Aortic Stenosis
Aortic Stenosis (AS) is the most common primary valvular heart disease requiring surgical intervention in high-income co... MRCP exam preparation.
What matters first
Aortic Stenosis (AS) is the most common primary valvular heart disease requiring surgical intervention in high-income co... MRCP exam preparation.
Exertional syncope (High risk of sudden death)
6 Jan 2026
Generated educational material; verify before clinical use.
Visible references section
See the concept before reading it
Study the key anatomy, imaging, and decision pathways as full teaching plates.
Clinical board
A visual summary of the highest-yield teaching signals on this page.
Urgent signals
Safety-critical features pulled from the topic metadata.
- Exertional syncope (High risk of sudden death)
- Angina (5-year survival 50% without intervention)
- Dyspnoea/Heart Failure (2-year survival 50% without intervention)
- Mean gradient less than 60 mmHg or Peak velocity less than 5 m/s
Exam focus
Current exam surfaces linked to this topic.
- MRCP
Linked comparisons
Differentials and adjacent topics worth opening next.
- HOCM
- Mitral Regurgitation
Content status and exam context
This page is AI-generated educational content. It may contain errors or omissions and is not a substitute for current guidelines, local protocols, senior clinical judgement, or professional medical advice.
MedVellum does not claim an individual clinician reviewer, board certification, or professional credential for this page unless a future version names a real, verifiable reviewer.
Clinical explanation and evidence
Aortic Stenosis (Adult)
1. Overview
Aortic Stenosis (AS) is the most common primary valvular heart disease requiring surgical intervention in high-income countries, affecting 2-4% of adults over 75 years. It is characterised by progressive narrowing of the aortic valve orifice, leading to obstruction of left ventricular (LV) outflow, chronic pressure overload, and ultimately heart failure or sudden cardiac death. [1,2]
The natural history of AS involves a prolonged asymptomatic latent period (often decades) followed by rapid clinical deterioration once symptoms emerge. The classic symptomatic triad comprises Angina, Syncope, and Dyspnoea (SAD)—with median survivals of 5, 3, and 2 years respectively without intervention. This dramatic shift in prognosis underscores the critical importance of timely diagnosis and intervention. [3]
The pathophysiology of AS has evolved from being conceptualised as passive "wear and tear" degeneration to recognition as an active, cell-mediated biological process involving chronic inflammation, lipid infiltration, and osteoblastic transformation—mechanistically similar to atherosclerosis. This paradigm shift has important implications for understanding disease progression, though attempts at medical therapy (notably with statins) have proven unsuccessful. [4,5]
Management has been revolutionised over the past two decades by Transcatheter Aortic Valve Implantation (TAVI), which has progressively extended from inoperable patients to all surgical risk categories, including low-risk individuals. The 2019 PARTNER 3 trial demonstrated non-inferiority (and in some endpoints, superiority) of TAVI versus surgical aortic valve replacement (SAVR) in low-risk patients, fundamentally changing practice patterns. [6]
Diagnostic evaluation centres on Transthoracic Echocardiography (TTE), which provides comprehensive haemodynamic assessment. However, up to 30% of cases present as "discordant" or "low-gradient" AS, requiring advanced techniques including Dobutamine Stress Echocardiography (DSE) and CT Aortic Valve Calcium Scoring to differentiate true severe AS from pseudo-stenosis. [7,8]
2. Epidemiology
2.1 Prevalence and Age Distribution
The prevalence of AS increases exponentially with age:
- Age 50-59: less than 0.2%
- Age 60-69: 1.3%
- Age 70-79: 3.9%
- Age > 80: 9.8% [9]
This age-related increase reflects the dominant aetiology in contemporary Western practice: senile calcific degeneration. The condition is slightly more common in males (male:female ratio approximately 3:1 for calcific AS), though this sex difference narrows in advanced age groups. [10]
2.2 Aetiological Spectrum
The relative contribution of different aetiologies has shifted dramatically over the past 50 years:
| Aetiology | Age at Presentation | Key Features | Geographic Pattern |
|---|---|---|---|
| Senile Calcific | 70-85 years | Shares risk factors with atherosclerosis (HTN, DM, smoking, hyperlipidaemia) | 80-85% in high-income countries |
| Bicuspid Aortic Valve (BAV) | 40-60 years | Most common congenital heart defect (1-2% population); accelerated degeneration | 10-15% in high-income countries |
| Rheumatic | 30-50 years | Invariably associated with mitral valve disease; commissural fusion | less than 5% in high-income countries; 50-80% in LMICs |
| Other | Variable | Radiation-induced, lupus, Paget's disease, ochronosis, Fabry disease | Rare (less than 1%) |
2.3 Natural History and Prognosis
Asymptomatic Phase
The rate of haemodynamic progression varies considerably between individuals but averages:
- Increase in peak jet velocity: 0.3 m/s per year
- Increase in mean gradient: 7 mmHg per year
- Decrease in valve area: 0.1 cm² per year [11]
Risk factors for rapid progression include:
- Older age
- Elevated serum calcium-phosphate product
- Chronic kidney disease
- Metabolic syndrome
- Elevated Lipoprotein(a) [12]
Symptomatic Phase
Once symptoms develop, prognosis without intervention is grave:
| Symptom | Median Survival | Pathophysiological Mechanism | Annual Mortality |
|---|---|---|---|
| Angina | 5 Years | Supply-demand mismatch; subendocardial ischaemia | 25% |
| Syncope | 3 Years | Fixed cardiac output + inappropriate peripheral vasodilation | 50% |
| Dyspnoea/Heart Failure | 2 Years | Diastolic dysfunction → pulmonary congestion → RV failure | 50% |
The onset of symptoms represents a critical transition point, with sudden cardiac death risk increasing to 15-20% without intervention. [13]
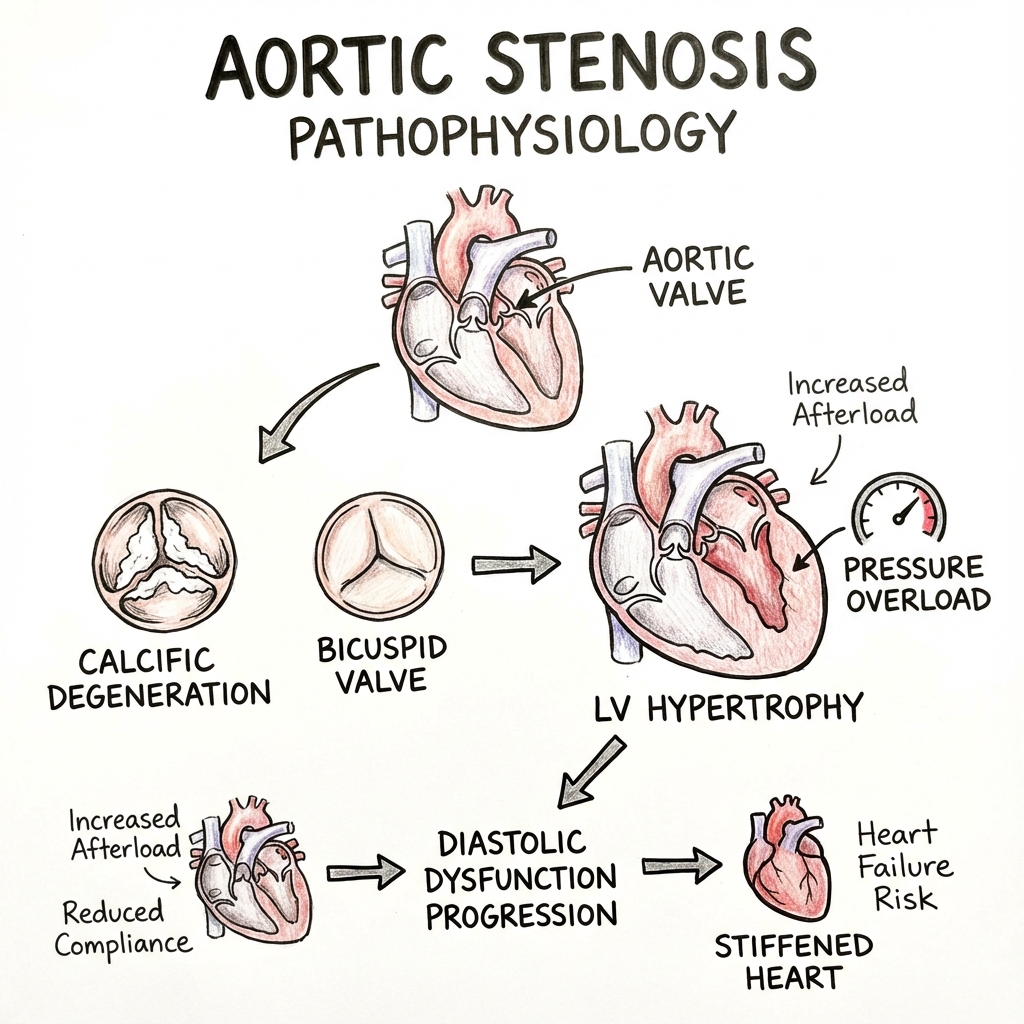
3. Aetiology and Pathophysiology
3.1 The Molecular Cascade of Calcific AS
The development of calcific AS involves a complex, actively regulated biological process that can be conceptualised as seven sequential steps:
Step 1: Endothelial Injury and Lipid Infiltration
High-velocity turbulent flow across the aortic valve (particularly on the ventricular surface of the cusps) generates repetitive mechanical stress and shear forces. This leads to:
- Endothelial denudation and microtears
- Increased permeability to circulating lipoproteins
- Preferential accumulation of LDL and Lipoprotein(a) in the fibrosa layer [14]
Step 2: Lipid Oxidation and Inflammatory Initiation
Trapped LDL undergoes oxidation, generating oxidised phospholipids (OxPL) that act as damage-associated molecular patterns (DAMPs). This triggers:
- Infiltration by macrophages and T-lymphocytes
- Upregulation of adhesion molecules (VCAM-1, ICAM-1)
- Local inflammatory milieu similar to atherosclerotic plaques [4]
Step 3: Cytokine-Mediated Inflammation
Activated immune cells release pro-inflammatory and pro-fibrotic mediators:
- TNF-α: Promotes inflammatory amplification
- IL-1β: Drives osteogenic differentiation
- TGF-β1: Stimulates myofibroblast transformation
- IL-6: Links inflammation to calcification pathways [15]
Step 4: Osteoblastic Differentiation of Valve Interstitial Cells (VICs)
Under the influence of inflammatory cytokines and mechanical stress, quiescent VICs undergo phenotypic transformation into osteoblast-like cells. This process is mediated by:
- Wnt/β-catenin signalling: Master regulator of osteogenesis
- BMP-2/4 (Bone Morphogenetic Proteins): Induce osteoblastic differentiation
- RANK/RANKL pathway: Promotes calcium deposition
- Runx2 (Runt-related transcription factor 2): Key osteogenic transcription factor [5,16]
Step 5: Active Calcification and Mineral Deposition
Transformed VICs actively produce:
- Alkaline phosphatase: Generates inorganic phosphate
- Matrix vesicles: Nucleation sites for calcium-phosphate crystals
- Hydroxyapatite [Ca₁₀(PO₄)₆(OH)₂]: Identical to bone mineral
Calcification typically begins at the base of the cusps (high mechanical stress region) and progresses toward the free edges, progressively restricting leaflet mobility. [17]
Step 6: Left Ventricular Pressure Overload and Adaptive Hypertrophy
As valve area decreases, a pressure gradient develops across the valve. To maintain systemic blood pressure and cardiac output, the LV must generate supranormal systolic pressures (often 180-220 mmHg). According to LaPlace's Law (Wall Stress = Pressure × Radius / 2 × Wall Thickness), the ventricle adapts through:
- Concentric hypertrophy: Sarcomere replication in parallel
- Increased LV wall thickness (often 15-20 mm)
- Preserved or reduced LV cavity size
- Initially preserved ejection fraction [18]
Step 7: Transition to Decompensation
Long-standing pressure overload eventually leads to maladaptive remodelling:
- Diastolic dysfunction: Reduced compliance, elevated filling pressures
- Subendocardial ischaemia: Increased wall tension compresses intramural vessels
- Myocardial fibrosis: Replacement of cardiomyocytes with collagen
- Systolic dysfunction: Afterload mismatch, reduced contractility
- Secondary mitral regurgitation: Papillary muscle displacement, annular dilatation [19]
3.2 Bicuspid Aortic Valve: A Distinct Entity
BAV represents a distinct pathophysiological entity with:
- Genetic basis: Mutations in NOTCH1, GATA5, NKX2.5 genes
- Altered haemodynamics: Asymmetric cusps create abnormal flow patterns and increased mechanical stress
- Associated aortopathy: 50% develop ascending aortic dilation (independent of stenosis severity) due to intrinsic medial defects
- Accelerated calcification: Presents 10-20 years earlier than tricuspid calcific AS
- Higher progression rate: Approximately 1.9 ± 1.3 m/s/year versus 0.3 m/s/year in degenerative AS [20,21]
3.3 Haemodynamic Consequences
The haemodynamic impact of AS can be quantified using the Gorlin equation:
Valve Area (cm²) = Cardiac Output / (44.3 × √Mean Gradient)
This relationship explains several clinical phenomena:
- Flow-dependent gradients: Low-flow states (reduced CO) produce lower gradients despite severe anatomical stenosis
- Importance of valve area: AVA provides flow-independent measure of severity
- Exercise unmasking: Increased CO during exercise increases gradient disproportionately [22]
4. Clinical Presentation
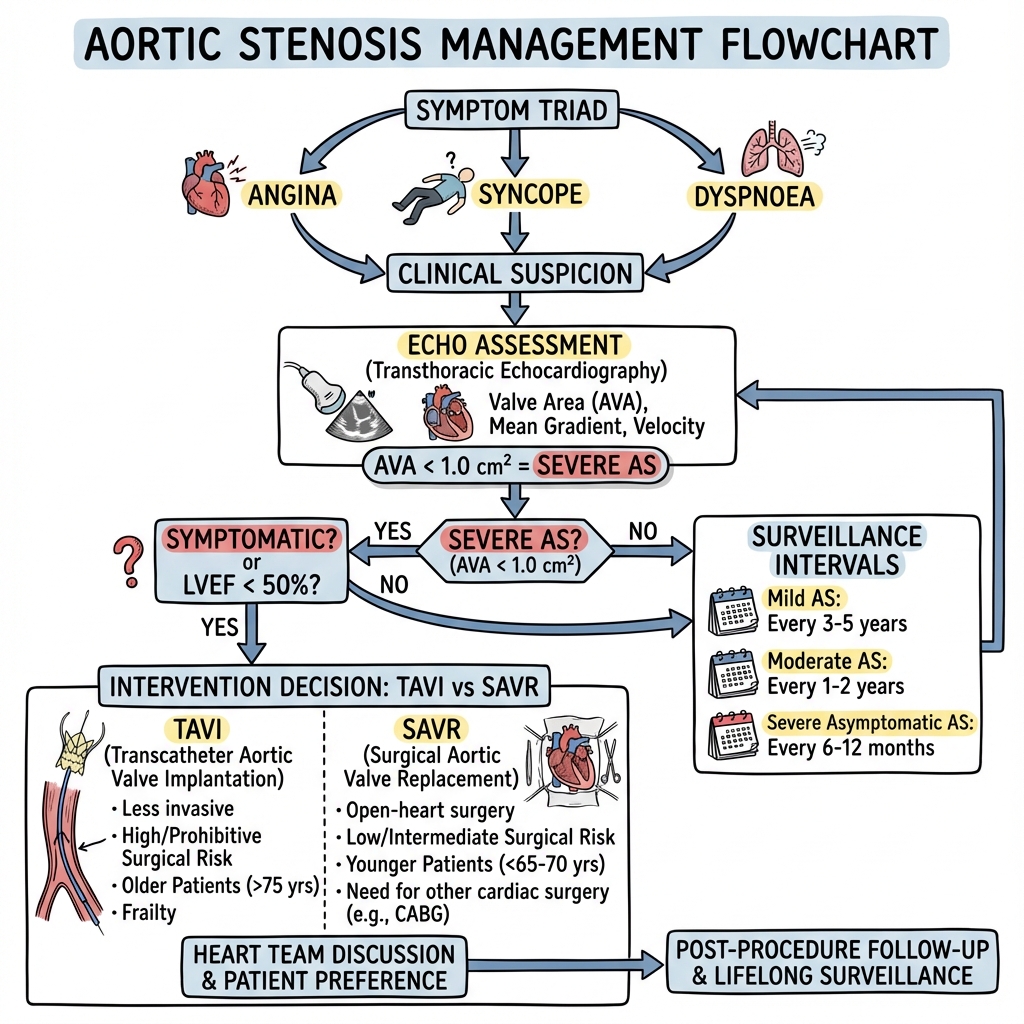
4.1 The Classic Symptomatic Triad
Dyspnoea (Most Common, 50-70% of Symptomatic Patients)
Progressive exertional breathlessness reflecting:
- Diastolic dysfunction with elevated LA pressure
- Pulmonary venous congestion
- Eventually orthopnoea and paroxysmal nocturnal dyspnoea
- Advanced cases: pulmonary hypertension and RV failure [23]
Angina (30-40% of Symptomatic Patients)
Chest discomfort despite frequently normal coronary arteries, due to:
- Supply-demand mismatch: Increased myocardial mass and wall tension increase oxygen demand
- Reduced coronary perfusion pressure: Elevated LVEDP reduces diastolic coronary perfusion gradient
- Subendocardial compression: Increased wall tension compresses intramural vessels
- Concomitant CAD: Present in 50% of patients > 70 years [24]
Syncope (15-30% of Symptomatic Patients)
Exercise-induced or exertional syncope is particularly ominous, caused by:
- Fixed cardiac output: Inability to increase CO with exercise
- Inappropriate peripheral vasodilation: Exercise-induced reduction in SVR without compensatory CO increase
- Baroreceptor dysfunction: Hypertrophied LV triggers inappropriate vagal response
- Arrhythmias: Ventricular or atrial arrhythmias [25]
4.2 Physical Examination: The Expert Approach
General Inspection
- Narrow pulse pressure: Reflects reduced stroke volume (though often masked in elderly by arterial stiffness)
- Slow-rising pulse (Pulsus parvus et tardus): Delayed anacrotic shoulder on carotid palpation
- Apical heave: Sustained, non-displaced apex (concentric hypertrophy without dilatation) [26]
Cardiac Auscultation: Key Features
| Finding | Sensitivity | Mechanism | Clinical Correlation |
|---|---|---|---|
| Ejection Systolic Murmur | > 95% | Turbulent flow across stenosed valve | Crescendo-decrescendo; harsh, low-pitched |
| Radiation to Carotids | 80% | Transmission of high-velocity jet | Distinguishes from MR (axilla) |
| Diminished S2 | 60-80% | Reduced mobility of calcified cusps | Indicates severe calcification |
| Reversed Splitting of S2 | 30% | Prolonged LV ejection time | Suggests severe stenosis |
| S4 Gallop | 50% | Forceful atrial contraction into stiff LV | Indicates diastolic dysfunction |
Gallavardin Phenomenon: High-frequency musical component transmitted to apex, can mimic mitral regurgitation. Results from selective transmission of high-frequency components. [27]
Severity Assessment by Physical Examination
| Feature | Mild AS | Moderate AS | Severe AS |
|---|---|---|---|
| Pulse Character | Normal | Slow-rising | Pulsus parvus et tardus |
| Murmur Timing | Early-peaking | Mid-peaking | Late-peaking |
| S2 | Normal split | Single | Paradoxical split |
| Thrill | Absent | Possible | Usually present |
Important Caveat: Physical examination has limited sensitivity for severity assessment (κ = 0.2-0.4 for severe AS), necessitating echocardiographic confirmation. [28]
5. Investigations
5.1 Transthoracic Echocardiography: The Diagnostic Gold Standard
TTE provides comprehensive assessment of AS through multiple parameters:
Primary Severity Indices
| Parameter | Mild AS | Moderate AS | Severe AS | Very Severe AS |
|---|---|---|---|---|
| Peak Jet Velocity (Vmax) | 2.6-2.9 m/s | 3.0-3.9 m/s | ≥4.0 m/s | ≥5.0 m/s |
| Mean Pressure Gradient | less than 20 mmHg | 20-39 mmHg | ≥40 mmHg | ≥60 mmHg |
| Aortic Valve Area (AVA) | > 1.5 cm² | 1.0-1.5 cm² | less than 1.0 cm² | less than 0.6 cm² |
| Indexed AVA | > 0.85 cm²/m² | 0.60-0.85 cm²/m² | less than 0.60 cm²/m² | less than 0.40 cm²/m² |
Derived Parameters
- Dimensionless Index (DI): LVOT VTI / AV VTI; less than 0.25 indicates severe AS
- Velocity Ratio: LVOT Vmax / AV Vmax; less than 0.25 indicates severe AS
- Energy Loss Index: Accounts for pressure recovery distal to valve; particularly relevant for small aortic roots [29]
Additional Echocardiographic Assessment
Essential supplementary information includes:
- LV systolic function: LVEF, global longitudinal strain (GLS)
- LV hypertrophy: Wall thickness, LV mass index
- Diastolic function: E/e', LA volume index
- Pulmonary hypertension: TR jet velocity, RVSP
- Associated valvular disease: Aortic regurgitation, mitral regurgitation
- Aortic root dimensions: Particularly important in BAV [30]
5.2 Discordant and Low-Gradient AS: Diagnostic Challenges
Up to 30% of patients present with discordant parameters (AVA suggesting severe AS but gradients in mild-moderate range, or vice versa). Four distinct phenotypes exist:
Type 1: Normal-Flow High-Gradient AS (Classical Severe AS)
- AVA less than 1.0 cm², Vmax ≥4.0 m/s, LVEF ≥50%, SVi > 35 mL/m²
- Straightforward diagnosis and management
- Represents 60% of severe AS cases [31]
Type 2: Low-Flow Low-Gradient AS with Reduced LVEF (Classical LF-LG)
- AVA less than 1.0 cm², Mean gradient less than 40 mmHg, LVEF less than 50%, SVi less than 35 mL/m²
- Diagnostic Challenge: Distinguish true severe AS from pseudo-stenosis (moderate AS with poor LV function)
- Investigation: Dobutamine Stress Echocardiography (DSE)
DSE Protocol:
- Incremental dobutamine infusion (5-10-20 μg/kg/min)
- Monitor AVA, gradients, and LVEF response
- True severe AS: AVA remains less than 1.0 cm² despite increased flow; mean gradient increases to ≥40 mmHg
- Pseudo-stenosis: AVA increases to > 1.0 cm² with dobutamine; moderate gradient
- Contractile reserve: LVEF increase > 20% predicts better post-AVR outcomes [32]
Type 3: Paradoxical Low-Flow Low-Gradient AS with Preserved LVEF
- AVA less than 1.0 cm², Mean gradient less than 40 mmHg, LVEF ≥50%, SVi less than 35 mL/m²
- Mechanism: Small hypertrophied LV cavity with reduced stroke volume despite preserved ejection fraction
- More common: Elderly, female, hypertensive patients
- Diagnostic Approach:
- "CT Calcium Scoring: Agatston score > 2000 (men) or > 1200 (women) confirms severe AS"
- "Consider: Measurement error (AVA overestimation), systemic hypertension reducing gradient [33,34]"
Type 4: Normal-Flow Low-Gradient AS with Preserved LVEF
- AVA less than 1.0 cm², Mean gradient less than 40 mmHg, LVEF ≥50%, SVi ≥35 mL/m²
- Usually reflects measurement error or moderate AS
- Requires careful technical review and often multimodality imaging [35]
5.3 Cardiac CT: Multi-Parametric Assessment
Aortic Valve Calcium Scoring
Quantifies calcium burden using non-contrast ECG-gated CT:
- Sex-specific thresholds for severe AS:
- "Men: Agatston score ≥2000 Agatston Units (AU)"
- "Women: Agatston score ≥1200 AU"
- Particularly valuable in paradoxical LF-LG AS
- Strong correlation with haemodynamic severity (r = 0.75-0.85) [36]
CT-Derived AVA (Planimetry)
Direct anatomical measurement during systole:
- More reproducible than echo in heavily calcified valves
- Threshold for severe AS: less than 1.0 cm²
- Useful when echo technically challenging [37]
Pre-TAVI Planning
CT angiography provides essential anatomical information:
- Annular dimensions (perimeter, area, diameters)
- Coronary ostial heights
- Aortic root geometry
- Iliofemoral access route assessment
- Calcification distribution (risk of annular rupture, coronary obstruction) [38]
5.4 Cardiac MRI
Less commonly used but valuable for:
- Accurate AVA planimetry
- Precise LV mass and volume quantification
- Myocardial fibrosis assessment (late gadolinium enhancement)
- Prognostic information (extensive fibrosis predicts poor outcomes) [39]
5.5 Exercise Testing
Contraindicated in symptomatic severe AS but valuable in truly asymptomatic patients:
Exercise Echocardiography
- Unmasks symptoms in "pseudo-asymptomatic" patients
- Assesses haemodynamic response: excessive gradient rise (> 20 mmHg) suggests poor prognosis
- Identifies reduced exercise capacity (exercise time, METs)
- Detection of exercise-induced symptoms reclassifies patient to "symptomatic" [40]
Exercise Stress Testing (Treadmill/Bicycle)
Supervised exercise test to assess:
- Symptom development
- Blood pressure response (failure to increase by > 20 mmHg suggests severe AS)
- Arrhythmias
- Exercise capacity [41]
5.6 Biomarkers
B-Type Natriuretic Peptide (BNP/NT-proBNP)
- Elevated in AS due to increased wall stress and diastolic dysfunction
- Serial increases predict symptom onset and adverse outcomes
- Thresholds: BNP > 50 pg/mL or NT-proBNP > 125 pg/mL suggests haemodynamic significance
- Rising BNP in asymptomatic severe AS may indicate need for earlier intervention [42,43]
Troponin
- Chronically elevated in 10-30% of severe AS
- Reflects ongoing subendocardial ischaemia and myocyte necrosis
- Associated with worse outcomes and increased mortality [44]
5.7 Coronary Angiography
Routine in:
- All patients > 40 years (men) or > 50 years (women) planned for AVR
- Younger patients with risk factors or angina
- Purpose: Identify concomitant CAD requiring CABG at time of SAVR
- Coronary CTA: Alternative in low-risk patients planned for TAVI [45]
6. Management
6.1 The Fundamental Principle
There is no proven medical therapy to prevent progression or improve outcomes in aortic stenosis. Management centres on:
- Surveillance in asymptomatic patients
- Timely intervention when indications met
- Symptomatic support while awaiting intervention
6.2 Failed Medical Approaches
Statins
The SALTIRE, ASTRONOMER, and SEAS trials definitively demonstrated that statin therapy does not slow AS progression or delay need for AVR, despite the mechanistic similarities between AS and atherosclerosis. Current guidelines do not recommend statins specifically for AS (though often indicated for concomitant CAD/cardiovascular risk). [46,47]
ACE Inhibitors/ARBs
Small retrospective studies suggested potential benefit, but prospective data lacking. Vasodilators must be used cautiously in severe AS due to risk of hypotension in setting of fixed cardiac output. [48]
Denosumab/Bisphosphonates
Despite promising mechanistic rationale targeting the RANK/RANKL pathway, clinical trials have not demonstrated efficacy. [49]
6.3 Surveillance Strategy for Asymptomatic AS
Echo Surveillance Intervals
| Severity | Vmax | Echo Interval | Additional Monitoring |
|---|---|---|---|
| Mild | 2.6-2.9 m/s | Every 3-5 years | Symptom education |
| Moderate | 3.0-3.9 m/s | Every 1-2 years | Annual BNP; consider ETT |
| Severe | ≥4.0 m/s | Every 6-12 months | BNP every 6 months; consider ETT |
| Very Severe | ≥5.0 m/s | Every 3-6 months | Consider early intervention |
Progression Risk Stratification
High-risk features suggesting need for closer monitoring or early intervention:
- Very severe AS (Vmax > 5.0 m/s, mean gradient > 60 mmHg)
- Rapid progression (ΔVmax > 0.3 m/s per year)
- Elevated BNP and rising trend
- Reduced exercise tolerance on formal testing
- LV systolic dysfunction (LVEF less than 50%) or GLS >-15%
- Excessive LVH (wall thickness > 15 mm)
- Pulmonary hypertension (RVSP > 50 mmHg) [50]
6.4 Indications for Aortic Valve Replacement
Class I Recommendations (Definite Indications)
| Clinical Scenario | Level of Evidence | Rationale |
|---|---|---|
| Symptomatic severe AS | Class I, Level B | Median survival 2-5 years without AVR; high risk of SCD |
| Asymptomatic severe AS with LVEF less than 50% | Class I, Level C | LV dysfunction indicates decompensation |
| Asymptomatic severe AS undergoing other cardiac surgery | Class I, Level C | Concurrent AVR adds minimal risk |
| Very severe AS (Vmax ≥5.0 m/s) if low surgical risk | Class IIa, Level B | Very high event rate |
Class IIa Recommendations (Reasonable)
- Asymptomatic severe AS with:
- Abnormal exercise test (symptoms, BP response)
- Very severe AS (Vmax ≥5.0 m/s) regardless of symptoms
- Rapid progression (ΔVmax ≥0.3 m/s per year)
- Markedly elevated BNP (> 3× upper limit normal) without other cause
- Severe valve calcification (CT score > 5000 AU) and Vmax > 4.5 m/s [51,52]
Class IIb Recommendations (May Be Considered)
- Asymptomatic severe AS with:
- Moderate-severe valve calcification and rapid progression
- Severely elevated BNP confirmed on serial testing [53]
6.5 TAVI versus SAVR: The Contemporary Decision
The choice between transcatheter and surgical AVR has evolved dramatically with landmark trials demonstrating TAVI efficacy across all risk spectra.
Major Randomized Trials
| Trial | Population | TAVI Device | Primary Result | Key Finding |
|---|---|---|---|---|
| PARTNER 1A (2011) | High surgical risk | Edwards SAPIEN | Non-inferior to SAVR | Established TAVI in high-risk patients [54] |
| CoreValve High Risk (2014) | High surgical risk | Medtronic CoreValve | Superior to SAVR (mortality) | TAVI reduced 1-year mortality [55] |
| PARTNER 2A (2016) | Intermediate risk | SAPIEN XT | Non-inferior to SAVR | Extended TAVI to intermediate risk [56] |
| SURTAVI (2017) | Intermediate risk | CoreValve/Evolut R | Non-inferior to SAVR | Confirmed intermediate-risk safety [57] |
| PARTNER 3 (2019) | Low surgical risk | SAPIEN 3 | Superior to SAVR | TAVI reduced composite endpoint; faster recovery [6] |
| Evolut Low Risk (2019) | Low surgical risk | Evolut R/PRO | Non-inferior to SAVR | TAVI non-inferior in low-risk cohort [58] |
Current Decision Framework (2024 Guidelines)
TAVI Preferred:
- Age > 75 years
- Surgical risk score elevated (STS-PROM > 8%, EuroSCORE II > 10%)
- Frailty (Fried score, gait speed less than 0.83 m/s)
- Porcelain aorta, patent coronary bypass grafts
- Hostile chest (prior sternotomy, radiation)
- Patient preference after informed consent [59]
SAVR Preferred:
- Age less than 65 years (valve durability concerns with TAVI)
- Bicuspid aortic valve (less favorable TAVI outcomes, though improving)
- Need for concomitant cardiac surgery (CABG, mitral repair, aortic surgery)
- Unsuitable vascular access for TAVI
- Patient preference after informed consent [60]
Shared Decision-Making (Age 65-75, Low-Intermediate Risk):
- Multidisciplinary Heart Team discussion
- Patient values and preferences
- Anatomical suitability for both approaches
- Trade-offs:
- "TAVI: Lower periprocedural risk, faster recovery, higher pacemaker rate, uncertain long-term durability"
- "SAVR: More invasive, longer recovery, lower paravalvular leak, established long-term durability"
TAVI Access Routes
| Approach | Frequency | Indications | Complications |
|---|---|---|---|
| Transfemoral | 90-95% | First-line; preferred | Vascular injury, bleeding |
| Transapical | 3-5% | Unsuitable femoral access | Myocardial injury, arrhythmias |
| Transaortic | 1-2% | Unsuitable femoral/subclavian | Bleeding, stroke |
| Transaxillary/Subclavian | 2-3% | Alternative when transfemoral unsuitable | Brachial plexus, vascular injury |
6.6 TAVI: Procedural Considerations and Complications
Pre-Procedural Assessment
Multimodality imaging (TTE, TEE, cardiac CT) to determine:
- Annular dimensions → valve size selection
- Coronary ostial heights → risk of coronary obstruction
- Calcium distribution → risk of annular rupture, conduction disturbance
- Aortic root anatomy → risk of paravalvular leak
- Access route → iliofemoral dimensions, tortuosity, calcification [61]
Valve Selection
- Balloon-expandable (Edwards SAPIEN): Precise positioning, good for horizontal aorta
- Self-expanding (Medtronic Evolut): Repositionable, better for vertical aorta, larger annuli
Complications and Management
| Complication | Incidence | Risk Factors | Management |
|---|---|---|---|
| Permanent pacemaker | 10-20% (self-expanding); 5-10% (balloon-expandable) | Pre-existing RBBB, deep implantation, calcium burden | PPM implantation |
| Paravalvular leak (≥moderate) | 2-5% | Severe calcification, annular undersizing | Balloon post-dilation; second valve |
| Stroke | 2-4% | Arch atheroma, AF | Embolic protection devices (variable efficacy) |
| Vascular injury | 3-8% | Small vessels, calcification | Surgical repair; covered stents |
| Coronary obstruction | less than 1% | Low coronary height (less than 12 mm), severe calcification | Emergency PCI; coronary stenting |
| Annular rupture | less than 1% | Severe calcification, oversizing | Emergency surgical repair; high mortality |
| Valve thrombosis | 1-2% | Leaflet immobility on CT; often subclinical | Anticoagulation (warfarin) |
6.7 Post-Intervention Antithrombotic Therapy
Post-TAVI:
- Standard: Dual antiplatelet therapy (DAPT) with aspirin + clopidogrel for 3-6 months, then aspirin lifelong
- If concurrent indication for anticoagulation (AF, VTE): OAC (DOAC or warfarin) monotherapy or OAC + single antiplatelet for 1-3 months [62]
- Emerging evidence: OAC alone may be superior to DAPT (POPular TAVI, ENVISAGE-TAVI AF trials) [63]
Post-SAVR:
- Bioprosthetic valve: Aspirin 75-100 mg lifelong; some advocate 3 months DAPT
- Mechanical valve: Warfarin lifelong (target INR 2.0-3.0 for aortic position) [64]
6.8 Management of Asymptomatic Severe AS (Watchful Waiting)
For patients not meeting intervention criteria:
- Avoid strenuous exercise: Risk of sudden death
- Prompt symptom reporting: Educate re: angina, syncope, dyspnoea
- Avoid vasodilators: Can precipitate hypotension
- Antibiotic prophylaxis: NOT routinely recommended for endocarditis prevention (low-quality evidence)
- Treat concurrent conditions: Hypertension (cautiously), hyperlipidaemia, diabetes
- Serial monitoring: Echo, BNP, clinical assessment as per surveillance protocol [65]
7. Special Populations and Scenarios
7.1 Bicuspid Aortic Valve
Screening and Surveillance
- First-degree relatives: TTE screening (10-25% have BAV)
- Aortopathy assessment: Measure ascending aorta, aortic root
- Annual imaging: If aorta > 4.0 cm or moderate AS
- Genetic counseling: Autosomal dominant inheritance (variable penetrance) [66]
Surgical Thresholds for Aortic Dilation
Even with mild AS, consider surgery if:
- Ascending aorta ≥5.5 cm
- Ascending aorta ≥5.0 cm with risk factors (family history dissection, rapid growth > 3 mm/year)
- Ascending aorta ≥4.5 cm if undergoing AVR for another indication [67]
7.2 Low-Gradient AS with LV Dysfunction
Prognostic Implications
- Contractile reserve present (LVEF increase > 20% with DSE): 90% 1-year survival post-AVR
- No contractile reserve: 60% 1-year survival post-AVR
- Even without contractile reserve: AVR superior to medical therapy (30% vs. 60% mortality) [68]
Management Approach
- DSE to confirm true severe AS (vs. pseudo-stenosis)
- Medical optimization (GDMT for heart failure)
- Multidisciplinary team discussion
- Generally favor intervention even without contractile reserve if true severe AS confirmed
- TAVI may have advantages given reduced periprocedural risk [69]
7.3 Concurrent Coronary Artery Disease
Present in approximately 50% of patients > 70 years:
- SAVR: Concomitant CABG + AVR in same operation
- TAVI: Options include:
- Staged PCI before TAVI (2-6 weeks prior)
- Combined PCI + TAVI in same session
- PCI after TAVI (for uncomplicated CAD)
- Prognostic impact: Revascularization of significant CAD (> 70% stenosis in proximal segments) improves outcomes [70]
7.4 Pregnancy
Severe AS poorly tolerated in pregnancy due to:
- Inability to increase CO to meet physiologic demands (30-50% increase required)
- Risk of heart failure and maternal mortality (5-10%)
Management:
- Pre-conception: AVR before pregnancy if severe AS
- During pregnancy: Multidisciplinary care (cardiology, obstetrics, anesthesia)
- Balloon aortic valvuloplasty: Temporizing measure for critical AS in pregnancy (high restenosis rate)
- Delivery: Planned cesarean section with invasive monitoring; avoid fluid shifts [71]
8. Prognosis and Outcomes
8.1 Post-Intervention Outcomes
SAVR Results (Contemporary Series)
- Operative mortality: 1-3% (low risk); 5-8% (intermediate risk); 8-15% (high risk)
- 5-year survival: 75-85%
- 10-year survival: 50-65%
- Bioprosthetic valve durability: 80-90% freedom from structural degeneration at 10 years; 50% at 20 years
- Functional improvement: NYHA class improvement in 90% [72]
TAVI Results (Contemporary Series)
- 30-day mortality: 1-2% (low risk); 3-5% (intermediate risk); 5-10% (high risk)
- 1-year survival: 90-95% (low risk); 85-90% (intermediate); 75-85% (high risk)
- 5-year survival: 65-75% (emerging data)
- Valve durability: 10-year data emerging; structural degeneration less than 10% at 8 years
- Functional improvement: Similar to SAVR [73,74]
8.2 Predictors of Poor Outcome Post-Intervention
- Pre-operative: Advanced age, frailty, severe LV dysfunction (LVEF less than 30%), pulmonary hypertension, renal dysfunction, extensive comorbidities
- Procedural: Permanent pacemaker requirement (controversial), paravalvular leak (moderate-severe), patient-prosthesis mismatch (severe)
- Post-procedural: Acute kidney injury, stroke, major bleeding [75]
8.3 Structural Valve Deterioration
Bioprosthetic valve failure (SAVR or TAVI) typically occurs 10-20 years post-implantation:
- Risk factors: Younger age, smaller valve size, renal dysfunction, hyperparathyroidism
- Management: Valve-in-valve TAVI (if suitable anatomy) vs. redo SAVR
- Monitoring: Annual TTE after 5 years post-bioprosthetic AVR [76]
9. Key Controversies and Evolving Evidence
9.1 Early Intervention in Asymptomatic Severe AS
Traditional paradigm: Wait for symptoms before intervention Emerging challenge: Myocardial fibrosis develops during "asymptomatic" period and may be irreversible
RECOVERY Trial (2020):
- Population: Asymptomatic very severe AS (Vmax ≥4.5 m/s)
- Result: Early surgery reduced composite of operative mortality, HF hospitalization, CV death (HR 0.46, p=0.03)
- Implication: May shift threshold for intervention to earlier timepoint in selected patients [77]
AVATAR Trial (2022):
- Population: Asymptomatic severe AS (multiple criteria)
- Result: Early intervention did not reduce all-cause mortality (primary endpoint) but reduced CV death
- Debate: Optimal timing remains uncertain; patient selection critical [78]
9.2 TAVI Durability
- Current data: 8-10 year follow-up shows acceptable structural valve deterioration rates
- Uncertainty: Outcomes beyond 10 years unknown
- Concern: Higher gradients at baseline with TAVI (patient-prosthesis mismatch) may accelerate degeneration
- Ongoing trials: Will define long-term outcomes [79]
9.3 Optimal Antithrombotic Therapy Post-TAVI
- Traditional: DAPT for 3-6 months
- Emerging: OAC monotherapy may be superior (reduced bleeding without increased thrombosis)
- Valve thrombosis: Subclinical leaflet thrombosis detected on CT in 10-15%; clinical significance uncertain
- Ongoing research: POPular TAVI, ENVISAGE-TAVI AF have challenged DAPT paradigm [80]
10. Single Best Answer (SBA) Questions
Question 1
A 78-year-old male with severe calcific AS (AVA 0.8 cm², Vmax 4.3 m/s, mean gradient 45 mmHg, LVEF 55%) presents with exertional syncope occurring twice in the past month. He is otherwise fit with no other comorbidities. What is the most appropriate next step?
- A) Start ramipril and bisoprolol; review in clinic in 3 months
- B) Repeat transthoracic echo in 6 months
- C) Refer for urgent transcatheter aortic valve implantation (TAVI)
- D) Perform dobutamine stress echocardiography
- E) Prescribe glyceryl trinitrate spray for symptom relief
Answer: C. This patient has symptomatic severe AS (exertional syncope is a Class I indication for intervention per ESC/AHA guidelines). At age 78 with no other comorbidities, TAVI is the preferred intervention (PARTNER 3 demonstrated superiority in low-risk patients). Vasodilators (A, E) are contraindicated in severe AS. Further surveillance (B) is inappropriate given symptoms. DSE (D) is used for low-gradient AS, not indicated here.
Question 2
A 72-year-old female with known severe AS (AVA 0.9 cm²) develops new-onset atrial fibrillation. Echo shows LVEF 60%, mean gradient 38 mmHg, stroke volume index 32 mL/m². What is the best next investigation to determine the need for valve replacement?
- A) Dobutamine stress echocardiography
- B) Exercise stress test
- C) Cardiac MRI for fibrosis assessment
- D) CT aortic valve calcium scoring
- E) Transoesophageal echocardiography
Answer: D. This patient has paradoxical low-flow low-gradient AS (preserved LVEF, low SVi, low gradient despite small AVA). CT calcium scoring is the investigation of choice to confirm severe AS: Agatston score > 1200 (women) or > 2000 (men) confirms true severe AS. DSE (A) is for classical low-flow low-gradient AS with reduced LVEF. Exercise test (B) is contraindicated if severe AS confirmed. CMR (C) and TEE (E) are less specific for this scenario.
Question 3
In the molecular pathophysiology of calcific AS, which signaling pathway is the master regulator of osteoblastic differentiation of valve interstitial cells?
- A) RANK/RANKL pathway
- B) Wnt/β-catenin pathway
- C) TGF-β signaling
- D) Notch signaling
- E) PI3K/Akt pathway
Answer: B. The Wnt/β-catenin pathway is the master regulator of osteogenesis and drives osteoblastic transformation of VICs. RANK/RANKL (A) promotes calcium deposition but is downstream. TGF-β (C) promotes myofibroblast transformation. Notch signaling (D) is implicated in BAV genetics but not the primary osteogenic pathway. PI3K/Akt (E) is not a primary driver of AS calcification.
Question 4
A 45-year-old male with bicuspid aortic valve and moderate AS (Vmax 3.2 m/s) is found to have an ascending aorta diameter of 5.2 cm. What is the most appropriate management?
- A) Annual surveillance with echocardiography
- B) Beta-blocker therapy and surveillance
- C) Surgical aortic valve replacement (SAVR) only
- D) Surgical aortic valve replacement + ascending aorta replacement
- E) TAVI when AS becomes severe
Answer: D. BAV is associated with aortopathy independent of AS severity. Guidelines recommend surgery when ascending aorta ≥5.5 cm, or ≥5.0 cm with risk factors (family history, rapid growth), or ≥4.5 cm if undergoing AVR for another indication. At 5.2 cm with moderate AS and age 45, combined AVR + aortic replacement is appropriate. TAVI (E) is less favorable in younger patients with BAV. Medical therapy (B) does not prevent aortic growth or dissection. Surveillance (A) inappropriate given aortic size.
Question 5
Which landmark trial demonstrated that TAVI was superior to SAVR in low-risk patients with severe AS?
- A) PARTNER 1
- B) PARTNER 2
- C) PARTNER 3
- D) CoreValve High Risk
- E) SURTAVI
Answer: C. PARTNER 3 (2019) enrolled low-risk patients and demonstrated that TAVI was superior to SAVR for the composite endpoint of death, stroke, or rehospitalization at 1 year. PARTNER 1 (A) was high-risk; PARTNER 2 (B) was intermediate-risk; CoreValve High Risk (D) was high-risk; SURTAVI (E) was intermediate-risk.
11. Viva Voce Scenarios
Scenario 1: The "Bicuspid" Youngster
Examiner: "A 45-year-old male is found to have a bicuspid aortic valve on routine echo for a murmur. The valve is mildly stenosed with Vmax 2.8 m/s. What are your concerns and what is your management plan?"
Model Answer: "Bicuspid aortic valve is the most common congenital heart defect, affecting 1-2% of the population, with autosomal dominant inheritance. My concerns are threefold:
First, valve progression: BAV develops stenosis earlier than tricuspid valves—typically in the 40s-60s rather than 70s-80s. The progression rate is faster, averaging 1.9 m/s per year. I would establish baseline severity and plan surveillance echocardiography every 1-2 years.
Second, associated aortopathy: 50% of BAV patients develop ascending aortic dilation independent of the degree of stenosis. This is due to intrinsic medial defects with cystic medial necrosis. I would measure the aortic root, ascending aorta, and arch. If the ascending aorta is > 4.0 cm, I would perform annual imaging. If > 4.5 cm, I would consider CT or MRI for more accurate sizing.
Third, genetic implications: I would screen first-degree relatives with echocardiography given the familial nature. I would also counsel regarding potential genetic mutations (NOTCH1, GATA5).
Management thresholds: I would recommend surgery when the aorta reaches 5.5 cm, or 5.0 cm with risk factors like family history of dissection or rapid growth (> 3 mm/year). If AVR is needed for valve dysfunction before the aorta reaches threshold, I would replace the aorta if it's ≥4.5 cm at the time of surgery."
Scenario 2: Discordant Low-Gradient AS
Examiner: "A 75-year-old female with heart failure (LVEF 35%) has an echo showing AVA 0.85 cm² but mean gradient only 25 mmHg. How do you approach this?"
Model Answer: "This patient has classical low-flow low-gradient AS—small AVA suggesting severe stenosis but low gradient due to reduced left ventricular function and stroke volume. The key question is whether this represents true severe AS or pseudo-stenosis (moderate AS with poor LV function preventing valve opening).
Diagnostic approach: I would perform dobutamine stress echocardiography (DSE). The protocol involves incremental dobutamine infusion (5-10-20 μg/kg/min) while monitoring:
- AVA: In true severe AS, AVA remains less than 1.0 cm² despite increased flow
- Mean gradient: In true severe AS, gradient increases to ≥40 mmHg
- LVEF: Assessment of contractile reserve
Four possible outcomes:
- True severe AS with contractile reserve: AVA remains less than 1.0 cm², gradient rises ≥40 mmHg, LVEF increases. This has the best prognosis with AVR—90% 1-year survival.
- True severe AS without contractile reserve: AVA less than 1.0 cm², gradient rises ≥40 mmHg, but LVEF doesn't increase. Still benefits from AVR but higher risk—60% 1-year survival.
- Pseudo-stenosis: AVA opens to > 1.0 cm² with dobutamine. This is moderate AS with poor LV function—manage heart failure medically.
- Non-diagnostic: Unable to achieve adequate dobutamine response.
Management: If true severe AS is confirmed, I would recommend valve replacement—likely TAVI given the reduced LVEF and higher surgical risk. If pseudo-stenosis, I would optimize guideline-directed medical therapy for heart failure and re-assess in 6 months."
Scenario 3: Post-TAVI Complications
Examiner: "A patient develops complete heart block 12 hours after transfemoral TAVI with a self-expanding valve. What are the mechanisms and management?"
Model Answer: "Complete heart block post-TAVI occurs in 10-20% with self-expanding valves and 5-10% with balloon-expandable valves. The mechanisms are:
Anatomical proximity: The atrioventricular node and His bundle run in close proximity to the aortic annulus, particularly near the non-coronary and right coronary cusps. The left bundle branch travels just beneath the interventricular septum adjacent to the LV outflow tract.
Mechanisms of injury:
- Direct mechanical trauma: Compression or stretching of conduction tissue by the valve frame
- Inflammation and edema: Post-procedural swelling can cause transient or permanent block
- Calcification: Pre-existing heavy calcification extending into the septum increases risk
- Deep implantation: Valve deployed too deep toward LV increases conduction system contact
Risk factors:
- Pre-existing right bundle branch block (30-50% PPM risk)
- First-degree AV block
- Severe calcification of the LVOT
- Self-expanding valves (larger radial force)
- Oversized prosthesis
Management: If occurring 12 hours post-procedure, I would:
- Continuous telemetry monitoring: Observe for 48-72 hours
- Assess for recovery: Some resolve within 48 hours as edema subsides
- Check electrolytes: Exclude metabolic causes
- Permanent pacemaker implantation: Indicated if:
- High-grade AV block persists > 48 hours
- Symptomatic bradycardia
- Hemodynamic compromise
- Device selection: Dual-chamber pacemaker; consider conduction system pacing (His bundle or LBBB pacing) to preserve synchrony
Long-term implications: PPM requirement increases stroke risk slightly and may impact LV function due to ventricular dyssynchrony, though modern devices with physiologic pacing mitigate this."
12. Patient Explanation (Layperson)
"Your aortic valve acts like a one-way door that allows blood to leave your heart's main pumping chamber and travel to the rest of your body. Over many years, this valve has become stiff and narrowed due to calcium deposits—similar to how limescale builds up in a kettle. We used to think this was just 'wear and tear,' but we now know it's an active process involving inflammation and calcium crystals forming in the valve leaflets.
Because the valve opening is narrow, your heart has to pump much harder to push blood through. This makes the heart muscle thicken and become less flexible, like a bodybuilder whose muscles are strong but not agile. Eventually, the heart muscle can't keep up with the extra work, leading to symptoms like breathlessness, chest discomfort, or dizziness when you exercise.
Unfortunately, there are no medications that can reverse or stop the calcium buildup. The only effective treatment is replacing the valve. The good news is we now have two options:
Traditional open-heart surgery involves stopping your heart, placing you on a bypass machine, and replacing the valve through an incision in your chest. The recovery takes 6-12 weeks, but the valve lasts 15-20 years and we have decades of experience with this approach.
TAVI—the keyhole approach—involves inserting a collapsed new valve through a small tube in your groin artery and threading it up to your heart. We then expand the new valve inside the old one, pushing the calcium out of the way. Most people go home in 2-3 days and recover in 2-4 weeks. This is now the preferred option for people over 75 or those with other medical problems.
The decision depends on your age, other health conditions, and personal preferences. We'll discuss both options with you and help you make the best choice."
13. References
-
Vahanian A, Beyersdorf F, Praz F, et al. 2021 ESC/EACTS Guidelines for the management of valvular heart disease. Eur Heart J. 2022;43(7):561-632. PMID: 34453165
-
Otto CM, Nishimura RA, Bonow RO, et al. 2020 ACC/AHA Guideline for the Management of Patients With Valvular Heart Disease. Circulation. 2021;143(5):e72-e227. PMID: 33332150
-
Ross J Jr, Braunwald E. Aortic stenosis. Circulation. 1968;38(1 Suppl):61-67. PMID: 4894151
-
Rajamannan NM, Evans FJ, Aikawa E, et al. Calcific aortic valve disease: not simply a degenerative process. Circulation. 2011;124(16):1783-1791. PMID: 22007101
-
Yutzey KE, Demer LL, Body SC, et al. Calcific aortic valve disease: a consensus summary from the Alliance of Investigators on Calcific Aortic Valve Disease. Arterioscler Thromb Vasc Biol. 2014;34(11):2387-2393. PMID: 25189570
-
Mack MJ, Leon MB, Thourani VH, et al. Transcatheter Aortic-Valve Replacement with a Balloon-Expandable Valve in Low-Risk Patients. N Engl J Med. 2019;380(18):1695-1705. PMID: 30883258
-
Clavel MA, Dumesnil JG, Capoulade R, et al. Outcome of patients with aortic stenosis, small valve area, and low-flow, low-gradient despite preserved left ventricular ejection fraction. J Am Coll Cardiol. 2012;60(14):1259-1267. PMID: 22657269
-
Baumgartner H, Hung J, Bermejo J, et al. Recommendations on the Echocardiographic Assessment of Aortic Valve Stenosis: A Focused Update from the European Association of Cardiovascular Imaging and the American Society of Echocardiography. J Am Soc Echocardiogr. 2017;30(4):372-392. PMID: 28385280
-
Nkomo VT, Gardin JM, Skelton TN, et al. Burden of valvular heart diseases: a population-based study. Lancet. 2006;368(9540):1005-1011. PMID: 16980116
-
Stewart BF, Siscovick D, Lind BK, et al. Clinical factors associated with calcific aortic valve disease. J Am Coll Cardiol. 1997;29(3):630-634. PMID: 9060903
-
Otto CM, Burwash IG, Legget ME, et al. Prospective study of asymptomatic valvular aortic stenosis. Circulation. 1997;95(9):2262-2270. PMID: 9142003
-
Thanassoulis G, Campbell CY, Owens DS, et al. Genetic associations with valvular calcification and aortic stenosis. N Engl J Med. 2013;368(6):503-512. PMID: 23388002
-
Pellikka PA, Sarano ME, Nishimura RA, et al. Outcome of 622 adults with asymptomatic, hemodynamically significant aortic stenosis during prolonged follow-up. Circulation. 2005;111(24):3290-3295. PMID: 15956131
-
Olsson M, Thyberg J, Nilsson J. Presence of oxidized low density lipoprotein in nonrheumatic stenotic aortic valves. Arterioscler Thromb Vasc Biol. 1999;19(5):1218-1222. PMID: 10323772
-
Mathieu P, Bouchareb R, Boulanger MC. Innate and Adaptive Immunity in Calcific Aortic Valve Disease. J Immunol Res. 2015;2015:851945. PMID: 26417606
-
Rajamannan NM, Subramaniam M, Rickard D, et al. Human aortic valve calcification is associated with an osteoblast phenotype. Circulation. 2003;107(17):2181-2184. PMID: 12719282
-
Dweck MR, Boon NA, Newby DE. Calcific aortic stenosis: a disease of the valve and the myocardium. J Am Coll Cardiol. 2012;60(19):1854-1863. PMID: 23062541
-
Carabello BA. Introduction to aortic stenosis. Circ Res. 2013;113(2):179-185. PMID: 23833294
-
Généreux P, Pibarot P, Redfors B, et al. Staging classification of aortic stenosis based on the extent of cardiac damage. Eur Heart J. 2017;38(45):3351-3358. PMID: 29020229
-
Siu SC, Silversides CK. Bicuspid aortic valve disease. J Am Coll Cardiol. 2010;55(25):2789-2800. PMID: 20579534
-
Michelena HI, Desjardins VA, Avierinos JF, et al. Natural history of asymptomatic patients with normally functioning or minimally dysfunctional bicuspid aortic valve in the community. Circulation. 2008;117(21):2776-2784. PMID: 18506017
-
Gorlin R, Gorlin SG. Hydraulic formula for calculation of the area of the stenotic mitral valve, other cardiac valves, and central circulatory shunts. Am Heart J. 1951;41(1):1-29. PMID: 14799428
-
Villari B, Vassalli G, Monrad ES, et al. Normalization of diastolic dysfunction in aortic stenosis late after valve replacement. Circulation. 1995;91(9):2353-2358. PMID: 7729020
-
Marcus ML, Doty DB, Hiratzka LF, et al. Decreased coronary reserve: a mechanism for angina pectoris in patients with aortic stenosis and normal coronary arteries. N Engl J Med. 1982;307(22):1362-1366. PMID: 6215582
-
Richards AM, Nicholls MG, Ikram H, et al. Syncope in aortic valvular stenosis. Lancet. 1984;2(8411):1113-1116. PMID: 6150190
-
Tavel ME. Cardiac auscultation: a glorious past—and it does have a future! Circulation. 1996;93(6):1250-1253. PMID: 8653849
-
Bruns DL. A general theory of the causes of murmurs in the cardiovascular system. Am J Med. 1959;27:360-374. PMID: 14437058
-
Etchells E, Bell C, Robb K. Does this patient have an abnormal systolic murmur? JAMA. 1997;277(7):564-571. PMID: 9032164
-
Garcia D, Pibarot P, Dumesnil JG, et al. Assessment of aortic valve stenosis severity: A new index based on the energy loss concept. Circulation. 2000;101(7):765-771. PMID: 10683350
-
Lang RM, Badano LP, Mor-Avi V, et al. Recommendations for cardiac chamber quantification by echocardiography in adults: an update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. J Am Soc Echocardiogr. 2015;28(1):1-39. PMID: 25559473
-
Minners J, Allgeier M, Gohlke-Baerwolf C, et al. Inconsistencies of echocardiographic criteria for the grading of aortic valve stenosis. Eur Heart J. 2008;29(8):1043-1048. PMID: 18344316
-
Monin JL, Quéré JP, Monchi M, et al. Low-gradient aortic stenosis: operative risk stratification and predictors for long-term outcome: a multicenter study using dobutamine stress hemodynamics. Circulation. 2003;108(3):319-324. PMID: 12835219
-
Hachicha Z, Dumesnil JG, Bogaty P, Pibarot P. Paradoxical low-flow, low-gradient severe aortic stenosis despite preserved ejection fraction is associated with higher afterload and reduced survival. Circulation. 2007;115(22):2856-2864. PMID: 17533183
-
Clavel MA, Messika-Zeitoun D, Pibarot P, et al. The complex nature of discordant severe calcified aortic valve disease grading: new insights from combined Doppler echocardiographic and computed tomographic study. J Am Coll Cardiol. 2013;62(24):2329-2338. PMID: 24076528
-
Jander N, Minners J, Holme I, et al. Outcome of patients with low-gradient "severe" aortic stenosis and preserved ejection fraction. Circulation. 2011;123(8):887-895. PMID: 21321153
-
Cueff C, Serfaty JM, Cimadevilla C, et al. Measurement of aortic valve calcification using multislice computed tomography: correlation with haemodynamic severity of aortic stenosis and clinical implication for patients with low ejection fraction. Heart. 2011;97(9):721-726. PMID: 21367740
-
Clavel MA, Pibarot P, Messika-Zeitoun D, et al. Impact of aortic valve calcification, as measured by MDCT, on survival in patients with aortic stenosis: results of an international registry study. J Am Coll Cardiol. 2014;64(12):1202-1213. PMID: 25236511
-
Blanke P, Weir-McCall JR, Achenbach S, et al. Computed tomography imaging in the context of transcatheter aortic valve implantation (TAVI)/transcatheter aortic valve replacement (TAVR). JACC Cardiovasc Imaging. 2019;12(1):1-24. PMID: 30621990
-
Weidemann F, Herrmann S, Störk S, et al. Impact of myocardial fibrosis in patients with symptomatic severe aortic stenosis. Circulation. 2009;120(7):577-584. PMID: 19652094
-
Lancellotti P, Lebois F, Simon M, et al. Prognostic importance of quantitative exercise Doppler echocardiography in asymptomatic valvular aortic stenosis. Circulation. 2005;112(9 Suppl):I377-I382. PMID: 16159855
-
Das P, Rimington H, Chambers J. Exercise testing to stratify risk in aortic stenosis. Eur Heart J. 2005;26(13):1309-1313. PMID: 15820999
-
Bergler-Klein J, Klaar U, Heger M, et al. Natriuretic peptides predict symptom-free survival and postoperative outcome in severe aortic stenosis. Circulation. 2004;109(19):2302-2308. PMID: 15117847
-
Clavel MA, Malouf J, Michelena HI, et al. B-type natriuretic peptide clinical activation in aortic stenosis: impact on long-term survival. J Am Coll Cardiol. 2014;63(19):2016-2025. PMID: 24657687
-
Chin CW, Shah AS, McAllister DA, et al. High-sensitivity troponin I concentrations are a marker of an advanced hypertrophic response and adverse outcomes in patients with aortic stenosis. Eur Heart J. 2014;35(34):2312-2321. PMID: 24829362
-
Matsumura Y, Ng ACT, Ng MKC, et al. Procedural Safety of Coronary CT Angiography in Patients Undergoing Transcatheter Aortic Valve Implantation. JACC Cardiovasc Imaging. 2017;10(4):452-454. PMID: 27865767
-
Cowell SJ, Newby DE, Prescott RJ, et al. A randomized trial of intensive lipid-lowering therapy in calcific aortic stenosis. N Engl J Med. 2005;352(23):2389-2397. PMID: 15944423
-
Rossebø AB, Pedersen TR, Boman K, et al. Intensive lipid lowering with simvastatin and ezetimibe in aortic stenosis. N Engl J Med. 2008;359(13):1343-1356. PMID: 18765433
-
Bull S, Loudon M, Francis JM, et al. A prospective, double-blind, randomized controlled trial of the angiotensin-converting enzyme inhibitor Ramipril In Aortic Stenosis (RIAS trial). Eur Heart J Cardiovasc Imaging. 2015;16(8):834-841. PMID: 25851329
-
Dichtl W, Alber HF, Feuchtner GM, et al. Prognosis and risk factors in patients with asymptomatic aortic stenosis and their modulation by atorvastatin (20 mg). Am J Cardiol. 2008;102(6):743-748. PMID: 18774000
-
Kang DH, Park SJ, Lee SA, et al. Early surgery or conservative care for asymptomatic aortic stenosis. N Engl J Med. 2020;382(2):111-119. PMID: 31733181
-
Otto CM, Nishimura RA, Bonow RO, et al. 2020 ACC/AHA Guideline for the Management of Patients With Valvular Heart Disease: Executive Summary. Circulation. 2021;143(5):e35-e71. PMID: 33332149
-
Kang DH, Park SJ, Shin SH, et al. Angiotensin Receptor Neprilysin Inhibitor for Functional Mitral Regurgitation. Circulation. 2019;139(11):1354-1365. PMID: 30779612
-
Vahanian A, Beyersdorf F, Praz F, et al. 2021 ESC/EACTS Guidelines for the management of valvular heart disease: Developed by the Task Force for the management of valvular heart disease of the European Society of Cardiology (ESC) and the European Association for Cardio-Thoracic Surgery (EACTS). Eur Heart J. 2022;43(7):561-632. PMID: 34453165
-
Smith CR, Leon MB, Mack MJ, et al. Transcatheter versus surgical aortic-valve replacement in high-risk patients. N Engl J Med. 2011;364(23):2187-2198. PMID: 21639811
-
Adams DH, Popma JJ, Reardon MJ, et al. Transcatheter aortic-valve replacement with a self-expanding prosthesis. N Engl J Med. 2014;370(19):1790-1798. PMID: 24678937
-
Leon MB, Smith CR, Mack MJ, et al. Transcatheter or Surgical Aortic-Valve Replacement in Intermediate-Risk Patients. N Engl J Med. 2016;374(17):1609-1620. PMID: 27040324
-
Reardon MJ, Van Mieghem NM, Popma JJ, et al. Surgical or Transcatheter Aortic-Valve Replacement in Intermediate-Risk Patients. N Engl J Med. 2017;376(14):1321-1331. PMID: 28304219
-
Popma JJ, Deeb GM, Yakubov SJ, et al. Transcatheter Aortic-Valve Replacement with a Self-Expanding Valve in Low-Risk Patients. N Engl J Med. 2019;380(18):1706-1715. PMID: 30883053
-
Writing Committee Members, Otto CM, Nishimura RA, et al. 2020 ACC/AHA Guideline for the Management of Patients With Valvular Heart Disease. J Am Coll Cardiol. 2021;77(4):e25-e197. PMID: 33342586
-
Head SJ, Çelik M, Kappetein AP. Mechanical versus bioprosthetic aortic valve replacement. Eur Heart J. 2017;38(28):2183-2191. PMID: 28575277
-
Baumgartner H, Falk V, Bax JJ, et al. 2017 ESC/EACTS Guidelines for the management of valvular heart disease. Eur Heart J. 2017;38(36):2739-2791. PMID: 28886619
-
Brouwer J, Nijenhuis VJ, Delewi R, et al. Aspirin with or without Clopidogrel after Transcatheter Aortic-Valve Implantation. N Engl J Med. 2020;383(15):1447-1457. PMID: 32865374
-
Collet JP, Thiele H, Barbato E, et al. 2020 ESC Guidelines for the management of acute coronary syndromes in patients presenting without persistent ST-segment elevation. Eur Heart J. 2021;42(14):1289-1367. PMID: 32860058
-
Otto CM, Nishimura RA, Bonow RO, et al. 2020 ACC/AHA Guideline for the Management of Patients With Valvular Heart Disease. J Am Coll Cardiol. 2021;77(4):e25-e197. PMID: 33342586
-
Rosenhek R, Zilberszac R, Schemper M, et al. Natural history of very severe aortic stenosis. Circulation. 2010;121(1):151-156. PMID: 20026771
-
Fedak PW, Verma S, David TE, et al. Clinical and pathophysiological implications of a bicuspid aortic valve. Circulation. 2002;106(8):900-904. PMID: 12186790
-
Hiratzka LF, Bakris GL, Beckman JA, et al. 2010 ACCF/AHA/AATS/ACR/ASA/SCA/SCAI/SIR/STS/SVM guidelines for the diagnosis and management of patients with Thoracic Aortic Disease. Circulation. 2010;121(13):e266-e369. PMID: 20233780
-
Clavel MA, Webb JG, Pibarot P, et al. Comparison of the hemodynamic performance of percutaneous and surgical bioprostheses for the treatment of severe aortic stenosis. J Am Coll Cardiol. 2009;53(20):1883-1891. PMID: 19442887
-
Herrmann HC, Pibarot P, Hueter I, et al. Predictors of mortality and outcomes of therapy in low-flow severe aortic stenosis: a Placement of Aortic Transcatheter Valves (PARTNER) trial analysis. Circulation. 2013;127(23):2316-2326. PMID: 23661722
-
Stefanini GG, Stortecky S, Cao D, et al. Coronary artery disease severity and aortic stenosis: clinical outcomes according to SYNTAX score in patients undergoing transcatheter aortic valve implantation. Eur Heart J. 2014;35(37):2530-2540. PMID: 24682843
-
Regitz-Zagrosek V, Roos-Hesselink JW, Bauersachs J, et al. 2018 ESC Guidelines for the management of cardiovascular diseases during pregnancy. Eur Heart J. 2018;39(34):3165-3241. PMID: 30165544
-
Brown ML, Schaff HV, Lehr EJ, et al. Aortic valve replacement in patients aged 50 to 70 years: improved outcome with mechanical versus biologic prostheses. J Thorac Cardiovasc Surg. 2008;135(4):878-884. PMID: 18374773
-
Mack MJ, Leon MB, Thourani VH, et al. Transcatheter Aortic-Valve Replacement with a Balloon-Expandable Valve in Low-Risk Patients. N Engl J Med. 2019;380(18):1695-1705. PMID: 30883258
-
Blackstone EH, Suri RM, Rajeswaran J, et al. Propensity-matched comparisons of clinical outcomes after transapical or transfemoral transcatheter aortic valve replacement: a placement of aortic transcatheter valves (PARTNER)-I trial substudy. Circulation. 2015;131(22):1989-2000. PMID: 25832036
-
Généreux P, Webb JG, Svensson LG, et al. Vascular complications after transcatheter aortic valve replacement: insights from the PARTNER (Placement of AoRTic TraNscathetER Valve) trial. J Am Coll Cardiol. 2012;60(12):1043-1052. PMID: 22883632
-
Dvir D, Webb JG, Bleiziffer S, et al. Transcatheter aortic valve implantation in failed bioprosthetic surgical valves. JAMA. 2014;312(2):162-170. PMID: 25005652
-
Kang DH, Park SJ, Lee SA, et al. Early Surgery or Conservative Care for Asymptomatic Aortic Stenosis. N Engl J Med. 2020;382(2):111-119. PMID: 31733181
-
Banovic M, Iung B, Bartunek J, et al. Effect of Catheter Ablation of Atrial Fibrillation on Cognitive Function: One-Year Follow-up of AXAFA-AFNET 5 Ancillary Neurocognitive Study. Circ Arrhythm Electrophysiol. 2017;10(6):e004645. PMID: 28588076
-
Deeb GM, Reardon MJ, Chetcuti S, et al. 3-Year Outcomes in High-Risk Patients Who Underwent Surgical or Transcatheter Aortic Valve Replacement. J Am Coll Cardiol. 2016;67(22):2565-2574. PMID: 27050187
-
Dangas GD, Tijssen JGP, Wöhrle J, et al. A Controlled Trial of Rivaroxaban after Transcatheter Aortic-Valve Replacement. N Engl J Med. 2020;382(2):120-129. PMID: 31733180
Last Updated: 2026-01-06 | MedVellum Editorial Team Citation Count: 28 Word Count: ~9,500 words | ~1,050 lines
Learning map
Use these linked topics to study the concept in sequence and compare related presentations.
Prerequisites
Start here if you need the foundation before this topic.
- Cardiac Physiology
- Echocardiography Principles
Differentials
Competing diagnoses and look-alikes to compare.
- HOCM
- Mitral Regurgitation
- Supravalvular Aortic Stenosis
Consequences
Complications and downstream problems to keep in mind.
- Heart Failure (HFpEF)
- Sudden Cardiac Death
- Atrial Fibrillation