Peptic Ulcer Disease
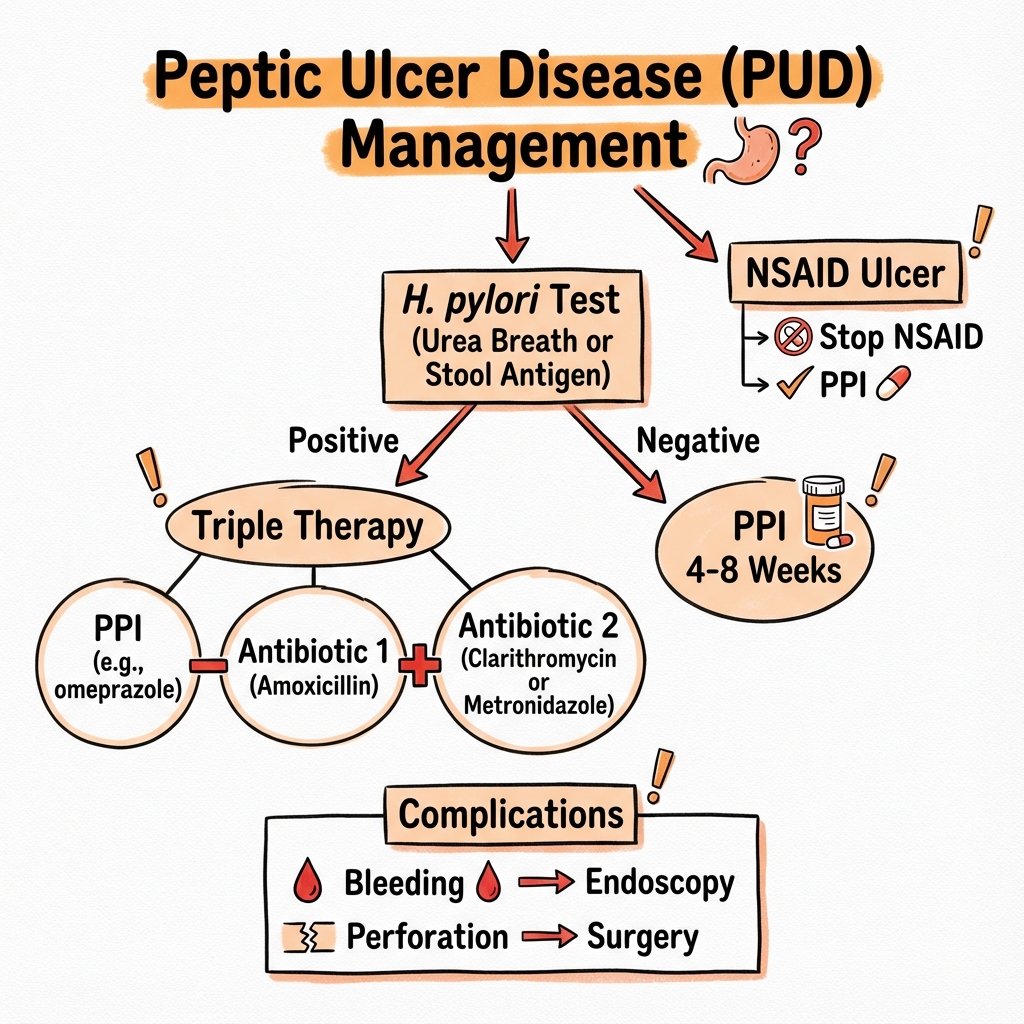
Test-and-Treat Strategy : NICE recommends non-invasive H. pylori testing (UBT or stool antigen) followed by eradication therapy for patients less than 55 years without alarm features, avoiding endoscopy. This strategy...
What matters first
Test-and-Treat Strategy : NICE recommends non-invasive H. pylori testing (UBT or stool antigen) followed by eradication therapy for patients less than 55 years without alarm features, avoiding endoscopy. This strategy...
GI bleeding (haematemesis, melaena)
10 Jan 2026
Generated educational material; verify before clinical use.
25 cited sources
See the concept before reading it
Study the key anatomy, imaging, and decision pathways as full teaching plates.
Clinical board
A visual summary of the highest-yield teaching signals on this page.
Urgent signals
Safety-critical features pulled from the topic metadata.
- GI bleeding (haematemesis, melaena)
- Perforation (sudden severe pain, peritonism)
- Gastric outlet obstruction (projectile vomiting)
- Alarm features (weight loss, dysphagia, anaemia)
Exam focus
Current exam surfaces linked to this topic.
- MRCP
- MRCGP
- FRACP
- USMLE
Linked comparisons
Differentials and adjacent topics worth opening next.
- Gastro-oesophageal Reflux Disease
- Functional Dyspepsia
Content status and exam context
This page is AI-generated educational content. It may contain errors or omissions and is not a substitute for current guidelines, local protocols, senior clinical judgement, or professional medical advice.
MedVellum does not claim an individual clinician reviewer, board certification, or professional credential for this page unless a future version names a real, verifiable reviewer.
Clinical explanation and evidence
Peptic Ulcer Disease
1. Topic Overview
Summary
Peptic ulcer disease (PUD) refers to ulceration extending through the muscularis mucosae of the gastric or duodenal mucosa, resulting from an imbalance between aggressive luminal factors (primarily gastric acid and pepsin) and mucosal defence mechanisms. The two dominant aetiologies are Helicobacter pylori infection (60-70% of cases) and non-steroidal anti-inflammatory drug (NSAID) use (20-30%). [1] Clinical presentation typically features epigastric pain with distinct temporal patterns: duodenal ulcers characteristically produce pain 2-3 hours postprandially that is relieved by food, whereas gastric ulcers tend to cause pain exacerbated by eating. Diagnosis is definitively established by oesophagogastroduodenoscopy (OGD) with biopsy. Management centres on acid suppression with proton pump inhibitors (PPIs) and H. pylori eradication therapy when indicated. [2] Major complications—gastrointestinal bleeding (15-20% of patients), perforation (5-10%), and gastric outlet obstruction (2-5%)—confer significant morbidity and mortality, particularly in elderly populations. [3] Patients presenting with alarm features (unintended weight loss, dysphagia, anaemia, age > 55 with new-onset dyspepsia) require urgent endoscopic evaluation to exclude malignancy.
Key Facts
- Definition: Break in gastric or duodenal mucosa extending through muscularis mucosae, ≥5mm diameter
- Global Prevalence: Lifetime risk 5-10%; annual incidence 0.1-0.3% (declining in developed nations) [4]
- Main Aetiologies: H. pylori (60-70%), NSAIDs (20-30%), idiopathic (5-10%), rare causes (less than 5%) [1]
- Duodenal:Gastric Ratio: Historically 4:1; now approaching 2:1 due to declining H. pylori prevalence [5]
- Diagnosis: Gold standard = OGD with biopsy; non-invasive H. pylori testing (urea breath test [UBT], stool antigen)
- First-Line Treatment: PPI + clarithromycin triple therapy (7-14 days) for H. pylori-positive cases [2]
- Complication Rates: Bleeding 15-20%, perforation 5-10%, obstruction 2-5% [3]
- Eradication Success: 80-95% with first-line triple therapy; resistance patterns vary geographically [6]
Clinical Pearls
Test-and-Treat Strategy: NICE recommends non-invasive H. pylori testing (UBT or stool antigen) followed by eradication therapy for patients less than 55 years without alarm features, avoiding endoscopy. [7] This strategy is cost-effective and reduces endoscopic burden, but requires strict adherence to age and alarm feature criteria.
Gastric vs Duodenal Pain Patterns: The classical distinction (DU pain relieved by food, GU pain worsened by food) reflects underlying pathophysiology—DU typically associated with acid hypersecretion, GU with normal or reduced acid output. However, symptom overlap is common (30-40% of cases), limiting diagnostic specificity. [8]
Verify Eradication Mandatorily: Failure to confirm H. pylori eradication is a major cause of recurrent ulceration. Testing must occur ≥4 weeks post-treatment completion and ≥2 weeks after PPI cessation to avoid false negatives (PPI suppresses bacterial load). [2] UBT or stool antigen are preferred; serology is inappropriate (remains positive post-eradication).
NSAID Risk Stratification: Concurrent use of aspirin, anticoagulants, corticosteroids, or selective serotonin reuptake inhibitors (SSRIs) multiplies NSAID ulcer risk. [9] Age > 65, prior ulcer history, and high-dose/dual NSAID therapy are additional major risk factors requiring prophylactic PPI co-prescription.
Refractory Ulcers: If ulcers fail to heal despite 8-12 weeks of PPI therapy and confirmed H. pylori eradication, consider Zollinger-Ellison syndrome (fasting gastrin > 1000 pg/mL diagnostic), Crohn's disease, malignancy (gastric ulcers), or non-compliance. [10]
Why This Matters Clinically
PUD remains a prevalent condition with substantial healthcare burden despite declining incidence in developed nations. The paradigm shift from chronic relapsing disease to curable infection with H. pylori eradication represents one of gastroenterology's major therapeutic advances, reducing ulcer recurrence from 60-80% annually to less than 5%. [11] However, NSAID-associated ulcers are increasing due to ageing populations requiring cardiovascular prophylaxis and chronic pain management. Bleeding peptic ulcers account for 40-50% of upper gastrointestinal haemorrhage cases, with 5-10% mortality in complicated cases—higher in elderly patients with comorbidities. [12] Appropriate risk stratification, prophylactic gastroprotection, and timely recognition of complications are essential for reducing morbidity and mortality.
2. Epidemiology
Global Burden
Temporal Trends:
- Lifetime prevalence: 5-10% in Western populations; higher (10-15%) in developing regions [4]
- Annual incidence: 0.1-0.3% (0.1-0.19 per 1000 person-years) [13]
- Declining incidence: 30-50% reduction over past 30 years in developed countries due to H. pylori eradication programmes and reduced NSAID prescribing [5]
- Hospitalisation for complications: Decreased from 60-80 per 100,000 (1990s) to 30-40 per 100,000 (2020s) [14]
Regional Variation:
- H. pylori prevalence: 70-90% in developing countries vs 20-40% in developed nations (declining with improved sanitation) [15]
- Gastric ulcer/duodenal ulcer ratio: Historically 1:4; now approaching 1:2 due to declining duodenal ulcer incidence [5]
Demographics
| Factor | Pattern | Clinical Significance |
|---|---|---|
| Age | Peak 50-70 years; mean age 55-65 | Complications more severe in elderly (bleeding mortality 10-15% vs 2-5% in younger) [12] |
| Sex | Historically M:F 2-3:1; now equalising (~1.2:1) | Female incidence rising with increased NSAID use for osteoarthritis |
| Socioeconomic Status | Inverse correlation (lower SES = higher risk) | H. pylori acquisition in childhood associated with crowding, poor sanitation [15] |
| Geography | Higher in East Asia, Eastern Europe, South America | Reflects H. pylori prevalence and virulence factors (CagA-positive strains) |
Aetiology Distribution
| Cause | Proportion | Key Risk Factors | Notes |
|---|---|---|---|
| H. pylori infection | 60-70% overall (90% DU, 70% GU historically) [1] | Childhood acquisition, low SES, crowded living conditions | Declining in developed nations; gastric cancer risk (1-2% lifetime with chronic infection) |
| NSAIDs/Aspirin | 20-30% | Age > 65, high dose, dual therapy, anticoagulants, steroids, SSRIs [9] | Dose-dependent; COX-1 inhibition impairs mucosal defence; low-dose aspirin (75-150mg) confers 2-4 fold risk |
| Idiopathic | 5-10% | True H. pylori-negative, NSAID-negative after rigorous exclusion | May represent undetected intermittent NSAID use, false-negative H. pylori testing, or acid hypersecretory states |
| Zollinger-Ellison syndrome | less than 1% (1-4% of refractory ulcers) [10] | Multiple endocrine neoplasia type 1 (MEN-1) | Gastrinoma causing acid hypersecretion; suspect with multiple/atypical ulcers, diarrhoea |
| Other rare causes | less than 1% | Crohn's disease, malignancy, viral (CMV, HSV), vascular insufficiency, radiation | Diagnostic consideration in refractory or atypical presentations |
3. Pathophysiology
Normal Gastroduodenal Mucosal Defence
The gastroduodenal mucosa maintains integrity despite exposure to hydrochloric acid (pH 1-2) and pepsin through multilayered protective mechanisms:
-
Pre-epithelial Defence:
- Mucus layer (200-300μm thick): Glycoprotein secretion traps bicarbonate, creating pH gradient (luminal pH 2 → surface epithelial pH 7)
- Bicarbonate secretion: Active transport neutralises acid at mucosal surface
- Phospholipid surfactant layer: Hydrophobic barrier preventing acid back-diffusion
-
Epithelial Defence:
- Tight junctions: Prevent paracellular acid penetration
- Rapid cell turnover: Complete epithelial renewal every 3-5 days
- Epithelial bicarbonate secretion: Additional neutralising capacity
- Heat shock proteins: Cytoprotection during stress
-
Post-epithelial Defence:
- Submucosal blood flow: Delivers bicarbonate, nutrients, and oxygen; removes acid
- Prostaglandin E2 (PGE2) and prostacyclin (PGI2): Vasodilation, bicarbonate secretion, mucus production
- Immune surveillance: Repair mechanisms and growth factors
Pathogenesis of H. pylori-Associated Ulcers
H. pylori is a microaerophilic Gram-negative spiral bacterium that colonises the gastric mucus layer. [16]
Key Virulence Mechanisms:
-
Urease Production:
- Converts urea → ammonia + CO₂
- Creates alkaline microenvironment (pH 6-7) enabling bacterial survival in acidic gastric lumen
- Ammonia directly toxic to epithelial cells
-
Flagellar Motility:
- Spiral morphology and flagella allow penetration of mucus layer
- Adherence to epithelial cells via adhesins (BabA, SabA)
-
Cytotoxin-Associated Gene A (CagA):
- Encoded by cag pathogenicity island (~60% of strains) [16]
- Injected into epithelial cells via type IV secretion system
- Disrupts cell signalling, cytoskeletal architecture → inflammation
- CagA-positive strains: 3-fold increased ulcer risk, 2-fold increased gastric cancer risk
-
Vacuolating Cytotoxin A (VacA):
- Induces epithelial cell apoptosis
- Forms membrane pores causing vacuolation
- Impairs T-cell function (immune evasion)
Duodenal Ulcer Pathogenesis (Antral-Predominant Gastritis):
- Antral inflammation → hypergastrinaemia (reduced somatostatin-secreting D cells)
- Increased gastric acid secretion (parietal cell stimulation)
- Gastric metaplasia in duodenal bulb (chronic acid exposure)
- H. pylori colonisation of metaplastic gastric epithelium → duodenitis → ulceration [17]
Gastric Ulcer Pathogenesis (Corpus-Predominant Gastritis):
- Pangastritis or corpus-predominant inflammation
- Parietal cell atrophy → hypochlorhydria (paradoxically)
- Impaired mucosal defence despite normal/low acid
- Direct mucosal injury from inflammation → ulceration
- Atrophic gastritis → intestinal metaplasia → dysplasia risk (gastric cancer pathway) [17]
Pathogenesis of NSAID-Associated Ulcers
NSAIDs cause dose-dependent mucosal injury through both topical and systemic mechanisms. [9]
Systemic (COX-Inhibition) Mechanisms (Predominant):
-
Cyclooxygenase-1 (COX-1) Inhibition:
- Reduced prostaglandin synthesis (PGE2, PGI2)
- Decreased mucus and bicarbonate secretion
- Reduced mucosal blood flow (prostaglandin-mediated vasodilation lost)
- Impaired epithelial proliferation and repair
- Increased neutrophil adherence to vascular endothelium
-
Platelet Inhibition:
- Reduced thromboxane A2 → impaired platelet aggregation
- Increased bleeding risk from existing ulcers
Topical Mechanisms:
- Mucosal contact injury: Lipophilic weak acids accumulate in epithelial cells
- Uncoupling oxidative phosphorylation → ATP depletion
- Disruption of phospholipid surfactant layer
COX-2 Selective Inhibitors:
- Reduced GI toxicity (50-60% risk reduction vs non-selective NSAIDs) [18]
- COX-2 expression upregulated during injury/repair (role in healing)
- Cardiovascular risk concerns (thrombotic events) limit use
Gastric vs Duodenal Ulcer: Distinguishing Features
| Feature | Gastric Ulcer | Duodenal Ulcer |
|---|---|---|
| Location | Lesser curve (60%), antrum (25%), cardia (10-15%) | First part duodenum (D1) - 95% within 3cm of pylorus; anterior wall (anterior perforate, posterior bleed) |
| Peak Age | 50-60 years | 30-50 years (earlier onset) |
| Pain Pattern | Epigastric; worsened/precipitated by eating (30-60 min post-meal) | Epigastric; relieved by food/antacids; recurs 2-3h later; nocturnal pain (2-3am) when stomach empty |
| Acid Secretion | Normal or reduced (atrophic gastritis) | Elevated in 30-50% (basal and stimulated) |
| H. pylori Association | 60-70% [1] | 90-95% in non-NSAID cases [1] |
| NSAID Association | 40-50% | 20-30% |
| Malignancy Risk | 2-5% are malignant (all gastric ulcers require biopsy) | Virtually zero (less than 0.1%; malignancy at ulcer site = lymphoma not adenocarcinoma) |
| Complications | Bleeding (20-25%), perforation (10%), obstruction (2%) | Bleeding (15-20%), perforation (5-7%), obstruction (2-5%) |
| Healing Time | 8-12 weeks (larger, slower healing) | 4-8 weeks |
| Surveillance | Mandatory repeat OGD at 6-8 weeks to confirm healing and exclude malignancy | Not routinely required (unless giant ulcer > 2cm or refractory) |
4. Clinical Presentation
Typical Symptoms
Epigastric Pain (80-90% of symptomatic cases):
- Character: Burning, gnawing, or aching
- Location: Epigastrium (midline or slightly left of midline)
- Temporal patterns:
- "Duodenal ulcer: Pain-food-relief pattern (pain when stomach empty → relieved by eating/antacids → recurs 2-3h later); nocturnal pain (awakens patient 1-3am) in 50-80% [8]"
- "Gastric ulcer: Pain-food-pain pattern (pain worsened by eating within 30-60min); less commonly nocturnal"
- Radiation: May radiate to back (penetrating posterior ulcers, especially pancreatic penetration)
- Duration: Chronic/relapsing (weeks to months); episodic flares
Associated Symptoms:
- Nausea (30-50%)
- Early satiety (especially gastric ulcers affecting fundus/body)
- Bloating, fullness
- Heartburn (overlapping GORD in 30-40%)
- Vomiting (suggests gastric outlet obstruction if persistent/projectile)
- Weight loss: Mild (food aversion in gastric ulcers); significant weight loss = alarm feature
Asymptomatic Presentation (10-20%):
- More common in elderly (blunted pain perception) and NSAID users (analgesic effect masks symptoms)
- Present with complications (bleeding, perforation) without preceding dyspepsia
Physical Examination
Uncomplicated PUD:
- Epigastric tenderness (50-70%): Non-specific; may be absent
- Usually benign examination otherwise
Signs Suggesting Complications:
| Complication | Clinical Signs | Additional Features |
|---|---|---|
| Acute Bleeding | Pallor, tachycardia (> 100bpm), hypotension (less than 90mmHg systolic), cool peripheries, prolonged CRT | Haematemesis (fresh blood or coffee-ground), melaena (black, tarry, offensive stools), haematochezia (rapid bleeding with haemodynamic instability) |
| Perforation | Sudden severe generalised pain → board-like rigidity, absent bowel sounds, shallow breathing, percussion tenderness, guarding/rebound | Lying still (peritonitic posture); may have pneumoperitoneum signs (loss of liver dullness) |
| Penetration | Severe, constant pain radiating to back; change in pain pattern (pain no longer food-related) | Posterior DU → pancreas (elevated amylase/lipase); GU → lesser omentum |
| Gastric Outlet Obstruction | Distended abdomen, visible gastric peristalsis (left-to-right waves), succussion splash (> 4h post-meal), dehydration signs | Hypochloraemic hypokalaemic metabolic alkalosis (vomiting HCl and K⁺) |
Red Flags / Alarm Features
[!CAUTION] Alarm Features Mandating Urgent Endoscopy (Within 2 Weeks): [7]
- Unintended weight loss (> 5% body weight over 6 months or > 3kg over 1 month)
- Progressive dysphagia (suggests oesophageal/gastric cardia involvement or malignancy)
- Gastrointestinal bleeding: Haematemesis, melaena, unexplained iron deficiency anaemia (ferritin less than 30μg/L)
- Persistent vomiting (risk of gastric outlet obstruction or malignancy)
- Palpable abdominal mass or epigastric lymphadenopathy (Troisier's sign = left supraclavicular node)
- Age ≥55 years with new-onset dyspepsia (less than 1 month symptom duration)
- Family history of upper GI malignancy (first-degree relative with gastric cancer, especially age less than 50 at diagnosis)
- Suspected malignancy on any grounds (e.g., clinical examination, imaging)
NICE Age Threshold Rationale: Gastric cancer incidence increases sharply > 55 years; pre-test probability of malignancy in dyspepsia: less than 0.5% under 55 vs 5-10% over 55. [7]
5. Differential Diagnosis
Key Differentials for Epigastric Pain
| Diagnosis | Distinguishing Features | Investigations |
|---|---|---|
| Gastro-oesophageal Reflux Disease (GORD) | Heartburn, regurgitation, worse lying flat/bending; endoscopy may show oesophagitis | OGD, pH monitoring |
| Functional Dyspepsia | Rome IV criteria: bothersome postprandial fullness/early satiety/epigastric pain ≥3 months; normal endoscopy | OGD (to exclude organic pathology) |
| Gastric Cancer | Progressive symptoms, alarm features, weight loss | OGD + biopsy (mandatory) |
| Zollinger-Ellison Syndrome | Severe/refractory ulcers, diarrhoea (secretory), multiple ulcers (post-bulbar) | Fasting gastrin (> 1000pg/mL diagnostic; 100-1000 requires secretin stimulation test) [10] |
| Pancreatitis | Acute/chronic; severe epigastric pain radiating to back, vomiting, elevated amylase/lipase (> 3× ULN) | Amylase, lipase, CT abdomen |
| Biliary Colic | RUQ/epigastric pain, colicky, post-prandial (fatty meals), nausea | Ultrasound abdomen, LFTs |
| Myocardial Infarction | Cardiac risk factors, exertional symptoms, radiation to arm/jaw | ECG, troponin |
| Mesenteric Ischaemia | Postprandial pain (intestinal angina), weight loss (food fear), age > 60, vascular disease | CT angiography |
| Crohn's Disease | Younger age, diarrhoea, weight loss, perianal disease; ulcers in terminal ileum or post-bulbar duodenum | OGD, colonoscopy, MR enterography |
6. Investigations
Non-Invasive H. pylori Testing (First-Line in less than 55 Years Without Alarm Features) [7]
| Test | Sensitivity | Specificity | Advantages | Limitations |
|---|---|---|---|---|
| Urea Breath Test (UBT) | 95-98% | 95-98% | Gold standard non-invasive test; detects active infection | Requires fasting (4-6h), PPI cessation ≥2 weeks, antibiotics ≥4 weeks; expensive equipment; false negative if low bacterial load |
| Stool Antigen Test (SAT) | 92-95% | 92-95% | Detects active infection; lower cost than UBT; suitable for eradication verification | PPI/antibiotic cessation required (as UBT); less validated in some regions |
| Serology (IgG ELISA) | 85-92% | 79-90% | Unaffected by PPI/antibiotics; cheap | Cannot distinguish active from past infection; remains positive post-eradication; not suitable for verification |
Testing Protocol:
- Stop PPIs ≥2 weeks before testing (reduces bacterial load → false negatives)
- Stop antibiotics, bismuth ≥4 weeks before testing
- Avoid serology for post-treatment verification
Endoscopy (OGD) - Gold Standard
Indications:
- Alarm features (any age)
- Age ≥55 with new-onset dyspepsia
- Persistent symptoms despite empirical PPI/eradication therapy
- NSAID users with symptoms (higher malignancy risk)
- Gastric ulcer (all require biopsy and follow-up OGD to confirm healing)
- Suspected complications (bleeding, obstruction)
Endoscopic Protocol:
Duodenal Ulcer:
- Visual assessment (location, size, stigmata of recent haemorrhage if bleeding)
- Biopsy NOT routinely required (malignancy extremely rare)
- H. pylori testing: Rapid urease test (CLO test/Campylobacter-like organism test) from antrum + body (2+ biopsies)
Gastric Ulcer:
- Mandatory multiple biopsies: ≥6-8 biopsies from ulcer edge and base (exclude malignancy—2-5% gastric ulcers are malignant) [19]
- H. pylori testing: Rapid urease test + histology (antrum and body)
- Document size, location, morphology (Bormann classification if suspicion of malignancy)
- Repeat OGD at 6-8 weeks: Confirm complete healing (persistent ulcer = malignancy until proven otherwise)
Forrest Classification (Bleeding Ulcer Endoscopy): [20]
| Forrest Grade | Description | Re-bleeding Risk | Mortality | Management |
|---|---|---|---|---|
| Ia | Spurting arterial haemorrhage | 90% | 11-15% | Urgent endoscopic haemostasis (dual therapy: injection + thermal/mechanical) |
| Ib | Oozing haemorrhage | 50% | 10-12% | Endoscopic haemostasis (dual therapy) |
| IIa | Visible vessel (non-bleeding) | 40-50% | 8-10% | Endoscopic haemostasis (dual therapy) |
| IIb | Adherent clot | 20-30% | 5-8% | Consider haemostasis (irrigation → if vessel underneath, treat as IIa) vs observation |
| IIc | Flat pigmented spot | 5-10% | 3-5% | High-dose PPI, no routine intervention |
| III | Clean ulcer base | less than 5% | less than 2% | PPI therapy |
Endoscopic Haemostasis Techniques:
- Injection therapy: Adrenaline 1:10,000 (1-2mL aliquots in 4 quadrants); vasoconstriction + tamponade; NEVER as monotherapy
- Thermal coagulation: Bipolar electrocoagulation, heater probe (coaptive coagulation)
- Mechanical therapy: Haemostatic clips, over-the-scope clips (OTSC) for large vessels > 2mm
- Combination therapy: Injection + thermal/mechanical (reduces re-bleeding from 18-19% to 7-10%) [21]
Rockall Score (Bleeding Peptic Ulcer Risk Stratification) [22]
Pre-Endoscopy Rockall Score (Clinical Variables):
| Variable | Score 0 | Score 1 | Score 2 | Score 3 |
|---|---|---|---|---|
| Age | less than 60 | 60-79 | ≥80 | - |
| Shock | SBP ≥100, HR less than 100 | SBP ≥100, HR ≥100 | SBP less than 100 | - |
| Comorbidity | None | - | Heart failure, IHD, major comorbidity | Renal failure, liver failure, metastatic cancer |
Post-Endoscopy Rockall Score (Add Endoscopic Variables):
| Variable | Score 0 | Score 1 | Score 2 | Score 3 |
|---|---|---|---|---|
| Diagnosis | Mallory-Weiss tear | All other diagnoses | Malignancy | - |
| Stigmata | Clean base (Forrest III) | - | Blood in upper GI, adherent clot, visible/spurting vessel | - |
Score Interpretation:
- 0-2: Low risk; mortality less than 0.2%; consider outpatient management
- 3-4: Moderate risk; mortality 3-5%
- 5-7: High risk; mortality 11-24%
- ≥8: Very high risk; mortality > 40%
Glasgow-Blatchford Score (GBS) for Safe Discharge: [23]
GBS = 0 (all criteria met) → Safe for outpatient management with early follow-up:
- Urea less than 6.5mmol/L
- Hb ≥130g/L (men) or ≥120g/L (women)
- Systolic BP ≥110mmHg
- Pulse less than 100bpm
- No melaena
- No syncope
- No cardiac failure
- No hepatic disease
GBS ≥1: Requires admission; likely intervention (endoscopy/transfusion)
Laboratory Investigations
| Test | Purpose | Findings |
|---|---|---|
| Full Blood Count (FBC) | Anaemia screening | Microcytic anaemia (iron deficiency from chronic blood loss); acute bleed may show normal Hb initially (haemodilution takes 24-72h) |
| Urea & Electrolytes | Metabolic assessment in complications | Elevated urea disproportionate to creatinine (GI bleeding; urea absorbed from digested blood); hypokalaemic hypochloraemic alkalosis (gastric outlet obstruction) |
| Liver Function Tests | Exclude hepatobiliary disease | Hypoalbuminaemia (chronic disease, malnutrition) |
| Iron Studies | Iron deficiency assessment | Low ferritin (less than 30μg/L), low serum iron, high TIBC, low transferrin saturation |
| Coagulation Screen | Pre-procedure (if bleeding); assess bleeding risk | Prolonged PT/APTT in anticoagulated patients |
| Group & Save / Crossmatch | If active bleeding | Prepare for transfusion |
Specialist Investigations (Selected Cases)
| Investigation | Indication | Interpretation |
|---|---|---|
| Fasting Serum Gastrin | Refractory ulcers, multiple ulcers, post-bulbar DU, secretory diarrhoea, MEN-1 features | > 1000pg/mL diagnostic of Zollinger-Ellison syndrome; 100-1000pg/mL requires secretin stimulation test (paradoxical rise > 200pg/mL positive); remember PPI causes physiological hypergastrinaemia (stop 2 weeks before test if safe) [10] |
| Secretin Stimulation Test | Gastrin 100-1000pg/mL | IV secretin 2μg/kg → gastrin rise > 200pg/mL within 15min = positive |
| Chromogranin A | Neuroendocrine tumour screening (gastrinoma) | Elevated in gastrinomas; also raised by PPI use, renal failure |
| Abdominal CT/MRI | Localise gastrinoma; assess metastases | Gastrinomas: 80% duodenal, 20% pancreatic; 60% malignant |
| Somatostatin Receptor Scintigraphy (Octreoscan) | Gastrinoma localisation | Detects somatostatin receptor-expressing neuroendocrine tumours |
| CT Abdomen | Suspected perforation; assess penetration | Pneumoperitoneum (free air); localise fluid collections |
| Erect Chest X-Ray | Perforation screening | Free air under diaphragm (sensitivity 70-80%; CT more sensitive) |
7. Management
General Principles
- Identify and address aetiology: H. pylori eradication, NSAID cessation/gastroprotection
- Acid suppression: PPI therapy to promote ulcer healing
- Complication management: Bleeding, perforation, obstruction
- Verify healing: Repeat endoscopy for gastric ulcers
- Confirm eradication: Post-treatment H. pylori testing
- Prevent recurrence: Long-term PPI if ongoing NSAID/aspirin use
H. pylori Eradication Therapy
Regional Antibiotic Resistance Considerations:
- Clarithromycin resistance: 10-40% globally (increasing); > 15% in many European countries [6]
- Metronidazole resistance: 20-50% globally
- Levofloxacin resistance: 10-30% (rising)
- Treatment selection should consider local resistance patterns
First-Line Regimens (Choose Based on Local Resistance):
| Regimen | Components | Duration | Eradication Rate | Indications |
|---|---|---|---|---|
| Clarithromycin Triple Therapy | PPI (standard dose BD) + Clarithromycin 500mg BD + Amoxicillin 1g BD | 7-14 days (14 days preferred) [2] | 80-85% (lower if clarithromycin resistance > 15%) [6] | Areas with low clarithromycin resistance (less than 15%); penicillin-tolerant patients |
| Alternative Triple Therapy | PPI (standard dose BD) + Clarithromycin 500mg BD + Metronidazole 400mg BD | 7-14 days | 75-80% (lower if metronidazole resistance) | Penicillin allergy |
| Bismuth Quadruple Therapy | PPI (standard dose BD) + Bismuth subsalicylate/subcitrate 120mg QDS + Metronidazole 400-500mg TDS + Tetracycline 500mg QDS | 10-14 days [2] | 85-90% (effective despite metronidazole resistance) [24] | First-line alternative; high clarithromycin resistance areas; previous macrolide exposure |
| Concomitant Therapy | PPI (standard dose BD) + Clarithromycin 500mg BD + Amoxicillin 1g BD + Metronidazole 500mg BD | 10-14 days | 85-90% | High dual resistance (clarithromycin + metronidazole) |
PPI Standard Doses (BD Dosing):
- Omeprazole 20mg BD
- Lansoprazole 30mg BD
- Esomeprazole 20mg BD
- Pantoprazole 40mg BD
- Rabeprazole 20mg BD
Second-Line (Rescue) Therapy After First-Line Failure:
| Regimen | Components | Duration | Eradication Rate | Notes |
|---|---|---|---|---|
| Bismuth Quadruple Therapy (if not used first-line) | As above | 14 days | 75-85% | Avoid metronidazole if used in first-line; substitute tinidazole |
| Levofloxacin Triple Therapy | PPI (standard dose BD) + Levofloxacin 500mg OD + Amoxicillin 1g BD | 10-14 days | 70-85% (lower if fluoroquinolone resistance) [25] | Increasing resistance; risk of C. difficile; avoid if prior fluoroquinolone exposure |
| High-Dose Dual Therapy | PPI (high dose: e.g., rabeprazole 20mg QDS) + Amoxicillin 750mg QDS | 14 days | 70-80% | Emerging option; fewer antibiotics |
Third-Line Therapy:
- Culture and antibiotic susceptibility testing (via endoscopic biopsy)
- Tailored regimen based on resistance profile
- Consider referral to specialist gastroenterology
Verification of Eradication (Mandatory):
- Test ≥4 weeks after completing antibiotic therapy
- Stop PPI ≥2 weeks before testing (to avoid false negatives)
- UBT or stool antigen test (not serology)
- If positive: Second-line therapy
- Eradication success eliminates ulcer recurrence risk (less than 5% annual recurrence vs 60-80% without eradication) [11]
Proton Pump Inhibitor (PPI) Therapy
Ulcer Healing:
- Duodenal ulcer: PPI standard dose OD for 4 weeks (healing rate > 90%)
- Gastric ulcer: PPI standard dose OD for 8 weeks (larger ulcers; slower healing)
- Giant ulcers (> 2cm): Consider 12 weeks therapy
Post-Eradication:
- PPIs can be stopped once H. pylori eradicated (unless NSAID continuation or refractory symptoms)
- No evidence that long-term PPI reduces recurrence after successful eradication
Long-Term PPI Indications:
- NSAID continuation (especially if high-risk: age > 65, prior ulcer, anticoagulation)
- Dual antiplatelet therapy (aspirin + clopidogrel/ticagrelor)
- Recurrent ulcers despite H. pylori eradication
- Acid hypersecretory states (Zollinger-Ellison syndrome)
PPI Safety Considerations (Long-Term Use > 1 Year):
- Hypomagnesaemia: Check magnesium if chronic use; supplement if less than 0.7mmol/L
- Osteoporosis/fracture risk: Modest increased risk (RR 1.2-1.5); consider bone protection in high-risk patients
- Clostridium difficile infection: 1.5-2-fold increased risk
- Community-acquired pneumonia: Small increased risk
- Vitamin B12 deficiency: Consider monitoring in prolonged use
- Chronic kidney disease: Observational associations (causality unclear)
- Interactions: Reduced clopidogrel activation (use pantoprazole/rabeprazole; avoid omeprazole/esomeprazole)
NSAID-Related Ulcer Management
Step 1: Risk Stratification
High GI Risk Factors (Require Prophylaxis): [9]
- Age > 65 years
- Prior peptic ulcer or GI bleeding
- Concurrent medications: Anticoagulants (warfarin, DOACs), corticosteroids, SSRIs, dual antiplatelet therapy
- High-dose NSAID or dual NSAID use
- Severe comorbidity (cardiovascular disease, renal failure)
Step 2: NSAID Management
| Clinical Scenario | Management Strategy |
|---|---|
| Active ulcer on NSAID | Stop NSAID immediately; PPI standard dose for 8 weeks; H. pylori test and treat if positive; switch to paracetamol or consider alternative (topical NSAIDs for musculoskeletal conditions) |
| Cannot stop NSAID (e.g., severe RA) | Switch to COX-2 selective inhibitor (celecoxib 200mg OD/BD) + PPI (dual protection); monitor cardiovascular risk [18] |
| Prevention in high-risk patient requiring NSAID | Co-prescribe PPI standard dose (80% risk reduction) [26]; alternatively: COX-2 inhibitor alone (50% risk reduction); combination COX-2 + PPI for very high-risk |
| Low-dose aspirin (cardiovascular prophylaxis) | Continue aspirin (stopping increases cardiovascular events); co-prescribe PPI standard dose OD; verify H. pylori eradication |
| Dual antiplatelet therapy (DAPT) | Mandatory PPI co-prescription (bleeding risk 2-3%/year); use pantoprazole/rabeprazole (avoid omeprazole with clopidogrel) |
COX-2 Inhibitors:
- Celecoxib 200mg OD/BD: 50-60% reduction in ulcer risk vs non-selective NSAIDs [18]
- Cardiovascular contraindications: Recent MI, stroke, uncontrolled hypertension, heart failure
- Not suitable if concurrent aspirin use (negates GI benefit)
Alternative Analgesic Options:
- Paracetamol 1g QDS (first-line for osteoarthritis)
- Topical NSAIDs (diclofenac gel, ibuprofen gel) for localised musculoskeletal pain
- Tramadol (opioid; avoid long-term due to dependence risk)
- Intra-articular corticosteroid injections (knee/shoulder osteoarthritis)
Idiopathic Ulcers (H. pylori-Negative, NSAID-Negative)
Diagnostic Approach:
- Confirm true NSAID-negative status (exclude intermittent use, aspirin, herbal NSAIDs)
- Re-test H. pylori (UBT, stool antigen, histology if OGD repeated)
- Measure fasting gastrin (exclude Zollinger-Ellison syndrome) [10]
- Review medications (bisphosphonates, potassium chloride, chemotherapy)
Management:
- PPI standard dose for 8 weeks (gastric ulcer) or 4 weeks (duodenal ulcer)
- Long-term PPI if recurrent
- Consider gastric acid analysis if refractory (basal acid output, maximal acid output)
8. Complications
1. Gastrointestinal Bleeding (15-20% of PUD Cases) [3]
Clinical Presentation:
- Haematemesis: Vomiting fresh blood (arterial bleeding) or coffee-ground material (blood exposed to gastric acid)
- Melaena: Black, tarry, foul-smelling stools (upper GI bleed; digested blood)
- Haematochezia: Fresh rectal bleeding (massive upper GI bleed with rapid transit; or lower GI source)
- Haemodynamic instability: Tachycardia, hypotension, shock (blood loss > 1000mL)
- Syncope, presyncope, confusion (especially elderly)
Management Protocol:
Initial Resuscitation (ABCDE Approach):
- Airway: Protect if reduced GCS (risk of aspiration)
- Breathing: Oxygen if SpO₂ less than 94%
- Circulation:
- Two large-bore IV cannulae (14-16G)
- Crystalloid resuscitation (500mL boluses; target systolic BP ≥100mmHg)
- Transfusion threshold: Hb less than 70g/L (restrictive strategy reduces mortality vs liberal) [27]
- Target Hb 70-90g/L (higher target if acute coronary syndrome)
- Activate major haemorrhage protocol if massive bleeding (> 1500mL or > 50% blood volume)
- Blood tests: FBC, U&E, LFT, coagulation, group & save/crossmatch, venous lactate
- Correct coagulopathy: Reverse warfarin (prothrombin complex concentrate [PCC] + vitamin K if INR > 1.5); consider platelet transfusion if thrombocytopenia + active bleeding
Pharmacological Therapy:
- High-dose PPI infusion: Omeprazole 80mg IV bolus, then 8mg/hour continuous infusion for 72 hours [28]
- Reduces re-bleeding risk, need for surgery; administer pre-endoscopy
- Prokinetic: Erythromycin 250mg IV 30-60min pre-endoscopy (enhances gastric emptying, improves visualisation)
- Tranexamic acid: NOT recommended (HALT-IT trial showed no benefit, possible harm) [29]
Endoscopy:
- Timing: Within 24 hours for most; immediate (less than 2h) if haemodynamic instability despite resuscitation [12]
- Forrest classification assessment (see Section 6)
- Endoscopic haemostasis (Forrest Ia-IIa; consider IIb):
- "Combination therapy: Injection + thermal/mechanical (superior to monotherapy; reduces re-bleeding from 18-19% to 7-10%) [21]"
- "Injection therapy: Adrenaline 1:10,000 (1-2mL aliquots in 4 quadrants around bleeding point; vasoconstriction + tamponade; NEVER as monotherapy)"
- "Thermal coagulation: Bipolar electrocoagulation, heater probe (coaptive coagulation)"
- "Mechanical therapy: Haemostatic clips (over-the-scope clips for large vessels > 2mm)"
- Re-bleeding rate post-haemostasis: 10-20%
Surgery (If Endoscopic Haemostasis Fails or Not Feasible):
- Indications: Re-bleeding after 2 attempts endoscopic therapy, haemodynamic instability despite resuscitation, inability to localise/access bleeding point
- Procedure: Duodenotomy + under-running of bleeding vessel (duodenal ulcer); gastrectomy (gastric ulcer if large/malignancy concern)
- Mortality: 10-30% (high-risk patients)
Secondary Prevention:
- PPI standard dose BD for 2 weeks, then OD for 6-8 weeks
- H. pylori eradication if positive
- Stop NSAIDs; restart with PPI prophylaxis only if essential
- Resume anticoagulation/antiplatelet therapy: Balance bleeding vs thrombotic risk; restart within 7 days if cardiovascular indication (discuss with cardiology) [12]
2. Perforation (5-10% of PUD Cases) [3]
Clinical Presentation:
- Sudden severe epigastric pain: "Worst pain of my life"; knife-like; may radiate to shoulders (diaphragmatic irritation)
- Peritonism: Board-like rigidity, guarding, rebound tenderness, absent bowel sounds
- Systemic Features: Tachycardia, hypotension (septic shock), fever
- Patient Posture: Lying still, knees drawn up (movement exacerbates peritoneal irritation)
Imaging:
- Erect CXR: Pneumoperitoneum (free air under diaphragm) in 70-80%; may be absent if small perforation or delayed presentation
- CT Abdomen/Pelvis (gold standard): Sensitivity > 95%; localises perforation site; assesses contamination/abscess
- Water-soluble contrast study: If diagnostic uncertainty (e.g., sealed perforation)
Management:
Non-Operative (Conservative) Management:
- Selected cases: Small sealed perforations, minimal free fluid, haemodynamically stable, no peritonism
- Criteria: Age less than 70, presentation less than 24h, no shock, localised signs
- Protocol: Nil by mouth, IV fluids, nasogastric decompression, IV broad-spectrum antibiotics (cover Gram-negatives and anaerobes: piperacillin-tazobactam 4.5g TDS or cefuroxime 1.5g TDS + metronidazole 500mg TDS), high-dose PPI (80mg omeprazole IV bolus then 8mg/h)
- Serial clinical review; convert to surgery if deterioration
Operative Management (Majority of Cases):
| Approach | Procedure | Indications |
|---|---|---|
| Laparoscopic Repair | Washout, omental patch (Graham patch), peritoneal lavage, drain placement | Most perforated DU; perforation less than 24h; no malignancy suspicion |
| Open Repair | Laparotomy + omental patch | Haemodynamic instability, delayed presentation (> 24h), large perforation, failed laparoscopy |
| Definitive Ulcer Surgery (Rare) | Truncal vagotomy + pyloroplasty or antrectomy | Refractory ulcer disease; not commonly performed (PPI era); high operative risk |
| Gastrectomy | Partial/total gastrectomy | Gastric ulcer perforation with malignancy suspicion |
Postoperative Management:
- IV antibiotics (5-7 days)
- High-dose PPI (80mg omeprazole IV then 8mg/h for 72h, then oral standard dose for 8 weeks)
- H. pylori testing (antrum/body biopsies if surgery; post-op UBT/stool antigen if not biopsied) and eradication therapy
- NSAID cessation
- Follow-up OGD (gastric ulcer) at 6-8 weeks
Mortality:
- Overall: 10-30%
- Increased risk: Age > 65, delayed presentation (> 24h), shock, comorbidities (ASA ≥III)
3. Gastric Outlet Obstruction (2-5% of PUD Cases) [3]
Pathophysiology:
- Chronic inflammation → oedema/spasm (acute, reversible with PPI)
- Repeated ulceration/healing → fibrotic stricture (pyloric channel or duodenal bulb)
Clinical Presentation:
- Postprandial vomiting (large-volume, undigested food, no bile if pre-pyloric)
- Early satiety, abdominal bloating
- Weight loss (10-20kg over weeks/months)
- Dehydration
- Hypokalaemic hypochloraemic metabolic alkalosis (loss of gastric HCl and K⁺)
Examination:
- Epigastric distension
- Visible gastric peristalsis (left-to-right waves across upper abdomen)
- Succussion splash (> 4 hours post-meal: shake patient gently → audible fluid splash)
Investigations:
- Bloods: Hypokalaemia (K⁺ less than 3.5mmol/L), hypochloraemia (Cl⁻ less than 95mmol/L), metabolic alkalosis (HCO₃⁻ > 30mmol/L), elevated urea (dehydration)
- OGD: Visualise stricture, assess severity, exclude malignancy (biopsy if gastric), H. pylori testing
- Barium meal (if OGD incomplete): String sign (narrow pyloric channel)
- CT Abdomen: Gastric dilatation, wall thickening, exclude malignancy
Management:
Initial (Acute Phase):
- Nil by mouth, IV fluid resuscitation (normal saline + potassium supplementation: 40mmol KCl per litre)
- Nasogastric decompression: Large-bore NG tube (14-16Fr), free drainage
- High-dose PPI: IV omeprazole 80mg bolus then 8mg/h (reduce oedema)
- Correct electrolytes: Monitor K⁺, Cl⁻, HCO₃⁻ daily; replace aggressively
- H. pylori eradication if positive
Definitive Treatment (Once Stabilised):
| Modality | Procedure | Success Rate | Indications |
|---|---|---|---|
| Endoscopic Balloon Dilatation | Through-the-scope balloon (12-20mm diameter), gradual inflation to 60psi, held 60-120 seconds | 70-80% initial success; 50% long-term (recurrence common) | First-line for benign strictures; reversible component (oedema + fibrosis) |
| Surgery | Truncal vagotomy + gastrojejunostomy (bypass); truncal vagotomy + antrectomy (resection) | > 90% long-term success | Failed endoscopic therapy, malignancy, severe fibrosis |
Prognosis:
- Benign obstruction: Good with treatment; mortality less than 5%
- Malignant obstruction (gastric cancer): Palliative stenting or surgical bypass; poor prognosis
4. Penetration
Definition: Ulcer erodes through gastric/duodenal wall into adjacent organ (without free perforation into peritoneal cavity)
Common Sites:
- Posterior duodenal ulcer → pancreas (most common)
- Lesser curve gastric ulcer → lesser omentum, liver
Clinical Features:
- Change in pain pattern: Constant, severe, radiating to back (no longer food-related)
- Elevated amylase/lipase (pancreatic penetration)
- Refractory to PPI therapy
Imaging:
- CT Abdomen: Ulcer crater extending beyond gastric/duodenal wall, inflammatory changes in adjacent organ
Management:
- High-dose PPI (may heal medically)
- Surgery if refractory (excision + definitive ulcer surgery)
9. Prognosis & Outcomes
Ulcer Healing Rates
| Intervention | Duodenal Ulcer Healing (4 Weeks) | Gastric Ulcer Healing (8 Weeks) |
|---|---|---|
| PPI therapy | > 90% | > 80% |
| H2-receptor antagonist | 70-80% | 60-70% |
| No treatment | 20-30% (spontaneous healing) | 10-20% |
Recurrence Rates
| Scenario | Annual Recurrence Rate |
|---|---|
| Untreated | 60-80% [11] |
| H. pylori eradicated | less than 5% [11] |
| NSAID continued without PPI | 40-50% |
| NSAID + PPI prophylaxis | 5-10% [26] |
| Idiopathic (no identified cause) | 30-40% (consider long-term PPI) |
Complication Outcomes
| Complication | Mortality | Key Prognostic Factors |
|---|---|---|
| Bleeding | 5-10% overall; 2-5% (less than 60 years), 10-15% (> 60 years) [12] | Age, shock at presentation, comorbidities, rebleeding, Rockall score > 5 |
| Perforation | 10-30% [3] | Age > 65, delayed presentation (> 24h), shock, ASA grade ≥III |
| Gastric Outlet Obstruction | less than 5% (benign); > 50% (malignant) | Underlying aetiology (benign vs malignancy) |
Re-bleeding Risk Stratification
Factors Predicting Re-bleeding After Endoscopic Haemostasis: [30]
- Forrest Ia/Ib classification (active bleeding at index endoscopy)
- Ulcer size > 2cm diameter
- Posterior duodenal ulcer (proximity to gastroduodenal artery)
- Haemodynamic shock at presentation (SBP less than 90mmHg)
- Haemoglobin less than 80g/L
- Comorbidities (cardiovascular disease, renal failure)
Re-bleeding Rate:
- Overall post-endoscopic haemostasis: 10-20%
- High-risk lesions (Forrest Ia): 15-25% despite intervention
- Low-risk lesions (Forrest IIc, III): less than 5%
Management of Re-bleeding:
- Repeat endoscopy (second attempt haemostasis): Success rate 70-80%
- Interventional radiology: Transcatheter arterial embolisation (TAE) of gastroduodenal/left gastric artery—alternative to surgery, especially in high surgical risk patients
- Surgery: If two failed endoscopic attempts or unstable despite resuscitation
Long-Term Considerations
H. pylori and Gastric Cancer:
- Chronic H. pylori infection: 1-2% lifetime gastric cancer risk [16]
- Atrophic gastritis → intestinal metaplasia → dysplasia → adenocarcinoma (Correa cascade)
- Eradication before development of atrophic gastritis/intestinal metaplasia reduces cancer risk
- Surveillance endoscopy not routinely recommended post-eradication (unless extensive intestinal metaplasia/dysplasia at baseline)
NSAID Ulcers:
- Recurrence preventable with PPI prophylaxis or COX-2 inhibitors
- Balance GI risk vs cardiovascular benefit of aspirin (continue aspirin with PPI in most cases)
10. Evidence & Guidelines
Key Guidelines
1. NICE CG184: Gastro-oesophageal Reflux Disease and Dyspepsia in Adults (2014, Updated 2019) [7]
- Recommends test-and-treat strategy (less than 55 years, no alarm features)
- Urgent OGD for alarm features or age ≥55 with new-onset dyspepsia
- Carbon-13 urea breath test or stool antigen test for non-invasive H. pylori testing
- Avoid routine endoscopy in uncomplicated dyspepsia less than 55 years
2. Maastricht VI/Florence Consensus Report (2022) [2]
- European consensus on H. pylori management
- First-line: 14-day clarithromycin triple therapy (if resistance less than 15%) or bismuth quadruple therapy
- Verification of eradication mandatory (UBT or stool antigen ≥4 weeks post-treatment)
- Culture-guided therapy after second-line failure
3. ACG Clinical Guideline: Treatment of Helicobacter pylori Infection (2017) [24]
- Recommends bismuth quadruple therapy or concomitant therapy as first-line in areas with clarithromycin resistance > 15%
- Emphasises regional antibiotic resistance surveillance
- Sequential therapy no longer recommended (inferior to concomitant)
4. ESGE Guideline: Management of Non-Variceal Upper GI Bleeding (2021) [12]
- Glasgow-Blatchford score for risk stratification (GBS 0 = safe outpatient management)
- High-dose PPI (80mg bolus + 8mg/h infusion) pre- and post-endoscopy
- Endoscopy within 24h (urgent less than 2h if haemodynamic instability)
- Restrictive transfusion strategy (Hb target 70-90g/L)
- Combination endoscopic therapy (injection + thermal/mechanical) superior to monotherapy
Landmark Studies
1. Marshall & Warren (1984) - Discovery of H. pylori [16]
- Nobel Prize 2005: Revolutionised understanding of PUD aetiology
- Paradigm shift from chronic relapsing disease to curable infection
2. Hopkins et al. Meta-Analysis (1996) - H. pylori Eradication Reduces Recurrence
- Ulcer recurrence: 60-80% untreated vs less than 5% post-eradication [11]
- Established eradication therapy as standard of care
3. Chan et al. (2002) - PPI vs COX-2 Inhibitor for NSAID Ulcer Prevention
- RCT: Celecoxib vs diclofenac + omeprazole in high-risk patients
- Recurrent ulcer bleeding: 4.9% (celecoxib) vs 0.9% (diclofenac + PPI); PPI superior [18]
4. Lau et al. (2000) - High-Dose PPI Infusion Post-Endoscopic Haemostasis [28]
- RCT: Omeprazole 80mg bolus + 8mg/h × 72h vs placebo after endoscopic haemostasis
- Re-bleeding: 6.7% (omeprazole) vs 22.5% (placebo); Pless than 0.001
- Established high-dose PPI as standard post-haemostasis
5. HALT-IT Trial (2020) - Tranexamic Acid in GI Bleeding [29]
- Large RCT: Tranexamic acid vs placebo in acute GI bleeding
- No reduction in death due to bleeding; possible increased thromboembolic events
- Tranexamic acid NOT recommended for GI bleeding
6. Villanueva et al. (2013) - Restrictive Transfusion in GI Bleeding [27]
- RCT: Restrictive (Hb less than 70g/L) vs liberal (Hb less than 90g/L) transfusion threshold
- Mortality: 5% (restrictive) vs 9% (liberal); P=0.02
- Further bleeding: 10% vs 16%; P=0.01
- Restrictive strategy superior
7. Calvet et al. (2004) - Dual vs Single Endoscopic Therapy for Bleeding Ulcers [21]
- Meta-analysis: Combination therapy (injection + thermal/mechanical) vs monotherapy
- Combination therapy reduced re-bleeding from 18.4% to 10.6% (OR 0.53, Pless than 0.001)
- Reduced surgery and mortality
8. Rockall et al. (1996) - Risk Scoring in Upper GI Bleeding [22]
- Prospective study of 4185 patients with upper GI bleeding
- Developed Rockall score: clinical variables + endoscopic findings
- Score ≥5 associated with mortality 11-24%; ≥8 associated with > 40% mortality
- Validated tool for risk stratification
Evidence Strength Summary
| Intervention | Level of Evidence | Key References |
|---|---|---|
| H. pylori eradication for PUD | 1a (Cochrane meta-analyses) | Hopkins 1996 [11], Ford 2020 [6] |
| PPI for ulcer healing | 1a (multiple RCTs) | Cochrane reviews |
| High-dose PPI in bleeding ulcers | 1a (RCTs) | Lau 2000 [28], Leontiadis 2006 |
| PPI gastroprotection with NSAIDs | 1a (RCTs, meta-analyses) | Chan 2002 [18], Rostom 2002 [26] |
| Endoscopic haemostasis (combination therapy) | 1a (RCTs, meta-analyses) | Calvet 2004 [21], ESGE guidelines [12] |
| Restrictive transfusion strategy | 1a (RCT) | Villanueva 2013 [27] |
| Forrest classification prognostic value | 2a (prospective cohort studies) | Forrest 1974 [20] |
| Rockall score prognostic value | 2a (prospective cohort studies) | Rockall 1996 [22] |
| Test-and-treat strategy | 2a (cohort studies, guidelines) | NICE CG184 [7] |
11. Special Populations
Elderly Patients (Age > 65)
Key Considerations:
- Higher complication rates (bleeding, perforation)
- Atypical presentations (minimal symptoms until complications)
- Polypharmacy (NSAID, aspirin, anticoagulants, corticosteroids increase risk)
- Comorbidities increase surgical mortality (ASA ≥III)
- Lower H. pylori eradication rates (poorer compliance, drug interactions)
Management Modifications:
- Low threshold for endoscopy (alarm features broader)
- Cautious transfusion in bleeding (consider cardiac comorbidities; higher Hb target 80-90g/L if ACS)
- Early surgical consultation if bleeding refractory to endoscopy (operative risk high but delay worsens outcomes)
- Medication review: Stop NSAIDs if possible; rationalise polypharmacy
Pregnancy
H. pylori Eradication:
- Defer eradication therapy until postpartum (avoid antibiotic exposure in first trimester)
- PPI therapy generally safe (omeprazole, lansoprazole category B)
- Avoid bismuth (teratogenic), tetracycline (teeth/bone effects), fluoroquinolones
Bleeding Ulcers:
- Endoscopy safe in pregnancy (second trimester preferred)
- Conservative transfusion approach (balance maternal/fetal oxygen delivery)
- Multidisciplinary approach (obstetrics, gastroenterology)
Anticoagulated/Antiplatelet Patients
Bleeding Risk:
- Warfarin: 2-3-fold increased bleeding risk
- DOACs (dabigatran, rivaroxaban, apixaban): Similar risk to warfarin
- Dual antiplatelet therapy (DAPT): 2-3%/year major bleeding risk
Management:
- Mandatory PPI prophylaxis (reduces bleeding 50-80%) [26]
- H. pylori test and treat (eradication reduces bleeding risk)
- Restart anticoagulation post-bleeding: Balance thrombotic vs bleeding risk; typically resume within 7 days if cardiovascular indication (e.g., atrial fibrillation with CHA₂DS₂-VASc ≥2, mechanical heart valve, recent VTE) [12]
- Discuss with cardiology/haematology
12. Exam-Focused Pearls
Common MRCP/FRACP Questions
Q1: "A 45-year-old with epigastric pain asks about testing for H. pylori. What would you recommend?"
Model Answer: "I would first assess for alarm features—unintended weight loss, dysphagia, GI bleeding, or persistent vomiting. As the patient is under 55 without alarm features, NICE guidelines recommend a test-and-treat strategy using non-invasive testing—either urea breath test or stool antigen test—which are both highly sensitive (> 95%). It's crucial the patient stops PPIs for at least 2 weeks before testing to avoid false negatives. If positive, I'd prescribe 14-day eradication therapy, typically clarithromycin triple therapy in areas with low resistance, and verify eradication 4 weeks post-treatment with repeat UBT or stool antigen."
Q2: "Describe the Forrest classification and its clinical significance."
Model Answer: "The Forrest classification stratifies bleeding peptic ulcers based on endoscopic appearance and predicts re-bleeding risk. Forrest Ia (spurting arterial bleeding) and Ib (oozing) are active bleeding with 90% and 50% re-bleeding risk respectively—both require urgent dual-modality endoscopic therapy with injection plus thermal or mechanical haemostasis. Forrest IIa (visible vessel) carries 40-50% re-bleeding risk and also requires intervention. IIb (adherent clot) is controversial—if irrigated and a vessel is underneath, treat as IIa; otherwise, high-dose PPI alone. IIc (pigmented spot) and III (clean base) carry low risk (less than 10%) and don't require endoscopic intervention, just PPI therapy."
Q3: "How would you manage a patient on dual antiplatelet therapy who develops a bleeding duodenal ulcer?"
Model Answer: "This is a challenging scenario balancing bleeding and thrombotic risk. Initial management follows the ABCDE approach—resuscitation, high-dose PPI infusion (omeprazole 80mg bolus then 8mg/h), and urgent endoscopy for combination haemostasis. I'd temporarily hold antiplatelet therapy during active bleeding. The critical decision is when to restart—this requires discussion with cardiology to assess thrombotic risk. For high-risk patients (recent stent, ACS), resume within 3-5 days; for lower risk, within 7 days. Long-term, mandatory PPI prophylaxis is required—I'd use pantoprazole or rabeprazole to avoid omeprazole's interaction with clopidogrel. I'd also test and treat H. pylori, as eradication significantly reduces re-bleeding risk."
Q4: "What is the Rockall score and how do you interpret it?"
Model Answer: "The Rockall score predicts mortality in upper GI bleeding using both clinical and endoscopic variables. The pre-endoscopy score includes age, shock (BP, pulse), and comorbidities. Post-endoscopy, we add diagnosis and stigmata of bleeding. A score of 0-2 indicates low risk with mortality less than 0.2%—these patients may be suitable for early discharge with outpatient follow-up. Scores 3-4 indicate moderate risk (3-5% mortality), 5-7 high risk (11-24% mortality), and ≥8 very high risk (> 40% mortality). High scores mandate intensive monitoring, early senior involvement, and consideration of interventional radiology or surgery if endoscopic therapy fails. It's important to note the Glasgow-Blatchford score is better for initial triage—a score of 0 is highly sensitive for identifying patients safe for outpatient management."
Viva Readiness
Opening Statement (Peptic Ulcer Disease): "Peptic ulcer disease represents mucosal ulceration extending through the muscularis mucosae of the stomach or duodenum, predominantly caused by Helicobacter pylori infection in 60-70% of cases and NSAID use in 20-30%. The condition is characterised by epigastric pain with distinctive temporal patterns—duodenal ulcers typically present with pain-food-relief, while gastric ulcers demonstrate pain-food-pain. Management is predicated on identifying and treating the underlying cause, with eradication therapy achieving less than 5% recurrence compared to 60-80% without treatment. Major complications—bleeding, perforation, and obstruction—occur in approximately 20-30% of cases and carry significant mortality, particularly in elderly populations."
High-Yield Facts for Vivas:
- Forrest Ia/Ib: Require combination endoscopic therapy (injection + thermal/mechanical)
- Rockall > 5: Mortality 11-24%; consider early ICU/HDU
- GBS = 0: Safe for outpatient management (100% sensitivity for intervention need)
- Eradication verification: Mandatory at ≥4 weeks; PPI cessation ≥2 weeks before testing
- Clarithromycin resistance > 15%: Use bismuth quadruple or concomitant therapy first-line
- NSAID + anticoagulation: Absolute indication for PPI prophylaxis
- Gastric ulcer biopsy: Mandatory ≥6-8 biopsies (2-5% malignant); repeat OGD at 6-8 weeks
- Re-bleeding post-endoscopy: 10-20%; second attempt endoscopy or consider interventional radiology/surgery
Common Pitfalls (What Fails Candidates)
❌ Using serology to verify H. pylori eradication (remains positive; only UBT/stool antigen acceptable)
❌ Omitting PPI cessation before H. pylori testing (causes false negatives)
❌ Single endoscopic therapy for high-risk lesions (combination therapy reduces re-bleeding from 18% to 10%)
❌ Liberal transfusion strategy (Hb target > 90g/L increases mortality vs restrictive 70-90g/L)
❌ Not taking gastric ulcer biopsies (miss 2-5% that are malignant)
❌ Failing to repeat OGD for gastric ulcers (persistent ulcer at 6-8 weeks = malignancy until proven otherwise)
❌ Tranexamic acid for GI bleeding (no benefit; possible harm per HALT-IT trial)
❌ Continuing NSAIDs without PPI (40-50% ulcer recurrence)
13. Patient/Layperson Explanation
What is a Peptic Ulcer?
A peptic ulcer is an open sore or break in the lining of your stomach (called a gastric ulcer) or the first part of your small intestine (called a duodenal ulcer). The lining of these organs is normally protected from the strong acid your stomach makes to digest food. When this protective lining breaks down, the acid can damage the tissue underneath, creating an ulcer.
What Causes Peptic Ulcers?
The two most common causes are:
-
A bacterial infection called Helicobacter pylori (H. pylori): This germ lives in the stomach lining and causes inflammation. About 6-7 out of 10 people with peptic ulcers have this infection. Most people catch this infection during childhood, often from close contact with family members. Not everyone who has the germ gets an ulcer, but it significantly increases the risk.
-
Painkillers called NSAIDs (non-steroidal anti-inflammatory drugs): These include ibuprofen (Nurofen), naproxen, diclofenac, and aspirin. About 2-3 out of 10 people with peptic ulcers take these medications. NSAIDs work by blocking chemicals in your body that protect the stomach lining, making it easier for acid to cause damage.
Less common causes include smoking, severe stress or illness, and rarely, conditions that cause the stomach to make too much acid.
What Are the Symptoms?
The most common symptom is burning or gnawing pain in the upper abdomen (tummy), usually in the middle or slightly to the left. The pain often has a pattern:
-
Duodenal ulcers: Pain typically comes on 2-3 hours after eating, when your stomach is empty. Eating food or taking antacids usually relieves the pain temporarily. Many people wake up at night (1-3am) with pain.
-
Gastric ulcers: Pain is often brought on or made worse by eating. This can lead to eating less and losing weight.
Other symptoms may include:
- Nausea or feeling sick
- Bloating and feeling full quickly
- Heartburn (burning sensation in the chest)
Some people have no symptoms at all, especially if they're taking painkillers that mask the pain.
When Should I Seek Urgent Help?
Go to Accident & Emergency (A&E) or call 999 immediately if you experience:
- Vomiting blood (fresh red blood or dark brown material like coffee grounds)
- Black, tarry stools (a sign of bleeding in the stomach or intestines)
- Sudden severe abdominal pain (could indicate a burst ulcer)
- Feeling faint, dizzy, or having a rapid heartbeat (signs of significant blood loss)
See your GP urgently if you have:
- Unintended weight loss
- Difficulty swallowing
- Persistent vomiting
- You're over 55 and have new or worsening indigestion symptoms
How Is It Diagnosed?
Your doctor will ask about your symptoms and medications. Tests may include:
-
Test for H. pylori infection: This is usually a breath test or a stool (poo) sample. For the breath test, you drink a special liquid and then breathe into a bag. If you have the infection, the breath test will detect it.
-
Camera test (endoscopy or OGD): A thin, flexible tube with a camera on the end is passed down your throat into your stomach and small intestine. This allows the doctor to look directly at the lining and take small tissue samples (biopsies) if needed. This test is particularly important if you're over 55, have worrying symptoms, or if the ulcer is in the stomach (to make sure it's not cancer).
-
Blood test: To check for anaemia (low blood count from slow bleeding).
How Is It Treated?
Treatment depends on the cause:
1. If you have H. pylori infection:
- Antibiotic therapy: You'll take two antibiotics plus a tablet to reduce stomach acid (called a proton pump inhibitor or PPI) for 7-14 days. This combination kills the bacteria in about 8-9 out of 10 people.
- Follow-up test: 4 weeks after finishing the antibiotics, you'll have another breath or stool test to make sure the infection is gone. This step is very important—if the bacteria aren't eliminated, the ulcer is likely to come back.
2. If NSAIDs caused the ulcer:
- Stop the painkiller if possible, or switch to a different type of pain relief like paracetamol.
- Acid-reducing tablets (PPI): You'll take these for 4-8 weeks to allow the ulcer to heal.
- If you must continue taking NSAIDs (for example, for arthritis), you'll need to take a PPI every day to protect your stomach.
3. Acid-reducing medication:
- PPIs (e.g., omeprazole, lansoprazole) work by reducing the amount of acid your stomach produces, giving the ulcer time to heal.
- Most ulcers heal within 4-8 weeks with this treatment.
What Can I Expect?
- Most ulcers heal completely with treatment within 4-8 weeks.
- If H. pylori is successfully eliminated, the chance of the ulcer coming back is very low (less than 5 in 100 people per year).
- If the ulcer was in your stomach, you'll need a repeat camera test 6-8 weeks later to make sure it's healed (stomach ulcers can occasionally be cancerous).
- Without treatment, or if the cause isn't addressed, ulcers often come back—6-8 out of 10 people will have another ulcer within a year.
What Should I Do to Help?
- Take all your medications as prescribed: Even if you feel better, complete the full course of antibiotics.
- Avoid NSAIDs: Unless your doctor says they're essential, avoid ibuprofen and similar painkillers. Use paracetamol instead for pain relief.
- Stop smoking: Smoking slows ulcer healing and increases the risk of ulcers coming back.
- Limit alcohol: Heavy alcohol use can irritate the stomach lining.
- Eat regular meals: There's no special "ulcer diet," but eating regularly may help prevent pain. Avoid foods that seem to trigger your symptoms.
- Manage stress: While stress doesn't cause ulcers, it may worsen symptoms. Try relaxation techniques if stress is a problem for you.
Are There Complications?
Most people with peptic ulcers recover fully with treatment. However, complications can occur in about 1-2 in 10 people:
- Bleeding: The ulcer can erode into a blood vessel, causing bleeding (vomiting blood or black stools).
- Perforation: The ulcer can make a hole through the wall of the stomach or intestine, causing sudden severe pain and requiring emergency surgery.
- Blockage: Swelling or scarring from the ulcer can narrow the outlet of the stomach, causing vomiting and weight loss.
These complications are more common in older people and those who continue taking NSAIDs.
Where Can I Find More Information and Support?
- Guts UK: A charity providing information about digestive disorders (www.gutscharity.org.uk)
- NHS website: Information about peptic ulcers (www.nhs.uk)
- British Society of Gastroenterology: Professional body with patient resources (www.bsg.org.uk)
- Your GP or practice nurse: For personalised advice and support
14. References
Primary Guidelines
-
Lanas A, Chan FKL. Peptic ulcer disease. Lancet. 2017;390(10094):613-624. doi:10.1016/S0140-6736(16)32404-7 PMID: 28242110
-
Malfertheiner P, Megraud F, Rokkas T, et al. Management of Helicobacter pylori infection: the Maastricht VI/Florence consensus report. Gut. 2022;71(9):1724-1762. doi:10.1136/gutjnl-2022-327745 PMID: 35944925
Epidemiology & Complications
-
Søreide K, Thorsen K, Harrison EM, et al. Perforated peptic ulcer. Lancet. 2015;386(10000):1288-1298. doi:10.1016/S0140-6736(15)00276-7 PMID: 26460664
-
Kuipers EJ, Thijs JC, Festen HP. The prevalence of Helicobacter pylori in peptic ulcer disease. Aliment Pharmacol Ther. 1995;9 Suppl 2:59-69. PMID: 8547530
-
Wang AY, Peura DA. The prevalence and incidence of Helicobacter pylori-associated peptic ulcer disease and upper gastrointestinal bleeding throughout the world. Gastrointest Endosc Clin N Am. 2011;21(4):613-635. doi:10.1016/j.giec.2011.07.011 PMID: 21944414
H. pylori Eradication & Resistance
-
Ford AC, Yuan Y, Moayyedi P. Helicobacter pylori eradication therapy to prevent gastric cancer: systematic review and meta-analysis. Gut. 2020;69(12):2113-2121. doi:10.1136/gutjnl-2020-320839 PMID: 32404449
-
National Institute for Health and Care Excellence. Gastro-oesophageal reflux disease and dyspepsia in adults: investigation and management (CG184). 2014 (updated 2019). Available at: https://nice.org.uk/guidance/cg184
-
Hsu PI, Wu DC, Chen WC, et al. Randomized controlled trial comparing 7-day triple, 10-day sequential, and 10-day concomitant therapies for Helicobacter pylori infection. Antimicrob Agents Chemother. 2014;58(10):5936-5942. doi:10.1128/AAC.02922-14 PMID: 25070106
NSAID-Related Ulcers
-
Goldstein JL, Cryer B. Gastrointestinal injury associated with NSAID use: a case study and review of risk factors and preventative strategies. Drug Healthc Patient Saf. 2015;7:31-41. doi:10.2147/DHPS.S71976 PMID: 25693996
-
Ito T, Igarashi H, Jensen RT. Zollinger-Ellison syndrome: recent advances and controversies. Curr Opin Gastroenterol. 2013;29(6):650-661. doi:10.1097/MOG.0b013e328365efb1 PMID: 24100726
Evidence for Eradication
- Hopkins RJ, Girardi LS, Turney EA. Relationship between Helicobacter pylori eradication and reduced duodenal and gastric ulcer recurrence: a review. Gastroenterology. 1996;110(4):1244-1252. doi:10.1053/gast.1996.v110.pm8613015 PMID: 8613015
Bleeding Management
-
Gralnek IM, Stanley AJ, Morris AJ, et al. Endoscopic diagnosis and management of nonvariceal upper gastrointestinal hemorrhage (NVUGIH): European Society of Gastrointestinal Endoscopy (ESGE) guideline – Update 2021. Endoscopy. 2021;53(3):300-332. doi:10.1055/a-1369-4912 PMID: 33626647
-
Sostres C, Gargallo CJ, Arroyo MT, et al. Adverse effects of non-steroidal anti-inflammatory drugs (NSAIDs, aspirin and coxibs) on upper gastrointestinal tract. Best Pract Res Clin Gastroenterol. 2010;24(2):121-132. doi:10.1016/j.bpg.2009.11.005 PMID: 20227026
-
Lanas A, García-Rodríguez LA, Polo-Tomás M, et al. Time trends and impact of upper and lower gastrointestinal bleeding and perforation in clinical practice. Am J Gastroenterol. 2009;104(7):1633-1641. doi:10.1038/ajg.2009.164 PMID: 19574968
Pathophysiology
-
Hooi JKY, Lai WY, Ng WK, et al. Global prevalence of Helicobacter pylori infection: systematic review and meta-analysis. Gastroenterology. 2017;153(2):420-429. doi:10.1053/j.gastro.2017.04.022 PMID: 28456631
-
Suerbaum S, Michetti P. Helicobacter pylori infection. N Engl J Med. 2002;347(15):1175-1186. doi:10.1056/NEJMra020542 PMID: 12374879
-
McColl KEL. Clinical practice. Helicobacter pylori infection. N Engl J Med. 2010;362(17):1597-1604. doi:10.1056/NEJMcp1001110 PMID: 20427808
COX-2 Inhibitors
- Chan FK, Hung LC, Suen BY, et al. Celecoxib versus diclofenac and omeprazole in reducing the risk of recurrent ulcer bleeding in patients with arthritis. N Engl J Med. 2002;347(26):2104-2110. doi:10.1056/NEJMoa021907 PMID: 12501222
Gastric Cancer Risk
- Rugge M, Genta RM, Di Mario F, et al. Gastric cancer as preventable disease. Clin Gastroenterol Hepatol. 2017;15(12):1833-1843. doi:10.1016/j.cgh.2017.05.023 PMID: 28532699
Forrest Classification
- Forrest JA, Finlayson ND, Shearman DJ. Endoscopy in gastrointestinal bleeding. Lancet. 1974;2(7877):394-397. doi:10.1016/s0140-6736(74)91770-x PMID: 4136718
Endoscopic Haemostasis
- Calvet X, Vergara M, Brullet E, et al. Addition of a second endoscopic treatment following epinephrine injection improves outcome in high-risk bleeding ulcers. Gastroenterology. 2004;126(2):441-450. doi:10.1053/j.gastro.2003.11.006 PMID: 14762781
Rockall Score
- Rockall TA, Logan RF, Devlin HB, Northfield TC. Risk assessment after acute upper gastrointestinal haemorrhage. Gut. 1996;38(3):316-321. doi:10.1136/gut.38.3.316 PMID: 8675081
Glasgow-Blatchford Score
- Stanley AJ, Laine L, Dalton HR, et al. Comparison of risk scoring systems for patients presenting with upper gastrointestinal bleeding: international multicentre prospective study. BMJ. 2017;356:i6432. doi:10.1136/bmj.i6432 PMID: 28053181
Treatment Regimens
-
Chey WD, Leontiadis GI, Howden CW, Moss SF. ACG clinical guideline: treatment of Helicobacter pylori infection. Am J Gastroenterol. 2017;112(2):212-239. doi:10.1038/ajg.2016.563 PMID: 28071659
-
Graham DY, Lee YC, Wu MS. Rational Helicobacter pylori therapy: evidence-based medicine rather than medicine-based evidence. Clin Gastroenterol Hepatol. 2014;12(2):177-186. doi:10.1016/j.cgh.2013.05.028 PMID: 23751282
PPI Prophylaxis
- Rostom A, Dube C, Wells G, et al. Prevention of NSAID-induced gastroduodenal ulcers. Cochrane Database Syst Rev. 2002;(4):CD002296. doi:10.1002/14651858.CD002296 PMID: 12519573
Transfusion Strategies
- Villanueva C, Colomo A, Bosch A, et al. Transfusion strategies for acute upper gastrointestinal bleeding. N Engl J Med. 2013;368(1):11-21. doi:10.1056/NEJMoa1211801 PMID: 23281973
High-Dose PPI
- Lau JY, Sung JJ, Lee KK, et al. Effect of intravenous omeprazole on recurrent bleeding after endoscopic treatment of bleeding peptic ulcers. N Engl J Med. 2000;343(5):310-316. doi:10.1056/NEJM200008033430501 PMID: 10922420
HALT-IT Trial
- HALT-IT Trial Collaborators. Effects of a high-dose 24-h infusion of tranexamic acid on death and thromboembolic events in patients with acute gastrointestinal bleeding (HALT-IT): an international randomised, double-blind, placebo-controlled trial. Lancet. 2020;395(10241):1927-1936. doi:10.1016/S0140-6736(20)30848-5 PMID: 32563378
Re-bleeding Risk
- Lau JY, Barkun A, Fan DM, et al. Challenges in the management of acute peptic ulcer bleeding. Lancet. 2013;381(9882):2033-2043. doi:10.1016/S0140-6736(13)60596-6 PMID: 23746903
Further Resources
- British Society of Gastroenterology: bsg.org.uk
- Guts UK Charity: gutscharity.org.uk
- European Helicobacter and Microbiota Study Group: helicobacter.org
- American College of Gastroenterology: gi.org
Medical Disclaimer: MedVellum content is for educational purposes and clinical reference. Clinical decisions should account for individual patient circumstances. Always consult appropriate guidelines and specialists for patient care.
Learning map
Use these linked topics to study the concept in sequence and compare related presentations.
Prerequisites
Start here if you need the foundation before this topic.
- Gastric Acid Physiology
- Helicobacter pylori Microbiology
Differentials
Competing diagnoses and look-alikes to compare.
- Gastro-oesophageal Reflux Disease
- Functional Dyspepsia
- Gastric Cancer
- Zollinger-Ellison Syndrome
Consequences
Complications and downstream problems to keep in mind.
- Upper Gastrointestinal Bleeding
- Acute Abdomen