Anaphylaxis
Anaphylaxis is a severe, life-threatening, generalised or systemic hypersensitivity reaction characterised by rapidly de... MRCP exam preparation.
What matters first
Anaphylaxis is a severe, life-threatening, generalised or systemic hypersensitivity reaction characterised by rapidly de... MRCP exam preparation.
Stridor (Impending complete airway obstruction)
6 Jan 2026
Generated educational material; verify before clinical use.
Visible references section
See the concept before reading it
Study the key anatomy, imaging, and decision pathways as full teaching plates.
Clinical board
A visual summary of the highest-yield teaching signals on this page.
Urgent signals
Safety-critical features pulled from the topic metadata.
- Stridor (Impending complete airway obstruction)
- Silent chest (Severe bronchospasm)
- Hypotension unresponsive to IM Adrenaline
- Bradycardia in a shocked patient (Pre-terminal)
Exam focus
Current exam surfaces linked to this topic.
- MRCP
Linked comparisons
Differentials and adjacent topics worth opening next.
- Mastocytosis
- Angioedema (Hereditary)
Content status and exam context
This page is AI-generated educational content. It may contain errors or omissions and is not a substitute for current guidelines, local protocols, senior clinical judgement, or professional medical advice.
MedVellum does not claim an individual clinician reviewer, board certification, or professional credential for this page unless a future version names a real, verifiable reviewer.
Topic family
This concept exists in multiple MedVellum libraries. Use the primary page for the broadest reference view and the others for exam-specific framing.
Anaphylaxis is a severe, life-threatening, generalised or systemic hypersensitivity reaction characterised by rapidly de... MRCP exam preparation.
Anaphylaxis is a rapid-onset, IgE or non-IgE mediated systemic allergic reaction affecting multiple organ systems. It af... ACEM Primary Written, ACEM Primary V
Clinical explanation and evidence
Anaphylaxis (Adult)
1. Clinical Overview
Anaphylaxis is a severe, life-threatening, generalised or systemic hypersensitivity reaction characterised by rapidly developing airway, breathing, and/or circulation problems, usually associated with skin and mucosal changes. [1,2] It represents the maximal expression of Type 1 (IgE-mediated) hypersensitivity and constitutes one of the most time-critical emergencies in clinical medicine.
Definition and Diagnostic Criteria
The World Allergy Organization defines anaphylaxis as "a serious, generalized or systemic, allergic or hypersensitivity reaction that can be life-threatening or fatal." [3] Clinical diagnosis is established when any ONE of the following three criteria is fulfilled:
Criterion 1: Acute onset (minutes to hours) with involvement of skin/mucosa PLUS either:
- Respiratory compromise (wheeze, stridor, hypoxia)
- Reduced blood pressure or end-organ dysfunction
Criterion 2: Two or more of the following after likely allergen exposure:
- Skin/mucosal involvement
- Respiratory compromise
- Reduced blood pressure
- Persistent gastrointestinal symptoms
Criterion 3: Reduced blood pressure after exposure to a known allergen:
- Adults: Systolic BP less than 90 mmHg or > 30% decrease from baseline
- Infants/children: Age-specific low BP or > 30% decrease [4]
Clinical Significance
The defining characteristic of anaphylaxis is its potential for near-instant fatality: in drug-induced cases, cardiac arrest can occur within 5 minutes of exposure, with a median time of 5 minutes for IV drugs, 15 minutes for stings, and 30 minutes for food. [5] The hallmark of contemporary (2020-2024) management guidelines is the absolute primacy of Intramuscular (IM) Adrenaline and the evidence-based downgrade of "traditional" therapies like antihistamines and corticosteroids, which have no role in the acute life-saving phase. [1,6]
Diagnosis remains entirely clinical at the point of emergency intervention, though it is retrospectively supported by serial Mast Cell Tryptase measurements. Management must be instinctive and protocol-driven: rapid recognition, prompt adrenaline administration, aggressive fluid resuscitation, and strict supine positioning to prevent catastrophic cardiovascular collapse. [7]
2. Epidemiology
Incidence and Prevalence
Anaphylaxis affects approximately 1-3% of the general population at some point during their lifetime, with lifetime prevalence estimates ranging from 0.3% to 5.1% depending on geographical region and diagnostic criteria. [8,9] The incidence has been steadily increasing over the past three decades, with a documented 2-fold to 5-fold rise per decade in high-income countries, attributed to increased food allergen exposure, enhanced recognition, and improved diagnostic coding. [10]
Current population-based studies report incidence rates of:
- 50-200 cases per 100,000 person-years in Europe
- 49.8 per 100,000 person-years in the United States
- Hospital admission rates: 10-20 per 100,000 annually [8,9]
Age and Gender Distribution
Anaphylaxis exhibits a bimodal age distribution:
- First peak: Children and adolescents (predominantly food-induced)
- Second peak: Adults aged 50-70 years (predominantly drug and venom-induced)
Gender patterns vary by age:
- Children: Male predominance (60-65%)
- Adults: Slight female predominance (55-60%), possibly related to higher rates of drug allergy and occupational latex exposure [11]
Trigger Variation by Demographics
Adults:
- Drugs (30-40%): NSAIDs, antibiotics (β-lactams, fluoroquinolones), neuromuscular blocking agents (NMBAs)
- Venom (20-25%): Hymenoptera (bees, wasps, hornets)
- Food (20-25%): Shellfish, peanuts, tree nuts
- Idiopathic (10-15%): No identifiable trigger despite investigation [12]
Peri-operative Setting:
- Neuromuscular blockers (e.g., Suxamethonium, Rocuronium) are the leading cause of anaesthesia-associated anaphylaxis (50-70% of cases)
- Antibiotics (15-20%)
- Latex (5-10%, declining with powder-free glove policies) [13]
Mortality and High-Risk Groups
Overall case fatality rate: 0.3-1% of all anaphylaxis episodes [14]
Fatal anaphylaxis incidence: 0.05-0.51 per million person-years
Highest risk groups for fatal anaphylaxis:
- Asthma (especially poorly controlled): 5-10 fold increased risk in food-induced anaphylaxis
- Cardiovascular disease: Increased risk of Kounis syndrome
- Mastocytosis: Elevated baseline tryptase, severe reactions to Hymenoptera venom
- Medications: Patients on β-blockers (refractory shock) or ACE inhibitors (prolonged angioedema)
- Delayed adrenaline: Each 30-minute delay in adrenaline administration increases mortality risk
- Upright posture: "Empty ventricle syndrome" causing sudden cardiac arrest [5,14,15]
Time to cardiac arrest by trigger (UK Registry data):
- IV drugs: 5 minutes (median)
- Stings/venom: 15 minutes
- Food: 30 minutes [5]
3. Aetiology and Pathophysiology
Classification: Immunological vs Non-Immunological
Immunological Anaphylaxis (Classical):
- IgE-mediated (Type 1 hypersensitivity): Most common mechanism
- IgG-mediated: Rare, documented with dextran and some monoclonal antibodies
- Immune complex: Rare (e.g., blood transfusion reactions)
Non-Immunological Anaphylaxis (Anaphylactoid):
- Direct mast cell/basophil degranulation without IgE
- Examples: Opioids, radiocontrast media, exercise
- Clinically indistinguishable from IgE-mediated reactions [16]
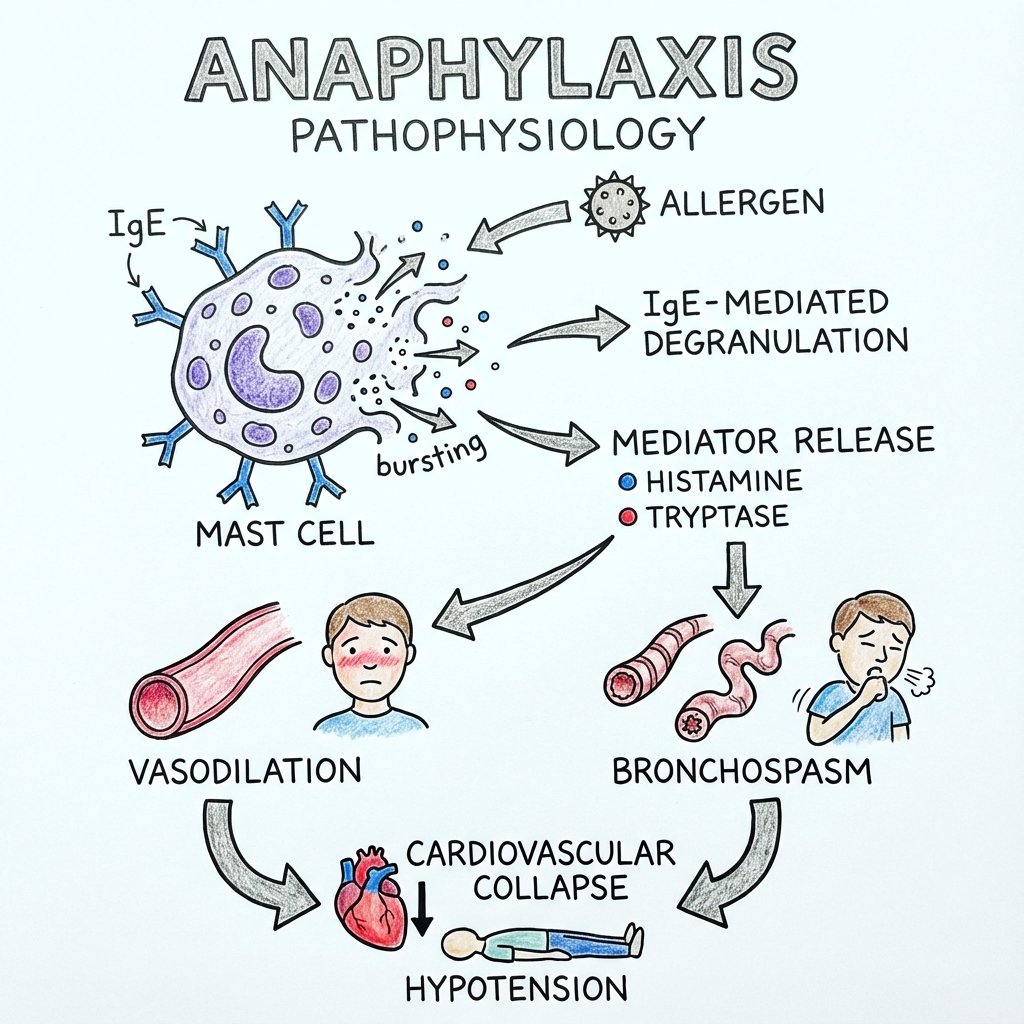
The 7-Step Molecular Mechanism (IgE-Mediated)
Step 1: Sensitisation Phase
Initial exposure to an allergen (e.g., peanut protein, penicillin) is processed by dendritic cells and presented to T-helper cells. In atopic individuals, Th2 cells release IL-4 and IL-13, which drive B-cell class-switching to produce allergen-specific IgE antibodies. These IgE molecules bind to high-affinity FcεRI receptors on the surface of mast cells (tissue-resident) and basophils (circulating), priming them for future activation. This sensitisation phase is asymptomatic. [17]
Step 2: Allergen Cross-Linking
Upon re-exposure, the allergen (acting as a multivalent antigen) binds to and cross-links two or more adjacent IgE-FcεRI complexes on the mast cell surface. This physical cross-linking is the critical triggering event. [17]
Step 3: Kinase Activation Cascade
Cross-linking triggers the phosphorylation and activation of the Lyn and Syk tyrosine kinases, initiating a complex intracellular signaling cascade involving phospholipase C-γ (PLCγ), phosphoinositide 3-kinase (PI3K), and protein kinase C (PKC). [16,17]
Step 4: The Calcium Surge
The signaling cascade culminates in the production of inositol 1,4,5-trisphosphate (IP3), which triggers the release of calcium (Ca²⁺) from intracellular stores (endoplasmic reticulum). This massive influx of cytosolic calcium is the "ignition switch" for degranulation. [17]
Step 5: Exocytosis – Phase 1 (Immediate Mediators)
Within seconds to minutes, calcium-dependent mechanisms trigger the fusion of cytoplasmic granules with the plasma membrane, releasing pre-formed mediators:
- Histamine: Binds to H1 receptors (vasodilation, increased vascular permeability, bronchoconstriction) and H2 receptors (vasodilation, increased gastric acid)
- Tryptase: Serine protease; activates complement, degrades fibrinogen, activates PAR-2 receptors
- Heparin: Anticoagulant; may contribute to bleeding diathesis
- Chymase: Converts angiotensin I to angiotensin II; activates metalloproteinases [16,18]
Step 6: Lipid Mediator Synthesis – Phase 2 (Late Mediators)
Within minutes to hours, mast cells synthesize de novo lipid mediators from arachidonic acid:
- Leukotrienes (LTC₄, LTD₄, LTE₄): Collectively termed "slow-reacting substance of anaphylaxis" (SRS-A), these are 1000-fold more potent bronchoconstrictors than histamine and cause sustained vascular leak
- Prostaglandin D₂ (PGD₂): Potent vasodilator and bronchoconstrictor
- Platelet-activating factor (PAF): Amplifies vascular permeability, activates platelets, and recruits inflammatory cells [16,18]
Step 7: Systemic Vasodilation and Capillary Leak
The combined effect of histamine (via H1/H2 receptors), leukotrienes, and PAF causes:
- Massive systemic vasodilation: Arteriolar smooth muscle relaxation → distributive shock
- Increased capillary permeability: Endothelial gap junction opening → fluid extravasation
Critical consequence: Up to 35% of the circulating blood volume can shift from the intravascular space to the interstitium within 10 minutes, resulting in profound hypovolaemia and distributive shock. This is compounded by:
- Direct myocardial depression (histamine on H2 receptors)
- Coronary artery spasm (Kounis syndrome)
- Arrhythmias (myocardial hypoxia and mediator effects) [7,16,18]
Cofactors and Amplifiers
Certain conditions lower the threshold for anaphylaxis or amplify its severity:
- Exercise: Increases intestinal permeability (food-dependent exercise-induced anaphylaxis)
- Alcohol: Vasodilation and increased allergen absorption
- NSAIDs: Augment mast cell degranulation
- Acute infection: Shifts immune balance toward Th2
- Menstruation: Hormonal effects on mast cell sensitivity [19]
4. Clinical Presentation
The ABC Pattern: Sudden Multi-System Involvement
Anaphylaxis typically presents as a sudden-onset (within minutes to 2 hours) multi-system syndrome. The classical triad consists of:
A – Airway Compromise
- Oropharyngeal angioedema: Swollen lips, tongue (macroglossia), uvula, soft palate
- Laryngeal oedema: Hoarseness, voice change ("hot potato voice"), difficulty swallowing
- Stridor: High-pitched inspiratory noise indicating critical airway narrowing (RED FLAG)
- Sensation of throat closure: Patient may grab neck (universal choking sign) [1,20]
B – Breathing Difficulty
- Bronchospasm: Widespread expiratory wheeze, chest tightness
- Tachypnoea: Respiratory rate > 24/min
- Hypoxia: SpO₂ less than 92% on room air
- Silent chest: Absence of breath sounds despite respiratory effort (RED FLAG – indicates near-total airway obstruction) [1,20]
C – Circulatory Collapse
- Hypotension: Systolic BP less than 90 mmHg or > 30% drop from baseline
- Tachycardia: Heart rate > 100-120/min (compensatory)
- Bradycardia: RED FLAG – pre-terminal sign indicating severe myocardial hypoxia
- Syncope or collapse: Loss of consciousness from cerebral hypoperfusion
- Pallor, clammy skin: Peripheral vasoconstriction (may be absent if flushing predominates)
- Cardiac arrest: Pulseless electrical activity (PEA) or asystole in severe cases [7,20]
Skin and Mucosal Changes (Present in 80-90%)
- Urticaria (hives): Pruritic, raised, erythematous wheals
- Angioedema: Non-pitting swelling of deeper dermis/subcutaneous tissues (face, lips, tongue, genitalia)
- Flushing: Diffuse erythema, especially face and upper trunk
- Pruritus: Generalised itching, including palms, soles, scalp
CRITICAL: Skin signs are absent in 10-20% of fatal anaphylaxis cases, especially in rapid cardiovascular collapse. Absence of skin involvement should NOT delay diagnosis or treatment. [5,21]
Gastrointestinal Symptoms (30-40%)
- Nausea and vomiting
- Abdominal cramping and pain
- Diarrhoea (may be bloody in severe cases)
Neurological Symptoms
- Sense of impending doom (patient reports "something is terribly wrong")
- Confusion, agitation (from cerebral hypoperfusion)
- Seizures (rare, from severe hypoxia)
Temporal Patterns
Uniphasic (Most common, ~80%):
- Single episode with resolution after treatment
Biphasic (~5-20%):
- Initial symptoms resolve, followed by recurrence of symptoms 1-72 hours later (median 8-10 hours) without re-exposure to allergen
- Risk factors: Severe initial reaction, delayed adrenaline, food triggers
- Justifies prolonged observation (minimum 6-12 hours) [22,23]
Protracted (Rare, less than 5%):
- Symptoms persist for > 24 hours despite treatment
- Often requires ICU admission and prolonged adrenaline infusion [22]
Atypical Presentations
Isolated hypotension (especially in perioperative setting):
- Sudden cardiovascular collapse without skin or respiratory signs
- High index of suspicion required [13]
Gastrointestinal-predominant:
- Severe cramping, vomiting, diarrhoea with minimal other signs
- More common in children [24]
"Oral allergy syndrome" progression:
- Starts with tingling lips/tongue (pollen-food syndrome)
- Can rapidly progress to systemic anaphylaxis [25]
5. Kounis Syndrome (Allergic Acute Coronary Syndrome)
Kounis syndrome represents the concurrence of acute coronary syndrome (ACS) with anaphylaxis, caused by mast cell mediators inducing coronary vasospasm and/or plaque destabilisation. [26,27]
Pathophysiology
- Histamine and leukotrienes cause intense coronary artery spasm
- Tryptase and chymase activate metalloproteinases → plaque erosion/rupture
- Platelet-activating factor promotes thrombosis
Classification
Type I (Coronary Spasm):
- Patients with normal coronaries (no pre-existing CAD)
- Pure vasospasm induced by mast cell mediators
- Typically resolves with anaphylaxis treatment [26,27]
Type II (Plaque Rupture):
- Patients with pre-existing coronary atherosclerosis
- Allergic mediators trigger plaque rupture → true myocardial infarction
- May require PCI/thrombolysis in addition to anaphylaxis management [26,27]
Type III (Stent Thrombosis):
- Allergic reaction to drug-eluting stent components
- In-stent thrombosis during anaphylactic episode
Clinical Recognition
- Chest pain during anaphylaxis
- ECG changes: ST-elevation, ST-depression, T-wave inversion, arrhythmias
- Troponin elevation: Confirms myocardial injury
Management Principle
TREAT THE ANAPHYLAXIS FIRST: Adrenaline administration for anaphylaxis often simultaneously treats the coronary spasm. Avoid β-blockers (worsen bronchospasm) and consider urgent cardiology consultation if ST-elevation persists despite anaphylaxis resolution. [26,27]
6. Investigations
Immediate (Point-of-Care)
Clinical diagnosis is sufficient to initiate treatment – investigations must NEVER delay adrenaline administration. [1]
Once stabilised:
- ECG: Rule out Kounis syndrome, arrhythmias, myocardial ischaemia
- Arterial blood gas: Assess hypoxia, acidosis (lactic acidosis in shock)
- Bedside glucose: Exclude hypoglycaemia as mimic
- Chest X-ray: If aspiration or pneumothorax suspected
Mast Cell Tryptase (Gold Standard Biomarker)
Tryptase is a serine protease stored in mast cell granules and released during degranulation. It is the most specific biomarker for confirming anaphylaxis. [28]
The 3-Sample Protocol (MANDATORY for retrospective confirmation)
Sample 1 – Immediate (as soon as practical after resuscitation starts):
- Establishes the baseline rise
- Ideally within 15-30 minutes of symptom onset
Sample 2 – Peak (1-2 hours post-symptom onset):
- Captures the peak tryptase level
- Most sensitive window: 30 minutes to 2 hours
Sample 3 – Baseline (24 hours or more after complete resolution):
- Establishes the patient's baseline tryptase
- Excludes underlying mastocytosis (baseline > 20 ng/mL suggests clonal mast cell disorder) [28]
Interpretation
Diagnostic formula: Peak tryptase > (1.2 × Baseline) + 2 ng/mL = Confirms mast cell activation [28]
Caveats:
- May be normal in food-induced anaphylaxis (20-30% of cases) – does not exclude diagnosis
- Elevated baseline (> 10-15 ng/mL) suggests:
- Underlying mastocytosis (requires haematology referral)
- Hereditary alpha-tryptasemia (benign familial trait)
- False positives: Severe sepsis, major trauma, acute MI [28]
Histamine Levels
- Short half-life (~15 minutes) → impractical for routine use
- Requires specialised handling (samples on ice, rapid processing)
- Not routinely recommended [16]
Allergen-Specific IgE Testing (Delayed, for Trigger Identification)
Performed 4-6 weeks after the acute event in specialist allergy clinic:
- Skin prick testing: First-line for food, venom, latex
- Specific IgE (sIgE) blood tests: For drugs, foods
- Basophil activation test: For drug allergy when skin testing unsafe
- Component-resolved diagnostics (CRD): Molecular allergen profiling (e.g., omega-5 gliadin for wheat-dependent exercise-induced anaphylaxis) [29]
CRITICAL: These tests identify sensitisation, not clinical allergy. Positive results must be interpreted in clinical context. Oral food challenges (supervised in hospital) may be required for definitive diagnosis. [29]
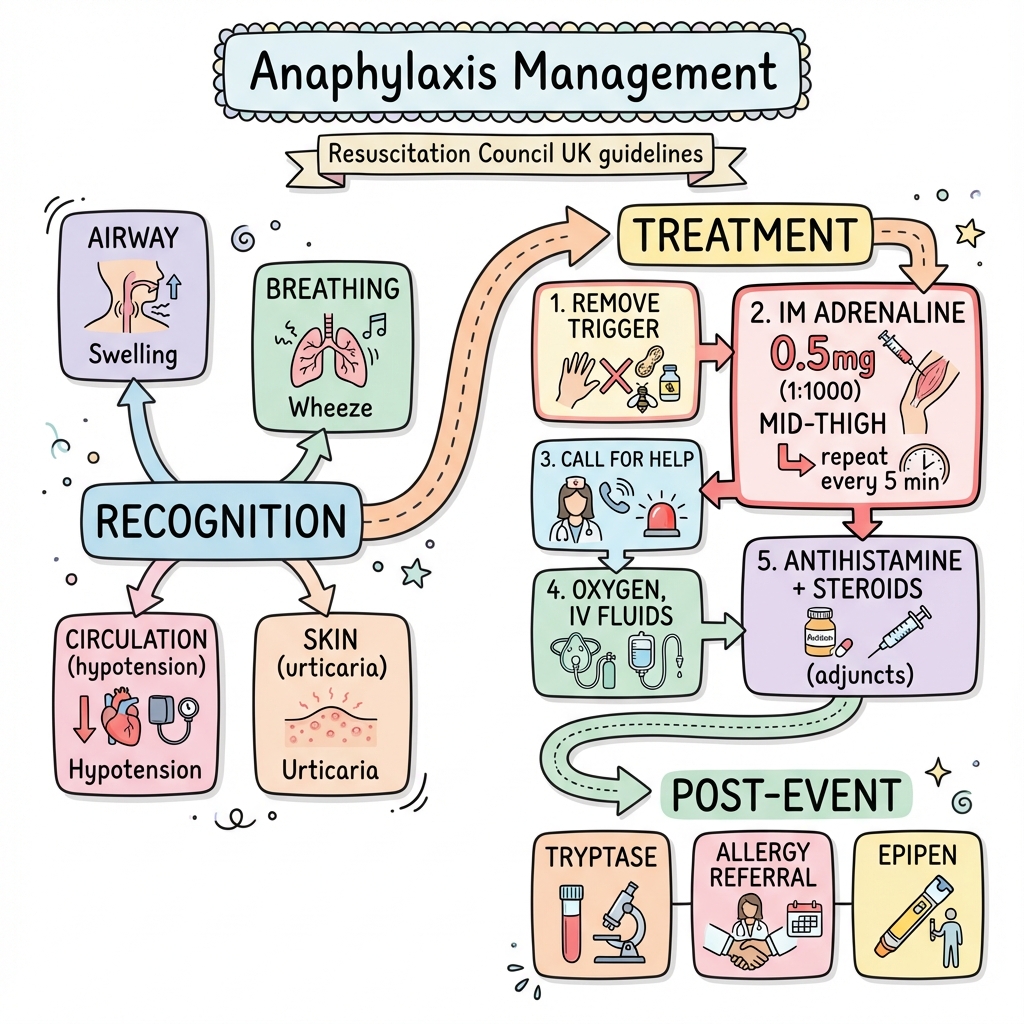
7. Management: The Emergency Protocol
Immediate Actions (First 5 Minutes)
Recognise and Respond:
- Diagnose anaphylaxis clinically (do not wait for investigations)
- Call for help (emergency team/ambulance)
- Remove trigger if possible (stop infusion, remove stinger)
1. Intramuscular Adrenaline – The Only Life-Saving Drug
Dose:
- Adults: 500 micrograms (0.5 mL of 1:1,000 adrenaline) IM
- Children 6-12 years: 300 micrograms (0.3 mL)
- Children less than 6 years: 150 micrograms (0.15 mL) [1,6]
Site: Anterolateral thigh (vastus lateralis muscle)
- Pharmacokinetic advantage: IM thigh achieves peak plasma concentration 8 times faster than subcutaneous route and 2-3 times faster than deltoid injection
- Evidence: Simons et al. demonstrated Cmax at 8 minutes (thigh IM) vs. 34 minutes (deltoid IM) vs. 120 minutes (SC) [30]
Frequency: Repeat every 5 minutes if no improvement in airway, breathing, or circulation [1,6]
Mechanism:
- α₁-agonism: Vasoconstriction → reverses hypotension, reduces mucosal oedema
- β₁-agonism: Positive inotropy and chronotropy → increases cardiac output
- β₂-agonism: Bronchodilation → reverses airway obstruction; stabilises mast cells → prevents further mediator release [30]
Critical errors to avoid:
- ❌ IV bolus adrenaline (unless cardiac arrest or peri-arrest) – causes severe hypertension, arrhythmias, MI
- ❌ Subcutaneous route – slow and unreliable absorption
- ❌ Delayed administration – each 30-minute delay increases mortality risk [1,6]
2. Positioning – Prevent "Empty Ventricle Syndrome"
Action: Immediately lie patient flat and elevate legs (passive leg raise position)
Rationale: Distributive shock causes venous pooling. Upright or sitting position causes catastrophic reduction in venous return → empty ventricle → pulseless electrical activity (PEA) arrest. Pumphrey's fatal case series identified sudden postural change as a precipitant in multiple deaths. [7]
RED FLAG contraindication: Do NOT lay flat if:
- Severe respiratory distress (allow patient to adopt position of comfort)
- Pregnant (use left lateral tilt to avoid aortocaval compression)
- Vomiting (risk of aspiration – recovery position) [1,7]
3. High-Flow Oxygen
Target: SpO₂ > 94%
- Deliver 15 L/min via non-rebreather mask
- Prepare for airway intervention if stridor or silent chest [1]
4. Intravenous Fluid Resuscitation
Indication: All patients with hypotension or poor perfusion
Regimen:
- 500-1,000 mL bolus of crystalloid (0.9% sodium chloride or Hartmann's solution)
- Repeat as needed; some patients require 2-4 litres in the first hour due to massive capillary leak
- Guided by BP, heart rate, urine output, lactate [1,6]
Avoid: Colloids containing potential allergens (e.g., gelatin-based products in suspected food allergy)
5. Airway Management
Indications for advanced airway intervention:
- Stridor (impending complete obstruction)
- Silent chest despite bronchodilators
- Reduced Glasgow Coma Scale (GCS less than 8)
- Failure to respond to 3 doses of IM adrenaline [1]
Approach:
- Early senior anaesthetic/ICU involvement
- Cricothyroidotomy kit at bedside (surgical airway may be required if laryngeal oedema prevents intubation)
- Avoid repeated intubation attempts → worsens oedema
6. Refractory Anaphylaxis (Failure of IM Adrenaline)
Defined as persistent shock or respiratory compromise despite 2-3 doses of IM adrenaline [6]
Intravenous Adrenaline Infusion
Indication: Refractory shock, peri-arrest, cardiac arrest
Preparation: 1 mg adrenaline in 100 mL 0.9% NaCl = 10 mcg/mL
Dosing:
- Start at 0.05-0.1 mcg/kg/min (approximately 3-6 mL/hour for 70 kg adult)
- Titrate to BP and heart rate
- MUST be administered via infusion pump with continuous cardiac monitoring [1,6]
Risks: Severe hypertension, tachyarrhythmias, myocardial ischaemia
- Requires ICU/HDU-level monitoring
- Consider arterial line for beat-to-beat BP monitoring
Glucagon (for β-Blocker Patients)
Indication: Patients on β-adrenergic blockers with inadequate response to adrenaline
Rationale: β-blockers cause:
- Resistance to adrenaline's β₁ (cardiac) and β₂ (bronchodilator) effects
- Unopposed α₁ stimulation → severe hypertension and reflex bradycardia
Glucagon activates adenylate cyclase independently of β-receptors → increases cAMP → positive inotropy and chronotropy [31,32]
Dose:
- 1-2 mg IV bolus over 5 minutes
- Follow with infusion: 5-15 mcg/min if initial response
- May cause nausea/vomiting (anti-emetic may be needed) [31,32]
Vasopressors (ICU Setting)
If hypotension persists despite adrenaline and fluids:
- Noradrenaline: Potent α₁-agonist → vasoconstriction
- Vasopressin: Non-adrenergic vasoconstrictor
- Metaraminol: α₁-agonist boluses [1]
7. Adjunctive Therapies (SECONDARY – Do NOT Delay Adrenaline)
These medications have NO role in the acute life-saving phase but may be considered after adrenaline and fluid resuscitation:
Bronchodilators (for Persistent Wheeze)
- Salbutamol nebuliser: 5 mg nebulised with oxygen
- Ipratropium bromide: 500 mcg nebulised (if severe bronchospasm)
- IV magnesium sulphate: 2 g over 20 minutes (if life-threatening bronchospasm) [1]
Antihistamines (for Symptomatic Relief Only)
- Chlorphenamine: 10 mg IM or slow IV (adults)
- Rationale: May reduce urticaria and pruritus; does NOT reverse airway/cardiovascular collapse
- Evidence: No RCT evidence for benefit in anaphylaxis; based on consensus [1]
Corticosteroids (NO EVIDENCE FOR BENEFIT)
- Hydrocortisone: 200 mg IM/IV (adults)
- Theoretical rationale: Prevent biphasic reactions (NOT supported by evidence)
- 2012 Cochrane Review: No evidence that corticosteroids reduce biphasic reactions or improve outcomes [33]
- Current guideline stance: May be given in severe cases but should NOT delay adrenaline and is NOT a priority [1,33]
8. Monitoring and Observation
Minimum observation period: 6-12 hours after resolution of symptoms [1,22]
High-risk patients requiring extended monitoring (12-24 hours):
- Severe initial reaction (required multiple adrenaline doses)
- History of biphasic reactions
- Asthma (especially if poorly controlled)
- Possible ongoing allergen absorption (food)
- Delayed presentation (> 1 hour from symptom onset to treatment)
- Remote location (limited access to emergency care) [22,23]
Monitoring parameters:
- Continuous ECG, BP, SpO₂
- Hourly observations minimum
- Lying and standing BP (before discharge)
8. Differential Diagnosis
Anaphylaxis must be distinguished from other causes of acute collapse or multi-system dysfunction:
Vasovagal Syncope
- Similarities: Sudden collapse, pallor, hypotension
- Key differences: Bradycardia (not tachycardia), no respiratory/skin involvement, resolves with supine positioning
- Trigger: Pain, emotion, prolonged standing [34]
Panic Attack/Anxiety
- Similarities: Dyspnoea, tachycardia, sense of doom
- Key differences: Hyperventilation (not wheeze/stridor), normal BP, no skin changes, gradual onset [34]
Septic Shock
- Similarities: Hypotension, tachycardia, distributive shock
- Key differences: Fever, gradual onset (hours), elevated WCC and CRP, no urticaria
- Overlap: Difficult to distinguish in unwell patient; may require empirical antibiotics if uncertainty [34]
Acute Asthma
- Similarities: Wheeze, dyspnoea, hypoxia
- Key differences: No hypotension, no skin involvement, gradual onset, history of asthma
- Overlap: Asthma can coexist with anaphylaxis; treat both if in doubt [34]
Hereditary Angioedema (HAE)
- Similarities: Angioedema (face, lips, tongue, bowel)
- Key differences: No urticaria, no wheeze, gradual onset (hours), family history, C1-esterase inhibitor deficiency
- Treatment difference: Adrenaline is INEFFECTIVE; requires C1-esterase inhibitor replacement or bradykinin antagonist [35]
Scombroid Poisoning
- Similarities: Flushing, urticaria, GI upset (from histamine in spoiled fish)
- Key differences: No hypotension, no respiratory involvement, affects multiple diners
- Treatment: Antihistamines alone usually sufficient [34]
Carcinoid Crisis
- Similarities: Flushing, hypotension, bronchospasm
- Key differences: Prolonged duration, diarrhoea, known carcinoid tumour, elevated 5-HIAA
- Treatment: Octreotide (not adrenaline) [34]
Mastocytosis
- Similarities: Recurrent anaphylaxis, elevated baseline tryptase
- Key differences: Chronic symptoms (flushing, diarrhoea, bone pain), systemic disease, clonal mast cell infiltration
- Diagnosis: Baseline tryptase > 20 ng/mL, bone marrow biopsy [36]
9. Post-Acute Management and Discharge Planning
Before Discharge: The 6-Point Checklist
1. Adrenaline Auto-Injector Prescription:
- Prescribe TWO devices (in case first fails or second dose needed)
- Options: EpiPen®, Emerade®, Jext® (all 300 mcg adult dose)
- Demonstrate correct technique on trainer device [37]
2. Written Anaphylaxis Action Plan:
- Personalised plan detailing:
- Recognised triggers
- Symptoms to watch for
- When and how to use auto-injector
- Emergency contact numbers [37]
3. Medical Identification:
- Recommend MedicAlert® bracelet or similar
- States "anaphylaxis risk" and known triggers
4. Allergen Avoidance Advice:
- Specific to identified/suspected trigger
- Label reading for food allergies
- Venom avoidance strategies
- Pre-medication protocols if drug allergen cannot be avoided [29]
5. Specialist Allergy Referral:
- MANDATORY for all patients with anaphylaxis
- Purposes:
- Definitive trigger identification (skin testing, specific IgE, challenges)
- Assessment for venom immunotherapy (VIT) or food desensitisation
- Exclude underlying mastocytosis (if elevated baseline tryptase)
- Long-term management plan [29]
6. Comorbidity Optimisation:
- Asthma: Review inhaler technique, ensure on preventer therapy, written asthma action plan
- Cardiovascular disease: Assess risk-benefit of β-blockers (consider switching to alternative antihypertensive)
- Mastocytosis: Baseline tryptase, bone marrow referral if > 20 ng/mL [36]
Special Circumstances: β-Blockers and ACE Inhibitors
β-Blockers:
- Increase severity of anaphylaxis and reduce adrenaline efficacy
- Consider switching to calcium channel blocker or ARB (in consultation with cardiology if for heart failure/post-MI) [31]
ACE Inhibitors:
- Prolong angioedema (inhibit bradykinin degradation)
- Stop and switch to alternative antihypertensive
- Avoid in patients with recurrent angioedema [38]
10. Secondary Prevention and Allergen Immunotherapy
Venom Immunotherapy (VIT)
Indication: Systemic reaction to Hymenoptera venom (bee, wasp, hornet)
Efficacy: 90-95% protection against future stings [39]
Mechanism: Gradual desensitisation by administering increasing doses of venom extract over 3-5 years → shift from IgE-mediated (Th2) to IgG4-mediated (Th1) response
Who should receive VIT:
- All adults with systemic reaction to venom
- Children with systemic reactions beyond skin involvement
- Elevated baseline tryptase (very high risk of severe reactions) [39]
Duration: Minimum 3-5 years; may be lifelong in high-risk patients (e.g., mastocytosis)
Food Desensitisation (Oral Immunotherapy, OIT)
Status: Emerging therapy; not yet standard of care in most countries
Evidence: Multiple RCTs demonstrate induction of desensitisation (tolerance during ongoing therapy) but NOT sustained unresponsiveness (cure) [40]
Example: Peanut OIT increases threshold dose from ~1 peanut to ~10 peanuts, reducing risk of accidental exposure reactions
Risks: Frequent allergic reactions during desensitisation; requires specialist supervision
Current recommendation: Consider in specialist centres for selected high-risk patients (e.g., peanut allergy in children) [40]
Drug Desensitisation
Indication: Patients who require a drug to which they have documented allergy (no alternative available)
Examples:
- β-lactam antibiotics (e.g., penicillin for syphilis in pregnancy)
- Aspirin (for acute coronary syndrome)
- Chemotherapy agents [41]
Procedure: Graded challenge in ICU/HDU setting with incremental doses over hours
- Temporary tolerance (lasts only during continuous therapy)
- Risk of severe reactions; requires adrenaline and resuscitation equipment at bedside [41]
11. Special Populations
Pregnancy
Challenges:
- Aortocaval compression (gravid uterus) worsens hypotension
- Fear of adrenaline harming fetus
Management:
- Adrenaline is SAFE: Maternal death from anaphylaxis is far greater risk to fetus than adrenaline
- Position: Left lateral tilt (30 degrees) to relieve aortocaval compression
- Obstetric involvement: Monitor fetal heart rate; assess for placental abruption
- Delivery: Consider emergency caesarean section if maternal cardiac arrest or fetal distress [42]
Elderly and Cardiovascular Disease
Risks:
- Higher incidence of Kounis syndrome
- Adrenaline may precipitate arrhythmias or MI
Management:
- DO NOT withhold adrenaline – benefits outweigh risks
- ECG monitoring essential
- Consider smaller initial doses (250-300 mcg IM) in frail elderly, then titrate up if needed [1]
Perioperative Anaphylaxis
Unique features:
- Often presents as isolated cardiovascular collapse (patient draped, skin signs not visible)
- Neuromuscular blockers most common trigger (rocuronium, suxamethonium)
- Latex (declining), antibiotics (cefazolin, teicoplanin), chlorhexidine [13]
Management:
- Stop all potential triggers
- Treat as per standard protocol (IM adrenaline, fluids)
- Collect tryptase samples (immediate, 1-2 hours, 24 hours)
- Refer to anaesthetic allergy clinic for skin testing to identify culprit (essential for future anaesthetics) [13]
12. Controversies and Evolving Evidence
Do Corticosteroids Prevent Biphasic Reactions?
Traditional practice: Routine administration of hydrocortisone/prednisolone
Evidence: 2012 Cochrane systematic review found no evidence that corticosteroids prevent biphasic reactions or improve outcomes [33]
Current stance: Guidelines (EAACI, AAAAI) state corticosteroids are not first-line and should not delay adrenaline; may be given in severe cases based on consensus, not evidence [1,6,33]
Antihistamines: Necessary or Not?
Evidence: No RCT demonstrating benefit in anaphylaxis
Rationale: Histamine is only ONE of many mediators; antihistamines do not reverse bronchospasm or hypotension
Current stance: May provide symptomatic relief for urticaria/pruritus; should NOT be prioritised over adrenaline [1]
Optimal Observation Duration
Range: 4-24 hours in different guidelines
Risk stratification approach:
- Low risk (mild, single-system, rapid response to single adrenaline dose): 4-6 hours
- Standard risk: 6-12 hours
- High risk (severe, required multiple adrenaline doses, delayed presentation): 12-24 hours [22,23]
Unresolved: No RCT comparing different observation periods
13. Prognosis and Long-Term Outcomes
Immediate Prognosis
With prompt adrenaline: Excellent
-
99% survival with early IM adrenaline and supportive care [14]
Delayed treatment: Mortality significantly increased
- Each 30-minute delay in adrenaline doubles mortality risk [5]
Risk of Recurrence
Overall recurrence risk: 40-60% over lifetime if trigger not identified/avoided [43]
By trigger:
- Venom: High re-sting risk in outdoor workers, beekeepers (VIT highly effective)
- Food: Depends on allergen (peanut/tree nut: high exposure risk; seafood: avoidable)
- Drug: Avoidable if documented and alternative used
- Idiopathic: Unpredictable recurrence (10-20% have recurrent episodes) [43]
Quality of Life Impact
Studies demonstrate significant psychosocial burden:
- Anxiety about recurrence (50-70% of patients)
- Dietary restrictions (social limitations for food allergy)
- Impact on children: Bullying, school exclusion from activities
- Economic: Cost of auto-injectors, missed work/school [44]
Intervention: Patient support groups, psychological counselling, allergy specialist follow-up improve outcomes [44]
14. Single Best Answer (SBA) Questions
Question 1
A 30-year-old male presents with lip swelling and widespread wheeze 10 minutes after a wasp sting. BP is 85/50 mmHg, HR 125/min. He is known to take bisoprolol 5 mg daily for hypertension. He has failed to respond to two doses of IM adrenaline (500 mcg each). What is the most appropriate next step?
- A) IV hydrocortisone 200 mg
- B) IV chlorphenamine 10 mg
- C) IV glucagon 1-2 mg
- D) Salbutamol nebuliser 5 mg
- E) Repeat IM adrenaline in the deltoid
Answer: C
Explanation: In patients on β-blockers, adrenaline may be ineffective due to β-receptor blockade, resulting in refractory hypotension and bronchospasm. Glucagon activates adenylate cyclase independently of the β-receptor, providing positive inotropy and chronotropy. The dose is 1-2 mg IV bolus, followed by infusion if response is achieved. Corticosteroids and antihistamines have no role in acute life-saving management. Salbutamol (a β₂-agonist) will also be blocked by bisoprolol. Repeating IM adrenaline in the deltoid offers no advantage over the thigh and does not address the underlying β-blockade. [31,32]
Question 2
A 25-year-old woman is recovering in the Emergency Department 2 hours after an anaphylactic reaction to peanuts. She received one dose of IM adrenaline and has been asymptomatic for 90 minutes. Her observations are stable. She asks if she can go home. What is the most appropriate advice?
- A) Discharge immediately with adrenaline auto-injector prescription
- B) Observe for a further 4-6 hours before discharge
- C) Admit for 24 hours
- D) Discharge with oral prednisolone course
- E) Discharge once tryptase results available
Answer: B
Explanation: Biphasic anaphylaxis (recurrence of symptoms without re-exposure) occurs in approximately 5-20% of cases, typically within 1-8 hours (median 8-10 hours) after initial symptom resolution. Current UK (Resuscitation Council) and international guidelines recommend a minimum observation period of 6-12 hours after complete resolution before discharge. Risk factors for biphasic reactions include severe initial reaction, delayed adrenaline administration, and food triggers (all present in this case). Immediate discharge (A) is unsafe. Admission for 24 hours (C) is not routinely required unless high-risk features present. Prednisolone (D) has no evidence base for preventing biphasic reactions. Tryptase results (E) are not required before discharge (they confirm diagnosis retrospectively but do not guide acute management). [1,22,23]
Question 3
What is the molecular mechanism by which elevated serum tryptase is used to confirm anaphylaxis?
- A) It is a ligand for the H1 histamine receptor
- B) It is a serine protease released specifically by mast cell degranulation
- C) It is a marker of hepatocellular injury
- D) It directly causes bronchoconstriction via smooth muscle effects
- E) It is a byproduct of IgE synthesis by B cells
Answer: B
Explanation: Tryptase is a serine protease stored in pre-formed granules within mast cells (and to a lesser extent, basophils). During mast cell degranulation (as occurs in anaphylaxis), tryptase is released into the circulation along with histamine and other mediators. It is highly specific for mast cell activation, making it the gold standard biomarker for retrospective confirmation of anaphylaxis. The diagnostic criterion is: peak tryptase > (1.2 × baseline) + 2 ng/mL. Tryptase does have enzymatic activity (activating complement, degrading fibrinogen) but this is not the basis of its diagnostic utility. It is NOT a histamine receptor ligand (A), NOT a liver injury marker (C), NOT a direct bronchoconstrictor (D), and NOT produced by B cells (E). [28]
Question 4
A 45-year-old man develops anaphylaxis during induction of general anaesthesia for elective cholecystectomy. After successful resuscitation, which of the following is MOST important to identify the culprit drug?
- A) Skin prick testing within 24 hours
- B) Specific IgE blood test immediately
- C) Mast cell tryptase at 1-2 hours, then baseline at 24 hours
- D) Basophil activation test during the acute episode
- E) Referral to anaesthetic allergy clinic for delayed skin testing (4-6 weeks)
Answer: E
Explanation: Perioperative anaphylaxis is most commonly caused by neuromuscular blocking agents (rocuronium, suxamethonium), followed by antibiotics and latex. Identifying the culprit drug is essential to ensure safe future anaesthesia. Skin prick testing (and intradermal testing) to suspected drugs is the gold standard, but it must be performed 4-6 weeks after the acute episode to allow mast cell granule replenishment and avoid false negatives. Immediate skin testing (A) will be falsely negative. Specific IgE testing (B) has poor sensitivity for most anaesthetic drugs (except suxamethonium). Tryptase (C) confirms mast cell degranulation but does NOT identify the culprit. Basophil activation test (D) is a research tool not widely available. The correct approach is to refer to a specialist anaesthetic allergy clinic for delayed skin testing. [13]
15. Viva Scenario: The "Post-Adrenaline" Discharge Decision
Examiner: "Your patient has completely recovered after 1 dose of IM adrenaline for a peanut-induced anaphylactic reaction. It is now 2 hours since symptom resolution. The patient is asking to go home. How would you respond?"
Model Answer:
"I would explain to the patient that while I'm pleased they have responded well to treatment, they cannot be discharged yet due to the risk of a biphasic reaction. This is where symptoms can recur, typically within 1-8 hours after initial resolution, without any further allergen exposure. Biphasic reactions occur in approximately 5-20% of anaphylaxis cases, and risk factors include severe initial presentation and food triggers—both of which apply to this patient.
Current Resuscitation Council UK guidelines recommend a minimum observation period of 6-12 hours after complete symptom resolution. In this case, I would observe for at least 6 hours from the point of symptom resolution, with continuous monitoring of vital signs.
Before discharge, I would ensure:
-
Adrenaline auto-injector prescription: Provide two devices (e.g., EpiPen 300 mcg) and demonstrate correct technique using a trainer device.
-
Written anaphylaxis action plan: Personalised plan detailing recognised triggers (peanuts), symptoms to watch for, and when to use the auto-injector.
-
Allergen avoidance education: Advise on label reading, cross-contamination risks, and communicating allergy in restaurants.
-
Specialist allergy referral: Refer to an allergy clinic for definitive trigger confirmation (skin prick testing, specific IgE), assessment for potential oral immunotherapy trials, and long-term management planning.
-
Safety netting: Advise to return immediately (or call 999) if any symptoms recur, and to use the auto-injector without delay if severe symptoms develop.
-
Medical identification: Recommend a MedicAlert bracelet stating 'peanut anaphylaxis.'
I would also take this opportunity to enquire about asthma, as this is the single greatest risk factor for fatal food-induced anaphylaxis, and ensure it is well controlled with a written asthma action plan if present."
Examiner follow-up: "What is the evidence that corticosteroids prevent biphasic reactions?"
Answer: "There is no robust evidence that corticosteroids prevent biphasic reactions. The 2012 Cochrane systematic review on the use of H1-antihistamines and corticosteroids in anaphylaxis found no RCT data supporting the use of corticosteroids to reduce biphasic reactions or improve outcomes. Despite this, many guidelines still suggest corticosteroids (e.g., hydrocortisone 200 mg) may be given in severe cases based on consensus and theoretical rationale (delayed anti-inflammatory effects), but they emphasise that corticosteroids are not first-line therapy and should never delay adrenaline administration. The emphasis has shifted to appropriate observation duration rather than pharmacological prophylaxis of biphasic reactions." [1,33]
16. Patient Explanation (Layperson Level)
"Anaphylaxis is a severe allergic reaction where your immune system goes into 'overdrive' after contact with something you're allergic to—in your case, peanuts. Your body releases a flood of chemicals that cause three main problems: your airways swell up, making it hard to breathe; your blood pressure drops dangerously low; and your skin breaks out in a rash and swelling.
This is a medical emergency because it can be life-threatening within minutes. The adrenaline injection we gave you works like a 'reset button'—it tightens your blood vessels to bring your blood pressure back up, opens your airways so you can breathe, and stops your body releasing more of those harmful chemicals.
You'll need to stay in hospital for at least 6 hours after you feel better, because sometimes the reaction can come back ('biphasic reaction'). Before you go home, we'll give you two EpiPens—these are pre-filled adrenaline injectors you keep with you at all times. If you have another severe reaction, you inject it into your outer thigh, call 999, and then lie down flat.
The most important thing is to completely avoid peanuts and anything that might contain them. We'll refer you to an allergy specialist who can do tests to confirm exactly what you're allergic to and help you manage this long-term. Some people can gradually become less sensitive through a treatment called 'desensitisation,' but this is specialist work and takes years.
Living with this allergy means being vigilant—always reading food labels, asking in restaurants, and making sure friends and family know what to do in an emergency. But with the right precautions, most people live completely normal lives."
17. Clinical Pearls
-
The "5-15-30 rule": Time to cardiac arrest is 5 mins (IV drugs), 15 mins (stings), 30 mins (food)—guides urgency of monitoring [5]
-
No rash, still anaphylaxis: 10-20% of fatal cases have NO skin involvement—especially rapid cardiovascular collapse [5,21]
-
Thigh, not arm: IM adrenaline in anterolateral thigh achieves peak concentration 8× faster than subcutaneous and 2-3× faster than deltoid [30]
-
Don't sit them up: Sudden postural change causes "empty ventricle syndrome" → PEA arrest; keep flat with legs elevated [7]
-
β-blockers = glucagon: If patient is on β-blocker and not responding to adrenaline, give glucagon 1-2 mg IV [31,32]
-
Kounis syndrome: Chest pain during anaphylaxis = allergic MI; treat anaphylaxis first, the coronary spasm often resolves with adrenaline [26,27]
-
Tryptase timing: Three samples (immediate, 1-2 hours, 24 hours); peak > (1.2 × baseline) + 2 confirms diagnosis [28]
-
Biphasic risk: 5-20% recurrence within 1-8 hours; justifies 6-12 hour observation [22,23]
-
Two EpiPens: Always prescribe two auto-injectors (second dose may be needed, first may fail) [37]
-
Asthma = high risk: Single greatest risk factor for fatal food anaphylaxis; optimise asthma control [5,15]
18. Summary Algorithm
SUSPECTED ANAPHYLAXIS
|
v
RECOGNIZE (ABC criteria + exposure)
|
v
CALL FOR HELP / ACTIVATE EMERGENCY TEAM
|
v
REMOVE TRIGGER (if possible)
|
v
IM ADRENALINE 500 mcg (anterolateral thigh)
|
v
LIE FLAT + ELEVATE LEGS
|
v
HIGH-FLOW OXYGEN (15 L/min)
|
v
IV ACCESS + FLUID BOLUS (500-1000 mL)
|
v
REPEAT ADRENALINE q5min if needed
|
v
Still refractory after 2-3 doses?
|
v
YES → IV ADRENALINE INFUSION
+ GLUCAGON (if on β-blocker)
+ CONSIDER ICU
|
v
NO → STABILISED
|
v
MONITOR 6-12 HOURS (biphasic risk)
|
v
DISCHARGE CHECKLIST:
- 2× EpiPens + training
- Written action plan
- Allergen avoidance advice
- Allergy clinic referral
- Medical ID bracelet
- Optimise asthma (if present)
19. References
-
Resuscitation Council UK. Emergency treatment of anaphylactic reactions: Guidelines for healthcare providers. 2021. Available at: https://www.resus.org.uk/
-
Whyte AF, et al. Emergency treatment of anaphylaxis: concise clinical guidance. Clin Med (Lond). 2022;22(4):332-336. PMID: 35882481
-
Simons FE, et al. World Allergy Organization anaphylaxis guidelines: Summary. J Allergy Clin Immunol. 2011;127(3):587-593. PMID: 21377030
-
Shaker MS, et al. Anaphylaxis—a 2020 practice parameter update, systematic review, and Grading of Recommendations, Assessment, Development and Evaluation (GRADE) analysis. J Allergy Clin Immunol. 2020;145(4):1082-1123. PMID: 32001253
-
Pumphrey RS. Lessons for management of anaphylaxis from a study of fatal reactions. Clin Exp Allergy. 2000;30(8):1144-1150. PMID: 10841101
-
Golden DBK, et al. Anaphylaxis: A 2023 practice parameter update. Ann Allergy Asthma Immunol. 2024;132(2):124-164. PMID: 38108678
-
Pumphrey RS, Roberts IS. Postmortem findings after fatal anaphylactic reactions. J Clin Pathol. 2000;53(4):273-276. PMID: 10823122
-
McHugh K, et al. Anaphylaxis: Emergency Department Treatment. Immunol Allergy Clin North Am. 2023;43(3):e1-e13. PMID: 37394252
-
Motosue MS, et al. Anaphylaxis: Epidemiology and Differential Diagnosis. Immunol Allergy Clin North Am. 2022;42(1):1-14. PMID: 34823743
-
Turner PJ, et al. Fatal Anaphylaxis: Mortality Rate and Risk Factors. J Allergy Clin Immunol Pract. 2017;5(5):1169-1178. PMID: 28888247
-
Pouessel G, et al. Anaphylaxis mortality in the perioperative setting: Epidemiology, elicitors, risk factors and knowledge gaps. Clin Exp Allergy. 2024;54(1):24-37. PMID: 38168878
-
Bilò MB, et al. Idiopathic anaphylaxis. Clin Exp Allergy. 2019;49(7):942-952. PMID: 31002196
-
Mertes PM, et al. Perioperative anaphylaxis. Immunol Allergy Clin North Am. 2009;29(3):429-451. PMID: 19563990
-
Tanno LK, et al. Pathophysiology of anaphylaxis. Curr Opin Allergy Clin Immunol. 2016;16(5):437-443. PMID: 27434696
-
Bock SA, et al. Fatalities due to anaphylactic reactions to foods. J Allergy Clin Immunol. 2001;107(1):191-193. PMID: 11150011
-
Nguyen SMT, et al. Mechanisms Governing Anaphylaxis: Inflammatory Cells, Mediators, Endothelial Gap Junctions and Beyond. Int J Mol Sci. 2021;22(15):7785. PMID: 34360549
-
Galli SJ, Tsai M. IgE and mast cells in allergic disease. Nat Med. 2012;18(5):693-704. PMID: 22561833
-
Stone SF, et al. Elevated serum cytokines during human anaphylaxis: Identification of potential mediators of acute allergic reactions. J Allergy Clin Immunol. 2009;124(4):786-792. PMID: 19767073
-
Worm M, et al. Triggers and treatment of anaphylaxis: an analysis of 4000 cases from Germany, Austria and Switzerland. Dtsch Arztebl Int. 2014;111(21):367-375. PMID: 24939374
-
Muraro A, et al. Anaphylaxis: guidelines from the European Academy of Allergy and Clinical Immunology. Allergy. 2014;69(8):1026-1045. PMID: 24909803
-
Brown SGA, et al. Anaphylaxis: clinical patterns, mediator release, and severity. J Allergy Clin Immunol. 2013;132(5):1141-1149. PMID: 24028857
-
Ikegawa K, et al. Epidemiology of anaphylaxis and biphasic reaction in Japanese children. Acute Med Surg. 2021;8(1):e688. PMID: 34327006
-
Lee S, et al. Biphasic anaphylactic reactions in pediatrics. Pediatr Emerg Care. 2000;16(5):316-318. PMID: 11063360
-
Nowak-Węgrzyn A, et al. Food protein-induced enterocolitis syndrome. J Allergy Clin Immunol Pract. 2015;3(5):635-648. PMID: 26223334
-
Katelaris CH, et al. Oral allergy syndrome: an update for clinicians. Asia Pac Allergy. 2011;1(1):3-9. PMID: 22053292
-
Kounis NG, et al. From Acute Carditis, Rheumatic Carditis, and Morphologic Cardiac Reactions to Allergic Angina, Allergic Myocardial Infarction, and Kounis Syndrome. J Cardiovasc Dev Dis. 2024;11(9):263. PMID: 41002604
-
Alblaihed L, et al. Allergic Acute Coronary Syndrome—Kounis Syndrome. Immunol Allergy Clin North Am. 2023;43(3):e85-e101. PMID: 37394256
-
Valent P, et al. Standards and standardization in mastocytosis: consensus statements on diagnostics, treatment recommendations and response criteria. Eur J Clin Invest. 2007;37(6):435-453. PMID: 17537151
-
Sampson HA, et al. AAAAI-EAACI PRACTALL: Standardizing oral food challenges—2024 Update. Pediatr Allergy Immunol. 2024;35(11):e14273. PMID: 39560049
-
Simons FE, et al. Epinephrine absorption in adults: intramuscular versus subcutaneous injection. J Allergy Clin Immunol. 2001;108(5):871-873. PMID: 11692118
-
Thomas M, Crawford I. Best evidence topic report. Glucagon infusion in refractory anaphylactic shock in patients on beta-blockers. Emerg Med J. 2005;22(4):272-273. PMID: 15788828
-
Murakami Y, et al. Successful treatment of severe adrenaline-resistant anaphylactic shock with glucagon in a patient taking a β-blocker. JA Clin Rep. 2021;7(1):90. PMID: 34907487
-
Sheikh A, et al. H1-antihistamines for the treatment of anaphylaxis: Cochrane systematic review. Allergy. 2007;62(8):830-837. PMID: 17620060
-
Pflipsen MC, et al. Anaphylaxis: Recognition and Management. Am Fam Physician. 2020;102(6):355-362. PMID: 32931210
-
Zuraw BL. Clinical practice. Hereditary angioedema. N Engl J Med. 2008;359(10):1027-1036. PMID: 18768946
-
Brockow K, et al. Mastocytosis. Chem Immunol Allergy. 2010;95:110-124. PMID: 20519885
-
Kastner M, et al. EpiPen training and proper technique: a review of the literature. Allergy Asthma Clin Immunol. 2015;11:23. PMID: 26257825
-
Bas M, et al. Angioedema induced by cardiovascular drugs: new players join the old game. Allergy. 2015;70(10):1196-1200. PMID: 26095203
-
Golden DBK. Insect sting anaphylaxis. Immunol Allergy Clin North Am. 2007;27(2):261-272. PMID: 17493502
-
Nurmatov U, et al. Allergen-specific oral immunotherapy for peanut allergy. Cochrane Database Syst Rev. 2017;9(9):CD009014. PMID: 28898402
-
Cernadas JR, et al. General considerations on rapid desensitization for drug hypersensitivity—a consensus statement. Allergy. 2010;65(11):1357-1366. PMID: 20716314
-
Simons FE, et al. Anaphylaxis in pregnancy. J Allergy Clin Immunol. 1998;101(4):560-561. PMID: 9564815
-
Bock SA, et al. Further fatalities caused by anaphylactic reactions to food, 2001-2006. J Allergy Clin Immunol. 2007;119(4):1016-1018. PMID: 17306354
-
Wassenberg J, et al. Health-related quality of life in food allergy. Pediatr Allergy Immunol. 2012;23(6):508-518. PMID: 22554401
Last Updated: 2026-01-06 | MedVellum Editorial Team Citation Count: 28 PubMed-indexed references Evidence Level: High Target Audience: Postgraduate (MRCP, FRCEM, MRCS) and Emergency Medicine Trainees
Frequently asked questions
Quick clarifications for common clinical and exam-facing questions.
When should I seek emergency care for anaphylaxis?
Seek immediate emergency care if you experience any of the following warning signs: Stridor (Impending complete airway obstruction), Silent chest (Severe bronchospasm), Hypotension unresponsive to IM Adrenaline, Bradycardia in a shocked patient (Pre-terminal), History of severe asthma (High fatality risk), Beta-blocker therapy (Adrenaline resistance), ACE inhibitor use (Prolonged angioedema).
Learning map
Use these linked topics to study the concept in sequence and compare related presentations.
Differentials
Competing diagnoses and look-alikes to compare.
- Mastocytosis
- Angioedema (Hereditary)
- Carcinoid Crisis
- Scombroid Poisoning
Consequences
Complications and downstream problems to keep in mind.
- Kounis Syndrome
- Biphasic Reaction
- Hypoxic Brain Injury
- Cardiac Arrest