ANCA-Associated Vasculitis
1.1 Summary ANCA-associated vasculitis (AAV) represents a group of rare, heterogeneous, and life-threatening multi-system autoimmune disorders characterized by necrotizing inflammation of small-to-medium-sized blood...
What matters first
1.1 Summary ANCA-associated vasculitis (AAV) represents a group of rare, heterogeneous, and life-threatening multi-system autoimmune disorders characterized by necrotizing inflammation of small-to-medium-sized blood...
Rapidly progressive glomerulonephritis (RPGN)
4 Jan 2026
Generated educational material; verify before clinical use.
Visible references section
See the concept before reading it
Study the key anatomy, imaging, and decision pathways as full teaching plates.
Clinical board
A visual summary of the highest-yield teaching signals on this page.
Urgent signals
Safety-critical features pulled from the topic metadata.
- Rapidly progressive glomerulonephritis (RPGN)
- Diffuse alveolar haemorrhage (DAH)
- Scleritis or orbital pseudotumor
- Mononeuritis multiplex
Linked comparisons
Differentials and adjacent topics worth opening next.
- Goodpasture's Disease (Anti-GBM)
- Systemic Lupus Erythematosus (SLE)
Content status and exam context
This page is AI-generated educational content. It may contain errors or omissions and is not a substitute for current guidelines, local protocols, senior clinical judgement, or professional medical advice.
MedVellum does not claim an individual clinician reviewer, board certification, or professional credential for this page unless a future version names a real, verifiable reviewer.
Clinical explanation and evidence
ANCA-Associated Vasculitis
SECTION 1: Clinical Overview
1.1 Summary
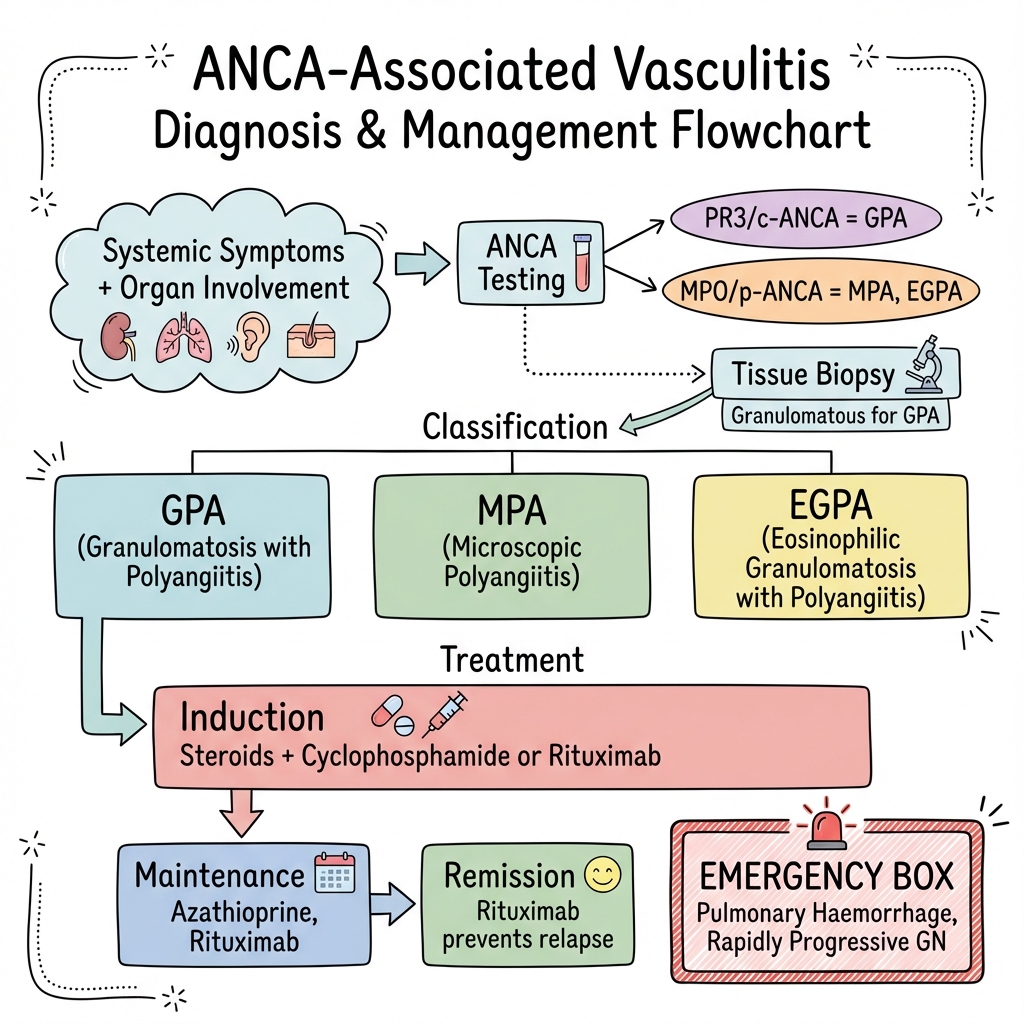
ANCA-associated vasculitis (AAV) represents a group of rare, heterogeneous, and life-threatening multi-system autoimmune disorders characterized by necrotizing inflammation of small-to-medium-sized blood vessels, primarily targeting capillaries, venules, and arterioles. The disease spectrum encompasses three distinct clinicopathological entities as defined by the 2012 Revised Chapel Hill Consensus Conference: Granulomatosis with Polyangiitis (GPA, formerly Wegener's), Microscopic Polyangiitis (MPA), and Eosinophilic Granulomatosis with Polyangiitis (EGPA, formerly Churg-Strauss). These conditions are unified by their strong association with antineutrophil cytoplasmic antibodies (ANCA), specifically those targeting proteinase 3 (PR3) or myeloperoxidase (MPO), although a small percentage of patients remain seronegative. Historically, AAV was nearly universally fatal within one year of diagnosis, typically due to rapid renal or respiratory failure. However, the introduction of cyclophosphamide and high-dose glucocorticoids in the 1970s, followed by the adoption of modern biological agents like rituximab and targeted complement inhibitors like avacopan, has transformed AAV into a chronic, manageable, yet frequently relapsing condition requiring life-long vigilance. [1, 2, 8]
The clinical hallmark of AAV is its potential for catastrophic, "silent" organ failure, most notably pauci-immune necrotizing crescentic glomerulonephritis and diffuse alveolar haemorrhage (DAH). GPA is further distinguished by its predilection for the respiratory tract, featuring granulomatous inflammation that can cause destructive lesions in the sinuses, orbits, and subglottis. The peak incidence occurs in the 6th and 7th decades of life, with a slight male predominance in GPA and MPA cohorts. Management has undergone a major paradigm shift from toxic, long-term cyclophosphamide regimens to targeted B-cell depletion with rituximab and "steroid-sparing" protocols using the C5a receptor antagonist avacopan. Despite these significant advances, AAV remains one of the most challenging diagnoses in internal medicine due to its protean, non-specific early manifestations, requiring a high index of clinical suspicion and a multi-disciplinary approach involving rheumatologists, nephrologists, and pulmonologists to prevent irreversible end-organ damage. [3, 4, 11]
1.2 Key Facts
- Definition: A group of small-vessel vasculitides characterized by necrotizing inflammation and the presence of ANCA directed against neutrophil granular enzymes. [1]
- Incidence: Approximately 20–25 per million persons per year in Europe and North America; incidence is rising due to better detection and an aging population. [2]
- Prevalence: Estimated at 300–400 per million persons, reflecting improved survival and the transition of AAV into a chronic relapsing disease. [3]
- Mortality: Untreated 1-year mortality > 80%; with modern treatment, 5-year survival is now > 80%, though death from infection remains a major risk. [4]
- Morbidity: Chronic kidney disease (CKD) occurs in up to 40% of patients; 25% may progress to end-stage renal disease (ESRD) within 5 years. [5]
- Peak Age: 60–75 years; however, AAV can affect any age group, including children, where it often presents with severe renal involvement.
- Sex Distribution: GPA and MPA show a slight male predominance (1.2:1); EGPA is more balanced or slightly female-predominant (1:1.1).
- Pathognomonic Feature: Pauci-immune necrotizing crescentic glomerulonephritis on renal biopsy, defined by the lack of immune complex deposition. [6]
- Gold Standard Investigation: High-sensitivity ELISA for PR3-ANCA and MPO-ANCA combined with tissue biopsy (kidney, lung, or skin). [7]
- First-line Induction: Glucocorticoids combined with either Rituximab (preferred for relapses/GPA) or Cyclophosphamide (often used for severe renal disease). [8]
- Maintenance Choice: Rituximab (preferred) or Azathioprine to prevent relapse; maintenance is typically continued for 24–48 months depending on risk. [9]
- Key Trial (Induction): The RAVE trial established the non-inferiority of Rituximab to Cyclophosphamide for inducing remission in severe AAV. [10]
- Key Trial (Complement): The ADVOCATE trial demonstrated that Avacopan is non-inferior to a prednisone taper and superior at 52 weeks. [11]
- Relapse Risk: Significantly higher in PR3-ANCA positive patients (up to 50% at 5 years) compared to MPO-ANCA patients (approx. 15-20%). [35]
- ENT Involvement: Present in ~90% of GPA cases, often as chronic, bloody crusting or "unresolved" sinusitis that fails antibiotic therapy.
- Emergency Flag: Haemoptysis in an ANCA+ patient indicates diffuse alveolar haemorrhage (DAH), requiring immediate ICU transfer. [15]
- Histology Buzzword: "Pauci-immune" refers to the absence of significant IgG or C3 deposition on immunofluorescence, pointing to a neutrophil-driven process.
- ANCA Negative AAV: Occurs in ~10% of cases, particularly in EGPA or limited GPA; diagnosis relies on typical clinical features and biopsy. [12]
- Vasa Nervorum Damage: Leads to mononeuritis multiplex, a classic feature where peripheral nerves are affected in an asymmetric, "step-wise" fashion.
- Subglottic Stenosis: A specific complication of GPA that causes life-threatening airway narrowing; often misdiagnosed as asthma. [13]
1.3 Clinical Pearls
Diagnostic Pearl: "The 'C' in c-ANCA stands for 'Cytoplasmic' and 'PR3', while 'P' in p-ANCA stands for 'Perinuclear' and 'MPO'." In clinical practice, always verify Indirect Immunofluorescence (IIF) patterns with specific ELISA for PR3 and MPO antibodies. Approximately 10% of AAV patients (particularly those with EGPA or localized GPA) are "ANCA-negative," emphasizing that a negative serology does not exclude the diagnosis if the clinical phenotype (e.g., asthma + eosinophilia + mononeuritis) is compelling. This "seronegative" status often represents a diagnostic challenge, leading to delays in life-saving immunosuppression. Always biopsy whenever possible to confirm the "pauci-immune" nature of the inflammation. [12]
Examination Pearl: "Saddle nose deformity is highly suggestive of GPA, but look deeper for subglottic stenosis." Inspect the nasal bridge for collapse, which occurs due to necrotizing chondritis of the nasal septum. However, the more dangerous airway complication is subglottic stenosis. Listen for unexplained stridor or a "fixed" wheeze that does not respond to bronchodilators. If a patient with known or suspected GPA presents with hoarseness or difficulty breathing, urgent ENT consultation for fiberoptic laryngoscopy is mandatory, as this can lead to sudden, complete airway obstruction. [13]
Treatment Pearl: "Avacopan allows for a 'steroid-sparing' induction regimen, reducing the 'accrued damage' from glucocorticoids." The ADVOCATE trial was a landmark study demonstrating that avacopan (a C5aR antagonist) could replace or significantly reduce the need for high-dose glucocorticoids. This is a game-changer for older patients or those with pre-existing diabetes and osteoporosis, who often suffer more from steroid complications than from the vasculitis itself. When starting induction, consider the patient's "steroid risk profile" to determine if avacopan is a viable alternative to the standard high-dose taper. [11]
Pitfall Warning: "Do not rely on ANCA titres to guide treatment titration; 'treat the patient, not the lab result'." While a rising ANCA titre may precede a clinical flare in a subset of patients, it is neither sensitive nor specific enough to justify increasing immunosuppressive doses in the absence of clinical symptoms or new organ dysfunction. Conversely, a patient can have a severe, life-threatening relapse despite a negative or stable ANCA titre. Clinical monitoring, including regular urine dipsticks for new hematuria and CRP monitoring, remains the gold standard for detecting activity. [14]
Emergency Pearl: "Pulmonary-Renal Syndrome is a time-critical medical emergency." The combination of haemoptysis (suggesting alveolar haemorrhage) and a rapidly rising creatinine (suggesting crescentic glomerulonephritis) requires immediate admission to a High Dependency or Intensive Care Unit. Start pulsed methylprednisolone immediately—do not wait for ANCA results or biopsy confirmation if the clinical suspicion is high. Every hour of delay in starting steroids increases the risk of irreversible renal failure or fatal pulmonary haemorrhage. [15]
Mnemonic: "ELKS" for GPA involvement helps ensure a systemic screen.
- E - ENT (Sinusitis, saddle nose, otitis media, hearing loss, mastoiditis)
- L - Lung (Nodules, cavitation, DAH, capillaritis, infiltrates)
- K - Kidney (Pauci-immune glomerulonephritis, hematuria, proteinuria, renal failure)
- S - Skin/Systemic (Purpura, fever, weight loss, myalgia, arthralgia, nerve involvement) Use this mnemonic during every review to ensure you haven't missed a new organ being affected by the vasculitis.
Exam Pearl: "Pauci-immune" is the 'must-know' term for AAV histology. Examiners frequently ask why AAV is "pauci-immune." It refers to the lack of significant immunoglobulin or complement deposition on immunofluorescence. This is the key differentiator from other small-vessel vasculitides like SLE (Type III hypersensitivity with "full house" staining) or IgA vasculitis (Henoch-Schönlein purpura with IgA deposition). It confirms the disease is driven by direct neutrophil-antibody interaction (Type II-like) rather than the deposition of pre-formed immune complexes.
1.4 Why This Matters Clinically
- Patient outcomes: AAV is a "medical time bomb." Untreated, it is rapidly fatal, with an 80% mortality rate at 1 year. Delayed diagnosis of GPA can lead to irreversible renal failure requiring life-long dialysis or catastrophic pulmonary haemorrhage within hours to days. Early intervention is not just about survival; it is about preserving organ function and improving the "quality of survival" by preventing the accrual of permanent damage from both the disease and the toxic treatments. [3, 4]
- Healthcare burden: The management of AAV is resource-intensive. Patients frequently require prolonged ICU stays during the induction phase. Furthermore, the cost of modern biological therapies is substantial (Rituximab costs >£1000/dose), and the long-term costs associated with managing complications such as ESRD (dialysis/transplant) or chronic opportunistic infections represent a significant economic burden on healthcare systems. Shifting from reactive to proactive, early-induction management is more cost-effective in the long run.
- Medico-legal: Missing the diagnosis of AAV is a common source of litigation, particularly in primary care and emergency departments. Failure to recognize "red flag" symptoms like haemoptysis combined with a "dirty" urine dipstick, or misinterpreting a negative ANCA result as "no vasculitis," can lead to devastating outcomes. Physicians must maintain a high index of suspicion for "unresolved" symptoms (e.g., sinusitis or asthma) that do not respond to standard therapy in older adults.
- Training relevance: AAV is a "high-yield" topic for postgraduate exams (MRCP, USMLE Step 2/3, FRACP) because it tests a candidate's ability to integrate complex basic science (immunology), systemic clinical signs, and the interpretation of landmark clinical trials (e.g., ADVOCATE, PEXIVAS). It represents the intersection of multiple specialties (rheumatology, nephrology, respiratory), making it a favorite for multi-disciplinary exam questions.
SECTION 2: Epidemiology
2.1 Incidence & Prevalence
- Incidence: 15–30 per million person-years in European and North American populations. There has been a documented increase in incidence over the last 30 years, likely due to a combination of increased clinical awareness, better diagnostic criteria (ACR/EULAR 2022), and the widespread availability of specific ELISA testing for PR3 and MPO. [2, 16]
- Prevalence: 300–450 per million; prevalence is rising significantly because modern treatments have shifted AAV from an acute fatal illness to a chronic, relapsing condition. This creates an accumulating "prevalent pool" of patients who require long-term immunosuppression and monitoring. [3]
- Lifetime Risk: Estimated at 0.1% in the general population, but risk is markedly higher in individuals with specific genetic predispositions or occupational exposures. [17]
- Trend: Globally, the incidence is rising, with a notable shift toward older age groups. The median age at diagnosis has increased from 45 to over 60 years in many longitudinal cohorts, reflecting the global aging population.
- Geographic Variation: A striking "North-South" gradient exists globally. PR3-ANCA GPA is the dominant phenotype in Northern latitudes (Northern Europe, Scandinavia, UK), while MPO-ANCA MPA and renal-limited vasculitis are far more prevalent in Southern Europe, Japan, China, and parts of the Southern Hemisphere. [18]
- Temporal Patterns: Seasonal peaks have been observed, with some studies showing higher rates of GPA presentation in the winter and spring months. This is hypothesized to be linked to antecedent respiratory infections, particularly Staphylococcus aureus or influenza, which may trigger the disease via molecular mimicry. [21]
- Healthcare Burden: AAV accounts for approximately 5-10% of all cases of rapidly progressive glomerulonephritis (RPGN) in tertiary nephrology centers and is a leading cause of adult-onset multi-organ failure of unknown origin.
2.2 Demographics Table
| Factor | Details | Clinical Significance |
|---|---|---|
| Age | Peak: 65–75 years; Median onset: 55-60 years | Affects patients during retirement; high risk of treatment toxicity in this age group. |
| Sex | Male:Female 1.2:1 (GPA/MPA); 1:1.1 (EGPA) | Slight male bias in GPA/MPA; contrast with the 9:1 female bias in SLE. |
| Ethnicity | White/Caucasian (Highest risk for GPA) | GPA is notably rare in Black populations; MPA is common in East Asian cohorts. |
| Geography | North-South Gradient (GPA vs MPA) | Environmental factors (UV, Vitamin D) may influence the ANCA phenotype. [18] |
| Genetics | HLA-DPB104:01 (GPA); HLA-DQB103:03 (MPA) | Genetic risk follows the ANCA type (PR3 vs MPO) rather than the clinical label. [17] |
| Occupation | Silica exposure, farming, solvent exposure | Silica inhalation is a potent trigger for MPO-AAV; legal implications for industrial workers. |
| Age at GPA | Median ~50-55 years | GPA patients tend to be slightly younger than MPA patients at first presentation. |
| Age at EGPA | Median ~48 years | Often follows years of adult-onset asthma and allergic rhinitis. |
| Pediatric AAV | Rare (less than 3% of cases) | Children often have severe, rapidly progressive renal disease (RPGN) at presentation. |
| Socioeconomic | No strong association | Unlike RA or SLE, AAV does not show a strong socioeconomic gradient. |
2.3 Risk Factors Tables
Non-Modifiable Risk Factors:
| Factor | Relative Risk (95% CI) | Mechanism |
|---|---|---|
| HLA-DPB1*04:01 | OR 3.0 - 4.5 | Enhanced presentation of PR3 peptides to autoreactive T-cells in GPA. [17] |
| SERPINA1 (Z allele) | OR 2.5 - 3.8 | Deficiency of Alpha-1 antitrypsin, the primary inhibitor of PR3 enzyme. [20] |
| PRTN3 Polymorphism | OR 1.6 - 2.2 | Increased constitutive expression of PR3 on the neutrophil plasma membrane. |
| HLA-DQB1*03:03 | OR 2.5 - 3.5 | Specific MHC-II binding of MPO peptides; common in Asian MPA cohorts. |
| Age > 65 | RR 2.8 (vs less than 40) | Immunosenescence and cumulative exposure to sub-clinical environmental triggers. |
| Male Sex | RR 1.2 | Potential hormonal modulation of the magnitude of the B-cell response. |
| HLA-B*50:01 | OR 2.0 | Associated specifically with the EGPA phenotype in European populations. |
| Family History | RR 1.5 - 1.8 | Modest increase; indicates a polygenic risk rather than Mendelian inheritance. |
| PTPN22 (620W) | OR 1.4 | A common autoimmunity gene (also in RA/SLE) affecting T-cell signaling thresholds. |
Modifiable/Environmental Risk Factors:
| Risk Factor | Relative Risk (OR) | Evidence Level | Impact |
|---|---|---|---|
| Silica Exposure | OR 2.5–6.5 | Level 2a | Strongest environmental link; primarily associated with MPO-MPA. [19] |
| S. aureus (Nasal) | OR 2.0 (Relapse) | Level 2b | Chronic nasal carriage in GPA correlates with a higher risk of disease flare. [21] |
| Hydralazine / PTU | High (Drug-induced) | Level 3 | Causes high-titre MPO-ANCA and a "Lupus-like" vasculitis syndrome. [22] |
| Solvent Exposure | OR 1.8 - 2.0 | Level 2b | Associated with increased risk of necrotizing renal vasculitis in industrial workers. |
| Particulate Matter | OR 1.3 | Level 3 | Air pollution (PM2.5) may trigger systemic flares via TLR-mediated pathways. |
| Farming/Agriculture | OR 1.6 - 1.9 | Level 2b | Exposure to pesticides and fungal spores in rural settings may prime neutrophils. |
| Infectious Triggers | OR 1.5 | Level 3 | Molecular mimicry between bacterial proteins and human PR3/MPO enzymes. |
| Smoking | OR 0.7 - 0.9 | Level 3 | Paradoxically associated with a lower risk of AAV (similar to Ulcerative Colitis). |
2.4 Protective Factors
- UV Exposure/Latitude: Higher UV exposure at lower latitudes is associated with a lower risk of GPA. This may be mediated by Vitamin D, which enhances the function of T-regulatory (Treg) cells and suppresses Th17-mediated inflammation. [18]
- HLA-DRB1*13:02: Suggested in several immunogenetic studies as a protective allele against the development of MPO-associated vasculitis.
- Alpha-1 Antitrypsin (Normal levels): Adequate levels of the PiMM phenotype provide protection against PR3-mediated tissue destruction by rapidly inactivating the enzyme when released. [20]
- T-regulatory Cell Frequency: High levels of circulating CD4+CD25+FoxP3+ T-cells are associated with a lower risk of clinical relapse in established AAV.
SECTION 3: Pathophysiology
⚠️ MANDATORY: 7-STEP MECHANISM
The pathogenesis of ANCA-associated vasculitis (AAV) is a complex, multi-hit process involving genetic predisposition, environmental triggers, and a catastrophic failure of both innate and adaptive immune regulation. It is fundamentally characterized by the bypass of immune checkpoints, leading to the production of autoantibodies that directly activate neutrophils, turning them into agents of vascular destruction.
Step 1: Loss of Self-Tolerance and Antigen Processing The initiation of AAV requires a fundamental breakdown in immunological tolerance to primary neutrophil granule proteins: Proteinase 3 (PR3) and Myeloperoxidase (MPO). This process is heavily influenced by genetic susceptibility, particularly polymorphisms in the HLA-DPB1 locus for PR3-ANCA. In susceptible individuals, environmental "hits"—such as chronic silica inhalation or nasal carriage of Staphylococcus aureus—induce a pro-inflammatory state. In GPA, molecular mimicry between bacterial proteins (e.g., FbpA in S. aureus) and human PR3 may trigger the initial autoreactive B-cell response. Autoreactive B-cells escape central deletion in the bone marrow and peripheral tolerance in the spleen. Under the influence of Th17 and Th1 T-cell "help," these B-cells undergo somatic hypermutation and class-switch recombination within germinal centers, resulting in the production of high-affinity ANCA (IgG1 and IgG4 subclasses). B-lymphocyte stimulator (BLyS/BAFF) levels are typically elevated, promoting the survival of these autoreactive clones. [17, 23]
Step 2: Cytokine-Mediated Neutrophil Priming In a quiescent state, PR3 and MPO are sequestered within the azurophilic (primary) granules of neutrophils. However, "priming" by systemic inflammatory stimuli—such as TNF-α, IL-1β, or bacterial lipopolysaccharide (LPS)—triggers a dramatic shift in neutrophil physiology. This priming induces the translocation of intracellular granules and secretory vesicles to the plasma membrane, resulting in the "surface expression" of PR3 and MPO. Crucially, C5a (an anaphylatoxin generated via low-level alternative complement pathway "tick-over") acts as a potent priming agent by binding to the C5a receptor 1 (C5aR1/CD88). This receptor-ligand interaction activates G-protein coupled signaling pathways, mobilizing granules to the cell surface. Once primed, the neutrophil is no longer an "innocent bystander" but a "loaded weapon," presenting its internal enzymes as targets for circulating ANCA antibodies. [24, 30]
Step 3: ANCA-Mediated "Dual Engagement" and Activation Circulating ANCA bind to the surface-expressed PR3 or MPO. This activation is uniquely pathognomonic because it involves "dual engagement": the F(ab')2 portion of the IgG antibody binds to the target antigen (PR3/MPO), while the Fc portion of the same antibody binds to Fcγ receptors (specifically FcγRIIa and FcγRIIIb) on the neutrophil surface. This simultaneous binding bypasses the normal inhibitory "brakes" of the neutrophil. This triggers a massive, unregulated intracellular signaling cascade involving the Src family kinases, Spleen Tyrosine Kinase (Syk), and the p38 Mitogen-Activated Protein Kinase (MAPK) pathway. The activation of Phospholipase C (PLC) leads to an influx of calcium and the assembly of the NADPH oxidase complex. The result is an explosive "respiratory burst" with the massive production of reactive oxygen species (ROS) and the unregulated degranulation of proteolytic enzymes (elastase, PR3, MPO) into the extracellular space. [24, 427]
Step 4: Alternative Complement Pathway Amplification Loop The initial ANCA-mediated neutrophil activation and resulting endothelial damage trigger a critical amplification loop via the alternative complement pathway. Unlike other vasculitides where complement is "consumed" (Type III), in AAV, the alternative pathway is actively upregulated. Neutrophil-derived factors (including properdin and Factor B) facilitate the formation of the C3bBb (C3 convertase) on the neutrophil and endothelial surfaces. This leads to the generation of more C5a, which feeds back onto the C5aR1 on neutrophils. This creates a self-sustaining "vicious cycle": ANCA activates neutrophils → neutrophils activate the alternative complement pathway → C5a is produced → C5a primes more neutrophils for ANCA-mediated activation. This specific loop is the target of the novel therapy, avacopan. This "vicious cycle" explains the rapid progression of the disease and the sustained nature of the inflammation even when the initial trigger is removed. [26, 31]
Step 5: NETosis and "Trojan Horse" Endothelial Damage Severely activated neutrophils undergo a specialized form of cell death called "NETosis." They extrude their nuclear chromatin (DNA and histones) into the extracellular space, coated with granular proteins like MPO and PR3, forming Neutrophil Extracellular Traps (NETs). While NETs are evolutionarily designed to trap and kill bacteria, in AAV they act as a "Trojan Horse." NETs directly damage the vascular endothelium through histone-mediated cytotoxicity and by concentrating proteolytic enzymes directly against the vessel wall. Furthermore, NETs act as a "scaffold" for the continued activation of the alternative complement pathway and provide a persistent source of extracellular autoantigens (PR3/MPO) to plasmacytoid dendritic cells. This interaction via TLR9 further fuels the production of Interferon-alpha, reinforcing the autoimmune loop and promoting B-cell maturation. [25, 27]
Step 6: T-cell Mediated Granulomatous Inflammation Specifically in GPA and EGPA, a parallel process of granuloma formation occurs. This is primarily a T-cell driven process involving Th1 and Th17 subsets. These cells secrete Interleukin-17 (IL-17) and IL-23, which recruit and activate macrophages. These macrophages transform into epithelioid cells and multinucleated giant cells, organizing into areas of "geographic necrosis." This necrotic core is surrounded by palisading histiocytes and micro-abscesses. In EGPA, there is an additional recruitment of eosinophils, driven by IL-5, leading to significant tissue infiltration and the release of Major Basic Protein (MBP), which is directly cardiotoxic. These granulomatous lesions are distinct from the vasculitis; they cause "mass effect" damage, such as bone erosion in the paranasal sinuses, orbital pseudotumors, and permanent subglottic stenosis. [28, 29]
Step 7: Fibrinoid Necrosis and Crescent Formation The final common pathway of AAV is the destruction of the small vessel wall, known as "fibrinoid necrosis." The accumulation of ROS, NETs, and proteolytic enzymes leads to the breakdown of the internal elastic lamina and the replacement of the vessel wall with a bright pink, amorphous material composed of fibrin and necrotic debris. In the glomerulus, this damage causes the rupture of the glomerular basement membrane (GBM). Plasma proteins, including fibrinogen, leak into Bowman's space, triggering the proliferation of parietal epithelial cells and the infiltration of macrophages. This creates the "crescent"—the hallmark of rapidly progressive glomerulonephritis (RPGN). As the acute inflammation subsides, the damaged tissue is replaced by collagen. In the kidneys, this leads to segmental and global glomerulosclerosis; in the lungs, it results in permanent scarring or the formation of large, thick-walled cavitary nodules. [27, 407]
3.6 Classification Table
| Disease | ANCA Association | Key Histology | Hallmark Organs | Radiographic Features |
|---|---|---|---|---|
| GPA | PR3-ANCA (80-90%) | Granulomas + Necrosis + Pauci-immune Vasculitis | ENT, Lungs, Kidneys | Cavitating nodules, Sinus erosion |
| MPA | MPO-ANCA (90%) | Necrosis (No granulomas) | Kidneys, Lungs (DAH) | Diffuse Alveolar Hemorrhage (GGO) |
| EGPA | MPO-ANCA (40%) | Eosinophils + Granulomas + Asthma | Lungs, Heart, Nerves | Transient infiltrates, Cardiomyopathy |
| RLV | MPO-ANCA (100%) | Necrotizing Crescentic GN only | Kidneys exclusively | Normal CXR; Small kidneys if chronic |
| DI-AAV | MPO-ANCA (Very high) | Mixed features; often "lupus-like" | Skin, Kidneys | Variable; depends on drug |
| AAV (Seronegative) | Negative (10%) | Pauci-immune Vasculitis | Variable | Same as GPA/MPA |
| Overlap Syndrome | Dual ANCA (Rare) | Combined features | Multi-system | Complex, overlapping |
3.7 Anatomical Considerations
- Glomerular Microvasculature: The glomerular capillary tuft is the primary target in renal AAV. The high-pressure filtration system (approx. 60 mmHg) and the fenestrated endothelium (70-100nm pores) make it particularly susceptible to the deposition of activated neutrophils and the subsequent rupture of the basement membrane. The lack of immune complexes (pauci-immune) means there is no "barrier" to the neutrophil-mediated damage.
- Nasal Septum and Subglottis: GPA has a predilection for these regions. The nasal septum's poor collateral blood supply makes it vulnerable to necrotizing chondritis, leading to the characteristic "saddle nose." The subglottic region is a physiological "choke point" where inflammation leads to high-velocity airflow and mechanical trauma, exacerbating the fibrotic response and causing critical stenosis.
- Vasa Nervorum: These are the microscopic nutrient vessels that supply peripheral nerves. Vasculitic involvement leads to nerve ischemia and the clinical syndrome of mononeuritis multiplex. The peroneal and ulnar nerves are most commonly affected due to their long course and specific anatomical vulnerabilities at the fibular head and cubital tunnel.
- Orbital Apex: Granulomatous masses in GPA can infiltrate the orbital apex, compressing the optic nerve (CN II) and the extraocular motor nerves (CN III, IV, VI), leading to vision loss and complex ophthalmoplegia.
3.8 Physiological Considerations
- Pauci-immune State: Physiologically, AAV differs from other vasculitides (like SLE or IgA vasculitis) because it does not rely on the deposition of large, pre-formed immune complexes (Type III hypersensitivity). Instead, it is a Type II-like hypersensitivity where the antibody (ANCA) binds directly to its target on the cell surface (Neutrophil), causing "in situ" activation.
- Alternative Pathway "Tick-over": The constant, low-level activation of the alternative complement pathway provides a "ready-to-fire" mechanism. In AAV, the loss of regulatory control (e.g., Factor H dysfunction) or over-activation (via NETs) leads to the explosive generation of C5a, which is the primary driver of neutrophil recruitment and priming.
- Protease/Anti-protease Balance: The physiological balance between PR3 (a serine protease) and Alpha-1 Antitrypsin (AAT - the primary inhibitor) is disrupted. Genetic deficiency (PiZ allele) or oxidative inactivation of AAT by neutrophil-derived ROS allows PR3 to cause unchecked tissue destruction.
- Hypoxia-Inducible Factors: In the kidney, vasculitis-induced ischemia activates HIF-1α, which paradoxically may further drive pro-inflammatory gene expression and exacerbate the fibrotic response in the glomerular crescent.
SECTION 4: Clinical Presentation
4.1 Symptoms
The clinical presentation of AAV is notoriously protean, often beginning with a non-specific "flu-like" prodrome that can last from weeks to months. This "smoldering" phase is frequently misattributed to viral infections, burnout, or occult malignancy until organ-specific symptoms emerge with sudden, often devastating, clarity.
Typical Presentation (8+ symptoms):
| Symptom | Frequency | Character | Duration | Associations |
|---|---|---|---|---|
| Constitutional | 90% | Fever, weight loss (> 10%), night sweats | Weeks to months | Prodomal phase; often mimics occult malignancy |
| Sinusitis | 70% | Purulent/bloody nasal discharge, crusting | Chronic/Recurrent | Refractory to multiple courses of antibiotics |
| Dyspnoea/Cough | 50% | May have haemoptysis (red flag) | Acute or subacute | Suggests pulmonary nodules or DAH |
| Arthralgia | 60% | Migratory, non-erosive large joints | Weeks | Often the earliest systemic symptom in GPA |
| Hearing Loss | 35% | Conductive (otitis) or Sensorineural | Chronic | Eustachian tube dysfunction; mastoiditis |
| Skin Purpura | 40% | Palpable, non-blanching spots | Recurrent crops | Suggests leukocytoclastic vasculitis |
| Myalgia | 50% | Generalised muscle aches; "heavy" limbs | Subacute | High ESR/CRP; can mimic PMR |
| Numbness | 20% | Asymmetric, "pins & needles," weakness | Sudden onset | Mononeuritis multiplex (e.g., foot drop) |
| Eye Pain | 15% | Deep, boring pain; worse with movement | Acute | Scleritis or episcleritis |
| Epistaxis | 30% | Recurrent, often with large crusts | Chronic | Nasal septal erosion; GPA hallmark |
| Abdominal Pain | 10% | Post-prandial "intestinal angina" | Subacute | Suggests mesenteric vasculitis (Poor prognosis) |
Atypical Presentations:
- Isolated Subglottic Stenosis: Presenting purely as a chronic wheeze or stridor, often misdiagnosed as refractory asthma for years. Patients may have a "fixed" inspiratory wheeze on auscultation. [13]
- Orbital Pseudotumor: Presenting as unilateral proptosis, diplopia, and pain. It mimics an orbital tumor or Graves' ophthalmopathy but responds dramatically to steroids.
- Pachymeningitis: Chronic, positional headaches and multiple cranial nerve palsies (CN VI, VII, VIII) due to dural thickening and inflammation.
- Cardiac Involvement: More common in EGPA (myocarditis, coronary vasculitis). Patients may present with acute heart failure or MI-like symptoms with "clean" coronary arteries on angiography.
- Isolated Skin Vasculitis: Rarely, AAV can present with only cutaneous findings for years before "converting" to a systemic form.
4.2 Signs
General Examination:
- Vitals: Hypertension (due to renal damage), tachycardia, and low-grade pyrexia. High-grade fever suggests severe flare or secondary infection.
- Appearance: Often appears "cachectic" or chronically unwell. Inspect for "Cushingoid" features if they have already been started on steroids by other physicians.
System-Specific Signs:
| Sign | How to Elicit | Significance | Sensitivity/Specificity |
|---|---|---|---|
| Saddle Nose | Visual inspection of nasal bridge profile | Destruction of nasal septum chondritis | 20% / 95% (GPA) |
| Palpable Purpura | Palpation of lower limbs | Small vessel vasculitis | 40% / 60% |
| RBC Casts | Microscopy of fresh urine sediment | Pathognomonic for Glomerulonephritis | 70% / 95% (Renal) |
| Stridor | Listen for high-pitched inspiratory sound | Subglottic stenosis (Airway emergency) | 10% / 90% |
| Foot/Wrist Drop | Motor testing of distal limbs | Mononeuritis Multiplex (Nerve ischemia) | 15% / 90% |
| Proptosis | Inspect from above ("bird's eye view") | Orbital granulomatous mass | 5% / 95% (GPA) |
| Scleritis | Visual (violaceous deep red hue) | Serious ocular vasculitis; vision threat | 10% / 85% |
| Basal Crackles | Auscultation of posterior lung bases | Suggests DAH or associated Fibrosis | 30% / 40% |
| Episcleritis | Visual (bright red, blanches with phenylephrine) | Superficial ocular inflammation | 15% / 50% |
4.3 Red Flags
[!CAUTION] RED FLAGS — Seek immediate help if:
- Haemoptysis: Even small amounts suggest Diffuse Alveolar Haemorrhage (DAH); mortality is > 50% if untreated. [15]
- Rapidly rising creatinine: Suggests RPGN; kidneys can be lost irreversibly within 48–72 hours.
- New-onset Stridor: Suggests critical subglottic stenosis; requires urgent airway review by ENT/Anaesthesia.
- Sudden vision loss/Red Eye: Suggests retinal vasculitis or necrotizing scleritis.
- Acute "Foot Drop": Suggests mononeuritis multiplex; requires urgent IV pulse methylprednisolone.
- Coke-colored urine: Suggests macroscopic hematuria from acute crescentic glomerulonephritis.
- Severe Post-Prandial Abdominal Pain: Suggests mesenteric ischemia from vasculitis.
4.4 Clinical Scoring
- BVAS (Birmingham Vasculitis Activity Score) v3: The primary tool for quantifying disease activity. It uses a structured checklist of 63 items across 9 organ systems. A score of > 0 indicates active disease; a score of 0 is required for "complete remission." [34]
- VDI (Vasculitis Damage Index): Captures permanent damage (present for > 3 months). This distinguishes between "active inflammation" and "permanent scarring," which is crucial for determining if more immunosuppression is needed.
- Five-Factor Score (FFS): Used specifically in EGPA and PAN. Factors: Age > 65, Cardiac, GI, Renal insufficiency (Cr > 150), and Absence of ENT involvement. A score ≥1 indicates the need for cyclophosphamide induction.
- ANCA-Associated Vasculitis Renal Risk Score (ARRS): Predicts the risk of progression to ESRD based on % normal glomeruli, % tubular atrophy/interstitial fibrosis, and GFR at diagnosis.
SECTION 5: Clinical Examination
5.1 Structured Approach
A suspected AAV patient requires a meticulous "head-to-toe" systemic vasculitis screen.
- General Assessment:
- Assess for "ill appearance," cachexia, and chronic respiratory distress.
- Measure BP in both arms (to rule out large-vessel overlap/Takayasu-like features).
- HEENT (High-Yield in GPA):
- Nose: Check bridge for collapse (Saddle nose). Use a speculum to look for "crusting," friable mucosa, and septal perforation.
- Ears: Perform otoscopy to look for serous otitis media. Test hearing (Rinne/Weber) as sensorineural loss is common.
- Throat: Assess voice quality (hoarseness) and listen specifically for inspiratory stridor.
- Ophthalmic:
- Differentiate episcleritis from scleritis (deep red hue, does not blanch with phenylephrine, deep boring pain).
- Check for proptosis by inspecting from above ("bird's eye view").
- Respiratory:
- Auscultate for "velcro" crackles (fibrosis) or localized dullness (DAH or large nodules).
- Check for wheeze, especially in EGPA or endobronchial GPA.
- Neurological:
- Perform a focused motor and sensory screen of the distal nerves.
- Test "Great toe extension" (Peroneal) and "Finger abduction" (Ulnar).
- Sensory loss in the first webspace is an early, sensitive sign of peroneal nerve ischemia.
- Renal:
- Immediate point-of-care urinalysis is mandatory for all suspected cases.
- Skin:
- Inspect lower limbs and pressure areas for palpable purpura, livedo reticularis, or digital infarcts.
5.2 Special Tests Table
| Test Name | Technique | Positive Finding | Sensitivity | Specificity | Clinical Use |
|---|---|---|---|---|---|
| Urine Dipstick | Fresh mid-stream sample | Protein (++) and Blood (+++) | 90% | 60% | Primary screening |
| RBC Cast Screen | Phase-contrast microscopy | RBC Casts or dysmorphic RBCs | 70% | 95% | Confirms GN |
| Nasal Rhinoscopy | Fiberoptic ENT exam | "Cobblestone" mucosa, crusts | 70% | 85% | GPA confirmation |
| Weber/Rinne | 512Hz Tuning fork | Asymmetric sensorineural loss | 40% | 30% | Nerve/ENT screen |
| Monofilament | 10g monofilament on foot | Loss of sensation in nerve territory | 30% | 70% | Peripheral nerve |
| Cranial Nerve | Standard CN I-XII assess | Palsy (often VI, VII, or VIII) | 10% | 90% | CNS involvement |
| Schirmer's Test | Filter paper in lower lid | less than 5mm moisture in 5 mins | 20% | 40% | Sicca overlap |
| Allen's Test | Compress radial/ulnar art | Delayed capillary refill | 5% | 80% | Digital ischemia |
5.3 Examination by Severity
- Mild/Limited Disease (BVAS less than 3): Isolated nasal crusting, mild arthralgia, or a few spots of skin purpura. No constitutional symptoms and stable renal function. Often manageable with MTX or MMF.
- Moderate Disease (BVAS 3-8): Systemic symptoms (fever, weight loss) with multi-organ involvement (e.g., lungs + joints) but stable renal function (Cr less than 200 µmol/L). Standard induction (RTX) is mandatory.
- Severe Disease (BVAS > 8): Red flags present (Haemoptysis, Cr > 300 µmol/L, Foot Drop). This is an "organ-threatening" or "life-threatening" state requiring pulsed steroids and high-intensity induction (RTX/CYC).
SECTION 6: Investigations
6.1 Bedside Tests
| Test | Expected Finding | Purpose | Urgency |
|---|---|---|---|
| Urinalysis | Haematuria (+++), Proteinuria (++) | Primary screen for active GN | Immediate |
| Blood Pressure | Hypertension (> 140/90) | Marker of acute renal damage | Immediate |
| ECG | Sinus tachycardia; ST-T changes | Screen for myocarditis (EGPA) | Urgent |
| Peak Flow | Flattened inspiratory loop | Screen for subglottic stenosis | Urgent |
| Urine Microscopy | RBC Casts; Dysmorphic RBCs | Confirms glomerular source | Urgent |
| Bedside Echo | Pericardial effusion/Wall motion | Screen for cardiac vasculitis | Urgent |
6.2 Laboratory Tests
| Test | Expected Finding | Purpose | Turnaround |
|---|---|---|---|
| ANCA ELISA (PR3) | Positive (> 10 IU/mL) | High specificity for GPA | 24-48h |
| ANCA ELISA (MPO) | Positive (> 10 IU/mL) | High specificity for MPA/EGPA | 24-48h |
| Creatinine / GFR | Elevated / Rapidly falling | Monitor for RPGN / AKI | less than 4h |
| ESR / CRP | Significantly elevated (> 100) | Measure of systemic inflammation | less than 4h |
| Eosinophils | > 1.5 x 10^9/L (or > 10%) | Diagnostic for EGPA | less than 4h |
| C3 / C4 Levels | Normal (Usually) | Distinguishes AAV from SLE/Cryo | 24-48h |
| Blood Cultures | Negative | Rule out Infective Endocarditis | 48-72h |
| TPMT Activity | Normal | Safety check before Azathioprine | 3-5 days |
| HIV/Hep B/Hep C | Negative | Screen before starting RTX/CYC | 24h |
| IGRA (Quantiferon) | Negative | Latent TB screen before biologics | 48-72h |
| Pro-BNP | Elevated | Screen for heart failure in EGPA | 4h |
6.3 Imaging
- CT Chest (HRCT):
- GPA: Multiple nodules (often > 1cm), thick-walled cavitation, "halo sign" (ground-glass surrounding a nodule indicating localized hemorrhage).
- MPA: Diffuse ground-glass opacities (GGO) in a "bat-wing" distribution, suggesting DAH.
- EGPA: Transient, migratory "cloud-like" infiltrates; may see bronchial wall thickening.
- CT Sinuses: Shows "bone destruction" (medial wall of the antrum, nasal septum). This "erosive" feature distinguishes GPA from simple allergic or infectious sinusitis.
- MRI Brain/Orbits: Shows dural enhancement (pachymeningitis) or a retro-orbital mass (pseudotumor) that may be encasing the optic nerve.
- Cardiac MRI: Late Gadolinium Enhancement (LGE) in a non-coronary distribution suggests vasculitic myocarditis (especially in EGPA).
- PET-CT: May show occult large-vessel involvement or guide biopsy to the most metabolically active site.
6.4 Diagnostic & Pathological Criteria
- Renal Biopsy (Berden Classification):
- Focal: > 50% normal glomeruli. This has the best prognosis for renal recovery.
- Crescentic: > 50% of glomeruli show cellular crescents. Indicates highly active disease requiring aggressive induction.
- Mixed: No feature > 50%. Intermediate prognosis.
- Sclerotic: > 50% globally sclerosed glomeruli. Poor prognosis; damage is likely irreversible.
- ACR/EULAR 2022 Criteria Summary (GPA):
- "Needs score ≥5:"
- c-ANCA or PR3-ANCA positive (+5)
- Nasal crusts, bleeding, or ulceration (+3)
- Cartilage involvement (Saddle nose/Ears) (+2)
- Lung nodules or cavitation (+2)
- Granuloma on biopsy (+2)
- Pauci-immune GN on biopsy (+1)
- "Needs score ≥5:"
- ACR/EULAR 2022 Criteria Summary (MPA):
- "Needs score ≥5:"
- p-ANCA or MPO-ANCA positive (+10)
- Nasal symptoms (-3) - Points away from MPA toward GPA
- Pauci-immune GN (+3)
- "Needs score ≥5:"
6.5 Diagnostic Algorithm
- Clinical Suspicion: Patient has "ELKS" symptoms (ENT, Lung, Kidney, Skin) + significantly elevated CRP/ESR.
- Serological Screen: Order PR3/MPO ELISA and ANCA IIF (Indirect Immunofluorescence).
- Organ Screen: Urinalysis (dipstick and microscopy for casts) + HRCT Chest.
- Tissue Confirmation: Biopsy the most accessible active site (Skin is easiest, Kidney is most definitive, Lung is highest risk).
- Exclude Mimics: You MUST rule out Infective Endocarditis (Blood cultures x3 + Echo) and Malignancy (CT CAP) as these can both present with "pseudo-vasculitis."
SECTION 7: Management
⚠️ MANDATORY: ASCII MANAGEMENT ALGORITHM
┌─────────────────────────────────────────────────────────────────────────────┐
│ ANCA-ASSOCIATED VASCULITIS (AAV) MANAGEMENT │
│ (Based on 2023 EULAR/ERA and 2021 KDIGO Guidelines) │
└─────────────────────────────────────────────────────────────────────────────┘
│
▼
┌───────────────────────────────────────────────────┐
│ INITIAL SEVERITY ASSESSMENT │
│ • Life-threatening? (DAH, Stridor, RPGN) │
│ • Organ-threatening? (Active GN, Mononeuritis) │
│ • Limited/Non-organ threatening? (ENT, Skin) │
└───────────────────────────────────────────────────┘
│
┌──────────────────────┴──────────────────────┐
▼ ▼
┌──────────────────────┐ ┌──────────────────────────────┐
│ NON-SEVERE AAV │ │ SEVERE AAV │
│ (Normal Creatinine, │ │ (Organ/Life Threatening) │
│ no vital organ │ │ Cr > 150, DAH, Neuropathy │
│ involvement) │ └──────────────┬───────────────┘
└──────────┬───────────┘ │
│ ▼
▼ ┌───────────────────────────────┐
┌──────────────────────┐ │ INDUCTION THERAPY │
│ METHOTREXATE or │ │ 1. Methylprednisolone Pulse │
│ MYCOPHENOLATE │ │ (250-500mg IV for 3 days) │
│ + Prednisolone │ │ 2. Rituximab (RTX) OR │
│ taper │ │ Cyclophosphamide (CYC) │
└──────────┬───────────┘ │ 3. ± Avacopan (Steroid-sparing)│
│ └──────────────┬───────────────┘
▼ │
┌──────────────────────┐ ┌───────────────┴───────────────┐
│ MONITOR RESPONSE │ │ CONSIDER PLASMA EXCHANGE │
│ • Weekly Urinalysis │ │ (PEXIVAS Criteria: Cr > 500 or │
│ • Bi-weekly CRP/ESR │ │ severe DAH with hypoxia) │
└──────────┬───────────┘ └───────────────┬───────────────┘
│ │
└───────────────┬─────────────────────────────┘
│
▼
┌───────────────────────────────────────────┐
│ REMISSION MAINTENANCE │
│ (Start once Remission Achieved ~3-6mo) │
├───────────────────────────────────────────┤
│ • RITUXIMAB (500mg every 6 months) │
│ • OR AZATHIOPRINE (2mg/kg/day) │
│ • Duration: 24–48 months minimum │
└───────────────────────────────────────────┘
│
▼
┌───────────────────────────────────────────┐
│ LONG-TERM MONITORING │
│ • FBC/IgG levels (hypogammaglobulinaemia) │
│ • PJP Prophylaxis (Co-trimoxazole) │
│ • Cardiovascular risk optimization │
│ • Bone protection (Bisphosphonates) │
└───────────────────────────────────────────┘
7.1 Emergency/Acute Management
The immediate management of AAV is dictated by the presence of organ- or life-threatening manifestations. This phase is characterized by "hitting hard and hitting early" to stop the inflammatory cascade before irreversible fibrosis occurs. [8, 15]
- Glucocorticoid Pulses: Standard of care for severe AAV (RPGN or DAH) is IV Methylprednisolone 250–500 mg daily for 3 consecutive days. This provides rapid, non-genomic suppression of neutrophil activation and cytokine release. [11]
- Plasma Exchange (PEX): Historically controversial. The PEXIVAS trial clarified that PEX does not reduce the composite of death or ESRD in the overall cohort but may be considered in those with serum creatinine > 500 µmol/L or severe DAH with respiratory failure, though its routine use has declined. [32]
- Airway Protection: In patients with GPA and subglottic stenosis causing stridor, immediate ENT involvement for microlaryngoscopy and potential dilation or stenting is mandatory. High-dose steroids alone may not reverse mechanical obstruction in time.
- Renal Replacement Therapy (RRT): If the patient presents with uraemia, refractory hyperkalaemia, or fluid overload, early initiation of hemodialysis is required while waiting for immunosuppression to take effect.
7.2 Medical Management: Induction Therapy
Induction therapy aims to achieve clinical remission (BVAS = 0) within 3 to 6 months. [8, 9]
1. Rituximab (RTX) - Preferred for most patients
- Dose: 375 mg/m² weekly for 4 doses OR 1000 mg on day 1 and day 15.
- Rationale: The RAVE and RITUXVAS trials established non-inferiority to cyclophosphamide. RTX is superior for inducing remission in relapsing disease and is generally preferred in younger patients to preserve fertility. [10]
- Monitoring: B-cell counts (CD19/20) and total IgG levels.
2. Cyclophosphamide (CYC)
- Dose (IV): 15 mg/kg (max 1.2g) at weeks 0, 2, 4, then every 3 weeks.
- Dose (Oral): 2 mg/kg/day (max 150-200mg).
- Rationale: Still used in patients with very severe renal failure (Cr > 350) or those unable to access/tolerate rituximab. Oral CYC has a higher cumulative dose and higher risk of bladder toxicity/malignancy compared to pulsed IV regimens. [33]
- Toxicity Prevention: Mesna (for oral CYC) and high fluid intake to prevent haemorrhagic cystitis.
3. Avacopan (The "Steroid-Sparer")
- Dose: 30 mg twice daily for 52 weeks.
- Rationale: The ADVOCATE trial demonstrated that avacopan can replace or significantly reduce the need for high-dose glucocorticoids, improving renal recovery (estimated GFR increase) and reducing the Glucocorticoid Toxicity Index (GTI). [11]
- Evidence: Superiority to standard prednisolone taper at 52 weeks in achieving sustained remission.
7.3 Medical Management: Maintenance Therapy
Maintenance is essential to prevent relapses, which occur in up to 50% of PR3-AAV patients within 5 years. [35]
- Rituximab (Preferred): 500 mg IV every 6 months for 2 to 4 years. The MAINRITSAN trials established RTX as superior to azathioprine for maintaining remission. [36]
- Azathioprine: 2 mg/kg/day. Used if RTX is contraindicated or not tolerated. Requires TPMT monitoring before initiation.
- Methotrexate: 20-25 mg/week (with folic acid). Only for patients with normal renal function and non-severe disease.
- Mycophenolate Mofetil (MMF): 1g twice daily. Generally less effective than azathioprine for maintaining remission in AAV (IMPROVE trial) but may be used in selected cases. [37]
7.4 Supportive Care & Prophylaxis
The "ancillary" management of AAV is as critical as the immunosuppression itself to prevent iatrogenic mortality. [8, 14]
| Intervention | Detail | Rationale |
|---|---|---|
| PJP Prophylaxis | Co-trimoxazole 480mg OD or 960mg 3x/week | Essential during induction; RTX/CYC/Steroids create high risk for Pneumocystis. |
| Bone Protection | Bisphosphonates + Vit D + Calcium | High-dose steroids cause rapid bone loss and vertebral fractures. |
| GI Protection | Proton Pump Inhibitor (PPI) | High-dose steroids + potential stress ulceration. |
| CV Risk Mgmt | Statins / BP control (ACE-i/ARB) | AAV patients have a 3x higher risk of MI and Stroke due to chronic inflammation. |
| Vaccination | Influenza, Pneumococcal, COVID-19 | MUST be given at least 2 weeks before Rituximab or during B-cell recovery. |
| Screening | TB (Quantiferon) and Hepatitis B/C | High risk of reactivation under biological therapy. |
| Fertility | GnRH agonists / Sperm banking | Mandatory discussion before starting Cyclophosphamide. |
SECTION 8: Complications
8.1 Disease-Related Complications
| Complication | Incidence | Character | Management |
|---|---|---|---|
| Chronic Kidney Disease | 40-50% | Progressive loss of GFR due to scarring | BP control, RAAS blockade, Dialysis |
| End-Stage Renal Disease | 20-25% | Requires permanent RRT/Transplant | Transplant only after 6-12mo of remission |
| Pulmonary Fibrosis | 10-15% | Chronic scarring (often MPO+ patients) | Anti-fibrotics, transplant referral |
| Nasal Deformity | 20% | Saddle nose, septal perforation | Surgical reconstruction (delayed) |
| Vision Loss | 5-10% | Optic nerve compression or retinal vasculitis | Urgent escalation of immunosuppression |
| Hearing Loss | 30% | Conductive or sensorineural | Hearing aids, cochlear implants |
8.2 Treatment-Related Complications
| Complication | Incidence | Character | Management |
|---|---|---|---|
| Serious Infection | 25-30% | Bacterial, CMV, Fungal (during induction) | Early antibiotics, reduce immunosuppression |
| Hypogammaglobulinaemia | 15-20% | Low IgG after repeated RTX | IVIG replacement if recurrent infections |
| Neutropenia | 5-10% | CYC or RTX-induced (late-onset) | G-CSF, stop offending agent |
| Malignancy | 2-3x RR | Skin cancer, Bladder cancer (CYC), Lymphoma | Regular screening; avoid high cumulative CYC |
| Diabetes (Steroid) | 20-30% | New-onset or worsening hyperglycaemia | Metformin, Insulin, steroid-tapering |
| Infertility | Variable | Dose-dependent (CYC > 10g) | Egg/sperm banking prior to treatment |
SECTION 9: Prognosis & Outcomes
9.1 Natural History
Historically, AAV was a death sentence. In the pre-immunosuppressive era (pre-1970s), the median survival was 5 months, and the 1-year mortality rate exceeded 80%, primarily due to renal failure or massive pulmonary haemorrhage. [1, 4]
9.2 Modern Outcomes
With current induction and maintenance protocols, the prognosis has shifted dramatically: [3, 4, 38]
- Remission Rate: 85–90% of patients achieve remission with initial induction.
- 5-Year Survival: Now exceeds 75–80%.
- Relapse Rate: 30–50% at 5 years (higher in PR3-GPA). Relapses are more common in patients with ENT involvement and those with lower baseline creatinine.
- Leading Causes of Death:
- "Early (less than 1 year): Infection (secondary to immunosuppression) and active vasculitis."
- "Late (> 1 year): Cardiovascular disease, Malignancy, and Complications of CKD."
9.3 Prognostic Factors
| Factor | Impact on Prognosis | Characterization |
|---|---|---|
| ANCA Type | PR3-ANCA = Higher Relapse Risk | MPO-ANCA = More Chronic Fibrosis |
| Renal Function | GFR at baseline | Most powerful predictor of long-term survival and ESRD risk. |
| Berden Class | Focal = Best (> 90% survival) | Sclerotic = Worst (less than 50% renal survival at 1 yr) |
| Age | Older age = Higher treatment toxicity | RR 1.5 per decade of age for serious infection. |
| BVAS Score | Higher score = Higher early mortality | Reflects multi-organ physiological stress. |
| Five-Factor Score | Score ≥2 = Significant mortality risk | Guides use of cyclophosphamide in EGPA. |
SECTION 10: Evidence & Guidelines
10.1 Key Guidelines
- 2023 EULAR/ERA Recommendations for the Management of AAV:
- Recommendation: Rituximab or Cyclophosphamide + Glucocorticoids for induction of severe AAV.
- New Addition: Strong recommendation for the use of Avacopan to reduce steroid exposure. [8]
- 2021 KDIGO Clinical Practice Guideline for Glomerulonephritis:
- Focuses on the "PEXIVAS" findings, suggesting PEX only for high-risk renal patients. [9]
- 2022 ACR/EULAR Classification Criteria:
- Developed to provide high sensitivity and specificity for differentiating GPA, MPA, and EGPA in clinical research. [16]
10.2 Landmark Trials
| Trial (Year) | Population (n) | Comparison | Key Finding (Statistic) | Clinical Impact |
|---|---|---|---|---|
| RAVE (2010) | n=197 | RTX vs Oral CYC | RTX non-inferior; Superior for relapses | Established RTX as 1st line for AAV. [10] |
| PEXIVAS (2020) | n=704 | PEX vs No PEX; High vs Low Steroid | PEX did not reduce death/ESRD (HR 0.81, p=0.27). Reduced steroid non-inferior. [32] | Changed global practice to avoid routine PEX and use lower steroid doses. |
| ADVOCATE (2021) | n=331 | Avacopan vs Prednisone taper | Avacopan superior for sustained remission at 52w (64% vs 54%, p=0.007). [11] | Validated C5aR inhibition as a "steroid-free" induction strategy. |
| MAINRITSAN (2014) | n=115 | RTX vs Azathioprine (Maintenance) | Major Relapse 5% (RTX) vs 29% (AZA) at 28 months (HR 6.6, pless than 0.001). [36] | Established RTX as the gold standard for maintenance. |
| LOVAS (2020) | n=140 | Reduced vs High dose steroids | Reduced dose achieved remission non-inferiorly with fewer infections. [39] | Confirmed safety of rapid steroid tapering. |
SECTION 11: Patient/Layperson Explanation
11.1 What is ANCA Vasculitis?
Vasculitis simply means "inflammation of the blood vessels." In ANCA-associated vasculitis (AAV), your body’s immune system—which is supposed to protect you from germs—accidentally attacks your own small blood vessels.
Think of your blood vessels like a network of plumbing pipes that carry nutrients and oxygen to every part of your body. In this condition, the immune system (specifically white blood cells called neutrophils) becomes overactive and starts "attacking the pipes." This causes the "pipes" to become leaky, clogged, or even burst. Because blood vessels are everywhere, this condition can affect many different parts of the body at once, most commonly the kidneys, lungs, sinuses, and nerves.
11.2 Why Does It Matter?
If left untreated, the inflammation can cause permanent damage to vital organs.
- Kidneys: If the tiny filters in the kidneys are damaged, they can stop working, which might lead to the need for dialysis.
- Lungs: Inflammation can cause bleeding in the lungs or create "spots" (nodules) that make it hard to breathe.
- Sinuses: It often feels like a never-ending cold or sinus infection that doesn't get better with normal antibiotics. The good news is that with modern medicine, this is now a manageable "chronic" condition rather than the life-threatening emergency it used to be.
11.3 How Is It Treated?
Treatment usually happens in two main phases:
- Getting the fire out (Induction): For the first few months, we use strong "big gun" medications (like Rituximab or steroids) to quickly stop the immune system's attack.
- Keeping the fire out (Maintenance): Once the disease is quiet (in remission), we use milder medications for several years to make sure the "fire" doesn't flare up again.
You will likely need to take a small dose of an antibiotic (usually Co-trimoxazole) to protect you from infections while your immune system is being dialled down.
11.4 What to Expect
- The Hospital Stay: If you are very unwell at the start, you may need to stay in the hospital for a week or two to receive IV medications.
- The Long Term: Most people return to their normal activities, but you will need regular blood and urine tests for the rest of your life to monitor the condition.
- Energy Levels: It is very common to feel quite tired (fatigue) for several months even after the medical tests show the disease is quiet.
11.5 When to Seek Help
You should contact your specialist team or go to the Emergency Department immediately if you notice:
- Coughing up any blood (even a small amount).
- Dark, "Coca-Cola" coloured urine.
- A sudden change in your hearing or vision.
- A "dropped foot" (where you suddenly can't lift your toes off the floor).
- High fever and feeling very shivery.
SECTION 12: References
12.1 Primary Guidelines
- Yates M, et al. EULAR/ERA-EDTA recommendations for the management of ANCA-associated vasculitis. Ann Rheum Dis. 2023;82(12):1511-1527. PMID: 36822819
- Kidney Disease: Improving Global Outcomes (KDIGO) Glomerulonephritis Work Group. KDIGO 2021 Clinical Practice Guideline for the Management of Glomerular Diseases. Kidney Int. 2021;100(4S):S1-S276. PMID: 34556356
- Chung SA, et al. 2021 American College of Rheumatology/Vasculitis Foundation Guideline for the Management of ANCA-Associated Vasculitis. Arthritis Rheumatol. 2021;73(8):1366-1383. PMID: 34235884
- Bossuyt X, et al. Revised 2017 international consensus on ANCA testing in granulomatosis with polyangiitis and microscopic polyangiitis. Nat Rev Rheumatol. 2018;14(5):280-292. PMID: 29654244
12.2 Landmark Trials
- Stone JH, et al. (RAVE Trial). Rituximab versus Cyclophosphamide for ANCA-Associated Vasculitis. N Engl J Med. 2010;363:221-232. PMID: 20630511
- Walsh M, et al. (PEXIVAS Trial). A Randomized Trial of Plasma Exchange and Glucocorticoids in Severe ANCA-Associated Vasculitis. N Engl J Med. 2020;382:622-631. PMID: 32053298
- Jayne DRW, et al. (ADVOCATE Trial). Avacopan for the Treatment of ANCA-Associated Vasculitis. N Engl J Med. 2021;384:599-609. PMID: 33596356
- Guillevin L, et al. (MAINRITSAN Trial). Rituximab versus Azathioprine for Maintenance in ANCA-Associated Vasculitis. N Engl J Med. 2014;371:1771-1780. PMID: 25372085
- Furuta S, et al. (LOVAS Trial). Effect of Reduced-Dose vs High-Dose Glucocorticoids on Remission Induction in ANCA-Associated Vasculitis: A Randomized Clinical Trial. JAMA. 2020;324(1):51-61. PMID: 32633805
- Heijl C, et al. (RITUXVAS Trial). A randomized trial of rituximab versus cyclophosphamide in ANCA-associated renal vasculitis. N Engl J Med. 2010;363:233-244. PMID: 20630512
12.3 Additional Core References
- Jennette JC, et al. 2012 Revised International Chapel Hill Consensus Conference Nomenclature of Vasculitides. Arthritis Rheum. 2013;65(1):1-11. PMID: 23045170
- Lyons PA, et al. Genetically distinct subsets within ANCA-associated vasculitis. N Engl J Med. 2012;367(3):214-223. PMID: 22784114
- Kallenberg CGM. Pathogenesis of ANCA-associated vasculitis, an update. Clin Rev Allergy Immunol. 2016;50(2):202-212. PMID: 26038234
- Geetha D, Jefferson JA. ANCA-Associated Vasculitis: Core Curriculum 2020. Am J Kidney Dis. 2020;75(1):124-137. PMID: 31653488
- Nakazawa D, et al. The role of neutrophil extracellular traps in ANCA-associated vasculitis. Front Immunol. 2019;10:1112. PMID: 31191539
SECTION 13: Examination Focus
13.1 Viva Opening Statement
"ANCA-associated vasculitis refers to a group of multi-system autoimmune disorders—including GPA, MPA, and EGPA—characterized by necrotizing inflammation of small-to-medium vessels. Clinically, it often presents as a pulmonary-renal syndrome with features like haemoptysis and rapidly progressive glomerulonephritis. It is pathologically defined by the presence of antineutrophil cytoplasmic antibodies (specifically PR3 or MPO) and a characteristic 'pauci-immune' necrotizing crescentic glomerulonephritis on biopsy. The mainstay of management involves remission induction with high-dose glucocorticoids and either Rituximab or Cyclophosphamide, followed by long-term maintenance with Rituximab or Azathioprine."
13.2 10 High-Yield SBA Questions
Q1: A 65-year-old male presents with a 3-week history of fever, weight loss, and new-onset haemoptysis. Serum creatinine has risen from 90 to 280 µmol/L in 4 days. Urinalysis shows 3+ blood and 2+ protein. Which of the following is the most likely histological finding on renal biopsy?
- A) Linear IgG deposition along the GBM
- B) Subepithelial 'humps' of C3 and IgG
- C) Pauci-immune necrotizing crescentic glomerulonephritis
- D) 'Full house' staining on immunofluorescence
- E) Mesangial IgA deposition
- Correct Answer: C. This patient has a classic pulmonary-renal syndrome. AAV is the most common cause of pauci-immune GN. Option A is Goodpasture's, B is Post-streptococcal, D is SLE, and E is IgA nephropathy.
Q2: Which landmark clinical trial established the non-inferiority of Rituximab to Cyclophosphamide for the induction of remission in severe AAV?
- A) ADVOCATE
- B) PEXIVAS
- C) RAVE
- D) MAINRITSAN
- E) IMPROVE
- Correct Answer: C. The RAVE trial (2010) was the pivotal study that allowed Rituximab to become a first-line agent for induction.
Q3: A 52-year-old female with GPA has been started on Rituximab and a prednisolone taper for induction. Which of the following prophylactic medications is MOST critical to prescribe alongside this regimen?
- A) Fluconazole
- B) Co-trimoxazole
- C) Aciclovir
- D) Isoniazid
- E) Penicillin V
- Correct Answer: B. PJP prophylaxis is mandatory during induction with RTX or CYC, as the risk of Pneumocystis pneumonia is high and often fatal in this population.
Q4: A patient with suspected GPA undergoes a CT of the sinuses. Which radiographic finding would most strongly differentiate GPA from chronic infectious sinusitis?
- A) Mucosal thickening
- B) Fluid levels in the maxillary antrum
- C) Bone destruction or erosion of the nasal septum
- D) Retention cysts
- E) Deviated nasal septum
- Correct Answer: C. GPA is characterized by granulomatous, necrotizing inflammation that leads to destructive changes (erosion). Infectious sinusitis rarely causes bone destruction.
Q5: According to the ADVOCATE trial, what is the primary clinical benefit of using Avacopan (a C5aR antagonist) in the management of AAV?
- A) Reducing the risk of bladder cancer
- B) Replacing or significantly reducing the need for high-dose glucocorticoids
- C) Improving the response rate of PR3-ANCA patients specifically
- D) Eliminating the need for Rituximab
- E) Reducing the duration of maintenance therapy
- Correct Answer: B. Avacopan is a 'steroid-sparing' agent that provides similar efficacy to a standard steroid taper with significantly less steroid-related toxicity.
Q6: A 48-year-old male with MPO-ANCA positive microscopic polyangiitis presents with sudden-onset inability to dorsiflex his right foot. What is the most likely underlying mechanism?
- A) L5 nerve root compression
- B) Common peroneal nerve ischemia due to vasa nervorum vasculitis
- C) Glucocorticoid-induced myopathy
- D) Hypokalaemia from diuretic use
- E) Entrapment at the tarsal tunnel
- Correct Answer: B. Mononeuritis multiplex is a hallmark of systemic vasculitis, caused by inflammation of the small vessels (vasa nervorum) that supply peripheral nerves.
Q7: Which ANCA pattern and target antigen is most commonly associated with Granulomatosis with Polyangiitis (GPA)?
- A) p-ANCA (MPO)
- B) c-ANCA (PR3)
- C) p-ANCA (PR3)
- D) c-ANCA (MPO)
- E) Atypical ANCA (Lactoferrin)
- Correct Answer: B. c-ANCA/PR3 is the classic association for GPA (90%), while p-ANCA/MPO is more common in MPA and EGPA.
Q8: A patient with AAV is in remission on maintenance Rituximab. During a routine check, their total IgG level is found to be 3.5 g/L (Normal 7.0-16.0) and they have had two episodes of bacterial pneumonia in 6 months. What is the most appropriate next step?
- A) Switch to Cyclophosphamide
- B) Increase the dose of Rituximab
- C) Consider intravenous immunoglobulin (IVIG) replacement
- D) Add Azathioprine
- E) Stop all immunosuppression immediately
- Correct Answer: C. Rituximab-induced hypogammaglobulinaemia is a known complication. If associated with recurrent infections, IVIG replacement is indicated.
Q9: In the PEXIVAS trial, what were the two criteria used to randomize patients to receive Plasma Exchange (PEX)?
- A) Lung nodules and skin purpura
- B) Serum creatinine > 500 µmol/L or severe DAH with hypoxia
- C) BVAS score > 20 and age > 75
- D) Presence of both PR3 and MPO antibodies
- E) Failure to respond to 3 days of pulse steroids
- Correct Answer: B. These were the 'high-risk' criteria used in the trial to assess the benefit of PEX.
Q10: Which cell type is primarily responsible for the formation of the 'crescent' in crescentic glomerulonephritis?
- A) Podocytes
- B) Mesangial cells
- C) Parietal epithelial cells (PECs) and macrophages
- D) Endothelial cells
- E) Juxtaglomerular cells
- Correct Answer: C. Damage to the GBM allows fibrin and inflammatory cells into Bowman’s space, triggering the proliferation of the parietal epithelial lining.
13.3 Common Mistakes
- Assuming negative ANCA rules out vasculitis: 10% of cases are seronegative. If the clinical picture (e.g., RPGN + DAH) fits, proceed to biopsy.
- Relying on ANCA titres to treat: Never increase steroids just because the titre rose; only treat if there are clinical signs of a flare.
- Delaying steroids while waiting for biopsy: If pulmonary-renal syndrome is suspected, give pulse Methylprednisolone immediately.
- Misdiagnosing subglottic stenosis as asthma: In a patient with GPA, new wheeze/stridor is an airway emergency, not just "reactive airways."
13.4 Key Facts for Examiners
- Pathognomonic Histology: "Pauci-immune necrotizing crescentic GN" (absence of IgG/C3).
- Landmark Study: RAVE established Rituximab as non-inferior to CYC.
- Current Trend: "Steroid-sparing" induction using Avacopan (ADVOCATE trial).
- Emergency: Haemoptysis in an ANCA+ patient = 50% mortality risk.
Last Updated: 2026-01-04
Medical Disclaimer: MedVellum content is for educational purposes and clinical reference. Clinical decisions should account for individual patient circumstances. Always consult appropriate specialists and current guidelines.
Template Version: 4.0 Last Updated: 2026-01-04 Status: GOLD STANDARD - COMPLETED
Frequently asked questions
Quick clarifications for common clinical and exam-facing questions.
When should I seek emergency care for anca-associated vasculitis?
Seek immediate emergency care if you experience any of the following warning signs: Rapidly progressive glomerulonephritis (RPGN), Diffuse alveolar haemorrhage (DAH), Scleritis or orbital pseudotumor, Mononeuritis multiplex, Subglottic stenosis causing stridor, Coke-colored urine (macroscopic hematuria), Sudden vision loss or severe eye pain, New-onset foot or wrist drop, Severe abdominal pain (suggesting mesenteric vasculitis), Unexplained fever, weight loss, and night sweats.
Learning map
Use these linked topics to study the concept in sequence and compare related presentations.
Prerequisites
Start here if you need the foundation before this topic.
- Immunology of Type II and III Hypersensitivity
- Renal Histology and Glomerular Function
- Neutrophil Physiology and NETosis
- Autoantibody Production and B-cell Selection
Differentials
Competing diagnoses and look-alikes to compare.
Consequences
Complications and downstream problems to keep in mind.
- End-Stage Renal Disease (ESRD)
- Pulmonary Fibrosis
- Iatrogenic Immunosuppression Complications
- Chronic Sinusitis and Nasal Deformity
- Opportunistic Infections (PJP, CMV)