HIV Infection (Adult)
Human Immunodeficiency Virus (HIV) is a lentivirus (subfamily of retroviruses) that targets the human immune system, specifically CD4+ T-lymphocytes (helper T cells), macrophages, and dendritic cells. Progressive...
What matters first
Human Immunodeficiency Virus (HIV) is a lentivirus (subfamily of retroviruses) that targets the human immune system, specifically CD4+ T-lymphocytes (helper T cells), macrophages, and dendritic cells. Progressive...
CD4 count less than 200 cells/μL (high risk opportunistic infections)
6 Jan 2026
Generated educational material; verify before clinical use.
Visible references section
See the concept before reading it
Study the key anatomy, imaging, and decision pathways as full teaching plates.
Clinical board
A visual summary of the highest-yield teaching signals on this page.
Urgent signals
Safety-critical features pulled from the topic metadata.
- CD4 count less than 200 cells/μL (high risk opportunistic infections)
- CD4 count less than 50 cells/μL (critical immunosuppression)
- Severe headache with fever (cryptococcal meningitis)
- Progressive visual loss (CMV retinitis)
Linked comparisons
Differentials and adjacent topics worth opening next.
- Infectious Mononucleosis
- Acute Hepatitis
Content status and exam context
This page is AI-generated educational content. It may contain errors or omissions and is not a substitute for current guidelines, local protocols, senior clinical judgement, or professional medical advice.
MedVellum does not claim an individual clinician reviewer, board certification, or professional credential for this page unless a future version names a real, verifiable reviewer.
Clinical explanation and evidence
HIV Infection (Adult)
1. Clinical Overview
Summary
Human Immunodeficiency Virus (HIV) is a lentivirus (subfamily of retroviruses) that targets the human immune system, specifically CD4+ T-lymphocytes (helper T cells), macrophages, and dendritic cells. [1] Progressive destruction of these cells leads to profound immunodeficiency, leaving affected individuals vulnerable to opportunistic infections and malignancies that define Acquired Immunodeficiency Syndrome (AIDS). [2]
The introduction of combination antiretroviral therapy (ART) in the mid-1990s transformed HIV from an invariably fatal diagnosis into a manageable chronic condition. [3] People living with HIV (PLHIV) who achieve sustained virological suppression on ART can expect near-normal life expectancy and quality of life. [4] However, late diagnosis remains a critical challenge associated with high morbidity and mortality. [5]
Foundation Concept: U=U (Undetectable = Untransmittable)
This paradigm-shifting principle is now central to HIV medicine and prevention: individuals with HIV who maintain an undetectable viral load (less than 50 copies/mL) through adherent ART cannot sexually transmit the virus to others. [6]
The U=U concept is supported by:
- PARTNER Study (2016): Zero HIV transmissions among 58,000 condomless sexual acts in serodifferent couples where the HIV-positive partner had viral suppression. [6]
- PARTNER2 Study (2018): Extended follow-up in MSM couples confirmed zero transmissions over 77,000 acts. [7]
- Opposites Attract Study (2018): Confirmed findings in predominantly MSM cohort. [8]
This evidence has profound implications for clinical counselling, stigma reduction, and public health messaging.
Clinical Pearls
Acute Retroviral Syndrome (Seroconversion Illness): Occurs in 50-80% of new infections, typically 2-4 weeks post-exposure. [9] Presents as a mononucleosis-like illness with fever (80-90%), maculopapular rash (40-80%), pharyngitis (50-70%), and lymphadenopathy (40-70%). [9] The triad of fever + rash + pharyngitis in a sexually active adult should trigger HIV testing.
High Viral Load, Negative Antibody: During the "window period" (first 2-4 weeks), antibody tests may be negative despite extremely high viral loads (often > 100,000 copies/mL). [9] Fourth-generation antigen/antibody combination tests that detect p24 antigen significantly narrow this window.
Indicator Conditions: Clinical presentations that should prompt HIV testing include:
- Seborrhoeic dermatitis (severe/refractory)
- Oral or vaginal candidiasis (recurrent)
- Oral hairy leukoplakia (EBV-driven white corrugated plaques on lateral tongue)
- Herpes zoster in adults less than 50 years
- Unexplained lymphadenopathy, weight loss (> 10%), or prolonged fever
- Any sexually transmitted infection
- Hepatitis B or C infection
- Lymphoma
- Anal or cervical cancer
Immune Reconstitution Inflammatory Syndrome (IRIS): Paradoxical clinical deterioration occurring weeks to months after ART initiation, due to restoration of pathogen-specific inflammatory responses. [10] Most common with underlying TB, cryptococcal infection, or CMV. Risk factors include CD4 less than 50 cells/μL and rapid viral load decline. Management involves continuing ART while treating the underlying opportunistic infection; corticosteroids may be beneficial in severe cases.
2. Epidemiology
Global Burden
As of 2022: [11]
- 38.4 million people living with HIV worldwide
- 1.3 million new infections annually (down from 3.2 million at peak in 1997)
- 650,000 AIDS-related deaths annually (down from 2.0 million at peak in 2004)
- 29.8 million (76%) accessing antiretroviral therapy
Regional Disparities:
- Sub-Saharan Africa accounts for 67% of global PLHIV (25.6 million)
- Eastern and Southern Africa most severely affected (20.6 million, 54% of global total)
- Adult (15-49 years) HIV prevalence exceeds 10% in several Southern African countries
United Kingdom Epidemiology
UK data (2022): [12]
- 105,200 people living with diagnosed HIV
- Estimated 4,200 undiagnosed (4% of total PLHIV)
- 2,700 new HIV diagnoses in 2022
- Late diagnosis (CD4 less than 350 cells/μL) in 42% of new diagnoses
Risk Groups:
- Gay, bisexual, and other men who have sex with men (GBMSM): 45% of PLHIV, 49% of new diagnoses
- Heterosexual acquisition: 50% of new diagnoses (predominantly Black African communities)
- People who inject drugs (PWID): less than 2% of new diagnoses (success of harm reduction)
Geographical Distribution:
- London accounts for 38% of UK PLHIV
- High prevalence areas: London, Brighton, Manchester, Birmingham
Transmission Routes
Sexual Transmission (predominant globally): [13]
- Receptive anal intercourse: 1.4% per-exposure risk (highest)
- Insertive anal intercourse: 0.11% per exposure
- Receptive vaginal intercourse: 0.08% per exposure
- Insertive vaginal intercourse: 0.04% per exposure
Risk multiplied by:
- Acute/primary infection (viral load > 100,000: 10-26× increased risk)
- Concurrent STIs (genital ulcer disease: 2-5× increased risk)
- Advanced HIV in source (low CD4, high viral load)
Parenteral Transmission:
- Sharing injection equipment: 0.63% per injection episode
- Needlestick injury (healthcare): 0.23% per exposure (0.09% with hollow-bore needle)
- Blood transfusion: > 90% (now extremely rare in high-income countries with screening)
Vertical Transmission (Mother-to-Child): [14]
- Without intervention: 15-45% transmission risk
- With optimal ART + viral suppression + caesarean section + formula feeding: less than 0.1%
- Breastfeeding in untreated mothers: additional 10-20% transmission risk
3. Pathophysiology
Viral Structure and Classification
HIV is a lentivirus (slow virus) within the retrovirus family. Two types exist:
- HIV-1: Globally predominant, more virulent (four groups: M, N, O, P; group M causes pandemic)
- HIV-2: Primarily West Africa, slower progression, lower transmissibility
Viral Components:
- Envelope glycoproteins: gp120 (surface) and gp41 (transmembrane) - mediate cell entry
- Core proteins: p24 (capsid), p17 (matrix), p7 (nucleocapsid)
- Enzymes: Reverse transcriptase, integrase, protease
- Genome: Two copies of single-stranded RNA (9.8 kb)
Viral Entry and Replication Cycle
1. Attachment and Entry: [15]
- gp120 binds to CD4 receptor on target cells (T-helper cells, macrophages, dendritic cells)
- Conformational change exposes binding sites for co-receptors (CCR5 or CXCR4)
- CCR5 tropism predominates in early infection
- CXCR4 tropism emerges in ~50% during late disease (associated with faster progression)
- gp41-mediated membrane fusion allows viral core entry
Genetic Resistance:
- CCR5-Δ32 deletion (homozygous: 1% Northern Europeans): near-complete protection against R5-tropic HIV
- CCR5-Δ32 heterozygosity: slower progression if infected
2. Reverse Transcription:
- Viral reverse transcriptase (RNA-dependent DNA polymerase) synthesizes complementary DNA (cDNA) from viral RNA template
- High error rate (no proofreading): 1 error per 10,000 nucleotides → generates viral diversity and drug resistance
- RNase H component degrades original RNA template
- Second DNA strand synthesized to form double-stranded proviral DNA
3. Integration:
- Proviral DNA enters nucleus within pre-integration complex
- Viral integrase cleaves host chromosomal DNA and inserts provirus
- Integrated provirus = permanent infection (cannot be eliminated with current therapies)
- Establishes latent reservoir in resting CD4+ memory T cells (half-life: 44 months)
4. Transcription and Translation:
- Host RNA polymerase II transcribes proviral DNA
- Viral Tat protein enhances transcriptional efficiency (100-1000×)
- Rev protein facilitates nuclear export of unspliced viral RNA
5. Assembly and Budding:
- Viral proteins and genomic RNA assemble at cell membrane
- Immature virions bud from cell surface (using host ESCRT machinery)
- Viral protease cleaves Gag and Gag-Pol polyproteins → mature infectious virion
- Each infected CD4+ T cell produces ~10,000 viral particles before cell death
Viral Replication Dynamics:
- Generation time: ~1-2 days
- Plasma half-life of free virions: ~30 minutes
- Daily production in untreated infection: ~10^10 virions
- Daily CD4+ T-cell turnover: ~10^9 cells (normal ~2×10^9 total CD4 pool)
Mechanisms of CD4+ T-Cell Depletion
1. Direct Cytopathic Effects: [16]
- Viral replication disrupts cell metabolism and membrane integrity
- Accumulation of unintegrated viral DNA triggers cell death
- Syncytia formation (cell fusion via gp120-CD4 interaction) in X4-tropic virus
2. Indirect Mechanisms:
- Chronic immune activation: Persistent inflammation drives T-cell exhaustion and apoptosis
- Bystander apoptosis: Uninfected CD4+ cells undergo activation-induced cell death
- Reduced thymic output: HIV infection of thymic tissue impairs naïve T-cell production
- Autoimmune destruction: Anti-CD4 antibodies and cytotoxic T-cell responses against infected cells
3. Gut-Associated Lymphoid Tissue (GALT) Depletion: [17]
- Massive early depletion of gut CD4+ T cells (50-80% loss in first weeks)
- Disrupts intestinal barrier integrity → microbial translocation
- Chronic exposure to bacterial products (LPS) drives systemic immune activation
- Key driver of HIV-associated inflammation and non-AIDS comorbidities
Immunological Progression
Acute/Primary Infection (Weeks 0-12):
- Explosive viral replication: plasma HIV RNA peaks at 10^5-10^7 copies/mL
- CD4 count transiently drops (may fall to less than 200 cells/μL)
- HIV-specific immune responses emerge (weeks 2-8)
- Viral set point established by 6-12 months (predicts disease progression)
Chronic/Latent Phase (Years):
- Appears clinically quiescent but ongoing high-level viral replication
- CD4 count declines at average 50-80 cells/μL/year (varies widely)
- Persistent immune activation and inflammation
- Gradual loss of immune competence
AIDS (Advanced HIV):
- CD4 less than 200 cells/μL or AIDS-defining illness
- Severe immunodeficiency permits opportunistic infections and malignancies
- Without ART: median survival 1-2 years from AIDS diagnosis
Predictors of Rapid Progression: [18]
- High viral set point (> 100,000 copies/mL)
- Low CD4 nadir
- Advanced age at infection
- Co-infections (HCV, TB)
- CCR5-Δ32 heterozygosity protective (slower progression)
- HLA-B27 and HLA-B57 associated with slower progression (elite controllers)
4. Clinical Presentation
4.1 Primary HIV Infection (Acute Retroviral Syndrome)
Timeline: 2-4 weeks post-exposure (range: 1-6 weeks). [9]
Clinical Features (in order of frequency):
- Fever (80-90%): Often high-grade, may be accompanied by rigors
- Fatigue and malaise (70-90%)
- Pharyngitis (50-70%): Non-exudative, may resemble viral pharyngitis
- Rash (40-80%): Maculopapular, erythematous, affects trunk and face; may involve palms/soles (cf. secondary syphilis)
- Myalgia and arthralgia (50-70%)
- Lymphadenopathy (40-70%): Generalized, tender, mobile
- Headache (40-70%): May indicate aseptic meningitis
- Nausea, vomiting, diarrhoea (30-60%)
- Oral or genital ulceration (10-35%)
- Night sweats (50%)
- Weight loss (> 5kg in 70%)
Severe Manifestations (uncommon):
- Aseptic meningitis (5-10%): lymphocytic CSF, may have cranial neuropathies
- Guillain-Barré syndrome
- Brachial neuritis
- Myelopathy
- Oesophageal ulceration
Laboratory Findings:
- Very high HIV RNA (typically > 100,000, often > 1 million copies/mL)
- Negative or indeterminate HIV antibody (window period)
- Low CD4 count (transient immunosuppression)
- CD8 lymphocytosis (reactive cytotoxic response)
- Thrombocytopenia (30-40%)
- Elevated transaminases (mild)
- Atypical lymphocytes on blood film
Diagnostic Testing:
- 4th generation antigen/antibody test: Detects p24 antigen before antibody seroconversion (reduces window to 2-3 weeks)
- HIV RNA PCR: Definitive during window period; quantifies viral load
- Confirmation: Western blot or HIV-1/HIV-2 differentiation assay
Clinical Significance:
- Acute infection = highly infectious (high viral load, genital tract shedding)
- Often misdiagnosed as infectious mononucleosis, influenza, or viral pharyngitis
- Severity of acute illness correlates with viral set point and progression rate
4.2 Clinical Latency (Chronic Asymptomatic Phase)
Duration: Variable (median 8-10 years without treatment; range: 2-20+ years). [18]
Characteristics:
- Clinically asymptomatic or minimal symptoms
- Ongoing viral replication (viral load stable at "set point")
- Progressive CD4 decline (average 50-80 cells/μL/year)
- Persistent generalized lymphadenopathy (PGL) in 30-50%
- Detectable HIV antibodies (seroconversion complete by 3-12 weeks post-infection)
"Elite Controllers" (rare, less than 1%): [19]
- Maintain undetectable viral load (less than 50 copies/mL) without ART
- HLA-B27 and HLA-B57 overrepresented
- Vigorous HIV-specific CD8+ T-cell responses
- Still at risk of inflammation-related complications and low-level viremia
4.3 Symptomatic HIV Disease
Early Symptomatic (CD4 200-500 cells/μL):
Constitutional Symptoms:
- Persistent fever (> 1 month)
- Night sweats (drenching, requiring change of clothes)
- Unintentional weight loss (> 10% body weight)
- Chronic diarrhoea (> 1 month)
Mucocutaneous Manifestations:
- Oral candidiasis (thrush): Creamy white plaques on buccal mucosa, tongue, palate (removable)
- Oral hairy leukoplakia: Non-removable white corrugated plaques on lateral tongue (EBV-driven)
- Seborrhoeic dermatitis: Severe, refractory to topical therapy
- Pruritic papular eruption: Intensely itchy papules on trunk, extremities
- Herpes zoster (shingles): Especially multidermatomal or recurrent
- Recurrent HSV: Severe, persistent, atypical presentations
Haematological:
- Thrombocytopenia (HIV-associated ITP)
- Anaemia (normochromic normocytic; multifactorial)
- Leukopenia (neutropenia, lymphopenia)
Neurological:
- HIV-associated neurocognitive disorder (HAND): Impaired concentration, memory, executive function
- Peripheral neuropathy (distal symmetric polyneuropathy)
4.4 AIDS-Defining Illnesses (CD4 less than 200 cells/μL)
Respiratory:
- Pneumocystis jirovecii pneumonia (PCP): Most common OI; dry cough, exertional dyspnoea, fever, desaturation on exertion; CXR may be normal or show bilateral interstitial infiltrates
- Pulmonary tuberculosis: Often atypical (upper lobe cavitation less common); high incidence of extrapulmonary and disseminated TB
- Recurrent bacterial pneumonia (≥2 episodes in 12 months)
Neurological:
- Cerebral toxoplasmosis: Ring-enhancing lesions (typically multiple); headache, focal deficit, seizures, fever
- Cryptococcal meningoencephalitis: Subacute headache, fever, altered mental status; high CSF opening pressure (> 25 cmH₂O)
- Primary CNS lymphoma: Solitary or multiple mass lesions; EBV-associated
- Progressive multifocal leukoencephalopathy (PML): JC virus-driven demyelination; focal deficits, cognitive decline; MRI shows non-enhancing white matter lesions
- HIV-associated dementia: Severe cognitive impairment affecting daily function (now rare with ART)
- CMV encephalitis/radiculopathy
Gastrointestinal:
- Oesophageal candidiasis: Dysphagia, odynophagia, retrosternal chest pain
- Chronic cryptosporidiosis (> 1 month): Severe watery diarrhoea, malabsorption
- CMV colitis: Bloody diarrhoea, abdominal pain; colonoscopy shows ulceration
Ocular:
- CMV retinitis: Progressive visual loss; "pizza" retina with haemorrhages and exudates; leading cause of blindness in untreated HIV
Disseminated Infections:
- Disseminated Mycobacterium avium complex (MAC): Fever, night sweats, weight loss, hepatosplenomegaly, cytopenias (CD4 less than 50 cells/μL)
- Disseminated histoplasmosis, coccidioidomycosis (endemic areas)
Malignancies (AIDS-Defining): [20]
- Kaposi sarcoma (HHV-8/KSHV-associated): Violaceous cutaneous lesions, oral lesions, visceral involvement
- Non-Hodgkin lymphoma: High-grade B-cell (Burkitt, diffuse large B-cell, primary CNS)
- Invasive cervical carcinoma (HPV-associated)
Other:
- Wasting syndrome (> 10% weight loss + chronic diarrhoea or fever > 30 days)
- HIV-associated nephropathy (HIVAN): Proteinuria, rapid progression to ESRD
CD4 Count Stratification of Opportunistic Infections
| CD4 Count | Typical Opportunistic Infections |
|---|---|
| less than 200 cells/μL | PCP, disseminated histoplasmosis, coccidioidomycosis |
| less than 150 cells/μL | Disseminated TB, Candida oesophagitis |
| less than 100 cells/μL | Cerebral toxoplasmosis, cryptococcal meningitis, PML |
| less than 50 cells/μL | Disseminated MAC, CMV retinitis/colitis |
5. Investigations
5.1 Diagnostic Testing
Fourth-Generation Antigen/Antibody Combination Assay (Standard Screening): [21]
- Detects HIV-1/HIV-2 antibodies + p24 antigen
- Sensitivity > 99.5% after seroconversion
- Window period: ~18-45 days (antibody alone: 3-12 weeks)
- Recommended testing algorithm:
- Reactive screening test → confirmatory differentiation assay (HIV-1/HIV-2 antibody)
- If differentiation assay negative or indeterminate → HIV RNA PCR
Point-of-Care Rapid Tests (Antibody-Only):
- Fingerprick or oral fluid sample
- Result in 15-30 minutes
- Sensitivity 99%, specificity 99.8%
- Window period: ~3 months (antibody only)
- Reactive result requires laboratory confirmation
HIV-1 RNA Quantification (Viral Load):
- Confirms diagnosis (especially during window period or if serology indeterminate)
- Baseline assessment and monitoring on ART
- Detectable range: 20-10,000,000 copies/mL
- Target on ART: less than 50 copies/mL (undetectable)
HIV-1/HIV-2 Differentiation:
- Distinguishes HIV-1 from HIV-2 (implications for treatment: HIV-2 naturally resistant to NNRTIs)
When to Test for HIV: [22]
- Universal opt-out testing in high-prevalence areas (diagnosed prevalence > 2 per 1,000)
- Indicator conditions (see Clinical Pearls)
- All new presentations to sexual health/GUM clinics
- All pregnant women (antenatal screening)
- Before starting immunosuppressive therapy
- New diagnosis of hepatitis B, hepatitis C, tuberculosis
- All patients with lymphoma
5.2 Baseline Assessment After Diagnosis
Mandatory Initial Investigations: [23]
Staging and Prognostic:
- CD4+ T-cell count: Defines stage, guides prophylaxis, predicts prognosis
- HIV-1 RNA viral load: Baseline level, confirms active replication
- HIV-1 resistance genotyping: Detects transmitted drug resistance (15-20% in UK) to guide ART choice
Pharmacogenetic Screening:
- HLA-B*5701 testing: Screen BEFORE abacavir use (5-8% prevalence in Caucasians)
- Positive → contraindication to abacavir (risk of potentially fatal hypersensitivity reaction)
- Negative → abacavir can be used safely
Co-Infection Screening:
- Hepatitis B serology: HBsAg, anti-HBs, anti-HBc (10-15% HBV co-infection; influences ART choice)
- Hepatitis C serology: Anti-HCV antibody + HCV RNA if positive (5-10% co-infection; treat HCV before or during ART)
- Syphilis serology: Treponemal EIA + RPR/VDRL
- Tuberculosis screening: Symptom enquiry; CXR; IGRA (QuantiFERON/T-SPOT) in high-risk or CD4 less than 200
- Toxoplasma gondii IgG: Identifies need for primary prophylaxis if CD4 less than 100 and seronegative
General Health:
- Full blood count, renal function (eGFR), liver function tests
- Fasting lipid profile, fasting glucose (baseline cardiovascular risk)
- Urinalysis (HIVAN screening)
- Pregnancy test (women of childbearing potential)
Cervical/Anal Screening:
- Cervical cytology (annual; high HPV-related cervical cancer risk)
- Consider anal cytology/high-resolution anoscopy in MSM (anal cancer risk)
Other:
- CMV IgG (nearly universal in PLHIV; informs prophylaxis decisions)
- Vitamin D level (deficiency common)
- Bone density (DEXA scan) if age > 50 or other risk factors
Opportunistic Infection Screening (if CD4 less than 200):
- Serum cryptococcal antigen (CrAg) if CD4 less than 100 (detects subclinical infection; pre-emptive therapy reduces meningitis)
5.3 Monitoring on Antiretroviral Therapy
Viral Load Monitoring: [23]
- Baseline, then at 4, 12, and 24 weeks after ART initiation
- Target: less than 50 copies/mL by 24 weeks (> 90% achieve this)
- Once suppressed: every 3-6 months
- Virological failure: Confirmed viral load > 200 copies/mL (suggests resistance or non-adherence)
- Persistent low-level viremia (50-200 copies/mL) may warrant genotyping
CD4 Monitoring:
- Baseline, then every 3-6 months in first year
- Once stable and viral load suppressed: can be stopped if CD4 consistently > 350 cells/μL
- CD4 recovery: typically increases 50-150 cells/μL in first year
Renal Function:
- Every 3-6 months (tenofovir nephrotoxicity)
- eGFR, urinalysis, urine protein:creatinine ratio
Hepatic Function:
- Every 6-12 months
- More frequent if HBV/HCV co-infection or hepatotoxic ART
Lipid Profile and Cardiovascular Risk:
- Every 6-12 months (ART and HIV both increase CV risk)
- Framingham or QRISK3 cardiovascular risk assessment
Bone Density:
- DEXA scan at baseline if risk factors; repeat every 2-5 years
- HIV and some ARVs (tenofovir disoproxil fumarate) decrease bone density
Adherence Assessment:
- At every visit (most important predictor of virological success)
- Pharmacy refill data, self-report, viral load as objective measure
6. Management
6.1 General Principles
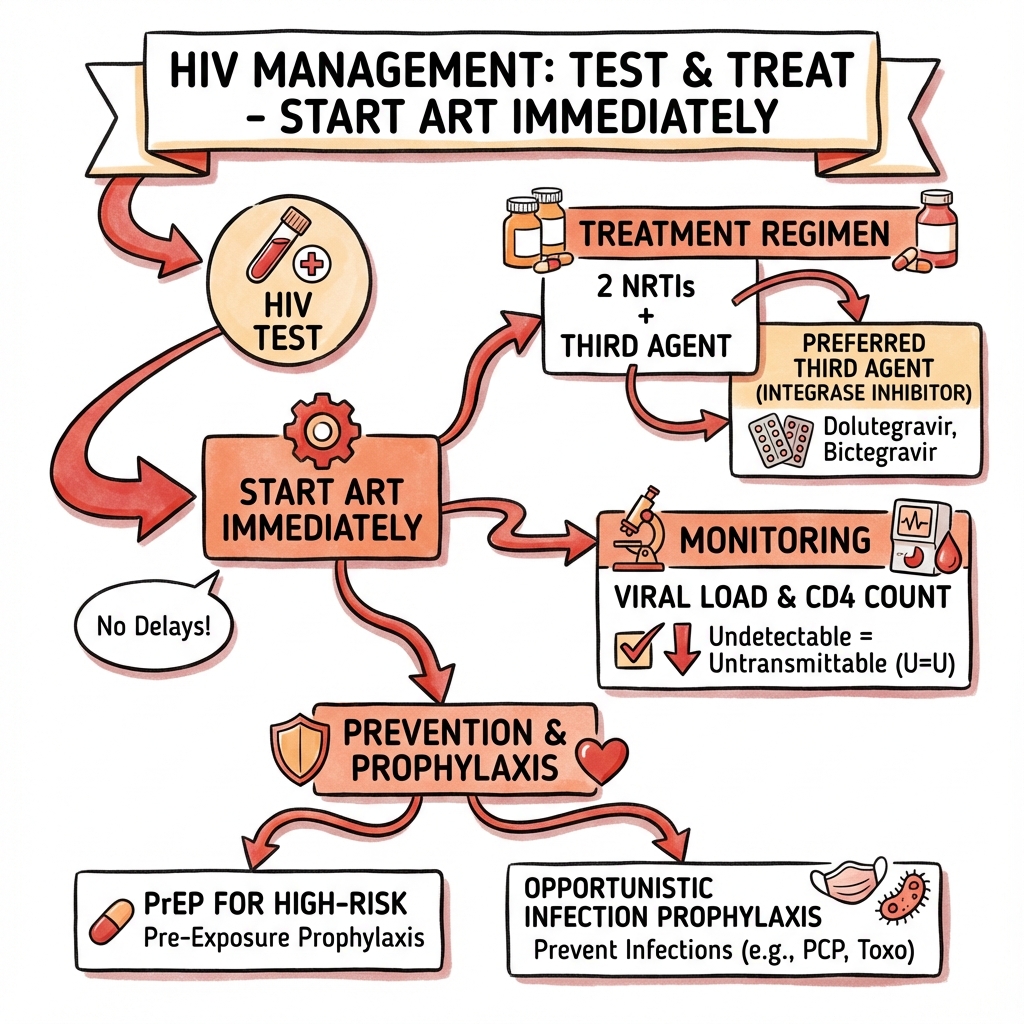
Universal "Test and Treat" Strategy: [24,25]
- Initiate ART in all people living with HIV, regardless of CD4 count
- Supported by START trial (2015): immediate ART (CD4 > 500) vs deferred (CD4 less than 350) showed 57% reduction in serious AIDS and non-AIDS events [24]
- Benefits: individual health, prevention of transmission (Treatment as Prevention)
Goals of ART:
- Suppress viral load to less than 50 copies/mL (undetectable)
- Restore immune function (CD4 recovery)
- Prevent opportunistic infections
- Reduce HIV-related and non-HIV-related morbidity and mortality
- Prevent transmission (U=U)
- Improve quality of life
When to Start ART:
- Immediately (within days of diagnosis if patient ready)
- Same day initiation in acute/primary HIV (high viral load = high transmission risk)
- Urgently if AIDS-defining illness, CD4 less than 200, pregnancy, HBV co-infection requiring treatment
- Delay ART only if: active TB meningitis (IRIS risk), cryptococcal meningitis (induction therapy first), patient not ready
6.2 Antiretroviral Drug Classes
Mechanism of Action by Drug Class: [26]
| Class | Mechanism | Examples | Key Points |
|---|---|---|---|
| Nucleoside/Nucleotide Reverse Transcriptase Inhibitors (NRTIs) | Competitive inhibition + chain termination of reverse transcription | Tenofovir (TAF/TDF), Emtricitabine (FTC), Abacavir (ABC), Lamivudine (3TC) | Backbone of ART; require intracellular phosphorylation; mitochondrial toxicity |
| Non-Nucleoside Reverse Transcriptase Inhibitors (NNRTIs) | Non-competitive allosteric inhibition of reverse transcriptase | Efavirenz (EFV), Rilpivirine (RPV), Doravirine (DOR) | Low genetic barrier (single mutation = resistance); CNS side effects (EFV) |
| Integrase Strand Transfer Inhibitors (INSTIs) | Block integration of proviral DNA into host genome | Dolutegravir (DTG), Bictegravir (BIC), Raltegravir (RAL) | Preferred anchor drug; high barrier to resistance; excellent tolerability |
| Protease Inhibitors (PIs) | Inhibit viral protease (prevents maturation of virions) | Darunavir (DRV), Atazanavir (ATV) | Require boosting with ritonavir/cobicistat; metabolic side effects |
| Entry/Fusion Inhibitors | Block viral entry (CCR5 antagonist, fusion inhibitor) | Maraviroc (MVC), Enfuvirtide (T20) | Salvage therapy; tropism testing required (MVC) |
| Post-Attachment Inhibitors | Blocks CD4 binding and conformational change | Ibalizumab | Salvage therapy; IV administration |
| Capsid Inhibitors | Disrupts capsid-mediated functions | Lenacapavir | Long-acting (6-monthly injection); newest class |
6.3 Recommended First-Line Regimens (UK/BHIVA 2022)
Standard Regimen (Preferred): [23]
Integrase Inhibitor-Based (INSTI) + 2 NRTIs:
Option 1: Bictegravir/Tenofovir Alafenamide/Emtricitabine (BIC/TAF/FTC)
- Single-tablet regimen (STR): One pill once daily
- Excellent efficacy (> 90% viral suppression at 48 weeks)
- High barrier to resistance
- Minimal drug interactions
- Well-tolerated
- Safe in renal impairment (TAF has less nephrotoxicity than TDF)
Option 2: Dolutegravir + Tenofovir Disoproxil Fumarate/Emtricitabine (DTG + TDF/FTC)
- DTG: 50mg once daily
- TDF/FTC: single-tablet (Truvada)
- Most cost-effective (generic DTG and TDF/FTC available)
- Excellent efficacy and high barrier to resistance
- Monitor renal function (TDF nephrotoxicity)
Option 3: Dolutegravir + Abacavir/Lamivudine (DTG + ABC/3TC)
- Avoid if HLA-B*5701 positive (abacavir hypersensitivity)
- Avoid if high cardiovascular risk (conflicting data on ABC and MI risk)
- Avoid if baseline viral load > 100,000 copies/mL (higher failure rate)
- Single-tablet regimen (Triumeq) available
Alternative Regimens:
Doravirine/Tenofovir Disoproxil Fumarate/Lamivudine (DOR/TDF/3TC):
- NNRTI-based regimen
- Well-tolerated (fewer CNS side effects than efavirenz)
- Fewer drug interactions than other NNRTIs
Darunavir/Cobicistat/Tenofovir Alafenamide/Emtricitabine (DRV/c/TAF/FTC):
- PI-based regimen
- High barrier to resistance
- Reserve for specific situations (e.g., high baseline resistance, pregnancy)
6.4 Special Situations
Pregnancy: [27]
- Continue or start ART immediately (regardless of trimester)
- Goal: Undetectable viral load at delivery (less than 50 copies/mL) → less than 0.1% vertical transmission
- Preferred regimen: Integrase inhibitor-based
- "Historical concern re: DTG and neural tube defects (Tsepamo study) largely resolved (absolute risk 0.19% vs 0.11%)"
- "Current recommendation: DTG can be used (counsel re: minimal increased NTD risk if conception on DTG)"
- Avoid efavirenz (teratogenic in primates; first-trimester neural tube defects)
- Delivery mode:
- Vaginal delivery if viral load less than 50 copies/mL
- Caesarean section if viral load > 50 copies/mL or unknown
- Infant prophylaxis: Zidovudine (AZT) for 4 weeks if mother suppressed; intensified regimen if high-risk
- Breastfeeding: Avoid in high-income countries (formula feeding recommended)
Tuberculosis Co-Infection: [28]
- TB treatment takes priority; start ART within 2 weeks (8 weeks if TB meningitis - delay reduces IRIS risk)
- Drug interactions: Rifampicin is potent enzyme inducer
- Avoid standard-dose PIs and most NNRTIs
- "Preferred: Dolutegravir 50mg twice daily (rifampicin reduces DTG levels)"
- "Alternative: Efavirenz 600mg daily (inducer-inducer interaction)"
- Monitor for IRIS (20-30% incidence)
Hepatitis B Co-Infection (10% of PLHIV): [23]
- Always include HBV-active NRTIs in regimen:
- Tenofovir (TDF or TAF) + Emtricitabine or Lamivudine
- Stopping these agents → risk of HBV flare and hepatic decompensation
- Monitor HBV DNA, ALT, HBeAg/anti-HBe
Hepatitis C Co-Infection:
- Treat HCV with direct-acting antivirals (DAAs)
- Check for drug interactions with ART (significant with PIs)
- ART improves HCV treatment outcomes
Chronic Kidney Disease:
- Avoid or dose-adjust: Tenofovir disoproxil fumarate (TDF) - prefer TAF
- Consider: Abacavir/lamivudine or TAF-containing regimens
- Dose adjustments for renally cleared drugs (e.g., emtricitabine, lamivudine)
Cardiovascular Disease:
- Avoid abacavir if established CVD or high Framingham risk (> 20%)
- Manage traditional CV risk factors aggressively (statins, antihypertensives)
- Some PIs worsen lipid profile
6.5 Opportunistic Infection Prophylaxis
Primary Prophylaxis (preventing first episode):
Pneumocystis jirovecii Pneumonia (PCP):
- Indication: CD4 less than 200 cells/μL
- Agent: Co-trimoxazole 960mg (DS) once daily (or 960mg thrice weekly)
- Alternative: Dapsone 100mg daily (check G6PD first), atovaquone 1500mg daily, nebulised pentamidine 300mg monthly
- Stop when: CD4 > 200 cells/μL for > 3 months on ART
Toxoplasma gondii:
- Indication: CD4 less than 100 cells/μL and toxoplasma IgG positive
- Agent: Co-trimoxazole 960mg daily (also covers PCP)
- Stop when: CD4 > 200 cells/μL for > 3 months
Mycobacterium avium Complex (MAC):
- Indication: CD4 less than 50 cells/μL
- Agent: Azithromycin 1250mg once weekly (or 600mg daily clarithromycin)
- Stop when: CD4 > 100 cells/μL for > 3 months on ART
Cryptococcus:
- Screening with serum CrAg if CD4 less than 100 cells/μL
- If CrAg positive but asymptomatic (no meningitis): pre-emptive fluconazole 800mg daily × 2 weeks, then 400mg daily
Tuberculosis (Latent TB Treatment):
- Indication: IGRA positive or high TB exposure risk, especially if CD4 less than 350
- Regimen: Isoniazid 300mg + pyridoxine 25mg daily × 6 months, or 3-month rifampicin + isoniazid
Secondary Prophylaxis (preventing recurrence after treatment):
- Continue until immune reconstitution (generally CD4 > 200 cells/μL for > 3 months)
6.6 Monitoring and Adherence
Adherence is Critical: [29]
-
95% adherence required for durable viral suppression (modern INSTIs more forgiving)
- Suboptimal adherence → virological failure → drug resistance → limited future options
Barriers to Adherence:
- Complexity of regimen (number of pills, dosing frequency)
- Side effects (real or perceived)
- Stigma and lack of disclosure
- Mental health issues (depression, substance use)
- Socioeconomic factors (housing instability, food insecurity)
Strategies to Support Adherence:
- Single-tablet regimens (improve adherence vs multi-pill)
- Once-daily dosing
- Tailored counseling and education
- Mobile phone reminders, pill boxes
- Directly observed therapy (DOT) in selected cases
- Peer support groups
- Address mental health and substance use
6.7 Treatment Failure and Resistance Testing
Virological Failure: Confirmed viral load > 200 copies/mL on two occasions. [23]
Approach:
- Assess adherence (most common cause)
- Review drug interactions (PPI reducing absorption, rifampicin inducing metabolism)
- Resistance testing: HIV-1 genotype while on failing regimen (requires viral load > 500 copies/mL for success)
- Switch regimen based on resistance profile and treatment history
Resistance Patterns:
- NNRTIs: Low genetic barrier (single mutation confers high-level resistance, often cross-resistance)
- NRTIs: Thymidine analogue mutations (TAMs), M184V (lamivudine/emtricitabine resistance)
- INSTIs: High genetic barrier (DTG, BIC require multiple mutations); RAL lower barrier
- PIs: High genetic barrier (especially darunavir)
Salvage Regimens:
- Require expert consultation
- Use ≥2 fully active agents from different classes
- May include newer agents (lenacapavir, ibalizumab, fostemsavir)
7. Complications
7.1 AIDS-Defining Opportunistic Infections
Pneumocystis jirovecii Pneumonia (PCP):
- Incidence: Most common OI in HIV (CD4 less than 200)
- Presentation: Subacute onset (weeks), dry cough, exertional dyspnoea, fever, desaturation on exertion
- Investigations:
- CXR: Bilateral interstitial infiltrates ("bat-wing"), may be normal (20%)
- "CT: Ground-glass opacification"
- Induced sputum or BAL for immunofluorescence or PCR (sensitivity 90-95%)
- "ABG: Hypoxia, increased A-a gradient"
- LDH elevated (non-specific)
- Treatment: [30]
- "First-line: Co-trimoxazole 120mg/kg/day IV (divided TDS-QDS) × 21 days"
- Adjunctive corticosteroids if PaO₂ less than 9.3 kPa or A-a gradient > 4.7 kPa (prednisolone 40mg BD × 5 days, then wean)
- "Alternatives: IV pentamidine, atovaquone (mild-moderate)"
- Prognosis: Mortality 10-20% with treatment, 100% without
Cerebral Toxoplasmosis:
- Pathogen: Toxoplasma gondii (reactivation of latent infection; CD4 less than 100)
- Presentation: Headache, fever, focal neurological deficit (hemiparesis, ataxia), seizures, altered mental status
- Investigations:
- "MRI: Multiple ring-enhancing lesions with oedema (typically basal ganglia, corticomedullary junction)"
- Toxoplasma IgG positive (> 95%)
- "Differential: Primary CNS lymphoma (usually solitary, EBV PCR in CSF positive)"
- Empirical treatment often given (LP risky with mass effect)
- Treatment:
- Pyrimethamine 200mg loading, then 50-75mg daily + sulfadiazine 1g QDS + folinic acid 15mg daily × 6 weeks
- "Alternative: Co-trimoxazole high-dose"
- Radiological improvement expected within 2 weeks (if no improvement → LP + brain biopsy for lymphoma)
- Secondary prophylaxis: Continue until CD4 > 200 for > 3 months
Cryptococcal Meningoencephalitis:
- Pathogen: Cryptococcus neoformans (environmental fungus; CD4 less than 100)
- Presentation: Subacute headache (95%), fever, altered mental status, cranial nerve palsies; often minimal neck stiffness
- Investigations:
- "Serum CrAg: Highly sensitive screening (> 95%); if positive → LP mandatory"
- "Lumbar puncture: Opening pressure often very high (> 25 cmH₂O), India ink stain (70% sensitive), CrAg CSF (> 99% sensitive), culture (gold standard)"
- "CT/MRI brain (before LP): Often normal; may show hydrocephalus, cryptococcomas"
- Treatment: [31]
- "Induction (2 weeks): Liposomal amphotericin B 3-4mg/kg/day IV + flucytosine 100mg/kg/day PO (divided QDS)"
- "Consolidation (8 weeks): Fluconazole 400mg daily"
- "Maintenance: Fluconazole 200mg daily until CD4 > 200 for > 3 months"
- "Manage raised ICP (key to reducing mortality): Daily therapeutic LP if opening pressure > 25 cmH₂O (remove sufficient CSF to halve opening pressure)"
- Delay ART for 4-6 weeks (reduce IRIS risk)
- Prognosis: Mortality 20-30% even with treatment
CMV Disease:
- Retinitis (most common):
- CD4 less than 50 cells/μL
- "Presentation: Floaters, visual field defects, progressive painless visual loss"
- Fundoscopy: "Pizza" retina - retinal haemorrhages, exudates, necrosis
- "Treatment: Ganciclovir or foscarnet (induction then maintenance); valganciclovir PO alternative"
- "Ophthalmology emergency: Risk of retinal detachment"
- Colitis: Bloody diarrhoea, abdominal pain; colonoscopy shows ulceration; biopsy shows CMV inclusion bodies
- Oesophagitis: Odynophagia, dysphagia
- Radiculopathy: Ascending flaccid paralysis (cauda equina)
Mycobacterium avium Complex (MAC):
- Disseminated disease: CD4 less than 50 cells/μL
- Presentation: Fever, night sweats, weight loss, diarrhoea, hepatosplenomegaly, lymphadenopathy, cytopenias
- Investigations: Blood cultures (mycobacterial), CT abdomen (mesenteric lymphadenopathy)
- Treatment: Clarithromycin 500mg BD + ethambutol 15mg/kg daily ± rifabutin; treat for 12 months minimum
7.2 AIDS-Defining Malignancies
Kaposi Sarcoma (KS):
- Pathogen: HHV-8 (KSHV)
- Presentation: Violaceous/purple macules, plaques, or nodules (skin, oral mucosa); may involve GI tract, lungs
- Staging: Limited cutaneous vs extensive (visceral, oedema, constitutional symptoms)
- Treatment: ART alone (may induce regression); chemotherapy (liposomal doxorubicin, paclitaxel) for extensive disease
Non-Hodgkin Lymphoma:
- High-grade B-cell (Burkitt, diffuse large B-cell, primary CNS lymphoma)
- EBV-associated
- Treatment: Chemotherapy (R-CHOP) + ART
Invasive Cervical Carcinoma:
- HPV-associated
- Screening vital (annual cervical cytology in women with HIV)
7.3 Immune Reconstitution Inflammatory Syndrome (IRIS)
Definition: Paradoxical clinical deterioration due to restoration of immune responses after ART initiation. [10]
Epidemiology: 10-25% of ART initiators, especially if CD4 less than 50 cells/μL.
Risk Factors:
- Low CD4 nadir (less than 50 cells/μL)
- High baseline viral load
- Rapid viral load decline
- Pre-existing subclinical opportunistic infection
Common IRIS Presentations:
- TB-IRIS (most common): Fever, worsening respiratory symptoms, lymphadenopathy, paradoxical enlargement of TB lesions (2-12 weeks post-ART)
- Cryptococcal-IRIS: Worsening meningitis symptoms, cryptococcomas
- CMV-IRIS: Uveitis, vitritis (after CMV retinitis treated)
- Mycobacterial IRIS (MAC, leprosy)
- Herpes zoster
- Sarcoidosis, autoimmune conditions
Management:
- Continue ART (unless life-threatening IRIS)
- Treat underlying opportunistic infection
- NSAIDs for mild symptoms
- Corticosteroids for severe IRIS (prednisone 1mg/kg, taper over weeks)
- Consider temporary ART interruption only if severe CNS IRIS
7.4 Non-AIDS Complications
Cardiovascular Disease: [32]
- 1.5-2× increased risk of myocardial infarction vs HIV-negative (independent of traditional risk factors)
- Mechanisms: Chronic inflammation, immune activation, endothelial dysfunction, ART effects (some PIs, abacavir)
- Management: Aggressive CV risk modification, statins (beware drug interactions)
Chronic Kidney Disease:
- HIV-associated nephropathy (HIVAN): Collapsing FSGS, rapid progression to ESRD (especially Black African ancestry)
- Tenofovir nephrotoxicity (TDF > TAF)
- Screen with eGFR and urinalysis
Bone Disease:
- Osteopenia/osteoporosis (2-3× increased fracture risk)
- Causes: HIV itself, ART (tenofovir, PIs), traditional risk factors
- DEXA screening and vitamin D/calcium supplementation
Neurocognitive Impairment:
- HIV-associated neurocognitive disorder (HAND): Spectrum from asymptomatic to dementia
- Prevalence: 30-50% mild impairment even with viral suppression
- Pathogenesis: Ongoing CNS inflammation, co-morbidities
Malignancies (Non-AIDS-Defining):
- Increased risk: Anal cancer (HPV), lung cancer, Hodgkin lymphoma, hepatocellular carcinoma (HBV/HCV)
- Screening and prevention important
Accelerated Aging:
- Premature age-related comorbidities (frailty, polypharmacy)
- Chronic inflammation ("inflammaging")
8. Prognosis and Outcomes
Life Expectancy with ART
Modern Era (2015 onwards): [4]
- Individuals diagnosed with HIV at age 20 who start ART promptly (CD4 > 350) can expect to live to ~78 years (approaching general population life expectancy)
- Those starting ART with CD4 200-349: life expectancy ~73 years
- Late diagnosis (CD4 less than 200): life expectancy reduced by ~10-13 years
Key Predictors of Outcome:
- CD4 count at ART initiation (most important)
- Age at diagnosis
- Viral suppression (undetectable viral load)
- Adherence to ART
- Co-morbidities (HBV, HCV, cardiovascular disease)
Late Diagnosis
Definition: CD4 less than 350 cells/μL at diagnosis. [5]
UK Statistics:
- 42% of new diagnoses are "late" (CD4 less than 350)
- 25% have CD4 less than 200 (very late, presenting with AIDS)
Consequences:
- 10-fold higher mortality in first year vs early diagnosis
- Increased AIDS-related morbidity
- Missed opportunities for prevention (higher transmission during undiagnosed period)
- Higher healthcare costs
Strategies to Reduce Late Diagnosis:
- Expanded HIV testing in healthcare settings (opt-out testing in high-prevalence areas)
- Testing for indicator conditions
- Community testing initiatives
- Routine testing in primary care, emergency departments
U=U and Transmission Prevention
- Undetectable viral load on ART = zero risk of sexual transmission [6,7,8]
- Transformed HIV prevention, reduced stigma
- Allows PLHIV to have unprotected sex with partners without transmitting virus
- Enables conception and pregnancy without transmission risk (vertical transmission less than 0.1% with viral suppression)
9. Prevention
9.1 Pre-Exposure Prophylaxis (PrEP)
Definition: Antiretroviral medication taken by HIV-negative individuals at risk of HIV acquisition. [33]
Efficacy:
- Tenofovir disoproxil fumarate/emtricitabine (TDF/FTC, Truvada): > 90% efficacy in MSM with adherent use
- iPrEx trial: 44% reduction in HIV incidence (intention-to-treat), 92% in adherent users
- PROUD trial (UK): 86% reduction in HIV incidence vs deferred PrEP
Regimens:
- Daily PrEP: TDF/FTC 1 tablet daily (continuous protection)
- Event-driven (on-demand) PrEP: "2-1-1" dosing (2 pills 2-24h before sex, 1 pill 24h after, 1 pill 48h after) - efficacy demonstrated in MSM (IPERGAY trial: 86% reduction)
Indications (UK BASHH/BHIVA):
- MSM or transgender individuals with condomless anal sex
- HIV-negative partner in serodifferent relationship (if HIV-positive partner not virally suppressed)
- Heterosexuals from high-prevalence region with condomless sex
- People who inject drugs sharing equipment
Monitoring:
- Baseline: HIV test (exclude HIV), renal function, HBV serology, STI screen
- Follow-up: HIV test every 3 months, renal function every 6-12 months, STI screening
Discontinuation: Stop if HIV acquired (test every 3 months); can stop if no longer at risk.
9.2 Post-Exposure Prophylaxis (PEP)
Definition: Short course of ART after potential HIV exposure to prevent infection. [34]
Indications:
- Condomless sex with person known or likely to be HIV-positive
- Needlestick injury or mucous membrane exposure (healthcare)
- Sexual assault
- Sharing injecting equipment
Timing:
- Must start within 72 hours of exposure (ideally less than 24 hours)
- Efficacy decreases with delay
Regimen (UK):
- Tenofovir/emtricitabine + raltegravir (or dolutegravir) for 28 days
- Alternative: TDF/FTC + darunavir/ritonavir
Monitoring:
- Baseline HIV test (exclude infection)
- Follow-up HIV testing at 4, 8, and 12 weeks (exclude seroconversion)
- Renal function at baseline
- STI screen
9.3 Prevention of Vertical Transmission
Interventions: [27]
- Maternal ART (suppress viral load to less than 50 copies/mL)
- Mode of delivery: Vaginal if viral load less than 50; elective caesarean if > 50 or unknown
- Infant prophylaxis: Zidovudine 4 weeks (if maternal viral load suppressed); combination ART if high risk
- Infant feeding: Formula feeding in high-income countries (avoid breastfeeding)
Outcomes:
- Vertical transmission rate less than 0.1% with full interventions (vs 15-45% without)
9.4 Public Health Measures
- Test and Treat: Diagnose and treat all PLHIV → reduces community viral load → reduces transmission
- Condom distribution
- Needle exchange programmes (harm reduction for PWID)
- Education and stigma reduction
- Partner notification and testing
10. Evidence and Guidelines
Key Guidelines
| Guideline | Organisation | Year | Key Recommendations |
|---|---|---|---|
| Management of HIV in Adults | BHIVA (UK) | 2022 | Start ART in all PLHIV regardless of CD4; prefer INSTI-based regimens [23] |
| Adult and Adolescent ART | DHHS (US) | 2023 | Integrase inhibitor + 2 NRTIs first-line; same-day ART initiation supported [26] |
| Consolidated Guidelines on HIV Prevention, Testing, and Treatment | WHO | 2021 | Universal test-and-treat; dolutegravir-based regimens preferred [35] |
| HIV Testing | NICE (UK) | 2016 | Expanded testing in high-prevalence areas; indicator condition testing [22] |
Landmark Trials
START Trial (Strategic Timing of Antiretroviral Treatment) (2015): [24]
- Question: When to start ART - immediately (CD4 > 500) vs deferred (CD4 less than 350)?
- Result: Immediate ART reduced serious AIDS events by 72% and serious non-AIDS events by 39%
- Impact: Established "treat all" regardless of CD4 count
PARTNER/PARTNER2 Studies (2016, 2018): [6,7]
- Question: Can HIV be transmitted if viral load undetectable?
- Result: Zero transmissions in 135,000 condomless sex acts (upper 95% CI: 0.23/100 person-years)
- Impact: Established U=U (Undetectable = Untransmittable)
TEMPRANO Trial (2015): [25]
- Question: Early ART in resource-limited settings?
- Result: Immediate ART (vs deferred to CD4 less than 500) reduced severe morbidity by 44% in West Africa
- Impact: Reinforced early ART benefit across settings
NAMSAL Trial (2019):
- Question: Dolutegravir vs efavirenz in Sub-Saharan Africa?
- Result: DTG superior efficacy, better tolerability
- Impact: DTG became WHO preferred first-line
ADVANCE Trial (2019):
- Question: TAF vs TDF + dolutegravir vs efavirenz?
- Result: DTG + TAF superior virological suppression, better bone/renal safety vs TDF
- Impact: TAF preferred in settings with renal/bone concerns
11. Patient and Layperson Explanation
What is HIV?
HIV (Human Immunodeficiency Virus) is a virus that attacks your immune system - specifically the cells that help your body fight infections. Over time, HIV destroys these cells, making it harder for your body to defend itself against infections and some cancers.
Is HIV the same as AIDS?
No. HIV is the virus. AIDS (Acquired Immunodeficiency Syndrome) is the late stage of HIV infection, when the immune system is severely damaged. Most people with HIV today do not have AIDS and never will, as long as they take treatment.
How is HIV treated?
HIV is treated with antiretroviral therapy (ART) - a combination of medications (usually 1-3 pills) taken once daily. ART:
- Stops the virus from multiplying
- Allows your immune system to recover
- Prevents HIV from progressing to AIDS
- Prevents transmission to others
Can HIV be cured?
Not yet. ART controls HIV but does not eliminate it completely. The virus remains in your body in a dormant state. If you stop treatment, the virus will start multiplying again. Research into a cure is ongoing.
What is "undetectable"?
After starting ART, the amount of virus in your blood (viral load) drops dramatically. When it reaches very low levels (less than 50 copies/mL), it's called undetectable. This means:
- The virus cannot be detected by standard tests
- Your immune system can recover
- You cannot pass HIV to sexual partners (U=U: Undetectable = Untransmittable)
Can I have children?
Yes. With undetectable viral load on ART, the risk of passing HIV to your partner or baby is extremely low (less than 0.1%). With proper medical care during pregnancy and delivery, mothers with HIV can have healthy, HIV-negative babies.
Will I live a normal life?
Yes. People diagnosed early and taking ART consistently can expect a near-normal life expectancy and quality of life. You can work, have relationships, have children, and participate fully in life. The key is:
- Taking medication every day
- Regular monitoring with your HIV clinic
- Maintaining a healthy lifestyle
What about side effects?
Modern HIV medications have far fewer side effects than older drugs. Most people tolerate them very well. Some may experience mild nausea or headache initially, which usually resolves. Serious side effects are rare. Your doctor will monitor you regularly.
Do I need to tell people?
You have no legal obligation to disclose your HIV status to most people (exceptions: sexual partners in some jurisdictions, blood/organ donation). It's your personal choice who to tell. Many people choose to share with close friends, family, or partners, but it's entirely up to you.
How do I take care of myself?
- Take your medications every day (never miss doses)
- Attend regular clinic appointments
- Don't smoke; limit alcohol
- Eat a balanced diet and stay active
- Manage stress and mental health
- Practice safe sex (prevents other STIs)
- Get vaccinated (flu, pneumococcal, hepatitis)
12. Examination Focus
High-Yield MRCP/Postgraduate Topics
Diagnosis and Window Period:
- Fourth-generation tests detect p24 antigen + antibodies (window period ~3-4 weeks)
- Point-of-care tests (antibody only) have longer window (~3 months)
- Acute retroviral syndrome: high viral load, negative/indeterminate antibody
Indicator Conditions (should prompt HIV test):
- Seborrhoeic dermatitis (severe), oral candidiasis, hairy leukoplakia
- Herpes zoster (less than 50 years), unexplained lymphadenopathy/weight loss
- Any STI, hepatitis B/C, tuberculosis, lymphoma
CD4 Count Stratification (opportunistic infection risk):
- less than 200: PCP, histoplasmosis
- less than 100: Toxoplasmosis, cryptococcal meningitis
- less than 50: MAC, CMV disease
First-Line ART:
- Standard: 2 NRTIs (tenofovir + emtricitabine) + integrase inhibitor (dolutegravir or bictegravir)
- Single-tablet regimens improve adherence
- HLA-B*5701 testing mandatory before abacavir (hypersensitivity risk)
Opportunistic Infection Prophylaxis:
- PCP: Co-trimoxazole if CD4 less than 200
- Toxoplasma: Co-trimoxazole if CD4 less than 100 and toxoplasma IgG+
- MAC: Azithromycin if CD4 less than 50
IRIS (Immune Reconstitution Inflammatory Syndrome):
- Paradoxical worsening after ART initiation (immune system "wakes up")
- Most common with TB, cryptococcal infection, CMV
- Management: Continue ART, treat OI, consider corticosteroids
U=U (Undetectable = Untransmittable):
- Viral load less than 50 copies/mL on ART → zero sexual transmission risk
- PARTNER/PARTNER2 studies: zero transmissions in 135,000 condomless acts
PrEP (Pre-Exposure Prophylaxis):
- Tenofovir/emtricitabine for HIV-negative individuals at high risk
-
90% efficacy with adherence
- Daily or event-driven (2-1-1) dosing
PEP (Post-Exposure Prophylaxis):
- Start within 72 hours (ideally less than 24h) after high-risk exposure
- TDF/FTC + raltegravir or dolutegravir for 28 days
Common Exam Scenarios
SBA Question: "A 28-year-old MSM presents with fever, rash, and sore throat 3 weeks after unprotected sex. Monospot test is negative. What is the next best investigation?"
- Answer: HIV antigen/antibody test (4th generation) - suspect acute retroviral syndrome
SBA Question: "Before starting abacavir in HIV treatment, which test is mandatory?"
- Answer: HLA-B*5701 (positive = contraindication due to hypersensitivity risk)
SBA Question: "A patient with HIV (CD4 80) presents with multiple ring-enhancing brain lesions. What is the most likely diagnosis?"
- Answer: Cerebral toxoplasmosis (vs primary CNS lymphoma - usually solitary)
SBA Question: "What is the target viral load on effective ART?"
- Answer: less than 50 copies/mL (undetectable)
Clinical Case: "Patient on ART for 6 months, CD4 risen from 50 to 150, develops fever, cough, and worsening lymphadenopathy. Diagnosis?"
- Answer: IRIS (TB-IRIS most likely) - immune reconstitution revealing subclinical TB
Viva Topics
Antiretroviral Drug Classes:
- NRTIs: Chain terminators (tenofovir, emtricitabine, abacavir)
- INSTIs: Block integration (dolutegravir, bictegravir - high barrier to resistance)
- NNRTIs: Allosteric inhibition (efavirenz, rilpivirine - low genetic barrier)
- PIs: Prevent virion maturation (darunavir - require ritonavir boosting)
Opportunistic Infection Treatment:
- PCP: Co-trimoxazole + adjunctive corticosteroids (if PaO₂ less than 9.3 kPa)
- Toxoplasmosis: Pyrimethamine + sulfadiazine + folinic acid; empirical treatment given
- Cryptococcal meningitis: Amphotericin + flucytosine; manage raised ICP with serial LPs
START Trial: Immediate ART (CD4 > 500) vs deferred (CD4 less than 350) → 57% reduction in serious events with immediate ART → "treat all" strategy
PARTNER Study: Serodifferent couples, HIV-positive partner with undetectable viral load → zero transmissions in 58,000 condomless sex acts → established U=U
13. References
Primary Sources
-
Fauci AS, Lane HC. Human Immunodeficiency Virus Disease: AIDS and Related Disorders. In: Kasper DL, et al, editors. Harrison's Principles of Internal Medicine. 21st ed. New York: McGraw-Hill; 2022.
-
Deeks SG, Overbaugh J, Phillips A, Buchbinder S. HIV infection. Nat Rev Dis Primers. 2015;1:15035. doi:10.1038/nrdp.2015.35
-
Palella FJ Jr, Delaney KM, Moorman AC, et al. Declining morbidity and mortality among patients with advanced human immunodeficiency virus infection. N Engl J Med. 1998;338(13):853-860. doi:10.1056/NEJM199803263381301
-
Trickey A, May MT, Vehreschild JJ, et al. Survival of HIV-positive patients starting antiretroviral therapy between 1996 and 2013: a collaborative analysis of cohort studies. Lancet HIV. 2017;4(8):e349-e356. doi:10.1016/S2352-3018(17)30066-8
-
Katz IT, Maughan-Brown B. Improved life expectancy of people living with HIV: who is left behind? Lancet HIV. 2017;4(8):e324-e326. doi:10.1016/S2352-3018(17)30086-3
-
Rodger AJ, Cambiano V, Bruun T, et al. Sexual activity without condoms and risk of HIV transmission in serodifferent couples when the HIV-positive partner is using suppressive antiretroviral therapy. JAMA. 2016;316(2):171-181. doi:10.1001/jama.2016.5148
-
Rodger AJ, Cambiano V, Bruun T, et al. Risk of HIV transmission through condomless sex in serodifferent gay couples with the HIV-positive partner taking suppressive antiretroviral therapy (PARTNER): final results of a multicentre, prospective, observational study. Lancet. 2019;393(10189):2428-2438. doi:10.1016/S0140-6736(19)30418-0
-
Bavinton BR, Pinto AN, Phanuphak N, et al. Viral suppression and HIV transmission in serodiscordant male couples: an international, prospective, observational, cohort study. Lancet HIV. 2018;5(8):e438-e447. doi:10.1016/S2352-3018(18)30132-2
-
Cohen MS, Shaw GM, McMichael AJ, Haynes BF. Acute HIV-1 Infection. N Engl J Med. 2011;364(20):1943-1954. doi:10.1056/NEJMra1011874
-
Haddow LJ, Easterbrook PJ, Mosam A, et al. Defining immune reconstitution inflammatory syndrome: evaluation of expert opinion versus 2 case definitions in a South African cohort. Clin Infect Dis. 2009;49(9):1424-1432. doi:10.1086/630208
-
UNAIDS. Global HIV & AIDS statistics — Fact sheet 2023. Available at: https://www.unaids.org/en/resources/fact-sheet
-
UK Health Security Agency. HIV annual data tables to end of December 2022. Published 2023.
-
Patel P, Borkowf CB, Brooks JT, Lasry A, Lansky A, Mermin J. Estimating per-act HIV transmission risk: a systematic review. AIDS. 2014;28(10):1509-1519. doi:10.1097/QAD.0000000000000298
-
Townsend CL, Byrne L, Cortina-Borja M, et al. Earlier initiation of ART and further decline in mother-to-child HIV transmission rates, 2000-2011. AIDS. 2014;28(7):1049-1057. doi:10.1097/QAD.0000000000000212
-
Wilen CB, Tilton JC, Doms RW. HIV: cell binding and entry. Cold Spring Harb Perspect Med. 2012;2(8):a006866. doi:10.1101/cshperspect.a006866
-
Doitsh G, Greene WC. Dissecting how CD4 T cells are lost during HIV infection. Cell Host Microbe. 2016;19(3):280-291. doi:10.1016/j.chom.2016.02.012
-
Brenchley JM, Schacker TW, Ruff LE, et al. CD4+ T cell depletion during all stages of HIV disease occurs predominantly in the gastrointestinal tract. J Exp Med. 2004;200(6):749-759. doi:10.1084/jem.20040874
-
Langford SE, Ananworanich J, Cooper DA. Predictors of disease progression in HIV infection: a review. AIDS Res Ther. 2007;4:11. doi:10.1186/1742-6405-4-11
-
Deeks SG, Walker BD. Human immunodeficiency virus controllers: mechanisms of durable virus control in the absence of antiretroviral therapy. Immunity. 2007;27(3):406-416. doi:10.1016/j.immuni.2007.08.010
-
Shiels MS, Islam JY, Rosenberg PS, Hall HI, Jacobson E, Engels EA. Projected Cancer Incidence Rates and Burden of Incident Cancer Cases in HIV-Infected Adults in the United States Through 2030. Ann Intern Med. 2018;168(12):866-873. doi:10.7326/M17-2499
-
Branson BM, Owen SM, Wesolowski LG, et al. Laboratory Testing for the Diagnosis of HIV Infection: Updated Recommendations. CDC; 2014.
-
National Institute for Health and Care Excellence. HIV testing: increasing uptake among people who may have undiagnosed HIV. NICE guideline [NG60]. Published December 2016.
-
British HIV Association. British HIV Association guidelines for the treatment of HIV-1-positive adults with antiretroviral therapy 2022. HIV Med. 2022;23 Suppl 4:2-26. doi:10.1111/hiv.13353
-
INSIGHT START Study Group. Initiation of Antiretroviral Therapy in Early Asymptomatic HIV Infection. N Engl J Med. 2015;373(9):795-807. doi:10.1056/NEJMoa1506816
-
TEMPRANO ANRS 12136 Study Group. A Trial of Early Antiretrovirals and Isoniazid Preventive Therapy in Africa. N Engl J Med. 2015;373(9):808-822. doi:10.1056/NEJMoa1507198
-
Panel on Antiretroviral Guidelines for Adults and Adolescents. Guidelines for the Use of Antiretroviral Agents in Adults and Adolescents with HIV. Department of Health and Human Services. Updated 2023.
-
British HIV Association. BHIVA guidelines for the management of HIV in pregnancy and postpartum 2018 (2020 third interim update). HIV Med. 2020;21 Suppl 6:1-85. doi:10.1111/hiv.12959
-
British HIV Association. BHIVA guidelines for the treatment of tuberculosis (TB) in adults living with HIV 2019. HIV Med. 2019;20 Suppl 6:s2-s83. doi:10.1111/hiv.12792
-
Paterson DL, Swindells S, Mohr J, et al. Adherence to protease inhibitor therapy and outcomes in patients with HIV infection. Ann Intern Med. 2000;133(1):21-30. doi:10.7326/0003-4819-133-1-200007040-00004
-
Thomas CF Jr, Limper AH. Pneumocystis pneumonia. N Engl J Med. 2004;350(24):2487-2498. doi:10.1056/NEJMra032588
-
Perfect JR, Dismukes WE, Dromer F, et al. Clinical practice guidelines for the management of cryptococcal disease: 2010 update by the Infectious Diseases Society of America. Clin Infect Dis. 2010;50(3):291-322. doi:10.1086/649858
-
Feinstein MJ, Hsue PY, Benjamin LA, et al. Characteristics, Prevention, and Management of Cardiovascular Disease in People Living With HIV: A Scientific Statement From the American Heart Association. Circulation. 2019;140(2):e98-e124. doi:10.1161/CIR.0000000000000695
-
McCormack S, Dunn DT, Desai M, et al. Pre-exposure prophylaxis to prevent the acquisition of HIV-1 infection (PROUD): effectiveness results from the pilot phase of a pragmatic open-label randomised trial. Lancet. 2016;387(10013):53-60. doi:10.1016/S0140-6736(15)00056-2
-
Mayer KH, Mimiaga MJ, Cohen D, et al. Tenofovir DF plus lamivudine or emtricitabine for nonoccupational postexposure prophylaxis (NPEP) in a Boston Community Health Center. J Acquir Immune Defic Syndr. 2008;47(4):494-499. doi:10.1097/QAI.0b013e31816e6c8e
-
World Health Organization. Consolidated guidelines on HIV prevention, testing, treatment, service delivery and monitoring: recommendations for a public health approach. Geneva: WHO; 2021.
Medical Disclaimer: MedVellum content is for educational purposes and clinical reference. Clinical decisions should account for individual patient circumstances and current guidelines. Always consult appropriate specialists and use the most recent evidence-based protocols for patient care.
Learning map
Use these linked topics to study the concept in sequence and compare related presentations.
Prerequisites
Start here if you need the foundation before this topic.
- CD4 T-Cell Immunology
- Retroviral Replication
Consequences
Complications and downstream problems to keep in mind.
- Pneumocystis Pneumonia
- Cryptococcal Meningitis
- Toxoplasmosis
- CMV Retinitis
- Kaposi Sarcoma
- Tuberculosis