Irritable Bowel Syndrome
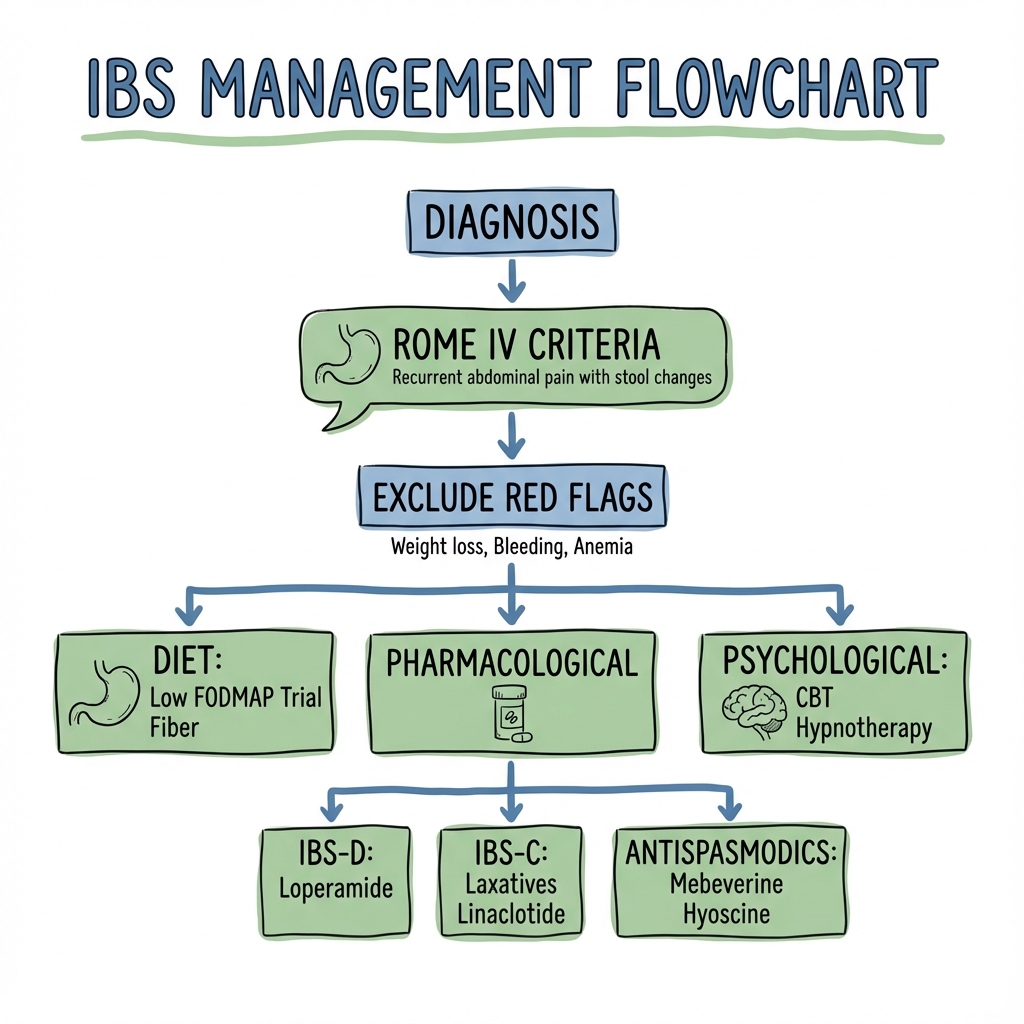
Diagnosis is clinical, based on the Rome IV criteria, which require recurrent abdominal pain at least 1 day per week in the last 3 months, associated with two or more of: (1) related to defecation, (2) associated with...
What matters first
Diagnosis is clinical, based on the Rome IV criteria, which require recurrent abdominal pain at least 1 day per week in the last 3 months, associated with two or more of: (1) related to defecation, (2) associated with...
Unintentional weight loss
11 Jan 2026
Generated educational material; verify before clinical use.
Visible references section
See the concept before reading it
Study the key anatomy, imaging, and decision pathways as full teaching plates.
Clinical board
A visual summary of the highest-yield teaching signals on this page.
Urgent signals
Safety-critical features pulled from the topic metadata.
- Unintentional weight loss
- Rectal bleeding
- Age greater than 50 with new symptoms
- Family history of colorectal cancer or IBD
Content status and exam context
This page is AI-generated educational content. It may contain errors or omissions and is not a substitute for current guidelines, local protocols, senior clinical judgement, or professional medical advice.
MedVellum does not claim an individual clinician reviewer, board certification, or professional credential for this page unless a future version names a real, verifiable reviewer.
Clinical explanation and evidence
Irritable Bowel Syndrome
1. Clinical Overview
Summary
Irritable bowel syndrome (IBS) is a chronic functional gastrointestinal disorder characterised by recurrent abdominal pain associated with defecation or change in bowel habit, in the absence of detectable organic disease. IBS is classified as a disorder of gut-brain interaction (DGBI), reflecting the complex interplay between visceral hypersensitivity, altered gut motility, gut-brain axis dysfunction, and microbiome alterations. [1,2]
Diagnosis is clinical, based on the Rome IV criteria, which require recurrent abdominal pain at least 1 day per week in the last 3 months, associated with two or more of: (1) related to defecation, (2) associated with change in stool frequency, or (3) associated with change in stool form. [2] Symptom onset must be at least 6 months prior to diagnosis. IBS is subtyped based on predominant stool pattern using the Bristol Stool Scale: IBS-C (constipation-predominant), IBS-D (diarrhoea-predominant), IBS-M (mixed), or IBS-U (unsubtyped). [2]
IBS is a diagnosis of exclusion in patients with red flag features. Essential investigations include FBC, CRP/ESR, and coeliac serology (tissue transglutaminase IgA with total IgA). [3,4] Faecal calprotectin should be checked in diarrhoea-predominant IBS to exclude inflammatory bowel disease. [3]
Management is multifaceted and symptom-directed. First-line approaches include dietary modification (particularly low FODMAP diet), lifestyle advice, and explanation of the condition. [3,5] Pharmacotherapy is tailored to the predominant symptom: antispasmodics (mebeverine, peppermint oil) for pain and bloating; osmotic laxatives (macrogol) or secretagogues (linaclotide) for constipation; loperamide for diarrhoea; and neuromodulators (low-dose tricyclic antidepressants or SSRIs) for refractory pain. [3,6] Psychological therapies, particularly cognitive behavioural therapy (CBT) and gut-directed hypnotherapy, have strong evidence and are recommended for patients with inadequate response to first-line measures. [7,8]
IBS is a chronic, relapsing-remitting condition that does not progress to structural bowel disease or increase colorectal cancer risk. [9] However, it significantly impacts quality of life, work productivity, and healthcare utilisation. [10]
Key Facts
- Definition: Functional disorder of gut-brain interaction characterised by recurrent abdominal pain and altered bowel habit (Rome IV criteria) [2]
- Global Prevalence: 9.2% by Rome III criteria; 3.8% by Rome IV criteria (more restrictive) [11]
- Demographics: Female:male ratio 1.5-2:1; peak age 20-40 years; lower prevalence in elderly [11,12]
- Subtypes: IBS-D (diarrhoea-predominant), IBS-C (constipation-predominant), IBS-M (mixed), IBS-U (unsubtyped) [2]
- Pathophysiology: Multifactorial—visceral hypersensitivity, altered motility, gut-brain axis dysfunction, microbiome dysbiosis, low-grade inflammation [1,13]
- Gold Standard Diagnosis: Clinical diagnosis using Rome IV criteria; investigations to exclude organic disease [2,3]
- First-line Treatment: Education, dietary modification (low FODMAP diet 50-80% response), symptom-directed pharmacotherapy [3,5]
- Psychological Comorbidity: Anxiety or depression in up to 30% of IBS patients; strongly influences quality of life [14]
- Prognosis: Chronic, relapsing-remitting; does not progress to IBD or colorectal cancer; 30-50% improve over time [9]
Clinical Pearls
Rome IV Pearl: The Rome IV criteria are more restrictive than Rome III, requiring abdominal pain at least 1 day/week (not just 3 days/month). This reduces prevalence but improves specificity. [2,11]
Coeliac Pearl: NICE and BSG guidelines recommend testing all patients meeting IBS criteria for coeliac disease with tissue transglutaminase IgA and total IgA, as up to 4% may have undiagnosed coeliac disease. [3,4]
FODMAP Pearl: Low FODMAP diet is effective in 50-80% of IBS patients, but requires structured dietitian-led implementation with elimination phase (4-8 weeks) followed by systematic reintroduction to identify individual triggers. [5,15]
Red Flag Pearl: IBS is a diagnosis of exclusion in patients aged over 50 with new symptoms. Any red flag (rectal bleeding, weight loss, nocturnal symptoms, family history of IBD/colorectal cancer, anaemia) mandates investigation with colonoscopy and appropriate imaging. [3]
Psychological Pearl: The gut-brain axis is central to IBS pathophysiology. CBT and gut-directed hypnotherapy have equivalent efficacy to pharmacotherapy and are recommended by international guidelines. [7,8,14]
Post-infectious Pearl: Approximately 10% of patients develop IBS following acute gastroenteritis (post-infectious IBS), suggesting a role for persistent gut microbiome changes and low-grade inflammation. [16]
Bile Acid Pearl: In IBS-D refractory to first-line therapy, consider bile acid malabsorption (BAM)—trial of bile acid sequestrant (cholestyramine, colesevelam) can be diagnostic and therapeutic. [3]
Neuromodulator Pearl: Low-dose tricyclic antidepressants (amitriptyline 10-30mg nocte) are effective for pain-predominant IBS through visceral analgesic effects, not antidepressant action. Start low, titrate slowly. [6]
Why This Matters Clinically
IBS is the most common functional gastrointestinal disorder, affecting 1 in 10 adults globally, and accounts for 25-50% of gastroenterology referrals. [10,12] Despite its benign prognosis, IBS substantially impairs quality of life, with severity comparable to inflammatory bowel disease and diabetes. [10]
The condition incurs significant economic costs: patients with IBS have 50% higher healthcare utilisation and work absenteeism than matched controls, with annual direct costs estimated at $1-2 billion in the UK and $20-30 billion in the USA. [10] Indirect costs from productivity loss are even greater.
Effective management requires confident clinical diagnosis (avoiding over-investigation), tailored symptom-directed therapy, and recognition of psychological comorbidity. The availability of evidence-based dietary (FODMAP), pharmacological (neuromodulators, secretagogues), and psychological interventions (CBT, hypnotherapy) means most patients can achieve meaningful symptom control. [3,5,7,8]
Recognising when to investigate versus when to diagnose confidently based on Rome IV criteria, and understanding the role of the gut-brain axis, are critical skills for both primary care physicians and gastroenterologists.
2. Epidemiology
Prevalence
Global Prevalence
A systematic review and meta-analysis of 57 studies across 38 countries (423,362 participants) found pooled IBS prevalence of 9.2% (95% CI 7.6-10.8%) using Rome III criteria. [11] When the more restrictive Rome IV criteria were applied (6 studies, 82,476 participants), prevalence was lower at 3.8% (95% CI 3.1-4.5%). [11]
Earlier meta-analysis using any symptom-based criteria (81 studies, 260,960 participants) reported global pooled prevalence of 11.2% (95% CI 9.8-12.8%), with substantial variation by country (1.1% to 45%). [12]
The Rome Foundation Global Study (73,076 respondents across 33 countries) found that 40.3% of internet respondents and 20.7% of household survey respondents met criteria for at least one functional gastrointestinal disorder; IBS accounted for a significant proportion. [17]
Regional Variation
Prevalence varies significantly by region, even when uniform diagnostic criteria are applied: [11,12]
- Europe: 7-10%
- North America: 10-15%
- South America: 15-20%
- Asia: 5-10% (lower in some Southeast Asian countries)
- Africa: Limited data; estimates 5-15%
This geographic variation persists even with identical methodology, suggesting true environmental, dietary, genetic, or cultural differences beyond diagnostic criteria. [1,11]
Healthcare-Seeking Behaviour
Only 30-40% of individuals meeting IBS criteria seek medical attention. [12] Those who consult healthcare tend to have more severe symptoms, greater psychological comorbidity, and poorer quality of life. [10]
Demographics
Age
- Peak incidence: 20-40 years [11,12]
- Onset after 50: Less common; warrants thorough investigation to exclude organic disease [3]
- Elderly: Lower prevalence; new diagnosis in elderly should prompt colonoscopy [3]
- Adolescents/young adults: IBS is common; Rome IV criteria validated in this age group [18]
Sex
IBS shows female predominance across all studies: [11,12]
- Female:male ratio: 1.5-2:1
- Odds ratio: Women have 1.46-1.67 times higher odds of IBS than men (95% CI 1.33-1.82) [11,12]
- Subtype differences: IBS-C more common in women; IBS-D more common in men [11]
Hormonal factors may play a role: women report symptom fluctuation with menstrual cycle, and prevalence is highest in reproductive years. [1]
Socioeconomic Status
Limited high-quality data. Some studies suggest lower socioeconomic status associates with higher prevalence, but findings are inconsistent. [12]
Risk Factors
| Factor | Association | Evidence |
|---|---|---|
| Female sex | OR 1.5-2.0 | Consistent across all studies [11,12] |
| Age less than 50 years | OR 0.75 for age >50 vs less than 50 | Decreases with age [12] |
| Post-infectious gastroenteritis | 10-15% develop post-infectious IBS | Risk factors: female, younger age, severity of infection, psychological distress [16] |
| Anxiety and depression | Present in 30% of IBS patients | Bidirectional relationship; psychological factors predict IBS development [14] |
| Adverse childhood events | Increased risk (OR ~2-3) | Physical, sexual, or emotional abuse associated with adult IBS [1] |
| Antibiotic exposure | Alters microbiome; may increase risk | Observational data; causality unclear [13] |
| Family history of IBS | 2-3x increased risk | Suggests genetic predisposition or shared environment [1] |
| Stress | Symptom exacerbation | Chronic stress associated with symptom onset and flares [14] |
Burden of Disease
Quality of Life
IBS significantly impairs health-related quality of life across physical, psychological, and social domains. [10] Severity is comparable to chronic conditions such as inflammatory bowel disease, diabetes, and depression. [10]
Healthcare Utilisation
Patients with IBS have: [10]
- 50% higher healthcare consultations than matched controls
- 2-3x higher prescription costs
- Higher rates of unnecessary investigations and surgical procedures (e.g., appendicectomy, cholecystectomy)
Economic Impact
- Direct costs: Healthcare consultations, investigations, medications
- "UK: £1-2 billion annually [10]"
- "USA: $20-30 billion annually [10]"
- Indirect costs: Work absenteeism, presenteeism (reduced productivity while at work), unemployment
- IBS patients take 3x more sick days than healthy controls [10]
- Indirect costs exceed direct costs by 2-3 fold [10]
3. Pathophysiology
Mechanism Overview
IBS is a multifactorial disorder of gut-brain interaction. No single pathophysiological mechanism explains all cases; rather, IBS arises from variable combinations of visceral hypersensitivity, altered motility, gut-brain axis dysfunction, microbiome dysbiosis, immune activation, and genetic predisposition. [1,13]
1. Visceral Hypersensitivity
Definition: Heightened perception of normal visceral stimuli, leading to pain at lower thresholds than healthy individuals. [1]
Evidence:
- Rectal balloon distension studies demonstrate lower pain thresholds in IBS patients compared to controls [1]
- Functional MRI shows altered central pain processing with increased activation of pain-related brain regions (anterior cingulate cortex, insula) during visceral stimulation [1]
- Peripheral sensitisation: increased nociceptor excitability in gut wall
- Central sensitisation: amplified pain signaling in spinal cord and brain
Clinical relevance: Explains why IBS patients experience pain from normal bowel contractions and gas that would be painless in healthy individuals.
2. Altered Gut Motility
Mechanism: Abnormal patterns of intestinal contractility contribute to constipation or diarrhoea. [1]
Patterns: [1]
- IBS-D: Increased colonic motility, accelerated transit, reduced water absorption → diarrhoea
- IBS-C: Decreased colonic motility, delayed transit, increased water absorption → constipation
- IBS-M: Fluctuating motility patterns
Evidence: Colonic transit studies show faster transit in IBS-D and slower transit in IBS-C compared to controls. [1] However, motility patterns do not correlate perfectly with symptoms, indicating other mechanisms are involved.
3. Gut-Brain Axis Dysfunction
Concept: Bidirectional communication between the enteric nervous system (ENS) and central nervous system (CNS) via neural (vagus nerve), endocrine (HPA axis), and immune pathways. [1,14]
Mechanisms: [1,14]
- CNS to gut: Psychological stress → HPA axis activation → corticotropin-releasing hormone (CRH) release → increased gut permeability, motility, and visceral sensitivity
- Gut to CNS: Gut inflammation, microbiome metabolites, and visceral signals influence mood, cognition, and pain perception
- Dysregulated stress response: IBS patients show exaggerated HPA axis response to stress
Clinical relevance: Explains stress-symptom relationship and efficacy of psychological therapies and neuromodulators. [7,8,14]
4. Gut Microbiome Dysbiosis
Findings: IBS patients show altered gut microbiome composition, diversity, and function compared to healthy controls. [13]
Patterns: [13]
- Reduced diversity (less microbial richness)
- Altered Firmicutes:Bacteroidetes ratio
- Changes in metabolites: altered short-chain fatty acids (SCFAs), increased gas production
Post-infectious IBS: 10-15% of patients develop IBS following acute gastroenteritis. [16] Risk factors include severity of infection, female sex, younger age, and psychological stress. [16] Persistent microbiome changes and low-grade inflammation may underlie symptoms. [16]
Therapeutic implications: Low FODMAP diet alters microbiome and reduces fermentation; probiotics have inconsistent evidence. [5,13]
5. Low-Grade Inflammation and Immune Activation
Evidence: [13,16]
- Increased mast cells in colonic mucosa of IBS patients, particularly post-infectious IBS
- Elevated pro-inflammatory cytokines (IL-6, IL-8, TNF-alpha) in some patients
- Increased intestinal permeability ("leaky gut") in subset of patients
- Activated immune cells in proximity to enteric nerves → sensitisation
Clinical relevance: Provides rationale for anti-inflammatory therapies under investigation; explains post-infectious IBS.
6. Dietary Factors: FODMAPs
FODMAP: Fermentable Oligosaccharides, Disaccharides, Monosaccharides And Polyols. [5,15]
Mechanism: [5,15]
- Poorly absorbed in small intestine
- Rapidly fermented by colonic bacteria → gas production (H₂, CO₂, CH₄)
- Osmotic effect → water drawn into bowel lumen
- Result: bloating, distension, altered bowel habit
High FODMAP foods: Onion, garlic, wheat, lactose-containing dairy, apples, pears, stone fruits, legumes, high-fructose corn syrup, sorbitol/mannitol (artificial sweeteners). [5,15]
Evidence: Randomised controlled trials show low FODMAP diet reduces IBS symptoms in 50-80% of patients. [5,15]
7. Genetic Predisposition
Evidence: [1]
- Twin studies: concordance higher in monozygotic (17%) than dizygotic (8%) twins
- Family clustering: 2-3x increased risk in first-degree relatives
- Candidate genes: polymorphisms in serotonin transporter (5-HTT), TNF-alpha, toll-like receptors, and bile acid synthesis genes
Clinical relevance: IBS is polygenic; no single gene accounts for disease, but genetic factors modulate susceptibility.
Integrated Model
IBS arises from interplay of:
- Genetic predisposition → susceptibility to visceral hypersensitivity and altered motility
- Environmental triggers → infection, antibiotics, stress, diet
- Microbiome dysbiosis → altered fermentation, immune activation
- Gut-brain axis dysfunction → amplified pain signaling, stress-symptom cycle
- Psychological factors → anxiety, depression, catastrophising → worsened symptoms and quality of life
No single mechanism explains all cases; IBS is best understood as a spectrum of disorders with heterogeneous pathophysiology. [1,13]
Molecular Mechanisms (Advanced Detail)
1. Serotonin Signaling and Gut Motility
Role of 5-HT (serotonin): [1,27]
- Source: 95% of body's serotonin is in gut; produced by enterochromaffin cells (EC cells) in intestinal mucosa
- Function: Regulates gut motility, secretion, visceral sensation
- Receptors:
- "5-HT3: Mediates nociception (pain), secretion; activation → nausea, pain"
- "5-HT4: Prokinetic; stimulates peristalsis, accelerates transit"
IBS-D pathophysiology: [1,27]
- Increased post-prandial 5-HT release → accelerated colonic transit → diarrhoea
- Polymorphisms in serotonin transporter gene (SLC6A4) → reduced 5-HT reuptake → prolonged 5-HT signaling
IBS-C pathophysiology: [1,27]
- Reduced 5-HT4 receptor signaling → impaired prokinetic drive → slow transit
- Therapeutic target: 5-HT4 agonists (prucalopride) restore prokinetic function
Therapeutic implications: [27]
- 5-HT3 antagonists (alosetron, ondansetron): Block 5-HT3 → reduce pain, secretion; effective in IBS-D (limited availability due to ischaemic colitis risk with alosetron)
- 5-HT4 agonists (prucalopride, tegaserod): Enhance motility; effective in IBS-C
2. Intestinal Barrier Function and Permeability
Concept: Intestinal epithelium forms selective barrier; tight junctions (TJ) between enterocytes regulate permeability. [13,28]
IBS findings: [13,28]
- Increased intestinal permeability ("leaky gut") in subset of IBS patients (particularly post-infectious IBS)
- Mechanisms:
- Reduced expression of TJ proteins (occludin, claudin-1, ZO-1)
- Inflammatory mediators (TNF-α, IL-6, IL-8) disrupt TJ integrity
- Stress → corticotropin-releasing hormone (CRH) → mast cell degranulation → histamine, tryptase → TJ disruption
Consequences: [13,28]
- Increased luminal antigen exposure → immune activation → inflammation
- Bacterial translocation → low-grade endotoxemia
- Visceral hypersensitivity → amplified pain signaling
Evidence: [28]
- Lactulose/mannitol ratio test (permeability marker) elevated in 30-40% of IBS patients
- Confocal endomicroscopy shows disrupted TJ structure in IBS
Therapeutic targeting: [28]
- Probiotics (specific strains) may improve barrier function
- Glutamine, zinc supplementation under investigation
- Mast cell stabilisers (under research)
3. Mast Cell Activation and Neuroimmune Crosstalk
Mechanism: [13,29]
- Mast cells (immune cells) are increased in colonic mucosa of IBS patients, particularly near enteric nerves
- Triggers: Stress, food antigens, infections → mast cell degranulation
- Mediators released: Histamine, tryptase, serotonin, prostaglandins, cytokines (IL-6, TNF-α)
- Effect: Sensitisation of enteric nerves → visceral hypersensitivity, altered motility
Evidence: [13,29]
- Mast cell density correlates with IBS symptom severity
- Proximity of mast cells to enteric nerves predicts pain intensity
- Histamine levels elevated in IBS colonic mucosa
Therapeutic targeting: [29]
- Mast cell stabilisers: Ketotifen, sodium cromoglycate (limited evidence; under investigation)
- Antihistamines: May reduce abdominal pain in subset of patients
4. Bile Acid Dysregulation
Normal physiology: [23,30]
- Bile acid synthesis: Liver produces primary bile acids (cholic acid, chenodeoxycholic acid) from cholesterol
- Enterohepatic circulation: Bile acids secreted into duodenum → absorbed in terminal ileum (95% reabsorption) → returned to liver
- Regulation: FGF19 (ileal hormone) inhibits hepatic bile acid synthesis via negative feedback
IBS-D pathophysiology (Bile Acid Malabsorption - BAM): [23,30]
- Mechanism: Impaired ileal bile acid reabsorption → excess bile acids reach colon → stimulate colonic secretion, motility → diarrhoea
- Causes in IBS:
- "Primary (idiopathic): Reduced expression of ileal bile acid transporter (IBAT/ASBT)"
- "Increased hepatic bile acid synthesis: Low FGF19 levels → unregulated bile acid production"
- Prevalence: 25-30% of IBS-D patients have BAM [23,30]
Diagnostic approaches: [23,30]
- SeHCAT scan (75-selenium-homocholic acid taurine): Measures bile acid retention at 7 days; less than 15% = BAM (gold standard; limited availability in UK, not available USA)
- Serum 7α-hydroxy-4-cholesten-3-one (C4): Bile acid synthesis marker; elevated in BAM (research tool, not widely available)
- Empirical trial: Bile acid sequestrant (cholestyramine, colesevelam) for 2-4 weeks; therapeutic response confirms diagnosis
Management: [23,30]
- Bile acid sequestrants: Cholestyramine 4g OD-TDS, colesevelam 625mg BD; binds bile acids, reduces colonic stimulation
- Response rate: 60-70% in proven BAM
5. Gut Microbiome: Specific Mechanisms
Dysbiosis patterns in IBS: [13,31]
- Reduced diversity: Lower microbial richness (fewer bacterial species)
- Altered phyla: Increased Firmicutes:Bacteroidetes ratio (inconsistent across studies)
- Specific changes:
- "IBS-D: Reduced Bifidobacterium, increased Firmicutes; altered bile acid metabolism"
- "IBS-C: Increased methane-producing archaea (Methanobrevibacter smithii) → methane slows transit [24]"
- Functional changes: Altered production of short-chain fatty acids (SCFAs: acetate, propionate, butyrate), increased gas production (H₂, CH₄, CO₂)
Metabolomic studies: [31]
- IBS patients show altered faecal metabolites: reduced butyrate (anti-inflammatory SCFA), increased branched-chain fatty acids, altered tryptophan metabolism
- Butyrate deficiency: Impairs colonocyte energy metabolism, barrier function, anti-inflammatory signaling
Microbiota-gut-brain axis: [31]
- Microbial metabolites influence CNS via vagus nerve, immune signaling, neuroactive compounds
- SCFAs: Butyrate crosses blood-brain barrier, modulates neurotransmitters (GABA, serotonin)
- Tryptophan metabolism: Gut bacteria metabolise tryptophan → indoles, kynurenine → influence mood, pain perception
Probiotics: Strain-specific effects: [13,31]
- Effective strains (meta-analyses):
- "Bifidobacterium infantis 35624: Reduces pain, bloating, normalises IL-10/IL-12 ratio"
- Lactobacillus plantarum 299 v: Reduces bloating, improves gut transit
- "Multi-strain combinations (VSL#3): Mixed evidence; some benefit in IBS-D"
- Mechanisms: Restore microbial diversity, produce SCFAs, enhance barrier function, modulate immune response
- Evidence quality: Moderate; significant heterogeneity across studies; benefits modest
6. Genetic Susceptibility: Precision Medicine Approach
Heritability: [1,32]
- Twin studies: 25-30% heritability (genetic contribution)
- Familial clustering: 2-3x increased risk in first-degree relatives
Candidate genes (polymorphisms associated with IBS): [1,32]
- Serotonin pathway:
- "SLC6A4 (serotonin transporter): 5-HTTLPR polymorphism → altered 5-HT reuptake → IBS-D association"
- "HTR3 (5-HT3 receptor): Variants associated with IBS-D"
- Inflammatory/immune genes:
- "IL-6, TNF-α: Pro-inflammatory cytokine polymorphisms → increased inflammation"
- "TLR9 (Toll-like receptor 9): Innate immune recognition; variants associated with post-infectious IBS"
- Barrier function:
- "CDH1 (E-cadherin), TJP1 (ZO-1): Tight junction protein variants → increased permeability"
- Bile acid metabolism:
- "KLBH (β-Klotho): FGF19 receptor; variants associated with bile acid malabsorption in IBS-D"
Genome-wide association studies (GWAS): [32]
- Large GWAS (53,000 IBS cases, 433,000 controls) identified 6 genetic loci associated with IBS, including genes involved in ion channel function, neuronal signaling, and immune regulation
- Effect sizes small; IBS is polygenic (many genes, each with minor effect)
Clinical implication: [32]
- Genetic testing not yet clinically useful (predictive value low)
- Future: Precision medicine may enable subtype stratification, targeted therapy based on genetic/metabolomic signatures
7. Neurogastroenterology: Central and Peripheral Mechanisms
Enteric Nervous System (ENS): [1]
- "Second brain": 200-600 million neurons in gut wall; regulates motility, secretion, blood flow independently of CNS
- Neurotransmitters: Acetylcholine (excitatory), nitric oxide (inhibitory), vasoactive intestinal peptide (VIP), substance P (nociceptive)
Central sensitisation: [1,33]
- Definition: Amplification of pain signals in CNS; enhanced responsiveness of nociceptive neurons in spinal cord and brain
- Mechanisms:
- Increased excitability of dorsal horn neurons (spinal cord)
- Reduced descending inhibitory control from brainstem (pain modulation pathways)
- "Altered brain processing: fMRI studies show increased activation of pain-related regions (anterior cingulate cortex, insula, prefrontal cortex) during rectal distension"
- Clinical manifestation: Hyperalgesia (increased pain from painful stimulus), allodynia (pain from non-painful stimulus)
Hypothalamic-Pituitary-Adrenal (HPA) axis dysregulation: [14,33]
- Normal stress response: Hypothalamus → CRH → pituitary → ACTH → adrenal cortex → cortisol
- IBS patients: Exaggerated HPA axis response to stress; dysregulated cortisol rhythms
- Effects on gut:
- CRH increases gut permeability, motility, visceral sensitivity
- Cortisol modulates immune function, inflammation
- Bidirectional: Gut inflammation/dysbiosis → vagal afferents → brain → HPA activation → further gut dysfunction
Brain-gut axis imaging studies: [33]
- Structural changes: IBS patients show reduced grey matter density in pain-processing regions
- Functional changes: Altered connectivity between prefrontal cortex (cognitive control), limbic system (emotion), and brainstem (autonomic regulation)
- Implication: IBS involves central nervous system dysfunction, not just peripheral gut abnormality
Emerging Biomarkers and Diagnostic Tools
Faecal biomarkers: [34]
- Calprotectin: Validated to exclude IBD (less than 50 μg/g); 95% NPV for IBD
- Lactoferrin: Similar to calprotectin; less widely used
- Volatile organic compounds (VOCs): Breath/faecal VOCs differ in IBS vs healthy controls; experimental stage
- Faecal microbiome profiling: Not yet clinically validated; research tool
Serum biomarkers: [34]
- Anti-CdtB antibodies: Elevated in post-infectious IBS-D; marker of immune-mediated IBS (research stage)
- Anti-vinculin antibodies: Associated with IBS-M; experimental
- FGF19: Low levels correlate with bile acid malabsorption; not widely available
Future diagnostics: [34]
- Metabolomics: Faecal/serum metabolite profiling to identify IBS subtypes
- Genetic/epigenetic markers: Stratify patients by molecular signature
- AI-assisted diagnosis: Machine learning algorithms integrating clinical, microbiome, metabolomic data
Subtypes: Pathophysiological Differences
| Subtype | Predominant Pathophysiology |
|---|---|
| IBS-D | Rapid colonic transit, bile acid malabsorption (subset), microbiome dysbiosis, post-infectious inflammation |
| IBS-C | Slow colonic transit, pelvic floor dysfunction (subset), methane-producing archaea (Methanobrevibacter smithii) |
| IBS-M | Fluctuating motility, visceral hypersensitivity dominant |
| Post-infectious IBS | Persistent inflammation, immune activation, microbiome changes |
Detailed Subtype Comparison
IBS-D (Diarrhoea-Predominant)
Clinical characteristics: [1,2]
- Loose, watery stools (\u003e25% Bristol 6-7)
- Urgency (compelling need to defecate); often post-prandial
- Faecal incontinence in severe cases
- May have nocturnal diarrhoea (unusual; suggests organic disease if new)
Pathophysiological mechanisms: [1,13]
- Rapid colonic transit: Accelerated gut motility reduces water absorption
- Bile acid malabsorption (BAM): 25-30% of IBS-D have BAM; bile acids stimulate colonic secretion and motility [23]
- Microbiome dysbiosis: Reduced Bifidobacterium, increased Firmicutes; altered bile acid metabolism [13]
- 5-HT (serotonin) dysregulation: Increased post-prandial 5-HT release → faster transit [1]
- Post-infectious: 10-15% develop IBS-D after gastroenteritis; persistent low-grade inflammation [16]
Therapeutic approach: [3,6]
- Dietary: Low FODMAP diet; reduce caffeine, alcohol, high-fat meals
- First-line: Loperamide 2-4mg PRN; antispasmodics (peppermint oil, mebeverine)
- Second-line: Bile acid sequestrant (cholestyramine, colesevelam) if refractory; trial for 2-4 weeks [3]
- Third-line: Rifaximin (non-absorbable antibiotic; modulates microbiome) — used in USA, limited UK availability [23]
- Neuromodulators: TCAs (amitriptyline) — anticholinergic effect slows transit; beneficial in IBS-D [6]
Clinical pearl: In refractory IBS-D, consider bile acid malabsorption (BAM). SeHCAT scan (if available) shows \u003c15% retention at 7 days. Empirical trial of bile acid sequestrant is both diagnostic and therapeutic. [3,23]
IBS-C (Constipation-Predominant)
Clinical characteristics: [1,2]
- Hard, lumpy stools (\u003e25% Bristol 1-2)
- Straining, incomplete evacuation
- Infrequent bowel movements (\u003c3/week)
- Abdominal pain often relieved by defecation
Pathophysiological mechanisms: [1,13]
- Slow colonic transit: Reduced motility → prolonged stool retention → increased water absorption → hard stools
- Pelvic floor dysfunction (dyssynergic defecation): 30-40% of IBS-C; paradoxical anal sphincter contraction during defecation [3]
- Methane production: Methanobrevibacter smithii (archaea) produces methane → slows transit; associated with constipation [24]
- Serotonin deficiency: Reduced 5-HT4 receptor signaling → impaired prokinetic drive [1]
- Visceral hypersensitivity: Distension from retained stool → bloating, pain
Therapeutic approach: [3,6]
- Dietary: Soluble fibre (ispaghula/psyllium, oats); avoid insoluble fibre (bran) if bloating severe. Low FODMAP diet.
- First-line: Osmotic laxative — macrogol (polyethylene glycol) 1-2 sachets daily; well-tolerated
- Second-line: Secretagogues — linaclotide 290mcg OD (guanylate cyclase-C agonist; increases intestinal secretion, accelerates transit) [19,25]
- Second-line: Prokinetic — prucalopride 2mg OD (5-HT4 agonist) [3]
- If pelvic floor dysfunction: Biofeedback therapy (pelvic floor retraining); anorectal manometry + balloon expulsion test for diagnosis [3]
- Neuromodulators: SSRIs (sertraline, citalopram) — prokinetic effect; beneficial in IBS-C with anxiety/depression [6]
Clinical pearl: If IBS-C refractory to laxatives, assess for pelvic floor dysfunction. Anorectal manometry shows paradoxical anal contraction. Biofeedback therapy is effective. [3]
IBS-M (Mixed)
Clinical characteristics: [1,2]
- Alternating constipation and diarrhoea (\u003e25% both Bristol 1-2 and 6-7)
- Fluctuating stool pattern over days to weeks
- Pain and bloating prominent
Pathophysiological mechanisms: [1]
- Fluctuating motility: Variable colonic transit; rapid → diarrhoea; slow → constipation
- Visceral hypersensitivity dominant: Central pain amplification; symptoms disproportionate to motility changes
Therapeutic approach: [3,6]
- Challenge: Cannot use constipating or pro-diarrhoeal agents consistently
- Focus on pain/bloating: Antispasmodics (peppermint oil, mebeverine), low FODMAP diet
- Neuromodulators: TCAs (low-dose amitriptyline) or SSRIs depending on predominant subtype at time
- Psychological therapy: CBT, gut-directed hypnotherapy — particularly effective in IBS-M due to visceral hypersensitivity [7,8]
IBS-U (Unsubtyped)
Definition: Meets Rome IV criteria but stool pattern does not fit IBS-C, IBS-D, or IBS-M. [2]
Management: Symptom-directed; same as IBS-M. [3]
Post-Infectious IBS (PI-IBS)
Definition: IBS developing after acute gastroenteritis (bacterial, viral, or parasitic). [16]
Incidence: 10-15% of patients with infectious gastroenteritis develop PI-IBS. [16]
Risk factors: [16]
- Female sex
- Younger age
- Severity of infection (prolonged diarrhoea, fever)
- Antibiotic use during infection
- Psychological stress at time of infection
Pathophysiology: [13,16]
- Persistent low-grade inflammation: Increased mast cells, activated T-cells in gut mucosa
- Microbiome changes: Persistent dysbiosis after infection
- Increased intestinal permeability: Disrupted tight junctions
- Visceral hypersensitivity: Inflammatory mediators sensitise enteric nerves
Clinical course: [16]
- Often IBS-D subtype
- May improve spontaneously over 6-12 months in some patients
- 50% still symptomatic at 6 years
Management: Same as IBS-D; some evidence for probiotics in PI-IBS, but inconsistent. [3,13]
4. Clinical Presentation
Rome IV Diagnostic Criteria
Definition: Recurrent abdominal pain, on average, at least 1 day per week in the last 3 months, associated with two or more of the following: [2]
- Related to defecation (may increase or decrease pain)
- Associated with change in frequency of stool
- Associated with change in form (appearance) of stool
Timing: Criteria fulfilled for the last 3 months with symptom onset at least 6 months prior to diagnosis. [2]
Rome IV vs Rome III: Rome IV requires pain at least 1 day/week (previously 3 days/month), making criteria more restrictive and reducing prevalence. [2,11]
IBS Subtypes (Bristol Stool Scale)
Subtyping is based on stool form on days when patient has abnormal bowel movements: [2]
| Subtype | Definition (Bristol Stool Scale) |
|---|---|
| IBS-C (Constipation-predominant) | >25% of stools Bristol type 1-2 (hard/lumpy) AND less than 25% Bristol type 6-7 (loose/watery) |
| IBS-D (Diarrhoea-predominant) | >25% of stools Bristol type 6-7 (loose/watery) AND less than 25% Bristol type 1-2 (hard/lumpy) |
| IBS-M (Mixed) | >25% of stools Bristol type 1-2 AND >25% Bristol type 6-7 |
| IBS-U (Unsubtyped) | Meets Rome IV criteria but stool pattern does not fit IBS-C, IBS-D, or IBS-M |
Note: Subtypes can change over time; re-evaluation may be needed. [2]
Typical Symptoms
| Symptom | Characteristics | Prevalence |
|---|---|---|
| Abdominal pain | Crampy, lower abdomen (left iliac fossa, suprapubic); relieved by defecation; worse after eating | 100% (diagnostic criterion) [2] |
| Bloating | Subjective sensation of abdominal fullness; worse by evening; often relieved overnight | 80-90% [1] |
| Visible distension | Objective increase in abdominal girth; may be disproportionate to gas volume | 50-60% [1] |
| Altered bowel habit | Diarrhoea, constipation, or alternating; urgency; incomplete evacuation | 100% (diagnostic criterion) [2] |
| Mucus in stool | Passage of clear/white mucus with stool; benign finding | 50% [1] |
| Urgency | Sudden compelling need to defecate; common in IBS-D | 60-70% in IBS-D [1] |
| Incomplete evacuation | Sensation of incomplete rectal emptying; common in IBS-C | 50-60% in IBS-C [1] |
| Straining | Difficulty passing stool; common in IBS-C | 50-60% in IBS-C [1] |
Symptom Patterns
Temporal patterns: [1]
- Chronic, relapsing-remitting course
- Symptoms fluctuate over days to weeks
- Often worse during periods of stress
- May improve during holidays/relaxation
- Nocturnal symptoms (waking from sleep) are atypical and warrant investigation [3]
Meal-related: [1]
- Postprandial pain and urgency common (exaggerated gastrocolic reflex)
- Symptoms often worse after large meals or high-FODMAP foods
Psychological: [14]
- Symptoms exacerbate with stress, anxiety, depression
- Bidirectional relationship: symptoms worsen mood; poor mood worsens symptoms
Red Flags (Exclude Organic Disease)
[!CAUTION] Red Flags Requiring Investigation
- Age >50 years with new symptoms (risk of colorectal cancer)
- Unintentional weight loss (malignancy, IBD, coeliac disease)
- Rectal bleeding (colorectal cancer, IBD, polyps) — excludes blood on tissue from haemorrhoids
- Iron deficiency anaemia (malignancy, coeliac disease, IBD)
- Family history of colorectal cancer or inflammatory bowel disease (especially first-degree relative, age less than 50)
- Nocturnal symptoms waking patient from sleep (IBD, malignancy)
- Progressive symptoms (worsening over weeks to months)
- Palpable abdominal or rectal mass (malignancy)
- Unexplained fever (IBD, infection)
- Severe diarrhoea (>6 loose stools/day, dehydration)
Action: Any red flag mandates investigation with colonoscopy, imaging, and appropriate laboratory tests before diagnosing IBS. [3]
Extraintestinal Symptoms
IBS frequently coexists with other functional somatic syndromes: [1,14]
- Gynaecological: Dysmenorrhoea, chronic pelvic pain (overlap with endometriosis)
- Urological: Urgency, frequency, nocturia, interstitial cystitis
- Musculoskeletal: Fibromyalgia, chronic fatigue syndrome
- Neurological: Migraine, tension headache
- Psychiatric: Anxiety, depression, somatisation disorder
Clinical implication: High symptom burden and psychological comorbidity predict poorer response to therapy; multidisciplinary approach recommended. [14]
Differential Diagnosis
IBS is a diagnosis of exclusion. Consider: [3]
Inflammatory/Structural Bowel Disease
| Condition | Key Features | Investigation | Distinguishing Points |
|---|---|---|---|
| Inflammatory bowel disease (Crohn's, UC) | Weight loss, blood in stool, nocturnal symptoms, raised inflammatory markers | Faecal calprotectin (\u003e100 μg/g), colonoscopy with biopsy, imaging (MR enterography) | CRP/ESR elevated, faecal calprotectin raised, endoscopic/histological inflammation [3] |
| Microscopic colitis (collagenous, lymphocytic) | Older age (\u003e50), chronic watery diarrhoea, normal colonoscopy | Colonoscopy with biopsies (essential even if normal mucosa) | Normal endoscopy; diagnosis requires histology showing lymphocytic infiltrate or subepithelial collagen band [3] |
| Coeliac disease | Diarrhoea, bloating, weight loss, iron deficiency anaemia | Tissue transglutaminase IgA + total IgA; if positive → duodenal biopsy | TTG-IgA positive; villous atrophy on duodenal biopsy; HLA-DQ2/DQ8 positive [3,4] |
| Colorectal cancer | Age \u003e50, rectal bleeding, weight loss, change in bowel habit (\u003c3 months), anaemia | Colonoscopy ± CT colonography | Red flags present; mass on imaging/endoscopy [3] |
Functional/Motility Disorders
| Condition | Key Features | Investigation | Distinguishing Points |
|---|---|---|---|
| Bile acid malabsorption (BAM) | Chronic watery diarrhoea (often urgency, post-prandial), IBS-D refractory to first-line therapy | SeHCAT scan (\u003c15% retention at 7 days); empirical trial bile acid sequestrant | 25-30% of IBS-D have BAM; responds to cholestyramine/colesevelam [3,23] |
| Small intestinal bacterial overgrowth (SIBO) | Bloating (severe), diarrhoea, weight loss, flatulence | Hydrogen/methane breath test; aspirate culture (gold standard, rarely done) | Controversial overlap with IBS; evidence for treating SIBO in IBS is weak [3] |
| Lactose intolerance | Bloating, diarrhoea after dairy; ethnic predisposition (African, Asian) | Lactose hydrogen breath test; empirical lactose-free diet (2 weeks) | Symptom improvement with lactose avoidance; breath test positive [3] |
| Chronic idiopathic constipation | Constipation without pain; does not meet Rome IV criteria for IBS | Colonic transit study (if severe); anorectal manometry (if pelvic floor dysfunction) | Pain not predominant; treated similarly to IBS-C [3] |
Endocrine/Metabolic
| Condition | Key Features | Investigation | Distinguishing Points |
|---|---|---|---|
| Hyperthyroidism | Diarrhoea, weight loss, palpitations, heat intolerance | TSH (suppressed), free T4/T3 (elevated) | Systemic features; abnormal thyroid function tests [3] |
| Hypothyroidism | Constipation, weight gain, fatigue, cold intolerance | TSH (elevated), free T4 (low) | Systemic features; abnormal thyroid function tests [3] |
| Diabetes mellitus (autonomic neuropathy) | Diarrhoea (often nocturnal), constipation, long-standing diabetes | HbA1c, glucose; autonomic function tests | Known diabetes; other autonomic features (orthostatic hypotension, gastroparesis) [3] |
Malabsorption/Pancreatic
| Condition | Key Features | Investigation | Distinguishing Points |
|---|---|---|---|
| Pancreatic insufficiency | Steatorrhoea (pale, floating, foul-smelling stools), weight loss | Faecal elastase (\u003c200 μg/g); imaging (CT/MRI pancreas) | Weight loss, steatorrhoea; low faecal elastase; chronic pancreatitis or pancreatic cancer [3] |
| Small bowel Crohn's disease | Weight loss, abdominal pain, diarrhoea, mouth ulcers, perianal disease | MR enterography, capsule endoscopy, inflammatory markers | Weight loss, raised CRP; small bowel inflammation on imaging [3] |
Gynaecological (Women)
| Condition | Key Features | Investigation | Distinguishing Points |
|---|---|---|---|
| Endometriosis | Pelvic pain (cyclic, related to menses), dysmenorrhoea, dyspareunia | Pelvic ultrasound, MRI pelvis, laparoscopy (gold standard) | Cyclical pain; pelvic tenderness; imaging shows endometriotic deposits [3] |
| Ovarian cancer | Bloating, pelvic pain, urinary symptoms, early satiety; age \u003e50 | CA-125, pelvic ultrasound | Persistent symptoms \u003c1 year; raised CA-125; pelvic mass on imaging [3] |
Medication-Induced
- Opioids: Constipation (chronic use)
- Proton pump inhibitors (PPIs): Diarrhoea (microscopic colitis association), SIBO
- Antibiotics: Diarrhoea (microbiome disruption)
- Metformin: Diarrhoea (osmotic effect)
- NSAIDs: Diarrhoea, small bowel ulceration
- SSRIs: Diarrhoea (prokinetic effect)
Clinical approach to differentials: [3]
- First-line tests (all patients): FBC, CRP, TTG-IgA + total IgA, faecal calprotectin (IBS-D)
- Red flags: Colonoscopy ± imaging
- Refractory IBS-D: Consider BAM (trial bile acid sequestrant), SIBO (breath test), microscopic colitis (colonoscopy with biopsy)
- Refractory IBS-C: Anorectal manometry (pelvic floor dysfunction), colonic transit study (slow transit constipation)
5. Clinical Examination
General Principles
Physical examination in IBS is typically normal. The purpose is to exclude organic disease, identify red flags, and reassure the patient. [3]
Examination Findings
| Component | Typical Finding in IBS | Red Flags |
|---|---|---|
| General inspection | Well-appearing, normal weight | Cachexia, pallor (anaemia) |
| Vital signs | Normal | Fever (IBD, infection), tachycardia (severe diarrhoea, dehydration) |
| Hands | Normal | Clubbing (IBD, coeliac disease), koilonychia (iron deficiency) |
| Mouth | Normal | Aphthous ulcers (IBD, coeliac disease) |
| Abdomen inspection | Normal or mild distension | Visible distension (obstruction, ascites), scars (previous surgery) |
| Abdominal palpation | Soft, non-tender or mild lower abdominal tenderness | Guarding, rigidity, rebound (peritonitis), palpable mass (malignancy, IBD) |
| Percussion | Normal (resonant over gas-filled bowel) | Shifting dullness (ascites) |
| Auscultation | Normal bowel sounds | Absent (obstruction), high-pitched (obstruction) |
| Rectal examination | Normal; stool may be present | Blood on glove (malignancy, IBD), palpable mass (rectal cancer), impacted stool (severe constipation) |
| Perianal inspection | Normal | Skin tags, fissures, fistulae (Crohn's disease) |
Key Examination Steps
- Weight: Document baseline; unintentional weight loss is red flag [3]
- Abdominal examination: Inspect, palpate (masses, tenderness), percuss (ascites), auscultate
- Rectal examination: Mandatory if red flags (bleeding, age >50, family history); assess for masses, blood [3]
- Extraintestinal examination: If symptoms suggest (e.g., joints in suspected IBD)
Clinical Significance of Normal Examination
A normal examination in a patient meeting Rome IV criteria, without red flags, supports IBS diagnosis and avoids unnecessary investigation. [3] Over-investigation can reinforce illness behaviour and increase patient anxiety. [3]
6. Investigations
General Principles
IBS is a clinical diagnosis based on Rome IV criteria. Investigations are performed to exclude organic disease, not to confirm IBS. [3]
Key principle: In young patients (less than 50 years) with typical IBS symptoms and no red flags, limited investigations are appropriate. [3] Over-investigation is costly, anxiety-provoking, and may lead to incidental findings requiring further work-up. [3]
First-Line Investigations (All Patients)
Recommended for all patients meeting Rome IV criteria: [3,4]
| Test | Purpose | Interpretation |
|---|---|---|
| Full blood count (FBC) | Exclude anaemia (malignancy, coeliac disease, IBD) | Normal in IBS; low Hb or MCV warrants investigation |
| C-reactive protein (CRP) or ESR | Exclude inflammation (IBD, infection) | Normal in IBS (less than 5 mg/L); elevated suggests IBD or infection |
| Tissue transglutaminase IgA (TTG-IgA) + total IgA | Exclude coeliac disease | TTG-IgA positive + normal total IgA → likely coeliac; confirm with duodenal biopsy. IgA deficiency → check TTG-IgG or EMA-IgG |
| Faecal calprotectin (if diarrhoea-predominant) | Exclude IBD | less than 50 μg/g: IBD unlikely; 50-100 μg/g: equivocal (repeat or investigate if clinical concern); >100 μg/g: IBD likely, refer for colonoscopy [3] |
NICE/BSG recommendation: These tests have high yield and are cost-effective. [3,4]
Second-Line Investigations (If Red Flags or Atypical Features)
| Test | Indication | Interpretation |
|---|---|---|
| Colonoscopy ± biopsy | Age >50 with new symptoms, rectal bleeding, family history colorectal cancer/IBD, raised calprotectin, anaemia | Rule out malignancy, IBD, microscopic colitis (requires biopsy even if normal mucosa) |
| Thyroid function (TSH, free T4) | Hyperthyroidism (diarrhoea), hypothyroidism (constipation) | Abnormal TSH warrants treatment; recheck symptoms after correction |
| Stool microscopy, culture, ova, cysts, parasites | Diarrhoea-predominant IBS, travel history, infectious symptoms | Exclude Giardia, Cryptosporidium, bacterial infection |
| Faecal elastase | Weight loss, steatorrhoea (pale, floating stools) | less than 200 μg/g suggests pancreatic insufficiency |
| Hydrogen/methane breath test | Suspected lactose intolerance, SIBO (if bloating predominant) | Positive lactose breath test → trial lactose-free diet; SIBO evidence controversial [3] |
| SeHCAT scan (if available) | IBS-D refractory to first-line therapy; suspect bile acid malabsorption | less than 15% retention at 7 days indicates BAM; trial bile acid sequestrant |
Third-Line/Specialist Investigations
| Test | Indication | Notes |
|---|---|---|
| CT/MRI abdomen-pelvis | Abdominal mass, unexplained weight loss, suspected Crohn's disease | Excludes malignancy, IBD (MR enterography preferred for small bowel) |
| Small bowel capsule endoscopy | Suspected small bowel pathology (Crohn's, tumour) not visible on CT/MRI | Specialist investigation |
| Colonic transit study | Severe IBS-C refractory to laxatives; consider pelvic floor dysfunction | Sitz marker study or wireless motility capsule |
| Anorectal manometry + balloon expulsion | Suspected pelvic floor dysfunction (dyssynergic defecation) in IBS-C | May guide biofeedback therapy |
What NOT to Do
Avoid over-investigation: [3]
- Routine imaging (CT, ultrasound) in typical IBS without red flags is not indicated
- Repeated colonoscopy without new symptoms or red flags is not indicated
- Extensive allergy testing (IgG food panels) is not evidence-based and should be avoided
- Routine SIBO breath testing is controversial; not recommended by major guidelines [3]
Clinical pearl: Over-investigation can increase patient anxiety, reinforce illness behaviour, and lead to iatrogenic harm from incidental findings. Confident clinical diagnosis and reassurance are therapeutic. [3]
7. Management
General Principles
IBS management is multifaceted, individualised, and symptom-directed. No single therapy works for all patients. Goals are: [3]
- Establish therapeutic relationship: Validate symptoms, explain gut-brain interaction
- Reassure: IBS does not progress to IBD or cancer
- Set realistic expectations: Aim for symptom control, not cure; chronic relapsing-remitting condition
- Tailor to predominant symptom: Pain/bloating, constipation, diarrhoea
- Address psychological comorbidity: Screen for anxiety, depression; consider psychological therapy
Evidence base: NICE, BSG, ACG, and Rome Foundation guidelines provide evidence-based recommendations. [3,4,6]
Management Algorithm
ROME IV DIAGNOSIS CONFIRMED, RED FLAGS EXCLUDED
↓
┌──────────────────────────────────────────────────────────────┐
│ PATIENT EDUCATION & REASSURANCE │
│ - Explain IBS: functional disorder, not dangerous │
│ - Gut-brain axis: stress-symptom link │
│ - Chronic, relapsing-remitting; does not → IBD/cancer │
│ - Set realistic goals: symptom control, not cure │
└──────────────────────────────────────────────────────────────┘
↓
┌──────────────────────────────────────────────────────────────┐
│ LIFESTYLE & DIETARY ADVICE │
│ - Regular meals (avoid skipping), adequate hydration │
│ - Limit caffeine, alcohol, fizzy drinks │
│ - Fibre: soluble fibre (ispaghula) if constipation; │
│ avoid insoluble fibre (bran) if bloating │
│ - Identify trigger foods: food diary │
│ - Regular physical activity (30 min/day, 5 days/week) │
└──────────────────────────────────────────────────────────────┘
↓
┌──────────────────────────────────────────────────────────────┐
│ LOW FODMAP DIET (Dietitian-led) │
│ - Elimination phase: 4-8 weeks strict low FODMAP │
│ - Reintroduction: Systematic challenge to identify triggers │
│ - Personalisation: Long-term modified diet based on │
│ individual tolerance │
│ - Response rate: 50-80% achieve symptom improvement [5,15] │
└──────────────────────────────────────────────────────────────┘
↓
┌──────────────────────────────────────────────────────────────┐
│ SYMPTOM-DIRECTED PHARMACOTHERAPY (First-line) │
├──────────────────────────────────────────────────────────────┤
│ PAIN / BLOATING: │
│ - Antispasmodics: mebeverine 135mg TDS, hyoscine butyl- │
│ bromide 10-20mg TDS, peppermint oil 1-2 caps TDS │
│ │
│ IBS-C (CONSTIPATION): │
│ - Osmotic laxative: macrogol (Movicol) 1-2 sachets daily │
│ - Avoid stimulant laxatives (senna) long-term │
│ │
│ IBS-D (DIARRHOEA): │
│ - Loperamide 2-4mg PRN (after loose stool, max 16mg/day) │
│ - Avoid prophylactic use; risk of constipation rebound │
└──────────────────────────────────────────────────────────────┘
↓
IF INADEQUATE RESPONSE
↓
┌──────────────────────────────────────────────────────────────┐
│ SYMPTOM-DIRECTED PHARMACOTHERAPY (Second-line) │
├──────────────────────────────────────────────────────────────┤
│ REFRACTORY IBS-C: │
│ - Linaclotide 290mcg OD (take 30 min before breakfast) │
│ Guanylate cyclase-C agonist; secretagogue [19] │
│ - Prucalopride 2mg OD (5-HT4 agonist; prokinetic) │
│ │
│ REFRACTORY IBS-D: │
│ - Bile acid sequestrant: cholestyramine 4g OD-TDS, │
│ colesevelam 625mg BD (if suspected BAM) [3] │
│ - Eluxadoline 100mg BD (μ-opioid agonist/δ-antagonist) │
│ Caution: pancreatitis risk; contraindicated if no │
│ gallbladder, alcohol excess, pancreatitis history │
│ │
│ PAIN-PREDOMINANT IBS (any subtype): │
│ - Tricyclic antidepressant (TCA): amitriptyline 10mg │
│ nocte, titrate to 30mg (max 50mg). Start low, go slow. │
│ Visceral analgesic effect, not antidepressant. [6,20] │
│ NNT = 4 for symptom improvement [6] │
│ - SSRI (if anxiety/depression comorbid or TCA not │
│ tolerated): sertraline 50mg OD, citalopram 20mg OD [6] │
└──────────────────────────────────────────────────────────────┘
↓
IF INADEQUATE RESPONSE OR
PSYCHOLOGICAL COMORBIDITY
↓
┌──────────────────────────────────────────────────────────────┐
│ PSYCHOLOGICAL THERAPIES (Evidence-based) │
│ - Cognitive Behavioural Therapy (CBT): 6-12 sessions; │
│ addresses maladaptive thoughts, coping strategies. │
│ NNT = 4 for global symptom improvement [7,8] │
│ - Gut-directed Hypnotherapy: 7-12 sessions; induces │
│ relaxation, modulates visceral perception. Efficacy │
│ equivalent to CBT; long-term benefit. [7,8,21] │
│ - Mindfulness-based therapy: Reduces stress, improves │
│ quality of life [8] │
│ - Psychodynamic/interpersonal therapy: Limited evidence │
│ │
│ Referral criteria: Moderate-severe symptoms, inadequate │
│ response to pharmacotherapy, significant anxiety/depression │
└──────────────────────────────────────────────────────────────┘
Dietary Management
General Dietary Advice (All Patients)
Evidence-based recommendations: [3,5]
- Regular meals: Eat 3 meals/day; avoid skipping meals or grazing
- Adequate hydration: 1.5-2 litres water/day
- Limit triggers:
- "Caffeine (coffee, tea, energy drinks): exacerbates urgency, anxiety"
- "Alcohol: gut irritant; worsens diarrhoea"
- "Fizzy drinks: gas production → bloating"
- Fibre modification:
- "Soluble fibre (ispaghula/psyllium, oats, linseeds): beneficial in IBS-C; softens stool [3]"
- "Insoluble fibre (bran, wholegrain wheat): may worsen bloating; avoid if bloating predominant [3]"
- Eat slowly: Reduces aerophagia (swallowed air) → less bloating
Low FODMAP Diet
FODMAP: Fermentable Oligosaccharides (fructans, GOS), Disaccharides (lactose), Monosaccharides (fructose), And Polyols (sorbitol, mannitol). [5,15]
Rationale: High FODMAP foods are poorly absorbed, rapidly fermented by gut bacteria, and osmotically active → gas, bloating, pain, altered bowel habit. [5,15]
Evidence: Meta-analysis of RCTs shows low FODMAP diet reduces global IBS symptoms in 50-80% of patients. [5,15] Efficacy comparable to first-line pharmacotherapy. [5]
Implementation: [5,15]
Phase 1: Elimination (4-8 weeks)
- Strict avoidance of high FODMAP foods
- Dietitian-supervised for adherence and nutritional adequacy
Phase 2: Reintroduction (6-8 weeks)
- Systematic challenge with individual FODMAP groups (e.g., lactose week 1, fructose week 2, etc.)
- Identify personal triggers
Phase 3: Personalisation (long-term)
- Liberalised diet avoiding only proven triggers
- Prevents unnecessary dietary restriction and maintains microbiome diversity
High FODMAP foods to avoid (elimination phase): [5,15]
- Oligosaccharides: Onion, garlic, wheat, rye, legumes (beans, lentils)
- Disaccharides: Milk, yogurt, soft cheese (lactose)
- Monosaccharides: Apples, pears, mango, honey, high-fructose corn syrup
- Polyols: Stone fruits (peaches, plums, cherries), mushrooms, cauliflower, artificial sweeteners (sorbitol, mannitol, xylitol)
Low FODMAP alternatives: [5,15]
- Grains: Rice, oats, quinoa, gluten-free bread
- Dairy: Lactose-free milk, hard cheese, butter
- Protein: Meat, fish, eggs, tofu
- Fruit: Banana, blueberries, strawberries, oranges, grapes
- Vegetables: Carrot, cucumber, lettuce, tomato, potato, courgette
Key principle: FODMAP diet requires dietitian supervision. Self-directed diets often fail due to poor adherence or overly restrictive approach. [5,15]
Pharmacotherapy
Antispasmodics (Pain/Bloating)
| Drug | Dose | Mechanism | Evidence | Side Effects |
|---|---|---|---|---|
| Mebeverine | 135mg TDS (before meals) | Smooth muscle relaxant | Cochrane review: modest benefit for pain; NNT ~5 [3] | Generally well-tolerated; rare hypersensitivity |
| Hyoscine butylbromide | 10-20mg TDS-QDS PRN | Anticholinergic (blocks muscarinic receptors) | Reduces cramping; short-acting | Dry mouth, blurred vision, urinary retention |
| Peppermint oil (enteric-coated) | 1-2 capsules TDS (before meals) | Calcium channel blocker; smooth muscle relaxant | Meta-analysis: effective for global IBS symptoms; NNT = 3-4 [22] | Heartburn (use enteric-coated), perianal burning |
Clinical use: [3]
- First-line for pain and bloating
- Peppermint oil has best evidence but often underutilised [22]
- Can be used PRN or regularly; trial 4 weeks before switching
Laxatives (IBS-C)
| Drug | Dose | Mechanism | Evidence | Notes |
|---|---|---|---|---|
| Macrogol (polyethylene glycol) | 1-2 sachets daily | Osmotic laxative; retains water in bowel | First-line for IBS-C; well-tolerated [3] | Dissolve in water; adjust dose to response |
| Lactulose | 10-20ml BD | Osmotic (non-absorbable disaccharide) | Effective but causes bloating/flatulence; avoid in IBS [3] | Avoid: worsens bloating |
| Linaclotide | 290mcg OD (30 min before breakfast) | Guanylate cyclase-C agonist; increases intestinal secretion, accelerates transit | RCTs: effective for IBS-C; improves pain, stool frequency, quality of life. NNT = 6-7 [19] | Diarrhoea (10-20%); expensive; second-line |
| Prucalopride | 2mg OD | 5-HT4 agonist (prokinetic) | Licensed for chronic constipation; evidence in IBS-C [3] | Headache, nausea; second-line |
Clinical use: [3]
- Start macrogol; titrate dose
- If inadequate response after 4-6 weeks, consider linaclotide or prucalopride
- Avoid stimulant laxatives (senna, bisacodyl) long-term; may worsen pain
Antidiarrhoeals (IBS-D)
| Drug | Dose | Mechanism | Evidence | Notes |
|---|---|---|---|---|
| Loperamide | 2-4mg PRN after loose stool (max 16mg/day) | Opioid receptor agonist; slows transit, increases water absorption | Effective for diarrhoea; no effect on pain [3] | Use PRN, not prophylactically; risk of constipation rebound |
| Cholestyramine | 4g OD-TDS (before meals) | Bile acid sequestrant | Trial if suspected bile acid malabsorption (BAM) in refractory IBS-D [3,23] | Bloating, constipation; interferes with drug absorption (take other meds 1h before or 4h after) |
| Colesevelam | 625mg 1-3 tablets BD | Bile acid sequestrant | Better tolerated than cholestyramine [3,23] | Expensive; similar efficacy |
| Eluxadoline | 100mg BD (with food) | μ-opioid receptor agonist + δ-antagonist | RCTs: effective for IBS-D; reduces pain, urgency [3] | Pancreatitis risk (1-2%); contraindicated: no gallbladder, alcohol excess, pancreatitis history. Not widely available. |
| Rifaximin | 550mg TDS for 14 days | Non-absorbable antibiotic; modulates gut microbiome | TARGET trials: effective for IBS-D; reduces bloating, pain, loose stools. NNT = 10-11 [23,25] | USA FDA-approved; limited UK availability. Can repeat courses. |
Clinical use: [3]
- Loperamide first-line for diarrhoea; PRN use
- If refractory, trial bile acid sequestrant for 2-4 weeks
- Eluxadoline: specialist use only
- Rifaximin: see detailed section below
Rifaximin: Evidence and Clinical Use
Mechanism: [23,25]
- Non-absorbable antibiotic (rifamycin derivative)
- \u003c1% systemic absorption; acts locally in gut
- Modulates gut microbiome: reduces gas-producing bacteria, alters bile acid metabolism
- Anti-inflammatory effects: reduces mucosal cytokines
Evidence: [23,25,26]
- TARGET 1 \u0026 2 trials (RCTs, n=1260): Rifaximin 550mg TDS for 14 days vs placebo in IBS-D [23]
- "Primary endpoint: Adequate relief of global IBS symptoms"
- "Results: 40.8% rifaximin vs 31.2% placebo (NNT = 10-11)"
- "Benefit sustained: 10 weeks post-treatment"
- TARGET 3 trial: Repeat treatment effective in relapsers (up to 2 additional 14-day courses) [26]
- Meta-analysis (7 RCTs, n=1803): Rifaximin superior to placebo for global symptoms (RR 1.57, 95% CI 1.22-2.01); particularly effective for bloating [25]
Clinical use: [23,25]
- Indication: IBS-D refractory to dietary modification and loperamide
- Dose: 550mg TDS for 14 days
- Response: Assess at 4 weeks post-treatment
- Repeat courses: If symptoms recur, can repeat (up to 2 additional courses); efficacy maintained [26]
- Side effects: Minimal (non-absorbable); nausea (rare), headache
- Resistance: Not a concern (non-absorbable; does not select for systemic resistance)
Availability: [3,23]
- USA: FDA-approved for IBS-D (2015); widely used
- UK: Not routinely available on NHS; not in NICE/BSG guidelines
- Rationale for limited UK use: Modest NNT (10-11), expensive, antibiotic stewardship concerns
Clinical pearl: Rifaximin is particularly effective for bloating-predominant IBS-D. Consider if bloating is the most troublesome symptom despite dietary measures. [23,25]
Neuromodulators (Central-Acting Agents)
Definition: Medications that modulate central pain processing, gut-brain axis, and visceral hypersensitivity. [6,20]
Mechanism: [6,20]
- Tricyclic antidepressants (TCAs): Block reuptake of norepinephrine and serotonin; anticholinergic effects slow gut transit; central analgesic action (not antidepressant)
- SSRIs: Block serotonin reuptake; prokinetic effect via 5-HT4 activation; anxiolytic action
- SNRIs: Block norepinephrine and serotonin reuptake; central pain modulation
Evidence: [6,20]
- TCAs: Meta-analysis (12 RCTs, n=787): NNT = 4 for global symptom improvement; effective for pain-predominant IBS
- SSRIs: Meta-analysis (8 RCTs, n=514): NNT = 6-8; less effective than TCAs for pain; beneficial if anxiety/depression comorbid
- SNRIs: Limited evidence; duloxetine (60mg OD) shows promise in refractory IBS with pain
| Drug | Dose | Subtype | Evidence | Side Effects |
|---|---|---|---|---|
| Amitriptyline | 10mg nocte, titrate to 30mg (max 50mg) | IBS-D, pain-predominant | NNT = 4; gold standard TCA [6,20] | Dry mouth, constipation, drowsiness, weight gain. Caution: cardiac arrhythmia (baseline ECG if age >50, cardiac history) |
| Nortriptyline | 10-25mg nocte | IBS-D, pain-predominant | Similar efficacy to amitriptyline; fewer anticholinergic effects [6] | Less constipation than amitriptyline |
| Sertraline | 50mg OD | IBS-C with anxiety/depression | NNT = 6-8; prokinetic + anxiolytic [6] | Nausea (transient), diarrhoea, insomnia |
| Citalopram | 20mg OD | IBS-C with anxiety/depression | NNT = 6-8 [6] | Similar to sertraline |
| Duloxetine (SNRI) | 30-60mg OD | Refractory IBS with chronic pain | Small RCTs show benefit; not widely used [35] | Nausea, dry mouth, fatigue |
Clinical use: [6,20]
- First-line (pain): Amitriptyline 10mg nocte; titrate slowly (increase 10mg every 2-4 weeks) to minimise side effects
- Duration: Continue for 6-12 months if effective; attempt gradual withdrawal after symptom stabilisation
- Counselling: Explain analgesic mechanism (not treating depression); effects may take 2-4 weeks; start low, go slow
- TCAs: Favour in IBS-D (anticholinergic effect slows transit)
- SSRIs: Favour in IBS-C (prokinetic effect); particularly beneficial if anxiety/depression comorbid
Contraindications: [6]
- TCAs: Cardiac arrhythmia, recent MI, severe liver disease, urinary retention, narrow-angle glaucoma
- SSRIs/SNRIs: MAOI use (within 14 days), uncontrolled bleeding disorder
Emerging and Investigational Therapies
1. Linaclotide (Guanylate Cyclase-C Agonist)
Mechanism: [19,36]
- Activates guanylate cyclase-C receptors in intestinal epithelium → increased cyclic GMP → chloride/bicarbonate secretion → fluid secretion, accelerated transit
- Reduces visceral hypersensitivity via analgesic effect on colonic afferent neurons
Evidence: [19,36]
- Phase 3 RCTs (n=1604): Linaclotide 290mcg OD vs placebo in IBS-C
- "Primary endpoint: Composite (≥30% reduction in abdominal pain + ≥1 complete spontaneous bowel movement increase/week)"
- "Results: 33.6% linaclotide vs 21.0% placebo; NNT = 6-7"
- "Sustained benefit: 26 weeks"
- Cochrane review (2018): Linaclotide superior to placebo for global symptoms, pain, stool frequency, quality of life
Dosing: [19,36]
- IBS-C: 290mcg once daily, 30 minutes before breakfast, on empty stomach
- Onset: 1-2 weeks
Side effects: [19,36]
- Diarrhoea: 10-20% (dose-dependent; usually transient; improves with continued use)
- Abdominal pain: Mild, transient
- Bloating: Paradoxical worsening in some patients
Availability: [3,36]
- UK: Available; NICE does not recommend routine use on NHS (cost-effectiveness concerns); specialist prescription
- USA: FDA-approved (2012); widely used
Clinical use: [3,19,36]
- Indication: IBS-C refractory to macrogol, fibre; moderate-severe symptoms
- Cost-effectiveness: Expensive (£40-50/month UK); reserve for refractory cases
- Predictors of response: Severe bloating, pain-predominant IBS-C
2. Plecanatide (Guanylate Cyclase-C Agonist)
Mechanism: Similar to linaclotide; activates GC-C receptors → intestinal secretion, accelerated transit [37]
Evidence: [37]
- Phase 3 RCTs (n=2189): Plecanatide 3mg OD vs placebo in IBS-C
- "Response rate: 30% plecanatide vs 17% placebo (composite endpoint: pain + bowel movements)"
- "NNT: 7-8"
Availability: [37]
- USA: FDA-approved (2017); alternative to linaclotide
- UK/Europe: Not available
Comparison to linaclotide: [37]
- Similar efficacy and side effect profile
- Lower rate of severe diarrhoea (plecanatide 3mg vs linaclotide 290mcg)
3. Tenapanor (NHE3 Inhibitor)
Mechanism: [38]
- Inhibits sodium-hydrogen exchanger 3 (NHE3) in intestinal epithelium → reduces sodium absorption → water retention in lumen → softer stools, accelerated transit
- Minimally absorbed (less than 1%); local action
Evidence: [38]
- Phase 3 RCTs (n=1394): Tenapanor 50mg BD vs placebo in IBS-C
- "Response rate: 36.5% tenapanor vs 23.7% placebo"
- "NNT: 8"
- "Sustained benefit: 26 weeks"
Side effects: [38]
- Diarrhoea: 15-20% (less than linaclotide)
- Abdominal distension: Transient
Availability: [38]
- USA: FDA-approved (2019)
- UK/Europe: Not available
4. 5-HT3 Antagonists (Ondansetron, Alosetron)
Mechanism: [39]
- Block 5-HT3 receptors → reduce colonic transit, secretion, visceral sensation
- Particularly effective in IBS-D
Alosetron: [39]
- Evidence: Multiple RCTs show efficacy in IBS-D (NNT = 8-10)
- Availability: USA FDA-approved (women with severe IBS-D refractory to other therapy); restricted prescribing due to ischaemic colitis risk (1 in 1000)
- UK/Europe: Withdrawn due to safety concerns
Ondansetron: [39]
- Evidence: RCT (n=120): Ondansetron 4-8mg TDS vs placebo in IBS-D; improved stool consistency, urgency, frequency
- Off-label use: UK/Europe; not licensed for IBS but used in refractory IBS-D
- Dosing: Start 4mg OD-TDS; titrate to response (max 24mg/day in divided doses)
- Side effects: Constipation (dose-limiting; 20-30%), headache
- Safety: No ischaemic colitis risk (unlike alosetron)
Clinical use: [39]
- Indication: Severe IBS-D refractory to loperamide, dietary measures
- Monitoring: Assess stool frequency/consistency weekly; reduce dose if constipation develops
- Duration: Long-term use acceptable; no tachyphylaxis
5. Probiotics: Evidence-Based Recommendations
Mechanism: [13,31]
- Restore microbial diversity, produce SCFAs (butyrate), enhance barrier function, modulate immune response
Evidence summary: [31,40]
- Meta-analyses: Probiotics superior to placebo for global IBS symptoms (RR 0.79, 95% CI 0.72-0.87); NNT = 7-10
- Heterogeneity: High; benefits vary by strain, dose, formulation
- Effective strains:
- "Bifidobacterium infantis 35624 (Align®): RCT shows reduced pain, bloating, normalised IL-10/IL-12 ratio"
- Lactobacillus plantarum 299 v: RCT shows reduced bloating, improved pain
- "Saccharomyces boulardii: RCT shows benefit in IBS-D"
- "Multi-strain (VSL#3, Symprove): Mixed evidence; some benefit in IBS-D/IBS-M"
Clinical use: [3,31,40]
- Evidence quality: Moderate; benefits modest (10-20% absolute improvement over placebo)
- Trial approach: Consider in mild-moderate IBS; trial 4-8 weeks; continue if symptomatic benefit
- Strain-specific: Use evidence-based strains; avoid generic "probiotic" products without trial data
- Safety: Generally safe; rare case reports of bacteraemia in immunocompromised
NICE/BSG position: [3,4]
- NICE (2017): Probiotics may be trialled for 4 weeks; no specific strain recommended
- BSG (2021): Weak recommendation; certain strains (B. infantis, L. plantarum) may provide modest benefit
6. FMT (Faecal Microbiota Transplantation)
Rationale: [41]
- Microbiome dysbiosis is central to IBS pathophysiology; FMT aims to restore healthy microbiome
Evidence: [41]
- Systematic review (7 RCTs, n=471): FMT vs placebo in IBS
- "Heterogeneous results: 2 RCTs showed benefit; 5 showed no difference"
- "Overall: No consistent evidence of benefit"
- Challenges: Variability in donor microbiome, delivery method (colonoscopy vs capsule), dose, frequency
Clinical use: [3,41]
- Current recommendation: Not recommended for IBS (insufficient evidence)
- Research ongoing: Trials investigating "super-donors" (high engraftment donors), repeated dosing, specific microbiome signatures
7. Complementary and Alternative Medicine (CAM)
Acupuncture: [42]
- Evidence: Small RCTs and meta-analyses suggest modest benefit for global IBS symptoms; high risk of bias, placebo effect
- Mechanism: Uncertain; may modulate visceral sensitivity, autonomic function
- Clinical use: May offer benefit in motivated patients; low risk
Herbal therapies: [42]
- Peppermint oil: Evidence-based (see antispasmodics section); effective NNT = 3-4 [22]
- STW 5 (Iberogast®): German herbal preparation; RCTs show benefit for dyspepsia/IBS overlap; safety concerns (hepatotoxicity)
- Chinese herbal medicine: Limited high-quality RCTs; individualised formulations difficult to standardise
- Turmeric (curcumin): Small RCT (n=207) suggests benefit for IBS symptoms; anti-inflammatory effects
Clinical use: [42]
- Evidence quality: Low-moderate; peppermint oil has best evidence
- Safety: Herbal ≠ safe; potential drug interactions, hepatotoxicity (STW 5)
- Position: Discuss with patients; may trial peppermint oil; caution with other herbal preparations
8. Neuromodulation: Emerging Non-Pharmacological Therapies
Transcutaneous Electrical Nerve Stimulation (TENS): [43]
- Mechanism: Electrical stimulation of abdominal wall → modulates visceral pain via gate control theory
- Evidence: Small RCTs show modest benefit for abdominal pain; limited data
- Clinical use: Experimental; not widely used
Sacral Nerve Stimulation (SNS): [43]
- Mechanism: Electrical stimulation of S3 nerve root → modulates gut-brain signaling, pelvic floor function
- Evidence: Case series show benefit in severe refractory IBS-C with pelvic floor dysfunction; no large RCTs
- Clinical use: Experimental; specialist centres only
Vagal Nerve Stimulation (VNS): [43]
- Mechanism: Stimulation of vagus nerve → modulates gut-brain axis, inflammation
- Evidence: Preclinical; early-phase human trials ongoing
- Clinical use: Investigational only
8. Complications
Short-term Complications
IBS is a functional disorder; there are no direct medical complications leading to organ damage, cancer, or mortality. [9]
However, aggressive symptom management can cause iatrogenic complications: [3]
- Overuse of laxatives: Electrolyte disturbance (hypokalaemia with stimulant laxatives)
- Overuse of loperamide: Rebound constipation, rarely toxic megacolon (if very high dose)
- Eluxadoline: Pancreatitis (1-2% risk) [3]
Long-term Complications
Quality of Life Impairment
IBS significantly impairs health-related quality of life across all domains (physical, psychological, social, work). [10] Severity is comparable to chronic diseases such as IBD, diabetes, chronic pain syndromes. [10]
Domains affected: [10]
- Physical: Pain, fatigue, sleep disturbance
- Psychological: Anxiety, depression, poor self-image
- Social: Avoidance of social activities, travel, relationships due to fear of symptoms
- Work: Absenteeism (3x higher than controls), presenteeism (reduced productivity) [10]
Psychological Comorbidity
Up to 30% of IBS patients have comorbid anxiety or depression. [14] Bidirectional relationship: psychological distress worsens IBS symptoms; chronic IBS symptoms worsen psychological distress. [14]
Impact: [14]
- Poorer response to therapy
- Higher healthcare utilisation
- Greater disability
Management implication: Screen for anxiety/depression; treat concurrently with psychological therapy or SSRIs. [3,14]
Healthcare Utilisation and Economic Burden
IBS patients have: [10]
- 50% higher healthcare consultations
- 2-3x prescription costs
- Increased unnecessary investigations and surgeries (e.g., cholecystectomy for "biliary-type pain", appendicectomy)
Total economic burden (direct + indirect costs): [10]
- UK: £1-2 billion/year
- USA: $20-30 billion/year
Social and Occupational Impact
- Work absenteeism: 3x higher than matched controls [10]
- Unemployment: Some patients unable to maintain employment due to symptom severity
- Social isolation: Fear of symptoms (urgency, incontinence) leads to avoidance of travel, dining out, social events
Does IBS Progress to Organic Disease?
No. IBS does not increase risk of: [9]
- Inflammatory bowel disease (Crohn's disease, ulcerative colitis)
- Colorectal cancer
- Small bowel disease
- Mortality
Evidence: Long-term cohort studies (10-20 year follow-up) show no excess risk of structural bowel disease in IBS patients. [9]
Clinical implication: Reassure patients that IBS, while chronic and burdensome, is not dangerous and does not lead to cancer or serious disease. [3]
9. Prognosis and Outcomes
Natural History
IBS is a chronic, relapsing-remitting condition. [9]
Course: [9]
- Symptoms fluctuate over days, weeks, months
- Periods of exacerbation (often stress-related) alternate with periods of remission
- 30-50% of patients report spontaneous improvement over 5-10 years
- 10-20% report worsening symptoms
- 30-40% have stable symptoms
Factors predicting poor prognosis: [9,14]
- Severe baseline symptoms
- Comorbid anxiety, depression, somatisation
- Long symptom duration (>5 years)
- Catastrophising, poor coping strategies
- High healthcare utilisation
Factors predicting good prognosis: [9]
- Mild-moderate symptoms
- Good insight, adaptive coping
- Engagement with therapy (dietary, psychological)
- Absence of psychological comorbidity
Treatment Outcomes
Dietary Interventions
Low FODMAP diet: [5,15]
- Response rate: 50-80% achieve ≥50% reduction in symptoms
- Time to response: 2-4 weeks
- Predictors of response: Bloating predominant, short symptom duration, adherence to dietitian advice
Pharmacotherapy
| Therapy | Response Rate | NNT | Notes |
|---|---|---|---|
| Antispasmodics (mebeverine, peppermint oil) | 40-60% | 4-5 | Modest benefit; peppermint oil best evidence [22] |
| Laxatives (macrogol) | 60-70% in IBS-C | — | Well-tolerated; first-line IBS-C [3] |
| Linaclotide (IBS-C) | 50-60% | 6-7 | Effective but expensive; second-line [19] |
| Loperamide (IBS-D) | 60-80% for diarrhoea | — | No effect on pain [3] |
| Tricyclic antidepressants (amitriptyline) | 50-60% | 4 | Effective for pain; long-term use acceptable [6,20] |
| SSRIs | 40-50% | 6-8 | Less evidence than TCAs; useful if anxiety/depression [6] |
Key point: Pharmacotherapy is symptom control, not cure. Most patients require ongoing therapy; some can stop after achieving stability. [3]
Psychological Therapies
| Therapy | Response Rate | NNT | Long-term Benefit |
|---|---|---|---|
| Cognitive Behavioural Therapy (CBT) | 50-70% | 4 | Sustained at 6-12 months [7,8] |
| Gut-directed hypnotherapy | 50-70% | 4 | Sustained at 1-5 years [7,21] |
| Mindfulness-based therapy | 40-60% | — | Improves quality of life [8] |
Key point: Psychological therapies have durable benefit; effects persist after therapy ends. [7,8,21] Cost-effective long-term. [8]
Quality of Life Outcomes
With effective treatment: Most patients achieve meaningful improvement in quality of life, even if symptoms do not completely resolve. [10]
Domains that improve: [10]
- Physical function (less pain, better energy)
- Psychological (reduced anxiety, improved mood)
- Social (less avoidance, improved relationships)
- Work productivity (less absenteeism)
Predictors of QOL improvement: [10,14]
- Symptom reduction (even partial)
- Treatment of comorbid anxiety/depression
- Engagement with psychological therapy
- Realistic expectations (symptom control, not cure)
Long-term Follow-up
Primary care: Most patients managed in primary care; annual review or as needed for symptom flares. [3]
Specialist follow-up: Reserve for: [3]
- Severe, refractory symptoms despite optimal therapy
- Diagnostic uncertainty (concern for organic disease)
- Trial of specialist therapies (linaclotide, eluxadoline, biofeedback)
- Psychological therapy access
Re-evaluation: If significant change in symptoms (new red flags, marked worsening), re-investigate to exclude new pathology. [3]
10. Evidence and Guidelines
Key Guidelines
-
NICE Clinical Guideline CG61. Irritable bowel syndrome in adults: diagnosis and management. National Institute for Health and Care Excellence. 2008 (updated 2017). [3]
- UK gold standard for IBS management
- Emphasises clinical diagnosis, limited investigations, symptom-directed therapy
-
Vasant DH, Paine PA, Black CJ, et al. British Society of Gastroenterology guidelines on the management of irritable bowel syndrome. Gut. 2021;70(7):1214-1240. [PMID: 33903147] [4]
- Comprehensive UK guideline; updates NICE
- Evidence-based recommendations for diet, pharmacotherapy, psychological therapy
-
Lacy BE, Pimentel M, Brenner DM, et al. ACG Clinical Guideline: Management of Irritable Bowel Syndrome. Am J Gastroenterol. 2021;116(1):17-44. [PMID: 33315591] [6]
- USA guideline; includes therapies available in USA (e.g., rifaximin, eluxadoline)
-
Mearin F, Lacy BE, Chang L, et al. Bowel Disorders (Rome IV). Gastroenterology. 2016;150:1393-1407. [PMID: 27144627] [2]
- Defines Rome IV diagnostic criteria for functional bowel disorders, including IBS
-
Ford AC, Lacy BE, Talley NJ. Irritable Bowel Syndrome. N Engl J Med. 2017;376(26):2566-2578. [PMID: 28657875]
- Authoritative review of IBS pathophysiology, diagnosis, management
Key Evidence: Epidemiology
-
Oka P, Parr H, Barberio B, et al. Global prevalence of irritable bowel syndrome according to Rome III or IV criteria: a systematic review and meta-analysis. Lancet Gastroenterol Hepatol. 2020;5(10):908-917. [PMID: 32702295] [11]
- Pooled prevalence: 9.2% (Rome III), 3.8% (Rome IV); female predominance 1.5:1
-
Lovell RM, Ford AC. Global prevalence of and risk factors for irritable bowel syndrome: a meta-analysis. Clin Gastroenterol Hepatol. 2012;10(7):712-721. [PMID: 22426087] [12]
- Meta-analysis: 11.2% global prevalence; female OR 1.67; lower in elderly
-
Sperber AD, Bangdiwala SI, Drossman DA, et al. Worldwide Prevalence and Burden of Functional Gastrointestinal Disorders, Results of Rome Foundation Global Study. Gastroenterology. 2021;160(1):99-114. [PMID: 32294476] [17]
- 73,076 respondents, 33 countries; 40% have functional GI disorder
-
Black CJ, Ford AC. Global burden of irritable bowel syndrome: trends, predictions and risk factors. Nat Rev Gastroenterol Hepatol. 2020;17(8):473-486. [PMID: 32296140] [1]
- Comprehensive review of global IBS burden, quality of life, economic impact
Key Evidence: Pathophysiology
-
Enck P, Aziz Q, Barbara G, et al. Irritable bowel syndrome. Nat Rev Dis Primers. 2016;2:16014. [PMID: 27159638] [13]
- Detailed pathophysiology: visceral hypersensitivity, motility, microbiome, immune activation
-
Ford AC, Sperber AD, Corsetti M, Camilleri M. Irritable bowel syndrome. Lancet. 2020;396(10263):1675-1688. [PMID: 33049221]
- Authoritative review; pathophysiology, diagnosis, treatment
-
Klem F, Wadhwa A, Prokop LJ, et al. Prevalence, Risk Factors, and Outcomes of Irritable Bowel Syndrome After Infectious Enteritis: A Systematic Review and Meta-analysis. Gastroenterology. 2017;152(5):1042-1054. [PMID: 27894741] [16]
- Post-infectious IBS develops in 10% after gastroenteritis; risk factors identified
Key Evidence: Dietary Management
-
Halmos EP, Power VA, Shepherd SJ, et al. A diet low in FODMAPs reduces symptoms of irritable bowel syndrome. Gastroenterology. 2014;146(1):67-75. [PMID: 24076059] [15]
- RCT: Low FODMAP diet superior to standard Australian diet for IBS symptoms
-
Schumann D, Klose P, Lauche R, et al. Low fermentable, oligo-, di-, mono-saccharides and polyol diet in the treatment of irritable bowel syndrome: A systematic review and meta-analysis. Nutrition. 2018;45:24-31. [PMID: 29129233] [5]
- Meta-analysis: Low FODMAP diet effective for global IBS symptoms, abdominal pain, bloating
Key Evidence: Pharmacotherapy
-
Ford AC, Lacy BE, Harris LA, et al. Effect of Antidepressants and Psychological Therapies in Irritable Bowel Syndrome: An Updated Systematic Review and Meta-Analysis. Am J Gastroenterol. 2019;114(1):21-39. [PMID: 30177784] [6]
- TCAs: NNT = 4; SSRIs: NNT = 6-8; psychological therapies: NNT = 4
-
Ruepert L, Quartero AO, de Wit NJ, et al. Bulking agents, antispasmodics and antidepressants for the treatment of irritable bowel syndrome. Cochrane Database Syst Rev. 2011;(8):CD003460. [PMID: 21833945] [20]
- Cochrane review: TCAs effective (NNT = 4); antispasmodics modest benefit
-
Rao SSC, Lembo AJ, Shiff SJ, et al. A 12-week, randomized, controlled trial with a 4-week randomized withdrawal period to evaluate the efficacy and safety of linaclotide in irritable bowel syndrome with constipation. Am J Gastroenterol. 2012;107(11):1714-1724. [PMID: 22986440] [19]
- RCT: Linaclotide effective for IBS-C (pain, stool frequency, quality of life)
-
Khanna R, MacDonald JK, Levesque BG. Peppermint oil for the treatment of irritable bowel syndrome: a systematic review and meta-analysis. J Clin Gastroenterol. 2014;48(6):505-512. [PMID: 24100754] [22]
- Meta-analysis: Peppermint oil effective for global IBS symptoms; NNT = 3-4
Key Evidence: Psychological Therapies
-
Ford AC, Lacy BE, Harris LA, et al. Effect of Antidepressants and Psychological Therapies in Irritable Bowel Syndrome: An Updated Systematic Review and Meta-Analysis. Am J Gastroenterol. 2019;114(1):21-39. [PMID: 30177784] [7]
- Psychological therapies (CBT, hypnotherapy) effective; NNT = 4; equivalent to pharmacotherapy
-
Black CJ, Thakur ER, Houghton LA, et al. Efficacy of psychological therapies for irritable bowel syndrome: systematic review and network meta-analysis. Gut. 2020;69(8):1441-1451. [PMID: 32276950] [8]
- Network meta-analysis: CBT and gut-directed hypnotherapy most effective; long-term benefit
-
Miller V, Carruthers HR, Morris J, et al. Hypnotherapy for irritable bowel syndrome: an audit of one thousand adult patients. Aliment Pharmacol Ther. 2015;41(9):844-855. [PMID: 25689324] [21]
- Long-term follow-up: gut-directed hypnotherapy produces sustained benefit at 1-5 years
Key Evidence: Mental Health Comorbidity
- Staudacher HM, Black CJ, Teasdale SB, et al. Irritable bowel syndrome and mental health comorbidity - approach to multidisciplinary management. Nat Rev Gastroenterol Hepatol. 2023;20(9):582-596. [PMID: 37268741] [14]
- Comprehensive review: anxiety/depression in 30% of IBS; integrated care approach recommended
Key Evidence: Prognosis and Burden
-
Canavan C, West J, Card T. The epidemiology of irritable bowel syndrome. Clin Epidemiol. 2014;6:71-80. [PMID: 24523597] [9]
- Natural history: chronic, relapsing-remitting; does not progress to IBD or cancer
-
Drossman DA, Chang L, Bellamy N, et al. Severity in irritable bowel syndrome: a Rome Foundation Working Team report. Am J Gastroenterol. 2011;106(10):1749-1759. [PMID: 21747417] [10]
- IBS significantly impairs quality of life; severity comparable to IBD, diabetes
11. Patient Explanation
What is IBS?
Irritable bowel syndrome (IBS) is a common condition affecting the gut (bowel). It causes abdominal pain, bloating, and changes in your bowel habit — either diarrhoea, constipation, or both.
IBS is a "functional" disorder, which means the gut is sensitive and not working quite as it should, but there is no damage, inflammation, or disease that we can see on scans or tests.
How common is it?
Very common. About 1 in 10 people have IBS. It often starts in young adulthood (20s-30s) and affects women more than men.
What causes it?
We don't fully understand the cause, but IBS involves several factors:
- Sensitive gut: Your gut is more sensitive to normal activity (like stretching from gas or food passing through), so you feel pain or discomfort when others wouldn't.
- Gut-brain connection: There's a strong link between your gut and your brain. Stress, anxiety, or worry can make symptoms worse.
- Diet: Certain foods (like onions, garlic, wheat, beans, apples) can be hard for your gut to digest, causing bloating and pain.
- Gut bacteria: The balance of bacteria in your gut may be different in IBS.
Is it serious?
No. IBS is uncomfortable and can affect your quality of life, but it is not dangerous. It does not lead to:
- Cancer
- Inflammatory bowel disease (Crohn's or ulcerative colitis)
- Damage to your bowel
How is it diagnosed?
IBS is diagnosed based on your symptoms, using specific criteria (called "Rome IV criteria"). Your doctor will:
- Ask about your symptoms (pain, bloating, bowel habit)
- Examine your tummy
- Do some simple blood tests to rule out other conditions (e.g., coeliac disease, inflammation)
If you're over 50, or have "red flag" symptoms (like blood in your stool, weight loss, or a family history of bowel cancer), you may need further tests like a camera test (colonoscopy) to make sure there's nothing else going on.
How is it treated?
There's no cure for IBS, but there are many treatments that can help control your symptoms:
-
Diet changes:
- Eat regular meals; don't skip meals.
- Drink plenty of water (1.5-2 litres/day).
- Cut down on caffeine, alcohol, and fizzy drinks.
- Try a "low FODMAP diet" — this involves avoiding certain foods that are hard to digest (like onions, wheat, apples). A dietitian can help guide you through this.
-
Medicines:
- For pain/bloating: Antispasmodic tablets (like mebeverine) or peppermint oil capsules.
- For constipation: Laxatives (like Movicol).
- For diarrhoea: Anti-diarrhoea tablets (like loperamide).
- For ongoing pain: Low-dose antidepressants (used for pain relief, not depression) can help.
-
Stress management:
- IBS symptoms often get worse with stress. Talking therapies (like CBT — cognitive behavioural therapy) or relaxation techniques (like hypnotherapy) can really help.
-
Exercise: Regular physical activity (like walking, cycling, swimming) can improve gut function and reduce stress.
What can I do to help myself?
- Keep a symptom diary: Track what you eat and when symptoms occur. This can help identify trigger foods.
- Manage stress: Try relaxation techniques, yoga, or mindfulness.
- Don't avoid foods unnecessarily: Cutting out lots of foods can make things worse. Work with a dietitian if you're unsure.
- Stay active: Regular exercise helps your gut and your mood.
Will it get better?
IBS is a long-term condition. Symptoms often come and go — you may have good weeks and bad weeks. Many people improve over time, especially with the right treatment. About 1 in 3 people with IBS find their symptoms improve or even go away after several years.
The key is finding what works for you — whether it's diet, medicines, or stress management.
When should I see a doctor?
See your doctor if:
- You have new symptoms or your symptoms change.
- You have "red flag" symptoms: blood in your stool, unexplained weight loss, severe pain, or symptoms that wake you at night.
- Your symptoms are affecting your quality of life and you need more help.
Where can I get more information?
- IBS Network (UK): www.theibsnetwork.org — charity supporting people with IBS
- Guts UK: www.gutscharity.org.uk — information on digestive conditions
- NHS website: www.nhs.uk/conditions/irritable-bowel-syndrome-ibs/
12. References
-
Black CJ, Ford AC. Global burden of irritable bowel syndrome: trends, predictions and risk factors. Nat Rev Gastroenterol Hepatol. 2020;17(8):473-486. PMID: 32296140
-
Mearin F, Lacy BE, Chang L, et al. Bowel Disorders. Gastroenterology. 2016;150(6):1393-1407. PMID: 27144627
-
NICE Clinical Guideline CG61. Irritable bowel syndrome in adults: diagnosis and management. National Institute for Health and Care Excellence. 2008 (updated 2017). Available at: www.nice.org.uk/guidance/cg61
-
Vasant DH, Paine PA, Black CJ, et al. British Society of Gastroenterology guidelines on the management of irritable bowel syndrome. Gut. 2021;70(7):1214-1240. PMID: 33903147
-
Schumann D, Klose P, Lauche R, et al. Low fermentable, oligo-, di-, mono-saccharides and polyol diet in the treatment of irritable bowel syndrome: A systematic review and meta-analysis. Nutrition. 2018;45:24-31. PMID: 29129233
-
Lacy BE, Pimentel M, Brenner DM, et al. ACG Clinical Guideline: Management of Irritable Bowel Syndrome. Am J Gastroenterol. 2021;116(1):17-44. PMID: 33315591
-
Ford AC, Lacy BE, Harris LA, et al. Effect of Antidepressants and Psychological Therapies in Irritable Bowel Syndrome: An Updated Systematic Review and Meta-Analysis. Am J Gastroenterol. 2019;114(1):21-39. PMID: 30177784
-
Black CJ, Thakur ER, Houghton LA, et al. Efficacy of psychological therapies for irritable bowel syndrome: systematic review and network meta-analysis. Gut. 2020;69(8):1441-1451. PMID: 32276950
-
Canavan C, West J, Card T. The epidemiology of irritable bowel syndrome. Clin Epidemiol. 2014;6:71-80. PMID: 24523597
-
Drossman DA, Chang L, Bellamy N, et al. Severity in irritable bowel syndrome: a Rome Foundation Working Team report. Am J Gastroenterol. 2011;106(10):1749-1759. PMID: 21747417
-
Oka P, Parr H, Barberio B, et al. Global prevalence of irritable bowel syndrome according to Rome III or IV criteria: a systematic review and meta-analysis. Lancet Gastroenterol Hepatol. 2020;5(10):908-917. PMID: 32702295
-
Lovell RM, Ford AC. Global prevalence of and risk factors for irritable bowel syndrome: a meta-analysis. Clin Gastroenterol Hepatol. 2012;10(7):712-721. PMID: 22426087
-
Enck P, Aziz Q, Barbara G, et al. Irritable bowel syndrome. Nat Rev Dis Primers. 2016;2:16014. PMID: 27159638
-
Staudacher HM, Black CJ, Teasdale SB, et al. Irritable bowel syndrome and mental health comorbidity - approach to multidisciplinary management. Nat Rev Gastroenterol Hepatol. 2023;20(9):582-596. PMID: 37268741
-
Halmos EP, Power VA, Shepherd SJ, et al. A diet low in FODMAPs reduces symptoms of irritable bowel syndrome. Gastroenterology. 2014;146(1):67-75. PMID: 24076059
-
Klem F, Wadhwa A, Prokop LJ, et al. Prevalence, Risk Factors, and Outcomes of Irritable Bowel Syndrome After Infectious Enteritis: A Systematic Review and Meta-analysis. Gastroenterology. 2017;152(5):1042-1054. PMID: 27894741
-
Sperber AD, Bangdiwala SI, Drossman DA, et al. Worldwide Prevalence and Burden of Functional Gastrointestinal Disorders, Results of Rome Foundation Global Study. Gastroenterology. 2021;160(1):99-114. PMID: 32294476
-
Hreinsson JP, Tornblom H, Tack J, et al. Factor Analysis of the Rome IV Criteria for Major Disorders of Gut-Brain Interaction (DGBI) Globally and Across Geographical, Sex, and Age Groups. Gastroenterology. 2023;164(7):1211-1222. PMID: 36889555
-
Rao SSC, Lembo AJ, Shiff SJ, et al. A 12-week, randomized, controlled trial with a 4-week randomized withdrawal period to evaluate the efficacy and safety of linaclotide in irritable bowel syndrome with constipation. Am J Gastroenterol. 2012;107(11):1714-1724. PMID: 22986440
-
Ruepert L, Quartero AO, de Wit NJ, et al. Bulking agents, antispasmodics and antidepressants for the treatment of irritable bowel syndrome. Cochrane Database Syst Rev. 2011;(8):CD003460. PMID: 21833945
-
Miller V, Carruthers HR, Morris J, et al. Hypnotherapy for irritable bowel syndrome: an audit of one thousand adult patients. Aliment Pharmacol Ther. 2015;41(9):844-855. PMID: 25689324
-
Khanna R, MacDonald JK, Levesque BG. Peppermint oil for the treatment of irritable bowel syndrome: a systematic review and meta-analysis. J Clin Gastroenterol. 2014;48(6):505-512. PMID: 24100754
-
Pimentel M, Lembo A, Chey WD, et al. Rifaximin therapy for patients with irritable bowel syndrome without constipation. N Engl J Med. 2011;364(1):22-32. PMID: 21208106
-
Kunkel D, Basseri RJ, Makhani MD, et al. Methane on breath test is associated with constipation: a systematic review and meta-analysis. Dig Dis Sci. 2011;56(6):1612-1618. PMID: 21286935
-
Menees SB, Maneerattannaporn M, Kim HM, Chey WD. The efficacy and safety of rifaximin for the irritable bowel syndrome: a systematic review and meta-analysis. Am J Gastroenterol. 2012;107(1):28-35. PMID: 22045120
-
Lembo A, Pimentel M, Rao SS, et al. Repeat treatment with rifaximin is safe and effective in patients with diarrhea-predominant irritable bowel syndrome. Gastroenterology. 2016;151(6):1113-1121. PMID: 27528626 | DOI: 10.1053/j.gastro.2016.08.003
-
Beattie DT, Smith JA. Serotonin pharmacology in the gastrointestinal tract: a review. Naunyn Schmiedebergs Arch Pharmacol. 2008;377(3):181-203. PMID: 18398601 | DOI: 10.1007/s00210-008-0276-9
-
Bednarska O, Walter SA, Casado-Bedmar M, et al. Vasoactive Intestinal Polypeptide and Mast Cells Regulate Increased Passage of Colonic Bacteria in Patients With Irritable Bowel Syndrome. Gastroenterology. 2017;153(4):948-960. PMID: 28583826 | DOI: 10.1053/j.gastro.2017.06.051
-
Barbara G, Stanghellini V, De Giorgio R, et al. Activated mast cells in proximity to colonic nerves correlate with abdominal pain in irritable bowel syndrome. Gastroenterology. 2004;126(3):693-702. PMID: 14988823 | DOI: 10.1053/j.gastro.2003.11.055
-
Vijayvargiya P, Camilleri M, Chedid V, et al. Analysis of Postprandial Serum Bile Acids in Patients With Bile Acid Diarrhea and Irritable Bowel Syndrome With Diarrhea. Clin Gastroenterol Hepatol. 2020;18(12):2704-2711. PMID: 32061870 | DOI: 10.1016/j.cgh.2020.02.015
-
Pittayanon R, Lau JT, Yuan Y, et al. Gut Microbiota in Patients With Irritable Bowel Syndrome-A Systematic Review. Gastroenterology. 2019;157(1):97-108. PMID: 30940523 | DOI: 10.1053/j.gastro.2019.03.049
-
Bonfiglio F, Zheng T, Garcia-Etxebarria K, et al. Female-Specific Association Between Variants on Chromosome 9 and Self-Reported Diagnosis of Irritable Bowel Syndrome. Gastroenterology. 2018;155(1):168-179. PMID: 29596934 | DOI: 10.1053/j.gastro.2018.03.064
-
Mayer EA, Labus JS, Tillisch K, et al. Towards a systems view of IBS. Nat Rev Gastroenterol Hepatol. 2015;12(10):592-605. PMID: 26303674 | DOI: 10.1038/nrgastro.2015.121
-
Houghton LA, Heitkemper M, Crowell MD, et al. Age, Gender, and Women's Health and the Patient. Gastroenterology. 2016;150(6):1332-1343. PMID: 27144632 | DOI: 10.1053/j.gastro.2016.02.017
-
Brennan BP, Fogarty KV, Roberts JL, et al. Duloxetine in the treatment of irritable bowel syndrome: an open-label pilot study. Hum Psychopharmacol. 2009;24(5):423-428. PMID: 19548294 | DOI: 10.1002/hup.1038
-
Chey WD, Lembo AJ, Lavins BJ, et al. Linaclotide for irritable bowel syndrome with constipation: a 26-week, randomized, double-blind, placebo-controlled trial to evaluate efficacy and safety. Am J Gastroenterol. 2012;107(11):1702-1712. PMID: 22986437 | DOI: 10.1038/ajg.2012.254
-
Miner PB Jr, Koltun WD, Wiener GJ, et al. A Randomized Phase III Clinical Trial of Plecanatide, a Uroguanylin Analog, in Patients With Chronic Idiopathic Constipation. Am J Gastroenterol. 2017;112(4):613-621. PMID: 28244497 | DOI: 10.1038/ajg.2016.611
-
Chey WD, Rosenbaum DP. Tenapanor Treatment of Patients With Constipation-Predominant Irritable Bowel Syndrome: A Phase 2, Randomized, Placebo-Controlled Efficacy and Safety Trial. Am J Gastroenterol. 2017;112(5):763-774. PMID: 28291240 | DOI: 10.1038/ajg.2017.41
-
Garsed K, Chernova J, Hastings M, et al. A randomised trial of ondansetron for the treatment of irritable bowel syndrome with diarrhoea. Gut. 2014;63(10):1617-1625. PMID: 24334242 | DOI: 10.1136/gutjnl-2013-305989
-
Ford AC, Harris LA, Lacy BE, et al. Systematic review with meta-analysis: the efficacy of prebiotics, probiotics, synbiotics and antibiotics in irritable bowel syndrome. Aliment Pharmacol Ther. 2018;48(10):1044-1060. PMID: 30294792 | DOI: 10.1111/apt.15001
-
El-Salhy M, Hatlebakk JG, Gilja OH, et al. Efficacy of faecal microbiota transplantation for patients with irritable bowel syndrome in a randomised, double-blind, placebo-controlled study. Gut. 2020;69(5):859-867. PMID: 31852769 | DOI: 10.1136/gutjnl-2019-319630
-
Chumpitazi BP, Kearns GL, Shulman RJ. Review article: the physiological effects and safety of peppermint oil and its efficacy in irritable bowel syndrome and other functional disorders. Aliment Pharmacol Ther. 2018;47(6):738-752. PMID: 29372567 | DOI: 10.1111/apt.14519
-
Farmer AD, Ruffle JK, Aziz Q. The role of neuromodulation in functional GI disorders. Curr Gastroenterol Rep. 2015;17(6):22. PMID: 25939463 | DOI: 10.1007/s11894-015-0447-5
13. Examination Focus
MRCP/FRACP Viva Points
"Irritable bowel syndrome is a functional disorder of gut-brain interaction, affecting 9% of adults globally by Rome IV criteria. Diagnosis is clinical: recurrent abdominal pain at least 1 day/week for 3 months, associated with two or more of — related to defecation, change in stool frequency, or change in stool form. Subtype as IBS-D, IBS-C, IBS-M, or IBS-U based on Bristol Stool Scale. IBS is a diagnosis of exclusion if red flags present. Essential investigations: FBC, CRP, coeliac serology, and faecal calprotectin if diarrhoea-predominant. Management is symptom-directed: low FODMAP diet (50-80% response), antispasmodics (mebeverine, peppermint oil) for pain, laxatives (macrogol) or linaclotide for IBS-C, loperamide for IBS-D, and low-dose tricyclic antidepressants (amitriptyline) for pain-predominant IBS. Psychological therapies — CBT and gut-directed hypnotherapy — are as effective as pharmacotherapy, with NNT of 4. IBS is chronic, relapsing-remitting, but does not progress to IBD or colorectal cancer."
High-Yield Exam Topics
Rome IV Criteria: [2]
- Abdominal pain ≥1 day/week for 3 months
- Associated with 2+: related to defecation, change in stool frequency, change in stool form
- Onset ≥6 months prior
Subtypes: [2]
- IBS-C: >25% Bristol 1-2, less than 25% Bristol 6-7
- IBS-D: >25% Bristol 6-7, less than 25% Bristol 1-2
- IBS-M: >25% both hard and loose
- IBS-U: Does not fit above
Pathophysiology (5 mechanisms): [1,13]
- Visceral hypersensitivity
- Altered gut motility
- Gut-brain axis dysfunction
- Microbiome dysbiosis
- Low-grade inflammation
Investigations: [3,4]
- First-line: FBC, CRP/ESR, TTG-IgA + total IgA, faecal calprotectin (IBS-D)
- Red flags: colonoscopy, imaging
Pharmacotherapy (NNT): [6,20,22]
- Peppermint oil: NNT = 3-4
- TCAs (amitriptyline): NNT = 4
- Linaclotide (IBS-C): NNT = 6-7
Psychological therapy (NNT): [7,8]
- CBT: NNT = 4
- Gut-directed hypnotherapy: NNT = 4
Low FODMAP diet: [5,15]
- 50-80% response rate
- Dietitian-led: elimination (4-8 weeks) → reintroduction → personalisation
Common MRCP/FRACP Exam Mistakes
- ❌ Forgetting coeliac serology: NICE/BSG recommend in all IBS patients [3,4]
- ❌ Over-investigating typical IBS: Causes anxiety, reinforces illness behaviour [3]
- ❌ Missing red flags: Age >50, bleeding, weight loss, family history → investigate [3]
- ❌ Diagnosing IBS in elderly without investigation: Always exclude organic disease [3]
- ❌ Not considering bile acid malabsorption in refractory IBS-D: Trial bile acid sequestrant [3]
- ❌ Forgetting psychological therapies: CBT and hypnotherapy have strong evidence (NNT = 4) [7,8]
- ❌ Using TCAs at antidepressant dose: Analgesic effect at low dose (10-30mg) [6,20]
- ❌ Prescribing low FODMAP without dietitian: Self-directed diets often fail [5,15]
Viva Voce Scenarios (MRCP/FRACP)
Scenario 1: Diagnostic Approach to IBS
Examiner: "A 28-year-old woman presents with 9 months of crampy lower abdominal pain, bloating, and alternating constipation and diarrhoea. How would you approach this?"
Model Answer: "This presentation suggests a functional bowel disorder, most likely irritable bowel syndrome. I would take a detailed history to assess if she meets Rome IV diagnostic criteria: recurrent abdominal pain at least 1 day per week for the last 3 months, associated with two or more of — related to defecation, change in stool frequency, or change in stool form, with symptom onset at least 6 months ago. I'd establish her predominant stool pattern using the Bristol Stool Scale to subtype as IBS-C, IBS-D, IBS-M, or IBS-U.
Critically, I would screen for red flag features: age over 50 with new symptoms, unintentional weight loss, rectal bleeding, nocturnal symptoms waking her from sleep, family history of colorectal cancer or inflammatory bowel disease, and examine for anaemia or abdominal mass.
If no red flags, I would perform limited first-line investigations: FBC, CRP, tissue transglutaminase IgA with total IgA to exclude coeliac disease, and faecal calprotectin if diarrhoea-predominant to exclude IBD. NICE and BSG guidelines recommend coeliac serology in all patients meeting IBS criteria, as up to 4% have undiagnosed coeliac disease.
If investigations normal and she meets Rome IV criteria without red flags, I would confidently diagnose IBS and avoid over-investigation, which can increase anxiety and reinforce illness behaviour."
Examiner: "What if she were 55 years old with otherwise identical symptoms?"
Model Answer: "Age over 50 with new onset bowel symptoms is a red flag. Even if she meets Rome IV criteria, I would investigate with colonoscopy to exclude colorectal cancer, polyps, microscopic colitis, or inflammatory bowel disease before diagnosing IBS. The threshold for investigation is much lower in this age group."
Scenario 2: Management of Refractory IBS-D
Examiner: "A 35-year-old man with established IBS-D has tried a low FODMAP diet and loperamide with partial improvement. He still has 4-5 loose stools daily with urgency. What would you do next?"
Model Answer: "This is refractory IBS-D. I would first ensure he has had adequate dietary intervention — low FODMAP diet should be dietitian-led with proper elimination and reintroduction phases; self-directed diets often fail. I'd confirm he's using loperamide correctly: 2-4mg after each loose stool, not prophylactically, maximum 16mg per day.
If adherence to first-line therapy is good, I would consider bile acid malabsorption, which affects 25-30% of IBS-D patients. In the UK, if SeHCAT scanning is available, less than 15% retention at 7 days confirms bile acid malabsorption. Alternatively, an empirical trial of a bile acid sequestrant — cholestyramine 4g once daily before breakfast or colesevelam 625mg twice daily — is both diagnostic and therapeutic. If symptoms improve significantly within 2-4 weeks, bile acid malabsorption is likely.
If bile acid sequestrant is ineffective or not tolerated, I would consider low-dose tricyclic antidepressant therapy. Amitriptyline 10mg nocte, titrated slowly to 30mg if needed, has visceral analgesic effects and anticholinergic properties that slow gut transit. The NNT is 4 for symptom improvement. I would explain this is for pain modulation, not depression, and effects take 2-4 weeks.
If symptoms remain refractory, I would assess for psychological comorbidity — anxiety and depression are present in 30% of IBS patients — and consider referral for cognitive behavioural therapy or gut-directed hypnotherapy, both of which have NNT of 4 and produce durable benefit.
In specialist centres, rifaximin 550mg three times daily for 14 days is an option; it's FDA-approved in the USA for IBS-D and has an NNT of 10-11, particularly effective for bloating. However, it's not routinely available on the NHS in the UK."
Examiner: "What about 5-HT3 antagonists?"
Model Answer: "Ondansetron, a 5-HT3 antagonist, can be used off-label for severe refractory IBS-D. It reduces colonic transit and secretion. Dosing is 4-8mg three times daily, titrated to response. The main side effect is constipation, which is dose-limiting in 20-30%. Unlike alosetron, which was associated with ischaemic colitis and is restricted in the USA, ondansetron does not carry this risk and is generally safe for long-term use."
Scenario 3: IBS-C Management Algorithm
Examiner: "A 42-year-old woman with IBS-C has been taking macrogol 2 sachets daily for 6 months with minimal improvement. She still has hard stools, straining, and abdominal pain 3 days per week. What are the next steps?"
Model Answer: "This is refractory IBS-C. I would first optimise her laxative regimen — ensure she's adequately hydrated (1.5-2 litres per day), taking macrogol regularly, and has tried dose titration (up to 3-4 sachets daily if needed). I would also ensure she's had a trial of soluble fibre such as psyllium or ispaghula, which softens stool; insoluble fibre like bran should be avoided if bloating is severe.
If optimised first-line therapy fails, I would consider second-line secretagogues. Linaclotide 290mcg once daily, taken 30 minutes before breakfast on an empty stomach, is a guanylate cyclase-C agonist that increases intestinal fluid secretion, accelerates transit, and has analgesic effects on visceral pain. The NNT is 6-7 for the composite endpoint of pain reduction and increased bowel movements. The main side effect is diarrhoea in 10-20%, usually transient. Linaclotide is expensive and NICE does not recommend routine NHS use, so it's typically a specialist prescription for severe refractory cases.
Alternatively, prucalopride 2mg once daily, a 5-HT4 agonist, is a prokinetic licensed for chronic constipation with evidence in IBS-C. Side effects include headache and nausea.
If she has features suggesting pelvic floor dysfunction — excessive straining, sensation of incomplete evacuation, need for manual manoeuvres — I would refer for anorectal manometry and balloon expulsion testing. If dyssynergic defecation is confirmed (paradoxical anal contraction during attempted defecation), biofeedback therapy is highly effective.
For pain-predominant IBS-C with anxiety or depression, I would consider an SSRI such as sertraline 50mg daily, which has prokinetic effects and anxiolytic benefits. The NNT is 6-8.
Psychological therapy — particularly CBT or gut-directed hypnotherapy — should be offered if inadequate response to pharmacotherapy, with NNT of 4 and sustained long-term benefit."
Scenario 4: Post-Infectious IBS
Examiner: "A 30-year-old woman had bacterial gastroenteritis 6 months ago while travelling in India. Since then, she has persistent loose stools 4-5 times daily, abdominal pain, and bloating. Stool cultures are negative. What is your differential and approach?"
Model Answer: "The key feature here is persistent gastrointestinal symptoms following acute infectious gastroenteritis, raising the possibility of post-infectious IBS. Approximately 10-15% of patients develop IBS after gastroenteritis. Risk factors include female sex, younger age, severity of the initial infection, antibiotic use, and psychological stress at the time of infection.
The pathophysiology involves persistent low-grade inflammation, increased mast cells in the gut mucosa, microbiome dysbiosis, increased intestinal permeability, and sensitisation of enteric nerves, leading to visceral hypersensitivity.
However, before diagnosing post-infectious IBS, I must exclude persistent infection or alternative diagnoses:
- Persistent infection: Repeat stool microscopy, culture, ova, cysts, and parasites — particularly Giardia lamblia, which can cause prolonged diarrhoea post-travel and may require three stool samples for detection
- Small intestinal bacterial overgrowth (SIBO): Possible following gastroenteritis; hydrogen/methane breath testing, though evidence for treating SIBO in IBS is weak
- Inflammatory bowel disease: Faecal calprotectin; if elevated (>100 μg/g), proceed to colonoscopy
- Coeliac disease: Tissue transglutaminase IgA with total IgA
If investigations are negative and she meets Rome IV criteria, I would diagnose post-infectious IBS-D. Management is the same as standard IBS-D: low FODMAP diet, loperamide, consideration of bile acid malabsorption with trial of bile acid sequestrant if refractory, and low-dose amitriptyline for pain.
There is some evidence for probiotics in post-infectious IBS, particularly Bifidobacterium infantis or Saccharomyces boulardii, though benefits are modest. The natural history of post-infectious IBS is variable — some patients improve spontaneously over 6-12 months, but 50% remain symptomatic at 6 years."
Scenario 5: IBS and Psychological Comorbidity
Examiner: "A 38-year-old man with IBS-M has tried dietary modification and antispasmodics with minimal benefit. He mentions he's been 'stressed and anxious' recently. How does this influence your management?"
Model Answer: "The gut-brain axis is central to IBS pathophysiology, and psychological factors — particularly anxiety, depression, and chronic stress — are present in up to 30% of IBS patients. There is a bidirectional relationship: psychological distress worsens IBS symptoms through HPA axis activation, increased gut permeability, and visceral hypersensitivity; conversely, chronic IBS symptoms impair quality of life and worsen mood.
I would formally screen for anxiety and depression using validated tools such as the GAD-7 and PHQ-9. If significant psychological comorbidity is identified, this should be addressed concurrently with IBS management, as it predicts poorer response to pharmacotherapy alone.
First-line psychological therapy for IBS includes cognitive behavioural therapy (CBT) and gut-directed hypnotherapy. Both have strong evidence with an NNT of 4 for global symptom improvement, equivalent to pharmacotherapy. CBT typically involves 6-12 sessions addressing maladaptive thought patterns, catastrophising, and developing coping strategies. Gut-directed hypnotherapy involves 7-12 sessions inducing relaxation and modulating visceral perception. These therapies produce durable benefit, with effects sustained at 1-5 years post-treatment.
If pharmacotherapy is preferred or as an adjunct, I would consider a neuromodulator. Given his anxiety, an SSRI such as sertraline 50mg daily would provide anxiolytic benefit as well as modest IBS symptom improvement (NNT 6-8). Alternatively, if pain is predominant, low-dose amitriptyline 10-30mg nocte has visceral analgesic effects (NNT 4), though I would explain this is not treating his anxiety and may need concurrent SSRI or psychological therapy.
Mindfulness-based stress reduction is another evidence-based option, improving quality of life and reducing symptom severity.
The key is recognising that effective IBS management in patients with psychological comorbidity requires an integrated, multidisciplinary approach addressing both gut and brain."
Scenario 6: Red Flags and When to Investigate
Examiner: "A 50-year-old man presents with 4 months of lower abdominal pain, bloating, and looser stools. He thinks he may have IBS. What features in the history would concern you?"
Model Answer: "This patient meets the age threshold where new bowel symptoms warrant investigation. Age over 50 is a red flag, as colorectal cancer risk increases significantly. I would screen for additional red flags:
- Unintentional weight loss: Suggests malignancy, inflammatory bowel disease, coeliac disease, or malabsorption
- Rectal bleeding: Not blood on tissue from haemorrhoids, but blood mixed with stool, suggesting colorectal cancer, polyps, or IBD
- Iron deficiency anaemia: Right-sided colonic cancer, coeliac disease, IBD
- Family history: First-degree relative with colorectal cancer (especially if diagnosed under age 50) or inflammatory bowel disease
- Nocturnal symptoms: Diarrhoea or pain waking him from sleep is unusual for IBS and suggests organic disease such as IBD
- Progressive symptoms: Worsening over weeks to months, rather than fluctuating course
- Palpable mass: Abdominal or rectal mass on examination
If any red flag is present, or simply because he is over 50 with new symptoms, I would investigate with:
- Colonoscopy: To exclude colorectal cancer, polyps, inflammatory bowel disease, or microscopic colitis (which requires biopsies even if mucosa looks normal endoscopically)
- FBC: To detect anaemia
- CRP/ESR: Elevated in IBD or malignancy
- Coeliac serology: TTG-IgA with total IgA
- Faecal calprotectin: If diarrhoea-predominant; >100 μg/g indicates IBD likely
IBS is a diagnosis of exclusion in this age group. Only after excluding organic disease can I confidently diagnose IBS."
Scenario 7: Microscopic Colitis vs IBS-D
Examiner: "A 58-year-old woman has chronic watery diarrhoea. Colonoscopy shows normal-appearing mucosa. The endoscopist didn't take biopsies. What would you do?"
Model Answer: "This is a critical error. Microscopic colitis — which includes collagenous colitis and lymphocytic colitis — is a cause of chronic watery diarrhoea, particularly in older adults, and is diagnosed histologically despite normal endoscopic appearance.
I would repeat colonoscopy with multiple biopsies from the colon (at least 2 from each segment: caecum, ascending, transverse, descending, sigmoid, rectum), as microscopic colitis can have patchy distribution. Histology in collagenous colitis shows a thickened subepithelial collagen band (>10 μm) and lymphocytic infiltrate. Lymphocytic colitis shows intraepithelial lymphocytosis (>20 lymphocytes per 100 epithelial cells) without collagen band thickening.
Microscopic colitis is associated with autoimmune conditions (coeliac disease, thyroid disease), certain medications (NSAIDs, PPIs, SSRIs), and smoking. Treatment includes budesonide (corticosteroid with high first-pass metabolism; 9mg daily for 8 weeks is highly effective), bile acid sequestrants (cholestyramine), and loperamide.
The distinction from IBS-D is important because microscopic colitis has specific effective treatments, whereas IBS-D management is symptom-directed. The lesson: always biopsy normal-appearing mucosa in chronic diarrhoea, especially in older adults."
OSCE/Clinical Exam Scenarios
History-taking station: Patient with abdominal pain and altered bowel habit
- Explore Rome IV criteria (pain frequency, relationship to defecation, stool changes)
- Screen for red flags (age, bleeding, weight loss, family history)
- Assess psychological comorbidity (anxiety, depression, stress)
- Identify trigger foods and dietary patterns
Explanation station: Explain IBS diagnosis and management to patient
- Reassure: benign, does not progress to cancer/IBD
- Explain gut-brain axis, stress-symptom link
- Discuss dietary modification (low FODMAP), pharmacotherapy, psychological therapy
- Set realistic expectations: symptom control, not cure
Data interpretation: Colonoscopy report showing normal mucosa in 55-year-old with diarrhoea
- Differential: IBS-D vs microscopic colitis
- Next step: Ensure biopsies taken (microscopic colitis requires histology even if normal mucosa)
- If biopsies normal: IBS-D; manage with loperamide, consider bile acid sequestrant
Clinical Pearls for Examinations
Pearl 1: Rome IV vs Rome III
- Rome IV requires pain ≥1 day/week (Rome III: ≥3 days/month)
- This makes Rome IV more restrictive: prevalence 3.8% (Rome IV) vs 9.2% (Rome III)
- Always state "Rome IV criteria" in exams to demonstrate up-to-date knowledge
Pearl 2: Coeliac Serology is Mandatory
- NICE and BSG recommend TTG-IgA + total IgA in all IBS patients
- 4% of patients meeting IBS criteria have undiagnosed coeliac disease
- Missing this is a common exam failure point
Pearl 3: Faecal Calprotectin Cutoffs
- less than 50 μg/g: IBD unlikely (95% NPV)
- 50-100 μg/g: Equivocal; repeat or investigate if clinical concern
- >100 μg/g: IBD likely; refer for colonoscopy
- Specificity: Normal calprotectin does not rule out microscopic colitis (requires biopsy)
Pearl 4: Low FODMAP Requires Dietitian
- Self-directed FODMAP diets have poor adherence and efficacy
- Must be three-phase: elimination (4-8 weeks) → reintroduction → personalisation
- Long-term restriction without reintroduction risks nutritional deficiency and microbiome diversity loss
Pearl 5: TCA Dosing
- Analgesic dose: 10-30mg nocte (amitriptyline)
- Antidepressant dose: 75-150mg
- IBS benefit is from visceral analgesia and anticholinergic effects, not antidepressant action
- "Start low, go slow" to minimise side effects
Pearl 6: Bile Acid Malabsorption
- Present in 25-30% of IBS-D
- SeHCAT scan less than 15% retention = BAM (gold standard; limited UK availability, not available USA)
- Empirical trial: Cholestyramine 4g OD for 2-4 weeks; response confirms diagnosis
- Response rate 60-70% in proven BAM
Pearl 7: Psychological Therapies are Evidence-Based
- CBT and gut-directed hypnotherapy: NNT = 4 (equivalent to TCAs, better than SSRIs)
- Produces durable benefit: sustained at 1-5 years
- Not just for "psychological" patients; effective in all IBS subtypes
- Underutilised due to access issues, but should be offered per guidelines
Pearl 8: IBS Does Not Progress
- Critical reassurance point: IBS does not increase risk of IBD, colorectal cancer, or mortality
- Long-term cohort studies (10-20 years) show no excess risk of structural bowel disease
- However, new symptoms or red flags in established IBS warrant re-evaluation
Pearl 9: Bloating and Visible Distension
- Bloating (subjective sensation): 80-90% of IBS patients
- Visible distension (objective abdominal girth increase): 50-60%
- Mechanism: Visceral hypersensitivity (bloating without distension), gas production (FODMAPs, SIBO), impaired gas transit
- Low FODMAP diet and rifaximin particularly effective for bloating
Pearl 10: Overlap with Functional Dyspepsia
- 30-40% of IBS patients also meet criteria for functional dyspepsia
- Symptom overlap: epigastric pain, bloating, early satiety
- Management: Treat both; PPI trial for dyspepsia, IBS-directed therapy for lower GI symptoms
Last Updated: 2026-01-09