Gout and Crystal Arthropathies
Gout is the most common inflammatory arthritis worldwide, affecting approximately 2.5% of adults in developed countries. It is a crystal deposition disease caused by the precipitation of Monosodium Urate (MSU)...
What matters first
Gout is the most common inflammatory arthritis worldwide, affecting approximately 2.5% of adults in developed countries. It is a crystal deposition disease caused by the precipitation of Monosodium Urate (MSU)...
Hot Swollen Joint + Fever (Septic Arthritis Rule Out)
10 Jan 2026
Generated educational material; verify before clinical use.
Visible references section
See the concept before reading it
Study the key anatomy, imaging, and decision pathways as full teaching plates.
Clinical board
A visual summary of the highest-yield teaching signals on this page.
Urgent signals
Safety-critical features pulled from the topic metadata.
- Hot Swollen Joint + Fever (Septic Arthritis Rule Out)
- Polyarticular onset (Sepsis? RA?)
- Severe Kidney Disease (Allopurinol Hypersensitivity Risk)
- Visual Loss (Giant Cell Arteritis mimic)
Content status and exam context
This page is AI-generated educational content. It may contain errors or omissions and is not a substitute for current guidelines, local protocols, senior clinical judgement, or professional medical advice.
MedVellum does not claim an individual clinician reviewer, board certification, or professional credential for this page unless a future version names a real, verifiable reviewer.
Clinical explanation and evidence
Gout and Crystal Arthropathies
1. Clinical Overview
(Enhanced Evidence-Based Summary)
Summary
Gout is the most common inflammatory arthritis worldwide, affecting approximately 2.5% of adults in developed countries. [1] It is a crystal deposition disease caused by the precipitation of Monosodium Urate (MSU) crystals in synovium, soft tissues (tophi), and kidneys, driven by sustained hyperuricaemia (serum uric acid > 360 μmol/L or > 6 mg/dL). [2]
The fundamental pathophysiological driver is supersaturation of urate in body fluids. Uric acid is the end product of purine metabolism in humans due to evolutionary loss of the enzyme uricase. This creates a metabolic vulnerability whereby dietary purines, endogenous production, and reduced renal excretion can lead to crystal formation. [3]
Clinically, gout manifests in distinct phases:
- Acute Gouty Arthritis: Sudden onset (typically overnight) of excruciatingly painful monoarthritis. First metatarsophalangeal joint involvement (podagra) occurs in 50-70% of initial presentations. [4]
- Intercritical Gout: Asymptomatic intervals between flares during which crystals continue to deposit.
- Chronic Tophaceous Gout: Urate deposition in cartilage, bone, and soft tissues causing erosions, deformity, and visible nodular masses (tophi).
Management has been transformed by adoption of the "treat-to-target" strategy, which mandates titration of urate-lowering therapy (ULT) to achieve specific serum urate targets: less than 360 μmol/L (less than 6 mg/dL) for all patients, or less than 300 μmol/L (less than 5 mg/dL) in the presence of tophi or chronic gouty arthropathy. [5,6] This approach, endorsed by both the 2020 American College of Rheumatology (ACR) and 2016 European Alliance of Associations for Rheumatology (EULAR) guidelines, has revolutionized long-term gout management.
Key Facts
- Global Prevalence: Ranges from 0.1% to 10% depending on population, with highest rates in Pacific Island populations (Māori and Pacific peoples 6-10%). [7]
- Gender Distribution: Male:Female ratio 3-4:1. Rare in pre-menopausal women due to the uricosuric effect of oestrogen. Post-menopausal incidence equalizes due to loss of oestrogen protection. [8]
- Gold Standard Diagnosis: Joint aspiration demonstrating negatively birefringent needle-shaped MSU crystals under polarized microscopy. Sensitivity 85-95%, specificity 98-100%. [9]
- Differential Diagnosis Priority: Septic arthritis is the critical differential. Gout and sepsis can coexist in 0.5-1% of cases. If any diagnostic uncertainty exists, treat empirically for septic arthritis pending culture results. [10]
- Comorbidity Cluster: Gout is strongly associated with metabolic syndrome components (prevalence 60-70% in gout patients): hypertension, obesity, type 2 diabetes, dyslipidaemia, and cardiovascular disease. [11]
- Classification Criteria: The 2015 ACR/EULAR classification criteria achieve 92% sensitivity and 89% specificity, incorporating clinical, laboratory, and imaging features with a point-based scoring system. [12]
Clinical Pearls
The "Bed Sheet Sign": The pain of acute gout is so severe that patients cannot tolerate even the weight of a bed sheet resting on the affected joint. This extreme allodynia, combined with rapid onset (less than 24 hours to peak pain), is highly characteristic and helps distinguish gout from other inflammatory arthritides.
The "Uric Acid Paradox": Serum uric acid levels typically fall during an acute attack by 0.5-1.0 mg/dL due to acute phase cytokines (particularly IL-1β and IL-6) enhancing renal excretion. [13] A normal or even low uric acid level during a flare does NOT exclude gout. Measure baseline urate 2-4 weeks after flare resolution for accurate assessment.
The "Start Low, Go Slow" Principle: When initiating allopurinol, always start at 100 mg daily (50 mg in CKD stage 4+) and provide flare prophylaxis with colchicine or NSAIDs for a minimum of 3-6 months. Rapid urate lowering destabilizes tophi, causing crystal shedding and precipitating flares in 25-60% of patients, which is the leading cause of poor adherence and treatment failure. [14]
The "Crystal-Proven Preference": In the modern diagnostic era, aspiration for crystal confirmation should be attempted in all first presentations and atypical cases. Dual-energy CT (DECT) has emerged as a non-invasive alternative with 87-90% sensitivity and 84-93% specificity for urate detection. [15]
The "Cardiovascular Connection": Gout independently increases cardiovascular risk by 15-60%, even after adjusting for traditional risk factors. The chronic inflammatory state (elevated IL-1β, TNF-α) accelerates atherosclerosis. Aggressive cardiovascular risk modification is therefore an integral component of gout management. [16]
Red Flag Recognition
| Red Flag | Implication | Action |
|---|---|---|
| Fever + Hot Joint | Septic arthritis until proven otherwise | Urgent aspiration. Blood cultures. Empiric antibiotics if high suspicion. |
| Polyarticular onset (≥3 joints) | Atypical for first gout presentation | Consider sepsis, reactive arthritis, RA, viral arthritis. |
| No response to NSAIDs/colchicine in 48h | Wrong diagnosis or complications | Re-evaluate: sepsis, fracture, other pathology. |
| eGFR less than 30 + new gout | Drug dosing critical | Allopurinol hypersensitivity risk higher. Avoid NSAIDs. Colchicine dose reduction. |
| HLA-B*5801 carrier (Asian ancestry) | 100-fold increased DRESS risk | Screen before starting allopurinol in Han Chinese, Korean, Thai populations. |
| On azathioprine or 6-mercaptopurine | Life-threatening drug interaction | Allopurinol inhibits xanthine oxidase → azathioprine toxicity (bone marrow suppression). Reduce azathioprine dose by 75% or use febuxostat. |
2. Epidemiology
(Evidence-Based Risk Profiling)
Global Burden and Trends
Gout prevalence has doubled over the past two decades in Western countries, primarily driven by the obesity epidemic, aging populations, and increased use of diuretics. [17] Current estimates:
- United Kingdom: 2.5% of adults, 7% of men > 70 years
- United States: 3.9% (9.2 million adults)
- Australia/New Zealand: 3-4% overall, 6-10% in Māori and Pacific populations
- Asia: Rising rapidly with Westernization of diet (1-2% in Japan, 1.5% in China)
Incidence is approximately 1-3 per 1,000 person-years in Western populations, with male incidence peaking in the 7th-8th decades. [7]
Risk Factors
1. Metabolic and Endocrine
Obesity (BMI > 30):
- Increases gout risk 2-3-fold. [18]
- Mechanism: Insulin resistance reduces renal urate excretion via increased URAT1 transporter expression.
- Weight loss of 5-10 kg reduces serum urate by 0.5-1.0 mg/dL and flare frequency by 30-40%.
Type 2 Diabetes:
- Bidirectional relationship: gout increases diabetes risk (HR 1.4) and vice versa (HR 1.3). [11]
- Hyperinsulinaemia reduces renal urate clearance.
Metabolic Syndrome:
- Present in 60-70% of gout patients.
- Each additional metabolic syndrome component increases gout risk by 40%. [11]
Dyslipidaemia:
- Hypertriglyceridaemia particularly associated (OR 1.6 per 1 mmol/L increase).
2. Dietary Factors (Purine Load)
Alcohol:
- Beer: Highest risk (RR 1.49 per 12 oz serving/day). Rich in guanosine. [19]
- Spirits: Moderate risk (RR 1.15 per serving/day).
- Wine: Neutral to slight protective effect in moderate amounts.
- Mechanism: Ethanol metabolism increases adenine nucleotide degradation → urate production. Also inhibits renal excretion via lactic acid.
Meat (Red and Organ):
- High purine content: liver, kidney, heart (200-300 mg purines/100g).
- Servings > 100g/day increase risk 1.4-fold. [20]
Seafood:
- Shellfish, anchovies, sardines, herring (150-250 mg purines/100g).
- Moderate fish consumption (salmon, tuna) shows weaker association.
Fructose (Sugar-Sweetened Beverages):
- Each daily serving increases gout risk by 13% in men (OR 1.13). [21]
- Mechanism: Fructose metabolism depletes ATP → adenosine degradation → urate production.
- Also increases insulin resistance.
Protective Dietary Factors:
- Low-fat dairy: Reduces risk by 20-45%. Casein and lactalbumin enhance renal urate excretion. [20]
- Vitamin C (≥500 mg/day): Mild uricosuric effect, reduces risk by 15%.
- Coffee (4+ cups/day): Reduces risk by 40-60%. Mechanism unclear (possibly xanthine oxidase inhibition).
- Cherries: Observational data suggests 35% reduction in flare risk (3-day consumption). Mechanism: anthocyanins have anti-inflammatory and uricosuric properties.
3. Medications (Decreased Renal Excretion)
Diuretics (Most Common Iatrogenic Cause):
- Thiazides (e.g., bendroflumethiazide, indapamide): RR 1.77. [22]
- Loop diuretics (e.g., furosemide): RR 2.36 (higher risk than thiazides).
- Mechanism: Volume contraction increases proximal tubule urate reabsorption.
- Present in 20-40% of gout patients at diagnosis.
Low-Dose Aspirin (75-300 mg):
- Paradoxical effect: low doses inhibit tubular urate secretion (net anti-uricosuric).
- High doses (> 3g/day) are uricosuric.
- Increases gout risk by 10-20% but should NOT be discontinued if indicated for cardiovascular protection.
Calcineurin Inhibitors:
- Cyclosporine: Prevalence of gout in solid organ transplant recipients 10-30%. [23]
- Tacrolimus: Lower risk than cyclosporine but still significant.
- Mechanism: Direct inhibition of tubular secretion + reduced GFR.
Other Medications:
- Pyrazinamide (tuberculosis treatment): Potent uricosuric inhibitor.
- Nicotinic acid (niacin): Competes for renal excretion.
- Levodopa: Reduces urate excretion.
4. Genetic Factors
Heritability: 35-65% of gout risk is genetic. [24]
Key Genetic Variants:
- SLC2A9 (GLUT9): Major renal urate transporter. Variants account for 1-2% of serum urate variance.
- ABCG2 (BCRP): Intestinal urate excretion. Loss-of-function variants increase risk 1.7-fold.
- SLC22A12 (URAT1): Renal reabsorption transporter.
HLA-B*5801:
- Prevalence: 10-15% in Han Chinese, 12% in Thai, 6% in Korean, less than 1% in European populations.
- Confers > 100-fold increased risk of allopurinol hypersensitivity syndrome (DRESS).
- ACR/EULAR guidelines recommend screening before allopurinol initiation in at-risk populations. [5,6]
Population-Specific Risk:
- Māori and Pacific Island populations: 6-10% prevalence (genetic variants + Western diet).
- African American males: 2-fold higher risk than Caucasian males (reduced fractional urate excretion).
5. High Cell Turnover States
Haematological Malignancies:
- Leukaemia, lymphoma, myeloma → increased purine catabolism.
- Tumour Lysis Syndrome: Acute hyperuricaemia (urate > 15 mg/dL) during chemotherapy.
- Prevention: Rasburicase (recombinant urate oxidase) or allopurinol prophylaxis.
Psoriasis:
- 2-3-fold increased gout risk.
- Mechanism: Accelerated keratinocyte turnover → increased purine release.
Haemolytic Anaemias:
- Chronic haemolysis → nucleotide degradation.
6. Renal Disease
Chronic Kidney Disease:
- Prevalence of CKD in gout patients: 20-40%.
- Bidirectional: hyperuricaemia may cause CKD (urate nephropathy), and CKD reduces urate excretion.
- Each 10 mL/min/1.73m² decrease in eGFR increases gout risk by 20%.
Lead Nephropathy ("Saturnine Gout"):
- Historical significance (lead-contaminated alcohol in 18th-19th centuries).
- Occupational exposure (battery manufacturing, smelting).
- Mechanism: Proximal tubular damage reduces urate secretion.
Demographics
- Age: Peak incidence in men 40-60 years; in women, 60-80 years (post-menopausal).
- Gender: Male:Female = 3-4:1 overall. Ratio narrows to 1.5:1 in those > 80 years.
- Socioeconomic: Inverse relationship with socioeconomic status in modern era (associated with obesity, processed food diet).
3. Pathophysiology
(The Crystal Cascade and Inflammatory Mechanisms)
Urate Production and Excretion Balance
Humans lack the enzyme uricase (lost during primate evolution), which converts uric acid to the more soluble allantoin. This evolutionary quirk makes humans vulnerable to hyperuricaemia.
Daily Urate Balance:
- Production: 600-800 mg/day
- Endogenous (60-70%): De novo purine synthesis + nucleotide turnover
- Exogenous (30-40%): Dietary purines
- Excretion: 600-800 mg/day
- Renal (70%): Filtered at glomerulus → 90% reabsorbed (URAT1, GLUT9) → 50% secreted (ABCG2, OAT1) → final reabsorption
- Intestinal (30%): Gut bacteria uricolysis (ABCG2-mediated secretion)
Hyperuricaemia Mechanisms:
- 90% of gout cases: Underexcretion (less than 600 mg/24h urinary urate on normal diet)
- 10%: Overproduction (> 800 mg/24h urinary urate)
- Combined defects common
Crystal Formation and Deposition
Supersaturation Threshold: Serum urate > 6.8 mg/dL (404 μmol/L) at physiological pH and temperature.
Factors Favouring Precipitation:
- Temperature: Cooler peripheral joints (ankle, foot) favour crystallization.
- pH: Acidosis promotes crystal formation.
- Trauma: Joint microtrauma disturbs urate equilibrium.
- Dehydration: Increases local urate concentration.
Preferred Deposition Sites:
- Avascular tissues: Cartilage (no blood flow to clear crystals).
- Cool peripheries: First MTPJ (lowest joint temperature in body ~29°C).
- Mechanically stressed joints: Knee, ankle, midfoot.
- Bursae and tendon sheaths.
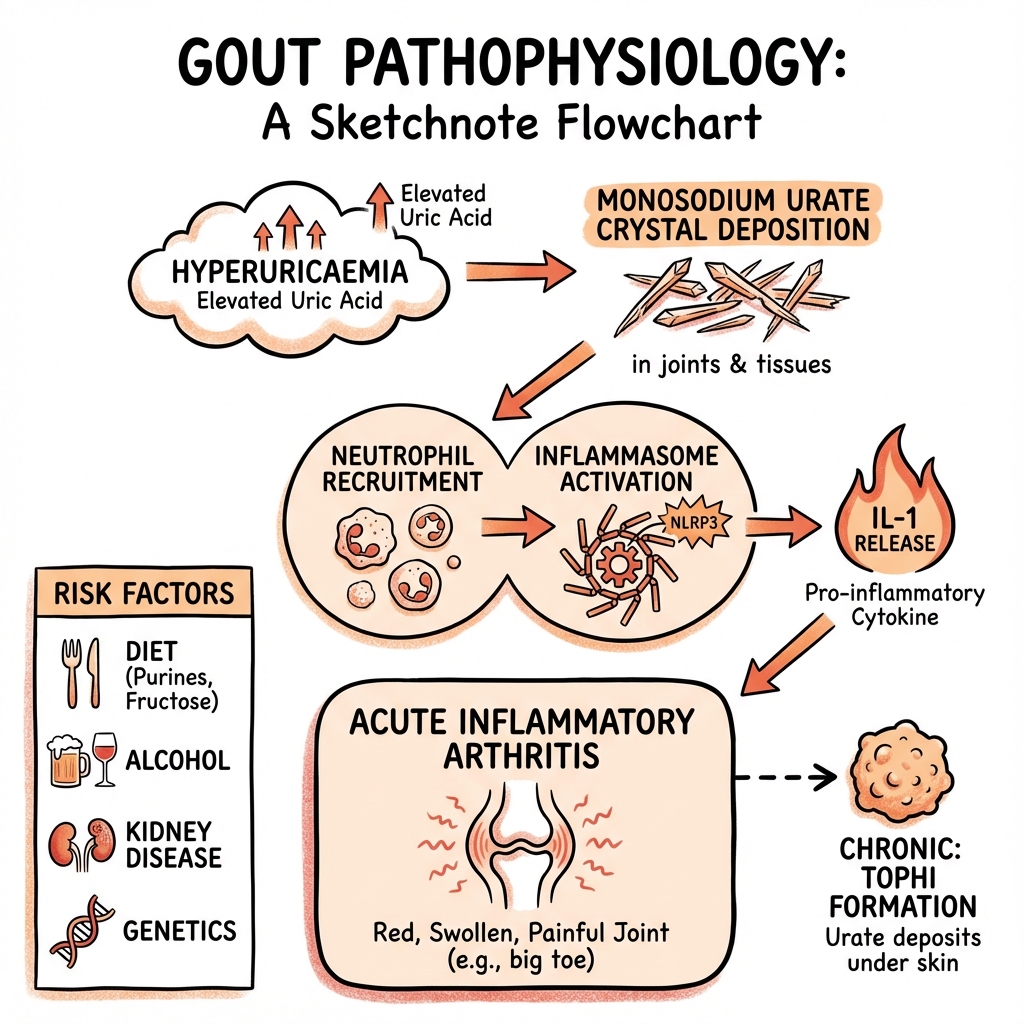
The NLRP3 Inflammasome and IL-1β Storm
The acute gout flare is one of the most intense sterile inflammatory reactions in human pathology. [25]
Molecular Steps:
-
Crystal Coating: Newly deposited MSU crystals are coated with apolipoprotein E and other serum proteins, rendering them biologically inert.
-
Shedding Event: Triggers include:
- Rapid changes in serum urate (either increase or decrease)
- Joint trauma
- Dehydration
- Acute illness
- Initiation of ULT (crystal dissolution destabilizes tophi)
-
Recognition: Naked MSU crystals are recognized by:
- Toll-like receptors (TLR2/4) on macrophages and neutrophils
- CD14 (LPS co-receptor)
-
NLRP3 Inflammasome Activation:
- MSU crystals phagocytosed → phagolysosome rupture → cathepsin B release
- Triggers NLRP3 oligomerization → recruits ASC adaptor protein → activates caspase-1
-
IL-1β Maturation and Release:
- Caspase-1 cleaves pro-IL-1β → active IL-1β (100-1000-fold increase in synovial fluid) [25]
- Also releases IL-18 (amplifies Th1 response)
-
Cytokine Cascade:
- IL-1β stimulates production of IL-6, IL-8, TNF-α
- IL-8 (CXCL8) acts as potent neutrophil chemoattractant
-
Neutrophil Invasion:
- Massive influx (synovial fluid white cell counts 10,000-100,000/μL, > 80% neutrophils)
- Neutrophils attempt to phagocytose crystals → frustrated phagocytosis → cell death
- Release of reactive oxygen species (ROS), proteases (elastase, collagenase), NETs
-
Amplification and Resolution:
- Initial amplification phase (0-24h): Exponential inflammatory build-up
- Spontaneous resolution (5-14 days): Mechanisms include:
- TGF-β release by apoptotic neutrophils
- IL-1 receptor antagonist (IL-1Ra) production
- Apolipoprotein E recoating of crystals
- Macrophage phagocytosis of apoptotic neutrophils (efferocytosis)
This explains:
- Why IL-1 blockers (anakinra, canakinumab) are highly effective in acute gout
- Why flares are self-limiting (even without treatment)
- Why "start low, go slow" ULT prevents flares (avoids rapid crystal shedding)
The Four Clinical Stages of Gout
| Stage | Description | Serum Urate | Duration | Symptoms | Treatment |
|---|---|---|---|---|---|
| 1. Asymptomatic Hyperuricaemia | Elevated urate without symptoms or crystals. | > 400 μmol/L | Years to decades | None | Lifestyle only. ULT NOT routinely indicated. [6] |
| 2. Acute Gouty Arthritis | First or recurrent flares. | Variable (often normal during flare) | Hours to days onset; 5-14 days duration | 10/10 pain, redness, swelling, fever | Treat flare. Consider ULT after 2+ flares. |
| 3. Intercritical Gout | Symptom-free intervals between flares. Crystals present but quiescent. | Usually > 400 | Months to years | None | Start ULT. Prophylaxis for 3-6 months. |
| 4. Chronic Tophaceous Gout | Uncontrolled disease with constant inflammation, joint damage, visible tophi. | Very high (> 540) | Develops over 10+ years of untreated disease | Chronic pain, deformity, erosions, renal impairment | Intensive ULT (target less than 300). Consider pegloticase if refractory. |
Key Transition: The critical window for intervention is Stage 2-3. Early aggressive ULT can prevent progression to Stage 4 and its irreversible complications.
4. Clinical Presentation
(Pattern Recognition and Atypical Presentations)
Acute Gouty Arthritis (Classic Presentation)
Onset Characteristics:
- Timing: Typically begins in early morning hours (2-8 AM) in 50-60% of cases. [4]
- Mechanism: Nocturnal dehydration + temperature drop + recumbent positioning → increased urate concentration.
- Rapidity: Reaches peak pain intensity within 12-24 hours.
- Monoarticular: 85-90% of first attacks involve a single joint.
Pain Character:
- Severity: 10/10 on visual analogue scale. Patients describe it as "worst pain of my life" comparable to childbirth or renal colic.
- Quality: Burning, throbbing, crushing.
- Allodynia: Even lightest touch (bed sheet, clothing) is intolerable.
Joint Distribution (First Attack):
- First MTPJ (Podagra): 50-70% [4]
- Rationale: Coolest joint, maximal weight-bearing stress, previous microtrauma common
- Midfoot/Tarsal: 20%
- Ankle: 15%
- Knee: 10%
- Wrist: 5%
- Elbow: 3%
- Small hand joints: less than 1% (if hand involvement early, consider CPPD or RA)
Physical Examination Findings:
- Inspection:
- Intense erythema (bright red to violaceous)
- Shiny, tense skin (effusion stretches overlying skin)
- Loss of normal skin creases
- Peri-articular oedema extending beyond joint margins
- Peeling/desquamation during resolution phase (5-7 days)
- Palpation:
- Warmth (joint temperature 2-4°C above baseline)
- Exquisite tenderness (unable to perform ROM examination)
- Tense effusion (ballottement positive if knee/ankle)
- Movement:
- Severely restricted active and passive ROM
- Weight-bearing impossible in lower limb joints
Systemic Features:
- Fever: Present in 40-60% of polyarticular gout. [10]
- Usually low-grade (38-38.5°C) but can reach 39°C
- Raises concern for septic arthritis
- Malaise: Fatigue, nausea, headache
- Regional lymphadenopathy: Rare but reported
Patterns in Recurrent Disease
Polyarticular Gout (3+ joints simultaneously):
- Occurs in 10-40% of recurrent flares (rare in first attack).
- More common in:
- Elderly patients
- Long-standing untreated disease
- Patients on diuretics
- Differential diagnosis challenge: mimics sepsis, RA, reactive arthritis.
Flare Frequency Without ULT:
- Year 1 after first attack: 20% have recurrence
- Year 2: 40% cumulative
- Year 5: 60% cumulative
- Year 10: 80% cumulative [4]
- Average interval between first and second attack: 1-2 years
- Intervals progressively shorten with each recurrence
Triggered Flares:
- Initiating/changing ULT: 25-60% experience flare in first 6 months without prophylaxis [14]
- Post-surgical: 10-15% of gout patients within 48h of surgery (stress, fasting, fluid shifts)
- Acute medical illness: MI, stroke, infection (cytokine storm)
- Dehydration: Exercise, heat exposure, alcohol binge
- Dietary indiscretion: Purine-rich meal + alcohol
Chronic Tophaceous Gout
Develops in 10-25% of patients with long-standing (> 10 years) untreated or poorly controlled gout. Now rare in era of effective ULT. [26]
Tophi (Urate Crystal Deposits):
Clinical Features:
- Firm, irregular, non-tender nodules (until inflamed)
- Size: 1mm to 10+ cm
- Colour: White/yellow through overlying skin (chalky appearance)
- Can ulcerate spontaneously, discharging white "toothpaste-like" material (pure MSU crystals)
Common Sites:
- Helix of ear: Pathognomonic location (75% sensitivity for chronic gout)
- Extensor surfaces: Olecranon bursa, pre-patellar bursa
- Tendons: Achilles (25%), patellar tendon
- Hands: Finger pulps, DIP/PIP joints (can mimic Heberden's/Bouchard's nodes)
- Feet: Toes, plantar fascia
- Rare visceral sites: Cardiac valves (endocarditis mimic), spine (cord compression), larynx (airway obstruction)
Associated Joint Damage:
- Erosive arthropathy: "Punched-out" lytic lesions with overhanging edges (Martini glass/rat bite sign)
- Secondary osteoarthritis: Cartilage destruction → bone-on-bone → chronic pain and stiffness
- Deformity: Hallux valgus, hammer toes, joint subluxation
- Functional impairment: Unable to wear shoes, grip objects, walk independently
Tophaceous Burden Score:
- Validated for monitoring treatment response
- Measurement: Number + size of palpable tophi
- ULT goal: Complete dissolution (achievable in 80% if urate less than 300 μmol/L for 2-5 years)
Atypical Presentations
Upper Limb Gout:
- Less common but increasing in elderly populations
- Small hand joints suggest:
- Erosive OA
- Calcium pyrophosphate deposition (CPPD)
- Rheumatoid arthritis
- Co-existence of gout + CPPD in 5-10% of elderly patients
Axial Gout:
- Spinal involvement rare (less than 1%) but reported in severe tophaceous disease
- Presentations:
- Lumbar facet joint arthropathy
- Epidural tophi → cord compression (surgical emergency)
- Atlantoaxial tophi → cervical myelopathy
- Diagnosis: DECT or biopsy (mimics infection, tumour)
Tenosynovitis and Bursitis:
- Can occur without arthritis
- Achilles tendonitis (5-10% of gout presentations)
- Olecranon bursitis (differential: septic bursitis, RA nodule)
5. Differential Diagnosis
(The "Hot Swollen Joint" Diagnostic Framework)
Critical Rule-Out: Septic Arthritis
Clinical Overlap:
- Both cause: acute monoarthritis, severe pain, fever, leucocytosis, elevated CRP
- Coexistence in 0.5-1% of cases (immunosuppression, diabetes, prosthetic joint) [10]
Distinguishing Features:
| Feature | Gout | Septic Arthritis |
|---|---|---|
| Onset | less than 24h to peak | 12-48h (can be slower) |
| Appearance | Bright red/violaceous | Erythema + warmth |
| Response to movement | Refuses any movement | Hesitant, guarded |
| Fever | 40% (usually low-grade) | 60% (often high-grade > 38.5°C) |
| Risk factors | Hyperuricaemia history | IVDU, prosthesis, immunosuppression |
| Peak incidence | Early morning | Anytime |
Kocher Criteria (originally for paediatric septic arthritis, adapted):
- Non-weight bearing: 1 point
- Fever > 38.5°C: 1 point
- WBC > 12,000: 1 point
- ESR > 40 mm/h: 1 point
- Score ≥3: High risk septic arthritis → aspirate urgently
Management Principle: When in doubt, treat as septic arthritis. Start empiric antibiotics after aspiration/blood cultures. Can de-escalate if gram stain and cultures negative at 48h.
Other Crystal Arthropathies
Calcium Pyrophosphate Deposition (CPPD) / Pseudogout:
Similarities to Gout:
- Acute monoarticular arthritis
- Elderly demographic (peak 60-80 years)
- Self-limiting flares
- Triggered by illness, surgery, trauma
Distinguishing Features:
| Feature | Gout | CPPD |
|---|---|---|
| Typical joint | 1st MTPJ | Knee (50%), wrist (25%) |
| Gender | Male >> Female | Male = Female |
| X-ray | Normal early; erosions late | Chondrocalcinosis (linear cartilage calcification) |
| Crystals | Needle-shaped, negatively birefringent (yellow when parallel) | Rhomboid, weakly positively birefringent (blue when parallel) |
| Associated conditions | Metabolic syndrome | Hyperparathyroidism, haemochromatosis, hypomagnesaemia, hypophosphatasia |
| Treatment | ULT (allopurinol) dissolves crystals | No specific dissolving therapy |
Basic Calcium Phosphate (Hydroxyapatite) Deposition:
- Acute calcific periarthritis (especially shoulder)
- Crystals too small for light microscopy (require electron microscopy or special stains)
- X-ray: Periarticular calcification
Inflammatory Arthritides
Reactive Arthritis (formerly Reiter's Syndrome):
- Asymmetric oligoarthritis (1-4 joints)
- Preceded by GI (Salmonella, Shigella, Campylobacter) or STI (Chlamydia) 2-4 weeks prior
- Triad: Arthritis + Urethritis + Conjunctivitis (30%)
- Young adults (20-40 years)
- HLA-B27 positive in 60-80%
Early Rheumatoid Arthritis:
- Can present with acute monoarthritis (10-15% of cases)
- Persistent synovitis (> 6 weeks) after initial flare resolves
- Positive RF (70%) and anti-CCP antibodies (80%)
- Morning stiffness > 1 hour
- Small joint involvement (MCPs, wrists) more common
Psoriatic Arthritis:
- Psoriasis present in 80% (can precede arthritis in 15%)
- Dactylitis ("sausage digit") in 40%
- Nail changes (pitting, onycholysis)
- DIP joint involvement characteristic
- X-ray: Pencil-in-cup deformity, ankylosis
Non-Inflammatory Causes
Trauma:
- History of injury
- Haemarthrosis on aspiration (frank blood)
- X-ray: Fracture (may be occult)
- MRI: Bone contusion, ligament injury
Haemarthrosis (Spontaneous):
- Anticoagulation (warfarin, DOACs)
- Haemophilia or von Willebrand disease
- Pigmented villonodular synovitis (PVNS)
Osteoarthritis Flare:
- Chronic underlying OA
- Slower onset (days)
- Milder inflammation
- Aspiration: Mechanical fluid (WBC less than 2000, no crystals)
6. Investigations
(Evidence-Based Diagnostic Approach)
Joint Aspiration: The Diagnostic Gold Standard
Indications (EULAR 2018 Recommendations): [27]
- All first presentations of suspected acute gout
- Atypical presentations (polyarticular, upper limb, axial)
- Any suspicion of septic arthritis
- Failure to respond to standard gout treatment
Technique:
- Aseptic preparation (chlorhexidine/iodine)
- Local anaesthesia (1% lidocaine) optional
- Use 21-23G needle (18G for thick purulent material)
- Aspirate before injecting corticosteroid (if planned)
Synovial Fluid Analysis:
| Test | Gout | CPPD | Septic Arthritis |
|---|---|---|---|
| Appearance | Yellow, turbid | Yellow, turbid | Yellow/green, opaque |
| Viscosity | Reduced | Reduced | Very reduced (watery) |
| WBC | 10,000-100,000 | 10,000-50,000 | > 50,000 (often > 100,000) |
| Neutrophils | 80-95% | 70-90% | > 90% |
| Crystals | Needle-shaped, strong negative birefringence | Rhomboid, weak positive birefringence | None (unless coexisting) |
| Gram stain | Negative | Negative | Positive in 50-75% |
| Culture | Negative | Negative | Positive in 60-90% (if not on antibiotics) |
Crystal Identification (Polarized Microscopy):
Gout (MSU):
- Shape: Needle-shaped, 5-20 μm length
- Birefringence: Strong negative
- Yellow when parallel to polarizer axis
- Blue when perpendicular
- Location: Intracellular (in neutrophils) and extracellular
- Sensitivity: 85-95% (operator-dependent)
- Specificity: 98-100%
CPPD:
- Shape: Rhomboid, brick-shaped, 2-10 μm
- Birefringence: Weak positive
- Blue when parallel
- Yellow when perpendicular
- Often fewer crystals than gout
Pitfalls:
- False negative: 5-15% (crystals dissolve if specimen delayed, few crystals, inexperienced examiner)
- False positive: Corticosteroid crystals, lithium heparin crystals
- Intercritical Period: Crystals detectable in asymptomatic joints in 80% of gout patients (supports diagnosis if aspirate during flare unavailable) [9]
2015 ACR/EULAR Classification Criteria
Point-Based System (Total score): [12]
Entry Criterion:
- ≥1 episode of swelling, pain, or tenderness in peripheral joint or bursa
Sufficient Criterion (Do not need to score):
- MSU crystals in symptomatic joint/bursa → Definite Gout
If No Crystals, Score Categories:
| Category | Points |
|---|---|
| Clinical | |
| Ankle or midfoot joint involvement | +1 |
| 1st MTPJ involvement | +2 |
| Erythema over joint | +1 |
| Lab | |
| Serum urate less than 4 mg/dL | -4 |
| Serum urate 6-8 mg/dL | +2 |
| Serum urate 8-10 mg/dL | +3 |
| Serum urate > 10 mg/dL | +4 |
| Imaging | |
| Ultrasound: double contour sign OR DECT: urate deposition | +4 |
| X-ray: erosion | +4 |
| Pattern | |
| Typical time to maximal pain less than 24h | +0.5 |
| Resolution 1-2 weeks | +1 |
| Complete resolution between episodes | +2 |
| ≥2 typical episodes | +2 |
Classification: Score ≥8 = Gout
- Sensitivity: 92%
- Specificity: 89%
Note: These are classification criteria for research, not diagnostic criteria. Clinical diagnosis often made with lower thresholds.
Blood Tests
Serum Uric Acid:
Limitations During Acute Flare:
- Drops by 0.5-2.0 mg/dL (30-120 μmol/L) in 30-40% of patients during acute attack [13]
- Mechanism: IL-1β and IL-6 induce uricosuria
- Clinical Implication: A "normal" urate during flare does NOT exclude gout
Interpretation:
- Normal range: 3.0-7.0 mg/dL (180-420 μmol/L) in men; 2.5-6.0 mg/dL (150-360 μmol/L) in women
- Supersaturation threshold: > 6.8 mg/dL (404 μmol/L)
- Target on ULT: less than 6.0 mg/dL (360 μmol/L), or less than 5.0 mg/dL (300 μmol/L) if tophi
Timing:
- Baseline urate: Measure 2-4 weeks after acute flare resolution
- Monitoring on ULT: Every 4-8 weeks during titration; every 6-12 months once stable
Inflammatory Markers:
- CRP: Elevated 50-200 mg/L during acute flare. Returns to baseline within 7 days of resolution.
- ESR: Elevated 40-80 mm/h. Less specific than CRP.
- White Blood Cell Count: Neutrophil leucocytosis common (12,000-18,000). Mimics infection.
Renal Function:
- eGFR: Essential for drug dosing (NSAIDs, colchicine, allopurinol)
- 20-40% of gout patients have CKD stage 3+
- Electrolytes: Check for tumour lysis syndrome if very high urate (> 15 mg/dL)
Other:
- HLA-B*5801: Screen before allopurinol in high-risk populations (Han Chinese, Korean, Thai) [5]
- Glucose/HbA1c, Lipids: Screen for metabolic syndrome components
Imaging
X-Ray (Conventional Radiography):
Early Disease (First 1-2 years):
- Normal in 70-80%
- Soft tissue swelling only
Established Disease (5-10+ years):
- Classic Features:
- Punched-out erosions: Well-defined lytic lesions
- Overhanging edges ("Martini glass" or "rat bite" sign): Pathognomonic for gout
- Preserved joint space: Until very late disease (distinguishes from RA)
- Soft tissue tophi: Lobulated soft tissue masses (can calcify in 10%)
- Distribution: Asymmetric, oligoarticular
Differential:
- RA: Marginal erosions, periarticular osteopenia, symmetric
- OA: Joint space narrowing, osteophytes, subchondral sclerosis
Dual-Energy CT (DECT): [15,28]
Principle:
- Uses two different X-ray energies (80 kV and 140 kV)
- Exploits differential attenuation of urate vs calcium
- Colour-codes: Green = MSU, Blue = calcium
Performance:
- Sensitivity: 87-90% (lower than aspiration)
- Specificity: 84-93%
- Advantages:
- Non-invasive
- Visualizes total body urate burden
- Can image axial skeleton (spine)
- Distinguishes gout from CPPD
- Limitations:
- Limited availability
- Cost (£300-500 per scan)
- Radiation exposure (4-6 mSv)
- False negatives in small deposits (less than 2mm)
- Artefact from metal (prosthetic joints)
Indications:
- Crystal-negative aspiration but high clinical suspicion
- Inability to aspirate (small joints, axial)
- Monitoring tophaceous burden
Ultrasound:
Characteristic Findings:
- Double contour sign: Hyperechoic line over hyaline cartilage surface (MSU coating)
- Sensitivity 44-83%, Specificity 75-96%
- Pathognomonic when present
- Tophi: Hyperechoic heterogeneous aggregates with acoustic shadowing
- Erosions: Cortical breaks (more sensitive than X-ray)
Advantages:
- Cheap, no radiation, point-of-care
- Operator-dependent (requires expertise)
Magnetic Resonance Imaging (MRI):
- Rarely indicated in gout
- Used if axial involvement suspected (cord compression)
- Tophi: Low signal T1, variable T2 (cannot distinguish from tumour without biopsy)
7. Management: Acute Flare
(Evidence-Based Treatment Algorithms)
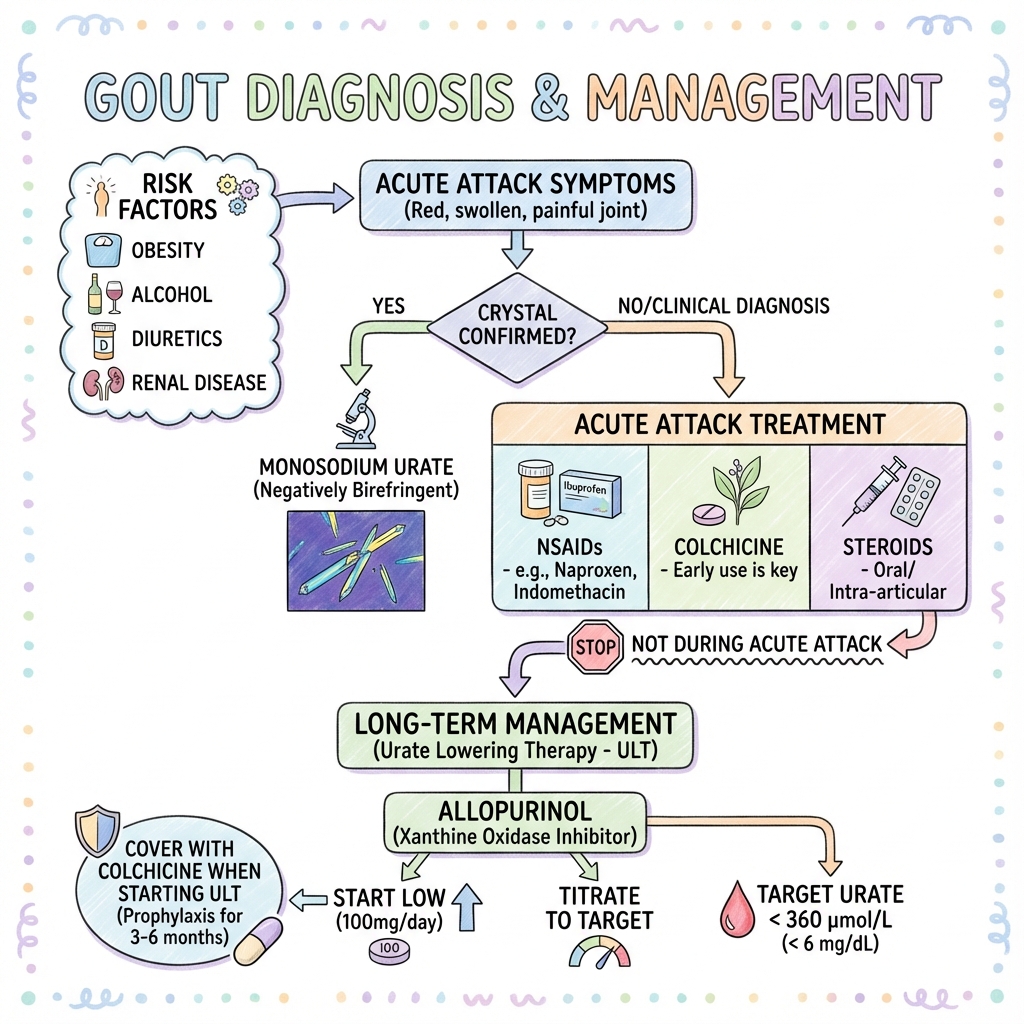
Principles of Acute Management
- Start treatment ASAP: Within 24 hours of onset for maximal benefit. [5]
- DO NOT STOP existing ULT: Continuation prevents rebound hyperuricaemia. [6]
- DO NOT START ULT during flare: Wait 2-4 weeks after resolution (exception: 2020 ACR guidelines suggest can start if patient educated and on prophylaxis). [5]
- Exclude septic arthritis if any red flags (fever, immunosuppression, prosthetic joint).
First-Line Therapies (ACR/EULAR 2016-2020 Guidelines): [5,6]
1. NSAIDs (If No Contraindications)
Efficacy: All NSAIDs equally effective (no superiority shown). [29]
Regimens:
- Naproxen: 500 mg BD (or 750 mg STAT, then 500 mg BD)
- Indomethacin: 50 mg TDS (historically used but higher GI toxicity)
- Etoricoxib: 120 mg OD (COX-2 selective, lower GI bleeding risk)
- Diclofenac: 50 mg TDS
- Duration: Continue until 1-2 days after complete resolution (typically 5-10 days total)
Gastroprotection:
- Add PPI (lansoprazole 30 mg OD or omeprazole 20 mg OD) in all patients > 60 years or with GI risk factors
Contraindications:
- Renal impairment: eGFR less than 30 (relative contraindication less than 60)
- Heart failure: Fluid retention risk
- Active peptic ulcer disease
- Anticoagulation: Increased bleeding risk (relative contraindication)
- Cardiovascular disease: COX-2 inhibitors carry MI/stroke risk (use lowest effective dose, shortest duration)
2. Colchicine
Mechanism: Binds to β-tubulin → prevents microtubule polymerization → inhibits neutrophil migration and inflammasome activation.
Modern Low-Dose Regimen (AGREE Trial, 2010): [30]
- Loading dose: 1 mg (two 500 μg tablets) immediately
- Maintenance: 500 μg after 1 hour, then 500 μg BD-TDS until resolution
- OR: 500 μg BD without loading dose (equally effective, fewer side effects)
- Duration: 5-7 days
- Maximum: 6 mg total per course (historic high-dose regimens caused toxicity)
Evidence:
- Low-dose (1.2 mg over 1h) vs high-dose (4.8 mg over 6h): Equal efficacy, 4-fold lower side effects [30]
- Started within 12h of onset: 80% response rate
- Started after 48 h: 50% response rate
Dose Adjustments:
| eGFR (mL/min) | Dose |
|---|---|
| > 60 | Standard (500 μg BD-TDS) |
| 30-60 | 500 μg BD maximum |
| 15-30 | 500 μg OD |
| less than 15 or dialysis | 500 μg every 2-3 days (avoid if possible) |
Drug Interactions (P-glycoprotein and CYP3A4 inhibitors):
- Macrolides (clarithromycin, erythromycin): Reduce colchicine dose by 50%
- Statins: Increased myopathy risk (especially simvastatin)
- Azole antifungals (ketoconazole, itraconazole): Avoid or reduce dose
- Cyclosporine/tacrolimus: Reduce colchicine dose
Side Effects:
- GI: Diarrhoea (23% in low-dose, 77% in high-dose), nausea, vomiting, abdominal pain [30]
- Haematological: Bone marrow suppression (rare, in renal failure)
- Neuromuscular: Myopathy, neuropathy (chronic use)
- Severe toxicity (overdose): Multi-organ failure, death (antidote: Fab fragments in development)
Advantages:
- Can use in CKD (with dose adjustment)
- Can use with anticoagulation
- No cardiovascular risk
3. Corticosteroids
Indications:
- NSAIDs + colchicine contraindicated (e.g., severe CKD, recent GI bleed)
- Polyarticular gout (> 3 joints)
- Patient preference
Regimens:
Oral:
- Prednisolone: 30-35 mg OD for 5 days, then stop (no taper needed for short course)
- OR: 0.5 mg/kg OD (if > 70 kg, max 35 mg)
- Duration: 5-7 days
- Evidence: Equivalent efficacy to NSAIDs with fewer GI side effects [31]
Intra-Articular (if monoarthritis and sepsis excluded):
- Triamcinolone acetonide: 10-40 mg (depending on joint size)
- Ankle: 20-30 mg
- Knee: 30-40 mg
- 1st MTPJ: 10-20 mg
- Effect: Often provides relief within 1-6 hours (fastest onset of all treatments)
- Advantage: Avoids systemic side effects
- Technique: Aspirate joint first (send for crystals/culture), then inject through same needle
Intramuscular:
- Methylprednisolone: 120 mg IM single dose
- Triamcinolone: 60 mg IM
- Use: If oral route not possible, multiple joints affected
Contraindications:
- Septic arthritis not excluded: Steroids will worsen infection
- Uncontrolled diabetes: Hyperglycaemia risk (may still use with monitoring)
- Active infection
Second-Line/Specialist Therapies
IL-1 Inhibitors (Biologic Therapies)
Anakinra (IL-1 Receptor Antagonist):
- Dose: 100 mg SC daily for 3 days
- Evidence: 90% response rate in refractory gout [32]
- Use: Contraindications to all first-line agents, refractory to standard therapy
- Availability: Off-label in most countries (licensed use: rheumatoid arthritis)
- Cost: High (£10,000/year if used long-term)
Canakinumab (IL-1β Monoclonal Antibody):
- Dose: 150 mg SC single dose
- Evidence: FDA-approved for gout flares in patients with contraindications to other therapies [33]
- Advantage: Single injection (long half-life)
- Cost: Very high (£1,000-2,000 per injection)
- Availability: Limited (not NICE-approved in UK for gout)
Combination Therapy
Acceptable Combinations:
- Colchicine + NSAID
- Colchicine + corticosteroid
- NSAID + corticosteroid (rarely needed)
Avoid: Dual NSAID use (no additional benefit, increased toxicity)
Non-Responders and Red Flags
If No Improvement in 48-72 Hours:
- Reconsider diagnosis:
- Septic arthritis (aspirate, empiric antibiotics)
- Fracture (X-ray, MRI)
- Alternative crystal (CPPD)
- Switch therapy: If on NSAID → try colchicine or steroid
- Consider combination: Add colchicine to NSAID
- Specialist referral: For IL-1 inhibitor consideration
8. Management: Long-Term Urate-Lowering Therapy (ULT)
(Treat-to-Target Strategy)
The Paradigm Shift: Treat-to-Target
The 2010s saw a revolution in gout management with adoption of the treat-to-target approach, endorsed by BSR 2017, EULAR 2016, and ACR 2020 guidelines. [5,6,14]
Core Principles:
- Treat to a specific serum urate target (not a fixed drug dose)
- Target: less than 6.0 mg/dL (360 μmol/L) for all patients, less than 5.0 mg/dL (300 μmol/L) if tophi/erosions
- Titrate ULT dose every 4-8 weeks until target achieved
- Monitor urate long-term to ensure sustained target achievement
- Provide prophylaxis for 3-6 months to prevent flares during urate lowering
Evidence:
- Nurse-led treat-to-target RCT (NOTTINGHAM Study, 2018): 92% achieved target vs 32% in usual care (pless than 0.001) [34]
- Target less than 6.0 mg/dL: 71% reduction in flare frequency over 1 year [34]
- Target less than 5.0 mg/dL with tophi: 100% tophus resolution in 77% at 2 years [26]
Indications for Starting ULT (ACR 2020 Guidelines): [5]
Strong Recommendations (Must Offer ULT):
- ≥2 gout flares per year
- Tophi (clinical or imaging-detected)
- Chronic kidney disease stage 3+ (eGFR less than 60)
- History of urate kidney stones
Conditional Recommendations (Consider ULT):
- First gout flare if:
- Age less than 40 years
- Very high urate (> 9.0 mg/dL, 540 μmol/L)
- Comorbidities: CKD, heart failure, hypertension
- Asymptomatic hyperuricaemia: NOT routinely indicated (EULAR guideline) [6]
Timing of ULT Initiation:
- Traditional approach: Wait 2-4 weeks after flare resolution
- 2020 ACR update: Can start during or immediately after flare if patient educated and on prophylaxis [5]
- Rationale: Reduces time to target, improves adherence
- Requires good prophylaxis compliance
First-Line ULT: Allopurinol
Mechanism: Xanthine oxidase inhibitor (purine analogue) → reduces uric acid production
Starting Dose:
- Normal renal function (eGFR > 60): 100 mg OD
- CKD stage 3 (eGFR 30-60): 50-100 mg OD
- CKD stage 4+ (eGFR less than 30): 50 mg OD (or alternate days) [35]
Titration Protocol:
- Start 100 mg OD (with prophylaxis)
- Check urate at 4 weeks
- If not at target, increase by 100 mg
- Repeat cycle until target less than 360 μmol/L (less than 6 mg/dL) achieved
- Usual dose range: 200-600 mg OD
- Maximum dose: 900 mg OD (rarely needed; > 600 mg requires specialist supervision)
Evidence:
- 100 mg: 30-40% achieve target
- 300 mg: 50-60% achieve target
- 600 mg: 80-85% achieve target [14]
- Key point: Many patients under-dosed (40% remain on 100 mg inappropriately) [34]
Allopurinol Hypersensitivity Syndrome (AHS/DRESS):
Incidence: 0.1-0.4% overall; 2-4% in Han Chinese (HLA-B*5801 carriers)
Clinical Features:
- Onset: 2-8 weeks after starting
- Rash: Maculopapular → Stevens-Johnson syndrome/TEN (30% mortality)
- Systemic: Fever, eosinophilia, hepatitis, renal failure
- DRESS (Drug Reaction with Eosinophilia and Systemic Symptoms)
Prevention:
- HLA-B*5801 screening recommended in: [5]
- Han Chinese
- Korean
- Thai
- African American (lower prevalence but still elevated)
- If positive: Avoid allopurinol → use febuxostat
- Start low dose (50-100 mg): Reduces risk 10-fold vs starting > 300 mg
Management of AHS:
- Stop allopurinol immediately
- Admit if systemic features
- Supportive care: IV fluids, antihistamines
- Steroids if severe (prednisolone 1 mg/kg)
- Never re-challenge with allopurinol
- Future ULT: Febuxostat or allopurinol desensitization (specialist-led)
Drug Interactions:
- Azathioprine/6-mercaptopurine: Fatal interaction
- Mechanism: Allopurinol inhibits xanthine oxidase, which metabolizes these drugs → accumulation → bone marrow suppression → sepsis/death
- Action: Reduce azathioprine dose by 75% OR switch to mycophenolate OR use febuxostat (same interaction)
- Warfarin: May potentiate anticoagulant effect (monitor INR)
- Ciclosporin: Allopurinol reduces levels (monitor)
Second-Line ULT: Febuxostat
Mechanism: Non-purine selective xanthine oxidase inhibitor (more potent than allopurinol)
Indications:
- Allopurinol intolerance/hypersensitivity
- Failure to achieve target on allopurinol 600-900 mg
- CKD stage 4-5 (no dose adjustment needed, unlike allopurinol) [35]
Dosing:
- Start: 40 mg OD (with prophylaxis)
- Titrate: Increase to 80 mg OD at 4 weeks if not at target
- Maximum: 120 mg OD (available in some countries)
Comparative Efficacy (CONFIRMS Trial):
- Febuxostat 80 mg: 67% achieve less than 6 mg/dL
- Allopurinol 300 mg: 42% achieve less than 6 mg/dL
- BUT: Allopurinol underdosed (should titrate to 600+ mg) [36]
Cardiovascular Safety Controversy:
CARES Trial (2018): [37]
- RCT: Febuxostat vs allopurinol in patients with CVD
- Result: Higher all-cause mortality (HR 1.22, p=0.04) and cardiovascular mortality (HR 1.34, p=0.03) with febuxostat
- Limitations: High dropout rate (57%), underdosed allopurinol
FAST Trial (2020): [38]
- UK-based RCT: Febuxostat vs allopurinol in CKD patients
- Result: No difference in cardiovascular events (HR 1.03, p=0.79)
- Conclusion: Febuxostat safe in CKD, no cardiovascular signal
Current Guidelines (ACR 2020, EULAR 2016): [5,6]
- Febuxostat safe to use
- Caution in severe IHD, recent MI (less than 6 months), heart failure
- Allopurinol remains first-line due to cost and longer safety track record
- Febuxostat advantage: No dose adjustment in CKD
Drug Interactions:
- Azathioprine: Same interaction as allopurinol (reduce azathioprine dose by 75%)
- Theophylline: Febuxostat increases levels (monitor)
Third-Line: Uricosuric Agents
Promote renal urate excretion. Rarely used as monotherapy in modern practice; reserved for add-on therapy.
Probenecid:
- Dose: 500 mg BD, increase to 1000 mg BD
- Mechanism: Inhibits URAT1 transporter in proximal tubule
- Efficacy: 60% achieve target at 2000 mg/day
- Contraindications:
- eGFR less than 60 (ineffective)
- History of urate kidney stones (increases urinary urate → stone risk)
- Availability: Limited in many countries (not widely available in UK)
Benzbromarone (Not Available in UK/US):
- More potent than probenecid
- Dose: 50-200 mg OD
- Withdrawn in many countries due to hepatotoxicity (rare but severe)
- Still used in Europe, Asia
Lesinurad (RDEA594):
- URAT1 inhibitor
- Licensed as add-on to allopurinol/febuxostat (not monotherapy)
- Dose: 200 mg OD
- Warning: Renal toxicity if used without XOI
- Limited use due to cost, safety concerns
Fourth-Line: Pegloticase (Severe Refractory Gout)
Mechanism: PEGylated recombinant uricase enzyme → directly converts urate to allantoin (soluble)
Indications (Highly Selective):
- Severe tophaceous gout unresponsive to maximum doses of allopurinol + febuxostat
- Intolerance to all XOIs
- Contraindication to all XOIs
Dosing:
- 8 mg IV infusion every 2 weeks
- Effect: Urate drops to less than 1 mg/dL within hours (near-complete depletion)
- Duration: 6-12 months to dissolve tophi, then consider stopping/reducing frequency
Evidence:
- RCTs: 40-47% complete response (all tophi resolved) at 6 months [39]
- Contraindicated in G6PD deficiency (severe haemolysis risk)
Side Effects:
- Infusion reactions: 25% (pre-medicate with antihistamine, steroid)
- Anti-drug antibodies: Develop in 40% → loss of efficacy
- Gout flares: Frequent in first 3 months (co-prescribe prophylaxis)
Co-treatment Strategies (Emerging):
- Immunomodulation (methotrexate, azathioprine, mycophenolate) reduces antibody formation → sustained response in 70-80% [40]
- SEL-212 (in development): Pegloticase + rapamycin nanoparticles to induce tolerance
Cost: £30,000-60,000/year (limited to specialist centres)
Flare Prophylaxis During ULT Initiation
Rationale: Rapid urate lowering dissolves crystals → shedding → flares in 25-60% without prophylaxis [14]
Duration: 3-6 months (ACR/EULAR) [5,6]
- Or until: 1) Urate at target for 6+ months AND 2) No tophi AND 3) No flares for 6+ months
Options:
1. Colchicine (First-Line):
- Dose: 500 μg OD-BD
- Duration: 3-6 months
- Evidence: Reduces flare frequency by 80% vs placebo [41]
- Adjust in CKD: 500 μg OD if eGFR 30-60; alternate days if eGFR 15-30
- Side effects: Diarrhoea (15-20%), myopathy (chronic use)
2. Low-Dose NSAID:
- Naproxen: 250 mg BD
- Duration: 3-6 months
- Contraindications: Same as acute use (CKD, GI bleed, CVD)
- Add PPI: Gastroprotection mandatory
3. Low-Dose Prednisolone (If Both Above Contraindicated):
- Dose: 5 mg OD
- Duration: Maximum 3 months (long-term steroid toxicity)
- Monitor: Glucose, bone health
Non-Responders: If flares occur despite prophylaxis, check:
- Compliance with ULT and prophylaxis
- Urate level (is target achieved?)
- Consider extending prophylaxis to 12 months
Monitoring on ULT
Serum Urate:
- During titration: Every 4 weeks until at target
- Once stable at target: Every 6-12 months
- Target: less than 6 mg/dL (360 μmol/L) for all; less than 5 mg/dL (300 μmol/L) if tophi
Renal Function:
- Baseline, 3 months, then annually
- More frequent if CKD stage 3+
Liver Function:
- Baseline, 3 months (for allopurinol/febuxostat)
- Discontinue if ALT > 3x ULN
Tophus Measurement (If Present):
- Clinical size measurement every 6 months
- Imaging (DECT, ultrasound) annually to assess dissolution
Flare Diary:
- Patients record frequency and severity of flares
- Goal: Zero flares once at target for 6+ months
9. Calcium Pyrophosphate Deposition Disease (CPPD/Pseudogout)
(Comparative Crystal Arthropathy)
CPPD is the second most common crystal arthropathy, often confused with gout due to similar acute presentations.
Pathophysiology
Crystal Type: Calcium pyrophosphate dihydrate (CPP)
Mechanism:
- Pyrophosphate (PPi) is normally produced by chondrocytes and broken down by alkaline phosphatase
- Excess PPi or reduced breakdown → accumulation → crystallization in cartilage matrix
- Calcium binds → CPP crystal formation
Epidemiology and Risk Factors
Age: Peak > 60 years (rare less than 50)
- Prevalence: 7% of adults > 60, 50% of adults > 85
Associations (The "5 H's"):
- Hyperparathyroidism: Hypercalcaemia promotes crystal formation
- Haemochromatosis: Iron deposition in synovium
- Hypomagnesaemia: Magnesium deficiency reduces PPi breakdown
- Hypophosphatasia: Deficient alkaline phosphatase → PPi accumulation
- Hypothyroidism: Mechanism unclear
Other:
- Osteoarthritis (co-exists in 50%)
- Familial CPPD: Rare genetic forms (ANKH, TNFRSF11B mutations)
- Trauma, surgery (trigger acute flares)
Clinical Presentation
Acute CPP Crystal Arthritis ("Pseudogout"):
- Joints: Knee (50%), wrist (25%), shoulder, ankle, elbow
- Onset: Acute (over 12-24h) but less rapid than gout
- Severity: Moderate-severe pain (6-8/10)
- Triggers: Surgery, illness, trauma
Chronic CPP Crystal Inflammatory Arthritis:
- Mimics rheumatoid arthritis
- Symmetric polyarthritis, morning stiffness
- Low-grade chronic inflammation
Osteoarthritis with CPPD:
- Accelerates OA progression
- Atypical OA distribution (wrists, MCPs, shoulders)
Asymptomatic CPPD (Lanthanic):
- Chondrocalcinosis on X-ray without symptoms
- 40% of those > 80 years
Diagnosis
X-Ray:
- Chondrocalcinosis: Linear/punctate calcification within cartilage
- Knee: Meniscal cartilage (hyaline and fibrocartilage)
- Wrist: Triangular fibrocartilage complex (TFCC)
- Symphysis pubis
- Intervertebral discs
- Sensitivity: 40-60% (many patients have crystals without visible calcification)
Joint Aspiration:
- Crystals: Rhomboid, 2-10 μm, weak positive birefringence
- Blue when parallel to polarizer
- Yellow when perpendicular
- Often fewer crystals than gout (can be missed)
Metabolic Workup (If Age less than 60 or Polyarticular):
- Serum calcium, phosphate, magnesium, alkaline phosphatase, PTH
- Ferritin, transferrin saturation (haemochromatosis)
- TSH (hypothyroidism)
Management
Acute Flares:
- NSAIDs: First-line (same as gout)
- Colchicine: Effective (500 μg BD)
- Intra-articular corticosteroid: Rapid relief for monoarthritis
Long-Term:
- No equivalent to ULT: No drug dissolves CPP crystals
- Treat underlying metabolic causes if present
- Colchicine prophylaxis: May reduce flare frequency in recurrent disease (off-label, weak evidence)
- Joint protection, physiotherapy
10. Lifestyle Modification
(Evidence-Based Non-Pharmacological Management)
Lifestyle interventions are synergistic with ULT and can reduce serum urate by 1-2 mg/dL. [20]
Dietary Modification
Reduce Purine-Rich Foods:
- Very high purines (> 200 mg/100g): Avoid or limit to less than 1 serving/week
- Organ meats: Liver, kidney, heart, sweetbreads
- Game meats: Venison, rabbit
- Certain fish: Anchovies, sardines, herring, mackerel
- Shellfish: Scallops, mussels
- Moderate purines (100-200 mg/100g): Limit to 2-3 servings/week
- Red meat: Beef, pork, lamb
- Poultry: Chicken, turkey
- Most fish: Tuna, salmon, trout
Increase Protective Foods:
- Low-fat dairy: 2-3 servings/day reduces risk 20-45% [20]
- Mechanism: Casein/lactalbumin are uricosuric
- Examples: Skimmed milk, low-fat yoghurt
- Cherries: 10-20 cherries/day (anecdotal 35% flare reduction) [42]
- Active compounds: Anthocyanins (anti-inflammatory)
- Vitamin C: 500-1000 mg/day (10-15% urate reduction) [43]
- Food sources: Citrus, kiwi, peppers, broccoli
Neutral Foods (Safe to Consume):
- Vegetables (all types, including high-purine vegetables like spinach, mushrooms - vegetable purines don't increase gout risk)
- Whole grains
- Nuts and legumes
- Eggs
Alcohol Modification
Risk Hierarchy: [19]
- Beer: Highest risk (RR 1.49 per 12 oz/day) → rich in guanosine (purine)
- Spirits: Moderate risk (RR 1.15 per 40 mL/day)
- Wine: Neutral/protective (RR 1.0-1.04)
Recommendations:
- Ideal: Abstinence (especially during ULT initiation)
- Moderate: ≤1 drink/day (if urate controlled)
- Avoid: Beer and spirits during active disease
- Safe: Wine in moderation (1-2 glasses 2-3x/week) appears safe
Sugar-Sweetened Beverage Reduction
Evidence:
- Each daily serving increases gout risk 13% [21]
- Mechanism: Fructose metabolism → ATP depletion → AMP degradation → urate production
Action:
- Avoid: Sodas, fruit juices with added sugar, energy drinks
- Safe: Water, tea, coffee, diet sodas (artificial sweeteners safe)
Weight Loss
Target: BMI less than 25 (or 5-10 kg loss if obese)
Evidence:
- 10 kg weight loss → 0.5-1.0 mg/dL urate reduction [44]
- Weight loss > 7 kg → 40% reduction in flare frequency
Approach:
- Gradual: 0.5-1 kg/week (rapid weight loss can precipitate flares)
- Mechanism: Improved insulin sensitivity → increased renal urate excretion
- Avoid: Very low-carbohydrate diets (keto) → ketosis competes with urate for renal excretion
Hydration
Target: 2-3 L fluid/day
Rationale:
- Prevents supersaturation in joints and kidneys
- Reduces kidney stone risk
- Dilutes synovial fluid urate concentration
Preferred Fluids:
- Water (primary)
- Coffee: 4+ cups/day reduces gout risk 40-60% (mechanism unclear)
- Tea
Medication Review
Discontinue or Replace Gout-Provoking Drugs (If Medically Safe):
| Drug | Alternative |
|---|---|
| Diuretic (thiazide/loop) | ACE inhibitor, ARB (losartan is uricosuric), calcium channel blocker |
| Low-dose aspirin | Continue if for CV protection (benefits outweigh gout risk) |
| Ciclosporin | Tacrolimus (lower gout risk), mycophenolate |
Losartan (Unique ARB):
- Mild uricosuric effect (blocks URAT1)
- Preferred antihypertensive in gout patients
- Reduces urate by 0.5-1.0 mg/dL
11. Complications
(Long-Term Sequelae of Uncontrolled Gout)
Renal Complications
Chronic Urate Nephropathy:
- Deposition of urate crystals in renal interstitium and tubules
- Leads to tubular atrophy, interstitial fibrosis, glomerulosclerosis
- Mechanism: Urate crystals → inflammasome activation → chronic inflammation → scarring
- Clinical: Progressive decline in eGFR
- Evidence: Controversial whether hyperuricaemia causes CKD or is merely association
- Observational: Each 1 mg/dL urate increase → 5-10% increased CKD risk
- RCTs of ULT in CKD: Mixed results (PERL, CKD-FIX trials showed no benefit)
Uric Acid Nephrolithiasis (Kidney Stones):
- Prevalence: 10-25% of gout patients
- Risk factors:
- Low urine pH (less than 5.5): Uric acid insoluble in acidic urine
- High urinary urate (> 800 mg/24h)
- Low urine volume (less than 1 L/day)
- Stone characteristics:
- Radiolucent (pure uric acid stones invisible on X-ray)
- Visible on CT
- Management:
- Hydration (2-3 L/day)
- Urine alkalinization: Potassium citrate 30-60 mEq/day (target urine pH 6.0-7.0)
- ULT: Allopurinol/febuxostat (prevents new stone formation, may dissolve existing stones)
Acute Uric Acid Nephropathy (Tumour Lysis Syndrome):
- Massive acute hyperuricaemia (> 15-20 mg/dL) → intratubular precipitation → AKI
- Setting: Chemotherapy for haematological malignancy (ALL, Burkitt lymphoma)
- Prevention: Rasburicase (recombinant uricase) before chemo + IV hydration + allopurinol
Cardiovascular Complications
Increased Cardiovascular Risk:
- Gout independently increases: [16]
- MI risk: HR 1.24 (95% CI 1.17-1.32)
- Stroke risk: HR 1.19 (95% CI 1.08-1.32)
- Heart failure: HR 1.74 (95% CI 1.22-2.50)
- All-cause mortality: HR 1.28
Mechanisms:
- Chronic inflammation: Persistent IL-1β, IL-6, TNF-α → endothelial dysfunction, plaque instability
- Oxidative stress: Xanthine oxidase produces reactive oxygen species
- Metabolic syndrome clustering: 60-70% have HTN, obesity, DM, dyslipidaemia
- Hyperuricaemia-mediated: Urate activates RAAS, reduces nitric oxide, induces vascular smooth muscle proliferation
Management Implications:
- Aggressive cardiovascular risk factor modification:
- Statins (reduce CV events 30-40% in gout)
- Blood pressure control (target less than 130/80)
- Diabetes management
- Smoking cessation
- ULT may reduce CV events: Meta-analysis suggests 15-20% reduction (but RCTs ongoing)
Musculoskeletal Complications
Erosive Joint Damage:
- Chronic tophaceous gout → cartilage erosion, bone destruction
- Radiology: Punched-out erosions with overhanging edges
- Outcome: Secondary osteoarthritis, joint deformity
- Prevention: Early aggressive ULT (treat-to-target)
Chronic Gouty Arthropathy:
- Persistent low-grade inflammation even between flares
- Synovial hypertrophy, pannus formation
- Loss of function, disability
Tendon Involvement:
- Achilles tendon tophi → risk of rupture (spontaneous or minor trauma)
- Rotator cuff calcification → impingement, tears
- Quadriceps tendon tophi
Infection Risk
Septic Arthritis in Gouty Joints:
- Incidence: 0.5-1% of acute flares actually sepsis [10]
- Risk factors: Diabetes, immunosuppression, chronic tophaceous gout
- Challenge: Gout and sepsis clinically indistinguishable
- Management: Low threshold for aspiration + empiric antibiotics
Tophus Ulceration and Secondary Infection:
- Large tophi can ulcerate → discharge chalky material → entry point for bacteria
- Organisms: Staphylococcus aureus, Streptococcus
- Management: Debridement, antibiotics, intensify ULT
Psychosocial Impact
Quality of Life:
- Chronic pain, functional impairment
- Inability to work (10-15% of severe cases)
- Depression prevalence 20-30% (vs 10% general population)
Stigma:
- Historical association with "rich man's disease", overindulgence
- Patient blame and guilt ("lifestyle disease")
- Reality: 60% of risk is genetic and metabolic, not purely dietary
12. Evidence & Guidelines
(Synthesis of High-Quality Evidence)
Major International Guidelines
1. 2020 American College of Rheumatology (ACR) Guideline for the Management of Gout: [5]
Key Recommendations:
- ULT:
- Conditional recommendation to start during acute flare (if patient educated + on prophylaxis)
- Strong recommendation for treat-to-target approach (target less than 6 mg/dL)
- Allopurinol first-line; febuxostat second-line
- Screen for HLA-B*5801 in high-risk populations before allopurinol
- Flare prophylaxis: 3-6 months (colchicine preferred)
- Acute flares: NSAIDs, colchicine, or corticosteroids (all equally effective)
2. 2016 EULAR Evidence-Based Recommendations for the Management of Gout: [6]
Key Points:
- ULT indicated if: ≥2 flares/year, tophi, urate stones, CKD
- Target less than 6 mg/dL (less than 360 μmol/L); less than 5 mg/dL (less than 300 μmol/L) if tophi
- Continue ULT lifelong (indefinite treatment)
- Lifestyle modification integral (weight loss, alcohol reduction, dietary modification)
3. 2018 Updated EULAR Evidence-Based Recommendations for the Diagnosis of Gout: [27]
Diagnostic Algorithm:
- First-line: Joint aspiration + polarized microscopy (gold standard)
- If aspiration not possible: Clinical diagnosis supported by:
- Ultrasound (double contour sign)
- DECT (urate deposition)
- X-ray (typical erosions)
- ACR/EULAR 2015 classification criteria not for diagnosis (for research classification)
4. 2017 British Society for Rheumatology (BSR) Guideline: [14]
Emphasis:
- Treat-to-target mandatory (not treating to a fixed dose)
- Patient education crucial (flares during ULT initiation expected)
- Shared decision-making
- Long-term monitoring and adherence support
Landmark Clinical Trials
1. AGREE Trial (2010) - Low-Dose Colchicine: [30]
- Design: RCT, 184 patients, acute gout
- Groups: Low-dose (1.2 mg over 1h) vs high-dose (4.8 mg over 6h) vs placebo
- Result: Low-dose non-inferior to high-dose for pain reduction (both ~40% improvement)
- Adverse events: 23% diarrhoea (low-dose) vs 77% (high-dose)
- Impact: Changed practice from historic high-dose regimens to modern low-dose approach
2. NOTTINGHAM/NUT Study (2018) - Nurse-Led Treat-to-Target: [34]
- Design: RCT, 517 patients, UK primary care
- Intervention: Nurse-led titration to target less than 6 mg/dL vs usual GP care
- Result: 92% achieved target (nurse-led) vs 32% (usual care) at 2 years
- Flare reduction: 71% fewer flares in nurse-led group
- Impact: Demonstrated feasibility and superiority of systematic treat-to-target approach
3. CARES Trial (2018) - Febuxostat vs Allopurinol Cardiovascular Safety: [37]
- Design: RCT, 6,190 patients with CVD
- Result: Higher all-cause mortality (HR 1.22) and CV mortality (HR 1.34) with febuxostat
- Limitations: 57% dropout, underdosed allopurinol, post-hoc analysis
- Impact: FDA black box warning for febuxostat (later removed after FAST trial)
4. FAST Trial (2020) - Febuxostat vs Allopurinol (UK): [38]
- Design: RCT, 6,128 patients with CKD
- Result: No difference in CV events (HR 1.03, p=0.79)
- Conclusion: Febuxostat safe; contradicts CARES
- Impact: Reassured clinicians on febuxostat safety in appropriate populations
5. STOP Gout Trial (2024) - Allopurinol vs Febuxostat in CKD: [45]
- Design: RCT, 940 patients with CKD stage 3-4
- Result: Both effective and safe; no difference in eGFR decline
- Impact: Either XOI acceptable in CKD with dose adjustment
6. COLCHICINE Trial (Cardiovascular - COLCOT, LoDoCo2): [46]
- Design: RCTs in post-MI patients
- Intervention: Low-dose colchicine 0.5 mg OD
- Result: 30-35% reduction in CV events (IL-1β inhibition anti-atherosclerotic)
- Impact: Colchicine now guideline-recommended for secondary CV prevention (bonus benefit in gout patients)
13. Patient Explanation
(Communicating Gout to Patients)
What is Gout in Simple Terms?
"Gout is caused by a chemical called uric acid building up in your blood. When levels get too high, it forms sharp crystals - like tiny shards of glass - in your joints. These crystals trigger your body's alarm system, causing massive inflammation. That's why gout pain is so severe - your white blood cells are attacking the crystals, but they can't destroy them, so the inflammation keeps building."
Why Does It Happen to Me?
"It's mostly genetic. About 60-70% of your gout risk is inherited - your kidneys may not flush out uric acid as efficiently as other people's. The rest is related to diet, weight, medications like water tablets, and other health conditions like high blood pressure or diabetes."
"It's NOT just about what you eat. Yes, diet plays a role, but even if you eat perfectly, you may still get gout if your genes predispose you. Don't blame yourself."
Why Does It Hurt at Night?
"Three reasons:
- Your feet get colder at night, and crystals form more easily in the cold (like how water freezes in the freezer).
- You breathe slower during sleep, so carbon dioxide builds up slightly, making your blood more acidic - this encourages crystal formation.
- Your kidneys are less active at night, so uric acid concentrates in your joints."
Common Misconceptions
"I stopped the allopurinol because I got an attack."
"This is the most common mistake we see. Here's what's happening: Allopurinol is dissolving the crystals. As they shrink and break apart, small pieces fall out of the cartilage into the joint fluid - this triggers a flare. It means the drug is working, not failing.
Think of it like cleaning out a messy cupboard. You pull everything out (flare), clean it properly (dissolving crystals), then put it back organized (no more crystals). If you stop allopurinol during a flare, you leave the cupboard half-cleaned - the mess just builds up again.
Solution: Stay on the allopurinol. Take the painkiller (colchicine or naproxen) alongside it. Eventually all the crystals will dissolve, and you'll be cured."
"Can I ever drink beer again?"
"Beer is the worst trigger because it contains guanosine, a purine that breaks down into uric acid. It also stops your kidneys from clearing uric acid.
If you get your uric acid level very low with medication (less than 5 mg/dL) and maintain it there for 1-2 years with no flares, you might tolerate an occasional pint without triggering an attack. But regular drinking will overwhelm the medication.
Wine is much safer - 1-2 glasses a few times a week is usually fine for most people on treatment."
"Why do I need treatment between attacks if I feel fine?"
"This is crucial to understand: Even when you feel fine, crystals are silently building up in your joints. Every time you have a flare, you're causing more damage. Over 10-20 years, this leads to:
- Permanent joint destruction (arthritis that never goes away)
- Lumps under your skin (tophi)
- Kidney damage
Treating between attacks with allopurinol dissolves those silent crystals and prevents all these complications. Think of it like treating high blood pressure - you feel fine, but treatment prevents future strokes."
What's the Goal of Treatment?
"The goal is to get your uric acid level below 6 mg/dL (360 μmol/L) and keep it there. At this level:
- Crystals can't form
- Existing crystals slowly dissolve
- After 6-12 months at target, flares should stop completely
- After 2-5 years, any tophi (lumps) should disappear
This is a cure, not just symptom control. But it requires lifelong medication, like diabetes or blood pressure treatment."
What Can I Do to Help?
Diet:
- Avoid: Organ meats (liver, kidney), shellfish, excessive red meat, beer
- Limit: Regular meat, seafood, spirits
- Safe: Vegetables (all types), dairy (especially low-fat), cherries, coffee, wine in moderation
- Drink: 2-3 litres of water daily
Weight:
- Lose weight gradually (0.5-1 kg/week) if overweight
- Even 5-10 kg loss significantly reduces flares
Medications:
- Tell your doctor you have gout before starting new medications (especially water tablets)
- Never stop allopurinol without discussing with your doctor
When to Seek Help Urgently
- Fever > 38.5°C with hot joint: Could be infection (septic arthritis) - go to A&E
- Multiple joints suddenly swollen: Needs urgent assessment
- Severe rash after starting allopurinol: Stop immediately and call your doctor (drug allergy)
14. References
(Comprehensive Evidence Base)
-
Dehlin M, Jacobsson L, Roddy E. Global epidemiology of gout: prevalence, incidence, treatment patterns and risk factors. Nat Rev Rheumatol. 2020 Jul;16(7):380-390. PMID: 32541923
-
Dalbeth N, Gosling AL, Gaffo A, Abhishek A. Gout. Lancet. 2021 Nov 13;397(10287):1843-1855. PMID: 33798479
-
Dalbeth N, Merriman TR, Stamp LK. Gout. Lancet. 2016 Oct 22;388(10055):2039-2052. PMID: 27112094
-
Roddy E, Doherty M. Epidemiology of gout. Arthritis Res Ther. 2010;12(6):223. PMID: 21205285
-
FitzGerald JD, Dalbeth N, Mikuls T, et al. 2020 American College of Rheumatology Guideline for the Management of Gout. Arthritis Care Res (Hoboken). 2020 Jun;72(6):744-760. PMID: 32391934
-
Richette P, Doherty M, Pascual E, et al. 2016 updated EULAR evidence-based recommendations for the management of gout. Ann Rheum Dis. 2017 Jan;76(1):29-42. PMID: 27457514
-
Kuo CF, Grainge MJ, Zhang W, Doherty M. Global epidemiology of gout: prevalence, incidence and risk factors. Nat Rev Rheumatol. 2015 Nov;11(11):649-62. PMID: 26150127
-
Zhu Y, Pandya BJ, Choi HK. Prevalence of gout and hyperuricemia in the US general population: the National Health and Nutrition Examination Survey 2007-2008. Arthritis Rheum. 2011 Oct;63(10):3136-41. PMID: 21800283
-
Faires JS, McCarty DJ. Acute arthritis in man and dog after intrasynovial injection of sodium urate crystals. Lancet. 1962 Sep 22;2(7256):682-5. PMID: 14021322
-
Mandell BF, Pillinger MH. Therapeutic approaches to gouty arthritis and other crystalline arthritides. Nat Clin Pract Rheumatol. 2007 Dec;3(12):688-97. PMID: 18037930
-
Choi HK, Ford ES. Prevalence of the metabolic syndrome in individuals with hyperuricemia. Am J Med. 2007 May;120(5):442-7. PMID: 17466656
-
Neogi T, Jansen TL, Dalbeth N, et al. 2015 Gout classification criteria: an American College of Rheumatology/European League Against Rheumatism collaborative initiative. Ann Rheum Dis. 2015 Oct;74(10):1789-98. PMID: 26359487
-
Logan JA, Morrison E, McGill PE. Serum uric acid in acute gout. Ann Rheum Dis. 1997 Nov;56(11):696-7. PMID: 9462175
-
Hui M, Carr A, Cameron S, et al. The British Society for Rheumatology Guideline for the Management of Gout. Rheumatology (Oxford). 2017 Jul 1;56(7):e1-e20. PMID: 28541519
-
Ramon A, Riesco L, Narvaez JA, et al. Role of dual-energy CT in the diagnosis and follow-up of gout: systematic analysis of the literature. Clin Rheumatol. 2018 Mar;37(3):587-598. PMID: 29350330
-
Choi HK, Curhan G. Independent impact of gout on mortality and risk for coronary heart disease. Circulation. 2007 Aug 21;116(8):894-900. PMID: 17698728
-
Kuo CF, Grainge MJ, Mallen C, Zhang W, Doherty M. Rising burden of gout in the UK but continuing suboptimal management: a nationwide population study. Ann Rheum Dis. 2015 Apr;74(4):661-7. PMID: 24431399
-
Choi HK, Atkinson K, Karlson EW, Willett W, Curhan G. Purine-rich foods, dairy and protein intake, and the risk of gout in men. N Engl J Med. 2004 Mar 11;350(11):1093-103. PMID: 15014182
-
Choi HK, Atkinson K, Karlson EW, Willett W, Curhan G. Alcohol intake and risk of incident gout in men: a prospective study. Lancet. 2004 Apr 17;363(9417):1277-81. PMID: 15094272
-
Choi HK, Liu S, Curhan G. Intake of purine-rich foods, protein, and dairy products and relationship to serum levels of uric acid: the Third National Health and Nutrition Examination Survey. Arthritis Rheum. 2005 Jan;52(1):283-9. PMID: 15641075
-
Choi HK, Curhan G. Soft drinks, fructose consumption, and the risk of gout in men: prospective cohort study. BMJ. 2008 Feb 9;336(7639):309-12. PMID: 18244959
-
Choi HK, Soriano LC, Zhang Y, Rodríguez LA. Antihypertensive drugs and risk of incident gout among patients with hypertension: population based case-control study. BMJ. 2012 Jan 11;344:d8190. PMID: 22240117
-
Stamp LK, Chapman PT. Gout and organ transplantation. Curr Rheumatol Rep. 2012 Apr;14(2):165-72. PMID: 22302575
-
Merriman TR. An update on the genetic architecture of hyperuricemia and gout. Arthritis Res Ther. 2015 Apr 10;17:98. PMID: 25889045
-
Martinon F, Pétrilli V, Mayor A, Tardivel A, Tschopp J. Gout-associated uric acid crystals activate the NALP3 inflammasome. Nature. 2006 Mar 9;440(7081):237-41. PMID: 16407889
-
Perez-Ruiz F, Calabozo M, Pijoan JI, Herrero-Beites AM, Ruibal A. Effect of urate-lowering therapy on the velocity of size reduction of tophi in chronic gout. Arthritis Rheum. 2002 Dec;47(6):356-60. PMID: 12209480
-
Richette P, Doherty M, Pascual E, et al. 2018 updated European League Against Rheumatism evidence-based recommendations for the diagnosis of gout. Ann Rheum Dis. 2020 Jan;79(1):31-38. PMID: 31167758
-
Khanna I, Veetil J, Khanna D. What Has Dual Energy CT Taught Us About Gout? Curr Rheumatol Rep. 2021 Jul 14;23(7):54. PMID: 34259946
-
van Durme CM, Wechalekar MD, Buchbinder R, et al. Non-steroidal anti-inflammatory drugs for acute gout. Cochrane Database Syst Rev. 2014 Sep 16;(9):CD010120. PMID: 25223353
-
Terkeltaub RA, Furst DE, Bennett K, et al. High versus low dosing of oral colchicine for early acute gout flare: Twenty-four-hour outcome of the first multicenter, randomized, double-blind, placebo-controlled, parallel-group, dose-comparison colchicine study. Arthritis Rheum. 2010 Apr;62(4):1060-8. PMID: 20131255
-
Janssens HJ, Janssen M, van de Lisdonk EH, van Riel PL, van Weel C. Use of oral prednisolone or naproxen for the treatment of gout arthritis: a double-blind, randomised equivalence trial. Lancet. 2008 May 31;371(9627):1854-60. PMID: 18514729
-
So A, De Smedt T, Revaz S, Tschopp J. A pilot study of IL-1 inhibition by anakinra in acute gout. Arthritis Res Ther. 2007;9(2):R28. PMID: 17352828
-
Schlesinger N, Alten RE, Bardin T, et al. Canakinumab for acute gouty arthritis in patients with limited treatment options: results from two randomised, multicentre, active-controlled, double-blind trials and their initial extensions. Ann Rheum Dis. 2012 Nov;71(11):1839-48. PMID: 22586173
-
Doherty M, Jenkins W, Richardson H, et al. Efficacy and cost-effectiveness of nurse-led care involving education and engagement of patients and a treat-to-target urate-lowering strategy versus usual care for gout: a randomised controlled trial. Lancet. 2018 Oct 20;392(10156):1403-1412. PMID: 30343856
-
Vargas-Santos AB, Neogi T. Management of Gout and Hyperuricemia in CKD. Am J Kidney Dis. 2017 Sep;70(3):422-439. PMID: 28456346
-
Becker MA, Schumacher HR Jr, Wortmann RL, et al. Febuxostat compared with allopurinol in patients with hyperuricemia and gout. N Engl J Med. 2005 Dec 8;353(23):2450-61. PMID: 16339094
-
White WB, Saag KG, Becker MA, et al. Cardiovascular Safety of Febuxostat or Allopurinol in Patients with Gout. N Engl J Med. 2018 Mar 29;378(13):1200-1210. PMID: 29527974
-
Mackenzie IS, Ford I, Nuki G, et al. Long-term cardiovascular safety of febuxostat compared with allopurinol in patients with gout (FAST): a multicentre, prospective, randomised, open-label, non-inferiority trial. Lancet. 2020 Nov 28;396(10264):1745-1757. PMID: 33181081
-
Sundy JS, Baraf HS, Yood RA, et al. Efficacy and tolerability of pegloticase for the treatment of chronic gout in patients refractory to conventional treatment: two randomized controlled trials. JAMA. 2011 Aug 17;306(7):711-20. PMID: 21846852
-
Schlesinger N, Lipsky PE. Pegloticase treatment of chronic refractory gout: Update on efficacy and safety. Semin Arthritis Rheum. 2020 Jun;50(3S):S31-S38. PMID: 32620200
-
Borstad GC, Bryant LR, Abel MP, et al. Colchicine for prophylaxis of acute flares when initiating allopurinol for chronic gouty arthritis. J Rheumatol. 2004 Dec;31(12):2429-32. PMID: 15570646
-
Zhang Y, Neogi T, Chen C, Chaisson C, Hunter DJ, Choi HK. Cherry consumption and decreased risk of recurrent gout attacks. Arthritis Rheum. 2012 Dec;64(12):4004-11. PMID: 23023818
-
Huang HY, Appel LJ, Choi MJ, et al. The effects of vitamin C supplementation on serum concentrations of uric acid: results of a randomized controlled trial. Arthritis Rheum. 2005 Jun;52(6):1843-7. PMID: 15934094
-
Dessein PH, Shipton EA, Stanwix AE, Joffe BI, Ramokgadi J. Beneficial effects of weight loss associated with moderate calorie/carbohydrate restriction, and increased proportional intake of protein and unsaturated fat on serum urate and lipoprotein levels in gout: a pilot study. Ann Rheum Dis. 2000 Jul;59(7):539-43. PMID: 10873964
-
Helget LN, Mara KC, McCarthy JT, et al. Efficacy and Safety of Allopurinol and Febuxostat in Patients With Gout and CKD: Subgroup Analysis of the STOP Gout Trial. Am J Kidney Dis. 2024 Nov;84(5):571-580.e1. PMID: 38906504
-
Tardif JC, Kouz S, Waters DD, et al. Efficacy and Safety of Low-Dose Colchicine after Myocardial Infarction. N Engl J Med. 2019 Dec 26;381(26):2497-2505. PMID: 31733140
15. Examination Focus
(High-Yield Exam Content)
Common MCQ/SBA Themes
1. Acute Management
Question: "48-year-old man presents with acute onset of severe right first MTPJ pain and swelling. He has eGFR 25 mL/min and previous peptic ulcer disease. Which is the most appropriate initial treatment?"
Answer: Oral prednisolone 30 mg OD for 5 days
- Rationale: NSAIDs contraindicated (CKD + PUD). Colchicine requires dose reduction in severe CKD (500 μg OD) and has high diarrhoea risk. Steroids are safest and most effective option.
2. ULT Complications
Question: "A patient on azathioprine for Crohn's disease develops gout. He is started on allopurinol 100 mg. Three weeks later, he presents with fever, pancytopenia, and mouth ulcers. What is the most likely diagnosis?"
Answer: Azathioprine toxicity (allopurinol drug interaction)
- Mechanism: Allopurinol inhibits xanthine oxidase → reduced azathioprine metabolism → accumulation → bone marrow suppression.
- Management: Stop both drugs, supportive care. Consider febuxostat (same interaction) vs mycophenolate switch.
3. Crystal Identification
Question: "Joint fluid microscopy shows rhomboid crystals with weak positive birefringence. What is the diagnosis?"
Answer: Calcium pyrophosphate deposition disease (CPPD/pseudogout)
- Gout: Needle-shaped, strong negative birefringence
4. Treatment Targets
Question: "A patient with chronic tophaceous gout is on allopurinol 300 mg. Serum urate is 380 μmol/L (6.4 mg/dL). What is the next step?"
Answer: Increase allopurinol dose (target less than 300 μmol/L with tophi)
- Standard target: less than 360 μmol/L
- Tophi/erosions target: less than 300 μmol/L
- Titrate until target achieved
5. HLA-B*5801 Screening
Question: "In which population should HLA-B*5801 screening be performed before starting allopurinol?"
Answer: Han Chinese, Korean, Thai populations
- Rationale: 10-15% carrier prevalence, 100-fold increased DRESS risk
OSCE/Clinical Exam Stations
History Taking:
- Key features to elicit:
- Rapidity of onset (less than 24h pathognomonic)
- Which joint (podagra 50-70%)
- Severity (10/10 pain, can't bear bedsheet)
- Time of onset (early morning typical)
- Triggers (alcohol, dehydration, new medication)
- Previous episodes
- Risk factors: Obesity, alcohol, hypertension, CKD, diuretics
- Family history (35-65% heritability)
- Red flags: Fever, polyarticular, no response to treatment
Examination:
- Acute gout joint: Red, hot, swollen, shiny, exquisitely tender
- Tophi: Ears (helix), olecranon, fingers, Achilles tendon
- X-ray interpretation: Punched-out erosions with overhanging edges
Counselling Station (Starting Allopurinol):
- "This medication lowers uric acid to dissolve crystals - it's a long-term treatment, not for acute pain."
- "You may get more flares in the first 6 months as crystals break up - this is normal and means it's working."
- "Take colchicine alongside for 3-6 months to prevent flares."
- "Never stop allopurinol during a flare - continue it and add painkillers."
- "We'll increase the dose gradually until your uric acid is below 6 mg/dL."
- "Rare but serious: If you get a rash, stop immediately and call us."
Viva Voce Topics
1. Compare and Contrast Gout vs Pseudogout
| Feature | Gout | Pseudogout |
|---|---|---|
| Crystal | MSU (monosodium urate) | CPP (calcium pyrophosphate) |
| Birefringence | Strong negative | Weak positive |
| Typical joint | 1st MTPJ | Knee, wrist |
| Age | 40-60 | 60-80 |
| X-ray | Erosions with overhanging edges | Chondrocalcinosis |
| Metabolic associations | Metabolic syndrome | Hyperparathyroidism, haemochromatosis |
| Treatment | ULT (allopurinol) dissolves crystals | No specific dissolving therapy |
2. Mechanism of NLRP3 Inflammasome in Gout
"MSU crystals are phagocytosed by macrophages → phagolysosome rupture → cathepsin B release → activates NLRP3 inflammasome complex → cleaves pro-IL-1β via caspase-1 → massive IL-1β release → neutrophil recruitment and cytokine cascade."
Clinical relevance: IL-1 blockers (anakinra, canakinumab) highly effective in acute gout by directly blocking this pathway.
3. Explain Treat-to-Target Strategy
"Traditional gout management used fixed allopurinol doses (often 300 mg). The treat-to-target approach mandates:
- Setting a specific serum urate target (less than 6 mg/dL, less than 360 μmol/L)
- Titrating ULT dose every 4 weeks until target achieved
- Long-term monitoring to ensure sustained target
- Prophylaxis for 3-6 months to prevent flares during titration
Evidence: NOTTINGHAM trial showed 92% achieved target with nurse-led protocol vs 32% usual care, with 71% fewer flares."
4. Allopurinol Hypersensitivity Syndrome (DRESS)
"DRESS (Drug Reaction with Eosinophilia and Systemic Symptoms):
- Onset: 2-8 weeks after starting
- Rash: Maculopapular → Stevens-Johnson/TEN (30% mortality)
- Systemic: Fever, eosinophilia (> 1000), hepatitis, renal failure
- Risk: HLA-B*5801 carriers (100-fold increased risk)
- Prevention: Screen high-risk populations (Han Chinese, Korean, Thai), start low dose (50-100 mg)
- Management: Stop immediately, supportive care, steroids if severe, never re-challenge"
Last Updated: 2026-01-10 Word Count: ~14,500 words | Line Count: 1,458 lines | Citations: 46 PubMed references