Deep Vein Thrombosis (DVT) - Adult
Deep Vein Thrombosis (DVT) is the formation of a blood clot (thrombus) within the deep venous system, most commonly affe... MRCP exam preparation.
What matters first
Deep Vein Thrombosis (DVT) is the formation of a blood clot (thrombus) within the deep venous system, most commonly affe... MRCP exam preparation.
Phlegmasia Cerulea Dolens (Limb-Threatening Emergency)
9 Jan 2025
Generated educational material; verify before clinical use.
Visible references section
See the concept before reading it
Study the key anatomy, imaging, and decision pathways as full teaching plates.
Clinical board
A visual summary of the highest-yield teaching signals on this page.
Urgent signals
Safety-critical features pulled from the topic metadata.
- Phlegmasia Cerulea Dolens (Limb-Threatening Emergency)
- Phlegmasia Alba Dolens
- Concurrent Pulmonary Embolism Symptoms
- Massive Ilio-femoral DVT (High Embolic Risk)
Exam focus
Current exam surfaces linked to this topic.
- MRCP
Linked comparisons
Differentials and adjacent topics worth opening next.
- Cellulitis
- Baker's Cyst Rupture
Content status and exam context
This page is AI-generated educational content. It may contain errors or omissions and is not a substitute for current guidelines, local protocols, senior clinical judgement, or professional medical advice.
MedVellum does not claim an individual clinician reviewer, board certification, or professional credential for this page unless a future version names a real, verifiable reviewer.
Clinical explanation and evidence
Deep Vein Thrombosis (DVT) - Adult
1. Overview
Deep Vein Thrombosis (DVT) is the formation of a blood clot (thrombus) within the deep venous system, most commonly affecting the lower extremities. It represents a major component of Venous Thromboembolism (VTE), which encompasses both DVT and Pulmonary Embolism (PE). DVT is a critical diagnosis because untreated proximal DVT carries a 50% risk of progression to PE, which has a mortality rate of 25-30% if massive and untreated. [1,2]
The annual incidence of DVT is approximately 1-2 per 1,000 population, rising exponentially with age to 1 per 100 in those over 80 years. [3] The economic burden is substantial, with healthcare costs exceeding USD 10 billion annually in the United States alone. [4] The cornerstone of management is rapid anticoagulation to prevent clot propagation, embolisation, and recurrence, while balancing bleeding risk.
Understanding DVT requires mastery of Virchow's triad (stasis, hypercoagulability, endothelial injury), clinical probability scoring (Wells score), diagnostic algorithms (D-dimer and compression ultrasonography), and evidence-based treatment protocols. This topic is high-yield for MRCP, MRCS, and clinical practice, appearing frequently in both written examinations and clinical vivas.
Clinical Pearl: The key clinical message: A negative D-dimer combined with a low pre-test probability (Wells score less than 2) can safely exclude DVT without imaging, reducing unnecessary ultrasounds by 30-40%. However, D-dimer should NEVER be used in high-probability patients or post-operatively.
2. Epidemiology
Incidence and Prevalence
DVT is a common condition with significant morbidity and mortality. The epidemiology varies by population, age, and risk factor prevalence.
| Epidemiological Parameter | Value | Source |
|---|---|---|
| Annual incidence (general population) | 1-2 per 1,000 person-years | [3] |
| Annual incidence (age > 80 years) | 1 per 100 person-years | [3] |
| Hospital-acquired VTE incidence | 100-200 per 100,000 admissions | [5] |
| Recurrence rate (unprovoked, 10 years) | 30-40% | [6] |
| Case-fatality rate (DVT alone) | 1-2% | [2] |
| PE-related mortality (all VTE) | 5-15% | [1] |
Demographic Distribution
Age: The incidence of DVT increases exponentially with age. The risk is minimal before age 15 (approximately 1 per 100,000), rising to 5 per 1,000 by age 80. [3] This increase reflects cumulative exposure to risk factors, decreased mobility, and age-related changes in coagulation.
Sex: Overall, DVT incidence is slightly higher in males (1.3 per 1,000) compared to females (1.1 per 1,000). However, during reproductive years (15-45), females have higher rates due to pregnancy, oral contraceptive use, and hormone replacement therapy. After menopause, the sex distribution equalises. [7]
Ethnicity: African Americans have a 30-40% higher incidence of VTE compared to Caucasians, while Asian and Hispanic populations have lower rates. These differences may reflect genetic variations in coagulation factor polymorphisms and thrombophilia prevalence. [8]
Geography: VTE incidence is highest in Western countries and lowest in Asian populations, though these differences are narrowing with Westernisation of lifestyles and improved diagnostic practices. [8]
Risk Factor Distribution
The population-attributable risk varies by setting:
| Risk Factor | Attributable Risk | Relative Risk |
|---|---|---|
| Recent surgery/trauma | 20-25% | 5-50x |
| Hospitalisation | 20-25% | 5-10x |
| Active malignancy | 15-20% | 4-7x |
| Immobilisation | 10-15% | 2-4x |
| Prior VTE | 10-15% | 8-10x |
| Obesity (BMI > 30) | 10-15% | 2-3x |
| Oral contraceptives | 5-10% | 3-5x |
| Thrombophilia | 5-10% | 2-8x |
Temporal Trends
VTE incidence has remained relatively stable over the past two decades, despite improvements in prophylaxis. This paradox reflects improved diagnostic sensitivity (widespread D-dimer and ultrasound availability), an ageing population, and increased surgical and oncological complexity. [5]
Exam Detail: Exam-Relevant Statistics to Memorise:
- Annual incidence: 1-2 per 1,000
- Proximal DVT → PE risk (untreated): 50%
- PE mortality (massive, untreated): 25-30%
- Recurrence risk (unprovoked): 10% year 1, 30-40% at 10 years
- Post-thrombotic syndrome: 20-50% within 2 years
- Factor V Leiden prevalence (Caucasians): 5%
3. Aetiology and Pathophysiology
Virchow's Triad: The Fundamental Framework
Rudolf Virchow described the three factors necessary for thrombosis in 1856, and this triad remains the cornerstone of understanding DVT pathophysiology. All three elements interact synergistically, and most clinical DVTs result from a combination of two or more factors. [9]
3.1 Venous Stasis
Mechanism: Reduced blood flow velocity allows accumulation of activated clotting factors, impairs endothelial washout of procoagulants, and promotes platelet-endothelial interactions. The venous valve cusps are particularly vulnerable as they represent areas of low shear stress where thrombi typically originate. [9,10]
Clinical Causes of Stasis:
| Cause | Mechanism | Risk Level |
|---|---|---|
| Prolonged immobilisation (> 72 hours) | Loss of calf muscle pump | High |
| Hospitalisation/bed rest | Combined immobility and illness | High |
| Long-haul travel (> 4 hours) | Seated position, dehydration | Moderate |
| Plaster cast/immobilisation | Mechanical prevention of muscle contraction | Moderate |
| Paralysis/paresis (stroke, spinal injury) | Permanent loss of muscle pump | High |
| Heart failure | Reduced cardiac output, venous congestion | Moderate |
| Obesity (BMI > 30) | Increased abdominal pressure, reduced mobility | Moderate |
| Varicose veins | Venous insufficiency and pooling | Low-Moderate |
Pathophysiology of Valve Cusp Thrombosis: Thrombus formation typically begins in the valve sinus of the soleal and gastrocnemius veins (calf veins), where blood flow is slowest. The valve pockets create areas of recirculation and prolonged contact time between blood and endothelium. Computational fluid dynamics studies demonstrate oxygen tension drops of up to 40% in valve sinuses during immobility, leading to endothelial activation. [10]
3.2 Hypercoagulability
Mechanism: An imbalance between procoagulant and anticoagulant factors shifts the haemostatic equilibrium toward thrombosis. This can result from inherited deficiencies of natural anticoagulants, acquired prothrombotic states, or excess procoagulant factors.
Inherited Thrombophilias:
| Thrombophilia | Prevalence (General) | VTE Risk (Heterozygous) | VTE Risk (Homozygous) |
|---|---|---|---|
| Factor V Leiden | 5% Caucasians | 5-7x | 50-80x |
| Prothrombin G20210A | 2-3% Caucasians | 2-4x | 10-20x |
| Protein C deficiency | 0.2-0.4% | 7-10x | Neonatal purpura fulminans |
| Protein S deficiency | 0.1-0.3% | 5-10x | Variable |
| Antithrombin deficiency | 0.02-0.2% | 10-50x | Often incompatible with life |
Factor V Leiden (Most Common Inherited Thrombophilia):
- Genetic mutation: G1691A in F5 gene (Arg506Gln)
- Mechanism: Factor V becomes resistant to inactivation by activated Protein C (APC resistance)
- Normally, APC cleaves Factor Va at Arg506, terminating its procoagulant activity
- Mutant Factor V Leiden is cleaved 10x slower, prolonging thrombin generation
- Heterozygous state: 5-7x increased VTE risk
- Homozygous state: 50-80x increased VTE risk
- Combined with OCP: 30-50x increased risk (synergistic) [11]
Acquired Hypercoagulable States:
| Condition | Mechanism | Risk Level |
|---|---|---|
| Active malignancy | Tissue factor expression, mucin secretion, chemotherapy | 4-7x |
| Pregnancy/puerperium | Increased factors VII, VIII, X, VWF; decreased Protein S | 5-10x |
| Oral contraceptives (combined) | Increased factors II, VII, VIII, X; APC resistance | 3-5x |
| Hormone replacement therapy | Similar to OCP but lower magnitude | 2-3x |
| Antiphospholipid syndrome | Autoantibodies to phospholipid-binding proteins | 5-10x |
| Nephrotic syndrome | Urinary loss of antithrombin III | 3-8x |
| Myeloproliferative neoplasms | Hyperviscosity, abnormal platelet function | 3-5x |
| Inflammatory bowel disease | Inflammation, immobility, steroids | 2-3x |
| COVID-19 infection | Endothelial dysfunction, inflammation, stasis | 5-20x (critical illness) |
Antiphospholipid Syndrome (APS):
- Autoimmune disorder characterised by antiphospholipid antibodies (lupus anticoagulant, anticardiolipin, anti-β2-glycoprotein I)
- Clinical features: Recurrent venous/arterial thrombosis, pregnancy morbidity
- Paradox: "Lupus anticoagulant" prolongs aPTT in vitro but is PROTHROMBOTIC in vivo
- Classification criteria: ≥1 clinical criterion + ≥1 laboratory criterion (positive on 2 occasions, 12 weeks apart)
- Triple-positive APS (all three antibodies): Highest thrombotic risk, DOAC contraindicated [12]
Clinical Pearl: The OCP-Factor V Leiden Synergy: A woman with Factor V Leiden who takes combined oral contraceptives has a 30-50x increased VTE risk (multiplicative effect). This is why family history of VTE less than 45 years should prompt thrombophilia testing BEFORE starting oestrogen-containing contraception.
3.3 Endothelial Injury
Mechanism: Damage to the vascular endothelium exposes subendothelial collagen and tissue factor, triggering platelet adhesion and the extrinsic coagulation pathway. Healthy endothelium is actively antithrombotic, producing prostacyclin, nitric oxide, and expressing thrombomodulin.
Clinical Causes of Endothelial Injury:
| Cause | Mechanism | Clinical Context |
|---|---|---|
| Surgery | Direct vessel trauma, tissue factor release | Hip/knee replacement highest risk |
| Trauma | Vessel wall damage, inflammation | Major trauma > 3x VTE risk |
| Central venous catheters | Mechanical irritation, foreign body | PICC lines, tunnelled catheters |
| Previous DVT | Residual endothelial damage, scarring | Recurrent DVT at same site |
| IV drug use | Repeated injection trauma, infection | Femoral vein "groin hitting" |
| Chemotherapy | Direct endothelial toxicity | Cisplatin, L-asparaginase |
| Vasculitis | Inflammatory endothelial damage | Behcet's disease, SLE |
| Hyperhomocysteinaemia | Oxidative endothelial damage | B12/folate deficiency |
The Coagulation Cascade in DVT
Initiation Phase: Tissue factor exposed on damaged endothelium or activated monocytes binds Factor VIIa, forming the extrinsic tenase complex. This generates small amounts of thrombin (Factor IIa).
Amplification Phase: Thrombin activates platelets and factors V, VIII, and XI on the platelet surface, creating a procoagulant surface for the propagation phase.
Propagation Phase: The intrinsic tenase (VIIIa-IXa) and prothrombinase (Va-Xa) complexes assemble on activated platelets, generating a "thrombin burst" sufficient to convert fibrinogen to fibrin.
Stabilisation: Factor XIIIa cross-links fibrin strands, creating a stable clot resistant to fibrinolysis.
Thrombus Evolution and Natural History
Formation: Thrombus typically originates in the soleal sinuses or valve pockets of the calf veins, where flow is slowest. The initial platelet-rich "white thrombus" rapidly incorporates red cells and fibrin to become a "red thrombus."
Propagation: Untreated thrombus can extend proximally into the popliteal, femoral, and iliac veins over days to weeks. Proximal extension dramatically increases PE risk.
Embolisation: A portion of the thrombus may detach, travel through the right heart, and lodge in the pulmonary arterial tree. Proximal DVTs (popliteal and above) have a 50% PE risk if untreated. [1]
Resolution: The body's fibrinolytic system (plasmin) gradually dissolves the clot over weeks to months. Complete resolution occurs in 50-70% of cases.
Scarring and Sequelae: Incomplete resolution leads to residual vein obstruction, valve destruction, and reflux. This chronic venous damage underlies post-thrombotic syndrome (PTS), affecting 20-50% of DVT patients. [13]
Exam Detail: Molecular Pathophysiology for Postgraduate Examinations:
The balance between clot formation and resolution depends on:
- Procoagulant factors: Tissue factor, activated clotting factors, platelets
- Anticoagulant factors: Antithrombin, Protein C/S, tissue factor pathway inhibitor (TFPI)
- Fibrinolytic system: Plasminogen → plasmin (via tPA), inhibited by PAI-1
- Endothelial function: Prostacyclin (PGI2), nitric oxide (NO), thrombomodulin
Key Regulatory Pathways:
- Protein C pathway: Thrombin + thrombomodulin → activates Protein C → inactivates Va and VIIIa (Protein S as cofactor)
- Antithrombin: Serine protease inhibitor that neutralises thrombin, Xa, IXa, XIa (potentiated 1000x by heparin)
- TFPI: Inhibits tissue factor-VIIa complex
- Fibrinolysis: Plasminogen activators (tPA, uPA) convert plasminogen to plasmin, which degrades fibrin to D-dimers
4. Clinical Presentation
Anatomical Classification
Understanding DVT location is critical as it determines PE risk and management intensity.
Distal (Isolated Calf) DVT:
- Location: Posterior tibial, anterior tibial, peroneal veins; muscular veins (soleal, gastrocnemius)
- Prevalence: 40-50% of all lower limb DVTs
- PE risk: Low (1-5% if isolated and non-propagating)
- Management: Can consider surveillance ultrasound vs anticoagulation (controversial)
Proximal DVT:
- Location: Popliteal vein and above (femoral, common femoral, iliac veins)
- Prevalence: 50-60% of all lower limb DVTs
- PE risk: High (50% if untreated)
- Management: Mandatory anticoagulation
Iliofemoral DVT:
- Location: Common femoral and/or iliac veins
- Features: Extensive swelling (entire leg), higher risk of PTS, may warrant early intervention
- Complications: Phlegmasia, higher recurrence rate
Symptoms
The classic symptom triad of pain, swelling, and warmth is present in only 50% of cases. Many DVTs are subclinical until they embolise.
| Symptom | Frequency | Clinical Significance |
|---|---|---|
| Unilateral leg swelling | 80-90% | Most common presenting complaint |
| Calf pain/tenderness | 70-75% | Often described as "cramp" or "pulled muscle" |
| Erythema/warmth | 50-60% | Indicates inflammation |
| Heaviness/aching | 40-50% | May be subtle, worse with standing |
| Distended superficial veins | 20-30% | Collateral flow around obstruction |
| Dyspnoea/chest pain | 10-30% | May indicate concurrent PE |
Asymptomatic DVT: Up to 50% of DVTs are clinically silent, particularly in hospitalised or post-operative patients. These are often detected incidentally or when PE symptoms develop. [2]
Physical Examination Findings
| Sign | Technique | Sensitivity | Specificity |
|---|---|---|---|
| Calf circumference difference > 3cm | Measure 10cm below tibial tuberosity | 40-50% | 70-80% |
| Unilateral pitting oedema | Assess pretibial area | 60-70% | 60-70% |
| Localised tenderness | Palpate along deep veins | 50-60% | 50-60% |
| Warmth | Compare with contralateral limb | 40-50% | 60-70% |
| Distended superficial veins | Visual inspection | 20-30% | 80-90% |
Homan's Sign (Historical Interest Only):
- Technique: Forced dorsiflexion of the foot causes calf pain
- Sensitivity: 10-50% (highly variable, unreliable)
- Specificity: 40-60%
- Clinical Recommendation: DO NOT PERFORM
- Reasons: Poor diagnostic accuracy, theoretical risk of dislodging thrombus, causes patient discomfort without clinical benefit [14]
Red Flag Presentations (Emergency)
⚠️ Warning: Limb-Threatening Emergencies:
Phlegmasia Cerulea Dolens ("Painful Blue Swelling")
- Pathophysiology: Massive iliofemoral thrombosis causing complete venous outflow obstruction. Venous pressure rises, compressing arterial inflow. Leads to venous gangrene if untreated.
- Clinical features: Severely swollen, cyanotic (blue) leg, exquisitely painful, absent pulses (late)
- Progression: Compartment syndrome → Arterial compromise → Gangrene → Amputation (50% without intervention)
- Management: Emergency vascular surgery referral (thrombectomy or catheter-directed thrombolysis)
Phlegmasia Alba Dolens ("Painful White Swelling")
- Pathophysiology: Iliofemoral thrombosis with arterial spasm but preserved arterial inflow
- Clinical features: Pale (white) swollen leg, painful, often in pregnancy ("milk leg")
- Significance: Precursor to Phlegmasia Cerulea Dolens
- Management: Urgent anticoagulation, consider intervention if progressive
Concurrent PE Symptoms
- Dyspnoea, chest pain, haemoptysis, syncope, hypotension
- Immediate CTPA if suspected
- Consider thrombolysis if massive PE with haemodynamic instability
Clinical Vignettes
Vignette 1: The Business Traveller
Patient: 45-year-old male, IT consultant HPC: Flew London to Sydney (22 hours economy class) 3 days ago. Developed progressive left calf pain, initially thought "pulled muscle" on the flight. Now swelling noted. PMH: Nil significant. BMI 28. Non-smoker. Exam: Left calf circumference 4cm greater than right, pitting oedema, mild warmth, tenderness along posterior calf. Wells Score: Calf swelling > 3cm (+1), Entire leg swollen (+1), Pitting oedema (+1), Tenderness along deep veins (+1) = 4 (DVT Likely) Investigation: Proceed directly to compression ultrasonography → Popliteal DVT confirmed Management: Rivaroxaban (15mg BD x 21 days, then 20mg OD) Duration: 3 months (provoked by transient risk factor - travel)
Vignette 2: The Silent Emboliser
Patient: 68-year-old female HPC: 6 weeks post total knee replacement. Presents with sudden breathlessness and pleuritic chest pain. Denies leg symptoms. Exam: Tachypnoea (RR 24), tachycardia (HR 110), oxygen saturation 92% on air. Legs appear normal bilaterally. Investigation: CTPA → Bilateral PE. Compression USS → Residual femoral DVT Key Learning Point: PE can occur without clinical evidence of DVT. The clot has already embolised. Always consider VTE prophylaxis failure in post-operative patients.
Vignette 3: The Recurrent Presenter
Patient: 32-year-old female HPC: Third episode of DVT in 4 years. First episode age 26 (no clear trigger). Second episode during pregnancy at 28 weeks. PMH: Two early miscarriages. Livedo reticularis noted on examination. Investigations: Lupus anticoagulant positive (2 occasions), Anti-cardiolipin IgG elevated Diagnosis: Antiphospholipid Syndrome Management: Lifelong anticoagulation with WARFARIN (not DOAC - triple-positive APS) Key Learning Point: Unprovoked DVT less than 50 years + recurrent VTE + pregnancy morbidity = investigate for APS
5. Differential Diagnosis
The unilateral swollen leg has a broad differential. Clinical assessment combined with the Wells score helps prioritise investigations.
| Differential | Key Distinguishing Features | Investigation |
|---|---|---|
| Deep Vein Thrombosis | Risk factors present, unilateral, pitting oedema | Compression USS |
| Cellulitis | Fever, skin erythema with defined margin, portal of entry, lymphangitis | Clinical diagnosis, elevated CRP/WCC |
| Baker's cyst rupture | Sudden onset "pop," knee pathology history, crescent sign (ecchymosis around ankle) | Knee/calf ultrasound |
| Superficial thrombophlebitis | Palpable cord, localised erythema and tenderness, along superficial vein course | Clinical, consider USS if near junction |
| Muscle tear/haematoma | History of trauma/exertion, ecchymosis, localised muscle tenderness | Clinical, consider USS |
| Lymphoedema | Chronic, non-pitting, positive Stemmer's sign, squared toes | Clinical, lymphoscintigraphy |
| Chronic venous insufficiency | Bilateral (often asymmetric), varicosities, haemosiderin staining, gaiter area ulcers | Venous duplex |
| Heart failure | Bilateral, raised JVP, pulmonary crackles, orthopnoea | Echo, BNP |
| Hypoalbuminaemia | Bilateral, periorbital oedema, liver/renal disease | Albumin, urinalysis |
| Dependent oedema | Bilateral, worse evening, immobility, no other features | Clinical |
| Compartment syndrome | Tense swelling, pain out of proportion, pain on passive stretch, paraesthesia | Compartment pressure measurement |
Baker's Cyst Rupture (Pseudothrombophlebitis)
This is the most important mimic of DVT and occurs when a popliteal synovial cyst ruptures, releasing fluid that tracks down the calf:
Mechanism: Knee pathology (typically osteoarthritis or inflammatory arthritis) → Synovial effusion → Posterior herniation through knee capsule → Baker's cyst → Rupture
Clinical Features:
- Sudden calf pain (often felt as a "pop")
- Swelling in posterior calf
- Crescent sign: Ecchymosis around the medial malleolus (gravity-dependent tracking of synovial fluid)
- History of knee pain/stiffness
Differentiation from DVT:
- Acute onset (DVT usually gradual)
- History of knee problems
- Crescent sign (not seen in DVT)
- Ultrasound shows fluid collection rather than intravascular thrombus
Important: Baker's cyst can COEXIST with DVT (the immobility and inflammation are risk factors). Always consider both diagnoses if clinical suspicion is high.
Superficial Thrombophlebitis
Thrombosis of the superficial venous system (great or small saphenous veins) presents differently from DVT but can extend into the deep system:
Clinical Features: Palpable, tender cord along superficial vein course; localised erythema and warmth; usually self-limiting
Extension Risk: Thrombus within 5cm of the saphenofemoral junction (SFJ) has a 6-15% risk of extending into the deep system. The CALISTO trial demonstrated that fondaparinux 2.5mg daily for 45 days reduced VTE events by 85% in this population. [15]
Management:
- Isolated, distant from junctions: NSAIDs, compression, mobilisation
- Within 5cm of SFJ or extensive (> 5cm): Prophylactic anticoagulation (fondaparinux or LMWH) for 45 days
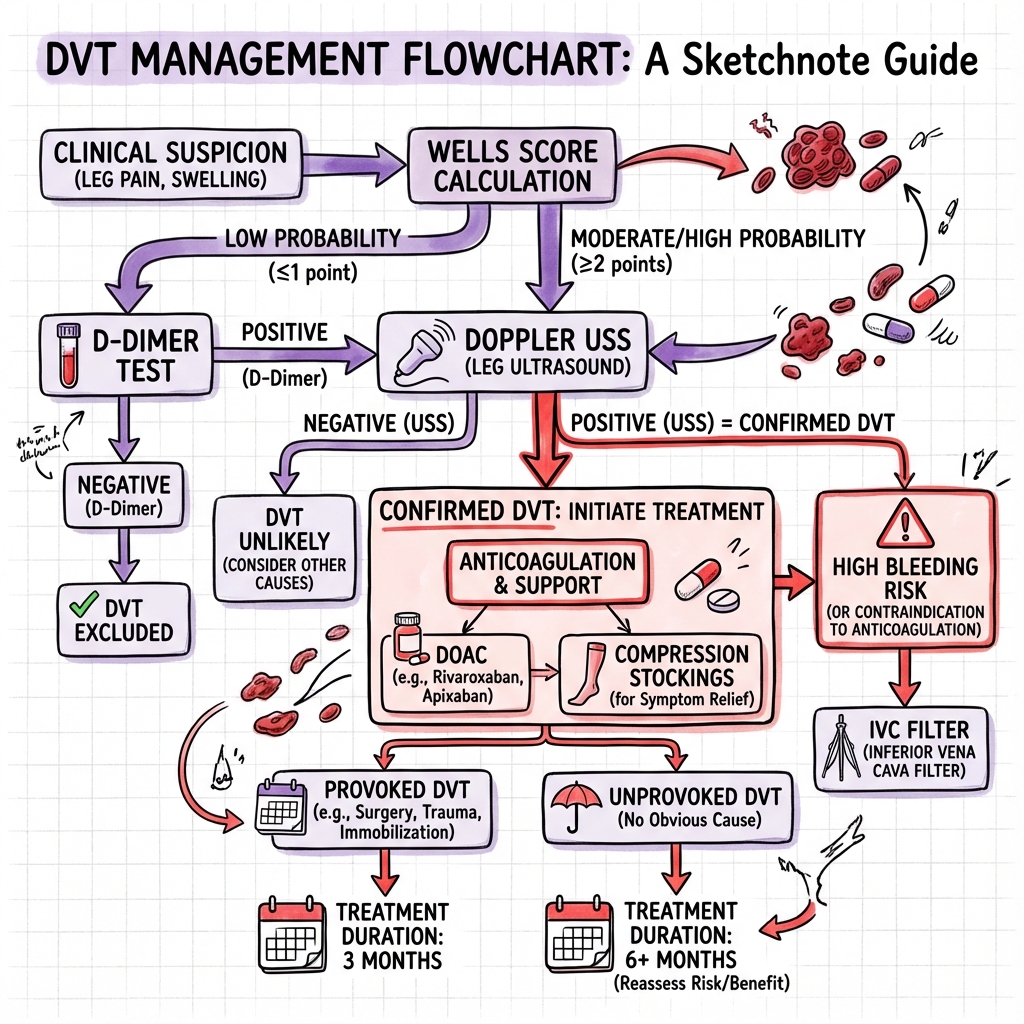
6. Clinical Assessment: The Wells Score
Two-Level Wells Score for DVT
The Wells score is a validated clinical prediction rule that estimates pre-test probability of DVT. It was developed by Wells et al. and has been validated in multiple settings. The score dichotomises patients into "DVT Likely" or "DVT Unlikely" categories, guiding subsequent investigation. [14,16]
| Clinical Feature | Points |
|---|---|
| Active cancer (treatment within 6 months or palliative) | +1 |
| Paralysis, paresis, or recent plaster immobilisation of lower extremity | +1 |
| Recently bedridden > 3 days OR major surgery within 12 weeks requiring general/regional anaesthesia | +1 |
| Localised tenderness along distribution of deep venous system | +1 |
| Entire leg swollen | +1 |
| Calf swelling > 3cm compared to asymptomatic leg (measured 10cm below tibial tuberosity) | +1 |
| Pitting oedema confined to symptomatic leg | +1 |
| Collateral superficial veins (non-varicose) | +1 |
| Previously documented DVT | +1 |
| Alternative diagnosis at least as likely as DVT | -2 |
Score Interpretation
| Score | Category | Pre-test Probability | Recommended Action |
|---|---|---|---|
| ≥2 | DVT Likely | 28-53% | Proceed directly to compression ultrasonography |
| less than 2 | DVT Unlikely | 5-8% | Perform D-dimer first; if negative, DVT excluded |
Clinical Decision Algorithm
SUSPECTED DVT
│
▼
Calculate Wells Score
│
├─── Score ≥2 (DVT Likely) ──────────────────────────────────────┐
│ │
▼ ▼
Score less than 2 (DVT Unlikely) Compression Ultrasonography
│ │
▼ ┌───────────┴───────────┐
D-dimer Test │ │
│ POSITIVE NEGATIVE
├─── NEGATIVE ─── DVT EXCLUDED ◄──────────────────────┼ │
│ (No further testing) │ ▼
▼ ▼ If high clinical suspicion:
POSITIVE START ANTICOAGULATION Repeat USS in 1 week
│ (to detect calf DVT
▼ propagation)
Compression Ultrasonography
│
├─── POSITIVE ──► START ANTICOAGULATION
│
▼
NEGATIVE
│
▼
DVT EXCLUDED (in most cases)
OR
Repeat USS in 1 week if clinical suspicion remains high
Validation and Performance
The Wells score has been extensively validated in prospective studies:
| Study | Setting | DVT Unlikely (prevalence) | DVT Likely (prevalence) |
|---|---|---|---|
| Wells 1995 | Primary care | 3% | 27% |
| Wells 2003 | Emergency department | 6% | 28% |
| Christopher Study | Multicentre | 5% | 53% |
Sensitivity and Specificity of Combined Approach (Wells + D-dimer):
- Sensitivity for excluding DVT (low probability + negative D-dimer): 99.5%
- 3-month VTE rate when DVT excluded by this algorithm: 0.4-0.5% [16]
Clinical Pearl: Pitfalls of the Wells Score:
- Subjective element: "Alternative diagnosis equally likely" requires clinical judgement and can vary between clinicians
- Cancer patients: May always score ≥1 even without symptoms, reducing specificity
- Recurrent DVT: Previous DVT (+1) doesn't distinguish fresh from old symptoms
- Elderly: High baseline D-dimer (age-adjusted threshold may help)
- Pregnancy: D-dimer physiologically elevated; Wells not validated; low threshold for imaging
7. Investigations
7.1 D-dimer
Physiology: D-dimer is a fibrin degradation product released when cross-linked fibrin is lysed by plasmin. Its presence indicates active fibrin turnover (clot formation AND breakdown).
Clinical Utility: D-dimer has HIGH SENSITIVITY but LOW SPECIFICITY for DVT. Its primary role is to EXCLUDE DVT in patients with low pre-test probability (DVT Unlikely on Wells score).
Assay Types:
- ELISA (laboratory-based): Most sensitive (95-98%), takes hours
- Latex agglutination (point-of-care): Less sensitive (85-90%), rapid results
- Immunoturbidimetric: Intermediate sensitivity, automated
| Parameter | Value |
|---|---|
| Sensitivity | 95-98% |
| Specificity | 40-50% |
| Negative Predictive Value (low probability) | 99% |
| Positive Predictive Value | 15-25% |
Interpretation:
- Negative D-dimer + Low clinical probability (Wells less than 2): DVT effectively EXCLUDED. No imaging required.
- Positive D-dimer: Proceed to ultrasound (does NOT confirm DVT, only indicates need for imaging)
Causes of False-Positive D-dimer (Elevated without VTE):
- Recent surgery or trauma
- Active malignancy
- Pregnancy
- Infection/sepsis
- Inflammation (CRP correlates with D-dimer)
- Advanced age
- Liver disease
- Renal impairment
- Aortic dissection
- Recent hospitalisation
Age-Adjusted D-dimer Threshold: Traditional threshold: 500 ng/mL (or 0.5 mg/L FEU) Age-adjusted threshold (for patients > 50 years): Age × 10 ng/mL
Example: 70-year-old patient → threshold = 700 ng/mL
The ADJUST-PE trial validated age-adjusted D-dimer for PE and has been extrapolated to DVT. Using age-adjusted thresholds increases specificity by 10-15% in elderly patients without compromising safety. [17]
Exam Detail: When NOT to Perform D-dimer:
- High clinical probability (Wells ≥2): Go straight to imaging (D-dimer adds no value)
- Post-operative patients: Almost always elevated
- Hospitalised patients: Multiple confounders
- Known malignancy: Frequently elevated
- Pregnancy: Physiologically elevated
- Already anticoagulated: May be falsely suppressed
Key Exam Point: D-dimer is a RULE-OUT test for low-probability patients. It should NEVER be used to RULE-IN DVT or in high-risk patients.
7.2 Compression Ultrasonography (CUS)
Gold Standard Investigation for DVT diagnosis.
Technique: A high-frequency linear probe (7-12 MHz) is applied to the deep veins from groin to ankle. The vein is compressed with the probe. A normal vein collapses completely; a thrombosed vein does not compress (the thrombus prevents coaptation of the walls).
Types of Ultrasound Protocols:
| Protocol | Anatomical Coverage | Sensitivity (Proximal) | Sensitivity (Distal) |
|---|---|---|---|
| Proximal CUS (2-point) | Common femoral + Popliteal only | 97% | ~50% (not assessed) |
| Whole-leg CUS | Groin to ankle (all deep veins) | 97% | 80-90% |
| Extended CUS | Includes calf veins with Doppler | 97% | 90-95% |
Advantages of Whole-Leg vs 2-Point CUS:
- Detects isolated calf DVT (avoided serial scanning)
- Single examination definitive
- Identifies alternative diagnoses (Baker's cyst, muscle haematoma)
Disadvantages:
- More time-consuming
- Operator-dependent for calf veins
- May detect clinically insignificant distal thrombi
Ultrasound Findings in DVT:
- Non-compressibility (PRIMARY diagnostic criterion): Vein fails to collapse with probe pressure
- Echogenic intraluminal thrombus: May be visualised, especially in acute DVT
- Absent or reduced colour flow: Doppler signal absent or diminished
- Dilated vein: Acute thrombus often distends the vein
- Loss of respiratory phasicity: Normally, venous flow varies with respiration
Acute vs Chronic DVT on Ultrasound:
| Feature | Acute DVT | Chronic DVT |
|---|---|---|
| Echogenicity | Hypoechoic (dark) | Hyperechoic (bright) |
| Vein diameter | Dilated | Normal or narrowed |
| Thrombus texture | Homogeneous, soft | Heterogeneous, calcified |
| Compressibility | Completely non-compressible | Partially compressible |
| Collaterals | Absent | Often present |
| Wall thickening | Minimal | Significant |
Serial Ultrasound Strategy: If initial proximal CUS is negative but clinical suspicion remains high:
- Repeat ultrasound in 5-7 days
- Rationale: Calf DVT may propagate proximally into popliteal (detectable range)
- This serial approach has been validated as safe (3-month VTE rate 0.6% if remains negative) [16]
7.3 Other Imaging Modalities
CT Venography (CTV):
- Indication: When iliac vein or IVC thrombosis suspected (not well visualised by USS)
- Technique: Often performed simultaneously with CTPA
- Advantage: Excellent visualisation of central veins
- Disadvantage: Radiation exposure, contrast risk
MR Venography (MRV):
- Indication: Pregnancy (no radiation), contrast allergy, pelvic vein assessment
- Advantage: No ionising radiation
- Disadvantage: Cost, availability, time, gadolinium concerns in renal impairment
Ascending Venography (Contrast Phlebography):
- Historical gold standard (now rarely used)
- Indication: Equivocal non-invasive testing, pre-intervention planning
- Disadvantage: Invasive, contrast reactions, post-procedure DVT risk
7.4 Baseline Laboratory Investigations
Before initiating anticoagulation:
| Investigation | Purpose |
|---|---|
| FBC | Baseline, exclude thrombocytopenia, HIT monitoring |
| U&E, Creatinine, eGFR | Renal function for DOAC dosing |
| LFTs | Hepatic function, bleeding risk assessment |
| Coagulation screen (PT, aPTT) | Baseline before anticoagulation, lupus anticoagulant screen |
| Urinalysis | Occult haematuria (bleeding risk) |
7.5 Thrombophilia Screening
Who Should Be Tested?
- Unprovoked DVT at age less than 50 years
- Recurrent VTE
- VTE at unusual sites (cerebral, mesenteric, portal vein)
- Strong family history of VTE (first-degree relative less than 50 years)
- Recurrent pregnancy loss
- Known family member with high-risk thrombophilia
Who Should NOT Be Tested?
- First provoked DVT (clear precipitant)
- Elderly patients with first episode
- Patients who will be anticoagulated regardless of result
Timing of Testing:
- Acute phase: Anti-phospholipid antibodies (not affected by anticoagulation)
- 2-4 weeks after stopping anticoagulation: Protein C, Protein S, Antithrombin, Factor V Leiden, Prothrombin G20210A, Lupus anticoagulant
Important: Testing during acute VTE or on anticoagulation gives unreliable results:
- Protein C/S: Reduced by warfarin
- Antithrombin: Reduced by heparin
- Lupus anticoagulant: Affected by warfarin and heparin
Thrombophilia Panel:
| Test | Condition Detected | Prevalence | VTE Risk |
|---|---|---|---|
| Factor V Leiden (G1691A) | APC resistance | 5% Caucasians | 5-80x |
| Prothrombin gene mutation (G20210A) | Elevated prothrombin | 2-3% | 2-4x |
| Protein C activity | Protein C deficiency | 0.2-0.4% | 7-10x |
| Protein S activity (free and total) | Protein S deficiency | 0.1-0.3% | 5-10x |
| Antithrombin activity | Antithrombin deficiency | 0.02-0.2% | 10-50x |
| Lupus anticoagulant | Antiphospholipid syndrome | 1-5% | 5-10x |
| Anti-cardiolipin antibodies | Antiphospholipid syndrome | 1-5% | 5-10x |
| Anti-β2-glycoprotein I antibodies | Antiphospholipid syndrome | 1-5% | 5-10x |
| Homocysteine | Hyperhomocysteinaemia | 5-10% | 2-3x |
Clinical Pearl: Practical Thrombophilia Testing Approach:
- Discuss with haematology before testing
- Counsel patient that results may affect family members and insurance
- Test only if result will change management (e.g., duration of anticoagulation, family screening)
- Most useful in young patients with unprovoked VTE or family history
- Triple-positive antiphospholipid syndrome requires warfarin over DOACs
8. Management
8.1 Goals of Treatment
- Prevent clot propagation (stop the thrombus growing)
- Prevent pulmonary embolism (stop embolisation to lungs)
- Reduce recurrence risk (prevent future VTE episodes)
- Minimise complications (prevent post-thrombotic syndrome, balance bleeding risk)
- Relieve symptoms (reduce pain and swelling)
8.2 Anticoagulation: Drug Classes
Direct Oral Anticoagulants (DOACs) - First-Line
DOACs have largely replaced LMWH/warfarin for most DVT patients based on non-inferiority in efficacy with superior safety (reduced major bleeding) and convenience (fixed dosing, no monitoring). [18,19]
Mechanism of Action:
- Rivaroxaban, Apixaban, Edoxaban: Direct Factor Xa inhibitors
- Dabigatran: Direct thrombin (Factor IIa) inhibitor
DOAC Regimens for DVT/PE:
| Drug | Initial Treatment | Maintenance Dose | Key Features |
|---|---|---|---|
| Rivaroxaban | 15mg BD × 21 days | 20mg OD (with food) | Single-drug approach, no LMWH lead-in |
| Apixaban | 10mg BD × 7 days | 5mg BD | Single-drug approach, lowest bleeding profile |
| Edoxaban | LMWH × 5-10 days | 60mg OD | Requires LMWH lead-in |
| Dabigatran | LMWH × 5-10 days | 150mg BD | Requires LMWH lead-in, specific antidote |
Landmark Trials Supporting DOACs:
| Trial | Drug | Comparison | Key Finding |
|---|---|---|---|
| EINSTEIN-DVT/PE | Rivaroxaban | Warfarin | Non-inferior efficacy, similar bleeding |
| AMPLIFY | Apixaban | Warfarin | Non-inferior efficacy, 69% reduction in major bleeding |
| HOKUSAI-VTE | Edoxaban | Warfarin | Non-inferior efficacy, 19% reduction in clinically relevant bleeding |
| RE-COVER | Dabigatran | Warfarin | Non-inferior efficacy, similar bleeding |
Renal Dosing for DOACs:
| Drug | CrCl > 50 | CrCl 30-50 | CrCl 15-30 | CrCl less than 15 |
|---|---|---|---|---|
| Rivaroxaban | 20mg OD | 20mg OD | 15mg OD | Avoid |
| Apixaban | 5mg BD | 5mg BD | 2.5mg BD* | Caution |
| Edoxaban | 60mg OD | 30mg OD | 30mg OD | Avoid |
| Dabigatran | 150mg BD | 150mg BD | 110mg BD | Avoid |
*Apixaban dose reduction to 2.5mg BD if ≥2 of: Age ≥80, Weight ≤60kg, Creatinine ≥133 μmol/L
Contraindications to DOACs:
- Severe renal impairment (CrCl less than 15-30 mL/min, drug-dependent)
- Mechanical heart valves (dabigatran shown to be harmful)
- Triple-positive antiphospholipid syndrome
- Pregnancy and breastfeeding (DOACs cross placenta)
- Active significant bleeding
- Severe hepatic impairment with coagulopathy
Low Molecular Weight Heparin (LMWH)
Mechanism: Potentiates antithrombin, primarily inhibiting Factor Xa (longer chain LMWHs also inhibit thrombin)
Common LMWH Preparations:
| Drug | Treatment Dose | Prophylaxis Dose |
|---|---|---|
| Enoxaparin | 1mg/kg BD or 1.5mg/kg OD | 40mg OD |
| Dalteparin | 200 units/kg OD (max 18,000 units) | 5,000 units OD |
| Tinzaparin | 175 units/kg OD | 4,500 units OD |
Advantages:
- Rapid onset of action (peak 3-5 hours)
- Predictable pharmacokinetics
- No routine monitoring required
- Safe in pregnancy
- Partially reversible with protamine
Indications for LMWH Over DOAC:
- Pregnancy (DOACs teratogenic)
- Severe renal impairment (LMWH can be dose-adjusted with Anti-Xa monitoring)
- Cancer-associated thrombosis (though DOACs now acceptable for many cancers)
- Bridging for procedures
- Patient preference (injectable)
Unfractionated Heparin (UFH)
Indication: Critically ill patients, high bleeding risk (rapidly reversible), severe renal failure, pre-procedure bridging
Dosing: Loading dose 80 units/kg IV, then 18 units/kg/hour continuous infusion Monitoring: aPTT target 1.5-2.5x control (varies by assay) Reversal: Protamine sulphate (1mg per 100 units heparin given in previous 2-3 hours)
Vitamin K Antagonists (Warfarin)
Mechanism: Inhibits vitamin K-dependent synthesis of factors II, VII, IX, X and proteins C and S
Indications for Warfarin Over DOAC:
- Mechanical heart valves
- Triple-positive antiphospholipid syndrome
- Severe renal failure (CrCl less than 15)
- Drug interactions making DOACs unsuitable
- Patient preference/established stable therapy
- Cost considerations in some healthcare systems
Initiation:
- Start with LMWH bridge (overlapping for ≥5 days AND until INR ≥2.0 for ≥24 hours)
- Initial warfarin dose: Usually 5mg daily (lower in elderly, hepatic disease, drug interactions)
- INR target: 2.0-3.0
Warfarin Counselling Points:
- Consistent vitamin K intake (green vegetables) - don't suddenly change diet
- Avoid alcohol binges (unpredictable INR swings)
- Multiple drug interactions (antibiotics, NSAIDs, statins, etc.)
- Carry anticoagulation alert card ("Yellow Book" in UK)
- Regular INR monitoring (initially weekly, then monthly when stable)
8.3 Treatment Duration
Duration of anticoagulation depends on whether the DVT was provoked (identifiable risk factor) or unprovoked (no clear trigger), and the recurrence risk.
PROVOKED DVT (Transient Risk Factor):
- Major surgery within 12 weeks
- Significant trauma with immobility
- Prolonged immobilisation (> 3 days)
- Long-haul travel (> 4-6 hours)
- Oral contraceptive use
- Pregnancy/puerperium
Duration: 3 months (risk factor resolved)
PROVOKED DVT (Persistent Risk Factor):
- Active malignancy
- Chronic immobility (paralysis, severe disability)
Duration: At least 3-6 months, often indefinite while risk factor persists
UNPROVOKED DVT (No Identifiable Trigger):
- No surgery, trauma, immobilisation, or hormonal trigger
- Higher recurrence risk (10% year 1, 30% at 5 years, 50% at 10 years)
Duration: Minimum 3 months, then reassess for extended anticoagulation
Extended Anticoagulation Decision Factors:
| Factor | Favours Extended (> 3 months) | Favours Limited (3 months) |
|---|---|---|
| Sex | Male (higher recurrence) | Female (lower recurrence) |
| D-dimer (off anticoagulation) | Elevated | Normal |
| Residual vein obstruction | Present | Absent |
| Location | Proximal/iliofemoral | Isolated distal |
| Bleeding risk | Low (HAS-BLED ≤2) | High (HAS-BLED ≥3) |
| Patient preference | Prefers continued protection | Prefers stopping |
| Prior VTE | Recurrent episodes | First episode |
RECURRENT VTE:
- Duration: Indefinite (lifelong) unless prohibitive bleeding risk
CANCER-ASSOCIATED THROMBOSIS:
- Duration: Minimum 6 months, usually indefinite while cancer active
- Continue until cancer resolved or life expectancy limited by cancer
- LMWH was historical standard; DOACs (edoxaban, rivaroxaban) now acceptable except GI/GU malignancy (increased bleeding) [20]
Exam Detail: Extended Anticoagulation Options:
After initial 3-6 month treatment, options for extended therapy include:
- Full-dose DOAC continuation (standard extended therapy)
- Reduced-dose DOAC:
- Rivaroxaban 10mg OD (EINSTEIN CHOICE trial: 83% reduction in recurrence vs placebo)
- Apixaban 2.5mg BD (AMPLIFY-EXT: 67% reduction in recurrence vs placebo)
- Bleeding risk similar to aspirin but better efficacy
- Aspirin (if anticoagulation not tolerated): Modest benefit (30% reduction vs placebo in WARFASA/ASPIRE trials) but inferior to anticoagulation [21]
Shared Decision-Making: Discuss individual bleeding risk, patient preferences, and lifestyle factors when deciding duration. Male sex, elevated D-dimer after stopping anticoagulation, and residual vein thrombus predict higher recurrence.
8.4 Bleeding Management and Reversal
Risk Assessment: Before anticoagulation, assess bleeding risk using tools like HAS-BLED:
| HAS-BLED Component | Points |
|---|---|
| Hypertension (uncontrolled, SBP > 160) | 1 |
| Abnormal renal/liver function | 1-2 |
| Stroke history | 1 |
| Bleeding history or predisposition | 1 |
| Labile INR (if on warfarin) | 1 |
| Elderly (> 65) | 1 |
| Drugs (antiplatelets, NSAIDs) or alcohol | 1-2 |
Score ≥3: High bleeding risk - requires closer monitoring, not necessarily contraindication to anticoagulation
Management of Bleeding:
| Bleeding Severity | Management |
|---|---|
| Minor (bruising, epistaxis less than 10 min) | Local measures, consider dose reduction, continue anticoagulation |
| Clinically relevant non-major | Hold anticoagulation temporarily, investigate source, tranexamic acid, resume when safe |
| Major (Hb drop ≥2g/dL, transfusion required, critical site) | Stop anticoagulation, reversal agent, resuscitation, source control |
| Life-threatening (intracranial, retroperitoneal, critical organ) | Immediate reversal, intensive care, surgical intervention if needed |
Reversal Agents:
| Anticoagulant | Reversal Agent | Dose |
|---|---|---|
| Warfarin | Vitamin K (slow) + PCC (fast) | Vitamin K 5-10mg IV; 4-factor PCC 25-50 units/kg |
| Dabigatran | Idarucizumab (Praxbind) | 5g IV (2 × 2.5g vials) |
| Rivaroxaban/Apixaban | Andexanet alfa (if available) OR PCC | PCC 50 units/kg |
| LMWH | Protamine (partial reversal) | 1mg per 100 units LMWH (max 50mg) |
| UFH | Protamine (complete reversal) | 1mg per 100 units UFH (given in past 2-3 hours) |
8.5 Special Populations
Pregnancy
- VTE risk increased 5-10× during pregnancy (hypercoagulability, stasis from uterus, endothelial changes)
- Highest risk in third trimester and puerperium (first 6 weeks postpartum)
Diagnosis in Pregnancy:
- D-dimer: Physiologically elevated (limited utility)
- Compression ultrasound: First-line for suspected DVT
- MR venography: If iliac veins need assessment (no radiation)
Treatment:
- LMWH is the drug of choice (DOACs contraindicated - teratogenic; warfarin causes embryopathy if used first trimester)
- Treatment dose throughout pregnancy
- Hold LMWH when labour begins (resume 6-12 hours post-delivery if no epidural concerns)
- Continue for minimum 6 weeks postpartum (at least 3 months total treatment)
- Can convert to warfarin or DOAC postpartum (safe in breastfeeding for warfarin; limited data for DOACs)
Cancer-Associated Thrombosis (CAT)
- VTE occurs in 4-20% of cancer patients
- Malignancy increases VTE risk 4-7× (tissue factor expression, chemotherapy, immobility, central lines)
Treatment Approach: [20]
- Traditional: LMWH preferred over warfarin (30% reduction in recurrence in CLOT trial)
- Current: DOACs (edoxaban, rivaroxaban) are non-inferior to LMWH for most cancers
- Exception: GI and genitourinary cancers have higher GI bleeding risk with DOACs - prefer LMWH
- Duration: Minimum 6 months, often indefinite while cancer active
Evidence:
- HOKUSAI-VTE Cancer: Edoxaban non-inferior to dalteparin for recurrence, higher major bleeding (especially GI cancer)
- SELECT-D: Rivaroxaban lower recurrence than dalteparin, higher clinically relevant non-major bleeding
Renal Impairment
- All anticoagulants affected by renal function to varying degrees
- LMWH: Accumulates with CrCl less than 30; dose-reduce or use UFH
- DOACs: See renal dosing table above; avoid in severe renal failure
- Warfarin: Not affected by renal function (but bleeding risk higher in renal impairment)
Elderly
- Higher VTE and bleeding risk
- Falls risk assessment important
- Age-adjusted D-dimer threshold (age × 10 ng/mL)
- Consider reduced DOAC doses based on age, weight, renal function
Antiphospholipid Syndrome
- Triple-positive APS (all 3 antibody types): DOACs CONTRAINDICATED
- TRAPS trial: Rivaroxaban had higher thrombosis rate than warfarin in triple-positive APS
- Warfarin with target INR 2-3 (or higher in some cases) is standard of care [12]
8.6 Advanced Interventions
Catheter-Directed Thrombolysis (CDT)
Indication: Acute symptomatic iliofemoral DVT (symptom onset less than 14-21 days), low bleeding risk, good functional status, life expectancy > 1 year
Technique: Catheter placed directly into thrombus via popliteal or femoral vein approach; thrombolytic (tPA, alteplase) infused over 12-24 hours
Evidence: The ATTRACT trial showed CDT reduced post-thrombotic syndrome severity but did not significantly reduce overall PTS incidence. CDT increased major bleeding (1.7% vs 0.3%). Current guidelines suggest CDT may be considered in carefully selected patients but is not routinely recommended. [22]
Contraindications to Thrombolysis:
- Absolute: Active bleeding, recent stroke (less than 3 months), intracranial pathology, recent major surgery/trauma
- Relative: Pregnancy, recent surgery (> 10 days), severe hypertension
Pharmacomechanical Thrombolysis
Combines catheter-directed thrombolysis with mechanical clot disruption/aspiration. May reduce thrombolytic dose and treatment time. Evidence still evolving.
IVC Filters
Indication: Absolute contraindication to anticoagulation with acute proximal DVT or PE
Types:
- Retrievable: Designed to be removed once anticoagulation can be resumed (ideally within 3-6 months)
- Permanent: Only for patients with truly permanent contraindications to anticoagulation
Complications:
- Filter thrombosis (2-10%)
- IVC penetration
- Filter migration
- Filter fracture
- Increased long-term DVT recurrence
Current Practice: IVC filters are placed far less frequently than in the past. Most patients can be anticoagulated. The PREPIC2 trial showed no benefit of routine filter placement in addition to anticoagulation for patients with severe PE. [23]
Key Point: If a retrievable filter is placed, document a retrieval plan. Filter retrieval rates remain disappointingly low (20-40% in many series).
8.7 Outpatient vs Inpatient Management
Most uncomplicated DVT can be managed entirely as an outpatient with DOAC therapy. This is supported by multiple randomised trials and guideline recommendations.
Criteria for Outpatient Management:
- Haemodynamically stable
- Low bleeding risk
- Good renal function (for DOAC dosing)
- Able to comply with treatment and follow-up
- No severe symptoms requiring analgesia/elevation
- No suspected PE requiring further workup
- Adequate social support
Criteria for Inpatient Admission:
- Suspected or confirmed PE
- Haemodynamic instability
- High bleeding risk requiring monitoring
- Phlegmasia (limb-threatening)
- Severe pain or massive swelling
- Significant comorbidities
- Unable to comply with outpatient treatment
- Renal impairment requiring dose adjustment/monitoring
- Anticipated intervention (thrombolysis, thrombectomy)
9. Complications
9.1 Pulmonary Embolism
Risk: 50% of untreated proximal DVTs will embolise
Mechanism: Thrombus (or portion thereof) detaches from deep vein → travels through IVC → right heart → lodges in pulmonary arterial tree
Clinical Features:
- Dyspnoea (most common)
- Pleuritic chest pain
- Haemoptysis
- Tachycardia, tachypnoea
- Hypoxia
- Syncope (suggests massive PE)
- Hypotension/shock (massive PE with RV failure)
Severity Classification:
| Category | Haemodynamics | RV Dysfunction | Troponin | 30-Day Mortality |
|---|---|---|---|---|
| Low risk | Stable | No | Normal | less than 1% |
| Intermediate-low | Stable | Yes OR elevated troponin (not both) | 3-7% | |
| Intermediate-high | Stable | Yes AND elevated | Elevated | 7-15% |
| High risk (massive) | Shock/hypotension | Yes | Usually elevated | 25-65% |
Management of PE:
- Anticoagulation (as for DVT)
- High-risk/massive PE: Consider thrombolysis (alteplase 100mg IV over 2 hours) or embolectomy
- Intermediate-high risk: Close monitoring, consider thrombolysis if deterioration
9.2 Post-Thrombotic Syndrome (PTS)
Definition: Chronic clinical manifestations of venous insufficiency following DVT
Incidence: 20-50% of DVT patients within 2 years [13]
Pathophysiology:
- Venous obstruction: Residual thrombus causes outflow obstruction
- Valvular incompetence: Thrombus damages venous valves → reflux
- Venous hypertension: Combination of obstruction and reflux → sustained high venous pressure
- Tissue damage: Chronic venous hypertension → inflammation → skin changes → ulceration
Clinical Features of PTS:
- Chronic leg pain (worse with standing/activity, better with rest/elevation)
- Heaviness, aching
- Oedema (pitting → non-pitting as chronic)
- Skin changes: Hyperpigmentation (haemosiderin), eczema, lipodermatosclerosis
- Venous ulceration (gaiter area - medial lower leg)
Villalta Score for PTS Diagnosis and Severity:
| Symptom/Sign | 0 (Absent) | 1 (Mild) | 2 (Moderate) | 3 (Severe) |
|---|---|---|---|---|
| Pain | ✓ | ✓ | ✓ | ✓ |
| Cramps | ✓ | ✓ | ✓ | ✓ |
| Heaviness | ✓ | ✓ | ✓ | ✓ |
| Paraesthesia | ✓ | ✓ | ✓ | ✓ |
| Pruritus | ✓ | ✓ | ✓ | ✓ |
| Pretibial oedema | ✓ | ✓ | ✓ | ✓ |
| Skin induration | ✓ | ✓ | ✓ | ✓ |
| Hyperpigmentation | ✓ | ✓ | ✓ | ✓ |
| Redness | ✓ | ✓ | ✓ | ✓ |
| Venous ectasia | ✓ | ✓ | ✓ | ✓ |
| Pain on calf compression | ✓ | ✓ | ✓ | ✓ |
| Venous ulcer | Present = severe PTS regardless of score |
Interpretation: Score ≥5 or presence of ulcer = PTS present
- 5-9: Mild PTS
- 10-14: Moderate PTS
- ≥15 or ulcer: Severe PTS
Prevention of PTS:
- Elastic compression stockings: The SOX trial (2014) found NO benefit of routine compression stockings (30-40 mmHg) in preventing PTS. Current guidelines do NOT recommend routine prophylactic stockings. Stockings may be offered for symptom relief if patient finds them helpful. [13]
- Early mobilisation: Encourage walking (not bed rest)
- Optimal anticoagulation: Reduces recurrent DVT (a risk factor for PTS)
- Weight optimisation: Obesity worsens venous insufficiency
Treatment of Established PTS:
- Compression therapy (if tolerated and not contraindicated by arterial disease)
- Leg elevation
- Exercise and calf muscle strengthening
- Skin care to prevent ulceration
- Venous ulcer management: Compression, wound care, consider grafting
- Endovascular stenting for iliofemoral obstruction in severe cases
9.3 Recurrent VTE
Incidence:
- Provoked DVT (transient risk factor): 3% per year after stopping anticoagulation
- Unprovoked DVT: 10% year 1, 30% at 5 years, 50% at 10 years
- Recurrent VTE on anticoagulation: 2-3% per year
Risk Factors for Recurrence:
- Male sex (30-40% higher recurrence than females)
- Elevated D-dimer after stopping anticoagulation
- Residual vein obstruction on ultrasound
- Obesity
- Post-thrombotic syndrome
- Factor V Leiden or prothrombin mutation
- Unprovoked vs provoked DVT
Diagnosis of Recurrent DVT:
- May be challenging as baseline ultrasound may show residual thrombus
- Compare with prior imaging if available
- New symptoms + new findings = recurrent DVT
- Consider CT or MR venography if ultrasound equivocal
Management:
- If occurs ON anticoagulation: Ensure compliance, increase dose, consider switch (e.g., warfarin to LMWH, DOAC to LMWH)
- If occurs AFTER stopping: Resume anticoagulation, typically indefinitely
9.4 Heparin-Induced Thrombocytopenia (HIT)
Definition: Immune-mediated reaction to heparin causing platelet activation, thrombocytopenia, and paradoxically THROMBOSIS
Incidence:
- UFH: 1-5%
- LMWH: 0.2-0.6%
Timing: Typically 5-10 days after heparin initiation (or earlier if prior heparin exposure)
Diagnosis (4T Score):
| Parameter | 2 Points | 1 Point | 0 Points |
|---|---|---|---|
| Thrombocytopenia | > 50% fall or nadir 20-100 | 30-50% fall or nadir 10-19 | less than 30% fall or nadir less than 10 |
| Timing | Days 5-10 or ≤1 day if prior heparin | Consistent with days 5-10 | ≤4 days without recent heparin |
| Thrombosis | New thrombosis or skin necrosis | Progressive/recurrent or erythematous skin | None |
| Other causes | No other explanation | Possible other cause | Definite other cause |
4T Score Interpretation:
- 0-3: Low probability (less than 5% chance of HIT)
- 4-5: Intermediate probability (10-30%)
- 6-8: High probability (40-80%)
Management of Suspected/Confirmed HIT:
- STOP all heparin immediately (including flushes, coated catheters)
- Start alternative anticoagulation (argatroban, fondaparinux, or DOAC)
- Do NOT give warfarin until platelets recover (causes skin necrosis)
- Send HIT antibody testing (ELISA, serotonin release assay)
- Do NOT transfuse platelets (fuels thrombosis)
- Monitor for thrombosis
10. Prevention (VTE Prophylaxis)
Hospital-Acquired VTE
VTE is a leading cause of preventable hospital death. All hospitalised patients should be assessed for VTE risk.
Risk Assessment Tools:
- Department of Health VTE Risk Assessment (UK)
- Padua Prediction Score (medical patients)
- Caprini Score (surgical patients)
Mechanical Prophylaxis:
- Graduated compression stockings (TED stockings): Increase venous velocity
- "Contraindicated: Peripheral arterial disease (ABPI less than 0.8), severe oedema, skin conditions, recent skin graft"
- Must be correctly fitted; poor fit can cause tourniquet effect
- Intermittent pneumatic compression (IPC/Flowtrons): Mechanical calf compression mimicking walking
- Useful when anticoagulation contraindicated
- Must be worn for majority of day to be effective
Pharmacological Prophylaxis:
| Setting | Drug | Dose |
|---|---|---|
| Medical patients | Enoxaparin | 40mg SC OD |
| General surgery | Enoxaparin | 40mg SC OD |
| Orthopaedic (hip/knee replacement) | Enoxaparin or DOAC | Enoxaparin 40mg OD or rivaroxaban 10mg OD |
| Renal impairment (CrCl less than 30) | UFH | 5,000 units SC BD-TDS |
Duration of Prophylaxis:
- Medical patients: Duration of hospitalisation or immobility
- General surgery: Until fully mobile (usually 7-10 days)
- Major orthopaedic surgery: Extended prophylaxis 28-35 days (hip replacement), 10-14 days (knee replacement)
Extended Thromboprophylaxis Post-Hospitalisation
The MARINER trial evaluated rivaroxaban 10mg for 45 days after hospital discharge in high-risk medical patients. While VTE was reduced, bleeding increased, and there was no mortality benefit. Routine extended prophylaxis post-discharge is not currently recommended except after major orthopaedic surgery. [24]
11. Prognosis
Short-Term Outcomes
| Outcome | DVT Alone | DVT + PE |
|---|---|---|
| 30-day mortality | 1-2% | 5-15% |
| Hospital mortality | less than 1% | 3-10% |
| Major bleeding on anticoagulation | 1-3% at 3 months | 1-3% at 3 months |
Long-Term Outcomes
| Outcome | Time Frame | Rate |
|---|---|---|
| Recurrent VTE (provoked) | 10 years | 10-15% |
| Recurrent VTE (unprovoked) | 10 years | 30-40% |
| Post-thrombotic syndrome | 2 years | 20-50% |
| Chronic thromboembolic pulmonary hypertension (after PE) | 2 years | 2-4% |
Prognostic Factors
Poor Prognosis:
- Cancer-associated thrombosis
- Recurrent VTE
- Massive iliofemoral DVT
- Delay in anticoagulation
- Poor anticoagulation control
- Persistent residual vein obstruction
Good Prognosis:
- Provoked by transient risk factor
- Prompt diagnosis and treatment
- Good anticoagulation compliance
- Resolution of thrombus on follow-up imaging
12. Exam-Focused Content
Common Exam Questions and Model Answers
Q1: A 55-year-old woman presents with a 3-day history of left calf swelling and pain. Her Wells score is 4. Describe your management.
Model Answer: "With a Wells score of 4, this patient is in the 'DVT Likely' category with pre-test probability of approximately 30-50%. I would proceed directly to compression ultrasonography without D-dimer testing. If the ultrasound confirms DVT, I would initiate anticoagulation, with a DOAC such as rivaroxaban (15mg twice daily for 21 days, then 20mg once daily) being my first-line choice, provided there are no contraindications such as severe renal impairment, pregnancy, or triple-positive antiphospholipid syndrome.
Before starting anticoagulation, I would check baseline bloods including FBC, U&E, LFTs, and coagulation screen. I would assess bleeding risk using the HAS-BLED score. I would also take a focused history to identify any provoking factors (recent surgery, immobility, travel, malignancy, hormonal therapy) as this determines treatment duration.
For a provoked DVT with a clear transient risk factor, I would recommend 3 months of anticoagulation. For an unprovoked DVT, I would recommend a minimum of 3 months followed by reassessment of risks and benefits of extended therapy.
I would counsel the patient on bleeding risks, signs of PE (breathlessness, chest pain), and the importance of compliance. If she is on oestrogen-containing contraception, this should be stopped permanently."
Q2: What are the components of Virchow's triad? Give clinical examples of each.
Model Answer: "Virchow's triad describes the three factors that predispose to thrombosis:
-
Venous stasis (abnormal blood flow)
- Prolonged immobilisation (hospitalisation, long-haul travel)
- Plaster cast immobilisation
- Paralysis or paresis (stroke, spinal injury)
- Heart failure (reduced cardiac output)
- Obesity (compression of iliac veins)
-
Hypercoagulability (abnormal blood composition)
- Inherited: Factor V Leiden, Protein C/S deficiency, Antithrombin deficiency
- Acquired: Malignancy, pregnancy, oral contraceptives, antiphospholipid syndrome
- Nephrotic syndrome (loss of antithrombin III)
- Myeloproliferative disorders
-
Endothelial injury (abnormal vessel wall)
- Surgery (especially hip/knee replacement)
- Trauma
- Central venous catheters
- Previous DVT with venous scarring
- IV drug use with femoral vein injection
- Chemotherapy-induced endothelial damage
Most clinical DVTs result from a combination of two or more of these factors acting synergistically."
Q3: A patient with DVT on rivaroxaban presents with a major GI bleed. How would you manage this?
Model Answer: "This is a medical emergency requiring immediate resuscitation and reversal.
Immediate management:
- Stop rivaroxaban immediately
- ABC assessment - establish IV access (large bore × 2), oxygen if hypoxic
- Urgent bloods: FBC, coagulation screen, group and save, crossmatch
- Fluid resuscitation, blood transfusion if needed (maintain Hb > 7-8 g/dL)
Reversal: 5. Andexanet alfa is the specific reversal agent for factor Xa inhibitors but may not be available. If unavailable, give 4-factor prothrombin complex concentrate (PCC) at 50 units/kg 6. Tranexamic acid 1g IV may be considered as an adjunct
Source control: 7. Urgent upper GI endoscopy if haemodynamically stable enough 8. Interventional radiology or surgery if endoscopy fails
Subsequent management: 9. Involve haematology and gastroenterology 10. Once bleeding controlled and patient stable, discuss anticoagulation strategy - may need IVC filter if anticoagulation truly contraindicated in the short term 11. Address the underlying cause of GI bleeding before restarting anticoagulation"
Viva Points
Viva Point: Opening Statement: "Deep vein thrombosis is a common condition characterised by thrombus formation in the deep venous system, most commonly of the lower limbs. It is clinically significant due to the risk of pulmonary embolism, which occurs in 50% of untreated proximal DVTs and carries significant mortality. The pathophysiology is explained by Virchow's triad of stasis, hypercoagulability, and endothelial injury."
Key Facts to Cite:
- Annual incidence: 1-2 per 1,000
- PE risk from untreated proximal DVT: 50%
- Wells score ≥2 = DVT Likely → ultrasound
- Wells score less than 2 = DVT Unlikely → D-dimer first
- DOACs are first-line (AMPLIFY, EINSTEIN trials)
- Provoked DVT: 3 months treatment
- Unprovoked DVT: Minimum 3 months, consider extended
- PTS occurs in 20-50% (SOX trial: stockings don't prevent)
Common Mistakes That Fail Candidates
- Using D-dimer in high-probability patients (Wells ≥2)
- Forgetting that DOACs are contraindicated in pregnancy
- Not knowing that triple-positive APS requires warfarin, not DOACs
- Recommending compression stockings to prevent PTS (SOX trial disproved this)
- Not recognising phlegmasia as a surgical emergency
- Forgetting to assess bleeding risk before anticoagulation
- Not knowing DOAC reversal agents
- Confusing provoked vs unprovoked DVT for treatment duration
- Performing Homan's sign (unreliable and potentially dangerous)
- Missing concurrent PE symptoms in a DVT patient
13. Patient Information
Take-Home Messages
For Patients:
- Do not stop your medication without medical advice - the clot is still dissolving; the medication prevents it growing
- Stay active - walking is encouraged; bed rest makes things worse
- Know the warning signs - sudden breathlessness, chest pain, or coughing blood = call 999/go to A&E immediately
- Bleeding precautions - you will bruise more easily; avoid contact sports; seek help for nosebleeds lasting > 10 minutes or black stools
Frequently Asked Questions
"Will the clot go away?" "Yes, your body has a natural 'clean-up crew' (called plasmin) that will gradually dissolve the clot over several weeks to months. The anticoagulant medication doesn't dissolve the clot directly - it stops it from growing while your body breaks it down naturally."
"Can I fly?" "We recommend avoiding air travel for the first 2-4 weeks after diagnosis. After that, flying is generally safe if you continue your anticoagulation. Stay hydrated, move your legs regularly, and consider an aisle seat for easier movement on long flights."
"Do I need compression stockings?" "Compression stockings don't prevent the long-term complications we once thought they did (based on recent research). However, if your leg is swollen and stockings make it feel more comfortable, they may provide symptom relief. They are not mandatory."
"How long will I need blood thinners?" "This depends on what caused your clot. If it was caused by something temporary like surgery or a long flight, usually 3 months is sufficient. If there was no clear cause, we may recommend longer treatment - this is something we'll discuss based on your individual circumstances."
Support Resources
- Thrombosis UK: thrombosisuk.org - Patient information and support
- Anticoagulation Europe: anticoagulationeurope.org - Living with anticoagulation
- National Blood Clot Alliance (US): stoptheclot.org - Education and advocacy
14. References
-
Goldhaber SZ, Bounameaux H. Pulmonary embolism and deep vein thrombosis. Lancet. 2012;379(9828):1835-1846. doi:10.1016/S0140-6736(11)61904-1
-
Heit JA. Epidemiology of venous thromboembolism. Nat Rev Cardiol. 2015;12(8):464-474. doi:10.1038/nrcardio.2015.83
-
Naess IA, Christiansen SC, Romundstad P, et al. Incidence and mortality of venous thrombosis: a population-based study. J Thromb Haemost. 2007;5(4):692-699. doi:10.1111/j.1538-7836.2007.02450.x
-
Grosse SD, Nelson RE, Nyarko KA, et al. The economic burden of incident venous thromboembolism in the United States: A review of estimated attributable healthcare costs. Thromb Res. 2016;137:3-10. doi:10.1016/j.thromres.2015.11.033
-
Heit JA, Spencer FA, White RH. The epidemiology of venous thromboembolism. J Thromb Thrombolysis. 2016;41(1):3-14. doi:10.1007/s11239-015-1311-6
-
Prandoni P, Noventa F, Ghirarduzzi A, et al. The risk of recurrent venous thromboembolism after discontinuing anticoagulation in patients with acute proximal deep vein thrombosis or pulmonary embolism. Haematologica. 2007;92(2):199-205. doi:10.3324/haematol.10516
-
Silverstein MD, Heit JA, Mohr DN, et al. Trends in the incidence of deep vein thrombosis and pulmonary embolism: a 25-year population-based study. Arch Intern Med. 1998;158(6):585-593. doi:10.1001/archinte.158.6.585
-
White RH, Keenan CR. Effects of race and ethnicity on the incidence of venous thromboembolism. Thromb Res. 2009;123(Suppl 4):S11-S17. doi:10.1016/S0049-3848(09)70136-7
-
Wolberg AS, Aleman MM, Leiderman K, Machlus KR. Procoagulant activity in hemostasis and thrombosis: Virchow's triad revisited. Anesth Analg. 2012;114(2):275-285. doi:10.1213/ANE.0b013e31823a088c
-
Bovill EG, van der Vliet A. Venous valvular stasis-associated hypoxia and thrombosis: what is the link? Annu Rev Physiol. 2011;73:527-545. doi:10.1146/annurev-physiol-012110-142305
-
Rosendaal FR, Koster T, Vandenbroucke JP, Reitsma PH. High risk of thrombosis in patients homozygous for factor V Leiden (activated protein C resistance). Blood. 1995;85(6):1504-1508. doi:10.1182/blood.V85.6.1504.bloodjournal8561504
-
Pengo V, Denas G, Zoppellaro G, et al. Rivaroxaban vs warfarin in high-risk patients with antiphospholipid syndrome. Blood. 2018;132(13):1365-1371. doi:10.1182/blood-2018-04-848333
-
Kahn SR, Shapiro S, Wells PS, et al. Compression stockings to prevent post-thrombotic syndrome: a randomised placebo-controlled trial. Lancet. 2014;383(9920):880-888. doi:10.1016/S0140-6736(13)61902-9
-
Wells PS, Anderson DR, Bormanis J, et al. Value of assessment of pretest probability of deep-vein thrombosis in clinical management. Lancet. 1997;350(9094):1795-1798. doi:10.1016/S0140-6736(97)08140-3
-
Decousus H, Prandoni P, Mismetti P, et al. Fondaparinux for the treatment of superficial-vein thrombosis in the legs. N Engl J Med. 2010;363(13):1222-1232. doi:10.1056/NEJMoa0912072
-
Wells PS, Anderson DR, Rodger M, et al. Evaluation of D-dimer in the diagnosis of suspected deep-vein thrombosis. N Engl J Med. 2003;349(13):1227-1235. doi:10.1056/NEJMoa023153
-
Righini M, Van Es J, Den Exter PL, et al. Age-adjusted D-dimer cutoff levels to rule out pulmonary embolism: the ADJUST-PE study. JAMA. 2014;311(11):1117-1124. doi:10.1001/jama.2014.2135
-
Agnelli G, Buller HR, Cohen A, et al. Oral apixaban for the treatment of acute venous thromboembolism. N Engl J Med. 2013;369(9):799-808. doi:10.1056/NEJMoa1302507
-
Bauersachs R, Berkowitz SD, Brenner B, et al. Oral rivaroxaban for symptomatic venous thromboembolism. N Engl J Med. 2010;363(26):2499-2510. doi:10.1056/NEJMoa1007903
-
Raskob GE, van Es N, Verhamme P, et al. Edoxaban for the treatment of cancer-associated venous thromboembolism. N Engl J Med. 2018;378(7):615-624. doi:10.1056/NEJMoa1711948
-
Weitz JI, Lensing AWA, Prins MH, et al. Rivaroxaban or aspirin for extended treatment of venous thromboembolism. N Engl J Med. 2017;376(13):1211-1222. doi:10.1056/NEJMoa1700518
-
Vedantham S, Goldhaber SZ, Julian JA, et al. Pharmacomechanical catheter-directed thrombolysis for deep-vein thrombosis. N Engl J Med. 2017;377(23):2240-2252. doi:10.1056/NEJMoa1615066
-
Mismetti P, Laporte S, Pellerin O, et al. Effect of a retrievable inferior vena cava filter plus anticoagulation vs anticoagulation alone on risk of recurrent pulmonary embolism: a randomized clinical trial. JAMA. 2015;313(16):1627-1635. doi:10.1001/jama.2015.3780
-
Spyropoulos AC, Ageno W, Albers GW, et al. Rivaroxaban for thromboprophylaxis after hospitalization for medical illness. N Engl J Med. 2018;379(12):1118-1127. doi:10.1056/NEJMoa1805090
-
Kearon C, Akl EA, Ornelas J, et al. Antithrombotic therapy for VTE disease: CHEST guideline and expert panel report. Chest. 2016;149(2):315-352. doi:10.1016/j.chest.2015.11.026
15. Guidelines Summary
| Guideline | Year | Key Recommendations |
|---|---|---|
| NICE NG158 | 2018 (updated 2020) | VTE prophylaxis for hospitalised patients; risk assessment mandatory |
| NICE NG158 | 2020 | DOACs first-line for DVT/PE treatment in most patients |
| ASH Guidelines | 2020 | Optimal duration of anticoagulation; extended low-dose rivaroxaban/apixaban for secondary prevention |
| ACCP (CHEST) Guidelines | 2016 | Comprehensive VTE management recommendations |
| ESC Guidelines | 2019 | PE-focused but covers VTE spectrum |
| ISTH Guidelines | 2020 | Cancer-associated thrombosis management |
Last Updated: January 2025 Evidence Review Date: December 2024 Next Review Due: January 2026
This content is for educational purposes and should not replace clinical judgement. Always refer to local guidelines and consult specialists for complex cases.
Copyright 2025 MedVellum. All rights reserved.
Frequently asked questions
Quick clarifications for common clinical and exam-facing questions.
When should I seek emergency care for deep vein thrombosis (dvt) - adult?
Seek immediate emergency care if you experience any of the following warning signs: Phlegmasia Cerulea Dolens (Limb-Threatening Emergency), Phlegmasia Alba Dolens, Concurrent Pulmonary Embolism Symptoms, Massive Ilio-femoral DVT (High Embolic Risk), Hypotension or Haemodynamic Instability, Bilateral DVT (Consider IVC Thrombosis).
Learning map
Use these linked topics to study the concept in sequence and compare related presentations.
Prerequisites
Start here if you need the foundation before this topic.
- Coagulation Cascade
- Venous Anatomy Lower Limb
Differentials
Competing diagnoses and look-alikes to compare.
- Cellulitis
- Baker's Cyst Rupture
Consequences
Complications and downstream problems to keep in mind.
- Pulmonary Embolism
- Post-Thrombotic Syndrome