Chronic Obstructive Pulmonary Disease (COPD) in Adults
Chronic Obstructive Pulmonary Disease (COPD) is a common, preventable, and treatable heterogeneous lung condition charac... MRCP, USMLE, FRACP exam preparation.
What matters first
Chronic Obstructive Pulmonary Disease (COPD) is a common, preventable, and treatable heterogeneous lung condition charac... MRCP, USMLE, FRACP exam preparation.
Acute worsening of dyspnea at rest
11 Jan 2026
Generated educational material; verify before clinical use.
Visible references section
See the concept before reading it
Study the key anatomy, imaging, and decision pathways as full teaching plates.
Clinical board
A visual summary of the highest-yield teaching signals on this page.
Urgent signals
Safety-critical features pulled from the topic metadata.
- Acute worsening of dyspnea at rest
- New-onset confusion or altered mental status (Hypercapnia)
- Use of accessory respiratory muscles
- Peripheral cyanosis or saturations less than 88% on room air
Exam focus
Current exam surfaces linked to this topic.
- MRCP, USMLE, FRACP
Content status and exam context
This page is AI-generated educational content. It may contain errors or omissions and is not a substitute for current guidelines, local protocols, senior clinical judgement, or professional medical advice.
MedVellum does not claim an individual clinician reviewer, board certification, or professional credential for this page unless a future version names a real, verifiable reviewer.
Clinical explanation and evidence
SECTION 1: Clinical Overview
1.1 Summary
Chronic Obstructive Pulmonary Disease (COPD) is a common, preventable, and treatable heterogeneous lung condition characterized by chronic respiratory symptoms including dyspnea, cough, and sputum production. These symptoms are primarily due to abnormalities of the airways (bronchitis, bronchiolitis) and/or alveoli (emphysema) that cause persistent, often progressive, airflow obstruction. [1,2]
Globally, COPD is a leading cause of morbidity and mortality, currently ranking as the third leading cause of death worldwide, with an estimated 3.2 million deaths annually. The disease is defined physiologically by a post-bronchodilator FEV1/FVC ratio of less than 0.70. [1] Clinical significance lies in its progressive nature and the high frequency of acute exacerbations (AECOPD), which significantly accelerate lung function decline and increase mortality risk.
Management has shifted from a reactive approach to a proactive, individualized strategy focusing on symptom reduction and risk mitigation (exacerbation prevention) using the GOLD ABE assessment tool introduced in 2023. [3,4] This represents a significant paradigm shift that merges the previous Groups C and D into a single "E" category for frequent exacerbators, simplifying clinical decision-making while maintaining prognostic accuracy.
Prognosis is generally poor if the primary insult (usually tobacco smoke) continues, but it can be significantly improved with smoking cessation, pulmonary rehabilitation, and optimized triple therapy (ICS/LABA/LAMA) where indicated. Recent landmark trials have demonstrated that triple therapy not only reduces exacerbations but also provides mortality benefit, fundamentally changing COPD management. [5,6]
1.2 Key Facts
- Definition: A heterogeneous lung condition characterized by chronic respiratory symptoms due to abnormalities of the airways and/or alveoli that cause persistent airflow obstruction.
- Incidence: Approximately 300-400 cases per 100,000 person-years in adults over 40.
- Prevalence: Global prevalence is estimated at 10.3% (95% CI 8.2%–12.8%) among individuals aged 30–79 years. [7]
- Mortality: Responsible for approximately 5-6% of all deaths globally; 1-year mortality after hospitalization for AECOPD is nearly 25%.
- Morbidity: Leading cause of "Years Lived with Disability" (YLD), with high rates of depression, skeletal muscle dysfunction, and cardiovascular co-morbidity.
- Peak Age: Most commonly diagnosed in the 5th to 7th decades of life; prevalence increases sharply with age.
- Sex Distribution: Historically higher in males, but prevalence in females is now equal or higher in many high-income countries due to changing smoking patterns.
- Pathognomonic Feature: There is no single pathognomonic feature, but the combination of chronic cough and "Hoover's sign" (inward movement of lower rib cage during inspiration) is highly suggestive.
- Gold Standard Investigation: Post-bronchodilator spirometry showing FEV1/FVC less than 0.70. [8]
- First-line Treatment: Long-acting bronchodilators (LAMA or LABA, or LAMA+LABA combination for symptomatic patients).
- Second-line Treatment: Triple therapy (ICS + LAMA + LABA) for patients with frequent exacerbations and high blood eosinophil counts.
- Key Complication: Acute Exacerbation of COPD (AECOPD), often triggered by viral or bacterial infections, leading to acute respiratory failure.
1.3 Clinical Pearls
Diagnostic Pearl: "The 0.70 Fixed Ratio Caveat" While FEV1/FVC less than 0.70 is the diagnostic threshold, this may lead to overdiagnosis in the elderly (due to natural lung aging) and underdiagnosis in young adults. The GOLD Science Committee now recommends considering Lower Limit of Normal (LLN) values in borderline cases, especially in non-smokers and those under 45 or over 65 years of age. [8]
Examination Pearl: "Hoover's Sign Significance" Observe the lower costal margins during inspiration. Inward movement (Hoover's sign) indicates severe hyperinflation and flattening of the diaphragm. This finding is clinically important because it correlates with a higher work of breathing and increased severity of airflow obstruction on spirometry, predicting worse outcomes.
Treatment Pearl: "The Eosinophil Threshold" Blood eosinophil counts should guide the use of Inhaled Corticosteroids (ICS). Patients with less than 100 cells/µL derive little benefit from ICS and face increased pneumonia risk, whereas those with > 300 cells/µL or > 100 cells/µL with frequent exacerbations show significant reduction in exacerbation rates with ICS. [9]
Pitfall Warning: "Oxygen over-prescription in AECOPD" Avoid high-flow oxygen in patients with suspected hypercapnic respiratory failure. Target SpO2 88–92% rather than > 94%. Excessive oxygen reduces hypoxic drive and worsens V/Q mismatch (Haldane effect), potentially leading to life-threatening respiratory acidosis.
Mnemonic: "The 6-M Management of COPD" Monitoring (Spirometry/Symptom scores), Modification of risk (Smoking cessation), Medication (LAMA/LABA/ICS), Motion (Pulmonary rehab), Microbes (Vaccinations/Antibiotics for AECOPD), and Mentality (Treating anxiety/depression).
Emergency Pearl: "The 'Silent Chest' Danger" In an acute exacerbation, a "silent chest" on auscultation is more ominous than loud wheezing. It indicates that airflow is so restricted that it cannot generate sound. This requires immediate aggressive bronchodilation and consideration for non-invasive ventilation (NIV).
Exam Pearl: "Alpha-1 Antitrypsin Screening" The WHO recommends screening every patient diagnosed with COPD at least once, regardless of age or smoking history, though it is particularly vital in those under 45 or with basal emphysema on imaging.
1.4 Why This Matters Clinically
- Patient outcomes: Failure to diagnose COPD early leads to irreversible loss of lung function. Delayed recognition of exacerbations increases the risk of intubation and death, as each severe exacerbation permanently lowers the patient's baseline functional status.
- Healthcare burden: COPD is one of the most common reasons for emergency department visits and hospital readmissions. The economic cost is staggering, driven by hospitalizations, long-term oxygen therapy, and lost productivity in the working-age population.
- Medico-legal: Failure to screen for Alpha-1 Antitrypsin deficiency in young patients or improper titration of oxygen leading to hypercapnic coma are common areas of clinical negligence. Documentation of smoking cessation advice is also a key quality metric.
- Training relevance: COPD is a "core" topic for all medical boards (USMLE, MRCP) because it integrates physiology (acid-base balance), pharmacology (receptor agonism/antagonism), and chronic disease management.
SECTION 2: Epidemiology
2.1 Incidence & Prevalence
- Incidence: Estimated at 9.2 cases per 1,000 person-years in adults over 45 years.
- Prevalence: Global prevalence is approximately 10.1% using GOLD criteria, though this varies significantly by region. Major population studies have confirmed substantial geographic variation, with higher rates in low-middle income countries (LMICs) due to biomass fuel exposure. [7]
- Lifetime Risk: For a 40-year-old smoker, the lifetime risk of developing COPD is approximately 25-30%.
- Trend: Increasing globally due to aging populations and continued high smoking rates in developing nations, despite declines in the West. Asia-Pacific region shows particularly rapid increases. [7]
- Geographic Variation: Higher in Eastern Europe and parts of Asia (e.g., China) due to high smoking prevalence and indoor air pollution from biomass fuels.
- Temporal Patterns: Hospitalizations for AECOPD peak in winter months, correlating with viral respiratory infection seasons.
- Healthcare Burden: Accounts for 1 in 8 emergency hospital admissions in the UK and similar proportions in the US, representing a major strain on healthcare resources.
2.2 Demographics Table
| Factor | Details | Clinical Significance |
|---|---|---|
| Age | Peak: 65-75 years; rare under 40 | Accumulation of environmental insults and lung aging. |
| Sex | Historically 3:1 (M:F), now nearly 1:1 in many regions | Women may be more susceptible to tobacco smoke damage. |
| Ethnicity | Higher prevalence reported in Caucasian and African American populations | May reflect differences in tobacco exposure and genetics. |
| Geography | High in low-middle income countries (LMICs) | Linked to biomass fuel use and poorly regulated pollution. |
| Socioeconomic | Strong inverse correlation with income/education | Higher exposure to pollutants and lower access to inhalers. |
| Occupation | Mining, construction, and textile workers | Occupational dusts/chemicals contribute 15-20% of cases. |
2.3 Risk Factors Tables
Non-Modifiable Risk Factors:
| Factor | Relative Risk (95% CI) | Mechanism | Clinical Relevance |
|---|---|---|---|
| Age > 65 | RR 3.5 (2.8-4.2) | Cumulative exposure and cellular senescence. | Screen all patients > 40 with risk factors. |
| Alpha-1 Antitrypsin Deficiency | RR 20.0+ (for ZZ phenotype) | Unchecked neutrophil elastase destroys alveolar walls. | Test all COPD diagnosed less than 45 years or with basal emphysema. |
| Female Sex | RR 1.2 (1.1-1.3) | Smaller airways and potential hormonal influences. | Women may have greater susceptibility to smoke. |
| Family History | RR 1.9 (1.5-2.4) | Polygenic risk scores influencing lung development. | Stronger predictor if first-degree relative less than 40 years. |
| Prematurity/Low Birth Weight | RR 1.6 (1.3-1.9) | Failure to reach peak lung function in early adulthood. | Respiratory follow-up for high-risk infants. |
| Genetic Polymorphisms | RR 1.3-2.0 | Variants in MMP-12, HHIP, FAM13A genes | Research area; not yet clinically actionable. |
Modifiable Risk Factors:
| Risk Factor | Relative Risk (95% CI) | Evidence Level | Intervention Impact | Population Attributable Risk |
|---|---|---|---|---|
| Cigarette Smoking | RR 10.0-25.0 | Level 1a | Cessation slows FEV1 decline significantly. | 50-70% of COPD cases |
| Biomass Fuel Exposure | RR 2.2 (1.9-2.5) | Level 1b | Improved ventilation reduces risk in LMICs. | 25-35% in developing countries |
| Occupational Dust | RR 1.4 (1.2-1.7) | Level 2a | PPE and regulation reduce lung function loss. | 15-20% of COPD cases |
| Air Pollution (PM2.5) | RR 1.2 (1.1-1.3) | Level 2a | Public health policy impact on incidence. | 5-10% population level |
| History of Tuberculosis | RR 2.5 (2.1-3.0) | Level 2b | Post-infectious structural lung damage. | Significant in endemic areas |
| Childhood Respiratory Infections | RR 1.8 (1.4-2.3) | Level 2b | Permanent airway remodeling | Prevention via vaccination |
| Cannabis Smoking | RR 2.5 (1.5-4.1) | Level 3 | Similar tar load to cigarettes | Emerging risk factor |
2.4 Protective Factors
- Smoking Cessation: RR 0.4 compared to continued smokers for mortality.
- Physical Activity: Higher baseline activity levels correlate with slower FEV1 decline and fewer hospitalizations.
- Vaccination (Flu/Pneumo): Reduces the risk of severe exacerbations and related mortality by approximately 50%.
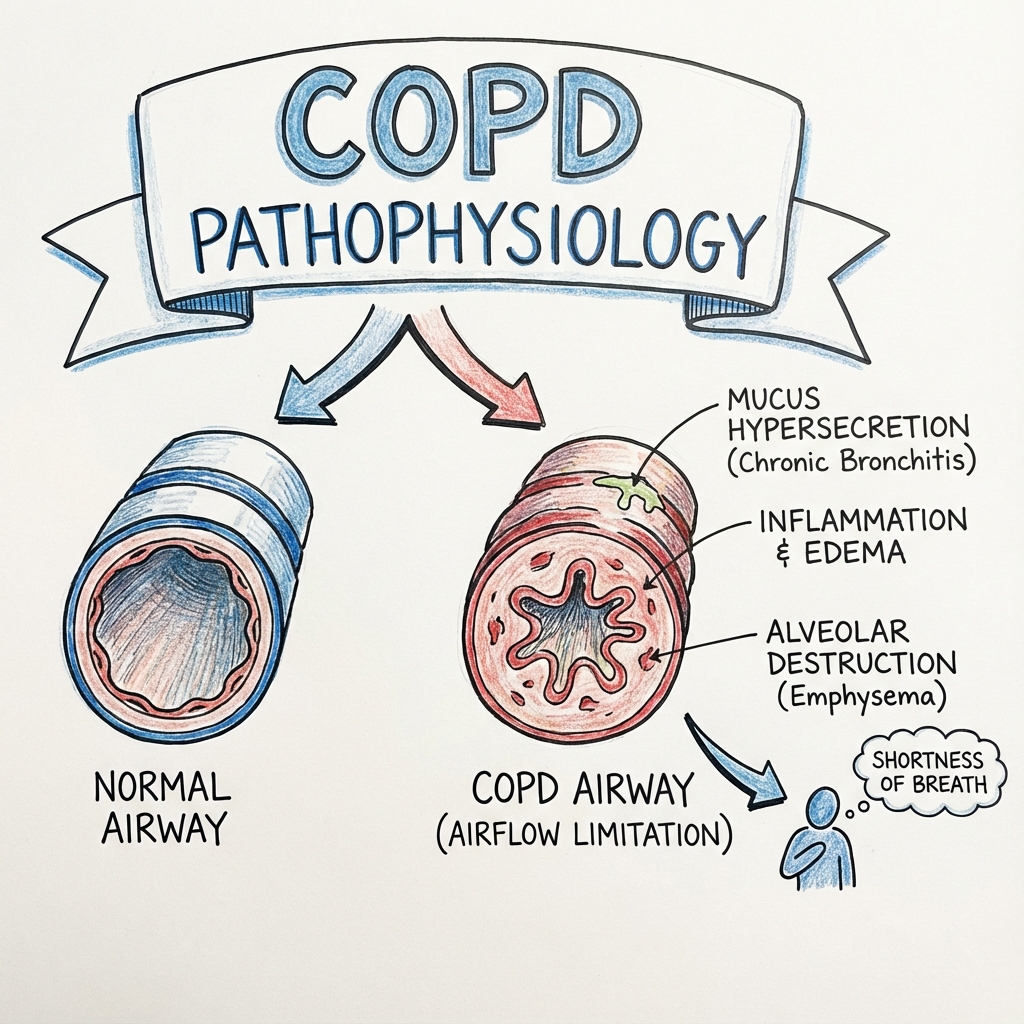
SECTION 3: Pathophysiology
3.1 Step 1: Initiating Event - Oxidative Stress and Epithelial Injury
The process begins with the chronic inhalation of noxious particles, most commonly from cigarette smoke (containing > 7,000 chemicals) or biomass fuels. These irritants introduce reactive oxygen species (ROS) directly into the airway and induce the endogenous production of ROS by epithelial cells and macrophages.
- Molecular Basis: ROS activate the Nuclear Factor-kappa B (NF-κB) pathway and the Mitogen-Activated Protein Kinase (MAPK) cascade.
- Receptor Activation: Pattern Recognition Receptors (PRRs) such as Toll-Like Receptor 4 (TLR4) are activated by damage-associated molecular patterns (DAMPs) from dying cells.
- Cellular Response: Airway epithelial cells undergo squamous metaplasia and lose ciliary function. They begin secreting pro-inflammatory mediators like IL-8 and TNF-α within hours of exposure.
- Susceptibility: Individuals with polymorphisms in antioxidant genes (e.g., Glutathione S-transferase) are more susceptible to this initial oxidative hit.
3.2 Step 2: Early Pathological Changes - Inflammatory Recruitment
The initial epithelial signal triggers a massive recruitment of inflammatory cells into the lung parenchyma and small airways.
- Cellular Infiltration: Unlike asthma (eosinophilic), the inflammation in COPD is predominantly Neutrophilic and CD8+ T-lymphocyte driven.
- Chemokine Gradient: High levels of CXCL8 (IL-8) and LTB4 act as potent chemoattractants for neutrophils.
- Vascular Changes: Increased vascular permeability leads to mucosal edema.
- Mucus Hypersecretion: Inflammatory mediators stimulate Goblet cell hyperplasia and submucosal gland hypertrophy, mediated by the EGFR signaling pathway. This leads to the "Chronic Bronchitis" phenotype.
- Reversibility: At this stage, the process may be partially reversible if the trigger (smoke) is removed, but the inflammatory "memory" often persists.
3.3 Step 3: Established Disease - Protease-Antiprotease Imbalance
As inflammation becomes chronic, the lung environment shifts toward a state of proteolytic dominance, which is the hallmark of emphysema.
- Protease Release: Activated neutrophils and macrophages release Matrix Metalloproteinases (MMP-9, MMP-12) and Neutrophil Elastase.
- Antiprotease Inhibition: Oxidative stress inactivates protective antiproteases like Alpha-1 Antitrypsin (AAT) and Secretory Leukoprotease Inhibitor (SLPI).
- Alveolar Destruction: Elastase breaks down elastin fibers in the alveolar walls, leading to permanent enlargement of airspaces distal to the terminal bronchioles.
- Functional Consequences: Loss of elastic recoil causes the small airways to collapse during expiration, leading to gas trapping and dynamic hyperinflation. This is the primary cause of exertional dyspnea.
3.4 Step 4: Small Airway Remodeling and Fibrosis
Simultaneous with alveolar destruction, the small airways (less than 2mm diameter) undergo structural changes that increase resistance.
- Peribronchiolar Fibrosis: Chronic inflammation triggers TGF-β release, stimulating fibroblasts to deposit collagen around the bronchioles.
- Lumen Narrowing: The combination of wall thickening, luminal mucus plugs, and loss of radial traction (from destroyed surrounding alveoli) leads to fixed airway obstruction.
- Point of No Return: Once significant peribronchiolar fibrosis and alveolar septal loss have occurred, the airflow obstruction becomes irreversible by bronchodilators.
- Vascular Remodeling: Hypoxia-induced pulmonary vasoconstriction and intimal thickening of small pulmonary arteries begin, laying the groundwork for pulmonary hypertension.
3.5 Step 5: Systemic Effects and End-Stage Complications
COPD is no longer viewed as a disease "limited to the lungs" but as a systemic inflammatory syndrome.
- Systemic Inflammation: Pro-inflammatory cytokines (IL-6, TNF-α) spill into the systemic circulation, leading to "cachexia" and skeletal muscle wasting.
- Cor Pulmonale: Chronic hypoxia leads to permanent pulmonary hypertension, which causes Right Ventricular Hypertrophy and eventually right-sided heart failure.
- Metabolic Consequences: Increased risk of osteoporosis, type 2 diabetes, and ischemic heart disease due to shared inflammatory pathways. [10]
- Outcome: Mortality usually results from acute respiratory failure during an exacerbation, cardiovascular events, or the development of bronchogenic carcinoma (favored by the chronic inflammatory milieu).
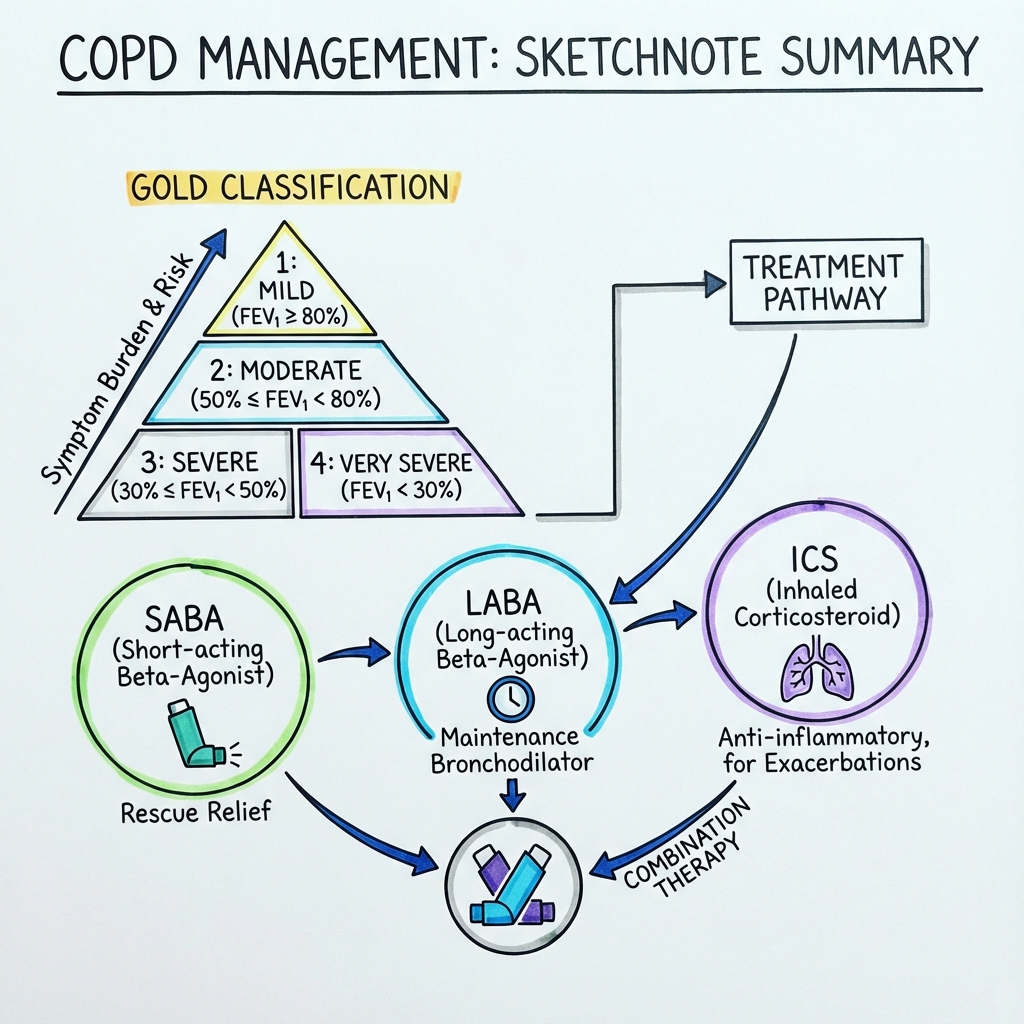
3.6 Classification/Staging (GOLD 2024)
GOLD Spirometric Grading (Based on Post-Bronchodilator FEV1):
| Grade | Severity | FEV1 (% Predicted) | Clinical Context |
|---|---|---|---|
| GOLD 1 | Mild | ≥ 80% | Often asymptomatic or mild cough. |
| GOLD 2 | Moderate | 50% – 79% | Dyspnea on exertion; seeks medical care. |
| GOLD 3 | Severe | 30% – 49% | Significant limitation in QoL; frequent AECOPD. |
| GOLD 4 | Very Severe | less than 30% | Chronic respiratory failure; cor pulmonale. |
GOLD ABE Assessment (Clinical Staging):
The 2023 GOLD update introduced a simplified ABE classification, merging previous Groups C and D into a single Group E for frequent exacerbators. This evidence-based change reflects the understanding that exacerbation history is the primary driver of treatment decisions. [3,4]
| Category | Exacerbations/Year | Symptoms (mMRC or CAT) | Recommended Initial Rx |
|---|---|---|---|
| A | 0 or 1 (no hosp) | mMRC 0-1; CAT less than 10 | A bronchodilator |
| B | 0 or 1 (no hosp) | mMRC ≥ 2; CAT ≥ 10 | LABA + LAMA |
| E | ≥ 2 (or 1 hosp) | Any symptom score | LABA + LAMA (consider ICS if EOS > 300) |
3.7 Anatomical Considerations
- Acinus: The functional unit of the lung; emphysema is classified as centriacinar (smoking-related, upper lobes) or panacinar (AATD-related, lower lobes).
- Small Airways: The "silent zone" of the lung where resistance is high but symptoms are late to appear.
- Diaphragm: Becomes flattened due to hyperinflation, losing its mechanical advantage and forcing reliance on accessory muscles (sternocleidomastoid, scalenes).
3.8 Physiological Considerations
- V/Q Mismatch: Areas of high ventilation/low perfusion (dead space) in emphysema; areas of low ventilation/high perfusion (shunt) in chronic bronchitis.
- Compliance: Increased in emphysema (easy to inflate, hard to deflate); decreased in areas of fibrosis.
- Work of Breathing: Increases 5-10 fold in severe COPD compared to healthy individuals.
3.9 COPD Phenotypes and Personalised Medicine
The recognition that COPD is a heterogeneous disease has led to phenotype-based approaches to management. Recent evidence emphasizes the importance of biomarker-guided therapy, particularly blood eosinophils for ICS decisions. [9,11]
Major Clinical Phenotypes:
| Phenotype | Characteristics | Imaging | Treatment Implications |
|---|---|---|---|
| Emphysema-Predominant ("Pink Puffer") | Thin, barrel chest, minimal cough, severe dyspnea, pursed-lip breathing | CT: bullae, low attenuation areas | Consider LVRS/EBV if upper lobe predominant |
| Chronic Bronchitis-Predominant ("Blue Bloater") | Overweight, chronic productive cough, cyanosis, peripheral edema | CT: bronchial wall thickening | Roflumilast, mucolytics, consider azithromycin |
| Frequent Exacerbator | ≥2 exacerbations/year despite therapy | Variable | Optimize to triple therapy, prophylactic antibiotics |
| ACOS (Asthma-COPD Overlap) | Significant reversibility (> 400mL FEV1), atopy, eosinophilia | Bronchial thickening | Higher dose ICS, consider biologics |
| Rapid Decliner | FEV1 decline > 40mL/year | Progressive changes | Aggressive risk factor modification |
Eosinophilic vs Non-Eosinophilic COPD:
The blood eosinophil count has emerged as a key biomarker for guiding ICS therapy, with multiple trials demonstrating differential response based on eosinophil levels. [9,11]
| Feature | Eosinophilic (Blood Eos > 300) | Non-Eosinophilic (Blood Eos less than 100) |
|---|---|---|
| Prevalence | 30-40% of COPD patients | 35-40% of COPD patients |
| Exacerbation trigger | Often viral | Often bacterial |
| Response to ICS | Good (NNT 3-5) | Poor (NNT > 30) |
| Pneumonia risk on ICS | Moderate | High |
| Preferred therapy | Triple therapy (ICS/LABA/LAMA) | Dual bronchodilator (LABA/LAMA) |
Overlap Syndromes:
- COPD-OSA Overlap: 10-15% prevalence, higher cardiovascular mortality, treat both conditions
- COPD-Bronchiectasis Overlap: 30-50% in severe COPD, more frequent exacerbations, consider long-term macrolides
- COPD-ILD Overlap (CPFE): Combined pulmonary fibrosis and emphysema, severe pulmonary hypertension, poor prognosis
3.10 Molecular Biomarkers and Treatable Traits
Emerging evidence supports a "treatable traits" approach to COPD management, identifying specific molecular phenotypes that respond to targeted therapies. [12,13]
Key Biomarkers in Clinical Use:
| Biomarker | Clinical Utility | Threshold | Treatment Implication |
|---|---|---|---|
| Blood Eosinophils | ICS response predictor | > 300 cells/µL | Strong indication for ICS |
| Fibrinogen | Exacerbation risk | > 350 mg/dL | Increased risk, intensify monitoring |
| CRP | Bacterial infection | > 40 mg/L | Consider antibiotics in AECOPD |
| NT-proBNP | Heart failure differentiation | > 300 pg/mL | Suggests cardiac contribution |
| AAT level | Genetic predisposition | less than 11 µmol/L | Consider augmentation therapy |
Emerging Biomarkers (Research Stage):
- Serum surfactant protein D (SPD): Marker of alveolar damage
- Club cell protein 16 (CC16): Small airway damage marker
- Exhaled volatile organic compounds (VOCs): Metabolic fingerprinting
- MicroRNAs: Regulatory molecules in inflammation
SECTION 4: Clinical Presentation
4.0 Differential Diagnosis
COPD must be distinguished from other causes of chronic dyspnea and airflow limitation. The differential is broad and includes both respiratory and cardiac etiologies.
Key Differential Diagnoses:
| Condition | Distinguishing Features | Diagnostic Test | Management Difference |
|---|---|---|---|
| Asthma | Younger onset, atopy, significant reversibility (> 12% AND > 400mL FEV1), variable symptoms, normal DLCO | Bronchodilator response, FeNO, eosinophilia | ICS first-line, complete reversibility possible |
| Heart Failure | Orthopnea, PND, elevated JVP, S3 gallop, bilateral basal crackles, cardiomegaly on CXR | NT-proBNP > 300 pg/mL, echocardiogram | Diuretics, ACE inhibitors, beta-blockers |
| Bronchiectasis | Copious purulent sputum, digital clubbing, coarse crackles, CT bronchiectasis | HRCT chest (dilated bronchi) | Airway clearance, prolonged antibiotics for exacerbations |
| Interstitial Lung Disease | Fine bi basal inspiratory crackles, restrictive pattern on PFTs, reduced DLCO | HRCT (reticular pattern, honeycombing) | Antifibrotics, avoid oxygen unless hypoxemic |
| Pulmonary Embolism | Acute onset, pleuritic chest pain, hemoptysis, risk factors for VTE | D-dimer, CTPA | Anticoagulation |
| Lung Cancer | Hemoptysis, weight loss, clubbing, CXR mass/nodule | CT chest, biopsy | Surgery/chemotherapy/radiotherapy |
| Tuberculosis | Night sweats, weight loss, hemoptysis, apical infiltrates, risk factors | Sputum AFB, CXR apical disease | Anti-TB therapy for 6 months |
| Obesity Hypoventilation | BMI > 30, hypercapnia at rest, normal spirometry | ABG showing hypercapnia, sleep study | Weight loss, CPAP/BiPAP |
| Panic Disorder | Acute episodic dyspnea, hyperventilation, normal examination and spirometry | Clinical diagnosis, psych assessment | CBT, SSRIs |
Red Flags Suggesting Alternative Diagnosis:
- Age less than 40 years with no smoking history → consider asthma, bronchiectasis, CF
- Hemoptysis → rule out lung cancer, TB, bronchiectasis
- Significant weight loss → malignancy, TB
- Bilateral ankle edema out of proportion to dyspnea → heart failure
- Finger clubbing → lung cancer, bronchiectasis, ILD (NOT seen in COPD alone)
- Inspiratory crackles → ILD, heart failure, pneumonia
- Unilateral reduced breath sounds → pneumothorax, effusion, collapse
4.1 Symptoms
| Symptom | Frequency | Character | Duration | Associations |
|---|---|---|---|---|
| Dyspnea | > 90% | Progressive, persistent, worse with exercise | Years | Activity limitation |
| Chronic Cough | 70-80% | May be intermittent; often "smoker's cough" | > 3 months/year | Sputum production |
| Sputum Production | 60-70% | Mucoid; becomes purulent during AECOPD | Chronic | Morning peak |
| Wheezing | 40-50% | Expiratory; varies in intensity | Episodic | Chest tightness |
| Fatigue | 50-60% | Generalized tiredness | Constant | Depression |
| Weight Loss | 15-20% | Unintentional; "pulmonary cachexia" | Months | Severe emphysema |
| Ankle Swelling | 10-15% | Pitting edema | Persistent | Cor pulmonale |
| Morning Headache | 5-10% | Dull, bifrontal | On awakening | Hypercapnia (CO2 retention) |
4.2 Signs
General Examination:
- Tachypnea (RR > 20)
- Use of accessory muscles (sternocleidomastoid)
- Pursed-lip breathing (to maintain PEP and prevent airway collapse)
- Central or peripheral cyanosis
- Low BMI (in emphysema-predominant)
System-Specific Signs:
| Sign | How to Elicit | Significance | Sensitivity/Specificity |
|---|---|---|---|
| Barrel Chest | Visual inspection (increased AP diameter) | Severe hyperinflation | 50% / 90% |
| Hyper-resonance | Percussion of chest wall | Gas trapping/Emphysema | 40% / 85% |
| Decreased Breath Sounds | Auscultation | Reduced airflow/Alveolar loss | 60% / 80% |
| Coarse Crackles | Auscultation (early inspiratory) | Secretions in large airways | 40% / 70% |
| Hoover's Sign | Observe lower rib cage on inspiration | Diaphragmatic flattening | 30% / 95% |
| Raised JVP | Inspection of neck veins | Right heart failure (Cor pulmonale) | 50% / 90% |
4.3 Red Flags
[!CAUTION] RED FLAGS — Seek immediate help if:
- Sudden worsening of shortness of breath: Indicates AECOPD, PE, or Pneumothorax.
- New onset confusion/lethargy: Sign of severe hypercapnia and respiratory acidosis.
- Chest pain: May indicate comorbid myocardial infarction or pleurisy.
- Inability to complete a sentence: Indicates severe respiratory distress.
- Fever and purulent sputum: Suggests acute bacterial infection/pneumonia.
- Hemoptysis: Always requires exclusion of bronchogenic carcinoma.
- Cyanosis of lips or fingernails: Indicates life-threatening hypoxemia.
4.4 Clinical Scoring
- mMRC Dyspnea Scale: 0 (only with strenuous exercise) to 4 (too dyspneic to leave the house).
- CAT (COPD Assessment Test): 8-item questionnaire; scores > 10 indicate high symptom burden.
- BODE Index: Predicts mortality using BMI, Obstruction (FEV1), Dyspnea (mMRC), and Exercise (6-minute walk test).
SECTION 5: Clinical Examination
5.1 Structured Approach
- Inspection: Assess for "Pink Puffer" (thin, tachypneic, pursed lips) vs "Blue Bloater" (overweight, cyanotic, edematous) phenotypes. Look for nicotine staining on fingers.
- Vitals: Check SpO2 (target 88-92% if CO2 retainer), RR, and HR (tachycardia common due to beta-agonist use or hypoxia).
- Palpation: Trachea (usually midline), Apex beat (may be impalpable due to hyperinflation). Check for peripheral edema.
- Percussion: Generally hyper-resonant; loss of cardiac and hepatic dullness due to lung expansion.
- Auscultation: Prolonged expiratory phase; quiet breath sounds; expiratory wheeze.
5.2 Special Tests Table
| Test Name | Technique | Positive Finding | Sensitivity | Specificity | Clinical Use |
|---|---|---|---|---|---|
| Forced Expiratory Time | Time the duration of a full forced expiration | > 6 seconds | 70% | 85% | Bedside screening for obstruction |
| 6-Minute Walk Test | Distance walked on flat ground in 6 min | less than 350 meters | High | Moderate | Prognosticating/Pulmonary rehab |
| Schamroth Window | Oppose dorsal surfaces of distal phalanges | Loss of diamond window | Low | High | Checking for clubbing (rare in COPD) |
| Jugular Venous Distension | Patient at 45 degrees, observe IJV | > 3cm above sternal angle | Moderate | High | Screening for Cor Pulmonale |
| Asterixis | Extend wrists with fingers spread | Brief, jerky flapping | Moderate | High | Detecting CO2 retention/Encephalopathy |
| Pitting Edema | Press over medial malleolus for 5s | Persistent indentation | Moderate | Moderate | Assessing right heart failure |
5.3 Examination by Severity
- Mild: Physical exam may be completely normal.
- Moderate: Prolonged expiration and wheezing on exertion.
- Severe: Barrel chest, Hoover's sign, use of accessory muscles, and distant heart sounds.
SECTION 6: Investigations
6.1 Bedside Tests
| Test | Expected Finding | Purpose | Urgency |
|---|---|---|---|
| Pulse Oximetry | 88-94% | Baseline oxygenation | Immediate |
| ECG | P-pulmonale, RAD, RVH | Screen for Cor Pulmonale | Immediate |
| Sputum Culture | Neutrophils, H. influenzae | Guide antibiotic therapy | Within 24h |
| Peak Flow | Reduced (but less useful than FEV1) | Monitoring, not diagnosis | Routine |
| Blood Glucose | Elevated (if on steroids) | Monitor steroid side effects | Routine |
6.2 Laboratory Tests
| Test | Expected Finding | Purpose | Turnaround |
|---|---|---|---|
| FBC | Polycythemia or Anemia | Chronic hypoxia or chronic disease | 4 hours |
| Eosinophil Count | > 300 cells/µL | Guide ICS therapy | 4 hours |
| Alpha-1 Antitrypsin | less than 11 µmol/L | Screen for genetic deficiency | 3-5 days |
| ABG | pH less than 7.35, pCO2 > 45 | Assess respiratory failure/acidosis | 15 mins |
| CRP | Elevated in AECOPD | Assess for bacterial infection | 1 hour |
| U&Es | Hypokalemia (if on SABA) | Monitor diuretic/beta-agonist SEs | 2 hours |
| NT-proBNP | Elevated | Differentiate from Heart Failure | 4 hours |
| Theophylline Level | Target 10-20 mcg/mL | If patient is on methylxanthines | 12 hours |
6.3 Imaging
| Modality | Key Findings | Indication | Sensitivity/Specificity |
|---|---|---|---|
| Chest X-ray | Flattened diaphragm, hyperlucency | Rule out pneumonia/pneumothorax | 50% / 90% |
| HRCT Chest | Bullae, "dirty" lungs, airway thickening | Assess emphysema phenotype | 95% / 95% |
| Echocardiogram | RV dilation, TR jet | Assess for Pulmonary Hypertension | 70% / 80% |
| Ventilation/Perfusion | Mismatched defects | Rule out PE during AECOPD | 80% / 80% |
| DXA Scan | Reduced bone mineral density | Long-term steroid use monitoring | N/A |
6.4 Diagnostic Criteria
Diagnosis is confirmed by Spirometry:
- Requirement: Post-bronchodilator FEV1/FVC less than 0.70. [8]
- Procedure: Administer 400 mcg Salbutamol; repeat spirometry after 15-20 minutes.
- Alternative: FEV1/FVC < Lower Limit of Normal (LLN) to avoid overdiagnosis in elderly.
Detailed Spirometry Interpretation:
| Parameter | Normal | Mild COPD | Moderate | Severe | Very Severe |
|---|---|---|---|---|---|
| FEV1/FVC | > 0.70 | less than 0.70 | less than 0.70 | less than 0.70 | less than 0.70 |
| FEV1 % predicted | ≥80% | ≥80% | 50-79% | 30-49% | less than 30% |
| Clinical significance | - | Often asymptomatic | Dyspnea on exertion | Severely limited | Respiratory failure risk |
Post-Bronchodilator Testing Protocol:
- Baseline spirometry performed (pre-bronchodilator)
- Administer 400 mcg salbutamol via MDI with spacer OR 2.5mg nebulized
- Wait 15-20 minutes for bronchodilator effect
- Repeat spirometry
- Significant reversibility: > 12% AND > 200mL increase in FEV1 (suggests asthma component)
- Diagnosis requires FEV1/FVC less than 0.70 POST-bronchodilator
Spirometry Quality Criteria:
- Minimum 3 acceptable maneuvers
- Variation between best two FEV1 and FVC values less than 150mL
- Good start of test (back-extrapolated volume less than 5% of FVC or 150mL)
- No early termination
- Peak flow achieved early in exhalation
When to Consider LLN Instead of Fixed 0.70 Ratio:
- Age > 65 years (avoids overdiagnosis)
- Age less than 45 years (avoids underdiagnosis)
- Never-smokers with borderline values
- Patients with diagnostic uncertainty
SECTION 7: Management
⚠️ MANDATORY: ASCII MANAGEMENT ALGORITHM
┌─────────────────────────────────────────────────────────────┐
│ STABLE COPD MANAGEMENT ALGORITHM │
└─────────────────────────────────────────────────────────────┘
│
▼
┌───────────────────────────────┐
│ DIAGNOSIS (FEV1/FVC less than 0.7) │
│ • Smoking Cessation Advice │
│ • Vaccinations (Flu/Pneumo) │
└───────────────────────────────┘
│
▼
┌───────────────────────────────┐
│ ASSESS SYMPTOMS (mMRC/CAT)│
│ & EXACERBATION HISTORY │
└───────────────────────────────┘
│
┌─────────────────────┼─────────────────────┐
▼ ▼ ▼
┌───────────────┐ ┌───────────────┐ ┌───────────────┐
│ GROUP A │ │ GROUP B │ │ GROUP E │
│ (Low symptoms,│ │(High symptoms,│ │(Frequent │
│ 0-1 AECOPD) │ │ 0-1 AECOPD) │ │ exacerbations)│
└───────────────┘ └───────────────┘ └───────────────┘
│ │ │
▼ ▼ ▼
┌───────────────┐ ┌───────────────┐ ┌───────────────┐
│ Single Broncho│ │ LABA + LAMA │ │ LABA + LAMA │
│ (LAMA or LABA)│ │ Combination │ │ (Initial Rx) │
└───────────────┘ └───────────────┘ └───────────────┘
│ │ │
▼ ▼ ▼
┌─────────┐ ┌─────────┐ ┌────────────────┐
│Response?│ │Response?│ │Blood Eos > 300? │
└────┬────┘ └────┬────┘ └───────┬────────┘
Yes │ No Yes │ No Yes │ No
│ │ │
▼ ▼ ▼
┌───────────────┐ ┌───────────────┐ ┌───────────────┐
│ MAINTAIN │ │ ESCALATE │ │ADD ICS (TRIPLE)│
│ • Review tech │ │ • To LAMA+LABA│ │ • ICS+LAMA+LABA│
│ • Pulmonary │ │ • Review tech │ │ • Review QoL │
│ Rehab │ │ • Pulm Rehab │ │ • Monitor Pneum│
└───────────────┘ └───────────────┘ └───────────────┘
│
▼
┌───────────────────────────────┐
│ ADVANCED THERAPIES │
│ • Long-term Oxygen (LTOT) │
│ • Roflumilast (if FEV1 less than 50%) │
│ • Azithromycin (Prophylaxis) │
│ • Lung Volume Reduction (LVR) │
└───────────────────────────────┘
7.1 Emergency/Acute Management (AECOPD)
An acute exacerbation of COPD is defined as an acute worsening of respiratory symptoms that results in additional therapy. Exacerbations are the leading cause of hospitalization, healthcare costs, and mortality in COPD. Recent guidelines emphasize early recognition and prompt treatment initiation. [14]
AECOPD Severity Classification:
| Severity | Clinical Features | Management Setting | Mortality Risk |
|---|---|---|---|
| Mild | Increased SABA use, no change in activities | Outpatient | less than 1% |
| Moderate | SABA increase + antibiotic or oral steroid, reduced activities | Outpatient or ED | 2-5% |
| Severe | Hospital admission required, significant breathlessness at rest | Hospital ward | 8-15% |
| Life-Threatening | Respiratory failure requiring NIV/intubation or ICU | ICU | 25-30% |
Immediate Actions (ABCDE approach):
A - Airway:
- Ensure patency
- Suction secretions if excessive
- Consider oro/nasopharyngeal airway if reduced GCS
- Assess for upper airway obstruction (stridor, tumor)
B - Breathing:
- Controlled oxygen therapy - This is CRITICAL
- Use Venturi mask 24% or 28% to achieve SpO2 88-92%
- AVOID high-flow oxygen - causes worsening hypercapnia
- If patient brought in on high-flow, REDUCE immediately
- Recheck ABG after 30-60 minutes of controlled oxygen
- Nebulized bronchodilators:
- Salbutamol 5mg (can repeat every 20-30 min initially)
- Ipratropium bromide 500mcg (can repeat every 4-6 hours)
- Can be driven by air + supplemental nasal O2 if hypoxemic
- If severely hypercapnic, use air-driven nebulizers only
- Respiratory rate and pattern assessment
- RR > 30 = severe
- Paradoxical breathing = exhaustion, impending respiratory failure
- "Silent chest" = life-threatening bronchospasm
C - Circulation:
- IV access (18G minimum)
- Fluid balance: judicious fluids only (avoid overload in cor pulmonale)
- Monitor for arrhythmias (AF common, triggered by hypoxia and beta-agonists)
- ECG: look for RVH (p-pulmonale, RAD), ischemia, arrhythmias
- Blood pressure: hypotension suggests sepsis or PE
D - Disability:
- GCS assessment (confusion = hypercapnic encephalopathy)
- ABG mandatory if acidotic or pH unknown:
- pH less than 7.35 = acidemia, likely NIV needed
- pH less than 7.25 = severe, consider ICU
- pCO2 > 45 mmHg = hypercapnia
- Compare to previous ABGs if available (chronic vs acute retention)
- Check for asterixis (CO2 flap) - bilateral hand tremor
E - Exposure:
- Temperature (fever suggests infection)
- Check calves for DVT (PE common precipitant)
- Look for peripheral edema (cor pulmonale)
- Check for cachexia, muscle wasting
Emergency Medications:
| Drug | Dose | Route | Timing | Evidence | Monitoring |
|---|---|---|---|---|---|
| Prednisolone | 30-40mg | PO | Daily x 5 days | REDUCE trial (5 days non-inferior to 14 days) [15] | Blood glucose, BP |
| Hydrocortisone | 100mg | IV | QDS x 48h then switch to oral | If unable to take oral | As above |
| Amoxicillin | 500mg | PO | TDS x 5 days | If purulent sputum (Anthonisen criteria) [16] | Allergy history |
| Doxycycline | 100mg | PO | BD x 5 days | Alternative to amoxicillin | Photosensitivity |
| Clarithromycin | 500mg | PO/IV | BD x 5-7 days | If Mycoplasma/Legionella suspected | QT interval |
| Co-amoxiclav | 625mg | PO | TDS x 5 days | If previous isolates resistant | C. diff risk |
| Aminophylline | 5mg/kg (max 500mg) | IV | Loading over 20 min, then infusion | ONLY if poor response to nebulizers | ECG, levels, K+ |
Anthonisen Criteria for Antibiotics in AECOPD:
Type 1 (all 3 present) - TREAT: Increased dyspnea + increased sputum volume + increased sputum purulence [16] Type 2 (2 of 3) - TREAT: As above Type 3 (1 of 3 + one of): fever, URI, wheeze - CONSIDER: Clinical judgment
Common Pathogens and Antibiotic Choice:
| Pathogen | Frequency | First-Line | Alternative |
|---|---|---|---|
| H. influenzae | 30-40% | Amoxicillin, doxycycline | Co-amoxiclav, macrolide |
| S. pneumoniae | 20-30% | Amoxicillin | Macrolide, cephalosporin |
| M. catarrhalis | 10-20% | Co-amoxiclav, doxycycline | Macrolide, fluoroquinolone |
| Viral (Rhinovirus, Influenza) | 30-40% | Supportive (consider oseltamivir for flu) | - |
| P. aeruginosa | 5-10% (in severe COPD) | Ciprofloxacin | Piperacillin-tazobactam |
Escalation Criteria (Call for ICU/NIV):
Within 1 hour if ANY of:
- pH less than 7.35 on controlled oxygen
- pCO2 > 45 mmHg with acidosis
- Deteriorating mental status
- Respiratory rate > 30 with exhaustion
- Inability to maintain SpO2 > 85% despite high FiO2
Immediate Intubation Criteria:
- Cardiac/respiratory arrest
- GCS ≤8 (unable to protect airway)
- Severe hemodynamic instability (SBP less than 90 despite fluids)
- Worsening acidosis despite 2 hours NIV (pH less than 7.20)
- Multi-organ failure
7.2 Conservative Management
Smoking Cessation - THE MOST IMPORTANT INTERVENTION:
Smoking cessation is the ONLY intervention that slows FEV1 decline and reduces mortality at ALL COPD stages. Every clinical encounter should address smoking.
The "5 A's" Framework:
| Step | Action | Detail | Documentation |
|---|---|---|---|
| Ask | About tobacco use | "Do you currently smoke or use any tobacco products?" | Record pack-years, quit attempts |
| Advise | Urge to quit | Clear, personalized advice: "Quitting smoking is the most important thing you can do for your lungs" | Advice given, date |
| Assess | Willingness to quit | "On a scale of 0-10, how ready are you to quit in the next 30 days?" | Readiness score |
| Assist | Aid in quitting | Pharmacotherapy + behavioral support | Prescriptions, referrals |
| Arrange | Follow-up contact | Within 1 week of quit date, then ongoing | F/U appointments |
Evidence-Based Pharmacotherapy for Smoking Cessation:
| Medication | Mechanism | Dose | Duration | NNT | Contraindications | Side Effects |
|---|---|---|---|---|---|---|
| Varenicline | Partial nicotinic agonist | 0.5mg OD x3d, then 0.5mg BD x4d, then 1mg BD | 12 weeks (24 if needed) | 10 | Seizures, severe psychiatric illness | Nausea, abnormal dreams, mood changes |
| Bupropion SR | Norepinephrine/dopamine reuptake inhibitor | 150mg OD x3d, then 150mg BD | 7-12 weeks | 17 | Seizures, eating disorders, MAOIs | Insomnia, dry mouth, seizure risk |
| Nicotine Replacement (patch) | Nicotine delivery without combustion | 21mg/24h x 6wks, then taper | 8-12 weeks | 14 | Recent MI, unstable angina | Skin irritation, insomnia |
| Nicotine Replacement (gum/lozenge) | As above | 2-4mg PRN (8-12 pieces/day) | 12 weeks | 14 | As above | Jaw ache, hiccups, dyspepsia |
| Combination NRT | Higher quit rates | Patch + gum/lozenge | 12 weeks | 8 | As above | Combined side effects |
Comparative Efficacy:
- Varenicline: 22% quit rate at 1 year (Best efficacy)
- Combination NRT: 20% quit rate
- Bupropion: 18% quit rate
- Single NRT: 16% quit rate
- Behavioral support alone: 12% quit rate
- No intervention: 5% quit rate
Pulmonary Rehabilitation:
Pulmonary rehabilitation is a comprehensive intervention combining exercise training, education, and behavioral modification. It is a Grade A recommendation in all COPD guidelines with Level 1a evidence for efficacy. [17,18]
Components of Comprehensive PR Program:
| Component | Duration/Frequency | Content | Evidence Level |
|---|---|---|---|
| Exercise Training | 6-8 weeks, 2-3x/week | Aerobic + resistance training | 1a |
| Education | Throughout program | Disease understanding, inhaler technique | 1a |
| Nutritional Assessment | Initial + ongoing | Weight optimization, protein supplementation | 1b |
| Psychological Support | As needed | Depression/anxiety screening and treatment | 1b |
| Self-Management | Ongoing | Action plans, exacerbation recognition | 1a |
Indications for Pulmonary Rehabilitation:
- Primary: Any COPD patient with mMRC ≥2 OR CAT ≥10
- Post-exacerbation: Within 1 month of hospital discharge for AECOPD (reduces readmissions) [17]
- Pre-operative: Before lung surgery (improves outcomes)
Exercise Prescription:
| Exercise Type | Intensity | Duration | Frequency | Progression |
|---|---|---|---|---|
| Aerobic (walking/cycling) | 60-80% peak work rate | 20-30 minutes | 3-5x/week | Increase by 10% weekly |
| Resistance Training | 60-70% 1-RM | 8-12 reps x 2-3 sets | 2-3x/week | Increase weight when > 12 reps easy |
| Inspiratory Muscle Training | 30% MIP | 15 minutes | Daily | Increase load weekly |
Outcomes from PR (Cochrane Meta-Analysis): [17,18]
- 6-Minute Walk Distance: Mean increase 44 meters (clinically significant: ≥30m)
- Quality of Life (SGRQ): Mean improvement 7 units (MCID: 4 units)
- Dyspnea (mMRC): Improvement by 0.8 units
- Exacerbations: 40% reduction in hospitalizations if done post-AECOPD
Vaccinations:
- Annual Influenza vaccination reduces exacerbation risk
- Pneumococcal vaccination (PPSV23 and PCV13)
- COVID-19 vaccination
- Tdap (for Pertussis protection)
Nutrition:
- High-protein diet for cachectic patients (BMI less than 21)
- Small frequent meals to avoid diaphragmatic pressure
- Nutritional supplementation if BMI less than 21
7.3 Medical Management
First-Line Drug Table:
| Drug | Class | Dose | Device | Frequency | Duration | Key Side Effects | Monitoring | Evidence |
|---|---|---|---|---|---|---|---|---|
| Tiotropium | LAMA | 18 mcg or 5 mcg | HandiHaler/Respimat | OD | Chronic | Dry mouth, urinary retention | Glaucoma risk, voiding | UPLIFT trial |
| Salmeterol | LABA | 50 mcg | DPI/MDI | BD | Chronic | Tremor, palpitations, hypokalemia | HR, K+, ECG | TORCH trial |
| Umeclidinium | LAMA | 62.5 mcg | Ellipta DPI | OD | Chronic | Dry mouth, constipation | Voiding issues | EMAX trial |
| Formoterol | LABA | 12 mcg | DPI/MDI | BD | Chronic | Tachycardia, muscle cramps | ECG if cardiac history | FLAME trial [19] |
| Indacaterol | Ultra-LABA | 75-150 mcg | Breezhaler | OD | Chronic | Cough post-inhale, headache | Baseline FEV1 | SPARK trial |
| Glycopyrronium | LAMA | 50 mcg | Breezhaler | OD | Chronic | Dry mouth, nasopharyngitis | UTI symptoms | GLOW trials |
| Olodaterol | Ultra-LABA | 5 mcg | Respimat | OD | Chronic | Nasopharyngitis | Blood glucose | TONADO trials |
Fixed-Dose Combination Inhalers (LABA/LAMA):
The FLAME trial demonstrated superiority of LABA/LAMA over ICS/LABA for exacerbation prevention in non-eosinophilic COPD, fundamentally changing treatment algorithms. [19]
| Product | Components | Dose | Device | Frequency | NNT (Exacerbation Prevention) |
|---|---|---|---|---|---|
| Anoro Ellipta | Umeclidinium/Vilanterol | 62.5/25 mcg | Ellipta | OD | 8-12 vs LAMA alone |
| Ultibro Breezhaler | Glycopyrronium/Indacaterol | 50/110 mcg | Breezhaler | OD | 10-15 vs LABA alone |
| Duaklir | Aclidinium/Formoterol | 340/12 mcg | Genuair | BD | 12-18 vs monotherapy |
| Spiolto Respimat | Tiotropium/Olodaterol | 5/5 mcg | Respimat | OD | 9-14 vs tiotropium |
| Bevespi Aerosphere | Glycopyrronium/Formoterol | 9/4.8 mcg | MDI | BD | Similar to other LABA/LAMA |
Second-Line/Alternative/Add-on Drugs:
| Drug | Indication | Dose | Route | Evidence | Contraindications | NNT/NNH |
|---|---|---|---|---|---|---|
| Fluticasone | Triple therapy (EOS > 300) | 250-500 mcg BD | Inhaled | IMPACT trial [5] | Recurrent pneumonia | NNT 15 for exacerbations |
| Budesonide | Triple therapy | 320 mcg BD | Inhaled | ETHOS trial [6] | Active TB | NNT 12 for exacerbations |
| Roflumilast | Chronic bronchitis, FEV1 less than 50% | 500 mcg OD | PO | REACT trial [20] | Severe liver disease | NNT 8 for exacerbations |
| Azithromycin | Frequent exacerbators (> 2/year) | 250mg 3x/week | PO | COLUMBUS trial [21] | QT > 450ms, hearing loss | NNT 3 for exacerbations |
| Carbocisteine | High sputum burden | 750mg TDS | PO | PEACE trial | Active peptic ulcer | NNT 20 for exacerbations |
| Theophylline | Refractory symptoms | 200-400mg BD | PO | Limited evidence | Arrhythmias, seizures | Not for exacerbations |
| N-Acetylcysteine (NAC) | Antioxidant therapy | 600mg BD | PO | PANTHEON trial | Limited benefit | NNT > 30 |
Triple Therapy Evidence Summary:
The landmark IMPACT and ETHOS trials revolutionized COPD management by demonstrating mortality benefits with triple therapy (ICS/LABA/LAMA):
- IMPACT Trial (2018): 10,355 patients, triple therapy (fluticasone/umeclidinium/vilanterol) reduced moderate-to-severe exacerbations by 15% vs LABA/LAMA and 25% vs ICS/LABA [5]
- ETHOS Trial (2020): First trial to show all-cause mortality reduction with triple therapy (budesonide/glycopyrrolate/formoterol), 24% mortality reduction vs LABA/LAMA [6]
- FLAME Trial (2016): Demonstrated LABA/LAMA superior to ICS/LABA for exacerbation prevention in non-eosinophilic COPD [19]
These trials established triple therapy as the standard of care for Group E patients with elevated eosinophils, representing a paradigm shift in COPD management.
Eosinophil-Guided ICS Therapy:
Blood eosinophil count has emerged as a key biomarker for predicting ICS response, with multiple post-hoc analyses and meta-analyses confirming differential treatment effects. [9,11]
| Blood Eosinophil Count | ICS Recommendation | Evidence | Clinical Action |
|---|---|---|---|
| less than 100 cells/µL | Avoid or withdraw ICS | Minimal benefit, increased pneumonia risk | Use LABA/LAMA only |
| 100-300 cells/µL | Consider if ≥2 exacerbations/year | Modest benefit | Individualize decision |
| > 300 cells/µL | Strongly recommend ICS | Significant exacerbation reduction | Triple therapy if symptomatic |
| > 400 cells/µL | Very strong indication | NNT 2-3 for preventing exacerbations | Consider early escalation |
7.4 Surgical Management
Indications for Surgery:
- Severe emphysema (predominantly upper lobe)
- FEV1 20-45% predicted
- DLCO > 20% predicted
- Completion of pulmonary rehabilitation
- Six-minute walk distance > 140m
Procedure Description:
- Lung Volume Reduction Surgery (LVRS): Removal of the most diseased hyperinflated lung tissue to allow better expansion of healthier tissue. [22]
- Endobronchial Valves (EBV): One-way valves placed via bronchoscopy to collapse hyperinflated lobes (less invasive than LVRS). [22]
- Lung Transplantation: Considered in GOLD 4 patients with BODE score 7-10.
7.5 Long-Term Oxygen Therapy (LTOT)
Long-term oxygen therapy is one of only two pharmacological interventions (along with smoking cessation) that has proven mortality benefit in COPD.
Indications for LTOT (BTS/ATS Guidelines):
| Criterion | PaO2 | SpO2 | Additional Requirements |
|---|---|---|---|
| Absolute | ≤7.3 kPa (55 mmHg) | ≤88% | At rest, on room air, stable state |
| Relative | 7.3-8.0 kPa (55-60 mmHg) | 88-92% | PLUS evidence of: |
| - Pulmonary hypertension (echo) | |||
| - Peripheral edema (cor pulmonale) | |||
| - Polycythemia (Hct > 55%) | |||
| Nocturnal | SpO2 less than 90% for > 30% sleep time | - | Consider if daytime borderline |
| Exertional | SpO2 less than 88% on 6MWT | - | Ambulatory oxygen if benefits demonstrated |
Evidence Base for LTOT:
- MRC Trial (1981): > 15 hours/day oxygen improved survival vs no oxygen in hypoxemic COPD
- NOTT Trial (1980): 24-hour oxygen superior to 12-hour nocturnal oxygen for survival
- LOTT Trial (2016): NO mortality benefit for moderate desaturation (SpO2 89-93%) - critical for avoiding over-prescription
LTOT Prescription Details:
| Parameter | Specification | Rationale |
|---|---|---|
| Flow rate | Titrate to achieve PaO2 > 8 kPa (60 mmHg) or SpO2 > 90% | Usually 1-4 L/min at rest |
| Duration | ≥15 hours/day (ideally 24h if tolerated) | Mortality benefit requires sustained use |
| Delivery method | Nasal cannula preferred | Better compliance than masks |
| Reassessment | 60-90 days after initiation | Confirm stable hypoxemia, not transient AECOPD |
| Ambulatory O2 | If desaturation less than 88% on 6MWT | Improved exercise capacity and QoL |
LTOT Monitoring:
- ABG or pulse oximetry at rest and with exertion every 6-12 months
- Check compliance (hour meter on concentrator)
- Screen for cor pulmonale (JVP, edema, echo)
- Assess for polycythemia (FBC)
Contraindications/Cautions:
- Absolute: Continued smoking (fire risk)
- Relative: Poorly controlled psychiatric illness, cognitive impairment preventing safe use
- Caution: Presence of bullae (theoretical pneumothorax risk)
7.6 Non-Invasive Ventilation (NIV)
NIV is the cornerstone of management for acute hypercapnic respiratory failure in AECOPD, with robust mortality and intubation reduction benefits.
Acute NIV for AECOPD (Hospital Setting):
Indications:
- Respiratory acidosis: pH 7.25-7.35 AND pCO2 > 45 mmHg (6 kPa)
- Persistent hypercapnia despite optimal medical therapy (1 hour trial)
- Clinical signs of respiratory distress despite controlled oxygen
Contraindications:
- Absolute: Cardiac/respiratory arrest, facial trauma/burns, upper GI surgery
- Relative: Vomiting, confusion/agitation, inability to protect airway, copious secretions
NIV Settings (BiPAP Mode):
| Parameter | Initial Setting | Titration | Target |
|---|---|---|---|
| IPAP | 12-14 cmH2O | Increase by 2 cmH2O every 10-15 min | 18-25 cmH2O |
| EPAP | 4-5 cmH2O | Usually keep at 4-6 | 4-6 cmH2O |
| Backup rate | 12-15 breaths/min | Adjust to patient comfort | Patient-triggered ideally |
| FiO2 | Titrate to SpO2 88-92% | Minimize while maintaining target | SpO2 88-92% |
NIV Success Criteria (After 1-2 hours):
- Improvement in pH (> 7.30 or increase by ≥0.03)
- Reduction in PaCO2
- Reduction in respiratory rate
- Improvement in mental status
- Patient tolerance
NIV Failure Criteria (Intubation Required):
- Worsening acidosis (pH less than 7.25) despite 2 hours NIV
- Deteriorating mental status (GCS less than 8)
- Cardiovascular instability
- Unable to tolerate NIV
- Worsening hypoxemia
Evidence for Acute NIV:
- Cochrane Review (2017): NIV reduces mortality (NNT 10), intubation (NNT 4), and hospital stay in AECOPD
- NIV should be initiated within 60 minutes of meeting criteria
- Failure rate approximately 20-30%
Chronic Domiciliary NIV:
Indications (Evolving Evidence):
- Persistent hypercapnia (pCO2 > 55 mmHg/7.3 kPa) despite optimal therapy
- Recurrent hospitalizations for hypercapnic AECOPD
- Overlap syndrome (OSA + COPD)
Chronic NIV Settings:
- Higher pressures than acute (IPAP 20-30 cmH2O, EPAP 4-6 cmH2O)
- Target: normalize or reduce PaCO2, improve sleep quality
- Use requires specialist respiratory sleep center
7.7 Disposition and Follow-Up
Hospital Admission Criteria (DECAF Score Validation):
| Criterion | Assessment | Threshold | Action |
|---|---|---|---|
| Severe Dyspnea | At rest, unable to complete sentences | RR > 30 | Admit |
| Acute Confusion | AMT less than 8 or new confusion | AVPUAlert only (A) | Admit to HDU |
| Acidemia | ABG pH | less than 7.35 | Admit, consider NIV |
| Atrial Fibrillation | New onset or rapid rate | HR > 120 | Admit for monitoring |
| Peripheral Edema | New or worsening | Significant | Admit (? cor pulmonale) |
| Comorbidities | IHD, HF, DM complications | Unstable | Admit |
| Social Factors | Lives alone, poor support | Unable to cope | Admit or acute care at home |
| Failed Outpatient | No improvement after 48h oral therapy | - | Admit |
DECAF Score for Mortality Risk:
| Component | Points | Assessment |
|---|---|---|
| Dyspnea (eMRCD 5a or 5b) | 1 | Too breathless to leave room or dress |
| Eosinopenia (less than 0.05 x10⁹/L) | 1 | Blood eosinophils less than 50/µL |
| Consolidation on CXR | 1 | Infiltrate present |
| Acidemia (pH less than 7.30) | 1 | Severe respiratory acidosis |
| Atrial Fibrillation | 1 | On admission ECG |
DECAF Mortality Interpretation:
- 0-1 points: Low risk (less than 3% in-hospital mortality) - consider discharge
- 2 points: Intermediate risk (9% mortality) - admit to ward
- 3-6 points: High risk (31-50% mortality) - admit to HDU/ICU
ICU/HDU Admission Criteria:
- pH less than 7.30 despite 1 hour NIV
- Respiratory arrest or imminent
- Hemodynamic instability requiring vasopressors
- Altered consciousness (GCS less than 12)
- Multi-organ failure
Discharge Criteria:
| Domain | Criteria | Verification |
|---|---|---|
| Clinical Stability | No use of SABA for 4+ hours | Observation period |
| Oxygenation | SpO2 > 90% on room air OR stable on home oxygen | Pulse oximetry |
| Mobility | Able to walk across room | 6-minute walk or equivalent |
| Self-Care | Able to eat, drink, sleep adequately | Self-report + nursing assessment |
| Inhaler Technique | Demonstrated correct use | Pharmacist/nurse check |
| Understanding | Can recognize worsening and action plan | Teach-back method |
| Follow-Up | Appointment within 2-4 weeks | Scheduled before discharge |
| Home Support | Adequate or arranged | Social work assessment if needed |
Post-Discharge Care Bundle:
- Within 48 hours: Phone call to check symptoms, medications
- Within 7 days: Respiratory nurse or GP visit
- Within 30 days: Pulmonary rehabilitation referral
- Within 90 days: Specialist respiratory follow-up
Preventing Readmissions (Evidence-Based Strategies):
- Pulmonary rehabilitation within 1 month (RR 0.56 for readmission) [17]
- Discharge bundle implementation (inhaler training, action plan, follow-up) reduces 30-day readmissions by 25%
- Telephone follow-up programs (RR 0.75 for readmissions)
- Early supported discharge schemes for selected patients
SECTION 8: Complications
8.1 Immediate Complications
| Complication | Incidence | Presentation | Risk Factors | Management |
|---|---|---|---|---|
| Acute Resp Failure | 15-20% | pH less than 7.35, pCO2 > 45 | Severe obstruction | NIV or Intubation |
| Pneumothorax | 2-5% | Sudden pain/dyspnea | Bullous emphysema | Chest drain |
| Arrhythmias (AF) | 10-15% | Palpitations | Hypoxia, Beta-agonists | Rate control/O2 |
| PE | 5-10% | Unexplained dyspnea | Immobility | Anticoagulation |
| Acute Heart Fail | 10% | Orthopnea, rales | Comorbid CAD | Diuretics |
8.2 Early Complications
- Pneumonia: Incidence increased 2-fold in patients on ICS
- Steroid-induced Myopathy: Weakness of respiratory muscles after high-dose systemic steroids
- Hyperglycemia: Common during AECOPD treatment with Prednisolone
8.3 Late Complications
- Cor Pulmonale: Right-sided heart failure due to chronic pulmonary hypertension
- Secondary Polycythemia: Hct > 55% due to chronic hypoxemia; increases stroke risk
- Lung Cancer: COPD is an independent risk factor for bronchogenic carcinoma
- Osteoporosis: Due to smoking, inactivity, and steroid use
8.4 Comorbidities in COPD
COPD rarely exists in isolation. Comorbidities are present in > 95% of COPD patients and significantly impact outcomes. Recent evidence emphasizes the importance of treating cardiovascular comorbidities aggressively. [10]
Major Comorbidities and Management:
| Comorbidity | Prevalence in COPD | Impact | Screening | Management Considerations |
|---|---|---|---|---|
| Cardiovascular Disease | 30-50% | Leading cause of death in mild-moderate COPD [10] | ECG, echocardiogram if indicated | Beta-blockers NOT contraindicated; cardioselective preferred |
| Ischemic Heart Disease | 20-30% | 2-3x higher risk vs general population | Stress test if symptomatic | Treat as per guidelines; beta-blockers safe in COPD |
| Heart Failure | 20-25% | Overlapping symptoms complicate diagnosis | NT-proBNP, echo | Diuretics, ACEI, beta-blockers; avoid NSAIDs |
| Atrial Fibrillation | 15-20% | Increases exacerbation risk | ECG on admission for AECOPD | Rate control; anticoagulation as per CHA2DS2-VASc |
| Hypertension | 40-50% | Shared risk factors with COPD | BP monitoring | All antihypertensives safe; avoid beta-blockers only if bronchospasm |
| Diabetes Mellitus | 15-20% | Worsened by systemic steroids | HbA1c, glucose monitoring | Monitor closely during AECOPD; adjust insulin |
| Osteoporosis | 35-60% | Fracture risk increased 2-5x | DEXA scan if age > 50 or long-term steroids | Bisphosphonates, vitamin D, weight-bearing exercise |
| Depression | 25-40% | Associated with poor adherence and outcomes | PHQ-9 screening | SSRIs safe; pulmonary rehabilitation helps |
| Anxiety | 20-35% | Exacerbates dyspnea perception | GAD-7 screening | CBT, pulmonary rehabilitation, SSRIs |
| GERD | 30-60% | May trigger exacerbations | Clinical diagnosis, endoscopy if alarm features | PPIs, lifestyle modifications |
| Anemia | 10-20% | Worsens dyspnea and exercise capacity | FBC, ferritin, B12 | Iron supplementation, investigate cause |
| Cachexia/Sarcopenia | 20-40% in severe COPD | Independent predictor of mortality | BMI, FFMI, handgrip strength | High-protein diet, anabolic steroids in select cases |
| Chronic Kidney Disease | 10-15% | Adjust drug doses | eGFR annually | Careful with nephrotoxic drugs, adjust theophylline |
| Sleep Disorders (OSA) | 10-15% overlap | Increases cardiovascular risk | Epworth score, sleep study | CPAP or BiPAP for overlap syndrome |
| Cognitive Impairment | 25% in severe COPD | Affects self-management | MMSE/MoCA | Simplify regimens, involve caregivers |
| Lung Cancer | 4-6x increased risk | Leading cause of death after COPD | Low-dose CT screening if age 55-80, ≥30 pack-years | Per lung cancer guidelines |
Comorbidity Assessment Tools:
- Charlson Comorbidity Index: Predicts 10-year survival
- COTE Index (COPD-specific Comorbidity Test): 12-item index predicting mortality
- Comprehensive Geriatric Assessment: For elderly COPD patients (> 70 years)
Management Principles for Comorbid COPD:
- Treat each condition according to guidelines - no "COPD exception" for most drugs
- Beta-blockers are SAFE in COPD (even in severe COPD) - do not withhold for cardiac indications
- ACE inhibitors may cause cough but do not worsen COPD - persevere if cardiac indication
- Statins reduce exacerbations and cardiovascular events - consider in all COPD with CVD [10]
- Screen for depression/anxiety - significantly improves QoL and adherence
- Optimize bone health - DEXA scan for all on long-term ICS or recurrent steroids
- Low-dose CT lung cancer screening for eligible patients (55-80 years, ≥30 pack-years)
SECTION 9: Prognosis & Outcomes
9.1 Natural History
Without treatment, COPD is characterized by a steady decline in FEV1 (approx 50-100 mL/year in active smokers vs 20-30 mL/year in non-smokers). Mortality is highly linked to the frequency of exacerbations.
9.2 Outcomes with Treatment
| Variable | Outcome | Notes |
|---|---|---|
| 5-year survival | 40-70% | Highly dependent on GOLD stage |
| Mortality Reduction | ~20% | Achieved with LTOT in hypoxemic patients |
| Exacerbation Rate | 30-50% reduction | With optimized triple therapy [5,6] |
| QoL Improvement | Significant | Measured by SGRQ or CAT scores |
9.3 Prognostic Factors
Good Prognosis Factors:
- Successful smoking cessation (RR 0.5 for mortality)
- Higher baseline FEV1 (> 50%)
- High levels of physical activity
- Normal BMI (21-25)
- Reversibility on spirometry (> 12% and 200mL)
Poor Prognosis Factors:
- BODE Score > 7
- Frequent exacerbations (≥2 per year)
- Chronic hypercapnia (pCO2 > 45)
- Pulmonary hypertension on Echo
- Severe cachexia (BMI less than 18)
SECTION 10: Evidence & Guidelines
10.1 Key Guidelines
Guideline 1: GOLD Report (2024) [1,3,4]
- Organization: Global Initiative for Chronic Obstructive Lung Disease
- Key Recommendation 1: Shift to ABE assessment tool (combining C and D into E)
- Key Recommendation 2: Triple therapy (LABA+LAMA+ICS) preferred for Group E with Eos > 300
- Link: goldcopd.org
Guideline 2: NICE Guideline NG115 (2019/Updated 2023)
- Organization: National Institute for Health and Care Excellence (UK)
- Key Recommendation: Emphasizes pulmonary rehabilitation and smoking cessation at every stage
10.2 Landmark Trials
TRIAL 1: IMPACT (2018) [5]
- Study Design: Phase III RCT, n = 10,355
- Intervention: Triple therapy (Fluticasone/Umeclidinium/Vilanterol)
- Control: Dual therapy (ICS/LABA or LAMA/LABA)
- Key Finding: Triple therapy resulted in a significantly lower rate of moderate-to-severe exacerbations (0.91 vs 1.07/1.21)
- DOI: 10.1056/NEJMoa1713901
TRIAL 2: ETHOS (2020) [6]
- Intervention: Triple therapy (Budesonide/Glycopyrrolate/Formoterol)
- Key Finding: Triple therapy reduced the rate of moderate or severe exacerbations and, crucially, showed a reduction in all-cause mortality (24% reduction vs LABA/LAMA)
- DOI: 10.1056/NEJMoa1916046
TRIAL 3: FLAME (2016) [19]
- Intervention: LAMA/LABA (Indacaterol/Glycopyrronium) vs ICS/LABA (Salmeterol/Fluticasone)
- Key Finding: LAMA/LABA was superior to ICS/LABA in preventing exacerbations, regardless of eosinophil count
- DOI: 10.1056/NEJMoa1516385
TRIAL 4: REDUCE (2013) [15]
- Finding: 5 days of systemic corticosteroids is non-inferior to 14 days for AECOPD
- DOI: 10.1001/jama.2013.5023
10.3 Evidence Strength Table
| Intervention | Evidence Level | Key Evidence | Recommendation Grade |
|---|---|---|---|
| Smoking Cessation | 1a | Multiple Cohorts | Grade A |
| LAMA+LABA | 1a | FLAME Trial [19] | Grade A |
| Triple Therapy | 1a | IMPACT/ETHOS [5,6] | Grade A |
| Pulmonary Rehab | 1a | Cochrane Review [17,18] | Grade A |
| LTOT (> 15h/day) | 1b | NOTT Trial | Grade A |
SECTION 11: Patient/Layperson Explanation
11.1 What is COPD?
COPD stands for Chronic Obstructive Pulmonary Disease. Think of your lungs like a tree with many branches (airways) ending in tiny balloons (air sacs). In COPD, the "branches" get clogged with mucus and become swollen, and the "balloons" lose their stretchiness or pop. This makes it very hard to push air out of your lungs, leaving you feeling short of breath, as if you are breathing through a straw.
11.2 Why Does It Matter?
While the damage to the lungs cannot be fully reversed, the symptoms can be managed. If left untreated, COPD makes simple tasks like walking to the mailbox or getting dressed very difficult. It also makes you more likely to get serious chest infections that could land you in the hospital.
11.3 How Is It Treated?
- Stopping Smoking: This is the most important step to keep your lungs from getting worse
- Inhalers: These act like "WD-40" for your airways, helping them stay open so you can breathe easier
- Pulmonary Rehab: This is a special exercise program that teaches your muscles how to use oxygen more efficiently
- Vaccines: Getting your flu and pneumonia shots prevents the infections that cause "flare-ups"
11.4 When to Seek Help
Call your doctor or go to the emergency room if:
- You are so short of breath you cannot speak
- Your lips or fingernails look blue or gray
- You feel confused or unusually sleepy
- Your rescue inhaler (usually blue) isn't helping
SECTION 12: References
-
Global Initiative for Chronic Obstructive Lung Disease (GOLD). Global Strategy for the Diagnosis, Management, and Prevention of COPD. 2024. Available at: goldcopd.org
-
Sharma M, et al. Global Initiative for Chronic Obstructive Lung Disease (GOLD) 2023 Guidelines Reviewed. Open Respir Med J. 2024. doi:10.2174/0118743064279064231227070344 PMID: 38660684
-
Cornelius T, et al. Clinical guideline highlights for the hospitalist: GOLD COPD update 2024. J Hosp Med. 2024 Sep. doi:10.1002/jhm.13416 PMID: 38797887
-
Suissa S, et al. Guidelines for the Pharmacologic Treatment of COPD 2023: Canada versus GOLD. COPD. 2024 Dec. doi:10.1080/15412555.2023.2292613 PMID: 38329461
-
Lipson DA, et al. Once-Daily Single-Inhaler Triple versus Dual Therapy in Patients with COPD (IMPACT). N Engl J Med. 2018;378(18):1671-1680. doi:10.1056/NEJMoa1713901 PMID: 29668352
-
Rabe KF, et al. Triple Inhaled Therapy at Two Glucocorticoid Doses in Moderate-to-Very-Severe COPD (ETHOS). N Engl J Med. 2020;383(1):35-48. doi:10.1056/NEJMoa1916046 PMID: 32579807
-
Wang C, et al. Prevalence and risk factors of chronic obstructive pulmonary disease in China (the China Pulmonary Health [CPH] study). Lancet. 2018;391(10131):1706-1717. doi:10.1016/S0140-6736(18)30841-9 PMID: 29650248
-
Singh D, et al. GOLD Science Committee recommendations for the use of pre- and post-bronchodilator spirometry for the diagnosis of COPD. Eur Respir J. 2025;65(2):2401603. doi:10.1183/13993003.01603-2024 PMID: 39638416
-
Mathioudakis AG, et al. Rethinking Blood Eosinophils for Assessing Inhaled Corticosteroids Response in COPD: A Post Hoc Analysis From the FLAME Trial. Chest. 2024;166(5):987-997. doi:10.1016/j.chest.2024.06.3790 PMID: 38992490
-
Polman R, et al. Cardiovascular disease and risk in COPD: a state of the art review. Expert Rev Cardiovasc Ther. 2024 Apr-May. doi:10.1080/14779072.2024.2333786 PMID: 38529639
-
Cheng SL. Blood eosinophils and inhaled corticosteroids in patients with COPD: systematic review and meta-analysis. Int J Chron Obstruct Pulmon Dis. 2018;13:2775-2784. doi:10.2147/COPD.S175017 PMID: 30233168
-
Xie C, et al. Toward precision medicine in COPD: phenotypes, endotypes, biomarkers, and treatable traits. Respir Res. 2025 Sep 29. doi:10.1186/s12931-025-03356-w PMID: 41024111
-
Pleasants RA, et al. Current Perspectives of Pharmacotherapies for COPD. Respir Care. 2023 Jul. doi:10.4187/respcare.10952 PMID: 37353337
-
Qian Y, et al. Analyses of Factors Associated with Acute Exacerbations of Chronic Obstructive Pulmonary Disease: A Review. Int J Chron Obstruct Pulmon Dis. 2023. doi:10.2147/COPD.S433183 PMID: 38034468
-
Leuppi JD, et al. Short-term vs conventional glucocorticoid therapy in acute exacerbations of COPD: the REDUCE randomized clinical trial. JAMA. 2013;309(21):2223-31. doi:10.1001/jama.2013.5023 PMID: 23695288
-
Anthonisen NR, et al. Antibiotic therapy in exacerbations of chronic obstructive pulmonary disease. Ann Intern Med. 1987;106(2):196-204. doi:10.7326/0003-4819-106-2-196 PMID: 3492164
-
Puhan MA, et al. Pulmonary rehabilitation following exacerbations of chronic obstructive pulmonary disease. Cochrane Database Syst Rev. 2016;12:CD005305. doi:10.1002/14651858.CD005305.pub4 PMID: 27930803
-
Lamberton CE, et al. Review of the Evidence for Pulmonary Rehabilitation in COPD: Clinical Benefits and Cost-Effectiveness. Respir Care. 2024 May 28. doi:10.4187/respcare.11541 PMID: 38503466
-
Wedzicha JA, et al. Indacaterol-Glycopyrronium versus Salmeterol-Fluticasone for COPD (FLAME). N Engl J Med. 2016;374(23):2222-2234. doi:10.1056/NEJMoa1516385 PMID: 27182917
-
Martinez FJ, et al. Effect of roflumilast on exacerbations in patients with severe chronic obstructive pulmonary disease uncontrolled by combination therapy (REACT). Lancet. 2015;385(9971):857-866. doi:10.1016/S0140-6736(14)62410-7 PMID: 25684586
-
Albert RK, et al. Azithromycin for Prevention of Exacerbations of COPD. N Engl J Med. 2011;365(8):689-698. doi:10.1056/NEJMoa1104623 PMID: 21864166
-
Criner GJ, et al. A Randomized Trial of Zephyr Endobronchial Valves for Severe Emphysema. Am J Respir Crit Care Med. 2018;198(9):1151-1164. doi:10.1164/rccm.201803-0590OC PMID: 29787288
SECTION 13: Examination Focus
13.1 Common Exam Questions
USMLE Step 2/3 High-Yield:
-
"A 65-year-old smoker presents with worsening dyspnea and FEV1/FVC of 0.62. What is the most important intervention to improve survival?"
- Answer: Smoking cessation (and LTOT if PaO2 ≤55 mmHg). Only these two interventions have proven mortality benefit.
-
"A COPD patient on LABA/LAMA has had 3 exacerbations in the past year. Blood eosinophils are 350 cells/µL. What is the next step?"
- Answer: Add ICS to current regimen (triple therapy). Eosinophils > 300 predict ICS response. [9,11]
-
"During an AECOPD, a patient on 4L O2 has SpO2 of 94%, but ABG shows pH 7.28, PaCO2 58 mmHg. What is the most appropriate action?"
- Answer: Reduce oxygen to target SpO2 88-92% AND initiate NIV. High-flow oxygen worsens hypercapnia in COPD.
MRCP Part 1/2 Favorites:
-
"Which trial showed that LAMA/LABA is superior to ICS/LABA for exacerbation prevention?"
- Answer: The FLAME trial (2016). [19]
-
"A patient with COPD has a blood eosinophil count of 50 cells/µL. What is the risk of adding an ICS?"
- Answer: Increased risk of pneumonia (NNH 20) with minimal benefit in exacerbation reduction.
-
"A 55-year-old presents with COPD and basal-predominant emphysema on CT. What test should be ordered?"
- Answer: Alpha-1 antitrypsin level (suspect AAT deficiency with basal emphysema and age less than 60).
-
"What is the proven mortality benefit of 5-day vs 14-day oral corticosteroids in AECOPD?"
- Answer: Non-inferior outcomes (REDUCE trial) - use 5 days to minimize side effects. [15]
FRACP/MRCP PACES Clinical Stations:
-
Station: "Examine this patient's respiratory system"
- Findings: Barrel chest, hyperresonance, reduced breath sounds, prolonged expiration
- Presentation: "This patient has features consistent with COPD, characterized by hyperinflation and airflow obstruction. I would like to complete my examination with spirometry and assess for cor pulmonale with JVP and ankle edema."
-
Station: "This patient has been admitted with an exacerbation of COPD. Discuss management."
- Structure: A-E assessment → Controlled oxygen → Nebulizers → Steroids and antibiotics → Assess for NIV criteria → Disposition
13.2 Viva Opening Statements and Model Answers
Opening Statement for COPD: "COPD is a common, preventable, and treatable heterogeneous lung condition characterized by persistent, often progressive, airflow limitation due to airway and/or alveolar abnormalities. It is the third leading cause of death worldwide. The diagnosis requires post-bronchodilator spirometry demonstrating FEV1/FVC less than 0.70. Management is now personalized based on symptom burden and exacerbation risk using the GOLD ABE classification, with smoking cessation being the single most important intervention."
High-Yield Viva Questions and Model Answers:
Q: "Tell me about the GOLD 2024 classification changes."
A: "The GOLD 2024 update introduced the ABE classification system, merging the previous C and D groups into a single 'E' category for frequent exacerbators. Group A has low symptoms and low exacerbation risk (0-1 non-hospitalized exacerbations), Group B has high symptoms but low exacerbation risk, and Group E has ≥2 exacerbations or ≥1 hospitalization regardless of symptoms. This simplified approach better guides initial bronchodilator therapy." [3,4]
Q: "What is the evidence for triple therapy in COPD?"
A: "Two landmark trials established triple therapy. The IMPACT trial (2018) with 10,355 patients showed triple therapy reduced exacerbations by 15% vs LABA/LAMA. More importantly, the ETHOS trial (2020) was the first to demonstrate all-cause mortality reduction with triple therapy - a 24% reduction vs LABA/LAMA. Triple therapy is now recommended for Group E patients, particularly those with blood eosinophils > 300 cells/µL." [5,6]
Q: "When would you prescribe long-term oxygen therapy?"
A: "LTOT is indicated when PaO2 is ≤55 mmHg (7.3 kPa) or SpO2 ≤88% at rest on room air in stable state. It's also indicated if PaO2 is 55-60 mmHg with evidence of pulmonary hypertension, peripheral edema, or polycythemia. The evidence comes from the MRC and NOTT trials from the 1980s showing mortality benefit with ≥15 hours/day usage. Critically, the recent LOTT trial showed NO benefit for moderate desaturation (SpO2 89-93%), so we must be strict with criteria to avoid over-prescription."
Q: "How do you manage acute hypercapnic respiratory failure in AECOPD?"
A: "I use a systematic approach. First, controlled oxygen targeting SpO2 88-92% using a Venturi mask at 24-28%. Second, bronchodilators via nebulizer - salbutamol 5mg plus ipratropium 500mcg. Third, systemic corticosteroids - prednisolone 30-40mg for 5 days. Fourth, antibiotics if increased sputum purulence suggesting bacterial infection. If after 1 hour of optimal therapy the patient has pH less than 7.35 with rising PaCO2, I would initiate NIV with BiPAP settings of IPAP 12-15 and EPAP 4-5, titrating upwards. NIV has strong evidence reducing intubation and mortality in this setting from the Cochrane review."
Q: "When should you screen for alpha-1 antitrypsin deficiency?"
A: "WHO recommends screening every COPD patient at least once. However, I specifically suspect and prioritize screening in: patients diagnosed less than 45 years, never/minimal smokers with COPD, those with basal-predominant emphysema on CT, family history of early COPD or liver disease, and patients with unexplained liver disease. The test is a simple blood level - if low, confirm with Pi phenotyping. ZZ homozygotes have highest risk and may benefit from augmentation therapy if FEV1 30-65% predicted."
Q: "What is your approach to a COPD patient with recurrent exacerbations despite optimal inhaled therapy?"
A: "I would use a systematic approach. First, verify inhaler technique and adherence - poor technique causes 70% of treatment failures. Second, check blood eosinophils - if > 300, ensure on triple therapy with ICS. Third, consider prophylactic therapy: azithromycin 250mg three times weekly reduces exacerbations by 30% (COLUMBUS trial), particularly in non-eosinophilic patients. [21] Roflumilast 500mcg daily if FEV1 less than 50% and chronic bronchitis phenotype. [20] Fourth, ensure pulmonary rehabilitation enrollment. Fifth, assess and treat comorbidities - GERD, OSA, heart failure all increase exacerbation risk. Finally, in severe refractory cases, consider endobronchial valves if upper lobe-predominant emphysema. [22]"
13.3 Key Facts to Memorize for Exams
Numbers You Must Know:
- FEV1/FVC cutoff: less than 0.70 (post-bronchodilator)
- LTOT indication: PaO2 ≤55 mmHg OR ≤60 mmHg + cor pulmonale
- Target SpO2 in AECOPD: 88-92%
- Steroids for AECOPD: 30-40mg prednisolone for 5 days (not 10-14 days) [15]
- NIV initiation threshold: pH less than 7.35 with pCO2 > 45 mmHg
- Eosinophil thresholds: less than 100 (avoid ICS), 100-300 (consider), > 300 (strongly recommend ICS) [9,11]
- 6-minute walk MCID: 30 meters
- FEV1 decline in smokers: 50-100 mL/year vs 20-30 mL/year in non-smokers
- BODE score: BMI, Obstruction (FEV1), Dyspnea (mMRC), Exercise (6MWT) - range 0-10, > 7 indicates high mortality
Trials You Must Know:
- IMPACT (2018): Triple therapy reduces exacerbations vs dual therapy [5]
- ETHOS (2020): Triple therapy reduces mortality [6]
- FLAME (2016): LABA/LAMA > ICS/LABA for exacerbations [19]
- REDUCE (2013): 5 days steroids non-inferior to 14 days [15]
- LOTT (2016): No benefit of oxygen for moderate desaturation
- MRC/NOTT (1980s): LTOT improves survival in severe hypoxemia
- UPLIFT (2008): Tiotropium improves QoL but not mortality
- TORCH (2007): ICS/LABA improves QoL and reduces exacerbations
13.4 Common Mistakes That Fail Candidates
❌ CRITICAL ERRORS:
- Recommending ICS as monotherapy (always must be combined with LABA; monotherapy increases pneumonia risk without benefit)
- Using high-flow oxygen (> 2L or SpO2 > 94%) in AECOPD before checking ABG (causes CO2 retention and narcosis)
- Stating "pink puffer/blue bloater" as diagnostic criteria (outdated phenotypic descriptions, NOT diagnostic)
- Forgetting to mention smoking cessation as THE most important intervention
- Recommending oxygen for moderate desaturation (SpO2 89-93%) - LOTT trial showed no benefit
- Saying beta-blockers are contraindicated in COPD (they are SAFE and should be used for cardiac indications) [10]
❌ COMMON PITFALLS:
- Not checking post-bronchodilator spirometry (must be post-bronchodilator for diagnosis) [8]
- Forgetting to screen for alpha-1 antitrypsin in young patients (less than 45 years)
- Not adjusting oxygen target (should be 88-92%, NOT 94-98% as in other conditions)
- Ordering inappropriate investigations (CT chest NOT needed for diagnosis; spirometry is gold standard)
- Forgetting pulmonary rehabilitation (Level 1a evidence, often overlooked) [17,18]
- Not considering NIV early enough in AECOPD (should be within 1 hour if pH less than 7.35)
13.5 Exam Mnemonics and Memory Aids
**"COPD"
- Key Management Points:**
- Cessation of smoking (most important!)
- Oxygen therapy (if severely hypoxemic)
- Pulmonary rehabilitation (for all with mMRC ≥2)
- Drugs (bronchodilators ± ICS based on phenotype)
**"GOLD ABE"
- Classification:**
- A: Asymptomatic (low symptoms, low risk)
- B: Breathless (high symptoms, low risk)
- E: Exacerbator (frequent exacerbations, any symptoms)
"LTOT" Criteria to Remember:
- Less than 55 mmHg PaO2 (absolute indication)
- Three (55-60 mmHg) if cor pulmonale, edema, or polycythemia
- Only after 60-90 days stability (not during acute exacerbation)
- Time requirement: > 15 hours/day for mortality benefit
**"AECOPD Treatment"
- SBAO:**
- Steroids (prednisolone 30-40mg x 5 days)
- Bronchodilators (nebulized SABA + SAMA)
- Antibiotics (if purulent sputum)
- Oxygen (controlled, target 88-92%)
Last Updated: 2026-01-11
Medical Disclaimer: MedVellum content is for educational purposes and clinical reference. Clinical decisions should account for individual patient circumstances. Always consult appropriate specialists and current guidelines.
Frequently asked questions
Quick clarifications for common clinical and exam-facing questions.
When should I seek emergency care for chronic obstructive pulmonary disease (copd) in adults?
Seek immediate emergency care if you experience any of the following warning signs: Acute worsening of dyspnea at rest, New-onset confusion or altered mental status (Hypercapnia), Use of accessory respiratory muscles, Peripheral cyanosis or saturations less than 88% on room air, Inability to speak in full sentences, Paradoxical chest wall movement, New-onset peripheral edema (Cor pulmonale), Silent chest on auscultation during exacerbation, Respiratory acidosis (pH less than 7.35) with hypercapnia.