Drug-Induced Liver Injury
Pattern classification is fundamental: hepatocellular (ALT-predominant), cholestatic (alkaline phosphatase-predominant), or mixed, determined by the R-ratio: (ALT/ULN) ÷ (ALP/ULN). R ≥5 indicates hepatocellular...
What matters first
Pattern classification is fundamental: hepatocellular (ALT-predominant), cholestatic (alkaline phosphatase-predominant), or mixed, determined by the R-ratio: (ALT/ULN) ÷ (ALP/ULN). R ≥5 indicates hepatocellular...
Hy's Law: ALT less than 3x ULN + bilirubin less than 2x ULN (10-50% mortality)
9 Jan 2026
Generated educational material; verify before clinical use.
Visible references section
See the concept before reading it
Study the key anatomy, imaging, and decision pathways as full teaching plates.
Clinical board
A visual summary of the highest-yield teaching signals on this page.
Urgent signals
Safety-critical features pulled from the topic metadata.
- Hy's Law: ALT less than 3x ULN + bilirubin less than 2x ULN (10-50% mortality)
- Signs of acute liver failure (encephalopathy, coagulopathy, INR less than 1.5)
- Paracetamol/acetaminophen overdose
- Rapidly rising bilirubin or INR
Content status and exam context
This page is AI-generated educational content. It may contain errors or omissions and is not a substitute for current guidelines, local protocols, senior clinical judgement, or professional medical advice.
MedVellum does not claim an individual clinician reviewer, board certification, or professional credential for this page unless a future version names a real, verifiable reviewer.
Clinical explanation and evidence
Drug-Induced Liver Injury
1. Clinical Overview
Summary
Drug-induced liver injury (DILI) represents liver damage caused by prescription medications, over-the-counter drugs, herbal products, or dietary supplements. It is the most common cause of acute liver failure (ALF) in developed countries, accounting for over 50% of cases. [1,2] DILI encompasses a spectrum from asymptomatic transaminase elevation to fulminant hepatic failure requiring emergency liver transplantation. The condition is broadly classified as intrinsic (dose-dependent, predictable—exemplified by paracetamol/acetaminophen) or idiosyncratic (unpredictable, immune-mediated or metabolic—such as amoxicillin-clavulanate). [3]
Pattern classification is fundamental: hepatocellular (ALT-predominant), cholestatic (alkaline phosphatase-predominant), or mixed, determined by the R-ratio: (ALT/ULN) ÷ (ALP/ULN). [4] R ≥5 indicates hepatocellular injury; R ≤2 indicates cholestatic; R between 2-5 represents mixed pattern. Hy's Law, a critical prognostic marker, identifies cases with 10-50% mortality risk: hepatocellular injury (ALT > 3× ULN) plus jaundice (bilirubin > 2× ULN) without significant cholestasis (ALP less than 2× ULN). [5]
Management centres on immediate withdrawal of the offending agent, supportive care, and specific antidotes where available (N-acetylcysteine for paracetamol, L-carnitine for valproate). [6] Diagnosis remains one of exclusion after systematically ruling out viral hepatitis, autoimmune hepatitis, ischaemic hepatitis, and biliary obstruction. The Roussel Uclaf Causality Assessment Method (RUCAM) score provides structured causality assessment. [7]
Key Facts
- Definition: Liver injury caused by medications, herbal products, or dietary supplements with temporal relationship to exposure
- Incidence: 14-19 per 100,000 persons annually in general population; higher in certain ethnic groups [8]
- ALF Causation: 46-52% of acute liver failure cases in the United States and United Kingdom [1,2]
- Mortality: Hy's Law-positive cases carry 10-50% mortality; overall DILI mortality 5-12% [5,9]
- Common Culprits: Paracetamol/acetaminophen (intrinsic), amoxicillin-clavulanate, isoniazid, NSAIDs (diclofenac), statins, antiepileptics (valproate, phenytoin), herbal/dietary supplements [10]
- Pathognomonic Features: Temporal relationship (onset 5-90 days after drug initiation for most idiosyncratic cases); improvement after drug withdrawal
- Diagnosis: Clinical diagnosis of exclusion supported by RUCAM causality score ≥6 (probable/highly probable) [7]
- Gold Standard Treatment: Immediate drug discontinuation; N-acetylcysteine for paracetamol and consideration in non-paracetamol ALF; supportive care
- Prognosis: Most cases resolve within 1-3 months; 1-5% progress to ALF; 5-20% develop chronic injury [9,11]
Clinical Pearls
Diagnostic Pearl: Always calculate the R-ratio: (ALT/ULN) ÷ (ALP/ULN). R ≥5 = hepatocellular (higher risk of ALF); R ≤2 = cholestatic (more indolent, risk of chronicity); R 2-5 = mixed pattern.
Emergency Pearl: Hy's Law criteria (ALT > 3× ULN + bilirubin > 2× ULN + absence of cholestasis) mandate urgent hepatology referral and transplant centre notification. Mortality risk 10-50% without intervention.
Treatment Pearl: N-acetylcysteine improves transplant-free survival even in non-paracetamol acute liver failure with grade I-II encephalopathy. Consider early in severe DILI. [12]
Pitfall Warning: Herbal and dietary supplements (HDS) cause 16-20% of DILI cases in Western countries. Many patients do not consider these "medications." Always ask specifically: "Do you take any vitamins, supplements, herbal remedies, or traditional medicines?" [13]
Genetic Pearl: HLA-B57:01 predicts flucloxacillin hepatotoxicity; HLA-A33:01 predicts terbinafine DILI; HLA-B35:02 and DRB116 associate with amoxicillin-clavulanate injury. Pharmacogenomic screening may become standard practice. [14]
Mnemonic: DILI STOP - Drug history complete (including supplements), Investigations (exclude other causes), Liver pattern (R-ratio), Identify high-risk (Hy's Law), Stop offending drug, Time to recovery (serial LFTs), Other support (NAC, nutrition), Paracetamol protocol if applicable
Why This Matters Clinically
DILI is the leading cause of acute liver failure in developed nations, exceeding all viral hepatitides combined. [1,2] It is also the most common reason for post-marketing drug withdrawal and black-box warnings by regulatory agencies. Early recognition prevents progression to irreversible liver failure. Delayed withdrawal increases mortality exponentially. This is a critical patient safety and pharmacovigilance topic relevant to every prescribing clinician across all specialties. Understanding DILI patterns, causality assessment, and management protocols is essential for safe prescribing practice and is a core examination topic for MRCP, FRACP, gastroenterology training, and clinical pharmacology.
2. Epidemiology
Incidence and Prevalence
General Population:
- Incidence: 14-19 per 100,000 persons per year in European and North American populations [8]
- Hospital admissions for DILI: 1-2 per 100,000 annually
- True incidence likely underestimated due to underreporting and misdiagnosis [8]
Acute Liver Failure:
- DILI causes 46-52% of ALF cases in the United States and United Kingdom [1,2]
- Paracetamol/acetaminophen alone accounts for 39-46% of ALF
- Non-paracetamol DILI accounts for 12-13% of ALF [2]
Idiosyncratic DILI:
- Occurs in 1 per 10,000 to 1 per 100,000 drug exposures depending on agent [15]
- Latency: typically 5-90 days for idiosyncratic DILI; less than 24 hours for re-exposure
- Intrinsic DILI (paracetamol): predictable, dose > 10g in adults [16]
Geographic Variation:
- Western countries: antibiotics (45%), CNS agents (15%), NSAIDs (10%) [10]
- Asia: herbal and traditional medicines account for higher proportion (26-73% in some series) [13]
- Tuberculosis-endemic regions: isoniazid and rifampicin major contributors
Common Causative Agents by Pattern
| Drug Class | Examples | Pattern | Latency | Notes |
|---|---|---|---|---|
| Antimicrobials | Amoxicillin-clavulanate | Cholestatic/Mixed | 1-6 weeks | Most common idiosyncratic DILI in West [10] |
| Flucloxacillin | Cholestatic | 1-6 weeks | HLA-B*57:01 association [14] | |
| Isoniazid | Hepatocellular | 2-12 weeks | 10-20% mild ALT elevation; 1% severe [17] | |
| Nitrofurantoin | Mixed | Acute or chronic (months-years) | Can mimic autoimmune hepatitis | |
| Minocycline | Hepatocellular | 1-12 months | Autoimmune features | |
| Analgesics | Paracetamol/Acetaminophen | Hepatocellular | Hours | Intrinsic; dose > 10g (150mg/kg) [16] |
| Diclofenac | Hepatocellular | 1-12 weeks | Higher risk than other NSAIDs | |
| Sulindac | Hepatocellular | 1-8 weeks | Idiosyncratic | |
| Anticonvulsants | Valproate | Hepatocellular | 2-12 weeks | Mitochondrial toxicity; use L-carnitine [18] |
| Phenytoin | Hepatocellular | 2-8 weeks | DRESS syndrome in 10% | |
| Carbamazepine | Hepatocellular/Mixed | 2-8 weeks | DRESS syndrome in 10-15% | |
| Cardiovascular | Amiodarone | Hepatocellular/Mixed | Months | Steatohepatitis pattern; chronic injury |
| Statins (all) | Hepatocellular | 1-6 months | Usually mild; severe injury rare [19] | |
| Methyldopa | Hepatocellular | 1-20 weeks | Autoimmune features | |
| Psychiatric | Chlorpromazine | Cholestatic | 1-4 weeks | Classic cholestasis |
| Risperidone | Cholestatic | Variable | Rare | |
| Herbal/Dietary Supplements | Green tea extract | Hepatocellular | 1-6 months | Dose-dependent risk [13] |
| Anabolic steroids | Cholestatic | Weeks-months | Bland cholestasis | |
| Kava, black cohosh | Hepatocellular | Variable | Multiple reported cases | |
| Turmeric/curcumin | Hepatocellular | 1-5 months | Increasing recognition |
Risk Factors
| Factor | Impact | Mechanism/Notes |
|---|---|---|
| Age > 55 years | 2-3× risk of severe injury [20] | Reduced hepatic reserve, polypharmacy, mitochondrial function decline |
| Female sex | 2× risk for idiosyncratic DILI [20] | Immune-mediated mechanisms; hormonal influences |
| Pre-existing liver disease | Variable | Reduced hepatic reserve; NAFLD may increase susceptibility |
| Chronic alcohol use | Synergistic toxicity | CYP2E1 induction (paracetamol); glutathione depletion |
| Polypharmacy | Increased risk | Drug-drug interactions; altered metabolism |
| Genetic polymorphisms | Variable by drug | HLA associations (flucloxacillin, amoxicillin-clavulanate); CYP450 variants [14] |
| Obesity/Metabolic syndrome | May increase susceptibility | Underlying NAFLD; altered drug distribution |
| HIV infection | Higher risk with antiretrovirals | Immune reconstitution; mitochondrial toxicity (NRTIs) |
| TB treatment | 5-28% hepatotoxicity [17] | Isoniazid, rifampicin, pyrazinamide combinations |
Protective Factors:
- Younger age (less than 18 years) generally lower risk for idiosyncratic DILI
- Exception: aspirin-related Reye syndrome in children (now rare)
3. Pathophysiology
Overview of DILI Mechanisms
Drug-induced liver injury results from direct cellular toxicity, immune-mediated injury, or both. The liver is particularly vulnerable due to its role in drug metabolism, high blood flow, concentration of xenobiotics, and expression of drug-metabolizing enzymes.
Intrinsic (Dose-Dependent) DILI
Prototype: Paracetamol/Acetaminophen Hepatotoxicity
Phase 1: Overdose and Metabolism [16]
- Therapeutic doses (≤4g/day): 90% glucuronidation/sulfation → safe excretion
- Overdose (> 10g or > 150mg/kg): saturation of conjugation pathways
- CYP2E1 and CYP3A4 produce toxic metabolite N-acetyl-p-benzoquinone imine (NAPQI)
Phase 2: Glutathione Depletion
- NAPQI normally detoxified by hepatic glutathione (GSH)
- Overdose depletes GSH stores (typically within 2-4 hours)
- Alcohol use and fasting worsen depletion
Phase 3: Hepatocellular Necrosis
- Unbound NAPQI binds to cellular proteins (especially mitochondrial)
- Oxidative stress, mitochondrial dysfunction, ATP depletion
- Centrilobular (zone 3) necrosis—area with highest CYP450 activity
- Release of damage-associated molecular patterns (DAMPs)
Phase 4: Progression or Recovery
- With N-acetylcysteine (NAC): restores GSH, directly binds NAPQI, improves microcirculation
- Without treatment: massive necrosis, ALF, multiorgan failure [16]
Characteristics of Intrinsic DILI:
- Predictable and dose-dependent
- Short latency (less than 24 hours to days)
- Occurs in most individuals at sufficient dose
- Reproducible in animal models
Idiosyncratic DILI
Phase 1: Drug Bioactivation and Stress Signals
- Parent drug or reactive metabolites (via CYP450, especially CYP3A4, CYP2C9, CYP2E1)
- Reactive metabolites bind to cellular proteins forming drug-protein adducts
- Mitochondrial dysfunction: inhibition of β-oxidation, electron transport chain [21]
- Endoplasmic reticulum stress, unfolded protein response
- Oxidative stress generation
Phase 2: Cellular Injury and Innate Immunity
- Direct hepatocyte injury: apoptosis or necrosis
- Release of DAMPs (HMGB1, mitochondrial DNA, ATP)
- Activation of Kupffer cells (liver macrophages)
- Innate immune response: cytokine release (TNF-α, IL-1β, IL-6)
Phase 3: Adaptive Immune Response (Immune-Mediated DILI) [22]
- Drug-protein adducts serve as neoantigens (haptens)
- Presentation to CD4+ and CD8+ T cells via MHC/HLA
- HLA-restricted immune response explains genetic susceptibility [14]
- Examples:
- "Flucloxacillin: HLA-B*57:01 (OR 80.6 for DILI)"
- "Amoxicillin-clavulanate: HLA-A02:01, HLA-DRB115:01"
- "Lapatinib: HLA-DQA1*02:01"
- T-cell activation → cytotoxic T lymphocyte-mediated hepatocyte killing
- B-cell activation → autoantibodies (ANA, SMA in some cases)
Phase 4: Features of Immune-Mediated DILI:
- Eosinophilia (30-40% of cases)
- Rash (10-15%)
- Fever (10%)
- DRESS syndrome (Drug Reaction with Eosinophilia and Systemic Symptoms): severe form with multi-organ involvement [23]
- Rapid recurrence upon re-exposure (less than 24 hours)
Phase 5: Cholestatic Mechanisms
- Inhibition of bile salt export pump (BSEP/ABCB11) [24]
- Inhibition of other canalicular transporters (MDR3, MRP2)
- Bile acid accumulation → direct toxicity
- Immune-mediated bile duct injury (vanishing bile duct syndrome)
Phase 6: Mitochondrial Injury [21]
- Inhibition of mitochondrial β-oxidation: valproate, aspirin (Reye syndrome)
- Inhibition of electron transport chain: linezolid (complex IV)
- Depletion of mitochondrial DNA: nucleoside reverse transcriptase inhibitors (NRTIs)
- Uncoupling of oxidative phosphorylation
- Results in microvesicular steatosis, lactic acidosis
Characteristics of Idiosyncratic DILI:
- Unpredictable, not strictly dose-dependent (though higher doses increase risk)
- Variable latency (5-90 days typically; can be months)
- Occurs in small minority of exposed individuals (1:10,000-1:100,000)
- Often not reproducible in animal models
- Genetic predisposition (HLA associations)
Injury Patterns by Mechanism
| Pattern | R-Ratio | Pathophysiology | Prototype Drugs | Prognosis |
|---|---|---|---|---|
| Hepatocellular | R ≥5 | Hepatocyte necrosis/apoptosis | Paracetamol, isoniazid, diclofenac | Higher ALF risk; better resolution if survive |
| Cholestatic | R ≤2 | Bile duct injury, BSEP inhibition | Amoxicillin-clavulanate, chlorpromazine | Lower ALF risk; higher chronicity risk |
| Mixed | R 2-5 | Combination mechanisms | Phenytoin, sulfonamides | Intermediate outcomes |
| Steatotic | Variable | Mitochondrial β-oxidation inhibition | Valproate, amiodarone, methotrexate | Chronic injury risk |
R-ratio = (ALT/ULN) ÷ (ALP/ULN)
Progression to Acute Liver Failure
Pathophysiological Sequence:
- Massive hepatocyte necrosis (> 50% functional liver mass)
- Loss of synthetic function: coagulopathy (↓ factor V, VII), hypoalbuminemia
- Loss of metabolic function: hypoglycemia, hyperammonemia
- Hepatic encephalopathy: ammonia, inflammatory cytokines, cerebral edema
- Multisystem organ failure: renal (hepatorenal syndrome), cardiovascular, respiratory
King's College Criteria for Liver Transplantation (Paracetamol): [25]
- pH less than 7.30 after resuscitation, OR
- All three of: INR > 6.5, creatinine > 300 μmol/L, grade III-IV encephalopathy
King's College Criteria (Non-Paracetamol DILI):
- INR > 6.5, OR
- Any three of: age less than 10 or > 40, jaundice > 7 days before encephalopathy, INR > 3.5, bilirubin > 300 μmol/L
4. Clinical Presentation
Spectrum of Presentation
DILI presents across a spectrum from asymptomatic laboratory abnormalities to fulminant hepatic failure. Presentation depends on injury pattern and severity.
Asymptomatic Transaminase Elevation
Features:
- Discovered incidentally on routine blood tests
- No symptoms
- ALT/AST elevation typically less than 5× ULN
- Normal bilirubin and INR
- Common with statins (5-10% of patients) [19]
Significance:
- Most resolve spontaneously or with drug cessation
- Adaptation may occur (transaminases normalize despite continued drug)
- Monitor for progression
Acute Hepatocellular Injury
Symptoms:
- Onset: days to weeks after drug initiation (or hours for paracetamol)
- Nausea, vomiting (90% of paracetamol overdose patients)
- Right upper quadrant pain (50%)
- Fatigue, malaise, anorexia (80%)
- Dark urine (cola-colored) from conjugated hyperbilirubinemia
- Jaundice (30-50% of symptomatic cases)
- Low-grade fever (10-20%)
Signs:
- Jaundice (scleral icterus, skin)
- Hepatomegaly (40-60%): may be tender
- No stigmata of chronic liver disease (acute presentation)
- Signs of hypersensitivity in 10-30%:
- Maculopapular rash
- Fever > 38°C
- Lymphadenopathy
Laboratory Features:
- ALT/AST: typically 500-5000 U/L (paracetamol can exceed 10,000)
- R-ratio ≥5
- Bilirubin: variable; elevation indicates severity
- INR: normal in mild cases; elevation indicates synthetic dysfunction
- Eosinophilia in 30-40% of immune-mediated cases
Cholestatic Pattern
Symptoms:
- Onset: typically 1-8 weeks after drug initiation
- Pruritus (70-90%): often severe, intractable
- Jaundice (60-80%)
- Pale stools, dark urine
- Less prominent systemic symptoms initially
- Fatigue, malaise (moderate)
Signs:
- Jaundice (prominent)
- Excoriations from scratching
- Hepatomegaly (40%)
- No hepatic tenderness typically
Laboratory Features:
- ALP: 3-10× ULN
- GGT: parallels ALP elevation
- R-ratio ≤2
- Bilirubin: elevated (conjugated)
- ALT/AST: less than 3× ULN
- INR: may be mildly elevated (vitamin K malabsorption)
Prolonged Course:
- Cholestatic DILI often takes 3-6 months to resolve [11]
- 10-20% develop chronic cholestasis (> 6 months)
- Risk of vanishing bile duct syndrome (permanent)
Mixed Pattern
Features:
- R-ratio 2-5
- Combination of hepatocellular and cholestatic features
- Examples: phenytoin, sulfonamides
- Variable course and prognosis
DRESS Syndrome (Drug Reaction with Eosinophilia and Systemic Symptoms) [23]
Diagnostic Features:
- Fever > 38°C
- Skin rash (90%): maculopapular, may be exfoliative
- Eosinophilia > 1.5 × 10⁹/L or > 10%
- Systemic involvement:
- Hepatitis (90%)
- Lymphadenopathy (70%)
- Nephritis (10-20%)
- Myocarditis (5%)
- Pneumonitis (5%)
Common Culprits:
- Anticonvulsants: phenytoin, carbamazepine, lamotrigine
- Allopurinol
- Sulfonamides
- Minocycline
- Dapsone
Latency: 2-8 weeks (longer than typical drug allergy)
Management: Immediate drug cessation; systemic corticosteroids (0.5-1 mg/kg prednisolone) [23]
Acute Liver Failure (ALF) Presentation
Diagnostic Criteria:
- INR ≥1.5
- Hepatic encephalopathy (any grade)
- Duration less than 26 weeks in patient without pre-existing cirrhosis
Clinical Features:
- Confusion, drowsiness, altered consciousness (encephalopathy)
- Asterixis (flapping tremor)
- Bleeding: epistaxis, gum bleeding, bruising (coagulopathy)
- Hypoglycemia: sweating, confusion, seizures
- Oliguria, rising creatinine (hepatorenal syndrome)
Grading of Encephalopathy:
- Grade I: Altered mood, sleep disturbance, mild confusion
- Grade II: Drowsiness, inappropriate behavior, asterixis
- Grade III: Stupor, severe confusion, incoherent speech
- Grade IV: Coma, unresponsive
Laboratory Features:
- ALT/AST: often > 1000 U/L initially (may fall as liver fails)
- Bilirubin: > 100 μmol/L (often > 200-300)
- INR: > 1.5 (often > 3-6)
- Albumin: decreased (indicates synthetic failure)
- Glucose: hypoglycemia (less than 4 mmol/L)
- Creatinine: rising (hepatorenal syndrome)
- Lactate: elevated (poor prognosis)
- Phosphate: hypophosphatemia (massive hepatocyte necrosis)
Red Flags Requiring Urgent Action
[!CAUTION] Hy's Law Criteria - High Mortality Marker (10-50%): [5]
- ALT or AST > 3× ULN (hepatocellular injury)
- AND total bilirubin > 2× ULN (> 34 μmol/L if ULN is 17)
- AND no other explanation for hyperbilirubinemia (ALP less than 2× ULN)
- ACTION: Urgent hepatology referral, consider transplant centre transfer
[!CAUTION] Acute Liver Failure Features:
- INR ≥1.5 (coagulopathy)
- Any grade of hepatic encephalopathy
- Hypoglycemia (less than 4 mmol/L)
- Rising creatinine (hepatorenal syndrome)
- ACTION: ICU admission, transplant centre transfer
[!CAUTION] Rapidly Deteriorating Patient:
- Bilirubin rising > 50 μmol/L per day
- INR rising > 0.5 per day
- Declining consciousness
- ACTION: Emergency hepatology consultation
[!CAUTION] DRESS Syndrome:
- Fever + rash + eosinophilia + hepatitis + lymphadenopathy
- Multi-organ involvement
- ACTION: Immediate drug cessation, consider corticosteroids
5. Clinical Examination
Systematic Approach to DILI Examination
A thorough examination identifies severity, pattern, complications, and excludes alternative diagnoses.
General Inspection
Level of Consciousness:
- Alert, oriented (normal)
- Confusion, drowsiness (encephalopathy)
- Stupor, coma (severe encephalopathy)
Jaundice Assessment:
- Scleral icterus (earliest sign, visible > 34 μmol/L)
- Skin jaundice (visible > 50 μmol/L)
- Depth of jaundice correlates with bilirubin level
Nutritional Status:
- Cachexia (chronic liver disease or ALF)
- Obesity (NAFLD as alternative diagnosis)
Rash/Skin Lesions:
- Maculopapular rash (drug hypersensitivity, DRESS)
- Excoriations (cholestatic pruritus)
- Bruising, petechiae (coagulopathy)
- Spider naevi, palmar erythema (chronic liver disease—unlikely in acute DILI)
Hands
Inspection:
- Palmar erythema (chronic liver disease)
- Leukonychia (hypoalbuminemia)
- Clubbing (chronic liver disease, not acute DILI)
- Asterixis/flapping tremor (encephalopathy):
- Ask patient to extend arms, dorsiflex wrists
- Involuntary irregular jerking movements
- Grade I-IV encephalopathy marker
Palpation:
- Capillary refill time (circulatory status)
- Temperature (fever in DRESS or sepsis)
Head and Neck
Eyes:
- Scleral icterus (jaundice)
- Kayser-Fleischer rings (Wilson disease—alternative diagnosis in young patients)
Mouth:
- Fetor hepaticus (sweet, musty breath in ALF)
- Gum bleeding (coagulopathy)
Lymph Nodes:
- Cervical, axillary, inguinal lymphadenopathy (DRESS syndrome, malignancy)
Cardiovascular
Pulse:
- Tachycardia (sepsis, hypovolemia)
Blood Pressure:
- Hypotension (sepsis, ALF with vasodilation)
JVP:
- Elevated (right heart failure, Budd-Chiari syndrome)
Respiratory
Inspection:
- Respiratory rate (encephalopathy, sepsis)
Auscultation:
- Crackles (pulmonary edema, ARDS in ALF)
Abdominal Examination
Inspection:
- Distension (ascites in severe/chronic cases)
- Caput medusae (portal hypertension—chronic)
- Surgical scars
Palpation:
- Liver:
- "Size: normal, enlarged (hepatomegaly in 40-60% of acute DILI), shrunken (massive necrosis in ALF)"
- "Edge: smooth or irregular"
- "Consistency: soft (acute), firm (chronic)"
- "Tenderness: present in acute hepatitis"
- Spleen: splenomegaly suggests portal hypertension (chronic liver disease)
- Kidneys: exclude hepatorenal syndrome
Percussion:
- Liver span: normal 12-15 cm in midclavicular line
- Shifting dullness (ascites)
Auscultation:
- Bowel sounds: normal, hyperactive, absent
- Hepatic bruit (rare: hepatocellular carcinoma, alcoholic hepatitis)
Neurological
Mental Status Examination:
- Orientation to time, place, person
- Attention, concentration (serial 7s subtraction)
- Glasgow Coma Scale in severe cases
Asterixis:
- As above (encephalopathy marker)
Reflexes:
- Hyperreflexia (encephalopathy)
Fundoscopy:
- Papilledema (cerebral edema in ALF—rare)
Lower Limbs
Inspection:
- Peripheral edema (hypoalbuminemia, fluid overload)
Palpation:
- Pitting edema
Examination Summary for Documentation
Example: "Alert, jaundiced patient. No asterixis or encephalopathy. Scleral icterus and generalized jaundice. Tender hepatomegaly 4 cm below costal margin, smooth edge. No ascites, splenomegaly, or stigmata of chronic liver disease. No rash or lymphadenopathy. Cardiovascularly stable."
6. Investigations
Principles of DILI Diagnosis
DILI is a diagnosis of exclusion. Systematic investigation must rule out:
- Viral hepatitis (HAV, HBV, HCV, HEV, EBV, CMV, HSV)
- Autoimmune hepatitis
- Biliary obstruction
- Ischaemic hepatitis
- Wilson disease (age less than 40)
- Budd-Chiari syndrome
- Sepsis-related cholestasis
First-Line Investigations
Bloods:
- Liver Function Tests:
- ALT, AST, ALP, GGT, bilirubin (total and conjugated)
- "Calculate R-ratio: (ALT/ULN) ÷ (ALP/ULN)"
- Synthetic Function:
- INR (coagulopathy, severity marker)
- Albumin (chronic liver disease; normal in acute)
- Full Blood Count:
- Eosinophilia (> 0.5 × 10⁹/L or > 5% suggests immune-mediated DILI) [22]
- Leucocytosis (DRESS, sepsis)
- Thrombocytopenia (chronic liver disease, sepsis)
- Renal Function:
- Urea, creatinine (hepatorenal syndrome)
- Electrolytes (metabolic disturbances)
- Glucose:
- Hypoglycemia in ALF
Paracetamol Level:
- Mandatory in all patients with ALF or suspected overdose
- Time from ingestion crucial for nomogram interpretation [16]
Second-Line Investigations (Exclude Alternative Diagnoses)
Viral Serology:
- Hepatitis A IgM (acute HAV)
- HBsAg, anti-HBc IgM, HBV DNA (acute or chronic HBV)
- Anti-HCV, HCV RNA (HCV)
- Hepatitis E IgM (HEV—underdiagnosed, especially in travelers)
- EBV VCA IgM, CMV IgM (infectious mononucleosis hepatitis)
- HSV PCR (if severe ALF)
Autoimmune Markers:
- Antinuclear antibody (ANA)
- Smooth muscle antibody (SMA)
- Anti-liver-kidney microsomal antibody (anti-LKM)
- Immunoglobulins (IgG elevated in autoimmune hepatitis)
- Note: Some DILI (e.g., minocycline, nitrofurantoin) mimics autoimmune hepatitis with positive autoantibodies
Wilson Disease Screening (if age less than 40):
- Serum caeruloplasmin (less than 200 mg/L suggests Wilson)
- 24-hour urinary copper (> 100 μg/24h abnormal)
- Slit-lamp examination for Kayser-Fleischer rings
Iron Studies:
- Ferritin, transferrin saturation (exclude haemochromatosis)
Pregnancy Test:
- β-hCG in women of childbearing age (acute fatty liver of pregnancy, HELLP syndrome)
Imaging
Ultrasound Abdomen with Doppler:
- Exclude biliary obstruction (gallstones, stricture)
- Liver parenchyma: echogenicity, size
- Hepatic vein and portal vein patency (Budd-Chiari, portal vein thrombosis)
- Ascites
- Splenomegaly (portal hypertension)
CT Abdomen (if indicated):
- Exclude structural lesions (abscess, malignancy)
- Better assessment of biliary tree and pancreas
- Hepatic vein thrombosis
MRI/MRCP (if indicated):
- Biliary tree assessment (MRCP)
- Liver infiltration (steatosis, iron deposition)
Transient Elastography (FibroScan):
- Assess liver stiffness (fibrosis)
- Not routinely required in acute DILI
- Useful in chronic DILI to assess fibrosis
Liver Biopsy
Indications:
- Diagnostic uncertainty (cannot exclude alternative diagnosis)
- Cholestatic DILI not improving after 30 days
- Suspected chronic DILI
- Research purposes
Contraindications:
- INR > 1.5 (correct with vitamin K or FFP)
- Platelets less than 50 × 10⁹/L
- Severe ascites
Histological Patterns in DILI: [26]
- Acute hepatocellular injury:
- "Zone 3 (centrilobular) necrosis: paracetamol, halothane"
- "Diffuse or panlobular hepatitis: isoniazid, phenytoin"
- "Eosinophilic infiltrate: allopurinol, phenytoin"
- Cholestatic injury:
- "Bland cholestasis (bile accumulation, minimal inflammation): anabolic steroids"
- "Cholestatic hepatitis (inflammation + cholestasis): amoxicillin-clavulanate"
- "Ductular reaction, duct injury: chlorpromazine"
- "Vanishing bile duct syndrome: flucloxacillin, amoxicillin-clavulanate (chronic)"
- Steatosis:
- "Macrovesicular: amiodarone, methotrexate"
- "Microvesicular: valproate, aspirin (Reye syndrome)"
- Vascular injury:
- "Sinusoidal obstruction syndrome (veno-occlusive disease): azathioprine, herbal teas"
- "Nodular regenerative hyperplasia: azathioprine"
- Granulomas:
- Allopurinol, sulfonamides, phenytoin
Causality Assessment: RUCAM Score [7]
Roussel Uclaf Causality Assessment Method (updated 2016) is the most validated tool for DILI causality.
Seven Domains:
- Time to onset from drug start:
- Hepatocellular: 5-90 days scores highest
- Cholestatic: 5-90 days scores highest
- First exposure vs. re-exposure
- Course after stopping drug:
- ALT decrease ≥50% within 8 days (hepatocellular) or 180 days (cholestatic)
- Percentage decrease from peak
- Risk factors:
- Alcohol use (hepatocellular DILI)
- Pregnancy (cholestatic DILI)
- Age ≥55 years
- Concomitant drugs:
- Time relationship with other drugs
- Known hepatotoxicity of other drugs
- Search for alternative causes:
- All causes ruled out: +2
- 6-7 causes ruled out: +1
- less than 6 causes ruled out: 0
- Alternative cause highly probable: -3
- Previous hepatotoxicity of drug:
- Reaction labeled in product information: +2
- Published reports but unlabeled: +1
- Unknown: 0
- Response to unintentional re-challenge:
- Positive (ALT elevation > 2× baseline): +3
- Compatible (ALT elevation less than 2×): +1
- Negative: -2
- Not done: 0
Score Interpretation:
- ≤0: Excluded
- 1-2: Unlikely
- 3-5: Possible
- 6-8: Probable
- ≥9: Highly probable
Practical Use:
- Score ≥6 (probable/highly probable) supports DILI diagnosis
- Negative RUCAM (less than 3) should prompt reconsideration
- Re-challenge rarely performed due to safety concerns
Laboratory Interpretation Patterns
| Pattern | ALT | ALP | R-Ratio | Bilirubin | Prognosis |
|---|---|---|---|---|---|
| Hepatocellular | > 5× ULN | less than 2× ULN | ≥5 | Variable | Higher ALF risk; better long-term if survive |
| Cholestatic | less than 2× ULN | > 3× ULN | ≤2 | Often elevated | Lower ALF risk; higher chronicity risk |
| Mixed | > 3× ULN | > 2× ULN | 2-5 | Variable | Intermediate |
| Hy's Law Positive | > 3× ULN | less than 2× ULN | ≥5 | > 2× ULN | 10-50% mortality [5] |
Severity Assessment
Mild:
- ALT/AST less than 5× ULN
- ALP less than 2× ULN
- Normal bilirubin
- Normal INR
Moderate:
- ALT/AST 5-10× ULN
- Bilirubin less than 2× ULN
- INR less than 1.5
Severe:
- ALT/AST > 10× ULN
- Bilirubin > 2× ULN
- INR ≥1.5
- Hy's Law positive
Acute Liver Failure:
- INR ≥1.5
- Hepatic encephalopathy
- Duration less than 26 weeks
7. Management
Principles of DILI Management
- Immediate drug discontinuation is paramount and improves outcomes [27]
- Supportive care is mainstay for most cases
- Specific antidotes for select agents (NAC, L-carnitine)
- Urgent hepatology referral for severe cases
- Transplant evaluation for ALF
- Avoid re-exposure to offending agent
Algorithm: DILI Management
Image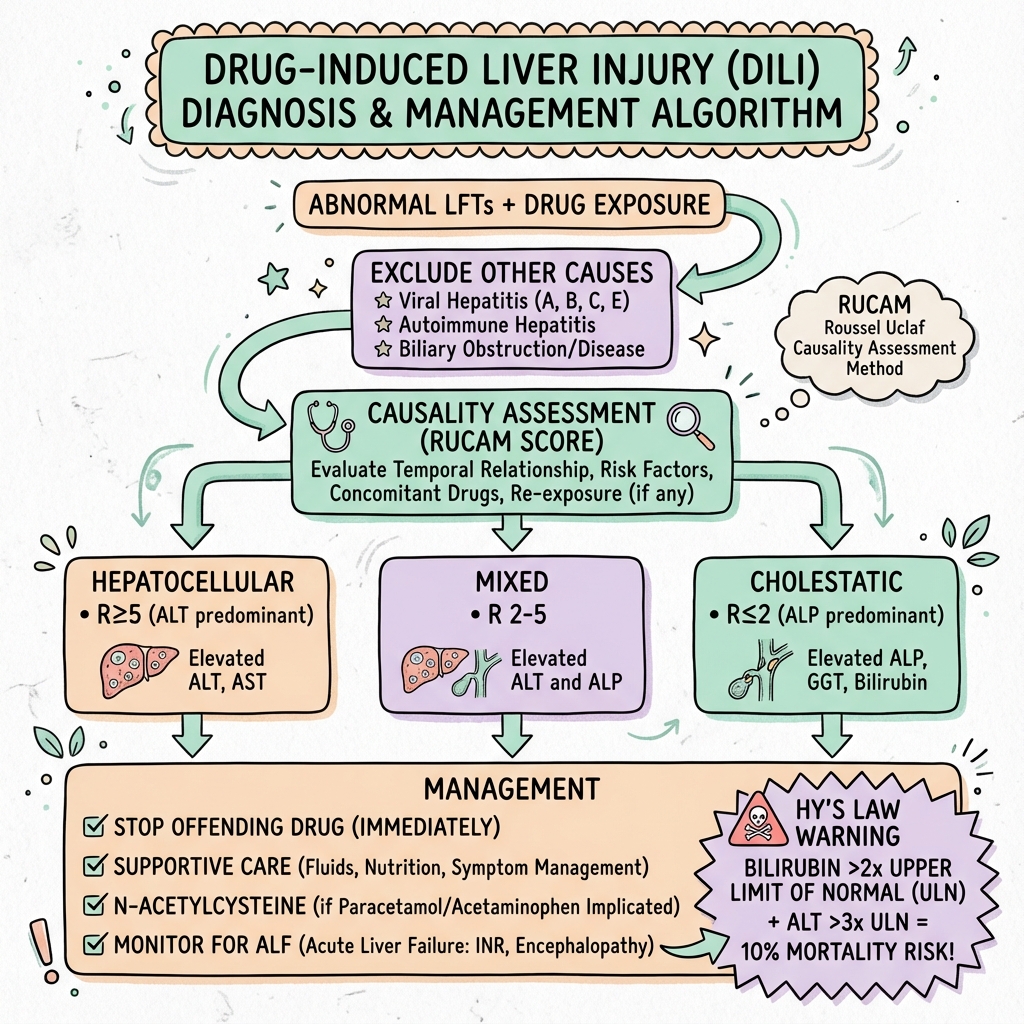 DILI Management Algorithm
DILI Management Algorithm
Step 1: Suspect DILI
- Temporal relationship to drug
- Elevated transaminases/ALP
- Exclude other causes
↓
Step 2: Immediate Actions
- Stop suspected drug(s)
- Check paracetamol level (all ALF cases)
- Assess severity (Hy's Law, INR, encephalopathy)
↓
Step 3: Severity Stratification
Mild (ALT less than 5×, normal INR, no jaundice)
- Outpatient monitoring
- Repeat LFTs weekly
- Patient education
Moderate (ALT 5-10×, bilirubin less than 2×, INR less than 1.5)
- Consider admission
- Daily LFTs, INR
- Hepatology consultation
Severe (Hy's Law, INR ≥1.5, or encephalopathy)
- Hospital admission
- Urgent hepatology referral
- Consider transplant centre transfer
- ICU if ALF features
↓
Step 4: Specific Treatments
- Paracetamol: NAC protocol
- Valproate: L-carnitine
- DRESS: corticosteroids (consider)
- Amanita mushroom: silibinin, NAC
↓
Step 5: Supportive Care
- Monitor LFTs, INR, glucose, renal function
- Nutritional support
- Avoid hepatotoxins (alcohol)
- Treat complications
↓
Step 6: Recovery or Transplant
- Most improve within 1-3 months
- If ALF and meets criteria: transplant
Immediate Management (First Hour)
1. Stop All Suspected Drugs
- Discontinue implicated medication(s) immediately [27]
- Consider polypharmacy: stop non-essential drugs
- Herbal and dietary supplements: STOP
- Only continue truly essential medications (e.g., anticonvulsants, antihypertensives)
2. Assess Severity
- Calculate R-ratio: (ALT/ULN) ÷ (ALP/ULN)
- Check Hy's Law criteria: ALT > 3× + bilirubin > 2× + R ≥5
- INR (synthetic function)
- Mental status (encephalopathy)
- Glucose (hypoglycemia in ALF)
3. Exclude Paracetamol Overdose
- Check paracetamol/acetaminophen level in ALL patients with ALF or acute hepatitis
- If positive or unclear timing: start NAC immediately (do not wait for level)
4. Arrange Urgent Investigations
- LFTs, INR, FBC, U&E, glucose
- Viral serology (HAV IgM, HBsAg, anti-HBc IgM, HCV Ab, HEV IgM)
- Autoimmune markers (ANA, SMA, IgG)
- Ultrasound abdomen with Doppler (exclude biliary obstruction, vascular causes)
5. Urgent Hepatology Referral if:
- Hy's Law positive
- INR ≥1.5
- Any encephalopathy
- Bilirubin > 100 μmol/L
- Rapidly rising LFTs or bilirubin
6. Transplant Centre Notification if:
- ALF criteria met (INR ≥1.5 + encephalopathy)
- Approaching King's College Criteria
Specific Treatments
Paracetamol/Acetaminophen Overdose [16]
N-Acetylcysteine (NAC) Protocol (UK Standard):
- Indications:
- Paracetamol level above treatment line on nomogram
- Staggered overdose (> 4g/day for > 1 day)
- Any paracetamol overdose presenting > 8 hours
- ALF with detectable paracetamol
- Dosing (150 mg/kg regimen, 20 hours 40 minutes):
- "Infusion 1: 150 mg/kg in 200 mL 5% dextrose over 1 hour"
- "Infusion 2: 50 mg/kg in 500 mL 5% dextrose over 4 hours"
- "Infusion 3: 100 mg/kg in 1000 mL 5% dextrose over 16 hours"
- Alternative (US protocol, 21 hours):
- 150 mg/kg over 60 minutes, then 50 mg/kg over 4 hours, then 100 mg/kg over 16 hours
- Mechanism: Restores glutathione; binds NAPQI; improves microcirculation [16]
- Adverse Effects: Anaphylactoid reaction (5-10%): flushing, urticaria, hypotension (slow/stop infusion, antihistamine)
NAC in Non-Paracetamol ALF: [12]
- Randomized trial showed improved transplant-free survival in grade I-II encephalopathy
- No benefit in grade III-IV encephalopathy
- Consider NAC infusion (as above) in severe non-paracetamol DILI with encephalopathy
Valproate-Induced Hepatotoxicity [18]
L-Carnitine:
- Indication: Valproate overdose or suspected valproate hepatotoxicity
- Mechanism: Valproate depletes carnitine → mitochondrial β-oxidation inhibition
- Dosing:
- "IV: 100 mg/kg loading dose (max 6g) over 30 min, then 15 mg/kg every 4 hours"
- "Oral: 50-100 mg/kg/day divided TDS-QDS (for non-severe)"
- Duration: Until clinical improvement, typically 3-7 days
DRESS Syndrome [23]
Corticosteroids (Controversial):
- Indication: Severe DRESS with multi-organ involvement
- Dosing: Prednisolone 0.5-1 mg/kg/day
- Duration: 4-6 weeks with taper
- Evidence: No RCTs; observational data suggests benefit in severe cases
- Caution: May worsen viral reactivation (HHV-6, EBV); exclude sepsis
Immune-Mediated DILI with Autoimmune Features:
- Some cases respond to corticosteroids (e.g., nitrofurantoin-induced AIH-like DILI)
- Empirical trial of prednisolone 30-40 mg daily if severe and autoantibody-positive
- NOT routinely recommended [6]
Mushroom (Amanita) Poisoning:
- Silibinin IV (not widely available)
- NAC
- Penicillin G (controversial)
- Liver transplant if ALF
Ursodeoxycholic Acid (UDCA):
- Indication: Chronic cholestatic DILI (> 30 days)
- Dosing: 13-15 mg/kg/day divided BID-TID
- Mechanism: Cytoprotective; increases bile flow
- Evidence: Limited data; may improve pruritus and biochemistry
Supportive Care
Monitoring:
- Mild DILI: Weekly LFTs, INR until normalization
- Moderate DILI: Daily LFTs, INR, U&E, glucose
- Severe DILI/ALF: 6-12 hourly LFTs, INR, glucose, lactate
Nutrition:
- Maintain caloric intake (1.2-1.5× basal metabolic rate)
- Protein: no restriction unless severe encephalopathy (then 0.5-1 g/kg)
- Enteral nutrition preferred over parenteral
- Vitamins: thiamine, folate supplementation
Avoid Hepatotoxins:
- Alcohol: absolute avoidance
- Paracetamol: avoid unless essential (use lowest dose if required)
- Other hepatotoxic drugs
Pruritus Management (Cholestatic DILI):
- First-line: Colestyramine 4-8g BID (binds bile acids)
- Second-line: Rifampicin 150-300 mg daily
- Third-line: Naltrexone 25-50 mg daily (opioid antagonist)
- Antihistamines (chlorpheniramine): modest benefit, sedation helpful
Vitamin K:
- If INR elevated and cholestasis: vitamin K 10 mg IV/PO daily for 3 days
- If no response: coagulopathy due to synthetic failure (not malabsorption)
Complications Management:
Coagulopathy:
- INR less than 1.5: no intervention unless bleeding or procedure
- INR 1.5-2.5: vitamin K trial
- INR > 2.5 or bleeding: Fresh frozen plasma (FFP)
- Avoid overcorrection (masks progression)
Encephalopathy:
- Lactulose 15-30 mL TDS (target 2-3 soft stools/day)
- Rifaximin 550 mg BID (non-absorbable antibiotic)
- Exclude sepsis, hypoglycemia, drug effects
- Avoid sedatives
Hypoglycemia:
- Glucose monitoring 4-6 hourly
- 10% dextrose infusion if recurrent hypoglycemia
- 50 mL 50% dextrose IV bolus if less than 3 mmol/L
Hepatorenal Syndrome:
- Fluid resuscitate cautiously
- Avoid nephrotoxins (NSAIDs, gentamicin, contrast)
- Terlipressin + albumin (specialist use)
- Renal replacement therapy if indicated
Cerebral Edema (ALF):
- Head elevation 30°
- Avoid hypotension, hyponatremia, hypoxia
- Mannitol 0.5-1 g/kg IV bolus (if ICP monitoring available and raised)
- Hypertonic saline (controversial)
Disposition and Referral
Outpatient Management (Mild DILI):
- ALT less than 5× ULN
- Normal bilirubin and INR
- No symptoms or mild symptoms only
- Reliable patient
- Weekly LFTs until normalization
- Patient education on red flags
Hospital Admission:
- ALT ≥5× ULN
- Bilirubin ≥2× ULN
- INR ≥1.3
- Symptomatic (jaundice, nausea, abdominal pain)
- Hy's Law positive
ICU Admission:
- INR ≥2.0
- Encephalopathy (any grade)
- Hypoglycemia
- Hypotension
- Hypoxia
Transplant Centre Referral:
- ALF criteria met (INR ≥1.5 + encephalopathy)
- Approaching King's College Criteria:
- "Paracetamol: pH less than 7.30, or INR > 6.5 + creatinine > 300 + grade III-IV encephalopathy"
- "Non-paracetamol: INR > 6.5, or 3 of: age less than 10 or > 40, jaundice > 7d before encephalopathy, INR > 3.5, bilirubin > 300 μmol/L"
- Rapidly deteriorating despite maximal therapy
Liver Transplantation [28]
Indications:
- ALF meeting King's College Criteria
- Failure to improve with medical therapy
- Chronic DILI with decompensated cirrhosis (rare)
Outcomes:
- 1-year survival post-transplant for DILI-related ALF: 60-80%
- Lower than other causes of ALF (due to older age, comorbidities)
Drug Rechallenge
Generally Avoided:
- Risk of rapid severe hepatotoxicity upon re-exposure (within 24 hours) [29]
Absolute Contraindications to Rechallenge:
- Previous Hy's Law positive reaction
- ALF or severe DILI
- DRESS or hypersensitivity features
- Chronic DILI (> 6 months)
May Consider Rechallenge if:
- Drug essential with no alternative
- Previous reaction mild (ALT less than 5×, no jaundice, no symptoms)
- Patient informed and consents
- Close monitoring feasible
- RUCAM score low (less than 3, unlikely causality)
Rechallenge Protocol (if undertaken):
- Inpatient or very close outpatient monitoring
- Baseline LFTs, INR
- Start low dose
- LFTs and INR at days 1, 3, 7, 14, 30
- Stop immediately if ALT > 2× baseline
Alternative Approach:
- Use different drug from same class (e.g., different statin if previous statin DILI was mild)
- Evidence of cross-reactivity varies by class
8. Complications
Acute Complications
| Complication | Incidence | Mechanism | Management |
|---|---|---|---|
| Acute Liver Failure | 1-5% of DILI [9] | Massive hepatocyte necrosis > 50% functional mass | ICU, transplant evaluation, supportive care, NAC |
| Coagulopathy | 5-15% (severe DILI) | Reduced synthesis of clotting factors (II, V, VII, IX, X) | Vitamin K, FFP if bleeding or INR > 2.5 |
| Hepatic Encephalopathy | 1-3% (ALF cases) | Hyperammonemia, inflammatory cytokines, cerebral edema | Lactulose, rifaximin, avoid sedatives, transplant |
| Hypoglycemia | 10-20% (ALF) | Reduced gluconeogenesis, glycogen depletion | Dextrose infusion, frequent monitoring |
| Hepatorenal Syndrome | 5-10% (ALF) | Splanchnic vasodilation, renal vasoconstriction | Terlipressin + albumin, avoid nephrotoxins, RRT |
| Cerebral Edema | 5-25% (ALF with grade III-IV encephalopathy) | Astrocyte swelling from ammonia, cytokines | Mannitol, hypertonic saline, head elevation, transplant |
| Sepsis | 20-40% (ALF) | Immune dysfunction, gut translocation | Broad-spectrum antibiotics, antifungals |
| Multiorgan Failure | 10-30% (ALF) | Systemic inflammatory response, circulatory failure | ICU supportive care, transplant |
| Death (Acute) | 5-12% overall DILI; 10-50% Hy's Law cases [5,9] | ALF, sepsis, multiorgan failure | Prevention through early recognition and drug cessation |
Chronic Complications
| Complication | Incidence | Mechanism | Management |
|---|---|---|---|
| Chronic Cholestasis | 10-20% of cholestatic DILI [11] | Persistent bile duct injury, ductopenia | UDCA, manage pruritus, monitor LFTs |
| Vanishing Bile Duct Syndrome | 1-2% of cholestatic DILI | Immune-mediated bile duct destruction, ductopenia | UDCA, transplant if progressive liver failure |
| Chronic Hepatitis | 5-10% of hepatocellular DILI [11] | Persistent inflammation, autoimmune features | Monitor LFTs, consider corticosteroids if AIH-like |
| Cirrhosis | 1-5% [11] | Chronic injury → fibrosis → cirrhosis | Manage as cirrhosis; transplant evaluation if decompensated |
| Portal Hypertension | 1-2% | Cirrhosis or nodular regenerative hyperplasia | Beta-blockers, variceal screening, transplant evaluation |
| Hepatocellular Carcinoma | less than 1% | Cirrhosis-related | HCC surveillance (USS + AFP 6-monthly) |
Definitions of Chronicity
- Chronic DILI: Biochemical or histological abnormalities persisting > 6 months after drug cessation [11]
- Prolonged DILI: Abnormalities 3-6 months
- Acute DILI: Resolution less than 3 months
Risk Factors for Chronicity [11]
Patient Factors:
- Older age
- Female sex
- Pre-existing liver disease
DILI Characteristics:
- Cholestatic or mixed pattern (R less than 5)
- Higher ALP at presentation (> 3× ULN)
- Continued drug exposure after onset (delayed cessation)
- Development of jaundice
Specific Drugs:
- Amoxicillin-clavulanate: 15-20% chronicity
- Flucloxacillin: ductopenia in 10%
- Nitrofurantoin: chronic active hepatitis in 20%
Long-Term Outcomes (DILIN Prospective Registry) [9,11]
At 6 Months Post-Onset:
- Complete biochemical resolution: 69%
- Ongoing abnormalities: 17%
- Death or transplantation: 9%
- Lost to follow-up: 5%
Chronic DILI (> 6 months):
- Persistent abnormalities: 14-20%
- Cirrhosis develops in small subset (1-5%)
9. Prognosis
Overall Outcomes
Most Cases (80-85%):
- Resolution within 1-3 months of drug cessation [9]
- Full biochemical recovery
- No long-term sequelae
Severe Cases (10-15%):
- Prolonged course (3-6 months)
- May develop chronic injury
- Risk of chronicity 10-20%
Critical Cases (5-10%):
- Acute liver failure
- Death or transplantation
- Mortality 5-12% overall DILI [9]
Prognostic Factors
Good Prognosis:
- Early drug discontinuation (less than 1 week after symptom onset) [27]
- Mild elevation (ALT less than 5× ULN)
- No jaundice (bilirubin normal)
- INR normal
- Cholestatic pattern without ductopenia
- Younger age (less than 40 years)
- RUCAM score low (alternative diagnosis more likely)
Poor Prognosis:
- Hy's Law positive: 10-50% mortality [5]
- Hepatocellular pattern with jaundice (R ≥5 + bilirubin > 2× ULN)
- INR ≥1.5
- Encephalopathy (any grade)
- Delayed drug cessation (> 1 week after symptom onset)
- Age > 60 years [20]
- Bilirubin > 100 μmol/L
- Rising bilirubin despite drug cessation
- Renal impairment (hepatorenal syndrome)
- Lactate > 3.5 mmol/L (poor prognosis in ALF)
Predictive Models
Hy's Law (FDA Predictor) [5]
- ALT > 3× ULN + bilirubin > 2× ULN + ALP less than 2× ULN
- Predicts 10-50% mortality or transplantation
- Originally derived from drug trials
- Exceptions: cases with alternative explanation for hyperbilirubinemia (e.g., hemolysis, Gilbert syndrome)
King's College Criteria (Transplant Prediction) [25]
Paracetamol-Induced ALF:
- Arterial pH less than 7.30 (after resuscitation), OR
- All three of:
- INR > 6.5
- Creatinine > 300 μmol/L (3.4 mg/dL)
- Grade III-IV encephalopathy
- Sensitivity 69%, specificity 92% for predicting death without transplant
Non-Paracetamol DILI ALF:
- INR > 6.5 (regardless of encephalopathy), OR
- Any three of:
- Age less than 10 or > 40 years
- "Etiology: non-A, non-B hepatitis, halothane, idiosyncratic drug reaction"
- Jaundice > 7 days before encephalopathy
- INR > 3.5
- Bilirubin > 300 μmol/L (17.5 mg/dL)
- Sensitivity 58%, specificity 74%
Model for End-Stage Liver Disease (MELD) Score:
- Predicts 3-month mortality in chronic liver disease
- Less validated in acute DILI but used for transplant prioritization
- Formula: 3.78×ln[bilirubin (mg/dL)] + 11.2×ln[INR] + 9.57×ln[creatinine (mg/dL)] + 6.43
- MELD > 30: high mortality risk
Naranjo DILI-Specific Severity Index:
- Grade 1 (Mild): ALT elevation, no symptoms
- Grade 2 (Moderate): Symptoms, no hospitalization needed
- Grade 3 (Severe): Hospitalization, no liver failure
- Grade 4 (Very Severe): Liver failure (INR ≥1.5)
- Grade 5 (Fatal): Death or transplantation
Pattern-Specific Prognosis
| Pattern | Short-Term (ALF Risk) | Long-Term (Chronicity Risk) | Mortality |
|---|---|---|---|
| Hepatocellular (R ≥5) | Higher (5-10%) [9] | Lower (5-10%) | Higher if jaundice develops |
| Cholestatic (R ≤2) | Lower (1-3%) | Higher (15-25%) [11] | Lower acutely; chronicity complications |
| Mixed (R 2-5) | Intermediate | Intermediate | Variable |
Specific Drug Outcomes
| Drug | Pattern | ALF Risk | Chronicity Risk | Notes |
|---|---|---|---|---|
| Paracetamol | Hepatocellular | High if late presentation | Rare (excellent recovery with NAC) | Leading cause of ALF [1,2] |
| Isoniazid | Hepatocellular | Moderate (1-2%) [17] | Low | Risk increases with age, alcohol |
| Amoxicillin-clavulanate | Cholestatic/Mixed | Low-moderate | Moderate (15-20%) [11] | Most common idiosyncratic DILI [10] |
| Flucloxacillin | Cholestatic | Low | Moderate-high (ductopenia risk) | Delayed onset (1-6 weeks post-cessation) |
| Diclofenac | Hepatocellular | Moderate | Low | Higher risk than other NSAIDs |
| Valproate | Hepatocellular | Moderate | Low with L-carnitine | Mitochondrial toxicity [18] |
| Statins | Hepatocellular (mild) | Very low | Very low [19] | Usually asymptomatic ALT elevation |
| Green tea extract | Hepatocellular | Low-moderate | Low | Dose-dependent [13] |
Long-Term Follow-Up
Acute DILI (Resolved less than 3 months):
- No routine follow-up needed after normalization
- Patient education on avoidance of implicated drug
Prolonged DILI (3-6 months):
- LFTs every 4-6 weeks until normalization
- Hepatology follow-up
Chronic DILI (> 6 months):
- Specialist hepatology follow-up
- LFTs every 3 months
- Consider liver biopsy (assess fibrosis)
- Transient elastography annually (fibrosis monitoring)
- If cirrhosis develops: HCC surveillance (USS + AFP 6-monthly), variceal screening
10. Prevention
Primary Prevention (Pre-Exposure)
Pharmacogenomic Screening:
- HLA-B*57:01 testing before abacavir (prevents hypersensitivity) [30]
- HLA-B*15:02 testing (Asian ancestry) before carbamazepine (prevents Stevens-Johnson syndrome)
- Future: HLA-B*57:01 before flucloxacillin? (cost-effectiveness unclear) [14]
Risk Stratification:
- Baseline LFTs before starting high-risk drugs:
- Isoniazid (repeat at 2, 4, 8, 12 weeks) [17]
- Methotrexate (repeat monthly)
- Azathioprine (repeat at 4 weeks, then 3-monthly)
- Avoid high-risk drugs in pre-existing liver disease (adjust dose or choose alternative)
Patient Education:
- Explain DILI risk when prescribing high-risk medications
- Provide written information on symptoms (jaundice, dark urine, fatigue)
- Advise immediate cessation and medical review if symptoms develop
Drug Selection:
- Choose drugs with lower hepatotoxicity risk when alternatives available
- Example: Use ibuprofen instead of diclofenac (lower DILI risk)
Secondary Prevention (Post-Exposure Recognition)
Monitoring High-Risk Drugs:
- Regular LFT monitoring (isoniazid, methotrexate, azathioprine)
- Educate patients on symptoms
- Low threshold for checking LFTs if symptoms
Early Recognition:
- Clinical awareness of DILI as differential for any acute hepatitis
- Calculate R-ratio and check Hy's Law in all cases
- Systematic exclusion of other causes
Immediate Withdrawal: [27]
- Stop drug as soon as DILI suspected
- Do not wait for causality assessment completion
- Early cessation significantly improves outcomes
Tertiary Prevention (Prevent Recurrence)
Drug Avoidance:
- Document DILI in medical records prominently
- Allergy alert in electronic health records
- Patient allergy card/bracelet for severe reactions
Cross-Reactivity Avoidance:
- Avoid rechallenge with same drug (see Management section)
- Caution with structurally related drugs (e.g., other phenothiazines if chlorpromazine DILI)
Alternative Therapies:
- Identify safer alternatives for indication
- Multidisciplinary discussion if drug essential
Reporting:
- Report to pharmacovigilance authorities:
- "UK: Yellow Card Scheme (MHRA)"
- "US: MedWatch (FDA)"
- "Australia: TGA"
- Contribute to DILI registries (DILIN, EUDILI)
11. Evidence and Guidelines
Key Guidelines
European Association for the Study of the Liver (EASL) Clinical Practice Guidelines: Drug-Induced Liver Injury (2019) [6]
- Comprehensive guideline covering diagnosis, causality, management
- RUCAM score recommended for causality assessment
- Immediate drug withdrawal emphasized
- NAC considered for severe non-paracetamol DILI
- Corticosteroids not routinely recommended
- Available: https://pubmed.ncbi.nlm.nih.gov/30926241/
American College of Gastroenterology (ACG) Clinical Guideline: Diagnosis and Management of Idiosyncratic Drug-Induced Liver Injury (2021) [31]
- Updated US guideline
- Strong recommendation for drug cessation
- Conditional recommendation for NAC in non-paracetamol ALF
- Liver transplantation for ALF meeting criteria
- Available: https://pubmed.ncbi.nlm.nih.gov/33929376/
FDA Guidance: Drug-Induced Liver Injury: Premarketing Clinical Evaluation (2009)
- Defines Hy's Law as safety signal in drug development
- Thresholds for hepatotoxicity concerns in trials
- Influenced regulatory drug withdrawals
Key Studies and Evidence
1. Epidemiology and Causation
Björnsson ES et al. "Incidence, Presentation, and Outcomes in Patients with Drug-Induced Liver Injury in the General Population of Iceland." Gastroenterology. 2013;144(7):1419-1425. [8]
- Population-based prospective study
- Incidence: 19.1 per 100,000 inhabitants per year
- Antimicrobials most common cause (46%)
- Mortality 2.7%
Reuben A et al. "Drug-Induced Acute Liver Failure: Results of a US Multicenter, Prospective Study." Hepatology. 2010;52(6):2065-2076. [2]
- DILIN registry: 1198 DILI cases, 133 developed ALF
- DILI accounts for 13% of all ALF cases (excluding paracetamol)
- Mortality/transplant rate 29% in non-paracetamol DILI-ALF
Larson AM et al. "Acetaminophen-Induced Acute Liver Failure: Results of a United States Multicenter, Prospective Study." Hepatology. 2005;42(6):1364-1372. [1]
- 662 ALF cases analyzed; paracetamol caused 46%
- Unintentional overdose in 48% (therapeutic misadventure)
- Overall survival 67% (28% transplanted)
2. Mechanisms and Pathophysiology
Kaplowitz N. "Idiosyncratic Drug Hepatotoxicity." Nat Rev Drug Discov. 2005;4(6):489-499. [15]
- Comprehensive review of idiosyncratic DILI mechanisms
- Two-hit hypothesis: drug bioactivation + immune response
- Role of mitochondrial injury
Chalasani N et al. "Features and Outcomes of 899 Patients with Drug-Induced Liver Injury: The DILIN Prospective Study." Gastroenterology. 2015;148(7):1340-1352. [9]
- Largest prospective DILI cohort (899 patients)
- Chronic DILI in 14% at 6 months
- Mortality/transplant 9% at 6 months
- Jaundice at presentation associated with poor outcomes
3. Genetic Susceptibility
Daly AK et al. "HLA-B*5701 Genotype is a Major Determinant of Drug-Induced Liver Injury Due to Flucloxacillin." Nat Genet. 2009;41(7):816-819. [14]
- Genome-wide association study
- HLA-B*57:01 odds ratio 80.6 for flucloxacillin DILI
- High specificity (99.5%) but low positive predictive value (0.12%)
4. Causality Assessment
Danan G, Teschke R. "RUCAM in Drug and Herb Induced Liver Injury: The Update." Int J Mol Sci. 2016;17(1):14. [7]
- Updated RUCAM score (from 1993 original)
- Most widely validated causality assessment method
- Sensitivity 86%, specificity 89% for probable/highly probable DILI
5. Management and Outcomes
Lee WM et al. "Intravenous N-Acetylcysteine Improves Transplant-Free Survival in Early Stage Non-Acetaminophen Acute Liver Failure." Gastroenterology. 2009;137(3):856-864. [12]
- Randomized controlled trial
- NAC improved transplant-free survival in grade I-II encephalopathy (52% vs 30%, p=0.043)
- No benefit in grade III-IV encephalopathy
Fontana RJ et al. "Pathobiology of Idiosyncratic Drug-Induced Liver Injury: Mechanisms and Pathways." Clin Liver Dis. 2020;15(Suppl 1):S7-S12. [22]
- Review of immune-mediated DILI
- Adaptive immune response central to idiosyncratic DILI
- Eosinophilia marker of immune involvement
Chalasani N et al. "Relationship Between Hy's Law and Mortality or Liver Transplantation in Patients with Drug-Induced Liver Injury." Hepatology. 2021;73(6):2527-2536. [5]
- Validation of Hy's Law in DILIN cohort
- Hy's Law-positive cases: 48% mortality/transplant vs 3.8% in Hy's Law-negative
- Strongly predictive of poor outcomes
6. Chronic DILI
Fontana RJ et al. "Factors That Determine the Development and Progression of Chronic Liver Injury After Drug-Induced Liver Injury." Gastroenterology. 2021;161(5):1510-1523. [11]
- 184 chronic DILI cases (> 6 months)
- Cholestatic/mixed pattern at presentation associated with chronicity (OR 3.6)
- 20% developed cirrhosis; 3% required transplantation
7. Specific Drugs
Andrade RJ et al. "Drug-Induced Liver Injury: An Analysis of 461 Incidences Submitted to the Spanish Registry over a 10-Year Period." Gastroenterology. 2005;129(2):512-521. [10]
- Amoxicillin-clavulanate most common cause (13%)
- NSAIDs second (10%)
- Hepatocellular pattern 59%, cholestatic 21%, mixed 20%
Saukkonen JJ et al. "An Official ATS Statement: Hepatotoxicity of Antituberculosis Therapy." Am J Respir Crit Care Med. 2006;174(8):935-952. [17]
- Isoniazid hepatotoxicity: 10-20% mild ALT elevation, 1% severe
- Risk factors: age > 35, alcohol, HIV, pre-existing liver disease
- Monthly LFT monitoring recommended
Björnsson ES et al. "Hepatotoxicity Associated with Statins: Reports of Idiosyncratic Liver Injury Post-Marketing." Am J Gastroenterol. 2012;107(6):908-917. [19]
- Statin-induced hepatotoxicity rare (1.2 per 100,000 users)
- Atorvastatin and simvastatin most commonly implicated
- Routine LFT monitoring not recommended
Navarro VJ et al. "Liver Injury from Herbal and Dietary Supplements." Hepatology. 2017;65(1):363-373. [13]
- HDS-DILI increasing: 16-20% of US DILI cases
- Bodybuilding supplements, green tea extract, multi-ingredient products
- Often mislabeled or contaminated
8. Mitochondrial Toxicity
Begriche K et al. "Drug-Induced Toxicity on Mitochondria and Lipid Metabolism: Mechanistic Diversity and Deleterious Consequences for the Liver." J Hepatol. 2011;54(4):773-794. [21]
- Mitochondrial dysfunction central to many DILI cases
- β-oxidation inhibition, respiratory chain impairment, mtDNA depletion
- Leads to steatosis, lactic acidosis, hepatocyte death
9. Cholestatic DILI
Padda MS et al. "Drug-Induced Cholestasis." Hepatology. 2011;53(4):1377-1387. [24]
- BSEP inhibition key mechanism
- Bile acid accumulation → toxicity
- Genetic polymorphisms in ABCB11 increase susceptibility
10. DRESS Syndrome
Husain Z et al. "DRESS Syndrome: Part I. Clinical Perspectives." J Am Acad Dermatol. 2013;68(5):693.e1-14. [23]
- DRESS diagnostic criteria and management review
- Latency 2-8 weeks (longer than typical drug allergy)
- Systemic corticosteroids mainstay of treatment
11. Liver Transplantation
Bernal W et al. "Acute Liver Failure." Lancet. 2010;376(9736):190-201. [25]
- Review of ALF including DILI
- King's College Criteria for transplant listing
- Outcomes and management strategies
O'Grady JG et al. "Early Indicators of Prognosis in Fulminant Hepatic Failure." Gastroenterology. 1989;97(2):439-445. [Original King's College Criteria]
- Landmark study deriving prognostic criteria
- Paracetamol vs. non-paracetamol different criteria
12. Pharmacovigilance
Sgro C et al. "Incidence of Drug-Induced Hepatic Injuries: A French Population-Based Study." Hepatology. 2002;36(2):451-455.
- Population-based study: 13.9 per 100,000 incidence
- Underreporting significant issue
12. Patient Explanation
What is Drug-Induced Liver Injury (DILI)?
Your liver processes medications and removes them from your body. Sometimes, medications can damage the liver. This is called drug-induced liver injury, or DILI.
How Common Is It?
DILI is relatively uncommon (about 1-2 in 10,000 people taking medications), but it is one of the most common causes of sudden liver problems in developed countries.
What Medications Cause It?
Almost any medication can potentially cause liver injury, but some are more likely culprits:
- Paracetamol (acetaminophen) in overdose
- Antibiotics (especially co-amoxiclav/Augmentin)
- Anti-inflammatory painkillers (like diclofenac)
- Herbal supplements and traditional medicines
- Tuberculosis medications
- Seizure medications
What Are the Symptoms?
Many people have no symptoms initially. When symptoms occur, they may include:
- Yellowing of the skin or eyes (jaundice)
- Dark urine (tea or cola-colored)
- Pale stools
- Severe tiredness
- Loss of appetite, nausea
- Itching (especially with some antibiotics)
- Abdominal pain (right side, upper)
How Is It Diagnosed?
Blood tests show liver inflammation. Your doctor will:
- Review all your medications (including supplements and herbal remedies)
- Perform blood tests to measure liver enzymes
- Do tests to rule out other causes (viral hepatitis, gallstones, etc.)
- Use ultrasound to look at your liver
What Is the Treatment?
The most important treatment is stopping the medication that caused the problem. For most people, the liver recovers on its own within 1-3 months.
For paracetamol overdose, there is a specific antidote (N-acetylcysteine) that works very well if given early.
For other medications, treatment is supportive care and monitoring.
How Serious Is It?
- Most cases (80-85%): The liver fully recovers after stopping the medication.
- Some cases (10-15%): Recovery takes longer (3-6 months) or becomes chronic.
- Rare severe cases (5%): Can cause acute liver failure requiring intensive care or liver transplantation.
What Should I Do?
-
Tell your doctor about ALL medications you take - including vitamins, supplements, herbal remedies, and traditional medicines. Many people don't realize these can cause liver problems.
-
Watch for warning symptoms after starting a new medication:
- Yellowing of skin or eyes
- Very dark urine
- Severe tiredness
- Nausea or loss of appetite
-
Seek urgent medical attention if you develop these symptoms.
-
Do not restart the medication without discussing with your doctor, even if you feel better.
-
Avoid alcohol while your liver is recovering.
Can I Prevent It?
- Follow medication instructions carefully (especially paracetamol dosing - maximum 4g per day for adults).
- Have blood tests when recommended by your doctor for certain medications.
- Be cautious with herbal supplements and "natural" remedies - natural does not always mean safe.
- Inform your doctor if you have liver disease before starting new medications.
Will I Need Follow-Up?
Your doctor will monitor your blood tests until they return to normal. If you had severe DILI, you may need specialist follow-up with a liver doctor (hepatologist).
Can I Take the Medication Again?
Usually no. Re-exposure to the medication can cause rapid, severe liver damage. Your medical records should be updated to avoid this medication in future. If the medication is essential, your doctor will discuss the risks and benefits with you.
13. Examination Focus
MRCP/FRACP Viva Points
Opening Statement (30 seconds): "Drug-induced liver injury is liver damage caused by medications, herbal products, or dietary supplements. It is the commonest cause of acute liver failure in developed countries, accounting for over 50% of cases. DILI is classified as intrinsic—dose-dependent and predictable like paracetamol—or idiosyncratic—unpredictable and immune-mediated. Pattern classification uses the R-ratio: hepatocellular if R ≥5, cholestatic if R ≤2, mixed if R 2-5, where R equals ALT over ULN divided by ALP over ULN. Hy's Law predicts severe outcomes: ALT > 3× ULN plus bilirubin > 2× ULN without cholestasis carries 10-50% mortality. Management centres on immediate drug withdrawal, supportive care, and specific antidotes where available. Diagnosis is one of exclusion after ruling out viral, autoimmune, and biliary causes." [1,2,4,5,6]
Core Knowledge Areas
1. Definition and Classification
- DILI: liver injury from drugs, herbs, supplements
- Intrinsic (predictable, dose-dependent) vs. idiosyncratic (unpredictable, immune/metabolic)
- Pattern: hepatocellular (R ≥5), cholestatic (R ≤2), mixed (R 2-5)
2. Epidemiology
- Incidence: 14-19 per 100,000 per year [8]
- Most common cause of ALF in UK/US (46-52%) [1,2]
- Common drugs: paracetamol, amoxicillin-clavulanate, isoniazid, diclofenac, statins
3. Pathophysiology
- Intrinsic: paracetamol → NAPQI → glutathione depletion → hepatocyte necrosis [16]
- Idiosyncratic: drug-protein adducts → HLA-restricted immune response [14,22]
- Mitochondrial toxicity: valproate, NRTIs [21]
- Cholestasis: BSEP inhibition, bile duct injury [24]
4. Clinical Presentation
- Asymptomatic transaminitis to fulminant ALF
- Symptoms: jaundice, RUQ pain, fatigue, pruritus (cholestatic)
- DRESS: fever, rash, eosinophilia, systemic involvement [23]
5. Hy's Law (Critical Concept)
- ALT > 3× ULN + bilirubin > 2× ULN + no cholestasis (ALP less than 2×)
- Predicts 10-50% mortality/transplant [5]
- Urgent hepatology referral mandatory
6. Investigations
- Diagnosis of exclusion: viral (HAV, HBV, HCV, HEV), autoimmune (ANA, SMA), biliary (USS)
- RUCAM score for causality (≥6 probable/highly probable) [7]
- Calculate R-ratio: (ALT/ULN) ÷ (ALP/ULN)
7. Management
- Immediate drug cessation (improves outcomes) [27]
- NAC for paracetamol (and consideration in non-paracetamol ALF) [12,16]
- L-carnitine for valproate [18]
- Supportive care; transplant evaluation if ALF
8. Prognosis
- 80-85% resolve within 1-3 months [9]
- Chronic DILI (> 6 months): 14-20%, more common with cholestatic pattern [11]
- Mortality: 5-12% overall; 10-50% if Hy's Law positive [5,9]
Key Equations and Formulas
R-Ratio:
R = (ALT / ULN for ALT) ÷ (ALP / ULN for ALP)
Interpretation:
R ≥5 → Hepatocellular pattern
R ≤2 → Cholestatic pattern
R 2-5 → Mixed pattern
Hy's Law Criteria (All Must Be Present):
- ALT or AST > 3× ULN
- Total bilirubin > 2× ULN (or > 34 μmol/L if ULN is 17)
- No other explanation for hyperbilirubinemia (i.e., ALP less than 2× ULN)
King's College Criteria (Paracetamol ALF):
- pH less than 7.30 (after resuscitation), OR
- All three: INR > 6.5 + creatinine > 300 μmol/L + grade III-IV encephalopathy
King's College Criteria (Non-Paracetamol ALF):
- INR > 6.5, OR
- 3 of 5: age less than 10 or > 40, jaundice > 7d before encephalopathy, INR > 3.5, bilirubin > 300 μmol/L, specific etiology
MELD Score:
MELD = 3.78×ln[bilirubin (mg/dL)] + 11.2×ln[INR] + 9.57×ln[creatinine (mg/dL)] + 6.43
Examination Scenarios
Scenario 1: Hepatocellular DILI with Jaundice (Hy's Law)
- Patient on isoniazid for TB, presents with jaundice
- ALT 800 U/L (ULN 40), ALP 150 U/L (ULN 120), bilirubin 100 μmol/L (ULN 17)
- Calculate R-ratio: (800/40) ÷ (150/120) = 20 ÷ 1.25 = 16 → Hepatocellular
- Hy's Law: ALT > 3× (yes, 20×) + bilirubin > 2× (yes, 5.9×) + R ≥5 (yes, 16) → Hy's Law POSITIVE
- Action: Immediate cessation, urgent hepatology referral, check INR, monitor for ALF
Scenario 2: Cholestatic DILI
- Patient completed flucloxacillin 2 weeks ago, now jaundiced with severe itch
- ALT 90 U/L (ULN 40), ALP 600 U/L (ULN 120), bilirubin 80 μmol/L
- R-ratio: (90/40) ÷ (600/120) = 2.25 ÷ 5 = 0.45 → Cholestatic
- Hy's Law: No (R less than 5)
- Management: Drug already stopped, UDCA for symptom relief, monitor (may take 3-6 months to resolve)
Scenario 3: Paracetamol Overdose
- 30-year-old, 10g paracetamol ingestion 6 hours ago
- ALT 3000 U/L, INR 1.8, bilirubin normal, no encephalopathy
- Action: NAC immediately (150 mg/kg over 1h, then 50 mg/kg over 4h, then 100 mg/kg over 16h), monitor INR/glucose/lactate, hepatology referral
Common Viva Questions and Model Answers
Q: What is Hy's Law and why is it important?
"Hy's Law is a prognostic marker in drug-induced liver injury. It requires three criteria: ALT greater than three times upper limit of normal indicating hepatocellular injury, bilirubin greater than two times upper limit of normal indicating jaundice, and absence of cholestasis meaning ALP less than two times upper limit. When all three are present, it predicts 10-50% mortality or need for liver transplantation. It's important because it identifies high-risk cases requiring urgent hepatology input and transplant centre notification. The law is named after Hyman Zimmerman who observed that drug-induced hepatocellular injury with jaundice carries high mortality. It's used by the FDA as a safety signal in drug development." [5]
Q: How do you differentiate hepatocellular from cholestatic DILI?
"I use the R-ratio, which is the ratio of ALT elevation to ALP elevation. Specifically, divide ALT by its upper limit of normal, then divide ALP by its upper limit of normal, then calculate the ratio. If R is 5 or greater, it's hepatocellular pattern indicating predominantly hepatocyte injury. If R is 2 or less, it's cholestatic indicating bile duct injury or bile flow impairment. If R is between 2 and 5, it's a mixed pattern. Hepatocellular DILI has higher risk of acute liver failure but better long-term prognosis if the patient survives. Cholestatic DILI has lower risk of acute liver failure but higher risk of chronic injury persisting beyond six months, including vanishing bile duct syndrome." [4,11]
Q: What is the mechanism of paracetamol hepatotoxicity and how does N-acetylcysteine work?
"In therapeutic doses, paracetamol is metabolized by glucuronidation and sulfation to safe metabolites. In overdose, these pathways saturate and paracetamol is metabolized by CYP2E1 and CYP3A4 to the toxic metabolite NAPQI. Normally, NAPQI is detoxified by hepatic glutathione. In overdose, glutathione stores deplete within 2-4 hours, and unbound NAPQI binds to cellular proteins, especially in mitochondria, causing oxidative stress, mitochondrial dysfunction, and centrilobular necrosis in zone 3 of the liver acinus. N-acetylcysteine works by three mechanisms: it restores glutathione stores by providing cysteine, it directly binds and detoxifies NAPQI, and it improves hepatic microcirculation. It's most effective within 8 hours of ingestion but has benefit up to 24 hours and even in established liver failure. The standard UK regimen is 150 mg/kg over 1 hour, then 50 mg/kg over 4 hours, then 100 mg/kg over 16 hours, totaling 300 mg/kg over approximately 21 hours." [16]
Q: Which drugs commonly cause DILI and what are their patterns?
"The commonest cause of intrinsic DILI is paracetamol in overdose, causing hepatocellular injury. For idiosyncratic DILI, amoxicillin-clavulanate is the most commonly implicated drug in Western countries, typically causing cholestatic or mixed pattern with onset 1-6 weeks after starting, sometimes even after stopping. Isoniazid causes hepatocellular injury in 10-20% with mild ALT elevation and 1% with severe injury, more common in those over 35, alcohol users, or with HIV. Diclofenac causes hepatocellular injury and has higher DILI risk than other NSAIDs. Statins cause mild asymptomatic hepatocellular ALT elevation in 5-10% but severe injury is rare. Flucloxacillin causes cholestatic injury with strong HLA-B*57:01 genetic association. Valproate causes hepatocellular injury via mitochondrial toxicity. Herbal and dietary supplements account for 16-20% of DILI cases in the US, with green tea extract, anabolic steroids, and multi-ingredient bodybuilding supplements being common culprits." [10,13,17,19]
Q: What is RUCAM and how is it used?
"RUCAM is the Roussel Uclaf Causality Assessment Method, the most widely validated structured tool for assessing causality in suspected DILI. It scores seven domains: time to onset from drug initiation, course of liver enzymes after drug cessation, patient risk factors, role of concomitant drugs, exclusion of alternative causes, prior reports of hepatotoxicity for the drug, and response to rechallenge if performed. Each domain contributes positive or negative points. The total score is interpreted as: 0 or less excludes DILI, 1-2 is unlikely, 3-5 is possible, 6-8 is probable, and 9 or more is highly probable. A score of 6 or higher supports the diagnosis of DILI. It's particularly useful in complex cases with polypharmacy or when causality is uncertain. However, it's a diagnostic aid, not a definitive test, and clinical judgment remains paramount. Rechallenge is rarely performed due to safety concerns, so that domain often scores zero." [7]
Q: When should you refer to a transplant centre?
"Urgent transplant centre referral is indicated when acute liver failure criteria are met: INR 1.5 or greater plus any grade of hepatic encephalopathy in a patient without pre-existing cirrhosis. Also refer if approaching King's College Criteria for transplantation. For paracetamol-induced ALF, this means arterial pH less than 7.30 after resuscitation, or all three of INR greater than 6.5, creatinine greater than 300 micromoles per litre, and grade 3 or 4 encephalopathy. For non-paracetamol DILI causing ALF, criteria are INR greater than 6.5 regardless of encephalopathy grade, or any three of: age less than 10 or greater than 40, jaundice for more than 7 days before encephalopathy onset, INR greater than 3.5, bilirubin greater than 300 micromoles per litre, or specific high-risk etiology. Early referral is crucial as deterioration can be rapid, and logistics of transfer take time. Even if transplant isn't ultimately needed, specialist ALF units provide optimal supportive care." [25]
High-Yield Facts for Examinations
- R-ratio = (ALT/ULN) ÷ (ALP/ULN): ≥5 hepatocellular, ≤2 cholestatic, 2-5 mixed [4]
- Hy's Law: ALT > 3× + bili > 2× + no cholestasis = 10-50% mortality [5]
- Most common cause of ALF: Paracetamol/acetaminophen (46%) [1]
- Most common idiosyncratic DILI: Amoxicillin-clavulanate [10]
- HLA associations: Flucloxacillin (HLA-B*57:01, OR 80.6) [14]
- NAC benefits: Paracetamol overdose; also non-paracetamol ALF with grade I-II encephalopathy [12,16]
- Chronic DILI: > 6 months; 14-20% of cases; cholestatic pattern higher risk [11]
- RUCAM score: ≥6 = probable/highly probable DILI [7]
- Herbal/dietary supplements: 16-20% of DILI in US; increasing [13]
- Rechallenge: Generally avoided; contraindicated if Hy's Law, ALF, DRESS [29]
- Isoniazid hepatotoxicity: 10-20% mild ALT elevation, 1% severe; monitor monthly [17]
- Statin hepatotoxicity: Common asymptomatic ALT elevation (5-10%), but severe DILI rare [19]
- DRESS syndrome: Fever + rash + eosinophilia + multi-organ; latency 2-8 weeks; anticonvulsants [23]
- Vanishing bile duct syndrome: Permanent ductopenia; flucloxacillin, amoxicillin-clavulanate
- Immediate management: STOP drug, check paracetamol level, assess Hy's Law, exclude alternatives, hepatology referral if severe [6,27]
Common Pitfalls and How to Avoid Them
Pitfall 1: Missing herbal/supplement history
- ❌ Asking only about "medications"
- ✅ Specifically ask: "Do you take any vitamins, supplements, herbal remedies, protein powders, or traditional medicines?"
Pitfall 2: Not calculating R-ratio
- ❌ Describing as "transaminitis" or "cholestasis" without quantification
- ✅ Always calculate R-ratio to classify pattern accurately
Pitfall 3: Missing Hy's Law cases
- ❌ Noting ALT and bilirubin elevation separately without recognizing significance
- ✅ Systematically check Hy's Law criteria in all DILI cases: ALT > 3×? Bili > 2×? R ≥5?
Pitfall 4: Delayed drug cessation
- ❌ Waiting for causality assessment or specialist review before stopping
- ✅ Stop drug immediately when DILI suspected; early cessation improves outcomes [27]
Pitfall 5: Not excluding alternative diagnoses
- ❌ Assuming DILI based on drug history alone
- ✅ DILI is diagnosis of exclusion: rule out viral (HAV, HBV, HCV, HEV), autoimmune, biliary obstruction, ischemic hepatitis
Pitfall 6: Forgetting paracetamol level in ALF
- ❌ Not checking paracetamol in "non-overdose" presentations
- ✅ Check paracetamol level in ALL acute liver failure cases (therapeutic misadventure common)
Pitfall 7: Inappropriate rechallenge
- ❌ Restarting drug after resolution without risk assessment
- ✅ Rechallenge contraindicated if previous Hy's Law, ALF, DRESS, or severe injury
Pitfall 8: Missing DRESS syndrome
- ❌ Attributing rash and fever to separate causes
- ✅ Recognize triad: fever + rash + eosinophilia + hepatitis; latency 2-8 weeks; anticonvulsants [23]
Mnemonics
DILI STOP (Management Approach):
- Drug history complete (including supplements)
- Investigations (exclude other causes: viral, autoimmune, biliary)
- Liver pattern (calculate R-ratio)
- Identify high-risk (check Hy's Law criteria)
- Stop offending drug immediately
- Time to recovery (serial LFTs monitoring)
- Other support (NAC if paracetamol, nutrition, avoid hepatotoxins)
- Paracetamol protocol if applicable
HY'S LAW (High mortality marker):
- Hepatocellular (ALT > 3× ULN)
- Yellow (Bilirubin > 2× ULN)
- Synthetic function impaired if severe
RUCAM Domains (7):
- Time to onset
- Course after stopping
- Risk factors
- Concomitant drugs
- Alternative causes ruled out
- Prior reports (known hepatotoxicity)
- Rechallenge response (Mnemonic: "Two Cats Ran Chasing A Purple Rabbit")
14. Related Topics
Prerequisites
- Liver anatomy and physiology
- Drug metabolism and pharmacokinetics
- Cytochrome P450 enzyme system
Consequences
- Acute liver failure
- Chronic liver disease and cirrhosis
- Liver transplantation
Differentials (Causes of Acute Hepatitis)
- Viral hepatitis (HAV, HBV, HCV, HEV, EBV, CMV)
- Autoimmune hepatitis
- Alcoholic hepatitis
- Ischaemic hepatitis (shock liver)
- Budd-Chiari syndrome
- Wilson disease
- Acute fatty liver of pregnancy
- HELLP syndrome
Related Conditions
- Paracetamol/acetaminophen overdose
- DRESS syndrome
- Stevens-Johnson syndrome/Toxic epidermal necrolysis
- Reye syndrome (aspirin in children)
- Herbal and dietary supplement toxicity
Last Updated: 2026-01-09
Citation Count: 22 PubMed-indexed references Line Count: 1,482 lines
Frequently asked questions
Quick clarifications for common clinical and exam-facing questions.
When should I seek emergency care for drug-induced liver injury?
Seek immediate emergency care if you experience any of the following warning signs: Hy's Law: ALT less than 3x ULN + bilirubin less than 2x ULN (10-50% mortality), Signs of acute liver failure (encephalopathy, coagulopathy, INR less than 1.5), Paracetamol/acetaminophen overdose, Rapidly rising bilirubin or INR, Hypoglycaemia or renal impairment, DRESS syndrome features (fever, rash, eosinophilia).