Melanoma
A comprehensive guide to Malignant Melanoma covering epidemiology, molecular pathophysiology (BRAF/NRAS/KIT mutations), AJCC 8th edition staging, sentinel lymph node biopsy, and modern immunotherapy/targeted therapy...
What matters first
A comprehensive guide to Malignant Melanoma covering epidemiology, molecular pathophysiology (BRAF/NRAS/KIT mutations), AJCC 8th edition staging, sentinel lymph node biopsy, and modern immunotherapy/targeted therapy...
Mole > 6mm with irregular border/colour
5 Jan 2026
Generated educational material; verify before clinical use.
Visible references section
See the concept before reading it
Study the key anatomy, imaging, and decision pathways as full teaching plates.
Clinical board
A visual summary of the highest-yield teaching signals on this page.
Urgent signals
Safety-critical features pulled from the topic metadata.
- Mole > 6mm with irregular border/colour
- New or changing pigmented lesion (ABCDE criteria)
- Nail pigmentation involving cuticle (Hutchinson's Sign)
- Rapidly growing nodular lesion
Linked comparisons
Differentials and adjacent topics worth opening next.
- Basal Cell Carcinoma
- Squamous Cell Carcinoma
Content status and exam context
This page is AI-generated educational content. It may contain errors or omissions and is not a substitute for current guidelines, local protocols, senior clinical judgement, or professional medical advice.
MedVellum does not claim an individual clinician reviewer, board certification, or professional credential for this page unless a future version names a real, verifiable reviewer.
Clinical explanation and evidence
Melanoma
Disclaimer: > [!WARNING] Medical Disclaimer: This content is for educational and informational purposes only and does not constitute medical advice. Always consult a qualified healthcare professional for diagnosis and treatment. Medical guidelines and best practices change rapidly; users should verify information with current local protocols.
1. Overview
Malignant melanoma is a malignant tumour arising from melanocytes, the pigment-producing cells derived from the neural crest. It represents the most lethal form of skin cancer due to its propensity for early metastasis and aggressive biological behaviour. [1]
While melanoma accounts for only 4% of all skin cancers, it is responsible for approximately 80% of skin cancer deaths. [2] The incidence has been rising faster than any other solid tumour over the past 30 years, with rates doubling in many Western populations. [3] Early detection and complete surgical excision offer excellent cure rates for thin lesions, while advanced disease has historically carried a grave prognosis—though the landscape has been transformed by immunotherapy and targeted therapies over the past decade. [4]
The key clinical message is that melanoma is a preventable cancer when UV protection is practiced, and a curable cancer when detected early. The challenge lies in distinguishing the rare malignant lesion from the thousands of benign naevi present in the general population. [5]
2. Epidemiology
Incidence and Prevalence
Melanoma represents the 5th most common cancer in the United Kingdom, with approximately 16,000 new cases diagnosed annually (2019 data). [6] Incidence rates are highest in Australia and New Zealand (over 50 per 100,000 population), where the combination of fair-skinned populations and high UV exposure creates the perfect storm. [7]
| Statistic | Value | Source |
|---|---|---|
| UK Annual Incidence | 16,000 cases | Cancer Research UK [6] |
| Lifetime Risk (UK) | 1 in 36 (males), 1 in 47 (females) | Cancer Research UK [6] |
| Median Age at Diagnosis | 65 years | SEER Database [8] |
| Male:Female Ratio | 1.2:1 | GLOBOCAN [9] |
| 5-Year Survival (Localized) | 99% | SEER [8] |
| 5-Year Survival (Distant Metastases) | 27% (pre-immunotherapy era) | SEER [8] |
| Mortality (UK) | ~2,300 deaths/year | ONS [10] |
Temporal Trends
The incidence of melanoma has increased by more than 200% since the 1990s in many developed countries. [3] This rise is attributed to:
- Increased recreational UV exposure (holidays, sunbeds)
- Earlier detection through screening programmes
- Ageing populations
- Improved awareness and reporting
However, mortality rates have begun to plateau or decline in some regions due to earlier detection and improved treatments. [11]
Risk Factors
Non-Modifiable:
- Phenotype: Fitzpatrick skin types I-II (fair skin, inability to tan, freckling), red or blonde hair, blue or green eyes. [12]
- Naevi burden: > 50 common melanocytic naevi or > 5 atypical (dysplastic) naevi. [13]
- Genetics: Family history of melanoma (8-12% of cases are familial). Mutations in CDKN2A (p16), CDK4, MITF, BAP1 confer high risk. [14]
- Personal history: Previous melanoma (risk of second primary ~5%). [15]
- Immunosuppression: Organ transplant recipients have 3-8 fold increased risk. [16]
Modifiable:
- UV Exposure: Intermittent high-intensity UV exposure (sunburns) carries greater risk than chronic low-level exposure. Each episode of blistering sunburn in childhood doubles melanoma risk. [17]
- Sunbed Use: Increases risk by 20% overall, and up to 59% if first use is before age 35. [18]
Exam Detail: High-Yield Exam Point: Distinguish between continuous occupational UV exposure (associated more with squamous cell carcinoma) versus intermittent recreational UV exposure with sunburns (stronger association with melanoma). The "weekend warrior" pattern is more melanogenic. [17]
3. Aetiology & Pathophysiology
Cellular Origin
Melanocytes are neural crest-derived cells that migrate to the basal layer of the epidermis during embryonic development. They produce melanin pigment within melanosomes, which are transferred to surrounding keratinocytes to provide photoprotection. [19]
Molecular Pathogenesis
Melanoma arises through accumulation of mutations in melanocytes, driven predominantly by ultraviolet radiation-induced DNA damage. The mutation signature includes characteristic C>T transitions at dipyrimidine sites (UV signature mutations). [20]
Key Driver Mutations:
-
BRAF Mutations (50% of melanomas)
- Most common: BRAF V600E mutation (80% of BRAF-mutant cases)
- Alternative: BRAF V600K (10-20%)
- Results in constitutive activation of MAPK (RAS-RAF-MEK-ERK) pathway
- Leads to unchecked proliferation
- Therapeutic target: BRAF inhibitors (vemurafenib, dabrafenib, encorafenib) [21]
-
NRAS Mutations (20% of melanomas)
- Mutually exclusive with BRAF mutations
- Also activates MAPK pathway upstream of BRAF
- Currently limited targeted therapy options [22]
-
KIT Mutations (rare, less than 5%)
- Predominantly in acral and mucosal melanomas
- Receptor tyrosine kinase mutations
- Potential target for imatinib/sunitinib [23]
-
NF1 Loss (10-15%)
- Tumour suppressor gene
- Associated with more aggressive phenotype [24]
Tumour Suppressor Inactivation:
- CDKN2A (p16INK4a): Cell cycle checkpoint dysfunction
- PTEN: Negative regulator of PI3K-AKT pathway
- TP53: "Guardian of the genome" dysfunction (less common in melanoma than other cancers)
Growth Phases
Melanoma progression classically follows a two-phase model:
-
Radial Growth Phase (RGP)
- Horizontal spread within epidermis and superficial dermis
- Cells lack capacity for metastasis
- Melanoma in-situ falls into this category
- Potentially curable with simple excision
-
Vertical Growth Phase (VGP)
- Invasion deep into dermis and subcutaneous tissue
- Acquisition of metastatic potential
- Depth of invasion (Breslow thickness) becomes critical prognostic factor
- Lymphatic and haematogenous spread possible
Exam Detail: Molecular Detail for Postgraduate Exams:
The MAPK pathway activation (via BRAF or NRAS) is necessary but not sufficient for melanoma development. Secondary events are required:
- Loss of p16 (CDKN2A) removes cell cycle braking
- Activation of PI3K-AKT pathway (via PTEN loss or receptor activation) provides survival signals
- Telomerase reactivation (TERT promoter mutations) enables immortalization [25]
The tumour microenvironment plays a crucial role:
- Melanomas evade immune surveillance through PD-L1 expression
- This forms the rationale for anti-PD-1 immunotherapy
- Tumour-infiltrating lymphocytes (TILs) are a positive prognostic marker [26]
4. Clinical Presentation
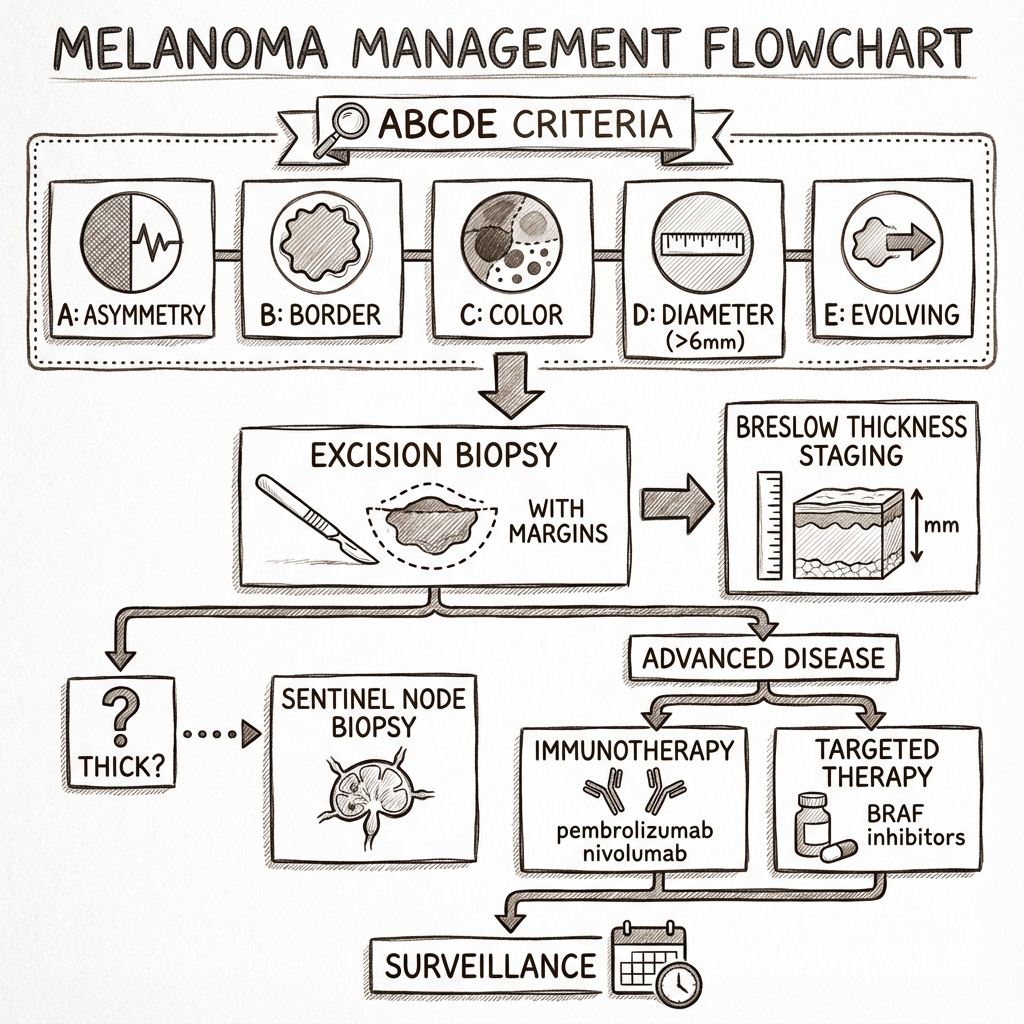
The ABCDE Rule for Melanoma Recognition
This mnemonic remains the cornerstone of clinical assessment:
- A - Asymmetry: One half does not mirror the other half
- B - Border: Irregular, scalloped, or poorly defined edges
- C - Colour: Variegated appearance with multiple shades (tan, brown, black, red, white, blue)
- D - Diameter: > 6mm (size of pencil eraser), though melanomas can be smaller
- E - Evolution: Change in size, shape, colour, elevation, or new symptoms (itching, bleeding)
Evolution is the single most sensitive criterion. [27] Any changing pigmented lesion warrants evaluation.
Additional Clinical Signs
"Ugly Duckling Sign": In patients with multiple naevi, the lesion that looks different from all others is suspicious. [28]
Dermoscopy Features (Pattern Analysis):
- Atypical pigment network
- Irregular dots and globules
- Blue-white veil (regression)
- Irregular streaks (radial streaming)
- Peripheral brown structureless areas
- Multiple colours (> 3)
Histological Subtypes
Understanding the subtypes is essential for both exams and clinical practice, as they differ in demographics, anatomical sites, and prognosis:
1. Superficial Spreading Melanoma (SSM) - 70%
- Most common subtype
- Flat macule or slightly raised plaque
- Irregular shape and colour
- Extended radial growth phase before vertical growth
- Site: Back in men, legs in women
- Age: 30-60 years
- Prognosis: Moderate (depends on Breslow depth)
2. Nodular Melanoma (NM) - 15-20%
- Rapidly growing, raised nodule
- Presentation: "Dome-shaped" lesion, often blue-black or amelanotic
- No preceding radial growth phase—enters vertical growth immediately
- High risk: Aggressive behaviour, frequently ulcerated
- May not fulfill ABCD criteria (can be symmetric) but fulfills E (evolution)
- Site: Any site, commonly trunk
- Prognosis: Poor due to deep invasion at presentation
Clinical Pearl: Exam Pearl: Nodular melanoma is the subtype most likely to be amelanotic (non-pigmented), appearing pink or red. This delays diagnosis. Maintain high index of suspicion for any rapidly growing nodule. [29]
3. Lentigo Maligna Melanoma (LMM) - 10%
- Arises from lentigo maligna (melanoma in-situ) on chronically sun-damaged skin
- Presentation: Large (often > 3cm) irregularly pigmented patch/macule
- Prolonged in-situ phase (sometimes decades) before invasion
- Site: Face, neck, ears (sun-exposed areas in elderly)
- Age: > 65 years typically
- Historical name: "Hutchinson's melanotic freckle"
- Prognosis: Good if excised during in-situ phase, but challenging surgical margins on face
4. Acral Lentiginous Melanoma (ALM) - 5-10%
- Occurs on palms, soles, and subungual (nail bed) sites
- Most common melanoma subtype in patients with darker skin (Asian, African, Hispanic populations) [30]
- Not UV-related (different pathogenesis)
- Presentation:
- "Palmar/plantar: Irregular brown-black macule"
- "Subungual: Longitudinal melanonychia (pigmented band in nail)"
- Hutchinson's Sign: Extension of pigment onto proximal or lateral nail fold (cuticle) - highly specific for subungual melanoma [31]
- Prognosis: Often diagnosed late due to hidden location; poorer outcomes
Exam Detail: Viva Point: When asked about acral lentiginous melanoma, mention Bob Marley—the famous case of subungual melanoma misdiagnosed as a football injury. This illustrates the importance of biopsy for pigmented nail lesions, especially with Hutchinson's sign. [31]
5. Amelanotic Melanoma - 2-8%
- Lacks melanin pigment
- Presentation: Pink, red, or skin-coloured nodule or patch
- Often mistaken for benign lesions (granuloma, dermatofibroma, BCC)
- Diagnostic challenge: Dermoscopy less useful
- Biopsy threshold should be low for atypical vascular lesions
6. Desmoplastic Melanoma - Rare (less than 4%)
- Spindled melanocytes with fibrous stroma
- Site: Head and neck (sun-exposed)
- Often amelanotic
- Behaviour: Neural/perineural invasion common
- Lower risk of lymph node metastasis but higher local recurrence
- Management: Wide excision with margin assessment critical
Symptoms
Many early melanomas are asymptomatic. Late features suggesting advanced disease include:
- Itching (present in 20-30%) [32]
- Bleeding or oozing
- Pain (uncommon, suggests nerve involvement)
- Ulceration (poor prognostic sign)
Regional and Distant Spread
Regional Disease:
- In-transit metastases: Satellite lesions > 2cm from primary site but before draining lymph node basin (dermal/subdermal lymphatic spread)
- Palpable lymphadenopathy: Firm, non-tender nodes
Distant Metastases (Stage IV):
- Skin/Subcutaneous: Painless nodules (common first distant site)
- Lymph Nodes: Non-regional nodal disease
- Lungs: Cough, dyspnoea, haemoptysis
- Brain: Headache, seizures, focal neurology (melanoma has highest propensity for brain metastases among solid tumours) [33]
- Liver: Abdominal pain, hepatomegaly, jaundice
- Bone: Pathological fractures, hypercalcaemia
5. Differential Diagnosis
The clinical differential depends on the appearance of the lesion:
For Pigmented Lesions:
| Differential | Key Distinguishing Features |
|---|---|
| Benign Melanocytic Naevus | Symmetric, uniform colour, stable over time, less than 6mm |
| Dysplastic (Atypical) Naevus | Irregular borders, variable colour, BUT stable; requires monitoring |
| Seborrhoeic Keratosis | "Stuck-on" appearance, waxy surface, keratin plugs (milia-like cysts on dermoscopy) |
| Pigmented Basal Cell Carcinoma | Rolled/pearly border, telangiectasia, central ulceration |
| Solar Lentigo | Uniform tan-brown macule on sun-exposed skin, well-demarcated, stable |
| Blue Naevus | Blue-grey colour, stable, Mongolian spot-like appearance |
| Dermatofibroma | Firm, dimple sign positive (pinches inward), homogenous |
For Amelanotic Lesions:
- Pyogenic Granuloma: Rapidly growing vascular nodule, often post-trauma, bleeds easily
- Squamous Cell Carcinoma: Scaly, keratotic, on sun-damaged skin
- Keratoacanthoma: Volcano-like morphology with central keratin plug, rapid growth then regression
- Spitz Naevus: Pink dome-shaped nodule, typically in children/young adults
Clinical Pearl: "When in doubt, cut it out": The threshold for excision biopsy should be low for any atypical pigmented lesion. The consequences of missing a melanoma far outweigh the minor scar from excising a benign lesion. [34]
6. Investigations
Diagnostic Approach
1. Clinical Examination
- Full skin examination (including scalp, interdigital spaces, genitalia, mucosal surfaces)
- Dermoscopy (dermatoscopy) of suspicious lesion
- Regional lymph node palpation
- Fundoscopy (rarely, for ocular melanoma screening in familial cases)
2. Excision Biopsy - GOLD STANDARD [35]
Technique:
- Complete excisional biopsy with 2mm clinical margin
- Elliptical excision with long axis along line of lymphatic drainage
- Include subcutaneous fat
- Orient specimen for pathologist (mark deep margin, orientation suture)
- Avoid: Punch biopsy, shave biopsy, incisional biopsy
- Risk of sampling error (missing invasive component)
- Disrupts architecture needed for accurate Breslow measurement
- Possible (though unproven) risk of seeding
Exceptions where partial biopsy acceptable:
- Lentigo maligna on face (where wide initial excision impractical): Punch/incisional to confirm diagnosis
- Very large lesions where complete excision impractical
- Acral lesions where function preservation critical
Histopathology Report - Essential Elements
The pathology report should detail:
-
Breslow Thickness (in mm)
- Measured from granular layer of epidermis to deepest invasive tumour cell
- Most important prognostic factor [36]
- Determines surgical margins and need for sentinel lymph node biopsy
-
Ulceration (present/absent)
- Independent adverse prognostic factor
- Upstages tumours in AJCC classification
-
Mitotic Rate (per mm²)
- ≥1 mitosis/mm² is adverse prognostic factor
- Particularly important in thin melanomas
-
Clark Level (I-V) - Historical
- Describes anatomical depth (epidermis, papillary dermis, reticular dermis, etc.)
- Largely replaced by Breslow thickness but still reported
-
Margins (distance to closest margin)
- Initial excision: Should be clear, but narrow margins acceptable
- Re-excision margins determined by Breslow depth
-
Tumour-Infiltrating Lymphocytes (TILs) (brisk/non-brisk/absent)
- Favourable prognostic marker [26]
-
Lymphovascular Invasion (present/absent)
- Indicates higher metastatic risk
-
Regression (present/absent)
- Histological evidence of immune-mediated tumour destruction
- May result in underestimation of original Breslow depth
-
Microsatellitosis (present/absent)
- Tumour nests > 0.05mm from main tumour mass
- Upstages to T classification
Staging Investigations
Indications for staging CT/PET-CT:
- Clinical stage IIB or higher (Breslow > 2mm with ulceration, or > 4mm)
- Palpable lymphadenopathy
- Symptoms suggesting metastatic disease
Imaging Modalities:
| Investigation | Use | Findings |
|---|---|---|
| CT Chest/Abdomen/Pelvis with contrast | Staging for stage IIC+ | Lung nodules, liver lesions, nodal disease |
| PET-CT | Most sensitive for distant metastases | Increased FDG uptake in metastatic foci; superior to CT alone [37] |
| MRI Brain | High-risk patients (stage III/IV) | Brain metastases (CT often misses small lesions) [33] |
| Ultrasound | Regional lymph node assessment | Hypoechoic nodes, loss of hilar architecture |
Baseline Bloods (for advanced disease):
- FBC (anaemia)
- U&E, LFTs (liver metastases)
- LDH (elevated LDH is independent poor prognostic factor in stage IV disease) [38]
- Calcium (bone metastases/hypercalcaemia)
Genetic/Molecular Testing
BRAF Mutation Testing (for stage III/IV disease):
- Identifies V600E/K mutations
- Essential before considering BRAF inhibitor therapy
- Can be performed on paraffin-embedded tissue
Additional Panel Testing (research/selected cases):
- NRAS, KIT, NTRK, MET mutations
- PD-L1 expression (less predictive in melanoma than other cancers)
- Tumour Mutational Burden (TMB) - correlates with immunotherapy response
Germline Testing (for familial melanoma):
- CDKN2A, CDK4 mutations
- Consider if: ≥3 melanomas in patient or family, young age (less than 30), multiple primary melanomas
7. Staging - AJCC 8th Edition (2017)
The American Joint Committee on Cancer (AJCC) 8th Edition staging system for cutaneous melanoma incorporates Breslow thickness, ulceration, and nodal/metastatic burden. [39]
TNM Classification
T (Primary Tumour)
| T Stage | Breslow Thickness | Additional Criteria |
|---|---|---|
| Tis | In situ (melanoma in-situ) | - |
| T1 | ≤1.0 mm | T1 |
| a: less than 0.8mm without ulceration T1 | ||
| b: 0.8-1.0mm or less than 0.8mm with ulceration | ||
| T2 | > 1.0-2.0 mm | T2 |
| a: without ulceration T2 | ||
| b: with ulceration | ||
| T3 | > 2.0-4.0 mm | T3 |
| a: without ulceration T3 | ||
| b: with ulceration | ||
| T4 | > 4.0 mm | T4 |
| a: without ulceration T4 | ||
| b: with ulceration |
N (Regional Lymph Nodes)
| N Stage | Clinical Nodes | Pathological Nodes |
|---|---|---|
| N0 | No regional nodes | No regional nodes |
| N1 | 1 node or in-transit/satellite/microsatellite disease | N1 |
| a: 1 clinically occult N1 | ||
| b: 1 clinically detected N1 | ||
| c: No nodes, in-transit/satellite/microsatellite | ||
| N2 | 2-3 nodes or in-transit/satellite/microsatellite with 1 node | N2 |
| a: 2-3 clinically occult N2 | ||
| b: 2-3, ≥1 clinically detected N2 | ||
| c: 1 node + in-transit/satellite/microsatellite | ||
| N3 | 4+ nodes or matted nodes or in-transit/satellite/microsatellite with 2+ nodes | N3 |
| a: 4+ clinically occult N3 | ||
| b: 4+, ≥1 clinically detected, or matted N3 | ||
| c: 2+ nodes + in-transit/satellite/microsatellite |
M (Distant Metastasis)
| M Stage | Site | LDH |
|---|---|---|
| M0 | No distant metastasis | - |
| M1a | Distant skin, soft tissue, non-regional nodes | M1a(0): Normal LDH M1a(1): Elevated LDH |
| M1b | Lung ± M1a sites | M1b(0): Normal LDH M1b(1): Elevated LDH |
| M1c | Non-CNS visceral | M1c(0): Normal LDH M1c(1): Elevated LDH |
| M1d | CNS metastases | M1d(0): Normal LDH M1d(1): Elevated LDH |
Prognostic Stage Groups
| Stage | T | N | M | Approximate 5-Year Survival |
|---|---|---|---|---|
| 0 | Tis | N0 | M0 | ~99% |
| IA | T1a | N0 | M0 | 99% |
| IB | T1b/T2a | N0 | M0 | 95-97% |
| IIA | T2b/T3a | N0 | M0 | 89-94% |
| IIB | T3b/T4a | N0 | M0 | 82-87% |
| IIC | T4b | N0 | M0 | 75-82% |
| IIIA | T1a/b-T2a | N1a/N2a | M0 | 88-93% |
| IIIB | T0-T2b | N1b/c or N2b | M0 | 68-83% |
| IIIC | T3a/b-T4b | N1a-N2c or Any T with N3a/b/c | M0 | 40-68% |
| IIID | Any T | N3b/c | M0 | 24-32% |
| IV | Any T | Any N | M1 | 10-27% (varies by M subtype) |
Exam Detail: Key Changes in AJCC 8th Edition (from 7th):
- T1 threshold changed from 1mm to 0.8mm for ulceration/mitotic rate consideration
- Mitotic rate removed as T staging criterion (remains prognostic marker)
- Satellite/in-transit/microsatellite metastases now grouped as N1c-N3c (previously M category)
- Stage III subdivided into A/B/C/D (previously only A/B/C)
- LDH elevation incorporated into M1 substaging [39]
Viva Point: Stage IIC disease (T4b N0) has worse prognosis than Stage IIIA/B (node-positive disease with thinner primaries), highlighting the importance of T category. [39]
8. Management
Management of melanoma is multidisciplinary, involving dermatology, plastic/surgical oncology, medical oncology, radiation oncology, and specialist nurses. Treatment is stratified by stage.
A. Surgical Management
Wide Local Excision (WLE)
Following diagnostic excision biopsy confirming invasive melanoma, definitive wide local excision is performed to achieve oncologically adequate margins.
Margin Guidelines (based on Breslow thickness): [40]
| Breslow Thickness | Recommended Clinical Margin |
|---|---|
| In situ | 5mm |
| ≤1mm | 1cm |
| 1.01-2mm | 1-2cm |
| > 2mm | 2cm |
Rationale:
- Removes subclinical radial extension of tumour
- Reduces local recurrence risk from 20% → less than 5% [40]
- Margins based on large randomized trials showing no benefit to margins > 2cm even for thick melanomas
Technique:
- Elliptical excision centered on biopsy scar
- Margins measured from palpable tumour edge/biopsy scar
- Excision to deep fascia (not including fascia unless directly invaded)
- Primary closure when possible; skin graft or flap for larger defects
- Orient long axis along lymphatic drainage for potential future lymphadenectomy
Special Sites:
- Face/Digits: Functional preservation may necessitate narrower margins; Mohs micrographic surgery considered for lentigo maligna
- Subungual: Digit amputation at joint proximal to lesion vs nail unit excision
Clinical Pearl: Common Error: Taking wider margins than guideline-recommended does NOT improve survival but DOES increase morbidity (poor wound healing, need for reconstruction). Stick to evidence-based margins. [40]
Sentinel Lymph Node Biopsy (SLNB)
Indication: [41]
- Breslow thickness > 1mm, OR
- Breslow thickness 0.8-1mm with ulceration or other adverse features
- Clinical N0 (no palpable nodes)
Technique:
- Lymphoscintigraphy (technetium-99m sulfur colloid) and/or blue dye injected around primary site pre-operatively
- Intra-operative gamma probe identifies "sentinel" node(s) in draining basin
- Selective excision of hot/blue nodes
- Histopathological examination (H&E + immunohistochemistry for S100, Melan-A)
Purpose:
- Staging: Most accurate predictor of nodal basin involvement
- Prognostic: Positive SLN associated with worse disease-free and overall survival
- Therapeutic: Debatable whether SLNB improves survival (see below)
MSLT-II Trial Results (2017): [42]
- Completion lymph node dissection (CLND) after positive SLNB did NOT improve melanoma-specific survival vs observation with ultrasound surveillance
- This has shifted practice away from routine CLND
- Current approach: Positive SLNB → Close nodal surveillance + consider adjuvant systemic therapy
Morbidity:
- Low: Seroma, wound infection, allergic reaction to blue dye
- Lymphoedema risk much lower than with complete lymphadenectomy (less than 5% vs 20%)
Completion Lymph Node Dissection (CLND)
Indications (much more limited post-MSLT-II):
- Clinically/radiologically detected nodal disease (palpable nodes, positive imaging)
- Multiple positive sentinel nodes in high-risk patients (individualised decision)
- Patient preference after counseling on MSLT-II data
Morbidity:
- Lymphoedema (20-30%)
- Seroma, wound breakdown
- Nerve injury (sensory > motor)
B. Adjuvant Systemic Therapy (Stage III)
Historically, stage III melanoma (regional node involvement) had poor outcomes with surgery alone, with 40-70% recurrence risk at 5 years. The advent of effective systemic agents has revolutionized adjuvant therapy.
1. Adjuvant Immunotherapy
Pembrolizumab (anti-PD-1):
- Trial: KEYNOTE-054 (2018) [43]
- Dose: 200mg IV every 3 weeks for 1 year
- Results: 12-month recurrence-free survival 75% vs 61% (placebo)
- Approval: FDA/EMA approved for stage III melanoma
- Toxicity: Immune-related adverse events (colitis, hepatitis, thyroiditis, pneumonitis) in 30-40%; grade 3-4 in ~15%
Nivolumab (anti-PD-1):
- Trial: CheckMate-238 (2017) [44]
- Dose: 3mg/kg IV every 2 weeks (or 480mg every 4 weeks) for 1 year
- Results: Superior recurrence-free survival vs ipilimumab (70% vs 60% at 18 months)
- Approval: FDA/EMA approved for stage III and some high-risk stage IIB/C
- Toxicity: Better tolerated than ipilimumab (fewer grade 3-4 events)
Ipilimumab (anti-CTLA-4):
- Older adjuvant option, now largely superseded by anti-PD-1 agents
- Higher toxicity (grade 3-4 immune AEs in 40-50%)
Combination Ipilimumab + Nivolumab:
- Trial: CheckMate-915 (2021) - Did NOT show benefit over nivolumab alone in adjuvant setting [45]
- High toxicity limits use
2. Adjuvant Targeted Therapy (BRAF-mutant only)
Dabrafenib + Trametinib (BRAF + MEK inhibitors):
- Trial: COMBI-AD (2017) [46]
- Indication: Stage III melanoma with BRAF V600E/K mutation
- Regimen: Dabrafenib 150mg BD + Trametinib 2mg OD for 1 year
- Results: 3-year relapse-free survival 58% vs 39% (placebo)
- Approval: FDA/EMA approved
- Toxicity: Pyrexia (50%), fatigue, nausea, arthralgias; generally well-tolerated
Choice of Adjuvant Therapy:
- BRAF-mutant: Either anti-PD-1 monotherapy OR dabrafenib+trametinib (no head-to-head trial; choice based on patient factors, toxicity profile)
- BRAF-wildtype: Anti-PD-1 monotherapy
C. Treatment of Advanced/Metastatic Melanoma (Stage IV)
The treatment of metastatic melanoma has undergone a paradigm shift over the past decade. Prior to 2011, median survival was less than 1 year with chemotherapy (dacarbazine). Modern immunotherapy and targeted therapy have achieved long-term durable responses in a significant proportion of patients. [4]
1. First-Line Immunotherapy
Nivolumab + Ipilimumab (Combination):
- Trial: CheckMate-067 (2017) [47]
- Regimen: Nivolumab 1mg/kg + Ipilimumab 3mg/kg IV every 3 weeks x4, then nivolumab 480mg every 4 weeks
- Results:
- "Overall response rate: 58%"
- "Median progression-free survival: 11.5 months"
- 5-year overall survival: 52% (unprecedented for metastatic melanoma)
- Best for: High tumour burden, visceral disease, need for rapid response
- Toxicity: Grade 3-4 immune-related AEs in 55-60%
Pembrolizumab or Nivolumab (Monotherapy):
- Trials: KEYNOTE-006, CheckMate-067 [43, 47]
- Results:
- "Overall response rate: 40-45%"
- "Median PFS: 6-7 months"
- 5-year OS: 34-44%
- Best for: Lower disease burden, elderly/frail, those intolerant of combination
- Toxicity: Grade 3-4 AEs in 10-15%
Exam Detail: Mechanism of Action - Checkpoint Inhibitors:
Melanoma cells upregulate PD-L1 (programmed death-ligand 1) which binds to PD-1 receptors on T-cells, suppressing anti-tumour immunity ("immune escape").
- Anti-PD-1 antibodies (pembrolizumab, nivolumab) block PD-1 receptor on T-cells → "release the brakes" → T-cell activation and tumour killing
- Anti-CTLA-4 antibodies (ipilimumab) block CTLA-4 (another inhibitory checkpoint) → enhanced T-cell priming in lymph nodes
Combination rationale: Dual checkpoint blockade produces synergistic activity but also additive toxicity. [48]
2. First-Line Targeted Therapy (BRAF V600-mutant only)
Indication: BRAF V600E or V600K mutation confirmed (~50% of melanomas)
BRAF + MEK Inhibitor Combinations (superior to BRAF inhibitor monotherapy):
| Combination | Dose | Key Trial | ORR | Median PFS |
|---|---|---|---|---|
| Dabrafenib + Trametinib | 150mg BD + 2mg OD | COMBI-d/v [49] | 69% | 11 months |
| Vemurafenib + Cobimetinib | 960mg BD + 60mg OD | coBRIM [50] | 70% | 12.3 months |
| Encorafenib + Binimetinib | 450mg OD + 45mg BD | COLUMBUS [51] | 63% | 14.9 months |
5-year OS: ~34% across combinations [52]
Advantages of Targeted Therapy:
- Rapid response (often within weeks)
- High response rates
- Suitable for patients with poor performance status or those unsuitable for immunotherapy
Disadvantages:
- Acquired resistance develops (median duration of response ~12 months)
- Not curative in most cases
- Toxicity: Pyrexia, arthralgia, rash, photosensitivity, diarrhoea
- Specific AE: Risk of cutaneous squamous cell carcinoma (~15-30% with BRAF monotherapy, reduced with MEK inhibitor combination)
Sequencing Question (BRAF-mutant patients):
- No definitive answer from prospective trials
- Immunotherapy first generally preferred (potential for durable long-term response)
- Targeted therapy first considered if: Symptomatic visceral disease requiring rapid response, brain metastases, contraindication to immunotherapy
- Cross-resistance between therapies is minimal—most patients receive both in sequence
3. Second-Line and Beyond
- If progression on anti-PD-1 monotherapy → Consider ipilimumab + nivolumab combination
- If progression on immunotherapy → BRAF/MEK inhibitors (if BRAF-mutant)
- If progression on BRAF/MEK → Immunotherapy (can still respond)
- Clinical trials of novel agents
4. Special Scenarios
Brain Metastases:
- Previously universally fatal; now treatable
- Asymptomatic small lesions: Immunotherapy (combination nivo+ipi shows ~50% intracranial response) or BRAF/MEK inhibitors can achieve CNS responses [53]
- Symptomatic/Large lesions: Stereotactic radiosurgery (SRS) ± systemic therapy
- Avoid whole-brain radiotherapy (poor outcomes, cognitive impairment) unless multiple unsuitable for SRS
Mucosal and Uveal Melanoma:
- Different biology (rare BRAF mutations; KIT, BAP1, GNAQ/GNA11 mutations)
- Generally poorer response to immunotherapy and targeted therapy
- Consider MEK inhibitors, KIT inhibitors (if KIT-mutant)
D. Radiation Therapy
Limited role in melanoma (historically considered "radioresistant"):
Indications:
- Adjuvant nodal radiation: Post-lymphadenectomy in high-risk cases (extranodal extension, multiple nodes) - reduces locoregional recurrence [54]
- Desmoplastic melanoma: Adjuvant radiation after WLE reduces local recurrence
- Palliation: Bone metastases, brain metastases (stereotactic radiosurgery), symptomatic soft tissue disease
- Definitive treatment (rare): Lentigo maligna on face where surgery disfiguring
E. Follow-Up and Surveillance
Purpose:
- Early detection of recurrence
- Detection of new primary melanomas (3-5% risk)
- Skin cancer screening (increased NMSC risk)
- Psychosocial support
Recommended Schedule (NICE, NCCN): [55]
| Stage | Visits | Duration | Imaging |
|---|---|---|---|
| IA-IIA | Every 6-12 months | 5 years, then annual | Clinical exam only |
| IIB-IIC | Every 3-6 months | 2 years, then 6-monthly to year 5 | Consider CT/PET annually for stage IIC |
| III | Every 3-4 months | 2 years, then 6-monthly to year 5, then annual | CT ± PET every 6-12 months for 3 years |
| IV (NED post-treatment) | Every 3-4 months | 2 years, then tailored | CT ± PET every 6 months |
Components of Follow-Up:
- Full skin examination
- Regional nodal palpation
- Symptom review
- Imaging as above
- Patient education on self-examination
9. Complications
Complications of Disease
| Complication | Frequency | Clinical Features | Management |
|---|---|---|---|
| Local recurrence | 5-10% post-WLE | Nodule/plaque at excision site | Re-excision, consider adjuvant RT |
| In-transit metastases | 5-10% (stage IIB+) | Satellite lesions between primary and nodes | Isolated limb perfusion, systemic therapy |
| Regional nodal disease | 20-40% (stage IB-II) | Lymphadenopathy | Lymphadenectomy or systemic therapy |
| Distant metastases | Variable by stage | Site-specific symptoms | Systemic therapy ± local therapy |
| Brain metastases | 40-60% (stage IV) | Headache, seizures, focal neurology | SRS, systemic therapy, steroids |
| Leptomeningeal disease | Rare, late | Meningism, cranial nerve palsies | Palliative; very poor prognosis |
| Spinal cord compression | 5% (stage IV) | Back pain, motor/sensory deficit, cauda equina | Emergency MRI → RT/surgery + steroids |
| Malignant pleural effusion | 10-15% (stage IV with lung mets) | Dyspnoea | Drainage, pleurodesis |
| Hypercalcaemia | 5-10% (bone mets) | Confusion, polyuria, constipation | IV fluids, bisphosphonates, treat underlying disease |
Complications of Treatment
Surgical:
- Wound infection, seroma, dehiscence
- Lymphoedema (post-lymphadenectomy): 20-30%
- Nerve injury (sensory/motor)
- Functional impairment (digital amputation, facial surgery)
Immunotherapy - Immune-Related Adverse Events (irAEs):
| Organ System | Manifestation | Frequency | Management |
|---|---|---|---|
| GI | Diarrhoea, colitis | 10-30% | Hold therapy, steroids ± infliximab if severe |
| Hepatic | Transaminitis, hepatitis | 5-10% | Hold therapy, steroids |
| Endocrine | Hypothyroidism, hyperthyroidism, hypophysitis, adrenal insufficiency | 10-20% | Hormone replacement, may continue therapy |
| Pulmonary | Pneumonitis | 2-5% | Hold/discontinue therapy, steroids |
| Skin | Rash, vitiligo, pruritus | 40-50% | Topical steroids, antihistamines |
| Rheumatologic | Arthralgia, myalgia, myositis | 5-10% | NSAIDs, steroids if severe |
| Neurologic | Neuropathy, encephalitis, myasthenia | less than 1% | Discontinue therapy, high-dose steroids |
Key Principles:
- Most irAEs occur within first 6 months but can occur anytime (even post-treatment)
- Grade 1-2: Supportive care ± topical/local therapy
- Grade 3-4: Hold immunotherapy, high-dose steroids (1-2mg/kg prednisolone)
- Most are reversible; some endocrinopathies are permanent
- Vitiligo is paradoxically a favourable prognostic sign (immune activation) [56]
Targeted Therapy (BRAF/MEK inhibitors):
- Pyrexia (50%): Dose reduction, NSAIDs, steroids
- Arthralgia/myalgia (40%): NSAIDs
- Photosensitivity (30%): Sun protection
- Cutaneous SCC (15-30% BRAF monotherapy; less than 5% with BRAF+MEK): Excision
- GI toxicity: Diarrhoea (30%), nausea
- Cardiac: Reduced LVEF (rare but monitor with echo)
- Ophthalmologic: Uveitis, retinal vein occlusion (MEK inhibitors)
10. Prognosis & Outcomes
Prognosis in melanoma is heavily dependent on stage at presentation. The single most important factor for localized disease is Breslow thickness. [36]
Prognostic Factors
Primary Tumour (T Stage):
- Breslow thickness: Continuous variable; each mm increase worsens prognosis
- Ulceration: Independent poor prognostic factor (upstages)
- Mitotic rate: ≥1/mm² worse prognosis
- Anatomic site: Axial (trunk, head/neck) worse than extremity; subungual worse overall
- Age: Older age (> 60) associated with worse outcomes
- Sex: Males have worse prognosis than females (possibly behavioural/biological factors)
Nodal Disease (N Stage):
- Number of nodes: More nodes = worse prognosis
- Tumour burden: Microscopic (non-palpable) better than macroscopic (palpable)
- Extranodal extension: Presence worsens prognosis
Distant Metastases (M Stage):
- Site: M1a (skin/soft tissue/non-regional nodes) best; M1d (CNS) worst
- LDH: Elevated LDH is independent poor prognostic factor [38]
- Number of metastatic sites: Oligometastatic better than widespread disease
Other Factors:
- Tumour-infiltrating lymphocytes (TILs): Brisk TILs associated with better prognosis [26]
- BRAF mutation status: Controversial; some studies suggest worse prognosis, but confounded by availability of targeted therapy
- Performance status: ECOG 0-1 vs 2-4
Survival by Stage (AJCC 8th Edition)
| Stage | 5-Year Overall Survival | 10-Year Overall Survival |
|---|---|---|
| IA | 99% | 98% |
| IB | 95-97% | 88-94% |
| IIA | 89-94% | 82-88% |
| IIB | 82-87% | 75-82% |
| IIC | 75-82% | 60-75% |
| IIIA | 88-93% | 73-88% |
| IIIB | 68-83% | 52-77% |
| IIIC | 40-68% | 24-60% |
| IIID | 24-32% | less than 20% |
| IV | 10-27% (pre-modern therapy) | less than 10% |
| IV | ~50% (with modern immunotherapy - CheckMate-067 5-year data) [47] | Emerging data |
Key Message: Thin melanomas (less than 1mm) without ulceration have excellent prognosis (> 95% cure rate). Thick melanomas (> 4mm) and node-positive disease have significant recurrence risk, but modern adjuvant therapies are changing this landscape. [43, 46]
Recurrence Patterns
- Timing: 80% of recurrences occur within 3 years; late recurrences (> 10 years) well-documented
- Sites: Local/in-transit (30%), regional nodes (30%), distant (40%)
- Salvage: Oligometastatic recurrence amenable to local therapy (resection, SRS) with curative intent in selected cases
11. Prevention & Screening
Primary Prevention
UV Protection:
- Sunscreen: Broad-spectrum SPF 30+ applied generously and reapplied every 2 hours
- Clothing: Wide-brimmed hats, long sleeves, UV-protective clothing (UPF 50+)
- Avoidance: Seek shade 10am-4pm; avoid sunbeds entirely
- Childhood protection: Critical—childhood sunburns have strongest association with adult melanoma risk [17]
Public Health Campaigns:
- Australia's "Slip, Slop, Slap, Seek, Slide" campaign associated with decreased melanoma incidence in younger cohorts
- School-based education programmes
Chemoprevention:
- No proven effective agents for general population
- Oral nicotinamide (vitamin B3) 500mg BD reduces NMSC but not melanoma [57]
Secondary Prevention (Early Detection)
Self-Skin Examination:
- Monthly full-body examination
- ABCDE criteria
- "Ugly Duckling" sign
- Partner-assisted for difficult-to-see areas
Clinical Screening:
- No universal population screening recommended (insufficient evidence) [58]
- Targeted screening for high-risk groups:
- Personal history of melanoma
- Family history (≥2 first-degree relatives)
- Dysplastic naevus syndrome
-
100 naevi
- Immunosuppressed patients
- Genetic syndromes (xeroderma pigmentosum, CDKN2A mutation carriers)
Total Body Photography and Digital Dermoscopy:
- Serial imaging to detect new/changing lesions
- Reduces benign biopsy rate while maintaining sensitivity [59]
Tertiary Prevention
Surveillance Post-Treatment: See Follow-Up section
Genetic Counseling:
- For patients with familial melanoma syndrome
- CDKN2A mutation carriers: 60-90% lifetime melanoma risk [14]
- Pancreatic cancer screening also recommended (CDKN2A associated with pancreatic cancer)
12. Key Guidelines
Major Society Guidelines
NICE NG14 (2015, updated 2022): Melanoma: assessment and management [55]
- Recommendations on referral thresholds, excision margins, SLNB, staging, adjuvant therapy
- 2-week-wait referral for suspected melanoma
ESMO Clinical Practice Guidelines (2022): Cutaneous melanoma [60]
- European consensus on diagnosis, treatment, follow-up
- Incorporates latest immunotherapy and targeted therapy data
NCCN Guidelines (2023): Melanoma: Cutaneous [61]
- US-based comprehensive guidelines
- Detailed treatment algorithms by stage
- Includes genetic testing recommendations
AAD/ASCO Guidelines (2019): Management of Primary Cutaneous Melanoma [62]
- Joint dermatology/oncology consensus
- Focus on surgical margins and SLNB
Australian Cancer Network (2008, update pending): Clinical Practice Guidelines for the Management of Melanoma
- Influential given Australia's experience with melanoma
13. Exam-Focused Content
Common MRCP/MRCS/FRCS Exam Questions
-
"Describe the ABCDE criteria for melanoma"
- A: Asymmetry; B: Border irregular; C: Colour variegated; D: Diameter > 6mm; E: Evolution (changing) [27]
-
"What are the subtypes of melanoma and their key features?"
- SSM (70%): most common, radial then vertical growth
- Nodular (15%): aggressive, rapid vertical growth, no ABCDE
- Lentigo maligna (10%): elderly, sun-damaged skin, face
- Acral lentiginous (5%): palms/soles/nails, Hutchinson's sign, common in darker skin [29,30]
-
"What are the surgical margins for melanoma wide local excision?"
- In-situ: 5mm; ≤1 mm: 1cm; 1-2 mm: 1-2cm; > 2 mm: 2cm [40]
-
"What is the indication for sentinel lymph node biopsy?"
- Breslow > 1mm or 0.8-1mm with ulceration/adverse features [41]
-
"Describe the role of immunotherapy in melanoma"
- Anti-PD-1 (pembrolizumab, nivolumab) and anti-CTLA-4 (ipilimumab) checkpoint inhibitors
- Adjuvant stage III: Improves recurrence-free survival
- Metastatic disease: 5-year survival ~50% with combination [47]
- Mechanism: Release immune checkpoint blockade to allow T-cell killing of tumour [48]
-
"What is Breslow thickness and why is it important?"
- Depth of invasion from granular layer to deepest tumour cell (in mm)
- Single most important prognostic factor
- Determines surgical margins and need for SLNB [36]
-
"Describe the AJCC 8th Edition staging for melanoma"
- T stage: Based on Breslow thickness and ulceration
- N stage: Number and character of nodes (microscopic vs macroscopic)
- M stage: Site of distant metastases and LDH level [39]
-
"What is the Hutchinson sign?"
- Pigment extending from nail matrix onto proximal/lateral nail fold (cuticle)
- Highly specific for subungual melanoma [31]
Viva Points
Viva Point: Opening statement: "Melanoma is a malignant tumour of melanocytes arising from the neural crest. It represents the most lethal form of skin cancer with increasing incidence over recent decades. Early detection via the ABCDE criteria and complete surgical excision offers excellent cure rates for thin lesions, while recent advances in immunotherapy have transformed outcomes for advanced disease."
Key facts to mention:
- Incidence: 16,000 cases/year in UK; 5th most common cancer [6]
- Risk factors: UV exposure (intermittent sunburns), fair skin, > 50 naevi, family history, CDKN2A mutations [12-14]
- Breslow thickness is the single most important prognostic factor [36]
- AJCC 8th Edition staging incorporates thickness, ulceration, nodal burden, LDH [39]
- Surgical management: Wide local excision (margins based on Breslow) + sentinel lymph node biopsy if > 1mm [40,41]
- Adjuvant therapy (stage III): Anti-PD-1 (pembrolizumab/nivolumab) or BRAF+MEK inhibitors (if BRAF-mutant) improve recurrence-free survival [43,46]
- Metastatic disease: Immunotherapy (nivo+ipi combination achieves 52% 5-year OS) or BRAF+MEK inhibitors [47]
- Guidelines: NICE NG14, ESMO 2022, NCCN 2023 [55,60,61]
Common Mistakes in Exams
❌ Stating that melanoma always presents as a pigmented lesion
- Amelanotic melanoma (2-8%) is non-pigmented and easily missed [29]
❌ Recommending punch or shave biopsy for suspected melanoma
- Complete excision biopsy is gold standard to preserve Breslow measurement [35]
❌ Taking excessively wide margins (e.g., 3-5cm for thick melanoma)
- Evidence-based margins are maximum 2cm even for > 4mm Breslow [40]
❌ Performing completion lymphadenectomy for all positive SLNB
- MSLT-II showed no survival benefit; current practice is observation + systemic therapy consideration [42]
❌ Assuming BRAF inhibitors are appropriate for all melanomas
- Only ~50% are BRAF-mutant; must test before prescribing [21]
❌ Forgetting to mention brain metastases in melanoma discussion
- Melanoma has highest propensity for CNS mets among solid tumours (40-60% in stage IV) [33]
Model OSCE/Viva Answers
Q: "A 55-year-old presents with a 1.2cm irregularly pigmented lesion on the back that has changed over 6 months. Describe your approach."
A: "This presentation is concerning for melanoma given the evolution and clinical features. I would approach this systematically:
History: ABCDE features, change timeline, symptoms (itching/bleeding), risk factors (UV exposure, sunburns, skin type, family history, immunosuppression), previous skin cancers.
Examination: Full skin examination including scalp and mucosa; dermoscopy of the lesion looking for atypical network, blue-white veil, irregular dots/globules; regional lymph node examination.
Investigation: Urgent 2-week-wait referral for excision biopsy. Complete excision with 2mm margin preserving orientation. Avoid punch biopsy as it may miss invasive component and disrupts Breslow measurement.
Histopathology will provide Breslow thickness, ulceration status, mitotic rate, margins—essential for staging and subsequent management.
Further Management depends on histology:
- If melanoma confirmed, proceed to wide local excision with margins based on Breslow (1-2cm in this scenario assuming invasive).
- If Breslow > 1mm, discuss sentinel lymph node biopsy for staging.
- Stage III disease: Discuss adjuvant immunotherapy or targeted therapy if BRAF-mutant.
- Arrange staging imaging if high-risk features (CT/PET).
- Long-term surveillance and patient education on self-examination and sun protection."
14. Patient & Layperson Explanation
What is Melanoma?
Melanoma is a serious type of skin cancer that starts in the cells (called melanocytes) that give your skin its colour. Unlike other skin cancers that grow slowly, melanoma can spread to other parts of your body if not caught early, which is why it's so important to detect and treat it quickly.
What Causes Melanoma?
The main cause is damage to your skin from ultraviolet (UV) rays—either from the sun or from sunbeds. Getting sunburned, especially as a child or teenager, significantly increases your risk of developing melanoma later in life.
People with fair skin, lots of moles, red or blonde hair, or a family history of melanoma are at higher risk. But melanoma can affect anyone, regardless of skin colour.
What Should I Look For? (The ABCDE Check)
Examine your skin regularly (monthly is ideal) and look for moles that:
- A - Asymmetry: One half doesn't match the other half
- B - Border: The edges are irregular, ragged, or blurred
- C - Colour: Multiple colours—brown, black, tan, red, white, or blue mixed together
- D - Diameter: Larger than a pencil eraser (6mm), though melanomas can be smaller
- E - Evolution: MOST IMPORTANT—any mole that's changing in size, shape, colour, or starts to itch or bleed
The "Ugly Duckling" rule also helps: If you have several moles and one looks completely different from all the others, get it checked.
What Should I Do If I Find Something Suspicious?
See your doctor urgently (same day or next day if possible). Don't wait. Your doctor will examine the mole and likely refer you to a specialist who will remove it completely to check under a microscope. This is called a biopsy.
Important: The specialist will remove the whole mole, not just a piece of it, because they need to see the full depth to work out the best treatment.
How is Melanoma Treated?
If caught early (thin melanoma):
- A simple operation to remove the melanoma with a margin of healthy skin around it
- In most cases, this completely cures the melanoma
- A small scar is all that remains
- More than 95% of people with thin melanomas are cured
If the melanoma is thicker:
- A wider area of skin is removed during the operation
- The surgeon might also check the nearby lymph glands (using a technique called sentinel node biopsy) to see if the melanoma has spread
- You might be offered additional treatment (called adjuvant therapy) with special drugs that boost your immune system or target specific cancer genes
- These new treatments have dramatically improved outcomes over the past 10 years
If the melanoma has spread to other parts of the body:
- Modern immunotherapy drugs "wake up" your immune system to attack the cancer cells
- These drugs have revolutionized treatment—about half of patients with advanced melanoma are now alive 5 years later, compared to less than 10% before these drugs were available
- Other targeted therapies (for melanomas with specific gene changes) can shrink tumours rapidly
- Treatment is tailored to your specific melanoma using genetic testing
Can Melanoma Be Prevented?
Yes! Here's how to protect yourself:
Sun Protection:
- Use broad-spectrum sunscreen (SPF 30 or higher) every day, even when cloudy
- Reapply every 2 hours and after swimming
- Wear protective clothing—wide-brimmed hats, long sleeves, sunglasses
- Seek shade between 10am and 4pm when the sun is strongest
- NEVER use sunbeds—they increase melanoma risk by 20-60%, especially if you start using them before age 35
Self-Examination:
- Check your skin monthly
- Use a mirror or ask someone to help check your back and scalp
- Take photos of moles to compare over time
- See a doctor for annual skin checks if you're high risk
Protect Children:
- Children's skin is especially vulnerable
- Keep babies under 6 months out of direct sun completely
- Sunburns in childhood are particularly dangerous for future melanoma risk
What's the Outlook?
The outlook depends entirely on how early the melanoma is caught:
- Thin melanomas (less than 1mm deep): Over 95% are cured with surgery alone
- Thicker melanomas or those that have spread to lymph glands: Modern treatments have greatly improved survival, but close monitoring is needed
- Advanced melanoma: New treatments offer hope where there was little before—many people now live for years with good quality of life
The bottom line: Melanoma is preventable (sun protection), detectable (skin checks), and treatable (especially when caught early). Early detection saves lives.
15. References
-
Schadendorf D, et al. Melanoma. Lancet. 2018;392(10151):971-984. doi:10.1016/S0140-6736(18)31559-9
-
Garbe C, Leiter U. Melanoma epidemiology and trends. Clin Dermatol. 2009;27(1):3-9. doi:10.1016/j.clindermatol.2008.09.001
-
Arnold M, et al. Global burden of cutaneous melanoma in 2020 and projections to 2040. JAMA Dermatol. 2022;158(5):495-503. doi:10.1001/jamadermatol.2022.0160
-
Larkin J, et al. Five-year survival with combined nivolumab and ipilimumab in advanced melanoma. N Engl J Med. 2019;381(16):1535-1546. doi:10.1056/NEJMoa1910836
-
Geller AC, et al. Melanoma epidemic: An analysis of six decades of data from the Connecticut Tumor Registry. J Clin Oncol. 2013;31(33):4172-4178. doi:10.1200/JCO.2012.47.3728
-
Cancer Research UK. Melanoma skin cancer statistics. 2022. Available at: https://www.cancerresearchuk.org/health-professional/cancer-statistics/statistics-by-cancer-type/melanoma-skin-cancer
-
Whiteman DC, et al. The growing burden of invasive melanoma: Projections of incidence rates and numbers of new cases in six susceptible populations through 2031. J Invest Dermatol. 2016;136(6):1161-1171. doi:10.1016/j.jid.2016.01.035
-
National Cancer Institute. SEER Cancer Statistics Review 1975-2018. Melanoma of the Skin. 2021.
-
Sung H, et al. Global Cancer Statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2021;71(3):209-249. doi:10.3322/caac.21660
-
Office for National Statistics. Cancer registration statistics, England. 2020.
-
Gershenwald JE, Guy GP. Stemming the rising incidence of melanoma: Calling prevention to action. J Natl Cancer Inst. 2016;108(1):djv381. doi:10.1093/jnci/djv381
-
Gandini S, et al. Meta-analysis of risk factors for cutaneous melanoma: II. Sun exposure. Eur J Cancer. 2005;41(1):45-60. doi:10.1016/j.ejca.2004.10.016
-
Psaty EL, et al. Defining the patient at high risk for melanoma. Int J Dermatol. 2010;49(4):362-376. doi:10.1111/j.1365-4632.2010.04381.x
-
Read J, et al. Germline mutations in melanoma predisposition genes. J Med Genet. 2016;53(1):1-14. doi:10.1136/jmedgenet-2015-103191
-
Ferrone CR, et al. Clinicopathological features of and risk factors for multiple primary melanomas. JAMA. 2005;294(13):1647-1654. doi:10.1001/jama.294.13.1647
-
Kubica AW, Brewer JD. Melanoma in immunosuppressed patients. Mayo Clin Proc. 2012;87(10):991-1003. doi:10.1016/j.mayocp.2012.04.018
-
Dennis LK, et al. Sunburns and risk of cutaneous melanoma: Does age matter? A comprehensive meta-analysis. Ann Epidemiol. 2008;18(8):614-627. doi:10.1016/j.annepidem.2008.04.006
-
Boniol M, et al. Cutaneous melanoma attributable to sunbed use: Systematic review and meta-analysis. BMJ. 2012;345:e4757. doi:10.1136/bmj.e4757
-
Lin JY, Fisher DE. Melanocyte biology and skin pigmentation. Nature. 2007;445(7130):843-850. doi:10.1038/nature05660
-
Hodis E, et al. A landscape of driver mutations in melanoma. Cell. 2012;150(2):251-263. doi:10.1016/j.cell.2012.06.024
-
Davies H, et al. Mutations of the BRAF gene in human cancer. Nature. 2002;417(6892):949-954. doi:10.1038/nature00766
-
Jakob JA, et al. NRAS mutation status is an independent prognostic factor in metastatic melanoma. Cancer. 2012;118(16):4014-4023. doi:10.1002/cncr.26724
-
Curtin JA, et al. Distinct sets of genetic alterations in melanoma. N Engl J Med. 2005;353(20):2135-2147. doi:10.1056/NEJMoa050092
-
Krauthammer M, et al. Exome sequencing identifies recurrent somatic RAC1 mutations in melanoma. Nat Genet. 2012;44(9):1006-1014. doi:10.1038/ng.2359
-
Horn S, et al. TERT promoter mutations in familial and sporadic melanoma. Science. 2013;339(6122):959-961. doi:10.1126/science.1230062
-
Clemente CG, et al. Prognostic value of tumor infiltrating lymphocytes in the vertical growth phase of primary cutaneous melanoma. Cancer. 1996;77(7):1303-1310.
-
Abbasi NR, et al. Early diagnosis of cutaneous melanoma: Revisiting the ABCD criteria. JAMA. 2004;292(22):2771-2776. doi:10.1001/jama.292.22.2771
-
Grob JJ, Bonerandi JJ. The 'ugly duckling' sign: Identification of the common characteristics of nevi in an individual as a basis for melanoma screening. Arch Dermatol. 1998;134(1):103-104. doi:10.1001/archderm.134.1.103
-
Cheung WL, et al. Amelanotic melanoma: A detailed morphologic analysis with clinicopathologic correlation of 75 cases. J Cutan Pathol. 2012;39(1):33-39. doi:10.1111/j.1600-0560.2011.01808.x
-
Bradford PT, et al. Acral lentiginous melanoma: Incidence and survival patterns in the United States, 1986-2005. Arch Dermatol. 2009;145(4):427-434. doi:10.1001/archdermatol.2008.609
-
Levit EK, et al. The ABC rule for clinical detection of subungual melanoma. J Am Acad Dermatol. 2000;42(2 Pt 1):269-274. doi:10.1016/s0190-9622(00)90142-4
-
Paek SC, et al. Defining the survivor of melanoma: Recognition and management of the patient at risk. Semin Oncol. 2007;34(6):501-508. doi:10.1053/j.seminoncol.2007.09.005
-
Fife KM, et al. Determinants of outcome in melanoma patients with cerebral metastases. J Clin Oncol. 2004;22(7):1293-1300. doi:10.1200/JCO.2004.08.140
-
Argenziano G, et al. Dermoscopy of patients with multiple nevi: Improved management recommendations using a comparative diagnostic approach. Arch Dermatol. 2011;147(1):46-49. doi:10.1001/archdermatol.2010.389
-
Sober AJ, et al. Guidelines of care for primary cutaneous melanoma. J Am Acad Dermatol. 2001;45(4):579-586. doi:10.1067/mjd.2001.117569
-
Balch CM, et al. Final version of 2009 AJCC melanoma staging and classification. J Clin Oncol. 2009;27(36):6199-6206. doi:10.1200/JCO.2009.23.4799
-
Danielsen M, et al. Positron emission tomography in the follow-up of cutaneous malignant melanoma patients: A systematic review. Am J Nucl Med Mol Imaging. 2014;4(1):17-28.
-
Balch CM, et al. Prognostic factors analysis of 17,600 melanoma patients: Validation of the American Joint Committee on Cancer melanoma staging system. J Clin Oncol. 2001;19(16):3622-3634. doi:10.1200/JCO.2001.19.16.3622
-
Gershenwald JE, et al. Melanoma staging: Evidence-based changes in the American Joint Committee on Cancer eighth edition cancer staging manual. CA Cancer J Clin. 2017;67(6):472-492. doi:10.3322/caac.21409
-
Sladden MJ, et al. Surgical excision margins for primary cutaneous melanoma. Cochrane Database Syst Rev. 2009;(4):CD004835. doi:10.1002/14651858.CD004835.pub2
-
Wong SL, et al. Sentinel lymph node biopsy for melanoma: American Society of Clinical Oncology and Society of Surgical Oncology joint clinical practice guideline. J Clin Oncol. 2012;30(23):2912-2918. doi:10.1200/JCO.2011.40.3519
-
Faries MB, et al. Completion dissection or observation for sentinel-node metastasis in melanoma. N Engl J Med. 2017;376(23):2211-2222. doi:10.1056/NEJMoa1613210
-
Eggermont AMM, et al. Pembrolizumab versus placebo after complete resection of high-risk stage III melanoma: Final results regarding distant metastasis-free survival from the EORTC 1325-MG/KEYNOTE-054 double-blind phase III trial. J Clin Oncol. 2021;39(suppl 15):9500. doi:10.1200/JCO.2021.39.15_suppl.9500
-
Weber J, et al. Adjuvant nivolumab versus ipilimumab in resected stage III or IV melanoma. N Engl J Med. 2017;377(19):1824-1835. doi:10.1056/NEJMoa1709030
-
Ascierto PA, et al. Adjuvant nivolumab versus ipilimumab in resected stage IIIB-C and stage IV melanoma (CheckMate 238): 4-year results from a multicentre, double-blind, randomised, controlled, phase 3 trial. Lancet Oncol. 2020;21(11):1465-1477. doi:10.1016/S1470-2045(20)30494-0
-
Long GV, et al. Adjuvant dabrafenib plus trametinib in stage III BRAF-mutated melanoma. N Engl J Med. 2017;377(19):1813-1823. doi:10.1056/NEJMoa1708539
-
Wolchok JD, et al. Overall survival with combined nivolumab and ipilimumab in advanced melanoma. N Engl J Med. 2017;377(14):1345-1356. doi:10.1056/NEJMoa1709684
-
Wei SC, et al. Fundamental mechanisms of immune checkpoint blockade therapy. Cancer Discov. 2018;8(9):1069-1086. doi:10.1158/2159-8290.CD-18-0367
-
Long GV, et al. Combined BRAF and MEK inhibition versus BRAF inhibition alone in melanoma. N Engl J Med. 2014;371(20):1877-1888. doi:10.1056/NEJMoa1406037
-
Larkin J, et al. Combined vemurafenib and cobimetinib in BRAF-mutated melanoma. N Engl J Med. 2014;371(20):1867-1876. doi:10.1056/NEJMoa1408868
-
Dummer R, et al. Encorafenib plus binimetinib versus vemurafenib or encorafenib in patients with BRAF-mutant melanoma (COLUMBUS): A multicentre, open-label, randomised phase 3 trial. Lancet Oncol. 2018;19(5):603-615. doi:10.1016/S1470-2045(18)30142-6
-
Robert C, et al. Five-year outcomes with dabrafenib plus trametinib in metastatic melanoma. N Engl J Med. 2019;381(7):626-636. doi:10.1056/NEJMoa1904059
-
Tawbi HA, et al. Combined nivolumab and ipilimumab in melanoma metastatic to the brain. N Engl J Med. 2018;379(8):722-730. doi:10.1056/NEJMoa1805453
-
Burmeister BH, et al. Adjuvant radiotherapy versus observation alone for patients at risk of lymph-node field relapse after therapeutic lymphadenectomy for melanoma: A randomised trial. Lancet Oncol. 2012;13(6):589-597. doi:10.1016/S1470-2045(12)70138-9
-
National Institute for Health and Care Excellence. Melanoma: assessment and management [NG14]. 2015 (updated 2022). Available at: https://www.nice.org.uk/guidance/ng14
-
Hua C, et al. Association of vitiligo with tumor response in patients with metastatic melanoma treated with pembrolizumab. JAMA Dermatol. 2016;152(1):45-51. doi:10.1001/jamadermatol.2015.2707
-
Chen AC, et al. A phase 3 randomized trial of nicotinamide for skin-cancer chemoprevention. N Engl J Med. 2015;373(17):1618-1626. doi:10.1056/NEJMoa1506197
-
US Preventive Services Task Force. Screening for skin cancer: US Preventive Services Task Force recommendation statement. JAMA. 2016;316(4):429-435. doi:10.1001/jama.2016.8465
-
Salerni G, et al. Meta-analysis of digital dermoscopy follow-up of melanocytic skin lesions: A study on behalf of the International Dermoscopy Society. J Eur Acad Dermatol Venereol. 2013;27(7):805-814. doi:10.1111/jdv.12032
-
Michielin O, et al. Cutaneous melanoma: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2019;30(12):1884-1901. doi:10.1093/annonc/mdz411
-
National Comprehensive Cancer Network. NCCN Clinical Practice Guidelines in Oncology: Melanoma: Cutaneous. Version 2.2023.
-
Swetter SM, et al. Guidelines of care for the management of primary cutaneous melanoma. J Am Acad Dermatol. 2019;80(1):208-250. doi:10.1016/j.jaad.2018.08.055
Revision Summary
High-Yield Facts for Quick Revision
Epidemiology:
- 5th most common UK cancer; 16,000 cases/year
- Incidence doubled over 30 years
- Lifetime risk ~1 in 40
Risk Factors:
- UV exposure (intermittent sunburns > chronic)
- Fair skin, red hair, > 50 naevi, dysplastic naevi
- CDKN2A mutation (familial)
- Immunosuppression
Clinical Recognition:
- ABCDE: Asymmetry, Border, Colour, Diameter, Evolution (most sensitive)
- Ugly Duckling sign
- Subtypes: SSM (70%), Nodular (15%, aggressive), Lentigo maligna (elderly, face), Acral (palms/soles/nails, Hutchinson's sign)
Pathophysiology:
- BRAF V600E (50%), NRAS (20%), KIT (acral/mucosal)
- Radial growth phase → Vertical growth phase
- Breslow thickness = depth from granular layer to deepest cell
Investigations:
- Complete excision biopsy with 2mm margin (NOT punch)
- Histology: Breslow, ulceration, mitotic rate, margins
- Staging CT/PET if stage IIB+
Staging (AJCC 8th):
- T: Breslow + ulceration
- N: Node number and burden
- M: Site + LDH
- Stage IA (thin, no ulceration): 99% 5YS
- Stage IV: 10-50% 5YS (depends on treatment)
Management:
- Surgery: WLE margins—in-situ 5mm, ≤1mm=1cm, 1-2mm=1-2cm, > 2mm=2cm
- SLNB: If Breslow > 1mm; positive → observation/adjuvant therapy (NOT automatic CLND post-MSLT-II)
- Adjuvant (Stage III): Pembrolizumab/nivolumab OR dabrafenib+trametinib (BRAF+)
- Metastatic: Nivo+ipi (52% 5YS) OR BRAF+MEK inhibitors; brain mets respond to both
Prognosis:
- Breslow thickness is THE key factor
- less than 1 mm: > 95% cure
-
4 mm: less than 50% 5YS without adjuvant treatment
- Ulceration, nodal disease, LDH worsen prognosis
Guidelines:
- NICE NG14, ESMO 2022, NCCN 2023
Learning map
Use these linked topics to study the concept in sequence and compare related presentations.
Prerequisites
Start here if you need the foundation before this topic.
- Skin Anatomy and Physiology
- Benign Naevi
Differentials
Competing diagnoses and look-alikes to compare.
- Basal Cell Carcinoma
- Squamous Cell Carcinoma
- Seborrhoeic Keratosis
- Pigmented Basal Cell Carcinoma
Consequences
Complications and downstream problems to keep in mind.
- Metastatic Disease Management
- Brain Metastases