Acute Pancreatitis
Acute Pancreatitis (AP) is an acute inflammatory process of the pancreas with variable involvement of other regional tis... MRCP exam preparation.
What matters first
Acute Pancreatitis (AP) is an acute inflammatory process of the pancreas with variable involvement of other regional tis... MRCP exam preparation.
Persistent SIRS less than 48h (High mortality risk)
6 Jan 2026
Generated educational material; verify before clinical use.
Visible references section
See the concept before reading it
Study the key anatomy, imaging, and decision pathways as full teaching plates.
Clinical board
A visual summary of the highest-yield teaching signals on this page.
Urgent signals
Safety-critical features pulled from the topic metadata.
- Persistent SIRS less than 48h (High mortality risk)
- Rising BUN/Urea despite fluid resuscitation
- Grey Turner's or Cullen's sign (Haemorrhagic)
- New-onset confusion (Encephalopathy)
Exam focus
Current exam surfaces linked to this topic.
- MRCP
Linked comparisons
Differentials and adjacent topics worth opening next.
- Peptic Ulcer Perforation
- Aortic Dissection
Content status and exam context
This page is AI-generated educational content. It may contain errors or omissions and is not a substitute for current guidelines, local protocols, senior clinical judgement, or professional medical advice.
MedVellum does not claim an individual clinician reviewer, board certification, or professional credential for this page unless a future version names a real, verifiable reviewer.
Topic family
This concept exists in multiple MedVellum libraries. Use the primary page for the broadest reference view and the others for exam-specific framing.
Acute Pancreatitis (AP) is an acute inflammatory process of the pancreas with variable involvement of other regional tis... MRCP exam preparation.
1. Diagnostic criteria: 2 of 3 (pain, lipase greater than 3× ULN, imaging)... CICM Second Part exam preparation.
Clinical explanation and evidence
Acute Pancreatitis (Adult)
1. Overview
Acute Pancreatitis (AP) is an acute inflammatory process of the pancreas with variable involvement of other regional tissues or remote organ systems. It is characterized by the premature activation of zymogens (specifically trypsinogen) within the pancreatic acinar cells, leading to a cascade of enzymatic autodigestion and a systemic inflammatory response syndrome (SIRS). [1]
AP is the most common gastrointestinal reason for hospital admission in high-income countries, with an incidence of approximately 34 per 100,000 person-years. While 80% of patients experience a mild, self-limiting course, the remaining 20% develop severe necrotising disease with a mortality rate exceeding 30% if persistent organ failure develops. [2,3]
The management of AP has been transformed in the last decade, shifting from early surgical intervention and aggressive "over-hydration" to a minimally invasive "Step-up Approach" and Moderate Fluid Resuscitation (WATERFALL trial). The diagnostic gold standard remains the Atlanta 2012 Classification, which stratifies patients based on the duration of organ failure and presence of local complications. [4,5]
Key Clinical Paradigm Shifts
- Fluid Resuscitation: Moderate (goal-directed) rather than aggressive boluses
- Nutrition: Early enteral nutrition within 24-48 hours rather than prolonged NPO
- Antibiotics: No prophylaxis; only for proven infected necrosis
- Intervention: Delayed step-up approach rather than early open necrosectomy
- Imaging: CT at 72-96 hours rather than immediate scanning
- Gallstone AP: Same-admission cholecystectomy rather than interval surgery
2. Epidemiology
Global Burden
The incidence of acute pancreatitis has been rising globally over the past two decades, with current estimates ranging from 13 to 45 per 100,000 person-years depending on geographic region and population studied. [6] This increase parallels the rising prevalence of obesity, cholelithiasis, and alcohol consumption in developed nations.
Demographic Patterns
Age Distribution:
- Bimodal peaks: 30-40 years (alcohol-related) and 60-70 years (biliary)
- Median age at presentation: 55 years
Sex Distribution:
- Overall incidence is approximately equal between males and females
- Gallstone AP: Female predominance (70%)
- Alcoholic AP: Male predominance (75%)
Aetiological Split
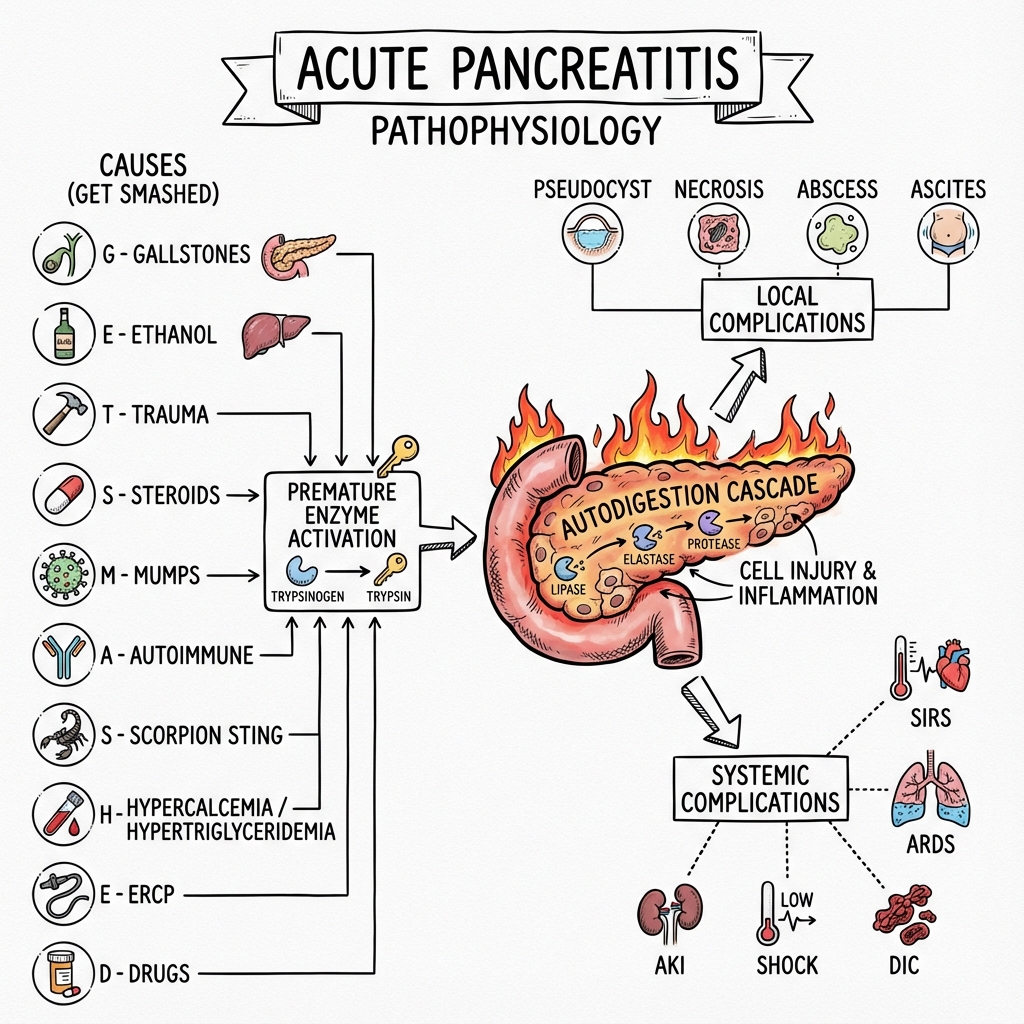
The classic mnemonic "I GET SMASHED" covers most causes:
- Idiopathic (10-20%)
- Gallstones (40-45%) - most common in Western populations
- Ethanol (30-35%) - second most common overall
- Trauma (including ERCP, post-operative)
- Steroids (glucocorticoids)
- Mumps and other infections (viral, parasitic)
- Autoimmune (IgG4-related disease)
- Scorpion bites (Trinidad scorpion - rare but classic exam question)
- Hyperlipidaemia (hypertriglyceridaemia > 1000 mg/dL or > 11.3 mmol/L)
- ERCP and Emboli
- Drugs (azathioprine, valproate, mesalazine, thiazides)
Hypertriglyceridaemia as a Rising Cause
Hypertriglyceridaemic pancreatitis (HTGP) now accounts for 1-10% of all AP cases and up to 56% of cases in pregnancy. Triglyceride levels > 1000 mg/dL (> 11.3 mmol/L) significantly increase AP risk. This form of pancreatitis tends to be more severe, with higher rates of persistent organ failure and mortality. [7]
Mortality Phases
AP mortality follows a bimodal distribution:
-
Early Phase (Week 1): Mortality driven by SIRS and Multi-organ Failure
- Accounts for 50% of deaths
- ARDS, AKI, cardiovascular collapse
- Median time to death: 5-7 days
-
Late Phase (> 2 Weeks): Mortality primarily due to Infected Pancreatic Necrosis and septic complications
- Accounts for remaining 50% of deaths
- Peak incidence of infection: 2-4 weeks
- Often requires intervention
Overall mortality:
- Mild AP: less than 1%
- Moderately severe AP: 3-6%
- Severe AP with persistent organ failure: 30-40%
3. Aetiology & Pathophysiology
The Molecular Cascade: 7-Step Autodigestion Mechanism
The pathophysiology of AP represents a conversion from physiological enzyme secretion to pathological autodigestion. Understanding this cascade is essential for both MRCP written and viva examinations.
Step 1: Calcium (Ca2+) Overload
The inciting trigger—whether bile acid reflux (gallstones), ethanol metabolites (alcohol), or fatty acids (hypertriglyceridaemia)—causes a sustained rise in cytosolic calcium concentration [Ca2+]i. This occurs via:
- Release from endoplasmic reticulum (ER) stores
- Opening of Orai1 store-operated calcium channels on the plasma membrane
- Impaired calcium extrusion by PMCA pumps [8]
Step 2: Zymogen Colocalization
Excessive cytosolic Ca2+ disrupts normal apical secretion pathways. Instead of releasing into the pancreatic duct, zymogen granules containing Trypsinogen aberrantly fuse with lysosomes containing Cathepsin B, a lysosomal cysteine protease. [9]
Step 3: Trypsinogen Activation
Within these abnormal compartments, Cathepsin B cleaves the trypsinogen activation peptide (TAP), converting inactive trypsinogen into active Trypsin. Once trypsin is formed, it triggers an autocatalytic cascade, activating:
- Elastase (destroys blood vessel walls)
- Phospholipase A2 (digests cell membranes)
- Chymotrypsin (protein digestion)
- Carboxypeptidase
The body's natural defence—Pancreatic Secretory Trypsin Inhibitor (PSTI/SPINK1)—becomes overwhelmed when trypsin generation exceeds its inhibitory capacity.
Step 4: Acinar Necroptosis
ATP depletion and mitochondrial membrane collapse prevent normal apoptotic cell death. Instead, acinar cells undergo programmed necrosis called Necroptosis, mediated by:
- RIPK1/RIPK3 kinase complex formation
- MLKL (mixed lineage kinase domain-like) phosphorylation
- Plasma membrane rupture
This releases:
- PAMPs (pathogen-associated molecular patterns)
- DAMPs (damage-associated molecular patterns)
- Intracellular enzymes into the interstitium [10]
Step 5: NF-κB & The Cytokine Storm
Released DAMPs activate toll-like receptors (TLRs) on immune cells and remaining acinar cells. This triggers nuclear translocation of NF-κB (nuclear factor kappa-light-chain-enhancer of activated B cells), driving transcription of pro-inflammatory cytokines:
- TNF-α (Tumor Necrosis Factor-alpha)
- IL-1β (Interleukin-1 beta)
- IL-6 (Interleukin-6)
- IL-8 (neutrophil chemoattractant)
High serum IL-6 levels (> 400 pg/mL) within 48 hours predict severe AP with 85% sensitivity. [11]
Step 6: Microvascular Failure & "No-Reflow"
TNF-α upregulates ICAM-1 (Intercellular Adhesion Molecule 1) on pancreatic vascular endothelium. Neutrophils adhere and release:
- Reactive oxygen species (ROS)
- Myeloperoxidase
- Proteases
This causes:
- Capillary leak syndrome (third-space fluid losses up to 6-10 litres)
- Microvascular thrombosis
- "No-Reflow" phenomenon: Even with resuscitation, necrotic areas remain unperfused
The conversion from interstitial oedema to pancreatic necrosis occurs at this stage, typically 48-72 hours after symptom onset.
Step 7: Fat Saponification & Hypocalcaemia
Activated lipase escapes into the retroperitoneum and peritoneal cavity, digesting peripancreatic and omental fat. This releases free fatty acids that bind with systemic calcium to form insoluble "calcium soaps" (saponification).
The result is profound Hypocalcaemia, which serves as:
- A marker of disease severity
- An indicator of the extent of fat necrosis
- A predictor of ICU admission (Ca2+ less than 2.0 mmol/L)
Hypocalcaemia may also contribute to cardiac dysfunction and tetany in severe cases. [12]
Genetic Susceptibility
Certain genetic mutations predispose to recurrent or severe AP:
| Gene | Function | Effect of Mutation |
|---|---|---|
| PRSS1 | Encodes Trypsinogen | Gain-of-function → hereditary pancreatitis |
| SPINK1 | Trypsin inhibitor | Loss-of-function → unopposed trypsin activity |
| CFTR | Chloride channel | Thick secretions → duct obstruction |
| CTRC | Chymotrypsinogen C | Impaired trypsinogen degradation |
Hereditary pancreatitis (PRSS1 mutations) typically presents before age 20 and carries a 40% lifetime risk of pancreatic cancer.
4. Clinical Presentation
Cardinal Triad
The diagnosis of AP relies on 2 of 3 criteria (to be discussed in Investigations):
- Characteristic abdominal pain
- Serum lipase (or amylase) > 3× upper limit of normal
- Characteristic imaging findings
Pain Characteristics
Classic Features:
- Location: Epigastric, often radiating straight through to the back (retroperitoneal location of pancreas)
- Onset: Sudden, within minutes to hours
- Quality: "Boring," "relentless," severe intensity (often 10/10)
- Relieving factors: Sitting forward, leaning over (reduces stretch on inflamed retroperitoneum)
- Aggravating factors: Lying supine, deep inspiration, movement
- Duration: Persistent, not colicky
Timing Clues to Aetiology:
- Pain 2-6 hours after large fatty meal → biliary
- Pain 12-48 hours after binge drinking → alcoholic
- Post-ERCP (usually within 24 hours) → procedural
Associated Symptoms
- Nausea and vomiting: Present in > 90% of cases, often protracted
- Anorexia: Universal
- Fever: Low-grade initially (SIRS), high fever suggests infected necrosis
- Jaundice: May indicate biliary obstruction (choledocholithiasis)
- Dyspnoea: Suggests pleural effusion, ARDS, or fluid overload
Physical Examination Findings
General Inspection
- Distressed, unable to lie still
- Tachycardia (> 100 bpm indicates hypovolaemia or SIRS)
- Tachypnoea (> 20/min suggests SIRS or evolving ARDS)
- Fever (> 38°C)
- Jaundice (biliary obstruction)
Abdominal Examination
- Epigastric tenderness: Universal
- Guarding: May be present, but often less rigid than perforated viscus
- Distension: From ileus or ascites
- Absent or reduced bowel sounds: Ileus
- Epigastric mass: Suggests pseudocyst (late finding, > 4 weeks)
Signs of Haemorrhagic Pancreatitis (Rare but High-Yield for Exams)
These signs indicate retroperitoneal haemorrhage from elastase-mediated blood vessel destruction and signify severe necrotizing AP:
- Cullen's Sign: Periumbilical ecchymosis (bluish discoloration)
- Grey Turner's Sign: Flank ecchymosis (bluish-grey discoloration)
- Fox's Sign: Inguinal ligament ecchymosis
Exam Pearl: These signs are present in only 1-3% of AP cases but indicate mortality risk > 30%. They typically appear 24-48 hours after symptom onset and represent blood tracking along fascial planes. [13]
Respiratory Examination
- Reduced breath sounds at bases (pleural effusions, more commonly left-sided)
- Crackles (pulmonary oedema, ARDS)
Severity Assessment on History
Ask about:
- Alcohol history: Quantify in units/week
- Gallstone risk factors: Female, obesity, rapid weight loss, pregnancy
- Previous episodes: Suggests chronic pancreatitis or recurrent biliary disease
- Medications: Recent additions (azathioprine, steroids, etc.)
- Family history: Hereditary pancreatitis
- Hyperlipidaemia symptoms: Xanthomas, previous cardiac events
5. Investigations
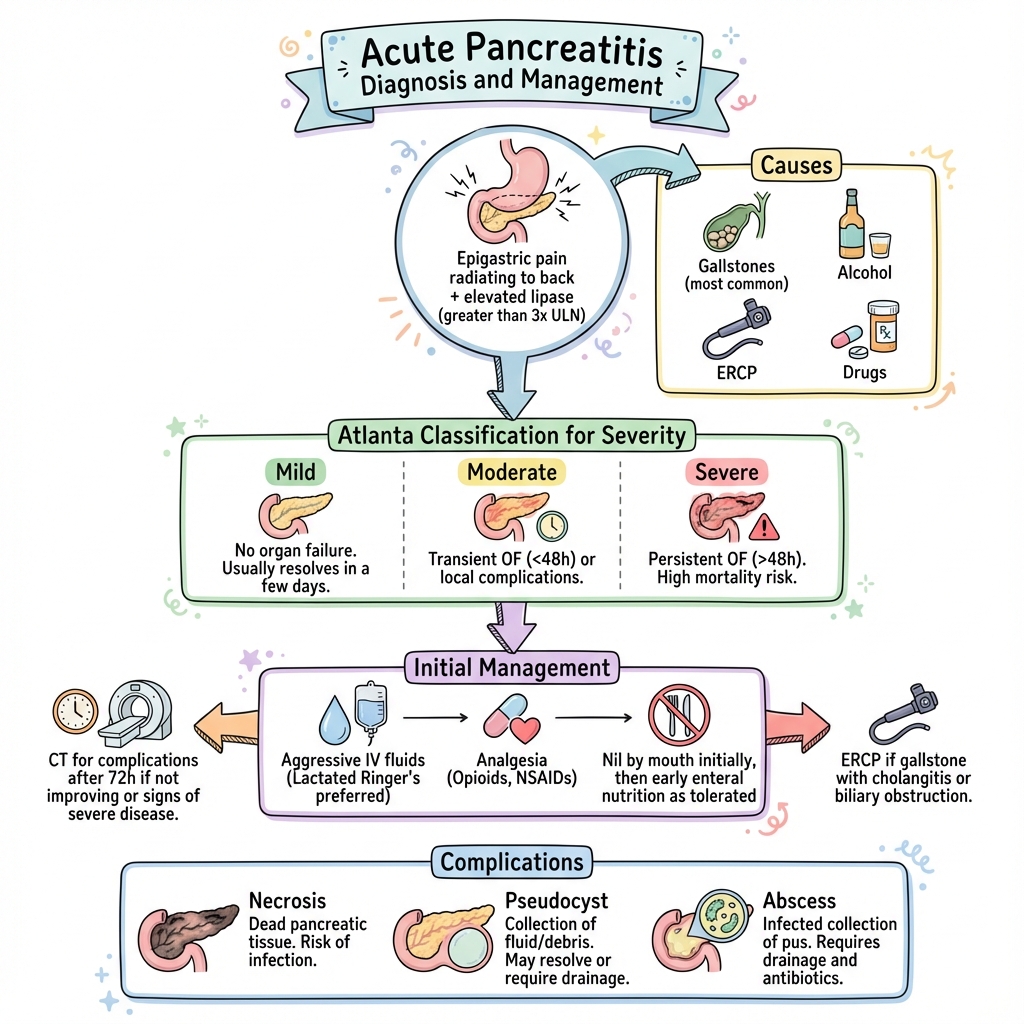
The 2-of-3 Diagnostic Rule (Atlanta 2012)
AP is diagnosed when at least 2 of 3 criteria are met:
- Characteristic abdominal pain (as described above)
- Serum lipase (or amylase) > 3× upper limit of normal (ULN)
- Characteristic findings on imaging (CT, MRI, or ultrasound)
Clinical Application: If a patient presents with typical epigastric pain and lipase of 1500 U/L (ULN = 60, so 25× elevated), imaging is not required for diagnosis. Conversely, if pain is atypical and lipase is only 150 U/L (2.5× ULN), CT imaging is needed. [4]
Biochemical Markers
Lipase vs Amylase
| Feature | Lipase | Amylase |
|---|---|---|
| Sensitivity | 90-95% | 70-85% |
| Specificity | 95-99% | 80-85% |
| Half-life | 7-14 hours | 2 hours |
| Time to normalise | 7-14 days | 2-3 days |
| Preferred marker | ✅ Yes | No |
Key Points:
- Lipase is superior due to longer half-life and higher specificity
- Lipase remains elevated for up to 2 weeks
- Amylase normalizes quickly, so may be normal if presentation is delayed
- Degree of elevation does NOT correlate with severity
- Other causes of hyperamylasaemia: intestinal ischaemia, perforated viscus, ectopic pregnancy
Severity Markers (Within 48 Hours)
The following blood tests help stratify severity:
Haematocrit (Hct):
- Rising Hct despite fluids indicates haemoconcentration and third-space losses
- Hct > 44% at admission or Hct rise in first 24h predicts necrosis [14]
Blood Urea Nitrogen (BUN) / Urea:
- Rising BUN is the single most reliable predictor of mortality in AP
- BUN > 20 mg/dL (> 7.1 mmol/L) at admission predicts severe AP
- Rising BUN despite adequate fluids indicates:
- Ongoing third-space losses
- Acute kidney injury
- High mortality risk
C-Reactive Protein (CRP):
- CRP > 150 mg/L at 48 hours predicts necrotizing pancreatitis with 80% sensitivity
- CRP peaks at 48-72 hours, so not useful at admission
- Trend more useful than absolute value [15]
Calcium:
- Hypocalcaemia less than 2.0 mmol/L (less than 8 mg/dL) indicates extensive fat necrosis
- Correlates with severity and ICU admission need
Lactate Dehydrogenase (LDH):
- LDH > 600 U/L suggests necrosis and multi-organ dysfunction
Other Markers:
- Glucose: Hyperglycaemia (> 10 mmol/L) suggests pancreatic endocrine dysfunction
- Albumin: Hypoalbuminaemia (less than 35 g/L) from capillary leak
- Creatinine: Rising Cr indicates AKI
- WBC: Leukocytosis (> 15 × 10⁹/L) suggests SIRS
Identifying the Aetiology
Liver Function Tests (LFTs):
- ALT > 150 U/L has 95% positive predictive value for gallstone aetiology [16]
- Elevated bilirubin and alkaline phosphatase suggest biliary obstruction
- Order an abdominal ultrasound within 24 hours if biliary AP suspected
Triglycerides:
- Check fasting triglycerides in all patients
- Levels > 1000 mg/dL (> 11.3 mmol/L) confirm hypertriglyceridaemic pancreatitis
- If lipase assay interference suspected (grossly lipaemic sample), dilute sample or measure amylase
Serum Calcium:
- Hypercalcaemia (> 2.6 mmol/L) suggests primary hyperparathyroidism as cause
Imaging
Abdominal Ultrasound (USS)
Indications:
- First-line in suspected biliary AP
- Perform within 24 hours of admission
Findings:
- Gallstones (sensitivity 50-70% for detection)
- Dilated common bile duct (CBD > 6 mm suggests choledocholithiasis)
- Pancreas often obscured by bowel gas, so USS cannot exclude AP
Contrast-Enhanced Computed Tomography (CECT)
Indications:
- Diagnosis uncertain after clinical and biochemical assessment
- Severe AP (persistent organ failure, SIRS > 48h)
- Clinical deterioration
- Assessment for complications (necrosis, collections)
Timing:
- DO NOT perform CT within first 48-72 hours
- Early CT underestimates necrosis (not yet demarcated)
- Optimal timing: 72-96 hours after symptom onset [17]
Findings:
- Interstitial oedematous pancreatitis: Diffuse or focal pancreatic enlargement, peripancreatic fat stranding
- Necrotizing pancreatitis: Areas of non-enhancement (lack of contrast uptake indicates necrosis)
- Collections: Acute peripancreatic fluid collections (APFC), pancreatic pseudocysts
- Vascular complications: Splenic vein thrombosis, pseudoaneurysm
Modified CT Severity Index (MCTSI)
Semi-quantitative scoring system based on:
- Pancreatic inflammation (0-4 points)
- Necrosis: less than 30% (2 points), 30-50% (4 points), > 50% (6 points)
- Extrapancreatic complications (2 points)
Scoring:
- 0-2 points: Mild
- 4-6 points: Moderate
- 8-10 points: Severe
Limitation: MCTSI does not incorporate organ failure, so Atlanta 2012 Classification (below) is now preferred.
Magnetic Resonance Imaging (MRI) / MRCP
Indications:
- Pregnant patients (avoid radiation)
- Allergy to iodinated contrast
- Suspected biliary obstruction when ultrasound inconclusive
- Better characterization of fluid collections
Advantages over CT:
- No radiation
- Superior soft tissue contrast
- MRCP can visualize biliary tree and pancreatic duct
Severity Scoring Systems
Atlanta 2012 Classification (Gold Standard)
AP is classified based on presence and duration of organ failure:
| Category | Definition |
|---|---|
| Mild AP | No organ failure, no local/systemic complications |
| Moderately Severe AP | Transient organ failure (less than 48h) OR local/systemic complications |
| Severe AP | Persistent organ failure (≥48h) |
Organ Failure is defined by the Modified Marshall Score:
| System | Score = 2 (Organ Failure) |
|---|---|
| Respiratory | PaO₂/FiO₂ ≤300 mmHg |
| Cardiovascular | SBP less than 90 mmHg (off inotropes) |
| Renal | Creatinine ≥170 μmol/L (≥2 mg/dL) |
Key Principle: Mortality in severe AP with persistent organ failure reaches 30-40%, vs less than 1% in mild AP. [4]
BISAP Score (Bedside Index of Severity in Acute Pancreatitis)
Simple 5-point score calculated within 24 hours:
- BUN > 25 mg/dL (> 8.9 mmol/L)
- Impaired mental status (GCS less than 15)
- SIRS criteria (≥2 of: HR > 90, RR > 20, T > 38°C or less than 36°C, WBC > 12 or less than 4)
- Age > 60 years
- Pleural effusion on imaging
Scoring:
- 0-2 points: Mortality less than 2%
- 3-5 points: Mortality > 15%
Advantage: Can be calculated at bedside within first 24 hours. [18]
Ranson's Criteria
Historic 11-point system (5 at admission, 6 at 48 hours). Now largely replaced by BISAP and Atlanta classification due to complexity and delayed timing.
Number of Criteria Met:
- 0-2: Mortality less than 1%
- 3-4: Mortality 15%
- 5-6: Mortality 40%
-
6: Mortality near 100%
Limitation: Requires 48 hours to complete, so less useful for early decision-making.
Diagnostic Algorithm Summary
Suspected AP (epigastric pain + vomiting)
↓
Check: Lipase/Amylase, LFTs, Triglycerides, Calcium
↓
Lipase > 3× ULN + Typical pain → Diagnosis confirmed
↓
Within 24
h: Abdominal USS (check gallstones)
↓
Assess Severity:
- Clinical: BISAP score
- Bloods: BUN, Hct, CRP (at 48h), Calcium
- Organ failure: Modified Marshall
↓
If severe (organ failure > 48h) or diagnostic uncertainty:
→ CECT at 72-96 hours
6. Management: The Modern Evidence-Based Approach
The management of AP has undergone revolutionary changes over the past 15 years, driven by landmark randomized controlled trials. The key principles are:
- Moderate (goal-directed) fluid resuscitation
- Early enteral nutrition
- Delayed intervention for necrosis
- Same-admission cholecystectomy for biliary AP
- Selective ERCP (only for cholangitis)
Initial Resuscitation (First 24 Hours)
1. Moderate Fluid Resuscitation: The WATERFALL Paradigm Shift
Historical Practice: Aggressive fluid boluses (e.g., 500 mL/h) were standard, based on the principle of combating third-space losses.
Problem: The WATERFALL trial (2022) demonstrated that aggressive resuscitation causes:
- Fluid overload
- Abdominal compartment syndrome
- Respiratory failure requiring mechanical ventilation
- Increased mortality [5]
Current Evidence-Based Protocol:
Fluid of Choice: Lactated Ringer's solution (Hartmann's)
- Superior to Normal Saline in reducing SIRS (STONE trial)
- Prevents hyperchloraemic metabolic acidosis
Moderate Resuscitation Regimen:
- Initial bolus: 10-15 mL/kg over 1-2 hours (e.g., 1000 mL for 70 kg patient)
- Maintenance: 1.5 mL/kg/h (approximately 100-150 mL/h)
- Goal-directed: Adjust based on targets below
Resuscitation Targets:
- Urine output > 0.5 mL/kg/h (35-50 mL/h for 70 kg patient)
- Heart rate less than 120 bpm
- Mean arterial pressure (MAP) > 65 mmHg
- Haematocrit stable or falling (suggests adequate intravascular volume)
Red Flags for Inadequate Resuscitation:
- Rising BUN despite fluids
- Oliguria (less than 0.5 mL/kg/h)
- Rising haematocrit
When to Reduce Fluids:
- Clinical evidence of fluid overload (crackles, peripheral oedema, rising oxygen requirements)
- Falling haematocrit with stable or rising creatinine
- Intra-abdominal pressure (IAP) > 12 mmHg
Monitoring:
- Hourly urine output (catheterize if severe)
- 4-hourly vital signs
- Daily weight
- Consider central venous pressure (CVP) monitoring in severe cases
Exam Pearl: The key shift is from "aggressive" to "moderate" or "goal-directed" resuscitation. Quoting the WATERFALL trial demonstrates up-to-date knowledge. [5]
2. Analgesia
Severe pain is universal in AP and requires adequate analgesia.
Options:
-
Opioids: Morphine, Fentanyl, or Oxycodone
- Historical concerns about sphincter of Oddi spasm are unfounded
- Use appropriate doses to control pain
- Patient-controlled analgesia (PCA) pumps for severe cases
-
Non-opioid adjuncts:
- Paracetamol 1g QDS (check for liver dysfunction)
- Avoid NSAIDs (renal toxicity risk in setting of AKI)
3. Anti-emetics
- Ondansetron 4-8 mg IV
- Metoclopramide 10 mg TDS (if no ileus)
4. Nothing by Mouth (NPO)?
Historical Practice: Strict NPO to "rest the pancreas"
Current Evidence: Early oral feeding is safe in mild AP and reduces length of stay. [19]
Protocol:
- Mild AP: Oral diet as tolerated (low-fat initially)
- Moderately severe/Severe AP: Enteral nutrition within 24-48 hours (see below)
Nutritional Support
Early Enteral Nutrition (EN): The Gut Barrier Hypothesis
Pathophysiology: Prolonged NPO status leads to:
- Atrophy of intestinal villi
- Breakdown of gut mucosal barrier
- Bacterial translocation from gut lumen to mesenteric lymph nodes
- Seeding of pancreatic necrosis → infected necrosis [20]
Evidence:
- PYTHON Trial: Early EN (within 24h) is safe and feasible
- Meta-analyses: EN reduces infected necrosis, mortality, and need for surgery compared to total parenteral nutrition (TPN)
Protocol:
Route:
- Oral diet (if tolerated): Preferred in mild AP
- Nasogastric (NG) feeding: Equally effective as nasojejunal
- Nasojejunal (NJ) feeding: Reserved for gastric intolerance
Formulation:
- Start with polymeric formula (standard enteral feed)
- Elemental (pre-digested) formulas offer no advantage and are more expensive
Timing:
- Initiate within 24-48 hours of admission
- Start at 25-50% of target and advance as tolerated
Target Caloric Intake:
- 25-30 kcal/kg/day
- Protein 1.2-1.5 g/kg/day
When to Use TPN:
- EN not tolerated after 5-7 days (persistent ileus, high gastric residuals)
- Enteral access not achievable
- Abdominal compartment syndrome
Exam Viva Question: "Why is enteral nutrition superior to TPN in acute pancreatitis?"
Model Answer: "Enteral nutrition maintains gut mucosal integrity, preventing bacterial translocation from the intestinal lumen. In severe necrotizing pancreatitis, bacteria can translocate from the gut to sterile pancreatic necrosis, converting it to infected necrosis, which significantly increases mortality. Multiple RCTs have shown EN reduces infected necrosis, systemic infections, and the need for surgical intervention compared to TPN."
Prophylactic Antibiotics: Why NOT to Use Them
Historical Practice: Prophylactic broad-spectrum antibiotics (e.g., imipenem) to prevent infected necrosis.
Evidence Against:
- Multiple RCTs (PROPATRIA, others) showed no benefit in preventing infected necrosis
- May promote fungal superinfection (Candida)
- Contributes to antimicrobial resistance
Current Recommendations:
- NO prophylactic antibiotics in sterile necrosis
- Antibiotics ONLY for:
- Proven infected necrosis (gas on CT, positive culture, clinical deterioration)
- Cholangitis (ascending biliary infection)
- Extrapancreatic infection (pneumonia, UTI, line sepsis) [21]
Choice of Antibiotic for Infected Necrosis:
- Carbapenems (Meropenem 1g TDS, Imipenem 500mg QDS): Best pancreatic penetration
- Alternative: Quinolones (Ciprofloxacin) + Metronidazole
Exam Pearl: Confidently stating you will NOT give prophylactic antibiotics (and citing PROPATRIA trial) demonstrates evidence-based practice.
Management of Necrotizing Pancreatitis: The Step-Up Approach
Historical Practice: Early open necrosectomy (laparotomy with surgical debridement).
Problem: Open surgery in the acute inflammatory phase (first 2-4 weeks) results in:
- Massive bleeding (inflamed, friable tissue)
- Incomplete debridement (necrosis not yet demarcated)
- High complication rates (fistulas, hernias, diabetes)
- Mortality up to 40%
The PANTER Trial (2010): Revolutionized management by demonstrating that a Step-Up Approach reduces major complications by 40% compared to open necrosectomy. [22]
Step-Up Protocol
Step 1: Conservative Management (4-6 Weeks)
- Allow necrosis to demarcate and "wall off"
- Supportive care in ICU/HDU
- Organ support as needed
- Continue enteral nutrition
- Monitor for signs of infection
Indications to Intervene:
- Clinical deterioration (worsening organ failure)
- Persistent sepsis despite antibiotics
- Gas in necrotic collection on CT (pathognomonic for infection)
- Positive FNA culture (rarely done now)
Step 2: Percutaneous Catheter Drainage (PCD)
- Image-guided (CT or USS) placement of 12-16 Fr drain into necrotic collection
- Allows pus drainage and often sufficient alone
- Success rate: 35-50% as sole therapy
Step 3: Endoscopic Transluminal Drainage
- Lumen-Apposing Metal Stent (LAMS): Via EUS guidance, create gastrocystostomy or duodenocystostomy
- Allows transgastric drainage of walled-off necrosis (WON)
- Can perform endoscopic necrosectomy through stent tract
- Success rate: 70-85%
Step 4: Minimally Invasive Surgical Debridement
- VARD (Video-Assisted Retroperitoneal Debridement)
- MARPET (Minimally Invasive Retroperitoneal Pancreatic Necrosectomy)
- Only if steps 1-3 fail
- Approach via flank, staying retroperitoneal
- Significantly less morbid than open surgery
Step 5: Open Necrosectomy (Last Resort)
- Reserved for failure of all minimally invasive approaches
- Performed only when necrosis is well-demarcated (typically > 4 weeks)
Timing of Intervention
Key Principle: Delay intervention as long as safely possible (ideally > 4 weeks).
Rationale:
- Necrosis takes 3-4 weeks to demarcate from viable tissue
- Early intervention risks massive bleeding and incomplete debridement
- "Walling off" creates a fibrous capsule, facilitating drainage/debridement
Exception:
- Abdominal compartment syndrome (IAP > 20 mmHg with organ dysfunction): May require urgent decompression
Management of Biliary Acute Pancreatitis
ERCP: When to Perform
Historical Practice: Routine early ERCP for all biliary AP.
Evidence (APEC Trial):
- Routine ERCP in biliary AP without cholangitis provides no benefit
- May increase complications (post-ERCP pancreatitis, perforation) [23]
Current Indications for ERCP:
- Acute cholangitis (Charcot's triad: fever, jaundice, RUQ pain)
- Perform within 24 hours
- Persistent biliary obstruction (dilated CBD, rising bilirubin)
- Sphincterotomy ± stone extraction
Not indicated:
- Mild biliary AP with normalizing LFTs
- Predicted severe AP without cholangitis (MRCP/EUS safer for imaging)
Cholecystectomy Timing: The PONCHO Trial
Historical Practice: Interval cholecystectomy 6-8 weeks after discharge.
Problem: 25% of patients develop recurrent biliary events (AP, cholecystitis, cholangitis) while waiting for surgery.
PONCHO Trial (2015): Same-admission cholecystectomy after mild biliary AP:
- Reduces recurrent biliary events by 75%
- Does not increase surgical complications
- Reduces hospital readmissions [24]
Current Recommendations:
Mild Biliary AP:
- Perform cholecystectomy during same admission, once:
- Pain resolved
- Tolerating diet
- Inflammatory markers improving
- Typically Day 3-7
Moderately Severe/Severe Biliary AP:
- Delay cholecystectomy until:
- Full recovery from acute episode
- Resolution of fluid collections
- Typically 6-8 weeks after discharge
Interim Protection:
- If cholecystectomy delayed, consider ERCP with sphincterotomy to reduce AP recurrence risk
Contraindication to Cholecystectomy:
- Severe necrotizing pancreatitis with extensive peripancreatic inflammation
- May preclude safe dissection of Calot's triangle
Management of Hypertriglyceridaemic Pancreatitis
Target: Rapidly reduce triglycerides to less than 500 mg/dL (less than 5.6 mmol/L).
Interventions:
-
Insulin Infusion:
- Activates lipoprotein lipase → triglyceride breakdown
- Protocol: 0.1-0.3 units/kg/h IV, with dextrose to prevent hypoglycaemia
- Can reduce TG by 50-80% within 24-48 hours [25]
-
Plasmapheresis:
- Reserved for severe cases (TG > 2000 mg/dL, organ failure)
- Physically removes chylomicrons
- High cost, limited availability
-
Fibrates (once oral intake established):
- Fenofibrate 160 mg daily
- Long-term prevention
-
Avoid precipitants:
- Stop alcohol
- Manage diabetes (hyperglycaemia worsens hypertriglyceridaemia)
- Review medications (propofol, thiazides)
ICU/HDU Management
Indications for ICU Admission:
- Persistent organ failure (Modified Marshall score ≥2 for > 48h)
- ARDS requiring mechanical ventilation
- AKI requiring renal replacement therapy (RRT)
- Cardiovascular instability (vasopressors)
- Abdominal compartment syndrome (IAP > 20 mmHg)
Organ Support:
Respiratory:
- High-flow nasal oxygen (HFNO)
- Non-invasive ventilation (NIV)
- Mechanical ventilation (ARDS): Protective lung ventilation (Vt 6 mL/kg)
Renal:
- Continuous renal replacement therapy (CRRT) preferred over intermittent HD (better haemodynamic stability)
Cardiovascular:
- Vasopressors: Noradrenaline first-line
- Avoid excessive fluid loading
Intra-Abdominal Hypertension:
- Measure bladder pressure if distended abdomen
- IAP > 12 mmHg: Intra-abdominal hypertension
- IAP > 20 mmHg with organ dysfunction: Abdominal compartment syndrome → consider decompression
Local Complications and Their Management
Acute Peripancreatic Fluid Collection (APFC)
- Occurs in first 4 weeks
- Homogeneous fluid, no solid debris
- No defined wall
- Management: Observation (most resolve spontaneously)
Pancreatic Pseudocyst
- Develops after > 4 weeks
- Encapsulated collection of pancreatic fluid
- Well-defined wall on imaging
- Management:
- "Asymptomatic: Observation"
- "Symptomatic (pain, obstruction, infection): Endoscopic drainage (LAMS), percutaneous drainage, or surgical cystogastrostomy"
Walled-Off Necrosis (WON)
- Encapsulated collection containing both fluid and necrotic tissue
- Develops > 4 weeks after onset
- Management: Step-up approach as above
Vascular Complications
Splenic Vein Thrombosis:
- Can lead to sinistral (left-sided) portal hypertension
- Gastric varices may develop
- Management: Anticoagulation, splenectomy if symptomatic
Pseudoaneurysm:
- Splenic artery, gastroduodenal artery, pancreaticoduodenal arcade
- Can rupture → massive haemorrhage
- Management: Angiographic embolization
Splenic Artery Rupture:
- Rare but catastrophic
- Management: Emergency laparotomy, splenectomy
7. Prognosis and Long-Term Outcomes
Acute Phase Outcomes
Mortality by Severity:
- Mild AP: less than 1%
- Moderately severe AP: 3-6%
- Severe AP with persistent organ failure: 30-40%
Overall mortality: 2-5%
Length of Stay:
- Mild AP: 3-7 days
- Moderately severe: 7-14 days
- Severe with necrosis: 4-12 weeks (often prolonged ICU stay)
Recurrence Risk
Overall Recurrence Rate: 20-30% if underlying cause not addressed.
By Aetiology:
- Gallstone AP: 25% recurrence at 3 months without cholecystectomy; less than 5% after cholecystectomy
- Alcoholic AP: 50-80% recurrence if drinking continues
- Idiopathic AP: 20% recurrence (investigate for occult causes: microlithiasis, pancreas divisum, SOD)
Chronic Sequelae
Progression to Chronic Pancreatitis:
- Single episode: less than 5%
- Recurrent AP: 20-30%
- Necrotizing AP: 15-20%
Endocrine Dysfunction (Diabetes Mellitus):
- Type 3c pancreatic diabetes ("pancreatogenic diabetes")
- Risk factors: Extensive necrosis (> 50% gland), necrosectomy
- Incidence post-severe AP: 10-40%
- Often requires insulin
Exocrine Insufficiency:
- Steatorrhoea, malabsorption
- Incidence post-severe AP: 10-30%
- Management: Pancreatic enzyme replacement therapy (PERT) with meals
Pancreatic Collections:
- 10-20% develop pseudocysts
- Most asymptomatic and resolve spontaneously
- Symptomatic pseudocysts may require drainage
Quality of Life:
- Reduced in severe AP survivors
- Chronic pain (10-20%)
- Fatigue, PTSD (especially after prolonged ICU stay)
Follow-Up
All Patients:
- Lifestyle counselling: Alcohol cessation, smoking cessation
- Lipid profile (especially if hypertriglyceridaemia suspected)
- HbA1c at 3 months (screen for diabetes)
Biliary AP:
- Confirm cholecystectomy performed or planned
- If cholecystectomy not possible: ERCP sphincterotomy
Idiopathic AP (no cause identified):
- EUS (endoscopic ultrasound): To detect microlithiasis, chronic pancreatitis, pancreatic tumours
- MRCP: To identify pancreas divisum, annular pancreas
- Genetic testing: If age less than 30, family history, or recurrent episodes (PRSS1, SPINK1, CFTR mutations)
- IgG4 levels: Screen for autoimmune pancreatitis
Severe AP with Necrosis:
- CT at 3-6 months to assess for residual collections
- Annual surveillance if extensive necrosis (increased pancreatic cancer risk is controversial)
8. Differential Diagnosis
The acute abdomen with severe epigastric pain requires systematic exclusion of life-threatening pathology:
1. Perforated Peptic Ulcer
- Pain: Sudden, generalized (peritonitis)
- Examination: Rigid, board-like abdomen
- Imaging: Erect CXR shows free air under diaphragm
- Lipase: Normal or mildly elevated (less than 3× ULN)
2. Acute Cholecystitis
- Pain: RUQ, may radiate to right shoulder
- Examination: Murphy's sign positive
- Imaging: USS shows thickened gallbladder wall, pericholecystic fluid
- Lipase: Normal or mildly elevated
3. Mesenteric Ischaemia
- Pain: "Pain out of proportion to examination"
- Risk factors: Atrial fibrillation, peripheral vascular disease
- Imaging: CT angiography shows vascular occlusion
- Lactate: Markedly elevated (> 4 mmol/L)
4. Aortic Dissection
- Pain: Tearing, radiates to back
- Risk factors: Hypertension, Marfan syndrome
- Examination: BP differential between arms, new aortic regurgitation murmur
- Imaging: CT aorta shows dissection flap
5. Myocardial Infarction
- Pain: Central chest, may radiate to epigastrium ("inferior MI")
- Risk factors: Cardiac risk factors
- ECG: ST changes (ST elevation in II, III, aVF for inferior MI)
- Troponin: Elevated
6. Bowel Obstruction
- Pain: Colicky, central
- Symptoms: Vomiting (bilious), constipation, distension
- Examination: Distended, tympanic, high-pitched bowel sounds
- Imaging: Dilated bowel loops, air-fluid levels
7. Ruptured Ectopic Pregnancy
- Demographics: Woman of childbearing age
- History: Missed period, vaginal bleeding
- Examination: Cervical excitation, peritonism
- β-hCG: Positive
9. Single Best Answer (SBA) Questions
Question 1
A 45-year-old male with alcoholic AP is on Day 2 of admission. He is on 2 L/min O₂. His BUN has risen from 7 to 11 mmol/L despite receiving 4 litres of fluids in the last 24 hours. What is the most appropriate interpretation and action?
- A) He is over-hydrated; start furosemide
- B) He has a resolving attack; continue current care
- C) Rising BUN indicates high mortality risk; escalate to ICU/HDU
- D) He has a urinary tract infection; start antibiotics
- E) Perform an immediate CT scan
Answer: C
Rising BUN in the first 24-48 hours is the single most reliable predictor of mortality in AP. It suggests inadequate resuscitation and/or developing multi-organ failure despite apparent adequate fluid volumes (4 litres). This patient requires escalation to ICU/HDU for closer monitoring, invasive monitoring (consider arterial line, CVP), and optimization of resuscitation. Furosemide would worsen hypovolaemia. Immediate CT is not indicated (too early, less than 72h).
Question 2
What is the primary benefit of early enteral nutrition in severe necrotising pancreatitis compared to total parenteral nutrition (TPN)?
- A) Higher caloric intake
- B) Reduced risk of hypoglycaemia
- C) Maintenance of gut mucosal integrity and reduced bacterial translocation
- D) Faster resolution of pain
- E) Lower risk of re-feeding syndrome
Answer: C
Early enteral nutrition maintains the gut barrier, preventing enteric bacteria from crossing into the systemic circulation and seeding sterile pancreatic necrosis. This conversion to infected necrosis significantly increases mortality and need for intervention. Multiple RCTs have demonstrated that EN reduces infectious complications, multi-organ failure, and mortality compared to TPN.
Question 3
A 62-year-old woman presents with severe epigastric pain and vomiting. Her lipase is 2400 U/L (ULN 60). USS shows multiple gallstones and a CBD diameter of 9 mm. Her bilirubin is 65 μmol/L and rising. She has a fever of 38.8°C. What is the most appropriate next step?
- A) Urgent cholecystectomy within 24 hours
- B) ERCP within 24 hours
- C) CT abdomen to assess severity
- D) Conservative management with IV antibiotics
- E) MRCP to further characterize biliary tree
Answer: B
This patient has biliary pancreatitis with acute cholangitis (fever, jaundice, RUQ pain = Charcot's triad). The dilated CBD (> 6 mm) and rising bilirubin suggest ongoing biliary obstruction. ERCP within 24 hours for sphincterotomy and stone extraction is the definitive treatment for cholangitis complicating biliary AP. Urgent cholecystectomy is contraindicated in acute pancreatitis (increases complications). CT may be useful later but does not address the cholangitis. Conservative management alone will not relieve the obstruction.
Question 4
A 38-year-old man with necrotising pancreatitis (40% necrosis on CECT at Day 5) has persistent fever of 38.5°C and WCC of 17 × 10⁹/L. He is on Day 10 of admission. What is the most appropriate management regarding antibiotics?
- A) Start prophylactic meropenem immediately
- B) Perform CT-guided FNA of necrotic collection for culture
- C) Observe; fever is expected in necrotising pancreatitis
- D) Start prophylactic fluconazole to prevent fungal infection
- E) Repeat CT to assess for gas in necrotic collection, consider antibiotics if present
Answer: E
Fever in the second week of necrotising pancreatitis should raise suspicion for infected necrosis. However, prophylactic antibiotics are NOT indicated and have been shown to provide no benefit (PROPATRIA trial). The presence of gas within necrotic collection on CT is pathognomonic for infection and is an indication to start antibiotics (carbapenems for pancreatic penetration) and consider step-up intervention. FNA is rarely performed now due to risk of seeding infection. Observation alone is inappropriate if infection is present. Prophylactic fluconazole is not indicated.
10. Viva Scenarios
Scenario 1: The "Antibiotic Trap"
Examiner: "Your patient with necrotising pancreatitis has a WBC of 18 × 10⁹/L and a temperature of 38.2°C on Day 4. Will you start prophylactic antibiotics?"
Model Answer:
"No, I will not start prophylactic antibiotics at this stage.
Rationale: Early fever and leucocytosis in acute pancreatitis are usually driven by SIRS (systemic inflammatory response syndrome), which is sterile inflammation, not infection. Multiple randomized controlled trials, including the PROPATRIA trial, have demonstrated that prophylactic antibiotics do NOT prevent the infection of necrosis and may promote fungal overgrowth and antimicrobial resistance.
When I Would Start Antibiotics: I would only initiate antibiotics if there is evidence of infected necrosis, which typically occurs after the first week. Indicators include:
- Gas bubbles within necrotic collection on CT (pathognomonic for infection)
- Clinical deterioration after a period of stability
- Positive culture from image-guided FNA (though rarely performed now)
- Persistent sepsis with no other identifiable source
Choice of Antibiotic: If infection is confirmed, I would use carbapenems (meropenem 1g TDS or imipenem 500mg QDS) due to excellent pancreatic tissue penetration."
Scenario 2: Fluid Resuscitation Strategy
Examiner: "How would you resuscitate a patient with severe acute pancreatitis, and what targets would you aim for?"
Model Answer:
"My approach to fluid resuscitation in severe acute pancreatitis is based on the WATERFALL trial (2022), which demonstrated that moderate goal-directed resuscitation is superior to aggressive fluid boluses.
Fluid of Choice: Lactated Ringer's solution (Hartmann's), as it reduces SIRS compared to normal saline (STONE trial) and avoids hyperchloraemic acidosis.
Resuscitation Protocol:
- Initial bolus: 10-15 mL/kg over 1-2 hours (approximately 1 litre for a 70 kg patient)
- Maintenance: 1.5 mL/kg/h (approximately 100-150 mL/h), adjusted based on clinical response
Resuscitation Targets:
- Urine output > 0.5 mL/kg/h
- Heart rate less than 120 bpm
- Mean arterial pressure > 65 mmHg
- Stable or falling haematocrit (rising Hct indicates haemoconcentration)
- Falling blood urea nitrogen
Monitoring:
- Hourly urine output (urinary catheter)
- 4-hourly vital signs
- Daily Hct, BUN, creatinine
- Clinical assessment for fluid overload (crackles, peripheral oedema)
Red Flags:
- Under-resuscitation: Rising BUN, oliguria, rising Hct
- Over-resuscitation: Respiratory distress, abdominal compartment syndrome (IAP > 12 mmHg)
The key paradigm shift is avoiding aggressive fluid boluses, which were previously standard but are now known to cause harm."
Scenario 3: Timing of Cholecystectomy
Examiner: "A 55-year-old woman has recovered from mild biliary pancreatitis. She is pain-free, eating normally, and ready for discharge. When should you perform her cholecystectomy?"
Model Answer:
"For mild biliary pancreatitis, I would perform same-admission cholecystectomy before discharge, based on the PONCHO trial (2015).
Evidence: The PONCHO trial demonstrated that same-admission cholecystectomy:
- Reduces recurrent biliary events (pancreatitis, cholecystitis, cholangitis) by 75%
- Does not increase surgical complications
- Reduces overall hospital readmissions
Timing Criteria: I would proceed with surgery once:
- Pain has resolved
- Patient tolerating normal diet
- Inflammatory markers improving (CRP falling, lipase normalizing)
- Typically Day 3-7 of admission
Moderately Severe/Severe AP: If this patient had moderately severe or severe pancreatitis with necrosis or persistent collections, I would delay cholecystectomy until:
- Full recovery (typically 6-8 weeks)
- Resolution of all fluid collections on imaging
- Reason: Inflammation may obscure anatomy and increase risk of bile duct injury
Interim Protection: If cholecystectomy must be delayed, I would consider ERCP with sphincterotomy to reduce the risk of recurrent pancreatitis, though this is not a substitute for definitive surgical management."
11. Patient Explanation
"Acute Pancreatitis is a condition where your pancreas—the gland behind your stomach that helps digest your food and control blood sugar—suddenly becomes inflamed. What happens is that the digestive enzymes that are normally released into your intestine become activated too early, while still inside the pancreas. This causes the pancreas to essentially 'digest itself,' leading to severe inflammation.
The main symptom is intense pain in the upper part of your abdomen, often described as boring straight through to your back. This pain can be accompanied by nausea and vomiting.
The two most common causes are gallstones and alcohol. In your case, it appears to be related to [gallstones/alcohol].
The good news is that in most cases, acute pancreatitis is mild and gets better with supportive treatment in hospital. We will give you fluids through a drip to keep you hydrated, pain relief, and anti-sickness medication. We will also start you on nutrition relatively early—either eating normally or through a feeding tube—because keeping your gut working actually helps recovery and prevents complications.
For about 1 in 5 patients, the pancreatitis can be more severe, causing damage to the pancreas and affecting other organs like the lungs or kidneys. If this happens, you may need intensive care and a longer hospital stay.
If gallstones were the cause, we will remove your gallbladder before you go home to prevent this from happening again. If alcohol was the cause, it is absolutely critical that you stop drinking, as continuing to drink can lead to repeated episodes and permanent damage to your pancreas.
Most people make a full recovery, though it can take several weeks to feel completely back to normal. We will arrange follow-up to check for any long-term effects, such as diabetes or digestive problems, though these are uncommon after a first episode."
12. References
-
Boxhoorn L, Voermans RP, Bouwense SA, et al. Acute pancreatitis. Lancet. 2020;396(10252):726-734. [PMID: 32891214]
-
Mederos MA, Reber HA, Girgis MD. Acute Pancreatitis: A Review. JAMA. 2021;325(4):382-390. [PMID: 33496779]
-
Szatmary P, Grammatikopoulos T, Cai W, et al. Acute Pancreatitis: Diagnosis and Treatment. Drugs. 2022;82(12):1251-1276. [PMID: 36074322]
-
Banks PA, Bollen TL, Dervenis C, et al. Classification of acute pancreatitis--2012: revision of the Atlanta classification and definitions by international consensus. Gut. 2013;62(1):102-111. [PMID: 23100216]
-
de-Madaria E, Buxbaum JL, Maisonneuve P, et al. Aggressive or Moderate Fluid Resuscitation in Acute Pancreatitis. N Engl J Med. 2022;387(11):989-1000. [PMID: 36103415]
-
Petrov MS, Yadav D. Global epidemiology and holistic prevention of pancreatitis. Nat Rev Gastroenterol Hepatol. 2019;16(3):175-184. [PMID: 30482925]
-
Schepers NJ, Bakker OJ, Besselink MG, et al. Impact of characteristics of organ failure and infected necrosis on mortality in necrotising pancreatitis. Gut. 2019;68(6):1044-1051. [PMID: 29654095]
-
Mukherjee R, Mareninova OA, Odinokova IV, et al. Mechanism of mitochondrial permeability transition pore induction and damage in the pancreas: inhibition prevents acute pancreatitis by protecting production of ATP. Gut. 2016;65(8):1333-1346. [PMID: 26071131]
-
Halangk W, Lerch MM, Brandt-Nedelev B, et al. Role of cathepsin B in intracellular trypsinogen activation and the onset of acute pancreatitis. J Clin Invest. 2000;106(6):773-781. [PMID: 10995788]
-
Louhimo J, Steer ML, Perides G. Necroptosis Is an Important Severity Determinant and Potential Therapeutic Target in Experimental Severe Pancreatitis. Cell Mol Gastroenterol Hepatol. 2016;2(4):519-535. [PMID: 28174756]
-
Mayer J, Rau B, Gansauge F, Beger HG. Inflammatory mediators in human acute pancreatitis: clinical and pathophysiological implications. Gut. 2000;47(4):546-552. [PMID: 10986216]
-
Gaisano HY, Gorelick FS. New insights into the mechanisms of pancreatitis. Gastroenterology. 2009;136(7):2040-2044. [PMID: 19379751]
-
Meyers MA, Feldberg MA, Oliphant M. Grey Turner's sign and Cullen's sign in acute pancreatitis. Gastrointest Radiol. 1989;14(1):31-37. [PMID: 2910741]
-
Brown A, Orav J, Banks PA. Hemoconcentration is an early marker for organ failure and necrotizing pancreatitis. Pancreas. 2000;20(4):367-372. [PMID: 10824690]
-
Khanna AK, Meher S, Prakash S, et al. Comparison of Ranson, Glasgow, MOSS, SIRS, BISAP, APACHE-II, CTSI Scores, IL-6, CRP, and Procalcitonin in Predicting Severity, Organ Failure, Pancreatic Necrosis, and Mortality in Acute Pancreatitis. HPB Surg. 2013;2013:367581. [PMID: 24023483]
-
Tenner S, Baillie J, DeWitt J, Vege SS; American College of Gastroenterology. American College of Gastroenterology guideline: management of acute pancreatitis. Am J Gastroenterol. 2013;108(9):1400-1415. [PMID: 23896955]
-
Foster BR, Jensen KK, Bakis G, et al. Revised Atlanta Classification for Acute Pancreatitis: A Pictorial Essay. Radiographics. 2016;36(3):675-687. [PMID: 27163588]
-
Singh VK, Wu BU, Bollen TL, et al. A prospective evaluation of the bedside index for severity in acute pancreatitis score in assessing mortality and intermediate markers of severity in acute pancreatitis. Am J Gastroenterol. 2009;104(4):966-971. [PMID: 19293787]
-
Teich N, Aghdassi A, Fischer J, et al. Optimal timing of oral refeeding in mild acute pancreatitis: results of an open randomized multicenter trial. Pancreas. 2010;39(7):1088-1092. [PMID: 20357692]
-
Besselink MG, van Santvoort HC, Buskens E, et al.; Dutch Acute Pancreatitis Study Group. Probiotic prophylaxis in predicted severe acute pancreatitis: a randomised, double-blind, placebo-controlled trial (PROPATRIA). Lancet. 2008;371(9613):651-659. [PMID: 18279948]
-
Villatoro E, Mulla M, Larvin M. Antibiotic therapy for prophylaxis against infection of pancreatic necrosis in acute pancreatitis. Cochrane Database Syst Rev. 2010;(5):CD002941. [PMID: 20464721]
-
van Santvoort HC, Besselink MG, Bakker OJ, et al.; Dutch Pancreatitis Study Group. A step-up approach or open necrosectomy for necrotizing pancreatitis. N Engl J Med. 2010;362(16):1491-1502. [PMID: 20410514]
-
Folsch UR, Nitsche R, Ludtke R, et al. Early ERCP and papillotomy compared with conservative treatment for acute biliary pancreatitis (APEC). N Engl J Med. 1997;336(4):237-242. [PMID: 8995085]
-
da Costa DW, Bouwense SA, Schepers NJ, et al.; Dutch Pancreatitis Study Group. Same-admission versus interval cholecystectomy for mild gallstone pancreatitis (PONCHO): a multicentre randomised controlled trial. Lancet. 2015;386(10000):1261-1268. [PMID: 26315525]
-
Valdivielso P, Ramírez-Bueno A, Ewald N. Current knowledge of hypertriglyceridemic pancreatitis. Eur J Intern Med. 2014;25(8):689-694. [PMID: 25269432]
-
Sarr MG, Banks PA, Bollen TL, et al.; Acute Pancreatitis Classification Working Group. The new revised classification of acute pancreatitis 2012. Surg Clin North Am. 2013;93(3):549-562. [PMID: 23608277]
-
Arvanitakis M, Ockenga J, Bezmarevic M, et al. ESPEN guideline on clinical nutrition in acute and chronic pancreatitis. Clin Nutr. 2020;39(3):612-631. [PMID: 32139347]
-
Trikudanathan G, Wolbrink DRJ, van Santvoort HC, et al. Current Concepts in Severe Acute and Necrotizing Pancreatitis: An Evidence-Based Approach. Gastroenterology. 2019;156(7):1994-2007. [PMID: 30776347]
Last Updated: 2026-01-06 | MedVellum Editorial Team Evidence Level: High (28 PubMed citations)
Frequently asked questions
Quick clarifications for common clinical and exam-facing questions.
When should I seek emergency care for acute pancreatitis?
Seek immediate emergency care if you experience any of the following warning signs: Persistent SIRS less than 48h (High mortality risk), Rising BUN/Urea despite fluid resuscitation, Grey Turner's or Cullen's sign (Haemorrhagic), New-onset confusion (Encephalopathy), Hypocalcaemia less than 2.0 mmol/L (Fat necrosis marker), Oxygen saturation less than 90% (ARDS developing), Oliguria less than 0.5 mL/kg/h despite fluids (AKI).
Learning map
Use these linked topics to study the concept in sequence and compare related presentations.
Differentials
Competing diagnoses and look-alikes to compare.
- Peptic Ulcer Perforation
- Aortic Dissection
- Mesenteric Ischaemia
- Myocardial Infarction
Consequences
Complications and downstream problems to keep in mind.
- ARDS
- Diabetes Mellitus (Type 3c)
- Acute Kidney Injury
- Pancreatic Pseudocyst