Sarcoidosis
Diagnostic Pearl : Löfgren's Syndrome (erythema nodosum, bilateral hilar lymphadenopathy, polyarthritis/arthralgia) is highly specific (95%) for sarcoidosis. In this specific setting, biopsy is generally not required...
What matters first
Diagnostic Pearl : Löfgren's Syndrome (erythema nodosum, bilateral hilar lymphadenopathy, polyarthritis/arthralgia) is highly specific (95%) for sarcoidosis. In this specific setting, biopsy is generally not required...
Cardiac sarcoidosis (arrhythmias, heart block, sudden death risk)
6 Jan 2026
Generated educational material; verify before clinical use.
Visible references section
See the concept before reading it
Study the key anatomy, imaging, and decision pathways as full teaching plates.
Clinical board
A visual summary of the highest-yield teaching signals on this page.
Urgent signals
Safety-critical features pulled from the topic metadata.
- Cardiac sarcoidosis (arrhythmias, heart block, sudden death risk)
- Neurosarcoidosis (cranial neuropathy, seizures, hypothalamic dysfunction)
- Severe hypercalcaemia (renal failure, confusion)
- Progressive pulmonary fibrosis (Stage IV disease)
Content status and exam context
This page is AI-generated educational content. It may contain errors or omissions and is not a substitute for current guidelines, local protocols, senior clinical judgement, or professional medical advice.
MedVellum does not claim an individual clinician reviewer, board certification, or professional credential for this page unless a future version names a real, verifiable reviewer.
Clinical explanation and evidence
Sarcoidosis
1. Clinical Overview
Summary
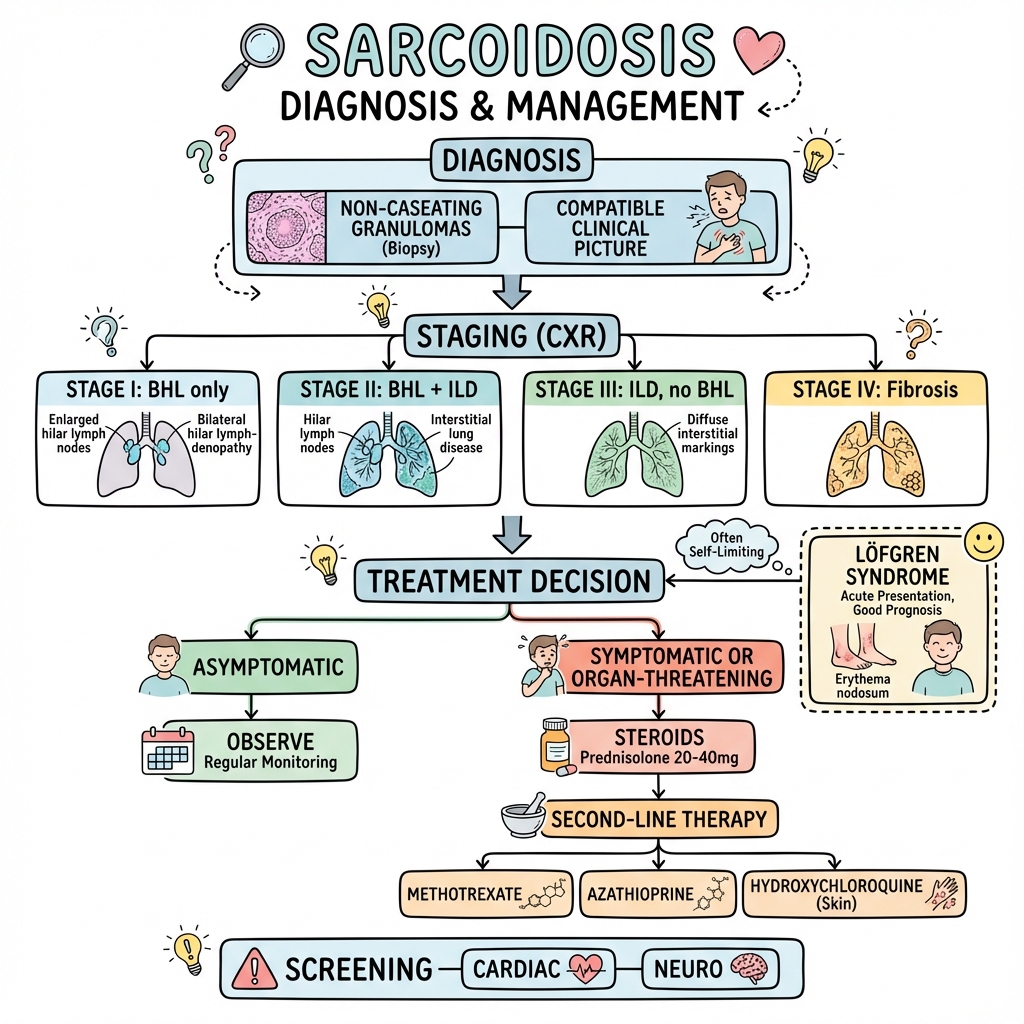
Sarcoidosis is a multisystem granulomatous disorder of unknown aetiology, characterised histologically by the presence of non-caseating epithelioid cell granulomas in affected organs. While it predominantly affects the lungs (90% of cases) and intrathoracic lymph nodes, it is a systemic disease that can involve the eyes, skin, liver, spleen, heart, and nervous system. The disease follows a variable course, ranging from asymptomatic incidental findings (often bilateral hilar lymphadenopathy) to progressive fibrosis and organ failure. It most commonly presents in young adults (20–40 years), with a second peak in women over 50. Diagnosis requires a compatible clinical and radiological presentation, histological evidence of non-caseating granulomas, and the rigorous exclusion of other granulomatous diseases, particularly tuberculosis and fungal infections. [1,2]
Key Facts
- Definition: Systemic inflammatory disease characterised by non-caseating granulomas.
- Hallmark: Accumulation of T lymphocytes (CD4+ Th1) and mononuclear phagocytes in affected organs.
- Most Common Organ: Lungs (> 90%), followed by skin (25%), eyes (25%), and liver (20%). [2]
- Diagnostic Gold Standard: Histopathological demonstration of non-caseating granulomas in a compatible clinical context, with exclusion of other causes. [1]
- Treatment Threshold: Asymptomatic disease often requires no treatment. Corticosteroids are the first-line therapy for organ-threatening or symptomatic disease. [3]
- Prognosis: Approximately two-thirds of patients achieve spontaneous remission. 10–30% develop chronic or progressive disease. [4]
Clinical Pearls
Diagnostic Pearl: Löfgren's Syndrome (erythema nodosum, bilateral hilar lymphadenopathy, polyarthritis/arthralgia) is highly specific (95%) for sarcoidosis. In this specific setting, biopsy is generally not required for diagnosis (ATS 2020 Guideline). [1]
Management Pearl: Do not treat the X-ray. Asymptomatic Stage I (BHL) and Stage II (BHL + infiltrates) sarcoidosis have high rates of spontaneous remission. Treatment is reserved for symptoms or organ threat. [3]
Safety Pearl: All patients with a new diagnosis of sarcoidosis must have an ECG and ophthalmology assessment to screen for silent cardiac and ocular involvement. [2]
Pitfall Warning: Always exclude tuberculosis (TB) and lymphoma. TB granulomas are typically caseating (necrotising), but non-caseating granulomas can also be seen in TB. Culture and molecular testing (PCR) are mandatory in endemic areas or high-risk populations.
Why This Matters Clinically
Sarcoidosis mimics many common conditions (TB, lymphoma, lung cancer). Misdiagnosis leads to inappropriate treatment (e.g., steroids for TB can be fatal). Conversely, recognising sarcoidosis allows for reassuring observation in many cases, sparing patients the toxicity of long-term immunosuppression. However, missing cardiac or neurological involvement can lead to sudden death or permanent disability.
2. Epidemiology
Incidence and Prevalence
The epidemiology of sarcoidosis varies significantly by geography, race, and sex.
- Global Incidence: 2–10 cases per 100,000 per year.
- Highest Prevalence:
- "Northern Europe: 10–40 per 100,000 (especially Scandinavia)."
- "African Americans: 35–80 per 100,000 (3-4x higher than Caucasian Americans). [5]"
- Lowest Prevalence: East Asia (Japan, Korea).
Demographics
- Age: Classic peak at 20–39 years. A second peak is observed in women aged 50–65 years (post-menopausal).
- Sex: Slight female predominance overall.
- Ethnicity Impact:
- African Americans tend to have more severe, chronic, and extrapulmonary disease (skin, bone, eye).
- White Europeans/Scandinavians are more likely to present with acute, resolving disease (Löfgren's syndrome). [6]
Genetic Factors
- Familial Clustering: Increased risk in first-degree relatives (odds ratio ~5).
- HLA Associations:
- "HLA-DRB1*03: Strongly associated with Löfgren's syndrome and good prognosis (spontaneous resolution)."
- "HLA-DRB1*15 and HLA-DRB1*14: Associated with chronic course and extra-thoracic involvement. [7]"
3. Aetiology and Pathophysiology
Aetiology: The "Unknown Antigen" Hypothesis
Sarcoidosis is believed to result from an exaggerated immune response to an unknown antigen in genetically susceptible individuals.
- Infectious Agents: Mycobacterium tuberculosis (mCAT catalase-peroxidase protein), Propionibacterium acnes, viruses. No single agent has been definitively proven.
- Environmental Triggers: Insecticides, mould, agricultural dust, silica, fire service employment (e.g., World Trade Center responders). [2]
- Autoimmunity: Vimentin has been identified as a potential autoantigen in some cases.
Molecular Pathophysiology
The core pathological process is a distinct T-helper 1 (Th1) immune response.
Exam Detail: #### Immunopathogenesis Steps
-
Antigen Presentation:
- Antigen Presenting Cells (APCs), specifically alveolar macrophages and dendritic cells, phagocytose the unknown antigen.
- Antigen is presented to CD4+ T cells via HLA class II molecules.
-
T-Cell Activation (The Sarcoid Alveolitis):
- CD4+ T cells proliferate and differentiate into Th1 and Th17 effector cells.
- This leads to a high CD4/CD8 ratio in the bronchoalveolar lavage (BAL) fluid (typically > 3.5).
-
Cytokine Storm:
- IL-2: Drives T-cell proliferation (lymphocytosis).
- IFN-γ (Interferon-gamma): Crucial for macrophage activation and granuloma formation.
- TNF-α (Tumour Necrosis Factor-alpha): Key driver of granuloma maintenance and progression. (Target for infliximab).
- IL-12 and IL-18: Promote Th1 differentiation.
-
Granuloma Formation:
- Activated macrophages transform into epithelioid cells.
- Epithelioid cells fuse to form multinucleated giant cells (Langhans type or Foreign Body type).
- The central core of macrophages/giant cells is surrounded by a rim of lymphocytes (CD4+ and CD8+) and fibroblasts.
- Non-caseating: Unlike TB, the centre does not undergo necrosis (caseation), although small areas of fibrinoid necrosis can occur (necrotising sarcoid granulomatosis).
-
Resolution vs. Fibrosis:
- In many cases, regulatory T cells (Tregs) suppress the response, and granulomas resolve.
- In chronic disease, a shift towards Th2 cytokines (IL-4, IL-13) and TGF-β promotes fibroblast recruitment, collagen deposition, and irreversible fibrosis. [8]
The Granuloma
The sarcoid granuloma is a metabolically active structure.
- 1,25-dihydroxyvitamin D production: Macrophages within the granuloma express 1-alpha-hydroxylase, converting 25(OH)D to active 1,25(OH)2D, leading to hypercalcaemia and hypercalciuria. [9]
- ACE Production: Epithelioid cells produce Angiotensin Converting Enzyme (ACE), reflecting the total granuloma burden (granuloma load).
4. Clinical Features
Sarcoidosis is the "great mimicker". Presentation can be systemic (fever, weight loss, fatigue) or organ-specific.
Pulmonary Involvement (90%)
- Symptoms: Dry cough (often persistent), dyspnoea (exertional), chest pain (vague, retrosternal).
- Signs: Lung examination is often normal. Crackles are rare unless significant fibrosis is present. Wheeze may indicate endobronchial involvement.
- Functional: Restrictive defect with reduced gas transfer (DLCO) is classic. Airway obstruction can occur.
Cutaneous Involvement (25%)
- Erythema Nodosum (EN): Tender, red nodules on anterior shins. Associated with acute sarcoidosis (Löfgren's). Biopsy of EN shows septal panniculitis, not granulomas.
- Lupus Pernio: Violaceous, indurated plaques on nose, cheeks, ears. Pathognomonic for chronic, fibrotic sarcoidosis. Associated with upper respiratory tract involvement and bone cysts. Poor prognosis. [10]
- Maculopapular Eruptions: Common on face, neck, trunk.
- Scar Sarcoidosis: Infiltration of old scars/tattoos (become raised, red, and tender). A highly specific sign.
Ocular Involvement (25%)
- Can cause blindness if untreated.
- Uveitis: Anterior (pain, red eye, photophobia) or Posterior (painless blurring, floaters).
- Keratoconjunctivitis sicca: Dry eyes (lacrimal gland infiltration).
- Heerfordt's Syndrome (Uveoparotid Fever): Uveitis + Parotitis + Facial Nerve Palsy + Fever. [2]
Cardiac Involvement (5-25%)
- Typically clinically silent (only 5% symptomatic), but autopsy studies show 25% involvement.
- Manifestations:
- "Conduction abnormalities: Complete heart block, bundle branch blocks (granulomas in septum)."
- "Ventricular arrhythmias: VT, VF (scarring)."
- "Heart Failure: Restrictive or dilated cardiomyopathy."
- "Sudden Cardiac Death: Can be the first presentation. [11]"
Neurological Involvement (5-10%)
- Cranial Neuropathies: Facial nerve (VII) palsy is most common (often bilateral). Optic neuritis (II).
- Meningeal: Aseptic meningitis (chronic).
- Parenchymal: Space-occupying lesions (seizures), hypothalamic/pituitary dysfunction (diabetes insipidus).
- Peripheral: Mononeuritis multiplex, small fibre neuropathy (painful). [12]
Lymphatic (30-40%)
- Painless lymphadenopathy (cervical, axillary, inguinal, epitrochlear).
Other Organs
- Liver: Hepatomegaly, elevated ALP/GGT. Usually asymptomatic but can cause cirrhosis/portal hypertension.
- Spleen: Splenomegaly, hypersplenism (thrombocytopenia).
- Calcium: Hypercalcaemia (10%), Hypercalciuria (30%), Nephrocalcinosis, Renal stones.
- Musculoskeletal: Arthralgia (ankles/knees), acute polyarthritis (Löfgren's), dactylitis (sausage digits), bone cysts (lace-like pattern on X-ray).
- Parotid: Bilateral painless swelling.
5. Investigations and Diagnosis
The ATS/ERS 2020 Guideline emphasises three pillars for diagnosis:
- Compatible clinical/radiological presentation.
- Evidence of non-caseating granulomas.
- Exclusion of alternative causes. [1]
1. Imaging
Chest X-Ray (CXR) - Scadding Staging
Used for prognostic stratification, not just diagnosis.
| Stage | Radiographic Findings | Frequency at Presentation | Spontaneous Remission |
|---|---|---|---|
| 0 | Normal chest radiograph | 5-10% | N/A |
| I | Bilateral Hilar Lymphadenopathy (BHL) only | 50% | 60-90% (Good) |
| II | BHL + Pulmonary Infiltration | 25-30% | 40-70% (Fair) |
| III | Pulmonary Infiltration without BHL | 15% | 10-20% (Poor) |
| IV | Pulmonary Fibrosis (honeycombing, volume loss) | 5-10% | 0% (Irreversible) |
High-Resolution CT (HRCT)
More sensitive than CXR.
- Typical Findings: Micronodules with perilymphatic distribution (along fissures, bronchovascular bundles, subpleural). Upper/mid-zone predominance.
- Lymphadenopathy: Hilar and mediastinal ("potato nodes"), often with calcification ("eggshell" calcification).
- Fibrosis: Traction bronchiectasis, architectural distortion, honeycombing (Stage IV).
- Alveolar Sarcoid: Ground-glass opacities (active alveolitis) or consolidation (galaxy sign). [13]
FDG-PET/CT
- Useful for identifying occult disease sites for biopsy.
- Critical for diagnosing Cardiac Sarcoidosis (patchy uptake, mismatch with perfusion). [11]
2. Tissue Diagnosis (Biopsy)
Biopsy is required for most cases, except classical Löfgren's syndrome.
- Bronchoscopy:
- "Endobronchial Ultrasound (EBUS-TBNA): Gold standard for sampling hilar/mediastinal nodes. High yield (> 80%)."
- "Transbronchial Biopsy (TBB): Samples lung parenchyma."
- "Endobronchial Biopsy: If mucosal lesions are seen."
- Other Sites: Skin, palpable lymph nodes, lacrimal gland, conjunctiva.
- Blind Biopsies: E.g., minor salivary gland or conjunctival biopsy (lower yield, used if bronchoscopy unsafe).
3. Laboratory Tests
- Serum ACE: Elevated in 60-70%. Reflects total granuloma burden. Low sensitivity and specificity. Useful for monitoring serial trends rather than diagnosis.
- Calcium: Serum calcium (hypercalcaemia) and 24h urinary calcium (hypercalciuria).
- Renal Function: Creatinine (renal sarcoid or nephrocalcinosis).
- Liver Function: Elevated ALP/GGT (hepatic infiltration).
- Full Blood Count: Lymphopenia (sequestration), mild anaemia, thrombocytopenia (splenomegaly).
- Inflammatory Markers: ESR often elevated; CRP usually normal unless acute (EN) or infection.
4. Specialised Tests
- ECG: Mandatory for all patients (look for AV block, bundle branch block).
- Echocardiogram: If cardiac symptoms or abnormal ECG.
- Cardiac MRI: Gold standard for diagnosing cardiac sarcoidosis (Late Gadolinium Enhancement).
- Pulmonary Function Tests (PFTs):
- Restriction (reduced FVC, TLC).
- Reduced diffusion capacity (DLCO) - often the first parameter to fall.
- Obstruction (in endobronchial disease).
Exam Detail: #### Diagnostic Confidence (ATS 2020)
- "Definite: Compatible clinical features + biopsy showing granulomas + exclusion of other causes."
- "Probable: Compatible features + biopsy not performed/non-diagnostic + typical clinical course (e.g., Löfgren's)."
- "Possible: Atypical features, awaiting confirmation."
Note on Löfgren's: The guideline states biopsy is not needed if the presentation is classic Löfgren's (EN + BHL). [1]
6. Management
Management is guided by the ERS 2021 Treatment Guideline. [3]
To Treat or Not To Treat?
- Do NOT treat: Asymptomatic Stage I/II pulmonary sarcoidosis (observation is standard). Asymptomatic mild LFT abnormalities.
- DO treat:
- "Organ-threatening disease: Eye, Heart, Neuro, Kidney."
- "Severe pulmonary disease: Worsening symptoms (dyspnoea) or deteriorating PFTs (> 10% drop in FVC or > 15% drop in DLCO)."
- "Significant constitutional symptoms: Disabling fatigue, fever, weight loss."
- Hypercalcaemia.
- Disfiguring cutaneous lesions (Lupus pernio).
Pharmacotherapy
1. First-Line: Corticosteroids
- Pulmonary Sarcoidosis: Prednisolone 20–40 mg/day orally.
- Continue for 4–6 weeks, then slow taper.
- "Maintenance: 5–10 mg/day."
- "Total duration: usually 12 months minimum to prevent relapse."
- Acute Sarcoidosis (Löfgren's): NSAIDs alone often suffice. Short course steroids if severe.
- Cardiac/Neuro: Higher doses (1 mg/kg) or IV Methylprednisolone pulses. [3]
2. Second-Line: Antimetabolites (Steroid-Sparing)
Indicated if: steroid toxicity, inability to taper prednisone less than 10mg/day, or steroid failure.
- Methotrexate (MTX): Gold standard second-line.
- "Dose: 10–25 mg weekly + Folic acid."
- "Toxicity: Hepatotoxicity, marrow suppression, pneumonitis (rare)."
- Azathioprine: 50–150 mg daily. Alternative to MTX.
- Leflunomide: 10–20 mg daily. Usually third choice.
- Mycophenolate Mofetil: Often used for neurosarcoidosis. [3, 14]
3. Third-Line: Biologics (TNF-alpha Inhibitors)
For refractory disease despite steroids + antimetabolite.
- Infliximab: Chimeric monoclonal antibody.
- "Dose: 3–5 mg/kg IV at weeks 0, 2, 6, then every 4–8 weeks."
- "High efficacy for: Lupus pernio, Neurosarcoidosis, Refractory pulmonary disease."
- Adalimumab: Fully human monoclonal antibody. SC injection.
- Note: Etanercept is not effective in sarcoidosis. [3, 15]
Advanced Therapy
- Lung Transplantation: For end-stage pulmonary fibrosis (Stage IV). Recurrence in the transplanted lung occurs but is rarely clinically significant.
- Pacemaker/ICD: For high-grade heart block or VT risk in cardiac sarcoidosis.
7. Special Situations
Pregnancy and Sarcoidosis
Sarcoidosis frequently affects women of childbearing age.
- Outcomes: Pregnancy outcomes are generally good. Sarcoidosis does not increase the risk of congenital malformations.
- Disease Course:
- Many women (60%) experience improvement or stability during pregnancy due to increased endogenous cortisol.
- Relapse is common postpartum (within 3-6 months).
- Management:
- "Chest X-rays: Avoid if possible, but safe with shielding if clinical suspicion is high."
- "Medications:"
- Prednisolone: Safe (category B). Enzyme 11β-HSD2 in the placenta deactivates 90% of prednisolone, protecting the fetus.
- Methotrexate: CONTRAINDICATED (teratogenic). Must stop 3 months before conception.
- Hydroxychloroquine: Generally considered safe.
- Infliximab: Often continued until 3rd trimester.
- Preeclampsia: Some studies suggest a slightly increased risk. [16]
Sarcoidosis-Associated Fatigue
Fatigue is a major burden, affecting up to 80% of patients, even those with "inactive" disease.
- Causes:
- Active granulomatous inflammation (cytokines IL-1, IL-6, TNF).
- Sleep apnoea (common in sarcoidosis).
- Small fibre neuropathy.
- Depression/Anxiety.
- Hypothyroidism.
- Management:
- Treat active disease.
- Screen for OSA.
- "Stimulants: Methylphenidate or Modafinil (off-label, limited evidence)."
- "Exercise: Pulmonary rehabilitation improves fatigue scores. [17]"
Small Fibre Neuropathy (SFN)
Often overlooked.
- Symptoms: Burning pain, paraesthesiae, autonomic dysfunction (POTS, gastroparesis), but normal nerve conduction studies (which assess large fibres).
- Diagnosis: Skin biopsy (reduced intra-epidermal nerve fibre density).
- Treatment: IVIG, Gabapentin, Anti-TNF agents. [18]
8. Pharmacology in Depth
Exam Detail: #### Mechanism of Action of Key Agents
1. Glucocorticoids (Prednisolone)
- "Mechanism: Bind to cytosolic glucocorticoid receptor (GR). The complex translocates to the nucleus."
- "Genomic Effect: "
- Transrepression: Inhibits transcription factors AP-1 and NF-κB, switching off pro-inflammatory genes (IL-1, IL-2, TNF-α, IFN-γ).
- Transactivation: Up-regulates anti-inflammatory genes (Annexin A1).
- "Sarcoid-Specific: Induces apoptosis of Th1 lymphocytes and inhibits granuloma formation."
2. Methotrexate (MTX)
- "Mechanism: Folate antagonist. Inhibits Dihydrofolate Reductase (DHFR), interfering with DNA synthesis in rapidly dividing cells (lymphocytes)."
- "Anti-inflammatory: Increases extracellular Adenosine, a potent anti-inflammatory mediator that binds to A2A receptors on immune cells, downregulating TNF-α."
3. Infliximab (Anti-TNF)
- "Mechanism: Chimeric monoclonal antibody (IgG1) against TNF-α."
- "Action: Binds soluble and transmembrane TNF-α."
- "Granuloma Effect: TNF-α is critical for maintaining the structural integrity of the granuloma. Blockade leads to granuloma disintegration."
- "Caution: Reactivation of latent TB (granuloma breakdown). Screening with IGRA/Mantoux is mandatory pre-treatment."
9. Patient Information Layer
What is Sarcoidosis?
Sarcoidosis is a condition where tiny lumps called "granulomas" develop in the body's tissues. It's like the immune system is overreacting to something, but we don't know exactly what triggers it yet.
Key Points for Patients
- It is NOT cancer.
- It is NOT contagious. You cannot catch it from or give it to anyone.
- Most people get better. About 2 out of 3 people recover completely without any long-term problems.
- Treatment isn't always needed. If your symptoms are mild, your doctor might just monitor you. The medicines (steroids) can have side effects, so we only use them when necessary.
When to Seek Help
Contact your specialist if you notice:
- Worsening breathlessness.
- Palpitations (heart fluttering) or fainting.
- Changes in your vision (blurred vision, red eye).
- Severe fatigue or joint pain.
10. Historical Note
- Jonathan Hutchinson (1877): Described the first case of cutaneous sarcoidosis ("Mortimer's Malady").
- Ernest Besnier (1889): Described "Lupus Pernio" (purple nose).
- Caesar Boeck (1899): Coined the term "Sarkoid" (flesh-like) and noted the histological resemblance to sarcoma but benign nature.
- Jorgen Schaumann (1917): Recognised it as a systemic disease. Hence the old name: Besnier-Boeck-Schaumann Disease.
11. Clinical Monitoring Checklist
| Frequency | Clinical Assessment | Investigations |
|---|---|---|
| Every Visit | Dyspnoea score (MRC), Cough severity, Palpitations, Vision check, Skin check, Weight | Oxygen saturation (resting/exertion) |
| 3-6 Monthly | Fatigue score, Joint symptoms | CXR (if pulmonary), Spirometry (FVC, FEV1), ACE, Renal function, Calcium |
| Annual | Full systems review | ECG (Mandatory), Ophthalmology review (if on hydroxychloroquine or history of uveitis), Vitamin D (25-OH and 1,25-OH), 24h Urinary Calcium |
| As Needed | Neurological symptoms | HRCT (if functional decline), Cardiac MRI (if palpitations/syncope), Holter monitor |
12. Prognosis and Complications
Prognosis
- Spontaneous Remission: 60–70% of all patients.
- "Stage I: > 80% remission."
- "Stage II: 60% remission."
- "Stage III: 20% remission."
- Chronic/Progressive: 10–30%.
- Mortality: 1–5%. Most common causes of death are:
- Advanced Pulmonary Fibrosis (Respiratory Failure).
- Cardiac Sarcoidosis (Sudden Death).
- Neurosarcoidosis. [4]
Complications
- Pulmonary Hypertension: Due to fibrosis or vascular granulomas. Poor prognostic sign.
- Aspergilloma: Fungal ball forming in fibrotic cavities (Stage IV). Risk of massive haemoptysis.
- Bronchiectasis: Traction bronchiectasis from fibrosis.
13. Differential Diagnosis
| Condition | Distinguishing Features |
|---|---|
| Tuberculosis | Caseating granulomas, positive culture/PCR, necrotic centres, upper lobe cavitation. |
| Lymphoma | B-symptoms prominent, specific cytology/histology, clonal population. |
| Lung Cancer | Mass lesion, distinct histology. Sarcoid-like reaction can occur in draining nodes of cancer. |
| Berylliosis | Occupational history (aerospace, electronics), positive BeLPT test. Identical histology to sarcoid. |
| Hypersensitivity Pneumonitis | Exposure history (birds, mould), centrilobular nodules, air trapping, poorly formed granulomas. |
| GPA (Wegener's) | ANCA positive, necrotising vasculitis, renal involvement (glomerulonephritis). |
| Common Variable Immunodeficiency (CVID) | Can cause GLILD (Granulomatous-Lymphocytic Interstitial Lung Disease). Check immunoglobulins. |
14. References
-
Crouser ED, Maier LA, Wilson KC, et al. Diagnosis and Detection of Sarcoidosis. An Official ATS Clinical Practice Guideline. Am J Respir Crit Care Med. 2020;201(8):e26-e51. PMID: 32293205
-
Iannuzzi MC, Rybicki BA, Teirstein AS. Sarcoidosis. N Engl J Med. 2007;357(21):2153-2165. PMID: 18032765
-
Baughman RP, Valeyre D, Korsten P, et al. ERS clinical practice guidelines on treatment of sarcoidosis. Eur Respir J. 2021;58(6):2004079. PMID: 34140301
-
Statement on sarcoidosis. Joint Statement of the American Thoracic Society (ATS), the European Respiratory Society (ERS) and the World Association of Sarcoidosis and Other Granulomatous Disorders (WASOG) adopted by the ATS Board of Directors and by the ERS Executive Committee, February 1999. Am J Respir Crit Care Med. 1999;160(2):736-755. PMID: 10430755
-
Arkema EV, Cozier YC. Epidemiology of sarcoidosis: current findings and future directions. Ther Adv Chronic Dis. 2018;9(11):227-240. PMID: 30416705
-
Ungprasert P, Crowson CS, Matteson EL. Epidemiology and Clinical Characteristics of Sarcoidosis: An Update from a Population-Based Cohort Study. Mayo Clin Proc. 2017;92(2):189-200. PMID: 28160872
-
Grunewald J, Spagnolo P, Wahlström J, Eklund A. Immunogenetics of sarcoidosis. Curr Opin Pulm Med. 2015;21(5):503-509. PMID: 26186483
-
Patterson KC, Chen ES. The pathogenesis of pulmonary sarcoidosis and implications for treatment. Chest. 2018;153(6):1432-1442. PMID: 29248431
-
Nunes H, Bouvry D, Soler P, Valeyre D. Sarcoidosis. Orphanet J Rare Dis. 2007;2:46. PMID: 18036239
-
Spagnolo P, Rossi G, Trisolini R, et al. Pulmonary sarcoidosis. Lancet Respir Med. 2018;6(5):389-402. PMID: 29656983
-
Birnie DH, Sauer WH, Bogun F, et al. HRS expert consensus statement on the diagnosis and management of arrhythmias associated with cardiac sarcoidosis. Heart Rhythm. 2014;11(7):1305-1323. PMID: 24819193
-
Stern BJ, Royal W 3rd, Gelfand JM, et al. Definition and Diagnostic Criteria of Neurosarcoidosis: From the Neurosarcoidosis Consortium Consensus Group. JAMA Neurol. 2018;75(12):1546-1553. PMID: 30167655
-
Lynch JP 3rd, Ma YL, Koss MN, White ES. Pulmonary sarcoidosis. Semin Respir Crit Care Med. 2007;28(1):53-74. PMID: 17330192
-
Hamzeh N, Voelker A, Forssén A, et al. Efficacy of mycophenolate mofetil in sarcoidosis. Respir Med. 2014;108(11):1663-1669. PMID: 25262299
-
Baughman RP, Drent M, Kavuru M, et al. Infliximab therapy in patients with chronic sarcoidosis and pulmonary involvement. Am J Respir Crit Care Med. 2006;174(7):795-802. PMID: 16809633
-
Hadid T, Alsawah F, Littlefield A, et al. Sarcoidosis and pregnancy: pregnancy outcomes and maternal complications. Respir Med. 2015;109(5):652-653. PMID: 25801931
-
Drent M, Lower EE, De Vries J. Sarcoidosis-associated fatigue. Eur Respir J. 2012;40(1):255-263. PMID: 22753856
-
Tavee J, Culver D. Sarcoidosis and small-fiber neuropathy. Curr Pain Headache Rep. 2011;15(3):201-206. PMID: 21286873
15. Examination Focus
Viva Questions & Model Answers
Q: What is Löfgren's syndrome and how does it influence management?
A: Löfgren's syndrome is an acute form of sarcoidosis presenting with the triad of erythema nodosum, bilateral hilar lymphadenopathy, and polyarthritis (often ankles). It is significant because it has an excellent prognosis (> 90% spontaneous remission). Biopsy is typically not required if the presentation is classic. Management is usually supportive with NSAIDs; steroids are rarely needed unless symptoms are severe.
Q: How do you stage sarcoidosis on CXR?
A: Using the Scadding staging system:
- Stage 0: Normal.
- Stage I: Bilateral hilar lymphadenopathy (BHL) only.
- Stage II: BHL + pulmonary infiltrates.
- Stage III: Pulmonary infiltrates without BHL.
- Stage IV: Pulmonary fibrosis. Note that staging does not always correlate with symptom severity but correlates with prognosis (Stage I best, Stage IV worst).
Q: When would you initiate corticosteroid treatment?
A: Treatment is indicated for:
- Organ-threatening disease (Cardiac, Neuro, Eye, severe hypercalcaemia).
- Progressive symptomatic pulmonary disease (worsening lung function or dyspnoea).
- Disfiguring skin disease (Lupus pernio). Asymptomatic pulmonary sarcoidosis (Stage I/II) should be observed, not treated.
Q: What is the significance of the CD4:CD8 ratio in BAL?
A: A ratio > 3.5 is highly specific (94-96%) for sarcoidosis, but not diagnostic in isolation. It reflects the Th1 alveolitis. A normal or low ratio does not exclude the diagnosis.
Q: What are the features of cardiac sarcoidosis?
A: It can present with conduction defects (AV block), ventricular arrhythmias (VT), or heart failure (cardiomyopathy). It is a leading cause of death in sarcoidosis. Diagnosis involves ECG, Echo, and Cardiac MRI (LGE) or PET-CT.
Common Exam Pitfalls
- Misdiagnosis: Treating "sarcoid" without biopsy, only to miss Lymphoma or Tuberculosis.
- Overtreatment: Starting steroids for asymptomatic BHL (Stage I).
- Undertreatment: Failing to screen for cardiac or eye involvement.
- Calcium: Forgetting that hypercalcaemia is mediated by 1,25-dihydroxyvitamin D produced by the granuloma itself.
Last Updated: 2026-01-06