Minimal Change Disease (Adult)
Minimal change disease (MCD) is a primary glomerular disorder characterised by nephrotic-range proteinuria with normal g... MRCP exam preparation.
What matters first
Minimal change disease (MCD) is a primary glomerular disorder characterised by nephrotic-range proteinuria with normal g... MRCP exam preparation.
Thromboembolism (DVT, PE, renal vein thrombosis) - risk 5-25% in nephrotic syndrome
9 Jan 2026
Generated educational material; verify before clinical use.
Visible references section
See the concept before reading it
Study the key anatomy, imaging, and decision pathways as full teaching plates.
Clinical board
A visual summary of the highest-yield teaching signals on this page.
Urgent signals
Safety-critical features pulled from the topic metadata.
- Thromboembolism (DVT, PE, renal vein thrombosis) - risk 5-25% in nephrotic syndrome
- Severe hypoalbuminaemia (less than 20 g/L) requiring prophylactic anticoagulation
- AKI in setting of nephrotic syndrome
- Infection risk (spontaneous bacterial peritonitis, cellulitis, pneumonia)
Exam focus
Current exam surfaces linked to this topic.
- MRCP
Content status and exam context
This page is AI-generated educational content. It may contain errors or omissions and is not a substitute for current guidelines, local protocols, senior clinical judgement, or professional medical advice.
MedVellum does not claim an individual clinician reviewer, board certification, or professional credential for this page unless a future version names a real, verifiable reviewer.
Clinical explanation and evidence
Minimal Change Disease (Adult)
1. Clinical Overview
Summary
Minimal change disease (MCD) is a primary glomerular disorder characterised by nephrotic-range proteinuria with normal glomerular architecture on light microscopy and diffuse podocyte foot process effacement on electron microscopy. While MCD accounts for 80-90% of idiopathic nephrotic syndrome in children, it represents only 10-15% of cases in adults, with focal segmental glomerulosclerosis (FSGS) and membranous nephropathy being more prevalent in this age group. [1,2]
The pathogenesis involves immune dysregulation, particularly T-cell dysfunction, leading to production of circulating permeability factors that directly injure podocytes and disrupt the glomerular filtration barrier. This results in massive, highly selective proteinuria (predominantly albumin) with preservation of larger plasma proteins. [3] Unlike proliferative glomerulonephritides, patients typically maintain normal renal function, lack haematuria, and remain normotensive at presentation.
MCD demonstrates exceptional steroid responsiveness—80-90% of adults achieve complete remission with corticosteroids, though treatment courses are longer than in children (16 weeks versus 8-12 weeks). [4] However, the disease is characterised by high relapse rates (50-70%), with 30-50% becoming steroid-dependent or frequently relapsing, necessitating steroid-sparing immunosuppression. [5] Despite frequent relapses, long-term renal prognosis remains excellent, with progression to end-stage kidney disease occurring in less than 5% of true MCD cases (steroid-resistant cases often represent FSGS on re-biopsy). [6]
Secondary MCD in adults may be triggered by NSAIDs, lithium, interferon, ampicillin, or associated with Hodgkin lymphoma, thymoma, and rarely other malignancies. [7] Recognition of secondary forms is crucial as treatment must address the underlying cause.
Key Facts
- Definition: Glomerular disease causing nephrotic syndrome with normal light microscopy and diffuse podocyte foot process effacement on electron microscopy
- Incidence: Adults 0.5-1 per 100,000 per year; accounts for 10-15% of adult nephrotic syndrome (versus 80-90% in children)
- Peak Demographics: Bimodal distribution in adults—young adults (20-30 years) and elderly (> 65 years)
- Pathognomonic Feature: Massive proteinuria (> 3.5 g/24h) with highly selective proteinuria, normal light microscopy, diffuse foot process effacement (> 80% podocytes) on electron microscopy
- Gold Standard Investigation: Renal biopsy mandatory in adults (unlike children where empirical treatment is standard)
- First-line Treatment: Prednisolone 1 mg/kg/day (maximum 80 mg) for minimum 4 weeks, extended to 16 weeks if needed
- Steroid Response: 80-90% achieve complete remission (adults) versus 90-95% (children); response slower in adults (4-16 weeks versus 2-4 weeks)
- Relapse Rate: 50-70% experience at least one relapse; 30-50% become steroid-dependent or frequently relapsing
- Prognosis: Excellent—progression to ESKD less than 5% (primarily occurs in steroid-resistant cases, often FSGS on re-evaluation)
Clinical Pearls
Age-Related Diagnostic Pearl: In adults presenting with nephrotic syndrome, always perform renal biopsy before initiating treatment. Unlike children, adults have higher rates of FSGS, membranous nephropathy, and secondary causes that require histological confirmation. Empirical steroid therapy without biopsy is inappropriate in adults.
Treatment Pearl: Adults require longer steroid courses than children for optimal response—maintain full-dose prednisolone for at least 4 weeks, and up to 16 weeks before declaring steroid resistance. Premature cessation or tapering is a common cause of apparent treatment failure. [4]
Thrombosis Prevention Pearl: Nephrotic syndrome is a hypercoagulable state. Prophylactic anticoagulation should be considered when serum albumin less than 20 g/L, especially in adults who have additional risk factors (immobility, obesity, prior VTE). Renal vein thrombosis occurs in 5-25% and may be clinically silent. [8]
Secondary Cause Pearl: Always inquire about NSAID use (including over-the-counter ibuprofen), recent interferon therapy, and screen for malignancy (especially Hodgkin lymphoma in younger adults, solid tumours in elderly). Drug-induced MCD typically resolves with cessation of offending agent. [7]
Relapse Recognition Pearl: Patients should monitor urine with dipstick at home during intercurrent illness or stress. Early detection of relapse (proteinuria 2-3+) allows early re-introduction of low-dose steroids, potentially preventing full nephrotic syndrome recurrence.
Pitfall Warning: Steroid-resistant MCD (failure to remit after 16 weeks of adequate therapy) often represents FSGS missed on initial biopsy due to sampling error. Re-biopsy should be strongly considered, as management and prognosis differ significantly. [9]
Mnemonic: MCD = Minimal on light microscopy, Complete response to steroids (90%), Diffuse foot process effacement on EM
Why This Matters Clinically
MCD represents the paradigm of steroid-responsive glomerular disease and demonstrates reversible podocyte injury. Understanding its pathophysiology informs therapeutic approaches to other podocytopathies. Complications of nephrotic syndrome—particularly thromboembolism and infection—require proactive prevention and can cause significant morbidity even when the underlying glomerular disease is highly treatable. The challenge in adult MCD is not achieving remission but managing frequent relapses and minimising cumulative steroid toxicity through judicious use of steroid-sparing agents. Distinguishing true MCD from early or focal FSGS has critical prognostic implications and requires expertise in renal pathology.
2. Epidemiology
Incidence and Prevalence
Children:
- Incidence: 2-7 per 100,000 children per year
- Accounts for 80-90% of idiopathic nephrotic syndrome in children aged 2-6 years [1]
Adults:
- Incidence: 0.5-1 per 100,000 adults per year
- Accounts for 10-15% of adult idiopathic nephrotic syndrome (versus 35-40% FSGS, 30-35% membranous nephropathy) [2]
- Prevalence difficult to estimate due to relapsing-remitting nature
Demographics
| Factor | Details |
|---|---|
| Age Distribution | Bimodal in adults: peak 20-30 years, second peak > 65 years |
| Paediatric Peak | 2-6 years (80% of childhood nephrotic syndrome) |
| Sex Ratio | Children: Male:Female 2:1; Adults: approximately 1:1 |
| Ethnicity | Higher incidence in South Asian populations; lower in Black populations compared to FSGS |
| Seasonal Variation | Some evidence of increased incidence following upper respiratory tract infections |
Geographic Variation
- Higher reported incidence in Asian countries (particularly Japan, India)
- May reflect genetic susceptibility and/or environmental factors
- Registry data from Europe and North America show consistent 10-15% proportion of adult nephrotic syndrome
Risk Factors and Associations
Primary (Idiopathic) MCD Associations
| Association | Relative Risk/Notes | Evidence |
|---|---|---|
| Atopy | Strong association with asthma, eczema, allergic rhinitis, food allergies | Suggests immune dysregulation [3] |
| Recent URTI | May trigger initial presentation or relapse | Viral triggers postulated |
| HLA Associations | HLA-DR7, HLA-B12 in some populations | Genetic susceptibility markers |
| Immunisations | Rarely reported following vaccinations | Temporal association, causality unclear |
| Allergic Reactions | Bee stings, food allergens occasionally implicated | Case reports |
Secondary MCD Causes
Drug-Induced MCD (10-15% of adult MCD): [7]
| Drug Class | Common Agents | Mechanism | Time to Onset |
|---|---|---|---|
| NSAIDs | Ibuprofen, naproxen, diclofenac | Most common drug cause; COX-2 inhibitors also implicated | Weeks to months |
| Antibiotics | Ampicillin, rifampicin, cephalosporins | Rare | Variable |
| Lithium | Mood stabiliser | Interferes with podocyte function | Months to years |
| Interferon | IFN-α, IFN-β | Immune-mediated | Weeks to months |
| Bisphosphonates | Pamidronate | Rare association | Variable |
| Immunotherapy | Checkpoint inhibitors (PD-1, PD-L1 inhibitors) | Immune-related adverse event | Weeks to months |
Malignancy-Associated MCD: [10]
| Malignancy | Association Strength | Screening Recommendations |
|---|---|---|
| Hodgkin Lymphoma | Strongest association (classic paraneoplastic syndrome) | Age-appropriate screening, particularly in young adults with B symptoms |
| Non-Hodgkin Lymphoma | Weaker association | Consider in appropriate clinical context |
| Thymoma | Case reports | Rare, consider if mediastinal mass |
| Solid Tumours | Very rare (lung, colon, prostate reported) | Routine age-appropriate cancer screening |
Other Associations:
- Mycoplasma pneumoniae infection
- Syphilis (rare)
- HIV (rare, more commonly associated with collapsing FSGS)
3. Pathophysiology
Molecular and Cellular Mechanisms
Step 1: Immune Dysregulation and T-Cell Dysfunction
T-Cell Abnormalities: MCD is fundamentally an immune-mediated disorder, though the precise initiating event remains incompletely understood. [3]
- CD4+ T-cell polarisation: Shift towards Th2 phenotype with increased IL-4, IL-13 production
- Regulatory T-cell (Treg) dysfunction: Reduced number and/or function of CD4+CD25+FOXP3+ Tregs
- T-cell activation: Abnormal response to antigenic stimulation, possibly viral or allergen-triggered
- B-cell involvement: Emerging evidence suggests B-cells may contribute (explaining rituximab efficacy)
Evidence Base:
- T-cell supernatants from MCD patients cause proteinuria when injected into rats [3]
- Remission with T-cell suppressive therapies (corticosteroids, calcineurin inhibitors, cyclophosphamide)
- Association with atopy and allergic disorders suggests dysregulated immune responses
Step 2: Circulating Permeability Factor(s) Production
The Permeability Factor Hypothesis:
Dysfunctional immune cells produce one or more circulating factors that directly injure podocytes. [11]
Proposed Permeability Factors:
- Angiopoietin-like 4 (ANGPTL4): Upregulated in podocytes in MCD; causes proteinuria in experimental models
- Hemopexin: Increased in relapsing nephrotic syndrome
- Cardiotrophin-like cytokine factor 1 (CLCF1): Implicated in post-transplant recurrence studies
- CD80 (B7-1): Controversial; initially proposed but not consistently validated
Evidence:
- Plasma exchange sometimes induces remission (removing circulating factors)
- Recurrence of MCD within hours in kidney transplant recipients (circulating factor affecting new kidney)
- Plasmapheresis can reduce proteinuria in some patients
Current Understanding: Likely multiple factors rather than single "permeability factor"; factors may vary between patients and disease stages.
Step 3: Podocyte Injury and Foot Process Effacement
Podocyte Structure and Function:
Podocytes are highly specialised glomerular epithelial cells with interdigitating foot processes connected by slit diaphragms. The slit diaphragm contains nephrin, podocin, CD2AP, and other proteins forming the final barrier to protein filtration. [12]
Mechanisms of Foot Process Effacement in MCD:
-
Actin Cytoskeleton Disruption:
- Permeability factors activate podocyte signalling pathways (Rho GTPases, phospholipase C)
- Reorganisation of actin cytoskeleton from organised bundles to diffuse network
- Retraction and flattening of foot processes
-
Slit Diaphragm Disruption:
- Redistribution or internalisation of nephrin and podocin
- Widening of filtration slits
- Loss of size-selective barrier function
-
Loss of Negative Charge:
- Reduction in glomerular polyanion (heparan sulphate proteoglycans)
- Loss of charge-selective barrier (albumin is negatively charged)
-
Podocyte Adaptation vs Injury:
- MCD: Reversible adaptive response (foot processes reform with treatment)
- FSGS: Irreversible injury with podocyte detachment and segmental sclerosis
Key Distinction MCD vs FSGS:
- MCD: Diffuse (> 80% podocytes), reversible effacement; podocytes remain viable
- FSGS: Focal injury with podocyte loss, extracellular matrix deposition, irreversible scarring
Step 4: Disruption of Glomerular Filtration Barrier
The Three-Layer Barrier:
- Fenestrated Endothelium: Intact in MCD
- Glomerular Basement Membrane (GBM): Structurally normal in MCD
- Podocyte Slit Diaphragm: Primary site of dysfunction in MCD
Consequence: Highly Selective Proteinuria
- Selective proteinuria: Predominantly albumin (69 kDa) and small proteins
- Larger proteins retained: IgG (150 kDa) relatively preserved
- Selectivity Index: Ratio of IgG clearance to transferrin clearance
- less than 0.1: Highly selective (typical of MCD)
-
0.2: Non-selective (suggests FSGS or other glomerulopathies)
Massive Proteinuria:
- Often 5-15 g/day (versus nephrotic threshold of 3.5 g/day)
- Can exceed 20 g/day in severe cases
- Proteinuria magnitude does not correlate with prognosis in MCD
Step 5: Systemic Consequences of Massive Proteinuria (Nephrotic Syndrome)
Hypoalbuminaemia:
- Urinary albumin loss exceeds hepatic synthetic capacity
- Serum albumin typically 15-25 g/L (normal 35-50 g/L)
- Reduced oncotic pressure drives oedema formation
Oedema Formation:
- Underfill mechanism: Reduced plasma oncotic pressure → fluid shift to interstitium → reduced effective circulating volume → RAAS activation → sodium retention
- Overfill mechanism: Primary renal sodium retention (epithelial sodium channel upregulation) → expanded plasma volume → oedema
- Reality: Both mechanisms contribute
Hyperlipidaemia: [13]
- Increased hepatic lipoprotein synthesis (compensatory response to hypoalbuminaemia)
- Decreased lipid catabolism (loss of lipid-regulating proteins in urine)
- Elevated total cholesterol (often 10-15 mmol/L), LDL, VLDL, triglycerides
- HDL often normal or reduced
- Lipiduria (oval fat bodies, Maltese crosses on polarised microscopy)
Hypercoagulability: [8]
- Prothrombotic changes:
- Loss of antithrombin III, protein C, protein S in urine
- Increased hepatic synthesis of fibrinogen, factors V, VII, VIII
- Platelet hyperaggregability
- Hyperviscosity from hypoalbuminaemia and hyperlipidaemia
- Reduced fibrinolysis
- Thromboembolism risk: 5-25% in nephrotic syndrome
- Deep vein thrombosis
- Pulmonary embolism
- Renal vein thrombosis (may be asymptomatic)
- Arterial thrombosis (rare)
Immunodeficiency:
- Loss of immunoglobulins (especially IgG)
- Loss of complement factors (especially factor B, properdin)
- Impaired opsonisation and cell-mediated immunity
- Increased infection risk: encapsulated organisms (Streptococcus pneumoniae, Haemophilus influenzae), spontaneous bacterial peritonitis, cellulitis
Other Metabolic Consequences:
- Vitamin D deficiency (loss of vitamin D binding protein)
- Hypothyroidism (loss of thyroxine-binding globulin; usually subclinical)
- Trace element deficiencies (zinc, copper)
- Protein malnutrition (if prolonged nephrotic syndrome)
Step 6: Remission with Corticosteroid Therapy
Mechanisms of Steroid Action in MCD:
-
Immunosuppression:
- Suppression of T-cell activation and proliferation
- Reduction in circulating permeability factor production
- Restoration of Treg function
-
Direct Podocyte Effects:
- Podocytes express glucocorticoid receptors
- Stabilisation of actin cytoskeleton
- Upregulation of nephrin and podocin expression
- Anti-apoptotic effects on podocytes
-
Anti-inflammatory Effects:
- Reduction in cytokine production (IL-4, IL-13)
- Decreased vascular permeability
Timeline of Response:
- Children: Proteinuria resolves 2-4 weeks (90-95%)
- Adults: Proteinuria resolves 4-16 weeks (80-90%)
- Delayed response in adults may reflect chronic podocyte changes requiring longer recovery
Foot Process Recovery:
- Electron microscopy shows gradual reformation of foot processes over weeks
- Complete structural normalisation may lag behind proteinuria resolution
- Reversibility confirms MCD diagnosis (versus irreversible FSGS)
Histopathology
Light Microscopy (LM)
Classic Finding: Normal Appearance
- Glomeruli appear entirely normal in size, cellularity, and architecture
- No mesangial proliferation
- No endocapillary proliferation
- No crescent formation
- No sclerosis or segmental lesions
- Tubules and interstitium: Normal or minimal acute tubular injury
Caveat:
- Up to 20% may show mild mesangial prominence (not true proliferation)
- Mild tubular lipid vacuolation may be present (reflecting lipiduria)
- Mild interstitial oedema
- Acute tubular injury if presenting with AKI
Differential on LM:
- If sclerotic lesions present → FSGS (may be sampling error if focal)
- If mesangial proliferation → IgA nephropathy, post-infectious GN
- If GBM thickening → membranous nephropathy
Immunofluorescence (IF)
Classic Finding: Negative or Non-specific
- Typically completely negative for IgG, IgA, IgM, C3, C1q, kappa, lambda
- May show trace mesangial IgM deposition (10-20% of cases)
- Trace C3 may be present
Significance:
- Absence of immune deposits distinguishes MCD from immune complex-mediated glomerulonephritis
- Prominent IF positivity suggests alternative diagnosis (membranous, lupus nephritis, IgA nephropathy)
Electron Microscopy (EM)
Pathognomonic Finding: Diffuse Podocyte Foot Process Effacement [12]
- Extent: > 80% of podocytes show effacement (versus focal in FSGS)
- Pattern: Foot processes appear flattened and fused, replacing normal interdigitating pattern
- Slit Diaphragm: Widened or obliterated filtration slits
GBM Appearance:
- Normal thickness (300-400 nm in adults)
- No subepithelial, subendothelial, or intramembranous deposits
- No lamellation or splitting
Other EM Features:
- Podocyte cytoplasm may show microvillous transformation (apical surface irregularities)
- Increased podocyte vacuolation (lipid)
- Endothelial cells: Normal
- Mesangium: Normal or minimal expansion
Diagnostic Criteria:
- Light microscopy: Normal glomeruli
- Immunofluorescence: Negative or trace non-specific staining
- Electron microscopy: Diffuse foot process effacement without immune deposits
4. Clinical Presentation
Symptom Constellation
Presenting Symptoms (Adults)
| Symptom | Frequency | Characteristics | Timeline |
|---|---|---|---|
| Peripheral Oedema | 95-100% | Initially dependent (ankles, feet), progresses to legs, sacrum if bed-bound | Days to weeks |
| Periorbital Oedema | 70-80% | More prominent in morning, improves through day | Days to weeks |
| Frothy/Foamy Urine | 60-80% | Bubbles persist; reflects massive proteinuria | Concurrent with oedema |
| Weight Gain | 80-90% | Rapid (2-10 kg), fluid retention | Days to weeks |
| Reduced Urine Output | 40-60% | Oliguria may occur | Variable |
| Abdominal Distension | 30-50% | Ascites in severe cases | Weeks |
| Fatigue/Malaise | 70-80% | Non-specific, may reflect hypoalbuminaemia | Variable |
| Dyspnoea | 20-40% | Pulmonary oedema rare; pleural effusions if severe | Severe cases |
| Diarrhoea | 10-20% | Intestinal oedema | Severe cases |
Symptoms Typically ABSENT in MCD (Distinguish from Other GN)
- ❌ Macroscopic haematuria (if present, consider IgA nephropathy, post-infectious GN, vasculitis)
- ❌ Hypertension at presentation (suggests membranous, FSGS, proliferative GN)
- ❌ Oliguria/AKI (uncommon unless severe nephrotic syndrome; suggests alternative diagnosis)
- ❌ Constitutional symptoms (fever, weight loss, night sweats—if present, consider malignancy, vasculitis)
- ❌ Arthralgia/rash (suggests lupus or systemic disease)
Physical Examination Findings
General Appearance
- Appears cushingoid if previously treated with steroids
- Appears volume-overloaded but may have reduced effective circulating volume
Vital Signs
- Blood Pressure: Typically normal (130/80 mmHg or less)
- Hypertension suggests FSGS, membranous nephropathy, or other diagnosis
- Heart Rate: May be elevated if significant intravascular depletion
- Respiratory Rate: Increased if pulmonary oedema/pleural effusions
Focused Examination
Fluid Status Assessment:
| Site | Findings | Severity Indicator |
|---|---|---|
| Periorbital | Puffiness, especially morning | Mild-moderate |
| Hands | Difficulty removing rings, puffy fingers | Mild |
| Ankles/Feet | Pitting oedema (grade depth 0-4+) | All severities |
| Sacral | In bed-bound patients | Moderate-severe |
| Genital | Scrotal or labial oedema | Moderate-severe |
| Abdominal | Shifting dullness, fluid wave (ascites) | Severe |
| Pulmonary | Reduced breath sounds at bases (effusions) | Severe |
Pitting Oedema Grading:
- 1+: Slight indentation, disappears rapidly
- 2+: Indentation 2-4 mm, disappears in 10-15 seconds
- 3+: Indentation 4-6 mm, lasts > 1 minute
- 4+: Deep indentation > 6 mm, lasts 2-5 minutes
Cardiovascular Examination:
- Jugular venous pressure: Typically not elevated (distinguishes from heart failure)
- Apex beat: Non-displaced
- Heart sounds: Normal; no murmurs
- Peripheral pulses: Present; check for asymmetry (DVT consideration)
Respiratory Examination:
- Reduced breath sounds at bases (pleural effusions)
- Dullness to percussion at bases
- No crackles typically (pulmonary oedema rare unless severe)
Abdominal Examination:
- Shifting dullness (ascites)
- Non-tender unless spontaneous bacterial peritonitis
- No hepatosplenomegaly (if present, consider alternative diagnoses)
- Umbilical herniation may occur with severe ascites
Skin Examination:
- Pallor (if significant hypoalbuminaemia)
- Striae (if previous steroid exposure)
- No rash (presence suggests systemic disease)
- Examine legs for signs of DVT (asymmetric swelling, warmth, erythema, tenderness)
Other Systems:
- Fundoscopy: No hypertensive or diabetic changes (unlike diabetic nephropathy)
- Neurological: Normal
Clinical Patterns and Phenotypes
Age-Related Presentation Differences
Young Adults (20-40 years):
- Often sudden onset
- Severe nephrotic syndrome (proteinuria > 10 g/day)
- Excellent steroid response
- Higher relapse rates
- Consider secondary causes: NSAIDs, Hodgkin lymphoma
Elderly (> 65 years):
- May have more insidious onset
- Higher frequency of secondary causes (NSAIDs, malignancy)
- Slower to respond to steroids
- Greater steroid toxicity concerns
- Higher thromboembolism risk
Red Flags and Clinical Warning Signs
[!CAUTION] IMMEDIATE ASSESSMENT REQUIRED
Thromboembolism:
- Unilateral leg swelling, pain, erythema (DVT)
- Acute dyspnoea, pleuritic chest pain, tachycardia (PE)
- Flank pain, haematuria, asymmetric renal function (renal vein thrombosis)
Infection:
- Fever with abdominal pain and ascites (spontaneous bacterial peritonitis)
- Cellulitis in oedematous limbs
- Pneumonia (encapsulated organisms)
Severe Hypoalbuminaemia:
- Albumin less than 15 g/L: High risk complications
- Anasarca (generalised oedema)
Acute Kidney Injury:
- Oliguria, rising creatinine
- Consider: Volume depletion, sepsis, AIN (if recent antibiotics), acute tubular necrosis
Atypical Features Suggesting Alternative Diagnosis:
- Macroscopic haematuria → IgA nephropathy, vasculitis, post-infectious GN
- Hypertension → FSGS, membranous nephropathy
- Systemic symptoms → Malignancy, lupus, vasculitis
- Renal impairment → FSGS, membranous with thrombosis
- Prolonged steroid resistance → FSGS (re-biopsy indicated)
5. Diagnostic Approach
Clinical Diagnosis
Nephrotic Syndrome Criteria:
- Heavy proteinuria: > 3.5 g/24h or urine protein:creatinine ratio (uPCR) > 300 mg/mmol
- Hypoalbuminaemia: Serum albumin less than 25 g/L
- Oedema
- Hyperlipidaemia (often present but not mandatory for diagnosis)
MCD-Specific Clinical Clues:
- Normotensive
- No haematuria
- Normal renal function (eGFR typically > 60 ml/min/1.73m²)
- Highly selective proteinuria (if measured)
Investigations
Initial Laboratory Workup
Urine Studies:
| Test | Finding in MCD | Clinical Significance |
|---|---|---|
| Urinalysis (Dipstick) | Proteinuria 3-4+; no haematuria | Screen for nephrotic-range proteinuria |
| Urine Microscopy | Hyaline casts, oval fat bodies, fatty casts; no RBC casts | Distinguish from glomerulonephritis |
| Spot uPCR | > 300 mg/mmol (> 350 mg/mmol confirms nephrotic) | Quantify proteinuria; easier than 24h |
| 24-Hour Urine Protein | > 3.5 g/day (often 5-15 g/day in MCD) | Gold standard quantification |
| Urine Albumin | Predominantly albumin | Highly selective proteinuria |
| Selectivity Index | less than 0.1 (highly selective) | IgG clearance / transferrin clearance; supports MCD |
Serum Studies:
| Test | Expected Finding | Purpose |
|---|---|---|
| Serum Albumin | less than 25 g/L (often 15-20 g/L) | Confirms hypoalbuminaemia; severity marker |
| Total Protein | Reduced (less than 60 g/L) | Reflects protein loss |
| Urea and Electrolytes | Usually normal eGFR (> 60 ml/min/1.73m²) | Assess renal function; AKI rare |
| Lipid Profile | Total cholesterol > 10 mmol/L, elevated LDL, VLDL, TG | Nephrotic syndrome component |
| Complement (C3, C4) | Normal | Exclude complement-mediated GN (MPGN, post-infectious, lupus) |
Extended Workup (Adults)
Exclude Secondary Causes:
| Investigation | Purpose | When Indicated |
|---|---|---|
| Serum/Urine Protein Electrophoresis | Exclude myeloma | All adults > 40 years; younger if suggestive features |
| Serum Free Light Chains | Exclude myeloma | As above |
| Hepatitis B/C Serology | Exclude viral-associated GN | All adults with nephrotic syndrome |
| HIV Test | Exclude HIV-associated nephropathy (HIVAN) | Risk factors or unexplained nephrotic syndrome |
| ANA, anti-dsDNA | Exclude SLE | Younger patients, systemic features |
| ANCA | Exclude vasculitis | Haematuria, systemic symptoms |
| Cryoglobulins | Exclude cryoglobulinaemic GN | Hepatitis C positive, systemic symptoms |
Malignancy Screening (Paraneoplastic MCD): [10]
| Age Group | Screening |
|---|---|
| Young Adults (less than 40) | - Detailed history for B symptoms - Chest X-ray (Hodgkin lymphoma, thymoma) - CT chest if lymphadenopathy/mediastinal mass suspected |
| Elderly (> 60) | - Age-appropriate cancer screening - CT chest/abdomen/pelvis if constitutional symptoms - Colonoscopy, PSA (men), mammography (women) as per guidelines |
Renal Biopsy
Indications in Adults:
✅ MANDATORY in all adults with nephrotic syndrome before treatment
Rationale:
- 10-15% of adult nephrotic syndrome is MCD (versus 80-90% in children)
- FSGS (35-40%) and membranous nephropathy (30-35%) more common in adults
- Management and prognosis differ significantly
- Cannot reliably distinguish clinically
Biopsy Technique:
- Percutaneous under ultrasound guidance
- Native kidney biopsy (lower pole right kidney typically)
- Adequate sample: Minimum 10 glomeruli for light microscopy
- Tissue for light microscopy (LM), immunofluorescence (IF), and electron microscopy (EM)
Histological Diagnosis of MCD:
| Modality | Finding |
|---|---|
| Light Microscopy | Normal glomeruli (no proliferation, sclerosis, or crescents) |
| Immunofluorescence | Negative or trace non-specific IgM/C3 |
| Electron Microscopy | Diffuse podocyte foot process effacement (> 80% of capillary loops) |
Contraindications to Biopsy:
- Uncontrolled hypertension (SBP > 160 mmHg despite treatment)
- Bleeding diathesis (uncorrected)
- Single functioning kidney (relative)
- Uncooperative patient
When NOT to Biopsy (Paediatrics):
In children aged 1-10 years with typical nephrotic syndrome (no haematuria, no hypertension, normal complement, normal renal function), empirical steroid therapy without biopsy is standard practice. Biopsy if:
- Age less than 1 year or > 12 years
- Atypical features (haematuria, hypertension, low complement, renal impairment)
- Steroid resistance (no remission after 4-8 weeks)
- Steroid-dependent or frequently relapsing (before second-line therapy)
Imaging
Renal Ultrasound:
- Findings in MCD: Normal-sized or enlarged kidneys (due to oedema); increased echogenicity; preserved corticomedullary differentiation
- Purpose: Assess kidney size, exclude structural abnormalities, pre-biopsy assessment
Doppler Ultrasound:
- Indication: Suspected renal vein thrombosis (flank pain, haematuria, asymmetric renal function)
- Findings if RVT: Enlarged kidney, reduced/reversed diastolic flow, thrombus in renal vein
CT/MRI:
- Not routinely required for MCD diagnosis
- CT venography: If renal vein thrombosis suspected and Doppler inconclusive
- CT chest/abdomen/pelvis: Malignancy screening if paraneoplastic MCD suspected
6. Management
Treatment Algorithm
Image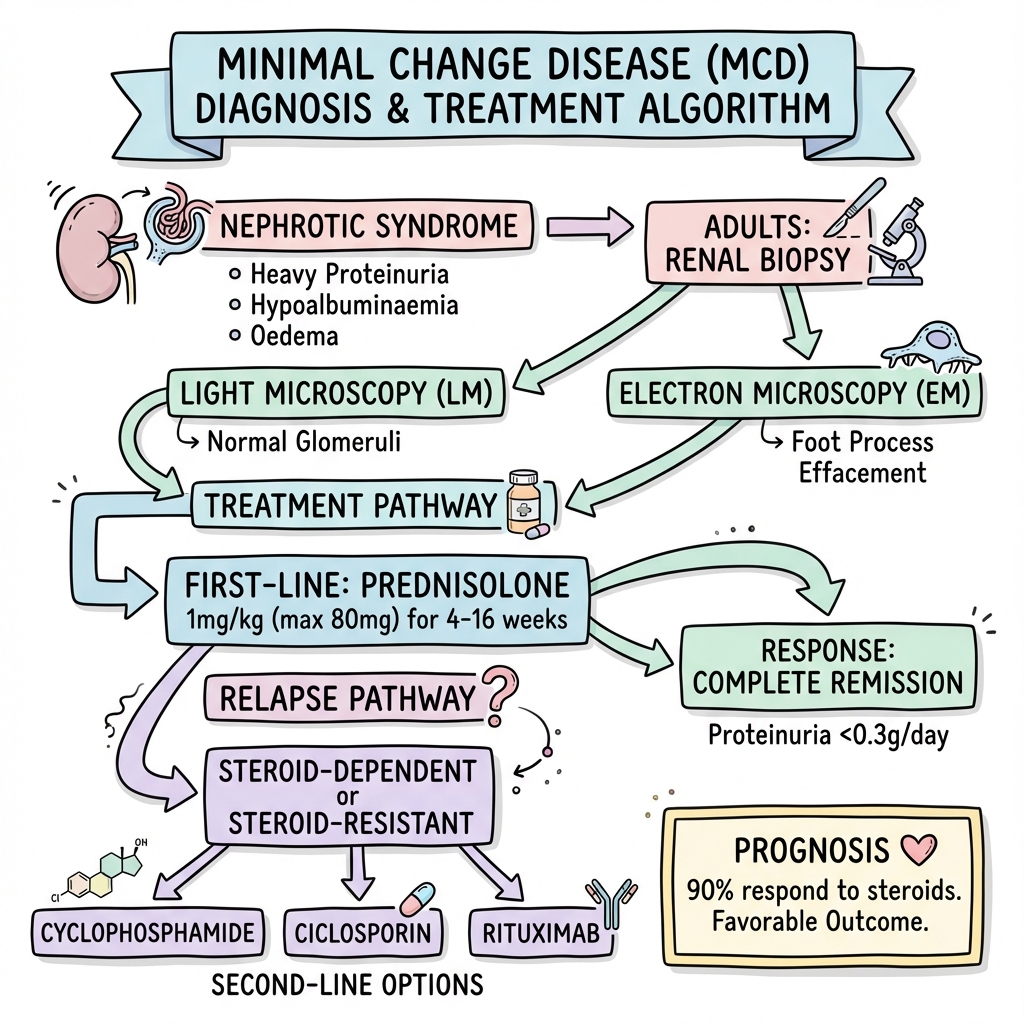 Minimal Change Disease Adult Management Algorithm
Minimal Change Disease Adult Management Algorithm
Adult Nephrotic Syndrome
↓
Renal Biopsy
↓
MCD Diagnosed (LM normal, EM diffuse foot process effacement)
↓
┌────────────────────────────────────────┐
│ Exclude Secondary Causes: │
│ - Stop NSAIDs, lithium, interferon │
│ - Screen for malignancy (Hodgkin, etc) │
│ - Treat underlying cause if identified │
└────────────────────────────────────────┘
↓
┌────────────────────────────────────────┐
│ Supportive Care: │
│ - Sodium restriction, diuretics │
│ - Thromboprophylaxis if albumin less than 20g/L │
│ - Infection prevention │
│ - Lipid management │
└────────────────────────────────────────┘
↓
FIRST-LINE TREATMENT
Prednisolone 1 mg/kg/day (max 80 mg)
Continue minimum 4 weeks
↓
Response at 4 weeks?
↓
┌────────┴────────┐
YES NO
│ │
Complete Continue prednisolone
Remission up to 16 weeks total
uPCR less than 30 │
│ ↓
Taper over Response by 16 weeks?
12 weeks │
│ ┌────┴────┐
│ YES NO
│ │ │
│ Taper STEROID-RESISTANT
│ Re-biopsy to exclude FSGS
│ Second-line: CNI + low-dose steroid
│ Consider: Rituximab
↓
Monitor for Relapse
(Home urine dipstick during illness)
│
Relapse occurs (50-70%)
│
Treat relapse with prednisolone
│
┌───┴────────────────────────┐
│ │
Infrequent Relapse Frequent Relapse OR
(less than 2 in 6 months) Steroid-Dependent
Treat each relapse SECOND-LINE THERAPY
│
┌───┴───────────────┐
│ │
Cyclophosphamide Calcineurin Inhibitor
2 mg/kg × 8-12 wks (Ciclosporin or Tacrolimus)
(Use ONCE only) OR Rituximab
OR Mycophenolate
First-Line Treatment: Corticosteroids
Prednisolone Regimen for Adults [4]
Induction Phase:
- Dose: Prednisolone 1 mg/kg/day (maximum 80 mg daily)
- Duration: Minimum 4 weeks; continue up to 16 weeks if no response
- Timing: Single morning dose (reduces insomnia, mimics physiological cortisol)
Rationale for Prolonged Treatment in Adults:
- Adults respond more slowly than children (median 8 weeks versus 2-4 weeks)
- 80-90% achieve remission by 16 weeks
- Premature cessation risks missing late responders
Tapering Phase (Once Remission Achieved):
- Continue full dose for 2 weeks after remission
- Taper by 10 mg every 1-2 weeks
- Total treatment duration: Typically 16-24 weeks (longer than paediatric 8-12 weeks)
Alternative Regimens:
- High-dose pulse methylprednisolone (controversial; not standard)
Response Definitions
| Category | Definition | Timeline |
|---|---|---|
| Complete Remission | uPCR less than 30 mg/mmol or 24h protein less than 0.3 g/day | 4-16 weeks |
| Partial Remission | > 50% reduction in proteinuria but still > 30 mg/mmol | Variable |
| Steroid Resistance | Failure to achieve remission after 16 weeks adequate therapy | 16 weeks |
| Relapse | uPCR > 300 mg/mmol (or 24h protein > 3.5 g) after remission | Variable post-remission |
| Frequent Relapser | ≥2 relapses in 6 months OR ≥4 relapses in 12 months | 6-12 months |
| Steroid-Dependent | Relapse during steroid taper OR within 2 weeks of stopping | During/post-taper |
Expected Response Rates [1,2,4]
| Population | Complete Remission Rate | Median Time to Remission |
|---|---|---|
| Children | 90-95% | 2-4 weeks |
| Adults | 80-90% | 4-16 weeks (median 8 weeks) |
Predictors of Steroid Response:
- ✅ Younger age at onset
- ✅ Normal renal function at presentation
- ✅ Highly selective proteinuria
- ❌ Older age (> 60 years): Slower response
- ❌ Male sex (weaker predictor)
Steroid Side Effects (Particularly Relevant in Adults)
Short-term (During High-Dose Phase):
- Hyperglycaemia/steroid-induced diabetes
- Hypertension
- Insomnia, mood changes, psychosis
- Gastritis/peptic ulcer
- Increased appetite, weight gain
- Acne, moon facies, striae
- Proximal myopathy
Long-term/Cumulative:
- Osteoporosis and fracture risk [14]
- Cataracts
- Avascular necrosis (hips, knees)
- Growth retardation (children)
- Cushingoid features
Infection Risk:
- Opportunistic infections (Pneumocystis, tuberculosis if prolonged/high-dose)
- Reactivation of latent TB, hepatitis B
Steroid Risk Mitigation
Bone Protection: [14]
- Calcium 1000-1200 mg daily
- Vitamin D 800-1000 IU daily
- Bisphosphonates if prednisolone ≥7.5 mg for > 3 months (alendronate 70 mg weekly)
- Baseline DEXA scan if prolonged treatment anticipated
GI Protection:
- Proton pump inhibitor if risk factors (age > 60, prior ulcer, concurrent NSAIDs)
Glucose Monitoring:
- Baseline HbA1c
- Monitor glucose if risk factors for diabetes
Infection Prevention:
- Avoid live vaccines during immunosuppression
- Pneumocystis prophylaxis (co-trimoxazole) if prednisolone > 20 mg for > 1 month (controversial; not universal practice)
Growth Monitoring (Children):
- Height and weight at each visit
- Alternate-day steroids reduce growth impact
Treatment of Relapse
Relapse Incidence:
- 50-70% of MCD patients experience ≥1 relapse [5]
- Higher in children than adults
- May be triggered by infections, stress, allergen exposure
Relapse Treatment:
- First Relapse: Treat as initial episode (prednisolone 1 mg/kg, taper over 12-16 weeks)
- Subsequent Infrequent Relapses: Same approach
- Frequent Relapses: Consider second-line steroid-sparing therapy
Patient Self-Management:
- Home urine dipstick monitoring during intercurrent illness
- Early detection allows prompt treatment
- Some protocols: Patient-initiated low-dose prednisolone (0.5 mg/kg) at first sign of proteinuria
Second-Line (Steroid-Sparing) Therapies
Indications for Second-Line Therapy
- Steroid-Dependent Disease: Relapses during taper or within 2 weeks of stopping
- Frequently Relapsing Disease: ≥2 relapses in 6 months or ≥4 in 12 months
- Steroid-Resistant Disease: No remission after 16 weeks adequate steroid therapy
- Steroid Toxicity: Intolerable side effects necessitating dose reduction
Cyclophosphamide
Indication: Steroid-dependent or frequently relapsing MCD [15]
Regimen:
- Dose: 2 mg/kg/day orally for 8-12 weeks (maximum cumulative 168 mg/kg)
- Alternative: IV pulse therapy (500-750 mg/m² monthly × 6 months)
Efficacy:
- 60-70% achieve sustained remission
- Relapse-free period: Often 1-2 years post-treatment
- CRITICAL: Use ONCE only (cumulative toxicity)
Adverse Effects:
- Bone Marrow Suppression: Leukopenia (monitor FBC weekly); increased infection risk
- Gonadal Toxicity: Oligospermia/azoospermia (men); premature ovarian failure (women, especially > 25 years)
- Haemorrhagic Cystitis: Encourage high fluid intake
- Malignancy Risk: Bladder cancer, lymphoma (long-term risk)
- Teratogenicity: Contraception mandatory
Monitoring:
- FBC weekly during treatment
- Discontinue if WCC less than 3.0 × 10⁹/L or neutrophils less than 1.5 × 10⁹/L
Counselling:
- Discuss fertility preservation (sperm banking, oocyte cryopreservation) before treatment
Calcineurin Inhibitors (Ciclosporin, Tacrolimus)
Indication: Steroid-dependent, frequently relapsing, or steroid-resistant MCD [16]
Ciclosporin:
- Dose: 3-5 mg/kg/day in divided doses (target trough level 100-150 ng/ml)
- Duration: Typically 6-12 months, then taper
Tacrolimus:
- Dose: 0.05-0.1 mg/kg/day in divided doses (target trough 5-10 ng/ml)
- Duration: As above
Efficacy:
- 80-85% achieve remission (including steroid-resistant cases)
- Relapse common on withdrawal (60-70%)
- Often requires prolonged therapy (1-2 years)
Adverse Effects:
- Nephrotoxicity: Acute (reversible vasoconstriction) and chronic (interstitial fibrosis)—monitor creatinine closely
- Hypertension: Common (40-60%); requires treatment
- Tremor: Fine tremor, especially hands
- Gingival Hyperplasia: Ciclosporin more than tacrolimus
- Hirsutism: Ciclosporin more than tacrolimus
- Hyperkalemia, Hypomagnesaemia
- Increased Malignancy Risk: Skin cancer (sun protection essential)
Monitoring:
- Trough levels (ciclosporin: 100-150 ng/ml; tacrolimus: 5-10 ng/ml)
- Creatinine, eGFR (monthly initially, then 3-monthly)
- Blood pressure (each visit)
- Magnesium, potassium
Rituximab (Anti-CD20 Monoclonal Antibody)
Indication: Steroid-dependent or frequently relapsing MCD; increasingly used as alternative to cyclophosphamide [17]
Mechanism:
- B-cell depletion (CD20+ B-cells)
- May reduce circulating permeability factors
- Exact mechanism in MCD unclear (MCD traditionally considered T-cell disease)
Regimen:
- Dose: 375 mg/m² IV weekly × 4 doses (standard lymphoma protocol)
- Alternative: Two doses 375 mg/m² separated by 2 weeks
Efficacy:
- 60-80% achieve sustained remission
- Relapse-free period: Median 12-18 months
- May repeat dosing if relapse after initial response
Adverse Effects:
- Infusion Reactions: Fever, chills, hypotension (usually first infusion; premedicate with antihistamine, paracetamol)
- Immunosuppression: Hypogammaglobulinaemia; increased infection risk
- Rare: Progressive multifocal leukoencephalopathy (PML), hepatitis B reactivation
Advantages over Cyclophosphamide:
- No gonadal toxicity (preferred in young adults desiring fertility)
- Can be repeated
- Generally well-tolerated
Monitoring:
- CD19/CD20 counts (confirm B-cell depletion)
- Immunoglobulin levels (monitor for hypogammaglobulinaemia)
- Hepatitis B serology pre-treatment (risk of reactivation)
Mycophenolate Mofetil (MMF)
Indication: Steroid-dependent or frequently relapsing MCD (less robust evidence than above agents)
Regimen:
- Dose: 1-2 g daily in divided doses
- Duration: 6-12 months minimum
Efficacy:
- 40-60% sustained remission (lower than other agents)
- Often used in combination with low-dose steroids or CNI
Adverse Effects:
- GI disturbance (diarrhoea, nausea)
- Bone marrow suppression (monitor FBC)
- Teratogenicity (contraception mandatory)
Role:
- Less effective than cyclophosphamide, CNI, or rituximab
- May be useful in patients intolerant of other agents
Approach to Steroid-Resistant MCD [9]
Definition: Failure to achieve remission after 16 weeks of adequate prednisolone therapy
Key Consideration: Is it truly MCD?
- Steroid resistance uncommon in true MCD (less than 5%)
- Many "steroid-resistant MCD" cases are FSGS on re-biopsy (focal lesions missed on initial biopsy due to sampling error)
Management:
-
Re-biopsy: Strongly consider repeat biopsy to confirm diagnosis
- Sample more glomeruli if possible
- FSGS may be focal; requires multiple glomeruli for detection
-
If Confirmed MCD:
- First-line: Calcineurin inhibitor (ciclosporin or tacrolimus) + low-dose prednisolone
- Alternative: Rituximab
- Rarely: Combination therapy (e.g., CNI + MMF)
-
If FSGS on Re-biopsy:
- Management changes to FSGS protocols
- Prognosis worse (30-40% progress to ESKD)
Supportive Care and Symptomatic Management
Oedema Management
Non-Pharmacological:
- Sodium Restriction: less than 2 g sodium daily (less than 5 g salt)
- Fluid Restriction: If severe hyponatraemia (typically not required)
- Elevate Legs: Reduce dependent oedema
- Compression Stockings: May reduce peripheral oedema (caution: may increase venous stasis if severe)
Diuretics:
| Agent | Dose | Mechanism | Notes |
|---|---|---|---|
| Furosemide | 40-240 mg daily (divide dose) | Loop diuretic | First-line; may require high doses due to hypoalbuminaemia |
| Bumetanide | 1-5 mg daily | Loop diuretic | More potent than furosemide |
| Metolazone | 2.5-10 mg daily | Thiazide-like | Add if loop resistance; synergistic effect |
| Spironolactone | 25-100 mg daily | Aldosterone antagonist | May help if secondary hyperaldosteronism |
Diuretic Resistance:
- Common in nephrotic syndrome (hypoalbuminaemia reduces diuretic delivery to tubules)
- Strategies:
- Increase dose
- Add second agent (sequential nephron blockade: loop + thiazide)
- IV Albumin + IV Furosemide: Temporary measure in severe cases
- Risk: Intravascular volume expansion may worsen hypertension, pulmonary oedema
- Not recommended for routine use
Monitoring:
- Daily weights
- Fluid balance charts
- Electrolytes (risk of hypokalaemia, hyponatraemia, hypomagnesaemia)
Thromboprophylaxis [8]
Rationale:
- Nephrotic syndrome is prothrombotic (loss of antithrombin III, protein C/S; increased procoagulant factors)
- Thromboembolism incidence: 5-25% in nephrotic syndrome
- Renal vein thrombosis may be asymptomatic
Risk Stratification:
| Risk Category | Criteria | Recommendation |
|---|---|---|
| High Risk | Albumin less than 20 g/L + immobility/prior VTE/malignancy | Prophylactic anticoagulation (LMWH or warfarin) |
| Moderate Risk | Albumin 20-25 g/L | Consider prophylaxis; mobilise actively |
| Lower Risk | Albumin > 25 g/L | Mobilisation; thromboprophylaxis if hospitalised/immobile |
Agents:
- LMWH (enoxaparin 40 mg SC daily): Easier dosing than warfarin; no monitoring required for prophylaxis
- Warfarin (target INR 2-3): If prolonged anticoagulation anticipated
- DOACs: Emerging data; less experience in nephrotic syndrome (protein binding affects drug levels)
Duration:
- Continue while albumin less than 20-25 g/L
- Discontinue once remission achieved and albumin normalises
Treatment of Established VTE:
- Therapeutic anticoagulation (LMWH followed by warfarin, or DOAC)
- Duration: Minimum 3-6 months; consider extended if recurrent or persistent risk factors
Infection Prevention and Management
Increased Infection Risk:
- Loss of immunoglobulins (especially IgG)
- Loss of complement factors
- Immunosuppressive therapy
Common Infections:
- Bacterial: Spontaneous bacterial peritonitis (SBP), cellulitis, pneumonia (encapsulated organisms: S. pneumoniae, H. influenzae)
- Viral: Varicella-zoster reactivation (if on steroids)
- Opportunistic: Pneumocystis jirovecii (if prolonged high-dose steroids)
Prevention:
- Pneumococcal Vaccination: PCV13 followed by PPV23 (8 weeks later)
- Influenza Vaccine: Annual (inactivated vaccine)
- Avoid Live Vaccines: During immunosuppression (MMR, varicella, yellow fever)
- Prophylactic Antibiotics: Generally not recommended routinely
- "Exception: Consider co-trimoxazole if prednisolone > 20 mg for > 1 month (PCP prophylaxis)"
Management:
- Low Threshold for Antibiotics: Fever or signs of infection should prompt early empirical treatment
- SBP: High suspicion if fever + abdominal pain + ascites; diagnostic paracentesis (neutrophils > 250/mm³); treat with ceftriaxone or cefotaxime
Hyperlipidaemia Management [13]
Rationale:
- Universal in nephrotic syndrome (cholesterol often 10-15 mmol/L)
- Increased cardiovascular risk with prolonged hyperlipidaemia
- Usually resolves with remission of nephrotic syndrome
Management:
- During Active Nephrotic Syndrome: Dietary modification; statins controversial (proteinuria may improve lipids more than statins)
- Persistent Hyperlipidaemia After Remission: Statin therapy (atorvastatin 20-40 mg daily)
- Children: Generally avoid statins unless persistent severe hyperlipidaemia after remission
Nutritional Management
Protein Intake:
- Recommendation: Normal protein intake (0.8-1 g/kg/day)
- Avoid: High protein diets (do not replace urinary losses; may worsen proteinuria)
- Severe Hypoalbuminaemia: Nutritional support; consider dietitian involvement
Vitamin D Supplementation:
- Loss of vitamin D binding protein in urine → vitamin D deficiency
- Supplementation: Cholecalciferol 1000-2000 IU daily
Other Micronutrients:
- Monitor and replace if deficient (zinc, copper)
Blood Pressure Management
MCD Typically Normotensive:
- Hypertension suggests alternative diagnosis (FSGS, membranous) or complication
If Hypertension Develops:
- Target: less than 130/80 mmHg
- First-line: ACE inhibitor or ARB (antiproteinuric effect)
- Add calcium channel blocker or diuretic as needed
Disposition and Follow-Up
Inpatient Admission Indications:
- Severe oedema (anasarca, respiratory compromise, refractory to outpatient diuretics)
- Complications: Thromboembolism, infection (SBP, severe cellulitis), AKI
- Initiation of IV therapies (albumin + diuretics, IV methylprednisolone)
- Social factors (inability to manage at home)
Outpatient Management:
- Mild-moderate oedema
- Stable renal function
- No complications
Follow-Up Schedule:
| Phase | Frequency | Monitoring |
|---|---|---|
| Active Treatment | Every 2-4 weeks | Weight, BP, oedema, uPCR, albumin, creatinine, glucose |
| Tapering Steroids | Every 4-6 weeks | As above + signs of relapse |
| Remission | Every 3-6 months | uPCR, creatinine; screen for relapse |
| Second-Line Therapy | Every 2-4 weeks initially | Drug levels (CNI), toxicity monitoring |
Patient Education:
- Home urine dipstick monitoring (especially during illness)
- Recognise signs of relapse (oedema, frothy urine)
- Thromboembolism symptoms (leg swelling, dyspnoea)
- Infection symptoms (fever, abdominal pain)
- Steroid side effects
- Importance of adherence and gradual taper (not abrupt cessation)
7. Complications
Complications of Nephrotic Syndrome
Thromboembolism [8]
| Type | Incidence | Presentation | Diagnosis | Management |
|---|---|---|---|---|
| DVT | 5-15% | Unilateral leg swelling, pain, erythema | Doppler USS lower limb | Therapeutic anticoagulation (LMWH → warfarin or DOAC) |
| Pulmonary Embolism | 2-5% | Dyspnoea, pleuritic chest pain, tachycardia | CTPA | Therapeutic anticoagulation; may require ICU |
| Renal Vein Thrombosis | 5-25% | Often asymptomatic; flank pain, haematuria if acute | Doppler USS, CT/MR venography | Anticoagulation ≥6 months |
| Arterial Thrombosis | Rare | Depends on site (stroke, MI, limb ischaemia) | Imaging | Anticoagulation + antiplatelet |
Risk Factors for Thromboembolism:
- Albumin less than 20 g/L (strongest predictor)
- Immobility
- Central venous catheters
- Prior VTE
- Malignancy
- Genetic thrombophilia
Infection [18]
| Type | Organisms | Presentation | Management |
|---|---|---|---|
| Spontaneous Bacterial Peritonitis (SBP) | E. coli, Streptococcus pneumoniae, Klebsiella | Fever, abdominal pain, ascites | Diagnostic tap (neutrophils > 250/mm³); ceftriaxone 2 g IV daily |
| Cellulitis | Streptococcus, Staphylococcus | Erythema, warmth, swelling in oedematous limb | Flucloxacillin ± benzylpenicillin |
| Pneumonia | S. pneumoniae, H. influenzae | Fever, cough, dyspnoea | CXR; amoxicillin or co-amoxiclav |
| Pneumocystis Pneumonia | Pneumocystis jirovecii | Dry cough, dyspnoea (if prolonged high-dose steroids) | High-resolution CT; co-trimoxazole high-dose |
Prevention:
- Pneumococcal and influenza vaccination
- Low threshold for antibiotics
- Avoid unnecessary invasive procedures
Acute Kidney Injury
Causes in MCD Context:
-
Hypovolaemia:
- Over-diuresis
- Reduced effective circulating volume (severe hypoalbuminaemia)
- Management: Careful fluid and diuretic management; IV fluids if prerenal
-
Acute Tubular Necrosis (ATN):
- Sepsis
- Nephrotoxic drugs (NSAIDs, aminoglycosides, contrast)
- Management: Supportive; discontinue nephrotoxins
-
Acute Interstitial Nephritis (AIN):
- Drug reaction (antibiotics, NSAIDs, PPIs)
- Management: Discontinue offending drug; may require steroids
-
Renal Vein Thrombosis:
- May cause AKI
- Management: Anticoagulation
Monitoring:
- Regular creatinine, eGFR
- Urinalysis (if haematuria develops, consider alternative diagnosis or complication)
Hyperlipidaemia and Cardiovascular Risk
- Acute Phase: Universal; cholesterol may exceed 15 mmol/L
- Long-term: Usually resolves with remission
- Cardiovascular Risk: Unclear if short-term hyperlipidaemia increases long-term CV risk; prolonged nephrotic syndrome may contribute
Protein Malnutrition
- Rare unless prolonged nephrotic syndrome
- Loss of proteins other than albumin (transferrin, immunoglobulins)
- May require nutritional support
Vitamin D Deficiency and Bone Disease
- Loss of vitamin D binding protein → vitamin D deficiency → secondary hyperparathyroidism, bone disease
- Management: Vitamin D supplementation; monitor calcium, phosphate, PTH
Treatment-Related Complications
Corticosteroid Complications [14]
Metabolic:
- Diabetes mellitus (5-10% develop steroid-induced diabetes)
- Dyslipidaemia
- Weight gain, central obesity
- Growth retardation (children)
Cardiovascular:
- Hypertension
- Increased CV risk (prolonged use)
Musculoskeletal:
- Osteoporosis and fractures (10-20% if prolonged use)
- Avascular necrosis (hips, knees)—rare but serious
- Proximal myopathy
Gastrointestinal:
- Peptic ulcer disease
- Pancreatitis (rare)
Ophthalmological:
- Posterior subcapsular cataracts (prolonged use)
- Glaucoma
Neuropsychiatric:
- Mood changes, insomnia, anxiety
- Psychosis (rare)
Infection:
- Opportunistic infections
- TB reactivation
- Varicella-zoster reactivation
Other:
- Cushingoid features (moon face, buffalo hump, striae)
- Skin thinning, easy bruising
- Acne
Mitigation: See steroid risk mitigation section above
Cyclophosphamide Toxicity [15]
- Bone marrow suppression (10-20%): Monitor FBC weekly
- Gonadal toxicity (30-60% in adults): Discuss fertility preservation
- Haemorrhagic cystitis (5-10%): Encourage high fluid intake; mesna if high-dose IV
- Malignancy: Bladder cancer, lymphoma (long-term risk, 1-2%)
- Infection: Increased risk during treatment
Calcineurin Inhibitor Toxicity [16]
- Nephrotoxicity: Acute (reversible) and chronic (interstitial fibrosis); monitor creatinine closely
- Hypertension (40-60%): Treat aggressively
- Metabolic: Hyperglycaemia, hyperlipidaemia, hyperuricaemia (gout)
- Neurotoxicity: Tremor (common), seizures (rare)
- Cosmetic: Gingival hyperplasia, hirsutism (ciclosporin > tacrolimus)
- Malignancy: Skin cancer (sun protection)
Rituximab Toxicity [17]
- Infusion reactions (30-50% first infusion): Premedicate
- Hypogammaglobulinaemia: Monitor immunoglobulin levels
- Infection risk
- Rare: PML, hepatitis B reactivation, neutropenia
8. Prognosis and Long-Term Outcomes
Renal Outcomes
| Population | Complete Remission Rate | Relapse Rate | Progression to ESKD |
|---|---|---|---|
| Children | 90-95% | 60-70% (at least one relapse) | less than 5% |
| Adults | 80-90% | 50-70% | less than 5% (true MCD) |
Long-Term Renal Prognosis:
- Excellent if true MCD: Progression to ESKD rare (less than 5%)
- Steroid-resistant cases: Often FSGS on re-biopsy; 30-40% progress to ESKD
- Most patients maintain normal renal function long-term despite relapses [6]
Relapse Patterns [5]
Relapse Incidence:
- First relapse: 50-70% within 5 years
- Multiple relapses: 30-50% become steroid-dependent or frequently relapsing
Relapse Risk Factors:
- Younger age at onset
- Male sex (children)
- Delayed time to first remission
- Higher initial proteinuria
Relapse Outcomes:
- Most relapses respond to repeat steroid courses
- Each relapse carries cumulative steroid toxicity risk
- Steroid-sparing agents reduce relapse frequency
Response to Treatment Categories and Outcomes
| Category | Proportion | Long-Term Outcome |
|---|---|---|
| Infrequent Relapsers | 30-40% | Excellent; treat relapses as they occur; minimal cumulative steroid toxicity |
| Frequent Relapsers / Steroid-Dependent | 30-50% | Good renal outcome with steroid-sparing therapy; manage cumulative immunosuppression toxicity |
| Steroid-Resistant | 5-10% | Often FSGS on re-biopsy; 30-40% progress to ESKD if true FSGS; CNI may induce remission if true MCD |
Cardiovascular and Mortality Outcomes
Cardiovascular Risk:
- Short-term hyperlipidaemia during nephrotic syndrome
- Long-term CV risk unclear; likely related to steroid exposure and cumulative cardiovascular risk factors
Mortality:
- Low in MCD
- Mortality primarily related to complications (thromboembolism, infection, treatment toxicity) rather than renal failure
Quality of Life
Factors Affecting QoL:
- Frequent relapses requiring repeated treatment
- Steroid side effects (cosmetic, metabolic, psychological)
- Dietary restrictions (sodium)
- Frequent medical visits and monitoring
Strategies to Improve QoL:
- Steroid-sparing agents to reduce cumulative steroid dose
- Patient education and self-management (home urine monitoring)
- Psychological support
Prognostic Factors
Favourable Prognosis
- Rapid response to steroids (less than 4 weeks)
- Complete remission
- Infrequent relapses
- Childhood onset
- Highly selective proteinuria
Unfavourable Prognosis (Suggesting FSGS Rather Than MCD)
- Steroid resistance (no remission after 16 weeks)
- Persistent haematuria
- Hypertension
- Impaired renal function at presentation
- Older age at onset (> 60 years)
- Non-selective proteinuria
Post-Transplant Outcomes
MCD in Transplant Recipients:
- MCD can recur in kidney transplant, but recurrence rare (less than 5%)
- If recurrence: Responds to steroids (immunosuppression already on board)
- Contrast with FSGS: 30-40% recurrence, often immediate and severe
9. Special Populations and Considerations
Pregnancy and MCD
Pre-Conception Counselling:
- Advise achieving remission before conception
- Risk of relapse during pregnancy (30-50%)
- Discontinue teratogenic medications (cyclophosphamide, MMF, CNI controversial)
Management During Pregnancy:
- Prednisolone safe (does not cross placenta significantly)
- Monitor for relapse (may mimic pre-eclampsia)
- Increased thromboembolism risk (consider prophylactic LMWH)
- Pregnancy outcomes generally good if in remission
Distinguishing Relapse from Pre-Eclampsia:
- Relapse: Proteinuria, oedema, but normotensive
- Pre-eclampsia: Proteinuria, oedema, hypertension, often > 20 weeks gestation
Elderly Patients (> 65 years)
Epidemiology:
- Second peak of adult MCD incidence
- Higher frequency of secondary causes (NSAIDs, malignancy)
Challenges:
- Greater steroid toxicity (osteoporosis, fractures, diabetes, infections)
- Slower response to treatment
- Polypharmacy and drug interactions
Management Modifications:
- Thorough malignancy screening
- Aggressive bone protection (bisphosphonates)
- Lower threshold for steroid-sparing agents
- Close monitoring for steroid complications
Secondary MCD
Drug-Induced MCD [7]
NSAIDs:
- Most common drug cause
- May occur with short-term or chronic use
- Management: Discontinue NSAID; spontaneous remission in 50-70%; steroids if persistent
Other Drugs:
- Lithium, interferon, ampicillin, bisphosphonates, checkpoint inhibitors
- Management: Discontinue drug; consider steroids if no spontaneous remission
Malignancy-Associated MCD [10]
Hodgkin Lymphoma:
- Classic paraneoplastic association
- MCD may precede, coincide with, or follow lymphoma diagnosis
- Management: Treat lymphoma; MCD often remits with cancer treatment; may require steroids concurrently
Other Malignancies:
- Non-Hodgkin lymphoma, thymoma, solid tumours (rare)
- Screening: Age-appropriate; high suspicion if constitutional symptoms
Paediatric MCD (Brief Overview for Context)
Differences from Adults:
- Much higher incidence (80-90% of childhood nephrotic syndrome)
- Faster response to steroids (2-4 weeks)
- Empirical steroid treatment standard (no biopsy if typical presentation)
- Higher relapse rates
- Shorter steroid courses (8-12 weeks total)
- Growth concerns with prolonged steroids
10. Evidence Base and Guidelines
Key Clinical Practice Guidelines
-
KDIGO 2021 Clinical Practice Guideline for the Management of Glomerular Diseases [1]
- Comprehensive guideline covering MCD diagnosis and management
- Recommendations for corticosteroid regimens in adults and children
- Guidance on second-line therapies
- Key Recommendations:
- Adults: Prednisolone 1 mg/kg/day for minimum 4 weeks, up to 16 weeks
- Renal biopsy mandatory in adults before treatment
- Second-line agents: Cyclophosphamide, CNI, rituximab for steroid-dependent disease
-
Renal Association UK Clinical Practice Guidelines: Glomerulonephritis
- UK-specific guidance aligned with KDIGO
- Emphasis on multidisciplinary care
-
European Rare Kidney Disease Reference Network (ERKNet) Guidelines
- Focus on rare glomerular diseases including adult MCD
Landmark Studies and Key Evidence
Pathophysiology and Mechanisms
Shalhoub Hypothesis (1974): [3]
- Seminal hypothesis proposing T-cell derived circulating permeability factor
- Basis: Response to T-cell immunosuppression, association with atopy
- Evidence: T-cell supernatants induce proteinuria in experimental models
- PMID: 4549040 (historical reference)
Podocyte Biology and Foot Process Effacement: [12]
- Understanding of podocyte structure and slit diaphragm proteins (nephrin, podocin)
- Reversible actin cytoskeleton reorganisation in MCD versus irreversible injury in FSGS
- Key Review: Vivarelli M et al. Minimal Change Disease. Clin J Am Soc Nephrol. 2017;12(2):332-345. PMID: 27940460
Corticosteroid Treatment
KDIGO Meta-Analysis (2021): [1]
- Adults require longer steroid courses than children (16 weeks versus 8 weeks)
- 80-90% remission rate in adults
- Relapse rate 50-70%
PREDNOS Trials (Paediatric):
- Compared different steroid regimens in childhood nephrotic syndrome
- Longer steroid courses reduce relapse rates
- Standard: 12 weeks total (6 weeks daily, 6 weeks alternate-day)
Rituximab in MCD
Ruggenenti P et al. Rituximab in Steroid-Dependent or Frequently Relapsing Idiopathic Nephrotic Syndrome. J Am Soc Nephrol. 2014;25(4):850-863. PMID: 24480825 [17]
- Multicentre RCT
- Rituximab reduced relapses and steroid exposure in steroid-dependent nephrotic syndrome
- 60-80% sustained remission
Iijima K et al. Rituximab for Childhood-Onset, Complicated, Frequently Relapsing Nephrotic Syndrome or Steroid-Dependent Nephrotic Syndrome: A Multicentre, Double-Blind, Randomised, Placebo-Controlled Trial. Lancet. 2014;384(9950):1273-1281. PMID: 24965823
- Paediatric RCT
- Rituximab significantly reduced relapse rate
- Emerging as steroid-sparing option
Calcineurin Inhibitors
Systematic Review and Meta-Analysis:
- Ciclosporin and tacrolimus effective in steroid-dependent and steroid-resistant MCD [16]
- 80-85% achieve remission
- High relapse rate on withdrawal (60-70%)
Thromboembolism Risk
Kerlin BA et al. Epidemiology and Risk Factors for Thromboembolic Complications of Childhood Nephrotic Syndrome: A Midwest Pediatric Nephrology Consortium (MWPNC) Study. J Pediatr. 2009;155(1):105-110. PMID: 19394032 [8]
- Thromboembolism incidence: 1.8-5% (paediatric); higher in adults
- Risk highest when albumin less than 20 g/L
Mahmoodi BK et al. Association of Mild to Moderate Chronic Kidney Disease With Venous Thromboembolism: Pooled Analysis of Five Prospective General Population Cohorts. Circulation. 2012;126(16):1964-1971. PMID: 22977131
- Nephrotic syndrome increases VTE risk 3-8 fold
Long-Term Outcomes
Waldman M et al. Adult Minimal-Change Disease: Clinical Characteristics, Treatment, and Outcomes. Clin J Am Soc Nephrol. 2007;2(3):445-453. PMID: 17699450 [6]
- Long-term follow-up of adult MCD patients
- 90% achieve remission; less than 5% progress to ESKD
- Relapse rate 50-70%; 30-50% become steroid-dependent
Vivarelli M et al. Minimal Change Disease. Clin J Am Soc Nephrol. 2017;12(2):332-345. PMID: 27940460 [2]
- Comprehensive review of MCD epidemiology, pathophysiology, management
- Excellent long-term renal prognosis despite frequent relapses
Ongoing Research and Emerging Therapies
Investigational Therapies:
- Ofatumumab: Alternative anti-CD20 antibody
- ACTH (Adrenocorticotropic Hormone): May have direct podocyte effects beyond steroid induction
- Galactose: Anecdotal reports in recurrent FSGS post-transplant; mechanism unclear
- Abatacept (CTLA4-Ig): Targets CD80; controversial results
Research Priorities:
- Identification of circulating permeability factor(s)
- Biomarkers to predict steroid response and relapse
- Precision medicine approaches based on genetic/molecular profiling
- Novel podocyte-protective therapies
11. Differential Diagnosis
Conditions to Consider in Adult Nephrotic Syndrome
| Condition | Key Distinguishing Features | Histology | Management Differs? |
|---|---|---|---|
| Focal Segmental Glomerulosclerosis (FSGS) | Hypertension common; haematuria may be present; lower steroid response (30-40%); impaired renal function | LM: Segmental sclerosis; EM: Focal foot process effacement | Yes: Worse prognosis; different second-line therapies |
| Membranous Nephropathy | Older adults; gradual onset; associated with malignancy, autoimmune disease; anti-PLA2R antibodies (70%) | LM: Thickened GBM; IF: Granular IgG and C3; EM: Subepithelial deposits | Yes: Different treatment (rituximab first-line if high-risk) |
| Diabetic Nephropathy | History of diabetes; gradual proteinuria progression; diabetic retinopathy often present | LM: Mesangial expansion, Kimmelstiel-Wilson nodules; GBM thickening | Yes: Glycaemic control, RAAS blockade; no steroids |
| Amyloidosis (AL or AA) | Systemic symptoms; hepatosplenomegaly; restrictive cardiomyopathy; monoclonal protein (AL) | LM: Congo red positive; EM: Fibrils | Yes: Treat underlying cause (chemotherapy for AL; anti-inflammatory for AA) |
| Lupus Nephritis (Class V) | Systemic lupus features; positive ANA, anti-dsDNA; low complement | LM: Variable; IF: Full house (IgG, IgA, IgM, C3, C1q); EM: Subepithelial deposits | Yes: Immunosuppression for lupus (hydroxychloroquine, MMF, cyclophosphamide) |
| IgA Nephropathy | Haematuria (macro or micro); history of synpharyngitic haematuria | LM: Mesangial proliferation; IF: Dominant IgA deposition | Yes: RAAS blockade; steroids only if high risk |
| Post-Infectious Glomerulonephritis | Recent infection (strep throat, skin); low C3 (recovers); haematuria, hypertension | LM: Proliferative GN; IF: C3, IgG; EM: Subepithelial humps | Yes: Supportive; steroids not indicated |
Clinical Clues to Distinguish MCD from Other Causes
Strongly Suggests MCD:
- ✅ Normotensive
- ✅ No haematuria
- ✅ Normal renal function
- ✅ Highly selective proteinuria
- ✅ Rapid onset
- ✅ History of atopy
Suggests Alternative Diagnosis:
- ❌ Hypertension → FSGS, membranous, diabetic nephropathy
- ❌ Haematuria → IgA nephropathy, vasculitis, post-infectious GN
- ❌ Impaired renal function → FSGS, diabetic nephropathy, amyloidosis
- ❌ Systemic symptoms → Lupus, amyloidosis, malignancy
- ❌ Low complement → Lupus, post-infectious GN, MPGN
Key Principle: In adults, renal biopsy is MANDATORY to distinguish MCD from other causes of nephrotic syndrome. Clinical features alone are insufficient.
12. Patient Information and Counselling
What is Minimal Change Disease?
Minimal change disease is a condition affecting the filters in your kidneys (called glomeruli). These filters normally prevent protein from leaking into your urine. In MCD, the filters become "leaky" and allow large amounts of protein (especially albumin) to pass into the urine.
Under a standard microscope, the kidney filters look almost normal (hence "minimal change"). However, using a powerful electron microscope, doctors can see tiny changes in the cells that make up the filter (called podocytes).
What Causes It?
The exact cause is unknown in most cases (called "primary" or "idiopathic" MCD). It is thought to involve the immune system producing substances that damage the kidney filters.
In some adults, MCD can be triggered by:
- Medications (especially anti-inflammatory painkillers like ibuprofen)
- Infections
- Rarely, certain cancers (especially Hodgkin lymphoma)
What Are the Symptoms?
- Swelling (oedema): Usually starts around the eyes (especially in the morning) and ankles, and can spread to the legs, abdomen, and other areas
- Frothy or bubbly urine: Due to protein in the urine
- Weight gain: From fluid retention
- Fatigue: Feeling tired and unwell
How is it Diagnosed?
- Urine tests: To measure protein in urine
- Blood tests: To check protein levels (albumin), kidney function, and cholesterol
- Kidney biopsy: In adults, a small sample of kidney tissue is taken (under local anaesthetic) and examined under a microscope. This is essential to confirm the diagnosis.
What is the Treatment?
Steroids (Prednisolone):
- The main treatment is steroid tablets (prednisolone), usually for 4-6 months
- About 80-90% of adults respond to steroids and achieve remission (protein in urine returns to normal)
- Steroids take longer to work in adults than in children (may take 2-4 months)
Side Effects of Steroids:
- Increased appetite and weight gain
- Mood changes, difficulty sleeping
- Increased blood sugar (risk of diabetes)
- Weakened bones (you may need calcium, vitamin D, and bone-strengthening tablets)
- Increased infection risk
If Steroids Don't Work or You Relapse:
- About half of patients have a relapse (the condition comes back) after stopping steroids
- Other medications may be used, such as cyclophosphamide, ciclosporin, tacrolimus, or rituximab
Supportive Treatment:
- Water tablets (diuretics) to reduce swelling
- Low-salt diet
- Blood thinners if protein levels are very low (to prevent blood clots)
- Cholesterol-lowering tablets if needed
What is the Outlook?
- Excellent long-term outlook: MCD responds very well to treatment, and progression to kidney failure is rare (less than 5%)
- Relapses are common: About 50-70% of people have at least one relapse, but these usually respond to treatment again
- Normal kidney function long-term: Most people maintain normal kidney function despite relapses
What Should I Watch For?
Contact your doctor urgently if you develop:
- Sudden increase in swelling
- Difficulty breathing
- Fever or signs of infection
- Leg pain, swelling, or redness (may indicate blood clot)
- Blood in urine
Monitor at home:
- Use urine dipsticks to check for protein during illnesses (your doctor can provide these)
- Weigh yourself daily during active disease
- Report early signs of relapse (frothy urine, swelling) to your doctor
Lifestyle and Self-Care
- Diet: Reduce salt intake (avoid processed foods, don't add salt to meals)
- Fluid: Limit fluids if swelling is severe (ask your doctor)
- Protein: Normal protein intake (do not restrict protein)
- Infections: Avoid people with infections; get flu and pneumonia vaccines
- Medications: Avoid anti-inflammatory painkillers (ibuprofen, naproxen) unless approved by your doctor
- Pregnancy: Discuss with your doctor before conceiving; need to adjust medications
Questions to Ask Your Doctor
- Do I need a kidney biopsy?
- How long will I need to take steroids?
- What are the signs of relapse?
- Do I need blood thinners to prevent clots?
- When should I come back for follow-up?
- Can I take over-the-counter painkillers?
- What vaccines do I need?
13. Clinical Viva and Examination Preparation
Viva Stem Question
"A 28-year-old man presents with a 2-week history of ankle swelling and frothy urine. Blood pressure 125/78 mmHg. Urinalysis shows 4+ proteinuria, no haematuria. Serum albumin 18 g/L, creatinine 78 µmol/L. What is your differential diagnosis and how would you investigate?"
Model Answer:
"This patient has nephrotic syndrome, defined by heavy proteinuria, hypoalbuminaemia, and oedema. In a young adult, the differential includes minimal change disease, focal segmental glomerulosclerosis, and membranous nephropathy. Secondary causes such as diabetic nephropathy, lupus, and amyloidosis should also be considered.
Key features favouring MCD are the young age, normotensive, normal renal function, and absence of haematuria. However, in adults, renal biopsy is mandatory to distinguish MCD from FSGS and membranous nephropathy, as management and prognosis differ.
Investigations:
- Quantify proteinuria: 24-hour urine protein or urine protein:creatinine ratio (expect > 300 mg/mmol)
- Exclude secondary causes: Complement C3/C4 (low in lupus, post-infectious GN), glucose (diabetes), serum/urine electrophoresis (myeloma in older patients), hepatitis B/C and HIV serology, ANA and ANCA if systemic features
- Renal ultrasound pre-biopsy
- Renal biopsy: Essential in adults; MCD shows normal light microscopy, negative immunofluorescence, and diffuse foot process effacement on electron microscopy
Treatment would be prednisolone 1 mg/kg/day for a minimum of 4 weeks, extended to 16 weeks if needed. I would also provide supportive care: sodium restriction, diuretics for oedema, and consider thromboprophylaxis given severe hypoalbuminaemia. Expected remission rate is 80-90%, though relapse occurs in 50-70%."
High-Yield Facts for Exams
Epidemiology:
- Children: 80-90% of nephrotic syndrome
- Adults: 10-15% of nephrotic syndrome (FSGS and membranous more common)
Pathophysiology:
- T-cell dysfunction → circulating permeability factor → podocyte injury → foot process effacement → massive proteinuria
Histology (MUST KNOW):
- LM: Normal (hence "minimal change")
- IF: Negative or trace IgM
- EM: Diffuse podocyte foot process effacement (pathognomonic)
Clinical:
- Normotensive, no haematuria, normal renal function (distinguishes from other GN)
- Highly selective proteinuria (albumin predominant)
Management:
- Adults: Prednisolone 1 mg/kg/day × 4-16 weeks (longer than children)
- Remission: 80-90% adults, 90-95% children
- Relapse: 50-70%
- Second-line: Cyclophosphamide, CNI (ciclosporin/tacrolimus), rituximab
Complications:
- Thromboembolism (5-25%), especially if albumin less than 20 g/L
- Infection (loss of immunoglobulins and complement)
- Steroid toxicity
Prognosis:
- Excellent: less than 5% progress to ESKD (steroid-resistant often FSGS on re-biopsy)
Common Examination Mistakes
❌ Mistake 1: Treating adult nephrotic syndrome empirically without biopsy
- ✅ Correct: Biopsy mandatory in adults (unlike children with typical presentation)
❌ Mistake 2: Stopping steroids too early (e.g., at 4 weeks) if no response
- ✅ Correct: Adults may take up to 16 weeks to respond; continue steroids
❌ Mistake 3: Forgetting thromboprophylaxis
- ✅ Correct: Consider anticoagulation if albumin less than 20 g/L
❌ Mistake 4: Calling steroid-resistant MCD "treatment failure"
- ✅ Correct: Re-biopsy to exclude FSGS (sampling error); true MCD rarely steroid-resistant
❌ Mistake 5: Not screening for secondary causes in adults
- ✅ Correct: NSAIDs, malignancy (Hodgkin), drugs—always inquire and screen
Viva Questioning Themes
Pathophysiology:
- "What is the pathogenesis of MCD?"
- "Why does foot process effacement cause proteinuria?"
- "Why is proteinuria 'selective' in MCD?"
Diagnosis:
- "How do you distinguish MCD from FSGS clinically?" (Answer: Cannot reliably; biopsy required)
- "What are the biopsy findings in MCD?"
- "Why biopsy adults but not children?"
Management:
- "What is the steroid regimen for adult MCD?"
- "How do you define steroid resistance?"
- "What are the options for steroid-dependent disease?"
Complications:
- "Why is nephrotic syndrome a hypercoagulable state?"
- "What infections are patients at risk of?"
Prognosis:
- "What is the long-term renal prognosis?"
- "What proportion relapse?"
MRCP/FRACP-Style MCQ Example
A 32-year-old woman presents with generalised oedema and frothy urine. BP 118/72 mmHg. Investigations: Serum albumin 16 g/L, creatinine 65 µmol/L, urine protein 8.2 g/24h. Renal biopsy shows normal glomeruli on light microscopy, negative immunofluorescence, and diffuse foot process effacement on electron microscopy. She is started on prednisolone 60 mg daily. After 6 weeks, proteinuria persists (uPCR 450 mg/mmol). What is the most appropriate next step?
A. Stop prednisolone and start cyclophosphamide B. Continue prednisolone for a further 10 weeks C. Perform repeat renal biopsy D. Add ciclosporin to prednisolone E. Refer for plasma exchange
Answer: B. Continue prednisolone for a further 10 weeks
Rationale: Adults with MCD may take up to 16 weeks to respond to steroids (versus 4 weeks in children). This patient has completed only 6 weeks. Steroid resistance is defined as failure to remit after 16 weeks of adequate therapy. The appropriate next step is to continue prednisolone up to 16 weeks total before considering second-line therapy or re-biopsy.
14. References
-
KDIGO Glomerular Diseases Work Group. KDIGO 2021 Clinical Practice Guideline for the Management of Glomerular Diseases. Kidney Int. 2021;100(4S):S1-S276. PMID: 34556300
-
Vivarelli M, Massella L, Ruggiero B, Emma F. Minimal Change Disease. Clin J Am Soc Nephrol. 2017;12(2):332-345. PMID: 27940460
-
Shalhoub RJ. Pathogenesis of lipoid nephrosis: a disorder of T-cell function. Lancet. 1974;2(7880):556-560. PMID: 4140273
-
Hogan J, Radhakrishnan J. The treatment of minimal change disease in adults. J Am Soc Nephrol. 2013;24(5):702-711. PMID: 23547108
-
Waldman M, Crew RJ, Valeri A, et al. Adult minimal-change disease: clinical characteristics, treatment, and outcomes. Clin J Am Soc Nephrol. 2007;2(3):445-453. PMID: 17699450
-
Shinzawa M, Yamamoto R, Nagasawa Y, et al. Comparison of methylprednisolone plus prednisolone with prednisolone alone as initial treatment in adult-onset minimal change disease: a retrospective cohort study. Clin J Am Soc Nephrol. 2014;9(7):1040-1048. PMID: 24742480
-
Tsakopoulos M, Gjyzari E, Kalligeros M, Shemin D. Drug-induced minimal change disease: A systematic review. J Nephrol. 2023;36(6):1625-1642. PMID: 37682269
-
Kerlin BA, Ayoob R, Smoyer WE. Epidemiology and pathophysiology of nephrotic syndrome-associated thromboembolic disease. Clin J Am Soc Nephrol. 2012;7(3):513-520. PMID: 22344511
-
Maas RJ, Deegens JK, Smeets B, Moeller MJ, Wetzels JF. Minimal change disease and idiopathic FSGS: manifestations of the same disease. Nat Rev Nephrol. 2016;12(8):445-456. PMID: 27297766
-
Audard V, Larousserie F, Grimbert P, et al. Minimal change nephrotic syndrome and classical Hodgkin's lymphoma: report of 21 cases and review of the literature. Kidney Int. 2006;69(12):2251-2260. PMID: 16672911
-
Kaneko K. Pathogenesis of minimal change nephrotic syndrome: a still unresolved enigma. Pediatr Int. 2013;55(3):277-281. PMID: 23731357
-
Faul C, Asanuma K, Yanagida-Asanuma E, Kim K, Mundel P. Actin up: regulation of podocyte structure and function by components of the actin cytoskeleton. Trends Cell Biol. 2007;17(9):428-437. PMID: 17804239
-
Vaziri ND. Disorders of lipid metabolism in nephrotic syndrome: mechanisms and consequences. Kidney Int. 2016;90(1):41-52. PMID: 27165817
-
Briot K, Roux C. Glucocorticoid-induced osteoporosis. RMD Open. 2015;1(1):e000014. PMID: 26509049
-
Ponticelli C, Edefonti A, Ghio L, et al. Cyclosporin versus cyclophosphamide for patients with steroid-dependent and frequently relapsing idiopathic nephrotic syndrome: a multicentre randomized controlled trial. Nephrol Dial Transplant. 1993;8(12):1326-1332. PMID: 8159343
-
Li X, Li H, Chen J, et al. Tacrolimus as a steroid-sparing agent for adults with steroid-dependent minimal change nephrotic syndrome. Nephrol Dial Transplant. 2008;23(6):1919-1925. PMID: 18252698
-
Ruggenenti P, Ruggiero B, Cravedi P, et al. Rituximab in steroid-dependent or frequently relapsing idiopathic nephrotic syndrome. J Am Soc Nephrol. 2014;25(4):850-863. PMID: 24480825
-
Ogi M, Yokoyama H, Tomosugi N, et al. Risk factors for infection and immunoglobulin replacement therapy in adult nephrotic syndrome. Am J Kidney Dis. 1994;24(3):427-436. PMID: 8079965
-
Hladunewich MA, Cattran D, Sethi S, et al. Glomerular disease in pregnancy. Clin J Am Soc Nephrol. 2014;9(11):1944-1953. PMID: 24925967
-
Rauen T, Michaelis A, Floege J, Mertens PR. Case series of idiopathic minimal change disease in adults: the role of drug treatment and malignancies. BMC Nephrol. 2013;14:186. PMID: 24004612
-
Takei T, Itabashi M, Moriyama T, et al. Short-term steroid therapy for adult minimal-change nephrotic syndrome. Nephrol Dial Transplant. 2001;16(10):1994-1999. PMID: 11572887
-
Crew RJ, Radhakrishnan J, Appel G. Complications of the nephrotic syndrome and their treatment. Clin Nephrol. 2004;62(4):245-259. PMID: 15524053