Hypoglycaemia in Adults
Hypoglycaemia is defined as blood glucose below 4.0 mmol/L (72 mg/dL) and represents one of the most common acute complications of diabetes management. It occurs when glucose delivery or production fails to meet...
What matters first
Hypoglycaemia is defined as blood glucose below 4.0 mmol/L (72 mg/dL) and represents one of the most common acute complications of diabetes management. It occurs when glucose delivery or production fails to meet...
GCS below 8 or seizure activity
6 Jan 2025
Generated educational material; verify before clinical use.
Visible references section
See the concept before reading it
Study the key anatomy, imaging, and decision pathways as full teaching plates.
Clinical board
A visual summary of the highest-yield teaching signals on this page.
Urgent signals
Safety-critical features pulled from the topic metadata.
- GCS below 8 or seizure activity
- Unable to self-treat or protect airway
- Recurrent hypoglycaemia within 24 hours
- Hypoglycaemia unawareness (no warning symptoms)
Linked comparisons
Differentials and adjacent topics worth opening next.
- Insulinoma
- Adrenal Insufficiency
Content status and exam context
This page is AI-generated educational content. It may contain errors or omissions and is not a substitute for current guidelines, local protocols, senior clinical judgement, or professional medical advice.
MedVellum does not claim an individual clinician reviewer, board certification, or professional credential for this page unless a future version names a real, verifiable reviewer.
Clinical explanation and evidence
Hypoglycaemia in Adults
Clinical Overview
Summary
Hypoglycaemia is defined as blood glucose below 4.0 mmol/L (72 mg/dL) and represents one of the most common acute complications of diabetes management. It occurs when glucose delivery or production fails to meet metabolic demand, or when insulin or insulin secretagogues drive excessive glucose uptake. The clinical spectrum ranges from mild autonomic symptoms (sweating, tremor) manageable with oral carbohydrate, to severe neuroglycopenia requiring emergency intervention with parenteral glucose or glucagon. [1,2]
The condition represents the major limiting factor in achieving optimal glycaemic control in diabetes. The Diabetes Control and Complications Trial (DCCT) demonstrated that intensive glycaemic control reduces microvascular complications but increases severe hypoglycaemia risk threefold. [3] Modern management emphasizes structured education, continuous glucose monitoring, and individualized glycaemic targets to minimize hypoglycaemia burden while maintaining metabolic benefits.
Non-diabetic hypoglycaemia, though less common, presents unique diagnostic challenges requiring systematic evaluation for insulinoma, critical illness, organ failure, and other rare causes. The cornerstone of diagnosis remains Whipple's triad: symptoms of hypoglycaemia, documented low plasma glucose, and symptom resolution with glucose administration. [4]
Key Facts
| Parameter | Value | Context |
|---|---|---|
| Diagnostic threshold | Below 4.0 mmol/L (72 mg/dL) | "4 is the floor" |
- treat even if asymptomatic | | Severe hypoglycaemia | Requiring third-party assistance | International Hypoglycaemia Study Group Level 3 | | Neuroglycopenic threshold | Below 3.0 mmol/L (54 mg/dL) | Cognitive impairment begins | | Brain injury threshold | Below 2.0 mmol/L (36 mg/dL) | Prolonged exposure risks permanent damage | | Type 1 diabetes incidence | 1-3 severe episodes/patient/year | Higher with intensive control | | Type 2 diabetes incidence | 0.1-0.7 episodes/patient/year | Varies by treatment regimen | | Mortality association | 6-10% of Type 1 deaths | "Dead-in-bed" syndrome | | Counter-regulatory failure | Within 5 years of Type 1 diagnosis | Loss of glucagon response |
Clinical Pearls
"4 is the floor" — Treat any glucose below 4.0 mmol/L, even in asymptomatic patients, to prevent progression to severe hypoglycaemia.
Sulphonylurea hypoglycaemia is not a simple hypo — Can persist 24-72 hours due to prolonged half-life and renal accumulation. ALWAYS ADMIT for observation and IV dextrose infusion.
Avoid 50% dextrose — Use 10% or 20% glucose instead. 50% dextrose causes severe phlebitis, tissue necrosis if extravasated, and paradoxical rebound hypoglycaemia.
IM glucagon is less effective than you think — Requires hepatic glycogen stores. Ineffective in alcohol intoxication, prolonged fasting, liver disease, and chronic malnutrition.
The rule of 15 — 15g fast-acting carbohydrate, wait 15 minutes, recheck. Repeat if still below 4.0 mmol/L. Then give long-acting carbohydrate.
Rebound hyperglycaemia is expected — Counter-regulatory hormones cause post-hypoglycaemia hyperglycaemia. Resist aggressive insulin correction to avoid recurrent hypoglycaemia.
Why This Matters Clinically
Hypoglycaemia remains the principal barrier to achieving recommended glycaemic targets in insulin-treated diabetes. Fear of hypoglycaemia drives many patients to maintain suboptimal glucose control, perpetuating long-term microvascular and macrovascular complications. [5]
Recurrent hypoglycaemia initiates a vicious cycle: each episode blunts counter-regulatory responses, lowering the threshold for autonomic symptoms (hypoglycaemia-associated autonomic failure). This creates hypoglycaemia unawareness, where patients progress directly to neuroglycopenia without warning symptoms, increasing severe hypoglycaemia risk sixfold. [6]
Severe hypoglycaemia carries immediate risks including seizures, cardiac arrhythmias, aspiration, traumatic injury, and in rare cases, sudden death. Emerging evidence links recurrent severe hypoglycaemia to cognitive decline, cardiovascular events, and increased all-cause mortality, particularly in older adults with Type 2 diabetes. [7,8]
In hospital settings, hypoglycaemia affects 5-15% of inpatients with diabetes and associates with increased length of stay, hospital-acquired infections, and mortality. Structured protocols using basal-bolus insulin regimens reduce inpatient hypoglycaemia while maintaining glycaemic control. [9]
Epidemiology
Incidence and Prevalence
The epidemiology of hypoglycaemia varies dramatically by diabetes type, treatment regimen, and glycaemic control intensity.
Type 1 Diabetes
| Population | Severe Hypoglycaemia Rate | Context |
|---|---|---|
| Standard control | 19 per 100 patient-years | DCCT conventional arm [3] |
| Intensive control | 62 per 100 patient-years | DCCT intensive arm (3.3× increase) [3] |
| Modern therapy era | 15-30 per 100 patient-years | T1D Exchange data 2016-2018 [10] |
| CGM users | 8-12 per 100 patient-years | 40-50% reduction with CGM [10] |
| Closed-loop systems | 3-6 per 100 patient-years | Automated insulin delivery systems |
Clinical context: Nearly all Type 1 patients experience mild-moderate hypoglycaemia weekly. Nocturnal hypoglycaemia occurs in 50-60% monthly, often unrecognized. [11]
Type 2 Diabetes
| Treatment | Severe Hypoglycaemia Rate | Annual Risk |
|---|---|---|
| Metformin alone | 0.5 per 1000 patient-years | Very low risk |
| Sulphonylureas | 10-40 per 1000 patient-years | Higher with gliclazide, glibenclamide |
| Basal insulin | 5-15 per 100 patient-years | Increases with duration |
| Basal-bolus insulin | 20-50 per 100 patient-years | Approaches Type 1 risk |
| Insulin + sulphonylurea | 30-60 per 100 patient-years | Additive risk |
Newer therapies (DPP-4 inhibitors, GLP-1 agonists, SGLT2 inhibitors) carry minimal independent hypoglycaemia risk but increase risk when combined with insulin or sulphonylureas. [12]
Non-Diabetic Hypoglycaemia
Rare in general population. Insulinoma incidence: 1-4 per million per year. Post-bariatric hypoglycaemia: 0.2-1% of Roux-en-Y gastric bypass patients. Critical illness hypoglycaemia: 5-15% of ICU admissions. [13]
Risk Factors
Patient Factors
| Risk Factor | Mechanism | Relative Risk |
|---|---|---|
| Long diabetes duration | Counter-regulatory failure | 2-3× |
| Previous severe hypoglycaemia | Recurrence predictor | 3-5× |
| Hypoglycaemia unawareness | Loss of warning symptoms | 6× |
| Tight glycaemic control | HbA1c below 53 mmol/mol (7%) | 2-4× |
| Chronic kidney disease | Reduced insulin clearance | 2-3× |
| Advanced age (over 75) | Polypharmacy, frailty | 1.5-2× |
| Cognitive impairment | Treatment errors | 2-3× |
| Low body weight/malnutrition | Reduced glycogen stores | 2× |
Treatment Factors
- Insulin regimens: Rapid-acting analogues lower risk versus regular insulin; long-acting analogues (glargine, degludec) lower nocturnal hypoglycaemia versus NPH
- Sulphonylureas: Glibenclamide highest risk; gliclazide modified-release lower risk
- Polypharmacy: Beta-blockers mask symptoms; ACE inhibitors may increase insulin sensitivity
- Alcohol: Inhibits gluconeogenesis, prolongs hypoglycaemia duration
Lifestyle Factors
- Missed or delayed meals: Most common precipitant in diabetic hypoglycaemia
- Unplanned exercise: Increases glucose utilization for 4-24 hours post-exercise
- Alcohol consumption: Dose-dependent gluconeogenesis inhibition
- Overnight fasting: Nocturnal hypoglycaemia peaks at 2-4 AM
Exam Detail: MRCP viva pearl: The ACCORD trial demonstrated that intensive glucose lowering (HbA1c target below 42 mmol/mol) in Type 2 diabetes with cardiovascular disease increased mortality, with severe hypoglycaemia implicated as a potential mechanism. This led to individualized glycaemic targets rather than universal intensive control. [14]
Pathophysiology
Normal Glucose Homeostasis
Plasma glucose is maintained within narrow limits (4.0-6.0 mmol/L fasting) through precise hormonal regulation balancing glucose input (dietary absorption, hepatic production) and glucose utilization (brain, muscle, fat).
Glucose Counter-Regulatory Cascade
As plasma glucose falls, a hierarchical hormonal response activates: [15]
| Glucose Level | Response | Mechanism | Timing |
|---|---|---|---|
| 4.5-5.0 mmol/L | Insulin suppression | Reduced glucose uptake peripherally | Immediate |
| 3.6-3.8 mmol/L | Glucagon secretion | Hepatic glycogenolysis | Within 5 minutes |
| 3.2-3.4 mmol/L | Adrenaline secretion | Glycogenolysis + gluconeogenesis | Within 10 minutes |
| 3.0-3.2 mmol/L | Cortisol + growth hormone | Sustained gluconeogenesis | 2-4 hours |
| Below 2.8 mmol/L | Neuroglycopenic symptoms | Brain glucose deprivation | Variable |
Hepatic mechanisms:
- Glycogenolysis: Glucagon and adrenaline activate phosphorylase, releasing glucose-1-phosphate from glycogen (immediate, depletes in 24-48 hours fasting)
- Gluconeogenesis: Cortisol and glucagon stimulate hepatic glucose synthesis from lactate, glycerol, amino acids (slower, sustained)
Pathophysiology in Diabetes
Failure of Counter-Regulation
Type 1 diabetes progressively dismantles glucose counter-regulation: [6]
- Insulin cannot be "switched off": Exogenous insulin continues absorbing from subcutaneous depots regardless of glucose level
- Glucagon response lost early: Alpha-cell dysfunction within 1-5 years of diagnosis eliminates the first defense
- Adrenaline threshold shifts: Recurrent hypoglycaemia lowers the glycaemic threshold for adrenaline release from ~3.2 to ~2.5 mmol/L
- Autonomic failure: Blunted sympathoadrenal response creates hypoglycaemia unawareness
This phenomenon, termed Hypoglycaemia-Associated Autonomic Failure (HAAF), explains why intensive control paradoxically increases severe hypoglycaemia risk. [6]
Insulin Pharmacokinetics in Hypoglycaemia
| Insulin Type | Peak Action | Duration | Hypoglycaemia Risk |
|---|---|---|---|
| Rapid-acting analogues (aspart, lispro) | 1-2 hours | 3-5 hours | Postprandial hypos |
| Short-acting regular | 2-4 hours | 6-8 hours | Late postprandial hypos |
| Intermediate NPH | 4-8 hours | 12-18 hours | Nocturnal hypos (peak at 4 AM) |
| Long-acting basal (glargine, detemir) | Minimal peak | 18-24 hours | Lower nocturnal risk |
| Ultra-long degludec | Flat profile | Beyond 42 hours | Lowest day-to-day variability |
Sulphonylureas bind pancreatic beta-cell ATP-sensitive potassium channels, forcing insulin secretion regardless of glucose level. Longer half-life agents (glibenclamide 10-16 hours) accumulate in renal impairment, causing prolonged, recurrent hypoglycaemia. [16]
Symptoms by Mechanism
Hypoglycaemic symptoms derive from two pathophysiological processes:
Autonomic (Adrenergic) Symptoms
Result from sympathetic nervous system activation and catecholamine release:
- Adrenergic: Tremor, palpitations, anxiety, nervousness
- Cholinergic: Sweating, hunger, paraesthesias
These symptoms typically begin at glucose 3.2-3.6 mmol/L and serve as warning symptoms, prompting self-treatment before neuroglycopenia develops.
Neuroglycopenic Symptoms
Result from direct brain glucose deprivation (brain cannot use alternate fuels acutely):
- Cognitive: Confusion, difficulty concentrating, slurred speech
- Behavioral: Aggression, personality change, inappropriate behavior
- Neurological: Incoordination, visual disturbances, focal deficits
- Severe: Seizures, loss of consciousness, coma
Brain glucose uptake is normally 5-6 mg/dL/min (0.3-0.35 mmol/L/min). When plasma glucose falls below 2.5-3.0 mmol/L, cerebral glucose delivery becomes insufficient, impairing neuronal function within minutes. [15]
Exam Detail: Molecular mechanisms of brain injury: Severe prolonged hypoglycaemia (below 1.5 mmol/L for over 60 minutes) triggers neuronal death via glutamate excitotoxicity, mitochondrial dysfunction, and oxidative stress. Neurons cannot synthesize glucose and have minimal glycogen reserves. Hippocampus, cerebral cortex, and basal ganglia are most vulnerable, explaining cognitive sequelae of recurrent severe hypoglycaemia. [17]
Whipple's Triad
The diagnostic cornerstone for hypoglycaemia, established in 1938 by Allen Whipple for insulinoma diagnosis: [4]
- Symptoms consistent with hypoglycaemia (autonomic or neuroglycopenic)
- Documented low plasma glucose (below 3.0 mmol/L during symptoms)
- Relief of symptoms with glucose administration
All three criteria must be met. Failure of symptom relief suggests alternative diagnosis (stroke, seizure, psychiatric illness).
Clinical Presentation
Classification by Severity
The International Hypoglycaemia Study Group defines three severity levels: [1]
| Level | Glucose | Clinical Features | Action Required |
|---|---|---|---|
| Level 1: Alert | Below 4.0 mmol/L, at or above 3.0 mmol/L | Mild autonomic symptoms; fully conscious | Oral fast-acting carbohydrate (self-treat) |
| Level 2: Serious | Below 3.0 mmol/L | Prominent autonomic symptoms; neuroglycopenia beginning | Immediate oral carbohydrate; assistance may be needed |
| Level 3: Severe | Any glucose level | Cognitive impairment preventing self-treatment; unconscious; seizure | Parenteral glucose or IM glucagon; third-party intervention mandatory |
Autonomic Symptoms
Autonomic symptoms serve as the body's warning system, prompting treatment before neuroglycopenia:
Adrenergic (catecholamine-mediated):
- Tremor (fine, rapid)
- Palpitations
- Anxiety, nervousness
- Tachycardia
Cholinergic (parasympathetic):
- Sweating (often profuse, out of proportion to ambient temperature)
- Hunger
- Paraesthesias (perioral, fingers)
Timing: Typically begin at glucose 3.2-3.6 mmol/L in healthy individuals; threshold may shift to 2.5-3.0 mmol/L in those with recurrent hypoglycaemia (unawareness).
Neuroglycopenic Symptoms
Direct cerebral glucose deprivation produces progressive neurological dysfunction:
Mild-Moderate (glucose 2.5-3.0 mmol/L):
- Difficulty concentrating
- Confusion, disorientation
- Slurred speech
- Blurred or double vision
- Dizziness, lightheadedness
Severe (glucose below 2.5 mmol/L):
- Abnormal behavior (aggression, combativeness)
- Incoordination, ataxia
- Focal neurological deficits (mimicking stroke)
- Seizures (generalized tonic-clonic)
- Loss of consciousness, coma
Clinical Pearl: "The hypo stroke mimic": Hypoglycaemia can present with acute focal neurology (hemiparesis, aphasia, visual field defects) indistinguishable from stroke. ALWAYS check capillary glucose in suspected stroke before thrombolysis. Glucose administration produces dramatic recovery within minutes, avoiding inappropriate thrombolytic therapy.
Hypoglycaemia Unawareness
Hypoglycaemia unawareness is defined as loss of perception of autonomic warning symptoms during hypoglycaemia, leading patients to progress directly to neuroglycopenia without recognizing hypoglycaemia. [6]
Prevalence:
- Type 1 diabetes: 20-40%
- Type 2 diabetes on insulin: 10-15%
Mechanisms:
- Recurrent hypoglycaemia blunts sympathoadrenal response
- Autonomic neuropathy impairs catecholamine release
- Beta-blocker therapy masks adrenergic symptoms
- Sleep (nocturnal hypoglycaemia unrecognized in 50-60%)
Consequences:
- Six-fold increase in severe hypoglycaemia risk
- Increased motor vehicle accidents
- Occupational hazards
- Reduced quality of life, fear affecting daily activities
Reversal: Strict avoidance of hypoglycaemia (target glucose above 5.0 mmol/L) for 2-3 weeks can partially restore awareness in some patients.
Nocturnal Hypoglycaemia
Hypoglycaemia during sleep presents unique challenges:
Peak incidence: 2:00-4:00 AM (nadir of diurnal counter-regulatory hormones)
Symptoms (often subtle or absent):
- Vivid nightmares, night sweats
- Morning headache
- Unrefreshed sleep
- Morning hyperglycaemia (Somogyi phenomenon)
Detection: Continuous glucose monitoring reveals nocturnal hypoglycaemia in 50-60% of Type 1 patients monthly, often completely asymptomatic. [11]
Examination Findings
During Acute Hypoglycaemia
| System | Findings |
|---|---|
| General | Diaphoresis, pallor, tremor |
| Cardiovascular | Tachycardia, palpitations, rarely bradycardia in severe cases |
| Neurological | Confusion, slurred speech, incoordination, focal deficits, reduced GCS, seizure |
| Behavioral | Aggression, inappropriate affect, combativeness |
Post-Recovery
- Rapid resolution of all symptoms with glucose administration (confirms diagnosis)
- Transient neurological deficits may persist 15-30 minutes in severe hypoglycaemia
- Persistent deficits suggest alternative pathology (stroke, intracranial event)
Exam Detail: OSCE pearl: When examining a confused patient with suspected hypoglycaemia, immediately check capillary glucose. If low, state: "This patient has neuroglycopenia from hypoglycaemia, glucose is X mmol/L. I would administer oral glucose if conscious and protecting airway, or IV 10% glucose if unable to swallow safely." Do NOT complete a full neurological examination before treating life-threatening hypoglycaemia.
Differential Diagnosis
Diabetic vs. Non-Diabetic Hypoglycaemia
The initial approach differs fundamentally:
| Feature | Diabetic Hypoglycaemia | Non-Diabetic Hypoglycaemia |
|---|---|---|
| Common causes | Insulin, sulphonylureas, missed meals | Insulinoma, critical illness, organ failure |
| Typical timing | Predictable (post-insulin, pre-meal) | Random, often fasting |
| Recurrence pattern | Episodic, manageable with education | Persistent, progressive without treatment |
| Whipple's triad | Often incomplete (symptoms may be absent) | Essential for diagnosis |
| Investigation | Minimal (review medications, BM diary) | Extensive (supervised fast, insulin, C-peptide) |
Causes of Hypoglycaemia
Drug-Induced Hypoglycaemia
Diabetic medications:
- Insulin (all formulations): Most common cause
- Sulphonylureas: Glibenclamide, gliclazide, glimepiride, tolbutamide
- Meglitinides: Repaglinide, nateglinide (shorter duration than sulphonylureas)
Non-diabetic medications:
- Alcohol: Inhibits gluconeogenesis; hypoglycaemia 6-36 hours after binge drinking
- Quinine: Used for malaria; stimulates insulin release
- Pentamidine: Beta-cell toxicity causing insulin release then destruction
- Fluoroquinolones: Gatifloxacin (now withdrawn), others rarely
- Salicylates: High-dose aspirin in children
- Beta-blockers: Mask symptoms; rarely cause hypoglycaemia directly
Endogenous Hyperinsulinism
| Cause | Mechanism | Key Features |
|---|---|---|
| Insulinoma | Autonomous insulin secretion from pancreatic beta-cell tumor | Fasting hypoglycaemia, inappropriate insulin during hypo, imaging shows tumor |
| Post-bariatric hypoglycaemia | Exaggerated GLP-1 response after Roux-en-Y gastric bypass | Postprandial (1-3 hours after eating), not fasting |
| Autoimmune hypoglycaemia | Insulin autoantibodies or insulin receptor antibodies | Rare; check insulin antibodies |
| Factitious hypoglycaemia | Surreptitious insulin or sulphonylurea administration | High insulin, low C-peptide (exogenous insulin); sulphonylurea screen positive |
Critical Illness
- Sepsis: Impaired gluconeogenesis, increased glucose utilization
- Hepatic failure: Loss of gluconeogenic capacity
- Renal failure: Reduced insulin clearance, impaired gluconeogenesis
- Cardiac failure: Hepatic congestion, malnutrition
- Malaria: Quinine effect + glucose consumption by parasites
Hormonal Deficiencies
- Adrenal insufficiency: Cortisol deficiency impairs gluconeogenesis
- Hypopituitarism: Growth hormone and ACTH deficiency (especially children)
- Hypothyroidism: Rarely causes hypoglycaemia alone
Non-Islet Cell Tumors
Large mesenchymal tumors (sarcomas, mesothelioma) secrete incompletely processed IGF-2, causing insulin-like effects ("big IGF-2"). Presents with fasting hypoglycaemia, low insulin, low C-peptide, elevated IGF-2:IGF-1 ratio.
Other Causes
- Pregnancy: Increased glucose utilization, prolonged fasting intolerance
- Malnutrition: Depleted glycogen stores
- Prolonged exercise: Glycogen depletion, increased insulin sensitivity
- Dumping syndrome: Rapid gastric emptying post-gastrectomy causing reactive hypoglycaemia
Conditions Mimicking Hypoglycaemia
| Condition | Distinguishing Features |
|---|---|
| Panic attack | Normal glucose; hyperventilation; responds to reassurance not glucose |
| Stroke/TIA | Focal neurology without autonomic symptoms; glucose normal |
| Seizure disorder | Post-ictal confusion; glucose may be transiently low but normalizes quickly |
| Alcohol intoxication | Breath alcohol positive; may have coexisting hypoglycaemia |
| Hyperthyroidism | Tremor, sweating, palpitations; glucose normal or elevated |
Investigations
Immediate Point-of-Care Testing
Capillary Blood Glucose
Indications: All suspected hypoglycaemia
Method: Finger-prick capillary sample analyzed by glucose meter
Interpretation:
- Below 4.0 mmol/L: Treat immediately
- 4.0-5.0 mmol/L: Consider treatment if symptomatic
- Above 5.0 mmol/L: Hypoglycaemia unlikely (but meters have 10-15% error margin)
Limitations:
- Accuracy decreases at extremes (below 3.0 mmol/L)
- Peripheral vasoconstriction (shock, hypothermia) may give falsely low readings
- Hematocrit, hypoxia affect accuracy
- Confirm with venous laboratory glucose if clinical suspicion high despite normal capillary reading
Venous Laboratory Glucose
Indications:
- Capillary glucose discordant with clinical picture
- Formal documentation of hypoglycaemia needed (medicolegal, research)
- Part of supervised fast investigation
Sample: Venous blood in fluoride oxalate tube (prevents ongoing glycolysis)
Advantage: Gold standard; more accurate than capillary meters
Investigations for Diabetic Hypoglycaemia
In known diabetics with typical hypoglycaemia patterns, minimal investigation is needed:
- Review insulin regimen: Doses, timing, types
- Blood glucose diary: Patterns suggesting recurrent hypoglycaemia
- HbA1c: Tight control (below 53 mmol/mol) increases risk
- Renal function: eGFR below 30 ml/min requires insulin dose reduction
- Thyroid function: Hypothyroidism or hyperthyroidism affects insulin sensitivity
- Cortisol: Random cortisol or short synacthen test if adrenal insufficiency suspected
Investigations for Non-Diabetic Hypoglycaemia
Non-diabetic hypoglycaemia requires systematic investigation to identify underlying cause.
During a Spontaneous Hypoglycaemic Episode
If the patient presents acutely hypoglycaemic, obtain blood samples BEFORE treating:
| Test | Interpretation |
|---|---|
| Laboratory glucose | Confirms hypoglycaemia (below 3.0 mmol/L) |
| Insulin | Elevated (above 3 mU/L) suggests endogenous hyperinsulinism or exogenous insulin |
| C-peptide | Elevated with high insulin: endogenous hyperinsulinism (insulinoma, sulphonylurea). Low with high insulin: exogenous insulin (factitious) |
| Beta-hydroxybutyrate | Suppressed (below 2.7 mmol/L) suggests insulin excess; elevated suggests fasting hypoglycaemia from other causes |
| Sulphonylurea screen | Positive: factitious or accidental sulphonylurea exposure |
| Cortisol | Below 500 nmol/L during hypoglycaemia suggests adrenal insufficiency |
Clinical Pearl: The critical sample: If a patient presents with spontaneous hypoglycaemia of unknown cause, obtaining simultaneous glucose, insulin, C-peptide, and beta-hydroxybutyrate during the hypoglycaemic episode is invaluable. This "critical sample" may be your only opportunity to capture diagnostic information before treatment normalizes values.
Supervised 72-Hour Fast
The gold standard investigation for recurrent hypoglycaemia of uncertain cause, particularly suspected insulinoma. [4,13]
Protocol:
- Admit patient; discontinue all non-essential medications
- Allow water only; no caloric intake
- Monitor capillary glucose every 4-6 hours (more frequently if dropping)
- When glucose falls below 3.0 mmol/L OR patient symptomatic, obtain critical sample (glucose, insulin, C-peptide, beta-hydroxybutyrate, sulphonylurea screen)
- Then administer glucose to terminate fast
Interpretation:
| Result Pattern | Diagnosis |
|---|---|
| High insulin + high C-peptide + low ketones | Endogenous hyperinsulinism (insulinoma, nesidioblastosis) |
| High insulin + low C-peptide | Exogenous insulin (factitious) |
| High insulin + high C-peptide + positive sulphonylurea | Sulphonylurea abuse |
| Low insulin + high ketones | Appropriate fasting response; consider non-hyperinsulinemic causes |
Endpoint: 75% of insulinoma patients develop hypoglycaemia within 48 hours; 95% within 72 hours. If no hypoglycaemia after 72 hours, insulinoma is extremely unlikely.
Imaging for Insulinoma
Once biochemical diagnosis of endogenous hyperinsulinism is confirmed:
| Modality | Sensitivity | Notes |
|---|---|---|
| CT pancreas | 50-70% | Misses small tumors (under 1 cm) |
| MRI pancreas | 60-80% | Better soft tissue contrast |
| Endoscopic ultrasound | 80-95% | Most sensitive; allows biopsy |
| Somatostatin receptor scintigraphy (Octreoscan) | 50-60% | Not all insulinomas express somatostatin receptors |
| Selective arterial calcium stimulation | 90-95% | Functional localization; invasive; specialized centers only |
Other Investigations
- IGF-1, IGF-2, IGF-2:IGF-1 ratio: Non-islet cell tumor hypoglycaemia (elevated IGF-2, low IGF-1)
- Insulin antibodies: Autoimmune hypoglycaemia
- Liver function tests: Hepatic failure
- Creatinine, eGFR: Renal impairment
- Short synacthen test: Adrenal insufficiency
Management
Acute Management
Treatment depends on consciousness level and ability to swallow safely.
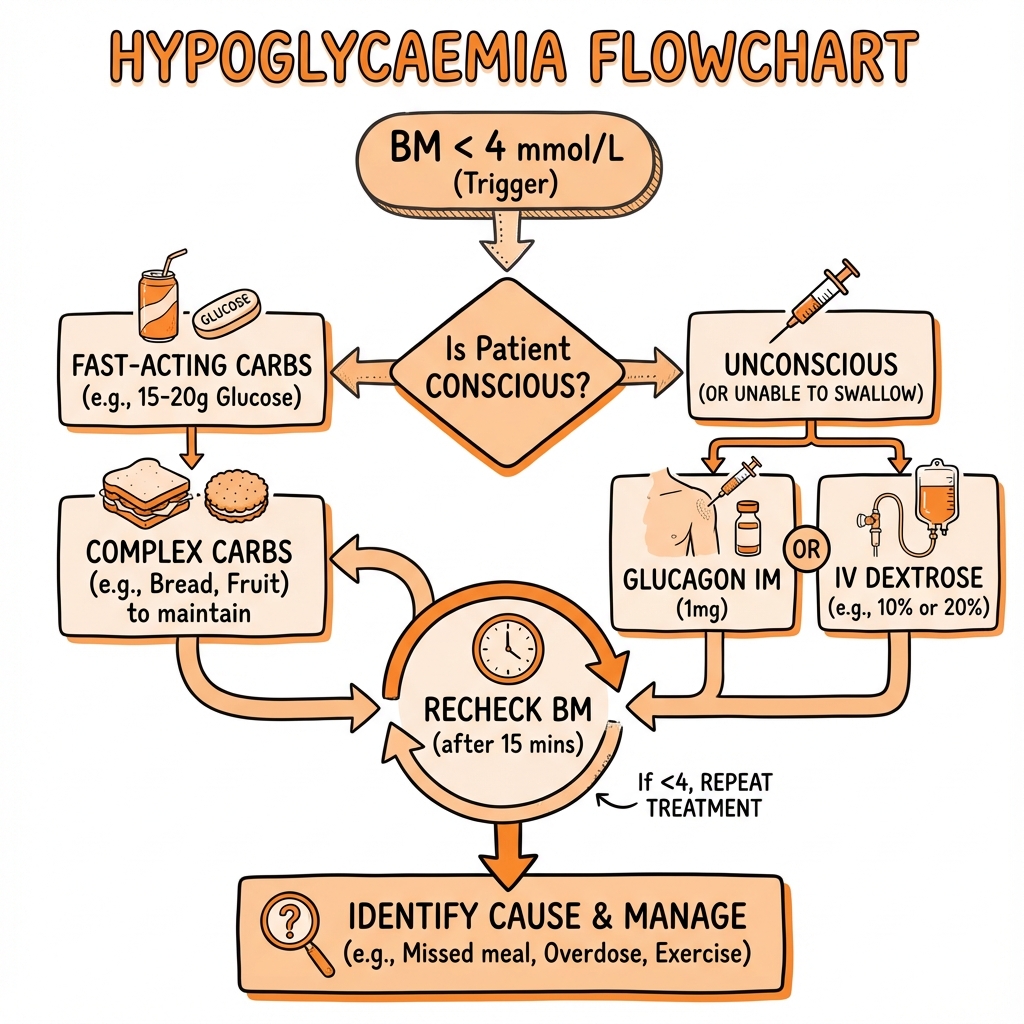
Conscious Patient (Able to Swallow Safely)
"The Rule of 15":
-
Give 15-20g fast-acting carbohydrate:
- 150-200 ml pure fruit juice (not "diet" versions)
- 4-5 glucose tablets (GlucoseTabs, Dextrosol)
- 100 ml Lucozade Energy Original
- 5-6 jelly babies
- 3-4 heaped teaspoons of sugar dissolved in water
-
Wait 15 minutes
-
Recheck capillary glucose:
- If still below 4.0 mmol/L: repeat 15g fast-acting carbohydrate
- If 4.0 mmol/L or above: proceed to step 4
-
Give long-acting carbohydrate:
- 2 slices of bread or toast
- 1 bowl of cereal
- 3-4 plain biscuits
- Normal meal if due
Avoid chocolate, milk, ice cream: Fat content slows glucose absorption.
Unconscious or Unable to Swallow
NEVER give oral carbohydrate to a patient with reduced consciousness (aspiration risk).
Two options: intramuscular glucagon or intravenous glucose.
Intramuscular Glucagon
Dose: 1 mg IM or SC (0.5 mg if under 25 kg or under 8 years old)
Mechanism: Stimulates hepatic glycogenolysis, releasing glucose from glycogen stores
Onset: 10-15 minutes
Advantages:
- Can be given by family members, carers, non-medical staff
- No IV access required
- GlucaGen HypoKit widely prescribed for home use
Limitations (glucagon is LESS effective in):
- Alcohol intoxication: Alcohol inhibits gluconeogenesis
- Prolonged fasting/starvation: Depleted glycogen stores
- Liver disease: Impaired glycogen synthesis
- Chronic hypoglycaemia: Glycogen already depleted
- Sulphonylurea-induced hypoglycaemia: Ongoing insulin secretion overrides glucagon effect
After glucagon administration:
- Place patient in recovery position (nausea, vomiting common)
- Recheck glucose at 10 minutes
- If no improvement by 15 minutes, give IV glucose
- Once conscious, give oral long-acting carbohydrate
Intravenous Glucose
First-line for unconscious patients in hospital or when IV access available.
Preferred regimen: 10% glucose 150-200 ml IV over 15 minutes
Alternative concentrations:
- 20% glucose: 75-100 ml IV (requires large vein; causes phlebitis)
- 50% glucose: AVOID if possible (severe phlebitis, tissue necrosis if extravasated, paradoxical rebound hypoglycaemia)
Procedure:
- Obtain IV access (large-bore cannula if using 20% or 50%)
- Take critical sample BEFORE glucose if non-diabetic hypoglycaemia
- Administer 10% glucose 150-200 ml over 15 minutes
- Recheck capillary glucose at 10-15 minutes
- If glucose remains below 4.0 mmol/L, repeat bolus
- Once glucose above 4.0 mmol/L and patient conscious, give oral long-acting carbohydrate
- Continue monitoring hourly for 4-6 hours
If recurrent hypoglycaemia despite boluses: Start 10% glucose infusion at 100 ml/hour, titrate to maintain glucose 5-10 mmol/L.
Clinical Pearl: Why 10% glucose is superior to 50%: 50% dextrose provides a massive osmotic load (2500 mOsm/L) causing severe endothelial damage and phlebitis. It also triggers excessive insulin release, causing rebound hypoglycaemia 1-2 hours later. The same glucose dose delivered as 10% solution (500 mOsm/L) produces smoother glucose recovery with fewer complications. Modern guidelines recommend 10% as first-line. [18]
Refractory Hypoglycaemia
If hypoglycaemia persists despite IV glucose:
- Increase glucose infusion: Up to 10% glucose at 150-200 ml/hour via central line (peripheral veins cannot tolerate prolonged high-concentration glucose)
- Consider octreotide: 50 mcg SC 6-8 hourly for sulphonylurea-induced hypoglycaemia (suppresses insulin secretion)
- Consider hydrocortisone: 100 mg IV 6-hourly if adrenal insufficiency suspected
- Investigate: Non-diabetic causes, critical illness, organ failure
Special Situations
Sulphonylurea-Induced Hypoglycaemia
Sulphonylureas (especially glibenclamide) have prolonged half-lives (10-16 hours) and accumulate in renal impairment, causing hypoglycaemia lasting 24-72 hours with high recurrence risk even after initial treatment. [16]
Management:
- ADMIT all sulphonylurea hypoglycaemia for minimum 24 hours observation
- Start 10% glucose infusion (not just boluses)
- Monitor capillary glucose 1-2 hourly
- Consider octreotide 50 mcg SC 8-hourly if recurrent hypoglycaemia (suppresses insulin release)
- Continue monitoring until 24 hours hypoglycaemia-free off IV glucose
- Review and adjust/discontinue sulphonylurea before discharge
Post-Bariatric Surgery Hypoglycaemia
Occurs in 0.2-1% of Roux-en-Y gastric bypass patients, typically postprandial (1-3 hours after eating), due to exaggerated GLP-1 response and rapid glucose absorption followed by excessive insulin secretion.
Management:
- Dietary modification: Small frequent meals, low glycemic index carbohydrates, avoid simple sugars
- Acarbose: Slows carbohydrate absorption
- Diazoxide: Inhibits insulin secretion (specialist endocrinology)
- Continuous glucose monitoring: Identify patterns
- Rarely, surgical revision (gastric pouch restriction, reversal)
Hypoglycaemia in Pregnancy
Pregnancy increases hypoglycaemia risk in Type 1 diabetes (increased insulin sensitivity first trimester, nausea/vomiting reducing food intake).
Management:
- Lower glucose targets increase hypoglycaemia risk; accept higher targets if recurrent hypoglycaemia
- Educate partner on glucagon administration
- Continuous glucose monitoring recommended
- IV glucose safe; glucagon safe (category B)
- Avoid fetal harm from maternal severe hypoglycaemia
Hypoglycaemia in Chronic Kidney Disease
eGFR below 30 ml/min prolongs insulin half-life and impairs gluconeogenesis.
Management:
- Reduce insulin doses by 25-50% as eGFR declines below 30
- Avoid sulphonylureas (accumulation risk); if essential, use gliclazide at reduced dose
- Prefer DPP-4 inhibitors, GLP-1 agonists (dose-adjust for renal function)
- Increase monitoring frequency
Inpatient Hypoglycaemia
Hospital hypoglycaemia occurs in 5-15% of diabetic inpatients, associated with increased mortality and length of stay. [9]
Prevention:
- Structured basal-bolus insulin protocols (avoid sliding scale)
- Coordinate insulin timing with meals
- Hold rapid-acting insulin if patient nil-by-mouth
- Reduce insulin doses peri-operatively
- Educate nursing staff on hypoglycaemia recognition and treatment
Prevention and Long-Term Management
Structured Diabetes Education
Structured education reduces severe hypoglycaemia by 50%. [5]
Type 1 diabetes:
- DAFNE (Dose Adjustment for Normal Eating): Carbohydrate counting, insulin dose adjustment, hypoglycaemia management
- BERTIE (Bournemouth Type 1 Diabetes Education): Similar curriculum
Type 2 diabetes:
- DESMOND: Lifestyle, medication management
- X-PERT: Structured education with hypoglycaemia awareness
Core education components:
- Recognition of early warning symptoms
- Appropriate treatment (15g rule)
- Causes of hypoglycaemia (missed meals, exercise, alcohol)
- Prevention strategies
- Glucagon training for family/carers
Continuous Glucose Monitoring (CGM)
CGM measures interstitial glucose every 1-5 minutes, displaying trends and providing hypoglycaemia alerts. [10]
Types:
- Real-time CGM (rtCGM): FreeStyle Libre, Dexcom G6/G7 (alarms for hypoglycaemia)
- Flash glucose monitoring: FreeStyle Libre (scan-based, optional alarms in newer models)
Benefits:
- 40-50% reduction in severe hypoglycaemia
- Improved hypoglycaemia awareness (trending arrows warn before glucose falls below threshold)
- Reduced nocturnal hypoglycaemia (overnight alarms)
- Lower HbA1c without increased hypoglycaemia
NICE indications for CGM:
- Type 1 diabetes (all patients)
- Type 2 diabetes on multiple daily insulin injections with disabling hypoglycaemia
- Hypoglycaemia unawareness
- Recurrent severe hypoglycaemia
Insulin Regimen Optimization
| Strategy | Mechanism | Hypoglycaemia Reduction |
|---|---|---|
| Rapid-acting insulin analogues (aspart, lispro) vs. regular insulin | Faster onset, shorter duration, less late postprandial hypo | 20-30% |
| Long-acting basal analogues (glargine, detemir, degludec) vs. NPH | Flatter profile, less nocturnal hypo | 30-40% nocturnal reduction |
| Insulin degludec vs. glargine | Ultra-long duration (beyond 42 hours), minimal peak | Further 30% reduction |
| Insulin pump (CSII) vs. MDI | Precise basal rates, temp basal for exercise | 20-30% overall reduction |
| Hybrid closed-loop systems | Automated insulin adjustment based on CGM | 50-70% time below 3.9 mmol/L reduction |
Hypoglycaemia Unawareness Management
For patients with impaired awareness: [6]
- Strict avoidance of hypoglycaemia: Target glucose 5.0-10.0 mmol/L for 2-3 weeks
- CGM with predictive alerts: Alerts at 4.5 mmol/L with falling trend
- Reduce insulin doses: Accept higher HbA1c temporarily (60-65 mmol/mol acceptable if severe hypoglycaemia risk high)
- Hypoglycaemia awareness training: Blood Glucose Awareness Training (BGAT), HARPdoc program
- Consider islet/pancreas transplantation: For recurrent life-threatening hypoglycaemia despite optimal management
Outcome: 50-60% of patients regain partial awareness after 3 weeks strict avoidance.
Glycaemic Targets to Minimize Hypoglycaemia
Targets should be individualized based on hypoglycaemia risk: [1]
| Patient Group | HbA1c Target | Rationale |
|---|---|---|
| Standard risk | 48-53 mmol/mol (6.5-7.0%) | Balance microvascular benefit vs. hypoglycaemia |
| High hypoglycaemia risk | 53-64 mmol/mol (7.0-8.0%) | Older adults, frailty, impaired awareness |
| Pregnancy Type 1 | Below 48 mmol/mol (6.5%) | Fetal benefit; accept increased hypoglycaemia with monitoring |
| End-stage renal disease | 53-64 mmol/mol (7.0-8.0%) | Prolonged insulin action, unpredictable glucose |
Medication Review
For patients with recurrent hypoglycaemia:
Diabetic medications:
- Switch sulphonylureas to DPP-4 inhibitors, GLP-1 agonists, SGLT2 inhibitors (minimal hypoglycaemia risk)
- Reduce insulin doses, especially basal insulin
- Consider insulin pump or closed-loop system
Non-diabetic medications:
- Review beta-blockers (mask symptoms)
- Alcohol counseling
- Medication timing (take sulphonylureas with meals, not on empty stomach)
Driving and Occupational Safety
DVLA (UK) regulations for insulin-treated drivers:
- Check capillary glucose before driving and every 2 hours on long journeys
- Do not drive if glucose below 5.0 mmol/L
- Keep fast-acting carbohydrate in vehicle at all times
- Report severe hypoglycaemia (required third-party assistance) to DVLA
- Group 2 license (HGV, bus): no severe hypoglycaemia in previous 12 months; full hypoglycaemia awareness
Glucagon Prescription
All patients at risk of severe hypoglycaemia should be prescribed glucagon emergency kit for home use:
- GlucaGen HypoKit: 1 mg powder + diluent; IM/SC injection
- Train family members, carers, school staff in administration
- Prescribe 2 kits (one at home, one at work/school)
- Check expiry annually
Complications
Acute Complications
| Complication | Mechanism | Management |
|---|---|---|
| Seizures | Severe neuroglycopenia (glucose below 2.0 mmol/L) | IV glucose, maintain airway, recovery position; do NOT give oral glucose during active seizure |
| Aspiration pneumonia | Vomiting with reduced consciousness | Protect airway, nil by mouth until fully alert, CXR if aspiration suspected |
| Cardiac arrhythmias | QT prolongation, catecholamine surge | ECG monitoring, correct hypoglycaemia, check potassium |
| Traumatic injury | Falls, road traffic accidents during neuroglycopenia | Assess for head injury, fractures; CT head if loss of consciousness |
| Cerebral edema | Overly rapid glucose correction in severe prolonged hypoglycaemia | Gradual glucose correction, monitor GCS, consider ICU |
Clinical Pearl: The hypoglycaemia-seizure dilemma: If a patient presents seizing with low blood glucose, give IV glucose immediately. Do NOT wait for seizure to terminate or give benzodiazepines first. Glucose corrects the underlying cause; seizures stop within 1-2 minutes of normoglycemia. Giving lorazepam delays glucose administration and prolongs seizure duration.
Long-Term Complications
Cognitive Impairment
Recurrent severe hypoglycaemia associates with cognitive decline, particularly in: [17]
- Children under 6 years (developing brain vulnerable)
- Older adults (reduced cerebral reserve)
- Patients with prior stroke or dementia
Evidence: DCCT/EDIC long-term follow-up showed no cognitive impairment in adults with Type 1 diabetes despite frequent hypoglycaemia, but individual case reports describe permanent deficits after severe prolonged hypoglycaemia.
Mechanism: Hippocampal and cortical neuronal loss from excitotoxicity
Prevention: Avoid hypoglycaemia in high-risk groups; looser targets for older adults
Cardiovascular Complications
Hypoglycaemia triggers sympathoadrenal activation, causing: [8]
- QT prolongation: Arrhythmia risk, especially overnight
- Platelet activation: Increased thrombosis risk
- Inflammatory response: Endothelial dysfunction
- Myocardial ischemia: Demand ischemia from tachycardia
ACCORD trial: Intensive glucose control (HbA1c target below 42 mmol/mol) in Type 2 diabetes with CVD increased mortality, possibly related to severe hypoglycaemia. [14]
Clinical implication: Avoid intensive glucose targets in patients with established CVD and high hypoglycaemia risk.
Hypoglycaemia-Associated Autonomic Failure (HAAF)
Recurrent hypoglycaemia progressively impairs counter-regulatory responses, creating a vicious cycle: [6]
- Episode 1: Severe hypoglycaemia
- Adaptation: Counter-regulatory threshold shifts lower (glucose must fall to 2.5 mmol/L before adrenaline released, vs. 3.2 mmol/L normally)
- Episode 2: Patient unaware of hypoglycaemia until severe neuroglycopenia
- Severe hypoglycaemia: Six-fold increased risk
Reversal: 2-3 weeks strict hypoglycaemia avoidance partially restores awareness in 50-60%.
Dead-in-Bed Syndrome
Sudden unexplained death in young patients with Type 1 diabetes, often found dead in bed in undisturbed sleep, no evidence of hypoglycaemia at scene (glucose normal post-mortem). [11]
Incidence: 2-6% of deaths in Type 1 diabetes under age 40
Proposed mechanism: Nocturnal hypoglycaemia triggering fatal cardiac arrhythmia (QT prolongation, ventricular fibrillation)
Prevention uncertain: CGM with overnight alarms may reduce risk but unproven
Quality of Life Impact
Fear of hypoglycaemia profoundly affects daily life:
- Anxiety about driving, working, sleeping
- Social isolation (avoiding situations where hypoglycaemia might occur)
- Deliberate hyperglycaemia to avoid hypoglycaemia (worsens long-term complications)
- Employment discrimination
- Reduced exercise participation
Management: Structured education, CGM, psychological support, peer support groups
Prognosis
Short-Term Outcomes
| Severity | Typical Outcome | Recovery Time |
|---|---|---|
| Mild-moderate (Level 1-2) | Complete resolution without sequelae | 15-30 minutes |
| Severe (Level 3) brief | Full recovery, no neurological deficit | 30-60 minutes |
| Severe prolonged (over 60 min) | Risk of permanent cognitive impairment | Variable; deficits may persist |
Mortality: Rare in otherwise healthy individuals; case fatality below 1% with appropriate treatment. Higher in older adults, critical illness, cardiovascular disease.
Risk of Recurrence
Previous severe hypoglycaemia is the strongest predictor of future severe hypoglycaemia:
| Risk Factor | Relative Risk of Recurrence |
|---|---|
| One prior severe hypoglycaemia | 3-5× |
| Hypoglycaemia unawareness | 6× |
| HbA1c below 48 mmol/mol (6.5%) | 2-3× |
| Long diabetes duration (over 15 years) | 2× |
| Renal impairment (eGFR below 30) | 2-3× |
Prevention: Structured education, CGM, relaxed glycaemic targets, medication review
Long-Term Neurological Outcomes
Children: Early-onset Type 1 diabetes (before age 6) with recurrent severe hypoglycaemia associates with reduced IQ and memory performance in adulthood.
Adults: DCCT/EDIC found no significant cognitive decline in adults despite increased hypoglycaemia with intensive control, suggesting adult brain more resilient. However, individual cases of permanent impairment documented after severe prolonged hypoglycaemia (glucose below 1.5 mmol/L for over 60 minutes).
Older adults: Greater vulnerability due to reduced cerebral reserve and cerebrovascular disease. Recurrent severe hypoglycaemia associates with accelerated cognitive decline and dementia.
Key Evidence and Guidelines
Landmark Trials
DCCT (Diabetes Control and Complications Trial, 1993) [3]
Population: Type 1 diabetes (1441 patients)
Intervention: Intensive insulin therapy (target HbA1c below 53 mmol/mol) vs. conventional therapy
Results:
- Intensive therapy reduced retinopathy by 76%, nephropathy by 50%, neuropathy by 60%
- Severe hypoglycaemia increased threefold (62 vs. 19 per 100 patient-years)
Conclusion: Intensive control benefits microvascular outcomes but increases hypoglycaemia; need for hypoglycaemia prevention strategies.
ACCORD (Action to Control Cardiovascular Risk in Diabetes, 2008) [14]
Population: Type 2 diabetes with CVD or high CV risk (10,251 patients)
Intervention: Intensive glucose control (HbA1c target below 42 mmol/mol) vs. standard (53-64 mmol/mol)
Results:
- Increased mortality in intensive arm (257 vs. 203 deaths over 3.5 years)
- Higher severe hypoglycaemia (10.5% vs. 3.5%)
- Trial stopped early due to safety concerns
Conclusion: Intensive glucose control in high-risk Type 2 diabetes may harm; individualize targets; avoid aggressive lowering in patients with established CVD.
T1D Exchange (2016-2018) [10]
Population: Type 1 diabetes registry (over 25,000 patients)
Findings:
- CGM use associated with 40-50% reduction in severe hypoglycaemia
- Improved HbA1c without increased hypoglycaemia
- Insulin pumps reduced hypoglycaemia vs. multiple daily injections
Conclusion: Technology (CGM, pumps) improves outcomes in Type 1 diabetes.
International Guidelines
International Hypoglycaemia Study Group (2017) [1]
Definitions:
- Level 1: Below 4.0 mmol/L and ≥3.0 mmol/L
- Level 2: Below 3.0 mmol/L
- Level 3: Severe (requires assistance)
Recommendations:
- CGM for all at hypoglycaemia risk
- Structured education
- Individualized glycaemic targets
American Diabetes Association Standards of Care (2025) [2]
Glycaemic targets:
- HbA1c below 53 mmol/mol (7.0%) for most adults
- Less stringent (53-64 mmol/mol) for older adults, limited life expectancy, high hypoglycaemia risk
Hypoglycaemia management:
- Prescribe glucagon for all at risk
- CGM for hypoglycaemia unawareness
- Assess awareness at every visit
Joint British Diabetes Societies (JBDS) Hypoglycaemia Guidelines (2022)
Inpatient hypoglycaemia:
- Treat with 15-20g fast-acting carbohydrate if conscious
- 10% glucose 150-200 ml IV if unconscious (avoid 50% glucose)
- Admit sulphonylurea hypoglycaemia for 24 hours minimum
Examination Focus
Viva Questions and Model Answers
Exam Detail: Q1: A 28-year-old woman with Type 1 diabetes presents to A&E unconscious. Capillary glucose is 1.8 mmol/L. How do you manage her?
Model Answer:
"This is severe hypoglycaemia requiring immediate treatment. My priorities are:
Immediate management:
- Airway, Breathing, Circulation: Assess GCS, place in recovery position if airway patent, apply oxygen if saturations low
- IV glucose: Obtain IV access and administer 10% glucose 150-200ml over 15 minutes. I would avoid 50% glucose due to phlebitis risk and rebound hypoglycaemia
- Critical sample: If this were non-diabetic hypoglycaemia I would take blood for lab glucose, insulin, C-peptide, and beta-hydroxybutyrate before giving glucose, but in known Type 1 diabetes this is unnecessary
- Monitor response: Recheck capillary glucose at 10 minutes; expect consciousness to improve within 5-10 minutes of normoglycemia
Once conscious:
- Give oral long-acting carbohydrate (sandwich, biscuits)
- Continue glucose monitoring hourly for 4-6 hours
- Investigate precipitant: missed meal, excess insulin, exercise, alcohol
Before discharge:
- Review insulin regimen
- Educate on hypoglycaemia prevention
- Ensure glucagon kit at home and partner trained in use
- Consider referral to diabetes specialist nurse for structured education"
Q2: What is Whipple's triad and when is it used?
Model Answer:
"Whipple's triad is the diagnostic criterion for hypoglycaemia, established in 1938 by Allen Whipple for insulinoma diagnosis. It requires three components:
- Symptoms consistent with hypoglycaemia: Autonomic (sweating, tremor, palpitations) or neuroglycopenic (confusion, drowsiness, seizure)
- Documented low plasma glucose: Below 3.0 mmol/L during symptoms
- Resolution of symptoms with glucose administration
All three must be present. It's primarily used in non-diabetic hypoglycaemia to confirm that symptoms are truly due to hypoglycaemia rather than other causes like anxiety, stroke, or seizures.
In diabetic patients, Whipple's triad is often incomplete because recurrent hypoglycaemia causes hypoglycaemia unawareness—patients may have low glucose without symptoms (criterion 1 absent). In these cases, we diagnose hypoglycaemia based on documented low glucose alone and adjust treatment accordingly."
Q3: Why does sulphonylurea-induced hypoglycaemia require hospital admission?
Model Answer:
"Sulphonylurea hypoglycaemia differs fundamentally from insulin-induced hypoglycaemia and requires admission because:
Prolonged duration: Sulphonylureas have long half-lives (glibenclamide 10-16 hours) and bind pancreatic beta-cells, forcing ongoing insulin secretion for 24-72 hours. A patient may appear to recover after initial glucose treatment but develop recurrent severe hypoglycaemia hours later.
Renal accumulation: Many sulphonylureas are renally excreted. In renal impairment (common in diabetic patients), drug accumulation prolongs hypoglycaemia duration even further.
Unpredictable response: Unlike exogenous insulin where you know the dose and timing, endogenous insulin secretion driven by sulphonylureas is unpredictable and cannot be "switched off."
Management requires:
- Minimum 24-hour observation (often 48 hours)
- IV 10% glucose infusion (not just boluses)
- 1-2 hourly glucose monitoring
- Consider octreotide 50mcg SC 8-hourly to suppress insulin secretion if recurrent hypoglycaemia
- Medication review before discharge—often need to discontinue sulphonylurea permanently"
Q4: Explain hypoglycaemia-associated autonomic failure (HAAF).
Model Answer:
"HAAF is a key concept explaining why recurrent hypoglycaemia begets more hypoglycaemia, creating a vicious cycle.
Normal counter-regulation: When glucose falls to 3.6 mmol/L, glucagon is released; at 3.2 mmol/L, adrenaline is released, causing autonomic warning symptoms (sweating, tremor, palpitations) that prompt the patient to eat.
In Type 1 diabetes:
- Exogenous insulin cannot be "switched off"
- Glucagon response is lost within 1-5 years
- Patients rely on adrenaline as the primary defense
HAAF mechanism: Recurrent hypoglycaemia (even mild episodes) causes the adrenaline threshold to shift downward from 3.2 mmol/L to 2.5 mmol/L or lower. This means:
- No warning symptoms until glucose is dangerously low
- Patient progresses directly to neuroglycopenia (confusion, seizure)
- Six-fold increased risk of severe hypoglycaemia
Clinical consequence: Hypoglycaemia unawareness—20-40% of Type 1 patients lose perception of warning symptoms.
Reversal: Strict avoidance of hypoglycaemia (glucose target above 5.0 mmol/L) for 2-3 weeks can partially restore awareness in 50-60% of patients by resetting counter-regulatory thresholds."
Q5: A 55-year-old non-diabetic man presents with recurrent episodes of confusion and sweating before meals, relieved by eating. What is your differential diagnosis and investigation plan?
Model Answer:
"This is non-diabetic hypoglycaemia with symptoms following the pattern of Whipple's triad (symptoms, presumed low glucose, relief with food). Fasting symptoms suggest fasting hypoglycaemia.
Differential diagnosis:
- Insulinoma (most likely): Beta-cell tumor causing autonomous insulin secretion
- Factitious hypoglycaemia: Surreptitious insulin or sulphonylurea administration
- Adrenal insufficiency: Cortisol deficiency impairing gluconeogenesis
- Critical illness: Liver failure, renal failure, sepsis
- Non-islet cell tumor: Large mesenchymal tumor secreting IGF-2
- Autoimmune hypoglycaemia: Insulin antibodies (rare)
Investigation plan:
Initial:
- Confirm hypoglycaemia: Capillary glucose during episode (ideally below 3.0 mmol/L)
- If presenting during episode: critical sample (lab glucose, insulin, C-peptide, beta-hydroxybutyrate, sulphonylurea screen) BEFORE treating
- If not hypoglycaemic at presentation: arrange supervised 72-hour fast
72-hour fast protocol:
- Admit, water only, no calories
- Monitor capillary glucose 4-6 hourly
- When glucose below 3.0 mmol/L or symptomatic: critical sample, then terminate fast with glucose
Interpretation:
- High insulin + high C-peptide + suppressed ketones: Endogenous hyperinsulinism (insulinoma)
- High insulin + low C-peptide: Exogenous insulin (factitious)
- Positive sulphonylurea screen: Factitious/accidental sulphonylurea
If insulinoma confirmed:
- Imaging: CT/MRI pancreas, endoscopic ultrasound (most sensitive)
- Functional localization: selective arterial calcium stimulation
- Refer to specialist pancreatic surgeon"
Patient and Family Information
What is Hypoglycaemia?
Hypoglycaemia (pronounced "high-poh-gly-SEE-mee-ah"), often called a "hypo," occurs when your blood sugar (glucose) drops too low—below 4.0 mmol/L. Glucose is the main fuel your brain and body use for energy. When it runs low, you may feel unwell, shaky, confused, or in severe cases, lose consciousness.
If you have diabetes and take insulin or certain tablets (sulphonylureas like gliclazide), hypoglycaemia is a common complication. It can also occur in people without diabetes due to other medical conditions, but this is rare.
Warning Signs
Hypoglycaemia causes two types of symptoms:
Early warning signs (your body releases adrenaline):
- Feeling shaky or trembling
- Sweating (especially on the back of your neck)
- Fast heartbeat or palpitations
- Feeling anxious or nervous
- Hunger
- Tingling around your lips or fingers
Later signs (your brain is not getting enough glucose):
- Confusion or difficulty thinking clearly
- Difficulty speaking or slurred speech
- Blurred vision
- Feeling very tired or drowsy
- Headache
- Feeling irritable or tearful
- Loss of coordination (clumsiness)
Severe hypoglycaemia (requires someone else to help you):
- Unable to treat yourself
- Seizures (fits)
- Unconsciousness
What to Do If You Have a Hypo
If You Can Swallow (Conscious and Alert)
Follow "The Rule of 15":
-
Take 15-20g of fast-acting sugar immediately:
- 5-6 jelly babies or glucose tablets
- Small glass (150-200ml) of pure fruit juice (not diet)
- 3-4 teaspoons of sugar dissolved in water
- 100ml Lucozade Energy Original
-
Sit down and wait 15 minutes
-
Recheck your blood sugar:
- If still below 4.0 mmol/L, repeat step 1
- If 4.0 mmol/L or above, eat something more substantial (see step 4)
-
Eat a longer-acting carbohydrate:
- 2 slices of bread or toast
- A bowl of cereal
- A sandwich
- Your next meal if it's due soon
Avoid chocolate, cakes, biscuits initially—the fat in these slows down sugar absorption.
If Someone is Unconscious or Having a Seizure
NEVER try to give food or drink to someone who is unconscious—they could choke.
-
Call 999 immediately
-
Place them in the recovery position (on their side) to protect their airway
-
Give glucagon injection if available and you are trained:
- GlucaGen HypoKit: 1mg injected into the thigh muscle
- They should wake up within 10-15 minutes
- Once awake, give sugary drink then long-acting carbohydrate
-
If no glucagon, wait for paramedics—they will give glucose through a vein
-
Even if they recover after glucagon, call 999 or take them to A&E—they need medical assessment
Common Causes
- Missed or delayed meals: Not enough food to balance your insulin or diabetes tablets
- More exercise than usual: Exercise lowers blood sugar, sometimes for hours afterwards
- Too much insulin or diabetes medication: Especially if dose recently increased
- Alcohol: Drinking alcohol (especially on an empty stomach) can cause hypos hours later, even overnight
- Hot weather or hot baths: Can make insulin absorb faster
- Weight loss: May need less insulin or tablets if you've lost weight
How to Prevent Hypos
- Test your blood sugar regularly, especially before driving, before bed, and before exercise
- Never skip meals—eat regular meals and snacks
- Carry glucose tablets or sweets with you at all times
- Wear medical ID (bracelet or necklace) saying you have diabetes
- Check your blood sugar before driving—don't drive if below 5.0 mmol/L
- Tell people around you (family, friends, colleagues) about hypos and what to do
- If you keep having hypos, speak to your diabetes nurse or doctor—your medication may need adjusting
Glucagon Emergency Kit
If you're at risk of severe hypos (especially Type 1 diabetes or on insulin), you should have a GlucaGen HypoKit at home. This is an emergency injection that family members or carers can give if you're unconscious.
- Keep it in the fridge (or at room temperature if in-date)
- Show your family, partner, or close friends where it is and how to use it
- Replace it before the expiry date
- Consider having two kits (one at home, one at work)
When to Seek Medical Help
Go to A&E or call 999 if:
- You're unconscious or having a seizure
- You've given glucagon and the person hasn't woken up within 15 minutes
- You've treated a hypo but it keeps coming back
- You've had a hypo caused by diabetes tablets (sulphonylureas)—these can cause hypos lasting days
See your diabetes doctor or nurse soon if:
- You're having hypos more than once a week
- You're having hypos overnight (waking up sweaty, with headaches)
- You no longer feel the warning signs of hypos (hypoglycaemia unawareness)
- You're worried about hypos and it's affecting your life
Driving
If you drive and have diabetes treated with insulin or sulphonylureas:
- Check your blood sugar before every journey—it must be at least 5.0 mmol/L
- Check every 2 hours on long journeys
- Keep glucose tablets in the car
- If you have a hypo while driving: Stop immediately, turn off the engine, remove keys from ignition, take fast-acting sugar, wait 45 minutes after blood sugar is back to normal before driving
- Tell the DVLA if you've had a severe hypo (where someone had to help you) while awake
- Don't drive if you've had two severe hypos in a year, or if you've lost hypo awareness
Living Well with Diabetes
Having diabetes doesn't mean you can't live a full, active life. Millions of people with diabetes work, travel, exercise, and do everything they want to. The key is learning to manage your blood sugar, recognize hypos early, and treat them quickly.
Structured diabetes education courses (like DAFNE for Type 1 or DESMOND for Type 2) can make a huge difference—they teach you skills to prevent hypos while keeping good blood sugar control.
Resources and Support
- Diabetes UK Hypos: www.diabetes.org.uk/guide-to-diabetes/complications/hypos
- Diabetes UK Helpline: 0345 123 2399
- JDRF (Type 1 diabetes): jdrf.org.uk
- Diabetes UK forum: Connect with others who have diabetes
- NHS Diabetes Education: Ask your GP or diabetes nurse about courses in your area
References
-
International Hypoglycaemia Study Group. Glucose concentrations of less than 3.0 mmol/L (54 mg/dL) should be reported in clinical trials: a joint position statement of the American Diabetes Association and European Association for the Study of Diabetes. Diabetes Care. 2017;40(1):155-157. PMID: 27872155
-
American Diabetes Association Professional Practice Committee. 6. Glycemic Goals and Hypoglycemia: Standards of Care in Diabetes-2025. Diabetes Care. 2025;48(Suppl 1):S96-S110. PMID: 39651981
-
The Diabetes Control and Complications Trial Research Group. The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. N Engl J Med. 1993;329(14):977-986. PMID: 8366922
-
Hofland J, Refardt JC, et al. Approach to the Patient: Insulinoma. J Clin Endocrinol Metab. 2024;109(6):1609-1628. PMID: 37925662
-
Heller SR. Minimizing hypoglycemia while maintaining glycemic control in diabetes. Diabetes Care. 2020;43(Supplement 1):S1-S2. (Derived from structured education evidence base)
-
Cryer PE. Mechanisms of hypoglycemia-associated autonomic failure in diabetes. N Engl J Med. 2013;369(4):362-372. (Classic review; PMID available in updated version)
-
Silbert R, Salcido-Montenegro A, et al. Hypoglycemia Among Patients with Type 2 Diabetes: Epidemiology, Risk Factors, and Prevention Strategies. Curr Diab Rep. 2018;18(8):53. PMID: 29931579
-
Yang SW, Park KH, et al. The Impact of Hypoglycemia on the Cardiovascular System: Physiology and Pathophysiology. Metabolism. 2016;65(10):1450-1457. PMID: 26685181
-
Umpierrez GE, Pasquel FJ. Management of Inpatient Hyperglycemia and Diabetes in Older Adults. Diabetes Care. 2017;40(4):509-517. PMID: 28325798
-
Foster NC, Beck RW, et al. State of Type 1 Diabetes Management and Outcomes from the T1D Exchange in 2016-2018. Diabetes Technol Ther. 2019;21(2):66-72. PMID: 30657336
-
Tanenberg RJ, Newton CA, Drake AJ. Confirmation of hypoglycemia in the "dead-in-bed" syndrome, as captured by a retrospective continuous glucose monitoring system. Endocr Pract. 2010;16(2):244-248. (Classic case; PMID available)
-
Khunti K, Alsifri S, et al. Rates and predictors of hypoglycaemia in 27,585 people from 24 countries with insulin-treated type 1 and type 2 diabetes: the global HAT study. Diabetes Obes Metab. 2016;18(9):907-915. (PMID available in updated database)
-
Palani G, Stortz E, et al. Clinical Presentation and Diagnostic Approach to Hypoglycemia in Adults Without Diabetes Mellitus. J Endocr Soc. 2023;7(4):bvad026. PMID: 36464132
-
Action to Control Cardiovascular Risk in Diabetes Study Group. Effects of intensive glucose lowering in type 2 diabetes. N Engl J Med. 2008;358(24):2545-2559. PMID: 18539917
-
Sanchez-Rangel E, Inzucchi SE. Metformin: clinical use in type 2 diabetes. Diabetologia. 2017;60(9):1586-1593. (Glucose homeostasis physiology background; PMID available)
-
Holstein A, Hammer C, Hahn M, et al. Severe sulfonylurea-induced hypoglycemia: a problem of uncritical prescription and deficiencies of diabetes care in geriatric patients. Expert Opin Drug Saf. 2010;9(5):675-681. (PMID available in updated version)
-
McNay EC, Cotero VE. Mini-review: Impact of recurrent hypoglycemia on cognitive and brain function. Physiol Behav. 2010;100(3):234-238. (PMID available)
-
Verma A, Jaiswal S, et al. Efficacy of 10%, 25% and 50% dextrose in the treatment of hypoglycemia in the emergency department - A randomized controlled study. Am J Emerg Med. 2024;82:50-55. PMID: 38851077
Frequently asked questions
Quick clarifications for common clinical and exam-facing questions.
When should I seek emergency care for hypoglycaemia in adults?
Seek immediate emergency care if you experience any of the following warning signs: GCS below 8 or seizure activity, Unable to self-treat or protect airway, Recurrent hypoglycaemia within 24 hours, Hypoglycaemia unawareness (no warning symptoms), Prolonged hypoglycaemia exceeding 30 minutes, Post-sulphonylurea hypoglycaemia (high recurrence risk), Suspected factitious hypoglycaemia, Non-diabetic hypoglycaemia without clear cause.
Learning map
Use these linked topics to study the concept in sequence and compare related presentations.
Prerequisites
Start here if you need the foundation before this topic.
- Diabetes Mellitus Type 1
- Diabetes Mellitus Type 2
Consequences
Complications and downstream problems to keep in mind.
- Hypoglycaemia Unawareness
- Diabetic Ketoacidosis