Upper Gastrointestinal Bleeding (Adult)
Upper gastrointestinal bleeding (UGIB) is a common and potentially life-threatening medical emergency defined as bleedin... MRCP exam preparation.
What matters first
Upper gastrointestinal bleeding (UGIB) is a common and potentially life-threatening medical emergency defined as bleedin... MRCP exam preparation.
Hematemesis (bright red or coffee-ground)
9 Jan 2026
Generated educational material; verify before clinical use.
Visible references section
See the concept before reading it
Study the key anatomy, imaging, and decision pathways as full teaching plates.
Clinical board
A visual summary of the highest-yield teaching signals on this page.
Urgent signals
Safety-critical features pulled from the topic metadata.
- Hematemesis (bright red or coffee-ground)
- Melena (black, tarry, foul-smelling stools)
- Hemodynamic instability (SBP less than 90 mmHg, HR >100 bpm)
- Syncope or near-syncope
Exam focus
Current exam surfaces linked to this topic.
- MRCP
Linked comparisons
Differentials and adjacent topics worth opening next.
- Lower Gastrointestinal Bleeding
Content status and exam context
This page is AI-generated educational content. It may contain errors or omissions and is not a substitute for current guidelines, local protocols, senior clinical judgement, or professional medical advice.
MedVellum does not claim an individual clinician reviewer, board certification, or professional credential for this page unless a future version names a real, verifiable reviewer.
Clinical explanation and evidence
Upper Gastrointestinal Bleeding (Adult)
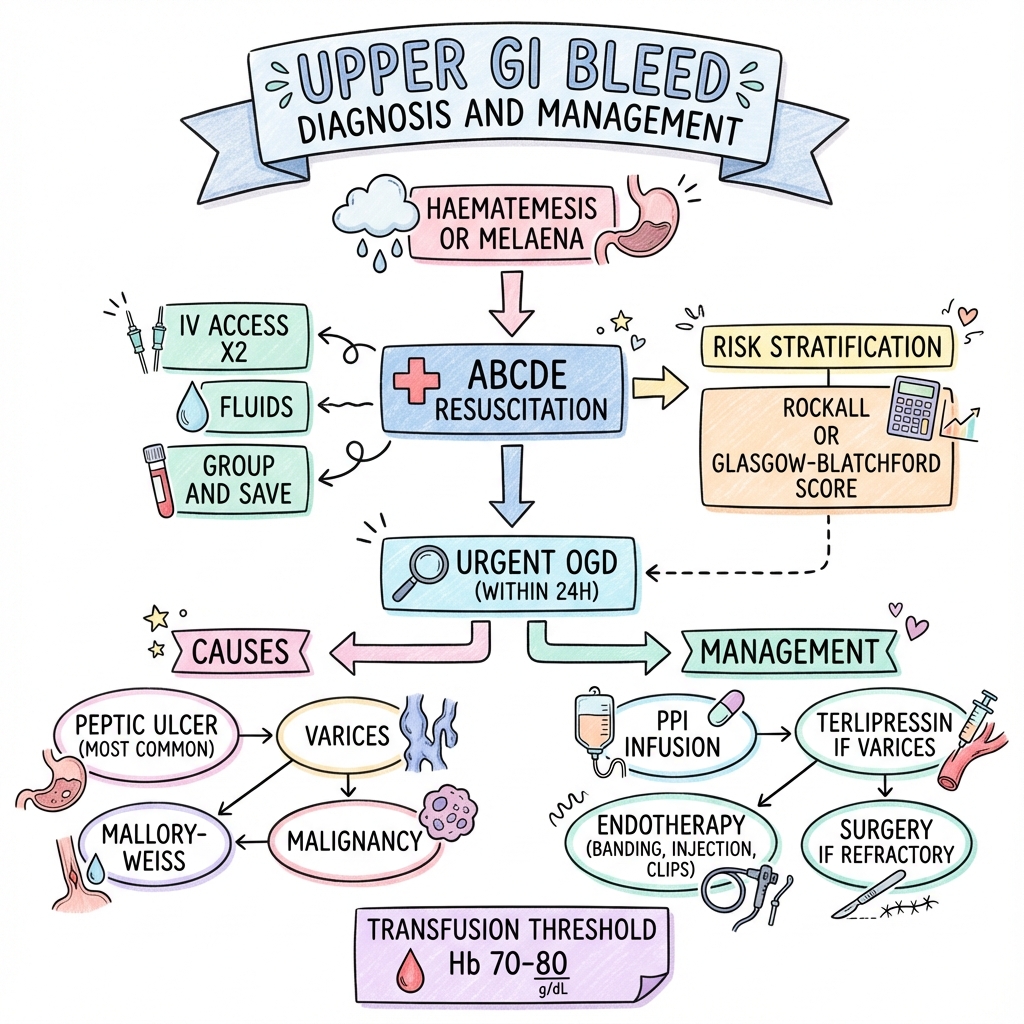
1. Clinical Overview
Summary
Upper gastrointestinal bleeding (UGIB) is a common and potentially life-threatening medical emergency defined as bleeding originating proximal to the ligament of Treitz (the duodenojejunal junction). It represents one of the most frequent gastroenterological emergencies worldwide, with an annual incidence of 50-150 per 100,000 adults. [1,2] UGIB typically presents with hematemesis (vomiting of blood, either bright red or coffee-ground appearance) or melena (black, tarry, foul-smelling stools), though massive upper GI hemorrhage may present with hematochezia (passage of bright red blood per rectum) due to rapid transit time.
The condition is broadly categorized into two major groups based on etiology: non-variceal and variceal bleeding. Non-variceal causes account for approximately 80-90% of cases and include peptic ulcer disease (40-50%), gastric and duodenal erosions (15-20%), Mallory-Weiss tears (5-10%), vascular lesions including Dieulafoy lesions, and upper GI malignancies. [3] Variceal bleeding, accounting for 10-20% of cases, occurs in the context of portal hypertension, most commonly secondary to liver cirrhosis, and carries significantly higher mortality rates (15-20%) compared to non-variceal bleeding (5-10%). [4,5]
The management of UGIB requires a rapid, systematic, and evidence-based approach. Initial assessment follows the ABCDE protocol with emphasis on early recognition of hemodynamic instability and immediate resuscitation. Risk stratification using validated scoring systems—particularly the Glasgow-Blatchford Score (GBS) at presentation and the Rockall Score post-endoscopy—guides decisions regarding admission, transfusion, and timing of endoscopy. [6,7] The Glasgow-Blatchford Score has emerged as the preferred pre-endoscopy tool, with a score of 0 having excellent negative predictive value for the need for intervention, allowing safe outpatient management in carefully selected patients.
Contemporary management emphasizes restrictive transfusion strategies (target hemoglobin 70-90 g/L), high-dose proton pump inhibitor therapy for non-variceal bleeding, and early endoscopy (within 24 hours for most patients, within 12 hours for high-risk patients). [8] For suspected variceal bleeding, immediate vasoactive drug administration (terlipressin or somatostatin analogues) and prophylactic antibiotics are critical interventions that should be initiated before endoscopy. [9] Tranexamic acid, despite theoretical benefits, has been shown not to reduce mortality in gastrointestinal bleeding and carries increased thromboembolic risk. [10]
Despite advances in endoscopic therapy and critical care, mortality from UGIB has remained relatively stable over recent decades at approximately 5-10%, largely due to increasing patient age and comorbidity burden, particularly cardiovascular and renal disease. [2] The increasing use of antiplatelet agents (aspirin, clopidogrel) and anticoagulants (warfarin, direct oral anticoagulants) has also contributed to both increased incidence and severity of bleeding episodes.
Key Facts
- Definition: Bleeding from the gastrointestinal tract proximal to the ligament of Treitz (duodenojejunal junction)
- Incidence: 50-150 per 100,000 adults per year, with geographic variation [1,2]
- Mortality: 5-10% overall; 15-20% for variceal bleeding; significantly higher in elderly and those with comorbidities [4,5]
- Common Causes:
- Peptic ulcer disease (40-50%)
- Gastritis/Erosive disease (15-20%)
- Esophageal/gastric varices (10-20%)
- Mallory-Weiss tears (5-10%)
- Vascular lesions including Dieulafoy (5%)
- Malignancy (2-5%)
- Risk Stratification Tools:
- Glasgow-Blatchford Score (pre-endoscopy; guides admission and intervention need) [6]
- Rockall Score (post-endoscopy; predicts mortality and rebleeding) [7]
- First-line Investigation: Urgent upper GI endoscopy (esophagogastroduodenoscopy, OGD)
- Transfusion Threshold: Hemoglobin less than 70 g/L (or less than 80 g/L in patients with acute coronary syndrome or significant cardiovascular disease) [8]
- Emergency Management Priorities:
- ABCDE assessment and resuscitation
- Large-bore IV access (2 × 14-16G cannulae)
- Crystalloid resuscitation
- Blood transfusion (restrictive strategy)
- Correction of coagulopathy
- Risk stratification
- Endoscopy within 24 hours (or 12 hours if high-risk)
Clinical Pearls
"Resuscitate before you investigate" — Hemodynamic stability must be achieved before endoscopy. Never delay fluid resuscitation or blood transfusion while waiting for the gastroenterologist. Unstable patients may require intubation and airway protection prior to endoscopy.
"The urea:creatinine ratio is a diagnostic clue" — An isolated rise in blood urea nitrogen (BUN) or urea, disproportionate to creatinine (ratio >100:1 or >36:1 in SI units), strongly suggests an upper GI source. This occurs due to digestion of blood proteins in the small intestine with absorption of nitrogenous waste products.
"GBS 0 can go home" — A Glasgow-Blatchford Score of 0 has a negative predictive value exceeding 99% for the need for intervention (endoscopic therapy, transfusion, or surgery) and identifies patients suitable for safe outpatient management with early elective endoscopy. [6]
"Variceal bleeding is a different beast" — If variceal bleeding is suspected (history of cirrhosis, chronic liver disease, stigmata including jaundice, ascites, spider naevi), immediately start vasoactive drugs (terlipressin 2mg IV every 4 hours) and prophylactic antibiotics (ceftriaxone 1g IV daily) before endoscopy. This approach reduces mortality, rebleeding, and infection rates. [9,14]
"High-risk stigmata demand therapy" — Forrest classification guides endoscopic intervention. Active spurting or oozing (Forrest Ia/Ib) and visible vessel (Forrest IIa) require immediate endoscopic hemostasis. Adherent clot (Forrest IIb) carries intermediate risk and often benefits from clot removal and treatment of underlying lesion. [11]
"Avoid tranexamic acid" — The HALT-IT trial definitively showed that tranexamic acid does not reduce mortality in gastrointestinal bleeding and increases venous thromboembolism risk. It should not be used routinely in UGIB. [10]
"Restrictive transfusion saves lives" — The landmark Villanueva trial demonstrated that restrictive transfusion (threshold Hb less than 70 g/L, target 70-90 g/L) significantly reduces mortality and rebleeding compared to liberal transfusion, particularly in variceal bleeding. [8]
Why This Matters Clinically
Upper gastrointestinal bleeding remains a leading cause of emergency hospital admission and a significant source of morbidity and mortality worldwide. Despite major advances in pharmacotherapy, endoscopic techniques, and critical care, the overall mortality rate has plateaued at 5-10% over the past three decades. This stability reflects the changing demographic of UGIB patients: an aging population with increasing comorbidity burden, particularly cardiovascular disease, chronic kidney disease, and malignancy. [2]
The widespread use of aspirin for cardiovascular prophylaxis, dual antiplatelet therapy following percutaneous coronary intervention, and direct oral anticoagulants for atrial fibrillation has fundamentally altered the epidemiology of UGIB. While Helicobacter pylori eradication programs have reduced peptic ulcer-related bleeding in younger populations, medication-associated bleeding (NSAIDs, antiplatelet agents, anticoagulants) now predominates, particularly in the elderly. [12]
Rapid recognition of high-risk patients, adherence to evidence-based resuscitation and transfusion protocols, appropriate timing of endoscopy, and effective endoscopic hemostasis are essential for optimizing outcomes. Equally important is the post-bleeding management: Helicobacter pylori eradication, appropriate gastroprotection, and careful consideration of when to restart anticoagulant and antiplatelet therapy to balance bleeding and thrombotic risks. [13]
2. Epidemiology
Incidence & Prevalence
- Incidence: 50-150 per 100,000 person-years in Western countries, with significant geographic variation [1,2]
- Higher rates in Northern Europe and North America (100-180/100,000)
- Lower rates in Asia (40-60/100,000), though increasing with NSAID use
- Trends Over Time:
- Overall incidence has remained stable or slightly declined over past 20 years
- Peptic ulcer-related UGIB declining (due to H. pylori eradication and PPI use)
- Medication-related UGIB increasing (NSAIDs, antiplatelet agents, anticoagulants)
- Variceal bleeding incidence stable, reflecting persistent burden of cirrhosis
- Hospitalizations:
- One of the most common reasons for emergency gastrointestinal admission
- Account for approximately 50,000-70,000 hospital admissions annually in the UK
- Estimated healthcare costs of £155-180 million annually in the UK alone
- Seasonal Variation: Some evidence for increased incidence in winter months, possibly related to NSAID use for musculoskeletal conditions and respiratory infections
Demographics
| Factor | Details |
|---|---|
| Age | Median age of presentation 65-70 years; incidence and mortality increase exponentially with age. Peak incidence in 7th-8th decades. Younger patients (age less than 60) have significantly better outcomes. [2] |
| Sex | Male:female ratio approximately 2:1 to 2.5:1 across all age groups. Higher in men due to increased prevalence of H. pylori, NSAID use, and alcohol consumption. |
| Socioeconomic Status | Higher incidence in lower socioeconomic groups, linked to H. pylori prevalence, smoking, alcohol use, and NSAID consumption. [15] |
| Ethnicity | Higher rates in Black and Hispanic populations in US studies; Asian populations have lower H. pylori-associated bleeding but increasing NSAID-related bleeding. |
| Comorbidities | Mortality strongly correlated with comorbidity burden. Cardiovascular disease, chronic kidney disease, liver cirrhosis, malignancy, and respiratory disease are major prognostic factors. Death is often due to decompensation of underlying disease rather than exsanguination. [2] |
Risk Factors
Non-Modifiable:
- Advanced age (>60 years)
- Male sex
- Previous history of UGIB or peptic ulcer disease (strongest predictor of recurrence)
- History of H. pylori infection
- Blood group O (associated with increased peptic ulcer risk)
Modifiable/Environmental:
| Risk Factor | Relative Risk | Mechanism | Clinical Notes |
|---|---|---|---|
| NSAID Use | 3-5× increased risk | COX-1 inhibition reduces gastric mucosal prostaglandin synthesis, impairing mucosal defense. Dose-dependent effect. | Risk persists with all NSAIDs including selective COX-2 inhibitors (though lower). Short-term use (even 1-7 days) increases risk. |
| H. pylori Infection | 2-3× for peptic ulcer bleeding | Chronic active gastritis, disruption of mucus layer, increased acid secretion in duodenal ulcer. | Eradication reduces rebleeding risk by 75%. Testing and eradication mandatory after ulcer bleeding. [13] |
| Anticoagulants | 2-3× increased risk and severity | Warfarin, DOACs increase both risk and severity of bleeding. Not causative but unmask underlying lesions and worsen hemorrhage. | Reversal agents: Vitamin K and PCC for warfarin; idarucizumab for dabigatran; andexanet alfa for Xa inhibitors. |
| Antiplatelet Agents | Aspirin 2-3×; DAPT 3-5× | Irreversible platelet inhibition (aspirin) or P2Y12 receptor blockade (clopidogrel, ticagrelor). Combined effect with DAPT. | Aspirin alone increases risk modestly; dual antiplatelet therapy substantially increases risk. |
| Alcohol Excess | 2-4× increased risk | Direct mucosal injury (gastritis), increases gastric acid secretion, portal hypertension (cirrhosis), coagulopathy. | Both acute intoxication and chronic use are risk factors. |
| Smoking | 1.5-2× increased risk | Impairs ulcer healing, increases acid secretion, reduces mucosal blood flow, decreases prostaglandin synthesis. | Dose-dependent; cessation improves healing. |
| Corticosteroids | Controversial; 1.5-2× when combined with NSAIDs | Possible impairment of mucosal defense; most evidence suggests increased risk only when combined with NSAIDs. | Routine PPI prophylaxis recommended if combined with NSAIDs. |
| SSRIs | 1.5-2× increased risk | Platelet serotonin depletion impairs hemostasis. Synergistic with NSAIDs and antiplatelet agents. | Consider gastroprotection in high-risk patients. |
| Anticoagulation plus Antiplatelet | 5-13× increased risk | Synergistic effect on bleeding risk. | Requires careful risk-benefit assessment; PPI prophylaxis strongly recommended. |
Medical Conditions Increasing Risk:
- Chronic kidney disease (impaired platelet function, uremia)
- Liver cirrhosis (portal hypertension, coagulopathy, thrombocytopenia)
- Critical illness (stress ulceration in ICU patients)
- Previous gastric surgery (marginal ulceration)
- Zollinger-Ellison syndrome (gastrinoma)
- Systemic mastocytosis (histamine excess)
3. Aetiology & Pathophysiology
Causes of Upper GI Bleeding
UGIB is classified into non-variceal (80-90%) and variceal (10-20%) sources.
Non-Variceal Causes
| Cause | Frequency | Key Features |
|---|---|---|
| Peptic Ulcer Disease | 40-50% | Most common cause. Gastric ulcers (usually lesser curve, antrum) and duodenal ulcers (first part of duodenum). Associated with H. pylori and NSAIDs. |
| Gastric/Duodenal Erosions | 15-20% | Superficial mucosal breaks not penetrating muscularis mucosa. Often NSAID-related, alcohol, or stress-induced. Usually multiple. |
| Mallory-Weiss Tear | 5-10% | Longitudinal mucosal laceration at gastroesophageal junction from forceful vomiting/retching. Often alcohol-related. Usually self-limiting. |
| Esophagitis | 5-8% | Severe reflux esophagitis (LA grade C/D), pill esophagitis (bisphosphonates, NSAIDs, potassium), infectious (CMV, HSV in immunocompromised). |
| Dieulafoy Lesion | 1-2% | Large caliber submucosal artery (1-3mm) that erodes through intact mucosa. Causes torrential intermittent arterial bleeding. Most common in proximal stomach, lesser curve. |
| Vascular Ectasia | 2-5% | Gastric antral vascular ectasia (GAVE/watermelon stomach), angiodysplasia. Associated with cirrhosis, chronic kidney disease, aortic stenosis. |
| Upper GI Malignancy | 2-5% | Gastric adenocarcinoma, lymphoma, GIST. Usually chronic bleeding; acute hemorrhage uncommon. |
| Post-Endoscopic | 1-2% | Post-polypectomy, post-EMR/ESD (endoscopic mucosal resection/submucosal dissection), post-PEG insertion. |
| Aortoenteric Fistula | less than 1% | Rare but catastrophic. Secondary (post-AAA repair) or primary. Herald bleed followed by massive hemorrhage. High index of suspicion in previous aortic surgery. |
Variceal Causes
| Type | Frequency | Features |
|---|---|---|
| Esophageal Varices | 90% of variceal bleeding | Secondary to portal hypertension (HVPG >12 mmHg). Size (small/medium/large) and red signs predict bleeding risk. |
| Gastric Varices | 10% of variceal bleeding | Classified by Sarin classification: GOV1 (gastroesophageal, continuation of esophageal), GOV2 (fundal, continuation), IGV1 (isolated fundal, most common and highest bleeding risk). |
| Portal Hypertensive Gastropathy | Associated finding | Mosaic pattern on endoscopy. Chronic oozing rather than acute hemorrhage. |
Molecular and Cellular Pathophysiology
Peptic Ulcer Disease: Acid-Peptic Injury
Normal Mucosal Defense: The gastroduodenal mucosa maintains integrity through multiple protective mechanisms:
- Mucus-bicarbonate layer: Gel layer traps bicarbonate creating pH gradient (luminal pH 1-2 → mucosal surface pH 6-7)
- Epithelial barrier: Tight junctions between epithelial cells prevent H+ back-diffusion
- Mucosal blood flow: Delivers oxygen, nutrients, bicarbonate; removes H+ and toxins
- Prostaglandins (PGE2, PGI2): Maintain blood flow, stimulate mucus/bicarbonate secretion, promote epithelial proliferation
NSAID-Induced Ulceration:
- NSAIDs inhibit cyclooxygenase-1 (COX-1) → decreased prostaglandin synthesis
- Reduces mucus and bicarbonate secretion
- Decreases mucosal blood flow
- Impairs epithelial repair and proliferation
- Direct topical injury to epithelium
- Uncouples oxidative phosphorylation in mitochondria
H. pylori-Induced Ulceration:
- Chronic active gastritis with neutrophil infiltration
- Urease production → ammonia → direct mucosal injury and increased gastrin release
- Disruption of mucus layer by bacterial proteases and phospholipases
- Inflammatory cytokines (IL-1β, IL-8, TNF-α) impair mucosal defense
- Increased acid secretion in duodenal ulcer (antral-predominant gastritis → increased gastrin)
- Gastric ulcer: corpus-predominant gastritis → atrophy → decreased mucosal resistance
Bleeding Mechanism:
- Progressive ulcer deepening through mucosa → submucosa → erosion into submucosal or deeper arteries
- Gastroduodenal artery branches most commonly involved
- Acid-pepsin digestion of exposed vessel wall prevents effective hemostasis
- Arterial bleeding more severe than venous; vessel size correlates with bleeding severity
Variceal Bleeding: Portal Hypertension Hemodynamics
Portal Hypertension Pathophysiology:
- Normal portal pressure: 5-10 mmHg (HVPG 3-5 mmHg)
- Portal hypertension: HVPG >5 mmHg
- Varix formation: HVPG >10 mmHg
- High risk of variceal bleeding: HVPG >12 mmHg
Hemodynamic Changes:
- Increased resistance: Cirrhosis → hepatic fibrosis → sinusoidal compression → increased intrahepatic resistance (structural component)
- Intrahepatic vasoconstriction: Decreased nitric oxide, increased endothelin-1, increased sympathetic tone (functional component)
- Splanchnic vasodilation: Increased NO, prostacyclin, glucagon → increased portal blood flow (hyperdynamic circulation)
- Collateral formation: Portosystemic shunts develop, including esophageal varices (left gastric vein → esophageal venous plexus)
Variceal Rupture:
- Laplace's law: Wall tension = (Pressure × Radius) / Wall thickness
- Increased pressure, increased varix diameter, and thin vessel walls → high wall tension
- Variceal pressure exceeds wall tensile strength → rupture
- Exacerbating factors: straining, coughing, large meals increasing splanchnic flow
Impaired Hemostasis in Cirrhosis:
- Thrombocytopenia (splenic sequestration, decreased thrombopoietin)
- Decreased clotting factor synthesis (II, VII, IX, X, protein C/S)
- Dysfibrinogenemia
- Increased fibrinolysis
- These create a "rebalanced" but fragile hemostatic state that decompensates with bleeding
Mallory-Weiss Tear Pathophysiology
- Forceful vomiting/retching → acute increase in intra-abdominal pressure
- Sudden distension of gastroesophageal junction during retching
- Mucosal laceration parallel to axis of esophagus (longitudinal)
- Typically at or just below gastroesophageal junction (85%)
- Tear extends through mucosa and submucosa but not muscularis propria
- Bleeding from submucosal vessels
- Majority (80-90%) stop spontaneously due to local tamponade
Dieulafoy Lesion Pathophysiology
- Congenital persistence of large caliber (1-3mm diameter) submucosal artery
- Artery runs unusually close to mucosal surface
- Minor mucosal erosion (possibly from acid, NSAIDs, local trauma) exposes arterial lumen
- No associated ulcer crater
- Causes pulsatile arterial hemorrhage
- Intermittent bleeding: clot forms → dislodges → rebleeds
- Difficult to diagnose endoscopically between bleeding episodes (no visible lesion when not bleeding)
The Physiological Response to Hemorrhage
The body's response to acute blood loss follows predictable stages:
Stage 1: Hypovolemia and Reduced Cardiac Output
- Intravascular volume depletion
- Decreased venous return → decreased preload
- Decreased stroke volume and cardiac output
- Tissue hypoperfusion begins
Stage 2: Sympathetic Activation (Compensated Shock)
- Baroreceptors (carotid sinus, aortic arch) detect decreased pressure
- Activation of sympathetic nervous system
- Catecholamine release (epinephrine, norepinephrine)
- Physiological effects:
- Tachycardia (increased HR to maintain cardiac output)
- Peripheral vasoconstriction (skin, muscle, splanchnic) → blood shunted to vital organs
- Maintains blood pressure initially (compensated state)
- Increased myocardial contractility
Stage 3: Transcapillary Refill
- Arteriolar vasoconstriction reduces capillary hydrostatic pressure
- Oncotic pressure gradient draws interstitial fluid into intravascular space
- This process takes 6-12 hours
- Critical clinical implication: Initial hemoglobin may be falsely normal/high despite significant blood loss
- True anemia becomes apparent only after fluid shifts or crystalloid resuscitation
Stage 4: Renal Compensation
- Decreased renal perfusion → activation of RAAS (renin-angiotensin-aldosterone system)
- Aldosterone → sodium and water retention
- ADH (vasopressin) release → water retention
- Oliguria develops (urine output less than 0.5 mL/kg/hr)
Stage 5: Decompensation (Uncompensated Shock)
- Compensatory mechanisms fail with ongoing bleeding (typically >30-40% blood volume loss)
- Hypotension develops
- Severe tissue hypoxia → metabolic acidosis → lactic acidosis
- Cellular injury and organ dysfunction:
- Acute kidney injury
- Myocardial ischemia/infarction (especially with underlying coronary disease)
- Hepatic ischemia
- Cerebral hypoperfusion
- Coagulopathy develops (dilutional, consumptive, hypothermia)
- Death from cardiovascular collapse or multi-organ failure
Classification of Hemorrhagic Shock (ATLS):
| Class | Blood Loss | HR | BP | RR | Urine Output | Mental Status |
|---|---|---|---|---|---|---|
| I | less than 15% (less than 750 mL) | less than 100 | Normal | 14-20 | >30 mL/hr | Anxious |
| II | 15-30% (750-1500 mL) | 100-120 | Normal | 20-30 | 20-30 mL/hr | Anxious, mildly confused |
| III | 30-40% (1500-2000 mL) | 120-140 | Decreased | 30-40 | 5-15 mL/hr | Confused |
| IV | >40% (>2000 mL) | >140 | Severely decreased | >35 | Anuric | Lethargic, unresponsive |
4. Clinical Presentation
Cardinal Symptoms
Hematemesis
- Vomiting of blood, indicating upper GI source
- Fresh red blood: Active, ongoing bleeding; suggests arterial source or very rapid bleeding
- "Coffee-ground" vomitus: Blood altered by gastric acid; suggests slower bleeding or bleeding that has stopped
- Volume of hematemesis roughly correlates with severity but is unreliable (some swallowed blood, dilution with gastric contents)
- Large volume hematemesis (>500 mL) indicates severe, life-threatening bleeding
Melena
- Black, tarry, sticky, foul-smelling (offensive odor) stools
- Requires 50-100 mL of blood in upper GI tract
- Indicates upper GI bleeding in >90% of cases (can be small bowel or right colon)
- Bacterial degradation of hemoglobin → hematin (black pigment)
- Odor is characteristic: "sweet, sickly, distinctive" – once smelled, never forgotten
- May persist for 48-72 hours after bleeding has stopped
- Clinical caveat: Iron supplements, bismuth, activated charcoal cause black stools but without tarry consistency or characteristic odor
Hematochezia
- Passage of bright red blood or maroon stools per rectum
- Usually indicates lower GI source BUT
- Massive UGIB with rapid transit can present as hematochezia (10-15% of UGIB)
- Suggests severe bleeding with hemodynamic instability
- Requires urgent resuscitation and early endoscopy
Associated Symptoms
| Symptom | Significance | Associated Diagnosis |
|---|---|---|
| Dyspepsia/Epigastric pain | Suggests peptic ulcer disease | PUD, gastritis |
| Pain relieved by eating | Classic for duodenal ulcer | Duodenal ulcer |
| Pain worsened by eating | More suggestive of gastric ulcer | Gastric ulcer |
| Vomiting preceding hematemesis | Suggests Mallory-Weiss tear | Mallory-Weiss tear (forceful vomiting → tear) |
| Retching/vomiting then bleeding | Classic presentation | Mallory-Weiss |
| Dysphagia | Esophageal pathology | Esophagitis, esophageal varices, malignancy |
| Weight loss | Red flag for malignancy | Gastric cancer, lymphoma |
| Syncope/Presyncope | Indicates significant hypovolemia (>15-20% blood volume loss) | Severe bleeding |
| Confusion/Altered mental status | Severe hypovolemia OR hepatic encephalopathy in cirrhotics | Shock, encephalopathy |
| Chest pain/SOB | Myocardial ischemia from anemia/hypotension | ACS precipitated by bleeding |
Signs on Examination
General Inspection
- Pallor: Conjunctival, palmar crease pallor suggests chronic or severe acute anemia
- Diaphoresis: Cold, clammy, sweaty skin indicates shock
- Restlessness/Agitation: Early sign of hypovolemia and tissue hypoperfusion
- Confusion/Drowsiness: Severe hypovolemia or hepatic encephalopathy
Vital Signs
- Tachycardia: Often the earliest sign of volume depletion
- HR >100 bpm suggests >15% blood loss
- HR >120 bpm suggests >30% blood loss
- Hypotension: Systolic BP less than 90 mmHg indicates significant hemorrhage (>30% blood loss, Class III shock)
- Late sign; compensatory mechanisms maintain BP initially
- Postural Hypotension: >20 mmHg drop in systolic BP or >10 mmHg drop in diastolic BP on standing
- Suggests 15-20% volume loss
- May be only sign in young, fit patients with good compensatory reserve
- Tachypnea: Respiratory rate >20/min; compensation for metabolic acidosis
- Hypoxia: SpO2 less than 94%; suggests aspiration, pulmonary edema from resuscitation, or severe shock
Cardiovascular Examination
- Weak, thready pulse
- Prolonged capillary refill time (>2 seconds)
- Cool peripheries (peripheral vasoconstriction)
- Jugular venous pressure: Low/not visible (hypovolemia)
Abdominal Examination
- Epigastric tenderness: Suggests peptic ulcer disease (present in 40-60% of PUD bleeding)
- Peritonism (guarding, rigidity, rebound tenderness): Red flag for perforation – rare but requires urgent surgical consultation
- Hepatomegaly: Chronic liver disease, malignancy
- Splenomegaly: Portal hypertension, cirrhosis
- Ascites: Cirrhosis, portal hypertension; suggests variceal bleed
Stigmata of Chronic Liver Disease (Suggests Variceal Bleed)
- Jaundice: Scleral icterus, skin yellowing
- Spider naevi: >5 in distribution of SVC (chest, arms, face)
- Palmar erythema: Reddening of thenar and hypothenar eminences
- Dupuytren's contracture: Alcohol-related
- Gynaecomastia: Hyperestrogenism in cirrhosis
- Testicular atrophy: Hypogonadism in cirrhosis
- Caput medusae: Dilated periumbilical veins (portosystemic shunt)
- Asterixis (flapping tremor): Hepatic encephalopathy
- Fetor hepaticus: Sweet, musty breath odor; hepatic encephalopathy
Digital Rectal Examination (DRE)
- Mandatory in all suspected GI bleeding
- Confirms presence of melena or hematochezia
- Assesses for rectal/colonic masses
5. Risk Stratification Scores
Risk stratification is critical for determining appropriate level of care, timing of endoscopy, and need for intervention.
Glasgow-Blatchford Score (GBS)
Purpose: Pre-endoscopy risk stratification to identify patients requiring intervention (transfusion, endoscopic therapy, surgery)
Timing: Calculated at presentation, before endoscopy
Parameters and Scoring:
| Parameter | Finding | Score |
|---|---|---|
| Blood Urea (mmol/L) | 6.5-8.0 | 2 |
| 8.0-10.0 | 3 | |
| 10.0-25.0 | 4 | |
| ≥25.0 | 6 | |
| Hemoglobin (g/L) - Men | 120-129 | 1 |
| 100-119 | 3 | |
| less than 100 | 6 | |
| Hemoglobin (g/L) - Women | 100-119 | 1 |
| less than 100 | 6 | |
| Systolic BP (mmHg) | 100-109 | 1 |
| 90-99 | 2 | |
| less than 90 | 3 | |
| Other markers | Pulse ≥100 bpm | 1 |
| Melena present | 1 | |
| Syncope | 2 | |
| Hepatic disease | 2 | |
| Heart failure | 2 |
Total Score Range: 0-23
Interpretation:
- GBS = 0: Very low risk; NPV >99% for need for intervention
- Safe for outpatient management with early elective endoscopy
- May not require hospital admission
- GBS = 1: Low risk; consider outpatient or short observation
- GBS ≥2: Admission required
- GBS ≥6: High risk; likely to need intervention (transfusion, endoscopy, surgery)
- GBS ≥12: Very high risk; requires high-dependency or ICU-level care
Advantages:
- Does not require endoscopy
- Excellent negative predictive value (identifies low-risk patients)
- No subjective variables
- Validated in multiple populations [6]
Limitations:
- Does not predict mortality directly
- Does not distinguish variceal from non-variceal bleeding
Rockall Score
Purpose: Predict mortality and rebleeding risk after endoscopy
Timing: Complete score requires endoscopy findings
Pre-Endoscopy (Clinical) Rockall Score:
| Variable | Score 0 | Score 1 | Score 2 | Score 3 |
|---|---|---|---|---|
| Age | less than 60 | 60-79 | ≥80 | - |
| Shock | No shock (SBP≥100, HRless than 100) | Tachycardia (SBP≥100, HR≥100) | Hypotension (SBPless than 100) | - |
| Comorbidity | None | - | Heart failure, IHD, major comorbidity | Renal failure, liver failure, malignancy |
Complete (Post-Endoscopy) Rockall Score adds:
| Variable | Score 0 | Score 1 | Score 2 |
|---|---|---|---|
| Diagnosis | Mallory-Weiss, no lesion | All other diagnoses | Malignancy |
| Stigmata of bleeding | None, dark spot | - | Blood in upper GI, adherent clot, visible/spurting vessel |
Total Score Range: 0-11
Interpretation:
- Score 0-2: Low risk; mortality less than 0.2%, rebleeding 5%
- Score 3-4: Moderate risk; mortality 5%, rebleeding 11%
- Score 5-6: High risk; mortality 11%, rebleeding 25%
- Score ≥7: Very high risk; mortality 17-41%, rebleeding 42%
Advantages:
- Predicts mortality
- Validated predictor of rebleeding
- Incorporates endoscopic findings
Limitations:
- Requires endoscopy for complete score
- Pre-endoscopy (clinical) score alone has limited utility
- Complex to calculate at bedside
AIMS65 Score
Alternative simple score using:
- Albumin less than 30 g/L (1 point)
- INR >1.5 (1 point)
- Mental status alteration (1 point)
- Systolic BP ≤90 mmHg (1 point)
- Age ≥65 years (1 point)
Score ≥2 associated with increased mortality. Less commonly used than GBS/Rockall.
6. Clinical Examination: The ABCDE Approach
In acute UGIB, systematic assessment follows Advanced Life Support principles.
A - Airway
Assessment:
- Is the patient protecting their own airway?
- Risk of aspiration in massive hematemesis
- Decreased GCS (less than 8) compromises airway protection
- Active vomiting of large volumes of blood
Actions:
- Maintain patency: Head tilt, chin lift, jaw thrust if needed
- Suction equipment immediately available
- Recovery position (left lateral) if decreased consciousness and ongoing hematemesis
- Consider early intubation if:
- GCS less than 8 (inability to protect airway)
- Massive ongoing hematemesis
- Respiratory failure
- Need for emergency endoscopy in unstable patient
- Senior anesthetic support early if airway concerns
B - Breathing
Assessment:
- Respiratory rate (normal 12-20/min; tachypnea >20 suggests compensation for acidosis)
- SpO2 (target >94%)
- Work of breathing, accessory muscle use
- Auscultate chest: Crackles (aspiration pneumonia, pulmonary edema from resuscitation)
Actions:
- High-flow oxygen (15L via non-rebreather mask) initially to all patients with significant bleeding
- Titrate to target SpO2 94-98% (88-92% in COPD)
- Arterial blood gas if evidence of respiratory failure or severe shock (assess lactate, metabolic acidosis)
C - Circulation (The Priority)
Assessment:
- Heart rate, rhythm (tachycardia; AF common in elderly)
- Blood pressure (hypotension late sign)
- Capillary refill time (normal less than 2 seconds)
- Peripheral temperature (cool peripheries indicate vasoconstriction/shock)
- Urine output (less than 0.5 mL/kg/hr indicates poor perfusion)
Actions:
-
IV Access: Two large-bore cannulae (14G or 16G) in antecubital fossae
- Grey (16G) or orange (14G) cannulae
- Allows rapid fluid administration
- Consider central venous access if peripheral access difficult or massive transfusion anticipated
- Intraosseous access if unable to secure IV access rapidly
-
Bloods:
- Full blood count (baseline Hb – may be falsely normal initially)
- Urea & electrolytes (elevated urea:creatinine ratio suggests UGIB)
- Liver function tests (assess for chronic liver disease)
- Coagulation screen (PT, APTT, INR – essential if on anticoagulants or liver disease)
- Lactate (marker of tissue hypoperfusion)
- Group & Save (all patients)
- Crossmatch 2-4 units if evidence of significant bleeding or hemodynamic instability
-
Fluid Resuscitation:
- Crystalloid bolus: 500 mL 0.9% sodium chloride or Hartmann's solution over 15 minutes
- Reassess; repeat if still hemodynamically unstable
- Target MAP ≥65 mmHg, HR less than 100 bpm
- Avoid over-resuscitation: Excessive crystalloid increases rebleeding risk and portal pressure in variceal bleeding
- Permissive hypotension (SBP 90-100 mmHg) acceptable in ongoing bleeding until definitive hemostasis, unless cerebrovascular or coronary disease
-
Blood Transfusion:
- Restrictive strategy: Threshold Hb less than 70 g/L; target 70-90 g/L [8]
- Liberal strategy (Hb less than 80 g/L): Only in patients with acute coronary syndrome or symptomatic cardiovascular disease
- Order blood early; activate major hemorrhage protocol if massive bleeding
- Use leucodepleted, CMV-negative if immunocompromised
-
Correct Coagulopathy:
- Platelets: Transfuse if less than 50 × 10⁹/L (target >50)
- Fresh Frozen Plasma (FFP): If PT ratio >1.5; dose 10-15 mL/kg
- Prothrombin Complex Concentrate (PCC): For warfarin reversal (50 units/kg) plus Vitamin K 5-10 mg IV
- Cryoprecipitate: If fibrinogen less than 1.5 g/L
- Reversal agents:
- Warfarin: PCC + Vitamin K
- Dabigatran: Idarucizumab 5g IV
- Rivaroxaban/Apixaban: Andexanet alfa (if available) or PCC
-
Activate Major Hemorrhage Protocol if:
- Ongoing massive bleeding
- Hemodynamic instability despite initial resuscitation
- Expected need for >4 units PRBC
- Provides blood products in balanced ratios (PRBC:FFP:Platelets = 1:1:1)
D - Disability
Assessment:
- GCS (Glasgow Coma Scale): Reduced by hypovolemia (cerebral hypoperfusion) or hepatic encephalopathy
- Pupil size and reactivity
- Blood glucose (exclude hypoglycemia)
- Signs of hepatic encephalopathy: Asterixis, confusion, drowsiness
Actions:
- Treat hypoglycemia if present
- Consider hepatic encephalopathy if known liver disease and reduced consciousness (lactulose, rifaximin)
- Protect airway if GCS less than 8
E - Exposure
Assessment:
- Full abdominal examination: Tenderness, peritonism, hepatosplenomegaly, ascites
- Stigmata of chronic liver disease (see above)
- Digital Rectal Examination (DRE): Mandatory to confirm melena/hematochezia
Actions:
- Keep patient warm (prevent hypothermia which worsens coagulopathy)
- Nasogastric tube insertion controversial:
- Not routinely recommended
- May be useful to clear stomach before endoscopy in massive bleeding
- Avoid in suspected variceal bleeding (may precipitate further bleeding)
7. Investigations
Bedside Tests
| Test | Findings | Significance |
|---|---|---|
| ECG | Tachycardia, ischemic changes (ST depression, T wave inversion) | Myocardial ischemia from anemia/hypotension; baseline for comparison |
| ABG | Metabolic acidosis (↓pH, ↓HCO₃, ↑lactate) | Severity of shock; lactate >4 mmol/L indicates severe hypoperfusion |
| Urine Output | Oliguria (less than 0.5 mL/kg/hr or less than 30 mL/hr) | Renal hypoperfusion; acute kidney injury |
Laboratory Investigations
| Test | Expected Findings | Clinical Notes |
|---|---|---|
| Full Blood Count | ↓Hb (may be normal initially), ↓MCV (if chronic), ↓platelets (cirrhosis) | Hb may lag true blood loss by 6-24 hours until fluid shifts. Serial Hb measurements essential. |
| Urea & Electrolytes | ↑Urea disproportionate to creatinine | Urea:creatinine ratio >100:1 (or >36:1 SI units) suggests UGIB. Blood proteins digested → absorbed → increased urea. |
| Liver Function Tests | ↑Bilirubin, ↓Albumin, ↑ALT/AST (chronic liver disease) | Identify cirrhosis. Hypoalbuminemia (less than 30 g/L) is poor prognostic sign. |
| Coagulation Screen | ↑PT/INR (warfarin, liver disease), ↑APTT (heparin, liver disease) | Essential to guide reversal. INR >1.5 requires correction. |
| Lactate | ↑Lactate (>2 mmol/L; >4 mmol/L severe) | Marker of tissue hypoperfusion and shock. Guides resuscitation adequacy. |
| Group & Save | ABO and Rh typing, antibody screen | Allows rapid crossmatch if transfusion needed. |
| Crossmatch | Compatibility testing | Crossmatch 2-4 units PRBC if significant bleeding. |
Endoscopy (Esophagogastroduodenoscopy, OGD)
The gold standard for diagnosis and therapy.
Timing of Endoscopy
| Clinical Scenario | Timing | Rationale |
|---|---|---|
| Suspected variceal bleeding | Urgent: Within 12 hours | Early variceal band ligation reduces rebleeding and mortality. [14] |
| Hemodynamically unstable | Urgent: After initial resuscitation, within 12-24 hours | Allows diagnosis and therapeutic intervention. Resuscitate first. |
| Hemodynamically stable | Standard: Within 24 hours | Optimal balance of safety and effectiveness. [11] |
| GBS = 0, low risk | Elective: Outpatient within 1-2 weeks | Safe to defer; very low intervention risk. |
Pre-Endoscopy Preparation:
- NPO (nil by mouth) for 2-4 hours if possible (aspiration risk vs. urgency)
- Correct coagulopathy (target INR less than 2.5, platelets >50 × 10⁹/L)
- Prokinetic agent (erythromycin 250 mg IV 30-60 min pre-procedure) may improve visualization by promoting gastric emptying if large blood clot burden
- Consider airway protection (intubation) if massive hematemesis, decreased GCS, or high aspiration risk
- Ensure resuscitation equipment and IV access secured
Endoscopic Findings & Forrest Classification (Peptic Ulcers)
Forrest Classification predicts rebleeding risk and guides therapy:
| Class | Description | Rebleeding Risk (Without Therapy) | Endoscopic Therapy Required |
|---|---|---|---|
| Ia | Active spurting hemorrhage | 90% | Yes - Urgent |
| Ib | Active oozing hemorrhage | 70-80% | Yes - Urgent |
| IIa | Non-bleeding visible vessel | 50% | Yes |
| IIb | Adherent clot | 25-30% | Consider - Remove clot and treat underlying lesion |
| IIc | Hematin-covered base (flat pigmented spot) | 10% | No |
| III | Clean-based ulcer | less than 5% | No |
Endoscopic Therapeutic Techniques
Injection Therapy:
- Dilute adrenaline (epinephrine) 1:10,000 or 1:20,000
- Mechanism: Local vasoconstriction, volume tamponade
- Inject 1-2 mL aliquots in 4 quadrants around bleeding point
- Should NOT be used as monotherapy (high rebleeding rate); combine with mechanical or thermal therapy
Mechanical Therapy:
- Hemoclips (through-the-scope clips): Mechanical apposition of bleeding vessel; effective for Forrest Ia, Ib, IIa
- Endoloop: Detachable snare for pedunculated lesions
- Hemostatic powders (Hemospray): Topical agent forming mechanical barrier; useful for diffuse oozing or when other methods fail
Thermal Therapy:
- Heater probe: Contact thermal coagulation
- Bipolar electrocoagulation (Gold Probe, Bicap): Electrical coagulation
- Argon plasma coagulation (APC): Non-contact thermal; useful for vascular ectasias
Variceal-Specific Therapy:
- Esophageal variceal band ligation (EVL): First-line; bands applied from gastroesophageal junction upward
- Sclerotherapy: Injection of sclerosant (ethanolamine, sodium tetradecyl sulfate); second-line due to higher complication rate
- Gastric variceal obliteration: Cyanoacrylate glue injection (N-butyl-2-cyanoacrylate) for fundal varices (GOV2, IGV1)
Imaging
CT Angiography (CTA)
Indications:
- Ongoing bleeding with negative or non-diagnostic endoscopy
- Hemodynamically unstable patient unable to undergo endoscopy safely
- Suspected aortoenteric fistula
Findings:
- Active contrast extravasation (requires active bleeding >0.5-1 mL/min)
- Vascular malformations
- Masses
Advantages:
- Non-invasive; rapid
- Can localize bleeding site
- May guide interventional radiology
Limitations:
- Requires active bleeding at time of scan
- Contrast nephropathy risk in AKI
- Radiation exposure
Interventional Radiology
Angiography with Embolization:
- Reserved for refractory bleeding despite endoscopic therapy
- Selective catheterization of bleeding vessel (left gastric, gastroduodenal artery)
- Coil embolization or gelfoam
- Success rate 50-90%; rebleeding risk 10-30%
- Risk of bowel ischemia (especially if concurrent hypotension)
Other Imaging
- Chest X-ray (CXR): Erect CXR if suspected perforation (free air under diaphragm)
- Abdominal X-ray: Limited utility; may show bowel obstruction or toxic megacolon (rare complications)
8. Management
Management Algorithm
ACUTE UPPER GI BLEEDING SUSPECTED
↓
┌──────────────────────────────────────────────────┐
│ IMMEDIATE ASSESSMENT & RESUSCITATION (ABCDE) │
│ • Airway: Protect if GCS less than 8, massive hematemesis│
│ • Breathing: High-flow O₂, target SpO₂ >94% │
│ • Circulation: 2× Large-bore IVs (14-16G) │
│ - Bloods: FBC, U&E, LFT, Coag, G&S, Lactate │
│ - Crystalloid bolus if SBP less than 90 or HR >100 │
│ - Crossmatch 2-4 units PRBC │
│ • Disability: GCS, glucose, encephalopathy? │
│ • Exposure: Full exam, DRE for melena │
└──────────────────────────────────────────────────┘
↓
┌──────────────────────────────────────────────────┐
│ RISK STRATIFICATION (Glasgow-Blatchford Score) │
├──────────────────────────────────────────────────┤
│ GBS = 0: Low risk → Outpatient OGD │
│ GBS ≥ 1: Admit for inpatient management │
│ GBS ≥ 6: High risk → HDU/ICU, urgent OGD │
└──────────────────────────────────────────────────┘
↓
┌──────────────────────────────────────────────────┐
│ TRANSFUSION STRATEGY │
├──────────────────────────────────────────────────┤
│ • RESTRICTIVE: Hb less than 70 g/L (target 70-90 g/L) │
│ • LIBERAL: Hb less than 80 g/L if ACS/CVD │
│ • Platelets: less than 50 × 10⁹/L │
│ • FFP: INR >1.5 │
│ • Reverse anticoagulation (PCC for warfarin) │
└──────────────────────────────────────────────────┘
↓
┌──────────────────────────────────────────────────┐
│ PRE-ENDOSCOPY MEDICAL THERAPY │
├──────────────────────────────────────────────────┤
│ SUSPECTED NON-VARICEAL: │
│ • Hold NSAIDs, anticoagulants │
│ • PPI can be given but benefit pre-OGD limited │
│ │
│ SUSPECTED VARICEAL (Cirrhosis/CLD stigmata): │
│ • TERLIPRESSIN 2 mg IV every 4 hours (or │
│ Somatostatin 250 mcg bolus + 250 mcg/hr) │
│ • PROPHYLACTIC ANTIBIOTICS: Ceftriaxone 1g IV │
│ • DO NOT delay OGD for pharmacotherapy │
└──────────────────────────────────────────────────┘
↓
┌──────────────────────────────────────────────────┐
│ UPPER GI ENDOSCOPY (OGD) │
├──────────────────────────────────────────────────┤
│ TIMING: │
│ • Suspected variceal: Within 12 hours │
│ • Unstable/high-risk: Within 12-24 hours │
│ • Stable: Within 24 hours │
│ • GBS 0: Elective outpatient │
│ │
│ THERAPEUTIC INTERVENTION: │
│ • High-risk stigmata (Forrest Ia, Ib, IIa): │
│ - Adrenaline injection + mechanical/thermal │
│ - Hemoclips, heater probe, APC │
│ • Varices: Band ligation (EVL) or sclerotherapy │
│ • Gastric varices: Cyanoacrylate injection │
└──────────────────────────────────────────────────┘
↓
┌──────────────────────────────────────────────────┐
│ POST-ENDOSCOPY MANAGEMENT │
├──────────────────────────────────────────────────┤
│ NON-VARICEAL ULCER BLEEDING: │
│ • High-dose PPI: Omeprazole 80 mg IV bolus then │
│ 8 mg/hr infusion × 72 hrs (or 40 mg BD PO) │
│ • H. pylori testing (CLO test, histology) │
│ • Repeat OGD if high-risk stigmata │
│ • Gastric ulcer: Repeat OGD in 6-8 weeks with │
│ biopsy to exclude malignancy │
│ │
│ VARICEAL BLEEDING: │
│ • Continue vasoactive drugs × 2-5 days │
│ • Continue antibiotics × 7 days │
│ • Secondary prophylaxis: Non-selective │
│ β-blocker (propranolol, carvedilol) │
│ • Repeat EVL every 2-4 weeks until obliteration │
│ • TIPS if refractory bleeding │
└──────────────────────────────────────────────────┘
↓
┌──────────────────────────────────────────────────┐
│ REBLEEDING (Occurs in 10-15%) │
├──────────────────────────────────────────────────┤
│ • Repeat resuscitation (ABCDE) │
│ • Repeat OGD with endoscopic therapy │
│ • If refractory (2nd endoscopic failure): │
│ - Interventional radiology (angioembolization)│
│ - Surgery (rare; high mortality) │
│ - TIPS (for variceal bleeding) │
└──────────────────────────────────────────────────┘
↓
┌──────────────────────────────────────────────────┐
│ DISCHARGE & FOLLOW-UP │
├──────────────────────────────────────────────────┤
│ • H. pylori eradication therapy (if positive) │
│ • Long-term PPI if ongoing NSAID/aspirin │
│ • Plan for restarting anticoagulation (assess │
│ thrombotic vs. bleeding risk; usually 7-14d) │
│ • Repeat OGD for gastric ulcer (rule out CA) │
│ • Variceal surveillance (repeat EVL, β-blocker) │
└──────────────────────────────────────────────────┘
Resuscitation & Transfusion Strategy
Fluid Resuscitation
Crystalloids:
- 0.9% sodium chloride or Hartmann's solution (balanced crystalloid)
- Bolus 500 mL over 15 minutes if hemodynamically unstable
- Reassess and repeat as needed
- Target: MAP ≥65 mmHg, HR less than 100 bpm, urine output >0.5 mL/kg/hr
Avoid Over-Resuscitation:
- Excessive crystalloid increases rebleeding risk (increased portal pressure, dilutional coagulopathy)
- Permissive hypotension (SBP 90-100 mmHg) acceptable in ongoing bleeding until hemostasis achieved
- Exception: Maintain adequate perfusion pressure if known cerebrovascular or coronary artery disease
Blood Transfusion: Restrictive vs. Liberal
Landmark Evidence: Villanueva et al. (2013) [8]
- RCT in 921 patients with acute UGIB
- Restrictive (Hb threshold less than 70 g/L) vs. Liberal (Hb threshold less than 90 g/L)
- Results: Restrictive strategy associated with:
- Reduced mortality (5% vs. 9%, p=0.02)
- Reduced rebleeding (10% vs. 16%, p=0.01)
- Reduced adverse events
- Benefit most pronounced in variceal bleeding
Current Transfusion Thresholds:
- Restrictive (Standard): Transfuse if Hb less than 70 g/L; target 70-90 g/L
- Liberal (Selective): Transfuse if Hb less than 80 g/L in:
- Acute coronary syndrome
- Symptomatic cardiovascular disease
- Ongoing bleeding with hemodynamic instability
Mechanism of Harm with Liberal Transfusion:
- Increased blood volume → increased portal pressure → increased variceal bleeding
- Transfusion increases blood viscosity and induces vasoconstriction
- Dilutional effect on coagulation factors
Platelet Transfusion
- Threshold: less than 50 × 10⁹/L
- Target: >50 × 10⁹/L
- 1 adult therapeutic dose (ATD) typically raises platelet count by 20-30 × 10⁹/L
- Consider higher threshold (>70) for ongoing severe bleeding or planned high-risk intervention
Coagulation Factor Replacement
Fresh Frozen Plasma (FFP):
- Indication: PT ratio >1.5 or fibrinogen less than 1.5 g/L
- Dose: 10-15 mL/kg (typically 4 units for average adult)
- Contains all clotting factors
Cryoprecipitate:
- Indication: Fibrinogen less than 1.5 g/L
- Rich in fibrinogen, Factor VIII, vWF, Factor XIII
- Dose: 2 pools (10 units)
Prothrombin Complex Concentrate (PCC):
- Rapid reversal of warfarin (contains Factors II, VII, IX, X)
- Dose: 25-50 units/kg depending on INR
- Give with Vitamin K 5-10 mg IV (slow reversal over 12-24 hours)
Anticoagulant Reversal
| Anticoagulant | Reversal Agent | Dose | Notes |
|---|---|---|---|
| Warfarin | PCC + Vitamin K | PCC 25-50 u/kg IV + Vit K 5-10 mg IV | PCC provides immediate reversal; Vitamin K required for sustained effect |
| Dabigatran | Idarucizumab | 5 g IV (2 × 2.5g boluses) | Specific reversal agent; highly effective |
| Rivaroxaban, Apixaban | Andexanet alfa (if available) or PCC | 400-800 mg IV or PCC 50 u/kg | Andexanet alfa is specific Xa inhibitor reversal but limited availability; PCC second-line |
| Heparin (UFH) | Protamine sulfate | 1 mg per 100 units heparin | Give slowly (max 50 mg); risk of allergic reaction |
| LMWH | Protamine (partial) | 1 mg per 100 anti-Xa units | Less effective than for UFH; partial reversal only |
Pharmacotherapy
Proton Pump Inhibitors (PPIs)
Pre-Endoscopy:
- Controversial; limited evidence for benefit before endoscopy
- May reduce stigmata of recent hemorrhage but does not affect mortality or rebleeding
- Can be given but should not delay endoscopy
Post-Endoscopy (NON-VARICEAL ulcer bleeding with high-risk stigmata):
Landmark Evidence: Lau et al. (2000) [16]
- High-dose IV omeprazole after endoscopic therapy for bleeding peptic ulcers
- Reduced rebleeding (6.7% vs. 22.5%, pless than 0.001)
- Reduced need for surgery
Dosing:
- IV regimen (preferred for high-risk): Omeprazole 80 mg IV bolus, then 8 mg/hr continuous infusion for 72 hours
- Oral regimen (alternative): Omeprazole 40 mg BD (twice daily) PO/NG
- After 72 hours: Transition to oral PPI (omeprazole 20-40 mg daily, lansoprazole 30 mg daily)
- Continue for 4-8 weeks (duodenal ulcer) or 8-12 weeks (gastric ulcer)
Mechanism:
- Increases gastric pH >6, optimal for platelet aggregation and clot stability
- Inhibits pepsin activity (pepsin degrades clot)
PPI NOT indicated post-endoscopy if:
- Forrest IIc or III (low-risk stigmata)
- Non-ulcer source (Mallory-Weiss, vascular ectasia)
Vasoactive Drugs (VARICEAL Bleeding)
Must be started immediately if variceal bleeding suspected (before endoscopy).
Terlipressin:
- Synthetic vasopressin analogue
- Dose: 2 mg IV every 4 hours for 2-5 days
- Mechanism: Splanchnic vasoconstriction → decreased portal blood flow → reduced portal pressure
- Reduces mortality and rebleeding [14]
- Side effects: Coronary/peripheral ischemia (caution in IHD), hyponatremia
Somatostatin:
- Dose: 250 mcg IV bolus, then 250 mcg/hr continuous infusion for 2-5 days
- Mechanism: Splanchnic vasoconstriction, reduced portal flow
- Fewer cardiovascular side effects than terlipressin
- Limited availability in some countries
Octreotide:
- Synthetic somatostatin analogue
- Dose: 50 mcg IV bolus, then 50 mcg/hr continuous infusion
- Longer half-life than somatostatin
- Used widely in US; less evidence than terlipressin
Prophylactic Antibiotics (VARICEAL Bleeding)
Mandatory in all cirrhotic patients with GI bleeding.
Landmark Evidence:
- Reduces bacterial infections (11% vs. 26%)
- Reduces rebleeding
- Reduces mortality (9% vs. 20%) [14]
- Spontaneous bacterial peritonitis common in variceal bleeding
Dosing:
- Ceftriaxone 1 g IV once daily for 7 days (first-line)
- Norfloxacin 400 mg BD PO (alternative if low risk)
- Start immediately on presentation
- Continue for 7 days or until discharge
Tranexamic Acid: DO NOT USE
HALT-IT Trial (2020) [10]:
- Large RCT (12,009 patients with GI bleeding)
- Tranexamic acid (1g bolus + 3g infusion over 24h) vs. placebo
- Results:
- No reduction in mortality (18.5% vs. 19.4%, p=0.07)
- No reduction in bleeding deaths
- Increased venous thromboembolism (0.8% vs. 0.4%, p=0.008)
- Conclusion: Tranexamic acid should NOT be used in UGIB
Endoscopic Therapy
See "Investigations: Endoscopy" section above for detailed techniques.
Key Principles:
- Dual therapy superior to monotherapy (adrenaline injection alone insufficient; must combine with mechanical or thermal)
- Forrest Ia, Ib, IIa require therapy
- Forrest IIb (adherent clot): Remove clot and treat underlying lesion
- Forrest IIc, III: No therapy needed; low rebleeding risk
Refractory or Recurrent Bleeding
Definition: Ongoing bleeding despite endoscopic therapy, or rebleeding after initial control.
Incidence: 10-15% of cases; usually within 72 hours.
Management:
- Resuscitate: Repeat ABCDE assessment, ensure adequate IV access, transfusion
- Repeat endoscopy: Second-look OGD with repeat hemostatic therapy (success rate 50-70%)
- Interventional radiology: Angiography with embolization (if available and patient suitable)
- Surgery (last resort; high mortality 15-30%):
- Peptic ulcer: Under-running of bleeding vessel, vagotomy + pyloroplasty, partial gastrectomy
- Varices: Esophageal transection (rare; TIPS preferred)
- TIPS (Transjugular Intrahepatic Portosystemic Shunt): For refractory variceal bleeding
- Creates shunt between hepatic and portal veins → reduces portal pressure
- Salvage therapy for failed endoscopic/pharmacologic therapy
- Risk of hepatic encephalopathy (30-40%)
Special Considerations: Restarting Anticoagulation and Antiplatelet Therapy
Key Principle: Balance thrombotic risk vs. rebleeding risk.
Antiplatelet Therapy (Aspirin, Clopidogrel, DAPT):
- High thrombotic risk (recent ACS, coronary stent less than 1 year, especially less than 3 months): Resume aspirin within 1-3 days if hemostasis achieved [17]
- Moderate risk (stable CAD, prior MI, stroke): Resume within 7 days
- DAPT: Discuss with cardiology; aspirin can often resume early, consider delaying P2Y12 inhibitor
- Long-term PPI mandatory if continuing antiplatelet therapy post-UGIB
Anticoagulation (Warfarin, DOACs):
- High thrombotic risk (mechanical heart valve, AF with CHA₂DS₂-VASc ≥5, recent VTE): Resume within 7-14 days
- Moderate risk (AF with CHA₂DS₂-VASc 2-4): Resume within 14-21 days
- Low risk: Resume after 4 weeks or consider withholding
- Consider switching warfarin to DOAC (lower bleeding risk in some studies)
Post-Discharge Management
All Patients:
- Helicobacter pylori eradication if positive (see below)
- Avoid NSAIDs; use alternative analgesia (paracetamol, weak opioids)
- Gastroprotection with PPI if ongoing aspirin/NSAID requirement
- Lifestyle modification: Smoking cessation, reduce alcohol
- Follow-up to ensure ulcer healing
Peptic Ulcer Disease:
- Gastric ulcer: Repeat OGD in 6-8 weeks with multiple biopsies to exclude malignancy (gastric cancer can present as ulcer)
- Duodenal ulcer: Repeat OGD not routinely required (malignancy rare)
Variceal Bleeding:
- Secondary prophylaxis: Non-selective β-blocker (propranolol, carvedilol) to reduce portal pressure
- Repeat EVL every 2-4 weeks until variceal obliteration
- Treat underlying liver disease
- Consider liver transplant assessment if decompensated cirrhosis
9. Helicobacter pylori: Testing and Eradication
Importance: H. pylori present in 60-90% of peptic ulcer bleeding (higher in duodenal than gastric). Eradication reduces ulcer recurrence by 75% and rebleeding risk significantly. [13]
Testing During Acute Bleeding
At Index Endoscopy:
- Rapid Urease Test (CLO test, Campylobacter-like organism test): Biopsy specimen placed in urea-containing gel; urease production by H. pylori changes pH → color change. Fast (minutes to hours). Sensitivity reduced in acute bleeding (50-70% vs. >95% normally) due to blood dilution, recent PPI use.
- Histology: Antral and corpus biopsies; hematoxylin-eosin or special stains (Giemsa, Warthin-Starry). Sensitivity 80-90%; gold standard.
Post-Discharge (if acute tests negative):
- Urea Breath Test (UBT): Non-invasive; sensitivity/specificity >95%. Perform 4-8 weeks after stopping PPI (false negative if on PPI).
- Stool Antigen Test: Non-invasive; sensitivity/specificity 90-95%. Less affected by PPI than UBT.
- Serology: Detects IgG antibodies. Cannot distinguish active from past infection. Not recommended for diagnosis post-treatment (remains positive for years).
Eradication Therapy
First-Line (if low clarithromycin resistance less than 15%):
- PAC Regimen (Proton Pump Inhibitor + Amoxicillin + Clarithromycin):
- PPI (omeprazole 20 mg, lansoprazole 30 mg, or equivalent) BD
- Amoxicillin 1 g BD
- Clarithromycin 500 mg BD
- "Duration: 14 days (superior to 7 days)"
First-Line (if high clarithromycin resistance or penicillin allergy):
- Bismuth Quadruple Therapy:
- PPI BD
- Bismuth subsalicylate 525 mg QID or subcitrate
- Metronidazole 400 mg TDS
- Tetracycline 500 mg QID
- "Duration: 14 days"
Alternative First-Line:
- Concomitant Therapy (PPI + amoxicillin + clarithromycin + metronidazole for 14 days)
- Sequential Therapy (PPI + amoxicillin × 5 days, then PPI + clarithromycin + metronidazole × 5 days)
Confirmation of Eradication:
- Urea Breath Test at least 4 weeks after completing therapy (and 2 weeks off PPI)
- Essential in all patients with ulcer bleeding
If Eradication Fails:
- Second-line therapy (different antibiotic regimen)
- Consider culture and antibiotic sensitivity testing
- Levofloxacin-based triple therapy (PPI + amoxicillin + levofloxacin)
10. Complications
Acute Complications (During Bleeding Episode)
| Complication | Incidence | Mechanism | Management |
|---|---|---|---|
| Rebleeding | 10-15% (within 72 hrs) | Clot dislodgement, failed initial hemostasis | Repeat endoscopy, IR embolization, surgery |
| Hypovolemic Shock | 10-20% | Severe blood loss → tissue hypoperfusion | Aggressive resuscitation, transfusion, vasopressors if refractory |
| Acute Kidney Injury | 15-20% | Prerenal (hypovolemia), ATN (prolonged hypoperfusion) | Fluid resuscitation, avoid nephrotoxins, may require RRT |
| Myocardial Infarction | 2-5% | Anemia + hypotension → myocardial ischemia (especially with underlying CAD) | Transfusion (liberal threshold if ACS), cardiology input, balance transfusion vs. antiplatelet risk |
| Aspiration Pneumonia | 5-10% | Massive hematemesis → aspiration of blood/gastric contents | Airway protection, antibiotics, respiratory support |
| Hepatic Encephalopathy | 20-30% in cirrhotics | Protein load (blood) in gut → ammonia production | Lactulose, rifaximin, treat precipitant |
| Spontaneous Bacterial Peritonitis | 10-15% in cirrhotics | Bacterial translocation during bleed | Prophylactic antibiotics (ceftriaxone) |
| Cerebrovascular Event | less than 2% | Hypoperfusion or thrombosis (if holding anticoagulation) | Supportive care, careful anticoagulation management |
| Disseminated Intravascular Coagulation (DIC) | Rare | Massive transfusion, prolonged shock | Treat underlying cause, replace clotting factors, platelets |
| Death | 5-10% overall; 15-20% variceal | Exsanguination, multi-organ failure, comorbidity decompensation | Intensive resuscitation, early definitive hemostasis |
Endoscopic Procedure-Related Complications
| Complication | Incidence | Risk Factors | Management |
|---|---|---|---|
| Perforation | less than 1% | Aggressive therapy, friable tissue, therapeutic intervention | Surgical repair (often); some small perforations managed conservatively (NBM, antibiotics, clips) |
| Worsening Bleeding | 1-2% | Inappropriate therapy, trauma | Repeat hemostatic therapy, consider alternative modality |
| Aspiration | 1-3% | Decreased GCS, massive hematemesis | Prevention: Airway protection pre-procedure if high risk. Treatment: Antibiotics, respiratory support |
Long-Term Complications
| Complication | Incidence | Mechanism | Prevention/Management |
|---|---|---|---|
| Recurrent Ulcer Disease | 30-50% without H. pylori eradication; 5-10% with eradication | Persistent H. pylori, ongoing NSAID use | H. pylori eradication, avoid NSAIDs, long-term PPI if needed |
| Gastric Cancer (if gastric ulcer) | 2-5% | Ulcerated gastric adenocarcinoma misdiagnosed as benign | Mandatory repeat OGD with biopsy at 6-8 weeks for gastric ulcer |
| Recurrent Variceal Bleeding | 60% at 1 year without prophylaxis | Persistent portal hypertension | Secondary prophylaxis: β-blockers, repeat EVL, TIPS if refractory |
| Chronic Anemia | Variable | Recurrent/persistent occult bleeding | Iron supplementation, investigate source, address underlying cause |
11. Prognosis
Overall Mortality
- Overall mortality: 5-10% [2,4]
- Variceal bleeding: 15-20% (significantly higher than non-variceal)
- Non-variceal bleeding: 5-8%
Mortality trends: Has remained relatively stable over past 30 years despite advances in therapy, reflecting:
- Aging population
- Increased comorbidity burden
- Medication-related bleeding (anticoagulants, antiplatelets)
Predictors of Mortality
Patient Factors:
- Age >60 years (strongest predictor; mortality doubles with each decade)
- Comorbidities: Cardiovascular disease, chronic kidney disease, malignancy, cirrhosis
- Hemodynamic instability at presentation (SBP less than 90 mmHg, HR >100 bpm)
- In-hospital onset of bleeding (mortality 3-4× higher)
Bleeding Factors:
- Variceal source (vs. non-variceal)
- High-risk stigmata on endoscopy (Forrest Ia, Ib)
- Large volume bleeding
- Transfusion requirement >4 units
Laboratory Factors:
- Hemoglobin less than 80 g/L
- Albumin less than 30 g/L
- Creatinine elevation (acute kidney injury)
- Lactate >4 mmol/L
- INR >1.5
Rebleeding Risk
Incidence: 10-15% overall; highest risk in first 72 hours
Predictors of Rebleeding:
- Endoscopic stigmata (Forrest classification): Ia/Ib 70-90%, IIa 50%, IIb 25-30% without therapy
- Ulcer characteristics: Size >2 cm, posterior duodenal ulcer (gastroduodenal artery), lesser curve gastric ulcer (left gastric artery)
- Variceal size: Large varices with red signs
- Failed initial hemostasis
- Inadequate PPI therapy post-ulcer bleeding
- Continued NSAID/anticoagulant use
Impact of Rebleeding:
- Mortality 10× higher in patients who rebleed
- Increases length of hospital stay
- Necessitates repeat intervention
Rockall Score Mortality Prediction
| Rockall Score | Mortality | Rebleeding Risk |
|---|---|---|
| 0-2 | less than 0.2% | 5% |
| 3-4 | 5% | 11% |
| 5-6 | 11% | 25% |
| ≥7 | 17-41% | 42% |
Long-Term Outcomes
If H. pylori eradicated and NSAIDs avoided:
- Ulcer recurrence: less than 5% at 1 year
- Rebleeding: less than 3% at 1 year
If H. pylori persists or NSAIDs continued:
- Ulcer recurrence: 30-50% at 1 year
- Rebleeding: 10-15% at 1 year
Variceal Bleeding:
- Rebleeding risk 60% at 1 year without secondary prophylaxis
- Reduced to 20-30% with β-blockers + repeat EVL
- 5-year survival for cirrhotic patients with variceal bleed: 30-50% (largely dependent on underlying liver disease severity)
12. Evidence & Guidelines
Landmark Trials
-
Villanueva C, et al. (2013) - Restrictive vs. Liberal Transfusion in UGIB [8]
- N Engl J Med. 2013;368(1):11-21. PMID: 23281973. DOI: 10.1056/NEJMoa1211801
- RCT, 921 patients with acute UGIB
- Restrictive (Hb less than 70 g/L) vs. Liberal (Hb less than 90 g/L) transfusion
- Results: Restrictive strategy reduced mortality (5% vs. 9%, HR 0.55, p=0.02), rebleeding (10% vs. 16%, p=0.01), and adverse events
- Conclusion: Restrictive transfusion strategy superior; now standard of care
-
Lau JY, et al. (2000) - High-Dose PPI After Endoscopic Therapy [16]
- N Engl J Med. 2000;343(5):310-316. PMID: 10933737. DOI: 10.1056/NEJM200008033430501
- RCT, 240 patients with bleeding peptic ulcers after successful endoscopic therapy
- High-dose IV omeprazole (80 mg bolus + 8 mg/hr × 72h) vs. placebo
- Results: Reduced rebleeding (6.7% vs. 22.5%, pless than 0.001), surgery (0.8% vs. 6.7%, p=0.01)
- Conclusion: High-dose PPI reduces rebleeding after endoscopic ulcer therapy
-
HALT-IT Trial (2020) - Tranexamic Acid in GI Bleeding [10]
- Lancet. 2020;395(10241):1927-1936. PMID: 32563442. DOI: 10.1016/S0140-6736(20)30848-5
- RCT, 12,009 patients with acute GI bleeding
- Tranexamic acid (1g bolus + 3g over 24h) vs. placebo
- Results: No reduction in death (18.5% vs. 19.4%, RR 0.99, p=0.07), increased VTE (0.8% vs. 0.4%, p=0.008)
- Conclusion: Tranexamic acid should NOT be used in GI bleeding
-
Gralnek IM, et al. (2021) - ESGE Guideline on Non-Variceal UGIB [11]
- Endoscopy. 2021;53(3):300-332. PMID: 33561863. DOI: 10.1055/a-1310-0898
- Updated European guideline on diagnosis and management
- Key recommendations: Early endoscopy (less than 24h), dual therapy for high-risk stigmata, restrictive transfusion, high-dose PPI
-
Barkun AN, et al. (2019) - International Consensus on Non-Variceal UGIB [18]
- Ann Intern Med. 2019;171(11):805-822. PMID: 31634906. DOI: 10.7326/M19-1795
- International consensus group recommendations
- Emphasizes risk stratification (GBS), pre-endoscopy resuscitation, endoscopic dual therapy, post-endoscopy PPI
-
de Franchis R, et al. (2022) - Baveno VII Consensus on Portal Hypertension [19]
- J Hepatol. 2022;76(4):959-974. PMID: 35120870. DOI: 10.1016/j.jhep.2021.12.022
- Updated recommendations for variceal bleeding management
- Key points: Vasoactive drugs + antibiotics pre-endoscopy, EVL preferred over sclerotherapy, TIPS for refractory bleeding
-
Stanley AJ, Laine L (2019) - Management of Acute UGIB [20]
- BMJ. 2019;364:l536. PMID: 30910853. DOI: 10.1136/bmj.l536
- Comprehensive review and practical guidance
- Algorithm-based approach to risk stratification and management
Clinical Practice Guidelines
NICE CG141 (2012, updated 2016): Acute Upper Gastrointestinal Bleeding in Over 16s [21]
- Pre-endoscopy risk assessment with validated tools (GBS)
- Immediate endoscopy (less than 2 hours) if hemodynamically unstable after resuscitation
- Routine endoscopy within 24 hours
- Clip or thermal coagulation with adrenaline injection (not adrenaline alone)
- Restrictive transfusion (Hb 70-100 g/L)
ESGE Guideline (2021): Endoscopic Management of Non-Variceal UGIH [11]
- GBS recommended for pre-endoscopy risk stratification
- Endoscopy within 24 hours for most; less than 12 hours for high-risk
- Dual therapy (injection + mechanical/thermal) for Forrest Ia, Ib, IIa
- High-dose PPI (80 mg IV bolus + 8 mg/hr infusion) after endoscopic therapy
- Consider second-look endoscopy for very high-risk lesions
ASGE Guideline: Management of Patients with Ulcer Bleeding
- Similar recommendations to ESGE
- Emphasizes importance of H. pylori testing and eradication
- Guidance on restarting antiplatelet/anticoagulant therapy
British Society of Gastroenterology: Management of Variceal Hemorrhage [22]
- Tripathi D, et al. Gut. 2015;64(11):1680-1704. PMID: 25887380
- Vasoactive drugs (terlipressin) + antibiotics (ceftriaxone) immediately on admission
- Urgent endoscopy (less than 12 hours)
- EVL preferred over sclerotherapy
- TIPS for refractory bleeding or high-risk patients
Quality Indicators for UGIB Management
Evidence-based quality metrics:
- Time to risk stratification: GBS calculated within 4 hours of presentation
- Time to endoscopy: >80% of patients undergo endoscopy within 24 hours; high-risk within 12 hours
- Appropriate transfusion: Restrictive strategy (Hb less than 70 g/L) in >80% of patients
- Dual endoscopic therapy: Used in >90% of high-risk stigmata (Forrest Ia, Ib, IIa)
- Post-endoscopy PPI: High-dose IV PPI in >90% of ulcer bleeding with high-risk stigmata
- H. pylori testing: Performed in >95% of peptic ulcer bleeding
- H. pylori eradication: Eradication therapy prescribed and confirmation testing arranged in >90% of H. pylori-positive cases
- Anticoagulant reversal: Appropriate reversal agents administered within 4 hours when indicated
- Variceal bleeding: Vasoactive drugs + antibiotics in >90% of suspected variceal bleeding before endoscopy
- Rebleeding management: Repeat endoscopy or escalation to IR/surgery within 12 hours of rebleeding
13. Patient Education & Counseling
Understanding Upper GI Bleeding
Simple Explanation: "Upper gastrointestinal bleeding means bleeding from your stomach, esophagus (food pipe), or the first part of your small intestine. This can happen due to ulcers, irritation of the stomach lining, or enlarged blood vessels (varices). The main signs are vomiting blood or passing black, tar-like stools."
Medication Safety
NSAIDs (Non-Steroidal Anti-Inflammatory Drugs):
- Avoid ibuprofen, naproxen, diclofenac unless specifically directed by your doctor
- These medications increase risk of stomach ulcers and bleeding
- Use paracetamol (acetaminophen) for pain relief instead
- If NSAIDs are essential, take with a stomach-protecting medication (PPI)
Aspirin and Blood Thinners:
- If prescribed aspirin for heart protection, discuss with your doctor before stopping (stopping may increase heart attack/stroke risk)
- Your doctor will carefully decide when to restart blood thinners after your bleeding has stopped
- Never adjust doses yourself
Helicobacter pylori Eradication
Why It Matters: "Helicobacter pylori is a bacteria that lives in your stomach and causes ulcers. If we don't get rid of it, your ulcer will come back in 70% of cases. With successful treatment, the recurrence risk drops to less than 5%."
Treatment Course:
- Take ALL antibiotics exactly as prescribed for the full 14 days, even if feeling better
- Complete the course even if you experience minor side effects (nausea, diarrhea, metallic taste)
- Avoid alcohol during treatment (interacts with metronidazole)
- You will need a breath test 4-6 weeks after finishing antibiotics to confirm the bacteria is gone
Warning Signs - When to Seek Immediate Help
Return to the Emergency Department immediately if you experience:
- Vomiting blood (bright red or coffee-ground appearance)
- Black, tarry, sticky stools with offensive smell
- Fresh red blood in stools
- Feeling faint, dizzy, or lightheaded
- Rapid heartbeat or shortness of breath
- Severe abdominal pain
- Confusion or altered mental state
Lifestyle Modifications
Alcohol:
- Reduce or eliminate alcohol consumption (irritates stomach lining, increases ulcer risk, worsens liver disease)
- If you have liver disease with varices, complete abstinence is essential
Smoking:
- Smoking impairs ulcer healing and increases recurrence risk
- Smoking cessation resources and support should be offered
Diet:
- No specific "ulcer diet" required
- Avoid foods that trigger symptoms (spicy, fatty foods for some people)
- Small, regular meals may be more comfortable than large meals
- Caffeine and chocolate may worsen symptoms in some individuals
Follow-Up Care
Gastric Ulcer:
- You will need a repeat endoscopy (camera test) in 6-8 weeks
- This is essential to ensure the ulcer has healed and to rule out stomach cancer (which can look like an ulcer)
- Multiple biopsies (small tissue samples) will be taken
Duodenal Ulcer:
- Repeat endoscopy usually not needed (cancer very rare)
- Focus on H. pylori eradication and avoiding NSAIDs
Variceal Bleeding:
- You will need repeat endoscopy every 2-4 weeks to treat varices until they are gone
- Take prescribed beta-blocker medication daily (reduces pressure in the varices)
- Manage underlying liver disease
- Consider liver transplant assessment if cirrhosis is advanced
Medication After Discharge
Proton Pump Inhibitor (PPI):
- Continue for 4-12 weeks depending on ulcer location and size
- Take 30 minutes before breakfast for best effect
- Long-term use (>8 weeks) only if ongoing need (continuing aspirin/NSAID, severe reflux disease)
- Discuss duration with your doctor
14. Differential Diagnosis
When a patient presents with hematemesis or melena, consider:
Upper GI Sources (Proximal to Ligament of Treitz)
| Diagnosis | Key Distinguishing Features | Prevalence |
|---|---|---|
| Peptic Ulcer Disease | Epigastric pain, history of NSAID use or H. pylori, relieved/worsened by eating | 40-50% |
| Gastric/Duodenal Erosions | NSAID use, alcohol, stress (ICU patients), usually less severe bleeding | 15-20% |
| Esophageal/Gastric Varices | Known cirrhosis, stigmata of chronic liver disease, massive bleeding | 10-20% |
| Mallory-Weiss Tear | Forceful vomiting/retching preceding hematemesis, often alcohol-related | 5-10% |
| Esophagitis | Heartburn, dysphagia, odynophagia; pill esophagitis (bisphosphonates, NSAIDs) | 5-8% |
| Dieulafoy Lesion | Massive intermittent arterial bleeding, no ulcer on endoscopy, proximal stomach lesser curve | 1-2% |
| Gastric Antral Vascular Ectasia (GAVE) | Chronic iron-deficiency anemia, "watermelon stomach" on endoscopy, portal hypertension or systemic sclerosis | 1-2% |
| Upper GI Malignancy | Weight loss, early satiety, anemia, dysphagia; usually chronic bleeding, acute hemorrhage uncommon | 2-5% |
| Aortoenteric Fistula | Previous AAA repair, herald bleed followed by massive hemorrhage, high mortality | less than 1% |
Lower GI Sources That May Mimic UGIB
| Diagnosis | Key Features |
|---|---|
| Right Colon Bleeding | Can cause melena if slow transit; more commonly hematochezia |
| Small Bowel Bleeding | Obscure GI bleeding; angiodysplasia, Meckel's diverticulum, small bowel tumors |
Non-GI Causes of "Blood-Stained" Vomitus
| Diagnosis | Key Distinguishing Features |
|---|---|
| Epistaxis with Swallowed Blood | History of nosebleed, blood in nasopharynx, no hematemesis |
| Hemoptysis | Coughing up blood (not vomiting), frothy/bright red, respiratory symptoms |
| Factitious (Self-Induced) | Inconsistent history, psychiatric history, blood not digested |
Red Flag Features Requiring Urgent Assessment
- Aortoenteric fistula: History of aortic graft surgery, herald bleed (small bleed followed by massive hemorrhage), pulsatile abdominal mass
- Gastric cancer: Weight loss, early satiety, persistent vomiting, dysphagia, palpable mass
- Perforation: Severe, sudden-onset abdominal pain, peritonism, free air on imaging
15. Examination Viva & OSCE Scenarios
Opening Statement (Structured Approach)
Example Viva Opening: "Upper gastrointestinal bleeding is a common medical emergency defined as bleeding from a source proximal to the ligament of Treitz, presenting with hematemesis or melena. It affects approximately 100 per 100,000 adults annually with an overall mortality of 5-10%, rising to 15-20% for variceal bleeding. The commonest cause is peptic ulcer disease, accounting for 40-50% of cases. My approach would involve immediate resuscitation using an ABCDE framework, risk stratification with the Glasgow-Blatchford Score, and early endoscopy within 24 hours for diagnosis and therapy."
Common Viva Questions & Model Answers
Q1: How would you initially assess and manage a patient presenting with hematemesis?
Model Answer: "I would use a systematic ABCDE approach:
Airway: Assess for airway compromise, particularly if massive ongoing hematemesis or reduced GCS. Have suction available and consider early intubation if GCS less than 8.
Breathing: Assess respiratory rate, oxygen saturation, and auscultate for aspiration. Provide high-flow oxygen.
Circulation: This is the priority. I would:
- Establish two large-bore IV cannulae (14 or 16 gauge)
- Take bloods including FBC, U&E, LFTs, coagulation screen, group and save, and crossmatch
- Start fluid resuscitation with crystalloid boluses if hemodynamically unstable
- Note the elevated urea to creatinine ratio which suggests upper GI bleeding
- Apply restrictive transfusion strategy with threshold of hemoglobin below 70 g/L
Disability: Assess GCS and look for signs of hepatic encephalopathy if cirrhosis suspected.
Exposure: Perform full abdominal examination and digital rectal examination to confirm melena and assess for stigmata of chronic liver disease.
I would then risk-stratify using the Glasgow-Blatchford Score and arrange urgent endoscopy within 24 hours, or within 12 hours if high-risk features or suspected variceal bleeding."
Q2: What is the Glasgow-Blatchford Score and how do you interpret it?
Model Answer: "The Glasgow-Blatchford Score is a validated pre-endoscopy risk stratification tool that predicts the need for intervention—namely transfusion, endoscopic therapy, or surgery. It does not require endoscopy findings, making it immediately applicable at presentation.
The score uses eight variables: blood urea, hemoglobin (gender-specific cutoffs), systolic blood pressure, heart rate above 100, presence of melena, syncope, hepatic disease, and heart failure. Scores range from 0 to 23.
The key interpretation is that a score of zero has a negative predictive value exceeding 99% for needing intervention, identifying patients safe for outpatient management with elective endoscopy. A score of 6 or higher indicates high risk requiring admission and likely intervention. This tool is superior to clinical judgment alone in identifying low-risk patients."
Q3: Describe the Forrest classification and its clinical significance.
Model Answer: "The Forrest classification categorizes peptic ulcer bleeding based on endoscopic appearance, predicting rebleeding risk and guiding endoscopic therapy decisions.
It consists of three main categories:
- Forrest I (Active bleeding): Ia is spurting hemorrhage (90% rebleeding risk), Ib is oozing (70-80% risk)
- Forrest II (Recent bleeding): IIa is a non-bleeding visible vessel (50% risk), IIb is an adherent clot (25-30% risk), IIc is a flat pigmented spot (10% risk)
- Forrest III: Clean ulcer base with less than 5% rebleeding risk
Classes Ia, Ib, and IIa definitely require endoscopic therapy with dual modality treatment—adrenaline injection combined with either mechanical therapy like clips or thermal therapy like heater probe. Adrenaline alone is insufficient. Class IIb with adherent clot should have the clot removed and underlying lesion treated if high-risk stigmata present. Classes IIc and III are low-risk and do not require endoscopic therapy."
Q4: What are the key differences in managing variceal versus non-variceal upper GI bleeding?
Model Answer: "Variceal bleeding requires specific management distinct from non-variceal bleeding:
Variceal-specific interventions:
- Immediate vasoactive drugs before endoscopy—terlipressin 2mg IV every 4 hours or somatostatin analogs—which reduce portal pressure and control bleeding
- Prophylactic antibiotics—ceftriaxone 1g IV daily—proven to reduce mortality, infection, and rebleeding in cirrhotic patients
- More urgent endoscopy—within 12 hours rather than 24
- Specific endoscopic therapy—esophageal variceal band ligation or sclerotherapy for esophageal varices, cyanoacrylate injection for gastric varices
- Different transfusion targets—restrictive strategy is especially important as overtransfusion increases portal pressure
- Salvage options—if refractory, TIPS (transjugular intrahepatic portosystemic shunt) rather than surgery
Additionally, variceal bleeding requires treatment of precipitants like spontaneous bacterial peritonitis, and initiation of secondary prophylaxis with non-selective beta-blockers plus repeat band ligation. The mortality is significantly higher at 15-20% compared to 5-8% for non-variceal bleeding."
Q5: What did the HALT-IT trial demonstrate and what are the implications?
Model Answer: "The HALT-IT trial, published in Lancet in 2020, was a large multicenter randomized controlled trial of over 12,000 patients with acute gastrointestinal bleeding. It compared tranexamic acid—a 1g bolus followed by 3g infusion over 24 hours—versus placebo.
The results showed that tranexamic acid did not reduce mortality (18.5% versus 19.4%, p=0.07), did not reduce death from bleeding, but significantly increased venous thromboembolism risk (0.8% versus 0.4%, p=0.008).
The implication is clear: tranexamic acid should not be used routinely in gastrointestinal bleeding, despite theoretical benefits. This contrasts with its proven efficacy in trauma and postpartum hemorrhage. The trial definitively answered the question and changed practice—tranexamic acid is now contraindicated in this context."
Q6: When and how would you restart anticoagulation after an upper GI bleed?
Model Answer: "This requires careful individualized risk-benefit assessment balancing thrombotic risk versus rebleeding risk.
High thrombotic risk patients—such as mechanical heart valves, AF with CHA2DS2-VASc score of 5 or higher, or recent VTE—should restart anticoagulation within 7-14 days once hemostasis is secured.
Moderate risk patients—AF with CHA2DS2-VASc 2-4—can resume at 14-21 days.
Low risk patients may defer for 4 weeks or withhold altogether if bleeding risk outweighs benefit.
For antiplatelet therapy post-acute coronary syndrome or recent stenting (especially within 3 months), aspirin should be restarted within 1-3 days given the very high thrombotic risk, ideally after discussion with cardiology.
All patients restarting antithrombotic therapy should receive long-term PPI gastroprotection. I would also ensure H. pylori eradication if positive, counsel avoidance of NSAIDs, and arrange appropriate follow-up. Some patients may benefit from switching from warfarin to a DOAC given lower bleeding risk in some studies."
Common OSCE Scenarios
Scenario 1: Acute Management Station
- Patient with massive hematemesis, tachycardia, hypotension
- Tasks: ABCDE assessment, establish IV access, order investigations, initiate resuscitation, arrange urgent endoscopy
- Examiner looks for: Systematic approach, recognition of shock, appropriate fluid/blood product use, early escalation
Scenario 2: History Taking Station
- Patient presenting with melena for 2 days, on aspirin and NSAIDs for arthritis
- Tasks: Focused history, risk factor assessment, medication review, assess hemodynamic status
- Examiner looks for: Screening for red flags, NSAID/aspirin history, previous ulcer disease, features of chronic liver disease
Scenario 3: Counseling Station
- Post-endoscopy counseling after diagnosis of H. pylori-positive duodenal ulcer
- Tasks: Explain diagnosis, explain H. pylori eradication, medication counseling, lifestyle advice, follow-up plan
- Examiner looks for: Clear communication, checking understanding, medication safety (avoid NSAIDs), importance of compliance and confirmation testing
Scenario 4: Data Interpretation
- Endoscopy images showing Forrest IIa ulcer, varices, Dieulafoy lesion
- Tasks: Describe findings, classify lesion, predict rebleeding risk, outline management
- Examiner looks for: Correct interpretation, Forrest classification knowledge, appropriate therapy selection
Common Mistakes to Avoid
❌ Waiting for hemoglobin to drop before transfusing shocked patient — Hb lags true blood loss; clinical assessment of shock is key
❌ Using adrenaline injection alone — Must combine with mechanical (clips) or thermal therapy; adrenaline monotherapy has high rebleeding rate
❌ Delaying endoscopy in variceal bleeding to "stabilize" — Vasoactive drugs and antibiotics should be started immediately, and endoscopy performed within 12 hours even if unstable (after basic resuscitation)
❌ Over-transfusing — Liberal transfusion increases mortality and rebleeding, especially in variceal bleeding; adhere to restrictive strategy (Hb less than 70 g/L)
❌ Forgetting digital rectal examination — Essential to confirm melena and support diagnosis; frequently omitted
❌ Not testing or eradicating H. pylori — Failure to eradicate increases recurrence from 70% to less than 5%; must test and confirm eradication
❌ Giving tranexamic acid — HALT-IT trial showed no benefit and increased VTE risk; should not be used
❌ Not giving prophylactic antibiotics in cirrhotic patients — Reduces mortality, SBP, and rebleeding; mandatory in all cirrhotics with GI bleeding
16. References
-
Hearnshaw SA, et al. Acute upper gastrointestinal bleeding in the UK: patient characteristics, diagnoses and outcomes in the 2007 UK audit. Gut. 2011;60(10):1327-1335. PMID: 21490373. DOI: 10.1136/gut.2010.228437
-
Lanas A, et al. Time trends in upper gastrointestinal bleeding mortality across 56 countries. Aliment Pharmacol Ther. 2019;49(11):1331-1341. PMID: 30957278. DOI: 10.1111/apt.15241
-
van Leerdam ME. Epidemiology of acute upper gastrointestinal bleeding. Best Pract Res Clin Gastroenterol. 2008;22(2):209-224. PMID: 18346679. DOI: 10.1016/j.bpg.2007.10.011
-
Jairath V, Barkun AN. The overall approach to the management of upper gastrointestinal bleeding. Gastrointest Endosc Clin N Am. 2015;25(3):425-443. PMID: 26142029. DOI: 10.1016/j.giec.2015.02.002
-
Garcia-Tsao G, Bosch J. Management of varices and variceal hemorrhage in cirrhosis. N Engl J Med. 2010;362(9):823-832. PMID: 20200386. DOI: 10.1056/NEJMra0901512
-
Stanley AJ, et al. Comparison of risk scoring systems for patients presenting with upper gastrointestinal bleeding: international multicentre prospective study. BMJ. 2017;356:i6432. PMID: 28053181. DOI: 10.1136/bmj.i6432
-
Rockall TA, et al. Risk assessment after acute upper gastrointestinal haemorrhage. Gut. 1996;38(3):316-321. PMID: 8675081. DOI: 10.1136/gut.38.3.316
-
Villanueva C, et al. Transfusion strategies for acute upper gastrointestinal bleeding. N Engl J Med. 2013;368(1):11-21. PMID: 23281973. DOI: 10.1056/NEJMoa1211801
-
Wells M, et al. The management of acute upper gastrointestinal bleeding. BMJ. 2022;378:e070439. PMID: 36041763. DOI: 10.1136/bmj-2022-070439
-
HALT-IT Trial Collaborators. Effects of a high-dose 24-h infusion of tranexamic acid on death and thromboembolic events in patients with acute gastrointestinal bleeding (HALT-IT): an international randomised, double-blind, placebo-controlled trial. Lancet. 2020;395(10241):1927-1936. PMID: 32563442. DOI: 10.1016/S0140-6736(20)30848-5
-
Gralnek IM, et al. Endoscopic diagnosis and management of nonvariceal upper gastrointestinal hemorrhage (NVUGIH): European Society of Gastrointestinal Endoscopy (ESGE) Guideline - Update 2021. Endoscopy. 2021;53(3):300-332. PMID: 33561863. DOI: 10.1055/a-1310-0898
-
Lanas A, et al. Non-variceal upper gastrointestinal bleeding. Nat Rev Dis Primers. 2018;4:18020. PMID: 29892030. DOI: 10.1038/nrdp.2018.20
-
Chey WD, et al. ACG Clinical Guideline: Treatment of Helicobacter pylori Infection. Am J Gastroenterol. 2017;112(2):212-239. PMID: 28071659. DOI: 10.1038/ajg.2016.563
-
Tripathi D, et al. UK guidelines on the management of variceal haemorrhage in cirrhotic patients. Gut. 2015;64(11):1680-1704. PMID: 25887380. DOI: 10.1136/gutjnl-2015-309262
-
Laine L, Yang H, Chang SC, Datto C. Trends for incidence of hospitalization and death due to GI complications in the United States from 2001 to 2009. Am J Gastroenterol. 2012;107(8):1190-1195. PMID: 22688850. DOI: 10.1038/ajg.2012.168
-
Lau JY, et al. Effect of intravenous omeprazole on recurrent bleeding after endoscopic treatment of bleeding peptic ulcers. N Engl J Med. 2000;343(5):310-316. PMID: 10933737. DOI: 10.1056/NEJM200008033430501
-
Douketis JD, et al. Perioperative management of antithrombotic therapy: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012;141(2 Suppl):e326S-e350S. PMID: 22315266. DOI: 10.1378/chest.11-2298
-
Barkun AN, et al. Management of Nonvariceal Upper Gastrointestinal Bleeding: Guideline Recommendations From the International Consensus Group. Ann Intern Med. 2019;171(11):805-822. PMID: 31634906. DOI: 10.7326/M19-1795
-
de Franchis R, et al. Baveno VII - Renewing consensus in portal hypertension. J Hepatol. 2022;76(4):959-974. PMID: 35120870. DOI: 10.1016/j.jhep.2021.12.022
-
Stanley AJ, Laine L. Management of acute upper gastrointestinal bleeding. BMJ. 2019;364:l536. PMID: 30910853. DOI: 10.1136/bmj.l536
-
NICE. Acute upper gastrointestinal bleeding in over 16 s: management. Clinical Guideline [CG141]. 2012 (updated 2016). Available at: https://www.nice.org.uk/guidance/cg141
-
Tripathi D, et al. UK guidelines on the management of variceal haemorrhage in cirrhotic patients. Gut. 2015;64(11):1680-1704. PMID: 25887380. DOI: 10.1136/gutjnl-2015-309262
Summary
Upper gastrointestinal bleeding is a common medical emergency requiring immediate systematic assessment, evidence-based resuscitation (restrictive transfusion strategy with hemoglobin threshold less than 70 g/L), risk stratification using the Glasgow-Blatchford Score, and timely endoscopy for diagnosis and therapy. Non-variceal bleeding (80-90% of cases), most commonly from peptic ulcers, is managed with dual endoscopic therapy (adrenaline plus mechanical/thermal modality) for high-risk stigmata and high-dose PPI infusion post-endoscopy. Variceal bleeding (10-20% of cases) requires immediate vasoactive drugs (terlipressin) and prophylactic antibiotics before endoscopy, with band ligation as first-line endoscopic therapy. H. pylori eradication is mandatory for peptic ulcer bleeding. Tranexamic acid should not be used. Overall mortality remains 5-10%, with variceal bleeding carrying higher mortality (15-20%). Rebleeding occurs in 10-15% and dramatically worsens prognosis, necessitating repeat endoscopy, interventional radiology, or surgery.
Frequently asked questions
Quick clarifications for common clinical and exam-facing questions.
When should I seek emergency care for upper gastrointestinal bleeding (adult)?
Seek immediate emergency care if you experience any of the following warning signs: Hematemesis (bright red or coffee-ground), Melena (black, tarry, foul-smelling stools), Hemodynamic instability (SBP less than 90 mmHg, HR >100 bpm), Syncope or near-syncope, Altered mental status (suggests severe hypovolemia or encephalopathy), Active, ongoing bleeding, Severe abdominal pain (suggests perforation).
Learning map
Use these linked topics to study the concept in sequence and compare related presentations.
Prerequisites
Start here if you need the foundation before this topic.
- Gastrointestinal Anatomy
Consequences
Complications and downstream problems to keep in mind.
- Hypovolemic Shock
- Hepatic Encephalopathy