Urinary Tract Infection in Adults
Urinary tract infection (UTI) is bacterial infection of any component of the urinary system, most commonly the bladder (... MRCP exam preparation.
What matters first
Urinary tract infection (UTI) is bacterial infection of any component of the urinary system, most commonly the bladder (... MRCP exam preparation.
Signs of pyelonephritis (loin pain, fever less than 38CC)
17 Jan 2026
Generated educational material; verify before clinical use.
Visible references section
See the concept before reading it
Study the key anatomy, imaging, and decision pathways as full teaching plates.
Clinical board
A visual summary of the highest-yield teaching signals on this page.
Urgent signals
Safety-critical features pulled from the topic metadata.
- Signs of pyelonephritis (loin pain, fever less than 38CC)
- Signs of sepsis (SIRS criteria)
- Acute urinary retention
- Male UTI (complicated by definition)
Exam focus
Current exam surfaces linked to this topic.
- MRCP
Linked comparisons
Differentials and adjacent topics worth opening next.
- Acute Interstitial Nephritis
- Urethritis and Sexually Transmitted Infections
Content status and exam context
This page is AI-generated educational content. It may contain errors or omissions and is not a substitute for current guidelines, local protocols, senior clinical judgement, or professional medical advice.
MedVellum does not claim an individual clinician reviewer, board certification, or professional credential for this page unless a future version names a real, verifiable reviewer.
Clinical explanation and evidence
Urinary Tract Infection in Adults
1. Clinical Overview
Summary
Urinary tract infection (UTI) is bacterial infection of any component of the urinary system, most commonly the bladder (cystitis) or kidneys (pyelonephritis). UTI represents one of the most frequent bacterial infections encountered in clinical practice, with an estimated 150 million cases worldwide annually. [1] Women are disproportionately affected, with 50-60% experiencing at least one UTI during their lifetime. [2] The vast majority of community-acquired UTIs are caused by uropathogenic Escherichia coli (75-95%), which ascends from the perineum via the urethra to colonise the bladder. [3]
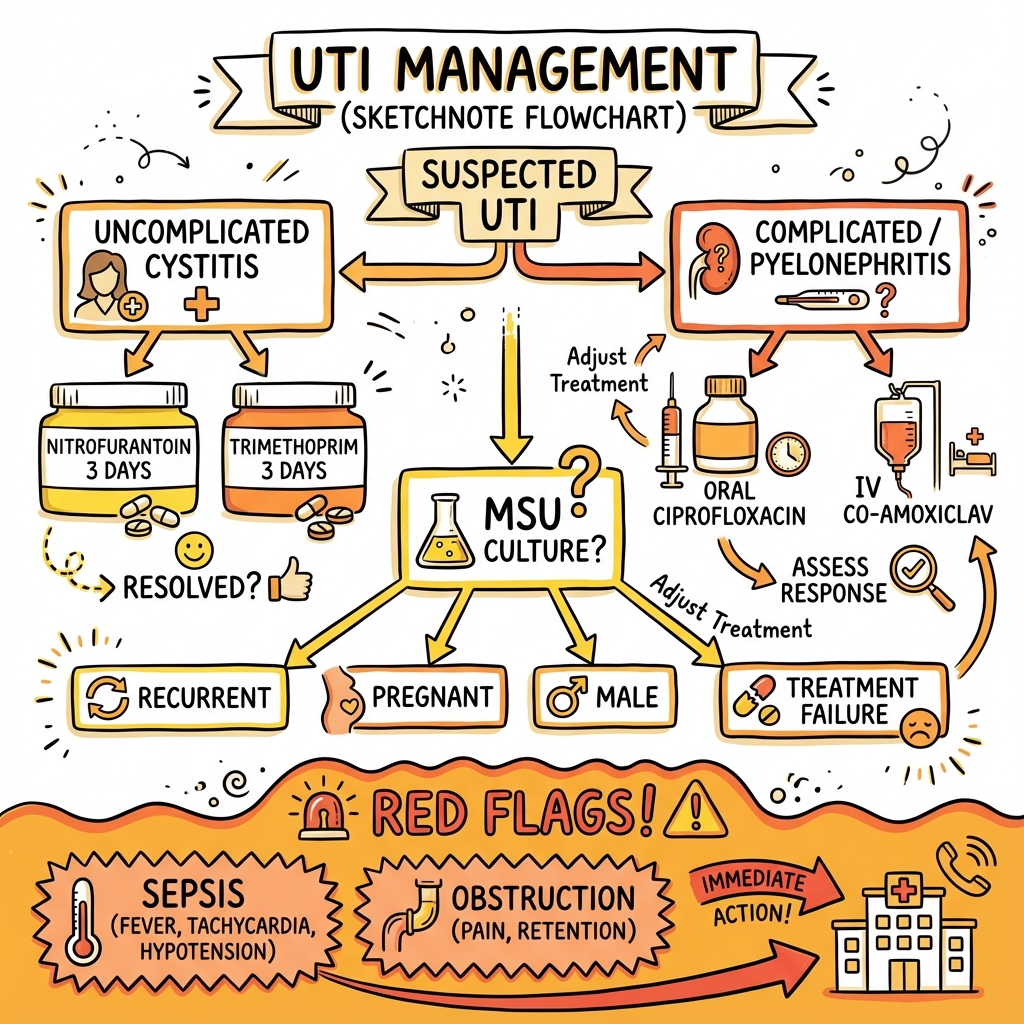
Lower UTI (cystitis) presents with dysuria, urinary frequency, urgency, suprapubic pain, and occasionally haematuria, typically without systemic features. Upper UTI (pyelonephritis) manifests with loin pain, fever, rigors, nausea, and vomiting, representing a more severe infection with potential for bacteraemia and sepsis. The distinction between uncomplicated and complicated UTI is critical for management: uncomplicated UTI occurs in otherwise healthy, non-pregnant women with normal urinary anatomy, while complicated UTI occurs in males, pregnant women, or those with structural abnormalities, immunosuppression, catheters, or renal transplants. [4]
Diagnosis is predominantly clinical for uncomplicated cystitis in women, though urine dipstick provides rapid supportive evidence (positive nitrites and leucocyte esterase). Urine culture is mandatory for complicated UTI, pregnancy, treatment failure, or recurrent infections, with significant bacteriuria defined as greater than 10^5 colony-forming units per millilitre (CFU/mL) on midstream urine. [5] Empirical antibiotic therapy remains the cornerstone of management, with short-course treatment (3 days) of nitrofurantoin or trimethoprim highly effective for uncomplicated cystitis. [6] Pyelonephritis requires longer treatment duration (7-14 days) with broader-spectrum agents such as fluoroquinolones or co-amoxiclav.
A crucial and often misunderstood concept is asymptomatic bacteriuria, defined as the presence of bacteria in urine without clinical symptoms. Treatment is only indicated in pregnancy (due to risk of pyelonephritis and preterm labour) and before urological procedures; unnecessary treatment in other contexts drives antimicrobial resistance without clinical benefit. [7] Recurrent UTI, defined as two or more infections in six months or three or more in twelve months, affects approximately 20-30% of women after an initial UTI and requires investigation for underlying structural abnormalities and consideration of preventative strategies. [8]
Key Facts
- Definition: Bacterial infection of the urinary tract; lower UTI (cystitis: bladder) or upper UTI (pyelonephritis: kidneys)
- Incidence: 150 million cases globally per year; 50-60% of women will experience at least one UTI in their lifetime [1,2]
- Demographics: Female:Male ratio approximately 30:1 in young adults; ratio equalises in elderly due to prostatic disease and catheterisation
- Dominant Pathogen: Escherichia coli accounts for 75-95% of community-acquired UTIs [3]
- Key Distinction: Uncomplicated (healthy non-pregnant women) vs complicated (males, pregnancy, structural abnormalities, immunosuppression, catheters)
- Diagnostic Gold Standard: Urine culture with ≥10^5 CFU/mL significant bacteriuria (though clinical diagnosis often sufficient for simple cystitis)
- First-Line Treatment (Uncomplicated): Nitrofurantoin 100mg BD for 3 days OR Trimethoprim 200mg BD for 3 days [6]
- First-Line Treatment (Pyelonephritis): Ciprofloxacin 500mg BD for 7 days OR co-amoxiclav 625mg TDS for 10-14 days
- Treatment Duration: 3 days for uncomplicated cystitis, 7 days for male UTI, 7 days for pregnancy, 7-14 days for pyelonephritis
- Prognosis: Excellent with treatment; spontaneous resolution in 25-40% of uncomplicated cases; 20-30% recurrence rate within 6 months
Clinical Pearls
Short Course Pearl: Uncomplicated cystitis in women requires only 3 days of treatment. Meta-analyses demonstrate equivalence to 5-7 day courses for clinical cure (OR 0.98, 95% CI 0.71-1.35), while shorter duration reduces side effects and antimicrobial resistance. [6]
Nitrofurantoin Pearl: Nitrofurantoin maintains exceptionally low resistance rates (typically less than 5% for E. coli) due to multiple mechanisms of action and limited use outside UTI. However, avoid if eGFR less than 45 mL/min/1.73m² (reduced urinary concentration and efficacy) and at term in pregnancy (theoretical risk of neonatal haemolysis). [9]
Male UTI Pearl: ALL UTIs in men are considered complicated by definition due to the longer urethra and requirement for an underlying anatomical or functional abnormality. Require minimum 7 days treatment and investigation (ultrasound, post-void residual, PSA if over 50 years). [4]
Asymptomatic Bacteriuria Pearl: Do NOT treat asymptomatic bacteriuria except in pregnancy (20-30% progress to pyelonephritis; risk of preterm labour and low birth weight) and before urological procedures with mucosal trauma. [7] Treatment in other contexts (elderly, catheterised patients, diabetics) provides no benefit and drives resistance.
Catheter Pearl: Catheter-associated bacteriuria is universal after 30 days of catheterisation. Only treat if patient has clinical symptoms (fever, new delirium, haemodynamic instability). Bacteriuria or pyuria alone is NOT an indication for antibiotics. [10]
Dipstick Pearl: Negative nitrites do NOT exclude UTI. Nitrites are produced by gram-negative organisms (E. coli, Klebsiella, Proteus) but NOT by Staphylococcus saprophyticus, Enterococcus, or Pseudomonas. Leucocyte esterase is more sensitive but less specific.
Pregnancy Pearl: Nitrofurantoin is safe in pregnancy except at term (avoid after 36 weeks). Trimethoprim is relatively contraindicated in first trimester (folate antagonist; theoretical teratogenic risk). Fluoroquinolones are absolutely contraindicated throughout pregnancy.
Recurrence vs Relapse Pearl: Recurrence (new infection with different organism or same organism after greater than 2 weeks) is far more common than relapse (same organism within 2 weeks, suggesting treatment failure or persistent source).
Why This Matters Clinically
UTI is one of the most common reasons for antibiotic prescription in primary and secondary care, accounting for approximately 15% of all community-prescribed antibiotics. [11] Inappropriate diagnosis (especially of asymptomatic bacteriuria), suboptimal antibiotic choice, and excessive treatment duration are major drivers of antimicrobial resistance, particularly fluoroquinolone resistance in E. coli which has increased from less than 5% to greater than 15-20% in many regions over two decades. [12]
Antimicrobial stewardship in UTI requires: (1) accurate distinction between symptomatic infection and asymptomatic bacteriuria, (2) appropriate use of urine culture to guide therapy in complicated cases, (3) selection of narrow-spectrum agents with local susceptibility patterns in mind, (4) shortest effective treatment duration, and (5) avoidance of fluoroquinolones as first-line agents to preserve their efficacy for resistant infections and pyelonephritis. The clinical and public health impact of judicious UTI management cannot be overstated in the era of escalating antimicrobial resistance.
2. Epidemiology
Incidence and Prevalence
Urinary tract infection is the second most common infection in humans after respiratory tract infection. Global annual incidence is estimated at 150 million cases, with significant geographic and demographic variation. [1] Population-based studies demonstrate the following epidemiological patterns:
| Population | Annual Incidence | Lifetime Risk | Notes |
|---|---|---|---|
| Women (18-40 years) | 0.5-0.7 episodes per person-year | 50-60% | Peak incidence in sexually active years [2] |
| Women (postmenopausal) | Increases with age | 20% prevalence in > 65 years | Associated with oestrogen deficiency, prolapse |
| Men (18-40 years) | 0.006 episodes per person-year | 5-8% | Rare in healthy young men |
| Men (> 60 years) | Increases 20-fold | 20% over 65 years | Prostatic disease, catheterisation |
| Pregnant women | 2-10% symptomatic UTI | 2-10% ASB progresses to pyelonephritis | Higher risk in second and third trimesters |
| Hospitalised patients | 40% of nosocomial infections | — | Predominantly catheter-associated [10] |
| Nursing home residents | 30-50% prevalence (mostly ASB) | — | High catheterisation rates, functional impairment |
The dramatic female predominance (30:1 ratio in young adults) reflects anatomical differences: the female urethra is approximately 4 cm long compared to 20 cm in males, facilitating bacterial ascension. This ratio equalises in the elderly as prostatic hypertrophy, bladder dysfunction, and catheterisation increase male UTI risk. [13]
Risk Factors
| Category | Risk Factors | Mechanism/Notes |
|---|---|---|
| Female-Specific | Sexual activity (risk increases 60-fold) [14] | Mechanical introduction of periurethral bacteria |
| Spermicide use | Alters vaginal flora, reduces lactobacilli | |
| New sexual partner | Exposure to new bacterial strains | |
| Menopause/oestrogen deficiency | Vaginal pH rises, lactobacilli decrease, uroepithelial changes | |
| Pregnancy | Ureteric dilatation (progesterone), incomplete bladder emptying | |
| Maternal history of UTI | Genetic factors (e.g., TLR polymorphisms, blood group antigens) | |
| Anatomical | Short urethra (females) | Reduced physical barrier to ascension |
| Vesicoureteric reflux | Retrograde flow of infected urine to kidneys | |
| Urinary tract obstruction | Stones, strictures, tumours; impaired washout | |
| Bladder diverticula | Incomplete emptying, urinary stasis | |
| Neurogenic bladder | Incomplete emptying, high residual volumes | |
| Iatrogenic | Urinary catheterisation | Risk 3-7% per day; biofilm formation [10] |
| Recent urological instrumentation | Mucosal trauma, bacterial introduction | |
| Genitourinary surgery | Disrupted anatomy, stents, drains | |
| Host Factors | Diabetes mellitus | Glycosuria (bacterial substrate), impaired neutrophil function, autonomic neuropathy |
| Immunosuppression | Impaired cellular immunity; transplant recipients, chemotherapy, biologics | |
| Renal transplantation | Immunosuppression, obstructed transplant ureter, VUR | |
| Chronic kidney disease | Impaired immune function, incomplete bladder emptying if uraemic | |
| Male-Specific | Benign prostatic hyperplasia | Incomplete emptying, high post-void residual (PVR > 100mL) [15] |
| Prostatitis | Prostatic inflammation and oedema causing obstruction | |
| Uncircumcised status | Higher periurethral bacterial colonisation (controversial) | |
| Anal intercourse | Rectal flora exposure | |
| Behavioural | Delayed voiding | Prolonged bladder colonisation, reduced washout |
| Inadequate fluid intake | Concentrated urine, reduced flushing | |
| Poor perineal hygiene | Increased periurethral bacterial load |
Classification
The most clinically important classification distinguishes uncomplicated from complicated UTI, as this determines investigation strategy, treatment duration, and need for imaging.
| Category | Definition | Examples | Clinical Implications |
|---|---|---|---|
| Uncomplicated UTI | Cystitis in otherwise healthy, premenopausal, non-pregnant woman with normal urinary tract anatomy | Community-acquired cystitis in 25-year-old woman | Empirical 3-day treatment; no imaging; urine culture optional |
| Complicated UTI | UTI in patient with factors increasing risk of treatment failure or severe infection | Males, pregnancy, structural abnormalities, catheters, immunosuppression, renal transplant, diabetes | Urine culture mandatory; 7-14 day treatment; consider imaging; investigate underlying cause |
| Lower UTI (Cystitis) | Infection confined to bladder; no systemic features | Dysuria, frequency, urgency, suprapubic pain | Oral antibiotics; outpatient management |
| Upper UTI (Pyelonephritis) | Infection of renal pelvis and kidney parenchyma | Loin pain, fever > 38°C, rigors, nausea/vomiting | Longer treatment (7-14 days); may require IV antibiotics and admission |
| Recurrent UTI | ≥2 UTIs in 6 months OR ≥3 UTIs in 12 months | Woman with 3 episodes in 8 months | Investigation for underlying cause; consider prophylaxis [8] |
| Catheter-Associated UTI (CAUTI) | Symptomatic UTI in patient with indwelling catheter or within 48h of removal | Fever in catheterised patient | Only treat if symptomatic; change catheter; extended treatment |
| Asymptomatic Bacteriuria (ASB) | ≥10^5 CFU/mL on culture WITHOUT symptoms | Elderly patient, catheterised patient | Do NOT treat except pregnancy or before urological procedure [7] |
Alternative Classifications:
- By severity: Mild (outpatient oral), moderate (oral or IV), severe (sepsis, requiring admission)
- By acquisition: Community-acquired vs healthcare-associated (nosocomial)
- By anatomy: Cystitis, urethritis, prostatitis, epididymo-orchitis, pyelonephritis
3. Aetiology and Pathophysiology
Causative Organisms
Uropathogenic bacteria typically originate from the bowel flora and colonise the periurethral area before ascending the urethra to cause infection.
| Organism | Frequency | Setting | Virulence Factors | Antibiotic Considerations |
|---|---|---|---|---|
| Escherichia coli | 75-95% | Community-acquired uncomplicated UTI [3] | Type 1 fimbriae (bladder adhesion), P fimbriae (kidney adhesion), haemolysin, aerobactin (iron acquisition) | Increasing resistance to trimethoprim (20-30%) and fluoroquinolones (15-20%); nitrofurantoin remains highly susceptible [12] |
| Staphylococcus saprophyticus | 5-10% | Young sexually active women (second most common) [16] | Surface proteins for uroepithelial adhesion | Usually susceptible to common agents; does NOT produce nitrites on dipstick |
| Klebsiella pneumoniae | 5-10% | Healthcare-associated, diabetics | Polysaccharide capsule, siderophores | Increasing ESBL production (10-30% in some regions) |
| Proteus mirabilis | 5% | Males, catheterised patients, stone formers | Urease (alkalinises urine, promotes struvite stones), swarming motility | Intrinsically resistant to nitrofurantoin and tigecycline |
| Enterococcus faecalis | 5-10% | CAUTI, healthcare-associated, elderly | Biofilm formation, aggregation substance | Intrinsically resistant to cephalosporins; VRE emerging problem |
| Pseudomonas aeruginosa | < 5% | CAUTI, complicated UTI, nosocomial | Biofilm formation, multiple efflux pumps | Intrinsically resistant to most oral agents; requires ciprofloxacin or aminoglycosides |
| Group B Streptococcus (GBS) | < 5% | Pregnant women, diabetics | Capsule | Penicillin-sensitive; important in pregnancy |
ESBL-Producing Organisms: Extended-spectrum beta-lactamase (ESBL)-producing Enterobacteriaceae (predominantly E. coli and Klebsiella) are increasingly prevalent, particularly in recurrent UTI, recent antibiotic exposure, healthcare contact, and travel to endemic areas (Indian subcontinent). ESBL organisms are resistant to penicillins, cephalosporins, and aztreonam; carbapenems (ertapenem, meropenem) are first-line for severe infections, though nitrofurantoin and fosfomycin often retain activity for cystitis. [17]
Pathophysiological Sequence
Step 1: Periurethral Colonisation
The initial event is colonisation of the periurethral area and vaginal introitus with uropathogenic bacteria from the rectal reservoir. Women with recurrent UTI demonstrate higher periurethral and vaginal colonisation with E. coli compared to controls. Factors promoting colonisation include:
- Vaginal pH changes (reduced lactobacilli)
- Spermicide use (disrupts normal flora)
- Oestrogen deficiency (postmenopausal vaginal atrophy)
- Sexual activity (mechanical transfer of bacteria)
Step 2: Urethral Ascension
Bacteria ascend the urethra to reach the bladder. The short female urethra (4 cm vs 20 cm in males) facilitates this process, explaining the dramatic sex difference in UTI incidence. Sexual intercourse causes mechanical massage of periurethral bacteria into the bladder ("honeymoon cystitis"). [14] In males, UTI almost always implies an underlying abnormality (obstruction, catheter, instrumentation) as the long urethra provides effective defence in health.
Step 3: Bladder Colonisation and Invasion
Upon reaching the bladder, uropathogenic bacteria must overcome multiple host defences:
- Urinary washout: Regular micturition physically removes bacteria (voiding 6-8 times daily with complete emptying)
- Bladder mucus layer: Glycosaminoglycans coat uroepithelium, providing physical barrier
- Antimicrobial peptides: Defensins and cathelicidin in urine inhibit bacterial growth
- Uroepithelial shedding: Infected superficial cells exfoliate, removing adherent bacteria
- Lactoferrin and secretory IgA: Antimicrobial proteins in urine
E. coli overcomes these defences using type 1 fimbriae, which bind to mannosylated uroplakin receptors on bladder epithelium. This triggers bacterial invasion into superficial umbrella cells, where E. coli can form intracellular bacterial communities (IBCs) protected from antibiotics and immune clearance. [18] Bacteria may also invade deeper into the bladder wall, forming quiescent intracellular reservoirs that seed recurrent infections.
Step 4: Inflammatory Response
Bacterial adhesion and invasion trigger toll-like receptor (TLR) signalling, particularly TLR4 (recognises LPS from gram-negative bacteria). This activates NF-κB, inducing production of pro-inflammatory cytokines (IL-6, IL-8, TNF-α) and chemokines that recruit neutrophils to the bladder. Neutrophil transmigration into urine causes pyuria. The inflammatory response generates classical UTI symptoms:
- Dysuria: Inflammatory mediators sensitise bladder nerve endings
- Frequency and urgency: Inflammatory oedema reduces functional bladder capacity; irritation of detrusor muscle causes uninhibited contractions
- Suprapubic pain: Bladder wall inflammation and distension
Step 5: Potential for Ascending Pyelonephritis
In 20-30% of untreated UTIs, bacteria ascend the ureters to reach the renal pelvis and parenchyma, causing pyelonephritis. [19] Risk factors include:
- Vesicoureteric reflux (retrograde flow during voiding)
- High bacterial load and virulence (P fimbriae mediate kidney adhesion)
- Delayed treatment
- Obstruction (preventing downward urinary flow)
- Pregnancy (progesterone-induced ureteric dilatation and hypomotility)
Pyelonephritis triggers a systemic inflammatory response with fever, rigors, and potential bacteraemia. Severe cases may progress to renal abscess, perinephric abscess, emphysematous pyelonephritis (especially in diabetics), or septic shock.
Host Susceptibility Factors
Genetic factors influence UTI susceptibility. Blood group antigens are expressed on uroepithelial cells; individuals with P1 antigen and non-secretors of ABH antigens demonstrate increased E. coli adhesion and recurrent UTI risk. [20] Polymorphisms in genes encoding TLR4, CXCR1 (IL-8 receptor), and other innate immune components affect UTI susceptibility and pyelonephritis risk. These genetic factors partly explain why some women experience frequent recurrent UTI despite normal anatomy and behaviour.
4. Clinical Presentation
Lower UTI (Cystitis)
Cystitis presents with local bladder symptoms WITHOUT systemic features (no fever, no loin pain). The classic triad is dysuria, frequency, and urgency.
| Symptom | Frequency | Characteristics | Clinical Notes |
|---|---|---|---|
| Dysuria | 80-90% | Burning or stinging pain during micturition, typically at start or throughout voiding | External dysuria (pain before urine passes through urethra) suggests vulvovaginitis or urethritis |
| Frequency | 75-85% | Passing small volumes frequently (> 8 times per 24 hours) | Daytime and nocturnal frequency; distinguish from polyuria (large volumes) |
| Urgency | 70-80% | Sudden compelling desire to void, difficulty deferring | May be accompanied by urge incontinence |
| Suprapubic pain | 50-60% | Dull ache or pressure in lower abdomen, worse with bladder filling | Tenderness on palpation of suprapubic region |
| Haematuria | 20-30% | Visible (macroscopic) or non-visible (microscopic) blood in urine | Alarming to patients but common in UTI; resolve with treatment |
| Cloudy/malodorous urine | 40-50% | Offensive smell, turbid appearance | Due to pyuria and bacteriuria; subjective |
| Incomplete emptying | 30-40% | Sensation of residual urine after voiding | May indicate detrusor dysfunction or obstruction if persistent |
Absence of systemic symptoms is key: No fever, no loin pain, no rigors, no nausea/vomiting. If present, consider upper UTI (pyelonephritis).
Upper UTI (Pyelonephritis)
Pyelonephritis manifests with systemic features in addition to urinary symptoms, reflecting renal parenchymal infection and inflammatory response.
| Feature | Frequency | Characteristics | Clinical Significance |
|---|---|---|---|
| Loin/flank pain | 80-90% | Unilateral or bilateral dull ache or sharp pain in costovertebral angle | Indicates renal involvement; distinguish from renal colic (colicky, radiating) |
| Fever | 80-90% | Temperature > 38°C, often > 39°C with rigors | Marker of systemic infection; persistent fever suggests complication (abscess) |
| Rigors | 60-70% | Shaking chills, teeth chattering | Indicates bacteraemia; associated with temperature spikes |
| Nausea and vomiting | 50-60% | Often prominent, may prevent oral intake | May require IV fluids and antiemetics |
| Lower UTI symptoms | 70-80% | Dysuria, frequency, urgency often precede systemic features by 24-48 hours | Progression from cystitis to pyelonephritis |
| Malaise and myalgia | 60-70% | Generalised feeling of unwellness, body aches | Non-specific but common |
Red Flag Features
[!CAUTION] RED FLAGS REQUIRING URGENT ASSESSMENT
- Fever > 38.5°C with rigors: Suggests pyelonephritis or bacteraemia
- Loin pain: Indicates upper UTI; risk of abscess or obstruction
- Sepsis criteria: Tachycardia, tachypnoea, hypotension, altered mental status (SIRS/qSOFA)
- Acute urinary retention: Complete inability to void; requires catheterisation
- Macroscopic haematuria in male or > 40 years: Investigate for malignancy after infection treated
- Pregnancy: All UTIs in pregnancy require culture and treatment (risk of preterm labour)
- Immunosuppression: Transplant recipients, biologics, chemotherapy (risk of fungal UTI, resistant organisms)
- Known structural abnormality: Single kidney, ileal conduit, neuropathic bladder (high complication risk)
- Recent urological procedure: Instrumentation within 7 days (iatrogenic introduction)
- Failed empirical therapy: Persistent symptoms after 48-72 hours of appropriate antibiotics (resistant organism or complication)
- Male patient with UTI symptoms: Complicated by definition; requires investigation
Atypical Presentations
Elderly Patients: Older adults may present with non-specific features rather than classic UTI symptoms. Delirium (acute confusion) is frequently attributed to UTI in the elderly, but asymptomatic bacteriuria is highly prevalent (20-50%) and is often an incidental finding rather than cause of confusion. [21] Genuine UTI in elderly may present with:
- New or worsening confusion (delirium)
- Falls
- Functional decline
- Urinary incontinence (new or worsened)
- Anorexia
- Fever (often absent even with infection)
Catheterised Patients: Distinguishing catheter-associated UTI (CAUTI) from asymptomatic bacteriuria is challenging. All long-term catheterised patients have bacteriuria (universal by 30 days). [10] Only treat if patient has symptoms attributable to UTI:
- Fever without other source
- New suprapubic or loin pain
- Acute delirium
- Rigors or haemodynamic instability
- Visible haematuria (if not explained by trauma)
Pyuria, malodorous urine, and cloudy urine are NOT indications for treatment in catheterised patients.
Diabetic Patients: Diabetes increases UTI risk and severity. Diabetic patients may develop:
- Emphysematous cystitis (gas-forming organisms; bladder wall gas on CT)
- Emphysematous pyelonephritis (life-threatening; gas in renal parenchyma; often requires nephrectomy)
- Renal abscess
- Papillary necrosis (sloughed renal papillae causing obstruction and haematuria)
Men: UTI is rare in healthy young men. Presence of UTI symptoms should prompt consideration of:
- Urethritis (discharge, STI exposure)
- Prostatitis (perineal pain, obstructive symptoms, tender prostate on DRE)
- Epididymo-orchitis (scrotal pain and swelling)
- Bladder outlet obstruction (BPH, stricture)
- Underlying malignancy (bladder, prostate)
5. Clinical Examination
Lower UTI (Uncomplicated Cystitis)
Examination findings are typically minimal or normal in uncomplicated cystitis.
General Inspection:
- Patient appears well, not systemically unwell
- Afebrile (temperature less than 37.5°C)
- Normal haemodynamic parameters
Abdominal Examination:
- Suprapubic tenderness on palpation (50% of cases)
- No loin tenderness
- No masses or organomegaly
- Bladder not palpable (unless retention)
Specific Examinations:
- External genitalia: Inspect for vulvovaginitis, discharge (alternative diagnosis)
- No costovertebral angle (renal angle) tenderness
Upper UTI (Pyelonephritis)
General Inspection:
- Patient appears unwell, flushed
- Fever (temperature > 38°C, often 39-40°C)
- Tachycardia (heart rate > 100 bpm)
- Signs of dehydration (reduced skin turgor, dry mucous membranes) if vomiting
Vital Signs:
- Temperature: > 38°C (may spike to 40°C)
- Heart rate: Tachycardia proportionate to fever
- Blood pressure: Usually normal; hypotension suggests sepsis
- Respiratory rate: May be elevated if septic
Abdominal Examination:
- Renal angle tenderness: Tenderness or pain on percussion over costovertebral angle (Murphy's punch sign); may be unilateral or bilateral
- Loin tenderness on palpation
- Suprapubic tenderness (if concurrent cystitis)
- Assess for peritonism (rare; suggests abscess or perforation)
Systemic Examination:
- Hydration status
- Evidence of sepsis (capillary refill time, peripheral perfusion, mental status)
Special Examination Scenarios
Male UTI:
- Digital rectal examination (DRE): Assess prostate size, consistency, and tenderness
- "Benign prostatic hyperplasia: Enlarged, smooth, non-tender"
- "Prostatitis: Exquisitely tender, boggy, warm; avoid vigorous massage (risk of bacteraemia)"
- "Prostate cancer: Irregular, hard, nodular"
- Genitalia: Exclude epididymo-orchitis, urethritis, phimosis
- Assess for urinary retention: Palpable bladder, dullness to percussion suprapubically
Pregnant Women:
- Standard examination as above
- Assess for contractions (preterm labour)
- Check fetal heart rate if available
- Examine for signs of preterm rupture of membranes
Catheterised Patients:
- Inspect catheter site for trauma, bleeding
- Assess catheter patency (debris, blockage)
- Examine urine in bag (colour, clarity, visible blood)
- Suprapubic or loin tenderness if genuine CAUTI
6. Differential Diagnosis
Urinary symptoms are non-specific and occur in multiple conditions. Accurate differential diagnosis prevents inappropriate antibiotic use.
| Differential Diagnosis | Key Distinguishing Features | Diagnostic Approach |
|---|---|---|
| Urethritis (STI) | Urethral discharge, dysuria without frequency, sexual history, gradual onset | Urethral swab for chlamydia/gonorrhoea; first-pass urine NAAT |
| Vulvovaginitis (Candida, BV) | External dysuria, vaginal discharge, pruritus, no frequency | Vaginal examination, high vaginal swab |
| Pelvic Inflammatory Disease | Pelvic pain, dyspareunia, cervical motion tenderness, adnexal tenderness | Bimanual examination, pregnancy test, swabs |
| Interstitial Cystitis (Painful Bladder Syndrome) | Chronic pelvic pain, frequency, urgency WITHOUT infection on culture, pain relieved by voiding | Diagnosis of exclusion; cystoscopy shows glomerulations; Hunner's ulcers |
| Bladder Cancer | Painless visible haematuria (typically), older patient (> 50 years), smoking history, no dysuria | Urine cytology, flexible cystoscopy, CT urogram |
| Renal Calculi (Colic) | Severe colicky loin-to-groin pain, haematuria, nausea/vomiting, pain comes in waves | Non-contrast CT KUB (gold standard); ultrasound |
| Acute Pyelonephritis vs Renal Abscess | Persistent fever despite 48-72h antibiotics, persistent bacteraemia | Contrast-enhanced CT abdomen/pelvis |
| Prostatitis | Perineal pain, obstructive voiding symptoms, tender prostate on DRE, male patient | DRE, urine culture, consider prostate massage cultures (after acute phase) |
| Acute Urinary Retention | Sudden inability to void, severe suprapubic pain, palpable bladder | Bladder scan (> 300mL); catheterisation |
| Atrophic Vaginitis (Postmenopausal) | Vaginal dryness, dyspareunia, postmenopausal, no infection on culture | Vaginal examination (pale, thin epithelium); trial of topical oestrogen |
| Acute Interstitial Nephritis | Drug exposure (NSAIDs, PPIs, antibiotics), rash, eosinophilia, AKI | Urine eosinophils, renal biopsy if uncertain |
| Appendicitis | Right-sided abdominal pain (McBurney's point), anorexia, migration of pain, fever | Inflammatory markers, CT abdomen/pelvis |
| Diverticulitis | Left iliac fossa pain, altered bowel habit, fever, older patient | CT abdomen/pelvis with contrast |
Key Clinical Clues:
- Urethral discharge = NOT UTI → Consider STI (chlamydia, gonorrhoea, trichomoniasis)
- External dysuria (pain before urine passes through urethra) = NOT UTI → Consider vulvovaginitis, dermatitis
- Haematuria without dysuria/frequency = NOT UTI → Consider cancer, stones, glomerulonephritis
- Symptoms > 7 days despite negative culture = NOT UTI → Consider interstitial cystitis, overactive bladder
- Recurrent "UTI" with negative cultures = NOT UTI → Consider interstitial cystitis, urethral syndrome
7. Investigations
Bedside Investigations
Urine Dipstick
Rapid point-of-care test providing supportive evidence within 2 minutes. Interpretation requires understanding of test characteristics.
| Dipstick Finding | Interpretation | Sensitivity | Specificity | Clinical Notes |
|---|---|---|---|---|
| Nitrites | Presence of nitrite-reducing bacteria (E. coli, Klebsiella, Proteus) | 40-60% | 85-98% | HIGH specificity: Positive nitrites strongly support UTI. False negatives: S. saprophyticus, Enterococcus, Pseudomonas do NOT produce nitrites; inadequate bladder dwell time (less than 4 hours); dietary factors [22] |
| Leucocyte esterase | Enzyme released by lysed neutrophils; indicates pyuria | 75-90% | 60-80% | HIGH sensitivity: Negative LE makes UTI less likely. False positives: Contamination, sterile pyuria (TB, interstitial nephritis, stones) |
| Both positive | — | 80-90% | 85-90% | Combined: Positive predictive value 80-90% for UTI |
| Both negative | — | — | 90-95% | Combined: Negative predictive value 90-95%; UTI unlikely |
| Blood | Haematuria (intact RBCs) or haemoglobin/myoglobin | Variable | Variable | Common in UTI (20-30%); persistent haematuria after treatment requires investigation for cancer/stones |
| Protein | Proteinuria | Variable | Variable | Mild proteinuria common in UTI; heavy proteinuria suggests glomerular disease |
Dipstick Limitations:
- Cannot differentiate UTI from asymptomatic bacteriuria
- Poor positive predictive value in low-prevalence populations (e.g., asymptomatic screening)
- Should NOT be performed in asymptomatic patients
- Does NOT replace clinical diagnosis in uncomplicated cystitis
Vital Signs and Observations:
- Temperature (fever > 38°C suggests pyelonephritis)
- Blood pressure and heart rate (sepsis screening)
- Oxygen saturations (if septic)
- Capillary blood glucose (especially if diabetic; assess control)
Laboratory Investigations
**Urine Culture "Urine Culture Interpretation and Reporting Standards
Urine culture remains the gold standard for microbiological confirmation of UTI and is essential in complicated cases, treatment failures, and pregnancy. Understanding culture interpretation, including quantitative bacteriology, mixed growth patterns, and reporting timelines, is crucial for antimicrobial stewardship.
Collection Technique:
- Midstream urine (MSU): Gold standard for non-catheterised patients
- Clean perineum (avoid antiseptics which may inhibit bacterial growth)
- Discard first portion of urine (washes urethral commensals)
- Collect midstream portion in sterile container
- Process within 4 hours or refrigerate (prevents overgrowth of contaminants)
- Catheter specimen: From catheter sampling port (NOT from bag)
- Suprapubic aspirate: Gold standard if MSU unobtainable (rarely used)
Interpretation:
- Significant bacteriuria: ≥10^5 CFU/mL of single uropathogen (classic definition) [5]
- Lower thresholds may be significant: ≥10^3 CFU/mL in symptomatic women; ≥10^2 CFU/mL in catheter specimen
- Mixed growth (≥3 organisms): Usually contamination; repeat sample
- Sterile pyuria (WBCs but no growth): Consider TB, partially treated UTI, urethritis, interstitial nephritis, stones
Quantitative Bacteriology Thresholds:
The classic threshold of ≥10^5 CFU/mL (100,000 colony-forming units per millilitre) was established by Kass in 1956 and remains the standard definition of significant bacteriuria. [5] However, this threshold was derived from asymptomatic bacteriuria studies in pregnancy and may lack sensitivity in symptomatic acute cystitis. Modern evidence demonstrates that lower thresholds are clinically significant in specific contexts:
| Clinical Context | Significant Threshold | Rationale |
|---|---|---|
| Symptomatic women (cystitis) | ≥10^3 CFU/mL | Acute cystitis may present with lower bacterial counts due to recent voiding, high urinary flow, or early infection [39] |
| Symptomatic men | ≥10^3 CFU/mL | Lower threshold reflects difficulty in obtaining uncontaminated samples; longer urethra reduces contamination risk |
| Catheterised patients (CAUTI) | ≥10^2 CFU/mL | Catheter provides direct access; biofilm bacteria release into urine at lower counts |
| Suprapubic aspirate | ANY growth | Suprapubic aspiration bypasses urethral contamination; any bacterial growth is pathological |
| Asymptomatic bacteriuria | ≥10^5 CFU/mL on two consecutive samples | Reduces false positives from transient colonisation or single contaminated samples |
Mixed Growth and Contamination:
Mixed growth, defined as three or more bacterial species, occurs in 10-20% of urine samples and almost always represents contamination from perineal or vaginal flora. Repeat sampling with careful midstream technique typically yields a single uropathogen. However, genuinely polymicrobial UTI (two organisms) occurs in 5-10% of complicated UTI, particularly in catheterised patients, patients with urinary tract abnormalities, or following urological instrumentation. Enterococcus faecalis and Escherichia coli are the most common polymicrobial combination. [40]
Sterile Pyuria:
Pyuria (white blood cells in urine, detected by leucocyte esterase on dipstick or greater than 10 WBCs per high-power field on microscopy) without bacterial growth on culture is termed sterile pyuria. Differential diagnoses include:
| Cause | Key Features | Diagnostic Approach |
|---|---|---|
| Partially treated UTI | Recent antibiotics (within 48 hours) | Clinical history; repeat culture after antibiotic course |
| Urogenital tuberculosis | Chronic symptoms, night sweats, weight loss, haematuria | Three early-morning urine samples for acid-fast bacilli (AFB) and TB culture/PCR |
| Urethritis (STI) | Urethral discharge, dysuria without frequency, sexual risk | Urethral swab or first-pass urine NAAT for chlamydia/gonorrhoea |
| Interstitial nephritis | Drug exposure (NSAIDs, PPIs, antibiotics), AKI, rash, eosinophilia | Urine eosinophils, renal biopsy if persistent |
| Renal calculi | Colicky loin pain, haematuria | Non-contrast CT KUB |
| Appendicitis/diverticulitis | Adjacent inflammation causing bladder irritation | CT abdomen/pelvis |
| Interstitial cystitis | Chronic pelvic pain, frequency, urgency; negative cultures repeatedly | Cystoscopy (glomerulations, Hunner's ulcers) |
Sterile pyuria in the context of systemic symptoms (fever, weight loss, night sweats) should prompt investigation for genitourinary tuberculosis, particularly in patients from endemic regions or with HIV. Three early-morning urine samples (first void of the day, when bacterial load is highest) should be sent for Ziehl-Neelsen staining, mycobacterial culture, and TB PCR (GeneXpert). Sensitivity of urine microscopy for TB is low (40-60%), hence the requirement for three samples. [41]
Antibiotic Susceptibility Reporting:
Urine culture results typically include antibiotic susceptibility testing, guiding targeted therapy and antimicrobial stewardship. Reporting follows standardised categories:
- Sensitive (S): Organism is inhibited by standard doses of the antibiotic; clinical success expected
- Intermediate (I): Organism may be inhibited by increased doses or if antibiotic concentrates in urine (applies to some agents like nitrofurantoin)
- Resistant (R): Organism is not inhibited by achievable drug concentrations; treatment failure likely
Modern laboratories use EUCAST (European Committee on Antimicrobial Susceptibility Testing) or CLSI (Clinical and Laboratory Standards Institute) breakpoints, which differ slightly and may lead to discrepant reporting between centres. Nitrofurantoin, for example, achieves very high urinary concentrations (200-400 mg/L), meaning organisms reported as "intermediate" by blood-based breakpoints may still be clinically susceptible for cystitis (though not for pyelonephritis, where tissue penetration matters). [42]
Resistance Patterns and ESBL Detection:
Extended-spectrum beta-lactamase (ESBL)-producing Enterobacteriaceae are increasingly prevalent in UTI, particularly in patients with recurrent infections, recent antibiotic exposure (especially cephalosporins or fluoroquinolones), healthcare contact, or travel to high-prevalence regions (Indian subcontinent, Middle East, Southeast Asia). ESBL organisms are resistant to penicillins, cephalosporins (including third-generation agents like ceftriaxone), and aztreonam, but typically remain susceptible to carbapenems (ertapenem, meropenem) and often to nitrofurantoin and fosfomycin for cystitis. [17]
Laboratory detection of ESBL involves phenotypic testing: if an organism shows reduced susceptibility to third-generation cephalosporins, confirmatory testing with clavulanic acid (a beta-lactamase inhibitor) is performed. ESBL detection should trigger:
- Treatment adjustment: Carbapenems for severe infection (pyelonephritis, urosepsis); nitrofurantoin or fosfomycin for uncomplicated cystitis if susceptible
- Infection control: Contact precautions in hospital settings (ESBL colonisation may persist for months)
- Antimicrobial stewardship review: Avoid unnecessary cephalosporins and fluoroquinolones in this patient going forward
Culture Reporting Timelines:
Understanding culture timelines is essential for clinical decision-making:
- Preliminary results (18-24 hours): Gram stain and preliminary identification (Gram-negative rods, Gram-positive cocci); allows early adjustment from empirical therapy
- Final identification and sensitivities (48-72 hours): Species confirmation and full antibiotic susceptibility panel
- Slow-growing organisms (5-7 days): Mycobacterium tuberculosis, fungi (Candida species require 2-3 days; moulds may require 5-7 days)
For patients empirically treated, clinical response (resolution of symptoms, defervescence) should be assessed at 48-72 hours. If the patient is improving, empirical therapy can be continued even if subsequent culture shows "intermediate" susceptibility, as clinical response supersedes in vitro testing in uncomplicated cases. Conversely, if no improvement occurs despite in vitro susceptibility, consider non-adherence, inadequate dosing, renal impairment affecting drug concentrations, or complication (abscess, obstruction). [43]
Blood Tests (if systemically unwell or pyelonephritis)
| Test | Expected Findings | Clinical Use |
|---|---|---|
| Full Blood Count | Neutrophilia (raised WCC, typically 12-20 × 10^9/L); left shift | Marker of bacterial infection; baseline if septic |
| C-Reactive Protein (CRP) | Elevated (typically 50-200 mg/L in pyelonephritis) | Marker of inflammation; trend with treatment response |
| Urea and Electrolytes (U&E) | May show AKI (raised creatinine) in pyelonephritis/sepsis; dehydration (raised urea) | Assess renal function; guide drug dosing (e.g., nitrofurantoin contraindicated if eGFR less than 45) |
| Blood Cultures | Positive in 20-30% of pyelonephritis cases | Identify bacteraemia; guide antibiotic choice if resistant organism |
| Lactate | Elevated if septic (> 2 mmol/L) | Marker of tissue hypoperfusion and sepsis severity |
| Procalcitonin | Elevated in bacterial infection | Increasingly used to differentiate bacterial from non-bacterial inflammation; guides antibiotic duration |
Pregnancy Test: All women of childbearing age with UTI symptoms should have pregnancy excluded (management differs significantly).
Imaging
Ultrasound Renal Tract
Indications:
- Male UTI (assess for obstruction, post-void residual, prostatic size)
- Recurrent UTI in women (identify stones, hydronephrosis, structural abnormalities)
- Pyelonephritis not responding to 48-72 hours of antibiotics (exclude abscess, obstruction)
- Suspected obstruction (hydronephrosis)
- Acute kidney injury in context of UTI
Findings:
- Hydronephrosis (ureteric/bladder outlet obstruction)
- Renal or bladder calculi
- Post-void residual volume (greater than 100 mL abnormal; greater than 300 mL significant retention)
- Renal abscess (hypoechoic collection)
- Prostatic enlargement
Technique and Optimization: Post-void residual (PVR) measurement requires bladder scanning immediately after the patient voids completely. Timing is critical: scanning more than 10 minutes post-void reduces accuracy due to ongoing urine production. A PVR of 50-100 mL is borderline; greater than 100 mL is abnormal and suggests incomplete emptying (detrusor weakness, obstruction, or neurogenic bladder). In men over 50 years with recurrent UTI, measuring PVR combined with prostatic volume assessment (transabdominal or transrectal) guides management decisions regarding alpha-blockers or surgical intervention for benign prostatic hyperplasia. [33]
Renal ultrasound has 85-90% sensitivity for detecting hydronephrosis but only 20-40% sensitivity for detecting renal calculi less than 5 mm. [34] If stone disease is suspected (colicky pain, haematuria), non-contrast CT KUB is the investigation of choice.
CT Abdomen/Pelvis with Contrast
Indications:
- Complicated pyelonephritis (persistent fever, failure to respond to antibiotics, severe sepsis)
- Suspected renal or perinephric abscess
- Emphysematous pyelonephritis (gas in renal parenchyma; diabetics)
- Suspected obstructing stone with infection (pyonephrosis; urological emergency)
- Recurrent UTI with suspected anatomical abnormality not seen on ultrasound
Findings:
- Pyelonephritis: Striated nephrogram, perinephric fat stranding, delayed excretion
- Renal abscess: Rim-enhancing collection within kidney
- Perinephric abscess: Collection in perinephric space
- Emphysematous pyelonephritis: Gas within renal parenchyma (surgical emergency)
- Stones, hydronephrosis, anatomical variants
Diagnostic Criteria for Complicated Pyelonephritis on CT: The presence of gas in the renal parenchyma (emphysematous pyelonephritis) or collecting system (emphysematous pyelitis) is diagnostic of severe infection, almost exclusively occurring in diabetic patients with poorly controlled glucose. CT classification systems exist: Class 1 (gas in collecting system only), Class 2 (gas in renal parenchyma without abscess), Class 3A (extension to perinephric space), Class 3B (extension to pararenal space), Class 4 (bilateral involvement or solitary kidney). [35] Class 1 and 2 may respond to medical management with broad-spectrum IV antibiotics and tight glycaemic control; Class 3 and 4 often require nephrectomy due to mortality rates of 40-60% with medical management alone.
Renal abscess appears as a low-attenuation collection with peripheral rim enhancement on contrast CT. Abscesses larger than 3-5 cm typically require percutaneous or surgical drainage in addition to prolonged antibiotics (4-6 weeks total), whereas smaller abscesses may respond to antibiotics alone. Percutaneous drainage under CT or ultrasound guidance has replaced open surgical drainage as first-line intervention, with success rates of 80-90%. [36]
Non-Contrast CT KUB (Kidney-Ureter-Bladder)
Indication: Suspected renal calculi (more sensitive than ultrasound)
Non-contrast CT KUB is the gold standard for diagnosing renal and ureteric calculi, with sensitivity and specificity both exceeding 95%. [34] This is superior to ultrasound (which misses small stones and ureteric stones) and intravenous urography (which is now rarely used). The investigation is rapid (less than 5 minutes), does not require contrast, and provides definitive stone size, location, and density (Hounsfield units), which predict stone composition and likelihood of spontaneous passage. Stones less than 5 mm have 90% chance of spontaneous passage; stones greater than 7 mm have less than 10% chance and typically require intervention (shock wave lithotripsy, ureteroscopy, or percutaneous nephrolithotomy).
Cystoscopy
Indications:
- Recurrent UTI in women (exclude bladder pathology, interstitial cystitis)
- Persistent haematuria after UTI treated (exclude bladder cancer)
- Suspected foreign body (e.g., encrusted catheter, suture)
Flexible cystoscopy is performed under local anaesthetic (intraurethral lidocaine gel) and allows direct visualisation of the urethra, bladder mucosa, and ureteric orifices. It is indicated in women with recurrent UTI if initial investigations (ultrasound, urine culture) are unrevealing, particularly if over 40 years or if haematuria is present. Cystoscopic findings in interstitial cystitis include glomerulations (petechial haemorrhages on bladder distension) and Hunner's ulcers (in 10% of cases). Bladder cancer typically presents as papillary or solid lesions; biopsy is performed at the time of cystoscopy. [37]
DMSA Scan (Renal Scintigraphy)
Indication: Assess for renal scarring after recurrent pyelonephritis (predominantly paediatric use)
Technetium-99m dimercaptosuccinic acid (DMSA) renal scintigraphy is the gold standard for detecting renal cortical scarring resulting from pyelonephritis or vesicoureteric reflux. It is primarily used in children following febrile UTI to identify those at risk of progressive renal damage, hypertension, and chronic kidney disease. In adults, DMSA scanning may be considered in young adults with recurrent pyelonephritis to assess cumulative renal damage and guide decisions regarding prophylactic antibiotics or surgical correction of reflux. The technique involves intravenous injection of DMSA, which is taken up by functioning renal tubular cells; areas of scarring or acute inflammation appear as photopenic defects (cold spots). Acute pyelonephritis and chronic scarring can be differentiated by repeating the scan 6 months later: acute inflammatory changes resolve, whereas scars persist. [38]
8. Management
Management strategy is determined by UTI classification (uncomplicated vs complicated), severity (cystitis vs pyelonephritis), and patient factors (pregnancy, immunosuppression).
General Principles
-
Antimicrobial Stewardship:
- Use narrowest-spectrum antibiotic with local susceptibility data
- Shortest effective duration (avoid excessive courses)
- Reserve fluoroquinolones for pyelonephritis and resistant infections
- Send urine culture before antibiotics if complicated UTI (adjust based on sensitivities)
-
Symptomatic Management:
- Adequate analgesia (paracetamol, NSAIDs if appropriate)
- Encourage high fluid intake (2-3 L/day) to promote urinary washout
- Avoid bladder irritants (caffeine, alcohol, spicy foods)
-
Safety Netting:
- Advise return if no improvement within 48 hours
- Red flag symptoms (fever, loin pain, vomiting, inability to tolerate oral fluids)
Uncomplicated Cystitis in Women
First-Line Antibiotics (3-day course): [6,9]
| Antibiotic | Dose | Advantages | Disadvantages | Resistance Rates |
|---|---|---|---|---|
| Nitrofurantoin | 100mg BD (modified-release) or 50mg QDS (immediate-release) | Low resistance (typically less than 5%); minimal impact on gut flora; safe in pregnancy (except at term) | Contraindicated if eGFR less than 45; nausea common; pulmonary fibrosis with long-term use; ineffective for pyelonephritis | 2-5% |
| Trimethoprim | 200mg BD | Low cost; well-tolerated; once daily option (300mg OD) | Rising resistance (20-30% in many regions); contraindicated first trimester pregnancy (folate antagonist) | 20-30% |
| Fosfomycin | 3g single dose | Single-dose convenience; low resistance; broad spectrum | Expensive; less evidence than nitrofurantoin/trimethoprim; may be inferior to 5-day courses [23] | 5-10% |
| Pivmecillinam | 400mg TDS | Low resistance; effective; available in some European countries | Not available in all countries (UK, Scandinavia); GI side effects | 5-8% |
Second-Line Options (if first-line contraindicated or resistant):
- Cefalexin 500mg BD for 3 days
- Amoxicillin/clavulanate 625mg TDS for 3 days (if culture confirms susceptibility)
Avoid as First-Line:
- Fluoroquinolones (ciprofloxacin, levofloxacin): Reserve for pyelonephritis and complicated UTI; drive resistance
- Amoxicillin: 50% E. coli resistance due to beta-lactamase production
Duration: 3 days is optimal. Meta-analyses demonstrate non-inferiority of 3-day vs 5-7 day courses for clinical cure (OR 0.98, 95% CI 0.71-1.35) with lower adverse effects and antimicrobial resistance. [6]
Evidence: The landmark Huttner et al. JAMA trial (2018) compared 5-day nitrofurantoin to single-dose fosfomycin in 513 women with uncomplicated cystitis. Clinical resolution at day 28 was superior with 5-day nitrofurantoin (70% vs 58%, difference 12%, 95% CI 3-21%, p=0.004), establishing multidose nitrofurantoin as preferred over single-dose fosfomycin. [23]
Male UTI
ALL UTIs in men are complicated by definition and require investigation for underlying cause.
Antibiotic Therapy (7-day course):
- First-line: Trimethoprim 200mg BD for 7 days OR Nitrofurantoin 100mg BD for 7 days
- Second-line: Ciprofloxacin 500mg BD for 7 days (if resistant or suspected prostatitis)
- Prostatitis suspected: Requires longer duration (4-6 weeks); use fluoroquinolone or trimethoprim (prostatic penetration)
Mandatory Investigations:
- Urine culture (identify organism and sensitivities)
- Renal tract ultrasound (post-void residual, prostatic size, hydronephrosis)
- Consider PSA if > 50 years (after infection cleared; acute infection elevates PSA)
- Consider sexual health screening if less than 35 years (urethritis/STI more likely than UTI)
Specific Causes to Address:
- Benign prostatic hyperplasia (BPH): Alpha-blocker (tamsulosin), 5-alpha-reductase inhibitor (finasteride), or surgery if severe
- Urethral stricture: Urological referral for dilation or urethroplasty
- Bladder outlet obstruction: Assess with uroflowmetry and post-void residual
Pregnancy
UTI in pregnancy (symptomatic or asymptomatic bacteriuria) requires treatment due to risk of pyelonephritis (20-30% if untreated), preterm labour, and low birth weight. [7,24]
Antibiotic Choices (7-day course):
| Antibiotic | Trimester | Safety Profile |
|---|---|---|
| Nitrofurantoin | First and second trimester | SAFE; avoid at term (> 36 weeks) due to theoretical neonatal haemolysis risk |
| Cefalexin | All trimesters | SAFE; first-line if nitrofurantoin contraindicated |
| Amoxicillin | All trimesters | SAFE if culture confirms susceptibility (high resistance rates) |
| Trimethoprim | Second and third trimester only | AVOID first trimester (folate antagonist; theoretical neural tube defect risk); safe in second/third trimester |
| Fluoroquinolones | ALL trimesters | CONTRAINDICATED (cartilage toxicity) |
| Tetracyclines | ALL trimesters | CONTRAINDICATED (teeth staining, bone growth inhibition) |
Management Protocol:
- All pregnant women: Urine culture at first antenatal booking (screen for asymptomatic bacteriuria)
- If positive: Treat with 7-day course and repeat culture 1 week post-treatment (test of cure)
- Recurrent UTI in pregnancy: Monthly urine cultures for remainder of pregnancy
- Pyelonephritis in pregnancy: Hospital admission, IV antibiotics (cefuroxime or gentamicin), obstetric involvement
Pyelonephritis
Upper UTI requires longer treatment duration and broader-spectrum antibiotics with renal parenchymal penetration.
Severity Assessment:
| Severity | Criteria | Management Setting |
|---|---|---|
| Mild | Low-grade fever, tolerate oral fluids, haemodynamically stable | Outpatient with oral antibiotics; safety-net advice |
| Moderate | High fever, vomiting but able to take oral antibiotics | Consider admission for 24h observation; oral or IV antibiotics |
| Severe | Sepsis criteria, unable to tolerate oral, hypotension, severe vomiting/dehydration | Hospital admission; IV antibiotics; fluid resuscitation |
Antibiotic Regimens:
Oral (Outpatient - Mild Pyelonephritis):
- Ciprofloxacin 500mg BD for 7 days (first-line if local resistance less than 10%)
- Co-amoxiclav 625mg TDS for 10-14 days (if fluoroquinolone resistance or contraindication)
- Cefalexin 500mg TDS for 10-14 days (alternative; less evidence)
Intravenous (Inpatient - Moderate to Severe Pyelonephritis):
- Gentamicin 5-7 mg/kg IV once daily PLUS amoxicillin 1g TDS (broad spectrum; covers Enterococcus)
- Ceftriaxone 1-2g IV once daily (if gentamicin contraindicated; does NOT cover Enterococcus)
- Co-amoxiclav 1.2g IV TDS (alternative; good renal penetration)
If ESBL suspected (risk factors: recurrent UTI, recent antibiotics, healthcare exposure, travel to endemic areas):
- Ertapenem 1g IV once daily OR Meropenem 1g IV TDS
Duration: 7 days for mild pyelonephritis, 10-14 days for severe or complicated cases. Recent meta-analysis suggests short-course (7-day) therapy may be non-inferior to longer courses in selected patients. [25]
Admission Criteria:
- Unable to tolerate oral antibiotics (vomiting)
- Sepsis or haemodynamic instability
- Pregnancy
- Immunosuppression
- Single kidney or known renal impairment
- Suspicion of obstruction or abscess
- Failed outpatient management
Step-Down Therapy: Switch from IV to oral antibiotics when:
- Afebrile for 24 hours
- Tolerating oral fluids and medications
- Improving inflammatory markers
Continue oral antibiotics to complete 10-14 days total.
Catheter-Associated UTI (CAUTI)
Diagnostic Criteria (ALL required):
- Indwelling catheter OR catheter removed within 48 hours
- Clinical symptoms attributable to UTI (fever without other source, new suprapubic/loin pain, acute delirium, rigors)
- Urine culture with ≥10^3 CFU/mL of uropathogen
Do NOT treat if: Asymptomatic bacteriuria (universal in long-term catheters), pyuria alone, malodorous urine alone
Management:
- Change catheter before starting antibiotics (biofilm on old catheter harbours bacteria)
- Antibiotic therapy (7 days):
- Empirical: As per local hospital guidelines (typically broader spectrum due to Pseudomonas and Enterococcus risk)
- First-line: Ciprofloxacin 500mg BD OR co-amoxiclav 625mg TDS
- Adjust based on culture sensitivities
- Remove catheter if possible (catheter-free status prevents recurrence)
- If catheter required: Consider intermittent self-catheterisation if feasible (lower infection rate than indwelling)
Prevention:
- Avoid unnecessary catheterisation (urinary catheter bundles)
- Use smallest bore catheter for shortest duration
- Maintain closed drainage system
- Consider alternatives (condom catheter, intermittent catheterisation)
Recurrent UTI
Defined as ≥2 UTIs in 6 months or ≥3 UTIs in 12 months. Affects 20-30% of women after initial UTI. [8]
Investigation:
- Urine culture during symptomatic episodes (identify organism pattern)
- Renal tract ultrasound (exclude stones, obstruction, post-void residual > 100mL)
- Consider cystoscopy if haematuria, age > 50, or risk factors for bladder cancer
- Assess for diabetes, immunosuppression
- Post-void residual measurement (incomplete emptying)
Non-Antibiotic Preventative Strategies:
| Strategy | Evidence | Mechanism | Recommendations |
|---|---|---|---|
| Behavioural Measures | Moderate | Reduce bacterial load and colonisation time | Void after intercourse; adequate hydration (6-8 glasses/day); avoid delayed voiding; wipe front-to-back |
| Cranberry Products | Moderate | Proanthocyanidins inhibit E. coli adhesion to uroepithelium | Meta-analysis shows 26% reduction in UTI recurrence (RR 0.74, 95% CI 0.55-0.97) with cranberry vs placebo [26]; requires high-dose standardised products |
| D-Mannose | Moderate | Mannose binds type 1 fimbriae, preventing E. coli bladder adhesion | RCT showed D-mannose 2g daily non-inferior to nitrofurantoin 50mg daily for prophylaxis [27]; well-tolerated alternative |
| Vaginal Oestrogen (Postmenopausal) | Strong | Restores vaginal pH, promotes lactobacilli, improves uroepithelial integrity | Topical oestrogen (cream, pessary, ring) reduces UTI recurrence by 50% in postmenopausal women [28]; no systemic absorption |
| Probiotics (Lactobacillus) | Weak | Vaginal flora restoration | Limited evidence; some trials show benefit with specific strains |
| Increased Fluid Intake | Weak | Urinary dilution and washout | Advise 1.5-2L daily; some evidence of benefit |
| Methenamine Hippurate | Weak | Converted to formaldehyde in acidic urine (bactericidal) | Some benefit in post-menopausal women; requires acidic urine (vitamin C co-administration) |
Antibiotic Prophylaxis (Specialist Initiation):
Reserved for women with recurrent UTI despite behavioural measures, when non-antibiotic strategies have failed.
Regimens:
- Continuous low-dose prophylaxis: Nitrofurantoin 50mg nocte OR Trimethoprim 100mg nocte for 6-12 months
- Post-coital prophylaxis: Single dose of nitrofurantoin 50mg or trimethoprim 100mg after intercourse (if UTI temporally related to sexual activity)
Evidence: Cochrane review demonstrates antibiotic prophylaxis reduces UTI recurrence (RR 0.21, 95% CI 0.13-0.34) but with adverse effects and antimicrobial resistance concerns. [29]
Risks: Antimicrobial resistance, candidiasis, GI side effects, C. difficile infection
Follow-up: Review efficacy and adverse effects at 3 and 6 months; consider stopping after 6-12 months and reassess recurrence pattern.
Asymptomatic Bacteriuria (ASB)
Presence of ≥10^5 CFU/mL on urine culture WITHOUT urinary symptoms.
Prevalence:
- Healthy premenopausal women: 1-5%
- Postmenopausal women: 10-15%
- Elderly women (> 70 years): 20-50%
- Catheterised patients: 100% by 30 days
DO NOT TREAT EXCEPT:
- Pregnancy (20-30% progress to pyelonephritis; risk of preterm labour) [7]
- Before urological procedures involving mucosal trauma (TURP, cystoscopy with biopsy, stone manipulation)
DO NOT TREAT in:
- Elderly patients (no benefit; increases resistance and C. difficile risk)
- Diabetic patients (no benefit demonstrated)
- Catheterised patients (re-bacteriuria occurs within days)
- Renal transplant recipients (beyond first 3 months post-transplant)
- Patients with spinal cord injury and neuropathic bladder
Evidence: Multiple RCTs and meta-analyses demonstrate NO benefit of treating ASB in non-pregnant populations, with harms including adverse effects, antimicrobial resistance, and C. difficile infection. [7,21]
9. Complications
| Complication | Incidence | Risk Factors | Clinical Features | Management |
|---|---|---|---|---|
| Acute Pyelonephritis | 20-30% of untreated cystitis [19] | Delayed treatment, vesicoureteric reflux, pregnancy, diabetes | Loin pain, fever, rigors, nausea/vomiting | 7-14 days antibiotics; IV if severe; admission if septic |
| Urosepsis | 10-20% of pyelonephritis | Elderly, immunosuppressed, obstruction, delayed treatment | SIRS criteria, hypotension, organ dysfunction | Sepsis 6 bundle; IV broad-spectrum antibiotics; fluid resuscitation; ICU if shocked |
| Renal Abscess | 1-2% of pyelonephritis | Diabetes, S. aureus bacteraemia, obstruction | Persistent fever despite antibiotics, loin mass | CT diagnosis; antibiotics (4-6 weeks); percutaneous/surgical drainage if > 3cm |
| Perinephric Abscess | 0.5% of pyelonephritis | Extension of renal abscess, haematogenous spread | Persistent fever, loin pain, sepsis | CT diagnosis; percutaneous or surgical drainage; prolonged antibiotics |
| Emphysematous Pyelonephritis | Rare; almost exclusively diabetics | Poorly controlled diabetes, obstruction, immunosuppression | Severe sepsis, gas in renal parenchyma on CT | Urological emergency; IV antibiotics; often requires nephrectomy; mortality 20-40% |
| Papillary Necrosis | Rare; diabetics, sickle cell, analgesic nephropathy | Diabetes, NSAIDs, sickle cell disease | Loin pain, haematuria, passage of sloughed tissue, AKI | IV antibiotics; ureteric stenting if obstruction; analgesia |
| Chronic Pyelonephritis | 5-10% with recurrent UTIs | Recurrent pyelonephritis, vesicoureteric reflux, obstruction | Scarred, shrunken kidneys; hypertension; CKD | Prevent recurrent infections; treat hypertension; monitor renal function |
| Bacteraemia | 20-30% of pyelonephritis | Pyelonephritis, instrumentation, CAUTI | Positive blood cultures; may be asymptomatic or cause sepsis | IV antibiotics guided by blood culture sensitivities |
| Acute Kidney Injury | 10-15% of severe pyelonephritis | Sepsis, obstruction, volume depletion, nephrotoxic drugs | Raised creatinine, oliguria | Fluid resuscitation; treat infection; avoid nephrotoxins; may require RRT if severe |
| Preterm Labour (Pregnancy) | 20-30% if pyelonephritis in pregnancy | UTI/pyelonephritis in pregnancy | Contractions, cervical change before 37 weeks | IV antibiotics; tocolytics if appropriate; obstetric management |
| Chronic UTI Symptoms | 10-20% | Inadequate treatment, recurrent infections | Persistent dysuria, frequency despite negative cultures | Exclude interstitial cystitis, chronic pelvic pain syndrome; may need cystoscopy |
Preventing Complications:
- Early recognition and treatment of UTI
- Longer treatment duration for pyelonephritis (avoid inadequate 5-day courses)
- Investigation of male UTI and recurrent UTI to identify and correct underlying causes
- Aggressive management of UTI in high-risk groups (pregnancy, diabetes, immunosuppression, single kidney)
- Appropriate use of IV antibiotics in severe cases
10. Prognosis and Outcomes
Uncomplicated Cystitis
Natural History:
- Spontaneous resolution in 25-40% of cases without antibiotics (though symptoms prolonged)
- With appropriate antibiotics: Clinical resolution in 85-95% within 48-72 hours
- Microbiological cure (negative culture): 80-90% at 1 week post-treatment
Recurrence:
- 20-30% experience recurrent UTI within 6 months of initial episode [8]
- 40-50% experience at least one further UTI within 12 months
- Most recurrences are reinfections (new organisms) rather than relapse (same organism)
Long-Term:
- No long-term renal sequelae from uncomplicated cystitis
- Quality of life significantly impaired during symptomatic episodes
- Recurrent UTI associated with psychological distress, work absence, sexual dysfunction
Pyelonephritis
With Treatment:
- 90-95% clinical resolution with appropriate antibiotics
- Fever typically resolves within 48-72 hours
- Complete recovery in most cases without long-term sequelae
Without Treatment:
- High risk of progression to sepsis (20-30%)
- Mortality in untreated pyelonephritis: 10-20%
- Risk of renal abscess formation: 5-10%
Recurrence:
- 10-15% recurrence rate within 1 year
- Higher if underlying structural abnormality (VUR, stones, obstruction)
Long-Term:
- Recurrent pyelonephritis → chronic pyelonephritis → renal scarring → CKD in 5-10%
- Hypertension may develop secondary to renal scarring
Special Populations
Pregnancy:
- Untreated asymptomatic bacteriuria: 20-30% progress to pyelonephritis
- Pyelonephritis in pregnancy: 20-30% risk of preterm labour; 10% risk of low birth weight
- With treatment: Outcomes similar to general obstetric population
Elderly:
- Higher mortality from urosepsis (10-20% in elderly vs 2-5% in younger adults)
- Increased risk of complications (bacteraemia, AKI, delirium)
- Higher recurrence rates due to comorbidities
Immunosuppressed:
- Higher risk of severe infection, bacteraemia, and resistant organisms
- Mortality from urosepsis: 15-30%
- Require prolonged antibiotic courses and close monitoring
Prognostic Factors
Favorable Prognosis:
- Young, healthy, non-pregnant woman
- Uncomplicated cystitis
- Prompt treatment with appropriate antibiotics
- No structural abnormalities
Poor Prognosis:
- Delayed treatment (> 7 days from symptom onset)
- Immunosuppression (transplant, chemotherapy, HIV, biologics)
- Structural abnormalities (obstruction, stones, VUR)
- Sepsis at presentation
- Extremes of age (neonates, elderly > 75 years)
- Resistant organisms (ESBL, carbapenemase-producing Enterobacteriaceae)
11. Prevention and Screening
Primary Prevention
Behavioural Modifications:
- Adequate hydration (1.5-2L daily; 6-8 glasses)
- Regular, complete bladder emptying (avoid delayed voiding)
- Post-coital voiding within 30 minutes
- Front-to-back wiping after defecation
- Avoid vaginal douching and spermicide use
- Cotton underwear (though evidence weak)
- Avoid prolonged wetness (wet swimwear, tight synthetic clothing)
Postmenopausal Women:
- Topical vaginal oestrogen (cream, pessary, or ring) for women with recurrent UTI [28]
- Restores vaginal pH, promotes lactobacilli, improves uroepithelial integrity
- Reduces recurrence by approximately 50%
Men:
- Treat benign prostatic hyperplasia (alpha-blockers, 5-ARIs, surgery)
- Optimise bladder emptying (void sitting down to reduce post-void residual)
- Avoid urethral instrumentation when possible
Screening
Screening for Asymptomatic Bacteriuria:
Recommended:
- Pregnant women: Screen at first antenatal booking (12-16 weeks) with urine culture; treat if positive
- Before urological procedures with mucosal trauma (TURP, cystoscopy with biopsy)
NOT Recommended (no benefit demonstrated):
- Elderly patients (community-dwelling or institutionalised)
- Diabetic patients
- Catheterised patients
- Patients with spinal cord injury
- Renal transplant recipients (beyond 3 months post-transplant)
- Premenopausal non-pregnant women
Catheter Care (Prevention of CAUTI)
Evidence-Based Strategies: [10]
- Avoid unnecessary catheterisation: Use only when indicated (urinary retention, perioperative monitoring, palliative care); daily review of necessity
- Aseptic insertion technique: Sterile equipment, antiseptic cleaning, adequate lubrication
- Closed drainage system: Maintain closed system; minimise disconnections
- Position drainage bag below bladder level: Prevent retrograde flow
- Empty drainage bag regularly: Before two-thirds full; avoid bag touching floor
- Remove catheters as soon as possible: Each day increases infection risk by 3-7%
- Consider alternatives: Intermittent self-catheterisation (lower infection rate); condom catheters (males); prompted voiding
Ineffective Strategies (not recommended):
- Antimicrobial/silver-coated catheters (limited benefit)
- Routine catheter changes (no benefit if functioning)
- Antibiotic prophylaxis during catheterisation (drives resistance)
- Bladder washouts/irrigation (no benefit; may increase infection)
12. Key Guidelines and Evidence
International Guidelines
-
NICE NG109 (2018): Urinary tract infection (lower): antimicrobial prescribing
- Recommends nitrofurantoin or trimethoprim as first-line for uncomplicated cystitis
- 3-day course for women; 7-day course for men
- Avoid fluoroquinolones as first-line
-
NICE NG111 (2018): Pyelonephritis (acute): antimicrobial prescribing
- Ciprofloxacin or co-amoxiclav for 7-14 days depending on severity
- Admission criteria specified (sepsis, inability to tolerate oral, pregnancy)
-
EAU Guidelines on Urological Infections (2024) [30]
- Comprehensive European guideline covering uncomplicated and complicated UTI
- Emphasises antimicrobial stewardship and avoiding fluoroquinolones for simple cystitis
- Defines complicated UTI and provides antibiotic recommendations based on local resistance
-
IDSA/ESCMID Guidelines (2011): International Clinical Practice Guidelines for Uncomplicated Cystitis and Pyelonephritis [31]
- Landmark guideline defining uncomplicated vs complicated UTI
- Evidence-based antibiotic recommendations and duration
- Addresses asymptomatic bacteriuria (treat only in pregnancy and before urological procedures)
-
AUA/CUA/SUFU Guideline (2019): Recurrent Uncomplicated Urinary Tract Infections in Women [32]
- Evidence-based strategies for recurrent UTI prevention
- Non-antibiotic interventions (behavioural, cranberry, D-mannose, vaginal oestrogen)
- Antibiotic prophylaxis as last resort
-
Cochrane Review (2019): Antibiotics for asymptomatic bacteriuria in pregnancy [7]
- Antibiotic treatment reduces risk of pyelonephritis (RR 0.23, 95% CI 0.13-0.41)
- Reduces risk of preterm birth and low birth weight
- Supports universal screening and treatment in pregnancy
Landmark Evidence
Short-Course Antibiotic Therapy:
- Milo et al. BMJ (2005): Meta-analysis of 32 trials (9,605 women) comparing 3-day vs 5-7 day antibiotic courses for uncomplicated cystitis. No difference in clinical cure (OR 0.98, 95% CI 0.71-1.35); lower adverse effects with 3-day courses. [6]
Nitrofurantoin vs Fosfomycin:
- Huttner et al. JAMA (2018): RCT of 513 women; 5-day nitrofurantoin superior to single-dose fosfomycin for clinical resolution (70% vs 58%, p=0.004). [23]
Cranberry for Prevention:
- Fu et al. J Nutr (2017): Meta-analysis of 7 RCTs (1,498 participants) showed cranberry products reduce UTI recurrence (RR 0.74, 95% CI 0.55-0.97), particularly in women with recurrent UTI. [26]
D-Mannose for Prevention:
- Kranjčec et al. World J Urol (2014): RCT of 308 women; D-mannose 2g daily non-inferior to nitrofurantoin 50mg daily for preventing recurrent UTI (15% vs 20% recurrence, p=NS). [27]
Vaginal Oestrogen in Postmenopausal Women:
- Perrotta et al. Menopause (2008): Meta-analysis demonstrated topical vaginal oestrogen reduces UTI recurrence by 50% in postmenopausal women (RR 0.50, 95% CI 0.34-0.74). [28]
Asymptomatic Bacteriuria - Harm of Treatment:
- Nicolle et al. Clin Infect Dis (2005): Landmark review establishing that treatment of ASB in non-pregnant populations provides no benefit and increases antimicrobial resistance. [7]
Antibiotic Resistance Trends:
- Klingeberg et al. Dtsch Arztebl Int (2018): German study showing E. coli resistance to trimethoprim 27%, fluoroquinolones 15%, but nitrofurantoin only 2%; emphasises importance of nitrofurantoin as first-line. [12]
CAUTI Prevention:
- Flores-Mireles et al. Top Spinal Cord Inj Rehabil (2019): Comprehensive review of CAUTI pathophysiology and prevention strategies; emphasises catheter removal as most effective prevention. [10]
13. Patient Communication and Explanation
What is a Urinary Tract Infection?
A urinary tract infection (UTI) is a bacterial infection affecting your bladder (cystitis) or kidneys (pyelonephritis). It is extremely common, especially in women. Bacteria, usually from the bowel (most commonly E. coli), travel up the urethra (the tube from which you pass urine) and multiply in the bladder, causing inflammation and symptoms.
What are the Symptoms?
Bladder Infection (Cystitis):
- Burning or stinging pain when passing urine (dysuria)
- Needing to pass urine frequently, including at night
- Sudden urges to urinate that are difficult to hold
- Lower abdominal discomfort or pressure
- Urine that looks cloudy, smells unpleasant, or contains blood
Kidney Infection (Pyelonephritis):
- All the above symptoms, plus:
- Fever, shivering, and feeling generally unwell
- Pain in your back or side (loin pain)
- Nausea and vomiting
How is it Diagnosed?
Most bladder infections in women are diagnosed based on your symptoms alone. Your doctor may ask you to provide a urine sample for a quick dipstick test, which can support the diagnosis. If you have a kidney infection, are pregnant, are male, or have recurrent infections, a urine sample will be sent to the laboratory to identify the specific bacteria and guide treatment.
Treatment
Bladder Infection:
- Short course of antibiotics (usually 3 days)
- Common antibiotics: nitrofurantoin or trimethoprim
- Drink plenty of fluids (water, dilute juice)
- Paracetamol or ibuprofen for pain and discomfort
Kidney Infection:
- Longer course of antibiotics (7-14 days)
- May require hospital admission if severe
When to Seek Urgent Help
Contact your doctor or seek urgent medical attention if you develop:
- High fever (temperature above 38°C) with shivering
- Severe back or side pain
- Persistent vomiting preventing you from keeping fluids down
- No improvement after 2 days of antibiotics
- Blood in your urine (if not already present)
- Inability to pass urine
Prevention
To Reduce Your Risk of UTI:
- Drink plenty of fluids (6-8 glasses of water daily)
- Wipe from front to back after using the toilet
- Urinate soon after sexual intercourse (within 30 minutes)
- Don't delay going to the toilet when you feel the need
- Avoid perfumed soaps or bubble baths in the genital area
If You Have Recurrent Infections:
- Your doctor may recommend additional measures such as vaginal oestrogen cream (if you are postmenopausal), cranberry products, or D-mannose supplements
- In some cases, low-dose antibiotic prophylaxis may be considered
Common Questions
Q: Can I have sex with a UTI? A: Sexual intercourse may be uncomfortable and could potentially worsen symptoms. It's generally advisable to wait until symptoms have resolved.
Q: Do I need to finish the full course of antibiotics even if I feel better? A: Yes. Completing the full prescribed course (usually 3 days for simple bladder infections) ensures the infection is fully cleared and reduces the risk of antibiotic resistance.
Q: Can cranberry juice prevent UTIs? A: Some evidence suggests cranberry products may reduce the risk of recurrent UTIs in some women, but the effect is modest. Cranberry products are not a substitute for antibiotics to treat an active infection.
Q: Why do I keep getting UTIs? A: Some women are more prone to UTIs due to anatomical factors, hormonal changes (especially after menopause), sexual activity, or genetic susceptibility. Your doctor can assess for underlying causes and discuss preventative strategies.
Q: Should I get tested if I have no symptoms but my urine looks cloudy? A: No. If you have no symptoms, testing is not usually helpful (except in pregnancy). Cloudy urine can be normal, and bacteria in urine without symptoms (asymptomatic bacteriuria) does not usually require treatment.
14. Examination Focus (Postgraduate Level)
MRCP PACES / Clinical Stations
Scenario: 25-year-old woman presents with 2-day history of dysuria, frequency, and suprapubic discomfort. No fever or loin pain.
Expected Approach:
-
History:
- Urinary symptoms (dysuria, frequency, urgency, haematuria)
- Systemic symptoms (fever, loin pain, rigors) → exclude pyelonephritis
- Previous UTIs (recurrence pattern)
- Sexual history (new partner, post-coital timing)
- Vaginal symptoms (discharge, pruritus) → exclude vaginitis/STI
- Pregnancy status (LMP, contraception)
- Medical history (diabetes, immunosuppression)
- Drug history (recent antibiotics, spermicide use)
-
Examination:
- General: Appears well, afebrile, haemodynamically stable
- Abdomen: Suprapubic tenderness; no loin tenderness; no masses
-
Investigations:
- Urine dipstick: Positive nitrites and leucocyte esterase → supports UTI
- Urine culture: NOT routinely required for uncomplicated cystitis (clinical diagnosis sufficient)
- Pregnancy test: If applicable
-
Diagnosis:
- Uncomplicated acute cystitis
-
Management:
- First-line: Nitrofurantoin 100mg BD for 3 days OR trimethoprim 200mg BD for 3 days
- Safety-net advice: Return if no improvement in 48 hours, or if fever/loin pain develops
- Lifestyle advice: Adequate hydration, post-coital voiding
- Avoid: Urine culture (not indicated); fluoroquinolones (reserve for pyelonephritis)
Viva Voce Questions and Model Answers
Q1: How would you distinguish between cystitis and pyelonephritis?
A: "Cystitis presents with local bladder symptoms—dysuria, frequency, urgency, and suprapubic pain—without systemic features. Examination reveals suprapubic tenderness but no fever or loin tenderness. Pyelonephritis, conversely, is an upper urinary tract infection presenting with systemic features: fever above 38°C, rigors, loin pain, and often nausea and vomiting. Examination demonstrates renal angle tenderness and fever. Cystitis requires 3-day treatment, whereas pyelonephritis requires 7-14 days with broader-spectrum antibiotics and may necessitate hospital admission if severe."
Q2: What defines complicated UTI, and why is this distinction important?
A: "Complicated UTI is defined as infection occurring in a patient with factors that increase risk of treatment failure or severe infection. This includes males, pregnant women, patients with structural urinary tract abnormalities, urinary catheters, immunosuppression, diabetes, renal transplantation, or recent instrumentation. The distinction is crucial because complicated UTI requires: (1) mandatory urine culture to guide therapy, (2) longer treatment duration (minimum 7 days), (3) investigation for underlying anatomical or functional abnormalities, and (4) broader-spectrum empirical antibiotics in some cases. In contrast, uncomplicated cystitis in women can be diagnosed and treated empirically without culture."
Q3: A 72-year-old man in a nursing home has a catheter and cloudy urine. Urine culture grows 10^6 E. coli. He is afebrile and has no new symptoms. Do you treat?
A: "No. This is asymptomatic bacteriuria, which is universal in long-term catheterised patients. Treatment is only indicated if the patient has clinical symptoms attributable to UTI—fever without alternative source, new suprapubic or loin pain, acute delirium, or haemodynamic instability. Pyuria and bacteriuria alone, even with high colony counts, are not indications for antibiotics in catheterised patients. Unnecessary treatment drives antimicrobial resistance, causes adverse effects including C. difficile infection, and provides no clinical benefit. The appropriate management here is clinical observation without antibiotics."
Q4: What is the evidence regarding treatment of asymptomatic bacteriuria in pregnancy?
A: "Asymptomatic bacteriuria occurs in 2-10% of pregnancies and, if untreated, progresses to pyelonephritis in 20-30% of cases. A Cochrane systematic review of 14 trials demonstrated that antibiotic treatment significantly reduces pyelonephritis risk (RR 0.23, 95% CI 0.13-0.41), preterm birth, and low birth weight. [7] Consequently, all pregnant women should be screened with urine culture at first antenatal booking, and positive cultures should be treated with a 7-day course of pregnancy-safe antibiotics—nitrofurantoin (avoid at term), cefalexin, or amoxicillin if susceptible. Test-of-cure culture is performed one week post-treatment, and women with recurrence require monthly surveillance cultures for the remainder of pregnancy."
Q5: Why is nitrofurantoin preferred over trimethoprim for uncomplicated cystitis?
A: "Nitrofurantoin is increasingly favoured due to antimicrobial stewardship considerations. E. coli resistance to trimethoprim has risen substantially, now 20-30% in many regions, whereas nitrofurantoin resistance remains low at 2-5%. [12] Nitrofurantoin has multiple mechanisms of action, reducing resistance development, and has minimal impact on gut flora, thus preserving the microbiome. Additionally, it is safe in pregnancy except at term. However, nitrofurantoin is contraindicated if eGFR is less than 45 mL/min/1.73m² due to inadequate urinary concentration and efficacy, and it does not achieve therapeutic renal parenchymal levels, rendering it ineffective for pyelonephritis. The landmark JAMA trial by Huttner et al. (2018) also demonstrated 5-day nitrofurantoin superior to single-dose fosfomycin. [23]"
Q6: What non-antibiotic strategies have evidence for preventing recurrent UTI?
A: "Several non-antibiotic strategies have robust evidence. First, vaginal oestrogen in postmenopausal women reduces recurrence by approximately 50% (RR 0.50, 95% CI 0.34-0.74) by restoring vaginal pH and promoting lactobacilli. [28] Second, cranberry products containing proanthocyanidins reduce recurrence by 26% (RR 0.74, 95% CI 0.55-0.97), though high-dose standardised products are required. [26] Third, D-mannose 2g daily has been shown in RCT to be non-inferior to nitrofurantoin 50mg daily for prophylaxis with fewer side effects. [27] Behavioural measures—post-coital voiding, adequate hydration, and avoiding delayed voiding—are recommended, though evidence is less robust. Antibiotic prophylaxis with low-dose nitrofurantoin or trimethoprim is reserved for women who fail non-antibiotic strategies, due to resistance concerns."
Q7: A 28-year-old woman presents with dysuria. Urine culture grows greater than 10^5 S. saprophyticus. Why might the urine dipstick have been negative for nitrites?
A: "Staphylococcus saprophyticus does not produce nitrite reductase, the enzyme that converts urinary nitrate to nitrite. Consequently, urine dipstick nitrites will be negative despite genuine UTI. S. saprophyticus is the second most common cause of uncomplicated cystitis in young, sexually active women (5-10% of cases) after E. coli. It typically occurs in late summer and autumn. Leucocyte esterase is usually positive, reflecting pyuria. The diagnosis is confirmed by urine culture showing greater than 10^5 CFU/mL of S. saprophyticus. First-line treatment is the same as for E. coli UTI—nitrofurantoin or trimethoprim for 3 days."
Common Viva Mistakes
❌ Treating asymptomatic bacteriuria in elderly or catheterised patients (no benefit; drives resistance)
❌ Using 7-day antibiotic course for uncomplicated cystitis in women (3 days is sufficient and evidence-based)
❌ Failing to investigate male UTI (ALL male UTIs are complicated and require ultrasound and investigation)
❌ Prescribing nitrofurantoin in renal impairment (eGFR less than 45) (ineffective; inadequate urinary concentration)
❌ Using trimethoprim in first trimester pregnancy (folate antagonist; theoretical teratogenicity)
❌ Prescribing fluoroquinolones as first-line for uncomplicated cystitis (reserve for pyelonephritis; antimicrobial stewardship)
❌ Diagnosing UTI in elderly based solely on confusion (ASB prevalence 20-50%; require other supportive features)
❌ Failing to send urine culture in complicated UTI (mandatory to guide therapy and detect resistance)
❌ Not recognising ESBL risk factors (recurrent UTI, recent antibiotics, travel to endemic areas, healthcare contact)
❌ Inadequate duration for pyelonephritis (requires 7-14 days, not 3-5 days)
High-Yield Examination Facts
- E. coli causes 75-95% of community-acquired UTIs [3]
- 3-day antibiotic course for uncomplicated cystitis (non-inferior to longer courses) [6]
- Nitrofurantoin contraindicated if eGFR less than 45 mL/min/1.73m²
- All male UTIs are complicated by definition (require investigation)
- Asymptomatic bacteriuria: treat ONLY in pregnancy and before urological procedures [7]
- Nitrites: high specificity (85-98%), low sensitivity (40-60%) [22]
- Cranberry reduces UTI recurrence by 26% (RR 0.74) [26]
- Vaginal oestrogen reduces recurrence by 50% in postmenopausal women [28]
- Pyelonephritis in pregnancy: 20-30% risk of preterm labour [24]
- CAUTI: only treat if symptomatic (not bacteriuria/pyuria alone) [10]
- Recurrent UTI definition: ≥2 in 6 months OR ≥3 in 12 months [8]
- Significant bacteriuria: ≥10^5 CFU/mL on MSU [5]
- S. saprophyticus: second most common in young women; does NOT produce nitrites [16]
- ESBL organisms: treat with carbapenem (ertapenem, meropenem) if severe [17]
- Nitrofurantoin resistance: 2-5%; trimethoprim resistance: 20-30% [12]
15. References
-
Flores-Mireles AL, Walker JN, Caparon M, Hultgren SJ. Urinary tract infections: epidemiology, mechanisms of infection and treatment options. Nat Rev Microbiol. 2015;13(5):269-284. doi:10.1038/nrmicro3432
-
Foxman B. Epidemiology of urinary tract infections: incidence, morbidity, and economic costs. Am J Med. 2002;113 Suppl 1A:5S-13S. doi:10.1016/s0002-9343(02)01054-9
-
Klingeberg A, Noll I, Willems S, et al. Antibiotic-Resistant E. coli in Uncomplicated Community-Acquired Urinary Tract Infection. Dtsch Arzteblatt Int. 2018;115(29-30):494-500. doi:10.3238/arztebl.2018.0494
-
Bonkat G, Bartoletti R, Bruyère F, et al. EAU Guidelines on Urological Infections. European Association of Urology. 2024. https://uroweb.org/guidelines/urological-infections
-
Kass EH. Asymptomatic infections of the urinary tract. Trans Assoc Am Physicians. 1956;69:56-64.
-
Milo G, Katchman EA, Paul M, Christiaens T, Baerheim A, Leibovici L. Duration of antibacterial treatment for uncomplicated urinary tract infection in women. Cochrane Database Syst Rev. 2005;(2):CD004682. doi:10.1002/14651858.CD004682.pub2
-
Smaill FM, Vazquez JC. Antibiotics for asymptomatic bacteriuria in pregnancy. Cochrane Database Syst Rev. 2019;11(11):CD000490. doi:10.1002/14651858.CD000490.pub4
-
Anger J, Lee U, Ackerman AL, et al. Recurrent Uncomplicated Urinary Tract Infections in Women: AUA/CUA/SUFU Guideline. J Urol. 2019;202(2):282-289. doi:10.1097/JU.0000000000000296
-
Mahdizade Ari M, Davoodian P, Rajaei B, Ghasemi A, Dadvand H, Amid P. Nitrofurantoin: properties and potential in treatment of urinary tract infection: a narrative review. Front Cell Infect Microbiol. 2023;13:1148603. doi:10.3389/fcimb.2023.1148603
-
Flores-Mireles AL, Hreha TN, Hunstad DA. Pathophysiology, Treatment, and Prevention of Catheter-Associated Urinary Tract Infection. Top Spinal Cord Inj Rehabil. 2019;25(3):228-240. doi:10.1310/sci2503-228
-
Public Health England. English surveillance programme for antimicrobial utilisation and resistance (ESPAUR) Report 2019 to 2020. GOV.UK. Published 2020.
-
Klingeberg A, Noll I, Willems S, et al. Antibiotic-Resistant E. coli in Uncomplicated Community-Acquired Urinary Tract Infection. Dtsch Arzteblatt Int. 2018;115(29-30):494-500. doi:10.3238/arztebl.2018.0494
-
Geerlings SE. Clinical Presentations and Epidemiology of Urinary Tract Infections. Microbiol Spectr. 2016;4(5). doi:10.1128/microbiolspec.UTI-0002-2012
-
Foxman B, Barlow R, D'Arcy H, Gillespie B, Sobel JD. Urinary tract infection: self-reported incidence and associated costs. Ann Epidemiol. 2000;10(8):509-515. doi:10.1016/s1047-2797(00)00072-7
-
Choi JB, Hong SH, Kim JH, Bae JH. Complicated urinary tract infection in patients with benign prostatic hyperplasia. J Infect Chemother. 2021;27(9):1350-1356. doi:10.1016/j.jiac.2021.05.007
-
Raz R, Colodner R, Kunin CM. Who are you--Staphylococcus saprophyticus? Clin Infect Dis. 2005;40(6):896-898. doi:10.1086/428353
-
Peirano G, Pitout JDD. Extended-Spectrum β-Lactamase-Producing Enterobacteriaceae: Update on Molecular Epidemiology and Treatment Options. Drugs. 2019;79(14):1529-1541. doi:10.1007/s40265-019-01180-3
-
Mulvey MA, Schilling JD, Hultgren SJ. Establishment of a persistent Escherichia coli reservoir during the acute phase of a bladder infection. Infect Immun. 2001;69(7):4572-4579. doi:10.1128/IAI.69.7.4572-4579.2001
-
Gupta K, Hooton TM, Naber KG, et al. International clinical practice guidelines for the treatment of acute uncomplicated cystitis and pyelonephritis in women: A 2010 update by the Infectious Diseases Society of America and the European Society for Microbiology and Infectious Diseases. Clin Infect Dis. 2011;52(5):e103-e120. doi:10.1093/cid/ciq257
-
Sheinfeld J, Schaeffer AJ, Cordon-Cardo C, Rogatko A, Fair WR. Association of the Lewis blood-group phenotype with recurrent urinary tract infections in women. N Engl J Med. 1989;320(12):773-777. doi:10.1056/NEJM198903233201205
-
Nicolle LE, Gupta K, Bradley SF, et al. Clinical Practice Guideline for the Management of Asymptomatic Bacteriuria: 2019 Update by the Infectious Diseases Society of America. Clin Infect Dis. 2019;68(10):e83-e110. doi:10.1093/cid/ciy1121
-
Deville WL, Yzermans JC, van Duijn NP, Bezemer PD, van der Windt DA, Bouter LM. The urine dipstick test useful to rule out infections. A meta-analysis of the accuracy. BMC Urol. 2004;4:4. doi:10.1186/1471-2490-4-4
-
Huttner A, Kowalczyk A, Turjeman A, et al. Effect of 5-Day Nitrofurantoin vs Single-Dose Fosfomycin on Clinical Resolution of Uncomplicated Lower Urinary Tract Infection in Women: A Randomized Clinical Trial. JAMA. 2018;319(17):1781-1789. doi:10.1001/jama.2018.3627
-
Ansaldi Y, Gonser S. Urinary tract infections in pregnancy. Clin Microbiol Infect. 2023;29(10):1249-1253. doi:10.1016/j.cmi.2022.08.016
-
Zahavi I, Eliakim-Raz N, Yahav D. Short vs. long antibiotic treatment for pyelonephritis and complicated urinary tract infections: a living systematic review and meta-analysis of randomized controlled trials. Clin Microbiol Infect. 2025;31(8):1054-1063. doi:10.1016/j.cmi.2025.02.015
-
Fu Z, Liska D, Talan D, Chung M. Cranberry Reduces the Risk of Urinary Tract Infection Recurrence in Otherwise Healthy Women: A Systematic Review and Meta-Analysis. J Nutr. 2017;147(12):2282-2288. doi:10.3945/jn.117.254961
-
Kranjčec B, Papeš D, Altarac S. D-mannose powder for prophylaxis of recurrent urinary tract infections in women: a randomized clinical trial. World J Urol. 2014;32(1):79-84. doi:10.1007/s00345-013-1091-6
-
Perrotta C, Aznar M, Mejia R, Albert X, Ng CW. Oestrogens for preventing recurrent urinary tract infection in postmenopausal women. Cochrane Database Syst Rev. 2008;(2):CD005131. doi:10.1002/14651858.CD005131.pub2
-
Albert X, Huertas I, Pereiró II, Sanfélix J, Gosalbes V, Perrota C. Antibiotics for preventing recurrent urinary tract infection in non-pregnant women. Cochrane Database Syst Rev. 2004;(3):CD001209. doi:10.1002/14651858.CD001209.pub2
-
Kranz J, Schmidt S, Wagenlehner F, Schneidewind L. European Association of Urology Guidelines on Urological Infections: Summary of the 2024 Guidelines. Eur Urol. 2024;86(1):27-41. doi:10.1016/j.eururo.2024.03.035
-
Gupta K, Hooton TM, Naber KG, et al. International clinical practice guidelines for the treatment of acute uncomplicated cystitis and pyelonephritis in women: A 2010 update by the Infectious Diseases Society of America and the European Society for Microbiology and Infectious Diseases. Clin Infect Dis. 2011;52(5):e103-e120. doi:10.1093/cid/ciq257
-
Schmiemann G, Bleidorn J, Haefeli WE, Knopf H. The Diagnosis, Treatment, and Prevention of Recurrent Urinary Tract Infection. Dtsch Arzteblatt Int. 2024;121(11):369-376. doi:10.3238/arztebl.m2024.0058
-
Ahmed H, Davies F, Francis N, et al. Long-term antibiotics for prevention of recurrent urinary tract infection in older adults: systematic review and meta-analysis of randomised trials. BMJ Open. 2017;7(5):e015233. doi:10.1136/bmjopen-2016-015233
-
Smith RC, Rosenfield AT, Choe KA, et al. Acute flank pain: comparison of non-contrast-enhanced CT and intravenous urography. Radiology. 1995;194(3):789-794. doi:10.1148/radiology.194.3.7862980
-
Huang JJ, Tseng CC. Emphysematous pyelonephritis: clinicoradiological classification, management, and outcome. Arch Intern Med. 2000;160(6):797-805. doi:10.1001/archinte.160.6.797
-
Coelho RF, Schneider-Monteiro ED, Mesquita JL, Mazzucchi E, Marmo Lucon A, Srougi M. Renal and perinephric abscesses: analysis of 65 consecutive cases. World J Surg. 2007;31(2):431-436. doi:10.1007/s00268-006-0452-x
-
Hanno PM, Erickson D, Moldwin R, Faraday MM. Diagnosis and treatment of interstitial cystitis/bladder pain syndrome: AUA guideline amendment. J Urol. 2015;193(5):1545-1553. doi:10.1016/j.juro.2015.01.086
-
Piepsz A, Blaufox MD, Gordon I, et al. Consensus on renal cortical scintigraphy in children with urinary tract infection. Semin Nucl Med. 1999;29(2):160-174. doi:10.1016/s0001-2998(99)80005-6
-
Stamm WE, Counts GW, Running KR, Fihn S, Turck M, Holmes KK. Diagnosis of coliform infection in acutely dysuric women. N Engl J Med. 1982;307(8):463-468. doi:10.1056/NEJM198208193070802
-
Talan DA, Stamm WE, Hooton TM, et al. Comparison of ciprofloxacin (7 days) and trimethoprim-sulfamethoxazole (14 days) for acute uncomplicated pyelonephritis in women: a randomized trial. JAMA. 2000;283(12):1583-1590. doi:10.1001/jama.283.12.1583
-
Moussa OM, Eraky I, El-Far MA, et al. Rapid diagnosis of genitourinary tuberculosis by polymerase chain reaction and non-radioactive DNA hybridization. J Urol. 2000;164(2):584-588. doi:10.1016/s0022-5347(05)67417-6
-
Andrews JM. Determination of minimum inhibitory concentrations. J Antimicrob Chemother. 2001;48 Suppl 1:5-16. doi:10.1093/jac/48.suppl_1.5
-
Koningstein FN, Verbon A, Stobberingh E, et al. Urine culture-guided treatment shortens hospital stay and increases treatment success for hospitalized patients with urinary tract infections: a randomized controlled trial. Clin Infect Dis. 2023;76(3):e948-e956. doi:10.1093/cid/ciac635
Frequently asked questions
Quick clarifications for common clinical and exam-facing questions.
When should I seek emergency care for urinary tract infection in adults?
Seek immediate emergency care if you experience any of the following warning signs: Signs of pyelonephritis (loin pain, fever less than 38CC), Signs of sepsis (SIRS criteria), Acute urinary retention, Male UTI (complicated by definition), Pregnancy (risk of preterm labour), Catheter-associated infection with systemic features, Recurrent UTI requiring investigation, Failed empirical antibiotic therapy, Immunocompromised patient, Known structural urological abnormality.
Learning map
Use these linked topics to study the concept in sequence and compare related presentations.
Prerequisites
Start here if you need the foundation before this topic.
- Renal Anatomy and Physiology
- Antibiotic Principles
Differentials
Competing diagnoses and look-alikes to compare.
- Acute Interstitial Nephritis
- Urethritis and Sexually Transmitted Infections
- Bladder Cancer
Consequences
Complications and downstream problems to keep in mind.
- Urosepsis and Septic Shock
- Chronic Kidney Disease