Major Depressive Disorder in Adults
Major Depressive Disorder (MDD) is a common, serious, and potentially life-threatening psychiatric condition characteriz... MRCP, PLAB exam preparation.
What matters first
Major Depressive Disorder (MDD) is a common, serious, and potentially life-threatening psychiatric condition characteriz... MRCP, PLAB exam preparation.
Active suicidal ideation with plan, intent, or access to means
9 Jan 2026
Generated educational material; verify before clinical use.
Visible references section
See the concept before reading it
Study the key anatomy, imaging, and decision pathways as full teaching plates.
Clinical board
A visual summary of the highest-yield teaching signals on this page.
Urgent signals
Safety-critical features pulled from the topic metadata.
- Active suicidal ideation with plan, intent, or access to means
- Psychotic features (mood-congruent hallucinations or delusions)
- Severe psychomotor retardation or catatonic features
- Complete refusal of food or fluids with dehydration
Exam focus
Current exam surfaces linked to this topic.
- MRCP
- PLAB
- MRCPsych
- FRANZCP
Linked comparisons
Differentials and adjacent topics worth opening next.
- Bipolar Disorder
- Persistent Depressive Disorder
Content status and exam context
This page is AI-generated educational content. It may contain errors or omissions and is not a substitute for current guidelines, local protocols, senior clinical judgement, or professional medical advice.
MedVellum does not claim an individual clinician reviewer, board certification, or professional credential for this page unless a future version names a real, verifiable reviewer.
Clinical explanation and evidence
SECTION 1: Clinical Overview
1.1 Summary
Major Depressive Disorder (MDD) is a common, serious, and potentially life-threatening psychiatric condition characterized by persistent depressed mood and/or anhedonia accompanied by neurovegetative symptoms lasting at least two weeks. [1] MDD represents a syndrome of biological, psychological, and social dysfunction that profoundly impairs an individual's ability to function across all life domains—occupational, interpersonal, and self-care. The Global Burden of Disease Study 2019 identified MDD as the second leading cause of years lived with disability worldwide, affecting approximately 280 million people globally. [2]
The pathophysiology of MDD involves complex interactions between genetic vulnerability (heritability 35-40%), hypothalamic-pituitary-adrenal (HPA) axis dysregulation, monoamine neurotransmitter deficiency, reduced neurotrophic support (particularly Brain-Derived Neurotrophic Factor), neuroinflammation, and disrupted neural network connectivity. [3] These biological substrates interact with psychological vulnerabilities (negative cognitive schemas, rumination) and social stressors (loss, isolation, trauma) in a biopsychosocial model.
Diagnosis is clinical, based on DSM-5 or ICD-11 criteria, with validated screening instruments (PHQ-9) facilitating detection in primary care. The differential diagnosis is broad and includes bipolar disorder, persistent depressive disorder (dysthymia), adjustment disorder, grief reactions, substance-induced mood disorders, and mood disorders secondary to medical conditions (hypothyroidism, stroke, malignancy). [4]
Management follows a stepped-care model stratified by severity. Mild depression may respond to active monitoring and low-intensity psychological interventions. Moderate-to-severe depression requires pharmacotherapy (first-line: SSRIs) combined with evidence-based psychotherapy (CBT, IPT). Treatment-resistant cases benefit from switching antidepressant class, augmentation strategies (lithium, atypical antipsychotics), or neuromodulation (ECT, rTMS, esketamine). [5] Prognosis with adequate treatment is generally favorable, though recurrence rates are substantial (50% after one episode, 80% after two, 90% after three), necessitating maintenance therapy in recurrent cases. [6]
Suicide remains the most devastating complication, with lifetime suicide mortality of 4-7% in those hospitalized for MDD. Comprehensive suicide risk assessment and safety planning are mandatory components of care. [7]
1.2 Key Facts
| Domain | Key Fact | Source |
|---|---|---|
| Definition | ≥5 symptoms (including depressed mood or anhedonia) for ≥2 weeks causing significant functional impairment | DSM-5 [1] |
| Global Prevalence | 4.4% point prevalence (322 million affected) | WHO 2017 [2] |
| 12-Month Prevalence | 7.1% in US adults | NIMH 2020 [8] |
| Lifetime Prevalence | 16.6% (US), 10-20% globally | NCS-R [9] |
| Incidence | 3.0 per 100 person-years (women), 1.8 per 100 (men) | PMID: 29102357 [10] |
| Sex Ratio | Female:Male = 2:1 | Consistent across cultures [9] |
| Peak Onset Age | 18-29 years | DSM-5 [1] |
| Disability Burden | 2nd leading cause of YLDs globally | GBD 2019 [2] |
| Suicide Mortality | 4-7% lifetime in hospitalized patients | PMID: 15629203 [7] |
| Remission Rate | 30-40% with first antidepressant trial | STAR*D [11] |
| Recurrence Risk | 50% after 1 episode, 80% after 2, 90% after 3 | PMID: 10232312 [6] |
| First-Line Pharmacotherapy | SSRIs (sertraline, escitalopram) | NICE NG222 [5] |
| First-Line Psychotherapy | CBT, IPT, Behavioral Activation | APA Guidelines [12] |
| Gold Standard for TRD | Electroconvulsive Therapy (ECT) | Cochrane 2019 [13] |
| Screening Tool | PHQ-9 (Sensitivity 88%, Specificity 88%) | PMID: 11556941 [14] |
1.3 Clinical Pearls
Diagnostic Pearl: "The Core Dyad" Diagnosis requires at least one of: (1) depressed mood OR (2) anhedonia. Neither alone is sufficient without the other plus additional symptoms. Anhedonia—the inability to experience pleasure even from previously enjoyed activities—has higher specificity for MDD and predicts better response to biological treatments. [3]
Screening Pearl: "PHQ-2 then PHQ-9" The two-item PHQ-2 (depressed mood + anhedonia) has 97% sensitivity for detecting MDD. A positive screen (score ≥3) should trigger full PHQ-9 completion. This sequential approach optimizes clinical efficiency. [14]
Treatment Pearl: "Start Low, Go Slow, But Go" Initiate SSRIs at standard starting doses (sertraline 50mg, escitalopram 10mg). Therapeutic effect requires 2-4 weeks at adequate dose. If partial response at 4-6 weeks, titrate to maximum tolerated dose before switching. Premature switching is the most common prescribing error. [15]
Safety Pearl: "The Therapeutic Lag Paradox" Physical energy and motivation often improve before mood lifts (typically by week 1-2). This "therapeutic window" paradoxically increases suicide risk as patients regain capacity to act on persistent suicidal thoughts. Enhanced monitoring during weeks 2-4 is critical. [16]
Diagnostic Pitfall: "The Bipolar Trap" Always screen for prior manic/hypomanic episodes before initiating antidepressants. The Mood Disorder Questionnaire (MDQ) screens for bipolar disorder. SSRI monotherapy in undiagnosed bipolar disorder can trigger manic switch (10-15% risk) or rapid cycling, worsening long-term prognosis. [17]
Exam Mnemonic: "SIG E CAPS" (Prescribe Energy Capsules) Sleep disturbance, Interest decreased (anhedonia), Guilt/worthlessness, Energy decreased, Concentration impaired, Appetite change, Psychomotor change, Suicidal ideation. This covers the nine DSM-5 criteria efficiently for examinations. [1]
Elderly Pearl: "Depression vs. Dementia" In elderly patients presenting with cognitive complaints: Depression (pseudodementia) features rapid onset, "I don't know" responses, preserved attention to orientation, and subjective distress about deficits. Dementia features insidious onset, confabulation, disorientation, and lack of insight. Depression-related cognitive impairment is reversible with treatment. [18]
Severity Pearl: "The Melancholic Phenotype" Melancholic features (complete anhedonia, diurnal variation with morning worsening, early morning awakening, psychomotor disturbance, excessive guilt, weight loss) predict superior response to biological treatments (antidepressants, ECT) versus psychotherapy alone. [1]
1.4 Why This Matters Clinically
Patient Outcomes: Untreated depression leads to "kindling"—a neurobiological phenomenon where each episode lowers the threshold for subsequent episodes and increases treatment resistance. Early, adequate treatment prevents this progressive deterioration. Suicide is the 10th leading cause of death overall and the 2nd leading cause in ages 15-34. [7]
Healthcare Burden: MDD costs the global economy over $1 trillion annually in lost productivity and healthcare expenditure. In the UK, depression accounts for 70 million lost working days annually. The WHO predicts depression will become the leading cause of disability by 2030. [2]
Medico-Legal Considerations: Suicide risk assessment documentation is one of the most scrutinized areas in medical malpractice litigation. Every clinical encounter with a depressed patient requires documented assessment of suicidal ideation, intent, plan, access to means, and protective factors. Failure to assess or document appropriately creates significant liability exposure. [19]
Examination Relevance: MDD appears in every major medical examination (MRCP, USMLE, PLAB, MRCPsych). High-yield areas include DSM-5 criteria, PHQ-9 interpretation, SSRI pharmacology, serotonin syndrome recognition, suicide risk stratification, and ECT indications.
SECTION 2: Epidemiology
2.1 Incidence and Prevalence
Global Epidemiology:
- Point Prevalence: 4.4% globally (322 million people affected) [2]
- 12-Month Prevalence: 7.1% US adults, 6.9% European adults [8]
- Lifetime Prevalence: 16.6% (US), 10-20% internationally [9]
- Incidence: Annual incidence 3.0% in women, 1.8% in men [10]
Temporal Trends:
- 18.4% increase in global prevalence from 2005-2015 [2]
- COVID-19 pandemic associated with 25% increase in depression prevalence globally [20]
- Concerning increase in adolescent and young adult depression over past 15 years
Geographic Variation:
- Higher reported rates in high-income countries (likely reflecting diagnostic access)
- Highest disease burden in low-middle income countries (due to treatment gap)
- Seasonal Affective Disorder (SAD) shows winter peak at northern latitudes
Healthcare Burden:
- 2nd leading cause of years lived with disability (YLDs) globally [2]
- Accounts for 10% of all non-fatal disability burden
- 50% of patients with MDD never receive treatment (treatment gap) [2]
2.2 Demographics Table
| Factor | Details | Clinical Significance | Evidence |
|---|---|---|---|
| Age | Peak: 18-29 years; bimodal with second peak 40-50 years | Early onset predicts more chronic course and higher recurrence | DSM-5 [1] |
| Sex | Female:Male = 2:1; ratio narrows after age 65 | Hormonal transitions (puberty, postpartum, perimenopause) are high-risk periods | [9] |
| Ethnicity | Similar prevalence across groups; higher chronicity in minorities | Disparities in access to culturally competent care; lower treatment rates | [9] |
| Marital Status | Higher in separated/divorced; lowest in married | Social support is protective; relationship conflict is risk factor | [9] |
| Urban/Rural | Higher in urban environments | Social isolation, noise, pollution, pace of life as stressors | [10] |
| Socioeconomic | Inverse correlation with income and education | Poverty as chronic psychosocial stressor; reduced treatment access | [9] |
| Employment | Higher in unemployed; high-stress/low-control jobs at risk | Bidirectional relationship: depression causes and results from unemployment | [10] |
2.3 Risk Factor Tables
Non-Modifiable Risk Factors:
| Risk Factor | Relative Risk (95% CI) | Mechanism | Evidence Level |
|---|---|---|---|
| Family history (1st degree relative) | RR 2.8 (2.3-3.4) | Polygenic inheritance; heritability 35-40% | Level 1a [3] |
| Female sex | RR 1.9 (1.7-2.2) | Estrogen/progesterone effects on 5-HT system; psychosocial factors | Level 1a [9] |
| Prior depressive episode | RR 4.0 (3.2-5.1) | Neurobiological kindling; residual symptoms | Level 1a [6] |
| Early childhood adversity | RR 2.5 (1.9-3.2) | Epigenetic changes to HPA axis regulation | Level 1a [3] |
| Chronic medical illness | RR 2.0-3.0 | Inflammatory mechanisms; shared pathophysiology | Level 1a [21] |
| Personality traits (neuroticism) | RR 2.0 (1.7-2.4) | Cognitive vulnerability; stress sensitivity | Level 2a [3] |
Modifiable Risk Factors:
| Risk Factor | Relative Risk (95% CI) | Evidence Level | Intervention Impact |
|---|---|---|---|
| Substance use disorder | RR 2.1 (1.8-2.5) | Level 1a | Sobriety improves treatment response by 50% |
| Physical inactivity | RR 1.4 (1.2-1.6) | Level 1b | Regular exercise equivalent to SSRI in mild MDD |
| Poor social support | RR 2.3 (1.9-2.8) | Level 2a | Social prescribing reduces relapse rates |
| Obesity (BMI > 30) | RR 1.5 (1.3-1.8) | Level 1a | Weight loss reduces inflammatory markers |
| Chronic insomnia | RR 2.1 (1.7-2.6) | Level 1b | CBT-I reduces depression incidence by 50% |
| Stressful life events | RR 2.5 (2.0-3.1) | Level 1a | Stress management and coping skills training |
| Unemployment | RR 1.8 (1.5-2.2) | Level 2a | Vocational rehabilitation as adjunct therapy |
2.4 Protective Factors
| Protective Factor | Mechanism | Evidence |
|---|---|---|
| Strong social connections | Buffering effect against stress; oxytocin release | Level 1b |
| Regular physical activity | BDNF upregulation; anti-inflammatory effects | Level 1a |
| Adequate sleep (7-9 hours) | Circadian rhythm regulation; emotional processing | Level 1b |
| Purpose and meaning | Psychological resilience; reduced rumination | Level 2b |
| Religious/spiritual involvement | Community support; coping mechanisms | Level 2b |
| High emotional intelligence | Emotional regulation; stress coping | Level 2b |
| Mediterranean diet | Anti-inflammatory; gut-brain axis effects | Level 1b [22] |
SECTION 3: Pathophysiology
3.1 Etiology Overview: The Biopsychosocial Model
MDD arises from complex interactions between biological vulnerability, psychological factors, and social/environmental stressors. No single etiological factor is sufficient; rather, MDD represents a "final common pathway" of multiple converging mechanisms. [3]
Genetic Factors (35-40% heritability):
- Twin studies demonstrate concordance of 37% (monozygotic) vs. 18% (dizygotic)
- Genome-wide association studies identify > 100 risk loci, each with small effect
- Key genes implicated: SLC6A4 (serotonin transporter), BDNF, FKBP5, CRHR1
- Gene-environment interactions crucial: 5-HTTLPR polymorphism moderates impact of early adversity
Epigenetic Mechanisms:
- Childhood adversity produces lasting epigenetic modifications
- Hypermethylation of glucocorticoid receptor (NR3C1) gene promoter
- Altered stress response persists into adulthood
- These changes are potentially reversible with treatment
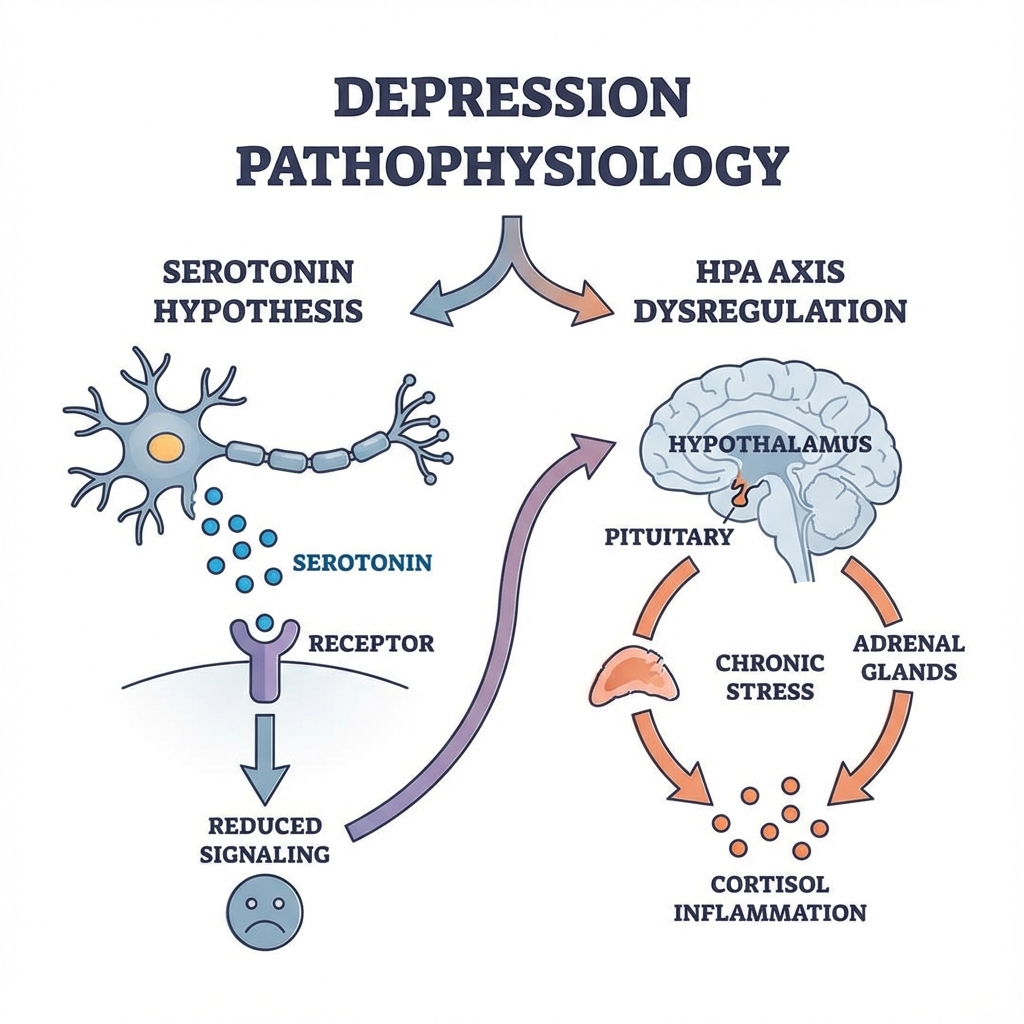
3.2 The Hypothalamic-Pituitary-Adrenal (HPA) Axis Dysregulation
The HPA axis is the body's central stress response system, and its dysregulation is one of the most replicated findings in MDD. [3]
Normal Stress Response:
- Stressor perceived → Amygdala activation
- Hypothalamus releases Corticotropin-Releasing Hormone (CRH)
- CRH stimulates anterior pituitary to release ACTH
- ACTH stimulates adrenal cortex to release cortisol
- Cortisol provides negative feedback to hypothalamus/pituitary (terminating response)
Pathological Changes in MDD:
- Glucocorticoid Receptor Resistance: Chronic stress leads to downregulation of GR in hippocampus and hypothalamus
- Failed Negative Feedback: Elevated cortisol fails to suppress CRH/ACTH release
- Chronic Hypercortisolemia: Sustained cortisol elevation is neurotoxic
- Dexamethasone Suppression Test: 50% of melancholic patients fail to suppress cortisol following dexamethasone
Clinical Correlates:
- Hypercortisolemia correlates with depression severity
- Explains insomnia (cortisol disrupts sleep architecture)
- Explains hippocampal atrophy (cortisol is neurotoxic to hippocampal neurons)
- Normalization of HPA axis predicts sustained remission
3.3 The Monoamine Hypothesis
The monoamine hypothesis remains foundational, though now understood as reflecting downstream consequences rather than primary causation. [3]
Core Premise:
- Depression results from functional deficiency of monoamine neurotransmitters: serotonin (5-HT), norepinephrine (NE), and dopamine (DA)
- Antidepressants increase monoamine availability → clinical improvement
Evidence Supporting:
- Reserpine (depletes monoamines) → causes depression
- Monoamine oxidase inhibitors (prevent monoamine breakdown) → antidepressant
- SSRIs/SNRIs (block reuptake) → antidepressant
- Tryptophan depletion → transient mood lowering in remitted patients
Specific Neurotransmitter Roles:
| Neurotransmitter | Key Pathways | Clinical Symptoms When Deficient |
|---|---|---|
| Serotonin (5-HT) | Raphe nuclei → limbic system, cortex | Anxiety, obsessionality, irritability, suicidality |
| Norepinephrine (NE) | Locus coeruleus → cortex, limbic system | Fatigue, psychomotor retardation, concentration deficits |
| Dopamine (DA) | VTA → nucleus accumbens (mesolimbic) | Anhedonia, amotivation, psychomotor retardation |
Limitations of Monoamine Hypothesis:
- Monoamine levels increase within hours; clinical effect takes weeks
- 30-40% of patients do not respond to monoaminergic drugs
- No consistent evidence of baseline monoamine deficiency in depressed patients
3.4 The Neurotrophic Hypothesis
Modern understanding emphasizes neuroplasticity and neurotrophic factor dysregulation as central to MDD pathophysiology. [3]
Brain-Derived Neurotrophic Factor (BDNF):
- BDNF is essential for neuronal survival, dendritic arborization, synaptic plasticity, and neurogenesis
- Serum BDNF levels are reduced in MDD and correlate with depression severity
- Postmortem studies show reduced BDNF in hippocampus and PFC of depressed individuals
Mechanism of BDNF Reduction:
- Chronic stress → elevated cortisol
- Cortisol suppresses BDNF gene transcription
- Reduced BDNF → dendritic spine retraction, reduced synaptogenesis
- Hippocampal and prefrontal volume reduction
- Impaired emotional regulation and cognitive function
Neuroplasticity Effects:
- Hippocampal volume reduction (10-15% in recurrent MDD)
- Prefrontal cortical thinning
- Reduced synaptic density
- These changes are partially reversible with treatment (antidepressants increase BDNF)
Therapeutic Implications:
- Antidepressants increase BDNF expression (explains therapeutic lag)
- Ketamine rapidly increases BDNF and synaptogenesis
- Exercise is a potent BDNF inducer
3.5 Neuroinflammation and Immune Dysregulation
Depression is increasingly understood as a systemic inflammatory condition with prominent central immune activation. [21]
Evidence for Inflammation:
- Elevated pro-inflammatory cytokines (IL-1β, IL-6, TNF-α, CRP) in 30-50% of MDD patients
- Meta-analyses confirm effect sizes of d=0.5-0.7 for cytokine elevations
- Anti-inflammatory treatments show antidepressant effects in trials
- Medical conditions with high inflammation (autoimmune, cardiovascular) have high depression comorbidity
Mechanisms of Inflammation-Induced Depression:
-
Kynurenine Pathway Activation:
- Inflammatory cytokines activate indoleamine 2,3-dioxygenase (IDO)
- Tryptophan shunted from serotonin synthesis toward kynurenine
- Results in: (a) reduced serotonin, (b) increased quinolinic acid (neurotoxic NMDA agonist)
-
Microglial Activation:
- Microglia shift from surveying to pro-inflammatory phenotype
- Release cytokines, reactive oxygen species
- Impair synaptic pruning and neuroplasticity
-
Blood-Brain Barrier Disruption:
- Peripheral cytokines access CNS
- Activates central inflammatory cascades
Clinical Correlates:
- Treatment-resistant depression associated with higher inflammatory markers
- Anti-inflammatory augmentation may benefit inflammatory subtype
- Exercise and Mediterranean diet reduce inflammation
3.6 Neural Circuit Dysregulation
Functional neuroimaging reveals disrupted connectivity in specific neural networks in MDD. [3]
Key Networks Affected:
| Network | Normal Function | Abnormality in MDD | Clinical Manifestation |
|---|---|---|---|
| Default Mode Network (DMN) | Self-referential thought, memory | Hyperconnectivity, overactivity | Rumination, negative self-focus |
| Central Executive Network (CEN) | Working memory, goal-directed behavior | Hypoconnectivity, reduced activity | Poor concentration, indecision |
| Salience Network (SN) | Detecting and filtering salient stimuli | Altered function | Negative attentional bias |
| Reward Network | Processing pleasure, motivation | Reduced activity in nucleus accumbens | Anhedonia, amotivation |
Specific Regional Abnormalities:
- Prefrontal Cortex: Reduced activity in dorsolateral PFC (executive function)
- Amygdala: Hyperactive in response to negative stimuli; persistent after stimulus offset
- Hippocampus: Volume reduction; impaired contextual memory and emotional regulation
- Anterior Cingulate Cortex: Altered activity; involved in emotional processing and pain
3.7 Classification and Staging
DSM-5 Severity Specifiers:
| Severity | Criteria | Functional Impact | Treatment Implications |
|---|---|---|---|
| Mild | 5-6 symptoms; minor functional impairment | Works with effort; social life maintained | Active monitoring, low-intensity intervention |
| Moderate | Symptoms and impairment between mild and severe | Significant work/social difficulty | Pharmacotherapy and/or psychotherapy |
| Severe | Most symptoms; marked functional impairment | Unable to work/socialize; may not self-care | Combined treatment; consider hospitalization |
DSM-5 Specifiers:
| Specifier | Key Features | Clinical Significance |
|---|---|---|
| With Anxious Distress | Tension, restlessness, worry, fear of loss of control | Poorer treatment response; higher suicide risk |
| With Mixed Features | ≥3 manic symptoms (but not meeting mania criteria) | Consider bipolar spectrum; cautious with AD |
| With Melancholic Features | Complete anhedonia, diurnal variation, psychomotor disturbance | Better response to biological treatments |
| With Atypical Features | Mood reactivity, hypersomnia, hyperphagia, rejection sensitivity | MAOIs may be superior; SSRIs effective |
| With Psychotic Features | Delusions and/or hallucinations (typically mood-congruent) | Requires antipsychotic + antidepressant or ECT |
| With Catatonia | Immobility, mutism, negativism, posturing | Benzodiazepines first-line; consider ECT |
| With Peripartum Onset | Onset during pregnancy or within 4 weeks postpartum | Screen for infanticide risk; breastfeeding considerations |
| With Seasonal Pattern | Regular temporal relationship (typically winter) | Light therapy effective |
Course Specifiers:
- Single Episode: First lifetime episode
- Recurrent: ≥2 episodes with ≥2 months remission between
- In Partial Remission: Residual symptoms; full criteria not met
- In Full Remission: No significant symptoms for ≥2 months
SECTION 4: Clinical Presentation
4.1 DSM-5 Diagnostic Criteria
Criterion A: Five or more of the following symptoms present during the same 2-week period, representing a change from previous functioning. At least one symptom is either (1) depressed mood or (2) loss of interest or pleasure.
| Symptom | DSM-5 Description | Clinical Features |
|---|---|---|
| 1. Depressed Mood | Most of the day, nearly every day; subjective or observed | "Sad," "empty," "hopeless," "low"; may be irritable in children/adolescents |
| 2. Anhedonia | Markedly diminished interest or pleasure in all or almost all activities | Loss of enjoyment in hobbies, sex, food, social activities |
| 3. Weight/Appetite Change | Significant weight loss/gain (> 5% in month) or appetite change | Typically decreased in melancholic; increased in atypical |
| 4. Sleep Disturbance | Insomnia or hypersomnia nearly every day | Insomnia common; early morning awakening in melancholic |
| 5. Psychomotor Change | Agitation or retardation (observable, not subjective) | Slowed speech/movement OR restlessness, pacing |
| 6. Fatigue | Fatigue or loss of energy nearly every day | "Leaden paralysis," everything feels effortful |
| 7. Worthlessness/Guilt | Feelings of worthlessness or excessive/inappropriate guilt | May be delusional in severe cases |
| 8. Concentration | Diminished ability to think or concentrate, or indecisiveness | "Brain fog," difficulty making decisions |
| 9. Suicidal Ideation | Recurrent thoughts of death, suicidal ideation, attempt, or plan | Range from passive ("better off dead") to active (specific plan) |
Criterion B: Symptoms cause clinically significant distress or impairment in social, occupational, or other important areas of functioning.
Criterion C: Episode not attributable to physiological effects of a substance or another medical condition.
Criterion D: Not better explained by schizoaffective disorder, schizophrenia, schizophreniform disorder, delusional disorder, or other specified/unspecified schizophrenia spectrum disorders.
Criterion E: There has never been a manic or hypomanic episode (unless substance-induced or attributable to another medical condition).
4.2 Symptom Details
| Symptom | Frequency | Characteristics | Associations | Severity Indicator |
|---|---|---|---|---|
| Depressed mood | 95% | Pervasive, persistent, qualitatively different from normal sadness | Diurnal variation (worse AM in melancholic) | Duration and pervasiveness |
| Anhedonia | 85-90% | Complete inability to experience pleasure | Social withdrawal, loss of libido | Completeness (partial vs total) |
| Sleep disturbance | 80-85% | Initial/middle/terminal insomnia or hypersomnia | Fatigue, concentration impairment | Terminal insomnia = melancholic |
| Fatigue/anergia | 80-85% | "Leaden paralysis," exhaustion without exertion | Reduced self-care, work impairment | Inability to get out of bed |
| Concentration impairment | 70-75% | "Brain fog," difficulty reading, watching TV | Work errors, accidents | Pseudodementia in elderly |
| Appetite/weight change | 65-70% | Decreased or increased (atypical); comfort eating | > 5% weight change in month | Refusal to eat = emergency |
| Worthlessness/guilt | 60-65% | Excessive rumination on perceived failures | May be delusional | Guilt over trivial matters |
| Psychomotor change | 50-55% | Observable slowing OR agitation | Melancholic feature if marked | Catatonia if extreme |
| Suicidal ideation | 50-60% | Passive thoughts → active ideation → plan → intent | Prior attempts, hopelessness | Plan + intent = emergency |
4.3 Signs on Examination
| Sign | How to Elicit | Finding in MDD | Sensitivity | Specificity |
|---|---|---|---|---|
| Psychomotor retardation | Observe gait, spontaneous movement, response latency | Slowed movements, delayed responses (> 3 seconds) | 40% | 90% |
| Psychomotor agitation | Observe for restlessness | Hand-wringing, pacing, inability to sit still | 20% | 85% |
| Reduced eye contact | Clinical observation | Avoids gaze, downcast eyes | 60% | 50% |
| Flattened affect | Observe emotional range during interview | Restricted or absent affective response | 70% | 60% |
| Omega sign (Veraguth fold) | Observe forehead | Furrowing creating inverted omega shape | 20% | 95% |
| Reduced speech | Listen to spontaneous speech | Low volume, monotone, long pauses | 55% | 75% |
| Self-neglect | Observe grooming, hygiene | Unkempt, unwashed, unchanged clothes | 40% | 70% |
| Weight change | Serial weights, observe for loose clothing | Loss > 5% body weight in month | 50% | 80% |
| Self-harm scars | Inspect arms, thighs | Healed or fresh lacerations | N/A | N/A |
4.4 Atypical and Special Presentations
Atypical Depression (DSM-5 specifier):
- Mood reactivity (can be cheered up temporarily)
- Hypersomnia (> 10 hours/day)
- Hyperphagia/weight gain
- Leaden paralysis
- Interpersonal rejection sensitivity
Masked/Somatized Depression:
- Primary presentation with physical symptoms
- Chronic pain, fatigue, GI symptoms, headache
- Denial of psychological symptoms
- More common in elderly, certain cultures
Psychotic Depression:
- Mood-congruent delusions (guilt, poverty, nihilism, somatic)
- Cotard syndrome (belief that one is dead or organs are rotting)
- Auditory hallucinations (derogatory voices)
- Requires antipsychotic + antidepressant OR ECT
Depression with Anxious Distress:
- Feeling keyed up or tense
- Feeling unusually restless
- Difficulty concentrating due to worry
- Fear that something awful may happen
- Feeling of loss of control
- Poorer treatment response; higher suicide risk
4.5 Red Flags
Suicidal Emergency:
- Active suicidal ideation with specific plan
- Intent to act on suicidal thoughts
- Access to lethal means (firearms, medications, heights)
- Recent suicide attempt or significant self-harm
- Expressing goodbyes, giving away possessions
Psychotic Emergency:
- Command hallucinations to harm self or others
- Persecutory delusions leading to defensive violence
- Delusions of guilt requiring "punishment"
- Cotard syndrome (nihilistic delusions)
Physical Emergency:
- Catatonic features (immobility, mutism, posturing)
- Complete refusal of food/fluids > 48 hours
- Severe dehydration or electrolyte disturbance
- Self-neglect with medical complications
High-Risk Transitions:
- Recent psychiatric discharge (first 30 days highest risk)
- Sudden unexplained improvement (may indicate decision made)
- Agitation after starting antidepressant (akathisia)
SECTION 5: Suicide Risk Assessment
5.1 Epidemiology of Suicide in MDD
- Lifetime suicide mortality in MDD: 4-7% (hospitalized patients); 2-4% (outpatients) [7]
- Suicide attempts: 10-15% of MDD patients will attempt suicide
- Suicidal ideation: Present in 50-60% of MDD patients at some point
- Completed suicide ratio: 25:1 attempts to completions (overall); 3:1 in elderly men
5.2 Risk Stratification Framework
Acute Risk Factors (Modifiable):
| Factor | Description | Intervention |
|---|---|---|
| Suicidal ideation with plan | Specific method, time, place identified | Hospitalization |
| Access to lethal means | Firearms, stockpiled medications, heights | Means restriction |
| Acute intoxication | Alcohol/substance use lowers inhibition | Medical stabilization |
| Severe hopelessness | "Nothing will ever improve" | Intensive treatment |
| Acute psychotic symptoms | Command hallucinations, delusions | Antipsychotic treatment |
| Recent discharge | First 7-30 days post-hospitalization | Enhanced follow-up |
| Agitation/akathisia | Extreme restlessness, medication-induced | Medication adjustment |
Chronic Risk Factors (Static):
| Factor | Relative Risk | Notes |
|---|---|---|
| Prior suicide attempt | 40x | Single strongest predictor |
| Family history of suicide | 4-6x | Genetic and learned behavior |
| Male sex | 3-4x | Higher completion rate despite lower attempt rate |
| Older age (> 65) | 2-3x | Higher lethality; less warning |
| Chronic medical illness | 2-3x | Especially pain, terminal illness, disability |
| Substance use disorder | 6x | Comorbidity dramatically increases risk |
| Personality disorder (BPD) | 4x | Impulsivity, emotional dysregulation |
| LGBTQ+ identity | 2-4x | Minority stress, discrimination |
Protective Factors:
- Reasons for living (children, responsibilities)
- Religious/cultural beliefs against suicide
- Strong social support
- Therapeutic alliance
- Limited access to means
- Fear of death/pain
- Future orientation (plans, goals)
5.3 Structured Assessment Tools
Columbia Suicide Severity Rating Scale (C-SSRS):
- Gold standard for suicide assessment in research and clinical practice
- Assesses ideation severity, intensity, and behavior
- Categorizes ideation intensity (1-5 scale)
- Documents specific suicide attempts with details
Questions to Ask:
- "Have you had thoughts of being better off dead, or of hurting yourself?"
- "Have you actually had thoughts of killing yourself?"
- "Have you thought about how you might do this?"
- "Have you had any intention of acting on these thoughts?"
- "Have you done anything to prepare?" (Obtained means, written note, said goodbyes)
PHQ-9 Item 9:
- "Thoughts that you would be better off dead, or of hurting yourself"
- Any positive response requires further assessment
- Score of 1+ triggers clinical risk assessment
5.4 Documentation Requirements
Every encounter with a depressed patient should document:
| Element | Content | Purpose |
|---|---|---|
| Ideation | Presence, frequency, intensity of suicidal thoughts | Baseline and monitoring |
| Intent | Degree of desire to act on thoughts | Risk level determination |
| Plan | Specific method, time, location | Immediacy of risk |
| Access to means | Availability of firearms, medications, etc. | Means restriction planning |
| Protective factors | Reasons for living, supports, beliefs | Treatment leverage |
| Risk level assessment | Low, moderate, high, imminent | Guides disposition |
| Safety plan | Coping strategies, contacts, crisis resources | Intervention |
| Clinical decision | Rationale for outpatient vs. hospitalization | Medico-legal protection |
5.5 Safety Planning
Stanley-Brown Safety Planning Intervention:
- Warning signs: Personal triggers and symptoms of crisis
- Internal coping strategies: What patient can do alone to distract/calm
- Social contacts for distraction: People and places that provide positive distraction
- People to ask for help: Specific individuals who can provide support in crisis
- Professional resources: Therapist, psychiatrist, crisis line, ED
- Means restriction: Specific plan to reduce access to lethal means
SECTION 6: Differential Diagnosis
6.1 Psychiatric Differential Diagnosis
| Condition | Key Distinguishing Features | Investigation |
|---|---|---|
| Bipolar I/II Disorder | History of manic or hypomanic episodes | MDQ, collateral history, mood chart |
| Persistent Depressive Disorder | Chronic (> 2 years) but less severe symptoms | Timeline, severity assessment |
| Adjustment Disorder | Clear stressor, symptoms less than 6 months after resolution | Timeline, stressor identification |
| Anxiety Disorders | Anxiety primary, depression secondary | Symptom onset order, predominance |
| PTSD | Trauma history, re-experiencing, avoidance | Trauma screening (PC-PTSD-5) |
| Grief/Bereavement | Recent loss, focus on deceased, waves of sadness | Timeline, content of thoughts |
| Schizoaffective Disorder | Psychosis persists outside mood episodes | Longitudinal history |
| Personality Disorders | Chronic patterns, interpersonal dysfunction | Longitudinal history, PDQ-4 |
| Substance Use Disorder | Symptoms during intoxication/withdrawal | Toxicology, drinking history |
6.2 Medical Differential Diagnosis
| Condition | Clinical Clues | Investigation |
|---|---|---|
| Hypothyroidism | Weight gain, cold intolerance, constipation | TSH, free T4 |
| Hyperthyroidism | Weight loss, tremor, palpitations, anxiety | TSH, free T4, T3 |
| Vitamin B12 deficiency | Macrocytic anemia, neuropathy, elderly | Serum B12, MMA |
| Folate deficiency | Macrocytic anemia, malnutrition | Serum/RBC folate |
| Vitamin D deficiency | Fatigue, bone pain, seasonality | 25-OH Vitamin D |
| Anemia | Fatigue, pallor, dyspnea | CBC, iron studies |
| Stroke | Sudden onset, focal neurology, post-stroke | CT/MRI brain |
| Parkinson's disease | Bradykinesia, rigidity, tremor | Clinical exam, DaTscan |
| Multiple sclerosis | Neurological symptoms, young onset | MRI brain/spine, LP |
| Traumatic brain injury | History of head trauma, personality change | CT/MRI, neuropsych |
| Sleep apnea | Snoring, witnessed apneas, daytime sleepiness | Polysomnography |
| Chronic fatigue syndrome | Post-exertional malaise, unrefreshing sleep | Clinical criteria |
| Cushing's syndrome | Truncal obesity, striae, easy bruising | 24h urine cortisol, DST |
| Addison's disease | Fatigue, hypotension, hyperpigmentation | AM cortisol, ACTH stim |
| Hypercalcemia | Polyuria, constipation, confusion | Serum calcium, PTH |
| HIV/AIDS | Risk factors, immunosuppression | HIV serology |
| Neurosyphilis | Psychiatric symptoms, neurological signs | RPR/VDRL, LP |
| Pancreatic cancer | New-onset depression, weight loss, older age | CT abdomen, CA19-9 |
6.3 Medication-Induced Depression
| Drug Class | Examples | Mechanism |
|---|---|---|
| Corticosteroids | Prednisone, dexamethasone | HPA axis dysregulation |
| Beta-blockers | Propranolol (lipophilic) | CNS effects, fatigue |
| Interferon-alpha | Hepatitis C treatment | Cytokine-induced depression |
| Isotretinoin | Acne treatment | Mechanism unclear |
| Hormonal contraceptives | Combined pill, progestins | Hormone modulation |
| Antiepileptics | Levetiracetam, phenobarbital | CNS depression |
| Opioids | Chronic use | Reward pathway dysregulation |
| Benzodiazepines | Chronic use | CNS depression |
| Antipsychotics | Haloperidol, risperidone | Dopamine blockade |
| Antihistamines | Diphenhydramine | CNS effects |
SECTION 7: Investigations
7.1 Routine Baseline Investigations
| Test | Rationale | Expected Finding | Urgency |
|---|---|---|---|
| TSH | Rule out thyroid dysfunction | Normal (0.4-4.0 mIU/L) | Routine |
| Free T4 | If TSH abnormal | Normal | Routine |
| CBC | Anemia, infection | Normal | Routine |
| CMP (U&Es, glucose) | Baseline, SIADH risk with SSRIs | Normal | Routine |
| Vitamin B12 | Deficiency mimics depression | > 300 pg/mL | Routine |
| Folate | Deficiency, MTHFR considerations | Normal | Routine |
| Vitamin D (25-OH) | Deficiency associated with depression | > 30 ng/mL optimal | Routine |
| LFTs | Baseline before medication | Normal | Routine |
| HbA1c | Metabolic syndrome screening | less than 5.7% normal | Routine |
| Lipid panel | Metabolic syndrome, atypical antipsychotic baseline | — | Routine |
7.2 Targeted Investigations
| Test | Indication | Rationale |
|---|---|---|
| Urine drug screen | Suspected substance use, young patients | Rule out substance-induced depression |
| HIV serology | Risk factors present | HIV-associated depression |
| Syphilis serology | Risk factors, neurological symptoms | Neurosyphilis |
| Cortisol (AM) | Clinical features of Cushing's or Addison's | Adrenal pathology |
| Calcium/PTH | Symptoms suggestive of hypercalcemia | Hypercalcemia causes psychiatric symptoms |
| ESR/CRP | Inflammatory symptoms, treatment-resistance | Inflammatory subtype |
| ANA | Systemic symptoms, young women | SLE, autoimmune causes |
7.3 Neuroimaging
| Modality | Indication | Expected Findings |
|---|---|---|
| MRI Brain | First episode > 60 years, atypical features, cognitive symptoms, focal neurology | Rule out structural pathology; may show hippocampal atrophy |
| CT Head | Acute onset, trauma, neurological signs | Rule out mass, hemorrhage, stroke |
| EEG | Atypical symptoms, paroxysmal features, suspected seizures | Rule out temporal lobe epilepsy |
7.4 Pre-Treatment ECG
Indications:
- Baseline before starting antidepressant with QTc risk (citalopram, escitalopram > 20mg, TCAs)
- Personal or family history of cardiac disease
- Age > 65 years
- Concurrent QTc-prolonging medications
QTc Thresholds:
- Normal: less than 450ms (men), less than 460ms (women)
- Borderline: 450-470ms (men), 460-480ms (women)
- Prolonged: > 470ms (men), > 480ms (women)
- Avoid citalopram/escitalopram if QTc > 500ms
SECTION 8: Screening and Assessment Instruments
8.1 Patient Health Questionnaire-9 (PHQ-9)
Description: 9-item self-report questionnaire based on DSM-IV criteria, validated for DSM-5. Gold standard for depression screening in primary care. [14]
Scoring:
- Each item scored 0-3 (Not at all → Nearly every day)
- Total score range: 0-27
| Score | Severity | Provisional Diagnosis | Treatment Recommendation |
|---|---|---|---|
| 0-4 | Minimal | None | None |
| 5-9 | Mild | Minor depression | Watchful waiting, psychoeducation |
| 10-14 | Moderate | Major depression (moderate) | Antidepressant or psychotherapy |
| 15-19 | Moderately Severe | Major depression | Combined treatment |
| 20-27 | Severe | Major depression (severe) | Pharmacotherapy + psychotherapy; consider referral |
Psychometric Properties:
- Sensitivity: 88% (at cutoff ≥10)
- Specificity: 88% (at cutoff ≥10)
- Positive Predictive Value: 57%
- Negative Predictive Value: 98%
Item 9 (Suicidal Ideation):
- Any score ≥1 requires clinical risk assessment
- "Thoughts that you would be better off dead, or of hurting yourself in some way"
8.2 PHQ-2 Screening
Items:
- "Little interest or pleasure in doing things"
- "Feeling down, depressed, or hopeless"
Scoring: 0-6 total; score ≥3 is positive screen (triggers full PHQ-9)
Performance:
- Sensitivity: 97% for MDD
- Specificity: 67%
- Recommended as first-step screening in all patients
8.3 Hamilton Depression Rating Scale (HAM-D/HDRS)
Description: 17-item clinician-administered scale. Gold standard for clinical trials.
Scoring:
- 0-7: Normal
- 8-13: Mild depression
- 14-18: Moderate depression
- 19-22: Severe depression
- ≥23: Very severe depression
Use: Research, monitoring treatment response in specialty settings. 50% reduction in HAM-D score = treatment response.
8.4 Other Validated Instruments
| Instrument | Items | Use | Population |
|---|---|---|---|
| Beck Depression Inventory (BDI-II) | 21 | Self-report; therapy monitoring | General adult |
| Geriatric Depression Scale (GDS) | 15 | Screening in elderly | Age ≥65 years |
| Edinburgh Postnatal Depression Scale (EPDS) | 10 | Perinatal screening | Pregnant/postpartum women |
| Montgomery-Åsberg Depression Rating Scale (MADRS) | 10 | Clinician-rated; sensitive to change | Clinical trials |
| Quick Inventory of Depressive Symptoms (QIDS) | 16 | Self or clinician-rated | General adult |
| Mood Disorder Questionnaire (MDQ) | 13 | Screening for bipolar disorder | Rule out bipolar |
SECTION 9: Management
9.1 Principles of Treatment
Goals of Treatment:
- Response: ≥50% reduction in symptom severity
- Remission: Minimal or no residual symptoms (PHQ-9 less than 5, HAM-D less than 7)
- Recovery: Sustained remission ≥4-6 months
- Prevention of relapse: Maintain gains during continuation phase
- Prevention of recurrence: Long-term prevention in recurrent MDD
Treatment Phases:
| Phase | Duration | Goal | Medication Strategy |
|---|---|---|---|
| Acute | 8-12 weeks | Achieve remission | Initiate and optimize dose |
| Continuation | 4-9 months | Prevent relapse | Maintain effective dose |
| Maintenance | ≥1 year to lifelong | Prevent recurrence | Continue indefinitely in recurrent MDD |
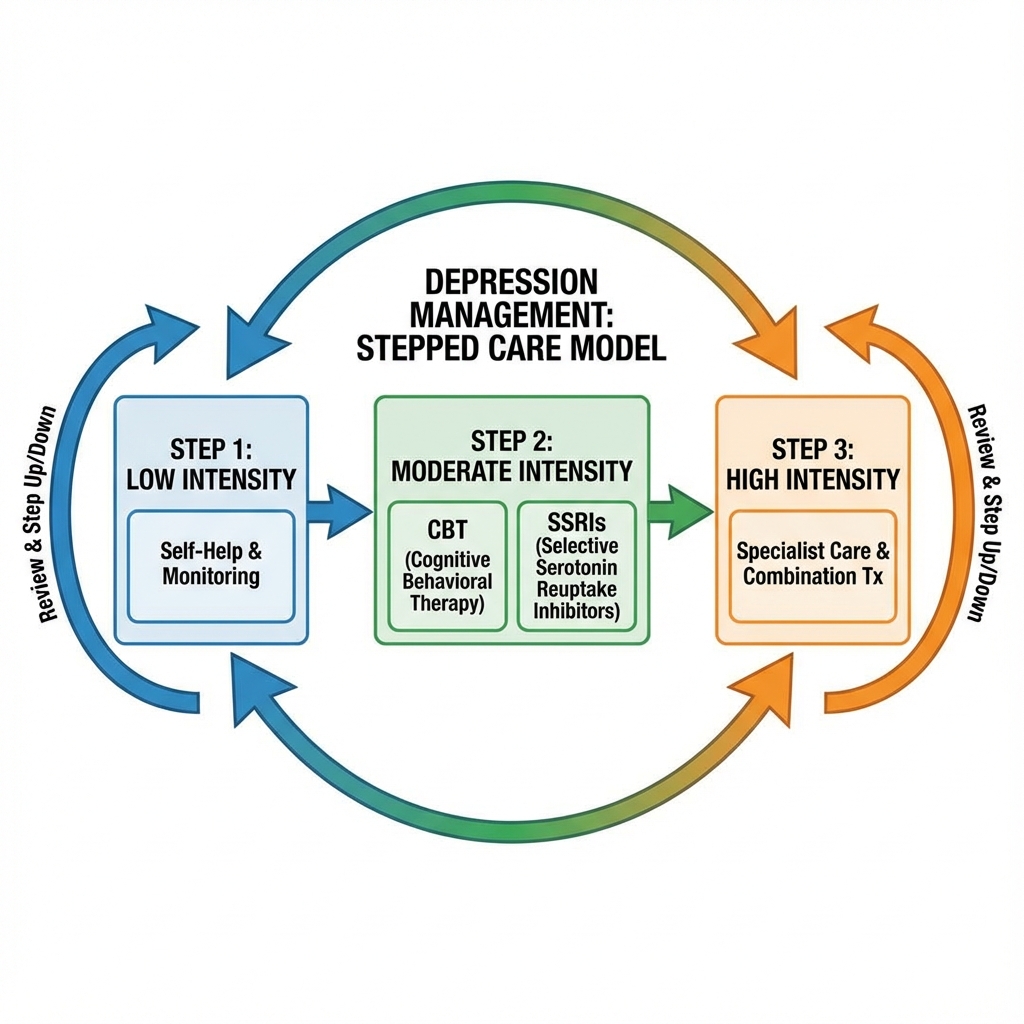
9.2 Stepped Care Algorithm
┌─────────────────────────────────────────────────────────────────────────┐
│ ADULT DEPRESSION MANAGEMENT ALGORITHM │
│ (NICE NG222 / APA Based) │
└─────────────────────────────────────────────────────────────────────────┘
│
▼
┌─────────────────────────────────┐
│ INITIAL ASSESSMENT │
│ • Confirm diagnosis (DSM-5) │
│ • PHQ-9 score + severity │
│ • SUICIDE RISK ASSESSMENT │
│ • Rule out bipolar (MDQ) │
│ • Rule out medical causes │
│ • Assess functional impairment │
└─────────────────────────────────┘
│
┌─────────────────┼─────────────────┐
▼ ▼ ▼
┌─────────────────┐ ┌─────────────────┐ ┌─────────────────┐
│ MILD (PHQ 5-9) │ │ MODERATE (10-19)│ │ SEVERE (≥20) │
│ │ │ │ │ OR PSYCHOTIC │
└────────┬────────┘ └────────┬────────┘ └────────┬────────┘
│ │ │
▼ ▼ ▼
┌─────────────────┐ ┌─────────────────┐ ┌─────────────────┐
│ STEP 1: │ │ STEP 2: │ │ STEP 3: │
│ • Active │ │ • SSRI + HIGH- │ │ • Urgent Psych │
│ monitoring │ │ INTENSITY │ │ referral │
│ • Psychoeducat- │ │ PSYCHOTHERAPY │ │ • Consider │
│ ion │ │ (CBT or IPT) │ │ hospitalizat- │
│ • Guided self- │ │ • OR Antidepres-│ │ ion │
│ help │ │ sant alone │ │ • Pharmacother- │
│ • Exercise │ │ (shared │ │ apy + therapy │
│ • Behavioral │ │ decision) │ │ • +Antipsychot- │
│ activation │ │ │ │ ic if │
│ • Review 2-4 │ │ │ │ psychotic │
│ weeks │ │ │ │ │
└────────┬────────┘ └────────┬────────┘ └────────┬────────┘
│ │ │
┌────────▼────────┐ ┌────────▼────────┐ ┌────────▼────────┐
│ RESPONSE? │ │ RESPONSE BY │ │ RESPONSE? │
│ (2-4 weeks) │ │ 4-6 WEEKS? │ │ (4-6 weeks) │
└────────┬────────┘ └────────┬────────┘ └────────┬────────┘
Yes │ No Yes │ No Yes │ No
│ │ │
▼ ▼ ▼
┌─────────────────┐ ┌─────────────────┐ ┌─────────────────┐
│ Continue or │ │ Optimize: │ │ TREATMENT │
│ graduate to │ │ • Increase dose │ │ RESISTANT: │
│ Step 2 if │ │ • Ensure │ │ • ECT (1st │
│ persists │ │ adherence │ │ choice if │
│ │ │ • Address │ │ severe) │
│ │ │ comorbidity │ │ • Esketamine │
│ │ │ │ │ • Lithium │
│ │ │ If no response: │ │ augmentation │
│ │ │ • Switch SSRI │ │ • Atypical │
│ │ │ • Switch class │ │ antipsychotic │
│ │ │ (SNRI, TCA) │ │ augmentation │
│ │ │ • Augment (Li+, │ │ • rTMS │
│ │ │ aripiprazole) │ │ • VNS/DBS │
└─────────────────┘ └─────────────────┘ └─────────────────┘
│
▼
┌─────────────────────────────────┐
│ CONTINUATION PHASE │
│ • Maintain effective treatment │
│ for 6-12 months after │
│ remission │
│ • Same dose that achieved │
│ remission │
│ • Monitor for relapse │
└─────────────────────────────────┘
│
▼
┌─────────────────────────────────┐
│ MAINTENANCE PHASE │
│ • Consider lifelong Rx if: │
│ - ≥3 prior episodes │
│ - 2 episodes + risk factors │
│ - Severe or chronic episodes │
│ • Gradual taper (25%/month) │
│ if discontinuing │
└─────────────────────────────────┘
9.3 Pharmacological Management
9.3.1 First-Line Antidepressants: SSRIs
SSRIs are first-line due to favorable efficacy-to-side-effect ratio, safety in overdose, and once-daily dosing. [4]
Mechanism of Action:
- Selectively inhibit serotonin (5-HT) reuptake transporter (SERT)
- Increase synaptic 5-HT availability
- Downstream effects: receptor desensitization, BDNF upregulation, neurogenesis
- Therapeutic effect requires 2-4 weeks (receptor and neuroplastic changes)
SSRI Comparison Table:
| Drug | Starting Dose | Target Dose | Max Dose | Half-life | Key Characteristics |
|---|---|---|---|---|---|
| Sertraline | 50 mg/day | 100-150 mg | 200 mg | 26 h | First choice; safest post-MI; GI side effects |
| Escitalopram | 10 mg/day | 10-20 mg | 20 mg | 27-32 h | Most selective; well tolerated; QTc at high doses |
| Fluoxetine | 20 mg/day | 20-40 mg | 80 mg | 4-6 days | Long half-life; activating; fewer WD symptoms |
| Citalopram | 20 mg/day | 20-40 mg | 40 mg | 35 h | QTc prolongation risk; max 20mg if > 60 years |
| Paroxetine | 20 mg/day | 20-40 mg | 50 mg | 21 h | Short half-life; worse WD; anticholinergic |
| Fluvoxamine | 50 mg/day | 100-200 mg | 300 mg | 15 h | CYP1A2 inhibitor; used more for OCD |
SSRI Side Effects:
| Side Effect | Onset | Frequency | Management |
|---|---|---|---|
| GI upset (nausea, diarrhea) | Days 1-7 | 25-30% | Take with food; usually transient |
| Headache | Week 1 | 15-20% | Usually transient; analgesics PRN |
| Insomnia/agitation | Week 1-2 | 10-15% | Give in morning; consider adjunct hypnotic |
| Sexual dysfunction | Weeks 2-4 | 30-50% | May persist; switch or add bupropion |
| Weight gain | Months | 10-20% | Monitor; consider switch |
| Emotional blunting | Weeks | 20-30% | Dose reduction; consider switch |
| Hyponatremia (SIADH) | Weeks | 1-2% | Elderly at risk; monitor Na+ |
| Bleeding risk | — | Increased | Caution with NSAIDs, anticoagulants |
| Discontinuation syndrome | On stopping | 20-50% | Taper gradually (25%/month) |
9.3.2 Second-Line Antidepressants
| Drug Class | Examples | Mechanism | Indications | Key Considerations |
|---|---|---|---|---|
| SNRIs | Venlafaxine 75-225mg, Duloxetine 60-120mg | 5-HT + NE reuptake inhibition | Failed SSRI; melancholic; pain comorbidity | HTN monitoring; worse WD |
| Mirtazapine | 15-45mg | α2-antagonist, 5-HT2/3 antagonist, H1 antagonist | Insomnia; weight loss; elderly | Sedation; weight gain |
| Bupropion | 150-450mg | NE/DA reuptake inhibition | Fatigue; sexual dysfunction; smoking | Contraindicated in seizures, eating disorders |
| TCAs | Amitriptyline, Nortriptyline | Multiple neurotransmitter effects | Treatment-resistant; chronic pain | Lethal in OD; ECG monitoring |
| MAOIs | Phenelzine, Tranylcypromine | Monoamine oxidase inhibition | Treatment-resistant; atypical depression | Dietary restrictions; drug interactions |
9.3.3 Augmentation Strategies
For partial response after adequate trial (≥8 weeks at therapeutic dose):
| Strategy | Agent | Dose | Evidence Level | Notes |
|---|---|---|---|---|
| Lithium | Lithium carbonate | 600-900mg (level 0.6-0.8) | Level 1a | Gold standard augmentation; renal/thyroid monitoring |
| Atypical antipsychotic | Aripiprazole | 2-10mg | Level 1a | FDA-approved augmentation; metabolic monitoring |
| Quetiapine | 50-300mg | Level 1a | Sedation; weight gain | |
| Olanzapine | 5-20mg | Level 1b | Weight gain; combine with fluoxetine (Symbyax) | |
| Thyroid | Liothyronine (T3) | 25-50mcg | Level 1b | Especially in women; monitor TFTs |
| Buspirone | Buspirone | 15-60mg | Level 2b | 5-HT1A agonist |
9.3.4 Treatment-Resistant Depression (TRD)
Definition: Failure to respond to ≥2 adequate antidepressant trials (different classes, adequate dose, 6-8 weeks each). [15]
Evaluation:
- Confirm diagnosis (rule out bipolar, medical causes)
- Assess adherence
- Optimize current treatment
- Address comorbidities (substance use, personality disorder, anxiety)
- Consider pharmacogenomic testing
TRD Treatment Options:
| Treatment | Evidence | Response Rate | Considerations |
|---|---|---|---|
| ECT | Level 1a | 50-70% | Gold standard for severe TRD; see Section 9.4 |
| Esketamine (Spravato) | Level 1b | 54-70% | FDA-approved for TRD; intranasal; REMS program |
| rTMS | Level 1a | 30-50% | Non-invasive; daily sessions x 4-6 weeks |
| IV Ketamine | Level 2a | 60-70% | Rapid onset; off-label; requires supervision |
| VNS | Level 2b | 20-30% | Implantable; adjunctive; slow onset |
| MAOI | Level 1b | 50-60% | Underused; dietary restrictions |
9.4 Electroconvulsive Therapy (ECT)
ECT remains the most effective treatment for severe depression and is the gold standard for treatment-resistant depression. [13]
Mechanism:
- Controlled induction of generalized seizure under anesthesia
- Increases BDNF, promotes neuroplasticity
- Modulates neurotransmitter systems
- Resets HPA axis function
- Precise mechanism unknown but efficacy well-established
Indications:
- Severe depression with psychotic features
- Severe depression with catatonia
- High suicide risk requiring rapid response
- Food/fluid refusal with medical compromise
- Treatment-resistant depression (failed ≥2 medication trials)
- Prior positive response to ECT
- Pregnancy (safer than some medications)
- Medical frailty precluding medications
Contraindications:
- Absolute: Recent MI (less than 3 months), recent stroke, intracranial mass with increased ICP
- Relative: Severe pulmonary disease, unstable angina, retinal detachment, pregnancy (not absolute)
Procedure:
- Pre-procedure fasting (NPO 8 hours)
- IV access, ECG monitoring, pulse oximetry, BP
- Short-acting anesthetic (propofol, methohexital, etomidate)
- Muscle relaxant (succinylcholine)
- Bite block insertion
- Electrode placement (bilateral or unilateral)
- Brief-pulse electrical stimulus (typically 0.5-2 seconds)
- Induced seizure (target 20-60 seconds motor; 25-120 seconds EEG)
- Recovery and monitoring
Treatment Course:
- Acute: 6-12 sessions (typically 3x weekly)
- Continuation: Weekly → biweekly → monthly
- Maintenance: Monthly sessions for relapse prevention
Side Effects:
| Side Effect | Frequency | Duration | Management |
|---|---|---|---|
| Confusion (postictal) | 100% | Minutes-hours | Observation |
| Headache | 30-50% | Hours | Simple analgesia |
| Nausea | 10-20% | Hours | Antiemetics |
| Muscle soreness | 10-20% | Days | Analgesia |
| Anterograde amnesia | Variable | Weeks-months | Usually resolves |
| Retrograde amnesia | Variable | Weeks to permanent | Pre-treatment counseling |
| Cognitive dysfunction | Variable | Usually transient | Unilateral placement; modified technique |
Efficacy:
- Response rate: 50-70% in TRD
- Remission rate: 35-50%
- Faster response than medication (often within 1-2 weeks)
- Superior to medication in severe, psychotic, and catatonic depression
9.5 Psychotherapy
9.5.1 Cognitive Behavioral Therapy (CBT)
CBT is the most extensively studied and evidence-based psychotherapy for depression. [12]
Theoretical Framework:
- Depression maintained by negative cognitive patterns
- Beck's cognitive triad: negative views of self, world, and future
- Cognitive distortions: all-or-nothing thinking, catastrophizing, mind-reading, should statements
- Behavioral component: reduced positive reinforcement due to withdrawal/inactivity
Key Techniques:
- Behavioral Activation: Scheduling pleasurable and mastery activities
- Cognitive Restructuring: Identifying and challenging negative automatic thoughts
- Behavioral Experiments: Testing beliefs through real-world experiments
- Thought Records: Documenting situations, thoughts, emotions, alternative thoughts
- Problem-Solving: Structured approach to addressing life problems
- Graded Task Assignment: Breaking overwhelming tasks into manageable steps
Treatment Parameters:
- Duration: 12-20 sessions (typically weekly)
- Format: Individual (most evidence), group (cost-effective), computerized (iCBT)
- Delivered by: Trained therapists (psychologists, counselors, CBT therapists)
Efficacy:
- Comparable to antidepressants for mild-moderate depression
- Combined treatment superior for severe depression
- Lower relapse rates than medication alone after treatment ends
- Effect size: d = 0.6-0.8 vs. control
9.5.2 Other Evidence-Based Psychotherapies
| Therapy | Description | Evidence Level | Best Suited For |
|---|---|---|---|
| Interpersonal Therapy (IPT) | Focuses on interpersonal disputes, role transitions, grief, deficits | Level 1a | Relationship issues; life transitions; grief |
| Behavioral Activation (BA) | Pure behavioral approach; activity scheduling | Level 1a | Severe depression; anhedonia; simpler delivery |
| Mindfulness-Based Cognitive Therapy (MBCT) | Mindfulness + CBT elements | Level 1a | Relapse prevention in recurrent MDD |
| Problem-Solving Therapy | Structured problem-solving approach | Level 1b | Primary care; practical orientation |
| Short-Term Psychodynamic Therapy | Insight-oriented; unconscious conflicts | Level 2a | Preference for exploration; personality issues |
9.6 Other Treatments
9.6.1 Repetitive Transcranial Magnetic Stimulation (rTMS)
- Non-invasive neuromodulation using magnetic pulses
- FDA-approved for TRD (2008)
- Targets left dorsolateral prefrontal cortex (DLPFC)
- Sessions: 20-30 daily sessions over 4-6 weeks
- Response rate: 30-50%; remission 15-30%
- Minimal side effects (scalp discomfort, headache)
- Newer protocols: theta-burst (shorter sessions), accelerated (multiple daily sessions)
9.6.2 Intranasal Esketamine (Spravato)
- S-enantiomer of ketamine; NMDA receptor antagonist
- FDA-approved for TRD (2019) and MDD with suicidal ideation (2020)
- Mechanism: rapid glutamatergic effects; BDNF release; synaptogenesis
- Administration: Supervised nasal spray (56mg or 84mg); twice weekly initially, then weekly/biweekly
- Requires 2-hour observation due to dissociative effects, sedation, BP elevation
- Response often seen within hours to days
- REMS program required (registration, monitoring)
9.6.3 Light Therapy
- First-line for Seasonal Affective Disorder (SAD)
- 10,000 lux bright light for 30 minutes each morning
- Onset of action: 1-2 weeks
- Side effects: Headache, eye strain (rare)
- May augment antidepressants in non-seasonal depression
9.6.4 Exercise
- Evidence equivalent to antidepressants for mild-moderate depression
- Mechanism: BDNF upregulation, endorphin release, anti-inflammatory effects
- Prescription: Moderate-intensity aerobic exercise, 30-45 minutes, 3-5x weekly
- Benefits beyond depression: cardiovascular health, metabolic health, sleep
SECTION 10: Special Populations
10.1 Elderly Depression (Late-Life Depression)
Epidemiology: 15-20% of elderly community dwellers; 30-45% in nursing homes
Special Considerations:
- Presentation: More somatic complaints, cognitive symptoms, anxiety, less expressed sadness
- "Pseudodementia": Depression-related cognitive impairment mimics dementia (but is reversible)
- Vascular depression: Associated with cerebrovascular disease and executive dysfunction
- Comorbidity: Medical illness, polypharmacy, social isolation, bereavement
Assessment:
- Geriatric Depression Scale (GDS-15) validated for this population
- Screen for cognitive impairment (MoCA, MMSE)
- Medical workup: TSH, B12, metabolic panel, consider neuroimaging
Treatment Modifications:
- "Start low, go slow, but go": Lower starting doses but titrate to full therapeutic doses
- SSRIs preferred (sertraline, escitalopram, citalopram max 20mg)
- Avoid anticholinergic drugs (TCAs, paroxetine)
- ECT is safe and highly effective in elderly
- Address psychosocial factors: isolation, loss, role changes
10.2 Perinatal Depression
Epidemiology:
- Prenatal depression: 10-15%
- Postpartum depression: 10-15% (peak 4-6 weeks postpartum)
- Postpartum psychosis: 0.1-0.2% (medical emergency)
Screening: Edinburgh Postnatal Depression Scale (EPDS) at prenatal and postnatal visits
Treatment in Pregnancy:
- Untreated depression itself carries risks (preterm birth, low birth weight, bonding)
- SSRIs generally considered acceptable (sertraline preferred)
- Paroxetine avoided in first trimester (cardiac defects)
- ECT safe and effective
- Psychotherapy (CBT, IPT) first-line for mild-moderate
Treatment in Breastfeeding:
- Sertraline: Low milk transfer, minimal infant serum levels (preferred)
- Paroxetine: Low milk transfer
- Fluoxetine: Higher transfer, long half-life (caution)
Postpartum Psychosis:
- Psychiatric emergency requiring immediate hospitalization
- Screen for infanticide risk
- Treatment: Antipsychotic + mood stabilizer (often becomes bipolar diagnosis)
10.3 Depression with Comorbid Medical Illness
Cardiovascular Disease:
- Depression is independent risk factor for MI and mortality
- Sertraline is safest post-MI (SADHART trial)
- Avoid TCAs (cardiotoxic)
Diabetes:
- Bidirectional relationship; each doubles risk of the other
- Depression impairs diabetes self-care
- SSRIs may improve glycemic control
Chronic Pain:
- High comorbidity; shared pathophysiology
- Duloxetine, TCAs effective for both
Cancer:
- Depression common (25-50%); often undertreated
- Treat aggressively; improves quality of life and possibly survival
SECTION 11: Prognosis
11.1 Natural History
Episode Duration:
- Untreated episode: Median 6-9 months; 10-20% become chronic (> 2 years)
- Treated episode: Response typically by 4-6 weeks; remission by 8-12 weeks
Recurrence:
- After 1 episode: 50% recurrence risk
- After 2 episodes: 70-80% recurrence risk
- After 3 episodes: 90% recurrence risk [6]
Chronicity:
- 15-20% have chronic course (continuous or minimal remission)
- Risk factors: Early onset, severity, comorbidity, inadequate treatment
11.2 Prognostic Factors
Favorable Prognosis:
- Acute onset
- Identifiable precipitant
- Good premorbid functioning
- Strong social support
- Full inter-episode recovery
- Treatment adherence
- Single episode
Poor Prognosis:
- Insidious onset
- Chronic course
- Comorbid personality disorder
- Comorbid substance use
- Residual symptoms
- Multiple prior episodes
- Early onset
- Psychotic features (higher recurrence)
11.3 Mortality
Suicide:
- Lifetime mortality: 4-7% in hospitalized patients; 2-4% in outpatients [7]
- 15-25% of patients with recurrent MDD will attempt suicide
All-Cause Mortality:
- Depression associated with 1.8x increased all-cause mortality
- Cardiovascular mortality increased 1.5-2x
- Mechanisms: Behavioral (smoking, inactivity, non-adherence), biological (inflammation, HPA dysregulation)
SECTION 12: Complications
12.1 Psychiatric Complications
| Complication | Incidence | Risk Factors | Prevention/Management |
|---|---|---|---|
| Suicide/self-harm | 10-15% attempt | Prior attempt, male, hopelessness | Risk assessment, safety planning |
| Treatment resistance | 30% | Chronicity, comorbidity, severity | Optimize treatment, ECT |
| Recurrence | 50-90% | Prior episodes, residual symptoms | Maintenance treatment |
| Chronicity | 15-20% | Early onset, personality disorder | Aggressive early treatment |
| Substance use disorder | 30% | Self-medication, genetics | Integrated treatment |
12.2 Medical Complications
| Complication | Mechanism | Prevention |
|---|---|---|
| Cardiovascular disease | Inflammation, behavioral | Treat depression, lifestyle |
| Diabetes | Behavioral, HPA dysregulation | Metabolic monitoring |
| Obesity | Atypical symptoms, medications | Diet, exercise, drug choice |
| Osteoporosis | SSRIs affect bone metabolism | Vitamin D, calcium, weight-bearing exercise |
12.3 Treatment Complications
| Complication | Agent | Presentation | Management |
|---|---|---|---|
| Serotonin syndrome | SSRI + MAOI/serotonergic | Hyperthermia, rigidity, clonus, AMS | Stop agents, supportive, cyproheptadine |
| SSRI discontinuation | Any SSRI (worst: paroxetine) | Dizziness, nausea, "brain zaps" | Gradual taper (25%/month) |
| Hyponatremia | SSRIs (elderly) | Confusion, seizures | Monitor Na+, hold SSRI |
| QTc prolongation | Citalopram, TCAs | Arrhythmia risk | ECG monitoring, dose limits |
| Manic switch | Any antidepressant (undiagnosed bipolar) | Mania/hypomania symptoms | Stop antidepressant, mood stabilizer |
SECTION 13: Evidence and Guidelines
13.1 Major Guidelines
NICE NG222 (2022) - Depression in Adults: [5]
- Key recommendations:
- Shared decision-making regarding treatment choice
- "For mild: Active monitoring, low-intensity interventions, physical activity"
- "For moderate-severe: Combination of antidepressant + high-intensity psychological therapy"
- Antidepressant choice based on side effect profile and patient preference
- Continuation treatment for 6+ months after remission
- Gradual withdrawal when discontinuing
APA Practice Guideline (2010, updated 2019): [12]
- Pharmacotherapy or evidence-based psychotherapy as initial treatment
- Combined treatment for moderate-severe or chronic depression
- Sequential treatment strategies for inadequate response
- Maintenance treatment for recurrent depression
CANMAT Guidelines (2016, updated 2023):
- First-line: SSRIs, SNRIs, bupropion, mirtazapine
- Second-line: TCAs, trazodone, MAOIs
- Adjunctive: Aripiprazole, quetiapine, lithium
13.2 Landmark Trials
STAR*D (Sequenced Treatment Alternatives to Relieve Depression) - 2006 [11]
- Design: Large-scale, multi-step, pragmatic trial (n=3,671)
- Question: What is the effectiveness of sequenced treatments in real-world patients?
- Key Findings:
- "Level 1 (Citalopram): 33% remission"
- "Cumulative remission after 4 steps: 67%"
- Each subsequent step had lower remission rates
- Augmentation and switching equally effective
- Impact: Established evidence for sequential treatment strategies; showed need for multiple trials
Cipriani et al. Lancet Meta-Analysis - 2018 [4]
- Design: Network meta-analysis of 522 RCTs (116,477 participants)
- Question: Comparative efficacy and acceptability of 21 antidepressants
- Key Findings:
- All 21 antidepressants more effective than placebo
- "Most effective: Amitriptyline, mirtazapine, venlafaxine"
- "Most tolerable: Agomelatine, citalopram, escitalopram, fluoxetine"
- "Best balance: Escitalopram, sertraline"
- Impact: Definitive evidence that antidepressants work; guides drug selection
SADHART (Sertraline Antidepressant Heart Attack Randomized Trial) - 2002
- Design: RCT of sertraline vs. placebo in post-MI depression (n=369)
- Key Finding: Sertraline safe and effective post-MI
- Impact: Established SSRI safety in cardiovascular disease
UK ECT Review Group Cochrane Review - 2019 [13]
- Design: Systematic review and meta-analysis
- Key Finding: ECT superior to sham ECT and pharmacotherapy for depression
- Impact: Confirms ECT efficacy; gold standard for severe/TRD
PANDA Trial - 2019
- Design: Primary care RCT of sertraline vs. placebo (n=655)
- Key Findings: Sertraline improved anxiety symptoms by 6 weeks; depression improvement by 12 weeks
- Impact: Supports SSRI use in primary care; highlights early anxiety improvement
SECTION 14: Examination Focus
14.1 High-Yield Examination Topics
| Topic | Examination | Key Points |
|---|---|---|
| DSM-5 criteria | All | SIG E CAPS mnemonic; core symptoms; 2-week duration |
| PHQ-9 scoring | USMLE, PLAB | Score thresholds; Item 9 interpretation |
| SSRI pharmacology | MRCP, USMLE | MOA, side effects, half-lives, drug interactions |
| Suicide risk assessment | All | Risk/protective factors; C-SSRS; safety planning |
| Serotonin syndrome | MRCP, USMLE | Recognition, triggers, management |
| ECT indications | MRCPsych, FRANZCP | Psychotic, catatonic, TRD, pregnancy |
| Bipolar screening | All | MDQ; risks of SSRI monotherapy in bipolar |
| Pseudodementia | MRCP, USMLE | Depression vs. dementia features |
| Perinatal depression | MRCOG, USMLE | EPDS; drug safety in pregnancy/lactation |
14.2 Sample Examination Questions
Question 1 (MRCP Style): A 68-year-old man presents with 6 weeks of low mood, poor concentration, and memory difficulties. He repeatedly answers "I don't know" to cognitive testing questions. His wife reports he no longer enjoys gardening.
What is the most likely diagnosis?
- A) Alzheimer's disease
- B) Major depressive disorder with pseudodementia
- C) Vascular dementia
- D) Frontotemporal dementia
- E) Lewy body dementia
Answer: B. "I don't know" responses with subjective distress about deficits and anhedonia are characteristic of depressive pseudodementia. Dementia typically shows confabulation and lack of insight.
Question 2 (USMLE Style): A 32-year-old woman with MDD on sertraline 100mg reports improvement in mood but persistent sexual dysfunction and emotional blunting. Which medication would be most appropriate to add?
- A) Lithium
- B) Bupropion
- C) Aripiprazole
- D) Mirtazapine
- E) Increase sertraline
Answer: B. Bupropion (NE/DA mechanism, no sexual side effects) can counteract SSRI-induced sexual dysfunction.
Question 3 (MRCPsych Style): Which feature would most strongly suggest electroconvulsive therapy as first-line treatment for major depressive disorder?
- A) Comorbid anxiety disorder
- B) First episode of depression
- C) Nihilistic delusions
- D) Atypical features
- E) Poor response to one SSRI
Answer: C. Psychotic features (including nihilistic delusions) are an indication for ECT as first-line treatment.
SECTION 15: References
-
American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5). Arlington, VA: American Psychiatric Publishing; 2013. ISBN: 978-0-89042-555-8
-
World Health Organization. Depression and Other Common Mental Disorders: Global Health Estimates. Geneva: World Health Organization; 2017. PMID: 28315433
-
Malhi GS, Mann JJ. Depression. Lancet. 2018;392(10161):2299-2312. PMID: 30396512
-
Cipriani A, Furukawa TA, Salanti G, et al. Comparative efficacy and acceptability of 21 antidepressant drugs for the acute treatment of adults with major depressive disorder: a systematic review and network meta-analysis. Lancet. 2018;391(10128):1357-1366. PMID: 29477251
-
National Institute for Health and Care Excellence. Depression in adults: treatment and management. NICE guideline [NG222]. 2022. https://www.nice.org.uk/guidance/ng222
-
Mueller TI, Leon AC, Keller MB, et al. Recurrence after recovery from major depressive disorder during 15 years of observational follow-up. Am J Psychiatry. 1999;156(7):1000-1006. PMID: 10401442
-
Bostwick JM, Pankratz VS. Affective disorders and suicide risk: a reexamination. Am J Psychiatry. 2000;157(12):1925-1932. PMID: 11097952
-
National Institute of Mental Health. Major Depression. Statistics. 2020. https://www.nimh.nih.gov/health/statistics/major-depression
-
Kessler RC, Bromet EJ. The epidemiology of depression across cultures. Annu Rev Public Health. 2013;34:119-138. PMID: 23514317
-
Lim GY, Tam WW, Lu Y, et al. Prevalence of Depression in the Community from 30 Countries between 1994 and 2014. Sci Rep. 2018;8(1):2861. PMID: 29434331
-
Rush AJ, Trivedi MH, Wisniewski SR, et al. Acute and longer-term outcomes in depressed outpatients requiring one or several treatment steps: a STAR*D report. Am J Psychiatry. 2006;163(11):1905-1917. PMID: 17074942
-
American Psychiatric Association. Practice Guideline for the Treatment of Patients with Major Depressive Disorder, Third Edition. Am J Psychiatry. 2010;167(10 Suppl):1-152. PMID: 21890579
-
UK ECT Review Group. Efficacy and safety of electroconvulsive therapy in depressive disorders: a systematic review and meta-analysis. Lancet. 2003;361(9360):799-808. PMID: 12642045
-
Kroenke K, Spitzer RL, Williams JB. The PHQ-9: validity of a brief depression severity measure. J Gen Intern Med. 2001;16(9):606-613. PMID: 11556941
-
McIntyre RS, Filteau MJ, Martin L, et al. Treatment-resistant depression: definitions, review of the evidence, and algorithmic approach. J Affect Disord. 2014;156:1-7. PMID: 24314926
-
Jick H, Kaye JA, Jick SS. Antidepressants and the risk of suicidal behaviors. JAMA. 2004;292(3):338-343. PMID: 15265848
-
Viktorin A, Lichtenstein P, Thase ME, et al. The risk of switch to mania in patients with bipolar disorder during treatment with an antidepressant alone and in combination with a mood stabilizer. Am J Psychiatry. 2014;171(10):1067-1073. PMID: 24935197
-
Koenig AM, Bhalla RK, Butters MA. Cognitive functioning and late-life depression. J Int Neuropsychol Soc. 2014;20(5):461-467. PMID: 24685173
-
Simon RI. Suicide risk assessment: Gateway to treatment and management. In: Simon RI, Hales RE, eds. Textbook of Suicide Assessment and Management. 2nd ed. American Psychiatric Publishing; 2012:3-28.
-
Santomauro DF, Mantilla Herrera AM, Shadid J, et al. Global prevalence and burden of depressive and anxiety disorders in 204 countries and territories in 2020 due to the COVID-19 pandemic. Lancet. 2021;398(10312):1700-1712. PMID: 34634250
-
Miller AH, Raison CL. The role of inflammation in depression: from evolutionary imperative to modern treatment target. Nat Rev Immunol. 2016;16(1):22-34. PMID: 26711676
-
Jacka FN, O'Neil A, Opie R, et al. A randomised controlled trial of dietary improvement for adults with major depression (the 'SMILES' trial). BMC Med. 2017;15(1):23. PMID: 28137247
Last Updated: 2026-01-09
Medical Disclaimer: MedVellum content is for educational purposes and clinical reference. Clinical decisions should account for individual patient circumstances. Always consult appropriate specialists and current guidelines. Emergency presentations require immediate clinical assessment.
Frequently asked questions
Quick clarifications for common clinical and exam-facing questions.
When should I seek emergency care for major depressive disorder in adults?
Seek immediate emergency care if you experience any of the following warning signs: Active suicidal ideation with plan, intent, or access to means, Psychotic features (mood-congruent hallucinations or delusions), Severe psychomotor retardation or catatonic features, Complete refusal of food or fluids with dehydration, Profound cognitive impairment (depressive pseudodementia), Command auditory hallucinations to self-harm, Recent discharge from psychiatric admission.
Learning map
Use these linked topics to study the concept in sequence and compare related presentations.
Prerequisites
Start here if you need the foundation before this topic.
- Neurotransmitter Pharmacology
- Psychiatric Assessment
Differentials
Competing diagnoses and look-alikes to compare.
- Bipolar Disorder
- Persistent Depressive Disorder
- Adjustment Disorder
Consequences
Complications and downstream problems to keep in mind.
- Suicide and Self-Harm
- Treatment-Resistant Depression