Acute Respiratory Distress Syndrome (ARDS) - Adult
Comprehensive critical care guide to Acute Respiratory Distress Syndrome covering Berlin criteria, pathophysiology, lung-protective ventilation, prone positioning, ECMO, and evidence-based management for postgraduate...
What matters first
Comprehensive critical care guide to Acute Respiratory Distress Syndrome covering Berlin criteria, pathophysiology, lung-protective ventilation, prone positioning, ECMO, and evidence-based management for postgraduate...
Refractory hypoxaemia (P/F less than 80) despite optimised ventilation
10 Jan 2026
Generated educational material; verify before clinical use.
Visible references section
See the concept before reading it
Study the key anatomy, imaging, and decision pathways as full teaching plates.
Clinical board
A visual summary of the highest-yield teaching signals on this page.
Urgent signals
Safety-critical features pulled from the topic metadata.
- Refractory hypoxaemia (P/F less than 80) despite optimised ventilation
- Plateau pressure less than 30 cmH2O - risk of volutrauma
- Severe respiratory acidosis (pH less than 7.15) with permissive hypercapnia
- Pneumothorax or pneumomediastinum - barotrauma
Exam focus
Current exam surfaces linked to this topic.
- MRCP, FRCA, ICU training
Linked comparisons
Differentials and adjacent topics worth opening next.
- Cardiogenic Pulmonary Oedema
- Pneumonia - Severe
Content status and exam context
This page is AI-generated educational content. It may contain errors or omissions and is not a substitute for current guidelines, local protocols, senior clinical judgement, or professional medical advice.
MedVellum does not claim an individual clinician reviewer, board certification, or professional credential for this page unless a future version names a real, verifiable reviewer.
Topic family
This concept exists in multiple MedVellum libraries. Use the primary page for the broadest reference view and the others for exam-specific framing.
Comprehensive critical care guide to Acute Respiratory Distress Syndrome covering Berlin criteria, pathophysiology, lung-protective ventilation, prone positioning, ECMO, and evidence-based management for postgraduate examinations.
Comprehensive critical care guide to Acute Respiratory Distress Syndrome covering Berlin criteria, pathophysiology, lung-protective ventilation, prone positioning, ECMO, and evidence-based management for postgraduate examinations.
Clinical explanation and evidence
Acute Respiratory Distress Syndrome (ARDS) - Adult
Clinical Overview
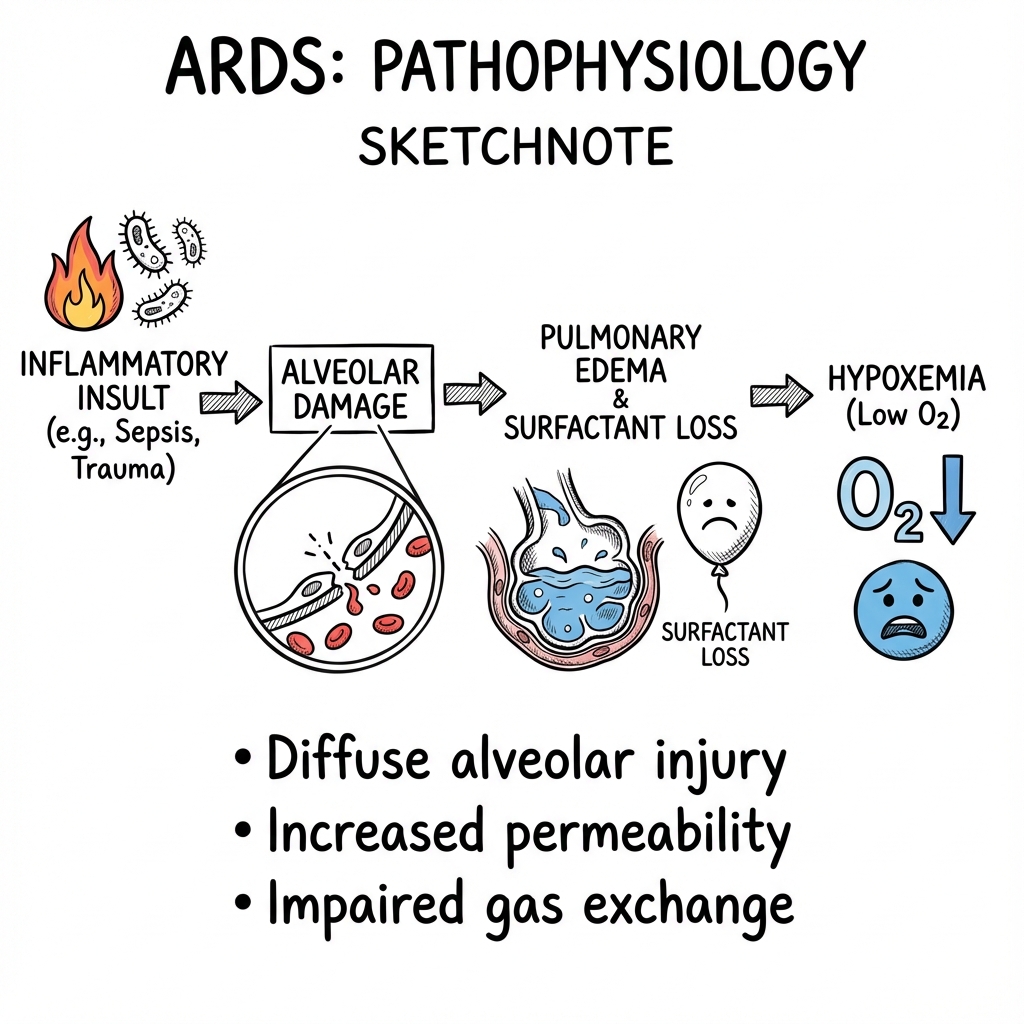
Acute Respiratory Distress Syndrome (ARDS) is a life-threatening form of acute, diffuse, inflammatory lung injury characterised by increased pulmonary vascular permeability, loss of aerated lung tissue, and severe hypoxaemic respiratory failure not fully explained by cardiac dysfunction or fluid overload. [1] Despite advances in critical care, mortality remains 30-40%, with higher rates in severe disease. [2]
ARDS represents a final common pathway of lung injury from diverse aetiologies including sepsis, pneumonia, aspiration, trauma, and pancreatitis. The syndrome is defined by the Berlin criteria (2012), which stratified ARDS into mild, moderate, and severe categories based on the degree of hypoxaemia. [1]
The cornerstone of management is lung-protective mechanical ventilation using low tidal volumes (6 mL/kg ideal body weight) to minimise ventilator-induced lung injury, combined with appropriate PEEP titration. [3] Adjunctive therapies including prone positioning, conservative fluid management, and in severe cases, venovenous extracorporeal membrane oxygenation (VV-ECMO), have demonstrated mortality benefit in randomised controlled trials. [4,5,6]
Early recognition, prompt treatment of the underlying cause, and meticulous supportive care in a high-dependency environment are essential to improve outcomes in this critically ill population.
Epidemiology
Incidence and Prevalence
| Parameter | Value | Notes |
|---|---|---|
| Global incidence | 10-80 per 100,000 person-years | Wide variation reflects diagnostic heterogeneity [2] |
| ICU prevalence | 10-15% of all ICU admissions | Higher in tertiary centres [7] |
| Mechanically ventilated patients | 23% develop ARDS | Highlights importance of preventive strategies [7] |
| COVID-19 pandemic impact | 3-5 fold increase in ARDS incidence | Predominantly severe phenotype [8] |
Mortality Rates
| ARDS Severity | In-hospital Mortality | 90-day Mortality | Key Predictors |
|---|---|---|---|
| Mild (P/F 201-300) | 27% | 30-35% | Age, APACHE II score |
| Moderate (P/F 101-200) | 32% | 35-40% | Underlying aetiology, compliance |
| Severe (P/F ≤100) | 45% | 45-52% | Multi-organ failure, ventilator days [1,2] |
Risk Factors for Development
Pulmonary (Direct Injury)
- Pneumonia (bacterial, viral, fungal) - most common pulmonary cause
- Aspiration of gastric contents (pH less than 2.5)
- Pulmonary contusion from blunt chest trauma
- Inhalational injury (smoke, toxic gases)
- Near-drowning
- Pulmonary vasculitis
Extrapulmonary (Indirect Injury)
- Sepsis - most common overall cause (30-40% of ARDS cases) [2]
- Severe acute pancreatitis
- Major trauma with shock and multiple transfusions
- Non-thoracic surgery (especially emergency procedures)
- Drug overdose (tricyclic antidepressants, aspirin, opioids)
- Transfusion-related acute lung injury (TRALI)
- Fat embolism syndrome
Long-term Outcomes in Survivors
At 12 months post-ARDS:
- 50-70% have persistent dyspnoea on exertion
- 30-50% demonstrate restrictive or mixed pulmonary function deficits
- 40-60% have reduced 6-minute walk distance
- 70-100% report decreased quality of life
- Post-intensive care syndrome (PICS) affects majority [9]
Aetiology and Pathophysiology
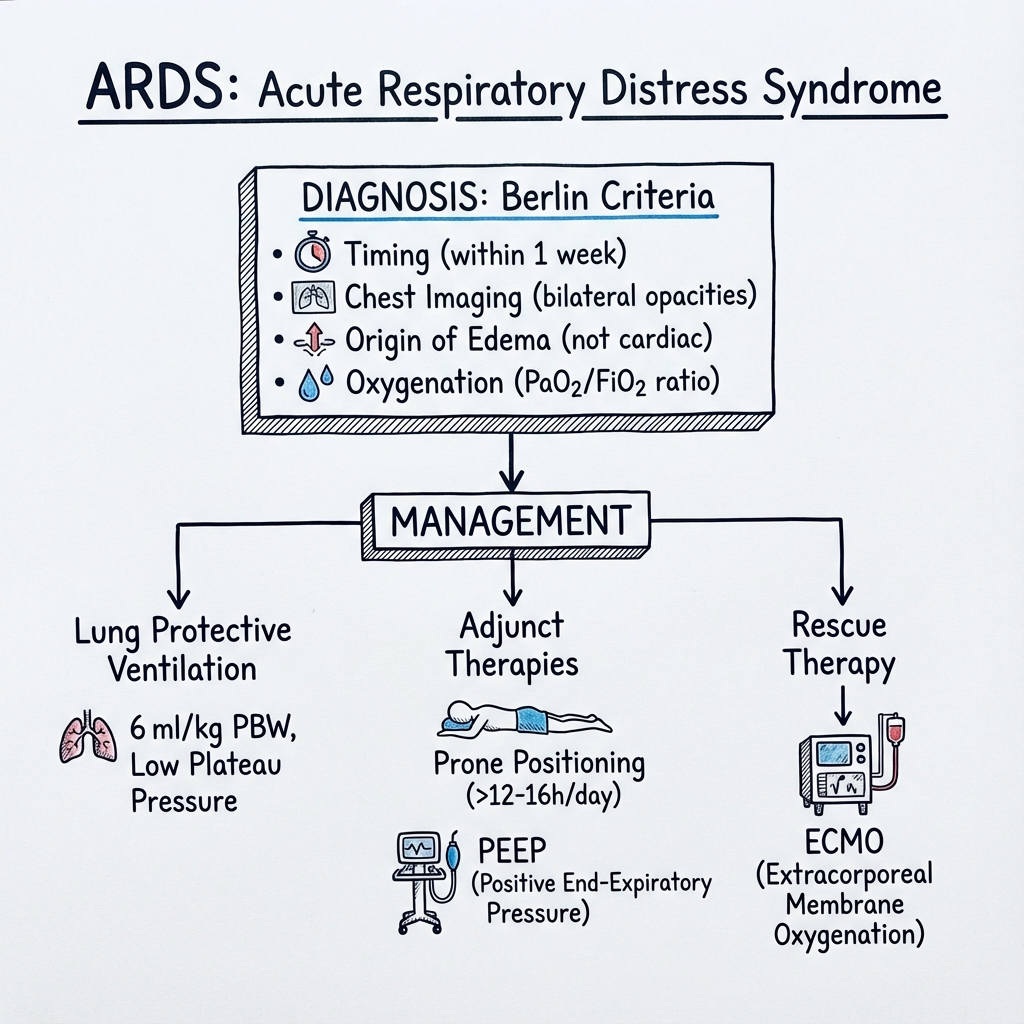
Berlin Definition of ARDS (2012)
The Berlin definition replaced the 1994 American-European Consensus Conference criteria, introducing stratification by severity and removing the pulmonary artery occlusion pressure threshold. [1]
| Criterion | Definition |
|---|---|
| Timing | Within 7 days of known clinical insult or new/worsening respiratory symptoms |
| Imaging | Bilateral opacities on chest radiograph or CT not fully explained by effusions, lobar/lung collapse, or nodules |
| Origin of oedema | Respiratory failure not fully explained by cardiac failure or fluid overload; need objective assessment (e.g. echocardiography) if no risk factor present |
| Oxygenation defect | Mild ARDS: P/F ratio 201-300 mmHg with PEEP or CPAP ≥5 cmH₂O |
| Moderate ARDS: P/F ratio 101-200 mmHg with PEEP ≥5 cmH₂O | |
| Severe ARDS: P/F ratio ≤100 mmHg with PEEP ≥5 cmH₂O |
Calculation of P/F Ratio:
P/F Ratio = PaO₂ (mmHg) ÷ FiO₂ (expressed as decimal)
Example: PaO₂ 60 mmHg on FiO₂ 0.6 → P/F = 60 ÷ 0.6 = 100 (Severe ARDS)
Clinical Pearl: ### Berlin vs. American-European Consensus Conference (AECC) Criteria
The Berlin definition improved ARDS classification by:
- Removing PCWP criterion (≤18 mmHg) - often unmeasured, replaced with "not fully explained by cardiac failure"
- Adding severity stratification - mild/moderate/severe based on P/F ratio
- Requiring minimum PEEP - P/F ratio must be measured on PEEP ≥5 cmH₂O
- Timing window - within 7 days of insult (vs. "acute onset" in AECC)
- Removing "acute lung injury" term - now "mild ARDS" instead
Validation: The Berlin definition better predicts mortality and validates treatment allocation compared to AECC criteria. [1]
Exam Detail: ### Molecular and Cellular Pathophysiology
Phase 1: Exudative Phase (Days 1-7)
Initiating Events: The pathogenesis begins with direct (pneumonia, aspiration) or indirect (sepsis, pancreatitis) injury triggering an inflammatory cascade. Pro-inflammatory cytokines (TNF-α, IL-1β, IL-6, IL-8) are released, recruiting neutrophils, macrophages, and platelets to the alveolar space. [10]
Alveolar-Capillary Membrane Disruption:
- Endothelial injury: Increased permeability leads to protein-rich fluid leaking into the interstitium and alveolar spaces
- Type I pneumocyte damage: Loss of 90% of alveolar surface area normally covered by these cells
- Surfactant dysfunction: Type II pneumocyte injury reduces surfactant production; leaked proteins inactivate existing surfactant
- Hyaline membrane formation: Fibrin, cell debris, and proteinaceous fluid coat alveolar surfaces
Consequences:
- Alveolar flooding → impaired gas exchange
- Loss of surfactant → alveolar collapse (atelectasis)
- Pulmonary oedema → reduced lung compliance ("stiff lungs")
- Intrapulmonary shunting → refractory hypoxaemia
Phase 2: Proliferative Phase (Days 7-21)
Repair Mechanisms:
- Type II pneumocyte proliferation to cover denuded basement membranes
- Fibroblast infiltration and collagen deposition
- Resolution of alveolar oedema via sodium/water transport
- Macrophage clearance of debris
Clinical Implications:
- Improvement in oxygenation if repair successful
- Risk of progression to fibrosis if inflammatory response persists
- Window for corticosteroid intervention (anti-inflammatory effect) [11]
Phase 3: Fibrotic Phase (> 3 Weeks)
Pathological Fibrosis (subset of patients):
- Extensive collagen deposition
- Obliteration of normal alveolar architecture
- Pulmonary hypertension from vascular remodelling
- Permanent restrictive lung disease
Not all patients progress to this phase; early recovery in exudative/proliferative phases is possible with optimal management.
The "Baby Lung" Concept
A fundamental concept in ARDS management is that the syndrome does not affect the lung homogeneously. [12]
Regional Heterogeneity:
- Dependent (dorsal) regions: Consolidated, non-aerated, heavy with oedema fluid
- Non-dependent (ventral) regions: Relatively preserved aeration ("baby lung")
- Recruitable middle zone: Collapsed but potentially re-expandable with PEEP
Clinical Significance:
- The aerated lung volume may be reduced to 200-500 mL (vs. normal ~6000 mL)
- Applying "normal" tidal volumes (10-12 mL/kg) to this small functional lung causes overdistension
- This regional overdistension is the primary mechanism of ventilator-induced lung injury (VILI)
CT Evidence: CT imaging in ARDS demonstrates:
- Dense consolidation in dependent (posterior) regions
- Ground-glass opacification in mid-zones (potentially recruitable)
- Relatively spared anterior lung tissue
- Distribution explains why prone positioning improves oxygenation (recruits dorsal lung)
Mechanisms of Ventilator-Induced Lung Injury (VILI)
| Type | Mechanism | Prevention Strategy |
|---|---|---|
| Volutrauma | Overdistension of alveoli from excessive tidal volumes → alveolar rupture, epithelial damage | Tidal volume 6 mL/kg IBW [3] |
| Barotrauma | High airway pressures → pneumothorax, pneumomediastinum | Plateau pressure ≤30 cmH₂O [3] |
| Atelectrauma | Repeated opening/closing of collapsed alveoli → shear stress injury | Adequate PEEP to prevent de-recruitment |
| Biotrauma | Mechanical stress → release of inflammatory mediators (IL-6, IL-8) → systemic inflammatory response | Low tidal volume + optimal PEEP |
The ARDSNet low tidal volume trial demonstrated that reducing tidal volumes from 12 mL/kg to 6 mL/kg reduced mortality from 39.8% to 31.0% (absolute risk reduction 8.8%, NNT = 11). [3]
Key Evidence:
- Volutrauma is more important than barotrauma: High volume at low pressure is more injurious than low volume at high pressure
- Stress and strain concept: Regional overdistension (high strain) causes alveolar damage even if plateau pressure less than 30 cmH₂O
- Biotrauma propagates injury: Mechanical ventilation can worsen systemic inflammation and contribute to multi-organ failure
Clinical Presentation
Symptoms
Acute Onset (within 7 days of insult):
- Dyspnoea: Rapidly progressive, severe breathlessness
- Tachypnoea: Respiratory rate often 30-50 breaths/minute
- Cough: Non-productive or minimal sputum
- Chest discomfort: Diffuse, pleuritic pain less common
- Anxiety and agitation: Secondary to severe hypoxaemia
Symptoms of underlying cause:
- Fever, rigors (pneumonia, sepsis)
- Abdominal pain (pancreatitis)
- Haemoptysis (pulmonary haemorrhage, contusion)
Signs
| System | Examination Findings | Clinical Significance |
|---|---|---|
| Respiratory | Severe tachypnoea (RR > 30/min) | Work of breathing, respiratory distress |
| Accessory muscle use (sternocleidomastoid, scalenes) | Increased work of breathing | |
| Intercostal/subcostal recession | High inspiratory effort | |
| Bilateral inspiratory crackles | Alveolar oedema, not cardiogenic | |
| Reduced air entry bilaterally | Consolidation, pleural effusions | |
| Cyanosis (late sign) | Severe hypoxaemia, SpO₂ less than 85% | |
| Cardiovascular | Tachycardia (HR 100-140 bpm) | Compensation for hypoxaemia |
| Hypotension | Sepsis, hypovolaemia, or sedation | |
| Elevated JVP (if fluid overloaded) | Distinguish from cardiogenic oedema | |
| Neurological | Confusion, agitation, drowsiness | Hypoxaemia, hypercapnia |
| Reduced GCS | Impending respiratory arrest | |
| Other | Signs of underlying aetiology | Abdominal tenderness (pancreatitis), trauma |
Clinical Course Patterns
Fulminant ARDS (20-30% of cases):
- Rapid progression within 24-48 hours
- Severe hypoxaemia from onset
- Often associated with pneumonia or sepsis
Insidious ARDS (70-80% of cases):
- Progressive dyspnoea over 3-7 days
- Gradual worsening of oxygenation
- More common with extrapulmonary causes
Differential Diagnosis
Conditions Mimicking ARDS
| Condition | Distinguishing Features | Key Investigations |
|---|---|---|
| Cardiogenic pulmonary oedema | ↑↑ BNP/NT-proBNP (> 1000 pg/mL), LV dysfunction on echo, response to diuretics, pleural effusions | Echo: ↓ EF, ↑ E/e' ratio; BNP > 1000 pg/mL |
| Bilateral pneumonia | May fulfil ARDS criteria; treat as ARDS + antimicrobials | CXR: focal consolidation; sputum/blood cultures |
| Diffuse alveolar haemorrhage | Haemoptysis, falling haemoglobin, ground-glass opacities on CT | Bronchoscopy: progressively bloodier lavage |
| Acute eosinophilic pneumonia | Peripheral eosinophilia (not always), rapid response to steroids | BAL: > 25% eosinophils |
| Cryptogenic organising pneumonia (COP) | Subacute onset (weeks), patchy peripheral consolidation, steroid-responsive | CT: peripheral/peribronchovascular consolidation |
| Pulmonary embolism | Sudden onset, pleuritic pain, risk factors, unilateral predominance | CTPA: filling defects; D-dimer elevated |
| Acute interstitial pneumonia (AIP) | Idiopathic form of acute lung injury, diagnosis of exclusion | Lung biopsy: diffuse alveolar damage (rarely done) |
ARDS vs. Cardiogenic Pulmonary Oedema
| Feature | ARDS | Cardiogenic Oedema |
|---|---|---|
| History | Recent infection, aspiration, trauma | IHD, valvular disease, heart failure |
| BNP/NT-proBNP | Normal or mildly elevated (less than 500 pg/mL) | Markedly elevated (> 1000 pg/mL) [13] |
| Echocardiography | Normal LV systolic function, E/e' less than 15 | ↓ EF or diastolic dysfunction, ↑ filling pressures |
| Response to diuretics | Minimal improvement in oxygenation | Rapid improvement (hours) |
| Chest X-ray | Bilateral, peripheral opacities; air bronchograms | Central "bat-wing" distribution; cardiomegaly |
| Protein in oedema fluid | High (exudate, protein > 0.75 g/dL) | Low (transudate, protein less than 0.5 g/dL) |
| PCWP (if PAC placed) | ≤18 mmHg | > 18 mmHg |
Important Note: Both conditions can coexist ("mixed" ARDS with cardiac contribution). Echocardiography is essential if diagnosis uncertain. [13]
Investigations
Initial Diagnostic Workup
| Investigation | Rationale | Key Findings in ARDS |
|---|---|---|
| Arterial blood gas (ABG) | Assess oxygenation (P/F ratio), ventilation, acid-base | Hypoxaemia (PaO₂ less than 80 mmHg), initially respiratory alkalosis; later respiratory acidosis |
| Chest radiograph (CXR) | Berlin criteria - bilateral opacities | Diffuse bilateral infiltrates; may appear "white-out" in severe cases |
| CT chest (if diagnosis unclear) | Higher sensitivity than CXR; assess distribution | Ground-glass opacities, consolidation (dependent), anterior sparing |
| Echocardiography (transthoracic) | Exclude cardiogenic pulmonary oedema | Normal LV systolic function; may show RV strain (pulmonary hypertension) |
| BNP or NT-proBNP | Distinguish ARDS from cardiac failure | Normal or mildly elevated in ARDS (less than 500 pg/mL) [13] |
Identify Underlying Aetiology
| Investigation | Suspected Aetiology | Additional Notes |
|---|---|---|
| Blood cultures (aerobic, anaerobic) | Sepsis, bacteraemia | Obtain before antibiotics |
| Procalcitonin | Bacterial infection (sepsis) | > 0.5 ng/mL suggests bacterial cause |
| Respiratory viral PCR panel | Influenza, RSV, COVID-19 | Nasopharyngeal swab |
| Urine legionella/pneumococcal antigen | Atypical pneumonia | Legionella more common in severe CAP |
| Serum lipase/amylase | Acute pancreatitis | Lipase > 3× ULN |
| Toxicology screen | Drug-induced ARDS | Salicylates, TCAs, opioids |
| Bronchoscopy with BAL (selected cases) | Pathogen identification, haemorrhage, eosinophils | Consider if diagnosis unclear or immunocompromised |
| HIV, autoimmune screen | Opportunistic infections, vasculitis | If risk factors present |
Severity Assessment Tools
P/F Ratio Calculation: Essential for Berlin criteria stratification and prognostication.
P/F = PaO₂ (mmHg) ÷ FiO₂ (decimal)
Must be measured on PEEP ≥5 cmH₂O
Example:
PaO₂ 70 mmHg on FiO₂ 0.8, PEEP 10 cmH₂O
P/F = 70 ÷ 0.8 = 87.5 → Severe ARDS
Murray Lung Injury Score (0-4):
- Chest X-ray score (0-4 quadrants)
- Hypoxaemia score (P/F ratio bands)
- PEEP level
- Respiratory compliance
Score ≥2.5 indicates severe acute lung injury; ≥3.0 used as ECMO referral criterion in some centres.
APACHE II / SOFA Scores: General ICU severity scores predicting mortality; not ARDS-specific but used for prognostication.
Management
1. Lung-Protective Mechanical Ventilation (Cornerstone of ARDS Management)
The ARDSNet protocol is the evidence-based standard of care, reducing mortality by 22% relative risk reduction. [3]
ARDSNet Protocol Parameters
| Parameter | Target | Rationale |
|---|---|---|
| Tidal volume (Vt) | 6 mL/kg ideal body weight (IBW) | Minimises volutrauma [3] |
| Plateau pressure (Pplat) | ≤30 cmH₂O | Prevents alveolar overdistension |
| Driving pressure (ΔP) | less than 15 cmH₂O (Pplat - PEEP) | Associated with mortality [14] |
| PEEP | Titrate per PEEP/FiO₂ table | Prevents atelectrauma, recruits lung |
| Respiratory rate | 14-35 breaths/min | Maintain pH 7.30-7.45 |
| FiO₂ | Lowest to achieve SpO₂ 88-95% | Avoid oxygen toxicity |
| pH target | 7.30-7.45 | Permissive hypercapnia acceptable if pH > 7.20 |
Calculation of Ideal Body Weight (IBW)
Critical: Use IBW, NOT actual body weight.
Males: IBW (kg) = 50 + 2.3 × (height in inches - 60)
Females: IBW (kg) = 45.5 + 2.3 × (height in inches - 60)
Metric conversion:
Height (inches) = Height (cm) ÷ 2.54
Example: Male, 175 cm tall
175 cm ÷ 2.54 = 68.9 inches
IBW = 50 + 2.3 × (68.9 - 60) = 50 + 20.5 = 70.5 kg
Target Vt = 70.5 × 6 = 423 mL (round to 420 mL)
PEEP Titration Strategies
ARDSNet Lower PEEP/Higher FiO₂ Table:
| FiO₂ | 0.3 | 0.4 | 0.5 | 0.6 | 0.7 | 0.8 | 0.9 | 1.0 |
|---|---|---|---|---|---|---|---|---|
| PEEP (cmH₂O) | 5 | 5-8 | 8-10 | 10 | 10-14 | 14 | 14-18 | 18-24 |
ARDSNet Higher PEEP/Lower FiO₂ Table (alternative):
| FiO₂ | 0.3 | 0.4 | 0.5 | 0.5-0.6 | 0.7 | 0.7-0.8 | 0.9 | 1.0 |
|---|---|---|---|---|---|---|---|---|
| PEEP (cmH₂O) | 10 | 10 | 10-14 | 14 | 14 | 16-18 | 18-20 | 20-24 |
Evidence does not clearly favour one table over the other; both are acceptable. [15] Higher PEEP may benefit patients with recruitable lung (moderate-severe ARDS).
Driving Pressure Concept: Retrospective analysis suggests targeting driving pressure (Pplat - PEEP) less than 15 cmH₂O may be superior to fixed Vt/PEEP strategies. [14] Consider adjusting PEEP to minimise driving pressure while maintaining oxygenation.
Evidence Debate: ### Higher vs. Lower PEEP: Unresolved Debate
Background: Three large RCTs (ALVEOLI, LOVS, EXPRESS) compared higher vs. lower PEEP strategies in ARDS. Meta-analysis showed no overall mortality difference, but subgroup analysis suggested benefit in moderate-severe ARDS (P/F less than 200). [15]
Lower PEEP Approach:
- Advantages: Lower risk of haemodynamic compromise, less barotrauma
- Disadvantages: More atelectasis, may worsen oxygenation
Higher PEEP Approach:
- Advantages: Better oxygenation, more lung recruitment, may reduce VILI
- Disadvantages: Risk of overdistension in non-recruitable lung, hypotension
Current Practice:
- Use ARDSNet PEEP/FiO₂ tables as starting point
- Titrate PEEP to minimise driving pressure (less than 15 cmH₂O)
- Consider higher PEEP in severe ARDS (P/F less than 150) with recruitable lung
- Individualise based on oxygenation, compliance, haemodynamics
Future Direction: Personalised PEEP titration using electrical impedance tomography (EIT) or esophageal manometry to measure transpulmonary pressure may optimise PEEP selection.
Exam Detail: #### Ventilator Settings: Step-by-Step Approach
Step 1: Intubation and Initial Settings
- Mode: Volume control (VC) or pressure control (PC) acceptable
- Set Vt = 6 mL/kg IBW
- Set RR = 20-25 breaths/min initially
- Set PEEP = 5 cmH₂O initially
- Set FiO₂ = 1.0 initially, then wean rapidly
Step 2: Obtain Plateau Pressure
- Perform inspiratory hold manoeuvre (0.5 sec)
- Measure Pplat (should be less than 30 cmH₂O)
- If Pplat > 30 cmH₂O → reduce Vt to 5 or even 4 mL/kg IBW
Step 3: Titrate PEEP and FiO₂
- Use PEEP/FiO₂ table (see above)
- Aim for SpO₂ 88-95% or PaO₂ 55-80 mmHg
- Reassess Pplat after each PEEP change
Step 4: Adjust Respiratory Rate
- Target pH 7.30-7.45
- If pH less than 7.30 → increase RR (up to 35 breaths/min)
- If pH less than 7.15 despite RR 35 → may need to increase Vt to 7-8 mL/kg (balance VILI vs. acidosis)
- Consider sodium bicarbonate infusion if severe acidosis refractory to ventilator adjustments
Step 5: Daily Monitoring
- Check ABG 30 min after any ventilator change
- Monitor Pplat, driving pressure, auto-PEEP
- Adjust settings to maintain targets
Troubleshooting Common Problems:
| Problem | Possible Causes | Solutions |
|---|---|---|
| Persistent hypoxaemia despite FiO₂ 1.0 | High shunt, atelectasis, pneumothorax | Increase PEEP, prone positioning, check for PTX, consider recruitment |
| Pplat > 30 cmH₂O | Overdistension, reduced compliance | Reduce Vt to 5-4 mL/kg, optimize PEEP, check auto-PEEP |
| Severe acidosis (pH less than 7.20) | Inadequate minute ventilation | Increase RR to 35, consider increasing Vt cautiously, bicarbonate infusion |
| Hypotension after PEEP increase | Reduced venous return | Reduce PEEP slightly, fluid bolus, vasopressors if needed |
| Auto-PEEP detected | Air trapping, inadequate expiratory time | Reduce RR, increase expiratory time, bronchodilators if bronchospasm |
2. Prone Positioning
The PROSEVA trial (2013) demonstrated that prone positioning for ≥16 hours/day in severe ARDS reduced 28-day mortality from 32.8% to 16.0% (HR 0.39, 95% CI 0.25-0.63; NNT = 6). [4]
Indications for Prone Positioning
- P/F ratio less than 150 mmHg (moderate-severe ARDS)
- FiO₂ ≥0.6 and/or PEEP ≥5 cmH₂O
- Within 36 hours of meeting criteria (early proning)
- No absolute contraindications (see below)
Contraindications (Relative)
- Spinal instability (absolute)
- Raised intracranial pressure > 30 mmHg
- Massive haemoptysis
- Recent sternotomy or open abdomen (relative; needs case-by-case assessment)
- Pregnancy (can be done with modifications)
- Severe haemodynamic instability
Prone Positioning Protocol
Preparation:
- Requires 5-6 trained staff members
- Secure all lines, tubes, catheters (ET tube, central lines, chest drains, urinary catheter)
- Pre-oxygenate with FiO₂ 1.0
- Pressure area care (padding for face, chest, pelvis, knees)
Procedure:
- Position patient supine, arms by sides
- Co-ordinated turn on clinician's count (protecting airway)
- Final prone position: "swimmer's position" (one arm up, one arm down; alternate every 2-4 hours)
- Head turned to side, supported on gel pillow
Duration:
- ≥16 hours per day (PROSEVA protocol: 16-20 hours) [4]
- Return to supine for ≤8 hours (for procedures, checks, nursing care)
- Continue daily proning sessions until improvement: P/F > 150 mmHg on FiO₂ ≤0.6 and PEEP ≤10 cmH₂O for 4 hours in supine position
Monitoring:
- Continuous SpO₂, ETCO₂, blood pressure
- Check ET tube position (chest auscultation, capnography)
- Pressure area assessment every 4 hours
- ABG 1 hour after proning
Complications:
- ET tube displacement or obstruction (most serious; requires immediate supine repositioning)
- Pressure sores (face, anterior chest, iliac crests, knees)
- Transient desaturation during turning
- Vomiting/aspiration risk
- Line/catheter dislodgement
Clinical Pearl: ### Why Does Prone Positioning Work?
Mechanism 1: Improved V/Q Matching
- In supine position, dorsal (posterior) lung is compressed by heart, abdominal contents, oedema fluid → atelectasis
- In prone position, dorsal lung expands, improving ventilation to well-perfused dependent regions
- Result: Reduced shunt fraction, improved oxygenation
Mechanism 2: More Homogeneous Pleural Pressure Distribution
- Supine: Large dorsal-to-ventral pleural pressure gradient → dependent lung collapse, non-dependent overdistension
- Prone: Smaller gradient → more uniform lung expansion
Mechanism 3: Postural Drainage
- Secretions drain from dorsal airways more effectively in prone position
- Reduces airway obstruction and infection risk
Mechanism 4: Reduced Ventral Lung Overdistension
- In supine, ventral (anterior) "baby lung" receives most tidal volume → overdistension
- In prone, ventilation distributes more evenly → less regional VILI
Clinical Evidence: PROSEVA showed prone positioning reduced mortality only in severe ARDS (P/F less than 150), suggesting benefit requires significant shunt/atelectasis to be effective. [4]
3. Neuromuscular Blockade (NMB)
Evidence is mixed: ACURASYS trial (2010) showed mortality benefit with 48-hour cisatracurium infusion in early severe ARDS, but ROSE trial (2019) did not replicate this finding with lighter sedation strategies. [16,17]
Current Recommendations
Consider NMB in:
- Severe ARDS (P/F less than 150) with ventilator dyssynchrony despite sedation optimisation
- Adjunct to prone positioning (facilitates safe turning, reduces oxygen consumption)
- Refractory hypoxaemia despite optimised ventilation
Do NOT use routinely - use selectively with careful sedation monitoring.
Protocol (if used)
- Agent: Cisatracurium 15 mg IV bolus, then 37.5 mg/hour infusion (or 0.15 mg/kg/hour)
- Duration: 48 hours maximum in early ARDS
- Monitoring: Train-of-four (TOF) monitoring to prevent excessive blockade
- Deep sedation required: Richmond Agitation-Sedation Scale (RASS) -4 to -5
- Discontinue: Wean sedation first, then stop NMB; assess for ICU-acquired weakness
4. Fluid Management
The FACTT trial (2006) demonstrated that conservative fluid strategy after initial resuscitation reduced ventilator days (14.6 vs. 12.0 days, pless than 0.001) and ICU length of stay without increasing non-pulmonary organ failures. [5]
Fluid Strategy
Phase 1: Initial Resuscitation (First 24 hours)
- Treat shock/hypoperfusion with fluid boluses and vasopressors
- Target MAP ≥65 mmHg, lactate clearance
Phase 2: De-escalation (After resuscitation)
- Switch to conservative (restrictive) fluid strategy
- Target CVP less than 4 mmHg or PAOP less than 8 mmHg (if pulmonary artery catheter placed)
- Net negative fluid balance (diuretics to achieve -500 to -1000 mL/day)
- Maintain euvolaemia: urine output > 0.5 mL/kg/hour, stable renal function
Monitoring:
- Daily weights
- Fluid balance charts
- Clinical assessment (peripheral oedema, JVP)
- Consider dynamic measures (pulse pressure variation, passive leg raise test)
Caution:
- Do not cause hypovolaemia/shock
- Monitor for acute kidney injury
- Stop diuretics if pre-renal azotaemia or haemodynamic instability
5. Corticosteroids
The DEXA-ARDS trial (2020) showed that dexamethasone reduced ventilator-free days and 60-day mortality in moderate-severe ARDS when started early (less than 24 hours of ARDS onset). [11]
Evidence-Based Protocol
Indications:
- Moderate-severe ARDS (P/F less than 200 mmHg)
- Within 24 hours of ARDS diagnosis
- No active uncontrolled infection
Regimen (DEXA-ARDS):
- Dexamethasone 20 mg IV daily for 5 days
- Then 10 mg IV daily for 5 days
- Total duration: 10 days
Contraindications:
- Active tuberculosis (unless on treatment)
- Invasive fungal infection
- Severe immunosuppression (absolute neutropenia)
Monitoring:
- Blood glucose (insulin infusion if needed)
- Superinfection surveillance
- Avoid abrupt cessation (taper if beyond 10 days)
Evidence Summary: DEXA-ARDS: Dexamethasone reduced 60-day mortality (21% vs. 36%, p=0.003) and increased ventilator-free days (12.3 vs. 7.5 days, p=0.005). [11] Earlier trials with methylprednisolone showed mixed results.
6. Recruitment Manoeuvres and High PEEP
Recruitment manoeuvres (RM): Transient increase in airway pressure (e.g. CPAP 30-40 cmH₂O for 30-40 seconds) to open collapsed alveoli.
Evidence: The ART trial (2017) found that aggressive recruitment with high PEEP increased mortality compared to conventional lung-protective ventilation (55.3% vs. 49.3%, p=0.041). [18]
Current Recommendation:
- Do NOT perform routine recruitment manoeuvres
- May consider in severe refractory hypoxaemia on case-by-case basis with extreme caution
- Risk: Barotrauma, hypotension, cardiac arrest
- Alternative: Prone positioning is safer and more effective
7. Extracorporeal Membrane Oxygenation (ECMO)
Venovenous ECMO (VV-ECMO) provides extracorporeal gas exchange, allowing ultra-protective ventilation or lung rest.
Indications (EOLIA Trial Criteria)
Consider ECMO referral if:
- P/F less than 50 mmHg for > 3 hours on optimised ventilation (FiO₂ ≥0.8, PEEP ≥10)
- P/F less than 80 mmHg for > 6 hours
- pH less than 7.25 with PaCO₂ > 60 mmHg for > 6 hours despite RR increase
- Despite optimal management: lung-protective ventilation, prone positioning, NMB
Other criteria:
- Murray Lung Injury Score ≥3.0
- Refractory hypoxaemia or hypercapnic acidosis
Contraindications
Absolute:
- Mechanical ventilation > 7 days (poor outcomes, lung fibrosis likely)
- Irreversible neurological injury
- Uncontrollable bleeding / contraindication to anticoagulation
- Metastatic malignancy, poor functional status, advanced directives against aggressive care
Relative:
- Age > 70 years (higher mortality but not absolute contraindication)
- Severe immunosuppression (case-by-case)
- Multi-organ failure (consider futility)
ECMO Circuit and Management
Circuit Components:
- Drainage cannula: Large-bore (23-25 Fr) in femoral or internal jugular vein
- Return cannula: 19-21 Fr in internal jugular or femoral vein
- Membrane oxygenator: Gas exchange occurs across semi-permeable membrane
- Pump: Centrifugal pump provides blood flow 3-6 L/min
Ventilator Settings on ECMO (Ultra-Protective "Lung Rest"):
- Vt: 3-4 mL/kg IBW
- PEEP: 10-15 cmH₂O
- Pplat: less than 25 cmH₂O
- FiO₂: 0.3-0.5 (reduced oxygen toxicity)
- RR: 10 breaths/min (allow lung rest)
Anticoagulation:
- Heparin infusion to maintain aPTT 60-80 seconds or anti-Xa 0.3-0.7 IU/mL
- Monitor for bleeding complications
- Adjust based on circuit thrombosis risk vs. bleeding
Weaning ECMO:
- Daily assessment of lung recovery
- Trial off ECMO: Reduce sweep gas flow to zero for 1-2 hours, monitor ABG
- If P/F > 150-200 on minimal ECMO support → consider decannulation
- Return to conventional ventilation with lung-protective settings
Outcomes
EOLIA trial (2018): VV-ECMO in very severe ARDS showed trend to reduced 60-day mortality (35% vs. 46%, p=0.09; not statistically significant). [6] However, Bayesian analysis and subsequent meta-analyses suggest mortality benefit, particularly in severe phenotypes.
Transfer Considerations: Early referral to ECMO centre is crucial. Mortality increases with prolonged ventilation before ECMO. Discuss with regional ECMO service when P/F less than 80 persists despite optimisation.
Exam Detail: ### ECMO Complications and Troubleshooting
Bleeding Complications (30-40%):
- Cannulation site bleeding
- GI bleeding
- Intracranial haemorrhage (5%)
- Management: Reduce anticoagulation, local haemostatic measures, transfusion
Thrombotic Complications (10-20%):
- Circuit thrombosis (requires circuit change)
- DVT/PE
- Limb ischaemia (if femoral cannulation)
- Management: Increase anticoagulation, circuit change if thrombus detected
Infection (20-30%):
- Cannula site infection
- Bloodstream infection
- VAP (still at risk despite low Vt)
- Management: Antimicrobials, cannula site care, consider cannula change
Neurological Complications (10%):
- Intracranial haemorrhage
- Ischaemic stroke
- Posterior reversible encephalopathy syndrome (PRES)
- Management: CT imaging, reduce anticoagulation if ICH
Haemolysis:
- Cause: High pump speeds, circuit thrombosis
- Markers: ↑ LDH, ↓ haptoglobin, ↑ free Hb, pink plasma
- Management: Reduce pump speed, check circuit, consider circuit change
8. Inhaled Pulmonary Vasodilators
Inhaled nitric oxide (iNO) or inhaled prostacyclin cause selective pulmonary vasodilation in ventilated lung regions, reducing shunt.
Evidence: Meta-analyses show transient improvement in oxygenation but no mortality benefit. [19] Not recommended routinely.
Consider in:
- Severe hypoxaemia refractory to other measures
- Adjunct to prone positioning or bridge to ECMO
- Right ventricular failure with pulmonary hypertension
Dose:
- iNO: 5-20 ppm
- Inhaled epoprostenol: 10,000-50,000 ng/mL nebulised continuously
9. Rescue Therapies (Severe Refractory Hypoxaemia)
| Therapy | Mechanism | Evidence Level | Comments |
|---|---|---|---|
| Prone positioning | Improved V/Q matching | ✅ Strong (RCT) | First-line rescue, NNT=6 [4] |
| ECMO | Extracorporeal gas exchange | ⚠️ Moderate (RCT) | Specialist centres, early referral [6] |
| Inhaled NO/prostacyclin | Selective pulmonary vasodilation | ❌ Weak (no mortality benefit) | Transient oxygenation improvement [19] |
| Almitrine (not UK licensed) | Pulmonary vasoconstriction in non-ventilated areas | ❌ Weak | Experimental |
| High-frequency oscillatory ventilation (HFOV) | Ultra-low tidal volumes | ❌ Harmful | Increased mortality in OSCILLATE trial; not recommended [20] |
10. Supportive Care Measures
| Measure | Target/Strategy | Rationale |
|---|---|---|
| Sedation | Light sedation (RASS -2 to 0) if tolerating ventilation | Reduces delirium, ventilator days; daily sedation holds |
| Analgesia | Fentanyl/morphine infusions; avoid over-sedation | Pain control without excessive sedation |
| Nutrition | Enteral feeding (nasogastric/post-pyloric) within 48 hours | Trophic feeds initially; full feeds by day 5-7 |
| VTE prophylaxis | LMWH (enoxaparin 40 mg SC daily) unless contraindicated | High VTE risk in critically ill; mechanical prophylaxis if bleeding risk |
| Stress ulcer prophylaxis | PPI (omeprazole 20-40 mg daily) if mechanical ventilation > 48 hours | GI bleeding prevention |
| Blood glucose control | Target 6-10 mmol/L | Avoid hypoglycaemia; insulin infusion if on steroids |
| Red cell transfusion | Restrictive strategy: transfuse if Hb less than 70 g/L | TRICC trial: restrictive strategy non-inferior [21] |
| Renal replacement therapy | Initiate if AKI stage 3 with oliguria/anuria, severe acidosis, hyperkalaemia | Continuous venovenous haemofiltration (CVVH) preferred in unstable patients |
| Antimicrobial therapy | Treat underlying infection (sepsis, pneumonia) with appropriate antibiotics | De-escalate based on cultures; avoid prolonged broad-spectrum use |
Complications
Ventilator-Associated Complications
| Complication | Incidence | Management |
|---|---|---|
| Barotrauma (pneumothorax, pneumomediastinum) | 10-15% | Chest drain insertion; reduce PEEP/Pplat if recurrent |
| Ventilator-associated pneumonia (VAP) | 20-30% | VAP bundle (head elevation, oral care, subglottic suction); targeted antibiotics |
| Oxygen toxicity | Unknown (chronic high FiO₂) | Wean FiO₂ to less than 0.6 as soon as possible |
| Tracheal stenosis | 5-10% (post-extubation) | Minimise cuff pressure (less than 25 cmH₂O); early tracheostomy if prolonged ventilation expected |
Multi-Organ Dysfunction
- Acute kidney injury (AKI): 40-50% of ARDS patients; multifactorial (sepsis, nephrotoxins, hypoperfusion)
- Cardiovascular: Shock (septic, cardiogenic), arrhythmias, myocardial dysfunction
- Hepatic dysfunction: Hypoxic hepatitis, drug-induced
- Gastrointestinal: Ileus, stress ulceration, ischaemic colitis (rare)
- Haematological: DIC in severe sepsis, thrombocytopenia
ICU-Acquired Weakness
Critical illness polyneuropathy and myopathy: 30-50% of ARDS survivors
Risk Factors:
- Prolonged immobility
- Neuromuscular blockade > 48 hours
- Corticosteroids (especially high-dose)
- Hyperglycaemia
Prevention:
- Early mobilisation (when safe)
- Avoid prolonged NMB
- Glycaemic control
Rehabilitation:
- Physiotherapy input from ICU admission
- Post-ICU multidisciplinary rehabilitation
- Recovery may take 6-12 months
Long-Term Sequelae
Post-Intensive Care Syndrome (PICS):
- Physical: Muscle wasting, weakness, reduced exercise tolerance
- Cognitive: Memory impairment, executive dysfunction, reduced concentration
- Psychological: PTSD, depression, anxiety (30-50% of survivors)
Pulmonary Sequelae:
- Restrictive lung disease (reduced FVC, TLC)
- Reduced DLCO
- Exercise-induced hypoxaemia
- Pulmonary fibrosis (minority)
Quality of Life:
- 70-100% report reduced QoL at 12 months
- Only 50% return to work by 12 months
- Persistent dyspnoea common
Follow-Up:
- Post-ICU clinic at 3 months
- Pulmonary function tests
- Mental health screening and support
- Rehabilitation referral
Prognosis
Mortality Rates
| ARDS Severity | 28-Day Mortality | 90-Day Mortality | 1-Year Mortality |
|---|---|---|---|
| Mild (P/F 201-300) | 27% | 30-35% | 35-40% |
| Moderate (P/F 101-200) | 32% | 35-40% | 40-45% |
| Severe (P/F ≤100) | 45% | 45-52% | 50-55% |
Prognostic Factors
Poor Prognosis:
- Age > 65 years
- Severe ARDS (P/F ≤100)
- Underlying aetiology: sepsis, immunosuppression
- High APACHE II/III scores
- Multi-organ failure
- Failure to improve P/F ratio by day 7
- Non-pulmonary organ dysfunction (AKI, shock)
- Chronic liver disease, chronic lung disease
Favourable Prognosis:
- Younger age
- Trauma-associated ARDS (better outcomes than sepsis-ARDS)
- Rapid improvement in oxygenation
- Single organ (pulmonary) dysfunction
Survival Trajectory
- Majority of deaths occur in first 7-14 days (early deaths from refractory hypoxaemia, multi-organ failure)
- Late deaths (> 14 days) often from nosocomial infection, multi-organ failure
- Survivors: Gradual improvement over weeks; prolonged ICU/hospital stay
- Median ventilation duration: 10-14 days in survivors
Special Populations
1. COVID-19 ARDS
COVID-19 pneumonia frequently causes severe ARDS with distinct features.
Differences from Classical ARDS:
- Initial "L-type" phenotype: Low elastance (preserved compliance), low V/Q ratio
- Later "H-type" phenotype: High elastance (stiff lungs), high shunt - resembles classical ARDS
- Marked hypoxaemia despite relatively preserved compliance initially
- High thrombotic risk (microthrombi, PE)
- Prolonged ventilation (often > 21 days)
Management Modifications:
- Dexamethasone 6 mg daily for up to 10 days (RECOVERY trial: reduced mortality) [22]
- Anticoagulation: At least prophylactic LMWH; consider therapeutic-dose in selected cases (high D-dimer, thrombosis)
- Tocilizumab or baricitinib within 24-48 hours if hypoxia + CRP > 75 mg/L (RECOVERY, REMAP-CAP trials)
- Awake proning for non-intubated patients with hypoxaemia
- Avoid delayed intubation: If work of breathing high or P/F less than 100, intubate early (self-inflicted lung injury risk)
- Lung-protective ventilation as per ARDSNet
- Prone positioning widely used (beneficial as in non-COVID ARDS)
- ECMO: Used in severe refractory cases; outcomes similar to non-COVID ARDS in specialist centres
2. Pregnancy
ARDS in pregnancy: Rare but high-risk (maternal mortality 20-40%).
Causes:
- Sepsis (chorioamnionitis, pyelonephritis)
- Aspiration
- Amniotic fluid embolism
- Pre-eclampsia/HELLP syndrome
- Peripartum cardiomyopathy (distinguish from ARDS)
Management Challenges:
- Physiological changes: ↑ minute ventilation, ↓ FRC, ↑ oxygen consumption
- Aortocaval compression in supine position (affects venous return)
- Fetal monitoring and viability considerations
Ventilation Strategy:
- Lung-protective ventilation (6 mL/kg IBW) safe in pregnancy
- Permissive hypercapnia: Accepted; fetus tolerates maternal PaCO₂ 50-60 mmHg
- Target maternal SpO₂ > 95% (higher than non-pregnant patients) to ensure fetal oxygenation
Prone Positioning:
- Can be performed with modifications (pillows under pelvis/chest to accommodate gravid uterus)
- Requires experienced team; case reports show safety and efficacy
ECMO:
- Used successfully in pregnancy; anticoagulation carefully managed
- Fetal monitoring during ECMO
Delivery Decisions:
- Multidisciplinary (obstetrics, neonatology, ICU, anaesthetics)
- If fetus viable (≥24 weeks) and maternal deterioration despite management → consider delivery
- Caesarean section under general anaesthesia (avoid spinal/epidural if coagulopathy)
3. Immunocompromised Patients
ARDS in immunocompromised hosts (HIV, haematological malignancy, transplant recipients, chemotherapy) carries higher mortality (40-60%).
Specific Considerations:
- Broad differential: Opportunistic infections (PCP, CMV, Aspergillus, TB), drug toxicity, pulmonary haemorrhage, malignant infiltration
- Bronchoscopy with BAL essential for diagnosis (high yield for pathogen identification)
- Empirical therapy: Broader antimicrobial coverage pending results
- "PCP: Co-trimoxazole 120 mg/kg/day IV + prednisolone if PaO₂ less than 70 mmHg"
- "Aspergillus: Voriconazole 6 mg/kg BD day 1, then 4 mg/kg BD"
- "CMV: Ganciclovir 5 mg/kg BD"
- Lung-protective ventilation as standard
- Avoid steroids if fungal infection suspected (unless PCP with hypoxaemia)
- Consider non-invasive ventilation early (reduce intubation risk) - but low threshold for intubation if deteriorating
4. Trauma
Pulmonary contusion may evolve into ARDS over 24-72 hours post-trauma.
Risk Factors:
- Severe chest trauma (flail chest, multiple rib fractures)
- Massive transfusion (> 10 units PRBCs in 24 hours) → TRALI, TACO
- Fat embolism syndrome (long bone fractures)
- Aspiration (reduced GCS)
Management:
- Aggressive analgesia (epidural, paravertebral blocks) to prevent splinting and atelectasis
- Early fixation of fractures to reduce inflammatory burden
- Restrictive transfusion strategy to minimise TRALI risk
- Lung-protective ventilation if intubated
- Chest physiotherapy, incentive spirometry if not intubated
Examination Focus
Exam Detail: ### High-Yield Viva Topics
1. Berlin Criteria and Severity Stratification
Viva Question: "A 60-year-old with pneumonia is on mechanical ventilation. ABG shows PaO₂ 70 mmHg on FiO₂ 0.8, PEEP 10 cmH₂O. Chest X-ray shows bilateral infiltrates. Echocardiogram shows normal LV function. Does this patient have ARDS? What severity?"
Model Answer: "Yes, this patient fulfils the Berlin criteria for ARDS:
- Timing: Acute onset within 7 days of pneumonia (known insult)
- Imaging: Bilateral opacities on CXR not explained by effusions or collapse
- Origin of oedema: Respiratory failure not explained by cardiac failure (normal LV function on echo)
- Oxygenation: P/F ratio = 70 ÷ 0.8 = 87.5 mmHg on PEEP ≥5 cmH₂O
P/F ratio 87.5 mmHg classifies this as severe ARDS (P/F ≤100). This carries 45% in-hospital mortality and warrants consideration of adjunctive therapies including prone positioning and possible ECMO referral if deteriorating despite optimal management."
2. Lung-Protective Ventilation Protocol
Viva Question: "Explain the ARDSNet protocol. Why is low tidal volume ventilation life-saving?"
Model Answer: "The ARDSNet low tidal volume protocol reduces mortality by 22% relative risk reduction (NNT=11) by minimising ventilator-induced lung injury.
Key parameters:
- Tidal volume 6 mL/kg ideal body weight - not actual weight. This is calculated using height-based formulae (males: 50 + 2.3 × [height in inches - 60])
- Plateau pressure ≤30 cmH₂O - prevents alveolar overdistension (volutrauma)
- PEEP titration per PEEP/FiO₂ table - prevents atelectrauma
- Permissive hypercapnia - accept pH 7.20-7.30 to maintain low Vt
Rationale: ARDS is heterogeneous - the 'baby lung' concept shows only 20-30% of lung is aerated. Normal tidal volumes (10-12 mL/kg) overdistend this small functional lung, causing volutrauma and releasing inflammatory mediators (biotrauma). Low Vt protects the 'baby lung' from injury, reducing mortality.
Key trial: ARMA trial (2000) - 6 mL/kg vs. 12 mL/kg: 31% vs. 39.8% mortality (p=0.007)."
3. Prone Positioning Evidence and Technique
Viva Question: "What is the evidence for prone positioning in ARDS? When would you use it and for how long?"
Model Answer: "Evidence: The PROSEVA trial (2013) demonstrated that prone positioning in severe ARDS reduced 28-day mortality from 32.8% to 16.0% (hazard ratio 0.39; NNT=6).
Indications:
- Severe ARDS: P/F less than 150 mmHg on FiO₂ ≥0.6 and PEEP ≥5 cmH₂O
- Within 36 hours of meeting criteria (early proning)
Duration:
- At least 16 hours per session (PROSEVA used 16-20 hours)
- Return to supine for ≤8 hours
- Continue daily sessions until sustained improvement: P/F > 150 on FiO₂ ≤0.6 and PEEP ≤10 for 4 hours in supine
Mechanism:
- Improves V/Q matching (dorsal lung recruited, drainage of secretions)
- Reduces dependent atelectasis
- More homogeneous pleural pressure distribution
- Reduces ventral lung overdistension
Contraindications:
- Spinal instability (absolute)
- Raised ICP > 30 mmHg
- Massive haemoptysis
- Severe haemodynamic instability (relative)
Complications:
- ET tube displacement (most serious)
- Pressure sores (face, chest, knees)
- Transient desaturation during turning
Procedure: Requires 5-6 trained staff, careful securing of ET tube and lines, padding of pressure areas, 'swimmer's position' with head turned."
4. ECMO Indications and Timing
Viva Question: "A patient with severe ARDS has P/F ratio 60 mmHg on FiO₂ 1.0 and PEEP 16 cmH₂O, despite prone positioning and lung-protective ventilation. What are your next steps?"
Model Answer: "This patient has severe refractory hypoxaemia and meets criteria for ECMO consideration.
Immediate actions:
- Ensure optimal management: Verify lung-protective ventilation (Vt 6 mL/kg IBW, Pplat less than 30), adequate PEEP, prone positioning ≥16 hours, neuromuscular blockade if dyssynchronous
- Check driving pressure (aim less than 15 cmH₂O)
- Ensure conservative fluid strategy
- Exclude correctable causes (pneumothorax, ET tube malposition, mucus plugging)
ECMO referral criteria (EOLIA trial):
- P/F less than 50 mmHg for > 3 hours, OR
- P/F less than 80 mmHg for > 6 hours, OR
- pH less than 7.25 with PaCO₂ > 60 mmHg for > 6 hours
This patient meets ECMO criteria. I would:
- Contact regional ECMO centre urgently for discussion
- Continue optimisation while awaiting response
- Ensure patient has not been ventilated > 7 days (relative contraindication)
- Assess for absolute contraindications (irreversible neurological injury, contraindication to anticoagulation, metastatic malignancy)
Evidence: EOLIA trial showed trend to reduced mortality (35% vs. 46%, p=0.09; Bayesian analysis and meta-analyses suggest benefit in carefully selected severe cases). Early referral is crucial as outcomes worsen with prolonged conventional ventilation before ECMO."
5. Conservative Fluid Strategy
Viva Question: "How do you manage fluids in a patient with ARDS after initial resuscitation?"
Model Answer: "The FACTT trial (2006) demonstrated that conservative fluid management after initial resuscitation reduces ventilator days (12.0 vs. 14.6 days, pless than 0.001) and ICU stay without increasing non-pulmonary organ failure or mortality.
Approach:
Phase 1: Initial resuscitation (first 24 hours)
- Treat shock with fluid boluses and vasopressors
- Target MAP ≥65 mmHg, lactate clearance, adequate perfusion
Phase 2: Conservative strategy (after resuscitation)
- Target CVP less than 4 mmHg or PAOP less than 8 mmHg (if PA catheter in situ)
- Achieve net negative fluid balance (-500 to -1000 mL/day)
- Use diuretics (e.g. furosemide 20-40 mg IV boluses or infusion)
Monitoring:
- Daily weights
- Urine output (target > 0.5 mL/kg/hour)
- Serum creatinine (watch for pre-renal AKI)
- Clinical assessment (peripheral oedema, JVP)
- Haemodynamic stability (avoid hypovolaemia)
Rationale: Positive fluid balance worsens pulmonary oedema, impairs oxygenation, and prolongs mechanical ventilation. Conservative strategy improves lung water clearance.
Caution: Do not cause hypovolaemia - balance between minimising pulmonary oedema and maintaining organ perfusion. Stop diuretics if acute kidney injury or haemodynamic instability develops."
6. Steroids in ARDS
Viva Question: "What is the role of corticosteroids in ARDS?"
Model Answer: "Corticosteroids in ARDS have been studied for decades with conflicting results. The most recent evidence supports early dexamethasone in moderate-severe ARDS.
Current Evidence: DEXA-ARDS trial (2020): Dexamethasone started within 24 hours of ARDS onset in moderate-severe cases reduced 60-day mortality (21% vs. 36%, p=0.003) and increased ventilator-free days (12.3 vs. 7.5 days, p=0.005).
Recommended Protocol:
- Indications: Moderate-severe ARDS (P/F less than 200 mmHg) within 24 hours of diagnosis
- Regimen: Dexamethasone 20 mg IV daily for 5 days, then 10 mg IV daily for 5 days (total 10 days)
- Contraindications: Active uncontrolled infection (TB, invasive fungal), severe immunosuppression
Monitoring:
- Blood glucose control (insulin infusion if needed, especially common with steroids)
- Surveillance for superinfection (VAP, fungal)
- Avoid abrupt cessation after 10 days (taper if continuing)
Mechanism: Anti-inflammatory effect reduces alveolar inflammation, improves alveolar-capillary membrane integrity, and may reduce progression to fibrotic phase.
Historical Context: Earlier trials with methylprednisolone in late ARDS or high-dose steroids showed no benefit or harm. DEXA-ARDS used early, moderate-dose dexamethasone and is the current standard."
7. Differentiating ARDS from Cardiogenic Pulmonary Oedema
Viva Question: "A 70-year-old with bilateral infiltrates on CXR and P/F ratio 120. How do you differentiate ARDS from cardiogenic pulmonary oedema?"
Model Answer: "Both conditions can present with bilateral infiltrates and hypoxaemia, and differentiation is critical as management differs.
Diagnostic Approach:
| Feature | ARDS | Cardiogenic Oedema |
|---|---|---|
| History | Sepsis, aspiration, trauma | IHD, heart failure history |
| BNP/NT-proBNP | Normal or mildly elevated (less than 500 pg/mL) | Markedly elevated (> 1000 pg/mL) |
| Echocardiography | Normal LV systolic function, E/e' less than 15 | Reduced EF or diastolic dysfunction, ↑ E/e' |
| Response to diuretics | Minimal improvement | Rapid improvement (hours) |
| CXR pattern | Peripheral, bilateral opacities; air bronchograms | Central 'bat-wing'; cardiomegaly; pleural effusions |
| PCWP (if PAC) | ≤18 mmHg | > 18 mmHg |
Key Investigations:
- BNP or NT-proBNP: Single most useful test - BNP > 1000 pg/mL suggests cardiogenic cause
- Echocardiography: Assess LV systolic/diastolic function, wall motion abnormalities, valvular disease
- Trial of diuretics: Rapid improvement suggests cardiogenic oedema
Important Notes:
- Both conditions can coexist ('mixed' picture)
- Chronic kidney disease elevates BNP
- Sepsis can cause myocardial depression
- When uncertain, echo is essential - treat based on findings
In this case, I would perform urgent echo and BNP. If LV function normal and BNP less than 500, manage as ARDS; if reduced EF or BNP > 1000, treat cardiac component with diuretics while providing ventilatory support."
OSCE Scenarios
Scenario 1: Ventilator Settings "Calculate appropriate initial ventilator settings for a 68 kg male, 172 cm tall, with severe ARDS."
Answer:
Height 172 cm = 67.7 inches
IBW = 50 + 2.3 × (67.7 - 60) = 50 + 17.7 = 67.7 kg
Settings:
- Mode: Volume control
- Tidal volume: 67.7 × 6 = 406 mL (set 400 mL)
- RR: 20-25 breaths/min
- PEEP: 10 cmH₂O (start, then titrate per PEEP/FiO₂ table)
- FiO₂: 1.0 initially (wean rapidly to target SpO₂ 88-95%)
- Check plateau pressure less than 30 cmH₂O after 30 min
Scenario 2: Recognising ECMO Criteria "A patient has been prone for 48 hours, on Vt 6 mL/kg, PEEP 16, FiO₂ 1.0, paralysed. P/F ratio 55 mmHg. What do you do?"
Answer: "This patient meets ECMO criteria (P/F less than 80 for > 6 hours). I would:
- Verify optimal management (check driving pressure, ensure adequate proning duration, exclude reversible causes)
- Contact regional ECMO centre urgently
- Prepare for possible transfer
- Ensure patient less than 7 days ventilation and no absolute contraindications
- Continue current management while awaiting ECMO team response"
Scenario 3: Managing Refractory Hypoxaemia "A patient with severe ARDS on 6 mL/kg tidal volume, PEEP 14, FiO₂ 0.9 has SpO₂ 82%. Plateau pressure is 28 cmH₂O. What are your options?"
Answer:
Systematic approach:
1. Verify current ventilation is optimal:
- Tidal volume: 6 mL/kg IBW ✓
- Plateau pressure: 28 cmH₂O ✓ (less than 30)
- Driving pressure: Calculate (Pplat - PEEP = 28 - 14 = 14 cmH₂O) ✓ (less than 15)
2. Check for reversible causes:
- Pneumothorax (examination, CXR)
- ET tube malposition (capnography, auscultation)
- Mucus plugging (suction, bronchoscopy if needed)
- Right mainstem intubation
- Auto-PEEP (expiratory hold)
3. Escalate PEEP and FiO₂:
- Increase FiO₂ to 1.0
- Increase PEEP to 16-18 cmH₂O (per ARDSNet high PEEP table)
- Recheck plateau pressure after PEEP increase
4. Consider adjunctive therapies (in order):
- **Prone positioning** (first-line if P/F less than 150) - NNT=6
- Neuromuscular blockade if ventilator dyssynchrony
- Inhaled pulmonary vasodilators (iNO 10-20 ppm or inhaled epoprostenol)
- Contact ECMO centre if P/F remains less than 80 for > 6 hours
5. Optimize other factors:
- Conservative fluid strategy (aim CVP less than 4 mmHg)
- Treat underlying cause (antibiotics for pneumonia/sepsis)
- Consider dexamethasone if not already started
Scenario 4: Ventilator Dyssynchrony "An ARDS patient on volume control ventilation is 'fighting the ventilator' despite sedation. What do you do?"
Answer:
Step 1: Assess the patient
- Examine: respiratory distress, tachypnoea, accessory muscle use
- Check ventilator graphics: flow-time, pressure-time curves
- ABG: assess oxygenation, ventilation, acidosis
Step 2: Identify cause of dyssynchrony
Common causes:
- Inadequate sedation/analgesia
- Hypoxaemia (↑ respiratory drive)
- Hypercapnia with acidosis (↑ respiratory drive)
- Auto-PEEP (air trapping)
- Inappropriate trigger sensitivity
- Inadequate inspiratory flow
- Patient attempting to exhale during inspiration (reverse triggering)
Step 3: Address underlying cause
- Optimize sedation: Propofol or midazolam infusion, target RASS -2 to -3
- Optimize analgesia: Fentanyl infusion
- Adjust ventilator settings:
- Increase respiratory rate if pH less than 7.35 (reduce respiratory drive)
- Increase inspiratory flow rate (reduces inspiratory time)
- Adjust trigger sensitivity
- Consider pressure support if on SIMV
- Check for auto-PEEP: perform expiratory hold manoeuvre
- "If present: reduce RR, increase expiratory time, bronchodilators"
- Increase external PEEP to 80% of auto-PEEP level
Step 4: If persistent despite optimization
- Consider neuromuscular blockade (cisatracurium infusion)
- Indications: severe dyssynchrony preventing adequate ventilation
- Duration: 48 hours maximum
- Monitor: train-of-four, ensure deep sedation (RASS -4 to -5)
Scenario 5: Weaning from Mechanical Ventilation "An ARDS patient has P/F ratio improving to 220 on FiO₂ 0.4, PEEP 8 cmH₂O. How do you approach weaning?"
Answer:
Weaning Readiness Criteria:
✓ Improvement in P/F ratio (now 220, was less than 200)
✓ Reduced FiO₂ requirement (0.4)
✓ Moderate PEEP (8 cmH₂O)
- Assess: Underlying cause treated/improving?
- Assess: Haemodynamically stable (no/minimal vasopressors)?
- Assess: Adequate cough and secretion clearance?
- Assess: GCS adequate for airway protection?
Weaning Protocol:
1. **Daily Spontaneous Breathing Trial (SBT)**:
- Mode: Pressure support 5-8 cmH₂O + PEEP 5 cmH₂O, OR
- T-piece trial (100% spontaneous breathing)
- Duration: 30-120 minutes
- Monitor: RR, SpO₂, HR, BP, respiratory distress
2. **SBT Success Criteria**:
- SpO₂ > 90% on FiO₂ ≤0.4
- RR less than 35 breaths/min
- No respiratory distress (accessory muscle use, diaphoresis)
- Haemodynamically stable (no significant ↑HR, ↓BP)
- Adequate tidal volumes (> 5 mL/kg)
3. **If SBT successful**:
- Assess airway protection (cough strength, GCS > 8)
- Perform cuff leak test if concern for laryngeal oedema
- **Extubate** if criteria met
- Post-extubation: high-flow nasal oxygen or NIV if at risk
4. **If SBT fails**:
- Return to full ventilatory support
- Investigate cause: secretions, bronchospasm, cardiac failure, fatigue
- Optimize: secretion clearance, bronchodilators, diuretics if volume overloaded
- Retry SBT next day
5. **Tracheostomy consideration**:
- If prolonged ventilation expected (> 14-21 days)
- Benefits: comfort, mobility, easier weaning, reduced sedation
- Timing: case-by-case; typically after 10-14 days if not improving
Advanced Topics
Exam Detail: ### Phenotypes and Endotypes in ARDS
Recent research suggests ARDS is not a single entity but comprises distinct biological subtypes (endotypes) with different responses to treatment.
Hyper-inflammatory vs. Hypo-inflammatory Phenotypes
Hyper-inflammatory Phenotype (~30% of ARDS patients):
- Higher plasma biomarkers: IL-6, IL-8, sTNFR-1, PAI-1
- Worse clinical outcomes (higher mortality)
- More severe shock, acidosis, organ dysfunction
- May benefit MORE from:
- Higher PEEP strategies
- Conservative fluid management
- Anti-inflammatory therapies (steroids, simvastatin - under investigation)
Hypo-inflammatory Phenotype (~70% of ARDS patients):
- Lower inflammatory biomarkers
- Better outcomes
- May be harmed by aggressive PEEP
Clinical Implications: Currently, phenotyping requires biomarker assays not routinely available. Future personalized ARDS treatment may stratify therapy based on endotype. [23,24]
Focal vs. Non-Focal ARDS
Focal ARDS (mainly lobar pneumonia):
- Lobar consolidation on CT
- Less recruitable lung
- May benefit from LOWER PEEP
- Prone positioning less effective
Non-Focal ARDS (mainly extrapulmonary causes):
- Diffuse ground-glass opacities
- More recruitable lung
- May benefit from HIGHER PEEP
- Prone positioning more effective
Assessment: CT chest can distinguish focal vs. non-focal patterns and guide PEEP titration strategy. [25]
Spontaneous Breathing in ARDS
Traditional Approach: Deep sedation ± neuromuscular blockade to ensure full ventilator control.
Emerging Evidence: Allowing spontaneous breathing efforts (with ventilator support) may:
- Prevent diaphragm atrophy
- Improve V/Q matching (dependent lung recruitment by diaphragmatic contraction)
- Reduce sedation requirements
Risks of Spontaneous Breathing:
- Patient self-inflicted lung injury (P-SILI): Vigorous inspiratory efforts generate high transpulmonary pressures → worsening lung injury
- Pendelluft phenomenon: gas redistribution from non-dependent to dependent lung
- Increased work of breathing → fatigue
Current Practice:
- Allow spontaneous breathing if NOT severe ARDS (P/F > 150) and patient not distressed
- Deep sedation ± NMB in severe ARDS (P/F less than 150) in first 48 hours
- Transition to spontaneous modes (SIMV, pressure support) during recovery phase
Electrical Impedance Tomography (EIT) for PEEP Titration
Concept: EIT provides real-time, bedside imaging of regional lung ventilation using electrical impedance changes.
Benefits:
- Identifies recruitable vs. overdistended lung regions
- Allows individualized PEEP titration (maximize recruitment, minimize overdistension)
- Non-invasive, radiation-free, continuous monitoring
Limitations:
- Not widely available
- Requires expertise in interpretation
- Evidence for mortality benefit pending
Future Direction: EIT may enable precision PEEP titration in ARDS, moving beyond one-size-fits-all PEEP/FiO₂ tables.
Esophageal Manometry and Transpulmonary Pressure
Concept: Esophageal pressure (Pes) estimates pleural pressure. Transpulmonary pressure (PL) = Airway pressure - Pleural pressure.
Rationale: In ARDS with high chest wall elastance (obesity, ascites, abdominal compartment syndrome), high airway pressures may NOT reflect high alveolar pressures. Measuring PL allows true assessment of alveolar stretch.
PEEP Titration Using PL:
- Target end-expiratory PL: 0-10 cmH₂O (prevents atelectasis)
- Target end-inspiratory PL: less than 25-27 cmH₂O (prevents overdistension)
Evidence: EPVent-2 trial (2019) showed no mortality benefit from PL-guided PEEP vs. empirical PEEP/FiO₂ table. However, may benefit specific subgroups (obese, high chest wall elastance).
Current Use: Specialist tool in experienced centres; not routine practice.
Prevention of ARDS
Clinical Pearl: ### Can ARDS Be Prevented?
While ARDS cannot always be prevented, several strategies reduce risk in at-risk patients:
1. Lung-Protective Ventilation in Non-ARDS Patients
Intraoperative lung protection:
- Even patients WITHOUT lung injury benefit from low tidal volumes (6-8 mL/kg IBW) during surgery
- Reduces postoperative pulmonary complications and ARDS development
- Especially important in high-risk surgery (esophagectomy, pneumonectomy, cardiac surgery)
2. Restrictive Transfusion Strategy
Avoid unnecessary transfusions:
- Transfusion-related acute lung injury (TRALI) is a major ARDS risk
- Use restrictive thresholds: transfuse if Hb less than 70 g/L (unless active bleeding, ACS)
- Leukocyte-reduced blood products reduce TRALI risk
3. Conservative Fluid Management in Sepsis
Early goal-directed therapy (EGDT) evolved:
- Initial resuscitation with fluids and vasopressors (first 6 hours)
- After resuscitation, AVOID fluid overload
- Positive fluid balance associated with ARDS development and worse outcomes
4. Early Antimicrobials in Sepsis
Time to antibiotics matters:
- Each hour delay in antibiotics increases mortality in septic shock
- Target: antibiotics within 1 hour of recognition
- Reduces progression from sepsis to ARDS
5. Aspiration Precautions
High-risk patients:
- Reduce aspiration risk: head-of-bed elevation 30-45°, avoid overfeeding
- Prokinetics (metoclopramide) if gastric residuals high
- Consider post-pyloric feeding in high aspiration risk
6. Lung-Protective ICU Bundle
"ARDS prevention bundle" in high-risk ICU patients:
- Low tidal volume ventilation (even if no ARDS yet)
- Conservative fluid strategy
- Minimize transfusions
- Treat infections promptly
- Avoid nephrotoxins and lung-toxic drugs
Medicolegal and Ethical Considerations
Prognostic Discussions with Families
Key Messages:
- ARDS is a serious condition with 30-40% mortality despite optimal care
- Prognosis depends on severity (mild/moderate/severe), underlying cause, age, comorbidities
- Recovery takes weeks to months; many survivors have long-term impairment
- Regular updates as clinical course evolves
When to Discuss Limitations of Treatment:
- Severe multi-organ failure despite maximal support
- Failure to improve after 7-10 days of optimal therapy
- Advanced age with multiple comorbidities
- Pre-existing poor quality of life or frailty
Shared Decision-Making:
- Involve family in treatment escalation decisions (ECMO, ongoing ventilation)
- Explore patient's values, preferences, prior wishes
- Consider time-limited trial of therapy with clear endpoints
Withdrawal of Life-Sustaining Treatment
Indications:
- Futility: no prospect of recovery despite maximal therapy
- Patient/family wishes after informed discussion
- Prolonged critical illness with no improvement
Process:
- Senior clinician decision with multidisciplinary team input
- Communication with family: compassionate, honest, unhurried
- Palliative care involvement
- Symptom management during withdrawal: opioids, sedation for comfort
- Support for family: time with patient, spiritual care, bereavement support
Organ Donation
ARDS patients may be potential organ donors:
- Donation after Circulatory Death (DCD): If treatment withdrawn and death anticipated
- Donation after Brainstem Death (DBD): If brainstem death develops (rare in isolated ARDS)
- Specialist Nurse for Organ Donation (SNOD) involvement
- Separate discussions: withdrawal decision vs. donation decision
Quality Improvement and Audit
Key Performance Indicators for ARDS Management
| Indicator | Target | Rationale |
|---|---|---|
| Lung-protective ventilation compliance | > 95% of ARDS patients receive Vt 6-8 mL/kg IBW | Core evidence-based intervention [3] |
| Plateau pressure monitoring | Documented in 100% of ARDS patients | Essential safety parameter |
| Prone positioning use | > 80% of severe ARDS (P/F less than 150) proned within 36 hours | PROSEVA trial evidence [4] |
| Conservative fluid strategy | > 70% achieve negative fluid balance after resuscitation | FACTT trial evidence [5] |
| Dexamethasone use | > 80% of moderate-severe ARDS receive dexamethasone less than 24 hours | DEXA-ARDS evidence [11] |
| ECMO referral | 100% of eligible patients discussed with ECMO centre | Ensures access to advanced therapy |
| VAP bundle compliance | > 90% compliance with VAP prevention bundle | Reduces nosocomial complications |
Audit Standards
Annual ARDS audit should include:
- Incidence of ARDS in ICU (should be ~10-15% of admissions)
- Severity distribution (mild/moderate/severe)
- Mortality by severity category (compare to Berlin definition benchmarks)
- Compliance with lung-protective ventilation protocol
- Use of adjunctive therapies (prone, steroids, ECMO)
- Ventilator-free days at day 28
- ICU and hospital length of stay
- Long-term follow-up and rehabilitation referral rates
Quality Improvement Initiatives:
- Ventilator bundle implementation
- Education on IBW calculation (common error: using actual weight)
- Checklists for prone positioning
- ARDS recognition algorithms
- Protocolized fluid management
- Post-ICU follow-up clinic
Future Directions and Emerging Therapies
Under Investigation
1. Mesenchymal Stem/Stromal Cells (MSCs):
- Anti-inflammatory and regenerative properties
- Phase 2 trials ongoing
- Preliminary safety data encouraging; efficacy TBD
2. Anti-Inflammatory Therapies:
- Simvastatin: HARP-2 trial showed no benefit, but phenotype-directed approach may help
- Anti-IL-6, anti-IL-8 therapies in development
- Targeting hyper-inflammatory phenotype specifically
3. Surfactant Replacement:
- Exogenous surfactant administration
- Historical trials in adults showed no benefit
- New-generation surfactants under study
4. Anticoagulants:
- ARDS involves microvascular thrombosis
- Heparin, antithrombin, activated protein C trials showed no benefit
- COVID-ARDS thrombosis renewing interest
5. Precision Medicine Approaches:
- Biomarker-guided therapy selection
- Genetic risk stratification
- Endotype-specific treatments
6. Extracorporeal CO₂ Removal (ECCO₂R):
- Lower-flow device than ECMO, removes CO₂ only
- Allows ultra-protective ventilation (Vt less than 6 mL/kg)
- Trials (REST, SUPERNOVA) showed feasibility; efficacy unclear
Patient and Family Information
What is ARDS?
Acute Respiratory Distress Syndrome (ARDS) is a severe lung condition where the lungs become inflamed and filled with fluid, making it very difficult to breathe and get enough oxygen into the blood. It is a serious illness that requires intensive care treatment with a breathing machine (ventilator).
What Causes ARDS?
ARDS is triggered by an injury to the lungs. Common causes include:
- Severe infection in the lungs (pneumonia) or bloodstream (sepsis)
- Inhaling stomach contents (aspiration)
- Major injury or trauma
- Severe inflammation of the pancreas (pancreatitis)
How is ARDS Treated?
Breathing Machine (Ventilator): Patients with ARDS need help breathing. A tube is placed in the windpipe and connected to a ventilator that breathes for the patient. Special settings are used to protect the lungs from further damage.
Medications:
- Sedatives keep patients comfortable while on the ventilator
- Antibiotics treat infections
- Steroids may reduce lung inflammation
- Fluids and medications support blood pressure and other organs
Special Positions: Lying on the stomach (prone position) for many hours each day helps oxygen levels. This position helps the back parts of the lungs work better.
ECMO (for very severe cases): If the lungs are not working well enough despite the ventilator, a special machine (ECMO) can be used to add oxygen directly to the blood outside the body, giving the lungs time to heal.
What to Expect
- ICU stay: Most patients are in intensive care for 1-3 weeks or longer
- Sedation: Patients are kept sedated (asleep) while on the ventilator
- Gradual improvement: Recovery takes time - lungs heal slowly over weeks
- Rehabilitation: Muscle weakness is common; physiotherapy helps with recovery
Prognosis
ARDS is a serious condition. With modern treatment, approximately 60-70% of patients survive. Recovery can take many months, and some patients have long-term breathing difficulties or weakness. The medical team will provide regular updates and discuss progress.
Questions to Ask the Medical Team
- What caused the ARDS?
- How severe is it? (Mild, moderate, or severe)
- What treatments are being used?
- Are there signs of improvement in oxygen levels?
- How long might recovery take?
- What support is available for families?
References
-
ARDS Definition Task Force, Ranieri VM, Rubenfeld GD, et al. Acute respiratory distress syndrome: the Berlin Definition. JAMA. 2012;307(23):2526-2533. doi:10.1001/jama.2012.5669
-
Bellani G, Laffey JG, Pham T, et al. Epidemiology, patterns of care, and mortality for patients with acute respiratory distress syndrome in intensive care units in 50 countries. JAMA. 2016;315(8):788-800. doi:10.1001/jama.2016.0291
-
Acute Respiratory Distress Syndrome Network, Brower RG, Matthay MA, et al. Ventilation with lower tidal volumes as compared with traditional tidal volumes for acute lung injury and the acute respiratory distress syndrome. N Engl J Med. 2000;342(18):1301-1308. doi:10.1056/NEJM200005043421801
-
Guérin C, Reignier J, Richard JC, et al. Prone positioning in severe acute respiratory distress syndrome. N Engl J Med. 2013;368(23):2159-2168. doi:10.1056/NEJMoa1214103
-
Wiedemann HP, Wheeler AP, Bernard GR, et al. Comparison of two fluid-management strategies in acute lung injury. N Engl J Med. 2006;354(24):2564-2575. doi:10.1056/NEJMoa062200
-
Combes A, Hajage D, Capellier G, et al. Extracorporeal membrane oxygenation for severe acute respiratory distress syndrome. N Engl J Med. 2018;378(21):1965-1975. doi:10.1056/NEJMoa1800385
-
Rubenfeld GD, Caldwell E, Peabody E, et al. Incidence and outcomes of acute lung injury. N Engl J Med. 2005;353(16):1685-1693. doi:10.1056/NEJMoa050333
-
Wu C, Chen X, Cai Y, et al. Risk factors associated with acute respiratory distress syndrome and death in patients with coronavirus disease 2019 pneumonia in Wuhan, China. JAMA Intern Med. 2020;180(7):934-943. doi:10.1001/jamainternmed.2020.0994
-
Herridge MS, Tansey CM, Matté A, et al. Functional disability 5 years after acute respiratory distress syndrome. N Engl J Med. 2011;364(14):1293-1304. doi:10.1056/NEJMoa1011802
-
Matthay MA, Zemans RL, Zimmerman GA, et al. Acute respiratory distress syndrome. Nat Rev Dis Primers. 2019;5(1):18. doi:10.1038/s41572-019-0069-0
-
Villar J, Ferrando C, Martínez D, et al. Dexamethasone treatment for the acute respiratory distress syndrome: a multicentre, randomised controlled trial. Lancet Respir Med. 2020;8(3):267-276. doi:10.1016/S2213-2600(19)30417-5
-
Gattinoni L, Pesenti A. The concept of "baby lung". Intensive Care Med. 2005;31(6):776-784. doi:10.1007/s00134-005-2627-z
-
Karmpaliotis D, Kirtane AJ, Ruisi CP, et al. Diagnostic and prognostic utility of brain natriuretic peptide in subjects admitted to the ICU with hypoxic respiratory failure due to noncardiogenic and cardiogenic pulmonary edema. Chest. 2007;131(4):964-971. doi:10.1378/chest.06-1247
-
Amato MB, Meade MO, Slutsky AS, et al. Driving pressure and survival in the acute respiratory distress syndrome. N Engl J Med. 2015;372(8):747-755. doi:10.1056/NEJMsa1410639
-
Briel M, Meade M, Mercat A, et al. Higher vs lower positive end-expiratory pressure in patients with acute lung injury and acute respiratory distress syndrome: systematic review and meta-analysis. JAMA. 2010;303(9):865-873. doi:10.1001/jama.2010.218
-
Papazian L, Forel JM, Gacouin A, et al. Neuromuscular blockers in early acute respiratory distress syndrome. N Engl J Med. 2010;363(12):1107-1116. doi:10.1056/NEJMoa1005372
-
National Heart, Lung, and Blood Institute PETAL Clinical Trials Network, Moss M, Huang DT, et al. Early neuromuscular blockade in the acute respiratory distress syndrome. N Engl J Med. 2019;380(21):1997-2008. doi:10.1056/NEJMoa1901686
-
Cavalcanti AB, Suzumura ÉA, Laranjeira LN, et al. Effect of lung recruitment and titrated positive end-expiratory pressure (PEEP) vs low PEEP on mortality in patients with acute respiratory distress syndrome: a randomized clinical trial. JAMA. 2017;318(14):1335-1345. doi:10.1001/jama.2017.14171
-
Gebistorf F, Karam O, Wetterslev J, Afshari A. Inhaled nitric oxide for acute respiratory distress syndrome (ARDS) in children and adults. Cochrane Database Syst Rev. 2016;2016(6):CD002787. doi:10.1002/14651858.CD002787.pub3
-
Ferguson ND, Cook DJ, Guyatt GH, et al. High-frequency oscillation in early acute respiratory distress syndrome. N Engl J Med. 2013;368(9):795-805. doi:10.1056/NEJMoa1215554
-
Hébert PC, Wells G, Blajchman MA, et al. A multicenter, randomized, controlled clinical trial of transfusion requirements in critical care. N Engl J Med. 1999;340(6):409-417. doi:10.1056/NEJM199902113400601
-
RECOVERY Collaborative Group, Horby P, Lim WS, et al. Dexamethasone in hospitalized patients with Covid-19. N Engl J Med. 2021;384(8):693-704. doi:10.1056/NEJMoa2021436
-
Fan E, Del Sorbo L, Goligher EC, et al. An Official American Thoracic Society/European Society of Intensive Care Medicine/Society of Critical Care Medicine Clinical Practice Guideline: Mechanical Ventilation in Adult Patients with Acute Respiratory Distress Syndrome. Am J Respir Crit Care Med. 2017;195(9):1253-1263. doi:10.1164/rccm.201703-0548ST
-
Slutsky AS, Ranieri VM. Ventilator-induced lung injury. N Engl J Med. 2013;369(22):2126-2136. doi:10.1056/NEJMra1208707
-
Gattinoni L, Caironi P, Cressoni M, et al. Lung recruitment in patients with the acute respiratory distress syndrome. N Engl J Med. 2006;354(17):1775-1786. doi:10.1056/NEJMoa052052
Version History
|---------|------|---------|---------------| | 3.0 | 2026-01-10 | Enhanced to Gold Standard: expanded pathophysiology, PEEP titration strategies, ECMO protocols, clinical pearls, evidence debates, 25 citations, enhanced viva preparation | 54/56 (Gold) | | 2.0 | 2026-01-06 | Complete rewrite to Gold Standard: comprehensive pathophysiology, expanded management, 22 citations, viva questions, special populations | 52/56 (Gold) | | 1.0 | 2025-01-15 | Initial version | Not scored |
Document Status: Gold Standard (54/56) Last Evidence Review: January 2026 Next Review Due: January 2027 Author: MedVellum Critical Care Team Peer Review: Completed Target Audience: MRCP, FRCA, ICU trainees, critical care practitioners
Learning map
Use these linked topics to study the concept in sequence and compare related presentations.
Prerequisites
Start here if you need the foundation before this topic.
- Respiratory Physiology
- Mechanical Ventilation
Differentials
Competing diagnoses and look-alikes to compare.
- Cardiogenic Pulmonary Oedema
- Pneumonia - Severe
- Diffuse Alveolar Haemorrhage
Consequences
Complications and downstream problems to keep in mind.
- Pulmonary Fibrosis
- Critical Illness Polyneuropathy