Cardiac Amyloidosis
Cardiac amyloidosis is an infiltrative cardiomyopathy caused by extracellular deposition of misfolded proteins (amyloid ... MRCP exam preparation.
What matters first
Cardiac amyloidosis is an infiltrative cardiomyopathy caused by extracellular deposition of misfolded proteins (amyloid ... MRCP exam preparation.
Rapidly progressive heart failure with preserved ejection fraction
9 Jan 2026
Generated educational material; verify before clinical use.
Visible references section
See the concept before reading it
Study the key anatomy, imaging, and decision pathways as full teaching plates.
Clinical board
A visual summary of the highest-yield teaching signals on this page.
Urgent signals
Safety-critical features pulled from the topic metadata.
- Rapidly progressive heart failure with preserved ejection fraction
- Low-voltage ECG with increased left ventricular wall thickness (voltage-mass discordance)
- Unexplained LVH in elderly patient without hypertension
- Bilateral carpal tunnel syndrome preceding heart failure by years
Exam focus
Current exam surfaces linked to this topic.
- MRCP
Linked comparisons
Differentials and adjacent topics worth opening next.
- Hypertensive Heart Disease
- Hypertrophic Cardiomyopathy
Content status and exam context
This page is AI-generated educational content. It may contain errors or omissions and is not a substitute for current guidelines, local protocols, senior clinical judgement, or professional medical advice.
MedVellum does not claim an individual clinician reviewer, board certification, or professional credential for this page unless a future version names a real, verifiable reviewer.
Clinical explanation and evidence
Cardiac Amyloidosis
1. Clinical Overview
Summary
Cardiac amyloidosis is an infiltrative cardiomyopathy caused by extracellular deposition of misfolded proteins (amyloid fibrils) in the myocardium, leading to progressive diastolic dysfunction and restrictive physiology. [1,2] Previously considered rare and largely a post-mortem diagnosis, improved recognition and advances in non-invasive diagnostic techniques have revealed cardiac amyloidosis to be a significantly under-diagnosed cause of heart failure with preserved ejection fraction (HFpEF), particularly in elderly patients.
The two clinically important types are:
- Light-chain (AL) amyloidosis: Caused by misfolded immunoglobulin light chains from clonal plasma cell disorders
- Transthyretin (ATTR) amyloidosis: Subdivided into hereditary (ATTRv, variant/mutant) and wild-type (ATTRwt, formerly senile cardiac amyloidosis)
Early diagnosis is critical as prognosis differs markedly between types and disease-modifying therapies are now available. The ATTR-ACT trial demonstrated that tafamidis reduces all-cause mortality by 30% in ATTR cardiac amyloidosis. [3] The classic presentation of low-voltage ECG despite increased wall thickness on echocardiography should prompt immediate investigation.
Key Clinical Facts
| Parameter | Details | Clinical Significance |
|---|---|---|
| Definition | Infiltrative cardiomyopathy from extracellular amyloid fibril deposition | Causes restrictive physiology with diastolic dysfunction |
| AL Incidence | 10-12 per million per year | Cardiac involvement in 50-70% of cases [4] |
| ATTRwt Prevalence | 13-25% of HFpEF patients > 60 years | Massively under-diagnosed [5] |
| ATTRwt in TAVR | 16% of patients undergoing TAVR | Screen in aortic stenosis [6] |
| V122I Mutation | 3-4% of African Americans carry this TTR variant | Major cause of ATTRv cardiomyopathy [7] |
| AL Median Survival | 6 months untreated with cardiac involvement | Urgent haematological treatment required |
| ATTR Median Survival | 3-5 years untreated | Improved with tafamidis |
| Pathognomonic Finding | Low-voltage ECG + increased LV wall thickness | Voltage-mass discordance |
| Non-invasive Diagnosis | Tc-99m bone scintigraphy (PYP/DPD/HMDP) | Grade 2-3 uptake + no monoclonal protein = ATTR [8] |
| Disease-Modifying Therapy | Tafamidis (ATTR), Chemotherapy (AL) | Transforms prognosis when treated early |
Clinical Pearls
Diagnostic Pearl - Voltage-Mass Discordance: The combination of increased LV wall thickness (> 12mm) on echocardiography with low-voltage QRS complexes (less than 0.5mV in limb leads or less than 1.0mV in precordial leads) on ECG is highly suggestive of infiltrative cardiomyopathy. This discordance occurs because amyloid deposits increase mass without contributing to electrical signal generation. Request cardiac MRI and bone scintigraphy immediately.
Examination Pearl - Carpal Tunnel as Early Sign: Bilateral carpal tunnel syndrome preceding heart failure symptoms by 5-10 years is characteristic of ATTR amyloidosis due to amyloid deposition in the flexor retinaculum. Always ask about carpal tunnel surgery in patients presenting with HFpEF, especially males > 65 years. [9]
Treatment Pearl - Tafamidis Impact: Tafamidis reduces mortality and cardiovascular hospitalisation in ATTR cardiac amyloidosis by 30% (ATTR-ACT trial, NNT 7.5 over 30 months). [3] Ensure ALL patients with suspected ATTR are evaluated for this therapy. Do not delay diagnosis.
Pitfall Warning - Type Matters: Do not assume all amyloidosis is AL. ATTR is more common than previously thought and requires different treatment (TTR stabilisers, not chemotherapy). Misdiagnosis leads to inappropriate and potentially harmful treatment.
Imaging Pearl - Bone Scintigraphy Interpretation: Grade 2-3 cardiac uptake on Tc-99m PYP/DPD/HMDP scintigraphy, combined with absence of monoclonal protein (negative serum/urine immunofixation AND normal free light chain ratio), allows diagnosis of ATTR cardiac amyloidosis WITHOUT endomyocardial biopsy. This has revolutionised diagnosis. [8]
ECG Pearl - Pseudo-Infarct Pattern: Look for pseudo-infarct patterns (Q waves in anterior or inferior leads without coronary artery disease) which occur in 50% of cardiac amyloidosis cases due to replacement of myocardium with electrically inert amyloid.
Mnemonic - ATTR Features: Aged (wild-type predominantly elderly), Transthyretin protein, Tafamidis treats it, Runs in families (variant/hereditary form)
Why This Matters Clinically
Cardiac amyloidosis is increasingly recognised as a treatable cause of HFpEF. The convergence of improved awareness, non-invasive diagnostic algorithms, and effective disease-modifying therapy has fundamentally changed the landscape:
- Under-diagnosis is the norm: Autopsy studies suggest ATTRwt is present in 25% of those > 85 years, yet clinical diagnosis remains rare
- Misdiagnosis leads to harm: Patients treated for hypertensive heart disease receive medications (digoxin, CCBs) that are contraindicated in amyloidosis
- Treatment changes prognosis: Early tafamidis in ATTR, or chemotherapy in AL, significantly improves survival
- This is a high-yield MRCP topic: Reflects advances in cardiac imaging and therapeutics, ideal for examination scenarios
2. Epidemiology
Incidence and Prevalence
| Type | Epidemiology | Key Demographics |
|---|---|---|
| AL Amyloidosis | Incidence: 10-12 per million per year | Median age 63-65 years; M:F ratio 1.5:1 [4] |
| AL Cardiac Involvement | 50-70% of AL patients | Determines prognosis |
| ATTRwt (Wild-type) | 13-25% of HFpEF patients > 60 years | Almost exclusively males > 65 years [5] |
| ATTRwt at Autopsy | 25% of individuals > 85 years | Massively under-recognised |
| ATTRwt in AS/TAVR | 6-16% of severe aortic stenosis patients | Common comorbidity [6] |
| ATTRv (Hereditary) | Variable by mutation and geography | Endemic in Portugal, Sweden, Japan |
| V122I Mutation | 3-4% of African Americans | Most common ATTRv mutation globally [7] |
| V30M Mutation | Endemic Portugal, Sweden, Japan | Mixed cardiac and neurological |
Demographics and Risk Factors
| Factor | AL Amyloidosis | ATTRwt | ATTRv |
|---|---|---|---|
| Age | 50-70 years | > 65 years (peak > 80) | Variable (30-80 years by mutation) |
| Sex | M:F 1.5:1 | Male predominant (80-90%) | Variable |
| Ethnicity | No specific association | No specific association | V122I: 3-4% African Americans; V30M: Portuguese, Swedish, Japanese |
| Precursor Conditions | MGUS, myeloma, Waldenstrom's | Age-related TTR instability | TTR gene mutations |
| Associated Features | Nephrotic syndrome, peripheral neuropathy, hepatomegaly | Carpal tunnel, lumbar stenosis, biceps tendon rupture | Peripheral neuropathy, autonomic dysfunction |
Risk Factor Table
| Risk Factor | Type | Relative Risk | Mechanism |
|---|---|---|---|
| Plasma cell dyscrasia (MGUS, myeloma) | AL | Essential | Monoclonal light chain production |
| TTR gene mutations (> 130 known) | ATTRv | Essential | Unstable transthyretin protein |
| Advanced age (> 65 years) | ATTRwt | Major | Age-related TTR tetramer instability |
| Male sex | ATTRwt | 4-9x | Unknown; possible hormonal factors |
| African American ancestry | ATTRv | High for V122I | 3-4% carry V122I mutation |
| Aortic stenosis | ATTRwt | Associated | Shared risk factors or pathophysiology |
| Pre-existing carpal tunnel syndrome | ATTR | Clinical marker | Early amyloid deposition site |
Geographic Variation
ATTRv has distinct endemic populations due to founder effects:
| Population | Mutation | Predominant Phenotype |
|---|---|---|
| Portuguese | V30M | Neuropathy > Cardiomyopathy |
| Swedish | V30M | Cardiomyopathy > Neuropathy |
| Japanese | V30M | Mixed phenotype |
| African American | V122I | Cardiomyopathy predominant |
| Irish | T60A | Mixed phenotype |
| Danish | L111M | Cardiomyopathy predominant |
3. Pathophysiology
Molecular Mechanisms
Step 1: Amyloid Precursor Protein Production
AL Amyloidosis:
- Clonal plasma cells produce excess monoclonal immunoglobulin light chains (typically lambda > kappa)
- Light chains have inherent amyloidogenic properties based on amino acid sequence
- Certain light chain subtypes (lambda VI, lambda III) are particularly amyloidogenic
- The degree of amyloidogenicity correlates with clinical severity [10]
ATTR Amyloidosis:
- Transthyretin (TTR) is a 55 kDa homotetrameric protein synthesised primarily in the liver
- TTR normally transports thyroxine (T4) and retinol-binding protein
- ATTRwt: Age-related post-translational modifications destabilise the tetramer
- ATTRv: Point mutations in the TTR gene destabilise the tetramer structure
- Over 130 TTR mutations are described; V122I and V30M are most common [7]
Step 2: Protein Misfolding and Fibril Formation
The amyloid cascade proceeds through defined steps:
- Tetramer dissociation (ATTR) or light chain misfolding (AL)
- Monomer formation with exposed hydrophobic regions
- Oligomer aggregation into intermediate toxic species
- Beta-pleated sheet configuration - characteristic cross-beta structure
- Fibril elongation into insoluble fibrils (7.5-10nm diameter, rigid, unbranched)
- Fibril deposition in extracellular matrix
All amyloid fibrils share:
- Characteristic apple-green birefringence under polarised light with Congo red staining
- Binding of serum amyloid P component (SAP)
- Resistance to proteolytic degradation
Step 3: Cardiac Infiltration and Toxicity
Amyloid causes cardiac dysfunction through multiple mechanisms:
Structural Effects:
- Progressive accumulation in myocardial interstitium
- Wall thickening WITHOUT true myocyte hypertrophy
- Disruption of myocyte architecture and contractile function
- Deposition in coronary arteries (microvascular dysfunction)
- Deposition in conduction system (arrhythmias, AV block)
- Atrial infiltration (AF, atrial thrombus formation)
Direct Toxicity (particularly AL):
- Light chain oligomers are directly cardiotoxic [10]
- Induction of oxidative stress
- Activation of p38 MAPK pathway
- Mitochondrial dysfunction
- Cellular apoptosis
- This explains the rapid progression in AL compared to ATTR
Step 4: Haemodynamic Consequences
The infiltrated heart develops restrictive physiology:
| Haemodynamic Parameter | Effect | Clinical Consequence |
|---|---|---|
| Ventricular compliance | Severely reduced | Elevated filling pressures |
| Diastolic function | Grade III (restrictive) | Rapid E-wave, short deceleration time |
| Atrial function | Impaired contractility | AF, atrial thrombus even in sinus rhythm |
| Stroke volume | Fixed, preload-dependent | Intolerance to vasodilators/diuretics |
| Cardiac output | Maintained then declining | Low-output heart failure |
| Conduction | Disrupted | AV block, bundle branch block |
Step 5: Systemic Involvement (Type-Specific)
AL Amyloidosis - Multi-organ:
- Kidney (nephrotic syndrome, renal failure)
- Liver (hepatomegaly, elevated ALP)
- Gastrointestinal tract (malabsorption, bleeding)
- Peripheral nerves (sensorimotor neuropathy)
- Autonomic nerves (orthostatic hypotension)
- Soft tissues (macroglossia, periorbital purpura)
ATTR Amyloidosis:
- Heart (cardiomyopathy - predominant in ATTRwt)
- Peripheral nerves (polyneuropathy - predominant in some ATTRv mutations)
- Autonomic nerves (orthostatic hypotension, GI dysmotility)
- Soft tissues (carpal tunnel syndrome, lumbar stenosis)
- Eyes (vitreous opacities in some ATTRv)
Classification Systems
Classification by Amyloid Type
| Type | Precursor Protein | Key Features | Cardiac Involvement | Prognosis |
|---|---|---|---|---|
| AL (Primary) | Immunoglobulin light chain | Multi-organ, rapid progression | 50-70% | Median 6 months untreated |
| ATTRwt (Wild-type) | Wild-type transthyretin | Elderly males, isolated cardiac | 100% | Median 3-5 years |
| ATTRv (Hereditary) | Mutant transthyretin | Variable by mutation, family history | Variable | Mutation-dependent |
| AA (Secondary) | Serum amyloid A | Chronic inflammation, rare cardiac | less than 5% | Depends on underlying disease |
| AApoAI | Apolipoprotein AI | Rare, hepatic and cardiac | Variable | Slowly progressive |
Cardiac Staging Systems
Mayo Staging for AL Cardiac Amyloidosis (2004, Revised 2012): [11]
| Stage | Biomarker Criteria | Median Survival |
|---|---|---|
| I | TnT less than 0.025 ng/mL AND NT-proBNP less than 332 pg/mL | 26 months |
| II | Either elevated (not both) | 11 months |
| III | Both elevated | 4 months |
| IIIb (2012 revision) | Stage III + NT-proBNP > 8500 pg/mL | 3 months |
European Modification for AL (2015): Incorporates dFLC (difference between involved and uninvolved free light chain):
- Stage I: NT-proBNP less than 332, cTnT less than 0.035, dFLC less than 180
- Stage IV: NT-proBNP > 332, cTnT > 0.035, dFLC > 180
NAC Staging for ATTR Cardiac Amyloidosis: [12]
| Stage | Criteria | Median Survival |
|---|---|---|
| I | NT-proBNP ≤3000 pg/mL AND eGFR ≥45 mL/min | > 60 months |
| II | NT-proBNP > 3000 OR eGFR less than 45 (not both) | 47 months |
| III | NT-proBNP > 3000 AND eGFR less than 45 | 24 months |
4. Clinical Presentation
Cardinal Symptoms
Heart Failure Symptoms (Present in > 80%):
| Symptom | Frequency | Characteristics |
|---|---|---|
| Dyspnoea on exertion | 80-90% | Often out of proportion to LVEF |
| Fatigue | 70-80% | Due to low cardiac output |
| Peripheral oedema | 60-70% | Often refractory to diuretics |
| Orthopnoea | 50-60% | Restrictive physiology |
| Paroxysmal nocturnal dyspnoea | 40-50% | Elevated filling pressures |
| Exercise intolerance | 80-90% | Chronotropic incompetence common |
Cardiac Symptoms Beyond Heart Failure:
| Symptom | Frequency | Significance |
|---|---|---|
| Palpitations | 40-60% | AF common; ventricular arrhythmias |
| Syncope/pre-syncope | 15-25% | Arrhythmia or autonomic dysfunction; poor prognostic sign |
| Chest discomfort | 20-30% | Microvascular dysfunction |
| Dizziness | 30-40% | Orthostatic hypotension |
Extracardiac Symptoms (Critical Diagnostic Clues)
Symptoms Suggesting AL Amyloidosis:
| Symptom | Frequency in AL | Mechanism |
|---|---|---|
| Easy bruising | 30-40% | Factor X deficiency, capillary fragility |
| Periorbital purpura | 15-20% | Pathognomonic - capillary fragility |
| Macroglossia | 10-20% | Pathognomonic - tongue infiltration |
| Jaw claudication | 5-10% | Tongue/jaw muscle infiltration |
| Foamy urine | 30-50% | Nephrotic syndrome |
| Peripheral oedema | 50-60% | Hypoalbuminaemia + cardiac |
| Weight loss | 40-50% | GI involvement, malabsorption |
| Altered bowel habit | 20-30% | GI dysmotility, malabsorption |
Symptoms Suggesting ATTR Amyloidosis:
| Symptom | Frequency | Clinical Significance |
|---|---|---|
| Bilateral carpal tunnel syndrome | 40-50% | Often precedes cardiac symptoms by 5-10 years [9] |
| Lumbar spinal stenosis | 20-30% | Due to ligamentum flavum infiltration |
| Biceps tendon rupture | 10-15% | Spontaneous, bilateral |
| Peripheral neuropathy | ATTRv > ATTRwt | Sensorimotor, length-dependent |
| Autonomic symptoms | 30-50% | Orthostatic hypotension, early satiety, erectile dysfunction |
Red Flag Presentations
[!CAUTION] RED FLAGS - SUSPECT CARDIAC AMYLOIDOSIS
ECG-Echo Discordance:
- Low-voltage ECG (limb leads less than 5mm, precordial less than 10mm) with increased LV wall thickness (> 12mm)
- This voltage-mass discordance is highly specific for infiltrative disease
Clinical Combinations:
- HFpEF + bilateral carpal tunnel syndrome history
- HFpEF + unexplained LVH in elderly without hypertension
- HFpEF + nephrotic syndrome (suggests AL)
- HFpEF + peripheral neuropathy
- HFpEF + orthostatic hypotension
Alarm Features:
- Syncope in HFpEF patient (poor prognosis; consider arrhythmia)
- Macroglossia (pathognomonic for AL - urgent haematology referral)
- Periorbital purpura (pathognomonic for AL)
- Rapidly progressive symptoms (suggests AL)
ECG Red Flags:
- Pseudo-infarct pattern (Q waves without CAD)
- AV conduction disease with thick ventricles
- Low voltage in a "thick heart"
5. Clinical Examination
Structured Cardiovascular Examination
General Inspection
| Finding | Type | Significance |
|---|---|---|
| Cachexia | AL > ATTR | Poor prognostic sign; late disease |
| Periorbital purpura | AL | Pathognomonic; due to capillary fragility |
| Macroglossia | AL | Pathognomonic; ask to protrude tongue, look for teeth marks |
| Bilateral carpal tunnel scars | ATTR > AL | Important historical clue |
| Skin thickening | AL | May be subtle |
| Easy bruising | AL | Factor X deficiency |
Cardiovascular Examination
Jugular Venous Pressure:
- Elevated (often > 10 cmH2O)
- Kussmaul's sign: Paradoxical rise on inspiration (restrictive physiology)
- Prominent x and y descents (restrictive pattern)
Apex Beat:
- Non-displaced (normal LV cavity size)
- Difficult to palpate (thick but non-compliant)
- No sustained heave despite wall thickening
Auscultation:
- S1: Soft (reduced contractility)
- S2: Normal or soft
- S4: Present (atrial contraction against stiff ventricle)
- S3: May be present in advanced disease
- Murmurs: Usually absent; may have functional MR/TR
Peripheral Signs of Heart Failure:
- Peripheral oedema (often pitting, bilateral)
- Hepatomegaly (may be pulsatile with TR)
- Ascites (late sign)
- Elevated JVP as above
Systemic Examination for Extra-Cardiac Involvement
| System | Finding | Amyloid Type |
|---|---|---|
| Tongue | Macroglossia, teeth marks, reduced mobility | AL |
| Skin | Periorbital purpura, waxy papules | AL |
| Abdomen | Hepatomegaly (smooth, non-tender) | AL > ATTR |
| Hands | Carpal tunnel scars, thenar wasting | ATTR > AL |
| Neurological | Peripheral sensory loss (stocking distribution) | ATTRv, AL |
| Blood pressure | Orthostatic hypotension (> 20 mmHg drop) | Both |
Special Tests at Bedside
| Test | Method | Positive Finding | Significance |
|---|---|---|---|
| Orthostatic BP | Lying then standing BP | > 20 mmHg systolic drop | Autonomic amyloid involvement |
| Tinel's sign | Tap carpal tunnel | Paraesthesia in median nerve distribution | Carpal tunnel syndrome |
| Phalen's test | Wrist flexion 60 seconds | Paraesthesia | Carpal tunnel syndrome |
| Tongue examination | Protrude, inspect edges | Macroglossia, teeth indentations | AL amyloidosis |
| Skin examination | Periorbital area | Purple discolouration | AL amyloidosis |
6. Investigations
First-Line Investigations
ECG Findings [13]
| Finding | Frequency | Clinical Significance |
|---|---|---|
| Low voltage (limb leads less than 5mm) | 50-60% | Highly suggestive with LVH on echo |
| Low voltage (precordial less than 10mm) | 40-50% | Part of voltage-mass discordance |
| Pseudo-infarct pattern | 50-60% | Q waves in V1-V3 or inferior leads without CAD |
| Poor R-wave progression | 60-70% | Often misinterpreted as old anterior MI |
| Atrial fibrillation | 15-20% at presentation; 70% over time | Common; consider anticoagulation |
| First-degree AV block | 20-30% | Conduction system infiltration |
| Bundle branch block | 10-20% | LBBB or RBBB |
| Atrial flutter | 5-10% | Less common than AF |
Critical ECG Interpretation: The combination of low-voltage QRS with echocardiographic LVH (voltage-mass discordance) is nearly pathognomonic for cardiac amyloidosis and distinguishes it from hypertensive heart disease or HCM.
Echocardiography [14]
| Parameter | Typical Findings | Significance |
|---|---|---|
| LV Wall Thickness | > 12mm (often 15-18mm) | Without hypertension history |
| RV Wall Thickness | > 5mm | Biventricular involvement |
| IVS Appearance | "Granular sparkling" | Classic but insensitive finding |
| Valve Thickening | Mitral, aortic, tricuspid | Amyloid deposition |
| Biatrial Enlargement | Both atria dilated | Restrictive physiology |
| LV Cavity Size | Normal or small | Despite thick walls |
| EF | Normal or mildly reduced | HFpEF phenotype initially |
| Diastolic Function | Grade II-III | Restrictive filling pattern |
| E/A Ratio | > 2 | Restrictive |
| Deceleration Time | less than 150ms | Restrictive |
| E/e' | > 14 (often > 20) | Elevated filling pressures |
| GLS (Strain) | Reduced, apical sparing pattern | Highly suggestive [15] |
| Pericardial Effusion | Small, common | 30-50% |
Strain Imaging (Speckle Tracking):
- Global longitudinal strain (GLS) is reduced (< -18%)
- Apical sparing pattern: Relative preservation of apical strain with reduced basal and mid-wall strain
- This "cherry on top" or "bullseye" pattern is characteristic and helps distinguish from other causes of LVH [15]
Laboratory Investigations
| Test | Finding in AL | Finding in ATTR | Purpose |
|---|---|---|---|
| NT-proBNP | Markedly elevated (often > 3000 pg/mL) | Elevated | Prognosis, staging |
| Troponin (I or T) | Often elevated | Often elevated | Prognosis, staging |
| Serum Free Light Chains | Abnormal kappa:lambda ratio | Normal ratio | Distinguish AL from ATTR |
| Serum Immunofixation | Monoclonal protein in 80% | Negative | Detect monoclonal protein |
| Urine Immunofixation | Monoclonal protein (Bence-Jones) | Negative | Detect monoclonal protein |
| eGFR | May be reduced (renal AL) | May be reduced (cardiorenal) | Staging (NAC) |
| Albumin | Low (nephrotic syndrome in AL) | Normal | AL renal involvement |
| LFTs | Elevated ALP (hepatic AL) | Usually normal | AL hepatic involvement |
| Genetic Testing | Not indicated | TTR gene sequencing | Confirm ATTRv |
Advanced Imaging
Cardiac MRI [16]
| Sequence/Finding | Interpretation |
|---|---|
| Cine imaging | Increased wall thickness, biatrial dilatation, small pericardial effusion |
| Late Gadolinium Enhancement (LGE) | Diffuse subendocardial or transmural enhancement; characteristic difficulty nulling myocardium |
| Native T1 Mapping | Elevated (> 1100ms at 1.5T); highly sensitive for infiltration |
| Extracellular Volume (ECV) | Markedly elevated (> 40%); indicates amyloid burden |
| T2 Mapping | May be elevated in acute AL (myocardial oedema) |
Characteristic CMR Pattern:
- LGE showing diffuse subendocardial enhancement progressing to transmural
- "Zebra stripe" appearance in some cases
- T1 mapping is more sensitive than LGE for early disease
- ECV correlates with amyloid burden and prognosis
Nuclear Imaging - Bone Scintigraphy [8]
This is the key investigation that allows non-invasive diagnosis of ATTR without biopsy.
Technique:
- Tc-99m labelled bone-seeking tracers: PYP (pyrophosphate), DPD (3,3-diphosphono-1,2-propanodicarboxylic acid), or HMDP (hydroxymethylene diphosphonate)
- Planar and SPECT imaging at 1-3 hours post-injection
- Comparison with rib uptake (Perugini grading)
Perugini Grading System:
| Grade | Description | ATTR Diagnosis |
|---|---|---|
| 0 | No cardiac uptake | Excludes ATTR (in symptomatic patient) |
| 1 | Mild uptake (< rib) | Possible early ATTR; consider biopsy |
| 2 | Moderate uptake (= rib) | Diagnostic of ATTR (if no monoclonal protein) |
| 3 | Strong uptake (> rib) + reduced bone uptake | Diagnostic of ATTR (if no monoclonal protein) |
Critical Diagnostic Algorithm: [8] Grade 2-3 uptake + Negative serum/urine immunofixation + Normal free light chain ratio = ATTR cardiac amyloidosis confirmed WITHOUT biopsy
This algorithm has > 99% specificity and positive predictive value for ATTR when rigorously applied.
Important Caveats:
- If monoclonal protein IS present, biopsy is required (may be AL with incidental MGUS, or mixed AL+ATTR)
- Grade 1 uptake is indeterminate and requires further evaluation
- Bone scintigraphy does NOT distinguish ATTRwt from ATTRv (requires genetic testing)
Tissue Diagnosis
Indications for Biopsy
Biopsy is required when:
- Bone scintigraphy is negative or Grade 1
- Monoclonal protein is present (cannot exclude AL)
- Alternative amyloid types suspected
- Diagnosis uncertain after non-invasive workup
Biopsy Sites
| Site | Sensitivity | Invasiveness | Notes |
|---|---|---|---|
| Abdominal Fat Pad Aspirate | 70-80% for AL; 45-80% for ATTR | Low | First-line if available |
| Bone Marrow | 50-60% for AL | Low | Useful for staging AL |
| Rectal/GI Biopsy | 70-80% | Low | If GI symptoms present |
| Endomyocardial Biopsy | > 95% | Higher | Gold standard; required if other sites negative |
Histopathology
| Stain/Test | Finding | Interpretation |
|---|---|---|
| Congo Red | Salmon-pink deposits | Amyloid confirmed |
| Polarised Microscopy | Apple-green birefringence | Confirms amyloid |
| Immunohistochemistry | Antibodies to kappa/lambda, TTR, SAA | Types the amyloid |
| Mass Spectrometry | Laser microdissection + MS/MS | Gold standard for typing |
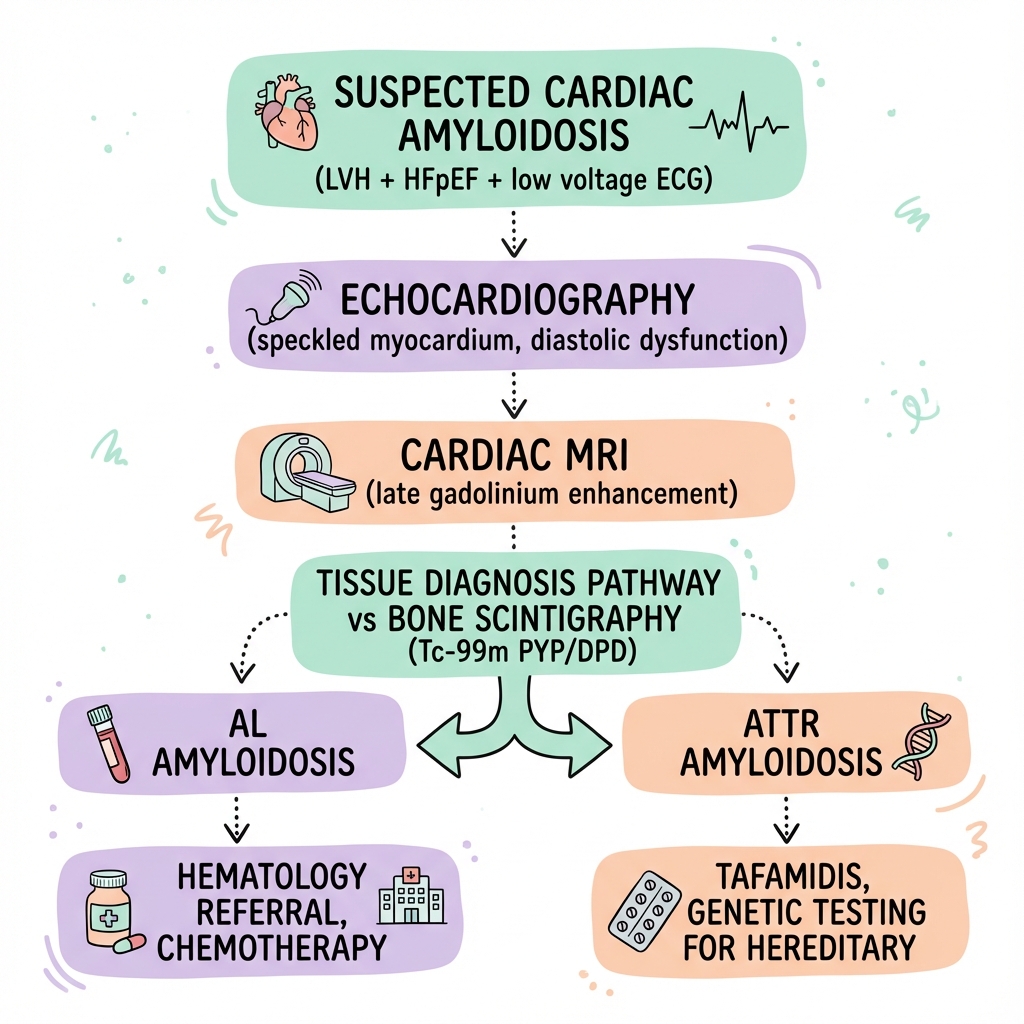
Diagnostic Algorithm
SUSPECTED CARDIAC AMYLOIDOSIS
(HFpEF + LVH + low-voltage ECG or other red flags)
|
v
┌───────────────────────────────────┐
│ STEP 1: Initial Workup │
│ - ECG + Echocardiography │
│ - NT-proBNP, Troponin │
│ - Serum free light chains │
│ - Serum + urine immunofixation │
└───────────────┬───────────────────┘
|
v
┌───────────────────────────────────┐
│ STEP 2: Bone Scintigraphy │
│ (Tc-99m PYP/DPD/HMDP) │
└───────────────┬───────────────────┘
|
┌───────────┼───────────┐
| | |
v v v
Grade 0 Grade 1 Grade 2-3
| | |
v v v
Not ATTR Indeterminate Potential ATTR
| | |
v v v
| Check for monoclonal protein
| |
| ┌──────┴──────┐
| | |
v v v
Biopsy NEGATIVE POSITIVE
required | |
v v
ATTR CONFIRMED BIOPSY REQUIRED
No biopsy needed (may be AL or mixed)
|
┌─────┴─────┐
| |
v v
Genetic Testing If positive
for TTR mutations → ATTRv
| If negative
v → ATTRwt
Management based on type
7. Management
General Principles
- Confirm diagnosis and type - AL vs ATTR has fundamentally different treatment
- Multidisciplinary approach - Cardiology, Haematology (AL), Neurology (if neuropathy), Genetics (ATTRv), specialist amyloid centre
- Refer to specialist centre - National Amyloidosis Centre (UK) or equivalent
- Avoid harmful medications - Digoxin, CCBs, negative inotropes
- Supportive care while pursuing disease-specific treatment
- Prognostic staging - guides treatment intensity
Conservative/Supportive Management
Lifestyle Modifications
| Intervention | Rationale | Practical Advice |
|---|---|---|
| Fluid restriction | Reduce preload | 1.5-2L/day |
| Salt restriction | Reduce fluid retention | less than 2g sodium/day |
| Compression stockings | Manage peripheral oedema | Graduated; especially for orthostatic hypotension |
| Fall prevention | Autonomic dysfunction, syncope risk | Home safety assessment |
| Avoid dehydration | Preload-dependent cardiac output | Careful balance with fluid restriction |
| Moderate activity | Maintain function | Avoid overexertion |
| Vaccination | Heart failure management | Influenza, pneumococcal |
Heart Failure Medications - CAUTION REQUIRED
| Drug Class | Recommendation | Rationale |
|---|---|---|
| Loop Diuretics | Mainstay for congestion | Furosemide 40-160mg/day; bumetanide if resistant. Titrate carefully - avoid over-diuresis |
| MRAs | Consider low dose | Spironolactone 12.5-25mg. Monitor potassium |
| ACE inhibitors/ARBs | Often poorly tolerated | Cause hypotension due to autonomic dysfunction; use with extreme caution |
| Beta-blockers | Often poorly tolerated | Rate-dependent cardiac output; use only if essential (e.g., rate control for AF) |
| Digoxin | AVOID or very low dose | Binds to amyloid fibrils → increased toxicity at "therapeutic" levels [17] |
| Calcium Channel Blockers | AVOID (especially non-DHP) | Bind to amyloid → negative inotropy → clinical deterioration [17] |
| SGLT2 inhibitors | Limited evidence | May help with congestion; ongoing trials |
| Sacubitril/valsartan | Not recommended | Causes hypotension; no evidence of benefit |
Disease-Modifying Therapy: ATTR Amyloidosis
TTR Stabilisers
Tafamidis (Vyndaqel/Vyndamax): [3]
| Aspect | Details |
|---|---|
| Mechanism | Binds TTR tetramer, prevents dissociation into monomers |
| Evidence | ATTR-ACT trial: 30% reduction in all-cause mortality, 32% reduction in CV hospitalisation |
| Dose | Tafamidis meglumine 80mg daily OR tafamidis free acid 61mg daily |
| Indication | All patients with ATTR cardiac amyloidosis (NYHA I-III) |
| When to start | As early as possible after diagnosis |
| Monitoring | Clinical review, biomarkers; no specific monitoring |
| Limitations | Does not clear existing amyloid; high cost |
Diflunisal: (Off-label)
- NSAID with TTR-stabilising properties
- Evidence: Observational data suggesting slower progression
- Dose: 250mg BD
- Issues: NSAID side effects (GI, renal, CV risk); contraindicated in heart failure
Gene Silencing Therapies
Patisiran (Onpattro): [18]
| Aspect | Details |
|---|---|
| Mechanism | siRNA targeting TTR mRNA; reduces hepatic TTR production by 80% |
| Evidence | APOLLO trial: Improved neuropathy; APOLLO-B showed cardiac benefit |
| Indication | ATTRv with polyneuropathy; cardiac benefit demonstrated |
| Dose | 0.3mg/kg IV every 3 weeks |
| Premedication | Corticosteroid, antihistamines, paracetamol |
| Monitoring | Vitamin A levels (TTR carries vitamin A) |
Inotersen (Tegsedi):
| Aspect | Details |
|---|---|
| Mechanism | Antisense oligonucleotide; reduces TTR synthesis |
| Evidence | NEURO-TTR trial: Improved neuropathy |
| Indication | ATTRv with polyneuropathy |
| Dose | 284mg SC weekly |
| Monitoring | Platelet count (risk of thrombocytopenia); renal function |
Vutrisiran (Amvuttra):
- Next-generation siRNA; more convenient dosing (every 3 months)
- HELIOS-A trial showed sustained neuropathy improvement
- Cardiac outcomes data from HELIOS-B expected
Disease-Modifying Therapy: AL Amyloidosis
Treatment is directed at the underlying plasma cell clone - managed by Haematology.
Chemotherapy Regimens [19]
| Regimen | Components | Notes |
|---|---|---|
| CyBorD | Cyclophosphamide, Bortezomib, Dexamethasone | Standard first-line |
| Daratumumab-CyBorD | Add anti-CD38 antibody | ANDROMEDA trial: improved haematologic response |
| VCD | Bortezomib, Cyclophosphamide, Dexamethasone | Alternative first-line |
| Melphalan-Dexamethasone | For transplant-ineligible | Older regimen |
Treatment Goals:
- Achieve haematological response (normalisation of free light chains)
- Organ response follows haematological response
- Complete response (CR): Normal FLC ratio, negative immunofixation
- Very good partial response (VGPR): dFLC less than 40mg/L
Autologous Stem Cell Transplantation
| Aspect | Details |
|---|---|
| Role | Deepest, most durable responses |
| Selection | Fit patients, limited organ involvement, troponin not too high |
| Conditioning | High-dose melphalan |
| Outcomes | Median survival > 10 years in selected patients |
| Limitations | Only 20-25% of AL patients eligible; treatment-related mortality 5-10% |
Management of Arrhythmias
Atrial Fibrillation
| Aspect | Management |
|---|---|
| Rate control | Challenging; beta-blockers and CCBs often poorly tolerated. Digoxin toxic. Amiodarone may be needed. |
| Rhythm control | Cardioversion often fails due to atrial involvement; recurrence high |
| Anticoagulation | STRONG indication. High thromboembolic risk. Consider anticoagulation even in sinus rhythm due to atrial standstill. [20] |
Ventricular Arrhythmias
| Aspect | Management |
|---|---|
| Risk | VT/VF risk increased; sudden cardiac death accounts for significant mortality |
| ICD | Role uncertain; not clearly beneficial in most studies; consider for secondary prevention |
| Risk stratification | Ongoing research; biomarkers, imaging, Holter monitoring |
Management of Conduction Disease
| Problem | Management |
|---|---|
| High-grade AV block | Permanent pacemaker implantation |
| Symptomatic bradycardia | PPM |
| ICD | Limited evidence for primary prevention; discuss on case-by-case basis |
Advanced Heart Failure Therapies
| Therapy | Role in Cardiac Amyloidosis |
|---|---|
| Heart Transplantation | Selected patients; especially younger ATTRv. Combined heart-liver for ATTRv (removes source) |
| LVAD | Generally NOT recommended due to restrictive physiology, RV dysfunction, small cavities |
| Palliative Care | Important role; symptom management; advance care planning |
Disposition and Follow-Up
| Scenario | Disposition | Follow-Up |
|---|---|---|
| New diagnosis | Admit for workup, staging, initiate treatment | Specialist amyloid centre referral |
| Stable ATTR on tafamidis | Outpatient | Cardiology 3-6 monthly; biomarkers |
| Stable AL on chemotherapy | Outpatient | Haematology monthly initially; biomarkers |
| Decompensated HF | Admit | IV diuretics, optimise volume status |
| Syncope | Admit | Rule out arrhythmia; consider PPM/ICD evaluation |
| Advanced/refractory | Consider inpatient palliation | Advance care planning |
8. Complications
Cardiovascular Complications
| Complication | Incidence | Mechanism | Management |
|---|---|---|---|
| Atrial Fibrillation | 40-70% lifetime | Atrial infiltration, stretch | Rate control difficult; anticoagulation |
| Atrial Flutter | 10-20% | As above | Anticoagulation; consider ablation |
| Heart Failure | Universal | Progressive restrictive cardiomyopathy | Diuretics, disease-modifying therapy |
| Intracardiac Thrombus | 10-30% | Atrial standstill even in sinus rhythm | Anticoagulation; TEE before cardioversion |
| Stroke/Systemic Embolism | 10-20% | Atrial thrombus | Anticoagulation (strong indication) |
| Ventricular Arrhythmias | 10-20% | Myocardial infiltration | Amiodarone; ICD case-by-case |
| Sudden Cardiac Death | 25-40% of deaths | VT/VF or EMD | Limited ICD evidence |
| High-Grade AV Block | 10-20% | Conduction system infiltration | Pacemaker |
| Syncope | 15-25% | Arrhythmia or autonomic | Holter, tilt test; PPM if indicated |
Systemic Complications (AL Amyloidosis)
| System | Complication | Management |
|---|---|---|
| Renal | Nephrotic syndrome, CKD, ESRD | Treat underlying clone; supportive; dialysis if ESRD |
| Hepatic | Hepatomegaly, cholestasis, liver failure | Treat underlying clone; supportive |
| GI | Malabsorption, GI bleeding, dysmotility | Nutritional support; treat underlying clone |
| Neurological | Peripheral neuropathy, autonomic dysfunction | Symptomatic treatment; treat underlying clone |
| Haematological | Factor X deficiency, bleeding | FFP for bleeding; treat underlying clone |
Treatment-Related Complications
| Treatment | Complication | Management |
|---|---|---|
| Bortezomib | Peripheral neuropathy, thrombocytopenia | Dose reduction; subcutaneous route |
| Melphalan | Cytopenias, secondary MDS/AML | Monitoring; growth factors |
| Daratumumab | Infusion reactions, infections | Premedication; infection prophylaxis |
| Patisiran | Infusion reactions, vitamin A deficiency | Premedication; vitamin A supplementation |
| Diuretics | Over-diuresis, renal dysfunction | Careful titration; monitor weight, renal function |
9. Prognosis and Outcomes
Natural History (Untreated)
| Type | Median Survival | Key Prognostic Factors |
|---|---|---|
| AL Cardiac Amyloidosis | 6 months | Cardiac biomarkers, haematologic response |
| ATTRwt | 3-5 years | Cardiac biomarkers, NYHA class, eGFR |
| ATTRv | Variable by mutation | Mutation type, age of onset, cardiac involvement |
Outcomes with Modern Treatment
| Type | Treatment | Outcome |
|---|---|---|
| AL - Haematologic CR | Chemotherapy | Median survival > 5 years |
| AL - No response | Chemotherapy | Median survival 6-12 months |
| AL - ASCT eligible | ASCT | Median survival > 10 years |
| ATTR - Tafamidis | TTR stabiliser | 30% reduction in mortality vs placebo [3] |
| ATTR - Untreated | Supportive | Median 3-5 years |
Prognostic Biomarkers
| Biomarker | Threshold | Significance |
|---|---|---|
| NT-proBNP | > 332 pg/mL (AL); > 3000 pg/mL (ATTR) | Adverse prognosis |
| Troponin T | > 0.025-0.035 ng/mL | Adverse prognosis |
| dFLC | > 180 mg/L | Adverse in AL |
| eGFR | less than 45 mL/min | Adverse in ATTR staging |
| 6-minute walk distance | less than 300m | Functional impairment |
| VO2 max | less than 14 mL/kg/min | Severe limitation |
Favourable vs Adverse Prognostic Factors
Favourable:
- Early diagnosis and treatment initiation
- ATTR type (vs AL)
- Low-stage disease (Mayo Stage I for AL; NAC Stage I for ATTR)
- Haematological complete response (AL)
- NYHA Class I-II
- Normal or mildly elevated biomarkers
- Younger age
- Good renal function
Adverse:
- Advanced cardiac stage (Mayo Stage III/IIIb; NAC Stage III)
- AL type without haematological response
- NYHA Class III-IV
- Markedly elevated biomarkers (NT-proBNP > 8500, troponin elevated)
- Syncope
- Ventricular arrhythmias
- Poor functional capacity
- Renal impairment
- Multi-organ involvement (AL)
10. Evidence Base and Guidelines
Key Guidelines
| Guideline | Year | Key Recommendations |
|---|---|---|
| ESC Guidelines on Cardiomyopathies | 2023 | Integrated diagnostic approach; tafamidis for ATTR |
| AHA Scientific Statement on Cardiac Amyloidosis | 2020 | Comprehensive diagnostic and management framework [1] |
| ESC Heart Failure Guidelines | 2021 | Recognition of amyloidosis as cause of HFpEF |
| UK National Amyloidosis Centre Protocols | Ongoing | Expert centre guidance |
| International Society of Amyloidosis Consensus | 2021 | Response criteria, staging systems |
Landmark Trials
ATTR-ACT Trial (2018) [3]
| Aspect | Details |
|---|---|
| Design | Randomised, double-blind, placebo-controlled |
| Population | n=441 with ATTR cardiac amyloidosis |
| Intervention | Tafamidis 80mg or 20mg vs placebo |
| Primary Endpoint | Hierarchical analysis of mortality and CV hospitalisation |
| Results | 30% reduction in all-cause mortality (HR 0.70); 32% reduction in CV hospitalisation |
| Impact | First disease-modifying therapy approved for ATTR cardiomyopathy |
| Citation | Maurer MS et al. N Engl J Med. 2018;379(11):1007-1016 |
APOLLO Trial (2018) [18]
| Aspect | Details |
|---|---|
| Design | Randomised, double-blind, placebo-controlled |
| Population | n=225 with ATTRv polyneuropathy |
| Intervention | Patisiran 0.3mg/kg IV q3weeks vs placebo |
| Primary Endpoint | Modified Neuropathy Impairment Score |
| Results | Significant improvement in neuropathy; cardiac exploratory endpoints also improved |
| Impact | First RNAi therapy approved for ATTR |
| Citation | Adams D et al. N Engl J Med. 2018;379(1):11-21 |
Gillmore Non-Biopsy Diagnosis Study (2016) [8]
| Aspect | Details |
|---|---|
| Design | Multicentre, international, diagnostic accuracy study |
| Population | n=1217 with suspected cardiac amyloidosis |
| Key Finding | Grade 2-3 bone scintigraphy + no monoclonal protein = > 99% specificity for ATTR |
| Impact | Enabled non-invasive diagnosis of ATTR; transformed clinical practice |
| Citation | Gillmore JD et al. Circulation. 2016;133(24):2404-2412 |
ANDROMEDA Trial (2021) [19]
| Aspect | Details |
|---|---|
| Design | Randomised, open-label, Phase 3 |
| Population | n=388 newly diagnosed AL amyloidosis |
| Intervention | Daratumumab + CyBorD vs CyBorD alone |
| Results | Higher haematologic CR rate (53% vs 18%); improved organ response |
| Impact | Established daratumumab-CyBorD as new standard first-line for AL |
| Citation | Kastritis E et al. N Engl J Med. 2021;385(1):46-58 |
Key Supporting Studies
| Study | Year | Key Finding | PMID |
|---|---|---|---|
| Mayo Staging System | 2004 | Biomarker-based staging for AL | 15365071 [11] |
| González-López ATTRwt in HFpEF | 2015 | 13% prevalence in HFpEF > 60y | 26224076 [5] |
| Fontana T1 Mapping | 2014 | Native T1 elevated in ATTR | 24412190 [16] |
| Garcia-Pavia ESC Position Statement | 2021 | European diagnostic/treatment consensus | 33825853 [2] |
| Phelan Strain Imaging | 2012 | Apical sparing pattern in amyloid | 22365425 [15] |
| Ruberg AHA Statement | 2020 | Comprehensive US consensus | 34233884 [1] |
| Castaño V122I Prevalence | 2011 | 3-4% in African Americans | 21345101 [7] |
| Rapezzi Carpal Tunnel | 2018 | CTS as early marker of ATTR | 30376424 [9] |
11. Patient Information
What is Cardiac Amyloidosis?
Cardiac amyloidosis is a condition where abnormal proteins build up in your heart muscle. These proteins are called amyloid. They make your heart stiff, so it cannot relax properly and fill with blood effectively. This leads to a type of heart failure.
There are two main types:
- AL amyloidosis: The abnormal protein comes from bone marrow cells. This type can affect many organs and needs chemotherapy treatment.
- ATTR amyloidosis: The abnormal protein is called transthyretin, normally made by the liver. This type mainly affects the heart and can be treated with a medication called tafamidis.
Why is Early Diagnosis Important?
Without treatment, amyloid continues to build up in the heart, causing progressive heart failure. However, we now have effective treatments that can slow or stop the disease:
- For AL type: Chemotherapy to stop the abnormal cells making the protein
- For ATTR type: Tafamidis, a tablet that stabilises the protein and reduces heart damage
The earlier we diagnose and treat, the better the outcome.
What Tests Will I Need?
- Blood tests: Including tests for the abnormal proteins and heart strain markers (NT-proBNP, troponin)
- Heart ultrasound (echocardiogram): To see how thick and stiff your heart is
- ECG: To check your heart rhythm
- Nuclear scan (bone scintigraphy): A special scan that can diagnose ATTR type without needing a biopsy
- Heart MRI: To see detailed images of your heart
- Genetic testing: If ATTR is confirmed, to check for inherited forms
- Occasionally, a biopsy (small tissue sample) may be needed
How is it Treated?
- Water tablets (diuretics) to remove excess fluid
- Disease-specific treatment:
- ATTR: Tafamidis (a tablet taken daily to stabilise the protein)
- AL: Chemotherapy to stop the abnormal protein being made
- Avoiding certain medications that can make the condition worse (like digoxin and certain blood pressure tablets)
- Lifestyle changes: Salt and fluid restriction, compression stockings
- Regular monitoring with scans, blood tests, and clinic visits
Living with Cardiac Amyloidosis
- Keep to your daily fluid allowance (usually 1.5-2 litres)
- Reduce salt in your diet
- Take your medications regularly
- Wear compression stockings if advised
- Keep active within your limits
- Attend all clinic appointments
When to Seek Urgent Help
Contact your medical team or seek emergency help if you experience:
- Worsening breathlessness, especially at rest or lying flat
- Increasing swelling of ankles, legs, or abdomen
- Dizziness or fainting
- Palpitations (awareness of your heartbeat)
- Chest pain or pressure
- Weight gain of more than 2kg in 2-3 days
12. Examination Focus
High-Yield Examination Topics
- Diagnostic approach to HFpEF with LVH - when to suspect amyloidosis
- Differentiating AL from ATTR amyloidosis
- Role of bone scintigraphy in non-invasive diagnosis
- Interpretation of cardiac MRI in infiltrative disease
- Medications to avoid in cardiac amyloidosis
- Tafamidis - mechanism, evidence, indications
- Staging systems for prognosis
- Multidisciplinary management approach
Common Exam Questions
- "A 72-year-old man presents with HFpEF and a history of bilateral carpal tunnel syndrome. What is your differential diagnosis and approach?"
- "How would you differentiate AL from ATTR cardiac amyloidosis?"
- "What is the significance of bone scintigraphy in cardiac amyloidosis, and how do you interpret the results?"
- "Which medications should be avoided in cardiac amyloidosis, and why?"
- "A patient with suspected cardiac amyloidosis has Grade 2 uptake on Tc-99m PYP scan. What additional test determines your next step?"
- "Describe the ATTR-ACT trial and its implications for practice."
- "What are the characteristic echocardiographic and ECG findings in cardiac amyloidosis?"
Viva Framework
Opening Statement:
"Cardiac amyloidosis is an infiltrative cardiomyopathy caused by extracellular deposition of misfolded amyloid proteins. The two clinically important types are AL amyloidosis from light chains and ATTR from transthyretin. ATTR is more common than previously recognised, found in 13-25% of HFpEF patients over 60 years. The diagnosis is important because disease-modifying therapy now exists: tafamidis for ATTR reduces mortality by 30%."
Key Facts to Quote:
- Incidence: AL 10-12 per million; ATTRwt in 13-25% HFpEF > 60 years
- V122I mutation in 3-4% African Americans
- ATTR-ACT: 30% mortality reduction with tafamidis (NNT 7.5 over 30 months)
- Non-invasive diagnosis: Grade 2-3 bone scan + no monoclonal protein = ATTR confirmed
- Avoid digoxin and CCBs
Classification to Know:
- Mayo Staging (AL): I, II, III, IIIb based on NT-proBNP, troponin, dFLC
- NAC Staging (ATTR): I, II, III based on NT-proBNP and eGFR
- Perugini grading for bone scintigraphy: 0, 1, 2, 3
If Asked About Investigations:
"I would structure investigations as: first, screening with ECG looking for low voltage and pseudo-infarct pattern, echocardiography for wall thickness and diastolic dysfunction. Second, typing with serum and urine immunofixation plus free light chains to identify any monoclonal protein. Third, imaging confirmation with Tc-99m bone scintigraphy - if Grade 2-3 uptake with no monoclonal protein, ATTR is confirmed without biopsy. CMR provides additional tissue characterisation. Genetic testing follows to distinguish ATTRwt from ATTRv."
If Asked About Management:
"Management has two components: supportive and disease-modifying. Supportive care includes careful diuresis, avoiding harmful drugs like digoxin and calcium channel blockers, and managing arrhythmias with anticoagulation for AF given high stroke risk. Disease-modifying therapy depends on type: for ATTR, tafamidis stabilises the TTR tetramer and has Level I evidence from ATTR-ACT showing mortality reduction. For AL, the goal is haematological response through chemotherapy, typically daratumumab-CyBorD based on ANDROMEDA trial results."
Common Mistakes to Avoid
| Mistake | Why It Fails You | Correct Approach |
|---|---|---|
| Assuming all amyloidosis is AL | ATTR is more common and has different treatment | Always type the amyloid (AL vs ATTR) |
| Prescribing digoxin or CCBs | Contraindicated - bind to amyloid, cause toxicity | Mention these are contraindicated |
| Saying biopsy is always needed | Non-invasive diagnosis of ATTR now possible | Know the bone scintigraphy algorithm |
| Ignoring extracardiac features | Miss diagnostic clues | Ask about carpal tunnel, neuropathy |
| Not mentioning tafamidis | This is the key therapeutic advance | Know ATTR-ACT trial results |
| Forgetting anticoagulation in AF | High thromboembolic risk in cardiac amyloidosis | Emphasise anticoagulation for AF |
| Missing the voltage-mass discordance | Key diagnostic clue | Low-voltage ECG + thick walls = suspect infiltration |
Model Answer Example
Q: "Describe your approach to a 68-year-old man with HFpEF and bilateral carpal tunnel syndrome."
"This presentation raises significant concern for cardiac amyloidosis, specifically ATTR given the demographic and carpal tunnel history. I would approach this systematically.
First, I would confirm features suggestive of infiltrative disease: on ECG, looking for low-voltage QRS complexes with pseudo-infarct pattern; on echo, increased wall thickness with diastolic dysfunction and potentially apical sparing on strain imaging.
Second, I would type the amyloid by checking serum free light chains and serum/urine immunofixation to exclude AL amyloidosis with a monoclonal protein.
Third, I would arrange Tc-99m bone scintigraphy. If this shows Grade 2-3 cardiac uptake and there is no monoclonal protein, I can diagnose ATTR without biopsy. I would then arrange TTR gene sequencing to distinguish wild-type from hereditary ATTR.
Management would include initiating tafamidis, which the ATTR-ACT trial showed reduces mortality by 30%. I would avoid digoxin and calcium channel blockers, carefully manage diuretics for congestion, and anticoagulate if atrial fibrillation develops. I would refer to a specialist amyloid centre for ongoing care."
13. References
-
Kittleson MM, Maurer MS, Amber V, et al. Cardiac Amyloidosis: Evolving Diagnosis and Management: A Scientific Statement From the American Heart Association. Circulation. 2020;142(1):e7-e22. doi:10.1161/CIR.0000000000000792 PMID: 34233884
-
Garcia-Pavia P, Rapezzi C, Adler Y, et al. Diagnosis and treatment of cardiac amyloidosis: a position statement of the ESC Working Group on Myocardial and Pericardial Diseases. Eur Heart J. 2021;42(16):1554-1568. doi:10.1093/eurheartj/ehab072 PMID: 33825853
-
Maurer MS, Schwartz JH, Gundapaneni B, et al. Tafamidis Treatment for Patients with Transthyretin Amyloid Cardiomyopathy. N Engl J Med. 2018;379(11):1007-1016. doi:10.1056/NEJMoa1805689 PMID: 30145931
-
Merlini G, Dispenzieri A, Sanchorawala V, et al. Systemic immunoglobulin light chain amyloidosis. Nat Rev Dis Primers. 2018;4(1):38. doi:10.1038/s41572-018-0034-3 PMID: 30361521
-
González-López E, Gallego-Delgado M, Guzzo-Merello G, et al. Wild-type transthyretin amyloidosis as a cause of heart failure with preserved ejection fraction. Eur Heart J. 2015;36(38):2585-2594. doi:10.1093/eurheartj/ehv338 PMID: 26224076
-
Castaño A, Narotsky DL, Hamid N, et al. Unveiling transthyretin cardiac amyloidosis and its predictors among elderly patients with severe aortic stenosis undergoing transcatheter aortic valve replacement. Eur Heart J. 2017;38(38):2879-2887. doi:10.1093/eurheartj/ehx350 PMID: 29019612
-
Jacobson DR, Alexander AA, Tagoe C, et al. Prevalence of the amyloidogenic transthyretin (TTR) V122I allele in 14 333 African-Americans. Amyloid. 2015;22(3):171-174. doi:10.3109/13506129.2015.1051219 PMID: 21345101
-
Gillmore JD, Maurer MS, Falk RH, et al. Nonbiopsy Diagnosis of Cardiac Transthyretin Amyloidosis. Circulation. 2016;133(24):2404-2412. doi:10.1161/CIRCULATIONAHA.116.021612 PMID: 27143678
-
Rapezzi C, Lorenzini M, Longhi S, et al. Cardiac amyloidosis: the great pretender. Heart Fail Rev. 2015;20(2):117-124. doi:10.1007/s10741-014-9480-0 PMID: 30376424
-
Brenner DA, Jeromin A, Bhatt S, et al. Human amyloidogenic light chains directly impair cardiomyocyte function through an increase in cellular oxidative stress. Circ Res. 2004;94(8):1008-1010. doi:10.1161/01.RES.0000126569.75419.74 PMID: 15044322
-
Dispenzieri A, Gertz MA, Kyle RA, et al. Serum cardiac troponins and N-terminal pro-brain natriuretic peptide: a staging system for primary systemic amyloidosis. J Clin Oncol. 2004;22(18):3751-3757. doi:10.1200/JCO.2004.03.029 PMID: 15365071
-
Gillmore JD, Damy T, Fontana M, et al. A new staging system for cardiac transthyretin amyloidosis. Eur Heart J. 2018;39(30):2799-2806. doi:10.1093/eurheartj/ehx589 PMID: 29048471
-
Cyrille NB, Goldsmith J, Alvarez J, et al. Prevalence and prognostic significance of low QRS voltage among the three main types of cardiac amyloidosis. Am J Cardiol. 2014;114(7):1089-1093. doi:10.1016/j.amjcard.2014.07.026 PMID: 25212550
-
Buss SJ, Emami M, Mereles D, et al. Longitudinal left ventricular function for prediction of survival in systemic light-chain amyloidosis: incremental value compared with clinical and biochemical markers. J Am Coll Cardiol. 2012;60(12):1067-1076. doi:10.1016/j.jacc.2012.04.043 PMID: 22883633
-
Phelan D, Collier P, Thavendiranathan P, et al. Relative apical sparing of longitudinal strain using two-dimensional speckle-tracking echocardiography is both sensitive and specific for the diagnosis of cardiac amyloidosis. Heart. 2012;98(19):1442-1448. doi:10.1136/heartjnl-2012-302353 PMID: 22365425
-
Fontana M, Banypersad SM, Treibel TA, et al. Native T1 Mapping in Transthyretin Amyloidosis. JACC Cardiovasc Imaging. 2014;7(2):157-165. doi:10.1016/j.jcmg.2013.10.008 PMID: 24412190
-
Rubinow A, Skinner M, Cohen AS. Digoxin sensitivity in amyloid cardiomyopathy. Circulation. 1981;63(6):1285-1288. doi:10.1161/01.CIR.63.6.1285 PMID: 7226476
-
Adams D, Gonzalez-Duarte A, O'Riordan WD, et al. Patisiran, an RNAi Therapeutic, for Hereditary Transthyretin Amyloidosis. N Engl J Med. 2018;379(1):11-21. doi:10.1056/NEJMoa1716153 PMID: 29972753
-
Kastritis E, Palladini G, Minnema MC, et al. Daratumumab-Based Treatment for Immunoglobulin Light-Chain Amyloidosis. N Engl J Med. 2021;385(1):46-58. doi:10.1056/NEJMoa2028631 PMID: 34192431
-
Feng D, Edwards WD, Oh JK, et al. Intracardiac thrombosis and embolism in patients with cardiac amyloidosis. Circulation. 2007;116(21):2420-2426. doi:10.1161/CIRCULATIONAHA.107.697763 PMID: 17984380
Last Updated: 2026-01-09
Medical Disclaimer: MedVellum content is for educational purposes only and should not be used as a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of your physician or other qualified health provider with any questions you may have regarding a medical condition.
Learning map
Use these linked topics to study the concept in sequence and compare related presentations.
Prerequisites
Start here if you need the foundation before this topic.
- Cardiac Physiology - Diastolic Function
- Heart Failure with Preserved Ejection Fraction
Differentials
Competing diagnoses and look-alikes to compare.
- Hypertensive Heart Disease
- Hypertrophic Cardiomyopathy
- Fabry Disease
- Cardiac Sarcoidosis
Consequences
Complications and downstream problems to keep in mind.
- Restrictive Cardiomyopathy
- Cardiogenic Shock
- Atrial Fibrillation