Acute Stroke in Adults
Critical Alerts "Time is Brain" : 1.9 million neurons lost per minute in large vessel occlusion FAST recognition : Face-Arm-Speech-Time enables immediate stroke pathway activation Thrombolysis window :...
What matters first
Critical Alerts "Time is Brain" : 1.9 million neurons lost per minute in large vessel occlusion FAST recognition : Face-Arm-Speech-Time enables immediate stroke pathway activation Thrombolysis window :...
Sudden onset focal neurological deficit (FAST positive)
9 Jan 2026
Generated educational material; verify before clinical use.
Visible references section
See the concept before reading it
Study the key anatomy, imaging, and decision pathways as full teaching plates.
Clinical board
A visual summary of the highest-yield teaching signals on this page.
Urgent signals
Safety-critical features pulled from the topic metadata.
- Sudden onset focal neurological deficit (FAST positive)
- Sudden 'worst headache of life' (Thunderclap headache suggesting SAH)
- Rapidly declining Level of Consciousness (GCS)
- New onset seizures following focal deficit
Linked comparisons
Differentials and adjacent topics worth opening next.
- Transient Ischaemic Attack (TIA)
- Hypoglycaemia
Content status and exam context
This page is AI-generated educational content. It may contain errors or omissions and is not a substitute for current guidelines, local protocols, senior clinical judgement, or professional medical advice.
MedVellum does not claim an individual clinician reviewer, board certification, or professional credential for this page unless a future version names a real, verifiable reviewer.
Clinical explanation and evidence
Acute Stroke in Adults
Quick Reference
Critical Alerts
- "Time is Brain": 1.9 million neurons lost per minute in large vessel occlusion [1]
- FAST recognition: Face-Arm-Speech-Time enables immediate stroke pathway activation
- Thrombolysis window: Alteplase/Tenecteplase within 4.5 hours of symptom onset [2]
- Thrombectomy window: 6-24 hours for large vessel occlusion with imaging selection (DAWN/DEFUSE-3 criteria) [3,4]
- Exclude hemorrhage first: Non-contrast CT mandatory before any reperfusion therapy
- Blood pressure targets: less than 185/110 mmHg pre-thrombolysis; permissive hypertension otherwise
- Door-to-needle target: less than 60 minutes for thrombolysis administration
- Door-to-groin puncture: less than 90 minutes for mechanical thrombectomy
Ischemic vs Hemorrhagic Stroke Differentiation
| Feature | Ischemic Stroke (85%) | Hemorrhagic Stroke (15%) |
|---|---|---|
| Onset | Sudden focal deficit | Sudden with severe headache |
| Headache | Uncommon at onset | Common, severe ("worst ever") |
| Vomiting | Uncommon | Common (increased ICP) |
| Consciousness | Often preserved initially | Often impaired early |
| CT appearance | Normal early; hypodensity later | Hyperdense (white) blood |
| Blood pressure | Variable, often elevated | Usually markedly elevated |
| Neck stiffness | Absent | May be present (SAH) |
| Treatment | Thrombolysis/Thrombectomy | BP control, reversal agents, surgery |
| Mortality | 10-15% at 30 days | 40-50% at 30 days |
Emergency Stroke Protocol
| Time | Action | Target |
|---|---|---|
| 0-10 min | ABCDE, Blood glucose, IV access | Immediate stabilization |
| 10-20 min | NIHSS score, Last known well time | Triage for reperfusion |
| 20-30 min | Non-contrast CT ± CTA | Hemorrhage exclusion, LVO identification |
| 30-60 min | Thrombolysis decision and administration | Door-to-needle less than 60 min |
| 60-90 min | Thrombectomy (if LVO) | Door-to-groin less than 90 min |
SECTION 1: Clinical Overview
1.1 Definition and Classification
Acute stroke is defined as a clinical syndrome characterized by the sudden onset of focal or global neurological deficits lasting more than 24 hours (or leading to death) with no apparent cause other than vascular origin. The World Health Organization (WHO) definition emphasizes the vascular aetiology and distinguishes stroke from transient ischaemic attack (TIA), which by modern tissue-based definition implies no evidence of acute infarction on neuroimaging. [1]
Classification by Pathophysiology:
| Type | Percentage | Mechanism | Key Features |
|---|---|---|---|
| Ischemic Stroke | 85% | Arterial occlusion (thrombotic or embolic) | Focal deficit in vascular territory |
| Intracerebral Hemorrhage | 10% | Vessel rupture into brain parenchyma | Severe headache, rapid deterioration |
| Subarachnoid Hemorrhage | 5% | Bleeding into subarachnoid space | Thunderclap headache, meningism |
1.2 Summary
Acute stroke remains the second leading cause of death worldwide and the leading cause of adult disability in developed countries. The incidence approaches 795,000 new or recurrent strokes annually in the United States, with approximately 15 million strokes occurring globally each year. [5] The concept of "Time is Brain" reflects the urgency of intervention, as approximately 1.9 million neurons are lost every minute during a large vessel occlusion. [1]
The management paradigm has evolved dramatically with the advent of reperfusion therapies. Intravenous thrombolysis with alteplase or tenecteplase remains the standard of care for eligible patients within 4.5 hours of symptom onset. [2] The landmark thrombectomy trials (MR CLEAN, ESCAPE, SWIFT PRIME, EXTEND-IA, REVASCAT) established mechanical thrombectomy as the most effective treatment in acute medicine for large vessel occlusion, with a number needed to treat of 2.6. [6,7]
Extended window thrombectomy, as demonstrated by the DAWN and DEFUSE-3 trials, allows intervention up to 24 hours in selected patients with favorable imaging profiles demonstrating salvageable penumbra. [3,4] Comprehensive stroke unit care with multidisciplinary input further improves outcomes compared to general medical ward management. [8]
1.3 Key Facts
| Parameter | Value | Source/Notes |
|---|---|---|
| Incidence | 200 per 100,000 person-years | Higher in elderly and minorities |
| Prevalence | 2.5% of adults | Approximately 9.4 million survivors in US |
| Lifetime risk | 1 in 4 adults over age 25 | WHO Global Burden of Disease [5] |
| Mortality (ischemic) | 10-15% at 30 days | Lower in specialized stroke units |
| Mortality (hemorrhagic) | 40-50% at 30 days | Higher mortality than ischemic |
| Disability | 50% of survivors | Reduced mobility over age 65 |
| Peak age | 75-85 years | Risk doubles each decade after 55 |
| Sex distribution | Men > Women (younger); Women > Men (older) | Women have higher lifetime risk |
| Time to intervention | Every 15 min delay = 1 month disability | HERMES meta-analysis [6] |
| Thrombolysis NNT | 10 | For mRS 0-1 at 3 months |
| Thrombectomy NNT | 2.6 | For reduction in disability by 1 mRS grade |
1.4 Clinical Pearls
Diagnostic Pearl - "The Sudden in Stroke": The most critical diagnostic feature is the temporal profile. True strokes reach maximal intensity within seconds to minutes. Gradual onset over hours suggests alternative diagnoses (malignancy, demyelination, metabolic derangement).
Examination Pearl - "Pronator Drift Test": Have the patient hold arms out with palms up and eyes closed. The affected side will slowly pronate and drift downward due to weakness in supinators and extensors — a sensitive indicator of subtle upper motor neuron weakness.
Treatment Pearl - "Permissive Hypertension": Do not aggressively lower blood pressure unless it exceeds 220/120 mmHg (or 185/110 if thrombolysing). Premature lowering collapses collateral flow to the penumbra, potentially extending the infarct. [9]
Pitfall Warning - "The Posterior Circulation Trap": Do not rely solely on FAST criteria. Posterior circulation strokes often present with the "5 Ds": Dizziness, Diplopia, Dysarthria, Dysphagia, Dystaxia. These are frequently missed in emergency settings.
Mnemonic - "BE FAST": Balance (loss), Eyes (vision loss), Face (drooping), Arms (weakness), Speech (difficulty), Time (call emergency services). This expanded mnemonic captures posterior circulation and sensory strokes.
Emergency Pearl - "Hypoglycemia is the Great Mimicker": Always check capillary blood glucose immediately. Hypoglycemia can perfectly mimic focal stroke syndromes, and administering thrombolysis to a hypoglycemic patient would be catastrophic.
Exam Pearl - "NIHSS Limitations": The NIH Stroke Scale is weighted toward anterior circulation (motor/speech). A high NIHSS usually indicates large territory stroke, but a LOW NIHSS does NOT exclude disabling stroke (e.g., isolated distal PCA infarct causing hemianopia).
Wake-up Stroke Pearl: 20-25% of strokes occur during sleep. MRI DWI-FLAIR mismatch or CT perfusion imaging can identify treatment candidates for reperfusion therapy. [10]
1.5 Why This Matters Clinically
Acute stroke represents a true medical emergency where the window for intervention is measured in minutes. Delayed or missed diagnosis results in loss of the opportunity for reperfusion, leading to massive neuronal death and permanent disability. This translates to significant loss of quality-adjusted life years (QALYs) and places an immense burden on patients, families, and healthcare systems.
Healthcare Economics: The cost of acute care, inpatient rehabilitation, and long-term nursing care for a single major stroke can exceed $140,000 in the first year alone. Lifetime costs approach $200,000-$300,000 per patient. The annual economic burden of stroke in the United States exceeds $56.5 billion. [5]
Medico-Legal Considerations: Stroke management is a high-risk area for litigation. Common grounds for claims include:
- Failure to recognize stroke symptoms
- Delay in ordering appropriate imaging
- Failure to administer thrombolysis within the evidence-based window
- Failure to refer for thrombectomy when indicated
- Inadequate documentation of treatment decisions and timing
Training Importance: Stroke is a cornerstone of medical education because it integrates anatomy (Circle of Willis, vascular territories), physiology (cerebral blood flow, autoregulation), pharmacology (thrombolytics, antiplatelets), and clinical reasoning under time pressure.
SECTION 2: Epidemiology
2.1 Incidence and Prevalence
| Parameter | Data | Clinical Significance |
|---|---|---|
| Annual incidence (USA) | 795,000 strokes | ~610,000 new, ~185,000 recurrent |
| Annual incidence (Global) | 15 million strokes | 5 million deaths, 5 million permanent disability |
| Prevalence (USA) | 9.4 million survivors | Leading cause of adult disability |
| Lifetime risk | 1 in 4 adults (age > 25) | Higher in women due to longer lifespan |
| Trend | Decreasing age-adjusted mortality | But increasing absolute numbers due to population aging |
| Geographic variation | "Stroke Belt" in SE USA | 20% higher mortality in southeastern states |
| Temporal pattern | Peak 6 AM to noon | Coincides with morning BP surge and platelet activation |
| Seasonal variation | Higher in winter months | Related to infection, dehydration, and cold |
Healthcare Burden: Direct and indirect costs of stroke in the US are estimated at $56.5 billion annually. This includes acute hospitalization, rehabilitation, long-term care, lost productivity, and caregiver burden. [5]
2.2 Demographics and Risk Stratification
| Factor | Details | Clinical Significance |
|---|---|---|
| Age | Peak 75-85 years; risk doubles every decade after 55 | Age is the strongest non-modifiable risk factor |
| Sex | Men > Women until age 85; then Women > Men | Women have higher mortality and worse functional outcomes |
| Ethnicity | Black and Hispanic populations have 2× risk vs White | Higher prevalence of hypertension and diabetes |
| Geography | High in Eastern Europe, SE Asia, SE USA | Linked to diet, smoking, healthcare access |
| Socioeconomic | Lower income associated with 30-50% higher risk | Limited access to prevention and healthy lifestyle |
| Occupation | High-stress, sedentary jobs increase risk | Shift work correlates with circadian disruption |
2.3 Risk Factors
Non-Modifiable Risk Factors:
| Factor | Relative Risk (95% CI) | Mechanism |
|---|---|---|
| Age (per decade) | RR 2.0 (1.8-2.2) | Arterial stiffening, cumulative vascular damage |
| Male sex | RR 1.3 (1.1-1.5) | Lack of protective estrogen in pre-menopausal years |
| Family history | RR 1.7 (1.4-2.1) | Genetic predisposition to HTN, DM, hyperlipidemia |
| Prior stroke/TIA | RR 10.0 (8.0-12.0) | Established high-burden atherosclerotic disease |
| Low birth weight | RR 1.2 (1.0-1.4) | Epigenetic programming of vascular resistance |
| Genetic conditions | Variable | CADASIL, sickle cell disease, Fabry disease |
Modifiable Risk Factors:
| Factor | Relative Risk | Population Attributable Risk | Intervention Impact |
|---|---|---|---|
| Hypertension | RR 4.0 (3.2-5.1) | 35-50% | 40% reduction with optimal BP control |
| Atrial fibrillation | RR 5.0 (4.0-6.2) | 15-20% | 60-70% reduction with anticoagulation |
| Smoking | RR 1.9 (1.7-2.2) | 12-14% | Risk returns to baseline 5 years after quitting |
| Diabetes mellitus | RR 2.3 (1.8-2.9) | 5-8% | Reduces micro/macrovascular complications |
| Hyperlipidemia | RR 1.5 (1.3-1.8) | 5-10% | 20-30% reduction with high-intensity statins |
| Obesity | RR 1.6 (1.4-1.9) | 5-8% | Weight loss improves all metabolic parameters |
| Physical inactivity | RR 1.4 (1.2-1.6) | 10-15% | Regular exercise reduces stroke risk by 27% |
| Excessive alcohol | RR 1.6 (1.4-1.9) | 3-5% | J-shaped curve; heavy use increases risk |
| Diet (poor quality) | RR 1.4 (1.2-1.7) | 10-15% | Mediterranean diet reduces risk by 20% |
2.4 Protective Factors
| Factor | Relative Risk Reduction | Mechanism |
|---|---|---|
| Physical activity | RR 0.73 | Improved endothelial function, weight control |
| Mediterranean diet | RR 0.80 | High polyphenols, healthy fats reduce oxidative stress |
| Moderate alcohol | RR 0.85 | U-shaped curve; > 2 drinks/day increases risk |
| Fruits and vegetables | RR 0.85 | Potassium, antioxidants, fiber |
| Fish consumption | RR 0.90 | Omega-3 fatty acids, anti-inflammatory effects |
SECTION 3: Pathophysiology
3.1 The Ischemic Cascade
Step 1: Initiating Event - Vascular Occlusion
The pathophysiology begins with abrupt cessation of cerebral blood flow (CBF) due to arterial occlusion. This may occur via:
- Thrombotic occlusion: In-situ thrombus formation over atherosclerotic plaque
- Embolic occlusion: Embolus from cardiac source (atrial fibrillation) or artery-to-artery (carotid stenosis)
- Small vessel occlusion: Lipohyalinosis of penetrating arteries (lacunar stroke)
CBF Thresholds:
| CBF Level | Consequence |
|---|---|
| Normal: 50-60 mL/100g/min | Normal neuronal function |
| 20-50 mL/100g/min | Electrical dysfunction begins |
| 10-20 mL/100g/min | Penumbra - reversible ischemia, neurons viable but dysfunctional |
| less than 10 mL/100g/min | Ischemic core - irreversible injury, neurons dying |
Step 2: Energy Failure and Ionic Disruption
When CBF falls below 10-12 mL/100g/min:
- ATP depletion: Aerobic metabolism fails within seconds
- Na+/K+ ATPase pump failure: Loss of ionic gradients
- Anoxic depolarization: Mass influx of Na+, Cl-, and water
- Cytotoxic edema: Cell swelling occurs within minutes
Step 3: Excitotoxicity and Calcium Overload
Depolarizing neurons release massive amounts of glutamate:
- NMDA/AMPA receptor activation: Massive Ca²⁺ influx
- Intracellular calcium levels rise 1000-fold
- Destructive enzyme activation: Calpains, lipases, endonucleases
- Mitochondrial damage: mPTP opening, cytochrome C release
Step 4: Oxidative Stress and Inflammation
Secondary injury cascade begins:
- ROS production: Superoxide, hydroxyl radicals
- Microglial activation: IL-1β, IL-6, TNF-α release via NF-κB pathway
- Adhesion molecule upregulation: ICAM-1, VCAM-1
- Leukocyte infiltration: Neutrophils (6-12 hours), macrophages (days)
Step 5: Blood-Brain Barrier Breakdown
Matrix metalloproteinase (MMP) release:
- MMP-9 degrades basal lamina: Tight junction proteins (claudin, occludin) disrupted
- Vasogenic edema: Plasma protein extravasation
- Hemorrhagic transformation: Red cell extravasation (especially post-thrombolysis)
- Mass effect: ICP elevation in large infarcts
Step 6: Programmed Cell Death
Penumbral cells may undergo apoptosis over hours to days:
- Caspase-3 activation: Intrinsic apoptotic pathway
- BAX/BCL-2 balance shifts: Pro-apoptotic signaling
- DNA fragmentation: Nuclear condensation
- Glial scar formation: Astrocyte proliferation at infarct periphery
3.2 The Ischemic Penumbra
The penumbra is the salvageable tissue surrounding the ischemic core:
┌─────────────────────────────────────────┐
│ ISCHEMIC CORE │
│ CBF less than 10 mL/100g/min │
│ Irreversible injury │
│ Infarction within minutes │
├─────────────────────────────────────────┤
│ PENUMBRA │
│ CBF 10-20 mL/100g/min │
│ Reversible ischemia │
│ SALVAGEABLE WITH REPERFUSION │
├─────────────────────────────────────────┤
│ OLIGEMIC ZONE │
│ CBF 20-50 mL/100g/min │
│ Electrical dysfunction │
│ Usually recovers spontaneously │
└─────────────────────────────────────────┘
Penumbra Duration: Varies from minutes to hours depending on:
- Collateral circulation adequacy
- Core size and location
- Systemic blood pressure
- Blood glucose levels
3.3 TOAST Classification (Ischemic Stroke Etiology)
| Type | Definition | % Cases | Key Features | Prognosis |
|---|---|---|---|---|
| Large artery atherosclerosis | > 50% stenosis of major vessel | 20-30% | Cortical/subcortical infarct, carotid bruit | High recurrence risk |
| Cardioembolism | Cardiac source of embolus | 20-25% | AF, valve disease, multiple territories | 20% mortality at 30 days |
| Small vessel (lacunar) | Lipohyalinosis of penetrating arteries | 15-25% | Pure motor/sensory, no cortical signs, less than 1.5cm | Good short-term recovery |
| Other determined etiology | Specific identifiable cause | 5-10% | Dissection, vasculitis, hypercoagulable, CADASIL | Depends on underlying cause |
| Cryptogenic/Undetermined | No cause found or multiple possible causes | 25-35% | Negative workup or competing etiologies | Variable; needs monitoring |
3.4 Hemorrhagic Stroke Pathophysiology
Intracerebral Hemorrhage (ICH):
- Hypertensive ICH: Rupture of lipohyalinotic microaneurysms in deep perforators (basal ganglia, thalamus, pons, cerebellum)
- Amyloid angiopathy: Lobar hemorrhages in elderly; high recurrence risk
- Secondary ICH: Tumors, vascular malformations, anticoagulation, hemorrhagic transformation
ICH Expansion: Hematoma expansion occurs in 30% of patients within 24 hours and is associated with worse outcomes. Early blood pressure control and coagulopathy reversal are critical. [11]
Subarachnoid Hemorrhage (SAH):
- 85% due to aneurysm rupture: Circle of Willis, ACOM most common
- 15% non-aneurysmal: Perimesencephalic, traumatic
- Complications: Vasospasm (peak days 4-14), hydrocephalus, rebleeding
SECTION 4: Clinical Presentation
4.1 Stroke Syndromes by Vascular Territory
Anterior Circulation (Carotid Territory):
| Syndrome | Vessel | Clinical Features |
|---|---|---|
| Total Anterior Circulation Syndrome (TACS) | MCA + ACA or ICA | All three of: Hemiplegia, Hemianopia, Higher cortical dysfunction (aphasia/neglect) |
| Partial Anterior Circulation Syndrome (PACS) | MCA cortical branches | Two of TACS criteria, OR isolated higher cortical dysfunction |
| Lacunar Syndrome (LACS) | Deep perforating arteries | Pure motor, Pure sensory, Sensorimotor, Ataxic hemiparesis, Dysarthria-clumsy hand |
Posterior Circulation (Vertebrobasilar Territory):
| Syndrome | Vessel | Clinical Features |
|---|---|---|
| Posterior Circulation Syndrome (POCS) | Vertebral, Basilar, PCA | Cranial nerve palsy + contralateral motor/sensory, Bilateral motor/sensory, Cerebellar signs, Isolated hemianopia |
| Basilar artery occlusion | Basilar artery | Quadriplegia, bulbar dysfunction, locked-in syndrome, coma |
| Cerebellar stroke | PICA, AICA, SCA | Ataxia, vertigo, nausea, headache, risk of herniation |
4.2 Specific Vascular Territory Syndromes
Middle Cerebral Artery (MCA) - Most Common:
| Feature | Dominant (Left) Hemisphere | Non-Dominant (Right) Hemisphere |
|---|---|---|
| Motor | Contralateral hemiparesis (face/arm > leg) | Contralateral hemiparesis (face/arm > leg) |
| Sensory | Contralateral hemisensory loss | Contralateral hemisensory loss |
| Visual | Contralateral homonymous hemianopia | Contralateral homonymous hemianopia |
| Language | Broca's (expressive) or Wernicke's (receptive) aphasia | Aprosodia (loss of emotional speech) |
| Attention | — | Hemispatial neglect (ignores left side) |
| Gaze | Eyes deviate toward lesion ("look at the lesion") | Eyes deviate toward lesion |
Anterior Cerebral Artery (ACA):
- Contralateral leg weakness > arm (motor strip distribution)
- Contralateral leg sensory loss
- Abulia (lack of will), apathy, mutism
- Urinary incontinence
- Alien hand syndrome (if corpus callosum involved)
Posterior Cerebral Artery (PCA):
- Contralateral homonymous hemianopia with macular sparing
- Visual agnosia
- Memory impairment (if hippocampus involved)
- Alexia without agraphia (dominant hemisphere)
Basilar Artery:
- Locked-in syndrome: Quadriplegia, facial paralysis, but preserved consciousness and vertical eye movements
- Coma if bilateral brainstem involvement
- Cranial nerve palsies
4.3 Lacunar Stroke Syndromes
| Syndrome | Location | Clinical Features |
|---|---|---|
| Pure Motor Hemiparesis | Posterior limb internal capsule, basis pontis | Equal weakness of face, arm, leg; no sensory/cortical signs |
| Pure Sensory Stroke | Thalamus (VPL/VPM nuclei) | Numbness/paresthesias face, arm, leg; no motor deficit |
| Sensorimotor Stroke | Thalamus + internal capsule | Combined motor and sensory deficit |
| Ataxic Hemiparesis | Posterior limb internal capsule, pons | Hemiparesis + ipsilateral cerebellar ataxia |
| Dysarthria-Clumsy Hand | Basis pontis, genu internal capsule | Dysarthria, facial weakness, hand clumsiness |
4.4 Symptoms Table
| Symptom | Frequency | Character | Vascular Territory |
|---|---|---|---|
| Hemiparesis | 80% | Sudden weakness | MCA (face/arm), ACA (leg) |
| Dysarthria | 60% | Slurred, "thick" speech | MCA, brainstem |
| Aphasia | 35% | Expressive (Broca) or Receptive (Wernicke) | Left MCA |
| Hemianopia | 25% | Visual field loss | MCA, PCA |
| Hemisensory loss | 40% | Numbness, tingling | MCA, thalamus |
| Ataxia | 20% | Incoordination | Cerebellum, brainstem |
| Vertigo | 15% | Room spinning | Posterior circulation |
| Headache | 15% (ischemic), 90% (hemorrhagic) | Variable severity | Hemorrhage > ischemia |
| Nausea/Vomiting | 15% | Associated with ICP elevation | Hemorrhage, posterior fossa |
| Decreased LOC | 10-15% | Obtundation, coma | Large infarct, hemorrhage, basilar |
4.5 Signs Table
| Sign | Technique | Positive Finding | Sensitivity | Specificity |
|---|---|---|---|---|
| Pronator drift | Arms extended, palms up, eyes closed | Pronation and drift downward | 79% | 90% |
| Facial droop | Smile, show teeth | Asymmetric lower face weakness | 85% | 80% |
| Dysphasia testing | Name objects, repeat phrases | Word-finding difficulty, paraphasic errors | 60% | 95% |
| Visual fields | Confrontation testing | Homonymous hemianopia | 50% | 98% |
| Babinski sign | Stroke lateral sole | Extensor plantar response (great toe up) | 50% | 99% |
| Gaze preference | Observe resting eye position | Eyes deviate toward lesion (ischemic) | 40% | 95% |
| Extinction | Simultaneous bilateral touch | Only feels one side | 30% | 99% |
| Finger-nose | Touch examiner's finger then nose | Dysmetria, past-pointing | 50% | 90% |
| NIHSS score | Standardized 11-item scale | Score 0-42, higher = more severe | High | High |
4.6 The NIH Stroke Scale (NIHSS)
The NIHSS is a standardized, validated 15-item assessment tool used to quantify stroke severity and guide treatment decisions. It has excellent inter-rater reliability (kappa 0.66-0.77) and strong correlation with infarct volume and clinical outcomes. [26]
Detailed Components and Scoring:
| Item | Test | Scoring (0 = Normal → Maximum = Worst) | Clinical Significance |
|---|---|---|---|
| 1a. Level of Consciousness | Alertness, responsiveness | 0=Alert; 1=Drowsy; 2=Stuporous; 3=Coma | LOC impairment suggests large infarct or brainstem involvement |
| 1b. LOC Questions | "What month?" "How old are you?" | 0=Both correct; 1=One correct; 2=Neither correct | Tests orientation; aphasic patients may score 2 |
| 1c. LOC Commands | "Close eyes" "Open/close hand" | 0=Both correct; 1=One correct; 2=Neither correct | Tests comprehension; differentiates aphasia from LOC |
| 2. Best Gaze | Horizontal eye movements | 0=Normal; 1=Partial palsy; 2=Forced deviation | Gaze deviation toward lesion (ischemia) or away (hemorrhage) |
| 3. Visual Fields | Confrontation testing all quadrants | 0=Normal; 1=Partial hemianopia; 2=Complete hemianopia; 3=Bilateral hemianopia | Posterior circulation or deep MCA involvement |
| 4. Facial Palsy | Show teeth, raise eyebrows, close eyes | 0=Normal; 1=Minor paralysis; 2=Partial paralysis; 3=Complete paralysis | Lower face worse = UMN; forehead sparing |
| 5a. Motor Arm - Left | Extend arm 90° (sitting) or 45° (lying) × 10 sec | 0=No drift; 1=Drift; 2=Some effort against gravity; 3=No effort against gravity; 4=No movement | Proximal weakness common in MCA strokes |
| 5b. Motor Arm - Right | Same as left | 0-4 (same as left) | Compare sides for asymmetry |
| 6a. Motor Leg - Left | Lift leg to 30° × 5 sec | 0=No drift; 1=Drift; 2=Some effort against gravity; 3=No effort against gravity; 4=No movement | ACA strokes cause leg > arm weakness |
| 6b. Motor Leg - Right | Same as left | 0-4 (same as left) | Assess for subtle asymmetry |
| 7. Limb Ataxia | Finger-nose-finger, heel-shin | 0=Absent; 1=Present in one limb; 2=Present in two limbs | Out of proportion to weakness = cerebellar |
| 8. Sensory | Pinprick face, arm, leg bilaterally | 0=Normal; 1=Mild loss; 2=Severe or total loss | Thalamic strokes cause dense hemisensory loss |
| 9. Best Language | Name objects, read sentences, describe picture | 0=Normal; 1=Mild aphasia; 2=Severe aphasia; 3=Mute/global aphasia | Dominant hemisphere (usually left) involvement |
| 10. Dysarthria | Read word list: "mama, tip-top, fifty-fifty" | 0=Normal; 1=Mild to moderate; 2=Severe; UN=Intubated | Cerebellar or brainstem pathology |
| 11. Extinction/Inattention | Simultaneous touch/visual stimuli | 0=No neglect; 1=Inattention to one modality; 2=Severe neglect | Non-dominant (usually right) hemisphere |
Total Score Range: 0-42 points
Score Interpretation and Clinical Implications:
| Score | Severity | Typical Deficits | Thrombolysis Decision | Thrombectomy Consideration |
|---|---|---|---|---|
| 0 | No stroke | Normal exam | Stroke mimic; do NOT lyse | Not indicated |
| 1-4 | Minor | Mild weakness, sensory loss | Shared decision-making; minor stroke protocol (DAPT) | Only if disabling deficit (e.g., isolated aphasia) |
| 5-15 | Moderate | Hemiparesis, hemianopia, aphasia | Strong indication for lysis | If LVO present on CTA |
| 16-20 | Moderate-severe | Dense hemiplegia ± global aphasia | Lyse AND urgent CTA for LVO | Very likely to have LVO |
| 21-42 | Severe | Total hemiplegia, coma, or global deficits | High hemorrhage risk but still beneficial if eligible | Likely LVO; check for large core |
Prognostic Value of NIHSS:
| Initial NIHSS | 3-Month Functional Independence (mRS 0-2) | 3-Month Mortality |
|---|---|---|
| 0-4 | 80-90% | 2-3% |
| 5-9 | 60-70% | 5-8% |
| 10-15 | 40-50% | 10-15% |
| 16-20 | 20-30% | 20-30% |
| > 20 | 5-10% | 40-60% |
Important NIHSS Pearls and Limitations:
Pearl - "The NIHSS is a Score, Not a Diagnosis": Use NIHSS to quantify severity and track change, but always correlate with clinical context. A patient with isolated severe aphasia (NIHSS 3-4) may be devastatingly disabled despite a "minor" score.
Limitation - Anterior Circulation Bias: The NIHSS is heavily weighted toward cortical (MCA) strokes. Motor and language items account for 23 of 42 points. Posterior circulation strokes (vertigo, ataxia, diplopia, isolated hemianopia) often score less than 5 despite significant disability.
Limitation - Right Hemisphere Underestimation: Left MCA strokes score higher due to aphasia and language testing (items 9-10). Right MCA strokes may have dense hemiplegia and neglect but score lower.
Limitation - Lacunar Strokes: Pure motor or pure sensory strokes may score 1-4 despite complete hemiplegia (if only motor item affected).
Pitfall - Dynamic Scoring: NIHSS can change rapidly with early recanalization (improving) or hemorrhagic transformation (worsening). Always re-score at 2 hours, 24 hours, and 7 days.
NIHSS and Treatment Thresholds:
- Thrombolysis: No absolute NIHSS cutoff, but typically withheld for NIHSS 0-1 unless disabling deficit
- Thrombectomy: Typically performed for NIHSS ≥6 with LVO (but can consider for lower scores if disabling)
- DAWN criteria: Requires NIHSS ≥10 (or ≥20 depending on age and core volume)
- DEFUSE-3: No strict NIHSS requirement, but enrolled NIHSS ≥6
Validated Modifications:
- Pediatric NIHSS (PedNIHSS): Adapted for children
- Modified NIHSS: Simplified versions exist but not widely used
- NIHSS-8: Excludes items 1a-c, 7, 9, 10, 11 for telemedicine
4.7 Red Flags and Emergencies
[!CAUTION] RED FLAGS — Seek immediate help if:
- Thunderclap headache: "Worst headache of my life" — suggests SAH
- Rapidly declining GCS: Suggests brainstem compression or massive hemorrhage
- Cushing's triad: Bradycardia + Hypertension + Irregular respirations = high ICP
- New onset seizure: Cortical irritation from hemorrhage or CVST
- Neck pain + Horner's syndrome: Carotid or vertebral artery dissection
- Bilateral weakness or bulbar signs: Basilar artery occlusion
- Symptoms progressing while under observation: Consider hemorrhagic transformation or edema
SECTION 5: Investigations
5.1 Immediate Bedside Tests
| Test | Purpose | Target/Normal | Urgency |
|---|---|---|---|
| Capillary blood glucose | Exclude hypoglycemia mimic | > 60 mg/dL (> 3.3 mmol/L) | STAT |
| SpO₂ | Prevent hypoxic injury | > 94% | Immediate |
| Blood pressure | Identify hypertensive emergency; pre-thrombolysis target | less than 185/110 if thrombolysis planned | Immediate |
| ECG | Detect atrial fibrillation, acute MI | Sinus rhythm | STAT |
| Temperature | Fever worsens outcomes | Normothermia | Immediate |
| GCS/NIHSS | Severity assessment | Document baseline | Immediate |
5.2 Laboratory Investigations
| Test | Purpose | Target/Finding | Turnaround |
|---|---|---|---|
| FBC | Platelets for thrombolysis eligibility | > 100,000/μL | 30 min |
| PT/INR | Coagulation status | INR less than 1.7 for thrombolysis | 30 min |
| APTT | If on heparin | less than 40 seconds | 30 min |
| Glucose | Hypoglycemia mimics stroke | 60-180 mg/dL | 30 min |
| Creatinine | Baseline renal function for contrast | Baseline | 45 min |
| Troponin | Concomitant ACS | Elevated in 15% of strokes | 60 min |
| HbA1c | Identify undiagnosed diabetes | less than 6.5% normal | 24 hours |
| Lipid panel | Secondary prevention baseline | LDL target less than 70 mg/dL | 24 hours |
| Toxicology | Cocaine/amphetamines in young patients | Negative | 4 hours |
| Pregnancy test | Contraindication assessment | Negative | 30 min |
Note: DO NOT delay thrombolysis for laboratory results other than glucose and platelet count (unless clinical suspicion of coagulopathy).
5.3 Neuroimaging
Non-Contrast CT (NCCT) — The First Essential Test:
| Finding | Significance | Timeframe |
|---|---|---|
| Hyperdense vessel sign | Clot in artery (MCA most common) | Immediate |
| Loss of grey-white differentiation | Early ischemic change | 1-3 hours |
| Sulcal effacement | Early edema | 2-6 hours |
| Hypodense region | Established infarct | 6-24 hours |
| Hyperdensity (white) | Hemorrhage | Immediate |
CT Angiography (CTA):
- Purpose: Identify large vessel occlusion (LVO)
- Sites: Internal carotid artery, M1/M2 MCA, basilar artery
- Critical for thrombectomy eligibility
CT Perfusion (CTP):
- Purpose: Identify penumbra for extended window treatment
- Core: CBF less than 30% of normal (irreversible)
- Penumbra: Tmax > 6 seconds (salvageable)
- Mismatch ratio: Target ratio > 1.8 and mismatch volume > 15 mL
MRI Brain:
| Sequence | Finding | Timeframe |
|---|---|---|
| DWI (Diffusion-weighted imaging) | Bright signal = restricted diffusion = acute ischemia | Minutes after onset |
| ADC map | Dark signal confirms true restriction | Minutes |
| FLAIR | Bright signal = established infarct (> 4.5 hours) | 4-6 hours |
| DWI-FLAIR mismatch | DWI+, FLAIR- suggests less than 4.5 hours | Used for wake-up stroke |
| MRA | Vessel occlusion or stenosis | Immediate |
| SWI/GRE | Microbleeds, hemorrhage | Immediate |
Imaging Protocol for Acute Stroke:
┌─────────────────────────────────────────────────────────────────┐
│ ACUTE STROKE IMAGING ALGORITHM │
└─────────────────────────────────────────────────────────────────┘
│
▼
┌──────────────────────────────┐
│ NON-CONTRAST CT HEAD │
│ (Target less than 20 min from │
│ arrival) │
└──────────────────────────────┘
│
┌────────────────────┴────────────────────┐
▼ ▼
┌─────────────────┐ ┌─────────────────┐
│ HEMORRHAGE │ │ NO HEMORRHAGE │
│ (Hyperdense) │ │ (Consider │
│ │ │ ischemia) │
└────────┬────────┘ └────────┬────────┘
│ │
▼ ▼
┌─────────────────┐ ┌────────────────────────────┐
│ ICH Protocol │ │ CT ANGIOGRAPHY (CTA) │
│ - BP control │ │ Identify Large Vessel │
│ - Reversal │ │ Occlusion (LVO) │
│ - Neurosurgery │ └────────────────────────────┘
└─────────────────┘ │
┌─────────────┴─────────────┐
▼ ▼
┌─────────────────┐ ┌─────────────────┐
│ LVO DETECTED │ │ NO LVO │
│ (ICA, M1, M2, │ │ │
│ Basilar) │ │ │
└────────┬────────┘ └────────┬────────┘
│ │
▼ ▼
┌──────────────────────────┐ ┌──────────────────┐
│ Consider Thrombectomy │ │ IV Thrombolysis │
│ ± CT Perfusion if │ │ if less than 4.5h │
│ Extended Window (6-24h) │ │ and eligible │
└──────────────────────────┘ └──────────────────┘
5.4 Cardiac Investigations
| Test | Indication | Findings |
|---|---|---|
| 12-lead ECG | All stroke patients | AF (5-10%), acute MI, LVH |
| Continuous telemetry | All stroke patients for ≥24-72 hours | Paroxysmal AF |
| Transthoracic Echo (TTE) | Suspected cardioembolic source | LV thrombus, valve disease, PFO |
| Transesophageal Echo (TEE) | High suspicion for cardiac source; young patient | LAA thrombus, aortic arch plaque, PFO |
| Prolonged cardiac monitoring | Cryptogenic stroke | Holter 24-48h; implantable loop recorder |
5.5 Vascular Imaging
| Modality | Purpose | Sensitivity/Specificity |
|---|---|---|
| Carotid Doppler ultrasound | Screen for carotid stenosis | 85%/90% for > 70% stenosis |
| CTA head and neck | Define vessel anatomy, stenosis, occlusion | 98%/95% |
| MRA head and neck | Non-contrast option | 90%/92% |
| Digital subtraction angiography (DSA) | Gold standard; invasive | 100% (by definition) |
SECTION 6: Management
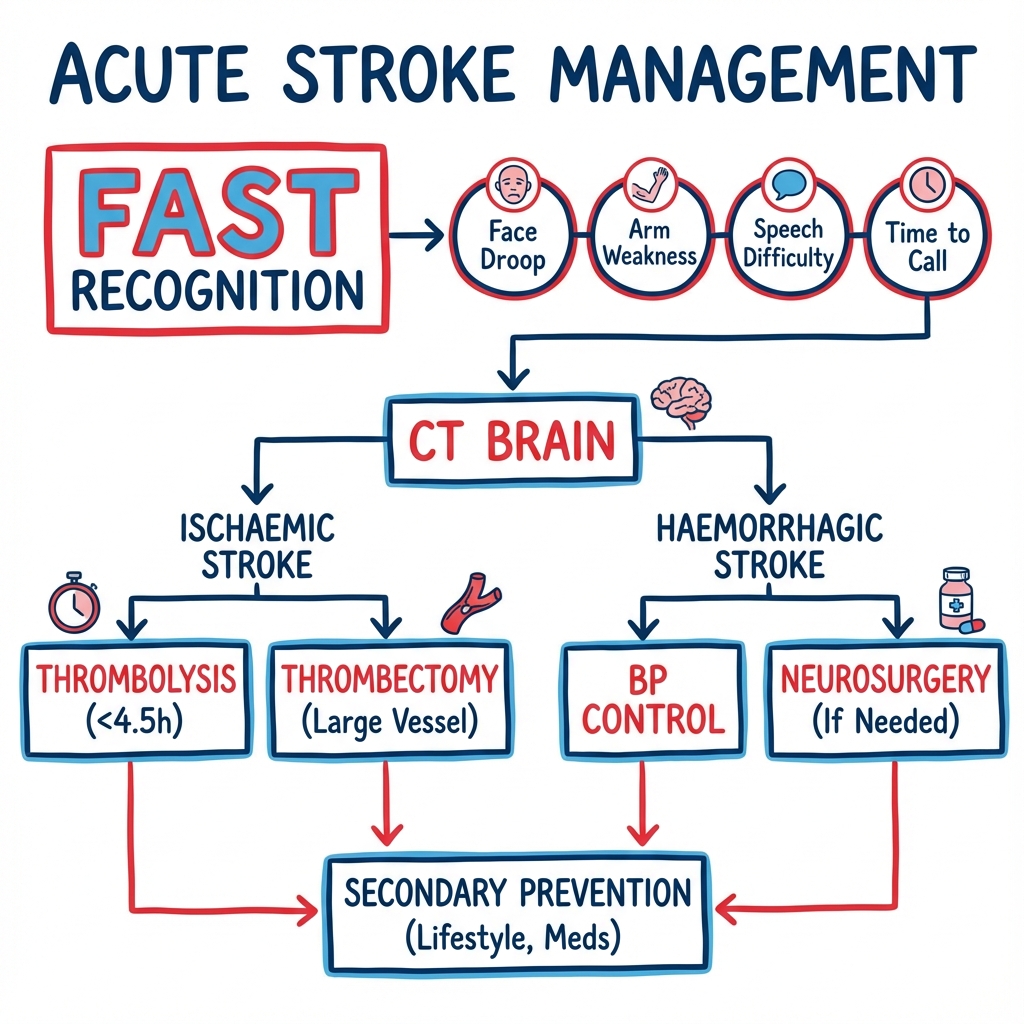
6.1 Acute Stroke Algorithm
┌─────────────────────────────────────────────────────────────────┐
│ ACUTE STROKE MANAGEMENT ALGORITHM │
└─────────────────────────────────────────────────────────────────┘
│
▼
┌───────────────────────────────┐
│ EMERGENCY TRIAGE (T=0) │
│ • ABCs, O₂ > 94%, Glucose │
│ • STAT Neurology Consult │
│ • Last Known Well (LKW) time │
│ • Activate Stroke Protocol │
└───────────────────────────────┘
│
▼
┌───────────────────────────────┐
│ IMMEDIATE IMAGING (Tless than 25m) │
│ • Non-contrast CT (NCCT) │
│ • CT Angiogram (CTA) │
│ • ± CT Perfusion (if > 6h) │
└───────────────────────────────┘
│
┌────────────────────┴────────────────────┐
▼ ▼
┌─────────────────┐ ┌─────────────────┐
│ HEMORRHAGE │ │ ISCHEMIA │
│ (On NCCT) │ │ (No blood) │
└────────┬────────┘ └────────┬────────┘
│ │
▼ ▼
┌─────────────────┐ ┌─────────────────────────────────┐
│ ICH PROTOCOL │ │ REPERFUSION ELIGIBLE? │
│ • SBP less than 140 mmHg │ │ • less than 4.5h for IV Thrombolysis │
│ • Reverse AC │ │ • less than 24h for Thrombectomy (LVO) │
│ • NSGY consult │ │ with imaging selection │
│ • ICU admission │ └────────────────┬────────────────┘
└─────────────────┘ │
┌────────────────┴────────────────┐
▼ ▼
┌─────────────────┐ ┌─────────────────┐
│ ELIGIBLE │ │ NOT ELIGIBLE │
│ │ │ │
│ • TNK or tPA │ │ • Aspirin 300mg │
│ • MT if LVO │ │ • Permissive │
│ • BP less than 185/110 │ │ Hypertension │
└────────┬────────┘ │ • Stroke Unit │
│ └────────┬────────┘
▼ │
┌─────────────────┐ ▼
│ POST-ACUTE │ ┌─────────────────┐
│ • ICU/Stroke U │ │ WORKUP/REHAB │
│ • NPO/Swallow │ │ • Echo/Tele │
│ • Serial Neuro │ │ • Statins │
│ • BP monitoring │ │ • PT/OT/SLP │
└─────────────────┘ └─────────────────┘
6.2 Intravenous Thrombolysis
Intravenous thrombolysis remains the cornerstone of acute ischemic stroke treatment, with a number needed to treat (NNT) of 10 for excellent functional outcome (mRS 0-1) at 3 months when administered within 3 hours, and NNT of 14 when given 3-4.5 hours. [2,13]
Mechanism of Action: Tissue plasminogen activator (tPA) converts plasminogen to plasmin, which enzymatically degrades fibrin in thrombi, thereby recanalization occluded vessels and restoring cerebral perfusion.
Drug Options and Evidence:
| Agent | Dose | Administration | Half-Life | Evidence | Advantages |
|---|---|---|---|---|---|
| Tenecteplase (TNK) | 0.25 mg/kg IV (max 25 mg) | Single bolus over 5-10 seconds | 20-24 minutes | AcT, EXTEND-IA TNK, TASTE: Non-inferior to alteplase [12,27] | Single bolus (faster), weight-based dosing, easier in pre-hospital, higher recanalization rates |
| Alteplase (tPA, rtPA) | 0.9 mg/kg IV (max 90 mg) | 10% bolus, then 90% infusion over 60 min | 4-6 minutes | NINDS, ECASS-III, IST-3: Gold standard since 1995 [13,21,22] | Most studied agent, widely available, proven efficacy |
Current Trend: Tenecteplase is increasingly preferred due to ease of administration and non-inferior efficacy. Some centers have completely transitioned to TNK as first-line agent.
6.2.1 Inclusion Criteria for Thrombolysis
Core Criteria (All Must Be Met):
- Clinical diagnosis of acute ischemic stroke causing measurable neurological deficit
- Onset time ≤4.5 hours from last known well (or wake-up stroke with DWI-FLAIR mismatch)
- Age: No upper age limit (benefit extends to > 80 years per IST-3)
- Imaging: Non-contrast CT or MRI excluding hemorrhage and mimics
- No absolute contraindications (see below)
Extended Window Indications (4.5-9 hours):
- Wake-up stroke with MRI showing DWI-positive, FLAIR-negative mismatch (WAKE-UP trial) [10]
- Unknown onset time with perfusion imaging showing salvageable penumbra
- Clinical improvement: If patient improving rapidly, may still benefit from lysis to prevent re-occlusion
6.2.2 Absolute Contraindications
Strict Contraindications (DO NOT LYSE):
| Category | Contraindication | Rationale |
|---|---|---|
| Imaging | Hemorrhage on CT or MRI | Would worsen bleeding |
| Imaging | Extensive early ischemic changes (> 1/3 MCA territory on CT, or ASPECTS less than 6) | High hemorrhagic transformation risk |
| Timing | Symptom onset > 4.5 hours without advanced imaging selection | Outside evidence-based window |
| Hemorrhage Risk | Active internal bleeding (GI, GU, retroperitoneal) | Systemic bleeding risk |
| Recent Bleeding | Intracranial hemorrhage ever (previous ICH, SAH, SDH) | Extremely high rebleed risk |
| Recent Surgery | Intracranial or spinal surgery within 3 months | Hemorrhage risk |
| Recent Trauma | Head trauma with loss of consciousness or skull fracture less than 3 months | Hemorrhage risk |
| Vascular | Intracranial arteriovenous malformation (AVM) | High rupture risk |
| Vascular | Intracranial aneurysm (unclipped/uncoiled) | High rupture risk |
| Vascular | Aortic arch dissection | Systemic dissection risk |
| Neoplasm | Known intracranial neoplasm | Hemorrhage into tumor |
| Blood Pressure | SBP > 185 mmHg or DBP > 110 mmHg despite treatment | Cannot be controlled pre-lysis |
| Coagulation | Platelet count less than 100,000/μL | Bleeding risk |
| Coagulation | INR > 1.7 or PT > 15 seconds | Warfarin effect |
| Coagulation | APTT > 40 seconds | Heparin effect |
| Coagulation | Therapeutic LMWH within 24 hours | Anti-Xa activity |
| Anticoagulation | Direct thrombin inhibitor or Factor Xa inhibitor use within 48 hours (unless normal assays) | Dabigatran: check thrombin time; Xa inhibitors: check anti-Xa level |
| Glucose | Blood glucose less than 50 mg/dL (2.8 mmol/L) | Hypoglycemia mimics stroke |
| Pregnancy | Pregnancy (relative - see below) | Bleeding risk, but may be outweighed by benefit |
| Clinical | Suspected aortic dissection | May propagate dissection |
| Clinical | Suspected infective endocarditis | Mycotic aneurysm rupture risk |
6.2.3 Relative Contraindications (Individualize Risk-Benefit)
Carefully Consider - May Still Lyse:
| Factor | Concern | Approach |
|---|---|---|
| Age > 80 years | Historical concern for bleeding | IST-3 showed benefit persists; age alone NOT a contraindication [22] |
| NIHSS less than 4 ("minor stroke") | Risk may outweigh benefit | Consider if deficit is disabling (aphasia, hemianopia, hand weakness) |
| NIHSS > 25 | Very high hemorrhage risk (~15%) | Still beneficial; discuss with family; consider thrombectomy |
| Rapidly improving symptoms | May spontaneously recanalize | Risk of re-occlusion; consider lysis if fluctuating or persistent deficit |
| Seizure at stroke onset | Todd's paresis vs true stroke | If deficit is clearly vascular territory, proceed |
| Major surgery or trauma less than 14 days | Bleeding at surgical site | Risk depends on site (compressible vs non-compressible) |
| GI or GU bleeding less than 21 days | Recurrent bleeding | If source controlled, may proceed |
| Arterial puncture at non-compressible site less than 7 days | Hematoma | Avoid if subclavian, femoral; neck may be compressible |
| Recent MI (less than 3 months) | Myocardial rupture (historical concern) | NOT a contraindication per modern guidelines [2] |
| Pregnancy | Theoretical fetal/maternal bleeding | Case reports of safe use; absolute indication may override |
| Prior stroke + diabetes | Historical NINDS exclusion | NOT a contraindication; no increased risk shown |
| Blood glucose 50-400 mg/dL | Extremes of glycemia | Correct glucose first; hyperglycemia worsens outcomes |
| Early CT hypodensity 1/3 MCA territory | Hemorrhagic transformation risk | Strong relative contraindication; consider ASPECTS score |
6.2.4 Pre-Thrombolysis Blood Pressure Management
Strict BP Control Required Before Lysis:
| Phase | Target | Management | Agents |
|---|---|---|---|
| Pre-thrombolysis | SBP less than 185 mmHg AND DBP less than 110 mmHg | Mandatory before bolus | Labetalol 10-20 mg IV over 1-2 min, repeat q10-20min (max 300 mg) OR Nicardipine 5 mg/h IV, titrate by 2.5 mg/h q5-15min (max 15 mg/h) |
| During infusion | SBP less than 180 mmHg AND DBP less than 105 mmHg | Monitor q15min × 2h | Continue IV antihypertensives |
| Post-thrombolysis (24h) | SBP less than 180 mmHg AND DBP less than 105 mmHg | Monitor q15min × 2h, then q30min × 6h, then q60min × 16h | Continue IV antihypertensives; transition to PO when stable |
If BP Cannot Be Controlled: DO NOT GIVE THROMBOLYSIS. Elevated BP during/after lysis increases symptomatic ICH risk from ~6% to > 15%.
Agents to AVOID: Sublingual nifedipine (unpredictable precipitous drop), hydralazine (unpredictable response).
6.2.5 Administration Protocol
Step-by-Step Checklist:
- ☑️ Confirm no contraindications: Review checklist
- ☑️ Confirm weight: Actual body weight in kg (use scale if possible)
- ☑️ Calculate dose: 0.9 mg/kg for alteplase (max 90 mg); 0.25 mg/kg for tenecteplase (max 25 mg)
- ☑️ BP control: Achieve SBP less than 185/110 mmHg
- ☑️ Consent: Discuss risks (6-7% symptomatic ICH) and benefits
- ☑️ Stop all antiplatelets/anticoagulants: Hold for 24 hours
- ☑️ Prepare ICU bed: For post-lysis monitoring
Alteplase Administration:
- Draw up 10% of total dose → give as IV bolus over 1 minute
- Draw up remaining 90% → infuse over 60 minutes via pump
- No other medications in same IV line during infusion
Tenecteplase Administration:
- Draw up total dose → give as IV bolus over 5-10 seconds
- Faster, simpler, fewer errors
6.2.6 Post-Thrombolysis Monitoring and Complications
Intensive Monitoring Protocol:
| Time | Action | Rationale |
|---|---|---|
| 0-2 hours | Neuro checks + BP q15min | Detect early deterioration (hemorrhage) |
| 2-8 hours | Neuro checks + BP q30min | ICH risk highest in first 24h |
| 8-24 hours | Neuro checks + BP q1h | Continue surveillance |
| 24 hours | Repeat CT head + Neuro exam | Assess for hemorrhagic transformation before starting antiplatelets |
Signs of Hemorrhagic Transformation:
- Sudden worsening of neurological deficit
- New severe headache
- Nausea/vomiting
- Decline in level of consciousness
- Seizure
Management of sICH (Symptomatic Intracranial Hemorrhage):
- STOP thrombolytic immediately
- Stat CT head (within 5 minutes)
- Type and cross for blood products
- Labs: PT, APTT, INR, fibrinogen, CBC
- Reverse fibrinolysis:
- Cryoprecipitate: 10 units IV (replaces fibrinogen; aim fibrinogen > 150 mg/dL)
- Tranexamic acid: 1000 mg IV over 10 min (antifibrinolytic; controversial)
- Platelets: 1-2 units if platelet count low or dysfunction
- Neurosurgery consult: For possible hematoma evacuation
- BP control: Target SBP less than 140-160 mmHg
Symptomatic ICH Rate: 6-7% with tPA (NINDS trial); slightly lower with TNK in some studies.
6.2.7 Outcomes of Thrombolysis
Efficacy (Pooled Meta-Analysis Data):
| Outcome | Placebo | Thrombolysis | NNT | ARR |
|---|---|---|---|---|
| Excellent outcome (mRS 0-1) at 3 months | 26% | 36% | 10 | 10% |
| Functional independence (mRS 0-2) at 3 months | 42% | 53% | 9 | 11% |
| Symptomatic ICH | 1% | 6-7% | -20 (NNH) | 5-6% |
| Mortality at 3 months | 19% | 17% | 50 | 2% |
Time-Dependency of Benefit:
| Time Window | NNT for mRS 0-1 | Absolute Benefit |
|---|---|---|
| 0-90 minutes | 4 | 25% |
| 90-180 minutes | 9 | 11% |
| 180-270 minutes | 14 | 7% |
| 270-360 minutes | 20 | 5% |
Every 15 minutes of delay = 4% lower odds of excellent outcome and 1 month of disability-free life lost. [28]
6.3 Mechanical Thrombectomy
Mechanical thrombectomy represents the most effective intervention in acute medicine, with a number needed to treat of 2.6 for one-grade improvement in mRS and NNT of 7 for functional independence (mRS 0-2). [6] The HERMES meta-analysis of 5 randomized trials demonstrated thrombectomy's superiority over medical therapy alone, establishing it as standard of care for large vessel occlusion.
Mechanism: Endovascular retrieval of thrombus using stent retrievers (Solitaire, Trevo) or direct aspiration catheters (Penumbra), achieving TICI 2b/3 reperfusion (≥50% or complete reperfusion) in 70-90% of cases.
6.3.1 Indications for Mechanical Thrombectomy
Core Inclusion Criteria (0-6 Hour Window):
-
Large Vessel Occlusion (LVO) on CTA/MRA:
- Internal carotid artery (ICA) — terminus or proximal
- M1 segment of middle cerebral artery (MCA)
- Proximal M2 MCA (select cases)
- Basilar artery (posterior circulation)
- Vertebral artery (select cases)
-
NIHSS ≥6 (evidence threshold from trials)
- Lower NIHSS may qualify if disabling deficit (e.g., dominant hand weakness, aphasia, hemianopia affecting driving)
-
Pre-stroke mRS 0-1 (functionally independent)
- Trials excluded patients with severe baseline disability
- Some flexibility for mRS 2 if patient/family preference
-
ASPECTS ≥6 on non-contrast CT
- Alberta Stroke Program Early CT Score
- Assesses extent of early ischemic change in MCA territory
- 10 regions scored; 1 point deducted for each region with hypodensity
- ASPECTS 6-10: Good collaterals, small core, favorable for thrombectomy
- ASPECTS 0-5: Large established infarct, high complication risk
-
Age: No strict upper limit, but trials enrolled mostly less than 85 years
-
Time from onset less than 6 hours from last known well
Basilar Artery Occlusion:
- More permissive criteria due to high mortality without intervention
- Consider up to 24 hours even without perfusion imaging
- NIHSS may be misleading (locked-in syndrome has variable NIHSS)
6.3.2 Extended Window Thrombectomy (6-24 Hours)
DAWN Trial Criteria (6-24 Hours): [3]
Enrollment based on clinical-core mismatch (DWI or CTP core volume):
| Age Group | NIHSS | Core Volume (mL) | Rationale |
|---|---|---|---|
| Age ≥80 | ≥10 | less than 21 mL | Small core despite moderate deficit → salvageable penumbra |
| Age less than 80 | ≥10 | less than 31 mL | Small core despite moderate deficit → salvageable penumbra |
| Age less than 80 | ≥20 | less than 51 mL | Severe deficit with moderate core → still salvageable tissue |
Additional DAWN Criteria:
- LVO: ICA or M1 MCA occlusion
- Pre-stroke mRS 0-1
- Last known well 6-24 hours prior
DAWN Results:
- 49% achieved mRS 0-2 (thrombectomy) vs 13% (control) — NNT = 2.8
- 90-day mortality: 19% vs 38%
DEFUSE-3 Trial Criteria (6-16 Hours): [4]
Enrollment based on perfusion mismatch (penumbra vs core ratio):
| Parameter | Threshold | Definition |
|---|---|---|
| Core volume | less than 70 mL | Ischemic core on CTP (CBF less than 30%) or DWI |
| Mismatch ratio | ≥1.8 | Tmax > 6s volume / Core volume ≥ 1.8 |
| Mismatch volume | ≥15 mL | Absolute difference between Tmax > 6s and core ≥15 mL |
Additional DEFUSE-3 Criteria:
- LVO: ICA or M1/M2 MCA occlusion
- Pre-stroke mRS 0-2
- NIHSS ≥6
- Age 18-90 years
- Last known well 6-16 hours prior
DEFUSE-3 Results:
- 45% achieved mRS 0-2 (thrombectomy) vs 17% (control) — NNT = 3.6
- Earlier reperfusion = better outcomes (each hour delay = 10% lower odds of good outcome)
6.3.3 Imaging Selection for Thrombectomy
CT Perfusion (CTP) Parameters:
| Map | Measurement | Threshold | Interpretation |
|---|---|---|---|
| CBF (Cerebral Blood Flow) | Relative CBF less than 30% of contralateral | Defines core | Irreversible injury |
| Tmax (Time to Maximum) | Tmax > 6 seconds | Defines penumbra | Tissue at risk but salvageable |
| CBV (Cerebral Blood Volume) | Reduced CBV | Supports core definition | Collateral failure |
| MTT (Mean Transit Time) | Prolonged MTT | Oligemia or penumbra | May recover spontaneously |
ASPECTS (Alberta Stroke Program Early CT Score):
10-point scale assessing MCA territory on non-contrast CT:
Supraganglionic Level (4 regions):
- M1: Anterior MCA cortex
- M2: MCA cortex lateral to insular ribbon
- M3: Posterior MCA cortex
- Insula
Ganglionic Level (6 regions):
- C: Caudate
- L: Lentiform nucleus (putamen)
- IC: Internal capsule
- M4: Anterior MCA immediately superior to M1
- M5: Lateral MCA territory superior to M2
- M6: Posterior MCA territory superior to M3
Scoring: Start at 10; subtract 1 for each region with hypodensity, loss of gray-white differentiation, or swelling.
| ASPECTS | Interpretation | Thrombectomy Recommendation |
|---|---|---|
| 8-10 | Minimal early ischemic change | Strong indication |
| 6-7 | Moderate early change | Reasonable indication |
| 0-5 | Extensive early change | Relative contraindication (high sICH risk) |
6.3.4 Thrombectomy Technique
Procedural Steps:
- Vascular Access: Femoral artery (most common) or radial artery
- Guide Catheter: Advanced to cervical ICA or vertebral artery
- Microcatheter Navigation: Across thrombus under fluoroscopy
- Reperfusion Technique:
- Stent Retriever: Deploy stent across clot, wait 3-5 min for integration, retrieve with aspiration
- Direct Aspiration (ADAPT): Large-bore catheter advanced to clot, direct suction applied
- Combined Technique: Stent retriever + proximal balloon guide catheter aspiration (SAVE technique)
- Assess Reperfusion: Angiography to determine TICI score
TICI (Thrombolysis in Cerebral Infarction) Score:
| Grade | Definition | Clinical Significance |
|---|---|---|
| 0 | No perfusion | Complete occlusion; treatment failed |
| 1 | Minimal perfusion past occlusion, no distal filling | Poor outcome |
| 2a | Partial perfusion less than 50% of territory | Suboptimal reperfusion |
| 2b | Partial perfusion 50-99% of territory | Target outcome; good prognosis |
| 2c | Near-complete perfusion with slow flow | Good outcome |
| 3 | Complete perfusion, normal flow | Optimal outcome |
Target: TICI 2b or 3 (achieved in 70-90% of modern cases)
Number of Passes: Minimize to reduce endothelial injury; most experts aim for ≤3 passes
6.3.5 Anesthesia for Thrombectomy
General Anesthesia (GA) vs Conscious Sedation (CS):
| Approach | Advantages | Disadvantages |
|---|---|---|
| Conscious Sedation | Faster workflow (no intubation), lower BP drops, ongoing neuro assessment, lower cost | Patient movement, airway compromise risk, aspiration risk |
| General Anesthesia | Immobile patient, secure airway, better imaging quality | Delays (intubation), BP instability, loss of neuro exam |
Current Evidence: Multiple RCTs (SIESTA, ANSTROKE, GOLIATH) show no difference in outcomes. Recent meta-analyses suggest possible benefit with CS due to faster times. [29]
Recommendation: Use conscious sedation unless:
- Patient agitated or uncooperative
- GCS less than 8 or loss of airway reflexes
- Posterior circulation stroke with risk of herniation
- Conversion to GA if aspiration occurs
6.3.6 Complications of Thrombectomy
| Complication | Incidence | Presentation | Management |
|---|---|---|---|
| Symptomatic ICH | 5-7% | Sudden neuro worsening | Stop procedure, BP control, neurosurgery |
| Vessel perforation | 1-2% | Extravasation of contrast | Reverse heparin, coil embolization |
| Arterial dissection | 1-2% | Flow limitation, occlusion | Stenting if flow-limiting |
| Vasospasm | 5-10% | Slowed flow, reduced perfusion | IA verapamil or nicardipine |
| Embolization to new territory | 5-8% | New deficit in different territory | Attempt retrieval if accessible |
| Groin hematoma | 3-5% | Groin swelling, Hb drop | Usually self-limited; vascular surgery if expanding |
Overall Safety: Serious adverse events in less than 10% of cases; benefit far outweighs risk.
6.3.7 Thrombectomy + Thrombolysis (Bridging Therapy)
Should We Give tPA Before Thrombectomy?
Yes (Bridging) - Standard practice in most stroke systems:
- May recanalize small vessel occlusions before thrombectomy
- No delay if systems run in parallel (tPA bolus → immediate transfer to angio)
- HERMES trials used bridging in majority
No (Direct Thrombectomy) - Some emerging evidence:
- DIRECT-MT, DEVT, SKIP trials: Direct thrombectomy non-inferior to bridging
- Avoids tPA-related sICH risk
- May be preferred if tPA contraindications (e.g., recent surgery)
Current Recommendation: Bridging therapy remains standard unless contraindications to tPA exist. Some centers practicing direct thrombectomy for select patients.
6.3.8 Posterior Circulation Thrombectomy
Basilar Artery Occlusion (BAO):
- High mortality without treatment (70-90%)
- More permissive time windows (up to 24h) due to devastating natural history
- Lower quality evidence (BEST, BASICS trials ongoing)
- Consider even with ASPECTS not applicable (use pc-ASPECTS for posterior circulation)
Indications:
- Basilar artery occlusion on CTA/MRA
- NIHSS variable (locked-in may have low NIHSS)
- Symptoms less than 24 hours (some centers extend further)
- No massive infarction on imaging
Outcomes: TICI 2b/3 reperfusion associated with 40-50% mRS 0-3 vs less than 10% without reperfusion.
6.3.9 Thrombectomy Outcomes
Efficacy Data (HERMES Meta-Analysis): [6]
| Outcome | Thrombectomy | Control | NNT | ARR |
|---|---|---|---|---|
| mRS 0-2 (functional independence) | 46% | 27% | 5.3 | 19% |
| 1-grade shift in mRS (any improvement) | — | — | 2.6 | 38% |
| Symptomatic ICH | 4% | 3% | -100 (NNH) | 1% |
| 90-day mortality | 15% | 19% | 25 | 4% |
Factors Associated with Better Outcome:
- TICI 2b/3 reperfusion achieved
- Shorter time to reperfusion (less than 6 hours best)
- Smaller baseline core volume
- Good collateral circulation
- Younger age
- Lower NIHSS at presentation
6.4 Intracerebral Hemorrhage Management
Blood Pressure Control:
| Guideline | Target SBP | Evidence |
|---|---|---|
| AHA/ASA 2022 | less than 140 mmHg (if presenting SBP 150-220) | INTERACT2, ATACH-2 showed safety; efficacy uncertain [11,14] |
| If presenting SBP > 220 | Aggressive reduction with continuous IV infusion | Limited evidence |
Reversal of Anticoagulation:
| Agent | Reversal | Dose |
|---|---|---|
| Warfarin | 4-factor PCC + Vitamin K | PCC 25-50 units/kg; Vitamin K 10 mg IV |
| Dabigatran | Idarucizumab | 5 g IV |
| Rivaroxaban/Apixaban | Andexanet alfa or 4-factor PCC | Andexanet: bolus + infusion; PCC 50 units/kg |
| Heparin | Protamine sulfate | 1 mg per 100 units heparin |
Surgical Intervention:
- Cerebellar hemorrhage > 3 cm with hydrocephalus or brainstem compression → Surgical evacuation
- Lobar hemorrhage with deterioration → Consider evacuation (STICH trials showed limited benefit for supratentorial ICH)
- IVH with hydrocephalus → External ventricular drain (EVD)
6.5 Blood Pressure Management in Acute Stroke
Blood pressure management in acute stroke is complex and context-dependent. Elevated BP is common (70-80% of patients) due to physiologic stress response, loss of cerebral autoregulation, and pre-existing hypertension. Premature or excessive lowering can extend infarct by collapsing penumbral perfusion; however, severe hypertension increases hemorrhagic transformation and cerebral edema risk.
6.5.1 Permissive Hypertension in Acute Ischemic Stroke (No Reperfusion Therapy)
Rationale: Elevated BP maintains collateral flow to ischemic penumbra. Cerebral autoregulation is disrupted in acute stroke, making perfusion pressure-dependent. [9]
Guidelines (AHA/ASA 2019): [2]
| BP Level | Action | Rationale |
|---|---|---|
| less than 220/120 mmHg | Permissive hypertension: DO NOT lower BP | Maintain penumbral perfusion via collaterals |
| ≥220/120 mmHg | Cautiously lower by 15% in first 24 hours | Balance hemorrhagic transformation risk vs perfusion |
| SBP > 220 mmHg with signs of end-organ damage | Lower BP to 180-200 mmHg cautiously | Prevent hypertensive complications (AKI, MI, pulmonary edema) |
Agents for Cautious Lowering (if SBP > 220 or DBP > 120):
| Agent | Dose | Onset | Duration | Advantages | Disadvantages |
|---|---|---|---|---|---|
| Labetalol | 10-20 mg IV over 1-2 min q10-20min (max 300 mg) | 5-10 min | 3-6 hours | Predictable, safe, dual alpha/beta blockade | Contraindicated in asthma, heart failure |
| Nicardipine | 5 mg/h IV infusion, titrate 2.5 mg/h q5-15min (max 15 mg/h) | 5-10 min | 30-40 min | Titratable, smooth reduction, cerebral vasodilation | Expensive, requires pump |
| Esmolol | 500 mcg/kg bolus, then 50-300 mcg/kg/min infusion | 1-2 min | 10-20 min | Ultra-short acting, easily titratable | Requires ICU monitoring |
| Hydralazine | 10-20 mg IV q4-6h PRN | 10-20 min | 3-8 hours | Inexpensive | AVOID: Unpredictable precipitous drops |
Avoid: Sublingual nifedipine (precipitous, uncontrolled drop → ischemia extension)
Pearl: Lower BP gradually. Aggressive lowering can precipitate stroke extension or new infarction in watershed zones.
6.5.2 BP Management for Thrombolysis Candidates
Strict Targets (MANDATORY):
| Phase | SBP Target | DBP Target | Monitoring Frequency |
|---|---|---|---|
| Pre-thrombolysis | less than 185 mmHg | less than 110 mmHg | Q5min until controlled, then q15min |
| During infusion (0-60 min) | less than 180 mmHg | less than 105 mmHg | Q15min continuously |
| Post-lysis (0-2 hours) | less than 180 mmHg | less than 105 mmHg | Q15min |
| Post-lysis (2-8 hours) | less than 180 mmHg | less than 105 mmHg | Q30min |
| Post-lysis (8-24 hours) | less than 180 mmHg | less than 105 mmHg | Q1h |
Critical Rule: If BP cannot be controlled to less than 185/110 mmHg, DO NOT give thrombolysis. Risk of symptomatic ICH increases from 6% to > 15% with uncontrolled hypertension.
Management Algorithm:
Pre-Thrombolysis BP > 185/110 mmHg
↓
Labetalol 10-20 mg IV push
↓
Recheck BP in 10 minutes
↓
┌──────────────────┐
↓ ↓
Still > 185/110 less than 185/110
↓ ↓
Repeat labetalol Proceed with lysis
or start Monitor q15min
nicardipine
infusion
↓
If still uncontrolled
after 300 mg labetalol
→ DEFER thrombolysis
Post-Thrombolysis BP Elevation:
- If SBP > 180 or DBP > 105 mmHg: Start IV nicardipine or labetalol
- If severe elevation (SBP > 230): Emergent CT to rule out hemorrhage, then aggressive IV therapy
6.5.3 BP Management in Intracerebral Hemorrhage (ICH)
Aggressive BP Lowering in ICH: Unlike ischemic stroke, ICH benefits from early intensive BP reduction to prevent hematoma expansion.
Evidence:
- INTERACT-2 (2013): SBP less than 140 mmHg safe but no significant functional benefit (primary outcome neutral) [9]
- ATACH-2 (2016): SBP 110-139 mmHg vs 140-179 mmHg → No benefit, possible harm in renal function [14]
AHA/ASA Guidelines for ICH (2015, updated 2022): [11]
| Presenting SBP | Target SBP | Timeframe | Rationale |
|---|---|---|---|
| 150-220 mmHg | less than 140 mmHg | Acutely, within 1-2 hours | Safe; may reduce hematoma expansion |
| > 220 mmHg | 140-160 mmHg | Continuous IV infusion | Very high BP worsens outcomes |
Contraindication to Aggressive Lowering:
- Large ICH with significant mass effect (CPP = MAP - ICP; lowering MAP may worsen CPP)
- Suspicion of elevated ICP (consider ICP monitoring)
Agents for ICH BP Control:
| Agent | Dose | Characteristics |
|---|---|---|
| Nicardipine | 5-15 mg/h IV infusion | First-line; smooth, titratable |
| Labetalol | 10-80 mg IV bolus q10min | Bolus therapy; predictable |
| Clevidipine | 1-2 mg/h IV, titrate q2-5min (max 21 mg/h) | Ultra-short acting calcium channel blocker |
| Enalaprilat | 0.625-1.25 mg IV q6h | ACE inhibitor; slower onset |
Monitoring: Continuous arterial line recommended for ICH with severe hypertension (SBP > 200) to allow real-time titration.
6.5.4 BP Management Post-Acute Phase (Days 2-7)
Ischemic Stroke: After the hyperacute phase (first 24-48h), begin gradual transition to long-term BP control.
Targets:
- Days 2-7: Aim SBP less than 140-160 mmHg (avoid rapid drops)
- After 7 days: Initiate guideline-directed long-term therapy (target less than 130/80)
Transition from IV to PO:
- If patient able to swallow and neurologically stable, transition to oral agents
- Common regimens: Amlodipine 5-10 mg daily, Lisinopril 10-20 mg daily, Losartan 50 mg daily
6.5.5 Special Considerations
Wake-Up Stroke: Permissive hypertension unless receiving thrombolysis/thrombectomy
Posterior Circulation Stroke: May tolerate lower BP better (brainstem has better autoregulation)
Lacunar Stroke: Often associated with chronic hypertension; permissive hypertension acutely, then aggressive long-term control
Stroke with Concurrent MI: Balanced approach; avoid extremes; cardiology co-management
Dissection: BP control to SBP less than 140 to prevent propagation (similar to aortic dissection principles)
6.6 General Acute Stroke Care
Beyond reperfusion therapy, meticulous supportive care in the first 24-72 hours significantly impacts outcomes. Stroke unit care reduces mortality and disability by 20-30% compared to general ward care, driven by protocol-based management, specialized nursing, and early rehabilitation. [8]
| Parameter | Target | Rationale | Monitoring |
|---|---|---|---|
| Airway | Intubation if GCS less than 8 or loss of protective reflexes | Prevent aspiration pneumonia (10-15% risk) | Continuous pulse oximetry |
| Oxygenation | SpO₂ > 94%; supplemental O₂ if less than 94% | Hypoxia worsens ischemic injury; avoid hyperoxia (free radical damage) | Continuous |
| Blood pressure (ischemic, no lysis) | Permissive hypertension ≤220/120 mmHg | Maintain collateral penumbral perfusion | Q1-2h × 24h, then Q4h |
| Temperature | Normothermia 36.5-37.5°C; treat fever > 37.5°C | Each 1°C increase worsens outcomes by ~30%; fever doubles mortality | Q4h; continuous if febrile |
| Glucose | 140-180 mg/dL (7.8-10 mmol/L) | Hyperglycemia worsens outcomes; hypoglycemia mimics stroke | Q4-6h or continuous CGM if on insulin |
| Positioning | Head of bed 0-30° | Flat positioning improves cerebral perfusion; elevate only if aspiration risk or dyspnea | As tolerated |
| Swallow Assessment | Formal dysphagia screen before PO intake | 37-78% of stroke patients have dysphagia; aspiration pneumonia in 10% | Before any PO; repeat if worsening |
| Nutrition | Enteral feeding within 48-72h if dysphagia | Malnutrition worsens outcomes; NG tube if prolonged NPO (> 48h) | Daily calorie count |
| DVT Prophylaxis | IPC immediately; LMWH 40 mg SC after 24-48h (if no hemorrhage) | DVT risk 10-20% without prophylaxis; PE 1-5% | Daily leg checks |
| Bladder Care | Avoid indwelling catheter if possible; intermittent catheterization preferred | Catheter-associated UTI increases LOS and mortality | Bladder scan; I/O monitoring |
| Mobilization | Early mobilization within 24-48h | Reduces DVT, pneumonia, and length of stay | PT/OT consult within 24h |
| Stroke Unit Admission | All stroke patients | 20-30% reduction in death and disability | Specialized nursing protocols |
Fever Management:
- Source control: Urinalysis, CXR, blood cultures
- Acetaminophen 650-1000 mg q6h PRN
- Cooling blankets if persistent fever > 38.5°C
- Avoid NSAIDs acutely (may interfere with antiplatelet effects)
Glucose Management:
- Insulin sliding scale if glucose > 180 mg/dL
- Avoid hypoglycemia (less than 70 mg/dL) — associated with worse outcomes
- Target HbA1c less than 7% long-term
6.7 Medical Management (First-Line Medications)
| Drug | Class | Dose | Indication | Key Considerations |
|---|---|---|---|---|
| Tenecteplase | Thrombolytic | 0.25 mg/kg IV bolus (max 25 mg) | Acute ischemic stroke less than 4.5h | Single bolus; easier than alteplase |
| Alteplase | Thrombolytic | 0.9 mg/kg (max 90 mg); 10% bolus, 90% over 60 min | Acute ischemic stroke less than 4.5h | Standard agent; requires infusion |
| Aspirin | Antiplatelet | 300 mg loading, then 75-100 mg daily | After hemorrhage excluded; within 24-48h | Give rectally if NPO |
| Clopidogrel | Antiplatelet | 75 mg daily (300 mg load for DAPT) | Minor stroke/TIA with high-risk features | DAPT for 21-90 days per POINT/CHANCE [15,16] |
| Atorvastatin | Statin | 80 mg daily | All ischemic stroke patients | High-intensity for LDL less than 70 mg/dL |
| Labetalol | Beta-blocker | 10-20 mg IV q10-20min | BP control pre/post thrombolysis | Max 300 mg |
| Nicardipine | Calcium channel blocker | 5-15 mg/h IV infusion | BP control | Titratable |
| Enoxaparin | LMWH | 40 mg SC daily | DVT prophylaxis (after 24-48h) | Hold if hemorrhage risk |
SECTION 7: Secondary Prevention
Secondary prevention is critical to reduce the 5-year recurrent stroke risk from 25-30% to 10-15%. The EXPRESS study demonstrated that immediate risk factor modification and treatment initiation within 24 hours reduces recurrent stroke by 80% in the first 90 days. [23] A comprehensive secondary prevention strategy addresses antiplatelet therapy, anticoagulation, statins, blood pressure control, diabetes management, lifestyle modification, and revascularization.
7.1 Antiplatelet Therapy
Antiplatelet therapy is the cornerstone of secondary prevention for non-cardioembolic ischemic stroke and TIA, reducing recurrent stroke risk by 20-25%.
Single Antiplatelet Therapy (Long-Term Maintenance):
| Agent | Dose | Evidence | Indication | Advantages |
|---|---|---|---|---|
| Aspirin | 75-325 mg daily (typically 81-100 mg) | Meta-analysis: 13% RRR for vascular events | First-line for most patients | Inexpensive, widely available, GI prophylaxis with PPI if needed |
| Clopidogrel | 75 mg daily | CAPRIE: 8.7% RRR vs aspirin (slightly superior) | If aspirin intolerant OR aspirin failure | Better GI tolerability than aspirin; preferred in PAD |
| Aspirin/Dipyridamole ER | 25/200 mg BID | ESPRIT, ESPS-2: Superior to aspirin alone | Alternative to clopidogrel | Headache limits tolerability (25% discontinuation) |
Recommendation: Clopidogrel 75 mg daily is preferred for most patients due to superior efficacy and tolerability compared to aspirin alone.
7.1.1 Dual Antiplatelet Therapy (DAPT) for Minor Stroke and High-Risk TIA
Indication: Minor stroke (NIHSS ≤3) or high-risk TIA presenting within 24 hours of symptom onset.
Definition of High-Risk TIA:
- ABCD² score ≥4, OR
- Symptomatic intracranial or extracranial stenosis ≥50%, OR
- Multiple ischemic lesions on DWI MRI
Evidence from Major Trials:
| Trial | Year | N | Regimen | Duration | Primary Outcome | NNT | sICH Risk |
|---|---|---|---|---|---|---|---|
| CHANCE [15] | 2013 | 5170 | ASA 75-300 mg + Clopidogrel 75 mg (300 mg load) | 21 days | 32% RRR for recurrent stroke at 90 days (8.2% vs 11.7%) | 29 | No increase |
| POINT [16] | 2018 | 4881 | ASA 50-325 mg + Clopidogrel 75 mg (600 mg load) | 90 days | 25% RRR for stroke/TIA/CV death (5.0% vs 6.5%) | 67 | Increased bleeding (0.9% vs 0.4%) |
| THALES [17] | 2020 | 11,016 | ASA 75-100 mg + Ticagrelor 180 mg load, then 90 mg BID | 30 days | 17% RRR for stroke/death (5.5% vs 6.6%) | 91 | Increased bleeding |
Interpretation:
- DAPT is most beneficial in the first 21-30 days post-event
- Longer duration (90 days) increases bleeding risk without additional benefit
- Loading dose of clopidogrel (300-600 mg) on day 1 is standard
Current Guideline Recommendation (AHA/ASA 2021): [2]
- Aspirin 75-100 mg + Clopidogrel 75 mg × 21 days, then single antiplatelet
- Start within 24 hours of minor stroke or high-risk TIA
- Avoid if plan for thrombolysis (wait 24h post-lysis)
- Avoid if plan for anticoagulation
DAPT Protocol:
Day 0 (Presentation):
- Aspirin 300 mg PO
- Clopidogrel 300 mg PO loading dose
Days 1-21:
- Aspirin 75-100 mg daily
- Clopidogrel 75 mg daily
After Day 21:
- STOP aspirin
- CONTINUE clopidogrel 75 mg daily long-term
Contraindications to DAPT:
- Recent ICH
- Active bleeding
- Severe thrombocytopenia (less than 50,000)
- Planned anticoagulation for AF
- High bleeding risk (HAS-BLED ≥3)
7.2 Anticoagulation for Atrial Fibrillation
Atrial fibrillation (AF) is present in 15-20% of ischemic strokes and confers a 5-fold increased risk of recurrent stroke. Anticoagulation reduces stroke risk by 60-70% compared to no treatment. [30]
CHA₂DS₂-VASc Score (Stroke Risk Stratification):
| Factor | Points |
|---|---|
| Congestive heart failure (or LVEF ≤40%) | 1 |
| Hypertension (≥140/90 or on treatment) | 1 |
| Age ≥75 years | 2 |
| Diabetes mellitus | 1 |
| Stroke/TIA/thromboembolism (prior event) | 2 |
| Vascular disease (MI, PAD, aortic plaque) | 1 |
| Age 65-74 years | 1 |
| Sex category (female) | 1 |
Maximum Score: 9 points
Annual Stroke Risk by CHA₂DS₂-VASc:
| Score | Annual Stroke Risk | Recommendation |
|---|---|---|
| 0 (men) / 1 (women) | 0.2-0.6% | No anticoagulation or aspirin |
| 1 (men) / 2 (women) | 0.6-2.2% | Consider anticoagulation (shared decision-making) |
| ≥2 (men) / ≥3 (women) | ≥2.2% | Anticoagulation strongly recommended |
Note: A history of stroke/TIA automatically qualifies for anticoagulation (score ≥2).
7.2.1 Choice of Anticoagulant
Direct Oral Anticoagulants (DOACs) — Preferred Over Warfarin:
| Agent | Class | Dose | Dose Reductions | Advantages vs Warfarin |
|---|---|---|---|---|
| Apixaban | Factor Xa inhibitor | 5 mg BID | 2.5 mg BID if 2 of: Age ≥80, weight ≤60 kg, Cr ≥1.5 | Lowest bleeding risk; BID dosing |
| Rivaroxaban | Factor Xa inhibitor | 20 mg daily with evening meal | 15 mg daily if CrCl 15-50 | Once-daily dosing; take with food |
| Edoxaban | Factor Xa inhibitor | 60 mg daily | 30 mg daily if CrCl 15-50, weight ≤60 kg, or P-gp inhibitors | Once daily; avoid if CrCl > 95 |
| Dabigatran | Direct thrombin inhibitor | 150 mg BID | 110 mg BID if age ≥80 or high bleeding risk | Reversal agent available (idarucizumab) |
Warfarin:
- Dose: Individualized to achieve INR 2.0-3.0
- Requires frequent monitoring
- Multiple drug/food interactions
- Reserve for: Severe CKD (CrCl less than 15), mechanical valves, rheumatic mitral stenosis
Evidence: RE-LY, ROCKET-AF, ARISTOTLE, ENGAGE AF-TIMI 48 trials all showed DOACs non-inferior or superior to warfarin with lower ICH risk. [30]
7.2.2 Timing of Anticoagulation After Stroke
The "1-3-6-12 Day Rule" (based on stroke size and hemorrhagic transformation risk):
| Stroke Severity (NIHSS) | Infarct Size | Timing to Start Anticoagulation |
|---|---|---|
| Minor (0-3) | TIA or small infarct | 1-3 days after event |
| Moderate (4-15) | Moderate infarct | 3-7 days after event |
| Severe (> 15) | Large infarct | 7-14 days after event (or individualized with repeat imaging) |
Rationale: Early anticoagulation (within 24-48h) in large infarcts increases hemorrhagic transformation risk. Delayed initiation balances recurrent stroke risk vs bleeding risk.
Factors Favoring Early Anticoagulation (consider 1-3 days):
- High thromboembolic risk (prior stroke, LV thrombus, recent MI)
- Mechanical heart valve
- Small infarct size on imaging
- No hemorrhagic transformation on 24h CT
Factors Favoring Delayed Anticoagulation (7-14 days):
- Large infarct (> 50% MCA territory or ASPECTS less than 6)
- Hemorrhagic transformation on imaging
- Uncontrolled hypertension (SBP > 180)
- High fall risk
Bridging Therapy: NOT recommended. Start DOAC directly without heparin bridge (BRIDGE trial showed no benefit and increased bleeding).
7.3 Statin Therapy
High-intensity statin therapy reduces recurrent stroke by 15-20% and all cardiovascular events by 20-30%, regardless of baseline LDL cholesterol. [18]
Recommendation: ALL ischemic stroke patients should receive high-intensity statin unless contraindicated.
High-Intensity Statins:
| Agent | Dose | LDL Reduction | Evidence |
|---|---|---|---|
| Atorvastatin | 40-80 mg daily | 50-60% | SPARCL: 16% RRR for recurrent stroke [18] |
| Rosuvastatin | 20-40 mg daily | 50-60% | Potent; use 20 mg if renal impairment |
LDL Target: less than 70 mg/dL (1.8 mmol/L), or ≥50% reduction from baseline.
2023 Update: AHA/ASA now recommends LDL less than 55 mg/dL for very high-risk patients (recurrent stroke, polyvascular disease).
Monitoring:
- Baseline: Lipid panel, ALT, CK
- Repeat lipid panel at 4-12 weeks to assess response
- ALT if symptoms of hepatotoxicity; discontinue if ALT > 3× ULN
- CK if symptoms of myopathy; discontinue if CK > 10× ULN or rhabdomyolysis
Statin Intolerance (5-10% of patients):
- Symptoms: Myalgias, weakness, elevated CK
- Management:
- Try alternative statin (rosuvastatin better tolerated)
- Lower dose with ezetimibe 10 mg to reach LDL goal
- PCSK9 inhibitors (evolocumab, alirocumab) if severe intolerance
7.4 Blood Pressure Management
Long-term blood pressure control is critical for secondary prevention. Hypertension is the most important modifiable risk factor for recurrent stroke (40% RRR with optimal control). [19]
Target: less than 130/80 mmHg (AHA/ASA 2021 guidelines) [2]
Exception: Bilateral carotid stenosis or vertebral stenosis — consider higher target (SBP 140-150) to maintain perfusion.
Timing: Initiate or resume antihypertensive therapy after the acute phase (48-72 hours), once patient neurologically stable. Avoid abrupt lowering in first 24 hours (risk of extending infarct).
7.4.1 Preferred Agents
First-Line Agents (Evidence-Based):
| Class | Agent | Dose | Evidence | Special Benefits |
|---|---|---|---|---|
| ACE Inhibitor | Lisinopril | 10-40 mg daily | HOPE trial: 32% RRR stroke | Renal protection in diabetes; improves endothelial function |
| ACE Inhibitor + Diuretic | Perindopril 4 mg + Indapamide 1.25 mg | Daily | PROGRESS: 28% RRR stroke [19] | Combination superior to monotherapy |
| ARB | Losartan | 50-100 mg daily | LIFE trial: 25% RRR stroke vs atenolol | If ACE inhibitor not tolerated (cough) |
| CCB | Amlodipine | 5-10 mg daily | Multiple trials | Good add-on therapy; no CNS side effects |
| Thiazide Diuretic | Chlorthalidone | 12.5-25 mg daily | SHEP, ALLHAT trials | Volume reduction; caution with electrolytes |
Avoid: Beta-blockers as first-line (less effective for stroke prevention compared to ACE-I/ARB).
Typical Regimen:
- Start: Lisinopril 10 mg + Amlodipine 5 mg
- Titrate: Lisinopril to 20-40 mg, Amlodipine to 10 mg
- Add: Chlorthalidone 12.5-25 mg if still above target
- Goal: SBP less than 130 mmHg, DBP less than 80 mmHg
Monitoring: Home BP monitoring preferred; target average less than 130/80.
7.5 Diabetes Management
Diabetes increases stroke risk 2-fold and worsens post-stroke outcomes. However, intensive glycemic control has NOT been proven to reduce recurrent stroke risk (ACCORD trial showed no stroke benefit with HbA1c less than 6% vs less than 7%).
Target HbA1c: 6.5-7.0% (individualized based on age, comorbidities, hypoglycemia risk)
Avoid: Hypoglycemia (less than 70 mg/dL) — associated with increased stroke risk and worse outcomes.
Preferred Agents (Cardiovascular Benefit):
| Class | Agent | Cardiovascular Benefit | Mechanism |
|---|---|---|---|
| GLP-1 Receptor Agonist | Semaglutide 0.5-1 mg SC weekly OR Dulaglutide 1.5 mg SC weekly | Reduces stroke by 26% (SUSTAIN-6, LEADER trials) | Weight loss, BP reduction, anti-inflammatory |
| SGLT2 Inhibitor | Empagliflozin 10-25 mg daily OR Dapagliflozin 10 mg daily | Reduces heart failure, may reduce stroke | Diuresis, BP reduction, renal protection |
| Metformin | 500-1000 mg BID | First-line for most patients | Insulin sensitizer, weight neutral |
Avoid Pioglitazone in stroke patients with heart failure (fluid retention).
7.6 Carotid Revascularization
Symptomatic carotid stenosis (stroke/TIA in the territory of a stenotic carotid artery) warrants urgent revascularization.
Indications for Carotid Endarterectomy (CEA) or Carotid Stenting (CAS):
| Stenosis Severity | Symptomatic (Stroke/TIA less than 6 months) | Asymptomatic |
|---|---|---|
| 70-99% | Strong indication: CEA/CAS within 2 weeks (14-day rule) | Consider if life expectancy > 5 years and low surgical risk |
| 50-69% | Moderate indication: CEA/CAS within 2 weeks (benefit less robust) | Generally not indicated |
| less than 50% | Medical management only | Medical management only |
Evidence:
- NASCET (North American Symptomatic Carotid Endarterectomy Trial): 70-99% stenosis → CEA reduced 2-year stroke risk from 26% to 9% (NNT=6)
- ECST (European Carotid Surgery Trial): Confirmed benefit for 70-99% stenosis
- EXPRESS Study: Early surgery (less than 14 days) reduced 90-day stroke risk by 80% vs delayed surgery [23]
7.6.1 CEA vs CAS
Carotid Endarterectomy (CEA):
- Gold standard surgical technique
- Lower periprocedural stroke risk
- Preferred in most patients
Carotid Artery Stenting (CAS):
- Percutaneous endovascular approach
- Higher periprocedural stroke risk (especially in elderly)
- Indications: High surgical risk, radiation-induced stenosis, restenosis post-CEA, anatomically inaccessible lesion
CREST Trial: CEA and CAS had similar long-term outcomes, but CEA had lower periprocedural stroke risk, while CAS had lower MI risk.
Recommendation: CEA preferred for most patients; CAS for selected high surgical risk cases.
7.7 Lifestyle Modifications
| Intervention | Recommendation | Impact on Stroke Risk |
|---|---|---|
| Smoking Cessation | Complete cessation | Risk returns to baseline in 5 years; immediate risk reduction starts within 1 year |
| Diet | Mediterranean diet (high in fruits, vegetables, whole grains, fish, olive oil) | 20-30% RRR for cardiovascular events; PREDIMED trial [30] |
| Physical Activity | 150 min/week moderate intensity (brisk walking) OR 75 min/week vigorous | 27% RRR for stroke |
| Weight Management | BMI 18.5-24.9 kg/m² | Obesity increases stroke risk 1.6-fold; weight loss improves all risk factors |
| Alcohol | ≤2 drinks/day (men), ≤1 drink/day (women) | J-shaped curve; moderate use may be protective, heavy use increases risk |
| Salt Restriction | less than 2.3 g/day sodium (less than 1.5 g ideal) | Lowers BP by 5-10 mmHg |
| Sleep | 7-9 hours/night; treat OSA if present | OSA increases stroke risk 2-fold; CPAP improves outcomes |
7.8 Patent Foramen Ovale (PFO) Closure
Indication: Cryptogenic stroke in patients age less than 60 with PFO and atrial septal aneurysm or large right-to-left shunt.
Evidence: CLOSE, RESPECT, REDUCE trials showed PFO closure superior to medical therapy alone in select patients (NNT=10-20). [30]
Procedure: Percutaneous transcatheter PFO closure device (Amplatzer, Gore Cardioform).
Post-Procedure: DAPT × 6 months, then aspirin indefinitely.
7.9 Comprehensive Secondary Prevention Checklist
Every ischemic stroke patient should have:
☑️ Antiplatelet therapy (aspirin, clopidogrel, or DAPT if minor stroke) ☑️ Anticoagulation if AF (DOAC preferred) ☑️ High-intensity statin (atorvastatin 40-80 mg) ☑️ Blood pressure control (less than 130/80 mmHg with ACE-I or ARB) ☑️ Diabetes management (HbA1c less than 7%, GLP-1 RA or SGLT2i if applicable) ☑️ Carotid imaging and revascularization if 50-99% symptomatic stenosis ☑️ Lifestyle counseling: smoking cessation, diet, exercise, weight ☑️ Swallow assessment and speech therapy if dysphagic/aphasic ☑️ PT/OT for rehabilitation ☑️ Depression screening and treatment (affects 30-50%) ☑️ Driving assessment (most countries require MD clearance post-stroke)
SECTION 8: Complications
8.1 Immediate Complications (0-72 hours)
| Complication | Incidence | Risk Factors | Presentation | Management |
|---|---|---|---|---|
| Hemorrhagic transformation | 6% (tPA); 1-2% (no tPA) | Large infarct, high BP, anticoagulation | Sudden deterioration, new headache | Stop anticoagulation, CT, supportive |
| Cerebral edema | 10-15% of large strokes | Large MCA infarct, young age | Decreased LOC, pupil changes | Osmotherapy, decompressive surgery |
| Seizures | 2-5% | Cortical involvement, hemorrhage | Focal or generalized seizures | Levetiracetam; avoid phenytoin |
| Aspiration pneumonia | 10-15% | Dysphagia, decreased LOC | Fever, hypoxia, infiltrate | NPO, antibiotics |
| DVT/PE | 2-5% with prophylaxis | Immobility, paralysis | Leg swelling, dyspnea | IPC, LMWH after 24-48h |
| Cardiac complications | 15-20% | Pre-existing heart disease | Arrhythmias, MI, heart failure | Telemetry, cardiology consult |
8.2 Malignant Middle Cerebral Artery Syndrome
Definition: Life-threatening edema following large MCA infarct, causing herniation.
Risk Factors:
- Age less than 60 years
- NIHSS > 15
-
50% MCA territory infarct
- Decreased LOC within 24 hours
Management:
- Decompressive hemicraniectomy: Within 48 hours
- DESTINY, DECIMAL, HAMLET trials: Reduces mortality from 78% to 29% [20]
- Age consideration: Greatest benefit in patients less than 60 years
8.3 Late Complications
| Complication | Timeframe | Incidence | Management |
|---|---|---|---|
| Post-stroke depression | Weeks to months | 30-50% | SSRIs; multidisciplinary support |
| Spasticity | Weeks | 20-40% | Physiotherapy, Baclofen, Botulinum toxin |
| Central post-stroke pain | Months | 5-10% (thalamic strokes) | Amitriptyline, Pregabalin |
| Post-stroke epilepsy | Months to years | 5-10% | Antiepileptic drugs |
| Vascular dementia | Years | 20-30% | Risk factor control; cognitive rehabilitation |
| Recurrent stroke | Ongoing | 3-5%/year with optimal treatment | Secondary prevention |
SECTION 9: Prognosis and Outcomes
9.1 Prognostic Factors
| Factor | Impact on Outcome |
|---|---|
| Age | Older age → worse functional outcome |
| Stroke severity (NIHSS) | Higher NIHSS → worse outcome |
| Time to treatment | Faster treatment → better outcome |
| Stroke type | Hemorrhagic → worse prognosis than ischemic |
| Infarct location | Brainstem and dominant hemisphere → worse |
| Collateral circulation | Good collaterals → better outcome |
| Reperfusion success | TICI 2b/3 → better outcome |
| Comorbidities | Diabetes, AF, heart failure → worse |
| Stroke unit care | Improves all outcomes |
9.2 Outcome Measures
Modified Rankin Scale (mRS):
| Score | Description |
|---|---|
| 0 | No symptoms |
| 1 | No significant disability; able to carry out all usual duties |
| 2 | Slight disability; unable to carry out all previous activities but able to look after own affairs |
| 3 | Moderate disability; requires some help but able to walk without assistance |
| 4 | Moderately severe disability; unable to walk without assistance and unable to attend to own bodily needs without assistance |
| 5 | Severe disability; bedridden, incontinent, requires constant nursing care |
| 6 | Dead |
9.3 Natural History and Treatment Outcomes
| Outcome | Without Treatment | With Treatment (Thrombolysis/Thrombectomy) |
|---|---|---|
| Functional independence (mRS 0-2) | 25% (LVO) | 40-50% |
| 30-day mortality (ischemic) | 10-15% | 8-12% |
| 1-year mortality (ischemic) | 20-25% | 12-15% |
| 30-day mortality (ICH) | 40-50% | 35-45% |
| Recurrent stroke (5 years) | 25-30% | 10-15% (with secondary prevention) |
SECTION 10: Evidence and Guidelines
10.1 Key Guidelines
AHA/ASA Guidelines for the Early Management of Acute Ischemic Stroke (2019, 2021 Update) [2]
- IV thrombolysis with alteplase or tenecteplase for eligible patients within 4.5 hours (Class I)
- Mechanical thrombectomy for LVO within 6 hours (Class I)
- Thrombectomy up to 24 hours with perfusion imaging selection (Class I)
- Dual antiplatelet therapy for minor stroke/high-risk TIA (Class I)
- Stroke unit care for all stroke patients (Class I)
European Stroke Organisation (ESO) Guidelines (2021)
- Recommendations align with AHA/ASA
- Emphasis on regional stroke networks and telestroke
10.2 Landmark Trials
Thrombolysis Trials:
| Trial | Year | N | Key Finding | PMID |
|---|---|---|---|---|
| NINDS rt-PA [13] | 1995 | 624 | tPA within 3h increased minimal/no disability by 30% at 3 months | 7477192 |
| ECASS III [21] | 2008 | 821 | Extended window to 4.5 hours with favorable outcomes | 18815396 |
| IST-3 [22] | 2012 | 3035 | Benefit extends to older patients and higher NIHSS | 22632908 |
Thrombectomy Trials:
| Trial | Year | N | Key Finding | PMID |
|---|---|---|---|---|
| MR CLEAN [7] | 2015 | 500 | First positive thrombectomy trial; NNT=7 for mRS 0-2 | 25517348 |
| ESCAPE | 2015 | 316 | Rapid workflow, NNT=4 for mRS 0-2 | 25671798 |
| SWIFT PRIME | 2015 | 196 | Stent retriever plus tPA superior | 25882376 |
| EXTEND-IA | 2015 | 70 | CT perfusion selection; 71% reperfusion | 25882376 |
| REVASCAT | 2015 | 206 | Benefit at 90 days | 25671797 |
| HERMES meta-analysis [6] | 2016 | 1287 | Pooled analysis; NNT=2.6 | 26898852 |
| DAWN [3] | 2018 | 206 | Extended window to 24h with clinical-core mismatch | 29129659 |
| DEFUSE-3 [4] | 2018 | 182 | Extended window to 16h with perfusion mismatch | 29414764 |
Secondary Prevention Trials:
| Trial | Year | N | Key Finding | PMID |
|---|---|---|---|---|
| CHANCE [15] | 2013 | 5170 | DAPT × 21 days reduced stroke by 32% | 23803136 |
| POINT [16] | 2018 | 4881 | DAPT × 90 days reduced stroke but increased bleeding | 29766750 |
| SPARCL [18] | 2006 | 4731 | High-dose atorvastatin reduced recurrent stroke by 16% | 16899775 |
| PROGRESS [19] | 2001 | 6105 | Perindopril ± indapamide reduced stroke by 28% | 11565518 |
SECTION 11: Patient and Layperson Explanation
11.1 What is a Stroke?
A stroke happens when blood flow to part of the brain is blocked (like a clogged pipe) or when a blood vessel in the brain bursts (like a burst pipe). When brain cells don't get blood, they start to die within minutes. This is why doctors call it a "brain attack" — just like a heart attack, but in the brain.
Two main types:
- Ischemic stroke (85%): A blood clot blocks an artery. This is like a traffic jam stopping all the cars.
- Hemorrhagic stroke (15%): A blood vessel bursts and bleeds into the brain. This is more dangerous and harder to treat.
11.2 How Do I Recognize a Stroke? (BE FAST)
| Letter | Sign | What to Look For |
|---|---|---|
| B | Balance | Sudden loss of balance or coordination |
| E | Eyes | Sudden vision problems in one or both eyes |
| F | Face | Ask the person to smile. Does one side droop? |
| A | Arms | Ask them to raise both arms. Does one arm drift down? |
| S | Speech | Ask them to repeat a simple phrase. Is speech slurred or strange? |
| T | Time | If you see ANY of these signs, call emergency services IMMEDIATELY |
Remember: Every minute matters! Treatments work best when given within hours. "Time is brain."
11.3 Why Does It Matter?
Every minute of a major stroke, nearly 2 million brain cells die. The sooner treatment is given:
- The more brain is saved
- The better the recovery
- The more likely you are to walk, talk, and live independently
11.4 What Happens at the Hospital?
- Immediate CT scan — to see if it's a clot or a bleed
- Blood tests — to check your blood sugar and clotting
- Clot-busting medicine — if it's a clot and you arrived in time
- Clot removal procedure — if there's a large clot, doctors can pull it out with a special wire
- Monitoring — in a specialized stroke unit
11.5 After a Stroke
- Rehabilitation: Physical therapy, occupational therapy, speech therapy
- Medications: Blood thinners, cholesterol medications, blood pressure medications
- Lifestyle changes: Stop smoking, healthy diet, exercise, control blood pressure and diabetes
- Follow-up: Regular appointments with your stroke team
SECTION 12: Examination Focus
12.1 Common Exam Questions (MRCP, USMLE, FRCS)
Question 1 (MRCP): A 72-year-old man presents 2 hours after sudden onset of right-sided weakness and speech difficulty. BP is 195/110 mmHg. CT head shows no hemorrhage. What is the most appropriate next step?
- Answer: Lower BP to less than 185/110 with IV labetalol or nicardipine, then administer IV tenecteplase or alteplase.
Question 2 (USMLE): A patient with acute ischemic stroke is found to have an M1 MCA occlusion on CTA. Symptom onset was 8 hours ago. What additional imaging is required to determine thrombectomy eligibility?
- Answer: CT perfusion (or MRI DWI/perfusion) to assess core infarct volume and penumbra (DAWN/DEFUSE-3 criteria).
Question 3 (FRCS): What is the most common cause of death within 1 week of a large MCA infarct?
- Answer: Cerebral edema and transtentorial herniation.
Question 4 (MRCP): A patient with atrial fibrillation presents 5 days after a minor ischemic stroke (NIHSS 2). When should anticoagulation be started?
- Answer: 1-3 days after minor stroke. Given NIHSS 2, consider starting at day 3-5 after repeat imaging excludes hemorrhagic transformation.
Question 5 (USMLE): What is the NIHSS component that is most commonly affected in a right MCA stroke but often underscores severity?
- Answer: Extinction/inattention (hemispatial neglect). The NIHSS underestimates right hemisphere strokes.
12.2 Viva Opening Statement
"Stroke is a medical emergency caused by the sudden interruption of cerebral blood flow, either due to arterial occlusion (ischemic, 85%) or vessel rupture (hemorrhagic, 15%). It is characterized by sudden-onset focal neurological deficits corresponding to a vascular territory. Recognition using FAST or BE-FAST criteria enables immediate activation of hyperacute stroke pathways. The cornerstone of ischemic stroke treatment is rapid reperfusion through intravenous thrombolysis within 4.5 hours and mechanical thrombectomy for large vessel occlusion up to 24 hours in selected patients. 'Time is brain' — every minute of delay results in loss of 1.9 million neurons. Secondary prevention with antiplatelets, statins, and blood pressure control reduces recurrence. Hemorrhagic stroke requires blood pressure control, reversal of anticoagulation, and consideration of surgical intervention."
12.3 High-Yield Facts for Exams
| Topic | Key Fact |
|---|---|
| Time is brain | 1.9 million neurons lost per minute in LVO |
| Thrombolysis window | 4.5 hours (alteplase/tenecteplase) |
| Thrombectomy window (standard) | 6 hours for anterior LVO |
| Thrombectomy extended window | Up to 24 hours with DAWN/DEFUSE-3 imaging criteria |
| Thrombectomy NNT | 2.6 for 1-grade improvement in mRS |
| BP target for thrombolysis | less than 185/110 pre-treatment; less than 180/105 post-treatment |
| BP target for ICH | SBP less than 140 mmHg |
| DAPT after minor stroke | Aspirin + Clopidogrel for 21 days |
| Carotid surgery timing | Within 2 weeks of symptomatic stenosis |
| Most common stroke territory | Middle cerebral artery (MCA) |
| Lacunar syndromes | 5 classic syndromes; no cortical signs |
| Posterior circulation | 5 Ds: Dizziness, Diplopia, Dysarthria, Dysphagia, Dystaxia |
SECTION 13: References
-
Saver JL. Time is brain—quantified. Stroke. 2006;37(1):263-266. PMID: 16339467 DOI: 10.1161/01.STR.0000196957.55928.ab
-
Powers WJ, Rabinstein AA, Ackerson T, et al. Guidelines for the Early Management of Patients With Acute Ischemic Stroke: 2019 Update to the 2018 Guidelines. Stroke. 2019;50(12):e344-e418. PMID: 31662037 DOI: 10.1161/STR.0000000000000211
-
Nogueira RG, Jadhav AP, Haussen DC, et al. Thrombectomy 6 to 24 Hours after Stroke with a Mismatch between Deficit and Infarct (DAWN). N Engl J Med. 2018;378(1):11-21. PMID: 29129157 DOI: 10.1056/NEJMoa1706442
-
Albers GW, Marks MP, Kemp S, et al. Thrombectomy for Stroke at 6 to 16 Hours with Selection by Perfusion Imaging (DEFUSE 3). N Engl J Med. 2018;378(8):708-718. PMID: 29414764 DOI: 10.1056/NEJMoa1713973
-
Virani SS, Alonso A, Aparicio HJ, et al. Heart Disease and Stroke Statistics—2021 Update. Circulation. 2021;143(8):e254-e743. PMID: 33501848 DOI: 10.1161/CIR.0000000000000950
-
Goyal M, Menon BK, van Zwam WH, et al. Endovascular thrombectomy after large-vessel ischaemic stroke: a meta-analysis of individual patient data from five randomised trials (HERMES). Lancet. 2016;387(10029):1723-1731. PMID: 26898852 DOI: 10.1016/S0140-6736(16)00163-X
-
Berkhemer OA, Fransen PS, Beumer D, et al. A randomized trial of intraarterial treatment for acute ischemic stroke (MR CLEAN). N Engl J Med. 2015;372(1):11-20. PMID: 25517348 DOI: 10.1056/NEJMoa1411587
-
Stroke Unit Trialists' Collaboration. Organised inpatient (stroke unit) care for stroke. Cochrane Database Syst Rev. 2013;(9):CD000197. PMID: 24026639 DOI: 10.1002/14651858.CD000197.pub3
-
Anderson CS, Heeley E, Huang Y, et al. Rapid blood-pressure lowering in patients with acute intracerebral hemorrhage (INTERACT2). N Engl J Med. 2013;368(25):2355-2365. PMID: 23713578 DOI: 10.1056/NEJMoa1214609
-
Thomalla G, Simonsen CZ, Boutitie F, et al. MRI-Guided Thrombolysis for Stroke with Unknown Time of Onset (WAKE-UP). N Engl J Med. 2018;379(7):611-622. PMID: 29766770 DOI: 10.1056/NEJMoa1804355
-
Hemphill JC 3rd, Greenberg SM, Anderson CS, et al. Guidelines for the Management of Spontaneous Intracerebral Hemorrhage. Stroke. 2015;46(7):2032-2060. PMID: 26022637 DOI: 10.1161/STR.0000000000000069
-
Campbell BCV, Mitchell PJ, Churilov L, et al. Tenecteplase versus Alteplase before Thrombectomy for Ischemic Stroke (EXTEND-IA TNK). N Engl J Med. 2018;378(17):1573-1582. PMID: 29694815 DOI: 10.1056/NEJMoa1716405
-
National Institute of Neurological Disorders and Stroke rt-PA Stroke Study Group. Tissue plasminogen activator for acute ischemic stroke. N Engl J Med. 1995;333(24):1581-1587. PMID: 7477192 DOI: 10.1056/NEJM199512143332401
-
Qureshi AI, Palesch YY, Barsan WG, et al. Intensive Blood-Pressure Lowering in Patients with Acute Cerebral Hemorrhage (ATACH-2). N Engl J Med. 2016;375(11):1033-1043. PMID: 27276234 DOI: 10.1056/NEJMoa1603460
-
Wang Y, Wang Y, Zhao X, et al. Clopidogrel with aspirin in acute minor stroke or transient ischemic attack (CHANCE). N Engl J Med. 2013;369(1):11-19. PMID: 23803136 DOI: 10.1056/NEJMoa1215340
-
Johnston SC, Easton JD, Farrant M, et al. Clopidogrel and Aspirin in Acute Ischemic Stroke and High-Risk TIA (POINT). N Engl J Med. 2018;379(3):215-225. PMID: 29766750 DOI: 10.1056/NEJMoa1800410
-
Johnston SC, Amarenco P, Denison H, et al. Ticagrelor and Aspirin or Aspirin Alone in Acute Ischemic Stroke or TIA (THALES). N Engl J Med. 2020;383(3):207-217. PMID: 32668111 DOI: 10.1056/NEJMoa1916870
-
Amarenco P, Bogousslavsky J, Callahan A 3rd, et al. High-dose atorvastatin after stroke or transient ischemic attack (SPARCL). N Engl J Med. 2006;355(6):549-559. PMID: 16899775 DOI: 10.1056/NEJMoa061894
-
PROGRESS Collaborative Group. Randomised trial of a perindopril-based blood-pressure-lowering regimen among 6,105 individuals with previous stroke or transient ischaemic attack. Lancet. 2001;358(9287):1033-1041. PMID: 11565518 DOI: 10.1016/S0140-6736(01)06178-5
-
Vahedi K, Hofmeijer J, Juettler E, et al. Early decompressive surgery in malignant infarction of the middle cerebral artery: a pooled analysis of three randomised controlled trials (DESTINY, DECIMAL, HAMLET). Lancet Neurol. 2007;6(3):215-222. PMID: 17303527 DOI: 10.1016/S1474-4422(07)70036-4
-
Hacke W, Kaste M, Bluhmki E, et al. Thrombolysis with alteplase 3 to 4.5 hours after acute ischemic stroke (ECASS III). N Engl J Med. 2008;359(13):1317-1329. PMID: 18815396 DOI: 10.1056/NEJMoa0804656
-
IST-3 Collaborative Group. The benefits and harms of intravenous thrombolysis with recombinant tissue plasminogen activator within 6 h of acute ischaemic stroke (the third international stroke trial [IST-3]): a randomised controlled trial. Lancet. 2012;379(9834):2352-2363. PMID: 22632908 DOI: 10.1016/S0140-6736(12)60768-5
-
Rothwell PM, Giles MF, Chandratheva A, et al. Effect of urgent treatment of transient ischaemic attack and minor stroke on early recurrent stroke (EXPRESS study). Lancet. 2007;370(9596):1432-1442. PMID: 17928046 DOI: 10.1016/S0140-6736(07)61448-2
-
Easton JD, Saver JL, Albers GW, et al. Definition and evaluation of transient ischemic attack. Stroke. 2009;40(6):2276-2293. PMID: 19423857 DOI: 10.1161/STROKEAHA.108.192218
-
Kernan WN, Ovbiagele B, Black HR, et al. Guidelines for the prevention of stroke in patients with stroke and transient ischemic attack. Stroke. 2014;45(7):2160-2236. PMID: 24788967 DOI: 10.1161/STR.0000000000000024
-
Lyden P, Brott T, Tilley B, et al. Improved reliability of the NIH Stroke Scale using video training. Stroke. 1994;25(11):2220-2226. PMID: 7974549 DOI: 10.1161/01.STR.25.11.2220
-
Katsanos AH, Safouris A, Sarraj A, et al. Intravenous Thrombolysis With Tenecteplase in Patients With Large Vessel Occlusions: Systematic Review and Meta-Analysis. Stroke. 2021;52(8):2681-2688. PMID: 34134545 DOI: 10.1161/STROKEAHA.120.033214
-
Saver JL, Fonarow GC, Smith EE, et al. Time to Treatment With Intravenous Tissue Plasminogen Activator and Outcome From Acute Ischemic Stroke. JAMA. 2013;309(23):2480-2488. PMID: 23780461 DOI: 10.1001/jama.2013.6959
-
Schönenberger S, Uhlmann L, Hacke W, et al. Effect of Conscious Sedation vs General Anesthesia on Early Neurological Improvement Among Patients With Ischemic Stroke Undergoing Endovascular Thrombectomy: A Randomized Clinical Trial (SIESTA). JAMA. 2016;316(19):1986-1996. PMID: 27785516 DOI: 10.1001/jama.2016.16623
-
Hindricks G, Potpara T, Dagres N, et al. 2020 ESC Guidelines for the diagnosis and management of atrial fibrillation. Eur Heart J. 2021;42(5):373-498. PMID: 32860505 DOI: 10.1093/eurheartj/ehaa612
Version History
| Version | Date | Changes |
|---|---|---|
| 1.0 | 2026-01-02 | Initial creation |
Last Updated: 2026-01-09
Frequently asked questions
Quick clarifications for common clinical and exam-facing questions.
When should I seek emergency care for acute stroke in adults?
Seek immediate emergency care if you experience any of the following warning signs: Sudden onset focal neurological deficit (FAST positive), Sudden 'worst headache of life' (Thunderclap headache suggesting SAH), Rapidly declining Level of Consciousness (GCS), New onset seizures following focal deficit, Signs of brainstem herniation (Cushing's triad), Blood pressure less than 220/120 mmHg, Within thrombolysis window (less than 4.5 hours from onset), Within thrombectomy window (less than 6-24 hours with imaging selection), Posterior circulation signs (vertigo, diplopia, ataxia, crossed signs), Neck pain with Horner's syndrome (arterial dissection).
Learning map
Use these linked topics to study the concept in sequence and compare related presentations.
Prerequisites
Start here if you need the foundation before this topic.
- Cerebrovascular Anatomy
- Neurological Examination
- NIHSS Assessment
Differentials
Competing diagnoses and look-alikes to compare.
- Transient Ischaemic Attack (TIA)
- Hypoglycaemia
- Seizure with Todd's Paresis
- Migraine with Aura
- Intracranial Mass Lesion
- Functional Neurological Disorder
Consequences
Complications and downstream problems to keep in mind.
- Stroke Rehabilitation
- Post-Stroke Complications
- Vascular Dementia