Parkinson's Disease
Parkinson's disease (PD) is a progressive neurodegenerative disorder characterised by the loss of dopaminergic neurons i... MRCP exam preparation.
What matters first
Parkinson's disease (PD) is a progressive neurodegenerative disorder characterised by the loss of dopaminergic neurons i... MRCP exam preparation.
Early falls (within first year — suggests atypical parkinsonism)
22 Dec 2025
Generated educational material; verify before clinical use.
Visible references section
See the concept before reading it
Study the key anatomy, imaging, and decision pathways as full teaching plates.
Clinical board
A visual summary of the highest-yield teaching signals on this page.
Urgent signals
Safety-critical features pulled from the topic metadata.
- Early falls (within first year — suggests atypical parkinsonism)
- Early severe autonomic failure (MSA)
- Poor or absent levodopa response (atypical parkinsonism)
- Rapid progression (doubling disability within 2-3 years)
Exam focus
Current exam surfaces linked to this topic.
- MRCP
Linked comparisons
Differentials and adjacent topics worth opening next.
- Essential Tremor
- Multiple System Atrophy
Content status and exam context
This page is AI-generated educational content. It may contain errors or omissions and is not a substitute for current guidelines, local protocols, senior clinical judgement, or professional medical advice.
MedVellum does not claim an individual clinician reviewer, board certification, or professional credential for this page unless a future version names a real, verifiable reviewer.
Clinical explanation and evidence
Parkinson's Disease
1. Topic Overview
Summary
Parkinson's disease (PD) is a progressive neurodegenerative disorder characterised by the loss of dopaminergic neurons in the substantia nigra pars compacta and the accumulation of α-synuclein-containing Lewy bodies. [1] It is the second most common neurodegenerative disorder after Alzheimer's disease, affecting approximately 1% of adults over 60 years of age and representing the fastest-growing neurological condition globally. [2]
The clinical hallmark is parkinsonism: the combination of bradykinesia (which must be present for diagnosis) with either rest tremor, rigidity, or both. [3] However, PD is now recognised as a multisystem disorder with prominent non-motor features including anosmia, constipation, REM sleep behaviour disorder (RBD), depression, anxiety, autonomic dysfunction, and cognitive impairment — many of which precede motor symptoms by years or decades. [4]
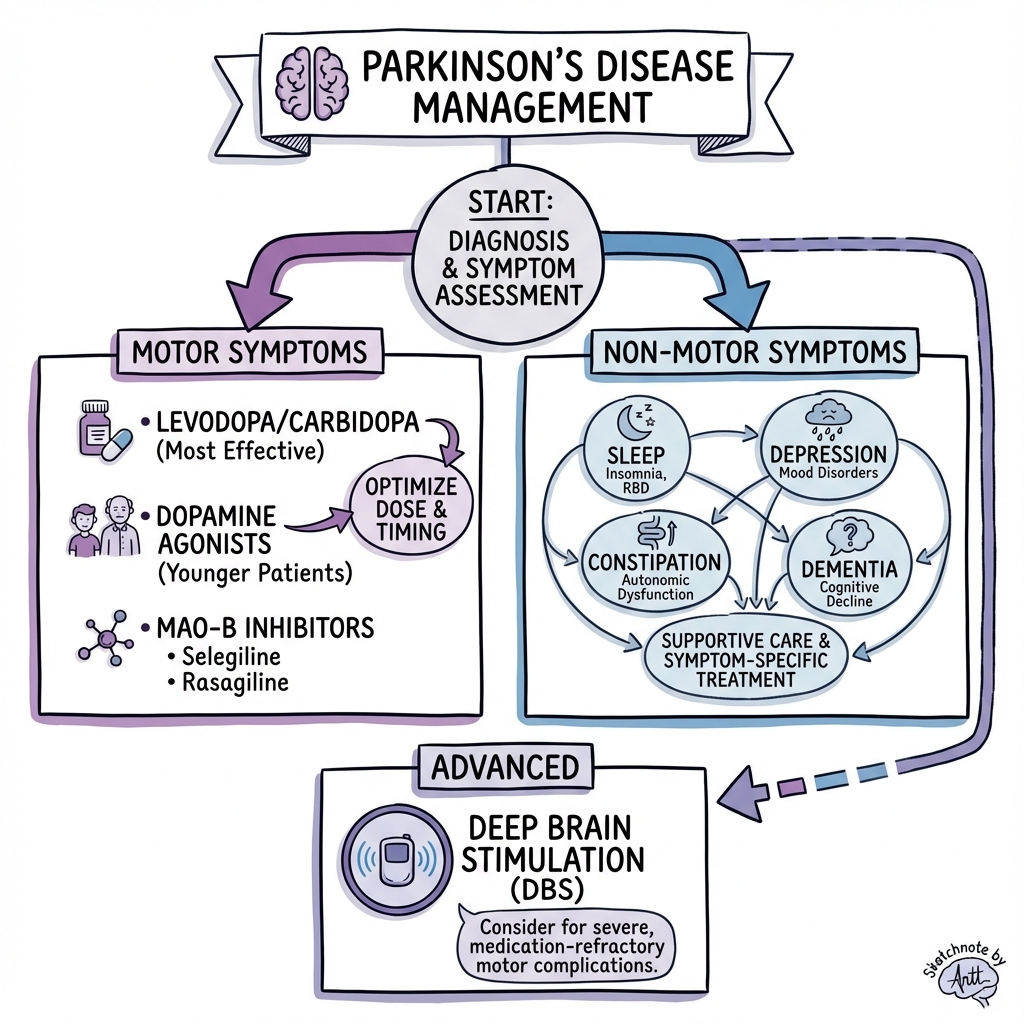
Diagnosis remains clinical, based on the Movement Disorder Society (MDS) Clinical Diagnostic Criteria. Treatment is symptomatic, with levodopa being the most effective agent for motor symptoms. Dopamine agonists, MAO-B inhibitors, and COMT inhibitors provide additional options. Advanced therapies including deep brain stimulation (DBS), levodopa-carbidopa intestinal gel (LCIG), and apomorphine infusions are available for patients with motor fluctuations refractory to oral medications. [5] Management requires a multidisciplinary approach addressing motor symptoms, non-motor features, falls prevention, and caregiver support.
Key Facts Card
| Domain | Key Information |
|---|---|
| Definition | Progressive neurodegenerative disorder due to dopaminergic neuron loss in substantia nigra pars compacta with Lewy body pathology |
| Prevalence | 1-2 per 1,000; ~1% of those > 60 years; ~6 million affected worldwide |
| Incidence | 10-20 per 100,000 per year; increasing with ageing populations |
| Male:Female | 1.5:1 |
| Mean onset | 60-65 years; 5-10% young-onset (less than 50 years) |
| Diagnostic criteria | MDS criteria: Bradykinesia PLUS rest tremor or rigidity |
| Hallmark pathology | Lewy bodies (α-synuclein aggregates) |
| First-line treatment | Levodopa with dopa decarboxylase inhibitor (carbidopa/benserazide) |
| Specialist referral | ALL patients with suspected PD should be referred untreated to a specialist |
Clinical Pearls
"Bradykinesia is Essential": You CANNOT diagnose Parkinson's disease without bradykinesia. It must be present along with at least one of: rest tremor or rigidity. Bradykinesia is not simply slowness — it requires progressive reduction in amplitude AND speed with repetitive movements (decrement). [3]
Asymmetry is a Hallmark: PD classically starts unilaterally and remains asymmetric even in advanced stages. Symmetric onset or early bilateral disease (within 3 years) should raise suspicion of atypical parkinsonism. [1]
Non-Motor Features Precede Motor Symptoms: The prodromal phase of PD can last 10-20 years. Anosmia, constipation, REM sleep behaviour disorder (RBD), depression, and anxiety often precede motor symptoms — and RBD carries an 80-90% risk of conversion to an α-synucleinopathy. [4,6]
Levodopa Response is Diagnostic: A clear, sustained response to adequate levodopa doses is a supportive criterion for PD diagnosis. Poor or absent levodopa response (despite adequate doses/duration) is an exclusion criterion and suggests atypical parkinsonism. [3]
Falls: Timing Matters: Falls within the first year of diagnosis are a red flag for atypical parkinsonism (especially PSP). In idiopathic PD, falls typically occur in moderate-to-advanced disease. [7]
Why This Matters Clinically
PD affects not just the patient but entire families. Early recognition enables timely specialist referral, accurate diagnosis (distinguishing PD from mimics), and initiation of appropriate symptomatic treatment — significantly improving quality of life. Understanding motor complications (wearing off, dyskinesias) allows proactive management adjustments. Recognising non-motor symptoms prevents undertreatment of depression, psychosis, autonomic dysfunction, and cognitive impairment. Finally, falls prevention through physiotherapy, home assessment, and medication optimisation reduces morbidity, hospitalisation, and mortality. [8]
2. Epidemiology
Incidence and Prevalence
| Parameter | Value | Notes | Ref |
|---|---|---|---|
| Global prevalence | ~6.1 million | Doubled since 1990 | [2] |
| Age-adjusted prevalence | 1-2 per 1,000 | General population | [1] |
| Prevalence > 60 years | ~1% | Rises steeply with age | [2] |
| Prevalence > 80 years | 3-4% | Highest in oldest old | [2] |
| Global incidence | 10-20 per 100,000/year | Standardised | [1] |
| Projected 2040 | > 12 million | The "Parkinson's Pandemic" | [2] |
PD is the fastest-growing neurological disorder in terms of prevalence, disability, and deaths — primarily driven by population ageing. [2]
Demographics
| Factor | Details |
|---|---|
| Age | Mean onset 60-65 years; risk doubles every decade after 60 |
| Young-onset PD | 5-10% of cases; onset less than 50 years; higher genetic contribution |
| Sex | Male predominance 1.5:1; possibly oestrogen-protective |
| Ethnicity | Higher in Caucasian/Hispanic populations; possibly lower in Asian/African populations (though may reflect ascertainment bias) |
| Geography | Higher in industrialised nations and pesticide-exposed regions |
Risk Factors
| Risk Factor | Relative Risk | Evidence | Notes |
|---|---|---|---|
| Age | Strongest factor | Level I | Risk doubles each decade after 60 |
| Male sex | 1.5x | Level I | Consistent across studies |
| Family history | 2-3x first-degree | Level II | 5-10% have affected relative |
| LRRK2 mutation | Variable (20-75% penetrance) | Level II | Most common genetic cause |
| GBA mutation | 5-8x | Level II | Strong genetic risk factor |
| Pesticide exposure | 1.5-2x | Level II | Paraquat, rotenone, organochlorines |
| Rural living/well water | 1.3-1.5x | Level III | Possible pesticide contamination |
| Head injury | 1.3-1.5x | Level II | Controversial; dose-response suggested |
| Melanoma | Bidirectional | Level II | Shared α-synuclein pathology? |
Protective Factors
| Factor | Relative Risk | Notes |
|---|---|---|
| Smoking | 0.4-0.6x | Inverse dose-response; mechanism unclear |
| Caffeine/Coffee | 0.6-0.7x | Adenosine antagonism may be protective |
| Physical activity | 0.6-0.7x | Neuroprotective; also symptomatic benefit |
| Urate levels (higher) | 0.5-0.7x | Antioxidant effects; intervention trials negative |
| NSAIDs | 0.7-0.8x | Anti-inflammatory mechanism; ibuprofen |
Genetic Factors
| Gene | Inheritance | Phenotype | Notes |
|---|---|---|---|
| SNCA | AD | Early-onset, rapid progression | α-synuclein gene; point mutations and multiplications |
| LRRK2 | AD | Classic PD; late-onset | Most common cause of autosomal dominant PD; G2019S common |
| GBA | Risk factor | Classic PD; higher dementia risk | Glucocerebrosidase; heterozygous carriers |
| PARK2 (Parkin) | AR | Young-onset; slow progression | Most common cause of young-onset AR PD |
| PINK1 | AR | Young-onset | Mitochondrial dysfunction |
| DJ-1 | AR | Young-onset | Rare |
| VPS35 | AD | Classic PD | Rare |
Exam Detail: Genetic Testing Indications:
- Young-onset PD (less than 40-50 years)
- Multiple affected family members
- Ashkenazi Jewish ancestry (LRRK2 G2019S, GBA)
- Research purposes or clinical trials
GBA Mutations:
- Present in 5-15% of PD patients
- Associated with earlier onset, faster progression, higher dementia risk
- Glucocerebrosidase enzyme dysfunction leads to lysosomal impairment and α-synuclein accumulation
3. Pathophysiology
Neuroanatomy of the Basal Ganglia
Normal Function:
The basal ganglia circuit modulates movement through the balance of direct and indirect pathways:
| Pathway | Effect | Neurotransmitters |
|---|---|---|
| Direct pathway | Facilitates movement | Dopamine D1 receptors (excitatory on pathway) |
| Indirect pathway | Inhibits movement | Dopamine D2 receptors (inhibitory on pathway) |
Key Structures:
- Striatum (caudate + putamen): Input nucleus
- Globus pallidus interna (GPi): Output nucleus
- Substantia nigra pars compacta (SNpc): Dopamine production
- Subthalamic nucleus (STN): Excitatory drive to GPi
Pathological Changes in PD
Step 1: Dopaminergic Neuron Loss
- Progressive degeneration of pigmented (neuromelanin-containing) neurons in the substantia nigra pars compacta
- By the time motor symptoms appear, 50-70% of SNpc neurons and 70-80% of striatal dopamine have been lost [1,9]
- Loss is most severe in the ventrolateral tier of SNpc (projecting to putamen → motor symptoms)
Step 2: Basal Ganglia Imbalance
- Reduced dopamine in striatum (especially putamen)
- Loss of D1 stimulation → underactive direct pathway
- Loss of D2 inhibition → overactive indirect pathway
- Net effect: Excessive GPi inhibitory output → hypokinesia
Step 3: α-Synuclein Aggregation and Lewy Body Formation
α-Synuclein is a presynaptic protein that misfolds and aggregates in PD:
| Structure | Composition | Location |
|---|---|---|
| Lewy bodies | Dense intracytoplasmic eosinophilic inclusions with pale halo | Substantia nigra, locus coeruleus, cortex |
| Lewy neurites | Abnormal α-synuclein-positive neurites | Processes and axons |
Exam Detail: Molecular Mechanisms of α-Synuclein Toxicity:
- Protein misfolding: Native α-synuclein → oligomers → protofibrils → fibrils
- Prion-like propagation: Cell-to-cell spread via exosomes and direct transmission
- Mitochondrial dysfunction: Impaired Complex I activity; oxidative stress
- Proteasome/lysosome dysfunction: Impaired protein degradation
- Neuroinflammation: Microglial activation; inflammatory cytokines
- Synaptic dysfunction: Impaired vesicle trafficking and neurotransmitter release
The "prion-like" propagation hypothesis explains the stereotyped caudal-to-rostral spread observed in Braak staging and is supported by evidence of Lewy pathology in transplanted fetal dopaminergic neurons. [9]
Braak Staging (α-Synuclein Pathology Progression)
| Stage | Anatomical Location | Clinical Correlate |
|---|---|---|
| Stage 1 | Olfactory bulb, anterior olfactory nucleus, dorsal motor nucleus of vagus | Anosmia, constipation |
| Stage 2 | Lower brainstem (locus coeruleus, raphe nuclei) | REM sleep behaviour disorder, depression, anxiety |
| Stage 3 | Substantia nigra pars compacta | Motor symptoms begin |
| Stage 4 | Amygdala, mesocortex | Mood disturbance, emotional changes |
| Stage 5 | Neocortex (prefrontal, association) | Cognitive impairment |
| Stage 6 | Primary neocortex (motor, sensory) | Dementia, severe disability |
This staging supports the concept of a prodromal phase where non-motor features precede motor symptoms by years. [4]
Other Neurotransmitter Systems Affected
| System | Pathology | Clinical Consequence |
|---|---|---|
| Noradrenergic (locus coeruleus) | Neuron loss | Depression, anxiety, orthostatic hypotension, cognitive impairment |
| Serotonergic (raphe nuclei) | Neuron loss | Depression, sleep disturbance |
| Cholinergic (nucleus basalis of Meynert, PPN) | Neuron loss | Cognitive impairment, dementia, gait/balance dysfunction |
| Autonomic (dorsal motor nucleus, sympathetic ganglia) | Neuron loss, Lewy pathology | Constipation, urinary dysfunction, orthostatic hypotension |
| Enteric nervous system | α-synuclein deposition | Constipation (often decades before motor onset) |
This multi-system involvement explains why PD is far more than a dopamine-deficiency motor disorder.
4. Clinical Presentation
Motor Features (Cardinal Features)
ESSENTIAL: Bradykinesia must be present, PLUS at least one of rest tremor or rigidity. [3]
Bradykinesia
| Aspect | Description |
|---|---|
| Definition | Slowness of movement initiation AND progressive reduction in speed AND amplitude with repetitive movements |
| Key feature | Decrement — movements become smaller and slower with repetition |
| Assessment | Finger tapping, hand open/close, pronation-supination, heel tapping, toe tapping |
| Clinical examples | Difficulty with buttons, slower handwriting, reduced facial expression, soft voice |
Examination Technique:
- Finger tapping: Thumb to index finger, as fast and big as possible (10-15 times)
- Hand movements: Open/close fist rapidly
- Pronation-supination: Piano-playing movements
- Heel/toe tapping: Foot on ground, tap heel then toe
Look for: Speed reduction, amplitude reduction, freezing/arrests, fatiguing
Rest Tremor
| Characteristic | Description |
|---|---|
| Frequency | 4-6 Hz (slow) |
| Type | "Pill-rolling" — thumb and index finger; supination-pronation |
| When present | At rest; limb fully supported |
| Suppressors | Voluntary movement, mental concentration on tremor |
| Enhancers | Mental distraction (counting backwards), contralateral movement |
| Distribution | Asymmetric; usually starts in one hand; may involve leg, jaw, lips (rarely head) |
Re-emergent tremor: Action tremor that appears after a latency (seconds to minutes) when maintaining a posture — represents rest tremor suppressed by movement, then re-emerging.
Distinguishing from Essential Tremor:
| Feature | Parkinson's Tremor | Essential Tremor |
|---|---|---|
| Type | Rest > postural/kinetic | Postural/kinetic > rest |
| Frequency | 4-6 Hz | 6-12 Hz |
| Distribution | Asymmetric; pill-rolling | Bilateral; flexion-extension |
| Head involvement | Rare | Common (titubation) |
| Alcohol response | Minimal | Often improves |
| Writing | Micrographia | Large, tremulous |
| DaTSCAN | Abnormal | Normal |
Rigidity
| Type | Description |
|---|---|
| Lead-pipe rigidity | Constant resistance throughout passive movement range |
| Cogwheel rigidity | Ratchety resistance (tremor superimposed on rigidity) |
| Activation (Froment's manoeuvre) | Rigidity enhanced by contralateral motor activity |
Rigidity is velocity-independent (cf. spasticity — velocity-dependent, clasp-knife phenomenon).
Postural Instability
| Aspect | Description |
|---|---|
| Onset | Late feature (typically after 5+ years) |
| Mechanism | Loss of postural reflexes |
| Assessment | Pull test (retropulsion test) |
| Significance | Major contributor to falls; does not respond well to dopaminergic therapy |
Pull Test Technique:
- Patient stands with feet slightly apart
- Examiner stands behind, warns patient of pull
- Brisk backward pull on shoulders
- Normal: 1-2 steps to recover
- Abnormal: > 2 steps, or would fall without catching
Important: Postural instability early in disease (within first 3 years) suggests atypical parkinsonism, especially PSP. [7]
Gait Abnormalities
| Feature | Description |
|---|---|
| Shuffling gait | Short steps, feet barely clearing ground |
| Reduced arm swing | Asymmetric; often first on affected side |
| Festination | Involuntary acceleration of steps (chasing centre of gravity) |
| Freezing of gait (FOG) | Sudden inability to initiate or continue walking; "feet stuck to floor" |
| En bloc turning | Multiple small steps to turn; not pivoting |
| Stooped posture | Flexed trunk, neck, elbows, knees |
| Start hesitation | Difficulty initiating first step |
Freezing of Gait (FOG):
- Affects 30-60% of PD patients
- Major risk factor for falls [10]
- Triggers: Doorways, narrow spaces, turning, dual-tasking, approaching a destination
- Often refractory to dopaminergic therapy (but can be OFF-state phenomenon)
- May respond to visual/auditory cueing strategies
Other Motor Signs
| Sign | Description |
|---|---|
| Hypomimia | Reduced facial expression ("masked facies") |
| Reduced blink rate | Normal 15-20/min; PD may be 5-10/min |
| Hypophonia | Soft, monotonous voice |
| Micrographia | Small, cramped handwriting with progressive reduction in size |
| Drooling (sialorrhea) | Reduced swallowing rather than excess production |
| Dysphagia | Pharyngeal phase dysfunction; risk of aspiration |
| Camptocormia | Severe truncal flexion when upright |
| Pisa syndrome | Lateral truncal flexion |
| Antecollis | Dropped head (neck flexion) |
| Striatal deformities | Hand dystonia (striatal hand) |
5. Non-Motor Features
Non-motor symptoms affect nearly ALL patients with PD and are often more disabling than motor symptoms. Many precede motor onset (prodromal markers). [4,6]
Prodromal Non-Motor Features
| Feature | Timeline Before Motor Onset | Conversion Risk | Ref |
|---|---|---|---|
| REM sleep behaviour disorder (RBD) | 5-20 years | 80-90% at 15 years | [6] |
| Hyposmia/anosmia | 5-10 years | ~10% at 5 years | [4] |
| Constipation | 10-20 years | Modest | [4] |
| Depression | 5-10 years | 2-3x risk | [4] |
| Excessive daytime somnolence | Variable | Associated | [4] |
Autonomic Dysfunction
| Domain | Symptoms | Pathophysiology | Management |
|---|---|---|---|
| Cardiovascular | Orthostatic hypotension, postprandial hypotension, supine hypertension | Cardiac sympathetic denervation | Midodrine, droxidopa, fludrocortisone, compression stockings |
| Gastrointestinal | Constipation (most common), gastroparesis, dysphagia, sialorrhea | ENS α-synuclein, vagal dysfunction | Macrogol, fibre, prokinetics |
| Urological | Urgency, frequency, nocturia, urge incontinence | Detrusor hyperreflexia, sphincter dyssynergia | Anticholinergics (caution: cognition), mirabegron |
| Sexual | Erectile dysfunction, reduced libido | Autonomic + medication effects | Sildenafil; caution with DA agonist hypersexuality |
| Thermoregulatory | Hyperhidrosis, heat intolerance | Autonomic dysregulation | |
| Seborrhoea | Oily skin, seborrhoeic dermatitis | Autonomic + dopamine effects | Topical antifungals |
Exam Detail: Orthostatic Hypotension in PD:
- Definition: SBP drop ≥20 mmHg or DBP ≥10 mmHg within 3 minutes of standing
- Prevalence: 30-50% of PD patients
- Causes: Disease (sympathetic denervation) + medications (levodopa, dopamine agonists)
- Cardiac MIBG scintigraphy: Reduced uptake indicates cardiac sympathetic denervation (supports PD vs MSA)
- Management: Non-pharmacological (fluids, salt, compression, rising slowly) → fludrocortisone → midodrine/droxidopa
Sleep Disorders
| Disorder | Prevalence | Features | Management |
|---|---|---|---|
| REM sleep behaviour disorder (RBD) | 30-50% | Vivid dreams with dream enactment; bed partner may be injured | Clonazepam 0.25-1mg, melatonin 3-12mg; bed safety |
| Insomnia | 60-80% | Sleep fragmentation, early waking, difficulty initiating sleep | Sleep hygiene, trazodone, melatonin |
| Excessive daytime somnolence | 30-50% | Uncontrollable sleepiness; sudden sleep attacks (esp. with DA agonists) | Address nocturnal sleep; reduce DA agonists; modafinil |
| Restless legs syndrome | 15-20% | Urge to move legs; worse at rest/evening | Dopamine agonists (may worsen over time) |
| Sleep-disordered breathing | 20-40% | OSA, central apnoea | CPAP if indicated |
| Nocturia | 60-80% | Disturbs sleep; autonomic component | See urological management |
Neuropsychiatric Features
| Feature | Prevalence | Notes | Management |
|---|---|---|---|
| Depression | 30-50% | Intrinsic to PD (serotonergic loss); also reactive | SSRIs, SNRIs, TCAs (caution in elderly) |
| Anxiety | 30-40% | Generalised, panic, social anxiety; may fluctuate with motor state | SSRIs; buspirone; CBT |
| Apathy | 30-40% | Distinct from depression; lack of motivation/initiative | Difficult to treat; optimise dopaminergics |
| Psychosis (hallucinations, delusions) | 30-60% | Visual hallucinations (benign → frightening); delusions (jealousy, paranoia) | Reduce/stop anticholinergics, amantadine, DA agonists → quetiapine or clozapine [11] |
| Impulse control disorders | 10-20% (on DA agonists) | Pathological gambling, hypersexuality, compulsive shopping, binge eating, punding, dopamine dysregulation syndrome | Reduce/stop DA agonists [12] |
Exam Detail: PD Psychosis — Management Hierarchy:
- Identify/treat triggers (infection, dehydration, metabolic)
- Reduce anticholinergics, amantadine
- Reduce/stop dopamine agonists
- Reduce levodopa (if tolerable)
- Add atypical antipsychotic: Clozapine (first-line, requires monitoring) or quetiapine [11]
- Pimavanserin (5-HT2A inverse agonist) — approved in some regions
Never use typical antipsychotics (haloperidol) or risperidone/olanzapine — will dramatically worsen parkinsonism
Cognitive Impairment and Dementia
| Stage | Criteria | Prevalence | Features |
|---|---|---|---|
| Mild Cognitive Impairment (PD-MCI) | Cognitive decline without significant functional impairment | 20-40% | Executive dysfunction, visuospatial, attention |
| PD Dementia (PDD) | Dementia developing > 1 year after motor onset | 40-80% by late disease | Executive, attention, visuospatial >> memory (cf. AD) |
Risk factors for PDD:
- Older age at onset
- PIGD phenotype (vs tremor-dominant)
- Severe motor symptoms
- Hallucinations
- GBA mutations
- Low education
Management of PDD:
- Rivastigmine (cholinesterase inhibitor) — Level I evidence (EXPRESS trial) [13]
- Donepezil — less evidence
- Memantine — limited evidence
Dementia with Lewy Bodies (DLB) vs PDD:
- Same underlying pathology (α-synucleinopathy)
- "1-year rule": If dementia precedes or begins within 1 year of motor symptoms → DLB; if motor symptoms precede dementia by > 1 year → PDD
- Clinically similar; DLB has more prominent fluctuations and visual hallucinations at onset
Pain
| Type | Prevalence | Features |
|---|---|---|
| Musculoskeletal | Most common | Rigidity-related; dystonia-related |
| Dystonic | Common in OFF states | Morning foot dystonia; painful cramping |
| Central/neuropathic | Less common | Burning, aching; poorly localised |
| Radicular | May be coincidental or related to posture | |
| Akathitic | Inner restlessness |
Fatigue
- Affects 40-60% of PD patients
- Often independent of motor disability, depression, sleep disturbance
- Difficult to treat; optimise modifiable factors
6. Clinical Examination
Structured Approach
General Observation (before touching the patient):
- Tremor at rest (hands, legs, jaw)
- Facial expression (hypomimia)
- Blink rate
- Posture (flexed trunk, neck)
- Spontaneous movements
Gait Assessment:
- Observe walking across the room
- Arm swing (asymmetric reduction)
- Stride length (short, shuffling)
- Turning (en bloc, multiple steps)
- Postural stability (pull test — done safely)
Upper Limb Examination:
- Tremor: At rest (hands in lap); re-emergent (arms outstretched)
- Rigidity: Passive flexion/extension of wrist and elbow; Froment's manoeuvre
- Bradykinesia: Finger tapping, hand open/close, pronation-supination
Lower Limb Examination:
- Tremor at rest
- Rigidity: Passive movement at ankle, knee, hip
- Bradykinesia: Heel tapping, toe tapping
Additional Signs:
- Voice assessment (hypophonia)
- Writing sample (micrographia)
- Rapid alternating movements
MDS-UPDRS (Unified Parkinson's Disease Rating Scale)
The MDS-UPDRS is the standard assessment tool: [14]
| Part | Domain | Items |
|---|---|---|
| Part I | Non-motor experiences of daily living | Cognitive, hallucinations, depression, anxiety, apathy, sleep, pain, urinary, constipation, fatigue |
| Part II | Motor experiences of daily living | Speech, saliva, swallowing, eating, dressing, hygiene, handwriting, hobbies, turning in bed, tremor, getting out of bed, walking, freezing |
| Part III | Motor examination | Speech, facial expression, rigidity, finger tapping, hand movements, pronation-supination, toe tapping, leg agility, arising from chair, gait, freezing, postural stability, posture, global spontaneity, rest tremor, action tremor |
| Part IV | Motor complications | Time OFF, functional impact OFF, time with dyskinesias, functional impact dyskinesias, complexity of fluctuations, painful dyskinesias |
Hoehn and Yahr Staging
| Stage | Description |
|---|---|
| 1 | Unilateral involvement only |
| 1.5 | Unilateral + axial involvement |
| 2 | Bilateral involvement without impairment of balance |
| 2.5 | Mild bilateral disease with recovery on pull test |
| 3 | Mild-to-moderate bilateral disease; some postural instability; physically independent |
| 4 | Severe disability; still able to walk/stand unassisted |
| 5 | Wheelchair-bound or bedridden unless aided |
Schwab and England ADL Scale
Quantifies functional independence from 0% (bedridden) to 100% (completely independent).
7. Differential Diagnosis
Atypical Parkinsonian Syndromes
| Condition | Distinguishing Features | Levodopa Response | Ref |
|---|---|---|---|
| Progressive Supranuclear Palsy (PSP) | Early falls (within 1 year), vertical supranuclear gaze palsy (esp. downgaze), axial rigidity > limb, frontal dementia, "surprised" facies | Poor | [7] |
| Multiple System Atrophy (MSA) | Early severe autonomic failure (OH, urinary), cerebellar signs (MSA-C), stridor, cold hands, anterocollis | Poor-moderate | [7] |
| Corticobasal Syndrome (CBS) | Asymmetric, apraxia, alien limb, cortical sensory loss, myoclonus, dystonia | Poor | [7] |
| Dementia with Lewy Bodies (DLB) | Dementia early (less than 1 year of motor), prominent fluctuations, visual hallucinations | Variable | [7] |
Other Causes of Parkinsonism
| Cause | Key Features | Investigation |
|---|---|---|
| Drug-induced parkinsonism | Antipsychotics, metoclopramide, prochlorperazine; symmetric; no tremor or rest tremor | Drug history; DaTSCAN normal |
| Vascular parkinsonism | Lower body predominant; gait > tremor; stepwise; CVD risk factors | MRI (basal ganglia infarcts, white matter disease) |
| Normal pressure hydrocephalus (NPH) | Triad: Gait apraxia, dementia, urinary incontinence | MRI (ventriculomegaly out of proportion); LP tap test |
| Essential tremor | Postural/kinetic tremor > rest; bilateral; family history; alcohol-responsive | DaTSCAN normal |
| Wilson's disease | Young onset (less than 40); liver disease; Kayser-Fleischer rings; psychiatric features | Ceruloplasmin, 24h copper, slit-lamp |
Red Flags for Atypical Parkinsonism
[!CAUTION] Red Flags — Consider Alternative Diagnosis:
- Falls within first year of symptom onset
- Poor or absent response to adequate levodopa (> 1000mg/day for > 3 months)
- Symmetric onset or early bilateral disease
- Early severe autonomic failure (syncope, urinary incontinence, erectile dysfunction)
- Early dementia (within first year)
- Vertical supranuclear gaze palsy
- Early severe bulbar dysfunction (dysarthria, dysphagia)
- Rapid progression (doubling disability in 3 years)
- Cerebellar signs (ataxia, nystagmus)
- Pyramidal signs (spasticity, hyperreflexia, Babinski)
- Inspiratory stridor
- Alien limb phenomenon, apraxia, cortical sensory loss
8. Investigations
Diagnosis is Clinical
The diagnosis of PD is clinical. There is no diagnostic blood test or routine imaging study. MRI is performed to exclude alternative diagnoses, not to confirm PD.
First-Line Investigations
| Test | Purpose | Expected Findings |
|---|---|---|
| Clinical assessment | Diagnosis | MDS criteria fulfilled |
| MRI Brain | Exclude alternatives | Normal in PD (may show atrophy in atypical syndromes) |
Specialist Investigations
| Investigation | Indication | Findings in PD | Notes |
|---|---|---|---|
| DaTSCAN (SPECT) | Diagnostic uncertainty; tremor-dominant presentation; differentiate from ET/drug-induced | Reduced striatal dopamine transporter uptake (asymmetric, putamen > caudate) | Does NOT distinguish PD from atypical parkinsonism |
| MIBG cardiac scintigraphy | Differentiate PD from MSA | Reduced cardiac uptake in PD (sympathetic denervation); preserved in MSA | Limited availability |
| Olfactory testing | Supportive | Reduced in PD | UPSIT, Sniffin' Sticks |
| Polysomnography | RBD confirmation | REM without atonia + dream enactment | Prodromal marker |
| Autonomic function tests | Quantify autonomic dysfunction | Variable | Head-up tilt, HR variability |
| Genetic testing | Young-onset (less than 40-50), family history, Ashkenazi ancestry | LRRK2, GBA, Parkin, etc. |
MDS Clinical Diagnostic Criteria (2015) [3]
Step 1: Parkinsonism is defined as:
- Bradykinesia (ESSENTIAL), AND
- At least one of: Rest tremor, Rigidity
Step 2: Establish criteria for PD:
| Criterion Type | Criteria |
|---|---|
| Supportive criteria (≥2 for "clinically established PD") | Clear beneficial response to dopaminergic therapy; Levodopa-induced dyskinesias; Rest tremor of a limb; Olfactory loss or cardiac sympathetic denervation (MIBG) |
| Absolute exclusion criteria (any = NOT PD) | Cerebellar abnormalities; Downward vertical supranuclear gaze palsy or selective slowing of downward saccades; Frontotemporal dementia or primary progressive aphasia within 5 years; Parkinsonian features restricted to legs for > 3 years; Treatment with dopamine blockers; Absence of response to high-dose levodopa; Unequivocal cortical sensory loss, limb apraxia, or aphasia; Normal DaTSCAN; Another condition more likely |
| Red flags (must be counterbalanced by supportive criteria) | Rapid progression; No motor progression over 5 years; Early bulbar dysfunction; Early inspiratory stridor; Severe autonomic failure early; Recurrent falls early; Disproportionate anterocollis/contractures early; No common NMS; Otherwise unexplained pyramidal tract signs; Bilateral symmetric parkinsonism |
Diagnostic certainty:
- Clinically established PD: Parkinsonism + ≥2 supportive criteria + no exclusion criteria + no red flags (or red flags counterbalanced)
- Clinically probable PD: Parkinsonism + no exclusion criteria + red flags balanced by supportive criteria
9. Management
Principles of Treatment
- Treatment is symptomatic — no proven disease-modifying therapy
- Individualised — consider age, symptom severity, comorbidities, patient preference
- Multidisciplinary — neurology, PD nurse specialists, physiotherapy, occupational therapy, speech therapy, psychology
- Specialist initiation — all patients with suspected PD should be referred untreated to a movement disorder specialist [8]
When to Start Treatment
- When symptoms cause functional impairment or distress
- There is NO evidence that delaying levodopa prevents motor complications
- Early treatment improves quality of life [15]
Pharmacological Treatment — Initial Therapy
Levodopa
| Aspect | Details |
|---|---|
| Mechanism | Dopamine precursor; crosses blood-brain barrier; converted to dopamine by AADC |
| Formulation | Combined with peripheral dopa decarboxylase inhibitor (carbidopa or benserazide) to prevent peripheral conversion and nausea |
| Efficacy | Most effective symptomatic treatment for motor symptoms |
| Starting dose | Co-careldopa 50/12.5mg TDS, titrate over weeks to 100/25mg TDS or more |
| Titration | Increase gradually based on response; typical doses 300-600mg/day initially |
Levodopa — Advantages:
- Most effective motor symptom control
- Generally well-tolerated
- No evidence that early use accelerates motor complications (PD MED trial) [15]
Levodopa — Disadvantages:
- Motor complications develop over time (wearing off, dyskinesias)
- Short half-life (~90 mins) necessitating multiple daily doses
Exam Detail: Levodopa Pharmacology:
- L-DOPA crosses the blood-brain barrier via large neutral amino acid transporter (competition with dietary protein)
- Peripheral dopa decarboxylase inhibitor (carbidopa/benserazide) blocks peripheral conversion → reduces nausea, allows more levodopa to reach brain
- Central conversion to dopamine in residual dopaminergic neurons
- Half-life ~90 minutes (hence need for frequent dosing)
- Bioavailability affected by gastric emptying, protein intake
Wearing Off Phenomenon:
- As disease progresses and dopaminergic neurons decline, brain cannot buffer levodopa levels
- "Short-duration response" — symptoms return as plasma levodopa falls
- Manifests as end-of-dose deterioration, morning akinesia
- Management: Increase frequency, add COMT inhibitor, add MAO-B inhibitor, CR formulations
Dopamine Agonists
| Agent | Formulation | Starting Dose | Notes |
|---|---|---|---|
| Pramipexole | Immediate-release or prolonged-release | 0.088mg TDS IR; 0.26mg OD PR | D2/D3 agonist |
| Ropinirole | Immediate-release or prolonged-release | 0.25mg TDS IR; 2mg OD PR | D2/D3 agonist |
| Rotigotine | Transdermal patch | 2mg/24hr, increase weekly | 24-hour delivery; D1/D2/D3 agonist |
| Apomorphine | SC injection or infusion | Rescue: titrated in clinic; Infusion: specialist | Potent D1/D2 agonist; advanced therapy |
Dopamine Agonists — Advantages:
- Longer half-life than levodopa → less pulsatile stimulation
- May delay motor complications when used as initial monotherapy (but less effective)
- Patch formulation (rotigotine) for stable delivery
Dopamine Agonists — Disadvantages:
- Less effective than levodopa for motor symptoms
- Higher rate of adverse effects: Nausea, dizziness, somnolence, hallucinations, oedema
- Impulse control disorders (ICDs): Pathological gambling, hypersexuality, compulsive shopping, binge eating (10-20% on DA agonists) [12]
- Sleep attacks — sudden irresistible sleep episodes (driving warning)
- Not preferred in elderly (hallucinations, cognitive effects)
[!WARNING] Impulse Control Disorders: Screen ALL patients starting dopamine agonists for ICDs at baseline and at every visit. Ask specifically about gambling, shopping, eating, sexual behaviour. ICDs may not be spontaneously disclosed due to shame. First-line management is dose reduction or discontinuation of dopamine agonist.
MAO-B Inhibitors
| Agent | Dose | Notes |
|---|---|---|
| Rasagiline | 1mg OD | Irreversible MAO-B inhibitor; monotherapy or adjunct |
| Selegiline | 5-10mg OD (morning) | Irreversible; metabolised to amphetamine derivatives (insomnia) |
| Safinamide | 50-100mg OD | Reversible MAO-B inhibitor + glutamate modulation; adjunct only |
MAO-B Inhibitors — Role:
- Monotherapy: Mild early disease (modest efficacy)
- Adjunct: Add to levodopa to reduce OFF time
Adverse effects:
- Generally well-tolerated
- Headache, nausea, insomnia (selegiline)
- Drug interactions: Avoid with pethidine, tramadol, SSRIs (serotonin syndrome risk — though rare with MAO-B selective doses)
Initial Treatment Choice — Summary
| Patient Profile | Suggested Initial Therapy | Rationale |
|---|---|---|
| Most patients | Levodopa | Most effective; no evidence early use causes harm [15] |
| Young patient (less than 70) with mild symptoms | Consider DA agonist or MAO-B inhibitor | May delay motor complications; will need levodopa eventually |
| Elderly patient or cognitive impairment | Levodopa | DA agonists higher risk of confusion, hallucinations, ICDs |
| Tremor-dominant, mild | MAO-B inhibitor or levodopa | Either reasonable |
Management of Motor Complications
Motor complications affect most patients after years on levodopa:
Wearing Off (End-of-Dose Deterioration)
| Strategy | Options |
|---|---|
| Increase levodopa frequency | More frequent, smaller doses |
| Add COMT inhibitor | Entacapone (with each levodopa dose), opicapone (once daily) |
| Add MAO-B inhibitor | Rasagiline, safinamide |
| Add dopamine agonist | If not already on one |
| CR levodopa formulations | Controlled-release (may be useful for nocturnal symptoms; less predictable absorption) |
| Levodopa-carbidopa intestinal gel (LCIG) | Continuous infusion — advanced therapy |
| Apomorphine | Intermittent SC injections for rescue; or continuous infusion |
| DBS | For suitable candidates |
Exam Detail: COMT Inhibitors:
- Block catechol-O-methyltransferase → reduce peripheral levodopa degradation → increase half-life and brain delivery
- Entacapone: 200mg with each levodopa dose (or as Stalevo — combination pill)
- Opicapone: 50mg once daily at bedtime (newer; longer acting)
- Tolcapone: More effective but requires LFT monitoring (hepatotoxicity risk)
- Main side effects: Diarrhoea (especially entacapone), discoloured urine, dyskinesias (if levodopa now "stronger")
Dyskinesias
| Type | Timing | Features | Management |
|---|---|---|---|
| Peak-dose dyskinesia | At peak levodopa effect (ON state) | Choreiform, often involving neck/trunk/limbs | Reduce individual levodopa doses; add amantadine; consider DBS |
| Biphasic dyskinesia | As levodopa kicking in or wearing off | Often dystonic; legs > arms; can be violent | Difficult; aim for stable plasma levels |
| OFF-period dystonia | During OFF state; often morning | Painful foot dystonia; leg cramping | Longer-acting preparations; early morning levodopa |
Amantadine for Dyskinesias:
- NMDA antagonist; antidyskinetic properties [16]
- Dose: 100mg OD–TDS (lower in renal impairment)
- Side effects: Livedo reticularis, ankle oedema, confusion, hallucinations
- ADS (amantadine extended-release) formulations available
Advanced Therapies (Device-Aided Therapies)
For patients with motor fluctuations and dyskinesias inadequately controlled by oral medications.
Deep Brain Stimulation (DBS)
| Aspect | Details |
|---|---|
| Targets | Subthalamic nucleus (STN) — most common; or Globus pallidus interna (GPi) |
| Mechanism | High-frequency electrical stimulation modulates basal ganglia circuitry |
| Indications | Motor fluctuations, dyskinesias, medication-refractory tremor in patients with good levodopa response |
| Evidence | Level 1b — RCTs including EARLYSTIM [17] |
Patient Selection for DBS:
| Inclusion Criteria | Exclusion Criteria |
|---|---|
| Idiopathic PD (good levodopa response) | Atypical parkinsonism |
| Motor fluctuations/dyskinesias despite optimised medications | Significant cognitive impairment (dementia) |
| Age typically less than 70-75 (relative) | Untreated psychiatric disease |
| Adequate cognition | Medical comorbidities precluding surgery |
| Realistic expectations | Symptoms not levodopa-responsive (postural instability, freezing, speech) |
DBS Outcomes:
- Reduces OFF time by 4-6 hours/day
- Reduces dyskinesias (STN: via medication reduction; GPi: direct effect)
- Improves quality of life
- Allows reduction in dopaminergic medications (STN > GPi)
- Does NOT improve: Levodopa-resistant symptoms (postural instability, freezing, dementia, speech)
- EARLYSTIM showed benefit of earlier DBS (within 3 years of motor complications) [17]
Complications of DBS:
- Surgical: Haemorrhage (1-2%), infection (3-5%), hardware problems
- Stimulation-related: Speech problems, balance issues, paraesthesias (usually adjustable)
- Cognitive/psychiatric: Depression, apathy, impulsivity (especially with STN)
Levodopa-Carbidopa Intestinal Gel (LCIG / Duodopa)
| Aspect | Details |
|---|---|
| Delivery | Continuous infusion via PEG-J tube into jejunum |
| Mechanism | Bypasses gastric emptying; continuous dopaminergic stimulation |
| Indications | Advanced fluctuations; DBS not suitable/wanted |
| Evidence | Level 1b [18] |
LCIG Complications:
- PEG-J tube issues: Dislodgement, blockage, infection, leakage
- Polyneuropathy (vitamin B12/B6 deficiency — monitor and supplement)
- Weight loss
- Cost
Apomorphine
| Formulation | Use |
|---|---|
| Intermittent SC injection (APO-go PEN) | Rescue for OFF episodes; onset ~10-15 minutes |
| Continuous SC infusion | Pump-based infusion for advanced fluctuations |
- Most potent dopamine agonist (D1/D2)
- Requires pre-treatment with domperidone (antiemetic) for initiation
- Side effects: Nausea (hence domperidone), nodules at injection sites, sedation, confusion, hallucinations
Non-Motor Symptom Management — Summary
| Symptom | First-Line | Second-Line |
|---|---|---|
| Depression | SSRI (sertraline, citalopram), SNRI (venlafaxine) | Mirtazapine, pramipexole (has antidepressant effect) |
| Anxiety | SSRI, buspirone | Benzodiazepines (short-term) |
| Psychosis | Reduce anticholinergics/amantadine → reduce DA agonist → reduce levodopa → add quetiapine or clozapine [11] | Pimavanserin |
| Dementia | Rivastigmine [13] | Donepezil |
| Orthostatic hypotension | Non-pharmacological (fluids, salt, compression) → midodrine → fludrocortisone | Droxidopa |
| Constipation | Macrogol, fibre, fluids | Lubiprostone, prucalopride |
| Sialorrhea | Botulinum toxin to salivary glands | Glycopyrrolate |
| RBD | Clonazepam 0.25-1mg, melatonin | Bed safety measures |
| Insomnia | Sleep hygiene, treat nocturnal motor symptoms | Melatonin, trazodone |
| Urinary urgency | Mirabegron (beta-3 agonist) | Anticholinergics (caution: cognition) |
10. Falls Prevention in Parkinson's Disease
Falls are a major cause of morbidity and mortality in PD, affecting 60-70% of patients. [10]
Risk Factors for Falls
| Category | Risk Factors |
|---|---|
| Disease-related | Disease duration/severity, PIGD phenotype, postural instability, freezing of gait, rigidity |
| Cognitive | Cognitive impairment, dual-tasking difficulty, impulsivity |
| Autonomic | Orthostatic hypotension |
| Medication | Polypharmacy, sedating medications, dopamine agonists (somnolence), OFF-state motor symptoms |
| Environmental | Home hazards, poor lighting, uneven surfaces |
| Previous falls | Strongest predictor of future falls |
Falls Assessment
- Falls history: Frequency, circumstances, injuries, fear of falling
- Motor assessment: Postural stability (pull test), freezing of gait, gait speed
- Cognitive screening: MoCA or similar
- Orthostatic BP measurement
- Medication review: Sedatives, anticholinergics, polypharmacy
- Vision assessment
- Home hazard assessment (OT referral)
- Footwear review
Falls Prevention Strategies
| Intervention | Evidence | Details |
|---|---|---|
| Physiotherapy | Level I | Balance training, strength, cueing strategies for FOG [10] |
| Cueing strategies | Level II | Visual cues (laser, lines on floor), auditory cues (metronome, music) for freezing |
| Exercise programs | Level I | Tai chi, dance (tango), resistance training |
| Home assessment (OT) | Level II | Remove hazards, improve lighting, grab rails |
| Medication optimisation | Level III | Reduce OFF time; treat OH; reduce sedatives |
| Hip protectors | Level III | May reduce fracture severity |
| Assistive devices | Expert opinion | Cane, walker (consider wheeled for FOG) |
| Treat orthostatic hypotension | Level II | See autonomic management |
| Cognitive-motor training | Level II | Dual-task training |
Freezing of Gait (FOG) Management
| Strategy | Description |
|---|---|
| Cueing | Visual cues (laser pointer attached to cane, stripes on floor); auditory cues (metronome, rhythmic music) |
| Attentional strategies | Focus on stepping over an imaginary object; exaggerated stepping |
| Medication optimisation | If OFF-state freezing: reduce OFF time; some patients have ON-state freezing (reduce levodopa) |
| DBS | May improve OFF-state freezing; variable effect on gait overall |
11. Multidisciplinary Team Management
PD is best managed by a specialist multidisciplinary team (MDT). NICE recommends access to the following: [8]
| Team Member | Role |
|---|---|
| Movement disorder specialist / Neurologist / Geriatrician | Diagnosis, medication management, advanced therapy decisions |
| Parkinson's disease nurse specialist | Education, medication adjustment, support, liaison |
| Physiotherapist | Gait training, balance, falls prevention, exercise prescription, cueing |
| Occupational therapist | ADL assessment, home assessment, equipment, work adaptations |
| Speech and language therapist (SALT) | Hypophonia (LSVT LOUD), swallowing assessment, communication aids |
| Dietitian | Nutrition, protein timing with levodopa, weight management |
| Psychologist / Psychiatrist | Depression, anxiety, cognitive support, ICD management |
| Social worker | Benefits, care packages, carer support |
| Palliative care | Advance care planning, symptom control in advanced disease |
Physiotherapy in PD
| Component | Details |
|---|---|
| Gait training | Increase stride length, arm swing; cueing strategies |
| Balance training | Weight shifting, perturbation-based training |
| Strength training | Resistance exercises; high-intensity shown beneficial |
| Flexibility | Stretching for rigidity, postural correction |
| Aerobic exercise | Cycling, walking, treadmill — neuroprotective effects suggested |
| Specific programs | LSVT BIG (high-amplitude movement), PD Warrior |
Speech Therapy
- LSVT LOUD: Evidence-based intensive treatment for hypophonia; focuses on loud phonation
- Swallowing assessment for dysphagia risk
- Alternative communication strategies
12. Prognosis and Outcomes
Natural History
PD is progressive but highly variable. Patients may live 15-20+ years from diagnosis. The rate of progression depends on:
| Factor | Better Prognosis | Worse Prognosis |
|---|---|---|
| Subtype | Tremor-dominant | PIGD (postural instability gait difficulty) |
| Age of onset | Younger | Older |
| Cognition | Intact | Early impairment |
| Motor response | Good levodopa response | Poor levodopa response |
| Genetics | Parkin (slow) | GBA (faster, dementia risk) |
Mortality
- Life expectancy reduced by approximately 2-5 years compared to age-matched controls
- Main causes of death: Pneumonia (aspiration), falls-related injuries, cardiovascular disease
- Dementia is a major determinant of quality of life and mortality in late disease
Motor Complications Timeline
| Complication | Typical Onset |
|---|---|
| Motor fluctuations (wearing off) | 40-50% by 5 years on levodopa |
| Dyskinesias | 30-50% by 5 years on levodopa |
| Falls | Variable; later in idiopathic PD (early = atypical) |
| Dementia (PDD) | Cumulative 40-80% by late disease |
Key Milestones
| Milestone | Average Time from Diagnosis |
|---|---|
| Hoehn & Yahr Stage 3 | 5-7 years |
| Hoehn & Yahr Stage 5 | 10-15 years |
| Care home requirement | Variable; later with good MDT support |
13. Exam-Focused Content
Common MRCP Questions
- "What are the cardinal features of Parkinson's disease?"
- "How do you differentiate PD from atypical parkinsonism?"
- "What is the role of DaTSCAN in diagnosis?"
- "Describe the management of motor fluctuations."
- "How would you manage PD psychosis?"
- "What are the side effects of dopamine agonists?"
- "Which patients are suitable for DBS?"
Viva Points
Viva Point: Opening Statement: "Parkinson's disease is a progressive neurodegenerative disorder characterised by the loss of dopaminergic neurons in the substantia nigra pars compacta, leading to bradykinesia, rigidity, and rest tremor, along with a wide spectrum of non-motor features. It is the second most common neurodegenerative disorder, affecting approximately 1% of people over 60 years."
Key Facts to Mention:
- Diagnosis is clinical: Bradykinesia PLUS tremor or rigidity (MDS criteria)
- Asymmetric onset is typical
- Levodopa is the most effective treatment (PD MED trial evidence)
- Non-motor features are often more disabling than motor symptoms
- Motor complications (wearing off, dyskinesias) develop with disease progression
- DBS is effective for motor fluctuations in appropriate candidates (EARLYSTIM)
- Falls are a major cause of morbidity — multidisciplinary approach essential
Evidence to Cite:
- MDS Clinical Diagnostic Criteria 2015
- PD MED trial (2014) — levodopa first-line
- EARLYSTIM (2013) — early DBS improves QoL
Red Flags to Mention:
- Early falls, poor levodopa response, symmetric onset, early autonomic failure, vertical gaze palsy
Common Mistakes (What Fails Candidates)
❌ Forgetting bradykinesia is essential — cannot diagnose PD with tremor and rigidity alone
❌ Confusing PD with essential tremor — know the differences (rest vs action tremor, DaTSCAN)
❌ Not knowing red flags — miss atypical parkinsonism
❌ Recommending dopamine agonists in elderly/cognitively impaired — high side effect risk
❌ Using typical antipsychotics for PD psychosis — will worsen parkinsonism dramatically
❌ Forgetting impulse control disorders — must screen all patients on dopamine agonists
❌ Not referring to specialist — all suspected PD should be seen by movement disorder specialist
Model Answer
Q: A 68-year-old man presents with a 2-year history of progressive right hand tremor and slowness. How would you approach this case?
"I would approach this systematically. First, I would take a detailed history focusing on:
- Motor symptoms: Tremor character (rest, action, or both), slowness, stiffness, difficulty with fine movements, gait changes
- Non-motor symptoms: Sleep disturbance (RBD), constipation, loss of smell, mood, cognition
- Medication history: Any dopamine-blocking agents (metoclopramide, antipsychotics)
- Family history: PD or tremor
On examination, I would assess the cardinal features:
- Bradykinesia: Finger tapping, hand open/close, looking for decrement
- Tremor: Rest vs postural/kinetic, frequency, pill-rolling character
- Rigidity: Lead-pipe, cogwheel
- Gait: Arm swing, stride length, turning, postural stability
Key examination findings that would support idiopathic PD: asymmetric bradykinesia with decrement, 4-6 Hz rest tremor, cogwheel rigidity, reduced arm swing on affected side.
I would look for red flags suggesting atypical parkinsonism: early falls, symmetric onset, poor response to levodopa, early autonomic failure, vertical gaze palsy.
Diagnosis is clinical using the MDS criteria. I would refer to a movement disorder specialist before initiating treatment, as per NICE guidance.
Investigations: I would arrange an MRI brain to exclude structural causes. DaTSCAN may be considered if there is diagnostic uncertainty (e.g., differentiating from essential tremor or drug-induced parkinsonism).
Treatment would be guided by symptom severity and patient preference. For most patients, levodopa remains the most effective option for motor symptoms, supported by the PD MED trial."
14. Patient/Layperson Explanation
What is Parkinson's Disease?
Parkinson's disease is a condition where certain nerve cells in your brain gradually stop working. These cells normally produce a chemical called dopamine, which helps control your movements. As these cells are lost, you may experience shaking (tremor), stiffness, and slowness of movement.
Parkinson's also affects other parts of the nervous system, which is why you might experience other symptoms like constipation, sleep problems, low mood, or changes in blood pressure.
Why Does It Matter?
Parkinson's is a progressive condition, meaning it changes over time. However, with the right treatment and support, most people with Parkinson's can maintain a good quality of life for many years. Treatment can significantly improve your symptoms.
What Are the Symptoms?
Main movement symptoms:
- Tremor (shaking) — usually when your hand is at rest
- Slowness of movement
- Stiffness in your arms and legs
- Balance problems (usually later on)
Other common symptoms:
- Loss of smell
- Constipation
- Sleep problems (vivid dreams, acting out dreams)
- Low mood or anxiety
- Tiredness
- Quieter voice
- Smaller handwriting
- Memory and thinking changes (may occur later)
How Is It Diagnosed?
Parkinson's is diagnosed by a specialist (neurologist or geriatrician) based on your symptoms and examination. There is no blood test for Parkinson's, but scans may be used to help in uncertain cases or to rule out other conditions.
How Is It Treated?
Medications:
The main treatments work by replacing or mimicking dopamine:
- Levodopa: The most effective medication; converted to dopamine in your brain
- Dopamine agonists: Mimic the effect of dopamine
- Other tablets: Help make your medications work better or last longer
Therapies:
- Physiotherapy — helps with movement, balance, and preventing falls
- Occupational therapy — helps with daily activities and home safety
- Speech therapy — helps with voice and swallowing
Surgery (for some people): Deep brain stimulation (DBS) involves implanting electrodes in the brain connected to a small device (like a pacemaker). This can help when medications aren't controlling symptoms well enough.
What to Expect
- Parkinson's progresses slowly — most people have many good years
- Medications work well, especially at first; they can be adjusted over time
- You may notice that medications work for shorter periods as years pass — this can be managed with adjustments
- Regular follow-up with your specialist team is important
- A team approach (doctors, nurses, therapists) helps maintain your independence
When to Seek Help
Contact your Parkinson's team, nurse, or GP if:
- Your symptoms are getting significantly worse
- You're having problems with medication timing or wearing off
- You experience hallucinations (seeing things that aren't there) or confusion
- Falls are increasing
- Mood is low and affecting your daily life
- You're struggling with swallowing
15. Evidence and Guidelines
Key Guidelines
| Guideline | Organisation | Year | Key Recommendations |
|---|---|---|---|
| Parkinson's Disease in Adults (NG71) | NICE | 2017 | Specialist referral; individualised treatment; MDT approach; levodopa or DA agonist as initial therapy; DBS for motor complications [8] |
| MDS Clinical Diagnostic Criteria | Movement Disorder Society | 2015 | Standardised criteria for clinical diagnosis; supportive and exclusion criteria [3] |
| MDS Evidence-Based Medicine Review | Movement Disorder Society | 2018 update | Evidence levels for pharmacological and non-pharmacological treatments |
Landmark Trials
| Trial | Year | Design | Key Findings | Impact |
|---|---|---|---|---|
| PD MED | 2014 | Large pragmatic RCT; initial treatment with levodopa vs levodopa-sparing (DA agonist or MAO-B inhibitor) | Levodopa provided better symptom control; no long-term disadvantage; levodopa-sparing did not delay need for levodopa or prevent motor complications [15] | Levodopa accepted as first-line for most patients |
| EARLYSTIM | 2013 | RCT; DBS + medical therapy vs best medical therapy alone in patients with early motor complications | DBS improved quality of life, motor function; mean 4.5 years disease duration [17] | Earlier consideration of DBS in suitable candidates |
| ELLDOPA | 2004 | RCT; different levodopa doses vs placebo | Higher doses more effective; no evidence of levodopa toxicity; suggested levodopa may slow progression (confounded by symptomatic effects) | Supported safety of levodopa |
| EXPRESS | 2004 | RCT; rivastigmine vs placebo in PD dementia | Rivastigmine improved cognition and ADLs [13] | Rivastigmine approved for PD dementia |
| ADAGIO | 2009 | RCT; rasagiline 1mg vs 2mg early vs delayed start | 1mg early start showed possible disease-modifying effect (not confirmed with 2mg); remains controversial | Interest in potential neuroprotection |
Evidence Level Summary
| Intervention | Evidence Level | Key Studies |
|---|---|---|
| Levodopa for motor symptoms | 1a | Meta-analyses, PD MED |
| Dopamine agonists | 1a | Multiple RCTs |
| MAO-B inhibitors | 1a | RCTs including ADAGIO |
| COMT inhibitors (adjunct) | 1a | RCTs |
| DBS for motor fluctuations | 1b | EARLYSTIM, PD SURG |
| Rivastigmine for PD dementia | 1b | EXPRESS |
| Clozapine for PD psychosis | 1b | RCTs |
| Physiotherapy | 1b | Multiple RCTs |
| Amantadine for dyskinesias | 1b | RCTs [16] |
16. References
-
Kalia LV, Lang AE. Parkinson's disease. Lancet. 2015;386(9996):896-912. doi:10.1016/S0140-6736(14)61393-3 PMID: 25904081
-
Dorsey ER, Sherer T, Okun MS, Bloem BR. The emerging evidence of the Parkinson pandemic. J Parkinsons Dis. 2018;8(s1):S3-S8. doi:10.3233/JPD-181474 PMID: 30584159
-
Postuma RB, Berg D, Stern M, et al. MDS clinical diagnostic criteria for Parkinson's disease. Mov Disord. 2015;30(12):1591-1601. doi:10.1002/mds.26424 PMID: 26474316
-
Schapira AHV, Chaudhuri KR, Jenner P. Non-motor features of Parkinson disease. Nat Rev Neurosci. 2017;18(7):435-450. doi:10.1038/nrn.2017.62 PMID: 28592904
-
Armstrong MJ, Okun MS. Diagnosis and treatment of Parkinson disease: a review. JAMA. 2020;323(6):548-560. doi:10.1001/jama.2019.22360 PMID: 32044947
-
Postuma RB, Iranzo A, Hu M, et al. Risk and predictors of dementia and parkinsonism in idiopathic REM sleep behaviour disorder: a multicentre study. Brain. 2019;142(3):744-759. doi:10.1093/brain/awz030 PMID: 30789229
-
Höglinger GU, Respondek G, Stamelou M, et al. Clinical diagnosis of progressive supranuclear palsy: the movement disorder society criteria. Mov Disord. 2017;32(6):853-864. doi:10.1002/mds.26987 PMID: 28467028
-
National Institute for Health and Care Excellence. Parkinson's disease in adults (NG71). 2017. nice.org.uk/guidance/ng71
-
Poewe W, Seppi K, Tanner CM, et al. Parkinson disease. Nat Rev Dis Primers. 2017;3:17013. doi:10.1038/nrdp.2017.13 PMID: 28332488
-
Allen NE, Schwarzel AK, Canning CG. Recurrent falls in Parkinson's disease: a systematic review. Parkinsons Dis. 2013;2013:906274. doi:10.1155/2013/906274 PMID: 23533953
-
Seppi K, Ray Chaudhuri K, Coelho M, et al. Update on treatments for nonmotor symptoms of Parkinson's disease-an evidence-based medicine review. Mov Disord. 2019;34(2):180-198. doi:10.1002/mds.27602 PMID: 30653247
-
Weintraub D, Koester J, Potenza MN, et al. Impulse control disorders in Parkinson disease: a cross-sectional study of 3090 patients. Arch Neurol. 2010;67(5):589-595. doi:10.1001/archneurol.2010.65 PMID: 20457959
-
Emre M, Aarsland D, Albanese A, et al. Rivastigmine for dementia associated with Parkinson's disease. N Engl J Med. 2004;351(24):2509-2518. doi:10.1056/NEJMoa041470 PMID: 15590953
-
Goetz CG, Tilley BC, Shaftman SR, et al. Movement Disorder Society-sponsored revision of the Unified Parkinson's Disease Rating Scale (MDS-UPDRS): scale presentation and clinimetric testing results. Mov Disord. 2008;23(15):2129-2170. doi:10.1002/mds.22340 PMID: 19025984
-
PD MED Collaborative Group. Long-term effectiveness of dopamine agonists and monoamine oxidase B inhibitors compared with levodopa as initial treatment for Parkinson's disease (PD MED): a large, open-label, pragmatic randomised trial. Lancet. 2014;384(9949):1196-1205. doi:10.1016/S0140-6736(14)60683-8 PMID: 24928805
-
Pahwa R, Tanner CM, Hauser RA, et al. ADS-5102 (amantadine) extended-release capsules for levodopa-induced dyskinesia in Parkinson disease (EASE LID study): a randomized clinical trial. JAMA Neurol. 2017;74(8):941-949. doi:10.1001/jamaneurol.2017.0943 PMID: 28548767
-
Schuepbach WMM, Rau J, Knudsen K, et al. Neurostimulation for Parkinson's disease with early motor complications (EARLYSTIM). N Engl J Med. 2013;368(7):610-622. doi:10.1056/NEJMoa1205158 PMID: 23406026
-
Olanow CW, Kieburtz K, Odin P, et al. Continuous intrajejunal infusion of levodopa-carbidopa intestinal gel for patients with advanced Parkinson's disease: a randomised, controlled, double-blind, double-dummy study. Lancet Neurol. 2014;13(2):141-149. doi:10.1016/S1474-4422(13)70293-X PMID: 24361112
-
Fox SH, Katzenschlager R, Lim SY, et al. International Parkinson and movement disorder society evidence-based medicine review: update on treatments for the motor symptoms of Parkinson's disease. Mov Disord. 2018;33(8):1248-1266. doi:10.1002/mds.27372 PMID: 29570866
-
Bloem BR, Okun MS, Klein C. Parkinson's disease. Lancet. 2021;397(10291):2284-2303. doi:10.1016/S0140-6736(21)00218-X PMID: 33848468
Medical Disclaimer: MedVellum content is for educational purposes and clinical reference. Clinical decisions should account for individual patient circumstances. Always consult appropriate guidelines and specialists for patient care.
Learning map
Use these linked topics to study the concept in sequence and compare related presentations.
Prerequisites
Start here if you need the foundation before this topic.
- Basal Ganglia Anatomy and Physiology
- Dopamine Neurotransmission
Differentials
Competing diagnoses and look-alikes to compare.
- Essential Tremor
- Multiple System Atrophy
- Progressive Supranuclear Palsy
- Drug-Induced Parkinsonism
Consequences
Complications and downstream problems to keep in mind.
- Parkinson's Disease Dementia
- Falls in the Elderly