Multiple Sclerosis (Adult)
Multiple Sclerosis (MS) is a chronic, immune-mediated inflammatory demyelinating disease of the central nervous system (... MRCP, Neurology Specialty exam prepa
What matters first
Multiple Sclerosis (MS) is a chronic, immune-mediated inflammatory demyelinating disease of the central nervous system (... MRCP, Neurology Specialty exam prepa
Neuromyelitis optica (NMO) misdiagnosis - requires different treatment
6 Jan 2026
Generated educational material; verify before clinical use.
Visible references section
See the concept before reading it
Study the key anatomy, imaging, and decision pathways as full teaching plates.
Clinical board
A visual summary of the highest-yield teaching signals on this page.
Urgent signals
Safety-critical features pulled from the topic metadata.
- Neuromyelitis optica (NMO) misdiagnosis - requires different treatment
- Acute cord compression mimicking transverse myelitis
- PML risk with natalizumab in JC virus positive patients
- Secondary autoimmunity with alemtuzumab
Exam focus
Current exam surfaces linked to this topic.
- MRCP
- Neurology Specialty
Linked comparisons
Differentials and adjacent topics worth opening next.
- Neuromyelitis Optica Spectrum Disorder (NMOSD)
- ADEM (Acute Disseminated Encephalomyelitis)
Content status and exam context
This page is AI-generated educational content. It may contain errors or omissions and is not a substitute for current guidelines, local protocols, senior clinical judgement, or professional medical advice.
MedVellum does not claim an individual clinician reviewer, board certification, or professional credential for this page unless a future version names a real, verifiable reviewer.
Clinical explanation and evidence
1. Clinical Overview
Definition and Significance
Multiple Sclerosis (MS) is a chronic, immune-mediated inflammatory demyelinating disease of the central nervous system (CNS) characterized by spatial and temporal dissemination of demyelinating lesions. It represents the most common non-traumatic neurological cause of disability in young adults worldwide. [1,2]
Core Pathological Features:
- Multifocal areas of demyelination (plaques) predominantly affecting white matter
- Inflammatory infiltrates with T-lymphocytes, B-lymphocytes, and activated microglia
- Axonal transection and neurodegeneration
- Blood-brain barrier disruption during active inflammation
- Gliotic scarring ("sclerosis") at lesion sites [1,3]
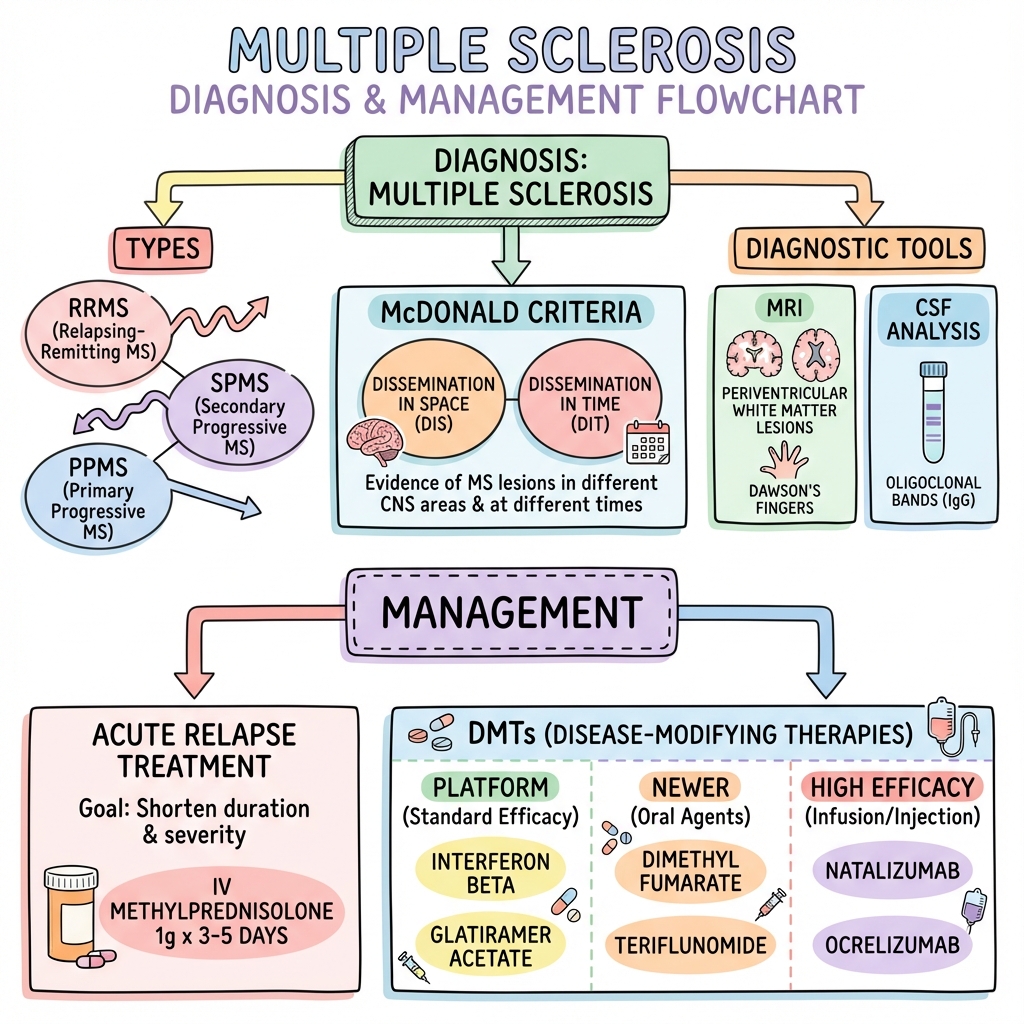
Clinical Hallmarks: The diagnosis requires demonstration of lesions disseminated in space (multiple CNS locations) and disseminated in time (occurring at different timepoints), as formalized in the McDonald Diagnostic Criteria. [4,5]
Clinical Significance:
- Prevalence: 100-300 per 100,000 in high-prevalence regions (increasing globally) [1,2]
- Median age of onset: 30 years (range 20-40 years)
- Female predominance: Female:Male ratio = 3:1 [2]
- Socioeconomic impact: Leading cause of non-traumatic disability in young adults
- Modern disease-modifying therapies (DMTs) have revolutionized outcomes, reducing disability accumulation by up to 50% in treated populations [6,7]
Key Clinical Associations and Eponyms
Classical Signs:
- Lhermitte's Sign: Electric shock-like sensation radiating down the spine with neck flexion, indicating posterior column demyelination in the cervical spinal cord
- Uhthoff's Phenomenon: Transient worsening of neurological symptoms with increased body temperature (exercise, hot bath, fever) due to temperature-dependent conduction block in demyelinated axons
- Internuclear Ophthalmoplegia (INO): Failure of adduction of the ipsilateral eye with nystagmus of the contralateral abducting eye, pathognomonic for medial longitudinal fasciculus (MLF) lesion in the brainstem [1,8]
Charcot's Triad (Historical): Nystagmus, intention tremor, and scanning (staccato) speech - originally described as characteristic of MS, though not commonly seen together in modern practice
Dawson's Fingers: Characteristic periventricular lesions on MRI oriented perpendicular to the lateral ventricles, reflecting perivenular inflammation [9]
"Do Not Miss" Warnings and Differential Diagnoses
⚠️ Warning: Critical Differential: Neuromyelitis Optica Spectrum Disorder (NMOSD)
NMOSD mimics MS but represents a distinct autoimmune condition targeting aquaporin-4 water channels. Differentiation is critical as MS disease-modifying therapies (particularly interferon-beta, fingolimod, and natalizumab) can worsen NMOSD. [10]
Red Flags for NMOSD:
- Severe, bilateral optic neuritis with prolonged visual loss or blindness
- Longitudinally extensive transverse myelitis (LETM) spanning ≥3 vertebral segments
- Area postrema syndrome (intractable hiccups, nausea, vomiting)
- Aquaporin-4 (AQP4) antibody positivity
- Asian, African, or Latin American ethnicity
- Brain MRI lesions predominantly in hypothalamus, brainstem (area postrema), or periependymal areas
Action: Test for AQP4-IgG antibodies in all suspected MS cases, especially with severe optic neuritis or extensive myelitis.
⚠️ Warning: Acute Spinal Cord Compression
Acute myelopathy presenting as transverse myelitis may represent cord compression requiring urgent neurosurgical intervention. Obtain urgent MRI spine before attributing acute cord syndrome to MS demyelination.
Red Flags:
- Severe progressive motor deficit over hours to days
- Spinal sensory level
- Acute bladder/bowel dysfunction
- Severe back pain
⚠️ Warning: Progressive Multifocal Leukoencephalopathy (PML)
PML is a severe, often fatal opportunistic CNS infection caused by JC virus reactivation in immunosuppressed patients receiving certain DMTs (particularly natalizumab). [11,12]
Risk Factors:
- Natalizumab therapy (highest risk: JC virus antibody positive, duration > 2 years, prior immunosuppressant exposure)
- Fingolimod, dimethyl fumarate (lower risk)
- Prolonged lymphopenia
Clinical Features:
- Subacute cognitive decline, personality change, motor deficits, visual disturbances
- Progressive neurological deterioration without fever
- MRI: Large, asymmetric, subcortical white matter lesions without mass effect or enhancement (unlike MS plaques)
Action: Screen JC virus antibody status before initiating natalizumab. Monitor with serial MRI and consider plasma exchange/immunoadsorption if PML suspected.
2. Epidemiology & Risk Factors
Incidence and Prevalence
Global Distribution:
- Worldwide prevalence: ~2.8 million people affected [1,2]
- Geographical gradient: Prevalence increases with latitude (latitude effect)
- "Low-prevalence regions (equatorial): 5-20 per 100,000"
- "Moderate-prevalence: 20-100 per 100,000"
- "High-prevalence regions (Scandinavia, Scotland, Canada, Northern USA): 100-300 per 100,000 [2]"
- Prevalence is increasing globally, partly due to improved diagnosis and survival [1,2]
Demographics:
- Age of onset: Peak incidence 20-40 years; rare before puberty or after age 60
- Sex distribution: Female:Male = 3:1 (increasing female predominance noted in recent decades) [2]
- Ethnicity: Highest in Caucasian populations of Northern European descent; lower in Asian, African, and Indigenous populations
Migration Studies: Migration before puberty adopts the risk of the new geographical location; migration after puberty retains risk of the country of origin, suggesting critical environmental exposures during childhood/adolescence. [1]
Risk Factors
Exam Detail: | Risk Factor | Relative Risk | Mechanism/Evidence | Level of Evidence | |:----------------|:------------------|:-----------------------|:----------------------| | Epstein-Barr Virus (EBV) | OR 32 (if no prior infection) | Nearly 100% of MS patients are EBV-seropositive. Molecular mimicry, polyclonal B-cell activation, and latent CNS infection hypothesized. [13] | Level I | | Low Vitamin D | HR 1.6-2.0 | Higher latitudes correlate with low UV exposure and reduced vitamin D synthesis. Vitamin D has immunomodulatory effects. [13] | Level II | | Smoking | HR 1.5 | Dose-dependent increased MS risk and accelerated progression to secondary progressive MS. [13] | Level I | | HLA-DRB1*15:01 | OR 3.0 | Strongest genetic association (MHC Class II allele). Polygenic inheritance with > 200 risk alleles identified. [1] | Level I | | Adolescent Obesity | OR 1.4-2.0 | Pro-inflammatory adipokine milieu hypothesized mechanism. [13] | Level II | | First-degree relative with MS | 15-20% lifetime risk | Polygenic inheritance pattern; concordance in monozygotic twins ~25-30%. [1] | Level II | | Other autoimmune diseases | Increased risk | Clustering with autoimmune thyroid disease, type 1 diabetes, inflammatory bowel disease. | Level II |
Environmental Hypotheses:
- "Hygiene Hypothesis": Reduced childhood infection exposure may predispose to autoimmunity
- Gut microbiome dysbiosis: Emerging evidence links altered gut microbiota composition to MS risk and disease activity [1]
- Diet: Low vitamin D, high salt intake, and Western diet patterns may increase risk
3. Pathophysiology
Molecular and Cellular Mechanisms
Exam Detail: Phase 1: Autoimmune Activation and CNS Infiltration
-
Peripheral Immune Activation:
- Autoreactive T-cells (CD4+ Th1 and Th17 subsets) escape thymic tolerance or become activated by environmental triggers (EBV, molecular mimicry)
- These cells target CNS myelin antigens (myelin basic protein, myelin oligodendrocyte glycoprotein, proteolipid protein) [1,3]
-
Blood-Brain Barrier (BBB) Disruption:
- Activated T-cells upregulate adhesion molecules (VLA-4, LFA-1) and interact with vascular endothelium
- Matrix metalloproteinases (MMPs) degrade tight junctions
- T-cells, B-cells, and macrophages cross the BBB into CNS parenchyma [3]
-
Inflammatory Cascade:
- T-cells release pro-inflammatory cytokines (IFN-γ, IL-17, TNF-α)
- Microglial activation and macrophage recruitment
- B-cells differentiate into plasma cells producing oligoclonal antibodies (detected as oligoclonal bands in CSF)
- Complement activation contributes to myelin destruction [1,3]
Phase 2: Demyelination
- Primary Target: Oligodendrocytes (myelin-producing cells) undergo apoptosis or direct antibody-mediated injury
- Myelin Breakdown: Phagocytosis of myelin debris by macrophages
- Axonal Exposure: Loss of myelin sheath exposes axons to inflammatory mediators
- Conduction Failure: Saltatory conduction is lost; exposed axons exhibit slowed conduction or complete conduction block [3]
Phase 3: Axonal Injury and Neurodegeneration
- Acute Axonal Transection: Inflammatory mediators (nitric oxide, reactive oxygen species, glutamate excitotoxicity) cause axonal transection within acute lesions
- Chronic Axonal Loss: Chronic demyelination leads to mitochondrial dysfunction, energy failure, and progressive axonal degeneration
- Neuronal Loss: Cortical demyelination and grey matter atrophy contribute to cognitive decline and disability progression
- "Irreversible Disability": Axonal loss is the pathological correlate of permanent neurological deficits [1,3]
Phase 4: Attempted Repair and Lesion Evolution
- Remyelination: Oligodendrocyte precursor cells (OPCs) may differentiate and remyelinate axons (partial or complete), explaining clinical recovery after relapses
- Incomplete Repair: Remyelination is often incomplete or fails in chronic lesions, leading to persistent "shadow plaques"
- Gliotic Scarring: Astrocytic gliosis forms the characteristic "sclerotic plaques" visible on pathology [3]
Lesion Distribution and Imaging Correlates
Characteristic Locations:
- Periventricular white matter: "Dawson's Fingers"
- lesions perpendicular to lateral ventricles along venules [9]
- Juxtacortical/Cortical: Lesions at grey-white matter junction or intracortical (U-fibers often spared in MS, unlike ADEM)
- Infratentorial: Brainstem (medial longitudinal fasciculus, pons, medulla), cerebellar peduncles
- Spinal cord: Cervical > thoracic; typically short (≤2 vertebral segments) and peripheral/lateral (unlike NMOSD with central, extensive lesions) [9]
- Optic nerve: Acute optic neuritis (often the first clinical event)
MRI Characteristics:
- T2/FLAIR: Hyperintense lesions
- T1 post-gadolinium: Enhancement indicates active inflammation and BBB breakdown (lesions enhance for ~4-6 weeks)
- T1 "black holes": Hypointense lesions representing severe axonal loss and tissue destruction (marker of poor prognosis)
- Spinal cord atrophy: Correlates with disability progression [9]
Clinical Phenotypes and Disease Course
Exam Detail: MS is classified into four clinical phenotypes based on disease trajectory: [1,2,4]
1. Clinically Isolated Syndrome (CIS)
- Definition: First clinical episode suggestive of CNS demyelination (e.g., optic neuritis, transverse myelitis, brainstem syndrome) without prior history
- Risk of Conversion to MS: 60-80% within 10-20 years if MRI shows asymptomatic lesions fulfilling McDonald criteria for dissemination in space [4]
- Management: High-risk CIS (MRI evidence of dissemination) may warrant early DMT initiation to delay second clinical event
2. Relapsing-Remitting MS (RRMS)
- Definition: Discrete attacks (relapses) with complete or partial recovery, followed by periods of clinical stability (remission)
- Frequency: 85% of MS cases at onset [1,2]
- Relapse Characteristics:
- Acute or subacute onset of new neurological symptoms lasting ≥24 hours
- Attacks separated by ≥30 days without new symptoms (to distinguish from fluctuations within a single relapse)
- Relapses may spontaneously remit over weeks to months
- Relapse Rate: Typically 0.5-1.5 relapses per year untreated; reduced by 30-70% with DMTs [6,7]
- Pathology: Predominantly inflammatory, with active lesions and BBB breakdown
3. Secondary Progressive MS (SPMS)
- Definition: Gradual neurological deterioration independent of relapses, following an initial RRMS phase
- Conversion: Historically 50% of RRMS patients transitioned to SPMS within 10 years; modern DMTs have significantly delayed this transition [1,2]
- Clinical Features:
- Progressive worsening over ≥6-12 months
- Relapses may or may not continue superimposed on progression
- Pathology: Shift from inflammation to neurodegenerative mechanisms; "smoldering inflammation" behind intact BBB, mitochondrial dysfunction, chronic axonal loss [3]
- Prognosis: DMT efficacy is reduced; siponimod and cladribine show modest benefit in active SPMS [14]
4. Primary Progressive MS (PPMS)
- Definition: Gradual neurological decline from onset without discrete relapses
- Frequency: 10-15% of MS cases [1,2]
- Clinical Features:
- Insidious onset (typically age > 40 years)
- Progressive myelopathy predominates (spastic paraparesis, bladder dysfunction)
- "MRI: Fewer brain lesions, more spinal cord involvement and atrophy"
- OCBs positive in ~80% (lower than RRMS)
- Pathology: Degenerative mechanisms predominate from onset; less inflammation [3]
- Prognosis: Poorer than RRMS; ocrelizumab is the only approved DMT for PPMS, reducing disability progression by ~24% [15]
Pregnancy and MS
Effects of Pregnancy on MS:
- Relapse Rate Reduction: Relapses decrease by ~70% during the third trimester (immunomodulatory effect of pregnancy with Th2 shift) [16]
- Postpartum Rebound: Increased relapse risk in the first 3-6 months postpartum (return to pre-pregnancy relapse rate or higher) [16]
- Long-Term Disability: Pregnancy does not worsen long-term disability outcomes [16]
DMT Considerations in Pregnancy:
- Safe: Glatiramer acetate, interferon-beta (may be continued if needed)
- High Risk/Contraindicated: Fingolimod, teriflunomide (teratogenic), cladribine, alemtuzumab
- Management Strategy: Consider treatment interruption during pregnancy for low-activity MS; resume postpartum. High-activity MS may require continuation of interferon-beta/glatiramer or use of IVIG/rituximab [16]
4. Clinical Presentation
Symptoms and Syndromes
MS symptoms vary widely based on lesion location. Clinical presentations may be monophasic (CIS), relapsing-remitting, or progressive.
Exam Detail: 1. Optic Neuritis (20-30% of first presentations)
- Symptoms:
- Subacute, unilateral painful visual loss over hours to days
- Pain exacerbated by eye movements (retro-orbital pain)
- Desaturation of color vision (especially red desaturation)
- Central scotoma (most common field defect)
- Signs:
- Relative afferent pupillary defect (RAPD) on affected side
- "Decreased visual acuity (range: mild blurring to complete blindness)"
- "Fundoscopy: Initially normal (retrobulbar neuritis) or optic disc swelling (papillitis); later optic atrophy (temporal pallor)"
- Recovery: 90% recover to ≥20/40 vision within 6 months
- Predictive Value: 50% develop MS within 15 years; risk increases to 70-80% if MRI shows asymptomatic brain lesions [1,8]
2. Sensory Syndromes (40-50% of presentations)
- Paresthesias/Dysesthesias: Numbness, tingling, "pins and needles," burning sensations
- "MS Hug": Tight band-like sensation around torso (indicates thoracic cord lesion)
- Posterior Column Dysfunction: Reduced proprioception, vibration sense (Romberg's sign positive); sensory ataxia
- Lhermitte's Sign: Pathognomonic for cervical posterior column demyelination [1,8]
3. Motor Syndromes
- Acute Myelopathy: Monoparesis, paraparesis, or quadriparesis depending on cord level
- Chronic Spasticity: Progressive stiffness, spasms, clonus (common in progressive MS)
- Weakness: Upper motor neuron pattern (pyramidal weakness affecting extensors of upper limbs, flexors of lower limbs)
- Fatigue: Disabling, disproportionate to exertion; affects 75-90% of MS patients (multifactorial: CNS inflammation, medication side effects, sleep disturbance, depression) [1,8]
4. Brainstem and Cerebellar Syndromes
- Internuclear Ophthalmoplegia (INO): Failure of ipsilateral adduction with contralateral abducting nystagmus (MLF lesion); bilateral INO is highly suggestive of MS in young adults [8]
- Other Cranial Neuropathies:
- Diplopia (abducens nerve palsy, INO)
- Facial numbness or pain (trigeminal neuralgia in young patient suggests MS)
- Facial weakness (Bell's palsy-like presentation)
- Vertigo (vestibular nucleus involvement)
- Dysphagia, dysarthria (bulbar involvement)
- Cerebellar Signs:
- Intention tremor (exacerbated by goal-directed movement)
- Dysmetria, dysdiadochokinesia
- Ataxic gait, truncal ataxia
- Scanning (staccato) speech
- Nystagmus (vestibular or cerebellar) [1,8]
5. Bladder, Bowel, and Sexual Dysfunction (50-80% over disease course)
- Bladder Dysfunction:
- Urinary urgency, frequency, urge incontinence (detrusor hyperreflexia)
- Urinary retention (detrusor-sphincter dyssynergia)
- Increased risk of urinary tract infections
- Bowel Dysfunction: Constipation (common), fecal urgency/incontinence (less common)
- Sexual Dysfunction: Erectile dysfunction, reduced libido, anorgasmia (multifactorial: neurological, fatigue, psychological) [1]
6. Cognitive and Neuropsychiatric Symptoms (40-65%)
- Cognitive Impairment:
- Information processing speed (most commonly affected)
- Working memory, attention, executive function
- Verbal fluency
- Visuospatial skills (less commonly affected)
- Depression: Affects ~50% (highest risk in the first year after diagnosis); increased suicide risk
- Euphoria/Pseudobulbar Affect: Pathological laughing/crying (less common, seen in advanced disease) [1,8]
7. Paroxysmal Symptoms
- Trigeminal Neuralgia: Severe facial pain in trigeminal distribution; MS should be considered in patients less than 40 years with trigeminal neuralgia
- Tonic Spasms: Brief (seconds to minutes), stereotyped painful muscle contractions (often limbs); may be provoked by hyperventilation or sensory stimuli
- Lhermitte's Sign: Provoked by neck flexion [1,8]
Uhthoff's Phenomenon
Clinical Significance: Transient worsening of pre-existing MS symptoms (or appearance of new symptoms) with increased core body temperature (exercise, hot bath, fever, hot weather).
Mechanism: Temperature-dependent conduction block in demyelinated axons; even a 0.5°C increase can impair saltatory conduction. [1]
Management: Cooling strategies (air conditioning, cooling vests), avoidance of heat exposure; does not represent a true relapse and resolves with temperature normalization.
5. Differential Diagnosis
Exam Detail: | Condition | Clinical Clues | Distinguishing Investigations | Key Differences | |:--------------|:-------------------|:----------------------------------|:--------------------| | Neuromyelitis Optica Spectrum Disorder (NMOSD) | Severe bilateral optic neuritis, LETM (≥3 vertebral segments), area postrema syndrome (hiccups, nausea) | AQP4-IgG antibody (70-80% seropositive), MRI brain often normal or atypical (hypothalamus, brainstem), spinal MRI shows LETM with central cord involvement | Distinct pathophysiology (astrocytopathy); MS DMTs may worsen NMOSD [10] | | MOG-Antibody Disease | Recurrent optic neuritis, myelitis, ADEM-like presentations; often pediatric onset | MOG-IgG antibody, MRI shows fluffy, large lesions; good response to steroids | Monophasic or relapsing; better prognosis than MS | | Acute Disseminated Encephalomyelitis (ADEM) | Monophasic encephalopathy (altered consciousness, seizures), multifocal deficits; follows infection/vaccination; pediatric | MRI: Large, bilateral, asymmetric lesions involving grey and white matter, thalamus, basal ganglia | Monophasic (vs. relapsing MS); encephalopathy is key feature [1] | | CNS Vasculitis (Primary or Secondary) | Systemic features (fever, weight loss, rash, arthritis), headache, stroke-like episodes, seizures | Elevated ESR/CRP, autoantibodies (ANA, ANCA), cerebral angiography (beading), brain/meningeal biopsy | Multi-organ involvement; angiographic abnormalities | | Neurosarcoidosis | Systemic sarcoidosis (lungs, skin, eyes), cranial neuropathies (especially facial nerve), meningeal enhancement | Elevated ACE, CXR (hilar lymphadenopathy), biopsy (non-caseating granulomas), MRI shows leptomeningeal enhancement | Systemic features; OCBs often absent | | Subacute Combined Degeneration (B12 Deficiency) | Dorsal and lateral column signs, peripheral neuropathy, macrocytic anemia, glossitis, neuropsychiatric symptoms | Low serum B12, elevated methylmalonic acid/homocysteine, MRI shows T2 hyperintensity in posterior columns | Spinal MRI: Inverted "V" sign; reversible if treated early | | CNS Lupus (SLE) | Multisystem features (rash, arthritis, serositis, renal), seizures, psychosis, strokes | Positive ANA, anti-dsDNA, low complement (C3, C4), MRI shows multiple small lesions (less periventricular than MS) | Systemic lupus features; CSF may show elevated protein without OCBs | | Lyme Neuroborreliosis | History of tick bite, erythema migrans rash, subacute meningoencephalitis, cranial neuropathies (especially facial), radiculopathy | Lyme serology (ELISA, Western blot), CSF shows lymphocytic pleocytosis, intrathecal Lyme antibody production | Endemic areas (North America, Europe); responds to antibiotics | | HTLV-1 Associated Myelopathy (HAM/TSP) | Progressive spastic paraparesis, bladder dysfunction, minimal sensory/upper limb involvement; endemic areas (Caribbean, Japan, Africa) | HTLV-1 serology, CSF antibodies, MRI shows spinal cord atrophy | Predominantly spinal; brain lesions rare | | Mitochondrial Disease (e.g., LHON, MELAS) | Bilateral optic neuropathy (LHON), stroke-like episodes (MELAS), family history (maternal inheritance), systemic features (diabetes, deafness, myopathy) | Serum lactate, mitochondrial DNA mutation analysis, muscle biopsy | Multisystem involvement; atypical MRI lesions (cortical, not periventricular) | | Cerebral Autosomal Dominant Arteriopathy with Subcortical Infarcts and Leukoencephalopathy (CADASIL) | Family history, recurrent strokes, migraines, subcortical dementia, anterior temporal lobe lesions on MRI | NOTCH3 gene mutation, skin biopsy (granular osmiophilic material in vessel walls) | Anterior temporal lobe involvement; family history of strokes/dementia |
6. Clinical Examination
Structured Neurological Examination for Suspected MS
Exam Detail: General Inspection:
- Gait (ataxic, spastic, or mixed)
- Mobility aids (cane, walker, wheelchair)
- Cognitive assessment (conversational fluency, attention)
Cranial Nerve Examination:
-
CN II (Optic Nerve):
- Visual Acuity: Snellen chart (reduced acuity suggests optic neuritis)
- Color Vision: Ishihara plates (red desaturation in optic neuritis)
- Visual Fields: Confrontation testing (central scotoma in optic neuritis)
- Pupils: Relative Afferent Pupillary Defect (RAPD) - swing light test (afferent defect on affected side in optic neuritis)
- Fundoscopy: Optic disc pallor/atrophy (chronic optic neuritis), papilledema (rare)
-
CN III, IV, VI (Oculomotor Function):
- Extraocular Movements: Assess for Internuclear Ophthalmoplegia (INO)
- Lesion in medial longitudinal fasciculus (MLF)
- Ipsilateral failure of adduction on lateral gaze
- Contralateral nystagmus of abducting eye
- Bilateral INO is highly suggestive of MS in young adults [8]
- Nystagmus: Horizontal, vertical, or rotatory (cerebellar/vestibular lesions)
- Extraocular Movements: Assess for Internuclear Ophthalmoplegia (INO)
-
CN V (Trigeminal):
- Facial sensation (light touch, pinprick in three divisions)
- Jaw jerk reflex (brisk in upper motor neuron lesion affecting corticobulbar tracts)
- Trigeminal neuralgia in young patient (less than 50 years) raises suspicion for MS
-
CN VII (Facial):
- Facial symmetry, eye closure, smile (unilateral facial weakness may occur in brainstem lesions)
-
CN VIII (Vestibulocochlear):
- Hearing (Weber and Rinne tests)
- Vestibular function (Dix-Hallpike maneuver if vertigo)
-
CN IX, X, XII (Bulbar):
- Dysarthria, dysphagia (ask patient to repeat "baby hippopotamus")
- Tongue deviation, fasciculations
Upper Limb Examination:
- Tone: Spasticity (velocity-dependent increased tone; "clasp-knife" phenomenon)
- Power: MRC grading; pyramidal pattern of weakness (extensors weaker than flexors)
- Reflexes: Brisk or pathologically brisk (hyperreflexia); clonus at wrist (rare)
- Coordination:
- Finger-nose test (intention tremor, dysmetria - cerebellar lesion)
- Dysdiadochokinesia (rapid alternating movements - cerebellar lesion)
- Sensation:
- Light touch, pinprick (spinothalamic)
- "Vibration (128 Hz tuning fork at bony prominences: wrist, sternum)"
- "Proprioception (joint position sense: index finger)"
Lower Limb Examination:
- Gait:
- Spastic gait (circumduction, scissoring)
- Ataxic gait (wide-based, unsteady)
- Foot drop (steppage gait from common peroneal nerve or pyramidal lesion)
- Tone: Spasticity, clonus at ankle (sustained ankle clonus indicates upper motor neuron lesion)
- Power: MRC grading; pyramidal pattern (hip flexors, ankle dorsiflexors weaker than extensors/plantar flexors)
- Reflexes: Brisk knee and ankle jerks; upgoing plantar response (Babinski sign) (upper motor neuron lesion)
- Coordination: Heel-shin test (intention tremor, dysmetria)
- Sensation:
- Establish sensory level (if transverse myelitis suspected)
- "Vibration: Test at sternum (normal) vs. ankles (reduced suggests posterior column lesion)"
- "Romberg's test: Positive if loss of proprioception (posterior column) or vestibular dysfunction"
Special Tests:
- Lhermitte's Sign: Ask patient to flex neck; positive if electric shock sensation down spine (cervical cord lesion)
- Romberg's Test: Stand with feet together, eyes closed; sway or fall indicates posterior column or vestibular dysfunction
Expanded Disability Status Scale (EDSS)
The EDSS is the most widely used disability scale in MS, ranging from 0 (normal) to 10 (death due to MS). It assesses eight functional systems (pyramidal, cerebellar, brainstem, sensory, bowel/bladder, visual, cerebral, other) and ambulation.
Key EDSS Milestones:
- 0: Normal neurological examination
- 2.0: Minimal disability in one functional system
- 4.0: Ambulatory without aid; able to walk ~500 meters without rest
- 6.0: Unilateral assistance (cane, crutch) required to walk ~100 meters
- 6.5: Bilateral assistance required
- 7.0: Wheelchair-bound; unable to walk > 5 meters
- 8.0: Restricted to bed but can use arms
- 10: Death due to MS
Clinical Significance: EDSS is used as a primary outcome in clinical trials; DMT efficacy is measured by reduction in sustained EDSS progression (e.g., ≥1.0 point increase sustained for ≥6 months).
7. Investigations
MRI Brain and Spinal Cord (Gold Standard for Diagnosis)
Exam Detail: MRI is the most sensitive and specific imaging modality for detecting MS lesions. [4,5,9]
Standard Protocol:
- Brain MRI: Axial T2-weighted, FLAIR, T1-weighted pre- and post-gadolinium contrast, diffusion-weighted imaging (DWI)
- Spinal Cord MRI: Sagittal and axial T2-weighted, T1-weighted post-gadolinium (if brain MRI inconclusive or spinal symptoms present)
T2/FLAIR Sequences (Detect Lesions):
- Hyperintense Lesions (bright on T2/FLAIR):
- Periventricular: "Dawson's Fingers"
- ovoid lesions perpendicular to lateral ventricles (following perivenular distribution)
- "Juxtacortical: Lesions abutting the cortex (grey-white matter junction); U-fibers typically spared (differentiates from ADEM)"
- "Infratentorial: Brainstem (pons, medulla, midbrain), cerebellar peduncles, middle cerebellar peduncles"
- "Spinal Cord: Short lesions (≤2 vertebral segments), peripheral/lateral location, asymmetric [9]"
T1 Post-Gadolinium Sequences (Detect Active Inflammation):
- Enhancing Lesions: Indicate active inflammation and BBB breakdown
- Enhancement persists for ~4-6 weeks after lesion formation
- Presence of enhancing lesions indicates active disease (useful for treatment decisions)
- "Black Holes" (T1 Hypointensities): Represent severe axonal loss and tissue destruction; marker of poor prognosis [9]
Diagnostic Criteria for MRI Lesions (McDonald Criteria 2017): [4,5]
Dissemination in Space (DIS): Requires ≥1 T2 lesion in at least 2 of 4 CNS regions:
- Periventricular
- Cortical or juxtacortical
- Infratentorial
- Spinal cord
Dissemination in Time (DIT): Demonstrated by one of the following:
- Simultaneous presence of gadolinium-enhancing and non-enhancing lesions at any time
- New T2 or gadolinium-enhancing lesion on follow-up MRI (compared to baseline scan at any time)
- Presence of CSF-specific oligoclonal bands (OCBs) can substitute for DIT
Implications:
- A single MRI scan showing both enhancing and non-enhancing lesions can fulfill DIT criteria, allowing diagnosis after a single clinical event (CIS).
- OCBs allow diagnosis without needing serial MRI scans.
Lumbar Puncture and Cerebrospinal Fluid (CSF) Analysis
Exam Detail: Indications:
- Diagnostic uncertainty
- Atypical presentations (e.g., older age, progressive from onset)
- Rule out alternative diagnoses (infection, malignancy)
- Fulfill McDonald criteria (OCBs substitute for DIT) [4,5]
CSF Findings in MS:
| Parameter | Finding | Interpretation |
|---|---|---|
| Oligoclonal Bands (OCBs) | Present in CSF but absent in serum | Intrathecal IgG synthesis; positive in ~95% of RRMS, ~80% of PPMS. Pathognomonic for CNS inflammation (not specific to MS; also seen in CNS infections, sarcoidosis). [1,4] |
| IgG Index | Elevated (> 0.7) | Indicates intrathecal IgG synthesis (alternative to OCBs) |
| Cell Count | Normal (less than 5 cells/μL) or mild lymphocytic pleocytosis (5-50 cells/μL) | Pleocytosis > 50 cells/μL suggests alternative diagnosis (NMOSD, infection, malignancy) |
| Protein | Normal or mildly elevated (less than 1 g/L) | Marked elevation suggests alternative diagnosis (Guillain-Barré, CIDP, malignancy) |
| Glucose | Normal | Low glucose suggests infection (bacterial/TB meningitis) |
CSF-Specific Oligoclonal Bands (OCBs):
- Detected by isoelectric focusing (comparing CSF and serum samples run in parallel)
- Interpretation:
- "Type 1: No bands (normal)"
- "Type 2: OCBs in CSF only (MS and other CNS inflammatory conditions)"
- "Type 3: OCBs in CSF and serum (systemic immune response)"
- "Type 4: Identical bands in CSF and serum (systemic disease, not MS)"
- "Type 5: Monoclonal bands (paraproteinemia)"
- Sensitivity: 95% in RRMS; specificity ~90% for CNS inflammatory disease (not MS-specific)
Evoked Potentials
Exam Detail: Evoked potentials detect subclinical lesions and can support the diagnosis of dissemination in space.
1. Visual Evoked Potentials (VEP):
- Indication: Suspected optic neuritis (past or present); evaluate visual pathway integrity
- Technique: Patient views alternating checkerboard pattern; EEG electrodes record visual cortex response
- Normal: P100 wave latency ~100 ms
- MS Finding: Delayed P100 latency (> 115 ms) indicates slowed conduction due to demyelination in optic nerve
- Interpretation: Prolonged VEP latency in asymptomatic eye suggests prior subclinical optic neuritis (supports dissemination in space)
2. Somatosensory Evoked Potentials (SSEP):
- Indication: Assess dorsal column/sensory pathway integrity
- Technique: Electrical stimulation of median or tibial nerve; record response at spinal cord and cortex
- MS Finding: Prolonged central conduction time (from spinal cord to cortex)
3. Brainstem Auditory Evoked Potentials (BAEP):
- Indication: Assess brainstem auditory pathways
- Technique: Auditory clicks delivered; record brainstem response
- MS Finding: Prolonged interpeak latencies
Current Role: Evoked potentials are less commonly used in the era of high-resolution MRI but may be helpful when MRI is contraindicated or equivocal.
Diagnostic Testing to Exclude Mimics
Exam Detail: Essential Tests in Suspected MS:
- AQP4-IgG (Aquaporin-4 Antibody): Rule out NMOSD
- MOG-IgG (Myelin Oligodendrocyte Glycoprotein Antibody): Rule out MOG-antibody disease
- Serum Vitamin B12 and Methylmalonic Acid: Rule out subacute combined degeneration
- Antinuclear Antibody (ANA), Anti-dsDNA, Complement (C3, C4): Rule out SLE
- ACE Level, CXR: Rule out sarcoidosis
- Lyme Serology (if endemic area or history of tick exposure): Rule out Lyme neuroborreliosis
- HTLV-1 Serology (if endemic area or progressive myelopathy): Rule out HAM/TSP
- HIV Test: Opportunistic infections can mimic MS
- Thyroid Function Tests: Thyroid disease is common in MS (comorbidity and DMT side effect with alemtuzumab)
8. Diagnosis: McDonald Criteria (2017 Revision)
Exam Detail: The McDonald Criteria provide standardized diagnostic criteria for MS, allowing earlier diagnosis and treatment initiation. [4,5]
Core Principles:
- Demonstrate Dissemination in Space (DIS): Lesions in multiple CNS locations
- Demonstrate Dissemination in Time (DIT): Lesions occurring at different timepoints
- Exclude alternative diagnoses ("no better explanation")
Criteria for MS Diagnosis After ≥2 Clinical Attacks:
| Clinical Attacks | Objective Lesions | Additional Requirements |
|---|---|---|
| ≥2 | ≥2 | None (clinical evidence sufficient) |
| ≥2 | 1 | DIS demonstrated by: (1) Additional clinical attack implicating different site, OR (2) MRI showing DIS |
| 1 | ≥2 | DIT demonstrated by: (1) Additional clinical attack, OR (2) MRI showing DIT, OR (3) CSF OCBs |
| 1 (CIS) | 1 | DIS and DIT demonstrated by MRI and/or CSF OCBs |
MRI Criteria for Dissemination in Space (DIS):
≥1 T2 lesion in at least 2 of 4 regions:
- Periventricular
- Cortical or juxtacortical
- Infratentorial
- Spinal cord
Note: Symptomatic lesions can be counted for DIS in patients with brainstem or spinal cord syndromes.
MRI Criteria for Dissemination in Time (DIT):
Demonstrated by one of the following:
- Simultaneous presence of gadolinium-enhancing and non-enhancing lesions on a single MRI (at any time)
- New T2 or gadolinium-enhancing lesion on follow-up MRI (compared to baseline, regardless of timing)
- CSF-specific oligoclonal bands (OCBs) can substitute for DIT
Special Scenarios:
Primary Progressive MS (PPMS) Diagnosis:
Requires 1 year of disability progression (retrospective or prospective) plus 2 of 3 criteria:
- DIS in brain: ≥1 T2 lesion in periventricular, cortical/juxtacortical, or infratentorial regions
- DIS in spinal cord: ≥2 T2 lesions
- CSF-specific OCBs
2024 Revision Update (McDonald Criteria 2024):
The 2024 revision (recently published) includes: [5]
- Optic nerve lesions can now count toward DIS (recognition of optic neuritis as a CNS lesion)
- Central vein sign and paramagnetic rim lesions recognized as highly specific imaging biomarkers (requires specialized MRI sequences)
- Kappa free light chains may be used as an alternative to OCBs
9. Management
Management Principles and Algorithm
Exam Detail: Goals of MS Management:
- Acute Relapse Treatment: Shorten duration and severity of relapses
- Disease Modification: Reduce relapse rate, MRI activity, and disability progression
- Symptomatic Management: Improve quality of life
- Multidisciplinary Support: Physical therapy, occupational therapy, psychological support, continence care
Algorithm:
[SUSPECTED MS]
↓
MRI Brain/Spine + CSF Analysis
↓
McDonald Criteria Fulfilled?
┌──────────┴──────────┐
NO YES
│ │
[Clinically Isolated [MS DIAGNOSIS]
Syndrome (CIS)] ↓
│ Phenotype Classification
High-risk MRI? ┌─────────┴─────────┐
(Asymptomatic RRMS PPMS/SPMS
lesions) │ │
↓ ↓ ↓
Consider DMT [ACUTE RELAPSE?] Ocrelizumab (PPMS)
vs. Monitor │ Siponimod (SPMS)
┌──────┴──────┐
NO YES (Disabling)
│ │
Supportive Methylprednisolone
Care 1g IV x3 days
(or 500mg PO x5)
│
↓
[DISEASE-MODIFYING THERAPY (DMT)]
Assess Disease Activity:
- Relapse frequency (ARR)
- MRI lesion burden/new lesions
- Disability progression (EDSS)
│
┌──────────────────┼──────────────────┐
Moderate High Efficacy Very High
Efficacy Needed Efficacy/Aggressive
(ARR ~0.5-1) (ARR > 1, ≥2 MRI) (Severe relapses, high burden)
│ │ │
Inj: Beta-IFN, Oral: Fingolimod, Infusion: Natalizumab,
Glatiramer Cladribine Ocrelizumab, Alemtuzumab
Oral: Teriflunomide,
Dimethyl fumarate
↓
[MONITORING & OPTIMIZATION]
Serial MRI (annually or PRN)
Clinical assessment (relapses, EDSS)
Safety monitoring (CBC, LFTs, JCV)
↓
Treatment failure? (breakthrough disease)
┌──────┴──────┐
NO YES
│ │
Continue Escalate/Switch DMT
DMT Consider higher efficacy agent
or alternative mechanism
1. Acute Relapse Management
Exam Detail: Definition of Relapse:
- New or worsening neurological symptoms lasting ≥24 hours
- Separated from previous relapse by ≥30 days
- Not explained by infection, fever, or other confounders (pseudorelapse)
Indications for Treatment:
- Disabling symptoms affecting function (e.g., severe visual loss, weakness affecting mobility, disabling ataxia)
- Not indicated for mild sensory relapses without functional impairment
Corticosteroid Therapy: [1,2]
Standard Regimen:
- Methylprednisolone 1g IV daily for 3 days (preferred)
- Alternative: Methylprednisolone 500 mg orally daily for 5 days (non-inferior efficacy, more convenient) [17]
Mechanism: Anti-inflammatory; stabilizes BBB, reduces edema, suppresses immune response
Evidence:
- Shortens duration of relapse and speeds recovery
- No effect on long-term disability or relapse rate (does not alter disease course) [17]
Oral Taper: Not routinely required; some clinicians use short oral prednisolone taper (e.g., 1 week) to reduce rebound symptoms
Gastroprotection: Proton pump inhibitor (PPI) during steroid course
Precautions:
- Exclude infection (especially UTI in patients with bladder dysfunction) before starting steroids
- Manage hyperglycemia in diabetic patients
- Monitor for steroid-induced psychosis (rare)
Plasma Exchange (PLEX) or Immunoadsorption:
Indication: Severe, disabling relapses not responding to corticosteroids (steroid-refractory relapse)
Protocol:
- 5-7 plasma exchanges over 10-14 days
- Evidence of benefit in fulminant demyelination and steroid-refractory relapses [1]
2. Disease-Modifying Therapies (DMTs)
Exam Detail: DMTs aim to reduce relapse rate, MRI activity (new/enhancing lesions), and disability progression. The concept of "No Evidence of Disease Activity" (NEDA-3) is used as a composite endpoint in clinical trials: [6,7]
- No relapses
- No MRI activity (no new/enlarging T2 or gadolinium-enhancing lesions)
- No confirmed disability progression (EDSS)
Principles of DMT Selection:
- Disease Activity: Higher disease activity (frequent relapses, active MRI lesions) favors high-efficacy DMTs
- Patient Factors: Age, comorbidities, pregnancy planning, tolerability, route of administration preference
- Safety Profile: Risk-benefit assessment (e.g., JC virus status for natalizumab, cardiac risk for fingolimod)
- Early Treatment: Evidence supports early DMT initiation to prevent irreversible disability [6,7]
Escalation vs. Induction Strategies:
- Escalation: Start with moderate-efficacy DMT; escalate to high-efficacy if inadequate response
- Induction: Start with high-efficacy DMT upfront in aggressive disease
MODERATE-EFFICACY DMTs (Reduce ARR by 30-50%)
1. Beta-Interferons (IFN-β)
- Examples: Interferon beta-1a (Avonex, Rebif), Interferon beta-1b (Betaseron, Extavia)
- Mechanism: Immunomodulatory (reduce T-cell activation, stabilize BBB)
- Route: Subcutaneous (SC) or intramuscular (IM) injection
- Efficacy: Reduce ARR by ~30%; reduce MRI activity
- Side Effects:
- Flu-like symptoms (fever, myalgia, headache) - often improve with time; premedicate with paracetamol/ibuprofen
- Injection site reactions (erythema, lipoatrophy)
- Hepatotoxicity (monitor LFTs)
- Leukopenia, thrombocytopenia (monitor CBC)
- Depression (relative contraindication in severe depression)
- Pregnancy: Considered safe; can be continued if needed [16]
- Monitoring: CBC, LFTs at baseline, 1 month, then every 3-6 months
2. Glatiramer Acetate (Copaxone)
- Mechanism: Immunomodulatory (synthetic amino acid copolymer; antigen decoy, induces regulatory T-cells)
- Route: Subcutaneous injection (daily or three times weekly)
- Efficacy: Reduce ARR by ~30%
- Side Effects:
- Injection site reactions (pain, erythema, lipoatrophy)
- Immediate post-injection reaction (flushing, chest tightness, palpitations, dyspnea) - benign, self-limiting
- Pregnancy: Considered safe [16]
- Monitoring: Minimal monitoring required
3. Teriflunomide (Aubagio)
- Mechanism: Inhibits dihydroorotate dehydrogenase (blocks pyrimidine synthesis, reduces lymphocyte proliferation)
- Route: Oral (daily)
- Efficacy: Reduce ARR by ~30-35%
- Side Effects:
- Diarrhea, nausea, alopecia (mild, reversible)
- Hepatotoxicity (monitor LFTs)
- Hypertension
- Teratogenic (Category X in pregnancy; requires contraception; long elimination half-life necessitates accelerated elimination protocol if pregnancy planned)
- Pregnancy: Contraindicated; washout required [16]
- Monitoring: CBC, LFTs monthly for 6 months, then every 6 months; blood pressure
4. Dimethyl Fumarate (Tecfidera)
- Mechanism: Activates Nrf2 pathway (antioxidant, anti-inflammatory)
- Route: Oral (twice daily)
- Efficacy: Reduce ARR by ~50%
- Side Effects:
- Flushing, gastrointestinal upset (diarrhea, nausea, abdominal pain) - often transient; take with food
- Lymphopenia (monitor absolute lymphocyte count; discontinue if less than 0.5 x 10⁹/L or sustained less than 0.8 x 10⁹/L)
- "Rare: PML (case reports in setting of prolonged severe lymphopenia)"
- Pregnancy: Limited data; avoid if possible
- Monitoring: CBC at baseline, every 3 months for 1 year, then every 6-12 months
HIGH-EFFICACY DMTs (Reduce ARR by 50-70%)
5. Fingolimod (Gilenya)
- Mechanism: Sphingosine-1-phosphate (S1P) receptor modulator; sequesters lymphocytes in lymph nodes (prevents CNS infiltration)
- Route: Oral (daily)
- Efficacy: Reduce ARR by ~50-55%; reduce disability progression
- Side Effects:
- Bradycardia and AV block (first-dose effect) - requires 6-hour cardiac monitoring with first dose
- Macular edema (monitor ophthalmology at baseline, 3-4 months)
- Lymphopenia, increased infection risk (VZV reactivation, cryptococcal meningitis - check VZV serology; vaccinate if seronegative before starting)
- PML (rare)
- Hepatotoxicity (monitor LFTs)
- Teratogenic
- Pregnancy: Contraindicated; 2-month washout required [16]
- Monitoring: Cardiac monitoring (ECG, telemetry) for 6 hours after first dose; ophthalmology at 3-4 months; CBC, LFTs; VZV serology
6. Cladribine (Mavenclad)
- Mechanism: Purine analog; selectively depletes lymphocytes (B and T cells); immune reconstitution therapy
- Route: Oral (short course: 2 treatment weeks in year 1, 2 treatment weeks in year 2; then no treatment - "treatment-free" period)
- Efficacy: Reduce ARR by ~55-60%; reduce disability progression; sustained benefit for ≥4 years after treatment [18]
- Side Effects:
- Lymphopenia (expected; nadirs at 2-3 months, recovers gradually)
- Increased infection risk (monitor for herpes zoster, opportunistic infections)
- Hepatotoxicity
- Teratogenic; contraception required for 6 months after last dose
- Theoretical malignancy risk (long-term data reassuring)
- Pregnancy: Contraindicated [16]
- Monitoring: CBC monthly for 6 months post-treatment, then every 3 months until lymphocyte recovery
7. Natalizumab (Tysabri)
- Mechanism: Monoclonal antibody against α4-integrin (VLA-4); blocks lymphocyte adhesion and migration across BBB
- Route: IV infusion (300 mg every 4 weeks)
- Efficacy: Reduce ARR by ~68%; reduce disability progression by ~42%; highly effective [19]
- Side Effects:
- "PML (Progressive Multifocal Leukoencephalopathy): JC virus reactivation; potentially fatal"
- Risk factors: JC virus antibody positive, duration > 2 years, prior immunosuppressant use
- Risk stratification: JC virus antibody-negative = ~0.1 per 1000; JC virus antibody-positive, > 2 years, prior immunosuppressant = ~10 per 1000 [11,12]
- Infusion reactions (headache, fatigue, urticaria)
- Increased infection risk
- "PML (Progressive Multifocal Leukoencephalopathy): JC virus reactivation; potentially fatal"
- PML Monitoring: Serial MRI (every 3-6 months), JC virus antibody index monitoring; educate patients on PML symptoms (cognitive decline, motor deficits, visual changes)
- Pregnancy: Discontinue before conception; limited data [16]
- Monitoring: JC virus antibody status every 6 months; MRI every 3-6 months
8. Ocrelizumab (Ocrevus)
- Mechanism: Monoclonal antibody against CD20 (B-cell depletion)
- Route: IV infusion (600 mg every 6 months)
- Efficacy: Reduce ARR by ~46-47% vs. interferon beta-1a; reduce disability progression; only DMT approved for PPMS (reduces disability progression by ~24% in PPMS) [15]
- Side Effects:
- Infusion reactions (pruritus, rash, urticaria, hypotension) - premedicate with methylprednisolone, antihistamine, paracetamol
- Increased infection risk (upper respiratory infections, urinary tract infections)
- "Rare: hepatitis B reactivation (screen HBV before starting)"
- Reduced immunoglobulin levels (monitor; consider Ig replacement if recurrent infections)
- Theoretical malignancy risk (particularly breast cancer signal in trials; requires ongoing surveillance)
- Pregnancy: Discontinue 6 months before conception; B-cell depletion in neonate if exposed in utero [16]
- Monitoring: Hepatitis B serology (HBsAg, anti-HBc, anti-HBs), immunoglobulins annually, CBC
9. Alemtuzumab (Lemtrada)
- Mechanism: Monoclonal antibody against CD52 (profound lymphocyte depletion, followed by immune reconstitution)
- Route: IV infusion (12 mg daily x5 days in year 1; 12 mg daily x3 days in year 2)
- Efficacy: Reduce ARR by ~55%; reduce disability progression; sustained benefit for ≥6-10 years [20]
- Side Effects:
- Infusion reactions (fever, rash, headache) - premedicate with corticosteroids, antihistamines, paracetamol
- "Secondary autoimmunity (most common and serious side effect):"
- Autoimmune thyroid disease (Graves', hypothyroidism) - 30-40%
- Immune thrombocytopenia (ITP) - 1-2% (can be severe/fatal)
- Goodpasture's syndrome (anti-GBM glomerulonephritis) - rare
- Infections (listeria, herpes zoster, tuberculosis reactivation)
- "Rare: stroke (immediate post-infusion)"
- Monitoring: Intensive monitoring required for ≥4 years post-treatment:
- Monthly CBC (ITP surveillance)
- Monthly urinalysis + serum creatinine (nephropathy surveillance)
- Thyroid function tests every 3 months
- Pregnancy: Contraindicated; 4-month washout required [16]
10. Ofatumumab (Kesimpta)
- Mechanism: Monoclonal antibody against CD20 (B-cell depletion)
- Route: Subcutaneous injection (20 mg every 4 weeks after loading)
- Efficacy: Reduce ARR by ~50-60% vs. teriflunomide; convenient SC administration (vs. IV ocrelizumab)
- Side Effects: Similar to ocrelizumab (infusion reactions, infections, reduced immunoglobulins)
THERAPIES FOR PROGRESSIVE MS
Siponimod (Mayzent):
- Indication: Secondary Progressive MS (SPMS) with active disease
- Mechanism: S1P receptor modulator (similar to fingolimod; S1P1 and S1P5 selectivity)
- Efficacy: Reduce disability progression by ~21% vs. placebo in active SPMS [14]
- Side Effects: Similar to fingolimod (bradycardia, macular edema, lymphopenia)
BTK Inhibitors (Emerging):
- Bruton's tyrosine kinase (BTK) inhibitors (e.g., tolebrutinib, fenebrutinib) show promise in progressive MS; trials ongoing
3. Symptomatic Management
Exam Detail: Spasticity:
- Pharmacological:
- "Baclofen (GABA-B agonist) 5-25 mg TDS (titrate gradually; side effects: sedation, weakness)"
- Tizanidine (α2-adrenergic agonist) 2-8 mg TDS
- Gabapentin 300-900 mg TDS
- Intrathecal baclofen pump (severe refractory spasticity)
- Botulinum toxin injections (focal spasticity)
- Non-pharmacological: Physiotherapy, stretching, hydrotherapy
Fatigue (affects 75-90%):
- Non-pharmacological (first-line): Energy conservation strategies, graded exercise, sleep hygiene, treat depression
- Pharmacological:
- Amantadine 100 mg BD (modest benefit)
- Modafinil 100-200 mg daily (off-label; variable efficacy)
Bladder Dysfunction:
- Detrusor Hyperreflexia (urgency, frequency):
- "Anticholinergics: Oxybutynin 2.5-5 mg TDS, Tolterodine 2-4 mg BD, Solifenacin 5-10 mg daily"
- "Beta-3 agonist: Mirabegron 25-50 mg daily"
- Detrusor-Sphincter Dyssynergia (incomplete emptying, retention):
- Intermittent self-catheterization (ISC)
- α-blockers (e.g., tamsulosin) if obstruction
- UTI Prophylaxis: If recurrent UTIs (cranberry, low-dose antibiotics)
Neuropathic Pain:
- First-line: Amitriptyline 10-75 mg nocte, Gabapentin 300-900 mg TDS, Pregabalin 75-300 mg BD
- Second-line: Duloxetine 60 mg daily, Carbamazepine 200-400 mg BD (especially for trigeminal neuralgia)
Trigeminal Neuralgia:
- First-line: Carbamazepine 200-400 mg BD (highly effective)
- Alternatives: Oxcarbazepine, Gabapentin, Pregabalin
- Surgical: Microvascular decompression (if refractory)
Tremor and Ataxia:
- Pharmacological: Limited efficacy (propranolol, primidone, gabapentin, clonazepam)
- Deep Brain Stimulation (DBS): Thalamic DBS for severe disabling tremor (selected cases)
- Occupational Therapy: Adaptive devices, weighted utensils
Depression and Anxiety:
- SSRIs: Sertraline 50-200 mg, Escitalopram 10-20 mg
- Cognitive Behavioral Therapy (CBT)
- Screen regularly: PHQ-9, GAD-7
Cognitive Impairment:
- Cognitive Rehabilitation: Memory strategies, attention training
- Pharmacological: Donepezil (off-label; limited evidence)
Walking Impairment:
- Fampridine (Dalfampridine) 10 mg BD (prolonged-release): Potassium channel blocker; improves walking speed in ~30-40% of patients (responders see ~25% improvement) [1]
- Physiotherapy, Mobility Aids: Canes, walkers, orthotics (AFO for foot drop)
Sexual Dysfunction:
- Erectile Dysfunction: Sildenafil, Tadalafil
- Female Sexual Dysfunction: Lubrication, counseling
- Address contributing factors: Fatigue, depression, relationship issues
4. Multidisciplinary Team Approach
Exam Detail: MS care requires coordinated multidisciplinary support:
- MS Specialist Nurse: Education, treatment coordination, relapse management, patient support
- Neurologist: Diagnosis, DMT initiation/monitoring, relapse treatment
- Physiotherapist: Exercise programs, gait training, spasticity management
- Occupational Therapist: Activities of daily living (ADL) assessment, adaptive equipment, home modifications
- Speech and Language Therapist: Dysphagia assessment, communication aids
- Continence Nurse/Urologist: Bladder/bowel management
- Neuropsychologist: Cognitive assessment, rehabilitation
- Psychiatrist/Psychologist: Depression, anxiety, adjustment disorder
- Social Worker: Benefits, disability support, caregiver support
- Dietitian: Nutritional counseling (obesity, dysphagia)
10. Complications and Prognosis
Complications
Exam Detail: | Complication | Frequency | Management | Prognosis | |:-----------------|:--------------|:---------------|:--------------| | Progressive Disability | 50% (historical RRMS to SPMS at 10-15 years; reduced with modern DMTs) | Early DMT, physiotherapy, mobility aids | Variable; EDSS ≥6.0 (requiring walking aid) in 25-30% at 20 years with treatment [1] | | Urosepsis | Common (bladder dysfunction in 50-80%) | Bladder management (ISC, anticholinergics), UTI prophylaxis | Preventable with proper bladder care | | Aspiration Pneumonia | Advanced disease | Dysphagia assessment, modified diet, PEG if needed | Leading cause of death in severe MS | | Falls and Fractures | High (ataxia, spasticity, impaired mobility) | Physiotherapy, home modifications, bone health (Vitamin D, bisphosphonates) | Preventable with multidisciplinary support | | Pressure Ulcers | Immobile patients | Pressure relief, skin care, nutrition | Preventable | | Deep Vein Thrombosis (DVT)/Pulmonary Embolism (PE) | Increased risk (immobility) | Early mobilization, compression stockings, anticoagulation prophylaxis if immobile | Preventable | | Osteoporosis | High (corticosteroid use, immobility, vitamin D deficiency) | Calcium, Vitamin D, bisphosphonates | Preventable | | Depression and Suicide | 50% depression; 2-fold increased suicide risk | Screening, SSRIs, CBT, psychiatric support | Treatable; early intervention critical | | Progressive Multifocal Leukoencephalopathy (PML) | Rare; natalizumab (highest risk), fingolimod, dimethyl fumarate | Immediate cessation of DMT, plasma exchange, immune reconstitution inflammatory syndrome (IRIS) management | 20-50% mortality; severe disability in survivors [11,12] | | Secondary Autoimmunity (Alemtuzumab) | Thyroid disease (30-40%), ITP (1-2%), Goodpasture's (less than 1%) | Intensive monitoring (monthly CBC, urinalysis, TFTs every 3 months for ≥4 years post-treatment); immunosuppression for ITP/Goodpasture | Thyroid disease manageable; ITP/Goodpasture can be severe [20] |
Prognosis
Exam Detail: Life Expectancy:
- Reduced by 7-10 years compared to general population (historical data)
- Leading causes of death: Infections (pneumonia, urosepsis), complications of immobility, suicide [1,2]
- Modern DMT era: Improved life expectancy expected (long-term data emerging)
Disability Outcomes:
Historical (Pre-DMT Era):
- 50% required walking aid (EDSS ≥6.0) at 15 years
- 50% transitioned to SPMS within 10 years of RRMS onset [1,2]
Modern (DMT Era):
- Early treatment reduces disability progression by 30-50%
- NEDA-3 (no relapses, no MRI activity, no disability progression) achievable in 40-60% of patients on high-efficacy DMTs at 2 years [6,7]
- Delayed transition to SPMS (estimated delay of 5-10 years with high-efficacy DMTs)
Prognostic Factors:
| Factor | Good Prognosis | Poor Prognosis |
|---|---|---|
| Sex | Female | Male |
| Age at Onset | Younger (less than 30 years) | Older (> 40 years) |
| Initial Symptoms | Sensory, optic neuritis | Motor, cerebellar, brainstem |
| Relapse Frequency | Low (less than 1 per year) | High (> 2 per year) |
| MRI Lesion Burden | Low lesion load | High lesion load (> 9 T2 lesions), "black holes" (T1 hypointensities) |
| Inter-relapse Interval | Long (> 1 year between first two relapses) | Short (less than 6 months) |
| Recovery from Relapses | Complete | Incomplete (residual deficits) |
| DMT Initiation | Early (within 1-2 years of diagnosis) | Delayed |
| CSF OCBs | Absent (uncertain significance) | Present (associated with confirmed MS diagnosis) |
11. Evidence & Guidelines
Guidelines
Exam Detail: 1. NICE NG127 (2022): Multiple Sclerosis in Adults: Management
- Recommends early DMT initiation for active RRMS
- Endorses shared decision-making for DMT selection
- Emphasizes multidisciplinary care and symptomatic management
- URL: https://www.nice.org.uk/guidance/ng127
2. Association of British Neurologists (ABN) Guidelines for MS (2022)
- Updated recommendations for DMT sequencing, monitoring, and switching
- PML risk stratification for natalizumab
- Pregnancy planning and DMT safety
3. European Academy of Neurology (EAN)/European Committee for Treatment and Research in Multiple Sclerosis (ECTRIMS) Guidelines
- Comprehensive European consensus on MS diagnosis and management
- Evidence-based DMT recommendations
4. American Academy of Neurology (AAN) Guidelines
- Evidence-based recommendations for DMT use in MS
Landmark Trials and Evidence
Exam Detail: 1. AFFIRM Trial (Natalizumab for RRMS)
- Design: Phase 3 RCT; natalizumab vs. placebo
- Result: Reduced ARR by 68%, disability progression by 42%, MRI lesions by 83%
- Citation: Polman CH, et al. N Engl J Med. 2006;354:899-910. [PMID: 16510744] [19]
2. OPERA I & II (Ocrelizumab for RRMS)
- Design: Phase 3 RCT; ocrelizumab vs. interferon beta-1a
- Result: Ocrelizumab superior; reduced ARR by 46-47%, disability progression by 40%, MRI activity by 95%
- Citation: Hauser SL, et al. N Engl J Med. 2017;376:221-234. [PMID: 28002688] [15]
3. ORATORIO (Ocrelizumab for PPMS)
- Design: Phase 3 RCT; ocrelizumab vs. placebo in PPMS
- Result: First DMT to show benefit in PPMS; reduced disability progression by 24% (first-ever positive trial in PPMS)
- Citation: Montalban X, et al. N Engl J Med. 2017;376:209-220. [PMID: 28002688] [15]
4. CLARITY (Cladribine for RRMS)
- Design: Phase 3 RCT; oral cladribine vs. placebo
- Result: Reduced ARR by 58%, disability progression by 33%, sustained benefit for ≥4 years
- Citation: Giovannoni G, et al. N Engl J Med. 2010;362:416-426. [PMID: 20089960] [18]
5. CARE-MS I & II (Alemtuzumab for RRMS)
- Design: Phase 3 RCT; alemtuzumab vs. interferon beta-1a
- Result: Alemtuzumab superior; reduced ARR by 55%, improved EDSS vs. baseline (unique among DMTs)
- Citation: Cohen JA, et al. Lancet. 2012;380:1819-1828. [PMID: 23122650] [20]
6. EXPAND (Siponimod for SPMS)
- Design: Phase 3 RCT; siponimod vs. placebo in SPMS
- Result: Reduced disability progression by 21% in SPMS with active disease
- Citation: Kappos L, et al. Lancet. 2018;391:1263-1273. [PMID: 29576505] [14]
7. McDonald Criteria 2017 Revision
- Landmark Consensus: Standardized MS diagnostic criteria; allows earlier diagnosis (after single clinical event if MRI/CSF criteria met)
- Citation: Thompson AJ, et al. Lancet Neurol. 2018;17:162-173. [PMID: 29275977] [4]
8. McDonald Criteria 2024 Revision
- Update: Includes optic nerve lesions, central vein sign, kappa free light chains
- Citation: Montalban X, et al. Lancet Neurol. 2025;24:119-131. [PMID: 40975101] [5]
12. Patient/Layperson Explanation
What is Multiple Sclerosis?
Multiple Sclerosis (MS) is a condition where your body's immune system mistakenly attacks the protective coating (called myelin) around the nerves in your brain and spinal cord. Think of myelin like the insulation around electrical wires - when it's damaged, the signals traveling along the nerves slow down or stop completely. This causes a wide variety of symptoms depending on which nerves are affected.
What Causes MS?
We don't know the exact cause, but MS is thought to result from a combination of genetic risk (having certain genes makes you more susceptible) and environmental triggers. Factors like low vitamin D (from less sunlight exposure), previous Epstein-Barr virus infection (the virus that causes glandular fever/mono), and smoking increase the risk.
What Are the Symptoms?
MS symptoms vary widely from person to person, but common symptoms include:
- Vision problems: Blurred vision, loss of color vision, or pain when moving the eyes
- Numbness or tingling: Often in the arms, legs, or face
- Weakness: Difficulty walking, muscle stiffness, or fatigue
- Balance and coordination problems: Dizziness, tremors, unsteady gait
- Bladder or bowel problems
- Cognitive changes: Memory difficulties, trouble concentrating
Symptoms may come and go (relapses and remissions) or gradually worsen over time.
How Is MS Diagnosed?
Your neurologist will use:
- MRI scans of your brain and spinal cord to look for areas of damage (lesions)
- Lumbar puncture (spinal tap) to analyze the fluid around your spinal cord for signs of inflammation
- Neurological examination to assess your vision, strength, coordination, and reflexes
What Are the Types of MS?
- Relapsing-Remitting MS (RRMS): Most common (85% at onset). You have flare-ups (relapses) of symptoms followed by periods of recovery. Between relapses, the disease is stable.
- Secondary Progressive MS (SPMS): After years of RRMS, some people develop gradual worsening of symptoms without clear relapses.
- Primary Progressive MS (PPMS): Symptoms gradually worsen from the start without relapses (10-15% of cases).
- Clinically Isolated Syndrome (CIS): A first episode of symptoms; may or may not develop into MS.
How Is MS Treated?
There is currently no cure for MS, but treatments can significantly slow the disease and manage symptoms:
1. Disease-Modifying Therapies (DMTs): These medications reduce the frequency of relapses, slow disability progression, and reduce new brain lesions on MRI. Options include injections, oral tablets, and intravenous infusions. Your neurologist will help you choose the best treatment based on your disease activity, side effects, and personal preferences.
2. Relapse Treatment: If you have a severe relapse (flare-up), high-dose steroids (methylprednisolone) can shorten the attack and speed recovery.
3. Symptom Management: Medications and therapies can help manage specific symptoms like muscle stiffness, pain, bladder problems, fatigue, and depression.
4. Lifestyle:
- Stay active: Regular exercise helps maintain strength, balance, and mood.
- Vitamin D: Many neurologists recommend vitamin D supplements (sunlight exposure is also beneficial).
- Quit smoking: Smoking worsens MS progression.
- Healthy diet: A balanced diet supports overall health.
Can I Have a Baby If I Have MS?
Yes! MS does not affect fertility, and pregnancy often improves MS symptoms (relapses are less frequent during pregnancy, especially in the third trimester). However:
- Some DMTs are not safe during pregnancy, so plan ahead with your neurologist to switch to safer options or pause treatment.
- Relapses may increase slightly in the first few months after delivery, but this can be managed.
Will I End Up in a Wheelchair?
Not necessarily. With modern treatments, many people with MS remain active and independent for decades. Early treatment with disease-modifying therapies significantly reduces the risk of disability. While some people do experience progressive disability, others have mild disease with minimal impact on daily life.
What Can I Do?
- Start treatment early: Evidence shows early DMT use prevents long-term disability.
- Stay informed: Understand your condition and work closely with your neurologist.
- Join support groups: Connecting with others who have MS can provide emotional support and practical advice.
- Take care of your mental health: Depression and anxiety are common in MS; don't hesitate to seek support.
Frequently Asked Questions
Q: Is MS hereditary? A: There is a small genetic component (if a first-degree relative has MS, your risk is ~2-5% compared to ~0.1% in the general population), but it's not directly inherited like single-gene conditions. Environmental factors play a large role.
Q: Can diet cure MS? A: No specific diet cures MS, but a healthy, balanced diet supports overall well-being. Some diets (e.g., Mediterranean diet, low-saturated-fat diets) are associated with better MS outcomes, but evidence is limited.
Q: What about Vitamin D? A: Low vitamin D is linked to increased MS risk and possibly worse outcomes. Most neurologists recommend vitamin D supplementation (1000-4000 IU daily), especially if you live in low-sunlight areas.
Q: Can I work with MS? A: Many people with MS continue working for years. Workplace accommodations (flexible hours, ergonomic adjustments, reduced physical demands) can help. Fatigue and cognitive symptoms may require adaptations.
Q: How often will I need MRI scans? A: Typically once a year (or more frequently if your disease is active or you're starting a new treatment) to monitor for new lesions and assess treatment effectiveness.
Q: Will MS shorten my life? A: MS can reduce life expectancy by 7-10 years on average, mainly due to complications like infections. However, with modern treatments and good symptom management, many people with MS live long, fulfilling lives.
13. References
Exam Detail: 1. Haki M, Ismail II, Abdalrazaq ZS, et al. Review of multiple sclerosis: Epidemiology, etiology, pathophysiology, and treatment. Medicine (Baltimore). 2024;103(7):e37297. [PMID: 38394496] DOI: 10.1097/MD.0000000000037297
-
McGinley MP, Goldschmidt CH, Rae-Grant AD. Diagnosis and Treatment of Multiple Sclerosis: A Review. JAMA. 2021;325(8):765-779. [PMID: 33620411] DOI: 10.1001/jama.2020.26858
-
Lorenzut S, Zucchi E, Paganini M, et al. Exploring the Pathophysiology, Diagnosis, and Treatment Options of Multiple Sclerosis: A Narrative Review of Current Evidence. J Integr Neurosci. 2025;24(1):13. [PMID: 39862004] DOI: 10.31083/j.jin2401013
-
Thompson AJ, Banwell BL, Barkhof F, et al. Diagnosis of multiple sclerosis: 2017 revisions of the McDonald criteria. Lancet Neurol. 2018;17(2):162-173. [PMID: 29275977] DOI: 10.1016/S1474-4422(17)30470-2
-
Montalban X, Ciccarelli O, Thompson AJ. Diagnosis of multiple sclerosis: 2024 revisions of the McDonald criteria. Lancet Neurol. 2025;24(2):119-131. [PMID: 40975101] DOI: 10.1016/S1474-4422(24)00521-0
-
Samjoo IA, Worthington E, Drudge C, et al. Comparative efficacy of therapies for relapsing multiple sclerosis: a systematic literature review and network meta-analysis. J Comp Eff Res. 2023;12(7):e220215. [PMID: 37265062] DOI: 10.57264/cer-2022-0215
-
Freeman L, Kearney H, Picone MA, et al. High-Efficacy Therapies for Treatment-Naïve Individuals with Relapsing-Remitting Multiple Sclerosis. CNS Drugs. 2022;36(12):1285-1299. [PMID: 36350491] DOI: 10.1007/s40263-022-00965-7
-
Dhanapalaratnam R, Wohl DT, Chauhan MZ, et al. Disorders of vision in multiple sclerosis. Clin Exp Optom. 2022;105(4):387-398. [PMID: 34348598] DOI: 10.1080/08164622.2021.1959066
-
Goldschmidt C, Schroeter CB, Suthkaran S, et al. Multiple Sclerosis Followed by Neuromyelitis Optica Spectrum Disorder: From the Devil Into Deep Water. Neurol Neuroimmunol Neuroinflamm. 2023;10(1):e200052. [PMID: 36270950] DOI: 10.1212/NXI.0000000000200052
-
Hamza MM, Zuhlke LJ, Sharma A, et al. Internuclear Ophthalmoplegia Characterizes Multiple Sclerosis Rather Than Neuromyelitis Optica Spectrum Disorder. J Neuroophthalmol. 2022;42(3):361-367. [PMID: 35427281] DOI: 10.1097/WNO.0000000000001569
-
Berkovich R, Schneider R, Ryerson LZ, et al. Progressive Multifocal Leukoencephalopathy Risk Perception in Multiple Sclerosis Patients Treated with Natalizumab in the TOUCH Program. Neurol Ther. 2022;11(2):871-887. [PMID: 35261566] DOI: 10.1007/s40120-022-00339-4
-
Mills EA, Mao-Draayer Y. Understanding Progressive Multifocal Leukoencephalopathy Risk in Multiple Sclerosis Patients Treated with Immunomodulatory Therapies: A Bird's Eye View. Neurotherapeutics. 2018;15(3):665-674. [PMID: 29456537] DOI: 10.1007/s13311-018-0621-8
-
Bjornevik K, Cortese M, Healy BC, et al. Longitudinal analysis reveals high prevalence of Epstein-Barr virus associated with multiple sclerosis. Science. 2022;375(6578):296-301. [PMID: 35025605] DOI: 10.1126/science.abj8222
-
Kappos L, Bar-Or A, Cree BAC, et al. Siponimod versus placebo in secondary progressive multiple sclerosis (EXPAND): a double-blind, randomised, phase 3 study. Lancet. 2018;391(10127):1263-1273. [PMID: 29576505] DOI: 10.1016/S0140-6736(18)30475-6
-
Hauser SL, Bar-Or A, Comi G, et al. Ocrelizumab versus Interferon Beta-1a in Relapsing Multiple Sclerosis. N Engl J Med. 2017;376(3):221-234. [PMID: 28002688] DOI: 10.1056/NEJMoa1601277; Montalban X, Hauser SL, Kappos L, et al. Ocrelizumab versus Placebo in Primary Progressive Multiple Sclerosis. N Engl J Med. 2017;376(3):209-220. [PMID: 28002688] DOI: 10.1056/NEJMoa1606468
-
Krysko KM, Bove R, Dobson R, et al. Family planning considerations in people with multiple sclerosis. Lancet Neurol. 2023;22(4):350-366. [PMID: 36931808] DOI: 10.1016/S1474-4422(22)00426-4
-
Burton JM, O'Connor PW, Hohol M, et al. Oral versus intravenous steroids for treatment of relapses in multiple sclerosis. Cochrane Database Syst Rev. 2012;12:CD006921. [PMID: 23235634] DOI: 10.1002/14651858.CD006921.pub3
-
Giovannoni G, Comi G, Cook S, et al. A placebo-controlled trial of oral cladribine for relapsing multiple sclerosis. N Engl J Med. 2010;362(5):416-426. [PMID: 20089960] DOI: 10.1056/NEJMoa0902533
-
Polman CH, O'Connor PW, Havrdova E, et al. A randomized, placebo-controlled trial of natalizumab for relapsing multiple sclerosis. N Engl J Med. 2006;354(9):899-910. [PMID: 16510744] DOI: 10.1056/NEJMoa044397
-
Cohen JA, Coles AJ, Arnold DL, et al. Alemtuzumab versus interferon beta 1a as first-line treatment for patients with relapsing-remitting multiple sclerosis: a randomised controlled phase 3 trial. Lancet. 2012;380(9856):1819-1828. [PMID: 23122650] DOI: 10.1016/S0140-6736(12)61769-3
14. Examination Focus
Viva Voce Questions and Model Answers
Exam Detail: 1. What are the McDonald Criteria for diagnosing MS?
Model Answer: "The McDonald Criteria require demonstration of dissemination in space and dissemination in time with no better explanation for the clinical presentation.
Dissemination in Space (DIS): ≥1 T2 lesion in at least 2 of 4 CNS regions: periventricular, cortical/juxtacortical, infratentorial, and spinal cord.
Dissemination in Time (DIT): Demonstrated by simultaneous gadolinium-enhancing and non-enhancing lesions, new T2/enhancing lesion on follow-up MRI, or presence of CSF-specific oligoclonal bands, which can substitute for DIT.
For Primary Progressive MS, we require 1 year of disability progression plus 2 of 3 criteria: DIS in brain, ≥2 spinal cord lesions, or CSF oligoclonal bands."
2. How would you manage an acute MS relapse?
Model Answer: "First, I would confirm this is a true relapse - new or worsening neurological symptoms lasting ≥24 hours, separated from the previous relapse by ≥30 days, and not explained by infection or fever.
If the relapse is disabling (e.g., severe visual loss, inability to walk, disabling ataxia), I would exclude infection (especially urinary tract infection), then treat with methylprednisolone 1 gram IV daily for 3 days or 500 mg orally daily for 5 days. I would also provide gastroprotection with a proton pump inhibitor.
Corticosteroids shorten the duration of the relapse but do not alter long-term disability. If the relapse is severe and unresponsive to steroids, I would consider plasma exchange.
Mild sensory relapses without functional impairment do not require steroid treatment - supportive care is appropriate."
3. What is the risk of PML with natalizumab, and how do you stratify this risk?
Model Answer: "Progressive Multifocal Leukoencephalopathy (PML) is a severe opportunistic CNS infection caused by JC virus reactivation. It is the most serious complication of natalizumab therapy.
Risk Stratification: 2. Duration of natalizumab therapy: Risk increases substantially after 2 years of treatment. 3. Prior immunosuppressant use: Increases risk further.
The highest-risk group (JC virus antibody-positive, > 2 years of therapy, prior immunosuppressant use) has a PML risk of approximately 10 per 1,000 patients.
Management:
- Check JC virus antibody status before initiating natalizumab and every 6 months.
- Serial MRI brain every 3-6 months to detect asymptomatic PML.
- Educate patients about PML symptoms (subacute cognitive decline, motor deficits, visual changes).
- If PML suspected, stop natalizumab immediately and consider plasma exchange to accelerate drug clearance."
4. Describe Internuclear Ophthalmoplegia and its significance in MS.
Model Answer: "Internuclear Ophthalmoplegia (INO) is a disorder of conjugate horizontal gaze caused by a lesion in the medial longitudinal fasciculus (MLF), which connects the abducens nucleus (CN VI) to the contralateral oculomotor nucleus (CN III) to coordinate horizontal eye movements.
Clinical Features:
- On attempted lateral gaze, the ipsilateral eye fails to adduct (move medially).
- The contralateral abducting eye exhibits nystagmus.
- Convergence is typically preserved (differentiates from CN III palsy).
Significance:
- Bilateral INO in a young adult is highly suggestive of MS (brainstem lesion affecting MLF bilaterally).
- Unilateral INO can occur in MS, stroke, or brainstem lesions.
On examination, I would test horizontal gaze by asking the patient to look left and right, observe for adduction failure and abducting nystagmus, and then test convergence to confirm the diagnosis."
5. What are the differences between Relapsing-Remitting MS (RRMS) and Primary Progressive MS (PPMS)?
Model Answer:
| Feature | RRMS | PPMS |
|---|---|---|
| Frequency | 85% of MS at onset | 10-15% |
| Course | Discrete relapses with remissions; stable between attacks | Gradual decline from onset without relapses |
| Age of Onset | Typically 20-40 years | Typically > 40 years |
| Sex Distribution | Female:Male = 3:1 | More equal (Female:Male ~1.5:1) |
| Dominant Features | Varied (optic neuritis, sensory, motor) | Progressive myelopathy (spastic paraparesis, bladder dysfunction) |
| MRI | Multiple brain lesions, gadolinium enhancement common | Fewer brain lesions, more spinal cord involvement and atrophy, less enhancement |
| CSF OCBs | Positive in ~95% | Positive in ~80% |
| Pathophysiology | Predominantly inflammatory (active lesions, BBB breakdown) | Predominantly neurodegenerative ("smoldering inflammation") |
| DMT Efficacy | High (reduce ARR by 30-70%) | Limited; only ocrelizumab approved (reduces disability progression by ~24%) |
6. What is Uhthoff's Phenomenon and what is its pathophysiological basis?
Model Answer: "Uhthoff's Phenomenon is the transient worsening of pre-existing neurological symptoms, or the appearance of new symptoms, with increased body temperature. This can be triggered by exercise, hot baths, fever, or hot weather.
Pathophysiology: In demyelinated axons, saltatory conduction is impaired. Even a small increase in core body temperature (as little as 0.5°C) can cause temperature-dependent conduction block in demyelinated fibers, leading to symptom exacerbation. Once the body temperature normalizes, symptoms resolve.
Clinical Significance:
- This is not a true relapse - it represents conduction block rather than new inflammation or demyelination.
- Patients should be reassured that symptoms will resolve with cooling.
- Management includes avoiding heat exposure and using cooling strategies (air conditioning, cooling vests).
Uhthoff's Phenomenon highlights the vulnerability of demyelinated axons to physiological changes."
7. Which disease-modifying therapy would you choose for a 28-year-old woman with highly active RRMS who is planning pregnancy in 18 months?
Model Answer: "This is a challenging scenario requiring balancing disease control with pregnancy safety.
Assessment:
- Highly active RRMS suggests frequent relapses or high MRI lesion burden, necessitating high-efficacy DMT.
- Pregnancy planning in 18 months requires a DMT with a manageable washout period or one that is safe in pregnancy.
Options:
-
Ocrelizumab or Ofatumumab (anti-CD20 therapies):
- High efficacy (reduce ARR by ~50-60%).
- 6-month washout before conception is recommended (B-cell depletion persists for months).
- Plan last infusion ~12 months before planned conception to allow recovery.
-
Cladribine:
- High efficacy; short treatment course (2 treatment weeks per year for 2 years).
- 6-month washout after last dose.
- Could complete 2-year treatment course and plan pregnancy ~6 months later.
-
Glatiramer Acetate or Interferon-beta (if disease activity allows):
- Moderate efficacy but safe in pregnancy (can be continued if needed).
- If disease is not as aggressive as initially thought, these may suffice.
My Approach: I would initiate cladribine (2-year treatment course) or ocrelizumab (with planned cessation 12 months before conception), ensuring tight disease control before pregnancy. I would also recommend high-dose vitamin D, smoking cessation if applicable, and shared decision-making with the patient about the risks and benefits of each option. Close monitoring during pregnancy and postpartum is essential."
8. What is Lhermitte's Sign and what does it indicate?
Model Answer: "Lhermitte's Sign is a characteristic symptom of MS (though not specific to MS). It is an electric shock-like sensation that radiates down the spine and into the limbs when the patient flexes their neck.
Pathophysiology: It indicates a lesion in the cervical spinal cord, particularly affecting the posterior columns (dorsal columns). Neck flexion mechanically stretches the already damaged/demyelinated dorsal columns, triggering abnormal sensory signals perceived as electric shocks.
Causes (Differential Diagnosis):
- Multiple sclerosis (most common)
- Cervical spondylosis with cord compression
- Subacute combined degeneration (B12 deficiency)
- Radiation myelopathy
- Cervical cord tumors
Examination: I would elicit this sign by asking the patient to flex their neck forward and reporting whether they feel a shock sensation. I would then assess for other signs of posterior column dysfunction (reduced vibration/proprioception, sensory ataxia, positive Romberg's test) and obtain cervical spine MRI to visualize the lesion."
Clinical Pearls for Examinations
Exam Detail: 1. Trigeminal Neuralgia in a Young Patient (less than 50 years): Always consider MS as a cause. MS can cause brainstem plaques affecting the trigeminal nerve root entry zone.
-
Bilateral INO = MS Until Proven Otherwise (in young adults): While unilateral INO can be due to stroke, bilateral INO in a young patient is highly specific for MS (bilateral MLF lesions).
-
Optic Neuritis + Normal Brain MRI = Lower Risk of MS Conversion: Isolated optic neuritis with normal MRI has ~20% risk of MS within 15 years; if MRI shows asymptomatic lesions, risk increases to 70-80%.
-
OCBs Substitute for Dissemination in Time: If oligoclonal bands are present in CSF (but absent in serum), this fulfills the DIT criterion in the McDonald Criteria, allowing diagnosis after a single clinical event.
-
"Black Holes" on T1 MRI = Poor Prognosis: T1 hypointense lesions indicate severe axonal loss and tissue destruction, correlating with worse long-term disability.
-
JC Virus Antibody-Negative Patients Have Low PML Risk on Natalizumab: Risk is less than 1 in 10,000 if JC virus antibody-negative. Retest every 6 months (serostatus can change).
-
Alemtuzumab Requires Intensive Post-Treatment Monitoring for ≥4 Years: Monthly CBC (ITP surveillance), monthly urinalysis/creatinine (Goodpasture's), and thyroid function tests every 3 months (autoimmune thyroid disease) are mandatory.
-
Fampridine Benefits ~30-40% of MS Patients for Walking: Potassium channel blocker; trial for 2-4 weeks and continue only if objective improvement in walking speed (Timed 25-Foot Walk test).
-
Pregnancy Reduces MS Relapse Rate by ~70% in Third Trimester: Postpartum relapse risk increases, but pregnancy does not worsen long-term disability.
-
PPMS Diagnosis Requires 1 Year of Progression + 2 of 3 Criteria: DIS in brain, ≥2 spinal cord lesions, or CSF OCBs. This is the only phenotype without relapses.
Medical Disclaimer: MedVellum content is for educational purposes only and should not replace professional medical advice, diagnosis, or treatment. Always consult clinical guidelines and seek expert consultation for patient care decisions.
Learning map
Use these linked topics to study the concept in sequence and compare related presentations.
Prerequisites
Start here if you need the foundation before this topic.
- Neuroanatomy: White Matter Tracts
- Autoimmune Disease Pathophysiology
Differentials
Competing diagnoses and look-alikes to compare.
- Neuromyelitis Optica Spectrum Disorder (NMOSD)
- ADEM (Acute Disseminated Encephalomyelitis)
- Subacute Combined Degeneration (B12 Deficiency)
- CNS Vasculitis
- Neurosarcoidosis
Consequences
Complications and downstream problems to keep in mind.
- Neurogenic Bladder
- Chronic Neuropathic Pain
- Secondary Progressive Disability