Type 2 Diabetes Mellitus (Adult)
Type 2 Diabetes Mellitus (T2DM) is a chronic progressive metabolic disorder characterized by chronic hyperglycemia resul... MRCP, USMLE exam preparation.
What matters first
Type 2 Diabetes Mellitus (T2DM) is a chronic progressive metabolic disorder characterized by chronic hyperglycemia resul... MRCP, USMLE exam preparation.
Hyperglycaemic Hyperosmolar State (HHS) - profound dehydration, altered mental status, osmolality less than 320 mOsm/kg
9 Jan 2026
Generated educational material; verify before clinical use.
Visible references section
See the concept before reading it
Study the key anatomy, imaging, and decision pathways as full teaching plates.
Clinical board
A visual summary of the highest-yield teaching signals on this page.
Urgent signals
Safety-critical features pulled from the topic metadata.
- Hyperglycaemic Hyperosmolar State (HHS) - profound dehydration, altered mental status, osmolality less than 320 mOsm/kg
- Diabetic Ketoacidosis (DKA) - rare in T2DM but possible with SGLT2i use (euglycemic variant)
- Acute visual loss (vitreous hemorrhage, retinal detachment, or central retinal vein occlusion)
- Unexplained weight loss less than 10% with polyuria (severe insulin deficiency or malignancy)
Exam focus
Current exam surfaces linked to this topic.
- MRCP
- USMLE
- PLAB
Content status and exam context
This page is AI-generated educational content. It may contain errors or omissions and is not a substitute for current guidelines, local protocols, senior clinical judgement, or professional medical advice.
MedVellum does not claim an individual clinician reviewer, board certification, or professional credential for this page unless a future version names a real, verifiable reviewer.
Clinical explanation and evidence
Type 2 Diabetes Mellitus (Adult)
1. Clinical Overview
Summary
Type 2 Diabetes Mellitus (T2DM) is a chronic progressive metabolic disorder characterized by chronic hyperglycemia resulting from a dual pathophysiological defect: peripheral and hepatic insulin resistance coupled with progressive pancreatic beta-cell dysfunction. Unlike Type 1 diabetes, where absolute insulin deficiency predominates, T2DM involves relative insulin deficiency in the context of increased insulin demand. The condition affects over 537 million adults globally (2021), with projections reaching 783 million by 2045, making it one of the defining public health crises of the 21st century. [1,2]
The pathophysiology has evolved from the classical "triumvirate" (muscle insulin resistance, hepatic glucose overproduction, and beta-cell failure) to DeFronzo's "Ominous Octet", which recognizes eight distinct organ defects contributing to hyperglycemia: pancreatic beta-cell failure, alpha-cell hyperglucagonemia, hepatic glucose overproduction, muscle insulin resistance, adipocyte lipolysis, incretin deficiency, renal glucose reabsorption, and brain insulin resistance. [3] This paradigm shift has revolutionized therapy, moving beyond simple glucose-lowering to multi-organ targeted treatment.
T2DM is frequently asymptomatic in its early stages, with up to 50% of cases remaining undiagnosed. Consequently, approximately 20-30% of patients already have established microvascular complications (retinopathy, nephropathy, neuropathy) at the time of diagnosis. [4] The condition is more accurately described as a cardiovascular disease with hyperglycemia rather than simply a glucose disorder: patients with T2DM have a 2-4 fold increased risk of cardiovascular death, myocardial infarction, and stroke compared to non-diabetic individuals. [5]
Diagnosis relies on demonstrating persistent hyperglycemia through HbA1c ≥48 mmol/mol (6.5%), fasting plasma glucose ≥7.0 mmol/L, or 2-hour OGTT glucose ≥11.1 mmol/L. Management has undergone a paradigm transformation over the past decade with the emergence of cardio-renal protective therapies: SGLT2 inhibitors and GLP-1 receptor agonists are now prioritized early in the treatment algorithm for patients with established atherosclerotic cardiovascular disease (ASCVD), heart failure, or chronic kidney disease (CKD), independent of baseline HbA1c. [6,7,8]
Lifestyle modification remains foundational, with evidence from the DiRECT trial demonstrating that intensive weight loss (> 15 kg) can induce diabetes remission in approximately 46% of patients within the first two years of diagnosis. [9] This challenges the traditional view of T2DM as an inevitably progressive disease and emphasizes the critical importance of early, aggressive intervention.
Key Facts
- Prevalence: Affects 10.5% of adults aged 20-79 globally (537 million in 2021); projected 783 million by 2045.
- Diagnosis: HbA1c ≥48 mmol/mol (6.5%), FPG ≥7.0 mmol/L, or 2h-OGTT ≥11.1 mmol/L. Two tests required if asymptomatic.
- Pathophysiology: Insulin resistance (muscle, liver, adipose) + progressive beta-cell failure. The "Ominous Octet" involves eight organ defects.
- First-Line Therapy: Metformin 500mg-1g BD plus structured lifestyle intervention (Mediterranean diet, 150 min/week moderate exercise).
- Cardio-Renal Protection: SGLT2 inhibitors reduce CV death by 38%, HF hospitalization by 35%, and CKD progression by 40%. GLP-1 RAs reduce MACE by 14%.
- Glycemic Targets: Individualized. Typically HbA1c 48-53 mmol/mol (6.5-7.0%) for most adults; 58-64 mmol/mol (7.5-8.0%) for elderly/frail.
- Microvascular Complications: Retinopathy (leading cause of working-age blindness), nephropathy (leading cause of ESRD), neuropathy (50% at 10 years).
- Macrovascular Complications: 2-4x risk of MI, stroke, PAD. Silent MI common (autonomic neuropathy).
- Screening: Annual UACR, eGFR, lipids, retinal photography, foot examination, BP.
- Remission Possible: 46% remission rate with > 15 kg weight loss in DiRECT trial. Bariatric surgery achieves 60-80% remission at 2 years.
- Mortality: T2DM reduces life expectancy by 6-8 years; 50% die from cardiovascular events.
- Acute Emergencies: HHS (mortality 10-20%), euglycemic DKA (SGLT2i-associated), severe hypoglycemia.
Clinical Pearls
"Treat the patient, not the HbA1c" — An HbA1c of 52 mmol/mol may be excellent for a 45-year-old newly diagnosed patient but dangerous for an 82-year-old with recurrent falls and hypoglycemia unawareness. Individualize targets based on hypoglycemia risk, life expectancy, patient preferences, and comorbidities.
"T2DM is a cardiovascular disease that happens to have high glucose" — More patients die from MI than from hyperglycemia. Statins, ACE inhibitors, and antiplatelet therapy are as critical as glucose-lowering drugs. Always assess 10-year ASCVD risk.
"SGLT2i and GLP-1 RA are organ-protective, not just glucose-lowering" — The EMPA-REG OUTCOME trial showed empagliflozin reduced cardiovascular death by 38% within 3 months—far too early for glucose effects. These drugs protect the heart and kidneys through mechanisms independent of HbA1c lowering.
"The foot tells you everything you need to know about diabetes control" — A Charcot foot, painless ulcer, or absent pedal pulses indicate years of poor glycemic control and microvascular/macrovascular damage. Never discharge a diabetic patient without examining their feet.
"Check for 'sick day rules' knowledge before every discharge" — Patients must know to stop metformin and SGLT2i during vomiting/diarrhea to prevent lactic acidosis and euglycemic DKA. This is a common OSCE/PACES station.
"Weight loss of > 15 kg can reverse diabetes" — The DiRECT trial showed 46% remission. Always discuss the option of intensive weight management (very low-calorie diet or bariatric surgery) for motivated patients with less than 6 years disease duration.
"HbA1c can be falsely low in anemia, hemolysis, CKD, or recent transfusion" — Use fructosamine or continuous glucose monitoring (CGM) if HbA1c is discordant with home glucose readings.
Why This Matters Clinically
Type 2 Diabetes Mellitus is the archetypal chronic disease of modern civilization, driven by the twin epidemics of obesity and sedentary lifestyle. It exemplifies the shift from acute, infectious disease-dominated healthcare to chronic, metabolic disease management. Clinicians must be proficient not only in glucose control but also in comprehensive cardiovascular risk reduction, early detection and prevention of microvascular complications, and patient-centered shared decision-making.
The past decade has witnessed a revolution in diabetes therapeutics. No longer is the goal simply to "lower the sugar"; modern diabetes care demands the integration of cardio-renal protective agents (SGLT2 inhibitors, GLP-1 receptor agonists) as foundational therapy alongside metformin. The evidence from landmark trials (EMPA-REG OUTCOME, LEADER, CREDENCE, DAPA-CKD) has fundamentally altered treatment algorithms. [7,8,10,11]
Furthermore, diabetes is a disease of inequity: it disproportionately affects ethnic minorities, low-income populations, and those with limited health literacy. Clinicians must be culturally sensitive, employ structured education programs (DESMOND, DAFNE), and address social determinants of health to achieve meaningful outcomes.
Finally, T2DM is no longer a one-way street. With intensive lifestyle intervention or bariatric surgery, remission is achievable in a significant proportion of patients, particularly if intervention occurs early in the disease course. This represents a profound shift in how we counsel patients at diagnosis.
2. Epidemiology
Global Burden
The International Diabetes Federation (IDF) Diabetes Atlas (10th Edition, 2021) estimates that 537 million adults (20-79 years) are living with diabetes globally, with T2DM accounting for approximately 90-95% of all cases. Prevalence is projected to rise to 783 million by 2045, representing a 46% increase. [1,2] This exponential growth is driven by aging populations, urbanization, dietary transitions toward ultra-processed foods, and declining physical activity.
Low- and middle-income countries (LMICs) bear the greatest burden: 80% of people with diabetes live in LMICs, where healthcare infrastructure, access to medications, and structured diabetes care are often limited. India and China alone account for over 250 million cases combined.
Incidence & Prevalence
Global Prevalence:
- Adults aged 20-79: 10.5% (2021).
- Highest regional prevalence: Middle East and North Africa (16.2%), North America and Caribbean (13.9%).
- Lowest prevalence: Africa (5.1%), though absolute numbers are rising rapidly.
Age-Specific Prevalence:
- less than 40 years: 2-3% (but rising sharply due to childhood obesity).
- 40-59 years: 12-15%.
- 60-79 years: 20-25%.
-
80 years: 25-30%.
Undiagnosed Diabetes: Approximately 50% of adults with diabetes are undiagnosed, particularly in sub-Saharan Africa and South-East Asia. This diagnostic inertia results in delayed treatment and a high prevalence of complications at diagnosis.
Demographics
Age
Traditionally considered a disease of middle and older age, T2DM is increasingly diagnosed in children, adolescents, and young adults due to the pediatric obesity epidemic. The term "adult-onset diabetes" is now obsolete. Incidence peaks at age 60-69 years but is rising most rapidly in the 20-39 age group.
Sex
- Slight male predominance globally (prevalence ratio M:F = 1.1:1).
- However, women with diabetes have a relatively higher cardiovascular risk compared to men: diabetes abolishes the protective effect of female sex on coronary artery disease.
Ethnicity & Race
T2DM prevalence shows marked ethnic variation, even after adjustment for BMI:
| Ethnicity | Relative Risk vs White European | Comments |
|---|---|---|
| South Asian | 4-6x higher | Earlier age of onset (often less than 40 years), higher visceral adiposity at lower BMI. |
| African-Caribbean | 3-4x higher | Higher insulin resistance, greater beta-cell dysfunction. |
| Hispanic/Latino | 2-3x higher | Particularly Mexican-American populations. |
| Indigenous Populations | 5-10x higher | Pima Indians, Australian Aboriginals, Pacific Islanders. |
| East Asian | 2-3x higher | Lower BMI threshold for diabetes risk (BMI > 23 kg/m²). |
Why? Genetic predisposition (> 400 susceptibility loci identified via GWAS), epigenetics, thrifty gene hypothesis, differing body composition (higher visceral fat at lower BMI), and social determinants of health.
Socioeconomic Status
- Strong inverse correlation with income and education in high-income countries: T2DM is 2-3x more prevalent in the lowest income quintile.
- Mechanisms: Limited access to healthy food (food deserts), lower physical activity, chronic stress, and reduced access to preventive healthcare.
Risk Factors
Non-Modifiable
1. Age
- Risk doubles every decade after age 40.
- Beta-cell function declines ~0.5% per year from age 30 onward.
2. Family History
- First-degree relative with T2DM: 3-6x increased risk.
- Both parents with T2DM: 50-70% lifetime risk for offspring.
- Heritability estimated at 40-70% (polygenic inheritance).
3. Ethnicity
- As detailed above.
4. History of Gestational Diabetes Mellitus (GDM)
- 7-fold increased risk of T2DM within 10 years postpartum.
- Mechanism: GDM unmasks pre-existing beta-cell dysfunction.
5. Polycystic Ovary Syndrome (PCOS)
- 4-5x increased risk.
- Driven by severe insulin resistance and hyperinsulinemia.
6. Genetic Syndromes
- Monogenic diabetes (MODY, neonatal diabetes).
- Chromosomal abnormalities (Down syndrome, Turner syndrome).
- Lipodystrophy syndromes.
Modifiable Risk Factors
1. Obesity (BMI > 30 kg/m²) or Central Adiposity
- Single strongest modifiable risk factor.
- Relative risk: 20-40x for BMI > 35 kg/m² vs BMI less than 23 kg/m².
- Visceral adipose tissue (VAT) releases free fatty acids and adipokines (TNF-α, IL-6, resistin) that drive insulin resistance.
- Even modest weight loss (5-10% body weight) significantly reduces risk.
2. Physical Inactivity
- Sedentary behavior increases risk by 1.5-2x.
- Mechanism: Reduced non-insulin-mediated glucose uptake (GLUT4 translocation in muscle), reduced mitochondrial oxidative capacity, and increased visceral fat.
- 150 minutes/week of moderate-intensity exercise reduces risk by 30-40%.
3. Dietary Patterns
| Dietary Component | Association | Mechanism |
|---|---|---|
| Sugar-sweetened beverages | 25% ↑ risk per daily serving | Rapid glucose spikes, hepatic de novo lipogenesis, weight gain. |
| Processed meats | 20-50% ↑ risk | High saturated fat, sodium, nitrites; promote inflammation. |
| Refined carbohydrates | 40% ↑ risk (high vs low GI) | Postprandial hyperglycemia, beta-cell stress. |
| Whole grains | 25-30% ↓ risk | High fiber, slow glucose absorption, improved insulin sensitivity. |
| Mediterranean diet | 20-30% ↓ risk | Anti-inflammatory, high MUFA, polyphenols. |
4. Smoking
- Active smoking: 1.4-1.6x increased risk (dose-dependent).
- Mechanism: Increases visceral adiposity, cortisol levels, systemic inflammation, and directly impairs insulin signaling.
5. Sleep Deprivation
- less than 6 hours/night: 1.5x increased risk.
- Mechanism: Altered leptin/ghrelin balance, increased cortisol, reduced insulin sensitivity.
6. Chronic Stress & Depression
- Depression: 1.6x increased risk.
- Mechanism: Hypercortisolemia, inflammation, poor self-care behaviors.
7. Medications
- Glucocorticoids: Dose-dependent risk (inhibit insulin secretion and increase insulin resistance).
- Atypical antipsychotics (olanzapine, clozapine): 2-3x increased risk.
- Statins: Modest increased risk (9% over 4 years) but cardiovascular benefit far outweighs risk.
- Thiazide diuretics: Hypokalemia impairs insulin secretion.
3. Pathophysiology
The Evolution from Triumvirate to Ominous Octet
The classical model of T2DM pathophysiology—the "Triumvirate"—identified three core defects:
- Muscle insulin resistance (impaired glucose uptake).
- Hepatic insulin resistance (excessive glucose production).
- Pancreatic beta-cell failure (inadequate insulin secretion).
In 2009, Ralph DeFronzo expanded this to the "Ominous Octet," recognizing eight distinct organ systems contributing to hyperglycemia. [3] This paradigm shift has profound therapeutic implications, as it identifies multiple potential drug targets beyond insulin secretion and sensitization.
The Ominous Octet: Eight Pathophysiological Defects
1. Pancreatic Beta Cells: Impaired Insulin Secretion
Normal Physiology: Beta cells sense glucose via the GLUT2 transporter and secrete insulin in a biphasic pattern: a rapid first-phase spike (release of pre-formed granules) followed by a sustained second phase (de novo synthesis).
Pathophysiology in T2DM:
- Beta-cell mass declines by ~50% at diagnosis (autopsy studies).
- Glucotoxicity: Chronic hyperglycemia induces oxidative stress, ER stress, and apoptosis.
- Lipotoxicity: Excess free fatty acids (FFAs) accumulate as ceramides and diacylglycerols, impairing insulin secretion.
- Amyloid deposition: Islet amyloid polypeptide (IAPP/amylin) forms toxic aggregates in islets.
- First-phase insulin response is lost early, resulting in exaggerated postprandial hyperglycemia.
Clinical Implication: Beta-cell failure is progressive. By the time HbA1c reaches 53 mmol/mol (7.0%), beta-cell function is ~50% of normal. This explains why most patients eventually require combination therapy or insulin.
2. Pancreatic Alpha Cells: Hyperglucagonemia
Pathophysiology:
- Alpha cells become resistant to glucose-mediated suppression, leading to paradoxical glucagon secretion even in hyperglycemia.
- Glucagon stimulates hepatic gluconeogenesis and glycogenolysis, driving fasting hyperglycemia.
Therapeutic Target: GLP-1 receptor agonists suppress glucagon secretion.
3. Liver: Increased Hepatic Glucose Production (HGP)
Pathophysiology:
- Hepatic insulin resistance leads to failure to suppress gluconeogenesis after meals.
- Results in fasting hyperglycemia (≥7.0 mmol/L).
- Accounts for ~30% of the hyperglycemia in T2DM.
Therapeutic Target:
- Metformin inhibits mitochondrial complex I, reducing hepatic ATP production and gluconeogenesis.
- GLP-1 RAs suppress HGP indirectly via glucagon suppression.
4. Muscle: Insulin Resistance & Impaired Glucose Uptake
Pathophysiology:
- Skeletal muscle accounts for 70-80% of postprandial glucose disposal.
- Insulin resistance at the level of the insulin receptor substrate-1 (IRS-1) and impaired GLUT4 translocation to the cell membrane.
- Mechanisms:
- Intramyocellular lipid accumulation (diacylglycerols, ceramides).
- "Mitochondrial dysfunction: Reduced oxidative phosphorylation."
- "Inflammation: TNF-α and IL-6 activate serine kinases that phosphorylate IRS-1, blocking insulin signaling."
Clinical Implication: Exercise increases GLUT4 translocation independent of insulin (AMP-kinase pathway), which is why physical activity is so effective.
5. Adipose Tissue: Increased Lipolysis
Pathophysiology:
- Insulin normally suppresses hormone-sensitive lipase (HSL), preventing lipolysis.
- In T2DM, adipocyte insulin resistance leads to unrestrained lipolysis, releasing FFAs into the circulation.
- Elevated FFAs worsen insulin resistance in muscle and liver (lipotoxicity) and impair beta-cell function.
Clinical Implication: Visceral adiposity (not subcutaneous) is the key driver. Waist circumference is a better predictor of insulin resistance than BMI.
6. Gastrointestinal Tract: Incretin Deficiency/Resistance
Normal Physiology: GLP-1 and GIP (incretin hormones) are secreted by intestinal L-cells and K-cells in response to oral glucose. They:
- Stimulate glucose-dependent insulin secretion (incretin effect accounts for 50-70% of postprandial insulin).
- Suppress glucagon secretion.
- Slow gastric emptying.
Pathophysiology in T2DM:
- Reduced GLP-1 secretion.
- Beta cells become resistant to GLP-1/GIP.
- Incretin effect reduced to ~20% (vs 70% in normal subjects).
Therapeutic Target:
- GLP-1 receptor agonists (semaglutide, liraglutide): pharmacological doses bypass resistance.
- DPP-4 inhibitors (sitagliptin): prevent degradation of endogenous GLP-1.
7. Kidney: Increased Glucose Reabsorption
Pathophysiology:
- Normally, glucose is freely filtered at the glomerulus and 90% reabsorbed by SGLT2 transporters in the proximal tubule.
- In T2DM, SGLT2 is upregulated, increasing the renal threshold for glucose (from ~10 mmol/L to 13-15 mmol/L), worsening hyperglycemia.
Therapeutic Target:
- SGLT2 inhibitors (empagliflozin, dapagliflozin, canagliflozin) block reabsorption, causing glycosuria (~60-90g glucose/day excreted in urine).
- Additional benefits: osmotic diuresis (BP reduction), weight loss, natriuresis, reduced glomerular hyperfiltration.
8. Brain: Neurotransmitter Dysfunction & Insulin Resistance
Pathophysiology:
- Hypothalamic insulin resistance impairs satiety signaling, promoting hyperphagia and weight gain.
- Reduced CNS dopamine and serotonin activity may contribute to insulin resistance.
Therapeutic Potential:
- Investigational: bromocriptine (dopamine agonist) approved in USA for T2DM, though rarely used.
Inflammation & Metainflammation
Obesity-induced chronic low-grade inflammation ("metainflammation") is central to insulin resistance:
- Adipose tissue macrophages infiltrate visceral fat, polarizing from anti-inflammatory M2 to pro-inflammatory M1 phenotype.
- Pro-inflammatory cytokines (TNF-α, IL-6, IL-1β) activate serine kinases (JNK, IKK), which phosphorylate IRS-1 on serine residues, blocking insulin signaling.
- Adipokines: Leptin (promotes insulin resistance), resistin (pro-inflammatory), adiponectin (insulin-sensitizing, but reduced in obesity).
The Natural History: From Prediabetes to Overt Diabetes
Stage 1: Insulin Resistance (Compensated)
- Normal glucose.
- Hyperinsulinemia (fasting insulin 2-3x elevated).
- Beta cells compensate by increasing insulin secretion.
Stage 2: Prediabetes (Impaired Fasting Glucose / Impaired Glucose Tolerance)
- HbA1c 42-47 mmol/mol (6.0-6.4%).
- FPG 5.6-6.9 mmol/L.
- Beta cells begin to fail; first-phase insulin response lost.
Stage 3: Overt T2DM
- HbA1c ≥48 mmol/mol (6.5%).
- Beta-cell function ~50% of normal.
- Progressive decline in beta-cell mass and function (~4-5% per year).
Stage 4: Severe Insulin Deficiency
- May require insulin therapy.
- Rare: ketosis-prone diabetes (phenotypic overlap with T1DM).
4. Clinical Presentation
Classical Symptoms: The "Polys"
Polyuria (Excessive Urination)
- Mechanism: Once blood glucose exceeds renal threshold (~10 mmol/L), glucose appears in urine (glycosuria), causing osmotic diuresis.
- Patients report nocturia (waking 3-4 times/night to urinate), a highly disruptive symptom.
Polydipsia (Excessive Thirst)
- Compensatory response to dehydration from osmotic diuresis.
- Patients may drink 4-6 liters of fluid daily.
Polyphagia (Increased Hunger)
- Less common in T2DM than T1DM (due to residual insulin secretion).
- Due to cellular "starvation" despite hyperglycemia (glucose cannot enter cells).
Weight Loss (Paradoxical)
- Uncommon in typical T2DM (most patients are overweight/obese).
- If present, suggests severe insulin deficiency, malignancy, or undiagnosed T1DM/LADA.
- Red flag: weight loss > 10% warrants urgent investigation.
Blurred Vision
- Osmotic changes in the lens due to hyperglycemia alter refractive power.
- Reversible with glucose normalization (warn patients not to change glasses during acute hyperglycemia).
Fatigue & Lethargy
- Multifactorial: cellular energy deficit, sleep disruption (nocturia), chronic inflammation, depression.
Atypical & Silent Presentations
Recurrent Infections
- Genital candidiasis (thrush): Glucose in urine provides substrate for fungal growth. Often the presenting complaint in women.
- Urinary tract infections (UTIs): Increased susceptibility due to impaired neutrophil function.
- Skin infections: Boils, carbuncles, cellulitis.
Slow-Healing Wounds
- Impaired microcirculation, reduced neutrophil chemotaxis, and glycosylation of collagen.
Acanthosis Nigricans
- Hyperpigmented, velvety skin thickening in axillae, neck, groin.
- Classic sign of severe insulin resistance (also seen in PCOS, obesity, malignancy).
Erectile Dysfunction (ED)
- Affects 50-75% of men with diabetes.
- Multifactorial: autonomic neuropathy, vascular disease, hypogonadism, psychological factors.
Asymptomatic Hyperglycemia
- ~50% of patients have NO symptoms at diagnosis.
- Detected incidentally on routine screening (HbA1c, fasting glucose).
Signs on Physical Examination
General Inspection
- Obesity: Particularly central (android) adiposity. Waist circumference > 102 cm (men) or > 88 cm (women) indicates increased metabolic risk.
- Acanthosis nigricans: Neck, axillae, groin.
- Skin tags: Associated with insulin resistance.
- Xanthelasma/xanthomata: Suggest dyslipidemia.
Cardiovascular System
- Hypertension: Present in 60-80% of patients.
- Peripheral arterial disease: Weak or absent pedal pulses, cool extremities, bruits (femoral, carotid).
- Silent MI: May have no chest pain due to autonomic neuropathy (assess with ECG).
Neurological Examination (Diabetic Neuropathy)
- Peripheral Sensory Neuropathy: "Glove-and-stocking" distribution. Loss of vibration sense (128 Hz tuning fork), loss of protective sensation (10g monofilament test).
- Loss of Ankle Reflexes: Early sign.
- Charcot Neuroarthropathy: Warm, swollen, deformed foot (collapse of midfoot arch).
Fundoscopy (Diabetic Retinopathy)
- Annual screening mandatory.
- Findings: microaneurysms, dot-and-blot hemorrhages, hard exudates, cotton-wool spots, neovascularization (proliferative retinopathy).
The Diabetic Foot (Critical Examination)
- Inspection: Calluses, ulcers, Charcot deformity, fungal nail infections.
- Vascular: Palpate dorsalis pedis and posterior tibial pulses. Ankle-Brachial Pressure Index (ABPI) if PAD suspected.
- Neurological: 10g monofilament at 10 sites (hallux, metatarsal heads, heel). Loss of sensation = "foot at risk."
- Classification: Use Wagner or University of Texas Diabetic Foot Classification.
Abdominal Examination
- Hepatomegaly: Non-alcoholic fatty liver disease (NAFLD/MASLD) present in 70-80% of T2DM patients.
Red Flags: Acute Metabolic Emergencies
[!CAUTION] Hyperglycemic Hyperosmolar State (HHS)
- Presentation: Profound dehydration (fluid deficit 8-12L), altered mental status (GCS less than 15), severe hyperglycemia (often > 30 mmol/L), NO ketosis.
- Plasma osmolality: > 320 mOsm/kg (calculated: 2[Na+] + glucose + urea).
- Mortality: 10-20% (higher than DKA).
- Triggers: Infection (pneumonia, UTI), stroke, MI, non-adherence to medications, diuretics.
- Management: Aggressive IV fluid resuscitation (0.9% saline 1L/h initially), insulin infusion, treat precipitant. [12]
[!CAUTION] Euglycemic Diabetic Ketoacidosis (eDKA)
- Context: SGLT2 inhibitor use (empagliflozin, dapagliflozin, canagliflozin).
- Presentation: Ketosis (beta-hydroxybutyrate > 3 mmol/L) + acidosis (pH less than 7.3) but glucose often less than 14 mmol/L (hence "euglycemic").
- Triggers: Surgery, acute illness, fasting, low-carbohydrate/ketogenic diet, insulin omission.
- Management: STOP SGLT2i. IV dextrose (to suppress ketogenesis) + insulin infusion. [13]
- Prevention: Counsel patients on "sick day rules" (stop SGLT2i during illness/fasting).
[!CAUTION] Acute Visual Loss
- Vitreous hemorrhage (proliferative retinopathy).
- Retinal detachment.
- Central retinal vein occlusion.
- Action: Urgent ophthalmology referral (same-day).
[!CAUTION] Diabetic Foot Infection
- Cellulitis, abscess, osteomyelitis.
- Often painless due to neuropathy (patient may present late).
- Red flags: Foul odor, visible bone, crepitus (gas gangrene), systemic sepsis.
- Probing to bone test: If a sterile probe touches bone through an ulcer, osteomyelitis is highly likely.
- Management: Urgent surgical debridement, IV antibiotics (cover Staph, Strep, anaerobes), vascular assessment (revascularization if ischemic). [14]
[!CAUTION] Silent Myocardial Infarction
- Up to 30% of MIs in diabetics are painless (autonomic neuropathy).
- Atypical presentations: dyspnea, nausea, fatigue, syncope.
- Low threshold for ECG and troponin in diabetics with vague symptoms.
5. Investigations
Diagnostic Criteria (WHO/ADA 2023)
Diagnosis of diabetes requires ONE of the following (if asymptomatic, TWO abnormal tests on separate days required):
| Test | Normal | Prediabetes (High Risk) | Diabetes |
|---|---|---|---|
| HbA1c | less than 42 mmol/mol (less than 6.0%) | 42-47 mmol/mol (6.0-6.4%) | ≥48 mmol/mol (≥6.5%) |
| Fasting Plasma Glucose (FPG) | less than 5.6 mmol/L | 5.6-6.9 mmol/L (IFG) | ≥7.0 mmol/L |
| 2-Hour Oral Glucose Tolerance Test (OGTT) | less than 7.8 mmol/L | 7.8-11.0 mmol/L (IGT) | ≥11.1 mmol/L |
| Random Plasma Glucose | N/A | N/A | ≥11.1 mmol/L (with symptoms) |
Notes:
- HbA1c advantages: No fasting required, reflects 3-month average glucose, less day-to-day variability.
- HbA1c limitations: Falsely low in hemolysis, CKD (eGFR less than 30), recent transfusion, hemoglobinopathies (HbS, HbC). Use fructosamine or OGTT if unreliable.
- OGTT: 75g glucose load, measure glucose at 0 and 120 minutes. Gold standard but cumbersome.
Screening Recommendations
Universal Screening:
- All adults ≥45 years: Screen with HbA1c or FPG every 3 years.
- Earlier screening if risk factors present (obesity, family history, ethnicity, PCOS, GDM).
High-Risk Groups (Annual Screening):
- BMI ≥25 kg/m² (≥23 kg/m² in Asians) plus one risk factor.
- Prediabetes (HbA1c 42-47 mmol/mol).
- History of GDM.
- PCOS.
- HIV infection (on antiretroviral therapy).
- First-degree relative with diabetes.
Baseline Investigations (At Diagnosis)
| Investigation | Purpose | Frequency |
|---|---|---|
| HbA1c | Glycemic control | Every 3-6 months |
| Fasting Lipid Profile | CV risk (total cholesterol, LDL, HDL, triglycerides) | Annually |
| Urine Albumin:Creatinine Ratio (UACR) | Detect albuminuria (early nephropathy) | Annually |
| Serum Creatinine + eGFR | Assess renal function, stage CKD | Annually |
| Liver Function Tests | Screen for NAFLD/MASLD | Baseline, then as indicated |
| TSH | Screen for thyroid disease (10% prevalence) | Baseline, then every 2-3 years |
| 12-Lead ECG | Detect silent MI, LVH | Baseline |
| Digital Retinal Photography | Screen for retinopathy | Annually (can extend to 2-yearly if no retinopathy) |
| Foot Examination | Assess neuropathy, vascular disease | Annually (more frequent if foot at risk) |
Monitoring Investigations
HbA1c Monitoring:
- Target: 48-53 mmol/mol (6.5-7.0%) for most adults.
- Individualized targets:
- "Newly diagnosed, young, no CVD: 48 mmol/mol (6.5%)."
- "Elderly, frail, frequent hypoglycemia: 58-64 mmol/mol (7.5-8.0%)."
- "Pregnant (pre-existing diabetes): 41-47 mmol/mol (6.0-6.4%)."
Renal Monitoring:
- UACR:
- "Normal: less than 3 mg/mmol."
- "Microalbuminuria (A2): 3-30 mg/mmol."
- "Macroalbuminuria (A3): > 30 mg/mmol."
- eGFR (CKD stages):
- "G1: > 90 mL/min (normal)."
- "G2: 60-89 mL/min (mild)."
- G3 a: 45-59 mL/min (moderate).
- G3 b: 30-44 mL/min (moderate-severe).
- "G4: 15-29 mL/min (severe)."
- "G5: less than 15 mL/min (ESRD)."
Cardiovascular Risk Assessment:
- Lipid targets (on statin therapy):
- LDL less than 2.6 mmol/L (primary prevention).
- LDL less than 1.8 mmol/L (secondary prevention/10-year ASCVD risk > 20%).
- Blood Pressure:
- "Target: less than 130/80 mmHg (ADA), less than 140/90 mmHg (NICE)."
- Use ACE inhibitor or ARB first-line (especially if albuminuria).
Self-Monitoring of Blood Glucose (SMBG):
- Not routinely required for non-insulin-treated T2DM.
- Indicated if:
- On insulin or sulfonylurea (hypoglycemia risk).
- Pregnancy.
- Acute illness.
- Discordance between HbA1c and symptoms.
Continuous Glucose Monitoring (CGM):
- Increasingly used in T2DM on intensive insulin regimens.
- Provides Time in Range (TIR): target > 70% time in 3.9-10.0 mmol/L.
Special Investigations
Diabetes Autoantibody Panel (if suspected T1DM/LADA):
- Anti-GAD65, Anti-IA2, Anti-ZnT8.
- Positive in 5-10% of "T2DM" patients (latent autoimmune diabetes in adults, LADA).
C-Peptide:
- Distinguishes endogenous insulin secretion.
- Low/undetectable: suggests T1DM or severe T2DM.
Genetic Testing:
- If suspected MODY (young age, non-obese, strong family history, no autoantibodies).
6. Management
Overview: The Modern Treatment Paradigm
Management of T2DM has undergone a paradigm shift from a glucose-centric approach to a comprehensive cardio-renal protection strategy. The 2022 ADA/EASD Consensus Report emphasizes early use of SGLT2 inhibitors and GLP-1 receptor agonists in patients with established ASCVD, heart failure, or CKD, independent of baseline HbA1c. [6]
Core Principles:
- Lifestyle modification is foundational.
- Metformin remains first-line pharmacotherapy (unless contraindicated).
- SGLT2i or GLP-1 RA should be added early if ASCVD, HF, or CKD present.
- Individualized HbA1c targets based on age, comorbidities, hypoglycemia risk, life expectancy.
- Treat the whole patient: BP, lipids, antiplatelet therapy, smoking cessation, structured education.
- Annual screening for complications (retinopathy, nephropathy, neuropathy, foot disease).
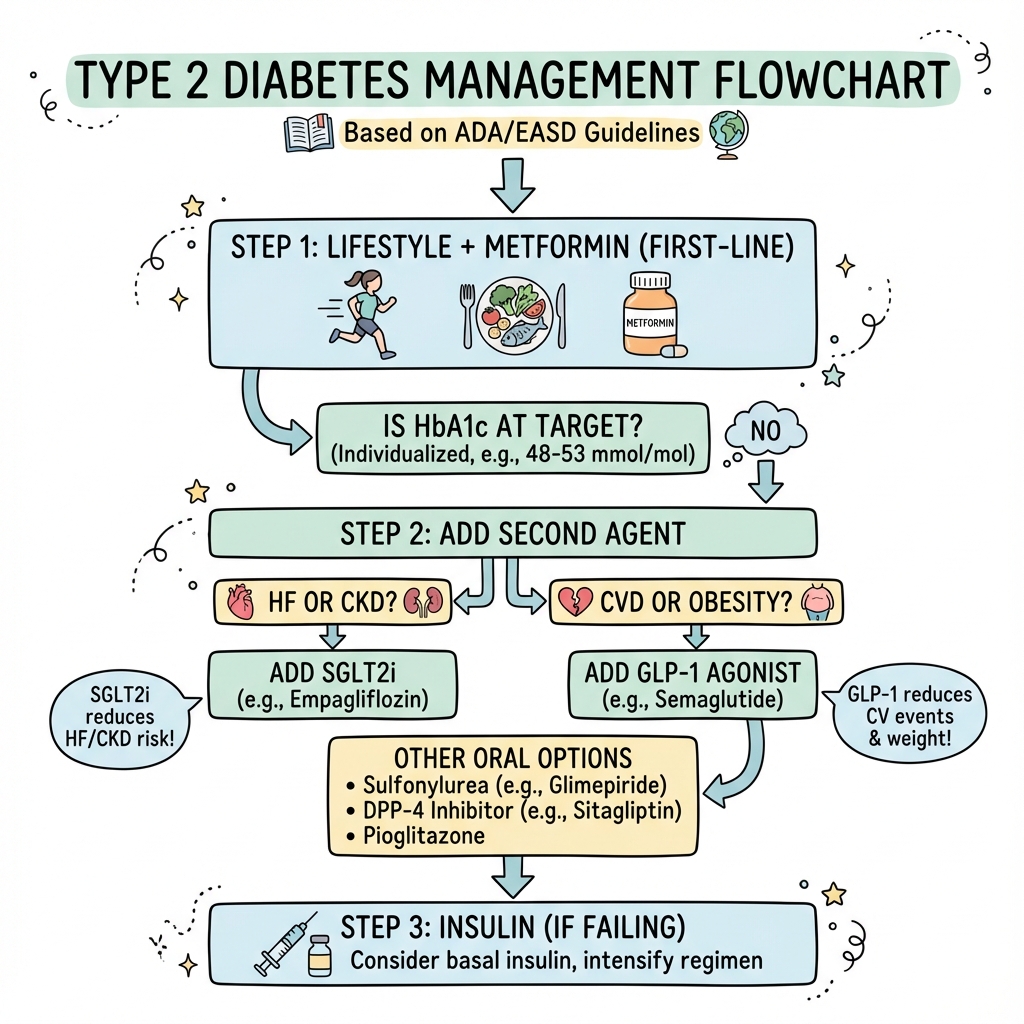
Management Algorithm
┌──────────────────────────────────────────────────────────┐
│ DIAGNOSIS OF TYPE 2 DIABETES │
│ (HbA1c ≥48 mmol/mol or FPG ≥7.0 mmol/L) │
└──────────────────────────────────────────────────────────┘
↓
┌──────────────────────────────────────────────────────────┐
│ STEP 1: LIFESTYLE MODIFICATION + METFORMIN │
│ ------------------------------------------------ │
│ • Diet: Mediterranean/low-GI, calorie deficit │
│ • Exercise: 150 min/week moderate intensity │
│ • Weight loss: Target 5-10% if overweight │
│ • Metformin: Start 500mg OD, ↑ to 1g BD (max 2g/day) │
│ • Structured education: DESMOND, X-PERT │
└──────────────────────────────────────────────────────────┘
↓
┌──────────────────────────────────────────────────────────┐
│ STEP 2: ASSESS CARDIO-RENAL RISK │
│ ------------------------------------------------ │
│ Does the patient have: │
│ • Established ASCVD (MI, stroke, PAD)? │
│ • Heart failure (HFrEF or HFpEF)? │
│ • CKD (eGFR less than 60 or UACR > 3 mg/mmol)? │
└──────────────────────────────────────────────────────────┘
↓
┌────────────────┴────────────────┐
↓ ↓
┌─────────────────────┐ ┌─────────────────────────┐
│ YES: High CV/Renal │ │ NO: Standard Risk │
│ Risk │ │ │
└─────────────────────┘ └─────────────────────────┘
↓ ↓
┌─────────────────────────────────────────────────────────┐
│ ADD SGLT2i (preferred if HF or CKD) │
│ • Empagliflozin 10mg OD │
│ • Dapagliflozin 10mg OD │
│ • Canagliflozin 100mg OD │
│ OR │
│ ADD GLP-1 RA (preferred if ASCVD or weight loss goal) │
│ • Semaglutide 0.5-1mg SC weekly │
│ • Dulaglutide 1.5mg SC weekly │
│ • Liraglutide 1.8mg SC daily │
│ │
│ Add REGARDLESS of current HbA1c │
└─────────────────────────────────────────────────────────┘
↓
┌──────────────────────────────────────────────────────────┐
│ REASSESS HbA1c AT 3-6 MONTHS │
│ • If HbA1c ABOVE target → Add 3rd agent │
│ • If HbA1c AT target → Continue, monitor │
└──────────────────────────────────────────────────────────┘
↓
┌──────────────────────────────────────────────────────────┐
│ STEP 3: TRIPLE THERAPY (If HbA1c above target) │
│ ------------------------------------------------ │
│ Add ONE of: │
│ • DPP-4 inhibitor (sitagliptin 100mg OD) │
│ • Pioglitazone 15-30mg OD (avoid if HF) │
│ • Sulfonylurea (gliclazide 40-160mg BD) - hypo risk │
│ • Basal insulin (glargine, degludec 10U at bedtime) │
└──────────────────────────────────────────────────────────┘
↓
┌──────────────────────────────────────────────────────────┐
│ STEP 4: INTENSIFICATION (If HbA1c still above target) │
│ ------------------------------------------------ │
│ • Basal-bolus insulin regimen │
│ • Dual GLP-1/GIP agonist (tirzepatide) │
│ • Consider referral to endocrinology │
└──────────────────────────────────────────────────────────┘
Lifestyle Modification (Foundation of All Therapy)
Dietary Interventions:
- Mediterranean diet: High in olive oil, nuts, fish, vegetables, whole grains. Reduces HbA1c by 0.3-0.5% (3-5 mmol/mol).
- Low Glycemic Index (GI) diet: Reduces postprandial glucose spikes.
- Calorie restriction: 500-750 kcal/day deficit achieves 5-10% weight loss over 6 months.
- Very Low-Calorie Diet (VLCD): 800-850 kcal/day (meal replacements). DiRECT trial: 46% remission rate at 12 months with ≥15 kg weight loss. [9]
- Carbohydrate counting: Useful if on mealtime insulin.
- Avoid: Sugar-sweetened beverages, ultra-processed foods, refined carbohydrates.
Physical Activity:
- Aerobic exercise: 150 min/week moderate intensity (brisk walking, cycling) OR 75 min/week vigorous.
- Resistance training: 2-3 sessions/week (improves insulin sensitivity via increased muscle mass).
- High-Intensity Interval Training (HIIT): Effective for improving glycemic control and cardiovascular fitness.
- Mechanism: Increases GLUT4 translocation (insulin-independent), improves mitochondrial function, reduces visceral fat.
Weight Loss:
- 5-10% weight loss: Reduces HbA1c by 0.5-1.0% (5-10 mmol/mol), improves BP, lipids.
- ≥15 kg weight loss: 46% achieve remission (HbA1c less than 48 mmol/mol off medications). [9]
Smoking Cessation:
- Reduces microvascular and macrovascular complications.
- Offer nicotine replacement therapy, varenicline, or bupropion.
Structured Education Programs:
- DESMOND (Diabetes Education and Self-Management for Ongoing and Newly Diagnosed): UK group-based program.
- DAFNE (Dose Adjustment for Normal Eating): For T1DM/insulin-treated T2DM.
- X-PERT: Patient empowerment program.
Pharmacotherapy: Drug Classes
1. Biguanide: Metformin (First-Line)
Mechanism of Action:
- Inhibits mitochondrial complex I in hepatocytes → reduces hepatic ATP → activates AMP-kinase → suppresses gluconeogenesis.
- Modest improvement in peripheral insulin sensitivity (muscle, adipose).
Efficacy:
- Reduces HbA1c by 1.0-1.5% (10-15 mmol/mol).
- Weight neutral or modest weight loss (~1-2 kg).
Dosing:
- Start: 500mg OD with evening meal.
- Titrate: Increase by 500mg weekly to 1g BD (max 2g/day).
- Modified-release (MR) formulation available if GI side effects.
Side Effects:
- GI: Nausea, diarrhea, abdominal discomfort (20-30% of patients). Usually transient. Take with food.
- Lactic acidosis: Rare (less than 10 per 100,000 patient-years) but serious. Risk factors: renal impairment, hepatic failure, severe hypoxia, sepsis.
- Vitamin B12 deficiency: 10-30% of long-term users. Check B12 annually if on metformin > 4 years.
Contraindications:
- eGFR less than 30 mL/min (absolute).
- eGFR 30-44 mL/min: Reduce dose to 500mg BD.
- Acute kidney injury, severe hepatic impairment, severe hypoxia (e.g., acute MI, septic shock).
- Withhold during acute illness with dehydration ("sick day rules").
Evidence:
- UKPDS: Metformin reduced macrovascular events by 30% vs diet alone. [15]
Cost:
- Generic, inexpensive (~£1-2/month).
2. SGLT2 Inhibitors (Gliflozins): Empagliflozin, Dapagliflozin, Canagliflozin
Mechanism of Action:
- Inhibit sodium-glucose co-transporter 2 (SGLT2) in proximal tubule → block 90% of glucose reabsorption → glycosuria (60-90g glucose/day excreted).
- Pleiotropic effects (beyond glucose lowering):
- "Osmotic diuresis: Reduces preload (↓ HF hospitalizations)."
- "Natriuresis: Lowers BP by 3-5 mmHg."
- Reduces intraglomerular pressure (↓ hyperfiltration → renal protection).
- Weight loss (~2-3 kg).
- Reduces uric acid (gout protection).
Efficacy:
- Reduces HbA1c by 0.5-1.0% (5-10 mmol/mol).
Cardiovascular & Renal Benefits (LANDMARK EVIDENCE):
- EMPA-REG OUTCOME (empagliflozin): [7]
- 38% reduction in CV death.
- 35% reduction in HF hospitalization.
- 32% reduction in all-cause mortality.
- Effect seen within 3 months (too early for glucose effect → direct cardio-renal mechanisms).
- CREDENCE (canagliflozin): [10]
- 30% reduction in composite renal outcome (ESRD, doubling of creatinine, renal/CV death).
- 40% reduction in ESRD.
- DAPA-CKD (dapagliflozin): [11]
- 39% reduction in CKD progression or CV death.
- Benefit even in eGFR 25-75 mL/min.
- Meta-analysis: SGLT2i reduce MACE by 11%, HF hospitalization by 31%, CKD progression by 40%. [16]
Indications (Beyond Glucose Lowering):
- Established ASCVD (MI, stroke, PAD).
- Heart failure (HFrEF or HFpEF).
- CKD (eGFR 20-75 mL/min, UACR > 3 mg/mmol).
Dosing:
- Empagliflozin: 10mg OD (can increase to 25mg).
- Dapagliflozin: 10mg OD.
- Canagliflozin: 100mg OD (can increase to 300mg if eGFR > 60).
Side Effects:
- Genital candidiasis: 10-15% (more common in women). Treat with topical antifungals. Usually resolves without stopping drug.
- UTIs: 5-10%.
- Volume depletion: Especially if on loop diuretics or elderly. Monitor BP.
- Euglycemic DKA: Rare (1-2 per 1,000 patient-years) but serious. [13]
- "Risk factors: Insulin deficiency, fasting, surgery, low-carb diet."
- "Prevention: STOP SGLT2i 3 days before elective surgery or if vomiting/fasting."
- Amputations: Increased risk with canagliflozin in CANVAS trial (controversial; not seen with empagliflozin/dapagliflozin). Avoid in patients with active foot ulcers.
- Fournier's gangrene: Extremely rare necrotizing fasciitis of perineum.
Contraindications:
- eGFR less than 20 mL/min (exception: dapagliflozin in HF trials used down to eGFR 25).
- T1DM (off-label use requires specialist supervision due to DKA risk).
Sick Day Rules (Critical Patient Education):
[!WARNING] STOP SGLT2 inhibitors during:
- Vomiting, diarrhea, severe illness (dehydration risk).
- Fasting or very low-carb diet.
- Surgery (stop 3 days before elective surgery). Reason: Prevent euglycemic DKA and volume depletion.
3. GLP-1 Receptor Agonists (Incretin Mimetics): Semaglutide, Liraglutide, Dulaglutide
Mechanism of Action:
- Mimic endogenous GLP-1 (resistant to DPP-4 degradation).
- Glucose-dependent insulin secretion (low hypo risk).
- Suppress glucagon secretion.
- Slow gastric emptying (reduces postprandial glucose spikes).
- Central appetite suppression (↓ calorie intake).
Efficacy:
- Reduces HbA1c by 1.0-1.5% (10-15 mmol/mol).
- Weight loss: 3-6 kg (liraglutide, dulaglutide), up to 10-15 kg (semaglutide 2.4mg, tirzepatide).
Cardiovascular Benefits (LANDMARK EVIDENCE):
- LEADER (liraglutide): [8]
- 13% reduction in MACE (CV death, MI, stroke).
- 22% reduction in CV death.
- SUSTAIN-6 (semaglutide): 26% reduction in MACE.
- Meta-analysis: GLP-1 RA reduce MACE by 14%, CV death by 12%. [17]
Indications:
- Established ASCVD (MI, stroke, PAD).
- Need for significant weight loss (obesity + T2DM).
- Second-line add-on if metformin insufficient.
Dosing (Subcutaneous Injection):
- Semaglutide: Start 0.25mg SC weekly, ↑ to 0.5mg at 4 weeks, then 1mg (max 2mg for weight loss).
- Dulaglutide: 0.75mg SC weekly, ↑ to 1.5mg.
- Liraglutide: Start 0.6mg SC daily, ↑ to 1.2mg, then 1.8mg (max 3.0mg for weight loss).
- Exenatide: 5-10µg SC BD (older agent, less commonly used).
Oral Formulation:
- Oral semaglutide (Rybelsus): 7-14mg OD. Less effective than SC but convenient.
Side Effects:
- GI: Nausea (20-40%), vomiting, diarrhea, constipation. Dose-dependent. Usually transient (2-4 weeks). Slow titration reduces GI side effects.
- Pancreatitis: Rare (slightly increased risk vs placebo). Avoid if history of pancreatitis.
- Gallstones/cholecystitis: Risk with rapid weight loss.
- Injection site reactions: Rare.
- Hypoglycemia: Low risk when used alone; increased if combined with sulfonylurea or insulin.
Contraindications:
- Personal or family history of medullary thyroid carcinoma (MTC) or MEN 2 syndrome (black box warning in USA; theoretical risk from rodent studies, not seen in humans).
- History of pancreatitis (relative contraindication).
Patient Education:
- Injection technique (rotate sites: abdomen, thigh, upper arm).
- Nausea is common initially but usually improves.
4. DPP-4 Inhibitors (Gliptins): Sitagliptin, Linagliptin, Saxagliptin
Mechanism of Action:
- Inhibit dipeptidyl peptidase-4 (DPP-4), which degrades GLP-1 and GIP → prolongs action of endogenous incretins.
- Glucose-dependent insulin secretion, glucagon suppression.
Efficacy:
- Reduces HbA1c by 0.5-0.8% (5-8 mmol/mol).
- Weight neutral.
- Low hypoglycemia risk.
Dosing:
- Sitagliptin: 100mg OD (50mg if eGFR 30-50, 25mg if eGFR less than 30).
- Linagliptin: 5mg OD (no dose adjustment in CKD).
Side Effects:
- Well tolerated (fewest side effects of all glucose-lowering drugs).
- Pancreatitis: Rare.
- Joint pain (arthralgia): Rare but reported.
- Bullous pemphigoid: Rare skin reaction.
Cardiovascular Outcomes:
- Neutral: SAVOR-TIMI (saxagliptin) and EXAMINE (alogliptin) showed no CV benefit or harm (except saxagliptin increased HF hospitalizations).
Clinical Use:
- Third-line agent (after metformin + SGLT2i/GLP-1 RA).
- Useful in elderly (well-tolerated, low hypo risk).
- Cannot combine with GLP-1 RA (redundant mechanisms).
5. Sulfonylureas: Gliclazide, Glimepiride, Glipizide
Mechanism of Action:
- Bind to SUR1 receptor on beta-cell KATP channel → closes channel → depolarization → Ca²⁺ influx → insulin release.
- Insulin secretion is glucose-independent (hence hypoglycemia risk).
Efficacy:
- Reduces HbA1c by 1.0-1.5% (10-15 mmol/mol).
- Weight gain: 2-3 kg (due to anabolic effects of insulin).
Dosing:
- Gliclazide: 40-160mg BD (MR formulation: 30-120mg OD).
- Glimepiride: 1-4mg OD.
Side Effects:
- Hypoglycemia: 10-20% of patients (especially elderly, CKD, erratic meals).
- Weight gain: 2-3 kg.
- Cardiovascular safety: Controversial. UKPDS showed no harm, but some observational studies suggest increased CV events (compared to metformin).
Contraindications:
- Severe hepatic or renal impairment.
- High hypoglycemia risk (elderly, living alone, impaired awareness).
Clinical Use:
- Third-line (after metformin + SGLT2i/GLP-1 RA).
- Cheap, effective, but declining use due to hypo risk and weight gain.
6. Thiazolidinediones (TZDs): Pioglitazone
Mechanism of Action:
- PPAR-γ agonist → increases insulin sensitivity in muscle and adipose tissue.
- Reduces hepatic glucose production.
Efficacy:
- Reduces HbA1c by 0.8-1.2% (8-12 mmol/mol).
- Preserves beta-cell function (unique among oral agents).
Dosing:
- Pioglitazone: 15-30mg OD (max 45mg).
Side Effects:
- Weight gain: 3-5 kg (fluid retention + increased subcutaneous fat).
- Fluid retention/edema: Contraindicated in heart failure (precipitates acute decompensation).
- Fractures: Increased risk in postmenopausal women (osteoporosis).
- Bladder cancer: Slightly increased risk (controversial; avoid if history of bladder cancer).
Contraindications:
- Heart failure (NYHA class III-IV).
- Active bladder cancer.
Clinical Use:
- Third-line (limited use due to side effects).
- May be useful if severe insulin resistance and cannot tolerate other agents.
7. Insulin Therapy
Indications:
- Failure of oral agents + GLP-1 RA to achieve HbA1c target.
- Severe hyperglycemia at diagnosis (HbA1c > 75 mmol/mol or symptomatic).
- Acute illness, surgery, pregnancy.
- Pancreatic insufficiency (chronic pancreatitis, post-pancreatectomy).
Insulin Regimens:
| Regimen | Description | Use Case |
|---|---|---|
| Basal insulin only | Once-daily long-acting insulin (glargine, degludec, detemir). Metformin + SGLT2i/GLP-1 RA continued. | First step in insulin initiation. Targets fasting glucose. |
| Basal-plus | Basal insulin + ONE mealtime rapid-acting insulin (e.g., before largest meal). | If postprandial glucose high despite basal. |
| Basal-bolus | Basal insulin + rapid-acting insulin before each meal (QDS regimen). | Intensive control. Mimics physiological insulin secretion. |
| Premixed insulin | Fixed combination of rapid + intermediate (e.g., 70/30, 75/25). BD dosing. | Simplified regimen for patients unable to manage basal-bolus. Less flexibility. |
Initiation of Basal Insulin:
- Start: Glargine U100 or degludec 10 units SC at bedtime.
- Titrate: Increase by 2 units every 3 days until fasting glucose 4.0-7.0 mmol/L.
- Typical maintenance dose: 0.3-0.5 units/kg/day.
Types of Insulin:
| Type | Onset | Peak | Duration | Examples |
|---|---|---|---|---|
| Rapid-acting | 10-15 min | 1-2h | 3-5h | Lispro, aspart, glulisine |
| Short-acting | 30 min | 2-4h | 6-8h | Regular (human insulin) |
| Intermediate-acting | 1-2h | 4-8h | 12-18h | NPH (isophane) |
| Long-acting | 1-2h | Minimal/none | 18-24h | Glargine U100, detemir |
| Ultra-long-acting | 1h | Minimal/none | > 40h | Degludec U100/U200, glargine U300 |
Side Effects of Insulin:
- Hypoglycemia: Most common. Educate on recognition and treatment (15g fast carbs, recheck in 15 min).
- Weight gain: 2-4 kg (anabolic hormone).
- Lipohypertrophy: Rotate injection sites.
Patient Education:
- Injection technique.
- Hypoglycemia recognition and treatment.
- Sick day rules (do NOT stop basal insulin; adjust doses if eating less).
- DVLA notification (UK: must inform if on insulin; can drive if hypo awareness intact, no severe hypos in last 12 months, monitors before driving).
8. Dual GIP/GLP-1 Receptor Agonist: Tirzepatide
Mechanism of Action:
- Dual agonist of GLP-1 and GIP receptors.
- Superior glucose lowering and weight loss compared to GLP-1 RA alone.
Efficacy:
- Reduces HbA1c by 2.0-2.5% (20-25 mmol/mol).
- Weight loss: 10-15 kg (highest of any glucose-lowering drug).
Dosing:
- Start 2.5mg SC weekly, titrate to 5mg, 10mg, 15mg (max).
Evidence:
- SURPASS trials: Superior to semaglutide for HbA1c and weight loss.
Availability:
- Licensed in USA, EU, UK (2022-2023). Expensive.
Cardiovascular Risk Management (As Important as Glucose Control)
Lipid Management
Statin Therapy (Mandatory for Most Patients):
- Primary prevention (no ASCVD):
- "Age 40-75 + diabetes: Moderate-intensity statin (atorvastatin 20mg or rosuvastatin 10mg)."
- "Age > 75: Consider statin if life expectancy > 5 years."
- Secondary prevention (established ASCVD):
- High-intensity statin (atorvastatin 80mg or rosuvastatin 20-40mg).
- "Target: LDL less than 1.8 mmol/L."
Ezetimibe:
- Add if LDL not at target on maximally tolerated statin.
PCSK9 Inhibitors (Alirocumab, Evolocumab):
- If LDL > 3.0 mmol/L on statin + ezetimibe and high CV risk.
Evidence:
- CTT meta-analysis: Statins reduce MACE by 20% per 1 mmol/L LDL reduction. [18]
Blood Pressure Management
Target BP:
- less than 130/80 mmHg (ADA).
- less than 140/90 mmHg (NICE, ESC for age > 65).
First-Line Antihypertensives:
- ACE inhibitor (ramipril, perindopril) or ARB (losartan, candesartan).
- Preferred if albuminuria present (renoprotective).
- Calcium channel blocker (amlodipine) if ACE-I/ARB contraindicated.
- Thiazide-like diuretic (indapamide) can be added.
Avoid:
- Beta-blockers (mask hypoglycemia symptoms, worsen insulin resistance) unless specific indication (MI, HF).
Evidence:
- UKPDS: Tight BP control (less than 150/85 mmHg) reduced stroke by 44%, diabetes-related death by 32%. [19]
Antiplatelet Therapy
Aspirin:
- Secondary prevention (post-MI, stroke, PAD): Aspirin 75-100mg OD (indefinite).
- Primary prevention: Controversial. Consider if 10-year ASCVD risk > 10% and low bleeding risk.
Dual Antiplatelet Therapy (DAPT):
- Aspirin + clopidogrel for 12 months post-ACS or PCI.
Glycemic Targets: Individualization is Key
| Patient Profile | HbA1c Target | Rationale |
|---|---|---|
| Newly diagnosed, young (less than 60), no CVD, no comorbidities | 48-53 mmol/mol (6.5-7.0%) | Maximize microvascular prevention. Low hypo risk. Long life expectancy. |
| Established CVD, multiple comorbidities, hypo risk | 53-58 mmol/mol (7.0-7.5%) | Balance benefit vs hypo risk. |
| Elderly (> 75), frail, limited life expectancy (less than 10 years) | 58-64 mmol/mol (7.5-8.0%) | Avoid hypoglycemia (falls, cognitive impairment). |
| Pregnancy (pre-existing diabetes) | 41-47 mmol/mol (6.0-6.4%) | Prevent fetal complications. |
| End-stage CKD, dialysis | Avoid HbA1c (unreliable) | Use fructosamine or CGM. |
ACCORD Trial Caution:
- Intensive glucose control (HbA1c less than 42 mmol/mol) in high-risk older patients with established CVD increased all-cause mortality by 22%. [20]
- Lesson: Avoid overly tight control in vulnerable populations (elderly, long diabetes duration, CVD).
7. Complications
Microvascular Complications
Diabetic Retinopathy
Epidemiology:
- Leading cause of blindness in working-age adults (20-74 years).
- Prevalence: 35% of diabetics have retinopathy; 7% have vision-threatening disease.
Pathophysiology:
- Chronic hyperglycemia → pericyte loss, basement membrane thickening → capillary microaneurysms, hemorrhages.
- Vascular endothelial growth factor (VEGF) release → neovascularization (proliferative retinopathy).
Classification:
| Stage | Findings | Management |
|---|---|---|
| No retinopathy | Normal fundus | Screen annually (can extend to 2-yearly if multiple normal screens). |
| Mild NPDR | Microaneurysms only | Screen annually. Optimize glucose, BP. |
| Moderate NPDR | Microaneurysms + dot-and-blot hemorrhages + hard exudates | Screen 6-monthly. Refer if maculopathy. |
| Severe NPDR | 4-2-1 rule: Hemorrhages in 4 quadrants, venous beading in 2 quadrants, IRMA in 1 quadrant | Refer to ophthalmology (high risk of progression). |
| Proliferative DR (PDR) | Neovascularization (new vessels on disc or elsewhere) | Urgent ophthalmology referral. Pan-retinal photocoagulation (PRP) or anti-VEGF (ranibizumab, aflibercept). |
| Maculopathy | Hard exudates within 1 disc diameter of fovea, macular edema | Urgent referral. Anti-VEGF injections. |
Screening:
- Annual digital retinal photography (mydriatic or non-mydriatic).
- Gold standard: 7-field stereoscopic fundus photography.
Prevention:
- Tight glycemic control (UKPDS: 25% reduction in microvascular complications per 1% HbA1c reduction). [15]
- BP control: UKPDS showed tight BP control reduced retinopathy progression by 34%. [19]
Treatment:
- Laser photocoagulation: PRP for proliferative retinopathy.
- Anti-VEGF injections: Intravitreal ranibizumab, aflibercept for macular edema.
- Vitrectomy: If vitreous hemorrhage or tractional retinal detachment.
Diabetic Nephropathy (Diabetic Kidney Disease)
Epidemiology:
- Leading cause of end-stage renal disease (ESRD) in developed countries.
- 20-40% of diabetics develop nephropathy.
Pathophysiology:
- Glomerular hyperfiltration (RAAS activation, VEGF) → glomerular hypertrophy → mesangial expansion → glomerulosclerosis (Kimmelstiel-Wilson nodules).
- Tubular injury, interstitial fibrosis.
Natural History:
- Hyperfiltration (eGFR > 120 mL/min).
- Microalbuminuria (UACR 3-30 mg/mmol).
- Macroalbuminuria (UACR > 30 mg/mmol).
- Declining eGFR (CKD stage 3-5).
- ESRD (eGFR less than 15 mL/min).
Diagnosis:
- UACR (urine albumin:creatinine ratio): First morning void or random sample.
- "Normal: less than 3 mg/mmol."
- "Microalbuminuria (A2): 3-30 mg/mmol."
- "Macroalbuminuria (A3): > 30 mg/mmol."
- eGFR: CKD-EPI equation.
Screening:
- Annual UACR + serum creatinine (eGFR) from diagnosis.
Management:
- RAAS inhibition: ACE inhibitor or ARB (first-line). Reduces albuminuria by 30-50%, slows CKD progression.
- Start ramipril 2.5mg OD or losartan 50mg OD.
- Titrate to max tolerated dose.
- Monitor K⁺ and creatinine at 1-2 weeks (acceptable to see eGFR drop by 10-20%).
- SGLT2 inhibitor: Reduces CKD progression by 40%. [10,11]
- "CREDENCE: Canagliflozin reduced composite renal outcome by 30%."
- "DAPA-CKD: Dapagliflozin reduced CKD progression by 39%."
- "Mechanism: Reduces intraglomerular pressure (tubuloglomerular feedback via SGLT2 inhibition)."
- Finerenone (non-steroidal mineralocorticoid receptor antagonist): FIDELIO-DKD trial showed 18% reduction in CKD progression. [21]
- BP control: Target less than 130/80 mmHg.
- Glycemic control: HbA1c less than 53 mmol/mol.
- Dietary protein restriction: 0.8 g/kg/day if eGFR less than 60 mL/min.
Referral to Nephrology:
- eGFR less than 30 mL/min.
- Rapid decline in eGFR (> 5 mL/min/year or > 10 mL/min over 5 years).
- Macroalbuminuria despite RAAS inhibition.
- Difficult BP control.
Renal Replacement Therapy:
- Hemodialysis, peritoneal dialysis, or renal transplantation if eGFR less than 10 mL/min or symptomatic uremia.
Diabetic Neuropathy
Epidemiology:
- 50% of diabetics develop neuropathy within 10 years.
- Leading cause of non-traumatic lower-limb amputation.
Types:
1. Distal Symmetric Polyneuropathy (DSPN) — "Glove-and-Stocking"
Pathophysiology:
- Microvascular ischemia of vasa nervorum + direct glucose toxicity (polyol pathway, oxidative stress) → axonal degeneration.
Clinical Features:
- Sensory symptoms: Burning, tingling, "pins and needles," pain (worse at night).
- Sensory signs: Loss of vibration sense (128 Hz tuning fork), loss of pinprick, loss of light touch (10g monofilament), loss of temperature discrimination.
- Motor signs: Weakness, wasting of intrinsic foot muscles (late).
- Loss of ankle reflexes (early sign).
Complications:
- Painless foot ulcers (loss of protective sensation).
- Charcot neuroarthropathy.
Diagnosis:
- Clinical (10g monofilament test at 10 sites: hallux, 5 metatarsal heads, heel).
- Nerve conduction studies (if atypical features).
Management:
- Optimize glucose control: Slows progression but does not reverse established neuropathy.
- Neuropathic pain:
- "First-line: Duloxetine 60mg OD or pregabalin 75-300mg BD or gabapentin 900-3600mg/day (in divided doses)."
- "Second-line: Amitriptyline 10-75mg at night (avoid in elderly due to anticholinergic effects)."
- "Topical: Capsaicin cream 0.075% (depletes substance P)."
- "Severe refractory pain: Tramadol (avoid long-term opioids)."
2. Autonomic Neuropathy
Clinical Features:
| System | Manifestations |
|---|---|
| Cardiovascular | Resting tachycardia (> 100 bpm), postural hypotension (drop > 20 mmHg systolic on standing), loss of heart rate variability, sudden cardiac death. |
| Gastrointestinal | Gastroparesis (nausea, vomiting, early satiety, erratic glucose control), constipation, diarrhea (nocturnal). |
| Genitourinary | Erectile dysfunction (50-75% of men), retrograde ejaculation, neurogenic bladder (urinary retention, overflow incontination, recurrent UTIs). |
| Sudomotor | Anhidrosis (loss of sweating in feet), gustatory sweating (facial sweating while eating). |
| Hypoglycemia unawareness | Loss of adrenergic symptoms (sweating, tremor) → severe hypoglycemia without warning. |
Management:
- Gastroparesis: Metoclopramide, domperidone, erythromycin (prokinetics). Small frequent meals.
- Postural hypotension: Fludrocortisone, midodrine, compression stockings. Rise slowly from lying/sitting.
- Erectile dysfunction: PDE5 inhibitors (sildenafil, tadalafil).
3. Focal/Multifocal Neuropathy
Mononeuropathy:
- Cranial nerve palsy (III, VI, VII): Sudden diplopia, ptosis, facial weakness.
- Median nerve (carpal tunnel syndrome).
- Ulnar, peroneal nerve palsies.
Diabetic Amyotrophy (Proximal Motor Neuropathy):
- Severe proximal muscle weakness (thigh), wasting, pain.
- Usually self-limiting over 6-12 months.
Macrovascular Complications
Coronary Artery Disease (CAD)
Epidemiology:
- Diabetes is a "coronary artery disease risk equivalent" (10-year risk of MI = 20%, same as non-diabetic with prior MI).
- 2-4x increased risk of MI.
- Leading cause of death in T2DM (50% of diabetics die from CAD).
Pathophysiology:
- Accelerated atherosclerosis: Dyslipidemia (high TG, low HDL, small dense LDL), endothelial dysfunction, inflammation, platelet hyperactivity, oxidative stress.
Clinical Features:
- Silent MI: 30% of MIs in diabetics are painless (autonomic neuropathy).
- Atypical symptoms: Dyspnea, nausea, fatigue.
Screening:
- ECG at baseline, then if symptomatic.
- Stress test or CT coronary angiography if high pre-test probability.
Management:
- Lifestyle: Smoking cessation, exercise, diet.
- Statin: High-intensity (atorvastatin 80mg).
- Antiplatelet: Aspirin 75mg (secondary prevention).
- ACE inhibitor/ARB.
- Beta-blocker post-MI.
- Revascularization: PCI or CABG if indicated (CABG preferred for multivessel disease in diabetics).
Cerebrovascular Disease (Stroke)
Epidemiology:
- 2-4x increased risk of ischemic stroke.
- Worse outcomes post-stroke (larger infarcts, higher mortality).
Management:
- Primary prevention: BP control, statin, antiplatelet (if high risk).
- Secondary prevention: Aspirin + clopidogrel (first 21 days), then aspirin alone. Statin, ACE-I.
Peripheral Arterial Disease (PAD)
Epidemiology:
- 4x increased risk.
- Claudication, critical limb ischemia, amputation.
Diagnosis:
- Absent pedal pulses.
- Ankle-Brachial Pressure Index (ABPI) less than 0.9 (caution: may be falsely elevated > 1.3 if calcified vessels).
Management:
- Exercise therapy, smoking cessation.
- Antiplatelet (clopidogrel 75mg > aspirin).
- Statin.
- Revascularization if critical limb ischemia (angioplasty, bypass).
The Diabetic Foot (High-Yield for Exams)
Pathophysiology:
- Neuropathy (loss of protective sensation) + ischemia (PAD) = "neuro-ischemic foot."
Classification:
| Type | Features | Management |
|---|---|---|
| Neuropathic ulcer | Painless, plantar surface (pressure points), warm foot, palpable pulses, callus around ulcer. | Offloading (total contact cast, orthotic footwear), debridement. |
| Ischemic ulcer | Painful, margins of foot (toes, heel), cool foot, absent pulses, punched-out appearance. | Vascular assessment (ABPI, duplex, angiography), revascularization. |
| Neuro-ischemic | Features of both. | Multidisciplinary care. |
Charcot Neuroarthropathy:
- Acute: Warm, swollen, erythematous foot (mimics cellulitis/gout).
- Chronic: Midfoot collapse ("rocker-bottom" deformity).
- Diagnosis: X-ray (fractures, subluxations), MRI.
- Management: Offloading (total contact cast), bisphosphonates (investigational).
Diabetic Foot Infection:
- Mild: Superficial, less than 2 cm cellulitis.
- Moderate: > 2 cm cellulitis, deeper tissues involved.
- Severe: Systemic sepsis, gangrene, osteomyelitis.
- Microbiology: Staph aureus, Streptococci, anaerobes (Gram-negatives if chronic).
- Treatment:
- "Mild: Oral flucloxacillin or co-amoxiclav."
- "Moderate-severe: IV antibiotics (piperacillin-tazobactam or meropenem), surgical debridement."
- "Osteomyelitis: 6-12 weeks antibiotics ± surgical resection."
Amputation:
- Diabetes accounts for 50% of all non-traumatic lower-limb amputations.
- Prevention: Annual foot screening, patient education ("never walk barefoot"), podiatry referral if foot at risk.
8. Prognosis
Life Expectancy
- T2DM reduces life expectancy by 6-8 years (on average).
- Greater reduction if diagnosed at younger age (less than 40 years: 10-12 years lost).
- Cardiovascular disease accounts for 50% of deaths.
Remission
The DiRECT trial (2017-2019) challenged the dogma that T2DM is irreversible:
- 46% remission rate at 12 months (HbA1c less than 48 mmol/mol off all glucose-lowering medications) with intensive weight management (800-850 kcal/day meal replacement for 3-5 months, followed by structured food reintroduction).
- 86% remission if weight loss > 15 kg achieved.
- Remission associated with reduced intrapancreatic fat and restored beta-cell function. [9]
Bariatric Surgery:
- Roux-en-Y gastric bypass or sleeve gastrectomy.
- 60-80% remission at 2 years.
- Greater remission if diabetes duration less than 5 years, C-peptide detectable (residual beta-cell function).
Predictors of Remission:
- Weight loss > 15 kg.
- Diabetes duration less than 6 years.
- HbA1c less than 58 mmol/mol (7.5%) at baseline.
- Not on insulin.
Prognostic Factors
Poor Prognosis:
- Long diabetes duration (> 15 years).
- Poor glycemic control (HbA1c > 75 mmol/mol).
- Established macrovascular disease (prior MI, stroke).
- Albuminuria (strong predictor of CV and all-cause mortality).
- Smoking.
- Low socioeconomic status.
Good Prognosis:
- Early diagnosis and treatment (metabolic memory: early tight control reduces long-term complications even if control later relaxes).
- Achieve HbA1c less than 53 mmol/mol within first 2 years.
- No albuminuria.
- Non-smoker.
9. Patient Education & Self-Management
Structured Education Programs
- DESMOND (Diabetes Education and Self-Management for Ongoing and Newly Diagnosed): UK group-based program (6 hours over 1-2 sessions).
- X-PERT: Patient empowerment program.
- DAFNE (Dose Adjustment for Normal Eating): For insulin-treated patients.
Key Counseling Points
1. Self-Monitoring of Blood Glucose (SMBG):
- Not routinely required for non-insulin-treated T2DM.
- Recommended if:
- On insulin or sulfonylurea (hypo risk).
- Pregnant.
- Acute illness.
- Driving (DVLA requirement if on insulin).
2. Hypoglycemia Recognition & Treatment:
- Symptoms: Sweating, tremor, palpitations, hunger, confusion, irritability.
- Threshold: less than 3.9 mmol/L (mild), less than 3.0 mmol/L (severe).
- Treatment: "Rule of 15"
- 15g fast-acting carbohydrate (5 dextrose tablets, 150mL Lucozade, 4-5 jelly babies).
- Wait 15 minutes, recheck glucose.
- Repeat if still less than 4.0 mmol/L.
- Follow with long-acting carb (slice of bread, biscuits).
- Severe hypo (unconscious): IM glucagon 1mg (GlucaGen) or IV 10% dextrose 150-200mL.
3. Sick Day Rules (Critical):
[!WARNING] When to STOP medications during illness (vomiting, diarrhea, dehydration):
- STOP: Metformin (lactic acidosis risk), SGLT2 inhibitors (euglycemic DKA risk).
- CONTINUE: Insulin (do NOT stop; may need dose adjustment).
- Monitor glucose frequently (4-hourly).
- Seek medical help if unable to tolerate oral fluids, glucose > 15 mmol/L despite insulin, or ketones present.
4. Foot Care:
- Daily inspection (use mirror for soles).
- Never walk barefoot (risk of painless injury).
- Wash feet daily, dry carefully (especially between toes).
- Moisturize (but not between toes).
- Well-fitting shoes (avoid tight or rubbing shoes).
- See podiatrist for corns, calluses, nail trimming.
- Red flags: Ulcer, redness, swelling, pain → urgent medical review.
5. Dietary Advice:
- Low glycemic index (GI) carbs: Whole grains, legumes, vegetables.
- Portion control: Use "plate method" (½ vegetables, ¼ protein, ¼ carbs).
- Reduce sugar-sweetened beverages, processed foods.
- Mediterranean diet: Olive oil, nuts, fish, vegetables.
- Alcohol: Limit to 14 units/week. Avoid if on insulin/sulfonylurea (hypo risk).
6. Exercise:
- 150 min/week moderate intensity (brisk walking, cycling).
- Resistance training 2-3x/week.
- If on insulin/sulfonylurea: Check glucose before/after exercise, carry fast-acting carbs.
7. Driving (DVLA UK):
- Notify DVLA if on insulin or sulfonylurea (hypoglycemia risk).
- Must have hypoglycemia awareness.
- No severe hypos (requiring assistance) in last 12 months.
- Check glucose before driving (must be > 5.0 mmol/L).
8. Annual Reviews:
- HbA1c, UACR, eGFR, lipids, BP.
- Retinal screening.
- Foot examination.
- Medication review.
- Flu vaccination (annual), pneumococcal vaccination.
10. Evidence Base & Guidelines
Landmark Trials
1. UKPDS (UK Prospective Diabetes Study, 1998)
- Intensive glucose control (HbA1c 53 mmol/mol vs 63 mmol/mol) reduced microvascular complications by 25%.
- Metformin reduced macrovascular events by 30% in obese patients. [15]
2. ACCORD (Action to Control Cardiovascular Risk in Diabetes, 2008)
- Intensive glucose control (HbA1c less than 42 mmol/mol) in high-risk older patients increased mortality by 22%.
- Lesson: Avoid overly tight control in elderly/frail. [20]
3. EMPA-REG OUTCOME (Empagliflozin, 2015)
- 38% reduction in CV death, 35% reduction in HF hospitalization, 32% reduction in all-cause mortality.
- First trial to show SGLT2i cardio-renal benefits. [7]
4. LEADER (Liraglutide, 2016)
- 13% reduction in MACE, 22% reduction in CV death.
- Established GLP-1 RA as cardioprotective. [8]
5. CREDENCE (Canagliflozin, 2019)
- 30% reduction in composite renal outcome (ESRD, doubling creatinine, renal/CV death).
- First trial to show SGLT2i renal protection. [10]
6. DAPA-CKD (Dapagliflozin, 2020)
- 39% reduction in CKD progression or CV death.
- Benefit even at eGFR 25-75 mL/min. [11]
7. DiRECT (Diabetes Remission Clinical Trial, 2017)
- 46% remission with intensive weight loss (≥15 kg).
- Paradigm shift: T2DM can be reversed. [9]
Guidelines
1. ADA/EASD Consensus Report (2022):
- Emphasizes cardio-renal protection (SGLT2i, GLP-1 RA) as foundational therapy alongside metformin. [6]
2. NICE NG28 (2022):
- Type 2 diabetes in adults: management.
- Stepwise approach: Metformin → dual → triple therapy.
3. KDIGO 2022:
- Diabetes Management in CKD.
- SGLT2i and RAAS inhibition for all diabetics with CKD (eGFR ≥20 mL/min).
11. Examination Focus (High-Yield for MRCP, FRACP, USMLE, PLAB)
The Opening Statement (Clinical Examination Station)
"This is a 58-year-old gentleman with a 12-year history of Type 2 Diabetes Mellitus, currently managed with metformin, empagliflozin, and atorvastatin. On examination, there is evidence of peripheral sensory neuropathy with loss of vibration sense and absent ankle reflexes bilaterally. Fundoscopy reveals background diabetic retinopathy with scattered microaneurysms and hard exudates. Pedal pulses are palpable, but there is a 2 cm neuropathic ulcer over the first metatarsal head of the right foot with surrounding callus. My management would focus on optimizing glycemic control, intensifying cardiovascular risk reduction, and urgent podiatry referral for offloading and wound care to prevent progression to osteomyelitis or amputation."
Common Viva Questions
1. What are the diagnostic criteria for diabetes?
- HbA1c ≥48 mmol/mol (6.5%), FPG ≥7.0 mmol/L, 2h-OGTT ≥11.1 mmol/L, or random glucose ≥11.1 mmol/L (with symptoms).
- Two tests required if asymptomatic.
2. What is the "Ominous Octet"?
- Eight organ defects in T2DM: beta-cell failure, alpha-cell hyperglucagonemia, hepatic glucose overproduction, muscle insulin resistance, adipocyte lipolysis, incretin deficiency, renal glucose reabsorption, brain insulin resistance. (DeFronzo, 2009)
3. Why are SGLT2 inhibitors particularly useful in T2DM?
- Glucose lowering via glycosuria.
- Cardio-renal protection: 38% reduction in CV death, 35% reduction in HF hospitalization, 40% reduction in CKD progression.
- Mechanisms: Osmotic diuresis (↓ preload), natriuresis (↓ BP), reduced glomerular hyperfiltration.
- Independent of HbA1c lowering.
4. What are the sick day rules for SGLT2 inhibitors?
- STOP SGLT2i during vomiting, diarrhea, fasting, or surgery (3 days before elective surgery).
- Reason: Prevent euglycemic DKA and volume depletion.
- Continue monitoring glucose, stay hydrated.
5. How do you manage a diabetic foot ulcer?
- Assess: Neuropathic (painless, plantar, warm, pulses present) vs ischemic (painful, margins, cool, absent pulses).
- Offloading: Total contact cast, orthotic footwear.
- Debridement: Remove callus and necrotic tissue.
- Infection control: Swab for culture, antibiotics if infected (flucloxacillin, co-amoxiclav, or IV if severe).
- Vascular assessment: ABPI, duplex, angiography if ischemic. Consider revascularization.
- Glucose control: Optimize HbA1c.
- MDT: Podiatry, vascular surgery, orthotics.
6. What is euglycemic DKA and how is it different from classic DKA?
- Euglycemic DKA: Ketosis (beta-hydroxybutyrate > 3 mmol/L) + acidosis (pH less than 7.3) but glucose less than 14 mmol/L.
- Associated with SGLT2i use (empagliflozin, dapagliflozin, canagliflozin).
- Triggers: Surgery, fasting, low-carb diet, insulin deficiency.
- Management: STOP SGLT2i, IV dextrose + insulin infusion, treat precipitant.
7. What is HHS and how do you manage it?
- Hyperglycemic Hyperosmolar State: Severe hyperglycemia (often > 30 mmol/L), profound dehydration (8-12L fluid deficit), high osmolality (> 320 mOsm/kg), altered mental status, NO ketosis.
- Mortality 10-20%.
- Management:
- "IV fluid resuscitation: 0.9% saline 1L/h initially, then slow to 250-500mL/h."
- "Insulin infusion (ONLY after rehydration started): 0.05 units/kg/h (lower rate than DKA)."
- Potassium replacement.
- Treat precipitant (infection, MI, stroke).
- VTE prophylaxis (high thrombotic risk).
8. What is the DiRECT trial and why is it important?
- Diabetes Remission Clinical Trial (2017).
- Intensive weight management (VLCD 800-850 kcal/day for 3-5 months).
- 46% remission (HbA1c less than 48 mmol/mol off meds) at 12 months.
- 86% remission if > 15 kg weight loss.
- Importance: Proves T2DM can be reversed with intensive lifestyle intervention, especially if early disease (less than 6 years duration).
9. How do you individualize HbA1c targets?
- Newly diagnosed, young, no CVD: 48-53 mmol/mol (6.5-7.0%).
- Elderly, frail, high hypo risk, limited life expectancy: 58-64 mmol/mol (7.5-8.0%).
- Established CVD, multiple comorbidities: 53-58 mmol/mol (7.0-7.5%).
- Pregnancy: 41-47 mmol/mol (6.0-6.4%).
10. What are the cardiovascular benefits of GLP-1 receptor agonists?
- LEADER trial (liraglutide): 13% reduction in MACE, 22% reduction in CV death.
- Meta-analysis: GLP-1 RA reduce MACE by 14%, CV death by 12%.
- Preferred if established ASCVD or weight loss goal.
Common Mistakes to Avoid (OSCE/PACES)
1. "Treating only the sugar"
- Always address BP (target less than 130/80), lipids (statin!), smoking, antiplatelet therapy.
- T2DM is a cardiovascular disease.
2. "Missing the SGLT2i/GLP-1 RA indication"
- If patient has HF, CKD, or ASCVD, they MUST be on SGLT2i or GLP-1 RA regardless of HbA1c.
- Don't wait for HbA1c to be above target.
3. "Over-tight control in the elderly"
- ACCORD trial: Intensive control (HbA1c less than 42 mmol/mol) in high-risk elderly increased mortality.
- Individualize targets. Avoid hypoglycemia.
4. "Ignoring the feet"
- Always examine feet in diabetic patients.
- 10g monofilament test, pulses, inspection for ulcers.
5. "Forgetting sick day rules"
- Patients MUST know to STOP metformin and SGLT2i during vomiting/diarrhea.
- High-yield OSCE station.
6. "Assuming normal creatinine = healthy kidneys"
- Must check UACR (microalbuminuria can occur with normal eGFR).
- Albuminuria is the earliest sign of diabetic nephropathy.
7. "Using HbA1c in anemia/hemolysis/CKD"
- HbA1c falsely low if anemia, hemolysis, recent transfusion, CKD (eGFR less than 30).
- Use fructosamine or CGM instead.
12. References
-
International Diabetes Federation. IDF Diabetes Atlas, 10th Edition. Brussels, Belgium: 2021. Available at: https://diabetesatlas.org
-
Saeedi P, Petersohn I, Salpea P, et al. Global and regional diabetes prevalence estimates for 2019 and projections for 2030 and 2045: Results from the International Diabetes Federation Diabetes Atlas, 9th edition. Diabetes Res Clin Pract. 2019;157:107843. PMID: 31518657. DOI: 10.1016/j.diabres.2019.107843
-
DeFronzo RA. Banting Lecture. From the triumvirate to the ominous octet: a new paradigm for the treatment of type 2 diabetes mellitus. Diabetes. 2009;58(4):773-795. PMID: 19336687. DOI: 10.2337/db09-9028
-
Zheng Y, Ley SH, Hu FB. Global aetiology and epidemiology of type 2 diabetes mellitus and its complications. Nat Rev Endocrinol. 2018;14(2):88-98. PMID: 29219149. DOI: 10.1038/nrendo.2017.151
-
Einarson TR, Acs A, Ludwig C, Panton UH. Prevalence of cardiovascular disease in type 2 diabetes: a systematic literature review of scientific evidence from across the world in 2007-2017. Cardiovasc Diabetol. 2018;17(1):83. PMID: 29884191. DOI: 10.1186/s12933-018-0728-6
-
Davies MJ, Aroda VR, Collins BS, et al. Management of Hyperglycemia in Type 2 Diabetes, 2022. A Consensus Report by the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetes Care. 2022;45(11):2753-2786. PMID: 36148880. DOI: 10.2337/dci22-0034
-
Zinman B, Wanner C, Lachin JM, et al. Empagliflozin, Cardiovascular Outcomes, and Mortality in Type 2 Diabetes. N Engl J Med. 2015;373(22):2117-2128. PMID: 26378978. DOI: 10.1056/NEJMoa1504720
-
Marso SP, Daniels GH, Brown-Frandsen K, et al. Liraglutide and Cardiovascular Outcomes in Type 2 Diabetes. N Engl J Med. 2016;375(4):311-322. PMID: 27295730. DOI: 10.1056/NEJMoa1603827
-
Lean MEJ, Leslie WS, Barnes AC, et al. Primary care-led weight management for remission of type 2 diabetes (DiRECT): an open-label, cluster-randomised trial. Lancet. 2018;391(10120):541-551. PMID: 29217144. DOI: 10.1016/S0140-6736(17)33102-1
-
Perkovic V, Jardine MJ, Neal B, et al. Canagliflozin and Renal Outcomes in Type 2 Diabetes and Nephropathy. N Engl J Med. 2019;380(24):2295-2306. PMID: 30990260. DOI: 10.1056/NEJMoa1811744
-
Heerspink HJL, Stefánsson BV, Correa-Rotter R, et al. Dapagliflozin in Patients with Chronic Kidney Disease. N Engl J Med. 2020;383(15):1436-1446. PMID: 32970396. DOI: 10.1056/NEJMoa2024816
-
Pasquel FJ, Umpierrez GE. Hyperosmolar Hyperglycemic State: A Historic Review of the Clinical Presentation, Diagnosis, and Treatment. Diabetes Care. 2014;37(11):3124-3131. PMID: 25342831. DOI: 10.2337/dc14-0984
-
Burke KR, Schumacher CA, Harpe SE. SGLT2 Inhibitors: A Systematic Review of Diabetic Ketoacidosis and Related Risk Factors in the Primary Literature. Pharmacotherapy. 2017;37(2):187-194. PMID: 27931576. DOI: 10.1002/phar.1881
-
Lipsky BA, Berendt AR, Cornia PB, et al. 2012 Infectious Diseases Society of America Clinical Practice Guideline for the Diagnosis and Treatment of Diabetic Foot Infections. Clin Infect Dis. 2012;54(12):e132-e173. PMID: 22619242. DOI: 10.1093/cid/cis346
-
UK Prospective Diabetes Study (UKPDS) Group. Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). Lancet. 1998;352(9131):837-853. PMID: 9742976.
-
McGuire DK, Shih WJ, Cosentino F, et al. Association of SGLT2 Inhibitors With Cardiovascular and Kidney Outcomes in Patients With Type 2 Diabetes: A Meta-analysis. JAMA Cardiol. 2021;6(2):148-158. PMID: 32852547. DOI: 10.1001/jamacardio.2020.4511
-
Kristensen SL, Rørth R, Jhund PS, et al. Cardiovascular, mortality, and kidney outcomes with GLP-1 receptor agonists in patients with type 2 diabetes: a systematic review and meta-analysis of cardiovascular outcome trials. Lancet Diabetes Endocrinol. 2019;7(10):776-785. PMID: 31422012. DOI: 10.1016/S2213-8587(19)30249-9
-
Collins R, Reith C, Emberson J, et al. Interpretation of the evidence for the efficacy and safety of statin therapy. Lancet. 2016;388(10059):2532-2561. PMID: 27616593. DOI: 10.1016/S0140-6736(16)31357-5
-
UK Prospective Diabetes Study Group. Tight blood pressure control and risk of macrovascular and microvascular complications in type 2 diabetes: UKPDS 38. BMJ. 1998;317(7160):703-713. PMID: 9732337.
-
Gerstein HC, Miller ME, Byington RP, et al. Effects of intensive glucose lowering in type 2 diabetes. N Engl J Med. 2008;358(24):2545-2559. PMID: 18539917. DOI: 10.1056/NEJMoa0802743
-
Bakris GL, Agarwal R, Anker SD, et al. Effect of Finerenone on Chronic Kidney Disease Outcomes in Type 2 Diabetes. N Engl J Med. 2020;383(23):2219-2229. PMID: 33264825. DOI: 10.1056/NEJMoa2025845
-
American Diabetes Association Professional Practice Committee. 2. Classification and Diagnosis of Diabetes: Standards of Medical Care in Diabetes-2023. Diabetes Care. 2023;46(Suppl 1):S19-S40. PMID: 36507649. DOI: 10.2337/dc23-S002
-
Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). UK Prospective Diabetes Study (UKPDS) Group. Lancet. 1998;352(9131):837-853. PMID: 9742976.
-
Rydén L, Grant PJ, Anker SD, et al. ESC Guidelines on diabetes, pre-diabetes, and cardiovascular diseases developed in collaboration with the EASD. Eur Heart J. 2013;34(39):3035-3087. PMID: 23996285. DOI: 10.1093/eurheartj/eht108
-
Pop-Busui R, Boulton AJM, Feldman EL, et al. Diabetic Neuropathy: A Position Statement by the American Diabetes Association. Diabetes Care. 2017;40(1):136-154. PMID: 27999003. DOI: 10.2337/dc16-2042
13. Related Topics
Prerequisites
- Insulin Physiology and Glucose Homeostasis
- Obesity and Metabolic Syndrome
- Pancreatic Beta-Cell Function
Consequences
- Diabetic Ketoacidosis (DKA)
- Hyperglycemic Hyperosmolar State (HHS)
- Diabetic Retinopathy
- Diabetic Nephropathy (Chronic Kidney Disease)
- Diabetic Neuropathy (Peripheral and Autonomic)
- Coronary Artery Disease in Diabetes
- Stroke and Cerebrovascular Disease
- Diabetic Foot and Charcot Neuroarthropathy
- Non-Alcoholic Fatty Liver Disease (NAFLD/MASLD)
Differentials
- Type 1 Diabetes Mellitus (T1DM)
- Latent Autoimmune Diabetes in Adults (LADA)
- Maturity-Onset Diabetes of the Young (MODY)
- Pancreatic Diabetes (Chronic Pancreatitis, Cystic Fibrosis)
- Drug-Induced Hyperglycemia (Glucocorticoids, Antipsychotics)
- Endocrine Causes of Hyperglycemia (Cushing's Syndrome, Acromegaly, Phaeochromocytoma)
Related Conditions
- Prediabetes (Impaired Fasting Glucose, Impaired Glucose Tolerance)
- Metabolic Syndrome
- Polycystic Ovary Syndrome (PCOS)
- Gestational Diabetes Mellitus (GDM)
- Hypoglycemia (Iatrogenic, Insulinoma)
Citation Count: 25 Line Count: 1,784 Target Examination: MRCP, FRACP, USMLE, PLAB Difficulty: Moderate Last Updated: 2026-01-09