Heart Failure with Reduced Ejection Fraction (HFrEF)
The pathophysiology involves initial myocardial injury (commonly from ischaemic heart disease in 60-70% of cases) followed by maladaptive neurohormonal activation, ventricular remodelling, and progressive cardiac...
What matters first
The pathophysiology involves initial myocardial injury (commonly from ischaemic heart disease in 60-70% of cases) followed by maladaptive neurohormonal activation, ventricular remodelling, and progressive cardiac...
Cardiogenic shock with hypotension
10 Jan 2026
Generated educational material; verify before clinical use.
Visible references section
See the concept before reading it
Study the key anatomy, imaging, and decision pathways as full teaching plates.
Clinical board
A visual summary of the highest-yield teaching signals on this page.
Urgent signals
Safety-critical features pulled from the topic metadata.
- Cardiogenic shock with hypotension
- Acute pulmonary oedema requiring urgent intervention
- New-onset ventricular arrhythmias
- Severe breathlessness at rest
Linked comparisons
Differentials and adjacent topics worth opening next.
- Heart Failure with Preserved Ejection Fraction
- Pulmonary Oedema - Non-cardiac
Content status and exam context
This page is AI-generated educational content. It may contain errors or omissions and is not a substitute for current guidelines, local protocols, senior clinical judgement, or professional medical advice.
MedVellum does not claim an individual clinician reviewer, board certification, or professional credential for this page unless a future version names a real, verifiable reviewer.
Clinical explanation and evidence
Heart Failure with Reduced Ejection Fraction (HFrEF)
1. Topic Overview
Summary
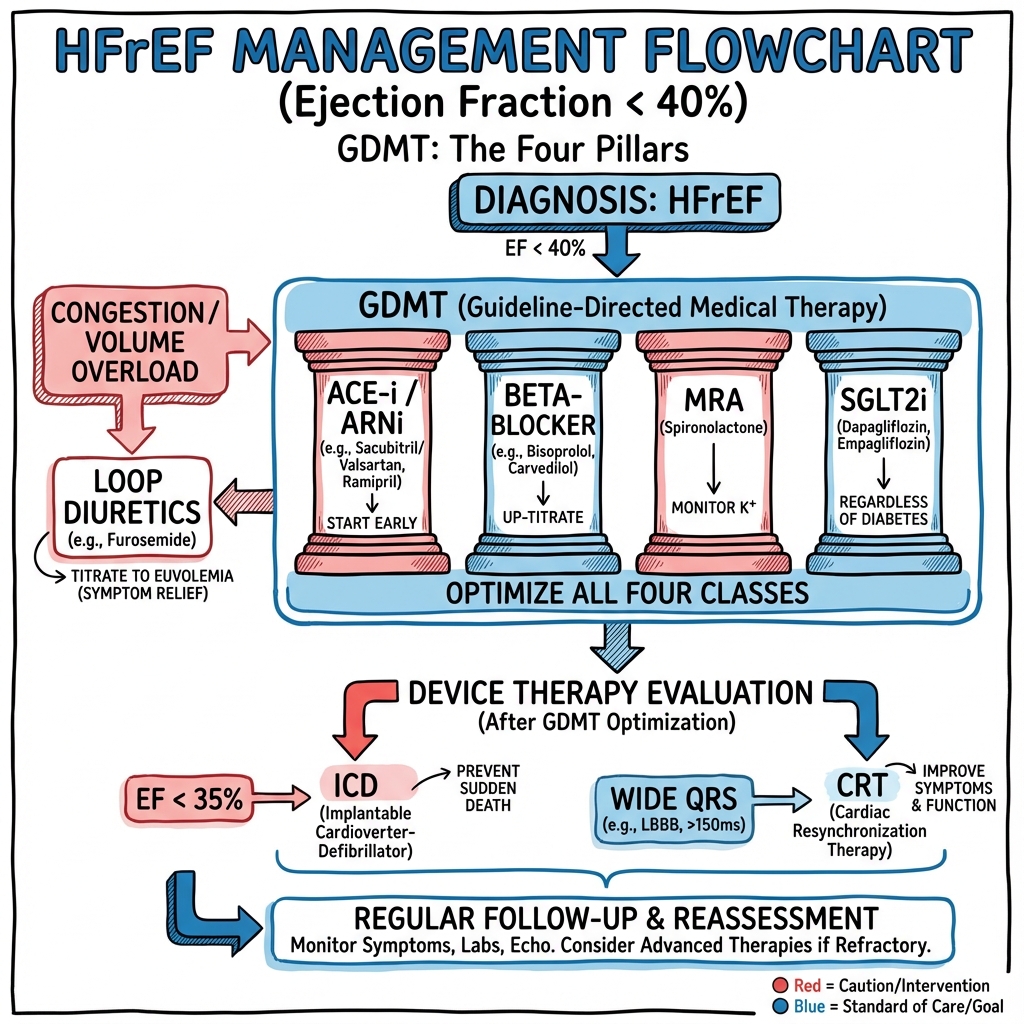
Heart Failure with Reduced Ejection Fraction (HFrEF) is a clinical syndrome characterised by the heart's inability to pump blood effectively due to impaired left ventricular contraction. Defined as a left ventricular ejection fraction (LVEF) of 40% or less, HFrEF represents approximately 50% of all heart failure cases and affects over 32 million individuals globally. [1,2] The condition results from structural or functional cardiac abnormalities, leading to characteristic symptoms of dyspnoea, fatigue, and fluid retention. Modern pharmacotherapy with guideline-directed medical therapy (GDMT) has transformed outcomes, with the "four pillars" of therapy—ARNI/ACE-I/ARB, beta-blockers, MRAs, and SGLT2 inhibitors—demonstrating significant mortality reduction when initiated simultaneously and titrated to target doses. [3,4]
The pathophysiology involves initial myocardial injury (commonly from ischaemic heart disease in 60-70% of cases) followed by maladaptive neurohormonal activation, ventricular remodelling, and progressive cardiac dysfunction. [5] Diagnosis requires clinical symptoms/signs plus objective evidence of reduced LVEF (≤40%) and elevated natriuretic peptides (BNP > 35 pg/mL or NT-proBNP > 125 pg/mL). [3] Without treatment, 5-year mortality approaches 50-60%, but optimal GDMT can reduce mortality by up to 60-70% and improve functional capacity significantly. [6,7]
Key Facts
- Definition: LVEF ≤40% with symptoms and/or signs of heart failure plus elevated natriuretic peptides [3]
- Global Burden: Affects 64 million people worldwide; prevalence 1-2% in developed countries, rising to 10-15% over age 70 [1,2]
- Mortality: 5-year mortality approximately 50% without optimal treatment; reduced to 15-25% with GDMT [6,7]
- Aetiology: Ischaemic heart disease (60-70%), non-ischaemic dilated cardiomyopathy (20-30%), valvular disease, hypertension [5]
- Treatment Revolution: Quadruple therapy (ARNI + BB + MRA + SGLT2i) reduces mortality by 60-70% compared to no treatment [3,4]
- Biomarkers: NT-proBNP > 125 pg/mL or BNP > 35 pg/mL diagnostic; higher levels predict worse prognosis [8]
- Device Therapy: ICD reduces sudden death by 23% (RRR); CRT improves mortality by 24% in appropriate candidates [9,10]
- Functional Classification: NYHA Class I-IV validated prognostic tool; each class increase doubles mortality risk [11]
Clinical Pearls
High-Yield Points for Clinical Practice:
- Initiate all four pillars simultaneously: Don't delay SGLT2i or MRA while up-titrating ACEi/ARB and beta-blockers [3,4]
- Titrate to target doses, not symptoms: 50% of mortality benefit lost when medications under-dosed [12]
- SGLT2 inhibitors benefit ALL HFrEF patients: Effective regardless of diabetes, renal function (eGFR ≥20), or baseline NYHA class [13,14]
- Iron deficiency affects 50% of patients: Treat if ferritin less than 100 ng/mL OR ferritin 100-299 with TSAT less than 20% using IV iron (improves QoL and reduces hospitalisation) [15]
- ARNI superior to ACEi: Sacubitril/valsartan reduces CV death/HF hospitalisation by 20% vs enalapril; consider first-line if no contraindications [16]
- Early device referral: Assess ICD/CRT eligibility after 3 months optimal GDMT if LVEF remains ≤35% [9,10]
- BNP-guided therapy: NT-proBNP reduction > 30% during titration predicts better outcomes [17]
- Avoid NSAIDs and glitazones: NSAIDs increase HF hospitalisation 2-fold; thiazolidinediones contraindicated [3]
Why This Matters Clinically
HFrEF is the leading cause of hospital admissions in patients over 65 years, accounting for over 1 million hospitalisations annually in the United States alone with healthcare costs exceeding $30 billion. [1,18] Despite advances in therapy, only 30-40% of eligible patients receive optimal GDMT doses, representing a major implementation gap. [12] Early diagnosis and prompt initiation of all four pillars dramatically improves survival and quality of life, with treated patients often experiencing functional improvement and even LVEF recovery (HFimpEF). [19]
The condition requires lifelong management with regular monitoring (clinical assessment, natriuretic peptides, renal function, electrolytes) and systematic medication optimisation. Understanding the evidence base for GDMT is essential for all physicians, as heart failure presentations occur across emergency medicine, general practice, geriatrics, and all medical specialties. Failure to recognise and treat HFrEF appropriately results in preventable mortality, morbidity, and healthcare expenditure.
2. Epidemiology
Incidence and Prevalence
| Metric | Value | Source | Notes |
|---|---|---|---|
| Global Prevalence | 64 million | [1] | Approximately 50% are HFrEF |
| Developed Countries | 1-2% of adults | [2] | Increases exponentially with age |
| Age over 70 | 10-15% | [2] | Peak prevalence group |
| Annual Incidence | 1-9 per 1000 | [2] | Varies by population and age |
| Lifetime Risk (55 years) | 33% men, 28% women | [2] | 1 in 3 will develop HF |
| HFrEF Proportion | 50% of HF cases | [1,3] | Equal to HFpEF in epidemiological studies |
Demographics
- Age: Mean age at diagnosis 70-75 years; incidence doubles with each decade over 45 [2]
- Sex: Slightly more common in males (55-60%); women more likely to have HFpEF [2]
- Ethnicity:
- "African Americans: 1.5× higher incidence, younger presentation (median 65 vs 72 years), worse outcomes [20]"
- "Hispanic populations: Intermediate risk between African American and Caucasian"
- "Genetic predisposition: TTN truncation variants in 15-25% of non-ischaemic DCM [5]"
- Geography: Higher prevalence in regions with high ischaemic heart disease burden (Eastern Europe, South Asia)
Risk Factors
Non-Modifiable:
- Age over 65 years (strongest risk factor) [2]
- Male sex (1.5× risk vs females for HFrEF) [2]
- Family history of cardiomyopathy (3-4× risk if first-degree relative) [5]
- Genetic mutations: TTN (titin, 15-25%), LMNA (lamin A/C, 5-10%), MYH7 (myosin heavy chain, 5%) [5]
Modifiable:
- Coronary artery disease (60-70% of cases): Prior MI strongest predictor [5]
- Hypertension: Long-standing HTN (especially systolic > 160 mmHg) increases risk 2-3× [2]
- Diabetes mellitus: 2-4× increased risk; earlier onset and worse prognosis [2]
- Obesity: BMI > 30 kg/m² increases risk 1.5-2× [2]
- Smoking: Current smoking doubles HF risk [2]
- Excessive alcohol: > 14 units/week associated with alcoholic cardiomyopathy [3]
- Cardiotoxic chemotherapy: Anthracyclines (dose-dependent), trastuzumab, tyrosine kinase inhibitors [21]
- Valvular heart disease: Especially severe aortic stenosis/regurgitation, mitral regurgitation [3]
Geographic and Temporal Variation
- High-income countries: Predominantly ischaemic aetiology (60-70%); declining incidence due to improved CAD management [1]
- Low/middle-income countries: Higher rates of rheumatic heart disease, Chagas disease, peripartum cardiomyopathy [1]
- Sub-Saharan Africa: Increased hypertensive heart disease and peripartum cardiomyopathy [1]
- Temporal trends: HFrEF incidence stable or declining; HFpEF increasing (ageing population, better HFrEF treatment) [2]
3. Pathophysiology
Mechanism Overview
HFrEF develops through a progressive cascade of events following initial myocardial injury, characterised by neurohormonal activation and maladaptive remodelling:
Stage 1: Initial Cardiac Injury
- Myocardial infarction: Loss of functional myocardium; scar formation prevents effective contraction
- Cardiomyopathy: Genetic mutations (sarcomeric proteins, cytoskeleton) or acquired (viral myocarditis, toxins)
- Chronic pressure/volume overload: Hypertension, valvular disease lead to sustained increased afterload/preload
- Cardiotoxic insults: Anthracyclines, alcohol, cocaine cause direct myocyte damage
Stage 2: Compensatory Mechanisms (Initially Beneficial)
Frank-Starling Mechanism:
- Increased preload (venous return) stretches myocardial fibres
- Optimises actin-myosin crossbridge formation
- Maintains stroke volume initially despite reduced contractility
Neurohormonal Activation:
- RAAS Activation: [5]
- Reduced renal perfusion → renin release
- Angiotensin II → vasoconstriction (increases afterload) + aldosterone secretion
- Aldosterone → salt/water retention (increases preload)
- Sympathetic Nervous System: [5]
- Baroreceptor-mediated catecholamine release
- Norepinephrine → increased heart rate (chronotropy) and contractility (inotropy)
- Short-term maintains cardiac output
Cardiac Hypertrophy:
- Increased wall stress triggers myocyte hypertrophy
- Attempts to normalise wall stress (Laplace's Law: wall stress ∝ pressure × radius / thickness)
Stage 3: Maladaptive Remodelling
Ventricular Remodelling: [5]
- Progressive LV dilation (increased end-diastolic and end-systolic volumes)
- Change from elliptical to spherical geometry (reduces ejection efficiency)
- Eccentric hypertrophy (sarcomeres added in series)
- Increased wall stress despite hypertrophy
Cellular Level Changes:
- Myocyte apoptosis and necrosis (programmed cell death pathways activated)
- Interstitial fibrosis (collagen deposition replaces functional tissue)
- Altered calcium handling (reduced SERCA2a expression → impaired relaxation)
- Mitochondrial dysfunction (energy depletion, increased ROS production)
Functional Consequences:
- Progressive reduction in LVEF (stroke volume declines relative to end-diastolic volume)
- Functional mitral regurgitation (annular dilation, papillary muscle displacement)
- Elevated filling pressures (reduced compliance, diastolic dysfunction)
- Reduced cardiac output (forward failure) and congestion (backward failure)
Stage 4: Clinical Heart Failure
- Symptoms of low output (fatigue, reduced exercise tolerance) and congestion (dyspnoea, oedema)
- Neurohormonal activation becomes chronically deleterious (vasoconstriction worsens afterload, fluid retention worsens congestion)
- Risk of arrhythmias (structural remodelling creates re-entry circuits; electrolyte disturbances; increased automaticity)
- Risk of sudden cardiac death (ventricular arrhythmias in 40-50% of HFrEF deaths) [9]
Key Molecular Pathways
| Pathway | Mediators | Pathological Effects | GDMT Target |
|---|---|---|---|
| RAAS Activation | Angiotensin II, Aldosterone | Vasoconstriction, sodium/water retention, myocardial fibrosis, vascular remodelling | ACEi/ARB/ARNI, MRA [3,16,22] |
| Sympathetic Activation | Norepinephrine, Epinephrine | Tachycardia, increased oxygen demand, β-receptor downregulation, arrhythmias | Beta-blockers [23] |
| Natriuretic Peptides | BNP, ANP, CNP | Counter-regulatory (natriuresis, vasodilation) but overwhelmed | ARNI (neprilysin inhibition) [16] |
| Inflammation | IL-1, IL-6, TNF-α | Myocardial fibrosis, cachexia, endothelial dysfunction | (No specific therapy) |
| Oxidative Stress | Reactive oxygen species | Myocyte dysfunction, apoptosis, mitochondrial damage | (Addressed indirectly by GDMT) |
| Endothelin System | Endothelin-1 | Vasoconstriction, myocardial hypertrophy | (Endothelin antagonists not proven beneficial) |
GDMT Mechanisms of Action
Understanding how each pillar interrupts pathophysiology: [3,4]
- ACEi/ARB/ARNI: Block RAAS → reduce afterload, reverse remodelling, reduce fibrosis, improve survival [16,22]
- Beta-blockers: Block sympathetic drive → reduce heart rate, reduce oxygen demand, reverse remodelling, anti-arrhythmic [23]
- MRA: Block aldosterone → reduce sodium/water retention, reduce myocardial fibrosis, reduce sudden death [22]
- SGLT2 inhibitors: Multiple mechanisms → improved myocardial energetics, reduced sodium reabsorption, anti-inflammatory, anti-fibrotic [13,14]
4. Clinical Presentation
Typical Presentation
Cardinal Symptoms:
- Dyspnoea: [3,11]
- "Initially exertional (NYHA II: ordinary activity limited)"
- "Progressive to rest (NYHA IV: symptoms at rest)"
- "Mechanism: Pulmonary congestion → reduced lung compliance → increased work of breathing"
- Orthopnoea: Breathlessness when lying flat (use multiple pillows)
- "Mechanism: Redistribution of lower limb oedema to central circulation → increased pulmonary capillary pressure"
- Paroxysmal Nocturnal Dyspnoea (PND): Waking gasping for breath after 1-2 hours sleep
- "Mechanism: Delayed pulmonary oedema accumulation while supine"
- Fatigue and Exercise Intolerance: Disproportionate tiredness, muscle weakness
- "Mechanism: Reduced cardiac output → skeletal muscle hypoperfusion, chronotropic incompetence"
- Fluid Retention: Peripheral oedema (ankles), weight gain (> 2 kg in 3 days)
- "Mechanism: RAAS activation → sodium/water retention; elevated venous pressures"
- Nocturia: Frequent nighttime urination
- "Mechanism: Supine position → improved renal perfusion → diuresis of accumulated daytime oedema"
Cardinal Signs:
- Elevated Jugular Venous Pressure (JVP): > 8 cm from sternal angle at 45° [3]
- Indicates elevated right atrial pressure and fluid overload
- Sensitivity 70%, Specificity 79% for elevated filling pressures [24]
- Displaced Apex Beat: Laterally (>mid-clavicular line) and inferiorly
- Indicates LV dilation; palpable in 5th/6th intercostal space
- Third Heart Sound (S3 Gallop): Low-pitched sound in early diastole
- "Mechanism: Rapid ventricular filling into non-compliant ventricle"
- Highly specific (> 90%) but insensitive (~30%) for HFrEF [24]
- Pulmonary Crackles: Bilateral fine inspiratory crackles at lung bases
- Indicates pulmonary oedema; moderate sensitivity/specificity (~60-70%) [24]
- Peripheral Oedema: Pitting oedema ankles (ambulatory) or sacrum (bed-bound)
- Bilateral distribution; unilateral suggests venous thrombosis
- Hepatomegaly: Tender, smooth hepatomegaly with hepatojugular reflux
- Indicates hepatic congestion; may progress to cardiac cirrhosis
NYHA Functional Classification
The New York Heart Association (NYHA) functional classification is a validated, widely used prognostic tool correlating symptoms with activity level: [11]
| Class | Description | Specific Activity Examples | Prognosis |
|---|---|---|---|
| I | No limitation of physical activity | Can climb multiple flights of stairs, run, perform vigorous exercise without symptoms | Excellent; 1-year mortality less than 5% [11] |
| II | Slight limitation of physical activity | Comfortable at rest and mild activity; ordinary activity (walking 2 blocks, climbing 1 flight) causes dyspnoea/fatigue | Good; 1-year mortality 5-10% [11] |
| III | Marked limitation of physical activity | Comfortable at rest; less than ordinary activity (walking less than 1 block, dressing) causes symptoms | Moderate; 1-year mortality 10-20% [11] |
| IV | Unable to carry out physical activity without discomfort | Symptoms at rest; any physical activity increases discomfort | Poor; 1-year mortality 20-40% [11] |
Clinical Application:
- NYHA class predicts mortality independently of LVEF [11]
- Used to guide device therapy (CRT indicated for NYHA II-IV ambulatory) [10]
- Each class increase approximately doubles mortality risk [11]
- Should be assessed at every follow-up visit
- Limitation: Subjective; inter-rater variability
Atypical Presentations
Elderly Patients (> 75 years):
- Confusion or cognitive decline: May be primary presentation due to cerebral hypoperfusion [2]
- Falls or functional decline: Weakness, deconditioning, postural hypotension
- Anorexia and cachexia: Cardiac cachexia (> 5% weight loss); poor prognostic sign
- Less pronounced dyspnoea: Reduced baseline activity masks exertional symptoms
- Delirium during decompensation: Acute confusional state
Young Adults (less than 50 years):
- Consider genetic/familial cardiomyopathy: Dilated cardiomyopathy, hypertrophic cardiomyopathy, arrhythmogenic right ventricular cardiomyopathy [5]
- Screen family members: First-degree relatives if genetic aetiology suspected
- Peripartum cardiomyopathy: Women presenting in last month of pregnancy or within 5 months postpartum; LVEF typically less than 45% [3]
Right Heart Failure Predominance:
- Elevated JVP, hepatomegaly, ascites, peripheral oedema with minimal pulmonary symptoms
- Consider: Pulmonary hypertension, RV infarction, congenital heart disease, tricuspid regurgitation
Red Flags and Emergencies
URGENT ASSESSMENT REQUIRED:
- Cardiogenic Shock: Hypotension (SBP less than 90 mmHg), cold/clammy extremities, altered mental status, oliguria less than 0.5 mL/kg/hr [3]
- Mortality > 40% without mechanical circulatory support; requires CCU/ICU
- Acute Pulmonary Oedema: Severe dyspnoea, tachypnoea > 30/min, SpO2 less than 90%, pink frothy sputum, bilateral crackles [3]
- Immediate intervention: Sit upright, oxygen, IV diuretics, consider NIV/CPAP
- Ventricular Arrhythmias: Sustained VT (> 30 seconds), VF, syncope, ICD shocks
- Risk of sudden cardiac death; requires telemetry, electrolyte correction, anti-arrhythmics
- Acute Coronary Syndrome: New chest pain, dynamic ECG changes, troponin rise
- Common precipitant of decompensation; consider urgent revascularisation
- Syncope: Loss of consciousness without warning
- High risk of sudden death; urgent arrhythmia evaluation, consider ICD
- Rapid AF with RVR: Heart rate > 130 bpm, haemodynamic compromise
- Rate control (beta-blocker, digoxin) or DCCV if unstable
5. Clinical Examination
Structured Examination Approach
A systematic cardiovascular examination is essential for assessing volume status, cardiac output, and identifying complications:
1. General Inspection
- Level of distress: Breathlessness at rest, use of accessory muscles, inability to speak in full sentences (indicates NYHA IV)
- Body habitus:
- Cachexia (> 5% unintentional weight loss) suggests advanced HF; poor prognostic sign [3]
- Obesity (consider HFpEF differential)
- Pallor: Anaemia present in 30-50% of HFrEF patients [15]
- Cyanosis: Central (perioral) indicates severe hypoxia
- Jaundice: Congestive hepatopathy (cardiac cirrhosis)
- Respiratory rate: Normal less than 20/min; > 25/min suggests decompensation
2. Vital Signs
- Blood Pressure:
- "Hypotension (SBP less than 90 mmHg): Advanced disease, cardiogenic shock, or over-diuresis"
- "Hypertension: Inadequate RAAS inhibition; risk factor for decompensation"
- "Narrow pulse pressure (less than 25 mmHg): Low cardiac output state"
- Heart Rate:
- "Tachycardia (> 100 bpm): Inadequate beta-blockade, anaemia, hyperthyroidism, or decompensation"
- "Bradycardia (less than 50 bpm): Excessive beta-blockade or conduction disease"
- "Irregularly irregular: Atrial fibrillation (present in 30-40% of HFrEF) [3]"
- Respiratory Rate: Elevated (> 20/min) in pulmonary congestion
- Oxygen Saturation: less than 92% on room air indicates significant congestion or concurrent lung disease
- Temperature: Fever suggests infection as precipitant
3. Cardiovascular Examination
Jugular Venous Pressure (JVP): [24]
- Technique: Patient at 45°, head turned left, measure vertical height from sternal angle
- Normal: less than 3 cm above sternal angle (~8 cm total height from right atrium)
- Elevated (> 8 cm): Indicates fluid overload, elevated right atrial pressure
- Waveform analysis:
- "Prominent V wave: Tricuspid regurgitation"
- "Kussmaul's sign (rise with inspiration): Constrictive pericarditis, restrictive cardiomyopathy"
Apex Beat:
- Position: Normally 5th intercostal space, mid-clavicular line
- Displaced: Laterally (>mid-clavicular) and inferiorly (6th/7th space) indicates LV dilation
- Character: Heaving (pressure overload), thrusting (volume overload), diffuse (dilated cardiomyopathy)
Heart Sounds:
- S1: May be soft (reduced contractility)
- S2: Widely split (LBBB); loud P2 (pulmonary hypertension)
- S3 Gallop: Pathognomonic of ventricular volume overload; best heard at apex with bell in left lateral position
- Specificity > 90% for elevated LVEDP [24]
- S4: Atrial kick into stiff ventricle (diastolic dysfunction component)
- Murmurs:
- "Pansystolic murmur at apex → lateral: Functional mitral regurgitation (present in 50% of moderate-severe HFrEF)"
- "Aortic stenosis murmur: May be primary cause or concurrent"
- "Tricuspid regurgitation: Pansystolic murmur at left sternal edge, louder with inspiration"
Peripheral Pulses:
- Carotid pulse: Small volume, slow rising (pulsus parvus et tardus if AS concurrent)
- Peripheral pulses: Weak, thready in low output states; assess for peripheral vascular disease
4. Respiratory Examination
- Tracheal position: Midline (deviated if large pleural effusion)
- Chest expansion: Symmetrical; reduced if large effusion
- Percussion:
- Resonant normally
- "Stony dull at bases: Pleural effusions (typically bilateral in HF; unilateral if right-sided suggests other cause)"
- Auscultation:
- "Crackles: Bilateral fine inspiratory crackles at bases (pulmonary oedema)"
- Extend up lung fields in severe congestion
- Wheeze: "Cardiac asthma" (oedematous bronchi); can mimic COPD/asthma
- "Reduced breath sounds: Pleural effusion"
- "Crackles: Bilateral fine inspiratory crackles at bases (pulmonary oedema)"
5. Abdominal Examination
- Hepatomegaly:
- Smooth, tender edge (acute congestion)
- Firm, irregular edge (cardiac cirrhosis if chronic)
- "Pulsatile liver: Severe tricuspid regurgitation"
- Ascites:
- Shifting dullness, fluid thrill
- Indicates advanced right heart failure, very high JVP
- Splenomegaly: Rare; consider concurrent portal hypertension
6. Peripheral Examination
- Peripheral Oedema:
- "Pitting oedema: Press for 5 seconds; skin indentation persists"
- "Distribution: Ankles/feet (ambulatory), sacrum/buttocks (bed-bound)"
- "Grading: Mild (ankles), Moderate (below knees), Severe (thighs/abdominal wall)"
- "Note: Absence doesn't exclude HF; CCB use can cause oedema independently"
- Peripheral Perfusion:
- "Temperature: Cool peripheries (vasoconstriction, low output)"
- "Capillary Refill: > 2 seconds abnormal (press nail bed 5 sec, measure refill time)"
- "Colour: Mottled, cyanotic in severe low output"
Special Tests and Manoeuvres
| Test | Technique | Positive Finding | Sensitivity/Specificity | Clinical Significance |
|---|---|---|---|---|
| Hepatojugular Reflux | Press firmly on RUQ for 15 seconds while observing JVP | Sustained JVP rise > 3 cm | 70% / 85% for elevated RA pressure [24] | Confirms elevated right-sided filling pressures |
| Abdominojugular Test | Alternative to hepatojugular; press periumbilical region | Sustained JVP rise > 3 cm | Similar to hepatojugular [24] | Easier to perform than hepatojugular |
| Valsalva Manoeuvre | Forced expiration against closed glottis for 15 sec, observe BP response | "Square wave" response (no phase IV overshoot) | High specificity for elevated LVEDP | Indicates reduced SV reserve; abnormal response specific for HF |
| Bendopnea Test | Ask patient to bend forward at waist as if tying shoelaces | Breathlessness within 30 seconds | Correlates with elevated PCWP [25] | Simple bedside test for congestion |
| Orthostatic Vitals | BP/HR supine and standing (after 3 min) | Drop > 20/10 mmHg or rise HR > 30 bpm | N/A | Assesses for over-diuresis or autonomic dysfunction |
6. Investigations
First-Line Investigations
A comprehensive initial workup is essential for diagnosis, aetiology determination, and baseline assessment before GDMT initiation:
| Investigation | Expected Findings in HFrEF | Clinical Use | Evidence |
|---|---|---|---|
| BNP | > 100 pg/mL (diagnostic threshold); > 400 pg/mL suggests severe HF | Diagnosis, prognosis, monitoring response | NPV > 95% if less than 35 pg/mL [8] |
| NT-proBNP | > 125 pg/mL (diagnostic); > 300 pg/mL if acute; > 450 pg/mL if > 50 years | Preferred in chronic HF; not cleared renally | NPV 98% if less than 125 pg/mL [8] |
| 12-Lead ECG | LBBB (QRS ≥120 ms), Q waves (prior MI), LVH (voltage criteria), AF, long QT | Identify aetiology, CRT eligibility, arrhythmias | Abnormal ECG in 90% of HFrEF [3] |
| Troponin (hs-Tn) | Mildly elevated (chronic myocyte turnover); significant rise suggests ACS | Exclude ACS; prognostic (elevated = worse outcome) | Elevated in 30-50% chronic HFrEF [3] |
| Full Blood Count | Anaemia (Hb less than 130 g/L men, less than 120 g/L women) in 30-50% | Identify treatable cause; anaemia worsens HF | Anaemia predicts mortality [15] |
| Urea & Electrolytes | ↑ Urea/creatinine (cardiorenal syndrome), ↓ Na+ (poor prognosis), ↑ K+ (RAAS inhibitors) | Monitor renal function, guide diuretics/RAAS inhibition | eGFR less than 60 in 30-50% at presentation [3] |
| Liver Function Tests | ↑ ALT, ↑ AST, ↑ bilirubin (hepatic congestion), ↑ GGT, ↓ albumin (chronic) | Assess hepatic congestion; exclude cirrhosis | Transaminases 2-3× ULN common in acute congestion |
| Thyroid Function | Hypo- or hyperthyroidism can cause or worsen HF | Screen for treatable cause | TSH abnormal in 5-10% [3] |
| HbA1c | Diabetes in 30-40% of HFrEF | Identify comorbidity; SGLT2i indicated regardless | Diabetes worsens prognosis |
| Lipid Profile | Assess cardiovascular risk; guide statin therapy | Primary/secondary prevention | If ischaemic aetiology |
| Iron Studies | Ferritin less than 100 ng/mL OR ferritin 100-299 with TSAT less than 20% = iron deficiency | IV iron indication (improves QoL, reduces hospitalisation) | Iron deficiency in 50% [15] |
Natriuretic Peptides: Detailed Interpretation
BNP (B-Type Natriuretic Peptide): [8]
- Released from ventricular myocytes in response to wall stretch
- Diagnostic thresholds:
- less than 35 pg/mL: HF very unlikely (NPV 95%)
- 35-100 pg/mL: Grey zone; consider if high pre-test probability
-
100 pg/mL: HF likely (acute presentation > 300 pg/mL)
-
400 pg/mL: Severe HF
- Causes of elevated BNP without HF: Pulmonary embolism, renal failure, atrial fibrillation, advanced age, sepsis
- Causes of falsely low BNP: Obesity (adipocytes clear BNP), flash pulmonary oedema (insufficient time for release)
NT-proBNP (N-Terminal pro-BNP): [8]
- Inactive cleavage product of proBNP; longer half-life than BNP
- Not cleared by neprilysin (use with ARNI therapy)
- Cleared renally (higher levels in renal impairment)
- Age-adjusted thresholds:
- less than 50 years: > 450 pg/mL diagnostic
- 50-75 years: > 900 pg/mL diagnostic
-
75 years: > 1800 pg/mL diagnostic
- "Acute presentation: > 300 pg/mL any age"
Prognostic Value: [17]
- Higher baseline BNP/NT-proBNP predicts worse outcomes (hospitalization, mortality)
- Reduction > 30% during GDMT titration predicts better prognosis
- Persistent elevation despite therapy indicates need for intensification
- Can guide therapy: BNP-guided treatment may reduce HF hospitalisations
Imaging
Transthoracic Echocardiography (ESSENTIAL): [3]
Core Parameters:
- LVEF Quantification: Biplane Simpson's method (gold standard); visual estimation acceptable if suboptimal images
- HFrEF defined as LVEF ≤40%
- "HFmrEF: 41-49%"
- "HFpEF: ≥50%"
- "HFimpEF: Was ≤40%, now > 40%"
- LV Dimensions:
- End-diastolic diameter (normal less than 55 mm)
- End-systolic diameter (normal less than 40 mm)
- LV mass (identify hypertrophy)
- Wall Motion Abnormalities:
- Regional (suggests ischaemic aetiology; e.g., inferolateral hypokinesis from LCx MI)
- Global hypokinesis (dilated cardiomyopathy)
- Valvular Assessment:
- "Aortic stenosis: Peak velocity, mean gradient, valve area (severe if AVA less than 1.0 cm²)"
- "Mitral regurgitation: Functional MR common (annular dilation, leaflet tethering); quantify severity (EROA, regurgitant volume)"
- "Tricuspid regurgitation: Estimate pulmonary artery pressure"
- Diastolic Function:
- E/A ratio, E/e', LA volume index
- Elevated filling pressures even in HFrEF (mixed systolic-diastolic dysfunction)
- Pulmonary Artery Pressure:
- Estimated from TR jet velocity
- Elevated (sPAP > 50 mmHg) indicates worse prognosis
CRT Assessment:
- Evaluate for mechanical dyssynchrony if QRS ≥120 ms (though QRS duration on ECG remains primary criterion) [10]
Chest X-ray: [3]
Typical Findings:
- Cardiomegaly: Cardiothoracic ratio (CTR) > 0.5 (cardiac width/thoracic width on PA film)
- Sensitivity 50%, specificity 80% for LV dilation
- Pulmonary Venous Congestion:
- Upper lobe blood diversion (cephalisation)
- Perihilar haziness
- Kerley B lines (interlobular septal thickening; horizontal lines at periphery)
- Pleural Effusions: Blunting of costophrenic angles; typically bilateral
- Alveolar Oedema: "Bat's wing" appearance (severe acute pulmonary oedema)
Utility:
- Excludes alternative diagnoses (pneumonia, malignancy, pneumothorax)
- Assesses response to diuresis
- Not required for diagnosis if echo available, but often first investigation in acute presentations
Cardiac MRI (Selected Cases): [3]
Indications:
- Precise LVEF measurement when echo suboptimal (especially obesity, COPD)
- Aetiology determination (ischaemic vs non-ischaemic):
- "Late Gadolinium Enhancement (LGE): Subendocardial/transmural pattern = ischaemic; mid-wall = non-ischaemic"
- Suspected infiltrative disease (amyloidosis, sarcoidosis, haemochromatosis)
- Myocarditis suspected (oedema sequences, LGE patterns)
- Viability assessment pre-revascularisation (LGE less than 50% transmural = viable)
Parameters:
- Most accurate LVEF quantification (gold standard)
- Tissue characterisation (T1/T2 mapping, extracellular volume)
- Fibrosis quantification
Coronary Angiography (Invasive): [3]
Indications:
- Suspected ischaemic aetiology (Q waves on ECG, regional wall motion abnormalities, cardiovascular risk factors)
- New-onset HFrEF (70% may have significant CAD)
- Angina symptoms
- Assess revascularisation candidacy
Findings:
- Obstructive CAD (> 70% stenosis or FFR less than 0.80)
- Prior MI (TIMI flow, collaterals)
Revascularisation Evidence:
- Benefit if viable myocardium and suitable anatomy for PCI/CABG
- No routine revascularisation without angina/viability [3]
Cardiopulmonary Exercise Testing (CPET):
Use:
- Objective exercise capacity (peak VO2)
- Advanced HF evaluation (transplant/LVAD candidacy: peak VO2 less than 12-14 mL/kg/min)
- Differentiate cardiac vs pulmonary limitation
Other Investigations:
- 24-hour Holter monitor: Assess arrhythmia burden, heart rate control on beta-blocker
- Sleep study: Screen for obstructive sleep apnoea (common comorbidity; worsens HF)
- Genetic testing: If dilated cardiomyopathy less than 50 years, family history, or syndromic features [5]
Diagnostic Criteria (ESC 2021)
Diagnosis of HFrEF requires ALL of the following: [3]
- Symptoms and/or signs of heart failure (dyspnoea, orthopnoea, oedema, elevated JVP, S3 gallop)
- LVEF ≤40% on echocardiography
- Elevated natriuretic peptides:
- BNP > 35 pg/mL OR
- NT-proBNP > 125 pg/mL
If natriuretic peptides borderline or unavailable, diagnosis can be made with clinical syndrome + LVEF ≤40%, but peptides strongly recommended.
7. Classification and Staging
EF-Based Classification (ESC/AHA)
The European Society of Cardiology and American Heart Association classify heart failure by ejection fraction: [3,4]
| Category | LVEF | Description | Therapeutic Implications |
|---|---|---|---|
| HFrEF | ≤40% | Reduced ejection fraction | Four-pillar GDMT (ARNI/ACEi/ARB + BB + MRA + SGLT2i); ICD/CRT if indicated |
| HFmrEF | 41-49% | Mildly reduced ejection fraction | GDMT beneficial; emerging evidence for SGLT2i, MRA |
| HFpEF | ≥50% | Preserved ejection fraction | SGLT2i, diuretics, BP/AF control; no proven mortality benefit for ACEi/BB/MRA |
| HFimpEF | Was ≤40%, now > 40% | Improved ejection fraction | Continue GDMT (may deteriorate if stopped); better prognosis than persistent HFrEF [19] |
Clinical Notes:
- ~50% of HF patients are HFrEF, 50% are HFpEF/HFmrEF [1,2]
- 20-40% of HFrEF patients improve EF to > 40% with GDMT (HFimpEF) [19]
- HFimpEF should continue GDMT indefinitely (relapse if stopped)
ACC/AHA Stages of Heart Failure
The American College of Cardiology/American Heart Association staging system emphasizes disease progression and prevention: [4]
| Stage | Description | Examples | Management Focus |
|---|---|---|---|
| A | At risk for HF but no structural heart disease, no symptoms | HTN, DM, CAD risk factors, family history of cardiomyopathy, cardiotoxic exposure | Primary prevention: Lifestyle (diet, exercise, smoking cessation), treat HTN/DM/hyperlipidaemia, statins, ACEi if CAD/DM [4] |
| B | Structural heart disease but no symptoms | Prior MI, LVH, asymptomatic valvular disease, LVEF ≤40% without symptoms | Secondary prevention: ACEi/ARB/ARNI + beta-blocker even if asymptomatic, statins, consider ICD if LVEF ≤35% [4] |
| C | Structural disease with current or prior symptoms of HF | LVEF ≤40% with dyspnoea, fatigue, oedema (current or past) | GDMT: Four pillars, titrate to target doses, ICD/CRT if indicated, sodium restriction, monitoring [3,4] |
| D | Advanced HF with severe symptoms despite maximal therapy | NYHA IV on GDMT, recurrent hospitalisations, inotrope-dependent, cardiogenic shock | Advanced therapies: LVAD evaluation, heart transplant assessment, palliative care, specialist HF unit [3,4] |
Key Point: Stages are progressive (A→B→C→D); cannot move backwards. Goals are to prevent progression and optimize stage-specific management.
INTERMACS Profiles (Advanced HF)
The Interagency Registry for Mechanically Assisted Circulatory Support profiles classify severity of advanced HF to guide timing of mechanical circulatory support: [4]
| Profile | Description | Clinical Scenario | Urgency | Typical Management |
|---|---|---|---|---|
| 1 - Cardiogenic Shock | "Crash and burn" | Critical hypoperfusion despite escalating inotropes/vasopressors; often mechanical ventilation | Immediate (less than 24 hours) | Emergent LVAD/transplant/ECMO |
| 2 - Progressive Decline | "Sliding on inotropes" | Declining on continuous IV inotropes; cannot wean | Urgent (within days) | Urgent LVAD/transplant |
| 3 - Stable Inotrope-Dependent | "Dependent stability" | Stable on low-dose inotropes but cannot wean | Semi-urgent (weeks) | LVAD bridge to transplant |
| 4 - Resting Symptoms | "Frequent flyer" | At home but frequent HF symptoms at rest; recurrent hospitalisations | Elective (months) | Optimization, consider LVAD/transplant |
| 5 - Exertion Intolerant | "Housebound" | NYHA IV; cannot perform ADLs without symptoms | Elective | Optimization, consider advanced therapies |
| 6 - Exertion Limited | "Walking wounded" | NYHA III; comfortable at rest but minimal activity causes symptoms | Variable | Outpatient optimization |
| 7 - Advanced NYHA III | "Placeholder" | NYHA III symptoms, stable on oral therapy | Not immediate | Continued outpatient GDMT |
Use in Practice:
- INTERMACS 1-3: Urgent evaluation for mechanical support
- INTERMACS 4-7: Careful optimization of GDMT; consider advanced therapies if no improvement
- Too sick (INTERMACS 1 with multi-organ failure) or too well (INTERMACS 5-7 with good QoL) may not benefit from LVAD
8. Management
Overview: The Four Pillars of GDMT
Modern HFrEF management is based on four pillars of guideline-directed medical therapy (GDMT), each independently reducing mortality and hospitalisation. All four should be initiated simultaneously or in rapid succession (within weeks) and titrated to target doses. [3,4,12]
CRITICAL PRINCIPLE: Initiate all four pillars early and titrate to target doses. 50% of mortality benefit is lost when medications are under-dosed. Do not wait months between adding therapies. [12]
Pillar 1: Renin-Angiotensin System Inhibition
Mechanism: Block RAAS → reduce angiotensin II-mediated vasoconstriction and aldosterone-mediated sodium retention → reduce afterload, reverse ventricular remodelling, reduce myocardial fibrosis. [16,22]
1A. Angiotensin Receptor-Neprilysin Inhibitor (ARNI) - FIRST-LINE
| Drug | Starting Dose | Target Dose | Monitoring | Evidence |
|---|---|---|---|---|
| Sacubitril/Valsartan | 49/51 mg BD | 97/103 mg BD | BP, K+, eGFR at 1-2 weeks, then 3-monthly | PARADIGM-HF: 20% ↓ CV death/HF hosp vs enalapril [16] |
PARADIGM-HF Trial (2014): [16]
- 8,442 patients, LVEF ≤40%, NYHA II-IV
- Primary outcome: 20% relative risk reduction in cardiovascular death or HF hospitalisation (21.8% vs 26.5%; HR 0.80, pless than 0.001)
- 16% reduction in all-cause mortality
- NNT = 21 over 27 months to prevent 1 event
Practical Points:
- Preferred over ACEi/ARB unless contraindications (pregnancy, angioedema history) [3]
- Washout period: Stop ACEi for 36 hours before starting ARNI (risk of angioedema if overlapping)
- Contraindications: History of angioedema, pregnancy, bilateral renal artery stenosis, severe aortic stenosis
- Titration: Start 49/51 mg BD; uptitrate every 2-4 weeks to 97/103 mg BD if tolerated (SBP > 95 mmHg, K+ less than 5.5 mmol/L, eGFR stable)
- If intolerant (hypotension, hyperkalaemia), downtitrate rather than stop
1B. ACE Inhibitors (Alternative if ARNI not tolerated/available)
| Drug | Starting Dose | Target Dose | Notes |
|---|---|---|---|
| Ramipril | 1.25-2.5 mg OD | 10 mg OD | Evidence from AIRE, TRACE |
| Enalapril | 2.5 mg BD | 10 mg BD | Used in SOLVD, comparator in PARADIGM-HF |
| Lisinopril | 2.5 mg OD | 20-40 mg OD | Once daily dosing |
| Perindopril | 2 mg OD | 8-16 mg OD | Once daily dosing |
Evidence: [22]
- Meta-analysis of ACEi in HFrEF: 23% reduction in mortality, 35% reduction in HF hospitalisation
- SOLVD (1991): Enalapril reduced mortality 16% vs placebo in LVEF less than 35%
- Mechanism: Block ACE → reduce angiotensin II and aldosterone → afterload reduction, reverse remodelling
Practical Points:
- Start low (especially if SBP less than 100 mmHg or elderly); uptitrate every 1-2 weeks
- Monitor K+ and creatinine at 1-2 weeks (acceptable for creatinine to rise less than 30% and K+ less than 5.5 mmol/L)
- Common side effect: Dry cough (10-15%); switch to ARB if troublesome
- Do not combine ACEi + ARB (increased risk hyperkalaemia, AKI, no added benefit)
1C. Angiotensin Receptor Blockers (If ACEi-intolerant)
| Drug | Starting Dose | Target Dose | Notes |
|---|---|---|---|
| Candesartan | 4-8 mg OD | 32 mg OD | CHARM trial |
| Valsartan | 40 mg BD | 160 mg BD | Val-HeFT trial |
| Losartan | 25 mg OD | 100 mg OD | Less evidence in HFrEF |
Evidence: [22]
- CHARM-Alternative: Candesartan vs placebo in ACEi-intolerant patients; 23% reduction in CV death/HF hospitalisation
- Non-inferior to ACEi but no cough (mechanism: don't inhibit bradykinin degradation)
Practical Points:
- Use only if ACEi-intolerant (cough, angioedema) or as valsartan component of ARNI
- Same monitoring as ACEi (BP, K+, creatinine)
Pillar 2: Beta-Blockers
Mechanism: Block sympathetic activation → reduce heart rate (increased diastolic filling time, reduced oxygen demand) → reverse remodelling, anti-arrhythmic, mortality reduction. [23]
| Drug | Starting Dose | Target Dose | Titration Schedule | Evidence |
|---|---|---|---|---|
| Bisoprolol | 1.25 mg OD | 10 mg OD | Double every 2 weeks | CIBIS-II: 34% ↓ mortality [23] |
| Carvedilol | 3.125 mg BD | 25 mg BD (50 mg BD if > 85 kg) | Double every 2 weeks | COPERNICUS: 35% ↓ mortality [23] |
| Metoprolol Succinate | 12.5-25 mg OD | 200 mg OD | Double every 2 weeks | MERIT-HF: 34% ↓ mortality [23] |
| Nebivolol | 1.25 mg OD | 10 mg OD | Double every 2 weeks | SENIORS (elderly > 70): ↓ death/hosp [23] |
Key Trials:
- CIBIS-II (1999): Bisoprolol vs placebo; 34% mortality reduction (HR 0.66) [23]
- MERIT-HF (1999): Metoprolol succinate; 34% mortality reduction, 41% reduction in sudden death [23]
- COPERNICUS (2002): Carvedilol in severe HF (LVEF less than 25%); 35% mortality reduction [23]
Practical Points:
- Initiate during compensated state: Do NOT start in acute decompensation (wait until euvolaemic)
- Uptitrate slowly: Double dose every 2 weeks if HR > 50 bpm, SBP > 90 mmHg, no worsening HF symptoms
- Target heart rate: 50-60 bpm at rest (resting HR > 70 bpm associated with worse outcomes)
- Transient worsening: Patients may feel worse initially (fatigue, fluid retention); reassure and continue if tolerated
- Do NOT stop abruptly: Rebound tachycardia, increased mortality risk
- Special case - Acute HF: Continue beta-blocker through acute decompensation if possible (unless cardiogenic shock); reduce dose if necessary but avoid stopping [3]
Pillar 3: Mineralocorticoid Receptor Antagonists (MRA)
Mechanism: Block aldosterone receptor → reduce sodium/water retention, reduce myocardial and vascular fibrosis, reduce potassium wasting, anti-arrhythmic (reduce sudden death). [22]
| Drug | Dose | Contraindications | Monitoring | Evidence |
|---|---|---|---|---|
| Spironolactone | 25 mg OD (may increase to 50 mg OD) | K+ > 5.0 mmol/L, eGFR less than 30 mL/min/1.73m², concurrent K+-sparing diuretics | K+, creatinine at 1 week, 4 weeks, 3-monthly | RALES: 30% ↓ mortality [22] |
| Eplerenone | 25 mg OD (may increase to 50 mg OD) | Same as spironolactone | Same as spironolactone | EMPHASIS-HF: 37% ↓ CV death/HF hosp [22] |
Key Trials:
- RALES (1999): Spironolactone 25 mg vs placebo in severe HF (NYHA III-IV); 30% mortality reduction (HR 0.70, pless than 0.001) [22]
- EMPHASIS-HF (2011): Eplerenone in NYHA II; 37% reduction in CV death/HF hospitalisation [22]
Practical Points:
- Essential in all patients unless contraindicated (high K+, severe renal impairment)
- Hyperkalaemia risk: Monitor K+ at 1 week and 4 weeks after initiation/dose change
- "If K+ 5.0-5.5: Reduce dose to 25 mg alternate days"
- "If K+ > 5.5: Stop MRA, recheck K+ in 3-5 days, restart at lower dose when less than 5.0"
- Eplerenone advantages: Selective MRA (less gynaecomastia, erectile dysfunction than spironolactone)
- Gynaecomastia: Occurs in 10% on spironolactone; switch to eplerenone
- Do not co-prescribe with potassium supplements unless recurrent hypokalaemia on diuretics
Pillar 4: SGLT2 Inhibitors
Mechanism: Multiple mechanisms → improved myocardial energetics (shift from glucose to ketone metabolism), osmotic diuresis (mild natriuretic effect), reduced inflammation and fibrosis, improved endothelial function, reduced sympathetic activation. [13,14]
| Drug | Dose | Key Points | Evidence |
|---|---|---|---|
| Dapagliflozin | 10 mg OD | Benefit regardless of diabetes status; safe to eGFR ≥25 | DAPA-HF: 26% ↓ CV death/HF worsening [13] |
| Empagliflozin | 10 mg OD | Benefit regardless of diabetes status; safe to eGFR ≥20 | EMPEROR-Reduced: 25% ↓ CV death/HF hosp [14] |
DAPA-HF Trial (2019): [13]
- 4,744 patients, LVEF ≤40%, NYHA II-IV, with or without diabetes
- Primary outcome: 26% reduction in CV death or worsening HF (16.3% vs 21.2%; HR 0.74, pless than 0.001)
- 18% reduction in CV death, 30% reduction in HF hospitalisation
- NNT = 21 over 18 months to prevent 1 primary outcome event
- Benefit consistent regardless of diabetes, baseline LVEF, NYHA class, or background therapy
EMPEROR-Reduced Trial (2020): [14]
- 3,730 patients, LVEF ≤40%
- Primary outcome: 25% reduction in CV death or HF hospitalisation (19.4% vs 24.7%; HR 0.75, pless than 0.001)
- Benefit extended to eGFR ≥20 mL/min/1.73m²
Practical Points:
- Add immediately: Do not delay for other GDMT; can add at any stage
- No diabetes required: Benefit independent of glycaemic status
- Minimal side effects: Well tolerated; rare genital infections (2-3%), volume depletion if over-diuresed
- Euglycaemic DKA: Very rare in HFrEF (more common in T1DM); ensure adequate hydration
- Renal function: Safe to eGFR ≥20-25; may continue even if eGFR declines below initiation threshold
Additional Medical Therapies
Diuretics (Symptom Control, NOT Mortality-Reducing): [3]
Loop Diuretics:
| Drug | Starting Dose | Maintenance Dose | Notes |
|---|---|---|---|
| Furosemide | 40 mg OD | 40-240 mg daily (divided doses if > 80 mg) | Short-acting; bioavailability 50% |
| Bumetanide | 1 mg OD | 1-5 mg daily | 40× more potent than furosemide |
| Torsemide | 10 mg OD | 10-100 mg daily | Better bioavailability (80%) |
Practical Points:
- Titrate to euvolaemia: Adjust dose to achieve dry weight (no oedema, no dyspnoea, flat JVP)
- Monitor closely: Daily weights at home (alert if > 2 kg gain in 3 days); flexible dosing ("traffic light" system: increase diuretic if weight rising)
- Diuretic resistance: If inadequate response, consider:
- Increase dose (max furosemide 240 mg BD)
- Add thiazide (metolazone 2.5-5 mg; sequential nephron blockade)
- IV diuretics if oral absorption impaired (gut oedema)
- Hypokalaemia: Common; monitor K+; MRA helps prevent (potassium-sparing effect)
- Over-diuresis: Causes hypotension, AKI, worsens renal function; balance is key
Ivabradine (If Channel Blocker): [3]
- Indication: Sinus rhythm, LVEF ≤35%, NYHA II-IV, resting HR ≥70 bpm despite maximized beta-blocker (or beta-blocker intolerant)
- Dose: 5 mg BD; increase to 7.5 mg BD if HR remains > 70 bpm
- SHIFT Trial (2010): 18% reduction in CV death/HF hospitalisation; driven by reduced HF hospitalisations
- Mechanism: Selective If channel inhibition in SA node → HR reduction without negative inotropy
- Side effects: Luminous visual phenomena (10-15%, transient), bradycardia
- Practical: Combination with beta-blocker superior to either alone for HR control
Hydralazine + Isosorbide Dinitrate (H-ISDN):
- Indication:
- "RAAS inhibitor intolerant (all classes: ACEi, ARB, ARNI contraindicated/intolerant)"
- African American patients as add-on to GDMT (evidence from A-HeFT)
- Dose: Hydralazine 37.5 mg TDS (target 75 mg TDS) + Isosorbide dinitrate 20 mg TDS (target 40 mg TDS)
- A-HeFT Trial (2004): In self-identified African Americans, addition of H-ISDN to standard therapy reduced mortality 43%
- Mechanism: Hydralazine (arterial vasodilator) + ISDN (venous and arterial vasodilator) → afterload/preload reduction
- Limitations: TDS dosing (compliance), headache, hypotension
Digoxin:
- Indication:
- Atrial fibrillation with rapid ventricular rate (rate control)
- Persistent HF symptoms despite GDMT in sinus rhythm (may reduce hospitalisation; no mortality benefit)
- Dose: 62.5-250 mcg OD (lower dose in elderly, renal impairment)
- DIG Trial (1997): No mortality benefit; 28% reduction in HF hospitalisations
- Caution: Narrow therapeutic window; toxicity (arrhythmias, GI symptoms, visual disturbances); check levels if toxicity suspected (target 0.5-0.9 ng/mL)
Vericiguat (Soluble Guanylate Cyclase Stimulator):
- Indication: Worsening HF (hospitalisation or IV diuretics in past 6 months) despite GDMT
- Dose: 2.5 mg OD; increase to 5 mg, then 10 mg (target) at 2-week intervals
- VICTORIA Trial (2020): 10% relative risk reduction in CV death/HF hospitalisation (modest benefit)
- Practical: Reserved for high-risk patients with recent decompensation; expensive; not first-line
Device Therapy
Device therapy is critical for selected HFrEF patients to prevent sudden cardiac death and improve contractility:
Implantable Cardioverter-Defibrillator (ICD)
Indication (Primary Prevention): [9]
- LVEF ≤35% despite ≥3 months optimal GDMT
- NYHA Class II-III (ambulatory)
- Life expectancy > 1 year with good functional status
Contraindications:
- NYHA IV with no plan for advanced therapies
- Recent MI (less than 40 days; allow time for remodelling)
- Recent revascularisation (less than 90 days)
Evidence:
- SCD-HeFT (2005): ICD vs placebo in NYHA II-III, LVEF ≤35%; 23% relative mortality reduction (HR 0.77, p=0.007) [9]
- MADIT-II (2002): ICD vs medical therapy post-MI, LVEF ≤30%; 31% mortality reduction
- NNT = 14 over 5 years to prevent 1 death
Types:
- Single-chamber ICD: RV lead only; for bradycardia pacing + defibrillation
- Dual-chamber ICD: RA + RV leads; better discrimination of atrial vs ventricular arrhythmias
- CRT-D: Biventricular pacing + defibrillation (if CRT criteria met)
Practical Points:
- Re-assess LVEF after 3 months GDMT (may improve to > 35%, obviating ICD need)
- Discuss risks (inappropriate shocks, infection, psychological impact) and benefits
- Subcutaneous ICD (S-ICD) option if no pacing needs
Cardiac Resynchronisation Therapy (CRT)
Indications (ESC/AHA 2021): [10]
Class I (Strongly Recommended):
- LVEF ≤35% despite ≥3 months optimal GDMT
- NYHA II-IV ambulatory
- QRS ≥150 ms with LBBB morphology (strongest indication; NNT = 5-6 to prevent 1 death)
Class IIa (Reasonable):
- QRS 130-149 ms with LBBB
- QRS ≥150 ms without LBBB (non-specific IVCD)
Class IIb (May Consider):
- Atrial fibrillation with criteria above (requires rate control or AV node ablation to ensure biventricular pacing > 95%)
- QRS less than 130 ms (generally not beneficial)
Evidence:
- CARE-HF (2005): CRT vs medical therapy; 36% mortality reduction [10]
- MADIT-CRT (2009): CRT-D vs ICD alone in NYHA I-II, QRS ≥130 ms; 34% reduction in HF events
- Meta-analysis: CRT reduces all-cause mortality 24%, HF hospitalisations 38%, improves NYHA class and QoL [10]
Mechanism:
- LBBB → ventricular dyssynchrony (septum contracts before lateral wall)
- Biventricular pacing (RV + LV leads) → synchronized contraction → improved ejection, reduced MR, reverse remodelling
Types:
- CRT-P: Pacing only (if no ICD indication)
- CRT-D: Pacing + defibrillation (most common; majority of patients meet both ICD and CRT criteria)
Practical Points:
- ~30% are "non-responders" (no symptomatic or echocardiographic improvement); LBBB + QRS ≥150 ms best predictors of response
- LV lead placement via coronary sinus (targets lateral/posterolateral wall)
- Requires echocardiographic optimization in some cases (AV and VV interval programming)
Advanced Therapies (Stage D Heart Failure)
Left Ventricular Assist Device (LVAD):
Indications:
- Bridge to transplant: Maintain organ perfusion while awaiting donor heart
- Destination therapy: No transplant candidacy; LVAD as permanent therapy
- Bridge to decision: Stabilize to assess transplant eligibility (e.g., while treating infection, improving nutrition)
Criteria:
- INTERMACS profiles 1-4 (cardiogenic shock to recurrent hospitalisations despite GDMT)
- LVEF less than 25%, peak VO2 less than 12 mL/kg/min, frequent hospitalisations, inotrope-dependent
Outcomes:
- Continuous-flow LVADs: 80% 1-year survival, 70% 2-year survival
- Complications: Bleeding, thrombosis, infection, device malfunction, stroke
Heart Transplantation:
Gold Standard for End-Stage HF:
- Criteria:
- Age typically less than 70 years (varies by centre)
- No active malignancy, infection, or irreversible pulmonary hypertension
- Good psychosocial support, compliance
- Peak VO2 less than 12-14 mL/kg/min or frequent hospitalisations despite maximal therapy
- Outcomes: 85-90% 1-year survival, 75% 5-year survival, 50% 10-year survival
- Limitation: Donor shortage (10% annual mortality on waiting list)
Palliative Care:
- Integrate early: Not just end-of-life; supportive care throughout disease trajectory
- Focus: Symptom control (dyspnoea management with opioids, anxiolytics), psychosocial support, advance care planning
- Indications for specialist palliative input:
- Recurrent hospitalisations despite GDMT
- NYHA IV symptoms
- Not candidate for advanced therapies
- Patient preference for comfort-focused care
Lifestyle and Non-Pharmacological Management
Patient Education: [3]
- Disease understanding: HF is chronic; lifelong treatment required
- Daily weights: Weigh same time daily; alert if > 2 kg gain in 3 days
- Symptom monitoring: Report worsening dyspnoea, oedema, fatigue
- Medication adherence: Explain importance of each medication
Dietary Modifications:
- Sodium restriction: less than 2-3 g/day (reduce fluid retention)
- Fluid restriction: 1.5-2 L/day if NYHA III-IV or hyponatraemia (controversial; individualize)
- Alcohol: Avoid if alcoholic cardiomyopathy; limit to less than 14 units/week otherwise
- Healthy diet: Mediterranean diet recommended (DASH diet for HTN)
Exercise:
- Cardiac rehabilitation: Improves exercise capacity, QoL, reduces hospitalisations
- Recommendation: 30 min moderate aerobic exercise 5×/week (walking, cycling)
- Resistance training: Light weights 2-3×/week
- Contraindication: Acute decompensation (wait until euvolaemic)
Vaccinations:
- Influenza vaccine: Annually (reduces HF hospitalisations)
- Pneumococcal vaccine: 13-valent PCV13 + 23-valent PPSV23
- COVID-19 vaccine: Recommended (HF patients high-risk for severe COVID)
Smoking Cessation and Weight Management:
- Smoking cessation mandatory (nicotine replacement, varenicline, behavioural support)
- Weight loss if obese (BMI > 30); improves symptoms and outcomes
Monitoring and Follow-Up
Frequency:
- Post-discharge: 1-2 weeks (high-risk readmission period)
- Uptitration phase: Every 1-2 weeks until target GDMT doses achieved
- Stable phase: 3-6 monthly
Monitoring Parameters:
| Parameter | Frequency | Action Threshold |
|---|---|---|
| Weight | Daily (patient home) | > 2 kg gain in 3 days → increase diuretic |
| BP | Each visit | SBP less than 90 mmHg → review GDMT doses |
| Heart Rate | Each visit | > 70 bpm → uptitrate beta-blocker |
| NYHA Class | Each visit | Worsening → investigate, optimize GDMT |
| NT-proBNP | 3-6 monthly | Rising → worsening HF, optimize therapy |
| U&E, Creatinine | 1-2 weeks (titration), 3 monthly (stable) | K+ > 5.5 or eGFR drop > 30% → adjust RAAS inhibitor/MRA |
| FBC | 6-12 monthly | Hb less than 120 g/L → iron studies, consider IV iron |
| Iron studies | 6-12 monthly | Ferritin less than 100 or TSAT less than 20% → IV iron |
| Echocardiography | Baseline, 3-6 months, then annually or if status change | LVEF improvement > 40% (HFimpEF); worsening → consider device therapy |
9. Complications
Acute Complications
| Complication | Incidence | Risk Factors | Clinical Features | Prevention | Management |
|---|---|---|---|---|---|
| Cardiogenic Shock | 5-10% of acute HF [3] | Acute MI, acute severe MR/AR, myocarditis | SBP less than 90 mmHg, cold/clammy, oliguria, altered consciousness | Optimal GDMT, revascularisation if ischaemic | IV inotropes (dobutamine), vasopressors (norepinephrine), urgent MCS (IABP, Impella, VA-ECMO), transplant assessment |
| Acute Pulmonary Oedema | 10-20% presentations [3] | Non-adherence, dietary indiscretion, AF with RVR | Severe dyspnoea, RR > 30, SpO2 less than 90%, pink frothy sputum | Medication adherence, daily weights, sodium restriction | Sit upright (45-90°), oxygen/NIV (CPAP), IV furosemide 40-80 mg bolus, consider IV GTN if SBP > 110 mmHg |
| Ventricular Arrhythmias | 40-50% of deaths [9] | Low EF (less than 25%), NSVT on Holter, long QT, electrolyte abnormalities | Palpitations, syncope, VT on monitor, ICD shocks | ICD if LVEF ≤35%, correct K+/Mg2+, avoid QT-prolonging drugs | DCCV if unstable VT; amiodarone load; electrolyte repletion; catheter ablation if refractory |
| Acute Kidney Injury | 25-30% hospitalisations [3] | Over-diuresis, NSAIDs, contrast, sepsis | ↑ Creatinine > 26 μmol/L in 48h or > 50% baseline, oliguria | Avoid nephrotoxins, judicious diuresis, monitor U&E | Hold ACEi/ARB/MRA temporarily; IV fluids if hypovolaemic; stop diuretics; treat sepsis; consider RRT if severe |
| Atrial Fibrillation | 30-40% of HFrEF [3] | LA dilation, elevated filling pressures | Irregular pulse, palpitations, haemodynamic compromise if RVR | Optimal GDMT (reverse remodelling), BB rate control | Rate control: Beta-blocker (1st line), digoxin (adjunct); Anticoagulation: CHA2DS2-VASc ≥1 (DOAC preferred); Rhythm control: Amiodarone if symptomatic, catheter ablation if refractory |
| Thromboembolism (VTE, Stroke) | 1-3%/year [3] | Low EF, AF, immobility, prior VTE | DVT/PE, stroke symptoms | Anticoagulation if AF (DOAC), mobilisation | Therapeutic anticoagulation; thrombolysis if massive PE/stroke within window |
Chronic Complications and Comorbidities
Cardiorenal Syndrome:
- Worsening renal function in 25-30% during hospitalisation [3]
- Mechanisms: Low cardiac output → renal hypoperfusion; venous congestion → ↑ renal venous pressure → ↓ GFR
- Management: Balance decongestion (diuresis) with renal perfusion; accept mild creatinine rise if decongesting; avoid over-diuresis
Anaemia and Iron Deficiency:
- Anaemia (Hb less than 130 g/L men, less than 120 g/L women) in 30-50% [15]
- Iron deficiency (ferritin less than 100 ng/mL OR ferritin 100-299 + TSAT less than 20%) in 50% [15]
- FAIR-HF Trial: IV ferric carboxymaltose improved symptoms, QoL, 6-min walk distance [15]
- AFFIRM-AHF: IV iron reduced HF hospitalisations post-discharge [15]
- Management: IV iron (ferric carboxymaltose 500-1000 mg) if iron deficient; repeat doses guided by ferritin/TSAT
Cardiac Cachexia:
- Unintentional weight loss > 5% over 6 months (excluding oedema)
- Poor prognosis (1-year mortality 50%)
- Mechanisms: Chronic inflammation (TNF-α, IL-6), reduced appetite, gut congestion (malabsorption)
- Management: Nutritional support, exercise (anabolic), treat underlying HF
Depression and Anxiety:
- Depression in 20-40% of HF patients [3]
- Associated with worse outcomes, poor adherence
- Screening: PHQ-9, GAD-7
- Management: SSRIs (sertraline safe in HF), CBT, cardiac rehabilitation
Cognitive Impairment:
- Present in 25-50% (especially elderly)
- Mechanisms: Cerebral hypoperfusion, emboli, small vessel disease
- Screen with MMSE/MoCA
- Management: Optimize cardiac output, control AF, BP management
Hepatic Congestion:
- Congestive hepatopathy ("cardiac liver"): ↑ transaminases (ALT/AST 2-3× ULN), ↑ bilirubin, ↑ INR
- Chronic: Cardiac cirrhosis (irreversible fibrosis)
- Management: Decongestion (diuretics); if cirrhotic, combined heart-liver transplant
10. Prognosis and Outcomes
Natural History (Untreated)
Without treatment, HFrEF has a grim prognosis: [6,7]
- 1-year mortality: 30-40%
- 5-year mortality: 50-60% (worse than many cancers)
- Mode of death: 50% sudden cardiac death (arrhythmic), 50% progressive pump failure
- Progressive functional decline (NYHA class deterioration)
- Frequent hospitalisations (QoL impact)
With Optimal GDMT
Modern therapy has revolutionized outcomes: [6,7,12]
- Mortality reduction: Each GDMT pillar reduces mortality 20-30%; combined effect 60-70% relative risk reduction vs no treatment [3,4]
- 5-year mortality: Reduced to 15-25% with optimal GDMT [6]
- Functional improvement: 40-60% improve by ≥1 NYHA class [3]
- EF improvement: 20-40% achieve LVEF > 40% (HFimpEF); continue GDMT (relapse if stopped) [19]
- Hospitalisations: Reduced 30-50% with GDMT + device therapy [3]
Prognostic Factors
Poor Prognosis:
- Lower EF (less than 25%)
- NYHA Class IV
- Elevated natriuretic peptides (NT-proBNP > 1000 pg/mL)
- Hyponatraemia (Na+ less than 135 mmol/L; indicates advanced neurohormonal activation)
- Renal dysfunction (eGFR less than 30 mL/min/1.73m²; cardiorenal syndrome)
- Anaemia (Hb less than 120 g/L)
- Diabetes mellitus
- Ischaemic aetiology (vs non-ischaemic)
- Wide QRS (> 150 ms; dyssynchrony)
- Reduced peak VO2 (less than 12 mL/kg/min; advanced HF)
- Cachexia (> 5% weight loss)
- Hypotension (SBP less than 100 mmHg)
- Frequent hospitalisations (> 2 per year)
Better Prognosis:
- Response to GDMT with EF improvement (HFimpEF)
- Non-ischaemic aetiology (especially if recent-onset; more likely to recover)
- Younger age (less than 60 years)
- Absence of comorbidities
- Good nutritional status
- Higher SBP (paradoxically; SBP less than 100 mmHg worse)
- Lower natriuretic peptides or declining levels with therapy
- Achievement of target GDMT doses [12]
Prognostic Scores:
- Seattle Heart Failure Model: Estimates 1-5 year survival based on clinical, lab, and medication variables (online calculator available)
- MAGGIC Risk Score: Mortality prediction based on age, EF, creatinine, NYHA, DM, etc.
Quality of Life Considerations
- Regular NYHA and symptom assessment (Kansas City Cardiomyopathy Questionnaire)
- Early goals of care discussions (advance care planning)
- Patient preferences regarding ICD, transplant, LVAD
- Integration of palliative care (not just end-of-life)
- Exercise rehabilitation improves QoL significantly (↑ 6-min walk, ↓ depression)
11. Evidence Base and Guidelines
Key Landmark Trials
RAAS Inhibition:
- CONSENSUS (1987): Enalapril vs placebo in NYHA IV; 27% mortality reduction at 6 months → first trial to show ACEi benefit
- SOLVD (1991): Enalapril vs placebo in LVEF less than 35%; 16% mortality reduction → established ACEi as standard
- PARADIGM-HF (2014): Sacubitril/valsartan vs enalapril; 20% reduction in CV death/HF hosp → ARNI became first-line [16]
Beta-Blockers:
- CIBIS-II (1999): Bisoprolol vs placebo; 34% mortality reduction [23]
- MERIT-HF (1999): Metoprolol succinate; 34% mortality reduction, 41% reduction in sudden death [23]
- COPERNICUS (2002): Carvedilol in severe HF; 35% mortality reduction despite LVEF less than 25% [23]
Mineralocorticoid Receptor Antagonists:
- RALES (1999): Spironolactone in NYHA III-IV; 30% mortality reduction [22]
- EMPHASIS-HF (2011): Eplerenone in NYHA II; 37% reduction in CV death/HF hosp [22]
SGLT2 Inhibitors:
- DAPA-HF (2019): Dapagliflozin vs placebo; 26% reduction in CV death/worsening HF regardless of diabetes [13]
- EMPEROR-Reduced (2020): Empagliflozin; 25% reduction in CV death/HF hosp [14]
Device Therapy:
- SCD-HeFT (2005): ICD vs placebo in LVEF ≤35%; 23% mortality reduction [9]
- CARE-HF (2005): CRT vs medical therapy; 36% mortality reduction in LBBB + QRS ≥120 ms [10]
Other Therapies:
- SHIFT (2010): Ivabradine in HR ≥70 bpm; 18% reduction in CV death/HF hosp
- DIG (1997): Digoxin; no mortality effect but 28% reduction in HF hospitalisations
- A-HeFT (2004): Hydralazine-ISDN in African Americans; 43% mortality reduction
Current Guidelines
| Guideline | Organisation | Year | Key Recommendations |
|---|---|---|---|
| ESC Heart Failure Guidelines | European Society of Cardiology | 2021 [3] | Four pillars of GDMT: ARNI (preferred) or ACEi/ARB + Beta-blocker + MRA + SGLT2i; initiate simultaneously and uptitrate to target doses; ICD if LVEF ≤35%; CRT if LBBB + QRS ≥130 ms |
| AHA/ACC/HFSA Guidelines | American Heart Association / American College of Cardiology / Heart Failure Society of America | 2022 [4] | Similar four-pillar approach; emphasis on SGLT2i benefit regardless of DM; ARNI first-line; uptitration critical; comprehensive team-based care |
| NICE NG106 | UK National Institute for Health and Care Excellence | 2018 (updated 2023) | ARNI first-line if tolerated; offer MRA and BB to all; SGLT2i for all; specialist HF MDT referral; ICD/CRT per established criteria |
| CCS Heart Failure Guidelines | Canadian Cardiovascular Society | 2021 | Four foundational therapies (ARNI + BB + MRA + SGLT2i); uptitration targets; natriuretic peptide-guided therapy; early device assessment |
Consensus Across Guidelines:
- Four pillars are foundational: All patients should receive ARNI/ACEi/ARB + BB + MRA + SGLT2i unless contraindicated
- ARNI preferred over ACEi: Sacubitril/valsartan first-line if no contraindications
- Titrate to target doses: 50% of benefit lost if under-dosed; prioritize dose escalation over number of agents [12]
- SGLT2i for all: Regardless of diabetes, NYHA class, or baseline therapy
- Device therapy: ICD if LVEF ≤35% after 3 months GDMT; CRT if LBBB + QRS ≥150 ms
- Multidisciplinary care: HF specialist nurse, pharmacist, dietitian, rehabilitation
12. Exam-Focused Sections
Common MRCP/FRACP Exam Questions
Written Exam (MCQ/SBA) Scenarios:
-
"A 68-year-old man with LVEF 28% is on enalapril 10 mg BD, bisoprolol 10 mg OD, and furosemide 40 mg OD. What additional therapy is MOST likely to reduce mortality?"
- Answer: SGLT2 inhibitor (dapagliflozin or empagliflozin) – add as 4th pillar; also add MRA
-
"A patient with HFrEF on maximal GDMT has LVEF 32%, LBBB with QRS 160 ms, NYHA III. What is the MOST appropriate next step?"
- Answer: Cardiac resynchronisation therapy (CRT-D) – meets criteria (LVEF ≤35%, LBBB, QRS ≥150 ms, NYHA II-IV)
-
"Which of the following medications is CONTRAINDICATED in HFrEF?"
- Answer: Non-dihydropyridine calcium channel blockers (diltiazem, verapamil) – negative inotropes, worsen HF; also thiazolidinediones
-
"A patient presents with HFrEF. NT-proBNP is 85 pg/mL. What is the MOST likely explanation?"
- Answer: Obesity (adipocytes clear BNP) OR flash pulmonary oedema (insufficient time for release) OR incorrect diagnosis
-
"What is the NNT for sacubitril/valsartan vs enalapril in PARADIGM-HF?"
- Answer: 21 over 27 months to prevent 1 CV death/HF hospitalisation [16]
Clinical Exam (PACES/Clinical) Scenarios:
- "Examine this patient's cardiovascular system. The patient is breathless at rest." (HFrEF with decompensation)
- Findings: Elevated JVP, displaced apex, S3 gallop, bilateral basal crackles, peripheral oedema
- Differential: Acute decompensated HF, consider precipitant (AF, ACS, non-adherence)
Data Interpretation:
- "Echo shows LVEF 32%, dilated LV (LVEDD 62 mm), moderate functional MR. ECG shows LBBB with QRS 165 ms. What device therapy is indicated?"
- Answer: CRT-D (meets CRT criteria: LVEF ≤35%, LBBB, QRS ≥150 ms; and ICD criteria: LVEF ≤35%)
Viva Voce Model Answers
Opening Statement for HFrEF Viva:
"Heart failure with reduced ejection fraction is a clinical syndrome characterised by the heart's inability to meet metabolic demands due to impaired left ventricular systolic function. It is defined by a left ventricular ejection fraction of 40% or less, with symptoms of heart failure and elevated natriuretic peptides. The condition affects approximately 32 million people globally and has a 5-year mortality of 50% without treatment, though modern guideline-directed medical therapy can reduce mortality by 60-70%."
Q: "How would you investigate a patient with suspected HFrEF?"
A: "I would take a systematic approach:
Initial investigations include NT-proBNP (if > 125 pg/mL, HF likely; negative predictive value 98% if less than 125), 12-lead ECG (identify LBBB for CRT eligibility, Q waves suggesting ischaemic aetiology, arrhythmias), and transthoracic echocardiography to confirm LVEF ≤40%, assess wall motion, quantify valvular disease, and estimate filling pressures.
Baseline bloods include FBC (anaemia in 30-50%), U&E (baseline renal function before RAAS inhibition), liver function (hepatic congestion), thyroid function (hypo/hyperthyroidism can cause HF), HbA1c (diabetes comorbidity), lipids (if ischaemic), troponin (exclude ACS), and iron studies (ferritin less than 100 or TSAT less than 20% warrants IV iron).
Aetiological investigations include coronary angiography if suspected ischaemic cause (most common), and cardiac MRI if aetiology unclear—late gadolinium enhancement differentiates ischaemic (subendocardial/transmural) from non-ischaemic (mid-wall) patterns."
Q: "What are the four pillars of GDMT in HFrEF?"
A: "The four foundational therapies, all of which independently reduce mortality, are:
-
RAAS inhibition: Sacubitril/valsartan (ARNI) is first-line, target 97/103 mg BD; reduces CV death/HF hospitalisation 20% vs enalapril in PARADIGM-HF. If ARNI not tolerated, use ACE inhibitor (ramipril 10 mg OD) or ARB (candesartan 32 mg OD).
-
Beta-blocker: Bisoprolol (target 10 mg OD), carvedilol (target 25 mg BD), or metoprolol succinate (target 200 mg OD); reduce mortality 34% (MERIT-HF, CIBIS-II). Target heart rate 50-60 bpm.
-
Mineralocorticoid receptor antagonist: Spironolactone or eplerenone 25-50 mg OD; reduce mortality 30% (RALES). Monitor potassium closely.
-
SGLT2 inhibitor: Dapagliflozin 10 mg OD or empagliflozin 10 mg OD; reduce CV death/HF events 25-26% (DAPA-HF, EMPEROR-Reduced) regardless of diabetes status.
All four should be initiated simultaneously or in rapid succession and titrated to target doses, as 50% of mortality benefit is lost if under-dosed."
Q: "When is an ICD indicated in HFrEF?"
A: "An ICD is indicated for primary prevention of sudden cardiac death in patients with:
- LVEF ≤35% despite at least 3 months of optimal GDMT
- NYHA Class II-III (ambulatory patients)
- Life expectancy > 1 year with good functional status
The evidence comes from SCD-HeFT, which showed a 23% relative reduction in mortality with ICD vs placebo in NYHA II-III patients with LVEF ≤35%, with a number needed to treat of 14 over 5 years.
Contraindications include NYHA IV with no plan for advanced therapies, recent MI within 40 days, or recent revascularisation within 90 days—these require a reassessment period to allow for potential EF recovery."
Q: "What are the criteria for cardiac resynchronisation therapy?"
A: "CRT is a Class I indication (strongly recommended) in patients with:
- LVEF ≤35% despite at least 3 months optimal GDMT
- NYHA Class II-IV ambulatory
- QRS ≥150 ms with LBBB morphology—this is the strongest indication, with a number needed to treat of 5-6 to prevent one death
The benefit is greatest with LBBB and wider QRS (≥150 ms). Evidence from CARE-HF showed a 36% mortality reduction, and MADIT-CRT showed a 34% reduction in HF events. CRT works by resynchronizing ventricular contraction, improving ejection efficiency, reducing mitral regurgitation, and promoting reverse remodelling.
Most patients receive CRT-D (combined pacing and defibrillator) as they meet criteria for both CRT and ICD. About 30% are non-responders, with LBBB and QRS ≥150 ms being the best predictors of response."
Common Mistakes to Avoid
❌ Don't delay SGLT2 inhibitors: Add immediately; don't wait for ACEi/BB uptitration ❌ Don't under-dose GDMT: Titrate to target doses; 50% of benefit lost if under-dosed [12] ❌ Don't use verapamil or diltiazem: Negative inotropes; contraindicated in HFrEF (dihydropyridine CCBs like amlodipine safe if HTN) ❌ Don't stop beta-blockers in acute decompensation: Continue if possible; reduce dose if necessary but avoid cessation (rebound tachycardia) ❌ Don't miss iron deficiency: Check iron studies; IV iron improves QoL and reduces hospitalisations if ferritin less than 100 or TSAT less than 20% [15] ❌ Don't forget to reassess LVEF: Check after 3 months GDMT (may improve to > 35%, affecting ICD need; 20-40% achieve HFimpEF) [19] ❌ Don't prescribe NSAIDs: Double HF hospitalisation risk; use paracetamol for analgesia ❌ Don't over-diurese: Can cause AKI, hypotension, worsening renal function; titrate to euvolaemia
13. Patient/Layperson Explanation
What is Heart Failure with Reduced Ejection Fraction?
Heart failure means your heart muscle has become weak and cannot pump blood as well as it should. "Reduced ejection fraction" means we can measure how much blood your heart pumps out with each beat, and in your case, it is lower than normal (under 40%, when normal is 55-70%). This doesn't mean your heart is about to stop—it means it's working less efficiently and needs help from medications.
What causes it?
The most common cause (about 2 out of 3 cases) is damage from a heart attack, which kills some of the heart muscle so it can't contract properly. Other causes include:
- Long-term high blood pressure (puts strain on the heart)
- Heart muscle disease (cardiomyopathy, sometimes genetic)
- Heart valve problems (leaky or narrowed valves)
- Infections or inflammation of the heart
- Alcohol excess or certain chemotherapy drugs
What are the symptoms?
- Breathlessness, especially when lying flat or during activity (climbing stairs, walking)
- Swelling in your ankles, legs, or tummy from fluid buildup
- Feeling very tired and lacking energy
- Waking up at night gasping for breath
- Needing to urinate more at night (your body gets rid of fluid when you lie down)
- Reduced appetite, feeling full quickly
How is it treated?
The very good news is that modern medications can dramatically improve your heart function and help you live much longer. Most patients will be prescribed four main types of tablets:
- ARNI or ACE inhibitor: Takes pressure off your heart by relaxing blood vessels (e.g., sacubitril/valsartan, ramipril)
- Beta-blocker: Slows your heart rate so it can pump more efficiently (e.g., bisoprolol, carvedilol)
- MRA: Removes excess fluid and protects your heart muscle (e.g., spironolactone)
- SGLT2 inhibitor: Originally for diabetes, but helps all heart failure patients by improving heart energy and reducing fluid (e.g., dapagliflozin, empagliflozin)
You'll also likely get a "water tablet" (diuretic) like furosemide to remove extra fluid and reduce breathlessness and swelling.
Some patients may need a special pacemaker-like device:
- ICD (defibrillator): Protects against dangerous heart rhythms
- CRT (biventricular pacemaker): Helps your heart pump more efficiently by coordinating both sides
What should I expect?
Many patients feel significantly better within weeks to months of starting treatment. Your breathlessness improves, swelling goes down, and you have more energy. Your heart function may even improve over time (this happens in 20-40% of patients). You will need regular check-ups with blood tests to monitor kidney function and potassium levels, and periodic heart scans (echocardiograms).
What do I need to do?
- Take all your medications every day: Even if you feel well; they're protecting your heart
- Weigh yourself daily: At the same time each day; tell your doctor if you gain > 2 kg (4-5 lbs) in 3 days
- Reduce salt: Aim for less than 2-3 g per day (avoid processed foods, don't add salt at table)
- Limit fluids: To 1.5-2 litres/day if advised
- Stay active: Gentle exercise like walking is very beneficial once your symptoms are stable
- Avoid alcohol: Especially if alcohol caused your heart failure; otherwise limit to small amounts
- Get vaccinations: Flu jab yearly, pneumonia jab, COVID vaccine
When to seek urgent help
Go to A&E or call 999 if you experience:
- Severe breathlessness that doesn't improve with rest or your usual medications
- Chest pain (may indicate heart attack)
- Fainting or feeling very faint
- Confusion or drowsiness
- Very fast or irregular heartbeat
- Rapid weight gain (> 2 kg in 3 days) with worsening swelling
What is the outlook?
Without treatment, heart failure is a serious condition. But with modern medications, many people live for many years with good quality of life. About 1 in 5 to 1 in 4 patients see their heart function improve back to normal or near-normal with treatment. The key is taking your medications as prescribed, monitoring your symptoms, and attending regular check-ups.
14. References
Primary Sources
-
Savarese G, Becher PM, Lund LH, et al. Global burden of heart failure: a comprehensive and updated review of epidemiology. Cardiovascular Research. 2023;118(17):3272-3287. doi:10.1093/cvr/cvac013
-
Virani SS, Alonso A, Benjamin EJ, et al. Heart Disease and Stroke Statistics—2021 Update: A Report From the American Heart Association. Circulation. 2021;143(8):e254-e743. doi:10.1161/CIR.0000000000000950
-
McDonagh TA, Metra M, Adamo M, et al. 2021 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure. European Heart Journal. 2021;42(36):3599-3726. doi:10.1093/eurheartj/ehab368
-
Heidenreich PA, Bozkurt B, Aguilar D, et al. 2022 AHA/ACC/HFSA Guideline for the Management of Heart Failure: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation. 2022;145(18):e895-e1032. doi:10.1161/CIR.0000000000001063
-
Schultheiss HP, Fairweather D, Caforio ALP, et al. Dilated cardiomyopathy. Nature Reviews Disease Primers. 2019;5(1):32. doi:10.1038/s41572-019-0084-1
-
Taylor CJ, Ordóñez-Mena JM, Roalfe AK, et al. Trends in survival after a diagnosis of heart failure in the United Kingdom 2000-2017: population based cohort study. BMJ. 2019;364:l223. doi:10.1136/bmj.l223
-
Conrad N, Judge A, Tran J, et al. Temporal trends and patterns in heart failure incidence: a population-based study of 4 million individuals. Lancet. 2018;391(10120):572-580. doi:10.1016/S0140-6736(17)32520-5
-
Januzzi JL, van Kimmenade R, Lainchbury J, et al. NT-proBNP testing for diagnosis and short-term prognosis in acute destabilized heart failure: an international pooled analysis of 1256 patients: the International Collaborative of NT-proBNP Study. European Heart Journal. 2006;27(3):330-337. doi:10.1093/eurheartj/ehi631
-
Bardy GH, Lee KL, Mark DB, et al. Amiodarone or an implantable cardioverter-defibrillator for congestive heart failure (SCD-HeFT). New England Journal of Medicine. 2005;352(3):225-237. doi:10.1056/NEJMoa043399
-
Cleland JGF, Daubert JC, Erdmann E, et al. The effect of cardiac resynchronization on morbidity and mortality in heart failure (CARE-HF). New England Journal of Medicine. 2005;352(15):1539-1549. doi:10.1056/NEJMoa050496
-
Raphael C, Briscoe C, Davies J, et al. Limitations of the New York Heart Association functional classification system and self-reported walking distances in chronic heart failure. Heart. 2007;93(4):476-482. doi:10.1136/hrt.2006.089656
-
Greene SJ, Butler J, Albert NM, et al. Medical Therapy for Heart Failure With Reduced Ejection Fraction: The CHAMP-HF Registry. Journal of the American College of Cardiology. 2018;72(4):351-366. doi:10.1016/j.jacc.2018.04.070
-
McMurray JJV, Solomon SD, Inzucchi SE, et al. Dapagliflozin in Patients with Heart Failure and Reduced Ejection Fraction (DAPA-HF). New England Journal of Medicine. 2019;381(21):1995-2008. doi:10.1056/NEJMoa1911303
-
Packer M, Anker SD, Butler J, et al. Cardiovascular and Renal Outcomes with Empagliflozin in Heart Failure (EMPEROR-Reduced). New England Journal of Medicine. 2020;383(15):1413-1424. doi:10.1056/NEJMoa2022190
-
Anker SD, Comin Colet J, Filippatos G, et al. Ferric carboxymaltose in patients with heart failure and iron deficiency (FAIR-HF). New England Journal of Medicine. 2009;361(25):2436-2448. doi:10.1056/NEJMoa0908355
-
McMurray JJ, Packer M, Desai AS, et al. Angiotensin-neprilysin inhibition versus enalapril in heart failure (PARADIGM-HF). New England Journal of Medicine. 2014;371(11):993-1004. doi:10.1056/NEJMoa1409077
-
Troughton RW, Frampton CM, Brunner-La Rocca HP, et al. Effect of B-type natriuretic peptide-guided treatment of chronic heart failure on total mortality and hospitalization: an individual patient meta-analysis. European Heart Journal. 2014;35(23):1559-1567. doi:10.1093/eurheartj/ehu090
-
Lesyuk W, Kriza C, Kolominsky-Rabas P. Cost-of-illness studies in heart failure: a systematic review 2004-2016. BMC Cardiovascular Disorders. 2018;18(1):74. doi:10.1186/s12872-018-0815-3
-
Wilcox JE, Fang JC, Margulies KB, Mann DL. Heart Failure With Recovered Left Ventricular Ejection Fraction: JACC Scientific Expert Panel. Journal of the American College of Cardiology. 2020;76(6):719-734. doi:10.1016/j.jacc.2020.05.075
-
Carnethon MR, Pu J, Howard G, et al. Cardiovascular Health in African Americans: A Scientific Statement From the American Heart Association. Circulation. 2017;136(21):e393-e423. doi:10.1161/CIR.0000000000000534
-
Zamorano JL, Lancellotti P, Rodriguez Muñoz D, et al. 2016 ESC Position Paper on cancer treatments and cardiovascular toxicity developed under the auspices of the ESC Committee for Practice Guidelines. European Heart Journal. 2016;37(36):2768-2801. doi:10.1093/eurheartj/ehw211
-
Pitt B, Zannad F, Remme WJ, et al. The effect of spironolactone on morbidity and mortality in patients with severe heart failure (RALES). New England Journal of Medicine. 1999;341(10):709-717. doi:10.1056/NEJM199909023411001
-
CIBIS-II Investigators and Committees. The Cardiac Insufficiency Bisoprolol Study II (CIBIS-II): a randomised trial. Lancet. 1999;353(9146):9-13. doi:10.1016/S0140-6736(98)11181-9
-
Stevenson LW, Perloff JK. The limited reliability of physical signs for estimating hemodynamics in chronic heart failure. JAMA. 1989;261(6):884-888. doi:10.1001/jama.1989.03420060100040
-
Thibodeau JT, Turer AT, Gualano SK, et al. Characterization of a novel symptom of advanced heart failure: bendopnea. JACC Heart Failure. 2014;2(1):24-31. doi:10.1016/j.jchf.2013.07.009
Guidelines and Consensus Documents
-
National Institute for Health and Care Excellence (NICE). Chronic heart failure in adults: diagnosis and management (NG106). London: NICE; 2018 (updated 2023). Available from: https://www.nice.org.uk/guidance/ng106
-
European Society of Cardiology. 2021 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure. Available from: https://www.escardio.org/Guidelines/Clinical-Practice-Guidelines/Acute-and-Chronic-Heart-Failure
-
American Heart Association/American College of Cardiology/Heart Failure Society of America. 2022 AHA/ACC/HFSA Guideline for the Management of Heart Failure. Available from: https://www.ahajournals.org/doi/10.1161/CIR.0000000000001063
Medical Disclaimer: MedVellum content is for educational purposes and clinical reference. It does not replace professional medical judgement. Always verify critical information with primary sources and consider individual patient factors. Guidelines and evidence evolve; check for updates before applying clinically.
Learning map
Use these linked topics to study the concept in sequence and compare related presentations.
Prerequisites
Start here if you need the foundation before this topic.
- Cardiac Physiology
Differentials
Competing diagnoses and look-alikes to compare.
- Heart Failure with Preserved Ejection Fraction
- Pulmonary Oedema - Non-cardiac
Consequences
Complications and downstream problems to keep in mind.
- Cardiogenic Shock
- Acute Pulmonary Oedema