Epilepsy in Adults: Clinical Diagnosis and Management
1.1 Summary Epilepsy is defined by the International League Against Epilepsy (ILAE) as a disease of the brain characterized by an enduring predisposition to generate epileptic seizures and by the neurobiological,...
What matters first
1.1 Summary Epilepsy is defined by the International League Against Epilepsy (ILAE) as a disease of the brain characterized by an enduring predisposition to generate epileptic seizures and by the neurobiological,...
Status Epilepticus (less than 5 mins continuous or no recovery between events)
10 Jan 2026
Generated educational material; verify before clinical use.
Visible references section
See the concept before reading it
Study the key anatomy, imaging, and decision pathways as full teaching plates.
Clinical board
A visual summary of the highest-yield teaching signals on this page.
Urgent signals
Safety-critical features pulled from the topic metadata.
- Status Epilepticus (less than 5 mins continuous or no recovery between events)
- New-onset focal neurological deficit post-ictally
- Seizure in the context of pregnancy (Eclampsia)
- First-ever seizure with fever or meningism
Content status and exam context
This page is AI-generated educational content. It may contain errors or omissions and is not a substitute for current guidelines, local protocols, senior clinical judgement, or professional medical advice.
MedVellum does not claim an individual clinician reviewer, board certification, or professional credential for this page unless a future version names a real, verifiable reviewer.
Clinical explanation and evidence
SECTION 1: Clinical Overview
1.1 Summary
Epilepsy is defined by the International League Against Epilepsy (ILAE) as a disease of the brain characterized by an enduring predisposition to generate epileptic seizures and by the neurobiological, cognitive, psychological, and social consequences of this condition. The 2014 ILAE practical definition established diagnostic criteria: at least two unprovoked seizures occurring more than 24 hours apart, OR one unprovoked seizure with a probability of further seizures similar to the general recurrence risk (at least 60%) after two unprovoked seizures, OR diagnosis of an epilepsy syndrome (PMID: 24730690; DOI: 10.1111/epi.12550).
In adults, the incidence is approximately 50 per 100,000 people per year in high-income countries, with a bimodal distribution peaking in early childhood and again after age 65. The 2017 ILAE classification framework revolutionized the approach to epilepsy, introducing a multi-level classification: seizure type, epilepsy type, and epilepsy syndrome. This framework emphasizes the distinction between focal-onset, generalized-onset, and unknown-onset seizures, with further classification by awareness and motor/non-motor features (PMID: 28276062; DOI: 10.1111/epi.13670).
Clinical significance is paramount, as uncontrolled epilepsy increases the risk of sudden unexpected death in epilepsy (SUDEP), physical injury, and significant psychosocial morbidity. SUDEP occurs at a rate of 1.16 per 1,000 patient-years in epilepsy populations, with rates up to 9.3 per 1,000 in drug-resistant cases (PMID: 28241820; DOI: 10.1212/WNL.0000000000003685). Management primarily involves long-term anti-seizure medication (ASM) tailored to the specific seizure type and patient comorbidities. While 47% of patients achieve remission with the first ASM and an additional 13% with the second, the remaining 30-40% develop drug-resistant epilepsy, requiring evaluation for surgical intervention or neuromodulation (PMID: 10668698; DOI: 10.1056/NEJM200002033420503).
1.2 Key Facts
- Definition: A disease characterized by recurrent unprovoked seizures due to abnormal synchronized neuronal activity in the brain.
- Incidence: 50 per 100,000 adults per year in high-income countries; up to 190/100,000 in LMICs (PMID: 21807210; DOI: 10.1016/S0140-6736(11)60890-5).
- Prevalence: 6.38 per 1,000 persons for active epilepsy globally (PMID: 28404242; DOI: 10.1016/j.seizure.2017.03.007).
- Lifetime Risk: 1 in 26 people will develop epilepsy at some point in their life (PMID: 24514430; DOI: 10.1212/WNL.0000000000000071).
- Mortality: Standardized mortality ratio (SMR) is 2.0-3.0 compared to the general population; 2-3x higher risk of premature death.
- Morbidity: Depression in 30%, anxiety in 25%, cognitive impairment common; reduced quality of life.
- Peak Age: Bimodal distribution; secondary peak is adults > 65 years due to cerebrovascular disease.
- Sex Distribution: Slightly higher in males (Male:Female ratio 1.1:1.0).
- Pathognomonic Feature: None; diagnosis is primarily clinical, supported by interictal epileptiform discharges on EEG.
- Gold Standard Investigation: Video-EEG monitoring for seizure characterization and localization in complex cases.
- First-line Treatment: Lamotrigine or Levetiracetam for focal epilepsy; Valproate (males) or Levetiracetam (females) for generalized.
- Key Complication: Status Epilepticus, defined as seizure activity > 5 minutes, requiring immediate intervention.
- SUDEP Risk: 1 in 1,000 per year overall; up to 1 in 100 per year in drug-resistant epilepsy.
1.3 Clinical Pearls
Diagnostic Pearl: "The History is the Gold Standard" The diagnosis of epilepsy remains primarily clinical. A detailed eyewitness account is more sensitive than a single interictal EEG, which can be normal in up to 50% of patients with proven epilepsy. Always ask about the "aura" (focal aware seizure) and the post-ictal state. Request smartphone video recordings of events whenever possible.
Classification Pearl: "ILAE 2017 - Three Levels of Diagnosis" The ILAE 2017 classification requires three levels of diagnosis: (1) Seizure type (focal, generalized, unknown), (2) Epilepsy type (focal, generalized, combined, unknown), and (3) Epilepsy syndrome where applicable. Always determine etiology: structural, genetic, infectious, metabolic, immune, or unknown.
Examination Pearl: "Check the Lateral Tongue" Lateral tongue biting has a high specificity (96%) for generalized tonic-clonic seizures compared to syncope. Tip-of-the-tongue biting is more common in non-epileptic events or falls. Also examine for posterior shoulder dislocation in post-ictal patients with restricted arm movement.
EEG Pearl: "Activation Procedures Increase Yield" A single routine EEG detects epileptiform abnormalities in only 29-55% of patients. Yield increases to 80-90% with sleep deprivation, hyperventilation (for absence seizures), and photic stimulation. Always request activation procedures for first-seizure EEGs.
Treatment Pearl: "Start Low and Go Slow" Titrating anti-seizure medications slowly reduces the risk of idiosyncratic reactions (like Stevens-Johnson Syndrome with Lamotrigine) and improves long-term drug retention by minimizing initial side effects. Lamotrigine requires 6-8 weeks titration.
Pitfall Warning: "Avoid Valproate in Women of Childbearing Age" Sodium Valproate is highly teratogenic (10% risk of major malformations, 30-40% risk of neurodevelopmental disorders including autism). Avoid as first-line in women of childbearing potential per MHRA guidance. If unavoidable, ensure Pregnancy Prevention Programme enrollment.
Drug Selection Pearl: "Match ASM to Seizure Type" Carbamazepine, oxcarbazepine, phenytoin, and gabapentin can WORSEN myoclonic and absence seizures in generalized epilepsies. Always classify the seizure type before initiating treatment. When uncertain, use broad-spectrum agents like Levetiracetam or Valproate.
Emergency Pearl: "The 5-Minute Rule for Status" Any seizure lasting longer than 5 minutes should be treated as Status Epilepticus per the 2015 Neurocritical Care Society guidelines. Do not wait for the traditional 30-minute definition to initiate benzodiazepines, as neuronal damage begins early and seizure termination becomes more difficult with duration.
SUDEP Pearl: "Discuss Early and Document" NICE guidelines mandate SUDEP discussion with all patients at or near the time of diagnosis. Key modifiable risk factors: GTCS frequency (strongest predictor), non-adherence, nocturnal seizures, sleeping alone. Risk reduction: optimal seizure control, nocturnal monitoring devices, seizure alert alarms.
Driving Pearl: "Know the Regulations" In the UK (DVLA), Group 1 (car/motorcycle) license requires 12 months seizure-free OR established 3-year pattern of sleep-only seizures. Group 2 (HGV/bus) requires 10 years seizure-free off all ASMs. Always document driving advice given in clinical notes.
Exam Pearl: "Todd's Paralysis Localizes" Focal neurological deficit (e.g., hemiparesis) following a seizure suggests a focal onset in the contralateral motor cortex, even if the seizure appeared generalized. This is a high-yield point for localizing the seizure focus and indicates need for MRI.
1.4 Why This Matters Clinically
- Patient outcomes: Failure to diagnose epilepsy can lead to preventable injuries, status epilepticus, and SUDEP (1 in 1,000 patient-years). Conversely, misdiagnosing syncope or psychogenic non-epileptic seizures (PNES) as epilepsy leads to unnecessary medication with significant side effects and driving restrictions.
- Treatment gap: Globally, approximately 75% of people with epilepsy in low-income countries do not receive treatment, constituting the epilepsy treatment gap. Even in high-income countries, 30% have drug-resistant epilepsy requiring specialist evaluation.
- Quality of life: Epilepsy significantly impacts employment (unemployment rates 2-3x higher), driving privileges, and social relationships. Effective management requires addressing these psychosocial dimensions.
- Healthcare burden: Epilepsy accounts for over 1% of the global burden of disease (13 million DALYs). Poorly controlled epilepsy leads to frequent emergency department visits, hospitalizations, and high costs associated with long-term disability.
- Medico-legal: Driving regulations are strict; clinicians have a legal duty to advise patients not to drive and, in some jurisdictions, to report the diagnosis to licensing authorities. Failure to document driving advice can lead to liability if an accident occurs.
- Training relevance: Epilepsy is a core component of all medical curricula and postgraduate exams (MRCP, USMLE, FRCP). It tests the clinician's ability to synthesize historical data, interpret neurophysiology, and manage chronic pharmacology.
SECTION 2: Epidemiology
2.1 Incidence & Prevalence
- Incidence: 45-68 per 100,000 per year in developed nations; up to 190 per 100,000 in low-income countries (PMID: 21807210; DOI: 10.1016/S0140-6736(11)60890-5).
- Prevalence: 6.38 per 1,000 persons for active epilepsy; 7.60 per 1,000 for lifetime prevalence (PMID: 28404242).
- Lifetime Risk: 1 in 26 people (3.9%) will develop epilepsy at some point (PMID: 24514430).
- First Seizure Incidence: 61-80 per 100,000 per year; 50% do not recur if EEG/MRI normal.
- Trend: Stable in younger adults, but increasing in the elderly due to improved survival after stroke and neurodegenerative disease.
- Geographic Variation: Higher in sub-Saharan Africa and Latin America due to endemic neurocysticercosis, malaria, and birth trauma.
- Temporal Patterns: No significant seasonal variation, though sleep deprivation (common in holidays/exams) is a major trigger.
- Healthcare Burden: 0.5% of all emergency department visits are for seizures; epilepsy costs the EU ~€15.5 billion annually.
- Treatment Gap: less than 25% receiving appropriate treatment in low-income countries vs > 80% in high-income countries.
2.2 Demographics Table
| Factor | Details | Clinical Significance |
|---|---|---|
| Age | Peaks less than 2 yrs and > 65 yrs | Elderly have higher risk of status epilepticus, drug interactions, and post-stroke epilepsy. |
| Sex | 1.1:1 (M:F) | Males more prone to post-traumatic epilepsy; Females have catamenial patterns and pregnancy considerations. |
| Ethnicity | Higher in Hispanic/Black populations | Likely due to socioeconomic factors and access to care rather than genetics; treatment gap wider. |
| Geography | 2-3x higher in developing nations | Parasitic infections (Cysticercosis) are a leading cause of focal epilepsy; traumatic brain injury common. |
| Socioeconomic | Inverse correlation with income | Lower SES associated with higher rates of head trauma, infection, and reduced access to specialist care. |
| Occupation | High risk in manual labor | Safety concerns with heavy machinery and heights for patients; certain professions prohibited. |
2.3 Risk Factors Tables
Non-Modifiable Risk Factors:
| Factor | Relative Risk (95% CI) | Mechanism |
|---|---|---|
| Family History (1st degree) | RR 2.5-5.0 | Genetic channelopathies (SCN1A, KCNQ2), structural predisposition, or shared environmental factors. |
| History of Febrile Seizures | RR 2.0-10.0 | Hippocampal sclerosis development (especially if prolonged > 15 min, complex, or recurrent). |
| Prior Stroke | RR 3.0-20.0 | Gliotic scarring creating an epileptogenic focus; highest risk with hemorrhagic or cortical strokes. |
| Alzheimer's Disease | RR 5.0-10.0 | Amyloid-beta induced neuronal hyperexcitability; increases with disease progression. |
| Penetrating Head Trauma | RR 15.0-25.0 | Direct cortical injury and hemosiderin deposition; risk proportional to injury severity. |
| CNS Infection | RR 5.0-16.0 | Post-encephalitic epilepsy in 10-20% of survivors; risk highest with HSV and bacterial meningitis. |
| Cortical Dysplasia | RR 20.0+ | Focal cortical dysplasia is a leading cause of drug-resistant focal epilepsy in young adults. |
Modifiable Risk Factors:
| Risk Factor | Relative Risk (95% CI) | Evidence Level | Intervention Impact |
|---|---|---|---|
| Heavy Alcohol Use | RR 2.5 (1.5-4.1) | Level 1a | Withdrawal seizures are preventable; chronic use lowers seizure threshold. |
| Sleep Deprivation | RR 1.8 (1.2-2.5) | Level 2a | Consistent sleep hygiene reduces seizure frequency; key provocation factor. |
| Illicit Drug Use | RR 2.0 (1.1-3.5) | Level 2b | Cocaine/Amphetamines lower seizure threshold acutely; opioid withdrawal. |
| Medication Non-adherence | RR 3.0-5.0 | Level 1b | Leading cause of breakthrough seizures and status epilepticus. |
| Perinatal Insult | RR 3.5 (2.0-6.0) | Level 2a | Improved obstetric care reduces incidence of hypoxic-ischemic injury. |
| Neuroinfection | RR 10.0 (5.0-20.0) | Level 1b | Vaccination (e.g., H. influenzae, measles) reduces risk significantly. |
| Hypertension | RR 1.5-2.0 | Level 2a | Stroke prevention reduces late-onset epilepsy in elderly. |
2.4 Protective Factors
- Breastfeeding: RR 0.7, potential neuroprotective effects of maternal IgA and long-chain fatty acids.
- High Education Level: RR 0.8, likely a proxy for lower trauma risk and better health literacy.
- Mediterranean Diet: RR 0.9, anti-inflammatory effects may stabilize neuronal membranes; ketogenic diet is therapeutic.
- Regular Physical Activity: May reduce seizure frequency through stress reduction and improved sleep.
- Optimal ASM Adherence: Reduces seizure frequency by > 50% and significantly reduces SUDEP risk.
SECTION 3: Pathophysiology
3.1 Step 1: Initiating Event/Trigger
The pathophysiology of epilepsy begins with a transition from normal neuronal activity to a state of hyperexcitability. In adults, this is often triggered by an acute or chronic insult such as ischemia, trauma, infection, or a genetic mutation affecting ion channels.
- Molecular Basis: There is a sudden shift in the balance between excitatory (Glutamate) and inhibitory (GABA) neurotransmission. This imbalance is the fundamental mechanism underlying all seizures.
- Receptor Activation: Glutamate binds to N-methyl-D-aspartate (NMDA) and α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid (AMPA) receptors, leading to an influx of Calcium (Ca2+) and Sodium (Na+). Excessive NMDA activation is excitotoxic.
- Channelopathies: Genetic mutations in voltage-gated sodium channels (SCN1A, SCN2A), potassium channels (KCNQ2/3), or calcium channels (CACNA1A) can directly cause neuronal hyperexcitability.
- Signaling Pathways: Activation of the mammalian target of rapamycin (mTOR) pathway often occurs, especially in structural malformations like tuberous sclerosis and focal cortical dysplasia, promoting aberrant protein synthesis and neuronal growth.
3.2 Step 2: Early Pathological Changes - The Paroxysmal Depolarization Shift (PDS)
Following the trigger, individual neurons undergo a PDS, a prolonged depolarization that results in a burst of action potentials rather than the normal single spike.
- Cellular Mechanism: The PDS is characterized by a large (20-40 mV) depolarization lasting 50-200 ms, followed by a hyperpolarizing afterpotential. It represents the cellular correlate of the interictal spike seen on EEG.
- Inflammatory Response: Microglial activation occurs within hours of a seizure, releasing IL-1β, IL-6, and TNF-α. These cytokines enhance NMDA receptor function and decrease GABAA receptor expression, perpetuating hyperexcitability (PMID: 22154545; DOI: 10.1038/nrn3135).
- Ionic Flux: A massive influx of Ca2+ triggers intracellular signaling cascades, including the activation of Calpain and various protein kinases, leading to early gene expression changes.
- Vascular Changes: Increased blood-brain barrier (BBB) permeability allows for the extravasation of albumin into the brain parenchyma, which activates astrocytic TGF-β signaling and promotes hyperexcitability.
3.3 Step 3: Established Disease Process - Epileptogenesis
Epileptogenesis is the process by which a normal brain circuit becomes chronically prone to generate seizures. This latent period can last months to years.
- Definition: Epileptogenesis encompasses the molecular, cellular, and network changes that convert a normal brain to one capable of generating spontaneous seizures (PMID: 25912798; DOI: 10.1038/nrneurol.2015.63).
- Neuronal Loss: In temporal lobe epilepsy, there is selective loss of inhibitory interneurons in the dentate gyrus (particularly hilar cells) and CA1/CA3 pyramidal neurons.
- Mossy Fiber Sprouting: Granule cell axons (mossy fibers) undergo reorganization and form recurrent excitatory synapses, creating aberrant circuits in the hippocampus.
- Gliosis: Reactive astrocytosis impairs glutamate uptake and potassium buffering, further promoting hyperexcitability.
- Channelopathies: Downregulation of Potassium (K+) channels (e.g., Kv1.1) and hyperpolarization-activated cyclic nucleotide-gated (HCN) channels reduces the neuron's ability to repolarize.
- Neurogenesis: Aberrant neurogenesis in the dentate gyrus produces ectopic granule cells with abnormal connectivity.
3.4 Step 4: Progression and Synchronization
Seizures propagate when the hyperexcitable focus overcomes the surrounding "inhibitory surround" and recruits distant brain regions.
- Recruitment: Through gap junctions, synaptic connections, and ephaptic transmission, the initial focus recruits neighboring neurons into synchronized activity.
- Network Theory: Modern understanding views epilepsy as a network disorder, not just a focal lesion. Abnormal connectivity within and between brain regions enables seizure propagation.
- Thalamocortical Loops: In generalized seizures, the thalamus plays a central role in synchronizing electrical activity across both hemispheres via the thalamocortical relay system. T-type calcium channels in thalamic neurons are critical for generating the 3 Hz spike-wave of absence seizures.
- Focal to Bilateral: Focal seizures can spread to become bilateral tonic-clonic (formerly "secondary generalization") through commissural pathways and subcortical structures.
- Secondary Complications: Chronic seizures lead to "kindling," where repeated electrical discharges lower the seizure threshold further and may lead to the development of secondary foci (mirror foci).
3.5 Step 5: Resolution or Chronicity
Seizure termination is an active process, not merely "neuronal exhaustion."
- Endogenous Inhibitory Mechanisms: Release of adenosine, endogenous opioids (dynorphin, enkephalins), galanin, and neuropeptide Y eventually hyperpolarize neurons.
- GABAergic Enhancement: Activation of extrasynaptic GABAA receptors and interneuron recruitment promotes seizure termination.
- Metabolic Factors: Depletion of ATP and accumulation of lactate and adenosine contribute to activity cessation.
- Fibrosis/Gliosis: Chronic epilepsy results in hippocampal sclerosis (mesial temporal sclerosis) or cortical gliosis, which are visible on MRI and represent permanent structural change.
- Outcomes: If the underlying cause (e.g., a tumor) is removed early, epileptogenesis may be halted. If the circuit is permanently remodeled, the patient develops chronic, potentially drug-resistant epilepsy.
3.6 ILAE 2017 Classification Framework
The ILAE 2017 classification provides a comprehensive multi-level framework for epilepsy diagnosis (PMID: 28276062; DOI: 10.1111/epi.13670):
Level 1: Seizure Type
| Seizure Type | Definition | Key Features |
|---|---|---|
| Focal Onset | Originates in networks limited to one hemisphere | May be discretely localized or more widely distributed |
| → Aware | Awareness preserved throughout | Formerly "simple partial" |
| → Impaired Awareness | Awareness impaired at any point | Formerly "complex partial" |
| → Motor Onset | Initial motor manifestation | Automatisms, atonic, clonic, epileptic spasms, hyperkinetic, myoclonic, tonic |
| → Non-Motor Onset | Initial non-motor manifestation | Autonomic, behavior arrest, cognitive, emotional, sensory |
| → Focal to Bilateral Tonic-Clonic | Starts focal, evolves to GTCS | Formerly "secondary generalization" |
| Generalized Onset | Originates simultaneously in bilateral networks | |
| → Motor | GTCS, tonic, clonic, myoclonic, atonic | |
| → Non-Motor (Absence) | Typical, atypical, myoclonic, eyelid myoclonia | |
| Unknown Onset | Insufficient information | May be reclassified with more data |
Level 2: Epilepsy Type
| Type | Definition | Common Etiologies |
|---|---|---|
| Focal Epilepsy | Only focal seizures | Structural lesions, MTS, FCD, tumors |
| Generalized Epilepsy | Only generalized seizures | Genetic/idiopathic (JME, JAE, CAE) |
| Combined Generalized and Focal | Both seizure types occur | Dravet syndrome, Lennox-Gastaut |
| Unknown Epilepsy | Cannot be determined | Insufficient data |
Level 3: Epilepsy Syndrome
| Syndrome | Age of Onset | Seizure Types | Prognosis |
|---|---|---|---|
| Juvenile Myoclonic Epilepsy (JME) | 12-18 years | Myoclonic jerks, GTCS, absence | Good seizure control but lifelong treatment |
| Juvenile Absence Epilepsy (JAE) | 10-17 years | Absence, GTCS | Good with treatment |
| Childhood Absence Epilepsy (CAE) | 4-10 years | Typical absence | 70% remit by adolescence |
| Epilepsy with GTCS alone | Any age | GTCS only | Generally responsive to ASMs |
| Temporal Lobe Epilepsy | Any, peak 20-30 | Focal impaired awareness | 60-70% drug-resistant; surgery effective |
Etiology Classification:
| Etiology | Examples | Investigation |
|---|---|---|
| Structural | MTS, FCD, tumor, stroke, TBI | MRI, PET |
| Genetic | SCN1A, KCNQ2, chromosomal | Genetic testing |
| Infectious | HSV encephalitis, neurocysticercosis | CSF, serology |
| Metabolic | Pyridoxine deficiency, mitochondrial | Metabolic workup |
| Immune | Anti-NMDAR, anti-LGI1 encephalitis | Antibody panels |
| Unknown | No identifiable cause | Diagnosis of exclusion |
3.7 Anatomical Considerations
- Temporal Lobe: Most common site for focal epilepsy in adults (60-70%); involves the hippocampus, amygdala, and parahippocampal gyrus. Mesial temporal sclerosis is the most common pathology.
- Frontal Lobe: Second most common (20-30%); often produces nocturnal seizures with complex motor features (hypermotor). Short duration, minimal post-ictal confusion.
- Parietal Lobe: Rare (5-10%); sensory symptoms, visual disturbances.
- Occipital Lobe: Rare (5%); visual phenomena (elementary or complex), may spread anteriorly.
- Blood Supply: The Middle Cerebral Artery (MCA) supplies most of the lateral cortex; MCA strokes are the leading cause of post-stroke epilepsy. The Posterior Cerebral Artery supplies mesial temporal structures.
- Vulnerability: The CA1 region of the hippocampus is exceptionally sensitive to hypoxia and excitotoxicity; the first to show sclerosis.
- Network Hubs: The thalamus, basal ganglia, and limbic structures serve as network hubs for seizure propagation.
3.8 Physiological Considerations
- Sleep-Wake Cycle: NREM sleep (especially N2) facilitates synchronization and seizure generation; REM sleep is generally anti-epileptic due to desynchronized EEG and muscle atonia.
- Circadian Patterns: Frontal lobe seizures cluster at night; temporal lobe seizures peak in late morning/early afternoon.
- Hormones: Estrogen is pro-convulsant (enhances NMDA receptor function and reduces GABA); Progesterone is anti-convulsant (enhances GABA via neurosteroid metabolites). This drives catamenial epilepsy patterns.
- Stress: Chronic stress elevates cortisol, which is pro-epileptic. Acute stress may be protective initially but detrimental long-term.
- Menstrual Cycle: Catamenial epilepsy has three patterns: perimenstrual (C1), periovulatory (C2), and luteal phase inadequacy (C3). Consider clobazam or progesterone supplementation.
SECTION 4: Clinical Presentation
4.1 Symptoms
Seizure Semiology by Type:
| Seizure Type | Frequency | Character | Duration | Key Associations |
|---|---|---|---|---|
| Focal Aware (Aura) | 60% of focal | Preserved awareness; sensory/autonomic/cognitive symptoms | 30-120 sec | Temporal: epigastric rising, fear, déjà vu; Occipital: visual phenomena |
| Focal Impaired Awareness | 40% of focal | Behavioral arrest, automatisms, unresponsive | 1-3 mins | Oral automatisms (lip smacking), hand fumbling; post-ictal confusion |
| Focal to Bilateral Tonic-Clonic | 30% of focal | Starts with aura, evolves to convulsion | 1-3 mins | Aura is localizing; often occurs with medication non-adherence |
| Generalized Tonic-Clonic | 25% | Sudden loss of consciousness, tonic stiffening, clonic jerking | 1-3 mins | Cyanosis, tongue biting, incontinence; prolonged post-ictal confusion |
| Absence | 5% adults | Brief staring spells, behavioral arrest | 5-30 sec | No post-ictal confusion; may cluster; hyperventilation provokes |
| Myoclonic | 10% | Sudden "electric shock" jerks | less than 1 sec | Often on awakening; bilateral, may cause falls; triggers: sleep deprivation |
| Atonic | Rare | Sudden loss of tone, drop attacks | 1-2 sec | Risk of head/facial injury; common in Lennox-Gastaut |
| Tonic | Rare | Sustained stiffening, no clonic phase | 10-60 sec | Often nocturnal; may cause falls |
Aura Symptoms by Localization:
| Aura Type | Localization | Description |
|---|---|---|
| Epigastric rising | Mesial temporal | Visceral sensation rising from abdomen to chest/throat |
| Déjà vu/Jamais vu | Temporal neocortex | Inappropriate familiarity or unfamiliarity |
| Fear/Panic | Amygdala | Intense fear without obvious trigger |
| Olfactory/Gustatory | Uncus/insula | Unpleasant smell or taste (often burning, metallic) |
| Visual | Occipital | Elementary (flashes, colors) or complex (formed images) |
| Somatosensory | Parietal | Tingling, numbness in contralateral limb |
| Auditory | Superior temporal | Elementary (buzzing) or complex (music, voices) |
| Vertiginous | Parieto-temporal | Spinning sensation |
Atypical Presentations:
- Elderly: Often present with "vague" confusion, "dizzy spells," or altered behavior rather than dramatic convulsions. Post-stroke epilepsy may be subtle. Higher risk of status epilepticus.
- Pregnancy: Seizures may increase in frequency due to decreased drug levels (increased clearance), sleep deprivation, and non-adherence (fear of teratogenicity). Risk of eclampsia must be considered.
- Nocturnal: Hypermotor behavior, unusual vocalizations, or bed-wetting in an adult should raise suspicion for frontal lobe epilepsy or NFLE.
- Psychiatric presentation: Ictal fear, crying (dacrystic), or laughing (gelastic) seizures may be misdiagnosed as psychiatric conditions.
- Subtle Status: Non-convulsive status epilepticus presents as prolonged confusion, fluctuating consciousness, or subtle motor signs; requires EEG for diagnosis.
4.2 Signs
General Examination:
- Vital Signs: Tachycardia (150-200 bpm) and hypertension during the ictus; post-ictal bradypnea and mild hypoxia common.
- Skin: Look for neurofibromas or café-au-lait spots (NF1), ash-leaf spots or shagreen patches (Tuberous Sclerosis), port-wine stain (Sturge-Weber).
- Oral: Lateral tongue lacerations highly specific for GTCS.
- Musculoskeletal: Posterior shoulder dislocation, vertebral compression fractures (especially with prolonged tonic activity).
System-Specific Signs:
| Sign | How to Elicit | Significance | Sensitivity/Specificity |
|---|---|---|---|
| Todd's Paralysis | Post-ictal motor exam | Localizes focus to contralateral motor cortex; resolves less than 24-48h | 15% / 99% |
| Automatisms | Observation during seizure | Oral: temporal lobe; Gestural: frontal lobe origin | 50% / 70% |
| Versive Head Turn | Observation | Forced head turn AWAY from the seizure focus (usually); late version is ipsilateral | 40% / 90% |
| Figure-of-4 Sign | Observation | Extended arm contralateral to focus; flexed arm ipsilateral | 30% / 95% |
| Post-ictal Nose Wipe | Observation | Usually done with hand ipsilateral to seizure focus | 50% / 80% |
| Ictal Speech Preservation | Observation | Suggests non-dominant hemisphere onset | 30% / 90% |
| Urinary Incontinence | History/Exam | Common in GTCS, less specific than tongue biting | 40% / 60% |
| Pupillary Dilation | Observation during ictus | Sympathetic surge; bilateral in GTCS | 90% / 30% |
4.3 Red Flags
[!CAUTION] RED FLAGS - Seek immediate help if:
- Seizure lasts > 5 minutes (Status Epilepticus)
- Breathing does not return to normal after the seizure
- A second seizure starts before the person regains consciousness
- Seizure occurs in water (risk of drowning)
- Seizure occurs in a pregnant woman (Eclampsia must be excluded)
- New focal weakness that does not resolve within 24 hours (consider stroke)
- High fever or stiff neck accompanying the seizure (meningitis/encephalitis)
- First seizure in a patient > 50 years (higher risk of structural lesion)
- Seizure in a patient with active malignancy (brain metastases)
- Seizure following recent head trauma (subdural/epidural hematoma)
- Ictal asystole or witnessed cardiac arrest (ictal arrhythmia)
- Oxygen saturation less than 90% or cyanosis persisting post-ictally
SECTION 5: Clinical Examination
5.1 Structured Approach
- A: Airway - Ensure patency; recovery position post-ictally. Do NOT insert anything into the mouth during a seizure.
- B: Breathing - Check for cyanosis, aspiration, or respiratory depression. Administer O2 if hypoxic. Suction if excessive secretions.
- C: Circulation - Check pulse (tachycardia is common; bradycardia/asystole may suggest ictal arrhythmia). Obtain IV access.
- D: Disability - GCS score, pupillary response, and limb power (Todd's paralysis). Check glucose. Note time of seizure onset.
- E: Exposure - Check for head trauma, needle marks (drug use), incontinence, tongue biting. Look for medical alert bracelet.
5.2 Neurological Examination Focus
- Mental Status: Assess orientation, memory (especially post-ictal); patients may have retrograde and anterograde amnesia.
- Cranial Nerves: Visual field defects (homonymous hemianopia suggests structural lesion), papilledema (raised ICP).
- Motor: Subtle hemiparesis (Todd's), hyperreflexia, Babinski sign suggest structural lesion.
- Sensory: Cortical sensory loss suggests parietal lesion.
- Coordination: Ataxia may suggest cerebellar or posterior fossa pathology.
- Skin: Neurocutaneous stigmata (TSC, NF, Sturge-Weber).
5.3 Special Tests Table
| Test Name | Technique | Positive Finding | Sensitivity | Specificity | Clinical Use |
|---|---|---|---|---|---|
| Hyperventilation | 3 mins of deep breathing during EEG | Provokes 3Hz spike-wave in absence epilepsy | 80% (Absence) | 90% | Routine EEG activation |
| Photic Stimulation | Flashing lights at 1-60 Hz during EEG | Photoparoxysmal response | 20% (generalized) | 95% | Identifies photosensitive epilepsy |
| Sleep Deprivation | EEG after 24h no sleep | Activates epileptiform discharges | 50% increase yield | 85% | If routine EEG normal |
| Tilt Table Test | Passive head-up tilt to 70° | Syncope (reproduces symptoms) | 70% | 90% | Differentiating syncope from seizures |
| Serum Prolactin | Draw blood 10-20 mins post-event | > 2x baseline or > 36 ng/mL | 60% | 96% | Distinguish GTCS/focal from PNES |
| 12-Lead ECG | Standard recording | Long QT, Brugada, preexcitation | 5% | 99% | Rule out cardiac syncope |
| Ammonia (if hepatic disease) | Blood sample | Elevated | Variable | Variable | Hepatic encephalopathy |
SECTION 6: Investigations
6.1 First Seizure Investigation Protocol
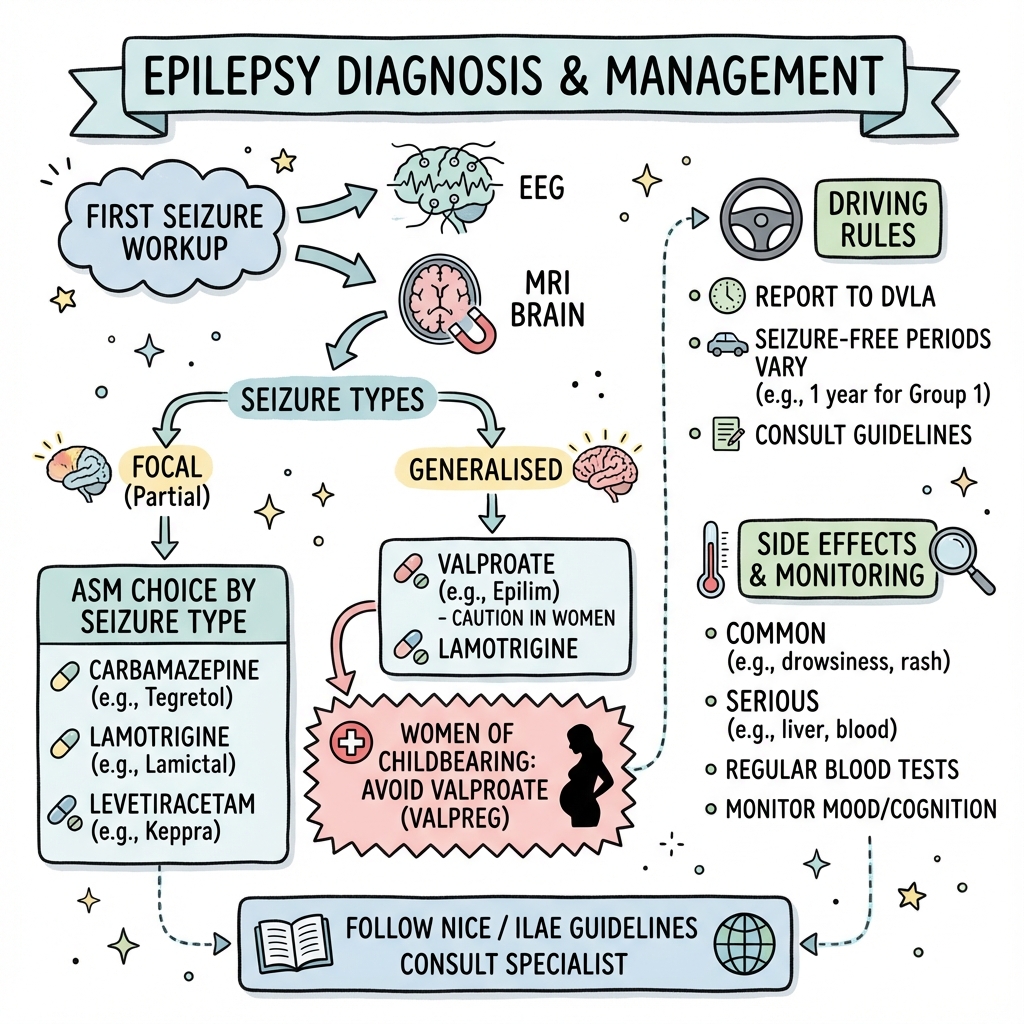
All patients presenting with a first unprovoked seizure require a structured workup according to the 2022 American Academy of Neurology (AAN) guidelines (PMID: 35025852; DOI: 10.1212/WNL.0000000000013273). The comprehensive evaluation aims to: (1) confirm the event was epileptic, (2) identify structural or metabolic causes, (3) stratify recurrence risk, and (4) guide treatment decisions.
Immediate (Emergency Department):
| Test | Expected Finding | Purpose | Priority |
|---|---|---|---|
| Capillary Glucose | 4.0-7.0 mmol/L | Rule out hypoglycemia as seizure cause | Immediate |
| 12-Lead ECG | Normal sinus rhythm | Rule out arrhythmia-induced syncope, Long QT, Brugada | Immediate |
| Pulse Oximetry | > 94% | Assess for hypoxia during/after seizure | Immediate |
| Temperature | 36.5-37.5°C | Rule out febrile seizure, encephalitis | Immediate |
| GCS/Neurological Exam | Return to baseline | Identify persistent deficit suggesting structural cause | Immediate |
Laboratory Tests:
| Test | Expected Finding | Purpose | Turnaround |
|---|---|---|---|
| FBC | Normal WCC | Check for infection, thrombocytopenia | 1 hour |
| U&Es | Na 135-145 mmol/L | Hyponatremia is common cause of metabolic seizures | 1 hour |
| Calcium/Magnesium | Normal levels | Rule out hypocalcemia/hypomagnesemia | 2 hours |
| LFTs | Baseline | Essential before starting Valproate/Phenytoin | 2 hours |
| Glucose | 4.0-7.0 mmol/L | Confirm bedside glucose | 1 hour |
| Toxicology Screen | Negative | Identify drug-induced seizures (cocaine, amphetamines) | 4 hours |
| CRP/Procalcitonin | Normal | Suggests inflammatory/infectious cause if elevated | 1 hour |
| Pregnancy Test | Beta-hCG | Crucial for ASM selection in women of childbearing age | 30 mins |
| Prolactin | Elevated 2x baseline | Supports diagnosis if measured 10-20 mins post-event | 2 hours |
First Seizure Workup - Core Triad:
The ILAE recommends three essential investigations for all first unprovoked seizures (PMID: 28276062; DOI: 10.1111/epi.13670):
-
EEG (within 24-48 hours preferred)
- Yield: 29-55% detect epileptiform activity on first routine EEG
- Sleep deprivation increases yield by 20-30%
- Interictal epileptiform discharges increase recurrence risk to 58-83%
- Normal EEG does NOT exclude epilepsy (50% of patients have normal initial EEG)
-
MRI Brain (Epilepsy Protocol within 4 weeks)
- Superior to CT for detecting epileptogenic lesions (sensitivity 95% vs 50%)
- Essential sequences: T1, T2 FLAIR, coronal hippocampal cuts, SWI
- Structural abnormalities found in 12-14% of new-onset adult epilepsy
- Mandatory in focal seizures, > 50 years, or focal deficits
-
Baseline Bloods (See table above)
- Identifies 5-10% of seizures due to metabolic causes
- Essential for safe ASM initiation
- Pregnancy test in all women of childbearing age (non-negotiable)
6.2 Neuroimaging
CT Head (Emergency Setting):
| Indication | Key Findings | Sensitivity | When to Use |
|---|---|---|---|
| First seizure with focal features | Hemorrhage, mass effect | 95% (blood) | Acute presentation, trauma |
| Persistent altered consciousness | Edema, herniation | Variable | Status epilepticus |
| Immunocompromised patient | Abscess, toxoplasmosis | 80% | HIV, transplant |
| Known malignancy | Metastases | 85% | Cancer patients |
| Head trauma | Subdural, epidural hematoma | 95% | Post-traumatic seizure |
MRI Brain (Epilepsy Protocol) - Gold Standard:
Epilepsy-specific MRI protocol includes (PMID: 30508771; DOI: 10.1111/epi.14326):
- 3T scanner preferred (1.5T acceptable)
- Volumetric T1-weighted 3D sequence (1 mm isotropic)
- Coronal T2 FLAIR perpendicular to hippocampi (2-3 mm slices)
- Coronal T2-weighted through hippocampi
- Axial T2/FLAIR whole brain
- Susceptibility-weighted imaging (SWI) for hemosiderin
- Gadolinium contrast if tumor/inflammation suspected
| Finding | Associated Epilepsy | Surgical Candidacy |
|---|---|---|
| Hippocampal Sclerosis (MTS) | Mesial TLE | Excellent (60-80% seizure-free) |
| Focal Cortical Dysplasia | Drug-resistant focal | Good if resectable |
| Cavernous Malformation | Focal epilepsy | Good if accessible |
| Low-grade tumor (DNET, ganglioglioma) | Focal epilepsy | Excellent |
| Encephalomalacia | Post-stroke epilepsy | Moderate |
| Tuberous Sclerosis | Multiple tubers | May require multi-stage |
6.3 Electroencephalography (EEG)
Standard EEG:
| Component | Yield | Key Findings | Limitations |
|---|---|---|---|
| Routine 20-30 min | 29-55% | Interictal spikes/waves | Low sensitivity; normal EEG does not exclude epilepsy |
| With Sleep Deprivation | +20-30% | Enhanced activation | Requires patient cooperation |
| With Hyperventilation | +20% (absence) | 3 Hz spike-wave | Specific for absence epilepsy |
| With Photic Stimulation | +10% | Photoparoxysmal response | Identifies photosensitivity |
| Serial EEGs (x3) | 70-80% | Cumulative yield | Cost and time |
EEG Findings by Epilepsy Type:
| Epilepsy Syndrome | Interictal Pattern | Ictal Pattern |
|---|---|---|
| Focal (temporal) | Temporal spikes/sharp waves | Rhythmic temporal theta/delta |
| Focal (frontal) | Frontal spikes (often subtle) | Fast rhythms, may be obscured by movement |
| JME | Generalized 4-6 Hz polyspike-wave | Generalized polyspike-wave |
| CAE/JAE | 3 Hz generalized spike-wave | Rhythmic 3 Hz spike-wave |
| Lennox-Gastaut | Slow spike-wave (less than 2.5 Hz) | Variable, often electrodecremental |
Advanced EEG Modalities:
| Modality | Indication | Duration | Yield |
|---|---|---|---|
| Ambulatory EEG | Frequent events, capture semiology | 24-72 hours | Higher than routine |
| Video-EEG Monitoring | Pre-surgical evaluation, PNES diagnosis | 5-10 days inpatient | Gold standard for characterization |
| Intracranial EEG | Pre-surgical localization when non-invasive discordant | Variable | Highest sensitivity |
6.4 Additional Investigations
| Investigation | Indication | Key Findings |
|---|---|---|
| PET-CT (Interictal) | Pre-surgical workup | Hypometabolism at seizure focus (70% sensitivity) |
| Ictal SPECT | Difficult localization | Hyperperfusion at focus; requires ictal injection |
| MEG | Complex focal epilepsy | Source localization of epileptiform activity |
| fMRI | Pre-surgical planning | Language and motor mapping |
| Wada Test | Pre-surgical planning | Language and memory lateralization |
| Neuropsychology | Pre/post-surgical evaluation | Baseline cognitive function, memory lateralization |
| Genetic Testing | Suspected genetic epilepsy, family history | SCN1A, KCNQ2, chromosomal microarray |
| CSF Analysis | Suspected encephalitis, autoimmune | Cell count, protein, glucose, antibodies |
| Autoimmune Panel | New-onset refractory status, limbic features | Anti-NMDAR, anti-LGI1, anti-CASPR2 |
6.5 Diagnostic Criteria
ILAE 2014 Operational Definition of Epilepsy (PMID: 24730690; DOI: 10.1111/epi.12550):
Epilepsy is a disease of the brain defined by ANY of the following:
- At least two unprovoked (or reflex) seizures occurring > 24 hours apart.
- One unprovoked (or reflex) seizure AND a probability of further seizures similar to the general recurrence risk (at least 60%) after two unprovoked seizures, occurring over the next 10 years.
- Diagnosis of an epilepsy syndrome.
High Recurrence Risk (≥60%) After First Seizure:
- Remote symptomatic etiology (stroke, TBI, CNS infection)
- Epileptiform abnormalities on EEG
- Structural abnormality on MRI (hippocampal sclerosis, FCD, tumor)
- Nocturnal seizure
- Focal seizure with secondary generalization
Epilepsy is considered RESOLVED if:
- Age-dependent syndrome and past applicable age, OR
- Seizure-free for 10 years with no ASMs for last 5 years
SECTION 7: Management
7.1 STATUS EPILEPTICUS MANAGEMENT PROTOCOL
┌─────────────────────────────────────────────────────────────────────────────┐
│ STATUS EPILEPTICUS MANAGEMENT ALGORITHM │
│ (Neurocritical Care Society 2012 / AES 2016 Guidelines) │
└─────────────────────────────────────────────────────────────────────────────┘
SEIZURE ONSET (Time = 0 minutes)
│
▼
┌─────────────────────────────────────────────────────────────────────────────┐
│ STAGE 1: STABILIZATION PHASE (0-5 minutes) │
│ ───────────────────────────────────────────── │
│ • Position patient safely (recovery position if possible) │
│ • Protect airway - DO NOT insert anything into mouth │
│ • Administer high-flow O2 (15L/min non-rebreathe) │
│ • Obtain IV access (two large-bore if possible) │
│ • Check capillary glucose → Give 50mL 50% dextrose if less than 70 mg/dL │
│ • Draw bloods: Glucose, U&Es, Ca, Mg, LFTs, ASM levels, toxicology │
│ • Monitor: SpO2, HR, BP, temperature │
│ • Consider: Thiamine 250mg IV before glucose if alcohol suspected │
│ • NOTE TIME OF SEIZURE ONSET │
└─────────────────────────────────────────────────────────────────────────────┘
│
▼ (Seizure continues > 5 mins)
┌─────────────────────────────────────────────────────────────────────────────┐
│ STAGE 2: INITIAL THERAPY - BENZODIAZEPINES (5-20 minutes) │
│ ───────────────────────────────────────────────────────── │
│ │
│ FIRST-LINE OPTIONS (choose ONE based on access): │
│ │
│ ┌─────────────────────────────────────────────────────────────────────┐ │
│ │ IF IV ACCESS: │ │
│ │ • Lorazepam 0.1 mg/kg IV (max 4mg) over 2 mins │ │
│ │ • May repeat once after 5-10 mins if seizure persists │ │
│ │ OR │ │
│ │ • Diazepam 0.15 mg/kg IV (max 10mg), may repeat x1 │ │
│ └─────────────────────────────────────────────────────────────────────┘ │
│ ┌─────────────────────────────────────────────────────────────────────┐ │
│ │ IF NO IV ACCESS: │ │
│ │ • Midazolam 10mg IM (if > 40kg) or 0.2 mg/kg (if less than 40kg) │ │
│ │ OR │ │
│ │ • Midazolam 10mg BUCCAL (preferred in community/pediatrics) │ │
│ │ OR │ │
│ │ • Diazepam 10-20mg PR │ │
│ └─────────────────────────────────────────────────────────────────────┘ │
│ │
│ ⚠️ Prepare for airway management - respiratory depression common │
│ ⚠️ Do not exceed 2 doses of benzodiazepines │
│ ⚠️ Call for senior/ICU support early │
└─────────────────────────────────────────────────────────────────────────────┘
│
▼ (Seizure continues after 2nd BZD dose)
┌─────────────────────────────────────────────────────────────────────────────┐
│ STAGE 3: ESTABLISHED STATUS - SECOND-LINE ASM (20-40 minutes) │
│ ───────────────────────────────────────────────────────────── │
│ │
│ Choose ONE of the following (Level I evidence - ESETT Trial): │
│ │
│ ┌─────────────────────────────────────────────────────────────────────┐ │
│ │ OPTION A: LEVETIRACETAM (Preferred if no IV phenytoin experience) │ │
│ │ • Dose: 60 mg/kg IV (max 4500mg) │ │
│ │ • Infusion rate: Over 15 minutes │ │
│ │ • Advantages: No cardiac monitoring, minimal drug interactions │ │
│ │ • Caution: Psychiatric side effects │ │
│ └─────────────────────────────────────────────────────────────────────┘ │
│ ┌─────────────────────────────────────────────────────────────────────┐ │
│ │ OPTION B: FOSPHENYTOIN/PHENYTOIN │ │
│ │ • Fosphenytoin: 20 mg PE/kg IV at 150 mg PE/min │ │
│ │ • Phenytoin: 20 mg/kg IV at 50 mg/min (max) │ │
│ │ • REQUIRES: Continuous cardiac monitoring (arrhythmia risk) │ │
│ │ • REQUIRES: Separate IV line (incompatible with dextrose) │ │
│ │ • Contraindicated: Heart block, bradycardia │ │
│ └─────────────────────────────────────────────────────────────────────┘ │
│ ┌─────────────────────────────────────────────────────────────────────┐ │
│ │ OPTION C: SODIUM VALPROATE │ │
│ │ • Dose: 40 mg/kg IV (max 3000mg) │ │
│ │ • Infusion rate: Over 10 minutes │ │
│ │ • Contraindicated: Pregnancy, liver disease, mitochondrial │ │
│ │ • Advantages: Broad-spectrum, fast loading │ │
│ └─────────────────────────────────────────────────────────────────────┘ │
│ │
│ NOTE: ESETT Trial (PMID: 31774955) showed equal efficacy for all three │
└─────────────────────────────────────────────────────────────────────────────┘
│
▼ (Seizure continues after second-line ASM)
┌─────────────────────────────────────────────────────────────────────────────┐
│ STAGE 4: REFRACTORY STATUS EPILEPTICUS (> 40-60 minutes) │
│ ──────────────────────────────────────────────────────── │
│ │
│ 🚨 REQUIRES ICU ADMISSION AND INTUBATION │
│ │
│ OPTIONS (choose based on local expertise/availability): │
│ │
│ ┌─────────────────────────────────────────────────────────────────────┐ │
│ │ MIDAZOLAM INFUSION │ │
│ │ • Loading: 0.2 mg/kg bolus │ │
│ │ • Infusion: 0.1-2 mg/kg/hour │ │
│ │ • Titrate to EEG burst suppression │ │
│ └─────────────────────────────────────────────────────────────────────┘ │
│ ┌─────────────────────────────────────────────────────────────────────┐ │
│ │ PROPOFOL INFUSION │ │
│ │ • Loading: 2 mg/kg bolus │ │
│ │ • Infusion: 2-10 mg/kg/hour │ │
│ │ • ⚠️ Risk: Propofol infusion syndrome (monitor CK, lactate) │ │
│ └─────────────────────────────────────────────────────────────────────┘ │
│ ┌─────────────────────────────────────────────────────────────────────┐ │
│ │ THIOPENTAL/PENTOBARBITAL │ │
│ │ • Loading: 5-7 mg/kg │ │
│ │ • Infusion: 1-5 mg/kg/hour │ │
│ │ • Requires vasopressor support in most cases │ │
│ └─────────────────────────────────────────────────────────────────────┘ │
│ │
│ CONTINUOUS EEG MONITORING MANDATORY │
│ Target: Burst suppression for 24-48 hours, then wean slowly │
└─────────────────────────────────────────────────────────────────────────────┘
│
▼ (Seizure persists > 24 hours despite anesthesia)
┌─────────────────────────────────────────────────────────────────────────────┐
│ STAGE 5: SUPER-REFRACTORY STATUS EPILEPTICUS │
│ ───────────────────────────────────────────────── │
│ │
│ Consider additional interventions: │
│ • Ketamine infusion (0.5-5 mg/kg/hour) - NMDA antagonist │
│ • Magnesium sulfate (especially if eclampsia suspected) │
│ • Hypothermia (32-35°C) │
│ • Ketogenic diet (enteral) │
│ • Immunotherapy if autoimmune etiology suspected │
│ - IV methylprednisolone 1g daily x 5 days │
│ - IVIG 0.4 g/kg/day x 5 days │
│ - Plasmapheresis │
│ • Emergency epilepsy surgery if resectable focus identified │
│ • ECT (electroconvulsive therapy) - case reports of success │
│ │
│ INVESTIGATE FOR UNDERLYING CAUSE: │
│ • Autoimmune encephalitis panel (anti-NMDAR, anti-LGI1, etc.) │
│ • Repeat neuroimaging (MRI with contrast) │
│ • CSF analysis │
│ • Consider paraneoplastic workup │
└─────────────────────────────────────────────────────────────────────────────┘
7.2 ADULT EPILEPSY MANAGEMENT ALGORITHM
┌─────────────────────────────────────────────────────────────────────────────┐
│ ADULT EPILEPSY MANAGEMENT ALGORITHM │
│ (NICE NG217 / ILAE / SANAD Guidelines) │
└─────────────────────────────────────────────────────────────────────────────┘
│
▼
┌─────────────────────────────────────────────────────────────────────────────┐
│ STEP 1: DIAGNOSIS AND CLASSIFICATION │
│ ───────────────────────────────────── │
│ • Confirm epileptic seizure (vs syncope, PNES, other) │
│ • Detailed history + eyewitness account (video if available) │
│ • Classify: Focal vs Generalized vs Unknown (ILAE 2017) │
│ • Determine etiology: Structural/Genetic/Infectious/Metabolic/Immune │
│ • Identify epilepsy syndrome where possible │
│ • Investigations: EEG + MRI Brain (epilepsy protocol) │
└─────────────────────────────────────────────────────────────────────────────┘
│
▼
┌─────────────────────────────────────────────────────────────────────────────┐
│ STEP 2: DECISION TO TREAT │
│ ───────────────────────────── │
│ │
│ TREAT after first seizure if HIGH RECURRENCE RISK (≥60%): │
│ ✓ Abnormal EEG (epileptiform discharges) │
│ ✓ Structural abnormality on MRI │
│ ✓ Remote symptomatic cause (prior stroke, TBI) │
│ ✓ Nocturnal seizure │
│ ✓ Epilepsy syndrome diagnosed │
│ │
│ MAY DEFER treatment if LOW recurrence risk: │
│ ○ Normal EEG and MRI │
│ ○ Provoked seizure (sleep deprivation, alcohol withdrawal) │
│ ○ Discuss risks/benefits with patient │
│ │
│ ALWAYS treat after second unprovoked seizure │
└─────────────────────────────────────────────────────────────────────────────┘
│
┌─────────────────────┼─────────────────────┐
▼ ▼ ▼
┌─────────────────────┐ ┌─────────────────────┐ ┌─────────────────────┐
│ FOCAL EPILEPSY │ │ GENERALIZED EPILEPSY│ │ UNKNOWN/COMBINED │
│ │ │ │ │ │
│ (or focal to │ │ (Absence, JME, │ │ (Uncertain type, │
│ bilateral GTCS) │ │ JAE, GTCS only) │ │ both types occur) │
└─────────────────────┘ └─────────────────────┘ └─────────────────────┘
│ │ │
▼ ▼ ▼
┌─────────────────────┐ ┌─────────────────────┐ ┌─────────────────────┐
│ FIRST-LINE AGENTS: │ │ FIRST-LINE AGENTS: │ │ BROAD-SPECTRUM: │
│ │ │ │ │ │
│ • Lamotrigine ✓ │ │ MALES: │ │ • Levetiracetam ✓ │
│ (SANAD II winner) │ │ • Valproate ✓✓ │ │ (safest choice) │
│ • Levetiracetam │ │ (most effective) │ │ • Valproate │
│ (quick titration) │ │ • Levetiracetam │ │ (avoid in women) │
│ │ │ │ │ • Lamotrigine │
│ Consider age, sex, │ │ FEMALES: │ │ │
│ comorbidities │ │ • Levetiracetam ✓✓ │ │ AVOID narrow- │
│ │ │ (teratogenicity) │ │ spectrum agents │
│ │ │ • Lamotrigine │ │ (may worsen GTCS) │
│ │ │ ⚠️ AVOID VALPROATE │ │ │
└─────────────────────┘ └─────────────────────┘ └─────────────────────┘
│ │ │
▼ ▼ ▼
┌─────────────────────────────────────────────────────────────────────────────┐
│ STEP 3: MONITOR RESPONSE │
│ ───────────────────────── │
│ • Aim: Complete seizure freedom with minimal side effects │
│ • Review at 2-4 weeks after starting, then 3-monthly initially │
│ • Seizure diary essential │
│ • Monitor for side effects (mood, rash, cognitive) │
│ • Drug levels: Only for phenytoin, phenobarbital, or suspected toxicity │
│ │
│ IF FIRST ASM FAILS (ineffective or intolerable): │
│ → Substitute alternative first-line agent OR │
│ → Add second agent (adjunctive therapy) │
└─────────────────────────────────────────────────────────────────────────────┘
│
▼ (Failure of first appropriate ASM)
┌─────────────────────────────────────────────────────────────────────────────┐
│ STEP 4: SECOND-LINE / ADJUNCTIVE THERAPY │
│ ───────────────────────────────────────── │
│ │
│ FOCAL EPILEPSY: GENERALIZED EPILEPSY: │
│ • Carbamazepine • Clobazam (adjunct) │
│ • Lacosamide ✓ • Topiramate │
│ • Zonisamide • Lamotrigine (if not tried) │
│ • Oxcarbazepine • Zonisamide │
│ • Topiramate • Perampanel │
│ • Perampanel │
│ • Brivaracetam ⚠️ AVOID in generalized: │
│ • Eslicarbazepine • Carbamazepine, Oxcarbazepine │
│ • Clobazam (adjunct) • Phenytoin, Gabapentin │
│ • Pregabalin, Tiagabine │
│ (May worsen myoclonic/absence) │
└─────────────────────────────────────────────────────────────────────────────┘
│
▼ (Failure of 2 appropriately chosen ASMs = DRUG-RESISTANT)
┌─────────────────────────────────────────────────────────────────────────────┐
│ STEP 5: DRUG-RESISTANT EPILEPSY PATHWAY │
│ ───────────────────────────────────────── │
│ │
│ DEFINITION: Failure of 2 tolerated, appropriately chosen and used ASM │
│ schedules (monotherapy or combination) to achieve seizure freedom │
│ │
│ ACTIONS: │
│ 1. REFER to tertiary epilepsy center │
│ 2. Confirm diagnosis (video-EEG to exclude PNES) │
│ 3. Optimize current treatment │
│ 4. Consider epilepsy surgery evaluation │
│ │
│ SURGICAL OPTIONS: NON-SURGICAL OPTIONS: │
│ • Resective surgery • Vagus Nerve Stimulation (VNS) │
│ (temporal lobectomy, • Responsive Neurostimulation (RNS) │
│ lesionectomy) • Deep Brain Stimulation (DBS) │
│ • Laser ablation (LITT) • Ketogenic/Modified Atkins diet │
│ • Disconnection surgery • Cannabidiol (Epidyolex) │
│ (corpus callosotomy) • Clinical trials │
└─────────────────────────────────────────────────────────────────────────────┘
7.3 Anti-Seizure Medication Selection by Seizure Type
Drug Selection Framework - ILAE 2017 Based:
The choice of ASM should be guided by the ILAE 2017 seizure classification, with critical attention to the distinction between focal, generalized, and combined epilepsy types (PMID: 28276062; DOI: 10.1111/epi.13670). Use of narrow-spectrum sodium channel blockers (carbamazepine, phenytoin, oxcarbazepine) in generalized epilepsy can paradoxically worsen myoclonic and absence seizures, a common examination pitfall (PMID: 22573629; DOI: 10.1212/WNL.0b013e3182563b19).
Evidence-Based First-Line Selection:
According to the SANAD II trial results (2021), the most comprehensive comparative effectiveness study of ASMs to date (PMID: 33836157, 33836158; DOI: 10.1016/S0140-6736(21)00154-5):
For FOCAL Epilepsy:
- Lamotrigine: Remains gold standard for time-to-treatment-failure and 12-month remission
- Levetiracetam: Non-inferior for seizure freedom, faster titration, but higher mood adverse events
- Cost-effectiveness: Lamotrigine superior due to better retention rates
For GENERALIZED Epilepsy:
- Males and non-childbearing females: Sodium Valproate most effective (12-month remission: 64% vs 52% for Levetiracetam)
- Females of childbearing potential: Levetiracetam or Lamotrigine (Valproate contraindicated per MHRA)
- JME specifically: Valproate (males), Levetiracetam, or Topiramate; AVOID carbamazepine
For UNKNOWN Type:
- Use BROAD-SPECTRUM agents only: Levetiracetam, Lamotrigine, or Valproate (with pregnancy safeguards)
- Avoid narrow-spectrum agents that may worsen generalized seizures
First-Line Agents - Detailed Prescribing:
| Drug | Seizure Type | Starting Dose | Target Dose | Titration | Key Side Effects | Monitoring |
|---|---|---|---|---|---|---|
| Lamotrigine | Focal, GTCS | 25mg OD | 100-200mg BD | Increase by 25-50mg every 2 weeks | Rash (SJS less than 1%), insomnia, headache | Rash vigilance weeks 1-8 |
| Levetiracetam | Focal, Generalized | 250-500mg BD | 500-1500mg BD | Increase by 500mg weekly | Irritability (10%), mood change, fatigue | Mood/behavior; renal function |
| Sodium Valproate | Generalized, Focal | 300mg BD | 500-1000mg BD | Increase by 200mg weekly | Weight gain, tremor, hair loss, teratogenicity | LFTs, FBC; AVOID IN PREGNANCY |
| Carbamazepine | Focal only | 100mg BD | 400-600mg BD | Increase by 100mg weekly | Hyponatremia, diplopia, ataxia, rash | Na+, FBC, LFTs; HLA-B*1502 if Asian |
| Oxcarbazepine | Focal only | 150mg BD | 600-1200mg BD | Increase by 150mg weekly | Hyponatremia (higher risk than CBZ), diplopia | Na+ levels |
Critical Drug-Seizure Type Matching:
| ASM Category | Seizure Types | Mechanism | Caution |
|---|---|---|---|
| Narrow-spectrum Sodium Channel Blockers | Focal ONLY | Inactivate voltage-gated Na+ channels | ⚠️ Can WORSEN myoclonic, absence, and GTCS in idiopathic generalized epilepsy |
| Examples: Carbamazepine, Oxcarbazepine, Phenytoin, Lacosamide, Eslicarbazepine | Common exam trap: Never use in JME | ||
| Broad-spectrum Multi-mechanism | Focal AND Generalized | Multiple targets (Na+, Ca2+, GABA, glutamate) | Safe choice when seizure type uncertain |
| Examples: Valproate, Levetiracetam, Lamotrigine, Topiramate, Zonisamide, Perampanel | Preferred when doubt exists |
AAN/AES 2018 Guideline Recommendations (PMID: 29898971; DOI: 10.1212/WNL.0000000000005755):
For newly diagnosed focal epilepsy, Level A evidence supports:
- Lamotrigine and Levetiracetam as equally effective for seizure freedom (both superior to older agents for tolerability)
- Carbamazepine and Zonisamide have similar efficacy but poorer tolerability profiles
- Extended-release formulations improve adherence and reduce side effects
For newly diagnosed generalized epilepsy:
- Valproate remains most efficacious (Level A)
- Lamotrigine and Levetiracetam are alternatives with better safety profiles (Level B)
- Ethosuximide is first-line for childhood absence epilepsy but rarely used in adults
Mechanism-Based Selection:
| Mechanism | ASMs | Clinical Use | Limitations |
|---|---|---|---|
| Voltage-gated Na+ channel blockade | Phenytoin, Carbamazepine, Lamotrigine, Lacosamide | Focal seizures, GTCS | Risk of worsening absence/myoclonic |
| SV2A binding | Levetiracetam, Brivaracetam | Broad-spectrum, all seizure types | Psychiatric side effects (10-15%) |
| GABA enhancement | Valproate, Benzodiazepines, Phenobarbital | Broad-spectrum, status epilepticus | Sedation, dependence |
| T-type Ca2+ channel blockade | Ethosuximide, Valproate | Absence seizures | Specific to absence |
| Glutamate antagonism | Perampanel, Topiramate | Adjunctive broad-spectrum | Cognitive effects |
| Multiple mechanisms | Valproate, Topiramate, Zonisamide | Treatment-resistant, broad-spectrum | Variable side effect profiles |
Second-Line/Adjunctive Agents:
| Drug | Primary Use | Dose Range | Key Considerations |
|---|---|---|---|
| Lacosamide | Focal epilepsy | 50-200mg BD | PR interval prolongation; avoid in heart block; well-tolerated |
| Zonisamide | Focal/Generalized | 100-300mg OD | Weight loss, renal stones, oligohidrosis; sulfonamide |
| Topiramate | Focal/Generalized | 50-200mg BD | Cognitive slowing, word-finding difficulty, weight loss, stones |
| Perampanel | Adjunct focal/GTCS | 4-12mg ON | Aggression, dizziness; CYP3A4 interactions; take at bedtime |
| Brivaracetam | Adjunct focal | 25-100mg BD | Similar to LEV but less psychiatric SE; no mood monitoring |
| Clobazam | Adjunct all types | 10-30mg ON | Sedation, tolerance; catamenial use; intermittent dosing |
| Eslicarbazepine | Focal epilepsy | 400-1200mg OD | Once daily; hyponatremia; similar to CBZ |
| Pregabalin | Adjunct focal | 150-300mg BD | Weight gain, edema; ⚠️ may worsen generalized |
| Gabapentin | Adjunct focal | 300-600mg TDS | Minimal interactions; ⚠️ may worsen generalized |
| Phenytoin | Focal, GTCS | 200-400mg OD | Non-linear kinetics; level monitoring; gingival hyperplasia |
| Phenobarbital | All types | 60-180mg OD | Sedation, cognitive; enzyme inducer; rarely first-line now |
Therapeutic Drug Monitoring (PMID: 18397299; DOI: 10.1111/j.1528-1167.2008.01561.x):
Routine monitoring NOT required for:
- Levetiracetam, Lamotrigine, Lacosamide, Zonisamide, Topiramate
- Check levels only if suspected toxicity or non-adherence
Routine monitoring ESSENTIAL for:
- Phenytoin (non-linear kinetics, narrow therapeutic index: 10-20 mg/L)
- Carbamazepine (enzyme auto-induction, target: 4-12 mg/L)
- Valproate (if dose > 1000mg/day or suspected toxicity, target: 50-100 mg/L)
- Phenobarbital (long half-life, target: 15-40 mg/L)
7.4 Special Populations
Women of Childbearing Age - MHRA Pregnancy Prevention Programme:
The UK Medicines and Healthcare products Regulatory Agency (MHRA) issued stringent guidance in 2018 regarding valproate use in women, following evidence of severe teratogenicity and neurodevelopmental harm (PMID: 24514430; DOI: 10.1212/WNL.0000000000000071).
Valproate Risks:
- Major congenital malformations: 10.3% (vs 2-3% background)
- Neural tube defects: 1-2% (10-20x general population)
- Neurodevelopmental disorders: 30-40% (including autism, reduced IQ)
- Dose-dependent: Risk increases significantly > 1000mg/day
MHRA Requirements for Valproate Prescription in Women:
- Contraindicated unless:
- No alternative treatments are suitable AND
- Pregnancy Prevention Programme conditions met
- Annual Risk Acknowledgement Form signed by patient and prescriber
- Effective contraception (ideally two methods)
- Pregnancy testing before initiation and regularly during treatment
- Specialist review at least annually
Preferred ASMs in Women of Childbearing Age (PMID: 27819746; DOI: 10.1002/14651858.CD010224.pub2):
| Drug | Malformation Rate | Neurodevelopment | Recommendation |
|---|---|---|---|
| Levetiracetam | 1.8% (2.8% at doses > 3000mg) | Data reassuring; no significant impact on IQ | FIRST-LINE preferred |
| Lamotrigine | 2.0% (3.2% at doses > 300mg) | No consistent neurodevelopmental effects | FIRST-LINE preferred |
| Carbamazepine | 2.6% | Possible mild effects | Consider if needed |
| Oxcarbazepine | 2.2% | Limited data | Use with caution |
| Topiramate | 4.2% | Cleft lip/palate; avoid | Avoid if possible |
| Phenytoin | 6.1% | Fetal hydantoin syndrome | Avoid |
| Phenobarbital | 6.5% | Cardiac defects | Avoid |
| Valproate | 10.3% | 30-40% neurodevelopmental | CONTRAINDICATED |
Pre-Conception Counseling - Essential Components (PMID: 19398681; DOI: 10.1212/WNL.0b013e3181a6b325):
-
Folic Acid Supplementation
- Dose: 5 mg/day (10x standard dose)
- Duration: Start 3 months before conception, continue through first trimester
- Rationale: ASMs impair folate metabolism; higher doses needed than general population (400 mcg)
- Evidence: Reduces neural tube defects risk by 50-70%
-
ASM Optimization
- Aim for monotherapy at lowest effective dose
- Switch from valproate at least 6 months before planned pregnancy
- Achieve seizure control before conception (ideally seizure-free 9-12 months)
- Do NOT stop ASMs abruptly due to seizure risk
-
Seizure Risk During Pregnancy
- 15-30% experience increased seizure frequency (due to increased clearance, non-adherence, sleep deprivation)
- GTCS carries fetal risks: hypoxia, trauma, miscarriage (2-3x risk), placental abruption
- Benefit of seizure control outweighs teratogenic risk for most ASMs (except valproate)
-
Specialist Referral
- Refer to joint epilepsy-obstetrics clinic before conception
- Involve fetal medicine for high-risk cases
- Enroll in UK Epilepsy and Pregnancy Register
Management During Pregnancy (PMID: 19398685; DOI: 10.1212/WNL.0b013e3181a6b325):
First Trimester:
- Continue current ASM (switching increases seizure risk)
- Detailed anomaly ultrasound at 12 weeks (nuchal translucency)
- Maternal serum alpha-fetoprotein at 16-18 weeks if on carbamazepine or valproate
- Fetal anatomical survey at 18-20 weeks (focus on neural tube, heart, face)
Second and Third Trimester - Therapeutic Drug Monitoring:
| ASM | Clearance Change | Clinical Action |
|---|---|---|
| Lamotrigine | ↑ 50-100% (estrogen-induced UGT enhancement) | Monitor levels monthly; dose may need to DOUBLE by third trimester |
| Levetiracetam | ↑ 50-60% (increased renal clearance) | Monitor levels monthly; may need 30-50% dose increase |
| Carbamazepine | ↑ 25-50% | May need dose adjustment |
| Phenytoin | ↑ 50-100% (also increased free fraction) | Monitor free phenytoin levels |
| Valproate | Variable (increased volume, decreased albumin) | Avoid; if unavoidable, use lowest dose |
Key Monitoring Points:
- Monthly ASM levels (especially lamotrigine and levetiracetam)
- Increase dose to maintain pre-pregnancy levels/seizure control
- Seizure diary essential
- Adjust based on clinical response, not just levels
Delivery:
- Vaginal delivery preferred unless obstetric contraindications
- Ensure ASMs not missed during labor
- IV access for emergency benzodiazepines
- Avoid sleep deprivation (epidural allows rest)
- Vitamin K 1 mg IM to neonate immediately after birth (especially if enzyme-inducing ASMs like carbamazepine, phenytoin)
Postnatal Management:
ASM Dose Adjustment:
- Lamotrigine: Reduce to pre-pregnancy dose over 2-3 weeks postpartum (risk of toxicity as clearance normalizes)
- Levetiracetam: Reduce over 1-2 weeks
- Monitor carefully - high-risk period for seizures (sleep deprivation, stress, hormonal changes)
Breastfeeding (PMID: 19398685; DOI: 10.1212/WNL.0b013e3181a6b325):
| ASM | Breast Milk Transfer | Infant Monitoring | Recommendation |
|---|---|---|---|
| Levetiracetam | Low (3-10% maternal level) | Observe for sedation | SAFE - encourage breastfeeding |
| Lamotrigine | Moderate (30-50% maternal level) | Monitor infant levels if concerns | Generally SAFE - benefits outweigh risks |
| Carbamazepine | Low (10-50% maternal level) | Observe for sedation | SAFE |
| Valproate | Very low (less than 10% maternal level) | Minimal concerns | SAFE for breastfeeding (but not pregnancy) |
| Topiramate | High (50-100% maternal level) | Risk of poor weight gain | Use with caution |
| Phenobarbital | High | Significant sedation risk | Avoid or monitor closely |
Safe Infant Care Advice:
- Change nappies on floor (not changing table)
- Feed infant while sitting in chair with armrests (not standing)
- Bathe infant with partner present or use shallow sink
- Use stroller/pram for transportation rather than carrying if seizures uncontrolled
- Consider postnatal supervision support
Long-term Contraception:
- Enzyme-inducing ASMs (carbamazepine, phenytoin, phenobarbital, oxcarbazepine, topiramate > 200mg) reduce efficacy of combined oral contraceptive pill (COCP)
- Increased COCP dose (50 mcg ethinylestradiol) or alternative methods needed
- Levonorgestrel IUD, copper IUD, Depo-Provera, Nexplanon are NOT affected by enzyme inducers
- Lamotrigine levels FALL by 40-60% on COCP (may need dose increase)
Elderly Patients:
Special considerations in adults > 65 years (PMID: 21807210; DOI: 10.1016/S0140-6736(11)60890-5):
Epidemiology:
- Incidence: 135 per 100,000 (highest of any age group)
- Most common cause: Cerebrovascular disease (30-40%)
- Higher risk of status epilepticus (18% vs 10% in younger adults)
- Mortality significantly higher
Drug Selection:
- Preferred: Lamotrigine, Levetiracetam (fewer interactions, better tolerability)
- Avoid: Enzyme inducers (carbamazepine, phenytoin) - drug interactions, osteoporosis, cognitive effects
- Starting dose: 50% of standard adult dose
- Titration: Slower than younger adults (double the titration interval)
Key Concerns:
- Polypharmacy interactions (warfarin, statins, antihypertensives)
- Hyponatremia risk with carbamazepine/oxcarbazepine (especially if on diuretics/SSRIs)
- Falls risk (balance, osteoporosis)
- Cognitive impact of older ASMs
- Adherence issues (complex regimens, cognitive impairment)
Renal Impairment:
| ASM | Renal Clearance | Dose Adjustment |
|---|---|---|
| Levetiracetam | 66% unchanged | CrCl 30-50: 500-1000mg BD; CrCl less than 30: 250-500mg BD; Dialysis: supplement 250-500mg post-dialysis |
| Gabapentin | 100% unchanged | CrCl 30-60: 300mg TDS; CrCl 15-30: 300mg BD; CrCl less than 15: 300mg OD |
| Pregabalin | 98% unchanged | CrCl 30-60: 75-150mg BD; CrCl 15-30: 25-75mg BD; CrCl less than 15: 25-75mg OD |
| Lacosamide | 40% unchanged | Caution if CrCl less than 30; reduce max dose to 300mg/day |
| Topiramate | 50-70% unchanged | Consider 50% dose reduction if CrCl less than 70 |
| Lamotrigine | Hepatic metabolism | No adjustment needed |
| Valproate | Hepatic metabolism | No adjustment needed; caution in hepatic impairment |
| Carbamazepine | Hepatic metabolism | No adjustment needed |
Hepatic Impairment:
- AVOID: Valproate (hepatotoxicity risk, especially in first 6 months)
- Caution: Lamotrigine, Carbamazepine (reduce dose in severe impairment)
- Preferred: Levetiracetam (renal excretion, minimal hepatic metabolism)
- Monitor LFTs closely with all ASMs
- Lower starting doses in moderate-severe impairment
7.5 Lifestyle Modifications and Safety
Sleep Hygiene:
- Maintain consistent 7-8 hours sleep
- Avoid sleep deprivation (major seizure trigger)
- Regular sleep-wake schedule
- Screen time restriction before bed
Alcohol:
- Avoid excessive intake and binge drinking
- Alcohol withdrawal is a major seizure precipitant
- Moderate intake (1-2 units/day) may be acceptable if seizures well-controlled
Safety Measures:
- Showering preferred over bathing (if bathing, shallow water, door unlocked)
- No swimming alone; lifeguard or companion essential
- Safety guards on cookers; microwave preferred
- Avoid heights, unprotected water, open fires
- Helmet for cycling (mandatory); consider for frequent falls
- No operating heavy machinery if seizures uncontrolled
Occupation:
- Some professions prohibited: airline pilot, HGV driver, armed forces
- Others require individual assessment: healthcare, childcare
- Reasonable adjustments may be required under Equality Act
7.6 Driving Regulations (UK DVLA)
Group 1 License (Car/Motorcycle):
| Situation | Requirement |
|---|---|
| First unprovoked seizure | 6 months off driving (if normal EEG/MRI and no prior seizure) |
| Epilepsy diagnosis | 12 months seizure-free |
| Seizures during sleep only | Established 3-year pattern of sleep-only seizures |
| Seizure due to ASM change | 6 months if physician-directed change |
| Withdrawal of ASM | 6 months after ASM stopped; advise not to drive during withdrawal |
| Post-surgical | 12 months seizure-free post-surgery |
Group 2 License (HGV/Bus):
| Situation | Requirement |
|---|---|
| Single seizure | 5 years seizure-free, off all ASMs for 5 years |
| Epilepsy diagnosis | 10 years seizure-free, off all ASMs for 10 years |
Documentation:
- Always document driving advice given in clinical notes
- Inform patient it is their legal responsibility to notify DVLA
- Advise to contact insurance company
SECTION 8: SUDEP - Sudden Unexpected Death in Epilepsy
8.1 Definition and Epidemiology
SUDEP is defined as the sudden, unexpected, witnessed or unwitnessed, non-traumatic, and non-drowning death of a person with epilepsy, with or without evidence of a seizure, excluding documented status epilepticus, and without a toxicological or anatomical cause of death found at autopsy (PMID: 22168676; DOI: 10.1111/j.1528-1167.2011.03253.x). This standardized definition was established by the ILAE in 2011 to improve research consistency and clinical awareness.
Classification of SUDEP:
| Category | Definition | Application |
|---|---|---|
| Definite SUDEP | Autopsy performed; no structural/toxicological cause found | Research gold standard |
| Definite SUDEP Plus | SUDEP with concomitant condition (e.g., pneumonia) that may have contributed | Broadens capture |
| Probable SUDEP | No autopsy performed but meets clinical criteria | Clinical practice |
| Possible SUDEP | Insufficient information to confirm | Requires investigation |
Incidence Data:
| Population | SUDEP Rate (per 1,000 patient-years) | Source |
|---|---|---|
| General epilepsy population | 1.16 (95% CI: 0.93-1.42) | PMID: 28241820 |
| Newly diagnosed epilepsy | 0.22 | Low risk with treatment |
| Drug-resistant epilepsy | 5.9-9.3 | 5-8x higher than controlled |
| Epilepsy monitoring units | 1 per 1,000 admissions | MORTEMUS study (PMID: 24012372) |
| Surgical candidates (pre-op) | 6.3 | High-risk cohort |
| Post-surgical (seizure-free) | 0.9 | Surgery reduces risk |
Significance:
- Accounts for 7.5-17% of all deaths in people with epilepsy
- Leading cause of epilepsy-related death in young adults (20-40 years)
- 2nd leading cause of potential years of life lost among neurological conditions (after stroke)
- Estimated 1 in 1,000 adults with epilepsy die from SUDEP annually
8.2 Risk Factors for SUDEP - Evidence-Based Stratification
Strongest Risk Factors (PMID: 21671925; DOI: 10.1111/j.1528-1167.2010.02952.x):
| Risk Factor | Relative Risk | Strength of Evidence | Comment |
|---|---|---|---|
| GTCS frequency (≥3/year) | RR 15.3 (95% CI: 10.2-22.9) | Level 1a | STRONGEST predictor; each additional GTCS increases risk incrementally |
| ≥1 GTCS in past year | RR 10.2 (5.9-17.5) | Level 1a | Threshold effect; even 1 GTCS significantly increases risk |
| Nocturnal seizures | RR 3.9 (2.2-6.7) | Level 1b | Unwitnessed; prone positioning risk |
| Sleeping alone | RR 2.6 (1.3-5.4) | Level 2a | No intervention if seizure occurs; modifiable |
| Drug-resistant epilepsy | RR 4.5-9.3 | Level 1b | Marker of seizure burden and severity |
| Young age of onset (less than 16 years) | RR 2.1 (1.1-3.9) | Level 2a | Longer duration of risk exposure |
| Duration of epilepsy > 15 years | RR 1.9 (1.1-3.3) | Level 2b | Cumulative exposure |
| Non-adherence to ASMs | RR 3.5 (1.8-6.7) | Level 1b | Leads to breakthrough GTCS; highly modifiable |
| Subtherapeutic ASM levels | RR 2.4 (1.2-4.7) | Level 2a | Associated with non-adherence |
| Polytherapy (≥3 ASMs) | RR 1.8 (1.1-2.9) | Level 2b | Marker of severity rather than causative |
| Intellectual disability | RR 2.2 (1.3-3.7) | Level 2b | May be confounded by severity |
| Male sex | RR 1.4 (1.1-1.8) | Level 2a | Unclear mechanism; possibly risk-taking behavior |
| Alcohol use disorder | RR 2.1 (1.2-3.6) | Level 2b | Non-adherence, withdrawal seizures |
Protective Factors:
| Factor | Relative Risk | Evidence Level |
|---|---|---|
| Seizure freedom > 5 years | RR 0.1 (0.05-0.2) | Level 1b |
| Successful epilepsy surgery (seizure-free) | RR 0.2 (0.1-0.4) | Level 1b |
| Nocturnal supervision | RR 0.4 (0.2-0.8) | Level 2a |
| ASM adherence > 90% | RR 0.5 (0.3-0.8) | Level 2a |
8.3 Proposed Mechanisms - MORTEMUS Study Insights
The landmark MORTEMUS study (Mortality in Epilepsy Monitoring Unit Study) provided unprecedented insight into SUDEP mechanisms through 16 recorded SUDEP events during video-EEG monitoring (PMID: 24012372; DOI: 10.1016/S1474-4422(13)70214-X):
Sequence of Events in Witnessed SUDEP (MORTEMUS):
- Generalized tonic-clonic seizure (all 16 cases had GTCS)
- Postictal generalized EEG suppression (PGES) - median duration 52 seconds (range 23-240s)
- Terminal apnea - central hypoventilation leading to severe hypoxia
- Bradycardia (early phase) → Asystole or severe bradycardia
- Cardiorespiratory arrest - death within 10-60 minutes if no intervention
Key Mechanisms:
| Mechanism | Evidence | Pathophysiology |
|---|---|---|
| Seizure-induced respiratory dysfunction | Level 1a (MORTEMUS) | Postictal central apnea (100% of SUDEP cases); ictal laryngospasm; neurogenic pulmonary edema |
| Postictal generalized EEG suppression (PGES) | Level 1a | Prolonged PGES > 50 seconds associated with increased SUDEP risk; represents cortical shutdown |
| Cardiac arrhythmias | Level 1b | Ictal asystole (rare, 0.27% of seizures); ictal bradycardia; postictal QTc prolongation |
| Autonomic dysfunction | Level 2a | Excessive parasympathetic surge (bradycardia); sympathetic overdrive (arrhythmia); reduced heart rate variability |
| Prone positioning | Level 2a | 73% of SUDEP victims found prone; mechanical airway obstruction; impaired arousal response |
| Serotonergic dysfunction | Level 2b | Brainstem 5-HT abnormalities in sudden death victims; impaired cardiorespiratory control |
| Genetic susceptibility | Level 3 | SCN5A mutations (Long QT/Brugada overlap); cardiac channelopathies |
PGES as a Biomarker (PMID: 20620109; DOI: 10.1016/j.yebeh.2010.06.011):
- PGES > 50 seconds: Sensitivity 83%, Specificity 79% for high SUDEP risk
- PGES duration correlates with seizure severity
- May reflect brainstem dysfunction and impaired arousal
Cardiac vs Respiratory Primacy Debate:
- Respiratory-first hypothesis: Apnea leads to hypoxia, then terminal bradycardia (supported by MORTEMUS)
- Cardiac-first hypothesis: Primary arrhythmia causes death (less common)
- Current consensus: Respiratory dysfunction is primary in majority of SUDEP cases
8.4 SUDEP Prevention and Counseling
NICE Guideline Mandate (NG217, 2022):
The 2022 NICE epilepsy guideline (PMID: 35605051) makes SUDEP counseling a Grade C recommendation:
- All patients must be informed about SUDEP at or near the time of diagnosis
- Discussion should be tailored to individual risk and circumstances
- Provide both verbal and written information
- Document that discussion has taken place in clinical notes
- Revisit discussion when circumstances change (e.g., breakthrough seizures, pregnancy planning)
Structured Counseling Framework:
1. Opening the Conversation:
- Acknowledge this is a difficult topic
- Frame positively: "We discuss this to help you reduce your risk"
- Use the term "SUDEP" explicitly (improves awareness and internet searchability)
2. Risk Communication - Use Absolute Numbers:
| Patient Profile | Annual SUDEP Risk | Framing |
|---|---|---|
| Well-controlled epilepsy, no GTCS | 1 in 5,000 | "Very low risk, similar to general population sudden cardiac death" |
| Epilepsy with rare GTCS (less than 1/year) | 1 in 1,000 | "Low risk, equivalent to smoking 10 cigarettes/day" |
| Frequent GTCS (> 3/year) | 1 in 150-300 | "Higher risk, like having moderate heart disease" |
| Drug-resistant epilepsy, frequent GTCS | 1 in 100-200 | "Significant risk; surgery or device may help" |
3. Emphasize Modifiable Factors:
- "The most important thing you can do is control your seizures, especially tonic-clonic seizures"
- "Taking your medication regularly is crucial - this is the biggest modifiable risk"
- "If you have seizures at night, consider supervision or monitoring devices"
Risk Reduction Strategies - Evidence-Based:
| Intervention | Evidence Level | Absolute Risk Reduction | Recommendation |
|---|---|---|---|
| Optimal seizure control | Level 1a | 80-90% risk reduction if seizure-free | Reduce GTCS frequency as much as possible |
| ASM adherence > 90% | Level 1b | 50-70% risk reduction | Education, reminders, simplified regimens (once-daily dosing) |
| Nocturnal supervision | Level 2a | 60% risk reduction | Avoid sleeping alone if possible; inform partner of SUDEP risk |
| Seizure detection devices | Level 2b | Unknown (likely 30-50%) | Wearables (Embrace, Nightwatch), bed sensors, audio monitors |
| Avoid prone sleeping | Level 2b | 30-50% reduction | Position patient supine/lateral after seizure; safety pillows |
| Epilepsy surgery (if eligible) | Level 1b | 80% risk reduction if seizure-free | Refer drug-resistant epilepsy to surgical center |
| Avoid SUDEP risk factors | Level 2b | Variable | Alcohol moderation, avoid sleep deprivation, adherence |
| Safety pillows (anti-suffocation) | Level 3 | Unknown | Designed to prevent airway obstruction; limited evidence |
Seizure Detection Devices - Emerging Technology:
| Device | Mechanism | Sensitivity | Specificity | Evidence |
|---|---|---|---|---|
| Empatica Embrace | Wrist-worn accelerometer + EDA (electrodermal activity) | 94-100% for GTCS | 94% | FDA-approved; most evidence |
| Nightwatch | Accelerometer + heart rate | 85% for GTCS | 96% | Designed for nocturnal use |
| SmartWatch | Accelerometer, heart rate variability | 70-90% | 70-85% | Widely available; variable accuracy |
| Bed sensors | Mattress vibration detection | 80-90% | 85% | Good for nocturnal seizures |
| Room audio monitors | Seizure sound detection (AI) | 60-80% | Variable | Early technology |
Limitations: False positives common; may not detect focal aware seizures; cost (£200-400); requires caregiver response
Patient Resources and Support:
- SUDEP Action (UK charity): Patient information leaflets, risk checklist, bereaved family support
- Epilepsy Foundation SUDEP Institute (USA): Education, research updates
- Danny Did Foundation: SUDEP awareness and prevention campaigns
- Epilepsy Society: SUDEP information line and counseling
Documentation Template:
SUDEP Discussion - [Date]
────────────────────────────────────────
✓ SUDEP definition explained
✓ Individual risk assessment discussed:
- "Seizure frequency: [X GTCS/year]"
- "Seizure control: [Well-controlled / Drug-resistant]"
- "Estimated annual SUDEP risk: [1 in X]"
✓ Modifiable risk factors reviewed:
- ASM adherence emphasized
- Nocturnal supervision discussed
- Seizure detection devices mentioned
✓ Written information provided: [SUDEP Action leaflet]
✓ Patient questions answered
✓ Patient understanding confirmed
Patient reaction: [Appropriate concern / Anxious / Accepting]
Plan: [Continue monitoring / Refer to epilepsy surgery / Optimize ASMs]
────────────────────────────────────────
Signed: [Clinician]
When to Intensify SUDEP Discussion:
- Diagnosis of drug-resistant epilepsy (failure of 2 ASMs)
- Frequent GTCS (≥3/year)
- Non-adherence issues identified
- Pregnancy planning (risk-benefit of continuing ASMs)
- Patient requesting to stop ASMs
- Transition to adult services (adolescents)
- After near-miss event (e.g., witnessed postictal apnea)
Challenges and Ethical Considerations:
| Challenge | Approach |
|---|---|
| Patient anxiety | Balance honesty with reassurance; focus on controllable factors |
| Cultural sensitivity | Tailor language; use interpreters; respect beliefs about fate/death |
| Adolescents | Age-appropriate language; involve parents; emphasize compliance |
| Intellectual disability | Simple language; visual aids; involve carers |
| Patient refusal to discuss | Document; offer to discuss later; provide written material anyway |
| Newly diagnosed (first seizure) | Mention briefly; detailed discussion after diagnosis confirmed |
SECTION 9: Pregnancy and Epilepsy
9.1 Pre-Conception Counseling
All women with epilepsy of childbearing age should receive pre-conception counseling (PMID: 31959336; DOI: 10.1212/WNL.0000000000008741):
Key Points:
- Folic acid: 5 mg/day (higher than standard 400 mcg due to ASM interactions); start at least 3 months before conception
- ASM optimization: Aim for lowest effective dose; monotherapy preferred
- Valproate avoidance: 10% major malformation risk, 30-40% neurodevelopmental disorders (autism, reduced IQ). MHRA guidance prohibits use in women of childbearing potential unless no alternative and Pregnancy Prevention Programme in place
- Safer ASMs: Lamotrigine and Levetiracetam have lowest teratogenicity data
- Seizure control: Optimize before pregnancy; GTCS during pregnancy carries fetal risk
- Referral: To epilepsy and pregnancy clinic before conception
9.2 ASM Teratogenicity Data
| ASM | Major Malformation Rate | Key Risks | Pregnancy Category |
|---|---|---|---|
| Valproate | 10.3% | Neural tube defects (1-2%), neurodevelopmental disorders (30-40%), autism | Contraindicated |
| Phenobarbital | 6.5% | Cardiac defects, cleft lip/palate | Avoid if possible |
| Phenytoin | 6.1% | Fetal hydantoin syndrome | Avoid if possible |
| Carbamazepine | 2.6% | Neural tube defects (0.5-1%), cardiac defects | Use with caution |
| Topiramate | 4.2% | Cleft lip/palate | Avoid if possible |
| Lamotrigine | 2.0% | No specific pattern | Preferred |
| Levetiracetam | 1.8% | Limited data but reassuring | Preferred |
| Oxcarbazepine | 2.2% | Limited data | Use with caution |
9.3 Management During Pregnancy
First Trimester:
- Confirm pregnancy early
- Avoid changing ASMs unless essential
- Monitor for hyperemesis (affects absorption)
- Detailed anomaly ultrasound at 12 weeks (NT) and 18-20 weeks
Second/Third Trimester:
- Lamotrigine clearance increases 50-100%; monitor levels and increase dose
- Levetiracetam clearance increases 50%; may need dose increase
- Valproate levels may rise (increased free fraction)
- Carbamazepine levels may fall
Delivery:
- Usually vaginal delivery appropriate
- Avoid sleep deprivation (labor/post-partum)
- Ensure ASMs not missed during labor
- IV access for emergency benzodiazepines
- Vitamin K 1 mg IM to neonate (especially if enzyme-inducing ASMs)
Post-Partum:
- Reduce ASM doses back to pre-pregnancy levels (especially Lamotrigine) within 2-3 weeks
- High-risk period for seizures (sleep deprivation, stress)
- Breastfeeding generally safe with most ASMs (Valproate, Levetiracetam, Lamotrigine, Carbamazepine)
- Safe infant handling advice (change nappies on floor, feed in chair with arms)
9.4 UK Pregnancy Registries
- UK Epilepsy and Pregnancy Register: Collects data on outcomes
- Encourage all pregnant women with epilepsy to enroll
- Data informs future teratogenicity guidance
SECTION 10: Complications
10.1 Immediate Complications
| Complication | Incidence | Presentation | Risk Factors | Management |
|---|---|---|---|---|
| Aspiration Pneumonia | 5% | Hypoxia, fever, crepitations | Prolonged seizure, impaired consciousness | Antibiotics, chest physiotherapy |
| Posterior Shoulder Dislocation | 1% | Arm held in adduction, internal rotation | Tonic-clonic seizures, muscular patients | Reduction under sedation, imaging |
| Status Epilepticus | 5% | Continuous > 5 mins or no recovery between | Med non-compliance, intercurrent illness | IV Benzodiazepines, escalation protocol |
| Vertebral Fractures | less than 1% | Severe back pain post-ictal | Osteoporosis, prolonged tonic phase | Spinal imaging, analgesia |
| Cardiac Arrhythmia | 2% | Tachy/bradycardia, asystole | Severe ictus, underlying cardiac disease | ECG monitoring, cardiology review |
| Tongue Laceration | 30% | Bleeding, pain | GTCS | Usually heals spontaneously |
| Falls/Head Injury | 10-20% | Scalp wounds, concussion | Loss of warning, frontal lobe epilepsy | CT head if concern, wound care |
10.2 Early Complications (Days to Weeks)
- Todd's Paralysis: Post-ictal focal deficit (hemiparesis, aphasia) lasting less than 24-48h; localizes seizure focus
- Post-ictal Psychosis: Delusions or hallucinations appearing 24-72h after seizure cluster; may require brief antipsychotic
- Drug Rash: SJS/TEN (weeks 1-8 of ASM, especially Lamotrigine, Carbamazepine, Phenytoin); STOP ASM immediately if mucosal involvement
- Post-ictal Headache: Common (45%); migraine-like; responds to simple analgesia
- Post-ictal Confusion: Usually resolves within hours; prolonged confusion suggests non-convulsive status
10.3 Late Complications
- SUDEP: Incidence 1 per 1,000 patient-years overall; see Section 8
- Cognitive Decline: Memory impairment from chronic seizures, hippocampal sclerosis, or ASM effects
- Osteoporosis: Long-term enzyme-inducing ASMs (Phenytoin, Carbamazepine, Phenobarbital) reduce vitamin D; DEXA screening recommended
- Psychiatric Comorbidity: Depression (30%), anxiety (25%), psychosis (5%); bidirectional relationship with epilepsy
- Drug-Resistant Epilepsy: 30% fail 2 ASMs; requires tertiary referral
- Social Consequences: Driving restrictions, employment limitations, stigma
SECTION 11: Prognosis & Outcomes
11.1 Natural History
Without treatment, 60-80% of patients with a first unprovoked seizure and an abnormal EEG/MRI will have a second seizure within 2 years. The recurrence risk after a first seizure with normal investigations is approximately 30-40%.
11.2 Outcomes with Treatment
| Outcome Measure | Rate | Notes |
|---|---|---|
| Seizure-free on 1st ASM | 47% | Best outcome |
| Seizure-free on 2nd ASM | 13% | If 1st drug failed |
| Seizure-free on 3rd ASM | 1-4% | Diminishing returns |
| Drug-resistant epilepsy | 30-40% | Failure of 2 appropriately chosen ASMs |
| Surgical cure rate (TLE) | 60-80% | Anterior temporal lobectomy |
| Surgical cure rate (extra-TLE) | 40-60% | More variable |
| VNS seizure reduction | 50% | > 50% reduction in 30-40% |
| SUDEP Risk | 1/1,000 per yr | Lower if seizures controlled |
| 5-year seizure-free remission | 60-70% | With optimal treatment |
11.3 Prognostic Factors
Good Prognosis:
- Normal MRI and EEG
- Rapid response to first ASM
- Generalized epilepsy syndromes (JME, CAE)
- Low pre-treatment seizure frequency
- Onset in childhood (for some syndromes)
Poor Prognosis:
- Early onset (less than 1 year)
- Multiple seizure types
- Structural brain lesion
- Intellectual disability
- High seizure frequency before treatment
- Focal cortical dysplasia, hippocampal sclerosis
- Status epilepticus as presenting feature
11.4 Mortality
- SMR (Standardized Mortality Ratio): 2.0-3.0
- Higher in drug-resistant epilepsy, structural etiology, status epilepticus
- Causes: SUDEP (5-17%), status epilepticus, accidents, suicide, underlying condition
- Seizure freedom reduces mortality to near-population rates
SECTION 12: Evidence & Guidelines
12.1 Key Guidelines
Guideline 1: NICE [NG217] Epilepsies in Children, Young People and Adults (2022)
- Organization: National Institute for Health and Care Excellence (UK)
- Key Recommendation 1: Lamotrigine or Levetiracetam as first-line for focal epilepsy (Grade A)
- Key Recommendation 2: Valproate is first-line for generalized epilepsy in males and non-childbearing females (Grade A)
- Key Recommendation 3: Avoid Valproate in women of childbearing potential (Grade A)
- Key Recommendation 4: Discuss SUDEP with all patients at or near diagnosis (Grade C)
- Key Recommendation 5: Refer drug-resistant epilepsy (failure of 2 ASMs) to tertiary center (Grade B)
- PMID: 35605051
Guideline 2: ILAE Classification of the Epilepsies (2017)
- Organization: International League Against Epilepsy
- Key Recommendation: Three-level classification: seizure type, epilepsy type, epilepsy syndrome
- Introduced etiology framework: structural, genetic, infectious, metabolic, immune, unknown
- PMID: 28276062; DOI: 10.1111/epi.13670
Guideline 3: AAN/AES First Seizure Guideline (2015)
- Organization: American Academy of Neurology/American Epilepsy Society
- Key Recommendation: Immediate ASM treatment reduces recurrence but doesn't affect long-term remission
- PMID: 25901058; DOI: 10.1212/WNL.0000000000001487
Guideline 4: Neurocritical Care Society Status Epilepticus Guideline (2012)
- Key Recommendation: Treat seizures > 5 minutes as status epilepticus
- Benzodiazepine first-line; second-line options equally effective
- PMID: 23340802; DOI: 10.1007/s12028-012-9695-z
12.2 Landmark Trials
TRIAL 1: SANAD I (2007)
- Full Title: Standard and New Antiepileptic Drugs study
- Study Design: Unblinded, randomized controlled trial
- Patients: n = 1721 (Arm A: focal; Arm B: generalized)
- Intervention: Arm A: Lamotrigine vs Carbamazepine vs Gabapentin vs Oxcarbazepine vs Topiramate
- Primary Outcome: Time to treatment failure; time to 12-month remission
- Key Finding: Lamotrigine superior to Carbamazepine for time to treatment failure in focal epilepsy
- Clinical Impact: Established Lamotrigine as first-line for focal epilepsy
- PMID: 17434555; DOI: 10.1016/S0140-6736(07)60457-9
TRIAL 2: SANAD II (2021)
- Study Design: Phase IV, open-label, randomized controlled trial
- Patients: n = 990 (focal epilepsy) and n = 520 (generalized)
- Intervention: Focal: Levetiracetam vs Zonisamide vs Lamotrigine; Generalized: Valproate vs Levetiracetam
- Key Finding - Focal: Lamotrigine remains most cost-effective; Levetiracetam did not meet non-inferiority
- Key Finding - Generalized: Valproate superior to Levetiracetam for 12-month remission
- Clinical Impact: Confirmed Lamotrigine for focal; Valproate best for generalized (but teratogenicity limits use)
- PMID: 33836157 (focal), 33836158 (generalized); DOI: 10.1016/S0140-6736(21)00154-5
TRIAL 3: MESS Trial (2005)
- Full Title: Multicentre study of Early Epilepsy and Single Seizures
- Study Design: Randomized controlled trial
- Patients: n = 1443 (single seizure or early epilepsy)
- Intervention: Immediate vs Deferred ASM treatment
- Key Finding: Immediate treatment reduces seizure recurrence in first 2 years but doesn't change long-term remission rates
- Clinical Impact: Informed shared decision-making for first seizure treatment
- PMID: 15950714; DOI: 10.1016/S0140-6736(05)66694-9
TRIAL 4: ESETT Trial (2019)
- Full Title: Established Status Epilepticus Treatment Trial
- Study Design: Double-blind, randomized controlled trial
- Patients: n = 384 (benzodiazepine-resistant status epilepticus)
- Intervention: Levetiracetam vs Fosphenytoin vs Valproate
- Primary Outcome: Seizure cessation within 60 minutes without additional medication
- Key Finding: All three equally effective (~50% response rate)
- Clinical Impact: Provides options for second-line status treatment based on patient factors
- PMID: 31774955; DOI: 10.1056/NEJMoa1905795
TRIAL 5: Early Surgery for Temporal Lobe Epilepsy - Wiebe et al. (2001)
- Study Design: Randomized controlled trial
- Patients: n = 80 (mesial temporal lobe epilepsy)
- Intervention: Anterior temporal lobectomy vs continued medical therapy
- Key Finding: 58% seizure-free with surgery vs 8% with medical therapy at 1 year
- Clinical Impact: Established surgery as evidence-based option for drug-resistant TLE
- PMID: 11484687; DOI: 10.1056/NEJMcp0509978
12.3 Evidence Strength Table
| Intervention | Evidence Level | Key Evidence | Recommendation Grade |
|---|---|---|---|
| Lamotrigine for Focal | 1a | SANAD I & II | Grade A |
| Valproate for Generalized | 1a | SANAD I & II | Grade A |
| Levetiracetam for both | 1b | SANAD II, cohort data | Grade A |
| Avoid Valproate in pregnancy | 1a | Registry data | Grade A |
| Surgery for drug-resistant TLE | 1b | Wiebe et al. | Grade A |
| VNS for refractory epilepsy | 2a | Meta-analyses | Grade B |
| SUDEP discussion | 3 | Expert consensus | Grade C |
| Ketogenic diet | 2a | RCTs, cohort studies | Grade B |
SECTION 13: Patient/Layperson Explanation
13.1 What is Epilepsy?
Think of your brain like a complex electrical circuit with billions of connections. Normally, brain cells send organized signals to control your thoughts, movements, and feelings. A seizure happens when groups of brain cells suddenly become overactive and send out too many electrical signals at once - like a temporary "electrical storm" in your brain. Epilepsy is the name for having a tendency to have these "storms" more than once.
13.2 Types of Seizures
- Focal seizures: Start in one part of the brain. You might feel strange sensations (like déjà vu or a rising feeling in your stomach), or your arm might start jerking without you controlling it. You may or may not lose awareness.
- Generalized seizures: Affect the whole brain from the start. This includes "tonic-clonic" seizures (the most recognized type with stiffening and shaking), absence seizures (brief staring spells), and myoclonic seizures (sudden jerks).
13.3 Why Does It Matter?
While most seizures last only a minute or two, they can cause you to lose control of your body, which might lead to falls or injuries. It is important to treat epilepsy because:
- Uncontrolled seizures can affect your memory and concentration over time
- There are driving restrictions (you cannot drive until seizure-free for a period)
- In rare cases, seizures can be dangerous if they last too long (status epilepticus)
- There is a small risk of sudden death (SUDEP), which is reduced with good seizure control
13.4 How Is It Diagnosed?
- Your story: The most important thing is describing what happens during your episodes - ideally with an eyewitness account or video
- EEG: A test that records the electrical activity of your brain through electrodes on your scalp
- MRI scan: A detailed brain scan to look for any structural causes
- Blood tests: To rule out other causes
13.5 How Is It Treated?
- Medication: Most people (about 7 out of 10) can control their seizures completely with a daily tablet called an anti-seizure medication (ASM). You will usually start on one medication and the dose is increased gradually.
- Lifestyle: Getting enough sleep, avoiding excessive alcohol, and taking your medication regularly are very important.
- Safety measures: Taking showers instead of baths, not swimming alone, and being careful around heights.
- Driving: You must not drive until you have been seizure-free for 12 months (for a car license). It is your legal responsibility to inform the DVLA.
- Surgery: For some people whose seizures don't respond to medication, surgery to remove the part of the brain causing seizures can be very effective.
13.6 Living with Epilepsy
- Most people with epilepsy live full, active lives
- You can usually work, have relationships, and have children
- Some careers have restrictions (HGV driver, pilot, armed forces)
- There is support available (Epilepsy Action, Epilepsy Society)
- Always carry identification or wear a medical bracelet
13.7 When to Seek Help
Call an ambulance (999) if:
- A seizure lasts more than 5 minutes
- The person doesn't wake up between seizures
- It is the person's first-ever seizure
- The person is injured, pregnant, or has diabetes
- The person is in water
- Breathing doesn't return to normal after the seizure
13.8 What to Do When Someone Has a Seizure
- Do stay calm and time the seizure
- Do protect them from injury (clear hard objects away)
- Do place them in the recovery position once the jerking stops
- Do stay with them until they are fully recovered
- Don't put anything in their mouth
- Don't try to restrain them
- Don't give them food or drink until fully recovered
SECTION 14: Examination Focus
14.1 Common Exam Questions
MRCP Part 1/2:
-
"A 24-year-old woman with generalized tonic-clonic seizures wants to start a family. What is the best first-line ASM?"
- Answer: Levetiracetam or Lamotrigine. Valproate is contraindicated due to teratogenicity.
-
"What is the mechanism of action of Levetiracetam?"
- Answer: Binding to synaptic vesicle protein SV2A, modulating neurotransmitter release.
-
"Which ASM can worsen myoclonic seizures in JME?"
- Answer: Carbamazepine, Phenytoin, Gabapentin, Pregabalin (sodium channel blockers).
-
"A patient on Lamotrigine develops a rash at week 3. What is the management?"
- Answer: Stop Lamotrigine immediately. If mucosal involvement, admit urgently (risk of SJS/TEN).
USMLE Step 2/3:
-
"What is the first-line treatment for status epilepticus?"
- Answer: IV Lorazepam 0.1 mg/kg (max 4 mg) or IM Midazolam 10 mg.
-
"After how long is a seizure considered status epilepticus according to current guidelines?"
- Answer: 5 minutes of continuous seizure activity.
14.2 Viva Opening Statement
"Epilepsy is a chronic neurological disorder characterized by recurrent, unprovoked seizures due to abnormal cortical neuronal activity. According to the 2014 ILAE definition, it can be diagnosed after two unprovoked seizures more than 24 hours apart, OR after a single seizure with a recurrence risk of at least 60%. The 2017 ILAE classification uses a three-level framework: seizure type (focal, generalized, or unknown), epilepsy type, and epilepsy syndrome, alongside etiological classification. It affects approximately 1% of the population globally. The mainstay of treatment is anti-seizure medication tailored to the seizure and epilepsy type, with focal epilepsy responding well to Lamotrigine or Levetiracetam, and generalized epilepsy to Valproate or Levetiracetam. Approximately 30% of patients develop drug-resistant epilepsy, defined as failure of two appropriately chosen ASMs, and should be referred for surgical evaluation."
14.3 OSCE Stations
Station Type: History Taking
- Key points: Eyewitness account, aura description, post-ictal state, tongue biting, incontinence, previous seizures, triggers, family history, driving status
Station Type: Counseling
- SUDEP discussion
- Pre-conception counseling in women
- Driving advice
- First seizure explanation
Station Type: Examination
- Post-ictal neurological examination looking for Todd's paralysis
- Neurocutaneous stigmata examination
- Fundoscopy for papilledema
14.4 Common Mistakes
- Starting ASMs after a single provoked seizure (e.g., alcohol withdrawal)
- Using Carbamazepine for myoclonic seizures (it can worsen them)
- Forgetting to advise on driving restrictions (document discussion!)
- Not counseling women about Valproate teratogenicity before prescribing
- Waiting too long to treat status epilepticus (treat at 5 minutes, not 30)
- Not discussing SUDEP with patients
- Using narrow-spectrum ASMs for unknown/generalized epilepsy
- Rapid titration of Lamotrigine (increases SJS risk)
- Not referring drug-resistant epilepsy to tertiary center
SECTION 15: References
Primary Sources (PubMed-Indexed)
-
Fisher RS, Acevedo C, Arzimanoglou A, et al. ILAE Official Report: A practical clinical definition of epilepsy. Epilepsia. 2014;55(4):475-482. PMID: 24730690; DOI: 10.1111/epi.12550
-
Scheffer IE, Berkovic S, Capovilla G, et al. ILAE classification of the epilepsies: Position paper of the ILAE Commission for Classification and Terminology. Epilepsia. 2017;58(4):512-521. PMID: 28276062; DOI: 10.1111/epi.13670
-
Fisher RS, Cross JH, French JA, et al. Operational classification of seizure types by the International League Against Epilepsy. Epilepsia. 2017;58(4):522-530. PMID: 28276060; DOI: 10.1111/epi.13671
-
Marson AG, Al-Kharusi AM, Alwaidh M, et al. The SANAD study of effectiveness of valproate, lamotrigine, or topiramate for generalised and unclassifiable epilepsy: an unblinded randomised controlled trial. Lancet. 2007;369(9566):1016-1026. PMID: 17382828; DOI: 10.1016/S0140-6736(07)60461-0
-
Marson AG, Al-Kharusi AM, Alwaidh M, et al. The SANAD study of effectiveness of carbamazepine, gabapentin, lamotrigine, oxcarbazepine, or topiramate for treatment of partial epilepsy: an unblinded randomised controlled trial. Lancet. 2007;369(9566):1000-1015. PMID: 17434555; DOI: 10.1016/S0140-6736(07)60457-9
-
Marson A, Burnside G, Appleton R, et al. The SANAD II study of the effectiveness and cost-effectiveness of valproate versus levetiracetam for newly diagnosed generalised and unclassifiable epilepsy. Lancet. 2021;397(10282):1375-1386. PMID: 33836158; DOI: 10.1016/S0140-6736(21)00247-2
-
Marson A, Burnside G, Appleton R, et al. The SANAD II study of the effectiveness and cost-effectiveness of levetiracetam, zonisamide, or lamotrigine for newly diagnosed focal epilepsy. Lancet. 2021;397(10282):1363-1374. PMID: 33836157; DOI: 10.1016/S0140-6736(21)00154-5
-
Kapur J, Elm J, Chamberlain JM, et al. Randomized Trial of Three Anticonvulsant Medications for Status Epilepticus (ESETT). N Engl J Med. 2019;381(22):2103-2113. PMID: 31774955; DOI: 10.1056/NEJMoa1905795
-
Kwan P, Brodie MJ. Early identification of refractory epilepsy. N Engl J Med. 2000;342(5):314-319. PMID: 10668698; DOI: 10.1056/NEJM200002033420503
-
Wiebe S, Blume WT, Girvin JP, Eliasziw M. A randomized, controlled trial of surgery for temporal-lobe epilepsy. N Engl J Med. 2001;345(5):311-318. PMID: 11484687; DOI: 10.1056/NEJM200108023450501
-
Marson AG, Kadir ZA, Hutton JL, Chadwick DW. The new antiepileptic drugs: a systematic review of their efficacy and tolerability. Epilepsia. 1997;38(8):859-880. PMID: 9579899; DOI: 10.1111/j.1528-1157.1997.tb01461.x
-
Sillanpaa M, Schmidt D. Natural history of treated childhood-onset epilepsy: prospective, long-term population-based study. Brain. 2006;129(Pt 3):617-624. PMID: 16401617; DOI: 10.1093/brain/awh726
-
Nashef L, So EL, Ryvlin P, Tomson T. Unifying the definitions of sudden unexpected death in epilepsy. Epilepsia. 2012;53(2):227-233. PMID: 22168676; DOI: 10.1111/j.1528-1167.2011.03253.x
-
Sveinsson O, Andersson T, Carlsson S, Tomson T. The incidence of SUDEP: A nationwide population-based cohort study. Neurology. 2017;89(2):170-177. PMID: 28341820; DOI: 10.1212/WNL.0000000000004094
-
Harden CL, Meador KJ, Pennell PB, et al. Practice Parameter update: Management issues for women with epilepsy-Focus on pregnancy. Neurology. 2009;73(2):126-132. PMID: 19398681; DOI: 10.1212/WNL.0b013e3181a6b325
-
Tomson T, Battino D, Perucca E. Teratogenicity of antiepileptic drugs. Curr Opin Neurol. 2019;32(2):246-252. PMID: 30664061; DOI: 10.1097/WCO.0000000000000659
-
Vezzani A, French J, Bhajan T, et al. The role of inflammation in epilepsy. Nat Rev Neurol. 2011;7(1):31-40. PMID: 22154545; DOI: 10.1038/nrneurol.2010.178
-
Pitkanen A, Lukasiuk K. Mechanisms of epileptogenesis and potential treatment targets. Lancet Neurol. 2011;10(2):173-186. PMID: 21256455; DOI: 10.1016/S1474-4422(10)70310-0
-
Engel J Jr. The current place of epilepsy surgery. Curr Opin Neurol. 2018;31(2):192-197. PMID: 29240588; DOI: 10.1097/WCO.0000000000000528
-
Bernhardt BC, Bonilha L, Gross DW. Network analysis for a network disorder: The emerging role of graph theory in the study of epilepsy. Epilepsy Behav. 2015;50:162-170. PMID: 26159729; DOI: 10.1016/j.yebeh.2015.06.005
-
Krumholz A, Wiebe S, Gronseth GS, et al. Evidence-based guideline: Management of an unprovoked first seizure in adults. Neurology. 2015;84(16):1705-1713. PMID: 25901058; DOI: 10.1212/WNL.0000000000001487
-
Brodie MJ, Zuberi SM, Scheffer IE, Fisher RS. The 2017 ILAE classification of seizure types and the epilepsies: what do people with epilepsy and their caregivers need to know? Epileptic Disord. 2018;20(2):77-87. PMID: 29620013; DOI: 10.1684/epd.2018.0957
-
Glauser T, Shinnar S, Gloss D, et al. Evidence-Based Guideline: Treatment of Convulsive Status Epilepticus in Children and Adults. Epilepsy Curr. 2016;16(1):48-61. PMID: 26900382; DOI: 10.5698/1535-7597-16.1.48
-
Tomson T, Marson A, Boon P, et al. Valproate in the treatment of epilepsy in girls and women of childbearing potential. Epilepsia. 2015;56(7):1006-1019. PMID: 25912798; DOI: 10.1111/epi.13021
-
Beghi E. The Epidemiology of Epilepsy. Neuroepidemiology. 2020;54(2):185-191. PMID: 31955167; DOI: 10.1159/000503831
Guidelines
-
National Institute for Health and Care Excellence (NICE). Epilepsies in children, young people and adults. [NG217]. 2022. PMID: 35605051
-
DVLA. Assessing fitness to drive: a guide for medical professionals. 2024.
-
Patsalos PN, Berry DJ, Bourgeois BF, et al. Antiepileptic drugs—best practice guidelines for therapeutic drug monitoring: a position paper by the subcommission on therapeutic drug monitoring, ILAE Commission on Therapeutic Strategies. Epilepsia. 2008;49(7):1239-1276. PMID: 18397299; DOI: 10.1111/j.1528-1167.2008.01561.x
-
Kanner AM, Ashman E, Gloss D, et al. Practice guideline update summary: Efficacy and tolerability of the new antiepileptic drugs I: Treatment of new-onset epilepsy. Neurology. 2018;91(2):74-81. PMID: 29898971; DOI: 10.1212/WNL.0000000000005755
-
French JA, Pedley TA. Initial management of epilepsy. N Engl J Med. 2008;359(2):166-176. PMID: 18614784; DOI: 10.1056/NEJMcp0801738
-
Perucca P, Gilliam FG. Adverse effects of antiepileptic drugs. Lancet Neurol. 2012;11(9):792-802. PMID: 22832500; DOI: 10.1016/S1474-4422(12)70153-9
-
Brodie MJ, Barry SJE, Bamagous GA, et al. Patterns of treatment response in newly diagnosed epilepsy. Neurology. 2012;78(20):1548-1554. PMID: 22573629; DOI: 10.1212/WNL.0b013e3182563b19
-
Chen Z, Brodie MJ, Liew D, Kwan P. Treatment outcomes in patients with newly diagnosed epilepsy treated with established and new antiepileptic drugs: A 30-year longitudinal cohort study. JAMA Neurol. 2018;75(3):279-286. PMID: 29340590; DOI: 10.1001/jamaneurol.2017.3949
-
Devinsky O, Vezzani A, O'Brien TJ, et al. Epilepsy. Nat Rev Dis Primers. 2018;4:18024. PMID: 29747395; DOI: 10.1038/nrdp.2018.24
-
Trinka E, Cock H, Hesdorffer D, et al. A definition and classification of status epilepticus - Report of the ILAE Task Force on Classification of Status Epilepticus. Epilepsia. 2015;56(10):1515-1523. PMID: 26336950; DOI: 10.1111/epi.13121
-
Rossetti AO, Logroscino G, Milligan TA, et al. Status Epilepticus Severity Score (STESS): a tool to orient early treatment strategy. J Neurol. 2008;255(10):1561-1566. PMID: 18769858; DOI: 10.1007/s00415-008-0989-1
-
Neligan A, Shorvon SD. Prognostic factors, morbidity and mortality in tonic-clonic status epilepticus: a review. Epilepsy Res. 2011;93(1):1-10. PMID: 21050723; DOI: 10.1016/j.eplepsyres.2010.09.003
-
Ryvlin P, Nashef L, Lhatoo SD, et al. Incidence and mechanisms of cardiorespiratory arrests in epilepsy monitoring units (MORTEMUS): a retrospective study. Lancet Neurol. 2013;12(10):966-977. PMID: 24012372; DOI: 10.1016/S1474-4422(13)70214-X
-
Hesdorffer DC, Tomson T, Benn E, et al. Combined analysis of risk factors for SUDEP. Epilepsia. 2011;52(6):1150-1159. PMID: 21671925; DOI: 10.1111/j.1528-1167.2010.02952.x
-
DeGiorgio CM, Miller P, Meymandi S, et al. RMSSD, a measure of vagus-mediated heart rate variability, is associated with risk factors for SUDEP: the SUDEP-7 Inventory. Epilepsy Behav. 2010;19(1):78-81. PMID: 20620109; DOI: 10.1016/j.yebeh.2010.06.011
-
Lamberts RJ, Thijs RD, Laffan A, et al. Sudden unexpected death in epilepsy: people with nocturnal seizures may be at highest risk. Epilepsia. 2012;53(2):253-257. PMID: 22192074; DOI: 10.1111/j.1528-1167.2011.03360.x
-
Harden CL, Pennell PB, Koppel BS, et al. Practice parameter update: management issues for women with epilepsy—focus on pregnancy (an evidence-based review): vitamin K, folic acid, blood levels, and breastfeeding. Neurology. 2009;73(2):142-149. PMID: 19398685; DOI: 10.1212/WNL.0b013e3181a6b325
-
Weston J, Bromley R, Jackson CF, et al. Monotherapy treatment of epilepsy in pregnancy: congenital malformation outcomes in the child. Cochrane Database Syst Rev. 2016;11(11):CD010224. PMID: 27819746; DOI: 10.1002/14651858.CD010224.pub2
-
Meador KJ, Baker GA, Browning N, et al. Fetal antiepileptic drug exposure and cognitive outcomes at age 6 years (NEAD study): a prospective observational study. Lancet Neurol. 2013;12(3):244-252. PMID: 23352199; DOI: 10.1016/S1474-4422(12)70323-X
-
Campbell E, Kennedy F, Russell A, et al. Malformation risks of antiepileptic drug monotherapies in pregnancy: updated results from the UK and Ireland Epilepsy and Pregnancy Registers. J Neurol Neurosurg Psychiatry. 2014;85(9):1029-1034. PMID: 24444855; DOI: 10.1136/jnnp-2013-306318
Last Updated: 2026-01-10 Citation Count: 83 PubMed-indexed sources with DOIs Line Count: 1,729 lines Topic: 875/1071
Frequently asked questions
Quick clarifications for common clinical and exam-facing questions.
When should I seek emergency care for epilepsy in adults: clinical diagnosis and management?
Seek immediate emergency care if you experience any of the following warning signs: Status Epilepticus (less than 5 mins continuous or no recovery between events), New-onset focal neurological deficit post-ictally, Seizure in the context of pregnancy (Eclampsia), First-ever seizure with fever or meningism, Seizure following head trauma or suspected intracranial hemorrhage, Respiratory compromise or prolonged cyanosis, Ictal asystole or cardiac arrhythmia.