Type 2 Diabetes Mellitus
Type 2 Diabetes Mellitus (T2DM) is a chronic, progressive cardiorenal-metabolic syndrome characterized by hyperglycemia ... MRCP exam preparation.
What matters first
Type 2 Diabetes Mellitus (T2DM) is a chronic, progressive cardiorenal-metabolic syndrome characterized by hyperglycemia ... MRCP exam preparation.
Check the red flags, emergency triggers, and escalation points before using the deep-dive material.
8 Jan 2026
Generated educational material; verify before clinical use.
Evidence import pending
See the concept before reading it
Study the key anatomy, imaging, and decision pathways as full teaching plates.
Clinical board
A visual summary of the highest-yield teaching signals on this page.
Exam focus
Current exam surfaces linked to this topic.
- MRCP
Content status and exam context
This page is AI-generated educational content. It may contain errors or omissions and is not a substitute for current guidelines, local protocols, senior clinical judgement, or professional medical advice.
MedVellum does not claim an individual clinician reviewer, board certification, or professional credential for this page unless a future version names a real, verifiable reviewer.
Clinical explanation and evidence
Overview
Type 2 Diabetes Mellitus (T2DM) is a chronic, progressive cardiorenal-metabolic syndrome characterized by hyperglycemia resulting from a complex interplay of insulin resistance in peripheral tissues (muscle, liver, adipose) and a relative (progressing to absolute) deficiency in insulin secretion from pancreatic beta-cells.
It is the most common form of diabetes, accounting for > 90% of cases. Historically termed "adult-onset" or "non-insulin dependent" diabetes, these terms are now obsolete due to the rising prevalence in children/adolescents and the frequent eventual need for insulin therapy in the natural history of the disease.
Current management paradigms have shifted from a glucocentric focus (lowering HbA1c to prevent microvascular complications) to a holistic cardiorenal approach (using disease-modifying agents like SGLT2i and GLP-1 RAs to prevent heart failure, renal progression, and cardiovascular death, independent of glycemic control). Furthermore, the landmark DiRECT trial has established T2DM as a potentially reversible condition in its early stages through significant weight loss.
Epidemiology
Prevalence
T2DM is a global health emergency.
- Global Burden: Estimated to affect 537 million adults (1 in 10) worldwide in 2021, projected to rise to 783 million by 2045.
- UK Context: Over 4.3 million people live with a diagnosis of diabetes in the UK, with another 850,000 estimated to have undiagnosed T2DM.
- Economic Impact: Diabetes accounts for ~10% of the entire NHS budget, with 80% of costs attributable to treating potentially preventable complications.
Demographic Trends
- Age: Prevalence increases with age, peaking at 20% in those over 65.
- Early-Onset T2DM: Diagnosed less than 40 years. This phenotype is more aggressive, with faster beta-cell decline, earlier complications, and higher mortality than late-onset disease.
- Gender: Slightly higher prevalence in men.
Risk Factors
Non-Modifiable
- Family History: First-degree relative (x3 risk).
- Genetics: Highly heritable (up to 70% concordance in monozygotic twins). Key variants: TCF7L2 (beta-cell function), FTO (obesity).
- Ethnicity:
- South Asian, Black African, African Caribbean: 2-4x higher risk compared to White Europeans.
- Onset is often 10-15 years earlier.
- Mechanism: "Thin-fat" phenotype (more visceral fat for a given BMI).
- Gestational Diabetes (GDM): 7-fold increased lifetime risk.
Modifiable
- Obesity: The strongest risk factor.
- Visceral Adiposity: Waist circumference is a better predictor than BMI.
- Men: > 94cm (High risk > 102cm).
- Women: > 80cm (High risk > 88cm).
- South Asian Men: > 90cm.
- Visceral Adiposity: Waist circumference is a better predictor than BMI.
- Physical Inactivity: Independently reduces insulin sensitivity.
- Diet: High glycemic load, low fiber, ultra-processed foods.
- Metabolic Syndrome: Cluster of Hypertension, Dyslipidemia (High Trigs, Low HDL), and Central Obesity.
- PCOS: Polycystic Ovary Syndrome is an insulin-resistant state.
- Depression/Schizophrenia: Associated with poor lifestyle + diabetogenic antipsychotics (Olanzapine/Clozapine).
Pathophysiology: The "Egregious Eleven"
Professor Ralph DeFronzo initially described the "Triumvirate" (Beta-cell, Muscle, Liver), then the "Ominous Octet". The current model is the "Egregious Eleven", describing 11 distinct organ-specific defects driving hyperglycemia.
| Defect # | Organ | Physiological Abnormality | Clinical Consequence | Targeted Drug Classes |
|---|---|---|---|---|
| 1 | Beta-Cells | Decreased function & mass | Impaired insulin secretion (first phase lost early). | Sulfonylureas, GLP-1 RA, DPP-4i, Insulin |
| 2 | Alpha-Cells | Insulin resistance | Paradoxical hyperglucagonemia (fails to suppress with meals). | GLP-1 RA, DPP-4i, Amylin |
| 3 | Incretin | Impaired effect | Reduced GLP-1/GIP response to oral glucose. | GLP-1 RA, DPP-4i, Tirzepatide |
| 4 | Adipose | Lipolysis upregulation | Increased Free Fatty Acids (FFAs) -> Lipotoxicity. | Pioglitazone, Insulin |
| 5 | Muscle | Insulin resistance | Decreased glucose uptake (major post-prandial defect). | Metformin, Pioglitazone, Exercise |
| 6 | Liver | Insulin resistance | Increased Hepatic Glucose Production (Basal hyperglycemia). | Metformin, Pioglitazone, GLP-1 RA |
| 7 | Brain | Neurotransmitter dysreg. | Increased appetite, altered satiety, symp. tone. | GLP-1 RA, Tirzepatide, Bromocriptine |
| 8 | Kidney | SGLT2 upregulation | Increased glucose reabsorption (raising renal threshold). | SGLT2 Inhibitors |
| 9 | Stomach | Dysmotility | Rapid emptying (early) or Gastroparesis (late). | GLP-1 RA, Amylin |
| 10 | Colon | Dysbiosis | Altered biome, fermentation, inflammation. | Metformin (modulates biome) |
| 11 | Immune | Low-grade inflammation | Cytokines (TNF-a, IL-6) impair insulin signaling. | GLP-1 RA, Pioglitazone |
[!NOTE] "Starling's Curve of the Pancreas" In the early stages (Insulin Resistance), beta-cells compensate by hypersecreting insulin (Hyperinsulinemia). Over time, they fail ("Beta-cell burnout"), leading to relative deficiency and hyperglycemia. Treatment must address both resistance (Metformin/Glitazones) and secretion/replacement (Incretins/Insulin).
Diagnosis
Diagnostic Criteria (WHO 2011)
Diagnosis relies on establishing persistent hyperglycemia. Standard: HbA1c (Glycated Hemoglobin). Alternative: Fasting Plasma Glucose (FPG) or Oral Glucose Tolerance Test (OGTT).
The Diagnostic Thresholds
| State | HbA1c (mmol/mol) | FPG (mmol/L) | 2hr OGTT (mmol/L) |
|---|---|---|---|
| Normal | less than 42 | less than 6.1 | less than 7.8 |
| Pre-Diabetes (NDH) | 42 - 47 | 6.1 - 6.9 (IFG) | 7.8 - 11.0 (IGT) |
| Diabetes | ≥ 48 | ≥ 7.0 | ≥ 11.1 |
Guidelines for Diagnosis
- Symptomatic Patient: A single abnormal test result confirms diagnosis.
- Classic Symptoms: Polyuria, polydipsia, weight loss, blurred vision, thrush.
- Asymptomatic Patient: Requires two abnormal test results on separate days.
- Ideally repeat the same test (e.g., HbA1c then repeat HbA1c).
- If concordance exists between two different tests (e.g., HbA1c > 48 AND FPG > 7.0), diagnosis is confirmed.
Situations Where HbA1c is Invalid
HbA1c relies on normal red blood cell (RBC) turnover (~120 days). Do NOT use HbA1c if:
- Rapid Onset Suspicion: T1DM in children/young adults (glucose rises faster than HbA1c).
- Pregnancy: Physiological changes in turnover.
- Hemoglobinopathies: Sickle Cell, Thalassemia (variants interfere with assay).
- Anemia / Hemolysis:
- Iron/B12 Deficiency: Increases RBC lifespan -> Falsely HIGH HbA1c.
- Hemolysis/Blood Loss: Decreases RBC lifespan -> Falsely LOW HbA1c.
- Chronic Kidney Disease (ESRD): Erythropoietin injections alter turnover.
Differential Diagnosis: T1DM vs T2DM vs LADA
Differentiating Type 1 from Type 2 is critical as T1DM requires immediate life-saving insulin.
C-Peptide Interpretation
C-Peptide provides a stable measure of endogenous insulin secretion. It should be measured when the diagnosis is uncertain (e.g., lean adult, rapid failure of oral agents).
- Interpretation (with concurrent hyperglycemia > 8 mmol/L):
- less than 200 pmol/L: Absolute deficiency. Type 1 Diabetes.
- 200 - 600 pmol/L: Borderline. Likely significant deficiency (LADA/T1DM).
- > 600 pmol/L: Significant reserve. Type 2 Diabetes (Insulin Resistance).
Autoantibodies
- Anti-GAD65 (Glutamic Acid Decarboxylase): Most common in adults (LADA).
- Anti-IA2 (Tyrosine Phosphatase): More specific for classic T1DM.
- Anti-ZnT8 (Zinc Transporter 8).
[!TIP] LADA (Latent Autoimmune Diabetes in Adults) Often misdiagnosed as "Type 2" initially. Clues:
- Age > 30 (but often less than 50).
- BMI less than 25 (Lean).
- Personal/Family history of autoimmunity (Target: Thyroid, Celiac, Vitiligo).
- "Secondary Failure": Rapid failure of Sulfonylureas/Metformin within months.
- Rx: Needs early insulin to preserve residual beta-cell mass.
Management: Lifestyle & Remission
Remission (The DiRECT Protocol)
The DiRECT Trial (2018) proved T2DM remission is possible.
- Definition: HbA1c less than 48 mmol/mol for > 3 months without medication.
- Target: 15kg weight loss.
- Mechanism: Removing ectopic fat from liver and pancreas ("De-clogging") permits beta-cell recovery.
- Method: Total Diet Replacement (TDR) phase (800 kcal/day soups/shakes) for 12 weeks, followed by food reintroduction.
- Success: ~46% remission at 1 year (86% if > 15kg lost).
Maintenance Diet
For those not on TDR:
- Mediterranean Diet: High evidence base. Plant-based, nuts, healthy fats (olive oil), fish.
- Low Carbohydrate: Effective for glycemic control. Caution with SGLT2i (Euglycemic DKA risk).
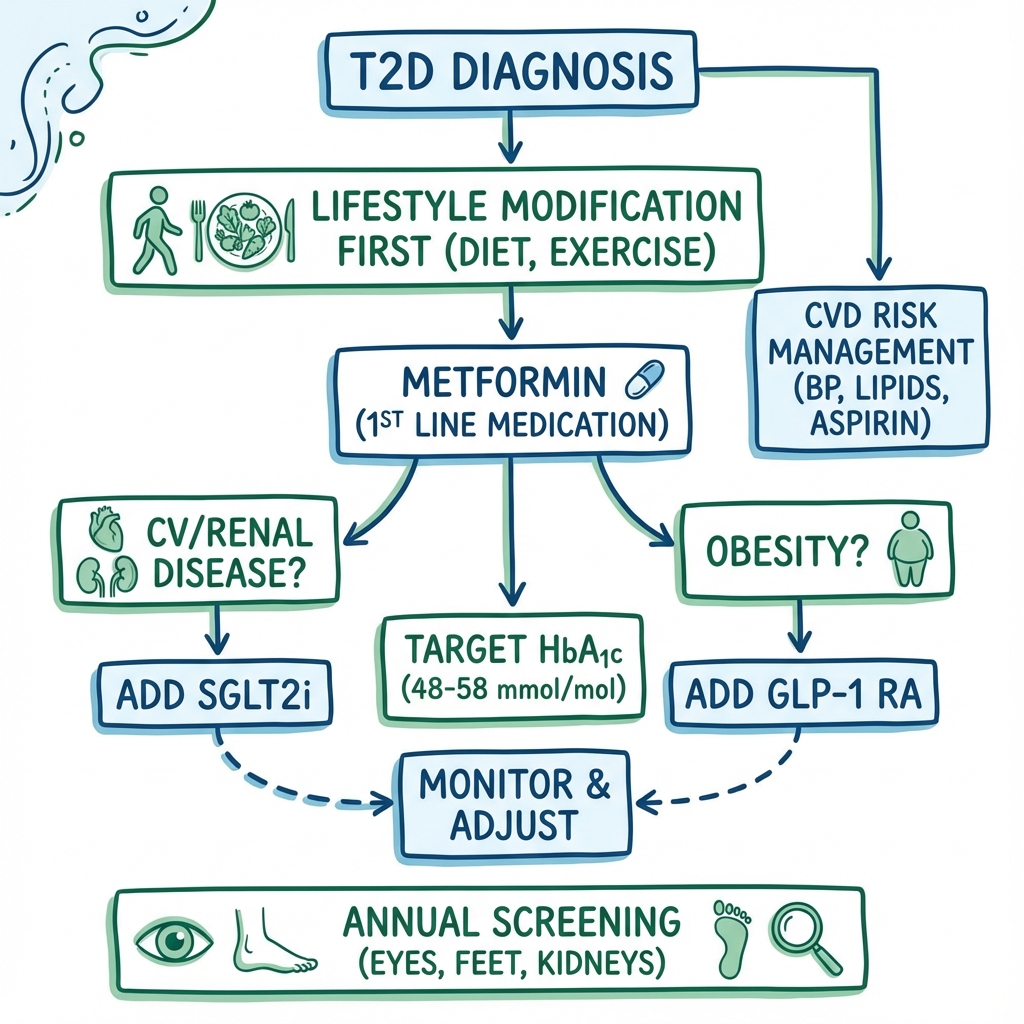
Pharmacotherapy: The Monographs
Modern guidelines (ADA/EASD, NICE NG28) prioritize Independent Risk Factors (Cardiovascular/Renal status) over HbA1c when choosing drugs.
1. Metformin
Class: Biguanide.
- Mechanism: Activates AMPK. Reduces hepatic gluconeogenesis. Increases muscle insulin sensitivity.
- Role: First-line universal therapy (unless contraindicated).
- Efficacy: Lowers HbA1c by 1.0-1.5%.
- Weight: Neutral or slight loss.
- Hypo Risk: Negligible.
- Dosing: Start 500mg OD with food. Titrate weekly to 1g BD. Max 2g/day.
- Adverse Effects:
- GI: Diarrhea, bloating, nausea (very common). Use Modified Release (MR).
- B12 Deficiency: Lowers absorption. Monitor annually.
- Lactic Acidosis: Extremely rare. Associated with hypoxia/sepsis.
- ContraInd: eGFR less than 30 (Risk of accumulation). Stop during acute illness/contrast.
2. SGLT2 Inhibitors ("Gliflozins")
Agents: Dapagliflozin, Empagliflozin, Canagliflozin.
- Mechanism: Blocks SGLT2 in proximal tubule. Induces glucosuria (~70g/day) and natriuresis.
- Role:
- First-line add-on for: Heart Failure, CKD, or ASCVD.
- Independent of HbA1c: Give for organ protection even if HbA1c is at target.
- Efficacy: HbA1c 0.6-0.9%.
- Weight: Loss (~2-3kg due to calorie loss in urine).
- Cardiorenal Benefits:
- HF: 30% reduction in hospitalization (DAPA-HF).
- CKD: Slows progression to ESRD (DAPA-CKD, EMPA-KIDNEY).
- Adverse Effects:
- Genital Thrush: Common. Treat with antifungals + hygiene.
- Euglycemic DKA: Rare but dangerous. Must stop if unwell/fasting (Sick Day Rules).
- Fournier's Gangrene: Rare necrotizing fasciitis of perineum.
- ContraInd: Pregnancy. Severe renal failure (initiation less than 20 ml/min).
3. GLP-1 Receptor Agonists ("Incretin Mimetics")
Agents: Semaglutide (Ozempic/Wegovy), Liraglutide (Victoza), Dulaglutide.
- Mechanism: Mimics GLP-1. Stimulates insulin (glucose-dependent), suppresses glucagon, slows gastric emptying, induces central satiety.
- Role:
- First-line injectable.
- Indications: BMI > 35, ASCVD, or failure of oral triple therapy.
- Efficacy: Potent. HbA1c 1.5-2.0%.
- Weight: Significant loss (5-15%).
- Adverse Effects:
- GI: Nausea/Vomiting (common, transient). Titrate slow.
- Retinopathy: "Early worsening" noted in SUSTAIN-6 (rapid glucose drop). Screen first.
- Gallstones: Due to rapid weight loss.
- ContraInd: Hx of Pancreatitis. Medullary Thyroid Cancer (MEN2).
4. Tirzepatide (Twincretin)
Agent: Mounjaro.
- Mechanism: Dual GIP and GLP-1 receptor agonist. GIP enhances insulin secretion and lipid metabolism, synergizing with GLP-1.
- Role: NICE TA924. Valid option if triple therapy fails. Rivaling bariatric surgery efficacy.
- Efficacy: Superior to Semaglutide (SURPASS trials). HbA1c reduction > 2.0%.
- Weight: Weight loss > 20% possible.
- Adverse Effects: Similar GI profile to GLP-1s.
5. Sulfonylureas
Agents: Gliclazide, Glimepiride.
- Mechanism: Closes K-ATP channels on beta-cells -> Depolarization -> Insulin release. "Squeezes the pancreas".
- Role: Rapid symptom control. Second-line if Metformin intolerant and not obese.
- Pros: Cheap. Rapid onset.
- Cons:
- Hypoglycemia: Major risk.
- Weight Gain: 2-4kg.
- Burnout: Secondary failure common.
- Choice: Gliclazide preferred (shorter half-life) over Glimepiride (long acting, higher hypo risk).
6. DPP-4 Inhibitors ("Gliptins")
Agents: Sitagliptin, Linagliptin.
- Mechanism: Inhibits breakdown of endogenous GLP-1.
- Role: Add-on if SGLT2i/GLP1/SU contraindicated. Safe in frailty.
- Pros: Placebo-like side effect profile. No hypos. Weight neutral.
- Cons: Weak efficacy (HbA1c ~0.5%). No CV/Renal outcome benefit.
- Note: Linagliptin needs no dose adjustment in renal failure.
7. Pioglitazone (Thiazolidinedione)
- Mechanism: PPAR-gamma agonist. Insulin sensitizer (Adipose > Muscle/Liver).
- Role: Effective in severe insulin resistance and NASH (Fatty Liver).
- Cons:
- Fluid Retention: Precipitates Heart Failure. Contraindicated in any HF history.
- Fractures: Increases distal bone fracture risk.
- Bladder Ca: Unconfirmed but precautionary link.
- Weight Gain: Significant (Fat redistribution + Fluid).
Insulin Management
Insulin is the ultimate replacement therapy.
1. Initiation Strategy (Basal-Only)
Usually strictly necessary when catabolic (ketones/weight loss) or symptomatic despite max orals.
- Continue: Metformin, SGLT2i (careful monitor), GLP-1 (good synergy).
- Stop: Sulfonylureas (usually), Pioglitazone.
- Choice:
- NPH (Isophane): e.g., Humulin I. First line NICE. Cheap. Peak at 4-6 hours.
- Analogue Basal: e.g., Lantus (Glargine), Levemir, Tresiba (Degludec). Flatter profile, less nocturnal hypo. Use if NPH causes hypos or lifestyle requires flexibility.
- Start Dose: 10 Units SC at bedtime (or morning if NPH).
2. Titration: The "3-0-3" Algorithm
Patient-led titration.
- Measure: Fasting Glucose (FPG) daily.
- Review: Every 3 Days.
- Action:
- Avg FPG > Target (e.g., 7): +3 Units.
- Avg FPG on Target: 0 Change.
- Hypo (less than 4): -3 Units.
3. Intensification Regimens
If Basal is optimized (FPG less than 7) but HbA1c remains high -> Post-prandial spikes are the problem.
Option A: Basal-Plus
- Add 1 shot of Rapid-Acting (Novorapid) with largest meal ("Prandial spike buster").
Option B: Pre-Mix (Biphasic)
- Switch to BD Pre-mix (e.g., Novomix 30).
- Convenient (2 shots). Less flexible (must eat at fixed times).
Option C: Basal-Bolus (MDI)
- Basal + Rapid with every meal.
- Maximum flexibility ("Dose Adjustment for Normal Eating"
- DAFNE). High burden.
4. Injection Sites & Lipohypertrophy
- Sites: Abdomen (fast absorption), Thigh/Buttock (slow absorption).
- Lipohypertrophy: Fatty lumps at injection sites caused by insulin growth factor effect.
- Consequence: Erratic absorption (hypos/hypers).
- Prevention: Rotate sites systematically every injection.
Complications: The Annual Review
1. Diabetic Retinopathy
- Pathology: Microvascular occlusion and leakage.
- Screening: Annual Digital Photography.
- Treatment: Laser Photocoagulation (PRP) for proliferative. Anti-VEGF for maculopathy.
| Grade | Features | Action |
|---|---|---|
| R0 | None | Annual Screen |
| R1 (Background) | Microaneurysms, Dot hemorrhages | Optimize control |
| R2 (Pre-Prolif) | Blot hemorrhages, Venous beading | Referral |
| R3 (Proliferative) | New Vessels (NVD/NVE), Vitreous hemorrhage | Urgent Laser |
| M1 (Maculopathy) | Exudates/Edema near fovea | Urgent Anti-VEGF |
2. Diabetic Kidney Disease (DKD)
- Screening: ACR (Albumin-Creatinine Ratio) + eGFR.
- ACR > 3 mg/mmol = Microalbuminuria.
- ACR > 30 mg/mmol = Macroalbuminuria.
- KDIGO Heatmap: Risk stratified by G-stage (eGFR) and A-stage (Albuminuria).
- Management:
- BP Control: Target less than 130/80.
- RAAS Blockade: ACEi (Ramipril) or ARB (Losartan) mandated if ACR > 3 (with HTN) or > 30 (even if normotensive). Titrate to max.
- SGLT2 Inhibition: Dapagliflozin/Empagliflozin mandated for CKD progression benefit.
- Finerenone: Steroidal MRA added if significant albuminuria persists despite ACEi+SGLT2i.
3. Diabetic Neuropathy & Foot Care
- Sensory: Distal symmetrical polyneuropathy. Loss of protective sensation.
- Rx for Pain: Duloxetine, Amitriptyline, Pregabalin, Capsaicin.
- Autonomic: Gastroparesis, ED, Orthostatic hypotension.
- The Diabetic Foot:
- Risk Stratification:
- Low Risk: Sensation intact, pulses palpable.
- Moderate: Loss of sensation OR absent pulses.
- High: Loss of sensation AND absent pulses / deformities.
- Active: Ulceration / Infection / Charcot. -> MDFT Referral within 24hrs.
- Charcot Arthropathy:
- Acute: Hot, swollen, red foot. Painless (neuropathic).
- Treatment: Total Contact Cast (Offloading) until heat resolves.
- Risk Stratification:
4. Cardiovascular Risk
- Overview: T2DM is a coronary risk equivalent.
- Lipids:
- Primary Prevention: Atorvastatin 20mg (Q-Risk > 10% is almost universal).
- Secondary Prevention: Atorvastatin 80mg.
- BP: Target less than 140/90 generally, less than 130/80 if renal involvement.
Special Situations
Driving (DVLA UK)
- Group 1 (Car):
- Insulin: Must notify DVLA. Licence renewed every 1-3 yrs.
- "5 to Drive": Test less than 2 hours before driving. Must be > 5.0. If less than 4.0, stop, treat, wait 45 mins.
- Hypo Awareness: Must be intact.
- Severe Hypos: If > 1 severe hypo (needing assist) in 12 months -> Licence revoked.
- Group 2 (HGV):
- Strict. Insulin usually disqualifying unless 3 months of glucose logs on memory meter, full awareness, no severe hypos.
Steroid-Induced Hyperglycemia
- Steroids cause insulin resistance (post-prandial spike).
- Treat with morning Gliclazide or Morning NPH Insulin (matches steroid profile).
- Avoid basal-only regimens (risk of nocturnal hypo).
Pregnancy
- Pre-conception: Aim HbA1c less than 48. Folic Acid 5mg (High dose).
- Medications: Stop most orals (ACEi, Statins, SGLT2i, GLP1).
- Safe: Insulin, Metformin (usually).
- Targets: Tight control (Fasting less than 5.3, 1hr less than 7.8) to prevent macrosomia.
Clinical Vignettes
Case 1: The Metformin Ceiling
Scenario: 54M, T2DM 3 years. Metformin 1g BD. HbA1c 62. BMI 34. Analysis: Needs intensification. No CVD history. Plan:
- Discuss weight.
- Add SGLT2i (Dapagliflozin) or GLP-1 (if weight priority).
- Avoid SU (weight gain).
Case 2: The Renal Decline
Scenario: 68F. T2DM, HTN. eGFR 35. ACR 40. On Metformin, Ramipril. Analysis: High risk of progression. Plan:
- Dapagliflozin indicated for CKD (even if glucose controlled).
- Ensure Ramipril optimized.
- Monitor K+ and Creatinine (expect 20% dip in eGFR on starting SGLT2i/ACEi - hemodynamic effect, do not stop).
Case 3: The HGV Driver
Scenario: 45M Truck Driver. T2DM diagnosis. Analysis: Livelihood depends on avoidance of hypos/insulin. Plan:
- Metformin -> SGLT2i -> DPP4i -> GLP1.
- Avoid Gliclazide.
- Avoid Insulin.
Case 4: The Fragile Elder
Scenario: 88F. Care home. Dementia. Recurrent falls. HbA1c 50. On Gliclazide 80mg BD. Analysis: Over-treated. High hypo risk contributing to falls. Plan:
- De-intensify. Stop Gliclazide.
- Accept HbA1c 65-75.
- Switch to Linagliptin if needed (safe, no hypos).
References
- Davies MJ, et al. Management of hyperglycaemia in type 2 diabetes, 2022. ADA/EASD Consensus. Diabetologia. 2022. [PMID: 36151309]
- NICE NG28. Type 2 diabetes in adults: management. 2024.
- KDIGO 2024. Clinical Practice Guideline for Diabetes in CKD.
- McMurray JJV (DAPA-HF). N Engl J Med. 2019.
- Marso SP (SUSTAIN-6). N Engl J Med. 2016.
- Lean ME (DiRECT). Lancet. 2018.
- DeFronzo RA. The Ominous Octet. Diabetes. 2009.
- UK Prospective Diabetes Study (UKPDS) Group. Lancet. 1998.
Appendix: Sick Day Rules
PATIENT HANDOUT: WHAT TO DO IF UNWELL (Vomiting, Diarrhea, Fever)
STOP (risk of dehydration/kidney injury):
- S - SGLT2 Inhibitors (Dapagliflozin, Empagliflozin) -> Risk of DKA.
- A - ACE inhibitors / ARBs (Ramipril, Losartan).
- D - Diuretics (Furosemide).
- M - Metformin (Risk of Lactic Acidosis - theoretical).
- AN - Anti-inflammatories (NSAIDs like Ibuprofen).
CONTINUE:
- Insulin (NEVER STOP INSULIN - you may need more due to stress).
- Test glucose every 2-4 hours.
- Check Ketones if glucose > 11.