Dementia
Diagnosis requires comprehensive assessment including cognitive testing (MMSE, MoCA, ACE-III), exclusion of reversible causes (B12 deficiency, hypothyroidism, normal pressure hydrocephalus, depression), and...
What matters first
Diagnosis requires comprehensive assessment including cognitive testing (MMSE, MoCA, ACE-III), exclusion of reversible causes (B12 deficiency, hypothyroidism, normal pressure hydrocephalus, depression), and...
Rapid decline (less than 6 months)
9 Jan 2026
Generated educational material; verify before clinical use.
Visible references section
See the concept before reading it
Study the key anatomy, imaging, and decision pathways as full teaching plates.
Clinical board
A visual summary of the highest-yield teaching signals on this page.
Urgent signals
Safety-critical features pulled from the topic metadata.
- Rapid decline (less than 6 months)
- Focal neurological signs
- Young onset (less than 65 years)
- Gait disturbance and urinary incontinence (NPH)
Linked comparisons
Differentials and adjacent topics worth opening next.
- Delirium
- Depression (Pseudodementia)
Content status and exam context
This page is AI-generated educational content. It may contain errors or omissions and is not a substitute for current guidelines, local protocols, senior clinical judgement, or professional medical advice.
MedVellum does not claim an individual clinician reviewer, board certification, or professional credential for this page unless a future version names a real, verifiable reviewer.
Clinical explanation and evidence
Dementia
1. Topic Overview
Summary
Dementia is a clinical syndrome characterised by progressive decline in cognitive function severe enough to interfere with daily activities and independent living. It is not a single disease but a syndrome resulting from various underlying pathologies. Alzheimer's disease accounts for 60-70% of cases, followed by vascular dementia (20%), dementia with Lewy bodies (10-15%), and frontotemporal dementia (2-5%). [1,2] The global prevalence is estimated at 5-7% in those aged ≥60 years, with numbers projected to reach 115 million worldwide by 2050. [3]
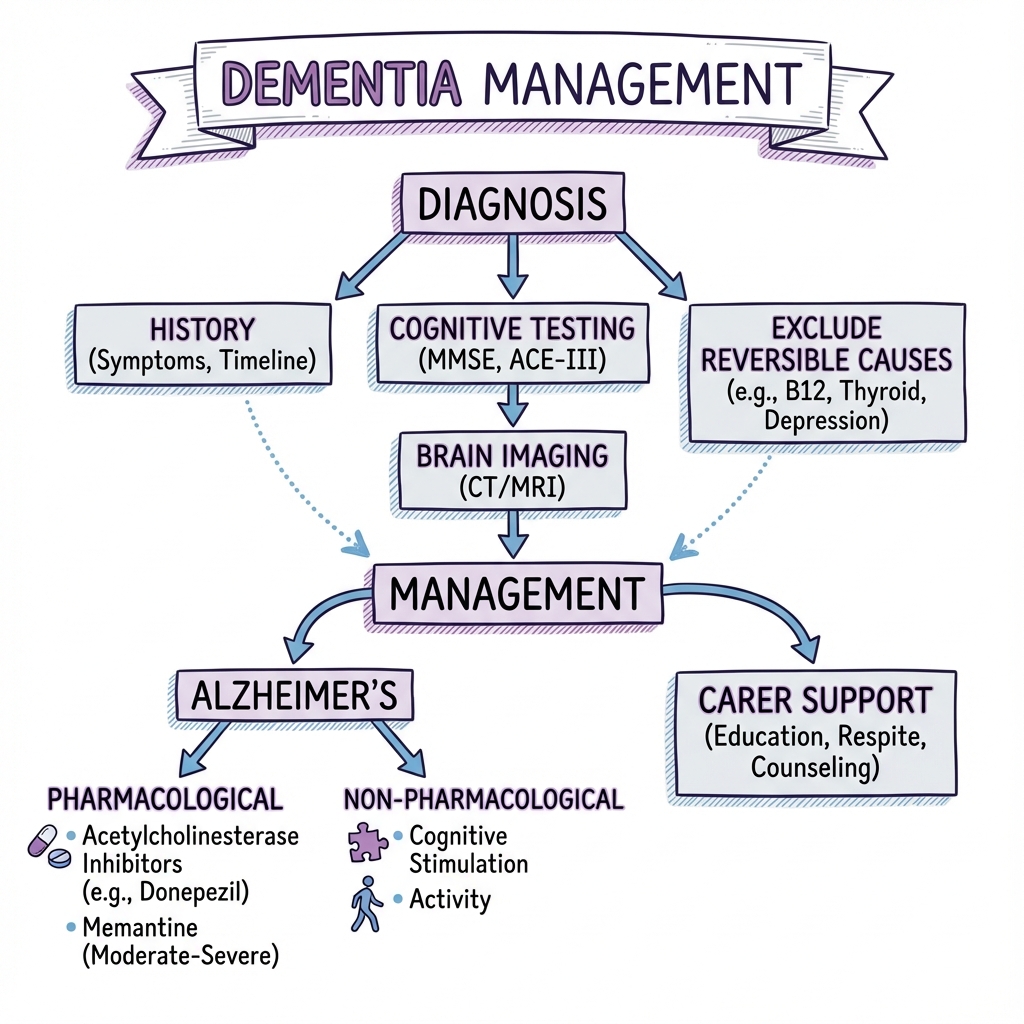
Diagnosis requires comprehensive assessment including cognitive testing (MMSE, MoCA, ACE-III), exclusion of reversible causes (B12 deficiency, hypothyroidism, normal pressure hydrocephalus, depression), and neuroimaging to exclude structural lesions and identify disease-specific patterns. [4] Management focuses on optimising function and quality of life through pharmacological interventions (acetylcholinesterase inhibitors for mild-moderate Alzheimer's, memantine for moderate-severe disease), non-pharmacological therapies (cognitive stimulation, reminiscence therapy), cardiovascular risk factor modification, and comprehensive support for patients and carers. [5]
Early diagnosis enables timely intervention, advance care planning while the patient retains capacity, and access to support services. The Lancet Commission identified 12 modifiable risk factors accounting for approximately 40% of dementia cases, emphasising the critical importance of prevention strategies across the lifespan. [6]
Key Facts
- Definition: Acquired progressive impairment in ≥2 cognitive domains (memory, executive function, language, visuospatial ability, praxis) severe enough to impair daily function
- Global Prevalence: 6.97% in adults ≥60 years; prevalence doubles every 5 years after age 65 [1]
- Alzheimer's Disease: 60-70% of cases; characterised by amyloid-beta plaques and neurofibrillary tau tangles [2]
- Vascular Dementia: 20% of cases; second most common type; stepwise decline associated with cerebrovascular disease [3]
- Dementia with Lewy Bodies: 10-15%; fluctuating cognition, visual hallucinations, parkinsonism, REM sleep behaviour disorder [7]
- Frontotemporal Dementia: 2-5%; younger onset (less than 65 years); behavioural variant or language variants [8]
- Reversible Causes: 5-10% have potentially reversible contributors (B12 deficiency, hypothyroidism, normal pressure hydrocephalus, depression) [9]
- Young-Onset Dementia: Prevalence 119 per 100,000 in ages 30-64 years; accounts for 3.9 million cases globally [10]
- Pharmacotherapy: AChEIs provide modest cognitive benefit (1.5-2 point MMSE improvement); memantine for moderate-severe disease [11,12]
- Prevention: 40% of dementia potentially preventable by addressing 12 modifiable risk factors [6]
Clinical Pearls
"Dementia is a Clinical Diagnosis": No single test diagnoses dementia. Diagnosis requires demonstrable decline from previous level of functioning in ≥2 cognitive domains, corroborated by informant history, affecting daily activities.
"Rule Out Reversible Causes First": Always exclude B12 deficiency, hypothyroidism, normal pressure hydrocephalus, and depression ("pseudodementia") before attributing symptoms to neurodegenerative disease. Structural imaging is mandatory to exclude subdural haematoma, tumours, and hydrocephalus.
"Dementia Subtype Determines Management": Lewy Body dementia has severe neuroleptic sensitivity (can cause fatal neuroleptic malignant syndrome). Frontotemporal dementia does not respond to acetylcholinesterase inhibitors. Vascular dementia requires aggressive cardiovascular risk factor control.
"Capacity Assessment is Time and Decision-Specific": A patient may have capacity for some decisions but not others. Assess capacity for each specific decision at the time it needs to be made. Dementia does not automatically mean loss of all capacity.
"Early Advance Care Planning is Essential": Discuss values, preferences, Lasting Power of Attorney (health and welfare), and Advance Decisions to Refuse Treatment while the patient can meaningfully participate. This is a clinical and ethical priority.
"Support the Carer": Carers experience high rates of depression (40%), anxiety, and burnout. Carer wellbeing directly impacts patient outcomes. Signpost to carer support services, respite care, and Admiral Nurses.
Why This Matters Clinically
Dementia is one of the leading causes of disability and dependency in older adults worldwide, with profound impacts on patients, families, and health systems. [3] Currently affecting over 55 million people globally, this number is projected to reach 152 million by 2050, driven by population ageing and increased life expectancy. [3] The condition carries enormous economic burden (estimated US$1.3 trillion in 2019) and is the seventh leading cause of death globally.
Early recognition enables:
- Access to evidence-based interventions that can improve quality of life and slow functional decline
- Identification and treatment of reversible causes (5-10% have contributory reversible factors) [9]
- Advance care planning while patients retain decision-making capacity
- Implementation of safety measures (driving assessment, safeguarding, financial protection)
- Carer education and support to reduce burnout and improve care quality
- Optimisation of comorbidity management (dementia complicates management of other conditions)
The Lancet Commission's identification of 12 modifiable risk factors accounting for 40% of dementia highlights the crucial importance of prevention across the life course. [6] Clinicians have opportunities at every stage—from managing midlife hypertension to promoting social engagement in older adults—to reduce dementia risk.
2. Epidemiology
Global Prevalence and Incidence
The global prevalence of dementia in adults aged ≥60 years is 6.97% (95% CI: 5.46-8.64 per 100 persons), translating to 55.2 million people living with dementia in 2020. [1,3] This represents a dramatic increase from 35.6 million in 2010, with projections suggesting 78 million by 2030 and 152 million by 2050. [3] The age-specific prevalence increases exponentially with advancing age, approximately doubling every five years after age 65. [1]
Age-Standardised Prevalence by Age Group: [1]
| Age Group | Prevalence per 100 persons | Population Impact |
|---|---|---|
| 50-59 | 0.27 | Rare |
| 60-69 | 2.0 | 1 in 50 |
| 70-79 | 6-8 | 1 in 15 |
| 80-84 | 15 | 1 in 7 |
| 85-89 | 25-30 | 1 in 4 |
| 90-94 | 40-45 | 1 in 2 |
| ≥95 | 50-60 | > 1 in 2 |
| ≥100 | 65.92 | 2 in 3 |
Global Distribution:
- Europe and North America: Higher age-standardised prevalence (7-8%) [3]
- Latin America: Highest regional prevalence (8.5%) [3]
- Sub-Saharan Africa: Lower reported prevalence (2-4%), likely reflecting under-diagnosis and lower life expectancy [3]
- Asia: 5-7%, with rapid increases projected due to demographic transition [3]
- Low- and middle-income countries: 58% of global dementia burden in 2020, projected to rise to 71% by 2050 [3]
Dementia Subtype Distribution: [2]
| Subtype | Proportion | Key Features |
|---|---|---|
| Alzheimer's Disease | 60-70% | Most common; gradual onset, prominent memory loss |
| Vascular Dementia | 15-20% | Second most common; stepwise decline, vascular risk factors |
| Mixed Dementia (AD + VaD) | 10-15% | Overlapping pathologies |
| Dementia with Lewy Bodies | 5-15% | Fluctuation, hallucinations, parkinsonism |
| Frontotemporal Dementia | 2-5% | Younger onset; behavioural or language predominant |
| Other (Parkinson's, prion, etc.) | 5-10% | Varied presentations |
Young-Onset Dementia (Age less than 65 Years)
Young-onset dementia (YOD) affects an estimated 3.9 million people aged 30-64 years globally, with age-standardised prevalence of 119 per 100,000 population in this age range. [10] Prevalence increases from 1.1 per 100,000 in ages 30-34 to 77.4 per 100,000 in ages 60-64. [10] Frontotemporal dementia and early-onset Alzheimer's (often with genetic contributions) are proportionally more common in YOD compared to late-onset dementia.
Demographics and Risk Factors
Sex Differences:
- Overall prevalence slightly higher in women (7.88% vs 5.61% in men) [1]
- Alzheimer's disease: Female preponderance (1.5-2:1 ratio), partly explained by longer life expectancy and possibly hormonal factors
- Vascular dementia: Male preponderance (1.8:1 in ages 60-69), reflecting higher cardiovascular disease burden [1]
- Dementia with Lewy bodies: Male preponderance (2:1)
Genetics:
- APOE ε4 allele: Strongest genetic risk factor for late-onset Alzheimer's disease; one copy increases risk 3-fold, two copies 8-12-fold [13]
- Autosomal dominant forms (rare, less than 1%): APP, PSEN1, PSEN2 mutations cause early-onset familial Alzheimer's
- Other genetic risks: TREM2 (innate immunity), CLU (apolipoprotein J), SORL1 variants
Modifiable Risk Factors (Lancet Commission 2020)
The Lancet Commission on Dementia Prevention, Intervention, and Care identified 12 modifiable risk factors across the life course accounting for approximately 40% of dementia worldwide: [6]
Early Life:
- Less education (7% population attributable fraction): Lower cognitive reserve
Midlife (45-65 years):
- Hearing loss (8%): Largest single modifiable factor; cochlear damage or social isolation
- Traumatic brain injury (3%): Repeated head trauma; seen in contact sports, falls
- Hypertension (2%): Systolic BP ≥130 mmHg; vascular damage and Aβ accumulation
- Excessive alcohol (1%): > 21 units/week increases risk
- Obesity (1%): BMI ≥30 kg/m²; midlife obesity more relevant than late-life
Later Life (≥65 years):
- Smoking (5%): Dose-dependent; cessation reduces risk
- Depression (4%): Bidirectional relationship; may be prodrome or risk factor
- Social isolation (4%): Lack of social contact; distinct from loneliness
- Physical inactivity (2%): Sedentary lifestyle
- Diabetes (1%): HbA1c > 7%; microvascular and macrovascular damage
- Air pollution (2%): PM2.5 and nitrogen dioxide exposure
3. Aetiology and Pathophysiology
Alzheimer's Disease
Alzheimer's disease is characterised by two hallmark neuropathological features: extracellular amyloid-beta (Aβ) plaques and intracellular neurofibrillary tangles composed of hyperphosphorylated tau protein. [2]
Amyloid Cascade Hypothesis:
- APP Processing: Amyloid precursor protein (APP) is abnormally cleaved by β-secretase and γ-secretase (instead of α-secretase)
- Aβ Production: Generates amyloid-beta peptides (Aβ40, Aβ42); Aβ42 is more prone to aggregation
- Plaque Formation: Aβ oligomers and fibrils accumulate extracellularly, forming amyloid plaques
- Neurotoxicity: Aβ oligomers impair synaptic function, trigger inflammatory responses, and induce neuronal stress
- Tau Pathology: Aβ accumulation promotes hyperphosphorylation of tau protein
- Neurofibrillary Tangles: Hyperphosphorylated tau aggregates intracellularly, disrupting microtubule stability and axonal transport
- Neuronal Death: Progressive synaptic loss, neuronal death, and brain atrophy
- Cholinergic Deficit: Degeneration of cholinergic neurons in nucleus basalis of Meynert, leading to acetylcholine deficiency (therapeutic target for AChEIs)
Anatomical Progression (Braak Staging):
- Stages I-II: Transentorhinal region (preclinical)
- Stages III-IV: Limbic system, hippocampus (mild-moderate dementia)
- Stages V-VI: Widespread neocortical involvement (severe dementia)
Neurochemistry:
- Cholinergic deficit: Reduced acetylcholine synthesis and neurotransmission
- Glutamatergic excitotoxicity: NMDA receptor overactivation (target for memantine)
- Neuroinflammation: Activated microglia and astrocytes; cytokine release
- Oxidative stress: Mitochondrial dysfunction, reactive oxygen species
Vascular Dementia
Vascular dementia results from chronic brain ischaemia due to cerebrovascular disease, leading to neuronal loss and white matter damage. [3] Multiple mechanisms contribute:
Pathological Subtypes:
- Multi-infarct dementia: Multiple large vessel territorial infarcts
- Strategic infarct dementia: Single infarct in critical location (thalamus, angular gyrus, hippocampus, caudate)
- Small vessel disease (most common):
- Lacunar infarcts (small deep infarcts)
- White matter hyperintensities (leukoaraiosis)
- Microbleeds and microinfarcts
- Blood-brain barrier dysfunction and chronic hypoperfusion
Risk Factors:
- Hypertension (most important modifiable factor)
- Diabetes mellitus
- Hyperlipidaemia
- Atrial fibrillation and cardiac embolic sources
- Smoking
- Previous stroke or TIA
Cognitive Profile:
- Executive dysfunction (planning, problem-solving) often more prominent than memory
- Processing speed reduced
- Stepwise decline pattern (but can be gradual with small vessel disease)
Dementia with Lewy Bodies (DLB)
DLB is characterised by abnormal intracellular aggregation of alpha-synuclein protein forming Lewy bodies in cortical and subcortical neurons. [7] Overlaps pathologically with Parkinson's disease dementia (PDD); distinction based on timing of motor vs cognitive symptoms.
Neuropathology:
- Cortical Lewy bodies (alpha-synuclein aggregates) in cingulate cortex, neocortex, amygdala, brainstem
- Cholinergic deficit (more severe than AD; explains better response to AChEIs)
- Dopaminergic neuron loss (substantia nigra) → parkinsonism
- Often coexistent AD pathology (mixed pathology common)
Core Clinical Features:
- Fluctuating cognition with variations in attention and alertness
- Recurrent visual hallucinations (well-formed, detailed)
- REM sleep behaviour disorder (acting out dreams; may precede dementia by years)
- Spontaneous parkinsonism (rigidity, bradykinesia; tremor less common than PD)
Severe Neuroleptic Sensitivity: DLB patients have heightened sensitivity to antipsychotic medications, which can precipitate severe parkinsonism, reduced consciousness, and neuroleptic malignant syndrome. Antipsychotics should be avoided or used with extreme caution.
Frontotemporal Dementia (FTD)
FTD encompasses a group of disorders characterised by selective degeneration of frontal and/or temporal lobes, typically presenting before age 65. [8]
Molecular Pathology:
- Tau proteinopathies (~40%): Mutations in MAPT gene
- TDP-43 proteinopathies (~50%): Mutations in GRN, C9orf72
- FUS proteinopathies (~5-10%)
Clinical Variants:
-
Behavioural variant FTD (bvFTD): Most common (~60%)
- Disinhibition, apathy, loss of empathy
- Compulsive behaviours, dietary changes (sweet preference)
- Executive dysfunction
- Memory relatively preserved early
- Frontal lobe atrophy
-
Semantic variant primary progressive aphasia (svPPA):
- Loss of word meaning and object knowledge
- Fluent but empty speech
- Surface dyslexia
- Anterior temporal lobe atrophy (left > right)
-
Non-fluent variant primary progressive aphasia (nfvPPA):
- Effortful, halting speech
- Agrammatism, phonological errors
- Comprehension relatively preserved
- Left posterior frontoinsular atrophy
Reversible and Treatable Causes
Approximately 5-10% of patients presenting with cognitive impairment have potentially reversible or contributory causes: [9]
| Cause | Mechanism | Diagnostic Test | Treatment |
|---|---|---|---|
| B12 Deficiency | Demyelination, homocysteine accumulation | Serum B12 (less than 200 pg/mL); MMA, homocysteine | B12 supplementation (IM or high-dose oral) |
| Hypothyroidism | Metabolic encephalopathy | TSH, free T4 | Levothyroxine replacement |
| Normal Pressure Hydrocephalus | Enlarged ventricles, impaired CSF circulation | MRI: ventriculomegaly disproportionate to atrophy; gait assessment | VP shunt (gait most responsive) |
| Depression (Pseudodementia) | Poor effort, psychomotor retardation | Clinical assessment; improves with treatment | Antidepressants, psychotherapy |
| Chronic Subdural Haematoma | Mass effect, ischaemia | CT/MRI brain | Surgical drainage if symptomatic |
| Brain Tumour | Mass effect, focal damage | MRI brain with contrast | Depends on tumour type |
| Neurosyphilis | Chronic meningovascular inflammation | Syphilis serology, CSF VDRL | Penicillin |
| HIV Dementia | Direct viral CNS infection | HIV serology, CD4 count | Antiretroviral therapy |
| Wernicke-Korsakoff | Thiamine deficiency | Clinical diagnosis; thiamine level | Parenteral thiamine |
| Medication Effects | Anticholinergic, sedative effects | Medication review | Deprescribing |
Triad of Normal Pressure Hydrocephalus (NPH):
- Gait disturbance (magnetic gait, wide-based, shuffling) — appears first
- Cognitive impairment (subcortical pattern: psychomotor slowing, executive dysfunction)
- Urinary incontinence (urgency, frequency) — appears late
NPH is potentially reversible with ventriculoperitoneal shunt, though response is variable and best for gait symptoms. [9]
4. Clinical Presentation
Diagnostic Criteria for Dementia
Dementia is a clinical diagnosis requiring: [4]
-
Cognitive decline from previous level of functioning in ≥2 cognitive domains:
- Memory (learning and recall)
- Executive function (planning, decision-making, working memory)
- Language (naming, word-finding, fluency)
- Visuospatial ability (orientation, spatial navigation)
- Praxis (learned motor skills)
- Social cognition (behavioural regulation, theory of mind)
-
Functional impairment: Cognitive deficits interfere with independence in everyday activities (IADLs initially, then ADLs)
-
Not better explained by delirium or other psychiatric disorder
-
Corroborated by informant history and objective cognitive assessment
Core Symptoms by Cognitive Domain
Memory Impairment:
- Early Alzheimer's: Episodic memory loss (recent events); difficulty forming new memories
- Repetitive questioning ("Have I told you this before?")
- Misplacing items in unusual locations (keys in freezer)
- Forgetting appointments, conversations
- Getting lost in familiar places
- Temporal gradient: Remote memories preserved longer than recent
Executive Dysfunction:
- Difficulty with complex tasks requiring planning and sequencing (cooking multi-step meals, managing finances)
- Poor judgment and decision-making
- Reduced mental flexibility
- Working memory deficits
- More prominent in vascular dementia and FTD
Language Impairment:
- Word-finding difficulty (anomia): "You know, the thing you use to..."
- Reduced verbal fluency
- Circumlocution (talking around missing words)
- Comprehension difficulties (in primary progressive aphasia)
- Difficulty following complex conversations
Visuospatial Impairment:
- Difficulty judging distances (parking, pouring liquids)
- Getting lost, even in familiar environments
- Difficulty reading (posterior cortical atrophy variant of AD)
- Constructional apraxia (difficulty copying shapes, clock drawing)
- Visual hallucinations (especially in DLB)
Behavioural and Psychological Symptoms of Dementia (BPSD):
- Apathy: Loss of interest, initiative, motivation (most common BPSD)
- Agitation and aggression: Verbal or physical; often response to unmet needs or environmental stressors
- Depression and anxiety: Present in 40-50% of dementia patients
- Psychosis: Delusions (theft, infidelity) and hallucinations
- Disinhibition: Socially inappropriate behaviour, impulsivity
- Sleep disturbance: Fragmented sleep, day-night reversal, REM sleep behaviour disorder (DLB)
- Wandering: Aimless or purposeful walking; risk of getting lost
Functional Decline
Instrumental Activities of Daily Living (IADLs) — affected early:
- Managing finances, paying bills
- Medication management
- Using telephone, technology
- Shopping, meal preparation
- Housework, home maintenance
- Transportation, driving
Activities of Daily Living (ADLs) — affected in moderate-severe stages:
- Bathing, showering
- Dressing, grooming
- Toileting, continence
- Transferring (bed to chair)
- Feeding
Clinical Features by Dementia Subtype
| Feature | Alzheimer's | Vascular | Lewy Body | Frontotemporal |
|---|---|---|---|---|
| Onset | Insidious, gradual | Acute/stepwise or gradual | Insidious | Insidious, often less than 65 years |
| Course | Progressive decline | Stepwise ± plateaus | Fluctuating | Progressive |
| Early feature | Episodic memory loss | Executive dysfunction | Fluctuation, hallucinations | Behaviour/language |
| Memory | Prominent early impairment | Variable; strategic infarcts | Relatively preserved early | Preserved until late |
| Executive function | Impaired moderate-late | Prominent early | Impaired | Severely impaired early |
| Hallucinations | Late, if at all | Uncommon | Visual, detailed, recurrent (early) | Rare |
| Parkinsonism | Late, if at all | Vascular parkinsonism | Early, spontaneous | Late, if at all |
| Gait | Normal until late | Early abnormality, falls | Parkinson-like | Normal early |
| MRI findings | Hippocampal atrophy, medial temporal lobe | Infarcts, WMH, lacunes | Relative preservation | Frontal/temporal atrophy |
| Treatment response | AChEIs, memantine | Vascular risk factors | AChEIs (good); avoid antipsychotics | SSRIs for behaviour; no AChEI benefit |
Red Flags Requiring Urgent Investigation
[!CAUTION] Red Flags — Features Suggesting Atypical or Rapidly Progressive Dementia:
- Rapid decline (less than 6 months): Consider Creutzfeldt-Jakob disease (CJD), autoimmune/paraneoplastic encephalitis, CNS vasculitis, malignancy
- Age less than 55 years: Genetic forms, FTD, inflammatory/infectious causes, metabolic disorders
- Focal neurological signs: Stroke, tumour, multiple sclerosis, CNS vasculitis
- Headache, fever, meningism: Encephalitis, meningitis, CNS vasculitis
- Seizures: Alzheimer's (uncommon but recognised), autoimmune encephalitis, structural lesions
- Myoclonus: CJD (jerky, startle-induced)
- Altered consciousness, delirium: Superimposed acute illness, medication toxicity, metabolic derangement
- Triad of gait + cognitive decline + incontinence: Normal pressure hydrocephalus (potentially reversible)
- Early prominent visual hallucinations: Dementia with Lewy bodies (neuroleptic sensitivity)
- Early severe behavioural change, disinhibition: Frontotemporal dementia
- Rapidly progressive ataxia + cognitive decline: CJD, paraneoplastic cerebellar degeneration
5. Clinical Examination
Structured Approach to Cognitive Assessment
General Observation:
- Appearance, hygiene, self-care
- Appropriateness of clothing for weather/occasion
- Level of engagement, eye contact
- Evidence of neglect or malnutrition
Screening Cognitive Assessment Tools:
1. Mini-Mental State Examination (MMSE) — 30 points
- Widely used, quick (~10 minutes)
- Domains: Orientation (10), registration (3), attention/calculation (5), recall (3), language (8), visuospatial (1)
- Interpretation:
- 24-30: Normal (but ceiling effect in educated individuals)
- 18-23: Mild dementia
- 10-17: Moderate dementia
- less than 10: Severe dementia
- Limitations: Insensitive to mild impairment, executive dysfunction, and FTD; heavily influenced by education and language
2. Montreal Cognitive Assessment (MoCA) — 30 points
- More sensitive for mild cognitive impairment and executive dysfunction [14]
- Domains: Visuospatial/executive (5), naming (3), attention (6), language (3), abstraction (2), delayed recall (5), orientation (6)
- Interpretation:
- ≥26: Normal (add 1 point if education ≤12 years)
- 18-25: Mild cognitive impairment or mild dementia
- less than 18: Dementia
- Advantages: Better detection of MCI, vascular dementia, FTD, DLB
3. Addenbrooke's Cognitive Examination-III (ACE-III) — 100 points
- Comprehensive (~20 minutes)
- Domains: Attention/orientation (18), memory (26), fluency (14), language (26), visuospatial (16)
- Interpretation:
- ≥88: Normal
- less than 88: Cognitive impairment; less than 82 sensitivity for dementia
- Subtype differentiation: VLOM ratio (Verbal fluency + Language / Orientation + Memory) helps distinguish FTD from AD
4. Six-Item Cognitive Impairment Test (6-CIT)
- Very brief (3-5 minutes); useful in primary care
- Orientation, memory, attention
- Score ≥8 suggests cognitive impairment
Clock Drawing Test:
- Draw clock face, place numbers, set time to "10 past 11"
- Assesses visuospatial ability, executive function, comprehension
- Sensitive for parietal lobe dysfunction, constructional apraxia
Verbal Fluency:
- Phonemic fluency: Name as many words beginning with 'F' in 60 seconds (normal > 15)
- Semantic fluency: Name as many animals in 60 seconds (normal > 20)
- Impaired in FTD, executive dysfunction
Neurological Examination
Cranial Nerves:
- Visual fields (posterior cortical atrophy)
- Eye movements (progressive supranuclear palsy: vertical gaze palsy)
- Primitive reflexes: Glabellar tap (non-habituating in parkinsonism), grasp reflex (frontal lobe disease), palmomental reflex
Motor Examination:
- Parkinsonism (DLB, Parkinson's disease dementia, vascular): Bradykinesia, rigidity, reduced arm swing
- Myoclonus (CJD): Jerky, startle-induced movements
- Focal weakness (vascular dementia, tumour): Upper motor neuron signs
Gait Assessment:
- Normal gait until late (AD)
- Apraxic gait (small steps, shuffling, difficulty initiating): Vascular dementia, NPH
- Magnetic gait (feet appear stuck to floor): NPH
- Parkinsonian gait (shuffling, reduced arm swing, stooped): DLB, Parkinson's disease dementia
- Ataxic gait (wide-based, unsteady): Alcohol-related dementia, cerebellar disease, CJD
- High-steppage gait (B12 deficiency with peripheral neuropathy)
Coordination and Sensation:
- Usually normal in early dementia
- Peripheral neuropathy: B12 deficiency, alcohol-related
- Posterior column loss: B12 deficiency (subacute combined degeneration)
Mood and Behavioural Assessment
Depression Screening:
- Geriatric Depression Scale (GDS): 15-item or 30-item version
- High prevalence of depression in dementia (40-50%)
- Depression can mimic or exacerbate cognitive impairment ("pseudodementia")
Neuropsychiatric Inventory (NPI):
- Comprehensive assessment of BPSD
- 12 domains: Delusions, hallucinations, agitation, depression, anxiety, euphoria, apathy, disinhibition, irritability, aberrant motor behaviour, sleep, appetite
Functional Assessment
Instrumental ADLs:
- Lawton IADL Scale: 8 domains (telephone, shopping, food preparation, housekeeping, laundry, transport, medications, finances)
Basic ADLs:
- Katz ADL Index or Barthel Index: Bathing, dressing, toileting, transferring, continence, feeding
Informant Questionnaires:
- Informant Questionnaire on Cognitive Decline in the Elderly (IQCODE): 16-item questionnaire assessing change over 10 years
6. Investigations
Mandatory Baseline Investigations (All Patients)
The primary goals of investigation are to: (1) exclude reversible causes, (2) identify dementia subtype, (3) exclude alternative diagnoses, and (4) assess for comorbidities.
Blood Tests: [4]
| Investigation | Purpose | Abnormalities to Detect |
|---|---|---|
| Full Blood Count (FBC) | Anaemia, infection | Macrocytic anaemia (B12, folate), leucocytosis (infection) |
| Renal Function (U&E) | Metabolic causes, medication monitoring | Uraemia, electrolyte disturbances |
| Liver Function Tests (LFTs) | Hepatic encephalopathy, alcohol | Elevated transaminases, GGT (alcohol) |
| Calcium | Hypercalcaemia (confusion) | Hypercalcaemia (primary hyperparathyroidism, malignancy) |
| Glucose or HbA1c | Diabetes, hypoglycaemia | Diabetes (risk factor), hypoglycaemia (reversible) |
| Thyroid Function (TSH, free T4) | Hypothyroidism (reversible) | Elevated TSH, low T4 |
| Vitamin B12 | B12 deficiency (reversible) | less than 200 pg/mL; if borderline, check methylmalonic acid, homocysteine |
| Folate | Folate deficiency | Low folate |
Additional Tests (If Clinically Indicated):
- Syphilis serology (VDRL, TPPA): Young onset, risk factors
- HIV serology: Risk factors, young onset
- Autoimmune screen (ANA, ANCA, anti-thyroid antibodies): Rapidly progressive, young onset, systemic features
- Paraneoplastic antibodies: Rapidly progressive, cancer history
- Copper, caeruloplasmin: Young onset (Wilson's disease)
- ESR, CRP: Inflammatory causes, vasculitis
- Urinalysis: UTI (common cause of delirium superimposed on dementia)
Neuroimaging
Structural Imaging — MANDATORY for all patients with dementia:
CT Brain (Non-Contrast):
- Indications: First-line structural imaging when MRI unavailable or contraindicated
- Findings:
- "Exclude structural causes: Subdural haematoma, tumour, hydrocephalus"
- Global atrophy (non-specific)
- "Vascular changes: Infarcts, white matter hypodensities"
- "NPH: Ventriculomegaly disproportionate to sulcal atrophy"
- Limitations: Less sensitive than MRI for early/subtle changes
MRI Brain (Preferred):
- Indications: Preferred modality for dementia assessment; higher sensitivity for pathological changes [15]
- Sequences: T1, T2, FLAIR, gradient echo (microbleeds), diffusion-weighted imaging (DWI)
- Findings by Subtype:
| Subtype | MRI Findings |
|---|---|
| Alzheimer's Disease | Medial temporal lobe atrophy (hippocampus, entorhinal cortex); posterior cingulate and precuneus atrophy; relative preservation of primary motor and sensory cortices |
| Vascular Dementia | Multiple infarcts (cortical and/or subcortical); extensive white matter hyperintensities (WMH); lacunar infarcts in basal ganglia, thalamus; microbleeds; strategic infarcts (thalamus, angular gyrus) |
| Dementia with Lewy Bodies | Relative preservation of medial temporal lobes (less atrophy than AD); posterior cortical atrophy; reduced occipital lobe metabolism |
| Frontotemporal Dementia | Frontal and/or anterior temporal lobe atrophy (often asymmetric); knife-edge atrophy; specific patterns for variants (bvFTD: frontal; svPPA: anterior temporal) |
| Normal Pressure Hydrocephalus | Ventriculomegaly (Evans index > 0.3: ventricular width/brain width); disproportionately enlarged ventricles vs sulcal atrophy; tight high convexity, dilated Sylvian fissures |
| CJD | DWI hyperintensity in cortical ribboning, basal ganglia (caudate, putamen) |
Functional and Molecular Imaging (Specialist Use):
FDG-PET (Fluorodeoxyglucose Positron Emission Tomography):
- Use: Differentiate dementia subtypes when diagnosis uncertain
- Findings:
- "AD: Hypometabolism in temporoparietal cortex, posterior cingulate (precuneus)"
- "FTD: Hypometabolism in frontal and/or anterior temporal lobes"
- "DLB: Hypometabolism in occipital cortex (cingulate island sign: preserved posterior cingulate)"
Amyloid PET Imaging:
- Tracers: Florbetapir, flutemetamol, florbetaben
- Use: Detect fibrillar amyloid-beta deposition; positive in AD, negative in FTD, DLB (may be positive if mixed pathology)
- Limitations: Positive scan does not confirm AD (amyloid can be present without dementia); expensive, limited availability
Tau PET Imaging:
- Emerging technique: Detects tau tangles; correlates better with cognitive decline than amyloid
- Research use: Not yet routine clinical practice
DaTscan (Ioflupane SPECT):
- Use: Differentiate DLB from AD and other non-Lewy body dementias [7]
- Mechanism: Assesses dopamine transporter density in striatum
- Findings:
- "DLB/Parkinson's: Reduced striatal uptake (abnormal scan)"
- "AD, FTD: Normal striatal uptake"
- Indication: Suspected DLB with uncertain diagnosis; guides management (neuroleptic sensitivity)
CSF Analysis
Indications (not routine; specialist use):
- Young-onset dementia (less than 55 years)
- Rapidly progressive dementia
- Suspicion of inflammatory, infectious, or autoimmune cause
- Atypical features
- Suspected CJD
CSF Biomarkers in Alzheimer's Disease:
- Decreased Aβ42: Reflects amyloid deposition in plaques
- Increased total tau: Neuronal damage
- Increased phosphorylated tau (p-tau): AD-specific tangle pathology
- Aβ42/Aβ40 ratio: Improved specificity
- Sensitivity/Specificity: ~85-90% for AD vs non-AD dementia [16]
CSF in Other Dementias:
- CJD: Elevated 14-3-3 protein, elevated total tau (very high), positive RT-QuIC (real-time quaking-induced conversion)
- Inflammatory/Autoimmune: Elevated protein, lymphocytic pleocytosis, oligoclonal bands, specific antibodies
Cognitive Assessment Tools (Already Covered in Examination Section)
Summary of Sensitivities for MCI/Dementia Detection: [14]
| Test | Sensitivity for Dementia | Sensitivity for MCI | Time to Administer |
|---|---|---|---|
| MMSE | 85% | 60% | 10 min |
| MoCA | 90% | 83% | 10 min |
| ACE-III | 93% | 85% | 20 min |
| 6-CIT | 80% | 65% | 3 min |
Genetic Testing
Indications:
- Strong family history (≥3 affected relatives, autosomal dominant pattern)
- Young-onset dementia (less than 60 years) with family history
- Patient request following genetic counselling
Genes Tested:
- Early-onset familial AD: APP, PSEN1, PSEN2
- FTD: MAPT, GRN, C9orf72
- APOE genotyping: NOT recommended for routine diagnosis (risk factor, not deterministic)
7. Management
Management of dementia is multifaceted, focusing on optimising cognitive function, managing behavioural and psychological symptoms, supporting independence and quality of life, treating comorbidities, and providing comprehensive carer support. [5]
General Principles
- Early diagnosis enables timely intervention and planning
- Shared decision-making involving patient (while they have capacity) and family
- Individualised care plans addressing cognitive, functional, behavioural, and social needs
- Multidisciplinary team approach: GP, memory clinic, old age psychiatry, neurology, occupational therapy, social services, Admiral Nurses
- Advance care planning while patient retains capacity
- Regular review and reassessment (every 6-12 months, more frequently if deteriorating)
Non-Pharmacological Interventions (First-Line for All Patients)
Non-pharmacological interventions form the foundation of dementia care and should be implemented for all patients regardless of severity. [5]
Cognitive Stimulation Therapy (CST):
- Evidence: Cochrane review shows benefits comparable to acetylcholinesterase inhibitors [17]
- Structure: Group sessions (7 participants), twice weekly for 7 weeks (14 sessions)
- Content: Themed activities (word games, reminiscence, music, current affairs)
- Benefits: Improves cognition (MMSE +1.7 points), quality of life
- NICE Recommendation: Offer to all people with mild-moderate dementia
Cognitive Rehabilitation:
- Goal-oriented, individualised therapy
- Patient identifies meaningful goals (remembering names, navigating home)
- Strategies developed collaboratively
Reminiscence Therapy:
- Discussion of past activities, events, experiences (often using photos, music, objects)
- Can be individual or group-based
- Improves mood, engagement, and quality of life
Physical Activity:
- Aerobic exercise 150 minutes/week (if able)
- Benefits: Physical health, mood, cognition, sleep, reduces falls risk
- Can be tailored (walking, dancing, gardening, seated exercises)
Occupational Therapy:
- Home environment assessment and modifications (safety, orientation aids)
- Adaptive equipment to maintain independence
- Activity scheduling and meaningful occupation
Behavioural Management for BPSD:
- Person-centred care: Understand unmet needs (pain, thirst, boredom, overstimulation)
- Environmental modifications: Reduce noise, clutter; increase lighting; orientation cues (clocks, calendars)
- Structured routines: Predictable daily schedule
- Music therapy: Personalised music playlists (reduces agitation)
- Validation therapy: Acknowledge emotions without correcting reality
- Dementia-friendly design: Clear signage, contrasting colours, familiar objects
Pharmacological Treatment
Acetylcholinesterase Inhibitors (AChEIs):
Three AChEIs are available for Alzheimer's disease; choice typically based on tolerability, formulation, and patient preference. [11]
| Drug | Dose | Mechanism | Formulations | Side Effects |
|---|---|---|---|---|
| Donepezil (Aricept) | Start 5 mg OD; increase to 10 mg after 4-6 weeks; (23 mg available but rarely used) | Reversible, non-competitive AChEI | Oral tablets, orodispersible | GI upset (N/V, diarrhoea), bradycardia, insomnia, muscle cramps, syncope |
| Rivastigmine (Exelon) | Oral: Start 1.5 mg BD; titrate to 3-6 mg BD (max 6 mg BD). Patch: Start 4.6 mg/24h; increase to 9.5 mg/24h | Pseudo-irreversible AChEI (also inhibits butyrylcholinesterase) | Oral capsules, liquid, transdermal patch | GI upset (dose-related, less with patch), weight loss, bradycardia |
| Galantamine (Reminyl) | Start 8 mg OD (modified release); increase to 16 mg OD after 4 weeks (max 24 mg OD) | Reversible AChEI + nicotinic receptor modulation | Oral tablets, modified-release capsules, liquid | GI upset, bradycardia, dizziness |
Indications:
- Alzheimer's disease: Mild to moderate (MMSE 10-26)
- Dementia with Lewy bodies: Rivastigmine preferred (better evidence); may improve cognition, hallucinations, and motor symptoms [7]
- Parkinson's disease dementia: Rivastigmine licensed
- NOT effective in FTD (may worsen behavioural symptoms)
- Vascular dementia: Evidence less robust; may be considered if mixed AD/VaD
Efficacy:
- Modest benefit: Mean improvement of 1.5-2 points on MMSE vs placebo [11]
- Functional benefit: Delays decline in ADLs by 6-9 months on average
- Not disease-modifying: Does not alter underlying pathology
- AD2000 Trial: Long-term benefits on institutionalisation or progression not demonstrated, but quality of life benefits recognised [18]
Monitoring:
- Baseline: Cognitive assessment (MMSE/MoCA), functional assessment, ECG (if cardiac history, bradycardia risk)
- Review at 3 months: Assess response (cognitive, functional, behavioural, carer feedback)
- Ongoing: 6-monthly reviews; continue if beneficial and tolerated
- Discontinuation: If no perceived benefit, severe dementia (MMSE less than 10), or intolerable side effects
Contraindications/Cautions:
- Sick sinus syndrome, bradycardia, heart block (especially rivastigmine and donepezil)
- Active peptic ulcer disease
- Severe asthma or COPD (cholinergic bronchospasm risk)
- Seizure history (may lower seizure threshold)
Memantine:
| Drug | Dose | Mechanism | Indication | Side Effects |
|---|---|---|---|---|
| Memantine (Ebixa, Namenda) | Start 5 mg OD; increase by 5 mg weekly to target 10 mg BD (20 mg total daily) | NMDA receptor antagonist (reduces glutamate excitotoxicity) | Moderate to severe AD (MMSE less than 15); can be added to AChEI | Dizziness, headache, confusion, constipation, hypertension |
Indications:
- Moderate-severe Alzheimer's disease (MMSE 10-19): Monotherapy or combination with AChEI
- Severe Alzheimer's disease (MMSE less than 10): Consider if behavioural symptoms or functional decline
- Intolerance to AChEIs: Alternative option
Efficacy:
- Modest benefit: Slows cognitive and functional decline vs placebo [12]
- Combination therapy (AChEI + memantine): Some evidence for additional benefit in moderate-severe AD
- Behavioural symptoms: May reduce agitation and aggression
Vascular Dementia Specific Management:
- No disease-modifying pharmacotherapy for vascular dementia
- Cardiovascular risk factor control (primary intervention):
- "Hypertension management: Target less than 140/90 mmHg (individualise for frail elderly)"
- "Diabetes control: HbA1c less than 7-8% (avoid hypoglycaemia in elderly)"
- "Statin therapy: For atherosclerotic disease, hyperlipidaemia"
- "Antiplatelet therapy: Aspirin or clopidogrel if history of stroke/TIA"
- "Anticoagulation: If atrial fibrillation (assess bleeding risk)"
- Smoking cessation
- Weight management, physical activity
- AChEIs: May be considered if mixed AD/VaD pathology suspected
Frontotemporal Dementia Specific Management:
- No proven pharmacological treatment for FTD
- SSRIs (e.g., sertraline, citalopram): May help with behavioural symptoms (disinhibition, compulsive behaviours, apathy) [8]
- Avoid AChEIs: Not effective; may worsen behavioural symptoms
- Focus on non-pharmacological behavioural interventions
Management of Behavioural and Psychological Symptoms of Dementia (BPSD)
BPSD affects up to 90% of people with dementia at some point and is a major cause of carer distress and institutionalisation. [5]
Stepwise Approach:
1. Identify and Treat Underlying Causes:
- Pain: Unrecognised pain (arthritis, constipation, UTI, pressure sores)
- Infection: UTI, chest infection
- Constipation or urinary retention
- Medication side effects: Anticholinergics, sedatives
- Environmental triggers: Overstimulation, unfamiliar surroundings
- Unmet needs: Hunger, thirst, need for toilet, boredom, loneliness
2. Non-Pharmacological Interventions (First-Line):
- Person-centred care, behavioural analysis (ABC: Antecedent-Behaviour-Consequence)
- Environmental modification
- Music therapy, aromatherapy, multisensory stimulation
- Carer education and support
3. Pharmacological Treatment (Only After Non-Pharmacological Approaches Have Failed):
Antipsychotics (Use With Extreme Caution):
- Indications: Severe agitation or psychosis causing significant distress or risk of harm, when non-pharmacological approaches have failed
- Drugs: Risperidone (licensed for short-term use in severe aggression in AD), quetiapine, olanzapine (second-line)
- Dose: Use lowest effective dose for shortest duration (review every 6 weeks; aim to discontinue after 12 weeks)
- "Risperidone: 0.25-1 mg BD"
- "Quetiapine: 25-100 mg daily"
- "Olanzapine: 2.5-5 mg daily"
- BLACK BOX WARNING: Increased risk of stroke (3-fold) and mortality (1.6-fold) in elderly with dementia [5]
- AVOID in DLB: Severe neuroleptic sensitivity; risk of neuroleptic malignant syndrome (use quetiapine with extreme caution if essential)
- Monitoring: Weekly for first 6 weeks (sedation, falls, extrapyramidal side effects, stroke signs), then monthly
Antidepressants:
- Indications: Comorbid depression, anxiety, agitation
- SSRIs preferred: Sertraline 50-100 mg, citalopram 10-20 mg (beware QTc prolongation > 20 mg)
- Mirtazapine: 15-30 mg (if sleep disturbance, appetite loss)
Other Agents:
- Trazodone: 50-100 mg (for agitation, sleep)
- Benzodiazepines: AVOID (paradoxical agitation, falls, worsening cognition); use only for acute severe distress, short-term
Driving and Dementia
- DVLA must be notified by the patient (legal requirement in UK)
- Mild dementia: May be able to continue driving if annual medical assessment and driving test passed
- Moderate-severe dementia: Driving should cease
- Clinician role: Advise patient and family; document advice; if patient continues to drive unsafely, breach confidentiality and inform DVLA
Capacity Assessment and Advance Care Planning
Mental Capacity Act (2005) Principles:
- Presume capacity unless proven otherwise
- Support people to make their own decisions
- Unwise decisions ≠ lack of capacity
- Best interests: If lacking capacity
- Least restrictive option
Assessing Capacity (Decision-Specific and Time-Specific): Can the person:
- Understand information relevant to the decision?
- Retain that information long enough to make the decision?
- Weigh up the information (consider pros and cons)?
- Communicate their decision?
Advance Care Planning (Initiate Early):
- Lasting Power of Attorney (LPA): Legal authority for someone to make decisions on patient's behalf (Health and Welfare; Property and Financial Affairs)
- Advance Decision to Refuse Treatment (ADRT): Legally binding refusal of specific treatments (must be in writing if refusing life-sustaining treatment)
- Advance Statement: Record of preferences and values (not legally binding but should be considered)
- Preferred Place of Care and Death
- DNACPR (Do Not Attempt Cardiopulmonary Resuscitation): Discuss sensitively; document clearly
Carer Support (Essential Component of Dementia Care)
Carers of people with dementia experience high rates of depression (40%), anxiety, physical health problems, and burnout. [5]
Interventions:
- Carer education programmes: Understanding dementia, communication strategies, behaviour management
- Respite care: Day centres, home care, short residential stays
- Carer support groups: Peer support, shared experiences
- Admiral Nurses: Specialist dementia nurses providing expert practical and emotional support to families
- Cognitive Behavioural Therapy (CBT) for carers: Reduces depression and anxiety
- Carer's assessment: Social services assessment of carer's own needs (separate from patient assessment)
- Financial support: Attendance Allowance, Carer's Allowance, Council Tax reduction
Signposting:
- Alzheimer's Society: Information, support groups, Dementia Advisers
- Dementia UK: Admiral Nurse service
- Age UK: Practical support and advice
Multidisciplinary Team Approach
| Discipline | Role |
|---|---|
| General Practitioner | Coordination, comorbidity management, prescription, carer support |
| Memory Clinic (Old Age Psychiatry/Neurology) | Diagnosis, treatment initiation, specialist review |
| Community Psychiatric Nurse (CPN) | Monitoring, medication support, BPSD management |
| Admiral Nurse | Specialist dementia nursing support for families |
| Occupational Therapist | Home adaptations, safety assessment, meaningful activities |
| Physiotherapist | Mobility, falls prevention, exercise programmes |
| Speech and Language Therapist | Communication strategies, swallowing assessment |
| Dietitian | Nutritional assessment, weight management |
| Social Worker | Care package, safeguarding, financial support, care home placement |
| Clinical Psychologist | Cognitive assessment, psychological therapy for patient/carer |
8. Complications
Dementia is associated with numerous medical, functional, and psychosocial complications that contribute to morbidity, mortality, and carer burden.
| Complication | Frequency | Pathophysiology/Risk Factors | Prevention | Management |
|---|---|---|---|---|
| Falls and Fractures | 50-60% fall annually | Gait disturbance, impaired judgment, medications, environmental hazards | Home safety assessment, strength/balance exercises, medication review, vision correction, hip protectors | Post-fall assessment, fracture management, falls clinic referral |
| Delirium | 30-50% | Acute illness, medications, surgery; superimposed on dementia | Prevent precipitants (infection, dehydration, polypharmacy) | Identify and treat underlying cause; avoid antipsychotics if possible |
| Aspiration Pneumonia | Common in advanced dementia | Dysphagia, impaired cough reflex, supine positioning | SALT assessment, modified diet (thickened fluids), upright positioning, oral care | Antibiotics, consider treatment escalation decisions |
| Malnutrition and Weight Loss | 30-40% | Forgetting to eat, apraxia (difficulty using utensils), dysphagia, depression, decreased sense of smell/taste | Regular meals, assistance with feeding, high-calorie supplements, pleasant dining environment | Dietitian review, treat reversible causes, consider PEG only if reversible cause and patient previously expressed wish |
| Urinary and Faecal Incontinence | 50-70% in moderate-severe | Functional (unable to find/reach toilet), detrusor overactivity, constipation with overflow | Prompted voiding, accessible toilets, continence pads | Exclude UTI, constipation; treat reversible causes; continence aids |
| Pressure Ulcers | 20-30% in advanced dementia | Immobility, malnutrition, incontinence | Repositioning, pressure-relieving mattresses, skin care, nutrition | Wound care, treat infection, pressure relief, nutrition support |
| Infections (UTI, pneumonia) | Very common | Functional decline, incontinence, aspiration, frailty | Hydration, hygiene, pneumococcal/influenza vaccination | Antibiotics; discuss treatment escalation and goals of care |
| Behavioural and Psychological Symptoms (BPSD) | 90% at some point | Neurodegeneration, unmet needs, environmental triggers | Person-centred care, environmental modification, meaningful activities | Non-pharmacological first; antipsychotics only if severe and other approaches failed |
| Wandering and Getting Lost | 40-60% | Disorientation, restlessness, searching for familiar places/people | GPS trackers, ID bracelets, door alarms, safe spaces to walk | Police alert if missing; consider care home if high risk |
| Elder Abuse and Neglect | 10-20% | Vulnerability, dependency, carer stress | Carer support, respite, safeguarding awareness | Safeguarding referral to local authority; police involvement if criminal |
| Carer Burnout | 40-50% | Physical and emotional demands, sleep deprivation, social isolation, financial strain | Respite care, carer support groups, education, Admiral Nurses | Psychological support, antidepressants if indicated, increased respite, care home placement if necessary |
| Medication Errors | Common | Forgetting to take medications, taking multiple doses, inability to manage complex regimens | Simplify regimens, blister packs, carer supervision | Medication review, dosette boxes, district nurse administration |
| Driving Accidents | Increased risk | Impaired judgment, visuospatial deficits, delayed reaction time | DVLA notification, driving assessment | Cease driving if unsafe; support with alternative transport |
| Financial Exploitation | 5-10% reported | Impaired judgment, vulnerability | LPA (Property and Financial Affairs), bank safeguards | Safeguarding referral, legal advice, Court of Protection |
End-of-Life Complications:
- Dysphagia and Feeding Difficulties: Ethical decisions about artificial nutrition and hydration
- Recurrent Infections: Discussions about antibiotic use, treatment escalation, palliative care
- Pain: Often under-recognised and undertreated (assess using pain scales for dementia: PAINAD, Abbey Pain Scale)
9. Prognosis and Outcomes
Natural History and Survival
Dementia is a progressive, ultimately fatal condition. Median survival from diagnosis varies by dementia subtype and age at diagnosis: [19]
| Dementia Subtype | Median Survival from Diagnosis | Range |
|---|---|---|
| Alzheimer's Disease | 8-10 years | 3-20 years |
| Vascular Dementia | 3-5 years | 2-10 years (often shorter due to cardiovascular comorbidity) |
| Dementia with Lewy Bodies | 5-8 years | 2-12 years |
| Frontotemporal Dementia | 6-8 years | 3-15 years (behavioural variant often shorter than language variants) |
| Young-Onset Dementia | Variable | Often longer survival (younger, fewer comorbidities), but faster progression in some genetic forms |
Factors Influencing Survival:
- Age at diagnosis: Younger onset often associated with longer survival but may have faster progression
- Functional status: Lower baseline ADL function → shorter survival
- Comorbidities: Cardiovascular disease, diabetes, chronic kidney disease reduce survival
- Severity at diagnosis: Advanced dementia at diagnosis → shorter survival
- Nutritional status: Malnutrition, weight loss → poor prognosis
- Complications: Recurrent falls, aspiration pneumonia, pressure ulcers → increased mortality
Functional Trajectory
Dementia progresses through stages, though individual variation is considerable:
Mild (MMSE 20-26):
- Duration: 2-4 years
- Cognition: Noticeable memory loss, word-finding difficulty, disorientation in unfamiliar places
- Function: IADL impairment (finances, medications, complex tasks); ADLs largely preserved
- Behaviour: Mood changes, apathy, occasional agitation
- Care needs: Supervision for IADLs, prompting
Moderate (MMSE 10-19):
- Duration: 2-10 years
- Cognition: Significant memory impairment, disorientation to time and place, impaired judgment
- Function: IADL dependency; ADL impairment (bathing, dressing, toileting require assistance)
- Behaviour: BPSD common (agitation, psychosis, wandering, sleep disturbance)
- Care needs: Daily support, supervision for safety, carer respite
Severe (MMSE less than 10):
- Duration: 1-3 years
- Cognition: Severe memory loss, minimal verbal communication, recognition loss (including family)
- Function: Total ADL dependency (feeding, mobility, continence)
- Behaviour: Apathy predominates; agitation may persist
- Physical: Immobility, contractures, dysphagia, incontinence, recurrent infections
- Care needs: 24-hour care, often residential/nursing home, end-of-life care planning
Prognostic Factors for Rapid Decline
- Older age at diagnosis
- Male sex (in some studies)
- Lower baseline MMSE
- Presence of psychotic symptoms (hallucinations, delusions)
- Extrapyramidal signs (rigidity, bradykinesia)
- Vascular risk factors and white matter disease
- Comorbidities (diabetes, stroke, heart failure)
- Rapid initial decline (predictive of continued rapid progression)
Quality of Life Considerations
Quality of life can be maintained even in advanced dementia through:
- Person-centred care focusing on comfort, dignity, and preferences
- Meaningful activities and social interaction
- Pain and symptom management
- Carer wellbeing (directly impacts patient QoL)
- Appropriate care setting (home vs residential care)
10. Prevention and Risk Reduction
Evidence-Based Prevention Strategies
The Lancet Commission (2020) estimated that 40% of dementia cases could be prevented or delayed by addressing 12 modifiable risk factors across the life course. [6] This represents a major public health opportunity.
Life-Course Approach to Dementia Prevention:
Early Life (Childhood, Adolescence):
- Education: Maximise educational attainment (7% PAF)
- Higher education builds cognitive reserve
- Continue learning throughout life
Midlife (45-65 years): 2. Hearing Loss: Prevent and treat (8% PAF)
- Use hearing protection in noisy environments
- Hearing aid use if hearing impairment detected
- Traumatic Brain Injury: Prevent head injury (3% PAF)
- Helmet use (cycling, motorcycling, contact sports)
- Falls prevention
- Hypertension: Control BP (2% PAF)
- Target less than 130/80 mmHg in midlife
- Antihypertensive medication if indicated
- Obesity: Maintain healthy weight (1% PAF)
- BMI 18.5-24.9 kg/m² in midlife
- Excessive Alcohol: Limit intake (1% PAF)
- ≤14 units/week (UK guidelines)
- Avoid binge drinking
Later Life (≥65 years): 7. Smoking: Cessation (5% PAF)
- Smoking cessation at any age beneficial
- Depression: Treat effectively (4% PAF)
- Screen and treat depression
- Psychological therapies, antidepressants
- Social Isolation: Maintain social contact (4% PAF)
- Social activities, community engagement
- Distinct from loneliness; focus on contact quantity
- Physical Inactivity: Regular exercise (2% PAF)
- 150 min/week moderate aerobic activity
- Resistance training twice weekly
- Diabetes: Optimise glycaemic control (1% PAF)
- HbA1c less than 7-8% (individualise in elderly)
- Prevent diabetes (weight management, exercise)
- Air Pollution: Reduce exposure (2% PAF)
- Policy-level interventions
- Avoid high-traffic areas for exercise
FINGER Trial (Multidomain Intervention)
The Finnish Geriatric Intervention Study to Prevent Cognitive Impairment and Disability (FINGER) was a landmark RCT demonstrating that multidomain lifestyle intervention can prevent cognitive decline in at-risk older adults. [20]
Intervention Components:
- Nutritional guidance (Mediterranean-style diet, fish, vegetables, healthy fats)
- Physical exercise (aerobic + strength training)
- Cognitive training (computer-based exercises)
- Social activity and vascular risk management
Results:
- Improved cognition in intervention group vs control (NTB total score improvement)
- Executive function and processing speed particularly benefited
- First RCT proof-of-concept that dementia risk is modifiable
Implications: Multidomain interventions targeting multiple risk factors simultaneously may be more effective than single-domain approaches. Worldwide FINGERS trials underway in diverse populations.
Pharmacological Prevention
No Proven Pharmacological Prevention:
- Statins: No evidence for primary prevention of dementia
- Antihypertensives: Benefit likely through BP control, not drug-specific effect
- NSAIDs: No benefit; potential harms
- Vitamin E, Ginkgo biloba: No evidence of benefit
- Hormone Replacement Therapy: Increased dementia risk if started after age 65
Screening for Dementia
Systematic population screening for dementia is NOT recommended (NICE, USPSTF):
- Uncertain benefit of early detection in asymptomatic individuals
- Risk of overdiagnosis and labelling
- No disease-modifying treatments available
Case-finding (opportunistic screening) may be appropriate:
- Individuals with subjective cognitive complaints
- Comorbid conditions (stroke, Parkinson's, depression)
- Presenting with functional decline, BPSD, or falls
11. Guidelines and Evidence
Key Clinical Guidelines
1. NICE NG97: Dementia — Assessment, Management and Support (2018, updated 2024) [4]
- Comprehensive guidance on diagnosis, pharmacological and non-pharmacological management
- Cognitive stimulation therapy recommended for all with mild-moderate dementia
- AChEIs for mild-moderate AD; memantine for moderate-severe AD
- Antipsychotics only for severe distress/risk of harm, after non-pharmacological approaches
2. NICE Technology Appraisals:
- TA217 (2018): Donepezil, galantamine, rivastigmine, and memantine for Alzheimer's
- Recommends AChEIs as options for mild-moderate AD
- Memantine for moderate AD (intolerant of AChEIs) or severe AD
3. Scottish Intercollegiate Guidelines Network (SIGN 157): Risk Reduction and Management of Dementia (2022)
- Evidence-based recommendations on risk factor modification
- Diagnosis and management of dementia subtypes
4. American Academy of Neurology (AAN) Guidelines:
- MCI practice guideline (2018)
- Dementia diagnosis and management
5. European Academy of Neurology/European Alzheimer's Disease Consortium Guidelines (2023)
- Diagnosis of mild cognitive impairment and dementia
Landmark Trials and Key Evidence
AD2000 Trial (Courtney et al., 2004) [18]
- Design: RCT of donepezil vs placebo in AD (565 patients, community-dwelling)
- Key Finding: Donepezil provided modest cognitive benefit (MMSE improvement) but no significant delay to institutionalisation or progression of disability at 3 years
- Clinical Impact: Tempered initial enthusiasm; AChEIs provide symptomatic benefit but are not disease-modifying. Manage patient and carer expectations.
FINGER Trial (Ngandu et al., 2015) [20]
- Design: RCT of multidomain lifestyle intervention in at-risk older adults (1,260 participants, Finland)
- Intervention: Diet, exercise, cognitive training, vascular risk management
- Key Finding: Multidomain intervention improved or maintained cognitive function vs control (NTB total score +0.20 SD)
- Clinical Impact: First proof-of-concept RCT that dementia is preventable through lifestyle modification. Worldwide FINGERS consortium now implementing similar trials.
Lancet Commission on Dementia Prevention, Intervention, and Care (2020) [6]
- Key Finding: 40% of dementia potentially preventable by addressing 12 modifiable risk factors across life course
- Clinical Impact: Public health focus on prevention; hearing loss identified as largest single modifiable factor (8% PAF)
Donepezil and Memantine for Moderate-to-Severe Alzheimer's Disease (Howard et al., 2012) [21]
- Design: RCT (295 patients with moderate-severe AD on donepezil), factorial design comparing continuation vs discontinuation of donepezil, and memantine vs placebo
- Key Finding: Continuing donepezil and adding memantine both showed benefit on cognition and function
- Clinical Impact: Supports continuation of AChEI and addition of memantine in moderate-severe AD
Cochrane Review: Cognitive Stimulation for Dementia (Woods et al., 2012) [17]
- Key Finding: CST improves cognition (similar magnitude to AChEIs) and quality of life in mild-moderate dementia
- Clinical Impact: CST recommended as first-line intervention (NICE NG97); non-pharmacological intervention with robust evidence
DaTscan in DLB Diagnosis (McKeith et al., 2007) [7]
- Key Finding: DaTscan has 78% sensitivity and 90% specificity for differentiating DLB from non-DLB dementias
- Clinical Impact: Useful adjunct in uncertain cases; helps identify patients with neuroleptic sensitivity
12. Viva and Exam Preparation
Common Viva Questions
Opening Statement:
"Dementia is a clinical syndrome characterised by acquired, progressive decline in two or more cognitive domains—typically memory, executive function, language, visuospatial ability, or social cognition—severe enough to impair daily functioning and independence. It is not a single disease but results from various underlying neurodegenerative and vascular pathologies. Alzheimer's disease is the most common cause, accounting for 60-70% of cases, followed by vascular dementia at 15-20%, dementia with Lewy bodies at 5-15%, and frontotemporal dementia at 2-5%. The global prevalence is approximately 7% in adults aged 60 and over, with numbers projected to reach 152 million by 2050. Diagnosis involves comprehensive cognitive assessment, exclusion of reversible causes, and neuroimaging. Management focuses on pharmacological interventions such as acetylcholinesterase inhibitors for Alzheimer's, non-pharmacological therapies including cognitive stimulation, and holistic support for patients and carers."
Q1: What are the causes of dementia?
A: Dementia is caused by a range of neurodegenerative, vascular, and other pathologies:
Neurodegenerative (Most Common):
- Alzheimer's disease (60-70%): Amyloid-beta plaques and tau neurofibrillary tangles
- Dementia with Lewy bodies (5-15%): Alpha-synuclein Lewy body deposition
- Frontotemporal dementia (2-5%): Tau or TDP-43 proteinopathies; frontal/temporal atrophy
- Parkinson's disease dementia: Alpha-synuclein pathology; dementia occurs > 1 year after motor symptoms
Vascular (15-20%): 5. Multi-infarct dementia: Multiple large vessel infarcts 6. Small vessel disease: Lacunar infarcts, white matter hyperintensities 7. Strategic infarct dementia: Single infarct in critical location (thalamus, hippocampus)
Mixed Pathology (10-15%): 8. Mixed Alzheimer's and vascular dementia: Overlapping pathologies
Reversible/Treatable (5-10%): 9. Metabolic: B12 deficiency, hypothyroidism, hypercalcaemia 10. Structural: Normal pressure hydrocephalus, subdural haematoma, brain tumour 11. Psychiatric: Depression ("pseudodementia") 12. Infectious: Neurosyphilis, HIV dementia 13. Toxic: Alcohol-related dementia, heavy metals, medications (anticholinergics)
Other: 14. Prion disease: Creutzfeldt-Jakob disease (rapid progression, myoclonus) 15. Huntington's disease: Autosomal dominant, chorea, psychiatric features
Q2: How would you investigate a patient presenting with memory loss?
A: I would take a systematic approach:
History:
- Cognitive symptoms: Nature, onset (gradual vs sudden), progression (gradual vs stepwise), affected domains (memory, language, executive, visuospatial)
- Functional impact: IADLs and ADLs
- Behavioural symptoms: Mood, personality change, psychosis, sleep, agitation
- Corroborative history from informant (essential)
- Medical history: Cardiovascular risk factors, stroke, head injury, depression, alcohol
- Medications: Anticholinergics, sedatives
- Family history: Early-onset dementia (genetic forms)
Examination:
- Cognitive assessment: MMSE, MoCA, or ACE-III
- Neurological exam: Parkinsonism, focal signs, primitive reflexes, gait
- Cardiovascular: BP, heart rhythm (AF), carotid bruits
- General: Nutrition, self-care, mood
Investigations:
Bloods (exclude reversible causes):
- FBC, U&E, LFTs, calcium, glucose, TSH, B12, folate
- Consider: Syphilis/HIV serology (young onset), autoimmune screen (rapid), ESR/CRP (inflammatory)
Neuroimaging (MANDATORY):
- MRI brain (preferred): Exclude structural causes (tumour, SDH, NPH), identify atrophy patterns (medial temporal/AD, frontal-temporal/FTD), vascular changes (infarcts, WMH)
- CT brain: If MRI contraindicated
Additional tests (if indicated):
- DaTscan: If suspected DLB (differentiate from AD)
- CSF analysis: Young onset, rapid progression, atypical (biomarkers: Aβ42, tau, 14-3-3)
- Genetic testing: Strong family history, early onset (APP, PSEN1, PSEN2, MAPT, GRN)
- EEG: If suspected seizures or CJD
Q3: What is your management approach to a patient with newly diagnosed Alzheimer's disease?
A: My management would be comprehensive and multidisciplinary:
1. Confirm Diagnosis and Subtype:
- Ensure diagnostic criteria met; exclude reversible causes; neuroimaging completed
2. Non-Pharmacological Interventions (First-Line for All):
- Cognitive Stimulation Therapy (CST): Group sessions, evidence-based (Cochrane review), improves cognition and QoL
- Physical activity: 150 min/week aerobic exercise
- Occupational therapy: Home safety assessment, adaptations, meaningful activities
- Social engagement: Activities, day centres
3. Pharmacological Treatment:
- Mild-moderate AD (MMSE 10-26): Acetylcholinesterase inhibitor
- Donepezil 5 mg OD → 10 mg OD, OR
- Rivastigmine (oral or patch), OR
- Galantamine
- Review at 3 months; continue if beneficial
- Moderate-severe AD (MMSE less than 20): Add memantine 10 mg BD (titrate from 5 mg OD)
- Treat comorbidities: Optimise vascular risk factors (BP, diabetes, lipids, antiplatelet if indicated)
4. Address BPSD:
- Non-pharmacological first (person-centred care, environmental modification, unmet needs)
- Pharmacological if severe (antidepressants for depression; antipsychotics only if severe agitation/psychosis and non-pharm failed, short-term, low dose)
5. Advance Care Planning (CRITICAL):
- Discuss while patient has capacity:
- Lasting Power of Attorney (health and welfare; property and financial affairs)
- Advance Decision to Refuse Treatment (if desired)
- Preferences for future care, resuscitation status
- Driving: Notify DVLA; arrange assessment
6. Carer Support:
- Education about dementia
- Carer support groups, Admiral Nurses
- Respite care
- Carer's assessment (social services)
7. Review and Monitor:
- 6-monthly reviews: Cognition, function, mood, BPSD, medication review, carer wellbeing
- Adjust management as disease progresses
8. End-of-Life Planning:
- Anticipatory care planning in moderate-severe dementia
- Palliative care referral when appropriate
Q4: How do you differentiate between Alzheimer's disease and dementia with Lewy bodies?
A: Differentiation is based on clinical features and investigations:
Clinical Features:
| Feature | Alzheimer's Disease | Dementia with Lewy Bodies |
|---|---|---|
| Onset of symptoms | Gradual, insidious | Insidious, but fluctuating course |
| Early prominent feature | Episodic memory loss | Fluctuating cognition, visual hallucinations |
| Hallucinations | Late, if at all | Early, recurrent visual hallucinations (well-formed, detailed) |
| Fluctuation | Stable, progressive decline | Marked fluctuations in attention and alertness (day-to-day or within-day) |
| Parkinsonism | Absent early; may occur late | Spontaneous parkinsonism (rigidity, bradykinesia; tremor less common) |
| REM sleep behaviour disorder | Absent | Acting out dreams (may precede dementia by years) |
| Neuroleptic sensitivity | Typical response | Severe sensitivity (risk of NMS; AVOID antipsychotics) |
| Autonomic dysfunction | Absent early | Orthostatic hypotension, syncope, constipation |
| Cognitive profile | Memory predominant | Visuospatial and executive dysfunction early; memory relatively preserved |
Investigations:
| Investigation | Alzheimer's Disease | Dementia with Lewy Bodies |
|---|---|---|
| MRI brain | Hippocampal and medial temporal lobe atrophy | Relative preservation of medial temporal lobes |
| DaTscan (SPECT) | Normal striatal dopamine transporter uptake | Reduced striatal uptake (abnormal scan) |
| FDG-PET | Temporoparietal, posterior cingulate hypometabolism | Occipital hypometabolism; cingulate island sign |
Diagnostic Criteria (DLB Consortium):
Core features (≥2 for probable DLB; 1 for possible DLB):
- Fluctuating cognition
- Recurrent visual hallucinations
- REM sleep behaviour disorder
- Spontaneous parkinsonism
Supportive features:
- Severe neuroleptic sensitivity
- Repeated falls, syncope, transient loss of consciousness
- Autonomic dysfunction
- Non-visual hallucinations
- Delusions
- Apathy, anxiety, depression
Management Implications:
- DLB: Rivastigmine (AChEI) often very effective; AVOID antipsychotics (use quetiapine with extreme caution only if essential)
- AD: AChEIs or memantine; antipsychotics safer (but still caution)
Q5: A 72-year-old man with dementia is wandering at night and becoming aggressive. How would you manage his behavioural symptoms?
A: I would use a structured approach to BPSD:
1. Assess for Underlying Causes (ABC Analysis):
- Antecedents: What triggers the behaviour? Time of day, specific situations, people?
- Medical causes:
- "Pain: Arthritis, constipation, UTI, pressure sores (use Abbey Pain Scale)"
- "Infection: UTI, chest infection"
- Constipation or urinary retention
- "Delirium: Acute superimposed illness"
- Medications: Anticholinergics, sedatives, new medications
- Environmental triggers: Overstimulation, noise, unfamiliar environment, unmet needs (hunger, thirst, boredom, toileting)
- Sleep hygiene: Day-night reversal, sleep apnoea
Investigations:
- Urinalysis and culture (UTI)
- FBC, CRP (infection)
- Medication review
- Pain assessment
2. Non-Pharmacological Interventions (First-Line):
- Person-centred care: Understand unmet needs (What is the patient trying to communicate?)
- Environmental modification:
- Reduce noise and clutter
- Increase daytime lighting, dim lights at night
- Orientation cues (clocks, calendars)
- Safe wandering space (secure garden, circular corridor)
- Structured routine: Predictable daily schedule
- Music therapy: Personalised playlists (reduces agitation)
- Exercise: Daytime physical activity (improves sleep)
- Avoid confrontation: Validation, distraction, reassurance
- Carer education: De-escalation techniques, communication strategies
3. Review Existing Dementia Medications:
- Is patient on AChEI? (May improve BPSD in DLB)
- Consider memantine if not already prescribed (may reduce agitation)
4. Pharmacological Treatment (Only if Non-Pharmacological Ineffective and Severe Distress/Risk):
Depression/Anxiety:
- Sertraline 50-100 mg OD, or
- Citalopram 10-20 mg OD (caution: QTc prolongation > 20 mg), or
- Mirtazapine 15-30 mg nocte (if sleep disturbance, appetite loss)
Agitation/Aggression (Severe, Failed Non-Pharm):
- Antipsychotics (LAST RESORT):
- Assess dementia subtype (AVOID if DLB)
- "Discuss risks with family: Stroke (3x risk), mortality (1.6x risk)"
- Risperidone 0.25-0.5 mg BD (licensed for short-term use in AD with severe aggression)
- Quetiapine 25-50 mg (if DLB, extreme caution)
- Use lowest dose, shortest duration (6-12 weeks maximum)
- Review weekly initially (falls, sedation, EPS, stroke signs)
- Attempt to discontinue after 12 weeks
Alternative:
- Trazodone 50-100 mg (for agitation, sleep; less evidence but safer than antipsychotics)
Avoid:
- Benzodiazepines (paradoxical agitation, falls, worsening cognition)
5. Carer Support:
- Respite care (urgent need if carer burnout)
- Admiral Nurse referral
- Carer education: Behaviour management strategies
- Consider increased care package or residential placement if severe and unmanageable at home
6. Safety:
- Safeguarding assessment: Is patient or others at risk?
- Home safety: Door alarms, motion sensors, remove hazards
7. Review and Reassess:
- Weekly initially, then fortnightly
- Document response to interventions
- Taper and discontinue antipsychotics if behaviour improves
High-Yield Facts for Exams
Epidemiology:
- Dementia prevalence doubles every 5 years after age 65
- 40% of dementia potentially preventable (Lancet Commission 2020)
- Hearing loss is single largest modifiable risk factor (8% PAF)
Diagnosis:
- MoCA more sensitive than MMSE for MCI and executive dysfunction
- MRI brain is MANDATORY to exclude structural causes and identify subtype
- DaTscan differentiates DLB from AD (reduced striatal uptake in DLB)
Reversible Causes (Remember "DEMENTIA"):
- Depression, Drugs (anticholinergics)
- Endocrine (hypothyroidism, hypercalcaemia)
- Metabolic (B12, folate, glucose)
- Emotional (depression)
- Normal Pressure Hydrocephalus (triad: gait, cognition, incontinence)
- Tumour, Trauma (subdural haematoma)
- Infection (syphilis, HIV)
- Alcohol
Management:
- CST has evidence comparable to AChEIs (Cochrane review)
- AChEIs improve MMSE by ~1.5-2 points vs placebo; modest benefit
- Memantine for moderate-severe AD (MMSE 10-19 or less than 10)
- Rivastigmine preferred for DLB; AVOID antipsychotics in DLB (neuroleptic sensitivity)
- Antipsychotics increase stroke risk 3-fold and mortality 1.6-fold in dementia; use only for severe BPSD after non-pharm failed
Red Flags:
- Rapid decline (less than 6 months) → CJD, autoimmune, malignancy
- Young onset (less than 55 years) → FTD, genetic, inflammatory
- Triad (gait + cognition + incontinence) → NPH (reversible)
- Early visual hallucinations → DLB (neuroleptic sensitivity)
13. Patient and Layperson Explanation
What is Dementia?
Dementia is a condition where your brain's ability to think, remember, and carry out everyday tasks gradually gets worse over time. It's caused by diseases that damage brain cells. Dementia is not a normal part of getting older—many people live into their 90s and beyond without developing dementia.
What Causes Dementia?
The most common cause is Alzheimer's disease (about 2 in 3 cases), where abnormal proteins build up in the brain and damage brain cells. Other causes include:
- Vascular dementia: Caused by problems with blood supply to the brain (like mini-strokes)
- Dementia with Lewy bodies: Caused by protein deposits in brain cells
- Frontotemporal dementia: Affects the front and sides of the brain; often starts younger (under 65)
Some causes of memory problems are treatable, like vitamin B12 deficiency, thyroid problems, or depression, which is why it's important to see a doctor.
What Are the Symptoms?
Early symptoms include:
- Forgetting recent events or conversations (asking the same question repeatedly)
- Difficulty finding words or following conversations
- Getting lost in familiar places
- Trouble with everyday tasks like managing money, cooking, or taking medications
- Changes in mood or personality (becoming withdrawn, anxious, or frustrated)
As dementia progresses:
- Needing help with personal care (washing, dressing)
- Difficulty recognising family and friends
- Confusion about time and place
- Changes in behaviour (agitation, wandering, sleep problems)
How is Dementia Diagnosed?
Your doctor will:
- Ask about your symptoms and talk to someone who knows you well (family member, friend)
- Do memory and thinking tests (like remembering words, drawing shapes, answering questions)
- Arrange blood tests to check for treatable causes (vitamin deficiencies, thyroid problems)
- Arrange a brain scan (CT or MRI) to look at your brain structure and rule out other problems
How is Dementia Treated?
There is currently no cure for most types of dementia, but treatments can help with symptoms and quality of life:
1. Medications:
- Tablets for memory (donepezil, rivastigmine, galantamine): Can help with memory and thinking for some people with Alzheimer's disease. They don't cure dementia but can slow down symptoms for a while.
- Memantine: For more advanced Alzheimer's disease
2. Activities and Therapies:
- Cognitive stimulation therapy: Group activities involving puzzles, discussions, music—proven to help memory and enjoyment
- Physical exercise: Walking, dancing, gardening—helps body and brain
- Social activities: Seeing friends, joining groups, day centres
- Reminiscence: Looking at old photos, listening to familiar music
3. Managing Other Symptoms:
- Treatment for low mood or anxiety
- Help with sleep problems
- Support if you become confused or agitated
4. Practical Support:
- Occupational therapist: Can make your home safer and easier to navigate
- Social worker: Can arrange care and support services
- Admiral Nurses: Specialist dementia nurses who support families
What Can I Do to Reduce My Risk?
Research shows that up to 4 in 10 cases of dementia could be prevented by:
- Staying physically active: Aim for 150 minutes of exercise per week (walking, swimming, dancing)
- Keeping your brain active: Reading, puzzles, learning new things
- Staying socially connected: Regular contact with family and friends
- Looking after your heart: Control blood pressure, cholesterol, and diabetes; don't smoke
- Protecting your hearing: Use hearing aids if needed
- Limiting alcohol: No more than 14 units per week
- Maintaining a healthy weight: Especially in midlife
- Treating depression: If you feel low, seek help
Living Well with Dementia
Many people live well with dementia, especially with the right support:
- Make plans early: While you can, discuss your wishes for future care, appoint someone you trust to make decisions if needed (Lasting Power of Attorney)
- Stay active and involved: Continue hobbies and activities you enjoy
- Ask for help: Don't struggle alone—many services are available
- Join support groups: Alzheimer's Society, Dementia UK, local groups
Support for Family and Carers
Caring for someone with dementia can be very demanding. Support is available:
- Respite care: Short breaks so you can rest
- Carer support groups: Meet others in similar situations
- Admiral Nurses: Specialist support for carers
- Financial help: Attendance Allowance, Carer's Allowance
When to Seek Help
See your doctor if you or someone you know has:
- Memory problems affecting daily life
- Difficulty with familiar tasks
- Confusion about time or place
- Changes in mood, personality, or behaviour
- Getting lost in familiar places
Early diagnosis helps:
- Access treatments and support sooner
- Plan for the future while you can
- Rule out treatable causes
14. References
Primary Guidelines
-
Cao Q, Tan CC, Xu W, et al. The prevalence of dementia: a systematic review and meta-analysis. J Alzheimers Dis. 2020;73(3):1157-1166. doi:10.3233/JAD-191092
-
Scheltens P, De Strooper B, Kivipelto M, et al. Alzheimer's disease. Lancet. 2021;397(10284):1577-1590. doi:10.1016/S0140-6736(20)32205-4
-
Prince M, Bryce R, Albanese E, et al. The global prevalence of dementia: a systematic review and metaanalysis. Alzheimers Dement. 2013;9(1):63-75.e2. doi:10.1016/j.jalz.2012.11.007
-
National Institute for Health and Care Excellence. Dementia: assessment, management and support for people living with dementia and their carers (NG97). 2018, updated 2024. Available at: https://www.nice.org.uk/guidance/ng97
-
Livingston G, Huntley J, Sommerlad A, et al. Dementia prevention, intervention, and care: 2020 report of the Lancet Commission. Lancet. 2020;396(10248):413-446. doi:10.1016/S0140-6736(20)30367-6
-
Livingston G, Huntley J, Sommerlad A, et al. Dementia prevention, intervention, and care: 2020 report of the Lancet Commission. Lancet. 2020;396(10248):413-446. doi:10.1016/S0140-6736(20)30367-6
-
McKeith IG, Boeve BF, Dickson DW, et al. Diagnosis and management of dementia with Lewy bodies: Fourth consensus report of the DLB Consortium. Neurology. 2017;89(1):88-100. doi:10.1212/WNL.0000000000004058
-
Olney NT, Spina S, Miller BL. Frontotemporal dementia. Neurol Clin. 2017;35(2):339-374. doi:10.1016/j.ncl.2017.01.008
-
Hejl A, Høgh P, Waldemar G. Potentially reversible conditions in 1000 consecutive memory clinic patients. J Neurol Neurosurg Psychiatry. 2002;73(4):390-394. doi:10.1136/jnnp.73.4.390
-
Hendriks S, Peetoom K, Bakker C, et al. Global prevalence of young-onset dementia: a systematic review and meta-analysis. JAMA Neurol. 2021;78(9):1080-1090. doi:10.1001/jamaneurol.2021.2161
-
Birks JS, Harvey RJ. Donepezil for dementia due to Alzheimer's disease. Cochrane Database Syst Rev. 2018;6(6):CD001190. doi:10.1002/14651858.CD001190.pub3
-
McShane R, Westby MJ, Roberts E, et al. Memantine for dementia. Cochrane Database Syst Rev. 2019;3(3):CD003154. doi:10.1002/14651858.CD003154.pub6
-
Liu CC, Liu CC, Kanekiyo T, et al. Apolipoprotein E and Alzheimer disease: risk, mechanisms and therapy. Nat Rev Neurol. 2013;9(2):106-118. doi:10.1038/nrneurol.2012.263
-
Nasreddine ZS, Phillips NA, Bédirian V, et al. The Montreal Cognitive Assessment, MoCA: a brief screening tool for mild cognitive impairment. J Am Geriatr Soc. 2005;53(4):695-699. doi:10.1111/j.1532-5415.2005.53221.x
-
Frisoni GB, Fox NC, Jack CR Jr, et al. The clinical use of structural MRI in Alzheimer disease. Nat Rev Neurol. 2010;6(2):67-77. doi:10.1038/nrneurol.2009.215
-
Blennow K, Zetterberg H. Biomarkers for Alzheimer's disease: current status and prospects for the future. J Intern Med. 2018;284(6):643-663. doi:10.1111/joim.12816
-
Woods B, Aguirre E, Spector AE, Orrell M. Cognitive stimulation to improve cognitive functioning in people with dementia. Cochrane Database Syst Rev. 2012;2:CD005562. doi:10.1002/14651858.CD005562.pub2
-
Courtney C, Farrell D, Gray R, et al. Long-term donepezil treatment in 565 patients with Alzheimer's disease (AD2000): randomised double-blind trial. Lancet. 2004;363(9427):2105-2115. doi:10.1016/S0140-6736(04)16499-4
-
Todd S, Barr S, Roberts M, Passmore AP. Survival in dementia and predictors of mortality: a review. Int J Geriatr Psychiatry. 2013;28(11):1109-1124. doi:10.1002/gps.3946
-
Ngandu T, Lehtisalo J, Solomon A, et al. A 2 year multidomain intervention of diet, exercise, cognitive training, and vascular risk monitoring versus control to prevent cognitive decline in at-risk elderly people (FINGER): a randomised controlled trial. Lancet. 2015;385(9984):2255-2263. doi:10.1016/S0140-6736(15)60461-5
-
Howard R, McShane R, Lindesay J, et al. Donepezil and memantine for moderate-to-severe Alzheimer's disease. N Engl J Med. 2012;366(10):893-903. doi:10.1056/NEJMoa1106668
-
Bai W, Chen P, Cai H, et al. Worldwide prevalence of mild cognitive impairment among community dwellers aged 50 years and older: a meta-analysis and systematic review of epidemiology studies. Age Ageing. 2022;51(8):afac173. doi:10.1093/ageing/afac173
Further Resources
- Alzheimer's Society: Information, support groups, Dementia Advisers. Available at: https://www.alzheimers.org.uk
- Dementia UK: Admiral Nurse service for specialist family support. Available at: https://www.dementiauk.org
- Age UK: Practical support and advice for older people. Available at: https://www.ageuk.org.uk
- NHS Dementia Guide: Patient information and local services. Available at: https://www.nhs.uk/conditions/dementia
Medical Disclaimer: MedVellum content is for educational purposes and clinical reference. Clinical decisions should account for individual patient circumstances. Always consult appropriate guidelines and specialists for patient care.
Learning map
Use these linked topics to study the concept in sequence and compare related presentations.
Prerequisites
Start here if you need the foundation before this topic.
- Cognitive Function Assessment
- Normal Aging vs Pathological Decline
Differentials
Competing diagnoses and look-alikes to compare.
- Delirium
- Depression (Pseudodementia)
- Mild Cognitive Impairment
Consequences
Complications and downstream problems to keep in mind.
- Delirium in Dementia
- End-of-Life Care Planning