Hyponatraemia in Adults
The clinical significance of hyponatraemia extends far beyond a laboratory abnormality. Acute symptomatic hyponatraemia ... MRCP exam preparation.
What matters first
The clinical significance of hyponatraemia extends far beyond a laboratory abnormality. Acute symptomatic hyponatraemia ... MRCP exam preparation.
Check the red flags, emergency triggers, and escalation points before using the deep-dive material.
6 Jan 2026
Generated educational material; verify before clinical use.
Visible references section
See the concept before reading it
Study the key anatomy, imaging, and decision pathways as full teaching plates.
Clinical board
A visual summary of the highest-yield teaching signals on this page.
Exam focus
Current exam surfaces linked to this topic.
- MRCP
Linked comparisons
Differentials and adjacent topics worth opening next.
- Hypernatraemia
- Cerebral Salt Wasting
Content status and exam context
This page is AI-generated educational content. It may contain errors or omissions and is not a substitute for current guidelines, local protocols, senior clinical judgement, or professional medical advice.
MedVellum does not claim an individual clinician reviewer, board certification, or professional credential for this page unless a future version names a real, verifiable reviewer.
Clinical explanation and evidence
1. Clinical Overview
Hyponatraemia is defined as a serum sodium concentration $less than 135$ mmol/L and represents the most common electrolyte disorder encountered in clinical practice, affecting 15-30% of hospitalized patients. [1,2] It reflects a relative excess of total body water compared to total body sodium, rather than a primary deficiency of sodium itself—a conceptual distinction critical to understanding its pathophysiology and management.
The clinical significance of hyponatraemia extends far beyond a laboratory abnormality. Acute symptomatic hyponatraemia constitutes a medical emergency capable of precipitating life-threatening cerebral oedema, seizures, respiratory arrest, and brainstem herniation within hours. [3] Conversely, even mild chronic hyponatraemia (130-134 mmol/L), long dismissed as clinically insignificant, is now recognized as an independent risk factor for gait instability, attention deficits, osteoporosis, fractures, and increased all-cause mortality. [4,7]
Management of hyponatraemia is notoriously complex and fraught with iatrogenic risk. The condition requires navigating between two potentially catastrophic outcomes: failure to adequately treat acute severe hyponatraemia risks cerebral herniation and death, while overly rapid correction of chronic hyponatraemia can precipitate Osmotic Demyelination Syndrome (ODS)—formerly termed central pontine myelinolysis—a devastating and often irreversible demyelinating condition that can result in locked-in syndrome. [3,9]
Clinical Pearl: The "Goldilocks" Electrolyte: Hyponatraemia management epitomizes the principle of therapeutic balance. A serum sodium that is too low endangers the patient through cerebral oedema and herniation, but raising it too rapidly is equally dangerous, risking permanent neurological devastation from ODS. The skilled clinician must navigate this narrow corridor with precision, guided by the dual beacons of symptom severity and chronicity.
Fundamental Clinical Classifications
The approach to hyponatraemia requires systematic classification along four independent axes:
-
Severity by Serum Sodium Concentration:
- Mild: 130-135 mmol/L
- Moderate: 125-129 mmol/L
- Severe: less than 125 mmol/L
- Profound: less than 120 mmol/L
-
Temporal Classification:
- Acute: less than 48 hours duration
- Chronic: > 48 hours duration
- Unknown duration: Treated as chronic to minimize ODS risk
-
Tonicity Classification:
- Hypotonic (True hyponatraemia): Low serum osmolality
- Isotonic (Pseudohyponatraemia): Normal serum osmolality
- Hypertonic (Translocational): Elevated serum osmolality
-
Volume Status Classification:
- Hypovolaemic: Total body sodium and water decreased (sodium deficit > water deficit)
- Euvolaemic: Total body sodium normal, water increased
- Hypervolaemic: Total body sodium and water increased (water excess > sodium excess)
Symptom-Based Risk Stratification
Beyond biochemical classification, clinical symptoms drive urgency of intervention:
- Severe symptoms (requiring emergency intervention): Vomiting, cardiorespiratory distress, abnormal somnolence, seizures, coma, Glasgow Coma Scale ≤8
- Moderate symptoms: Nausea without vomiting, confusion, headache
- Mild/absent symptoms: Asymptomatic or subtle findings (gait instability, concentration difficulties)
Exam Detail: The 48-Hour Threshold: The distinction between acute and chronic hyponatraemia is not arbitrary. Within 48 hours of onset, cerebral astrocytes begin adaptive mechanisms to combat cellular swelling by extruding organic osmolytes (glutamate, taurine, myo-inositol, creatine) and inorganic ions (potassium, chloride). This "brain volume regulation" reduces intracellular osmolality, limiting water influx and cerebral oedema. However, once these osmolytes are extruded, rapid normalization of serum sodium creates a hypertonic extracellular environment that draws water OUT of brain cells, causing cellular shrinkage, disruption of the blood-brain barrier, and demyelination—the pathophysiological basis of ODS.
2. Epidemiology
Hyponatraemia represents the most prevalent electrolyte disturbance across all healthcare settings, serving not merely as a biochemical finding but as a powerful prognostic marker for underlying disease severity and mortality risk.
| Setting/Population | Prevalence | Key Findings | Evidence Source |
|---|---|---|---|
| General Hospital Inpatients | 15-30% | Most common electrolyte disorder; prevalence increases with illness severity | [1,2] |
| Intensive Care Units | 30-40% | Associated with independent mortality risk (OR 1.5-2.0); marker of critical illness | [4] |
| Emergency Department | 10-15% | Often presenting feature of acute illness; requires urgent evaluation | [5] |
| Community/Ambulatory | 4-7% | Prevalence increases with age (> 65 years: 7-11%); often drug-induced | [5] |
| Nursing Homes/Elderly Care | 8-15% | Multifactorial: polypharmacy, reduced GFR, decreased thirst | [5] |
| Post-operative Patients | 4-6% | Hypotonic fluid administration, pain-induced ADH secretion | [6] |
| Marathon Runners | 10-15% | Exercise-associated hyponatraemia (EAH); fluid overload + non-osmotic ADH | [18] |
Mortality and Morbidity Impact
Hyponatraemia is not a benign finding. Meta-analytical evidence demonstrates:
- Mild chronic hyponatraemia (130-135 mmol/L): Hazard ratio (HR) for all-cause mortality 1.15-1.25 [6]
- Moderate hyponatraemia (125-129 mmol/L): HR for mortality 1.3-1.5 [6]
- Severe hyponatraemia (less than 125 mmol/L): HR for mortality 1.5-2.5 [6]
- Hospital-acquired hyponatraemia: Independently associated with increased length of stay (median +2.3 days), ICU admission, and in-hospital mortality [2]
Importantly, the association between hyponatraemia and mortality persists even after adjustment for comorbidities, suggesting that hyponatraemia itself—or the underlying dysregulation of water homeostasis it reflects—contributes to adverse outcomes rather than serving merely as a marker of illness severity.
Age and Sex Differences
- Women: 25-30% higher risk of hyponatraemia than men due to smaller body size, lower muscle mass (lower total body solute), and hormonal effects on ADH sensitivity [5]
- Elderly (> 65 years): Risk factors include reduced GFR, impaired urinary dilution, decreased thirst perception, polypharmacy (especially thiazides, SSRIs), and "tea and toast" diet (low solute intake) [5]
- Young women: Disproportionately affected by acute symptomatic hyponatraemia and ODS, possibly due to hormonal effects on cerebral adaptation [3]
Drug-Induced Hyponatraemia
Medications account for up to 30% of cases in ambulatory settings:
Highest Risk Medications:
- Thiazide diuretics: 10-30% incidence (higher than loop diuretics due to preserved medullary concentration gradient)
- SSRIs: 0.5-32% incidence (highly variable; highest in elderly)
- Carbamazepine/Oxcarbazepine: 5-40% incidence
- Proton pump inhibitors: 1-3% incidence (mechanism unclear)
- Desmopressin: Dose-dependent; high risk in elderly
- Cyclophosphamide: 2-20% (direct ADH release)
- Vincristine: 2-10%
- MDMA (Ecstasy): 10-15% of emergency presentations
Clinical Pearl: The "Asymptomatic" Myth: The term "asymptomatic hyponatraemia" is increasingly recognized as a misnomer. Prospective studies using objective gait assessment and neurocognitive testing demonstrate that even mild chronic hyponatraemia (130-134 mmol/L) is associated with measurable impairments in gait stability (increased sway area by 56%), attention (increased reaction time), and working memory. [7] In elderly patients, this translates to a 67% increased risk of falls and 2.1-fold increased fracture risk. The absence of overt neurological symptoms does not equate to an absence of harm.
3. Aetiology & Pathophysiology
Hyponatraemia is fundamentally a disorder of water balance rather than sodium balance. Understanding this principle is essential: in the vast majority of cases, hyponatraemia results from impaired renal free water excretion rather than sodium loss. The kidneys' ability to excrete free water is primarily regulated by the antidiuretic hormone (ADH, also known as arginine vasopressin or AVP) system and the thirst mechanism.
Normal Physiology of Water Homeostasis
The ADH-Aquaporin System
-
Osmotic Sensing: Specialized neurons in the anterior hypothalamus (organum vasculosum laminae terminalis, OVLT) act as osmoreceptors, detecting changes in plasma osmolality as small as 1-2%.
-
ADH Secretion: When plasma osmolality rises above the threshold (~280-295 mOsm/kg), the supraoptic and paraventricular nuclei of the hypothalamus secrete ADH into the posterior pituitary circulation.
-
Renal Action: ADH binds to V2 receptors on the basolateral membrane of principal cells in the renal collecting duct, activating adenylyl cyclase and increasing intracellular cAMP. This triggers protein kinase A (PKA)-mediated phosphorylation of aquaporin-2 (AQP2) water channels, promoting their insertion into the apical membrane.
-
Water Reabsorption: AQP2 channels permit passive water movement from the tubular lumen to the hypertonic medullary interstitium, concentrating the urine and conserving body water.
-
Negative Feedback: Water retention lowers plasma osmolality, suppressing ADH secretion when osmolality falls below ~280 mOsm/kg, permitting urinary dilution.
Non-Osmotic ADH Regulation
Critical to understanding hyponatraemia is recognizing that ADH secretion is governed not only by osmolality but also by non-osmotic stimuli that can override osmotic suppression:
- Volume/pressure: Baroreceptors in the carotid sinus, aortic arch, and left atrium detect effective arterial blood volume (EABV). A 10-15% reduction in EABV triggers exponential ADH release to preserve circulating volume, even at the cost of hypo-osmolality—the body "sacrifices" tonicity to maintain perfusion.
- Nausea: Potent stimulus for ADH release (explains hyponatraemia in hyperemesis, chemotherapy)
- Pain and stress: Activate ADH secretion
- Hypoxia and hypercapnia: Stimulate ADH release
- Upright posture: Mild ADH stimulus
Pathophysiological Classification
1. Hypovolaemic Hyponatraemia
Principle: Total body sodium and water are both decreased, but sodium deficit exceeds water deficit.
Mechanism: Volume depletion activates baroreceptors, triggering non-osmotic ADH secretion to preserve blood pressure. Simultaneously, the renin-angiotensin-aldosterone system (RAAS) is activated, promoting proximal sodium reabsorption and reducing distal delivery (impairing dilution capacity). Reduced effective circulating volume also stimulates thirst.
Causes:
Renal Sodium Loss (Urine Na > 20 mmol/L):
- Thiazide diuretics: Inhibit Na-Cl cotransporter in distal convoluted tubule; preserve medullary concentration gradient (unlike loop diuretics), permitting continued ADH-mediated water retention. Risk highest in first 2 weeks, elderly, women.
- Loop diuretics: Lower risk than thiazides (impair medullary gradient)
- Primary adrenal insufficiency (Addison's disease): Aldosterone deficiency → renal sodium wasting; cortisol deficiency → increased ADH secretion and sensitivity
- Salt-wasting nephropathies: Interstitial nephritis, medullary cystic disease, partial urinary tract obstruction
- Cerebral salt wasting (CSW): Renal sodium loss following CNS insult (subarachnoid haemorrhage, neurosurgery); mechanism involves brain natriuretic peptide (BNP)
- Osmotic diuresis: Hyperglycaemia, mannitol, urea (post-relief of obstruction)
Extra-Renal Sodium Loss (Urine Na less than 20 mmol/L):
- Gastrointestinal: Vomiting, diarrhoea, nasogastric drainage, enterocutaneous fistula
- Third spacing: Pancreatitis, bowel obstruction, peritonitis, burns, rhabdomyolysis
- Cutaneous: Excessive sweating (especially in cystic fibrosis)
Exam Detail: Thiazide vs. Loop Diuretics—Why the Difference?
Loop diuretics (furosemide, bumetanide) inhibit the Na-K-2Cl cotransporter in the thick ascending limb of the loop of Henle, disrupting the countercurrent multiplication mechanism that generates the medullary osmotic gradient. Without this gradient, ADH cannot effectively concentrate urine, limiting water retention and reducing hyponatraemia risk.
Thiazides, conversely, act distally (distal convoluted tubule), leaving the medullary gradient intact. ADH-mediated water reabsorption in the collecting duct can therefore proceed unimpaired, while thiazide-induced sodium loss triggers compensatory ADH secretion. Additionally, thiazides reduce GFR and enhance proximal tubular reabsorption, decreasing distal delivery and impairing dilution capacity. The combination renders thiazides the most common drug cause of severe hyponatraemia.
2. Euvolaemic Hyponatraemia
Principle: Total body water is increased while total body sodium remains essentially normal.
Mechanism: Excessive water retention due to either:
- Inappropriate ADH secretion (SIADH) relative to osmolality, or
- Water intake exceeding the kidney's maximum dilution capacity (primary polydipsia, beer potomania)
Causes:
Syndrome of Inappropriate Antidiuresis (SIAD/SIADH):
SIADH is a diagnosis of exclusion defined by:
- Hypotonic hyponatraemia (serum osmolality less than 275 mOsm/kg)
- Urine osmolality > 100 mOsm/kg (inappropriately concentrated)
- Urine sodium > 20 mmol/L (typically > 40 mmol/L) on normal diet
- Clinical euvolaemia (no oedema, hypovolaemia, or ascites)
- Normal thyroid, adrenal, and renal function
- No recent diuretic use
SIADH Aetiologies:
| Category | Examples | Notes |
|---|---|---|
| Malignancy | Small cell lung cancer (SCLC), pancreatic cancer, lymphoma, thymoma | Ectopic ADH secretion; SCLC in 10-15% of cases |
| Pulmonary | Pneumonia (bacterial, viral, TB), aspergillosis, empyema, pneumothorax, positive pressure ventilation | Mechanism unclear; inflammation-mediated? |
| CNS | Meningitis, encephalitis, abscess, head trauma, subdural haematoma, stroke, Guillain-Barré | Disruption of hypothalamic-pituitary axis |
| Drugs | See comprehensive table below | Most common reversible cause |
| Post-operative | Any major surgery | Pain, nausea, opioids, hypotonic fluids |
| Idiopathic | Diagnosis after exclusion | Common in elderly; may reflect age-related ADH dysregulation |
Comprehensive Drug Causes of SIADH:
| Drug Class | Specific Agents | Mechanism |
|---|---|---|
| Antidepressants | SSRIs (fluoxetine, sertraline, citalopram, escitalopram), SNRIs (venlafaxine, duloxetine), TCAs (amitriptyline), MAOIs | Serotonergic stimulation of ADH release; increased renal sensitivity |
| Anticonvulsants | Carbamazepine, oxcarbazepine, valproate, lamotrigine | Direct ADH release; enhanced renal tubular response |
| Antipsychotics | Typical (haloperidol, chlorpromazine) and atypical (risperidone, quetiapine, olanzapine) | Central ADH stimulation; dopamine antagonism |
| Chemotherapy | Cyclophosphamide, vincristine, vinblastine, cisplatin, ifosfamide, methotrexate | Direct ADH release; renal tubular damage |
| Opioids | Morphine, tramadol, fentanyl | Nausea + direct ADH secretion |
| Antiretrovirals | Didanosine, lamivudine | Mechanism unclear |
| PPIs | Omeprazole, esomeprazole, pantoprazole | Mechanism unknown; rare but reported |
| Ecstasy (MDMA) | Recreational | ADH release + excessive hypotonic fluid intake |
| Others | Desmopressin (DDAVP), oxytocin, bromocriptine, amiodarone, NSAIDs | Direct ADH agonism or secretion |
Exam Detail: Molecular Mechanism of SIADH:
In SIADH, circulating AVP (ADH) levels are elevated or "inappropriate" relative to the low serum osmolality. AVP binds to the V2 receptor, a G-protein coupled receptor (Gs) on the basolateral membrane of principal cells in the collecting duct. Receptor activation stimulates adenylyl cyclase, increasing intracellular cAMP. cAMP activates protein kinase A (PKA), which phosphorylates aquaporin-2 (AQP2) water channels residing in intracellular vesicles. Phosphorylation triggers vesicle trafficking to and fusion with the apical membrane, inserting AQP2 channels. Water moves passively down its osmotic gradient from the hypotonic tubular lumen into the hypertonic medullary interstitium (via basolateral AQP3 and AQP4). Chronic ADH exposure also increases AQP2 gene transcription, further amplifying water reabsorption. The net effect: concentrated urine (Uosm > 500 mOsm/kg), free water retention, and progressive dilutional hyponatraemia.
Endocrine Causes:
- Severe hypothyroidism: Reduced cardiac output and GFR → non-osmotic ADH; impaired free water clearance
- Glucocorticoid deficiency: Cortisol normally inhibits ADH secretion and enhances free water excretion; deficiency (secondary adrenal insufficiency, isolated ACTH deficiency) → SIADH-like picture. Distinguishing feature: typically normal aldosterone (no hyperkalemia if isolated glucocorticoid deficiency).
Primary Polydipsia:
- Psychogenic polydipsia: Compulsive water drinking (> 10-15 L/day), overwhelming renal dilution capacity (maximum ~12-15 L/day). ADH is appropriately suppressed; urine is maximally dilute (less than 100 mOsm/kg).
- Dipsogenic polydipsia: Abnormal thirst mechanism (hypothalamic lesion); patients drink excessively despite normal osmolality
Beer Potomania / "Tea and Toast" Syndrome:
- Mechanism: Kidneys require solute (electrolytes, urea) to excrete free water. Minimum urine osmolality is ~50 mOsm/kg. If daily solute intake is very low (e.g., 250 mOsm from beer or tea/toast diet), maximum urine volume = 250/50 = 5 L/day. Drinking > 5 L/day → positive water balance → hyponatraemia.
- Clinical setting: Chronic alcoholism (beer provides calories but minimal solute), elderly with poor intake, fad "tea cleanse" diets
- Urine osmolality: Often less than 100 mOsm/kg (appropriately dilute), but water excretion capacity is limited by solute, not ADH
- Danger: Reintroducing normal diet (solute) suddenly increases free water clearance capacity → rapid water diuresis → risk of overcorrection and ODS
Reset Osmostat:
- Mechanism: ADH regulation is intact but the osmotic threshold for ADH secretion is "reset" to a lower value (e.g., 265-270 mOsm/kg instead of 280-290). Patient regulates serum sodium around this new set point.
- Clinical features: Stable, chronic mild hyponatraemia (typically 125-132 mmol/L); patient can concentrate AND dilute urine appropriately relative to the new set point
- Associations: Pregnancy (physiological), quadriplegia, chronic malnutrition, advanced age
- Management implication: Attempting correction is futile; sodium will drift back to reset level
3. Hypervolaemic Hyponatraemia
Principle: Total body sodium AND water are both increased, but water excess exceeds sodium excess. The patient appears volume overloaded (oedema, ascites) but has reduced effective arterial blood volume (EABV).
Mechanism: In heart failure, cirrhosis, and nephrotic syndrome, reduced cardiac output or severe hypoalbuminaemia causes "underfilling" of the arterial circulation despite total body volume overload. Baroreceptors detect reduced EABV and activate compensatory mechanisms:
- RAAS activation: Angiotensin II stimulates proximal tubular sodium reabsorption, reducing distal delivery
- Non-osmotic ADH secretion: Water retention in collecting duct
- Sympathetic activation: Further promotes renal sodium retention
- Reduced GFR: Diminishes free water clearance
Net result: Avid sodium AND water retention, but water retention exceeds sodium retention → dilutional hyponatraemia.
Causes:
- Congestive heart failure: Severity of hyponatraemia correlates with NYHA class and mortality; marker of neurohormonal activation
- Liver cirrhosis with ascites: Portal hypertension → splanchnic vasodilation → reduced EABV; severe hyponatraemia (Na less than 125) predicts poor prognosis, hepatorenal syndrome
- Nephrotic syndrome: Severe hypoalbuminaemia (less than 20 g/L) → reduced oncotic pressure → EABV reduction
- Advanced chronic kidney disease: GFR less than 30 ml/min; impaired dilution capacity
Distinguishing Urine Sodium:
- Heart failure, cirrhosis: Urine Na typically less than 20 mmol/L (avid renal sodium retention)
- Renal failure: Urine Na > 20 mmol/L (kidneys cannot reabsorb sodium effectively despite signals)
4. Non-Hypotonic Hyponatraemia
These are NOT true hyponatraemia (intracellular osmolality is normal); serum sodium is low due to measurement artifact or osmotic shifts.
Pseudohyponatraemia (Isotonic):
- Mechanism: Severe hyperlipidaemia (triglycerides > 1500 mg/dL) or hyperproteinaemia (multiple myeloma, IVIg infusion) displaces plasma water volume. Indirect ion-selective electrodes (older laboratory methods) measure sodium concentration in whole plasma, yielding falsely low values. Sodium concentration in the plasma water phase is actually normal.
- Serum osmolality: Normal (275-295 mOsm/kg)
- Modern labs: Direct ion-selective electrodes (used in most contemporary analyzers) measure sodium activity directly and are NOT affected; pseudohyponatraemia is now rare
Translocational Hyponatraemia (Hypertonic):
- Mechanism: Accumulation of effective osmoles in the ECF (glucose, mannitol, contrast, glycerol) creates an osmotic gradient drawing water from ICF to ECF, diluting ECF sodium
- Serum osmolality: Elevated (> 295 mOsm/kg)
- Correction formula for hyperglycaemia:
- For every 5.5 mmol/L (100 mg/dL) rise in glucose above baseline, serum sodium falls by ~2.4 mmol/L
- Corrected Na = Measured Na + [2.4 × (Glucose - 5.5) / 5.5]
- "Alternative: Corrected Na = Measured Na + [1.6 × (Glucose - 5.5)]"
- Clinical relevance: Diabetic ketoacidosis, hyperosmolar hyperglycaemic state
- Treatment: Correcting hyperglycaemia will raise serum sodium; monitor to avoid overly rapid rise
4. Clinical Presentation
The clinical manifestations of hyponatraemia are predominantly neurological, reflecting osmotic shifts of water into brain cells. Symptom severity depends on:
- Absolute serum sodium level
- Rapidity of decline (acute vs. chronic)
- Patient-specific vulnerability (young women, elderly, chronic liver disease)
Acute Hyponatraemia (less than 48 Hours)
Rapid sodium decline overwhelms cerebral compensatory mechanisms. Water influx into astrocytes causes cytotoxic cerebral oedema, increased intracranial pressure, and risk of herniation.
Mild to Moderate (Na 125-134 mmol/L):
- Nausea (often the earliest symptom)
- Headache
- Malaise, fatigue
- Dizziness
- Confusion, disorientation
Severe (Na less than 125 mmol/L):
- Repeated vomiting
- Cardiorespiratory distress (Cheyne-Stokes breathing)
- Abnormal somnolence (Glasgow Coma Scale less than 13)
- Seizures (generalized tonic-clonic)
- Coma (GCS ≤8)
- Brainstem herniation:
- Decorticate/decerebrate posturing
- Fixed dilated pupils
- Respiratory arrest
- Death
High-Risk Populations for Acute Severe Symptoms:
- Post-operative patients (hypotonic IV fluids + ADH surge from pain/nausea)
- Marathon runners (exercise-associated hyponatraemia)
- Polydipsia (psychiatric patients, MDMA users)
- Post-TURP syndrome (glycine absorption)
- Oxytocin infusion during labour
Clinical Pearl: Sex Differences in Acute Hyponatraemia: Premenopausal women are disproportionately represented among cases of severe symptomatic acute hyponatraemia and resultant mortality. Proposed mechanisms include oestrogen-mediated inhibition of the cerebral Na-K-ATPase (impairing cellular ion extrusion), smaller brain volumes (less "room to swell"), and higher ADH levels during the luteal phase. Women also appear more susceptible to respiratory arrest prior to seizure activity, possibly due to hormonal effects on brainstem respiratory centres.
Chronic Hyponatraemia (> 48 Hours)
Over 48 hours, cerebral astrocytes extrude organic osmolytes (glutamate, taurine, myo-inositol, glutamine) and inorganic ions (K+, Cl-), reducing intracellular osmolality to match the hypotonic ECF. This brain volume regulation limits cerebral oedema, rendering chronic hyponatraemia better tolerated—but NOT benign.
Mild (Na 130-134 mmol/L):
- Often labelled "asymptomatic," but objective testing reveals:
- Gait instability (increased postural sway by 56% on balance platform testing) [7]
- Attention deficits (prolonged reaction times, reduced attention span)
- Subtle cognitive impairment (working memory, executive function)
- Consequence: 67% increased risk of falls; 2.1-fold increased fracture risk [7]
- Osteoporosis: Chronic hyponatraemia associated with increased osteoclast activity and decreased bone mineral density
Moderate (Na 125-129 mmol/L):
- Lethargy, fatigue
- Nausea without vomiting
- Anorexia
- "Brain fog," difficulty concentrating
- Muscle cramps
- Gait disturbance (observable)
Severe (Na less than 125 mmol/L):
- Confusion, disorientation
- Profound lethargy, somnolence
- Obtundation
- Seizures (less common than in acute, but possible if acute-on-chronic decline)
Acute-on-Chronic Hyponatraemia
Patients with chronic mild hyponatraemia who experience a rapid further decline (e.g., addition of a thiazide, diarrhoea, SIADH trigger) may present with severe symptoms despite the brain's prior adaptation. This scenario is particularly dangerous: the patient is symptomatic (favouring rapid correction) but has already extruded osmolytes (favouring slow correction to avoid ODS).
Exam Detail: Pathophysiology of Cerebral Adaptation and ODS:
In acute hyponatraemia, water influx into astrocytes triggers rapid compensatory mechanisms: activation of volume-regulated anion channels (VRACs) and extrusion of KCl within minutes to hours. Subsequently, over 24-48 hours, organic osmolytes (glutamate, taurine, myo-inositol) are lost via membrane transporters and metabolized. By 48 hours, brain water content nearly normalizes despite persistent hyponatraemia.
However, once osmolytes are lost, the brain becomes vulnerable to rapid correction. If serum sodium rises quickly (> 10 mmol/L per 24h), the ECF becomes acutely hypertonic relative to the osmolyte-depleted intracellular compartment. Water is drawn OUT of cells, causing:
- Cellular dehydration and shrinkage
- Disruption of the blood-brain barrier (BBB)
- Vascular congestion and microhaemorrhage
- Complement activation and microglial inflammation
- Oligodendrocyte apoptosis and demyelination
The pons is disproportionately affected due to its dense, interwoven white matter tracts and limited vascular supply. Extrapontine myelinolysis (basal ganglia, thalamus, cerebellum) may occur simultaneously. Symptoms typically emerge 2-6 days post-correction (the "biphasic" pattern): initial neurological improvement followed by devastating deterioration.
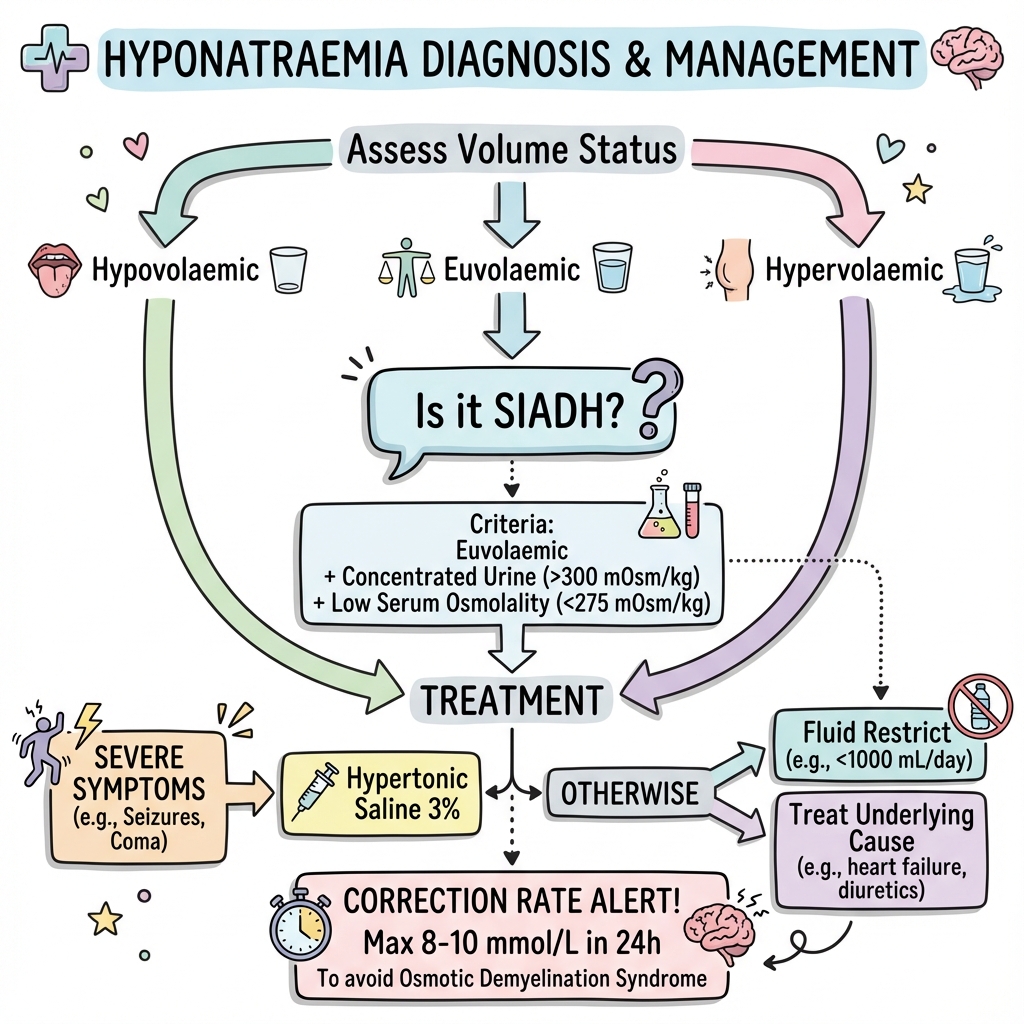
5. Differential Diagnosis & Volume Status Assessment
The diagnostic approach to hyponatraemia is algorithmic, with volume status assessment serving as the critical branch point. However, volume status determination is notoriously difficult and often inaccurate even among experienced clinicians.
Step-by-Step Diagnostic Algorithm
Step 1: Confirm True Hypotonic Hyponatraemia
Measure Serum Osmolality:
- Low (less than 275 mOsm/kg): TRUE hyponatraemia → Proceed to Step 2
- Normal (275-295 mOsm/kg): Pseudohyponatraemia
- "Check: Serum lipids (triglycerides > 1500 mg/dL), serum protein (total protein > 100 g/L), recent IVIg"
- "Action: No treatment required; measure sodium using direct ISE if available"
- High (> 295 mOsm/kg): Translocational (hypertonic) hyponatraemia
- "Check: Glucose, mannitol, glycerol, contrast agents"
- Calculate corrected sodium; treat underlying hyperosmolality
Step 2: Assess ADH Activity
Measure Urine Osmolality (on random or spot sample):
-
Maximally dilute (less than 100 mOsm/kg):
- "Interpretation: ADH is appropriately suppressed; kidneys are excreting free water normally"
- "Diagnosis: Primary polydipsia or beer potomania/low solute intake"
- "Distinguishing features:"
- Primary polydipsia: Urine sodium variable; history of excessive fluid intake
- Beer potomania: History of alcohol, poor nutrition; patient will develop aquaresis (water diuresis) upon reintroduction of normal diet → ODS risk
-
Inappropriately concentrated (> 100 mOsm/kg):
- "Interpretation: ADH is present (either appropriately due to volume depletion/EABV reduction, or inappropriately as in SIADH)"
- "Action: Proceed to Step 3"
Clinical Pearl: The Urine Osmolality Decision Point: A urine osmolality > 100 mOsm/kg in the setting of hypotonic hyponatraemia indicates that ADH is exerting an antidiuretic effect. The kidney SHOULD be producing dilute urine (less than 100 mOsm/kg) to excrete the excess water. Failure to do so implicates ADH excess or increased renal sensitivity to ADH. However, ADH may be appropriately elevated (volume depletion) or inappropriate (SIADH). Volume status assessment is therefore the next step.
Step 3: Assess Volume Status & Urine Sodium
Clinical Volume Assessment:
| Status | Clinical Signs | Caveats |
|---|---|---|
| Hypovolaemic | Dry mucous membranes, reduced skin turgor (> 2 sec tenting), orthostatic hypotension (BP drop > 20 mmHg systolic or > 10 mmHg diastolic upon standing), tachycardia, reduced JVP, decreased urine output | Sensitivity/specificity of individual signs is poor; orthostatic hypotension most reliable |
| Euvolaemic | Normal mucous membranes, normal skin turgor, normal BP and HR, no oedema, normal JVP | Diagnosis of exclusion; most difficult to determine clinically |
| Hypervolaemic | Peripheral pitting oedema (ankles, sacrum), ascites, elevated JVP (> 3 cm above sternal angle), pulmonary crackles, S3 gallop, pleural effusion | Clear signs; rarely diagnostic ambiguity |
Objective Volume Assessment (if available):
- Inferior vena cava diameter and collapsibility on ultrasound
- Central venous pressure (CVP) monitoring (if central line present)
- Response to fluid challenge (cautious in uncertain cases)
Measure Urine Sodium (spot urine; must be concurrent with serum):
| Volume Status | Urine Na | Interpretation | Differential Diagnoses |
|---|---|---|---|
| Hypovolaemic | less than 20 mmol/L | Appropriate renal sodium conservation; extra-renal losses | Vomiting, diarrhoea, third-spacing (pancreatitis, burns), remote diuretic use |
| Hypovolaemic | > 20 mmol/L | Inappropriate renal sodium wasting | Diuretics (current use), Addison's disease, cerebral salt wasting, salt-wasting nephropathy, bicarbonaturia (vomiting with alkalosis), ketonuria |
| Euvolaemic | > 20 mmol/L (usually > 40) | Renal sodium handling normal despite ADH; SIADH hallmark | SIADH, glucocorticoid deficiency, severe hypothyroidism, reset osmostat |
| Euvolaemic | less than 20 mmol/L | Low sodium intake or reset osmostat | Beer potomania, tea-and-toast, reset osmostat |
| Hypervolaemic | less than 20 mmol/L | Avid renal sodium retention due to reduced EABV | Heart failure, cirrhosis, nephrotic syndrome |
| Hypervolaemic | > 20 mmol/L | Kidney unable to retain sodium | Acute or chronic kidney disease |
Exam Detail: Urine Sodium Pitfalls:
-
Diuretic Use: A common confounder. Thiazides/loop diuretics increase urine sodium (> 20 mmol/L) even in the setting of volume depletion. If recent diuretic use, wait 24-48 hours (if clinically feasible) or measure urine chloride: less than 25 mmol/L suggests volume depletion (chloride is not increased by remote diuretic use).
-
Vomiting with Metabolic Alkalosis: Urine sodium may be elevated (> 20 mmol/L) due to obligatory excretion of sodium with bicarbonate (bicarbonaturia), despite volume depletion. Urine chloride less than 25 mmol/L confirms hypovolaemia.
-
Cerebral Salt Wasting (CSW) vs. SIADH: Both present with high urine sodium (> 40 mmol/L) and hyponatraemia post-neurosurgery or subarachnoid haemorrhage. Distinguishing features:
- CSW: Hypovolaemic (polyuria, negative fluid balance, weight loss, elevated haematocrit)
- SIADH: Euvolaemic (normal or positive fluid balance, stable weight, normal haematocrit)
- Treatment differs: CSW requires IV saline + oral salt; SIADH requires fluid restriction
- Diagnosis: Often requires careful monitoring of fluid balance and weights
Additional Diagnostic Investigations
| Test | Interpretation | Diagnostic Value |
|---|---|---|
| Serum Uric Acid | Low (less than 4 mg/dL or less than 0.24 mmol/L) in SIADH due to volume expansion and increased uric acid clearance; Elevated (> 5 mg/dL) in hypovolaemia due to increased proximal reabsorption | Supportive but not diagnostic |
| Serum Potassium | Hyperkalaemia suggests Addison's disease (combined mineralocorticoid and glucocorticoid deficiency) | High specificity for adrenal insufficiency |
| BUN/Creatinine Ratio | Elevated (> 20:1) in hypovolaemia (increased urea reabsorption); Normal in SIADH | Useful adjunct |
| 9am Cortisol | less than 100 nmol/L confirms adrenal insufficiency; > 500 nmol/L excludes; 100-500 requires ACTH stimulation test | Essential to exclude cortisol deficiency |
| TSH, Free T4 | Elevated TSH, low T4 in hypothyroidism | Severe hypothyroidism (myxoedema) can cause hyponatraemia |
| Lipids (Triglycerides) | > 1500 mg/dL can cause pseudohyponatraemia (if indirect ISE used) | Exclude pseudohyponatraemia |
| Chest X-ray | Small cell lung cancer, pneumonia, TB, lung abscess | Identify SIADH cause |
| CT Brain | Mass lesion, subdural haematoma, SAH, meningitis | CNS causes of SIADH or CSW |
SIADH: Diagnosis of Exclusion
SIADH can be definitively diagnosed only when the following criteria are met:
Essential Criteria (all required):
- Serum osmolality less than 275 mOsm/kg
- Urine osmolality > 100 mOsm/kg (inappropriate for low serum osmolality)
- Urine sodium > 20 mmol/L (typically > 40) on normal salt/protein diet
- Clinical euvolaemia
- Normal renal function (Creatinine less than 120 μmol/L)
- Normal thyroid function (TSH normal)
- Normal adrenal function (9am cortisol > 500 nmol/L or normal ACTH stim)
- No recent diuretic use (within 24-48 hours)
Supportive Findings:
- Low serum uric acid (less than 0.24 mmol/L)
- Low BUN (less than 7 mmol/L or less than 20 mg/dL)
- Fractional excretion of uric acid > 11%
- Failure to correct with 0.9% saline infusion
- Correction with fluid restriction
6. Investigations
Initial Laboratory Panel
Essential First-Line Tests (all patients):
- Serum sodium, potassium, chloride, bicarbonate
- Serum urea and creatinine
- Serum osmolality (measured, not calculated)
- Urine osmolality (concurrent with serum)
- Urine sodium (spot; concurrent with serum)
- Serum glucose
- Serum lipid profile (if pseudohyponatraemia suspected)
- Serum total protein (if pseudohyponatraemia suspected)
Calculated Serum Osmolality (for comparison):
\text{Calculated Osmolality (mOsm/kg)} = 2 \times \text{Na} + \text{Glucose} + \text{Urea}
(Units: Na in mmol/L, Glucose in mmol/L, Urea in mmol/L)
- Osmolar gap = Measured osmolality - Calculated osmolality
- Normal gap: less than 10 mOsm/kg
- Elevated gap suggests unmeasured osmoles (ethanol, methanol, ethylene glycol, mannitol)
Second-Line Tests (based on clinical suspicion):
| Indication | Tests |
|---|---|
| Suspected adrenal insufficiency | 9am cortisol, ACTH, ACTH stimulation test, plasma renin, aldosterone |
| Suspected hypothyroidism | TSH, Free T4 |
| Suspected SIADH | Serum uric acid, BUN, urine uric acid (fractional excretion), chest X-ray, CT chest/abdo |
| Post-neurosurgery/SAH | Daily fluid balance, weights, haematocrit (distinguish CSW vs. SIADH) |
| Suspected heart failure | BNP or NT-proBNP, echocardiography |
| Suspected cirrhosis | Liver function tests, albumin, coagulation, ultrasound liver |
Specialized Testing
Plasma ADH (Copeptin): Rarely required; research tool. Copeptin (C-terminal fragment of ADH precursor) is more stable and used as ADH surrogate. Interpretation complex.
Water Deprivation Test: Not used in hyponatraemia evaluation (used for diabetes insipidus workup).
Saline Infusion Test: Academic interest; rarely performed. Infusion of 2L 0.9% saline over 4 hours:
- SIADH: Serum sodium does NOT correct (urine remains concentrated)
- Hypovolaemia: Serum sodium corrects
7. Management
The management of hyponatraemia is guided by a symptom-first, aetiology-second approach. The presence of severe symptoms constitutes an emergency requiring immediate intervention regardless of chronicity or underlying cause. In the absence of severe symptoms, management is tailored to volume status, chronicity, and underlying aetiology, with overriding emphasis on avoiding overcorrection.
Emergency Management: Severe Symptomatic Hyponatraemia
Indications: ANY of the following:
- Seizures (active or recent)
- Coma (GCS ≤8)
- Cardiorespiratory distress (respiratory rate less than 8 or > 30, SaO2 less than 90%)
- Abnormal somnolence or obtundation (GCS 9-12)
Treatment Protocol (European Guidelines 2014): [8]
-
Hypertonic Saline Bolus Therapy:
- Administer 150 mL of 3% NaCl IV over 20 minutes
- Preparation: 3% NaCl = 30 g NaCl per litre = 513 mmol/L Na
- Expected effect: Raises serum sodium by ~2 mmol/L per bolus (in 70 kg adult)
-
Reassess at 20 Minutes:
- Check serum sodium
- Assess clinical response (improvement in consciousness, cessation of seizures)
-
Repeat if Needed:
- If symptoms persist: Give 2nd bolus of 150 mL 3% NaCl over 20 minutes
- Reassess at 20 minutes
- If symptoms STILL persist: Give 3rd bolus
-
Target:
- Raise serum sodium by 4-6 mmol/L in first 6 hours
- This modest rise is sufficient to reverse cerebral oedema and herniation risk
- Do NOT aim for full correction to normal
-
Cessation:
- STOP bolus therapy once symptoms improve OR target rise achieved
- Transition to maintenance management (see below based on aetiology)
-
Adjunctive Measures:
- Airway protection (intubation if GCS ≤8 or active seizure)
- Oxygen supplementation
- Benzodiazepines for seizures (lorazepam 4 mg IV or diazepam 10 mg IV)
- ICU/HDU admission for monitoring
Exam Detail: Why 150 mL Boluses? The Physiology:
The 150 mL bolus approach is designed to produce a predictable, controlled rise in serum sodium while minimizing calculation errors.
Calculation:
- 3% saline = 513 mmol Na per litre
- 150 mL = 0.15 L × 513 = 77 mmol Na delivered
- Total body water (TBW) = 0.6 × body weight (kg) = 0.6 × 70 kg = 42 L
- Expected rise in serum Na = 77 mmol / 42 L = 1.8 mmol/L
In practice, the rise is slightly higher (~2-2.5 mmol/L) due to concurrent urinary free water loss. Three boluses achieve 4-6 mmol/L rise. This approach is safer and more reproducible than continuous infusion calculations (e.g., "3 mL/kg over 1 hour"), which are error-prone and often result in inadvertent overcorrection.
Alternative for Smaller Patients or Children: 2 mL/kg of 3% saline over 10-20 minutes
Clinical Pearl: The 4-6 mmol/L Rule: A serum sodium rise of just 4-6 mmol/L is sufficient to reverse life-threatening cerebral oedema. For example, a patient with Na 110 mmol/L and seizures requires correction only to ~115 mmol/L acutely. Further correction to normal (135 mmol/L) is NOT necessary and introduces risk. The principle: "Enough to save the brain from swelling, not so much that you cause shrinking."
Non-Emergency Management Based on Volume Status
Once severe symptoms are addressed (or absent), management is dictated by volume status and aetiology.
A. Hypovolaemic Hyponatraemia
Principle: Correct volume depletion with isotonic saline. Volume resuscitation will suppress non-osmotic ADH secretion, permitting urinary free water excretion.
Treatment:
-
Isotonic Saline (0.9% NaCl):
- Initial rate: 1-2 litres over 4-6 hours, then reassess
- Monitor serum sodium every 2-4 hours
-
Danger: Rapid Overcorrection:
- When volume is restored, baroreceptors signal volume repletion
- ADH secretion abruptly ceases ("turning off the tap")
- Patient undergoes massive water diuresis (polyuria: 200-500 mL/hour of dilute urine)
- Serum sodium can rise precipitously (> 2 mmol/L per hour)
-
Prevention of Overcorrection:
- Monitor urine output: If > 100 mL/hour, increase monitoring frequency to every 1-2 hours
- Check serum sodium frequently: Every 1-2 hours during rapid diuresis
- If Na rising > 10 mmol/L in 24h or > 8 mmol/L in high-risk patients: Implement overcorrection rescue strategy (see below)
-
Specific Aetiologies:
- Addison's Disease: Requires glucocorticoid replacement (hydrocortisone 100 mg IV q6h initially) + saline. Correction of glucocorticoid deficiency will independently improve hyponatraemia.
- Cerebral Salt Wasting: Requires generous saline (3-4 L/day) + oral salt tablets (2-6 g/day). Differentiate from SIADH by volume status.
Clinical Pearl: Hypovolaemic Hyponatraemia is the Highest Risk for Overcorrection: Hypovolaemic hyponatraemia is paradoxically the most dangerous form to treat. Volume resuscitation rapidly corrects the stimulus for ADH secretion, unleashing a profound aquaresis. Patients can exceed safe correction limits (10 mmol/L per 24h) within 6-8 hours of starting saline. Close monitoring and proactive use of DDAVP (see overcorrection rescue) are often necessary.
B. Euvolaemic Hyponatraemia (SIADH)
First-Line: Fluid Restriction
- Rationale: Reducing water intake below obligate losses (urine + insensible) will create negative water balance, gradually raising serum sodium
- Target: Restrict total fluid intake to 500-1000 mL per day (must be less than 24h urine output + insensible losses [~500 mL/day])
- Efficacy: Succeeds in ~50-70% of cases; failure often due to non-adherence or urine osmolality > 500 mOsm/kg (kidneys too concentrated; not enough water excreted)
- Monitoring: Daily weights, serum sodium every 1-2 days initially
- Expected rate: 0.5-1 mmol/L per day rise (safe, slow correction)
Predictors of Fluid Restriction Failure:
- Urine osmolality > 500 mOsm/kg (highly concentrated; minimal free water excretion)
- Sum of urine (Na + K) > serum Na (indicates urine is effectively "hypertonic" relative to serum; infusion of saline would worsen hyponatraemia—the "desalination" phenomenon)
Exam Detail: The Desalination Phenomenon:
In severe SIADH, urine osmolality may exceed 600-800 mOsm/kg. If urine sodium + potassium concentration exceeds serum sodium (e.g., urine Na 80 + K 40 = 120 mmol/L vs. serum Na 118 mmol/L), then even isotonic (0.9%) saline is effectively hypotonic relative to the urine. Administering saline will result in net retention of free water (the sodium is excreted, but water is retained), paradoxically WORSENING hyponatraemia. This underscores why fluid restriction—not saline—is first-line for euvolaemic SIADH.
Second-Line Therapies (if fluid restriction fails or not tolerated):
-
Increased Solute Intake:
- Oral Salt Tablets: 2-6 g NaCl per day (1 g NaCl = 17 mmol Na). Increases obligate water excretion.
- Urea: 15-30 g per day (0.25-0.5 g/kg). Highly effective osmotic diuretic; generates osmotic diuresis, increasing free water clearance. Drawback: Poor palatability (bitter taste), nausea, limited availability.
- High-Protein Diet: Increases urea production (endogenous source)
-
Loop Diuretics (Furosemide):
- Mechanism: Inhibits thick ascending limb Na-K-2Cl transporter, impairing medullary concentration gradient; reduces maximum urine osmolality from ~1200 to ~300-400 mOsm/kg
- Dose: 20-40 mg daily
- Efficacy: Modest; requires concurrent solute/salt intake to avoid hypovolaemia
- Caution: Can cause hypokalaemia, hypovolaemia
-
Vasopressin Receptor Antagonists (Vaptans):
-
Tolvaptan (oral V2-receptor antagonist):
- Mechanism: Blocks ADH V2 receptor in collecting duct, preventing AQP2 insertion → aquaresis (free water excretion without electrolyte loss)
- Indications: SIADH refractory to fluid restriction; serum Na less than 125 mmol/L with symptoms; euvolaemic or hypervolaemic hyponatraemia
- Dose: 15 mg once daily initially; can uptitrate to 30-60 mg
- Efficacy: Highly effective; rapid rise in serum sodium (often 4-6 mmol/L in 24h)
- Contraindications: Hypovolaemia, anuric renal failure, hepatic impairment (black box warning for hepatotoxicity), concurrent strong CYP3A4 inhibitors
- Adverse Effects: Thirst, dry mouth, polyuria, hepatotoxicity (monitor LFTs monthly)
- Critical Warning: High risk of overcorrection. Monitor serum sodium every 2-4 hours during initiation. Do NOT combine with fluid restriction (exacerbates overcorrection risk). If Na rises > 8 mmol/L in 24h, STOP tolvaptan and implement overcorrection rescue.
- Cost: Expensive; restricted use in many healthcare systems
-
Conivaptan (IV V2/V1a antagonist): Used in USA; not widely available elsewhere
-
-
Demeclocycline:
- Mechanism: Tetracycline antibiotic that induces nephrogenic diabetes insipidus (interferes with ADH action on collecting duct)
- Dose: 600-1200 mg/day in divided doses
- Onset: Delayed (7-14 days)
- Efficacy: Variable and unpredictable
- Adverse Effects: Nephrotoxicity, photosensitivity, tooth discoloration
- Current Use: Largely replaced by vaptans; rarely used
Treatment of Underlying Cause:
- Malignancy: Chemotherapy for SCLC; hyponatraemia may resolve with tumour response
- Drugs: Discontinue offending agent (SSRI, carbamazepine, PPI, etc.)
- Pulmonary: Treat pneumonia, TB, aspergillosis
- CNS: Treat meningitis, abscess; SAH-related SIADH often resolves over weeks
C. Hypervolaemic Hyponatraemia
Principle: Total body sodium is elevated (causing oedema), but effective arterial blood volume is reduced (causing ADH release). Treatment focuses on negative sodium balance and cautious water restriction.
Treatment:
-
Fluid Restriction: 1000-1500 mL per day (less stringent than euvolaemic SIADH; severe restriction poorly tolerated and often ineffective)
-
Sodium Restriction: less than 2 g per day (88 mmol/day)
-
Loop Diuretics: Furosemide 40-120 mg daily (or higher in diuretic resistance)
- Goal: Negative sodium balance (reduce oedema)
- Caution: Overly aggressive diuresis can cause hypovolaemia and worsening ADH secretion
-
Treat Underlying Condition:
- Heart Failure: ACE inhibitors, beta-blockers, spironolactone (aldosterone antagonist reduces ADH drive)
- Cirrhosis: Large-volume paracentesis (if tense ascites), albumin infusion, midodrine/octreotide (increase EABV), consider TIPS if refractory
- Nephrotic Syndrome: Treat underlying glomerular disease; diuretics
-
Vaptans (Tolvaptan):
- Efficacy: Effective at raising serum sodium in heart failure and cirrhosis
- Mortality Benefit: None demonstrated in heart failure (EVEREST trial)
- Cirrhosis: Contraindicated (hepatotoxicity risk)
- Use: Reserved for severe, symptomatic hyponatraemia (Na less than 125) refractory to other measures
Prognosis: Hyponatraemia in heart failure and cirrhosis is a poor prognostic marker. Serum Na less than 125 mmol/L in cirrhosis predicts hepatorenal syndrome and mortality; consideration for liver transplant listing.
Special Clinical Scenarios
1. Exercise-Associated Hyponatraemia (EAH)
Epidemiology: Affects 10-15% of marathon/endurance athletes; severe cases (Na less than 120) in 1-3%
Pathophysiology:
- Excessive hypotonic fluid intake (sports drinks, water) driven by aggressive hydration advice or misperception of thirst
- Non-osmotic ADH secretion (stress, pain, nausea, upright posture during prolonged exercise)
- Result: Water retention exceeds sweat sodium losses → hyponatraemia
Clinical Presentation:
- Occurs during or within 24 hours post-race
- Mild: Nausea, headache, bloating, dizziness
- Severe: Confusion, seizures, coma, pulmonary oedema (volume overload)
Management:
-
Asymptomatic/Mild (Na > 125, no neurological symptoms):
- Oral hypertonic fluids (bouillon, broth) or oral salt tablets
- Restrict free water intake
- Monitor
-
Symptomatic (encephalopathy, seizures):
- "3% Hypertonic Saline: 100 mL IV bolus over 10 minutes; repeat as needed"
- "Do NOT use isotonic saline: These patients are often VOLUME OVERLOADED; 0.9% saline can worsen pulmonary oedema"
- Avoid hypotonic fluids entirely
Prevention:
- "Drink to thirst" education (avoid forced hydration schedules)
- Avoid NSAIDs (increase ADH secretion, reduce renal perfusion)
Clinical Pearl: EAH is a Volume Overload State: Contrary to intuition, marathon runners with EAH are typically volume overloaded (weight gain during race, peripheral oedema, pulmonary crackles). Administering isotonic or hypotonic fluids exacerbates this. Hypertonic saline is the only appropriate fluid; it raises serum sodium without adding excessive volume. This is a classic exam scenario: "collapsed runner, confused, Na 118"—answer is 3% saline, NOT 0.9% saline.
2. Beer Potomania / Tea-and-Toast Syndrome
Pathophysiology: Low dietary solute intake limits renal free water excretion capacity (see Aetiology section).
Clinical Presentation: Chronic, often asymptomatic hyponatraemia in malnourished individuals (alcoholism, elderly, eating disorders)
Diagnostic Clues:
- History of poor nutrition, chronic alcohol use
- Urine osmolality less than 100 mOsm/kg (appropriately dilute; not SIADH)
- Urine sodium often less than 20 mmol/L
Management:
-
Acute Phase:
- CAUTION: Reintroduction of normal diet (solute) will trigger massive aquaresis (the "refeeding aquaresis")
- Serum sodium can rise > 15-20 mmol/L in 24 hours → extremely high ODS risk
- Strategy: Gradual reintroduction of solute OR proactive DDAVP (see overcorrection rescue)
-
Monitoring:
- Check serum sodium every 2-4 hours after refeeding or IV saline administration
- Monitor urine output (expect polyuria: > 200 mL/hour)
-
Overcorrection Rescue (see below): Often required
-
Long-Term: Nutritional rehabilitation, alcohol cessation support
3. Perioperative Hyponatraemia
Risk Factors: Hypotonic IV fluids (0.45% saline, 5% dextrose), post-operative ADH surge (pain, nausea, opioids), prolonged surgery
Prevention:
- Isotonic Maintenance Fluids: Use Hartmann's, Plasma-Lyte, or 0.9% saline (avoid hypotonic fluids)
- Avoid Excessive Free Water: Limit dextrose-containing fluids unless treating hypoglycaemia
Management: As per general principles; consider post-operative SIADH
4. Pregnancy
Physiology: hCG and placental hormones reset osmostat ~5-10 mmol/L lower; serum sodium 130-135 mmol/L is physiological in pregnancy
Labour: Oxytocin has intrinsic antidiuretic activity (structural similarity to ADH). High-dose oxytocin infusions + large volumes of IV dextrose (free water) can cause acute symptomatic hyponatraemia and seizures (water intoxication).
Management:
- Stop oxytocin
- Restrict free water
- Hypertonic saline if severe symptoms
8. Correction Limits & Overcorrection Rescue
The Overcorrection Danger: Osmotic Demyelination Syndrome (ODS)
Overly rapid correction of chronic hyponatraemia is the most feared iatrogenic complication in electrolyte management, capable of causing irreversible neurological devastation.
Correction Limits (European/US Guidelines): [8,10]
- Standard Patients: Maximum rise 10 mmol/L in first 24 hours and 18 mmol/L in first 48 hours
- High-Risk Patients: Maximum rise 8 mmol/L in first 24 hours
High-Risk Groups for ODS:
- Chronic severe hyponatraemia (Na less than 105 mmol/L, or less than 110 present > 48h)
- Chronic alcoholism
- Advanced liver disease (cirrhosis, alcoholic hepatitis)
- Malnutrition (BMI less than 18.5, low albumin)
- Hypokalaemia (K less than 3.0 mmol/L; potassium repletion can precipitate rapid Na rise)
- Starting serum sodium less than 120 mmol/L
Exam Detail: Why 10 mmol/L? The Evidence:
The 10 mmol/L per 24h limit is derived from retrospective case series and systematic reviews showing that ODS occurs almost exclusively when correction exceeds 10-12 mmol/L in 24 hours. A landmark study by Sterns et al. (1986) demonstrated that patients corrected less than 10 mmol/L per 24h had near-zero ODS incidence, whereas those corrected > 12 mmol/L had 25-30% incidence. Subsequent guidelines (European 2014, US Expert Panel 2013) adopted 10 mmol/L as the safety threshold, with 8 mmol/L for high-risk groups to provide additional margin. However, no RCT data exist (performing such a trial would be unethical).
Overcorrection Rescue Strategy
Indications: Serum sodium has risen > 10 mmol/L in 24h (or > 8 mmol/L in high-risk) despite ongoing management.
Goal: Re-lower serum sodium to acceptable rise (less than 10 mmol/L from baseline) to prevent ODS.
Protocol: [9]
-
Stop All Active Correction Therapies:
- Discontinue hypertonic saline
- Stop isotonic saline (if hypovolaemic hyponatraemia)
- Discontinue tolvaptan/vaptans
-
Administer Free Water:
- IV 5% Dextrose: 1-2 litres over 2-4 hours (dextrose is metabolized, leaving free water)
- OR Oral Water: 1-2 litres over 2-4 hours (if patient able to drink safely)
- Goal: Dilute serum sodium
-
Administer Desmopressin (DDAVP):
- Dose: 2-4 mcg IV or SC
- Mechanism: Exogenous ADH analogue; re-induces antidiuresis, preventing urinary free water loss ("closing the floodgates")
- Repeat: Every 6-8 hours as needed to maintain antidiuresis
- Monitoring: Strict input/output; serum sodium every 2 hours
-
Target:
- Re-lower serum sodium by 2-3 mmol/L to bring total 24h rise back under 10 mmol/L
- Example: Baseline Na 115 → Rose to 127 at 18h (12 mmol/L rise, exceeds limit) → Target relowering to 125 (total rise 10 mmol/L)
-
Resume Cautious Correction:
- Once 24h window has "reset," resume correction at less than 10 mmol/L per subsequent 24h
Clinical Pearl: Proactive DDAVP "Clamp" Strategy: In high-risk scenarios (hypovolaemic hyponatraemia requiring saline, beer potomania refeeding), some centres employ a proactive DDAVP clamp from the outset: administer DDAVP 1-2 mcg IV every 6-8 hours to create a fixed antidiuresis, then administer small boluses of 3% saline (50-100 mL) to slowly, controllably raise serum sodium. This "DDAVP clamp + titrated hypertonic saline" approach allows precise control of correction rate, avoiding the unpredictable aquaresis that follows volume resuscitation. Requires ICU-level monitoring. [9]
Monitoring During Correction
Frequency:
- Severe symptomatic (receiving 3% saline boluses): Every 2 hours
- Hypovolaemic (receiving 0.9% saline): Every 1-2 hours (risk of rapid correction)
- SIADH (receiving tolvaptan): Every 2-4 hours during initiation
- Stable chronic (fluid restriction): Every 12-24 hours
Parameters to Monitor:
- Serum sodium, potassium
- Urine output (hourly if polyuria)
- Neurological status (GCS, seizure activity)
- Fluid balance (input/output chart, daily weights)
9. Complications
Osmotic Demyelination Syndrome (ODS)
Epidemiology: Rare; exact incidence unknown (likely underdiagnosed). Occurs almost exclusively with correction > 10-12 mmol/L per 24h.
Pathophysiology: (See Clinical Presentation section for detailed mechanism). Rapid correction of chronic hyponatraemia creates a hypertonic ECF relative to osmolyte-depleted brain cells → intracellular dehydration → oligodendrocyte apoptosis → demyelination.
Anatomical Distribution:
- Central Pontine Myelinolysis (CPM): Pons (most common; 60% of cases)
- Extrapontine Myelinolysis (EPM): Basal ganglia, thalamus, cerebellum, cerebral cortex (40%)
- Often both coexist
Clinical Presentation (Biphasic):
- Initial Phase (Days 1-2): Improvement in hyponatraemia symptoms (e.g., resolution of confusion)
- Latent Phase (Days 2-4): Patient appears stable or improving
- Deterioration Phase (Days 3-7):
- Pontine Symptoms (CPM):
- Dysarthria (slurred speech, difficulty articulating)
- Dysphagia (difficulty swallowing, risk of aspiration)
- Spastic quadriparesis (progressive limb weakness, increased tone, hyperreflexia)
- Pseudobulbar palsy (emotional lability, impaired facial expression)
- Locked-in Syndrome (severe cases): Patient is awake and aware but completely paralyzed except for vertical eye movements and blinking. Can hear and understand but cannot speak or move.
- Extrapontine Symptoms (EPM):
- Movement disorders (parkinsonism, dystonia, tremor)
- Behavioural changes (apathy, mutism, catatonia)
- Seizures
- Pontine Symptoms (CPM):
Diagnosis:
- MRI Brain: Modality of choice
- "Timing: Changes may not appear until 2-4 weeks post-correction"
- Findings: T2/FLAIR hyperintensity in pons ("trident" or "bat-wing" sign) or extrapontine regions (basal ganglia, thalamus)
- "Early MRI: May be normal; repeat at 2-4 weeks if clinical suspicion high"
- CT Brain: Insensitive; often normal
Prognosis:
- Mild Cases: Partial recovery over months to years; residual dysarthria, gait disturbance
- Moderate Cases: Significant disability; require long-term rehabilitation
- Severe Cases (Locked-in): Poor; many remain severely disabled or in vegetative state
- Mortality: 20-30% in severe cases
Treatment:
- No Proven Therapy: ODS is largely irreversible
- Re-lowering Sodium: Anecdotal reports suggest immediate re-induction of hyponatraemia (DDAVP + free water within 24-48h of overcorrection) may prevent or mitigate ODS, but evidence is limited
- Supportive Care: Rehabilitation (physiotherapy, speech therapy), aspiration precautions, PEG feeding if severe dysphagia
Prevention is the ONLY Cure: Strict adherence to correction limits.
Cerebral Oedema (from Hyponatraemia)
Mechanism: Acute hyponatraemia → water influx into astrocytes → cytotoxic oedema → increased intracranial pressure
Clinical Features: Headache, vomiting, seizures, coma, brainstem herniation signs (pupillary abnormalities, posturing, Cushing's reflex)
Diagnosis: CT or MRI brain (effacement of sulci, loss of grey-white differentiation, compression of ventricles)
Treatment: Hypertonic saline (see Emergency Management)
Prognosis: Excellent if treated promptly; fatal if herniation occurs
Rhabdomyolysis
Association: Severe symptomatic hyponatraemia (seizures) can cause rhabdomyolysis (muscle breakdown from prolonged seizure activity)
Diagnosis: Elevated CK (creatine kinase), myoglobinuria
Complications: Acute kidney injury (myoglobin nephrotoxicity), hyperkalaemia, hypocalcaemia
Treatment: IV fluids, treatment of hyponatraemia
10. Prognosis
Acute Symptomatic Hyponatraemia
- Untreated: High mortality (20-50% if severe symptoms; cerebral herniation)
- Treated Promptly with Hypertonic Saline: Excellent neurological recovery; mortality less than 5%
- Key Determinant: Speed of recognition and treatment
Chronic Hyponatraemia
Mild (130-134 mmol/L):
- Increased all-cause mortality (HR 1.15-1.25) [6]
- Increased falls and fracture risk (HR 2.1 for hip fracture) [7]
- Reversible neurocognitive deficits (gait improves with correction)
Moderate (125-129 mmol/L):
- Increased mortality (HR 1.3-1.5) [6]
- Higher fracture risk, longer hospital stays
Severe (less than 125 mmol/L):
- Increased mortality (HR 1.5-2.5) [6]
- ODS risk with correction
SIADH
Prognosis depends on underlying cause:
- Drug-induced: Resolves with drug cessation (weeks to months)
- Malignancy (SCLC): Poor; median survival 6-12 months (due to cancer, not SIADH)
- Pulmonary (pneumonia): Resolves with infection treatment (days to weeks)
- CNS: Variable; post-SAH SIADH often resolves over 2-6 weeks
- Idiopathic: Chronic; may require long-term fluid restriction or vaptans
Hyponatraemia in Heart Failure and Cirrhosis
- Independent predictor of mortality: Serum Na less than 130 correlates with NYHA class IV, hepatorenal syndrome, need for transplant
- Heart Failure: Na less than 125 associated with doubled 1-year mortality
- Cirrhosis: Na less than 125 predicts hepatorenal syndrome, variceal bleeding, mortality; used in MELD-Na score for transplant prioritization
11. Prevention & Screening
In-Hospital Prevention
-
Avoid Hypotonic Maintenance Fluids: Use isotonic fluids (0.9% saline, Hartmann's, Plasma-Lyte) for routine maintenance. Hypotonic fluids (0.45% saline, 5% dextrose alone) should be restricted to specific indications (hypernatraemia correction, free water deficit replacement).
-
Monitor High-Risk Patients: Daily electrolytes for:
- Post-operative patients (ADH surge)
- Patients on diuretics (especially thiazides)
- Patients on high-risk medications (SSRIs, carbamazepine, PPIs)
- Severe illness (pneumonia, meningitis)
- Neurological/neurosurgical patients (SAH, trauma)
-
Thiazide Monitoring: Check serum sodium at 2 weeks after thiazide initiation (peak incidence of hyponatraemia)
-
Educate Nursing Staff: Recognition of hyponatraemia symptoms (confusion, falls, nausea)
Community/Ambulatory Prevention
-
Medication Review: Elderly patients on SSRIs, thiazides, carbamazepine should have periodic sodium monitoring (e.g., 6-monthly)
-
Athlete Education: "Drink to thirst" campaigns to prevent EAH; avoid forced hydration
-
Chronic Disease Management: Optimize heart failure, cirrhosis management to reduce hyponatraemia incidence
Screening
No universal screening indicated, but consider checking serum sodium in:
- Elderly with falls or cognitive decline
- New-onset confusion (DDx: UTI, stroke, hyponatraemia)
- Cancer patients (SCLC, pancreatic) at diagnosis
- Chronic SSRI or diuretic users
12. Key Guidelines & Evidence
Major Clinical Practice Guidelines
-
European Society of Endocrinology / European Society of Intensive Care Medicine (2014): [8]
- Comprehensive guideline on diagnosis and treatment
- Defines acute vs. chronic (less than 48h vs. > 48h)
- Recommends 150 mL 3% NaCl boluses for severe symptoms
- Correction limits: 10 mmol/L per 24h (8 mmol/L in high-risk)
- Emphasizes DDAVP rescue for overcorrection
-
US Expert Panel Recommendations (Verbalis et al., 2013): [10]
- Similar principles to European guideline
- Supports urea for SIADH treatment (15-30 g/day)
- Describes DDAVP reactive and proactive strategies
- Provides detailed algorithms
-
UK NICE Guidance:
- No specific NICE guideline on hyponatraemia; defer to European guideline
Landmark Studies and Evidence
| Study | Design | Key Findings |
|---|---|---|
| Upadhyay et al., 2006 [1] | Review | Hyponatraemia prevalence 15-30% in hospital; most common electrolyte disorder |
| Renneboog et al., 2006 [7] | Prospective cohort | Mild chronic hyponatraemia associated with 67% increased falls, attention deficits, gait instability |
| Corona et al., 2013 [6] | Meta-analysis | Even moderate hyponatraemia (125-129) increases mortality (HR 1.3); dose-response relationship |
| Sterns et al., 2015 [3] | Expert review | Comprehensive pathophysiology of hyponatraemia and ODS; correction recommendations |
| Spasovski et al., 2014 [8] | Guideline | European consensus on diagnosis and treatment; 150 mL bolus protocol |
| Sood et al., 2013 [9] | Interventional study | DDAVP + hypertonic saline "clamp" method for controlled correction in high-risk patients |
| Schrier et al., 2006 [11] | RCT (SALT-1, SALT-2) | Tolvaptan effective for raising Na in SIADH, heart failure; aquaresis without electrolyte loss |
13. Examination Focus
Common MRCP Exam Scenarios
1. Data Interpretation Station (Written Exam):
Question: A 68-year-old woman is found to have Na 118, K 4.2, Urea 3.5, Creatinine 85 on routine bloods. Serum osmolality 250, urine osmolality 420, urine sodium 65. She takes bendroflumethiazide for hypertension. What is the most appropriate initial management?
Model Answer:
- Diagnosis: Thiazide-induced hyponatraemia, likely with features of SIADH (concentrated urine, high urine Na, low serum osmolality, euvolaemic clinically)
- Initial Management:
- Stop thiazide (essential; causative agent)
- Assess for symptoms: If asymptomatic/mild symptoms, no emergency treatment needed
- Fluid restriction: 1000 mL/day (first-line for SIADH-like picture)
- Monitor: Serum sodium daily; expect gradual rise over 3-7 days
- Correct hypokalaemia if present (potassium repletion can accelerate sodium rise; monitor closely)
- Exclude other causes: 9am cortisol, TSH (though thiazide is likely culprit)
2. PACES Station: Acute Medical Take:
Scenario: A 55-year-old man presents to A&E with a first seizure. He is post-ictal, confused. Na 112, K 3.8. No prior history. What are your immediate management steps?
Model Answer:
- Recognition: Severe symptomatic acute hyponatraemia (seizure, confusion, Na 112)
- Emergency Management:
- Airway, Breathing, Circulation: Ensure airway patent, oxygen, IV access
- 3% Hypertonic Saline Bolus: 150 mL IV over 20 minutes immediately
- Benzodiazepines: Lorazepam 4 mg IV for seizure (if ongoing or recurrent)
- Reassess at 20 minutes: Check serum sodium, neurological status; repeat bolus if symptoms persist (max 3 boluses)
- Target: Raise Na by 4-6 mmol/L in first 6 hours (to ~116-118)
- Monitor: ICU/HDU admission, serum Na every 2 hours, neuro obs
- Investigate Cause: History (new drugs? SIADH risk factors?), serum osmolality, urine osmolality/Na, CXR, CT head, cortisol, TFTs
- Avoid: Do NOT aim to correct to normal (135); do NOT use 0.9% saline alone (too slow for emergency)
3. History Taking Station (PACES 2):
Scenario: A 72-year-old woman presented with confusion. Na 120. You have corrected this to 125 with fluid restriction. Take a history to identify the underlying cause.
Key Questions:
- Drug History: "Have you started any new tablets recently? Do you take water tablets? Antidepressants? Tablets for pain or epilepsy?"
- Fluid Intake: "What do you normally drink? How much tea/coffee/water per day? Any alcohol?"
- Fluid Output: "Any vomiting or diarrhoea? Have you noticed changes in how much you're urinating?"
- Symptoms of SIADH Causes:
- Malignancy: "Any weight loss, cough, breathlessness, night sweats?"
- Pulmonary: "Recent chest infection? Pneumonia?"
- CNS: "Any headaches, neck stiffness, head injury?"
- Endocrine Screen: "Do you feel cold all the time? (hypothyroid) Any dizziness when standing? (Addison's) Any skin darkening?"
- Past Medical History: "Heart problems? Liver problems? Kidney problems?"
4. Viva Question:
Examiner: "A patient with known SIADH secondary to small cell lung cancer has failed fluid restriction. Her sodium remains 122. What are your next steps?"
Model Answer: "SIADH refractory to fluid restriction requires second-line therapy. I would consider:
- Compliance Check: Ensure patient is actually adhering to less than 1L/day fluid restriction; review fluid balance charts
- Urine Studies: Check urine osmolality and urine Na+K. If urine osmolality > 500 or urine (Na+K) > serum Na, fluid restriction is unlikely to succeed.
- Second-Line Options:
- Oral Urea (15-30 g/day): Osmotic diuresis; increases free water clearance. Highly effective but poor palatability.
- Salt Tablets (2-6 g/day NaCl): Increases solute load.
- Tolvaptan (V2 antagonist): 15 mg daily, titrate. Very effective but expensive, requires monitoring (hepatotoxicity risk, overcorrection risk). Monitor Na every 4 hours during initiation.
- Treat Underlying Cancer: Liaise with oncology; chemotherapy response may resolve SIADH.
- Monitoring: Regular serum Na, avoid overcorrection (limit 10 mmol/L per 24h)."
Common Exam Pitfalls
❌ Assuming all hyponatraemia requires saline: Euvolaemic SIADH worsens with 0.9% saline ("desalination") ❌ Correcting to normal (135) in acute setting: Only need 4-6 mmol/L rise to reverse cerebral oedema ❌ Ignoring correction limits: > 10 mmol/L per 24h → ODS risk ❌ Forgetting to check urine studies: Serum sodium alone does not diagnose aetiology ❌ Not recognizing CSW vs. SIADH: Both have high urine Na post-SAH; CSW is hypovolaemic (needs saline), SIADH euvolaemic (needs restriction) ❌ Using 0.9% saline in exercise-associated hyponatraemia: Patients are volume overloaded; use 3% saline
Viva Opening Statement
"Hyponatraemia reflects a disorder of water balance, not sodium balance. My approach prioritizes symptom severity first—severe symptoms (seizures, coma) are an emergency requiring immediate hypertonic saline regardless of chronicity. In non-emergency settings, I systematically classify by volume status and ADH activity to identify the underlying cause. The cornerstone of safe management is strict adherence to correction limits—maximum 10 mmol/L per 24 hours, or 8 mmol/L in high-risk patients—to prevent osmotic demyelination syndrome. Hypovolaemic hyponatraemia carries the highest overcorrection risk due to rapid ADH suppression after volume resuscitation, requiring close monitoring and often proactive DDAVP strategies."
High-Yield Facts for Vivas
- Severe symptomatic hyponatraemia: 150 mL 3% NaCl bolus over 20 min; target 4-6 mmol/L rise in 6h
- SIADH diagnosis: Hypotonic hyponatraemia + urine osm > 100 + urine Na > 40 + euvolaemia + normal thyroid/adrenal/renal function
- Thiazides > Loop diuretics for hyponatraemia risk (intact medullary gradient)
- Correction limit: 10 mmol/L per 24h; 8 mmol/L if high-risk (alcoholism, cirrhosis, malnutrition, K less than 3.0, Na less than 105)
- Overcorrection rescue: Stop active treatment + 5% dextrose + DDAVP 2-4 mcg IV
- ODS presentation: Biphasic; dysarthria, dysphagia, quadriparesis, locked-in syndrome 3-7 days post-correction
- CSW vs. SIADH: CSW is hypovolaemic (polyuria, negative balance); SIADH euvolaemic
- Beer potomania: Massive aquaresis upon refeeding → extreme ODS risk
- Exercise-associated hyponatraemia: Volume overload; use 3% saline, NOT 0.9% saline
- Desalination phenomenon: In severe SIADH, 0.9% saline can worsen hyponatraemia if urine (Na+K) > serum Na
14. Patient/Layperson Explanation
"Your blood test shows that the level of sodium—one of the important salts in your blood—is lower than normal. Sodium helps control the amount of water in and around your cells, especially in your brain.
When sodium is low, it usually means there's too much water in your blood, which dilutes the sodium. This can happen for several reasons, such as certain medications (like water tablets or antidepressants), drinking too much fluid, or medical conditions affecting your heart, liver, or kidneys.
Low sodium can make you feel tired, confused, or dizzy, and in severe cases, it can cause seizures. However, fixing it requires careful, gradual treatment. Raising your sodium level too quickly can damage your brain, so we need to correct it slowly and monitor you closely.
Treatment depends on the cause. It might involve restricting how much you drink, stopping certain medications, or in urgent situations, giving you a special salt solution through a drip. We'll check your blood regularly to make sure your sodium level is improving safely."
15. References
-
Upadhyay A, Jaber BL, Madias NE. Incidence and prevalence of hyponatremia. Am J Med. 2006;119(7 Suppl 1):S30-5. PMID: 16843082
-
Wald R, Jaber BL, Price LL, et al. Impact of hospital-associated hyponatremia on selected outcomes. Arch Intern Med. 2010;170(3):294-302. PMID: 20142578
-
Sterns RH. Disorders of plasma sodium—causes, consequences, and correction. N Engl J Med. 2015;372(1):55-65. PMID: 25551526
-
Funk GC, Lindner G, Druml W, et al. Incidence and prognosis of dysnatremias present on ICU admission. Intensive Care Med. 2010;36(2):304-11. PMID: 19847398
-
Hawkins RC. Age and gender as risk factors for hyponatremia and hypernatremia. Clin Chim Acta. 2003;337(1-2):169-72. PMID: 14556829
-
Corona G, Giuliani C, Parenti G, et al. Moderate hyponatremia is associated with increased risk of mortality: evidence from a meta-analysis. PLoS One. 2013;8(12):e80451. PMID: 24367479
-
Renneboog B, Musch W, Vandemergel X, et al. Mild chronic hyponatremia is associated with falls, unsteadiness, and attention deficits. Am J Med. 2006;119(1):71.e1-8. PMID: 16431193
-
Spasovski G, Vanholder R, Allolio B, et al. Clinical practice guideline on diagnosis and treatment of hyponatraemia. Eur J Endocrinol. 2014;170(3):G1-G47. PMID: 24569125
-
Sood L, Sterns RH, Hix JK, et al. Hypertonic saline and desmopressin: a simple strategy for safe correction of severe hyponatremia. Am J Kidney Dis. 2013;61(4):571-8. PMID: 23266328
-
Verbalis JG, Goldsmith SR, Greenberg A, et al. Diagnosis, evaluation, and treatment of hyponatremia: expert panel recommendations. Am J Med. 2013;126(10 Suppl 1):S1-42. PMID: 24074529
-
Schrier RW, Gross P, Gheorghiade M, et al. Tolvaptan, a selective oral vasopressin V2-receptor antagonist, for hyponatremia. N Engl J Med. 2006;355(20):2099-112. PMID: 17108345
-
Adrogué HJ, Madias NE. Hyponatremia. N Engl J Med. 2000;342(21):1581-9. PMID: 10824078
-
Ellison DH, Berl T. Clinical practice. The syndrome of inappropriate antidiuresis. N Engl J Med. 2007;356(20):2064-72. PMID: 17507705
-
Hannon MJ, Thompson CJ. The syndrome of inappropriate antidiuretic hormone: prevalence, causes and consequences. Eur J Endocrinol. 2010;162(Suppl 1):S5-12. PMID: 20164214
-
Palmer BF, Gates JR, Lader M. Causes and management of hyponatremia. Ann Pharmacother. 2003;37(11):1694-702. PMID: 14565800
-
Yasir M, Mechanic OJ. Syndrome of Inappropriate Antidiuretic Hormone Secretion. [Updated 2023]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2023.
-
Sterns RH, Nigwekar SU, Hix JK. The treatment of hyponatremia. Semin Nephrol. 2009;29(3):282-99. PMID: 19523575
-
Hew-Butler T, Rosner MH, Fowkes-Godek S, et al. Statement of the Third International Exercise-Associated Hyponatremia Consensus Development Conference, Carlsbad, California, 2015. Clin J Sport Med. 2015;25(4):303-20. PMID: 26102438
-
Hoorn EJ, Zietse R. Diagnosis and Treatment of Hyponatremia: Compilation of the Guidelines. J Am Soc Nephrol. 2017;28(5):1340-1349. PMID: 28381550
-
Burst V. Etiology and Epidemiology of Hyponatremia. Front Horm Res. 2019;52:24-35. PMID: 32097944
Learning map
Use these linked topics to study the concept in sequence and compare related presentations.
Prerequisites
Start here if you need the foundation before this topic.
- Renal Physiology: Water Handling
- Acid-Base Disorders
Differentials
Competing diagnoses and look-alikes to compare.
- Hypernatraemia
- Cerebral Salt Wasting
Consequences
Complications and downstream problems to keep in mind.
- Osmotic Demyelination Syndrome
- Cerebral Oedema