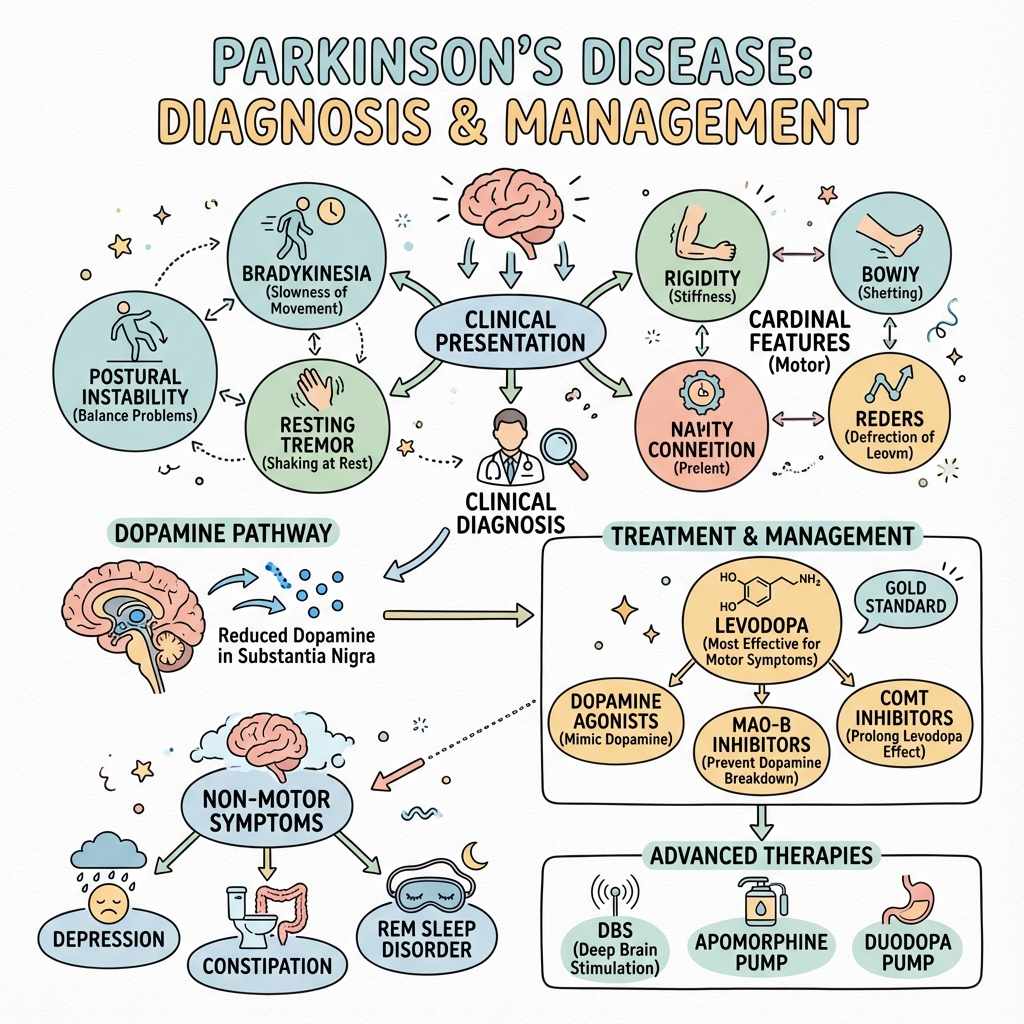
Parkinson's Disease (Adult)
Parkinson's disease (PD) is a progressive neurodegenerative disorder characterized by the selective loss of dopaminergic... MRCP, USMLE exam preparation.
What matters first
Parkinson's disease (PD) is a progressive neurodegenerative disorder characterized by the selective loss of dopaminergic... MRCP, USMLE exam preparation.
Rapid progression of gait impairment (suggests atypical parkinsonism)
9 Jan 2026
Generated educational material; verify before clinical use.
Visible references section
See the concept before reading it
Study the key anatomy, imaging, and decision pathways as full teaching plates.
Clinical board
A visual summary of the highest-yield teaching signals on this page.
Urgent signals
Safety-critical features pulled from the topic metadata.
- Rapid progression of gait impairment (suggests atypical parkinsonism)
- Early autonomic failure (suggests MSA)
- Early falls within first year (suggests PSP)
- Supranuclear vertical gaze palsy (pathognomonic for PSP)
Exam focus
Current exam surfaces linked to this topic.
- MRCP
- USMLE
- Medical Finals
Linked comparisons
Differentials and adjacent topics worth opening next.
- Essential Tremor
- Progressive Supranuclear Palsy
Content status and exam context
This page is AI-generated educational content. It may contain errors or omissions and is not a substitute for current guidelines, local protocols, senior clinical judgement, or professional medical advice.
MedVellum does not claim an individual clinician reviewer, board certification, or professional credential for this page unless a future version names a real, verifiable reviewer.
Clinical explanation and evidence
Parkinson's Disease (Adult)
1. Clinical Overview
Summary
Parkinson's disease (PD) is a progressive neurodegenerative disorder characterized by the selective loss of dopaminergic neurons in the substantia nigra pars compacta, resulting in striatal dopamine deficiency and the characteristic motor syndrome. [1,2] It is the second most common neurodegenerative disease after Alzheimer's disease, affecting over 6 million people worldwide with prevalence expected to double by 2040 due to population aging. [3]
The cardinal motor features comprise bradykinesia (mandatory for diagnosis), resting tremor, rigidity, and postural instability (the latter typically emerging in later disease stages). However, PD is now recognized as a multisystem alpha-synucleinopathy with extensive non-motor manifestations—including autonomic dysfunction, cognitive impairment, neuropsychiatric symptoms, sleep disorders, and sensory abnormalities—that often precede motor symptoms by 10-20 years during the "prodromal phase." [2,4]
The diagnosis remains fundamentally clinical, based on the Movement Disorder Society (MDS) Clinical Diagnostic Criteria, which require the presence of parkinsonism (bradykinesia plus tremor or rigidity), the absence of exclusion criteria, and the presence of supportive criteria. [5] Management is primarily symptomatic, centering on dopamine replacement therapy with levodopa (the most effective agent), dopamine agonists, and MAO-B inhibitors. While no disease-modifying therapy currently exists, substantial research is ongoing. Advanced therapies including deep brain stimulation (DBS), levodopa-carbidopa intestinal gel (LCIG), and subcutaneous apomorphine infusion provide significant benefit for patients with motor complications refractory to optimized oral therapy. [6,7]
Key Facts
| Domain | Key Information |
|---|---|
| Definition | Progressive alpha-synucleinopathy causing dopaminergic neurodegeneration in the substantia nigra pars compacta |
| Prevalence | 1-2% of population > 60 years; 4-5% > 85 years; ~6.2 million affected globally |
| Core Motor Features | Bradykinesia (required) + resting tremor and/or rigidity; postural instability in advanced stages |
| Pathological Hallmark | Alpha-synuclein-containing Lewy bodies and Lewy neurites with loss of pigmented neurons in SNpc |
| Diagnosis | Clinical diagnosis using MDS criteria; positive response to dopaminergic therapy is supportive |
| First-Line Treatment | Levodopa (most effective); alternatives include dopamine agonists or MAO-B inhibitors based on age, severity, and patient preference |
| Motor Complications | Wearing-off, on-off fluctuations, and dyskinesias develop in 40-50% of patients within 5 years of levodopa therapy |
| Life Expectancy | Modestly reduced (mortality ratio 1.5-2.0); death typically from aspiration pneumonia, falls, or cardiovascular disease |
Clinical Pearls
"Bradykinesia is the sine qua non of Parkinson's" — You cannot diagnose PD without bradykinesia, which must include slowness of movement AND progressive decrement in speed or amplitude during repetitive movements. A patient with tremor alone does not have Parkinson's disease.
"The 4-6 Hz pill-rolling tremor" — The classic PD tremor is a 4-6 Hz resting tremor that characteristically decreases or disappears during voluntary movement and during sleep. It is typically asymmetric and may have a re-emergent component when the posture is maintained.
"Watch the eyes and the gait for Parkinson-Plus" — Early falls (within the first year) or supranuclear vertical gaze palsy should immediately raise suspicion for Progressive Supranuclear Palsy. Early autonomic failure suggests Multiple System Atrophy.
"Non-motor symptoms often matter more to patients" — While clinicians focus on the motor syndrome, patients frequently report that depression, cognitive impairment, sleep fragmentation, fatigue, and autonomic dysfunction are more disabling than the motor symptoms.
"The honeymoon period typically lasts 3-5 years" — Most patients experience excellent symptom control with levodopa initially, but motor complications (fluctuations and dyskinesias) eventually develop in the majority. After 10 years, up to 90% will experience motor fluctuations. [8]
"Time matters for DBS" — The EARLYSTIM trial demonstrated that DBS is most beneficial when offered early in the course of motor complications, not as a therapy of last resort. [7]
Why This Matters Clinically
Parkinson's disease is a major cause of disability in the aging population, with profound impacts on quality of life, caregiver burden, and healthcare costs. Early and accurate diagnosis is crucial for:
- Initiating appropriate symptomatic treatment to maintain function and quality of life
- Providing accurate prognosis — distinguishing idiopathic PD (relatively good prognosis) from "Parkinson-plus" syndromes (poor prognosis)
- Avoiding harm — drug-induced parkinsonism from dopamine blockers requires cessation, not dopaminergic treatment
- Planning for the future — discussing driving, work capacity, advance care planning
- Timely referral for advanced therapies — recognizing the "levodopa-induced complications" phase for DBS or pump therapies
- Managing the whole patient — addressing the full spectrum of motor and non-motor symptoms
As the "Parkinson's pandemic" unfolds with projected doubling of cases by 2040, competence in PD diagnosis and management is essential for all physicians. [3]
2. Epidemiology
Incidence and Prevalence
Parkinson's disease demonstrates a strong age-dependent distribution, with incidence and prevalence rising exponentially after age 60. [1,9]
| Measure | General Population | Age > 60 Years | Age > 80 Years |
|---|---|---|---|
| Prevalence | 100-200 per 100,000 | 1,000-1,500 per 100,000 (1-1.5%) | 3,000-4,000 per 100,000 (3-4%) |
| Incidence | 8-18 per 100,000 person-years | 100-150 per 100,000 person-years | 200-300 per 100,000 person-years |
Global Burden: The Global Burden of Disease study documented 6.2 million individuals with PD worldwide in 2015, representing a 118% increase since 1990. This is the fastest-growing neurological disorder. [3]
Age of Onset:
- Late-onset PD: > 60 years (most common)
- Early-onset PD: 40-60 years
- Young-onset PD (YOPD): less than 40 years (~5% of cases; higher genetic contribution)
- Juvenile-onset PD: less than 21 years (rare; almost always genetic)
Demographics
| Factor | Details | Evidence |
|---|---|---|
| Age | Strongest risk factor; exponential increase after age 65; median onset age 60 years | [1,9] |
| Sex | Male predominance; male:female ratio 1.4-2.0:1 across most populations | Possibly due to estrogen neuroprotection [9] |
| Ethnicity | Highest rates in Hispanics and non-Hispanic whites; lower in Black and Asian populations | May reflect ascertainment bias or genetic factors [9] |
| Geography | Higher prevalence in industrialized countries; increasing in developing nations | Environmental factors and improved diagnosis [3] |
| Genetics | ~10-15% have positive family history; ~5-10% have identifiable monogenic forms | LRRK2, GBA, PRKN most common [10] |
Genetic Architecture of Parkinson's Disease
Understanding the genetic landscape is increasingly relevant for counseling and emerging therapies. [10]
Autosomal Dominant Genes
| Gene | Locus | Key Features | Frequency |
|---|---|---|---|
| SNCA (α-synuclein) | PARK1/4 | Point mutations or multiplications; aggressive course with dementia | Rare |
| LRRK2 | PARK8 | Most common genetic cause; classic PD phenotype; incomplete penetrance | 1-2% sporadic; 5-10% familial; 20-40% in Ashkenazi and North African Arab populations |
| VPS35 | PARK17 | Late-onset, classic phenotype | Very rare |
Autosomal Recessive Genes
| Gene | Locus | Key Features | Frequency |
|---|---|---|---|
| PRKN (Parkin) | PARK2 | Young-onset (less than 40); slow progression; excellent levodopa response; frequent dystonia | Most common early-onset cause |
| PINK1 | PARK6 | Young-onset; similar to PRKN | Second most common early-onset |
| DJ-1 | PARK7 | Young-onset; psychiatric features | Rare |
Major Susceptibility Gene
| Gene | Key Features | Clinical Significance |
|---|---|---|
| GBA (Glucocerebrosidase) | Most common genetic risk factor (5-10% of PD patients); heterozygous carriers have 5-8x increased risk; associated with earlier onset, faster progression, and increased cognitive decline | Target for disease-modifying therapy trials; genetic counseling implications [10] |
Risk Factors
Non-Modifiable Risk Factors:
- Age (primary risk factor)
- Male sex
- Family history (especially in YOPD)
- Specific genetic variants (GBA, LRRK2, SNCA, PRKN)
Modifiable/Environmental Risk Factors:
| Factor | Association | Proposed Mechanism | Evidence Level |
|---|---|---|---|
| Pesticide exposure | Increased risk (1.5-2.5x) | Rotenone and paraquat cause mitochondrial complex I inhibition; directly toxic to dopaminergic neurons | Strong [9] |
| Head trauma | Increased risk (1.5x) | Neuroinflammation; protein aggregation; blood-brain barrier disruption | Moderate |
| Well water | Increased risk | Possible pesticide/herbicide contamination | Moderate |
| Rural living | Increased risk | Agricultural chemical exposure | Moderate |
| Caffeine intake | Decreased risk (25-30%) | Adenosine A2A receptor antagonism; neuroprotective | Strong [9] |
| Smoking | Decreased risk (40-60%) | Paradoxical; mechanism unclear; possibly nicotine-mediated neuroprotection or MAO-B inhibition | Strong (but do not recommend) [9] |
| Physical exercise | Decreased risk (30-40%) | Enhanced neuroplasticity; increased BDNF; improved mitochondrial function | Strong [11] |
| Urate levels | Decreased risk (higher urate protective) | Antioxidant properties; scavenges reactive oxygen species | Moderate |
| NSAIDs (ibuprofen) | Decreased risk | Anti-inflammatory mechanisms | Weak-moderate |
Differential Diagnosis of Parkinsonism
Approximately 15-25% of patients initially diagnosed with PD are ultimately found to have an alternative diagnosis. [4]
| Condition | Distinguishing Features | Key Investigations |
|---|---|---|
| Essential Tremor | Action/postural tremor (not resting); symmetric; family history; head/voice involvement; improves with alcohol; no bradykinesia | Clinical; DaTscan shows normal uptake |
| Drug-Induced Parkinsonism | Symmetric; temporal relationship to dopamine blockers (antipsychotics, metoclopramide, prochlorperazine); resolves after drug cessation | Medication review; DaTscan typically normal |
| Vascular Parkinsonism | "Lower body parkinsonism" (gait predominant); stepwise progression; pyramidal signs; poor levodopa response; vascular risk factors | MRI shows extensive white matter disease/infarcts |
| Normal Pressure Hydrocephalus | Triad: gait apraxia, dementia, incontinence; "magnetic gait"; improves with CSF drainage | MRI shows ventriculomegaly; lumbar tap test |
| Multiple System Atrophy (MSA) | Early severe autonomic failure; cerebellar signs (MSA-C); stridor; poor levodopa response; rapid progression | MRI: "hot cross bun" (pons), "slit" putamen; MIBG normal |
| Progressive Supranuclear Palsy (PSP) | Early falls (within first year); supranuclear vertical gaze palsy; axial rigidity > limb; "surprised" facies; poor levodopa response | MRI: "hummingbird sign" (midbrain atrophy); "morning glory" sign |
| Corticobasal Syndrome (CBS) | Markedly asymmetric; limb apraxia; alien limb phenomenon; cortical sensory loss; myoclonus; cognitive impairment | MRI: asymmetric cortical atrophy |
| Dementia with Lewy Bodies (DLB) | Dementia onset before or within 1 year of motor symptoms (1-year rule); visual hallucinations; fluctuating cognition; REM sleep behavior disorder | DaTscan abnormal; MIBG reduced |
3. Pathophysiology
The Alpha-Synuclein Hypothesis
The central pathogenic event in Parkinson's disease is the misfolding and aggregation of alpha-synuclein (α-synuclein), a 140-amino acid protein normally found at presynaptic terminals, into insoluble fibrillar inclusions. [1,2]
Alpha-Synuclein Structure and Function
- Normal function: Involved in synaptic vesicle trafficking, neurotransmitter release, and synaptic plasticity
- Structure: Natively unfolded protein that adopts α-helical conformation when bound to membranes
- Aggregation cascade:
- Monomers → Oligomers (most toxic species) → Protofibrils → Mature fibrils
- Fibrils deposit as Lewy bodies (cytoplasmic) and Lewy neurites (in axons and dendrites)
- Oligomers impair mitochondrial function, proteasomal degradation, and membrane integrity
The Braak Hypothesis: Prion-like Propagation
Braak and colleagues proposed that α-synuclein pathology originates in peripheral sites and spreads centripetally to the brain via the vagus nerve. [12]
BRAAK STAGING OF PARKINSON'S DISEASE PATHOLOGY
Stage 1-2: PERIPHERAL/LOWER BRAINSTEM (PRODROMAL)
├── Olfactory bulb → Anosmia
├── Enteric nervous system → Constipation
├── Dorsal motor nucleus of vagus → Autonomic dysfunction
└── Lower brainstem → REM sleep behavior disorder
↓ Rostral progression via connected neurons
Stage 3-4: MIDBRAIN (MOTOR PHASE)
├── Substantia nigra pars compacta → Cardinal motor features
├── Locus coeruleus → Depression, attention deficits
├── Raphe nuclei → Sleep disorders, mood disturbance
└── Pedunculopontine nucleus → Gait and balance dysfunction
↓ Continued spread to higher structures
Stage 5-6: CORTICAL (ADVANCED)
├── Limbic structures → Neuropsychiatric symptoms
├── Neocortex → Cognitive impairment, dementia
└── Hippocampus → Memory dysfunction
Evidence for prion-like spread:
- Post-mortem studies of fetal dopamine neuron transplant recipients showed development of Lewy bodies in grafted neurons after 10-16 years
- α-synuclein fibrils can seed aggregation when injected into animal brains
- Truncal vagotomy associated with reduced PD risk
Substantia Nigra Degeneration
The substantia nigra pars compacta (SNpc) contains approximately 400,000-600,000 dopaminergic neurons in the healthy brain. [1,2]
Why SNpc Neurons Are Selectively Vulnerable
| Factor | Mechanism |
|---|---|
| High metabolic demand | Massive axonal arbor (each SNpc neuron can have > 1 million synapses in striatum); autonomous pacemaking requires high energy |
| Dopamine metabolism | Dopamine auto-oxidation produces reactive oxygen species; MAO-B metabolism generates hydrogen peroxide |
| Iron content | SNpc has highest brain iron concentration; iron catalyzes free radical formation (Fenton reaction) |
| Neuromelanin | Pigment accumulates iron and environmental toxins |
| Calcium handling | L-type calcium channels in SNpc neurons create high cytosolic calcium, stressing mitochondria |
| Limited antioxidant capacity | Relatively low glutathione compared to other brain regions |
Threshold for Symptom Onset
- Motor symptoms emerge when 50-60% of SNpc dopaminergic neurons are lost
- This corresponds to 70-80% depletion of striatal dopamine
- Compensatory mechanisms (increased dopamine turnover, receptor upregulation) maintain function initially
- Putamen is affected earlier and more severely than caudate
Mitochondrial Dysfunction and Oxidative Stress
Mitochondrial complex I deficiency is a consistent finding in PD. [2]
MITOCHONDRIAL DYSFUNCTION CASCADE
Environmental toxins (Rotenone, MPTP, Paraquat)
↓
Inhibit Complex I of electron transport chain
↓
↓ ATP production + ↑ Reactive Oxygen Species (ROS)
↓
┌───────────────────┼───────────────────┐
↓ ↓ ↓
Lipid peroxidation Protein oxidation DNA damage
↓ ↓ ↓
Membrane damage Protein aggregation Apoptosis signals
(α-synuclein)
↓
NEURONAL DEATH
Key genetic evidence linking mitochondria to PD:
- PINK1 and Parkin (recessive PD genes) function in mitophagy (clearance of damaged mitochondria)
- DJ-1 is a mitochondrial antioxidant
- MPTP (1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine) causes parkinsonism by inhibiting complex I
Basal Ganglia Circuitry: Direct and Indirect Pathways
Understanding basal ganglia physiology is essential for understanding PD symptoms and the mechanism of DBS. [1,2]
NORMAL BASAL GANGLIA FUNCTION
CORTEX
│
↓ Glutamate (+)
STRIATUM
/ \
D1 (direct) D2 (indirect)
↓ ↓
GPi/SNr ←──────── GPe
│ │
↓ ↓
THALAMUS STN
│ │
↓ ↓
CORTEX ←──────────────┘
DIRECT PATHWAY: Facilitates movement (D1 receptors)
Striatum ─(GABA)→ GPi/SNr ─(GABA)→ Thalamus → Cortex
Net effect: Disinhibition of thalamus → MOVEMENT
INDIRECT PATHWAY: Inhibits movement (D2 receptors)
Striatum ─(GABA)→ GPe ─(GABA)→ STN ─(Glutamate)→ GPi/SNr
Net effect: Increased inhibition of thalamus → NO MOVEMENT
Dopamine Depletion Effects
| Normal | Dopamine Depletion in PD |
|---|---|
| Balance between direct and indirect pathways | Underactivity of direct pathway (D1) |
| Smooth, voluntary movement | Overactivity of indirect pathway (D2) |
| Appropriate thalamocortical drive | Net result: Excessive GPi/SNr inhibition of thalamus |
| Reduced thalamocortical drive | |
| Clinical manifestation: Bradykinesia, rigidity |
Non-Dopaminergic Neurodegeneration
PD involves multiple neurotransmitter systems beyond dopamine, explaining the diverse non-motor symptoms. [2]
| Nucleus/System | Neurotransmitter | Symptoms from Degeneration |
|---|---|---|
| Locus coeruleus | Norepinephrine | Depression, anxiety, attention deficits, orthostatic hypotension |
| Raphe nuclei | Serotonin | Depression, sleep disorders, fatigue |
| Nucleus basalis of Meynert | Acetylcholine | Cognitive impairment, dementia, visual hallucinations |
| Pedunculopontine nucleus | Acetylcholine | Gait disorders, falls, REM sleep behavior disorder |
| Hypothalamus | Various | Sleep-wake disturbances, temperature dysregulation |
| Dorsal motor nucleus of vagus | Acetylcholine | GI dysmotility, constipation |
| Sympathetic ganglia | Norepinephrine | Orthostatic hypotension, cardiac denervation |
4. Clinical Presentation
Motor Symptoms: The Cardinal Features
The diagnosis of Parkinson's disease requires the presence of parkinsonism, defined as bradykinesia plus at least one of resting tremor or rigidity. [5]
1. Bradykinesia (Required for Diagnosis)
Bradykinesia is the defining motor feature and is mandatory for diagnosis. [5]
Definition: Slowness of movement initiation with progressive reduction in speed AND amplitude during repetitive movements (the "decremental" feature is pathognomonic).
| Feature | Clinical Assessment | Description |
|---|---|---|
| Finger tapping | Tap thumb and index finger repeatedly | Progressive reduction in amplitude and speed |
| Hand movements | Open and close fist repeatedly | Decrement and hesitations |
| Pronation-supination | Rapid alternating forearm movements | Irregular, slow, with arrests |
| Toe tapping | Tap foot on ground | Similar decrement pattern |
| Leg agility | Stomp foot up and down | Reduced amplitude over time |
| Global bradykinesia | Overall impression | Slowness in all movements |
Clinical Manifestations of Bradykinesia:
- Hypomimia ("masked facies"): Reduced facial expression and blink rate
- Hypophonia: Soft, monotonous voice
- Micrographia: Handwriting becomes progressively smaller
- Reduced arm swing: Usually unilateral initially
- Difficulty with fine motor tasks: Buttoning, using utensils
- Prolonged movement times: Everything takes longer
2. Resting Tremor
Present in 70-75% of patients at diagnosis; some never develop tremor ("akinetic-rigid" phenotype). [1,2]
| Characteristic | Description |
|---|---|
| Frequency | 4-6 Hz (slower than essential tremor's 8-12 Hz) |
| Type | Resting tremor; decreases with voluntary movement |
| Distribution | Usually begins unilaterally in upper limb; may spread to ipsilateral leg, then contralateral side |
| Classic appearance | "Pill-rolling" (thumb moves across fingers) |
| Re-emergent tremor | Postural tremor that appears after a few seconds of maintaining posture (unlike essential tremor which appears immediately) |
| Modulating factors | Increases with stress, mental concentration (counting backwards), and walking; absent during sleep |
Differentiating PD Tremor from Essential Tremor:
| Feature | Parkinson's Disease | Essential Tremor |
|---|---|---|
| Type | Resting | Action/Postural |
| Frequency | 4-6 Hz | 8-12 Hz |
| Onset | Asymmetric | Usually symmetric |
| Re-emergence | Yes (after latency) | No (immediate) |
| Alcohol response | No improvement | Often improves |
| Bradykinesia | Present | Absent |
| Head/voice tremor | Rare (chin = "yes-yes") | Common (head = "no-no") |
| DaTscan | Abnormal | Normal |
3. Rigidity
Increased resistance to passive movement, independent of velocity. [1,2]
| Type | Description | Mechanism |
|---|---|---|
| Lead-pipe rigidity | Constant resistance throughout range of motion | Tonic muscle activation |
| Cogwheel rigidity | Ratchety resistance (tremor superimposed on lead-pipe) | Tremor + rigidity |
| Activated rigidity | Enhancement with contralateral motor activity (Froment's maneuver) | Useful for detecting subtle rigidity |
Distribution: Usually begins unilaterally, affecting neck and proximal limbs; axial rigidity becomes prominent later.
Differentiation from Spasticity:
| Feature | Rigidity (PD) | Spasticity (UMN) |
|---|---|---|
| Velocity dependence | Independent | Velocity-dependent (clasp-knife) |
| Direction | Equal flexion/extension | Greater in flexors (arms) or extensors (legs) |
| Associated features | Bradykinesia, tremor | Hyperreflexia, Babinski sign |
| Distribution | Can be unilateral, any limb | Hemiparesis/paraparesis pattern |
4. Postural Instability
Usually a late feature; early postural instability suggests alternative diagnosis. [5]
- Pull test: Examiner stands behind patient and pulls shoulders backward; > 2 steps to recover or need for catching is abnormal
- Falls: Leading cause of morbidity in advanced PD
- Festination: Progressive acceleration of gait with shortening steps (trying to catch up with center of gravity)
- Freezing of gait (FOG): Sudden, transient inability to initiate or continue walking; often triggered by doorways, turns, or narrow spaces
Motor Phenotypes
Patients can be categorized into motor phenotypes with prognostic implications. [2,4]
| Phenotype | Features | Prognosis |
|---|---|---|
| Tremor-dominant | Prominent tremor, less bradykinesia/rigidity | Better prognosis, slower progression, less dementia |
| Akinetic-rigid (PIGD) | Predominant bradykinesia and rigidity, minimal tremor, early gait involvement | Worse prognosis, faster motor progression, higher dementia risk |
| Mixed | Elements of both | Intermediate |
Non-Motor Symptoms
Non-motor symptoms (NMS) affect virtually all PD patients and often cause greater disability than motor symptoms. [4,13]
Autonomic Dysfunction
| System | Symptoms | Pathophysiology |
|---|---|---|
| Cardiovascular | Orthostatic hypotension (30-50% of patients); supine hypertension | Cardiac sympathetic denervation; loss of vasomotor reflexes |
| Gastrointestinal | Constipation (80%); dysphagia; delayed gastric emptying | Enteric nervous system involvement; vagal dysfunction |
| Urinary | Urgency, frequency, nocturia; incontinence (late) | Detrusor hyperactivity; sphincter dysfunction |
| Sexual | Erectile dysfunction; reduced libido | Autonomic and hormonal factors |
| Sudomotor | Hyperhidrosis or anhidrosis; seborrhea | Thermoregulatory dysfunction |
Neuropsychiatric Manifestations
| Symptom | Prevalence | Key Features |
|---|---|---|
| Depression | 30-50% | May precede motor symptoms; often atypical; apathy can be mistaken for depression |
| Anxiety | 30-40% | Generalized anxiety, panic attacks, social phobia; often occurs with "off" periods |
| Apathy | 40% | Loss of motivation and initiative; distinct from depression |
| Psychosis | 20-40% (late) | Visual hallucinations (benign initially); paranoid delusions; drug-related or intrinsic |
| Cognitive impairment | Mild (25-30%); Dementia (25-80% over disease course) | Executive dysfunction, visuospatial deficits; point prevalence of dementia 30-40% |
| Impulse control disorders | 10-20% (with dopamine agonists) | Pathological gambling, hypersexuality, compulsive shopping, binge eating, punding |
Sleep Disorders
| Disorder | Description | Significance |
|---|---|---|
| REM sleep behavior disorder (RBD) | Acting out dreams (hitting, kicking); loss of REM atonia | Strong prodromal marker; 80% develop synucleinopathy within 15 years |
| Insomnia | Difficulty falling/staying asleep; early awakening | Multifactorial; very common |
| Excessive daytime sleepiness | "Sleep attacks" (especially with dopamine agonists) | Driving safety implications |
| Restless legs syndrome | Urge to move legs; worse at rest and evening | 15-20% of PD patients |
| Sleep fragmentation | Frequent awakenings; due to motor symptoms, nocturia | Very common |
Sensory and Pain Syndromes
| Type | Description |
|---|---|
| Hyposmia/Anosmia | Reduced/absent smell; present in > 80%; highly sensitive prodromal marker |
| Pain | Musculoskeletal (rigidity-related); dystonic pain (often in "off" periods); central/primary PD pain |
| Fatigue | Overwhelming tiredness unrelated to exertion; highly prevalent and disabling |
Prodromal Parkinson's Disease
Increasing recognition of the prodromal (pre-motor) phase has major implications for early identification and future neuroprotective trials. [4]
TIMELINE OF SYMPTOM EMERGENCE
20-10 years before motor onset:
├── Constipation
├── Depression/Anxiety
└── Anosmia/Hyposmia
10-5 years before motor onset:
├── REM sleep behavior disorder
├── Subtle cognitive changes
├── Seborrhea
└── Excessive daytime sleepiness
5-0 years before motor onset:
├── Subtle motor signs (reduced arm swing)
├── Urinary symptoms
├── Erectile dysfunction
└── Orthostatic hypotension
Motor onset:
├── Bradykinesia + tremor/rigidity
└── Asymmetric presentation
MDS Prodromal PD Criteria: Combine risk/protective factors (age, sex, family history, smoking, caffeine) with prodromal markers (RBD, hyposmia, constipation, depression, subtle motor signs, DaTscan) to calculate probability of prodromal PD.
Red Flags for Atypical Parkinsonism
[!CAUTION] Red Flags — Consider Atypical Parkinsonism (Parkinson-Plus):
- Rapid progression — Wheelchair dependence within 5 years
- Early falls — Within the first year of symptom onset (suggests PSP)
- Early severe autonomic failure — Symptomatic orthostatic hypotension or urinary incontinence (suggests MSA)
- Supranuclear vertical gaze palsy — Particularly downgaze limitation (pathognomonic for PSP)
- Poor or absent response to levodopa — Even at adequate doses (≥1000mg/day for ≥1 month)
- Symmetric onset — Idiopathic PD is almost always asymmetric at onset
- Early dementia — Cognitive decline within 1 year of motor onset (suggests DLB; use "1-year rule")
- Cerebellar signs — Ataxia, scanning speech, nystagmus (suggests MSA-C)
- Prominent pyramidal signs — Spasticity, hyperreflexia, Babinski (suggests atypical parkinsonism)
- Inspiratory stridor — Laryngeal dystonia (suggests MSA)
- Alien limb phenomenon — Limb acts independently of patient's will (suggests CBS)
- Disproportionate antecollis — Severe neck flexion (suggests MSA)
5. Clinical Examination
Systematic Neurological Examination for Parkinsonism
1. General Observation
| Feature | What to Look For |
|---|---|
| Hypomimia | Reduced facial expression, "masked facies," decreased blink rate |
| Tremor at rest | Pill-rolling tremor of hands in lap |
| Posture | Stooped posture, flexed neck and trunk (camptocormia if severe) |
| Speech | Hypophonia (soft), monotonous, tachyphemia (accelerated, mumbling) |
| Overall movement | Poverty of spontaneous movement; delayed initiation |
2. Tremor Assessment
| Position | Assessment | Significance |
|---|---|---|
| Rest | Hands resting in lap, supported | PD: Present; ET: Absent |
| Postural | Arms outstretched, fingers spread | PD: Re-emergent after latency; ET: Immediate onset |
| Action/Kinetic | Finger-to-nose testing | PD: Usually suppressed; ET: Present/worse |
| Mental activation | Count backward from 100 by 7s | PD: Tremor enhanced; ET: Variable |
3. Rigidity Assessment
| Joint | Technique | Notes |
|---|---|---|
| Wrist | Passive flexion/extension and pronation/supination | Feel for cogwheel or lead-pipe |
| Elbow | Passive flexion/extension | Often most prominent |
| Shoulder | Passive rotation and abduction | Assess for "frozen shoulder" |
| Neck | Passive rotation | Axial rigidity |
| Ankle | Passive dorsiflexion | Often overlooked |
| Froment's maneuver | Ask patient to move contralateral arm while testing | Activates or enhances subtle rigidity |
4. Bradykinesia Testing (MDS-UPDRS Items)
| Test | Instructions | Abnormal Findings |
|---|---|---|
| Finger tapping | "Tap your index finger and thumb together as fast and big as possible, 10 times" | Decrement in speed/amplitude; hesitations; arrests |
| Hand movements | "Open and close your hand as fast and big as possible" | Decrement; incomplete opening |
| Pronation-supination | "Turn your hand over and back as fast and fully as possible" | Irregular; halting; reduced amplitude |
| Toe tapping | "Tap your foot on the ground as fast and high as possible" | Decrement; freezing |
| Leg agility | "Stomp your foot up and down as fast and high as possible" | Fatiguing; reduced height |
5. Gait and Balance Assessment
| Test | Observation | Abnormal Findings |
|---|---|---|
| Walking | Walk 10 meters, turn, and return | Reduced arm swing (unilateral early); shuffling; short stride; festination |
| Turning | Turn 180 degrees | "En bloc" turning (multiple small steps) |
| Heel walking | Walk on heels | Assess for foot drop/dystonia |
| Tandem gait | Heel-to-toe walking | Imbalance (though not specific to PD) |
| Pull test | Stand behind patient, warn them, pull shoulders backward | Retropulsion; > 2 steps or caught by examiner = abnormal |
| Dual tasking | Walk while counting backward | Freezing; gait deterioration |
6. Additional Signs
| Sign | Description | Significance |
|---|---|---|
| Glabellar tap (Myerson's sign) | Tap glabella; positive if persistent blinking (failure to habituate) | Supports parkinsonism (not specific) |
| Micrographia | Ask patient to write a sentence | Progressive reduction in letter size |
| Hypomimia | Count spontaneous blinks; assess emotional expression | Reduced blink rate (less than 15/min); "reptilian stare" |
| Seborrhea | Inspect face | Oily, flaky skin common in PD |
| Sialorrhea | Observe for drooling | Due to reduced swallowing frequency |
7. Eye Movement Examination (Critical for Parkinson-Plus)
| Finding | Associated Diagnosis |
|---|---|
| Slow vertical saccades (especially downgaze) → Supranuclear gaze palsy | PSP |
| Square-wave jerks | PSP, MSA, cerebellar disease |
| Impaired smooth pursuit | Nonspecific |
| Oculogyric crisis | Drug-induced parkinsonism |
| Normal eye movements | Typical of idiopathic PD (eye movements preserved) |
Specific Named Signs
| Sign | Description | Notes |
|---|---|---|
| Pill-rolling tremor | Thumb and fingers move as if rolling a pill | Classic PD resting tremor |
| Cogwheel rigidity | Ratchety "catches" during passive movement | Tremor superimposed on rigidity |
| Festination | Gait progressively speeds up with shorter steps | Patient trying to catch center of gravity |
| Freezing of gait (FOG) | Sudden transient inability to move feet | Triggered by doorways, turns, narrow spaces |
| "En bloc" turning | Multiple small steps to turn rather than pivoting | Early sign of postural instability |
| Camptocormia | Severe flexion of the trunk while standing/walking | Trunk dystonia in PD |
| Pisa syndrome | Lateral trunk flexion | Drug-induced or disease-related |
| Anterocollis | Severe neck flexion | More common in MSA than PD |
6. Investigations
Clinical Diagnosis
Parkinson's disease remains a clinical diagnosis. There is no definitive diagnostic test.
The MDS Clinical Diagnostic Criteria for PD (2015) provide a standardized approach. [5]
MDS Clinical Diagnostic Criteria
Step 1: Establish Parkinsonism
- Bradykinesia (mandatory) PLUS
- Resting tremor and/or rigidity
Step 2: Apply Exclusion Criteria (Absolute; if present, PD is excluded)
- Cerebellar abnormalities
- Supranuclear downgaze palsy or selective slowing of downward saccades
- Probable behavioral variant frontotemporal dementia or primary progressive aphasia within first 5 years
- Parkinsonism restricted to lower limbs for > 3 years
- Treatment with dopamine receptor blockers or dopamine-depleting agents in a dose/time consistent with drug-induced parkinsonism
- Absence of observable response to high-dose levodopa despite at least moderate disease severity
- Unequivocal cortical sensory loss, clear limb ideomotor apraxia, or progressive aphasia
- Normal functional neuroimaging of presynaptic dopaminergic system (DaTscan)
- Documentation of alternative condition known to produce parkinsonism
Step 3: Count Supportive Criteria
- Clear and dramatic beneficial response to dopaminergic therapy
- Presence of levodopa-induced dyskinesia
- Rest tremor of a limb
- Olfactory loss or cardiac sympathetic denervation on MIBG scintigraphy
Step 4: Apply Red Flags (each red flag must be counterbalanced by a supportive criterion)
Diagnostic Certainty:
- Clinically Established PD: No absolute exclusion criteria, ≥2 supportive criteria, no red flags
- Clinically Probable PD: No absolute exclusion criteria, red flags counterbalanced by supportive criteria
Neuroimaging
1. Structural MRI Brain
| Purpose | Typical Findings |
|---|---|
| Rule out alternative diagnoses | Vascular disease, NPH, structural lesions |
| In idiopathic PD | Usually normal |
| MSA signs | "Hot cross bun" (pons); putaminal rim hyperintensity; cerebellar/pontine atrophy |
| PSP signs | "Hummingbird" or "penguin" sign (midbrain atrophy); "morning glory" sign (axial view) |
| CBS signs | Asymmetric cortical atrophy (frontoparietal) |
Advanced MRI techniques (research/specialized centers):
- SWI/Neuromelanin imaging: Loss of "swallow tail sign" in SNpc
- Diffusion tensor imaging: White matter tract changes
2. DaTscan (Ioflupane I-123 SPECT)
Assesses presynaptic dopamine transporter (DAT) density in the striatum. [14]
| Aspect | Details |
|---|---|
| Mechanism | I-123-ioflupane binds to presynaptic dopamine transporters |
| Normal appearance | Symmetric, comma-shaped uptake in both striata |
| PD appearance | Asymmetrically reduced uptake; "period" or "dot" instead of "comma" (putamen affected > caudate) |
| Clinical utility | Distinguishes degenerative parkinsonism (PD, MSA, PSP, DLB) from non-degenerative causes (essential tremor, drug-induced, psychogenic) |
| Limitation | Cannot distinguish between PD and Parkinson-plus syndromes (all show reduced uptake) |
| Indications | Diagnostic uncertainty between ET and PD; tremor-dominant cases; possible psychogenic parkinsonism |
3. Cardiac MIBG Scintigraphy
| Aspect | Details |
|---|---|
| Mechanism | I-123-MIBG is taken up by postganglionic sympathetic neurons |
| Normal | Symmetric cardiac uptake (heart:mediastinum ratio > 1.6) |
| In PD | Reduced cardiac uptake (postganglionic sympathetic denervation) |
| In MSA | Normal or mildly reduced (preganglionic lesion) |
| Utility | Helps distinguish PD/DLB (reduced) from MSA (preserved) |
Laboratory Investigations
Routine labs are not diagnostic but help exclude secondary causes:
| Test | Purpose |
|---|---|
| Ceruloplasmin, serum copper, 24-hour urine copper | Rule out Wilson's disease (mandatory in patients less than 50 years) |
| Thyroid function tests | Rule out hyperthyroidism (tremor) or hypothyroidism (motor slowing) |
| Vitamin B12 | Deficiency can cause peripheral neuropathy and cognitive changes |
| Comprehensive metabolic panel | General health assessment |
| MRI brain or CT head | Exclude structural lesions, NPH, vascular disease |
Genetic Testing
Consider in:
- Young-onset PD (less than 40-50 years)
- Strong family history
- Specific populations (Ashkenazi Jewish, North African Arab for LRRK2)
- Research or clinical trial eligibility
| Gene Panel | Comments |
|---|---|
| LRRK2 (G2019S) | Most common genetic cause; autosomal dominant; clinical trials available |
| GBA | Most common genetic risk factor; important for prognosis (faster progression) |
| PRKN, PINK1, DJ-1 | Recessive; consider in young-onset |
| SNCA | Rare; aggressive phenotype |
Additional Testing Based on Clinical Picture
| Scenario | Additional Investigations |
|---|---|
| Suspected MSA | Autonomic function tests, urodynamics, EMG of sphincter |
| Cognitive symptoms | Neuropsychological testing, MRI (hippocampal volume) |
| Atypical features | Lumbar puncture (CSF biomarkers in research settings) |
| Pre-DBS evaluation | Comprehensive neuropsychological testing, levodopa challenge, MRI, psychiatric evaluation |
7. Management
Principles of Management
Management of PD is symptomatic (no disease-modifying therapy is proven). Goals include:
- Maintain independence and quality of life
- Treat motor symptoms to acceptable patient-defined targets
- Recognize and manage non-motor symptoms
- Minimize treatment complications
- Plan for advanced disease
- Engage multidisciplinary team early
Treatment Initiation
When to start treatment: Treatment should begin when symptoms cause functional impairment or distress to the patient. There is no evidence that delaying treatment provides any protective benefit. [6,15]
INITIAL THERAPY DECISION ALGORITHM
PATIENT WITH NEWLY DIAGNOSED PD
(Bradykinesia + Tremor/Rigidity)
│
▼
┌───────────────────────────────┐
│ Are symptoms affecting │
│ quality of life or function? │
└───────────────────────────────┘
│ │
YES NO
│ │
▼ ▼
┌─────────────────┐ ┌─────────────────┐
│ Initiate │ │ Monitor; │
│ Pharmacotherapy │ │ Lifestyle │
│ │ │ modifications │
└─────────────────┘ │ (exercise) │
│ └─────────────────┘
▼
┌─────────────────────────────────────────────┐
│ CONSIDER PATIENT FACTORS: │
│ • Age │
│ • Severity of symptoms │
│ • Employment/activity demands │
│ • Cognitive status │
│ • Patient preference │
└─────────────────────────────────────────────┘
│
┌──────────┼──────────┐
│ │ │
▼ ▼ ▼
┌────────┐ ┌────────┐ ┌────────┐
│ YOUNG │ │ OLDER │ │ MILD │
│ (less than 60y) │ │ (> 70y) │ │ SYMPTS │
│ Active │ │ Cog. │ │ Any │
│ concern│ │ risk │ │ age │
│ for │ │ │ │ │
│ dyski- │ │ │ │ │
│ nesias │ │ │ │ │
└────────┘ └────────┘ └────────┘
│ │ │
▼ ▼ ▼
┌────────┐ ┌────────┐ ┌────────┐
│Dopamine│ │LEVODOPA│ │MAO-B │
│Agonist │ │(first- │ │Inhibitor│
│or │ │line if │ │ │
│MAO-B-I │ │QoL │ │Rasagi- │
│ │ │impact) │ │line │
└────────┘ └────────┘ └────────┘
Pharmacotherapy
1. Levodopa: The Gold Standard
Levodopa remains the most effective medication for motor symptoms of PD. [6,15]
| Aspect | Details |
|---|---|
| Mechanism | Dopamine precursor; crosses blood-brain barrier; converted to dopamine by aromatic L-amino acid decarboxylase (AADC) |
| Formulations | Carbidopa/Levodopa (Sinemet) or Benserazide/Levodopa (Madopar); peripheral decarboxylase inhibitor prevents peripheral conversion |
| Starting dose | 50mg levodopa TDS (with 12.5mg carbidopa) or equivalent |
| Titration | Increase by 50-100mg levodopa every 1-2 weeks to effect |
| Typical maintenance | 300-600mg/day in divided doses; some patients need 1000mg+ |
| Controlled-release | Sinemet CR, Madopar HBS — longer duration but less predictable absorption |
| Take timing | On empty stomach (30-60 min before meals) for best absorption; protein competes for absorption |
Adverse Effects:
| Category | Effects | Management |
|---|---|---|
| GI | Nausea, vomiting | Take with food (reduced absorption); domperidone (where available); ensure adequate carbidopa (> 75mg/day) |
| Cardiovascular | Orthostatic hypotension | Slow titration; hydration; compression stockings; midodrine/fludrocortisone if severe |
| Neuropsychiatric | Visual hallucinations, confusion (especially elderly) | Reduce dose; quetiapine or pimavanserin if needed |
| Motor complications | Wearing-off, dyskinesias (see below) | Adjust dosing; add adjunct therapies; consider advanced therapies |
| Dopamine dysregulation syndrome | Compulsive overuse of levodopa | Reduce access; behavioral intervention |
2. Dopamine Agonists
Directly stimulate dopamine receptors (D2/D3). [6,15]
| Agent | Half-life | Formulation | Notes |
|---|---|---|---|
| Pramipexole | 8-12 hours | IR (TDS), ER (once daily) | Common first-line agonist |
| Ropinirole | 6 hours | IR (TDS), ER (once daily) | Similar to pramipexole |
| Rotigotine | 5-7 hours | Transdermal patch (24h) | Useful if swallowing difficulty; continuous delivery |
| Apomorphine | 30-60 min | SC injection (rescue); SC infusion (continuous) | Potent; rapid onset; rescue for "off" periods |
Indications:
- Early monotherapy in younger patients (less than 60-65) to delay levodopa complications
- Adjunct to levodopa for motor fluctuations
- Apomorphine for refractory "off" periods
Adverse Effects:
| Effect | Details | Management |
|---|---|---|
| Impulse control disorders (ICDs) | Pathological gambling, hypersexuality, compulsive shopping, binge eating (10-20%); higher risk with higher doses and patient personality factors | SCREEN FOR ICDs REGULARLY; reduce/stop agonist; consider amantadine for dyskinesia offset |
| Dopamine agonist withdrawal syndrome | Anxiety, panic, depression, dysphoria, fatigue when stopping agonist | Taper slowly; warn patients |
| Somnolence/Sleep attacks | Sudden irresistible sleepiness (driving risk) | Warn about driving; may need to stop agonist |
| Peripheral edema | Lower limb swelling | Consider dose reduction; diuretics if needed |
| Hallucinations/Confusion | More common in elderly and cognitively impaired | Avoid agonists in these patients |
| Nausea | Similar to levodopa | Domperidone (not metoclopramide) |
| Ergot-specific (cabergoline, pergolide) | Valvular fibrosis, retroperitoneal fibrosis | Rarely used now; echocardiogram monitoring required |
[!CAUTION] Impulse Control Disorders — Dopamine agonists carry significant risk of ICDs. Screen all patients before starting and at each visit. Ask directly about gambling, excessive spending, hypersexuality, binge eating, and punding.
3. MAO-B Inhibitors
Inhibit monoamine oxidase type B, the enzyme that metabolizes dopamine in the brain. [6,15]
| Agent | Dose | Properties |
|---|---|---|
| Rasagiline | 1mg once daily | Irreversible MAO-B inhibitor; possible neuroprotective effect (inconclusive ADAGIO data) |
| Selegiline | 5mg BD or 10mg daily | Irreversible; metabolized to amphetamine derivatives (insomnia risk) |
| Safinamide | 50-100mg once daily | Reversible MAO-B inhibitor + glutamate modulation; used as adjunct |
Indications:
- Mild symptoms (can use as monotherapy)
- Adjunct to levodopa (extends duration, reduces "off" time)
Adverse Effects:
- Generally well tolerated
- Insomnia (selegiline > rasagiline)
- Headache
- Serotonin syndrome: Theoretical risk with SSRIs/SNRIs, though clinically rare at MAO-B selective doses
- Tyramine reaction: Very rare with MAO-B selective doses (dietary restriction unnecessary)
4. COMT Inhibitors
Block catechol-O-methyltransferase, reducing peripheral levodopa metabolism. [6,15]
| Agent | Dose | Properties |
|---|---|---|
| Entacapone | 200mg with each levodopa dose | Short-acting; commonly combined in Stalevo (levodopa/carbidopa/entacapone) |
| Opicapone | 50mg once daily | Long-acting; peripheral only; once-daily dosing |
| Tolcapone | 100-200mg TDS | Most potent; crosses BBB; requires LFT monitoring (hepatotoxicity risk) |
Indications:
- Wearing-off fluctuations (extends levodopa duration by 30-60 minutes per dose)
Adverse Effects:
- Dyskinesias (due to increased levodopa exposure — may need to reduce levodopa dose)
- Orange/brown discoloration of urine
- Diarrhea (common with entacapone; may be delayed onset)
- Hepatotoxicity (tolcapone — LFT monitoring required)
5. Amantadine
Originally an antiviral; mechanism in PD is complex (weak NMDA antagonist, dopamine release). [6]
| Indication | Details |
|---|---|
| Levodopa-induced dyskinesias | PRIMARY indication now; only oral medication proven to reduce dyskinesias |
| Early symptoms | Mild benefit on tremor and bradykinesia; effect may wane |
| Fatigue | May help |
Formulations:
- Immediate-release (100mg BID-TID)
- Extended-release (Gocovri, Osmolex): Specifically approved for dyskinesias; given at bedtime
Adverse Effects:
- Livedo reticularis (reticular purple discoloration of skin)
- Peripheral edema
- Hallucinations, confusion (especially in elderly — avoid if cognitive impairment)
- Anticholinergic effects
- Insomnia
6. Anticholinergics
Block muscarinic acetylcholine receptors; used for tremor. [6]
| Agent | Dose |
|---|---|
| Trihexyphenidyl (Benzhexol) | 1-2mg TDS |
| Benztropine | 0.5-2mg BD |
Indications: Tremor-dominant PD in young patients only
Adverse Effects: Cognitive impairment, confusion, hallucinations, urinary retention, constipation, dry mouth, blurred vision
[!CAUTION] Avoid anticholinergics in elderly patients — High risk of confusion, cognitive worsening, urinary retention, and falls. Use is limited to young, cognitively intact patients with tremor-dominant disease.
Motor Fluctuations and Dyskinesias
Motor complications develop in 40-50% of patients within 5 years of levodopa initiation and in up to 90% after 10 years. [8]
Types of Motor Fluctuations
| Type | Description | Mechanism |
|---|---|---|
| Wearing-off | Predictable return of symptoms before next dose (end-of-dose deterioration) | Short duration of levodopa response; reduced dopamine storage capacity |
| Delayed "on" | Slow or incomplete response to a dose | Impaired gastric emptying; protein competition |
| Dose failure | No response to a dose | Complete failure of absorption |
| On-off fluctuations | Unpredictable, sudden switches between good mobility and severe parkinsonism | Advanced disease; complex pharmacokinetics |
| Freezing of gait | Sudden inability to initiate or continue walking | Dopaminergic and non-dopaminergic; often worse in "off" |
Dyskinesias
| Type | Timing | Description | Management |
|---|---|---|---|
| Peak-dose dyskinesias | When levodopa levels are highest | Choreiform movements; usually not disabling initially | Reduce individual levodopa doses; add amantadine |
| Diphasic dyskinesias | At beginning and end of dose effect | Dystonic, repetitive, often stereotyped; affects legs | Difficult to treat; may benefit from more frequent levodopa |
| "Off" period dystonia | In "off" state, often early morning | Painful sustained contraction (often foot) | Morning controlled-release levodopa; apomorphine |
Management of Motor Fluctuations
WEARING-OFF MANAGEMENT ALGORITHM
Step 1: OPTIMIZE LEVODOPA
├── More frequent doses (smaller, more often)
├── Avoid taking with protein-rich meals
└── Consider controlled-release preparations
Step 2: ADD ADJUNCT THERAPY
├── COMT inhibitor (entacapone/opicapone)
│ → Extends levodopa duration by 30-60 min
├── MAO-B inhibitor (rasagiline/safinamide)
│ → Reduces "off" time by ~1 hour/day
└── Dopamine agonist
→ Longer half-life provides smoother effect
Step 3: ADDRESS DYSKINESIAS (if present)
├── Amantadine (especially extended-release)
├── Reduce levodopa dose (may worsen parkinsonism)
└── Consider advanced therapies
Step 4: CONSIDER ADVANCED THERAPIES
├── Deep brain stimulation
├── Levodopa-carbidopa intestinal gel (LCIG)
└── Subcutaneous apomorphine infusion
Advanced Therapies
For patients with motor fluctuations refractory to optimized oral therapy. [7]
Deep Brain Stimulation (DBS)
DBS is the most established advanced therapy for PD. [7]
Mechanism: High-frequency stimulation (typically 130-180 Hz) of deep brain structures disrupts pathological oscillatory activity and reduces output from the GPi or modulates STN function.
Targets:
- Subthalamic nucleus (STN): Most common; allows reduction in medication
- Globus pallidus interna (GPi): Preferred if significant dyskinesias; cognitive concerns
- Ventral intermediate nucleus of thalamus (VIM): Tremor only (rarely used in PD)
Indications:
- Motor fluctuations and dyskinesias not adequately controlled with optimized medical therapy
- Good response to levodopa (predicts DBS success)
- Disabling tremor unresponsive to medication
Contraindications:
- Significant cognitive impairment or dementia
- Uncontrolled psychiatric illness
- Medical comorbidities precluding surgery
- Parkinsonism not responsive to levodopa (suggests atypical parkinsonism)
- Advanced age is relative (no strict cutoff, but benefits diminish > 70-75 years)
Landmark Evidence:
- EARLYSTIM Trial (2013): DBS superior to best medical therapy in patients with early motor complications (mean disease duration 7.5 years); better quality of life at 2 years. [7]
| Outcome | DBS Benefits |
|---|---|
| "Off" time | Reduced by 2-4 hours/day |
| Dyskinesias | Reduced by 60-80% |
| Quality of life | Significantly improved |
| Medication reduction | 50-70% reduction in levodopa equivalent dose |
| Motor scores | 30-50% improvement in UPDRS motor scores |
Adverse Effects/Complications:
- Surgical: Hemorrhage (1-2%), infection (3-5%), lead malposition
- Stimulation-related: Dysarthria, paresthesias, mood changes, weight gain
- Hardware: Lead fracture, battery replacement (3-5 years for non-rechargeable)
What DBS Does NOT Improve:
- Cognitive symptoms (may worsen verbal fluency)
- Axial symptoms (gait freezing, postural instability) — often less responsive
- Non-motor symptoms
- "Off" symptoms that do not respond to levodopa
Levodopa-Carbidopa Intestinal Gel (LCIG/Duodopa)
Mechanism: Continuous intestinal infusion of levodopa gel via PEG-J tube directly into jejunum, bypassing variable gastric emptying.
| Aspect | Details |
|---|---|
| Indication | Severe motor fluctuations not responding to oral therapy |
| Delivery | Portable pump connected to PEG-J tube; 16-hour daytime infusion |
| Benefits | Reduces "off" time by 2-4 hours/day; reduces dyskinesias; continuous dopaminergic stimulation |
| Complications | PEG-J site problems (infection, granulation, dislocation); device issues; polyneuropathy (B12/B6 monitoring needed) |
Subcutaneous Apomorphine
Mechanism: Potent dopamine agonist delivered continuously via subcutaneous infusion.
| Aspect | Details |
|---|---|
| Indications | Motor fluctuations; alternative to DBS/LCIG |
| Delivery | Continuous SC infusion via portable pump |
| Benefits | Reduces "off" time; reduces levodopa dose |
| Adverse effects | SC nodules, skin reactions; neuropsychiatric (hallucinations, impulse control disorders); requires anti-emetic pretreatment |
Non-Motor Symptom Management
| Symptom | First-Line | Second-Line/Notes |
|---|---|---|
| Depression | SSRIs (sertraline, citalopram); SNRIs (venlafaxine) | TCAs (nortriptyline); dopamine agonists may help; ECT in severe cases |
| Anxiety | SSRIs/SNRIs; consider adjustment of dopaminergic therapy (anxiety often correlates with "off" periods) | Buspirone; benzodiazepines (short-term only) |
| Psychosis/Hallucinations | Remove offending drugs (anticholinergics > agonists > MAO-B > levodopa); quetiapine (off-label); pimavanserin (FDA-approved for PD psychosis) | Clozapine (efficacy proven but requires monitoring); avoid typical antipsychotics (worsen parkinsonism) |
| Dementia | Rivastigmine (cholinesterase inhibitor — only one proven for PD dementia) | Donepezil, galantamine (less evidence); avoid anticholinergics |
| Orthostatic hypotension | Non-pharmacological first (compression stockings, increased salt/water, rise slowly); midodrine; fludrocortisone; droxidopa | Reduce antihypertensives if possible; pyridostigmine |
| Constipation | Polyethylene glycol (Movicol/MiraLax); increased fiber and fluids; exercise | Lubiprostone; prucalopride |
| Sialorrhea (drooling) | Glycopyrrolate; botulinum toxin injection to salivary glands | Atropine sublingual drops |
| REM sleep behavior disorder | Melatonin (3-12mg at bedtime) | Clonazepam (0.5-2mg at bedtime) — use cautiously in elderly |
| Excessive daytime sleepiness | Optimize nighttime sleep; modafinil | Caffeine; review medications causing sedation |
| Pain | Optimize dopaminergic therapy (pain often correlates with "off" periods); analgesics | Duloxetine for neuropathic pain; onabotulinumtoxinA for dystonic pain |
Multidisciplinary Care
Optimal PD management requires a team approach:
| Discipline | Role |
|---|---|
| Neurologist/Movement disorders specialist | Diagnosis, medication management, advanced therapy decisions |
| PD nurse specialist | Education, monitoring, medication adjustment, patient support |
| Physiotherapist | Gait training, balance, exercise prescription, falls prevention |
| Occupational therapist | Home safety assessment, aids, adaptations, energy conservation |
| Speech and language therapist | Swallowing assessment, LSVT LOUD for hypophonia |
| Psychiatrist/Psychologist | Management of depression, anxiety, impulse control disorders, cognitive assessment |
| Dietitian | Nutrition, weight management, protein timing, constipation |
| Social worker | Carer support, benefits, care planning |
| Palliative care | Symptom management in advanced disease, advance care planning |
Exercise and Rehabilitation
Exercise is the only intervention with possible disease-modifying effects. [11]
| Type | Benefits | Examples |
|---|---|---|
| Aerobic exercise | Cardiovascular fitness, possible neuroprotection, improved motor function | Cycling, treadmill, swimming |
| Resistance training | Strength maintenance, mobility | Weight training, resistance bands |
| Balance training | Falls prevention | Tai chi, yoga, specific balance exercises |
| Amplitude-based training | Improved bradykinesia, hypomimia, hypophonia | LSVT BIG (physical therapy), LSVT LOUD (speech therapy) |
| Dance | Motor function, balance, quality of life | Tango, other dance forms |
| Boxing training | Non-contact boxing; motor function | Rock Steady Boxing |
Recommendation: 150 minutes/week of moderate-intensity exercise; include aerobic, strength, and balance components.
8. Complications
Motor Complications
As discussed above, motor fluctuations and dyskinesias are major complications of long-term levodopa therapy.
Risk factors for motor complications:
- Younger age at onset
- Higher levodopa doses
- Longer disease duration
- More severe disease
- Low body weight
Falls and Fractures
- Falls occur in 60-80% of patients during the disease course
- Leading cause of morbidity; hip fractures significantly increase mortality
- Multifactorial: postural instability, freezing of gait, orthostatic hypotension, cognitive impairment
- Prevention: physiotherapy, home assessment, medication review, treatment of OH
Aspiration Pneumonia
- Leading cause of death in PD
- Due to dysphagia (oropharyngeal) and reduced cough reflex
- Regular swallowing assessments; modified diet texture; speech therapy
Parkinson's Disease Dementia
- Point prevalence ~30-40%; cumulative risk up to 80% at 20 years
- Characteristically affects executive function, attention, and visuospatial skills (differs from Alzheimer's pattern)
- Visual hallucinations are common
- Rivastigmine is the only medication with proven efficacy [16]
Parkinson's Disease Psychosis
- Hallucinations (usually visual, well-formed) and delusions
- Continuum from "benign" insight-preserved hallucinations to delusional psychosis
- Drug-related (dopaminergic medications) but also intrinsic to disease
- Management: reduce offending medications; pimavanserin; quetiapine; clozapine
Impulse Control Disorders
- Pathological gambling, hypersexuality, compulsive shopping, binge eating, punding (stereotyped repetitive behaviors), hobbyism
- Strongly associated with dopamine agonist use; also occurs with levodopa
- Screen all patients at each visit; reduce or stop agonist; behavioral therapy
9. Prognosis
Disease Progression
- PD is slowly progressive over 10-25+ years
- Rate of progression varies considerably between individuals
- Factors associated with faster progression: older age at onset, akinetic-rigid phenotype, early cognitive impairment, minimal tremor
Hoehn and Yahr Staging
| Stage | Description | Typical Timeline |
|---|---|---|
| 1 | Unilateral involvement only | Diagnosis |
| 2 | Bilateral involvement without impairment of balance | 2-7 years |
| 3 | Mild to moderate bilateral disease; some postural instability; physically independent | 5-10 years |
| 4 | Severe disability; still able to walk or stand unassisted | 10-15 years |
| 5 | Wheelchair-bound or bedridden unless aided | Advanced disease |
Life Expectancy
- Modestly reduced compared to general population
- Mortality ratio ~1.5-2.0
- Primary causes of death:
- Aspiration pneumonia (most common)
- Falls and complications
- Cardiovascular disease
- Advanced dementia with complications
Predictors of Prognosis
| Better Prognosis | Worse Prognosis |
|---|---|
| Younger age at onset | Older age at onset |
| Tremor-dominant phenotype | Akinetic-rigid (PIGD) phenotype |
| Good levodopa response | Poor levodopa response |
| Absence of early cognitive impairment | Early cognitive impairment |
| Absence of early autonomic failure | Early autonomic failure |
| Asymmetric onset | Symmetric onset |
10. Evidence Summary
Landmark Trials
| Trial | Year | Key Finding |
|---|---|---|
| ELLDOPA | 2004 | Levodopa effective for symptoms; dyskinesias dose-dependent; no clear disease modification. [17] |
| PD MED | 2014 | In early PD, levodopa provides better quality of life than dopamine agonists or MAO-B inhibitors as initial therapy; no difference in motor complications at 7 years. [6] |
| EARLYSTIM | 2013 | DBS of STN superior to best medical therapy in patients with early motor complications (mean disease duration 7.5 years); significantly better quality of life. [7] |
| ADAGIO | 2009 | Rasagiline 1mg/day showed potential disease-modifying effect (delayed need for symptomatic therapy); higher dose (2mg) did not show same effect; results inconclusive. [18] |
| PRESTO/TEMPO | 2005 | Rasagiline reduces "off" time as adjunct to levodopa. |
| EXPRESS | 2007 | Rotigotine patch effective for motor fluctuations. |
| RECOVER | 2011 | Rotigotine patch improves early morning motor function and sleep. |
Major Guidelines
| Guideline | Key Recommendations |
|---|---|
| NICE NG71 (2017, updated 2023) | Levodopa first-line for those whose motor symptoms impact quality of life; dopamine agonists or MAO-B inhibitors alternative if milder symptoms or patient preference; offer DBS for refractory motor complications; multidisciplinary care; regular review of non-motor symptoms. [15] |
| MDS Evidence-Based Medicine Review (2018, 2019) | Comprehensive efficacy and safety ratings for all PD therapies — motor and non-motor. [6,13] |
| AAN Guidelines | Management of motor fluctuations and non-motor symptoms. |
| European Academy of Neurology | Diagnosis, early treatment, non-motor management. |
Emerging Therapies and Future Directions
| Approach | Status | Notes |
|---|---|---|
| α-synuclein immunotherapy | Phase 2 trials | Antibodies targeting extracellular α-synuclein; mixed results so far |
| GBA-targeting therapies | Phase 2 trials | Small molecules enhancing glucocerebrosidase activity for GBA-PD |
| LRRK2 inhibitors | Phase 1-2 trials | For LRRK2-associated PD |
| Gene therapy | Phase 1-2 trials | AAV-mediated delivery of GAD to STN; aromatic L-amino acid decarboxylase |
| Cell replacement therapy | Phase 1 trials | Stem cell-derived dopamine neuron transplantation |
| Focused ultrasound | FDA-approved for tremor | Unilateral thalamotomy; approved for tremor-dominant PD |
11. Patient Education
Key Counseling Points
- Chronic, progressive condition — Manage expectations; focus on maintaining function and quality of life
- Medication timing is crucial — Levodopa works best on empty stomach; timing matters as disease progresses
- Non-motor symptoms are real — Depression, anxiety, constipation, and fatigue are part of the disease
- Impulse control — Warn patients and families about ICDs (gambling, shopping, hypersexuality) with dopamine agonists
- Exercise is medicine — Regular exercise may slow functional decline
- Driving — Discuss driving safety; patients must notify relevant authorities (country-specific requirements)
- Advanced care planning — Consider preferences for advanced therapies and end-of-life care
Driving Considerations
- Must notify driving authority (DVLA in UK; equivalent elsewhere)
- Fitness to drive depends on motor and cognitive function
- Individual assessment required
- Sleep attacks (especially with dopamine agonists) are a specific concern
Support Resources
| Organization | Region |
|---|---|
| Parkinson's UK | United Kingdom |
| Parkinson's Foundation | United States |
| Michael J. Fox Foundation | United States/International |
| European Parkinson's Disease Association | Europe |
| Parkinson's Australia | Australia |
| Parkinson Society Canada | Canada |
Allied Health Programs
| Program | Description |
|---|---|
| LSVT LOUD | Intensive speech therapy program for hypophonia; 16 sessions over 1 month |
| LSVT BIG | Intensive physical therapy program for bradykinesia; amplitude-focused |
| PWR! Moves | Physical therapy program designed for PD |
| Rock Steady Boxing | Non-contact boxing training for motor symptoms |
| Dance for PD | Dance-based exercise program |
12. Examination Focus
The "Opening Statement" for a PD Case
"This patient presents with an asymmetric, 4-6 Hz resting tremor of the right hand with a pill-rolling component, associated with cogwheel rigidity and bradykinesia demonstrated by decremental finger tapping. The eye movements are normal, there are no cerebellar signs, and there is no evidence of early autonomic failure. The unilateral onset, resting tremor, and presence of bradykinesia are consistent with the clinical diagnosis of idiopathic Parkinson's disease. I would expect this patient to respond well to levodopa therapy."
Common Viva Questions and Model Answers
1. How do you distinguish Parkinson's disease from Essential Tremor?
| Feature | Parkinson's Disease | Essential Tremor |
|---|---|---|
| Tremor type | Resting | Action/Postural |
| Frequency | 4-6 Hz | 8-12 Hz |
| Onset | Asymmetric | Symmetric |
| Bradykinesia | Present (required) | Absent |
| Head/voice | Rare | Common |
| Alcohol | No effect | Improves |
| DaTscan | Abnormal | Normal |
2. What are the red flags for atypical parkinsonism?
- Early falls (PSP)
- Early severe autonomic failure (MSA)
- Supranuclear gaze palsy, especially downgaze (PSP)
- Poor levodopa response
- Symmetric onset
- Early dementia (DLB)
- Cerebellar signs (MSA-C)
3. What is the mechanism of levodopa? Levodopa is a dopamine precursor that crosses the blood-brain barrier (dopamine itself cannot). It is converted to dopamine by aromatic L-amino acid decarboxylase (AADC) in the brain. It is combined with a peripheral decarboxylase inhibitor (carbidopa or benserazide) to prevent peripheral conversion, reducing side effects (nausea, hypotension) and increasing CNS bioavailability.
4. Why do motor complications develop with levodopa?
- Pharmacokinetic factors: Short half-life of levodopa (90 minutes) leads to pulsatile dopamine receptor stimulation
- Pharmacodynamic factors: Loss of dopamine storage capacity in remaining neurons; postsynaptic receptor changes
- Disease progression: Fewer dopaminergic neurons to buffer fluctuations in dopamine levels
5. What are the indications for DBS in Parkinson's disease?
- Motor fluctuations and/or dyskinesias not adequately controlled with optimized oral therapy
- Good response to levodopa (predictor of DBS success)
- Medically fit for surgery
- No significant dementia or uncontrolled psychiatric illness
- Typically offered earlier in the course of motor complications (EARLYSTIM evidence)
6. What are the impulse control disorders, and how do you screen for them? ICDs include pathological gambling, hypersexuality, compulsive shopping, binge eating, and punding. Screen by asking directly: "Have you noticed any changes in behavior? Any new gambling, spending money, or sexual behaviors? Any repetitive activities like organizing or collecting?" More common with dopamine agonists but can occur with any dopaminergic therapy.
Common Mistakes to Avoid
- Diagnosing PD without bradykinesia — Tremor alone is NOT Parkinson's
- Missing the eyes — Always examine vertical gaze to rule out PSP
- Forgetting medication history — Always ask about dopamine blockers (antipsychotics, metoclopramide, prochlorperazine)
- Overlooking non-motor symptoms — Mention them in your management plan ("I would also screen for depression, sleep disorders, and cognitive impairment")
- Neglecting impulse control disorder screening — Essential when dopamine agonists are prescribed
- Forgetting the whole patient — PD management is multidisciplinary; mention physiotherapy, SALT, occupational therapy, Parkinson's nurse
13. Summary Tables
Quick Reference: Pharmacotherapy
| Drug Class | Examples | Primary Indication | Key Side Effect |
|---|---|---|---|
| Levodopa | Sinemet, Madopar | Most effective for motor symptoms | Motor complications (long-term) |
| Dopamine Agonists | Pramipexole, Ropinirole, Rotigotine | Young onset; adjunct for fluctuations | Impulse control disorders |
| MAO-B Inhibitors | Rasagiline, Selegiline, Safinamide | Mild symptoms; adjunct | Insomnia (selegiline) |
| COMT Inhibitors | Entacapone, Opicapone, Tolcapone | Wearing-off fluctuations | Diarrhea; dyskinesias |
| Amantadine | Symmetrel, Gocovri | Dyskinesias | Confusion (elderly) |
| Anticholinergics | Trihexyphenidyl | Tremor (young patients only) | Cognitive impairment |
Quick Reference: Motor Complications Management
| Problem | Solutions |
|---|---|
| Wearing-off | More frequent levodopa; COMT inhibitor; MAO-B inhibitor; dopamine agonist |
| Peak-dose dyskinesias | Reduce levodopa dose; add amantadine; consider DBS |
| Diphasic dyskinesias | More frequent levodopa; may need advanced therapy |
| Off-period dystonia | Morning controlled-release levodopa; apomorphine |
| Unpredictable fluctuations | Advanced therapies (DBS, LCIG, apomorphine pump) |
14. References
-
Kalia LV, Lang AE. Parkinson's disease. Lancet. 2015;386(9996):896-912. DOI: 10.1016/S0140-6736(14)61393-3. PMID: 25904358.
-
Poewe W, Seppi K, Tanner CM, et al. Parkinson disease. Nat Rev Dis Primers. 2017;3:17013. DOI: 10.1038/nrdp.2017.13. PMID: 28332491.
-
Dorsey ER, Sherer T, Okun MS, Bloem BR. The Emerging Evidence of the Parkinson Pandemic. J Parkinsons Dis. 2018;8(s1):S3-S8. DOI: 10.3233/JPD-181474. PMID: 30584159.
-
Bloem BR, Okun MS, Klein C. Parkinson's disease. Lancet. 2021;397(10291):2284-2303. DOI: 10.1016/S0140-6736(21)00218-X. PMID: 33848468.
-
Postuma RB, Berg D, Stern M, et al. MDS clinical diagnostic criteria for Parkinson's disease. Mov Disord. 2015;30(12):1591-1601. DOI: 10.1002/mds.26424. PMID: 26474316.
-
Fox SH, Katzenschlager R, Lim SY, et al. International Parkinson and Movement Disorder Society Evidence-Based Medicine Review: Update on treatments for the motor symptoms of Parkinson's disease. Mov Disord. 2018;33(8):1248-1266. DOI: 10.1002/mds.27372. PMID: 29570866.
-
Schuepbach WM, Rau J, Knudsen K, et al. Neurostimulation for Parkinson's disease with early motor complications. N Engl J Med. 2013;368(7):610-622. DOI: 10.1056/NEJMoa1205158. PMID: 23406026.
-
Olanow CW, Stocchi F. Levodopa: A new look at an old friend. Mov Disord. 2018;33(6):859-866. DOI: 10.1002/mds.27216. PMID: 29150893.
-
Simon DK, Tanner CM, Brundin P. Parkinson Disease Epidemiology, Pathology, Genetics, and Pathophysiology. Clin Geriatr Med. 2020;36(1):1-12. DOI: 10.1016/j.cger.2019.08.002. PMID: 31733690.
-
Blauwendraat C, Nalls MA, Singleton AB. The genetic architecture of Parkinson's disease. Lancet Neurol. 2020;19(2):170-178. DOI: 10.1016/S1474-4422(19)30287-X. PMID: 31521533.
-
Schenkman M, Moore CG, Kohrt WM, et al. Effect of High-Intensity Treadmill Exercise on Motor Symptoms in Patients With De Novo Parkinson Disease: A Phase 2 Randomized Clinical Trial. JAMA Neurol. 2018;75(2):219-226. DOI: 10.1001/jamaneurol.2017.3517. PMID: 29228079.
-
Braak H, Del Tredici K, Rüb U, et al. Staging of brain pathology related to sporadic Parkinson's disease. Neurobiol Aging. 2003;24(2):197-211. DOI: 10.1016/s0197-4580(02)00065-9. PMID: 12498954.
-
Seppi K, Ray Chaudhuri K, Coelho M, et al. Update on treatments for nonmotor symptoms of Parkinson's disease-an evidence-based medicine review. Mov Disord. 2019;34(2):180-198. DOI: 10.1002/mds.27602. PMID: 30653247.
-
Ba F, Martin WRW. Dopamine transporter imaging as a diagnostic tool for parkinsonism and related disorders in clinical practice. Parkinsonism Relat Disord. 2015;21(2):87-94. DOI: 10.1016/j.parkreldis.2014.11.007. PMID: 25487729.
-
National Institute for Health and Care Excellence. Parkinson's disease in adults: diagnosis and management. [NG71]. NICE, 2017 (updated 2023). Available at: https://www.nice.org.uk/guidance/ng71.
-
Emre M, Aarsland D, Albanese A, et al. Rivastigmine for dementia associated with Parkinson's disease. N Engl J Med. 2004;351(24):2509-2518. DOI: 10.1056/NEJMoa041470. PMID: 15590953.
-
Fahn S, Oakes D, Shoulson I, et al.; Parkinson Study Group. Levodopa and the progression of Parkinson's disease. N Engl J Med. 2004;351(24):2498-2508. DOI: 10.1056/NEJMoa033447. PMID: 15590952.
-
Olanow CW, Rascol O, Hauser R, et al.; ADAGIO Study Investigators. A double-blind, delayed-start trial of rasagiline in Parkinson's disease. N Engl J Med. 2009;361(13):1268-1278. DOI: 10.1056/NEJMoa0809335. PMID: 19776408.
-
Armstrong MJ, Okun MS. Diagnosis and Treatment of Parkinson Disease: A Review. JAMA. 2020;323(6):548-560. DOI: 10.1001/jama.2019.22360. PMID: 32044947.
-
PD MED Collaborative Group. Long-term effectiveness of dopamine agonists and monoamine oxidase B inhibitors compared with levodopa as initial treatment for Parkinson's disease (PD MED): a large, open-label, pragmatic randomised trial. Lancet. 2014;384(9949):1196-1205. DOI: 10.1016/S0140-6736(14)60683-8. PMID: 24928805.
-
Weintraub D, David AS, Evans AH, Grant JE, Stacy M. Clinical spectrum of impulse control disorders in Parkinson's disease. Mov Disord. 2015;30(2):121-127. DOI: 10.1002/mds.26016. PMID: 25370355.
-
Deuschl G, Schade-Brittinger C, Krack P, et al.; German Parkinson Study Group, Neurostimulation Section. A randomized trial of deep-brain stimulation for Parkinson's disease. N Engl J Med. 2006;355(9):896-908. DOI: 10.1056/NEJMoa060281. PMID: 16943402.
Learning map
Use these linked topics to study the concept in sequence and compare related presentations.
Prerequisites
Start here if you need the foundation before this topic.
- Basal Ganglia Anatomy and Physiology
- Dopamine Neurotransmission
- Movement Disorder Examination
Differentials
Competing diagnoses and look-alikes to compare.
- Essential Tremor
- Progressive Supranuclear Palsy
- Multiple System Atrophy
- Drug-Induced Parkinsonism
- Dementia with Lewy Bodies
- Corticobasal Syndrome
- Vascular Parkinsonism
Consequences
Complications and downstream problems to keep in mind.
- Parkinson's Disease Dementia
- Levodopa-Induced Dyskinesias
- Freezing of Gait