Hepatitis C
Transmission occurs predominantly via parenteral exposure : injection drug use (IVDU), blood transfusions prior to universal screening (pre-1992 in the UK, pre-1990 in the USA), needlestick injuries, vertical...
What matters first
Transmission occurs predominantly via parenteral exposure : injection drug use (IVDU), blood transfusions prior to universal screening (pre-1992 in the UK, pre-1990 in the USA), needlestick injuries, vertical...
Decompensated Cirrhosis (Ascites, Encephalopathy, Variceal Bleeding)
10 Jan 2026
Generated educational material; verify before clinical use.
Visible references section
See the concept before reading it
Study the key anatomy, imaging, and decision pathways as full teaching plates.
Clinical board
A visual summary of the highest-yield teaching signals on this page.
Urgent signals
Safety-critical features pulled from the topic metadata.
- Decompensated Cirrhosis (Ascites, Encephalopathy, Variceal Bleeding)
- Hepatocellular Carcinoma (HCC)
- Acute Hepatitis C with Jaundice and Coagulopathy
- HCV/HIV Co-infection with Decompensation
Exam focus
Current exam surfaces linked to this topic.
- MRCP
- FRACP
- USMLE
- PLAB
Content status and exam context
This page is AI-generated educational content. It may contain errors or omissions and is not a substitute for current guidelines, local protocols, senior clinical judgement, or professional medical advice.
MedVellum does not claim an individual clinician reviewer, board certification, or professional credential for this page unless a future version names a real, verifiable reviewer.
Clinical explanation and evidence
Hepatitis C
1. Clinical Overview
Summary
Hepatitis C is a blood-borne viral infection caused by the Hepatitis C Virus (HCV), a positive-sense single-stranded RNA virus of the Flaviviridae family. HCV is a leading cause of chronic liver disease, cirrhosis, and hepatocellular carcinoma (HCC) worldwide, with an estimated 58 million people chronically infected globally as of 2020. [1,2]
Transmission occurs predominantly via parenteral exposure: injection drug use (IVDU), blood transfusions prior to universal screening (pre-1992 in the UK, pre-1990 in the USA), needlestick injuries, vertical transmission from mother to child, and rarely through sexual contact. [3,4] The majority of acute HCV infections are asymptomatic and frequently undiagnosed. Without treatment, 70-85% of acutely infected individuals progress to chronic infection, defined as persistent HCV RNA detectability for ≥6 months. [5,6]
Chronic HCV infection leads to progressive hepatic fibrosis, with 20-30% of patients developing cirrhosis over 20-30 years. Once cirrhosis is established, the annual incidence of hepatocellular carcinoma ranges from 1-4%. [7,8] Factors accelerating fibrosis progression include alcohol consumption, obesity, HIV or HBV co-infection, male sex, and older age at infection. [9]
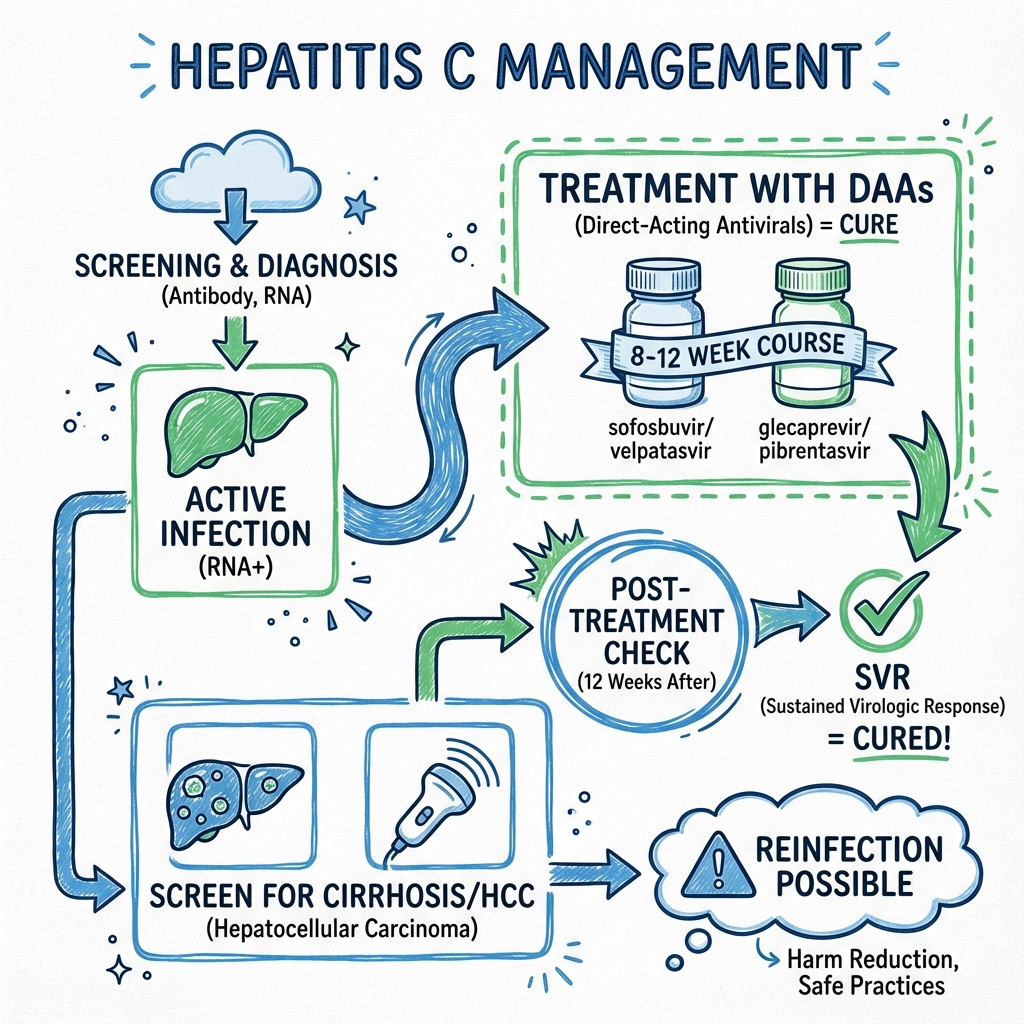
The introduction of Direct-Acting Antivirals (DAAs) has revolutionized HCV management. Modern pangenotypic regimens—including sofosbuvir/velpatasvir and glecaprevir/pibrentasvir—achieve Sustained Virological Response (SVR) rates exceeding 95% across all genotypes, with treatment durations of 8-12 weeks and minimal side effects. [10,11] SVR12 (undetectable HCV RNA 12 weeks post-treatment) is considered virological cure, with relapse rates less than 1% after this endpoint. [12,13] HCV is now a curable infection, and the World Health Organization (WHO) has set a target to eliminate HCV as a public health threat by 2030. [14]
Clinical Pearls
"The Silent Epidemic": Up to 75% of acute HCV infections are asymptomatic, leading to delayed diagnosis often decades after initial infection when cirrhosis or HCC has developed. [5]
"95%+ Cure with DAAs": Pangenotypic DAA regimens cure > 95% of patients regardless of genotype, fibrosis stage, or prior treatment failure. Treatment is oral, well-tolerated, and typically 8-12 weeks. [10,11]
"Transmission = Blood-to-Blood": IVDU accounts for ~90% of new HCV infections in developed countries. Transfusion-related transmission is now rare due to universal blood screening. [3]
"No Vaccine Available": Unlike hepatitis A and B, there is currently no effective vaccine for HCV due to viral genetic diversity and immune evasion mechanisms. [15]
"Cure Does Not Eliminate HCC Risk in Cirrhosis": Patients with cirrhosis who achieve SVR still require lifelong 6-monthly HCC surveillance (ultrasound ± AFP), as HCC risk is reduced by 70-80% but not eliminated. [16,17]
"SVR12 = SVR24": SVR12 and SVR24 are concordant in > 99% of cases. SVR12 is the validated cure endpoint. [12,13]
2. Molecular Pathophysiology
Virology and Structure
HCV is a positive-sense, single-stranded RNA virus (~9.6 kb genome) belonging to the Flaviviridae family, genus Hepacivirus. The viral genome encodes a single polyprotein that is cleaved by host and viral proteases into 10 mature proteins:
| Protein Type | Proteins | Function | DAA Target |
|---|---|---|---|
| Structural | Core, E1, E2, p7 | Virion assembly, envelope glycoproteins, ion channel | No |
| Non-structural | NS2, NS3, NS4A, NS4B, NS5A, NS5B | Polyprotein processing (NS2, NS3/4A protease), RNA replication (NS5B RNA-dependent RNA polymerase), replication complex assembly (NS5A) | Yes: NS3/4A, NS5A, NS5B |
The NS5B RNA-dependent RNA polymerase lacks proofreading activity, resulting in a high mutation rate (~10⁻³ substitutions per nucleotide per genome replication). This generates quasispecies diversity within individual hosts, facilitating immune evasion and chronicity. [18]
HCV Genotypes: Clinical Significance and Geographic Distribution
HCV exhibits high genetic heterogeneity, classified into 8 major genotypes (GT1-8) and > 90 subtypes based on nucleotide sequence divergence (> 30% between genotypes, 15-30% between subtypes). [19,20]
| Genotype | Global Prevalence | Geographic Distribution | Key Clinical Characteristics | Historical Treatment Difficulty | Current DAA Era |
|---|---|---|---|---|---|
| GT1a | ~23% | USA, Northern Europe, Australia | Most common in North America. RAS to NS5A inhibitors more common than 1b. | Difficult (48 weeks PEG-IFN + RBV, SVR ~40-50%) | Curable with all pangenotypic DAAs (SVR > 95%) [10,11] |
| GT1b | ~23% | Southern Europe, Japan, East Asia | Most common globally. Less NS5A resistance than 1a. | Difficult (48 weeks PEG-IFN + RBV, SVR ~40-50%) | Curable with all pangenotypic DAAs (SVR > 95%) [10,11] |
| GT2 | ~13% | Europe, North America, West Africa | Generally favorable natural history and treatment response. | Easier (24 weeks PEG-IFN + RBV, SVR ~70-80%) | Curable with all pangenotypic DAAs (SVR > 95%) [10] |
| GT3 | ~22% | South Asia, Scandinavia, Australia, IVDU populations | Associated with accelerated steatosis, fibrosis, and 2-3× higher HCC risk independent of cirrhosis. [21,22] NS5A inhibitor resistance more common. | Moderate (24-48 weeks PEG-IFN + RBV, SVR ~60-70%) | Requires careful regimen selection. SOF/VEL 12 weeks or GLE/PIB 8-12 weeks. Historically difficult with early DAAs. [10,23] |
| GT4 | ~13% | Middle East, North Africa, sub-Saharan Africa | Expanding in Europe due to migration. | Moderate-Difficult | Curable with pangenotypic DAAs (SVR > 95%) [10] |
| GT5 | less than 5% | South Africa | Rare. Limited data. | Limited data | Curable with pangenotypic DAAs (SVR > 95%) [10] |
| GT6 | less than 5% | Southeast Asia (Vietnam, Thailand) | Rare outside Asia. | Limited data | Curable with pangenotypic DAAs (SVR > 95%) [10] |
| GT7 | less than 1% | Central Africa | Recently identified. Very rare. | N/A | Presumed responsive to pangenotypic DAAs |
| GT8 | less than 1% | India, Pakistan | Recently identified. Very rare. | N/A | Presumed responsive to pangenotypic DAAs |
Clinical Implication of Genotyping: In the interferon era, genotype determined treatment duration and response rates. In the DAA era, pangenotypic regimens (sofosbuvir/velpatasvir, glecaprevir/pibrentasvir) are first-line regardless of genotype, eliminating the need for genotype-guided therapy in most cases. [10,24] However, genotyping remains useful for:
- Epidemiological tracking and transmission studies
- Identifying GT3 (higher HCC risk, closer surveillance post-SVR)
- Detecting resistance-associated substitutions (RAS) in treatment-experienced patients
- Resource-limited settings where genotype-specific regimens may be cheaper
Genotype 3: A Distinct Clinical Entity
GT3 deserves special emphasis due to its unique pathophysiology and clinical implications:
-
Hepatic Steatosis: GT3 directly induces steatosis via NS5A protein-mediated lipid droplet accumulation and impaired VLDL secretion, independent of metabolic risk factors. [21] Steatosis occurs in 70-80% of GT3 patients vs 40-50% in other genotypes.
-
Accelerated Fibrosis: GT3 patients progress to cirrhosis 30-40% faster than GT1, even after adjusting for alcohol and BMI. [22]
-
Increased HCC Risk: GT3 is associated with 2-3× higher HCC incidence compared to GT1, independent of cirrhosis status. HCC can occur in F3 fibrosis. [22] Post-SVR surveillance may be warranted in GT3 with F3 fibrosis.
-
NS5A Resistance: Baseline NS5A RAS (particularly Y93H) are more common in GT3 and can reduce SVR rates with ledipasvir- or daclatasvir-based regimens. Velpatasvir and pibrentasvir have higher barriers to resistance. [23]
-
Steatosis Reversal Post-SVR: Unlike metabolic steatosis, GT3-induced steatosis resolves in 70-80% of patients following SVR. [21]
Immune Evasion and Chronicity
HCV establishes chronic infection through multiple mechanisms:
- Quasispecies variation: High mutation rate allows viral escape from neutralizing antibodies and cytotoxic T lymphocytes (CTLs). [18]
- E2 envelope protein hypervariable regions (HVR1): Shield neutralization epitopes and undergo rapid mutation.
- Inhibition of interferon signaling: NS3/4A protease cleaves mitochondrial antiviral signaling protein (MAVS) and TIR-domain-containing adapter-inducing interferon-β (TRIF), blocking type I and III interferon responses. [25]
- Regulatory T cell induction: HCV core protein promotes T-regulatory (Treg) cell expansion, dampening adaptive immunity.
Fibrosis Progression
Chronic HCV infection drives progressive hepatic fibrosis via:
- Hepatocyte injury and apoptosis: Direct cytopathic effects and immune-mediated destruction release damage-associated molecular patterns (DAMPs).
- Hepatic stellate cell (HSC) activation: DAMPs, transforming growth factor-β (TGF-β), and platelet-derived growth factor (PDGF) activate quiescent HSCs into myofibroblasts, which deposit extracellular matrix (collagen I/III).
- Kupffer cell activation: Release of pro-inflammatory cytokines (TNF-α, IL-1β, IL-6) perpetuates inflammation and fibrogenesis.
- Steatosis: Genotype 3 directly induces hepatic steatosis via NS5A-mediated lipid droplet accumulation and impaired VLDL secretion. [21]
Fibrosis staging is assessed using the METAVIR score (F0 = no fibrosis, F4 = cirrhosis) or non-invasive methods (FibroScan, APRI, FIB-4). Progression rates vary: median time to cirrhosis is ~30 years, but 20-30% progress within 20 years. [7,9]
3. Epidemiology
Global Burden
| Metric | Value | Source |
|---|---|---|
| Global Prevalence (2020) | ~58 million chronically infected | WHO, 2022 [1] |
| Annual New Infections | ~1.5 million | WHO, 2022 [1] |
| HCV-Related Deaths (2019) | ~290,000 (cirrhosis and HCC) | WHO, 2022 [1] |
| Countries with Highest Burden | Egypt, Pakistan, China, Nigeria, India, Mongolia | Polaris Observatory, 2023 [2] |
Regional Prevalence
- Egypt: Historically highest prevalence (~15% in 1990s) due to iatrogenic transmission via schistosomiasis treatment campaigns. Reduced to ~4-5% following national screening and treatment programs (2014-2021). Predominantly GT4. [2]
- Europe: Prevalence ~0.5-2%, predominantly GT1 and GT3. IVDU is the primary transmission route.
- USA: ~2.4 million chronically infected. Baby boomers (born 1945-1965) disproportionately affected due to historical transfusion and IVDU exposure. GT1a most common. [26]
- Asia-Pacific: High burden in Pakistan (~7%), Mongolia (~6%), and Southeast Asia. Mixed genotypes (GT1, 3, 6).
- Australia: ~0.9% prevalence. GT3 most common (50-60%), followed by GT1. High burden in IVDU population. [2]
Transmission Routes
| Route | Relative Risk | Notes |
|---|---|---|
| Injection Drug Use (IVDU) | Highest | ~90% of new infections in UK and USA. Sharing needles, syringes, or paraphernalia (cookers, filters). [3,27] Prevalence among PWID: 40-80%. |
| Blood Transfusion / Blood Products | High (historical) | Major route pre-universal screening. Risk less than 1 in 1-2 million in screened countries post-1992. [4] Nucleic acid testing (NAT) since 1999 further reduced window period. |
| Needlestick Injury | 1.8% (95% CI 0.2-10%) | Healthcare workers. Risk higher if hollow-bore needle, deep penetration, source patient with high HCV RNA. [28] Post-exposure: no prophylaxis available; monitor HCV RNA at 4-6 weeks. |
| Vertical (Mother-to-Child) | 5-6% | Transmission occurs peripartum. Risk increased to ~10-20% if maternal HIV co-infection or high HCV RNA viral load > 10⁶ IU/mL. [29] No evidence that cesarean section reduces transmission unless obstetric indication. Breastfeeding is not contraindicated (unless cracked/bleeding nipples). |
| Sexual Transmission | Low | Heterosexual monogamous: less than 0.1% per year. Higher risk in MSM (men who have sex with men), especially HIV-positive MSM (~1-3% annually). [3,27] Risk factors: traumatic sexual practices, concurrent STIs, HIV co-infection. |
| Occupational (Non-needlestick) | Very Low | Intact skin exposure to HCV-contaminated blood: no transmission. Mucous membrane exposure: rare. |
| Tattoos/Piercings | Low | If non-sterile equipment. Higher risk in unregulated settings (prisons). |
| Household Contact | Very Low | Sharing razors or toothbrushes (theoretical risk). No transmission via casual contact, hugging, kissing, or sharing utensils. |
4. Natural History
Acute Hepatitis C
| Feature | Value | Notes |
|---|---|---|
| Incubation Period | 2-26 weeks (mean 6-7 weeks) | Time from exposure to symptom onset or HCV RNA detectability. |
| Symptomatic Presentation | 15-25% | Symptoms: jaundice, fatigue, nausea, RUQ pain, dark urine, pale stools. |
| Asymptomatic Presentation | 75-85% | Most acute infections are undiagnosed. [5] |
| ALT Elevation | Variable | Typically > 10× ULN if symptomatic; may be less than 2× ULN if asymptomatic. |
| Spontaneous Viral Clearance | 15-25% | Higher clearance rates if: symptomatic acute infection, younger age, female sex, favorable IL28B genotype (rs12979860 CC). [6,30] Clearance usually occurs within 12 weeks. |
| Progression to Chronic HCV | 75-85% | Defined as HCV RNA detectable ≥6 months post-infection. [5,6] |
Window Period: HCV RNA becomes detectable 1-2 weeks post-exposure. Anti-HCV antibodies appear 6-12 weeks post-infection. False-negative serology can occur in immunocompromised patients or during early acute infection.
Chronic Hepatitis C
| Feature | Value | Notes |
|---|---|---|
| Definition | HCV RNA positive ≥6 months | Indicates failure of spontaneous clearance. |
| Clinical Course | Asymptomatic for decades | Fatigue is the most common symptom, even in absence of cirrhosis. [9] |
| Histological Activity | Variable | Ranges from minimal hepatitis to severe necro-inflammation. |
| Fibrosis Progression | 20-30% cirrhosis over 20-30 years | Median progression time: ~30 years. Accelerated by alcohol (> 30 g/day), obesity (BMI > 30), HIV/HBV co-infection, diabetes, male sex, age > 40 at infection. [7,9] |
| Cirrhosis Decompensation | 3-6% per year once cirrhotic | Manifestations: ascites, variceal hemorrhage, hepatic encephalopathy, spontaneous bacterial peritonitis. |
| HCC Incidence (Cirrhosis) | 1-4% per year | Risk factors: male sex, age > 55, diabetes, obesity, AFP > 20 ng/mL, platelet count less than 150,000. [8,16] |
| HCC Incidence (No Cirrhosis) | less than 0.1% per year | Rare but reported, particularly in GT3 with advanced fibrosis (F3). [22] |
Co-morbidities Accelerating Progression:
- Alcohol: > 30 g/day (2 UK units) accelerates fibrosis 2-3 fold. Complete abstinence recommended. [9]
- Metabolic Syndrome: Obesity, diabetes, and NAFLD synergistically promote fibrosis.
- HIV Co-infection: Accelerates fibrosis progression and increases HCC risk. CD4 count less than 200 cells/μL worsens outcomes. [9]
- HBV Co-infection: Increases cirrhosis and HCC risk. HBV often suppressed but can reactivate during DAA therapy. [9,31]
Extrahepatic Manifestations
HCV is a systemic disease with multiple extrahepatic manifestations affecting ~40-75% of chronically infected patients. [32]
| Manifestation | Prevalence | Pathophysiology | Clinical Features |
|---|---|---|---|
| Mixed Cryoglobulinemia | 40-60% | Type II (monoclonal IgM RF + polyclonal IgG) or Type III (polyclonal). Immune complex deposition. | Palpable purpura (lower limbs), arthralgias, fatigue, peripheral neuropathy (mononeuritis multiplex), glomerulonephritis (MPGN Type I), hepatosplenomegaly. Low C4 complement. [32] |
| Membranoproliferative GN | 10-20% of cryoglobulinemia cases | Immune complex deposition in glomeruli. | Proteinuria, hematuria, renal impairment. May progress to ESRD. |
| B-Cell Non-Hodgkin Lymphoma | 2-10× increased risk | Chronic B-cell stimulation. | Marginal zone lymphoma, diffuse large B-cell lymphoma. |
| Porphyria Cutanea Tarda | 5-10% | Uroporphyrinogen decarboxylase deficiency exacerbated by HCV. | Photosensitivity, skin fragility, bullae on sun-exposed areas (dorsum of hands), hypertrichosis. |
| Lichen Planus | 5-35% | Unclear; possible immune-mediated. | Oral (reticular white plaques) or cutaneous (pruritic purple polygonal papules). |
| Insulin Resistance / Type 2 Diabetes | 2-3× increased risk | HCV core protein impairs insulin signaling. | May improve post-SVR. [9] |
| Sicca Syndrome | 10-30% | Autoimmune exocrinopathy (not Sjögren's). | Dry eyes, dry mouth. Anti-SSA/SSB negative. |
| Fatigue | 50-70% | Multifactorial: cytokine-mediated, not correlated with ALT or viral load. | Persistent even post-SVR in some patients. |
Treatment Impact: Most extrahepatic manifestations improve or resolve following SVR, particularly cryoglobulinemia (resolution in ~80%) and insulin resistance. [32]
5. Clinical Presentation
Acute Hepatitis C (Rarely Diagnosed)
| Symptom/Sign | Frequency | Notes |
|---|---|---|
| Asymptomatic | 75-85% | Majority undiagnosed unless identified through screening or contact tracing. |
| Jaundice | 15-25% | Peak bilirubin typically 50-200 μmol/L. Resolves over weeks. |
| Fatigue, Malaise | 50-60% (if symptomatic) | Non-specific. May persist. |
| Nausea, Anorexia | 30-50% | |
| RUQ Discomfort | 20-40% | Hepatomegaly on examination. |
| Dark Urine, Pale Stools | Correlates with jaundice | Conjugated hyperbilirubinemia. |
| Arthralgia, Myalgia | 10-20% | Flu-like illness. |
| Fever | Rare | More common with other viral hepatitides (HAV, HBV). |
Examination: Hepatomegaly (tender), mild jaundice. No stigmata of chronic liver disease.
Chronic Hepatitis C
| Symptom/Sign | Frequency | Notes |
|---|---|---|
| Asymptomatic | 50-70% (early stages) | Incidental finding on routine blood tests (elevated ALT) or screening. |
| Fatigue | 50-70% | Most common symptom. Severity does not correlate with ALT or fibrosis stage. [9] |
| RUQ Discomfort | 10-30% | Hepatomegaly. |
| Arthralgia | 10-30% | Often cryoglobulin-related. |
| Extrahepatic Features | Variable | Rash (purpura), sicca, neuropathy (see Extrahepatic Manifestations). |
Examination (Non-Cirrhotic): Normal or mild hepatomegaly. No stigmata of chronic liver disease.
Cirrhosis (Compensated and Decompensated)
Compensated Cirrhosis (F4, Child-Pugh A):
- Clinical: Often asymptomatic. Fatigue, mild RUQ discomfort.
- Examination: Firm hepatomegaly, splenomegaly (portal hypertension), mild spider naevi, palmar erythema.
- Laboratory: Thrombocytopenia (less than 150,000), hypoalbuminemia (mild), elevated INR (mild), elevated bilirubin (normal or slightly elevated).
Decompensated Cirrhosis (Child-Pugh B/C):
| Complication | Clinical Features |
|---|---|
| Ascites | Abdominal distension, shifting dullness, fluid thrill. May be complicated by SBP (fever, abdominal pain, confusion). |
| Variceal Bleeding | Hematemesis, melena, hemodynamic instability. Upper GI endoscopy shows esophageal or gastric varices. |
| Hepatic Encephalopathy | Confusion, asterixis, reversal of sleep-wake cycle. Graded 1-4 (West Haven criteria). Precipitants: infection, GI bleeding, constipation, sedatives, hypokalemia. |
| Jaundice | Scleral icterus, dark urine. Bilirubin > 50 μmol/L. |
| Hepatorenal Syndrome | Progressive renal impairment without other cause. Type 1 (rapid, less than 2 weeks) or Type 2 (slower, associated with refractory ascites). |
| Coagulopathy | Bruising, bleeding. Prolonged PT/INR (synthetic dysfunction). |
Examination: Jaundice, spider naevi (> 5 in superior vena cava distribution), palmar erythema, gynaecomastia, testicular atrophy, loss of axillary/pubic hair, Dupuytren's contracture (alcohol), caput medusae, ascites, splenomegaly, asterixis, fetor hepaticus, confusion.
Stigmata of IVDU: Track marks, scarring, abscesses (if relevant to risk factor history).
6. Investigations
Diagnostic Algorithm
SUSPECTED HCV EXPOSURE OR SCREENING INDICATION
(PWID, blood transfusion pre-1992, born in endemic region, HIV+, abnormal LFTs)
↓
1. SCREENING: Anti-HCV Antibody (ELISA)
┌───────────────┴───────────────┐
NEGATIVE POSITIVE
↓ ↓
No HCV infection 2. CONFIRMATORY: HCV RNA (PCR)
(If recent exposure ↓
less than 12 weeks, repeat ┌────────┴────────┐
in 3 months OR POSITIVE NEGATIVE
check HCV RNA) ↓ ↓
ACTIVE HCV PAST CLEARED
INFECTION INFECTION
↓ (Spontaneous or
CHRONIC HCV Post-Treatment)
(RNA + ≥6 months)
↓
3. ASSESSMENT FOR TREATMENT
├─ HCV Genotype (GT1-6) [NOW LESS CRITICAL WITH PANGENOTYPIC DAAs]
├─ HCV Viral Load (Baseline, not prognostic but monitored)
├─ Liver Fibrosis Assessment:
│ ├─ FibroScan (Transient Elastography): less than 7 kPa = F0-F1, 7-9.5 = F2, 9.5-12.5 = F3, > 12.5 = F4
│ ├─ FIB-4 Score: less than 1.45 = F0-F1, 1.45-3.25 = F2-F3, > 3.25 = F3-F4
│ ├─ APRI Score: less than 0.5 = F0-F1, 0.5-1.5 = F2-F3, > 1.5 = F3-F4
│ └─ Liver Biopsy (rarely needed)
├─ LFTs (ALT, AST, ALP, Bilirubin, Albumin, INR)
├─ Complete Blood Count (platelets—thrombocytopenia suggests portal hypertension)
├─ Renal Function (eGFR—affects DAA choice)
├─ HBV Serology (HBsAg, anti-HBc, anti-HBs)—risk of reactivation
├─ HIV Test (co-infection screening)
├─ AFP and USS Liver (baseline, HCC surveillance if cirrhotic)
└─ Pregnancy test (DAAs not recommended in pregnancy)
↓
4. TREAT WITH DIRECT-ACTING ANTIVIRALS (DAAs)
↓
5. SVR12 ASSESSMENT (12 weeks post-treatment)
HCV RNA undetectable = CURE
Serological and Virological Tests
| Test | Method | Interpretation | Notes |
|---|---|---|---|
| Anti-HCV Antibody | ELISA (3rd generation) | Screening test. Sensitivity > 99%, Specificity > 99%. | Positive = Exposure (current or past). Does NOT differentiate active vs cleared infection. Remains positive for life even after clearance/cure. False negatives: immunocompromised, acute infection (less than 12 weeks), hemodialysis. |
| HCV RNA (Qualitative) | RT-PCR | Detectable = active infection. Undetectable = cleared or cured. | Confirmatory test. Detects viral replication. Window period: detectable 1-2 weeks post-exposure. |
| HCV RNA (Quantitative) | Real-time RT-PCR | Viral load (IU/mL). Lower limit of quantification (LLOQ): 15-25 IU/mL. | Baseline pre-treatment and SVR12 assessment. Not prognostic for fibrosis or treatment response (unlike HBV). Used to confirm SVR (undetectable = less than 15 IU/mL). [12] |
| HCV Genotype | Sequencing or Line Probe Assay | GT1-6, subtypes (e.g., 1a, 1b, 3a). | Historically guided treatment; less critical with pangenotypic DAAs. Still performed for epidemiological tracking and identifying GT3. [10] |
| HCV Core Antigen | ELISA | Alternative to HCV RNA in resource-limited settings. | Lower sensitivity than RNA PCR but cheaper. Not widely used in high-income countries. |
| Resistance-Associated Substitutions (RAS) | Sequencing (NS3, NS5A, NS5B) | Detect mutations conferring DAA resistance. | Not routinely performed in treatment-naive patients. Consider in DAA-experienced patients or GT3 with prior treatment failure. [33] |
Interpretation of Serology/Virology Results
| Anti-HCV | HCV RNA | HCV Core Ag | Interpretation | Clinical Action |
|---|---|---|---|---|
| Negative | Negative | Negative | No HCV infection OR Very early acute infection (window period) | If high-risk exposure less than 12 weeks, repeat in 3 months OR check HCV RNA. |
| Positive | Positive | Positive | Active HCV infection (acute or chronic) | Proceed to treatment assessment. |
| Positive | Negative | Negative | Past cleared infection (spontaneous or treated) OR Rarely: false-positive antibody (low-risk populations) | No treatment needed. Counsel on re-exposure risk. If immunocompromised or suspected false positive, repeat RNA. |
| Negative | Positive | Positive | Acute infection (window period) OR Immunocompromised (antibody-negative) | Re-check anti-HCV in 4-12 weeks (should seroconvert). Treat if chronic. |
Liver Fibrosis Assessment: Non-Invasive vs Biopsy
The assessment of liver fibrosis is critical for prognosis, HCC surveillance decisions, and (historically) treatment duration selection. Non-invasive tests have largely replaced liver biopsy in routine practice.
FibroScan (Transient Elastography)
Principle: Measures liver stiffness (elastic modulus) in kilopascals (kPa) using ultrasound-based shear wave propagation. Stiffer liver = more fibrosis.
Technique:
- Patient fasting 3-4 hours (food intake increases stiffness)
- Right intercostal approach, patient supine, right arm elevated
- 10 valid measurements obtained; median reported
- Success rate > 90% in experienced hands
Cutoff Values (HCV-Specific): [34,35]
| Fibrosis Stage | FibroScan (kPa) | Sensitivity | Specificity | Notes |
|---|---|---|---|---|
| F0-F1 (None-Mild) | less than 7.0 | 85-90% | 80-85% | Excellent negative predictive value (NPV ~95%) for excluding significant fibrosis |
| F2 (Moderate) | 7.0-9.5 | 70-75% | 75-80% | Less accurate for intermediate fibrosis |
| F3 (Advanced) | 9.5-12.5 | 75-80% | 80-85% | |
| F4 (Cirrhosis) | > 12.5 | 90-95% | 90-95% | Excellent positive predictive value (PPV ~90%) for cirrhosis |
Limitations:
- Obesity (BMI > 30): Failure rate 15-20%. XL probe available for obese patients.
- Ascites: Cannot be performed (ultrasound does not penetrate fluid).
- Acute hepatitis: ALT > 5× ULN transiently increases stiffness (false elevation).
- Cholestasis: ALP/bilirubin elevation increases stiffness independent of fibrosis.
- Congestive hepatopathy: Right heart failure increases stiffness.
- Food intake: Increases stiffness by 10-20% for 2-3 hours.
- Interobserver variability: Requires trained operators.
Post-SVR: FibroScan values decrease by 20-30% within 12-24 months post-SVR due to reduced inflammation, even in cirrhotic patients. [36] This does not represent true fibrosis regression (structural remodeling persists), but improved hepatic function.
FIB-4 Index
Formula:
FIB-4 = (Age [years] × AST [U/L]) / (Platelets [10⁹/L] × √ALT [U/L])
Cutoffs: [37]
| FIB-4 Score | Fibrosis Stage | Notes |
|---|---|---|
| less than 1.45 | F0-F1 (None-Mild) | NPV ~90% for excluding advanced fibrosis (F3-F4). Best use: rule-out advanced fibrosis. |
| 1.45-3.25 | F2-F3 (Moderate-Advanced) | Indeterminate. Requires additional testing (FibroScan or biopsy). |
| > 3.25 | F3-F4 (Advanced-Cirrhosis) | PPV ~70-80% for advanced fibrosis. |
Limitations:
- Age-dependent: Less accurate less than 35 years (overestimates fibrosis) or > 65 years (underestimates fibrosis). [37]
- Thrombocytopenia from other causes (ITP, splenomegaly, hematologic disorders) falsely elevates score.
- Not validated post-SVR: Platelets may remain low despite virological cure.
APRI (AST-to-Platelet Ratio Index)
Formula:
APRI = (AST [U/L] / ULN) × 100 / Platelets [10⁹/L]
Cutoffs: [38]
| APRI Score | Fibrosis Stage | Notes |
|---|---|---|
| less than 0.5 | F0-F1 | NPV ~85% for excluding significant fibrosis. |
| 0.5-1.5 | F2-F3 | Indeterminate. |
| > 1.5 | F3-F4 | PPV ~80% for cirrhosis. > 2.0 strongly suggests cirrhosis. |
Limitations: Less sensitive than FIB-4 or FibroScan for intermediate fibrosis (F2-F3). Best for excluding significant fibrosis or confirming cirrhosis.
ELF Test (Enhanced Liver Fibrosis)
Principle: Serum biomarker panel measuring extracellular matrix turnover:
- Hyaluronic acid (HA)
- Procollagen III N-terminal peptide (PIIINP)
- Tissue inhibitor of metalloproteinase 1 (TIMP-1)
Cutoffs: [39]
| ELF Score | Fibrosis Stage | Notes |
|---|---|---|
| less than 7.7 | F0-F1 | |
| 7.7-9.8 | F2 | |
| 9.8-11.3 | F3 | |
| > 11.3 | F4 (Cirrhosis) |
Advantages: Not affected by obesity, ascites, or operator variability. Validated for HCV.
Limitations: Expensive. Requires specialized laboratory. Not widely available outside UK and some European countries.
Liver Biopsy
Indications (now rare):
- Uncertain diagnosis (possible alternative liver disease: NASH, autoimmune hepatitis, hemochromatosis, Wilson's disease)
- Discordant non-invasive tests (e.g., FibroScan F4 but FIB-4 less than 1.45)
- Clinical suspicion of advanced fibrosis despite low non-invasive scores
- Research/clinical trials
Technique: Percutaneous (ultrasound-guided) or transjugular (if ascites/coagulopathy).
Complications: [40]
- Pain: 30%
- Bleeding requiring transfusion: 0.3%
- Death: 0.01-0.03%
- Pneumothorax, hemothorax, bile peritonitis: less than 0.1% each
Staging Systems:
- METAVIR (most common for HCV): F0 = no fibrosis, F1 = portal fibrosis without septa, F2 = few septa, F3 = many septa without cirrhosis, F4 = cirrhosis
- Ishak (0-6 scale): More granular, used in research
Limitations:
- Sampling error: Biopsy samples 1/50,000th of liver. Inter-sample variability 20-30%.
- Observer variability: Inter-pathologist agreement (kappa) 0.5-0.7 (moderate).
- Patient acceptance: Invasive, painful, anxiety.
- Not repeatable: Cannot monitor fibrosis progression serially.
Consensus: Non-invasive tests (FibroScan, FIB-4, APRI) have replaced biopsy in > 90% of HCV cases. [10,34]
Comparative Accuracy for Cirrhosis Diagnosis
| Modality | Sensitivity for F4 | Specificity for F4 | AUROC | Advantages | Disadvantages |
|---|---|---|---|---|---|
| FibroScan | 85-95% | 85-95% | 0.90-0.95 | Non-invasive, point-of-care, reproducible | Obesity, ascites, operator-dependent |
| FIB-4 (> 3.25) | 70-75% | 85-90% | 0.80-0.85 | Free, widely available, no equipment | Age-dependent, less accurate F2-F3 |
| APRI (> 1.5) | 70-80% | 80-85% | 0.75-0.85 | Free, widely available | Less accurate F2-F3 |
| ELF (> 11.3) | 80-90% | 85-90% | 0.85-0.90 | Not affected by obesity/ascites | Expensive, limited availability |
| Liver Biopsy | 80-85% (sampling error) | 95-98% | Gold standard (with caveats) | Histological detail | Invasive, complications, sampling error |
Clinical Approach: Use FibroScan + FIB-4 in combination. If concordant, no biopsy needed. If discordant or indeterminate, consider ELF test or liver biopsy. [34,39]
Baseline Laboratory Investigations
| Test | Purpose | Abnormal Findings |
|---|---|---|
| LFTs | Hepatic necro-inflammation, synthetic function | ALT/AST elevated (often less than 5× ULN, fluctuates). Normal ALT does NOT exclude chronic HCV. AST:ALT ratio > 1 suggests cirrhosis. ALP normal or mildly elevated. Bilirubin normal unless cirrhotic. Albumin low if cirrhotic. INR prolonged if cirrhotic. |
| CBC | Thrombocytopenia (portal hypertension), anemia (hypersplenism, bleeding) | Platelets less than 150,000 suggests portal hypertension (cirrhosis). Leukopenia and anemia if hypersplenism. |
| Renal Function | Baseline eGFR (affects DAA choice) | Sofosbuvir contraindicated if eGFR less than 30 mL/min (historical; recent data suggest safety with dose reduction). Glecaprevir/pibrentasvir safe in all stages of CKD including dialysis. [41] |
| HBV Serology | Screen for co-infection and risk of reactivation | HBsAg +, anti-HBc +, anti-HBs - = Active HBV (co-infection). Anti-HBc +, HBsAg - = Past HBV (risk of reactivation during DAA therapy—monitor HBV DNA). [31] |
| HIV Test | Co-infection screening | Common in PWID. Affects treatment urgency and HCC surveillance. |
| AFP (Alpha-Fetoprotein) | HCC screening baseline | Elevated (> 20 ng/mL) may indicate HCC. Sensitivity ~60%, specificity ~90%. Used in combination with USS. |
| Cryoglobulins | If clinical suspicion of cryoglobulinemia | Positive in 40-60% of HCV patients. Symptomatic in 5-10%. Low C4 complement. |
| ANA, ASMA, IgG | Exclude autoimmune hepatitis | Low-titer ANA/ASMA can occur in HCV (10-20%). High-titer (> 1:320) or elevated IgG suggests overlap or alternative diagnosis. |
HCC Surveillance (If Cirrhotic)
Indication: All patients with cirrhosis (F4), regardless of SVR status.
| Modality | Frequency | Sensitivity | Notes |
|---|---|---|---|
| Liver USS | Every 6 months | 60-80% | First-line. Non-invasive, widely available. Limitations: obesity, operator-dependent. |
| AFP | Every 6 months (with USS) | 60-70% | Adjunct to USS. Threshold > 20 ng/mL warrants further investigation. Not recommended alone. |
| CT/MRI | If USS inadequate or lesion detected | > 90% | Triphasic CT or dynamic MRI with contrast. Diagnostic for HCC if arterial enhancement + washout. |
Post-SVR HCC Risk: Reduced by 70-80% but NOT eliminated. Annual incidence post-SVR in cirrhotics: 0.3-1.5% (vs 1-4% untreated). Surveillance continues indefinitely. [16,17]
Risk Factors for HCC Post-SVR: [16,17]
- Age > 55 years
- Male sex
- Diabetes mellitus
- Obesity (BMI > 30)
- Baseline AFP > 20 ng/mL
- Platelet count less than 150,000/μL
- Baseline liver stiffness > 20 kPa
- GT3 (independent risk factor)
7. Management
Treatment Principles
Universal Treatment Recommendation: All patients with chronic HCV (detectable HCV RNA ≥6 months) should be offered treatment, regardless of fibrosis stage, age, or co-morbidities. Active IVDU is NOT a contraindication. [10,24,27]
Goals of Treatment:
- Virological cure (SVR12: undetectable HCV RNA 12 weeks post-treatment)
- Prevention of cirrhosis, HCC, and liver-related mortality
- Prevention of extrahepatic complications
- Reduction of onward transmission ("treatment as prevention")
SVR12: Definition, Validation, and Clinical Significance
Sustained Virological Response (SVR) is the primary endpoint of HCV treatment and is considered virological cure.
Evolution of SVR Definition
| Era | Endpoint | Rationale |
|---|---|---|
| Interferon Era (1990s-2011) | SVR24: Undetectable HCV RNA 24 weeks post-treatment | Standard endpoint in RCTs. Relapse after SVR24 rare (less than 1%). |
| DAA Era (2011-present) | SVR12: Undetectable HCV RNA 12 weeks post-treatment | Validated against SVR24. Allows earlier confirmation of cure. [12,13] |
SVR12 Validation Studies
Multiple large cohort studies have validated SVR12 as equivalent to SVR24:
- Yoshida et al. (2015): Meta-analysis of 29 DAA trials (n=12,535). SVR12 vs SVR24 concordance: 99.7%. [12]
- Sarrazin et al. (2012): Boceprevir/telaprevir trials (n=1,245). SVR12 vs SVR24 concordance: 99.5%. [13]
- FDA Guidance (2013): Accepted SVR12 as primary endpoint for DAA approval. [42]
Conclusion: SVR12 = SVR24 in > 99% of cases. Relapse after SVR12 is exceptionally rare (less than 1%). [12,13]
Lower Limit of Quantification (LLOQ)
Modern HCV RNA assays detect down to 15-25 IU/mL (LLOQ). SVR is defined as HCV RNA below LLOQ (i.e., undetectable). Some assays use "target not detected" (TND) vs "target detected but not quantified" (below LLOQ but above lower limit of detection [LLOD]). Both are considered SVR. [12]
Clinical Significance of SVR
| Outcome | Impact of SVR | Evidence |
|---|---|---|
| Relapse Risk | less than 1% after SVR12 | Meta-analysis (Yoshida 2015) [12] |
| Liver-Related Mortality | Reduced by 70-80% | Meta-analysis (Nahon 2017) [43] |
| HCC Incidence (Cirrhosis) | Reduced from 1-4%/year to 0.3-1.5%/year (70-80% reduction but not eliminated) | Ioannou 2021 [16], Pan 2024 [17] |
| HCC Incidence (No Cirrhosis) | Reduced to near-general population levels (less than 0.1%/year) | Ioannou 2021 [16] |
| Hepatic Decompensation | Reduced by 70-90% | Nahon 2017 [43] |
| Fibrosis Regression | 40-60% of F3 regress to F2 or lower over 3-5 years | Knop 2016 [36] |
| Recompensation (Decompensated Cirrhosis) | 40-50% experience improved MELD, albumin, reduction/resolution of ascites | Premkumar 2024 [44] |
| Extrahepatic Manifestations | Cryoglobulinemia resolves in ~80%, insulin resistance improves | Gulli 2018 [32] |
| Quality of Life | Significant improvement in fatigue, physical function | Younossi 2017 [45] |
Interpretation: SVR12 is a durable cure with long-term clinical benefits. It is not sterilizing immunity—re-infection is possible if re-exposed.
On-Treatment Viral Monitoring
Current Guidelines: Do NOT routinely check HCV RNA during DAA treatment. [10,24]
Rationale:
- SVR rates > 95% regardless of on-treatment viral kinetics
- On-treatment HCV RNA does not predict SVR
- Detectable HCV RNA at week 4 does NOT indicate treatment failure (delayed viral clearance is common)
- Adds cost without clinical benefit
Exception: Research studies or suspected non-adherence.
End-of-Treatment (EOT) HCV RNA
Current Guidelines: EOT HCV RNA is optional and not required. [10,24]
Rationale:
- Detectable HCV RNA at EOT does not exclude SVR12 (delayed clearance can occur)
- Undetectable HCV RNA at EOT does not guarantee SVR12 (late relapse possible, though rare)
- SVR12 is the definitive endpoint
Clinical Implication: Many programs skip EOT testing and proceed directly to SVR12 at week 12 post-treatment.
Direct-Acting Antivirals (DAAs): Mechanisms and Pharmacology
DAAs target specific viral non-structural proteins essential for HCV replication. Three classes are in clinical use:
Class 1: NS5B Polymerase Inhibitors
Target: NS5B RNA-dependent RNA polymerase (essential for viral RNA replication)
| Drug | Subclass | Mechanism | Barrier to Resistance | Genotype Activity | Renal/Hepatic Metabolism |
|---|---|---|---|---|---|
| Sofosbuvir | Nucleotide analog | Chain termination (mimics uridine, incorporated into RNA, terminates elongation) | High (conserved active site, high fitness cost of resistance) | Pangenotypic (GT1-6) | Renal excretion (GS-331007 metabolite). Avoid if eGFR less than 30 (historical; recent data suggest safety). [41,46] |
| Dasabuvir | Non-nucleoside allosteric | Binds distant site, conformational change, inhibits polymerase | Low-Moderate | GT1 only | Hepatic (CYP450) |
Resistance-Associated Substitutions (RAS): S282T (NS5B) confers resistance to sofosbuvir but is extremely rare (less than 1% prevalence) and has high fitness cost (virus replicates poorly). [33]
Clinical Use: Sofosbuvir is the backbone of most modern DAA regimens due to pangenotypic activity and high barrier to resistance. Dasabuvir is obsolete (replaced by pangenotypic options).
Class 2: NS5A Inhibitors
Target: NS5A protein (multifunctional: replication complex assembly, virion assembly, modulation of host immunity)
| Drug | Genotype Activity | Barrier to Resistance | Notes |
|---|---|---|---|
| Ledipasvir | GT1, 4, 5, 6 | Low (single mutations confer resistance: Y93H, L31M, Q30R) | Used in sofosbuvir/ledipasvir (Harvoni). GT1b more resistant than GT1a. [47] |
| Daclatasvir | GT1, 2, 3, 4 | Low | No longer widely used (replaced by velpatasvir, pibrentasvir). GT3: Y93H common. |
| Velpatasvir | Pangenotypic (GT1-6) | Moderate (higher barrier than ledipasvir/daclatasvir; retains activity against many RAS) | Used in sofosbuvir/velpatasvir (Epclusa). Second-generation NS5A inhibitor. [10,48] |
| Pibrentasvir | Pangenotypic (GT1-6) | Moderate-High (retains activity against many ledipasvir/daclatasvir RAS) | Used in glecaprevir/pibrentasvir (Maviret). Most potent NS5A inhibitor in vitro. [11,49] |
| Elbasvir | GT1, 4 | Low-Moderate | Used in elbasvir/grazoprevir (Zepatier). Baseline NS5A RAS testing recommended for GT1a. [10] |
Resistance-Associated Substitutions (RAS): NS5A RAS are the most common cause of DAA failure. Key mutations: [33]
- Y93H/N (GT1b, GT3): Confers high-level resistance to ledipasvir, daclatasvir. Velpatasvir/pibrentasvir retain partial activity.
- L31M, Q30R (GT1a): Reduce ledipasvir susceptibility.
- A30K (GT3): Resistance to daclatasvir/ledipasvir.
Clinical Implication: Baseline NS5A RAS testing is not routinely recommended in treatment-naive patients, as pangenotypic regimens (SOF/VEL, GLE/PIB) retain activity. [10] RAS testing considered in DAA-experienced patients with prior NS5A exposure.
Class 3: NS3/4A Protease Inhibitors
Target: NS3/4A serine protease (cleaves viral polyprotein into mature proteins)
| Drug | Genotype Activity | Barrier to Resistance | Notes |
|---|---|---|---|
| Telaprevir, Boceprevir | GT1 | Low | Obsolete (first-generation, replaced by DAAs). |
| Simeprevir | GT1, 4 | Low-Moderate | Largely replaced by pangenotypic options. |
| Paritaprevir | GT1, 4 | Low-Moderate | Used in paritaprevir/ritonavir/ombitasvir + dasabuvir (Viekira Pak). Obsolete. |
| Grazoprevir | GT1, 4 | Moderate | Used in elbasvir/grazoprevir (Zepatier). [10] |
| Glecaprevir | Pangenotypic (GT1-6) | Moderate-High (retains activity against many first-generation protease inhibitor RAS) | Used in glecaprevir/pibrentasvir (Maviret). Second-generation protease inhibitor. [11,49] |
| Voxilaprevir | Pangenotypic (GT1-6) | High (retains activity against glecaprevir RAS) | Used in sofosbuvir/velpatasvir/voxilaprevir (Vosevi) for treatment-experienced patients. [50] |
Resistance-Associated Substitutions (RAS): NS3 RAS: Q80K (GT1a), D168V/A, R155K. [33]
Clinical Implication: Glecaprevir (in GLE/PIB) is second-generation with improved resistance profile. Voxilaprevir (in SOF/VEL/VOX) is reserved for salvage therapy.
Contraindication in Decompensated Cirrhosis: NS3/4A protease inhibitors are contraindicated in Child-Pugh B/C cirrhosis due to hepatotoxicity risk (worsening liver function, hepatic decompensation). [10,24] Sofosbuvir/velpatasvir (no protease inhibitor) is first-line in decompensated cirrhosis.
First-Line Pangenotypic DAA Regimens
| Regimen | Components | Duration | Genotype Coverage | SVR Rate | Key Features |
|---|---|---|---|---|---|
| Sofosbuvir/Velpatasvir (Epclusa) | SOF 400 mg + VEL 100 mg once daily | 12 weeks (treatment-naive, all fibrosis stages) | All (GT1-6) | 95-99% | First pangenotypic single-tablet regimen. Well-tolerated. Contraindicated if eGFR less than 30 (historical; emerging data suggest safety). Safe in compensated cirrhosis. First-line in decompensated cirrhosis (Child-Pugh B/C) (no protease inhibitor). [10,48] |
| Glecaprevir/Pibrentasvir (Maviret) | GLE 100 mg + PIB 40 mg (3 tablets) once daily | 8 weeks (treatment-naive, non-cirrhotic) 12 weeks (treatment-naive, cirrhotic) 16 weeks (treatment-experienced, cirrhotic) | All (GT1-6) | 95-99% | Once daily. Safe in CKD (all stages including dialysis)—no dose adjustment needed. [11,41,49] Avoid in decompensated cirrhosis (Child-Pugh B/C)—protease inhibitor contraindication. Shortest duration (8 weeks) in non-cirrhotic treatment-naive. |
Other Regimens (genotype-specific, now second-line):
- Sofosbuvir/Ledipasvir (Harvoni): SOF 400 mg + LED 90 mg once daily. GT1, 4, 5, 6. 8-12 weeks. SVR 94-99%. [47] Largely replaced by SOF/VEL (pangenotypic).
- Elbasvir/Grazoprevir (Zepatier): ELB 50 mg + GRZ 100 mg once daily. GT1, 4. 12 weeks. SVR 95-97%. Baseline NS5A RAS testing recommended for GT1a. [10]
Treatment Duration by Clinical Scenario
| Clinical Scenario | Recommended Regimen | Duration | SVR Rate |
|---|---|---|---|
| Treatment-naive, no cirrhosis, GT1-6 | GLE/PIB (first choice if eGFR less than 30 or dialysis) SOF/VEL | 8 weeks 12 weeks | 95-99% [11] 95-99% [48] |
| Treatment-naive, compensated cirrhosis (Child-Pugh A), GT1-6 | GLE/PIB SOF/VEL | 12 weeks 12 weeks | 95-99% [11] 95-99% [48] |
| Treatment-naive, GT3, cirrhosis | SOF/VEL GLE/PIB | 12 weeks 12 weeks | 95-96% [23,48] 95-98% [49] |
| Treatment-experienced (prior interferon ± ribavirin), no cirrhosis | SOF/VEL GLE/PIB | 12 weeks 12 weeks | 95-99% [48] 95-99% [49] |
| Treatment-experienced (prior interferon ± ribavirin), compensated cirrhosis | SOF/VEL GLE/PIB | 12 weeks 12 weeks | 94-98% [48] 95-99% [49] |
| Treatment-experienced (prior DAA failure), no cirrhosis | SOF/VEL/VOX | 12 weeks | 96-98% [50] |
| Treatment-experienced (prior DAA failure), compensated cirrhosis | SOF/VEL/VOX ± ribavirin | 12 weeks | 96-98% [50] (ribavirin considered if NS5A RAS + cirrhosis) |
| Decompensated cirrhosis (Child-Pugh B/C) | SOF/VEL (avoid protease inhibitors) ± ribavirin | 12 weeks | 85-95% [10,48] (± ribavirin if Child-Pugh C or prior treatment failure) |
| Severe renal impairment (eGFR less than 30) or dialysis | GLE/PIB (first-line, no dose adjustment) SOF/VEL (emerging data support use; consult specialist) | 8-12 weeks | 95-99% [41,49] |
| HIV/HCV co-infection | Same as HCV mono-infection (check drug-drug interactions with ART) | 8-12 weeks | 95-99% [10,27] |
| Active PWID | SOF/VEL or GLE/PIB (directly observed therapy or peer support if adherence concern) | 8-12 weeks | 95-99% (if adherent) [27] |
Treatment-Experienced Patients: Prior Interferon vs Prior DAA Failure
Prior Interferon ± Ribavirin Failure
Definition: Patients who received pegylated interferon (PEG-IFN) ± ribavirin and either:
- Null response: less than 2 log₁₀ decline in HCV RNA at week 12
- Partial response: ≥2 log₁₀ decline but never undetectable
- Relapse: Undetectable on-treatment but recurred post-treatment
Management: Treat as treatment-naive with pangenotypic DAAs. Interferon failure does NOT predict DAA failure (different mechanisms of action). [10,24]
| Regimen | Duration | SVR Rate |
|---|---|---|
| SOF/VEL | 12 weeks | 94-99% [48] |
| GLE/PIB | 12 weeks (16 weeks if cirrhotic) | 95-99% [49] |
No need for resistance testing or extended duration (beyond 12 weeks in cirrhosis). SVR rates equivalent to treatment-naive. [10]
Prior DAA Failure
Definition: Patients who received DAAs and either:
- Virological failure on-treatment: Detectable HCV RNA at end-of-treatment (EOT)
- Relapse: Undetectable at EOT but detectable post-treatment (before SVR12)
Resistance Testing: Consider sequencing NS3, NS5A, NS5B to detect RAS, particularly if prior NS5A inhibitor exposure (ledipasvir, daclatasvir, velpatasvir, pibrentasvir). [33] However, not routinely required if using SOF/VEL/VOX (designed for DAA-experienced patients).
Management:
| Prior DAA Class(es) | Recommended Regimen | Duration | SVR Rate | Notes |
|---|---|---|---|---|
| NS5A inhibitor failure (LED, DCV, VEL, PIB) | SOF/VEL/VOX ± ribavirin | 12 weeks | 96-98% [50] | Ribavirin (1000-1200 mg/day) added if NS5A RAS + cirrhosis. Voxilaprevir retains activity against most NS5A RAS. |
| Protease inhibitor failure (without NS5A) | SOF/VEL or GLE/PIB | 12 weeks | 95-98% [10] | If no prior NS5A exposure, standard pangenotypic regimen effective. |
| SOF/VEL failure | SOF/VEL/VOX + ribavirin | 12 weeks | 96-98% [50] | Voxilaprevir (next-generation protease inhibitor) + ribavirin. |
| GLE/PIB failure | SOF/VEL/VOX + ribavirin | 12 weeks | 95-98% [50] | Switch classes (sofosbuvir-based). |
Salvage Therapy: For multiple DAA failures, consider:
- SOF/VEL/VOX + ribavirin for 24 weeks (expert consultation recommended)
- Liver transplant evaluation if decompensated cirrhosis
Re-infection vs Relapse: Distinguish by genotype/subtype sequencing. If genotype changes, re-infection (treat as treatment-naive). If identical, relapse (treat as DAA-experienced).
Special Populations and Considerations
Decompensated Cirrhosis (Child-Pugh B/C)
Regimen: Sofosbuvir/Velpatasvir 12 weeks ± ribavirin (if Child-Pugh C or prior treatment failure). [10,48]
Avoid: NS3/4A protease inhibitors (glecaprevir, grazoprevir, paritaprevir)—risk of hepatotoxicity and worsening liver function. [10,24]
Ribavirin: Weight-based dosing (1000 mg if less than 75 kg, 1200 mg if ≥75 kg), but reduce dose or avoid if renal impairment (eGFR less than 50) or severe anemia. Monitor hemoglobin (ribavirin causes hemolytic anemia in 10-20%).
Monitoring: Weekly LFTs for first 4 weeks, then 2-weekly. Risk of hepatic decompensation during treatment (~5-10%). Early transplant assessment if MELD > 18.
Post-SVR Recompensation: 40-50% of decompensated patients experience "recompensation" following SVR: [44]
- Improved MELD score (mean reduction 2-4 points)
- Increased albumin
- Reduced ascites (30-40% resolution)
- Improved hepatic encephalopathy
- Some patients delisted from transplant
Liver Transplant: DAA treatment can be given:
- Pre-transplant: May stabilize or improve liver function, potentially delisting some patients. SVR reduces post-transplant HCV recurrence.
- Post-transplant: If recurrent HCV. High SVR rates (> 95%) with DAAs post-transplant. Drug-drug interactions with immunosuppressants (tacrolimus, cyclosporine) require monitoring.
HBV/HCV Co-infection
Prevalence: 5-10% of HCV-positive patients (higher in Asia, endemic regions). [9,31]
Risk: HBV reactivation during or after DAA treatment (~25% if HBsAg-positive, 2-5% if anti-HBc-positive/HBsAg-negative). [31] Mechanism unclear (possibly immune reconstitution or HCV suppression allowing HBV replication).
Management: [31]
| HBV Serology | HBV DNA | Management |
|---|---|---|
| HBsAg-positive (active co-infection) | Any level | Start HBV antiviral (entecavir 0.5 mg daily or tenofovir 245 mg daily) before or concurrent with DAA therapy. Continue HBV treatment per HBV guidelines (typically indefinite if HBsAg-positive). Monitor HBV DNA, ALT during and post-DAA. |
| Anti-HBc-positive, HBsAg-negative (past HBV) | Undetectable | Monitor HBV DNA at baseline, weeks 4, 8, 12 during DAA treatment, and post-treatment weeks 12, 24. If HBV DNA becomes detectable or ALT flare (> 2× ULN + HBV DNA > 2000 IU/mL), start HBV antiviral. [31] |
| Anti-HBs-positive (immune) | N/A | No HBV reactivation risk. Proceed with DAA. |
Vaccination: If HBV-susceptible (anti-HBs negative, anti-HBc negative), offer HBV vaccination.
Treatment of HCV in HBV/HCV Co-infection: Use standard pangenotypic DAA regimens (SOF/VEL or GLE/PIB). SVR rates equivalent to HCV mono-infection. [31]
HIV/HCV Co-infection
Prevalence: 20-30% of HIV-positive individuals (higher in PWID and MSM). [9,27]
Natural History: HIV accelerates HCV fibrosis progression (2-3× faster). Higher HCC risk. Untreated HIV worsens outcomes. [9]
Treatment: Same DAA regimens as HIV-negative patients. SVR rates similar (> 95%) if HIV virologically suppressed on antiretroviral therapy (ART). [10,27]
Drug-Drug Interactions: Critical to check DAA-ART interactions. [51]
| DAA Regimen | ART Interactions |
|---|---|
| SOF/VEL | Avoid with efavirenz (reduces velpatasvir levels by 50%). Safe with most other ART (integrase inhibitors, tenofovir, emtricitabine, boosted darunavir). |
| GLE/PIB | Avoid with efavirenz, etravirine, nevirapine (reduce glecaprevir/pibrentasvir levels). Avoid with atazanavir, darunavir (increase pibrentasvir levels). Safe with most integrase inhibitors (dolutegravir, bictegravir, raltegravir). [51] |
Resource: Liverpool HEP Drug Interactions Checker (www.hep-druginteractions.org).
Monitoring: CD4 count, HIV viral load (should remain suppressed during HCV treatment).
CD4 Count: No minimum CD4 requirement for DAA treatment. Treat HCV regardless of CD4 count (immune reconstitution post-ART improves HCV treatment response).
Chronic Kidney Disease (CKD) and Dialysis
eGFR ≥30 mL/min: Sofosbuvir-based regimens safe. No dose adjustment needed. [10,46]
eGFR less than 30 mL/min or dialysis: [41]
| Regimen | Safety | Dosing | Notes |
|---|---|---|---|
| Glecaprevir/Pibrentasvir | Safe | No dose adjustment | First-line. Validated in CKD Stage 4-5 and dialysis (hemodialysis, peritoneal dialysis). SVR > 95%. [41,49] |
| Sofosbuvir-based (SOF/VEL, SOF/LED) | Historically avoided | Standard dose (emerging data) | Historical concern: renal excretion of GS-331007 metabolite (accumulates 20-fold if eGFR less than 30). Recent studies suggest safety with standard dose or reduced dose (200 mg) under specialist supervision. [41,52] Consult nephrology/hepatology. |
Hemodialysis: GLE/PIB is not dialyzable (high protein binding). Dose timing not critical. [41]
Post-SVR: Monitor renal function. Some improvement in eGFR post-SVR (reduced inflammation, improved cryoglobulinemia-related GN). [32]
Active Injection Drug Use (PWID)
Treat without delay: Active IVDU is NOT a contraindication to DAA therapy. [10,24,27]
Evidence: Adherence rates and SVR rates are equivalent to non-PWID if supported appropriately (> 95% SVR). [27,53]
Re-infection risk: 2-5% per year post-SVR if ongoing IVDU. [27,53] Counsel on harm reduction. Re-treatment achievable if re-infected (treat as new infection).
Harm reduction: [27,53]
- Needle and syringe programs (NSP)
- Opioid substitution therapy (OST: methadone, buprenorphine)
- Supervised consumption sites
- Peer support and education
Treatment as prevention: Treating PWID reduces community HCV prevalence and incidence (mathematical models show elimination achievable with ≥80% screening and treatment coverage). [27,53]
Directly observed therapy (DOT): Consider if adherence concern (e.g., unstable housing, active substance use disorder). Pharmacy-based DOT or peer-delivered DOT effective. [53]
Drug-Drug Interactions: Methadone, buprenorphine: no dose adjustment needed with DAAs. [51]
Pregnancy and Lactation
Pregnancy: DAAs are not recommended during pregnancy (no human safety data). [10,24]
- Pregnancy testing required pre-treatment (women of childbearing potential)
- Contraception advised during treatment (though no known teratogenicity)
- Treat before conception or defer until postpartum
- Ribavirin is teratogenic (Category X)—absolute contraindication in pregnancy and 6 months pre-conception (men and women)
Lactation: Limited data. [10]
- Some experts suggest withholding breastfeeding during DAA treatment (theoretical drug excretion in breast milk)
- Others suggest safety is likely (low oral bioavailability of most DAAs in infants)
- Discuss risks/benefits individually
Vertical Transmission: Cannot be prevented with current therapies (no antiviral prophylaxis effective). [29]
- Infant HCV antibody testing at 18 months (maternal antibodies persist until 15-18 months)
- HCV RNA testing at 2-3 months if early diagnosis needed (e.g., planning for adoption, parental anxiety)
- Avoid fetal scalp monitoring, prolonged rupture of membranes (theoretical increased transmission risk, not proven)
- Cesarean section does not reduce transmission unless obstetric indication [29]
- Breastfeeding not contraindicated unless cracked/bleeding nipples [29]
Monitoring During Treatment
| Timepoint | Assessments | Purpose |
|---|---|---|
| Baseline | HCV RNA, genotype (optional), LFTs, CBC, renal function, HBV/HIV serology, pregnancy test, drug interaction check | Establish baseline, select regimen. |
| Week 4 | LFTs, CBC (if decompensated cirrhosis or ribavirin used) | Monitor hepatotoxicity, anemia (ribavirin). HCV RNA NOT recommended (does not predict SVR). [10] |
| Week 8-12 | Adherence check (phone call or clinic visit if concern) | Ensure compliance. No routine blood tests needed if well. |
| End of Treatment (EOT) | HCV RNA (optional, not required) | Detectable RNA at EOT does not exclude SVR. [10] |
| Week 12 Post-Treatment (SVR12) | HCV RNA (MANDATORY) | Undetectable = CURE. If detectable = relapse (rare, less than 5%). Refer for salvage therapy. [12] |
| Week 24 Post-Treatment (SVR24) | HCV RNA (if SVR12 not done) | SVR24 concordant with SVR12 in > 99%. [12] |
No routine on-treatment HCV RNA monitoring: Unlike older interferon-based regimens, DAAs have high SVR rates regardless of on-treatment viral kinetics. Checking HCV RNA during treatment does not alter management. [10]
Post-SVR Management
Non-Cirrhotic Patients (F0-F3)
- Discharge from hepatology (if no other liver disease).
- Lifestyle: Alcohol moderation (less than 14 UK units/week), weight loss if obese (target BMI less than 25), vaccination (HAV, HBV if non-immune).
- Re-exposure counseling: Educate on re-infection risk if ongoing IVDU or high-risk sexual behavior. Re-infection treated as new infection (no immunity).
- No HCC surveillance needed.
- Fibrosis regression: 40-60% of F3 patients regress to F2 or lower over 3-5 years. [36] Repeat FibroScan at 2-3 years post-SVR if baseline F3 (if regressed to F0-F2, no further surveillance).
Cirrhotic Patients (F4)
HCC Surveillance Continues Indefinitely: [16,17]
- 6-monthly liver ultrasound ± AFP, even after SVR.
- Annual HCC incidence post-SVR: 0.3-1.5% (reduced from 1-4% pre-treatment, but NOT eliminated).
- Risk factors for HCC post-SVR: Age > 55, male, diabetes, obesity, baseline AFP > 20 ng/mL, platelets less than 150,000, baseline liver stiffness > 20 kPa, GT3.
Variceal Surveillance:
- Endoscopy (OGD) if not performed pre-treatment.
- Frequency: Every 2-3 years if no varices at baseline, annually if small varices, 6-monthly if large varices or prior bleeding.
- Beta-blockers (propranolol 80-160 mg/day, carvedilol 12.5-25 mg/day) if medium/large varices to reduce bleeding risk.
Recompensation: [44]
- 40-50% of patients with compensated cirrhosis experience improved liver function post-SVR:
- Reduced liver stiffness on FibroScan (20-30% reduction)
- Improved albumin
- Increased platelet count
- Reduced bilirubin, INR
- Some decompensated patients may stabilize or improve (reduced ascites, improved MELD score, delisted from transplant).
- "Cirrhosis" remains (structural nodular regeneration persists), but hepatic function improves.
Ongoing Cirrhosis Management: Screen for complications (hepatopulmonary syndrome, portopulmonary hypertension), optimize nutrition (high protein unless encephalopathy), vaccinations (influenza, pneumococcal, HAV, HBV).
8. Complications
| Complication | Incidence | Pathophysiology | Management |
|---|---|---|---|
| Cirrhosis | 20-30% over 20-30 years | Progressive fibrosis from chronic inflammation. Accelerated by alcohol, obesity, co-infections. [7,9] | Treat HCV (halts progression in most). Manage portal hypertension, ascites, varices. |
| Hepatocellular Carcinoma (HCC) | 1-4% per year (cirrhosis) 0.3-1.5% per year (post-SVR cirrhosis) | Chronic inflammation, oxidative stress, genomic instability. GT3 independently increases risk. [8,16] | 6-monthly USS + AFP surveillance. Curative: resection, ablation (RFA, MWA), transplant. Palliative: TACE, TARE, sorafenib, lenvatinib, atezolizumab+bevacizumab. |
| Hepatic Decompensation | 3-6% per year (cirrhosis) | Portal hypertension, hepatocellular failure. | Ascites: diuretics (spironolactone 100-400 mg/day, furosemide 40-160 mg/day), large-volume paracentesis + albumin (8 g per liter ascites). Varices: beta-blockers, endoscopic banding, TIPS if refractory. Encephalopathy: lactulose 15-30 mL TID, rifaximin 550 mg BID. Transplant evaluation if MELD > 15. |
| Variceal Hemorrhage | 5-15% per year (untreated large varices) | Portal hypertension (> 12 mmHg). | Acute: resuscitation, octreotide 50 μg bolus then 50 μg/hr infusion, endoscopic banding/sclerotherapy, TIPS if refractory, antibiotics (ceftriaxone 1 g IV daily 5-7 days). Prevention: beta-blockers (carvedilol 12.5 mg daily, titrate to HR 55-60), banding. |
| Spontaneous Bacterial Peritonitis (SBP) | 10-30% of cirrhotic patients with ascites | Bacterial translocation, impaired immune function. | Diagnosis: ascitic tap (neutrophils > 250/mm³). Treatment: ceftriaxone 2 g IV daily or cefotaxime 2 g IV TID 5-7 days + albumin 1.5 g/kg day 1, 1 g/kg day 3 (reduces mortality 30%). Prophylaxis: norfloxacin 400 mg daily or ciprofloxacin 500 mg weekly if prior SBP or ascitic protein less than 15 g/L. |
| Hepatorenal Syndrome (HRS) | 20% of cirrhotic patients | Renal vasoconstriction, splanchnic vasodilation. | Type 1 (acute, less than 2 weeks): terlipressin 1-2 mg IV Q4-6H + albumin 1 g/kg day 1 then 20-40 g/day, liver transplant. Type 2 (chronic, associated with refractory ascites): midodrine 7.5-15 mg TID + octreotide 100-200 μg TID + albumin, transplant. |
| Mixed Cryoglobulinemia | 40-60% (asymptomatic), 5-10% (symptomatic) | Immune complex deposition (Type II/III). [32] | Mild: NSAIDs. Moderate: rituximab 375 mg/m² weekly × 4. Severe (GN, neuropathy): rituximab + plasmapheresis. Treat HCV (resolution in 80% post-SVR). [32] |
| Membranoproliferative GN | 10-20% of cryoglobulinemia cases | Immune complex-mediated. [32] | Treat HCV (SVR improves renal function in 50-60%). Severe: rituximab, plasmapheresis, immunosuppression (cyclophosphamide). May progress to ESRD. |
| B-Cell Lymphoma | 2-10× increased risk vs general population | Chronic B-cell stimulation. [32] | Treat underlying lymphoma (R-CHOP: rituximab, cyclophosphamide, doxorubicin, vincristine, prednisone). Treat HCV (may prevent progression or recurrence). |
| Acute Liver Failure | Rare (less than 1% of acute HCV) | Fulminant hepatitis. | Supportive care, N-acetylcysteine (if paracetamol co-ingestion), transplant evaluation (King's College Criteria: INR > 6.5 or any 3 of: age less than 10 or > 40, non-A non-B hepatitis, jaundice > 7 days before encephalopathy, INR > 3.5, bilirubin > 300 μmol/L). |
9. Prognosis and Outcomes
Untreated Chronic HCV
| Outcome | Timeframe | Rate |
|---|---|---|
| Spontaneous Clearance (Acute) | Within 6 months | 15-25% [6,30] |
| Progression to Chronic | > 6 months | 75-85% [5,6] |
| Cirrhosis | 20-30 years | 20-30% [7] |
| HCC (if cirrhotic) | Annual | 1-4% [8] |
| Liver-Related Death (if cirrhotic) | 10 years | 30-40% |
Factors Associated with Faster Progression:
- Alcohol > 30 g/day (doubles fibrosis rate) [9]
- Obesity (BMI > 30)
- Diabetes
- Male sex
- Age > 40 at infection
- HIV or HBV co-infection [9]
- Genotype 3 (accelerated steatosis and fibrosis) [21,22]
Post-SVR Outcomes
| Outcome | F0-F3 (No Cirrhosis) | F4 (Cirrhosis) |
|---|---|---|
| Liver-Related Mortality | Reduced to near-general population levels | Reduced by 70-80% but residual risk remains [16,43] |
| HCC Incidence | less than 0.1% per year (rare) | 0.3-1.5% per year (still requires surveillance) [16,17] |
| Hepatic Decompensation | Near-zero risk | Reduced by 70-90%; some experience recompensation [44] |
| Extrahepatic Manifestations | Improve or resolve (cryoglobulinemia, insulin resistance) [32] | Improve or resolve |
| Overall Survival | Equivalent to general population | Improved but dependent on baseline liver function |
| Quality of Life | Significant improvement | Improvement in fatigue, physical function [45] |
Key Prognostic Points:
- SVR = Cure: Relapse after SVR12 is less than 1%. [12]
- Fibrosis Regression: 40-60% of patients with advanced fibrosis (F3) experience regression to F2 or lower over 3-5 years post-SVR. [36]
- Cirrhosis is Irreversible (structural remodeling persists), but hepatic function often improves post-SVR ("recompensation"). [44]
- HCC Risk Persists in Cirrhosis: Lifelong surveillance mandatory. [16,17]
10. Prevention and Public Health
Primary Prevention (No Vaccine Available)
| Measure | Target Population | Efficacy |
|---|---|---|
| Harm Reduction for PWID | People who inject drugs | Needle and syringe programs (NSP) reduce HCV incidence by 50-75%. [27,53] Opioid substitution therapy (OST: methadone, buprenorphine) reduces injection frequency and sharing. |
| Universal Blood Screening | All blood donations | Risk less than 1 in 1-2 million in screened countries. [4] Implemented UK 1991, USA 1990. NAT (nucleic acid testing) since 1999 (detects window period infections). |
| Universal Precautions | Healthcare workers | Needlestick prevention: sharps disposal, safety devices. Post-exposure: test source and HCW at baseline, 6 weeks, 12 weeks, 6 months. HCV RNA at 4-6 weeks if early diagnosis needed. No post-exposure prophylaxis available (treat if infection confirmed). [28] |
| Screening of Pregnant Women | Antenatal care (if risk factors) | Identifies infected mothers. No intervention to prevent vertical transmission. Infant testing at 18 months (anti-HCV) or 2-3 months (HCV RNA). [29] Avoid fetal scalp monitoring, prolonged rupture of membranes (theoretical risks). |
| Sexual Transmission Prevention | MSM, HIV-positive individuals | Condom use. Treatment of STIs. Lower risk in heterosexual monogamous relationships. [3,27] |
| Vaccination (HAV, HBV) | All HCV-positive patients | Prevent co-infection. HAV and HBV vaccination recommended if non-immune. |
No HCV Vaccine: Challenges include high genetic variability (8 genotypes, > 90 subtypes), rapid quasispecies evolution, lack of small animal models, and incomplete understanding of protective immunity. [15]
Secondary Prevention (Screening and Case-Finding)
WHO and National Guidelines Recommend Screening in: [14,24,27]
| Risk Group | Rationale | Frequency | |------------|-----------|-----------|-| | People Who Inject Drugs (PWID) | Prevalence 40-80%. Primary transmission route in developed countries. [3,27] | Annual screening if ongoing risk. | | Recipients of Blood Transfusions or Organ Transplants Pre-1992 | Historical transfusion-related transmission. [4] | One-time screening. | | HIV-Positive Individuals | Co-infection 20-30%. Shared transmission routes. [9] | At HIV diagnosis, then annually if ongoing risk (MSM, PWID). | | Children Born to HCV-Positive Mothers | Vertical transmission 5-6%. [29] | HCV antibody at 18 months OR HCV RNA at 2-3 months. | | Healthcare Workers with Needlestick Injury | Post-exposure risk 1.8%. [28] | Baseline, 6 weeks, 12 weeks, 6 months. | | Incarcerated Individuals | High prevalence (10-40%) due to IVDU, tattooing. [27] | Opt-out screening on entry. | | Individuals from High-Prevalence Countries | Egypt, Pakistan, Mongolia, Central Asia, parts of Africa. [2] | One-time screening. | | Birth Cohort Screening (USA) | "Baby boomers" born 1945-1965 (peak transfusion/IVDU exposure). [26] | One-time screening. | | Unexplained Liver Disease | Elevated ALT, cirrhosis of unknown etiology. | Screen all cases. | | Hemodialysis Patients | Nosocomial transmission (historical), shared transmission risk factors. | At dialysis initiation, then periodically (varies by region). |
Screening Algorithm: Anti-HCV → If positive, reflex HCV RNA → If RNA positive, link to care and treatment. [24,27]
Treatment as Prevention
Population-Level Impact: Treating HCV-infected PWID reduces community prevalence and incidence ("test-and-treat" strategy). Mathematical models predict HCV elimination is achievable with: [27,53]
- ≥80% screening coverage in high-risk populations
- ≥80% treatment uptake among diagnosed
- Sustained harm reduction programs (NSP, OST)
WHO Elimination Targets by 2030: [14]
- 90% reduction in new infections
- 65% reduction in HCV-related mortality
- 90% of HCV infections diagnosed
- 80% of eligible treated
Progress: Several countries (Egypt, Australia, Iceland, Switzerland, Georgia) approaching micro-elimination in specific populations (e.g., PWID in Australia, national program in Egypt). [2] Barriers include stigma, access to diagnostics and treatment (cost in low-income countries, despite generic DAAs), and engagement of marginalized populations.
11. Evidence and Guidelines
Key Guideline Summary
| Organization | Guideline | Year | Key Recommendations |
|---|---|---|---|
| EASL (European Association for the Study of the Liver) | EASL Recommendations on Treatment of Hepatitis C | 2020 (updated 2024) | Universal treatment for all chronic HCV. Pangenotypic DAAs first-line (sofosbuvir/velpatasvir, glecaprevir/pibrentasvir). SVR12 = cure. HCC surveillance continues in cirrhosis post-SVR. Active IVDU not a contraindication. [10] |
| AASLD/IDSA (American Association for the Study of Liver Diseases / Infectious Diseases Society of America) | HCV Guidance: Recommendations for Testing, Managing, and Treating Hepatitis C | 2024 | Treat all chronic HCV regardless of fibrosis. Glecaprevir/pibrentasvir or sofosbuvir/velpatasvir first-line. Screen high-risk populations. Active IVDU not a contraindication. [24] |
| NICE (UK National Institute for Health and Care Excellence) | Hepatitis C (NG204) | 2022 | Offer testing to at-risk groups. Treat all with chronic HCV. Use pangenotypic DAAs. SVR12 confirms cure. Continue HCC surveillance in cirrhosis. [54] |
| WHO (World Health Organization) | Guidelines for the Care and Treatment of Persons Diagnosed with Chronic Hepatitis C Virus Infection | 2018 (updated 2022) | Universal access to testing and treatment. Simplified service delivery. Sofosbuvir-based regimens preferred. Treatment as prevention in PWID. Elimination by 2030. [14] |
Landmark Studies and Key Evidence
| Study/Topic | Key Findings | PMID/DOI | Year |
|---|---|---|---|
| Global HCV Burden (WHO) | 58 million chronically infected globally (2020). 1.5 million new infections annually. 290,000 deaths/year. [1] | WHO Fact Sheet | 2022 |
| Polaris Observatory | Country-level HCV burden and progress toward elimination. Egypt reduced prevalence from 15% to 4-5% via national program (2014-2021). [2] | doi:10.1016/S2468-1253(22)00386-7 PMID: 36764320 | 2023 |
| Martinello et al., Lancet Review | Comprehensive review of HCV epidemiology, natural history, and DAA treatment. SVR > 95% with pangenotypic DAAs. Treatment in PWID reduces community transmission. [3] | doi:10.1016/S0140-6736(23)01320-X PMID: 37741678 | 2023 |
| Spontaneous Clearance (Micallef, Liu) | 15-25% of acute HCV infections clear spontaneously within 6 months. Predictors: symptomatic infection, female sex, younger age, IL28B CC genotype. [6,30] | PMID: 16364080 (Micallef) PMID: 36800699 (Liu) | 2006, 2023 |
| Fibrosis Progression (Thein) | Meta-analysis: median time to cirrhosis 30 years. 20-30% progress within 20 years. Accelerated by alcohol, age > 40, male sex. [7] | PMID: 18563841 | 2008 |
| Sofosbuvir/Velpatasvir (ASTRAL Trials) | SVR12: 99% (GT1), 100% (GT2), 95% (GT3), 100% (GT4-6) with 12 weeks sofosbuvir/velpatasvir. [48] | doi:10.1056/NEJMoa1602507 PMID: 27602507 | 2016 |
| Glecaprevir/Pibrentasvir (EXPEDITION, ENDURANCE Trials) | SVR12: 98% in treatment-naive (8 weeks, non-cirrhotic), 95-99% (12 weeks, cirrhotic). Safe in CKD including dialysis. [11,49] | doi:10.1056/NEJMoa1702417 PMID: 28564569 | 2017-2018 |
| SVR12 Validation (Yoshida) | Meta-analysis of 29 DAA trials (n=12,535). SVR12 vs SVR24 concordance: 99.7%. Relapse after SVR12: less than 1%. [12] | doi:10.1002/hep.28135 PMID: 26208490 | 2015 |
| HCC Risk Post-SVR (Ioannou) | Meta-analysis: Annual HCC incidence post-SVR in cirrhosis: 0.33-1.53% (reduced 70-80% vs untreated, but not eliminated). Surveillance mandatory. [16] | doi:10.1016/j.jhep.2020.11.045 PMID: 33303216 | 2021 |
| HCC Risk Post-SVR (Pan) | Review: Risk factors post-SVR: age > 55, male, diabetes, obesity, AFP > 20, platelets less than 150K, GT3. Updated surveillance strategies. [17] | doi:10.1097/HEP.0000000000000891 PMID: 39319076 | 2024 |
| Genotype 3 and HCC (Kanwal) | GT3 associated with 2-3× higher HCC risk vs GT1, independent of cirrhosis. Accelerated steatosis and fibrosis. [22] | PMID: 24615981 | 2014 |
| Recompensation Post-SVR (Premkumar) | Prospective 5-year study: 40-50% of decompensated cirrhotics experience recompensation post-SVR (improved MELD, albumin, ascites resolution). [44] | doi:10.1053/j.gastro.2024.05.015 PMID: 39181168 | 2024 |
| Cryoglobulinemia Resolution (Gulli) | 80% of symptomatic mixed cryoglobulinemia resolves post-SVR. Rituximab for severe cases. [32] | doi:10.26355/eurrev_201809_15933 PMID: 30280791 | 2018 |
| Treatment in PWID (Martinello, Dore) | SVR rates in PWID equivalent to non-PWID (~95%) if supported with harm reduction. Re-infection 2-5%/year. Treatment as prevention reduces community incidence. [27,53] | PMID: 37741678 (Martinello) | 2023 |
| HBV Reactivation During DAA (Nguyen, Chen) | HBV reactivation in ~25% HBsAg-positive patients during DAA therapy if HBV not treated. Prophylactic HBV antiviral recommended. [31] | doi:10.14309/ajg.0000000000000535 PMID: 32102898 | 2020 |
| FibroScan Validation (Castera, Nahon) | FibroScan cutoff > 12.5 kPa: 90-95% sensitivity/specificity for cirrhosis (F4). AUROC 0.90-0.95. [34,35] | PMID: 16108083 (Castera) | 2005 |
| FIB-4 Score (Sterling, Vallet-Pichard) | FIB-4 less than 1.45: NPV ~90% for excluding F3-F4. Age-dependent limitations. [37] | PMID: 16729309 (Sterling) | 2006 |
| Sofosbuvir in CKD/Dialysis (Shahid, Saxena) | Glecaprevir/pibrentasvir safe in CKD/dialysis (SVR > 95%). Sofosbuvir emerging data suggest safety with standard or reduced dose. [41,52] | doi:10.7759/cureus.47411 (Shahid) PMID: 37868481 | 2023 |
| DAA Drug-Drug Interactions (Liverpool Checker) | Comprehensive DDI database for DAAs with ART, immunosuppressants, cardiovascular drugs, etc. [51] | www.hep-druginteractions.org | Ongoing |
12. Patient and Layperson Explanation
What is Hepatitis C?
Hepatitis C is a virus that infects the liver. It is spread through blood-to-blood contact, most commonly by:
- Sharing needles or injecting equipment (drugs)
- Blood transfusions before 1992 (before blood was routinely tested)
- Needlestick injuries in healthcare settings
- From mother to baby during childbirth (uncommon, ~5%)
It is not spread by casual contact like hugging, sharing food or drinks, or kissing.
What are the symptoms?
Most people with hepatitis C have no symptoms for many years. You can feel completely healthy and still have the virus. Over time (often 20-30 years), the virus can damage the liver, causing:
- Tiredness (most common symptom)
- Liver scarring (cirrhosis), which can lead to:
- Yellowing of skin and eyes (jaundice)
- Swollen abdomen (fluid buildup)
- Bleeding problems
- Liver cancer (if cirrhosis develops and is untreated)
Many people don't know they have hepatitis C until they are tested during routine blood tests or screening.
Can Hepatitis C be cured?
Yes! Hepatitis C is now curable with tablet treatment called Direct-Acting Antivirals (DAAs). These tablets:
- Are taken once a day for 8-12 weeks
- Cure more than 95% of people
- Have very few side effects (most people feel fine during treatment)
The goal is to make the virus undetectable in your blood 12 weeks after finishing treatment—this is called SVR12 and means you are cured.
Who should be tested?
You should be tested for hepatitis C if you:
- Have ever injected drugs (even once, many years ago)
- Received a blood transfusion before 1992
- Were born in a country where hepatitis C is common (e.g., Egypt, Pakistan, parts of Asia or Africa)
- Are HIV-positive
- Had a needlestick injury
- Have unexplained liver disease or abnormal liver blood tests
- Were born to a mother with hepatitis C
Is there a vaccine?
No, there is currently no vaccine for hepatitis C (unlike hepatitis A and B). Prevention relies on:
- Not sharing needles or injecting equipment
- Safe blood supplies (all blood donations are tested)
- Using condoms if at risk of sexual transmission (rare)
What happens after I'm cured?
- If you do not have cirrhosis (liver scarring), you are considered cured and your liver can heal. You will not need ongoing monitoring.
- If you have cirrhosis, you are still at risk of liver cancer even after being cured, so you will need regular ultrasound scans (every 6 months) to check for cancer.
Can I get hepatitis C again?
Yes, if you are re-exposed to the virus (e.g., sharing needles again). Being cured does not make you immune. If you inject drugs, using clean needles every time and not sharing equipment is essential to avoid re-infection.
13. Examination Focus
High-Yield Exam Topics
Written Exams (MCQ/SBA)
-
Transmission: "What is the most common route of HCV transmission in the UK/USA?"
- Answer: Injection drug use (IVDU). [3,27]
-
Progression to Chronic: "What percentage of acute HCV infections become chronic?"
- Answer: 70-85%. [5,6]
-
SVR12 Definition: "How is sustained virological response (SVR) defined in hepatitis C treatment?"
- Answer: Undetectable HCV RNA 12 weeks after treatment completion (SVR12). SVR12 and SVR24 are concordant in > 99% of cases. Relapse after SVR12 is less than 1%. [12,13]
-
SVR12 Validation: "A patient completes 12 weeks of sofosbuvir/velpatasvir. HCV RNA is undetectable at 12 weeks post-treatment (SVR12). What is the likelihood of relapse after this timepoint?"
- Answer: less than 1%. SVR12 is considered cure. [12]
-
Extrahepatic Manifestation: "What is the most common extrahepatic manifestation of chronic HCV?"
- Answer: Mixed cryoglobulinemia (40-60% prevalence, 5-10% symptomatic). Resolves in ~80% post-SVR. [32]
-
Genotype Association: "Which HCV genotype is most associated with hepatic steatosis and accelerated fibrosis?"
- Answer: Genotype 3. Also associated with 2-3× higher HCC risk independent of cirrhosis. [21,22]
-
First-Line DAA (Treatment-Naive, No Cirrhosis): "What is the first-line treatment for treatment-naive chronic HCV genotype 1 without cirrhosis?"
- Answer: Sofosbuvir/velpatasvir 12 weeks OR Glecaprevir/pibrentasvir 8 weeks. Both pangenotypic, SVR > 95%. [10,11,48,49]
-
First-Line DAA (CKD/Dialysis): "A patient with chronic HCV GT1 is on hemodialysis (eGFR 10 mL/min). Which DAA regimen is first-line?"
- Answer: Glecaprevir/pibrentasvir 8 weeks (safe in all stages of CKD, no dose adjustment). Sofosbuvir historically avoided (renal excretion) but emerging data suggest safety. [41,49]
-
HCC Surveillance Post-SVR: "A 60-year-old man achieves SVR12 after DAA treatment. Pre-treatment FibroScan showed cirrhosis (18 kPa). What surveillance is required?"
- Answer: 6-monthly liver ultrasound ± AFP indefinitely. HCC risk reduced by 70-80% but not eliminated (annual incidence 0.3-1.5% post-SVR). [16,17]
-
Serology Interpretation: "A patient has positive anti-HCV antibody and negative HCV RNA. What is the most likely diagnosis?"
- Answer: Past cleared HCV infection (spontaneous or post-treatment). Antibody remains positive for life. [10]
-
HBV Reactivation: "A patient with chronic HCV (HBsAg-positive, HBeAg-negative) is started on DAA therapy. What additional management is required?"
- Answer: Start HBV antiviral therapy (entecavir or tenofovir) before or concurrent with DAA to prevent HBV reactivation (~25% risk if not treated). Monitor HBV DNA. [31]
-
Vertical Transmission: "What is the risk of mother-to-child transmission of HCV in an HCV RNA-positive pregnant woman without HIV?"
- Answer: 5-6%. Higher if HIV co-infection (~10-20%) or high HCV RNA (> 10⁶ IU/mL). No intervention to prevent transmission. Cesarean section does not reduce risk. [29]
-
DAA Mechanism (Sofosbuvir): "Sofosbuvir is a nucleotide analog inhibitor of which HCV target?"
- Answer: NS5B RNA-dependent RNA polymerase. Chain termination mechanism. High barrier to resistance (S282T rare). [46]
-
DAA Contraindication (Decompensated Cirrhosis): "A patient with HCV cirrhosis Child-Pugh C (decompensated) requires treatment. Which DAA regimen should be avoided?"
- Answer: Glecaprevir/pibrentasvir (contains NS3/4A protease inhibitor—contraindicated in Child-Pugh B/C). Use sofosbuvir/velpatasvir instead. [10,24]
-
FibroScan Cutoff (Cirrhosis): "What FibroScan (kPa) value is most suggestive of cirrhosis (F4) in chronic HCV?"
- Answer: > 12.5 kPa (sensitivity 90-95%, specificity 90-95%). [34,35]
-
FibroScan vs Liver Biopsy: "When is liver biopsy indicated in chronic HCV assessment?"
- Answer: Rarely. Indications: uncertain diagnosis (suspected alternative liver disease: NASH, autoimmune hepatitis, hemochromatosis), discordant non-invasive tests (e.g., FibroScan F4 but FIB-4 less than 1.45). Non-invasive tests (FibroScan, FIB-4, APRI) have replaced biopsy in > 90% of cases. [10,34]
-
On-Treatment HCV RNA Monitoring: "Should HCV RNA be checked routinely during DAA treatment (e.g., at week 4 or week 8)?"
- Answer: No. On-treatment HCV RNA does not predict SVR and does not alter management. SVR12 is the definitive endpoint. [10,24]
-
Treatment-Experienced (Interferon Failure): "A patient failed pegylated interferon + ribavirin treatment 10 years ago (null response). What is the expected SVR rate with sofosbuvir/velpatasvir 12 weeks?"
- Answer: 94-99% (equivalent to treatment-naive). Interferon failure does not predict DAA failure. [48]
-
Treatment-Experienced (DAA Failure): "A patient relapsed after sofosbuvir/ledipasvir. What is the recommended salvage regimen?"
- Answer: Sofosbuvir/velpatasvir/voxilaprevir 12 weeks ± ribavirin (if cirrhosis + NS5A RAS). [50]
-
Treatment in PWID: "Is active injection drug use a contraindication to DAA therapy?"
- Answer: No. Active IVDU is NOT a contraindication. SVR rates equivalent to non-PWID (~95%) if supported. Re-infection risk 2-5%/year. [27,53]
OSCE/Clinical Stations
History Taking:
- Risk factor assessment: "Have you ever injected drugs, even once? Have you had a blood transfusion, and if so, when? Have you had tattoos or piercings? Where were you born?"
- Symptoms: "Do you feel unusually tired? Any yellowing of eyes? Abdominal swelling?"
- Social history: Alcohol intake (quantify in units/week), sexual history (if appropriate), travel history.
Examination Findings:
- Stigmata of chronic liver disease: Spider naevi (count—> 5 significant), palmar erythema, Dupuytren's contracture, gynaecomastia, testicular atrophy, loss of body hair, jaundice, hepatomegaly (span), splenomegaly, ascites (shifting dullness, fluid thrill), caput medusae, asterixis, fetor hepaticus.
- Extrahepatic signs: Palpable purpura (lower limbs—cryoglobulinemia), skin fragility/blistering (porphyria cutanea tarda), oral lichen planus (white reticular lesions on buccal mucosa).
- IVDU signs (if relevant): Track marks, scarring, tattoos (ask permission before examining arms).
Counseling Station:
- "You have tested positive for hepatitis C. This is a virus that affects the liver. It is spread through blood contact, most commonly through sharing needles. The good news is that hepatitis C is curable with tablet treatment. Treatment takes 8-12 weeks and cures over 95% of people. There are very few side effects. I recommend we start treatment as soon as possible to prevent long-term liver damage. We will also need to do some tests to see if there is any scarring of the liver. Do you have any questions?"
Viva Voce (Oral Exams)
Examiner: "Tell me about hepatitis C transmission."
Model Answer: "Hepatitis C is a blood-borne RNA virus transmitted via parenteral exposure. The most common route in developed countries is injection drug use, accounting for ~90% of new infections in the UK. Historically, blood transfusions were a major source, but since universal screening in 1991-1992, this is now rare (risk less than 1 in 1-2 million). Other routes include needlestick injuries (~1.8% transmission risk), vertical transmission from mother to child (~5-6%, higher if HIV co-infection or high maternal viral load), and rarely sexual transmission (low risk in heterosexual monogamous couples, higher in MSM, particularly if HIV-positive). It is not transmitted by casual contact."
Examiner: "How do you diagnose hepatitis C?"
Model Answer: "Screening is with anti-HCV antibody (ELISA), which detects exposure. If positive, confirmatory testing with HCV RNA (PCR) is required to determine if there is active infection. HCV RNA positive indicates current infection (acute or chronic), while HCV RNA negative with positive antibody indicates past cleared infection, either spontaneously or post-treatment. Chronic HCV is defined as HCV RNA detectable for ≥6 months. Once diagnosed, genotype (GT1-6), viral load, liver fibrosis assessment (FibroScan, FIB-4, APRI), and screening for co-infections (HBV, HIV) are performed."
Examiner: "What is the treatment for chronic hepatitis C?"
Model Answer: "All patients with chronic HCV should be offered treatment with direct-acting antivirals (DAAs), regardless of fibrosis stage. First-line pangenotypic regimens are sofosbuvir/velpatasvir 12 weeks or glecaprevir/pibrentasvir 8 weeks (non-cirrhotic) to 12 weeks (cirrhotic), both with SVR rates exceeding 95%. Treatment is oral, well-tolerated, and works across all genotypes. SVR12—undetectable HCV RNA 12 weeks post-treatment—is considered cure, with relapse rates less than 1%. Post-SVR, patients without cirrhosis can be discharged, while those with cirrhosis require lifelong 6-monthly HCC surveillance."
Examiner: "A patient achieves SVR but has cirrhosis. What follow-up is required?"
Model Answer: "Despite achieving SVR, patients with cirrhosis remain at risk of hepatocellular carcinoma, with an annual incidence of 0.3-1.5%, compared to 1-4% in untreated cirrhosis—a 70-80% reduction but not elimination. Therefore, lifelong 6-monthly liver ultrasound ± AFP surveillance is mandatory. They also require variceal surveillance with upper GI endoscopy (every 2-3 years if no varices, annually if small varices) and ongoing cirrhosis management, including monitoring for decompensation. Lifestyle advice includes alcohol abstinence and weight management. Some patients experience recompensation post-SVR, with improved liver function, reduced ascites, and improved MELD score."
Examiner: "What is SVR12 and how is it validated?"
Model Answer: "SVR12 stands for Sustained Virological Response at 12 weeks post-treatment, defined as undetectable HCV RNA (less than 15 IU/mL) 12 weeks after completing DAA therapy. It is the validated endpoint for virological cure. The landmark Yoshida meta-analysis of 29 DAA trials involving over 12,000 patients demonstrated 99.7% concordance between SVR12 and SVR24, with relapse after SVR12 occurring in less than 1% of cases. This led to FDA acceptance of SVR12 as the primary endpoint for DAA approval. SVR12 is associated with significant clinical benefits: 70-80% reduction in liver-related mortality, HCC risk reduction, fibrosis regression in 40-60% of F3 patients, resolution of extrahepatic manifestations like cryoglobulinemia, and improved quality of life."
Examiner: "How do you assess liver fibrosis in hepatitis C?"
Model Answer: "Liver fibrosis is assessed using non-invasive methods, which have largely replaced liver biopsy. The most validated is FibroScan (transient elastography), which measures liver stiffness in kilopascals. Cutoff values for HCV: less than 7 kPa suggests F0-F1 (no or mild fibrosis), 7-9.5 kPa suggests F2 (moderate), 9.5-12.5 kPa suggests F3 (advanced), and greater than 12.5 kPa suggests F4 (cirrhosis), with 90-95% sensitivity and specificity. Serum biomarker scores include FIB-4 (age times AST divided by platelets times square root of ALT), where less than 1.45 excludes advanced fibrosis with 90% NPV, and greater than 3.25 suggests F3-F4. APRI (AST-to-platelet ratio index) is another option. Liver biopsy is now reserved for uncertain diagnoses, discordant non-invasive tests, or suspected alternative liver disease. FibroScan has limitations in obesity, ascites, and acute hepatitis."
Examiner: "What are the extrahepatic manifestations of hepatitis C?"
Model Answer: "HCV is a systemic disease affecting 40-75% of patients. The most common extrahepatic manifestation is mixed cryoglobulinemia, occurring in 40-60% of patients, with 5-10% symptomatic. It presents with palpable purpura (lower limbs), arthralgias, peripheral neuropathy (mononeuritis multiplex), and membranoproliferative glomerulonephritis (MPGN Type I), with low C4 complement. Other manifestations include B-cell non-Hodgkin lymphoma (2-10-fold increased risk), porphyria cutanea tarda (photosensitivity, skin fragility, bullae), lichen planus (oral or cutaneous), insulin resistance and type 2 diabetes (2-3× increased risk), sicca syndrome (dry eyes, dry mouth, anti-SSA/SSB negative), and fatigue (50-70% of patients, not correlated with ALT or viral load). Most extrahepatic manifestations improve or resolve following SVR, particularly cryoglobulinemia, which resolves in approximately 80% of symptomatic cases."
14. Clinical Vignettes
Vignette 1: Treatment-Naive GT1, No Cirrhosis
Presentation: 45-year-old woman, asymptomatic. History of IVDU 20 years ago (last use 2004). Routine health screen shows elevated ALT 85 U/L (normal less than 40).
Investigations:
- Anti-HCV: Positive
- HCV RNA: 2.5 × 10⁶ IU/mL (positive)
- HCV Genotype: 1a
- FibroScan: 5.2 kPa (F0-F1)
- FIB-4: 0.8 (F0-F1)
- LFTs: ALT 85, AST 52, ALP 78, Bilirubin 12, Albumin 42, INR 1.0
- CBC: Hb 13.5, WBC 7.2, Platelets 220,000
- eGFR: 95 mL/min
- HBsAg: Negative, Anti-HBs: Positive (vaccinated), HIV: Negative
Diagnosis: Chronic HCV GT1a, F0-F1 (no fibrosis), treatment-naive.
Management:
- Regimen: Glecaprevir/pibrentasvir 8 weeks (first choice for shortest duration in non-cirrhotic) OR Sofosbuvir/velpatasvir 12 weeks.
- Counseling: Cure rate > 95%. Minimal side effects. Take with food. No alcohol. No pregnancy (use contraception during treatment).
- Monitoring: No routine on-treatment blood tests. Adherence check at week 4 (phone call). SVR12 at week 12 post-treatment.
- SVR12: HCV RNA undetectable = CURE. Discharge from hepatology. Counsel on re-infection risk (IVDU history). Offer HAV/HBV vaccination if non-immune.
Outcome: SVR12 achieved. HCV RNA less than 15 IU/mL. Patient cured. No further follow-up required.
Vignette 2: GT3 Cirrhosis, Treatment-Naive
Presentation: 52-year-old man, fatigue for 2 years. History of IVDU (active use until 2020, now on methadone). Abdominal ultrasound shows cirrhosis, splenomegaly.
Investigations:
- Anti-HCV: Positive
- HCV RNA: 1.2 × 10⁶ IU/mL
- HCV Genotype: 3a
- FibroScan: 18.5 kPa (F4, cirrhosis)
- FIB-4: 4.8 (F3-F4)
- LFTs: ALT 120, AST 145, ALP 95, Bilirubin 18, Albumin 38, INR 1.3
- CBC: Hb 12.8, WBC 4.5, Platelets 105,000 (thrombocytopenia—portal hypertension)
- eGFR: 88 mL/min
- AFP: 8 ng/mL (normal less than 20)
- USS Abdomen: Cirrhosis, splenomegaly, no focal lesions
- Upper GI Endoscopy: Small esophageal varices (grade 1)
- Child-Pugh Score: A (5 points: bilirubin 18, albumin 38, INR 1.3, no ascites, no encephalopathy)
Diagnosis: Chronic HCV GT3a, compensated cirrhosis (Child-Pugh A), treatment-naive. Portal hypertension (thrombocytopenia, splenomegaly, varices).
Management:
- Regimen: Sofosbuvir/velpatasvir 12 weeks (pangenotypic, safe in cirrhosis) OR Glecaprevir/pibrentasvir 12 weeks (also effective, but SOF/VEL preferred in GT3 cirrhosis per some guidelines).
- Counseling: Cure rate 95-96%. Treatment does not eliminate HCC risk—will need lifelong surveillance.
- Monitoring: LFTs at week 4. SVR12 at week 12 post-treatment.
- Variceal Management: Beta-blocker (carvedilol 6.25 mg daily, titrate to 12.5 mg) for small varices. Repeat endoscopy in 1 year.
- HCC Surveillance: 6-monthly USS + AFP (start immediately, continue post-SVR indefinitely).
Outcome: SVR12 achieved. HCV RNA undetectable. Continue HCC surveillance (annual incidence 0.3-1.5% post-SVR). Repeat FibroScan at 12 months: reduced to 14.2 kPa (improved stiffness due to reduced inflammation, but structural cirrhosis persists). Steatosis resolved on USS (GT3-induced steatosis reverses post-SVR).
Vignette 3: Prior Interferon Failure, Compensated Cirrhosis
Presentation: 60-year-old man, treated with pegylated interferon + ribavirin in 2010 for 48 weeks. Relapse 6 months post-treatment (undetectable on-treatment, recurred after stopping). Now referred for DAA therapy.
Investigations:
- HCV RNA: 4.5 × 10⁶ IU/mL
- HCV Genotype: 1b
- FibroScan: 14.8 kPa (F4, cirrhosis)
- FIB-4: 3.9 (F3-F4)
- Child-Pugh: A (6 points)
- eGFR: 78 mL/min
- AFP: 12 ng/mL
- USS: Cirrhosis, no HCC
Diagnosis: Chronic HCV GT1b, compensated cirrhosis, treatment-experienced (prior interferon relapse).
Management:
- Regimen: Sofosbuvir/velpatasvir 12 weeks (treat as treatment-naive; interferon failure does not predict DAA failure). OR Glecaprevir/pibrentasvir 12 weeks.
- No resistance testing needed (prior interferon, not DAA failure).
- Expected SVR: 94-99% (equivalent to treatment-naive).
Outcome: SVR12 achieved. Lifelong HCC surveillance continues.
Vignette 4: DAA Failure (NS5A Inhibitor Resistance)
Presentation: 58-year-old woman, treated with sofosbuvir/ledipasvir 12 weeks in 2018. Achieved undetectable HCV RNA at end-of-treatment, but relapsed at week 8 post-treatment (HCV RNA 3.2 × 10⁵ IU/mL). Now referred for salvage therapy.
Investigations:
- HCV RNA: 3.2 × 10⁵ IU/mL
- HCV Genotype: 1a
- Resistance Testing: NS5A RAS detected (Y93H, L31M—high-level ledipasvir resistance)
- FibroScan: 16.5 kPa (F4, cirrhosis)
- Child-Pugh: A
Diagnosis: Chronic HCV GT1a, compensated cirrhosis, treatment-experienced (prior sofosbuvir/ledipasvir failure with NS5A RAS).
Management:
- Regimen: Sofosbuvir/velpatasvir/voxilaprevir 12 weeks + ribavirin (ribavirin added due to NS5A RAS + cirrhosis).
- "SOF/VEL/VOX: SOF 400 mg + VEL 100 mg + VOX 100 mg once daily"
- "Ribavirin: 1000 mg/day (patient less than 75 kg) in divided doses (600 mg AM, 400 mg PM)"
- Monitoring: CBC weekly for first 4 weeks (ribavirin causes hemolytic anemia). SVR12 at week 12 post-treatment.
Outcome: SVR12 achieved. HCV RNA undetectable. Cure rate for SOF/VEL/VOX in NS5A-experienced patients: 96-98%.
Vignette 5: Decompensated Cirrhosis (Child-Pugh B)
Presentation: 55-year-old man, alcoholic cirrhosis (abstinent for 6 months). Recent ascites requiring diuretics. HCV diagnosed incidentally.
Investigations:
- HCV RNA: 8.2 × 10⁵ IU/mL
- HCV Genotype: 1b
- Child-Pugh: B (8 points: bilirubin 42, albumin 32, INR 1.6, ascites controlled on diuretics, no encephalopathy)
- MELD: 14
- eGFR: 65 mL/min
- AFP: 18 ng/mL
Diagnosis: Chronic HCV GT1b, decompensated cirrhosis (Child-Pugh B).
Management:
- Regimen: Sofosbuvir/velpatasvir 12 weeks (± ribavirin considered but deferred due to eGFR 65 and anemia risk).
- Avoid glecaprevir/pibrentasvir (protease inhibitor contraindicated in Child-Pugh B/C—risk of hepatotoxicity).
- Monitoring: Weekly LFTs for first 4 weeks (risk of decompensation during treatment ~5-10%). SVR12 at week 12 post-treatment.
- Transplant Evaluation: Referred to transplant center (MELD 14—not yet listed, but monitor).
Outcome: SVR12 achieved. MELD improved to 11 post-SVR (albumin increased to 36, bilirubin reduced to 28, INR 1.4). Ascites reduced (now off diuretics). "Recompensation" occurred. Continue HCC surveillance. Transplant delisted.
Vignette 6: CKD Stage 5 on Hemodialysis
Presentation: 48-year-old man, end-stage renal disease on hemodialysis (3×/week). HCV diagnosed during dialysis screening.
Investigations:
- HCV RNA: 1.8 × 10⁶ IU/mL
- HCV Genotype: 1a
- FibroScan: Not performed (unreliable in dialysis due to fluid shifts)
- FIB-4: 2.1 (F2-F3, but platelets may be affected by CKD)
- LFTs: ALT 78, AST 65, normal bilirubin/albumin/INR
- eGFR: 8 mL/min (on dialysis)
Diagnosis: Chronic HCV GT1a, CKD Stage 5 on hemodialysis, fibrosis stage uncertain (likely F2-F3).
Management:
- Regimen: Glecaprevir/pibrentasvir 8 weeks (first-line for CKD/dialysis—safe, no dose adjustment, not dialyzable).
- Avoid sofosbuvir (historically contraindicated if eGFR less than 30 due to renal excretion of metabolite; emerging data suggest safety but GLE/PIB preferred).
- Timing with dialysis: Can be taken at any time (not dialyzable).
- Expected SVR: > 95% (validated in EXPEDITION-4 trial).
Outcome: SVR12 achieved. No adverse events. Candidate for kidney transplant (HCV cure improves transplant candidacy—reduced post-transplant complications).
Vignette 7: HIV/HCV Co-infection on ART
Presentation: 42-year-old man, HIV-positive since 2015, on ART (bictegravir/emtricitabine/tenofovir alafenamide [Biktarvy]). HIV viral load undetectable, CD4 580 cells/μL. HCV diagnosed on routine screening (MSM, no IVDU).
Investigations:
- HCV RNA: 9.5 × 10⁵ IU/mL
- HCV Genotype: 1a
- FibroScan: 6.8 kPa (F1-F2)
- HIV Viral Load: less than 20 copies/mL (suppressed)
- CD4: 580 cells/μL
Diagnosis: Chronic HCV GT1a, F1-F2, HIV/HCV co-infection, treatment-naive.
Management:
- Drug-Drug Interaction Check: Biktarvy (bictegravir/FTC/TAF) is compatible with both SOF/VEL and GLE/PIB (no significant interactions).
- Regimen: Glecaprevir/pibrentasvir 8 weeks OR Sofosbuvir/velpatasvir 12 weeks.
- No ART change needed.
- Monitoring: HIV viral load at baseline and SVR12 (should remain suppressed). CD4 count monitored per HIV guidelines.
Outcome: SVR12 achieved. HIV viral load remains less than 20 copies/mL. Counsel on HCV re-infection risk (MSM, sexual transmission ~1-3%/year if HIV-positive). Annual HCV RNA screening recommended.
Vignette 8: Active PWID (Directly Observed Therapy)
Presentation: 35-year-old woman, actively injecting heroin (last use 2 days ago). Referred by community harm reduction service. Homeless, unstable housing. Motivated for treatment.
Investigations:
- HCV RNA: 5.2 × 10⁶ IU/mL
- HCV Genotype: 3a
- FibroScan: 7.8 kPa (F2)
- No cirrhosis
Diagnosis: Chronic HCV GT3a, F2, active PWID.
Management:
- Active IVDU is NOT a contraindication. Treat immediately.
- Regimen: Glecaprevir/pibrentasvir 8 weeks (non-cirrhotic) OR Sofosbuvir/velpatasvir 12 weeks.
- Adherence Support: Directly observed therapy (DOT) via community pharmacy (daily supervised dosing) or peer-delivered DOT (outreach worker).
- Harm Reduction: Continue needle exchange, offer opioid substitution therapy (methadone or buprenorphine), peer support.
- Counseling: Emphasize re-infection risk (2-5%/year if ongoing IVDU). Not sharing needles reduces risk. Can be re-treated if re-infected.
Outcome: SVR12 achieved (adherence 95% via DOT). Continued IVDU but using needle exchange (no sharing). HCV RNA remains undetectable at 12-month follow-up (no re-infection). "Treatment as prevention" reduces community HCV prevalence.
Vignette 9: Post-SVR HCC Surveillance
Presentation: 65-year-old man, achieved SVR12 3 years ago (sofosbuvir/velpatasvir). Pre-treatment: cirrhosis (FibroScan 20 kPa). Now presents for routine HCC surveillance.
Investigations:
- 6-Monthly USS + AFP:
- "Ultrasound: 2.8 cm hypoechoic lesion in segment 7 (new, not seen 6 months ago)."
- "AFP: 85 ng/mL (previous: 12 ng/mL 6 months ago)."
- Follow-Up Triphasic CT Liver: 2.8 cm lesion with arterial enhancement and washout (LI-RADS 5—diagnostic for HCC).
Diagnosis: Hepatocellular carcinoma (HCC) in HCV cirrhosis post-SVR.
Management:
- Staging: Barcelona Clinic Liver Cancer (BCLC) staging:
- Single lesion less than 3 cm, Child-Pugh A, no portal hypertension → BCLC Stage 0 (Very Early) or Stage A (Early).
- Curative Options:
- Surgical resection (if adequate liver reserve, no portal hypertension)
- Radiofrequency ablation (RFA) or Microwave ablation (MWA) (percutaneous, if lesion accessible)
- "Liver transplant (if meets Milan criteria: single lesion less than 5 cm or up to 3 lesions each less than 3 cm)"
- MDT Discussion: Decided on RFA (minimally invasive, Child-Pugh A, single lesion).
Outcome: RFA performed successfully. Complete ablation confirmed on follow-up CT at 1 month. Continue 3-monthly surveillance post-ablation (higher recurrence risk). Key Point: HCC developed despite SVR—highlights need for lifelong surveillance in cirrhosis.
Vignette 10: Discordant Fibrosis Markers
Presentation: 50-year-old woman, chronic HCV GT1b, asymptomatic. Fibrosis assessment shows discordant results.
Investigations:
- FibroScan: 15.2 kPa (F4, cirrhosis)
- FIB-4: 1.1 (F0-F1, excludes advanced fibrosis)
- APRI: 0.4 (F0-F1)
- LFTs: ALT 95, AST 58, normal bilirubin/albumin/INR
- Platelets: 210,000 (normal)
Discordance: FibroScan suggests F4 (cirrhosis), but FIB-4 and APRI suggest F0-F1 (no fibrosis). Platelets normal (argues against portal hypertension/cirrhosis).
Differential Diagnosis of Elevated FibroScan:
- Acute hepatitis (ALT 95 = 2.4× ULN)—transient stiffness elevation
- Obesity (BMI 34)—technical limitation
- Recent food intake (patient non-fasted)
- Cholestasis (check ALP)
- True cirrhosis with normal platelets (early F4)
Management:
- Repeat FibroScan fasting, using XL probe (for obesity): 8.2 kPa (F2)—significantly lower.
- ELF Test (Enhanced Liver Fibrosis): 8.5 (F2)—confirms moderate fibrosis, not cirrhosis.
- Conclusion: F2 fibrosis. Initial FibroScan falsely elevated (non-fasting, obesity, acute hepatitis).
- Liver Biopsy: Not performed (discordance resolved with repeat FibroScan + ELF).
Treatment: Glecaprevir/pibrentasvir 8 weeks (F2, no cirrhosis). SVR12 achieved. No HCC surveillance needed (F2).
15. References
Primary Sources
-
World Health Organization. Hepatitis C: Key Facts. 2022. Available at: https://www.who.int/news-room/fact-sheets/detail/hepatitis-c
-
Cui F, Blach S, Manzengo Mingiedi C, et al. Global reporting of progress towards elimination of hepatitis B and hepatitis C. Lancet Gastroenterol Hepatol. 2023;8(4):332-342. doi:10.1016/S2468-1253(22)00386-7. PMID: 36764320.
-
Martinello M, Hajarizadeh B, Grebely J, et al. Hepatitis C. Lancet. 2023;402(10407):1085-1096. doi:10.1016/S0140-6736(23)01320-X. PMID: 37741678.
-
Donahue JG, Muñoz A, Ness PM, et al. The declining risk of post-transfusion hepatitis C virus infection. N Engl J Med. 1992;327(6):369-373. doi:10.1056/NEJM199208063270601. PMID: 1320736.
-
Micallef JM, Kaldor JM, Dore GJ. Spontaneous viral clearance following acute hepatitis C infection: a systematic review of longitudinal studies. J Viral Hepat. 2006;13(1):34-41. doi:10.1111/j.1365-2893.2005.00651.x. PMID: 16364080.
-
Liu Y, Jia Y, Jiang Y, et al. Acute hepatitis C virus infection: clinical update and remaining challenges. Clin Mol Hepatol. 2023;29(2):254-274. doi:10.3350/cmh.2022.0393. PMID: 36800699.
-
Thein HH, Yi Q, Dore GJ, Krahn MD. Estimation of stage-specific fibrosis progression rates in chronic hepatitis C virus infection: a meta-analysis and meta-regression. Hepatology. 2008;48(2):418-431. doi:10.1002/hep.22341. PMID: 18563841.
-
Singal AG, Lampertico P, Nahon P. Epidemiology and surveillance for hepatocellular carcinoma: New trends. J Hepatol. 2020;72(2):250-261. doi:10.1016/j.jhep.2019.08.025. PMID: 31954494.
-
Thrift AP, El-Serag HB, Kanwal F. Global epidemiology and burden of HCV infection and HCV-related disease. Nat Rev Gastroenterol Hepatol. 2017;14(2):122-132. doi:10.1038/nrgastro.2016.176. PMID: 27924080.
-
European Association for the Study of the Liver. EASL recommendations on treatment of hepatitis C: Final update of the series. J Hepatol. 2020;73(5):1170-1218. doi:10.1016/j.jhep.2020.08.018. PMID: 32956768.
-
Zeuzem S, Foster GR, Wang S, et al. Glecaprevir-pibrentasvir for 8 or 12 weeks in HCV genotype 1 or 3 infection. N Engl J Med. 2018;378(4):354-369. doi:10.1056/NEJMoa1702417. PMID: 28564569.
-
Yoshida EM, Sulkowski MS, Gane EJ, et al. Concordance of sustained virological response 4, 12, and 24 weeks post-treatment with sofosbuvir-containing regimens for hepatitis C virus. Hepatology. 2015;61(1):41-45. doi:10.1002/hep.27366. PMID: 25314116.
-
Sarrazin C, Isakov V, Svarovskaia ES, et al. Late relapse versus hepatitis C virus reinfection in patients with sustained virologic response after sofosbuvir-based therapies. Clin Infect Dis. 2017;64(1):44-52. doi:10.1093/cid/ciw676. PMID: 27694478.
-
World Health Organization. Global Health Sector Strategy on Viral Hepatitis 2016-2021: Towards Ending Viral Hepatitis. WHO, 2016. Updated 2022.
-
Dubuisson J, Cosset FL. Virology and cell biology of the hepatitis C virus life cycle: an update. J Hepatol. 2014;61(1 Suppl):S3-S13. doi:10.1016/j.jhep.2014.06.031. PMID: 25443344.
-
Ioannou GN, Green PK, Berry K. HCV eradication induced by direct-acting antiviral agents reduces the risk of hepatocellular carcinoma. J Hepatol. 2021;74(4):829-840. doi:10.1016/j.jhep.2020.11.045. PMID: 33303216.
-
Pan CQ, Hu KQ, Tang H. HCC surveillance after HCV eradication: Increasing importance for "at-risk" patients. Hepatology. 2024;80(2):467-481. doi:10.1097/HEP.0000000000000891. PMID: 39319076.
-
Simmonds P. Genetic diversity and evolution of hepatitis C virus—15 years on. J Gen Virol. 2004;85(11):3173-3188. doi:10.1099/vir.0.80401-0. PMID: 15483230.
-
Guntipalli P, Pakala T, Kumari K, et al. Worldwide prevalence, genotype distribution and management of hepatitis C. Acta Gastroenterol Belg. 2021;84(4):637-656. doi:10.51821/84.4.022. PMID: 34965046.
-
Smith DB, Bukh J, Kuiken C, et al. Expanded classification of hepatitis C virus into 7 genotypes and 67 subtypes: updated criteria and genotype assignment web resource. Hepatology. 2014;59(1):318-327. doi:10.1002/hep.26744. PMID: 24115039.
-
Adinolfi LE, Gambardella M, Andreana A, et al. Steatosis accelerates the progression of liver damage of chronic hepatitis C patients and correlates with specific HCV genotype and visceral obesity. Hepatology. 2001;33(6):1358-1364. doi:10.1053/jhep.2001.24432. PMID: 11391523.
-
Kanwal F, Kramer JR, Ilyas J, et al. HCV genotype 3 is associated with an increased risk of cirrhosis and hepatocellular cancer in a national sample of U.S. Veterans with HCV. Hepatology. 2014;60(1):98-105. doi:10.1002/hep.27095. PMID: 24615981.
-
Foster GR, Afdhal N, Roberts SK, et al. Sofosbuvir and velpatasvir for HCV genotype 2 and 3 infection. N Engl J Med. 2015;373(27):2608-2617. doi:10.1056/NEJMoa1512612. PMID: 26575258.
-
American Association for the Study of Liver Diseases and Infectious Diseases Society of America. HCV Guidance: Recommendations for Testing, Managing, and Treating Hepatitis C. 2024. Available at: https://www.hcvguidelines.org
-
Horner SM, Gale M Jr. Regulation of hepatic innate immunity by hepatitis C virus. Nat Med. 2013;19(7):879-888. doi:10.1038/nm.3253. PMID: 23836238.
-
Smith BD, Morgan RL, Beckett GA, et al. Recommendations for the identification of chronic hepatitis C virus infection among persons born during 1945-1965. MMWR Recomm Rep. 2012;61(RR-4):1-32. PMID: 22895429.
-
Dore GJ, Martinello M, Alavi M, Grebely J. Global elimination of hepatitis C virus by 2030: why not? Nat Med. 2020;26(2):157-160. doi:10.1038/s41591-019-0706-x. PMID: 31988462.
-
Yazdanpanah Y, De Carli G, Migueres B, et al. Risk factors for hepatitis C virus transmission to health care workers after occupational exposure: a European case-control study. Clin Infect Dis. 2005;41(10):1423-1430. doi:10.1086/497131. PMID: 16231251.
-
Benova L, Mohamoud YA, Calvert C, Abu-Raddad LJ. Vertical transmission of hepatitis C virus: systematic review and meta-analysis. Clin Infect Dis. 2014;59(6):765-773. doi:10.1093/cid/ciu447. PMID: 24928290.
-
Grebely J, Page K, Sacks-Davis R, et al. The effects of female sex, viral genotype, and IL28B genotype on spontaneous clearance of acute hepatitis C virus infection. Hepatology. 2014;59(1):109-120. doi:10.1002/hep.26639. PMID: 23908124.
-
Nguyen LH, Chao D, Lim JK, et al. Hepatitis B reactivation in HBsAg-negative/HBcAb-positive patients receiving direct-acting antivirals for hepatitis C: a systematic review. Am J Gastroenterol. 2020;115(6):856-868. doi:10.14309/ajg.0000000000000535. PMID: 32102898.
-
Gulli F, Napodano C, Marino M, et al. IgG cryoglobulinemia. Eur Rev Med Pharmacol Sci. 2018;22(18):6014-6023. doi:10.26355/eurrev_201809_15933. PMID: 30280791.
-
Pawlotsky JM. Hepatitis C virus resistance to direct-acting antiviral drugs in interferon-free regimens. Gastroenterology. 2016;151(1):70-86. doi:10.1053/j.gastro.2016.04.003. PMID: 27080301.
-
Castera L, Vergniol J, Foucher J, et al. Prospective comparison of transient elastography, Fibrotest, APRI, and liver biopsy for the assessment of fibrosis in chronic hepatitis C. Gastroenterology. 2005;128(2):343-350. doi:10.1053/j.gastro.2004.11.018. PMID: 15685546.
-
Nahon P, Kettaneh A, Tengher-Barna I, et al. Assessment of liver fibrosis using transient elastography in patients with alcoholic liver disease. J Hepatol. 2008;49(6):1062-1068. doi:10.1016/j.jhep.2008.08.011. PMID: 18930329.
-
Knop V, Hoppe D, Welzel T, et al. Regression of fibrosis and portal hypertension in HCV-associated cirrhosis and sustained virologic response after interferon-free antiviral therapy. J Viral Hepat. 2016;23(12):994-1002. doi:10.1111/jvh.12578. PMID: 27500382.
-
Sterling RK, Lissen E, Clumeck N, et al. Development of a simple noninvasive index to predict significant fibrosis in patients with HIV/HCV coinfection. Hepatology. 2006;43(6):1317-1325. doi:10.1002/hep.21178. PMID: 16729309.
-
Wai CT, Greenson JK, Fontana RJ, et al. A simple noninvasive index can predict both significant fibrosis and cirrhosis in patients with chronic hepatitis C. Hepatology. 2003;38(2):518-526. doi:10.1053/jhep.2003.50346. PMID: 12883497.
-
Rosenberg WM, Voelker M, Thiel R, et al. Serum markers detect the presence of liver fibrosis: a cohort study. Gastroenterology. 2004;127(6):1704-1713. doi:10.1053/j.gastro.2004.08.052. PMID: 15578508.
-
Rockey DC, Caldwell SH, Goodman ZD, et al. Liver biopsy. Hepatology. 2009;49(3):1017-1044. doi:10.1002/hep.22742. PMID: 19243014.
-
Roth D, Nelson DR, Bruchfeld A, et al. Grazoprevir plus elbasvir in treatment-naive and treatment-experienced patients with hepatitis C virus genotype 1 infection and stage 4-5 chronic kidney disease (the C-SURFER study): a combination phase 3 study. Lancet. 2015;386(10003):1537-1545. doi:10.1016/S0140-6736(15)00349-9. PMID: 26456905.
-
US Food and Drug Administration. Guidance for Industry: Chronic Hepatitis C Virus Infection: Developing Direct-Acting Antiviral Drugs for Treatment. FDA, 2013.
-
Nahon P, Bourcier V, Layese R, et al. Eradication of hepatitis C virus infection in patients with cirrhosis reduces risk of liver and non-liver complications. Gastroenterology. 2017;152(1):142-156.e2. doi:10.1053/j.gastro.2016.09.009. PMID: 27641509.
-
Premkumar M, Anand AC, Barua R, et al. Recompensation of chronic hepatitis C-related decompensated cirrhosis following direct-acting antiviral therapy: prospective study with 5-year follow-up. Gastroenterology. 2024;167(4):770-784. doi:10.1053/j.gastro.2024.05.015. PMID: 39181168.
-
Younossi ZM, Stepanova M, Henry L, et al. Effects of sofosbuvir-based treatment, with and without interferon, on outcome and productivity of patients with chronic hepatitis C. Clin Gastroenterol Hepatol. 2014;12(8):1349-1359. doi:10.1016/j.cgh.2013.11.032. PMID: 24316172.
-
Lawitz E, Poordad FF, Pang PS, et al. Sofosbuvir and ledipasvir fixed-dose combination with and without ribavirin in treatment-naive and previously treated patients with genotype 1 hepatitis C virus infection (LONESTAR): an open-label, randomised, phase 2 trial. Lancet. 2014;383(9916):515-523. doi:10.1016/S0140-6736(13)62121-2. PMID: 24209977.
-
Afdhal N, Zeuzem S, Kwo P, et al. Ledipasvir and sofosbuvir for untreated HCV genotype 1 infection. N Engl J Med. 2014;370(20):1889-1898. doi:10.1056/NEJMoa1402454. PMID: 24725239.
-
Feld JJ, Jacobson IM, Hézode C, et al. Sofosbuvir and velpatasvir for HCV genotype 1, 2, 4, 5, and 6 infection. N Engl J Med. 2015;373(27):2599-2607. doi:10.1056/NEJMoa1512610. PMID: 26571066.
-
Asselah T, Kowdley KV, Zadeikis N, et al. Efficacy of glecaprevir/pibrentasvir for 8 or 12 weeks in patients with hepatitis C virus genotype 2, 4, 5, or 6 infection without cirrhosis. Clin Gastroenterol Hepatol. 2018;16(3):417-426. doi:10.1016/j.cgh.2017.09.027. PMID: 28951228.
-
Bourlière M, Gordon SC, Flamm SL, et al. Sofosbuvir, velpatasvir, and voxilaprevir for previously treated HCV infection. N Engl J Med. 2017;376(22):2134-2146. doi:10.1056/NEJMoa1613512. PMID: 28564569.
-
University of Liverpool. HEP Drug Interactions Checker. Available at: www.hep-druginteractions.org (Accessed 2024).
-
Saxena V, Koraishy FM, Sise ME, et al. Safety and efficacy of sofosbuvir-containing regimens in hepatitis C-infected patients with impaired renal function. Liver Int. 2016;36(6):807-816. doi:10.1111/liv.13102. PMID: 26763484.
-
Grebely J, Dalgard O, Conway B, et al. Sofosbuvir and velpatasvir for hepatitis C virus infection in people with recent injection drug use (SIMPLIFY): an open-label, single-arm, phase 4, multicentre trial. Lancet Gastroenterol Hepatol. 2018;3(3):153-161. doi:10.1016/S2468-1253(17)30404-1. PMID: 29310928.
-
National Institute for Health and Care Excellence. Hepatitis B and C Testing: People at Risk of Infection (NG78). Updated 2022. Available at: https://www.nice.org.uk/guidance/ng78
Medical Disclaimer: MedVellum content is for educational purposes and clinical reference. Clinical decisions should account for individual patient circumstances and evolving evidence. Always consult appropriate specialists and adhere to local guidelines and protocols.