Psoriasis
Plaque psoriasis (psoriasis vulgaris) accounts for 80-90% of cases and follows a relapsing-remitting course with significant impact on quality of life. Management follows a stepwise approach: topical therapy (vitamin...
What matters first
Plaque psoriasis (psoriasis vulgaris) accounts for 80-90% of cases and follows a relapsing-remitting course with significant impact on quality of life. Management follows a stepwise approach: topical therapy (vitamin...
Erythrodermic psoriasis (less than 90% BSA involvement)
9 Jan 2026
Generated educational material; verify before clinical use.
Visible references section
See the concept before reading it
Study the key anatomy, imaging, and decision pathways as full teaching plates.
Clinical board
A visual summary of the highest-yield teaching signals on this page.
Urgent signals
Safety-critical features pulled from the topic metadata.
- Erythrodermic psoriasis (less than 90% BSA involvement)
- Generalised pustular psoriasis (von Zumbusch)
- Severe systemic symptoms (fever, malaise, haemodynamic instability)
- Psoriatic arthritis with joint destruction risk
Linked comparisons
Differentials and adjacent topics worth opening next.
- Seborrhoeic Dermatitis
- Eczema
Content status and exam context
This page is AI-generated educational content. It may contain errors or omissions and is not a substitute for current guidelines, local protocols, senior clinical judgement, or professional medical advice.
MedVellum does not claim an individual clinician reviewer, board certification, or professional credential for this page unless a future version names a real, verifiable reviewer.
Clinical explanation and evidence
Psoriasis
1. Topic Overview
Summary
Psoriasis is a chronic, immune-mediated inflammatory skin disease characterised by well-demarcated erythematous plaques with adherent silvery-white scale, most commonly affecting extensor surfaces (elbows, knees), scalp, and nails. [1] It affects approximately 2-3% of the global population (approximately 125 million people worldwide) and represents a substantial burden due to its chronicity, visible nature, and association with multiple comorbidities. [1,2] The pathogenesis centres on dysregulated immune responses involving the IL-23/IL-17 axis and TNF-α, with genetic susceptibility (particularly HLA-C*06:02) and environmental triggers (infection, trauma, stress, medications) initiating disease. [2,3]
Plaque psoriasis (psoriasis vulgaris) accounts for 80-90% of cases and follows a relapsing-remitting course with significant impact on quality of life. [1] Management follows a stepwise approach: topical therapy (vitamin D analogues combined with corticosteroids) for mild disease (less than 10% body surface area), phototherapy or conventional systemic agents (methotrexate, ciclosporin) for moderate disease, and biologic therapies targeting specific cytokine pathways (anti-TNF-α, anti-IL-17, anti-IL-23) for moderate-to-severe disease. [1,4]
The disease extends beyond the skin, with 20-30% of patients developing psoriatic arthritis, and increased prevalence of cardiovascular disease, metabolic syndrome, inflammatory bowel disease, and depression. [2,5] Modern biologic therapies have revolutionised outcomes, with anti-IL-23 agents achieving ≥75% reduction in disease severity (PASI 75) in 85-90% of patients and near-complete clearance (PASI 90) in 75-82% at one year. [6,7] Early recognition, comprehensive assessment of disease burden and comorbidities, and personalised treatment selection are essential to optimise patient outcomes.
Key Facts
- Global Prevalence: 2-3% (125 million people worldwide) [1,2]
- Age of Onset: Bimodal distribution — Type 1 (peak 16-22 years, HLA-C*06:02-associated, stronger family history), Type 2 (peak 50-60 years, less genetic association) [2]
- Pathophysiology: IL-23/IL-17 axis central; keratinocyte hyperproliferation (cell cycle reduced from 28 days to 3-5 days) [2,3]
- Most Common Variant: Plaque psoriasis (80-90%) [1]
- Psoriatic Arthritis: Develops in 20-30%; nail disease is strongest predictor [5,8]
- Assessment Tools: PASI (Psoriasis Area and Severity Index), BSA (Body Surface Area), DLQI (Dermatology Life Quality Index), PEST (Psoriasis Epidemiology Screening Tool for arthritis) [1]
- First-Line Topical: Fixed-combination calcipotriol/betamethasone dipropionate [9]
- First-Line Biologic: Anti-IL-23 agents (guselkumab, risankizumab) show highest efficacy [6,7]
- Comorbidities: Cardiovascular disease (increased risk of MI, stroke), metabolic syndrome, depression, inflammatory bowel disease, uveitis [2,5]
Clinical Pearls
"Auspitz Sign": Removal of psoriatic scale reveals pinpoint bleeding from dilated capillaries in elongated dermal papillae — specific but not always present. [1]
"Koebner Phenomenon": New psoriatic lesions develop at sites of skin trauma (scratching, surgery, tattoos, sunburn) within 10-20 days — present in 25-50% of patients. [1,2]
"Nail Changes Predict Arthritis": Nail psoriasis (pitting, onycholysis, oil-drop sign) is the strongest clinical predictor of psoriatic arthritis development — screen all patients with nail disease. [8]
"Treat the Whole Patient": Psoriasis is a systemic inflammatory disease — screen for psoriatic arthritis (PEST questionnaire), cardiovascular risk factors, metabolic syndrome, and mental health. [2,5]
"Streptococcal Trigger for Guttate": Acute guttate psoriasis follows streptococcal pharyngitis in 50-80% of cases — consider throat swab and anti-streptolysin O titre. [10]
"Biologics as First-Line": AAD-NPF guidelines recommend biologics as first-line option for moderate-to-severe disease due to superior efficacy and safety profiles. [4]
Why This Matters Clinically
Psoriasis significantly impairs quality of life, with DLQI scores often exceeding those of diabetes, hypertension, and heart disease. [1] The visible nature of the disease contributes to stigmatisation, social isolation, and depression. [2] Beyond the skin, psoriasis is associated with serious comorbidities: patients with severe psoriasis have a 50% increased risk of cardiovascular mortality and are at increased risk of myocardial infarction, stroke, metabolic syndrome, diabetes, and inflammatory bowel disease. [5]
Early identification and treatment of psoriatic arthritis is critical to prevent irreversible joint damage and disability. [8] Modern biologic therapies targeting the IL-23/IL-17 axis can achieve near-complete skin clearance (PASI 90-100) in the majority of patients, transforming quality of life and potentially modulating systemic inflammation and comorbidity risk. [6,7] A holistic approach addressing skin disease, joint symptoms, comorbidities, and psychological well-being is essential to comprehensive care.
2. Epidemiology
Incidence & Prevalence
Psoriasis occurs worldwide with significant geographic and ethnic variation. [2]
| Measure | Value | Notes |
|---|---|---|
| Global Prevalence | 2-3% | Approximately 125 million people [1,2] |
| Range by Region | 0.1-11.4% | Higher in northern latitudes [2] |
| Annual Incidence | 50-140 per 100,000 | Varies by geography [2] |
| Plaque Psoriasis | 80-90% of cases | Most common variant [1] |
| Guttate Psoriasis | 2% | Often acute onset, post-streptococcal [10] |
| Pustular Psoriasis | less than 5% | Rare, potentially life-threatening [1] |
| Erythrodermic Psoriasis | less than 3% | Medical emergency [1] |
Geographic Variation:
- Highest prevalence: Northern Europe, North America (3-4%) [2]
- Lowest prevalence: Asia, South America (less than 1%) [2]
- Latitude gradient: Increased prevalence in northern latitudes (possibly related to UV exposure and vitamin D) [2]
Temporal Trends:
- Prevalence appears stable or slightly increasing in developed countries [2]
- Improved recognition and diagnosis may contribute to apparent increases [2]
Demographics
| Factor | Details |
|---|---|
| Age of Onset | Bimodal: Type 1 (16-22 years), Type 2 (50-60 years) [2] |
| Sex | Equal male:female ratio [2] |
| Ethnicity | Higher in Caucasians (2.5%); lower in Asians (0.4%), Africans (1.3%) [2] |
| Family History | 30-50% have affected first-degree relative [2] |
Type 1 (Early-Onset) Psoriasis:
- Peak onset 16-22 years [2]
- Strongly associated with HLA-C*06:02 [2,3]
- More severe disease, greater body surface area involvement [2]
- Stronger family history [2]
- Better response to biologics [2]
Type 2 (Late-Onset) Psoriasis:
- Peak onset 50-60 years [2]
- Weaker genetic association [2]
- Often less extensive disease [2]
- May be associated with medication triggers [2]
Risk Factors
Genetic Factors:
- HLA-C*06:02: Strongest genetic risk allele (odds ratio 9-23); present in 60% of psoriasis patients vs 10-15% of general population [2,3]
- Other GWAS Loci: > 80 susceptibility loci identified involving immune regulation, barrier function, and IL-23/IL-17 signalling [3]
- Familial Risk:
- "One affected parent: 10% offspring risk [2]"
- "Both parents affected: 50% offspring risk [2]"
- "Monozygotic twin concordance: 65-70% [2]"
- "Dizygotic twin concordance: 15-20% [2]"
Environmental Triggers:
- Infection:
- Streptococcal pharyngitis (guttate psoriasis trigger in 50-80%) [10]
- HIV (may trigger or exacerbate) [1]
- Trauma (Koebner phenomenon): Mechanical injury, sunburn, surgery, tattoos [1,2]
- Stress: Psychological stress precipitates flares in 40-80% [2]
- Medications:
- Beta-blockers, lithium, antimalarials (chloroquine), NSAIDs, tetracyclines [1,2]
- Interferon, TNF-α inhibitor withdrawal [2]
- Corticosteroid withdrawal (risk of rebound or pustular transformation) [1]
- Lifestyle Factors:
- Smoking (odds ratio 1.8-2.0; dose-dependent) [2]
- Alcohol (associated with more severe disease) [2]
- "Obesity (BMI > 30: odds ratio 1.5-2.2) [2,5]"
Comorbidity Associations (discussed in Section 8):
- Metabolic syndrome, cardiovascular disease, psoriatic arthritis, inflammatory bowel disease, depression [2,5]
3. Aetiology & Pathophysiology
Genetic Susceptibility
Psoriasis is a complex polygenic disease with > 80 susceptibility loci identified through genome-wide association studies (GWAS). [3]
Key Genetic Associations:
- HLA-C*06:02: Located within PSORS1 locus on chromosome 6p21; strongest risk allele (OR 9-23) [2,3]
- IL-23/IL-17 Pathway Genes: IL23A, IL23R, IL12B, TRAF3IP2, IL17RA [3]
- TNF-α Pathway: TNFAIP3, TNIP1 [3]
- NF-κB Signalling: CARD14, REL, NFKBIA [3]
- Barrier Function: LCE3B/3C deletion [3]
Molecular Pathophysiology
Psoriasis results from a complex interplay between innate and adaptive immunity, with IL-23 and IL-17 as central mediators. [2,3,11]
Step 1: Initiation Phase
- Trigger: Trauma, infection, stress, or medication activates innate immune response [2,3]
- Keratinocyte Activation: Release of antimicrobial peptides (LL-37, β-defensins) and self-nucleic acids (DNA, RNA) [3,11]
- Plasmacytoid Dendritic Cell (pDC) Activation: LL-37 complexes with self-DNA activate pDCs via TLR7/9 → production of type I interferons (IFN-α, IFN-β) [3,11]
Step 2: Dendritic Cell Maturation
- Myeloid Dendritic Cells (mDCs): Activated by TNF-α, IL-1β, and type I interferons [3]
- IL-23 Production: mDCs produce IL-23 (heterodimer of p19 and p40 subunits) [3,11]
- IL-12 Production: mDCs produce IL-12 (heterodimer of p35 and p40 subunits) [3]
- Migration: Activated DCs migrate to lymph nodes [3]
Step 3: T-Cell Activation and Differentiation
- Th17 Differentiation: IL-23 drives differentiation of naïve T cells into Th17 cells [3,11]
- Th1 Differentiation: IL-12 promotes Th1 cell differentiation [3]
- Innate Lymphoid Cells (ILC3): IL-23 also activates tissue-resident ILC3 cells that produce IL-17 and IL-22 without antigen-specific activation [11]
Step 4: Cytokine Cascade (Amplification)
- Th17/ILC3 Products:
- "IL-17A, IL-17F: Activate keratinocytes, neutrophils, and fibroblasts [3,11]"
- "IL-22: Promotes keratinocyte proliferation and blocks terminal differentiation [11]"
- Th1 Products:
- "IFN-γ: Enhances inflammatory cascade [3]"
- "TNF-α: Synergises with IL-17 [3]"
Step 5: Keratinocyte Response
- Hyperproliferation: Cell cycle accelerated from 28 days to 3-5 days [1,2]
- Abnormal Differentiation: Parakeratosis (nuclei retained in stratum corneum) [1]
- Cytokine Production: Keratinocytes amplify inflammation by producing IL-1, IL-6, TNF-α, IL-23, and chemokines (CXCL1, CXCL8, CCL20) [3,11]
- Antimicrobial Peptide Release: LL-37, β-defensins perpetuate dendritic cell activation [3]
Step 6: Neutrophil Recruitment
- Chemotaxis: IL-17 and CXCL8 recruit neutrophils [3]
- Munro's Microabscesses: Neutrophil collections in stratum corneum [1]
- Spongiform Pustules of Kogoj: Neutrophils within epidermis [1]
Step 7: Vascular Changes
- Angiogenesis: VEGF production leads to increased vascularity [3]
- Dilated Capillaries: Elongated dermal papillae with dilated capillary loops (visible as pinpoint bleeding in Auspitz sign) [1]
Perpetuation and Chronicity:
- Positive feedback loop: Keratinocyte-derived cytokines sustain dendritic cell and T-cell activation [3,11]
- Tissue-resident memory T cells (TRM) in skin maintain local immune responses even after clinical resolution [11]
- IL-17-producing TRM cells rapidly reactivate upon trigger exposure [11]
Classification of Psoriasis Variants
| Type | Frequency | Clinical Features | Pathophysiology Notes |
|---|---|---|---|
| Plaque (Vulgaris) | 80-90% | Well-demarcated erythematous plaques with silver scale; elbows, knees, scalp, sacrum [1,2] | Classic IL-23/IL-17 pathway [3] |
| Guttate | 2% | Multiple small (0.5-1.5cm) "raindrop" lesions; acute onset; trunk and proximal limbs [10] | Often post-streptococcal; molecular mimicry between streptococcal M-protein and keratin [10] |
| Inverse (Flexural) | 3-7% | Smooth, shiny, erythematous patches in intertriginous areas (axillae, groin, inframammary); minimal scale [1] | Moisture inhibits scale formation [1] |
| Pustular (Localised) | less than 5% | Palmoplantar pustulosis; sterile pustules on palms/soles [1] | IL-36 pathway dysregulation [12] |
| Pustular (Generalised) | less than 1% | von Zumbusch type; acute onset; widespread sterile pustules, fever, systemic upset; medical emergency [1,12] | IL-36RN mutations (deficiency of IL-36 receptor antagonist); unchecked IL-36-mediated inflammation [12] |
| Erythrodermic | less than 3% | > 90% BSA involvement; universal erythema, scale, systemic symptoms; thermoregulation failure [1] | Massive cytokine release; may complicate existing plaque psoriasis or result from medication withdrawal [1] |
| Nail Psoriasis | 50% | Pitting, onycholysis, subungual hyperkeratosis, oil-drop sign [1,8] | Involvement of nail matrix and nail bed; strong predictor of arthritis [8] |
4. Clinical Presentation
Symptoms
Cutaneous Symptoms:
- Pruritus: Present in 60-80% of patients; may be mild to severe [1,2]
- Burning or Soreness: Particularly in inflamed or fissured plaques [1]
- Bleeding: From scale removal or fissuring [1]
- Pain: In severe cases, especially palmoplantar or erythrodermic psoriasis [1]
Systemic Symptoms (indicate severe disease):
- Fatigue: Common, may relate to systemic inflammation or depression [2]
- Fever: In generalised pustular or erythrodermic psoriasis [1,12]
- Malaise: In severe flares [1]
Joint Symptoms (20-30% develop psoriatic arthritis) [5,8]:
- Arthralgia: Pain in joints (DIPs, PIPs, large joints, axial skeleton) [8]
- Stiffness: Particularly morning stiffness > 30 minutes [8]
- Swelling: "Sausage digit" (dactylitis) in fingers or toes [8]
- Enthesitis: Pain at tendon/ligament insertions (Achilles, plantar fascia) [8]
- Spinal Symptoms: Back pain, stiffness (spondylitis) [8]
Psychological Impact:
- Depression, anxiety, social withdrawal, reduced quality of life [2]
- DLQI scores in moderate-to-severe psoriasis often > 10 (large effect on life) [1]
Signs (Physical Examination Findings)
General Inspection:
- Distribution: Extensor surfaces (elbows, knees), scalp, sacrum, natal cleft [1,2]
- Symmetry: Typically bilateral and symmetrical [1]
- Morphology: Well-demarcated erythematous plaques with adherent silvery-white scale [1,2]
Plaque Characteristics:
- Size: Variable (small guttate lesions to large confluent plaques) [1]
- Colour: Salmon-pink to deep red; scale is silvery-white [1]
- Border: Sharp, well-demarcated (unlike eczema) [1]
- Surface: Thick, adherent scale [1]
Specific Signs:
| Sign | Description | Clinical Significance |
|---|---|---|
| Auspitz Sign | Pinpoint bleeding upon scale removal | Caused by dilated capillaries in elongated dermal papillae; specific but not always present [1] |
| Grattage Test (Candle Wax Sign) | Scratching plaque produces silvery scale resembling candle wax scrapings | Demonstrates parakeratotic scale [1] |
| Koebner Phenomenon | New psoriatic lesions at sites of trauma (scratch, surgery, sunburn) within 10-20 days | Occurs in 25-50%; indicates active disease [1,2] |
| Woronoff Ring | Blanched halo around resolving plaques | Indicates treatment response [1] |
Scalp Psoriasis (50-80% of patients) [1]:
- Thick, adherent, silvery scale
- Extension beyond hairline onto forehead ("headband distribution")
- Often spares hair follicles (non-scarring)
- May be only site of involvement
Nail Psoriasis (50% of patients; 80-90% of those with psoriatic arthritis) [1,8]:
| Finding | Location | Description |
|---|---|---|
| Pitting | Nail plate | Small depressions (1-2mm); most common sign [1] |
| Onycholysis | Distal nail | Separation of nail plate from nail bed [1] |
| Oil-Drop Sign | Nail bed | Yellow-brown discolouration resembling oil drop under nail [1] |
| Subungual Hyperkeratosis | Nail bed | Thickening under nail plate [1] |
| Splinter Haemorrhages | Nail bed | Longitudinal dark streaks [1] |
| Crumbling | Nail plate | Nail destruction in severe cases [1] |
Inverse (Flexural) Psoriasis:
- Smooth, shiny, erythematous patches in axillae, groin, inframammary, intergluteal areas [1]
- Minimal scale due to moisture [1]
- May be only manifestation [1]
Guttate Psoriasis:
- Multiple small (0.5-1.5cm) "raindrop" lesions [10]
- Sudden onset over days to weeks [10]
- Trunk and proximal extremities [10]
- Often follows streptococcal pharyngitis (2-3 weeks prior) [10]
Pustular Psoriasis:
- Palmoplantar: Sterile pustules on palms/soles, often painful [1]
- Generalised (von Zumbusch): Acute onset, widespread sterile pustules on erythematous base, fever, systemic upset; medical emergency [1,12]
- Acrodermatitis Continua of Hallopeau: Pustules on distal fingers/toes [1]
Erythrodermic Psoriasis:
-
90% BSA involvement [1]
- Universal erythema, exfoliation [1]
- Systemic symptoms: fever, chills, tachycardia, oedema [1]
- Risk of hypothermia, high-output cardiac failure, sepsis [1]
Red Flags (Requiring Urgent Assessment/Admission)
[!CAUTION] Red Flags:
- Erythrodermic psoriasis (> 90% BSA): Risk of thermoregulatory failure, high-output cardiac failure, infection [1]
- Generalised pustular psoriasis: Fever, systemic upset, haemodynamic instability; requires hospitalisation [1,12]
- Signs of sepsis: In pustular variants or erythroderma [1]
- Severe systemic symptoms: High fever, malaise, dehydration [1]
- Rapidly progressive disease: Despite treatment [1]
- Severe joint symptoms: Suggesting destructive psoriatic arthritis requiring urgent rheumatology referral [8]
5. Differential Diagnosis
Psoriasis must be distinguished from other papulosquamous and inflammatory skin conditions.
Key Differentials
| Condition | Key Distinguishing Features | Investigations to Differentiate |
|---|---|---|
| Seborrhoeic Dermatitis | Greasy yellow scale (vs silvery); predilection for seborrhoeic areas (scalp, nasolabial folds, eyebrows); less well-demarcated | Clinical; skin biopsy if uncertain [1] |
| Eczema (Atopic Dermatitis) | Flexural distribution (vs extensor); poorly demarcated; lichenification; excoriations; personal/family atopy | Clinical; IgE levels, patch testing if contact dermatitis suspected [1] |
| Tinea Corporis | Annular with central clearing; active scaly edge; usually asymmetric | KOH preparation, fungal culture [1] |
| Pityriasis Rosea | Herald patch; "Christmas tree" distribution on trunk; self-limiting (6-8 weeks); usually no recurrence | Clinical; VDRL/RPR if secondary syphilis suspected [1] |
| Pityriasis Rubra Pilaris | Orange-red colour; "islands of sparing"; palmoplantar keratoderma; follicular papules | Skin biopsy [1] |
| Cutaneous T-Cell Lymphoma (Mycosis Fungoides) | Patch stage: poorly demarcated patches; plaque stage: infiltrated plaques; may have atrophy; buttock/breast involvement | Skin biopsy with immunohistochemistry; T-cell receptor gene rearrangement [1] |
| Lichen Planus | Purple, polygonal, flat-topped papules; Wickham's striae; oral involvement common; very pruritic | Skin biopsy [1] |
| Secondary Syphilis | Polymorphic rash; palmoplantar involvement; lymphadenopathy; history of chancre | VDRL, TPPA, consider HIV testing [1] |
| Drug Eruption | Recent medication introduction; rapid onset; may have systemic symptoms | Drug history; withdrawal of suspect medication [1] |
| Subacute Cutaneous Lupus | Photosensitive; annular or psoriasiform plaques; ANA positive | ANA, anti-Ro, anti-La; skin biopsy [1] |
Guttate Psoriasis Differentials:
- Pityriasis rosea (herald patch, Christmas tree pattern)
- Secondary syphilis (palms/soles, lymphadenopathy)
- Drug eruption
- Viral exanthem
Inverse Psoriasis Differentials:
- Candidiasis (satellite pustules, KOH positive)
- Erythrasma (coral-red fluorescence under Wood's lamp)
- Seborrhoeic dermatitis
- Tinea cruris (asymmetric, active border)
Nail Psoriasis Differentials:
- Onychomycosis (fungal culture positive)
- Lichen planus (nail thinning, pterygium)
- Alopecia areata (geometric pitting)
6. Investigations
Clinical Diagnosis
Psoriasis is primarily a clinical diagnosis based on characteristic morphology, distribution, and history. [1] Investigations are used to:
- Exclude differentials
- Assess disease severity and extent
- Screen for comorbidities
- Monitor treatment safety
First-Line Investigations
For Diagnosis (if clinical uncertainty):
| Investigation | Indication | Findings |
|---|---|---|
| Skin Biopsy | Atypical presentation; exclude malignancy or other conditions | See Histopathology below [1] |
| Bacterial Throat Swab | Guttate psoriasis (assess for streptococcal infection) | Group A β-haemolytic streptococcus [10] |
| Anti-Streptolysin O (ASO) Titre | Guttate psoriasis (recent streptococcal infection) | Elevated > 200 IU/mL [10] |
| Fungal Microscopy (KOH) / Culture | Exclude tinea | Negative in psoriasis [1] |
Histopathology (Skin Biopsy) [1]
Classic Features:
- Parakeratosis: Nuclei retained in stratum corneum (abnormal keratinisation) [1]
- Hypergranulosis: Thinning or absence of granular layer [1]
- Acanthosis: Thickened epidermis with regular elongation of rete ridges [1]
- Munro's Microabscesses: Neutrophil collections in stratum corneum [1]
- Spongiform Pustules of Kogoj: Neutrophils within epidermis [1]
- Dilated Tortuous Capillaries: In elongated dermal papillae [1]
- Perivascular Lymphocytic Infiltrate: Dermis [1]
Assessment of Disease Severity
Body Surface Area (BSA) [1]:
- Patient's palm (including fingers) = 1% BSA
- Mild: less than 3% BSA
- Moderate: 3-10% BSA
- Severe: > 10% BSA
Psoriasis Area and Severity Index (PASI) [1]:
- Composite score (0-72) assessing erythema, induration, desquamation, and area involvement across four body regions
- PASI 75: 75% reduction from baseline (standard treatment efficacy endpoint)
- PASI 90: 90% reduction from baseline (high efficacy)
- PASI 100: Complete clearance
Dermatology Life Quality Index (DLQI) [1]:
- 10-item questionnaire assessing impact on quality of life (0-30)
- 0-1: No impact
- 2-5: Small impact
- 6-10: Moderate impact
- 11-20: Very large impact
- 21-30: Extremely large impact
Combined Assessment (defines treatment eligibility):
- Moderate-to-Severe Psoriasis: PASI > 10 OR BSA > 10% OR DLQI > 10 [4]
Screening for Comorbidities
All patients with psoriasis should be screened for associated comorbidities. [2,5]
Psoriatic Arthritis Screening:
- PEST (Psoriasis Epidemiology Screening Tool): 5-item questionnaire; ≥3 positive responses warrants rheumatology referral [8]
- Ever had a swollen joint?
- Has a doctor ever told you that you have arthritis?
- Do your fingernails or toenails have pits/holes?
- Have you had pain in your heel?
- Have you had a finger or toe swollen like a sausage?
- Examination: DIP joints, dactylitis, enthesitis (Achilles, plantar fascia), spinal mobility [8]
Cardiovascular and Metabolic Risk Screening:
- Lipid Profile: Total cholesterol, LDL, HDL, triglycerides [5]
- Fasting Glucose / HbA1c: Screen for diabetes [5]
- Blood Pressure: Hypertension screening [5]
- BMI and Waist Circumference: Obesity and metabolic syndrome [5]
- Cardiovascular Risk Score: QRISK3 or equivalent [5]
Mental Health Screening:
- Depression and anxiety screening (e.g., PHQ-9, GAD-7) [2]
Pre-Treatment Investigations (Before Systemic Therapy)
Conventional Systemics (Methotrexate, Ciclosporin, Acitretin) [1,4]:
| Test | Purpose |
|---|---|
| FBC | Baseline; monitor for myelosuppression (methotrexate) |
| U&E, eGFR | Baseline renal function (ciclosporin nephrotoxicity risk) |
| LFTs | Baseline liver function (methotrexate hepatotoxicity risk) |
| Hepatitis B & C Serology | Screen before methotrexate |
| Pregnancy Test | Essential before methotrexate, acitretin (teratogenic) |
| Chest X-Ray | Baseline (methotrexate pneumonitis risk) |
Biologic Therapy [1,4]:
| Test | Purpose |
|---|---|
| FBC, U&E, LFTs | Baseline |
| Hepatitis B (HBsAg, anti-HBc, anti-HBs) | Reactivation risk with immunosuppression |
| Hepatitis C Serology | Assess for chronic infection |
| HIV Test | Consider in high-risk populations |
| Tuberculosis Screening | Essential before anti-TNF or IL-23 inhibitors |
| - IGRA (QuantiFERON-TB Gold or T-SPOT.TB) | Preferred over TST (not affected by BCG) [13] |
| - Tuberculin Skin Test (TST) | Alternative; ≥5mm induration positive in immunosuppressed [13] |
| - Chest X-Ray | Assess for active or latent TB [13] |
| Pregnancy Test | Before treatment in women of childbearing potential |
| Varicella Zoster Serology | Consider; vaccinate if non-immune before immunosuppression |
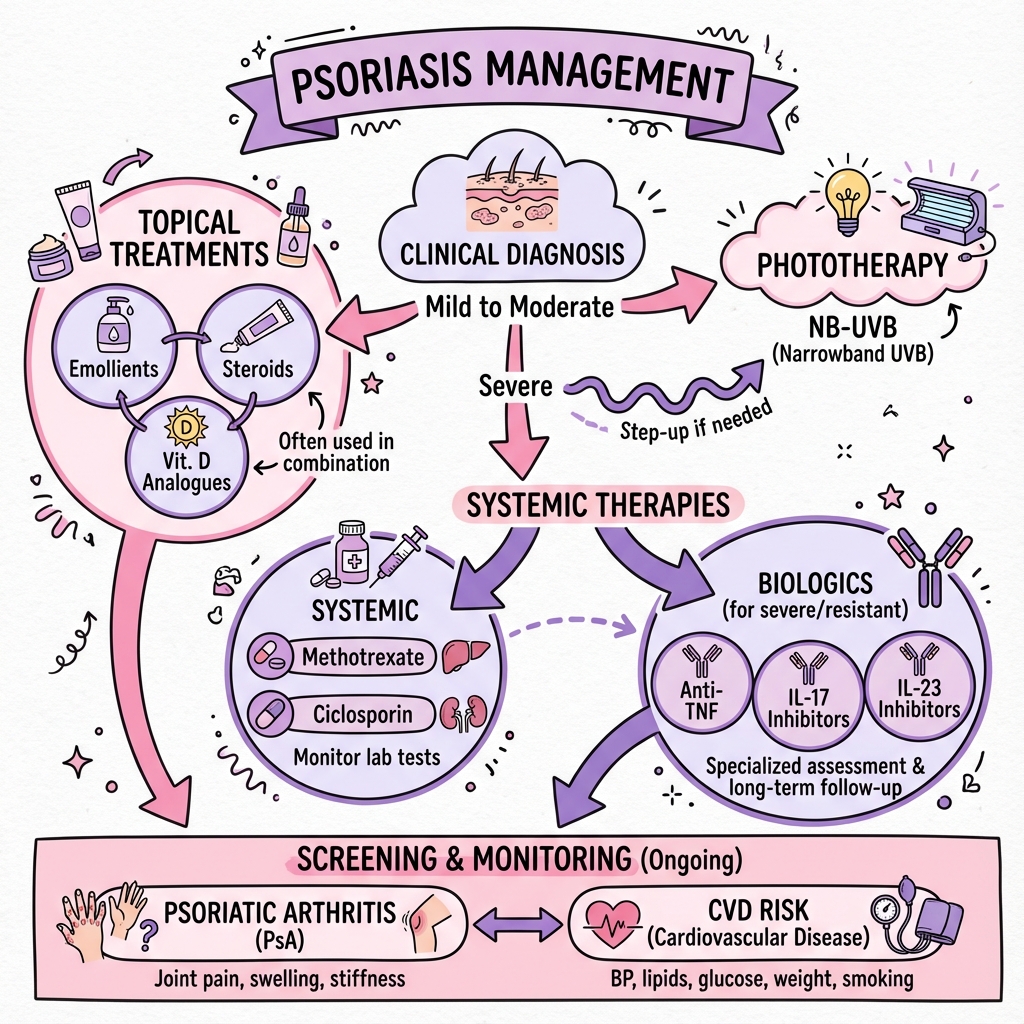
7. Management
Principles of Management
- Severity-Based Stepwise Approach: Topical → Phototherapy → Systemic → Biologics [1,4]
- Treat to Target: Aim for PASI 75-90 or DLQI ≤5 [4]
- Address Comorbidities: Screen and manage psoriatic arthritis, cardiovascular risk, mental health [2,5]
- Patient-Centred: Consider patient preferences, quality of life impact, practical considerations (e.g., injection vs oral) [4]
- Early Intervention: Particularly for joint symptoms to prevent irreversible damage [8]
- Lifestyle Modification: Weight loss, smoking cessation, alcohol reduction [2,5]
Management Algorithm
┌─────────────────────────────────────────────────┐
│ MILD PSORIASIS (less than 3-10% BSA, DLQI less than 10) │
└─────────────────────────────────────────────────┘
↓
┌─────────────────────────┐
│ TOPICAL THERAPY │
│ (First-Line) │
└─────────────────────────┘
│
│ • Vitamin D analogue + Corticosteroid
│ (calcipotriol/betamethasone)
│ • Emollients
│ • Consider rotational regimen
│
↓ (Inadequate response after 8-12 weeks)
│
┌─────────────────────────────────────────────────┐
│ MODERATE PSORIASIS (3-10% BSA, DLQI ≥10) │
└─────────────────────────────────────────────────┘
↓
┌─────────────────────────┐
│ PHOTOTHERAPY OR │
│ SYSTEMIC THERAPY │
└─────────────────────────┘
│
│ • Narrowband UVB (NB-UVB)
│ OR
│ • Methotrexate
│ • Ciclosporin
│ • Acitretin
│
↓ (Inadequate response, contraindication, or intolerance)
│
┌─────────────────────────────────────────────────┐
│ MODERATE-TO-SEVERE PSORIASIS │
│ (> 10% BSA OR PASI > 10 OR DLQI > 10) │
└─────────────────────────────────────────────────┘
↓
┌─────────────────────────┐
│ BIOLOGIC THERAPY │
│ (First-Line Option) │
└─────────────────────────┘
│
│ First-Line:
│ • Anti-IL-23 (guselkumab, risankizumab)
│ • Anti-IL-17 (secukinumab, ixekizumab)
│
│ Alternative:
│ • Anti-TNF (adalimumab, etanercept)
│ • IL-12/23 inhibitor (ustekinumab)
│
↓ (Inadequate response after 12-16 weeks)
│
└──→ Switch to alternative biologic
with different mechanism of action
Topical Therapy (Mild Psoriasis)
First-Line: Fixed-Combination Therapy [9]
| Agent | Formulation | Dosing | Efficacy | Notes |
|---|---|---|---|---|
| Calcipotriol 50µg/g + Betamethasone dipropionate 0.5mg/g | Gel, ointment, foam | Once daily; max 15g/day or 100g/week | PASI 75 at 8 weeks: 60% [9] | Synergistic; vitamin D reduces steroid side effects; avoid face/flexures with ointment [9] |
Individual Topical Agents:
| Class | Agent | Mechanism | Efficacy | Side Effects |
|---|---|---|---|---|
| Vitamin D Analogues | Calcipotriol, calcitriol, tacalcitol | Inhibit keratinocyte proliferation; promote differentiation [1] | Moderate efficacy; slower onset than steroids [1] | Irritation (10-20%); hypercalcaemia (rare if within dose limits) [1] |
| Topical Corticosteroids | Potent: betamethasone, mometasone; Very potent: clobetasol | Anti-inflammatory; suppress IL-17 and TNF-α [1] | Rapid improvement; tachyphylaxis with prolonged use [1] | Skin atrophy, striae, telangiectasia, rebound on withdrawal [1] |
| Calcineurin Inhibitors | Tacrolimus 0.1%, pimecrolimus 1% | Inhibit calcineurin → block T-cell activation [1] | Useful for face/flexures (no atrophy risk) [1] | Burning sensation (common); theoretical malignancy risk (not proven) [1] |
| Tar Preparations | Coal tar 1-5% | Anti-inflammatory; antiproliferative [1] | Modest efficacy; adjunct to phototherapy [1] | Messy, odour, staining, folliculitis [1] |
| Dithranol (Anthralin) | 0.1-2% cream | Antiproliferative; anti-inflammatory [1] | Effective but practical challenges [1] | Irritation, staining (skin, clothes, bath) [1] |
| Keratolytics | Salicylic acid 2-10% | Descaling [1] | Adjunct; improves penetration of other topicals [1] | Irritation; systemic absorption if large areas [1] |
Application Strategies:
- Rotational Therapy: Alternate agents to minimise side effects (e.g., steroid weekdays, vitamin D weekends) [1]
- Sequential Therapy: Potent steroid for rapid control → transition to vitamin D for maintenance [1]
- Emollients: Liberal use to reduce scale, pruritus, and improve penetration of active agents [1]
Specific Site Management:
- Scalp: Calcipotriol/betamethasone gel or foam; descaling with salicylic acid or coconut oil [1]
- Face/Flexures: Avoid potent steroids; use tacrolimus or mild steroid (hydrocortisone 1%) [1]
- Nails: Potent topical steroid; intralesional triamcinolone (consider if severe) [1]
Phototherapy (Moderate Psoriasis)
Narrowband UVB (NB-UVB) [1,14]
- Mechanism: Induces T-cell apoptosis; inhibits antigen presentation; promotes regulatory T cells [14]
- Indication: Moderate plaque psoriasis, guttate psoriasis [1,14]
- Protocol: 3 times/week; start at 70% of minimal erythema dose (MED); increase by 10-20% per session [14]
- Efficacy: PASI 75 in 60-70% at 12 weeks [14]
- Duration: Typically 20-30 sessions for clearance [14]
- Maintenance: Taper to 1-2 times/week or discontinue [14]
- Adverse Effects: Erythema, pruritus, xerosis; long-term: photoaging, skin cancer risk (dose-dependent) [14]
- Contraindications: Photosensitivity disorders, skin cancer history, immunosuppression [14]
PUVA (Psoralen + UVA) [1,14]
- Indication: Severe plaque psoriasis unresponsive to NB-UVB; palmoplantar psoriasis [14]
- Protocol: Oral or topical psoralen 2 hours before UVA exposure; 2-3 times/week [14]
- Efficacy: PASI 75 in 80-90% [14]
- Adverse Effects: Nausea (oral), phototoxicity, long-term skin cancer risk (higher than NB-UVB) [14]
- Monitoring: Cumulative UVA dose tracking; skin cancer surveillance [14]
Targeted Phototherapy (Excimer Laser 308nm) [1]:
- For localised plaques; reduces systemic UV exposure [1]
Conventional Systemic Therapy (Moderate-to-Severe Psoriasis)
Methotrexate [1,15]
| Parameter | Details |
|---|---|
| Mechanism | Folate antagonist; inhibits T-cell proliferation; induces adenosine (anti-inflammatory) [15] |
| Indication | Moderate-to-severe plaque psoriasis; psoriatic arthritis [1,15] |
| Dosing | Start 5-7.5mg once weekly (oral, SC, or IM); titrate by 2.5-5mg every 2-4 weeks to 15-25mg/week [15] |
| Co-Prescription | Folic acid 5mg daily (skip methotrexate day) to reduce side effects [15] |
| Efficacy | PASI 75 at 16 weeks: 40-60%; PASI 90: 20-35% [1,15] |
| Onset | 4-8 weeks [15] |
| Monitoring | FBC, LFTs, U&E: baseline, 1-2 weeks after each dose increase, then every 2-3 months [15] |
| Adverse Effects | Nausea (30%), hepatotoxicity (monitor transaminases; avoid alcohol), myelosuppression, pneumonitis (rare), teratogenicity [15] |
| Contraindications | Pregnancy, breastfeeding, significant hepatic or renal impairment, excessive alcohol, severe infection [15] |
| Drug Interactions | NSAIDs (reduce renal clearance), trimethoprim (additive folate antagonism) [15] |
Ciclosporin (Cyclosporine) [1,16]
| Parameter | Details |
|---|---|
| Mechanism | Calcineurin inhibitor; blocks T-cell activation [16] |
| Indication | Moderate-to-severe plaque psoriasis; rapid control needed; erythrodermic/pustular psoriasis [1,16] |
| Dosing | Start 2.5mg/kg/day (divided twice daily); increase by 0.5mg/kg every 2-4 weeks to max 5mg/kg/day [16] |
| Efficacy | PASI 75 at 12 weeks: 50-70% [16] |
| Onset | 2-4 weeks (rapid) [16] |
| Duration | Max continuous use: 1-2 years (nephrotoxicity risk) [16] |
| Monitoring | U&E, eGFR, BP: baseline, 2 weeks, then monthly; lipids: 3-monthly [16] |
| Adverse Effects | Nephrotoxicity (dose-dependent, often reversible), hypertension (50%), hypertrichosis, gingival hyperplasia, increased infection risk, hyperlipidaemia [16] |
| Contraindications | Uncontrolled hypertension, significant renal impairment, malignancy, serious infections [16] |
| Drug Interactions | Multiple via CYP3A4 (e.g., azole antifungals increase levels; rifampicin decreases levels) [16] |
Acitretin [1,17]
| Parameter | Details |
|---|---|
| Mechanism | Retinoid; normalises keratinocyte differentiation; anti-inflammatory [17] |
| Indication | Pustular psoriasis (first-line); erythrodermic psoriasis; adjunct to phototherapy [1,17] |
| Dosing | Start 10-25mg daily; usual maintenance 25-50mg daily [17] |
| Efficacy | PASI 75 at 12 weeks: 30-50% (less effective than methotrexate or biologics for plaque psoriasis) [17] |
| Onset | 8-12 weeks [17] |
| Monitoring | LFTs, lipids: baseline, 2-4 weeks, then 3-monthly [17] |
| Adverse Effects | Mucocutaneous (dry lips, skin, eyes — universal), hyperlipidaemia (25%), hepatotoxicity, hair loss, teratogenicity (extremely long half-life in adipose tissue) [17] |
| Contraindications | Pregnancy; women of childbearing potential must avoid pregnancy for 3 YEARS after discontinuation; hepatic/renal impairment [17] |
Apremilast [1,18]
| Parameter | Details |
|---|---|
| Mechanism | Phosphodiesterase-4 (PDE4) inhibitor; increases cAMP → reduces TNF-α, IL-17, IL-23 [18] |
| Indication | Moderate plaque psoriasis; psoriatic arthritis (useful if biologics unsuitable) [1,18] |
| Dosing | Titrate over 5 days to 30mg twice daily [18] |
| Efficacy | PASI 75 at 16 weeks: 30-40% (lower than biologics) [18] |
| Adverse Effects | Diarrhoea (15%), nausea (15%), headache, depression/suicidal ideation (monitor) [18] |
| Advantages | Oral; no monitoring required; safe in renal/hepatic impairment [18] |
Biologic Therapy (Moderate-to-Severe Psoriasis)
Biologics are recommended as first-line option for moderate-to-severe psoriasis (PASI > 10, BSA > 10%, or DLQI > 10) due to superior efficacy and acceptable safety. [4]
Anti-IL-23 Inhibitors (p19 Subunit) [6,7,19]
Mechanism: Selectively block IL-23 (p19 subunit) → inhibit Th17 differentiation and IL-17 production [6,7]
| Agent | Dosing | Efficacy (PASI 90 at Week 48-52) | Key Trials | Notes |
|---|---|---|---|---|
| Guselkumab | 100mg SC at weeks 0, 4, then every 8 weeks [6] | 70-76% [6] | VOYAGE 1 & 2 [6] | Superior to adalimumab; rapid onset; excellent long-term durability [6] |
| Risankizumab | 150mg SC at weeks 0, 4, then every 12 weeks [7] | 75-82% [7] | UltIMMa-1 & 2, IMMhance [7] | Highest PASI 90/100 rates; superior to ustekinumab and adalimumab; convenient dosing (every 12 weeks) [7] |
| Tildrakizumab | 100mg SC at weeks 0, 4, then every 12 weeks [19] | 55-62% [19] | reSURFACE 1 & 2 [19] | Less data than guselkumab/risankizumab [19] |
Efficacy Summary (Anti-IL-23): PASI 75 in 85-90%; PASI 90 in 70-82% at 1 year [6,7]
Anti-IL-17 Inhibitors [20,21]
Mechanism: Block IL-17A (± IL-17F) → inhibit keratinocyte activation and neutrophil recruitment [20,21]
| Agent | Dosing | Efficacy (PASI 90 at Week 12-16) | Key Trials | Notes |
|---|---|---|---|---|
| Secukinumab (anti-IL-17A) | 300mg SC at weeks 0, 1, 2, 3, 4, then every 4 weeks [20] | 55-65% [20] | ERASURE, FIXTURE [20] | First-in-class IL-17 inhibitor; approved for PsA, axial spondylitis [20] |
| Ixekizumab (anti-IL-17A) | 160mg SC at week 0, then 80mg at weeks 2, 4, 6, 8, 10, 12, then every 4 weeks [21] | 65-70% [21] | UNCOVER-1, 2, 3 [21] | Rapid onset (50% PASI 75 at week 2); higher PASI 90 than secukinumab [21] |
| Brodalumab (anti-IL-17RA) | 210mg SC at weeks 0, 1, 2, then every 2 weeks [1] | 60-70% [1] | AMAGINE-1, 2, 3 [1] | Blocks IL-17A, F, and E via receptor; risk of Candida, inflammatory bowel disease (caution) [1] |
Efficacy Summary (Anti-IL-17): PASI 75 in 75-85%; PASI 90 in 55-70% at 12-16 weeks [20,21]
Anti-TNF-α Inhibitors [1,22]
| Agent | Dosing | Efficacy (PASI 75 at Week 12-16) | Notes |
|---|---|---|---|
| Adalimumab | 80mg SC at week 0, then 40mg every 2 weeks from week 1 [22] | 70-80% | Fully human mAb; approved for PsA; lower PASI 90 than IL-23/IL-17 inhibitors [22] |
| Etanercept | 50mg SC twice weekly for 12 weeks, then 50mg weekly [1] | 45-55% | Fusion protein; lower efficacy than other biologics; biosimilars available [1] |
| Infliximab | 5mg/kg IV at weeks 0, 2, 6, then every 8 weeks [1] | 75-80% | Rapid onset; IV administration; immunogenicity risk [1] |
| Certolizumab Pegol | 400mg SC at weeks 0, 2, 4, then 200mg every 2 weeks [1] | 65-75% | PEGylated Fab fragment; approved for PsA; safe in pregnancy [1] |
Efficacy Summary (Anti-TNF): PASI 75 in 60-80%; PASI 90 in 25-45% [1,22]
Anti-IL-12/IL-23 Inhibitor (p40 Subunit) [1,23]
| Agent | Dosing | Efficacy | Notes |
|---|---|---|---|
| Ustekinumab | less than 100 | ||
| kg: 45mg SC at weeks 0, 4, then every 12 weeks; ≥100 | |||
| kg: 90mg [23] | PASI 75: 70-75%; PASI 90: 40-50% [23] | Blocks both IL-12 and IL-23 (p40 subunit); effective but superseded by selective anti-IL-23 agents [23] |
Biologic Selection Considerations:
| Factor | Preferred Agent(s) | Rationale |
|---|---|---|
| First-Line (No Prior Biologic) | Anti-IL-23 (risankizumab, guselkumab) [6,7] | Highest efficacy (PASI 90/100), excellent durability, convenient dosing [6,7] |
| Need for Rapid Clearance | Anti-IL-17 (ixekizumab) [21] | Fastest onset (50% achieve PASI 75 by week 2) [21] |
| Psoriatic Arthritis | Anti-IL-17 (secukinumab, ixekizumab) or anti-TNF [8,20] | Both effective for skin and joints; anti-IL-17 may be preferred for axial disease [8] |
| Inflammatory Bowel Disease | Anti-IL-23 or anti-TNF [1] | Avoid anti-IL-17 (may exacerbate IBD) [1] |
| Recurrent Candida Infections | Anti-IL-23 or anti-TNF [1] | Avoid anti-IL-17 (increases mucocutaneous candidiasis risk) [1] |
| Latent Tuberculosis | Anti-IL-17 or anti-IL-23 (lower risk) [13] | Anti-TNF has highest TB reactivation risk [13] |
| Pregnancy | Certolizumab pegol [1] | Does not cross placenta (Fab fragment) [1] |
| Cost Considerations | Biosimilar anti-TNF [1] | Lower cost but lower efficacy [1] |
Monitoring on Biologic Therapy:
- Pre-Treatment: See Section 6 (TB screening essential) [13]
- On Treatment:
- "FBC, U&E, LFTs: 3-6 monthly [1]"
- "Assess response: PASI at weeks 12-16; if inadequate response, consider switch [4]"
- Monitor for infections, injection site reactions, hypersensitivity [1]
- Annual skin cancer surveillance (especially if prior PUVA) [1]
Adverse Effects of Biologics:
- Infections: Upper respiratory tract infections (most common); opportunistic infections (TB, invasive fungal) rare [1]
- Injection Site Reactions: Common (10-20%); usually mild [1]
- Hypersensitivity: Rare; risk higher with chimeric antibodies (infliximab) [1]
- Anti-Drug Antibodies: May reduce efficacy (higher with infliximab, adalimumab) [1]
- Class-Specific:
- "Anti-IL-17: Mucocutaneous candidiasis (5-10%), potential IBD exacerbation [1]"
- "Anti-TNF: TB reactivation (highest risk), demyelination (rare), lupus-like syndrome, malignancy (controversial) [1]"
Special Scenarios
Erythrodermic Psoriasis [1]:
- Emergency: Admit for fluid resuscitation, temperature regulation, infection monitoring
- Treatment: Topical emollients, systemic ciclosporin or infliximab (rapid onset)
- Avoid: Corticosteroid withdrawal (precipitates erythroderma)
Generalised Pustular Psoriasis [1,12]:
- Emergency: Admit for supportive care, fluid resuscitation, infection screening
- Treatment: Ciclosporin, infliximab, or spesolimab (anti-IL-36 receptor; newly approved) [12]
- Note: IL36RN gene mutations in 20-30% [12]
Guttate Psoriasis [10]:
- First-Line: Topical therapy, phototherapy (NB-UVB very effective) [10]
- Streptococcal Infection: Treat with penicillin V or erythromycin if culture positive [10]
- Prognosis: 30% resolve spontaneously; 70% progress to chronic plaque psoriasis [10]
Pregnancy [1]:
- Safe: Emollients, narrowband UVB, ciclosporin (category C), certolizumab pegol [1]
- Avoid: Methotrexate (teratogen), acitretin (teratogen with 3-year washout), high-potency topical steroids (large areas) [1]
- Data Limited: Other biologics (limited human data; use if benefit outweighs risk) [1]
Children [1]:
- First-Line: Topical therapy (lower potency steroids, vitamin D analogues)
- Second-Line: Narrowband UVB
- Systemic: Methotrexate (off-label), ciclosporin, etanercept (approved ≥6 years), ustekinumab (approved ≥12 years)
Lifestyle and Patient Education
Lifestyle Modifications [2,5]:
- Weight Loss: Improves treatment response; reduces cardiovascular risk [5]
- Smoking Cessation: Reduces disease severity and improves biologic efficacy [2]
- Alcohol Reduction: Excessive alcohol associated with more severe disease and reduced treatment adherence [2]
- Stress Management: Cognitive behavioural therapy, mindfulness [2]
- Emollients: Daily use reduces scale, pruritus, and topical medication requirements [1]
Trigger Avoidance:
- Avoid skin trauma (Koebner phenomenon) [1]
- Review medications (beta-blockers, lithium, antimalarials, NSAIDs) with prescriber [1]
- Prompt treatment of streptococcal pharyngitis (especially in guttate psoriasis) [10]
Sun Exposure:
- Moderate sun exposure beneficial (vitamin D, UV effect) [1]
- Avoid sunburn (Koebner phenomenon risk) [1]
- Sunscreen important if on photosensitising medications or PUVA [1]
Patient Support:
- National Psoriasis Foundation (USA), Psoriasis Association (UK) [1]
- Psychological support for depression, anxiety, body image concerns [2]
8. Complications
Disease-Related Complications
| Complication | Prevalence | Screening/Management |
|---|---|---|
| Psoriatic Arthritis | 20-30% [5,8] | PEST questionnaire annually; rheumatology referral if positive; early DMARD therapy prevents joint destruction [8] |
| Cardiovascular Disease | 50% increased CV mortality in severe psoriasis [5] | QRISK3 assessment; manage hypertension, dyslipidaemia, diabetes; statin and aspirin if indicated [5] |
| Metabolic Syndrome | 30-50% (vs 20% general population) [5] | Screen lipids, glucose, BP, waist circumference; lifestyle modification; pharmacotherapy as needed [5] |
| Type 2 Diabetes Mellitus | Increased risk (OR 1.5-2.0) [5] | HbA1c screening; lifestyle modification; metformin if indicated [5] |
| Depression and Anxiety | 20-30% [2] | PHQ-9, GAD-7 screening; psychological support; antidepressants if indicated [2] |
| Inflammatory Bowel Disease | 2-3x increased risk [2] | Consider IBD if persistent GI symptoms; gastroenterology referral [2] |
| Uveitis | 7-20% (higher if PsA) [1] | Screen for eye pain, redness, photophobia; ophthalmology referral [1] |
| Chronic Kidney Disease | Increased risk (multifactorial: CVD, medications) [5] | Annual eGFR monitoring [5] |
| Non-Melanoma Skin Cancer | Increased with PUVA, immunosuppression [14] | Annual skin cancer surveillance [14] |
| Lymphoma | Slight increase (controversial; may relate to severity or treatment) [1] | No specific screening beyond standard care [1] |
Psoriatic Arthritis Patterns [8]:
- Oligoarticular Asymmetric (70%): less than 5 joints, asymmetric [8]
- Polyarticular Symmetric (15%): ≥5 joints, rheumatoid-like [8]
- Distal Interphalangeal (DIP) Predominant (5%): DIP joints, often with nail disease [8]
- Spondylitis (5%): Axial involvement ± peripheral arthritis [8]
- Arthritis Mutilans (less than 5%): Severe destructive arthropathy [8]
Key Features of Psoriatic Arthritis [8]:
- Dactylitis ("sausage digit") — 40% [8]
- Enthesitis (Achilles, plantar fascia) — 30-50% [8]
- Nail disease — 80-90% [8]
- Axial involvement — 20-40% [8]
Treatment-Related Complications
| Treatment | Major Complications | Monitoring | Management |
|---|---|---|---|
| Topical Corticosteroids | Skin atrophy, striae, telangiectasia, tachyphylaxis, rebound flare [1] | Clinical examination | Limit duration; use lowest effective potency; avoid abrupt discontinuation [1] |
| Methotrexate | Hepatotoxicity, myelosuppression, pneumonitis, teratogenicity [15] | FBC, LFTs every 2-3 months; CXR if respiratory symptoms [15] | Folic acid 5mg daily; reduce dose or stop if transaminases > 2x ULN; avoid alcohol [15] |
| Ciclosporin | Nephrotoxicity, hypertension, increased infection risk, malignancy (long-term) [16] | U&E, eGFR, BP monthly [16] | Reduce dose if creatinine > 30% above baseline; max 2 years continuous use [16] |
| Acitretin | Teratogenicity (3-year washout), hyperlipidaemia, hepatotoxicity [17] | LFTs, lipids every 3 months; pregnancy test [17] | Manage dyslipidaemia; avoid pregnancy for 3 years [17] |
| Biologics | Infections (TB, invasive fungal), malignancy (uncertain), hypersensitivity [1] | TB screening pre-treatment; monitor for infections [1,13] | Treat latent TB before biologic initiation; withhold biologic during serious infection [1,13] |
| PUVA | Skin cancer (dose-dependent; cumulative risk) [14] | Cumulative UVA dose tracking; annual skin examination [14] | Limit lifetime PUVA exposure; avoid if history of melanoma [14] |
9. Prognosis & Outcomes
Natural History
- Chronic Relapsing-Remitting Course: Psoriasis is lifelong with unpredictable flares and remissions [1,2]
- No Spontaneous Cure: Rare cases of prolonged remission, but disease typically persists [2]
- Guttate Variant: 30% resolve spontaneously within months; 70% evolve into chronic plaque psoriasis [10]
- Disease Progression: 20-30% develop psoriatic arthritis (median time 10 years after skin disease onset) [8]
Treatment Outcomes
Modern biologic therapies have transformed psoriasis outcomes, with majority of patients achieving near-complete clearance. [6,7]
Efficacy by Treatment Class (PASI 75 / PASI 90 at 1 Year):
| Treatment | PASI 75 | PASI 90 | PASI 100 | Notes |
|---|---|---|---|---|
| Anti-IL-23 (risankizumab, guselkumab) | 85-90% [6,7] | 75-82% [6,7] | 40-50% [6,7] | Highest efficacy; excellent durability [6,7] |
| Anti-IL-17 (secukinumab, ixekizumab) | 75-85% [20,21] | 55-70% [20,21] | 30-40% [20,21] | Rapid onset; high efficacy [20,21] |
| Anti-TNF (adalimumab) | 60-75% [22] | 35-45% [22] | 15-25% [22] | Lower efficacy than IL-23/IL-17 inhibitors [22] |
| Ustekinumab (anti-IL-12/23) | 70-75% [23] | 40-50% [23] | 20-30% [23] | Convenient dosing (every 12 weeks) [23] |
| Methotrexate | 40-60% [15] | 20-35% [15] | 10-15% [15] | Cost-effective; requires monitoring [15] |
| Ciclosporin | 50-70% [16] | 30-40% [16] | 10-20% [16] | Rapid onset; nephrotoxicity limits duration [16] |
| Apremilast | 30-40% [18] | 15-20% [18] | 5-10% [18] | Oral; no monitoring; lower efficacy [18] |
| NB-UVB | 60-70% [14] | 30-40% [14] | 10-20% [14] | Effective; requires clinic visits 3x/week [14] |
Quality of Life Improvement:
- Biologics achieving PASI 90 significantly improve DLQI (mean reduction 10-15 points) [1]
- Mental health and work productivity improve with skin clearance [2]
Long-Term Durability:
- Anti-IL-23: Excellent durability; 70-80% maintain PASI 90 at 2-3 years [6,7]
- Anti-IL-17: Good durability; 60-70% maintain PASI 90 at 2-3 years [20,21]
- Anti-TNF: Moderate durability; secondary loss of efficacy in 20-40% (anti-drug antibodies) [1]
Prognostic Factors
Good Prognosis (Higher Treatment Response):
- Early-onset (Type 1) psoriasis [2]
- HLA-C*06:02 positive [2]
- Limited body surface area involvement [1]
- No comorbidities [1]
- Good treatment adherence [1]
- Non-smoker [2]
- Normal BMI [5]
Poor Prognosis (Lower Treatment Response, Higher Comorbidity Risk):
- Severe, extensive disease (> 10% BSA) [1]
- Psoriatic arthritis [8]
- Obesity (BMI > 30) [5]
- Smoking [2]
- Excessive alcohol use [2]
- Metabolic syndrome [5]
- Poor treatment adherence [1]
- Anti-drug antibodies (biologics) [1]
Factors Predicting Psoriatic Arthritis Development [8]:
- Nail disease (strongest predictor) [8]
- Scalp involvement [8]
- Intergluteal/perianal involvement [8]
- Severe skin disease [8]
- Family history of PsA [8]
10. Prevention & Screening
Primary Prevention
No established primary prevention for psoriasis (genetic predisposition). [2]
Risk Reduction Strategies:
- Maintain healthy weight [5]
- Avoid smoking [2]
- Moderate alcohol consumption [2]
- Stress management [2]
Secondary Prevention (Flare Prevention)
Trigger Avoidance:
- Minimise skin trauma (Koebner phenomenon) [1]
- Prompt treatment of streptococcal infections (especially in children/young adults) [10]
- Review medications with potential to trigger psoriasis (beta-blockers, lithium, antimalarials) [1]
- Avoid abrupt corticosteroid withdrawal [1]
Maintenance Therapy:
- Continue topical therapy even when clear to prevent relapse [1]
- Biologics maintain remission; discontinuation often leads to relapse within 4-6 months [1]
Screening for Comorbidities
All patients with psoriasis should undergo regular screening for associated comorbidities. [2,5,8]
Annual Screening Protocol:
| Comorbidity | Screening Tool | Action if Positive |
|---|---|---|
| Psoriatic Arthritis | PEST questionnaire; examine joints, nails, entheses [8] | Rheumatology referral; early DMARD [8] |
| Cardiovascular Risk | QRISK3; BP; lipids; BMI [5] | Lifestyle modification; statin, antihypertensive if indicated [5] |
| Diabetes | HbA1c or fasting glucose [5] | Lifestyle modification; metformin if indicated [5] |
| Depression/Anxiety | PHQ-9, GAD-7 [2] | Psychological support; antidepressants if indicated [2] |
| Skin Cancer | Full skin examination (especially if prior PUVA) [14] | Dermatology review; biopsy suspicious lesions [14] |
11. Key Guidelines & Evidence
Major Clinical Guidelines
-
National Institute for Health and Care Excellence (NICE) NG153: Psoriasis: assessment and management (2012, updated 2017, 2023) [4]
- Recommends biologics as first-line option for moderate-to-severe psoriasis
- Stepwise approach: topical → phototherapy → systemic → biologics
- Available: nice.org.uk/guidance/ng153
-
American Academy of Dermatology – National Psoriasis Foundation (AAD-NPF): Guidelines of care for the management and treatment of psoriasis with biologics (2019, updated 2021) [4]
- Strongly recommends biologics for moderate-to-severe disease
- Supports biologic use in biologic-naïve patients
- Emphasises treat-to-target approach (PASI 75-90 or DLQI ≤5)
-
British Association of Dermatologists (BAD): Guidelines for the management of psoriasis (2017, updated 2020) [4]
- Comprehensive guidance across all severities
- Emphasises holistic approach including comorbidity screening
-
European S3-Guidelines on the systemic treatment of psoriasis vulgaris (2015, updated 2020) [4]
- European consensus on systemic and biologic therapies
- Detailed drug-specific recommendations
Landmark Trials
IL-23 Inhibitors:
-
VOYAGE 1 & 2 (Guselkumab) [6]
- Gordon KB, et al. Lancet. 2016;388(10042):402-411. PMID: 27343865
- Phase 3 RCTs; guselkumab vs adalimumab vs placebo
- Results: PASI 90 at week 16: guselkumab 73% vs adalimumab 50% (pless than 0.001)
-
UltIMMa-1 & UltIMMa-2 (Risankizumab) [7]
- Gordon KB, et al. Lancet. 2018;392(10148):650-661. PMID: 30097359
- Phase 3 RCTs; risankizumab vs ustekinumab vs placebo
- Results: PASI 90 at week 16: risankizumab 75% vs ustekinumab 42% (pless than 0.001)
-
IMMhance (Risankizumab) [7]
- Reich K, et al. Lancet. 2019;394(10213):576-586. PMID: 31427113
- Risankizumab vs adalimumab
- Results: PASI 90 at week 16: risankizumab 72% vs adalimumab 47% (pless than 0.001); sustained to week 44
IL-17 Inhibitors:
-
ERASURE & FIXTURE (Secukinumab) [20]
- Langley RG, et al. N Engl J Med. 2014;371(4):326-338. PMID: 25007392
- Phase 3 RCTs; secukinumab vs etanercept vs placebo
- Results: PASI 75 at week 12: secukinumab 300mg 82% vs etanercept 44% (pless than 0.001)
- First trial demonstrating superiority of IL-17 inhibition
-
UNCOVER-1, 2, 3 (Ixekizumab) [21]
- Gordon KB, et al. N Engl J Med. 2016;375(4):345-356. PMID: 27299809
- Phase 3 RCTs; ixekizumab vs etanercept vs placebo
- Results: PASI 75 at week 12: ixekizumab 89% vs etanercept 42%; rapid onset (50% achieved PASI 75 by week 2)
Conventional Systemics:
-
Methotrexate vs Placebo (METOP) [15]
- Heydendael VM, et al. Br J Dermatol. 2003;148(5):1049-1056. PMID: 12786837
- RCT demonstrating efficacy of methotrexate in moderate-to-severe psoriasis
-
Calcipotriol/Betamethasone Fixed Combination [9]
- Papp K, et al. J Eur Acad Dermatol Venereol. 2017;31(1):140-147. PMID: 27566848
- Superior efficacy to calcipotriol or betamethasone alone
Evidence Strength Summary
| Intervention | Level of Evidence | Key Supporting Evidence |
|---|---|---|
| Anti-IL-23 biologics | 1a (systematic reviews, meta-analyses, multiple high-quality RCTs) | VOYAGE, UltIMMa, IMMhance trials [6,7] |
| Anti-IL-17 biologics | 1a | ERASURE, FIXTURE, UNCOVER trials [20,21] |
| Anti-TNF biologics | 1a | Multiple phase 3 RCTs [22] |
| Methotrexate | 1b (high-quality RCTs) | METOP, observational studies [15] |
| Calcipotriol/betamethasone | 1a | Cochrane review, multiple RCTs [9] |
| Narrowband UVB | 1b | Multiple RCTs, systematic reviews [14] |
| Ciclosporin | 1b | Multiple RCTs [16] |
| Apremilast | 1a | ESTEEM trials [18] |
12. Common Exam Questions & Viva Points
Commonly Asked Questions
-
"What is psoriasis and what causes it?"
- Chronic immune-mediated inflammatory skin disease; IL-23/IL-17 axis central; genetic (HLA-C*06:02) and environmental triggers (infection, trauma, stress) [2,3]
-
"How do you assess the severity of psoriasis?"
- BSA (less than 3% mild, 3-10% moderate, > 10% severe), PASI score (0-72), DLQI (quality of life impact); moderate-to-severe defined as PASI > 10 OR BSA > 10% OR DLQI > 10 [1,4]
-
"What are the first-line treatments for mild and severe psoriasis?"
- Mild: Topical calcipotriol/betamethasone fixed combination [9]
- Severe: Biologics (anti-IL-23 or anti-IL-17) recommended as first-line option [4,6,7]
-
"What comorbidities are associated with psoriasis?"
- Psoriatic arthritis (20-30%), cardiovascular disease, metabolic syndrome, diabetes, depression, IBD, uveitis [2,5,8]
-
"How do you screen for psoriatic arthritis?"
- PEST questionnaire (≥3 positive responses warrants rheumatology referral); examine for dactylitis, enthesitis, nail disease [8]
-
"What is the mechanism of action of biologics used in psoriasis?"
- Anti-IL-23: Block IL-23 (p19 subunit) → inhibit Th17 differentiation and IL-17 production [6,7]
- Anti-IL-17: Block IL-17A → inhibit keratinocyte activation and neutrophil recruitment [20,21]
- Anti-TNF: Block TNF-α → reduce inflammation [22]
-
"What is the Koebner phenomenon?"
- Development of new psoriatic lesions at sites of skin trauma (scratching, surgery, tattoos, sunburn) within 10-20 days; occurs in 25-50% of patients; indicates active disease [1,2]
-
"What investigations are required before starting a patient on methotrexate?"
- FBC, U&E, LFTs, chest X-ray, hepatitis B/C serology, pregnancy test (women of childbearing potential); co-prescribe folic acid 5mg daily [15]
-
"What is the Auspitz sign?"
- Pinpoint bleeding upon removal of psoriatic scale, caused by dilated capillaries in elongated dermal papillae; specific but not always present [1]
-
"What triggers guttate psoriasis?"
- Streptococcal pharyngitis in 50-80% of cases (molecular mimicry between streptococcal M-protein and keratin); consider throat swab and ASO titre [10]
Viva Opening Statement
"Psoriasis is a chronic, immune-mediated inflammatory skin disease affecting 2-3% of the global population, characterised by well-demarcated erythematous plaques with silvery scale, most commonly on extensor surfaces. The pathogenesis centres on the IL-23/IL-17 axis, with genetic susceptibility (HLA-C*06:02) and environmental triggers. Plaque psoriasis accounts for 80-90% of cases. It is associated with significant comorbidities including psoriatic arthritis in 20-30%, cardiovascular disease, and metabolic syndrome. Management follows a stepwise approach from topical therapy for mild disease to biologic agents targeting IL-23 or IL-17 for moderate-to-severe disease, with anti-IL-23 inhibitors achieving PASI 90 in 75-82% of patients at one year." [1,2,3,6,7]
Common Mistakes (That Fail Candidates)
❌ Failing to screen for psoriatic arthritis: All patients with psoriasis require regular joint assessment; nail disease is strongest predictor [8]
❌ Not assessing cardiovascular risk: Psoriasis is a systemic inflammatory disease with 50% increased CV mortality in severe cases [5]
❌ Prescribing methotrexate without folic acid: Folic acid 5mg daily (skip methotrexate day) reduces side effects [15]
❌ Missing tuberculosis screening before biologics: IGRA and chest X-ray essential before anti-TNF or anti-IL-23 therapy [13]
❌ Abrupt corticosteroid withdrawal: Risk of rebound flare or transformation to pustular/erythrodermic psoriasis [1]
❌ Confusing psoriasis with eczema: Psoriasis is well-demarcated, extensor distribution, silvery scale; eczema is poorly demarcated, flexural, excoriated [1]
❌ Not recognising erythrodermic psoriasis as an emergency: > 90% BSA involvement requires admission for thermoregulation, fluid resuscitation, infection monitoring [1]
13. Patient/Layperson Explanation
What is Psoriasis?
Psoriasis is a long-term (chronic) skin condition where your immune system causes skin cells to grow too quickly. Normally, skin cells take about a month to replace themselves, but in psoriasis this happens in just 3-5 days. The extra skin cells build up as thick, scaly patches that can be itchy or sore. Psoriasis is not contagious — you cannot catch it from someone else or pass it to others.
Why Does It Matter?
- It's common: About 1 in 40 people have psoriasis
- It can be uncomfortable: The patches may itch, crack, or bleed
- It affects confidence: The visible patches can make people self-conscious
- It's more than skin deep: About 1 in 4 people with psoriasis also develop joint problems (psoriatic arthritis)
- It's linked to other health problems: People with psoriasis have a slightly higher risk of heart disease and diabetes
What Causes It?
Psoriasis is caused by a combination of:
- Your genes: It runs in families (if one parent has it, there's a 10% chance their child will too)
- Your immune system: Your body's defence system becomes overactive
- Triggers that can start or worsen flares:
- Stress
- Throat infections (especially in children)
- Skin injuries (cuts, scratches, sunburn)
- Some medications
- Smoking and heavy alcohol use
How is it Treated?
Treatment depends on how much of your body is affected:
1. Creams and Ointments (for mild psoriasis affecting less than 10% of your body)
- Vitamin D cream combined with a steroid cream (most effective)
- Moisturisers (use lots — they really help!)
- Usually applied once or twice daily
2. Light Therapy (for moderate psoriasis)
- Special ultraviolet (UV) light treatment at a hospital or clinic
- Usually 3 times per week for 8-12 weeks
- Can clear the skin in many people
3. Tablets or Oral Medicines (for moderate-to-severe psoriasis)
- Methotrexate: A tablet taken once a week (requires regular blood tests)
- Apremilast: A tablet taken twice daily (no blood tests needed)
4. Injection or Infusion Medicines (for moderate-to-severe psoriasis)
- Biologics: Very effective modern treatments given by injection every 4-12 weeks
- Target specific parts of the immune system causing psoriasis
- Examples: risankizumab, guselkumab, secukinumab
- Can clear the skin almost completely in most people (75-82% achieve 90% improvement)
What to Expect
- There's no cure, but treatments can control psoriasis very well
- It tends to come and go (flares and remissions)
- Modern treatments are very effective: Many people can achieve nearly clear skin with biologic injections
- Most people need long-term treatment to keep the skin clear
- Your doctor will monitor for other health problems like joint pain, high blood pressure, or diabetes
When to Seek Urgent Help
Contact your doctor urgently if:
- Psoriasis suddenly spreads to cover most of your body
- You develop a fever or feel generally unwell with your psoriasis
- You develop painful, swollen joints
- You have small pus-filled bumps (pustules) spreading across your skin
Lifestyle Tips
- Moisturise daily — this really helps reduce scaling and itching
- Avoid scratching or picking — this can trigger new patches
- Don't smoke — smoking makes psoriasis worse
- Reduce alcohol — heavy drinking is linked to more severe psoriasis
- Manage stress — stress can trigger flares
- Maintain a healthy weight — being overweight makes psoriasis harder to treat
- Get some sun (but don't burn!) — moderate sun exposure can help
Support and Information
- National Psoriasis Foundation (USA): psoriasis.org
- Psoriasis Association (UK): psoriasis-association.org.uk
- Support groups can help you connect with others living with psoriasis
14. References
Primary Guidelines
-
Armstrong AW, Read C. Pathophysiology, Clinical Presentation, and Treatment of Psoriasis: A Review. JAMA. 2020;323(19):1945-1960. doi:10.1001/jama.2020.4006. PMID: 32427307
-
Griffiths CEM, Armstrong AW, Gudjonsson JE, Barker JNWN. Psoriasis. Lancet. 2021;397(10281):1301-1315. doi:10.1016/S0140-6736(20)32549-6. PMID: 33812489
-
Yamanaka K, Yamamoto O, Honda T. Pathophysiology of psoriasis: A review. J Dermatol. 2021;48(6):722-731. doi:10.1111/1346-8138.15913. PMID: 33886133
-
National Institute for Health and Care Excellence. Psoriasis: assessment and management (NG153). 2012 (updated 2017, 2023). Available: https://www.nice.org.uk/guidance/ng153
Pathophysiology and Immunology
-
Boehncke WH, Schön MP. Psoriasis. Lancet. 2015;386(9997):983-994. doi:10.1016/S0140-6736(14)61909-7. PMID: 26025581
-
Singh R, Koppu S, Perche PO, Feldman SR. The Cytokine Mediated Molecular Pathophysiology of Psoriasis and Its Clinical Implications. Int J Mol Sci. 2021;22(23):12793. doi:10.3390/ijms222312793. PMID: 34884596
Comorbidities
-
Takeshita J, Grewal S, Langan SM, et al. Psoriasis and comorbid diseases: Epidemiology. J Am Acad Dermatol. 2017;76(3):377-390. doi:10.1016/j.jaad.2016.07.064. PMID: 28212759
-
Azuaga AB, Ramirez J, Cañete JD. Psoriatic Arthritis: Pathogenesis and Targeted Therapies. Int J Mol Sci. 2023;24(5):4901. doi:10.3390/ijms24054901. PMID: 36902329
Topical Therapy
- Papp K, Berth-Jones J, Kragballe K, Wozel G, de la Brassinne M. Scalp psoriasis: a review of current topical treatment options. J Eur Acad Dermatol Venereol. 2017;31(1):140-147. doi:10.1111/jdv.13859. PMID: 27566848
Guttate Psoriasis
- Weigle N, McBane S. Psoriasis. Am Fam Physician. 2013;87(9):626-633. PMID: 23668525
Pustular Psoriasis
- Bachelez H, Choon SE, Marrakchi S, et al. Inhibition of the Interleukin-36 Pathway for the Treatment of Generalized Pustular Psoriasis. N Engl J Med. 2019;380(10):981-983. doi:10.1056/NEJMc1811317. PMID: 30865801
Tuberculosis Screening
- Cantini F, Nannini C, Niccoli L, et al. Guidance for the management of patients with latent tuberculosis infection requiring biologic therapy in rheumatology and dermatology clinical practice. Autoimmun Rev. 2015;14(6):503-509. doi:10.1016/j.autrev.2015.01.011. PMID: 25633324
Phototherapy
- Almutawa F, Alnomair N, Wang Y, Hamzavi I, Lim HW. Systematic review of UV-based therapy for psoriasis. Am J Clin Dermatol. 2013;14(2):87-109. doi:10.1007/s40257-013-0015-y. PMID: 23456596
Systemic Therapy
-
Heydendael VM, Spuls PI, Opmeer BC, et al. Methotrexate versus cyclosporine in moderate-to-severe chronic plaque psoriasis. N Engl J Med. 2003;349(7):658-665. doi:10.1056/NEJMoa021359. PMID: 12917302
-
Mrowietz U, Kragballe K, Reich K, et al. Definition of treatment goals for moderate to severe psoriasis: a European consensus. Arch Dermatol Res. 2011;303(1):1-10. doi:10.1007/s00403-010-1080-1. PMID: 20857129
-
Menter A, Korman NJ, Elmets CA, et al. Guidelines of care for the management of psoriasis and psoriatic arthritis: Section 3. Guidelines of care for the management and treatment of psoriasis with topical therapies. J Am Acad Dermatol. 2009;60(4):643-659. doi:10.1016/j.jaad.2008.12.032. PMID: 19217694
-
Papp K, Reich K, Leonardi CL, et al. Apremilast, an oral phosphodiesterase 4 (PDE4) inhibitor, in patients with moderate to severe plaque psoriasis: Results of a phase III, randomized, controlled trial (Efficacy and Safety Trial Evaluating the Effects of Apremilast in Psoriasis [ESTEEM] 1). J Am Acad Dermatol. 2015;73(1):37-49. doi:10.1016/j.jaad.2015.03.049. PMID: 26089047
Biologic Therapy
-
Gordon KB, Blauvelt A, Papp KA, et al. Phase 3 Trials of Ixekizumab in Moderate-to-Severe Plaque Psoriasis. N Engl J Med. 2016;375(4):345-356. doi:10.1056/NEJMoa1512711. PMID: 27299809
-
Gordon KB, Strober B, Lebwohl M, et al. Efficacy and safety of risankizumab in moderate-to-severe plaque psoriasis (UltIMMa-1 and UltIMMa-2): results from two double-blind, randomised, placebo-controlled and ustekinumab-controlled phase 3 trials. Lancet. 2018;392(10148):650-661. doi:10.1016/S0140-6736(18)31713-6. PMID: 30097359
-
Reich K, Papp KA, Blauvelt A, et al. Tildrakizumab versus placebo or etanercept for chronic plaque psoriasis (reSURFACE 1 and reSURFACE 2): results from two randomised controlled, phase 3 trials. Lancet. 2017;390(10091):276-288. doi:10.1016/S0140-6736(17)31279-5. PMID: 28629948
-
Langley RG, Elewski BE, Lebwohl M, et al. Secukinumab in plaque psoriasis—results of two phase 3 trials. N Engl J Med. 2014;371(4):326-338. doi:10.1056/NEJMoa1314258. PMID: 25007392
-
Gordon KB, Blauvelt A, Papp KA, et al. Phase 3 Trials of Ixekizumab in Moderate-to-Severe Plaque Psoriasis. N Engl J Med. 2016;375(4):345-356. doi:10.1056/NEJMoa1512711. PMID: 27299809
-
Menter A, Tyring SK, Gordon K, et al. Adalimumab therapy for moderate to severe psoriasis: A randomized, controlled phase III trial. J Am Acad Dermatol. 2008;58(1):106-115. doi:10.1016/j.jaad.2007.09.010. PMID: 17936411
-
Leonardi CL, Kimball AB, Papp KA, et al. Efficacy and safety of ustekinumab, a human interleukin-12/23 monoclonal antibody, in patients with psoriasis: 76-week results from a randomised, double-blind, placebo-controlled trial (PHOENIX 1). Lancet. 2008;371(9625):1665-1674. doi:10.1016/S0140-6736(08)60725-4. PMID: 18486739
Medical Disclaimer: MedVellum content is for educational purposes and clinical reference. Clinical decisions should account for individual patient circumstances. Always consult current guidelines and specialist advice for complex cases.
Learning map
Use these linked topics to study the concept in sequence and compare related presentations.
Differentials
Competing diagnoses and look-alikes to compare.
- Seborrhoeic Dermatitis
- Eczema
- Tinea Corporis
Consequences
Complications and downstream problems to keep in mind.