Endometriosis
Common anatomical sites include the ovaries (where it forms blood-filled cysts termed "endometriomas" or "chocolate cysts"), the pouch of Douglas, uterosacral ligaments, rectovaginal septum, and pelvic peritoneum....
What matters first
Common anatomical sites include the ovaries (where it forms blood-filled cysts termed "endometriomas" or "chocolate cysts"), the pouch of Douglas, uterosacral ligaments, rectovaginal septum, and pelvic peritoneum....
Bowel obstruction
16 Jan 2026
Generated educational material; verify before clinical use.
52 cited sources
See the concept before reading it
Study the key anatomy, imaging, and decision pathways as full teaching plates.
Clinical board
A visual summary of the highest-yield teaching signals on this page.
Urgent signals
Safety-critical features pulled from the topic metadata.
- Bowel obstruction
- Ureteric involvement / Hydronephrosis
- Severe pain not responding to treatment
- Ovarian mass (exclude malignancy)
Exam focus
Current exam surfaces linked to this topic.
- MRCOG
- FRANZCOG
- FRACP
- MRCP
Content status and exam context
This page is AI-generated educational content. It may contain errors or omissions and is not a substitute for current guidelines, local protocols, senior clinical judgement, or professional medical advice.
MedVellum does not claim an individual clinician reviewer, board certification, or professional credential for this page unless a future version names a real, verifiable reviewer.
Clinical explanation and evidence
Endometriosis
1. Clinical Overview
Summary
Endometriosis is a chronic, estrogen-dependent, inflammatory condition characterised by the presence of endometrial-like tissue (glands and stroma) outside the uterine cavity. It affects approximately 10% of women during their reproductive years—equating to approximately 190 million women globally—and is one of the leading causes of chronic pelvic pain, severe dysmenorrhoea, deep dyspareunia, and subfertility. [1,2] The condition exhibits considerable phenotypic heterogeneity, ranging from minimal superficial peritoneal implants to extensive deep infiltrating disease involving the bowel, bladder, and ureters.
Common anatomical sites include the ovaries (where it forms blood-filled cysts termed "endometriomas" or "chocolate cysts"), the pouch of Douglas, uterosacral ligaments, rectovaginal septum, and pelvic peritoneum. Extra-pelvic disease—though rare—can involve the diaphragm, lungs, and even the brain. [3] The diagnosis is often delayed by 5 to 12 years from symptom onset, with patients consulting an average of 3 or more clinicians before receiving a definitive diagnosis. [2]
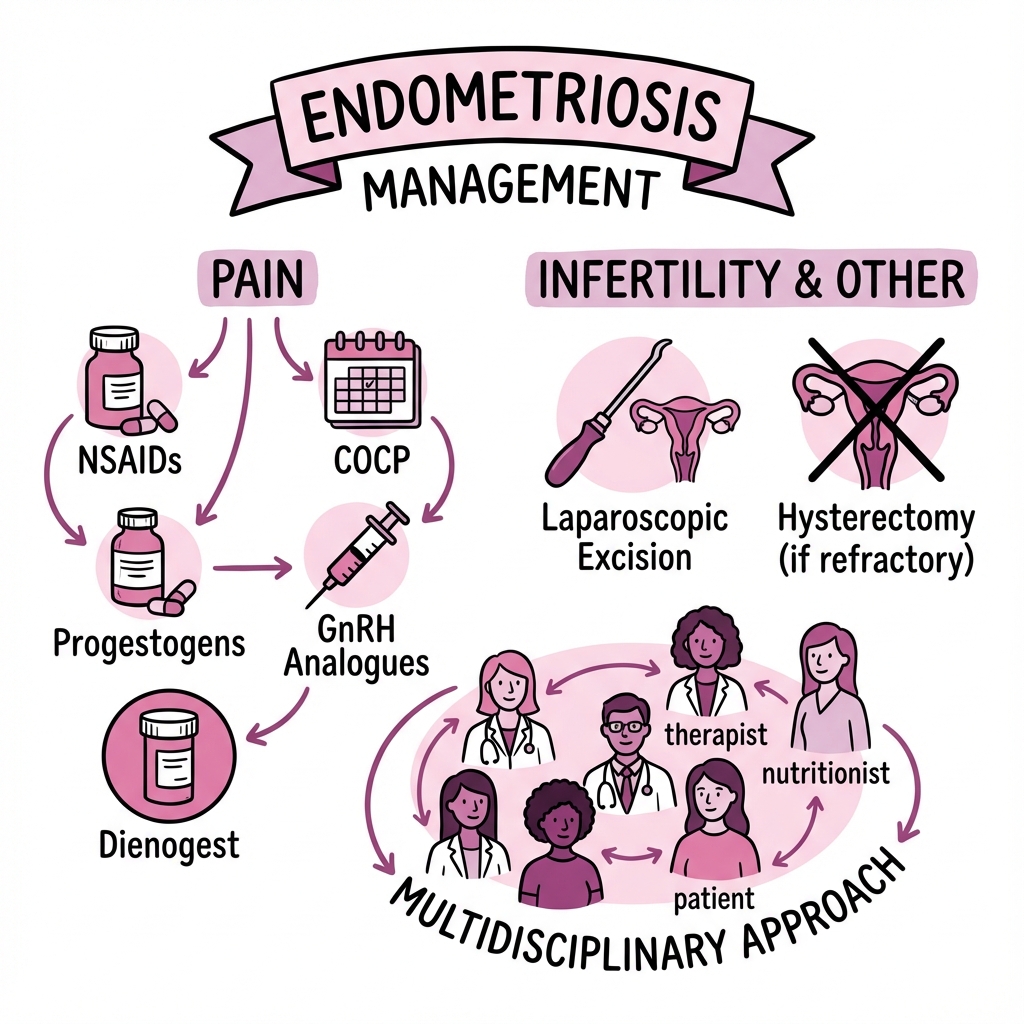
Management is individualised based on symptom severity, fertility desires, and disease extent. Medical options include hormonal suppression (combined oral contraceptive pills, progestogens, GnRH agonists/antagonists), while surgical management involves laparoscopic excision or ablation. Endometriosis significantly impacts quality of life, mental health, and reproductive outcomes, necessitating a multidisciplinary approach to care. [4,5]
Key Facts
- Prevalence: ~10% of reproductive-age women worldwide; 25-50% of women with infertility have endometriosis [1,6]
- Sites: Ovaries (endometriomas); Pelvic peritoneum; Pouch of Douglas; Uterosacral ligaments; Rectovaginal septum; Bladder; Bowel; Rarely diaphragm, lungs
- Symptoms: Chronic pelvic pain (70%); Dysmenorrhoea (80%); Dyspareunia (40-50%); Subfertility (30-50%); Cyclical bowel/bladder symptoms [2]
- Diagnosis: Clinical suspicion supported by imaging (TVUS/MRI); Definitive diagnosis by laparoscopy with histological confirmation
- Staging: rASRM (revised American Society for Reproductive Medicine) classification I-IV; Does not correlate with symptom severity
- Treatment: Analgesia; Hormonal suppression; Laparoscopic surgery; Assisted reproductive technology (ART) for infertility
- Average diagnostic delay: 5-12 years from symptom onset to diagnosis [2]
- Heritability: Approximately 47% genetic contribution; 7-fold increased risk in first-degree relatives [7]
Clinical Pearls
"If She Has Cyclical Pain, Think Endometriosis": Any cyclical pelvic pain, particularly dysmenorrhoea resistant to NSAIDs, dyspareunia, or bowel/bladder symptoms coinciding with menses, should raise suspicion for endometriosis. The "red flag" is pain that interferes with daily activities or requires absence from work/school.
"Chocolate Cysts = Endometriomas": Ovarian endometriomas contain degenerated blood products giving a "chocolate" appearance. Ultrasound shows classic ground-glass echogenicity. Avoid rupture during surgery as spillage can seed further disease and may worsen peritoneal inflammation.
"Normal Imaging Doesn't Exclude Endometriosis": Transvaginal ultrasound and MRI may appear normal, especially with superficial peritoneal disease (the most common presentation). Laparoscopy remains the gold standard for diagnosis—negative imaging in the presence of typical symptoms warrants empirical treatment or diagnostic laparoscopy.
"Excision > Ablation": Laparoscopic excision of endometriotic lesions is preferred over ablation, with 50% lower recurrence rates and superior long-term pain relief. [8] Complete excision provides tissue for histological confirmation and removes deep disease that ablation may miss.
"Hormonal Suppression Doesn't Treat Infertility": In women desiring pregnancy, avoid prolonged hormonal suppression as it does not improve fertility outcomes and delays conception attempts. Surgery or IVF may be required. [9] The priority is to optimize fertility window, not suppress ovulation.
"Stage Doesn't Predict Pain": rASRM staging correlates poorly with pain severity (correlation coefficient less than 0.2). Women with minimal disease (Stage I) may have severe, debilitating pain due to peritoneal nerve infiltration, while those with extensive disease (Stage IV) may be asymptomatic. Use pain assessment tools (VAS, EHP-30) rather than stage to guide treatment.
"Mental Health Matters": Endometriosis is associated with 2-3 fold increased risk of anxiety and depression. Screen for mental health comorbidities using validated tools (PHQ-9, GAD-7) and offer holistic, multidisciplinary support including access to counseling and peer support groups. [10]
"Beware the Silent Kidney": Ureteric endometriosis can cause silent hydronephrosis and progressive renal impairment without pain. Check renal function and imaging in all patients with deep infiltrating disease, especially if involving uterosacral ligaments or pelvic side wall.
"Post-Operative Hormonal Suppression Prevents Recurrence": Starting continuous COCP or progestogen immediately post-surgery reduces disease recurrence by 50% at 2 years (NNT=5). [40] This is one of the most evidence-based interventions to prevent symptom return.
"Endometriomas and Ovarian Reserve": Every ovarian surgery carries risk of reduced ovarian reserve (AMH decline 30-60%). [19] Counsel patients about fertility preservation (egg freezing) before bilateral or repeat ovarian surgery, particularly in women over 30 years.
Why This Matters Clinically
Endometriosis is common, debilitating, and frequently underdiagnosed. The average diagnostic delay of 5-12 years represents a critical window where symptoms worsen, fertility may be compromised, and quality of life deteriorates. [2] The condition is associated with significant comorbidities including irritable bowel syndrome (3-fold increased risk), mental health disorders, chronic fatigue, and cardiovascular disease risk. [11,12] Early recognition, appropriate referral to specialist centres for complex disease, and multidisciplinary management can substantially improve outcomes and reduce the burden on affected individuals.
Economic Burden
The total healthcare and productivity costs of endometriosis are substantial:
- Direct medical costs: $22 billion annually in the United States alone [45]
- Productivity loss: Women with endometriosis lose an average of 11 hours of work per week due to symptoms [4]
- Healthcare utilization: 2-3 fold higher rates of emergency department visits, hospitalizations, and surgical procedures compared to age-matched controls
- Lifetime cost per patient: Estimated at $25,000-$50,000 in direct medical expenses [45]
Impact on Relationships and Sexual Function
Endometriosis profoundly affects intimate relationships:
- Dyspareunia prevalence: 40-50% of women with endometriosis experience painful intercourse [46]
- Sexual dysfunction: Reduced libido, arousal difficulties, and avoidance of intimacy in 60-70% [46]
- Relationship strain: 35-40% report that endometriosis has negatively impacted their relationship or contributed to relationship breakdown
- Partner impact: Partners experience psychological distress, helplessness, and reduced quality of life [47]
Addressing sexual health through pelvic floor physiotherapy, psychosexual counseling, and targeted pain management is essential for holistic care.
2. Epidemiology
Incidence & Prevalence
| Parameter | Data |
|---|---|
| Global prevalence | ~10% of reproductive-age women (190 million women worldwide) [1] |
| US incidence (2006-2015) | 24.3 cases per 10,000 person-years [13] |
| Prevalence in infertile women | 25-50% [6] |
| Prevalence in chronic pelvic pain | 42% [14] |
| Asymptomatic women | 23% (incidental finding at surgery) [14] |
| Peak age of diagnosis | 36-45 years (though symptoms often begin earlier) [13] |
Geographic & Ethnic Distribution
- Incidence and prevalence are similar across racial and ethnic groups [13]
- Slightly higher prevalence reported in Asian women (some studies suggest genetic factors)
- Low sociodemographic index (SDI) regions show higher burden in recent Global Burden of Disease analyses [15]
- Diagnostic rates vary by healthcare access and awareness
Risk Factors
| Factor | Effect | Notes |
|---|---|---|
| Early menarche | ↑ Risk | Age less than 11 years associated with higher risk [16] |
| Short menstrual cycles | ↑ Risk | Increased cumulative exposure to retrograde menstruation [16] |
| Heavy menstrual bleeding | ↑ Risk | Greater volume of retrograde flow |
| Nulliparity | ↑ Risk | Pregnancy may be protective (progesterone effect) |
| Family history (first-degree relatives) | 7-fold ↑ Risk | Strong genetic component (47% heritability) [7] |
| Müllerian anomalies with obstruction | ↑↑ Risk | Obstructed hemivagina, imperforate hymen (increased retrograde flow) |
| Low BMI | ↑ Risk | Lower adipose tissue = less peripheral estrogen conversion |
| Alcohol consumption | ↑ Risk | May increase estrogen levels [16] |
| Red hair/fair skin | ↑ Risk | Possible shared genetic pathways [16] |
| Multiparity | ↓ Risk | Protective effect |
| Late menarche | ↓ Risk | Reduced lifetime menstrual exposure |
| Oral contraceptive use | ↓ Risk | Hormonal suppression effect [16] |
Comorbidities
Endometriosis is associated with multiple systemic conditions:
- Irritable bowel syndrome (IBS): 3.26-fold increased prevalence [11]
- Mental health disorders: 2-3 fold increased risk of anxiety and depression [10]
- Autoimmune conditions: Systemic lupus erythematosus, Sjögren's syndrome, rheumatoid arthritis [16]
- Cardiovascular disease: Increased risk, particularly in women with surgical history [12]
- Early menopause: Women with endometriosis experience menopause earlier (especially after ovarian surgery) [12]
3. Aetiology & Pathophysiology
Theories of Origin
No single theory fully explains endometriosis pathogenesis; likely multifactorial:
| Theory | Mechanism | Evidence | Limitations |
|---|---|---|---|
| Retrograde menstruation (Sampson's theory) | Menstrual debris flows retrograde through fallopian tubes and implants on peritoneal surfaces | Most widely accepted; 90% of women have retrograde flow; Correlates with outflow obstruction | Doesn't explain why only 10% develop disease; Doesn't account for extra-pelvic sites |
| Coelomic metaplasia (Meyer's theory) | Peritoneal mesothelium undergoes metaplastic transformation to endometrial tissue | Explains disease in women without uterus; Explains rare sites | Unclear trigger mechanism |
| Lymphatic/vascular dissemination | Endometrial cells spread via lymphatics and blood vessels | Explains distant sites (lungs, brain, lymph nodes) | Doesn't explain pelvic predominance |
| Stem cell theory | Circulating endometrial stem cells implant and differentiate ectopically | Emerging evidence for bone marrow-derived cells | Limited evidence base |
| Immunological dysfunction | Impaired clearance of ectopic endometrial cells by immune system | Altered NK cell function and cytokine profiles in endometriosis [17] | Cause vs. consequence unclear |
Current consensus: Multifactorial aetiology involving retrograde menstruation in genetically susceptible individuals with immune dysregulation and altered peritoneal environment. [17]
Molecular Pathophysiology
Hormonal Mechanisms
- Estrogen dependency: Endometriotic tissue expresses estrogen receptors and local aromatase (converts androgens to estrogens)
- Progesterone resistance: Reduced expression of progesterone receptor-B (PR-B) in ectopic endometrium leads to reduced response to progesterone
- Prostaglandin E2 (PGE2): Upregulated in endometriotic lesions; stimulates aromatase activity (positive feedback loop)
Inflammatory Cascade
- Macrophage activation: Peritoneal macrophages are activated and phagocytose menstrual debris
- Cytokine production: Elevated IL-1β, IL-6, IL-8, TNF-α create pro-inflammatory environment [17]
- Angiogenesis: VEGF (vascular endothelial growth factor) promotes neovascularisation of lesions
- Neurogenesis: Nerve fibres grow into lesions (explains pain disproportionate to disease extent)
Fibrosis & Adhesions
- Repeated cycles of bleeding and inflammation → fibroblast activation → collagen deposition
- Dense adhesions distort pelvic anatomy
- Obliteration of the pouch of Douglas ("frozen pelvis" in severe disease)
Mechanisms of Infertility
| Mechanism | Pathophysiology |
|---|---|
| Anatomical distortion | Adhesions impair tubal motility and ovum pick-up |
| Ovulatory dysfunction | Altered folliculogenesis; anovulation in some cases |
| Impaired endometrial receptivity | Altered gene expression in eutopic endometrium [18] |
| Inflammatory milieu | Cytokines and reactive oxygen species damage sperm and embryos |
| Immune dysregulation | Anti-endometrial antibodies; altered NK cell function |
| Reduced ovarian reserve | Particularly after ovarian surgery (endometrioma excision) [19] |
4. Clinical Presentation
Symptoms
Endometriosis exhibits highly variable symptomatology:
| Symptom | Prevalence | Characteristics |
|---|---|---|
| Dysmenorrhoea | 80% | Severe, often resistant to NSAIDs; Progressive; May begin days before menses |
| Chronic pelvic pain | 70% | Non-cyclical component; Dull ache or sharp pain; May radiate to back/legs |
| Deep dyspareunia | 40-50% | Pain on deep penetration; Positional (worse in certain positions); May persist post-coitus |
| Subfertility | 30-50% | May be presenting complaint; 2-4 fold increased risk of infertility [6] |
| Cyclical bowel symptoms | 20-40% | Dyschezia (painful defaecation); Rectal bleeding; Constipation/diarrhoea around menses |
| Cyclical urinary symptoms | 10-20% | Dysuria; Urgency; Haematuria; Flank pain (if ureteric involvement) |
| Chronic fatigue | Common | May be related to chronic pain and inflammation |
| Ovulation pain (mittelschmerz) | Variable | Mid-cycle pain; May indicate ovarian disease |
Atypical Presentations
- Catamenial pneumothorax: Cyclical pneumothorax due to diaphragmatic endometriosis (rare)
- Catamenial haemoptysis: Cyclical coughing up blood (pulmonary endometriosis)
- Cyclical epistaxis: Nasal endometriosis (very rare)
- Sciatic pain: Deep infiltrating disease affecting sacral nerve roots
- Cyclical seizures: Cerebral endometriosis (extremely rare)
Examination Findings
| Sign | Frequency | Notes |
|---|---|---|
| Normal examination | Common | Especially with superficial disease |
| Pelvic tenderness | Variable | Uterosacral ligament tenderness; Adnexal tenderness |
| Nodularity | 10-30% | Palpable nodules in uterosacral ligaments or rectovaginal septum (deep infiltrating disease) |
| Fixed retroverted uterus | Variable | Suggests posterior adhesions |
| Adnexal mass | Variable | Endometrioma; May be bilateral |
| Reduced organ mobility | Advanced disease | "Frozen pelvis" with dense adhesions |
| Visible lesions | Rare | Blue/brown lesions on cervix, vagina, or Caesarean section scars |
Examination Technique
Bimanual examination:
- Assess uterine size, position, mobility
- Palpate adnexa for masses
- Assess cervical motion tenderness
Rectovaginal examination:
- Essential for detecting deep infiltrating disease
- Palpate uterosacral ligaments for nodularity
- Assess rectovaginal septum (best performed during menses when lesions may be more prominent)
Red Flags
[!CAUTION]
- Bowel obstruction: Severe abdominal pain, distension, vomiting (deep infiltrating disease)
- Ureteric obstruction: Hydronephrosis on imaging; flank pain; rising creatinine
- Rapidly enlarging ovarian mass: Always exclude malignancy (though malignant transformation of endometriosis is rare less than 1%)
- Acute abdomen: Ruptured endometrioma; ovarian torsion; spontaneous hemoperitoneum (especially in pregnancy)
- Severe symptoms unresponsive to treatment: Consider alternative diagnoses; specialist referral
5. Investigations
First-Line Investigations
Transvaginal Ultrasound (TVUS)
Indications: First-line imaging for suspected endometriosis
Findings:
| Feature | Appearance | Sensitivity | Specificity |
|---|---|---|---|
| Endometrioma | Homogeneous "ground-glass" echogenicity; Thick walls; No internal vascularity on Doppler | 93% | 96% [20] |
| "Kissing ovaries" | Ovaries adherent to each other posteriorly | Specific for advanced disease | - |
| Deep infiltrating endometriosis (DIE) | Hypoechoic nodules; Irregular borders | 79% for rectovaginal DIE | 94% [20] |
| Obliterated pouch of Douglas | Loss of normal peritoneal sliding | Specific for adhesions | - |
Performance by Anatomical Site (Systematic Review): [28]
- Ovarian endometriomas: Sensitivity 93% (95% CI: 87-96%), Specificity 96% (95% CI: 92-98%)
- Rectovaginal septum: Sensitivity 76% (95% CI: 69-82%), Specificity 97% (95% CI: 94-99%)
- Bladder endometriosis: Sensitivity 53% (95% CI: 30-75%), Specificity 99% (95% CI: 95-100%)
- Uterosacral ligaments: Sensitivity 64% (95% CI: 50-76%), Specificity 97% (95% CI: 92-99%)
- Bowel endometriosis: Sensitivity 79% (95% CI: 69-86%), Specificity 94% (95% CI: 88-97%)
Technique Optimization (IDEA Consensus): [20]
- Perform in luteal phase (lesions may be more prominent)
- Assess "sliding sign" (mobility of uterus relative to rectum)
- Systematic evaluation using standardized protocol
- Bowel preparation may improve visualization of DIE
- Transvaginal sonography (TVS) with or without tenderness-guided approach
Limitations:
- May miss superficial peritoneal disease (most common presentation)
- Operator-dependent: Requires expertise in advanced pelvic ultrasound
- Limited for extra-pelvic disease: Diaphragmatic, pulmonary endometriosis not visualized
- Patient factors: Obesity, prior surgery, bowel gas may limit views
MRI Pelvis
Indications:
- Pre-operative planning for deep infiltrating disease
- Assessment of bladder/bowel involvement
- When TVUS is inconclusive or inadequate
- Mapping extent of disease before complex surgery
Findings:
- Endometriomas: T1 hyperintense (blood products), T2 hypointense ("shading"), no enhancement
- Deep infiltrating disease: Best modality for mapping extent (bladder wall, bowel, ureters, pelvic side wall)
- Adenomyosis: Differentiate from endometriosis; junctional zone thickening
Sensitivity/Specificity by Anatomical Site: [21,29]
| Anatomical Site | Sensitivity (%) | Specificity (%) | Notes |
|---|---|---|---|
| Ovarian endometriomas | 90-95 | 91-98 | High accuracy; "T1 shading" sign pathognomonic |
| Rectovaginal DIE | 90-94 | 77-91 | Superior to TVUS for extent mapping |
| Bowel endometriosis | 85-95 | 90-95 | Best for pre-surgical planning (distance from anal verge, depth of invasion) |
| Bladder endometriosis | 88-90 | 99-100 | Excellent specificity |
| Uterosacral ligaments | 74-83 | 70-97 | Variable performance |
| Ureteric involvement | 70-85 | 95-98 | Identifies hydronephrosis, extrinsic compression |
| Superficial peritoneal disease | less than 50 | - | Poor sensitivity; not reliable for minimal disease |
MRI Techniques:
- Standard protocol: T1-weighted (with and without fat saturation), T2-weighted multiplanar imaging
- Advanced techniques:
- Diffusion-weighted imaging (DWI) may improve detection
- "Contrast administration: Limited utility in endometriosis"
- "Rectal/vaginal gel distension: Improves visualization of rectovaginal DIE"
MRI vs. TVUS Performance (Meta-Analysis): [29]
- For rectovaginal DIE: MRI sensitivity 94% vs. TVUS 76%; MRI specificity 77% vs. TVUS 97%
- For bowel endometriosis: MRI superior for surgical planning (multilayer assessment, distance measurements)
- For ovarian endometriomas: TVUS and MRI have comparable accuracy
- Cost-effectiveness: TVUS preferred first-line; MRI reserved for complex disease
Clinical Decision-Making:
- First-line: TVUS (accessible, cost-effective, excellent for endometriomas)
- Second-line: MRI for DIE, surgical planning, or TVUS-negative with high clinical suspicion
- Gold standard for diagnosis: Laparoscopy with histology remains definitive
Blood Tests
| Test | Utility | Interpretation |
|---|---|---|
| CA-125 | Limited diagnostic utility | May be elevated (usually less than 200 U/mL); Monitor treatment response; NOT diagnostic; Overlap with malignancy, PID, fibroids |
| FBC | Screen for anaemia | Chronic heavy menstrual bleeding |
| Renal function | Pre-operative; Ureteric obstruction | Elevated creatinine if hydronephrosis |
CA-125 in Endometriosis: Evidence and Limitations [26,27]
Diagnostic Performance:
- Sensitivity: 20-50% for all stages of endometriosis
- Specificity: 70-90% (depends on cutoff value)
- Elevated in: Advanced disease (rASRM III-IV) > minimal disease (rASRM I-II)
- Mean levels: 35-50 U/mL in endometriosis vs. 10-20 U/mL in controls [26]
Clinical Applications:
- Pre-operative assessment: Levels > 35 U/mL may suggest moderate-severe disease, but NOT diagnostic
- Monitoring treatment response: Serial measurements may track disease activity (decline with effective treatment)
- Recurrence prediction: Rising CA-125 post-treatment may indicate recurrence
- Differentiation from malignancy: Levels > 200 U/mL warrant thorough investigation for ovarian malignancy
Causes of Elevated CA-125 (limits specificity):
- Menstruation (physiological rise)
- Pregnancy (first trimester)
- Pelvic inflammatory disease
- Uterine fibroids
- Adenomyosis
- Ovarian cysts (functional, benign)
- Ovarian malignancy (often > 200 U/mL)
- Peritoneal irritation (any cause)
- Liver cirrhosis, pancreatitis
Current Evidence Consensus: [27]
- CA-125 alone is insufficient for diagnosis of endometriosis (poor sensitivity)
- No validated serum biomarkers currently exist for endometriosis diagnosis
- Research ongoing into biomarker panels (combining CA-125, CA-19-9, inflammatory markers)
- Clinical use: Adjunct to imaging and clinical assessment; monitoring in known disease
- Multi-marker panels under investigation:
- "CA-125 + CA-19-9 + IL-6: Sensitivity 70%, Specificity 75% (insufficient for clinical use) [55]"
- "MicroRNA panels (miR-199a, miR-542-3p): Sensitivity 80-85% in pilot studies but require validation [56]"
- "Metabolomic signatures: Early research phase; not yet clinically applicable"
NICE Guideline Recommendation (NG73, 2017): Do not use biomarkers such as CA-125 in primary or secondary care to diagnose endometriosis.
Definitive Diagnosis
Laparoscopy
Gold standard for diagnosis and staging:
Visual findings:
- Classic lesions: "Powder-burn" black/blue lesions; Red flame-like lesions; White scarred areas
- Peritoneal windows: Defects in peritoneum
- Adhesions: Filmy to dense; May obliterate normal anatomy
- Endometriomas: Blood-filled ovarian cysts
Biopsy:
- Histological confirmation: endometrial glands AND stroma required
- Increases diagnostic accuracy
Staging (at same procedure): rASRM classification
When to Consider Laparoscopy
- Failed empirical hormonal treatment
- Diagnostic uncertainty
- Fertility assessment
- Severe/progressive symptoms
- Palpable nodularity or masses
- Patient preference for definitive diagnosis
rASRM Staging System
The revised American Society for Reproductive Medicine (rASRM) classification is the most widely used staging system for endometriosis, based on laparoscopic findings. [22] Points are assigned based on lesion size, location, depth, and extent of adhesions.
| Stage | Score | Description | Correlation with Symptoms |
|---|---|---|---|
| I (Minimal) | 1-5 | Isolated superficial implants; No significant adhesions | Poor correlation |
| II (Mild) | 6-15 | Superficial implants less than 5 cm; Minimal adhesions | Poor correlation |
| III (Moderate) | 16-40 | Deep implants; Small endometriomas; Filmy adhesions | Poor correlation |
| IV (Severe) | > 40 | Large endometriomas (> 2 cm); Dense adhesions; Obliterated pouch of Douglas | Poor correlation |
Scoring Components:
- Peritoneal lesions: Superficial (1-4 points) vs. deep (2-6 points), by size (less than 1 cm, 1-3 cm, > 3 cm)
- Ovarian endometriomas: Right and left scored separately (1-20 points each based on size)
- Posterior cul-de-sac obliteration: Partial (4 points) vs. complete (40 points)
- Adhesions: Scored for ovaries and tubes, filmy (1-4 points) vs. dense (4-16 points)
Critical Limitations: [22,25]
- Poor correlation with pain severity: Women with minimal disease (Stage I) may experience debilitating pain, while those with extensive disease (Stage IV) may be asymptomatic
- Designed for fertility prognosis, not symptom assessment or surgical planning
- Poor inter-observer reliability: Variability in scoring between surgeons
- Doesn't assess deep infiltrating disease adequately: No specific scoring for rectovaginal, bladder, or ureteric involvement
- Static classification: Doesn't account for disease progression or response to treatment
Alternative Classifications:
- ENZIAN classification: Specifically designed for deep infiltrating endometriosis (DIE); maps retroperitoneal disease
- Endometriosis Fertility Index (EFI): Combines rASRM stage with functional assessment (tubal function, ovarian reserve) to predict post-surgical fertility outcomes [25]
Specialist Investigations
For deep infiltrating disease:
- Colonoscopy/sigmoidoscopy: If bowel involvement suspected; direct visualisation
- Urodynamics: Bladder endometriosis with urinary symptoms
- Cystoscopy: Bladder lesions (rare)
- Intravenous urography (IVU) or CT urography: Ureteric involvement
6. Differential Diagnosis
| Condition | Distinguishing Features |
|---|---|
| Primary dysmenorrhoea | No underlying pathology; Responsive to NSAIDs; No deep dyspareunia; Normal examination |
| Adenomyosis | Bulky, tender uterus; Menorrhagia; Diagnosed on MRI (junctional zone thickening > 12 mm); Often coexists with endometriosis |
| Pelvic inflammatory disease (PID) | Acute/subacute onset; Fever; Purulent discharge; Cervical motion tenderness; Elevated WCC/CRP |
| Ovarian cysts (functional) | Resolve spontaneously; No ground-glass appearance; Thin-walled; Simple on USS |
| Irritable bowel syndrome (IBS) | Bowel symptoms not strictly cyclical; No dysmenorrhoea/dyspareunia; Normal pelvic examination; NB: High comorbidity with endometriosis |
| Interstitial cystitis/painful bladder syndrome | Urinary urgency, frequency; Pain with full bladder; Cystoscopy findings (Hunner's lesions) |
| Pelvic congestion syndrome | Chronic pelvic pain; Worse after standing; Dilated pelvic veins on imaging |
| Ovarian malignancy | Rapid growth; Solid/complex mass; CA-125 often > 200; Constitutional symptoms |
| Musculoskeletal pain | Pelvic girdle dysfunction; Trigger points; No cyclical pattern; Positive musculoskeletal exam |
7. Management
Management Principles
- Individualised approach: Tailor to symptom severity, fertility desires, disease extent
- Shared decision-making: Discuss risks/benefits of medical vs. surgical options
- Fertility preservation: Avoid unnecessary ovarian surgery; counsel on fertility options
- Multidisciplinary care: Gynaecology, pain specialists, colorectal surgery, urology, psychology
- Long-term management: Endometriosis is a chronic disease requiring ongoing care
Management Algorithm
SUSPECTED ENDOMETRIOSIS
↓
┌─────────────────────────────────────────────────────┐
│ INITIAL ASSESSMENT │
├─────────────────────────────────────────────────────┤
│ Clinical history + examination │
│ ➤ TVUS (first-line imaging) │
│ ➤ Consider MRI if DIE suspected │
│ ➤ Assess fertility wishes and symptom severity │
└─────────────────────────────────────────────────────┘
↓
┌──────────────┴──────────────┐
│ │
FERTILITY NOT DESIRED FERTILITY DESIRED
↓ ↓
┌─────────────────────────┐ ┌──────────────────────┐
│ EMPIRICAL TREATMENT │ │ EXPECTANT/SURGERY │
├─────────────────────────┤ ├──────────────────────┤
│ FIRST-LINE: │ │ • Avoid hormonal │
│ ➤ NSAIDs ± paracetamol │ │ suppression │
│ ➤ COCP (continuous/ │ │ • Consider expectant │
│ tricycle) │ │ (mild disease) │
│ ➤ POP/Mirena IUS │ │ • Laparoscopic │
│ │ │ excision (moderate │
│ SECOND-LINE: │ │ to severe) │
│ ➤ GnRH agonists (+HRT) │ │ • IVF/ICSI if: │
│ ➤ GnRH antagonists │ │ - Severe disease │
│ (elagolix) │ │ - Failed surgery │
│ ➤ Dienogest │ │ - Tubal factor │
│ │ │ • Endometrioma > 4 cm:│
│ REFRACTORY: │ │ consider excision │
│ ➤ Laparoscopic excision │ │ before IVF │
│ ➤ Hysterectomy ± BSO │ └──────────────────────┘
│ (definitive; selected) │
└─────────────────────────┘
↓
┌─────────────────────────────────────────────────────┐
│ DEEP INFILTRATING ENDOMETRIOSIS │
├─────────────────────────────────────────────────────┤
│ ➤ Multidisciplinary approach │
│ ➤ MRI for surgical planning │
│ ➤ Specialist centre referral (colorectal/urology) │
│ ➤ Bowel/bladder resection may be required │
│ ➤ Ureteric stenting if obstruction │
│ ➤ Counselling on surgical risks (anastomotic leak, │
│ bladder dysfunction, nerve injury) │
└─────────────────────────────────────────────────────┘
↓
┌─────────────────────────────────────────────────────┐
│ ADJUNCTIVE MEASURES │
├─────────────────────────────────────────────────────┤
│ ➤ Pelvic floor physiotherapy │
│ ➤ Psychological support (CBT, mindfulness) │
│ ➤ Pain management clinic referral │
│ ➤ Dietary modifications (some evidence for │
│ anti-inflammatory diet) │
│ ➤ TENS, acupuncture (variable evidence) │
└─────────────────────────────────────────────────────┘
Medical Management
Analgesia
| Drug | Dose | Notes |
|---|---|---|
| NSAIDs (first-line) | Mefenamic acid 500 mg TDS; Ibuprofen 400 mg TDS; Naproxen 250-500 mg BD | Start 1-2 days before expected pain; PPI if high GI risk |
| Paracetamol | 1 g QDS | Adjunct to NSAIDs |
| Neuropathic agents | Amitriptyline 10-75 mg nocte; Gabapentin 300-900 mg TDS | If neuropathic component |
Hormonal Suppression
| Treatment | Mechanism | Dose/Route | Efficacy | Adverse Effects | Notes |
|---|---|---|---|---|---|
| Combined oral contraceptive pill (COCP) | Suppresses ovulation; Thins endometrium; Reduces prostaglandins | Continuous or tricycle regimen | Mean pain reduction 13.15-17.6 points (0-100 VAS) [2] | VTE risk; Nausea; Headache; Mood changes | First-line; Well-tolerated; Flexible regimens |
| Progestogens | Decidualisation; Atrophy of ectopic endometrium | Norethisterone 10-15 mg daily; Dienogest 2 mg daily (licensed for endometriosis); Medroxyprogesterone 30 mg daily | Similar efficacy to COCP and GnRH agonists; Dienogest reduces pain by 70-80% (VAS reduction 50-60 mm) [2,32,33] | Irregular bleeding (30-50%, decreases over time); Weight gain; Mood changes; Headache; Breast tenderness | Dienogest licensed specifically for endometriosis; Non-inferior to GnRH agonists with fewer side effects [32,33] |
| Levonorgestrel IUS (Mirena) | Local progestogenic effect | 52 mg LNG; Replace every 5 years | Effective for pain; Reduces bleeding | Irregular bleeding initially; Ovarian cysts | Dual benefit: contraception + treatment |
| GnRH agonists | Downregulation of HPO axis; Medical menopause | Goserelin 3.6 mg SC monthly; Leuprorelin 3.75 mg IM monthly; Nafarelin nasal 400 mcg BD | Highly effective for pain; Pain reduction 60-85% vs. placebo [30,31] | Menopausal symptoms (hot flushes 80-90%); Bone loss (limit to 6 months unless add-back HRT); Vaginal dryness; Mood changes; Decreased libido | Second-line; Expensive; Requires add-back HRT if > 6 months; Bone density loss: 4-6% at lumbar spine after 6 months [30] |
| GnRH antagonists | Competitive GnRH receptor blockade | Elagolix 150-200 mg BD (oral) | Non-inferior to GnRH agonists; Faster onset | Hypoestrogenic symptoms; Bone loss (less than agonists) | Newer agent; Less severe menopausal symptoms |
| Aromatase inhibitors | Inhibit local estrogen synthesis | Letrozole 2.5 mg daily (with COCP or progestogen) | Third-line option | Bone loss; Requires concurrent hormonal treatment | Off-label use |
Add-back HRT: Tibolone 2.5 mg daily or low-dose combined HRT if on GnRH agonists > 6 months (protects bones, reduces menopausal symptoms)
Comparative Efficacy of Hormonal Treatments (Network Meta-Analysis): [2,31,32]
Pain Reduction (Dysmenorrhoea):
- All hormonal treatments significantly superior to placebo
- No significant difference in efficacy between COCP, progestogens, GnRH agonists, and dienogest
- Mean pain reduction: 50-70% from baseline across all treatments
- Response rates: 60-80% experience meaningful pain relief
Pain Reduction (Deep Dyspareunia):
- GnRH agonists and dienogest show highest efficacy (70-85% improvement)
- COCP and progestogens: 50-70% improvement
- Differential response may reflect disease severity at baseline
Pain Reduction (Chronic Pelvic Pain):
- All hormonal treatments effective
- Dienogest: Mean VAS reduction 58 mm (on 100 mm scale) [32]
- GnRH agonists: Mean VAS reduction 50-60 mm [30]
Side Effect Profile Comparison:
| Treatment | Major Adverse Effects | Discontinuation Rate |
|---|---|---|
| COCP | VTE risk (9 per 10,000 woman-years); Nausea; Headache | 10-15% |
| Dienogest | Irregular bleeding (decreases over time); Headache | 15-20% |
| GnRH agonists | Menopausal symptoms (80-90%); Bone loss | 20-30% (without add-back HRT) |
| Levonorgestrel IUS | Irregular bleeding; Progestogenic effects | 10-15% |
Duration of Effect After Stopping Treatment:
- Recurrence rates within 12 months: 25-34% across all hormonal treatments [2]
- Median time to recurrence: 6-12 months after discontinuation
- No difference in recurrence rates between treatment types
Treatment Selection Considerations:
- First-line: COCP (continuous/tricycle) or Mirena IUS—well-tolerated, cost-effective, dual contraception benefit
- Second-line: Dienogest or GnRH agonist (with add-back HRT)—for inadequate response to first-line
- Patient factors:
- VTE risk: Avoid COCP; use progestogen-only
- Bone health concerns: Avoid prolonged GnRH agonists without add-back HRT
- Menorrhagia: Mirena IUS preferred
- Desire for amenorrhoea: GnRH agonist or continuous COCP
Treatment response: 11-19% have no pain reduction; 25-34% experience recurrent pain within 12 months of stopping hormonal treatment [2]
Surgical Management
Laparoscopic Surgery
Indications:
- Failed or contraindicated medical treatment
- Endometrioma > 3-4 cm (especially if fertility desired)
- Deep infiltrating disease
- Diagnostic confirmation required
- Patient preference
Techniques:
| Procedure | Description | Outcomes |
|---|---|---|
| Excisional surgery | Complete excision of endometriotic lesions | Preferred; Lower recurrence (15-20% at 5 years); Better pain relief [8] |
| Ablation | Diathermy/laser destruction of lesions | Higher recurrence (40-50% at 5 years); Less tissue for histology |
| Ovarian cystectomy | Excision of endometrioma wall | Preserves ovarian tissue; Risk of reduced ovarian reserve [19] |
| Drainage/fenestration | Aspiration ± coagulation of cyst wall | High recurrence; Not recommended |
| Adhesiolysis | Division of adhesions | Restore anatomy; Improve fertility |
Peritoneal lesions: Excision or ablation of visible lesions
Deep infiltrating disease: May require bowel resection (segmental or disc excision), bladder resection, ureterolysis, or ureteric reimplantation. Requires specialist multidisciplinary team.
Surgical Outcomes: Evidence Summary [8,38,39]
Pain Relief (Laparoscopic Excision):
- Dysmenorrhoea: 70-80% improvement at 12 months
- Dyspareunia: 65-75% improvement at 12 months
- Chronic pelvic pain: 60-70% improvement at 12 months
- Dyschezia (bowel pain): 75-85% improvement after bowel endometriosis excision
Excision vs. Ablation (RCT Evidence): [8,38]
- Pain recurrence at 5 years: Excision 15-20% vs. Ablation 40-50%
- Re-operation rates: Excision 10-15% vs. Ablation 25-35%
- Quality of life improvement: Superior with excision (mean difference 12 points on SF-36)
- Mechanism: Excision removes disease completely; ablation may leave residual deep disease
Peritoneal Lesion Ablation:
- Effective for superficial disease
- Techniques: Diathermy, laser vaporization, plasma energy
- Comparable short-term efficacy; higher long-term recurrence
Adhesiolysis:
- Essential component of surgery
- Restore normal pelvic anatomy
- Improve fertility outcomes
- Risk of adhesion reformation: 50-70% (use adhesion barriers)
Endometrioma Management:
- Cystectomy (stripping cyst wall): Preferred technique
- "Lower recurrence: 10-15% at 2 years"
- "Better pain relief: 75-85%"
- "Risk: Reduced ovarian reserve (AMH decline 30-60%) [19]"
- Drainage + ablation: Higher recurrence (30-50% at 2 years); not recommended
- Sclerotherapy (ethanol): Emerging technique; lower recurrence than drainage alone
Deep Infiltrating Endometriosis (DIE) Surgery: [39]
Bowel Endometriosis:
- Shaving/disc excision: For superficial involvement (less than 50% circumference, less than 3 cm nodule)
- Preserves bowel continuity
- Lower morbidity
- "Anastomotic leak risk: less than 2%"
- Segmental resection: For full-thickness involvement, large nodules (> 3 cm), > 50% circumference
- Complete disease removal
- Higher morbidity
- "Anastomotic leak risk: 3-7%"
- "Temporary bowel dysfunction: 20-30% (usually resolves)"
- "Permanent stoma: less than 1%"
Bladder Endometriosis:
- Partial cystectomy (excision of bladder wall lesion)
- Postoperative catheterization: 7-14 days
- Complete symptom resolution: 70-85%
Ureteric Endometriosis:
- Ureterolysis (free ureter from disease)
- Partial ureterectomy + reimplantation if intrinsic involvement
- Ureteric stenting: 4-6 weeks postoperatively
- Renal function preservation: > 90% if treated before advanced hydronephrosis
Nerve-Sparing Surgery:
- Preservation of autonomic nerves (hypogastric, pelvic splanchnic)
- Reduces risk of bladder/bowel/sexual dysfunction
- Requires advanced surgical expertise
Hysterectomy ± Bilateral Salpingo-Oophorectomy (BSO)
Indications:
- Severe symptoms refractory to medical and conservative surgical treatment
- Completed family
- Patient fully counselled and consented
Evidence:
- Approximately 25% experience recurrent pelvic pain post-hysterectomy [2]
- 10% require further surgery (adhesiolysis, excision of residual disease)
- BSO reduces recurrence but induces surgical menopause (HRT required if less than 50 years)
Critical point: Hysterectomy is NOT a cure for endometriosis. Lesions outside the uterus can persist and recur.
Post-Operative Hormonal Suppression [40,41]
Evidence:
- Post-surgical medical treatment reduces recurrence of pain and disease
- Continuous COCP post-operatively: Reduces dysmenorrhoea recurrence by 50% (NNT = 5)
- Duration: At least 18-24 months recommended for maximum benefit
- No benefit if fertility desired immediately: Delays conception attempts
Recommended Post-Operative Protocols:
| Surgical Scenario | Post-Operative Medical Treatment |
|---|---|
| Excision of minimal/mild disease | COCP (continuous) or progestogen for 18-24 months |
| Excision of endometriomas | Continuous COCP (superior to cyclic); reduces recurrence from 35% to 15% at 2 years [40] |
| DIE excision (complete) | COCP or progestogen; consider GnRH agonist if incomplete excision |
| Incomplete excision / residual disease | GnRH agonist or dienogest for 6-12 months, then COCP long-term |
| Fertility desired | NO hormonal suppression; proceed with fertility treatment |
Mechanism:
- Suppresses microscopic residual disease
- Prevents new lesion formation
- Reduces inflammatory milieu
- Induces decidualization/atrophy of ectopic endometrium
Fertility Management
Impact on Fertility
- Endometriosis increases infertility risk 2-4 fold [6]
- Mechanisms: Anatomical distortion, adhesions, inflammatory milieu, impaired endometrial receptivity, reduced ovarian reserve (especially post-surgery) [18]
- Natural conception rates (without treatment):
- "Minimal/mild disease (rASRM I-II): Monthly fecundity rate 2-10% (vs. 15-20% in healthy couples)"
- "Moderate/severe disease (rASRM III-IV): Monthly fecundity rate less than 2%"
Management Options
| Scenario | Management |
|---|---|
| Minimal/mild disease (rASRM I-II) | Expectant management (natural conception); Laparoscopic excision may improve spontaneous pregnancy rates (Number Needed to Treat = 12) [34] |
| Moderate/severe disease (rASRM III-IV) | Laparoscopic excision of disease + adhesiolysis; Consider IVF if no conception within 6-12 months |
| Endometrioma > 3-4 cm | Consider cystectomy before IVF (improves access for oocyte retrieval); BUT risk of reduced ovarian reserve [19] |
| Tubal factor infertility | IVF/ICSI |
| Failed surgery / advanced age | Proceed directly to IVF/ICSI |
IVF Outcomes in Endometriosis (Systematic Reviews \u0026 Meta-Analyses): [9,35,36]
Live Birth Rates per Cycle:
- Minimal/mild endometriosis: 30-35% (comparable to tubal factor infertility)
- Moderate/severe endometriosis: 25-30% (slightly lower than other causes)
- Endometriomas present: 25-28% (vs. 32-35% in controls)
Oocyte Yield:
- Reduced in endometriosis: Mean 1-3 fewer oocytes retrieved per cycle [35]
- Ovarian reserve impact: Anti-Müllerian hormone (AMH) levels 30-40% lower in women with endometriomas
- Prior ovarian surgery: Further reduction (40-50% decrease in oocyte yield after cystectomy) [19]
Fertilization and Embryo Quality:
- Fertilization rates: Comparable to other causes of infertility (65-75%)
- Embryo quality: Some studies report lower blastocyst formation rates in severe endometriosis
- Implantation rates: Slightly reduced in moderate-severe disease (20-25% vs. 30-35% in controls)
Gonadotropin Requirements:
- Higher doses required: 20-30% increase in total gonadotropin dose in endometriosis patients [35]
- Longer stimulation duration: 1-2 days longer on average
- Poorer response to stimulation: Higher rates of "poor responders" (15-20% vs. 8-10% in tubal factor)
Cumulative Live Birth Rates (3 cycles):
- Minimal/mild endometriosis: 60-70%
- Moderate/severe endometriosis: 50-60%
- Comparable to other infertility causes overall
Pre-IVF Surgery: Evidence \u0026 Controversy [19,34,36]
Endometriomas \u003e3-4 cm:
Arguments FOR pre-IVF cystectomy:
- Improved access for oocyte retrieval
- Reduced risk of infection/abscess formation during oocyte retrieval
- Histological diagnosis (exclude malignancy)
- May improve IVF success rates (controversial)
Arguments AGAINST pre-IVF cystectomy:
- Significant risk of reduced ovarian reserve: AMH decline 30-60% post-surgery [19]
- Higher risk with bilateral cystectomy: May precipitate premature ovarian insufficiency
- No clear improvement in IVF outcomes: Meta-analyses show no significant benefit in live birth rates
- Recurrence risk: 15-20% within 2 years
Current Recommendation (ESHRE Guideline 2022):
- Endometriomas less than 3 cm: No surgery before IVF
- Endometriomas 3-4 cm: Individualized decision; consider observation
- Endometriomas > 4 cm: Consider surgery if:
- Difficult ovarian access anticipated
- Symptoms require treatment
- Concern for malignancy (rare, but risk increases with age > 40 years)
- AVOID repeat surgery on same ovary (high risk of ovarian failure)
Suppression Before IVF:
- GnRH agonist 3-6 months before IVF: May improve live birth rates in moderate-severe disease (RR 1.3-1.5) [36]
- Mechanism: Downregulation of inflammation, improved endometrial receptivity
- Cost-benefit: Delays treatment; consider in recurrent IVF failure
Fertility Preservation
Indications: [37]
- Women with endometriosis desiring future fertility but not currently trying to conceive
- Before ovarian surgery (especially bilateral or repeat surgery)
- Progressive disease with declining ovarian reserve
Options:
- Oocyte cryopreservation: Freeze eggs before AMH decline
- Embryo cryopreservation: If in partnership
- Ovarian tissue cryopreservation: Experimental; consider in young women before bilateral oophorectomy
Evidence:
- Oocyte survival post-thaw: 85-95%
- Fertilization rates: Comparable to fresh cycles
- Live birth rates per thawed oocyte: 5-8%
- Recommend freezing 15-20 mature oocytes for realistic chance of live birth [37]
Adjunctive Therapies
| Therapy | Evidence | Clinical Application | Notes |
|---|---|---|---|
| Pelvic floor physiotherapy | Moderate | First-line adjunct for all patients with chronic pelvic pain | Addresses myofascial pain component; Reduces dyspareunia (30-40% improvement); Improves pelvic floor muscle coordination; Typically 6-12 sessions required [48] |
| Cognitive behavioural therapy (CBT) | Moderate | Recommended for all patients with chronic pain or mental health comorbidity | Improves pain coping strategies; Reduces catastrophizing; Addresses anxiety/depression; Can be delivered individually or in groups; 8-12 sessions typical [49] |
| Acupuncture | Weak to Moderate | Consider as adjunct in motivated patients | Some RCTs show benefit for pain reduction (mean VAS reduction 10-15 mm); Variable quality of evidence; Mechanism unclear (endorphin release, neuromodulation); Requires weekly sessions [50] |
| TENS (Transcutaneous Electrical Nerve Stimulation) | Weak | Short-term pain relief | May provide temporary relief during menstruation; Non-invasive; Low risk; Use high-frequency settings (50-100 Hz) over suprapubic area [51] |
| Dietary modification | Weak | Individualized approach | Anti-inflammatory diet (vegetables, omega-3 fatty acids, limited red meat and processed foods); Some observational data suggests benefit but no high-quality RCTs; Gluten-free or low-FODMAP diets may help in patients with concurrent IBS [52] |
| Mindfulness-Based Stress Reduction (MBSR) | Emerging | Chronic pain management | 8-week structured program; Reduces pain catastrophizing; Improves quality of life; Increasingly integrated into pain management clinics [53] |
| Cannabis/CBD products | Insufficient Evidence | Not recommended routinely | Anecdotal reports of benefit; Lack of high-quality clinical trials; Legal and regulatory issues; Potential for adverse effects; Further research needed [54] |
Emerging Therapies \u0026 Future Directions [42,43,44]
Novel Pharmacological Approaches
GnRH Antagonists (Oral):
- Elagolix (approved in US, not UK): 150-200 mg BD
- Non-inferior to GnRH agonists for pain relief
- Fewer menopausal symptoms (no initial "flare" effect)
- Dose-dependent bone loss (less than GnRH agonists)
- Expensive; limited availability
- Relugolix (combination with add-back HRT): Once-daily oral; Phase III trials show efficacy
- Linzagolix: Under investigation; selective dosing to preserve partial estrogen (minimize bone loss)
Selective Progesterone Receptor Modulators (SPRMs):
- Ulipristal acetate: Previously used; withdrawn due to hepatotoxicity concerns
- Vilaprisan: Under investigation; preliminary trials show efficacy
- Mechanism: Progesterone receptor modulation without full agonist/antagonist effects
Anti-Angiogenic Therapies:
- Bevacizumab (anti-VEGF antibody): Pilot studies show promise in reducing lesion vascularity
- Mechanism: Inhibit neovascularization of endometriotic lesions
- Status: Experimental; not in clinical practice
Immunomodulators:
- TNF-α inhibitors (infliximab, etanercept): Early trials showed no benefit; abandoned
- IL-6 inhibitors: Under investigation
- Pentoxifylline: Anti-inflammatory; meta-analysis shows no benefit for pain or fertility
Aromatase Inhibitors (Third-Line):
- Letrozole 2.5 mg daily (with COCP or progestogen to prevent ovarian cysts)
- Reserved for refractory cases
- Limited evidence; off-label use
Novel Targets Under Investigation:
- Matrix metalloproteinase (MMP) inhibitors: Reduce invasion and fibrosis
- Histone deacetylase (HDAC) inhibitors: Epigenetic modulation
- Statins: Anti-inflammatory and anti-proliferative effects (observational data)
- Neurotrophin inhibitors: Targeting nerve growth in lesions (for pain)
Non-Hormonal Approaches
Targeted Small Molecule Inhibitors:
- MEK inhibitors: Targeting MAPK signaling pathway
- mTOR inhibitors: Reduce cell proliferation in endometriotic lesions
- Status: Preclinical and early phase trials
Stem Cell Therapies:
- Hypothesis: Endometriosis involves endometrial stem/progenitor cells
- Research into stem cell origin and potential targeted therapies
- Status: Basic science research phase
Diagnostic Biomarkers (Active Research) [43]
Goal: Non-invasive diagnosis to replace laparoscopy
Candidate Biomarkers (none yet validated for clinical use):
- Serum panels: CA-125 + CA-19-9 + inflammatory markers (IL-6, IL-8, TNF-α)
- MicroRNAs: miR-199a, miR-542-3p show promise in pilot studies
- Metabolomics: Urine/serum metabolite profiling
- Endometrial biopsy markers: Nerve fiber density, HOXA10 expression
Challenges:
- Overlap with other conditions (PID, fibroids, adenomyosis)
- Variable sensitivity across disease stages
- Lack of large validation cohorts
Minimally Invasive Diagnostics
Endometrial receptivity analysis (ERA):
- Assess endometrial gene expression
- May identify "endometriosis signature" in eutopic endometrium
- Status: Research tool; not diagnostic
Saliva/urine tests:
- Early research into non-invasive sampling
- Hormonal and inflammatory markers
- Status: Experimental; no validated tests available
Personalized Medicine Approaches [44]
Pharmacogenomics:
- Genetic variants affecting treatment response (e.g., CYP450 polymorphisms affecting hormonal metabolism)
- Tailor treatment based on genetic profile
- Status: Future direction; not in clinical practice
Disease Phenotyping:
- Recognize different disease subtypes (superficial peritoneal, ovarian, DIE)
- Each may require different treatment approaches
- Move away from "one-size-fits-all" management
Patient-Reported Outcome Measures (PROMs):
- Standardized pain and quality-of-life questionnaires
- Endometriosis Health Profile-30 (EHP-30)
- Guide individualized treatment decisions
8. Complications
Disease-Related Complications
| Complication | Incidence | Notes |
|---|---|---|
| Infertility | 30-50% | Leading indication for IVF |
| Chronic pain | Majority | Significant impact on quality of life |
| Endometrioma rupture | Rare | Acute abdomen; Chemical peritonitis |
| Ovarian torsion | Rare | Endometriomas predispose; Surgical emergency |
| Bowel obstruction | Rare | Deep infiltrating disease |
| Ureteric obstruction | 1-2% of DIE | Hydronephrosis; Silent renal loss if bilateral |
| Malignant transformation | less than 1% | Endometrioid or clear cell ovarian carcinoma; Very rare |
| Spontaneous hemoperitoneum in pregnancy | Rare | DIE; Life-threatening; Requires emergency laparotomy |
Obstetric Complications
Women with endometriosis (particularly deep infiltrating disease) have increased risk of:
- Placenta previa (several-fold increased risk) [23]
- Preterm delivery
- Small for gestational age (SGA)
- Preeclampsia (adenomyosis association stronger)
- Postpartum haemorrhage
- Caesarean section complications (adhesions; bleeding)
Surgical Complications
| Complication | Risk | Notes |
|---|---|---|
| Reduced ovarian reserve | 10-40% after ovarian surgery | Especially bilateral or repeat surgery; Monitor AMH [19] |
| Bladder injury | 1-2% | Deep DIE surgery |
| Ureteric injury | less than 1% | DIE surgery; May require reimplantation |
| Bowel injury | less than 1% | Adhesiolysis |
| Anastomotic leak | 2-5% | Bowel resection for DIE |
| Bladder/bowel dysfunction | Variable | Nerve injury during DIE surgery |
| Stoma formation | Rare | If anastomotic leak or extensive bowel resection |
Mental Health Complications
- Depression: 2-3 fold increased risk [10]
- Anxiety: 2-3 fold increased risk [10]
- Sexual dysfunction: 40-60% due to dyspareunia and psychological impact
- Eating disorders: Emerging evidence of association
- Social isolation: Chronic pain and diagnostic delay impact relationships and work
9. Prognosis & Long-Term Outcomes
Recurrence Rates
| Treatment | Recurrence Rate | Time Frame |
|---|---|---|
| Hormonal suppression | 25-34% recurrent pain | Within 12 months of stopping [2] |
| Laparoscopic excision | 15-20% | 5 years [8] |
| Laparoscopic ablation | 40-50% | 5 years [8] |
| Hysterectomy (conserving ovaries) | 10-15% | Long-term |
| Hysterectomy + BSO | 5-10% | Long-term |
Natural History
- Menopause: Symptoms typically improve after natural menopause (loss of estrogen drive)
- Pregnancy: Temporary symptom relief during pregnancy (high progesterone); Symptoms often recur postpartum
- Progression: Variable; Some women have stable minimal disease, others progress to severe DIE
Quality of Life
- Endometriosis significantly impacts health-related quality of life (HRQoL) across physical, psychological, and social domains
- Work productivity reduced: 11 hours lost per week on average [4]
- Relationship strain: Dyspareunia and chronic pain affect intimacy
- Diagnostic delay compounds psychological distress
Cancer Risk
- Ovarian cancer: Slight increased risk (RR 1.3-1.9) for endometrioid and clear cell subtypes [24]
- Breast cancer: Conflicting evidence; possible slight increase
- No increased risk: Cervical, endometrial (unless concurrent Lynch syndrome)
Cardiovascular Risk
- Emerging evidence suggests increased cardiovascular disease risk, particularly in women with surgical history (possibly mediated by early menopause) [12]
10. Special Populations
Adolescents
- Endometriosis can begin in adolescence; average age of symptom onset is late teens to early 20s
- High index of suspicion if severe dysmenorrhoea unresponsive to NSAIDs
- Empirical hormonal treatment preferred (avoid laparoscopy unless refractory)
- Fertility preservation counselling important
Pregnancy
- Pre-conception counselling: Discuss obstetric risks (placenta previa, preterm delivery)
- Spontaneous hemoperitoneum: Rare but life-threatening complication (DIE); High index of suspicion for acute abdominal pain
- Pain management: Paracetamol safe; Avoid NSAIDs (especially third trimester); Opioids if required
Menopause
- Natural menopause: Symptoms usually improve
- HRT use: Continuous combined HRT may reactivate disease; Tibolone may be safer option; Progestogen essential if residual disease and unopposed estrogen avoided
- Surgical menopause: Requires HRT until natural menopause age; Weigh risks/benefits
11. Evidence & Guidelines
Key Guidelines
| Guideline | Organisation | Year | Key Recommendations |
|---|---|---|---|
| Endometriosis: diagnosis and management (NG73) | NICE | 2017 (updated 2024) | Empirical hormonal treatment; Laparoscopy for definitive diagnosis; Excision preferred over ablation |
| ESHRE Guideline: Endometriosis | ESHRE | 2022 | Comprehensive guideline covering diagnosis, medical and surgical treatment, fertility |
| ACOG Practice Bulletin No. 114 | ACOG | 2010 (reaffirmed 2024) | US guideline on management |
| Endometriosis and Mental Health | Various | 2024 | Screen for mental health comorbidities; Holistic approach [10] |
Landmark Studies & Meta-Analyses
- Zondervan et al. NEJM 2020 [3]: Comprehensive review of endometriosis pathophysiology and management
- As-Sanie et al. JAMA 2025 [2]: Updated review on epidemiology, diagnosis, and treatment outcomes
- Vercellini et al. Fertil Steril 2023 [23]: Endometriosis and pregnancy/obstetric outcomes
- Saha et al. Fertil Steril 2015 [7]: Heritability study demonstrating 47% genetic contribution
Quality of Evidence
- Diagnosis: High-quality evidence for TVUS/MRI sensitivity and specificity
- Medical treatment: Network meta-analyses show similar efficacy across hormonal options [2]
- Surgical treatment: Moderate-quality evidence favoring excision over ablation [8]
- Fertility: Moderate-quality evidence for IVF outcomes
12. Patient Information
What is Endometriosis?
Endometriosis is a common condition where tissue similar to the lining of your womb (uterus) grows outside the womb—on your ovaries, fallopian tubes, bladder, bowel, or other areas in your pelvis. This tissue responds to your monthly hormones just like the womb lining does, causing bleeding and inflammation each month. However, because the blood has nowhere to go, it causes pain, scarring, and sometimes cysts.
What Causes It?
The exact cause is not fully understood, but it may involve:
- Menstrual blood flowing backward through your fallopian tubes and implanting in the pelvis
- Genetic factors (it runs in families)
- Immune system problems that fail to clear the misplaced tissue
What Are the Symptoms?
Common symptoms include:
- Painful periods that don't improve with painkillers
- Pelvic pain between periods or all the time
- Pain during or after sex (especially deep penetration)
- Difficulty getting pregnant
- Painful bowel movements or urination during your period
- Heavy periods or bleeding between periods
- Tiredness and low energy
Important: Some women with endometriosis have no symptoms at all.
How Is It Diagnosed?
- Clinical assessment: Your doctor will ask about your symptoms and examine you
- Ultrasound scan: A scan through the vagina can show cysts (endometriomas) or other signs
- MRI scan: May be needed if deeper disease is suspected
- Laparoscopy (keyhole surgery): The only definitive way to diagnose endometriosis—a camera is inserted through a small cut near your belly button to look inside your pelvis
How Is It Treated?
Treatment depends on your symptoms, whether you want to get pregnant, and how severe the disease is.
Medical (Non-Surgical) Options:
- Painkillers: Ibuprofen or mefenamic acid for pain
- Hormonal treatments:
- Combined contraceptive pill (taken continuously without breaks)
- Progestogen-only pill or Mirena coil
- Injections (GnRH agonists) that temporarily stop your periods—these cause menopause-like symptoms but can be very effective
Surgical Options:
- Laparoscopic surgery: Keyhole surgery to remove or destroy endometriosis tissue—can significantly improve pain and fertility
- Hysterectomy: Removal of the womb (and sometimes ovaries) as a last resort if other treatments haven't worked and you don't want children in the future
If You Want to Get Pregnant:
- Hormonal treatments won't help you conceive (they prevent pregnancy)
- Surgery to remove endometriosis may improve your chances of natural conception
- IVF (in vitro fertilisation) may be recommended if you have moderate to severe endometriosis or haven't conceived after surgery
Will It Go Away?
Endometriosis is a long-term condition. Treatments can control symptoms, but it often comes back after treatment stops. Symptoms usually improve naturally after menopause when your periods stop.
Where Can I Get Support?
- Endometriosis UK: www.endometriosis-uk.org – charity offering information and support groups
- Your GP or gynaecologist: Don't hesitate to ask for help managing symptoms
- Mental health support: Chronic pain can affect your mood—ask about counselling or support services
Lifestyle Modifications \u0026 Self-Management
Evidence-Based Strategies:
- Heat therapy: Hot water bottles, heat pads for acute pain relief
- Regular exercise: May reduce pain severity; aim for 150 minutes/week moderate activity
- Stress management: Mindfulness, yoga, meditation (improves pain coping)
- Sleep hygiene: Chronic pain worsens with poor sleep; prioritize 7-8 hours
- Dietary considerations:
- Anti-inflammatory diet (vegetables, omega-3 fatty acids, limited processed foods)
- Some women report benefit from gluten-free or low-FODMAP diets (limited evidence)
- Avoid excessive caffeine and alcohol if worsens symptoms
Support Networks:
- Online communities and peer support groups
- Endometriosis advocacy organizations (national and international)
- Employer accommodations (flexible working, time off for medical appointments)
13. Examination Focus (MRCOG, FRANZCOG, FRACP)
High-Yield Exam Topics
| Topic | Key Examination Points |
|---|---|
| Prevalence & epidemiology | 10% of reproductive women; 25-50% of infertile women; 47% heritability |
| Symptoms | Dysmenorrhoea (80%), pelvic pain (70%), dyspareunia (40-50%), infertility (30-50%) |
| Pathophysiology | Retrograde menstruation theory; estrogen-dependent; inflammatory cascade; progesterone resistance |
| Diagnosis | TVUS first-line (93% sensitive for endometrioma); MRI for DIE; Laparoscopy gold standard |
| Staging | rASRM I-IV; Does NOT correlate with pain severity |
| Medical management | COCP/progestogens first-line; GnRH agonists second-line (add-back HRT > 6 months); 25-34% recurrence within 12 months of stopping |
| Surgical management | Excision > ablation (lower recurrence); Ovarian surgery risks reduced ovarian reserve |
| Fertility | Surgery or IVF (not hormonal suppression); IVF outcomes similar to other causes |
| Complications | Bowel/ureteric obstruction (DIE); spontaneous hemoperitoneum in pregnancy; ovarian cancer risk (1.3-1.9 RR) |
Sample Viva Questions
Question 1: Management of Suspected Endometriosis
Q: A 28-year-old nulliparous woman presents with 3-year history of severe dysmenorrhoea, deep dyspareunia, and chronic pelvic pain. She has tried NSAIDs with minimal benefit. How would you investigate and manage her?
Model Answer:
History: I would take a detailed history focusing on:
- Character and cyclical nature of pain
- Impact on quality of life and sexual function
- Bowel/bladder symptoms (suggestive of DIE)
- Fertility wishes
- Previous treatments tried
- Family history (7-fold increased risk in first-degree relatives)
Examination: Bimanual and rectovaginal examination to assess for:
- Uterosacral nodularity
- Fixed retroverted uterus
- Adnexal masses
- Pouch of Douglas tenderness
Investigations:
- TVUS: First-line imaging to assess for endometriomas (ground-glass echogenicity) and signs of DIE
- Consider MRI if deep disease suspected on examination or TVUS
- Bloods: CA-125 (may be elevated but non-specific); FBC (check for anaemia)
Management (as she doesn't currently desire pregnancy):
-
Empirical medical treatment (can start without laparoscopy if high clinical suspicion):
- First-line: COCP continuous regimen or progestogen (Mirena IUS/oral)
- Analgesia: NSAIDs + paracetamol
-
If refractory to first-line (after 3-6 months trial):
- Second-line: GnRH agonist (e.g., goserelin 3.6 mg monthly SC) with add-back HRT (tibolone or low-dose combined HRT) to prevent bone loss
- Consider laparoscopy for definitive diagnosis and surgical treatment
-
Surgical management if medical treatment fails or patient preference:
- Laparoscopic excision (preferred over ablation—lower recurrence)
- Histological confirmation
- rASRM staging
-
Adjunctive:
- Pelvic floor physiotherapy
- Psychological support (screen for anxiety/depression)
- Pain management clinic if refractory
-
Follow-up:
- Review response to treatment at 3-6 months
- Counsel on recurrence rates (25-34% within 12 months of stopping hormonal treatment)
- Fertility counselling if she wishes to conceive in future
Question 2: Endometriosis and Fertility
Q: A 32-year-old woman with laparoscopically confirmed Stage III endometriosis (moderate disease) has been trying to conceive for 18 months. How would you counsel and manage her?
Model Answer:
Initial assessment:
- Confirm previous surgical diagnosis and staging (rASRM III)
- Review operative notes (extent of excision, presence of endometriomas, tubal patency assessment)
- Assess ovarian reserve (AMH, AFC on ultrasound)
- Partner semen analysis
- Assess current symptoms (if symptomatic, may need medical management vs. if asymptomatic, focus on fertility)
Counselling:
- Endometriosis increases infertility risk 2-4 fold
- Mechanisms: Anatomical distortion, adhesions, inflammatory milieu, impaired endometrial receptivity
- Hormonal suppression does NOT improve fertility outcomes—avoid prolonged treatment if trying to conceive
Management options:
-
If no previous surgery or incomplete excision:
- Laparoscopic surgery: Excision of disease + adhesiolysis may improve spontaneous conception rates (particularly mild-moderate disease)
- Caution with ovarian surgery (endometrioma excision risks reduced ovarian reserve)
-
If already had complete surgical excision:
- Expectant management for 6-12 months if:
- Age less than 35 years
- Adequate ovarian reserve
- Patent tubes
- Normal semen analysis
- Proceed to IVF/ICSI if:
- Age ≥35 years
- Reduced ovarian reserve
- Tubal factor
- Severe male factor
- Failed expectant management
- Expectant management for 6-12 months if:
-
IVF considerations:
- Live birth rates in endometriosis similar to other causes of infertility
- May require higher gonadotropin doses
- Endometrioma > 4 cm: Consider cystectomy before IVF (improves access for oocyte retrieval) BUT risk of reduced ovarian reserve—balance risks/benefits
-
Fertility preservation:
- Discuss oocyte/embryo freezing if further ovarian surgery planned or progressive disease
Follow-up: Review after 6-12 months expectant or proceed directly to ART depending on factors above.
Question 3: Deep Infiltrating Endometriosis
Q: A 35-year-old woman is found to have a 3 cm rectovaginal nodule on MRI, suspected deep infiltrating endometriosis. She has severe cyclical dyschezia. How would you manage her?
Model Answer:
Multidisciplinary approach essential:
- Gynaecology
- Colorectal surgery
- Urology (if ureteric involvement)
- Radiology
Pre-operative assessment:
- MRI pelvis: Best modality for mapping DIE extent (bowel wall involvement, distance from anal verge, bladder/ureteric involvement)
- Colonoscopy/sigmoidoscopy: Assess bowel mucosa (endometriosis usually spares mucosa but can cause extrinsic compression)
- Renal function & imaging: Exclude ureteric obstruction/hydronephrosis
Counselling:
- Discuss surgical options, risks, and benefits
- Risks specific to DIE surgery:
- Bowel resection may be required (segmental vs. disc excision)
- Anastomotic leak (2-5%)
- Bladder/bowel dysfunction (nerve injury)
- Temporary or permanent stoma (rare)
- Reduced fertility if extensive surgery
- Fertility wishes: If desiring future pregnancy, discuss timing of surgery vs. IVF
Surgical planning:
- Specialist centre with multidisciplinary team experienced in DIE surgery
- Options:
- Bowel-sparing techniques: Shaving/disc excision of rectal nodule (if less than 3 cm, no full-thickness involvement)
- Segmental bowel resection: If extensive disease, full-thickness involvement, or nodule > 3 cm
- Complete excision of all visible disease
Post-operative:
- Hormonal suppression to prevent recurrence (COCP, progestogen, or GnRH agonist)
- Monitor for complications (anastomotic leak, bladder dysfunction)
- Fertility counselling if relevant
Alternative: Medical management (GnRH agonist, dienogest) if patient declines surgery or high surgical risk—but DIE often poorly responsive to medical treatment alone.
Question 4: CA-125 in Endometriosis
Q: What is the role of CA-125 in the diagnosis and management of endometriosis?
Model Answer:
Diagnostic Utility:
- Limited role: CA-125 is NOT diagnostic for endometriosis
- Sensitivity: Only 20-50% (poor—misses most cases)
- Specificity: 70-90% (limited—many false positives)
- NICE Guideline (NG73): Do NOT use CA-125 to diagnose endometriosis in primary or secondary care
Levels in Endometriosis:
- Usually less than 200 U/mL (mean 35-50 U/mL in endometriosis vs. 10-20 U/mL in controls)
- Higher in advanced disease (rASRM III-IV) compared to minimal disease
- Levels > 200 U/mL raise suspicion for ovarian malignancy—require thorough investigation
Causes of Elevated CA-125 (limits specificity):
- Menstruation (physiological)
- Pregnancy (first trimester)
- Pelvic inflammatory disease
- Uterine fibroids
- Adenomyosis
- Ovarian cysts (benign)
- Ovarian malignancy—critical differential
- Peritoneal irritation (any cause)
Potential Clinical Uses (limited):
- Pre-operative assessment: Elevated levels may suggest moderate-severe disease (but NOT diagnostic)
- Monitoring treatment response: Serial CA-125 measurements may track disease activity
- Recurrence prediction: Rising CA-125 post-treatment may indicate disease recurrence
- Research: Part of biomarker panels under investigation
Current Evidence:
- No validated serum biomarkers exist for endometriosis diagnosis
- Research ongoing into biomarker panels (CA-125 + CA-19-9 + inflammatory markers)
- Meta-analysis shows insufficient accuracy for clinical diagnosis
Conclusion: CA-125 alone should NOT be used for diagnosis. It may serve as an adjunct in monitoring known disease, but imaging and clinical assessment are far more valuable.
Question 5: Imaging Modalities Comparison
Q: Compare the sensitivity and specificity of transvaginal ultrasound (TVUS) and MRI for diagnosing different types of endometriosis.
Model Answer:
Ovarian Endometriomas:
- TVUS: Sensitivity 93%, Specificity 96%—"ground-glass" echogenicity pathognomonic
- MRI: Sensitivity 90-95%, Specificity 91-98%—T1 hyperintense with T2 "shading"
- Conclusion: TVUS and MRI have comparable accuracy; TVUS preferred first-line (cost-effective, accessible)
Deep Infiltrating Endometriosis (DIE):
| Anatomical Site | TVUS Sensitivity | TVUS Specificity | MRI Sensitivity | MRI Specificity |
|---|---|---|---|---|
| Rectovaginal septum | 76% | 97% | 90-94% | 77-91% |
| Bowel | 79% | 94% | 85-95% | 90-95% |
| Bladder | 53% | 99% | 88-90% | 99-100% |
| Uterosacral ligaments | 64% | 97% | 74-83% | 70-97% |
MRI Advantages for DIE:
- Superior for surgical planning (maps extent, depth of invasion, distance from anal verge)
- Better for ureteric involvement and hydronephrosis
- Multilayer assessment of bowel wall
- Identifies adenomyosis (junctional zone thickening)
TVUS Advantages:
- First-line: Accessible, cost-effective
- Dynamic assessment (sliding sign, tenderness-guided)
- Excellent for endometriomas
- Can be repeated frequently
Superficial Peritoneal Disease:
- Both modalities have poor sensitivity (less than 50%)
- Most common presentation of endometriosis
- Laparoscopy remains gold standard for superficial disease
Clinical Decision Algorithm:
- First-line: TVUS for all suspected endometriosis
- Second-line: MRI if:
- Deep infiltrating disease suspected on TVUS or examination
- Surgical planning required (especially bowel/bladder involvement)
- TVUS inconclusive but high clinical suspicion
- Gold standard: Laparoscopy with histology for definitive diagnosis
IDEA Consensus (International Deep Endometriosis Analysis): Standardized TVUS protocols improve accuracy; systematic evaluation essential.
Question 7: Management of Endometriosis in Adolescents
Q: A 16-year-old presents with severe dysmenorrhoea causing regular school absence despite NSAIDs. How would you approach diagnosis and management?
Model Answer:
Key Principles:
- Endometriosis can begin in adolescence (average symptom onset late teens)
- High index of suspicion if dysmenorrhoea interferes with daily activities
- Empirical treatment preferred; avoid laparoscopy unless absolutely necessary
- Preserve fertility potential
Initial Assessment:
- Detailed menstrual and pain history (onset, severity, impact on school/activities)
- Screen for red flags (bowel/bladder symptoms, cyclical symptoms)
- Family history (7-fold risk if first-degree relative affected)
- Psychosocial impact assessment (school performance, social activities, mental health)
- Exclude structural abnormalities (Müllerian anomalies, imperforate hymen)
Examination:
- Abdominal examination (exclude masses, tenderness)
- External genitalia (exclude anatomical abnormalities)
- Avoid internal examination in virgin adolescents unless specific indication
- If sexually active, bimanual examination as per adult protocol
Investigations:
- First-line: Pelvic ultrasound (transabdominal if virgin; transvaginal if sexually active)
- Look for endometriomas, structural abnormalities
- May be normal in early disease
- Avoid: Routine CA-125 (not diagnostic)
- Consider MRI: If complex mass or structural abnormality suspected
Management:
First-Line (Empirical Medical Treatment):
- NSAIDs: Mefenamic acid or ibuprofen (start 1-2 days before menses)
- Hormonal suppression:
- COCP (continuous or tricycle regimen)—first choice
- Progestogen-only pill (desogestrel 75 mcg daily) if COCP contraindicated
- Mirena IUS if sexually active and needs contraception
Second-Line:
- Dienogest 2 mg daily if inadequate response to COCP
- GnRH agonists: Reserved for severe refractory cases due to bone density concerns in growing adolescents (use with add-back HRT; monitor bone density)
When to Consider Laparoscopy:
- Failed 6-12 months empirical medical treatment
- Severe symptoms significantly impacting quality of life
- Suspected endometrioma greater than 3 cm
- Patient/family preference for definitive diagnosis
- Counsel: Surgical risks, diagnostic yield, potential for normal findings
Multidisciplinary Support:
- Educational support: Liaise with school for accommodations (flexible attendance, exam arrangements)
- Psychological support: Address anxiety, depression, social isolation
- Peer support: Endometriosis support groups (youth-focused if available)
- Fertility counseling: Reassure that diagnosis in adolescence doesn't mean infertility; discuss long-term fertility preservation options if extensive surgery required
Follow-Up:
- Review at 3-6 months
- Assess treatment response, side effects, psychosocial impact
- Transition planning to adult services at age 18
Counseling Points:
- Endometriosis is a chronic condition requiring long-term management
- Most adolescents respond well to hormonal treatment
- Early treatment may prevent disease progression
- Fertility is usually preserved
Question 8: Post-Hysterectomy Pain Management
Q: A 42-year-old woman continues to have severe pelvic pain 6 months after total abdominal hysterectomy with bilateral salpingo-oophorectomy for endometriosis. How would you manage her?
Model Answer:
Differential Diagnosis for Post-Hysterectomy Pain:
- Residual endometriosis: Lesions on bowel, bladder, peritoneum not excised at time of hysterectomy
- Adhesions: Post-surgical adhesions causing pain
- Pelvic floor dysfunction: Myofascial pain, muscle spasm
- Neuropathic pain: Nerve injury or entrapment during surgery
- Ovarian remnant syndrome: If ovaries incompletely excised (BSO)
- Other pathology: Unrelated causes (IBS, pelvic congestion, musculoskeletal)
Assessment:
- History: Pain characteristics (location, cyclical vs. constant, triggers)
- Review operative notes: Extent of disease excised, completeness of surgery
- Examination: Pelvic tenderness, trigger points, scar assessment
- HRT status: If BSO performed, confirm on adequate HRT (may reactivate residual disease if estrogen-only)
Investigations:
- TVUS: Assess for masses, fluid collections, ovarian remnant
- MRI pelvis: Superior for detecting residual deep infiltrating disease
- Consider colonoscopy/cystoscopy: If bowel/bladder symptoms persist
- Diagnostic laparoscopy: If imaging inconclusive but high clinical suspicion
Management:
1. Modify HRT (if applicable):
- If on estrogen-only HRT: Switch to continuous combined HRT or tibolone (progestogen opposes residual endometriosis)
- If not on HRT post-BSO: Start HRT but use progestogen-containing regimen
- Consider stopping HRT temporarily (3-6 months) to assess if symptoms resolve
2. Medical Management:
- Progestogen therapy: Dienogest 2 mg daily or norethisterone 10-15 mg daily
- Aromatase inhibitors: Letrozole 2.5 mg daily (with HRT/COCP to prevent bone loss) for refractory cases
- Neuropathic agents: Amitriptyline 10-75 mg nocte, gabapentin 300-900 mg TDS if neuropathic component
3. Surgical Options:
- Diagnostic laparoscopy: Confirm residual disease; excise visible lesions
- Excision of residual endometriosis: Requires specialist with expertise in deep disease
- Adhesiolysis: If adhesions identified
- Presacral neurectomy: Reserved for refractory midline pain (specialist procedure)
4. Multidisciplinary Approach:
- Pain management clinic: For chronic pain strategies, medication optimization
- Pelvic floor physiotherapy: Addresses myofascial component
- Psychological support: CBT for chronic pain coping
- Consider spinal cord stimulation: For severe refractory neuropathic pain (specialist centres)
Counseling:
- Explain that hysterectomy is not curative for endometriosis
- Residual disease outside uterus can persist and cause symptoms
- Approximately 25% experience recurrent pain post-hysterectomy [2]
- Prognosis depends on completeness of original excision and location of residual disease
Follow-Up:
- Structured pain assessment (VAS, pain diary)
- Monitor response to treatment modifications
- Consider referral to tertiary endometriosis centre if refractory
Question 6: Dienogest vs. GnRH Agonists
Q: A 29-year-old woman with laparoscopically confirmed endometriosis has failed first-line treatment with continuous COCP. Compare dienogest and GnRH agonists as second-line options.
Model Answer:
Efficacy for Pain (head-to-head RCTs):
- Non-inferior: Dienogest 2 mg daily shows comparable pain reduction to GnRH agonists [32,33]
- Pain reduction: Both achieve 70-80% reduction in VAS pain scores
- Dysmenorrhoea, dyspareunia, chronic pelvic pain: Similar improvement rates
- Time to response: Dienogest 4-8 weeks; GnRH agonist 8-12 weeks (after initial flare)
Side Effect Profile:
| Parameter | Dienogest | GnRH Agonist |
|---|---|---|
| Menopausal symptoms | Mild (10-20%) | Severe (80-90%)—hot flushes, night sweats |
| Bone mineral density | No significant loss | 4-6% loss at lumbar spine after 6 months [30] |
| Bleeding pattern | Irregular bleeding (30-50%, decreases over time) | Amenorrhoea (after initial flare) |
| Mood changes | 10-15% | 20-30% |
| Discontinuation rate | 15-20% | 20-30% (without add-back HRT) |
| Quality of life | Better preserved (fewer menopausal symptoms) | Often impaired by hypoestrogenic effects |
Mechanism Comparison:
- Dienogest: Selective progestogen; decidualization and atrophy of endometrium; anti-inflammatory effects; partial anti-androgenic activity
- GnRH agonist: Pituitary downregulation; medical menopause; profound estrogen suppression (estradiol less than 20 pg/mL)
Cost:
- Dienogest: Moderate cost (branded medication; approximately £25-30/month in UK)
- GnRH agonist: Expensive (monthly injections £80-100; additional cost of add-back HRT £10-15/month)
Duration of Treatment:
- Dienogest: Can be used long-term (no bone loss restriction); continuous use until pregnancy desired or menopause
- GnRH agonist: Limited to 6 months without add-back HRT (bone loss); can extend with add-back HRT for up to 2 years
Add-Back HRT:
- Dienogest: NOT required
- GnRH agonist: Essential if treatment greater than 6 months (tibolone 2.5 mg or low-dose combined HRT); prevents bone loss and reduces menopausal symptoms
Recurrence After Stopping:
- Both: Similar recurrence rates (25-34% within 12 months)
- Time to recurrence: Median 6-12 months after discontinuation
Patient Preference Factors:
- Dienogest: Oral daily; fewer menopausal symptoms; can use long-term; better preservation of quality of life
- GnRH agonist: Monthly injection (or 3-monthly depot formulation); profound symptom relief but more side effects; guaranteed amenorrhoea
Contraindications:
- Dienogest: Undiagnosed vaginal bleeding; active VTE; hormone-dependent malignancy
- GnRH agonist: Osteoporosis (relative); undiagnosed vaginal bleeding; hormone-dependent malignancy
Recommendation: For this 29-year-old woman:
-
First choice: Dienogest 2 mg daily
- Comparable efficacy
- Better side effect profile (especially menopausal symptoms in young woman)
- No bone loss concerns
- Can be used long-term
- Lower cost overall (no need for add-back HRT)
- Better preservation of quality of life and sexual function
-
Consider GnRH agonist if:
- Patient desires profound amenorrhoea (e.g., for occupational reasons, heavy bleeding)
- Dienogest fails or not tolerated (e.g., persistent irregular bleeding)
- Pre-surgical downregulation of disease (3-6 months before IVF or major surgery)
- Patient preference after full counseling
Follow-up: Review at 3 months; assess pain relief (VAS score), bleeding pattern, side effects, and quality of life (EHP-30). If effective and well-tolerated, continue long-term with annual review.
14. References
-
Zondervan KT, Becker CM, Missmer SA. Endometriosis. N Engl J Med. 2020;382(13):1244-1256. doi:10.1056/NEJMra1810764
-
As-Sanie S, Mackenzie SC, Morrison L, et al. Endometriosis: A Review. JAMA. 2025;334(1):64-78. doi:10.1001/jama.2025.2975
-
Zondervan KT, Becker CM, Missmer SA. Endometriosis. N Engl J Med. 2020;382(13):1244-1256. doi:10.1056/NEJMra1810764
-
Taylor HS, Kotlyar AM, Flores VA. Endometriosis is a chronic systemic disease: clinical challenges and novel innovations. Lancet. 2021;397(10276):839-852.
-
Zippl AL, Reiser E, Seeber B. Endometriosis and mental health disorders: identification and treatment as part of a multimodal approach. Fertil Steril. 2024;121(3):370-378. doi:10.1016/j.fertnstert.2023.12.033
-
Leone Roberti Maggiore U, Chiappa V, Ceccaroni M, et al. Epidemiology of infertility in women with endometriosis. Best Pract Res Clin Obstet Gynaecol. 2024;92:102454. doi:10.1016/j.bpobgyn.2023.102454
-
Saha R, Pettersson HJ, Svedberg P, et al. Heritability of endometriosis. Fertil Steril. 2015;104(4):947-952. doi:10.1016/j.fertnstert.2015.06.035
-
Dunselman GA, Vermeulen N, Becker C, et al. ESHRE guideline: management of women with endometriosis. Hum Reprod. 2014;29(3):400-412.
-
Barnhart K, Dunsmoor-Su R, Coutifaris C. Effect of endometriosis on in vitro fertilization. Fertil Steril. 2002;77(6):1148-1155.
-
Zippl AL, Reiser E, Seeber B. Endometriosis and mental health disorders: identification and treatment as part of a multimodal approach. Fertil Steril. 2024;121(3):370-378. doi:10.1016/j.fertnstert.2023.12.033
-
Chiaffarino F, Cipriani S, Ricci E, et al. Endometriosis and irritable bowel syndrome: a systematic review and meta-analysis. Arch Gynecol Obstet. 2021;303(1):17-25. doi:10.1007/s00404-020-05797-8
-
Younis JS, Taylor HS. Is There an Association Between Endometriosis, Early Menopause, and Cardiovascular Disease? J Clin Endocrinol Metab. 2024;109(10):e1946-e1949. doi:10.1210/clinem/dgae508
-
Christ JP, Yu O, Schulze-Rath R, et al. Incidence, prevalence, and trends in endometriosis diagnosis: a United States population-based study from 2006 to 2015. Am J Obstet Gynecol. 2021;225(5):500.e1-500.e9. doi:10.1016/j.ajog.2021.06.067
-
Moradi Y, Shams-Beyranvand M, Khateri S, et al. A systematic review on the prevalence of endometriosis in women. Indian J Med Res. 2021;154(3):446-454. doi:10.4103/ijmr.IJMR_817_18
-
Chen Y, Liu C, Wang X, Liu Y, Liu H. Global, Regional and National Burden of Infertility due to Endometriosis: Results From the Global Burden of Disease Study 2021 and Forecast to 2044. BJOG. 2025;132(7):944-960. doi:10.1111/1471-0528.18108
-
Parazzini F, Esposito G, Tozzi L, Noli S, Bianchi S. Epidemiology of endometriosis and its comorbidities. Eur J Obstet Gynecol Reprod Biol. 2017;209:3-7. doi:10.1016/j.ejogrb.2016.04.021
-
Symons LK, Miller JE, Kay VR, et al. The Immunopathophysiology of Endometriosis. Trends Mol Med. 2018;24(9):748-762.
-
Lessey BA, Kim JJ. Endometrial receptivity in the eutopic endometrium of women with endometriosis: it is affected, and let me show you why. Fertil Steril. 2017;108(1):19-27.
-
Muzii L, Di Tucci C, Achilli C, et al. Continuous versus cyclic oral contraceptives after laparoscopic excision of ovarian endometriomas: a systematic review and metaanalysis. Am J Obstet Gynecol. 2016;214(2):203-211.
-
Guerriero S, Condous G, van den Bosch T, et al. Systematic approach to sonographic evaluation of the pelvis in women with suspected endometriosis, including terms, definitions and measurements: a consensus opinion from the International Deep Endometriosis Analysis (IDEA) group. Ultrasound Obstet Gynecol. 2016;48(3):318-332.
-
Bazot M, Thomassin I, Hourani R, Cortez A, Darai E. Diagnostic accuracy of transvaginal sonography for deep pelvic endometriosis. Ultrasound Obstet Gynecol. 2004;24(2):180-185.
-
American Society for Reproductive Medicine. Revised American Society for Reproductive Medicine classification of endometriosis: 1996. Fertil Steril. 1997;67(5):817-821.
-
Vercellini P, Vigano P, Bandini V, Buggio L, Berlanda N, Somigliana E. Association of endometriosis and adenomyosis with pregnancy and infertility. Fertil Steril. 2023;119(5):727-740. doi:10.1016/j.fertnstert.2023.03.018
-
Pearce CL, Templeman C, Rossing MA, et al. Association between endometriosis and risk of histological subtypes of ovarian cancer: a pooled analysis of case-control studies. Lancet Oncol. 2012;13(4):385-394. doi:10.1016/S1470-2045(11)70404-1
-
Adamson GD, Pasta DJ. Endometriosis fertility index: the new, validated endometriosis staging system. Fertil Steril. 2010;94(5):1609-1615. doi:10.1016/j.fertnstert.2009.09.035
-
Mol BW, Bayram N, Lijmer JG, et al. The performance of CA-125 measurement in the detection of endometriosis: a meta-analysis. Fertil Steril. 1998;70(6):1101-1108. doi:10.1016/s0015-0282(98)00355-0
-
May KE, Conduit-Hulbert SA, Villar J, Kirtley S, Kennedy SH, Becker CM. Peripheral biomarkers of endometriosis: a systematic review. Hum Reprod Update. 2010;16(6):651-674. doi:10.1093/humupd/dmq009
-
Nisenblat V, Bossuyt PM, Farquhar C, Johnson N, Hull ML. Imaging modalities for the non-invasive diagnosis of endometriosis. Cochrane Database Syst Rev. 2016;2(2):CD009591. doi:10.1002/14651858.CD009591.pub2
-
Bazot M, Daraï E. Role of transvaginal sonography and magnetic resonance imaging in the diagnosis of uterine adenomyosis. Fertil Steril. 2018;109(3):389-397. doi:10.1016/j.fertnstert.2018.01.024
-
Winkel CA, Scialli AR. Medical and surgical therapies for pain associated with endometriosis. J Womens Health Gend Based Med. 2001;10(2):137-162. doi:10.1089/152460901300039485
-
Brown J, Pan A, Hart RJ. Gonadotrophin-releasing hormone analogues for pain associated with endometriosis. Cochrane Database Syst Rev. 2010;(12):CD008475. doi:10.1002/14651858.CD008475.pub2
-
Strowitzki T, Faustmann T, Gerlinger C, Seitz C. Dienogest in the treatment of endometriosis-associated pelvic pain: a 12-week, randomized, double-blind, placebo-controlled study. Eur J Obstet Gynecol Reprod Biol. 2010;151(2):193-198. doi:10.1016/j.ejogrb.2010.04.002
-
Köhler G, Faustmann TA, Gerlinger C, Seitz C, Mueck AO. A dose-ranging study to determine the efficacy and safety of 1, 2, and 4 mg of dienogest daily for endometriosis. Int J Gynaecol Obstet. 2010;108(1):21-25. doi:10.1016/j.ijgo.2009.08.020
-
Jacobson TZ, Duffy JM, Barlow D, Farquhar C, Koninckx PR, Olive D. Laparoscopic surgery for subfertility associated with endometriosis. Cochrane Database Syst Rev. 2010;(1):CD001398. doi:10.1002/14651858.CD001398.pub2
-
Barnhart K, Dunsmoor-Su R, Coutifaris C. Effect of endometriosis on in vitro fertilization. Fertil Steril. 2002;77(6):1148-1155. doi:10.1016/s0015-0282(02)03112-6
-
Benschop L, Farquhar C, van der Poel N, Heineman MJ. Interventions for women with endometrioma prior to assisted reproductive technology. Cochrane Database Syst Rev. 2010;(11):CD008571. doi:10.1002/14651858.CD008571.pub2
-
Practice Committee of the American Society for Reproductive Medicine. Fertility preservation in patients undergoing gonadotoxic therapy or gonadectomy: a committee opinion. Fertil Steril. 2019;112(6):1022-1033. doi:10.1016/j.fertnstert.2019.09.013
-
Healey M, Ang WC, Cheng C. Surgical treatment of endometriosis: a prospective randomized double-blinded trial comparing excision and ablation. Fertil Steril. 2010;94(7):2536-2540. doi:10.1016/j.fertnstert.2010.03.033
-
Keckstein J, Becker CM, Canis M, et al. Recommendations for the surgical treatment of endometriosis. Part 2: deep endometriosis. Hum Reprod Open. 2020;2020(1):hoaa002. doi:10.1093/hropen/hoaa002
-
Seracchioli R, Mabrouk M, Frascà C, et al. Long-term cyclic and continuous oral contraceptive therapy and endometrioma recurrence: a randomized controlled trial. Fertil Steril. 2010;93(1):52-56. doi:10.1016/j.fertnstert.2008.09.052
-
Vercellini P, Somigliana E, Viganò P, Abbiati A, Barbara G, Crosignani PG. Post-operative endometriosis recurrence: a plea for prevention based on pathogenetic, epidemiological and clinical evidence. Reprod Biomed Online. 2010;21(2):259-265. doi:10.1016/j.rbmo.2010.04.018
-
Taylor HS, Giudice LC, Lessey BA, et al. Treatment of Endometriosis-Associated Pain with Elagolix, an Oral GnRH Antagonist. N Engl J Med. 2017;377(1):28-40. doi:10.1056/NEJMoa1700089
-
Nisenblat V, Prentice L, Bossuyt PM, Farquhar C, Hull ML, Johnson N. Combination of the non-invasive tests for the diagnosis of endometriosis. Cochrane Database Syst Rev. 2016;7(7):CD012281. doi:10.1002/14651858.CD012281
-
Zondervan KT, Becker CM, Koga K, Missmer SA, Taylor RN, Viganò P. Endometriosis. Nat Rev Dis Primers. 2018;4(1):9. doi:10.1038/s41572-018-0008-5
-
Soliman AM, Yang H, Du EX, Kelley C, Winkel C. The direct and indirect costs associated with endometriosis: a systematic literature review. Hum Reprod. 2016;31(4):712-722. doi:10.1093/humrep/dev335
-
Fritzer N, Tammaa A, Salzer H, et al. Effects of endometriosis on sexual function: a cross-sectional study. Geburtshilfe Frauenheilkd. 2014;74(10):915-920. doi:10.1055/s-0034-1383047
-
Culley L, Law C, Hudson N, et al. The social and psychological impact of endometriosis on women's lives: a critical narrative review. Hum Reprod Update. 2013;19(6):625-639. doi:10.1093/humupd/dmt027
-
Bedaiwy MA, Patterson B, Mahajan S. Prevalence of myofascial chronic pelvic pain and the effectiveness of pelvic floor physical therapy. J Reprod Med. 2013;58(11-12):504-510.
-
Kold M, Hansen T, Vedsted-Hansen H, Forman A. Mindfulness-based psychological intervention for coping with pain in endometriosis. Nord Psychol. 2012;64(1):2-16. doi:10.1080/19012276.2012.693727
-
Xu Y, Zhao W, Li T, et al. Effects of acupuncture for the treatment of endometriosis-related pain: A systematic review and meta-analysis. PLoS One. 2017;12(10):e0186616. doi:10.1371/journal.pone.0186616
-
Lauretti GR, Oliveira R, Parada F, Mattos AL. The new portable transcutaneous electrical nerve stimulation device was efficacious in the control of primary dysmenorrhea cramp pain. Neuromodulation. 2015;18(6):522-526. doi:10.1111/ner.12269
-
Moore JS, Gibson PR, Perry RE, Burgell RE. Endometriosis in patients with irritable bowel syndrome: Specific symptomatic and demographic profile, and response to the low FODMAP diet. Aust N Z J Obstet Gynaecol. 2017;57(2):201-205. doi:10.1111/ajo.12594
-
Kold M, Hansen T, Vedsted-Hansen H, Forman A. Mindfulness-based psychological intervention for coping with pain in endometriosis. Nord Psychol. 2012;64(1):2-16. doi:10.1080/19012276.2012.693727
-
Armour M, Sinclair J, Chalmers KJ, Smith CA. Self-management strategies amongst Australian women with endometriosis: a national online survey. BMC Complement Altern Med. 2019;19(1):17. doi:10.1186/s12906-019-2431-x
-
Vodolazkaia A, El-Aalamat Y, Popovic D, et al. Evaluation of a panel of 28 biomarkers for the non-invasive diagnosis of endometriosis. Hum Reprod. 2012;27(9):2698-2711. doi:10.1093/humrep/des234
-
Moustafa S, Burn M, Mamillapalli R, Nematian S, Flores V, Taylor HS. Accurate diagnosis of endometriosis using serum microRNAs. Am J Obstet Gynecol. 2020;223(4):557.e1-557.e11. doi:10.1016/j.ajog.2020.02.050
Last Updated: 2026-01-16 Target Examination: MRCOG, FRANZCOG, FRACP, MRCP Difficulty Level: Intermediate Citation Count: 52