
└─────────────────┘ └──────────────────────┘
↓ ↓
┌─────────────────┐ ┌──────────────────────┐
│ • Reassess for │ │ URGENT NEUROSURGICAL │
│ Alternative │ │ CONSULTATION │
│ Diagnosis │ │ (Immediate) │
│ • Neurology/ │ └──────────────────────┘
│ Urology │ ↓
│ Referral │ ┌──────────────────────┐
└─────────────────┘ │ CLASSIFICATION │
└──────────────────────┘
↓ ↓ ↓
┌─────────┐ ┌──────────┐ ┌─────────┐
│ CES-I │ │ CES-R │ │ Tumor/ │
│(Incomplete)│(Retention)│ │ Abscess/│
│ │ │ │ │ Hematoma│
└─────────┘ └──────────┘ └─────────┘
↓ ↓ ↓
┌───────────────┐ ┌──────────┐ ┌────────────┐
│ EMERGENCY │ │ URGENT │ │ EMERGENCY │
│ SURGERY │ │ SURGERY │ │ SURGERY │
│ (Same Day/ │ │ (Within │ │ (Same Day) │
│ Overnight) │ │ 24h) │ │ + Antibiotics│
│ │ │ │ │ (if abscess)│
│ GOAL: less than 24h │ │GOAL: less than 48h│ │ │
│ from symptom │ │ │ │ │
│ onset │ │ │ │ │
└───────────────┘ └──────────┘ └────────────┘
↓ ↓ ↓
┌───────────────────────────────────────┐
│ SURGICAL DECOMPRESSION │
│ • Wide Laminectomy │
│ • Discectomy/Mass Excision │
│ • Ensure Complete Neural Decompression│
└───────────────────────────────────────┘
↓
┌───────────────────────────────────────┐
│ POST-OPERATIVE MANAGEMENT │
│ • Catheter Care (May need weeks) │
│ • Bowel Regimen │
│ • Early Mobilization │
│ • Rehabilitation Referral │
│ • Urology Follow-Up (Urodynamics) │
│ • Psychological Support │
└───────────────────────────────────────┘
10. Surgical Management
Indications for Emergency Surgery
- CES-I (Incomplete): Bladder dysfunction (hesitancy, retention) but no painless overflow incontinence → BEST PROGNOSIS, operate immediately (same day/overnight).
- CES-R (Retention): Established painless retention with overflow → Operate urgently (within 24h); damage likely but prevents further deterioration.
- Progressive Motor Deficit: Rapidly worsening leg weakness over hours.
- Epidural Abscess: Emergency decompression + IV antibiotics.
- Epidural Hematoma: Emergency evacuation ± reversal of coagulopathy.
Contraindications to Surgery
- Relative: Severe medical comorbidities (ASA 4-5) where anesthesia risk prohibitive; palliative cancer with life expectancy less than 3 months (consider radiotherapy).
- Absolute: Patient/family refusal after informed consent; complete CES for > 7 days with no motor function (salvage surgery still considered to prevent further root damage, but functional recovery unlikely).
Surgical Approaches
Posterior Lumbar Decompression (Standard Approach)
- Positioning: Prone on radiolucent table; padding of pressure points; avoid abdominal compression (reduces epidural venous bleeding).
- Incision: Midline posterior incision centered over affected level(s); typically 6-10cm.
- Technique:
- Exposure: Subperiosteal dissection of paraspinal muscles from spinous processes and laminae bilaterally.
- Laminectomy: Wide removal of laminae (typically 1-2 levels, e.g., L4 and L5) and ligamentum flavum to expose thecal sac.
- Inspection: Identify compressive pathology (disc, tumor, hematoma, abscess).
- Discectomy (if disc herniation): Perform limited durotomy if necessary (rare); retract nerve roots gently; remove extruded/sequestered disc material; curette disc space to prevent recurrence (controversial—may increase instability).
- Tumor/Abscess Excision: Remove compressive lesion; send tissue for histology/culture.
- Hemostasis: Meticulous bipolar cautery; gelfoam/surgicel; consider drain placement.
- Closure: Reapproximate fascia; subcuticular skin closure.
- Duration: 1-3 hours depending on complexity.
- Instrumentation: Fusion generally NOT required for CES decompression unless concomitant instability (fracture, tumor causing vertebral collapse). [33]
Minimally Invasive Techniques
- Microdiscectomy: Smaller incision, tubular retractors, microscope/endoscope; faster recovery but requires expertise; may be inadequate for massive central disc (need wide visualization of thecal sac).
- Role in CES: Controversial; most surgeons prefer open wide decompression for CES to ensure complete neural decompression and visualization of all roots.
Intraoperative Considerations
- Anesthesia: General anesthesia; avoid long-acting muscle relaxants if neuromonitoring used.
- Neuromonitoring: SSEP, motor evoked potentials (MEP), EMG; alerts to iatrogenic nerve injury.
- CSF Leak: If dura torn during decompression, primary repair with 5-0/6-0 non-absorbable suture; fibrin glue; consider lumbar drain.
- Blood Loss: Typically 200-500ml; epidural venous plexus bleeding common; controlled with bipolar cautery, gelfoam, patient positioning.
Post-Operative Management
Immediate (0-48 hours)
- Monitoring: Neurovascular observations (motor, sensory, sphincter function); vitals; drain output.
- Analgesia: Multimodal (paracetamol, NSAIDs, opioids); avoid excessive opioids (constipation worsens bowel dysfunction).
- Catheter Care: Maintain urinary catheter; strict aseptic technique; monitor urine output and residual volumes.
- Mobilization: Sit out of bed day 1; mobilize with physiotherapy day 1-2; no spinal restrictions (unless fusion performed).
- VTE Prophylaxis: Mechanical (TED stockings, pneumatic compression); chemical (LMWH) once hemostasis confirmed (12-24h post-op).
- Bowel Regimen: Stool softeners (docusate); stimulant laxatives (senna); avoid constipation.
Early Recovery (2-14 days)
- Catheter Trial: Attempt catheter removal 3-5 days post-op; measure post-void residuals; if > 200ml, re-catheterize and retry in 3-5 days.
- Intermittent Self-Catheterization (ISC): Teach if unable to void spontaneously; reduces infection risk vs. indwelling catheter.
- Physiotherapy: Gait re-education; strengthening; manage foot drop with ankle-foot orthosis (AFO) if persistent.
- Wound Care: Remove drain when output less than 50ml/24h (typically day 2-3); remove sutures/staples day 10-14.
- Discharge Planning: May require inpatient rehabilitation if severe deficits; home modifications; community support.
Long-Term (Weeks to Months)
- Bladder Management: Urology referral; urodynamic studies at 3-6 months to assess detrusor function; options include ISC, indwelling catheter, suprapubic catheter, or sacral neuromodulation.
- Bowel Management: Gastroenterology referral; bowel regimen; consider transanal irrigation, sacral neuromodulation.
- Sexual Dysfunction: Urology/sexual health referral; phosphodiesterase-5 inhibitors (sildenafil) may help if vascular component; penile prosthesis if nerve damage complete.
- Pain Management: Neuropathic pain common (20-40%); gabapentin, pregabalin, duloxetine, amitriptyline; pain clinic referral.
- Psychological Support: Depression, anxiety, adjustment disorder common; counseling, CBT, antidepressants as needed.
- Vocational Rehabilitation: Return to work planning; occupational therapy assessment.
Surgical Complications
Intraoperative
- Dural Tear (5-10%): CSF leak; risk of headache, pseudomeningocele; repair primarily.
- Nerve Root Injury (1-2%): Iatrogenic traction/laceration; worsens motor/sensory deficit.
- Vascular Injury (less than 1%): Epidural vein injury (common, controlled with cautery); rare arterial injury (great vessel, segmental artery).
- Incomplete Decompression: Missed lateral recess stenosis, residual disc fragment; may require reoperation.
Early Post-Operative (0-6 weeks)
- Hematoma (1-2%): Epidural hematoma causing recurrent compression; presents with acute neurological deterioration; requires emergency evacuation.
- Wound Infection (2-5%): Superficial (erythema, discharge) or deep (abscess); treat with antibiotics ± debridement.
- CSF Leak/Pseudomeningocele (1-3%): Persistent CSF leak through wound; may require re-exploration and dural repair.
- Urinary Retention: Expected initially; persistence beyond 2 weeks suggests permanent denervation.
- VTE (1-2%): DVT/PE; prophylaxis reduces risk.
Late Post-Operative (> 6 weeks)
- Recurrent Disc Herniation (3-5% at 5 years): Same level; may require revision surgery.
- Adjacent Segment Degeneration: Accelerated degeneration at levels above/below decompression (controversial whether caused by surgery or natural history).
- Chronic Pain (20-40%): Neuropathic pain, failed back surgery syndrome; multifactorial.
- Permanent Neurological Deficit: Bladder dysfunction (20-50%), bowel dysfunction (10-30%), sexual dysfunction (30-60%), motor weakness (10-20%)—dependent on timing of decompression and severity at presentation. [34]
11. Prognosis and Outcomes
Factors Influencing Prognosis
Time to Decompression (Most Critical)
- less than 24 hours: 70-80% bladder recovery; 80-90% motor recovery. [5]
- 24-48 hours: 50-60% bladder recovery; 70-80% motor recovery. [6]
- > 48 hours: 30-40% bladder recovery; 60-70% motor recovery. [26]
- > 7 days: less than 20% bladder recovery; limited motor recovery. [27]
Severity at Presentation
- CES-I (Incomplete): Best prognosis; 60-70% achieve normal or near-normal bladder function. [35]
- CES-R (Retention with Overflow): Poor prognosis; 20-30% achieve normal bladder function; 40-50% require long-term ISC or indwelling catheter. [36]
Age
- Younger Patients (less than 50 years): Better neurological recovery potential.
- Elderly (> 70 years): Higher comorbidity burden; slower rehabilitation; higher surgical risk.
Degree of Canal Compromise
- > 75% Stenosis: Associated with worse outcomes; greater neural ischemia.
- Complete Canal Obliteration: Worse prognosis than partial compression.
Duration of Symptoms Before Onset of Sphincter Dysfunction
- Acute Presentation (hours to days of back pain before CES): Typically disc herniation; better outcomes with prompt surgery.
- Chronic Presentation (weeks to months of symptoms before CES): Typically tumor or stenosis; worse baseline neural health; poorer recovery.
Functional Outcomes by Domain
Bladder Function
- Complete Recovery: 30-50% overall (highly dependent on surgical timing). [34]
- Partial Recovery (ISC required): 30-40%.
- Permanent Catheter (Indwelling or Suprapubic): 10-20%.
- Recovery Timeline: Gradual over 6-24 months; most recovery in first 6 months.
Bowel Function
- Complete Recovery: 50-70% (better than bladder, as bowel has sympathetic + parasympathetic innervation and sacral reflexes may partly recover). [10]
- Partial Recovery (Manageable with Regimen): 20-30%.
- Permanent Incontinence/Colostomy: 5-10%.
Sexual Function
- Males:
- Erectile Dysfunction: 30-60% (variable recovery; phosphodiesterase inhibitors help in 20-40%). [37]
- Ejaculatory Dysfunction: 20-40%.
- Females:
- Lubrication/Orgasm Dysfunction: 20-50%.
- Recovery: Least likely to recover fully; profound impact on quality of life.
Motor Function
- Complete Recovery: 60-80% (motor recovery better than autonomic). [38]
- Partial Recovery (Residual Weakness): 15-25%; may require AFO for foot drop.
- Permanent Paralysis: less than 5% (rare with adequate decompression).
Pain
- Leg Pain Resolution: 70-90% (sciatica typically improves rapidly post-decompression).
- Back Pain Resolution: 50-70% (may persist due to disc degeneration, surgical scarring).
- Chronic Neuropathic Pain: 20-40% develop chronic neuropathic pain requiring long-term management. [39]
Quality of Life
- Return to Baseline Function: 30-40% overall.
- Significant Disability: 30-40% (require lifestyle modifications, unable to return to previous employment, sexual dysfunction, psychological impact).
- Severe Disability: 10-20% (wheelchair/catheter dependent, requires significant care).
Mortality
- Perioperative Mortality: less than 1% for disc herniation; higher for tumor (5-10%) and epidural abscess (5-15%) due to underlying disease. [40]
- Long-Term Mortality: Increased in patients with permanent disability due to complications (UTI, pressure ulcers, VTE, depression/suicide).
12. Medicolegal Considerations
Litigation Landscape
- Incidence: CES accounts for up to 40% of all spinal surgery litigation claims in the UK and Australia, despite being rare clinically. [11,20]
- Median Award: £500,000-£2,000,000 (UK); dependent on severity and age (loss of earnings, care costs). [41]
- Common Allegations:
- Failure to recognize red flags (delayed diagnosis).
- Failure to perform/arrange urgent MRI.
- Delay to surgery (e.g., operating at 72h when patient presented at 12h).
- Inadequate documentation (timing of symptom onset unclear).
- Failure to warn patient of red flags on discharge from ED.
Defensive Medicine and Guidelines
- BASS Standards (2018): British Association of Spine Surgeons published consensus standards to reduce variation and improve outcomes; emphasizes MRI availability 24/7, clear pathways, documentation. [28]
- NICE Guidance (UK): Recognizes CES as neurosurgical emergency; recommends emergency MRI and surgical referral.
- SBNS/BOA Guidance: Society of British Neurological Surgeons and British Orthopaedic Association endorse BASS standards.
Critical Documentation Elements
- Timing: Exact date and time of onset of each symptom (back pain, leg pain, bladder symptoms, saddle sensation loss). Use patient's own words.
- Red Flag Inquiry: Document that you specifically asked about bilateral sciatica, saddle numbness, bladder/bowel dysfunction.
- Bladder Scan Result: Record PVR volume and time of measurement.
- Examination Findings: Detailed motor, sensory, reflex, and rectal examination with grading (e.g., "Anal tone 3/5, reduced perianal sensation S4-5 bilaterally").
- Imaging: Time MRI requested, time performed, time results reviewed; documented discussion with radiologist/neurosurgeon.
- Decision-Making: Rationale for surgery timing; why emergency vs. urgent vs. delayed; document discussion with patient/family re: risks/benefits.
- Safety Netting: If discharging patient without CES diagnosis, document red flag advice given (e.g., "Return immediately if develop urinary retention, saddle numbness, or bilateral leg weakness").
High-Risk Scenarios for Litigation
- Missed Diagnosis in ED: Patient presents with back pain + 1 red flag, sent home without bladder scan or safety netting, returns 24h later with CES-R.
- Delayed MRI: Patient presents with CES-I, MRI delayed to next working day (36h), patient progresses to CES-R.
- Delayed Transfer: Patient in district hospital without neurosurgery; MRI confirms CES; delay of 24-48h transferring to tertiary center for surgery.
- Inadequate Consent: Patient not counseled pre-operatively about likely permanent bladder dysfunction; sues for "failure to warn."
13. Patient Education and Counseling
Explaining the Diagnosis
For Laypersons
"You have a condition called Cauda Equina Syndrome. At the bottom of your spinal cord, there are nerves that look like a horse's tail—that's called the 'cauda equina.' A disc in your back has popped out and is squashing these nerves. These nerves control your bladder, bowel, sexual function, and legs. If we don't relieve the pressure quickly by operating, the nerves can be permanently damaged, and you might need a catheter for life to empty your bladder, and lose control of your bowels. The surgery involves removing the part of the disc that is pressing on the nerves."
For Medical Students
"CES is compression of the lumbosacral nerve roots (L2-S5) below the conus medullaris. The classic presentation is bilateral sciatica, saddle anesthesia, and urinary retention. The key is distinguishing CES-I (incomplete, with bladder dysfunction but no established retention) from CES-R (retention with overflow incontinence). CES-I patients operated within 24h have the best prognosis. The surgery is a wide laminectomy and discectomy. This is a 'never miss' diagnosis; always do a bladder scan and DRE in back pain with red flags."
For Clinicians (Emergency/Primary Care)
"Maintain a low threshold for suspicion in any patient with back pain plus bilateral symptoms, saddle paresthesia, or new urinary symptoms. A bladder scan > 200ml post-void or reduced anal tone mandates emergency MRI (within 4h). Do not delay imaging or neurosurgical referral. Document timing meticulously. If discharging without imaging, provide explicit red flag advice and arrange urgent follow-up. CES is the highest litigation risk in spinal practice; err on the side of over-investigation."
Pre-Operative Counseling
Realistic Expectations
- Motor Recovery: "Your leg strength and pain will likely improve significantly after surgery, though it may take weeks to months."
- Bladder Recovery: "Your bladder function may improve, but recovery is slow (6-24 months) and may be incomplete. You might need to use a catheter temporarily or even long-term. The earlier we operate, the better your chances."
- Bowel Recovery: "Bowel function usually recovers better than bladder, though you may need a bowel regimen (laxatives, suppositories)."
- Sexual Function: "Sexual function may be affected, including erections, lubrication, and sensation. We can refer you to specialists to help manage this."
- Timeframe: "The surgery is urgent and will happen within the next few hours (or tonight). We cannot wait."
Surgical Risks
- Common: Pain, bleeding, infection (5%), CSF leak (5%), catheter for weeks-months.
- Uncommon: Nerve damage causing worsening deficit (1-2%), blood clots (1-2%), recurrent disc herniation (5% over years).
- Rare: Paralysis, death (less than 1%).
Alternative to Surgery
- "If we do not operate, the pressure on the nerves will continue and you will almost certainly have permanent bladder/bowel/sexual dysfunction and possibly paralysis. There is no effective non-surgical treatment for CES."
Post-Operative Counseling
Discharge Advice
- Wound Care: Keep clean and dry; remove dressing after 3 days; watch for signs of infection (redness, pus, fever).
- Activity: Gradual return to normal activity; avoid heavy lifting (> 5kg) for 6 weeks; no spinal restrictions for walking/sitting.
- Bladder/Bowel: Continue catheterization as instructed; follow up with urology in 4-6 weeks.
- Red Flags for Return: New leg weakness, loss of sensation, wound infection, severe headache (CSF leak), inability to pass urine (if catheter removed).
Long-Term Support
- Rehabilitation: Physiotherapy for strength/gait; occupational therapy for ADLs and home modifications.
- Continence Services: ISC training; pads/appliances; specialist continence nurse.
- Sexual Health: Urology/sexual health referral; psychological support.
- Psychological Support: Counseling; support groups (Cauda Equina Syndrome Association—CESA UK).
- Vocational: Phased return to work; occupational health assessment; disability benefits if unable to work.
14. Prevention and Risk Reduction
Primary Prevention (Population Level)
- Back Health Education: Ergonomic workplace design; manual handling training; core strengthening exercises.
- Obesity Management: Reduce mechanical load on lumbar spine.
- Smoking Cessation: Smoking accelerates disc degeneration.
Secondary Prevention (High-Risk Individuals)
- Early Treatment of Sciatica: Timely imaging and intervention for large disc herniations before progression to CES.
- Cancer Surveillance: Monitoring for spinal metastases in patients with known malignancy.
- Spinal Stenosis Management: Decompression surgery for severe stenosis before acute-on-chronic CES.
Tertiary Prevention (Post-CES)
- Recurrence Prevention: Avoid heavy lifting; core strengthening; maintain healthy weight.
- Complication Prevention: UTI prevention (aseptic ISC technique, hydration); pressure ulcer prevention (if wheelchair-bound); VTE prophylaxis during acute phase.
Healthcare System Level
- 24/7 MRI Access: All hospitals receiving emergency back pain patients must have 24/7 MRI capability or rapid transfer protocol.
- Clinical Pathways: Standardized CES pathways (triage tools, bladder scan protocols, emergency MRI slots, neurosurgical on-call).
- Clinician Education: Mandatory training for ED/primary care on CES red flags; regular audit of missed cases.
- Medico-Legal Risk Management: Robust documentation templates; safety netting advice templates; incident reporting and learning.
15. Special Populations
Pregnancy
- Risk: Disc herniation more common in pregnancy (hormonal ligamentous laxity, mechanical load, Valsalva during labor).
- Imaging: MRI safe in pregnancy (no ionizing radiation); contrast (gadolinium) avoided in pregnancy but CES diagnosis rarely requires contrast.
- Management: Surgical decompression still indicated urgently; caesarean section may be performed first if fetus viable and patient stable.
Elderly (> 75 years)
- Etiology: More likely tumor, stenosis, or fracture than disc herniation.
- Surgical Risk: Higher perioperative risk (cardiac, respiratory comorbidities); may require ICU post-operatively.
- Prognosis: Slower neurological recovery; higher risk of permanent disability; rehabilitation more prolonged.
Anticoagulated Patients
- Cause: Higher risk of epidural hematoma (spontaneous or post-spinal procedure).
- Management: Reverse anticoagulation urgently if hematoma suspected (vitamin K/PCC for warfarin; idarucizumab for dabigatran; andexanet alfa for rivaroxaban/apixaban); proceed to emergency decompression; resume anticoagulation post-operatively once hemostasis confirmed (discuss with hematology).
Immunocompromised (HIV, Transplant, Chemotherapy)
- Cause: Higher risk of epidural abscess, fungal infection, or opportunistic infection (TB, toxoplasmosis).
- Imaging: MRI with contrast to differentiate infection from tumor.
- Management: Emergency decompression + broad-spectrum antibiotics + antifungals/antivirals as indicated; send tissue for extensive microbiology/histopathology.
Previous Lumbar Surgery
- Risk: Epidural scarring complicates surgical dissection; higher risk of dural tear and nerve injury.
- Management: Experienced spinal surgeon required; longer operative time; consider neuromonitoring.
16. Emerging Research and Future Directions
Biomarkers for Early Detection
- Serum Neurofilament Light Chain (NfL): Elevated in acute nerve injury; potential blood test to quantify neural damage severity and predict prognosis. [42]
- Urinary Nerve Growth Factor (NGF): Elevated in neurogenic bladder; may predict recovery.
Advanced Imaging
- Diffusion Tensor Imaging (DTI): MRI technique visualizing nerve fiber integrity; may identify early ischemic damage before symptoms irreversible.
- MR Neurography: High-resolution imaging of individual nerve roots; may improve surgical planning.
Surgical Innovations
- Endoscopic Decompression: Fully endoscopic techniques reduce tissue trauma; early data suggest faster recovery, but efficacy in CES unclear (need wide decompression).
- Intraoperative Ultrasound: Real-time imaging to confirm complete decompression without residual compression.
Pharmacological Adjuncts
- Neuroprotective Agents: Steroids (controversial; not recommended for disc-related CES but used in tumor/abscess); riluzole (glutamate antagonist; under investigation).
- Regenerative Medicine: Stem cell therapy; nerve growth factors; experimental only.
Rehabilitation Technologies
- Sacral Neuromodulation: Electrical stimulation of sacral nerve roots to improve bladder/bowel control; FDA-approved for neurogenic bladder; may benefit CES patients with partial denervation.
- Exoskeletons: Robotic gait training for patients with persistent lower limb weakness.
Medicolegal and Policy
- AI Triage Tools: Machine learning algorithms to identify CES risk in ED; flag high-risk patients for urgent review.
- National Audits: UK National CES Audit (proposed) to track outcomes, delays, and litigation; drive quality improvement.
17. Examination Focus (Viva Vault)
FRCS/MRCS Oral Examination Questions
Q1: A 45-year-old man presents to ED with 3 days of back pain and bilateral leg pain. He mentions difficulty passing urine this morning. What is your differential diagnosis and immediate management?
Model Answer: "This presentation raises high suspicion for Cauda Equina Syndrome, but I would also consider acute lumbar radiculopathy, epidural abscess, spinal tumor, or Guillain-Barré syndrome. My immediate steps are:
- Focused history: Timing of urinary symptoms (hesitancy vs. retention vs. incontinence), saddle sensation, bowel dysfunction, trauma, cancer history, fever, IV drug use.
- Examination: Full lower limb neurological exam (motor L2-S1, sensory including saddle area, reflexes, perianal sensation, digital rectal exam for anal tone).
- Bladder scan: Measure post-void residual; > 200ml is highly suspicious.
- If PVR > 200ml or reduced anal tone, arrange emergency MRI lumbar spine within 4 hours.
- Keep patient nil by mouth and contact neurosurgery/spinal surgery immediately.
- If abscess suspected (fever, elevated CRP), start IV antibiotics while awaiting MRI."
Q2: Differentiate CES-I from CES-R and explain the prognostic significance.
Model Answer: "CES-I (Incomplete) is defined by altered urinary sensation and difficulty voiding—hesitancy, poor stream, reduced desire to void—but retention has not become established and the patient has not experienced painless overflow incontinence. They are still aware of their bladder status. Post-void residual is typically 200-500ml.
CES-R (Retention with overflow) is defined by established painless urinary retention where the bladder is mechanically paralyzed. The patient is unaware of bladder fullness and has experienced painless wetting. PVR is typically > 500ml.
The prognostic significance is critical: CES-I patients who undergo decompression within 24 hours have a 60-70% chance of achieving normal or near-normal bladder function. CES-R patients have only a 20-30% chance of normal bladder recovery, and 40-50% will require lifelong intermittent self-catheterization or indwelling catheter, even with urgent surgery. Therefore, identifying and operating on CES-I patients represents the best opportunity to preserve function. This is why we emphasize emergency MRI and same-day surgery for CES-I."
Q3: What is the evidence regarding timing of surgery in CES, and how would you counsel a patient presenting at 60 hours from symptom onset?
Model Answer: "The landmark evidence comes from several studies:
- Todd (2005) and Ahn et al. (2000) meta-analyses showed that decompression within 48 hours resulted in significantly better bladder and bowel outcomes compared to > 48h.
- More recently, Shapiro (2000) and Korse (2017) suggested that surgery within 24 hours is associated with even better outcomes, particularly for bladder recovery (70-80% vs. 50-60% for 24-48h).
- Beyond 48 hours, recovery rates drop to 30-40% for bladder function.
However, I would counsel the patient at 60 hours that while they are beyond the 'optimal' window, surgery is still indicated and may prevent further deterioration. Even delayed decompression can result in partial recovery and prevent progression. I would explain that permanent bladder dysfunction requiring catheterization is likely, but not certain. Surgery will also address leg pain and motor weakness, which typically recover better than autonomic function. I would arrange urgent surgery (within hours) and involve urology and rehabilitation teams for long-term planning. Importantly, I would document this discussion meticulously for medicolegal purposes."
Q4: Describe the surgical technique for decompression in a massive L4/5 central disc herniation causing CES-I.
Model Answer: "The patient is positioned prone on a radiolucent table with chest and pelvic supports to reduce abdominal pressure and minimize epidural venous bleeding. A midline incision is made centered over L4/5. Subperiosteal dissection exposes the L4 and L5 laminae bilaterally.
I perform a wide bilateral laminectomy of L4 and L5, removing the spinous processes, laminae, and ligamentum flavum to achieve generous exposure of the thecal sac. The key principle is adequate decompression of all nerve roots—I must visualize the thecal sac 'breathing' freely without compression.
Once the thecal sac is exposed, I identify the central disc herniation compressing the cauda equina. I perform a limited annulotomy if the disc is still contained, or remove extruded/sequestered fragments if free. Gentle retraction of the thecal sac and nerve roots may be necessary, using nerve root retractors carefully. I remove all disc material compressing the neural elements and gently curate the disc space to remove loose fragments, though aggressive discectomy is controversial as it may increase instability risk.
Hemostasis is achieved with bipolar cautery and hemostatic agents. I typically place a drain. Closure is in layers: fascia with absorbable sutures, subcuticular skin closure. Post-operatively, the patient is mobilized early, bladder function is monitored, and catheter trial is attempted after 3-5 days. I do not routinely fuse unless there is concomitant fracture or tumor-related instability."
Q5: What are the medicolegal pitfalls in CES management?
Model Answer: "CES represents the highest litigation risk in spinal surgery, accounting for 40% of neurosurgical claims. The key pitfalls are:
- Failure to diagnose: Missing red flags in ED—not asking about bladder/bowel symptoms, not performing bladder scan or DRE, discharging patient without safety netting.
- Delayed MRI: Patient presents with clear red flags, but MRI delayed to next day or beyond 4 hours; progression from CES-I to CES-R during delay.
- Delayed surgery: MRI confirms CES, but surgery delayed beyond 24-48h due to theater availability or transfer delays.
- Inadequate documentation: Timing of symptom onset not clearly recorded; red flag inquiry not documented; examination findings vague.
- Failure to warn: Patient discharged from ED without CES diagnosis, but not counseled about red flags and when to return immediately.
To mitigate these risks, I ensure:
- All back pain presentations have documented red flag screen.
- Bladder scan is mandatory if any suspicion.
- Emergency MRI within 4 hours for suspected CES.
- Clear communication with neurosurgery; documented discussion.
- Meticulous documentation using structured templates, including exact timings in patient's own words.
- Safety netting advice given verbally and in writing if discharging without CES diagnosis.
- Post-operative counseling about realistic expectations documented in consent process."
18. References
-
Todd NV. Cauda equina syndrome: the timing of surgery probably does influence outcome. Br J Neurosurg. 2005;19(4):301-6. doi:10.1080/02688690500305324
-
Gleave JR, Macfarlane R. Cauda equina syndrome: what is the relationship between timing of surgery and outcome? Br J Neurosurg. 2002;16(4):325-8. doi:10.1080/0268869021000032887
-
Ahn UM, Ahn NU, Buchowski JM, et al. Cauda equina syndrome secondary to lumbar disc herniation: a meta-analysis of surgical outcomes. Spine. 2000;25(12):1515-22. doi:10.1097/00007632-200006150-00010
-
Shapiro S. Medical realities of cauda equina syndrome secondary to lumbar disc herniation. Spine. 2000;25(3):348-52. doi:10.1097/00007632-200002010-00015
-
Srikandarajah N, Boissaud-Cooke MA, Clark S, Wilby MJ. Does early surgical decompression in cauda equina syndrome improve bladder outcome? Spine. 2015;40(8):580-3. doi:10.1097/BRS.0000000000000813
-
Korse NS, Pijpers JA, van Zwet E, et al. Cauda equina syndrome: presentation, outcome, and predictors with focus on micturition, defecation, and sexual dysfunction. Eur Spine J. 2017;26(3):894-904. doi:10.1007/s00586-017-4943-8
-
Podnar S. Epidemiology of cauda equina and conus medullaris lesions. Muscle Nerve. 2007;35(4):529-31. doi:10.1002/mus.20753
-
Germon T, Ahuja S, Casey ATH, et al. British Association of Spine Surgeons standards of care for cauda equina syndrome. Spine J. 2015;15(3S):S2-4. doi:10.1016/j.spinee.2015.01.006
-
Thanapalasingam C, Sabatino MJ, Thawrani DP, Raad M. Post-void residual volume as a screening tool for cauda equina syndrome: a systematic review. Spine J. 2020;20(11):1828-35. doi:10.1016/j.spinee.2020.06.006
-
DeLong WB, Polissar N, Neradilek B. Timing of surgery in cauda equina syndrome with urinary retention: meta-analysis of observational studies. J Neurosurg Spine. 2008;8(4):305-20. doi:10.3171/SPI/2008/8/4/305
-
Hoeritzauer I, Wood M, Copeland L, et al. What is the incidence of cauda equina syndrome? A systematic review. J Neurosurg Spine. 2020;32(2):150-61. doi:10.3171/2019.9.SPINE19586
-
Dionne N, Adeeb N, Moisi M, et al. Interobserver reliability of digital rectal examination in the assessment of anal sphincter tone. World Neurosurg. 2019;125:e1382-e1387. doi:10.1016/j.wneu.2019.02.090
-
Rider IS, Marra EM, Sloan TH. Cauda Equina Syndrome. 2023. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing. PMID: 28723029
-
Kostuik JP, Harrington I, Alexander D, et al. Cauda equina syndrome and lumbar disc herniation. J Bone Joint Surg Am. 1986;68(3):386-91. PMID: 3949835
-
Sun JC, Xu T, Chen KF, et al. Assessment of cauda equina syndrome progression pattern to improve diagnosis. Spine. 2014;39(7):596-602. doi:10.1097/BRS.0000000000000219
-
Porter RW, Hibbert C, Wellman P. Backache and the lumbar spinal canal. Spine. 1980;5(2):99-105. doi:10.1097/00007632-198003000-00002
-
Thongtrangan I, Le H, Park J, Kim DH. Cauda equina syndrome in patients with low lumbar fractures. Neurosurg Focus. 2004;16(6):e6. doi:10.3171/foc.2004.16.6.7
-
Bateman DK, Millhouse PW, Shahi N, et al. Epidural hematoma following epidural steroid injection: a systematic review. Spine. 2016;41(20):E1221-E1230. doi:10.1097/BRS.0000000000001673
-
Todd NV, Dickson RA. Standards of care in cauda equina syndrome. Br J Neurosurg. 2016;30(5):518-22. doi:10.1080/02688697.2016.1187254
-
Daniels EW, Gordon Z, French K, et al. Review of medicolegal cases for cauda equina syndrome: what factors lead to an adverse outcome for the provider? Orthopedics. 2012;35(3):e414-9. doi:10.3928/01477447-20120222-29
-
Saifuddin A, Burnett SJ, White J. The variation of position of the conus medullaris in an adult population. A magnetic resonance imaging study. Spine. 1998;23(13):1452-6. doi:10.1097/00007632-199807010-00005
-
Brouwers E, van de Meent H, Curt A, et al. Definitions of traumatic conus medullaris and cauda equina syndrome: a systematic literature review. Spinal Cord. 2017;55(10):886-90. doi:10.1038/sc.2017.61
-
Schizas C, Theumann N, Burn A, et al. Qualitative grading of severity of lumbar spinal stenosis based on the morphology of the dural sac on magnetic resonance images. Spine. 2010;35(21):1919-24. doi:10.1097/BRS.0b013e3181d359bd
-
Rydevik BL, Pedowitz RA, Hargens AR, et al. Effects of acute, graded compression on spinal nerve root function and structure. An experimental study of the pig cauda equina. Spine. 1991;16(5):487-93. doi:10.1097/00007632-199105000-00001
-
Olmarker K, Rydevik B. Pathophysiology of sciatica. Orthop Clin North Am. 1991;22(2):223-34. PMID: 1826546
-
Kohles SS, Kohles DA, Karp AP, et al. Time-dependent surgical outcomes following cauda equina syndrome diagnosis: comments on a meta-analysis. Spine. 2004;29(11):1281-7. doi:10.1097/00007632-200406010-00020
-
McCarthy MJ, Aylott CE, Grevitt MP, Hegarty J. Cauda equina syndrome: factors affecting long-term functional and sphincteric outcome. Spine. 2007;32(2):207-16. doi:10.1097/01.brs.0000251750.76492.93
-
Lavy C, James A, Wilson-MacDonald J, Fairbank J. Cauda equina syndrome. BMJ. 2009;338:b936. doi:10.1136/bmj.b936
-
Fraser S, Roberts L, Murphy E. Cauda equina syndrome: a literature review of its definition and clinical presentation. Arch Phys Med Rehabil. 2009;90(11):1964-8. doi:10.1016/j.apmr.2009.03.021
-
Bell DA, Collie D, Statham PF. Cauda equina syndrome—what is the correlation between clinical assessment and MRI scanning? Br J Neurosurg. 2007;21(2):201-3. doi:10.1080/02688690701317144
-
Fairbank JC, Pynsent PB. The Oswestry Disability Index. Spine. 2000;25(22):2940-52. doi:10.1097/00007632-200011150-00017
-
van Rijn JC, Klemetso N, Reitsma JB, et al. Observer variation in the evaluation of lumbar herniated discs and root compression: spiral CT compared with MRI. Br J Radiol. 2006;79(941):372-7. doi:10.1259/bjr/53801778
-
Aono H, Ohwada T, Hosono N, et al. Incidence of postoperative symptomatic epidural hematoma in spinal decompression surgery. J Neurosurg Spine. 2011;15(2):202-5. doi:10.3171/2011.3.SPINE10716
-
Chau AM, Xu LL, Pelzer NR, Gragnaniello C. Timing of surgical intervention in cauda equina syndrome: a systematic critical review. World Neurosurg. 2014;81(3-4):640-50. doi:10.1016/j.wneu.2013.11.007
-
Tandon PN, Sankaran B. Cauda equina syndrome due to lumbar disc prolapse. Indian J Orthop. 1967;1:112-9.
-
Qureshi A, Sell P. Cauda equina syndrome treated by surgical decompression: the influence of timing on surgical outcome. Eur Spine J. 2007;16(12):2143-51. doi:10.1007/s00586-007-0491-y
-
Bartolin Z, Krnić D,Uki I. Importance of urgent MRI in the diagnosis of cauda equina syndrome. Acta Clin Croat. 2012;51(4):663-6. PMID: 23540172
-
Gitelman A, Hishmeh S, Morelli BN, et al. Cauda equina syndrome: a comprehensive review. Am J Orthop. 2008;37(11):556-62. PMID: 19104682
-
Hussain SA, Gullan RW, Chitnavis BP. Cauda equina syndrome: outcome and implications for management. Br J Neurosurg. 2003;17(2):164-7. doi:10.1080/0268869031000108573
-
Hsu K, Zucherman J, White A, Wynne G. Deterioration of motion segments adjacent to lumbar spine fusions. Presented at the International Society for the Study of the Lumbar Spine, 1988.
-
Gardner A, Gardner E, Morley T. Cauda equina syndrome: a review of the current clinical and medicolegal position. Eur Spine J. 2011;20(5):690-7. doi:10.1007/s00586-010-1668-3
-
Kuhle J, Barro C, Andreasson U, et al. Comparison of three analytical platforms for quantification of the neurofilament light chain in blood samples: ELISA, electrochemiluminescence immunoassay and Simoa. Clin Chem Lab Med. 2016;54(10):1655-61. doi:10.1515/cclm-2015-1195
19. Key Takeaways
For Emergency Department Clinicians
- Low Threshold: Any back pain patient with bilateral leg symptoms, bladder/bowel symptoms, or saddle numbness warrants bladder scan and consideration of CES.
- Bladder Scan is Mandatory: PVR > 200ml in context of back pain is CES until proven otherwise.
- Emergency MRI: Must be available 24/7; do not delay to next working day.
- Safety Netting: If discharging, provide explicit verbal and written advice about red flags.
- Documentation: Record exact timing of symptom onset and red flag inquiry.
For Spinal Surgeons
- Time is Spine: Operate on CES-I within 24h for best outcomes; every hour counts.
- Wide Decompression: Ensure all nerve roots are visualized and decompressed; don't rush.
- Realistic Counseling: CES-R patients will likely have permanent bladder dysfunction; document this conversation.
- Multidisciplinary: Involve urology, rehabilitation, psychology early.
For Patients
- Red Flags: If you have severe back pain and develop numbness between your legs, difficulty passing urine, or weakness in both legs, go to hospital immediately.
- Recovery: Bladder recovery is slow (months to years) and may be incomplete; leg strength usually recovers better.
- Support: Join patient support groups (e.g., Cauda Equina Champions Charity UK); rehabilitation is key.
For Medical Students and Trainees
- Never Miss Diagnosis: CES is rare but devastating; always ask about bladder/bowel/saddle symptoms in back pain.
- CES-I vs. CES-R: The critical distinction is whether overflow incontinence has occurred; CES-I has best prognosis.
- Litigation Risk: CES is the highest litigation risk in spinal surgery; rigorous documentation and adherence to guidelines are essential.
Frequently asked questions
Quick clarifications for common clinical and exam-facing questions.
When should I seek emergency care for cauda equina syndrome?
Seek immediate emergency care if you experience any of the following warning signs: Bilateral Sciatica -> Central disc herniation, Saddle Anaesthesia -> Loss of S3-S5 sensation, Bladder Retention -> Painless overflow incontinence, Anal Tone Loss -> Inability to contract sphincter, Acute deterioration of neurological function, Progressive bilateral leg weakness.
Learning map
Use these linked topics to study the concept in sequence and compare related presentations.
Prerequisites
Start here if you need the foundation before this topic.
- Lumbar Spine Anatomy
- Spinal Cord and Nerve Root Anatomy
- Lower Limb Neurological Examination
Differentials
Competing diagnoses and look-alikes to compare.
- Conus Medullaris Syndrome
- Acute Spinal Cord Compression
- Peripheral Polyneuropathy
- Guillain-Barré Syndrome
Consequences
Complications and downstream problems to keep in mind.
- Neurogenic Bladder Dysfunction
- Chronic Neuropathic Pain Syndromes
- Neurological Sexual Dysfunction